Draft Screening Assessment Sodium ortho-phenylphenate (SOPP)
Official title: Draft Screening Assessment [1,1’-Biphenyl]-2-ol, sodium salt (Sodium ortho-phenylphenate; SOPP)
Chemical Abstracts Service Registry Number: 132-27-4
Environment and Climate Change Canada
Health Canada
February 2020
Synopsis
Pursuant to section 68 of the Canadian Environmental Protection Act, 1999 (CEPA), the Minister of the Environment and the Minister of Health have conducted a screening assessment of [1,1’-biphenyl]-2-ol, sodium salt, hereinafter referred to as sodium ortho-phenylphenate (SOPP). The Chemical Abstracts Service Registry Number (CAS RNFootnote 1) for SOPP is 132-27-4. This substance was identified as a priority for assessment on the basis of other human health concerns identified in 2006.
SOPP does not occur naturally in the environment. According to information submitted pursuant to a CEPA section 71 survey, the manufactured quantity reported in Canada in 2008 was in a range of 10 000 to 100 000 kg, and the import quantities were reported in a range of 1 000 to 10 000 kg.
SOPP is a material preservative agent. In Canada, SOPP is used in building or construction materials and in products available to consumers (such as tire and rubber lubricants), and it may be used as a component in food packaging materials and incidental additives or as a non-medicinal ingredient in natural health products and non-prescription drugs. It is also an active ingredient and a formulant in registered pest control products in Canada.
The ecological risk of SOPP was characterized using the ecological risk classification of organic substances (ERC), which is a risk-based approach that employs multiple metrics for both hazard and exposure, with weighted consideration of multiple lines of evidence for determining risk classification. Hazard profiles are based principally on metrics regarding mode of toxic action, chemical reactivity, food web-derived internal toxicity thresholds, bioavailability, and chemical and biological activity. Metrics considered in the exposure profiles include potential emission rate, overall persistence, and long-range transport potential. A risk matrix is used to assign a low, moderate or high level of potential concern for substances on the basis of their hazard and exposure profiles. Based on the outcome of the ERC analysis, SOPP is considered unlikely to be causing ecological harm.
Considering all available lines of evidence presented in this draft screening assessment, there is low risk of harm to the environment from SOPP. It is proposed to conclude that SOPP does not meet the criteria under paragraphs 64(a) or (b) of CEPA as it is not entering the environment in a quantity or concentration or under conditions that have or may have an immediate or long-term harmful effect on the environment or its biological diversity or that constitute or may constitute a danger to the environment on which life depends.
SOPP has been reviewed internationally through the International Agency for Research on Cancer, the FAO/WHO Joint Meeting on Pesticide Residues, the United States Environmental Protection Agency and the California Environmental Protection Agency. In 2008 SOPP was evaluated by Health Canada’s Pest Management Regulatory Agency. In laboratory studies, SOPP was found to be associated with an increased incidence of urinary bladder tumours. At lower doses, effects for SOPP and a structurally-related substance included decreased body weight gain and kidney effects.
The predominant source of exposure to SOPP from products available to consumers in Canada is from tire and rubber lubricants. There is also potential for exposure to SOPP to the general population from food, its use in food packaging materials and other products available to consumers.
A comparison of the estimate of exposure to SOPP from the use of tire and rubber lubricant with a critical effect level identified from laboratory studies, results in margin of exposure which is considered adequate to address uncertainties in the health effects and exposure databases. The risk to human health from exposure to SOPP from food packaging is considered to be very low and contribute negligibly to the overall dietary exposure of Canadians to SOPP.
On the basis of the information presented in this draft screening assessment, it is proposed to conclude that SOPP does not meet the criteria under paragraph 64(c) of CEPA as it is not entering the environment in a quantity or concentration or under conditions that constitute or may constitute a danger in Canada to human life or health.
It is therefore proposed to conclude that SOPP does not meet any of the criteria set out in section 64 of CEPA.
1. Introduction
Pursuant to section 68 of the Canadian Environmental Protection Act, 1999 (CEPA) (Canada 1999), the Minister of the Environment and the Minister of Health have conducted a screening assessment of [1,1’-Biphenyl]-2-ol, sodium salt, hereinafter referred to as sodium ortho-phenylphenate (SOPP), to determine whether this substance presents or may present a risk to the environment or to human health. This substance was considered a priority on the basis of other human health concerns (ECCC, HC [modified 2017]).
The substance currently being evaluated has been reviewed internationally by the International Agency for Research on Cancer Monographs Programme (IARC 1999), the FAO/WHO Joint Meeting on Pesticide Residues (JMPR 1999), the United States Environmental Protection Agency (EPA) (2006), and the California EPA (Cal EPA 2007). SOPP was also reviewed by Health Canada’s Pest Management Regulatory Agency (Health Canada 2008a,b). These assessments undergo rigorous review and were used to inform the health effects characterization in this screening assessment.
The ecological risk of SOPP was characterized using the ecological risk classification of organic substances (ERC) approach (ECCC 2016a). The ERC describes the hazard of a substance using key metrics, including mode of toxic action, chemical reactivity, food web-derived internal toxicity thresholds, bioavailability, and chemical and biological activity, and considers the possible exposure of organisms in the aquatic and terrestrial environments on the basis of such factors as potential emission rates, overall persistence, and long-range transport potential in air. The various lines of evidence are combined to identify substances as warranting further evaluation of their potential to cause harm to the environment or as having a low likelihood of causing harm to the environment.
This draft screening assessment includes consideration of information on chemical properties, environmental fate, hazards, uses and exposures, including additional information submitted by stakeholders. Relevant data for SOPP were identified up to January 2019. Empirical data from key studies as well as results from models were used to reach proposed conclusions. When available and relevant, information presented in assessments from other jurisdictions was considered.
This draft screening assessment was prepared by staff in the CEPA Risk Assessment Program at Health Canada and Environment and Climate Change Canada and incorporates input from other programs within these departments. The ecological portion of this assessment is based on the ERC document (published July 30, 2016), which was subject to an external review as well as a 60-day public comment period.
This draft screening assessment focuses on information critical to determining whether substances meet the criteria as set out in section 64 of CEPA by examining scientific information and incorporating a weight of evidence approach and precautionFootnote 2. This draft screening assessment presents the critical information and considerations on which the proposed conclusions are based.
2. Identity of substance
The Chemical Abstracts Service Registry Numbers (CAS RN), Domestic Substances List (DSL) name, and molecular structure for SOPP are presented in Table 2‑1.
| CAS RN | DSL name (common name; abbreviation) | Molecular structure and formula | Molecular weight (g/mol) |
|---|---|---|---|
| 132-27-4 | [1,1'-Biphenyl]-2-ol sodium salt (sodium ortho-phenylphenate; SOPP) | 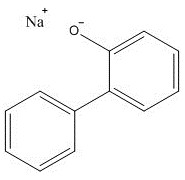 C12H10O.Na C12H10O.Na
|
192.19 |
2.1 Selection of analogue
A read-across approach using data from an analogue was used to inform the human health assessment. An analogue was selected that was structurally and/or functionally similar to the substance in this assessment (similar physical-chemical properties, toxicokinetics), and that had relevant empirical data that could be used to read-across to limited empirical data for the substance in this assessment. SOPP was found to be associated with limited empirical toxicity data. SOPP is the sodium salt of orthophenylphenol (OPP) and results from toxicity studies conducted with OPP were used in a read-across approach to inform the health effects assessment of SOPP. Identity information on OPP is presented in Table 2‑2. Physical and chemical properties and the read-across summary of toxicological data on ortho-phenylphenol (OPP) and SOPP can be found in Appendix A.
| CAS RN | DSL name (common name; abbreviation) | Chemical structure and molecular formula | Molecular weight (g/mol) |
|---|---|---|---|
| 90-43-7 | [1,1'-Biphenyl]-2-ol (Ortho-phenylphenate; OPP) |
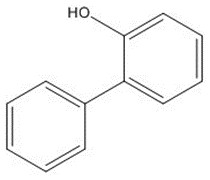 C12H10O C12H10O
|
170.2 |
3. Physical and chemical properties
A summary of chemical properties of SOPP are presented in Table 3-1. When experimental information was limited or not available for a property, data from analogues were used for read-across and/or (Q)SAR models were used to generate predicted values for the substance. Additional physical and chemical properties are reported in ECCC (2016b).
| Properties | SOPP | OPP | Reference |
|---|---|---|---|
| Vapour pressure (Pa) | 1.2 | 0.27 | ECHA c2007-2019 and HSDB 1983- |
| Water solubility (mg/L) | 1.00E+06 (pH 13.6) | 530 to 640 (pH 5-9) | ECHA c2007-2019 |
| log KOW | 2.95 | 3.18 | ECHA c2007-2019 |
| Half-life in air (hours) | - | 14 | US EPA 2006 |
Abbreviation: Kow, octanol-water partition coefficient.
4. Sources and uses
SOPP does not occur naturally in the environment (IARC 1999).
SOPP was included in a survey issued pursuant to section 71 of CEPA (Canada 2009). In 2008, SOPP was reported to be manufactured (10 000 to 100 000 kg) and imported (1 000 to 10 000 kg) into Canada (Environment Canada 2009).
On the basis of information submitted pursuant to Section 71 of CEPA (Environment Canada 2009), SOPP was reported to be used commercially in building or construction materials, with no consumer uses reported. It is used as a material preservative agent (Health Canada 2008a). Additional uses identified in Canada are presented in Table 4-1.
| Use | SOPP |
|---|---|
| Incidental additivea | Y |
| Food packaging materialsa | Y |
| Internal Drug Product Database as medicinal or non-medicinal ingredients in final Pharmaceutical, Disinfectant or Veterinary drug products in Canadab | Y |
| Natural Health Products Ingredients Databasec | Y |
| Licensed Natural Health Products Database as medicinal or non-medicinal ingredients in natural health products in Canadac | Y |
| Formulant and active ingredient in pest control products registered in Canadad | Y |
| Notified to be present in cosmetics under the Cosmetic Regulationse,f | N |
Abbreviation: Y, Yes, use was reported for this substance; N, No, use was not reported for this substance.
a Personal communication, e-mails from the Food Directorate (FD), Health Canada (HC) to the Existing Substances Risk Assessment Bureau (ESRAB), HC, dated Aug 2018; unreferenced.
b Personal communication, e-mails from the Therapeutic Products Directorate (TPD), HC to the ESRAB, HC, dated Aug 2018; unreferenced.
c Personal communication, e-mails from the Natural and Non-prescription Health Products Directorate (NNHPD), HC to the ESRAB, HC, dated Aug 2018; unreferenced.
d Personal communication, e-mails from the Pest Management Regulatory Agency (PMRA), HC to the ESRAB, HC, dated Aug 2018; unreferenced.
e Personal communication, emails from Consumer Product Safety Directorate (CPSD), HC to ESRAB, HC, dated August, 2018 and March 2019; unreferenced.
f Internationally, SOPP has been reported to be used as a preservative in cosmetics (SCCS 2015, 2018)
According to publicly available information and product material safety data sheets (MSDSs), SOPP was identified in tire and rubber lubricants available to consumers in Canada (MSDS 2015). It has also been reported to be used elsewhere as an intermediate for dyes, resins and rubber chemicals, as a germicide, as a preservative, a household disinfectant in dishwashing formulations and in food packaging (NTP 1991).
5. Potential to cause ecological harm
5.1 Characterization of ecological risk
The ecological risk of SOPP was characterized using the ecological risk classification of organic substances (ERC) approach (ECCC 2016a). The ERC is a risk-based approach that considers multiple metrics for both hazard and exposure, on the basis of weighted consideration of multiple lines of evidence for determining risk classification. The various lines of evidence are combined to discriminate between substances of lower or higher potency and lower or higher potential for exposure in various media. This approach reduces the overall uncertainty with risk characterization compared to an approach that relies on a single metric in a single medium (e.g., median lethal concentration [LC50]) for characterization. The following summarizes the approach, which is described in detail in ECCC (2016a).
Data on physical-chemical properties, fate (chemical half-lives in various media and biota, partition coefficients, and fish bioconcentration), acute fish ecotoxicity, and chemical import or manufacture volume in Canada were collected from the scientific literature, available empirical databases (e.g., OECD QSAR Toolbox 2016), and from responses to surveys conducted under section 71 of CEPA, or they were generated using selected (quantitative) structure-activity relationship ([Q]SAR) or mass-balance fate and bioaccumulation models. These data were used as inputs to other mass-balance models or to complete the substance hazard and exposure profiles.
Hazard profiles were based principally on metrics regarding mode of toxic action, chemical reactivity, food web-derived internal toxicity thresholds, bioavailability, and chemical and biological activity. Exposure profiles were also based on multiple metrics, including potential emission rate, overall persistence, and long-range transport potential. Hazard and exposure profiles were compared to decision criteria in order to classify the hazard and exposure potentials for each organic substance as low, moderate, or high. Additional rules were applied (e.g., classification consistency, margin of exposure) to refine the preliminary classifications of hazard or exposure.
A risk matrix was used to assign a low, moderate or high classification of potential risk for each substance on the basis of its hazard and exposure classifications. ERC classifications of potential risk were verified using a two-step approach. The first step adjusted the risk classification outcomes from moderate or high to low for substances that had a low estimated rate of emission to water after wastewater treatment, representing a low potential for exposure. The second step reviewed low risk potential classification outcomes using relatively conservative, local-scale (i.e., in the area immediately surrounding a point-source of discharge) risk scenarios, designed to be protective of the environment, to determine whether the classification of potential risk should be increased.
ERC uses a weighted approach to minimize the potential for both over and under classification of hazard and exposure and subsequent risk. The balanced approaches for dealing with uncertainties are described in greater detail in ECCC (2016a). The following describes two of the more substantial areas of uncertainty. Error with empirical or modelled acute toxicity values could result in changes in classification of hazard, particularly metrics relying on tissue residue values (i.e., mode of toxic action), many of which are predicted values from (Q)SAR models (OECD QSAR Toolbox 2016). However, the impact of this error is mitigated by the fact that overestimation of median lethality will result in a conservative (protective) tissue residue value used for critical body residue (CBR) analysis. Error with underestimation of acute toxicity will be mitigated through the use of other hazard metrics such as structural profiling of mode of action, reactivity and/or estrogen binding affinity. Changes or errors in chemical quantity could result in differences in classification of exposure as the exposure and risk classifications are highly sensitive to emission rate and use quantity. The ERC classifications thus reflect exposure and risk in Canada on the basis of what is calculated to be the current use quantity, and may not reflect future trends.
Critical data and considerations used to develop the substance-specific profile for SOPP, and the hazard, exposure and risk classification results, are presented in ECCC (2016b).
On the basis of low hazard and low exposure classifications according to information considered under ERC, SOPP was classified as having a low potential for ecological risk. It is unlikely that this substance is resulting in concerns for the environment in Canada.
6. Potential to cause harm to human health
6.1 Exposure assessment
Environmental media and food
SOPP does not occur naturally in the environment. No data have been identified for SOPP in relevant environmental media in Canada or elsewhere. The dissociation constant (pKa) of OPP indicates that SOPP is unstable in the environment and dissociates to form its conjugate acid OPP, and sodium hydroxide in water (ECHA c2007-2019). The dissociation to OPP is the predominant degradation pathway for SOPP, which is not expected to persist in the environment (ECHA c2007-2019). OPP is reported to be photolytically unstable in a neutral aqueous medium and to degrade completely in 14 days when exposed to sunlight (Health Canada 2008a; US EPA 2006). In addition, due to its physical and chemical properties (Table 3-1), SOPP is not expected to volatilize (US EPA 2014), is immobile in soils and is not likely to contaminate groundwater or surface water (Health Canada 2008a; US EPA 2006). Therefore, on the basis of the available information, it is expected that exposure of the general population to SOPP via water, air or soil is negligible.
SOPP may be used as a component in food packaging materials with the potential for direct food contact. The probable daily intake for SOPP from its use in food packaging materials is estimated to be 6.2 ng/kg bw/day for an adult aged 19 years and older. It may also be used as a component in incidental additives (e.g. cleaners, lubricants) used in food processing establishments. Exposure from its use in incidental additives is not expected, since there is no direct food contact with the incidental additive (Personal communication from the FD, HC to the ESRAB, HC, dated August 2018, unreferenced).
SOPP may be present as a residue on food from its use as a pest control product in Canada (Health Canada 2008a). The US EPA (2006) reported the highest estimated dietary exposure of SOPP for the U.S. population to be 0.021 mg/kg/day for children (aged 1 to 2 years). The US EPA assessment was considered to be applicable to the Canadian situation as stated in Heath Canada (2008a). In consideration of the contribution of the estimated dietary exposure to SOPP from its presence as pesticide residue in food, the estimated exposure to SOPP of the Canadian population from food packaging materials is considered to contribute negligibly to the overall dietary exposure to SOPP.
Products available to consumers
One product available to consumers that could reasonably be expected to be used by the general public was tire/rubber lubricant. The primary route of exposure is expected to be dermal as it is considered that these products are used outdoors and/or in a garage. Inhalation is expected to contribute a very small fraction of the overall exposure because of the high rate of air flow, the viscous matrices of these products, and the low vapour pressure of SOPP. Estimates for dermal exposure are presented in Table 6-1. A dermal absorption factor of 100% was assumed. Details are presented in Appendix B (Table B-1). Additional potential use scenarios for SOPP were considered (non-medicinal ingredient in natural health products, non-medicinal ingredient in non-prescription drugs) but resulted in lower exposure estimates than those presented in Table 6-1.
| Product scenario | Concentration (%) | Route of exposure | Per event exposure (systemic)a |
|---|---|---|---|
| Tire and rubber lubricant (19+ years) | 0.2b | Dermal | 0.02 mg/kg bw |
a Systemic exposure assuming 100% absorption through the dermal route.
b MSDS (2015).
6.2 Health effects assessment
OPP and its sodium salt (SOPP) were reviewed by the FAO/WHO JMPR (1999), the US EPA (US EPA 2006), the California EPA (Cal EPA 2007), and Health Canada’s Pest Management Regulatory Agency (Health Canada 2008a,b). IARC classified SOPP as possibly carcinogenic to humans (Group 2B) (IARC 1999). The US EPA and Health Canada reviews were used as primary sources to inform the health effects characterization in this screening assessment, and supplemented with information from the other reviews mentioned above. The US EPA has also published a work plan for OPP and its salts (2014), with additional details on studies. OPP and it sodium salt were also reviewed by Australia’s National Industrial Chemicals Notification and Assessment Scheme (NICNAS) (2016) and the European Commission Scientific Committee on Consumer Safety (SCCS 2015, 2018). The European Chemicals Agency (ECHA) has a registration dossier available for SOPP (ECHA c2007-2019). A literature search was conducted from January 1998 to January 2019 to account for international reviews which focused on effects based on uses in addition to pesticide uses (i.e., IARC 1999). No studies which could result in a different health effects characterization were found.
Following ingestion of a single gavage dose of SOPP (250 to 500 mg/kg bw), approximately 85% was found to be absorbed in rats and excreted via urine within 24 hours. Less than 8% of it was detected in tissue (including adipose, liver, kidneys, urinary bladder, stomach and intestine, brain and blood) at 24 hours and less than 1% detected at 7 days after dosing (IARC 1999; JMPR1999; Cal EPA 2007). It is rapidly excreted through the urine, but up to 26% and 4% of an oral dose in rats may also be excreted in bile and feces, respectively (Sato et al. 1988 cited in Cal EPA 2007). In mammals (including rodents and humans), orally absorbed SOPP produced metabolites such as sulfate and glucuronide conjugates of OPP, unconjugated phenylhydroquinone (PHQ), phenylbenzoquinone (PBQ), and 2,5-dihydroxybiphenyl (IARC 1999; JMPR 1999). In rats, the urinary concentrations of metabolites were greater in males than females (Nakao et al. 1983 and Morimoto et al. 1989 cited in IARC 1999 and JMPR 1999).
F344 rats (10/sex/group) were administered SOPP in the diet at 0, 1250, 5000, 10 000, 20 000, or 40 000 ppm (equal to 0, 86, 180, 350, 700, 1350 or 2450 mg/kg bw/day, respectively) for 13 weeks. Body weight gains decreased 15% to 17% in both sexes at 350 mg/kg bw/day and higher (Iguchi et al. 1979 cited in JMPR 1999). In males at 700 mg/kg bw/day and higher, there were decreased alanine aminotransferase (ALT) and aspartate aminotransferase (AST) activities, with increased relative liver weights observed at 1350 mg/kg bw/day and higher. Urinary bladder epithelium tumours (transitional papillomas and carcinomas in males, papillomas in females) were increased at 2450/1350 mg/kg bw/day and higher in males/females, with kidney inflammation (pyelonephritis) in both sexes at 2450 mg/kg bw/day and dose-related increase in urinary alkalinity (Iguchi et al. 1979 and Hiraga and Fujii 1981 cited in JMPR 1999, Cal EPA 2007 and SCCS 2015). In this assessment the no observed adverse effect level (NOAEL) is considered to be 180 mg/kg bw/day based on decreased body weight gain at 350 mg/kg bw/day and higher, in alignment with the JMPR (1999).
F344 rats were administered SOPP via diet in two carcinogenicity studies. In the first study, animals (50/sex/group) were treated at 0, 7000 or 20 000 ppm SOPP in diet for males (equivalent to 0, 270 or 770 mg/kg bw/day, respectively) and 0, 5000 or 10 000 ppm in diet for females (equivalent to 0, 224 or 466 mg/kg bw/day, respectively) for 104 weeks with a 2-week recovery period. In the second study, F344 rats (25/sex/group) were administered SOPP as above, with an additional lower 2500 ppm dose group (equivalent to 95/113 mg/kg bw/day males/females) and a 56-week recovery period (until all animals had died) (Fujii and Hiraga 1985 cited in IARC 1999 and Hiraga 1983 cited in Cal EPA 2007). The incidences of interstitial nephritis of the kidney were increased in both sexes at 224/270 mg/kg bw/day and higher in the first study and at 270/466 mg/kg bw/day and higher in the second study (males/females). There was a dose-related increase in focal atrophy of the pancreas in females at 224 mg/kg bw/day and above in the first study only. Urinary bladder papillomas and/or carcinomas were observed in both sexes in both studies only at 224/270 mg/kg bw/day and above. In both studies, body weights were decreased in females at 466 mg/kg bw/day and in males at 770 mg/kg bw/day, although body weight data were only available in summary form in the first study, and no individual data were available in the second study (Cal EPA 2007). The Cal EPA (2007) identified a lowest observed effect level at the lowest tested dose of 224 mg/kg bw/day based on increased incidences of interstitial nephritis in both sexes and studies, and increased incidences of pancreatic focal atrophy in females in the first study. The Cal EPA (2007) stated that the carcinogenicity component of this study was acceptable but not the chronic toxicity component due to insufficient data on hematology and ophthalmology.
Mice were less sensitive than rats to dietary doses of SOPP in both a 13 week and a chronic/carcinogenicity study. In a 13-week study, a NOAEL of 730/1021 mg/kg bw/day (males/females) was identified based on decreased body weight gain in males at 1581 mg/kg bw/day and higher, and increased relative liver weights in females at 1926 mg/kg bw/day and higher (Shibata et al. 1985 cited in Cal EPA 2007 and SCCS 2015, JMPR 1999). In a chronic/carcinogenicity study, at the lowest observed adverse effect level (LOAEL) of 480 mg/kg bw/day there was decreased body weight, increased alkaline phosphatase (ALP) activity, and decreased urine specific gravity in females (Hagiwara et al. 1984 cited in IARC 1999; Ito et al. 1983 cited in Cal EPA 2007). Although there were increased liver tumours in both sexes at 3009 mg/kg bw/day, no urinary bladder tumours were observed.
SOPP was overall not genotoxic in vitro but the data was mixed in vivo (reviewed in IARC 1999; Cal EPA 2007). While there were negative in vivo studies (including chromosome aberration, dominant lethal and Comet assays), cell transformation and DNA breaks were observed in the urinary bladder in male rats fed SOPP in the diet at 500 mg/kg bw/day and above for 1 week ) or 3 to 5 months, respectively. DNA breaks or adducts were also observed in mice orally and dermally exposed to single doses of 330 mg/kg bw and higher (IARC 1999; Sasaki et al. 2002; Cal EPA 2007; De Boeck et al. 2015).
The mode of action for urinary bladder carcinogenicity from SOPP is unclear. At high doses there could be saturation of phase II detoxification pathways, leading to increased levels of the metabolites PHQ or PBQ, which may induce carcinogenicity via non-genotoxic regenerative hyperplasia of the bladder, or by possible genotoxic mechanisms (IARC 1999; Cal EPA 2007; US CDC 2017). An alternative mechanism proposed by the Cal EPA (2007), is that there could be DNA damage mediated by reactive oxygen species during the conversion of PHQ to PBQ, and cell proliferation from chemically-induced cytotoxicity in the urothelium of the bladder.
The IARC has classified SOPP as possibly carcinogenic to humans (Group 2B) (IARC 1999). The US EPA (2006) and Health Canada (2008a) identified OPP and its salts as not likely to be carcinogenic below a specific dose range, without quantification of risk. They were considered “not likely to be carcinogenic to humans” below 200 mg/kg bw/day but “likely to be carcinogenic to humans” above 200 mg/kg bw/day (US EPA 2006).
In a developmental toxicity study, Jcl:ICR mice (20 pregnant females/group) were gavaged with SOPP in water at 0, 100, 200 or 400 mg/kg bw/day from gestation days (GDs) 7 to 15 and sacrificed on GD 18 (Ogata et al. 1978b cited in Cal EPA 2007 and Kwok and Silva 2013). At 100 mg/kg bw/day and higher, there were decreased fetal body weights in both sexes and increased litter incidences of cleft palate in fetuses and additional skeletal variations (Kwok and Silva 2013) in the presence of decreased maternal body weight gain. At 200 mg/kg bw/day there was increased maternal death with vaginal bleeding in decedent animals. The Cal EPA (2007) did not determine a maternal LOAEL because there were insufficient data to unambiguously distinguish the extent of decreased maternal body weight gain at 100 mg/kg bw/day (uterine weights not measured). Although a developmental LOAEL was identified at 100 mg/kg bw/day by the Cal EPA (2007), this study was considered unacceptable due to significant limitations in reporting, including lack of individual data (Cal EPA (2007).
Ortho-phenylphenol (OPP)
This section focuses on the reproductive, developmental, chronic toxicity/carcinogenicity and genotoxicity studies for OPP. SOPP is the sodium salt of OPP which has similar physical-chemical properties. SOPP and OPP are in a pH dependent equilibrium in aqueous solution (JMPR 1999; US EPA 2014) and the oral toxicokinetics are similar in mammalian species (JMPR 1999; Cal EPA 2007; NICNAS 2016), although there is increased urine alkalinity with SOPP (Appendix A).
Effects on the mammalian estrogen, androgen and thyroid pathways and immunotoxicity in mammals are not expected for OPP, so are not discussed further in this assessment (reviewed by the US EPA in 2015 and 2016, respectively). IARC (1999) classified OPP as Group 3 (not classifiable as to its carcinogenicity to humans). IARC (1999) stated that SOPP and OPP induce urinary bladder tumours mostly in male rats, with SOPP being more potent.
In a chronic/carcinogenicity study, mice (50/sex/group) were administered doses of OPP at 250 to 1000 mg/kg bw/day in diet for 2 years. A LOAEL of 250 mg/kg bw/day was identified by the US EPA (2014) based on increased absolute and relative liver weights and decreased absolute and relative spleen weights in both sexes, as well as increased liver masses or nodules at 500 mg/kg bw/day and higher) (Quast and McGuirk 1995 cited in JMPR 1999 and US EPA 2006, 2014). The Cal EPA (2007) also identified a LOAEL of 250 mg/kg bw/day from this study.
In a chronic toxicity/carcinogenicity study, F344 rats (46 to 50/sex/dose) were fed OPP in the diet at doses of 0, 800, 4000 or 8000/10 000 ppm (equal 0, 39/49, 200/248 or 402/648 mg/kg bw/day in males/females, respectively) for 2 years, with an interim sacrifice at 1 year (20/sex/group in the control and high dose groups and 10/sex/group at low and mid dose groups) (Wahle and Christenson 1996 cited in Cal EPA 2007 and US EPA 2014). The US EPA (2006, 2014) identified a NOAEL of 39 mg/kg bw/day based on decreased body weight, body weight gain, food consumption and food efficiency, increased clinical signs and gross pathological signs of toxicity at 200 mg OPP/kg bw/day and higher. The Cal EPA (2007) derived a benchmark dose (BMD10) of 222.8 mg/kg bw/day with 95% lower limit (BMDL10) of 185.2 mg/kg bw/day, based on the combined incidences of urinary bladder papillomas and carcinomas in male rats.
Genotoxicity studies conducted with OPP showed mixed results in vitro and in vivo (Cal EPA 2007; SCCS 2015). In contrast with the Cal EPA (2007), the US EPA (2006, 2014) considered that OPP was not genotoxic at doses which did not result in cytotoxicity. The mode of action of OPP resulting in increased urinary bladder tumours is unclear but proposed possibilities are those previously described for SOPP (IARC 1999; Cal EPA 2007; US CDC 2017).
In a two-generation reproductive toxicity study, Sprague Dawley (SD) rats (30/sex/group) received 0, 20, 100 or 500 mg/kg bw/day in the diet and there were two matings/generation to produce litters in the F1 and F2 generations (Eigenberg and Lake 1995 cited in Bomhard et al. 2002, Kwok and Silva 2013 and ECHA c2007-2019; US EPA 2006, 2014). The parental toxicity NOAEL was 100 mg/kg bw/day based on decreased body weights and body weight gain in both sexes, with kidney and urinary bladder related effects in males (including chronic inflammation in both and hyperplasia of ureters and urinary bladder), mortality (due to kidney failure in an adult male), and decreased body weight in 21-day old pups of both F1 and F2 generations at 500 mg/kg bw/day. The US EPA (2014) stated that the decreased pup weight was not due to lactational effects in the dams, but considered to be related to consumption of the treated food by pups. No reproductive adverse effects were noted in this study.
There were three developmental toxicity studies in mice, rats, and rabbits gavaged with OPP. In the rat developmental study (24 to 36 pregnant animals/group gavaged with OPP in cottonseed oil during GDs 6 to 15 and sacrificed on GD 21), the US EPA (2006, 2014) and Health Canada (2008a) selected a maternal NOAEL of 100 mg/kg bw/day for OPP based on decreased body weight gain, food consumption and food efficiency at 300 mg/kg bw/day OPP (John et al. 1978 cited in US EPA 2006, 2014 and Cal EPA 2007; Health Canada 2008a). The US EPA (2006, 2014) and Health Canada considered the rabbit developmental toxicity study as a co-critical developmental study, with a maternal NOAEL of 100 mg/kg bw/day based on effects in the kidneys (inflammation and tubular degeneration) at 250 mg/kg bw/day OPP (1 to 24 pregnant animals/group gavaged with OPP in corn oil during GDs 7 to 19 and sacrificed on GD 28) with no adverse fetal effects (Zablotny et al. 1991 cited in US EPA 2006 and Cal EPA 2007; Health Canada 2008a). There were no fetal effects in either of these studies up to 700 mg OPP/kg bw/day in rodents. In contrast, mice (21 pregnant animals/group gavaged with OPP in olive oil during GDs 7 to15 and sacrificed on GD 18) had severe fetal effects (including open eyelids, cleft palate, exencephalia) at the lowest tested dose of 1450 mg/kg bw/day (LOAEL), in the presence of maternal deaths (0, 4, 7 and 16 out of 21/group at 0, 1450, 1740 and 2100 mg/kg bw/day, respectively) (Ogata et al. 1978b cited in Cal EPA 2007 and Kwok and Silva 2013). The Cal EPA (2007) considered the developmental study in mice unacceptable because of doses selected which resulted in many maternal deaths and inadequate numbers of fetuses/group available for visceral and skeletal examinations.
The results of a 3-week dermal toxicity study in rats (Cal EPA 2007; US EPA 2006, 2014), were not considered in this assessment because comparison of physical-chemical properties between OPP and SOPP suggests that OPP has a lower dermal absorption, and SOPP is more dermally corrosive (SCCS 2015).
6.3 Characterization of risk to human health
Table 6-2 provides the relevant exposure estimate and critical effect level as well as the resultant MOE for the characterization of risk to human health from exposure to SOPP.
| Exposure scenario (age group with highest estimate) | Systemic exposurea | Critical effect level | Critical health effect endpoint | MOE |
|---|---|---|---|---|
| Per event dermal exposure from use of tire and rubber lubricant (19+ years) | 0.02 mg/kg bw | Oral NOAEL (maternal) = 100 mg /kg bw/day in developmental toxicity studies in rats and rabbits. | Maternal NOAEL of 100 mg /kg bw/day based on decreased body weight gain, food consumption and food efficiency at 300 mg /kg bw/day in rats and kidney effects in rabbits at 250 mg/kg bw/day (OPP, analogue). | 5000 |
Abbreviations: MOE, Margin of Exposure; NOAEL, no observed adverse effect level.
a Assumes 100% absorption through the oral and dermal routes.
A developmental toxicity study in mice conducted with SOPP (Ogata et al. 1978b) suggested potential for developmental effects that may occur in the presence of maternal toxicity, but given significant limitations in the study (including lack of individual data), it was not selected for use in risk characterization of per event dermal exposure to SOPP during use of a lubricant. Instead, consistent with an earlier Health Canada evaluation (Health Canada 2008a) and other international reviews (US EPA 2006), a NOAEL of 100 mg/kg bw/day was selected from developmental toxicity studies in rats and rabbits gavaged with the analogue OPP, for incidental oral short-term exposure to SOPP. This was based on maternal effects observed at higher doses in the absence of developmental toxicity. The MOE shown in Table 6-2 is considered adequate to address uncertainties in the health effects and exposure databases for SOPP.
The human health risk from chronic dietary exposure to pesticide food residues was addressed by Health Canada (2008a) in the re-evaluation of pesticidal uses of SOPP. This exposure was compared to an ADI of 0.39 mg/kg bw/day (based on the NOAEL of 39 mg/kg bw/day identified by the US EPA (2006)) and was found to be associated with an acceptable human health risk. The use of SOPP in food packaging material is considered to contribute negligibly to the overall dietary exposure of the general population to SOPP, and the risks to human health from exposure to SOPP from food packaging is considered to be very low.
While exposure of the general population to SOPP is not of concern at current levels, this substance is considered to have a health effect of concern on the basis of its potential hazard, due to its classification as an IARC Group 2B substance (possibly carcinogenic to humans) (IARC 1999).
6.4 Uncertainties in evaluation of risk to human health
The key sources of uncertainty are presented in the table below.
| Key source of uncertainty | Impact |
|---|---|
| No measured Canadian data in relevant environmental media for SOPP. | +/- |
| No dermal absorption data for SOPP. | + |
| No reproductive toxicity study or adequate developmental study by any route of exposure for SOPP. | +/- |
+/- = uncertainty with potential to cause over or under-estimation of risk; + = uncertainty with potential to cause over-estimation of risk
7. Conclusion
Considering all available lines of evidence presented in this draft screening assessment, there is low risk of harm to the environment from SOPP. It is proposed to conclude that SOPP does not meet the criteria under paragraphs 64(a) or (b) of CEPA as it is not entering the environment in a quantity or concentration or under conditions that have or may have an immediate or long-term harmful effect on the environment or its biological diversity or that constitute or may constitute a danger to the environment on which life depends.
On the basis of the information presented in this draft screening assessment, it is proposed to conclude that SOPP does not meet the criteria under paragraph 64(c) of CEPA as it is not entering the environment in a quantity or concentration or under conditions that constitute or may constitute a danger in Canada to human life or health.
It is therefore proposed to conclude that SOPP does not meet any of the criteria set out in section 64 of CEPA.
References
Bomhard EM, Brendler-Schwaab SY, Freyberger A, Herbold BA, Leser KH, Richter M. 2002. O-phenylphenol and its sodium and potassium salts: A toxicological assessment. Critical Reviews in Toxicology 32(6):551-626.
Brusick D. 2005. Analysis of genotoxicity and the carcinogenic mode of action for ortho-phenylphenol. Environmental and molecular mutagenesis 45:460-481.
Canada. 1999. Canadian Environmental Protection Act, 1999. S.C. 1999, c.33. Canada Gazette Part III, vol. 22, no. 3.
Canada, Dept. of the Environment. 2009. Canadian Environmental Protection Act, 1999: Notice with respect to certain inanimate substances (chemicals) on the Domestic Substances List [PDF]. Canada Gazette, Part I, vol. 143, no. 40, p. 2945-2956.
[Cal EPA] California Environmental Protection Agency. 2007. Ortho-phenylphenol (OPP) and sodium ortho-phenylphenate (SOPP) risk characterization document: Dietary exposure. Department of Pesticide Regulation. April 9, 2007. 225 pp. plus Appendices.
ChemIDplus [database]. 1993- . Bethesda (MD): US National Library of Medicine. [accessed 2018 August 28].
[ECCC] Environment and Climate Change Canada. 2016a. Science approach document: ecological risk classification of organic substances . Ottawa (ON): Government of Canada.
[ECCC] Environment and Climate Change Canada. 2016b. Supporting documentation: data used to create substance-specific hazard and exposure profiles and assign risk classifications. Gatineau (QC): ECCC. Information in support of the science approach document: ecological risk classification of organic substances. Available from: substances@ec.gc.ca.
[ECCC, HC] Environment and Climate Change Canada, Health Canada. [modified 2017 Mar 12]. Categorization . Ottawa (ON): Government of Canada. [accessed 2019 December 3].
[ECHA] European Chemicals Agency. c2007-2019. Registered substances database; search results for CAS RN 132-27-4 . Helsinki (FI): ECHA. [accessed 2019 January].
[EFSA] European Food Safety Authority. 2008. Conclusion on pesticide peer review regarding the risk assessment of the active substance 2-phenylphenol. EFSA Scientific Report (2008) 217:1-67.
Environment Canada. 2009. DSL Inventory Update data collected under the Canadian Environmental Protection Act, 1999, section 71: Notice with respect to certain inanimate substances (chemicals) on the Domestic Substances List. Data prepared by: Environment Canada, Health Canada; Existing Substances Program.
[EU] European Union. 2015. Regulation (EU) No. 528/2012 concerning the making available on the market and use of biocidal products, Assessment Report: Biphenyl-2-ol Product-type PT 2 (Preventol O Extra). March 2015. 76 pp.
Health Canada. 2008a. Proposed Re-evaluation Decision: 2-Phenylphenol and salts. No. PRVD2008-04; 25 Jan. 2008. Pest Management Regulatory Agency. 23 pp.
Health Canada. 2008b. Re-evaluation Decision: 2-Phenylphenol and salts. No. RVD2008-13; 7 April 2008. Pest Management Regulatory Agency. 5 pp.
Health Canada. 2013. Interim guidance on human health risk assessment for short-term exposure to carcinogens at contaminated sites [PDF] . Ottawa (ON): Health Canada. [accessed 2019 Jan.].
Health Canada. 2014. General exposure factor inputs for dietary, occupational, and residential exposure assessments [PDF]. No. SPN2014-01; 6 November 2014. Pest Management Regulatory Agency. 19 pp.
Health Canada. 2015. Food Consumption Table derived from Statistics Canada, Canadian Community Health Survey, Cycle 2.2, Nutrition (2004), Share file. Ottawa.
[IARC] International Agency for Research on Cancer. 1999. ortho-Phenylphenol and its sodium salt. International Agency for Research on Cancer (IARC) Monograph 73: 451-480.
[JMPR] Joint Meeting of the FAO Panel of Experts on Pesticide Residues in Food and the Environment and the WHO Core Assessment Group. 1999. Toxicological evaluations. 2-Phenylphenol and its sodium salt. Rome, 20-29 September 1999. International Programme on Chemical Safety. . [accessed 2019 Feb.].
[JMPR] Joint Meeting of the FAO Panel of Experts on Pesticide Residues in Food and the Environment and the WHO Core Assessment Group. 2000. 2-Phenylphenol and its sodium salt (056). Pp. 605-636 in Pesticide residues in food − 1999; Evaluations Part 1 − Residues. FAO Plant Production and Protection Paper 157 [PDF]. Sponsored jointly by FAO and WHO. Rome, 2000.
Kwok ESC, Silva M. 2013. Re-evaluation of developmental and reproductive toxicity of ortho-phenylphenol (OPP) and sodium ortho-phenylphenate (SOPP). Cell Dev Biol. 2(3):123. doi:10.4172/2168-9296.1000123.
[MSDS] Material Safety Data Sheet. 2015. 12097 Tire & Rubber Lube. Dixon, USA: Plews and Edelmann. [Accessed 2018 Sep 19].
[NTP] National Toxicology Program.1991. NTP Chemical Repository Data Sheet: o-Phenylphenol,
Research Triangle Park, NC; Cited in: World Health Organisation, International Agency for Research on Cancer. 1999. IARC Monographs on the evaluation of carcinogenic risks to humans. Volume 73.
[NICNAS] National Industrial Chemicals Notification and Assessments Scheme. (Australia). 2016. Human health Tier II assessment for biphenylol and its sodium salt .
OECD QSAR Toolbox. 2016. Paris (FR): Organisation for Economic Co-operation and Development, Laboratory of Mathematical Chemistry.
Sasaki YF, Kawaguchi S, Kamaya A, Ohshita M, Kabasawa K, Iwama K, Taniguchi K, Tsuda S. 2002. The comet assay with 8 mouse organs: results with 39 currently used food additives. Mutation Research/Genetic Toxicology and Environmental Mutagenesis 519:103-119.
[SCCS] Scientific Committee on Consumer Safety. 2015. Scientific opinion on o-phenylphenol, sodium o-phenylphenate and potassium o-phenylphenate. Report No. SCCS/1555/15. Revised version of 15 December 2015. 122 pp.
[SCCS] Scientific Committee on Consumer Safety. 2018. Addendum to the scientific opinion on o-phenylphenol, sodium o-phenylphenate and potassium o-phenylphenate (SCCS/1555/15); Here: the use a preservative of sodium o-phenylphenate, potassium o-phenylphenate, MEA o-phenylphenate (CAS n. 132-27-4, 13707-65-8, 84145-04-0). Report No. SCCS/1587/18. 36 pp.
[US CDC] United States Centers for Disease Control and Prevention. 2017. Biomonitoring summary; ortho-Phenylphenol, CAS No. 90-43-7 . 3 pp.
[US EPA] United States Environmental Protection Agency. 1984. Health and Environmental Effects Profile for 2-Phenylphenol. Environmental Criteria and Assessment Office, Office of Health and Environmental Assessment, Office of Research and Development. U.S. Environmental Protection Agency, Cincinnati, OH45268.
[US EPA] United States Environmental Protection Agency. 2006. Reregistration eligibility decision for 2-phenylphenol and salts (orthophenylphenol or OPP). Report No. 739-R-06-004; July 2006 [PDF]. Office of Prevention, Pesticides and Toxic Substances. 146 pp.
[US EPA] United States Environmental Protection Agency. 2011. Exposure Factors Handbook 2011 . National Center for Environmental Assessment. Office of Research and Development. U.S. Environmental Protection Agency, Washington, DC 20460.
[US EPA] United States Environmental Protection Agency. 2014. ortho-Phenyl phenol (oPP) and salts final work plan; Registration Review: Initial Docket Case Number 2575. Docket Number EPA-HQ_OPP-2013-0524 . March 2014. 74 pp.
[US EPA] United States Environmental Protection Agency. 2015. EDSP Weight of Evidence Conclusions on the Tier 1 Screening Assays for the List 1 Chemicals: O-phenylphenol (o-PP). 56 pp. + memorandum dated 29 June 2015.
[US EPA] United States Environmental Protection Agency. 2016. 2-Phenylphenol: Summary of Hazard and Science Policy Council (HASPOC) Meeting on July 21, 2016: Recommendations on the Requirements for Immunotoxicity Study (OCSPP 870.7800). Memorandum dated 30 August 2016. 6 pp.
Appendix A: Read-across from OPP to SOPP
| Chemical name | Ortho-phenylphenate (OPP) | Sodium ortho-phenylphenate (SOPP) |
|---|---|---|
| CAS RN | 90-43-7 | 132-27-4 |
| Role | Analogue | Target substance |
| Chemical structure | 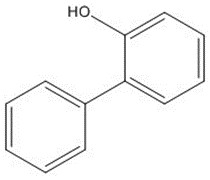 |
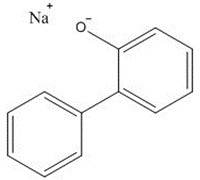 |
| Molecular weight (g/mol) | 170.2 | 192.2 |
| Vapour pressure (Pa at 25°C) | 0.27 | 1.2 |
| Water solubility (mg/L) | 530-640 (pH 5-9) | 1.00E+06 (pH 13.6) |
| Log Kow (dimensionless) | 3.18 | 2.95 |
| pKa | 9.55 | N/A |
| Absorption, distribution, metabolism, excretion | Single oral dose of OPP is 80% to 90% of OPP absorbed in rats, mice and goats within 24 hours (e.g., 85% to 86% absorption in male or female rats). A single gavage dose of radiolabelled OPP in rats resulted in less than 8% of it detected in tissue (including adipose, liver, kidneys, urinary bladder, stomach and intestine, brain and blood) at 24 h, with less than 1% detected at 7 days after dosing. Urinary metabolites (rodents, humans): sulfate and glucuronide conjugates of OPP, unconjugated phenylhydroquinone (PHQ), phenylbenzoquinone (PBQ), and 2,4'-dihydroxybiphenyl. In rats, the urinary concentrations of metabolites were greater in males than females. Excretion of OPP was rapid and complete (85-100%) in mice, rats, and humans and urine was the major excretion route in all species examined. In rats, up to 26% and 4% of an oral dose may also be excreted in bile and feces, respectively (IARC 1999; JMPR 1999; Cal EPA 2007; NICNAS 2016). | Single gavage dose of SOPP in rats is approximately 85% absorbed within 24 hours. A single gavage dose of radiolabelled SOPP in rats resulted in the same distribution results as those for OPP. Urinary metabolites (humans, rodents): sulfate and glucuronide conjugates of OPP, unconjugated PHQ, PBQ, and 2,5-dihydroxybiphenyl. In rats, the urinary concentrations of metabolites were greater in males than females. SOPP is primarily excreted in urine within 24 hours and the excretion results in bile and feces were the same as those for OPP, when SOPP was orally dosed in rats (IARC 1999; JMPR 1999; Cal EPA 2007; NICNAS 2016). |
| Eye and skin irritation, skin sensitization | Eye irritant, strong skin irritant, not a skin sensitizer (Bomhard et al. 2002). | Severe eye and skin corrosive, not a skin sensitizer (Bayer 1983 cited in ECHA c2007-2019; Toxicological Research Laboratory 1994 cited in ECHA c2007-2019; Bomhard et al. 2002). |
| Subchronic repeat dose toxicity (oral) | NR | 13-week rat study. Dose related increase in urinary alkalinity at all doses. Decreased body-weight gain at 350 mg/kg bw/day and higher. Increased relative kidney weight, decreased AST and ALT activities in males at 700 mg/kg bw/day and higher. NOAEL of 180 mg/kg bw/day (JMPR 1999), 350 mg/kg bw/day (Cal EPA 2007). 13-week mouse study. NOAEL = 730/1021 mg/kg bw/day based on decreased body weight gain in males at 1581 mg/kg bw/day and higher, and increased relative liver weights in females at 1926 mg/kg bw/day and higher (JMPR 1999; Cal EPA 2007) |
| Long-term repeat dose toxicity (oral, diet) | 2-year rat carcinogenicity study. Decreased body weight and body weight gain in both sexes, decreased food consumption in females and decreased food efficiency (sexes not specified), and increased clinical signs and gross pathological signs of toxicity in females at 200 mg OPP/kg bw/day. Increased incidence of urinary bladder papillomas and carcinomas in males (US EPA 2006, 2014; Cal EPA 2007); BMDL10 = 185.2 mg/kg bw/day (Cal EPA 2007). NOAEL of 39 mg/kg bw/day (LTD) considered protective of precursor events leading to development of bladder and liver tumours that occur at doses above 200 mg/kg/day (US EPA 2006). | 2-year rat chronic toxicity/carcinogenicity studies. , and At 224 mg/kg bw/day and higher, there was increased incidences of interstitial nephritis and increased incidences of pancreatic focal atrophy in females (Cal EPA 2007), with decreased body weights suggested at higher doses. Increased incidence of urinary bladder papillomas and/or carcinomas in both sexes at 224 mg/kg bw/day and higher (IARC 1999; Cal EPA 2007). 2-year mouse chronic toxicity/carcinogenicity study. In females at 480 mg/kg bw/day and higher in females there was decreased body weight, increased ALP activity, and decreased urine specific gravity. Increased incidence of liver tumours in both sexes at 3009 mg/kg bw/day, decreased body weight and urine specific gravity in males (IARC 1999; Cal EPA 2007). |
| Reproductive Toxicity (oral) | Rat 2-generation reproductive toxicity study. Parental NOAEL = 100 mg/kg bw/day based on decreased body weights and body weight gain in both sexes, with kidney and urinary bladder related effects in males (including chronic inflammation in both and hyperplasia of ureters and urinary bladder), mortality (due to kidney failure in an adult male), and decreased body weight in 21-day old pups at 500 mg/kg bw/day. Reproductive toxicity NOAEL = 500 mg/kg bw/day (HTD) (US EPA 2006, 2014). | Read-across from OPP. |
| Develop-mental Toxicity (oral) | Rat developmental toxicity study. Maternal NOAEL of 100 mg/kg bw/day (LTD) based on decreased body weight gain, food consumption and food efficiency at 300 mg/kg bw/day. Developmental NOAEL = 700 mg/kg bw/day (HTD) (Health Canada 2008a; US EPA 2006; Cal EPA 2007). Rabbit developmental toxicity study. Maternal NOAEL of 100 mg/kg bw/day (LTD) based on kidney effects (inflammation and tubular degeneration) at 250 mg/kg bw/day and above. Developmental NOAEL = 250 mg/kg bw/day (HTD) (Health Canada 2008a; US EPA 2006; Cal EPA 2007). | Read-across from rat and rabbit developmental toxicity studies using OPP. |
| Genotoxicity | Equivocal results in rodents in vivo and in mammalian cells in vitro (IARC 1999; Cal EPA 2007; SCCS 2015). Evidence for genotoxic potential supported by Cal EPA (2007), but not by US EPA (2006, 2014). | Overall not genotoxic in vitro but the data was mixed in vivo (reviewed in IARC 1999; Cal EPA 2007). |
| Carcino-genicity | Not classifiable (IARC Group 3) (IARC 1999). | Possibly carcinogenic to humans (IARC Group 2B) (IARC 1999). |
Abbreviations: ND, no data available; NR, Read-across not required for risk characterization; NOAEL, no observed adverse effect level; LOAEL, lowest observed adverse effect level; HTD, highest tested dose; LTD, lowest tested dose.
Appendix B: Dermal exposures to humans from products available to consumers
Sentinel exposure scenario assumptions are summarized in Table B-1. Dermal absorption was assumed to be 100%.
|
Exposure scenario |
Assumptions |
|---|---|
|
Tire and rubber lubricant (19+ years) |
Concentration of SOPP: 0.2% (MSDS 2015). A thin-film approach as outlined in the EPA Exposure factors handbook (US EPA 2011) was used. Estimated Exposure = (Concentration × SA × T × DSY) / BW It was assumed that exposure from handling a cloth coated in the product can be described as a thin film. This approach characterizes the dermal deposition from a mineral oil substance following handling of a rag saturated with the oil material, i.e., the mineral oil thickness ("thin film") estimated to remain on the skin (T) is 1.64 × 10−3 cm. This thickness was therefore assumed to apply to SOPP for characterizing dermal exposure for the application of the lubricant products. Assuming equal density of SOPP and the whole product (DSY) of 1302 mg/cm3 and an exposed skin surface area (SA) of 455 cm2 (half of both hands/palms), the dermal load was estimated to be 1.8 mg per 60-minute exposure event using a concentration of 0.2%. Using the selected body weight (BW) of 74 kg (considered to be representative of an average Canadian adult, 19+ years) (Health Canada 2015), dermal exposure was estimated to be 0.02 mg/kg-bw/event. |
