Screening assessment hexanoic acid, 2-ethyl-, calcium salt and hexanoic acid, 2-ethyl-, 2-ethylhexyl ester
Official title: Screening Assessment Hexanoic acid, 2-ethyl-, calcium salt and Hexanoic acid, 2-ethyl-, 2-ethylhexyl ester
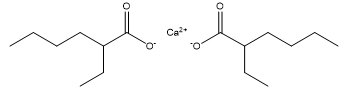
Hexanoic acid, 2-ethyl-, calcium salt
Chemical abstract service registry number 136-51-6
Cat. No: En14-348/2018E-PDF
ISBN: 978-0-660-28825-3
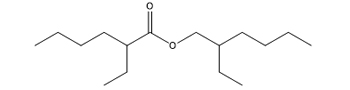
Hexanoic acid, 2-ethyl-, 2-ethylhexyl ester
Chemical abstract service registry number 7425-14-1
Cat. No: En14-349/2018E-PDF
ISBN: 978-0-660-28830-7
Environment and Climate Change Canada Health Canada
December 2018
Synopsis
Pursuant to sections 68 and 74 of the Canadian Environmental Protection Act, 1999 (CEPA), the Minister of the Environment and the Minister of Health have conducted a screening assessment of calcium 2-ethylhexanoate and 2-ethylhexyl 2-ethylhexanoate. These substances were identified as priorities for assessment as they met categorization criteria under subsection 73(1) of CEPA or were considered a priority on the basis of other human health concerns. The Chemical Abstracts Service Registry Numbers (CAS RNFootnote 1 ), their Domestic Substances List (DSL) names and their common names are listed in the table below.
CAS RN |
DSL name |
Common name |
|---|---|---|
136-51-6 |
Hexanoic acid, 2-ethyl-, calcium salt |
Calcium 2-ethylhexanoate |
7425-14-1 a |
Hexanoic acid, 2-ethyl-, 2-ethylhexyl ester |
2-Ethylhexyl 2-ethylhexanoate |
a This substance was not identified under subsection 73(1) of CEPA but was included in this assessment as it was considered as a priority on the basis of other human health concerns.
In 2011, there were no reports of manufacture of calcium 2-ethylhexanoate above the reporting threshold of 100 kg, but between 10 000 and 100 000 kg of calcium 2-ethylhexanoate was imported into Canada. There were no reports of manufacture or import for 2-ethylhexyl 2-ethylhexanoate above the reporting threshold of 100 kg for the same year. Calcium 2-ethylhexanoate is used predominantly as an additive in interior and exterior paints. It is also reported to be used in the manufacture of food packaging materials. 2-ethylhexyl 2-ethylhexanoate is an ingredient in cosmetics.
The ecological risks of calcium 2-ethylhexanoate and 2-ethylhexyl 2-ethylhexanoate were characterized using the ecological risk classification of organic substances (ERC). The ERC is a risk-based approach that employs multiple metrics for both hazard and exposure based on weighted consideration of multiple lines of evidence for determining risk classification. Hazard profiles based primarily on metrics regarding mode of toxic action, chemical reactivity, food web-derived internal toxicity thresholds, bioavailability, and chemical and biological activity are established. Metrics considered in the exposure profiles include potential emission rate, overall persistence, and long-range transport potential. A risk matrix is then used to assign a low, moderate or high level of potential concern for substances based on their hazard and exposure profiles. The ERC identified calcium 2-ethylhexanoate and 2-ethylhexyl 2-ethylhexanoate as having low potential to cause ecological harm.
Considering all available lines of evidence presented in this screening assessment, there is low risk of harm to organisms and the broader integrity of the environment from calcium 2-ethylhexanoate and 2-ethylhexyl 2-ethylhexanoate. It is concluded that calcium 2-ethylhexanoate and 2-ethylhexyl 2-ethylhexanoate do not meet the criteria under paragraphs 64(a) or (b) of CEPA as they are not entering the environment in a quantity or concentration or under conditions that have or may have an immediate or long-term harmful effect on the environment or its biological diversity or that constitute or may constitute a danger to the environment on which life depends.
In Canada, calcium 2-ethylhexanoate may be present in certain food packaging materials. 2-Ethylhexyl 2-ethylhexanoate was reported as a volatile component in a limited number of food samples collected outside of Canada with very low concentrations. Exposure of the general population to calcium 2-ethylhexanoate and 2-ethylhexyl 2-ethylhexanoate from food is expected to be negligible.
Dermal exposure to calcium 2-ethylhexanoate may occur from the use of interior and exterior household paints, and to 2-ethylhexyl 2-ethylhexanoate from the use of certain cosmetics, including foot lotion and face make-up. Substance-specific health effects data for the relevant route and durations of exposure were not identified. However, 2-ethylhexanoic acid (2-EHA) was selected as an analogue for characterization of potential health effects of both substances. Laboratory studies of exposure via the oral route identified liver and developmental effects. Margins of exposure between critical effect levels in laboratory studies and estimates of dermal exposure to calcium 2-ethylhexanoate were considered adequate to address uncertainties in the health effects and exposure databases. Margins of exposure between critical effect levels in laboratory studies and estimates of dermal exposure to 2-ethylhexyl 2-ethylhexanoate were considered potentially inadequate to address uncertainties in the health effects and exposure databases.
On the basis of available information, it is concluded that calcium 2-ethylhexanoate does not meet the criteria under paragraph 64(c) of CEPA as it is not entering the environment in a quantity or concentration or under conditions that constitute or may constitute a danger in Canada to human life or health.
On the basis of available information, it is concluded that 2-ethylhexyl 2-ethylhexanoate meets the criteria under paragraph 64(c) of CEPA as it is entering or may enter the environment in a quantity or concentration or under conditions that constitute or may constitute a danger in Canada to human life or health.
Therefore, it is concluded that calcium 2-ethylhexanoate does not meet any of the criteria under section 64 of CEPA and that 2-ethylhexyl 2-ethylhexanoate meets one or more of the criteria set out in section 64 of CEPA.
2-ethylhexyl 2-ethylhexanoate does not meet the persistence criteria but does meet the bioaccumulation criteria as set out in the Persistence and Bioaccumulation Regulations of CEPA.
1. Introduction
Pursuant to sections 68 and 74 of the Canadian Environmental Protection Act, 1999 (CEPA) (Canada 1999), the Minister of the Environment and the Minister of Health have conducted a screening assessment of calcium 2-ethylhexanoate and 2-ethylhexyl 2-ethylhexanoate to determine whether these substances present or may present a risk to the environment or to human health. These two substances were identified as priorities for assessment as they met categorization criteria under subsection 73(1) of CEPA or were considered a priority on the basis of other human health concerns (ECCC, HC [modified 2007]). Hexanoic acid, 2-ethyl-, 2-ethylhexyl ester has been identified by other jurisdictions as a concern due to developmental effects on laboratory animals.
This assessment includes two substances that are based on the parent structure 2-ethylhexanoic acid (2-EHA). 2-EHA was previously assessed under CEPA (Environment Canada, Health Canada 2011). Although not a part of the current assessment, 2-EHA is used as an analogue for read-across purposes, within the hazard context. The screening assessment report for 2-EHA (Environment Canada, Health Canada 2011) will be used to inform this assessment.
The ecological risks of calcium 2-ethylhexanoate and 2-ethylhexyl 2-ethylhexanoate were characterized using the ecological risk classification of organic substances (ERC) (ECCC 2016a). The ERC describes the hazard of a substance using key metrics including mode of action, chemical reactivity, food-web derived internal toxicity, bioavailability, and chemical and biological activity and considers the possible exposure of organisms in the aquatic and terrestrial environments based on factors including potential emission rates, overall persistence and long-range transport potential in air. The various lines of evidence are combined to identify substances which warrant further evaluation of their potential to cause harm to the environment or which have a low likelihood of causing harm to the environment.
This screening assessment includes consideration of information on chemical properties, environmental fate, hazards, uses and exposure, including additional information submitted by stakeholders. Relevant data were identified up to April 2016. Empirical data from key studies, as well as some results from models, were used to reach conclusions. When available and relevant, information presented in assessments from other jurisdictions was considered.
This screening assessment was prepared by staff in the CEPA Risk Assessment Program at Health Canada and Environment and Climate Change Canada and incorporates input from other programs within these departments. The draft of this screening assessment (published March 25, 2017) was subject to a 60-day public comment period. The ERC approach (published July 30, 2016) was peer-reviewed, and was also subject to a 60-day public comment period. While external comments were taken into consideration, the final content and outcome of the screening assessment remain the responsibility of Environment and Climate Change Canada and Health Canada.
This screening assessment focuses on information critical to determining whether a substance meets the criteria as set out in section 64 of CEPA by examining scientific information and incorporating a weight of evidence approach and precautionFootnote 2 . The screening assessment presents the critical information and considerations on which the conclusion is based.
2. Identity of substances
The Chemical Abstracts Service Registry Numbers (CAS RNFootnote 3 ), Domestic Substances List (DSL) names and common names for the individual substances are presented in Table 2‑1.
Calcium 2-ethylhexanoate and 2-ethylhexyl 2-ethylhexanoate are both based on the parent structure 2-EHA. Calcium 2-ethylhexanoate contains the conjugate base of 2-EHA, and 2-ethylhexyl 2-ethylhexanoate is the ester of 2-EHA and 2-ethylhexanol.
CAS RN |
DSL name (common name) |
Chemical structure and molecular formula |
Molecular weight (g/mol) |
|---|---|---|---|
136-51-6 |
Hexanoic acid, 2-ethyl-, calcium salt |
|
326.49 |
7425-14-1 |
Hexanoic acid, 2-ethyl-, 2-ethylhexyl ester |
|
256.43 |
3. Physical and chemical properties
A summary of modelled physical and chemical properties of the substances are presented in Table 3‑1. Additional physical and chemical properties are presented in ECCC 2016b. No empirical data were identified.
Property |
Value for calcium 2-ethylhexanoate |
Value for 2-ethylhexyl 2-ethylhexanoate |
|---|---|---|
Melting point (oC) |
116 |
21 |
Boiling point (oC) |
403 |
293 |
Water solubility (g/mL) |
5.06 × 10−6 |
4.93 × 10−8 |
Vapour pressure (Pa) |
1.15 × 10−4 (8.65 × 10−7 mm Hg) |
0.369 |
Henry’s law constant (Pa m3/mol) |
7.12 × 10−3 |
7.07 × 102 – 1.88 × 103 |
log Kow (dimensionless) |
3.88 |
6.61 |
log Koc (dimensionless) |
2.375a |
3.832a |
log Koc (dimensionless) |
2.613b |
4.516b |
Abbreviations: Kow, octanol–water partition coefficient; Koc, organic carbon–water partition coefficient
a Modelled using the MCI method
b Modelled using the Kow method
4. Sources
Both substances have been included in a survey pursuant to section 71 of CEPA (Canada 2012). Table 4‑1 presents a summary of the total manufacture and total import quantities for the substances.
Common name |
Total Manufacture (kg) |
Total lmports (kg) |
Survey Reference |
|---|---|---|---|
Calcium 2-ethylhexanoate |
0 |
10,000 - 100,000 |
Environment Canada 2013 |
2-Ethylhexyl 2-ethylhexanoate |
0 |
0 |
Environment Canada 2013 |
a Based on information submitted pursuant to section 71 of CEPA under Phase 2 of the DSL IU (Environment Canada 2013). Values reflect quantities reported in response to surveys. See survey for specific inclusions and exclusions (schedules 2 and 3) (Canada 2012).
In the United States, the national production volume for calcium 2-ethylhexanoate and 2-ethylhexyl 2-ethylhexanoate in 2011 was approximately 214 100 kilograms (472 600 pounds) and 35 300 kilograms (78 000 pounds), respectively (CDAT [modified 2014]).
2-Ethylhexyl 2-ethylhexanoate was reported as a naturally-occurring volatile component in certain food samples collected outside of Canada, including wild peanuts (Cherif et al. 2013), muskmelon (Priyanka et al. 2015), roasted barley (Bianchi et al. 2007), beef (Tansawat et al. 2013), and various types of herbs in the genera Heracleum (Tosun et al. 2008; Akcina et al. 2013; Radjabian et al. 2014), Tordylium (Özeka et al. 2007; Tosun et al. 2010) and Zosima (Razavi and Nejad-Ebrahimi 2010). The concentrations of 2-ethylhexyl 2-ethylhexanoate reported in those foods are generally very low.
5. Uses
In Canada, calcium 2-ethylhexanoate is imported for use as an additive for paints and coatings (Environment Canada 2013).
A recent survey did not identify uses of 2-ethylhexyl 2-ethylhexanoate (Environment Canada 2013). However, based on notifications submitted to Health Canada under the Cosmetic Regulations, 2-ethylhexyl 2-ethylhexanoate is used in certain cosmetic products in Canada, including face make-up and foot lotion (personal communication, email from Consumer Product Safety Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, dated January 20, 2016; unreferenced).
Additional uses for calcium 2-ethylhexanoate and 2-ethylhexyl 2-ethylhexanoate in Canada are listed in Table 5‑1.
Use |
Calcium 2-ethylhexanoate |
2-Ethylhexyl 2-ethylhexanoate |
|---|---|---|
Food additivea |
N |
N |
Food packaging materialsb |
Y (used in the manufacture of resins with direct food contact, and in printing inks with no direct food contact) |
N |
Incidental additivesb |
Y (component of no-rinse aid for the drying cycle of dishwasher machines) |
N |
Drug Product Databasec |
N |
N |
Natural Health Products Ingredients Databased |
N |
N |
Licensed Natural Health Products Database being present as a medicinal or non-medicinal ingredient in natural health products in Canadae |
N |
N |
List of Prohibited and Restricted Cosmetic Ingredientsf |
N |
N |
Notified to be present in cosmetics, based on notifications submitted under the Cosmetic Regulations to Health Canadag |
N |
Y |
Pest control products registered in Canadah |
Y (formulant; antifouling paints) |
N |
Abbreviations: N, NO; Y, YES
a Health Canada (modified 2013)
b Personal communication, emails from Food Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, dated February and April 2016; unreferenced
c DPD (modified 2015)
d NHPID (modified 2016)
e LNHPD (modified 2016)
f Health Canada (modified 2015)
g Personal communication, emails from Consumer Product Safety Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, dated December 2015 and January 2016; unreferenced
h Personal communication, email from Risk Management Bureau, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, dated February 5, 2016; unreferenced
Globally, metal 2-ethylhexanoates are primarily used as paint driers, which accelerate the drying process for paints and coatings (Landau 2000). Globally, 2-ethylhexyl 2-ethylhexanoate has reported uses as an emollient in cosmetics (Fiume et al. 2015; CosIng 2016). In the European Union, 2-ethylhexyl 2-ethylhexanoate is prohibited for use in cosmetic products due to its classification as a Category 2 reprotoxic substance (CosIng [modified 2015]). Criteria for assigning CMR classifications to substances in the European Union are described in European Union Regulation (EC) No 1272/2008 (EU 2008); the use of substances with Category 2 CMR classifications in cosmetic products is prohibited under European Union Regulation (EC) No 1223/2009 (EU 2009), although these substances may be used in cosmetic products if an evaluation by the Scientific Committee on Consumer Safety (SCCS) has found the substance safe for use in cosmetic products (EU 2009). As of December 2017, the SCCS has not carried out a safety evaluation of 2-ethylhexyl 2-ethylhexanoate in cosmetics.
6. Environmental fate and behaviour
6.1 Environmental persistence
Calcium 2-ethylhexanoate and 2-ethylhexyl 2-ethylhexanoate have a low predicted overall persistence (Pov) in the environment according to ERC (ECCC 2016b) and are not expected to be persistent in the environment.
6.2 Potential for bioaccumulation
On the basis of low bioconcentration factors and log Kow (ECCC 2016b) calcium 2-ethylhexanoate is not expected to significantly bioaccumulate in organisms. On the basis of a modelled bioconcentration factor value of about 40 000 L/kg and a log Kow of 6.6 (ECCC 2016b), 2-ethylhexyl 2-ethylhexanoate is expected to bioaccumulate significantly in organisms.
7. Potential to cause ecological harm
7.1 Characterization of ecological risk
The ecological risk of calcium 2-ethylhexanoate and 2-ethylhexyl 2-ethylhexanoate was characterized using the ecological risk classification of organic substances (ERC) (ECCC 2016a). The ERC is a risk-based approach that employs multiple metrics for both hazard and exposure based on weighted consideration of multiple lines of evidence for determining risk classification. The various lines of evidence are combined to discriminate between substances of lower or higher potency and lower or higher potential for exposure in various media. This approach reduces the overall uncertainty with risk characterization compared to an approach that relies on a single metric in a single medium (e.g., LC50) for characterization. The following summarizes the approach, which is described in detail in ECCC 2016a.
Data on physical-chemical properties, fate (chemical half-lives in various media and biota, partition coefficients, fish bioconcentration), acute fish ecotoxicity, and chemical import or manufacture volume in Canada were collected from scientific literature, from available empirical databases (e.g., OECD QSAR Toolbox) and from responses to surveys under section 71 of CEPA, or they were generated using selected quantitative structure-activity relationship (QSAR) or mass-balance fate and bioaccumulation models. These data were used as inputs to other mass-balance models or to complete the substance hazard and exposure profiles.
Hazard profiles based primarily on metrics regarding mode of toxic action, chemical reactivity, food web-derived internal toxicity thresholds, bioavailability, and chemical and biological activity were established. Exposure profiles were also composed of multiple metrics including potential emission rate, overall persistence, and long-range transport potential. Hazard and exposure profiles were compared to decision criteria in order to classify the hazard and exposure potential for each organic substance as low, moderate or high. Additional rules were applied (e.g., classification consistency, margin of exposure) to refine the preliminary classifications of hazard or exposure.
A risk matrix was used to assign a low, moderate or high classification of potential risk for each substance based on its hazard and exposure classifications. ERC classifications of potential risk were verified using a two-step approach. The first step adjusted the risk classification outcomes from moderate or high to low for substances that had a low estimated rate of emission to water after wastewater treatment, thus representing a low potential for exposure. The second step reviewed low risk potential classification outcomes using relatively conservative, local-scale (i.e., in the area immediately surrounding a point-source of discharge) risk scenarios, designed to be protective of the environment, to determine whether the classification of potential risk should be increased.
ERC uses a weighted approach to minimize the potential for both over and under classification of hazard and exposure and subsequent risk. The balanced approaches for dealing with uncertainties are described in greater detail in ECCC 2016a. The following describes two of the more substantial areas of uncertainty. Error in empirical or modeled acute toxicity values could result in changes in classification of hazard, particularly metrics relying on tissue residue values (i.e., mode of toxic action), many of which are predicted values from QSAR models. However, the impact of this error is mitigated by the fact that overestimation of median lethality will result in a conservative (protective) tissue residue value used for critical body residue (CBR) analysis. Error of underestimation of acute toxicity will be mitigated through the use of other hazard metrics such as structural profiling of mode of action, reactivity and/or estrogen binding affinity. Changes or errors in chemical quantity could result in differences in classification of exposure as the exposure and risk classifications are highly sensitive to emission rate and use quantity. The ERC classifications thus reflect exposure and risk in Canada based on what is believed to be the current use quantity, and may not reflect future trends.
Critical data and considerations used to develop the substance-specific profiles for calcium 2-ethylhexanoate and 2-ethylhexyl 2-ethylhexanoate, and the hazard, exposure and risk classification results, are presented in ECCC (2016b).
The hazard and exposure classifications for calcium 2-ethylhexanoate and 2-ethylhexyl 2-ethylhexanoate are summarized in Table 6.1.
Substance |
ERC hazard classification |
ERC exposure classification |
ERC classification |
|---|---|---|---|
Calcium 2-ethylhexanoate |
low |
low |
low |
2-Ethylhexyl 2-ethylhexanoate |
low |
low |
low |
Abbreviation: ERC, Ecological Risk Classification of organic substances
On the basis of low hazard and low exposure classifications according to ERC for calcium 2-ethylhexanoate and 2-ethylhexyl 2-ethylhexanoate, these substances were classified as having a low potential for ecological risk. It is therefore unlikely that these substances result in concerns for organisms or the broader integrity of the environment in Canada.
8. Potential to cause harm to human health
8.1 Exposure assessment
Environmental media
No reports of environmental monitoring of calcium 2-ethylhexanoate or 2-ethylhexyl 2-ethylhexanoate in environmental media or dust in Canada or elsewhere were identified.
Modelling using the quantities reported in Canadian commerce for 2011 (Environment Canada 2013) was performed with ChemCAN (ChemCAN 2003) to estimate environmental concentrations of calcium 2-ethylhexanoate. The Mixedwood Plains region in Ontario was chosen as it is the most industrialized and populated area of Canada. No removal of calcium 2-ethylhexanoate through wastewater treatment plants was assumed for the derivation of aquatic release estimates; this assumption was used as a first-pass in order to determine the most conservative estimate of exposure, without having to further refine. From the estimated environmental concentrations, upper-bound intake estimates were calculated for age groups ranging from 0–0.5 years to 60+ years. Intake estimates for all age groups were below 1 ng/kg/day, indicating that exposure to calcium 2-ethylhexanoate from environmental media is negligible. No further refinement to exposure estimates was therefore necessary.
Based on information submitted pursuant to section 71 of CEPA, there were no reports of manufacture or import of 2-ethylhexyl 2-ethylhexanoate above the reporting threshold of 100 kg (Environment Canada 2013). As such, exposure of the general population to 2-ethylhexyl 2-ethylhexanoate through environmental media is not expected.
Food
In Canada, calcium 2-ethylhexanoate is present in certain food packaging materials with direct food contact as a result of its potential use in the manufacture of resins. It can also be used in printing inks, but such use would have no direct contact with food. It was also identified as a component of a no-rinse aid used in the drying cycle of dishwasher machines. For those uses with direct food contact, exposure to the general population is expected to be negligible (personal communication, email from Food Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, dated April 15, 2016; unreferenced).
No occurrence data regarding 2-ethylhexyl 2-ethylhexanoate in food in Canada were identified. It was reported as a volatile component of certain foreign food samples such as wild peanuts (Cherif et al. 2013), muskmelon (Priyanka et al. 2015), roasted barley (Bianchi et al. 2007), and beef (Tansawat et al. 2013). Given the very low concentration present and the limited number of foods in which 2-ethylhexyl 2-ethylhexanoate has been detected, together with the fact that the substance is volatile, with expected losses during food preparation, the level of exposure from these sources is likely to be negligible. Also, it is unlikely that many of the foods in which 2-ethylhexyl 2-ethylhexanoate has been reported to be present would be consumed by the general population of Canadians (personal communication, email from Food Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, dated April 15, 2016; unreferenced).
On the basis of these considerations, exposure of the general population of Canadians to calcium 2-ethylhexanoate and 2-ethylhexyl 2-ethylhexanoate from food is considered negligible.
Products
Calcium 2-ethylhexanoate is also present as a drier in interior and exterior paints, including spray paints, which would be used infrequently. Concentrations of calcium 2-ethylhexanoate in paint were reported to be <0.5% w/w (Environment Canada 2013; Canada 2012). Based on the default exposure scenarios in ConsExpo (RIVM 2007), potential dermal exposure during brush painting is higher than that of spray painting. Accordingly, brush painting was chosen as the sentinel scenario to provide an upper-bounding estimate for dermal exposure to calcium 2-ethylhexnoate while painting. Conservative modelling using ConsExpo gave an estimated dermal exposure of 0.25 mg/kg per use, based on a solvent-rich paint containing 0.5% w/w of calcium 2-ethylhexanoate. Inhalation exposure was also considered for painting, including the use of spray paints (i.e., can or spray gun) and was modelled using conservative ConsExpo parameters. Given the low volatility of calcium 2-ethylhexanoate, dermal exposure was determined to be the primary route of exposure for painting scenarios.
Based on notifications submitted under the Cosmetics Regulations to Health Canada, 2-ethylhexyl 2-ethylhexanoate is used in certain cosmetic products in Canada, including face make-up and foot lotion (personal communication, email from Consumer Product Safety Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, dated January 20, 2016; unreferenced). Cosmetic uses could result in daily exposures. Conservative modelling using ConsExpo (ConsExpo 2006), including the assumption of 100% dermal absorption of 2-ethylhexyl 2-ethylhexanoate, gave estimated systemic exposures via the dermal route of exposure. Complete dermal absorption was used here so as to be equivalent to oral absorption (due to the selection of a critical oral toxicological study for risk characterization, as described below). These systemic exposures were determined to be 0.37 to 1.10 mg/kg-bw/day from the use of foot lotion containing 1–3% w/w of 2-ethylhexyl 2-ethylhexanoate (use frequency of 2 times per day), and 0.91 to 3.05 mg/kg-bw/day from the use of face make-up containing 3–10% w/w of 2-ethylhexyl 2-ethylhexanoate (use frequency of 1.8 times per day).
Details of the parameters used in modelling are provided in Appendix A.
8.2 Health effects assessment
Substance-specific health effects data were not identified for calcium 2-ethylhexanoate or 2-ethylhexyl 2-ethylhexanoate. However, Environment Canada and Health Canada (2011) summarized the health effects literature for related substances 2-EHA and 2-ethylhexanol (i.e., CAS RNs 149-57-5 and 104-76-7, respectively) and characterized the hazard for 2-EHA. 2-EHA and 2-ethylhexanol are considered relevant to the two substances currently being evaluated (see Identity of Substances section) and were used as potential analogues where critical health effects data were required. Use of 2-EHA as an analogue for these substances is supported by a recent assessment by the Australian Government Department of Health (AGDH 2013), which used a similar approach when assessing 2-ethylhexyl 2-ethylhexanoate. It is also consistent with the approach taken in the European Union, where the European Chemicals Agency classified 2-ethylhexyl 2-ethylhexanoate and calcium 2-ethylhexanoate as Reproductive Category 2 substances (suspected of damaging the unborn child [developmental toxicity]) under the Classification, Labelling and Packaging Regulation (ECHA c2007-2015); these GHS classifications are likewise based on read-across from 2-EHA (also a Reproductive Category 2 substance). As a result, the screening assessment by Environment Canada and Health Canada (2011) was used to inform the hazard characterization of calcium 2-ethylhexanoate and 2-ethylhexyl 2-ethylhexanoate.
A literature search for 2-EHA and 2-ethylhexanol was conducted from the year of the health effects literature cut-off identified in the above-noted report (i.e., January 2010) to February 2016. Although the Australian Government Department of Health’s assessment of 2-ethylhexyl 2-ethylhexanoate was consulted, it utilized the same studies as those described in Environment Canada and Health Canada (2011). No new health effects studies that could impact the risk characterization (i.e., result in different critical endpoints or lower points of departure than those stated in Environment Canada and Health Canada (2011) were identified.
The critical endpoints and corresponding effect levels for the analogues to be used for risk characterization, as cited directly from Environment Canada and Health Canada (2011), are summarized below.
For 2-ethylhexanol, a developmental study was conducted in rats, via the dermal route of exposure. Test animals were exposed during days 6-15 of gestation to 0, 252, 420, 840, 1680 or 2520 mg/kg-bw/day (25 dams per dose). The critical effect level and corresponding hazard endpoint was a NOAEL of 2520 mg/kg-bw/day, based on no treatment-related increases in external, visceral or skeletal malformations or variations in fetuses (decreased body weight gain in dams observed at 1680 mg/kg-bw/day) (Bushy Run 1989; Fisher et al. 1989; Tyl et al. 1992, as cited in Environment Canada, Health Canada 2011).
For 2-ethylhexanol, a developmental study was conducted in rats, via the oral route of exposure (gavage). Test animals were exposed during days 6-15 of gestation to 0, 130, 650 or 1300 mg/kg-bw/day (10 dams per dose). The critical effect level and corresponding hazard endpoint was a LOAEL of 1300 mg/kg-bw/day for skeletal malformations, variations, and retardations in fetuses (BASF AG 1991l, as cited in Environment Canada, Health Canada 2011).
For 2-EHA, a developmental study was conducted in rats, via the oral route of exposure (drinking water). Test animals were exposed during days 6-19 of gestation to 0, 100, 300 or 600 mg/kg-bw/day (20 or 21 dams per dose). The critical effect level and corresponding hazard endpoint was a LOAEL of 100 mg/kg-bw/day for skeletal variations (wavy ribs and reduced cranial ossifications) and skeletal malformations (club foot) in fetuses (Pennanen et al. 1992, as cited in Environment Canada, Health Canada 2011).
Although a dermal developmental study was identified for 2-ethylhexanol (see above), there were no similar dermal developmental studies for 2-EHA. When comparing the critical effect levels from the oral developmental studies for 2-EHA and 2-ethylhexanol, 2-EHA appears to be the more potent of the two hydrolysis products. Therefore, it was determined that the oral developmental study for 2-EHA would be selected as the basis for hazard and risk characterization rather than the dermal study for 2-ethylhexanol.
The developmental effects of 2-EHA exposure in laboratory animals appear to be secondary. According to Fiume et al. (2015), exposure to 2-EHA via the oral route causes liver toxicity in dams, and this initiates a multitude of effects which eventually leads to zinc accumulation in the liver. Zinc accumulation in dams results in a concomitant deficiency in the developing embryo, and it is suggested that this is the root cause of developmental toxicity. This is supported by the observation that zinc supplementation of dams results in reduced developmental toxicity, and further zinc deficiency results in more significant developmental effects in the fetuses. Additional support was found in a study that involved administering di-2-ethylhexyl terephthalate (DEHT), a 2-EHA precursor via 2-ethylhexanol, to rats. This resulted in a sufficiently slow metabolic conversion rate that allowed clearance of 2-EHA before sufficient levels could cause liver toxicity and resultant zinc accumulation (Fiume et al. 2015).
Environment Canada and Health Canada (2011) state that 2-EHA has been shown to induce peroxisome proliferation in rodents and further report that humans are generally expected to be less susceptible than rodents to peroxisome proliferation-related health effects induced by peroxisome proliferator-activated receptor alpha (PPARα). Although it is acknowledged that any developmental toxicity related to peroxisome proliferation is less likely to be relevant to humans, the exact nature of this mode of action remains to be established for 2-EHA. Indeed, according to Fiume et al. (2015), developmental toxicity from exposure to 2-EHA is, at least in part, related to hepatic zinc levels, and thus is unlikely to be related exclusively to peroxisome proliferation.
For 2-EHA, a short-term repeated-dose study was conducted in rats, via the oral route of exposure (diet). Test animals were exposed for 15 days to 0, 706, 1351 or 2276 mg/kg-bw/day (male) and 0, 756, 1411 or 2658 mg/kg-bw/day (female) (5 animals/sex/dose). The critical effect level and corresponding hazard endpoint was a LOAEL of 706 mg/kg-bw/day for a dose-dependent increase in liver weight (both sexes; accompanied by histological changes in the liver at the intermediate and highest doses) (Eastman Kodak 1987c, as cited in Environment Canada, Health Canada 2011).
With regards to absorption, Environment Canada and Health Canada (2011) cite toxicokinetic studies that indicate that 2-EHA and 2-ethylhexanol are rapidly and extensively absorbed from the gastrointestinal tract following oral exposure in rats (English et al. 1998, as cited in Environment Canada, Health Canada 2011; Deisinger et al. 1994, as cited in Environment Canada, Health Canada 2011). Dermal absorption in rats was determined to be less than oral uptake, having 50% absorption after 96 hours for 2-EHA (English et al. 1998, as cited in Environment Canada, Health Canada 2011), and 5 to 6% absorption after 96 hours for 2-ethylhexanol (Deisinger et al. 1994, as cited in Environment Canada, Health Canada 2011).
The primary metabolic pathway for 2-ethylhexyl 2-ethylhexanoate involves hydrolysis to form 2-EHA and 2-ethylhexanol. Because the current uses indicate that the predominant route of exposure is dermal, information on metabolism by skin esterases was considered. Although the majority of esterase activity in human skin is located in the epidermis and hair follicles (Tokudome et al. 2015), such activity has also been found in the stratum corneum (Beisson et al. 2001). This suggests that 2-ethylhexyl 2-ethylhexanoate may be hydrolyzed by esterases in the stratum corneum to form 2-EHA and 2-ethylhexanol, which are more readily absorbed through the skin than the parent ester.
8.3 Characterization of risk to human health
Based on reported esterase activity in the stratum corneum (see section 7.2), 2-ethylhexyl 2-ethylhexanoate may be hydrolyzed to 2-EHA and 2-ethylhexanol on human skin. As the extent of hydrolysis is unknown, it is conservatively assumed that all of 2-ethylhexyl 2-ethylhexanoate is hydrolyzed to 2-EHA, followed by complete absorption through the skin (i.e., assuming that absorption through the dermal route is equivalent to absorption through the oral route). 2-EHA was selected as the representative analogue for 2-ethylhexyl 2-ethylhexanoate as it is the more potent metabolite from ester hydrolysis, as evidenced by the health effects studies above.
To derive margins of exposure, critical effect levels for the selected analogue (i.e., 2-EHA) were compared to exposure estimates for both calcium 2-ethylhexanoate and 2-ethylhexyl 2-ethylhexanoate (Table 7‑1).
Exposure scenario |
Systemic exposure (mg/kg-bw) |
Critical effect level (mg/kg-bw/day) |
Critical health effect endpoint |
MOE |
|---|---|---|---|---|
Interior or exterior paint (calcium 2-ethylhexanoate) |
0.25 per application of calcium 2-ethylhexanoate |
LOAEL = 706 |
Short-term (increased liver weight) |
2820 |
Foot lotion (2-ethylhexyl 2-ethylhexanoate) |
0.37–1.10 per day of 2-ethylhexyl 2-ethylhexanoate [0.21–0.62 per day of 2-ethylhexanoic acid]a |
LOAEL = 100 |
Developmental (skeletal variations and malformations in fetuses) |
160–480 |
Face make-up (2-ethylhexyl 2-ethylhexanoate) |
0.91–3.05 per day of 2-ethylhexyl 2-ethylhexanoate [0.51–1.72 per day of 2-ethylhexanoic acid]a |
LOAEL = 100 |
Developmental (skeletal variations and malformations in fetuses) |
60–200 |
aAssuming complete hydrolysis, the mass of 2-ethylhexyl 2-ethylhexanoate is converted to an equivalent mass of 2-ethylhexanoic acid using a ratio of the molecular weights of these two substances (molecular weight of 2-ethylhexanoic acid is 144.21 g/mol).
For interior or exterior paint, a margin of exposure was not calculated for developmental health effects, as these were determined not to be relevant to this exposure scenario. Given that exposure to this product is a single event, metabolic clearance of 2-EHA is expected, thereby limiting liver toxicity in dams and the resultant chain of events leading to developmental toxicity in fetuses, based on knowledge of the mode of action for developmental toxicity of 2-EHA (Fiume et al. 2015).
The margin of exposure for interior or exterior paint is considered to be adequate to address uncertainties in the health effects and exposure databases.
The margins of exposure for foot lotion and face make-up are considered potentially inadequate to account for uncertainties in the health effects and exposure databases. In particular, the health effects endpoint (i.e., developmental toxicity) is severe, and the LOAEL of 100 mg/kg-bw/day was the lowest dose tested, indicating a potentially lower critical effect level.
While exposure of the general population to calcium 2-ethylhexanoate is not of concern at current levels, this substance is considered to have a health effect of concern based on its potential developmental toxicity. Therefore, there may be a concern for human health if exposure were to increase.
Uncertainties in evaluation of risk to human health
There is uncertainty in the extrapolation of toxicity between the oral and dermal routes of exposure, the dermal absorption of 2-ethylhexyl 2-ethylhexanoate, and the potential extent of hydrolysis on human skin. The conservative assumptions of complete hydrolysis to 2-EHA and equivalent absorption via the oral and dermal routes of exposure are likely to result in an overestimate of exposure. A reduction from 100% to 50% dermal absorption, based on the available study for 2-EHA (English et al. 1998, as cited in Environment Canada, Health Canada 2011), would not alter the outcome of the assessment.
There is also uncertainty regarding the mode of action for developmental toxicity following 2-EHA exposure. Effects related to PPARα-induced peroxisome proliferation are less likely to be relevant to humans as compared with rodents (Environment Canada, Health Canada 2011).
There is inherent uncertainty in the use of read-across hazard data from one substance to another. Although the analogue substances are known to have similar physical-chemical and/or toxicological profiles to the substances under consideration, it is possible that the health effects endpoints and/or critical effect values may vary.
9. Conclusion
Considering all available lines of evidence presented in this screening assessment, there is low risk of harm to organisms and the broader integrity of the environment from calcium 2-ethylhexanoate and 2-ethylhexyl 2-ethylhexanoate. It is concluded that calcium 2-ethylhexanoate and 2-ethylhexyl 2-ethylhexanoate do not meet the criteria under paragraphs 64(a) or (b) of CEPA as they are not entering the environment in a quantity or concentration or under conditions that have or may have an immediate or long-term harmful effect on the environment or its biological diversity or that constitute or may constitute a danger to the environment on which life depends.
On the basis of the adequacy of margins between estimates of exposure and critical effect levels observed in animal studies, it is concluded that calcium 2-ethylhexanoate does not meet the criteria under paragraph 64(c) of CEPA as it is not entering the environment in a quantity or concentration or under conditions that constitute or may constitute a danger in Canada to human life or health. On the basis of the potential inadequacy of margins between estimates of exposure and the critical effect level observed in the animal study, it is concluded that 2-ethylhexyl 2-ethylhexanoate meets the criteria under paragraph 64(c) of CEPA as it is entering or may enter the environment in a quantity or concentration or under conditions that constitute or may constitute a danger in Canada to human life or health.
Therefore, it is concluded that calcium 2-ethylhexanoate does not meet any of the criteria set out in section 64 of CEPA and that 2-ethylhexyl 2-ethylhexanoate meets one or more of the criteria set out in section 64 of CEPA.
2-ethylhexyl 2-ethylhexanoate does not meet the persistence criteria but does meet the bioaccumulation criteria as set out in the Persistence and Bioaccumulation Regulations of CEPA.
References
[AGDH] Australian Government Department of Health. 2013. Hexanoic acid, 2-ethyl-, 2-ethylhexyl ester. Sydney (AU): Department of Health, National Industrial Chemicals Notification and Assessment Scheme (NICNAS). Inventory Multi-tiered Assessment and Prioritisation (IMAP). [accessed 2016 Apr 1].
Akcina A, Seyisb F, Akcinc TA, Caycid YT, Coband AY. 2013. Chemical Composition and Antimicrobial
Activity of the Essential Oil of Endemic Heracleum platytaenium Boiss. from Turkey. J Essent Oil Res. 16(2):166–171.
Beisson F, Aoubala M, Marull S, Moustacas-Gardies A-M, Voultoury R, Verger R, Arondel V. 2001 Use of the tape stripping technique for directly quantifying esterase activities in human stratum corneum. Anal Biochem. 290: 179–185.
Bianchi F, Careri M, Conti C, Musci M, Vreuls R. 2007. Comparison of comprehensive two-dimensional gas chromatography-time-of-flight mass spectrometry and gas chromatography-mass spectrometry for the qualitative characterisation of roasted barley by solid-phase microextraction. J Sep Sci. 30:527–533.
Canada. 1999. Canadian Environmental Protection Act, 1999. S.C. 1999, c. 33. Canada Gazette Part III, vol. 22, no. 3.
Canada, Dept. of the Environment. 2012. Canadian Environmental Protection Act, 1999: Notice with respect to certain substances on the Domestic Substances List. Canada Gazette, Part I, vol. 146, no. 48, Supplement..
ChemCAN [level III fugacity model of 24 regions of Canada]. 2003. Ver. 6.00. Peterborough (ON): Trent University, Canadian Centre for Environmental Modelling and Chemistry..
[CDAT] Chemical Data Access Tool. [modified 2014 Jun]. Non-confidential 2012 Chemical Data Reporting Information: search results for CAS RNs 136-51-6 and 7425-14-1. Washington (DC): US Environmental Protection Agency. [accessed 2016 Jan 15].
Cherif AO, Messaouda MG, Pellerin I, Boukhchina S, Kallel H, Pepe C. 2013. Screening and Profiling of Hydrocarbon Components and Squalene in Developing Tunisian Cultivars and Wild Arachis hypogaea L. Species. J Am Oil Chem Soc. 90:675–686.
[ConsExpo] Consumer Exposure Model. 2006. Ver. 4.1. Bilthoven (NL): Rijksinstituut voor Volksgezondheid en Milieu [National Institute for Public Health and the Environment]..
[CosIng] Cosmetic Ingredients & Substances [database]. Brussels (BE): European Commission. [accessed 2016 Jan 13]..
[DPD] Drug Product Database [database]. [modified 2015 Jul 17]. Ottawa (ON): Health Canada. [accessed 2015 Oct 13]..
[ECHA] European Chemicals Agency. c2007-2015. Registered substances database; search results for CAS RN [149-57-5 and 7425-14-1]. Helsinki (FI): ECHA. [updated 2016 Mar 9; accessed 2016 Mar 9].
[ECCC] Environment and Climate Change Canada. 2016a. Ecological Science Approach Document: Ecological Risk Classification of Organic Substances. Ottawa (ON): Government of Canada.
[ECCC] Environment and Climate Change Canada. 2016b. Gatineau (QC): Data used to create substance-specific hazard and exposure profiles and assign risk classifications in the Ecological Risk Classification of organic substances. Gatineau (QC). Available from: substances@ec.gc.ca.
[ECCC, HC] Environment and Climate Change Canada, Health Canada. [modified 2007 Apr 20]. Categorization. Ottawa (ON): Government of Canada. [accessed 2015 Sep 25]..
Environment Canada. 2013. DSL Inventory Update data collected under the Canadian Environmental Protection Act, 1999, section 71: Notice with respect to certain substances on the Domestic Substances List. Data prepared by: Environment Canada, Health Canada; Existing Substances Program.
Environment Canada, Health Canada. 2011. Screening Assessment for the Challenge. Hexanoic acid, 2-ethyl-. Ottawa (ON): Health Canada. Available from:.
[EPI Suite] Estimation Programs Interface Suite for Microsoft Windows [estimation model]. c2000-2012. Ver. 4.11. Washington (DC): US Environmental Protection Agency, Office of Pollution Prevention and Toxics; Syracuse (NY): Syracuse Research Corporation..
[EU] European Union. 2008. Regulation (EC) No 1272/2008 of the European Parliament and of the Council of 16 December 2008 on classification, labelling and packaging of substances and mixtures, amending and repealing Directives 67/548/EEC and 1999/45/EC, and amending Regulation (EC) No 1907/2006. Off J Eur Union L. 353:1–1355..
[EU] European Union. 2009. Regulation (EC) No 1223/2009 of the European Parliament and of the Council of 30 November 2009 on cosmetic products. Off J Eur Union L. 342:59-209.
Fiume M et al. 2015. Safety Assessment of Alkyl Ethylhexanoates as Used in Cosmetics. Int J Toxicol. 34(S3):61S–73S.
Health Canada. [modified 2013 Jun 27]. Food additives permitted for use in Canada. Ottawa (ON): Health Canada. [accessed 2015 Nov 18].
Health Canada. [modified 2015 Dec 14]. Cosmetic Ingredient Hotlist: list of ingredients that are prohibited for use in cosmetic products. Ottawa (ON): Health Canada, Consumer Product Safety Directorate. [accessed 2016 Jan 20].
[IMO] International Maritime Organization. 2002. Anti-fouling systems. London (UK): International Maritime Organization [accessed 2016 Feb 18]..
Landau M. 2000. Driers and Metallic Soaps. Kirk-Othmer Encyclopedia of Chemical Technology. Online version. New York (NY): John Wiley and Sons, Inc. [accessed 2015 Dec 7]. [restricted access].
[LNHPD] Licensed Natural Health Products Database [database]. [modified 2016 July 14]. Ottawa (ON): Health Canada. [accessed 2015 Jul 28]..
[NHPID] Natural Health Products Ingredients Database [database]. [modified 2016 Apr 18]. Ottawa (ON): Health Canada. [accessed 2015 Oct 13].
Özeka T, Kürkçüoğlu M, Başer KHC, Tosunb A. 2007. Composition of the Essential Oils of Tordylium trachycarpum (Boiss.) AlEisawi et Jury and Tordylium hasselquistiae DC. Growing in Turkey. J Essent Oil Res. 19(5):410–412.
Priyanka D, Sindhoora S, Vijayanand P, Kulkarni SG, Nagaraian S. 2015. Influence of thermal processing on the volatile constituents of muskmelon puree. J Food Sci Tech. 52(5):3111–3116.
Radjabian T, Salimi A, Rahmani N. 2014. Essential-Oil Composition of the Fruits of Six Heracleum L. Species from Iran: Chemotaxonomic Significance. Chem Biodivers. 11:1945–1953.
Razavi SM, Nejad-Ebrahimi S. 2010. Chemical composition, allelopatic and antimicrobial potentials of the essential oil of Zosima absinthifolia (Vent.) Link fruits from Iran. Nat Prod Res. 24(12):1125–1130.
[RIVM] Rijksinstituut voor Volksgezondheid en Milieu [National Institute for Public Health and the Environment (NL)]. 2006. Cosmetics fact sheet: to assess the risks for the consumer: updated version for ConsExpo 4. Bilthoven (NL): RIVM. Report No.: 320104001/2006. [accessed 2016 Feb 19].
[RIVM] Rijksinstituut voor Volksgezondheid en Milieu [National Institute for Public Health and the Environment]. 2007. Paint products fact sheet: to assess the risks for the consumer: updated version for ConsExpo 4. Bilthoven (NL): RIVM. Report No.: 320104008/2007. [accessed 2016 Feb 25].
Tansawat R, Maughan CAJ, Ward RE, Martini S, Cornforth DP. 2013. Chemical characterisation of pasture- and grain-fed beef related to meat quality and flavour attributes. Int J Food Sci Tech. 48:484–495.
Tokudome Y, Katayanagi M, Hashimoto F. 2015. Esterase activity and intracellular localization in reconstructed human epidermal cultured skin models. Ann Dermatol. 27(3):269–274.
Tosun F, Kızılay ÇA, Erol K, Kılıç FS, Kürkçüoğlu M, Başer KHC. 2008. Anticonvulsant activity of furanocoumarins and the essential oil obtained from the fruits of Heracleum crenatifolium. Food Chem. 107(3):900–993.
Tosun A, Kürkçüoğlu M, Başer KHC. 2010. Composition of Tordylium aegyptiacum (L.) Lam. Essential Oil. J Essent Oil Res. 22(3):245–246.
Appendix A. Estimated exposure from use of paint and cosmetics, based on ConsExpo
Scenario |
Model parameters |
Estimated exposure |
|---|---|---|
Interior or exterior paint |
The scenario assumptions are based on a ConsExpo default dermal scenario for solvent rich paint (RIVM 2007), except noted below.
|
0.25 mg/kg/application of calcium 2-ethylhexanoate
|
Foot lotion |
The scenario assumptions are based on a ConsExpo default dermal scenario for antiperspirant foot cream (RIVM 2006), except noted below.
|
0.18–0.55 mg/kg/application; 0.37–1.10 mg/kg-bw/day of 2-ethylhexyl 2-ethylhexanoate |
Face make-up |
The scenario assumptions are based on a ConsExpo default dermal scenario for facial make-up (RIVM 2006), except noted below.
|
0.51–1.69 mg/kg/application; 0.91–3.05 mg/kg-bw/day of 2-ethylhexyl 2-ethylhexanoate |