Sea ice cycle: chapter 2
Growth
Once a sheet of ice has formed, it can increase in thickness by the freezing of water on its lower surface. This means that heat must be removed from the water.
When the air above the ice is colder than the water below the ice, heat is removed by conduction through the ice from the water to the air above. Thus, heat flows from the warm body (water) to the cold body (air). Scientists have discovered that the rate at which heat flows from the water is proportional to the temperature difference between air and water, and also inversely proportional to the thickness of the ice layer and the snow cover on top of the ice.
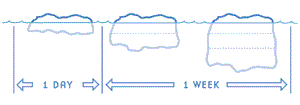
With very cold air temperatures, a thin sheet of ice will thicken quickly at first, perhaps 8 to 10 centimetres (3 to 4 inches) in the first 24 hours. The rate of growth gradually slows down as the ice thickens.
Besides growing on the bottom of the surface of an ice sheet, ice can also grow in-between the ice floes joining them together or by accumulation of slush, ice cakes, or ice floes on the windward shore.
Snow Cover
Snow cover acts as a blanket, slowing down the flow of heat from the water. The effectiveness of snow as an insulator, or blanket, depends mainly on how compacted it is. A layer of soft, fluffy, new fallen snow is an excellent insulator because of its high air content. On the other hand, extremely hard, compacted snow is a relatively poor insulator.
The table below gives the heat insulating qualities of various types of snow in terms of the thickness of ice that would have the same insulating effect.
| 5 cm of various snow types: | Equivalent to an ice thickness of: |
|---|---|
| Newly fallen soft snow | 264 - 381 cm |
| Slightly settled snow | 175 - 193 cm |
| Normal snow | 61 - 97 cm |
| Old snow | 41 - 61 cm |
| Hard packed snow | 24 - 31 cm |
| Extremely cold, wind swept snow | 19 - 24 cm |
It is, therefore apparent that a few centimetres of snow, especially loosely packed snow, on top of ice, will drastically slow down the rate of ice growth.
Structure of Sea Ice
When the temperature of surface water reaches the freezing point, tiny needle-like crystals or plates- about 10 mm across-of pure ice form. It is the continuous accumulation of those crystals on the surface that gives the water its oily appearance, and this soon results in a thin layer of slush ice.
Brine cells
As soon as the crystals form, the water in their immediate vicinity becomes a little saltier because the salt in the solution does not become part of the initial crystals. This saltier water, being denser than before the crystals formed, immediately tends to sink. As more and more crystals form, they will eventually become frozen to one another in such a way that tiny spaces or pockets will remain between groups of crystals. Some of the residual brine will become trapped in those pockets, which are known as brine cells.
If ice crystals form slowly on the water surface, most of the remaining brine is able to diffuse downward into the water underneath. Thus, when crystals freeze together, not as much brine is trapped and the ice will have lower salt content. However, when crystals freeze rapidly together, more brine is trapped in the brine cells before it can sink and mix with the water below. Hence, ice which forms rapidly is initially saltier than ice which forms more slowly.
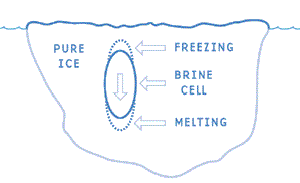
Some of the brine which becomes trapped in cells in the ice remains liquid at very low temperatures. As the temperature within the ice continues to fall, water is removed from the brine in the cells by the formation of ice crystals. Hence, the remaining brine becomes still saltier and the temperature must be lowered further before more ice crystals will form.