Adverse Events Following Immunization (AEFI) Quarterly Report for 2014 – Q4
Safety Assessment Summary for this Quarter:
- No significant vaccine safety concerns were identified in quarter 4 of 2014.
- All serious cases were reviewed and most found either to be expected (based on known vaccine-related adverse reactions) or to have alternate explanations, such as community acquired infection; exposures to other agents, such as antibiotics; or pre-existing problems like cardiac disease.
- As in previous quarters the total count of AEFI reports received was lower than that seen in previous quarters reflecting a gap in reporting from jurisdictions that are implementing new electronic reporting systems. We expect an increase in reports received in subsequent quarters to balance this gap.
- Two previously identified concerns in past quarterly reports were examined in more detail for the entire period of 2011 through the end of 2014 with the conclusion that:
- Rotavirus vaccines used in Canada from 2011 through the end of 2014 have an excellent safety profile with no unexpected concerns.
- The increase in reporting of Kawasaki syndrome, as noted in the Q3 report was related to initiation of systematic syndromic surveillance for vasculitis syndromes, including Kawasaki syndrome, by IMPACT in 2013. The overall frequency remained consistent with the known background rate of disease in the pre-school aged population and there was nothing to suggest a vaccine-specific concern.
Vaccines are closely monitored in Canada at all phases of the vaccine product 'life cycle' from discovery through market authorization (pre-market) and beyond, as people begin using them (post-market). Many stakeholders are involved in various activities related to vaccine safety assessment and monitoring including the federal government, provincial, territorial and local public health authorities, health care providers, vaccine industry and the public. The Public Health Agency of Canada (the Agency) conducts vigilant post-market safety surveillance through a national reporting system, the Canadian Adverse Events Following Immunization Surveillance System (CAEFISS).
The primary purpose of vaccine post market surveillance is to detect safety concerns. These concerns include a possible increase in the severity or frequency of expected reactions, or occurrence of one or more unexpected events (i.e. an event that is not consistent with Canadian product information or labelling). This allows immunization providers and public health immunization program providers to take public health action at the level of the:
- individual (e.g. further investigation to confirm a diagnosis and determine possible causes, consultation to rule out allergy to one or more vaccine components, or evaluate whether or not to give subsequent doses of a vaccine), and/or
- immunization program (e.g. investigation of a cluster of adverse events, review of procedures to ensure that vaccine storage requirements have been strictly followed, or consider a change in policy to adopt a less reactogenic vaccine).
The Agency also shares adverse event following immunization (AEFI) data with Health Canada, the vaccine regulator in Canada, to enable regulatory action related to vaccines marketed in Canada. These actions may include issuing communications to immunization providers or the public regarding the safety concern or requiring additional information or investigation by the vaccine distributor.
Regular vaccine safety surveillance quarterly reports summarizing CAEFISS data were initiated by the Agency in 2014. The first three Quarterly Reports, summarized all reports of adverse events following immunization (AEFI) received by the Agency in 2014 from Jan 1st to March 31st (Quarter 1), Apr 1st to June 30th (Quarter 2) and July 1st to Sept 30th, regardless of the date the vaccine was actually given. This Quarter 4 Report provides a similar summary for all reports received from Oct 1st to Dec 31st as well as cumulative data spanning the entire year of 2014. This provides a focus on each individual quarter but also tracks the trends as Quarterly Reports accumulate during the calendar year.
Both the current (Q4) and cumulative (all 2014) quarterly data are compared to an average of all reports received during the same periods of time (Quarter4 alone and cumulative from Jan 1st to Dec 31st for the average of the last three years (2011- 2013).
Notes on Interpretation: An AEFI is defined as "any untoward medical occurrence which follows immunization and which does not necessarily have a causal relationship with the usage of the vaccine. The adverse event may be any unfavorable or unintended sign, abnormal laboratory finding, symptom or disease". AEFI reports submitted to the Public Health Agency of Canada represent a suspicion, opinion or observation by the reporter as opposed to an assertion or proof that the vaccine may have caused the event. For most events a clear causal relationship is difficult to establish. Additional limitations to AEFI report data include varied reporting standards and underreporting, lack of certainty regarding the diagnostic validity of a reported event, missing information regarding other potential causes and other reporting biases. In short, this information should not be used to estimate the incidence of adverse events..
Results Highlighted for Quarter 4 of 2014
Cumulative counts included in this Quarterly Report (Q4) include reports received from October 1st to December 31st, 2014 and comparisons are made to the average number of reports received in the same quarter over the previous three calendar years (2011-2013). The reports analysed for 2014 were extracted from the CAEFISS database on 18-February-2015 and for 2011-2013 on 29-April-2014 by the Public Health Agency of Canada (the Agency).
Figure 1: Total AEFI Reports by calendar quarter and cumulative to end of each Quarter: 2014 vs. average for 2011-2013
A total of 691 AEFI reports were received by the Agency in Q4 of 2014. During the same periods for 2011, 2012 and 2013, the Agency received an average of 971 (range: 610-1288) AEFI reports. The lower reporting totals for Q4 in 2014 relative to previous years continues a trend seen in previous quarters of 2014. The cumulative total for the entire year of 2014 was 2408 reports compared to an average of 3519 for prior years (range 3167-4012). The main reason for the drop in 2014 was delayed report transmission from one or more provinces and territories to the Agency, for technical reasons relatedto implementing a new electronic AEFI reporting system. While reports have not yet been received by the Agency, they are reviewed and analysed in the reporting jurisdictions and any concerns communicated via the Vaccine Vigilance Working Group. There were none noted in 2014 and it is expected that the reports will be sent in the first or second quarter of 2015.

Text Equivalent
This is a combination bar and line graph. The bars show the total number of AEFI reports received each calendar quarter(Q) with Q1 encompassing January, February and March, Q2 April, May and Jun, Q3 July, August and September and Q4 October, November and December. The line shows the accumulated total reports to the end of each quarter. The primary y-axis on the left hand side of the graph is for the bar graph and shows the total count for each quarter, ranging from 0-1200. The secondary y-axis on the right hand side of the graph is for the line graph and shows the accumulated total count to the end of each quarter, ranging from 0 to 4000. The x-axis is quarter and ranges from Q1 to Q4. Two time periods are depicted on the graph.
The light purple bars shows the average total reports each quarter for 2011, 2012 and 2013: 1049 in Q1, 841 in Q2, 658 in Q3 and 971 in Q4. The dotted line shows the average cumulative total reports received by the end of each quarter for the same three years: 1890 by the end of Q2, 2548 by the end of Q3 and 3519 by the end of Q4.
The dark hatched purple bars show the total reports received each quarter for 2014: 760 in Q1, 455 in Q2, 502 in Q3 and 691 in Q4. The solid line shows that a cumulative total of 2408 reports were received by the end of Q4, 2014.
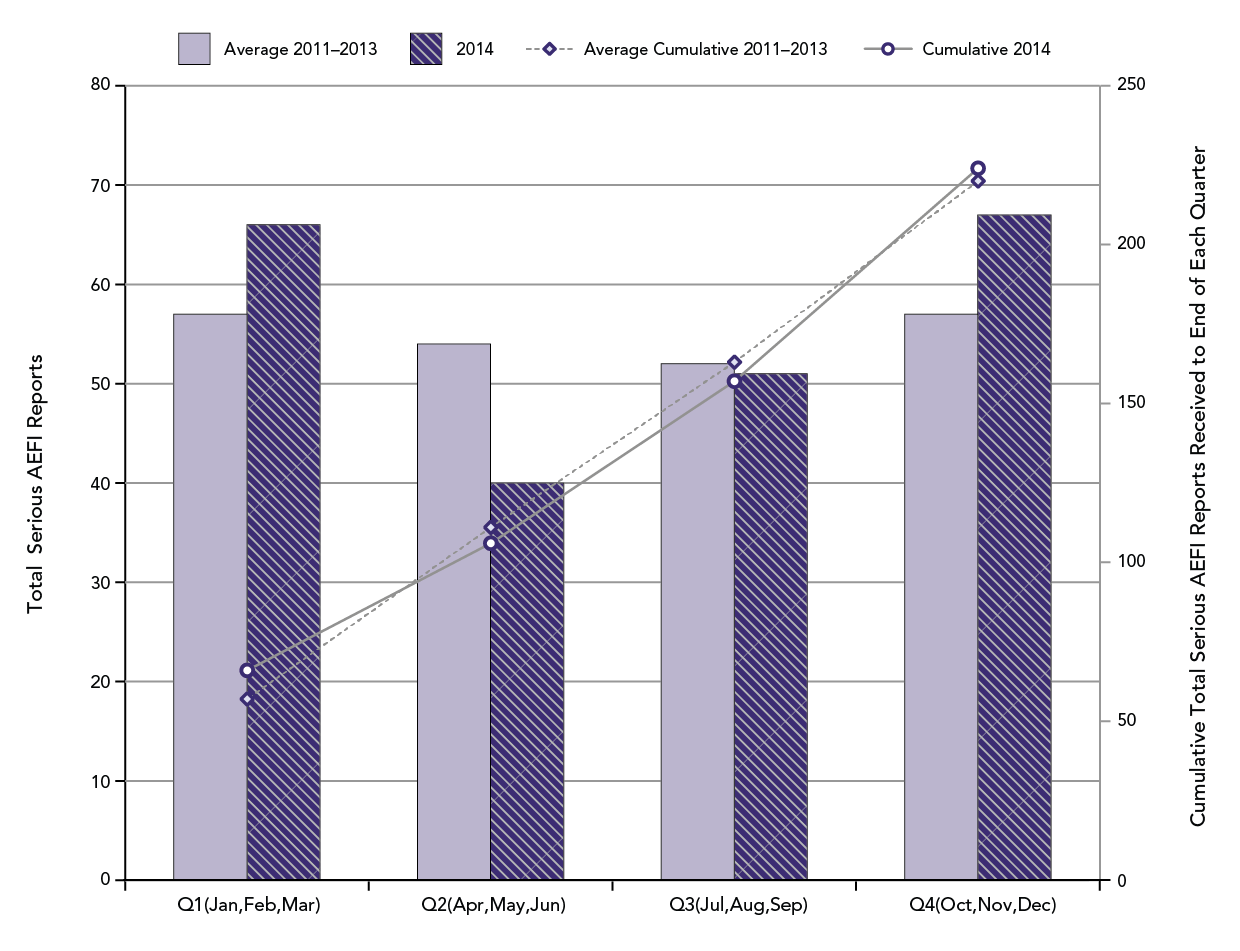
Figure 2: Total serious AEFI reports by calendar quarter and cumulative to end of each quarter: 2014 vs. average for 2011-2013.
A total of 67 AEFI reports received by the Agency in Q4 of 2014 were serious (9.7%% of all AEFI reports). During the same periods for 2011, 2012 and 2013, the Agency received an average of 57 (range: 38-75) serious AEFI reports representing from 5.7-6.2% of all AEFI reports received in the same quarter for these years. The cumulative total of 224 serious reports for all of 2014 was very similar to the average yearly count of 220 serious reports for 2011 – 2013 (range: 186-255).
Text Equivalent
This is a combination bar and line graph. The bars show the total number of serious AEFI reports received each calendar quarter(Q) with Q1 encompassing January, February and March, Q2 April, May and Jun, Q3 July, August and September and Q4 October, November and December. The line shows the accumulated total reports to the end of each quarter. The primary y-axis on the left hand side of the graph is for the bar graph and shows the total count for each quarter, ranging from 0-80. The secondary y-axis on the right hand side of the graph is for the line graph and shows the total count to the end of each quarter, ranging from 0 to 250. The x-axis is quarter and ranges from Q1 to Q4. Two time periods are depicted on the graph.
The light purple bars show the average total reports each quarter for 2011, 2012 and 2013: 57 in Q1, 54 in Q2, 52 in Q3 and 57 in Q4. The dotted line shows the average cumulative total reports received by the end of each quarter for the same three years: 111by the end of Q2, 163 by the end of Q3 and 220 by the end of Q4.
The dark hatched purple bars show the total reports received each quarter for 2014: 66 in Q1, 40 in Q2, 51 in Q3 and 67 in Q4. The solid line shows that a cumulative total of 224 reports were received by the end of Q4, 2014.
Frequency of serious and non-serious AEFI Reports by Age Group
There were a few changes in age distribution for serious AEFI reports received in Q4 for 2014 relative to previous years. Most notably, as shown in Table 1, the proportion of all serious reports for children under 1 year of age was higher in 2014 than previous years (31.3% versus 22.4%). This change was most likely due to an increase in serious reports following the administration of one or more of rota, DTaP-IPV-HB-Hib or pneu-C containing vaccines (see Table 3 below). Additionally the proportion of reports for 18 to <65 years was lower (7.5% versus 17.2%) but the absolute number of reports was small for both time periods (4 versus 10). Table 2 shows the distribution for reports received from January 1 to the end of Q4.For all of 2014 relative to previous years the distribution of serious reports was higher among children aged <1 year (34.8 versus 27.7%) but lower among children aged 1 to <2 years (26.1 versus 31.4%).
For non-serious AEFI reports, the age group distribution was similar in 2014 to previous years for Q4. For all of 2014 relative to previous years the proportion of non-serious reports dropped for children aged 1 to <2 years (10.5% versus 17.6%) and increased for 18-<65 year olds (35.7 versus 30.6%).
| Age Group | Serious Adverse Events (SAE) | Non-serious Adverse Events (non-SAE) | ||
|---|---|---|---|---|
| 2014 (% total) |
Average for 2011-2013 (% total) | 2014 (% total) |
Average for 2011-2013 (% total) | |
| Unknown | 1 (1.5) | 2 (3.4) | 3 (0.5) | 24 (2.6) |
| 65+ years | 3 (3.0) | 5 (8.6) | 76 (12.2) | 114 (12.5) |
| 18-<65 years | 4 (7.5) | 10 (17.2) | 246 (39.4) | 331 (36.2) |
| 7-<18 years | 4 (7.5) | 4 (6.9) | 101 (16.2) | 120 (13.1) |
| 2-<7 years | 10 (13.4) | 7 (12.1) | 60 (9.6) | 114 (12.5) |
| 1-<2 years | 17 (22.4) | 17 (29.3) | 61 (9.8) | 116 (12.7) |
| 0-<1 year | 28 (31.3) | 13 (22.4) | 77 (12.3) | 95 (10.4) |
| Age Group | Serious Adverse Events (SAE) | Non-serious Adverse Events (non-SAE) | ||
|---|---|---|---|---|
| 2014 (% total) |
Average for 2011-2013 (% total) | 2014 (% total) |
Average for 2011-2013 (% total) | |
| Unknown | 3 (1.3) | 2 (0.9) | 23 (1.1) | 81 (2.5) |
| 65+ years | 11 (4.91) | 18 (8.2) | 229 (10.5) | 279 (8.5) |
| 18-<65 years | 31 (13.8) | 26 (11.8) | 780 (35.7) | 1008 (30.6) |
| 7-<18 years | 16 (7.1) | 20 (9.1) | 388 (17.8) | 446 (13.5) |
| 2-<7 years | 26 (11.6) | 25 (11.4) | 270 (10.5) | 445 (13.5) |
| 1-<2 years | 59 (26.1) | 69 (31.4) | 230 (10.5) | 579 (17.6) |
| 0-<1 year | 78 (34.8) | 61 (27.7) | 264 (12.1) | 460 (13.9) |
Most common vaccines identified in Adverse Events Following Immunization (AEFI) Reports
| Number of Serious Adverse Event (SAE) Reports Received | Number of Non-Serious Adverse Event(non-SAE) Reports Received | |||
|---|---|---|---|---|
| 2014 | Average for 2011-2013 |
2014 | Average for 2011-2013 |
|
| Rota | 18 | 4 | 30 | 29 |
| DTaP-IPV-Hib | 13 | 14 | 52 | 99 |
| DTaP-HB-IPV-Hib | 17 | 5 | 26 | 31 |
| Pneu-C | 28 | 21 | 77 | 134 |
| Men-C | 14 | 16 | 63 | 105 |
| MMR | 6 | 9 | 37 | 92 |
| MMR-Var | 9 | 6 | 28 | 28 |
| Var | 2 | 4 | 26 | 66 |
| DTaP-IPV | 0 | 2 | 18 | 28 |
| Tdap-IPV | 0 | 1 | 8 | 15 |
| HPV | 1 | 3 | 28 | 47 |
| HB | 4 | 1 | 15 | 26 |
| Tdap | 0 | <1 | 54 | 59 |
| Inf | 7 | 17 | 224 | 386 |
| Zos | 1 | 1 | 25 | 17 |
| Pneu-P | 2 | 3 | 60 | 69 |
Table 3 lists the most commonly implicated vaccines among AEFI reports received for Q4 of 2014 relative to the 2011-2013 average reports received for the same quarter. The most notable change in the 4th quarter of 2014 relative to previous years was a higher number of serious AEFI reports following rotavirus vaccine (18 versus 4). This trend has been noted in previous quarterly reports, with an increase in intussusception reports in Q2 and Kawasaki syndrome in Q3. In contrast for Q4 there were no reports of intussusception and only 2 reports of Kawasaki syndrome. The spectrum of events reported was broad with no single type of event accounting for the observed increase in number.
The only other notable change was a drop in serious reports following influenza vaccine in Q4 of 2014 (total of 7 reports) relative to the average observed for Q4 from 2011-2013 (17 reports).
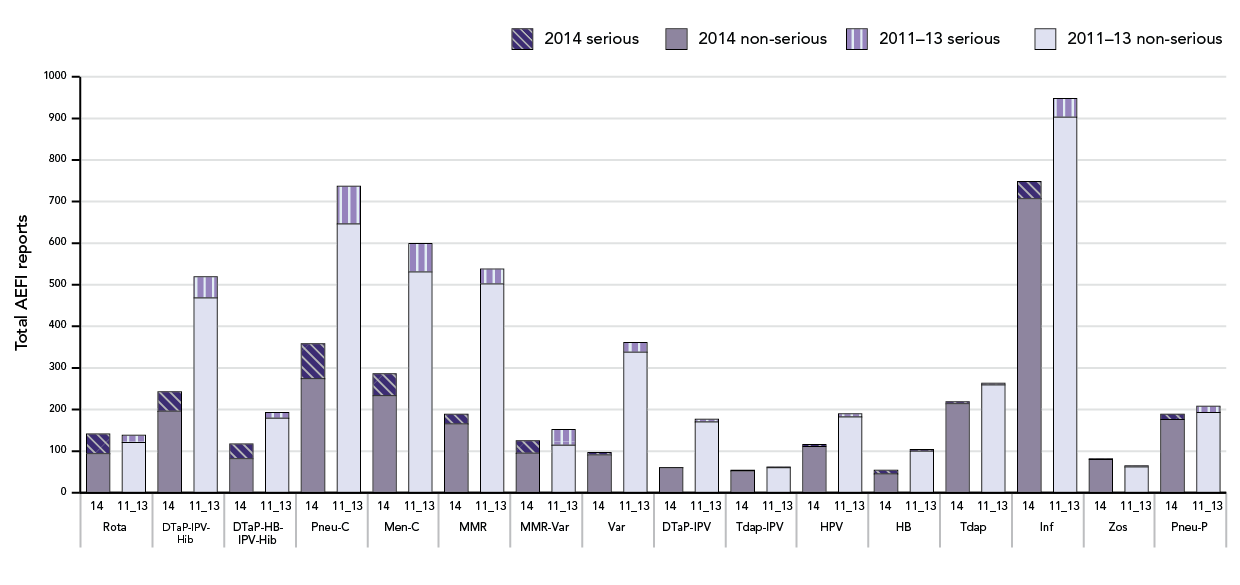
Figure 3 provides a cumulative summary of the most commonly implicated vaccines among AEFI reports received by the Agency throughout 2014 (January 1st to December 31st) compared to the average in 2011, 2012 and 2013 for the same time frame. As already noted in previous quarterly reports for 2014 there was a trend for a lower frequency of non-serious adverse event reports for most commonly administered childhood vaccines (DTaP-IPV-Hib, DTap-HB-IPV-Hib, Pneu-C, Men-C, MMR, Var and DTaP-IPV) relative to the previous three years. The reason for the lower numbers for childhood vaccines (as opposed to those given to adults) is primarily accounted for by delays in reporting from jurisdictions initiating new electronic AEFI reporting infrastructure. These will be captured in subsequent quarterly reports. In contrast the total serious AEFI reporting numbers are relatively unchanged for childhood vaccines, reflecting the significance of the contribution by the IMPACT active syndromic surveillance.
Figure 3: Most common vaccines identified in AEFIs reported from January 1st to end of 4th Quarter 2014 vs. average for 2011-2013.
Text Equivalent
This is a bar graph showing total numbers of non-serious and serious AEFI reports received for specific vaccines at the Public Health Agency to the end of the fourth quarter (December 31) of 2014 versus the average of reports received to the end of the fourth quarters of 2011, 2012 and 2013. The y-axis shows the number of reports, ranging from 0 to 1000. The x-axis shows specific vaccine groups based on the antigenic content rather than specific brand.
The vaccine groups shown and the number of non-serious/serious AEFI reports received for each to the end of the 4th quarter in a) 2014 versus (mauve bar for non-serious, dark-purple hatched bar for serious) b) the average for 2011, 2012 and 2013 (light purple bar for non-serious, vertical lined lilac bar for serious) respectively are: Rotavirus: a) 94/47 versus b) 121/17, DTaP-IPV-Hib: a) 196/47 versus b) 468/51; DTaP-HB-IPV-Hib: a) 82/35versus b) 179/14; pneumococcal conjugate: a) 274/84 versus b) 646/91; meningococcal conjugate C: a) 233/53 versus b) 531/68; MMR: a) 165/24 versus b) 502/36; MMRV: a) 95/30 versus b) 114/38; Varicella: a) 91/6 versus b) 338/23; DTaP-IPV: a) 60/1 versus b) 170/7; Tdap-IPV: a) 52/2 versus b) 60/2; HPV: a) 111/5 versus b) 182/8; HB: a) 45/9 versus b) 100/4; Tdap: a) 214/5 versus b) 259/4; Influenza: a) 707/41 versus b) 903/45; Zoster: a) 80/2 versus b) 62/3; Pneumococcal polysaccharide: a) 176/13 versus b) 193/15.
Adverse Events Following Immunization (AEFI) Reported by Main Reason and Seriousness
Classification of AEFIs for Report Purposes: all AEFI reports are reviewed daily by health professionals to assign a main reason for reporting and the associated severity. The classification adheres closely to the national AEFI reporting form in order to provide meaningful feedback to public health authorities, health professionals and the public regarding the adverse event reporting profile. The main categories used when reporting an AEFI, and those reflected on the graph below, are defined as:
- Reactions at or near the vaccination site: most reported events involve inflammation at or near the site of vaccination. Further details can be found in section 9(a) of the reporting form
- Allergic or Allergic-like Events: the most severe allergic event, anaphylaxis, is rare (≤1 episode per 100,000 doses administered). Further details can be found in section 9(b) of the reporting form.
- Neurologic events: seizures are the most commonly reported neurologic event especially in infants and toddlers who are prone to have seizures in association with fever. Such events are reported in Canada relatively frequently because they are actively sought and reported by the IMP-ACT network. Further details can be found in section 9(c) of the reporting form.
- Rash only: to be classified as 'rash only', the rash is the only sign or symptom reported. This would include hives that onset any time after immunization but are accompanied by no other signs or symptoms. Hives are a frequent occurrence in childhood and a cause is often not found.
- Other events specified on the CAEFISS report form: this could include other events such as persistent crying (continuous or unaltered crying ≥ 3 hours, intussusception, hypotonic-hyporesponsive episodes (HHE). Further details can be found in section 9(d) of the reporting form.
Events not clearly falling into one of the categories listed above are classified into one of four additional categories:
- Systemic events: these are primarily events involving many body systems often accompanied by fever. Any recognized syndromes are classified as systemic events and include such illnesses as Kawasaki syndrome, Steven-Johnson syndrome, Henoch-Schonlein purpura, fibromyalgia and serum sickness. In addition evidence for infection of one or more body parts (respiratory infection, bladder infection, etc.) are included in this category as are instances of rash that is non-allergic in origin and is accompanied by other signs and symptoms such as fever, cough and conjunctivitis. General symptoms such as fatigue, malaise, lethargy, headache, myalgias including influenza-like illnesses are classified as systemic events. Finally fever as the only adverse event reported is included in this category.
- Vaccination anxiety related events: this includes fainting (syncope) with or without seizure-like movements which may briefly accompany a fainting episode. Other events included in this category are clusters of events often seen within minutes of immunization indicative of anxiety such as pallor, shakiness, feeling cold, hyperventilation, etc.
- Miscellaneous other events: all other adverse events that don't fit into any of the above categories are captured as miscellaneous events. These may be further categorized by the predominant body system they fall under such as gastrointestinal, cardiac, genitourinary, etc.
- Vaccination error without an associated AEFI: on occasion reports are submitted of an error in immunization without any associated adverse event. Examples include use of an expired product or incorrect administration of a product (e.g. giving a vaccine into the muscle when it should have been injected subcutaneously).
The main reason for reporting serious (SAE) and non-serious (non-SAE) AEFI for Q4 of 2014 compared to the 2011-2013 average is shown in Table 4. The numbers of serious AEFI reports were roughly comparable in 2014 relative to the average for 2011-2013 in Q4.
| Main Reason for Reporting | Number of Serious Adverse Event (SAE) Reports | Number of Non-serious Adverse Event (non-SAE) Reports | ||
|---|---|---|---|---|
| 2014 | Average for 2011-2013 | 2014 | Average for 2011-2013 | |
| Reaction at or near the vaccination site | 7 | 7 | 241 | 342 |
| Allergic or allergic-like events | 3 | 3 | 97 | 149 |
| Neurologic events | 16 | 18 | 15 | 25 |
| Rash only | 2 | 1 | 117 | 182 |
| Other events specified on the CAEFISS form | 8 | 12 | 31 | 32 |
| Systemic events | 17 | 13 | 66 | 88 |
| Vaccination anxiety related events | 0 | 0 | 14 | 12 |
| Miscellaneous other events | 14 | 8 | 39 | 74 |
| Vaccination error without an associated AEFI | 0 | 0 | 4 | 21 |
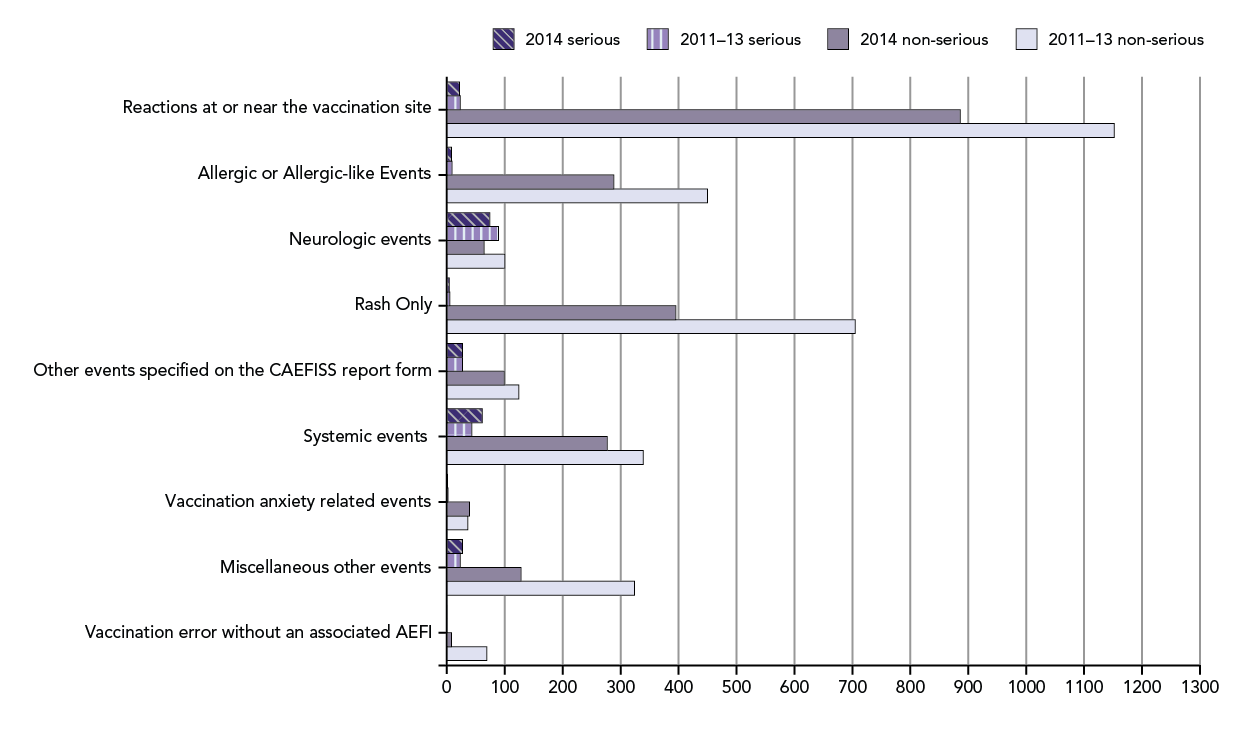
Figure 4 provides a cumulative summary of the same data for reports received during the entire year (January 1st to December 31st in 2014 compared to the average in 2011-2013. As noted in previous quarterly reports the total numbers of non-SAE reports were less in 2014 than previous years, but the distribution by main reason for reporting was similar. The most frequently reported non-SAE were reactions at or near the vaccination site, rash as the only event, allergic or allergic like events and systemic events.
Figure 4: Total AEFI reports received from January 1st to end of 4th Quarter by main reason for reporting and seriousness: 2014 vs. average for 2011-2013
Text Equivalent
This is a bar graph showing the total number of non-serious and serious reports categorized by the main reason for reporting received by the Public Health Agency of Canada to the end of the 4th quarter (December 31st) of 2014 (mauve bar for non-serious; diagonal hatched dark purple bar for serious) and the average of 2011, 2012 and 2013 for the same period (light purple bar for non-serious; vertical hatched lilac bar for serious).
The y-axis shows the 9 different categories of AEFI and the x-axis shows the total number of reports for each category ranging from 0 to 1300.
The respective totals for non-serious/ serious AEFI reports received by the end of Q4 were: reactions at or near the vaccination site: 2014: 886/22; 2011-13: 1152/23; allergic or allergic-like events: 2014: 288/8; 2011-13: 450/9; neurologic events: 2014: 64/74; 2011-13: 100/89; rash only (in other words rash without any other signs or symptoms): 2014: 395/4; 2011-2013: 705/5; Other events specified on CAEFISS report form: 2014: 99/27; 2011-13: 124/27; systemic events: 2014: 277/61; 2011-13: 339/43; vaccination anxiety related events: 2014: 39/1; 2011-13: 36/2; miscellaneous other events: 2014: 128/27; 2011-13: 324/23; vaccination error without an associated AEFI: 2014: 8/0; 2011-13: 69/0.
Analysis of Serious Adverse Events Following Immunization (SAE) Reports received by the Agency
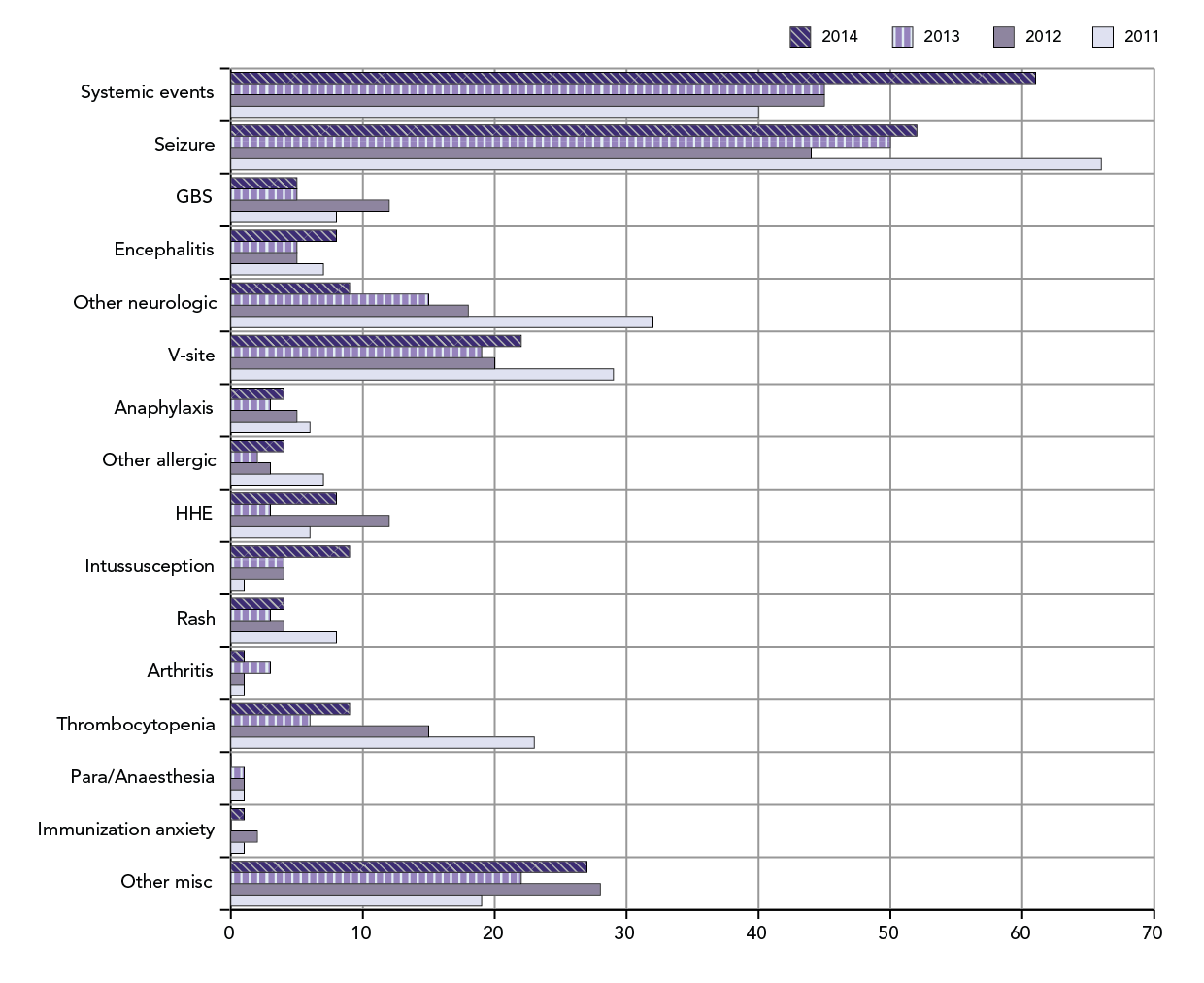
The type of serious adverse events (SAE) reported are described below. Table 5 summarizes the type of SAE reported in Q4 of 2014 compared to the 2011-2013 average. Figure 5 provides a cumulative summary of the same data collected for the entire year (Jan 1st - Dec 31st) in 2014 compared to the average in 2011-2013.
There were a total of 67 SAE reports in Q4 of 2014 (9.7% of 692 reports total). The average for Q4 from 2011 to 2013 was 57 serious reports (5.9% of 971 reports total). As noted in all previous quarterly reports for 2014, the single most common reason for reporting SAEs in Q4 were seizures usually accompanied by fever. Febrile seizures are actively sought by the pediatric hospital-based Immunization Monitoring Program, ACTive (IMPACT) network which regularly provides over half of all serious AEFI reported to PHAC for children. Febrile seizures are known to occur after immunization with live attenuated vaccines such as MMR or MMRV as often as once for every 2600 vaccinations given. The events are short-lived and not associated with long term adverse outcomes. The SAE report profile for Q4 2014 was not dissimilar to what has been seen in the same quarter in past years allowing for some small number variation.
| Types of SAE reports | 2011 | 2012 | 2013 | 2014 |
|---|---|---|---|---|
| Systemic events | 13 | 13 | 12 | 17 |
| Seizure | 12 | 6 | 7 | 13 |
| GBS | 5 | 3 | 2 | 0 |
| Encephalitis | 2 | 2 | 0 | 2 |
| Other neurologic | 5 | 6 | 5 | 1 |
| V-site | 12 | 6 | 2 | 7 |
| Anaphylaxis | 1 | 3 | 0 | 1 |
| Other allergic | 4 | 0 | 0 | 2 |
| HHE | 2 | 2 | 1 | 6 |
| Intussusception | 1 | 1 | 0 | 0 |
| Rash | 1 | 0 | 0 | 2 |
| Arthritis | 0 | 1 | 1 | 0 |
| Thrombocytopenia | 7 | 2 | 0 | 2 |
| Para/Anaesthesia | 1 | 0 | 0 | 0 |
| Immunization anxiety | 0 | 1 | 0 | 0 |
| Other misc | 7 | 12 | 6 | 14 |
Figure 5: Total serious AEFI reports received from January 1st to end of 4th Quarter by type of adverse event: 2014 vs. prior 3 years
Text Equivalent
This is a bar graph showing the total serious reports of specific AEFIs received to the end of the 4th quarter (December 31st) for each of four different years: 2011, 2012, 2013 and 2014. The y-axis lists the specific AEFI types as described below. The x-axis shows the total number of reports ranging from 0-70.
For each specific AEFI type the total reports received, respectively to the end of Q4 for 2011(mauve bar), 2012(dark purple bar), 2013(vertical hatched lilac bar) and 2014(diagonal hatched dark purple bar) were: systemic events: 40, 45, 45 and 61; seizure: 66, 44, 50 and 52 ; Guillain-Barre syndrome: 8, 12, 5 and 5 ; encephalitis: 7, 5, 5 and 8; other neurologic events: 32, 18, 15 and 9 ; reactions at or near the vaccination site: 29, 20, 19 and 22; anaphylaxis: 6, 5, 3 and 4 ; other allergic events: 7, 3, 2 and 4; hypotonic-hyporesponsive episodes: 6, 12, 3 and 8; intussusception: 1, 4, 4 and 9; rash: 8, 4, 3 and 4; arthritis: 1, 1, 3 and 1; thrombocytopenia: 23, 15, 6 and 9 ; Para/Anaesthesia: 1, 1, 1 and 0 ; immunization related anxiety: 1, 2, 0 and 1; other miscellaneous events: 19, 28, 22 and 27.
Spotlight on Vaccine Safety Concerns Identified in 2014 Quarterly Reports
Throughout 2014 there were only two possible vaccine safety issues identified in Quarterly reports. In Q2 there was an apparent increase relative to previous years in the frequency of reports of intussusception following rotavirus vaccine (5 in 2014 versus 0-2 in 2011, 2012 and 2013). In Q3 an increase in the frequency of reports of Kawasaki syndrome was noted (15 in 2014 vs 1-8 in 2011, 2012 and 2013). Neither trend was observed in Q4 for 2014. However, as this Q4 report contains information on reports received for the entire year of 2014 and the comparison years (2011-2013), summary data are presented below that span the entire period of 2011-2014 to enable a more comprehensive examination of reporting trends.
1. Rotavirus Vaccine Safety Profile: 2011-2014
Two rotavirus vaccines are licensed for use in Canada: Rotateq™ (Merck) since 2006 and Rotarix™ (GSK) since 2007. Publicly funded rotavirus vaccine programs were initiated in 2011 (British Columbia, Ontario, Quebec, Prince Edward Island), and expanded to include Saskatchewan and Yukon Territory in 2012 and Alberta and Manitoba in 2014. The dynamic nature of program introduction in Canada by jurisdiction impacts on reporting rates and thus complicates temporal comparisons. This clearly played a role for rotavirus vaccines because 2014 reporting frequency was being compared to an average for 2011-2013 when programs were not yet initiated or just being introduced.
Other key factors that influenced the quarterly reporting profiles for rotavirus vaccine include the presence of active surveillance by the IMPACT network and technical issues which have prevented jurisdictional reporting throughout 2014 and part of 2013. Table 6 shows the annual reporting frequency from 2011 through 2014 of both serious and non-serious adverse events involving rotavirus vaccines by reporting source.
| Reporting source | 2011 | 2012 | 2013 | 2014 | All Years | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| SAE | Non-SAE | SAE | Non-SAE | SAE | Non-SAE | SAE | Non-SAE | SAE | Non-SAE | |
| Public Health | 0 | 19 | 13 | 171 | 15 | 165 | 18 | 95 | 46 | 450 |
| IMPACT | 3 | 0 | 6 | 2 | 13 | 1 | 30 | 0 | 52 | 3 |
| Market authorization holder | 1 | 3 | 0 | 1 | 0 | 0 | 0 | 0 | 1 | 4 |
| Total | 4 | 22 | 19 | 174 | 28 | 166 | 48 | 95 | 99 | 457 |
| % serious | 15.4% | 9.8% | 14.4% | 33.6% | 17.8% | |||||
The increase in the total number of serious reports over time from 4 in 2011 to 48 in 2014 is expected given the rapid expansion in publicly funded programs and the presence of the IMPACT pediatric hospital based network which is actively looking for and reporting selected events including seizures, neurologic events and intussusception. However, the apparent increase in the proportion of serious reports is misleading and largely due to a drop in the non-serious reports from jurisdictions having technical difficulties in transferring reports to PHAC – as has been pointed out in all 2014 quarterly reports.
Table 7 shows the distribution of serious and non-serious reports classified by the primary reason for reporting along with an approximated reporting rate based on doses distributed. From 2011 to the end of 2014 over 2 million doses of rotavirus vaccine were distributed in Canada. During that time period a total of 555 AEFI reports were received for an overall reporting rate of 27.5/100,000 doses distributed. There were 99 serious reports (17.8% of all reports) for a serious AEFI reporting rate of 4.9/100,000 doses distributed. For intussusception, the reporting rate of 0.9 cases per 100,000 doses distributed is similar to what has been observed from postmarketing studies in other countries. Fifteen intussusception cases occurred within 21 days of immunization (reporting rate of 0.74/100000 doses distributed) and the remaining four onset from 42 to 155 days and thus were unlikely related to rotavirus vaccination.
| Primary Reason for Reporting | Total AEFI Reports |
Reporting rate/100,000 doses distributed |
% one vaccine |
Total Serious Reports (% all AEFI) |
|---|---|---|---|---|
| Vaccination site reaction | 27 | 1.30 | 3.7% | 0 |
| Anaphylaxis | 7 | 0.31 | 28.6% | 1 (14.3%) |
| Other Allergic event | 22 | 1.1 | 18.2% | 0 |
| Seizure | 18 | 0.9 | 0 | 13 (72.2%) |
| Other neurologic event | 6 | 0.3 | 0 | 5 (83.3%) |
| Intussusception | 19 | 0.9 | 36.8% | 16 (84.2%) |
| Haematochezia | 23 | 1.1 | 26.1% | 2 (8.7%) |
| Other gastrointestinal event | 121 | 6.0 | 22.3% | 4 (3.3%) |
| Kawasaki syndrome | 8 | 0.4 | 0 | 8 (100%) |
| Other systemic event | 100 | 4.9 | 10% | 26 (26%) |
| Hypotonic hyporesponsive episode | 36 | 1.8 | 5.6% | 14 (38.9%) |
| Persistent crying | 19 | 0.9 | 26.3% | 0 |
| Rash alone | 128 | 6.3 | 5.5% | 1 (0.8%) |
| Other event not listed above | 20 | 1.0 | 5% | 9 (45%) |
| Vaccination error with no adverse event | 1 | 0.05 | 0 | 0 |
| Total | 555 | 27.5 | 13% | 99 (17.8) |
In conclusion rotavirus vaccine has an excellent safety profile after four years of use in Canada’s publicly funded programs. As noted in the pre-licensure clinical trials, gastrointestinal complaints as well as rash as a solitary event are among the most common events reported and are usually non-serious.
2. Kawasaki Syndrome as an AEFI: 2011-2014
Kawasaki syndrome is an illness seen primarily in infants and young children in which there is widespread inflammation of blood vessels including coronary arteries. The cause is unknown and several different factors such as viral infection are thought to play a role. The background incidence in children aged less than 5 years ranges from 9 to 19 cases/100,000 population. Of note, in 2013 the IMPACT network initiated systematic active surveillance for vasculitis syndromes in children, including Kawasaki syndrome. Prior to 2013 IMPACT reporting was sporadic, if and when cases were called to the attention of nurse monitors. Thus the increase in reporting in 2014 relative to the average for 2011-2013 was artifactual based on initiation of a new surveillance target starting in 2013. On detailed review of individual reports there was no increase in reporting for vaccines administered in 2014 relative to previous years. Further the reporting frequency is within what would be expected by chance alone given the background incidence.
Page details
- Date modified: