Draft screening assessment terpenes and terpenoids monocyclic and bicyclic sesquiterpenes group
Official title: Draft Screening Assessment - Terpenes and Terpenoids - Monocyclic and Bicyclic Sesquiterpenes Group
Environment and Climate Change Canada
Health Canada
May 2021
Synopsis
Pursuant to section 68 or 74 of the Canadian Environmental Protection Act, 1999 (CEPA), the Minister of the Environment and the Minister of Health have conducted a screening assessment of 16 of 76 substances referred to collectively under the Chemicals Management Plan as the Terpenes and Terpenoids Group. These 16 substances were identified as priorities for assessment as they met categorization criteria under subsection 73(1) of CEPA or were considered a priority on the basis of other human health concerns. The 16 substances addressed in this draft screening assessment report will hereafter be referred to as the Monocyclic and Bicyclic Sesquiterpenes Group. It consists of 14 substances that have been divided into 3 subgroups based on chemical structure, properties, and/or toxicity, as well as 2 individual substances namely sandalwood oil and guaiazulene. The Chemical Abstracts Service Registry Numbers (CAS RNFootnote 1), their subgroup, their Domestic Substances List (DSL) names and the common name used in this assessment are listed in the table below.
| CAS RN | Subgroup | DSL name | Common name used in this assessment |
|---|---|---|---|
| 495-62-5 | 1 | Cyclohexene, 4-(1,5-dimethyl-4-hexenylidene)-1-methyl- | Bisabolene |
| 8001-61-4a,b | 1 | Balsams, copaiba | Copaiba balsam |
| 8007-08-7a,b | 1 | Oils, ginger | Ginger oil |
| 17627-44-0 | 1 | Cyclohexene, 4-(1,5-dimethyl-1,4-hexadienyl)-1-methyl- | alpha-Bisabolene |
| 65113-99-7 | 1 | 3-Cyclopentene-1-butanol, α,β,2,2,3-pentamethyl- | Sandalore |
| 107898-54-4 | 1 | 4-Penten-2-ol, 3,3-dimethyl-5-(2,2,3-trimethyl-3-cyclopenten-1-yl)- | Santol pentenol |
| 87-44-5 | 2 | Bicyclo[7.2.0]undec-4-ene, 4,11,11-trimethyl-8-methylene, [1R-(1R,4E,9S)]- | beta-Caryophyllene |
| 88-84-6 | 2 | Azulene, 1,2,3,4,5,6,7,8-octahydro-1,4-dimethyl-7-(1-methylethylidene)-, (1S-cis)- | Guaiene |
| 3691-12-1 | 2 | Azulene, 1,2,3,4,5,6,7,8-octahydro-1,4-dimethyl-7-(1-methylethenyl)-, [1S-(1α,4α,7α)]- | alpha-Guaiene |
| 4630-07-3 | 2 | Naphthalene, 1,2,3,5,6,7,8,8a-octahydro-1,8a-dimethyl-7-(1-methylethenyl)-, [1R-(1α,7β,8aα)]- | Valencene |
| 68917-29-3b | 2 | Terpenes and terpenoids, clove oil | T & T clove oil |
| 489-86-1 | 3 | 5-Azulenemethanol, 1,2,3,4,5,6,7,8-octahydro-α,α,3,8-tetramethyl-,[3S-(3α,5α,8α)]- | Guaiol |
| 639-99-6 | 3 | Cyclohexanemethanol, 4-ethenyl-α,α,4-trimethyl-3-(1-methylethenyl)-, [1R-(1α,3α,4β)]- | Elemol |
| 22451-73-6 | 3 | 5-Azulenemethanol, 1,2,3,3a,4,5,6,7-octahydro-α,α,3,8-tetramethyl-, [3S-(3α,3aβ,5α)]- | Bulnesol |
| 489-84-9 | Individual | Azulene, 1,4-dimethyl-7-(1-methylethyl)- | Guaiazulene |
| 8006-87-9a,b | Individual | Oils, sandalwood | Sandalwood oil |
a This substance was not identified under subsection 73(1) of CEPA but was included in this assessment as it was considered a priority on the basis of other human health concerns.
b This CAS RN is a UVCB (substances of unknown or variable composition, complex reaction products, or biological materials).
Terpenes are composed of repeating isoprene units and are classified according to the number of isoprene units they contain. Monoterpenes are the smallest and contain two isoprene units. Sesquiterpenes are larger and contain three isoprene units. Like monoterpenes, sesquiterpenes may be acyclic or cyclic, including many unique combinations. These substances are components of essential oils found in a wide variety of plants.
Most of the substances in the Monocyclic and Bicyclic Sesquiterpenes Group, except for alpha-guaiene, have been included in a survey issued pursuant to section 71 of CEPA, where none of the substances were reported to be manufactured above the reporting threshold of 100 kg. Santol pentenol, sandalore, and beta-caryophyllene were reported to be imported into Canada in quantities of up to 10 000 kg, while no imports of the other substances were reported. They are generally used as fragrances in self-care products (e.g., body lotion, massage products, hair care products, oral care products, drugs, non-prescription and natural health products), cleaning products, and air fresheners. They are also present in pest control products as formulants. In addition, some of them occur naturally in food and/or may be used as food flavouring agents.
The ecological risks of substances in the Monocyclic and Bicyclic Sesquiterpenes Group were characterized using the ecological risk classification of organic substances (ERC) approach, which is a risk-based approach that employs multiple metrics for both hazard and exposure, with weighted consideration of multiple lines of evidence for determining risk classification (ECCC 2016a). Hazard profiles are based principally on metrics regarding mode of toxic action, chemical reactivity, food web-derived internal toxicity thresholds, bioavailability, and chemical and biological activity. Metrics considered in the exposure profiles include potential emission rate, overall persistence, and long-range transport potential. A risk matrix is used to assign a low, moderate or high level of potential concern for substances on the basis of their hazard and exposure profiles. Based on the outcome of the ERC analysis, the 16 substances in the Monocyclic and Bicyclic Sesquiterpenes Group are considered unlikely to be causing ecological harm.
Considering all available lines of evidence presented in this draft screening assessment, there is low risk to the environment from substances in the Monocyclic and Bicyclic Sesquiterpenes Group. It is proposed to conclude that the 16 substances in the Monocyclic and Bicyclic Sesquiterpenes Group do not meet the criteria under paragraphs 64(a) or (b) of CEPA as they are not entering the environment in a quantity or concentration or under conditions that have or may have an immediate or long-term harmful effect on the environment or its biological diversity or that constitute or may constitute a danger to the environment on which life depends.
For the human health risk assessment, 14 of the substances in this group have been addressed under three subgroups, due to similarities in chemical structure, properties and/or toxicity, while the remaining two substances were addressed individually. An impact on human health from exposure to these substances from environmental media is not expected. Where applicable, exposures were characterized from use of self-care products, possible use as food flavouring agents, cleaning products, and air fresheners containing the monocyclic and bicyclic sesquiterpenes and are expected to be predominantly via the dermal and inhalation routes.
For subgroup 1, ginger oil, bisabolene, alpha-bisabolene, santol pentenol, sandalore, and copaiba balsam demonstrated low hazard potential. Therefore, the risk to human health was also considered to be low.
For subgroup 2 (beta-caryophyllene, T & T clove oil, guaiene, alpha-guaiene and valencene), hazard information for beta-caryophyllene was used to inform the hazard assessment. Beta-caryophyllene is the main component in T & T clove oil. It was also identified as a read-across analogue for guaiene, alpha-guaiene, and valencene. For beta-caryophyllene, critical health effects were in the lymphoid system, liver, and mesenteric lymph nodes. A comparison of estimated levels of exposure to guaiene and valencene from food, to beta-caryophyllene from food, cleaning products, air fresheners, and cold sore cream, to T & T clove oil from body lotion, massage oil, and mouthwash, and to alpha-guaiene from its potential use as a fragrance ingredient with critical effect levels results in margins that are considered adequate to address uncertainties in the health effects and exposure data. A comparison of estimated levels of exposure to T & T clove oil from its use as a body fragrance with critical effect levels results in margins that are considered potentially inadequate to address uncertainties in the health effects and exposure data.
For subgroup 3 (guaiol, bulnesol, elemol), read-across data from the analogue alpha-terpineol was used to inform the hazard assessment. Critical health effects were observed on the male reproductive system. A comparison of estimated levels of exposure to bulnesol and elemol from food with critical effect levels results in margins that are considered adequate to address uncertainties in the health effects and exposure data. In addition, calculated margins of exposure to elemol from its potential use as a fragrance ingredient are considered adequate to address uncertainties in the health effects and exposure data. Since there were no identified sources of exposure to the general population for guaiol, a qualitative approach to risk characterization was taken, and the risk to human health from guaiol was considered to be low.
For sandalwood oil, read-across data from the analogue, bisabolol was used to inform the hazard assessment. Critical health effects from the dermal route of exposure were a decrease in body weight gain, decrease in feed efficiency, decrease of absolute liver weight and increase in relative testes weight. Critical health effects from the oral route of exposure were sedation, ataxia, reduced feed intake, and reduction of body weight gain in females and a significant reduction in fetal number and increase in resorption rate. A comparison of estimated levels of exposure to sandalwood oil from food, massage oil (in individuals nine years and older), facial moisturizer, shampoo, acne medication (i.e., facial cleansing system), sunscreen, cleaning products, laundry detergent, and use in aromatherapy with critical effect levels results in margins that are considered adequate to address uncertainties in the health effects and exposure data. A comparison of estimated levels of exposure to sandalwood oil from body lotion, massage oil (in infants and children up to eight years), and use of the essential oil as a body fragrance with critical effect levels results in margins that are considered potentially inadequate to address uncertainties in the health effects and exposure data.
For guaiazulene, naphthalene was used as a read-across analogue to inform the hazard assessment. Critical health effects from the dermal route of exposure were effects on the testes and non-neoplastic lesions in the cervical lymph node, liver, thyroid, kidneys, urinary bladder, and skin. From the inhalation route of exposure, critical health effects were atrophy/disorganization of the olfactory epithelium and hyperplasia of the respiratory and transitional epithelium. A comparison of estimated levels of exposure to guaiazulene from body lotion, facial moisturizer, conditioner, and aftershave product with critical effect levels results in margins that are considered adequate to address uncertainties in the health effects and exposure data. A comparison of estimated levels of exposure by the dermal or inhalation route to guaiazulene from a hair perm or straightening product with critical effect levels results in margins that are considered potentially inadequate to address uncertainties in the health effects and exposure data.
Considering all the information presented in this draft screening assessment, it is proposed to conclude that bisabolene, copaiba balsam, ginger oil, alpha-bisabolene, sandalore, santol pentenol, beta-caryophyllene, guaiene, alpha-guaiene, valencene, guaiol, elemol, and bulnesol do not meet the criteria under paragraph 64(c) of CEPA as they are not entering the environment in a quantity or concentration or under conditions that constitute or may constitute a danger in Canada to human life or health.
Considering all the information presented in this draft screening assessment, it is proposed to conclude that T & T clove oil, sandalwood oil and guaiazulene meet the criteria under paragraph 64(c) of CEPA as they are entering or may enter the environment in a quantity or concentration or under conditions that constitute or may constitute a danger in Canada to human life or health.
Therefore, it is proposed to conclude that T & T clove oil, sandalwood oil and guaiazulene meet one or more of the criteria set out in section 64 of CEPA and that the remaining 13 substances in the Monocyclic and Bicyclic Sesquiterpenes Group do not meet any of the criteria set out in section 64 of CEPA.
It is also proposed that T & T clove oil and guaiazulene meet the persistence and bioaccumulation criteria and that sandalwood oil does not meet the persistence or bioaccumulation criteria as set out in the Persistence and Bioaccumulation Regulations of CEPA.
1. Introduction
Pursuant to section 68 or 74 of the Canadian Environmental Protection Act, 1999 (CEPA) (Canada 1999), the Minister of the Environment and the Minister of Health have conducted a screening assessment of 16 of 76 substances, referred to collectively under the Chemicals Management Plan as the Terpenes and Terpenoids Group, to determine whether these 16 substances present or may present a risk to the environment or to human health. These 16 substances were identified as priorities for assessment as they met categorization criteria under subsection 73(1) of CEPA or were considered a priority on the basis of other human health concerns (ECCC, HC 2017a).
Of the other 60 substances in the Terpenes and Terpenoids Group, 19 have been assessed in terms of risk to ecological and human health, and the decisions for these substances are provided in separate reports.Footnote 2 Decisions on the remaining 41 substances will be communicated in separate screening assessments. Therefore, these 60 substances are not further addressed in this report.
The 16 substances addressed in this draft screening assessment report will hereafter be referred to as the Monocyclic and Bicyclic Sesquiterpenes Group. Some substances are assessed in subgroups due to similarities in chemical structure, properties and/or toxicity. Given the potential for these substances to be used in similar ways and applications, the potential for risk to human health is assessed using similar exposure assumptions across the subgroups.
Subgroup 1 includes substances where a qualitative risk assessment approach was taken based on low hazard potential. For subgroup 2, hazard information for beta-caryophyllene was used to inform the risk characterization. Beta-caryophyllene is a discrete substance in subgroup 2, the main component of T & T clove oil, and was identified as a read-across analogue for guaiene, alpha-guaiene, and valencene. For subgroup 3, the read-across analogue, alpha-terpineol, was used to inform the hazard characterization. Sandalwood oil and guaiazulene were assessed individually.
The ecological risks of the substances in the Monocyclic and Bicyclic Sesquiterpenes Group were characterized using the ecological risk classification of organic substances (ERC) approach (ECCC 2016a). The ERC describes the hazard of a substance using key metrics, including mode of toxic action, chemical reactivity, food web-derived internal toxicity thresholds, bioavailability, and chemical and biological activity, and considers the possible exposure of organisms in the aquatic and terrestrial environments on the basis of such factors as potential emission rates, overall persistence, and long-range transport potential in air. The various lines of evidence are combined to identify substances as warranting further evaluation of their potential to cause harm to the environment or as having a low likelihood of causing harm to the environment.
Some substances in the Monocyclic and Bicyclic Sesquiterpenes Group or read-across analogues currently being evaluated have been reviewed by the United States Environmental Protection Agency (US EPA), the European Chemicals Agency (ECHA), the European Food Safety Authority (EFSA), the European Scientific Committee on Consumer Safety (SCCS), and the World Health Organization (WHO). In addition, naphthalene, a read-across analogue for guaiazulene, was previously reviewed by Environment Canada and Health Canada (2008). Reviews conducted by these institutions are used to inform the health effects characterization in this screening assessment.
This draft screening assessment includes consideration of information on chemical properties, environmental fate, hazards, uses and exposures, including additional information submitted by stakeholders. Relevant data were identified up to March 2019. Empirical data from key studies as well as some results from models were used to reach proposed conclusions.
Beta-caryophyllene and valencene have been identified in vaping products, also known as electronic cigarettes (US EPA, 2019). The assessment of risk to the general population from this use, including risk relative to that associated with conventional cigarettes, and possible options to mitigate risk associated with these products are being addressed through a separate legislative framework (HC [modified 2020]).
This draft screening assessment was prepared by staff in the CEPA Risk Assessment Program at Health Canada and Environment and Climate Change Canada and incorporates input from other programs within these departments. The human health portions of this assessment have undergone external peer review and/or consultation. Comments on the technical portions relevant to human health were received from Dr. Christine F. Chaisson, Dr. Ricardo Andrade Furtado, and Dr. George Burdock (Risk Sciences International). The ecological portion of this assessment is based on the ERC document (published July 30, 2016), which was subject to an external review and a 60-day public comment period. While external comments were taken into consideration, the final content and outcome of the screening assessment remain the responsibility of Health Canada and Environment and Climate Change Canada.
This draft screening assessment focuses on information critical to determining whether substances meet the criteria as set out in section 64 of CEPA by examining scientific information and incorporating a weight–of-evidence approach and precaution.Footnote 3 This draft screening assessment presents the critical information and considerations on which the proposed conclusions are based.
2. Identity of substances
The Chemical Abstracts Service Registry Numbers (CAS RN), Domestic Substances List (DSL) names and common names for the discrete substances and representative substances for UVCBs in the Monocyclic and Bicyclic Sesquiterpenes Group used to inform the human health assessments are presented in Table 2-1. The substances in this assessment have been divided into three subgroups based on their chemical structure, properties, and/or toxicity, and two individual substances.
Terpenes are simple hydrocarbons consisting of repeating five carbon isoprene units (Figure 2-1). Terpenoids are a modified class of terpenes with different functional groups and an oxidized methyl group moved or removed at various positions. Both terpenes and terpenoids are classified according to the number of isoprene units they contain (Caputi and Aprea 2011; Perveen 2018). Monoterpenes contain two isoprene units. The prefixes mono-, di-, tri-, or tetra- refer to one, two, three, and four terpene units, respectively. Sesquiterpenes and sesterpenes contain three and five isoprene units, respectively.
![Representative chemical structure of isoprene, with SMILES notation: [C(C=C)(=C)C]](/content/canadasite/en/environment-climate-change/services/evaluating-existing-substances/draft-screening-assessment-terpenes-terpenoids-monocyclic-bicyclic-sesquiterpenes/_jcr_content/par/mwsadaptiveimage/image.img.jpg/1612370788720.jpg)
These substances are the components of essential oils found in a wide variety of plants. Essential oils are mixtures of volatile organic compounds originating from a single botanical source and contribute to the flavour and fragrance of a plant. These plant-derived essential oils have many components which can be extracted from different parts of the plant (e.g., leaves, seeds, stems, flowers, roots, fruits, wood, bark, grass, gum, tree blossoms, bulbs, flower buds) (Tisserand and Young 2014). In addition, the concentration of these major components can be affected by different factors, such as origin of the plant, species, temperature, soil, and geography, and essential oils extracted from plants of the same genus and species can be chemically different even though their origin is the same.
| Subgroupa | CAS RN | DSL name(common name) | Chemical structure or representative chemical name(s), structure(s), and their range of concentration(s) in the essential oil and molecular formula |
|---|---|---|---|
| 1 | 8007-08-7 | Oils, gingerb(ginger oil) |
Zingiberene Alpha-curcumene Beta-sesquiphellandrene |
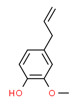
| 1 | 495-62-5 | Cyclohexene, 4-(1,5-dimethyl-4-hexenylidene)-1-methyl-(bisbolene) | Bisabolene C15H24 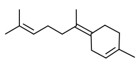 |
| 1 | 17627-44-0 | Cyclohexene, 4-(1,5-dimethyl-1,4-hexadienyl)-1-methyl-(alpha-bisabolene) | Alpha-bisabolene C15H24 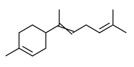 |
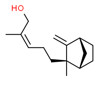
| 1 | 107898-54-4 | 4-Penten-2-ol, 3,3-dimethyl-5-(2,2,3-trimethyl-3-cyclopenten-1-yl)-(santol pentenol) | Santol pentenol C15H26O 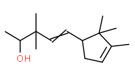 |
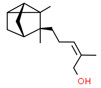
| 1 | 65113-99-7 | 3-Cyclopentene-1-butanol, α,β,2,2,3-pentamethyl-(sandalore) | Sandalore C14H26O 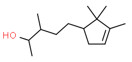 |
| 1 | 8001-61-4 | Balsams, copaibab(copaiba balsam) |
Beta-caryophyllene Alpha-Copaene trans-alpha-Bergamotene |
| 2 | 87-44-5 | Bicyclo[7.2.0]undec-4-ene, 4,11,11-trimethyl-8-methylene, [1R-(1R,4E,9S)]-(beta-caryophyllene) | Beta-caryophyllene C15H24 ![[C(=CCCC(C(C(C1(C)C)C2)C1)=C)(C2)C]](/content/dam/eccc/images/pded/terpenes-group-2/20210203-Table2-1-8.jpg) |
| 2 | 68917-29-3 | Terpenes and terpenoids, clove oilb(T & T clove oil) |
Beta-caryophyllene Eugenol |
| 2 | 88-84-6 | Azulene, 1,2,3,4,5,6,7,8-octahydro-1,4-dimethyl-7-(1-methylethylidene)-, (1S-cis)-(guaiene) | Guaiene C15H24 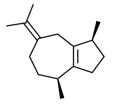 |
| 2 | 3691-12-1 | Azulene, 1,2,3,4,5,6,7,8-octahydro-1,4-dimethyl-7-(1-methylethenyl)-, [1S-(1α,4α,7α)]-(alpha-guaiene) | Alpha-guaiene C15H24 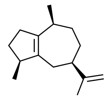 |
| 2 | 4630-07-3 | Naphthalene, 1,2,3,5,6,7,8,8a-octahydro-1,8a-dimethyl-7-(1-methylethenyl)-, [1R-(1α,7β,8aα)]-(valencene) | Valencene C15H24 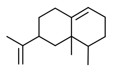 |
| 3 | 489-86-1 | 5-Azulenemethanol, 1,2,3,4,5,6,7,8-octahydro-α,α,3,8-tetramethyl-,[3S-(3α,5α,8α)]-(guaiol) | Guaiol C15H26O 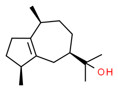 |
| 3 | 22451-73-6 | 5-Azulenemethanol, 1,2,3,3a,4,5,6,7-octahydro-α,α,3,8-tetramethyl-, [3S-(3α,3aβ,5α)]-(bulnesol) | Bulnesol C15H26O 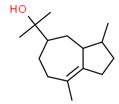 |
| 3 | 639-99-6 | Cyclohexanemethanol, 4-ethenyl-α,α,4-trimethyl-3-(1-methylethenyl)-, [1R-(1α,3α,4β)]- | ElemolC15H26O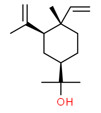 |
| Individual | 8006-87-9 | Oils, sandalwoodb(sandalwood oil) |
Alpha-Santalol Beta-Santalol |
| Individual | 489-84-9 | Azulene, 1,4-dimethyl-7-(1-methylethyl)-(guaiazulene) | Guaiazulene C15H18 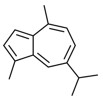 |
a The Monocyclic and Bicyclic Sesquiterpenes Group were assessed under 3 subgroups and 2 individual assessments. Subgroup 1 includes substances where a qualitative risk assessment approach was taken based on low hazard potential. For subgroup 2, hazard information for beta-caryophyllene was used to inform the risk characterization. Beta-caryophyllene is a discrete substance in subgroup 2, the main component of T & T clove oil, and was identified as a read-across analogue for guaiene, alpha-guaiene, and valencene. For subgroup 3, the read-across analogue, alpha-terpineol, was used to inform the hazard characterization. Sandalwood oil and guaiazulene were assessed individually.
b Substance of Unknown or Variable Composition, Complex Reaction Products and Biological Materials (UVCB). These substances are complex combinations of molecules that can originate in nature or are the result of chemical reactions and processes that take place during the distillation process. Given their complex and variable compositions, they could not practicably be formed by simply combining individual constituents.
c Concentration range of the main component(s) for Zingiber officinale Roscoe essential oil as cited in Tisserand and Young (2014), Rania et al. (2005), Pino et al. (2004), and Onyenekwe and Hashimoto (1999).
d Concentration range of the main component(s) for Copaifera langsdorffii/Copaifera officinalis essential oil as cited in Tisserand and Young (2005), and Swift (2005).
e Concentration range of the main component(s) for terpenes and terpenoids, clove oil, as cited in technical data sheets from two manufacturers (TDS 2018; Specification Sheet 2009).
f Concentration range of the main component(s) of Santalum album L., Santalum austrocaledonicum Vieill., and Santalum spicatum essential oil as cited in Tisserand and Young (2014), Xin-Hua et al. (2012), Brand et al. (2007) and Verghese et al. (1990).
2.1 Selection of analogues
A read-across approach using data from analogues or components of the target substances, where appropriate, has been used to inform the human health effects assessment and the risk characterization. Analogues were selected from a large list of substances having properties and structures similar to substances within this group (e.g., in terms of physical-chemical properties, toxicokinetics) and having empirical health effect data.
Analogue selection was based on analysis carried out using the Organisation for Economic Co-operation and Development (OECD) (quantitative) structure-activity relationship ([Q]SAR) toolbox version 4.2 (OECD QSAR Toolbox 2019). In the absence of empirical health effects data for bisabolene and alpha-bisabolene, the assessment was based on a read-across approach by EFSA that used limonene as an analogue to assess bisabolene and alpha-bisabolene (EFSA 2015b). Details of the read-across data chosen to inform the human health effects characterization of the substances in the Monocyclic and Bicyclic Sesquiterpenes Group are further discussed in the relevant sections of this report. Information on the identities and chemical structures of the analogues used to inform the human health assessment of the monocyclic and bicyclic sesquiterpenes (i.e., subgroup 3, sandalwood oil (UVCB) and guaiazulene) is presented in Table 2.2. Toxicological data on the analogues are presented in Appendix A.
| Subgroup or substance being assesseda | CAS RN for analogue | Common name | Chemical structure, molecular formula and SMILES | Molecular weight (g/mol) |
|---|---|---|---|---|
| 3 | 10482-56-1 | Alpha-terpineol | 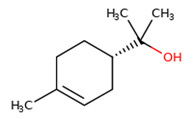 C10H18O C10H18OC10H18O CC1=CCC(CC1)C(C)(C)O |
154.25 |
| Sandalwood oil | 515-69-5 | Bisabolol | 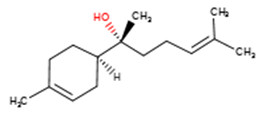 C15H26O C15H26OCC1=CCC(CC1)C(C)(CCC=C(C)C)O | 222.72 |
| Guaiazulene | 91-20-3 | Naphthalene |  C10H8 C10H8c1ccc2ccccc2c1 |
128.17 |
a Section 7.2.2.2 and 7.2.2.3 for bisabolene and alpha-bisabolene, Section 9.2 for subgroup 3, Section 10.2.3 for sandalwood oil, and Section 11.2.2 for guaiazulene.
3. Physical and chemical properties
A summary of physical and chemical property data of the substances in the Monocyclic and Bicyclic Sesquiterpenes Group is presented in Tables 3-1 to 3-4. As experimental information was not available, data from analogues were used for read-across, and/or (Q)SAR models (OECD 2019) were used to generate predicted values for the substance. Properties for the analogue substances are presented in Appendix A. Additional physical and chemical properties are reported in ECCC (2016b).
| Substance | Representative structure(s) common name (CAS RN) | Molecular weight (g/mol)a | Water solubility (mg/L)a | Vapour pressure (Pa)a | log Kowa |
|---|---|---|---|---|---|
| Oils, ginger | Zingiberene (495-60-3) | 204.19 | 1.50 x 10-2 (M) | 3.27(M) | 6.92(M) |
| Oils, ginger | Alpha-curcumene (644-30-4) | 202.17 | 1.80 x 10-2 (M) | 1.11(M) | 6.29(M) |
| Oils, ginger | Beta-sesquiphellandrene (20307-83-9) | 204.19 | 1.28 x 10-2 (M) | 4.23(M) | 6.99(M) |
| Bisabolene | N/A | 204.36 | 9.95 x 10-3 (M) | 2.93(M) | 7.12(M) |
| alpha-Bisabolene | N/A | 204.36 | 1.16 x 10-2 (M) | 2.11(M) | 7.05(M) |
| Santol pentenol | N/A | 222.37 | 2.71(M) | 1.66 x 10-2 (M) | 5.39(M) |
| Sandalore | N/A | 210.36 | 5.01(M) | 5.90 x 10-2 (M) | 5.15(M) |
| Copaiba balsam | Beta-caryophyllene (87-44-5) | 204.36 | 5.01 x 10-2 (M) | 4.16(M) | 6.30(M) |
| Copaiba balsam | Alpha-copaene (3856-25-5) | 204.36 | 3.16 x 10-1 (M) | 6.35(M) | 5.36(M) |
| Copaiba balsam | Trans-alpha-bergamotene (13474-59-4) | 204.36 | 2.99 x 10-2 (M) | 3.69(M) | 6.57(M) |
Abbreviations: N/A, not applicable; Kow, octanol-water partition coefficient
(M) Modelled
a US EPA 2012a
| Substance | Representative structure(s) common name (CAS RN) | Molecular weight (g/mol)a | Water solubility (mg/L)a | Vapour pressure (Pa)a | log Kowa |
|---|---|---|---|---|---|
| Beta-caryophyllene | N/A | 204.36 | 5.01 x 10-2 (M) | 4.16(M) | 6.30(M) |
| Terpenes and terpenoids, clove oil (T & T clove oil) | Beta-caryophyllene (87-44-5) | 204.36 | 5.01 x 10-2 (M) | 4.16(M) | 6.30(M) |
| Terpenes and terpenoids, clove oil (T & T clove oil) | Eugenol (97-53-0) | 164.20 | 754(M) | 1.26(M) | 2.27(M) |
| Guaiene | N/A | 204.35 | 1.92 x 10-2 (M) | 1.73(M) | 6.79(M) |
| alpha-Guaiene | N/A | 204.35 | 3.36 x 10-2 (M) | 3.27(M) | 6.51(M) |
| Valencene | N/A | 204.35 | 5.01 x 10-2 (M) | 4.41(M) | 6.30(M) |
Abbreviations: N/A, not applicable; Kow, octanol-water partition coefficient
(M) Modelled
a US EPA 2012a
| Substance | Representative structure(s) common name (CAS RN) | Molecular weight (g/mol)a | Water solubility (mg/L)a | Vapour pressure (Pa)a | log Kowa |
|---|---|---|---|---|---|
| Guaiol | N/A | 222.37 | 3.61(M) | 4.86 x 10-3 (M) | 5.24(M) |
| Bulnesol | N/A | 222.37 | 7.07(M) | 6.80 x 10-3 (M) | 4.90(M) |
| Elemol | N/A | 222.37 | 1.99(M) | 5.14 x 10-2 (M) | 5.54(M) |
Abbreviations: N/A, not applicable; Kow, octanol-water partition coefficient
(M) Modelled
a US EPA 2012a
| Substance | Representative structure(s) common name (CAS RN) | Molecular weight (g/mol)a | Water solubility (mg/L)a | Density (g/mL)a | Vapour pressure (Pa)a | log Kowa |
|---|---|---|---|---|---|---|
| Oils, sandalwood | Alpha-santalol (115-71-9) | 220.35 | 6.41(M) | 1.00(M) | 4.51 x 10-3 (M) | 4.96(M) |
| Oils, sandalwood | Beta-santalol (77-42-9) | 220.35 | 4.19(M) | 1.00(M) | 9.83 x 10-3 (M) | 5.18(M) |
| Guaiazulene | N/A | 198.30 | 1.12 x 10-1 (M) | 1.00 | 1.41(M) | 5.93(M) |
Abbreviations: N/A, not applicable; Kow, octanol-water partition coefficient
(M) Modelled
a US EPA 2012a
4. Sources and uses
All of the substances in the Monocyclic and Bicyclic Sesquiterpenes Group, except for alpha-guaiene, have been included in a survey issued pursuant to section 71 of CEPA (Canada 2012). Table 4-1 presents a summary of information reported on the total manufacture and total import quantities for the Monocyclic and Bicyclic Sesquiterpenes Group. Based on information submitted in response to a CEPA section 71 survey (Canada 2012), there were no reports of import or manufacture above the reporting threshold of 100 kg for ginger oil, bisabolene, alpha-bisabolene, copaiba balsam, T & T clove oil, guaiene, valencene, guaiol, bulnesol, elemol, sandalwood oil, and guaiazulene in 2011 (Environment Canada 2013). For santol pentenol and sandalore, there were no reports of manufacture above the reporting threshold of 100 kg in 2011, and between 100 and 1000 kg were imported into Canada during the same calendar year (Environment Canada 2013). For beta-caryophyllene, there were no reports of manufacture above the reporting threshold of 100 kg in 2011, and between 1000 and 10 000 kg of beta-caryophyllene were imported into Canada during the same calendar year (Environment Canada 2013).
| Common name | Total manufacture (kg)a | Total imports (kg)a | Reporting year |
|---|---|---|---|
| Ginger oil | NR | NR | 2011 |
| Bisabolene | NR | NR | 2011 |
| alpha-Bisabolene | NR | NR | 2011 |
| Santol pentenol | NR | 100 – 1000 kg | 2011 |
| Sandalore | NR | 100 – 1000 kg | 2011 |
| Copaiba balsam | NR | NR | 2011 |
| beta-Caryophyllene | NR | 1000 – 10 000 kg | 2011 |
| T & T clove oil | NR | NR | 2011 |
| Guaiene | NR | NR | 2011 |
| alpha-Guaiene | N/A | N/A | N/A |
| Valencene | NR | NR | 2011 |
| Guaiol | NR | NR | 2011 |
| Bulnesol | NR | NR | 2011 |
| Elemol | NR | NR | 2011 |
| Sandalwood oil | NR | NR | 2011 |
| Guaiazulene | NR | NR | 2011 |
Abbreviations: NR, no reports above the reporting threshold of 100 kg; N/A, not applicable, this substance was not included in the section 71 survey
a Values reflect quantities submitted in response to a CEPA section 71 survey (Canada 2012). See survey for specific inclusions and exclusions (schedules 2 and 3).
Information obtained pursuant to section 71 of CEPA indicated uses of bisabolene, alpha-bisabolene, santol pentenol, sandalore, and copaiba balsam in personal care products (Environment Canada 2013).
Additional uses for subgroup 1, subgroup 2, and sandalwood oil are outlined in Tables 4-2, 4-3, and 4-4, respectively.
| Use | Ginger oil | Bisabolene | Alpha-bisabolene | Santol Pentenol | Sandalore | Copaiba balsam |
|---|---|---|---|---|---|---|
| Food flavouring agenta | Y | Y | Y | N | N | Y |
| Food packaginga | N | N | N | N | N | Y (component in printing inks, no direct food contact) |
| Natural Health Products Ingredients Databaseb | Y (as ginger essential oil; MI, NMI) | N | N | N | N | Y (MI, NMI) |
| Licensed Natural Health Products Database being present as a medicinal or non-medicinal ingredient in natural health products in Canadab | Y (MI, NMI) | N | N | N | N | Y (MI, NMI) |
| Notified to be present in cosmetics, based on notifications submitted under the Cosmetic Regulations to Health Canadac | Y | N | N | N | Y | Y |
| Formulant in pest control products registered in Canadad | N | Y | Y | Y | Y | Y |
Abbreviations: Y, yes this use was reported for this substance; N, no this use was not reported for this substance; MI, medicinal ingredient; NMI, non-medicinal ingredient
a Personal communication, email communication from Food Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, 2015 and 2017; unreferenced
b Personal communication, email communication from Natural and Non-prescription Health Products Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, 2015; unreferenced
c Personal communication, email communication from Consumer and Hazardous Products Safety Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, 2015 and 2017; unreferenced
d Personal communication, email communication from Pest Management Regulatory Agency, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, 2015; unreferenced
| Use | Beta-Caryophyllene | T & T clove oil | Guaiene | Alpha-Guaiene | Valencene |
|---|---|---|---|---|---|
| Food flavouring agenta | Y | N | Y | N | Y |
| Natural Health Products Ingredients Databaseb | Y | N | N | N | Y |
| Licensed Natural Health Products Database being present as a medicinal or non-medicinal ingredient in natural health products in Canadab | Y (MI, NMI) | N | N | N | N |
| Notified to be present in cosmetics, based on notifications submitted under the Cosmetic Regulations to Health Canadac | Y | Y | N | N | N |
| Formulant in pest control products registered in Canadad | Y | Y | Y | Y | N |
Abbreviations: Y, yes this use was reported for this substance; N, no this use was not reported for this substance; MI, medicinal ingredient; NMI, non-medicinal ingredient
a Personal communication, email communication from Food Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, 2017; unreferenced
b Personal communication, email communication from Natural and Non-prescription Health Products Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, 2015 and 2017; unreferenced
c Personal communication, email communication from Consumer and Hazardous Products Safety Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, 2017 and October 2019; unreferenced
d Personal communication, email communication from Pest Management Regulatory Agency, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, 2015 and 2016; unreferenced
| Use | Details |
|---|---|
| Food flavouring agenta | Reported uses internationally in alcoholic and non-alcoholic beverages, baked goods, chewing gum, frozen dairy, and candy |
| Natural Health Products Ingredients Databaseb | MI (Sandalwood essential oil), NMI (Sandalwood oil) (flavour enhancer, fragrance ingredient) |
| Licensed Natural Health Products Database being present as a medicinal or non-medicinal ingredient in natural health products in Canadab | MI (Sandalwood essential oil), NMI (Sandalwood oil) |
| Notified to be present in cosmetics, based on notifications submitted under the Cosmetic Regulations to Health Canadac | Body lotions, fragrances, cleansers, massage products, and hair care products |
| Formulant in pest control products registered in Canadad | Formulant |
Abbreviations: MI, medicinal ingredient; NMI, non-medicinal ingredient.
a Personal communication, email communication from Food Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, 2015; unreferenced; Burdock 2010
b Personal communication, email communication from Natural and Non-prescription Health Products Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, February 2017; unreferenced
c Personal communication, email communication from Consumer and Hazardous Products Safety Directorate, to Existing Substances Risk Assessment Bureau, Health Canada, October 2019; unreferenced
d Personal communication, email communication from Pest Management Regulatory Agency, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, 2015; unreferenced
Subgroup 1 (ginger oil, bisabolene, alpha-bisabolene, santol pentenol, sandalore, copaiba balsam)
There are four discrete substances (bisabolene, alpha-bisabolene, santol pentenol, sandalore) and two UVCBs (ginger oil and copaiba balsam) in subgroup 1. Ginger oil is obtained by steam distillation of dried, ground ginger rhizomes. Ginger is cultivated in predominantly tropical and subtropical countries (Jamaica, India, Africa, southern China and Australia) from an herbaceous plant native to Asia, Zingiber officinale Roscoe (Burdock 2010). Copaiba balsam is obtained from the oleoresin of Copaifera species (Leguminoseae), which is grown mainly in northern and northeastern Brazil. The oleoresin obtained from the trunk of these species is a transparent liquid whose colour varies from yellow to light brown (Santiago et al. 2015). For copaiba balsam, the substance can be present in products available to consumers as a resin, oleoresin, or essential oil, all having a similar composition. The name copaiba balsam groups all subspecies of copaiba (i.e., Copaifera langsdorffii, Copaifera multijuga, Copaifera officinalis and Copaifera duckei).
Ginger oil is used in a number of products available to consumers such as body lotions, fragrances, cleansers, bath products, massage products, deodorants/antiperspirants, hair care products, and oral care products. Based on notifications submitted under the Cosmetic Regulations to Health Canada, Zingiber officinale (ginger), ginger oil, or ginger essential oil are used in over 250 products in Canada, with the majority (90%) of the products having a concentration of less than or equal to 3% (personal communication, email communication from the Consumer and Hazardous Products Safety Directorate, Health Canada, 2017; unreferenced). Ginger essential oil is also listed in the Natural Health Products Ingredients Database as a medicinal ingredient and non-medicinal ingredient as a flavour enhancer, fragrance ingredient, masking agent, skin-conditioning agent or tonicity agent (NHPID 2019). There are licensed natural health products, such as a topical pain reliever, oral antiseptic, that contain ginger essential oil as a non-medicinal ingredient (LNHPD 2018). In addition, according to the American Cleaning Institute (ACI), ginger oil is used as a fragrance in all-purpose cleaners, dish care products, and laundry care products (ACI 2018).
Copaiba balsam is used in a number of products available to consumers such as body and face lotions, massage products, cleansers, and hair care products. Based on notifications submitted to the Cosmetic Regulations to Health Canada, Balsam copaiba, Balsam copaiba resin, and Copaifera officinalis (Balsam Copaiba) resin is used in more than 60 products with the majority (> 85%) at concentrations less than or equal to 3% (personal communication, email communication from the Consumer and Hazardous Products Safety Directorate, Health Canada, 2015; unreferenced). Copaiba balsam is also reported as being used in cosmetics with film forming, masking, and perfuming functions (COSING 2018). Copaifera officinalis is listed in the Natural Health Products Ingredients Database as a homeopathic ingredient and a medicinal ingredient. Copaifera officinalis (Balsam copaiba) Resin is listed as a non-medicinal ingredient used as a film former or fragrance for products applied topically (NHPID 2019). Copaifera officinalis, Copaiba essential oil, and Copaiba are present as medicinal and homeopathic ingredients in some licensed natural health products. Copaifera officinalis (Copaiba balsam) Resin is present as a non-medicinal ingredient in licensed products such as topical pain relievers, acne medications, sunscreen and hair products (LNHPD 2018). In addition, information from the ACI’s website indicates potential use of copaiba balsam in household cleaning products (ACI 2017).
Based on notifications submitted under the Cosmetic Regulations to Health Canada, sandalore was reported in a limited number of products at concentrations up to 1%, and there were no reports for bisabolene, alpha-bisabolene, and santol pentenol (personal communication, email communication from the Consumer and Hazardous Products Safety Directorate, Health Canada, 2017; unreferenced). Bisabolene, alpha-bisabolene, santol pentenol and sandalore are fragrance ingredients used in consumer goods by the International Fragrance Association (IFRA 2016).
In Canada, bisabolene, alpha-bisabolene, santol pentenol, sandalore, and copaiba balsam were also reported to be used as formulants in pest control products.
Ginger oil and bisabolene have reported uses internationally as flavourings in food including alcoholic and non-alcoholic beverages, frozen dairy, gelatins/puddings, and soft candy (Burdock 2010). Ginger oil, bisabolene, and copaiba balsam are listed in the United States Food and Drug Administration (FDA) Substances Added to Food Inventory as flavouring agents (US FDA 2018; FCC 2018). Bisabolene is also listed as an antimicrobial agent, flavour enhancer or adjuvant, and copaiba balsam as an adjuvant (US FDA 2018). In addition, bisabolene and alpha-bisabolene are listed on the European Union (EU) List of Flavourings, and ginger oil, bisabolene, and copaiba balsam are listed in the Food Chemicals Codex as a flavouring agent (FCC 2018). No definitive information is available concerning the potential use of ginger oil, bisabolene, alpha-bisabolene, and copaiba balsam as food flavouring agents in Canada. However, since these substances are known to be used as food flavouring agents in the United States or Europe, it is possible that they are present as flavouring agents in foods sold in Canada (personal communication, email communication from Food Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, 2019; unreferenced).
Subgroup 2 (beta-caryophyllene, T & T clove oil, guaiene, alpha-guaiene, and valencene)
There are four discrete substances (beta-caryophyllene, guaiene, alpha-guaiene, and valencene) and one UVCB (T & T clove oil) in subgroup 2. Beta-caryophyllene is a natural bicyclic sesquiterpene and is a common constituent in many essential oils, especially clove oil, and essential oils of Cannabis sativa, rosemary, and hops (da Silva Oliveira 2018).
T & T clove oil is a by-product of the process of producing different clove oils. It contains beta-caryophyllene (minimum 70%) and eugenol (maximum 5%). The crude oil extracted from the leaves, bud and stem of plants from the Myrtaceae family is steam distilled and then further processed to produce various grades of clove oil and its derivatives (Ultra International B.V. 2018; TDS 2018; SS 2009). “Terpenes and terpenoids” refer to the results of the isolation of some fraction of terpene and terpenoid substances in a crude oil. Other synonyms for “terpenes and terpenoids” may be terpeneless, terpene, fractionated, rectified, or redistilled (Arctander 1960).
Beta-caryophyllene is used in a number of products available to consumers, such as body and face lotions and hair care products. Based on notifications submitted under the Cosmetic Regulations to Health Canada, beta-caryophyllene is used in a limited number of products in Canada in concentrations up to 0.1% (personal communication, email communication from the Consumer and Hazardous Products Safety Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, January 2017; unreferenced). Beta-caryophyllene is also present in several natural health products, such as a facial cleaner to treat acne and a topical product used to treat cold sores. According to the ACI, beta-caryophyllene is also used as a fragrance in liquid laundry detergent and detergent boosters (ACI 2018).
T & T clove oil is used in a number of products available to consumers, such as body lotions, fragrances, cleaners, bath products, massage products, deodorants/antiperspirants, hair care products, and oral care products. Based on notifications submitted under the Cosmetic Regulations to Health Canada, T & T clove oilFootnote 4 is used in over 600 products in Canada, with the majority (94%) of the products having a concentration of less than or equal to 3% (personal communication, email communication from the Consumer and Hazardous Products Safety Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, October 2019; unreferenced). Since there is no International Nomenclature of Cosmetic Ingredients name specific to T & T clove oil, there is a degree of uncertainty as to whether the notifications under the Cosmetic Regulations for the above-noted products contain T & T clove oil or clove oil. In the absence of any additional information, for the purposes of this assessment, it was considered that the above-noted products contain T & T clove oil. This is a conservative approach since the amount of beta-caryophyllene in traditional clove oil is much less than T & T clove oil (17% vs. >70%), and there is low hazard associated with eugenol, the main component in traditional clove oil (ECCC, HC 2018c).
Guaiene, T & T clove oil, alpha-guaiene, and valencene are also listed as fragrance ingredients used in consumer goods by the International Fragrance Association (IFRA 2017).
In Canada, beta-caryophyllene, T & T clove oil, guaiene, and alpha-guaiene are reported to be used as formulants in pest control products.
Beta-caryophyllene and valencene have been reported internationally to be used in a wide variety of prepared foods, including baked goods, frozen dairy products, chewing gum, and beverages (Burdock 2010). Guaiene is found naturally in patchouli oil and other essential oils in small quantities. It is reported to be found in lime peel oil, peppermint oil, calamus, lovage root, seed and leaf, and mangosteen (Garcinia mangostana L.) (Burdock 2010). Guaiene and valencene are permitted flavouring substances in the European Union (EFSA 2015b). Beta-caryophyllene, guaiene, and valencene are listed in the US FDA Substances Added to Food Inventory as flavouring agents or adjuvants (US FDA 2018). Beta-caryophyllene is also identified in the Food Chemicals Codex as a flavouring agent (FCC 2015). No definitive information is available concerning the potential use of beta-caryophyllene, guaiene or valencene as food flavouring agents in Canada. However, since these substances are known to be used as food flavouring agents in the United States, it is possible that they are present as flavouring agents in foods sold in Canada (personal communication, email communication from Food Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, 2019; unreferenced).
Furthermore, beta-caryophyllene and valencene have been identified in vaping products in the US (US EPA, 2019).
Subgroup 3 (guaiol, bulnesol, elemol)
There are three discrete substances in subgroup 3 (guaiol, bulnesol and elemol). They are sesquiterpenoid alcohols found in a number of essential oils including guaiacum wood, cypress pine, and guaiac wood oil (Bledsoe 2000; Petrov 2019).
In Canada, guaiol, bulnesol and elemol were not reported in cosmetic products based on notifications submitted under the Cosmetic Regulations to Health Canada. In Europe, guaiol and elemol have been reported to be used in cosmetics with a perfuming function (COSING 2018). They are also listed as fragrance ingredients used in consumer goods by the International Fragrance Association (IFRA 2017).
Elemol is used as an odour agent or fragrance ingredient in a range of products, including personal care products, cleaning products and air care products at a concentration of 50 ppm or less (follow-up to information obtained in a survey issued pursuant to a CEPA section 71 notice ([Canada 2012]).
Elemol was also reported to be used as a formulant in pest control products (personal communication, email communication from Pest Management Regulatory Agency, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, January 2016; unreferenced).
Elemol is a permitted flavouring substance in the European Union (EFSA 2015c). No definitive information is available concerning the potential use of elemol as a food flavouring agent in Canada. However, since this substance is known to be used as a food flavouring agent internationally, it is possible that it is present as a flavouring agent in foods sold in Canada (personal communication, email communication from Food Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, 2019; unreferenced).
Sandalwood oil
Sandalwood oil is used in a number of products available to consumers, such as body lotions, fragrances, cleansers, massage products, and hair care products. Based on notifications submitted under the Cosmetic Regulations to Health Canada, sandalwood oil,Footnote 5 is used in more than 650 products in Canada, with the majority (90%) of products having a concentration of less than or equal to 3% (personal communication, email communication from the Consumer and Hazardous Products Safety Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, October 2019; unreferenced).
Sandalwood oil is listed in the Natural Health Products Ingredients Database as a homeopathic ingredient, medicinal ingredient (Sandalwood essential oil) and non-medicinal ingredient with flavour enhancer and fragrance ingredient purposes (NHPID 2019). Santalum album (sandalwood) oil is present as a non-medicinal ingredient in some licensed natural health products, such as acne medications and sunscreens (LNHPD 2018).
According to the ACI, sandalwood oil is used as a fragrance in liquid all-purpose cleaners, dish care products, and laundry care products (ACI 2018). Sandalwood oil is also a formulant in pest control products (personal communication, email communication from the Pest Management Regulatory Agency, Health Canada, 2015; unreferenced).
Sandalwood oil has reported uses internationally in food including alcoholic and non-alcoholic beverages, baked goods, chewing gum, frozen dairy, and candy (Burdock 2010). Sandalwood oil is listed in the US FDA Substances Added to Food Inventory as a flavouring agent or adjuvant (US FDA 2018). No definitive information is available concerning the potential use of sandalwood oil as a food flavouring agent in Canada. However, since the substance is known to be used as a food flavouring agent in the United States, it is possible that it is present as a flavouring agent in foods sold in Canada (personal communication, email communication from Food Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, 2019; unreferenced).
Guaiazulene
Guaiazulene is a naturally occurring bicyclic sesquiterpene that is a component of various essential oils, such as guaiac wood oil and Matricaria chamomilla (Kourounakis et al. 1997).
Guaiazulene imparts a blue colour to cosmetics (Andersen 1999) and is used in a number of products available to consumers, such as body and facial moisturizers, shampoos, conditioners, bath products, hair removal after-care products, massage oils, antiperspirants, exfoliants, and makeup. Based on notifications submitted under the Cosmetic Regulations to Health Canada, guaiazulene is present in over 90 cosmetics with the majority (70%) of the products having a concentration of less than 0.1% (personal communication, email communication from Consumer and Hazardous Products Safety Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, October 2019).
Guaiazulene is listed in the Natural Health Products Ingredients Database as a non-medicinal ingredient with a colour additive and fragrance ingredient purpose (NHPID 2019). However, no licensed natural health products were identified (LNHPD 2018).
There is no information available to indicate that guaiazulene has any direct or indirect food uses in Canada or internationally (personal communication, email communication from Food Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, 2017; unreferenced).
5. Environmental fate and behaviour
5.1 Environmental persistence and potential for bioaccumulation
According to models used in ERC (ECCC 2016b), ginger oil, bisabolene, alpha-bisabolene, santol pentenol, sandalore, copaiba balsam, valencene, and sandalwood oil are not expected to persist in water, air, sediment or soil. The half-lives of beta-caryophyllene, T & T clove oil, guaiene, alpha-guaiene, guaiol, bulnesol, elemol, and guaiazulene indicate that these substances are expected to persist in water, sediment and soil, but are not expected to persist in air.
Given their low log Kow, low bioconcentration factors, and/or low bioaccumulation factors (ECCC 2016b), ginger oil, bulnesol, santol pentenol, sandalore, guaiol, elemol, and sandalwood oil are not expected to significantly bioaccumulate in organisms. Given their moderate to high log Kow (>4.2) and high bioaccumulation factors (>5000) (ECCC 2016b), bisabolene, alpha-bisabolene, copaiba balsam, beta-caryophyllene, T & T clove oil, guaiene, alpha-guaiene, valencene, and guaiazulene are expected to significantly bioaccumulate in organisms.
Therefore, it is expected that T & T clove oil, guaiene, alpha-guaiene, and guaiazulene will persist in the environment and bioaccumulate in organisms. Beta-caryophyllene, the main component of T & T clove oil which represents 73% to 80% of its composition, is also expected to persist in the environment and to have a high bioaccumulation potential.
6. Potential to cause ecological harm
6.1 Characterization of ecological risk
The ecological risks of the substances in the Monocyclic and Bicyclic Sesquiterpenes Group were characterized using the ecological risk classification of organic substances (ERC) approach (ECCC 2016a). The ERC is a risk-based approach that considers multiple metrics for both hazard and exposure, on the basis of weighted consideration of multiple lines of evidence for determining risk classification. The various lines of evidence are combined to discriminate between substances of lower or higher potency and lower or higher potential for exposure in various media. This approach reduces the overall uncertainty with risk characterization compared to an approach that relies on a single metric in a single medium (e.g., median lethal concentration) for characterization. The following summarizes the approach, which is described in detail in ECCC (2016a).
Data on physical-chemical properties, fate (chemical half-lives in various media and biota, partition coefficients, and fish bioconcentration), acute fish ecotoxicity, and chemical import or manufacture volume in Canada were collected from the scientific literature, from available empirical databases (e.g., OECD QSAR Toolbox 2014), and from responses to surveys issued pursuant to section 71 of CEPA, or they were generated using selected (quantitative) structure-activity relationship ([Q]SAR) or mass-balance fate and bioaccumulation models. These data were used as inputs to other mass-balance models or to complete the substance hazard and exposure profiles.
Hazard profiles were based principally on metrics regarding mode of toxic action, chemical reactivity, food web-derived internal toxicity thresholds, bioavailability, and chemical and biological activity. Exposure profiles were also based on multiple metrics, including potential emission rate, overall persistence, and long-range transport potential. Hazard and exposure profiles were compared to decision criteria in order to classify the hazard and exposure potentials for each organic substance as low, moderate, or high. Additional rules were applied (e.g., classification consistency, margin of exposure) to refine the preliminary classifications of hazard or exposure.
A risk matrix was used to assign a low, moderate or high classification of potential risk for each substance on the basis of its hazard and exposure classifications. ERC classifications of potential risk were verified using a two-step approach. The first step adjusted the risk classification outcomes from moderate or high to low for substances that had a low estimated rate of emission to water after wastewater treatment, representing a low potential for exposure. The second step reviewed low risk potential classification outcomes using relatively conservative, local-scale (i.e., in the area immediately surrounding a point-source of discharge) risk scenarios, designed to be protective of the environment, to determine whether the classification of potential risk should be increased.
ERC uses a weighted approach to minimize the potential for both over- and under-classification of hazard and exposure and of subsequent risk. The balanced approaches for dealing with uncertainties are described in greater detail in ECCC (2016a). The following describes two of the more substantial areas of uncertainty. Error with empirical or modeled acute toxicity values could result in changes in classification of hazard, particularly metrics relying on tissue residue values (i.e., mode of toxic action), many of which are predicted values from (Q)SAR models (OECD QSAR Toolbox 2014). However, the impact of this error is mitigated by the fact that overestimation of median lethality will result in a conservative (protective) tissue residue value used for critical body residue (CBR) analysis. Error with underestimation of acute toxicity will be mitigated through the use of other hazard metrics such as structural profiling of mode of action, reactivity and/or estrogen binding affinity. Changes or errors in chemical quantity could result in differences in classification of exposure as the exposure and risk classifications are highly sensitive to emission rate and use quantity. The ERC classifications thus reflect exposure and risk in Canada on the basis of what is estimated to be the current use quantity, and may not reflect future trends.
Critical data and considerations used to develop the substance-specific profiles for the substances in the Monocyclic and Bicyclic Sesquiterpenes Group, and the hazard, exposure and risk classification results are presented in ECCC (2016b).
The hazard and exposure classifications for the 16 substances in the Monocyclic and Bicyclic Sesquiterpenes Group are summarized in Table 5-1.
| Substance | ERC hazard classification | ERC exposure classification | ERC risk classification |
|---|---|---|---|
| Ginger oil | low | low | low |
| Bisabolene | low | low | low |
| alpha-Bisabolene | low | low | low |
| Santol pentenol | moderate | low | low |
| Sandalore | low | low | low |
| Copaiba balsam | low | moderate | low |
| beta-Caryophyllene | low | low | low |
| T & T clove oil | high | low | low |
| Guaiene | low | low | low |
| alpha-Guaiene | high | low | low |
| Valencene | high | low | low |
| Guaiol | low | low | low |
| Bulnesol | low | low | low |
| Elemol | low | low | low |
| Sandalwood oil | low | low | low |
| Guaiazulene | low | low | low |
On the basis of low hazard and low exposure classifications according to information considered under ERC, ginger oil, bisabolene, alpha-bisabolene, sandalore, beta-caryophyllene, guaiene, guaiol, bulnesol, elemol, sandalwood oil and guaiazulene were classified as having a low potential for ecological risk. It is unlikely that these substances are resulting in concerns for the environment in Canada.
According to information considered under ERC, santol pentenol was classified as having a low exposure potential. Santol pentenol was classified as having a moderate hazard potential on the basis of a moderate potential to cause adverse effects in aquatic food webs given its bioaccumulation potential. Santol pentenol was classified as having a low potential for ecological risk. The potential effects and how they may manifest in the environment were not further investigated due to the low exposure of this substance. On the basis of current use patterns, this substance is unlikely to be resulting in concerns for the environment in Canada.
According to information considered under ERC, copaiba balsam was classified as having a moderate exposure potential on the basis of a long overall persistence (the sum of chemical half-lives in all media weighted by the mass fraction of the chemical as determined using a multimedia fate model) and a moderate reported use volume according to a conservative quantity based on mean use quantity reported during the nomination of the substance to the DSL. Copaiba balsam was classified as having a low hazard potential and subsequently a low potential for ecological risk. Although the current use patterns result in a moderate exposure potential, considering its low hazard potential copaiba balsam is unlikely to be resulting in concerns for the environment in Canada.
According to information considered under ERC, T & T clove oil, alpha-guaiene and valencene were classified as having low exposure potentials. T & T clove oil, alpha-guaiene and valencene were classified as having high hazard potentials on the basis of a high potential to cause adverse effects in aquatic food webs given their bioaccumulation potential. T & T clove oil, alpha-guaiene and valencene were classified as having a moderate potential for ecological risk; however, the risk classification was decreased to low potential for ecological risk following the adjustment of risk classification based on current use quantities (see section 7.1.1 of the ERC approach document [ECCC 2016a]).The potential effects and how they may manifest in the environment were not further investigated due to the low exposure of these substances. On the basis of current use patterns, these substances are unlikely to be resulting in concerns for the environment in Canada.
7. Potential to cause harm to human health
For the health effect characterization of substances in the Monocyclic and Bicyclic Sesquiterpenes Group, preference was given to hazard data on the whole oil itself. In the absence of health effects data on the whole oil, health effects data for the major components present in the essential oil of interest were considered to inform the risk assessment. When there were no health effects data for the substance and/or major components in the whole oil, a read-across approach was taken.
7.1 Assessment of subgroup 1 (ginger oil, bisabolene, alpha-bisabolene, santol pentenol, sandalore, copaiba balsam)
7.1.1 Exposure assessment
Considering the low quantities of the subgroup 1 substances reported in response to a CEPA section 71 survey (Environment Canada 2013), exposure to these substances from environmental media is not expected.
There is potential dietary exposure to ginger oil, bisabolene, alpha-bisabolene, and copaiba balsam from their possible use as food flavouring agents. For ginger oil and copaiba balsam, exposure via the oral or dermal routes may also result from the use of natural health products intended for oral or topical use, respectively. Additionally, exposure via the dermal route may result from the use of certain cosmetics containing these substances (e.g., body lotions, cleansers, and hair care products). As the subgroup 1 substances are considered to be of low hazard potential, quantitative estimates of these potential exposures were not derived.
7.1.2 Health effects assessment of subgroup 1
Ginger oil
No international risk assessments were identified for ginger oil.
In a subchronic study, male and female Wistar rats (5/dose/sex) were administered 0, 100, 250, or 500 mg/kg bw/day ginger oil orally via gavage for 13 weeks (Jeena et al. 2011). No adverse effects were reported at any of the doses (Jeena et al. 2011). A no observed adverse effect level (NOAEL) of 500 mg/kg bw/day was established by the study authors (Jeena et al. 2011).
In a subchronic study, male and female Wistar rats (8/dose/sex) were administered 0, 600, 1000, 1400 or 1800 mg/kg bw/day ginger oil orally via gavage for 30 days. Rats treated with 1000 mg/kg bw/day and above had significant decreases in body weight and food intake as compared to the control group. The histological examination of livers presented diffuse clarification of hepatocytes, congestion and necrosis at 1400 and 1800 mg/kg bw/day (Biapa Nya et al. 2010). Based on these results, a NOAEL of 1000 mg/kg bw/day and a low observed adverse effect level (LOAEL) of 1400 mg/kg bw/day were established by the authors of the study.
In an Ames test, ginger oil was not mutagenic at 3 to 5 mg/plate in Salmonella typhimurium TA 98, TA 100, TA 102 and TA 1535 strains (Jeena et al. 2014). A single administration of an aqueous extract of ginger via gavage at doses of 500, 1000, 2000, 5000 or 10 000 mg/kg bw/day ginger oil in mice caused a significantly higher frequency of chromosomal aberrations in bone marrow cells at doses greater than 1000 mg/kg bw/day (Mukhopadhyay and Mukherjee 2000).
Based on available information, no health effects of concern are identified for ginger oil.
Bisabolene and Alpha-Bisabolene
Evaluation of bisabolene by the Joint FAO/WHO Expert Committee on Food Additives (JECFA) concluded that it does not present a safety concern as a food flavouring agent based on estimated levels of intake for Europe and the United States (0.2 μg/kg bw/day for both countries) (WHO 2005).
Due to the absence of empirical health effects data for bisabolene and alpha-bisabolene, the European Food Safety Authority based its assessment on a read-across approach with limonene, which is considered to be an analogue of bisabolene and alpha-bisabolene based on the structure and on available metabolism information (EFSA 2015b). Bisabolene, alpha-bisabolene and limonene were classified as cyclohexene hydrocarbons, and the EFSA committee concluded they were metabolized into innocuous metabolites (EFSA 2015b). Bisabolene, alpha-bisabolene and limonene are categorized as Class I under the Cramer classification. Class I substances are defined as substances of simple chemical structure with known metabolic pathways and innocuous end products that suggest a low order of oral toxicity. In addition to similarities in the chemical structure and physical-chemical properties, the applicability of limonene data to characterize the health risk potential of bisabolene and alpha-bisabolene was supported by similarities in their biological and chemical profiles in (Q)SAR model predictions (OECD QSAR Toolbox 2016).
Limonene was previously assessed under the Chemicals Management Plan in the screening assessment for Terpenes and Terpenoids, Acyclic, Monocyclic and Bicyclic Monoterpenes (ECCC, HC 2020) and no health effects of concern were identified for limonene. Accordingly, no health effects of concern are identified for bisabolene and alpha-bisabolene.
Santol pentenol
No international assessments were identified for santol pentenol. Limited empirical health effects data were identified.
In a study conducted according to OECD guideline 408, no effects were reported in male or female rats administered doses of up to 1000 mg/kg bw/day orally via gavage for 90 days. A NOAEL of greater than 1000 mg/kg bw/day was reported by the authors (ECHA Registration dossier 2015).
Similarly, in a second study conducted according to OECD guideline 421, no reproductive or developmental effects were reported in male or female rats or their pups, which were administered doses up to 1000 mg/kg bw/day orally by gavage for 90 days. A NOAEL of greater than 1000 mg/kg bw/day was reported by the authors (ECHA Registration dossier 2015).
Based on available information, no health effects of concern are identified for santol pentenol.
Sandalore
No international assessments were identified for sandalore. In addition, no mutagenicity or genotoxicity studies or dermal or inhalation toxicity studies were identified for sandalore.
In a 28-day repeated-dose subchronic toxicity study in which rats were administered sandalore oil by oral gavage at doses of 0, 35, 325 or 1000 mg/kg bw/day, no adverse effects were reported except for an increase in salivation in females and male rats treated with 325 and 1000 mg/kg bw/day (ECHA Registration dossier 2010). Rats of both sex treated with 325 and 1000 mg/kg bw/day were reported to show a significant increase in absolute and relative liver weights even after a 14-day recovery period. It was reported that microscopic examinations of livers revealed hepatocellular hypertrophy in females and males treated with 325 and 1000 mg/kg bw/day and in males treated with 35 mg/kg bw/day. Hypertrophy was also evident in the thyroid gland together with an increase in absolute and relative thyroid weights for females treated with 1000 mg/kg bw/day. However, the authors concluded that, in the absence of associated inflammatory or degenerative changes, hypertrophy in the liver and thyroid is an adaptive response. A significant increase in absolute and relative kidney weights with the presence of hyaline droplets in the tubules only in males in the 1000 mg/kg bw/day dose group was considered by the authors to be specific to male rats and not related to human health effects. In this study, the effects were reversible during the 14 day recovery period. The authors reported a NOAEL of 1000 mg/kg bw/day for toxicity (ECHA Registration dossier 2010). These conclusions aligned with our previous assessment on Acyclic, Monocyclic, and Bicyclic Monoterpenes (ECCC, HC 2020) and the US EPA risk assessment approach for human health (US EPA 1991).
In a reproductive and developmental study, in which male and females rats were administered sandalore oil by gavage at doses of 0, 100, 300 or 1000 mg/kg bw/day for 28 days, no mortality, changes in body weight, organ weight or food consumption, or effects on the reproductive system were reported (ECHA Registration dossier 2010). An increase in salivation and urine-stained abdominal fur were observed in male and female rats in the 1000 mg/kg bw/day dose group (ECHA Registration dossier 2010). However, the authors did not consider these effects to be treatment related. Similarly, no changes were noted in mating or fertility parameters, including fertility and gestation index, estrous cycle, sperm parameters, testes weight, and spermatogenesis. There were no changes reported in delivery or litter parameters as litter size and pup survival were unaffected by the treatment. There were also no clinical signs or gross lesions reported in the pups that could be attributed to maternal exposure to sandalore. The authors reported a NOAEL of 1000 mg/kg bw/day for reproductive and developmental toxicity (ECHA Registration dossier 2010).
Based on available information, no health effects of concern are identified for sandalore.
Copaiba balsam
No international assessments were identified for copaiba balsam. Copaiba balsam has not been classified with respect to its health effects by any national or international regulatory agency.
Limited information on health effects of copaiba balsam was identified in the literature.
In a developmental study conducted according to OECD guideline 414, pregnant female rats (25 per dose) were administered orally by gavage 0, 500, 1000 and 1250 mg/kg bw/day copaiba oleoresin (copaiba balsam) from gestational day (GD) 6 to GD 19 (Sachetti et al. 2011). Copaiba oleoresin was considered maternally toxic by the authors as it caused reduced food intake and body weight gain in dams at 1000 and 1250 mg/kg bw/day (Sachetti et al. 2011). However, it was reported that dams did not show any clinical signs of toxicity. Lower fetal body weight and increased occurrence of fetal skeleton variations such as incomplete frontal and thoracic ossification and unossified limbs at 1000 and 1250 mg/kg bw/day were observed at GD 20. No mortality or fetal malformations at any dose level were observed. Based on these results, a NOAEL of 500 mg/kg bw/day and a LOAEL of 1000 mg/kg bw/day were established for maternal and developmental toxicity by the authors of the study (Sachetti et al. 2011). Regarding the absence of clinical signs of toxicology in the dams, the decrease in food consumption during the treatment period at 1000 and 1250 mg/kg bw/day may be caused by the low palatability of the chemical and may be responsible for the decrease in body weights. Ossification in rodents occurs in the perinatal period (i.e., near the time of birth) and is strongly dependent on maternal food intake (DeSesso and Scialli 2018). Observations of reduced ossification are considered to be minor manifestations of developmental toxicity because they are transient and typically recover during the lactation period (DeSesso and Scialli 2018). The decrease in weight in dams and fetuses and the ossification delays in fetuses are not considered adverse in this report and a lowest observed effect level (LOEL) of 1250 mg/ kg bw/day is determined.
Male Wistar rats (5/dose) were orally administered 200, 500 or 2500 mg/kg bw/day of Copaifera multijuga, a subspecies of copaiba balsam, by gavage for 8 weeks and then mated with untreated females (2 females per male) (Gonçalves et al. 2014). No adverse effects were reported in males and their offspring. The authors concluded that oral exposure to C. multijuga oil for 8 weeks at 200, 500 or 2500 mg/kg bw/day did not cause reproductive toxicity, and an oral NOAEL for male toxicity of greater than 2500 mg/kg bw/day was suggested by the authors (Gonçalves et al. 2014).
In a reproductive and developmental study, female rats were administered a single daily dose of 28.6 mg/kg bw/day Copaifera duckei, a subspecies of copaiba balsam, intravaginally (2.5% in a vaginal cream) for 30 days before the pregnancy, 20 days during mating period and during the 20 days of pregnancy (total exposure = 70 days) (Lima et al. 2011). No adverse effects were reported in females and their offspring (Lima et al. 2011).
A single dose of copaiba oil resin, volatile or resin fraction (500, 1000 or 2000 mg/kg bw) administered by gavage to mice did not increase DNA damage in bone marrow and blood cells (Almeida et al. 2012). In a genotoxic study conducted according to OECD guideline 474, various copaiba resins administered by gavage to male Swiss mice did not increase micronuclei in bone marrow (Furtado et al. 2018). The frequency of micronuclei did not change after an exposure of Chinese hamster lung fibroblasts (V79 cells) to different copaiba resins (Furtado et al. 2018).
Based on available information, no health effects of concern are identified for copaiba balsam.
7.1.3 Risk characterization of subgroup 1
Based on available information, health effects of concern were not identified for subgroup 1. For that reason, points of departure were not defined and a qualitative approach to risk characterization was taken. Exposure to the general population to subgroup 1 is therefore considered to be of low risk to human health.
7.2 Assessment of subgroup 2 (beta-caryophyllene, T & T clove oil, guaiene, alpha-guaiene, and valencene)
7.2.1 Exposure assessment
Environmental media and food
Based on the low quantities of the substances reported to be used in Canada (Environment Canada 2013) and the low environmental exposure potential classification under ERC, exposure to T & T clove oil, guaiene, alpha-guaiene, and valencene from environmental media, including drinking water, is not expected.
The level III fugacity model known as ChemCAN (2003) was employed to derive predicted environmental concentrations of beta-caryophyllene for Canada using the upper-end of the reported volume range of 10 000 kg. The estimated concentrations in air, water, and soil were 1.01 x 10-2 ng/m3, 8.65 x 10-1 ng/L, and 4.71 x 10-3 ng/g, respectively. These estimated concentrations result in negligible exposure (i.e., less than 1 ng/kg bw/day) of the general population to beta-caryophyllene from environmental media.
Beta-caryophyllene has also been measured in indoor air in Canada in 36 homes with attached garages in Ottawa (January to February 2014, garage study) and in 54 homes in Nunavik (January to April 2018, Nunavik study) during the winter (Won 2019). Indoor air was sampled by deploying thermal desorption tubes (Tenax TA) passively over 2 or 7 days. In Ottawa, beta-caryophyllene was detected in 17% of the homes (i.e., 6 homes). However, all the samples had a concentration below the method quantification limit of 1.4 ng (approximately equal to 0.45 ng/m3). In Nunavik, beta-caryophyllene was detected in 9% of the homes (i.e., 5 homes) with a mean concentration of 0.9 µg/m3 and a range of 0.5 to 1.5 µg/m3. All other samples were below the limit of quantification (Won 2019).
Exposure to beta-caryophyllene from its presence in indoor air was estimated using the maximum measured air concentration of 1.5 µg/m3 from the Nunavik study described above (Won 2019). Exposure ranged from 1.09 x 10-3 to 3.06 x 10-4 mg/kg bw/day for all age groups (Appendix C, Table C-1).
No definitive information is available concerning the potential use of beta-caryophyllene, guaiene, and valencene in foods sold in Canada. However, since these substances are known to be used as food flavouring agents internationally, it is possible that they are present as flavouring agents in foods sold in Canada. Beta-caryophyllene, guaiene, and valencene have also been reported to occur in the various essential oils or plant-based extracts that are recognized as food flavouring agents internationally (Burdock 2010).
Beta-caryophyllene, guaiene, and valencene have been evaluated by JECFA for use as food flavouring agents (WHO 2005). It concluded that these substances present “no safety concern at current levels of intake when used as a flavouring agent.” EFSA has also evaluated beta-caryophyllene, guaiene, and valencene as food flavouring agents, also concluding that these flavouring substances would not present a safety concern at the estimated levels of intake (EFSA 2015a; EFSA 2015b). As part of these evaluations, JECFA and EFSA estimated the per capita intakes of these substances using a maximized survey-derived daily intake (MSDI) approach that is based on annual production volumes reported by the food industry in poundage surveys (NAS 1989; IOFI 1995; Lucas 1999 as cited in WHO 2005; EFSA 2015a; EFSA 2015b).
In the absence of data on the actual use, if any, of beta-caryophyllene, guaiene, and valencene as food flavouring agents in foods sold in Canada, the per capita intake estimates for the US population derived by JECFA of 8 x 10-3 mg/kg bw/day, 5 x 10-5 mg/kg bw/day, and 4 x 10-4 mg/kg bw/day are acceptable estimates of possible Canadian dietary exposure to beta-caryophyllene, guaiene, and valencene, respectively, from their use as food flavouring agents for the general population (1 year of age and older) (WHO 2005; personal communication, email communication from Food Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, May 2019; unreferenced).
Beta-caryophyllene, guaiene, and valencene are reported to occur naturally in foods (WHO 2005; Nijssen 2018) and are also reported to occur in a variety of essential oils, such as lemon peel oil, bitter orange oil, and curry leaf oil. There is expected to be little dietary exposure to these oils from their natural presence in the fruit peels and other plant materials used to obtain the essential oils.
Estimates of dietary exposure to beta-caryophyllene, guaiene, and valencene for the general Canadian population from their natural occurrence in food were derived by Health Canada’s Food Directorate using occurrence data sourced from the Volatile Compounds in Food database (Nijssen 2018). For each food and beverage category in the database, the highest concentration reported for each substance was conservatively applied to represent the food category. In cases where concentrations were of a similar magnitude between multiple related food categories, these foods were combined into a single category and the highest reported concentration of all applicable foods was applied to the entire category (e.g., all vegetables excluding potatoes).
The maximum beta-caryophyllene concentrations in food that were used in the assessment ranged from 0.001 ppm in chicken to 33 000 ppm in cinnamon, which was applied to all spices. Guaiene is reported to occur at up to 4 000 ppm in pimento berry (allspice) and at less than 1 000 ppm in ginger. The maximum guaiene concentration in allspice was applied to represent all spices and herbs. The maximum valencene concentrations range from 0.09 ppm in nuts (reported in macadamia nut) to 15.3 ppm in fruit juice (reported in orange juice).
Canadian dietary exposure to beta-caryophyllene, guaiene, and valencene from their natural occurrence in foods was estimated by multiplying the consumption of foods by the amount of each substance in those foods. Mean and 90th percentile food consumption estimates were based on individual one-day “all-persons” food intakes reported by respondents to the 2004 Canadian Community Health Survey (CCHS) for infants up to 12 months of ageFootnote 6 and the 2015 CCHS for all other age groups (Statistics Canada 2004 and 2015). The mean and 90th percentile dietary exposures estimated in this manner for various age groups are presented in Appendix B.
The mean dietary exposures for beta-caryophyllene, guaiene, and valencene from their natural occurrence in food ranged from 1.65 x 10-1 mg/kg bw/day (infants aged 6- to 12 months) to 1.52 mg/kg bw/day (4- to 8 year-olds), from 8.1 x 10-2 mg/kg bw/day (adults 19+ years) to 1.52 x 10-1 mg/kg bw/day (4- to 8-year-olds), and from 1.4 x 10-2 mg/kg bw/day (adults 19+ years) to 9.9 x 10-2 mg/kg bw/day (1- to 3-year-olds), respectively.
Products available to consumers
Beta-caryophyllene and T & T clove oil are present in products available to consumers. To evaluate the potential for exposure to beta-caryophyllene and T & T clove oil from cosmetics and natural health products (beta-caryophyllene only) applied by the dermal route, sentinel scenarios were selected based on a combination of use frequencies and reported concentrations of beta-caryophyllene and T & T clove oil in these products. These scenarios represented the highest exposure, relative to other dermally applied cosmetics or natural health products based on identified products reported to contain these substances. Exposures to T & T clove oil from the use of a body moisturizer, a massage oil, and an essential oil used as a fragrance product were considered to be the sentinel scenarios for dermal applications. Although beta-caryophyllene is also present in body moisturizer, exposure from body moisturizer containing T & T clove oil is greater than exposure to beta-caryophyllene based on the maximum concentration in the product and on the fact that beta-caryophyllene is the main component of T & T clove oil. Therefore, potential exposure to T & T clove oil is considered to be protective of potential exposures to beta-caryophyllene in body moisturizer. These data are summarized in Appendix C (Tables C-2 to C-4).
The highest daily exposures are therefore expected to occur from the use of a body moisturizer with a reported upper concentration of 3% and the use of T & T clove oil as an essential oil as a body fragrance and massage oil with an upper concentration of 100% (personal communication, email communication from Consumer and Hazardous Product Safety Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, October 2019; unreferenced). Although the upper concentration reported for massage oil containing T & T clove oil was 100%, massage oils are typically diluted prior to use. Thus, the maximum concentration of T & T clove oil in massage oil was assumed to be 3% (RIVM 2006).
Although there were no identified products for alpha-guaiene, it is used as a fragrance ingredient in consumer goods (IFRA 2017). Potential dermal exposure to this substance from its use as a fragrance ingredient was assessed using a representative product scenario with high dermal exposure estimates (i.e., body moisturizer). An assumed maximum concentration of 0.1% was used to derive the dermal exposure estimates. It was assumed that an individual substance in a fragrance mixture would be present at a maximum concentration of 10% and the maximum amount of fragrance in a cosmetic products would be 1% (HC 2016) (Table C-5).
Information from the ACI website indicates potential use of beta-caryophyllene as a fragrance in liquid laundry detergent and detergent boosters at an upper concentration of 5% (ACI 2018). To assess potential exposure to beta-caryophyllene from its use in household cleaning products, its use in liquid laundry detergent was assessed. It was assumed that beta-caryophyllene would be present at a maximum concentration of 5% as noted on the ACI website (ACI 2018).
Beta-caryophyllene is also present in cold sore medication. Exposure by the oral route from using a cold sore medication was quantified at a concentration of 20% (personal communication, email communication from Natural and Non-prescription Health Products Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, July and August, 2019; unreferenced). In addition, T & T clove oil is present in mouthwash at an upper concentration of 3% (personal communication, email communication from Consumer and Hazardous Product Safety Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, October 2019; unreferenced).
Beta-caryophyllene is also present in air fresheners as a fragrance. To assess potential exposure to beta-caryophyllene by the inhalation route, it was assumed that the amount of beta-caryophyllene in an air freshener would be equivalent to the amount of fragrance, which is up to 10% (MSDS 2015).
Systemic exposures from products available to consumers are summarized in Table 7-1.
Dermal absorption
For estimation of systemic exposure from potential dermal exposure to the subgroup 2 substances, a dermal absorption value of 20% was used for all subgroup 2 substances based on available information and the following considerations.
- A dermal absorption value of 20% for occluded skin conditions was used for the Acyclic, Monocyclic, and Bicyclic Monoterpenes Group (ECCC, HC 2020) based on in vitro human dermal absorption studies for geraniol, citronellol, and linalool (Gilpin et al. 2010; ECHA 2018). In those studies, dermal absorption ranged from 4.3% to 19.5% (mean dermal absorption value + 1 or 2 standard deviations based on variability (SCCS 2010) depending on whether the site is occluded, including skin-bound residues). Beta-caryophyllene, a representative subgroup 2 substance, has a similar molecular weight to geraniol, citronellol and linalool and also has a moderate vapour pressure. Geraniol, citronellol and linalool have moderate to high water solubility (10 to 10 000 mg/L) and log Kow (3-6), whereas beta-caryophyllene has low water solubility (0.01 to 10 mg/L) and high log Kow (6-8). This which would suggest that the dermal absorption potential of beta-caryophyllene would be less than geraniol, citronellol, and linalool.
- A limited in vitro dermal absorption study for copaiba oil containing 42% beta-caryophyllene, a subgroup 2 substance, in pig ear skin, suggested low dermal absorption of beta-caryophyllene (< 1% including skin bound residues) (Lucca et al. 2015).
Inhalation exposure
The subgroup 2 substances have vapour pressures ranging from 1.26 to 4.41 Pa and are considered to be volatile. Therefore, exposure by the inhalation route was also quantified for the sentinel dermal scenarios. To account for the amount of product absorbed by the dermal route, the product amount available for inhalation was adjusted by 80%. For body lotion, since the product amount for inhalation was adjusted for the exposed surface area and since this value was less than 80% of the product amount, no further adjustment was made to the product amount.
Total systemic exposure was calculated by summing the systemic exposure by the dermal and inhalation routes.
| Substance | Product scenario | Percent concentration in product | Daily systemic exposure (mg/kg bw/day)a |
|---|---|---|---|
| Beta-caryophyllene | NMI in cold sore cream (oral route) | 20% | 2.70 x 10-1 (adults) – 1.33 (2- to 3-year-olds) |
| Beta-caryophyllene | Washing and hanging hand-washed laundry (dermal and inhalation route) | 5% | 1.54 x 10-1 (adults) |
| Beta-caryophyllene | Air freshener (inhalation route) | 10% | 4.08 x 10-2 (adults) – 1.17 x 10-1 (infants) |
| T & T clove oil | Body lotion (dermal and inhalation route) | 3% | 8.54 x 10-1 (adults) – 1.93 (infants aged 0 to 5 months) |
| T & T clove oil | Massage oil (dermal and inhalation route) | 3% | 2.80 x 10-1 (adults) – 1.74 (infants aged 0 to 5 months) |
| T & T clove oil | Mouthwash (oral route) | 3% | 6.89 x 10-1 (adults) – 1.30 (4- to 8-year-olds) |
| T & T clove oil | Essential oil used as a body fragrance (dermal and inhalation route) | 100% | 1.31 (adults) – 3.81 (2- to 3-year-olds) |
| Alpha-guaiene | Body lotion (dermal and inhalation route) | 0.1% | 2.70 x 10-2 (adults) – 6.35 x 10-2 (infants aged 0 to 5 months) |
Abbreviations: NMI, non-medicinal ingredient
a A dermal absorption factor of 20% was used when extrapolating from the dermal to the systemic routes of exposure. Calculation details are in Appendix C.
7.2.2 Health effects assessment of subgroup 2
There are five substances in subgroup 2. Beta-caryophyllene is a discrete substance within subgroup 2, and it is also the main component in T & T clove oil.
T & T clove oil differs from clove oil in that T & T clove oil contains high levels of beta-caryophyllene (73% to 80%) and low levels of eugenol (0.5% to 5%) (TDS 2018; SS 2009). In contrast, clove oil (CAS RN 8000-34-8) contains 76% to 97% eugenol (CAS RN 97-53-0), 0.6% to 17% beta-caryophyllene, and trace amounts to 11% eugenyl acetate (CAS RN 93-28-7) (Tisserand and Young 2014; Jirovetz 2006). Clove oil was assessed by Environment and Climate Change Canada and Health Canada as part of the eugenol assessment as eugenol may be present in clove oil at concentrations up to 97%. Eugenol was considered to be a substance of low hazard potential, and therefore the risk for human health from exposure to eugenol or clove oil was considered to be low (ECCC, HC 2018c).
There is no hazard information available for T & T clove oil, alpha-guaiene or valencene. For guaiene, limited hazard information is available, consisting of skin sensitization and mutagenicity tests. There are three closely related isomers: alpha-, beta- and gamma-guaiene.
Beta-guaiene is not considered to be a skin sensitizer based on the findings of a maximization and repeated insult patch test in which human volunteers did not have a reaction following exposure to 2% beta-guaiene in petrolatum and 10% guaiene in 1:3 ethanol: diethyl phthalate (DEP), respectively. In addition, beta-guaiene is not considered mutagenic or clastogenic based on the findings of a bacterial reverse mutation assay and an in vitro micronucleus test conducted according to OECD guideline 471 and OECD guideline 487, respectively (RIFM 2017).
In the absence of hazard information for guaiene, alpha-guaiene, and valencene, EFSA identified beta-caryophyllene as a representative substance for guaiene and valencene, and guaiene as a representative substance for alpha-guaiene (EFSA 2015b). Therefore, beta-caryophyllene was used to inform the health effects assessment of these three substances. In addition, beta-caryophyllene was used to inform the health effects assessment of T & T clove oil as it is the main component of T & T clove oil.
Beta-caryophyllene
In a study conducted according to OECD guideline 408, male and female Wistar rats were administered 0, 150, 450 or 700 mg/kg bw/day of test material (equivalent to 0, 115.5, 346.5, or 539 mg/kg bw/day beta-caryophyllene) by gavage for 90 days followed by a 21-day recovery period. The authors of the study added a 28-day interim observation at the control and high-dose groups (Schmitt et al. 2016). Test material contained approximately 77% (wt/wt%) beta-caryophyllene, 1.28% (wt/wt%) eugenol and eugenol derivatives, and 21.72% (wt/wt%) other essential oils. No changes in body weight gain or food consumption were observed in either sex at any dose, No adverse effects were observed in either sex at any dose. Nasal discharge was considered incidental and not due to treatment with BCP as it was observed in the vehicle control group animals as well, but not after the recovery period. An increase in alkaline phosphatase (ALP) in the blood of high-dose females was observed. ALP is a biomarker of liver disease, so this may be a sign of liver damage at the highest dose. Lymphocyte and monocyte infiltration in control and high-dose females and vacuolar changes in the cytoplasm of hepatocytes in control and high-dose males were observed with similar incidence and severity. However, these effects could not be confirmed as the authors did not provide pictures for observation. Increases in liver weight in high-dose females and in spleen weight in low- and mid-dose males appeared to be adaptive because they were not observed in the recovery group. Authors collected lymph nodes but did not present any results. No adverse effects were observed following the administration of beta-caryophyllene or after the recovery period. A NOAEL of 700 mg/kg bw/day test material (539 mg/kg bw/day beta-caryophyllene) was determined for both male and female rats by the authors (Schmitt et al. 2016). Based on the presence of nasal discharge, which is a common symptom for viral infection, and vacuolar changes in the liver in control rats observed by authors of the study, it appears that some of the animals in the control groups (male and female) may have been ill during the study and may have altered the results.
In a study conducted according to OECD guideline 407, female Swiss mice were administered 0, 300 or 2000 mg/kg bw/day beta-caryophyllene (> 98.5% of purity) orally by gavage for 28 days (da Silva Oliveira et al. 2018). No adverse effects were observed. Authors concluded that the NOAEL for toxicity is greater than 2000 mg/kg bw/day, the highest dose tested (da Silva Oliveira et al. 2018).
Subchronic dietary administration of beta-caryophyllene at doses of 0, 222, 456 and 1367 mg/kg bw/day in male rats and 0, 263, 1033 or 4278 mg/kg bw/day in female rats for 90 days did not cause mortality, clinical signs of toxicity or ophthalmological changes (EFSA 2015b). Purity was up to 95%. Significant dose-related reductions were seen in body weight gain, food consumption and food efficiency in male and female rats in the mid- and high-dose groups (EFSA 2015b). An increase in platelet count was observed only in female rats, and a dose-dependent increase was seen in white blood cells and several other hematological parameters in male rats in the high-dose group. There were no histopathology findings correlating to these variations. Histopathological findings included an increase in absolute and relative liver weights along with centrilobular to midzonal hepatocellular hypertrophy in both sexes in the mid- and high-dose groups. Also, a significant increase was reported in absolute and relative kidney weights in males in the high-dose group and in female rats in the mid- and high-dose group. An increase in hyaline droplet accumulation or alpha2-globuline nephropathy was seen in male rats, which is considered a male rat specific effect, with little relevance for humans (EFSA 2015b). These conclusions aligned with the previous assessment on Acyclic, Monocyclic, and Bicyclic Monoterpenes (HC, ECCC 2020) and with the US EPA risk assessment approach for human health (US EPA 1991). Examination of the mesenteric lymph nodes revealed the presence of erythrocytes in the sinuses in male and female rats in the mid- and high-dose groups. Reduced spleen weights in high-dose males were considered to be related to general reductions in lymphoid system weights. Based on the lymphoid system changes in male rats and the pathological changes in the liver and mesenteric lymph nodes in both sexes, a NOAEL of 222 mg/kg bw/day was reported for beta-caryophyllene by the authors (EFSA 2015b).
In an in vivo study, exposure to 20, 200 and 2000 mg/kg did not produce any cytotoxic or genotoxic effects in blood cells in mice (Molina-Jasso et al. 2009). In an in vitro study, exposure to 100 µg/ml of beta-caryophyllene did not produce any cytotoxic or genotoxic effects in cultured human lymphocytes (Di Sotto et al. 2010).
7.2.3 Characterization of risk to human health
A NOAEL of 222 mg/kg bw/day has been identified for beta-caryophyllene for all routes of exposure based on lymphoid system changes in male rats, pathological changes in the liver and mesenteric lymph nodes in both sexes, and kidney effects in females in one of the 90-day oral studies (EFSA 2015b). No hazard data were identified for the dermal and inhalation routes of exposure to beta-caryophyllene. Therefore, the oral NOAEL of 222 mg/kg bw/day was used for characterization of risk along with route-to-route extrapolation.
Daily exposure estimates and resulting margins of exposure (MOEs) are summarized in Table 7-2.
| Substance | Exposure scenarioa | Systemic exposure (mg/kg bw/day) | MOEb |
|---|---|---|---|
| Beta-caryophyllene | Environmental media (indoor air) and food flavouring agent (1 year and older) | 8.31 x 10-3 (adults) – 9.09 x 10-3 (1-year-olds) | > 24 000 (all subpopulations) |
| Beta-caryophyllene | Systemic exposure by the oral route from cold sore cream (20%) | 2.70 x 10-1 (adults) – 1.33 (2- to 3-year-olds) | 167 (2- to 3-year-olds) – 821 (adults) |
| Beta-caryophyllene | Systemic exposure by the dermal and inhalation route from mixing, loading, washing and hanging hand-washed laundry (5%) | 1.54 x 10-1 (adults) | 1439 (adults) |
| Beta-caryophyllene | Systemic exposure by the inhalation route from an air freshener (10%) | 4.08 x 10-2 (adults) – 1.23 x 10-1 (2- to 3-year-olds) | 1810 (2- to 3-year-olds) – 5440 (adults) |
| T & T clove oil | Systemic exposure by the dermal and inhalation route from body lotion (3%) | 8.54 x 10-1 (adults) – 1.93 (infants aged 0 to 5 months) | 115 (infants aged 0 to 5 months) – 260 (adults) |
| T & T clove oil | Systemic exposure by the dermal and inhalation route from massage oil (3%) | 2.80 x 10-1 (adults) – 1.74 (infants aged 0 to 5 months) | 127 (infants aged 0 to 5 months) – 793 (adults) |
| T & T clove oil | Systemic exposure by the oral route from mouthwash (3%) | 6.89 x 10-1 (adults) – 1.30 (4- to 8-year-olds) | 170 (4- to 8-year-olds) – 322 (adults) |
| T & T clove oil | Systemic exposure by the dermal and inhalation route from use of the essential oil as a body fragrance (100%) | 1.31 (adults) – 3.81 (2- to 3-year-olds) | 58 (2- to 3-year-olds) – 169 (adults) |
| Alpha-guaiene (fragrance) | Systemic exposure by the dermal and inhalation route from body lotion (0.1%) | 2.70 x 10-2 (adults) – 6.35 x 10-2 (infants aged 0 to 5 months) | 3495 (infants aged 0 to 5 months) – 8209 (adults) |
| Guaiene | Food flavouring agent (dietary intake) (1 year and older) | 5 x 10-5 | 4 440 000 (1-year-olds and over) |
| Valencene | Food flavouring agent (dietary intake) (1 year and older) | 4 x 10-4 | 555 000 (1-year-olds and over) |
a Exposure scenario parameters and calculations for subgroup 2 are outlined in Appendix C.
b Margin of exposure calculated using the critical effect level (NOAEL = 222 mg/kg bw/day) based on lymphoid system changes in male rats, pathological changes in the liver and mesenteric lymph nodes in both sexes, and non-explained effects in female kidneys from a 90-day oral study conducted with beta-caryophyllene.
Calculated MOEs for beta-caryophyllene, guaiene, and valencene from food (based on their potential use as food flavouring agents) are considered adequate to address uncertainties in the health effects and exposure data. Additional intake of beta-caryophyllene, guaiene, and valencene from their natural occurrence in food was not identified as a concern for human health. For exposure to beta-caryophyllene from environmental media, cleaning products, air fresheners, and cold sore cream and to T & T clove oil from body lotion, massage oil, mouthwash, and its use as an essential oil as a body fragrance (9 years and older), the margin of exposure between the critical effect level and the estimate of exposure listed in Table 8-3 is considered adequate to account for uncertainties in the health effects and exposure data. In addition, the calculated MOE for alpha-guaiene from its potential use as a fragrance ingredient is considered adequate to address uncertainties in the health effects and exposure data.
The margin between the critical effect level and the estimate of daily exposure to T & T clove oil from its use as an essential oil as a body fragrance for 2- to 8-year-olds is considered potentially inadequate to account for uncertainties in the health effects and exposure data.
7.2.4 Uncertainties in evaluation of risk to human health
The key sources of uncertainty are presented in the table below.
| Key source of uncertainty | Impact |
|---|---|
| Exposure | N/A |
| There is a degree of uncertainty with the dermal absorption factor used for the subgroup 2 substances as it is based on consideration of available information. However, the use of a 20% dermal absorption factor is not expected to underestimate systemic exposures by the dermal route as it is an upper-bound estimate from the dermal absorption data available for the Acyclic, Monocyclic, and Bicyclic Monoterpenes Group and the Monocyclic and Bicyclic Sesquiterpenes Group. | +/- |
| As there are a number of fragrance substances (approximately 25) in the air freshener product, the assumption that the concentration of fragrance in the air freshener product is equivalent to the concentration beta-caryophyllene is a conservative estimate. | + |
| There is a degree of uncertainty as to whether the products available to consumers contain T & T clove oil or traditional clove oil. In the absence of any additional information, it was assumed that the products contain T & T clove oil. This is a conservative approach since the amount of beta-caryophyllene in traditional clove oil is much less than T & T clove oil (17% versus >70%) and there is low hazard associated with eugenol, the main component in traditional clove oil (ECCC, HC 2018c). | + |
| Hazard | N/A |
| There is limited information on repeated-dose effects via relevant routes of exposure and different durations for beta-caryophyllene. In addition, there are limited animal studies examining the repeated-dose toxicity of T & T clove oil, guaiene and valencene for the relevant routes of exposure (i.e., dermal, oral, inhalation). Hazard data from the main component, beta-caryophyllene, were used to inform the health effects assessment, where applicable. | +/- |
+ = uncertainty with potential to cause over-estimation of exposure/risk; - = uncertainty with potential to cause under-estimation of exposure/risk; +/- = unknown potential to cause over or under-estimation of risk.
7.3 Assessment of subgroup 3 (guaiol, bulnesol, elemol)
7.3.1 Exposure assessment of subgroup 3
Environmental media and food
Based on the low quantities (< 100 kg) of guaiol, bulnesol and elemol reported to be used in Canada (Environment Canada 2013), exposure to these substances from environmental media, including drinking water, is not expected.
Bulnesol was not identified to be used as a food flavouring agent in Canada or internationally. Elemol has been reported to occur in various essential oils or plant-based extracts that are also recognized as food flavouring agents internationally (Burdock 2010).
EFSA estimated the per capita intake of elemol from its use as a food flavouring in Europe to be 1.6 µg per day (0.027 µg/kg bw for a 60-kg person) using an MSDI approach that is based on annual production volumes reported by the food industry in poundage surveys (International Organization of the Flavour Industry 1995, as cited in EFSA 2015c). In the absence of data on the actual use, if any, of elemol as a food flavouring agent in foods sold in Canada, the per capita intake estimate derived by EFSA was used to represent possible Canadian dietary exposure of the general population 1 year of age and older to this substance from potential use as a food flavouring agent.
Bulnesol and elemol are reported to occur naturally in foods (WHO 2006; EFSA 2015c; Nijssen 2018) and are also reported to occur in a variety of essential oils, such as lemon peel oil, mandarin peel oil, bitter orange oil and curry leaf oil. There is expected to be little dietary exposure to these oils from their natural presence in the fruit peels and other plant material used to obtain the essential oils.
Estimates of dietary exposure to bulnesol and elemol for the general Canadian population from their natural occurrence in food were derived by Health Canada’s Food Directorate using occurrence data sourced from the Volatile Compounds in Food database (Nijssen 2018). For each food and beverage category in the database, the highest concentration reported for each substance was conservatively applied to represent the food category. In cases where concentrations were of a similar magnitude between multiple related food categories, these foods were combined into a single category and the highest reported concentration of all applicable foods was applied to the entire category (e.g., all vegetables excluding potatoes; all fruit juices).
Bulnesol is reported to occur in spices, with the maximum concentration of 4 500 ppm reported in basil and applied to represent all spices and herbs. The maximum elemol concentrations in foods that were used in the assessment ranged from 0.37 ppm in grapefruit juice (applied to represent all fruit juices) to 301 ppm in ginger (applied to represent all spices).
Canadian dietary exposure to bulnesol and elemol from their natural occurrence in foods was estimated by multiplying the consumption of foods by the amount of each substance reported in those foods. Mean and 90th percentile food consumption estimates were based on individual one-day “all-persons” food intakes reported by respondents to the 2004 Canadian Community Health Survey (CCHS) for infants up to 12 months of ageFootnote 7 and the 2015 CCHS for all other age groups (Statistics Canada 2004 and 2015). The mean dietary exposures estimated in this manner ranged from 8.2 x 10-2 mg/kg bw/day (14- to 18-year-olds) to 1.71 x 10-1 mg/kg bw/day (4- to 8-year- olds) for bulnesol and from 6 x 10-3 mg/kg bw/day (infants aged 6 to 12 months) to 2.7 x 10-2 mg/kg bw/day (4- to 8–year-olds) for elemol (Appendix B).
Products available to consumers
Elemol is used as an odour agent or fragrance ingredient in a range of products, including personal care products, cleaning products and air care products at a concentration of 50 ppm or less (follow-up to Section 71 information reported under Canada 2012 notice). To assess potential exposure to elemol from its use as a fragrance ingredient, a representative product scenario with high dermal exposure estimates (i.e., body moisturizer) was used with the maximum concentration of 50 ppm (0.005%) to derive dermal exposure estimates. Assuming complete absorption by the dermal route (i.e., 100% dermal absorption), systemic exposure estimates by the dermal route ranged from 6.76 x 10-3 for adults to 1.59 x 10-2 for infants aged 0 to 5 months .
7.3.2 Health effects assessment of subgroup 3
There is no empirical health effects data available for guaiol, bulnesol and elemol. No international assessments for guaiol, bulnesol and elemol, were available.
In the absence of hazard data on these substances, a read-across approach was taken, and hazard information on the analogue, alpha-terpineol, was used to inform the hazard assessment.
Alpha-terpineol is characterized by a six carbon ring including one double bond (cyclohexene), possessing multiple methyl groups and one alcohol group like elemol in subgroup 3, with the difference that alpha-terpineol is monocyclic whereas guaiol and bulnesol are bicyclic sesquiterpenes and have a seven carbon ring with one double bond (Dolejš et al. 1960). Analogue and target substances also have similar physical and chemical properties and are naturally found in essential oils extracted from Duguetia lanceolata or Cinnamomum illicioides (Isman 2000). Metabolism of both alpha-terpineol and elemol yields innocuous metabolites (OECD 2019). Toxicokinetics data is not available for guaiol and bulnesol but it is very likely they follow similar metabolism pathways to alpha-terpineol because the conjugation represents the major pathway of metabolism for these alcohols (Belsito et al. 2008). Bulnesol, guaiol, elemol and alpha-terpineol are categorized as Class I under the Cramer classification. Class I substances are defined as substances of simple chemical structure with known metabolic pathways and innocuous end products that suggest a low order of oral toxicity.
In a study conducted according to OECD guideline 422, three groups of 10 male and 10 female rats were administered 0, 60, 250 or 750 mg/kg bw/day alpha-terpineol by gavage for a minimum of 35 days (2 weeks before mating, throughout mating and gestation and until day 6 of lactation) (ECHA Registration dossier 2019b). The third group had 2 weeks of recovery. No clinical or toxic signs were recorded in males and females. A decrease in food consumption and body weight in males and females was observed only at the 750 mg/kg bw/day dose. At 750 mg/kg bw/day, blood urea, creatinine and plasma glucose levels were significantly higher. However, all of the blood parameters showed complete recovery after 2 weeks. Adaptive centrilobular hepatocyte hypertrophy in the liver of females in the 750 mg/kg bw/day group was not present after 2 weeks recovery and histopathological findings in the kidneys of males receiving 250 and 750 mg/kg bw/day also resolved after the end of dosing. At 750 mg/kg bw/day, reduced numbers or complete absence of spermatozoa accompanied by the presence of degenerate spermatogenic cells in ducts were observed in epididymides. Seminiferous tubular atrophy/degeneration was seen in the testes of all animals in the 750 mg/kg bw/day group accompanied by spermatid giant cells and seminiferous tubule vacuolation. At 750 mg/kg bw/day, none of the females mated with males became pregnant. The effects on the male reproductive tract would have been sufficient to prevent fertilization and render the males completely infertile. Similar findings were still present following the 2-week recovery period. There were no effects on female reproductive parameters such as estrous cycles, precoital interval or mating in any group and no effects on the number of implantations, post implantation survival index or live birth index for females at the mid and low doses. Based on the male reproductive effects, the NOAEL for reproductive toxicology was determined to be 250 mg/kg bw/day by the study’s authors (ECHA Registration dossier 2019b).
A comparative 2-week study was conducted where alpha-terpineol was administered orally either by diet or by gavage to male rats. Two groups of 5 male rats received terpineol orally by gavage at 500 or 750 mg/kg bw/day and two others via the diet at concentrations of 8000 or 12000 ppm alpha-terpineol for two weeks (ECHA Registration dossier 2019b). In the gavage groups, feed intake was reported to have decreased in both treatment groups during the treatment and at the end of the 2-week treatment period. In the diet groups, food intake was very low during the first days of the study but increased rapidly and total substance intake in the high-dose group remained slightly over 750 mg/kg bw/day during all of week 2. A clear decrease in body weight gain was reported in both groups. Effects on sperm motility were reported in gavage groups, while no effects were detected in the diet groups. Authors concluded that effects on the male reproductive tract depend of the mode of dose administration (ECHA Registration dossier 2019b). However, the exposure of alpha-terpineol was shorter in this study (14 days) in comparison with the previous study (35 days) and may not be representative of the effect on spermatogenesis seen in the previous study after 35 days of exposure.
Adverse effects were not reported in the reproductive male tract and liver in a repeated-dose study in male rats (10 animals/dose) in which administered doses of 0 or 622.65 mg/kg bw/day (12000 ppm) alpha-terpineol were administered by diet for 90 days (i.e., a whole period of spermatogenesis) (ECHA Registration dossier 2019b). Authors concluded that effects on male reproductive tract could only be observed because of the peak in concentration due to gavage dosing (ECHA Registration dossier 2019b). Authors of the study explained that in some cases, gavage administration creates pharmacokinetic circumstances that cannot be encountered in real conditions of exposure such as diet and can be considered in this case as a non-relevant route of exposure (ECHA Registration dossier 2019b). However, authors did not provide any measures of the chemical in the blood or urea of animals to substantiate this hypothesis.
In an OECD 414-compliant study, a terpineol mixture was administered by gavage to pregnant female rats (20/dose) at dose levels of 0, 60, 200, or 600 mg/kg bw/day from GD 6 to GD 19 (ECHA Registration dossier 2019b). This terpineol mixture contained alpha-terpineol as its main constituent (62% to 80%). The adjusted mean liver weights of females receiving 600 mg/kg bw/day was significantly higher, but there were no macroscopic abnormalities detected. No clinical signs or signs of reaction to treatment were noted in treated females. Food intake in the 600 mg/kg bw/day group was reported to be lower throughout the treatment period, but did not change in the 60 and 200 mg/kg bw/day groups. There was no reported effect of maternal treatment at any dose level on litter data except for embryo-fetal growth, which was slightly reduced at 600 mg/kg bw/day. In addition, mean placental weight was noted to be lower at 600 mg/kg bw/day. Placental, litter and fetal weights were reported to be unaffected at 60 and 200 mg/kg bw/day. In the 600 mg/kg bw/day group, there was a slightly higher incidence of incompletely ossified or unossified 5th and/or 6th sternebrae, but it may be related to the decrease in food consumption in dams and was not considered to constitute an adverse effect on development. On the basis of the results obtained in this study, the dose of 600 mg/kg bw/day terpineol mixture (equivalent to 480 mg/kg bw/day alpha-terpineol), the highest dose tested, was considered to be the NOAEL for maternal and developmental toxicity by study authors (ECHA Registration dossier 2019b).
In a study conducted according OECD guideline 413, a terpineol mixture was administered by inhalation-aerosol to male and female rats (10/sex/dose) at 0, 0.202, 0.572 or 2.23 mg/L (equivalent to 0, 202, 572 or 2230 mg/m3, respectively) for 6 hours per day, 5 days per week for 13 weeks (ECHA Registration dossier 2019b). Some animals in the control and high-dose groups had a recovery period of 4 weeks. No adverse effects were reported by the authors. On the basis of on these results, a NOAEC of 2.23 mg/L (equivalent to 2230 mg/m3), the highest dose tested, was established by study authors (ECHA Registration dossier 2019b).
Terpineol multiconstituent was found not to be mutagenic in bacterial reverse mutation assays with S. typhimurium strains TA 98, TA 100, TA 102, TA 1535 and TA 1537 with and without metabolic activation (ECHA Registration dossier 2019b).
Alpha-terpineol was not mutagenic in bacterial assays and did not induce gene mutations in mammalian cells either with or without exogenous metabolic activation (Belsito et al. 2008; Bhatia et al. 2008a). In a 14-day repeated dose study in 3 to 4 male rats, administration of a 1% alpha-terpineol supplemented ration caused a reduction in food intake, body weight and ApoA-1 (component of high density lipoprotein) levels. An increase in cholesterol and triacylglycerol levels and liver weight was also observed (Imaizumi et al. 1985).
Alpha-terpineol was evaluated for its potential to induce lung tumours in female A/He mice (Stoner et al. 1973). Mice were administered alpha-terpineol via intraperitoneal injection three times a week for 8 weeks for total cumulative doses of 1.9 and 9.6 g/kg (80 and 400 mg/kg bw/dose or 35 and 170 mg/kg bw/day). Mice were sacrificed 12 weeks following the last injection. At the 1.9 g/kg dose level, there were two deaths and tumours occurred in 3 of the 18 surviving animals. At the 9.6 g/kg dose level, there were five deaths and tumours occurred in 1 of the 15 surviving animals. There was no significant difference in tumour incidence between treated and control animals, and consequently alpha-terpineol was determined not to be carcinogenic in this study by the authors (Stoner et al. 1973).
7.3.3 Characterization of risk to human health
The critical effect level identified for alpha-terpineol from a 35-day reproductive toxicity gavage study is a NOAEL of 250 mg/kg bw/day. The effect level is based on complete infertility due to a reduced number or complete absence of spermatozoa in the presence of degenerate spermatogenic cells in the epididymides and seminiferous tubular atrophy/degeneration in the testes of male rats at 750 mg/kg bw/day (ECHA 2019b).
Daily exposure estimates and resulting MOEs are summarized in Table 7-4.
| Substancea | Exposure scenario | Daily systemic exposure (mg/kg bw/day) | MOEb |
|---|---|---|---|
| Elemol | Food flavouring agent (dietary intake) (children aged 1 year and over) | 2.70 x 10-5 | 9 259 259 |
| Elemol | Dermal exposure from body lotion (0.005%) (all subpopulations) | 6.76 x 10-3 (adults) – 1.59 x 10-2 (infants) | 15750 (infants) – 37000 (adults) |
a Exposure scenario parameters and calculations for elemol are outlined in Appendix D. Dermal absorption was assumed to be 100%.
b Margin of exposure calculated using the critical effect level (NOAEL = 250 mg/kg bw/day) based on reduced number or complete absence of spermatozoa in the presence of degenerate spermatogenic cells in the epididymides and seminiferous tubular atrophy/degeneration in the testes of males rats from a 35-day reproductive toxicity gavage study with terpineol.
The calculated MOE for elemol in food from its potential use in Canada as a food flavouring agent is considered adequate to address uncertainties in the health effects and exposure data. Additional intake of elemol from its natural occurrence in food, and natural occurrence of bulnesol in food, was not identified as a concern for human health. In addition, the calculated MOE for elemol from its potential use as a fragrance ingredient is considered adequate to address uncertainties in the health effects and exposure data.
Since there were no identified sources of exposure to the general population for guaiol, a qualitative approach to risk characterization was taken, and the risk to human health from guaiol was considered to be low.
7.3.4 Uncertainties in evaluation of risk to human health
The key sources of uncertainty are presented in the table below.
| Key source of uncertainty | Impact |
|---|---|
| Hazard | N/A |
| There is no hazard information for guaiol, bulnesol and elemol. Read-across data from the analogue, alpha-terpineol, was used to inform the risk assessment. | +/- |
+ = uncertainty with potential to cause over-estimation of exposure/risk; - = uncertainty with potential to cause under-estimation of exposure/risk; +/- = unknown potential to cause over or under-estimation of risk. The achieved margins of exposure were considered adequate to address uncertainties in the exposure and hazard databases.
7.4 Sandalwood oil
7.4.1 Exposure assessment of sandalwood oil
Environmental media and food
In consideration of the low quantities of the substance reported to be used in Canada (Environment Canada 2013), exposure to sandalwood oil from environmental media is not expected.
No definitive information is available concerning the potential use of sandalwood oil as a food flavouring agent in foods sold in Canada. However, since sandalwood oil is known to be used as a food flavouring agent internationally, it is possible that it is present as a flavouring agent in foods sold in Canada.
JECFA and EFSA have not reported exposure estimates for sandalwood oil when used as a flavouring agent, but Fenaroli’s Handbook of Flavour Ingredients (Burdock 2010) reports “individual’ consumption intake of this substance from its use as a food flavouring agent. Individual consumption intakes are a per capita estimate of intake based on an MSDI approach for the US population analogous to that employed by JECFA.
In the absence of data on the actual use, if any, of sandalwood oil as food flavouring agent in foods sold in Canada, the per capita intake estimates for the US population of 1.20 x 10-4 mg/kg bw/day reported in Fenaroli’s Handbook of Flavour Ingredients are acceptable estimates of possible Canadian dietary exposure to this substance from its use as a food flavouring agent for the general population 1 year of age and older (personal communication, email communication from Food Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, May 2019; unreferenced; Burdock 2010).
Products available to consumers
The dermal route of exposure is expected to be the predominant route of exposure from products available to consumers. Based on the low vapour pressure of the two main components of sandalwood oil, i.e., alpha-santalol (4.51 x 10-3 Pa) and beta-santalol (9.83 x 10-3 Pa), and the identified uses, the inhalation route is not expected to be a significant route of exposure.
To evaluate the potential for exposure to sandalwood oil from cosmetics applied by the dermal route, sentinel scenarios were selected based on a combination of use frequencies and reported concentrations of sandalwood oil in these products. Exposure to sandalwood oil from the use of a body moisturizer, massage oil, facial moisturizer, shampoo, and the use of the essential oil as a body fragrance were considered to be the sentinel scenarios for dermal applications. These data are summarized in Appendix E (Tables E-1 to E-5).
Thus, the highest daily exposure to sandalwood oil is expected to occur from the use of a body moisturizer with a reported upper concentration of 30% sandalwood oil. Although the upper concentration reported for massage oil was 100%, massage oils are typically diluted prior to use. Thus, the maximum concentration of sandalwood oil in massage oil was assumed to be 3% (RIVM 2006). Furthermore, dermal exposures from use of a facial moisturizer with an upper concentration of 3%, use of the essential oil as a body fragrance with an upper concentration of 100%, and use of a shampoo with an upper concentration of 10% were estimated (personal communication, email communication, from Consumer and Hazardous Products Safety Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, October 2019; unreferenced).
Sandalwood oil was also reported to be used as a non-medicinal ingredient in a facial cleansing system to treat acne and in a sunscreen at concentrations of up to 2% and 0.04%, respectively (May 2012; personal communication, email communication from Natural and Non-prescription Health Products Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, January 2019; unreferenced). Since the three components of the facial cleansing system are intended to be used together, daily exposures from a facial cleanser, facial moisturizer, and intensive spot treatment were summed. In addition, dermal exposure from use of sunscreen was also quantified (Appendix E, Table E-6 and E-7).
Sandalwood oil may also be purchased as a pure essential oil and used in aromatherapy. Inhalation exposure estimates from aromatherapy ranged from 2.24 x 10-1 for adults to 8 x 10-1 mg/kg bw/day for infants aged 0 to 5 months (Appendix E, Table E-8 and E-9). The aromatherapy exposure scenario was considered to be the sentinel scenario for potential inhalation exposure from the use of sandalwood oil in air fresheners, candles and incense.
Information from the ACI’s website indicates potential use of sandalwood oil as a fragrance in all-purpose cleaners and laundry care products at an upper concentration of 1% and 5%, respectively (ACI 2018). To assess potential exposure to sandalwood oil from its use in household cleaning products, the use of sandalwood oil in all-purpose cleaners and liquid laundry detergent was assessed. It was assumed that sandalwood oil would be present at a maximum concentration of 1% and 5% in all-purpose cleaners and liquid laundry detergents, respectively, as noted on the ACI website (ACI 2018) (Appendix E, Table E-8 and E-9).
Exposures from products available to consumers are summarized in Table 7-6.
| Scenario | Percent concentration in product | Daily exposure (mg/kg bw/day)a |
|---|---|---|
| Body lotion (dermal route) | 30% | 40.5 (adults) – 95.2 (infants aged 0 to 5 months) |
| Massage oil (dermal route) | 3% | 1.30 (adults) – 8.57 (infants aged 0 to 5 months) |
| Facial moisturizer (dermal route) | 3% | 7.23 x 10-1 (14- to 18-year-olds) – 1.22 (adults) |
| Essential oil used as body fragrance (dermal route) | 100% | 7.58 (adults) – 22.0 (2- to 3-year-olds) |
| Shampoo (dermal route) | 10% | 1.75 x 10-1 (adults) – 6.19 x 10-1 (infants aged 0 to 5 months) |
| Facial cleansing system (dermal route) | 2% | 7.39 x 10-1 (14- to 18-year-olds) – 1.23 (adults) |
| Sunscreen (dermal route) | 0.04% | 8.40 x 10-2 (9- to 13-year-olds) – 3.80 x 10-1 (infants aged 6 to 11 months) |
| Aromatherapy (systemic exposure by the inhalation route) | 100% | 2.24 x 10-1 (adults) – 6.46 x 10-1 (infants aged 0 to 5 months) |
| Mixing, loading and applying all-purpose cleaner (dermal route) | 1% | 5.00 x 10-2 (adults) |
| Exposure from contacting cleaned floors (dermal route) | 1% | 1.83 x 10-2 (1- to 2-year-olds) |
| Exposure from contacting cleaned floors (non-dietary oral route) | 1% | 1.38 x 10-3 (1- to 2-year-olds) |
| Mixing, loading, washing and hanging hand-washed laundry (dermal route) | 5% | 5.43 x 10-1 (adults) |
a Calculation details are in Appendix E.
7.4.2 Health effects assessment of sandalwood oil
Hazard assessment of sandalwood oil
Sandalwood oil has been approved as generally recognized as safe for use in food as a flavouring ingredient by the Flavor and Extract Manufacturers Association (FEMA) (Hall and Oser 1965). The US FDA approved sandalwood oil as a natural flavouring substance and natural substance used in conjunction with flavours (21CFR172.510). Limited empirical health effects information is available for sandalwood oil.
Lactating Swiss albino mice were administered 5 and 10 µl pure sandalwood oil/animal/day (138.9 and 277.7 mg/kg bw/day respectively) by gavage from day 1 of lactation for 14 or 21 days (Chaabra and Rao 1993). A significant decrease in hepatic cytochromes P-450 content was noted in pups and dams treated with 10 µl of sandalwood oil for 21 days. A decrease in hepatic cytochromes P-450 may indicate an inhibitory effect of sandalwood oil on cytochrome enzymes in the liver. From these results, it is not possible to determine which enzyme specifically is modified by sandalwood oil. Based on these results, sandalwood oil seems to pass through milk and may inhibit the hepatic xenobiotic metabolizing system in dams and pups (Chaabra and Rao 1993).
No sensitization reaction was observed when sandalwood oil was applied dermally at a concentration of 10% in petrolatum to 25 volunteers (Opdyke 1974).
Photoallergic reactions to 2% sandalwood oil were seen in 3 cases of 138 patients who were photopatch tested (Fotiades et al. 1995). In Japan, East Indian sandalwood oil is regarded as a high-risk skin sensitizer and potential cause of pigmented contact dermatitis (Nakayama 1998). Because of the risk of photoallergic reactions in people from Japan, a maximum use level of 2% is recommended (Tisserand and Young 2014).
Sandalwood oil was negative in spore Rec assay using H17 Rec+ and M45 Rec- in the presence or absence of metabolic activation (S9) (Burdock and Carabin 2008). Sandalwood was not cytotoxic or genotoxic in breast epithelial cells (Ortiz et al. 2016). In addition, sandalwood oil contains no known carcinogens (Burdock and Carabin 2008).
In order to inform the risk assessment, the hazard information available for the main components of sandalwood oil, i.e., alpha-santalol (41% to55%) and beta-santalol (24% to 27%), have been considered.
Hazard assessment of alpha- and beta-santalol
JECFA evaluated santalol (alpha and beta) and concluded that based on the current use levels of this compound in food there is no safety concern (WHO 2002). Alpha- and beta-santalol are permitted in the United States as synthetic flavouring substances or adjuvants that may be safely used in food in accordance with all the principles of good manufacturing practice (21CFR172.515).
The Research Institute for Fragrance Materials (RIFM) assessed the group of cyclic and non-cyclic terpene alcohols as fragrance ingredients, which covered some 30 substances classified as monoterpene, sesquiterpene and diteperne alcohols (Belsito et al. 2008). RIFM determined that all substances have close structural relationships and similar biochemical and toxicity profiles and participated in the same pathways of metabolic detoxification. Sufficient data were available from farnesol, bisabolol, linalool, menthol and alpha-terpineol (i.e., compounds that contain all key structural elements and potential sites of metabolism of all other members in the group) to demonstrate that the non-cyclic and cyclic terpenes share common metabolic pathways. A systemic NOAEL of 50 mg/kg bw/day was determined by RIFM to quantify human health risks from use of terpene alcohols. It was based on the lowest NOAEL from all available studies and was derived from an oral repeated dose toxicity study on linalool and citronellol (Belsito et al. 2008). The systemic NOAEL used by RIFM is based on a NOEL of 50 mg/kg bw/day from a repeated dose study on linalool reviewed in 2020 by ECCC and HC (ECCC, HC 2020). In this study, rats were given diets containing equal parts of citronellol and linalool (50 mg of each substance) for 12 weeks (ECCC, HC 2020). Because the study is based on a single dose of a mixture of citronellol and linalool, this dose is not considered to be an accurate NOAEL for assessing the human health risk of alpha- and beta-santalol.
In the absence of hazard data on these substances, a read-across approach was taken, and hazard information on the read-across analogue, bisabolol, was used to inform the hazard assessment.
Alpha- and beta-santalol are organic compounds classified as bicyclic sesquiterpene alkene alcohols and isomers of each other (97.4% of structural similarity on OECD QSAR Toolbox [2019]). Bisabolol, also known as levomenol, is a natural monocyclic sesquiterpene alkene alcohol. The target substances and the analogue are structurally similar, comprising a cyclic skeleton with a side chain including a double bond and a hydroxyl group. Bisabolol shows 75.5% structural similarity with alpha-santalol and 77% structural similarity with beta-santalol based on OECD QSAR Toolbox (2019). Sandalwood oil and bisabolol tested negative for mutagenicity in spore Rec assay using H17 Rec+ and M45 Rec- strains with their spores, and in Ames test, respectively. They also both tested negative for genotoxicity in breast epithelial cells (sandalwood oil) and in chromosome aberration assays using Chinese hamster V79 cells (bisabolol). Alpha- and beta-santalol and bisabolol are categorized as Class I under the Cramer classification. Class I substances are defined as substances of simple chemical structure with known metabolic pathways and innocuous end products that suggest a low order of oral toxicity.
In a 1999 safety assessment of bisabolol, the Cosmetic Ingredient Review (CIR) Expert Panel concluded that bisabolol is well absorbed following dermal application and can be a penetration enhancer (CIR 2017). WHO published a safety evaluation of aliphatic acyclic and alicyclic terpenoid tertiary alcohols and bisabolol is a part of the assessment (WHO 2011). WHO reviewed all studies available for bisabolol and determined a maternal and developmental NOAEL of 980 mg/kg bw/day (WHO 2011). WHO acknowledged the inhibitory effect of bisabolol on four human cytochrome P450 (CYP) enzymes in the liver (WHO 2011).
In a 28-day dermal toxicity study, bisabolol in an olive oil vehicle was applied dermally in a semi-occlusive dressing at doses of 50 (1%), 200 (4%), and 1000 (20%) mg/kg bw/day to the clipped skin of 10 Wistar rats (five each sex). Rats were exposed for 6 hours a day, 7 days a week, for 4 weeks (28 days). No treatment-related effects were noted in rats at 50 and 200 mg/kg bw/day. Transient moderate erythema and diffuse scale formation were noted in some female rats in the high-dose group. A significant increase in serum glucose in both sexes in the high-dose group and in serum calcium concentrations in high-dose males was observed. A significant decrease in mean absolute liver weight was noted in high-dose females and an increase in mean relative testes weight was noted in high-dose males. The changes were considered to result from the significant decreased mean terminal body weight of female and male high-dose rats by authors. Decreased liver weight may be an indication of inhibition of hepatic enzyme activity. Based on these results, the NOAEL for dermal effects was determined to be 200 mg/kg bw/day or 4% by authors (Bhatia et al. 2008b).
In a developmental toxicity study, pregnant rats received bisabolol (98%) daily via oral gavage on GD 6 through GD 15 at doses of 0, 0.250, 0.500, 1.0, or 3.0 ml/kg bw/day (equivalent to 0, 245, 490, 980 or 2940 mg/kg bw/day) (WHO 2011; Bhatia et al. 2008b). No effects on prenatal development were observed at doses up to 1.0 ml/kg (960 mg/kg bw/day). A significant reduction in fetal number and a subsequent increase in resorption rate were observed in the 3.0 ml/kg (2940 mg/kg bw/day) group. No deformities were noted. Slight sedation, ataxia, reduced feed intake, and reduction of body weight gain were observed in females in this dose group (2940 mg/kg bw/day). No information about the liver was provided. Based on these results, a NOAEL for maternal toxicity and developmental toxicity was determined at 980 mg/kg bw/day by the authors (WHO 2011; Bhatia et al. 2008b).
In a similar study, pregnant rabbits received 0, 0.3, 1.0, or 3.0 ml/kg bw/day (equivalent to 0, 294, 980 or 2940 mg/kg bw/day) bisabolol by gavage on GD 6 through GD 18 (WHO 2011; Bhatia et al. 2008b). Fetuses were removed on day 30 and examined. No adverse effects on either prenatal development or on the dams were noted at doses up to 1.0 ml/kg (980 mg/kg bw/day). A reduction in the number of fetuses was noted in the 3.0 ml/kg (2940 mg/kg bw/day) group; no dead fetuses or deformities were noted. Dams in this treatment group showed slight sedation and had reduced body weight gains. No information about the liver was reported. On the basis of these results, a NOAEL for maternal toxicity and developmental toxicity was determined at 980 mg/kg bw/day by authors (WHO 2011; Bhatia et al. 2008b).
The inhibitory effect of bisabolol on cytochromes P-450 such as CYP2D6, CYP1A2, CYP2C9 or CYP3A4 has been determined (Tisserand and Young 2014; WHO 2011). Cytochromes are the major enzymes involved in drug metabolism, accounting for about 75% of total metabolism. Most drugs undergo deactivation by cytochromes, either directly or by facilitated excretion from the body. Also, many substances are bioactivated by cytochromes to form their active compounds (Guengerich 2008). By inhibiting CYP2D6, CYP1A2, CYP2C9 or CYP3A4, bisabolol may induce a risk of interaction with drugs metabolized by these enzymes such as tricyclic antidepressants, antiarrhythmics, analgesics, antidiabetic, antiepileptic, antipsychotic, or chemotherapeutic drugs (Tisserand and Young 2014). Based on the decrease in hepatic cytochromes P-450 caused by sandalwood oil seen previously in Chaabra and Rao (1993), sandalwood oil and its analogue, bisabolol, which may increase the probability to induce adverse effects in vulnerable populations such as individuals with pre-existing health conditions.
In an Ames test using Salmonella typhimurium strains TA97a, TA98, TA100 and TA1535 with and without S9 activation conducted with bisabolol (in ethanol) at doses of 1.5 to 5000 µg/plate, no mutagenicity was observed (Bhatia et al. 2008b). Bisabolol was tested in the chromosome aberration assay using Chinese hamster V79 cells. Cells were incubated with 7.81, 15.63 or 31.25 µg bisabolol/ml in the presence of metabolic activation (S9) or 0.78, 1.56, or 3.13 µg bisabolol/ml without activation. In the second experiment, cells were incubated with 10, 20, 30 or 40 µg bisabolol/ml with activation or 2, 3 or 4 µg bisabolol/ml without activation. Bisabolol was negative in the assay (WHO 2011; Bhatia et al. 2008b).
7.4.3 Characterization of risk to human health
In the absence of hazard data on sandalwood oil and its main components, alpha- and beta-santalol, health effects information on the analogue bisabolol was used to characterize the risk to sandalwood oil.
A NOAEL of 200 mg/kg bw/day was selected from the short-term dermal study on bisabolol for risk characterization based not only on the critical health effects seen at 1000 mg/kg bw/day, such as decreased body weight gain, decreased feed efficiency, transient moderate skin erythema and diffuse scale formation in some females, decreased absolute live weight in females, and increased in relative testes weight in males in Bhatia et al. (2008b) but also on the decrease in hepatic cytochrome P-450 seen at 277.7 mg/kg bw/day sandalwood oil but not at 138.9 mg/kg bw/day in Chaabra and Rao (1993).
For the oral and inhalation routes of exposure, the NOAEL of 980 mg/kg bw/day from two developmental oral studies were used to characterize risk based on the critical health effects of slight sedation, ataxia, reduced feed intake, and reduction of body weight gain in females a significant reduction in fetal number and an increase in resorption rate (Bhatia et al. 2008b).
Daily exposure estimates and resulting MOEs are summarized in Table 7-7.
| Exposure scenarioa | Exposure (mg/kg bw/day) | MOEb |
|---|---|---|
| Food flavouring agent (dietary intake) (1-year-olds and older) | 1.20 x 10-4 | 416667 |
| Dermal exposure from body lotion (30%) (all subpopulations) | 40.5 (adults) – 95.2 (infants) | 2 (infant) – 5 (adults) |
| Dermal exposure from massage oil (3%) (all subpopulations) | 1.30 (adults) – 8.57 (infants) | 23 (infants) – 154 (adults) |
| Dermal exposure from facial moisturizer (3%) (14- to 18-year-olds; adults) | 7.26 x 10-1 (14- to 18-year-olds) – 1.22 (adults) | 164 (adults) – 276 (14- to 18-year-olds) |
| Dermal exposure from the essential oil used as a body fragrance (100%) (14- to 18-year-olds; 2- to 3-year-olds) | 7.45 (14- to 18-year-olds) – 22 (2- to 3-year-olds) | 9 (2- to 3-year-olds) – 27 (14- to 18-year-olds) |
| Dermal exposure from a shampoo product (10%) (all subpopulations) | 1.75 x 10-1 (adults) – 6.19 x 10-1 (infants aged 0 to 5 months) | 323 (infants aged 0 to 5 months) – 1140 (adults) |
| Dermal exposure from acne medication (facial cleansing system) (2%) (9- to 13-year-olds; adults) | 7.39 x 10-1 (9- to 13-year-olds) – 1.23 (adults) | 163 (adults) – 271 (9- to 13-year-olds) |
| Dermal exposure from sunscreen (0.04%) (all subpopulations) | 8.40 x 10-2 (9- to 13-year-olds) – 3.80 x 10-1 (infants aged 6 to 11 months) | 527 (infants aged 6 to 11 months) – 2381 (9- to 13-year-olds) |
| Dermal exposure from mixing, loading, and application of an all-purpose floor cleaner (1%) (adults) | 5.00 x 10-2 (adults) | 4000 (adults) |
| Dermal exposure from contacting cleaned floors (1%) (1- 2-year-olds) | 1.83 x 10-2 (1- to 2-year-olds) | 10 900 (1- 2-year-olds) |
| Systemic exposure by the non-dietary oral routes from contacting cleaned floors (1%) (1- 2-year-olds) | 1.38 x 10-3 (1- 2-year-olds) | > 700 000 (1- 2-year-olds) |
| Dermal exposure from mixing, loading, washing and hanging hand-washed laundry (5%) (adults) | 5.43 x 10-1 (adults) | 369 (adults) |
| Systemic exposure by the inhalation route from aromatherapy (100%) (all subpopulations) | 2.24 x 10-1 (adults) – 8.00 x 10-1 (1-year-olds) | 1225 (1-year-olds) – 4366 (adults) |
a Exposure scenario parameters and calculations for sandalwood oil are outlined in Appendix E. Dermal absorption was assumed to be 100%.
b For dermal exposure scenarios, the critical effect level (200 mg/kg bw/day) from a dermal short-term study is based on a decrease in body weight gain, decrease in feed efficiency, transient moderate skin erythema and diffuse scale formation in some females, decrease in absolute liver weight in females and increase in relative testes weight in males (Bhatia et al. 2008b). For inhalation and oral exposure scenarios, the critical effect level (980 mg/kg bw/day) from two oral developmental studies is based on slight sedation, ataxia, reduced feed intake, and reduction of body weight gain in females and a significant reduction in fetal number and increase in resorption rate (Bhatia et al. 2008b).
The MOEs between the critical effect level and the estimate of daily exposure to sandalwood oil from a body lotion, massage oil (i.e., infants and children up to 8 years old) and from use of the essential oil as a body fragrance are considered potentially inadequate to account for uncertainties in the health effects and exposure data.
For all other scenarios, the MOEs between the critical effect level and the estimate of exposure ranged from 154 to 10 667 646 and are considered to be adequate to account for uncertainties in the health effects and exposure data.
7.4.4 Uncertainties in evaluation of risk to human health
The key sources of uncertainty are presented in the table below.
| Key source of uncertainty | Impact |
|---|---|
| Hazard | N/A |
| There are no chronic, reproductive/developmental, genotoxicity or carcinogenicity animal studies for all routes of exposure for sandalwood oil and its main components (alpha- and beta-santalol). | +/- |
| In the absence of hazard information for sandalwood oil, and its main components (alpha- and beta-santalol), the read-across analogue, bisabolol, was used to inform the risk characterization. | +/- |
+ = uncertainty with potential to cause over-estimation of exposure/risk; - = uncertainty with potential to cause under-estimation of exposure risk; +/- = unknown potential to cause over or under-estimation of risk
7.5 Guaiazulene
7.5.1 Exposure assessment of guaiazulene
Environmental media
Given there are no reports of import or manufacturing above 100 kg in Canada and in consideration of the low quantities of the substance reported to be used in Canada (Environment Canada 2013), exposure to guaiazulene from environmental media, including drinking water, is not expected.
Indoor air was sampled for guaiazulene in 54 homes in Nunavik, Canada, during the winter (January to April 2018). However, it was not detected in any of the samples. The limit of quantification for guaiazulene was 1.5 ng (approximately equal to 0.34 ng/m3) (Won 2019).
Products available to consumers
Guaiazulene is present in products available to consumers. Exposure to guaiazulene from the use of a body lotion, facial moisturizer, conditioner, hair perm or straightening product, and aftershave were considered to be the sentinel scenarios for dermal applications. The data for guaiazulene are summarized in Appendix F (Tables F-1 to F-5).
The highest daily exposures to guaiazulene are expected to occur from the use of a body lotion with a reported upper concentration of 0.1% guaiazulene, a facial moisturizer with a reported upper concentration of 0.3%, a conditioner with a reported upper concentration of 1%, a hair perm or straightening product with a reported upper concentration of 1%, and an aftershave product with a reported upper concentration of 1% (personal communication, email communication from the Consumer and Hazardous Products Safety Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, October 2019; unreferenced).
Exposure is expected to be predominantly via the dermal route, but inhalation exposure was also quantified since guaiazulene is considered to be volatile.
Exposure estimates from products available to consumers for guaiazulene are summarized in Table 7-9.
| Scenario | Percent concentration in product | Dermal exposure (mg/kg bw/day)a | Inhalation exposure (mg/m3)a |
|---|---|---|---|
| Body lotion | 0.1% | 1.35 x 10-1 (adults) – 3.17 x 10-1 (infants aged 0 to 5 months) | 3.70 x 10-2 (adults) – 6.80 x 10-3 (infants aged 0 to 5 months) |
| Facial moisturizer | 0.3% | 1.22 x 10-1 (adults) – 7.86 x 10-2 (9- to 13-year-olds) | 2.70 x 10-2 (9- to 13-year-olds) – 4.30 x 10-2 (adults) |
| Conditioner | 1% | 1.95 x 10-2 (adults) – 3.47 x 10-2 (2- to 3-year-olds) | 1.10 x 10-2 (2- to 3-year-olds) – 4.10 x 10-2 (adults) |
| Hair perm straightening product | 1% | 2.16 (adults) – 5.74 (4- to 8-year-olds) | 7.50 x 10-2 (4- to 8-year-olds) – 1.40 x 10-1 (adults) |
| Aftershave | 1% | 3.24 x 10-1 (adults) – 5.48 x 10-1 (9- to 13-year-olds) | 7.70 x 10-3 (9- to 13-year-olds) – 1.20 x 10-2 (adults) |
a Calculation details are in Appendix F.
7.5.2 Health effects assessment of guaiazulene
The only health effects study available on guaiazulene showed in vitro cytotoxic and anti-proliferative activity but no genotoxic effects in healthy rat neuron cells (Togar et al. 2014).
In the absence of health effects data on guaiazulene, a read-across approach was taken, and hazard information on the analogue, naphthalene, was used to inform the hazard assessment.
Guaiazulene is an organic compound that is an alkylated derivative of azulene with an almost identical intensely blue colour. Guaiazulene and azulene have very similar structures (CIR 1999). Azulene is an organic compound and an isomer of naphthalene (CIR 1999). By moving one bond from the cycloheptatriene ring to the cyclopentadiene ring at a very high temperature, which may occur during a high temperature processing, azulene rearranges naturally to form naphthalene, which consists of a fused pair of benzene rings (CIR 1999). Naphthalene and guaiazulene show 87.1% structural similarity on OECD QSAR Toolbox (2019). All have comparable physicochemical properties and mechanistic profiles and tend to form a few common metabolites (using rat liver S9 metabolic simulator) that have structural features associated with the potential to act as developmental or reproductive toxicants. Guaiazulene and naphthalene have been classified in Class III under the Cramer classification. Class III substances are defined as substances with chemical structures that permit no strong initial impression of safety and that may suggest significant toxicity.
EC and HC have concluded that naphthalene was not genotoxic or carcinogenic and determined identical NOAELs and LOAELs (EC, HC 2008; US EPA 2008).
For oral exposure, a NOAEL of 100 mg/kg bw/day was identified based on significant decreases in body weights/body weight gains at 200 mg/kg bw/day (LOAEL) in a 13-week rat study (EC, HC 2008; US EPA 2008). In this study, 10 male and 10 female rats were administered naphthalene by gavage at doses of 0, 25, 50, 100, 200, or 400 mg/kg. Body weight gain decrements exceeding 10% without food intake being affected in both males and females administered 200 or 400 mg/kg bw/day and lesions in exposed male kidneys and exposed female thymuses were observed (EC, HC 2008; US EPA 2008).
For dermal exposure, a NOAEL of 300 mg/kg bw/day was identified based on a 90-day dermal toxicity study in rats where effects were noted only at the highest dose tested of 1000 mg/kg bw/day (EC, HC 2008; US EPA 2008). These effects included excoriated skin and papules in both sexes and atrophy of seminiferous tubules in males (EC, HC 2008; US EPA 2008). Other effects observed included non-neoplastic lesions in the cervical lymph node (hyperplasia), liver (hemosiderosis), thyroid (thyroglossal duct cysts), kidneys (pyelonephritis), urinary bladder (hyperplasia) and skin (acanthosis, hyperkeratosis) in females. As effects were seen only at the limit dose, the US EPA concluded that dermal toxicity is not likely a concern (EC, HC 2008; US EPA 2008).
In a 13-week whole body study in rats, a LOAEC of 10 mg/m3 was identified for inhalation exposure by the US EPA based on increased incidence and severity of nasal lesions (degeneration, atrophy and hyperplasia of basal cells of the olfactory epithelium; rosette formation of the olfactory epithelium; loss of bowman’s glands; hypertrophy of respiratory epithelium) (EC, HC 2008; US EPA 2008).
Environment Canada and Health Canada published a screening assessment for naphthalene in 2008 and concluded that naphthalene may pose a risk to human health (EC, HC 2008). The US EPA has classified naphthalene as Group C (“possible human carcinogen”) based on inadequate human carcinogenicity data and limited evidence of carcinogenicity after oral and inhalation exposures in experimental animals (US EPA 2008). The International Agency for Research on Cancer (IARC) has classified naphthalene as “possibly carcinogenic to humans” (Group 2B) on the basis of “inadequate evidence in humans” and “sufficient evidence in experimental animals” for determination of carcinogenicity (IARC 2002). In addition, it was considered that the risk of carcinogenicity by the inhalation route is lower than the risk of toxicity for naphthalene because of the weakness of the evidence that it may be carcinogenic in humans (observations of benign respiratory tumours in rodents observed at the highest dose, 157 mg/m3, compared to nasal lesions observed at the LOAEC of 10 mg/m3 determined by EC and HC) (EC, HC 2008).
A 90-day (6 hours/day, 5 days/week) inhalation study was conducted at 0, 0.1, 1, 10, and 30 ppm naphthalene vapour (0, 0.52, 5.2, 52, and 156 mg/m3 respectively) on Fisher 344 rats (10/group/sex) following US EPA guidelines, with a 4-week recovery group for rats exposed to 10 ppm (Dodd et al. 2012). Naphthalene exposure concentrations were measured by gas chromatography–mass spectrometry, and aerosol testing verified that solid particles were not present. A decrease in body weight and food/water consumption was observed only at 30 ppm. The absolute mean weights of the spleen, left testis, and thymic region of male rats exposed to 10 or 30 ppm were significantly lower than the control mean values, but no difference was noted for relative organ weights. In female rats, the absolute mean weights of the heart, liver, and thymic region were statistically significantly lower for all groups, but the magnitude of the decreases was not concentration-dependant. Relative to body weight, they were similar to control values. All statistically significant differences in organ weight values (absolute, relative to body weight or to brain) observed immediately following a 10 ppm exposure for 90 days were diminished in magnitude and were no longer statistically significantly different from control values following a 4-week recovery period. Mild hyperplasia and squamous metaplasia were observed in the respiratory epithelium of rats exposed to 10 or 30 ppm. Lesions in the olfactory epithelium were observed only in rats of the 10 or 30 ppm groups and consisted of degeneration, necrosis, areas of re-epithelialization and basal cell hyperplasia. Residual olfactory epithelial degeneration and basal cell hyperplasia were still evident after the 4-week recovery period at 10 ppm. No naphthalene-related effects were observed at 0.1 ppm or 1 ppm. A NOAEC of 5.2 mg/m3 (1 ppm) was determined based on atrophy/disorganization of the olfactory epithelium and hyperplasia of the respiratory and transitional epithelium observed at 52 mg/m3 (10 ppm) by study authors (Dodd et al. 2012; ECHA Registration dossier 2019a).
Although the mechanism is still not fully elucidated, EC, HC (2008) and US EPA (2008) reported that nongenotoxic mechanisms have been proposed for the possible carcinogenicity of naphthalene. EC, HC (2008) reported that the carcinogen effect was observed only in mice exposed to up to 157 mg/m3 and in rats exposed to 314 mg/m3 for 2 years in a National Toxicology Program (NTP) inhalation toxicity study. No difference in neoplastic effect was observed between control groups and the lowest dose, 52 mg/m3, in mice and rats (EC, HC 2008). The LOAEC for the inhalation route was determined to be 10 mg/m3 (Dodd et al. 2012), which is significantly lower than the dose at which carcinogenic effects were observed.
7.5.3 Characterization of risk to human health
Exposure of the general population to guaiazulene may occur through the use of products available to consumers (i.e., lotions, hair removal creams, conditioners, and aftershaves). Exposure is expected to be predominantly via the dermal route and possibly via the inhalation route.
For the dermal route of exposure, a NOAEL of 300 mg/kg bw/day was selected (EC, HC 2008; US EPA 2008).
For the inhalation route of exposure, a NOAEC of 5.2 mg/m3 was identified based on increased incidence and severity of nasal lesions (degeneration, atrophy and hyperplasia of basal cells of the olfactory epithelium; rosette formation of the olfactory epithelium; loss of Bowman’s glands; hypertrophy of respiratory epithelium) at a concentration of 10 mg/m3 (Dodd et al. 2012). As the toxic effects of guaiazulene were observed in the respiratory tract, it was considered appropriate to directly compare peak air concentrations from the exposure scenario to the NOAEC of 5.2 mg/m3. The non-carcinogenic NOAEC of 5.2 mg/m3 from naphthalene used to characterise the risk for guaiazulene is considered very conservative so it is not necessary to characterize the possible carcinogenicity of guaiazulene.
Table 7‑10 provides the relevant estimates of exposure to guaiazulene and the resultant MOEs.
| Exposure scenarioa | Exposure | MOEb |
|---|---|---|
| Dermal exposure from use of body lotion (0.1%) (all subpopulations) | 1.35 x 10-1 (adults) – 3.17 x 10-1 mg/kg bw/day (infants aged 0 to 5 months) | 950 (infants aged 0 to 5 months) – 2220 (adults) |
| Inhalation exposure from use of body lotion (0.1%) (all subpopulations) | 6.80 ×10⁻3 (infants aged 0 to 5 months) – 3.70 ×10⁻2 (adults) mg/m3 | 141 (adults) – 765 (infants aged 0 to 5 months) |
| Dermal exposure from use of facial moisturizer (0.3%) (9- to 13-year-olds; adults) | 7.26 x 10-2 (14- to 18-year-olds) – 1.22 x 10-1 (adults) mg/kg bw/day | 2467 (adults) – 4133 (14 to 18-year-olds) |
| Inhalation exposure from use of facial moisturizer (0.3%) (9- to 13-year-olds; adults) | 2.70 x 10-2 (9- to 13-year-olds) – 4.30 x 10-2 (adults) mg/m3 | 121 (adults) – 193 (9- to 13-year-olds) |
| Dermal exposure from use of conditioner (1%) (2- to 3-year-olds; adults) | 1.95 x 10-2 (adults) – 3.47 x 10-2 (2- to 3-year-olds) mg/kg bw/day | 8654 (2- to 3-year-olds) – 15406 (adults) |
| Inhalation exposure from use of conditioner (1%) | 1.10 x 10-2 (2- to 3-year-olds) – 4.10 x 10-2 (adults) mg/m3 | 127 (adults) – 473 (2- to 3-year-olds) |
| Dermal exposure from use of a hair perm or straightening product (1%) (4- to 8-year-olds; adults) | 2.16 (adults) – 5.74 (4- to 8-year-olds) mg/kg bw/day | 52 (4- to 8-year-olds) – 139 (adults) |
| Inhalation exposure from use of a hair perm or straightening product (1%) | 7.50 x 10-2 (4- to 8-year-olds) – 1.40 x 10-1 (adults) mg/m3 | 37 (adults) – 69 (4- to 8-year-olds) |
| Dermal exposure from an aftershave product (1%) (9- to 13-year-olds; adults) | 3.24 x 10-1 (adults) – 5.48 x 10-1 (9- to 13-year-olds) | 548 (9- to 13-year-olds) – 925(adults) |
| Inhalation exposure from an aftershave product (1%) (9- to 13-year-olds; adults) | 7.70 x 10-3 (9- to 13-year-olds) – 1.20 x 10-2 (adults) | 433 (adults) – 675 (9- to 13-year-olds) |
a Exposure scenario parameters and calculation for guaiazulene are outlined in Appendix F.
b For dermal exposure scenarios, the critical effect level of 300 mg/kg bw/day (NOAEL) from a 90-day dermal study with naphthalene is based on atrophy of seminiferous tubules in males, and non-neoplastic lesions in the cervical lymph node (hyperplasia), liver (hemosiderosis), thyroid (thyroglossal duct cysts), kidneys (pyelonephritis), urinary bladder (hyperplasia) and skin (acanthosis, hyperkeratosis) in females. For inhalation exposure scenarios, the critical effect level of 5.2 mg/m3 (NOAEC) from a subchronic (nose-only) neurotoxicity rat study with naphthalene is based on atrophy/disorganization of the olfactory epithelium and hyperplasia of the respiratory and transitional epithelium.
The MOE between the critical effect level and the estimate of daily exposure to guaiazulene by the dermal or inhalation route from a hair perm or straightening product is considered potentially inadequate to account for uncertainties in the health effects and exposure data.
For all other scenarios, the MOEs between the critical effect level and estimate of exposure ranged from 121 to 4133 and are considered adequate to account for uncertainties in the health effects and exposure data.
7.5.4 Uncertainties in evaluation of risk to human health
The key sources of uncertainties are presented in the table below.
| Key source of uncertainty | Impact |
|---|---|
| Hazard | N/A |
| There is no hazard data available for guaiazulene. The read-across analogue, naphthalene, was used to inform the risk assessment. | +/- |
+ = uncertainty with potential to cause over-estimation of exposure/risk; - = uncertainty with potential to cause under-estimation of exposure/risk; +/- = unknown potential to cause over or under-estimation of risk.
8. Conclusion
Considering all available lines of evidence presented in this draft screening assessment, there is low risk of harm to the environment from the substances in the Monocyclic and Bicyclic Sesquiterpenes Group. It is proposed to conclude that the 16 substances in the Monocyclic and Bicyclic Sesquiterpenes Group do not meet the criteria under paragraphs 64(a) or (b) of CEPA as they are not entering the environment in a quantity or concentration or under conditions that have or may have an immediate or long-term harmful effect on the environment or its biological diversity or that constitute or may constitute a danger to the environment on which life depends.
On the basis of the information presented in this draft screening assessment, it is proposed to conclude that bisabolene, copaiba balsam, ginger oil, alpha-bisabolene, sandalore, santol pentenol, beta-caryophyllene, guaiene, alpha-guaiene, valencene, guaiol, elemol, and bulnesol do not meet the criteria under paragraph 64(c) of CEPA as they are not entering the environment in a quantity or concentration or under conditions that constitute or may constitute a danger in Canada to human life or health.
On the basis of the information presented in this draft screening assessment, it is proposed to conclude that T & T clove oil, sandalwood oil, and guaiazulene meet the criteria under paragraph 64(c) of CEPA as they are entering or may enter the environment in a quantity or concentration or under conditions that constitute or may constitute a danger in Canada to human life or health.
Therefore, it is proposed to conclude that T & T clove oil, sandalwood oil, and guaiazulene meet one or more of the criteria set out in section 64 of CEPA and that the remaining 13 substances in the Monocyclic and Bicyclic Sesquiterpenes Group do not meet any of the criteria set out in section 64 of CEPA.
It is also proposed that T & T clove oil and guaiazulene meet the persistence and bioaccumulation criteria and that sandalwood oil does not meet the persistence or bioaccumulation criteria as set out in the Persistence and Bioaccumulation Regulations of CEPA.
References
[21CFR172.510] Code of Federal Regulations, Title 21 – Food and Drugs, Part 172 Food Additives Permitted for Direct Addition to Food for Human Consumption, Section 172.510 Synthetic flavoring substances and adjuvants. Revised as of April 1, 2019.
[21CFR172.515] Code of Federal Regulations, Title 21 – Food and Drugs, Part 172 Food Additives Permitted for Direct Addition to Food for Human Consumption, Section 172.515 Synthetic flavoring substances and adjuvants. Revised as of April 1, 2019.
[ACI] American Cleaning Institute [database]. 2017. Washington (DC): ACI [accessed 2017 December].
[ACI] American Cleaning Institute [database]. 2018. Washington (DC): ACI [accessed 2018 December].
[AFSSAPS] Agence Française de Sécurité Sanitaire des Produits de Santé. 2008. Recommandations relatives aux critères de qualité des huiles essentielles : contribution pour l’évaluation de la sécurité des produits cosmétiques contenant des huiles essentielles [PDF]. Saint-Denis : AFSSAPS (editor), collection Recommandations, 17 p., FRA.
Almeida MR, D’Arc Castania Darin J, Hernandes LC, Freiman de Souza Ramos M, Greggi Antunes LM, de Freitas O. 2012. Genotoxicity assessment of copaiba oil and its fractions in Swiss mice. Genet Mol Biol. 35(3):664-672.
Andersen FA 1999. Final report on the safety assessment of azulene. Int J Toxicol. 18(3 suppl):27-32.
Api AM, Belsito D, Bhatia S, Botelho D, Browne D, Bruze M, Burton Jr GA, Buschmann J, Calow P, Dagli ML, et al. 2016. RIFM fragrance ingredient safety assessment, elemol, CAS Registry Number 639-99-6. Food Chem Toxicol. 110:S16-S21.
Arctander S. 1960. Perfume and flavour material of natural origin. Elizabeth, NJ.
Belsito D, Bickers D, Bruze M, Calow P, Greim P, Hanifin JM, Rogers AE, Saurat JH, Sipes IG, Tagami H. 2008. A toxicologic and dermatologic assessment of cyclic and non-cyclic terpene alcohols when used as fragrance ingredients. Food Chem Toxicol. 46:S1-S71.
Bhatia SP, Letizia CS, Api AM. 2008a. Fragrance material review on alpha-terpineol. Food Chem Toxicol. 46:S280-S285.
Bhatia SP, McGinty D, Letizia CS, Api AM. 2008b. Fragrance material review on alpha-bisabolol. Food Chem Toxicol. 46:S72-S76.
Biapa Nya PC, Kuiate JR, Wankeu M, Ntiokam D. 2010. Acute and subacute toxicity studies of Zinginber Officinalis roscoe essential oil on mice (Swiss) and rats (Wistar). Afr J Pharm Pharmacol. 1:39-49.
Bledsoe JO. 2000. Terpenoids. In: Kirk‐Othmer, editor. Encyclopedia of Chemical Technology. 5th ed. Hoboken (NJ): Wiley.
Brand JE, Fox JED, Pronk G, Cornwell C. 2007. Comparison of oil concentration and oil quality from Santalum spicatum and S. album plantations, 8-25 years old, with those from mature S. spicatum natural stands. Aust For. 70(4):235-41.
Burdock GA, Carabin IG. 2008. Safety assessment of sandalwood oil (Santalum album L.). Food Chem Toxicol. 46:421-432.
Burdock GA. 2010. Fenaroli’s handbook of flavor ingredients. 6th ed. Boca Raton (FL): CRC Press.
Canada. [1978]. Food and Drug Regulations. C.R.C., c.870.
Canada. 1999. Canadian Environmental Protection Act, 1999. S.C. 1999, c.33. Canada Gazette Part III, vol. 22, no. 3.
Canada, Dept. of the Environment. 2012. Canadian Environmental Protection Act, 1999: Notice with respect to certain substances on the Domestic Substances List [PDF]. Canada Gazette, Part 1, vol. 146, no. 48, Supplement.
Caputi L, Aprea E. 2011. Use of terpenoids as natural flavouring compounds in food industry. Recent Pat Food Nutr Agric. 3:9-16.
Chaabra SK, Rao AR. 1993. Postnatal modulation of xenobiotic metabolizing enzymes in liver of mouse pups following transactional exposure to sandalwood oil. Nutr Res. 13,1191–1202.
ChemCAN [level III fugacity model of 24 regions of Canada]. 2003. Version 6.00. Peterborough (ON): Trent University, Canadian Centre for Environmental Modelling and Chemistry.
[CIR] Cosmetic Ingredient Review. 2017. Bisabolol. Int J Toxicol. 36:24S-25S.
[CIR] Cosmetic Ingredient Review. 1999. Final report on the safety assessment of azulene. Int J of Toxicol. 18(suppl.3):27-32.
[COSING] European Commission Database for Information on Cosmetic Substances and Ingredients. 2018. European Cosmetic ingredient inventory [database]. Brussels (BE): European Commission Cosmetics Directive [accessed 2018 December].
[CTFA] Cosmetic, Toiletry and Fragrance Association. 1983. Summary for the results of surveys of the amount and frequency of use of cosmetic products by women. Technical report. Washington (DC): CTFA.
da Silva Oliveira GL, Conceiçao Machado K, Conceiçao Machado K, Paula dos Santos C.L. da Silva A, Mendes Feitosa C, de Castro Almeida FR. 2018. Non-clinical toxicity of β-caryophyllene, a dietary cannabinoid: Absence of adverse effects in female Swiss mice. Regul Toxicol Pharmacol. 92:338-346.
DeSesso JM, Scialli AR. 2018. Bone development in laboratory mammals used in developmental toxicity studies. Birth Defects Res. 110:1157-1187.
Di Sotto A, Mazzanti G, Carbone G, Hreila P, Maffei F. 2010. Inhibition by β-caryophyllene of ethyl methanesulfonate-induced clastogenicity in cultured human lymphocytes. Mutat Res. 699:23-28.
Dodd DE, Wong BA, Gross EA, Miller RA. 2012. Nasal epithelial lesions in F344 rats following a 90-day inahaltion exposure to naphthalene. Inhal Toxicol 24:70-79.
Dolejš L, Mironov A, Šorm F. 1960. Structure of bulnesol stereochemistry of guaiol, nepetalinic acids and iridomyrmecins. Tetrahedron Letters 1(132):18-21.
[EC, HC] Environment Canada, Health Canada. 2008. Screening Assessment for the Challenge: Naphthalene (CAS RN 91-20-3). Ottawa (ON), Canada: Government of Canada.
[ECCC] Environment and Climate Change Canada. 2016a. Science approach document: ecological risk classification of organic substances. Ottawa (ON): Government of Canada.
[ECCC] Environment and Climate Change Canada. 2016b. Supporting documentation: data used to create substance-specific hazard and exposure profiles and assign risk classifications. Gatineau (QC): ECCC. Information in support of the science approach document: ecological risk classification of organic substances. Available from: substances@ec.gc.ca.
[ECCC, HC] Environment and Climate Change Canada, Health Canada. 2014. Approach for identification of chemicals and polymers as risk assessment priorities under Part 5 of the Canadian Environmental Protection Act, 1999 (CEPA 1999). Ottawa (ON): Government of Canada.
[ECCC, HC] Environment and Climate Change Canada, Health Canada. 2015. Identification of risk assessment priorities: results of the 2015 review. Ottawa (ON): Government of Canada.
[ECCC, HC] Environment and Climate Change Canada, Health Canada. [modified 2017 Mar 12]. Categorization. Ottawa (ON): Government of Canada. [accessed 2018 March].
[ECCC, HC] Environment and Climate Change Canada, Health Canada. 2018a. Rapid screening of substances with limited general population exposure [PDF]. Ottawa (ON): Government of Canada.
[ECCC, HC] Environment and Climate Change Canada, Health Canada. 2018b. Screening assessment: substances identified as being of low concern using the ecological risk classification of organic substances and the threshold of toxicological concern (TTC)-based approach for certain substances [PDF]. Ottawa (ON): Government of Canada.
[ECCC, HC] Environment and Climate Change Canada, Health Canada. 2018c. Screening Assessment Phenol, 2-methoxy-5-(2-propenyl)-(Eugenol) and Rose, Rosa canina, ext [PDF]. Ottawa (ON): Government of Canada.
[ECCC, HC] Environment and Climate Change Canada, Health Canada. 2020. Draft Screening Assessment Terpenes and Terpenoids (Acyclic/Monocyclic/Bicyclic Monoterpenes). Ottawa (ON): Government of Canada.
[ECHA Registration dossier] Registration dossier of European Chemicals Agency. 2010. Registration dossier for sandalore. Helsinki (FI): ECHA.
[ECHA Registration dossier] Registration dossier of European Chemicals Agency. 2015. Registration dossier for santol pentenol. Helsinki (FI): ECHA.
[ECHA Registration dossier] Registration dossier of European Chemicals Agency. 2018. Registration dossier for linalool. Helsinki (FI): ECHA.
[ECHA Registration dossier] Registration dossier of European Chemicals Agency. 2019a. Registration dossier for naphthalene. Helsinki (FI): ECHA.
[ECHA Registration dossier] Registration dossier of European Chemicals Agency. 2019b. Registration dossier for terpineol. Helsinki (FI): ECHA.
[EFSA] European Food Safety Authority. 2008. Flavouring Group Evaluation 25, (FGE.25): Aliphatic and aromatic hydrocarbons from chemical group 31. Scientific Opinion of the Panel on Food Additives, Flavourings, Processing Aids and Materials in Contact with Food. EFSA Journal. 918:1-109.
[EFSA] European Food Safety Authority. 2011a. Consideration of aliphatic and alicyclic and aromatic hydrocarbon evaluated by JECFA (63rd meeting) structurally related to aliphatic and aromatic hydrocarbons evaluated by EFSA in FGE.25Rev2. EFSA Journal. 13:4067.
[EFSA] European Food Safety Authority. 2011b. Aliphatic and aromatic hydrocarbons from chemical group 31. EFSA Journal. 9:2177.
[EFSA] European Food Safety Authority. 2011c. Scientific Opinion on Flavouring Group Evaluation 18, Revision 2 (FGE.18Rev2): Aliphatic, alicyclic and aromatic saturated and unsaturated tertiary alcohols, aromatic tertiary alcohols and their esters from chemical groups6 and 8. EFSA Journal. 9:1847.
[EFSA] European Food Safety Authority. 2015a. Consideration of aliphatic and alicyclic and aromatic hydrocarbon evaluated by JECFA (63rd meeting) structurally related to aliphatic and aromatic hydrocarbons evaluated by EFSA in FGE.25Rev2. EFSA Journal. 9:2178.
[EFSA] European Food Safety Authority. 2015b. Scientific Opinion on Flavouring Group Evaluation 78, Revision 2 (FGE.78Rev2): Consideration of aliphatic and alicyclic and aromatic hydrocarbon evaluated by JECFA (63rd meeting) structurally related to aliphatic and aromatic hydrocarbons evaluated by EFSA in FGE.25Rev2. EFSA Journal. 13:4067.
[EFSA] European Food Safety Authority. 2015c. Scientific Opinion on Flavouring Group Evaluation 18, revision 3 (FGE.18Rev3): Aliphatic, alicyclic and aromatic saturated and unsaturated tertiary alcohols, aromatic tertiary alcohols and their esters from chemical groups 6 and 8. EFSA Journal. 13(5):4118.
Environment Canada. 2013. DSL Inventory Update data collected under the Canadian Environmental Protection Act, 1999, section 71: Notice with respect to certain substances on the Domestic Substances List. Data prepared by: Environment Canada, Health Canada; Existing Substances Program.
[EPI Suite] Estimation Program Interface Suite for Microsoft Windows [estimation model]. c2000-2012. Ver. 4.11. Washington (DC): US Environmental Protection Agency, Office of Pollution Prevention and Toxics; Syracuse (NY): Syracuse Research Corporation.
[FCC] Food Chemicals Codex. 2018. Food Chemicals Codex, 11th Edition. Washington, D.C. National Academy Press.
Ficheux AS, Chevillotte G, Wesolek N, Morisset T, Dornic N, Bernard A, Bertho A, Romanet A, Leroy L, Mercat AC, et al. 2016. Consumption of cosmetic products by the French population. Second part: Amount data. Food Chem Toxicol. 90:130-141.
Ficheux AS, Wesolek N, Chevillotte G, Roudot AC. 2015. Consumption of cosmetic products by the French population. First part: Frequency data. Food Chem Toxicol. 78:159-169.
Florin I, Rutberg L, Curvall M, Enzell CR. 1980. Screening of tabacco smoke constituents for mutagenicity using the Ames' test. Toxicology. 15(3):219-232.
Fotiades J, Soter NA, Lim HW. 1995. Results of evaluation of 203 patients for photosensitivity in a 7.3-year period. J Am Acad Dermatol. 33(4):597-602.
Furtado RA, de Oliveira PF, Senedese JM, Ozelin SD, Ribeirao de Souza LD, Leandro LF, de Oliveira WL, Mangabeira da Silva JJ, Oliveira LC, Rogez H, et al. 2018. Assessment of genotoxic activity of oleoresins and leaves extracts of six Copaifera species for prediction of potential human risks. J Ethnopharmacol. 221:119-125.
Garcia-Hidalgo E, von Goetz N, Siegrist M, Hungerbühler K. 2017. Use-patterns of personal care and household cleaning products in Switzerland. Food Chem Toxicol. 99:24-39.
Gilpin S, Hui X, Maibach H. 2010. In vitro human skin penetration of geraniol and citronellol. Dermatitis 21: 41-48.
GlaxoSmithKline Consumer Healthcare Inc. [modified 2017]. Abreva frequently asked questions. Mississauga (ON): GlaxoSmithKline Consumer Healthcare Inc. [accessed 2019 Sep 4].
Gomes-Carneiro MR, Felzenszwalb I, Paumgartten FJ. 1998. Mutagenicity testing of (±)-camphor, 1, 8-cineole, citral, citronellal,(−)-menthol and terpineol with the Salmonella/microsome assay. Mutat Res. 416(1-2):129-136.
Gomez-Berrada MP, Gautier F, Parent-Massin D, Ferret PJ. 2013. Retrospective exposure data for baby and children care products: An analysis of 48 clinical studies. Food Chem Toxicol. 57:185–194.
Gonçalves ES, Silva JR, Gomes CL, Nery MBL, Navarro DMAF, Santos GKN, Silva-Neto JC, Costa-Silva JH, Araùjo AV, Wanderley AG. 2014. Effects of the oral treatment with Copaifera multijuga oil on reproductive performance of male Wistar rats. Revista Brasileira de Farmacognosia. 24:355-62.
Guengerish PF. 2008. Cytochrome P450 and Chemical Toxicology. Chem Res Toxicol. 21:70-83.
Hall RL, Oser BL. 1965. Recent progress in the consideration of flavoring ingredients under the Food Additives Amendment. III. GRAS (generally recognized as safe) substances. Food Technol. 19:151-197.
[HC] Health Canada. 2013. Supporting documentation: summary tables of health effects information for methylenediphenyl diisocyanates and diamines. Ottawa (ON): Health Canada. Information in support of the screening assessment for the MDI/MDA Substance Grouping. Available from: substances@ec.gc.ca.
[HC] Health Canada. 2016. Science approach document: threshold of toxicological concern (TTC)-based approach for certain substances. Ottawa (ON): Government of Canada.
[HC] Health Canada. 2019. Canadian exposure factors used in human health risk assessment. Unpublished report. Ottawa (ON): Government of Canada.
[HC] Health Canada. [modified 2020 Jul 14]. Vaping product regulations. Ottawa (ON): Government of Canada. [accessed 2020 Dec 4].
Heck JD, Vollmuth TA, Cifone MA, Jagannath DR, Myhr B, Curren RD. 1989. An evaluation of food flavoring ingredients in a genetic toxicity screening battery. Toxicologist. 9: 257-272.
Husnu Can Baser K, Buchbauer G. 2010. Handbook of essential oils. science, technology, and applications. 1st ed. London (UK): CRC Press and Taylor & Francis Group
[IARC] International Agency for Research on Cancer. 2002. IARC monographs on the evaluation of carcinogenic risks to human. Some traditional herbal medicines, some mycotoxins, naphthalene and styrene [PDF]. Volume 82. Lyon (FR): IARC.
[IFRA] International Fragrance Association Volume of Use Survey 2017: Transparency List. 2017. Geneva (CH): International Fragrance Association. [accessed 2018 December].
Imaizumi K, Hanada K, Mawatari K, Sugano M. 1985. Effect of essential oils on the concentration of serum lipids and apolipoproteins in rats. Agri Biol Chem. 49:2795-2796.
[IOFI] International Organization of the Flavour Industry 1995. European Inquiry on Volume of Use. Submitted to WHO by the Flavor and Extract Manufacturers’ Association of the United States, Washington DC, USA.
Isman MB. 2000. Plant essential oils for pest and disease management. J Crop Protec. 19:603-608.
Jeena K, Liju VB, Kuttan R. 2011. A preliminary 13-week oral toxicity study of ginger oil in male and female Wistar rats. Int J Toxicol. 30:662-670.
Jeena K, Liju VB, Kuttan R. 2014. Antimutagenic potential and modulation of carcinogen-metabolizing enzymes by ginger essential oil. Phytother Res. 28:849-855.
Jirovetz L, Buchbauer G, Stoilova I, Stoyanova A, Krastanov A, Schmidt E. 2006. Chemical composition and antioxidant properties of clove leaf essential oil. J Agric Food Chem 23: 6303-7.
Kourounakis AP, Rekka EA, Kourounakis PN. 1997. Antioxidant activity of guaiazulene and protection against paracetamol hepatotoxicity in rats. J Pharm Pharmacol. 49:938-942.
Lima CS, de Medeiros BJL, Favacho HAS, dos Santo KC, de Oliveira BR, Taglialegna JC, da Costa EVM, de Campos KJ,Carvalho JCT. 2011. Pre-clinic validation of vaginal cream containing copaiba oil (reproductive toxicology study). Phytomedecine. 18(12):1013-23.
[LNHPD] Licensed Natural Health Products Database [database]. [modified 2018 Feb 06]. Ottawa (ON): Government of Canada. [accessed 2019 April].
Loretz LG, Api AM, Barraj LM, Burdick J, Dressler WE, Gettings SD, Han Hsu H, Pan YHL, Re TA, Renskers KJ et al. 2005. Exposure data for cosmetic products: lipstick, body lotion, and face cream. Food Chem Toxicol. 43:279-291.
Loretz L, Api AM, Barraj L, Burdick J, Davis DA, Dressler W, Gilberti E, Jarrett G, Mann S, Pan YHL, et al. 2006. Exposure data for personal care products: Hairspray, spray perfume, liquid foundation, shampoo, body wash, and solid antiperspirant. Food Chem Toxicol. 44:2008-2018.
Loretz LG, Api AM, Babcock L, Barraj LM, Burdick J, Cater KC, Jarrett G, Mann S, Pan YHL, Re TA, et al. 2008. Exposure data for cosmetic products: Facial cleaner, hair conditioner, and eye shadow. Food Chem Toxicol. 46(5):1516-1524.
Lorillard Tobacco Company. 1982. Mutagenicity evaluation of alpha-terpineol in the mouse lymphoma mutation assay. LBI Project no. 20989. Unpublished report submitted by EFFA to FLAVIS Secretariat.
Lorillard Tobacco Company. 1983. Mutagenicity evaluation of alpha-terpineol in the Ames Salmonella/microsome plate test. LBI Project no. 20988. Unpublished report submitted by EFFA to FLAVIS Secretariat.
Lucas CD, Putnam JM, Hallagan JB. 1999. 1995 Poundage and technical effects update survey. Washington (DC): Flavor and Extract Manufacturers’ Association of the United States.
Lucca LG, de Matos SP, Borille BT, Dias DDO, Teixeira HF, Veiga Jr VF, Limberger RP, Koester LS. 2015. Determination of β-caryophyllene skin permeation/retention from crude copaiba oil (Copaifera multijuga Hayne) and respective oil-based nanoemulsion using a novel HS-GC/MS method. J Pharma Biomed Anal. 104:144-148.
Madyastha KM, Srivatsan V. 1988. Biotransformations of α-terpineol in the rat: its effects on the liver microsomal cytochrome P-450 system. Bull Environ Contam Toxicol. 41:17-25.
[MSDS] Material Safety Data Sheet. 2015. Glade Wax Melts (Pumpkin Spice). Racine WI: S.C. Johnson & Son. [accessed 2018 Mar 1].
May RL, Levenson C, So JJ, Rock JA. 2012. Single-centre, open-label study of a proprietary topical 0.5% salicylic acid-based treatment regimen containing sandalwood oil in adolescents and adults with mild to moderate acne. J Drugs Dermatol. 11(2):1403-1408.
Molina-Jasso D, Alvarez-Gonzalez I, Madrigal-Bujaidar E. 2009. Clastogenicity of beta-caryophyllene in mouse. Biol Pharm Bull. 32(3):520-522.
Mukhopadhyay MJ, Mukherjee A. 2000. Clastogenic effect of ginger rhizome in mice. Phytother Res. 14(7):555-557.
Nakayama H. 1998. Fragrance hypersensitivity and its control. In: Frosch PJ, Johansen JD, White IR, editors. Fragrances: beneficial and adverse effects. Berlin (DE): Springer.
[NAS] National Academy of Sciences, 1989. 1987 Poundage and technical effects update of substances added to food. Washington (DC): Committee on Food Additives Survey Data, Food and Nutrition Board, Institute of Medicine, National Academy of Sciences.
[NHPID] Natural Health Products Ingredients Database [database]. [modified 2019 Sep 26]. Ottawa (ON): Government of Canada. [accessed 2019 October].
Nijssen LM, Ingen-Visscher CA van, Donders JJH [eds]. 1963-2018. VCF (Volatile Compounds in Food): database, Version 16.6.1, 2018. Triskelion B.V. Zeist, Netherlands.
[OECD] Organisation for Economic Co-operation and Development. 2009. Emission scenario document on coating industry (paints, lacquers and varnishes) [PDF]. Paris (FR): OECD, Environment Directorate. (Series on Emission Scenario Documents No. 22; Report No.:ENV/JM/MONO(2009)24, JT03267833). [accessed 2018 Mar 1].
OECD QSAR Toolbox [Read-across tool]. 2014. Version 3.3. Paris (FR): Organisation for Economic Co-operation and Development, Laboratory of Mathematical Chemistry.
OECD QSAR Toolbox [Read-across tool]. 2016. Paris (FR): Organisation for Economic Co-operation and Development, Laboratory of Mathematical Chemistry.
OECD QSAR Toolbox [Read-across tool]. 2019. Version 4.2. Paris (FR): Organisation for Economic Co-operation and Development, Laboratory of Mathematical Chemistry.
Onyenekwe PC, Hashimoto S. 1999. The composition of the essential oil of dried Nigerian ginger (Zingiber officinale Roscoe). Eur Food Res Technol. 209(6):407-10.
Opdyke DLJ. 1974. Fragrance raw materials monographs: Sandalwood oil, East Indian. Food Chem Toxicol. 12:989-990.
Ortiz C, Morales L, Saster M, Haskins WE, Matta J. 2016. Cytotoxicity and genotoxicity assessment of sandalwood essential oil in human breast cell lines MCF-7 and MCF-10A. Evid Based Complement Alternat Med. 3696232.
Perveen S. 2018. Introductory Chapter: Terpenes and Terpenoids. In: IntechOpen. [accessed 2019 Dec 18].
Petrov P. 2019. Guaiol. Terpenes and Testing Magazine. [accessed 2019 Mar 15].
Pino JA, Marbot R, Rosado A, Batista A. 2004. Chemical composition of the essential oil of Zingiber officinale Roscoe L. from Cuba. J Essent Oil Res. 16(3):186-88.
[PMRA] Pest Management Regulatory Agency. 2010. Re-evaluation Decision Naphthalene. Ottawa (ON): Government of Canada.
Rania VK, Kumar A, Aggarwal KK. 2005. Essential oil composition of ginger (Zingiber officinale Roscoe) rhizomes from different places in India. J Essent. 8(2):187-191.
[RIFM] Research Institute for fragrance Materials, Inc. 2017. RIFM fragrance ingredient safety assessment, β-Guaiene, CAS Registry Number 88-84-6. Food Chem Toxicol. 110(Supplement 1):S9-S15.
[RIVM] Rijksinstituut voor Volksgezondheid en Milieu [National Institute for Public Health and the Environment (NL)]. 2006. Cosmetics fact sheet: to assess the risks for the consumer: updated version for ConsExpo 4. Bilthoven (NL): RIVM. Report No.: 320104001/2006.
[RIVM] Rijksinstituut voor Volksgezondheid en Milieu [National Institute for Public Health and the Environment (NL)]. 2014. General fact sheet: general default parameters for estimating consumer exposure–updated version 2014. Bilthoven (NL): RIVM. Report No.: 090013003/2014.
Sachetti CG, de Carvalho RR, Paumgartten FJR, Lameira OA, Caldas ED. 2011. Developmental toxicity of copaiba tree (Copaifera reticulate Ducke, Fabaceae) oleoresin in rat. Food Chem Toxicol. 49:1080-1085.
Santiago KB, Conti BJ, Andrade BFMT, Da Silva JJM, Rogez HLG, Crevelin EJ, De Moraes LAB, Veneziani R, Ambrosio SR, Bastos JK, et al. 2015. Immunomodulatory action of Copaifera spp oleoresins on cytokine production by human monocytes. Biomed Pharmacother. 70:12-18.
[SCCS] Scientific Committee on Consumer Safety. 2010. Basic criteria for the in vitro assessment of dermal absorption of cosmetic ingredients adopted by the SCCS at its 7th plenary meeting of 22 June 2010 [PDF]. Brussels (BE): European Commission, Health & Consumers.
[SCCS] Scientific Committee on Consumer Safety. 2015. The SCCS’s Notes of Guidance for the Testing of Cosmetic Ingredients and their Safety Evaluation [PDF]. 9th Revision. European Commission. Report No. SCCS/1564/15, Revised version of 25 April 2016.
Schmitt D, Levy R, Carrol B. 2016. Toxicological evaluation of β-caryophyllene oil: subchronic toxicity in rats. Int J Toxicol. 35(5):558-567.
Sharma M, Levenson C, Bell RH, Anderson, SA, Hudson JB, Collins CC, Cox ME. 2014. Suppression of Lipopolysaccharide‐stimulated Cytokine/Chemokine Production in Skin Cells by Sandalwood oil and Purified α‐santalol and β‐santalol. Phytother Res. 28(6):925-932.
[SS] Specification Sheet. 2009. Clove Terpenes 920 Indesso. Barcelona (Spain): Ventós.
Statistics Canada. 2004. Canadian Community Health Survey (CCHS) – Cycle 2.2: Nutrition. Share File. Ottawa (ON): Government of Canada. Estimates generated using SAS 9.3 [GRID] through SAS EG 5.1.
Statistics Canada. 2015. Canadian Community Health Survey (CCHS) 2015 Share File. Ottawa (ON): Government of Canada. Estimates generated using SAS 9.3 [GRID] through SAS EG 5.1.
Statistics Canada. 2017. Custom tabulation of grooming products data from the Canadian Health Measures Survey Cycle 2 (2010-2011). Prepared for Existing Substances Risk Assessment Bureau, Health Canada by Statistics Canada. Unpublished. Ottawa (ON): Government of Canada.
Stoner GD, Shimkin MB, Kniazeff AJ, Weisburger JH, Weisburger EK, Gori GB. 1973. Test for carcinogenicity of food additives and chemotherapeutic agents by the pulmonary tumor response in strain A mice. Cancer Res. 33:3069-3085.
Swift KAD. 2005. Flavours and Fragrances. Cambridge (UK): Woodhead Publishing Limited.
[TDS] Technical Data Sheet. 2018. Clove Terpenes. Barcelone (ES): Lluch Essence.
Tisserand R, Young R. 2014. Essential Oil Safety. 2nd ed. London (UK): Churchill Livingstone.
Togar B, Turkez H, Hacimuftuoglu A, Tatar A, Geyikoglu F. 2014. Guaiazulene biochemical activity and cytotoxic and genotoxic effects on rat neuron and N2a neuroblastom cells. J Intercult Ethnopharmacol. 4: 29-33.
Ultra International B.V. 2018. Product: Clove terpenes. Spijkenisse (NL): Ultra International B.V. [accessed 2018 December].
[US EPA] United States Environmental Protection Agency. 2008. Naphthalene: Phase 4 amendment: Response to comments in reference to “naphthalene: HED Chapter for the registration eligibility document (RED)”. Washington (DC): US EPA, Office of Research and Development.
[US EPA] United States Environmental Protection Agency. 2011. Exposure Factors Handbook 2011 Edition (Final. Washington (DC): US EPA. EPA/600/R-09/052F.
[US EPA] United States Environmental Protection Agency. 2012a. EPI Suite- Estimation Program Interface Suite for Microsoft Windows [estimation model]. C2000-2012. Version 4.11. Washington (DC): US Environmental Protection Agency, Office of Pollution Prevention and Toxics; Syracuse (NY): Syracuse Research Corporation.
[US EPA] United States Environmental Protection Agency. 2019. The Chemistry Dashboard. LIST: Terpenes in vape. Research Triangle Park (NC): US EPA, Chemical Safety for Sustainability Research Program.
[US FDA] US Food and Drug Administration. 2018. Substances Added to Food (formerly EAFUS). Silver Spring (MD): US FDA.
Verghese J, Sunny TP, Balakrishnan V. 1990. (+)-α-Santalol and (-)-β-Santalol (Z) Concentration, a New Quality Determinant of East Indian Sandalwood Oil. Flavour Frag J. 5:223-226.
[WHO] Joint FAO/WHO Expert Committee on Food Additives. 2002. Evaluation of certain food additives (#913). Fifty-ninth report of the Joint FAO/WHO Expert Committee on Food Additives [PDF]. Geneva (CH): World Health Organization.
[WHO] Joint FAO/WHO Expert Committee on Food Additives. 2005. Evaluation of certain food additives and certain contaminants (series 54). Sixty-third meeting of the Joint FAO/WHO Expert Committee on Food Additives [PDF]. Geneva (CH): World Health Organization.
[WHO] Joint FAO/WHO Expert Committee on Food Additives. 2011. Safety evaluation of certain food additives and contaminants (series 64). Seventy-third meeting of the Joint FAO/WHO Expert Committee on Food Additives [PDF]. Geneva (CH): World Health Organization.
Wilson AS, Davis CD, Williams DP, Buckpitt AR, Pirmohamed M, Park BK. 1996. Characterisation of the toxic metabolite(s) of naphthalene. Toxicology. 114:233-242.
Won D, Yang W, So S, Aubin D. 2019. Investigation of Sources and Indoor Air Levels of CMP3 Chemicals. Phase 1 – Analysis of Indoor Air Samples from Two Field Studies. Final Report. Ottawa (ON): National Research Council. 53 pp. Report No: A1-014282.1.
Wu X, Bennett DH, Ritz B, Cassady DL, Lee K, Hertz-Picciotto I. 2010. Usage pattern of personal care products in California households. Food Chem Toxicol. 48:3109-3119.
Xin-Hua Z, Teixeira da Silva JA, Yong-Xia J, Jian Y, Guo-Hua M. 2012. Essential Oils Composition from Roots of Santalum album L. J Essent. 15(1):1-6.
Appendices
Appendix A. Read across within subgroup 3, sandalwood oil and guaiazulene
| Chemical name | Guaiol | Bulnesol | Elemol | Alpha-terpineol |
|---|---|---|---|---|
| Role | Target | Target | Target | Analogue |
| CAS # | 489-86-1 | 22451-73-6 | 639-99-6 | 98-55-5 |
| Chemical structure | ![C[C@H]1CC[C@H](CC2=C1CC[C@@H]2C)C(C)(C)O](/content/dam/eccc/images/pded/terpenes-group-2/20210203-TableA-1-1.jpg) |
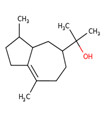 |
![CC(=C)[C@@H]1C[C@@H](CC[C@@]1(C)C=C)C(C)(C)O](/content/dam/eccc/images/pded/terpenes-group-2/20210203-TableA-1-3.jpg) |
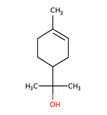 |
| Molecular weight (g/mol) | 222.37 | 222.37 | 222.37 | 154.25 |
| Water solubility (mg/L) | 3.61(M) | 7.07(M) | 1.99(M) | 710(E) |
| Vapour pressure (Pa) | 4.86 x 10-3 (M) | 6.80 x 10-3 (M) | 5.14 x 10-2 (M) | 5.64 at 24° C(E) |
| log Kow | 5.24(M) | 4.90(M) | 5.54(M) | 2.98(E) |
| Toxicokinetics and metabolism | ND | ND | Urinary metabolites: (-)-15- hydroxy-elemol. Metabolism via glucuronic acid/sulphate conjugation. Formation of epoxide intermediates is not anticipated.b | Allylic oxidation of the methyl group of α-terpineol, further oxidized to a carboxylic acid group. In a minor pathway, epoxidation and hydrolyzation to yield the triol metabolite 1,2,8-trihydroxy-p-menthane.c |
| Repeat dose toxicity (oral)a | ND | ND | ND | NOAEL=250 mg/kg bw/day (5-wk rat gavage study; testicular and epididymal toxicity leading to infertility at 750 mg/kg bw/day in males). |
| Repeat dose toxicity (inhalation) | ND | ND | ND | NOAEC>2.23 mg/L air (highest dose, snout-only 13-wk study in rats). |
| Reproductive and/or developmental toxicity (oral gavage) | ND | ND | ND | Male parental repro NOAEL=250 mg/kg bw/day; evidence of testicular and epididymal toxicity leading to infertility at highest dose tested of 750 mg/kg bw/day. This prevented the assessment of effects on female reproduction at 750 mg/kg bw/day so female repro NOAEL>250 mg/kg bw/day. Devo NOAEL>250 mg/kg bw/day in rats exposed daily throughout gestation and until day 6 of lactation; Male and female offspring were unaffected at 250 mg/kg bw/day |
| Developmental toxicity (oral gavage) | ND | ND | ND | NOAEL>600 mg/kg bw/day in rats exposed during GD 6-19 (14 days). |
| Genetic toxicity | ND | ND | Mutagenicity/ genotoxicity negative in Ames test in TA98, TA100, TA1535, TA1537 and E. coli strains.d | In vitro mutagenicity negative in 3 Ames test and 2 mouse lymphoma assays.e, f |
Abbreviations: ND, no data. Kow, octanol-water partition coefficient
E: experimental
M: modelled
A reference or references are cited if the information was not previously mentioned in the text of the report.
a The NOAEL observed for terpineol in this study is used as the POD in the risk characterization for read across of guaiol, bulnesol and elemol
b EFSA 2015
c Madyastha and Srivatsan 1988
d Api et al. 2016
e Florin et al. 1980, Heck et al. 1989, Lorillard Tobacco Company 1982, 1983
f Gomez-Carneiro et al. 1998
| Chemical name | Alpha-Santalol | Beta-Santalol | Bisabolol |
|---|---|---|---|
| Role | Target | Target | Analogue |
| CAS # | 115-71-9 | 77-42-9 | 515-69-5 |
| Chemical structure | ![C\C(=C\CC[C@]1(C)C2C[C@@H]3[C@H](C2)C13C)\CO (C)(C)O](/content/dam/eccc/images/pded/terpenes-group-2/20210203-TableA-2-1.jpg) |
![C\C(=C\CC[C@]1(C)[C@H]2CC[C@H](C2)C1=C)\CO)\CO (C)(C)O](/content/dam/eccc/images/pded/terpenes-group-2/20210203-TableA-2-2.jpg) |
(O)[C@@H]1CCC(=CC1)C)C)\CO)\CO (C)(C)O](/content/dam/eccc/images/pded/terpenes-group-2/20210203-TableA-2-1.jpg) |
| Molecular weight (g/mol) | 220.35 | 220.35 | 222.37 |
| Water solubility (mg/L) | 6.41(M) | 4.19(M) | 1.69(E) |
| Vapour pressure (Pa) | 4.51 x 10-3 (M) | 9.83 x 10-3 (M) | 0.018(M) |
| log Kow | 4.96(M) | 5.18(M) | 5.63(E) |
| Toxicokinetics and metabolism | ND | ND | ND |
| Repeat dose toxicity (oral) | ND | ND | ND |
| Repeat dose toxicity (inhalation) | ND | ND | ND |
| Repeat dose toxicity (dermal)* | ND | ND |
28 days semi-occluded at 0, 50, 200 or 1000 mg/kg bw/day on male and female rats. 50, 200 mg/kg body weight/day: no effects 1000 mg/kg body weight/day: decrease body weight gain, decrease feed efficiency, transient moderate skin erythema and diffuse scale formation in some females, decrease of absolute liver weight in females and increase of relative testes weight in males (NOAEL: 200 mg/kg body weight/day)a,b |
| Developmental toxicity (oral gavage)a | ND | ND | Pregnant rats received 0.250, 0.500, 1.0, or 3.0 ml/kg body weight/day bisabolol (equivalent to 0, 245, 490, 980 or 2940 mg/kg bw/day) on days 6 to15. NOAEL determined at 980 mg/kg bw/day based on slight sedation, ataxia, reduced feed intake, and reduction of body weight gain in females and a significant reduction in fetal number and increase in resorption rate at 2550 mg/kg bw/day.a,b Pregnant rabbits received 0.250, 0.500, 1.0, or 3.0 ml/kg body weight/day bisabolol (equivalent to 0, 294, 980 or 2940 mg/kg bw/day) on days 6 to18. NOAEL determined at 980 mg/kg bw/day based on slight sedation, ataxia, reduced feed intake, and reduction of body weight gain in females and a significant reduction in fetal number and increase in resorption rate at 2550 mg/kg bw/day.b |
| Genetic toxicity | Sandalwood oil was negative in spore Rec assay using H17 Rec+ and M45 Rec- in the presence of absence of metabolic activation (S9).c Sandalwood was not cytotoxic or genotoxic in breast epithelial cells.d | Sandalwood oil was negative in spore Rec assay using H17 Rec+ and M45 Rec- in the presence of absence of metabolic activation (S9).c Sandalwood was not cytotoxic or genotoxic in breast epithelial cells.d | Mutagenicity negative in Ames test in TA98, TA100 and TA1535, and Salmonella typhimurium and genotoxicity negative for chromosome aberration assay using Chinese hamster V79 cells.b |
Abbreviations: ND, no data. Kow, octanol-water partition coefficient
E: experimental
M: modelled
a The NOAEL observed for bisabolol in this study is used as the POD in the risk characterization for read across of sandalwood oil
b Bhatia et al. 2008b
c Burdock and Carabin 2008
d Ortiz et al. 2016
| Chemical name | Guaiazulene | Naphthalene |
|---|---|---|
| Role | Target | Analogue |
| CAS # | 489-84-9 | 91-20-3 |
| Chemical structure | 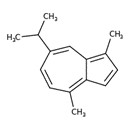 |
 |
| Molecular weight (g/mol) | 198.3 | 128.2 |
| Water solubility (mg/L) | 1.12 x 10-1 (M) | 31(E) |
| Vapour pressure (Pa) | 1.41(M) | 11.3(E) |
| Log Kow | 5.93(M) | 3.3(E) |
| Toxicokinetics and metabolism | ND | Naphthalene bioactivation varies considerably among species, gender and among different anatomical regions of the respiratory tract. In humans, it is readily absorbed and is metabolized by several cytochrome P450’s. Naphthalene and its metabolites can cross the placental barrier and consequently may affect foetal tissues. Naphthalene may undergo oxidative metabolism and is excreted in the form of naphtholic conjugates and thioether conjugates, which are derived from the initial product of oxidation in rodent. In human, the process is unknown.b |
| Repeat dose toxicity (oral) | ND | NOAEL of 100 mg/kg bw/day based on significant decreases in body weights/body weight gains at 200 mg/kg bw/day in a 13-week study (LOAEL).c |
| Repeat dose toxicity (inhalation) | ND | In a 13-week nose-only study in rats, a LOAEC of 10 mg/m3 was noticed based on increased incidence and severity of nasal lesions (degeneration, atrophy and hyperplasia of basal cells of the olfactory epithelium; rosette formation of the olfactory epithelium; loss of bowman’s glands; hypertrophy of respiratory epithelium).c NOAEC of 5.2 mg/m3 was identified in a similar inhalation study used to derive a NOAEC for nasal lesions following nose-only exposures.c |
| Repeat dose toxicity (dermal)a | ND | NOAEL of 300 mg/kg bw/day based on a 90-day dermal toxicity study in the rat where effects were noted only at the highest dose tested of 1000 mg/kg bw/day (US EPA 2008). These effects included excoriated skin and papules in both sexes and atrophy of seminiferous tubules in the males. Other effects observed included non-neoplastic lesions in the cervical lymph node (hyperplasia), liver (hemosiderosis), thyroid (thyroglossal duct cysts), kidneys (pyelonephritis), urinary bladder (hyperplasia) and skin (acanthosis, hyperkeratosis) in females.c |
| Genetic toxicity | ND | Naphthalene was negative in in vitro assays and in vivo assays and then not considerate as genotoxic.c |
Abbreviations: ND, no data. Kow, octanol-water partition coefficient
E: experimental
M: modelled
a The NOAEL observed for in naphthalene this study is used as the POD in the risk characterization for read across of guaiazulene
b Wilson et al. 1996
c US EPA 2008
Appendix B. Estimated dietary exposure from natural occurrence of monocyclic and bicyclic sesquiterpenes
| Subpopulation | Substance | Mean dietary exposure (µg/kg bw/day) | 90th percentile dietary exposure (µg/kg bw/day) |
|---|---|---|---|
| 0 to 6 months | Guaiene | 0 | 0 |
| 6 to 12 months | Guaiene | 0 | 0 |
| 1 to 3 years | Guaiene | 90 | 287 |
| 4 to 8 years | Guaiene | 152 | 361 |
| 9 to 13 years | Guaiene | 108 | 277 |
| 14 to 18 years | Guaiene | 73 | 165 |
| 19+ years | Guaiene | 81 | 137 |
| 0 to 6 months | beta-Caryophyllene | 0 | 0 |
| 6 to 12 months | beta-Caryophyllene | 165 | 519 |
| 1 to 3 years | beta-Caryophyllene | 1037 | 3242 |
| 4 to 8 years | beta-Caryophyllene | 1522 | 3733 |
| 9 to 13 years | beta-Caryophyllene | 1028 | 2691 |
| 14 to 18 years | beta-Caryophyllene | 694 | 1638 |
| 19+ years | beta-Caryophyllene | 735 | 1340 |
| 0 to 6 months | Valencene | 0 | 0 |
| 6 to 12 months | Valencene | 57 | 190 |
| 1 to 3 years | Valencene | 99 | 298 |
| 4 to 8 years | Valencene | 95 | 266 |
| 9 to 13 years | Valencene | 45 | 139 |
| 14 to 18 years | Valencene | 30 | 98 |
| 19+ years | Valencene | 14 | 54 |
| 0 to 6 months | Bulnesol | 0 | 0 |
| 6 to 12 months | Bulnesol | 0 | 0 |
| 1 to 3 years | Bulnesol | 101 | 322 |
| 4 to 8 years | Bulnesol | 171 | 406 |
| 9 to 13 years | Bulnesol | 122 | 311 |
| 14 to 18 years | Bulnesol | 82 | 185 |
| 19+ years | Bulnesol | 91 | 155 |
| 0 to 6 months | Elemol | 0 | 0 |
| 6 to 12 months | Elemol | 6 | 27 |
| 1 to 3 years | Elemol | 23 | 61 |
| 4 to 8 years | Elemol | 27 | 65 |
| 9 to 13 years | Elemol | 20 | 49 |
| 14 to 18 years | Elemol | 15 | 33 |
| 19+ years | Elemol | 14 | 26 |
Appendix C. Exposure parameters used to estimate exposure to subgroup 2 substances
| Age group | Air concentration (µg/m3) (24 hrs)a | Inhalation rate (m3/day)b | Body weight (kg)b | Inhalation exposure (mg/kg bw/day)c |
|---|---|---|---|---|
| 0-5 months | 1.5 | 3.7 | 6.3 | 8.81 x 10-4 |
| 6-11 months | 1.5 | 5.4 | 9.1 | 8.9 x 10-4 |
| 1 year | 1.5 | 8 | 11 | 1.09 x 10-3 |
| 2-3 years | 1.5 | 9.2 | 15 | 9.2 x 10-4 |
| 4-8 years | 1.5 | 11.1 | 23 | 7.24 x 10-4 |
| 9-13 years | 1.5 | 13.9 | 42 | 4.96 x 10-4 |
| 14-18 years | 1.5 | 15.9 | 62 | 3.85 x 10-4 |
|
Adults (19+ years) |
1.5 | 15.1 | 74 | 3.06 x 10-4 |
a Maximum value detected in indoor air monitoring in Nunavik homes (Won 2019).
b As cited in Heath Canada 2019.
c Calculated using the following formula: Inhalation exposure (mg/kg bw/day) = (Air concentration (1.5 µg/m3) * Inhalation rate (m3/day) * Conversion factor 1 mg/1000 µg))/ Body weight (kg)
| Age group | Mean product amount (g/app)a | Air concentration (mg/m3) (24 hrs)b | Dermal exposure (mg/kg bw/day)c | Inhalation exposure (mg/kg bw/day)d | Combined exposure (mg/kg bw/day)e |
|---|---|---|---|---|---|
| 0-5 months | 2 | 3.90 x 10-2 | 1.90 | 2.29 x 10-2 | 1.93 |
| 6-11 months | 2.5 | 4.90 x 10-2 | 1.65 | 2.91 x 10-2 | 1.68 |
| 1 year | 3.1 | 6.20 x 10-2 | 1.69 | 4.51 x 10-2 | 1.74 |
| 2-3 years | 4.1 | 7.90 x 10-2 | 1.64 | 4.85 x 10-2 | 1.69 |
| 4-8 years | 5 | 9.80 x 10-2 | 1.30 | 4.73 x 10-2 | 1.35 |
| 9-13 years | 7.7 | 1.50 x 10-1 | 1.10 | 4.96 x 10-2 | 1.15 |
| 14-18 years | 10 | 2.00 x 10-1 | 9.68 x 10-1 | 5.13 x 10-2 | 1.02 |
|
Adults (19+ years) |
10 | 2.10 x 10-1 | 8.11 x 10-1 | 4.29 x 10-2 | 8.54 x 10-1 |
a Based on Ficheux et al. 2016. Assumed frequency was 1 for all age groups. For inhalation modelling, product amount was adjusted for exposed surface area, see below for details.
b Air concentrations were modelled using ConsExpo exposure to vapour-evaporation-constant release model, and using the following parameters: exposure duration of 24 hours, emission duration of 24 hours, room volume of 58 m3, ventilation rate of 0.5/hr, surface area equal to exposure skin (assumed equal to arms, ¾ legs, hands, and ½ feet) (1325 cm2 for 0- to 5-month-olds, 1703 cm2 for 6- to 11-month-olds, 2070 cm2 for 1-year-olds, 2685 cm2 for 2- to 3-year-olds, 3813 cm2 for 4- to 8-year-olds, 5953 cm2 for 9- to 13-year-olds, 7655 cm2 for 14- to 18-year-olds, and 8543 cm2 for adults (19+ yrs) (HC 2019)) product amount adjusted for exposed surface area (0.93 g for 0- to 5-month-olds, 1.16 g for 6- to 11-month-olds, 1.45 g for 1-year-olds, 1.85 g for 2- to 3-year-olds, 2.30 for 4- to 8-year-olds, 3.61 g for 9- to 13-year-olds, 4.65 g for 14- to 18-year-olds, and 4.87 g for adults), molecular weight matrix of 1000 g/mol, and application temperature of 32°C.
c Dermal exposure (mg/kg bw/day) calculated using the following formula: [mean product (g/application) * mean daily frequency (applications/day) * product concentration (3%) * dermal absorption (20%) * conversion factor (1000 mg/g)] ÷ body weight (6.3 kg for 0- to 5-month-olds, 9.1 kg for 6- to 11-month-olds, 11 kg for 1-year-olds, 15 kg for 2- to 3-year-olds, 23 kg for 4- to 8-year-olds, 42 kg for 9- to 13-year-olds, 62 kg for 14- to 18-year-olds, and 74 kg for adults (19+ yrs) (HC 2019)).
d Inhalation exposure (mg/kg bw/day) calculated using the following formula: [Air concentration (mg/m3) (24 hrs time-weighted average) * Inhalation rate (m3/day) (3.7 m3/day for 0- to 5-month-olds, 5.4 m3/day for 6- to 11-month-olds, 8 m3/day for 1-year-olds, 9.2 m3/day for 2- to 3-year-olds, 11.1 m3/day for 4- to 8-year-olds, 13.9 m3/day for 9- to 13-year-olds, 15.9 m3/day for 14- to 18-year-olds, and 15.1 m3/day for adults (19+ yrs) (HC 2019))] ÷ body weight (6.3 kg for 0- to 5-month-olds, 9.1 kg for 6- to 11-month-olds, 11 kg for 1-year-olds, 15 kg for 2- to 3-year-olds, 23 kg for 4- to 8-year-olds, 42 kg for 9- to 13-year-olds, 62 kg for 14- to 18-year-olds, and 74 kg for adults (19+ yrs) (HC)).
e Combined exposure (mg/kg bw/day) = Dermal exposure (mg/kg bw/day) + Inhalation exposure (mg/kg bw/day)
| Age group | Mean product amount (g/day)a | Air concentration (mg/m3) (24 hrs)b | Dermal exposure (mg/kg bw/day)c | Inhalation exposure (mg/kg bw/day)d | Combined exposure (mg/kg bw/day)e |
|---|---|---|---|---|---|
| 0-5 months | 1.8 | 5.00 x 10-2 | 1.71 | 2.94 x 10-2 | 1.74 |
| 6-11 months | 1.8 | 5.30 x 10-2 | 1.19 | 3.15 x 10-2 | 1.22 |
| 1 years | 1.8 | 5.50 x 10-2 | 9.82 x 10-1 | 4.00 x 10-2 | 1.02 |
| 2-3 years | 1.8 | 5.80 x 10-2 | 7.20 x 10-1 | 3.56 x 10-2 | 7.56 x 10-1 |
| 4-8 years | 1.9 | 6.30 x 10-2 | 4.96 x 10-1 | 3.04 x 10-2 | 5.26 x 10-1 |
| 9-13 years | 2.3 | 7.50 x 10-2 | 3.29 x 10-1 | 2.48 x 10-2 | 3.53 x 10-1 |
| 14-18 years | 2.9 | 9.40 x 10-2 | 2.81 x 10-1 | 2.41 x 10-2 | 3.05 x 10-1 |
| Adults (19+ years) | 3.2 | 1.00 x 10-1 | 2.59 x 10-1 | 2.04 x 10-2 | 2.80 x 10-1 |
a Based on Ficheux et al. 2016. Assumed frequency of 1/day.
b Air concentrations were modelled using ConsExpo exposure to vapour-evaporation-constant release model, assuming 80% of the applied dose was available for evaporation (amount remaining on the skin surface following dermal absorption of the substance) (80% * product amount * 3%) and using the following parameters: exposure duration of 1 hr, emission duration of 1 hr, room volume of 20 m3, ventilation rate of 0.6/hr, release area equal to exposure surface area (total surface area minus head for 0- to 11-year-olds and total surface area minus one-half head and trunk for adults and adolescents) (3180 cm2 for 0-to 5-month-olds, 4090 cm2 for 6- to 11-month-olds, 4865 cm2 for 1-year-olds, 6225 cm2 for 2- to 3-year-olds, 8595 cm2 for 4- to 8-year-olds, 10395 cm2 for 9- to 13-year-olds, 13385 cm2 for 14- to 18-year-olds, and 14670 cm2 for adults (19+ yrs) (HC 2019)), molecular weight of 1000 g/mol, and application temperature of 32°C.
c Dermal exposure calculated using the following formula: (Mean product amount (g/application) * mean daily frequency * product concentration (3%) * dermal absorption (20%) * conversion factor (1000 mg/g)) ÷ Body weight (kg) (6.3 kg for 0- to 5-month-olds, 9.1 kg for 6- to 11-month-olds, 11 kg for 1-year-olds, 15 kg for 2- to 3-year-olds, 23 kg for 4- to 8-year-olds, 42 kg for 9- to 13-year-olds, 62 kg for 14- to 18-year-olds, and 74 kg for adults (19+ yrs) (HC 2019)).
d Inhalation exposure (mg/kg bw/day) calculated using the following formula: [Air concentration (mg/m3) (24 hrs time-weighted average) * Inhalation rate (m3/day) (3.7 m3/day for 0- to 5-month-olds, 5.4 m3/day for 6- to 11-month-olds, 8 m3/day for 1-year-olds, 9.2 m3/day for 2- to 3-year-olds, 11.1 m3/day for 4- to 8-year-olds, 13.9 m3/day for 9- to 13-year-olds, 15.9 m3/day for 14- to 18-year-olds, and 15.1 m3/day for adults (19+ yrs) (HC 2019))] ÷ body weight (6.3 kg for 0- to 5-month-olds, 9.1 kg for 6- to 11-month-olds, 11 kg for 1-year-olds, 15 kg for 2- to 3-year-olds, 23 kg for 4- to 8-year-olds, 42 kg for 9- to 13-year-olds, 62 kg for 14- to 18-year-olds, and 74 kg for adults (19+ yrs) (HC 2019)).
e Combined exposure (mg/kg bw/day) = dermal exposure (mg/kg bw/day) + inhalation exposure (mg/kg bw/day)
| Age group | Mean product amount (g/app)a | Mean daily frequencyb | Dermal exposure (mg/kg bw/day)c | Inhalation exposure (mg/kg bw/day)d | Systemic exposure (mg/kg bw/day)e |
|---|---|---|---|---|---|
| 2-3 years | 0.33 | 1.0 | 3.74 | 6.75 x 10-2 | 3.81 |
| 4-8 years | 0.33 | 1.0 | 2.44 | 5.31 x 10-2 | 2.49 |
| 9-13 years | 0.33 | 1.4 | 1.87 | 3.64 x 10-2 | 1.91 |
| 14-18 years | 0.33 | 1.4 | 1.27 | 2.82 x 10-2 | 1.29 |
|
Adults (19+ years) |
0.33 | 1.7 | 1.29 | 2.24 x 10-2 | 1.31 |
a Based on Loretz et al. 2006
b Based on Loretz et al. 2006; Statistics Canada 2017; Minimum daily frequency was set to 1.
c Dermal exposure (mg/kg bw/day) was calculated using the following formula: (Mean product amount (g/application) * mean daily frequency * product concentration (100%) * retention factor (0.85) * conversion factor (1000 mg/g) * dermal absorption (20%)) ÷ Body weight (kg) [15 kg (2- to 3-year-olds; 23 kg (4- to 8-year-olds; 42 kg (9- to 13-year-olds; 62 kg (14- to 18-year-olds; 74 kg (adults (19+ yrs) (HC 2019))]
d Inhalation exposure (mg/kg bw/day) was calculated using the following formula: (Air concentration (24 hrs average) (mg/m3) * inhalation rate (m3/day) [9.2 m3/day (2- to 3-year-olds); 11.1 m3/day (4- to 8-year-olds); 13.9 m3/day (9- to 13-year-olds); 15.9 m3/day (14- to 18-year-olds); 15.1 m3/day (adults (19+ yrs)] ÷ Body weight (kg) [15 kg (2- to 3-year-olds); 23 kg (4- to 8-year-olds); 42 kg (9- to 13-year-olds); 62 kg (14- to 18-year-olds); 74 kg (adults (19+ yrs) (HC 2019))]. Air concentrations were modelled using the instantaneous release model in ConsExpo assuming an exposure duration of 5 minutes, room volume of 10 m3 and ventilation rate of 0/hr.
e Systemic exposure (mg/kg bw/day) was calculated using the following formula: Dermal exposure (mg/kg bw/day) + Inhalation exposure (mg/kg bw/day)
| Age group | Mean product amount (g/app)a | Air concentration (mg/m3) (24 hrs)b | Dermal exposure (mg/kg bw/day)c | Inhalation exposure (mg/kg bw/day)d | Combined exposure (mg/kg bw/day)e |
|---|---|---|---|---|---|
| 0-5 months | 2 | 6.00 x 10-5 | 6.35 x 10-2 | 3.52 x 10-5 | 6.35 x 10-2 |
| 6-11 months | 2.5 | 8.60 x 10-5 | 5.49 x 10-2 | 5.10 x 10-5 | 5.50 x 10-2 |
| 1 years | 3.1 | 1.10 x 10-4 | 5.64 x 10-2 | 8.00 x 10-5 | 5.64 x 10-2 |
| 2-3 years | 4.1 | 1.00 x 10-4 | 5.47 x 10-2 | 6.13 x 10-5 | 5.47 x 10-2 |
| 4-8 years | 5 | 1.60 x 10-3 | 4.35 x 10-2 | 7.72 x 10-4 | 4.43 x 10-2 |
| 9-13 years | 7.7 | 3.90 x 10-4 | 3.67 x 10-2 | 1.29 x 10-4 | 3.68 x 10-2 |
| 14-18 years | 10 | 7.40 x 10-5 | 3.23 x 10-2 | 1.90 x 10-5 | 3.23 x 10-2 |
|
Adults (19+ years) |
10 | 7.60 x 10-5 | 2.70 x 10-2 | 1.55 x 10-5 | 2.70 x 10-2 |
a Based on Ficheux et al. 2016. Assumed frequency was 1 for all age groups. For inhalation modelling, product amount was adjusted for exposed surface area, see below for details.
b Air concentrations were modelled using ConsExpo exposure to vapour-evaporation-constant release model, and using the following parameters: exposure duration of 24 hours, emission duration of 24 hours, room volume of 58 m3, ventilation rate of 0.5/hr, surface area equal to exposure skin (assumed equal to arms, ¾ legs, hands, and ½ feet) (1325 cm2 for 0- to 5-month-olds, 1703 cm2 for 6- to 11-month-olds, 2070 cm2 for 1-year-olds, 2685 cm2 for 2- to 3-year-olds, 3813 cm2 for 4- to 8-year-olds, 5953 cm2 for 9- to 13-year-olds, 7655 cm2 for 14- to 18-year-olds, and 8543 cm2 for adults (19+ yrs) (HC 2019)) product amount adjusted for exposed surface area (0.93 g for 0- to 5-month-olds, 1.16 g for 6- to 11-month-olds, 1.45 g for 1-year-olds, 1.85 g for 2- to 3-year-olds, 2.30 for 4- to 8-year-olds, 3.61 g for 9- to 13-year-olds, 4.65 g for 14- to 18-year-olds, and 4.87 g for adults), molecular weight matrix of 1000 g/mol, and application temperature of 32°C.
c Dermal exposure (mg/kg bw/day) calculated using the following formula: [mean product (g/application) * mean daily frequency (applications/day) * product concentration (0.1%) * dermal absorption (20%) * conversion factor (1000 mg/g)] ÷ body weight (6.3 kg for 0- to 5-month-olds, 9.1 kg for 6- to 11-month-olds, 11 kg for 1-year-olds, 15 kg for 2- to 3-year-olds, 23 kg for 4- to 8-year-olds, 42 kg for 9- to 13-year-olds, 62 kg for 14- to 18-year-olds, and 74 kg for adults (19+ yrs) (HC 2019)).
d Inhalation exposure (mg/kg bw/day) calculated using the following formula: [Air concentration (mg/m3) (24 hrs time-weighted average) * Inhalation rate (m3/day) (3.7 m3/day for 0- to 5-month-olds, 5.4 m3/day for 6- to 11-month-olds, 8 m3/day for 1-year-olds, 9.2 m3/day for 2- to 3-year-olds, 11.1 m3/day for 4- to 8-year-olds, 13.9 m3/day for 9- to 13-year-olds, 15.9 m3/day for 14- to 18-year-olds, and 15.1 m3/day for adults (19+ yrs) (HC 2019))] ÷ body weight (6.3 kg for 0-to 5-month-olds, 9.1 kg for 6- to 11-month-olds, 11 kg for 1-year-olds, 15 kg for 2- to 3-year-olds, 23 kg for 4- to 8-year-olds, 42 kg for 9- to 13-year-olds, 62 kg for 14- to 18-year-olds, and 74 kg for adults (19+ yrs) (HC 2019)).
e Combined exposure (mg/kg bw/day) = Dermal exposure (mg/kg bw/day) + Inhalation exposure (mg/kg bw/day)
| Age group | Mean product amount ingested (g/application)a | Mean daily frequencyb | Body weight (kg)c | Systemic exposure (mg/kg bw/day)d |
|---|---|---|---|---|
| 4-8 years | 1.7 | 1 | 23 | 1.30 |
| 9-13 years | 1.7 | 1 | 42 | 7.14 x 10-1 |
| 14-18 years | 1.0 | 1 | 62 | 8.23 x 10-1 |
|
Adults (19+ years) |
1.0 | 1 | 74 | 6.89 x 10-1 |
a Based on Ficheux et al. 2016; SCCS 2015.
b Based on Ficheux et al. 2015. Minimum daily frequency was set to 1.
c As cited in Canadian exposure factors used in human health risk assessment (HC 2019).
d Systemic exposure calculated using the following formula: (Mean product amount ingested (g/application) * mean daily frequency * product concentration (3%) * conversion factor (1000 mg/g)) ÷ Body weight (kg)
| Exposure scenarioa | Age group | Concentration | Dermal exposure (mg/kg bw/day) | Inhalation exposure (mg/kg bw/day) | Combined exposure (mg/kg bw/day) |
|---|---|---|---|---|---|
| Mixing, loading, and hanging hand-washed laundry | Adults | 5% | 1.54 x 10-1 | 3.88 x 10-4 | 1.54 x 10-1 |
| Air freshener | Toddlers – Adults | 10% | N/A | 1.23 x 10-1 –4.08 x 10-2 | 1.23 x 10-1– 4.08 x 10-2 |
| NMI in a cold sore cream | Adults – 2- to 3-year-olds | 20% | 2.70 x 10-1 – 1.33 | N/A | 2.70 x 10-1 – 1.33 |
Abbreviations: N/A, not applicable
a Details on the method and parameters used to estimate dermal and inhalation exposure to beta-caryophyllene from products that are available to consumers are provided in Table A-4.
| Exposure scenario | Assumptionsa |
|---|---|
| Mixing and loading a liquid for hand washing and hanging hand washed laundry (adults) |
Concentration of beta-caryophyllene: 5% (ACI 2018) Mixing and loading (dermal): Mixing and loading (inhalation-exposure to vapour-evaporation-constant release model): Hand-washing (dermal): Hand-washing (inhalation-exposure to vapour evaporation-constant release): Hanging hand-washed laundry (dermal): Hanging hand-washed laundry (inhalation-exposure to vapour-evaporation-increasing release area): Combined exposure: Dermal (mixing/loading + hand washing + hanging machine washed laundry) + Inhalation (mixing/loading + hand-washing + hanging machine washed laundry) |
| Air freshener (wax melt) (inhalation) (all subpopulations) |
Concentration of beta-caryophyllene: 5% to 10% (MSDS 2015) Inhalation – exposure to vapour, constant rate scenario Inhalation exposure (mg/kg bw/day) = (air concentration (mg/m3 – 24 hrs) * daily inhalation rate (m3/day)) ÷ body weight (kg) |
| Non-medicinal ingredient in a cold sore cream |
Concentration of beta-caryophyllene: 20% Amount: 100 mg/day based on a 2000 mg product size that is intended for 3 outbreaks that last 10 days each (2000 mg/product ÷ 3 outbreaks/product ÷ 10 days/outbreak = 100 mg/day) (GlaxoSmithKline Consumer Healthcare Inc. 2017). Oral exposure (mg/kg bw/day) = (100 mg/day * product concentration (20%)) ÷ Body weight (kg) |
a Exposure to products was estimated using ConsExpo Web (2016). Exposure estimates were calculated based on default body weights and inhalation rates of 74 kg/15.1 m3/day for adults (19 years and older), 62 kg/15.9 m3/day for 14- to 18-year-olds, 42 kg/13.9 m3/day for 9- to 13-year-olds, 23 kg/11.1 m3/day for 4- to 8-year-olds, 15 kg/9.2 m3/day for 2- to 3-year-olds, 11 kg/8.0 m3/day for 1-year-olds, 9.1 kg/5.4 m3/day for infants aged 6 to 11 months, and 6.3 kg/3.7 m3/day for infants aged 0 to 5 months, respectively (HC 2019).
Appendix D. Exposure parameters used to estimate exposure to subgroup 3 substances
| Age group | Mean product amount (g/application)a | Body weight (kg)b | Dermal exposure (mg/kg bw/day)c |
|---|---|---|---|
| 0-5 months | 2 | 6.3 | 1.59 x 10-2 |
| 6-11 months | 2.5 | 9.1 | 1.37 x 10-2 |
| 1 year | 3.1 | 11 | 1.41 x 10-2 |
| 2-3 years | 4.1 | 15 | 1.37 x 10-2 |
| 4-8 years | 5 | 23 | 1.09 x 10-2 |
| 9-13 years | 7.7 | 42 | 9.17 x 10-3 |
| 14-18 years | 10 | 62 | 8.06 x 10-3 |
|
Adults (19+ years) |
10 | 74 | 6.76 x 10-3 |
a Based on Ficheux et al. 2016. Assumed frequency was 1 for all age groups.
b As cited in Canadian exposure factors used in human health risk assessment (HC 2019).
c Dermal exposure calculated using the following formula: (Mean product amount (g/application) * mean daily frequency * product concentration (0.005%) * dermal absorption (100%) * conversion factor (1000 mg/g)) ÷ Body weight (kg)
Appendix E. Exposure parameters used to estimate exposure to sandalwood oil
| Age group | Mean product amount (g/application)a | Body weight (kg)b | Dermal exposure (mg/kg bw/day)c |
|---|---|---|---|
| 0-5 months | 2 | 6.3 | 95.2 |
| 6-11 months | 2.5 | 9.1 | 82.4 |
| 1 year | 3.1 | 11 | 84.5 |
| 2-3 years | 4.1 | 15 | 82.0 |
| 4-8 years | 5 | 23 | 65.2 |
| 9-13 years | 7.7 | 42 | 55.0 |
| 14-18 years | 10 | 62 | 48.4 |
| Adults (19+ years) | 10 | 74 | 40.5 |
a Based on Ficheux et al. 2016. Assumed frequency was 1 for all age groups.
b As cited in Canadian exposure factors used in human health risk assessment (HC 2019).
c Dermal exposure calculated using the following formula: (Mean product amount (g/application) * mean daily frequency * product concentration (30%) * dermal absorption (100%) * conversion factor (1000 mg/g)) ÷ Body weight (kg)
| Age group | Mean product amount (g/application)a | Body weight (kg)b | Dermal exposure (mg/kg bw/day)c |
|---|---|---|---|
| 0-5 months | 1.8 | 6.3 | 8.57 |
| 6-11 months | 1.8 | 9.1 | 5.93 |
| 1 year | 1.8 | 11 | 4.91 |
| 2-3 years | 1.8 | 15 | 3.60 |
| 4-8 years | 1.9 | 23 | 2.48 |
| 9-13 years | 2.3 | 42 | 1.64 |
| 14-18 years | 2.9 | 62 | 1.40 |
| Adults (19+ years) | 3.2 | 74 | 1.30 |
a Based on Ficheux et al. 2016. Assumed frequency of 1/day.
b As cited in Canadian exposure factors used in human health risk assessment (HC 2019).
c Dermal exposure calculated using the following formula: (Mean product amount (g/application) * mean daily frequency * product concentration (3%) * dermal absorption (100%) * conversion factor (1000 mg/g)) ÷ Body weight (kg)
| Age group | Mean product amount (g/application)a | Mean daily frequencyb | Body weight (kg)c | Dermal exposure (mg/kg bw/day)d |
|---|---|---|---|---|
| 9-13 years | 1.1 | 1.0 | 42 | 7.86 x 10-1 |
| 14-18 years | 1.5 | 1.0 | 62 | 7.26 x 10-1 |
| Adults (19+ years) | 1.5 | 2.0 | 74 | 1.22 |
a Based on Ficheux et al. 2016
b Based on Loretz et al. 2005; Ficheux et al. 2015
c As cited in the Canadian exposure factors used in human health risk assessment (HC 2019).
d Systemic exposure calculated using the following formula: (Mean product amount (g/application) * mean daily frequency * product concentration (100%) * conversion factor (1000 mg/g)) ÷ Body weight (kg)
| Age group | Mean product amount (g/app)a | Mean daily frequencyb | Dermal exposure (mg/kg bw/day)c |
|---|---|---|---|
| 2-3 years | 0.33 | 1.0 | 22.0 |
| 4-8 years | 0.33 | 1.0 | 14.3 |
| 9-13 years | 0.33 | 1.4 | 11.0 |
| 14-18 years | 0.33 | 1.4 | 7.45 |
| Adults (19+ years) | 0.33 | 1.7 | 7.58 |
a Based on Loretz et al. 2006
b Based on Loretz et al. 2006; Statistics Canada 2017; Minimum daily frequency was set to 1.
c Dermal exposure (mg/kg bw/day) was calculated using the following formula: (Mean product amount (g/application) * mean daily frequency * product concentration (100%) * retention factor (0.85) * conversion factor (1000 mg/g) * dermal absorption (20%)) ÷ Body weight (kg) [15 kg (age 2- to 3-year-olds); 23 kg (4- to 8-year-olds); 42 kg (9- to 13-year-olds); 62 kg (14- to 18-year-olds); 74 kg (adults (19+ yrs) (HC 2019))]
| Age group | Mean product amount (g/application)a | Mean daily frequencyb | Retention factor | Body weight (kg)c | Dermal exposure (mg/kg bw/day)d |
|---|---|---|---|---|---|
| 0-5 months | 3.9 | 1.1 | 0.01 | 6.3 | 6.19 x 10-1 |
| 6-11 months | 5.6 | 1.0 | 0.01 | 9.1 | 6.15 x 10-1 |
| 1 year | 6.1 | 1.0 | 0.01 | 11 | 5.55 x 10-1 |
| 2-3 years | 7.9 | 1.0 | 0.01 | 15 | 5.27 x 10-1 |
| 4-8 years | 9.7 | 1.0 | 0.01 | 23 | 4.22 x 10-1 |
| 9-13 years | 7.5 | 1.0 | 0.01 | 42 | 1.79 x 10-1 |
| 14-18 years | 10.4 | 1.0 | 0.01 | 62 | 1.68 x 10-1 |
|
Adults (19+ years) |
11.8 | 1.0 | 0.01 | 74 | 1.75 x 10-1 |
a Based on Loretz et al. 2008; Ficheux et al. 2016; Gomez-Berrada et al. 2013.
b Based on Loretz et al. 2008; Ficheux et al. 2015; Wu et al. 2010; Gomez-Berrada et al. 2013. Minimum daily frequency was set to 1.
c As cited in Canadian exposure factors used in human health risk assessment (HC 2019).
d Dermal exposure calculated using the following formula: (Mean product amount (g/application) * mean daily frequency * product concentration (10%) * retention factor (0.01) * conversion factor (1000 mg/g)) ÷ Body weight (kg)
| Age group | Dermal exposure cleanser (mg/kg bw/day)a | Dermal exposure moisturizer (mg/kg bw/day)b | Dermal exposure spot treatment (mg/kg bw/day)c | Total dermal exposure (mg/kg bw/day)d |
|---|---|---|---|---|
| 9-13 years | 1.77 x 10-2 | 5.24 x 10-1 | 2.62 x 10-1 | 8.03 x 10-1 |
| 14-18 years | 1.28 x 10-2 | 4.84 x 10-1 | 2.42 x 10-1 | 7.39 x 10-1 |
| Adults (19+ years) | 1.43 x 10-2 | 8.11 x 10-1 | 4.05 x 10-1 | 1.23 |
a Daily exposure from a facial cleanser was calculated using the following formula: [Mean product amount (g/application (3.1 g for 9- to 13-year-olds and 3.3 g for 14- to 18-year-olds and adults (19+ yrs) based on Ficheux et al. 2016) * Mean frequency/day (1.2 for 9- to 13-year-olds and 14- to 18-year-olds and 1.6 for adults (19+ yrs) based on Ficheux et al. 2015 and Loretz et al. 2008) * Product concentration (2%) * Retention factor (0.01) * Conversion factor (1000 mg/g)] / Body weight (42 kg for 9- to 13-year-olds, 62 kg for 14- to 18-year-olds and 74 kg for adults (19+ yrs) (HC 2019).
b Daily exposure from a medicated moisturizer was calculated using the following formula: [(Mean product amount (g/application) (1.1 g for 9- to 13-year-olds, 1.5 g for 14- to 18-year-olds and adults (19+ yrs) based on Ficheux et al. 2016) * (Mean daily frequency) (1 for 9- to 13-year-olds and 14- to 18-year-olds, 2 for adults (19+ yrs) based on Loretz et al. 2005 and Ficheux et al. 2015) * Product concentration (2%) * Conversion factor (1000 mg/g)] / Body weight (42 kg for 9- to 13-year-olds, 62 kg for 14- to 18-year-olds and 74 kg for adults (19+ yrs) (HC 2019).
c Daily exposure from an intensive spot treatment was assumed to be 50% of exposure to the medicated moisturizer based on relative product amounts. The total product amount of the intensive spot treatment was half that of the medicated moisturizer. Daily exposure from an intensive spot treatment was calculated using the following formula: Daily exposure to medicated moisturizer (2%) * 50%
d Systemic total exposure calculated using the following formula: Daily exposure from facial cleanser (mg/kg bw/day) + Daily exposure from medicated moisturizer (mg/kg bw/day) + Daily exposure from an intensive spot treatment (mg/kg bw/day)
| Age group | Mean product amount (g/application)a | Mean daily frequencyb | Body weight (kg)c | Dermal exposure (mg/kg bw/day)d |
|---|---|---|---|---|
| 6-11 months | 5.4 | 1.6 | 9.1 | 3.80 x 10-1 |
| 1 year | 5.4 | 1.6 | 11 | 3.14 x 10-1 |
| 2-3 years | 5.4 | 1.6 | 15 | 2.30 x 10-1 |
| 4-8 years | 6.3 | 1.4 | 23 | 1.53 x 10-1 |
| 9-13 years | 6.3 | 1.4 | 42 | 8.40 x 10-2 |
| 14-18 years | 18.2 | 1.4 | 62 | 1.64 x 10-1 |
| Adults (19+) | 18.2 | 1.4 | 74 | 1.38 x 10-1 |
a Based on Ficheux et al. 2016
b Based on Ficheux et al. 2015
c As cited in the Canadian exposure factors used in human health risk assessment (HC 2019).
d Dermal exposure calculated using the following formula: (Mean product amount (g/application) * mean daily frequency * product concentration (10%) * dermal absorption (100%) * conversion factor (1000 mg/g)) ÷ Body weight (kg)
| Exposure scenarioa | Age group | Concentration | Dermal exposure (mg/kg bw/day) | Inhalation exposure (mg/kg bw/day) | Oral exposure (mg/kg bw/day) |
|---|---|---|---|---|---|
| Aromatherapy | Adults | 100% | N/A | 2.24 x 10-1 (1.1 mg/m3) | N/A |
| Aromatherapy | 0 to 5 months | 100% | N/A | 6.46 x 10-1 (1.1 mg/m3) | N/A |
| Mixing, loading, and hanging hand-washed laundry | Adults | 5% | 5.43 x 10-1 | N/A | N/A |
| Mixing, loading, and application of an all-purpose floor cleaner | Adults | 1% | 5.00 x 10-2 | N/A | N/A |
| Exposure from contacting cleaned floors | 1 to 2 years | 1% | 1.83 x 10-2 | N/A | 1.38 x 10-3 |
a Details on the method and parameters used to estimate dermal and inhalation exposure to sandalwood oil from products that are available to consumers are provided in Table C-5.
N/A Not applicable
| Exposure scenario | Assumptionsa |
|---|---|
| Aromatherapy |
Essential oil air freshener scenario in ConsExpo. Models the evaporation of an essential oil using an aroma lamp. Defaults were taken from the RIVM Cosmetics Fact Sheet (RIVM, 2006). Inhalation-evaporation-constant rate model: Inhalation exposure (mg/kg bw/day): (Air concentration (24 hrs; mg/m3) * Daily inhalation rate (m3/day)) ÷ Body weight (kg) |
|
Mixing and loading a liquid for hand washing and hanging hand washed laundry (adults) |
Concentration of sandalwood oil: 5% (ACI 2018) Mixing and loading (dermal): Hand-washing (dermal): Hanging hand-washed laundry (dermal): Combined exposure: mixing/loading + hand washing + hanging machine washed laundry |
|
Mixing, loading and application of an all-purpose floor cleaner (liquid) (adults) |
Concentration of sandalwood oil: 1% (ACI 2018). Mixing and loading (dermal): Application (dermal): Combined exposure: mixing/loading + application |
|
Exposure from contacting cleaned floors (toddler) |
Concentration of sandalwood oil: 1% (ACI 2018). Calculations based on the US EPA Residential SOPs (2012), Section 7. Dermal: Deposited residue (mg/cm2): Calculated assuming 14.4 g of product per 22 m2 of floor (ConsExpo Cleaning Fact Sheet, 2018) * 1000 mg/g * 1 m2/10000 cm2 Incidental oral (i.e., hand-to-mouth exposure): HR: hand residue loading (mg/cm2); calculated using the following algorithm: HR = [Faihands * Dermal exposure (mg) (calculated above)] / (SAH * 2) Faihands: 0.15 (unitless); fraction of active ingredient on hands compared to total surface residue from jazzercise study SAH: 150 cm2; typical surface area of one hand FM: 0.13 (unitless); fraction of hand mouthed per event |
a Exposure to products was estimated using ConsExpo Web (2016). Exposure estimates were calculated based on default body weights and inhalation rates of 74 kg/15.1 m3/day for adults (19 years and older), 62 kg/15.9 m3/day for 14- to 18-year-olds, 42 kg/13.9 m3/day for 9- to 13-year-olds, 23 kg/11.1 m3/day for 4- to 8-year-olds, 15 kg/9.2 m3/day for 2- to 3-year-olds, 11 kg/8.0 m3/day for 1-year-olds, 9.1 kg/5.4 m3/day for infants aged 6 to 11 months, and 6.3 kg/3.7 m3/day for infants aged 0 to 5 months (HC 2019).
Appendix F. Exposure parameters used to estimate exposure to guaiazulene
| Age group | Mean product amount (g/application)a | Body weight (kg)b | Dermal exposure (mg/kg bw/day)c | Peak inhalation exposure (mg/m3)d |
|---|---|---|---|---|
| 0-5 months | 2 | 6.3 | 3.17 x 10-1 | 6.80 x 10-3 |
| 6-11 months | 2.5 | 9.1 | 2.75 x 10-1 | 8.50 x 10-3 |
| 1 year | 3.1 | 11 | 2.82 x 10-1 | 1.10 x 10-2 |
| 2-3 years | 4.1 | 15 | 2.73 x 10-1 | 1.30 x 10-2 |
| 4-8 years | 5 | 23 | 2.17 x 10-1 | 2.20 x 10-2 |
| 9-13 years | 7.7 | 42 | 1.83 x 10-1 | 2.70 x 10-2 |
| 14-18 years | 10 | 62 | 1.61 x 10-1 | 3.50 x 10-2 |
| Adults (19+ years) | 10 | 74 | 1.35 x 10-1 | 3.70 x 10-2 |
a Mean product amount for dermal exposure based on Ficheux et al. 2016. Assumed frequency was 1 for all age groups.
b As cited in the Canadian exposure factors used in human health risk assessment (HC 2019).
c Dermal exposure calculated using the following formula: (Mean product amount (g/application) * mean daily frequency * product concentration (0.1%) * conversion factor (1000 mg/g)) ÷ Body weight (kg)
d Peak air concentrations were modelled using ConsExpo exposure to vapour, evaporation constant release model, using the following parameters: exposure duration of 24 hours, emission duration of 24 hour, room volume of 58 m3, release area equal to unclothed surface area of application (assumed equivalent to head, arms, hands and ¾ legs (8543 cm2 for adults, 7655 cm2 for 14- to 18-year-olds, 5953 cm2 for 9- to 13 -year-olds, 3813 cm2 for 4- to 8 -year-olds, 2685 cm2 for 2- to 3 -year-olds, 2070 cm2 for 1-year-olds, 1703 cm2 for 6- to 11-month-olds, and 1325 cm2 for 0- to 5-month-olds), product amount adjusted for exposed surface area (4.87 g for adults, 4.65 g for 14- to 18-year-olds, 3.61 g for 9- to 13-year-olds, 2.3 g for 4- to 8-year-olds, 1.85 g for 2- to 3-year-olds, 1.45 g for 1-year-olds, 1.16 g for 6- to 11-month-olds, and 0.93 g for 0- to 5-month-olds), ventilation rate of 0.5/hr, molecular weight matrix of 1000 g/mol, and application temperature of 32°C.
| Age group | Mean product amount (g/application)a | Frequencyb | Body weight (kg)c | Dermal exposure (mg/kg bw/day)d | Peak air concentration (mg/m3)e |
|---|---|---|---|---|---|
| 9-13 years | 1.1 | 1 | 42 | 7.86 x 10-2 | 2.70 x 10-2 |
| 14-18 years | 1.5 | 1 | 62 | 7.26 x 10-2 | 3.10 x 10-2 |
| Adults (19+ years) | 1.5 | 2 | 74 | 1.22 x 10-1 | 4.30 x 10-2 |
a As cited in Ficheux et al. 2016 for product amount.
b As cited in Ficheux et al. 2015 and Loretz et al. 2005
c As cited in Canadian exposure factors used in human health risk assessment (HC 2019)
d Dermal exposure calculated using the following formula: (Mean product amount (g/application) * mean daily frequency * product concentration (0.3%) * conversion factor (1000 mg/g)) ÷ Body weight (kg)
e Peak air concentrations were modelled using ConsExpo exposure to vapour, evaporation constant release model, using the following parameters: exposure duration of 24 hours for 9- to 18-year-olds and 12 hours for adults (frequency of 2 times daily), emission duration of 24 hours for 9- to 18-year-olds and 12 hours for adults, room volume of 20 m3, release area equal to surface area of application (assumed equivalent to face (585 cm2 for adults, 370 cm2 for 14- to 18-year-olds, 350 cm2 for 9- to 13-year-olds), ventilation rate of 0.6/hr, molecular weight matrix of 1000 g/mol, and application temperature of 32°C.
| Age group | Mean product amount (g/application)a | Retention factor | Dermal exposure (mg/kg bw/day)b | Peak air concentration (mg/m3)c |
|---|---|---|---|---|
| 2-3 years | 5.2 | 0.1 | 3.47 x 10-2 | 1.10 x 10-2 |
| 4-8 years | 7.8 | 0.1 | 3.39 x 10-2 | 2.10 x 10-2 |
| 9-13 years | 7.8 | 0.1 | 1.86 x 10-2 | 2.60 x 10-2 |
| 14-18 years | 10 | 0.1 | 1.61 x 10-2 | 3.00 x 10-2 |
| Adults (19+ years) | 13.1 | 0.1 | 1.95 x 10-2 | 4.10 x 10-2 |
a Based on Loretz et al. 2008 for adults, Ficheux et al. 2016 for 14- to 18-year-olds, 9- to 13-year-olds and 4- to 8-year-olds, and Garcia-Hidalgo et al. 2017 for 2- to 3-year-olds; Assumed frequency of 1 for all subpopulations except for adults where a frequency of 1.1 was assumed based on Loretz et al. 2008.
b Dermal exposure calculated using the following formula: (Mean product amount (g/application) * mean daily frequency * product concentration (1%) * retention factor (0.01) * conversion factor (1000 mg/g)) ÷ Body weight (kg) (15 kg for 2- to 3-year-olds, 23 kg for 4- to 8-year-olds, 42 kg for 9- to 13-year-olds, 62 kg for 24-18-year-olds, and 74 kg for adults) (HC 2019)
c Peak air concentrations were modelled using ConsExpo exposure to vapour, evaporation constant release model, assuming 100% of the applied dose was available for evaporation using the following parameters: exposure duration of 10 min, emission duration of 10 min (based on label instructions to leave-on dry hair for 10 minutes prior to rinsing), room volume of 10 m3 (bathroom), release area equal to surface area of application (assumed equivalent to half hands and half head for all subpopulations except for 2- to 3-year-olds were it was assumed to be half head (1040 cm2 for adults, 755 cm2 for 14- to 18-year-olds, and 655 cm2 for 9- to 13-year-olds, 520 cm2 for 4- to 8-year-olds and 275 cm2 for 2- to 3-year-olds), ventilation rate of 2/hr (bathroom), molecular weight matrix of 1000 g/mol, and application temperature of 32°C.
| Age group | Mean product amount (g/application)a | Retention factor | Dermal exposure (mg/kg bw/day)b | Peak air concentration (mg/m3)c |
|---|---|---|---|---|
| 4-8 years | 132 | 0.1 | 5.74 | 7.50 x 10-2 |
| 9-13 years | 152 | 0.1 | 3.62 | 8.60 x 10-2 |
| 14-18 years | 160 | 0.1 | 2.58 | 9.10 x 10-2 |
| Adults (19+ years) | 160 | 0.1 | 2.16 | 1.40 x 10-1 |
a Based on RIVM 2006 for adults and 14- to 18-year-olds, for 4- to 13-year-olds the product amount was calculated using a surface area adjustment.
b Dermal exposure calculated using the following formula: (Mean product amount (g/application) * product concentration (1%) * retention factor (0.1) * conversion factor (1000 mg/g)) ÷ Body weight (kg) (23 kg for 4- to 8-year-olds, 42 kg for 9- to 13-year-olds, 62 kg for 14- to 18-year-olds, and 74 kg for adults) (HC 2019)
c Peak air concentrations were modelled using ConsExpo exposure to vapour, evaporation constant release model, assuming 100% of the applied dose was available for evaporation (product amount/day * 1% concentration), using the following parameters: exposure duration of 1.5 hours (perm lotion is left on the hair for a maximum of 40 minutes prior to rinsing and the fixing lotion for a maximum of 15 minutes prior to rinsing; the 1.5 hour exposure duration is to estimate the time when the perm and fixing lotion are on the hair including rinsing time), emission duration of 1.5 hours, room volume of 58 m3, release area equal to surface area of application (assumed equivalent to half head for all subpopulations (585 cm2 for adults, 370 cm2 for 14- to 18-year-olds, and 350 cm2 for 9- to 13-year-olds, and 305 cm2 for 4- to 8-year-olds)), ventilation rate of 0.5/hr, molecular weight matrix assumption of 1000 g/mol, and application temperature of 32°C.
| Age group | Mean product amount (g/application)a | Body weight (kg)b | Dermal exposure (mg/kg bw/day)c | Peak air concentration (mg/m3)d |
|---|---|---|---|---|
| 9-13 years | 2.3 | 42 | 5.48 x 10-1 | 7.70 x 10-3 |
| 14-18 years | 2.4 | 62 | 3.87 x 10-1 | 8.10 x 10-3 |
| Adults 19+ years) | 2.4 | 74 | 3.24 x 10-1 | 1.20 x 10-2 |
a As cited in Ficheux et al. 2016 for product amount. Assumed frequency was 1 for all age groups.
b As cited in Canadian exposure factors used in human health risk assessment (HC 2019)
c Dermal exposure calculated using the following formula: (Mean product amount (g/application) * product concentration (1%) * conversion factor (1000 mg/g)) ÷ Body weight (kg)
d Peak air concentrations were modelled using ConsExpo exposure to vapour, evaporation constant release model, assuming 100% of the applied dose was available for evaporation (product amount/day * 1% concentration), and using the following parameters: exposure duration of 24 hours, emission duration of 24 hour, room volume of 20 m3, release area equal to surface area of application (assumed equivalent to one-quarter the surface area of the head to approximate one-half of the face (292.5 cm2 for adults, 185 cm2 for 14- to 18-year-olds, 175 cm2 for 9- to 13-year-olds), ventilation rate of 0.6/hr, molecular weight matrix of 100 g/mol (molecular weight matrix for an aftershave formula containing 42% water and 55% ethanol (RIVM 2006)) , and application temperature of 32°C.

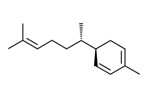
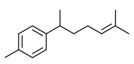
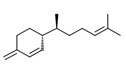
![[C(CC(C1C(C2)C(C)C)C3(C2)C)=C(C)C31]](/content/dam/eccc/images/pded/terpenes-group-2/20210203-Table2-1-9.jpg)
![CC1=CC[C@H]2C[C@@H]1[C@]2(C)CCC=C(C)C](/content/dam/eccc/images/pded/terpenes-group-2/20210203-Table2-1-10.jpg)