Screening assessment acetonitrile
Official title: Screening assessment - Acetonitrile
Chemical Abstracts Service Registry Number
75-05-8
Environment and Climate Change Canada
Health Canada
April 2021
Cat no.: En84-225/2021E-PDF
ISBN 978-0-660-37738-4
Synopsis
Pursuant to section 74 of the Canadian Environmental Protection Act, 1999 (CEPA), the Minister of the Environment and the Minister of Health have conducted a screening assessment of acetonitrile (Chemical Abstracts Service Registry Number [CAS RNFootnote 1 ] 75‑05‑8), which was identified as a priority for assessment as it met categorization criteria under subsection 73(1) of CEPA.
Acetonitrile was included in a survey issued pursuant to section 71 of CEPA. There were no reports of manufacture of acetonitrile above the reporting threshold of 100 kg during the 2011 calendar year in Canada. Acetonitrile was reported as being imported into Canada during the 2011 calendar year with a total volume in the range of 10 000 to 100 000 kg for use in laboratories.
Acetonitrile was measured in ambient and indoor air in Canada, as part of a series of residential air quality studies (2005-2010). Acetonitrile occurs naturally (for example, in coal tar, volcanic gas, and the combustion products of wood and other biomass) and is present in tobacco smoke.
The ecological risk of acetonitrile was characterized using the ecological risk classification of organic substances (ERC), which is a risk-based approach that employs multiple metrics for both hazard and exposure, with weighted consideration of multiple lines of evidence for determining the risk classification. Hazard profiles are based principally on metrics regarding mode of toxic action, chemical reactivity, food web-derived internal toxicity thresholds, bioavailability, and chemical and biological activity. Metrics considered in the exposure profiles include potential emission rate, overall persistence, and long-range transport potential. A risk matrix is used to assign a low, moderate or high level of potential concern for substances based on their hazard and exposure profiles. Based on the outcome of the ERC analysis, acetonitrile is considered unlikely to be causing ecological harm.
Considering all available lines of evidence presented in this screening assessment, there is low risk of harm to the environment from acetonitrile. It is concluded that acetonitrile does not meet the criteria under paragraphs 64(a) or (b) of CEPA as it is not entering the environment in a quantity or concentration or under conditions that have or may have an immediate or long-term harmful effect on the environment or its biological diversity or that constitute or may constitute a danger to the environment on which life depends.
Acetonitrile has been reviewed internationally through the Organization of Economic Co-operation and Development Cooperative Chemicals Assessment Programme (OECD) where it was determined that acetonitrile is not considered to be mutagenic, carcinogenic, nor a reproductive toxicant. The critical effect was based on haematological effects observed in mice following chronic inhalation exposure.
The general population of Canada may be exposed to acetonitrile from environmental media. A comparison of estimated exposure to acetonitrile from environmental media and critical effect levels results in margins of exposure that are considered adequate to address uncertainties in the health effects and exposure databases.
On the basis of the information presented in this screening assessment, it is concluded that acetonitrile does not meet the criteria under paragraph 64(c) of CEPA as it is not entering the environment in a quantity or concentration or under conditions that constitute or may constitute a danger in Canada to human life or health.
It is therefore concluded that acetonitrile does not meet any of the criteria set out in section 64 of CEPA.
1. Introduction
Pursuant to section 74 of the Canadian Environmental Protection Act, 1999 (CEPA) (Canada 1999), the Minister of the Environment and the Minister of Health have conducted a screening assessment of one of six substances (acetonitrile, Chemical Abstracts Service Registry Number [CAS RNFootnote 2 ] 75-05-8) referred to under the Chemical Management Plan as the Nitriles Group, to determine whether this substance presents or may present a risk to the environment or to human health. The substance was identified as a priority for assessment as it met categorization criteria under subsection 73(1) of CEPA (ECCC, HC [modified 2017]).
The other five substances (the CAS RN are listed in Table 1‑1, below) were considered in the ecological risk classification of organic substances (ERC) and the Threshold of Toxicological Concern (TTC)-based Approach for Certain Substances science approach documents (ECCC 2016a; Health Canada 2016) and were identified as being of low concern to both human health and the environment. As such, they are not further addressed in this report. Conclusions for these five substances are provided in the Substances Identified as Being of Low Concern on the basis of the Ecological Risk Classification of Organic Substances and the Threshold of Toxicological Concern (TTC)-based Approach for Certain Substances Screening Assessment (ECCC, HC 2018).
| CAS RN | Domestic Substances List name |
|---|---|
| 78-67-1 | Propanenitrile, 2,2’-azobis[2-methyl- |
| 13472-08-7 | Butanenitrile, 2,2’-azobis[2-methyl- |
| 61790-28-1 | Nitriles, tallow |
| 61790-29-2 | Nitriles, tallow, hydrogenated |
| 125328-64-5 | Nitriles, rape-oil, hydrogenated |
The ecological risk of acetonitrile was characterized using the ERC approach (ECCC 2016a). The ERC describes the hazard of a substance using key metrics including mode of toxic action, chemical reactivity, food web-derived internal toxicity thresholds, bioavailability, and chemical and biological activity and considers the possible exposure of organisms in the aquatic and terrestrial environments on the basis of factors such as potential emission rates, overall persistence and long-range transport potential in air. The various lines of evidence are combined to identify substances as warranting further evaluation of their potential to cause harm to the environment or as having a low likelihood of causing harm to the environment.
The substance, acetonitrile, has been reviewed internationally through the Organisation for Economic Co-operation and Development (OECD) Cooperative Chemicals Assessment Programme and there is an OECD SIAR (SIDS Initial Assessment Report) available. These assessments undergo rigorous review (including peer-review) and endorsement by international governmental authorities. Health Canada and Environment and Climate Change Canada are active participants in this process, and consider these assessments to be reliable. The OECD SIAR was used to inform the health effects characterization in this screening assessment.
This screening assessment includes consideration of information on chemical properties, environmental fate, hazards, uses and exposures, including additional information submitted by stakeholders. Relevant data were identified up to July 2018.
This screening assessment was prepared by staff in the CEPA Risk Assessment Program at Health Canada and Environment and Climate Change Canada and incorporates input from other programs within these departments. The ecological portion of this assessment is based on the ERC document (published July 30, 2016), which was subject to an external review as well as a 60-day public comment period. Additionally the draft of this screening assessment (published June 22, 2019) was subject to a 60-day public comment period. While external comments were taken into consideration, the final content and outcome of the screening assessment remain the responsibility of Health Canada and Environment and Climate Change Canada.
This screening assessment focuses on information critical to determining whether substances meet the criteria as set out in section 64 of CEPA by examining scientific information and incorporating a weight of evidence approach and precautionFootnote 3 . This screening assessment presents the critical information and considerations on which the conclusion is based.
2. Substance identity
The CAS RN, Domestic Substances List (DSL) name and structure for acetonitrile are presented in Table 2‑1.
| CAS RN | DSL name | Chemical structure and molecular formulaa | Molecular weight (g/mol)a |
|---|---|---|---|
| 75-05-8 | acetonitrile | 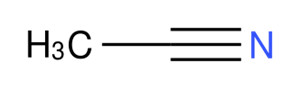 C2H3N
C2H3N | 41.1 |
a PubChem 2004- .
3. Physical and chemical properties
A summary of physical and chemical property data of acetonitrile are presented in Table 3‑1. Additional physical chemical properties are reported in ECCC 2016b.
| Property | Value (or range) | Key reference(s) |
|---|---|---|
| Physical state | Colourless liquid | ECHA 2018; Lewis 2007; Rumble 2018; SDS 2017 |
| Melting point (°C) | -41 to -48 | ECHA 2018; Lewis 2007; Rumble 2018; SDS 2017 |
| Vapour pressure (Pa) | 9.45 × 103 to 12.1 × 103 | ECHA 2018; Rumble 2018; SDS 2017 |
| Water solubility | Miscible | ECHA 2018; Lewis 2007; Rumble 2018; SDS 2017 |
| Log Kow (dimensionless) | -0.34 | Hansch and Anderson 1967 |
Abbreviations: Kow, octanol-water partition coefficient.
4. Sources and uses
Acetonitrile was included in a survey issued pursuant to section 71 of CEPA (Canada 2012). The substance was not reported to be manufactured above the reporting threshold of 100 kg during the 2011 calendar year in Canada, while total import quantities during that same period were reported in a range of 10 000 to 100 000 kg for use in laboratories (Environment Canada 2013)Footnote 4 . The substance is also present as a formulant in three pest control products registered in Canada (email from Pest Management Regulatory Agency, Canada, to Existing Substances Risk Assessment Bureau, Health Canada, dated April 5, 2018; unreferenced).
5. Potential to cause ecological harm
5.1 Characterization of ecological risk
The ecological risk of acetonitrile was characterized using the ecological risk classification of organic substances (ERC) approach (ECCC 2016a). The ERC is a risk-based approach that considers multiple metrics for both hazard and exposure, with weighted consideration of multiple lines of evidence for determining risk classification. The various lines of evidence are combined to discriminate between substances of lower or higher potency and lower or higher potential for exposure in various media. This approach reduces the overall uncertainty with risk characterization compared to an approach that relies on a single metric in a single medium (e.g., median lethal concentration) for characterization. The following summarizes the approach, which is described in detail in ECCC (2016a).
Data on physical-chemical properties, fate (chemical half-lives in various media and biota, partition coefficients, and fish bioconcentration), acute fish ecotoxicity, and chemical import or manufacture volume in Canada were collected from the scientific literature, available empirical databases (e.g., OECD [Q]SAR Toolbox 2014), and from responses to surveys conducted under section 71 of CEPA, or they were generated using selected quantitative structure-activity relationship ([Q]SAR) or mass-balance fate and bioaccumulation models. These data were used as inputs to other mass-balance models or to complete the substance hazard and exposure profiles.
Hazard profiles were established principally on metrics regarding mode of toxic action, chemical reactivity, food web-derived internal toxicity thresholds, bioavailability, and chemical and biological activity. Exposure profiles were also based on multiple metrics, including potential emission rate, overall persistence, and long-range transport potential. Hazard and exposure profiles were compared to decision criteria in order to classify the hazard and exposure potentials for each organic substance as low, moderate, or high. Additional rules were applied (e.g., classification consistency, margin of exposure) to refine the preliminary classifications of hazard or exposure.
A risk matrix was used to assign a low, moderate or high classification of potential risk for each substance on the basis of its hazard and exposure classifications. ERC classifications of potential risk were verified using a two-step approach. The first step adjusted the risk classification outcomes from moderate or high to low for substances that had a low estimated rate of emission to water after wastewater treatment, representing a low potential for exposure. The second step reviewed low risk potential classification outcomes using relatively conservative, local-scale (i.e., in the area immediately surrounding a point source of discharge) risk scenarios, designed to be protective of the environment, to determine whether the classification of potential risk should be increased.
ERC uses a weighted approach to minimize the potential for both over and under classification of hazard, exposure and subsequent risk. The balanced approaches for dealing with uncertainties are described in greater detail in ECCC (2016a). The following describes two of the more substantial areas of uncertainty. Error with empirical or modelled acute toxicity values could result in changes in classification of hazard, particularly metrics relying on tissue residue values (i.e., mode of toxic action), many of which are predicted values from (Q)SAR models (OECD QSAR Toolbox 2014). However, the impact of this error is mitigated by the fact that overestimation of median lethality will result in a conservative (protective) tissue residue value used for critical body residue analysis. Error with underestimation of acute toxicity will be mitigated through the use of other hazard metrics such as structural profiling of mode of action, reactivity and/or estrogen binding affinity. Changes or errors in chemical quantity could result in differences in classification of exposure as the exposure and risk classifications are highly sensitive to emission rate and use quantity. The ERC classifications thus reflect exposure and risk in Canada based on what is estimated to be the current use quantity, and may not reflect future trends.
Critical data and considerations used to develop the substance-specific profiles for acetonitrile, and the hazard, exposure and risk classification results are presented in ECCC (2016b).
On the basis of low hazard and low exposure classifications according to information considered under ERC, acetonitrile was classified as having a low potential for ecological risk. It is unlikely that this substance is resulting in concerns for the environment in Canada.
6. Potential to cause harm to human health
6.1 Exposure assessment
Natural occurrences of acetonitrile have been identified in the combustion products of wood and other biomass (ECB 2002; Yuan et al 2010), volcanic gas (Schwandner et al. 2004) and coal tar (Islas et al. 2002).
Acetonitrile was reported to be imported into Canada for use in laboratories (Environment Canada 2013). Possible environmental releases from uses in laboratories are unknown, but are expected to be low given expected disposal of this substance as hazardous waste, as well as data reported under NPRI (NPRI 2018a), and measured air concentrations.
No reports of acetonitrile measured in drinking water in Canada were identified. According to the National Pollutant Release Inventory (NPRI) database, there were no reported releases of acetonitrile to water or to municipal wastewater treatment systems for the reporting years 2011 to 2017 (NPRI 2018a).
In the NPRI database the average total annual release of acetonitrile to air was 40.25 tonnes for the reporting years 2006 to 2013, decreasing to an average of 18.25 tonnes for the reporting years 2014 to 2017. Industries reporting releases to air include those in the waste treatment and disposal, synthetic rubber manufacturing, and pharmaceutical manufacturing sectors (NPRI 2018b). The overall degradation half-life for acetonitrile in the troposphere has been estimated to be between 42 and 321 days (ECB 2002; US EPA 1987).
As part of a series of residential air quality studies, Health Canada (2010a,b, 2012, 2013) collected indoor and outdoor air samples for analysis of selected volatile organic compounds, including acetonitrile. Sampling took place during the summer and winter seasons in Edmonton (2010), Halifax (2009), Regina (2007), and Windsor (2005 to 2006). With the exception of the summer sampling periods for indoor air in Halifax and Regina, detection frequencies for acetonitrile were greater than 95% (with method detection limits of 0.047 µg/m3 or lower).
The Health Canada (2010a,b, 2012, 2013) air quality studies consisted of paired indoor-outdoor air measurements (i.e., simultaneous measurements were taken inside and outside the residence [e.g., a backyard] during sampling). Twenty-four-hour personal sampling was also conducted in Windsor. The 95th percentile personal sampling acetonitrile levels ranged from 0.85 to 1.58 µg/m3 and were typically higher in comparison to indoor and outdoor samples. Samples collected in Regina included residences of smokers (n=117) and non-smokers (n= 34). Samples from the other cities were exclusively residences of non-smokers. In the Regina samples, the ratio of indoor-air to outdoor-air acetonitrile levels for residences of non-smokers was less than 2, but as great as 18 for residences occupied by smokers. Tobacco smoke is a known source of acetonitrile (IPCS 1993; US EPA 1985) and is a contributing factor for acetonitrile levels, being on average, greater indoors compared to outdoors. As the acetonitrile concentrations detected in the indoor air of non-smoking homes is still higher than concentrations in outdoor air, this suggests an additional source(s) of acetonitrile originating from inside the residence.
In this assessment, the 95th percentile indoor air concentration based on a full data set (i.e., from residences comprised of smokers and exclusively non-smokers) is selected to estimate inhalation exposure to acetonitrile in indoor air. The highest 95th percentile for the only dataset that included residences with and without smokers present (Regina; summer sampling period) was 4.99 µg/m3 and is considered to be both a protective and representative exposure estimate for the general population. In comparison, the 95th percentile for residences from all four studies where no smoking was reported was in the range of 0.46 to 1.84 µg/m3 (Health Canada 2010a,b, 2012, 2013).
The 95th percentile concentration for outdoor air, reported in Halifax (winter sampling period; 1.26 µg/m3), was the highest 95th percentile outdoor air concentration reported (Health Canada 2010a,b, 2012, 2013) and used to estimate inhalation exposure to acetonitrile while outdoors.
The estimated daily intake for all age groups for acetonitrile via inhalation ranged from 0.92 to 3.29 µg/kg bw/day, with children aged 1 to 2 years old having the highest estimated exposure (See Appendix A. Estimated daily intake from inhalation exposures to humans for details).
6.2 Health effects assessment
OECD (1997) summarized the health effects literature and characterized the hazard for acetonitrile. The final assessment report from the OECD (SIAR) was published in 2002 by the European Chemicals Bureau (ECB) in the European Union (ECB 2002). The EU Risk Assessment Report (RAR) (ECB 2002) was used to inform the health effects characterization in this screening assessment, including selection of effect levels for critical endpoints.
A literature search was conducted for information more recent than the date of the last literature review, 1998, as reported in the EU report (ECB 2002). No health effects studies which could impact the risk characterization (i.e., result in different critical endpoints or lower points of departure than those stated in ECB 2002) were identified. In addition, reviews on acetonitrile from the US EPA (IRIS 1997), Australia (AGDH 2017) and Japan (J-CHECK c2010- ) were consulted as part of the extended literature search.
As reported by the ECB (2002), toxicokinetic data has shown that acetonitrile is well absorbed from the lungs and gastrointestinal tract and rapidly distributed throughout the body. Cyanide is the primary metabolite of acetonitrile, via cytochrome P450 metabolic biotransformation (ECB 2002; IRIS 1997- ).
In repeat dose inhalation exposure studies mice were found to be a sensitive species to acetonitrile (ECB 2002). The NTP (1996) conducted 13-week inhalation studies on both the F344/N rat and B6C3F1 mouse. In rats, groups of 10 males and females were exposed to 0, 100, 200, 400, 800 or 1600 ppm acetonitrile by inhalation for 6 hours per day, 5 days per week for 13 weeks. The same concentrations and dosing regimens were administered to groups of 10 male and 10 female B6C3F1 mice over 13 weeks. The results of these studies were used to inform dose selection for a subsequent 2 year inhalation study on F344/N rats and B3C6F1 mice. The 2-year inhalation study exposed groups of 60 male and 60 female B3C6F1 mice to 0, 50, 100 or 200 ppm acetonitrile for 6 hours a day for 5 days per week. In the same 2-year inhalation study, groups of 56 male and 56 female F344/N rats were exposed to concentrations of 0, 100, 200 or 400 ppm of acetonitrile for 6 hours per day, 5 days per week (NTP 1996). The ECB (2002) summarized the results of these studies and identified a NOAEC of 200 ppm (335 mg/m3) for the F344/N rat based on haematological effects, such as mean erythrocyte haemoglobin and mean erythrocyte volume, observed to be minimally lower than controls at 400 ppm (670 mg/m3) in the 2-year study.
The US EPA (1987) and the ECB (2002) reviewed the results of a 92 day unpublished inhalation study in B3C6F1 mice by Coate (1983). In the study, groups of 10 male and 10 female mice were exposed for 6 hours/day for 65 days over a 92 day period to concentrations of 0, 25, 50, 100, 200 and 400 ppm acetonitrile vapour (ECB 2002; US EPA 1987). A NOAEC of 100 ppm (168 mg/m3) was identified based on a significant decrease in blood urea nitrogen levels, hematocrits and erythrocytes, amongst other haematological effects, at higher doses.
Based on data from the NTP (1996) studies, as well as additional repeat dose inhalation effects observed from a number of unpublished studies, including the study by Coate (1983), evaluated as part of their toxicological assessment of acetonitrile, the ECB (2002) identified a NOAEC of 100 ppm (168 mg/m3) based on haematological effects, including statistically significant decreases in blood urea levels, red blood cell cholinesterase counts and hematocrits in female mice (ECB 2002; US EPA 1987). The selection of a NOAEC in mice of 100 ppm (168 mg/m3) as the critical effect for chronic repeat dose exposures to acetonitrile (ECB 2002; OECD 1997) is consistent with the value selected by other international agencies, including the US EPA (IRIS 1997- ), Japan (J-CHECK c2010‑ ) and Australia (AGDH 2017).
Acetonitrile is not considered to be mutagenic or carcinogenic based on available data (AGDH 2017; ECB 2002; IRIS 1997). The reproductive and developmental toxicity of acetonitrile has been examined in a number of studies. ECB 2002 reported that in most available studies, where reproductive and/or developmental effects were observed, the effects were associated with maternal toxicity. In rabbits administered acetonitrile by oral gavage, a NOAEL of 30 mg/kg bw/day for developmental effects was reported (highest dose tested), with a NOAEL for maternal toxicity reported at 15 mg/kg bw/day (mid dose). The maternal toxicity in this study was associated with reduced body weight gain during the exposure period (AGDH 2017; ECB 2002‑ ; IRIS 1997- ). Similarly, in two developmental toxicity inhalation studies reported for SD rats, a NOAEC for maternal toxicity of 1200 ppm (2015 mg/m3) was observed, with higher doses associated with effects on maternal body weights. The NOAEC for developmental toxicity from these studies was determined to be greater than 1200 ppm (2015 mg/m3). A NOAEC of 1500 ppm (2518 mg/m3) was reported in an inhalation study, in which an increase in the percentage of non-surviving implants and early embryonic reabsorptions were observed in SD rats exposed to the next concentration tested of 1800 ppm (3023 mg/m3). There were no other developmental effects observed at this dose, including no significant changes in the average number of implantation sites or incidences of skeletal variations (AGDH 2017; ECB 2002; IRIS 1997- ). The ECB (2002) concluded, based on the available information, that acetonitrile “is not a reproductive toxicant”.
6.3 Characterization of risk to human health
Empirical ambient and indoor air measurements were identified for acetonitrile in a series of residential air quality studies conducted in four cities across Canada, resulting in estimated daily intakes of acetonitrile ranging from 0.92 to 3.29 µg/kg bw/day, with children ages 1 to 2 years old having the highest estimated exposure.
To calculate margins of exposure (MOEs) for acetonitrile from inhalation the most appropriate toxicological endpoint was considered to be the NOAEC of 100 ppm (168 mg/m3) which was identified based on effects observed in a number of studies. The critical effect at this concentration was based on haematological effects observed in mice after chronic repeat dose inhalation exposures. The NOAEC of 100 ppm (168 mg/m3) is calculated to be equivalent to a 58.8 mg/kg bw/day.
Comparison of the estimated daily intake for acetonitrile with the selected critical effect levels results in MOEs ranging from 17 872 to 63 913. These MOEs are considered adequate to address uncertainties in the health effects and exposure databases.
6.4 Uncertainties in evaluation of risk to human health
No measured data were identified for drinking water in Canada or elsewhere.
7. Conclusion
Considering all available lines of evidence presented in this screening assessment, there is low risk of harm to the environment from acetonitrile. It is concluded that acetonitrile does not meet the criteria under paragraphs 64(a) or (b) of CEPA as it is not entering the environment in a quantity or concentration or under conditions that have or may have an immediate or long-term harmful effect on the environment or its biological diversity or that constitute or may constitute a danger to the environment on which life depends.
On the basis of the information presented in this screening assessment, it is concluded that acetonitrile does not meet the criteria under paragraph 64(c) of CEPA as it is not entering the environment in a quantity or concentration or under conditions that constitute or may constitute a danger in Canada to human life or health.
It is therefore concluded that acetonitrile does not meet any of the criteria set out in section 64 of CEPA.
References
[AGDH] Australian Government Department of Health. 2017. Human health tier II assessment for acetonitrile . Sydney (AU): Department of Health, National Industrial Chemicals Notification and Assessment Scheme (NICNAS). Inventory Multi-tiered Assessment and Prioritisation (IMAP) Group Assessment Report. [accessed 2018 July 19].
Canada. 1999. Canadian Environmental Protection Act, 1999. S.C. 1999, c.33. Canada Gazette Part III, vol. 22, no. 3.
Canada, Dept. of the Environment. 2012. Canadian Environmental Protection Act, 1999: Notice with respect to certain substances on the Domestic Substances List Canada Gazette, Part I, vol. 146, no. 48, Supplement.
Coate WB. 1983. 90-Day Subchronic toxicity study of acetonitrile in Fischer 344 rats. Final Report (revised). Submitted to National Toxicity Program by Hazelton Laboratories Inc. (cited in ECB 2002).
[ECB] European Chemicals Bureau. 2002. European Union risk assessment report: acetonitrile: CAS No. 75‑05-8. Luxembourg: Office for Official Publications of the European Communities. Report No.: EUR 19839 EN. [accessed 2018 May 15].
[ECCC] Environment and Climate Change Canada. 2016a. Science approach document: ecological risk classification of organic substances. Ottawa (ON): Government of Canada.
[ECCC] Environment and Climate Change Canada. 2016b. Data used to create substance-specific hazard and exposure profiles and assign risk classifications in the Ecological Risk Classification of organic substances. Gatineau (QC). Available from: substances@ec.gc.ca.
[ECCC, HC] Environment and Climate Change Canada, Health Canada. [modified 2017 Mar 12]. Categorization. Ottawa (ON): Government of Canada. [accessed 2018 July 20].
[ECCC, HC] Environment and Climate Change Canada, Health Canada. 2018. Screening assessment: substances identified as being of low concern using the ecological risk classification of organic substances and the threshold of toxicological concern (TTC)-based approach for certain substances. Ottawa (ON): Government of Canada.
[ECHA] European Chemicals Agency. c2007-2018. Registered substances database; search results for CAS RN 75-05-8. Helsinki (FI): ECHA. Substance Information: Chlorocresol. [updated 2018 July 7; accessed: 2018 July 20].
Environment Canada. 2013. DSL Inventory Update data collected under the Canadian Environmental Protection Act, 1999, section 71: Notice with respect to certain substances on the Domestic Substances List. Data prepared by: Environment Canada, Health Canada; Existing Substances Program.
Hansch C, Anderson SM. 1967. The effect of intramolecular hydrophobic bonding on partition coefficients. J Org Chem. 32(8):2583-2586.
Health Canada. 2010a. Regina exposure assessment study (2007): Data summary for volatile organic compound sampling [pdf]. Ottawa (ON): Government of Canada.
Health Canada. 2010b. Windsor exposure assessment study (2005-2006): Data summary for volatile organic compound sampling [pdf]. Ottawa (ON): Government of Canada.
Health Canada. 2012. Halifax exposure assessment study (2009): Data summary for volatile organic compound sampling. Ottawa (ON): Government of Canada.
Health Canada. 2013. Edmonton exposure assessment study (2010): Data summary for volatile organic compound sampling [pdf]. Ottawa (ON): Government of Canada.
Health Canada. 2015. Food Consumption Table derived from Statistics Canada, Canadian Community Health Survey, Cycle 2.2, Nutrition (2004), Share file. Ottawa.
Health Canada. 2016. Science approach document: threshold of toxicological concern (TTC)-based approach for certain substances. Ottawa (ON): Government of Canada.
[HSDB ] Hazardous Substances Data Bank [database]. 1983- . Search results for CAS RN 75-05-8
[IPCS] International Programme on Chemical Safety. 1993. Environmental Health Criteria 154: acetonitrile. Geneva (CH): United Nations Environment Programme, International Labour Organization, World Health Organization. [accessed 2018 July 19].
[IRIS] Integrated Risk Information System [database]. 1997- . Bethesda (MD): US National Library of Medicine. Toxicological Review of Acetonitrile (CAS No. 75-05-8) [pdf]. [accessed 2018 August 3].
Islas CA, Suelves I, Carter JF, Li W, Morgan TJ, Herod AA Kandiyoti R. 2002. Pyrolysis-gas chromatography/mass spectrometry of fractions separated from a low-temperature coal tar: an attempt to develop a general method for characterising structures and compositions of heavy hydrocarbon liquids. Rapid Commun. Mass Spectrom. 16:774-784.
[J-CHECK] Japan Chemicals Collaborative Knowledge database [database]. c2010- . Tokyo (JP): National Institute of Technology and Evaluation (NITE). Summary of initial risk assessment report Acetonitrile CAS No: 75-05-8 [pdf]. [accessed 2018 August 10].
Lewis, RJ Sr, editor. 2007. Hawley’s Condensed Chemical Dictionary. 15th ed. New York (NY): John Wiley & Sons.
[NPRI] National Pollutant Release Inventory [database]. 2018a. Search results for acetonitrile CAS RN 75-05-8. Ottawa (ON): Government of Canada. [accessed 2018 Jul 25].
[NPRI] National Pollutant Release Inventory [database]. 2018b. Search results for acetonitrile CAS RN 75-05-8. Ottawa (ON): Government of Canada. [accessed 2020 Nov 16].
[NTP 1996]. National Toxicology Program (NTP) 1996. NTP Technical Report on the Toxicology and Carcinogenesis Studies of Acetonitrile (CAS No. 75-05-8) in F344/N Rats and B6C3F1 Mice (Inhalation Studies) [pdf]. Technical Report Series No. 447.
[OECD] Organisation for Economic Co-operation and Development. 1997. SIDS Initial Assessment Profile (SIAP): Acetonitrile. SIAM [SIDS Initial Assessment Meeting] 6; 1997 June; Paris, France. [accessed 2018 August 2].
OECD QSAR Toolbox [Read-across tool]. 2014. Version 3.3. Paris (FR): Organisation for Economic Co-operation and Development, Laboratory of Mathematical Chemistry.
PubChem [database]. 2004- . Search for CAS RN 75-05-8. Bethesda (MD): US National Library of Medicine, National Center for Biotechnology Information. [updated 2018 July 14; accessed 2018 July 20].
Rumble JRR, editor. 2018. CRC handbook of chemistry and physics. 99th ed. Boca Raton (FL): CRC Press.
[SDS] Safety Data Sheet. 2017. Acetonitrile. Oakville (ON): Sigma-Aldrich. [accessed 2018 Jul 30].
Schwandner FM, Seward TM, Gize AP, Hall PA, Dietrich VJ. 2004. Diffuse emission of organic trace gases from the flank and crater of a quiescent active volcano (Vulcano, Aeolian Islands, Italy). J Geophys Res. 109(D4).
[US EPA] US Environmental Protection Agency. 1985. Health and Environmental Effect Profile for Acetonitrile. Washington (DC): Environmental Protection Agency (US). US EPA Report Nº EPA-600/X-85-357.
[US EPA] US Environmental Protection Agency. 1987. Health effects assessment for acetonitrile. US EPA Report Nº EPA-600/8-88-012. [as cited in ECB 2002].
[US EPA] US Environmental Protection Agency. 2011. Chapter 6: Inhalation Rates. Exposure Factors Handbook 2011 Edition (Final). U.S. Environmental Protection Agency: Washington, DC. EPA/600/R-09/052F.
[US EPA] US Environmental Protection Agency. 2018. Search results for Chemical Data Reporting: CAS RN 75-05-8. Washington (DC): Environmental Protection Agency (US). [accessed 2018 July 20].
Yuan B, Liu Y, Shao M, Lu S, Streets G. 2010. Biomass burning contributions to ambient VOCs species at a receptor site in the Pearl River delta (PRD). Environ Sci Technol. 44:4577-4582.
Appendix A. Estimated daily intake from inhalation exposures to humans
| Route of exposure | 0 to 5 monthsa | 6 to 11 monthsb | 1 yearc | 2 to 3 yearsd | 4 to 8 yearse | 9 to 13 yearsf | 14 to 18 yearsg | 19 years or olderh |
|---|---|---|---|---|---|---|---|---|
| Ambient airi | 0.09 | 0.09 | 0.11 | 0.09 | 0.07 | 0.05 | 0.04 | 0.03 |
| Indoor airj | 2.56 | 2.59 | 3.18 | 2.68 | 2.11 | 1.45 | 1.12 | 0.89 |
| Total intake | 2.65 | 2.68 | 3.29 | 2.77 | 2.18 | 1.49 | 1.16 | 0.92 |
a Assumed to weigh 6.3 kg (Health Canada 2015), to breathe 3.7 m3 of air per day (US EPA 2011 [modified]).
b Assumed to weigh 9.1 kg (Health Canada 2015), to breathe 5.4 m3 of air per day (US EPA 2011 [modified]).
c Assumed to weigh 11 kg (Health Canada 2015), to breathe 8.0 m3 of air per day (US EPA 2011 [modified]).
d Assumed to weigh 15 kg (Health Canada 2015), to breathe 9.2 m3 of air per day (US EPA 2011 [modified]).
e Assumed to weigh 23 kg (Health Canada 2015), to breathe 11.1 m3 of air per day (US EPA 2011 [modified]).
f Assumed to weigh 42 kg (Health Canada 2015), to breathe 13.9 m3 of air per day (US EPA 2011 [modified]).
g Assumed to weigh 62 kg (Health Canada 2015), to breathe 15.9 m3 of air per day (US EPA 2011 [modified]).
h Assumed to weigh 74 kg (Health Canada 2015), to breathe 15.1 m3 of air per day (US EPA 2011 [modified]).
i 1.26 µg/m3 as reported as the 95th percentile of 286 samples collected during the summer season from Halifax (NS) (Health Canada 2012). Canadians are assumed to spend 3 hours outdoors each day (Health Canada 1998).
j 4.99 µg/m3 as reported as the 95th percentile of 79 samples collected during the summer season from Regina (SK) (Health Canada 2010a). Canadians are assumed to spend 21 hours indoors each day (Health Canada 1998).
