The Georgia Basin-Puget Sound Airshed Characterization Report 2014: chapter 12
12. Atmospheric Deposition and Ecological Effects
Patrick Shaw, Chris Eckley, Donna Strang, Bill Taylor, Sarah Hanna (Environment Canada) and Julian Aherne (Trent University).
Air pollution can have profound effects on both aquatic and terrestrial ecosystems. At low pollution levels, these effects could include plant tissue damage (Krzyzanowski et al., 2006), loss of sensitive plant species and consequent shifts in community structure (Emmett, 2007, Geiser and Neitlich, 2007, Bobbink et al., 2010) , and changes in soil chemistry (Tomlinson, 2003). At high deposition levels, particularly of acidifying pollutants, there may be irreparable changes to soil chemistry through loss of important base cations (Jeziorski et al., 2008), and significant changes in surface water chemistry (Lawrence, 2002). Species vary widely in their sensitivity, but none are completely resistant to the effects of air pollutants.
In this chapter, the ecological impacts of sulphur and nitrogen species and hazardous air pollutants, such as organochlorines and mercury resulting from atmospheric deposition, will be examined
12.1 Atmospheric Deposition Overview
Atmospheric pollutants can be deposited onto surfaces such as buildings, trees, open ground, or surface waters. This can occur as wet deposition, where the pollutants are scavenged from the air by rain, snow, or fog, or as dry deposition, where the pollutants deposit as gases or as particulate matter. With dry deposition, particles are removed from the atmosphere by gravity at a rate determined by their size and density. The rate at which gases and particles are deposited to the surface is expressed as their deposition velocity, a value which is modelled mathematically for various contaminants in the atmosphere under different meteorological conditions (Vet et al., 2004).
As two distinct processes and monitoring methods are involved, separate calculations must be made for wet and dry deposition fluxes, the sum of which is the total contaminant flux over a given area. Wet deposition is calculated by measured ion concentrations in rainfall samples collected over some period at precipitation chemistry monitoring sites. Dry deposition monitoring is not practical to undertake directly, so the flux is inferred from measurements of ambient air concentrations. The dry deposition flux is the product of measured daily air concentrations (µg/m3) and modelled daily average deposition velocities (m/s) for each contaminant. The total flux or contaminant load over a given area is expressed as mass per unit area per unit of time. When integrated over a full year, the contaminant load is typically expressed in units of kg/ha/yr for sulphur and nitrogen, or as mg/ha/yr for trace contaminants such as mercury. To represent acidification potential where the contribution of a number of chemical species may be involved, the loading is frequently expressed in terms of chemical charge, typically equivalents per unit area per year (eq/ha/yr).
12.1.1 Adverse Effects of Sulphur and Nitrogen Deposition
Acidic deposition occurs when sulphur dioxide (SO2) and oxides of nitrogen (NOx) react with water vapour in the atmosphere to produce sulphuric acid (H2SO4) and nitric acid (HNO3). Sulphur dioxide is emitted during fossil fuel (oil and coal) combustion and smelting of pyrite-rich ore. Nitrogen oxides are mainly released during fuel combustion. Geographically, most acid rain falls in the eastern half of Canada and the U.S., since the largest emissions sources of acidic air pollutants are located in industrial regions of eastern North America, and high-level winds carry these pollutants generally from west to east. Acidic deposition has resulted in the acidification of both soils and surface waters worldwide, leading to adverse effects on both terrestrial and aquatic organisms.
In addition to acidic deposition, excess nitrogen is a growing concern worldwide (Fenn et al., 2003, Galloway et al., 2003, Bleeker et al., 2011) and has been identified as an important issue in BC (Schindler et al., 2006). While nitrogen is an essential nutrient, nitrogen enrichment beyond ecosystem needs or assimilative capacity can lead through a continuum to nitrogen saturation (see Figure 12.1 for alpine systems), and, depending on the nitrogen species involved can also cause acidification (Aber et al., 1989, Stoddard, 1994). The outcomes may include eutrophication, soil and surface water acidification (Aber et al., 1989, Stoddard, 1994) and changes in aquatic and terrestrial community composition, commonly associated with shifts towards nitrophilous plant species (Bergström and Jansson, 2006). Evidence of changes in community species composition have been observed in the diatom assemblages and vegetation communities of the Colorado Front Range (Bowman et al., 2006) and lichen species in the United States Pacific Northwest (Fenn et al., 2003, Geiser et al., 2010) .
Figure 12.1. Continuum of effects of chronic excess nitrogen deposition to alpine regions. After Porter and Johnson (2007).

Description of Figure 12.1
Figure 12.1 is a schematic with an arrow on the left indicating increasing nitrogen load from bottom to top and text describing the effects of chronic nitrogen deposition to alpine regions located along the arrow according to the level of nitrogen loading. No units or numerical scale are given. At the bottom, in the region of lowest nitrogen loading, there is increased lake and stream nitrate. Next is change in phytoplankton communities, which is quickly followed by change in lichen communities. Above change in lichen communities there is change in soil and foliar chemistry followed by change in alpine plant species. The top two effects are highlighted in red and are episodic acidification followed by chronic acidification.
12.1.2 Sensitivity to Sulphur and Nitrogen Deposition
Water and soils that lack natural alkalinity have low buffering capacity, low resistance to changes in pH and are vulnerable to acidic deposition. Surface waters in southwest British Columbia have high sensitivity to acidification, due to a dominance of poorly weatherable bedrock (Wiens et al., 1987).The geology of coastal British Columbia is dominated by the Coastal Intrusive Complex (Stowell and McClelland, 2000), an extensive region of highly resistant granitic bedrock. Vancouver Island, in contrast, is part of the western North American “Wrangellia Terrane” (Greene et al., 2005), bearing somewhat more extensive carbonates and more weatherable basaltic rocks. The buffering ability of the coastal bedrock may be summarized in general weathering classes (Figure 12.2) following Nanus et al.(2009): Class 1− low to no buffering (e.g. granite, quartzite, gneiss); Class 2 - moderate buffering (e.g. diorite, granodiorite); Class 3 − high buffering (e.g. basalt, gabbro); Class 4 - very high buffering (e.g. undifferentiated metamorphic rocks, olivine); and, Class 5 - extremely high buffering (e.g. limestone, marble, basic intrusive rocks). Poorly weatherable class 1 and 2 bedrocks dominate the south coast, while class 3 and higher are common on Vancouver Island (Figure 12.2).
Effects of excess nitrogen will be most evident in high elevation environments where thin soils, sparse vegetation and a short growing season will contribute to leaching to local water bodies (Baron et al., 2000). Vegetation community structure will play a large part in determining the nitrogen assimilative capacity of a landscape. For example, watersheds dominated by nitrogen-fixing species, such as red alder, may have sufficient nitrogen without atmospheric deposition (Compton et al., 2003). Denitrification processes may function under some circumstances to mitigate the effects of excess nitrogen (Aber et al., 1989).
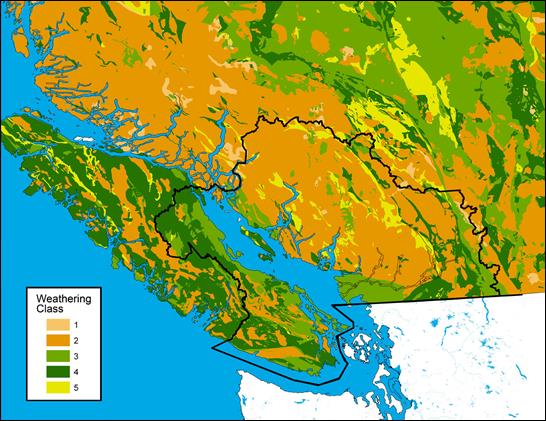
Figure 12.2. Map of southwestern BC showing bedrock weathering potential ranging from low buffering capacity (Class 1) to extremely high buffering capacity (Class 5). Assembled by Beverley Raymond, Environment Canada, based on the bedrock classification from Nanus et al. (2009) and basic bedrock geology from the BC Ministry of Mines and Petroleum Resources.
Description of Figure 12.2
Figure 12.2 is a map of southwestern British Columbia and northwestern Washington State. The Canadian portion of the Georgia Basin-Puget Sound airshed is outlined in black and the province is colored according to weathering class. There are five levels of weathering potential, ranging from low buffering capacity (Class 1) to extremely high buffering capacity (Class 5).
There are only a few very small Class 1 pockets within the Georgia Basin and these occur primarily along the eastern edge. Two very small Class 1 pockets occur in the vicinity of Whistler.
Class 2 weathering potential dominates much of the mainland part of the airshed north and east of the Fraser Valley. The Sunshine Coast between Howe Sound and Jervis Inlet is primarily weathering Class 2 as is most of the Coast Mountain area. Some smaller pockets occur on Vancouver Island and the Gulf Islands, notably on central Salt Spring and the Saanich Peninsula, as well as in a band running east from Port Renfrew along the San Juan River. Other areas of weathering Class 2 occur in the mountains around Cowichan Lake.
Class 3 weathering potential occurs throughout the lower Fraser Valley and along the Sunshine Coast north of Jervis Inlet. It also occurs along the east and south coasts of Vancouver Island, throughout most of the Gulf Islands, and in a patchy north-south band just east of Hope.
Class 4 weathering potential dominates the mountainous area to the east of Cultus Lake. It occurs in a patchy band to the east of Harrison Lake and again in the Hope area. Class 4 covers most of Texada Island and the mountainous areas to the west of Campbell River and Courtenay. It occurs patchily to the north of Port Renfrew and covers the Victoria and Malahat areas.
Class 5 weathering potential occurs only sporadically in a patchy band running north-by-northwest from Harrison Lake and in small patches around Howe Sound and Jervis Inlet.
12.2 Deposition Measurement Networks
Networks to measure the atmospheric deposition are present within the Georgia Basin airshed in both Washington State and British Columbia.
Historically, the province of British Columbia maintained a network of stations to measure atmospheric chemistry (the BC Precipitation Chemistry Monitoring Network - BCPCMN) for wet deposition. From 1983 to 2006 when the program was discontinued, up to 14 sites in southwestern British Columbia were monitored. Presently, only one active precipitation chemistry site remains in the province, located on Saturna Island in the southern Strait of Georgia. The current state of monitoring capability makes it nearly impossible to adequately evaluate current conditions, as well as changes and trends in fluxes of either acidifying of buffering substances in British Columbia. This shortcoming is discussed further in Chapter 15, “Summary of Key Findings and Implications”.
British Columbia’s sole precipitation chemistry monitoring site at Saturna Island is part of the Canadian Air and Precipitation Monitoring Network (CAPMoN), a national non-urban air quality monitoring network that measures the wet and dry deposition of acidifying chemicals, as well as ground-level ozone and some heavy metals. Daily rainwater samples are analyzed for hydride (acidity as pH), sulphate, nitrate, ammonium, chloride and base cations (such as calcium, magnesium, potassium and sodium) to estimate wet deposition, and daily filter pack (air) samples provide dry deposition. CAPMoN network stations are sited to ensure that the measurements are regionally representative and are not affected by nearby local sources of air pollution.
Deposition measurements in U.S. portion of the air basin are conducted through the National Atmospheric Deposition Program/National Trends Network (NADP/NTN). This is a cooperative effort amongst many different groups, including the State Agricultural Experiment Stations, U.S. Geological Survey, U.S. Department of Agriculture and other governmental and private entities. The purpose of the network is to collect data on the chemistry of precipitation for monitoring geographical and temporal long-term trends. Precipitation at each station is collected weekly and analyzed for hydride (acidity as pH), sulphate, nitrate, ammonium, chloride and base cations (such as calcium, magnesium, potassium and sodium). Dry deposition is estimated through the Clean Air Status Trends Network (CASTNET), which is considered the nation’s primary source for atmospheric data to estimate dry acidic deposition and to provide data on rural ozone levels. In the U.S., CASTNET is part of and complements the wet deposition database compiled by NADP. As in BC, the Pacific Northwest monitoring sites are sparse geographically. Within the U.S. portion of the Georgia Basin there is a NADP site at North Cascades National Park and a co-located NADP-CASTNET site to the south at Mt Rainier National Park.
12.3 Sulphur and Nitrogen Loading in Georgia Basin/Puget Sound
Until recently, the focus of research on sulphur and nitrogen loading has been in eastern Canada and the eastern U.S., where adverse environmental impacts have led to international agreements to reduce SO2 and NOx emissions. Only recently has this focus shifted to the west where concerns over the expansion of industry, transportation and urbanization have stimulated new research into the impacts of sulphur and nitrogen deposition. Much of this recent research has turned to understanding sources, developing depositional critical loads and monitoring ecosystem response to sulphur and nitrogen in the western half of the continent (Aherne and Shaw, 2010).
12.3.1 Deposition Modelling
Deposition estimates over large geographic regions make use of computer models which combine inventories of emission sources, observed or modelled meteorological conditions, and estimates of chemical dynamics in the atmosphere. One particular model, the Community Multiscale Air Quality Model (CMAQ: Byun and Schere, 2006) has found some valuable application in understanding depositional loadings in the Georgia Basin-Puget Sound.
Researchers at the University of British Columbia (UBC, 2007) conducted daily simulations over a geographic area (domain) covering much of SW BC and Washington State using CMAQ at a 4km x 4km resolution for much of 2004 to 2006, using the most current emission inventory from 2000. Hourly output was summarized and used to map total annual nitrogen and sulphur fluxes. Total sulphur deposition ranged from <1.0 to >30 kg/ha/yr, with the highest concentrations being found near marine shipping routes and major population centers (Figure 12.3). Particular hot-spots evident in northwest Washington State are related to local industrial activities, including an oil refinery at Cherry Point and an aluminum smelter at Ferndale. Most of the basin received less than 8 kg S ha/yr.
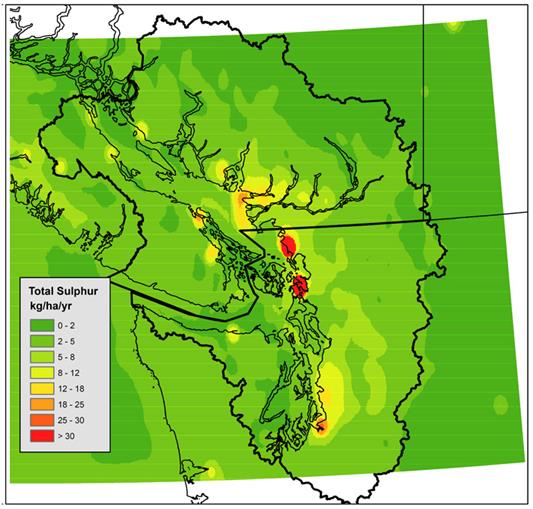
Figure 12.3 Modelled annual total sulphur deposition over the Georgia Basin/Puget Sound a 4 km by 4km grid resolution using a 2000 air emissions inventory. Map generated with CMAQdata from UBC (2007).
Description of Figure 12.3
Figure 12.3 is a map of southwestern British Columbia and northwestern Washington State. The Georgia Basin-Puget Sound airshed is outlined in black and most of airshed (with the exception of the very northern and southern extremities is colored by total sulphur deposition in kg/ha/yr. The increments are 0-2, 2-5, 5-8, 8-12, 12-18, 18-25, 25-30, and >30 kg/ha/yr.
Total sulphur deposition is less than 8 kg/ha/yr in most of the airshed. Pockets of deposition in the 8-18 kg/ha/yr range occur around Powel River, Campbell River, Courtenay, and near the south end of Buttle Lake in Strathcona Park. They also occur near Nanaimo, Duncan, Langdale, in the Strait of Georgia west of Metro Vancouver, along Burrard Inlet, around Port Angeles, and in the area from Everett to Tacoma. Pockets of deposition in the 18-25 kg/ha/yr range occur at Nanaimo, Vancouver, and in the area surrounding Tacoma. In the area immediately around Tacoma sulphur deposition is on the order of 25-30 kg/ha/yr. Two places have deposition over 30 kg/ha/yr and these are the area between Bellingham Bay and Birch Bay and the area around Anacortes covering Fidalgo Island and Padilla Bay.
Total annual nitrogen deposition ranged from <1 to >30 kg N/ha/yr for the 2005-2006 period (Figure 12.4), with most values below 12 kg N/ha/yr. Hot spots exceeding >30 kg N/ha/yr were indicated in the Fraser Valley, both in the south-western portion, associated with mixed farming activities, and in the central area where ammonia release from intensive poultry and livestock production drive very high deposition.
The outcome of the UBC work was largely corroborated in independent modelling on a coarser grid resolution for the Pacific Northwest states using CMAQ by Porter (2007). Annual deposition estimates were calculated from 10 years of modelling data (1990-1999). Particular hotspots for both nitrogen and sulphur deposition corresponded to major population centers (Vancouver, Seattle, Portland) and high-volume transportation corridors, but were less related to agricultural production perhaps because of the lower resolution.
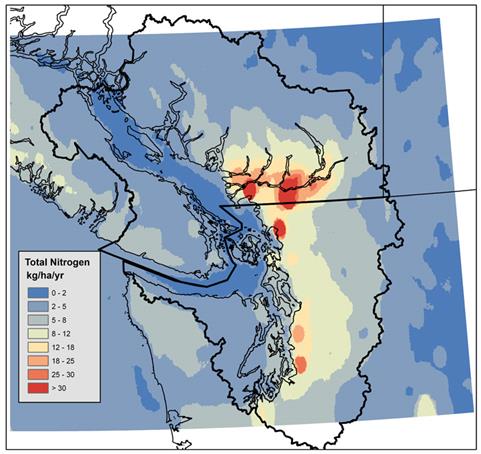
Figure 12.4. Modelled annual total nitrogen deposition over the Georgia Basin/Puget Sound for on a 4 km by 4km grid resolution using a 2000 air emissions inventory. Map generated with CMAQ data from UBC (2007).
Description of Figure 12-4
Figure 12.4 is a map of southwestern British Columbia and northwestern Washington State. The Georgia Basin-Puget Sound airshed is outlined in black and most of airshed (with the exception of the very northern and southern extremities is colored by total nitrogen deposition in kg/ha/yr. The increments are 0-2, 2-5, 5-8, 8-12, 12-18, 18-25, 25-30, and >30 kg/ha/yr.
Total nitrogen deposition is less than 2 kg/ha/yr over the Strait of Georgia and the Strait of Juan de Fuca. Vancouver Island, the Gulf Islands, the San Juan Islands, the Olympic Peninsula, the Cascades, and the Coast Mountains north of Howe Sound have total nitrogen deposition from 2-8 kg/ha/yr. The North Shore mountains, the area between Hope and Harrison Lake, and most of the area from the Cascades west to Puget Sound have deposition from 8-18 kg/ha/yr. The area around Olympia also has deposition from 8-18 kg/ha/yr. The area around Burrard Inlet and along the Fraser River from Burnaby to Agassiz (excluding Richmond) has nitrogen deposition on the order of 18-25 kg/ha/yr, as does the northern part of Seattle. Two areas with deposition on the order of 25-30 kg/ha/yr occur at Chilliwack and in Kent, Washington. Areas with total nitrogen deposition in excess of 30 kg/ha/yr occur in western Surrey, around Abbotsford, and around Bellingham.
12.3.2 Deposition Measurements in Georgia Basin and Puget Sound
During the period 2004-2006, there were three stations (CAPMoN × 1 and CASTNET × 2) in the Georgia Basin / Puget Sound monitoring wet and dry deposition: Saturna Island, Mt. Rainier National Park, and North Cascades National Park. Saturna Island had the highest annual total sulphur and total nitrogen deposition of the three sites for 2004-2006, while Mt. Rainier National Park has the lowest (Figure 12.5 ). Wet sulphur and nitrogen deposition is comparable among the three stations, but dry deposition estimates are significantly higher at the CAPMoN station. While this may be due to geographic and meteorological factors, such as elevation, rain days, it will also influenced by the method used by each network to estimate dry deposition (Schwede et al., 2011).
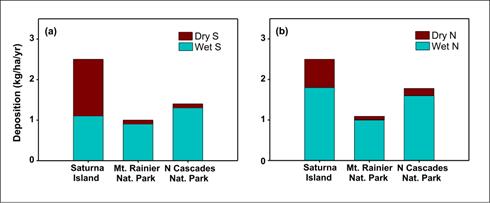
Figure 12.5. Mean annual (a) wet and dry sulphur deposition, and (b) wet and dry nitrogen deposition at three sites in the Pacific Northwest (kg/ha/yr) for 2004-2006 based on data from CAPMoN and CASTNET.
Description of Figure 12.5
Figure 12.5 is composed of two stacked bar charts. On the left there are bars showing dry and wet sulphur deposition in kg/ha/yr at Saturna Island, Mt. Rainier National Park, and North Cascades National Park. On the right there are bars showing dry and wet nitrogen deposition in kg/ha/yr at Saturna Island, Mt. Rainier National Park, and North Cascades National Park.
Wet sulphur deposition at Saturna Island is just over 1 kg/ha/yr, while dry sulphur deposition is approximately 1.5 kg/ha/yr. For Mt. Rainier National Park wet sulphur deposition is just under 1 kg/ha/yr, while dry sulphur deposition is approximately 0.1 kg/ha/yr. At North Cascades National Park wet sulphur deposition is approximately 1.2 kg/ha/yr and dry sulphur deposition is approximately 0.1 kg/ha/yr.
Wet nitrogen deposition at Saturna Island is approximately 1.8 kg/ha/yr, while dry nitrogen deposition is approximately 0.7 kg/ha/yr. For Mt. Rainier National Park wet nitrogen deposition is approximately 1 kg/ha/yr, while dry nitrogen deposition is approximately 0.1 kg/ha/yr. At North Cascades National Park wet nitrogen deposition is approximately 1.6 kg/ha/yr and dry nitrogen deposition is approximately 0.2 kg/ha/yr.
Compared to the period 1998-2000, all three sites experienced a decline in annual total (wet + dry) sulphur and nitrogen deposition during the 2004-2006 period. Annual total sulphur, decreased in the range of 27% to 48%, while annual total nitrogen decreases were more modest, ranging from 9% to 16% (R. Vet, pers. comm.).
At most CAPMoN sites in Canada, wet deposition predominates. One exception, however, is Saturna Island where the two processes contribute about equal amounts (see Figure 12.5). On a seasonal basis, Saturna Island shows higher fluxes of both wet and dry sulphur deposition in fall and winter. A seasonal pattern is less evident for nitrogen deposition (Vet et al., 2004).
Aherne et al. (2010) examined 18 years (1990-2007) of precipitation chemistry data from Saturna Island and compared it to three other CAPMoN sites in eastern Canada. They concluded that the Georgia Basin is less influenced by anthropogenic emissions of sulphur and nitrogen compared to eastern Canada, with Saturna Island showing significantly lower deposition of sulphate. Nonetheless, long term trends in sulphate, nitrate and pH for Saturna all showed improvement during the 18 years, mirroring the situation for eastern Canada, and suggesting that all regions have responded similarly to large scale emissions reductions under the Canada-U.S. Air Quality Agreement. Therefore, while coastal British Columbia does not have the high sulphate emissions typical of eastern Canada, its aquatic and forested ecosystems remain vulnerable to acid deposition because of the poor buffering properties of the soil and underlying geology (Wiens et al., 1987).
In the Puget Sound area, total sulphur and nitrogen deposition data are available from the monitoring site at Mt. Rainier National Park. Figure 12.6 and Figure 12.7 show the composition and annual trends for sulphur and nitrogen deposition respectively at the Mt. Rainier site (U.S. Environmental Protection Agency, 2010). For both sulphur and nitrogen, wet deposition is the dominant process at Mt. Rainier National Park. Data from NADP/NTN and CASTNET show decreasing trends in total sulphur and nitrogen during the period 1996-2008 in western Canada (MACTEC Engineering and Consulting, 2010). However, the trend is not as evident for National Parks sites on Mt. Rainier in the Puget Sound (Figure 12.6 and Figure 12.7).
For comparison with the Mount Rainier data, total sulphur deposition at the 17 monitoring stations in the western USA averaged between approximately 0.5 and 2 kg/ha/yr for the period of 1996 to 2008. For nitrogen the averaged total deposition was between approximately 1 and 4 kg/ha/yr for the same period (MACTEC Engineering and Consulting, 2010).
Figure 12.6. (a) Composition of total sulphur deposition by species for 2007 - 2009 at Mt Rainier National Park (b) Annual total sulphur deposition (kg/ha/yr) at Mt Rainier National Park (1996-2009). Only complete sample years are shown (U.S. Environmental Protection Agency, 2010).
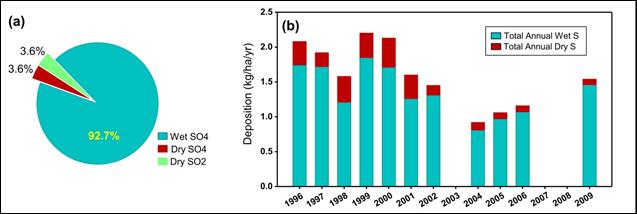
Description of Figure 12.6
Figure 12.6 is composed of two parts, on the left is a pie chart showing the composition of total sulphur deposition by species at Mt Rainier National Park and on the right is a stacked bar chart showing total annual wet and dry sulphur deposition by year in kg/ha/yr.
The pie chart on the left shows that 92.7% of total sulphur deposition is wet SO4, 3.6% is dry SO4, and 3.6% is dry SO2.
The bar chart on the right shows the following.
- In 1996 total wet sulphur deposition was approximately 1.75 kg/ha/yr and total dry sulphur deposition was approximately 0.3 kg/ha/yr.
- In 1997 total wet sulphur deposition was approximately 1.75 kg/ha/yr and total dry sulphur deposition was approximately 0.2 kg/ha/yr.
- In 1998 total wet sulphur deposition was approximately 1.25 kg/ha/yr and total dry sulphur deposition was approximately 0.3 kg/ha/yr.
- In 1999 total wet sulphur deposition was approximately 1.8 kg/ha/yr and total dry sulphur deposition was approximately 0.4 kg/ha/yr.
- In 2000 total wet sulphur deposition was approximately 1.75 kg/ha/yr and total dry sulphur deposition was approximately 0.5 kg/ha/yr.
- In 2001 total wet sulphur deposition was approximately 1.25 kg/ha/yr and total dry sulphur deposition was approximately 0.3 kg/ha/yr.
- In 2002 total wet sulphur deposition was approximately 1.3 kg/ha/yr and total dry sulphur deposition was approximately 0.15 kg/ha/yr.
- In 2004 total wet sulphur deposition was approximately 0.9 kg/ha/yr and total dry sulphur deposition was approximately 0.15 kg/ha/yr.
- In 2005 total wet sulphur deposition was approximately 1.0 kg/ha/yr and total dry sulphur deposition was approximately 0.1 kg/ha/yr.
- In 2006 total wet sulphur deposition was approximately 1.1 kg/ha/yr and total dry sulphur deposition was approximately 0.1 kg/ha/yr.
- In 2009 total wet sulphur deposition was approximately 1.4 kg/ha/yr and total dry sulphur deposition was approximately 0.1 kg/ha/yr.
Figure 12.7. (a) Composition of total nitrogen deposition by species for 2007 - 2009 at Mt Rainier National Park (b) Annual total nitrogen deposition (kg/ha/yr) at Mt Rainier National Park (1996-2009). Only complete sample years are shown (U.S. Environmental Protection Agency, 2010).
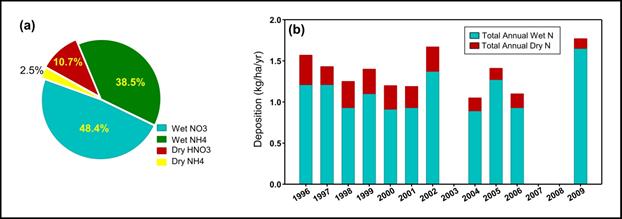
Description of Figure 12.7
Figure 12.7 is composed of two parts, on the left is a pie chart showing the composition of total nitrogen deposition by species and on the right is a stacked bar chart showing total annual wet and dry nitrogen deposition by year in kg/ha/yr.
The pie chart on the left shows that 48.4% of total nitrogen deposition is wet NO3, 38.5% is wet NH4, 10.7% is dry HNO3, and 2.5% is dry NH4.
The bar chart on the right shows the following.
- In 1996 total wet nitrogen deposition was approximately 1.2 kg/ha/yr and total dry nitrogen deposition was approximately 0.4 kg/ha/yr.
- In 1997 total wet nitrogen deposition was approximately 1.2 kg/ha/yr and total dry nitrogen deposition was approximately 0.2 kg/ha/yr.
- In 1998 total wet nitrogen deposition was approximately 0.9 kg/ha/yr and total dry nitrogen deposition was approximately 0.4 kg/ha/yr.
- In 1999 total wet nitrogen deposition was approximately 1.15 kg/ha/yr and total dry nitrogen deposition was approximately 0.4 kg/ha/yr.
- In 2000 total wet nitrogen deposition was approximately 0.9 kg/ha/yr and total dry nitrogen deposition was approximately 0.4 kg/ha/yr.
- In 2001 total wet nitrogen deposition was approximately 0.9 kg/ha/yr and total dry nitrogen deposition was approximately 0.3 kg/ha/yr.
- In 2002 total wet nitrogen deposition was approximately 1.3 kg/ha/yr and total dry nitrogen deposition was approximately 0.4 kg/ha/yr.
- In 2004 total wet nitrogen deposition was approximately 0.9 kg/ha/yr and total dry nitrogen deposition was approximately 0.2 kg/ha/yr.
- In 2005 total wet nitrogen deposition was approximately 1.25 kg/ha/yr and total dry nitrogen deposition was approximately 0.2 kg/ha/yr.
- In 2006 total wet nitrogen deposition was approximately 1.0 kg/ha/yr and total dry nitrogen deposition was approximately 0.2 kg/ha/yr.
- In 2009 total wet nitrogen deposition was approximately 1.6 kg/ha/yr and total dry nitrogen deposition was approximately 0.15 kg/ha/yr.
In addition to regular long term deposition monitoring, special studies were conducted from time to time that provided data over shorter time-frames. From 1999 to 2002, sampling was conducted at a number of sites by Environment Canada. Intensive nitrogen and sulphur measurements were undertaken through 2002 at three urban NAPS sites: Kitsilano, Rocky Point Park in Vancouver, and at the Topaz Park site in Victoria, and the rural CAPMoN site on Saturna Island. Another study involved intensive sampling between May 1999 and April 2001 at Chilliwack in the Lower Fraser Valley and at Cowichan on Vancouver Island (Hay et al., 2006). Total annual (wet and dry) sulphur deposition at four sites in the Georgia Basin varied between 2.8 at Victoria and 5.5 kg/ha/yr at Kitsilano during the study period (Figure 12.8). Total nitrogen (wet and dry) deposition among the same four sites ranged from 2.6 at Saturna Island to 8.2 kg/ha/yr at Kitsilano for the same period. However, at Chilliwack, the total nitrogen load increased to 30 kg/ha/yr owing to significant contributions of ammonia from nearby agricultural activities (Hay et al., 2006).
Figure 12.8 (a) Total annual wet and dry sulphur deposition at four sites in the Georgia Basin for 2002; (b) Total annual wet and dry nitrogen deposition at six sites in the Georgia Basin based on a compilation of available air and precipitation concentration data from monitoring networks and special studies conducted between 1999-2002 (Hay et al., 2006).
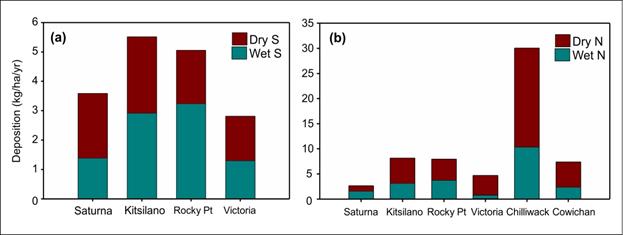
Description of Figure 12.8
Figure 12.8 is composed of two panels. In panel A on the left there is a stacked bar chart showing wet and dry sulphur deposition in kg/ha/yr for Saturna, Kitsilano, Rocky Point, and Victoria. In panel B on the right there is a stacked bar chart showing wet and dry nitrogen deposition in kg/ha/yr for Saturna, Kitsilano, Rocky Point, Victoria, Chilliwack, and Cowichan.
For Saturna total wet sulphur deposition was approximately 1.4 kg/ha/yr and total dry sulphur deposition was 2.1 kg/ha/yr. For Kitsilano total wet sulphur deposition was approximately 3.0 kg/ha/yr and total dry sulphur deposition was 2.5 kg/ha/yr. For Rocky Point total wet sulphur deposition was approximately 3.1 kg/ha/yr and total dry sulphur deposition was 1.9 kg/ha/yr. For Victoria total wet sulphur deposition was approximately 1.4 kg/ha/yr and total dry sulphur deposition was 1.5 kg/ha/yr.
For Saturna total wet nitrogen deposition was approximately 1.5 kg/ha/yr and total dry nitrogen deposition was 1.5 kg/ha/yr. For Kitsilano total wet nitrogen deposition was approximately 3.0 kg/ha/yr and total dry nitrogen deposition was 5.0 kg/ha/yr. For Rocky Point total wet nitrogen deposition was approximately 3.1 kg/ha/yr and total dry nitrogen deposition was 4.0 kg/ha/yr. For Victoria total wet nitrogen deposition was approximately 1.0 kg/ha/yr and total dry nitrogen deposition was 4.0 kg/ha/yr. For Chilliwack total wet nitrogen deposition was approximately 10 kg/ha/yr and total dry nitrogen deposition was 20 kg/ha/yr. For Cowichan total wet nitrogen deposition was approximately 3.0 kg/ha/yr and total dry nitrogen deposition was 5.0 kg/ha/yr.
Long-term precipitation chemistry measurements have been conducted in the UBC research forest near Maple Ridge, east of Vancouver (Feller, 2010). Bulk rainfall samples (collected in a permanently open sampler) have been collected on a weekly to biweekly basis from the late 1970s and analyzed for a range of ionic constituents. Bulk nitrogen deposition ranged from 4 to10 kg/ha/yr from 1980 to 2010. Bulk sulphate declined from a high of greater than 40 kg/ha/yr in the early 1980s to a measured average of about 10 kg/ha/yr in recent years. Not unexpectedly, rainfall conductivity has also declined while pH has risen over the same period.
12.4 Effects of Nitrogen & Sulphur Deposition on Georgia Basin Ecosystems
12.4.1 Acidification
Acidification of soils and forested ecosystems has gained much attention since the 1970s and 1980s. Impacts on forest health have been established, such as the depletion of base cations (calcium, magnesium and potassium) from soil which are essential nutrients for tree growth and vitality. The loss of base-cations also lessens the capacity of the soil to neutralize incoming acids thus increasing the acidity of water entering lakes and streams (Houle, 2004). Over time, there may be watershed-wide base cation depletion, with loss of key aquatic species (Jeziorski et al., 2008). For surface water, the buffering ability is expressed as “Acid Neutralizing Capacity” or ANC, and is typically calculated as the sum of base cations less the sum of acid anions ([Ca++Mg++K+] - [SO42-+NO3-]). Low ANC (< 50 µEq/L) is typical or highly sensitive waters.
In Canada, lakes and streams are considered acidified if the pH is below 6, a level at which negative biological impacts are expected to occur (Jeffries, 1997, Holt et al., 2003). Acidic surface waters are related to high concentrations of acid anions (e.g., sulphate: SO42-, nitrate: NO3-) and depleted base cation concentrations (calcium: Ca2+, magnesium: Mg2+, sodium: Na+, potassium: K+) resulting in low buffering and ANC. At low pH, the solubility of metals (e.g. Al3+) toxic to fish and other aquatic species may increase (Baker & Schofield, 1982). Although natural acidification is observed in areas with high concentrations of organic acids associated with dissolved organic carbon (DOC), if sulphur and nitrogen deposition persist, surface water acidification may continue, especially in lakes and streams surrounded by shallow, acid sensitive soils with depleted or no ANC.
12.4.2 Nitrogen Enrichment
Acidification due to excess reactive nitrogen will occur only at very high deposition fluxes, and other effects will be evident at lower rates (Henrys et al., 2011). In the U.S. Pacific Northwest, the National Park Service and the U.S. Forest Service are undertaking a program to develop critical loads based on eutrophication because elevated nitrogen may be causing unwanted effects at much lower deposition than is required for acidification (Baron, 2006). At this point, effects on lichens have been established (Geiser and Neitlich, 2007). A recent report completed for the National Park Service (E&S Environmental Chemistry, Inc., 2011) ranked Olympic, North Cascades and Mount Rainier National Parks at high risk from nitrogen enrichment. Work in Washington State (Fenn et al., 2007; Geiser and Neitlich, 2007; Geiser et al., 2010) has shown that this to be the case for lichens, and it may be true for vascular plants as well, particularly for alpine and subalpine species. Eutrophication may also be an issue in N-limited high elevation lakes, and studies in Washington State were underway in 2011 to investigate the implications of atmospheric nitrogen deposition (A. Paulson, pers. comm.).
12.5 Critical Loads
The impact of pollutant deposition is commonly assessed using the concept of an effects-based critical load (CL). A critical load is defined as ”the quantitative estimate of an exposure level below which significant harmful effects on specified sensitive elements in an ecosystem are not believed to occur based on present knowledge” (Nilsson et al., 1988).
Critical loads can be developed for any receptor, either physical or biological, with a suitable quantifiable response indicator. For example, aquatic critical loads are typically set to reflect the capacity of the water body to buffer incoming acidifying deposition and maintain ANC protective of biological communities (Henriksen and Posch, 2001). Soil critical loads are established to maintain ratios of base cations to aluminum to support healthy plant growth (Mongeon et al., 2010), or to maintain base cation levels (Nasr et al., 2010). Some critical loads for nitrogen deposition protect against loss of biodiversity (Geiser and Neitlich, 2007, Posch et al., 2011) or maintain aquatic phytoplankton community structure (Baron, 2006). When total (wet + dry) acidic (nitrogen and sulphur) deposition exceeds the critical load, the difference is defined as an exceedence, indicating potentially negative impacts on the specified ecosystem (Moran et al., 2008), or the N assimilative capacity of the system, in the case of eutrophication.
Aquatic and terrestrial critical loads are typically calculated using steady-state mass balance models or water chemistry models (Henriksen and Posch, 2001) and are often expressed in terms of charge loading, such as equivalents/ha/yr, since acidifying deposition can result from a number of possible chemical species. The conversions between the common units for key chemical species are presented in Table 12.1.
| Chemical Species | kg/ha/yr | eq/ha/yr | meq/m2/yr |
|---|---|---|---|
| SO42− |
1.0
|
20.8
|
2.08
|
| S |
1.0
|
62.4
|
6.24
|
| NO3− |
1.0
|
16.1
|
1.61
|
| NH4+ |
1.0
|
55.4
|
5.54
|
| N |
1.0
|
71.4
|
7.14
|
Description of Table 12.1
Table 12.1 gives the relationship (for five chemical species) between three different units used to express acidifying atmospheric deposition.
The first row of the table contains the headers “Chemical Species”, “kg/ha/yr”, “eq/ha/yr”, and “meq/m2/yr”. The first column of the table lists the chemical species for which the unit conversions are given. These are as follows:
- SO42−
- S
- NO3−
- NH4+
- N
The second column shows a value of 1.0 (kg/ha/yr) for all species. The third column gives the equivalent value in eq/ha/yr. The fourth column gives the equivalent value in meq/m2/yr.
12.5.1 Critical Loads for Soils
A number of factors affect the critical load; the key among them is the presence of weatherable bedrock in the watershed to supply buffering cations. Nasr et al. (2010) used the steady-state mass balance approach to track gains and losses in soil base cations in response to acidic deposition, soil weathering and land use practices across the Georgia Basin. Several data layers were synthesized in the study including the underlying geology, biogeoclimatic zones, elevation, soil attributes, temperature and precipitation. Modelled annual average total (wet and dry) sulphur and nitrogen deposition were obtained from CMAQ (see Figure 12.3 and Figure 12.4). In addition, geospatially interpolated nitrogen and sulphur and cation (Ca2+, Mg2+ and K+) wet deposition values were extrapolated from precipitation chemistry measurements from the BC Precipitation Chemistry Network now archived in the NatChem database.
Critical loads of acidity for soils based on a zero base cation depletion criterion were mapped for upland soils of the Georgia Basin (Figure 12.9), in addition to exceedences by total sulphur and nitrogen deposition (Figure 12.10). Land areas with critical load exceedences ranged from 13% of the Georgia Basin under NatChem interpolated wet deposition, and up to 32% under CMAQ modelled total (wet and dry) deposition. The highest exceedences were found in the northern uplands of the Lower Fraser Valley, where the soils are thin with low buffering capacity.
Mongeon et al. (2010) also assessed the potential impacts of sulphur and nitrogen deposition on forest soils in the Georgia Basin using the steady state mass balance approach. Site-specific critical loads were estimated for 19 forest plots using base cation weathering rates derived from the PROFILE model (Warfvinge and Sverdrup, 1992) in combination with site measured soil mineralogy. The critical chemical criterion associated with ecosystem protection was based on a base cation to aluminum ratio of ten to preserve soil base saturation. Soil aluminum concentrations were modelled using regional default and site-specific estimates of aluminum dissolution. Modelled CMAQ deposition during 2005-2006 exceeded critical load values at five to nine of the study sites (based on default and site-specific aluminum dissolution). While high elevation sites have acid-sensitive soils with poor weathering rates, critical loads were most often exceeded at sites near the Vancouver area due to the higher modelled acidic deposition. The results are similar to other acid sensitive regions in Canada.
Figure 12.9 Critical loads of acidity for soils (eq/ha/yr) in the Georgia Basin based on a zero base cation depletion criterion (Nasr et al., 2010).
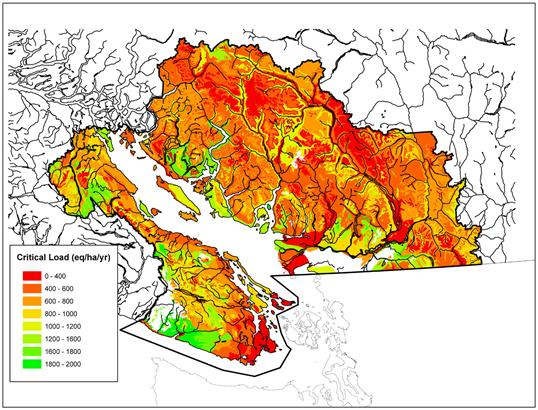
Description of Figure 12.9
Figure 12.9 is a map of southwestern British Columbia and northwestern Washington State. The Canadian portion of the Georgia Basin-Puget Sound airshed is outlined in black and is colored according to critical loads of acidity for soils in eq/ha/yr. The color scale is divided into 0-400, 400-600, 600-800, 800-1000, 1000-1200, 1200-1600, 1600-1800, and 1800-2000 eq/ha/yr.
Critical loads of 0-400 eq/ha/yr occur on southern Vancouver Island from the Saanich Peninsula south to Victoria and west to Sooke. They also occur around Mill Bay, on Mayne, Saturna, and Pender Islands, and on the south shore of Port San Juan at Port Renfrew. Smaller patches occur north of Cowichan Lake, in the mountains between Horne Lake and Comox Lake, and in Strathcona Park. On the mainland they occur in throughout Ladner and Delta, and throughout Burnaby and Coquitlam along the south shore of Burrard Inlet. The areas around Pitt and Harrison Lakes also have critical loads of 0-400 eq/ha/yr. The area to the east and to the south of Joffre Lakes as far south as Harrison Lake and the mountainous areas around the upper reaches of the Squamish River also have critical loads of 0-400 eq/ha/yr. A small patch also occurs along the Sunshine Coast from Powell River to Lund.
Critical loads of 400-800 eq/ha/yr occur along the length of the east coast of Vancouver Island from Campbell River south to Mill Bay, along the southwest coast from Sooke to Jordan River, and northwest from Port San Juan to Nitinat Lake. They also occur around Cowichan Lake and in Strathcona Park. Richmond (Lulu Island) has a critical load of 400-600 eq/ha/yr. In general the river drainages of the Coast Mountains have critical loads in the 400-1200 eq/ha/yr range, with the lower critical loads occurring at higher elevation and higher critical loads occurring along the river courses.
Critical loads of 1200 - 2000 eq/ha/yr occur along the southwest coast of Vancouver Island in the area bounded by Jordan River and San Juan River. They also occur just inland of the coast between the San Juan River and Nitinat Lake. The area between Cowichan Lake and Horne Lake also has large patches with critical loads of 1200 - 2000 eq/ha/yr, as does the area between Comox Lake and Buttle Lake. These critical loads also occur along the north arm of the Fraser River out to Point Grey, along the Chilliwack River, and around Alouette Lake. The Islands of Howe Sound and the Sunshine Coast from Langdale to Earl’s Cove are also have critical loads of 1200 - 2000 eq/ha/yr, as does much of the shore of Jervis Inlet and the inland area south of Powell Lake.
Figure 12.10. Exceedences (eq/ha/yr) of soil critical loads in the Georgia Basin based on zero base cation depletion criterion (Nasr et al., 2010) based on mean annual total sulphur deposition 2004-2006 from CMAQ. Positive values indicate regions where deposition is greater than the critical load, indicating potential negative impacts.
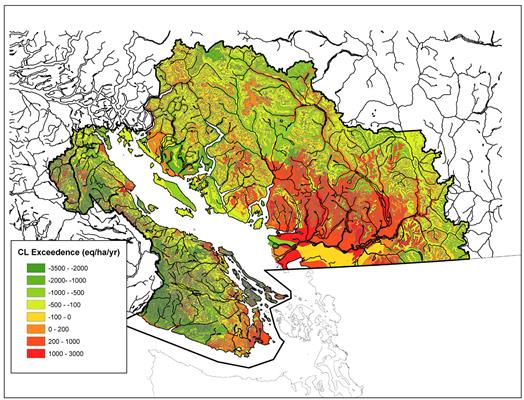
Description of Figure 12.10
Figure 12.10 is a map of southwestern British Columbia and northwestern Washington State. The Canadian portion of the Georgia Basin-Puget Sound airshed is outlined in black and is colored according to exceedances of soil critical loads in eq/ha/yr. The color scale is divided into -3500 to -2000, -20000 to -1000, -1000 to -500, -500 to -100, -100 to 0, 0 to 200, 200 to 1000, and 1000 to 3000 eq/ha/yr.
Exceedances in the range of 1000 to 3000 eq/ha/yr occur in Ladner and Delta, throughout Burnaby and Coquitlam along the south shore of Burrard Inlet, and along the drainages of Coquitlam, Pitt, and Harrison Lakes. They also occur south of the Fraser River between Abbotsford and Chilliwack and around Courtenay on Vancouver Island.
Exceedances in the 0 to 1000 eq/ha/yr range occur over Lulu Island, at White Rock, and throughout almost all of the northern uplands of the Lower Fraser Valley. They also occur in the mountainous areas between Howe Sound and Sechelt Inlet. They occur on the southern tip of Vancouver Island from the Saanich Peninsula, throughout the Victoria area, and as far west as Jordan River. Small areas around Nanaimo, Port Renfrew, and Campbell River also have exceedances in the 0 to 1000 eq/ha/yr range. The remainder of the airshed has deposition which is less than the critical load.
12.5.2 Critical Loads in Freshwater Lakes
In October 2008, Strang et al. (2010) conducted a survey of 72 high elevation lakes in the Georgia Basin to assess their acid sensitivity and to determine critical loads and exceedences of acidity. The lakes were mostly at higher elevations and known to have low buffering capacity and poor weathering rates (based on geological maps). Annual average total (wet and dry) sulphur and nitrogen deposition were modelled using CMAQ during the period 2004-2006 (Figure 12.3 and Figure 12.4).
The pH for the 72 lakes ranged from 5.4 to 7.3; almost 20% of the lakes had pH values less than 6.0. Acid neutralizing capacity varied greatly, with 21% of the lakes having less than 20 µeq/L, below a level which is protective of fish species. Based on ANC values, 34 (47%) of the lakes were classified as sensitive to acid deposition, 6 (8%) were insensitive, and 32 (44%) were moderately sensitive.
The median critical load based on the steady-state water chemistry model for the 72 lakes was approximately 70 meq/m2/yr, a value which is similar to acid-sensitive lakes in eastern Canada. Thirteen of the lakes (18%) received modelled sulphur deposition in excess of their critical load, suggesting that the lake catchments are unable to buffer acidic inputs, a probable consequence of the dominance of poorly weatherable bedrock and sparse vegetative land cover.
There has been some recent work on establishing nitrogen deposition critical loads for alpine lakes in the western U.S. (Saros et al., 2011) which follows on work conducted in Rocky Mountains National Park (Baron, 2006). Using paleolimnological analysis of diatom communities and shifts in species abundances in sediment cores, they determined a critical load of 1.4 kg N ha/yr to trigger nutrient enrichment effects. A similar U.S. Geological Survey study underway in the North Cascades National Park in Washington State (Paulson, 2011, pers. comm.) is also indicating a low threshold for nitrogen deposition effects.
12.5.3 Critical Load Estimates Using Lichen Communities
Geiser et al. (2010) studied the impact of nitrogen deposition on lichen communities in the western Washington and Oregon area. They used measured wet deposition levels from the NADP/NTN network, ambient airborne concentrations from the IMPROVE network, and modelled total deposition from CMAQ to develop critical loads based on lichen community composition and modelled precipitation. Critical loads were defined as a level nitrogen deposition associated with a measurable decline in the abundance of oligotrophic lichens (those adapted to low nutrient levels) and an increase in eutrophic lichens (those adapted to nutrient-rich environments), indicating more polluted conditions. Lichen community-based critical loads for total nitrogen deposition varied across north-western Oregon and western Washington from 3 to 9 kg/ha/yr with higher values occurring in areas with greater precipitation (Figure 12.11 reproduced from Geiser et al.(2010)).
Figure 12.11. Map of lichen community based critical loads for nitrogen in western Oregon and Washington. White shading indicates exceedences from 1994 to 2002 (from Geiser et al., 2010).

Description of Figure 12.11
Figure 12.11 is a relief map of western Washington State and northwestern Oregon State. The map is colored by lichen community based critical loads for nitrogen in kg/ha/yr. The color scale is divided into <2.7, 2.8 to 3.8, 3.9 to 4.8, 4.9 to 6.3, 6.4 to 7.2, 7.3 to 7.7, 7.8 to 8.6, 8.7 to 9.3, and >9.3 kg/ha/yr. Areas with critical load exceedances are shaded with white.
The only area with critical loads of <2.7 kg/ha/yr is a small region around Sequim on the Olympic Peninsula. Areas with critical loads in the range of 2.8 to 3.8 kg/ha/yr are the eastern shore of Puget Sound including Whidbey Island, the lowland region south and west of Tacoma, and the northeastern tip of the Olympic Peninsula from Port Angeles to Dabob Bay.
Areas with critical loads greater than 7.8 occur in the mountains of the central Olympic Peninsula, in the Casacades to the east of Puget Sound, and in the mountainous area just inland from the coast and south of the Columbia River. The remainder of the airshed has critical loads between 3.9 and 7.2 kg/ha/yr.
Crtical load exceedances occur in a corridor following Insterstate 5 and also extending out along the Columbia River. The I5 exceedance corridor stretches from the eastern shore of Puget Sound to the foothills of the Cascades and includes Whidbey Island and Bremmerton. It includes Olympia, Longview, and Portland/Vancouver and it is approximately 20 to 40km wide along its entire length.
Geiser et al. (2010) noted in particular that most of the study region received nitrogen deposition of only 1-2 kg/ha/yr in excess of pre-anthropogenic levels, concluding that there is still an opportunity to set regulations to limit nitrogen deposition to a level that will maintain intact natural lichen ecosystems. Figure 12.11 indicates areas where critical loads were exceeded from 1994 to 2002.
Raymond et al. (2010) surveyed tissue chemistry in 3 lichen and 1 moss species from a total of 57 sites in the Georgia Basin to map nitrogen and sulphur levels and compare the values with modelled deposition estimates. One lichen species, Platismatia glauca, was found at 56 of 57 sites with tissue nitrogen ranging from 0.22 to 0.91% and sulphur ranging from 0.05 to 0.22%. As shown in Figure 12.12, the pattern of tissue nitrogen in this lichen reflected closely the total nitrogen deposition fields from CMAQ modelling, with the highest levels at low elevation near major population centres and agricultural sources. The pattern for sulphur content was similarly close. As well, there was a high correlation between tissue N and S between the four species, showing that all responded similarly and any could be used in mapping regional deposition.
Figure 12.12 Nitrogen content (%) in the tissue of Platismatia glauca compared to CMAQ-modelled mean annual total nitrogen deposition (kg/ha/yr) during the period 2004-2006 (from Raymond et al., 2010).
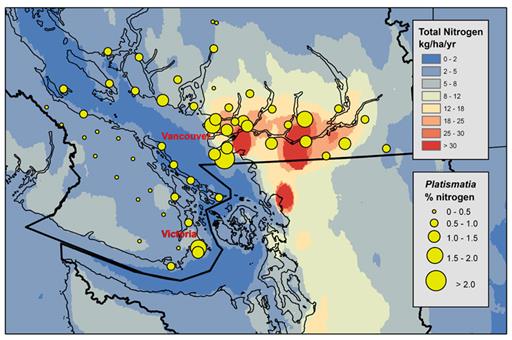
Description of Figure 12.12
Figure 12.12 is a map of southwestern British Columbia and northwestern Washington State. It extends from the south tip of Quadra Island in the north to Lynnwood in the south and from Barkley Sound in the west to approximately Princeton BC in the east. The Georgia Basin-Puget Sound airshed is outlined in black, although not all of the airshed is included in the map area. The map is colored by CMAQ-modelled mean annual total nitrogen deposition in kg/ha/yr which is also shown in Figure 12.4. The increments for the color mapping are 0-2, 2-5, 5-8, 8-12, 12-18, 18-25, 25-30, and >30 kg/ha/yr. Overlaid on the map are circles sized to indicate the percent nitrogen content in the tissue of Platismatia glauca. The nitrogen content increments are 0-0.5, 0.5-1.0, 1.0-1.5, 1.5-2.0, and > 2.0%.
As described in Figure 12.4, total nitrogen deposition is less than 2 kg/ha/yr over the Strait of Georgia and the Strait of Juan de Fuca. Vancouver Island, the Gulf Islands, the San Juan Islands, the Olympic Peninsula, the Cascades, and the Coast Mountains north of Howe Sound have total nitrogen deposition from 2-8 kg/ha/yr. The North Shore mountains, the area between Hope and Harrison Lake, and most of the area from the cascades west to Puget Sound have deposition from 8-18 kg/ha/yr. The area around Olympia also has deposition from 8-18 kg/ha/yr. The area around Burrard Inlet and along the Fraser River from Burnaby to Agassiz (excluding Richmond) has nitrogen deposition on the order of 18-25 kg/ha/yr, as does northern part of Seattle. Two areas with deposition on the order of 25-30 kg/ha/yr occur at Chilliwack and in Kent, Washington. Areas with total nitrogen deposition in excess of 30 kg/ha/yr occur in western Surrey, around Abbotsford, and around Bellingham.
On Vancouver Island Platismatia glauca sampling sites and the percent nitrogen content in the lichen tissue are as follows.
- Sooke, 0.5-1.0%
- Victoria, two sites with 1.0-1.5% and 1.5-2.0%
- central Saanich Peninsula 0.5-1.0%
- Sansum Narrows, 0.5-1.0%, Saturna Island, 0.5-1.0%
- south Galiano Island, 0.5-1.0%
- north Galiano Island, 0.5-1.0%
- Gabriola Island, 0.5-1.0%
- Qualicum Beach, 0.5-1.0%
- Hornby Island, 0.5-1.0%
- and Texada Island, 0.5-1.0%.
There were also thirteen additional sampling sites on Vancouver Island, they were inland from the coast and stretched between Langford and Fanny Bay, and all had Platismatia glaucapercent nitrogen content of 0-0.5%.
Sampling sites at Saltery Bay, Skookumchuk Narrows, Halfmoon Bay, Port Mellon, Bowen Island and Squamish had Platismatia glauca percent nitrogen content of 0.5-1.0%. A sampling site at Sechelt had Platismatia glauca percent nitrogen content of 1.0-1.5%. Two sites between Squamish and Whistler and one along the east side of Howe Sound had Platismatia glaucapercent nitrogen content of 0-0.5%.
In the lower mainland Platismatia glauca sampling sites and the percent nitrogen content in the lichen tissue are as follows.
- Vancouver, two sites with 1.0-1.5% and 1.5-2.0%
- Burnaby, 1.0-1.5%
- Coquitlam, one site with 1.0-1.5% and two with 1.5-2.0%
- Indian Arm two sites with 1.0-1.5%
- Pitt Lake, 1.0-1.5%
- Abbotsford, 1.5-2.0%
- Mission, 0.5-1.0%
- Stave Lake, 1.5-2.0%
- Chilliwack, 1.0-1.5%
- Cultus Lake, 0.5-1.0%
- Chilliwack south, 1.0-1.5%
- Harrison Hot Springs, 0.5-1.0%
- Agassiz, 0.5-1.0%
- Chilliwack Lake, 0.5-1.0%
- Ladner and Delta, two sites at 1.0-1.5% and one site at >2.0%
12.6 Hazardous Air Pollutants (HAPs)
Toxic or hazardous air pollutants (HAPs) form a broad category of chemical species that are associated with serious health risks and ecological impacts. Several of these contaminants are readily deposited onto soils or surface waters, where they can be taken up by plants and ingested by animals and can eventually be magnified up through the food chain. In this section, the deposition rates and trends for several HAPs measured in the Georgia Basin/Puget Sound airshed will be examined. These include mercury (Hg), polycyclic aromatic hydrocarbons (PAHs), polychlorinated biphenyls (PCBs), and polybrominated diphenyl ethers (PBDEs).
Polycyclic aromatic hydrocarbons are formed during the combustion of either fossil fuels or biomass. They are of concern because several PAHs have been identified as health hazards. Polychlorinated biphenyls and PBDEs belong to a class of compounds known as persistent organic pollutants (POPs). Polychlorinated biphenyls are synthetic compounds that were used in a variety of commercial and industrial applications and were banned in 2001 under the Stockholm Convention on Persistent Organic Pollutants. Polybrominated diphenyl ethers are used as flame retardants in a wide array of materials and have more recently come under some scrutiny and regulation. The physical and chemical properties of these and related compounds allow their transport in the environment, bioaccumulation in food chains, and initiation of a variety of short and long term toxic responses (Noël et al., 2009).
12.6.1 Mercury in Georgia Basin/Puget Sound
Mercury is released to the atmosphere primarily during coal combustion and the incineration of waste. Once emitted it can have a long atmospheric lifetime and be transported around the globe. Following deposition, mercury can be transformed to methyl mercury, which has increased toxicity and is the dominant form of mercury that accumulates in aquatic organisms and the humans and wildlife that consume them. Mercury methylation is predominantly a microbial process that occurs in aNOxic environments such as wetlands, sediments, and saturated soils and has been linked to the activity of sulphur reducing bacteria.
In Canada, atmospheric total gaseous mercury (TGM) concentrations and total mercury concentrations in precipitation are measured as part of CAPMoN to provide a better understanding of mercury trends and processes in the environment. Total gaseous mercury is measured in-situ at five minute intervals and quality controlled through Environment Canada’s Research Data Management and Quality Control System (RDMQ) before hourly data are archived. The wet deposition samples are collected weekly and are shipped to a central laboratory in the U.S. that analyzes all precipitation mercury samples in the U.S. and Canada that are part of the NADP/NTN’s Mercury Deposition Network (MDN). The objective of the MDN is to monitor the spatial and temporal trends of mercury in precipitation throughout North America, which can be used to identify inputs to lakes and streams.
Mercury Wet Deposition
Spatial Trends
As part of the MDN, total mercury concentrations in precipitation have been measured at four locations within the Puget Sound/Georgia Basin:
- WA18 - on National Oceanographic and Atmospheric Administration (NOAA) property near the urban area of Seattle along Puget Sound.
- WA03 - at the Makah Fish Hatchery in a remote region on the north-western tip of Washington State near the entrance to the Juan de Fuca Strait.
- BC06 - on Reifel Island located at the mouth of the Fraser River near the urban area of Vancouver.
- BC16 is located in the rural area of Saturna Island in the Strait of Juan de Fuca just north of the U.S./Canada border.
Annual Hg loading was higher at the Seattle/NOAA (WA18) site than Reifel Island (BC06) and the rural Makah Hatchery (WA03) site (Figure 12.13). Monitoring at the Saturna Island site was initiated during fall 2009 and a complete year of data from this site was not available for comparison with the other sites. The elevated loadings at the Seattle/NOAA site were not a function of higher precipitation amounts (annual averages of 807 mm at Seattle/NOAA, 813 mm at Reifel Island and 1509 mm at Wakah Hatchery), but instead reflected higher Hg concentrations at this site. Median concentrations for 2001 to 2003 were 6.5 ng/L at Seattle/NOAA (WA18) and 5.2 ng/L at Reifel Island (BC06). For 2008-2009, median concentrations were 9.7 ng/L at Seattle/NOAA compared to 3.2 ng/L at Makah Hatchery. Though a full year of data is not yet available for Saturna Island, the initial data from 2009 and 2010 indicate that it has median Hg concentrations similar to the remote Makah Hatchery site and significantly lower than the urban Seattle/NOAA site. Concentrations ranged from 4.2 ng/L at Saturna (BC16) to 3.1 ng/L at Makah (WA03) and 9.1 ng/L at Seattle/NOAA (WA18) for Sept 2009-June 2010.
Figure 12.13. Annual wet deposition of total mercury for three NADP-MDN sites in the Georgia Basin/Puget Sound area (adapted from NADP, 2010).
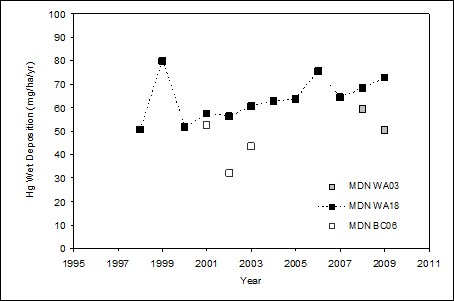
Note: WA03 = Makah Hatchery; WA18 = Seattle NOAA; BC06 = Reifel Island
Description of Figure 12.13
Figure 12.13 is a plot of mercury wet deposition in mg/ha/yr as a fuction of year (from 1995 to 2011) for the NADP-MDN sites at Makah Hatchery (WA03); Seattle NOAA (WA18); and Reifel Island (BC06).
For Makah Hatchery there is data for 2008 and 2009. The mercury wet deposition in these years was approximately 60 mg/ha/yr in 2008 and 50 mg/ha/yr in 2009.
For Seattle NOAA there is data for 1998 through 2009. In 1998 mercury wet deposition was approximately 50 mg/ha/yr, in 1999 this rose sharply to approximately 80 mg/ha/yr before falling back to approximately 50 mg/ha/yr in 2000. From 2000 to 2005 the level rose steadily from approximately 50 to approximately 60 mg/ha/yr. In 2006 there was a sharp rise to approximately 75 mg/ha/yr followed by a fall back to approximately 60 mg/ha/yr in 2007. From 2007 to 2009 the level rose steadily from approximately 60 to approximately 70 mg/ha/yr.
For Reifel Island there is data for 2001 through 2003. The mercury wet deposition in these years was approximately 50 mg/ha/yr in 2001, approximately 30 mg/ha/yr in 2002, and approximately 40 mg/ha/yr in 2003.
A separate study (Brandenberger et al., 2010) also looked at mercury deposition in the Puget Sound area. Their study included data from seven sites spread around the coast of Puget Sound during two periods from September-December 2008 and from February-June 2009. They found median total mercury fluxes of approximately 4-14 ng/m2/d (1.46- 5.11 µg/m2/yr, or 14.6 to 51.1 mg/ha/yr) with no statistical difference between the different sampling stations. They also found that on average 3.9% of the total mercury in precipitation was methyl mercury. They concluded that mercury fluxes in Puget Sound appear to be a combination from both longer-range regional sources and more localized sources in the industrial region of Tacoma.
A large study known as the Western Airborne Contaminants Assessment Project (WACAP) by Landers et al. (2008) detailed the ecological and biological impacts of hazardous air contaminants in eight national parks in the western U.S. and Alaska. In eight core parks, including Olympic and Mt. Rainier National Parks in Puget Sound area, air, snow, water, sediment, lichen, conifer needle and fish samples were collected. In 12 secondary parks, including North Cascades National Park, limited sampling was conducted. Mercury concentrations were analyzed in vegetation from Olympic and Mt. Rainier National Parks. In both Olympic and Rainier Parks, mercury concentrations in vegetation were in the mid to upper ranges compared to concentrations at other parks in the study. Low, but detectable, concentrations of PCBs were also found in vegetation in both these two and in North Cascades Park.
In Olympic National Park fish mercury concentrations were among the highest of all parks sampled, exceeding contaminant health thresholds for piscivorous (fish eating) mammals (otter, mink) and birds (kingfishers). In addition, some fish from both lakes sampled in Olympic National Park exceeded the human contaminant health threshold. In Mt. Rainier National Park, mercury concentrations in all fish from both lakes sampled exceeded contaminant health thresholds for birds (kingfishers), and some fish exceeded thresholds for piscivorous mammals (otter, mink). Mercury concentrations in some fish from one of the lakes in Mt. Rainier National Park exceeded contaminant health thresholds for humans. It is currently thought that most of the mercury entering these national parks arrives via atmospheric deposition from local, regional, and long-range sources (Landers et al., 2008). A subsequent study analyzed mercury and pesticide concentrations in fish collected from 14 lakes in the three national parks. Mercury was detected in fish from all lakes, with the highest concentration in fish from a lake at North Cascades National Park (Moran et al., 2007).
Snow samples and lake sediments were collected in Olympic and Mt. Rainer National Parks as part of WACAP. Snow samples provide a direct measure of annual atmospheric loading since snow is 90% of annual precipitation in the alpine sites where the samples were collected. Lake sediments can provide historic trends of contaminant loading to a particular watershed (Landers et al., 2008).
In Olympic National Park snow pack data were only available for 2004. In this year, mercury fluxes at two sites were quite high (approaching 104 ng/m2/yr or 100 mg/ha/yr), whereas mercury flux at a third site was somewhat less (closer to 103 ng/m2/yr or 10 mg/ha/yr). The high results were surprising, given that there are few known local or regional upwind mercury sources. One possible explanation is that deposition from regional sources to the east can reach the park on easterly airflows. Also, the median was calculated from only a few samples collected during a single year. In Mt. Rainer National Park, snow samples were collected from seven sites in the years 2003 through 2005. Mercury fluxes ranged from 102 to over103 ng/m2/yr or 1.0 mg/ha/yr to over 10 mg/ha/yr. Mercury measured in sediment cores showed a rapid percent enrichment near the surface (present time) of lakes sampled in Mt Rainer National Park. The source of this increase is unknown, but global warming, increased global background, and/or trans-Pacific sources could be responsible (Landers et al., 2008).
Temporal trends
The longest period of measurements exist at the WA18 Seattle/NOAA site which began in 1996 and are currently ongoing. Analyzing the data from 1996 to 2005 at this urban location, Prestbo and Gay (2009) found a significant decreasing trend in the mercury concentrations (-1.1%/year) and deposition. In 1996 and 1997 the volume weighted mean mercury concentrations were 19.8 and 17.5 ng/L and then decreased abruptly down to the range of 5.9 to 9.7 ng/L from 1998 to 2005. This large change is attributed decreased emissions from medical and municipal waste incinerators (Prestbo and Gay, 2009).
At all of the MDN sites, as well as the locations measured in Brandenberger et al. (2010), there was a strong positive correlation between the total Hg deposition and the precipitation amount. Seasonally, the WA18 Seattle/NOAA site (the location with the most extensive dataset) showed that the Hg concentrations were significantly higher during the spring and summer than the winter and fall (median values--spring: 11.5 ng/L; summer: 13.3 ng/L; fall: 5.7 ng/L; winter: 6.1 ng/L); however, the total deposition rates were significantly lower in the summer (median of 11 mg/ha) compared to other seasons due to lower amounts of rainfall (median values of 18 mg/ha for both fall and winter and 16 mg/ha for spring).
Mercury Atmospheric Concentrations
Mercury exists in the atmosphere in several forms/species (gaseous elemental Hg, gaseous oxidized Hg, and particle bound Hg), which have very different depositional velocities. Atmospheric Hg speciation measurements are conducted at several locations throughout Canada, however none are in or near the Georgia Basin/Puget Sound airshed. Atmospheric Hg measurements conducted within the airshed are of TGM, which does not include the particulate fraction. Total gaseous mercury has been measured at two of the locations where precipitation measurements were conducted: Reifel Island (1999 to 2004) and Saturna Island (2009 to present) (Figure 12.14). The Reifel Island site showed a decreasing trend of -0.055 ng m-3yr-1 during the five year period of measurements (Cole et al., 2013). Because measurements at this site were discontinued in 2004, it is unclear if this decreasing trend has continued during the ensuing decade. However, comparing the average concentration from Reifel Island (1.7 ± 0.3 ng/m3) to the more recent concentrations measured at Saturna Island (1.4 ± 0.3 ng/m3) indicate that they are not significantly different and that there haven’t been any major regional changes in mercury levels.
For the Saturna and Reifel Island sites, there were subtle but significant differences in the Hg concentrations between seasons; however the seasonal trends were different between the two sites. At Saturna, the highest concentrations were during the spring (1.6 ng/m3) followed by the summer (1.4 ng/m3), fall (1.2 ng/m3) and winter (1.1 ng/m3), whereas at Reifel Island the highest concentrations were during the winter and spring (each 1.7 ng/m3) and lowest during the summer and fall (each 1.6 ng/m3). It is possible that the seasonality is driven by westerly trans-pacific flows from industrial regions in Asia, since this source has been implicated for other atmospheric contaminants (Jaffe et al., 2003, Harner et al., 2005, Strode et al., 2008).
Figure 12.14. Atmospheric total gaseous mercury concentrations at Reifel Island (1999-2004) and Saturna Island (2009-2010).
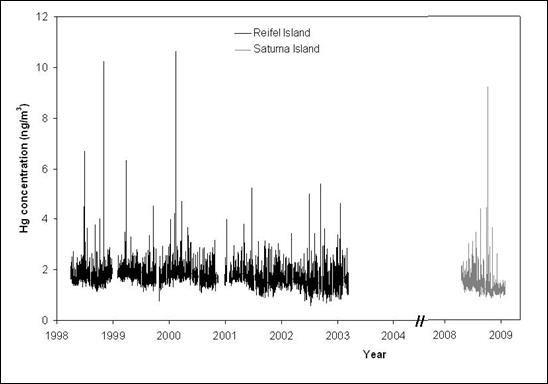
Description of Figure 12.14
Figure 12.14 is a plot of mercury concentration in ng/m3 as a function of year (from 1998 to 2009) for Reifel Island and Saturna Island. The y-axis scale is from 0 to 12 ng/m3 and the data has noise on the order of 1 ng/m3.
There is data from Reifel Island for 1999 through 2004 and no apparent trend can be seen. For Saturna Island there is data from 2009 to 2010 and again no trend is apparent. As stated in the text, the average concentrations from Reifel Island (1.7 ± 0.3 ng/m3) and Saturna Island (1.4 ± 0.3 ng/m3) were not significantly different.
12.6.2 Persistent Organic Pollutants
While the accumulation of Persistent Organic Pollutants (POPs) such as PCBs and PBDEs had been previously recorded in biota in the Puget Sound and Georgia Basin, the magnitude of the issue was highlighted in a study published in 2000, where regional killer whales were described as the “most contaminated cetaceans on earth” (Ross et al., 2000). While various direct discharges to marine waters of the Georgia Basin probably constitute a significant source of these contaminants, there are other areas of North America where atmospheric deposition is a large contributor. Polychlorinated biphenyls are legacy contaminants, which prior to their ban in the mid-1970s, were used for a wide range of industrial processes, including cooling and high-voltage electrical service. Polybrominated diphenyl ethers are flame-retardant chemicals which are used in consumer products, such as polyurethane foam, computer components, carpeting and textiles. PCBs and PBDEs have similar structures (Figure 12.15), with two phenyl rings and up to 209 congeners, each with different possible patterns of substitution of one to 10 chlorine or bromine atoms. The pattern and degree of substitution confers a range of physical characteristics, including volatility, solubility, bioaccumulation and persistence in the environment. Commercial formulations are composed of many different possible congeners which makes precise prediction of their fate a somewhat challenging exercise.
Figure 12.15 Basic structure of polychlorinated biphenyls (PCB) and polybrominated diphenyl ethers (PBDE).
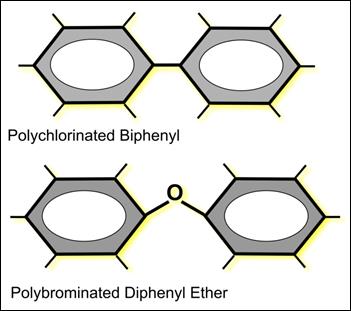
Description of Figure 12.15
Figure 12.15 is a drawing of the chemical structure of a polychlorinated biphenyl and a polybrominated diphenyl ether. Both have a biphenyl structure (two linked benzene rings). In a polychlorinated biphenyl the benzene rings can have any number of attached chlorine atoms. In a polybrominated diphenyl ether the linkage between the benzene rings is an ether linkage.
Noël et al. (2009) quantified atmospheric loadings of PCBs and PDBEs through a year-long sampling of both chemicals in gaseous and rainwater phases at two sites, one within the Strait of Georgia (Saturna Island) and one coastal reference site (Ucluelet). The final annual deposition estimates to the entire Strait of Georgia (8900 km2) were 3.5 ± 0.7 kg/yr for PCBs and 17.1 ± 6.1 kg/yr for PBDEs. Polychlorinated biphenyl levels at Saturna (3.9 mg/ha/yr) mirrored those at a paired site at Ucluelet (4.4 mg/ha/yr) on the far west coast of Vancouver Island, suggesting the two may represent a regional background concentration. In contrast, the PBDE levels differed between the sites both in total loading and composition. At the outer coast site, free from most urban contamination sources, PBDE deposition (both dry and wet) was 8.1 mg/ha/yr while the Saturna site was 19.1 mg/ha/yr. Polybrominated diphenyl ether composition differed greatly between rain and gas phases, with tetra and penta-brominated congeners dominating the gaseous and the commercial deca-brominated congener dominating in the rainfall. This presence of the deca-brominated form in precipitation was thought to be due to an association with particulates that are washed from the air with rainfall. Forty percent of air-mass back trajectories calculated for the Ucluelet site originated over Asia suggesting that a significant portion of the PBDEs at this remote site may have been the result of trans-Pacific transport (Noël et al., 2009).
Air concentrations of some legacy organochlorine pesticides have also been measured near the Strait of Georgia (Yang et al., 2007) and is summarized in Table 12.2. Passive air samplers consisting of a housing and prepared polyurethane foam (PUF) disks were installed on Cortes, Lasqueti and Saturna Islands. The PUF accumulates organic chemicals with a predictable rate over the exposure period, making it possible to estimate average air concentrations (Shoeib and Harner, 2002). Levels of all measured pesticides tended to be highest at the most southerly site on Saturna Island, closest to population centres and for those with some continuing use, such as Hexachlorocyclohexane (HCH) and Endosulphan.
Site |
ΣHCH pg/m3 |
ΣChlordane pg/ m3 |
ΣEndosulphan pg/ m3 |
HepEpox pg/ m3 |
Dieldrin pg/ m3 |
ΣPCB pg/ m3 |
|---|---|---|---|---|---|---|
| Cortes Island |
16.7 - 29.8
|
3.0 - 4.5
|
9.1 - 51.4
|
1.7 - 3.1
|
1.8 - 4.9
|
1.9 - 2.6
|
| Lasqueti lsland |
3.9 - 27.5
|
0.23 - 3.81
|
3.9 - 21.3
|
<DL - 1.6
|
<DL - 2.5
|
<DL - 2.5
|
| Saturna Island |
31.3 - 59.0
|
3.65 - 10.72
|
27.6 - 92.4
|
1.9 - 6.1
|
<DL - 13.5
|
2.1 - 4.4
|
Description of Table 12.2
Table 12.2 shows estimates of air concentrations of organochlorines in air from three sample sites in the Strait of Georgia.
The first row of the table contains the headers “Site”, “HCH (pg/m3)”, “Chlordane (pg/m3)”, “Endosulphan (pg/m3)”, “HepEpox (pg/m3)”, “Dieldrin (pg/m3)”, and “PCB (pg/m3)”. The first column of the table lists the three sample sites in the Strait of Georgia. These are Cortes Island, Lasqueti Island, and Saturna Island. Columns two through seven give the average concentration ranges of 4 quarterly PUF exposures for each organochlorine at each site.
Brandenberger et al. (2010) measured PAH and PDBE fluxes in Puget Sound. Most of the PAHs were pyrogenic in origin with mix of biomass and fossil fuel combustion sources. No strong seasonal trend was observed, however there were some site specific influences. Deposition fluxes at the urban site at Seattle/NOAA were 20-100 ng/m2/d (0.73 to 3.65 mg/ha/yr) while the other sites ranged from 5-50 ng/m2/d (0.18 to 1.83 mg/ha/yr). Polycyclic aromatic hydrocarbons fluxes were highest near an industrial site in Tacoma although the depositional fluxes of a majority of the PAHs in the urban/industrial area of Tacoma have decreased significantly (close to an order of magnitude for most) over the past two decades.
No strong seasonality or spatial trend was observed for PBDEs (Brandenberger et al., 2010), although fluxes were significantly higher near the urban/industrial Tacoma sites. Average fluxes were on the order of 5 - 7 ng/m2/d (0.18 to 0.26 mg/ha/yr, excluding Tacoma sites which were 3 - 4 times higher). Results indicate clearly that urban/industrial areas near Tacoma represent a regional hot spot for PBDE emissions.
In Mt. Rainier National Park, snow samples collected from seven sites in the years 2003 through 2005 showed PCB fluxes of approximately 101 to 102 ng/m2/yr (0.1 to 1.0 mg/ha/yr. Low concentrations of PCBs were detected in air samples from Olympic National Park, but not in air samples from Mt. Rainier or North Cascades National Parks. Low concentrations of PCBs were detected in vegetation at all three parks, with concentrations increasing in elevation. Because forest productivity is high, pesticides scrubbed from the air by vegetation probably contribute significant contaminant loads to the ecosystem via canopy through-fall and needle litter-fall. PBDE concentrations from fish in Golden Lake in Mt. Rainier National Park were the highest among all lakes in the study (Landers et al., 2008).
12.7 Data Gaps in Atmospheric Deposition
A fundamental data gap in assessing potential atmospheric deposition effects is the paucity of current wet or dry deposition measurements. The only current deposition chemistry monitoring in the Georgia Basin airshed is at Saturna Island, which is unfortunately not representative of the region as a whole. Sites at low and high elevation are needed to develop an understanding of changes in deposition related to elevation. While it is possible to model deposition fields using available historical data (Aherne et al.2010), the result is a poor substitute for current synoptic precipitation chemistry. While this information could be obtained using passive techniques, such as ion-exchange collectors (Fenn and Poth 2004; Proemse et al. 2012), the more complete solution need involve regular rainwater sampling and chemical analysis. In terms of critical load estimates and exceedence determination, the suite of variables would include nitrogen and sulphur species and base-cations.
In terms of mercury deposition, establishing a monitoring site located in a high rainfall location along the western BC coast is recommended for several reasons. Environment Canada Hg models (GRAHM) predict that some of the highest Hg deposition loads received in the entire country occur along the BC coast. This is driven not so much by high concentrations but by high rainfall. The Saturna CAPMon site, located in a rain shadow area, is not suitable at capturing this phenomenon, and so currently there are no data being collected that can assess the accuracy of the model's predictions that the West Coast is a deposition "hot spot". Furthermore, the MDN site located at Neah Bay (Makah Fish Hatchery), which was positioned to capture Hg deposition in a high rainfall location along the Pacific Coast in Washington, has recently (2012) been shut down, leaving a large knowledge gap to assess the potential inputs from trans-Pacific transport which are predicted to increase over time. The only existing MDN sites located in the PNW are the Seattle/NOAA site and the Saturna site. However, both of these sites are located in relatively lower rainfall areas for the region and would be expected to be affected by localized PNW urban/industrial sources. The most recently installed NAPS air quality monitoring site located at Amphitrite Point, Ucluelet, does fit the criteria for high rainfall and would likely make a suitable location for monitoring wet deposition of pollutants transported across the Pacific. The current monitoring program includes total gaseous Hg but does not have a precipitation monitoring component. Finally, in addition to measuring total mercury (THg) in precipitation, it is recommended that measurements of methyl mercury (MeHg) be added. A recent study conducted in Coastal California (Weiss-Penzias et al. 2012) discovered that there were surprisingly high MeHg concentrations in fog water/coastal precipitation. This may represent a previously unknown source of MeHg to coastal systems and is currently not being measured at any location in the PNW.
With the continued regional growth in transportation and agricultural emissions, the issue of nitrogen deposition impacts will continue to present a challenge in the airshed (Bergström and Jansson 2006; Schindler et al. 2006). Effects are clearly evident in lichen tissue chemistry (Raymond et al.2010) and lichen community structure (Geiser et al. 2010). While work to define effects-based critical loads for nitrogen is underway in the Puget Sound portion of the basin, including paleolimnological estimates (Saros et al. 2011), such work needs be conducted in BC to provide a suitable regional mapping. A relatively simple biomonitoring program using indicators, such as lichen tissue chemistry, lichen community structure or leaf/needle N-content (i.e. Augustin et al. 2005) could also be implemented to evaluate biological effects of N or S deposition.
All deposition effect assessments rely on accurate and current emission inventories coupled with atmospheric modelling to provide critical synoptic coverage. Emission inventories need to incorporate both point and non-point sources, the latter often being poorly characterized. For example, Hg emissions from mining operations with large footprints are not currently included into any of the existing inventories in either the U.S. or Canada. There need be a regional implementation of an atmospheric model such as CMAQ, AURAMS or similar which would guide monitoring network development. Model output would provide broad spatial deposition estimates for variables of concern, such as nitrogen, sulphur, mercury and could include ozone and particulates.
12.8 Chapter Summary
This chapter examined the patterns, levels and impacts of dry and wet deposition of contaminants to various environmental receptors, including soils, forests, vegetation and aquatic systems. Studies show that deposited compounds of sulphur and nitrogen, and hazardous air pollutants such as mercury, are found to have adverse effects on the ecosystems of Georgia Basin/Puget Sound airshed.
12.8.1 Summary of Sulphur and Nitrogen Deposition
Sulphur and nitrogen deposition can produce adverse effects in terrestrial and aquatic ecosystems through acidification and eutrophication of surface waters, reduced plant species diversity and possible declines in forest growth. As the portions of the Georgia Basin/Puget Sound airshed are characterized by soils with poor acid buffering capacity due to low bedrock weathering rates, the region is vulnerable to adverse environmental effects, even though regional emissions of acidifying compounds are lower than in eastern Canada and the U.S., where acid rain has a long history. Excess nitrogen deposition is also a concern for N-limited lakes, lichens and other alpine and sub-alpine vegetation.
Measurements of annual total sulphur loads at three remote sites in the basin (Saturna Island, Mount Rainier National Park, North Cascades National Park) for the period 2004-2006 ranged from 1.0-2.5 kg/ha/yr. Total nitrogen was slightly higher at 1.1-3.1 kg/ha/yr. Loads at the urban sites, such as those in Vancouver and Victoria, were higher with S deposition in the range of 2.5-5.5 kg/ha/yr and nitrogen about 8-8.5 ka/ha/yr. Compared to an earlier three year period, 1998-2000, all sites experienced a decline in annual total (wet + dry) sulphur and nitrogen. This downward trend was mirrored in eastern Canada (Aherne et al., 2010, Feller, 2010), suggesting that all regions have responded similarly to large-scale emissions reductions under the Canada-U.S. Air Quality Agreement.
CMAQ-modelled sulphur and nitrogen deposition loading for Georgia Basin/Puget Sound was in the range of 1-12 kg/ha/yr, except for certain hot spots near urban centres, shipping routes and areas of intense agricultural activity. Direct measurements taken between 1999 and 2002 at Chilliwack in the eastern Lower Fraser Valley show very high annual total nitrogen loading of roughly 30 kg/ha/yr associated with agricultural activity (Hay et al., 2006).
The concept of critical loads is applied to assess potential harmful levels of sulphur and nitrogen deposition to regional terrestrial and aquatic ecosystems. Aquatic critical loads were based on the capacity of the water body to buffer incoming acidifying deposition and maintain an ANC protective of biological communities or to assimilate deposited nitrogen so that there is no nitrogen-induced shift in diatom species composition. Soil critical loads were established to maintain ratios of base cations to aluminum to support healthy plant growth.
A number of studies conducted between 2005 and 2010 suggest that sulphur and nitrogen loads exceeded critical loads for various receptors in Georgia Basin/Puget Sound. One study (Nasr et al., 2010) found that 13% to 32% of the total surface area of Georgia Basin had sulphur and nitrogen loads that exceeded the critical load for soil acidity under present-day deposition levels. Site-specific study at 19 forest plots in SW BC showed similar results. The highest critical load exceedences were found in the northern uplands of the Lower Fraser Valley where deposition is relatively high and the soils are thin with low buffering capacity. In another study of 72 freshwater lakes in Georgia Basin, almost 20% of the lakes sampled had pH values less than 6.0, the level at which negative biological impacts are expected to occur (Strang et al., 2010). Thirteen of the lakes (18%) received sulphur deposition in excess of their critical load during 2005 - 2006. Other studies on forested ecosystems (Mongeon et al., 2010) and lichens (Geiser et al., 2010, Raymond et al., 2010) showed that these receptors experience regular exceedences of critical loads, particularly those close to or within the populated metropolitan areas under high atmospheric deposition.
12.8.2 Summary of Hazardous Air Pollutants
Toxic or hazardous air pollutants (HAPs) are associated with serious health risks and ecological impacts. Several HAPs are readily deposited onto soils or surface waters, where they can be taken up by plants and ingested by animals and can eventually be magnified up through the food chain. For example, mercury released to the atmosphere can have a long atmospheric lifetime and be transported around the globe. Following deposition, mercury can be transformed into methyl mercury, which has increased toxicity and is the dominant form of mercury that accumulates in aquatic organisms and the humans and wildlife that consume them.
In Mt. Rainer National Park, snow samples collected from seven sites during 2003-2005 showed mercury fluxes in a range of 1.0 mg/ha/yr to over 10 mg/ha/yr (Landers et al., 2008). Mercury concentrations in rainfall measured at two remote and two urban sites were found to be highest at the Seattle/NOAA urban site after accounting for differences in precipitation. There was no significant temporal trend in mercury concentrations at the Seattle/NOAA site based on records dating back to 1996.
Average concentrations of total gaseous mercury (TGM) measured at Reifel Island (1.7 ng/m3) and Saturna Island (1.4 ng/m3) were not significantly different despite the 5-year time period difference between the measurements, suggesting that atmospheric mercury concentrations have remained stable over the last decade.
In the WACAP program (Landers et al., 2008) the ecological and biological impacts of hazardous air contaminants were assessed in 20 national parks in the western U.S. and Alaska. In Olympic National Park, fish mercury concentrations were among the highest of all parks sampled. At both Olympic National Park and Mt. Rainier National Park, contaminant health thresholds were exceeded in some instances for otter, mink and kingfishers that consume fish. In addition, some fish from lakes sampled in both parks exceeded the human contaminant health threshold. It is currently thought that most of the mercury entering these national parks arrives via atmospheric deposition from local, regional and long-range sources.
Studies by Noël et al. (2009) and Brandenberger et al.(2010) show the presence and magnitude of Persistent Organic Pollutants such as PCBs and PDBEs in Georgia Basin and Puget Sound, with particularly high levels occurring in proximity to industrial sources in Puget Sound. Analysis of airmass back trajectories also suggests possible long range trans-Pacific transport of PDBEs to the basin.
12.9 References
Aber, J. D., Nadelhoffer, K. J., Steudler, P., and Melillo, J. M., 1989. Nitrogen saturation in northern forest ecosystems. BioScience 39: 378-386.
Aherne, J., Mongeon, A., and Watmough, S. A., 2010. Temporal and spatial trends in precipitation chemistry in the Georgia Basin, British Columbia. Journal of Limnology 69 (Suppl. 1): 4-10.
Aherne, J. and Shaw, D. P., 2010. Impacts of sulphur and nitrogen deposition in western Canada. Journal of Limnology 69 (Suppl. 1): 1-3.
Augustin, S., Bolte, A., Holzhausen, M., and Wolff, B., 2005. Exceedance of critical loads of nitrogen and sulphur and its relation to forest conditions. European Journal of Forest Research 124: 289-300.
Baron, J. S., 2006. Hindcasting nitrogen deposition to determine an ecological critical load. Ecological Applications 16: 433-439.
Baron, J. S., Rueth, H. M., Wolfe, A. M., Nydick, K. R., Allstott, E. J., Minear, J. T., and Moraska, B., 2000. Ecosystem responses to nitrogen deposition in the Colorado Front Range. Ecosystems 3: 352-368.
Bergström, A.-K. and Jansson, M., 2006. Atmospheric nitrogen deposition has caused nitrogen enrichment and eutrophication of lakes in the northern hemisphere. Global Change Biology 12: 635-643.
Bleeker, A., Hicks, W. K., Dentener, F., Galloway, J., and Erisman, J. W., 2011. N deposition as a threat to the World's protected areas under the Convention on Biological Diversity. Environmental Pollution 159: 2280-2288.
Bobbink, R., Hicks, K., Galloway, J., Spranger, T., Alkemade, R., Ashmore, M., Bustamante, M., Cinderby, S., Davidson, E., Dentener, F., Emmett, B., Erisman, J.-W., Fenn, M., Gilliam, F., Nordin, A., Pardo, L., and De Vries, W., 2010. Global assessment of nitrogen deposition effects on terrestrial plant diversity: a synthesis. Ecological Applications 20: 30-59.
Bowman, W. D., Gartner, J. R., Holland, K., and Wiedermann, M., 2006. Nitrogen critical loads for alpine vegetation and terrestrial ecosystem response: Are we there yet? Ecological Applications 16:1183-1193.
Brandenberger, J. M., Louchouarn, P., Kuo, L.-J., Crecelius, E. A., Cullinan, V., Gill, G. A., Garland, C., Williamson, J., and Dhammapala, R., 2010. Control of Toxic Chemicals in Puget Sound, Phase 3: Study of Atmospheric Deposition of Air Toxics to the Surface of Puget Sound. Report for the Washington Department of Ecology (Air Quality Program).
Byun, D. and Schere, K. L., 2006. Review of the governing equations, computational algorithms, and other components of the models-3 Community Multiscale Air Quality (CMAQ) modeling system. Applied Mechanics Reviews 59:51-76.
Cole, A.S., Steffen, A., Eckley, C., Narayan, J., Pilote, M., Graydon, J., Tordon, R., St. Louis, V. 2013. A survey of mercury in air and precipitation across Canada: patterns and trends. Conference poster presentation at International Conference on Mercury as a Global Pollutant (11th annual) Edinburgh, Scotland. July 2013.
Compton, J. E., Church, M. R., Larned, S. T., and Hogsett, W. E., 2003. Nitrogen export from forested watersheds in the Oregon Coast Range: The role of N2-fixing red alder. Ecosystems 6:773-785.
E&S Environmental Chemistry, Inc., Sullivan, T., McDonnell, T., McPherson, G., Mackey S., Moore D., 2011. Evaluation of the Sensitivity of Inventory and Monitoring National Parks to Nutrient Enrichment Effects from Atmospheric Nitrogen Deposition. Main Report. Prepared for Natural Resource Program Center, National Park Service, U.S. Department of the Interior. Natural Resource Report NPS/NPRC/ARD/NRR-2011/313.
Emmett, B. A., 2007. Nitrogen saturation of terrestrial ecosystems: Some recent findings and their implications for our conceptual framework. Water, Air, and Soil Pollution: Focus 7:99-109.
Feller, M. C., 2010. Trends in precipitation and streamwater chemistry in East Creek watershed in southwestern British Columbia, 1971-2008. Journal of Limnology 69 (Suppl. 1) 77-91.
Fenn, M. E. and Poth, M. A., 2004. Monitoring nitrogen deposition in throughfall using ion exchange resin columns: A field test in the San Bernardino Mountains. Journal of Environmental Quality 33: 2007-2014.
Fenn, M. E., Baron, J. S., Allen, E. B., Rueth, H. M., Nydick, K. R., Geiser, L., Bowman, W. D., Sickman, J. O., Meixner, T., Johnson, D. W., and Neitlich, P. N., 2003. Ecological effects of nitrogen deposition in the Western United States. BioScience 53: 404-420.
Fenn, M. E., Geiser, L., Bachman, R., Blubaugh, T. J., and Bytnerowicz, A., 2007. Atmospheric deposition inputs and effects on lichen chemistry and indicator species in the Columbia River Gorge, USA. Environmental Pollution 146: 77-91.
Galloway, J. N., Aber, J. D., Erisman, J. W., Seitzinger, S. P., Howarth, R. W., Cowling, E. B., and Cosby, B. J., 2003. The nitrogen cascade. BioScience 53: 341-356.
Geiser, L. H., Jovan, S. E., Glavich, D. A., and Porter, M. K., 2010. Lichen-based critical loads for atmospheric nitrogen deposition in Western Oregon and Washington Forests, USA. Environmental Pollution 158: 2412-2421.
Geiser, L. H. and Neitlich, P. N., 2007. Air pollution and climate gradients in western Oregon and Washington indicated by epiphytic macrolichens. Environmental Pollution 145: 203-218.
Greene, A. R., Scoates, J. S., and Weis, D., 2005. Wrangellia Terrane on Vancouver Island, British Columbia: Distribution of flood basalts with implications for potential Ni-Cu-PGE mineralization in southwestern British Columbia. Page 365 Geological Survey of BC. British Columbia Ministry of Energy and Mines, Victoria, British Columbia.
Harner, T., Shoeib, M., Kozma, M., Gobas, F. A. P. C., and Li, S. M., 2005. Hexachlorocyclohexanes and endosulfans in urban, rural, and high altitude air samples in the Fraser Valley, British Columbia: Evidence for trans-Pacific transport. Environmental Science & Technology 39: 724-731.
Hay, J., Raymond, B., and Vingarzan, R., 2006. Dry and wet atmospheric deposition at six sites in the Georgia Basin. Environment Canada internal report (unpublished).
Henriksen, A. and Posch, M., 2001. Steady-state models for calculating critical loads of acidity for surface waters. Water, Air, & Soil Pollution: Focus 1: 375-398.
Henrys, P. A., Stevens, C. J., Smart, S. M., Maskell, L. C., Walker, K. J., Preston, C. D., Crowe, A., Rowe, E., Gowing, D. J., and Emmett, B. A., 2011. Nitrogen impacts on vascular plants in Britain: An analysis of two national observation networks. Biogeosciences Discussions 8: 7441-7474.
Holt, C. A., Yan, N. D., and Somers, K. M., 2003. pH 6 as the threshold to use in critical load modeling for zooplankton community change with acidification in lakes of south-central Ontario: accounting for morphometry and geography. Canadian Journal of Fisheries and Aquatic Sciences 60: 151-158.
Jaffe, D., McKendry, I., Anderson, T., and Price, H., 2003. Six 'new' eisodes of trans-Pacific transport of air pollutants. Atmospheric Environment 37: 391-404.
Jeffries, D. S., 1997. 1997 Canadian Acid Rain Assessment Volume 3: The Effects on Canada's Lakes, Rivers and Wetlands. Environment Canada, Burlington, Ontario.
Jeffries, D. S. and R. Ouimet., 2004. Critical Loads: Are they being exceeded? Pages 341-369. Canadian Acid Deposition Science Assessment 2004. Environment Canada, Ottawa, Ontario.
Jeziorski, A., Yan, N. D., Paterson, A. M., DeSellas, A. M., Turner, M. A., Jeffries, D. S., Keller, B., Weeber, R. C., McNicol, D. K., Palmer, M. E., McIver, K., Arseneau, K., Ginn, B. K., Cumming, B. F., and Smol, J. P., 2008. The widespread threat of calcium decline in fresh waters. Science 322: 1374-1377.
Krzyzanowski, J., McKendry, I. G., and Innes, J. L., 2006. Evidence of elevated ozone concentrations on forested slopes of the Lower Fraser Valley, British Columbia, Canada. Water, Air, and Soil Pollution 173: 273-287.
Landers, D. H., Simonich, S. L., Jaffe, D. A., Geiser, L. H., Campbell, D. H., Schwindt, A. R., Schreck, C. B., Kent, M. L., Hafner, W. D., Taylor, H. E., Hageman, K. J., Usenko, S., Ackerman, L. K., Schrlau, J. E., Rose, N. L., Blett, T. F., and Erway, M. M., 2008. The Fate, Transport, and Ecological Impacts of Airborne Contaminants in Western National Parks (USA). EPA/600/R-07/138, Corvallis, Oregon.
Lawrence, G. B., 2002. Persistent episodic acidification of streams linked to acid rain effects on soil. Atmospheric Environment 36: 1589-1598.
MACTEC Engineering and Consulting, 2010. Clean Air Status and Trends Network (CASTNET) 2008 Annual Report. Prepared by MACTEC Engineering and Consulting, Inc. Prepared for: U.S. Environmental Protection Agency, Office of Air and Radiation, Clean Air Markets Division.
Mongeon, A., Aherne, J., and Watmough, S. A., 2010. Steady-state critical loads of acidity for forest soils in the Georgia Basin, British Columbia. Journal of Limnology 69 (Suppl. 1): 193-200.
Moran, M. D., Zheng, Q., Pavlovic, R., Cousineau, S., Bouchet, V. S., Sassi, M., Makar, P. A., Gong, W., and Stroud, C., 2008. Predicted acid deposition critical-load exceedances across Canada from a one-year simulation with a regional particulate-matter model. Page 20 pp in Proc. 15th Joint AMS/A&WMA Conf. on Applications of Air Pollution Meteorology. American Meteorological Society, Boston, New Orleans.
Moran, P. W., Aluru, N., Black, R. W., and Vijayan, M. M., 2007. Tissue contaminants and associated transcriptional response in trout liver from high elevation lakes of Washington. Environmental Science and Technology 41: 6591-6597.
Nanus, L., Williams, M. W., Campbell, D. H., Tonnessen, K. A., Blett, T., and Clow, D. W., 2009. Assessment of lake sensitivity to acidic deposition in national parks of the Rocky Mountains. Ecological Applications 19: 961-973.
Nasr, M., Castonguay, M., Oglivie, J., Raymond, B. A., and Arp, P. A., 2010. Modelling and mapping critical loads and exceedances for the Georgia Basin, British Columbia, using a zero base-cation depletion criterion. Journal of Limnology 69 (Suppl. 1): 181-192.
Niemi, D., 2004. Emissions of Pollutants Related to Acid Deposition in North America. Chapter 2. Pages 5-14. Canadian Acid Deposition Assessment, 2004. Environment Canada.
Nilsson, J., Grennfelt, P., and (Eds)., 1988. Critical loads for sulphur and nitrogen. Miljørapport 1988:15, NORD 1998:097, Nordic Council of Ministers, Copenhagen, Denmark: 418 pp.
Noël, M., Dangerfield, N., Hourston, R. A. S., Belzer, W., Shaw, P., Yunker, M. B., and Ross, P. S., 2009. Do trans-Pacific air masses deliver PBDEs to coastal British Columbia, Canada? Environmental Pollution 157: 3404-3412.
Porter, E. and Johnson, S., 2007. Translating science into policy: Using ecosystem thresholds to protect resources in Rocky Mountain National Park. Environmental Pollution 149:268-280.
Porter, M. K., 2007. Regional modeling of nitrogen, sulphur, and mercury atmospheric deposition in the Pacific Northwest. M.Sc. Washington State University, Pullman, Washington.
Posch, M., Aherne, J., and Hettelingh, J.-P., 2011. Nitrogen critical loads using biodiversity-related critical limits. Environmental Pollution 159: 2223-2227.
Prestbo E.M., Gay, D.A. , 2009. Wet deposition of mercury in the US and Canada, 1996-2005: Results and analysis of the NADP mercury deposition network (MDN). Atmospheric Environment 43: 4223-4233.
Proemse, B. C., Mayer, B., and Fenn, M. E., 2012. Tracing industrial sulfur contributions to atmospheric sulfate deposition in the Athabasca oil sands region, Alberta, Canada. Applied Geochemistry 27: 2425-2434.
Raymond, B. A., Bassingthwaighte, T., and Shaw, D. P., 2010. Measuring nitrogen and sulphur deposition in the Georgia Basin, British Columbia, using lichens and moss. Journal of Limnology 69 (Suppl. 1): 22-32.
Ross, P. S., Ellis, G. M., Ikonomou, M. G., Barrett-Lennard, L. G., and Addison, R. F., 2000. High PCB concentrations in free-ranging Pacific Killer Whales, Orcinus orca: Effects of age, sex and dietary preference. Marine Pollution Bulletin 40: 504-515.
Saros, J., Clow, D., Blett, T., and Wolfe, A., 2011. Critical nitrogen deposition loads in high-elevation lakes of the western US inferred from paleolimnological records. Water, Air and Soil Pollution 216: 193-202.
Schindler, D., Dillon, P., and Schreier, H., 2006. A review of anthropogenic sources of nitrogen and their effects on Canadian aquatic ecosystems. Biogeochemistry 79: 25-44.
Schwede, D., Zhang, L., Vet, R., and Lear, G., 2011. An intercomparison of the deposition models used in the CASTNET and CAPMoN networks. Atmospheric Environment 45: 1337-1346.
Shoeib, M., and Harner, T., 2002. Characterization and comparison of three passive air samplers for persistent organic pollutants. Environmental Science and Technology 36: 4142-4151.
Stoddard, J. L., 1994. Long-term changes in watershed retention of nitrogen: its causes and aquatic consequences. Pages 223-284 in L. A. Baker, editor. Environmental Chemistry of Lakes and Reservoirs. American Chemical Society, Washington, D.C.
Stowell, H. H. and McClelland, W. C., and editors., 2000. Tectonics of the Coast Mountains, southeastern Alaska and British Columbia. Geological Society of America, Boulder, Co.
Strang, D., Aherne, J., and Shaw, D. P., 2010. The hydrochemistry of high-elevation lakes in the Georgia Basin, British Columbia. Journal of Limnology 69 (Suppl. 1): 56-66.
Strode, S. A., Jaeglé, L., Jaffe, D. A., Swartzendruber, P. C., Selin, N. E., Holmes, C., and Yantosca, R. M., 2008. Trans-Pacific transport of mercury. Journal of Geophysical Research D: Atmospheres 113: D15305.
Tomlinson, G. H., 2003. Acidic deposition, nutrient leaching and forest growth. Biogeochemistry 65: 51-81.
UBC, 2007. Data extracted for Environment Canada from the output of the CMAQ model run at UBC for 1 March 2005 to 31 December 2006.
U.S. Environmental Protection Agency, 2010. Clean Air Status and Trends Network (CASTNET).
Vet, R., Brook, J., Ro, C., Shaw, M., Narayan, J., Zhang, L., Moran, M. D., and Lusis, M., 2004. Atmospheric Response to Past Emission Control Programs. Chapter 3. Pages 15-90. Canadian Acid Deposition Assessment, 2004. Environment Canada.
Warfvinge, P. and Sverdrup, H., 1992. Calculating critical loads of acid deposition with profile. A steady-state soil chemistry model. Water, Air, and Soil Pollution 63: 119-143.
Weiss-Penzias, P. S., Ortiz, Jr., C., Acosta, R. P., Heim, W., Ryan, J. P., Fernandez, D., Collett, Jr., J. L., and Flegal, A. R., 2012. Total and monomethyl mercury in fog water from the central California coast. Geographical Research Letters 39: L03804.
Wiens, J. H., Padbury, G. A., Fessenden, R. J., Wotton, D. L., Senyk , J. P., Paquin, E. and Holowaychuk, N., 1987. Sensitivity of western and northern Canada soils and geology to acidic inputs. Technical Committee for the Long-Range Transport of Atmospheric Pollutants in Western and Northern Canada., Coordinating Committee on Soil and Geology Sensitivity Mapping (Canada). Victoria, B.C.
Yang, S., and Lee, S.-C., 2007. Persistent organic pollutants in the Strait of Georgia air. University of Toronto, M.Env. Sci. internship report. Unpublished.