Summary report: Consultation on potential market for health products containing cannabis that would not require practitioner oversight
Contents
- 1.0 Introduction
- 2.0 Consumer feedback on potential market for CHPs
- 3.0 Industry feedback on potential market for CHPs
- 4.0 Feedback on the potential market for CHPs from all other interested parties
- 5.0 Need for scientific evidence
- 6.0 Next steps
1.0 Introduction
Health Canada is aware that some Canadians are interested in the potential therapeutic uses of cannabis for purposes such as pain relief for human and animal use, without the need for practitioner oversight.
Health Canada held a public consultation between June 19 and September 3, 2019 on the Potential Market for Cannabis Health Products (CHPs) that would not Require Practitioner Oversight. The consultation sought feedback from Canadians on the kinds of products they would be interested in if such products were made legally available in Canada. For the purposes of the consultation and for this summary report, Health Canada is referring to these products as "cannabis health products" (CHPs).
As part of the consultation, Health Canada held five information sessions by teleconference for parties interested in CHPs for human use or for use in animals. The aim was to support respondents in providing feedback. Comments were not solicited in these sessions.
The purpose of this summary report is to provide an overview of the comments received from Canadians. Health Canada intends to gather external scientific advice on the appropriate evidence standards required to demonstrate safety, efficacy and quality.
The information gathered through these activities will inform the next steps on a potential regulatory path forward.
1.1 Methodology
The consultation document was posted on the Health Canada website and distributed through different channels. In particular, stakeholders, including researchers as well as health and veterinary associations, the cannabis and health product industries, and government organizations, were notified by email. Health Canada’s stakeholder registry, the Consultation and Stakeholder Information Management System (CSIMS), was used to notify individuals or organizations interested in particular health topics. Canadians were also encouraged to participate through Health Canada social media platforms. Participants were asked to provide feedback by answering questions posed in the consultation, or by emailing a written submission.
Health Canada conducted qualitative analysis of the online responses and of the written submissions to identify major themes from the feedback received. This approach required a detailed review of each response. Once reviewed, Health Canada analysts developed a thematic framework by coding responses, a process used to classify and sort qualitative data. In addition, qualitative data analysis software was used to support analysis of the feedback from the online consultation, as responses were greater in volume than the written submissions. Results included in the report, and the figures, were generated from this process.
The sections of the report are grouped according to the themes of responses and appear in the same order as the questions posed in the online consultation. Consumer feedback was provided through the online consultation and common themes have been identified in section 2. As respondents from industry and other interested parties submitted comments through both the online consultation and written submissions, themes from both data sets are incorporated within their respective sections (3 and 4).
This report summarizes key points received from online consultation participants and written submissions. However, the views are not intended to be representative of the broader Canadian public.
To complement quantitative findings, qualitative descriptors are used throughout the report to refer to the approximate number of respondents who conveyed a particular theme. The following descriptors are ranked in the following order to provide a sense of reference for the reader: “few,” “some/others,” “several,” “many,” and “most/majority”.

Text description
Illustrating qualitative descriptors ranked in the following order from left to right: few, some/others, several, many, and most.
In addition to these descriptors, summary descriptors such as “overall” and “in general” or “generally" are used to summarize key themes conveyed by respondents.
1.2 Overview of consultation respondents
In total, 1,104 respondents completed the online consultation. Respondents were asked to identify as a consumer, industry representative or another type of interested party, such as a representative from a health or veterinary association.
- 62% of participants identified as consumers interested in purchasing CHPs.
- 23% as industry representatives interested in manufacturing or selling CHPs.
- 15% as other interested parties.
Questions posed in the online consultation differed for each participant type. However, all participants were asked to specify whether they were interested in CHPs for human use, for use in animals or for both purposes. A majority of both consumers (61%) and industry (62%) representatives indicated an interest in both CHPs for human use and for use in animals.
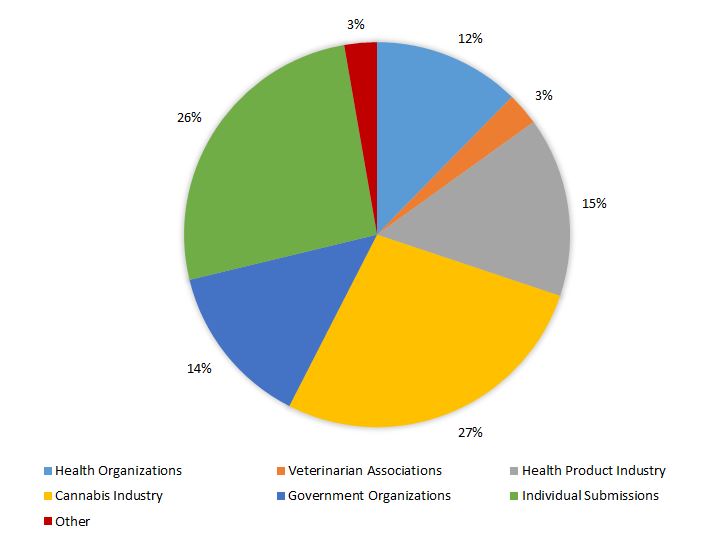
In addition to those who completed the online consultation, 73 written submissions were received from a range of organizations, including health organizations, veterinary associations, cannabis and health product industry, retailers, and other government organizations (refer to Figure 1).
Figure 1. Breakdown of written submissions (n=73) received in response to CHP consultation
Text description
| Submissions | By percentage |
|---|---|
| Cannabis Industry | 27 |
| Individual submissions | 26 |
| Health Product Industry | 15 |
| Government Organizations | 14 |
| Health Organizations | 12 |
| Veterinarian Associations | 3 |
| Other | 3 |
2.0 Consumer feedback on potential market for CHPs
Consumers comprised the majority of consultation participants, with a total of 684 respondents. Refer to Figure 2 for a breakdown of age and gender demographics.
Figure 2. Respondent demographics: Consumers (n=684) gender vs. age breakdown
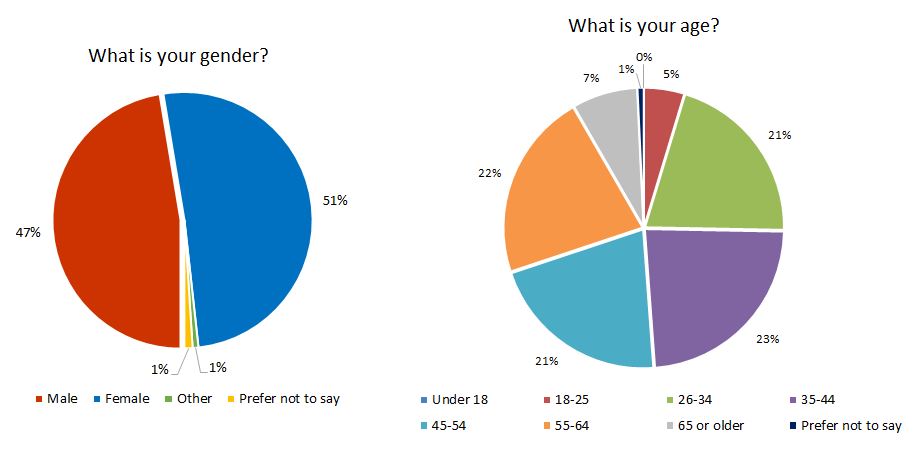
Text description
| By percentage | By numbers | |
|---|---|---|
| Male | 47 | 324 |
| Female | 51 | 348 |
| Prefer not to say | 1 | 7 |
| Other | 1 | 5 |
| By percentage | |
|---|---|
| 65 or older | 7 |
| 55-64 | 22 |
| 45-54 | 21 |
| 35-44 | 23 |
| 26-34 | 21 |
| 18-25 | 5 |
| Under 18 | 0 |
| Prefer not to say | 1 |
Questions put to consumers sought feedback on the types of CHPs they would be interested in purchasing without practitioner oversight and for what purposes. Overall, online consultation feedback from consumers indicated they believe there is a strong demand for CHPs, as cannabis is perceived to be an effective, natural and low-risk alternative to pharmaceutical drugs currently on the market.
2.1 Level of interest in purchasing a CHP intended to treat minor ailments
[Question 1: Would you be interested in purchasing a product approved by Health Canada that contains cannabis and is intended to treat minor ailments (e.g., a CHP approved to treat muscle pain)? Why or why not?]
Although the majority of respondents indicated interest in CHPs for both human use and use in animals, there were some differences in the levels of interest in purchasing these types of products. Of the 657 consumers who answered the first question, roughly:
- 93% demonstrated a positive level of interest in purchasing or learning more about CHPs to treat minor ailments.
- 2% of consumer respondents were strongly opposed.
- 5% indicated interest conditional on having further information.
Access to alternative products
Consumers who demonstrated a positive level of interest considered cannabis – particularly cannabidiol (CBD), a compound found in the cannabis plant – as a natural remedy. They noted their preference for greater access to natural alternatives to pharmaceutical drugs. Many shared anecdotes of their long-term experience using cannabis for therapeutic purposes. They believed they had experienced various health benefits as well as fewer side effects than they associated with opioids or synthetic drugs. Respondents encouraged Health Canada's approval of these types of products to ensure safety and quality, while allowing consumer choice and increased access to a wider array of products without practitioner oversight.
Lack of evidence
A small number of consumers were opposed to purchasing CHPs (2%). These consumers either felt there was not sufficient evidence to support unsupervised use of cannabis health products or were not interested in purchasing products due to the lack of evidence of the long-term health impacts.
Need for further information
About 5% of consumers felt they did not have enough information to indicate their level of support. They disclosed that if products are not affordable or not covered by insurance they would be more likely to, or will continue to, seek cheaper, unapproved products from the illicit market. Comments also reflected a need for more evidence and information on the safety and quality of CHPs to support consumers in making their own decisions for their health.
2.2 Common themes for use
[Questions 2 and 4: Are there particular purposes, or types of health claims, for which you are most likely to seek out CHPs (e.g., pain relief for sore muscles)? Are you aware of specific sub-populations or groups that would be particularly interested in CHPs? (e.g., seniors, those with chronic conditions) If so, for what purpose?]
Focus on pain relief and inflammation
Overall, consumers have a high interest in seeking out CHPs in a variety of formats and dosages for various purposes. The majority of consumers, approximately 85% (579 participants), described a desire for CHPs to treat issues related to pain and inflammation, such as joint or back pain, arthritis, migraines, or headaches. Mental health issues such as anxiety, depression or stress, were identified as a purpose of interest among 253 consumer respondents. Trouble sleeping was also a common purpose of interest (231 respondents).
Most respondents (approximately 82%) described more than one purpose of interest. As shown in Figure 3, consumers would also be interested in CHPs to helpFootnote 1 with:
- Gastrointestinal issues (e.g., nausea, indigestion, or issues with appetite).
- Skin conditions (e.g., psoriasis, eczema or skin care).
- General health.
- Muscle recovery or relaxation.
- Side effects to cancer treatments.
- Behavioural neurological disorders (e.g., autism, attention deficit hyperactivity disorder, multiple sclerosis or Parkinson's).
- Seizures.
- Other reasons such as concentration, energy, menopause, or other health benefits that were not specified.
Figure 3. What was heard: Consumer reasons to seek out cannabis health products
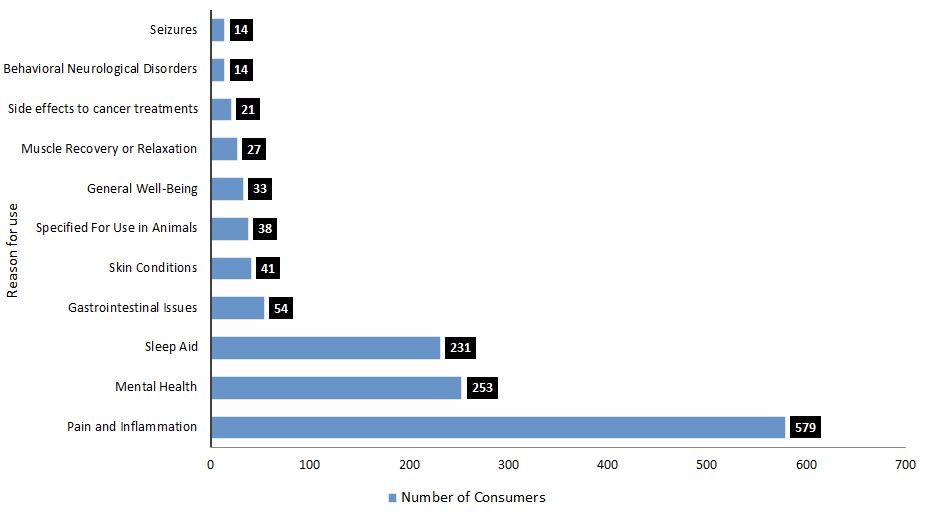
Text description
| Reason for use of cannabis health products | Number of consumers |
|---|---|
| Seizures | 14 |
| Behavioral neurological disorders | 14 |
| Side effects to cancer treatments | 21 |
| Muscle recovery or relaxation | 27 |
| General well-being | 33 |
| Specified for use in animals | 38 |
| Skin conditions | 41 |
| Gastrointestinal issues | 54 |
| Sleep aid | 231 |
| Mental health | 253 |
| Pain and inflammation | 579 |
Although feedback demonstrated interest in both CHPs for human use and for use in animals, respondents were not asked to distinguish between purposes of interest for human use or for use in animals. However, 38 consumer respondents specified interest in CHPs for animal use. Of this group, 17 indicated a particular interest in CHPs to treat pain (e.g., joint pain, osteoarthritis or sore muscles) for their pets and 13 were interested in CHPs to treat anxiety in their pets. While 12 respondents did not specify a purpose for use in animals, seizures or maintenance of fur or skin were other reasons of interest for CHPs for use in animals.
Sub-populations of interest
Most consumers noted current use of cannabis for a range of medical purposes. They felt Canadians would benefit from having access to approved products that are deemed safe by Health Canada and easily accessible. Specifically, about 76% of consumers were aware of sub-populations who would be interested in CHPs and felt anyone experiencing pain, trouble sleeping, or suffering from a chronic or mental health condition would benefit. In particular, seniors, veterans, pets, athletes, or women experiencing menopause were all groups of interest.
2.3 Preferred product formats for CHPs
[Question 3: Do you have preferred product formats for CHPs (e.g., cannabis extracts, topicals, tinctures, or others)?]
Comments focussing on safe and easy-to-use product formats, such as topicals or products for oral ingestion, were the most common responses to this question (Figure 4).
- 61% of consumers were interested in products that could be taken orally, particularly as extracts, tinctures, oils, capsules or in sublingual formats.Footnote 2
- 41% were interested in topicals, such as creams or lotions that would be easy to apply directly for joint pain or sore muscles.
- 24% either were interested in all formats or did not state a preference.
- 18% were interested in CHPs edibles or beverages that would be easy to use.
Roughly 10% of participants preferred to continue smoking or vaping cannabis, and others felt that product formats or dosing preferences would be dependent on the condition being treated. About 27% of participants specified they wanted CBD products in different formats and indicated a preference for easy-to-consume CBD products for both human use and for use in animals. While 4% were interested in CBD and tetrahydrocannabinol (THC) combination products, others did not want to experience intoxicating effects, emphasizing the need for THC-free product options.
Figure 4. What was heard: Product formats of interest among consumer respondents
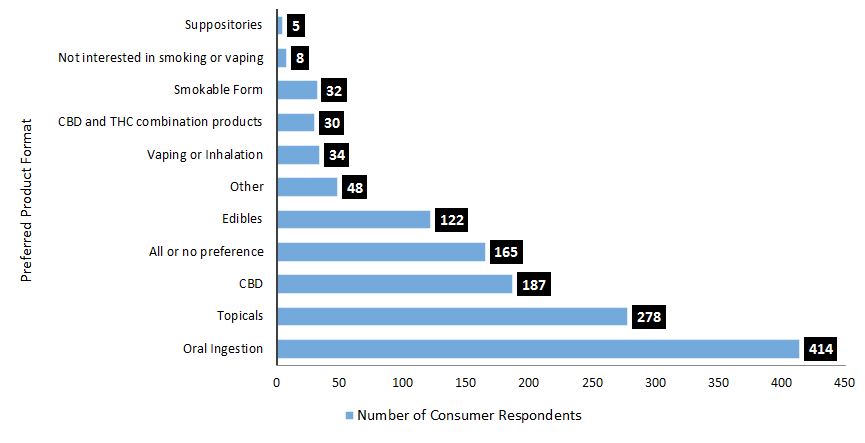
Text description
| Preferred product format | Number of consumers |
|---|---|
| Suppositories | 5 |
| Not interested in smoking or vaping | 8 |
| Smokable Form | 32 |
| CBD and THD combination products | 30 |
| Vaping or inhalation | 34 |
| Other | 48 |
| Edibles | 122 |
| All or no preference | 165 |
| CBD | 187 |
| Topicals | 278 |
| Oral ingestion | 414 |
2.4 Additional comments on the potential market for CHPs
[Question 5: Is there any additional feedback that you would like to share on the potential market for CHPs?]
Additional feedback from consumers affirmed high demand for making CHPs legally available for both human and animal use. While the focus of the consultation was on the potential market for CHPs, consumer participants also provided feedback related to the current rules and requirements for cannabis products under the Cannabis Act.Footnote 3
Need for fair regulations
500 respondents provided additional feedback. Of these, about 4% were mindful of the need for practitioner oversight to ensure safe and effective products, while 40% felt that CHPs required an appropriate level of regulatory controls. These respondents emphasized that regulations should support rather than hinder consumer choice and access, as stringent requirements risk stimulating the illicit market. As such, they advocated for fair regulations and further guidance, which would support consumer access and informed decision-making.
Place of sale and packaging and labelling requirements a concern for some consumers
Regarding the proposed parameters for CHPs outlined in the consultation document, place of sale was a concern among approximately 10% of consumers. In particular, they felt that the retail environment for CHPs should be similar to that of over-the-counter drugs or natural health products (NHPs), and should not be restricted to provincially or territorially authorized retailers or federally licensed cannabis sellers. Allowing CHPs to be sold at pharmacies, health stores or other retailers would improve availability and deter from the stigma associated with cannabis use. However, about 2% of respondents agreed that measures would need to be in place to prevent direct youth access, such as having CHPs behind the counter at pharmacies.
About 5% of consumers noted concerns with excessive packaging on current cannabis products and found labelling requirements too restrictive. These participants suggested some level of branding be allowed for CHPs so companies can support consumers in making informed decisions. Other respondents felt the controls put in place by Health Canada were necessary and would ensure quality consistency and safety.
Regulation of CBD
Consumers indicated a strong interest in products containing CBD throughout the online questionnaire. Moreover, 105 respondents provided additional comments in favour of access to CBD for therapeutic purposes without practitioner oversight. They recommended an alternative approach to the regulation of CBD, which – unlike THC – is not intoxicating. Generally, consumers expressed confusion regarding the current regulatory regime for cannabis, specifically the difference between the non-medical and medical frameworks. Participants suggested CBD should not be regulated the same way as other substances or products, such as THC, alcohol or other pharmaceutical drugs, which they view to be more harmful.
A small portion (10%) of respondents who provided additional feedback recommended a specific pathway for CHPs, such as regulating CBD as an NHP or over-the-counter drug. Less than 1% mentioned that they would prefer to access CHPs through a prescription or to have some level of oversight through a practitioner.
General consumer perspective
Overall, consumer participants felt that cannabis is safer or healthier to use than pharmaceuticals, and reported personal experiences of therapeutic benefits, particularly with CBD products. In general, respondents would prefer to access products approved for safety and efficacy without the need for additional practitioner oversight, rather than feeling constrained to other cannabis products. They would also like to learn more about the potential benefits of CHPs.
3.0 Industry feedback on potential market for CHPs
In total, 253 individuals indicated being a representative of an industry interested in manufacturing or selling CHPs, comprising 23% of all consultation participants. Participants were able to identify themselves as a representative of several types of industries as applicable (e.g., a representative of both an NHP and a non-prescription drug manufacturer). Consequently, the total number of represented industries (416) included in our analysis is higher than the number of respondents.
Figure 5 illustrates the types of industry represented.
- A majority of industry respondents indicated being a member of the NHP (39%) or cannabis industries (25%).
- Online consultation participants also included representatives of the prescription drug (3%), non-prescription drug (11%), and veterinary health product (12%) industry.
Figure 5. Representation of industry respondents
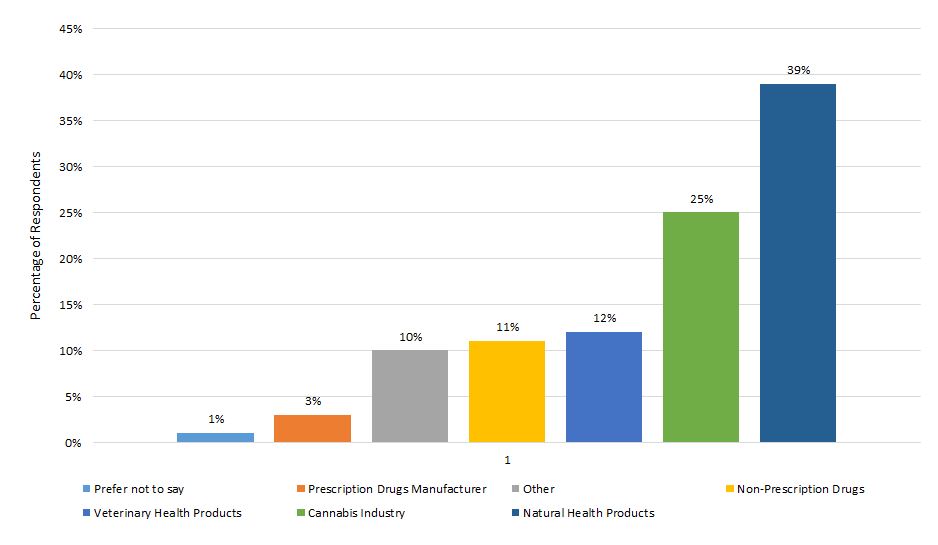
Text description
| Type of industry | By percentage |
|---|---|
| Natural health products | 39 |
| Cannabis | 25 |
| Veterinary health products | 12 |
| Non-prescription drugs | 11 |
| Other | 10 |
| Prescription drug manufacturer | 3 |
| Prefer not to say | 1 |
The overall feedback received from industry demonstrated a strong desire to bring to market a new range of cannabis products that would help displace the illicit market, meet Canadians' health needs and promote economic growth. The following section reflects themes identified in online consultation responses and written submissions from industry respondents.
3.1 Level of interest in manufacturing and selling CHPs on the Canadian market
[Question 1 for Industry: Are you interested in manufacturing and selling CHPs on the Canadian market? Why or why not?]
Approximately 78% of industry respondents expressed an interest in manufacturing or selling CHPs, 12% were not interested in those specific activities, and 11% preferred not to say. Of the total industry respondents who expressed an interest in manufacturing or selling CHPs, about 30% were interested in CHPs for human use, 5% specified an interest in CHPs for use in animals and 65% were interested in both.
Need to ensure consumer access to safe and effective products
Overall, company representatives across different industry groups indicated a need for a CHP pathway to displace the illicit market and protect the health and safety of consumers. The main reason of interest in CHPs by industry representatives is to ensure better consumer choice and access to safe, effective and quality products. They also viewed a potential market for CHPs as an opportunity to expand their business and generate more revenue. Industry comments demonstrated an eagerness to provide clear, consistent and evidence-based information on the potential health benefits and risks of cannabis. Refer to Figure 6 for the main reasons of interest in bringing CHPs to the Canadian market.
Figure 6. What was heard: Main reasons why industry representatives are interested in bringing CHPs to Canadian market
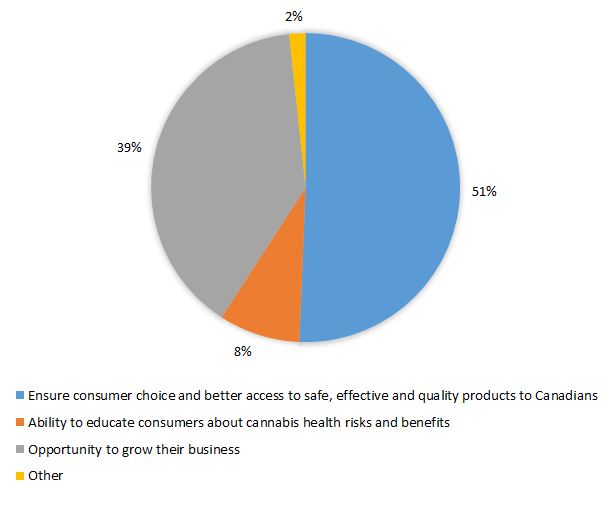
Text description
| Main reason interested in bringing cannabis health product | By percentage |
|---|---|
| Ensure consumer choice and better access to safe, effective and quality products to Canadians | 51 |
| Opportunity to grow their business | 39 |
| Ability to educate consumers about cannabis health risks and benefits | 8 |
| Other | 2 |
Reasons for lack of interest
Of the 12 % of respondents across different industry groups who indicated a lack of interest in selling or manufacturing CHPs, reasons included:
- Activities were simply not in their purview (e.g., companies offering consulting services).
- A lack of scientific evidence to support safety and efficacy of CHPs.
3.2 Identified products of interest to bring to market in Canada
[Question 2: If so, what products are you the most likely to pursue bringing to market in Canada? More specifically, what types of formats, with which cannabis ingredients, and with what health claims?]
Product formats of interest
Approximately 48% of industries represented, including 40% of veterinary industry respondents, indicated a specific interest in CBD-based products. Similar to what we heard from consumers, formats of preference included oral capsules, topicals (e.g., creams, lotions), extracts (e.g., oils, tinctures) and edible formats. In addition, 24 industry representatives indicated a desire to combine CBD with other cannabis-derived ingredients such as THC, flavonoids, terpenes, and approved NHPs (e.g., vitamins and minerals, essential oils, herbs).
Health claims of interest
With respect to health claims of interest, industry representatives from all groups emphasized their preference for providing CHPs for pain relief, inflammation, sleep disorders, and anxiety. Other areas of interest include skin conditions (e.g., eczema, psoriasis) and digestive issues.
Although 62% of industry representatives noted interest in both CHPs for human use and use in animals, approximately 7% of respondents across different industry groups specified interest in bringing CHPs to market for animal use. CBD oils and treats (e.g., soft chews) for pain and anxiety management were of particular interest.
3.3 Incentives or disincentives for seeking to bring a CHP to market in Canada
[Question 5&6: Are there elements of the proposed approach that would create a particular incentive or disincentive for seeking to bring a CHP to market in Canada? If so, what elements and why? Are there any other specific elements of the retail environment for cannabis that would present unique benefits or challenges to the sale of CHPs?]
A legal framework that provides convenient access to safe and effective CHPs without the oversight of a practitioner was a common theme among all industry groups. Generally, industry respondents expressed views that a new pathway for CHPs would increase economic opportunities, benefit consumers dealing with minor ailments and reduce the burden on practitioners.
Evidence standards a common theme
The proposed evidence standards for CHPs appeared to be both an incentive and a disincentive to bring CHPs into the market.
- 9 respondents from different industry groups highlighted the need for robust scientific evidence.
- 18 different industry representatives were in favour of less stringent requirements, to avoid discouraging research and innovation.
Of these respondents, a few representatives from NHP and veterinary health products (VHP) companies also mentioned that they would like to be able to use traditional evidence or make general health promotion claims on CHPs.
Place of sale a common concern
Overall, restricting the sale of CHPs to provincially and territorially authorized retailers or federally licensed sellers was identified in both online consultation responses and written submissions as a major disincentive for industry respondents. Similar to consumer feedback, many criticized the proposed retail approach, the lack of health training among employees and the risk of confusion for customers between non-medical cannabis products and CHPs.
Almost 40% of all types of industry respondents from the online consultations recommended that CHPs be sold in pharmacies, veterinary clinics or health stores. These respondents felt that having a health care professional such as a pharmacist or a veterinarian available to provide advice to consumers would support informed decision-making. About 58% of the written submissions received from the cannabis and the NHP industries also suggested pharmacy distribution of CHPs to ensure safe use of CHPs and help displace the illicit market.
Challenges with packaging and labelling requirements
About 50% of the total written submissions from the cannabis industry and a few written submissions from health product companies indicated the proposed packaging and labelling requirements would present challenges. They expressed a preference for a broader ability to promote and distinguish their products on the market. Respondents proposed that the requirements should be similar to any other NHP or over-the-counter drug to compete with the illicit market.
3.4 Additional feedback on the potential market for CHPs
[Question 7: Is there any additional feedback that you would like to share on the potential market for CHPs?]
Regulation of CBD
As additional feedback to the online consultation, 28% of cannabis industry representatives, 34% of NHP and 21% of the VHP company representatives took the opportunity to urge Health Canada to regulate CBD differently from THC. These respondents recommended a different approach for the regulation of CBD, citing both high market demand and their perception that CBD is a safe substance.
Similarly, about half of the written submissions from the cannabis and NHP industries proposed creating two distinct pathways for CHPs that appropriately reflect their risk profiles. Specifically, they suggested establishing two different pathways for CHPs with THC and CHPs with CBD. Furthermore, each potential pathway would have its own requirements for packaging and labelling, retail environment, youth access, and evidence standards.
Other recommendations
A few written submissions received from cannabis industry representatives also recommended that Health Canada publish specific CHP guidance documents on evidence requirements, permitted ingredients, and dosages to support research and development of potential CHPs.
Additional recommendations included the importance of regulatory monitoring, and future alignment and collaboration with the United States and Europe, particularly in terms of the importation and exportation of potential CHPs.
4.0 Feedback on the potential market for CHPs from all other interested parties
Out of all respondents to the online consultation, 167 (15%) identified themselves as other interested parties.
- 21% of participants indicated being a healthcare professional or association representative.
- 15% identified as a veterinary professional or association representative.
- 9% identified as academics or researchers.
- 5% represented provincial, territorial or municipal government organizations.
- 4% represented non-governmental organizations (NGOs).
- 3% identified as advocacy group representatives.
- 26% of participants specified being a part of “other groups” such as regulatory consultants or legal representatives.
A number of healthcare and veterinary associations and professionals suggested that discussions regarding CHPs are premature. Many of these respondents expressed significant health and safety concerns about CHPs, while levels of support and perspectives from the other respondents varied. Themes identified in the following section are based on feedback received from online consultation responses and written submissions.
4.1 Sub-populations or groups interested in CHPs
[Question 2 for other parties: Are you aware of specific sub-populations or groups that would be particularly interested in CHPs? (e.g., seniors, those with chronic conditions)? If so, what purpose?]
Approximately 69% of respondents from this group were aware of sub-populations or groups that would be interested in CHPs for both human use or use in animals. Similar to what we heard from consumers and industry representatives, different types of respondents described more than one purpose of interest. Comments indicated an interest primarily for pain relief, mental health purposes (e.g., anxiety) or as a sleep aid (refer to Figure 7). Of these respondents, 50% of healthcare professionals, 56% of researchers, and 32% of veterinary professionals indicated the most interest in pain relief. Healthcare professionals also demonstrated the greatest interest in mental health purposes (70%). Overall feedback from these respondents indicated that they perceived anyone suffering from chronic conditions or in pain would benefit from CHPs, particularly seniors, the aging population, or pets.
Other potential areas of benefit identified by the respondents included gastrointestinal issues, side effects of cancer treatments, neurological disorders (e.g., multiple sclerosis or Parkinson's), seizures, or other chronic conditions.Footnote 4
Figure 7. What was heard: Main reasons for use according to other interested parties
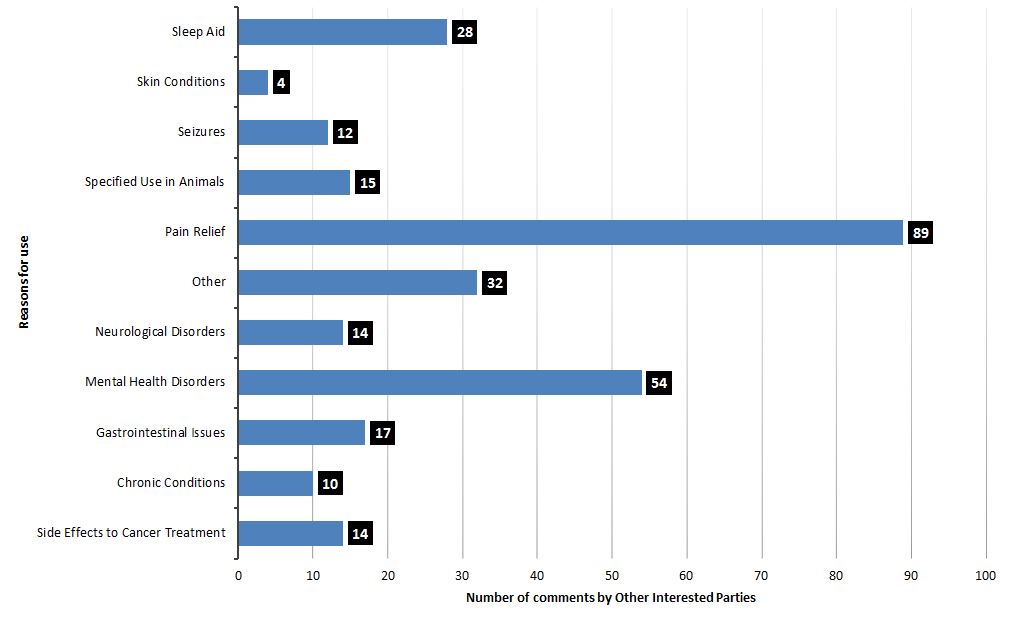
Text description
| Reason for use of cannabis health products | Number of comments by other interested parties |
|---|---|
| Sleep aid | 28 |
| Skin condition | 4 |
| Seizures | 12 |
| Specified use in animals | 15 |
| Pain relief | 89 |
| Other | 32 |
| Neurological disorder | 14 |
| Mental health disorder | 54 |
| Gastrointestinal issues | 17 |
| Chronic conditions | 10 |
| Side effects to cancer treatment | 14 |
4.2 Additional feedback on the potential market for CHPs
[Question 3: Is there any additional feedback that you would like to share on the potential market for CHPs?]
Health and safety concerns were a common theme among representatives from all other interested parties. In particular, 72% of the 39 representatives from healthcare associations or health professionals disclosed health and safety concerns. A shared concern among this group was the lack of scientific evidence on the potential negative effects of cannabis, particularly on the risks for women who are pregnant or breastfeeding, those with mental illness, or among youth. Healthcare association representatives highlighted the need for a cautious approach, and recommended having CHPs available only as scientific evidence becomes available to support (or refute) claims.
About 22% of veterinary professionals and associations who completed the online consultation also expressed health and safety concerns for pets, including the risk of toxicity, interactions with other medications, and side effects. Written submissions from medical professional associations noted public health concerns (e.g., ingestion by young children) with increased availability of CHPs. As a recommended measure, they advised strict packaging and labelling requirements be put in place.
Mental health associations also indicated in written submissions that any new pathway for CHPs should not encourage or increase consumption of cannabis for non-medical purposes. It was recommended that the strict controls put in place under the Cannabis Act to protect youth and prevent normalization of use be maintained.
Recommendations for practitioner oversight
Due to a lack of valid research and evidence and the need to protect youth, 40% of healthcare professional respondents and 33% of representatives from healthcare associations who completed the online consultation recommended the need for practitioner oversight for CHPs.
Feedback from online consultation responses indicated that 14% of veterinary professionals and 40% of representatives from veterinary associations were not interested in selling CHPs on the Canadian market due to a lack of evidence to support claims for use in animals. In addition, about 14% of veterinary professionals recommended the need for practitioner oversight for CHPs for use in animals. Notably, they suggested that the only veterinary use of cannabis should be under a valid veterinary-client-patient relationship. These views were also reflected in a written submission provided by a veterinary association. Suggestions were made to amend the current medical cannabis regime as a first step. These respondents also noted they currently feel limited in the guidance they are able to provide to clients regarding advice for pets.
Overall support for strong evidence requirements
Written submissions from representatives of health organizations indicated an overall support for strong evidence requirements for safety, efficacy and quality to support health claims. Many (44%) recommended CHPs be made available at a pharmacy where guidance or advice could be provided from a pharmacist. Recommendations to maintain strict packaging and labelling requirements were also reiterated in order to prevent ingestion or accidental consumption by children and to enhance consumer safety.
Of the online consultation responses and written submissions received from representatives of government organizations (e.g., provincial or territorial), some provided overall support for the proposed retail environment model and supported the need for strong evidence requirements for CHPs. Many government organization representatives also supported strong controls to prevent youth access, such as maintaining rules for packaging, labelling, and promotion. However, a few government respondents strongly opposed the introduction of CHPs and expressed several concerns including the lack of evidence on safety and efficacy, lack of compliance and enforcement measures and the need to restrict youth access.
Written submissions from two organizations that support the regulation of medical professionals recommended the maintenance of stringent compliance and enforcement measures to protect youth, such as restricting promotions and access. Representatives from these groups recommended that Health Canada consider developing a new type of product authorization number appropriate for these types of products that contain cannabis, rather than a Drug Identification Number (DIN) or Natural Product Number (NPN). These respondents supported the need for scientific evidence for the safety and efficacy of these products and recommended that the evidence standards and quality requirements –including packaging and labelling requirements –be similar to those applied to prescription drugs and non-prescription drugs. Specifically, they believed these requirements would be more appropriate for CHPs rather than those applied to NHPs. Additionally, these submissions advised Health Canada to consider how CHPs would be regulated in comparison to self-care products.
With respect to the retail environment, the two organizations that support the regulation of medical professionals were supportive of the proposed approach for provincial and territorial governments to determine place of sale. However, three written submissions from pharmacy organizations, including retailers, felt behind-the-counter at pharmacies would be the most appropriate to prevent youth access while allowing consumers access to a healthcare provider to support informed health decisions. Respondents welcomed further discussions and collaboration.
5.0 Need for scientific evidence
[Questions on scientific evidence for industry respondents and other interested parties: What scientific evidence is currently available/are you aware of scientific evidence that demonstrates the efficacy of cannabis in producing a specific health effect (e.g., relief of headaches, mild muscle pain)? Does your organization currently have scientific evidence of safety and efficacy without the oversight of a practitioner to support the approval of a CHP in Canada? Are you working to generate any evidence?]
Feedback regarding the status of scientific evidence varied, depending on the identity of the respondent. Consumers were not asked questions pertaining to scientific evidence, but did provide related views as part of the online consultation. This feedback indicated that consumers generally felt there was sufficient historical evidence on the health benefits of cannabis. A common theme among consumers was support for further research to provide consumers, retailers and the medical community with trusted, evidence-based information and guidance. There was a particular interest in having more guidance on different strains, as well as concentrations and dosages.
Overall, industry groups and representatives from healthcare organizations demonstrated support for further research and evidence on the safety and efficacy of cannabis in producing a specific health effect for humans or in animals. Written submissions from all government respondents emphasized the need for strong evidence requirements.
In general, industry respondents indicated they were working to generate evidence, and recommended further guidance from Health Canada, including guidance on how to conduct research involving cannabis.
6.0 Next steps
The purpose of this consultation was to seek feedback on the potential market for health products containing cannabis that would not require practitioner oversight.
Health Canada intends to create a scientific advisory committee in 2020 to seek external scientific advice that will support consideration of appropriate safety, efficacy and quality standards for health products containing cannabis that would be safe for use without practitioner oversight.
Scientific information shared through the consultation responses is being reviewed by Health Canada. Analysis will continue as further consultation and scientific advice is sought from external experts. Information gathered will help inform a potential path forward.
- Footnote 1
-
These views were shared by consumer respondents and include conditions that should be treated under a doctor's supervision. They are not health claims approved by Health Canada.
- Footnote 2
-
Respondents could provide more than one option, so percentages total to more than 100%.
- Footnote 3
-
The Cannabis Act creates a strict legal framework for controlling the production, distribution, sale and possession of cannabis across Canada. This includes the sale of cannabis products for medical purposes as well as for non-medical purposes.
- Footnote 4
-
These areas of potential benefit were identified by respondents only, and are not claims approved by Health Canada.