Screening Assessment for the Challenge
This page has been archived on the Web
Information identified as archived is provided for reference, research or recordkeeping purposes. It is not subject to the Government of Canada Web Standards and has not been altered or updated since it was archived. Please contact us to request a format other than those available.
Archived
Benzene, 1,3-bis(1-isocyanato-1-methylethyl)-
(Tetramethyl-m-xylylene diisocyanate)
Chemical Abstracts Service Registry Number
2778-42-9
Environment Canada
Health Canada
November 2008
Table of Contents
Pursuant to section 74 of the Canadian Environmental Protection Act, 1999 (CEPA 1999), the Ministers of the Environment and of Health have conducted a screening assessment on benzene, 1,3-bis(1-isocyanato-1-methylethyl)- (tetramethyl-m-xylylene diisocyanate), Chemical Abstracts Service Registry Number 2778-42-9. This substance was identified as a high priority for screening assessment and included in the Ministerial Challenge because it was found to meet the ecological categorization criteria for persistence, bioaccumulation potential and inherent toxicity to non-human organisms and is believed to be in commerce in Canada.
The substance tetramethyl-m-xylylene diisocyanate was not considered to be a high priority for assessment of potential risks to human health, based upon application of the simple exposure and hazard tools developed by Health Canada for categorization of substances on the Domestic Substances List. Therefore, this assessment focuses on information relevant to the evaluation of ecological risks.
The substance tetramethyl-m-xylylene diisocyanate is an organic substance that is used as an industrial intermediate which is incorporated into various polymers. The substance is not naturally produced in the environment. No reports of manufacture in or import into Canada of this substance at or above the reporting threshold of 100 kg in the 2005 or 2006 calendar year were received in response to notices published under section 71 of CEPA 1999. However, six Canadian and two foreign companies voluntarily reported having a stakeholder interest in this substance in 2005 and 2006. Further information was not provided.
Since there were no reports of import or manufacture at or above the reporting threshold of 100 kg in 2005 or 2006, releases of this substance into the Canadian environment are presumed to be very low. Tetramethyl-m-xylylene diisocyanate reacts with water and is expected to hydrolyze rapidly in the presence of water or moisture. It is semi-volatile, and may have a tendency towards sizable losses via advection when released into the air.
Based on its reactivity in the presence of moisture, tetramethyl-m-xylylene diisocyanate is not expected to be persistent in the environment. Additional consideration of the hydrolysis data for this compound suggests that this compound is rapidly degraded in water and moist environments, such as sediment and moist soils. Similarly, the role of hydrolysis and the potential metabolism/degradation of tetramethyl-m-xylylene diisocyanate in the gut has been considered further. The substance is thus no longer expected to meet the persistence or bioaccumulation criterion as set out in the Persistence and Bioaccumulation Regulations.
For this screening assessment, a conservative exposure scenario was selected in which an industrial operation discharges into the aquatic environment. The predicted environmental concentration in water (PEC) was many orders of magnitude below predicted no-effect concentrations (PNECs) calculated for fish.
In addition and where relevant, research and monitoring will support verification of assumptions used during the screening assessment.
Based on the information available, tetramethyl-m-xylylene diisocyanate does not meet any of the criteria set out in section 64 of the Canadian Environmental Protection Act, 1999.
The Canadian Environmental Protection Act, 1999 (CEPA 1999) (Canada 1999) requires the Minister of the Environment and the Minister of Health to conduct screening assessments of substances that have met the categorization criteria set out in the Act to determine whether these substances present or may present a risk to the environment or human health. Based on the results of a screening assessment, the Ministers can propose to take no further action with respect to the substance, to add the substance to the Priority Substances List (PSL) for further assessment, or to recommend that the substance be added to the List of Toxic Substances in Schedule 1 of the Act and, where applicable, the implementation of virtual elimination.
Based on the information obtained through the categorization process, the Ministers identified a number of substances as high priorities for action. These include substances that
- met all of the ecological categorization criteria, including persistence (P), bioaccumulation potential (B) and inherent toxicity to aquatic organisms (iT), and were believed to be in commerce in Canada; and/or
- met the categorization criteria for greatest potential for exposure (GPE) or presented an intermediate potential for exposure (IPE), and had been identified as posing a high hazard to human health based on classifications by other national or international agencies for carcinogenicity, genotoxicity, developmental toxicity or reproductive toxicity.
The Ministers therefore published a notice of intent in theCanada Gazette, Part I, on December 9, 2006 (Canada 2006a), that challenged industry and other interested stakeholders to submit, within specified timelines, specific information that may be used to inform risk assessment, and to develop and benchmark best practices for the risk management and product stewardship of those substances identified as high priorities.
The substance benzene, 1,3-bis(1-isocyanato-1-methylethyl)- was identified as a high priority for assessment of ecological risk as it had been found to be persistent, bioaccumulative and inherently toxic to aquatic organisms and is believed to be in commerce in Canada. The Challenge for this substance was published in theCanada Gazette on May 12, 2007 (Canada 2007). A substance profile was released at the same time. The substance profile presented the technical information available prior to December 2005 that formed the basis for categorization of this substance. As a result of the Challenge, no submissions of new information were received for this substance.
Although benzene, 1,3-bis(1-isocyanato-1-methylethyl)- was determined to be a high priority for assessment with respect to the environment, it did not meet the criteria for GPE or IPE and high hazard to human health based on classifications by other national or international agencies for carcinogenicity, genotoxicity, developmental toxicity or reproductive toxicity. Therefore, this assessment focuses principally on information relevant to the evaluation of ecological risks.
Under CEPA 1999, screening assessments focus on information critical to determining whether a substance meets the criteria for defining a chemical as toxic as set out in section 64 of the Act, where
"64. [...] a substance is toxic if it is entering or may enter the environment in a quantity or concentration or under conditions that
(a) have or may have an immediate or long-term harmful effect on the environment or its biological diversity;
(b) constitute or may constitute a danger to the environment on which life depends; or
(c) constitute or may constitute a danger in Canada to human life or health."
Screening assessments examine scientific information and develop conclusions by incorporating a weight of evidence approach and precaution.
This screening assessment includes consideration of information on chemical properties, hazards, uses and exposure, including the additional information submitted under the Challenge. Data relevant to the screening assessment of this substance were identified in original literature, review and assessment documents, stakeholder research reports and from recent literature searches, up to February 2008 for the ecological sections of the document. Key studies were critically evaluated; modelling results may have been used to reach conclusions. When available and relevant, information presented in hazard assessment from other jurisdictions was considered. The screening assessment does not represent an exhaustive or critical review of all available data. Rather, it presents the most critical studies and lines of evidence pertinent to the conclusion.
This screening assessment was prepared by staff in the Existing Substances Programs at Health Canada and Environment Canada and incorporates input from other programs within these departments. Additionally, the draft of this screening assessment was subject to a 60-day public comment period. The critical information and considerations upon which the assessment is based are summarized below.
For the purposes of this document, this substance will be referred to as tetramethyl-m-xylylene diisocyanate, which has been derived from the DSL inventory name. Other names and identifying characteristics of the substance are presented in Table 1.
| Chemical Abstracts Service Registry Number (CAS RN) | 2778-42-9 |
| Name on Domestic Substances List (DSL) | Benzene, 1,3-bis(1-isocyanato-1-methylethyl)- |
| Inventory names1 | Benzene, 1,3-bis(1-isocyanato-1-methylethyl)- (TSCA, AICS, ASIA-PAC, NZIoC, DSL) 1,3-Bis(1-isocyanato-1-methylethyl)benzene (EINECS, ENCS, ECL) |
| Other names | a,a,a',a'-Tetramethyl-m-phenylenedimethylene diisocyanate; a,a,a',a'-Tetramethyl-m-xylylene diisocyanate; 1,3-Bis(a-isocyanatoisopropyl)benzene; Isocyanic acid, a,a,a',a'-tetramethyl-m-xylylene ester; Isocyanic acid, m-phenylenediisopropylidene ester; m-Bis(1-isocyanato-1-methylethyl)benzene; m-TMXDI; Tetramethyl-m-xylylene diisocyanate |
| Chemical group (DSL stream) |
Discrete organics |
| Major chemical class or use | Isocyanate |
| Chemical formula | C14H16N2O2 |
| Chemical structure | 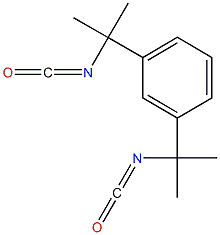 |
| Simplified Molecular Input Line Entry System (SMILES) | O=C=NC(c1cc(ccc1)C(N=C=O)(C)C)(C)C |
| Molecular mass | 244.30 g/mol |
[1] National Chemical Inventories (NCI), 2007: AICS (Australian Inventory of Chemical Substances); ECL (Korean Existing Chemicals List); EINECS (European Inventory of Existing Chemical Substances); ENCS (Japanese Existing and New Chemical Substances); TSCA (Toxic Substances Control Act Chemical Substance Inventory); ASIA-PAC (Asia-Pacific Substances Lists); NZIoC (New Zealand Inventory of Chemicals).
Table 2 contains experimental and modelled physical and chemical properties of tetramethyl-m-xylylene diisocyanate that are relevant to its environmental fate.
| Property | Type | Value[1] | Temperature (°C) | Reference |
|---|---|---|---|---|
| Melting point (°C) | Experimental | -10 | -- | Cytec Industries 2005 |
| Boiling point (°C) | Experimental | 292 | -- | Cytec Industries 2005 |
| Modelled | 320 | -- | MPBPWIN 2000 | |
| Density (kg/m3) | No information available | |||
| Vapour pressure (Pa) | Experimental | 0.43 (3.2E-3 mm Hg) |
25 | Cytec Industries 2005 |
| Modelled | 0.39 (2.98E-3 mm Hg) |
25 | MPBPWIN 2000 (using experimental boiling point and melting point in calculation) | |
| Modelled | 1.4E-2 (1.05E-4 mm Hg) |
25 | MPBPWIN 2000 | |
| Henry's Law Constant (Pa·m3/mol) | Modelled | 3.26E-1 (3.22 E-6 atm·m3/mol) |
25 | HENRYWIN 2000 |
| Log Kow (Octanol-water partition coefficient) (dimensionless) | Modelled | 4.74 | 25 | KOWWIN 2000 |
| Log Koc (Organic carbon partition coefficient) (dimensionless) | Modelled | 5.05 | PCKOCWIN 2000 | |
| Water solubility (mg/L) | Experimental | insoluble (reacts with water) | Cytec Industries 2005 | |
| Modelled | 2.289 | 25 | WSKOWWIN 2000 | |
| Modelled | 5.83 | 25 | WSKOWWIN 2000 (using experimental melting point in calculation) | |
| Modelled | 5.94 | 25 | WATERNT 2002 | |
| Other solubilities (g/L) | Experimental | n/a | ||
| Modelled | n/a | |||
| pKa (Acid dissociation constant) (dimensionless) |
Experimental | n/a | ||
| Modelled | n/a | |||
[1] If different, values in brackets represent the original values as reported by the authors or as estimated by the models.
It is important to note that the modelling technique used to estimate octanol-water partition coefficients (Kow) is a fragment addition method. The fragment addition method does not properly account for the rapid hydrolysis of the isocyanate groups on this compound, and calculates the log Kow based on the isocyanate groups being incorporated in the final Kow prediction. However, as the isocyanate groups are rapidly anticipated to hydrolyse to amines, the Kow prediction is not expected to be environmentally relevant. The log Kow for the compound containing diamines is predicted to be 1.89, as opposed to the log Kow prediction for the diisocyanate compound, which is 4.74. As a result, this chemical type introduces uncertainty into the quantitative structure-activity relationship (QSAR) estimates.
The substance tetramethyl-m-xylylene diisocyanate is not known to be naturally produced in the environment.
Information gathered from the CEPA 1999 section 71 notices for the calendar years 2005 and 2006 indicate that tetramethyl-m-xylylene diisocyanate was not manufactured in or imported into Canada in a quantity meeting the 100 kg reporting threshold (Canada 2006b; Environment Canada 2006; Environment Canada 2007a). Six Canadian and two foreign companies identified themselves as having a stakeholder interest in the substance.
Elsewhere, tetramethyl-m-xylylene diisocyanate has been identified as a High Production Volume (HPV) chemical by the Organisation for Economic Co-operation and Development (1997), as an HPV in the United States (Sponsor Cytec Industries), and as a Low Production Volume (LPV) chemical in the European Union.
No uses were identified as a result of the section 71 notice (Canada 2006b; Environment Canada 2006; Environment Canada 2007a). However, tetramethyl-m-xylylene diisocyanate is used as a monomer in polymers that are being imported into Canada, as confirmed by the New Substances program. Over 40 such polymers have been identified. Previously, "Organic Chemicals, Industrial" was identified as the one use for this substance on the DSL in 1984-1986.
Additionally, a number of uses have been identified through the U.S. HPV program. Tetramethyl-m-xylylene diisocyanate is a versatile aliphatic isocyanate with broad end-use applicability. Tetramethyl-m-xylylene diisocyanate is used as an industrial intermediate that is incorporated into various polymers to improve performance. This chemical imparts improved physical properties to polyurethane products, affording higher strength and improved adhesion, appearance and flexibility, resulting in more durable products. Application areas for the polymers incorporating tetramethyl-m-xylylene diisocyanate include specialty coatings, aqueous dispersions, automotive coatings, wood coatings, inks, sealants, adhesives, thermoplastic urethanes and lacquers. Common commercial products that may have been made using tetramethyl-m-xylylene diisocyanate include fabric and leather finishes, adhesives, automotive paints, printing inks, sealants and wood coatings. Tetramethyl-m-xylylene diisocyanate is approved by the United States Food and Drug Administration for use in food packaging under specific listings in the Code of Federal Regulations (CFR) Title 21 - Food and Drugs, Chapter I - Food and Drug Administration, Department of Health and Human Services (Cytec Industries 2005).
Since there were no reports of import or manufacture at or above the reporting threshold of 100 kg in 2006 in response to a section 71 notice (Environment Canada 2007a), releases of this substance to the Canadian environment are presumed to be very low.
According to Cytec Industries (2005), tetramethyl-m-xylylene diisocyanate is not expected to be widely dispersed in the environment as a result of its usage patterns, as it is anticipated that the substance is not used directly but rather reacted into the polymers to which it is added (Cytec Industries 2005).
Once it is incorporated in the polymer, the molecular structure of the monomer is modified and therefore the integral structure of tetramethyl-m-xylylene diisocyanate would not be maintained. Even if the polymers containing this substance as a monomer can be released to the environment at different steps of their life cycle and subsequently break down to smaller components, the substance tetramethyl-m-xylylene diisocyanate itself would not be released in its structural integrity. Residual material (unreacted tetramethyl-m-xylylene diisocyanate) is also not expected to be present in the polymer because of the reactivity of this substance (see Environmental Persistence section).
Based on its physical and chemical properties (Table 2) and the results of Level III fugacity modelling (Table 3), tetramethyl-m-xylylene diisocyanate is not expected to partition to any significant degree beyond the medium to which it is emitted. This is due to the high reactivity rate (hydrolysis) of the compound overriding intermedia exchange rates. Loss of the compound from the unit world is solely a result of reaction.
| Substance Released to | Percentage of substance partitioning into each compartment | |||
|---|---|---|---|---|
| Air | Water | Soil | Sediment | |
| Air (100%) | 100.0 | 0.0 | 0.0 | 0.0 |
| Water (100%) | 2.0 | 98.0 | 0.0 | 0.0 |
| Soil (100%) | 0.0 | 0.0 | 100.0 | 0.0 |
Tetramethyl-m-xylylene diisocyanate is expected to react rapidly in both water and soil, primarily as a result of hydrolysis and also in air, as a result of photo-oxidation, with rapid reactivity with hydroxyl radicals in the atmosphere anticipated. Abiotic reactions are the dominant loss processes in the unit world for releases to air, water and soil for this compound. However, for releases to air, based on this substance's semi-volatility, some losses from atmospheric transport, via advection, are expected.
Environmental Persistence
Table 4a presents the empirical degradation data for Tetramethyl-m-xylylene diisocyanate
| Medium | Fate process | Degradation value | Degradation endpoint (unit) | Reference |
|---|---|---|---|---|
| Air | Hydrolysis | 0.527 | Half-life (days) | Cytec Industries 2005 |
| Water | Hydrolysis | 13.7 | % ThOD (28 days) | Cytec Industries 2005 |
[*] ThOD = Theoretical Oxygen Demand
The empirical biodegradation data (Cytec Industries 2005), show 13.7 % biodegradation over 28 days in a ready-biodegradation test (OECD 301 D) for tetramethyl-m-xylylene diisocyanate (Table 4a). This indicates that the half-life in water and soil is longer than 182 days (6 months) (Environment Canada 2003), if hydrolysis is not considered. However, when hydrolysis is considered, this substance's half-life in water decreases significantly, as shown by empirical hydrolysis data from Cytec Industries (2005). A robust study summary for this study is presented in Appendix 1. Aliphatic diisocyanates are expected to react with water and form inert and insoluble polyureas under "normal" conditions (Infracor GmbH 2000; Sopac and Boltromejuk 1974). Similarly, aromatic diisocyanates, which are also reactive with water, have been observed to hydrolyze rapidly, forming insoluble polyureas (Pemberton and Tury 2004; Heimbach et al. 1996; Yakabe et al. 1999; Tury et al. 2003). Under certain conditions, particularly those exhibiting high dispersion coupled with low concentrations, there is potential for the formation of diamines as a result of hydrolysis (Sopac and Boltromejuk 1974).
Environment Canada considers hydrolysis to be a primary degradation process for most substances, and therefore is concerned with potential products of hydrolysis within the context of a screening assessment. It is anticipated that in addition to insoluble ureas forming as a result of the hydrolysis of tetramethyl-m-xylylene diisocyanate, there is a potential, under certain conditions (i.e., high dispersion and low concentrations), for the formation of tetramethyl-m-xylylene diamine; if it is assumed that hydrolysis products and reactions of tetramethyl-m-xylylene diisocyanate will be similar to those of other aliphatic and aromatic diisocyanates (Pemberton and Tury 2004; Heimbach et al. 1996; Yakabe et al. 1999; Tury et al. 2003).
Experimental persistence information was not found for tetramethyl-m-xylylene diamine compounds from a literature search and biodegradation modelling was conducted on this compound. The diamine hydrolysis product is not predicted to be persistent in water, with a half-life from the BIOWIN 2000 Ultimate Survey Model estimated at 37.5 days. Using the QSAR-based weight-of-evidence approach, tetramethyl-m-xylylene diamine is not expected to be persistent in water.
Table 4b provides the biodegradation predictions for tetramethyl-m-xylylene diisocyanate. However, the extent to which these biodegradation models reflect the rapid hydrolysis of this compound and therefore provide meaningful persistence estimations in water, soil and sediment, is uncertain.
In the atmosphere, modelled data indicate that tetramethyl-m-xylylene diisocyanate is rapidly degraded via reactions with hydroxyl radicals (see Table 4b). Information on reaction rates with other oxidative species was not found, although AOPWIN 2000 did not predict this compound to be reactive with ozone (Table 4b). Despite the high reactivity of diisocyanate compounds with water, it has been observed for other diisocyanate compounds, such as toluene diisocyanate, that reaction with atmospheric water was not a significant fate process (Tury et al. 2003). Based on the evidence available, it is believed that tetramethyl-m-xylylene diisocyanate should not be considered persistent in the atmosphere based on reactions with photogenerated hydroxyl radicals. In addition, since tetramethyl-m-xylylene diisocyanate is not persistent in air, it is not expected to have long-range transport potential (LRTP).
| Medium | Fate process | Degradation value | Degradation endpoint (unit) | Reference |
|---|---|---|---|---|
| Air | Atmospheric oxidation | 1.056 | Half-life (days) | AOPWIN 2000 |
| Air | Ozone reaction | Not reactive | Half-life (days) | AOPWIN 2000 |
| Water | Hydrolysis | < 10 | Half-life (minutes) | HYDROWIN 2000 |
| Water | Biodegradation | 60 | Half-life (days) | BIOWIN 2000, Ultimate survey |
| Water | Biodegradation | 0 | Probability | TOPKAT 2004 |
| Water | Biodegradation | 0.04 | Probability | BIOWIN 2000, MITI Non-linear |
Using an extrapolation ratio of 1:1:4 for a water: soil: sediment biodegradation half-life (Boethling et al. 1995), and the biodegradation half-life of 60 days obtained from the Ultimate survey model in BIOWIN (2000), the half-life in soil is also expected to be <182 days and the half-life in sediments < 365 days. This indicates that tetramethyl-m-xylylene diisocyanate is not expected to be persistent in soil and sediment.
The weight of evidence based on the data described above indicates that tetramethyl-m-xylylene diisocyanate does not meet the persistence criteria for air (half-life in air ≥ 2 days) and water or soil (half-life in soil and water ≥ 182 days) or sediments (half-life in sediments ≥ 365 days) as set out in the Persistence and Bioaccumulation Regulations (Canada 2000).
Potential for Bioaccumulation
Experimental bioaccumulation values for this chemical were not found. It has been well documented that other aliphatic diisocyanates--as well as aromatic diisocyanates which are expected to be similarly reactive compounds--hydrolyze rapidly in the presence of moisture (Infracor GmbH 2000, Sopac and Boltromejuk 1974, Pemberton and Tury 2004, Heimbach et al. 1996, Yakabe et al. 1999, Tury et al. 2003). Bioaccumulation models cannot take into account the hydrolysis potential of substances. Therefore, modelled values for bioaccumulation offer little supporting evidence for bioaccumulation for substances that undergo near instantaneous reaction in the environment. Also, based on the high reactivity of diisocyanates, should any uptake of the parent compound occur, it is anticipated that it would be hydrolyzed and/or metabolized in the gut at rates significant enough to counter any potential for bioaccumulation.
The high reactivity of tetramethyl-m-xylylene diisocyanate with water also makes the determination of an experimental log Kow difficult. As a result, the reliability of log Kow values used as input into the bioaccumulation models is questionable.
Since no experimental bioaccumulation factor (BAF) or bioconcentration factor (BCF) data for tetramethyl-m-xylylene diisocyanate were available, a QSAR-based weight-of-evidence approach (Environment Canada 2007b), was applied using the BAF and BCF models shown in Table 5.
| Test organism | Endpoint | Value wet weight (L/kg) | Reference |
|---|---|---|---|
| Fish | BAF | 6870 | Gobas BAF T2MTL (Arnot and Gobas 2003) |
| Fish | BCF | 2700 | Gobas BCF T2LTL (Arnot and Gobas 2003) |
| Fish | BCF | 10 326 | OASIS 2005 |
| Fish | BCF | 887 | BCFWIN 2000 |
The Modified Gobas BAF middle trophic level model produced a BAF of 6870 L/kg. The results of BCF model calculations seem to indicate that this substance could have a high bioconcentration potential. However, because of the reactive nature of diisocyanates, it is anticipated that both the log Kowpredictions, and the bioaccumulation models that depend on them, likely significantly overestimate the lipophilic nature of these compounds.
To the extent that hydrolysis of tetramethyl-m-xylylene diisocyanate may form tetramethyl-m-xylylene diamine, in addition to insoluble polyureas, it is worthwhile to consider the bioaccumulation potential of the diamine hydrolysis product. An experimental log Kow for tetramethyl-m-xylylene diamine was not found, however, an estimated log Kow of 1.89 was predicted by KOWWIN (2000). This indicates that the bioaccumulation potential of this hydrolysis product of tetramethyl-m-xylylene diamine is low. Analogous bioaccumulation data from the potential hydrolysis products of similarly reactive aromatic diisocyanates would seem to support the conclusion of low bioaccumulation potential for these substances, with empirical BCFs for toluene diamine for example found to range from <5 to <50 in carp (MITI 1992).
The weight of evidence indicates that the substance tetramethyl-m-xylylene diisocyanate does not meetthe bioaccumulation criteria (BCF or BAF 5000) as set out in thePersistence and Bioaccumulation Regulations (Canada 2000).
Ecological Effects Assessment
In the Aquatic Compartment
There is modelled and experimental evidence that tetramethyl-m-xylylene diisocyanate causes harm to aquatic organisms at relatively low concentrations (Tables 6a and 6b).
| Test organism | Type of test | Endpoint | Value (mg/L) | Reference |
|---|---|---|---|---|
| Algae | Acute (96 hours) | EC50[1] | 0.36[*] | Cytec Industries 2003 |
| Daphnia | Acute (48 hours) | LC50[2] | 5.2 | Cytec Industries 2005 |
| Daphnia | Acute (48 hours) | NOEC[3] | < 1.0 | Cytec Industries 2005 |
| Fish | Acute (96 hours) | LC50[2] | 0.67 | Cytec Industries 2005 |
| Fish | Acute (96 hours) | NOEC[3] | 0.32 | Cytec Industries 2005 |
[1] LC50 - The concentration of a substance that is estimated to cause some toxic sublethal effect on 50% of the test organisms.
[2] LC50 - The concentration of a substance that is estimated to be lethal to 50% of the test organisms.
[3] NOEC - The no-observed-effect concentration is the highest concentration in a toxicity test not causing a statistically significant effect in comparison to the controls.
[*] Categorization pivotal iT value
The empirical toxicity data for tetramethyl-m-xylylene diisocyanate indicate that this substance is highly hazardous to aquatic organisms, with an LC50 to fish (Pimephales promelas) of 0.67 mg/L and an observed EC50 to algae (biomass) of 0.36 mg/L (Cytec Industries 2005; Cytec Industries 2003). A number of other chronic and acute toxicity assays also provide evidence of the moderate to high hazard of this substance to aquatic organisms (Cytec Industries 2005). A robust study summary for the 2003 Cytek Industries study is presented in Appendix 1.
Based on the expected rapid hydrolysis of this substance, the role of tetramethyl-m-xylylene diisocyanate or that of a product of its hydrolysis in directly exerting these toxic effects, is uncertain. However, the evidence in either case suggests that tetramethyl-m-xylylene diisocyanate, its hydrolysis products, or a combination of the two, is highly hazardous to aquatic organisms.
A range of aquatic toxicity predictions were obtained from the various QSAR models considered (Table 6b). These results similarly indicate that the substance is moderately to highly hazardous to aquatic organisms.
| Test organism | Type of test | Endpoint | Value (mg/L) | Model/reference |
|---|---|---|---|---|
| Fish | Acute (96 hours) | LC50[1] | 1.35 | ECOSAR 2004 |
| ≤ 0.6 | TIMES 2007 (Canadian-POPs) for Unspecified Reactive Mode | |||
| 49.76 | AIES 2003-2005 | |||
| 1.2 | TOPKAT 2004 | |||
| Daphnia | Acute (48 hours) | EC50[2] | 1.2 | TOPKAT 2004 |
[1] LC50 - The concentration of a substance that is estimated to cause some toxic sublethal effect on 50% of the test organisms.
[2] LC50 - The concentration of a substance that is estimated to be lethal to 50% of the test organisms.
In Other Environmental Compartments
No suitable ecological effects studies were found for this compound in media other than water.
Ecological Exposure Assessment
No data concerning concentrations of tetramethyl-m-xylylene diisocyanate in environmental media (air, water, soil, sediment) in Canada have been identified. Environmental concentrations are, therefore, estimated from available information, including estimated substance quantities, release rates, and receiving water bodies.
Environment Canada's Industrial Generic Exposure Tool - Aquatic (IGETA) was employed to estimate the substance concentration (reasonable worst-case) in a generic water course receiving industrial effluents (Environment Canada 2008a). The generic scenario is designed to provide these estimates based on conservative assumptions regarding the amount of chemical processed and released, the number of processing days, the sewage treatment plant removal rate, and the size of the receiving watercourse. The tool models an industrial-release scenario based on loading data from sources such as industrial surveys and knowledge of the distribution of industrial discharges in the country, and calculates a predicted environmental concentration (PEC). The equation and inputs used to calculate the PEC in the receiving water course are described in Environment Canada (2008b). Assuming a use quantity of 100 kg (which is the threshold reporting value for the 2005 section 71 notice, but which was not reached by any facility in Canada), the predicted environmental concentration in water (PEC) was 0.0006 mg/L.
Characterization of Ecological Risk
A conservative predicted no-effect concentration (PNEC) was estimated based on the NOEC to fish. The 96-hour NOEC for tetramethyl-m-xylylene diisocyanate was 0.32 mg/L. A factor of 100 was then applied to account for uncertainty in extrapolating acute to chronic (long-term) toxicity and from laboratory results to the field. The resulting PNEC is 0.0032 mg/L. When compared to the conservative PEC calculated above for industrial releases to water, the resulting risk quotient (PEC/PNEC) is 0.0006/0.0032 = 0.19. Therefore, it is estimated that concentrations of tetramethyl-m-xylylene diisocyanate in surface waters in Canada resulting from point source releases appear unlikely to cause adverse effects on populations of aquatic organisms. There is a five-fold margin of safety afforded by the conservative risk quotient to account for uncertainty associated with calculation of the PEC and PNEC.
Based on the available information, tetramethyl-m-xylylene diisocyanate does not persist in the environment and is not bioaccumulative based on criteria defined in the Persistence and Bioaccumulation Regulations(Canada 2000). Furthermore, in water this substance is expected to undergo relatively rapid hydrolysis, forming insoluble polyureas and diamines. Information on concentrations of tetramethyl-m-xylylene diisocyanate in the environment has not been identified at this time. However, the experimental ecotoxicological data indicate that tetramethyl-m-xylylene diisocyanate, and its hydrolysis products, could cause harm to aquatic organisms at relatively low concentrations in the water. Risk quotients for aquatic exposures indicate that tetramethyl-m-xylylene diisocyanate concentrations likely do not exceed concentrations associated with effects, even when using conservative scenarios and assumptions. Therefore tetramethyl-m-xylylene diisocyanate is unlikely to be causing harm to populations of aquatic organisms in Canada.
Uncertainties in Evaluation of Ecological Risk
Information on concentrations of tetramethyl-m-xylylene diisocyanate in the Canadian environment is currently lacking. However, the lack of importation or manufacture of tetramethyl-m-xylylene diisocyanate in Canada at significant volumes suggests very low releases of this chemical into the Canadian environment.
Significant uncertainties are also associated with the evaluation of physical and chemical properties and aquatic toxicity. Water solubility and log Kow are principle considerations in determining the fate and bioavailability of a substance and estimating its aquatic toxicity. Experimentally derived values for these properties were not available for tetramethyl-m-xylylene diisocyanate, as the rapid hydrolysis of aliphatic diisocyanates prevents the measurement of these properties. Similarly, due to the reactivity of this compound in the presence of moisture, the reliability of the predictions of these properties is questionable. This also has implications for the estimated bioaccumulation potential, as that endpoint is estimated from log Kow.
There is also some uncertainty associated with the products of hydrolysis, as the assumptions regarding these products are based on observations and information from other aliphatic and aromatic diisocyanates (Infracor GmbH 2000, Sopac and Boltromejuk 1974, Pemberton and Tury 2004, Heimbach et al. 1996, Yakabe et al. 1999, Tury et al. 2003). However, information from Cytec Industries (2005) for tetramethyl-m-xylylene diisocyanate indicates that the reaction of this compound with water does result in the formation of insoluble polyureas.
The aquatic toxicity assessment for this substance has some uncertainty as it is not known if the aquatic toxicity empirically observed is a result of toxic effects exerted by tetramethyl-m-xylylene diisocyanate directly (e.g., as unreacted material), its hydrolysis products, or a combination of the two. The inherent toxicity for aquatic organisms may have an additional source of uncertainty in some situations, e.g., where these concentrations exceed the solubility of the chemical in water (either experimental or predicted). Given that concentrations for both the toxicity and water solubility often vary considerably (up to several orders of magnitude), it is acknowledged that these uncertainties exist.
For the exposure assessment, the predicted environmental concentration (PEC) accounts for concentrations in water only, so exposure through soils, suspended solids and sediments is not considered. However, given the current release scenarios and quantities used in Canada, exposure is not likely to be significant at this time. Potential releases from products that could contain this substance were not considered in the exposure scenario, adding uncertainty to the exposure characterization.
Based on the information presented in this screening assessment, it is concluded that tetramethyl-m-xylylene diisocyanate is not entering the environment in a quantity or concentration or under conditions that have or may have an immediate or long-term harmful effect on the environment or its biological diversity or that constitute or may constitute a danger to the environment on which life depends.
It is therefore concluded that tetramethyl-m-xylylene diisocyanate does not meet the definition of toxic as set out in section 64 of CEPA 1999. Additionally, tetramethyl-m-xylylene diisocyanate does not meet the criteria for persistence and bioaccumulation potential as set out in the Persistence and Bioaccumulation Regulations (Canada 2000).
[AIES] Artificial Intelligence Expert System. 2003-2005. Version 1.25. Ottawa (ON): Environment Canada. Model developed by Stephen Niculescu. Environment Canada, Existing Substances Division, New Substances Division, Ottawa, K1A 0H3.
[AOPWIN] Atmospheric Oxidation Program for Windows [Estimation Model]. 2000. Version 1.91. Washington (DC): US Environmental Protection Agency, Office of Pollution Prevention and Toxics; Syracuse (NY): Syracuse Research Corporation. Exposure Assessment Tools and Models.
Arnot JA, Gobas FAPC. 2003. A generic QSAR for assessing the bioaccumulation potential of organic chemicals in aquatic food webs. QSAR Comb Sci 22(3): 337-345.
[BCFWIN] BioConcentration Factor Program for Windows [Estimation Model]. 2000. Version 2.15. Washington (DC): US Environmental Protection Agency, Office of Pollution Prevention and Toxics; Syracuse (NY): Syracuse Research Corporation. Exposure Assessment Tools and Models.
[BIOWIN] Biodegradation Probability Program for Windows [Estimation Model]. 2000. Version 4.02. Washington (DC): US Environmental Protection Agency, Office of Pollution Prevention and Toxics; Syracuse (NY): Syracuse Research Corporation. Exposure Assessment Tools and Models.
Boethling, RS, Howard, PH, Beauman, JA, Larosche, ME 1995. Factors for intermedia extrapolations in biodegradability assessment. Chemosphere. 30(4): 741-752.
Canada. 1999. Canadian Environmental Protection Act, 1999. S.C., 1999, c. 33. Canada Gazette, Part III, Vol. 22, No. 3.
Canada. 2000. Canadian Environmental Protection Act: Persistence and Bioaccumulation Regulations, P.C. 2000-348, 23 March, 2000, SOR/2000-107, Canada Gazette, Part II, Vol. 134, No. 7.
Canada, Dept. of the Environment, Dept. of Health. 2006a.Canadian Environmental Protection Act, 1999: Notice of intent to develop and implement measures to assess and manage the risks posed by certain substances to the health of Canadians and their environment. Canada Gazette, Part I, Vol. 140, No. 49.
Canada, Dept. of the Environment, Dept. of Health. 2006b.Canadian Environmental Protection Act, 1999: Notice with respect to selected substances identified as priority for action. Canada Gazette, Part I, Vol. 140, No. 9.
Canada, Dept. of Environment. 2007. Canadian Environmental Protection Act, 1999: Notice with respect to certain Batch 2 Challenge substances. Canada Gazette, Part I, Vol. 141. No. 19.
Cytec Industries. 2003. Isocyanic acid, m-phenylenediiso-propylidene, CAS# 2778-42-9 [report on the Internet]..Washington (DC): US Environmental Protection Agency, High Production Volume (HPV) Challenge Program. [cited 2008 Aug].
Cytec Industries. 2005. Test plan for TMXDI® (meta) aliphatic isocyanate: Isocyanic acid, m-phenylenediiso-propylidene, CAS# 2778-42-9 [Internet]. Washington (DC): US Environmental Protection Agency, High Production Volume (HPV) Challenge Program. [cited 2008 Aug].
[ECOSAR] Ecological Structural Activity Relationships [Internet]. 2004. Version 0.99h. Washington (DC): US Environmental Protection Agency, Office of Pollution Prevention and Toxics; Syracuse (NY): Syracuse Research Corporation. Exposure Assessment Tools and Models.
Environment Canada 2003. Guidance Manual for the Categorization of Organic and Inorganic Substances on Canada's Domestic Substances List. Gatineau (QC): Environment Canada, Existing Substances Branch.
Environment Canada. 2006. Data for selected substances collected undertheCanadian Environmental Protection Act, 1999, Section71:Notice with respect to selected substances identified as priority for action. Prepared by: Environment Canada, Health Canada, Existing Substances Program.
Environment Canada. 2007a. Data for Batch 2 substances collected undertheCanadian Environmental Protection Act, 1999, Section71:Notice with respect to certain Batch 2 Challenge substances. Data prepared by: Environment Canada, Existing Substances Program.
Environment Canada. 2007b. Guidance for conducting ecological assessments under CEPA, 1999: science resource technical series: draft module on QSARs. Reviewed draft working document. Gatineau (QC): Environment Canada, Existing Substances Division.
Environment Canada. 2008a. Guidance for conducting ecological assessments under CEPA, 1999: science resource technical series, technical guidance module: the Industrial Generic Exposure Tool - Aquatic (IGETA). Working document. Gatineau (QC): Environment Canada, Existing Substances Division.
Environment Canada. 2008b. IGETA report: CAS RN 2778-42-9, 2008-03-03. Unpublished report. Gatineau (QC): Environment Canada, Existing Substances Division.
[EQC] Equilibrium Crieterion Model. 2003. Version 2.02. Peterborough (ON): Trent University, The Canadian Environmental Modelling Centre and Chemistry - EQC Model.
Heimbach F, Jaeger K, Sporenberg W. 1996. Fate and Biological Effects of Polymeric MDI (4,4-diphenylmethane diisocyanate and homologues) in small artificial ponds. Ecotox.Environ.Safety 33: 143-153
[HYDROWIN] Hydrolysis Rates Program for Microsoft Windows [Estimation Model]. 2000. Version 1.67. Washington (DC): US Environmental Protection Agency, Office of Pollution Prevention and Toxics; Syracuse (NY): Syracuse Research Corporation. Exposure Assessment Tools and Models.
Infracor GmbH 2000. Loslichkeits- und Abbauverhalten von Isophorondiisocyanat (IPDI) in Wasser. Letter from Dr W. Schleich to Degussa AG, dated 26 June 2000 [as cited in IUCLID data set for IPDI]
[KOWWIN] Octanol-Water Partition Coefficient Program for Microsoft Windows [Estimation Model]. 2000. Version 1.67. Washington (DC): US Environmental Protection Agency, Office of Pollution Prevention and Toxics; Syracuse (NY): Syracuse Research Corporation. Exposure Assessment Tools and Models.
MITI (Ministry of International Trade & Industry). 1992. Biodegradation and Bioaccumulation Data of Existing Chemicals Based on the CSCL Japan. Chemical Products Safety Division Basic Industries Bureau, Ministry of International Trade and Industry, Edited by Chemicals Inspection & Testing Institute, Japan.
[MPBPWIN] Melting Point Boiling Point Program for Microsoft Windows [Estimation Model]. 2000. Version 1.41. Washington (DC): US Environmental Protection Agency, Office of Pollution Prevention and Toxics; Syracuse (NY): Syracuse Research Corporation. Exposure Assessment Tools and Models.
[NCI] National Chemical Inventories [database on a CD-ROM]. 2007. Issue 1. Columbus (OH): American Chemical Society, Chemical Abstracts Service. [cited 2006 Dec 11]. National Chemical Inventory (NCI) System Requirements.
[OASIS Forecast] Optimized Approach based on Structural Indices Set [Internet]. 2005. Version 1.20. Bourgas (BG): Bourgas Prof. Assen Zlatarov University, Laboratory of Mathematical Chemistry. OASIS Software.
[PCKOCWIN] Organic Carbon Partition Coefficient Program for Windows [Estimation Model]. 2000. Version 1.66. Washington (DC): US Environmental Protection Agency, Office of Pollution Prevention and Toxics; Syracuse (NY): Syracuse Research Corporation. Exposure Assessment Tools and Models.
Pemberton D, Tury B. 2004. TDI Industry Risk Assessment: Sections on physico-chemical properties, environmental exposures, and environmental effects. GIL Report 2004/E.
Sopac ED, Boltromejuk LP. 1974. Gig. Sanit. 7:10-13 [as cited in IUCLID data for HDI]
[TIMES] TIssue MEtabolism Simulator [Computer Model]. 2007. Version 2.25. Bourgas (BG): Bourgas Prof. Assen Zlatarov University, Laboratory of Mathematical Chemistry. OASIS Software.
[TOPKAT] Toxicity Prediction Program [Internet]. 2004. Version 6.2. San Diego (CA): Accelrys Software Inc.
Tury B, Pemberton D, Bailey RE. 2003. Fate and Potential Environmental Effects of methylenediphenyl diisocyanate and toluene diisocyanate released into the atmosphere. J. Air & Waste Manage.Assoc. 53:61-66.
[WATERNT] Water Solubility Program [Estimation Model]. 2002. Version 1.00. Washington (DC): US Environmental Protection Agency, Office of Pollution Prevention and Toxics; Syracuse (NY): Syracuse Research Corporation. Exposure Assessment Tools and Models.
[WSKOWWIN] Water Solubility for Organic Compounds Program for Microsoft Windows [Estimation Model]. 2000. Version 1.41 Washington (DC): US Environmental Protection Agency, Office of Pollution Prevention and Toxics; Syracuse (NY): Syracuse Research Corporation. Exposure Assessment Tools and Models.
Yakabe Y, Henderson KM, Thompson WC, Pemberton D, Tury B, Bailey RE. 1999. Fate of methylenediphenyl diisocyanate and toluene diisocyanate in the aquatic environment. Environ Sci Technol. 33 (15): 2579-2583.
| Item | Yes | No |
|---|---|---|
| Reference: Cytec Industries. 2005. Test plan for TMXDI® (meta) aliphatic isocyanate: Isocyanic acid, m-phenylenediiso-propylidene, CAS# 2778-42-9 [Internet]. Washington (DC): US Environmental Protection Agency, High Production Volume (HPV) Challenge Program. [cited 2008 Aug]. Details on the study taken from IUCLID Dataset (201-15835), Cytek Industries, 21-01-2005 |
||
| Test Substance: CAS # and name (specify, but do not assess this item): 2778-42-9; Isocyanic acid, m-phenylenediiso-propylidene | ||
| Substance purity reported? (Y/N and specify) 99.29% | x | |
| Method | ||
| References (Y/N) | x | |
| OECD, EU, national, or other standard method? (Y/N) OECD Guide-line 111 "Hydrolysis as a Function of pH" | x | |
| If not a standard method, justification of the method/protocol provided? (Y/N) | n/a | |
| Test design / conditions | ||
| Study type (e.g., hydrolysis, biodegradation, etc. - specify, but do not assess): hydrolysis | ||
| Conditions type (aerobic or anaerobic - specify, but do not assess): n/a | ||
| Test medium (air, water, soil, or sediment - specify, but do not assess): water | ||
| Information on stability of the substance in the media of concern available? (do not assess this item): No | ||
| Information on controls (Y/N and specify, positive or negative) positive & negative | × | |
| Number of replicates (Y/N and specify) | × | |
| Temperature (Y/N and specify) 25°C | × | |
| Test duration (Y/N and specify) 24 hours | × | |
| Analytical method / technique used (Y/N) | × | |
| For photodegradation only | ||
| Reactants of gas-phase reactions (specify, but do not assess this item) | ||
| Light source (Y/N and specify) | ||
| Light spectrum and/or relative intensity based on sunlight intensity (Y/N) | ||
| For hydrolysis only | ||
| Measured concentrations reported? (Y/N) | × | |
| pH values reported? (Y/N and specify) | × | |
| For biodegradation only | ||
| Ready or inherent biodegradation? (specify, but do not assess this item) ready | ||
| Substance concentration (Y/N) | ||
| Inoculum source (Y/N) | ||
| Inoculum concentrationor number of microorganisms (Y/N) | ||
| Results | ||
| Endpoints / values / units (do not assess this item): t1/2 pH4: 0.4 h; t1/2 pH7: 0.4 h; t1/2 pH9: 0.3 h | ||
| Information on breakdown products available? (do not assess this item) No | ||
| Overall score: 7/10= 70% | ||
| EC Reliability code: 2 | ||
| Reliability category (high, satisfactory, low): Satisfactory | ||
| Comments: | ||
| Item | Yes | No |
|---|---|---|
| Reference: Cytec Industries. 2003. Isocyanic acid, m-phenylenediiso-propylidene, CAS# 2778-42-9 [report on the Internet]..Washington (DC): US Environmental Protection Agency, High Production Volume (HPV) Challenge Program. [cited 2008 Aug]. | ||
| Test Substance: CAS 2778-42-9 and name (specify, but do not assess this item): 2778-42-9; Isocyanic acid, m-phenylenediiso-propylidene | ||
| * Substance purity reported? (Y/N and specify) 98-99% | × | |
| Persistence/stability of test substance in aquatic solution reported? (Y/N) | × | |
| Method | ||
| References (Y/N) | × | |
| * OECD, EU, national, or other standard method? (Y/N) Based on methods outlined in the Committee on Methods for Toxicity Test with Aquatic Organisms, USEPA 660/3-75009. ABC Laboratories Protocol 7601. |
× | |
| If not a standard method, justification of the method/protocol provided? (Y/N) | n/a | |
| * GLP (Good Laboratory Practice) (Y/N) | × | |
| Test organisms (specify common and/or Latin names)(Pimephales promelas, Fathead minnow) | ||
| Latin or both Latin and common names reported? (Y/N) | × | |
| Life cycle age / stage of test organism (Y/N) | × | |
| Sex (Y/N) | × | |
| Length and/or weight of test organisms (Y/N) | × | |
| Number of test organisms per replicate (Y/N)10 | × | |
| Food type / feeding periods (acclimation/during test) (Y/N) | × | |
| Test design / conditions | ||
| Test type - acute or chronic (specify, but do not assess this item): acute | ||
| Experiment type (laboratory or field) specified? (Y/N) | × | |
| System type (static, semi-static, flow-through) (Y/N) static | × | |
| Negative or positive controls? (Y/N and specify) negative | × | |
| Number of replicates (including controls) and concentrations (Y/N) 10 per test concentration | × | |
| Exposure pathways (food, water, both) (Y/N) | × | |
| Exposure duration (Y/N and specify) 96h | × | |
| * Measured concentrations reported? | × | |
| Exposure media conditions (temperature, pH, electrical conductivity, hardness, TOC, DOC, DO, major cations and anions; other) (Y/N) | × | |
| Was pH within 6-9 range? (do not assess this item) yes | ||
| Was temperature within 5-28 °C range? (do not assess this item) yes | ||
| Photoperiod and light intensity (Y/N) | × | |
| Stock and test solution preparation (Y/N) | × | |
| Information on emulsifiers used for poorly soluble / unstable substances (Y/N) | n/a | |
| Analytical monitoring intervals (Y/N) daily fish observation and water quality parameters of temperature, dissolved oxygen and pH were measured throughout the test | × | |
| Statistical methods used (Y/N) | × | |
| Results | ||
| Toxicity endpoints / values / units (specify, but do not assess this item 96h NOEC 0.32 mg/L | ||
| Other endpoints reported (e.g. BCF/BAF, LOEC/NOEC, etc.) (specify, but do not assess this item): LC50 (96h) 0.67 mg/L | ||
| * Was toxicity value below the chemical's water solubility? (Y/N) water solubility unknown | n/a | |
| Other adverse effects (carcinogenicity, mutagenicity, etc. Do not assess this item) | n/a | |
| Score: major items - 3 / 4; overall score: 18/23=78% | ||
| EC Reliability code:2 | ||
| Reliability category (high, satisfactory, low): satisfactory | ||
| Comments: Water solubility was not reported; nominal concentrations were used. | ||