Drinking water screening value for perfluorononanoic acid (PFNA) – Technical summary

Download the alternative format
(PDF format, 164 KB, 5 pages)
Organization: Health Canada
Published: November 2020
A drinking water screening value of 20 ng/L (0.000020 mg/L) is established for perfluorononanoic acid (PFNA)
Screening values
Health Canada's screening values identify limits for contaminants in water that could be used as a source of drinking water. A lifetime of exposure to these contaminants up to the screening value, both by drinking the water or by using it for showering or bathing, is not expected to increase health risks for any Canadian, including children.
Screening values are established for contaminants that are not commonly found in Canadian drinking water (either source or treated) and therefore Guidelines for Canadian Drinking Water Quality are not established. Health Canada establishes screening values for contaminants at the request of federal departments, provinces and territories (jurisdictions). These requests are usually made when there is a concern for human health because the presence of a contaminant is suspected or detected in local source water and that contaminant does not have an established limit in drinking water. Since 2020, the technical summaries for screening values are typically published online when Health Canada expects that screening values may be needed by more than one stakeholder or jurisdiction.
Screening values do not replace or supersede existing regulations. However, screening values may help jurisdictions and the public understand the potential health effects of a contaminant.
Screening values are based on a review of scientific research and international regulatory information available at the time of their development. In addition, screening values are externally peer-reviewed to ensure scientific integrity.
Health Canada is committed to keeping pace with new science, including the potential health risks from contaminants that are not typically found in drinking water and do not have Guidelines for Canadian Drinking Water Quality. To this end, Health Canada includes contaminants with screening values in its cyclical prioritization of contaminants for full guideline development.
Exposure considerations
Identity and sources
Perfluorononanoic Acid (PFNA) belongs to a group of chemicals referred to as perfluoroalkyl substances. They have been used to make carpets, clothing, fabrics for furniture, paper packaging for food and other materials. They are also used for firefighting at airfields and in a number of industrial processes. Although PFNA is not manufactured in Canada, its past wide variety of uses, as well as its resistance to environmental degradation, has resulted in its detection in the environment, as well as in human blood samples.
Exposure
The major sources of exposure to PFNA are expected to be food and consumer products. However, drinking water can represent a significant source of exposure in the proximity of point sources such as fire fighting facilities where repeated fire training exercises occur or where large amounts of aqueous film-forming foams are used to extinguish fires fueled by flammable or combustible liquids. Additional point sources may include facilities where perfluoroalkylated substances (PFAS) or PFAS precursors (e.g., fluorotelomer alcohols) are stored and handled. Exposure to PFNA through bathing and showering is expected to be negligible due to its physical and chemical properties.
PFNA has not been detected in treated Canadian drinking water but has been found in Canadian surface water (<2.0 ng/L) and raw drinking water samples (<68 ng/L) (De Silva et al., 2009; Ministère du development durable, de l'environnement, de la faune et des parcs, 2012).
Health considerations
PFNA is rapidly absorbed following oral exposure and distributed to blood and liver. Similar to other PFAS, PFNA is chemically unreactive and not metabolized. Elimination is slow and occurs in urine and feces. Large inter-species differences exist between humans and other species: PFNA half-lives in the range of a few weeks have been measured in rodents whereas estimated elimination half-lives in humans are in the range of 2.1 to 4.3 years (Olsen et al., 2007; Bartell et al., 2010; Seals et al., 2011).
Over 40 epidemiological studies have investigated associations between serum PFNA concentrations and adverse health effects including liver, kidney and thyroid effects, immunological and metabolic effects, as well as reproductive and developmental effects. Positive associations were observed for effects including reduced liver and kidney function, lipidemia and immune functions. Due to study design, bias and confounders, epidemiological studies could not be used for dose-response assessment.
No studies have investigated chronic toxicity or carcinogenic potential of PFNA.
Selection of key study
In animal studies, oral exposure to PFNA was associated with adverse health effects of the liver and kidney as well as adverse reproductive effects. Hepatic toxicity in rats was the effect observed at the lowest level of exposure and was consistently reported at low doses across studies (Stump et al., 2008; Mertens et al., 2010; Wolf et al., 2010; Das et al., 2015) and serves as the critical (key) endpoint to establish the tolerable daily intake. Stump et al. 2008 was selected as the key study because it reported the lowest level of exposure at which toxic effects to the liver are observed.
Derivation of the screening value
The screening value of 20 ng/L has been derived based on the data available, as described below.
A benchmark dose modelling was performed on dose response data from Stump et al. (2008) which established a point of departure of 0.012 mg/kg body weight (bw) per day. This was further adjusted by a chemical-specific adjustment factor of 73 to account for the differences in toxic kinetics between human and rats based on literature data reported by Ohmori et al. (2003) and Zhang et al. (2013) resulting in a human equivalent point of departure (PODHED) of 1.64 × 10-4 mg/kg bw per day.
A screening value of 20 ng/L (rounded) can be derived following the standard equation:
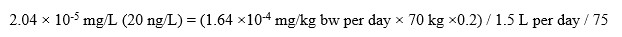
Text description
This equation calculates screening value for PFNA. The screening value is calculated by multiplying the point of departure for the human equivalent dose of 0.164 micrograms per kilogram of body weight per day by a body weight of 70 kilograms and by an allocation factor of 0.20 and then dividing by a volume of water ingested per day of 1.5 litres and by the safety factor applied to the point of departure for the human equivalent dose of 75. This equals a screening value of 20 nanograms per litre for PFAS.
Where:
- 1.64 ×10-4 bw/day is the PODHED for hepatocellular hypertrophy increase based on a 2- generation study in rats by Stump et al. (2008).
- 70 kg is taken as the mean body weight estimated for a Canadian adult.
- 0.2 is the default allocation factor for drinking water; the proportion of exposure to PFNA from drinking water, as opposed to other environmental media (i.e., food, air, soil, consumer products, etc.) in the absence of other exposure data, the default floor value used is 20% (Krishnan and Carrier, 2013).
- 1.5 L per day is the estimated daily volume of tap water by an adult.
- 75 is the safety factor applied to the PODHED (2.5 for interspecies toxicodynamic differences; 10 for intraspecies variability; 3 for database deficiencies).
International considerations
Drinking water quality guidelines, standards and/or guidance established by foreign governments or international agencies may vary due to the science available at the time of assessment, as well as the utilization of different policies and approaches, such as the choice of key study, and the use of different consumption rates, body weights and allocation factors.
New Jersey established a health-based maximum contaminant level for PFNA in drinking water of 13 ng/L based on increased liver weight in pregnant mice exposed to PFNA and a cumulative uncertainty factor of 1000 (New Jersey Water Quality Institute, 2015). The Texas Commission on Environmental Quality established a Tier 1 Residential Protective Concentration Level of 290 ng/L for PFNA (TCEQ, 2016). This value was based on a no observed adverse effects level of 1mg/kg bw per day for spleen apoptosis and a cumulative uncertainty factor of 1000 in addition to a factor of 81 to account for toxicokinetics (i.e., clearance rates) between rats and humans.
Contact us
For more information, please contact us at hc.water-eau.sc@canada.ca.
References
Bartell, S.M., Calafat, A.M., Lyu, C., Kato, K., Ryan, P.B. and Steenland, K. (2010). Rate of decline in serum PFOA concentrations after granular activated carbon filtration at two public water systems in Ohio and West Virginia. Environ. Health Perspect., 118(2): 222-228.
Das, K.P., Grey, B.E., Rosen, M.B., Wood, C.R., Tatum-Gibbs, K.R., Zehr, R.D., Strynar, M.J., Lindstrom, A.B. and Lau, C. (2015). Developmental toxicity of perfluorononanoic acid in mice. Reproductive Toxicology, 51: 133-144.
De Silva, A.O., Muir, D.C.G. and Mabury, S.A. (2009). Distribution of perfluorocarboxylate isomers in select samples from the North American environment. Environmental Toxicology and Chemistry, 28(9): 1801-1814.
Krishnan, K. and Carrier, R. (2013). The use of exposure source allocation factor in the risk assessment of drinking-water contaminants. J. Toxicol. Environ. Health, Part B: Crit. Rev., 16 (1): 39-51.
Lide, D.R. (Eds.) (1996). CRC Handbook of Chemistry and Physics, 77th Edition.
Mertens, J.J.W.M., Sved, D.W., Marit, G.B., Myers, N.R., Stetson, P.L., Murphy, S.R., Schmit, B., Shinohara, M. and Farr, C.H. (2010). Subchronic toxicity of S-111-S-WB in Sprague Dawley rats. Int. J. Toxicol., 29(4): 358-371.
Ministère du Développement durable, de l'Environnement, de la Faune et des Parcs. (2012). Les composés perfluorés dans les cours d'eau et l'eau potable du Québec méridional. Ministère du Développement durable, de l'Environnement, de la Faune et des Parcs, Direction du suivi de l'état de l'environnement.
New Jersey Water Quality Institute. (2018). Health-based maximum contaminant level support document: Perfluorononanoic acid (PFNA). New Jersey Water Quality Institute Health Effects Subcommittee.
Ohmori, K., Kudo, N., Katayama, K. and Kawashima, Y. (2003). Comparison of the toxicokinetics between perfluorocarboxylic acids with different carbon chain length. Toxicology. Toxicology, 184(2-3): 135-140.
Olsen, G.W., Burris, J.M., Ehresman, D.J., Froelich, J.W., Seacat, A.M., Butenhoff, J.L. and Zobel, L.R. (2007). Half-life of serum elimination of perfluorooctanesulfonate, perfluorohexanesulfonate, and perfluorooctanoate in retired fluorochemical production workers. Environ. Health Perspect., 115(9): 1298-1305.
Seals, R., Bartell, S.M. and Steenland, K. (2011). Accumulation and clearance of perfluorooctanoic acid (PFOA) in current and former residents of an exposed community. Environ. Health Perspect., 119(1): 119-124.
Stump, D.G., Holson, J.F., Murphy, S.R., Farr, C.H., Schmit, B. and Shinohara, M. (2008). An oral two-generation reproductive toxicity study of S-111-S-WB in rats. Reproductive Toxicology, 25(1): 7-20.
TCEQ. (2016). Toxicological evaluation of perfluoro compounds (e.g., PFOS, PFOA). Texas Commission on Environmental Quality. Available at https://www.tceq.texas.gov/toxicology#pfcs
Wolf, C.J., Zehr, R.D., Schmid, J.E., Lau, C. and Abbott, B.D. (2010). Developmental effects of perfluorononanoic acid in the mouse are dependent on peroxisome proliferator-activated receptor-alpha. PPAR Research, Article number 282896.
Zhang, T., Sun, H., Lin, Y., Qin, X., Zhang, Y., Geng, X. and Kannan, K. (2013a). Distribution of poly- and perfluoroalkyl substances in matched samples from pregnant women and carbon chain length related maternal transfer. Environ. Sci. Technol., 47(14): 7974-7981.
Zhang, Y., Beesoon, S., Zhu, L. and Martin, J.W. (2013b). Biomonitoring of perfluoroalkyl acids in human urine and estimates of biological half-life. Environmental science and technology. Environ. Sci. Technol., 47(18): 10619-10627.