Final Report - Audit of the Management of the Pesticide Regulatory Program - March 2015
Table of Contents
- Executive summary
- A - Introduction
- B - Findings, recommendations and management responses
- C - Conclusion
- Appendix A - Lines of enquiry and criteria
- Appendix B - Scorecard
- Appendix C - Estimated resources
- Appendix D - Submission categories
- Appendix E - Compliance and enforcement statistics
Executive summary
The focus of the audit was on the management control framework to support the Pesticide Regulatory Program. Health Canada's Pest Management Regulatory Agency (PMRA) was created in 1995 and received approximately $47 million in 2013-14 for the regulation of pest control products under the Pest Control Products Act. In the administration of this Act, the primary objective is to prevent unacceptable risks to people and the environment from the use of pest control products. The Act controls the import, sale, manufacture and use of pesticides. There are approximately 7,000 pesticides registered for use in Canada.
The objective of the audit was to assess the effectiveness of the management control framework as it relates to governance, risk management and internal controls for the Pesticide Regulatory Program. The audit was conducted in accordance with the Treasury Board Policy on Internal Audit and the International Standards for the Professional Practices of Internal Auditing. Sufficient and appropriate procedures were performed and evidence gathered to support the audit conclusion.
PMRA is governed by various external and internal committees. The roles and responsibilities within headquarters and between the regions are clear and understood. As well, the Pesticide Regulatory Program has a suitable risk management lens that drives its regulatory process. The program would benefit from updating its strategic plan.
Overall, the program is well managed, but would benefit from enhancing internal controls related to operational planning and resource allocation. Funding comes from the Program's A-base funding; through cost recovery from application and regulatory charges; and through funding agreements. Once fully operationalized, the time reporting system should allow PMRA to acquire baseline information to support its resource allocations and cost-recovery program.
PMRA has good processes for regulating new pesticides; however, it would benefit from strengthening its re-evaluation of older pesticides. PMRA is legislatively required to conduct re-evaluations of registered active ingredients and products on a 15-year cycle. As a result, PMRA should invest in this activity by developing a targeted re-evaluation work plan. The regulatory work will also be better supported as the Program updates its aging IM/IT infrastructure.
The audit notes that PMRA has been actively working to modernize its cost-recovery regime and has initiated an effort towards updating its fee structure. The Program is working to have the revised fee structure align with the User Fees Act.
PMRA partners well with the regional compliance and enforcement inspectors; however, inspectors would benefit from having front-line electronic tools to support field work. Outreach and engagement are key program activities but have been operating without an agency-wide outreach and stakeholder engagement strategy. Lastly, the audit recommends developing and implementing a performance management framework.
The audit makes six recommendations to further strengthen the management control framework for the Pesticide Regulatory Program.
A - Introduction
1. Background
Health Canada's Pest Management Regulatory Agency (PMRA) was created in 1995 and is responsible for the regulation of pest control products under the Pest Control Products Act. In the administration of this Act, the primary objective is to prevent unacceptable risks to people and the environment from the use of pest control products. The Act controls the import, sale, manufacture and use of pesticides in Canada. The responsibilities described in the Act include registering new pest control products, re-evaluating previously registered products and enforcing compliance with the Act, which includes reporting of incidents, and specifying pesticide maximum residue limits. In addition, other federal legislation must be considered during the registration and re-evaluation of pesticide products (for example, the Species at Risk Act).
Pesticides include:
- Herbicides used against weeds;
- Insecticides used against insects;
- Fungicides and antimicrobial agents used against fungus and other microorganisms;
- Material and wood preservatives;
- Animal and insect repellants;
- Insect and rodent controlling devices, such as mosquito zappers and mouse traps;
- Algaecides used to control algae in pools.
Before a pesticide can be registered in Canada, it must undergo testing to determine the potential risks posed to human health and the environment and the pesticide's value. Determining value includes a consideration of both the benefits of the product and the product's performance (efficacy). Part of the review is to ensure that the product works, to determine an acceptable use pattern and establish the directions for use of the product. PMRA determines if a pesticide meets the health and safety standards and regulates the use instructions and safety precautions that appear on the product's label. In Canada, the regulation of pesticide products is achieved using pre-market and post-market scientific assessment, compliance and enforcement activities and communication and outreach with stakeholders. While Health Canada plays a role in regulating pest control products, health and safety is achieved through a coordinated effort between the federal, provincial and territorial and municipal governments, using a combination of acts, regulations, guidelines, directives and bylaws. Provinces and territories may further regulate the sale, storage, transportation and disposal of pesticides, issue permits for them and monitor their use. They may also set further conditions on the use of pesticides or allow municipalities to do so.
Health Canada budgets approximately $47 million in funding to deliver the regulatory program. Funding comes from the Program's A-base funding; through cost recovery from application and regulatory charges; and through funding agreements (see Appendix C).
2. Audit objective
The objective of the audit was to assess the effectiveness of the management control framework as it relates to governance, risk management and internal controls for the Pesticide Regulatory Program.
3. Audit scope
The scope of the audit included activities that span the lifecycle of pesticide management, including pre-market evaluation, post-market re-evaluation, post-market compliance and enforcement, international collaboration and outreach activities. As such, the audit scope included the activities and resources of PMRA, as well as the applicable resources of the Regions and Programs Bureau. The scope of the audit focused on program activities undertaken during fiscal year 2013-14 to fiscal year 2014-15. The scope of the audit did not assess pesticide regulatory decisions.
4. Audit approach
The audit approach included a review of documentation, policies, standards, guidelines and frameworks; interviews and observation; and inquiry, testing and analysis. Regional field visits were conducted in Manitoba, Ontario and Quebec to examine the post-market compliance and enforcement activities undertaken within the Regions and Programs Bureau. These regions were chosen based on their coordinating role, materiality and volume of inspections.
The audit criteria, outlined in Appendix A, were derived from the Pest Control Products Act, regulatory directives, the User Fees Act and the Office of the Comptroller General Internal Audit Sector’s Audit Criteria Related to the Management Accountability Framework: A Tool for Internal Auditors(March 2011). The audit was conducted in accordance with the Government of Canada’s Policy on Internal Audit and examined sufficient, relevant, reliable and useful evidence and obtained sufficient information and explanations to provide a reasonable level of assurance in support of the audit conclusion.
5. Statement of conformance
In the professional judgment of the Chief Audit Executive, sufficient and appropriate procedures were performed and evidence gathered to support the accuracy of the audit conclusion. The audit findings and conclusion are based on a comparison of the conditions that existed as of the date of the audit against established criteria that were agreed upon with management. Further, the evidence was gathered in accordance with the Internal Auditing Standards for the Government of Canada and the International Standards for the Professional Practice of Internal Auditing. The audit conforms to the Internal Auditing Standards for the Government of Canada, as supported by the results of the quality assurance and improvement program.
B - Findings, recommendations and management responses
1. Governance
1.1 Governance
Audit criterion: The Pesticide Regulatory Program has a governance structure that supports the strategic direction.
In support of the complex regulatory environment, the audit expected to find a governance structure to support timely and informed strategic decision-making related to the Pesticide Regulatory Program.
External advisory structure
The Pest Management Regulatory Agency (PMRA) maintains two advisory bodies: the Pest Management Advisory Council and the Economic Management Advisory Committee. As provided under the legislation, the Pest Management Advisory Council consists of a diverse group of stakeholders, with representation from a variety of sectors (for example, industry, academia, non-governmental and legal). The council advises the Minister of Health, to ensure that PMRA programs are consistent with the needs of Canadians within the national and global health, environmental, social and economic context; provides recommendations on broad strategic directions, management and overall priorities for PMRA; and provides advice and a forum for the exchange of views of all key stakeholders on issues affecting the management of pest control products in Canada.
The Economic Management Advisory Committee is an external stakeholder group composed primarily of industry representatives, which provides advice to the PMRA Executive Director on specific ways to improve efficiency and cost-effectiveness without compromising health or environmental protection and while maintaining industry competitiveness. Under its terms of reference, this group meets a minimum of twice a year.
Internal governance structure
The Agency Management Committee (AMC) is the senior decision-making committee chaired by the Executive Director. This committee consists of the directors general and directors who report to the Executive Director. The committee is responsible for establishing and fulfilling the mission and vision of PMRA and supporting the strategic management of PMRA’s responsibilities and monitors results.
The Science Management Committee (SMC) is chaired by the Chief Registrar. This committee is comprised of the PMRA directors general and the Chair of the Science Operations Committee. The primary interest for this group is the development of consensus decisions on significant registration applications (for example, new active ingredients, major new uses), re-evaluations, special reviews and emergency registrations of pest control products.
The Science Operations Committee (SOC) is chaired by a director from the Registration Directorate and is composed of directors and section heads from the PMRA directorates responsible for product evaluation, re-evaluation and compliance and enforcement activities. Reporting to the SMC, this group’s primary focus is the timely coordination and science integration of major active ingredient and product decisions into new registrations and re-evaluations.
Governance of the national compliance program occurs through a joint PMRA/Regions and Programs Bureau (RAPB) three-tier structure: Executive Committee at PMRA Executive Director/RAPB Senior Director General level; Horizontal Committee (DG level); and the Pesticide Committee (Director/Manager level). As well, a working level group also exists to share information and address operational issues. These committees meet regularly to improve strategic and operational planning, promote coherent policy and program management, encourage discussion and analysis of compliance issues and provide a national focus to coordinate consistent delivery. Regular tri-weekly meetings are conducted involving the Section Heads with RAPB’s Pesticide Regulatory Program Lead Director and the program’s regional managers. These meetings focus on technical compliance and enforcement, product evaluation and re-evaluation, as well as operational and financial matters.
The audit found terms of reference for each of the committees, relevant agendas and records of decision. The document review, combined with interviews, demonstrates that PMRA and RAPB have implemented appropriate program governance structures to support the strategic direction.
Strategic planning
In the 2014-15 Report on Plans and Priorities, Health Canada (the Department) made several commitments related to advancing pesticide management. The Department committed to working with trading partners and other international regulatory organizations to reduce the regulatory compliance burden and to improving work in international science in support of prevention of the risks from the use of pest control products. It also committed to supporting more timely access to safer pesticides through effective processes for pre-market registrations, as well as post-market assessments, compliance and enforcement and outreach activities. In particular, it committed to continuing to implement the re-evaluation program based on a 15-year cycle, in accordance with the Pest Control Products Act; continuing to engage stakeholders while reviewing regulations and legislation, including the Pest Control Products Act; and ensuring relevance and managing risks associated with sustaining effective regulations in an evolving environment. In addition to the activities described above, emphasis was to be placed on continuing to develop a proposal for a revised cost-recovery framework for pesticides.
Given these commitments, there was an expectation that these activities would be highlighted in PMRA’s strategic plan and operational planning. A cross-examination between the Report on Plans and Priorities and the 2008-2013 PMRA Strategic Plan noted that PMRA would benefit from refreshing its strategic plan, since there were only two consistent priorities: working towards international regulatory cooperation and engaging stakeholders. Updating the strategic plan has been identified as a 2014-15 fiscal year deliverable; such an update would assist in aligning ongoing commitments.
Recommendation 1
It is recommended that the Executive Director, Pest Management Regulatory Agency, update the strategic plan (including the outreach strategy).
Management response
Management agrees with the recommendation.
PMRA is in the process of developing an updated Five-year Strategic Plan, including a strategy for outreach activities.
1.2 Roles and responsibilities
Audit criterion: The Pesticide Regulatory Program has documented roles and responsibilities.
In order to meet the broad objectives of the regulatory program and to deliver the program efficiently and effectively, it is critical to have clearly defined roles and responsibilities to support appropriate accountability. PMRA, supported by the RAPB, currently has four areas of operational focus: evaluation of new products, re-evaluation of registered products, compliance and enforcement and program management. Each of these areas is well-documented and executed by staff.
Evaluation of new products ($18.8 million)
Before a pesticide can be sold in Canada, applicants are required to provide sufficient data to show that the product does not pose unacceptable risks to health and the environment and that the product has value. The Registration Directorate screens new pesticide product submissions prior to the evaluation for health, environmental and value considerations in order to determine that submissions are complete and meet the format, content and fee requirements. The Health Evaluation Directorate evaluates data and other documentation submitted on pesticides (new and older products) to identify possible human health effects and establish the levels at which humans can be exposed to the products without harm. The directorate also makes recommendations for restricting human exposure and use to levels where there are no health risks or concerns.
The Environmental Assessment Directorate evaluates data and other documentation submitted on the environmental toxicology of products, as well as their environmental fate. To address environmental concerns that may arise from the intended use of a product, the directorate also makes recommendations for restrictions on use that lessen risk. The Value Assessment and Re-Evaluation Management Directorate has several responsibilities. It evaluates pesticide submissions to establish whether the product has acceptable value for the purposes claimed when the product is used according to label directions. This directorate is also responsible for coordinating submissions for minor use label expansions, research authorizations, research notifications and applications for emergency registrations. The Policy, Communications and Regulatory Affairs Directorate is responsible for the publication of regulatory decision documents for consultation purposes.
Re-evaluation of products ($8.8 million)
The Pest Control Products Act requires that pesticides undergo a re-evaluation every 15 years to ensure that registered products continue to meet modern scientific requirements. Re-evaluation involves a review of the pesticide active ingredients and their associated uses, in relation to updated science and data, to determine whether and under what conditions their continued registration is acceptable.
Compliance and enforcement activities ($6.6 million)
PMRA has overall responsibility for promoting, verifying and enforcing compliance with the Act and its regulations in collaboration with provincial and territorial governments, other federal departments and agencies and international regulatory authorities. In delivering on the compliance activities, PMRA receives direct support from Health Canada’s Regions and Programs Bureau (RAPB) staff, who contribute to program planning and development and are the primary agents of delivery of the National Pesticide Compliance Program (NPCP) across the regions. This involves undertaking compliance promotion and monitoring activities, responding to incidents and complaints and taking enforcement action on violations of the Pest Control Products Act, as required.
Outreach
In addition to the regulatory activities, PMRA also supports public awareness in relation to pest control products by informing the public and facilitating access to relevant information.
Program management and support ($12.6 million)
Program management and support is provided for each of the program activities. The Executive Director’s Office takes a leadership role in providing senior-level management within PMRA, in pursuit of its mandate, mission, strategic direction, operational goals and objectives. The Policy, Communications and Regulatory Affairs Directorate develops and implements federal policy and legislation for pest control products and works with other government bodies, grower groups and industry to facilitate information exchange. The Strategic Planning, Financial and Business Operations Division provides business management support services to PMRA, including strategic planning, financial management, continuous learning, audit and evaluation and all facilities-related activities.
2. Risk management
2.1 Risk management
Audit criterion: Risks and opportunities related to the delivery of the Pesticide Regulatory Program are identified, assessed and have mitigating strategies.
Program risks
For a large regulatory program, it is expected that management would identify overall program risks; that the risks would be assessed for likelihood of occurrence and magnitude of impact; and that a roll-up of the individual risks would be conducted to form a complete program risk analysis.
Program risks were identified with mitigation strategies in PMRA’s branch risk registry 2012-2013. PMRA has also reported program risks in its Strategic Plan 2008-2013. Each of the risks in the branch risk profile identified the risk factors, potential impacts and risk mitigation strategies. Examples of internal risks and mitigating strategies include financial constraints, keeping abreast of modern science, maintenance and support systems, public and stakeholder expectations and sustaining and growing partnerships.
PMRA reports that for new chemicals, the program relies heavily on a modern scientific risk assessment framework that is aligned with major regulators globally and includes collaboration with those regulators to ensure that Canadian decisions are made in consideration of the best science available.
PMRA recognizes the current program risks with the re-evaluation of older chemicals, specifically in relation to the nature of the older data and information that supports continued registration. Updating the data and information to modern standards and conducting new risk assessments is challenging and can require considerable time. Risk management strategies are also reliant on this data and information.
PMRA reports that its mitigating strategies include the development and implementation of risk mitigation measures, the development of national guidelines using a risk-based framework, the monitoring of pesticide health and environmental incidents, the monitoring of sales data, and taking regulatory action as required. However, the Program should monitor the mitigating strategies to ensure effectiveness.
Pesticide risk assessment and risk management
PMRA has committed to the constant evolution of its risk assessment and risk management approaches to ensure that its regulatory decisions continue to represent modern science and continue to follow accepted practices of major OECD countries globally. The framework used by PMRA relies on the use of conservative risk assessment approaches, and the use of appropriate risk mitigation measures to ensure that pest control products continue to meet international standards.
The audit found that risks related to the delivery of the Pesticides Regulatory Program are identified, assessed and have mitigating strategies.
3. Internal controls
3.1 Program planning
Audit criterion: Objectives, priorities and resource allocations are established to support the achievement of the Pesticide Regulatory Program's objectives.
According to Health Canada’s expenditure management framework, branches are responsible for determining how existing programs must be managed to meet expenditure targets and priorities. As such, it was expected that PMRA would have developed plans and budgets based on objectives and expected results. It was also expected that operational plans have been developed to guide the delivery and resourcing of all Pesticide Regulatory Program activities.
The audit examined operational plans developed at the branch and directorate level and the documents used to make decisions on resource allocations. The audit found an appropriate branch-level 2013-14 PMRA Operational Plan. The Plan included priorities, a summary of planned spending and a summary of key investments (for example, information technology initiatives). A cross-examination between the Departmental Operational Plan and PMRA Operational Plan noted clear linkages.
At the directorate level, detailed operational plans were to be developed. The audit expected to find planning information that was consistently and comprehensively articulated in directorate operational plans, including objectives and expected results; planned activities, milestones and timing; planned resources and activity assignments to support delivery of the activities; and planned outputs (and associated timing) of the activities. The audit found that directorate operational plans varied significantly in consistency and completeness in relation to these elements (see Recommendation 2).
In conjunction with PMRA planning efforts, the Regions and Programs Bureau (RAPB) contributes to the development of operational plans for the compliance and enforcement activity (see Section 3.3).
PMRA would benefit from having more consistent directorate-level operational plans to support more effectively inter-directorate regulatory activities and financial and human resource allocations.
Recommendation 2
It is recommended that the Executive Director, Pest Management Regulatory Agency, review the annual planning process to enhance its methods for tracking program-related costs and include all financial and non-financial information.
Management response
Management agrees with the recommendation.
PMRA continues to streamline its operational planning process and its corresponding plans to align better all financial and non-financial information. This work will support PMRA’s new cost-recovery regime and the reporting requirements under the User Fees Act.
3.2 Product evaluation and re-evaluation activities
Audit criterion: Product registration, evaluations and re-evaluations are prioritized, planned and executed.
PMRA carries out pre-market and post-market regulatory activities. The pre-market activities are conducted before a company can sell a new pest control product in Canada. Depending on the complexity of the submission, a complete evaluation can take anywhere from a number of weeks to about two years. The evaluation results either in the product being granted registration and allowed for sale and use in Canada or in the product being refused registration (see Figure 1). Moreover, the Pest Control Products Act requires that pesticides undergo a re-evaluation every 15 years, so that registered products continue to meet evolving scientific requirements.
Figure 1: Product Evaluation Process
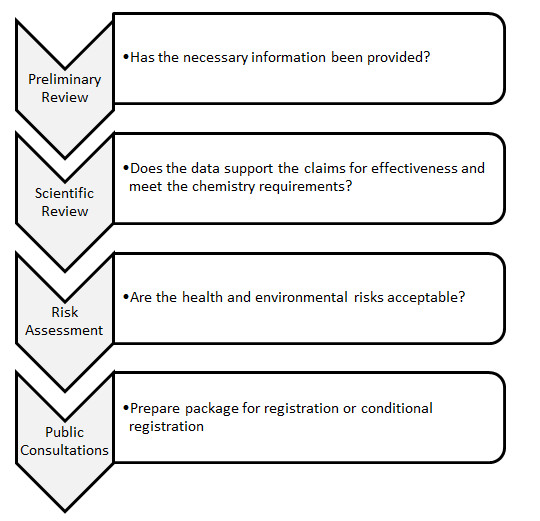
Text description
Figure 1 outlines the process for evaluating new pesticide product requests for registration.
The process for evaluation new pesticide product submissions asks three questions.
The preliminary review stage asks the questions: Has the necessary information been provided?
The scientific review stage asks the question: Does the data support the claims for effectiveness and meet the chemistry requirements?
The risk assessment stage asks the question: Are the health and environmental risks acceptable?
At the final stage, a bilingual consultation document or Proposed Registration Decision is published for all major decisions. The comments received during the consultation period are considered before rendering the final regulatory decision.
Once all the questions have been answered, PMRA determines whether or not a product should be granted full registration or conditional registration. A package is prepared that specifies the conditions of registration and includes detailed use instructions, so that the product can be used safely.
Pre-market activity
PMRA provides industry with detailed submission guidance and a pre-submission consultation service to help applicants prepare the data package. The Registration Directorate screens new pesticide product submissions, to ensure that they meet the format, content and fee requirements. All submissions are completed electronically. Evaluation of new active ingredients with accompanying new products may last up to 22 months, with a consultation period of 45 days.
The Health Evaluation Directorate evaluates the scientific data and documentation submitted on pesticides to identify possible human health effects and establish the levels at which humans can be exposed to the products without any harm. Areas of focus include toxicology, occupational and dietary exposure and food residue assessments. The directorate also makes recommendations for mitigation measures that reduce human health risks.
The Environmental Assessment Directorate evaluates the documentation submitted on the environmental toxicology of a product, as well as its environmental fate (what happens to the pesticide once it enters the environment). As well, to address environmental concerns that may arise from the intended use of a product, the directorate makes recommendations for restrictions on its use that would lessen the risk. This could include, for example, label statements outlining buffer zones, timing and frequency of applications and rate at which the product can be applied.
An application for registration of a pesticide must establish that the product has value for the purposes claimed when the product is used according to label directions. The Value Assessment and Re-evaluation Management Directorate evaluators carry out these assessments, which include an evaluation of the product’s contribution to pest management, as well as an assessment of its efficacy, its effects on host organisms, the health and environmental benefits and the social and economic impact. The "value" aspect of the assessment is linked to efficacy. For example, it looks at whether the product works as intended, improves crop yield or reduces damage by pests, depending on the intended use of the product.
Once all the component parts of a submission have been evaluated, PMRA determines whether or not a product should be granted registration. In all cases, conditions of registration are specified, including detailed use instructions, so that the product can be used safely.
Under the 2006 Pest Control Products Act, pesticides can be granted a conditional registration that is valid for three years. This regulatory tool takes the place of the previous "temporary registration" that had a validity period of only one year. The conditional registration allows a time frame during which additional information may be generated by an applicant for the purpose of satisfying any request by the Minister to provide additional information in support of a registration decision. The validity period of a conditional registration may be extended by two years when the registrant complies with the Section 12 notice for the purpose of providing the PMRA with the time needed to conduct an evaluation of the additional information, and arrive at a final decision. Full registration is granted when the required information has been submitted, reviewed and accepted. When PMRA grants a conditional registration, it issues a special notice which notes the additional information required.
In September 2014, it was reported to the Standing Committee on Agriculture and Forestry that 88 products remain conditionally registered. Of these, 28 have been conditionally registered for more than five years and eight of them for more than 10 years. However, PMRA reports that this number represents a decrease relative to the 13% of total products that were once being conditionally registered.
Emergency registrations allow the Minister to register a pest control product for a period not exceeding one year, for the emergency control of pest infestations that are seriously detrimental to public health, domestic animals, natural resources or other things. In 2013-14, PMRA reported 28 emergency registrations.
In April 2013, a new regulatory directive was issued concerning the submission process, which outlines performance timelines for managing applications for all submission categories (see Appendix C). The timelines vary based on the category and type of the submission. Most applications are typically reviewed in chronological order but timelines may be adjusted if there is a critical need or a grouping of related submissions.
Post-market activity
Once a product is registered, PMRA has various post-market work requirements, which include tracking conditional registrations, tracking emergency registrations, conducting 15-year re-evaluations and conducting special reviews.
For active ingredients registered after 1994, the Act requires a cyclical re-evaluation (that is every 15 years). Product re-evaluation involves a review of a pesticide’s active ingredients and its associated uses on the basis of updated science and data, to determine whether and under what conditions its continued registration is acceptable. Depending on the assessment results, Health Canada may retain the registration with no changes; retain the registration with amendments to the label to minimize risk concerns or retain the registration with confirmatory data requirements. In some cases, registration for all uses may be cancelled. In 2010, PMRA began to implement cyclical re-evaluation. For newer active ingredients with more modern data-bases, PMRA reports that it is taking a targeted approach to the re-evaluation, focussing only on those aspects of the health and environmental risk assessments that need to be updated.
On an annual basis, PMRA develops an overall internal work plan for the re-evaluation program. For each active ingredient within that work plan (under the 15-year statutory re-evaluation cycle), there is both an internal detailed project plan as well as a published work plan that includes a target time frame for proposed decisions. Management reports that the work plan is aligned with that of its international partners in order to leverage efforts across regulatory programs. A summary of re-evaluation progress is made available to the public through the PMRA annual reports.
Despite planned efforts, in management committee briefings, it was noted that some of the proposed re-evaluation decisions scheduled for 2013 would be late. Further, new re-evaluations slated for initiation in the last quarter of 2013 were not initiated as indicated in the internal plan; however, PMRA reports that it met the legislative obligation for initiation of these re-evaluations (that is, 15 years plus 1 year). In addition, the publication of numerous preliminary re-evaluation decisions did not meet the targets in the revised schedule.
The Office of the Auditor General has reported on the issue of evaluating older pesticides and noted improvements between 2003 and 2007 in the number of active ingredients re-evaluated. However, the 2008 audit also stated that PMRA still had to fully complete 235 active ingredient re-evaluations in two years, when it has taken six years to fully complete 166. Given the limitations of PMRA’s electronic system, it does not yet have an electronic tool to guide its re-evaluation work, including detailed time and cost estimates by which to gauge its level of effort and progress or a priority listing of the pesticides to be re-evaluated (see Recommendation 4).
Through interviews and document reviews, it was noted that PMRA is aware of the risks associated with the re-evaluation of older products, such as the difficulty in obtaining historical data, inconsistent labels and difficulties in getting information from registrants because the products are already on the market. As well, the delays in review allow products to remain for sale while they are still being re-evaluated. There were also concerns with the delays in implementing the results of re-evaluations and in phasing out existing products.
Pesticide active ingredients that were registered in Canada prior to December 31, 1994 needed to be re-evaluated to ensure that they continued to meet modern scientific standards. This consisted of 401 active ingredients and their registered end-use products. These re-evaluations began in the early 2000s as a part of the Chemical Management Plan. PMRA continues to work on these older chemicals.
PMRA is also concurrently engaged in special reviews of various active ingredients and their associated end-use products. According to the Act, special reviews are required when there are reasonable grounds to believe that the health or environmental risks or value are unacceptable or when an OECD member country prohibits all uses of a pesticide. To date, special reviews of 26 active ingredients have been announced and four have been completed.
Management reports that a guidance document on the approach for special reviews has been published, with internal standard operating procedures and timelines. It also reports that progress on the special reviews is monitored on a regular basis by senior managers and is reported in the annual report. The timeline for a special review varies on a case-by-case basis, depending on the aspect(s) of concern that must be considered, such as delays in obtaining documents from OECD-member countries. The audit requested information on performance against standards for these special reviews, including time and money spent, impact on evaluations and re-evaluations, but this information was not available. Management reports that the performance information was not available because this was a particularly large influx of special reviews.
Pesticide incident reports also provide valuable information that is taken into account during a product’s re-evaluation. In 2012, there were 1,960 pesticide incidents reported. The majority of the effects reported were minor in nature. Assessment of the pesticide incidents reported in 2012 led to several actions being taken by PMRA. These included the revision of label instructions; the development of best practices to reduce the exposure of bees to dust generated during the planting of treated seed; and continued monitoring for adverse effects related to particular pesticides.
PMRA reports that it manages pesticides on a product’s lifecycle. The approach could be strengthened by building product knowledge continuity from the time a pesticide product is presented for pre-market review to the time it is re-evaluated. To achieve this approach, PMRA should integrate pre- and post-market results and monitor incident reports to develop a work strategy. The Program reports that it has developed an integrated work plan for 2014-2018, which includes older re-evaluations, cyclical re-evaluations and special reviews.
Recommendation 3
It is recommended that the Executive Director, Pest Management Regulatory Agency, enhance the work plan to integrate better re-evaluation and special review activities.
Management response
Management agrees with the recommendation.
PMRA recognizes the importance of an integrated work plan for operational planning purposes. To this end, PMRA has recently drafted a three-year work plan that integrates both re-evaluation and special review activities. The work plan takes into consideration international re-evaluation timelines, so that PMRA is able to capitalize and coordinate efforts with its international partners.
Information technology
PMRA relies on information technology to aid in the delivery of its regulatory mandate. The primary application is the electronic Pesticide Regulatory System (ePRS), which is a submissions management system and document repository. While an important tool for PMRA, over the years the system has become difficult and costly to maintain and enhance, due to increased complexity and a customized configuration created through “hard-coded” programming of business rules.
Moreover, there are a number of core regulatory processes that are not fully supported by the system, so that PMRA staff have had to implement manual “work-arounds”. Examples of these gaps include the system's incapacity to support the full implementation of PMRA’s Management of Submissions Policy (launched in 2013); international joint product reviews and product re-evaluation processes; product sales reporting requirements, incident reporting requirements, current reporting requirements and the anticipated reporting requirements under the User Fees Act.
In response to these challenges, PMRA has begun to plan for the modernization of its information system environment. For example, a requirements definition exercise has been undertaken to define the key business processes and rules for managing new product evaluation processes. However, work remains to fully define PMRA’s business requirements across its various regulatory processes in order to develop and implement a comprehensive solution. Preliminary estimates for this initiative indicate an implementation cycle of four-plus years and a cost in excess of $8 million. PMRA is planning to make a new presentation in March 2015 that will respond to operational needs and reflect recent government guidance concerning new IM/IT systems.
Those responsible for managing registrations, evaluations and re-evaluations concur with the deficiencies in the existing system and the need to maintain their own manual and computerized spreadsheets to track progress against deadlines. They expect that the upgraded system will respond to management needs. In the interim, however, they use bilateral meetings and a “Trackers Club” of managers who discuss pressure points and potential solutions.
PMRA reports that while it is able to provide the status of evaluations and re-evaluations, it requires a significant amount of manual work and the maintenance of various spreadsheets and documents. As a result, providing a clear picture of the status of evaluations and re-evaluations to determine the progress against plans and identify where there are backlogs in the system is labour intensive and challenging.
Recommendation 4
It is recommended that the Executive Director, Pest Management Regulatory Agency, develop an integrated IM/IT strategy to support the Pesticide Regulatory Program.
Management response
Management agrees with the recommendation.
PMRA has been developing a multi-year IM/IT Strategy, including a four-year project to modernize its aging IM/IT system infrastructure. This project was approved by PMRA senior management in January 2015 and is planned to commence in early fiscal year 2015-16.
In addition, a farther-reaching, integrated IM/IT strategy will be produced by end of March 2015. It will address continued electronic business process improvement and increased transparency and availability of electronic information, driven by key initiatives such as the new cost-recovery regime, partnership initiatives and international electronic pesticide information standards.
3.3 Cost recovery
Audit criterion: The Pesticide Regulatory Program is in compliance with the requirements of pesticide user-fee regulations.
In response to stakeholder recommendations, pesticide regulatory fees were established in 1997. Currently, PMRA derives fee revenue from two sources: application fees and annual regulatory charges.
Application fees
For most types of applications, fees are determined by the data requirements associated with the application. The scientific data requirements are determined by the nature of the pesticide, the purpose of the application and the proposed use. A fee is charged for each type of scientific review required, such as chemical, toxicological and environmental fate. If an application relies on previously assessed data, there is no charge to applicants for the review of that data, although there are still some costs incurred by Health Canada.
Applications to register products containing new active ingredients may also be eligible for reduced fees if the anticipated revenue from sales within the first three years is less than 10 times the calculated application fee.
For some applications, a fixed or flat fee is charged. In addition, some pesticide products are currently exempt from application fees (other than the label review fee).
Annual charge
Registrants are required to pay an annual regulatory charge for each registered pest control product. The regulatory charge is set at $2,690 or 3% of annual sales of the pest control product, whichever is the lower amount, with a minimum fee of $75. During 2013-14, PMRA reports that it generated $7.9 million in revenue from the two fee types.
While the pesticide regulatory fees have not changed since they were put in place in 1997, PMRA asserts that in addition to inflationary impacts, the underlying regulatory work has evolved in terms of the increasing complexity of submissions and overall workload; the increasing international cooperation and global joint reviews; technology gaps; and ongoing developments in science and related policy.
In response, PMRA has initiated an effort towards modernizing its fee structure and fees. In order to meet the reporting requirements of the User Fees Act and associated Treasury Board Secretariat guidelines, PMRA will be reviewing its activity tracking system, to enable more timely and effective costing of activities for reporting purposes. As well, management reports that the activity tracking system will be used to validate program activity expenditures against operational plans. PMRA also reports that it is launching a renewed financial tracking system. Together, these systems will support improved reporting on program activities, costs and expenditures (see Recommendation 2 and Recommendation 6).
To date, PMRA has conducted a costing exercise, cost benefit and business impact analysis. It also developed an initial proposal for the specific fees to be levied, which were published in a stakeholder consultation document earlier in 2014. In addition, PMRA has developed preliminary investment estimates to identify the potential priority uses for additional user fee revenues to be generated. PMRA reports that it is implementing a renewed time tracking tool that goes beyond the current tracking of allocations and expenditures against costing codes. Going forward, this may assist in deriving fully costed activities and provide additional support for the cost-recovery process.
3.4 Compliance and enforcement activities
Audit criterion: Compliance and enforcement activities are prioritized, planned and executed.
Pesticide compliance and enforcement is a shared responsibility. PMRA is responsible for setting the strategic direction, establishing program priorities and policies and determining how those priorities are implemented across the country. The Compliance, Laboratory Services and Regional Operations Directorate (CLSROD) of PMRA, along with the Regions and Programs Bureau (RAPB), is responsible for verifying compliance with the Act, regulations and relevant policies and carrying out enforcement responses. Outreach to stakeholders is a joint responsibility.
Pesticide compliance and enforcement is guided by the Act and its regulations; PMRA’s Regulatory Directive 2007-02 Compliance Policy; and the Inspector’s Field Operating Manual. The manual in particular provides detailed information on PMRA, the legislation, product regulation, the inspector’s authorities and responsibilities and communication roles. It also provides guidance on the completion of inspections, enforcement responses, investigations and search warrants.
PMRA and RAPB have implemented a joint planning process to define compliance and enforcement priorities, programs and planned workloads, resulting in the annual National Pesticide Compliance Program (NPCP). The NPCP incorporates the national requirements, as well as the requirements particular to each region. Through this process, an environmental scan is developed, priorities are established, and an overall national operational plan is produced.
The audit conducted focus groups in three regions to validate the execution of the compliance and enforcement plan. Inspectors noted that the plan is executed; however, unforeseen events and emerging priorities may require adjusting approved plans. Inspectors also noted that the regional workload is often demanding and that more focused time on specific products would allow them to better identify and track regional trends.
Under the Pest Control Products Act, PMRA has access to various enforcement tools and powers of inspection, and penalties may reach up to $1 million. The audit conducted focus groups with the regional inspectors and notes that an enforcement continuum approach is applied to situations of non-compliance, taking into consideration the severity of the violation, the history and the harm. Some examples of tools available to inspectors include education (verbal and written), letters and penalties. The audit examined compliance and enforcement activities. The Program supplied data over three fiscal years (see Appendix E). For the period analyzed, enforcement actions increased by 35%. This can be explained primarily by the increase in denials of entry and requests to cease activity/remove product.
The audit finds that compliance and enforcement activities are prioritized, planned and executed. To further support the efficiency of this activity, regional inspectors would benefit from front-line electronic tools. Inspectors spend time in the field completing paper forms and transcribing pesticide registration numbers. Once back at the office, they input the same information into the compliance tracking database. Front-line tools, such as tablets with Internet access, would allow for searches of reference materials, expedite note-taking and report drafting and allow inspectors to educate clients on site.
Recommendation 5
It is recommended that the Senior Director General, Regions and Programs Bureau, develop a strategy for front-line electronic tools to support compliance and enforcement.
Management response
Management agrees with the recommendation.
Enabling efficient mobile inspection activities by investing in electronic tools is a priority for the compliance and enforcement function. The Regions and Programs Bureau has developed a strategy for the use of electronic tools and plans to deploy them by September 2015.
3.5 Outreach activities
Audit criterion: The objectives of the outreach activities are defined and the activities are planned and executed.
Public awareness of pest control products is a key activity for PMRA. It does this by informing the public, facilitating public access to relevant information and encouraging public participation in the decision-making process.
Within this role, PMRA serves the information needs of a broad and diverse set of audiences with a highly varied understanding of pesticides, pesticide regulations and potential pesticide risks. These audiences include, but are not limited to, pesticide manufacturers, distributors, sellers, growers, other users and the general Canadian public.
As such, PMRA develops communication products that inform Canadians on regulations related to pesticides and encourage Canadians to take part in consultations towards decision-making. In support of these strategies, PMRA has undertaken a variety of tactical outreach approaches. These include outreach activities undertaken and managed centrally as well as those undertaken by regional offices. Examples of outreach activities undertaken at headquarters include: developing and maintaining content on PMRA’s website; developing and executing an exhibit program focused on public events; developing and distributing periodic articles for publishing in various newspapers, magazines and newsletters; maintaining a call line that provides a channel for questions and requests from the public and other groups. Regional outreach activities may include targeted education initiatives, stakeholder presentations and tradeshow displays. Performance-related information regarding these activities should be collected and analyzed to inform decision-making (see Recommendation 6).
While these outreach efforts are recognized by PMRA management as having merit, management acknowledges the continuing challenge to adequately inform the public so that public perception of pesticides and chemicals is balanced. Management notes that it is difficult to know how much Canadians understand regarding Health Canada’s role in the evaluation and registration of pest control products. Management recognizes the continued need for a national outreach and stakeholder engagement strategy, including targeted audiences, key communication messages, communication channels and communication products and activities.
In addition, PMRA recognizes the importance of developing a robust method of measuring and monitoring the full extent of its outreach activities and resources used and addressing the impact of its outreach investments and activities. As a part of PMRA’s process to update its strategic plan, it would be important to include an outreach strategy (see Recommendation 1).
3.6 International collaboration
Audit criterion: International collaboration activities support the achievement of the Pesticide Regulatory Program's objectives.
PMRA plays a role in international efforts to encourage regulatory cooperation and, where possible, harmonize regulatory systems in an effort to facilitate consistency of approach and to have the most current science available to support regulatory decisions.
International collaboration is a key strategy for the Pesticide Regulatory Program. The strategic plan indicates that by contributing to and benefiting from science internationally, its assessments and decisions are able to integrate cutting-edge science; use a progressive approach based on level of anticipated risk; support an influential international voice; reach high international standards for protection of human health and the environment; and provide Canadians with timely access to safer and innovative pesticides.
The strategic plan further articulates supporting measures, such as advocating for the adoption of international regulatory standards and continuing to use global joint reviews, which is the preferred option for the evaluation of new pesticides and the re-evaluation of older pesticides.
PMRA undertakes a range of international collaboration activities across the organization, including participation in and contribution to a number of international forums, partnerships and working groups focused on regulatory co-operation and scientific best practices. Illustrative examples of international collaboration include supporting the OECD on working groups and targeted initiatives and contributing to the introduction of international environmental treaties such as the Stockholm and Rotterdam Conventions. As noted, PMRA also participates in and conducts global joint reviews for pesticide products and co-operative pesticide product re-evaluations with international regulatory partners.
Evidence of PMRA’s international collaboration activities is also documented in departmental accountability reports. The Program set out as a performance target that 80% of new pesticides be reviewed in collaboration with international partners. In addition, PMRA’s international collaboration efforts and achievements are highlighted in its Annual Report to Parliament and Directorate Operations Plans, which identify details of specific activities and deliverables.
The audit finds that international collaboration activities are used to support the achievement of program objectives.
3.7 Performance management framework
Audit criterion: Management has identified performance measures linked to planned results and performance is monitored against planned results.
Performance information is central to supporting effective management decision-making. Management and external stakeholders require performance information to determine the extent to which expected results have been achieved. As such, PMRA management, in concert with RAPB, should have a comprehensive performance management framework that is linked to planned results, regularly monitored and used to support management decision-making.
PMRA has developed various elements of a performance measurement framework to support the management of pesticide regulatory activities. PMRA has defined summary level performance indicators and targeted levels of performance linked to expected program results. These are reported in departmental accountability documents. Table 1 provides examples of the performance indicators presented in the 2013-14 departmental Report on Plans and Priorities, for illustrative purposes.
| Program expected results | Performance indicators | Targets |
|---|---|---|
| Industry meets regulatory requirements for new pesticides. | % of submissions that meet regulatory requirements. | 80% |
| Pesticides in the marketplace continue to meet modern scientific standards. | % of registered pesticides that are re-assessed according to the Re-evaluation Work Plan. | 80% |
| International collaboration is leveraged to maximize access to global science for the registration of pesticides. | % of new pesticides reviewed in collaboration with international partners. | 80% |
In addition, PMRA has defined a series of performance indicators and quarterly reporting of performance against key program areas, as part of a quarterly dashboard. Illustrative performance attributes include:
- Pre-market regulatory activities (for example, timeliness of completed Category A, B, C and generic submission reviews);
- Post-market regulatory activities (for example, completion rate (against plan) and timeliness of inspections and compliance verifications, volumes of product re-evaluation decisions, volumes and priority of incidents reported).
As required under the Act, PMRA produces an annual report that includes a qualitative and quantitative summary of program performance. In addition to the presentation of qualitative information related to pre-market and post-market regulatory activities, the annual report presents quantitative performance information on the timeliness of completed Category A, B and C submission reviews (see Appendix D for a description of the submission categories). These performance measurement elements provide an initial basis to support management evaluation of operations and decision-making; however, additional performance measurement methods and tools would support more comprehensive measurement of program inputs, activities, outputs and results.
Moreover, while logic models exist for many components of the Program, PMRA should integrate these in the development of a comprehensive logic model to demonstrate the relationship among program inputs, activities, program outputs and expected results. A program logic model would support PMRA management’s efforts to communicate more clearly its strategies and operations.
From an activity perspective, PMRA would benefit from setting out its performance measures, including systematic monitoring in key areas such as the timeliness and status of product re-evaluations and the scope and reach of PMRA’s outreach activities. From a results perspective, the audit notes that the Program would benefit from defining the expected results and measurement approaches related to its various pre- and post-market activities (for example, definition of the expected value added or the impact of compliance and enforcement activities). Enhancement of the current performance measurement framework would support PMRA management in its evaluation of program operations and decision-making and enable PMRA to better tell its performance story.
Recommendation 6
It is recommended that the Executive Director, Pest Management Regulatory Agency, develop a performance management framework to support pesticide regulatory decision-making.
Management response
Management agrees with the recommendation.
PMRA supports the strengthening of its performance management framework. Accordingly, it has commenced a mapping exercise of PMRA’s performance measures, indicators and commitments within the departmental PAA/PMF structure. This work will support the departmental Performance Measurement Initiative, as well as the reporting requirements of the User Fees Act.
C - Conclusion
The Pesticide Regulatory Program is governed by various external and internal committees. The roles and responsibilities of staff at headquarters and in the regions are clear and understood. As well, the Program has a suitable risk management lens that drives its regulatory process. However, the Program would benefit from updating its strategic plan.
Overall, the Program is well managed, but would benefit from enhancing internal controls related to operational planning and resource allocation. Funding comes from the Program’s A-base funding, through cost recovery from application and regulatory charges and through funding agreements. Once fully operationalized, the time reporting system should allow PMRA to acquire baseline information to support its resource allocations and cost-recovery program.
PMRA has good processes for regulating new pesticides; however, as part of the legislation, there is a requirement to conduct re-evaluations of products on a 15-year cycle. This work is challenging to conduct. As a result, PMRA would benefit from investing in this activity by developing a targeted re-evaluation work plan. PMRA has been actively working to modernize its cost-recovery regime and has initiated an effort towards updating its fee structure and fees to ensure that they align more closely with the objectives of the User Fees Act and associated Treasury Board of Canada Secretariat guidelines. The audit also recommends that the Program focus on updating its aging IM/IT infrastructure.
PMRA partners well with the regional compliance and enforcement inspectors. However, inspectors would benefit from having front-line electronic tools. Outreach is a key program but it has been operating without an agency-wide outreach and stakeholder engagement strategy. Lastly, the audit recommends developing and implementing a performance management framework.
Appendix A - Lines of enquiry and criteria
| Criteria title | Audit criteria |
|---|---|
| Line of enquiry 1: Governance | |
| 1.1 GovernanceTableau 1 note de bas de page 1 | The Pesticide Regulatory Program has a governance structure that supports the strategic direction. |
| 1.2 Roles and responsibilitiesTableau 1 note de bas de page 1 | The Pesticide Regulatory Program has documented roles and responsibilities. |
| Line of enquiry 2: Risk management | |
| 2.1 Risk managementTableau 1 note de bas de page 1 | Risks and opportunities related to the delivery of the Pesticide Regulatory Program are identified, assessed and have mitigating strategies. |
| Line of enquiry 3: Internal controls | |
| 3.1 Program planningTableau 1 note de bas de page 1 | Objectives, priorities and resource allocations are established to support the achievement of the Pesticide Regulatory Program's objectives. |
| 3.2 Product evaluation and re-evaluation activitiesTableau 1 note de bas de page 1,Tableau 1 note de bas de page 2 | Product registration, evaluations and re-evaluations are prioritized, planned and executed. |
| 3.3 Cost recoveryTableau 1 note de bas de page 1,Tableau 1 note de bas de page 2,Tableau 1 note de bas de page 3 | The Pesticide Regulatory Program is in compliance with the requirements of pesticide user-fee regulations. |
| 3.4 Compliance and enforcement activitiesTableau 1 note de bas de page 1,Tableau 1 note de bas de page 2 | Compliance and enforcement activities are prioritized, planned and executed. |
| 3.5 Outreach activitiesTableau 1 note de bas de page 1,Tableau 1 note de bas de page 2 | The objectives of the outreach activities are defined and the activities are planned and executed. |
| 3.6 International collaborationTableau 1 note de bas de page 4 | International collaboration activities support the achievement of the Pesticide Regulatory Program's objectives. |
| 3.7 Performance management frameworkTableau 1 note de bas de page 1 | Management has identified performance measures linked to planned results and performance is monitored against planned results. |
|
|
Appendix B - Scorecard
| Criterion | Rating | Conclusion | Rec # |
|---|---|---|---|
| Governance | |||
| 1.1 Governance | Needs minor improvement | External and internal governance committees are in place. However, the strategic plan needs to be updated, with better performance data. | 1 |
| 1.2 Roles and responsibilities | Satisfactory | The roles and responsibilities of PMRA staff and staff in Regions and Programs Bureau are clear and understood. | |
| Risk management | |||
| 2.1 Risk management | Needs minor improvement | Risks are identified, assessed and have mitigating strategies. However, performance measures to track the effectiveness of mitigating strategies should be developed. | |
| Internal controls | |||
| 3.1 Program management | Needs minor improvement | The overall branch operational plan is good. However, directorate plans vary and should be monitored to reflect progress against financial and work objectives. | 2 |
| 3.2 Product evaluation and re-evaluation activities | Evaluation – Needs minor improvement | There is a good, systematic evaluation process; however, there should be better performance measurement and monitoring. | Refer to rec. 6 |
| Re-evaluation – Needs moderate improvement | Re-evaluation lacks a systematic process and performance data, and is not integrated into the electronic system. | 3, 4 | |
| 3.3 Cost recovery | Needs minor improvement | The cost-recovery regime is being updated and a renewed time tracking tool is being implemented. | Refer to recs 2 and 6 |
| 3.4 Compliance and enforcement | Needs moderate improvement | Overall, the national compliance and enforcement plan is good. However, a strategy for front-line electronic tools should be developed to support compliance and enforcement. | 5 |
| 3.5 Outreach activities | Needs minor improvement | An national outreach and stakeholder engagement strategy should be developed. | Refer to rec. 1 |
| 3.6 International collaboration | Satisfactory | International collaboration activities support the Program’s objectives. | |
| 3.7 Performance management framework | Needs moderate improvement | The Program would benefit from improving performance measurement and monitoring of key regulatory activities and developing a program-wide logic model. | 6 |
Appendix C - Estimated resources
The following table provides a summary of the key PMRA and RAPB organizations and estimated resources involved in the delivery of Pesticide Regulatory Program activities for 2013-14. These figures are unaudited and are provided for illustration purposes.
| Organization | Activities | ||||
|---|---|---|---|---|---|
| Evaluation of new products | Re-evaluation of products | Compliance and enforcement | Program management and support | Total | |
| Expenditures for 2013-14 ($000) | |||||
| PMRA | |||||
| Executive Director’s Office (EDO) | $0 | $0 | $0 | $580 | $580 |
| Registration Directorate (RD) | $5,855 | $508 | $0 | $1,239 | $7,602 |
| Health Evaluation Directorate (HED) | $6,064 | $3,865 | $23 | $923 | $10,875 |
| Environmental Assessment Directorate (EAD) | $3,132 | $1,498 | $0 | $922 | $5,552 |
| Value Assessment and Re-evaluation Management Directorate (VRMD) | $3,739 | $2,941 | $24 | $888 | $7,592 |
| Compliance, Laboratory Services and Regional Operations Directorate (CLSROD) | $0 | $0 | $2,674 | $352 | $3,026 |
| Policy, Communications and Regulatory Affairs Directorate (PCRAD) | $11 | $11 | $0 | $4,793 | $4,815 |
| Strategic Planning, Financial and Business Operations Division (SPFBOD) | $1 | $0 | $0 | $2,952 | $2,953 |
| RAPB | |||||
| BC Region | $0 | $0 | $661 | $0 | $661 |
| Prairie Region | $0 | $0 | $1,126 | $0 | $1,126 |
| Ontario Region | $0 | $0 | $827 | $0 | $827 |
| Quebec Region | $0 | $0 | $659 | $0 | $659 |
| Atlantic Region | $0 | $0 | $571 | $0 | $571 |
| Total | $18,802 | $8,823 | $6,564 | $12,649 | $46,838 |
| % | 40% | 19% | 14% | 27% | 100% |
Appendix D - Submission categories
In keeping with the Revised Management of Submissions Policy, depending on the purpose of the application and the type of information required, every submission is assigned to one of the five following categories.
Category A
- New active ingredients or integrated system products, their related end-use products, and manufacturing-use products;
- Major new use of registered pest control products (defined as the addition of a new use site category to the use pattern for a specific registered active ingredient);
- Specification of import maximum residue limits (MRL) for an unregistered active ingredient.
Category B
- New pest control products containing registered active ingredients;
- Amendment to existing pest control products (for example, product chemistry, labelling);
- Conversion or renewal of conditional registration;
- Emergency registration;
- The addition of import MRLs for previously assessed active ingredients.
Category C
- Product registrations and amendments with no data requirements. These applications involve minor label or formulation reviews, such as product registration based on registered precedent products.
Category D
- Submissions within particular programs including:
- Import for Manufacture and Export Program (IMEP);
- Own Use Import (OUI);
- Grower Requested Own Use (GROU) Equivalency and import permits;
- User Requested Minor Use Label Expansion (URMULE);
- Registration Renewal;
- Discontinuations.
Category E
- Research authorizations for new active ingredients and new use(s) of registered active ingredients;
- Research notification for research carried out in Canada.
Appendix E - Compliance and enforcement statistics
| Activity | 2011-12 | 2012-13 | 2013-14 | Total |
|---|---|---|---|---|
| Education - written | 419 | 413 | 432 | 1,264 |
| Education - verbal | 104 | 124 | 103 | 331 |
| Enforcement letter | 46 | 38 | 62 | 146 |
| Compliance order and administrative monetary penalty (AMP) pursued | 2 | 1 | 0 | 3 |
| Compliance order (CO) | 1 | 6 | 1 | 8 |
| Notice of violation with warning issued | 0 | 0 | 1 | 1 |
| Notice of violation with penalty issued | 20 | 13 | 5 | 38 |
| Total | 592 | 595 | 604 | 1,791 |
| Penalty amount | $80,800 | $53,700 | $20,000 | $154,500 |
| Payment received | $40,400 | $26,850 | $6,820 | $74,070 |
| Action | 2011-12 | 2012-13 | 2013-14 | Total |
|---|---|---|---|---|
| Label modification recommended | 26 | 17 | 21 | 64 |
| Denial of entry | 63 | 123 | 159 | 345 |
| Denial of entry and border lockout | 3 | 2 | 0 | 5 |
| Seizure and detention | 1 | 0 | 0 | 1 |
| Forfeiture by consent | 18 | 5 | 4 | 27 |
| Requested to re-label product | 27 | 36 | 39 | 102 |
| Requested to cease activity/remove product | 319 | 290 | 402 | 1,011 |
| Ordered to cease activity/remove product | 35 | 82 | 39 | 156 |
| Ordered to re-label product | 0 | 2 | 2 | 4 |
| Total | 492 | 557 | 666 | 1,715 |
Page details
- Date modified: