Final Audit Report - Audit of the Transfer Payment to the Canadian Agency for Drugs and Technologies in Health
March 2013
Management response and action plan
Table of Contents
- Executive summary
- A - Introduction
- 1. Background
- 2. Audit objective
- 3. Audit scope
- 4. Audit approach
- 5. Statement of assurance
- B - Findings, recommendations and management responses
- C - Conclusion
- Appendix A - Lines of enquiry and criteria
- Appendix B - Scorecard
- Appendix C - Core business activities
- Appendix D - Board of directors
Executive summary
The proper management of government funds is an important aspect of grant and contribution oversight. This audit focussed on a conditional grant for $87.5M which was paid over a five-year period ending March 31, 2013 to the Canadian Agency for Drugs and Technologies in Health (the recipient). The recipient has received the funds from Health Canada to provide information and advice to health care system decision makers about the effectiveness and efficiency of drugs, medical devices and other health technologies. As the expiration of the conditional grant approaches, the Strategic Policy Branch will be moving to a five-year contribution agreement. Health Canada's conditional grant accounts for approximately 78 percent of the recipient's funding; provinces and territories account for the remaining 22 percent.
The objectives of the audit are to determine whether the Strategic Policy Branch has put in place an effective management control framework for the oversight and management of the funding agreement and whether the Canadian Agency for Drugs and Technologies in Health has complied with the terms and conditions of the funding agreement. The audit involved the conduct of sufficient and appropriate procedures and the gathering of evidence to support the audit conclusion.
The audit concludes that the Strategic Policy Branch had in place a control framework for the management of the grant. Overall, the grant was managed in a manner that established a reasonable combination of management practices and requirements for performance. However, the Branch recognized the limitation of a grant with regards to its ability to request more extensive monitoring related documentation. As such, the Branch has made a decision to move to a contribution agreement. This strategic decision will allow the Branch to request more fulsome documentation regarding the recipient's activities and related expenditures. Given this new arrangement, it will be important to update the risk profile of the recipient in light of the available tools inherent to a contribution agreement.
Secondly, while there is a control framework, it is important for the Strategic Policy Branch - Office of Pharmaceuticals Management Strategies - to formalize the documentation of the monitoring activity. Documented and systematic monitoring will also assist in providing better evidence of the required oversight for the release of payments.
Regarding the recipient, it has largely complied with the terms and conditions of the funding agreement and uses funding for the intended purposes. The audit found an accounting system in place to track financial transactions, policies and practices on procurement and payables, remuneration, travel, receivables and investments. However, it is recommended that Health Canada include in the contribution agreement the requirement to have the recipient report travel using travel expense claim forms and to adhere more closely to the Treasury Board's Travel Directive.
The audit report includes three recommendations that will serve to improve the controls for the management of the new funding agreement.
A. Introduction
1. Background
The Canadian Agency for Drugs and Technologies in Health (CADTH/the recipient), established in 1989 by Canada's Ministers of Health, is an independent not-for-profit organization funded by the federal, provincial and territorial governments of Canada. CADTH is one of Canada's leading sources of information and advice on the effectiveness and efficiency of drugs, medical devices and other health technologies.
CADTH's mandate is to deliver reliable, timely, evidence-based information to Canada's health care leaders on the effectiveness and efficiency of health technologies. Products and services available through CADTH are used by decision-makers within the ministries of health to support decisions on which medical devices and drugs to adopt and use. Such decisions are important to the quality, affordability and sustainability of health care in Canada while getting the best value from every health care dollar.
CADTH also plays a key collaborative role with Health Canada's Health Products and Food Branch by assisting in streamlining decisions for faster market access of health products and in exploring potential new joint activities (for example, accelerating assessments for health products for unmet medical needs; addressing the potential issue of antimicrobial resistance; reinforcing benefit-risk balance assessment; considering the usefulness or possibility of developing surveys to monitor the outcome of scientific review processes for health products; and improving the decision-making process by taking into account patient experience).
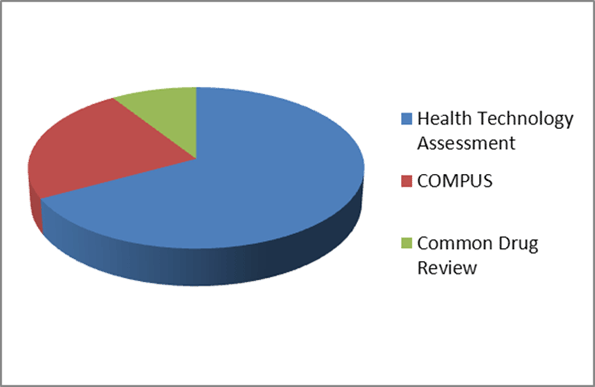
The conditional grant to CADTH is $87.5M over five years and is being renewed through a contribution agreement after March 31, 2013. Typically CADTH receives approximately $16M annually for funding towards three programs: Health Technology Assessment ($11M), Canadian Optimal Medication Prescribing and Utilization Service (COMPUS) ($4M - funded solely by Health Canada) and Common Drug Review ($1.5M). In addition, support towards the medical isotope project amounted to $3M over two years: 2010-11 and 2011-12. This project has been completed. Health Canada accounts for approximately 78 percent of CADTH's total funding while the provinces and territories account for the remaining 22 percent. (See Appendix C)
CADTH has a board of directors composed of an independent chair, a regional distribution of jurisdictional representatives and a number of non-jurisdictional representatives from health authorities, academia and the general public. Directors are elected by the Members of the Corporation who are the Deputy Ministers of Health from participating federal, provincial and territorial governments. The Board has overall responsibility for administering the affairs of the Corporation and for providing the strategic direction to guide CADTH's success as the Canadian "go to" provider of evidence and advice on the use of drugs and other health technologies (See Appendix D).
CADTH is accountable to Health Canada for the use of the federal assistance in accordance with the terms and conditions of the funding agreement. As such, the Strategic Policy Branch's Office of Pharmaceuticals Management Strategies is responsible for managing the funding agreement.
2. Audit objective
The objectives of the audit were to determine whether the Strategic Policy Branch (SPB) has put in place an effective management control framework for the oversight and management of the funding agreement; and whether CADTH has complied with the terms and conditions of the funding agreement.
3. Audit scope
The audit focused on SPB's management control framework for monitoring and administering the transfer payments to CADTH as well as CADTH's compliance to the funding agreement and financial management internal controls, specifically:
- SPB's controls surrounding the monitoring and the financial recording of the payments made according to the funding agreement;
- the financial management requirements of the recipient surrounding the administration of funds and the monitoring of expenditures; and
- the eligibility of expenses reported by the recipient for the two fiscal years ending March 31, 2011 and 2012, and the first quarter of 2012-13, and compliance with the terms and conditions of the funding agreement.
4. Audit approach
The audit methodology consisted of interviews, the examination of evidence supporting the internal control processes, the presentation of financial information for decision-making, an examination of the departmental management control framework; and an examination of the recipient's accounting system; internal control practices, costs eligibility (salary and benefit expenses, contracts, travel costs, and administration costs); financial reporting requirements (such as audited financial statements) and compliance to other terms and conditions of the funding agreement.
Work was performed at Health Canada's Strategic Policy Branch in Ottawa and at CADTH's premises in Ottawa, Ontario in November 2012.
5. Statement of assurance
In the professional judgment of the Chief Audit Executive, sufficient and appropriate procedures were performed and evidence gathered to support the accuracy of the audit conclusion. The audit findings and conclusion are based on a comparison of the conditions that existed as of the date of the audit, against established criteria that were agreed upon with management. Further, the evidence was gathered in accordance with the Internal Auditing Standards for the Government of Canada and the International Standards for the Professional Practice of Internal Auditing.
B. Findings, recommendations and management responses
1 Effective management control framework
1.1 Terms and conditions of the funding agreement
Audit criterion: The transfer payment complies with the funding proposal and the Directive on Transfer Payments (October 2008).
Funding proposal
A 2004 funding proposal established the named grant. In 2008, a second pan-Canadian funding proposal sought the authority to fund a few organizations encompassed under the Health Care Policy Contribution Program. While the recipient was referenced in that proposal, there was no requirement to seek Treasury Board approval for renewal of this funding. As such, an internal process was followed and signed by the Minister in March 2008 to renew the transfer payment through a conditional grant for $87.5M over a five-year period. The audit team examined: the 2004 and 2008 funding proposals; the documentation related to the internal approval; as well as two subsequent July 2011 funding agreement amendments (one for the inclusion of a special research project on isotopes project and a second administrative amendment). The audit concludes that there were no omissions or discrepancies between the various funding proposals and the transfer payment.
Policy and directive on transfer payments
On October 1, 2008, the Treasury Board (TB) implemented the Policy on Transfer Payments. The Secretariat noted that the funding agreements entered into before this date, would remain in effect until their expiry date. This is the situation for the transfer payment agreement between Health Canada and the recipient, which was signed five months prior to the new policy. While the grant was not subject to the updated policy, a comparison was performed between the Treasury Board policy, directive and the funding agreement in order to identify opportunities for improvements for the upcoming funding renewal.
Whereas the grant already has several conditions embedded, the audit analysis between the policy and the grant did note that it would be important for the Directorate to add the Minister's right to undertake a recipient audit (to provide compliance assurance) and to add the Minister's right of access to the recipient's documents and premises for the purpose of conducting an audit or monitoring compliance with the funding agreement. The Directorate has confirmed that these "rights" will form part of the new funding arrangement.
1.2 Monitoring activities and risk management
Audit criterion: The Strategic Policy Branch has a formal and rigorous approach to monitor the recipient's activities and manage risk.
The Directive on Transfer Payments allows departments to develop a measured response to risk throughout the management cycle of transfer payments. A risk-based approach should be applied when setting the funding agreement requirements related to cash management practices, reporting and monitoring.
In regard to the risk-based approach to recipient monitoring, the Directorate has implemented the Enterprise Risk Management - Agreement/Recipient Risk Assessment Tool (ERM-ARRAT) as of April 2010. The ERM-ARRAT tool is part of the Health Portfolio's framework for grants and contributions. The tool was developed as a result of the Policy on Transfer Payments, which aims to reduce the administrative burden on recipients by adopting monitoring, reporting and audit requirements that are proportionate to risk. In 2010-11, through the ERM-ARRAT instrument, the Directorate determined that the overall risk rating for the recipient was low. While the ERM-ARRAT is one tool for assessing risk, there are other elements found in the terms and conditions of the funding agreement that are not captured in the ERM-ARRAT exercise, such as privacy, lobbying, recognition of federal funding and official languages. Since the Directorate is moving to a new funding arrangement, and the fact that the risk profile for the recipient was established some time ago, there is an opportunity to refresh the risk assessment in support of the new contribution agreement.
Recommendation 1
It is recommended that the Assistant Deputy Minister, Strategic Policy Branch update the risk assessment for the recipient.
Management response
Management agrees with the recommendation.
The Office of Pharmaceuticals Management Strategies will update the Enterprise Risk Management - Agreement/Recipient Risk Assessment Tool assessment and will develop best practices for setting up risk assessment criteria and monitoring for organizations such as the Canadian Agency for Drugs and Technologies in Health, as well as develop a strategy to leverage existing best practices towards consistent and effective management of transfer payments across the Branch.
For monitoring purposes, the conditional grant required a cash flow schedule, an annual work plan (business plan), an annual progress report, financial statements and an independent evaluation report. For the period under review, it was noted that the financial reports received were not being referred to the Branch Senior Financial Officer for analysis until fall 2012. As a best practice, financial reports (audited financial statements, quarterly cash flow, business plans) should be referred to the Branch Senior Financial Officer for expert financial analysis. It would be important for the Directorate to systematically track reception of these key deliverables and forward them for analysis in support of its oversight role. Involving the Branch Senior Financial Officer will strengthen the financial management and accountability practices within the Department, provide for prudent management of resources in pursuit of departmental priorities, and will add an important level of accountability to the Directorate's oversight framework.
Beyond the funding agreement's reporting requirements, the Strategic Policy Branch's Office of Pharmaceuticals Management Strategies (OPMS) and other organizations within the Health Portfolio have been monitoring the activities of the recipient via committees. The Department's overall rationale for Board membership relates to: advancing federalism; strengthening health policy; and improved accountability. Federal involvement in entities like the Canadian Agency for Drugs and Technologies in Health (CADTH) is an expression of the federal interest and commitment to advance the health of Canadians. Board and committee memberships also give the opportunity of:
- liaising with the recipient's officers and Board members at meetings;
- influencing strategic decisions (for example, annual budget and strategic plan of the organization);
- enhancing the awareness of risks facing the recipient;
- ensuring that the recipient's activities fall within its corporate objectives; and
- monitoring and assessing the performance of key staff.
Currently, Health Canada's Deputy Minister is a member of the Conference of Deputy Ministers (the "owners" of the Corporation); the Assistant Deputy Minister is a member of CADTH's Board of Directors; the OPMS' Executive Director is a member of CADTH's Exchange Committee while another director sits on CADTH's Drug Policy Advisory Committee. In addition, four other managers from the Health Portfolio are members of CADTH's committees.
Oversight Framework Documents
- Description of the accountability provisions in the funding agreement;
- Annual cycle of deliverables;
- Accountability monitoring schedule (discretionary/time-limited activities);
- Ongoing due diligence activities;
- General obligations as articulated in the funding agreement
Through an examination of correspondence, it is evident that the Directorate has been monitoring via a working relationship with officials and committee involvement. Management notes that it has been receiving, reviewing and commenting on strategic documents. However, the Directorate would benefit from developing a documented risk-based oversight framework, including the assignment of oversight roles and responsibilities, the description of the applicable monitoring mechanisms to be employed, and the determination of the funding agreement conditions to be subjected to compliance oversight.
Recommendation 2
It is recommended that the Assistant Deputy Minister, Strategic Policy Branch document the approach to monitor the recipient's activities in order to effectively manage risk.
Management response
Management agrees with the recommendation.
While the Strategic Policy Branch has kept up with its monitoring duties, the Directorate recognizes that better documentation of such duties could be improved. In an effort to leverage from other established practices, the Directorate will work with other directorates to leverage suitable practices towards more effeciently and effectively managing transfer payments across the Branch.
1.3 Approval of payments
Audit criterion: Financial and non-financial reporting of the recipient is reviewed and approved in support of payments.
As a condition for payments, the recipient must meet the financial and non-financial reporting requirements specified in the funding agreement. The required reports in the grant include: an annual work plan (business plan), an annual progress report, a cash flow schedule and financial statements. Additionally, throughout the lifespan of the grant, further reporting requirements included an independent evaluation report, a strategic plan, a project plan, records of expenditures, and statements of work.
Although reporting documents were received, it was difficult to know if they were fully analysed by the Program prior to the release of payments. The Branch would benefit from using monitoring tools (such as checklists) in order to demonstrate that a thorough and systematic review is conducted prior to the release of payment. (See recommendation 2)
1.4 Performance measurement
Audit criterion: The Strategic Policy Branch management ensures that the recipient has conducted an evaluation of its work, with a directive to assess the implementation of the recommendations therein.
In year two of the agreement, the Conference of Deputy Ministers recommended an independent study to determine what alternative business and financing models might be considered for CADTH to best meet the needs of jurisdictions. This report made several recommendations starting with governance and the composition of the Board of Directors and CADTH's committees. This report has triggered some restructuring at CADTH in 2010 and will also have an impact on the next funding agreement.
The original funding agreement (signed in March 2008) required an independent evaluation to be completed by the recipient in 2011. For this evaluation to be objective, the Directorate worked in collaboration with the recipient to develop the statement of work to select the firm and orientate the work of the evaluation. As is often the case, this evaluation was performed in two phases. Phase 1 was delivered in December 2011 and contained 23 recommendations. In addition, an amending agreement was signed in January 2011 to fund the Isotope Project. The Strategic Policy Branch (SPB) required an evaluation of this project as well. A second amendment to the funding agreement extended the time limit for the delivery of Phase 2 of the evaluation to October 2012. The evaluations were performed as per the agreement requirements.
The recipient prepared a Management Action Plan in response to the 23 recommendations of the Phase 1 evaluation. The Phase 2 report, released at the time of the audit, indicates substantive progress on those recommendations. All evaluations and the Phase I Evaluation Management Response are posted on CADTH's website in compliance with the requirement of the funding agreement to make them public.
2. Terms and conditions of the funding agreements
2.1 Controls and management practices
Audit criterion: The recipient's internal controls and management practices are in compliance with the terms and conditions of the funding agreement and support due regard for economy and efficiency in spending decisions.
Good internal controls and management practices reduce financial risk. To that end, the audit examined the existence of policies and practices on procurement and payables, remuneration, travel, receivables and investments.
The Finance and Administration Department is responsible for the maintenance of internal controls for CADTH. This includes the monitoring of compliance of signing limits, the provision of guidance on signing authority questions and the training of individuals in the organization on signing authority and procurement.
The CADTH Policy for Signing and Commitment Authority (approved in April 2008) is extensive and applies to all expense categories for various levels. Only the President and executive staff are authorized to sign contracts and standing offers within the signing limits outlined in the Delegation of Financial Signing Authority matrix. The capital budget is established and approved by the Board annually.
The Delegation of Financial Signing Authority Matrix contains an extensive list of decisions and authority levels:
- Supplies and services, web site, meeting and special events expenses, the symposium, travel expenses, honoraria, corporate credit cards
- Human resources: training and development
- Fixed assets
- Investment of funds
- Other decisions such as the approval of the budget and of the audited Financial Statements by the Board of directors
The use of sole source (or single source) suppliers can only be used under limited circumstances for non-scientific contractors and where the purchase amount is over $5,000. The responsible member of the executive team and the Director, Finance and Administration must provide approval prior to a sole or single source commitment being made. Such contracts cannot be amended for any reason without the approval of the President & Chief Executive Officer. Purchase Orders are used to record expense commitments and reductions in commitments when payments are made.
Remuneration procedures cover the roles and responsibilities for the hiring, the establishment of pay scales, promotions, demotions or termination of employees, as well as other payroll changes. Payroll processing, the calculations and remittance of pay deductions, calculations and tracking of taxable benefits, and the preparation and submission of required tax forms are outsourced to a private firm. Changes to payroll runs are inputted by CADTH and reviewed after each pay.
The current travel policy has been in force since January 1, 2011 and replaces the one adopted in February 2007. This policy applies to staff and non-staff authorized to travel. The procedures require the pre-approval of staff travel by immediate supervisors while non-staff have their travel arranged through an administrative coordinator.
Currently, airfare, train travel and hotels are arranged through a travel agent that bills the recipient on a monthly basis. Expenses are also charged on corporate credit cards which are billed to CADTH separately. Travellers who use cash advances generally account separately for the use of their advance. Consequently, expenses for any given trip can be reported through three different sources, making the task of verifying the aspects of the travel claim challenging as well as inefficient to determine the total cost of each trip. One area that would assist the recipient would be to implement a travel expenses process so that each trip would be recorded onto a single travel expense claim.
Recommendation 3
It is recommended that the Assistant Deputy Minister, Strategic Policy Branch include in the contribution agreement wording to address travel procedures, rates and allowances.
Management response
Management agrees with the recommendation.
The Strategic Policy Branch - Office of Pharmaceuticals Management Strategies - will include wording in the contribution agreement to addresss travel procedures, rates and allowances.
2.2 Use of funds
Audit criterion: The recipient uses funds as per the terms and conditions and for the purposes intended in the funding agreement.
Much of the recipient's research is performed in-house. However, 40 percent of expenditures are for payments made to third parties (researchers and service providers). Approximately $3.1M of the recipient's expenditures are for professional service providers, researchers and other suppliers of goods and services, such communication and publications, computers and web maintenance. Advisory committees and annual symposia require some travel, as experts live across Canada. Therefore, the procurement, remuneration and travel transactions were tested. The audit also examined investments and some receivables.
A total of 29 transactions were selected, based on risk and materiality, from expenses incurred during fiscal years 2010-11, 2011-12 and the first quarter of 2012-13 under these categories: procurement, compensations and benefits, travel and other expenses.
The transaction testing demonstrated that the expenses claimed were supported, approved at the right level and charged to the correct program. Funding was used for the purpose of carrying out activities covered in the funding agreement.
2.3 Reporting requirements
Audit criterion: The recipient maintains its financial records and provides reports in accordance with the funding agreement, namely: a strategic plan, a project plan, an annual work plan, a progress report, a schedule of monthly payments, an independent evaluation, audited financial statetements prepared according to Generally Accepted Accounting Principles, as well as other reporting requirements.
The recipient must submit relevant information on its activities on an annual basis through its annual work plan (business plan), annual progress report, and via its financial statements. These documents, in addition to a two-phase independent evaluation and a strategic plan, represent the majority of the recipient's reporting requirements and are essential tools for OPMS to determine whether the recipient has used federal funding for the purposes set out in the funding agreement.
Further to the reporting requirements stated above, the conditional grant requirements also include cash flow schedules, records of expenditures, value-for-money reporting, statements of work and a project plan.
Strategic plan
The funding agreement stipulates that a strategic plan must be developed and submitted to the Conference of Deputy Ministers for approval, on or before December 31, 2010 and covering fiscal years 2011-13. The strategic plan was submitted and describes how the recipient intends to carry out its programs in order to achieve its mandate. As well, the strategic plan details an action plan to address the recommendations from the independent evaluation, and a strategy to achieve the objectives of the funding agreement.
Project plan
The project plan describes how the recipient intends to carry out its activities related to the project (Optimizing Health System Efficiency Initiative) in order to achieve its mandate through fiscal years 2010-12.
The project plan was delivered to the CADTH Board of Directors for approval on June 28, 2010 and received final approval. Therefore, this condition was met.
Annual work plan
The annual work plan and a proposed schedule of monthly payments must be provided for the Minister's review no later than February 28th for the following fiscal year. The annual work plan focuses on the recipient's budget, strategic objectives and related activities, outputs and intended outcomes for the fiscal year. Therefore, the recipient has complied with its obligations to provide the annual work plan and schedule of cash flow requirements to the Department.
Progress report
The funding agreement stipulates that the progress report must be submitted to the Minister, on or before October 1 in the subsequent fiscal year. The progress report is a report on performance which includes the recipient's objectives for a given fiscal year and the extent to which those objectives were met.
The progress reports contained an assessment of value-for-money, outcomes achieved, an overview of the significant expected results to be achieved in the upcoming fiscal year and were submitted to the Minister. Therefore, the recipient complied with this condition.
Schedule of monthly payments
The schedule of monthly payments was required in February of each fiscal year, to allow the Directorate to forecast monthly payments to the recipient in the following fiscal year. The recipient has developed and submitted to the Minister the schedule of monthly payments as required in the funding agreement. Therefore, the condition was met.
Independent evaluations
The funding agreement requires that an independent evaluation be carried out of the core business activities for the period of April 1, 2007 to January 31, 2012 and of the project's activities for the period of January 1, 2011 to March 31, 2012. Section 1.4 of this report addresses the topic of evaluations and their impact. The evaluations were carried out in compliance with the funding agreement.
Annual financial statements
The recipient is expected to properly and fully maintain its accounts, essential records, and records of transactions in relation to the funding received, in accordance with the Generally Accepted Accounting Principles applicable in Canada. Financial statements for fiscal year 2010-11 and fiscal year 2011-12 were audited by external auditors. Auditor reports for both fiscal years indicated that the accounting principles had been applied on a basis consistent with that of the preceding year and were in accordance with Generally Accepted Accounting Principles.
Other reporting requirements
The conditional grant requirements also included value-for-money reporting and statements of work. In addition, quarterly cash flow forecasts and records of expenditures have been required since the first amendment to the funding agreement (January 2011). However, as reported in section 1.2, they were requested by the Strategic Policy Branch in the fall of 2012.
Value-for-money requirements relate to the reporting of the recipient's activities and cost effectiveness and impact on the health system. The audit found that the recipient has developed, in conjunction with Health Canada, methods for reporting on the value-for-money of its activities.
Also, in conjunction with Health Canada, the recipient's developed statements of work, as outlined in the funding agreement, towards the evaluation and submitted them to the Minister.
2.4 Communication requirements
Audit criterion: The recipient's communication activities are in accordance with the terms and conditions of the funding agreement namely: the acknowledgement of financial contributions, official languages, and the protection of personal information.
Acknowledgement of funding
The recipient's Publishing Services group is responsible for ensuring that all relevant publications contain the information as required in the funding agreement. The mandatory statements such as the acknowledgement of funding and a disclaimer appears on the material developed for public distribution, including reports, project briefs, symposium programs, media releases, communication material, brochures, as well as being stated on the website.
The recipient has acknowledged the financial contributions received by Health Canada as per the terms and conditions of the funding agreement.
Official languages
To ensure a consistent approach for the translation of documents, the recipient's Publishing Services group issued a translation policy. The policy provides a product listing of what will and will not be translated and a rationale when translation is not provided. It was noted that the 2010-11 Annual Report was posted in English only on the recipient's website. In addition, on French pages, there are instances where links to the web sites of organizations that provided French versions led to the English versions or were not functioning. With the exception of the above noted observation, the recipient provides oral and written communications to the general public, as well as an active offering of its services to members of the public in both official languages of Canada.
Protection of personal information
Given the sensitivity of personal health information, the recipient was required by the funding agreement to incorporate the protection of personal information in its activities in accordance with applicable federal, provincial and territorial laws. The recipient has procedural documentation that deals with the handling of confidential and personal information, but since it deals with economic models with de-personalized data and 'proof of concept' information, personal or private information issues should not pose a serious risk.
As per the current terms and conditions, the recipient's protection of personal information complies with the terms and conditions of the funding agreement. However, the Directorate could benefit from strengthening the terms and conditions related to protection of personal information in the upcoming contribution agreement.
2.5 Legislation and policies
Audit criterion: The recipient complies with the federal legislation and policies stipulated in the agreements namely: conflict of interest, lobbying, dispute resolution, intellectual property, governing law, indemnification and limitation of liability, default, and general provisions.
Conflict of interest
The recipient has developed a Conflict of Interest Policy that has been in effect since 2008 and which applies to all permanent and term employees. It has also developed a conflict of interest procedure document that outlines the steps to be followed by the recipient regarding conflict of interest. In addition, at the time of hire, all employees are required to submit a conflict of interest declaration.
Further, a Conflict Of Interest Policy exists for Expert Committee and Panel Members. It requires that all members, as an annual practice, sign a declaration of conflicts. The recipient's Board of Director meetings also involve a declaration of potential conflict of interest process.
The recipient, however, has not made the Conflict of Interest Policy available to the public, as per the requirements of the funding agreement. This could be done best via the recipient's website. Additionally, moving forward towards the contribution agreement, it is advisable that the Directorate include within the terms and conditions, a clause related to the Conflict of Interest and Post-Employment Code for Public Office Holders, which is present in other funding agreements.
In conclusion, in spite of the above, the recipient has complied with the terms and conditions stated in the funding agreement related to conflict of interest.
Lobbying
The recipient follows the guidelines as set out in the funding agreement on lobbying and does not engage lobbyists. It has complied with the terms and conditions stated in the funding agreement related to lobbying.
Dispute resolution
Though there has not been any need for use of the dispute resolution process outlined within the funding agreement, the recipient has reported, in interviews, a willingness and desire to pursue resolution through negotiations or other appropriate dispute resolution procedures rather than to resort to litigation. The recipient has shown a willingness to comply with the dispute resolution provisions, should they be necessary, as per the terms and conditions of the funding agreement.
Intellectual property
Intellectual property is understood to be any new research findings that can be directed to any use and includes copyrighted material, patents, trade-marks, trade names, industrial designs and trade secrets. The current clause stipulates that any material produced by the recipient, while carrying out the activities under the funding agreement, or that result from the payment of the grant, remain the property of recipient, unless otherwise agreed to, per the funding agreement. Going forward the Directorate should review Health Canada's Intellectual Property Policy (May 2011) for alignment in the new contribution agreement. The recipient has complied with the terms and conditions stated in the funding agreement related to intellectual property.
Governing law
The recipient is incorporated under the Canada Corporations Act as a not-for-profit organization and retains active status with annual filings. The recipient governs its funding agreement by, and in accordance with the laws of the Province of Ontario and the laws of Canada.
Indemnification and limitation of liability
Clause 13 of the agreement describes the conditions for which the recipient shall indemnify and hold harmless Her Majesty the Queen in right of Canada from and against all claims, losses, and related damages attributable to or arising from any willful or negligent act in carrying out activities in relation to the funding agreement. As no such situations are known to have occurred, there was no audit procedure related to this section.
Default
Clause 14 of the agreement describes events that constitute a default, such as failure to perform and the consequences or rights of the Minister in such cases. As there are no known situations of defaults, there is no audit procedures related to that section.
General provisions
The general provisions deal with the obligations of the parties should the agreement expire or be terminated for any reason. Other provisions exclude the right to amend the agreement without the consent of the other party, the limits of the scope of the agreement to the parties and the non-establishment of principal and agent relationships. At the time of the « audit, there has been no such situations. In conclusion, the recipient complies with legislation, policies and other requirements stipulated in the funding agreement, including conflict of interest, registration of lobbyists, dispute resolution, intellectual property, governing law, indemnification and limitation of liability, default and general provisions.
Conclusion
The Strategic Policy Branch has in place a management control framework for the management of the grant. Overall, the grant was managed in a manner that established a reasonable combination of management practices and requirements for performance. The management of the grant has considered risks in the past. However, it will be important going forward to update the risk information related to the recipient. The updated risk information should be used to inform the new contribution agreement so that an appropriate balance between control and flexibility is established.
While there is a control framework, it is important for the Strategic Policy Branch - Office of Pharmaceuticals Management Strategies - to improve the overall effectiveness of the framework by documenting its monitoring framework and activity. Systematic documentation of the monitoring activity will also assist in providing better evidence of oversight required for the release of payments.
Regarding the recipient, it has largely complied with the terms and conditions of the funding agreement and uses funding for the intended purposes. The audit found an accounting system in place to track financial transactions, policies and practices on procurement and payables, remuneration, travel, receivables and investments. However, it is recommended that Health Canada include in the contribution agreement the requirement to have the recipient report travel using travel expense claim forms and to adhere more closely to the Treasury Board's Travel Directive.
The report includes three recommendations that will serve to improve the controls for the management of the contribution agreement that will be in force in April 2013.
Appendix A - Lines of enquiry and criteria
| Criteria Title | Audit Criteria | |
|---|---|---|
| The Strategic Policy Branch has put in place an effective management control framework for the management of the funding agreement. | ||
| 1.1 | Terms and conditions of the funding agreement | The transfer payment complies with the funding proposal and the Directive on Transfer Payments (October 2008). |
| 1.2 | Monitoring activities and risk management | The Strategic Policy Branch has a formal and rigorous approach to monitor the recipient's activities and manage risk. |
| 1.3 | Approval of payments | Financial and non-financial reporting of the recipient is reviewed and approved in support of payments. |
| 1.4 | Performance measurement | The Strategic Policy Branch management ensures that the recipient has conducted an evaluation of its work, and assess the implementation of recommendations therein. |
| The recipient complies with the terms and conditions of the funding agreement. | ||
| 2.1 | Controls and management practices | The recipient's controls and management practices are in compliance with the terms and conditions of the funding agreement, and support due regard for economy and efficiency in spending decisions. |
| 2.2 | Use of funds | The recipient uses funds as per the terms and conditions and for the purposes intended in the funding agreement. |
| 2.3 | Reporting requirements | The recipient maintains its financial records and provides reports in accordance with the funding agreement, namely: a strategic plan, a project plan, an annual work plan, a progress report, a schedule of monthly payments, an independent evaluation, audited financial statetements prepared according to Generally Accepted Accounting Principles, as well as other reporting requirements. |
| 2.4 | Communication requirements | The recipient's communication activities are in accordance with the terms and conditions of the funding agreement, namely: the acknowledgement of financial contributions, official languages, and the protection of personal information. |
| 2.5 | Legislation and policies | The recipient complies with the federal legislation and policies stipulated in the agreements namely: conflict of interest, lobbying, dispute resolution, intellectual property, governing law, indemnification and limitation of liability, default and general provisions. |
Appendix B - Scorecard
| Criterion | Rating | Conclusion | Rec # |
|---|---|---|---|
| The Strategic Policy Branch has put in place an effective management control framework for the management of the funding agreement. | |||
| 1.1 Terms and conditions of the funding agreement | Table 2 footnote NMI | The terms and conditions of the funding agreement are in line with the Treasury Board submissions; however, the next funding agreement should clarify the Minister's right to audit the recipient. | |
| 1.2 Monitoring activities and risk management | Table 2 footnote NMO | There is monitoring at the policy level; however, a formal risk-based approach to monitor the recipient's activities should be developed and monitoring activities should be documented. | 1 |
| 1.3 Approval of payments | Table 2 footnote NMO | Reporting by the recipient is reviewed and approved; however, more documentation that conditions are met before payment are recommended. | 2 |
| 1.4 Performance measurement | Table 2 footnote S | The Strategic Policy Branch ensures that evaluations are conducted and recommendations are followed. | |
| The recipient complies with the terms and conditions of the funding agreement. | |||
| 2.1 Controls and management practices | Table 2 footnote NMO | The recipient has controls and management practices in place to ensure good stewardship; however, travel expenses require stronger controls. | 3 |
| 2.2 Use of funds | Table 2 footnote NMI | The recipient uses funds as per the terms and conditions and for the purpose intended in the funding agreement. | |
| 2.3 Reporting requirements | Table 2 footnote S | The recipient maintains its financial records and provides reports in accordance with the terms and conditions of the funding agreement. | |
| 2.4 Communication requirements | Table 2 footnote NMI | Communication activities comply with the terms and conditions of the funding agreement. Some minor improvements to official languages were noted. | |
| 2.5 Legislation and policies | Table 2 footnote NMI | The recipient has complied with the following conditions: conflict of interest, lobbying, dispute resolution, intellectual property, governing law, indemnification and limitation of liability, default, and general provisions; however, some improvements are suggested on the next funding agreement. | |
Table 1 footnotes
|
|||
Appendix C - Core business activities
Common Drug Review
(Health Canada contributes 30 percent of the total funding)
The Common Drug Review (CDR) is a single process for undertaking reviews and providing listing recommendations - for new drugs (new chemical entities and new combination products) and new indications for previously approved CDR drugs - to participating federal, provincial and territorial drug benefit plans in Canada. The goal of the CDR is to objectively assess the clinical and pharmacoeconomic evidence for the newest drug therapies and newest therapies for new indications with old drugs coming to the Canadian market. All jurisdictions are participating except Quebec.
New chemical entities or new combination drug products (new drugs), first go through the Common Drug Review (CDR) process, housed at the Canadian Agency for Drugs and Technologies in Health (CADTH). CADTH is a national body that provides Canada's federal, provincial and territorial health care decisions with credible, impartial advice and evidence-based information about the effectiveness and efficiency of drugs and other health technologies. As part of the CDR process, the Canadian Expert Drug Advisory Committee (CEDAC), an independent advisory body composed of individuals with expertise in drug therapy and drug evaluation, makes recommendations to each of the participating federal, provincial, and territorial publicly funded drug plans regarding the listings on their formularies.
The formulary serves as a :
- guide to practitioners and pharmacists regarding drug products which are eligible for coverage under benefit plans;
- guide to professional committees in hospitals and institutions in the selection of drug products;
- guide to drug product interchangeability in respect of drug products that have been designated interchangeable under Ontario legislation; and
- comparative pricing guide for drug products.
Health Technology Assessments
(Health Canada contributes 90 percent of total funding)
A health technology assessment is a "multidisciplinary" activity that systematically examines the technical performance, safety, clinical efficacy and effectiveness, cost, cost-effectiveness, organisational implications, social consequences, legal and ethical considerations of the application of a health technology. The Health Technology Assessments support and inform effective, evidence-based decisions about health policy and purchasing, service management and clinical practice. Topics are selected based on criteria of national interest to the publicly funded health care system. These reports are comprehensive assessments of health care technologies, including drugs, devices, systems and services. These reports usually examine more mature technologies for which there is a larger and higher quality body of evidence available. The technologies' clinical effectiveness and cost-effectiveness and broader impact on both patient health and the health care system are examined. This may include information on the social, ethical and economic implications of their uses compared with alternative treatments, and information on patient indications, current treatment practices, regulatory issues, adverse effects and health services impact.
Canadian Optimal Medication Prescribing and Utilization Service
(Health Canada is the sole provider of funding)
The Canadian Optimal Medication Prescribing and Utilization Service (COMPUS) is a nationally coordinated program which provides a service to federal, provincial and territorial jurdiscitions. The goal of COMPUS is to optimize drug-related health outcomes and cost-effective use of drugs by identifying and promoting optimal drug prescribing and use. Where possible, COMPUS builds on existing applicable Canadian and international initiatives and research.
Isotopes project
(Federal participation: 100 percent of total activity costs)
Since 2007, the demand for medical isotopes has been critical and shortages could impose major challenges to the nuclear medicine community. The medical isotope at the centre of this global crisis is technetium-99m (99mTc), which is made from molybdenum. This isotope is used to diagnose and detect a number of conditions, including cancer and heart disease. Health Canada funded a study by CADTH to identify new and emerging technologies that may play a role in providing either solutions to the supply issue or alternate technologies that circumvent the need for nuclear reactor-based molybdenum-99 over the next five to 15 years.
Appendix D - Board of directors
Dr. Terrence Sullivan
Chair of the Board
Federal
Ms. Abby Hoffman
Assistant Deputy Minister, Strategic Policy Branch, Health Canada
Western provinces
Ms. Bernadette Preun
Assistant Deputy Minister of Provincial Programs and Services, Manitoba Health
Ms. Susan Williams
Assistant Deputy Minister, Health Policy and Service Standards Division of Alberta Health and Wellness
Territories and Nunavut
Ms. Sherri Wright
Assistant Deputy Minister of Health Services, Government of Yukon
Ontario
Ms. Diane McArthur
Assistant Deputy Minister and Executive Officer of Ontario Public Drug Programs
Atlantic provinces
Dr. Catherine Bradbury
Associate Deputy Minister, Department of Health and Community Services, Newfoundland and Labrador
Ms. Paula English
Chief, Program Standards and Quality, Nova Scotia Department of Health and Wellness
Health Authorities
Dr. Robert Halpenny
President & CEO, Interior Health, Kelowna, British Columbia
Dr. Édouard Hendriks
Vice-President, Medical and Academic Affairs, Horizon Health Network, New Brunswick
Academic
Dr. Tony Culyer
Ontario Research Chair in Health Policy and System Design, University of Toronto
Public
Ms. Sally Aileen Brown
Former CEO, Heart and Stroke Foundation of Canada
Dr. John M. Horne
Adjunct Professor, Royal Roads University, Victoria, British Columbia
Observer
Dr. Juan Roberto Iglesias
President and Chief Executive Officer, INESSS, Quebec
Source: Canadian Agency for Drugs and Technologies in Health
Page details
- Date modified: