Draft screening assessment - Diazenedicarboxamide
Official Title: Draft Screening Assessment Diazenedicarboxamide
Chemical Abstracts Service Registry Number:
123-77-3
Environment and Climate Change Canada
Health Canada
April 2019
Synopsis
Pursuant to section 74 of the Canadian Environmental Protection Act, 1999 (CEPA), the Minister of the Environment and the Minister of Health have conducted a screening assessment of diazenedicarboxamide under the Chemicals Management Plan. The Chemical Abstracts Service Registry Number (CAS RNFootnote 1) for diazenedicarboxamide is 123-77-3. This substance was identified as a priority for assessment as it met categorization criteria under subsection 73(1) of CEPA.
Diazenedicarboxamide does not occur naturally in the environment.
During the 2011 calendar year, diazenedicarboxamide was not reported to be manufactured in Canada above the reporting threshold of 100 kg. During that same period 100 000 to 1 000 000 kg of the substance were reported to be imported into Canada. Diazenedicarboxamide was reported to have commercial uses in the food and beverage industry, and in the manufacture of polyvinyl chloride for building or construction materials. The substance was also reported to be used in automotive manufacturing and fabrication of floor coverings in Canada.
Additional uses in Canada include its use as a permitted food additive, and as a component in polyvinyl chloride closure-based food packaging materials, such as liners and gaskets.
The ecological risk of diazenedicarboxamide was characterized using the ecological risk classification of organic substances (ERC), which is a risk-based approach that employs multiple metrics for both hazard and exposure, with weighted consideration of multiple lines of evidence for determining risk classification. Hazard profiles are based principally on metrics regarding mode of toxic action, chemical reactivity, food web-derived internal toxicity thresholds, bioavailability, and chemical and biological activity. Metrics considered in the exposure profiles include potential emission rate, overall persistence, and long-range transport potential. A risk matrix is used to assign a low, moderate or high level of potential concern for substances on the basis of their hazard and exposure profiles. Based on the outcome of the ERC analysis, diazenedicarboxamide is considered unlikely to be causing ecological harm.
Considering all available lines of evidence presented in this draft screening assessment, there is low risk of harm to the environment from diazenedicarboxamide. It is proposed to conclude that diazenedicarboxamide does not meet the criteria under paragraphs 64(a) or (b) of CEPA as it is not entering the environment in a quantity or concentration or under conditions that have or may have an immediate or long-term harmful effect on the environment or its biological diversity or that constitute or may constitute a danger to the environment on which life depends.
Diazenedicarboxamide has previously been assessed through the Organisation for Economic Co-operation and Development (OECD) Cooperative Chemicals Programme, and an OECD Screening and Information Dataset Initial Assessment Report (SIAR) is available. This report was used to inform the health effects section of this screening assessment. Effects in laboratory studies, including effects on the kidney, were observed at high doses. When used as a permitted food additive, diazenedicarboxamide decomposes relatively quickly in wet flour and when subjected to high temperature treatment. Due to typical baking conditions, dietary exposure from use as a permitted food additive is expected to be negligible. Exposure from its use in food packaging is not expected as there is no direct contact with the food.
There is a potential for dermal contact with diazenedicarboxamide via use of foam or rubber products manufactured with this substance. However, the residual levels of diazenedicarboxamide expected to be present in products available to consumers is expected to result in only minimal dermal exposure. Potential exposure via inhalation is not expected due to the negligible vapour pressure of the substance. On the basis of these considerations, exposure for the general population is expected to be minimal and the potential risk to human health is considered to be low.
On the basis of the information presented in this draft screening assessment, it is proposed to conclude that diazenedicarboxamide does not meet the criteria under paragraph 64(c) of CEPA as it is not entering the environment in a quantity or concentration or under conditions that constitute or may constitute a danger in Canada to human life or health.
Therefore, it is proposed to conclude that diazenedicarboxamide does not meet any of the criteria set out in section 64 of CEPA.
1. Introduction
Pursuant to section 74 of the Canadian Environmental Protection Act, 1999 (CEPA) (Canada 1999), the Minister of the Environment and the Minister of Health have conducted a screening assessment on diazenedicarboxamide to determine whether this substance presents or may present a risk to the environment or to human health. This substance was identified as a priority for assessment as it met categorization criteria under subsection 73(1) of CEPA (ECCC, HC [modified 2017]).
The ecological risk of diazenedicarboxamide was characterized using the ecological risk classification of organic substances (ERC) (ECCC 2016a). The ERC describes the hazard of a substance using key metrics, including mode of toxic action, chemical reactivity, food web-derived internal toxicity thresholds, bioavailability, and chemical and biological activity, and considers the possible exposure of organisms in the aquatic and terrestrial environments on the basis of such factors as potential emission rates, overall persistence, and long-range transport potential in air. The various lines of evidence are combined to identify substances as warranting further evaluation of their potential to cause harm to the environment or as having a low likelihood of causing harm to the environment.
This substance has been reviewed internationally through the Organisation for Economic Co-operation and Development (OECD) Cooperative Chemicals Assessment Programme, and there is an OECD Screening Information Dataset (SIDS) Initial Assessment Report (SIAR) available (OECD 2001). These assessments undergo rigorous review (including peer review) and endorsement by international governmental authorities. Health Canada and Environment and Climate Change Canada are active participants in this process and consider these assessments to be reliable. The OECD SIAR was used to inform the health effects characterization in this screening assessment.
This draft screening assessment includes consideration of information on chemical properties, environmental fate, hazards, uses and exposures, including additional information submitted by stakeholders. Relevant data were identified up to March 2018. Empirical data from key studies as well as results from models were used to reach proposed conclusions. When available and relevant, information presented in assessments from other jurisdictions was considered.
This draft screening assessment was prepared by staff in the CEPA Risk Assessment Program at Health Canada and Environment and Climate Change Canada, based on a draft developed by staff at Tetra Tech Incorporated, and incorporates input from other programs within these departments. The ecological portion of this assessment is based on the ERC document (published July 30, 2016), which was subject to an external review as well as a 60-day public comment period. While external comments were taken into consideration, the final content and outcome of this draft screening assessment remain the responsibility of Health Canada and Environment and Climate Change Canada.
This draft screening assessment focuses on information critical to determining whether the substance met the criteria as set out in section 64 of CEPA, by examining scientific information and incorporating a weight-of-evidence approach and precautionFootnote 2 . This draft screening assessment presents the critical information and considerations upon which the proposed conclusion is made.
2. Substance identity
The Chemical Abstracts Service Registry Number (CAS RNFootnote 3), Domestic Substances List (DSL) name and common names for diazenedicarboxamide are presented in Table 2‑1.
| CAS RN | DSL name (common names) | Chemical structure and molecular formula | Molecular weight (g/mol) |
|---|---|---|---|
| 123-77-3 | Diazenedicarboxamide (1,1-azobis[formamide]; 1,1’-azobispformamide]; azodicarbonamide; diazene-1,2-dicarboxamide) | 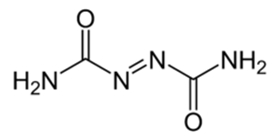 C2H4N4O2 C2H4N4O2 |
116.08 |
3. Physical and chemical properties
A summary of physical and chemical property data of diazenedicarboxamide is presented in Table 3-1. Available data are shown at standard temperatures and pressures. Additional physical chemical properties are reported in ECCC 2016b.
| Property | Value | Type of data | References |
|---|---|---|---|
| Melting point (oC) | 212 | Experimental | ChemIDPlus 2018 |
| Boiling point (oC) | 225 | Not Specified | PubChem 2004 |
| Water solubility (mg/L) | 35 | Experimental | ChemIDPlus 2018, OECD 2001 |
| Density (g/mL) | 1.65 | Not Specified | PubChem 2004 |
| Vapour pressure (Pa) | 2.5 × 10-8 | Experimental | ChemIDPlus 2018, OECD 2001 |
| Henry’s law constant (Pa m3/mol) | 8.3 × 10-8 | Estimated | PubChem 2004 |
| log Kow (dimensionless) | -3.89 to -1.7 | Estimated | ChemIDPlus 2018, OECD 2001 |
4. Sources and uses
Diazenedicarboxamide does not occur naturally in the environment.
Diazenedicarboxamide was included in a survey issued pursuant to a CEPA section 71 notice (Canada 2012). In Canada, diazenedicarboxamide was not reported to be manufactured above the reporting threshold of 100 kg during the 2011 calendar year. During that same period 100 000 to 1 000 000 kg of the substance were reported to be imported into Canada (Environment Canada 2013).
In Canada, the substance was reported to have commercial uses in the food and beverage industry, and in the manufacture of polyvinyl chloride materials for building or construction materials. Diazenedicarboxamide was also reported to have consumer uses in automotive manufacturing, and fabrication of floor coverings (Environment Canada 2013). Additional Canadian uses of diazenedicarboxamide are listed in Table 4‑1.
Use |
Details |
|---|---|
Permitted food additivea |
Permitted for use in bread, flour, and whole wheat flour at a maximum level of 45 ppm of flour. |
Food packaging materialsa |
Component in polyvinyl chloride closure based materials such as liners and gaskets |
Internal drug product database as medicinal or non-medicinal ingredients in final pharmaceutical, disinfectant or veterinary drug products in Canadab |
None |
Licensed Natural Health Products Database being present as a medicinal or non-medicinal ingredient in natural health products in Canadac |
None |
Notified to be present in cosmetics, based on notifications submitted under the Cosmetic Regulations Health Canadad |
No notifications |
Formulant in pest control products registered in Canadae |
Use as a formulant in two insecticides |
a Email from the Food Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, dated January 23, 2018; unreferenced.
b Email from Therapeutic Products Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, dated June 2015; unreferenced).
c LNHPD (modified 2018).
d Email from Consumer Product Safety Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, dated January 26, 2018; unreferenced.
e Email from Pest Management Regulatory Agency, Canada, to Existing Substances Risk Assessment Bureau, Health Canada, dated June 26, 2015; unreferenced.
Diazenedicarboxamide has been reported to be used as a foaming agent in the manufacture of plastics and rubber (OECD 2001). Its use as a foaming agent results in finished products that have improved functionality as an insulator, noise dampening barrier, ability to resist water, or offer lightweight solutions to increased structural performance (ECHA 2014). Plastic and rubber materials formulated with diazenedicarboxamide can be found in automobile interiors and in noise, vibration and harshness reduction systems (ECHA 2014). Building materials manufactured from diazenedicarboxamide-fabricated plastics include insulating material, weather stripping, water based adhesives, laminate flooring underlay, carpet backing, extruded plastic for door and window frames, and polyvinyl chloride foam core pipe (ECHA 2014, MSDS 2015, Schipper et al 1996).
5. Potential to cause ecological harm
5.1 Characterization of ecological risk
The ecological risk of diazenedicarboxamide was characterized using the ecological risk classification of organic substances (ERC) approach (ECCC 2016a). The ERC is a risk-based approach that considers multiple metrics for both hazard and exposure, with weighted consideration of multiple lines of evidence for determining risk classification. The various lines of evidence are combined to discriminate between substances of lower or higher potency and lower or higher potential for exposure in various media. This approach reduces the overall uncertainty with risk characterization compared to an approach that relies on a single metric in a single medium (e.g., LC50) for characterization. The following summarizes the approach, which is described in detail in ECCC (2016a).
Data on physical-chemical properties, fate (chemical half-lives in various media and biota, partition coefficients, and fish bioconcentration), acute fish ecotoxicity, and chemical import or manufacture volume in Canada were collected from the scientific literature, available empirical databases (e.g., OECD QSAR Toolbox), and responses to surveys conducted under section 71 of CEPA, or they were generated using selected quantitative structure-activity relationship (QSAR) or mass-balance fate and bioaccumulation models. These data were used as inputs to other mass-balance models or to complete the substance hazard and exposure profiles.
Hazard profiles were based principally on metrics regarding mode of toxic action, chemical reactivity, food web-derived internal toxicity thresholds, bioavailability, and chemical and biological activity. Exposure profiles were also based on multiple metrics including potential emission rate, overall persistence, and long-range transport potential. Hazard and exposure profiles were compared to decision criteria in order to classify the hazard and exposure potentials for each organic substance as low, moderate, or high. Additional rules were applied (e.g., classification consistency, margin of exposure) to refine the preliminary classifications of hazard or exposure.
A risk matrix was used to assign a low, moderate or high classification of potential risk for each substance on the basis of its hazard and exposure classifications. ERC classifications of potential risk were verified using a two-step approach. The first step adjusted the risk classification outcomes from moderate or high to low for substances that had a low estimated rate of emission to water after wastewater treatment, representing a low potential for exposure. The second step reviewed low risk potential classification outcomes using relatively conservative, local-scale (i.e., in the area immediately surrounding a point-source of discharge) risk scenarios, designed to be protective of the environment, to determine whether the classification of potential risk should be increased.
ERC uses a weighted approach to minimize the potential for both over and under classification of hazard, exposure and subsequent risk. The balanced approaches for dealing with uncertainties are described in greater detail in ECCC 2016a. The following describes two of the more substantial areas of uncertainty. Error with empirical or modeled acute toxicity values could result in changes in classification of hazard, particularly metrics relying on tissue residue values (i.e., mode of toxic action), many of which are predicted values from QSAR models. However, the impact of this error is mitigated by the fact that overestimation of median lethality will result in a conservative (protective) tissue residue value used for critical body residue (CBR) analysis. Error with underestimation of acute toxicity will be mitigated through the use of other hazard metrics such as structural profiling of mode of action, reactivity and/or estrogen binding affinity. Changes or errors in chemical quantity could result in differences in classification of exposure as the exposure and risk classifications are highly sensitive to emission rate and use quantity. The ERC classifications thus reflect exposure and risk in Canada according to what is believed to be the current use quantity and may not reflect future trends.
Critical data and considerations used to develop the substance-specific profiles for diazenedicarboxamide, and the hazard, exposure and risk classification results, are presented in ECCC (2016b).
On the basis of its low hazard and low exposure potential, according to information considered under ERC, diazenedicarboxamide was classified as having a low potential for ecological risk. It is unlikely that this substance results in concerns for the environment in Canada.
6. Potential to cause harm to human health
6.1 Exposure assessment
There is no reported manufacturing of diazenedicarboxamide in Canada. It is used primarily as a foaming agent during the manufacture of plastics and rubber during which it decomposes almost entirely (OECD 2001). Although releases to the environment are possible during manufacture of diazenedicarboxamide, significant releases of the substance to the environment during foaming of plastics and rubber are not expected (OECD 2001). No measured data regarding concentrations of diazenedicarboxamide in environmental media were identified in Canada, or elsewhere.
In Canada, diazenedicarboxamide (also known as azodicarbonamide) is a permitted food additive, as noted in the List of Permitted Bleaching, Maturing or Dough Conditioning Agents. The substance decomposes relatively quickly when exposed to typical baking conditions (wet flour and/or high temperature treatment); therefore, dietary exposure from use as a permitted food additive is expected to be negligible (email from the Food Directorate, Health Canada to the Existing Substances Risk Assessment Bureau, Health Canada, 23 Jan 2018; unreferenced).
In addition, the majority of flours offered for sale in Canada do not contain diazenedicarboxamide. When used, the substance is primarily added to bread and whole wheat flours which are not as widely used as all-purpose flours. Inhalation exposure during home baking is considered unlikely (email from the Food Directorate, Health Canada to the Existing Substances Risk Assessment Bureau, Health Canada, 29 Mar 2018; unreferenced).
In Canada, there is no expected exposure to diazenedicarboxamide when it is used as a component in food packaging as there is no potential for direct food contact with the food packaging component formulated with the substance (email from the Food Directorate, Health Canada to the Existing Substances Risk Assessment Bureau, Health Canada, 23 Jan 2018; unreferenced).
Although diazenedicarboxamide may be used as a foaming agent to manufacture products to which consumers could be exposed, including floor coverings, components of shoes, exercise mats, and artificial leather; 99.9% of the substance decomposes during manufacturing processes (OECD 2001). During high temperature processing, diazenedicarboxamide decomposes to form gases, primarily nitrogen, carbon
monoxide (together with some carbon dioxide) and ammonia and non-volatile residues such as biurea (ECHA 2012). Environment Agency Austria (2012) conducted a study in which 10 plastic parts were analyzed. Diazenedicarboxamide was detected in only one item (a door seal) at a concentration of 0.083% w/w.
While there is a potential for dermal contact with residual diazenedicarboxamide via contact with foam or rubber products manufactured with this substance, dermal exposure is expected to be minimal. Potential exposure via inhalation is not expected due to the negligible vapour pressure of the substance.
6.2 Health effects assessment
Health effects reviews on diazenedicarboxamide have been prepared by international organisations. These include the OECD SIDS SIAR for Azodicarboxamide (OECD 2001), the World Health Organization (WHO) Concise International Chemical Assessment Document 16 for Azodicarbonamide (WHO 1999), and the Joint FAO/WHO Expert Committee on Food Additives (JECFA 1965). A literature search was conducted from the year prior to the OECD publication date (June 2000). No health effects studies of diazenedicarboxamide were identified that result in lower NOAELs than those presented in OECD (2001). Therefore, the OECD SIAR (2001) is the primary source of hazard information used in this assessment.
Diazenedicarboxamide was not identified as posing a high hazard to human health based on classifications by other national or international agencies for carcinogenicity, genotoxicity, developmental toxicity or reproductive toxicity. Diazenedicarboxamide is on the European Chemicals Agency’s Candidate List of Substances of Very High Concern for Authorisation (ECHA [modified 2018]) based on respiratory sensitisation.
Subchronic (90-day) oral and inhalation studies in rodents showed low toxicity for diazenedicarboxamide. OECD (2001) identified the lowest oral NOAEL as 300 mg/kg bw/day, based on a LOAEL of 1000 mg/kg bw/day (highest dose tested) for observed maternal renal toxicity at the highest dose in rats during a 1-generation reproductive/developmental study of diazenedicarboxamide administered in corn oil via gavage. The same one-generation study (Hatano Research Institute, 2000) showed no treatment effect on reproductive success or on the viability, growth and development of offspring up to the highest oral dose (NOAEL of 1000 mg/kg bw/day). In the 90-day rodent inhalation study (Medinsky et al. 1990), no adverse effects on male reproductive organs or sperm morphology were observed at the highest exposure level (200 mg/m3). Oser et al. (1965) found no tumour development in a 2-year rat study of biurea, the predominant metabolite of diazenedicarboxamide, fed in a diet containing bread that was treated with 0, 750, 2370, or 7500 mg biurea/kg. WHO 1999 identified a NOAEL for the highest dose of the Oser et al. study of 450 mg biurea/kg bw/day. Negative results were reported for multiple in vitro and in vivo genotoxicity assays.
6.3 Characterization of risk to human health
Diazenedicarboxamide is not a naturally occurring substance and is not likely to be released in significant quantities into the Canadian environment. It is also reported to rapidly decompose during manufacturing processes, and when used as a permitted food additive. Inhalation exposure resulting from air-borne diazenedicarboxamide-containing flour during home-baking activities is unlikely. Potential exposure via inhalation is not expected due to the negligible vapour pressure of the substance. As a result, the potential for exposure of the general population to diazenedicarboxamide is expected to be minimal and the potential risk to human health is considered to be low.
6.4 Uncertainties in evaluation of risk to human health
Limited information on the concentration of the substance remaining in products available to consumers was identified as an uncertainty in this assessment of diazenedicarboxamide.
7. Conclusion
Considering all available lines of evidence presented in this draft screening assessment, there is low risk of harm to the environment from diazenedicarboxamide. It is proposed to conclude that diazenedicarboxamide does not meet the criteria under paragraphs 64(a) or (b) of CEPA as it is not entering the environment in a quantity or concentration or under conditions that have or may have an immediate or long-term harmful effect on the environment or its biological diversity or that constitute or may constitute a danger to the environment on which life depends.
On the basis of the information presented in this draft screening assessment, it is proposed to conclude that diazenedicarboxamide does not meet the criteria under paragraph 64(c) of CEPA as it is not entering the environment in a quantity or concentration or under conditions that constitute or may constitute a danger in Canada to human life or health.
Therefore, it is proposed to conclude that diazenedicarboxamide does not meet any of the criteria set out in section 64 of CEPA.
References
Canada. 1999. Canadian Environmental Protection Act, 1999. S.C. 1999, c.33. Canada Gazette Part III, vol. 128, no. 9.
Canada, Dept. of the Environment. 2012. Canadian Environmental Protection Act, 1999: Notice with respect to certain substances on the Domestic Substances List. [PDF] Canada Gazette, Part I, vol. 146, no. 48, Supplement.
ChemIDPlus. 2018. National Library of Medicine, Toxnet Database [database]. [accessed February 2018].
Environment Canada. 2013. DSL Inventory Update data collected under the Canadian Environmental Protection Act, 1999, section 71: Notice with respect to certain substances on the Domestic Substances List. Data prepared by: Environment Canada, Health Canada; Existing Substances Program.
[ECCC] Environment and Climate Change Canada. 2016a. Science Approach Document: Ecological Risk Classification of Organic Substances. Ottawa (ON): Government of Canada.
[ECCC] Environment and Climate Change Canada. 2016b. Data used to create substance-specific hazard and exposure profiles and assign risk classifications in the Ecological Risk Classification of organic substances. Gatineau (QC). Available from: substances@ec.gc.ca.
[ECCC, HC] Environment and Climate Change Canada, Health Canada. [modified 2017 Mar 12]. Categorization. Ottawa (ON): Government of Canada. [accessed 2017 August 9].
[ECHA] European Chemicals Agency. 2012. Annex XV Dossier: Diazene-1,2-dicarboxamide [C,C’ -azodi(formamide)) (ADCA). Proposal for Identification of a Substance as a CMR CAT 1A or 1B, PBT, vPvB or a Substance of an Equivalent Level of Concern. [accessed February 2018].
[ECHA] European Chemicals Agency. 2014. Response to public consultation of inclusion of substances in Annex XIV for Diazene-1,2-dicarboxamide [C,C’ -azodi(formamide)) (ADCA). [PDF] [accessed June 14, 2018].
[ECHA] European Chemicals Agency. [modified Jan 2018]. Candidate List of Substances of Very High Concern for Authorisation [Internet]. Helsinki (FI): European Chemicals Agency. [accessed 2018 Mar 08].
Environment Agency Austria. 2012a. Analyse von Produkten auf Azodicarbonamide und Semicarbazid. Prüfbericht Nr. 1207/0591,unpublished. [as cited in ECHA 2012].
Hatano Research Institute. 2000. Internal Report for the Environmental Health Bureau, The Japanese Ministry of Health and Welfare [as cited in OECD 2001].
[JECFA] Joint FAO/WHO Expert Committee on Food Additives. 1965. Toxicological evaluation of some antimicrobials, antioxidants, emulsifiers, stabilizers, flour-treatment agents, acids, and bases. WHO/Food Add/67.29, nos 17-91 on INCHEM (out of print). [accessed March 2018].
[LNHPD] Licensed Natural Health Products Database [database]. [modified February 2018]. Ottawa (ON): Health Canada. [accessed February 2018].
[MSDS] Material Safety. 2015. DGDB-3485NT. Dow Chemical. [accessed February 2018]
Medinsky, M.A.; Bechtold W.E.; Birnbaum, L.S.; Bond, J.A.; Burt, D.G.; Cheng, Y.S.; Gillett, N.A.; Gulati, D.K.; Hobbs, C.H. Pickrell, J.A. 1990. Effect of inhalated azodicarbonamide on F344/N Rats and B6C3F1 Mice with 2-week and 13-week inhalation exposures. Fundamental and Applied Toxicology 15, 308-319 [as cited in OECD 2001].
[OECD] Organisation for Economic Cooperation and Development. 2001. SIDS Initial Assessment Report: Azodicarboxamide. CAS No. 123-77-3. SIAM [SIDS Initial Assessment Meeting] 12; 2001; Paris, France. [Accessed February 2018].
Oser, B.L.; Oser, M. and Morgareidge, K. 1965. Studies of the safety of azodicarbonamide as a flour-maturing agent. Toxicol. Appl. Pharmacol. 7, 445-472 [as cited in OECD 2001].
PubChem [database]. 2004. Bethesdsa (MD): US National Library of Medicine, National Center for Biotechnology Information. Search results for Azodicarboxamide. [updated March 2018; accessed March 2018].
Schipper, P.S.; Black, J. and Dymek, T. 1996. Foamed rigid vinyl for building products. [PDF] Journal of Vinyl & Additive Technology. 2, 304-309.
[US EPA] US Environmental Protection Agency. 2018. Chemical Reporting Database. [Accessed February 2018].
[WHO] World Health Organization. 1999. Concise International Chemical Assessment Document 16 for Azodicarbonamide. Geneva, International Programme on Chemical Safety.
