ARCHIVED - The changing epidemiology of hepatitis A in British Columbia: using Health Authority follow-up data to inform policy and practice
15 October 2006 Volume 32 Number 20
SL Pollock, MSc, MD (1), A Sheikholeslami, MD,MHSc (1), B Edgar, MHSc (1), ST David, MHSc (1), JA Buxton, MBBS, MHSc, FRCPC (1)
-
Department of Epidemiology Services, British Columbia Centre for Disease Control, Vancouver, British Columbia
Infection with hepatitis A virus (HAV) causes fever, malaise, anorexia and abdominal discomfort, followed by jaundice in adults and school-aged children1,2. HAV is often asymptomatic in younger children. Transmission usually occurs through the fecal-oral route, via direct contact with infected persons or indirectly through ingestion of contaminated water or foods. Through surveillance and outbreak investigation, individuals and groups at high risk of HAV infection have been identified, and they include men who have sex with men (MSM)3, intravenous drug users (IDU), household contacts of hepatitis A cases, Aboriginal populations4 and travelers to endemic countries.
HAV vaccine is effective at preventing HAV and is often used as post-exposure prophylaxis and in response to outbreaks. Despite evidence of efficacy and the relative low cost of the vaccine, HAV vaccine strategies remain selectively targeted at groups at high risk of infection or at risk of severe complications of HAV (e.g. persons with hepatitis C). Vaccine strategies may not protect all members of high risk groups as a result of poor participation, cost barriers and lack of perceived risk and/or knowledge.
Sensitive occupations that involve a risk of HAV transmission involve food handlers, and health care and child care workers. Identification of a case of HAV in a food handler leads to public concern, and there may be huge costs associated with vaccinating potenial contacts. For example, following identification of an HAV-infected food handler in Vancouver in 2002, more than 6,000 persons received immune globulin5.
HAV vaccination policies in British Columbia (BC) have evolved in response to trends in HAV infection. These policy changes have included publicly funded vaccines for individuals with hemophilia A and B in 1994; IDU and persons with hepatitis C in 1998; and MSM, persons with chronic hepatitis B and/or chronic liver disease in 20016. Additionally, vaccine replaced immune globulin for post-exposure prophylaxis in 2002.
HAV infections are notifiable in BC under the BC Health Act. Cases are reported using the integrated Public Health Information System (iPHIS), and they include basic demographic information (age, sex, year of case and health authority of residence). Although HAV cases are followed up by their local health authority, follow-up data are not reported to the province. As a result, there is no clear provincial picture of the type of follow-up conducted, the burden of disease, the epidemiologic profile of cases or whether targeted immunization programs have affected the epidemiologic profile of cases.
The purpose of this study was to collect and collate HAV case follow-up data from the BC health authorities with the following objectives:
-
assess the follow-up methods used and the completeness of follow-up data;
-
measure the burden of HAV in BC;
-
conduct descriptive epidemiologic analysis of HAV; and
-
make recommendations to enhance HAV follow-up and explore new HAV vaccination strategies.
Methods
This was a retrospective descriptive analysis using non-nominal records of all HAV cases followed up by four of the five health authorities in BC between 1998 and 2004. Descriptive analyses were conducted to ascertain the burden of disease and epidemiologic trends. Follow-up data collected from the health authorities were compared with demographic data from iPHIS.
Cases occurring outside the review period and individuals listed on follow-up as “non-cases”, “unknown” cases or “missing” were excluded. Data were entered from the follow-up forms into EpiData, and descriptive statistics were calculated using Epi-Info. Data were analyzed according to a priori case definitions – only confirmed cases (HAV IgM reactive) or clinical cases (symptomatic contact of known case) were included.
Results
Demographic data on reported HAV cases were available for all five BC health authorities through iPHIS. Four of these five health authorities, which include 83% of the total BC population, provided follow-up data (2004 population data7).
HAV case follow-up across health authorities varied by method and data completeness. The time taken for case follow-up (excluding post-exposure prophylaxis administration), based on review of 145 consecutive case reports, ranged from 10 minutes to 8 hours, with a mean of 93 minutes and median of 60 minutes.
Comparison of demographic features: follow-up data vs iPHIS data
From the four health authorities that provided information, demographic data were available for 980 cases of HAV reported to iPHIS and for 805 confirmed or clinical HAV cases followed up by participating health authorities between 1998 and 2004 see Table 1).
The health authorities provided follow-up data on 82% of cases reported through the iPHIS database.
Table 1. HAV cases by age and sex, 1998-2004 (iPHIS and follow-up)
Health authority |
Number of cases |
Median age (range), years |
Male (n (%)) |
Female (n (%)) |
||||
iPHIS Follow-up |
iPHIS Follow-up* |
iPHIS |
iPHIS |
|||||
Fraser |
356 |
294 |
31 |
30 |
229 |
189 |
127 |
105 |
Vancouver Coastal |
426 |
348 |
35 |
35 |
304‡ (72%) |
259 (74%) |
119‡ (28%) |
89 (26%) |
Vancouver Island |
97 |
93 |
41 |
45 |
62 (64%) |
33 (61%) |
35 (36%) |
21 (39%) |
Northern |
101 |
70 |
25 |
22 |
47 (47%) |
33 (47%) |
54 (53%) |
37 (53%) |
Interior |
166 |
N/A |
37 |
N/A |
100 (60%) |
N/A |
66 (40%) |
N/A |
Totals |
1,146 §980 |
805 |
34 |
33 |
742 (65%) |
514 (67%) |
401 (35%) |
252 (33%) |
* Age available for 91% of total cases.
† Sex available for 95% of total cases.
‡ For 3 cases the sex was unknown.
§ Total number of cases in iPHIS excluding Interior Health.
HAV cases by occupation
Of those with known occupations, 47/568 (8.3%) were employed in occupations considered to be sensitive (see Table 2).
Occupation |
Number of cases |
Percentage of total cases followed up |
Food handler |
35 |
4.4% |
Health care worker |
9 |
1.1% |
Child care worker |
3 |
0.4% |
Retired |
30 |
3.7% |
Child < 14 years |
67 |
8.3% |
Unemployed |
70 |
8.7% |
Other |
354 |
44.0% |
Unknown/missing |
237 |
29.4% |
Contacts
Data on number of contacts immunized were available for 415 cases. Of these, 102/415 cases (24.6%) had no contacts who had been immunized, and 313/415 cases (75.4%) had at least one contact immunized. The mean number of contacts immunized per HAV case was 3.8 cases (median = 2; range = 0-46). Information from follow-up data did not include outbreaks of hepatitis A in which large groups of individuals had been immunized.
Risk factor groups
Of the 805 cases with follow-up information, a total of 99 cases identified themselves as MSM (83% from the Vancouver Coastal Health Authority), 208 cases had traveled to another country during the incubation period (68% to an HAV-endemic country), and 59 cases reported being IDU. Of those travel cases for which information was available on the country visited (n = 175), 43 cases travelled to Mexico, 37 cases to India, 28 to the United States and 10 to China. There were 10 cases who were of First Nations heritage. Ethnicity was unknown for 59.7% of cases (458/767); 51.1% of cases with known ethnicity (158/309) were of Canadian origin.
Figure 1 depicts HAV cases by selected risk factor group and year. The data show a decline in the number of cases reported for MSM and IDU groups over time, whereas case numbers for travelers remained fairly constant.
Figure 1. Risk factor groups for hepatitis A: British Columbia health authority follow-up data, 1998-2004.
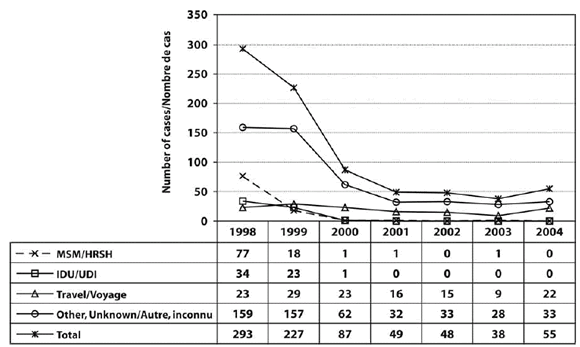
*Case numbers depicted in Figure 1 differ from numbers reported in text because of incomplete group data on risk factors reported by year.
Outcomes
Sixty-four HAV cases (8.1%) were hospitalized during the follow-up period. The mean age of these hospitalized cases was 33 years (range 6-69 years).
Two HAV cases died. Both were males (no known MSM) in the 40-49 age group. Both were hospitalized, and one of the two cases had chronic liver disease (hepatitis B and C).
Discussion
Provincial vaccination strategies targeted at HAV infections in BC have developed and evolved over time in response to identified risk factor groups. The results and conclusions from this report could be generalized to other Canadian provinces/territories, provided that their vaccination strategies are similar, in order to inform policy and procedure around HAV vaccination strategies.
These analyses were intended to describe trends in risk factors and burden of HAV cases across BC health authorities using follow-up data. Comparison between the iPHIS and follow-up data was performed to ensure that follow-up data were representative of the HAV cases reported in BC. One health authority did not provide follow-up data, and because only demographic data are collected through iPHIS an accurate comparison between the two data sources is challenging. Additionally, some of the iPHIS and follow-up cases had missing data (e.g. age, sex and occupation). Nevertheless, given that 82% of HAV cases recorded in iPHIS are receiving some degree of follow-up by health authorities, the follow-up process is encouraging. Some of the cases, such as non-residents of BC, would not have been entered into iPHIS and/or may have received some or all of their follow-up in their usual area of residence. The type of follow-up form used, the individual(s) completing the follow-up (environmental health officer versus public health nurse) and the time to complete the follow-up process are all factors that may contribute to the completeness of HAV case follow-up in a health authority.
By characterizing the burden of disease associated with HAV using standard data collection methods, it may be possible to target future vaccination strategies towards high-risk groups with the aim of decreasing the incidence of HAV infection. The overall numbers of HAV cases reported in BC have declined, likely because of immunization blitzes targeted at high-risk groups8 and subsequent targeted vaccination policies. For example, trends show a decrease in HAV rates following vaccine policies targeted at MSM and IDU groups, and travelers now represent a significant proportion of the known disease burden. There is currently no publicly funded vaccine offered to travelers in BC. Although HAV vaccine is recommended during travel to endemic countries, greater effort should be directed at encouraging HAV vaccine for at-risk travelers, including those originally from endemic countries. On follow-up, some of the HAV cases were identifed as originating from endemic countries (data not shown).
We have demonstrated that travelers now account for a significant proportion of the HAV disease burden in BC as a result of decreasing rates of HAV in MSM and IDU risk groups, corresponding to the implementation of publicly funded vaccination programs. Cost may be a major barrier to HAV vaccine uptake in some travelers, and rduced cost or free vaccine could improve uptake. However, other barriers may be more important, such as lack of knowledge about the vaccine, priority given to receipt of other vaccines, inadequate pre-trip counselling among those going to countries where HAV is endemic and/or lack of perceived risk. Identification of barriers and ways to improve uptake in travelers should be explored.
In addition to encouraging increased uptake of HAV vaccine, emphasis can be placed on improvement of pre-travel messages, including education regarding fecal-oral contamination and personal hygiene. Targeted media campaigns (TV commericals, brochures and posters) have worked well in the MSM and IDU risk groups.
Currently, follow-up questionnaires on HAV cases are not standardized among health authorities. Additionally, if large numbers of individuals are immunized in association with a potential HAV outbreak, these data are not being recorded on follow-up. For example, during the period 1998-2004 in BC, three events associated with an HAV case led to 6,400, 180 and 474 individuals subsequently being immunized. A standard provincial form is currently being developed through collaboration between the BC Centre for Disease Control and the health authorities in order to ensure that there is efficient and complete collection and collation of these data. In addition, individual health authorities can currently enter comments on individual cases into iPHIS for their own reference. Standardized data collection methods and follow-up procedures at both provinical and national levels may be used in the future to identify potential sources of HAV in clusters of cases and further inform HAV vaccination strategies.
Acknowledgements
We gratefully acknowledge the participating health authorities. Thanks go to Shaleen Krilow of the BC Centre for Disease Control for her assistance.
References
-
Health Canada. Canadian immunization guide, 6th edition. Ottawa: Health Canada, 2002.
-
Heymann DL, ed. Control of communicable diseases manual, 18th ed. American Public Health Association, 2004.
-
Allard R, Beauchemin J, Bedard L et al. Hepatitis A vaccination during an outbreak among gay men in Montreal, Canada, 1995-1997. J Epidemiol Community Health 2001;55:251-56.
-
Harb J, Lem M, Fyfe M et al. Hepatitis A in the northern interior of British Columbia: an outbreak among members of a First Nations community. CCDR 2000;26(19):157-61.
-
The BC Food Protection Association Newsletter. Spring 2003;1(6). URL: http://www.bcfpa.net/Attachments/Newsletters/ BCFPA Grapevine Vol. 1 No. 6 Spring 03.pdf
-
British Columbia Centre for Disease Control. Hepatitis A. In: Communicable disease control manual. Vancouver: BC Centre for Disease Control, 2005. URL: http://www.bccdc.org/content.php?item=192
-
BC statistics – population estimates and projections, P.E.O.P.L.E, run cycle 30. Vancouver: BC Stats, Ministry of Labour and Citizens’ Services. URL: http://www.bcstats.gov.bc.ca/DATA/pop/popstart.asp
-
Weatherill SA, Buxton JA, Daly PC. Immunization programs in non-traditional settings. Can J Public Health 2004;95(2):133-37.
Page details
- Date modified:
