Drinking water screening value for sulfolane - Technical summary
Download the alternative format
(PDF format, 554 KB, 5 pages)
Organization: Health Canada
Date published: November 2020
A drinking water screening value of 0.3 mg/L (300 µg/L) is established for Sulfolane
Screening values
Health Canada's screening values identify limits for contaminants in water that could be used as a source of drinking water. A lifetime of exposure to these contaminants up to the screening value, both by drinking the water or by using it for showering or bathing, is not expected to increase health risks for any Canadian, including children.
Screening values are established for contaminants that are not commonly found in Canadian drinking water (either source or treated) and therefore Guidelines for Canadian Drinking Water Quality are not established. Health Canada establishes screening values for contaminants at the request of federal departments, provinces and territories (jurisdictions). These requests are usually made when there is a concern for human health because the presence of a contaminant is suspected or detected in local source water and that contaminant does not have an established limit in drinking water. Since 2020, the technical summaries for screening values are typically published online when Health Canada expects that screening values may be needed by more than one stakeholder or jurisdiction.
Screening values do not replace or supersede existing regulations. However, screening values may help jurisdictions and the public understand the potential health effects of a contaminant.
Screening values are based on a review of scientific research and international regulatory information available at the time of their development. In addition, screening values are externally peer-reviewed to ensure scientific integrity.
Health Canada is committed to keeping pace with new science, including the potential health risks from contaminants that are not typically found in drinking water and do not have Guidelines for Canadian Drinking Water Quality. To this end, Health Canada includes contaminants with screening values in its cyclical prioritization of contaminants for full guideline development.
Exposure considerations
Identity and sources
Sulfolane is an industrial chemical of anthropogenic source. It is a colourless liquid primarily used as a solvent in gas treatment processes such as the removal of acid gases from natural gas, and elimination of hydrogen sulphide from shale and coal. Based on its physical and chemical properties, it is highly soluble in water and highly mobile in the subsurface. Its biodegradation in groundwater, where there is a lack of nutrients or under anaerobic conditions, may be very slow. As such, in areas where it is released in the environment, sulfolane may accumulate in nearby groundwater.
Exposure
Sulfolane exposure is expected to occur primarily through oral intake of drinking water. Due to its low volatility and low skin permeability constant, exposure through bathing and showering with drinking water is negligible. It partitions in water and hence dry inhalable dusts are not expected to contain sulfolane, exposures from manufactured items is unlikely, as it is not commonly used in manufactured items. Data on sulfolane levels in vegetation of a contaminated wetland suggest uptake of sulfolane by plants that may be consumed by humans although concentrations varied considerably across samples, species and parts of the plant. Maximum concentrations of sulfolane reported were 256 mg/kg and 185 mg/L sulfolane in plants and water, respectively (Headley et al., 1999; CCME, 2006).
Reports on the presence of sulfolane in water are limited to data collected in the vicinity of gas processing facilities in Western Canada and Alaska. Samples in monitoring wells and private well water near the refinery have been detected at levels as high as 319 μg/L and 946 μg/L, respectively.
No data are available on concentrations of sulfolane in Canadian drinking water.
Health considerations
Limited toxicokinetic data are available for sulfolane, which is readily absorbed through the oral route in blood via the gastrointestinal tract but not through human skin (Andersen et al., 1976). Urine appears to be the primary elimination pathway for sulfolane (Andersen et al., 1977).
Toxicological database for sulfolane is limited. No epidemiological studies are available and animal studies are limited to oral subchronic toxicity studies. Genotoxicity and mutagenicity evaluated in various in vitro tests are almost exclusively negative (CCME, 2006; NCEA 2012). There is a lack of information at this time to perform a cancer risk assessment.
Overall, oral exposure to sulfolane in experimental animals was associated with immunological, renal and reproductive and developmental effects (Huntingdon Life Science (HLS), 2001; Ministry of Health and Welfare Japan, 1996; Zhu et al., 1987).
Selection of key study
The endpoint observed at the lowest exposure doses and with the strongest causal link was determined to be the decreases in white blood cell (WBC) counts in female rats in the study by HLS (2001). For this reason, and because this study was also Good Laboratory Practices compliant, peer-reviewed with adequate methodological and data reporting and hence supported the derivation of the tolerable daily intake, HLS (2001) was selected as the key study.
Derivation of the screening value
A human equivalent dose of 3.37 mg/kg body weight (bw) per day, representing the 95% lower confidence limit on the benchmark dose (BMDL HED) has been derived by an expert panel established by Toxicology Excellence in Risk Assessment (TERA, 2014a; 2014b). This dose is based on a 13-week drinking water study that showed decreased WBC counts in rats (HLS, 2001). The expert panel was put together to review and discuss human health toxicity values for sulfolane for the Alaska Department of Environmental Conservation.
To derive the tolerable daily intake (TDI), an uncertainty factor of 270 (3 for interspecies differences upon using a human equivalent dose; 10 for human variability due to lack of information on sensitive population; 3 instead of 10 for extrapolation from a subchronic study because the severity of sulfolane effects did not increase over time, the effects may be reversible upon cessation of exposure (Ministry of Health And Welfare Japan, 1996) and sulfolane is not likely to accumulate in the body; 3 for database deficiencies as there are uncertainties surrounding the quality of the available data, and a two-generational reproductive study is missing), is applied to the BMDLHED resulting in a value of 0.0125 mg/kg bw per day.
A screening value of 0.3 mg/L (rounded) can be derived following the standard equation:
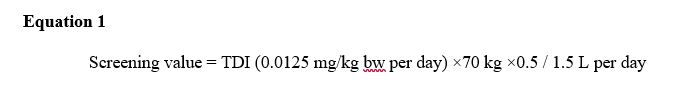
Equation 1 - Text Equivalent
Screening value: This equation calculates the sulfolane. The screening value for total for sulfolane is multiplied tolerable daily intake of 0.0125 mg/kg bw-day by the mean body weight estimated for a Canadian adult is 70 and by 50% based on usage patterning, environmental fate and physical-chemical properties then dividing it by 1.5 L the estimated daily volume of tap water consumed by an adult. Which equals a screening value of 0.3 mg/L.
Where:
- 0.0125 mg/kg bw/day is the TDI as derived above.
- 70 kg is taken as the mean body weight estimated for a Canadian adult.
- 0.5 is the allocation factor based on a conservative assumption that 50% of daily intake is attributable to drinking water. An allocation of 0.5 is recommended when there are only 2 potential significant sources of exposure (Krishnan and Carrier, 2013). Potential exposures of sulfolane are from drinking water and fruits and vegetables grown on sulfolane-contaminated sites.
- 1.5 L per day is the estimated daily volume of tap water by an adult.
International considerations
No regulatory limits for sulfolane in drinking water have been found in other countries. Sulfolane groundwater contamination in the city of North Pole, Alaska has prompted its inclusion in the United States National Toxicology Program where additional studies on sulfolane on its chronic effects as well as confirmation of its immunotoxicity (i.e., susceptibility to infection following reduced WBC counts) will be conducted.
Contact us
For more information, please contact us at hc.water-eau.sc@canada.ca.
References
Andersen, M., Jones, R.A., Kurlansik, L., Mehl, R.G. and Jenkins Jr., L.J. (1976). Sulfolane-induced convulsion in rodents. Chemical Pathology and Pharmacology, 15(3): 571-580.
CCME. (2006). Canadian environmental quality guidelines for sulfolane: water and soil. Scientific Supporting Document. Canadian Council of Ministers of the Environment, Canadian environmental quality guidelines, Winnipeg, Manitoba. Available at https://www.ccme.ca/files/Resources/supporting_scientific_documents/sulfolane_ssd_soil_water_1.1_e.pdf
Headley, J.V., Peru, K.M. and Dickson, L.C. (1999). Gas chromatographic-mass spectrometric determination of sulfolane in wetland vegetation to sour gas-contaminated groundwater. Journal of Chromatography, 859: 69-75.
HLS (Huntingdon Life Sciences). (2001). Sulfolane toxicity study by oral administration via the drinking water pathway to CD rats for 13 weeks. Huntingdon Life Sciences Ltd. Huntingdon, England.
Krishnan, K. and Carrier, R. (2013). The use of exposure source allocation factor in the risk assessment of drinking-water contaminants. J. Toxicol. Environ. Health, Part B: Crit. Rev., 16 (1): 39-51.
Lide, D.R. (Eds.) (1996). CRC Handbook of Chemistry and Physics, 77th Edition.
Ministry of Health and Welfare Japan. (1996) Sulfolane: 28 day repeat dose oral toxicity test. In: Toxicity testing reports of environmental chemicals. Tokyo, Japan. pp. 437−445. As cited in OECD (2004).
Ministry of Health and Welfare Japan. (1999). Sulfolane. In: Toxicity testing reports of environmental chemicals. Tokyo, Japan. pp. 473−481. As cited in OECD (2004).
NCEA. (2012). Provisional peer-reviewed toxicity values for Sulfolane (CASRN 126-33-0). Superfund Health Risk Technical Support Center, National Center for Environmental Assessment, Office of Research and Development, U.S. Environmental Protection Agency, Cincinnati, Ohio. Available at
http://dec.alaska.gov/spar/csp/sites/north-pole-refinery/docs/Sulfolane%20Final%20508.pdf
NTP. (2018a). Testing Status of Sulfolane 11054. National Toxicology Program, U.S. Department of Health and Human Services. https://ntp.niehs.nih.gov/testing/status/agents/ts-11054.html (last updated May 3rd, 2018).
NTP (2018b). Sulfolane Research Overview. National Toxicology Program, U.S. Department of Health and Human Services. https://ntp.niehs.nih.gov/results/areas/sulfolane/index.html (last updated May 9th, 2018).
TERA. (2014a). Report on the expert peer review of sulfolane reference doses for the Alaska Department of Environmental Conservation. Volume One. Toxicology Excellence for Risk Assessment. Expert panel: Andrew Maier, Susan Griffin, Richard Hertzberg, Michael Luster, Deborah Oudiz, Stephen Roberts. December 18, 2014. Available at https://www.tera.org/Peer/sulfolane/Sulfolane_peer_review_final_report.pdf
TERA. (2014b). Report on the expert peer review of sulfolane reference doses for the Alaska Department of Environmental Conservation. Volume Two. Toxicology Excellence for Risk Assessment. Expert panel: Andrew Maier, Susan Griffin, Richard Hertzberg, Michael Luster, Deborah Oudiz, Stephen Roberts. December 18, 2014. Available at https://www.tera.org/Peer/sulfolane/Volume%202%20Appendix.pdf
Zhu, Z.H., Sun, M.L., Li, Z.S., et al. (1987). An investigation of the maximum allowable concentration of sulfolane in surface water. J West China Univ Med Soc 18(4):376−380. Available at https://dec.alaska.gov/media/6254/sulfo-zhu-mac-transl.pdf.
