Page 6: Guidelines for Canadian Drinking Water Quality: Guideline Technical Document – Carbon Tetrachloride
Part II. Science and Technical Considerations - Continued
Canadians can be exposed to carbon tetrachloride through its presence in air and drinking water. In addition, certain segments of the population may be exposed through the use of specific consumer products or in occupational settings. Although some exposure data are available, they are not sufficient to modify the default allocation factor for drinking water of 20%.
Owing to its high volatilization from water, carbon tetrachloride concentrations are normally low in surface water (≤1 µg/L). However, in groundwater systems where volatilization and biodegradation are limited, concentrations may be higher if contamination has occurred in the vicinity and leaching has taken place.
Concentrations of carbon tetrachloride have been measured in various water sources at limited locations across Canada. In Québec, carbon tetrachloride was detected in 10 distribution systems at a maximum concentration of 1 µg/L between the years 2001 and 2005 (Tremblay and Robert, 2005).
In Ontario, carbon tetrachloride levels above 0.5 µg/L have very rarely been detected. In over 5700 analysis carried out over the period 2004-2009, levels above 0.5 µg/L were found in only two instances with the highest being 1.2 µg/L (Ontario Ministry of the Environment, 2010). Carbon tetrachloride was not detected in raw or treated groundwater or surface water from First Nations water supplies sampled in southern and northern Ontario during various years between 1996 and 2004 (Health Canada, 2005).
In Saskatchewan, carbon tetrachloride was not detected (detection limit 1 µg/L) in raw water, wells, or treated water sampled between 1992 and 2005 (Saskatchewan Environment, 2005).
Carbon tetrachloride is present in ambient air due to past and present releases resulting from production, disposal, or use (ATSDR, 2005). Recent surveys conducted on carbon tetrachloride in indoor and outdoor air in Canada have shown that mean levels are below 1 µg/m³.
Ambient air sampled at 17 rural and 40 urban sites across Canada (2004-2005), for a total of 6992 samples, showed an overall mean level of 0.60 µg/m³ (range 0.34-1.02 µg/m³) (Dann, 2006). Similar outdoor air levels were seen in a 2005 local survey of backyards of 48 homes in Windsor, Ontario, during the winter (range 0.47-0.72 µg/m³; overall mean 0.60 µg/m³) and summer (range 0.48-0.70 µg/m³; overall mean 0.59 µg/m³) (Health Canada, 2006a).
Simmonds et al. (1998) measured global atmospheric concentrations (lower troposphere) of carbon tetrachloride at five coastal monitoring stations around the world from 1978 to 1996. Peak levels were seen during 1989-1990, at 104.4 ppt (0.653 µg/m³). Less recent data from 1976 showed levels in North America ranging from 0.33 to 0.99 µg/m³, with a mean level of 0.86 µg/m³ (IPCS, 1999). Similar levels (0.87 µg/m³) were seen in the northern hemisphere between 1979 and 1981 (IPCS, 1999). Shah and Heyerdahl (1988) reported an average concentration of carbon tetrachloride of 0.168 ppb (1.1 µg/m³) in ambient air in the United States based on 4913 ambient air samples taken at various sites, and these levels have since been decreasing.
Member companies of the Canadian Chemical Producers' Association have reported that since 1992, emissions of carbon tetrachloride in Canada have been reduced by over 99%; the emissions went from 58.190 tonnes in 1992 down to 0.024 tonnes in 2004 (CCPA, 2006).
According to ATSDR (2005), carbon tetrachloride also appears to be a common contaminant of indoor air; the sources of exposure appear to be building materials or products, such as cleaning agents, used in the home. However, it should be noted that carbon tetrachloride has been eliminated from production, import, and export in Canada, and is on a list of restricted substances under the Canadian Environmental Protection Act (CEPA). Sources in Canada are therefore expected to be limited.
Indoor air levels were sampled in 48 homes in Windsor, Ontario, during the winter and summer of 2005 (Health Canada, 2006a). In winter, the concentration of carbon tetrachloride ranged from 0.035 to 3.31 µg/m³, with an overall mean value of 0.60 µg/m³; in summer, concentrations ranged from 0.24 to 7.30 µg/m³, with an overall mean of 0.72 µg/m³.
In the United States, typical indoor air concentrations of 1 µg/m³ were reported for 600 homes sampled in several states (Wallace, 1986), whereas slightly higher mean levels (2.6 µg/m³) were seen in another study in which 2120 indoor air samples were taken (although carbon tetrachloride was detected in less than half the samples) (Shah and Heyerdahl, 1988).
Data on the residues of carbon tetrachloride in Canadian food commodities were not available. In the past, carbon tetrachloride was used in agriculture as a grain fumigant; as a result, residues occurred in grain and food products such as bread prepared using the fumigated grain. In Canada, the Pest Management Regulatory Agency (PMRA, 2006) no longer supports the use of ozone-depleting chemicals such as carbon tetrachloride as formulants in pesticides; as a result, no new pesticide registrations or renewals will be issued for products containing them. Since carbon tetrachloride is no longer used for grain fumigation in Canada, and its use in other countries is limited, exposure to carbon tetrachloride via this route is not of concern.
No significant amount of carbon tetrachloride was found in food in the United States (U.S. FDA, 2003; ATSDR, 2005). In a total diet study summary from 1991 to 2001 (U.S. FDA, 2003), carbon tetrachloride residues were rarely detected. Detected concentrations of carbon tetrachloride ranged from 0.0040 to 0.0310 mg/kg. As a result, food does not represent a significant source of carbon tetrachloride exposure.
No studies have been located that measure human exposure from inhalation of carbon tetrachloride volatilized from tap water as a result of activities such as showering and bathing (IPCS, 1999). However, owing to carbon tetrachloride's high volatility, exposure by inhalation and through the skin during bathing and showering may also serve as important routes of exposure. Tancrède et al. (1992) studied the volatilization of various volatile organic compounds (VOCs) from tap water from household activities such as showering and found that at a temperature of 25°C, the fraction of volatilized carbon tetrachloride was approximately 40% and increased to over 70% when the temperature increased to either 33°C or 42°C (an ~50% increase in volatilization).
To assess the overall exposure to carbon tetrachloride in drinking water, the relative contribution of each exposure route is assessed through a multi-route exposure assessment approach (Krishnan, 2004). Contributions developed through this approach are expressed in litre equivalents (L-eq) per day. Both the dermal and inhalation routes of exposure for a VOC are considered significant if they contribute at least 10% of the drinking water consumption level (Krishnan, 2004).
Dermal exposure
To determine whether dermal exposure represents a significant route of exposure for carbon tetrachloride, tier 1 of the multi-route exposure assessment determines whether or not this route of exposure contributes a minimum of 10% of the drinking water consumption level (i.e., 10% of 1.5 L = 0.15 L). For a tier 1 goal of 0.15 L-eq, the skin permeability coefficient (Kp) for VOCs should be higher than 0.024 cm/h. Since the Kp for carbon tetrachloride of 0.16 cm/h is greater than 0.024 cm/h, exposure to carbon tetrachloride via dermal absorption from bathing or showering is considered significant. Tier 2 of the assessment is then used to calculate the L-eq value, using the following equation (Krishnan, 2004):

Figure 1 - Text Equivalent
The litre-equivalent value for the dermal absorption of carbon tetrachloride is 1.0 litre-equivalent per day. This value is calculated by multiplying the following factors: 0.16 centimetres per hour; 0.5 hours; 0.7; 18000 centimetres squared; and 0.001 litres per centimetre cubed.
where:
- Kp is the skin permeability coefficient of 0.16 cm/h (Krishnan, 2004);
- t is the duration time of the shower or bath assumed to be 0.5 h;
- Fabs is the fraction of dose absorbed assumed to be 0.7 (Krishnan, 2003a,b);
- A is the area of skin exposed assumed to be 18000 cm² for adults; and
- Cf is the conversion factor from cm³ to litres.
Inhalation exposure
A two-tier assessment was also used to evaluate the inhalation route of exposure. Similar to the approach used for dermal exposure, tier 1 of the assessment determines whether inhalation of carbon tetrachloride during bathing or showering is likely to contribute at least 10% of the drinking water consumption level. For a tier 1 goal of 0.15 L-eq, the air to water concentration (Fair:water) value for VOCs should be greater than 0.00063. Using the estimated Henry's law constant obtained from the U.S. EPA EPI Suite program (U.S. EPA, 2000), the Fair:water value for carbon tetrachloride was determined to be 0.0075. Since the Fair:water value is greater than 0.00063, exposure to carbon tetrachloride via inhalation from bathing or showering is considered to be significant. Tier 2 of the assessment calculates what the L-eq should be as a function of using the following formula (Krishnan, 2004):
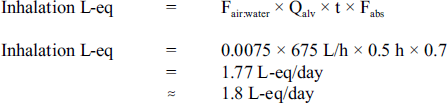
Figure 2 - Text Equivalent
The litre-equivalent value for the inhalation absorption of carbon tetrachloride is 1.8 litre-equivalents per day. This value is calculated by multiplying the following factors: 0.0075; 625 litres per hour; 0.5 hours; and 0.7.
where:
- Fair:water is the ratio (partitioning) of air to water concentrations of carbon tetrachloride;
- Qalv is the adult alveolar ventilation rate assumed to be at 675 L/h;
- t is the time of exposure duration assumed to be 0.5 h; and
- Fabs is the fraction absorbed, 0.7 (based on Krishnan, 2003a,b).
It should be noted that this multi-route exposure assessment is a conservative approach used to estimate the contribution that both the dermal and inhalation routes of exposure make towards total exposure. Using physiologically based pharmacokinetic (PBPK) modelling to estimate the L-eq contributions to the total daily dose from the dermal and inhalation pathways does not take into account exposure to carbon tetrachloride metabolites. Therefore, the approach does not place any "toxicological" weight on a particular route of exposure due to metabolite production.
Using the above approach, the L-eq/day exposure was calculated as 1.0 L-eq/day for the dermal route and 1.8 L-eq/day for the inhalation route. Adding these values to the standard Canadian drinking water consumption rate of 1.5 L/day results in a total litre-equivalent daily exposure of 4.3 L-eq/day.