Page 6: Guidelines for Canadian Drinking Water Quality: Guideline Technical Document – Benzene
Part II. Science and Technical Considerations - Continued
For non-smoking Canadians, exposure to benzene is primarily from air (98-99%), with a small proportion from drinking water (1-2%). Benzene has been detected in some foods, and some exposure may occur through automobile-related activities. Smokers may be exposed to significantly higher levels of benzene, in the range of 10 times the daily intake of a non-smoker.
Monitoring results are available for benzene in surface water and groundwater; if benzene is present, its levels are generally less than 1 µg/L.
In Alberta, benzene levels in municipal treated surface water ranged from 0.01 to 4.92 µg/L (mean 0.28 µg/L, 30 samples) for 26 locations from 1998 to mid-2005; levels in more than 96% of samples were less than 1 µg/L. Levels in municipal treated groundwater ranged from 0.01 to 0.23 µg/L (mean 0.097 µg/L, 15 samples) for 11 locations for the same period. These ranges represent 45 detects out of a total of 1500 samples. Approximately 60 samples of raw "feed"
water were analysed for benzene, and all samples had levels below the method detection limit (MDL); the reported MDLs ranged from 1.0 µg/L in 1988 to 0.1 µg/L from 1999 to 2005. Ambient benzene levels from six different river/stream sampling sites as part of an ambient monitoring program from 2000 to 2004 ranged from 0.02 to 0.42 µg/L; this range represented six detects out of a total of 860 samples (Alberta Department of Environment, 2005).
In Saskatchewan, levels of benzene in municipal treated surface water ranged from < 0.2 to < 1 µg/L (mean 0.25 µg/L, 30 samples) in nine locations from 1995 to 2005; levels in all samples were less than 1 µg/L. Municipal treated groundwater levels ranged from 0.1 to 1700 µg/LFootnote 2 (mean 0.71 µg/L, 34 samples) for 13 locations from 1995 to 2005; levels in more than 91% of samples were less than 1 µg/L. The level of benzene in raw (pretreated) water from two locations using surface water and one location using groundwater was 0.2 µg/L (three samples). Municipal treated water from a mixed surface water and groundwater source for three locations over the same period had benzene levels ranging from 0.1 to 1 µg/L (mean 0.46 µg/L, 12 samples); levels in all of the samples were less than 1 µg/L (Saskatchewan Department of Environment and Resource Management, 2005). The reported MDLs ranged from 0.0005 to 1 µg/L, depending on the analytical method used.
Benzene was not detected in raw or municipal treated surface water or groundwater in Newfoundland sampled between 1995 and 2005 (detection limit 1 µg/L) (Newfoundland and Labrador Department of Environment and Conservation, 2005).
In Ontario, the levels of benzene in municipal drinking water systems, using either groundwater or surface water, ranged from below the detection limit (0.05 µg/L) to 0.2 µg/L in 2277 treated water samples between January 2002 and March 2008. The highest benzene level detected in 2762 raw water samples during the same sampling period was 0.35 µg/L (Ontario Ministry of the Environment, 2008).
In Quebec, a total of 2388 samples from municipal treated drinking water were collected between 2001 and 2005 from 191 locations using either groundwater or surface water sources and analysed for benzene. In total, 26 samples taken from 21 locations contained benzene levels ranging from 0.03 to 3.6 µg/L (mean 0.35 µg/L), with only one sample of the 26 having a concentration greater than 1 µg/L. The remaining 2362 samples were reported as being below the limits of detection, with detection limits ranging from 0.03 to 2 µg/L, depending on the accredited laboratory used (Ministère du Développement durable, de l'Environnement et des Parcs du Québec, 2005).
The New Brunswick Analytical Services Laboratory analysed a total of 3903 samples for municipal surface water or groundwater supplies for benzene from January 2001 to May 2008. All samples were reported as non-detects (New Brunswick Department of Environment, 2008).
In Nova Scotia, levels of benzene in 104 municipally treated surface water or groundwater samples from 95 locations from 2001 to 2005 were all below the MDLs, which ranged from 0.5 to 1 µg/L (five samples out of the 104 total samples were reported as non-detects); levels of benzene in 27 raw (pretreated) surface water and groundwater samples were also reported as being below the MDL of 1 µg/L (six samples were reported as non-detects) (Nova Scotia Department of the Environment, 2005).
Benzene has been detected in a variety of foods. The U.S. Food and Drug Administration sponsored a 5-year study to determine the amount of volatile organics in food from 1996 to 2000. Benzene was found in a variety of foods, including dairy products-cheddar cheese, cream cheese, margarine, butter, sour cream; meats and fish-ground beef, bologna, hamburger, cheeseburger, pork, beef frankfurters, tuna canned in oil, chicken nuggets; desserts and baked goods-chocolate cake icing, sandwich cookie, chocolate chip cookies, graham crackers, sugar cookies, cake doughnuts with icing, apple pie, sweet roll danish, blueberry muffins; nuts and nut products-mixed nuts, peanut butter; fruits and vegetables-bananas, avocados, oranges, strawberries; and eggs. Concentrations of benzene in foods generally ranged from 1 to 190 µg/kg; some examples include the following: ground beef ( 9-190 µg/kg), bananas (11-132 µg/kg), carbonated cola (1-138 µg/kg), and coleslaw with dressing (11-102 µg/kg) (Fleming-Jones and Smith, 2003). Benzene detections in the above food types represented only a few detects per sample, indicating that food does not represent a significant source of benzene exposure. Further support is provided by a Canadian review of benzene exposures (Environment Canada and Health and Welfare Canada, 1993).
In another study by the U.S. FDA (2006), the Center for Food Safety and Applied Nutrition conducted an initial limited survey on benzene levels in beverages, with a focus on soft drinks that contain both benzoate salts and ascorbic or erythorbic acid. Over 100 soft drinks and other beverage samples were collected from retail stores in Maryland, Virginia, and Michigan. Two beverage products containing added benzoates and 27 beverage products containing both added benzoates and ascorbic acid had benzene levels above 1 µg/L. Four cranberry beverage products and one orange beverage product with added ascorbic acid and natural levels of benzoic acid (i.e., no added benzoates) also contained benzene above 1 µg/L. In general, however, most of the beverages sampled contained either no detectable benzene or levels below 1 µg/L. Exposure to heat and light can stimulate the formation of benzene in some beverages that contain benzoate salts and ascorbic acid (vitamin C). Benzoate salts are naturally present in some fruits and their juices, or sodium or potassium benzoate may be added to beverages to prevent the growth of bacteria, yeast, and moulds.
In general, mean benzene concentrations in ambient air were found to be highest at sites influenced by industrial sources and urban sites and lowest at rural and suburban sites (Environment Canada, 2001). Mean benzene concentrations in Canadian ambient air between 1989 and 1998 ranged from 1.8 to 3.6 µg/m3 for typical urban sites with no industrial source influences; mean benzene levels were found to range from as high as 10.3 µg/m3 at an urban site in Sault Ste. Marie, Ontario, influenced by emissions from a coke oven/iron and steel mill facility to as low as 0.3 µg/m3 at a rural remote site in Kejimkujik National Park, Nova Scotia (Environment Canada, 2001). Survey data from the period 1995-1997 showed mean concentrations at the urban/suburban sites ranging from 1.0 to 3.5 µg/m3, with approximately 78% (31 out of 40) of sites recording mean concentrations of less than 2.5 µg/m3 (Environment Canada, 2001). Mean concentrations for rural sites reportedly ranged from 0.3 and 0.8 µg/m3. Sites near roadways or industrial sources had mean benzene concentrations ranging from 4.1 to 13.1 µg/m3 (Environment Canada, 2001).
Benzene levels in indoor air are generally higher than those in outdoor air. Sources of benzene in indoor air include glues, paints, furniture wax, and some detergents. Zhu et al. (2005) measured indoor and outdoor air levels of benzene for 75 residences in Ottawa, Ontario, during the winter of 2002-2003. Indoor temperatures remained relatively constant (19 ± 2°C), and the majority of participating homes were single family homes located in residential areas using natural gas as the heating source. The average age of the houses was 37 years, with a range from newly constructed to over 100 years old. Roughly 13% (10 homes) of the homes in the study were homes with smokers. Mean indoor air levels of benzene were reported as 2.85 µg/m3 (range 0.025-20.99 µg/m3),Footnote 3 with a detection frequency of 97%; outdoor air sampling revealed a mean benzene level of 1.19 µg/m3 (0.025-16.88 µg/m3), with a detection frequency of 62%.
The general population may also be exposed to benzene through automobile-related activities and cigarette smoking. An average smoker (smoking 32 cigarettes per day with an average tar content) inhales approximately 1.8 mg of benzene per day, which is about 10 times the daily intake of a non-smoker; environmental tobacco smoke can also result in measurable increases in benzene intake (Wallace, 1989b,1996; Thomas et al., 1993). Duarte-Davidson et al. (2001) compared the daily doses of rural non-smokers, urban non-smokers, urban passive smokers (non-smokers exposed to secondhand smoke), and urban smokers and found very little difference between the rural non-smokers' estimated absorbed dose of 70-75 µg/day and the urban non-smokers' estimated absorbed dose of 89-95 µg/day; the absorbed dose for passive urban smokers was estimated to be 116-122 µg/day, whereas smokers were estimated to be exposed to 516-522 µg/day. On average, non-smokers in urban and rural environments have estimated benzene intakes of 1.15 and 1.5 µg/kg bw per day. Daily doses were determined using time-activity patterns and inhalation and absorption rates, in conjunction with measured benzene air concentrations.
Automobile-related activities can contribute to increased benzene intake through inhalation of gasoline fumes and from tailpipe emissions. Increased benzene exposure has been attributed to driving times, filling gas tanks, and indoor air of homes with attached garages (Wallace, 1989b). A 1990 German study analysed factors predicting human exposures to VOCs and found that cigarette smoking was the most significant determinant of benzene exposure; automobilerelated activities, such as refuelling and driving, were found to be the second highest source of benzene exposure (Hoffmann et al., 2000).
Benzene contamination of soil generally results from the spilling or leaking of gasoline or other benzene-containing petroleum products from containment vessels, such as underground storage tanks. The primary pathways responsible for benzene loss from soil are volatilization to the atmosphere, runoff to surface water and groundwater, and, to a much lesser extent, biodegradation (Environment Canada and Health and Welfare Canada, 1993). Hydrocarbondegrading microorganisms are ubiquitous in soil, and both sorbed and vapour-phase benzene are likely biodegraded under aerobic conditions (Rosenberg and Gutnick, 1981; English and Loehr, 1991); biodegradation practically ceases when conditions become anaerobic (Smith, 1990; Aelion and Bradley, 1991; Barbaro et al., 1991). Soil contamination does not lead directly to significant levels of human exposure to benzene, since benzene volatilizes rapidly from soil (IPCS, 1993). Benzene levels in the soil surrounding industrial facilities that produce or use benzene have been reported to range between < 2 and 191 µg/kg (U.S. EPA, 1979; IARC, 1982).
Exposure to benzene in drinking water was previously assessed (Health Canada, 1986) using ingestion as the only route of exposure. Owing to benzene's high volatility, exposure by inhalation and dermal absorption during bathing and showering may also serve as important routes of exposure. Lindstrom et al. (1994) carried out a study looking at the exposure to benzene while showering with gasoline-contaminated groundwater in a home in North Carolina. The groundwater had a measured benzene concentration of 292 µg/L. Three 20-minute showers on consecutive days were reported to have resulted in peak shower stall concentrations of 800- 1670 µg/m3, with bathroom concentrations reaching 370-500 µg/m3 and concentrations in the remainder of the house peaking 0.5-1 hours later at 40-140 µg/m3. The dose of benzene inhaled during the 20-minute shower ranged from 80 to 100 µg. A dermal dose of 160 µg was also determined using measured breath concentrations. The combined dose of about 250 µg from the 20-minute shower was found to be within the same magnitude of the mean total daily inhalation dose of about 200 µg for all non-smokers in the Total Exposure Assessment Methodology (TEAM) study (assuming 15 µg/m3 × 14 m3/day alveolar inspiration) (Wallace, 1987).
To assess the overall exposure to benzene in drinking water, the relative contribution of each exposure route is assessed through a multi-route exposure assessment approach (Krishnan, 2004). Contributions developed through this approach are expressed in litre-equivalents (L-eq) per day. Both the dermal and inhalation routes of exposure for a volatile organic chemical are considered significant if they contribute to at least 10% of the drinking water consumption level (Krishnan, 2004).
To determine whether dermal exposure represents a significant route of exposure for benzene, tier 1 of the multi-route exposure assessment determines whether or not this route of exposure contributes a minimum of 10% of the drinking water consumption level (i.e., 10% of 1.5 L = 0.15 L). For a tier 1 goal of 0.15 L-eq, the skin permeability coefficient (Kp) for benzene should be higher than 0.028 cm/h. Since the Kp for benzene of 0.14 cm/h (Nakai et al., 1997) is greater than 0.028 cm/h, exposure to benzene via dermal absorption from bathing or showering is considered significant. Tier 2 of the assessment is then used to calculate the litre-equivalent value, using the following equation (Krishnan, 2004):
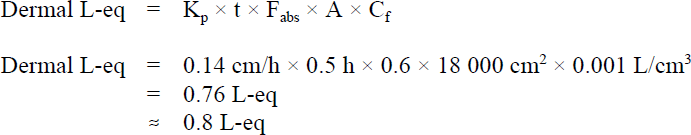
Figure 1 - Text Equivalent
The litre-equivalent value for the dermal absorption of benzene is 0.8 litre-equivalents per day. This value is calculated by multiplying the following factors: 0.14 centimetres per hour; 0.5 hours; 0.6; 18000 centimetres squared; and 0.001 litres per centimetre cubed.
where:
- Kp is the skin permeability coefficient of 0.14 cm/h (Nakai et al., 1997)
- t is the duration of the shower or bath, assumed to be 0.5 h
- Fabs is the fraction of dose absorbed, assumed to be 0.6 (Lindstrom et al., 1994)
- A is the area of skin exposed, assumed to be 18 000 cm2 for adults
- Cf is the conversion factor from cm3 to litres.
A two-tier assessment was also used to evaluate the inhalation route of exposure. Similar to the approach used for dermal exposure, tier 1 of the assessment determines whether the inhalation of benzene during bathing or showering is likely to contribute at least 10% of the drinking water consumption level. For a tier 1 goal of 0.15 L-eq, the air to water benzene concentration (Fair:water) value should be greater than 0.000 89. Using the estimated Henry's law constant (Kaw) obtained from the U.S. EPA's EPI Suite program (U.S. EPA, 2000), the Fair:water value for benzene was determined using the following equation by Krishnan (2004):
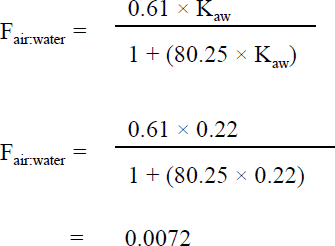
Figure 2 - Text Equivalent
The air to water benzene concentration is 0.0072. This value is calculated by dividing the product of 0.61 and 0.22 by the product of 80.25 and 0.22 added to 1.
where:
- Kaw is the unitless Henry's law constant of 0.22 at 25°C (U.S. EPA, 2000)
- 0.61 is 61% transfer efficiency (McKone and Knezovich, 1991)
- 80.25 is the ratio of the volume of air in an average bathroom (6420 L) to the average volume of water (80 L) used during the showering/bathing event (Krishnan, 2004).
Since the Fair:water value is greater than 0.000 89, exposure to benzene via inhalation from bathing or showering is considered to be significant. Tier 2 of the assessment calculates what the litre-equivalents should be as a function of using the following formula (Krishnan, 2004):
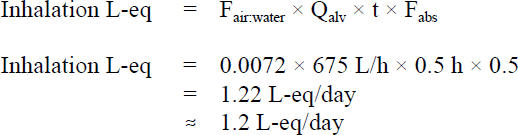
Figure 3 - Text Equivalent
The litre-equivalent value for the inhalation absorption of benzene is 1.2 litre-equivalents per day. This value is calculated by multiplying the following factors: 0.0072; 625 litres per hour; 0.5 hours; and 0.5.
where:
- Fair:water is the ratio (partitioning) of air to water benzene concentrations
- Qalv is the adult alveolar ventilation rate, assumed to be 675 L/h
- t is the exposure duration, assumed to be 0.5 h
- Fabs is the fraction absorbed, assumed to be 0.5 (Perbellini et al., 1988; Pekari et al., 1992; ATSDR, 2007).
It should be noted that this multi-route exposure assessment is a conservative approach used to estimate the contribution that both the dermal and inhalation routes of exposure make towards total exposure. Using physiologically based pharmacokinetic (PBPK) modelling to estimate the litre-equivalent contributions to the total daily dose from the dermal and inhalation pathways does not take into account exposure to benzene metabolites. Therefore, the approach does not place any "toxicological"
weight on a particular route of exposure due to metabolite production.
Using the above approach, the litre-equivalent exposure was calculated as 0.8 L-eq for the dermal route and 1.2 L-eq for the inhalation route. Adding these values to the standard Canadian drinking water consumption rate of 1.5 L/day results in a total litre-equivalent daily exposure of 3.5 L-eq.
In a Canadian review of benzene exposures (Environment Canada and Health and Welfare Canada, 1993), it was concluded that food and drinking water each contributed a total daily benzene intake of only 0.02 µg/kg bw; the total daily intake of benzene from airborne exposures was reported to be 2.4 µg/kg bw per day (3.3 µg/kg bw per day if exposed to cigarette smoke). It can therefore be concluded that airborne exposure accounts for an estimated 98-99% of total benzene intake for Canadian non-smokers.