Screening assessment acrylates and methacrylates group
Official title: Screening assessment acrylates and methacrylates group
Chemical abstracts service registry numbers
79-10-7, 79-41-4, 97-88-1, 103-11-7, 141-32-2, 7534-94-3
Environment and Climate Change Canada
Health Canada
October 2018
Cat. No.: En14-339/2018E-PDF
ISBN 978-0-660-27750-9
Synopsis
Pursuant to section 74 of the Canadian Environmental Protection Act, 1999 (CEPA), the Minister of the Environment and the Minister of Health have conducted a screening assessment of six of nine substances referred to collectively under the Chemicals Management Plan as the Acrylates and Methacrylates Group. These six substances were identified as priorities for assessment as they met categorization criteria under subsection 73(1) of CEPA. Three of the nine substances were subsequently determined to be of low concern through other approaches, and decisions for these substances are provided in separate reports. Footnote 1 , Footnote 2 Accordingly, this screening assessment addresses the six substances listed in the table below. The six substances addressed in this screening assessment will hereinafter be referred to as the Acrylates and Methacrylates Group.
CAS RNa |
Domestic Substances List name |
Common name |
79-10-7 |
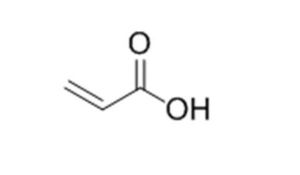
2-Propenoic acid |
Acrylic acid |
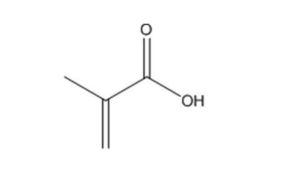
79-41-4 |
2-Propenoic acid, 2-methyl- |
Methacrylic acid |
97-88-1 |
2-Propenoic acid, 2-methyl-, butyl ester |
n-Butyl methacrylate |
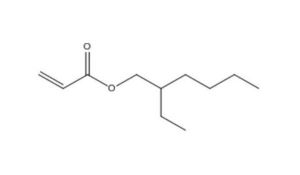
103-11-7 |
2-Propenoic acid, 2-ethylhexyl ester |
2-Ethylhexyl acrylate |
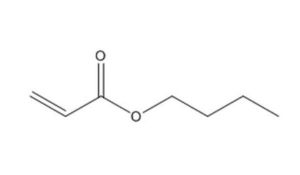
141-32-2 |
2-Propenoic acid, butyl ester |
Butyl acrylate |
7534-94-3 |
2-Propenoic acid, 2-methyl-, 1,7,7-trimethylbicyclo[2.2.1]hept-2-yl ester, exo- |
Isobornyl methacrylate |
a The Chemical Abstracts Service Registry Number (CAS RN) is the property of the American Chemical Society and any use or redistribution, except as required in supporting regulatory requirements and/or for reports to the Government of Canada when the information and the reports are required by law or administrative policy, is not permitted without the prior, written permission of the American Chemical Society.
Acrylic acid occurs naturally in marine algae, and methacrylic acid occurs naturally in oil from Roman chamomile. The other four substances do not occur naturally in the environment. Most of the substances in this group have many applications, including manufacture of polymers. According to information submitted pursuant to a survey under section 71 of CEPA, all six substances in the Acrylates and Methacrylates Group were imported into Canada in total quantities ranging from 10,000 to 22 million kg in the 2011 reporting year. In the same year, no Canadian manufacturing was reported for any of the six substances above the reporting threshold of 100 kg. Substances in the Acrylates and Methacrylates Group are used in commercial products and products available to consumers, including adhesives and sealants, paints and coatings, plastic and rubber materials, paper products, cosmetics, and building or construction materials.
The ecological risks of the substances in the Acrylates and Methacrylates Group were characterized using the Ecological Risk Classification of Organic Substances (ERC). The ERC is a risk-based approach that employs multiple metrics for both hazard and exposure, with weighted consideration of multiple lines of evidence for determining risk classification. Hazard profiles based principally on metrics regarding mode of toxic action, chemical reactivity, food web-derived internal toxicity thresholds, bioavailability, and chemical and biological activity are established. Metrics considered in the exposure profiles include potential emission rate, overall persistence, and long-range transport potential. A risk matrix is used to assign a low, moderate or high level of potential concern for substances on the basis of hazard and exposure profiles. The ERC identified the six substances in the Acrylates and Methacrylates Group as having low potential to cause ecological harm.
Considering all available lines of evidence presented in this screening assessment, there is low risk of harm to the environment from acrylic acid, methacrylic acid, n-butyl methacrylate, 2-ethylhexyl acrylate, butyl acrylate, and isobornyl methacrylate. It is concluded that acrylic acid, methacrylic acid, n-butyl methacrylate, 2-ethylhexyl acrylate, butyl acrylate, and isobornyl methacrylate do not meet the criteria under paragraphs 64(a) or (b) of CEPA as they are not entering the environment in a quantity or concentration or under conditions that have or may have an immediate or long-term harmful effect on the environment or its biological diversity or that constitute or may constitute a danger to the environment on which life depends.
For the general population of Canada, potential exposures to the Acrylates and Methacrylates Group from various environmental media and food were characterized. Estimates of exposure from use of products available to consumers, including cosmetics, were derived. Exposure is expected to occur mainly from products available to consumers.
The critical health effects for the Acrylates and Methacrylates Group are decreased body weight gain and liver and kidney toxicity at higher doses in laboratory studies. The substances are not considered to be carcinogenic, genotoxic or reproductive toxicants and do not cause developmental effects in the absence of maternal toxicity in laboratory studies.
Margins of exposure comparing effect levels for the critical health effects and the estimates of exposure from uses of products available to consumers were considered adequate to address uncertainties in the health effects and exposure databases for the substances in the Acrylates and Methacrylates Group.
On the basis of the information presented in this screening assessment, it is concluded that acrylic acid, methacrylic acid, n-butyl methacrylate , 2-ethylhexyl acrylate, butyl acrylate and isobornyl methacrylate do not meet the criteria under paragraph 64(c) of CEPA as they are not entering the environment in a quantity or concentration or under conditions that constitute or may constitute a danger in Canada to human life or health.
It is concluded that acrylic acid, methacrylic acid, n-butyl methacrylate, 2-ethylhexyl acrylate, butyl acrylate, and isobornyl methacrylate do not meet any of the criteria set out in section 64 of CEPA.
1. Introduction
Pursuant to section 74 of the Canadian Environmental Protection Act, 1999 (CEPA) (Canada 1999), the Minister of the Environment and the Minister of Health have conducted a screening assessment of six of nine substances referred to collectively under the Chemicals Management Plan as the Acrylates and Methacrylates Group, to determine whether these six substances present or may present a risk to the environment or to human health. These six substances were identified as priorities for assessment as they met categorization criteria under subsection 73(1) of CEPA (ECCC, HC [modified 2017]).
The other three substances (listed in Table 1‑1 below) were considered in the ecological risk classification of organic substances (ERC) Science Approach Document (ECCC 2016a) and in either the Threshold of Toxicological Concern (TTC)-based Approach for Certain Substances Science Approach Document (Health Canada 2016) or via the approach applied in the Rapid Screening of Substances with Limited General Population Exposure (ECCC, HC 2017a), and were identified as being of low concern to both human health and the environment. As such, they are not further addressed in this report. Conclusions for these three substances are provided in the Substances Identified as Being of Low Concern based on the Ecological Risk Classification of Organic Substances and the Threshold of Toxicological Concern (TTC)-based Approach for Certain Substances Draft Screening Assessment (ECCC, HC 2017b) and the Rapid Screening of Substances with Limited General Population Exposure Draft Screening Assessment (ECCC, HC 2017a). Accordingly, this screening assessment addresses the six substances listed in the table below. The six substances addressed in this screening assessment will hereinafter be referred to as the Acrylates and Methacrylates Group.
CAS RN |
Domestic Substances List name |
Approach under which the substance was addressed |
References |
122-68-9 |
2-Propenoic acid, 3-phenyl-, 3-phenylpropyl ester |
ERC/TTC |
ECCC, HC 2017b |
24448-20-2 |
2-Propenoic acid, 2-methyl-, (1-methylethylidene)bis(4, 1-phenyleneoxy-2,1-ethanediyl) ester |
ERC/Rapid Screening
|
ECCC, HC 2017a |
43048-08-4 |
2-Propenoic acid, 2-methyl-, (octahydro-4,7-methano-1H- indene-5,?-diyl)bis(methylene) ester |
ERC/Rapid Screening |
ECCC, HC 2017a |
The six other substances will be addressed in this screening assessment.
The substances in the Acrylates and Methacrylates Group were grouped together given their structural similarity, with the methacrylates having an additional methyl group. Acrylic acid, 2-ethylhexyl acrylate and methacrylic acid were reviewed by the European Commission (EC) (EC 2002a, 2002b, 2005). Acrylic acid, 2-ethylhexyl acrylate, butyl acrylate, methacrylic acid, n-butyl methacrylate and isobornyl methacrylate were reviewed by the Organisation for Economic Cooperation and Development (OECD) in Screening Information Dataset Initial Assessment Reports (SIARs) (OECD 2001a, 2001b, 2002a, 2002b, 2003, 2004a, 2004b, 2011b). These OECD assessments undergo rigorous review and endorsement by international governmental authorities. Health Canada and Environment and Climate Change Canada are active participants in this process and consider these assessments reliable. The EC Risk Assessment Reports and OECD SIARs inform the health effects characterization in this screening assessment.
The ecological risks of substances in the Acrylates and Methacrylates Group were characterized using the ERC approach (ECCC 2016a). The ERC describes the hazard of a substance using key metrics, including mode of toxic action, chemical reactivity, food web-derived internal toxicity thresholds, bioavailability, and chemical and biological activity, and considers the possible exposure of organisms in the aquatic and terrestrial environments on the basis of such factors as potential emission rates, overall persistence and long-range transport potential in air. The various lines of evidence are combined to identify substances as warranting further evaluation of their potential to cause harm to the environment or as having a low likelihood of causing harm to the environment.
This screening assessment includes consideration of information on chemical properties, environmental fate, hazards, uses, and exposures, including additional information submitted by stakeholders. Relevant data were identified up to December 2016. Empirical data from key studies as well as some results from models were used to reach proposed conclusions. When available and relevant, information presented in assessments from other jurisdictions was considered.
This screening assessment was prepared by staff in the CEPA Risk Assessment Program at Health Canada and Environment and Climate Change Canada and incorporates input from other programs within these departments. The ecological portion of this assessment is based on the ERC document (published July 30, 2016), which was peer-reviewed and subject to a 60-day public comment period. Additionally, the draft of this screening assessment (published September 23, 2017) was subject to a 60-day public comment period. While external comments were taken into consideration, the final content and outcome of the screening assessment remain the responsibility of Health Canada and Environment and Climate Change Canada.
This screening assessment focuses on information critical to determining whether substances meet the criteria as set out in section 64 of CEPA by examining scientific information and incorporating a weight of evidence approach and precaution.Footnote 3 This screening assessment presents the critical information and considerations on which the proposed conclusion is based.
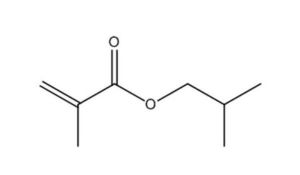
2. Identity of substances
The CAS RN, Domestic Substances List (DSL) names and common names and/or acronyms for the individual substances in the Acrylates and Methacrylates Group are presented in Table 2‑1. Given the structural similarities in their respective metabolic products, acrylic acid and two of its esters are listed first, followed by methacrylic acid and two of its esters.
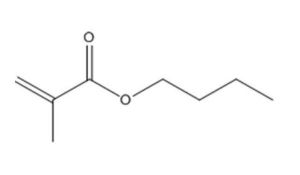
CAS RN |
DSL name |
Chemical structure and molecular formula |
Molecular weight (g/mol) |
|---|---|---|---|
79-10-7 |
2-Propenoic acid |
|
72.06 |
103-11-7 |
2-Propenoic acid, 2-ethylhexyl ester |
|
184.3 |
141-32-2 |
2-Propenoic acid, butyl ester |
|
128.2 |
79-41-4 |
2-Propenoic acid, 2-methyl- |
|
86.09 |
97-88-1 |
2-Propenoic acid, 2-methyl-, butyl ester |
|
142.2 |
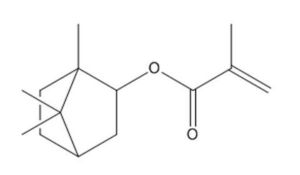
7534-94-3 |
2-Propenoic acid, 2-methyl-, 1,7,7-trimethylbicyclo [2.2.1]hept-2-yl ester, exo- |
|
222.3 |
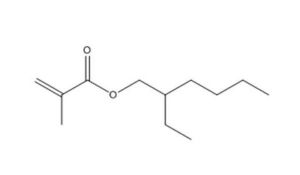
2.1 Selection of analogues and use of (Q)SAR models
CAS RN |
DSL or other name |
Chemical structure and molecular formula |
Molecular weight (g/mol) |
|---|---|---|---|
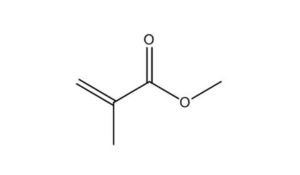
80-62-6 |
2-Propenoic acid, 2-methyl-, methyl ester |
|
100.1 |
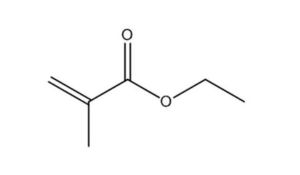
97-63-2 |
2-Propenoic acid, 2-methyl-, ethyl ester (Ethyl methacrylate) |
|
114.1 |
97-86-9 |
2-Propenoic acid, 2-methyl-, 2-methylpropyl ester (Isobutyl methacrylate) |
|
142.2 |
688-84-6 |
2-Propenoic acid, 2-methyl-, 2-ethylhexyl ester (2-Ethylhexyl methacrylate) |
|
198.3 |
3. Physical and chemical properties
A summary of the range of key physical and chemical properties of the substances in the Acrylates and Methacrylates Group are presented in Table 3‑1. Additional physical and chemical properties are presented in ECCC (2016b). Key physical and chemical properties for analogues of the Acrylates and Methacrylates Group are presented in Table 3-2.
Property |
Range |
Key reference |
Vapour pressure (mm Hg) |
0.011–5.45 |
EPI Suite c2000-2012 |
Water solubility (mg/L) |
2.89–1.00 × 106 |
EPI Suite c2000-2012 |
log Kow (dimensionless) |
0.35–4.76 |
EPI Suite c2000-2012 |
Abbreviations: Kow, octanol–water partition coefficient.
a Modelled values were used for isobornyl methacrylate and experimental values were used for the remaining substances in the Acrylates and Methacrylates Group.
Common name |
Property |
Value |
Key reference |
Methyl methacrylate |
Vapour pressure (mm Hg) |
38.5 |
EPI Suite c2000-2012 |
Water solubility (mg/L) |
1.5 × 104 |
||
log Kow (dimensionless) |
1.38 |
||
Ethyl methacrylate
|
Vapour pressure (mm Hg) |
20.6 |
EPI Suite c2000-2012 |
Water solubility (mg/L) |
5400 |
||
log Kow (dimensionless) |
1.94 |
||
Isobutyl methacrylate
|
Vapour pressure (mm Hg) |
3.63 |
EPI Suite c2000-2012 |
Water solubility (mg/L) |
1300 |
||
log Kow (dimensionless) |
2.66 |
||
2-Ethylhexyl methacrylate
|
Vapour pressure (mm Hg) |
0.0758 a |
EPI Suite c2000-2012 |
Water solubility (mg/L) |
5.92 a |
||
log Kow (dimensionless) |
4.54 |
Abbreviations: Kow, octanol–water partition coefficient.
a Modelled values were used.
4. Sources and uses
Acrylic acid occurs naturally in marine algae, and methacrylic acid occurs naturally in oil from Roman chamomile, whereas the other four substances do not occur naturally in the environment (IARC 1979; Merck Index 2006).
Use information for each of the substances identified in this section may reflect use as a reacted component of a polymer, in part or in whole, which may in turn be used in the manufacture of specific end products.
All of the substances in the Acrylates and Methacrylates Group have been included in a survey issued pursuant to a CEPA section 71 notice (Environment Canada 2013). Table 4‑1 presents a summary of the total reported import quantities for the substances in the Acrylates and Methacrylates Group in Canada for the 2011 calendar year. No manufacturing activity above the 100 kg reporting threshold was reported for any of the six substances in Canada.
Common name |
Total imports (kg) |
Acrylic acid |
443,024 |
2-Ethylhexyl acrylate |
1,000,000–10,000,000 |
Butyl acrylate |
21,634,074 |
Methacrylic acid |
1,000,000–10,000,000 |
n-Butyl methacrylate |
100,000–1,000,000 |
Isobornyl methacrylate |
10,000–100,000 |
a Values reflect quantities reported in response to a survey conducted under section 71 of CEPA (Environment Canada 2013). See survey for specific inclusions and exclusions (schedules 2 and 3).
Major Canadian commercial and consumer uses of the Acrylates and Methacrylates Group according to information submitted pursuant to a section 71 survey under CEPA are described (Environment Canada 2013). All substances in the Acrylates and Methacrylates Group are used as chemical intermediates to produce other substances. Acrylic acid, 2-ethylhexyl acrylate, and butyl acrylate are used in adhesives and sealants and in paper products, mixtures or manufactured items. The substances in the Acrylates and Methacrylates Group are all used in paints and coatings. Acrylic acid is used in floor coverings, water treatment, plastic and rubber materials, inks, toners and colourants, automotive products, cleaning and furnishing care, electrical and electronics, toys, playground and sporting equipment, building and construction materials, oil and natural gas extraction, and polymer manufacturing. 2-Ethylhexyl acrylate is used in ink, toner and colourants, lubricants and greases, and building and construction materials. Butyl acrylate is used in floor coverings, plastic and rubber materials, ink, toner and colourants, automotive products, toys, and playground and sporting equipment. Methacrylic acid is used in adhesives and sealants and building and construction materials. n-Butyl methacrylate is used in plastic and rubber materials, lubricants and greases, and automotive, aircraft and other transportation applications. Other use information has been identified for the substances in the Acrylates and Methacrylates Group, including adhesives for general purpose bonding, automobile repairs and maintenance, and markers intended for use by children (e.g. MSDS 2009, 2012, 2014a, 2014b).
Internationally, the substances in the Acrylates and Methacrylates Group are used in the manufacture of polymers, which in turn are used in products such as adhesives, paints, coatings, inks, plastics and textiles (EC 2002a, b, 2005; OECD 2001a, b, 2002a, b, 2003, 2004a, 2011b). For example, it is possible for residual monomers to be present in paints and coatings (EC 2002a, b, 2005; OECD 2001a, 2001b, 2002a, 2002b, 2003, 2004a, 2011b). Acrylic acid and butyl acrylate readily polymerize if not controlled by inhibitors (Arkema 2012a, 2012b).
In Canada, the six substances in the Acrylates and Methacrylates Group have been identified as being used in the manufacture of a variety of food packaging materials, including paper and paperboard, plastic films, can coatings, and inks. Acrylic acid, methacrylic acid, and 2-ethylhexyl acrylate have also been identified as components of incidental additivesFootnote 4 used in food processing plants (personal communication, emails from the Food Directorate, Health Canada, to the Existing Substances Risk Assessment Bureau, Health Canada, 2016; unreferenced).
Methacrylic acid is present as a non-medicinal ingredient in a non-prescription drug used as a laxative in Canada, while the remaining five substances are not listed in the Drug Product Database as being present in non-prescription drugs in Canada (DPD [modified 2015]; personal communication, emails from the Therapeutic Products Directorate, Health Canada, to the Existing Substances Risk Assessment Bureau, Health Canada, 2016; unreferenced). Specific information regarding quantity of methacrylic acid was not available (personal communication, emails from the Therapeutic Products Directorate, Health Canada, to the Existing Substances Risk Assessment Bureau, Health Canada, 2016; unreferenced). Methacrylic acid may be polymerized in the final product (Chang et al. 2016).
2-Ethylhexyl acrylate is listed in the Natural Health Products Ingredients Database as a non-medicinal ingredient as binder in natural health products; however, like the other substances in the Acrylates and Methacrylates Group, it is not listed in the Licensed Natural Health Products Database as being present in currently licensed natural health products in Canada (NHPID [modified 2018]; LNHPD [modified 2018]).
All six substances are also used in a variety of cosmetics in Canada, including nail polishes, nail adhesives, adhesive removers, bleaches, cleansers, conditioners, hair grooming products, makeup, and moisturizers (personal communication, emails from the Consumer Product Safety Directorate, Health Canada, to the Existing Substances Risk Assessment Bureau, Health Canada, 2016; unreferenced). Methacrylic acid is on the Cosmetic Ingredient Hotlist.Footnote 5 It is restricted for use in cosmetic products, and cosmetic uses of methacrylic acid at concentrations exceeding 5% require additional label warnings (Health Canada [modified 2015]).
The substances in the Acrylates and Methacrylates Group, with the exception of isobornyl methacrylate, are used as formulants in pesticides in Canada (personal communication, emails from the Pest Management Regulatory Agency, Health Canada, to the Existing Substances Risk Assessment Bureau, Health Canada, 2016; unreferenced).
5. Potential to cause ecological harm
5.1 Characterization of ecological risk
The ecological risks of the six substances in the Acrylates and Methacrylates Group were characterized using the ecological risk classification of organic substances (ERC) (ECCC 2016a). The ERC is a risk-based approach that considers multiple metrics for both hazard and exposure, with weighted consideration of multiple lines of evidence for determining risk classification. The various lines of evidence are combined to discriminate between substances of lower or higher potency and lower or higher potential for exposure in various media. This approach reduces the overall uncertainty with risk characterization compared to an approach that relies on a single metric in a single medium (e.g., LC50) for characterization. The following summarizes the approach, which is described in detail in ECCC (2016a).
Data on physical-chemical properties, fate (chemical half-lives in various media and biota, partition coefficients, and fish bioconcentration), acute fish ecotoxicity, and chemical import or manufacture volume in Canada were collected from scientific literature, from available empirical databases (e.g., OECD QSAR Toolbox), and from responses to surveys under section 71 of CEPA or were generated using selected quantitative structure-activity relationship (QSAR) or mass-balance fate and bioaccumulation models. These data were used as inputs to other mass-balance models or to complete the substance hazard and exposure profiles.
Hazard profiles were established principally on the basis of metrics regarding mode of toxic action, chemical reactivity, food web-derived internal toxicity thresholds, bioavailability, and chemical and biological activity. Exposure profiles were also established using multiple metrics, including potential emission rate, overall persistence, and long-range transport potential. Hazard and exposure profiles were compared to decision criteria in order to classify the hazard and exposure potentials for each organic substance as low, moderate, or high. Additional rules were applied (e.g., classification consistency, margin of exposure) to refine the preliminary classifications of hazard or exposure.
A risk matrix was used to assign a low, moderate or high classification of potential risk for each substance on the basis of hazard and exposure classifications. ERC classifications of potential risk were verified using a two-step approach. The first step adjusted the risk classification outcomes from moderate or high to low for substances that had a low estimated rate of emission to water after wastewater treatment, representing a low potential for exposure. The second step reviewed low risk potential classification outcomes using relatively conservative, local-scale (i.e., in the area immediately surrounding a point-source of discharge) risk scenarios, designed to be protective of the environment, to determine whether the classification of potential risk should be increased.
ERC uses a weighted approach to minimize the potential for both over-and under-classification of hazard, exposure and subsequent risk. The balanced approaches for dealing with uncertainties are described in greater detail in ECCC 2016a. The following describes two of the more substantial areas of uncertainty. Error in empirical or modeled acute toxicity values could result in changes in classification of hazard, particularly metrics relying on tissue residue values (i.e., mode of toxic action), many of which are predicted values from QSAR models. The impact of this error is mitigated, however, by the fact that overestimation of median lethality will result in a conservative (protective) tissue residue used for critical body residue (CBR) analysis. Error in underestimation of acute toxicity will be mitigated through the use of other hazard metrics, such as structural profiling of mode of action, reactivity and/or estrogen binding affinity. Changes or errors in chemical quantity could result in differences in classification of exposure as the exposure and risk classifications are highly sensitive to emission rate and use quantity. The ERC classifications thus reflect exposure and risk in Canada on the basis of what is believed to be the current use quantity, and may not reflect future trends.
Critical data and considerations used to develop the substance-specific profiles for the six substances in the Acrylates and Methacrylates Group, and the hazard, exposure and risk classification results, are presented in ECCC (2016b).
The hazard and exposure classifications for the six substances in the Acrylates and Methacrylates Group are summarized in Table 5-1.
Substance |
ERC hazard classification |
ERC exposure classification |
ERC risk classification |
Acrylic acid |
low |
low |
low |
2-Ethylhexyl acrylate |
low |
low |
moderate |
Butyl acrylate |
high |
low |
moderate |
Methacrylic acid |
low |
low |
low |
n-Butyl methacrylate |
low |
low |
low |
Isobornyl methacrylate |
moderate |
low |
low |
On the basis of low hazard and low exposure potential, acrylic acid, methacrylic acid, and n-butyl methacrylate were classified as having a low potential for ecological risk. It is unlikely that these substances result in concerns for the environment in Canada.
According to information considered under ERC, 2-ethylhexyl acrylate was classified as having a low hazard and low exposure potential, although with greater potential for local-scale exposures; therefore, this substance was classified as having a moderate potential for ecological risk. On the basis of current use patterns, this substance is unlikely to result in concerns for the environment in Canada.
According to information considered under ERC, butyl acrylate was classified as having a low exposure potential. Butyl acrylate was classified as having a high hazard potential on the basis of the agreement between the reactive mode of action and elevated toxic ratio, both of which suggest that this chemical is likely of high potency. In addition, structural alerts from the OECD toolbox identified this substance as being a potential protein binder. Butyl acrylate was classified as having a moderate potential for ecological risk. The potential effects and how they may manifest in the environment were not further investigated due to the low exposure of this substance. On the basis of current use patterns, this substance is unlikely to result in concerns for the environment in Canada.
According to information considered under ERC, isobornyl methacrylate was classified as having a low exposure potential. Isobornyl methacrylate was classified as having a moderate hazard potential on the basis of the reactive mode of action and potential to cause adverse effects in aquatic foodwebs given its bioaccumulation potential. In addition, structural alerts from the OECD toolbox identified this substance as being a potential protein binder. Isobornyl methacrylate was classified as having a low potential for ecological risk. The potential effects and how they may manifest in the environment were not further investigated due to the low exposure of this substance. On the basis of current use patterns, this substance is unlikely to result in concerns for the environment in Canada.
6. Potential to cause harm to human health
6.1 Exposure assessment
Potential exposures to substances in the Acrylates and Methacrylates Group from environmental media, food and products available to consumers are presented in this section. Additional details regarding the exposure scenarios for products available to consumers are summarized in Appendix B.
Environmental media and food
Two substances, acrylic acid and butyl acrylate, were reported as total releases (majority to air) at a rate of 0.032 and 2.4 tonnes/year in Canada, respectively, in 2016 (NPRI 2016). Using the 2011 total import quantities of these substances (Environment Canada 2013), their concentrations in environmental media were modelled under three theoretical release scenarios: 100% emission into air, water or soil (ChemCAN 2003). ChemCAN v6.00 simulations conservatively assumed that total import quantities were released into a single region of Canada, i.e., the Ontario Mixed-Wood Plain region, at a 100% emission factor and assuming 0% removal for wastewater treatment processes (for water releases). Theoretical total intakes were estimated for the six substances and for the three theoretical release scenarios. The theoretical total intakes of butyl acrylate from environmental media were estimated to be the highest of all six substances, with an intake of up to 0.077 µg/kg bw per day for formula-fed infants (0 to 6 months) on the basis of a 100% release scenario to water.
Dietary exposure, if any, from the use of substances in the Acrylates and Methacrylates Group in the manufacture of food packaging materials is expected to be less than 200 ng/kg bw per day. Dietary exposure, if any, from incidental additives is expected to be negligible (personal communication, emails from the Food Directorate, Health Canada, to the Existing Substances Risk Assessment Bureau, Health Canada, 2016; unreferenced).
Products available to consumers
Direct exposures from the use of products available to consumers were evaluated. Key studies and estimates of potential exposure are presented in this section. For each substance, exposures were estimated for use of products expected to represent the highest exposures to humans, and thus were considered protective of exposures from other products that have been identified. It is recognized that some of the concentrations of the individual substances used in the exposure scenarios may not reflect the actual residual concentration of unreacted substances remaining in products prior to use by consumers. However, concentration data were used as provided (see Tables 6-1 to 6-3 for details). Estimates of oral exposure to the substances in the Acrylates and Methacrylates Group are on the basis of the use of markers intended for children. Estimated oral exposures were quantified for acrylic acid and methacrylic acid (Table 6‑1). Estimates were calculated on a per event basis and were also averaged using use frequency to a daily estimate. Exposures were calculated using the highest exposed age group on a per body weight basis (toddlers 6 months to 4 years old).
Substance |
Product type |
Concentration (% by weight) |
Per event exposure (mg/kg bw) |
Daily exposure (mg/kg bw per day) |
Acrylic acid |
Water marker a |
5 |
0.16 |
0.0081 |
Methacrylic acid |
Marker ink b |
30 |
0.97 |
0.048 |
a MSDS 2014b.
b MSDS 2009.
Table 6‑2 presents estimated dermal exposures. Toddlers (6 months to 4 years old) were used to calculate the methacrylic acid exposure from marker ink. The scenarios for cosmetics were calculated on the basis of adults (20 to 59 years old). Exposure scenarios for nail products and adhesives were considered to represent intermittent i.e., per event exposures. Daily exposures and per event exposures were considered for other cosmetics given the frequencies of use.
For estimated potential exposures via the dermal route, 100% dermal absorption was conservatively used to characterize the exposures for acrylic acid, 2-ethylhexyl acrylate, butyl acrylate, methacrylic acid, and n-butyl methacrylate. Dermal absorption of isobornyl methacrylate was on the basis of 15% dermal absorption of neat methyl methacrylate through human epidermis in vitro with dermal load of 9430 µg/cm2 (Betts et al. 2006). After 10 hours, dermal absorption of methyl methacrylate was 15 or 0.56% for occluded or unoccluded exposures, respectively, but the latter was not used due to the high vapour pressure and expected evaporation of methyl methacrylate. Values for recovery and skin-bound residue were not described in the study. On the basis of their physical and chemical properties, in particular the larger size and lipophilicity of the isobornyl moiety, isobornyl methacrylate is expected to have lower dermal absorption than methyl methacrylate.
Substance |
Product type |
Concentration (% by weight) a |
Per event exposure (mg/kg bw) |
Daily exposure (mg/kg bw per day) |
Acrylic acid |
Face moisturizer |
3 |
0.51b |
0.91 b |
2-Ethylhexyl acrylate |
Facial cleanser |
10 |
0.037 b |
0.059 |
2-Ethylhexyl acrylate |
Press on manicure adhesive |
70 |
0.40 |
N/A |
Butyl acrylate |
Adhesive for eyes |
3 |
0.0038 b |
0.0046 b |
Butyl acrylate |
Nail polish |
3 |
0.068 b |
N/A |
Methacrylic acid |
Marker ink |
30 |
0.97 b |
0.048 b |
n-Butyl methacrylate |
Hair grooming gel |
8 |
0.21 b |
0.12 b |
n-Butyl methacrylate |
Adhesive in nails |
50 |
0.28 b |
N/A |
Isobornyl methacrylate |
Nail polish |
30 |
0.10 c |
N/A |
a Concentrations are on the basis of notifications submitted under the Cosmetic Regulations to Health Canada (personal communication, emails from the Consumer Product Safety Directorate, Health Canada, to the Existing Substances Risk Assessment Bureau, Health Canada, 2016; unreferenced).
b Dermal absorption is conservatively assumed to be 100%.
c Estimated systemic exposure values incorporate a dermal absorption factor of 15%.
Table 6‑3 presents the estimated inhalation exposures for nail products and do-it-yourself products. Per event exposure scenarios were calculated on the basis of adults (20 to 59 years old).
Substance |
Product type |
Concentration (% by weight) a |
Per event exposure (mg/m3) |
Acrylic acid |
Nail gel manicure preparation |
94 |
0.78 |
2-Ethylhexyl acrylate |
Press on manicure adhesive |
70 |
0.55 |
Butyl acrylate |
Nail polish |
3 |
0.44 |
Methacrylic acid |
Epoxy adhesive |
10 b |
3.1 |
n-Butyl methacrylate |
Adhesive in nails |
50 |
0.41 |
Isobornyl methacrylate |
Nail polish |
30 |
0.54 |
a Concentrations are on the basis of notifications submitted under the Cosmetic Regulations to Health Canada (personal communication, emails from the Consumer Product Safety Directorate, Health Canada, to the Existing Substances Risk Assessment Bureau, Health Canada, 2016; unreferenced).
b MSDS 2014a.
Other potential exposure scenarios for cosmetics and do-it-yourself products are considered to have lower exposure or are for specialized use where the general population exposure is expected to be lower than the scenarios presented in this section. Substances in the Acrylates and Methacrylates Group can be polymerized during the manufacturing of paints and coatings (EC 2002a, 2002b, 2005; OECD 2001a, 2001b, 2002a, 2002b, 2003, 2004a, 2011b). Considering the expected concentrations of these substances in the final products, consumer exposures from residual monomers are expected to be lower than those of the scenarios calculated in this assessment.
6.2 Health effects assessment
The health effects assessment for the Acrylates and Methacrylates Group was on the basis of the European Commission (EC) Risk Assessment Reports for acrylic acid (EC 2002a), 2-ethylhexyl acrylate (EC 2005), and methacrylic acid (EC 2002b). The OECD Screening Information Dataset (SIDS) Initial Assessment Reports were used to assess the health effects of butyl acrylate (OECD 2002a, 2002b), n-butyl methacrylate (OECD 2004a, 2004b), and isobornyl methacrylate (OECD 2011). When required, dose conversions were calculated using Health Canada’s reference values for intakes (Health Canada 1994). A literature search was conducted from 1996 to December 1, 2016; no significant new studies were identified that impacted the health effects assessment.
Substances in the Acrylates and Methacrylates Group are rapidly absorbed by oral (acrylic acid, 2-ethylhexyl acrylate, butyl acrylate, methacrylic acid, and n-butyl methacrylate), dermal (acrylic acid and butyl methacrylate), and inhalation (acrylic acid, methacrylic acid, and n-butyl methacrylate) routes (EC 2002a, 2002b, 2005; OECD 2002a, 2002b, 2003, 2004a).
Acrylic acid is rapidly metabolized to carbon dioxide and expired (EC 2002a), whereas 2-ethylhexyl acrylate and butyl acrylate are hydrolyzed to acrylic acid and to 2-ethylhexanol and butanol, respectively (OECD 2002b, 2003). Despite its physico-chemical properties, the systemic availability of methacrylic acid is estimated to be low (EC 2002b). n-Butyl methacrylate is rapidly metabolized to methacrylic acid and butanol (OECD 2004b). Limited data for isobornyl methacrylate are available, but in general, methacrylates metabolize to methacrylic acid and the corresponding alcohols (OECD 2011).
A read-across approach using data from analogues and the results of (Q)SAR models, where appropriate, have been used to inform the health effects assessment where insufficient health effects data were available. Analogues were selected on the basis of structure and/or function similarities to substances within this group (e.g., on the basis of physical-chemical properties) or they were identified as metabolites that had relevant empirical data that could be used. Details of the read-across data to inform the health effects assessments of the Acrylates and Methacrylates Group are provided below.
2-Ethylhexyl acrylate, butyl acrylate, and n-butyl methacrylate are skin sensitizers (EC 2005; OECD 2002b, 2003, 2004b).
Acrylic acid
The European Commission considered acrylic acid unlikely to be mutagenic on the basis of both experimental data and data from structurally related acrylic compounds. It is not considered to be a reproductive or developmental toxicant nor is it carcinogenic on the basis of long-term studies in animals (EC 2002a). A 90-day drinking water study in rats established a No Observed Adverse Effect Level (NOAEL) at the lowest dose of 83 mg/kg bw per day on the basis of decreased body weight gain in females only, decreased water consumption and increased kidney weights in both sexes and increased testes weights at 250 mg/kg bw per day (Bushy Run Research Center 1980, as cited in EC 2002a). A 12-month drinking water study in rats established a NOAEL of 40 mg/kg bw per day in males on the basis of decreased body weight gain and water consumption at 100 mg/kg bw per day, while a NOAEL of 375 mg/kg bw per day, the highest dose tested, was established for females (Hellwig 1993, as cited in EC 2002a).
No reproductive toxicity was observed in rats in a two-generation drinking water study at the highest dose of 460 mg/kg bw per day, with a NOAEL of 240 mg/kg bw per day for the parents (F0) on the basis of the critical effects of decreased food and drinking water consumption in females during the first 10 weeks through to lactation (Hellwig 1997, as cited in EC 2002a). Effects were observed in the offspring with a NOAEL of 53 mg/kg bw per day on the basis of decreased body weight (F1 and F2 pups) and the critical effect of decreased body weight gain (F1 pups from day 7 post-partum onwards) and decreased food and drinking water consumption.
In an inhalation study, no developmental toxicity was observed in rabbits exposed to acrylic acid, and a No Observed Adverse Effect Concentration (NOAEC) of 73 mg/m3 for maternal toxicity was established on the basis of the critical effects of dose-related perinasal/perioral wetness, nasal congestion, decreased body weight gain and decreased food consumption observed at the mid dose of 222 mg/m3 and higher (Neeper-Bradley et al. 1997).
In a dermal carcinogenicity study in rats, no treatment-related effects of skin irritation, toxicity, or skin tumours were observed with a NOAEL of 51 mg/kg bw per day, the highest dose tested (EC 2002a; BAMM 1990, 1991; TSCATS 1992).
2-Ethylhexyl acrylate
Experimental data from lifetime skin painting studies in male C3H/HeJ mice, with doses up to 1,081 mg/kg bw per day, found that 2-ethylhexyl acrylate induced skin tumours at concentrations that were highly irritating (21.5% equivalent to 269 mg/kg bw per day), but this was not confirmed in NMRI mice (Wenzel-Hartung 1989). Taking into account the negative experimental results from long-term oral and dermal animal studies with the cleavage product acrylic acid, 2-ethylhexyl acrylate is not considered carcinogenic (EC 2005). The European Commission determined that 2-ethylhexyl acrylate is not mutagenic in vivo on the basis of in vitro and limited in vivo data for the metabolites 2-ethylhexanol and acrylic acid (EC 2005).
In a developmental toxicity study in rats, inhalation exposure up to 750 mg/m3 did not show adverse effects on reproductive organs or embryo or fetal development (OECD 2003; EC 2005; Saillenfait et al. 1999a). The developmental study established a NOAEC of 563 mg/m3 (equivalent internally to 175 mg/kg bw per day) for maternal toxicity on the basis of the critical effect of slightly reduced food intake and lower maternal body weight gain at the high dose of 750 mg/m3.
In a dermal lifetime study in male mice only, a NO(A)EL of 1,081 mg/kg bw per day, the highest dose tested, was established on the basis of no systemic toxicity observed (Wenzel-Hartung 1989). In a 90-day whole body inhalation study in both male and female rats, a NOAEC of 225 mg/m3 (equivalent internally to 70 mg/kg bw per day) was established on the basis of the critical effects of elevated alanine transaminase and alkaline phosphatase in females at 750 mg/m3 and higher (BASF 1989, as cited in EC 2005).
Butyl acrylate
Butyl acrylate was not carcinogenic to rats via inhalation exposure up to 773 mg/m3, the highest dose tested, and showed no genotoxic effects in in vitro and in vivo assays (OECD 2002b). No reproductive studies are available, but a 90-day inhalation study for butyl acrylate did not show adverse effects to reproductive organs of rats (seminal vesicles, prostate, epididymis, uterus, testes or ovaries) at doses associated with mortality (BASF AG 1978, as cited in OECD 2002b). Repeat-dose studies by the oral and inhalation routes did not result in systemic toxicity (OECD 2002). A 90-day drinking water study in rats established a NOAEL of 111 mg/kg bw per day on the basis of no systemic toxicity observed at the highest dose and a NOAEL of 150 mg/kg bw per day on the basis of lack of systemic toxicity in a satellite group by gavage (Gorzinski 1982, as cited in OECD 2002b).
A developmental inhalation study in rats established a NOAEC of 130 mg/m3 on the basis of the critical effect of significant reduction of maternal body weight gain (gestational days 6 to 16, but comparable to controls at end of exposure period) and post-implantation loss at 720 mg/m3 (BASF AG 1979, as cited in OECD 2002b). Additional developmental studies established a NOAEL for maternal toxicity of 100 mg/kg bw per day in mice for gavage administration on the basis of mortality (1/30) at 1000 mg/kg bw per day. Maternal and fetal body weight gain was reduced at 1500 mg/kg bw per day and higher and increased number of resorptions and malformations was observed at 2500 mg/kg bw day and higher, with a NOAEL for developmental toxicity of 1000 mg/kg bw per day established (Rohm and Haas Co. 1982, as cited in OECD 2002b). An inhalation developmental study in rats established a Lowest Observed Adverse Effect Level (LOAEC) of 530 mg/m3 on the basis of significant reduction in absolute maternal body weight gain observed at all doses. Decreased fetal body weight was observed at the mid and high dose groups (Saillenfait 1999a, as cited in OECD 2002b). At concentrations where maternal toxicity was not observed, butyl acrylate did not cause developmental toxicity (OECD 2002b).
Methacrylic acid
Carcinogenicity data are not available for methacrylic acid. Data from the structurally related methyl methacrylate did not identify a concern for carcinogenicity or reproductive toxicity of methacrylic acid (EC 2002b, OECD 2001b). Methacrylic acid was negative in a bacterial gene mutation test. On the basis of this result, in conjunction with the lack of in vivo genotoxicity demonstrated for the structurally related methyl methacrylate, the European Commission considered that no further testing was needed (EC 2002b).
Data are not available for methacrylic acid for developmental toxicity but a developmental NOAEC was determined to be the highest dose tested (8436 mg/m3) in the presence of decreased maternal body weight gain in a developmental study with methyl methacrylate (Rohm and Haas 1991; Solomon et al. 1993, as cited in EC 2002b). In a 90-day whole body inhalation study, methacrylic acid administered to rats and mice resulted in nasal irritation and corrosion and a NOAEC of 1071 mg/m3 in rats at the highest dose tested and a NOAEC of 357 mg/m3 in mice (equivalent internally to 475 mg/kg bw per day) on the basis of the critical effect of reduced body weight gain at the high dose of 1071 mg/m3 which was also observed in the day 5 sacrifice group (CIIT 1984, as cited in EC 2002b). Data are not available for oral or dermal routes of exposure.
n-Butyl methacrylate
n-Butyl methacrylate was assessed by the OECD (2004a) as part of the assessment of the short-chain alkyl methacrylates, which show structure activity relationship with respect to mammalian toxicity and are rapidly metabolized to methacrylic acid and their corresponding alcohol, with methyl methacrylate used as a reference chemical. Carcinogenicity data are not available for n-butyl methacrylate. However, data from the structurally similar methyl methacrylate did not identify a concern for carcinogenicity (OECD 2004b). In vivo and in vitro assays show that n-butyl methacrylate is not genotoxic (OECD 2004b).
An inhalation developmental study in rats derived a NOAEC for developmental toxicity of 1773 mg/m3 on the basis of decreased fetal body weight at 3546 mg/m3 in the presence of maternal toxicity (Saillenfait 1999b, as cited in OECD 2004b). Repeat-dose studies for dermal route of exposure are not available for any of the short-chain alkyl methacrylates substance group. A 28-day inhalation study established a NOAEC of 1832 mg/m3 on the basis of lacrimation, eye squinting, laboured breathing and localized bilateral degeneration of the olfactory epithelium of the nasal cavity at 1744 mg/kg (Hagan et al. 1993, as cited in OECD 2004b). A developmental inhalation study in rats established a NOAEL for maternal toxicity of 591 mg/m3 (equivalent internally to 183 mg/kg bw per day) on the basis of the critical effect of decreased maternal body weight gain (gestational days 6-13) at 1773 mg/m3 (550 mg/kg bw per day) with the NOAEL of 3546 mg/m3 for developmental toxicity (Saillenfait 1999b, as cited in OECD 2004b).
A combined repeat-dose study with reproductive and developmental screening conducted by Ito et al. (1998) exposed rats to n-butyl methacrylate in sesame oil via gavage. In males exposed at 100 mg/kg bw per day and higher and in females exposed at 1000 mg/kg bw per day (highest dose tested), absolute and relative spleen weights were decreased with histopathological examination showing atrophy of the splenic red pulp. At 1000 mg/kg bw per day, relative kidney weights were increased as well as ketone bodies and occult blood in urine, prothrombin time and urea nitrogen in blood in males. A NO(A)EL of 30 mg/kg bw per day was established for repeat-dose toxicity in males, while a NO(A)EL of 300 mg/kg bw per day was established for repeat-dose toxicity in females and for developmental toxicity.
Isobornyl methacrylate
Data are not available for carcinogenicity for isobornyl methacrylate. Isobornyl methacrylate is not considered genotoxic under in vitro conditions and no in vivo studies were identified (OECD 2011). Data are not available for dermal or inhalation routes of exposure. Oral toxicity data for isobornyl methacrylate was used in the absence of dermal and inhalation toxicity data.
A combined reproductive developmental study administered isobornyl methacrylate by gavage in corn oil to rats and found no effects on developmental or reproductive parameters with effects limited to the liver and kidney. A NOAEL of 25 mg/kg bw per day for parental systemic toxicity was established on the basis of the critical effects of microscopic changes in the liver (biliary proliferation/hypertrophy associated with fibrosis and macrophages infiltration) and kidney (acidophilic globules in the cortical tubular epithelium) at the mid dose (100 mg/kg bw per day) and higher. At the high dose (500 mg/kg bw per day), statistically significant increases in liver weights in males and females were observed with disorganization of hepatic cords and necrosis in the parenchyma, as well as statistically significant increases in kidney weights in males (OECD 2011). In a 90-day dietary study in rats administered isobornyl methacrylate, a NOAEL was not established due to histopathological changes in the liver (biliary epithelial hyperplasia, bile duct hyperplasia) and kidney (hypertrophy of deep proximal convoluted tubules) noted at 50 mg/kg bw per day and above, as well as increased relative liver, kidney and testes weights at the high dose of 500 mg/kg bw per day. In a sub-chronic dietary study in dogs, a NOAEL of 95 mg/kg bw per day was derived on the basis of slightly increased blood urea nitrogen, increased relative liver weight, and slight degenerative changes in the epithelial cells of the kidney proximal convoluted tubules at 352 mg/kg bw per day.
6.3 Characterization of risk to human health
Exposures from environmental media and food are expected to be low or negligible, and risk is therefore considered to be low. The predominant source of exposure to the general population is expected to occur mainly from products available to consumers.
On the basis of the available information, substances in the Acrylates and Methacrylates Group are not considered genotoxic or carcinogenic. Characterization of risk to human health is on the basis of non-cancer effects.
The margins of exposure (MOEs) ranged from 94 to 329 for acrylic acid, 438 to greater than 18,000 for 2-ethylhexyl acrylate, 300 to 2,216 for butyl acrylate, 116 to 491 for methacrylic acid, 653 to 1,441 for n-butyl methacrylate, and 202 to 246 for isobornyl methacrylate. These MOEs are considered adequate to address uncertainties in the health effects and exposure databases (Appendix A).
6.4 Uncertainties in evaluation of risk to human health
Although there are some uncertainties in the health effects database (e.g., incomplete health effects database including lack of route- and duration-specific data) and some limitations in the exposure database (e.g., limited dermal absorption data), conservative approaches to characterizing exposure were taken, and the achieved margins of exposure are considered adequate to address these uncertainties.
7. Conclusion
Considering all available lines of evidence presented in this screening assessment, there is low risk of harm to the environment from acrylic acid, 2-ethylhexyl acrylate, butyl acrylate, methacrylic acid, n-butyl methacrylate, and isobornyl methacrylate. It is concluded that acrylic acid, 2-ethylhexyl acrylate, butyl acrylate, methacrylic acid, n-butyl methacrylate, and isobornyl methacrylate do not meet the criteria under paragraphs 64(a) or (b) of CEPA as they are not entering the environment in a quantity or concentration or under conditions that have or may have an immediate or long-term harmful effect on the environment or its biological diversity or that constitute or may constitute a danger to the environment on which life depends.
On the basis of the information presented in this screening assessment, it is concluded that acrylic acid, 2-ethylhexyl acrylate, butyl acrylate, methacrylic acid, n-butyl methacrylate, and isobornyl methacrylate do not meet the criteria under paragraph 64(c) of CEPA as they are not entering the environment in a quantity or concentration or under conditions that constitute or may constitute a danger in Canada to human life or health.
Therefore, it is concluded that acrylic acid, 2-ethylhexyl acrylate, butyl acrylate, methacrylic acid, n-butyl methacrylate, and isobornyl methacrylate do not meet any of the criteria set out in section 64 of CEPA.
References
Arkema. 2012a. Global Product Strategy Safety Summary. 2-Ethylhexyl Acrylate [PDF]. [accessed 2016 Nov 25].
Arkema. 2012b. Global Product Strategy Safety Summary. n-Butyl Acrylate [PDF]. [accessed 2016 Nov 25].
BAMM. 1990. Letter of December 14, 1990, with Final Report.
BAMM. 1991. Bushy Run Research Center. Unpublished report, October 4, 1991.
BASF AG. 1978. Report on the study of the subacute toxicity of n-butyl acrylate in the 13-week inhalation study on Sprague-Dawley rats, Department of Toxicology. Unpublished study (XXVI/352), May 30, 1978.
BASF AG. 1979. n-Butyl Acrylate: Prenatal inhalation toxicity in the rat. Department of Toxicology. Unpublished study (78/638), July 30, 1979.
BASF AG. 1989. Abteilung Toxikologie. Unpublished report (82/380), March 30, 1989.
Betts CJ, Dearman RJ, Heylings JR, Kimber I, Basketter DA. 2006. Skin sensitization potency of methyl methacrylate in the local lymph node assay: comparisons with guinea-pig data and human experience. Contact Dermatitis. 55:140-147.
Canada.1978. Food and Drug Regulations. C.R.C., c.870.
Canada. 1999. Canadian Environmental Protection Act, 1999. S.C. 1999, c.33. Canada Gazette Part III, vol. 22, no. 3.
Chang RK, Peng Y, Trivedi N, Johnson JR. 2016. Pharmaceutical Excipients: Polymethacrylates. [accessed 2017 Jan 9].
ChemCAN [level III fugacity model of 24 regions of Canada]. 2003. Version 6.00. Peterborough (ON): Trent University, Canadian Centre for Environmental Modelling and Chemistry.
[CIIT] Chemical Industry Institute of Toxicology. 1984. 90-Day vapour inhalation toxicity study of methacrylic acid in B6C3F1 mice, Sprague-Dawley rats and Fischer-344 rats. Toxicogenics study No. 420-1086.
[ConsExpo] Consumer Exposure Model. 2006. Version 4.1. Bilthoven (NL): Rijksinstituut voor Volksgezondheid en Milieu [National Institute for Public Health and the Environment].
[Danish EPA] Danish Environmental Protection Agency. 2008. Hansen PL, Tønning K, Malmgren-Hansen, B. 2008. Survey and health assessment of chemical substances in hobby products for children. Danish Ministry of the Environment, Environmental Protection Agency (Danish EPA). Survey of Chemical Substances in Consumer Products, No. 93.
[Dow] The Dow Chemical Company. 2014. Product Safety Assessment 2-Ethylhexyl Acrylate [PDF]. [accessed 2016 Nov 25].
[DPD] Drug Product Database [database]. [modified 2015 Jul 17]. Ottawa (ON): Government of Canada. [accessed 2016 Nov 25].
[EC] European Commission. 2002a. European Union Risk Assessment Report: acrylic acid: CAS No. 79-10-7. Luxembourg: Office for Official Publications of the European Communities. Report No.: EUR 19836. [accessed 2016 Nov 25].
[EC] European Commission. 2002b. European Union Risk Assessment Report: methacrylic acid: CAS No. 79-41-4. Luxembourg: Office for Official Publications of the European Communities. Report No.: EUR 19837. [accessed 2016 Nov 25].
[EC] European Commission. 2005. European Union Risk Assessment Report: 2-ethylhexyl acrylate: CAS No. 103-11-7. Luxembourg: Office for Official Publications of the European Communities. Report No.: EUR 21641. [accessed 2016 Nov 25].
[ECCC] Environment and Climate Change Canada. 2016a. Science approach document: ecological risk classification of organic substances.. Ottawa (ON): Government of Canada.
[ECCC] Environment and Climate Change Canada. 2016b. Data used to create substance-specific hazard and exposure profiles and assign risk classifications in the Ecological Risk Classification of organic substances. Gatineau (QC). Available from: substances@ec.gc.ca.
[ECCC, HC] Environment and Climate Change Canada, Health Canada. [modified 2017 Mar 12]. Categorization. Ottawa (ON): Government of Canada. [accessed 2016 Nov 25].
[ECCC, HC] Environment and Climate Change Canada, Health Canada. 2017a. Rapid screening of substances with limited general population exposure. Ottawa (ON): Government of Canada.
[ECCC, HC] Environment and Climate Change Canada, Health Canada. 2017b Draft screening assessment: substances identified as being of low concern using the ecological risk classification of organic substances and the threshold of toxicological concern (TTC)-based approach for certain substances. Ottawa (ON): Government of Canada.
Environment Canada. 2013. DSL Inventory Update data collected under the Canadian Environmental Protection Act, 1999, section 71: Notice with respect to certain substances on the Domestic Substances List. Data prepared by: Environment Canada, Health Canada; Existing Substances Program.
[EPI Suite] Estimation Program Interface Suite for Microsoft Windows [estimation model]. c2000-2012. Ver. 4.11. Washington (DC): US Environmental Protection Agency, Office of Pollution Prevention and Toxics; Syracuse (NY): Syracuse Research Corporation.
Ficheux AS, Morriset T, Chevillotte G, Postic C, Roudot AC. 2014. Probabilistic assessment of exposure to nail cosmetics in French consumers. Food Chem Toxicol. 66:36-43
Hagan JV, Bernacki HJ, Lomax LG. 1993. Butyl methacrylate: four-week vapor inhalation toxicity study in rats. Report No. 92R-143. Unpublished report. Spring House (PA): Toxicology Department Rohm and Haas Company.
Health Canada. 1994. Human Health Risk Assessment for Priority Substances. Ottawa (ON): Minister of Supply and Services Canada. Cat. No. En40-215/41E. [accessed 2016 Nov 25].
Health Canada. 1998. Exposure factors for assessing total daily intake of priority substances by the general population of Canada. Unpublished report. Ottawa (ON): Government of Canada.
Health Canada. [modified 2015 Dec 14]. Cosmetic Ingredient Hotlist: The List of Prohibited and Restricted Cosmetic Ingredients. Ottawa (ON): Government of Canada, Consumer Product Safety Directorate. [accessed 2016 Nov 25].
Health Canada. 2016. Science approach document: threshold of toxicological concern (TTC)-based approach for certain substances. Ottawa (ON): Government of Canada.
Hellwig J, Gembardt C, Murphy SR. 1997. Acrylic acid: Two-generation reproduction toxicity study in Wistar rats with continuous administration in the drinking water. Food Chem Toxicol. 35:859-868.
[IARC] IARC Working Group on the Evaluation of Carcinogenic Risks to Humans [PDF]. 1979. Some Monomers, Plastics and Synthetic Elastomers, and Acrolein.
Ito Y, Yamamoto Y, Hoshi S, Kawamura M, Ito M, Shimodaira Y. 1998. Combined repeat dose and reproductive/developmental toxicity screening test of butyl methacrylate by oral administration in rats. Research Institute for Animal Science in Biochemistry and Toxicology. Study on behalf of Ministry of Health and Welfare, Japan.
[LNHPD] Licensed Natural Health Products Database [database]. [modified 2018 Feb 6]. Ottawa (ON): Government of Canada. [accessed 2016 Nov 25].
Loretz LG, Api AM, Barraj LM, Burdick J, Dressler WE, Gettings SD, Han Hsu H, Pan YHL, Re TA, Renskers KJ, Rothenstein A, Scrafford CG, Sewall C. 2005. Exposure data for cosmetic products: lipstick, body lotion, and face cream. Food Chem Toxicol. 43:279-291.
Loretz LG, Api AM, Babcock L, Barraj LM, Burdick J, Cater KC, Jarrett G, Mann S, Pan YHL, Re TA, Renskers KJ, Scrafford CG. 2008. Exposure data for cosmetic products: facial cleanser, hair conditioner, and eye shadow. Food Chem Toxicol. 46:1516-1524.
Merck Index. 2006. The Merck index: an encyclopedia of chemicals, drugs, and biologicals. Whitehouse Station (NJ): Merck Research Laboratories. [accessed 2016 Nov 25].
[MSDS] Material Safety Data Sheet. 2009. Moxie Girlz Art-titude – Marker Ink (Blue). Van Nuys (CA): MGA Entertainment, Inc. [accessed 2016 Nov 25].
[MSDS] Material Safety Data Sheet. 2012. Bullseye Windshield Repair Kit [PDF]. Halton Hills (ON): ITW Permatex Canada. [accessed 2016 Nov 25].
[MSDS] Material Safety Data Sheet. 2014a. PC Perma Poxy 5 Minute Plastic Weld [PDF]. Halton Hills (ON): ITW Permatex Canada. [accessed 2018 Feb 20].
[MSDS] Material Safety Data Sheet. 2014b. Water Marker. Tara Toy. [accessed 2016 Nov 25].
Neeper-Bradley T, Fowler E, Pritts I, Tyler T. 1997. Developmental toxicity study of inhaled acrylic acid in New Zealand White rabbits. Food Chem Toxicol. 35:869-880
[NHPID] Natural Health Products Ingredients Database [database]. [modified 2018 Feb 21]. Ottawa (ON): Government of Canada. [accessed 2016 Nov 25].
[NPRI] National Pollutant Release Inventory [database]. 2016. Ottawa (ON): Government of Canada. [accessed 2017 Mar 8].
[OECD] Organisation for Economic Co-operation and Development. 2001a. SIDS initial assessment report: 2-Propenoic acid (Acrylic acid): CAS No. 79-10-7. SIAM [SIDS Initial Assessment Meeting] 13, 2001 November 6-9. [accessed 2016 Nov 11].
[OECD] Organisation for Economic Co-operation and Development. 2001b. SIDS initial assessment report: Methacrylic acid: CAS No. 79-41-4; 2001. United Nations Environment Programme (UNEP). [accessed 2016 Apr 25].
[OECD] Organisation for Economic Co-operation and Development. 2002a. Addendum to SIDS initial assessment report: n-Butyl Acrylate: CAS No. 141-32-2. SIAM [SIDS Initial Assessment Meeting] 15, 2002 October 22-25. [accessed 2016 May 9].
[OECD] Organisation for Economic Co-operation and Development. 2002b. SIDS initial assessment report: n-Butyl Acrylate: CAS No. 141-32-2. SIAM [SIDS Initial Assessment Meeting] 15, 2002 October 22-25. [accessed 2016 May 9].
[OECD] Organisation for Economic Co-operation and Development. 2003. SIDS initial assessment report: 2-Ethylhexylacrylate: CAS No. 103-11-7. SIAM [SIDS Initial Assessment Meeting] 16, 2003 May 27-30. [accessed 2016 May 9].
[OECD] Organisation for Economic Co-operation and Development. 2004a. SIDS initial assessment profile: Ethyl Methacrylate, Iso-Butyl Methacrylate, n-Butyl Methacrylate, 2-Ethylhexyl Methacrylate: CAS No. 97-63-2, 97-86-9, 97-88-1, 688-84-6. SIAM [SIDS Initial Assessment Meeting] 18, 2004 April 20-23. [accessed 2016 Jun 2].
[OECD] Organisation for Economic Co-operation and Development. 2004b. SIDS initial assessment report: Short Chain Alkyl Methacrylates: Ethyl Methacrylate, Iso-Butyl Methacrylate, n-Butyl Methacrylate, 2-Ethylhexyl Methacrylate: CAS No. 97-63-2, 97-86-9, 97-88-1, 688-84-6. SIAM [SIDS Initial Assessment Meeting] 18, 2004 April 20-23. [accessed 2016 Jun 2].
[OECD] Organisation for Economic Co-operation and Development. 2011. SIDS initial assessment report: Isobornyl Methacrylate (IBOMA): CAS No. 7534-94-3. SIAM [SIDS Initial Assessment Meeting] 32, 2011 April 19-21. [accessed 2016 May 11].
[RIVM] Rijksinstituut voor Volksgezondheid en Milieu [National Institute for Public Health and the Environment]. 2006. Cosmetics fact sheet: to assess the risks for the consumer: updated version for ConsExpo 4 [PDF]. Bilthoven (NL): RIVM. Report No.: 320104001/2006.
[RIVM] Rijksinstituut voor Volksgezondheid en Milieu [National Institute for Public Health and the Environment]. 2007. Do-it-yourself products fact sheet: to assess the risks for the consumer: updated version for ConsExpo 4 [PDF]. Bilthoven (NL): RIVM. Report No.: 320104001/2006.
Rohm & Haas Co. 1982. Teratological evaluation of n-Butyl Acrylate in CD-1 Mice. Research Triangle Institute, Contract No. N01-ES-6-2127, Sept. 13, 1982.
Saillenfait AM, Bonnet P, Gallissot F, Protois JC, Peltier A, Fabries JF. 1999a. Relative developmental toxicities of acrylates in rats following inhalation exposure. Toxicol Sci. 48:240-254.
Saillenfait, AM, Bonnet P, Gallissot F, Peltier A, Fabries JF. 1999b. Developmental toxicities of methacrylic acid, ethyl methacrylate, n-butyl methacrylate, and allyl methacrylate in rats following inhalation exposure. Toxicol Sci. 50:136-145.
[SDA] Soap and Detergent Association. 2005. Exposure and Risk Screening Methods for Consumer Product Ingredients [PDF]. [accessed 2016 Nov 25].
TSCATS. 1992. OTS 0510541-2, Old I.D. 8EHQ-0692-0592, New Doc I.D. 89-920000108, Bushy Run Research Center, Supplement.
Wenzel-Hartung RP, Brune H, Klimish HJ. 1989. Dermal oncogenicity study of 2-ethylhexyl acrylate by epicutaneous application in male C3H/HeJ mice. J Cancer Res Clin Oncol. 115(6):543-549.
Appendix A. Estimated margins of exposure
Presented below are all relevant exposure and hazard values for the Acrylates and Methacrylates Group, as well as the resultant margins of exposure (MOE) for determination of risk. Dermal absorption is conservatively assumed to be 100%, except for isobornyl methacrylate, for which dermal absorption was 15%.
Exposure scenario |
Estimated exposure |
Critical effect level |
Critical effect level |
MOE |
|---|---|---|---|---|
Water marker - toddler (per event, oral) |
0.16 mg/kg bw |
NOAEL (oral) = 53 mg/kg bw per day |
Decreased body weight gain in F1 generation |
329 |
Face moisturizer (daily, dermal) |
0.91 mg/kg bw per day |
NOAEL (oral) = 240 mg/kg bw per day |
Decreased food and drinking water in consumption F0 females |
264 |
Nail gel manicure preparation (per event, inhalation) |
0.78 mg/m3 |
NOAEC (inhalation) = 73 mg/m3 |
Decreased body weight gain, decreased food consumption, perinasal and perioral wetness, nasal congestion |
94 |
Exposure scenario |
Estimated exposure |
Critical effect level |
Critical effect level |
MOE |
|---|---|---|---|---|
Facial cleanser (daily, dermal) |
0.059 mg/kg bw per day |
NOAEL (inhalation) = 70 mg/kg bw per daya NO(A)EL (dermal) = 1,081 mg/kg bw per day |
Elevated alanine transaminase and alkaline phosphatase in females No systemic toxicity at highest dose |
1186 to >18000 |
Press on manicure adhesive (per event, dermal) |
0.40 mg/kg bw |
NOAEL (inhalation) = 175 mg/kg bw per daya NO(A)EL (dermal) = 1,081 mg/kg bw per day |
Maternal toxicity slight decrease food intake and body weight gain No systemic toxicity at highest dose |
438 to 2737 |
Press on manicure adhesive (per event, inhalation) |
0.55 mg/m3 |
NOAEC = 563 mg/m3 |
Maternal toxicity slight decrease food intake and body weight gain |
1024 |
a 1 mg/m3 in air is equal to 0.31 mg/kg bw per day (rat)
Exposure scenario |
Estimated exposure |
Critical effect level |
Critical effect level |
MOE |
|---|---|---|---|---|
Nail Polish (per event, dermal) |
0.068 mg/kg bw |
NOAEL(gavage) = 150 mg/kg bw per day |
No systemic toxicity |
2216 |
Nail Polish (per event, inhalation) |
0.44 mg/m3 |
NOAEC (inhalation) = 130 mg/m3 |
Deceased maternal body weight gain |
299 |
Exposure scenario |
Exposure estimate |
Critical Effect Level |
Critical effect level |
MOE |
|---|---|---|---|---|
Marker ink -toddler (per event, oral) |
0.97 mg/kg bw |
NOAEC (inhalation) = 475 mg/kg bw per daya |
Decreased body weight gain (mice) |
491 |
Marker ink -toddler (per event, dermal) |
0.97 mg/kg bw |
NOAEC (inhalation) = 475 mg/kg bw per daya |
Decreased body weight gain (mice) |
491 |
Epoxy adhesive (per event, inhalation) |
3.1 mg/m3 |
NOAEC (inhalation mice) = 357 mg/m3 |
Decreased body weight gain |
116 |
a 1 mg/m3 in air is equal to 1.33 mg/kg bw per day (mouse)
Exposure scenario |
Estimated exposure |
Critical effect level |
Critical effect level |
MOE |
|---|---|---|---|---|
Adhesive in nails (per event, dermal) |
0.28 mg/kg bw |
NOAEL (inhalation) = 183 mg/kg bw per daya |
Decreased maternal body weight gain |
653 |
Adhesive in nails (per event, inhalation) |
0.41 mg/m3 |
NOAEC (inhalation) = 591 mg/m3 |
Decreased maternal body weight gain |
1441 |
a 1 mg/m3 in air is equal to 0.31 mg/kg bw per day (rat)
Exposure scenario |
Systemic exposure |
Critical effect level |
Critical effect level |
MOE |
|---|---|---|---|---|
Nail polish (per event, dermal) |
0.10 mg/kg bw (15% dermal absorption applied) |
NOAEL (gavage) = 25 mg/kg bw per day |
Hypersalivation, microscopic finding of biliary proliferation /hypertrophy associated with fibrosis and macrophage infiltration; acidophilic globules in the cortical tubular epithelium of the kidney |
246 |
Nail polish (per event, inhalation) |
0.12 mg/kg bw (equivalent to 0.54 mg/m3) |
NOAEL (gavage) = 25 mg/kg bw per day |
Hypersalivation, microscopic finding of biliary proliferation /hypertrophy associated with fibrosis and macrophage infiltration; acidophilic globules in the cortical tubular epithelium of the kidney at 100 mg/kg bw |
202 |
Appendix B. Estimated human exposures to acrylates and methacrylates
Exposures were estimated using ConsExpo version 4.1 or algorithms from the model (RIVM 2006, 2007). Molecular weight and vapour pressure values were incorporated into the calculations (EpiSuite c2000-2012).
The cosmetic and do-it-yourself product scenarios were calculated on the basis of the default body weight (70.9 kg) and inhalation rate (16.2 m3/day) of an adult (20 to 59 years old) (Health Canada 1998), and the applicable use behaviours of an adult. The nail product scenarios that assumed products are used on both fingernails and toenails. Product amounts for inhalation scenarios are on the basis of mean amounts of products used, and product amounts for dermal scenarios are on the basis of amount on skin (Ficheux et al. 2014). The estimated dermal and inhalation exposure parameters for nail scenarios are described in Table B-1. Dermal adult exposure parameter assumptions for other cosmetics are described in Table B-2. Dermal absorption is conservatively assumed to be 100%, except for isobornyl methacrylate, for which dermal absorption was assumed to be 15%. Inhalation exposure for the epoxy adhesive is described in Table B-3.
Substance - Product |
Route |
Product amount (gram) |
Exposure and application duration (minute) |
2-Ethylhexyl acrylate – Press on manicure adhesive; n-Butyl methacrylate – Adhesive used in nails |
Dermal |
0.04 |
N/A |
Butyl acrylate – Nail polish; Isobornyl methacrylate – Nail polish |
Dermal |
0.16 |
N/A |
Acrylic acid – Nail gel manicure preparation; 2-Ethylhexyl acrylate – Press on manicure adhesive; n-Butyl methacrylate – Adhesive used in nails |
Inhalation |
0.18 |
7 |
Butyl acrylate – Nail polish; Isobornyl methacrylate – Nail polish |
Inhalation |
0.8 |
35 |
a Ventilation rate = 1/hr, room volume = 1 m3, molecular weight matrix = 124 g/mol, mass transfer rate = Langmuir’s method, release area for inhalation = 26.2 cm2, uptake fraction = 1.
Abbreviation: N/A, not applicable.
Substance - Product |
Product amount (gram) |
Retention factor |
Frequency (application per day) |
Acrylic acid – Face moisturizer |
1.2 |
1a |
1.8 |
2-Ethylhexyl acrylate – Facial cleanser |
2.58 |
0.01 |
1.6 |
Butyl acrylate – Adhesive for eyes b |
0.009 |
1a |
1.2 |
n-Butyl methacrylate – Hair grooming gel |
1.9 |
0.1c |
0.55 |
a Retention factor of 1 was used because products may not be washed off
b Utilized values from eye shadow scenario as conservative estimate of exposure
c Assumed a transfer factor of 0.1 from hair to scalp and no rinse-off (rinse-off factor = 1)
Substance - Product |
Parameters |
Methacrylic acid – Epoxy adhesive application a |
Applied amount: 20 gram Ventilation rate: 0.6 1/hr Uptake fraction = 1 Mass transfer rate = Thibodeaux’s method Molecular weight matrix = 3000 g/mol Exposure duration: 240 minute Release area: 500 cm2 Application duration: 10 minute Room volume: 20 m3 (RIVM 2007) |
Methacrylic acid – Epoxy adhesive mixing and loading a |
Applied amount: 20 gram Ventilation rate: 0.6 1/hr Uptake fraction = 1 Mass transfer rate = Thibodeaux’s method Molecular weight matrix = 3000 g/mol Exposure duration: 5 minute Release area: 20 cm2 Application duration: 5 minute Room volume: 1 m3 (RIVM 2007) |
a Concentration of up to 10% in one of two components
Oral and dermal exposures for markers in toys and children’s products were estimated on the basis of the default body weight, i.e., 15.5 kg, of a toddler (6 months to 4 years old) (Health Canada 1998) and the use behaviours of a toddler. For the per event exposure calculations, the estimated amount of ink per exposure is 50 mg (Danish EPA 2008). The fraction absorbed is assumed to be 1. For the daily exposure calculations, the ink laydown rate of 100 µg/cm and 25 cm of ink line per day is assumed (personal communication from the Art & Creative Materials Institute (ACMI), Duke University, to Health Canada, 2009; unreferenced). Hand-to-mouth and object-to-mouth exposures are covered in the estimate of daily exposure.