Caribou (Rangifer tarandus) COSEWIC assessment and status report 2014: chapter 2
Section 2
Wildlife Species Description and Significance
Name and Classification
- Scientific name:
- Rangifer tarandus Linnaeus 1758
- Class:
- Mammalia
- Order:
- Artiodactyla
- Family:
- Cervidae
- Subfamily:
- Capreolinae
- Common Names
- Caribou, Woodland Caribou (English); Caribou, Caribou des bois (French); Wah stzee (Dunne-za); Whudzih or wudzįįh (Tsek'ene/Tse Keh Nay/Sakani); Kwun Ba Whut'en (Carrier Sekani); Vadzaih (Gwichin); Ekwe (North Slavey); T'onzi/Tohzi/T'ondzi (North Slavey – woodland caribou); NodÍe (South Slavey - mountain caribou); MedzÍh (South Slavey - woodland caribou).
All caribou and reindeer in the world belong to one species, Rangifer tarandus. In Canada, Banfield (1961) classified caribou into four extant subspecies and one extinct subspecies based primarily on skull measurements and pelage, but also on antler shape and hoof shape. The four extant subspecies included “woodland caribou” (R.t. caribou), “barren-ground caribou” (R.t. groenlandicus), “Grant’s caribou” (R.t. granti) and “Peary caribou” (R.t. pearyi). “Dawson’s caribou” (R.t. dawsoni) were found on Haida Gwaii off the coast of BC before their extinction sometime during the early 1900s (Spalding 2000). Banfield’s (1961) subspecies classification is still the most commonly used taxonomy, largely because no alternative has been identified in a systematic way or has ever been broadly accepted. Caribou in western mountain regions of North America are included in Banfield’s (1961) “woodland” subspecies but have also been further categorized into ecotypes based on ecological conditions and adaptations. Ecotype names differ between jurisdictions, which leads to some confusion (see COSEWIC 2011). For example, in BC, caribou that live in areas of relatively shallow snowpacks and feed primarily on terrestrial lichens are called ‘northern’ ecotype caribou, while caribou that live in deep snow areas and feed primarily on arboreal lichens are ‘mountain’ ecotype caribou (Stevenson and Hatler 1985, Heard and Vagt 1998). In Alberta, caribou that feed primarily on terrestrial lichens and spend at least part of their annual cycle in the mountains are similar to BC’s ‘northern’ ecotype but are called ‘mountain’ caribou (ASRD&ACA 2010). There is wide recognition that a complete revision of the taxonomic entities within Rangifer is needed (COSEWIC 2011).
Controversy surrounding Banfield’s subspecies classification has been particularly pronounced for Woodland Caribou (R. t. caribou), because this includes caribou that occur in almost every available habitat type, despite exhibiting considerable variability in behaviour, ecology, and morphology (Geist 2007). To avoid problems associated with subspecies classification of caribou, the term “forest-dwelling caribou” will be used in this report to refer to caribou in Canada that remain south of treeline all year; “western mountain caribou” will be the collective term for caribou residing in the western mountains of North America. In addition, the term “population” will refer to caribou at the designatable unit (DU) level and “subpopulation” will be used when referring to individual herds.
Morphological Description
Caribou are mixed dark and lighter brown with a whitish mane and some white on their sides (Figure 1). Mature females and males usually weigh 110-150 kg and 160-210 kg, respectively, and height at the shoulder is 1.0-1.2 m. Some characteristics suggest that Rangifer is an ancient member of the family Cervidae (Banfield 1974). Both sexes can bear antlers although some females may lack antlers or have only one antler. A distinctive characteristic is large, rounded hooves that reduce sinking in snow and wetlands and act as shovels when digging for food under snow. The dew claws are large, widely spaced, and set back on the foot, which greatly increases weight-bearing area and reduces foot loads. Banfield (1961, 1974), Miller (1982), Kelsall (1984), Geist (1991),and Bergerud (2000) described physical features of caribou.

Photo: © Elena Jones
Long description for Figure 1
Photo of a group of Central Mountain caribou browsing among trees on snow-covered ground. These caribou, from the Kennedy Siding subpopulation, are mixed dark and lighter brown with a whitish mane and some white on their sides. The large dew claws of one animal are clearly visible.
From an Aboriginal traditional knowledge (ATK) perspective, “The participants described woodland caribou as being smaller in size than mountain woodland caribou and larger in size than the barren-ground caribou. The elders said that long ago, ‘They did not call the caribou either mountain woodland or boreal woodland.’ ‘They just knew they were different in their markings, colour and hoof prints.’ (Tulita elder)” (Sahtu Renewable Resources Board 2010).
Population Spatial Structure and Variability
Phylogenetic analyses of caribou based on mitochondrial DNA (mtDNA) studies provide information on genetic lineages, and studies of microsatellites on nuclear DNA provide information on genetic diversity and population structuring of forest-dwelling caribou. Available information focuses on caribou in west-central Alberta and east-central BC (Dueck 1998, McDevitt et al. 2009, Serrouya et al. 2012, Klütsch et al. 2012, Weckworth et al. 2012, Yannic et al. 2013), caribou in southeastern BC (Dueck 1998, Zittlau 2004, Serrouya et al. 2012) and caribou in Yukon and NT (Dueck 1998, Zittlau 2004, Kuhn et al. 2010, Weckworth et al. 2012), with limited information on caribou in northern and west-central BC (Zittlau 2004, Serrouya et al. 2012). In many studies, comparisons are made with subpopulations in other caribou DUs (COSEWIC 2011). Recent studies have contributed substantially to the understanding of phylogenetic and genetic structure of caribou in the western mountainous region of Canada, but further work is required to fill information gaps, especially for caribou in northern and west-central BC.
Phylogenetics
Phylogenetic analysis of caribou has revealed two distinct major haplogroups or clades, which suggest the isolation of caribou into two or three separate glacial refugia (Røed et al. 1991; Dueck 1998; McDevitt et al. 2009, COSEWIC 2011, Klütsch et al. 2012, Yannic et al. 2013). Dueck’s (1998) study of mtDNA suggested that all caribou in Canada originated from northern and southern clades (groups) isolated about 49 000 years ago during the Wisconsinan glaciation. Glacial advances occurred in the Canadian Rockies 75,000-64,000 and 20,000-11,000 years ago (Gadd 1986). Two groups of caribou were separated between those early and late Wisconsinan episodes and distinct clades evolved in isolation. After the retreat of the large continental glaciera, the two groups spread out across Canada and their distributions overlapped. Klütsch et al. (2012) suggested that three lineages of forest-dwelling caribou in Canada may have originated from three geographically separate refugia south of the Laurentide ice sheet: one in the Appalachians, one in the Wisconsin area, and one in the Rocky Mountains, with the caribou from the Rocky Mountain refugium spreading into the western mountainous region in Canada. Yannic et al. (2013) found that two phylogeographic lineages have remained genetically homogenous since diverging two interglacial-glacial cycles ago, and secondary contact between them occurred in central Canada about 8,000 years BP.
Caribou phylogeographical structure appears to be chiefly the result of postglacial expansions after the last glacial maximum, which is corroborated by evidence from the fossil record, ecological considerations, and physiogeography (Klütsch et al. 2012, Yannic et al. 2013). Many of the subpopulations situated in both west-central Alberta/east-central BC and southeastern BC contain haplotypes from both northern and southern clades (Dueck 1998, McDevitt et al. 2009, Yannic et al. 2013), indicating possible contact zones. The greatest mix of haplotypes is in the eastern mountains of BC and adjacent Alberta where 0-56% of haplotypes in analyzed samples in east-central BC/west-central Alberta and 1-67% of haplotypes in analyzed samples in southeastern BC are from the northern clade. McDevitt et al. (2009) found a positive association between migratory behaviour of individual caribou and belonging to membership in the northern clade (i.e. individual caribou that migrated were more likely to belong to the northern clade). Present-day forest-dwelling caribou apparently have evolved from both clades and introgression of DNA occurred where the two clades met and overlapped in distribution.
Genetic population structure
Microsatellite data paint a complex picture of genetic population structuring within and among caribou subpopulations in western Canada. Studies were inconsistent in that they each assessed a unique combination of subpopulations, and in terms of the character of loci being used, making it difficult to compare some of the differing results among studies. Figure 2 displays the geography of subpopulations referred to below (see also Designatable Units).
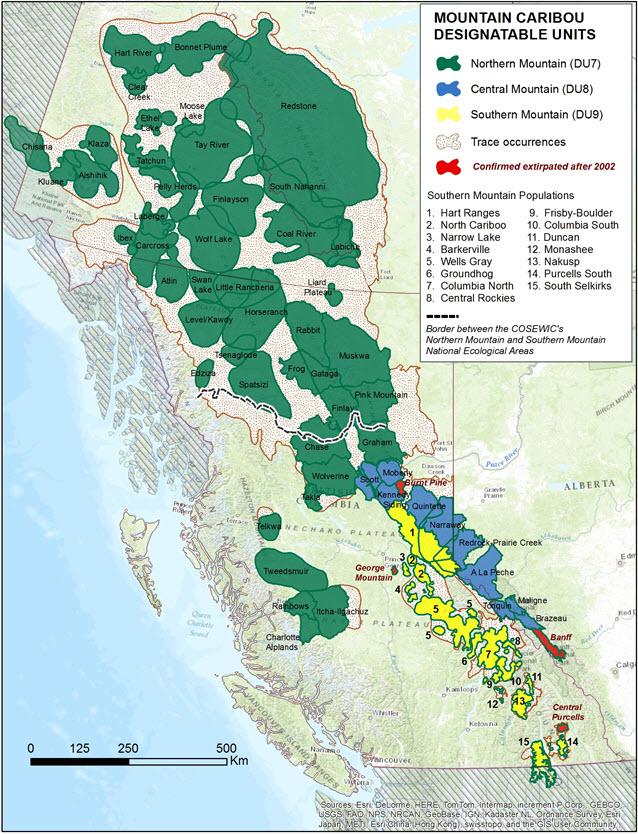
Map: © Bonnie Fournier 2013
Long description for Figure 2
Map of the distribution of subpopulations of western mountain caribou from the Northern Mountain, Central Mountain, and Southern Mountain designatable units. The range of each designatable unit is indicated by colour, with the ranges of subpopulations outlined and named, except for the Southern Mountain designatable unit, where subpopulations are numbered and identified in the legend. Other information includes areas confirmed extirpated after 2002 and trace occurrences. The border between COSEWIC's Northern Mountain and Southern Mountain National Ecological Areas is shown.
Serrouya et al. (2012) found that caribou subpopulations located north of the Peace River formed one distinct cluster. However, the two west-central BC subpopulations sampled (Itcha-Ilgachuz, Tweedsmuir) more closely resembled caribou south of the Peace River. Zittlau (2004) concluded that the two west-central BC subpopulations were more closely related to caribou in southeastern BC than to caribou sampled from northern BC. Of the subpopulations sampled from northern and west-central BC, the Itcha-Ilgachuz subpopulation had the lowest level of microsatellite diversity and was highly distinct (94% self assignment), likely due to its isolation (along with neighbouring Tweedsmuir-Entiako subpopulation) from other subpopulations (Zittlau 2004). The three sampled northern BC subpopulations (Atlin, Carcross, Cassiar) clustered together as did the three north-central BC subpopulations (Wolverine, Finlay, Chase). Zittlau (2004) also detected high microsatellite differentiation among three Yukon/BC subpopulations (Aishihik, Chisana, Wolf Lake) as well as high levels of genetic diversity within each of these subpopulations, which she hypothesized reflected the admixture of subpopulations in unglaciated regions during the last ice age. Kuhn et al. (2010) found four genetic clusters within subpopulations in Yukon. One of those clusters, which included Ibex, Carcross, and Atlin, was genetically distinct.
McDevitt et al. (2009) reported high differentiation that was significant (p<0.05) among subpopulations of caribou from east-central BC/west-central Alberta using mitochondrial data (FST 0.018 – 0.5619) and microsatellite data (FST 0.01-0.09). The A La Peche subpopulation clustered with Jasper and Banff National Park subpopulations, and the Red Rock Prairie Creek and Narraway (including Bearhole/Redwillow) subpopulations formed one cluster. The subpopulations in the northern part of the area (Quintette, Moberly, Burnt Pine, Kennedy Siding) clustered with the only southeastern BC subpopulation sampled (Hart Ranges - Parsnip portion). Conversely, Weckworth et al. (2012) found that the Jasper and Banff subpopulations clustered with one of the two southeastern BC subpopulations sampled (North Cariboo), while the A La Peche subpopulation clustered with other subpopulations from the area. Serrouya et al. (2012) found that the Peace River was an important source of genetic separation. The Graham subpopulation on the north side of the river was distinct from subpopulations to the south of the river. All subpopulations from east-central BC/west-central Alberta were included in the cluster that contained subpopulations south of the Peace River and north of the North Thompson River, except for the Jasper subpopulation, which was not well-classified into any cluster (Serrouya et al. 2012). Boreal caribou did not cluster with caribou from east-central BC/West-central Alberta in all three studies (McDevitt et al. 2009, Serrouya et al. 2012, Weckworth et al. 2012).
Serrouya et al. (2012) conducted the most extensive assessment of microsatellite genotypes of southeastern BC caribou (13 subpopulations). Zittlau (2004), McDevitt et al. (2009), and Weckworth et al. (2012) carried out separate studies, with a secondary focus on southeastern BC subpopulations. Some subpopulations in southeastern BC exhibit pronounced structuring, likely as a result of small populations that have experienced more genetic drift, although no meaningful genetic differentiation was detected between the Hart Ranges and the North Cariboo, nor among the Columbia North, Groundhog, Frisby-Boulder, Nakusp, and Duncan subpopulations (Serrouya et al. 2012). The lack of genetic differentiation for those 5 subpopulations could be due to historical movement between them, prior to a contemporary barrier to gene flow (Serrouya et al. 2012). Major river valleys serve as barriers to gene flow for southeastern BC subpopulations (Serrouya et al. 2012). Both Zittlau (2004) and Serrouya et al. (2012) found the Purcells South subpopulation to be genetically distinct among caribou subpopulations they sampled.
Designatable Units
COSEWIC defined designatable units (DUs) for caribou in Canada (COSEWIC 2011) using COSEWIC (2013) criteria for discreteness and evolutionary significance. Twelve DUs were defined based on currently available information on lines of evidence that included phylogenetics, genetic diversity and structure, morphology, movements, behaviour and life history strategies, and distribution.
In previous COSEWIC assessments (COSEWIC 2002; 2004), caribou in Canada were organized into eight “Nationally Significant Populations”. The subjects of this assessment belonged to two nationally significant populations: Northern Mountain and Southern Mountain (COSEWIC 2002) in the COSEWIC Northern and Southern National Ecological Areas, respectively (Figure 2). Based on more recent information on caribou ecology and genetics, COSEWIC (2011) re-organized these into three DUs: Northern Mountain, Central Mountain and Southern Mountain.
There are two major differences between the new DU structure (COSEWIC 2011) and that of the previous assessment (COSEWIC 2002). One change results from the reclassification of terrestrial lichen feeding/shallow snow caribou that were previously part of the Southern Mountain population. The new Southern Mountain DU, restricted to central and southeastern BC (Figure 2), now includes only the deep snow/arboreal lichen feeding ecotype. In contrast, all shallow snow/terrestrial lichen feeding caribou now belong to either the Central Mountain or Northern Mountain DUs. Subpopulations belonging to the new Central Mountain DU occur along the eastern side of the Rocky Mountains in west-central Alberta and east-central BC with some straddling the provincial border (Figure 2). Representing a division of the previous Southern Mountain 'population', subpopulations in this DU actually have more traits in common with Northern Mountain than Southern Mountain caribou. Central Mountain and Northern Mountain DU caribou share similar winter feeding behaviours and seasonal movement patterns, but they differ phylogenetically and are separated both genetically and geographically by the Peace River (see Population Spatial Structure and Variability). Northern Mountain DU subpopulations occur in the northern mountains of Yukon, the southern NT, and west-central and northern BC (Figure 2). This range extends further south into the Southern Mountain ecological area than did the former Northern Mountain population (COSEWIC 2002).
For all three western mountain caribou DUs, individual subpopulations that have been sampled are generally discrete from one another and adjacent populations, including those recognized as members of other DUs (see COSEWIC 2011). The Southern Mountain DU and Central Mountain DUs are discrete from other neighbouring DUs in that phylogenetically, caribou in those two DUs have both northern (Beringian-Eurasian [BEL]) and southern (North American [NAL]) lineages. Caribou sampled in the Northern Mountain DU all come from the BEL lineage. Caribou belonging to the Southern Mountain DU have a distinct behaviour related to their use of habitats found in steep, mountainous habitats with deep snowfall (with an accumulated snowpack of 2-5 m). These extreme snow conditions have led to a foraging strategy that is unique among cervids, that is, the exclusive reliance on arboreal lichens for 3-4 months of the year (COSEWIC 2011). Southern Mountain DU caribou are, therefore, discrete from Central and Northern Mountain DU caribou based on inherited traits, namely behavioural strategies and distributional patterns that have resulted from the steep terrain and high levels of snow accumulation (COSEWIC 2011).
With respect to significance, members of the Southern Mountain DU live and are adapted, at least behaviourally, to specific environments characterized by high-elevation forest communities that support abundant arboreal lichens. This group of caribou differs markedly from other caribou, including those in the adjacent DU8, as they have persisted in an ecological setting unique to the species that has given rise to local adaptations. As noted above, caribou in the Central Mountain and Northern Mountain DUs differ phylogenetically. Evidence from McDevitt et al. (2009) was suggestive of a “hybrid swarm” of two caribou lineages within the ice-free corridor that appeared along the eastern front of the Canadian Rockies (Central Mountain DU) producing a unique, mixed gene pool at the end of the Wisconsin glaciations ca. 14 000 years ago. Although some evidence indicates genetic relatedness between Northern Mountain DU subpopulations in west-central BC and caribou in the Central Mountain DU, the majority of subpopulations of Northern Mountain DU caribou are genetically different from Central Mountain DU caribou (Serrouya et al. 2012).
Further work needs to be conducted to assess phylogenetics and genetic population structure in the Northern Mountain DU in particular. Although all caribou in the 9 subpopulations of caribou that have been sampled in the Northern Mountain DU belonged to the northern clade (Dueck 1998, Zittlau 2004, Weckworth et al. 2012), this represents only 20% of the subpopulations, with 8 located in Yukon or along the Yukon/BC border. Only two of the 25 subpopulations in northern BC have been sampled, leaving a large gap in phylogenetic information for Northern Mountain DU. Further work is also needed to clarify genetic relationships in the Southern Mountain and Northern Mountain DUs. Based on limited samples, subpopulations in the west-central BC were found to be genetically more similar to subpopulations in the Southern Mountain DU (Zittlau 2004) and the Central Mountain DU (Serrouya et al. 2012) than to other Northern Mountain DU subpopulations. A comprehensive analysis across all DUs is required. There is modest overlap between DUs; however, these boundary areas are limited and there is little inter-mixing of caribou by use of seasonally distinct habitats.
Special Significance
Caribou are an iconic Canadian wildlife species appearing on the Canadian 25 cent coin, and are one of Canada’s most widely distributed large mammals (Species at Risk Public Registry 2013). Western mountain caribou display particular adaptations to weather and topographic conditions in mountainous forests along the western spine of the North American continent. By virtue of their reliance on large tracts of older forest, they are highly sensitive to forest clearing and are regarded as indicators of older and intact forest ecosystems. Over 90% of the distribution of western mountain caribou occurs in Canada (see Global Distribution).
In western Canada, caribou have played an important sustenance role for Aboriginal peoples, as well as for early fur traders and settlers (Birchwater 1993, Spalding 2000, Olsen et al. 2001, Botkin et al. 2005, Littlefield et al. 2007). Caribou are hunted recreationally and for sustenance in most of the Northern Mountain DU. Caribou are valued as trophy animals and resident and guided hunting of caribou are of economic importance in northern BC, Yukon and the Mackenzie Mountains in the NT. Caribou in all three DUs are also valued for wildlife tourism/wildlife viewing.
Distribution
Global Range
Caribou and reindeer are indigenous to arctic, sub-arctic, boreal, and sub-boreal biomes (Banfield 1961, Røed et al. 1991). Caribou in the three western mountain DUs occur mostly in Canada, with the exception of the South Selkirk subpopulation range in the Southern Mountain DU (extending into Idaho and Washington) and that of the Chisana subpopulation in the Northern Mountain DU (in Yukon extending into Alaska). No evaluation has been undertaken to clarify which, if any, other the other 27 caribou subpopulations in interior Alaska (e.g., Delta, Denali, etc.; ADFG 2011) belong to Northern Mountain Caribou DU. In the 19th century, western mountain caribou occurred as far south as the Salmon River in Idaho (Figure 3) but are now extirpated, except for the South Selkirk subpopulation; the last confirmed report of a caribou in Montana was in 1958 (US Fish and Wildlife Service 1993).
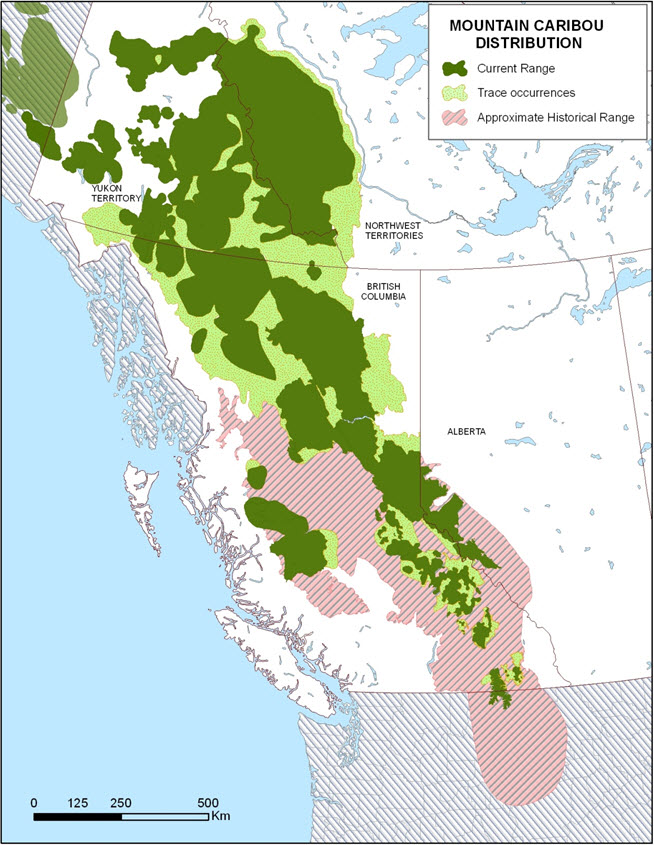
Photo: © Bonnie Fournier 2013
Long description for Figure 3
Map showing the approximate historical (colour and hatching) and current (colour) ranges of caribou in the western mountain designatable units in Canada and the United States. The majority of the range contraction has occurred in the southern portion of British Columbia.
Canadien Range
Historically, caribou in the mountain DUs of western Canada enjoyed a considerably broader distribution than they do currently (Figure 3). Caribou range in BC has been reduced by 20% since the early 1900s (Spalding 2000) with the major change in distribution occurring in the southern portion of the province. ATK holders have reported that caribou range has decreased in the Fraser and Columbia river basins (Hudson and Ignace 2004). In north-central BC, caribou used the Mount Milligan area in the late 1930s (McKay 1997, Santomauro et al. 2012) and in west-central BC, the Tsilhqot’in people used to hunt caribou in the early 1900s (Tsilhqot’in Nation v. British Columbia 2007). Caribou are no longer found in either of those areas. In Alberta, Dzus (2001) found that caribou occupied only about 39% of the generalized maximum historical range (Edmonds 1991); range has continued to recede since that time.
Northern Mountain DU (7)
Caribou in the Northern Mountain DU are distributed among 45 subpopulations ranging from west-central BC to Yukon and NT in the north, including 9 subpopulations in central BC that were part of the former Southern Mountain population of Woodland Caribou (Figure 2; Environment Canada 2014). In Yukon, 26 subpopulations of caribou in the Northern Mountain DU occupy much of the territory south of latitude 65o N. Two of these--Hart River and Bonnet Plume--overlap the winter range of the Porcupine herd of barren-ground caribou and the Forty-mile herd (no affixed DU). The Chisana subpopulation straddles the Yukon-Alaska border. Of the 26 subpopulations in Yukon, 5 are shared with NT and 6 are shared with BC (Figure 2).
Subpopulations in the southern part of the DU have relatively discrete ranges, while range overlap is more pronounced further north. Caribou from the Little Rancheria, Horseranch, Swan Lake and Level-Kawdy subpopulations share overlapping winter ranges, making it difficult to determine the subpopulation origin of caribou counted on the winter range without the aid of radio-collars (M. Williams, pers. comm. 2013). In Yukon, the Laberge subpopulation was only recently identified because it shares the same winter range as the Carcross subpopulation and was previously considered a part of that subpopulation (T. Hegel, pers. comm. 2013). Gaps between subpopulations tend to be wider in BC than in Yukon and NT. Search effort in some of those gaps has been limited but some are known to contain caribou (Thiessen 2009, McNay 2012, MacDonald and McNay 2013). It is unclear whether caribou found in areas between delineated ranges in northern BC belong to adjacent subpopulations or whether they belong to yet unidentified subpopulations (McNay 2012). In Yukon, caribou in the Klaza, Aishihik, Kluane, and Chisana subpopulations are separated from other subpopulations by the Yukon River (T. Hegel, pers. comm. 2013).
Caribou distribution in the northern portion of the Northern Mountain DU has experienced no reduction during the past century, but range recession has occurred in the southern portion of the DU in west-central and north-central BC (Figure 2). Historically, caribou were found throughout the interior plateau region of BC, but are largely absent from that area now (Figure 2, Spalding 2000). From 1984 to 1991, 52 caribou from the Itcha-Ilgachuz subpopulation were transplanted to unoccupied range in the Charlotte Alplands (Young et al. 2001), resulting in the re-establishment of some former range in that area.
Central Mountain DU (8)
The Central Mountain DU includes 10 extant subpopulations of caribou in east-central BC and west-central Alberta located in and around the Rocky Mountains, all of which belonged to the former Southern Mountain population of Woodland Caribou (Figure 2; Environment Canada 2014). There are now 3 subpopulations (Tonquin, Maligne, Brazeau) recognized within Jasper because caribou no longer move among those ranges (L. Neufeld, pers. comm. 2012). Caribou range in this DU recently receded with the death of all 5 individuals in the remnant Banff subpopulation in an avalanche in 2009 (Hebblewhite et al. 2010b). The Burnt Pine subpopulation was confirmed extirpated in 2014 (BC Ministry of Environment, unpublished data). Caribou in the A La Peche subpopulation have mostly abandoned their traditional annual migration to foothills winter range and now spend most of their time in the mountains, further reducing the amount of range occupied (ASRD&ACA 2010). Using historical records of caribou distribution, Edmonds and Bloomfield (1984) documented that caribou in the Alberta portion of the Central Mountain DU have disappeared or only remain as fragmented remnant populations.
Southern Mountain DU (9)
Caribou in the Southern Mountain DU are distributed across 15 extant subpopulations in southeastern BC, all of which belonged to the former Southern Mountain population of Woodland Caribou (Figure 2; Environment Canada 2014). About 80% of the population is found in ranges north of the North Thompson River (Serrouya et al. 2012). Since 2002, two additional subpopulations have been confirmed extirpated (George Mountain in 2003; Central Purcells in 2005), with several others having fewer than 10 individuals (see Population Sizes and Trends).
Wittmer et al. (2005a) reassessed distribution and subpopulation structure of Southern Mountain DU caribou and identified 17 subpopulations (excluding the South Selkirks) with a total range area of 28 357 km2. Since then, the Allen Creek subpopulation was combined with the Wells Gray subpopulation and some subpopulations were renamed, but those range areas are essentially the same as the current ranges. Wittmer et al.’s (2005a) total range represents a 54% reduction from the previous total range estimate of 61 290 km2 (excluding the South Selkirks; MCTAC 2002). Although the earlier total range estimate was based on known or suspected occupancy (MCTAC 2002), the reduction in range reflects a reduction in subpopulation numbers and increasing fragmentation and isolation of subpopulations, especially in the southern portion of the DU (Wittmer et al. 2005a).
Van Oort et al. (2011) assessed additional radio-collared caribou location data and found that the distribution of caribou in the subpopulations recognized by Wittmer et al. (2005a) plus South Selkirks were composed of 41 summer/fall composite ranges with almost no evidence of movements between them. They concluded that the subpopulations were not functioning as a metapopulation due to the lack of dispersal.
Extent of Occurrence and Area of Occupancy
As caribou population size decreases, they occupy less of their range (Bergerud 1996, Schaefer 2003). Caribou may also focus their use in a portion of their range in response to disturbance (Smith et al. 2000, Seip et al. 2007). Although it may be difficult to determine why the population is occupying less range, both situations indicate suboptimal conditions for caribou.
The current extent of occurrence (EO) was measured by fitting a minimum convex polygon around the current range of each DU, including trace occurrences (Figure 2). The EO for the three DUs is: Northern Mountain 1,050,174 km2, Central Mountain 85,986 km2, and Southern Mountain 118,240 km2. The index of area of occupancy, based on a 2 x 2 km2 grid placed on the current subpopulation boundaries (excluding trace occurrences) is: Northern Mountain 523 672 km2, Central Mountain 46 144 km2, and Southern Mountain 46 324 km2. It is likely that the area of occupancy is actually considerably smaller, if measured as “the smallest area essential at any stage to the survival of existing populations” (IUCN Standards and Petitions Subcommittee 2013). However, this was not possible to calculate at the DU scale, as many essential wintering and/or calving ranges for individual subpopulations are unknown or have not been mapped. Both measures of range extent are known to have declined over the past decade, as evidenced by declines or extirpations of several subpopulations (see Population Sizes and Trends). However, the revision of DU boundaries since the 2002 assessment (see Designatable Units) precludes quantification of this change.
Search Effort
The distribution maps of forest-dwelling caribou in the Northern Mountain, Central Mountain, and Southern Mountain DUs are based on range maps provided by each jurisdiction in 2013. For most subpopulations, range areas have been delineated using positions from radio-collared caribou.
In the Central Mountain and Southern Mountain DUs, extensive radio- and GPS-collared caribou studies have been conducted over several years resulting in thousands of telemetry positions (e.g. Wittmer et al. 2005a, Jones 2007, ASRD&ACA 2010, van Oort et al. 2011, Williamson-Ehlers 2012, Seip and Jones 2013). Most of the radio-telemetry studies in the Central Mountain DU were conducted after 2002, so ranges reflect current distribution and range use. In the Central Mountain DU, all but the Scott subpopulation have recent information on range use and distribution (S. McNay, pers. comm. 2013). In the Southern Mountain DU, Wittmer et al. (2005a) delineated current ranges based on radio-collared caribou studies conducted mostly in the mid- to late 1990s and early 2000s.
In the Northern Mountain DU, most subpopulations have had at least some radio-collared caribou on which to base distributions (Environment Canada 2012). Subpopulations with limited information on ranges and distribution include: Bonnet Plume, Tay River, Moose Lake, Gataga, Rabbit, Edziza, and Tsenaglode. The Frog, Gataga, and Rabbit ranges in BC are adjacent to areas of trace occurrences. Recent surveys of caribou and other species in those areas indicate that caribou are using those areas (Thiessen 2009, McNay 2012, MacDonald and McNay 2013). Twelve caribou were recently radio-collared in this area, which will help refine subpopulation boundaries (MacDonald and McNay 2013). In Yukon, most of the caribou ranges and surrounding areas have been surveyed for caribou or for other species so overall distribution is assumed to be mostly known, notwithstanding the dynamic nature of range occupancy (T. Hegel, pers. comm. 2013).
Habitat
Habitat Requirements
Topography, climate, and winter feeding habits divide caribou in the western mountainous region of Canada into two general ecotypes (Stevenson and Hatler 1985, Heard and Vagt 1998). In the Northern Mountain and Central Mountain DUs, snow depths are relatively shallow and caribou feed primarily on terrestrial lichens either in low-elevation forest-dominated habitats or on rounded wind-swept peaks (Johnson et al. 2001, Jones et al. 2007). In the Southern Mountain DU, deep snow causes caribou to eat arboreal lichens found in mid- to high-elevation mountainous habitats (Terry et al. 2000).
In general, caribou require large seasonal ranges. These large areas allow caribou populations to segregate from one another (horizontally and altitudinally) from predators and other ungulates, reducing predation on both adults and calves (Bergerud and Page 1987). Also, access to a broad distribution of habitat allows caribou populations to alter their range use in response to natural disturbance processes (e.g. fire, insects, weather/snow conditions) and human activities such as forest harvesting, mining and mineral exploration, oil and gas development, and recreation activities (McKay 1997, MCTAC 2002, NCTAC 2004, McNay et al. 2008, Environment Canada 2012). High disturbance (anthropogenic and natural) levels and young forest cover within a population range can both preclude options for caribou shifts to suitable habitat and lead to changes in ecological interactions within the system at large; the result is lower survival rates (Wittmer et al. 2007, Williamson-Ehlers et al. 2013).
During winter, caribou require large patches of winter habitat including low-elevation mature pine forests with abundant terrestrial lichens (Central and Northern Mountain DUs), low-elevation mature forests (pine or spruce) or forested wetlands with abundant arboreal lichens (Central and Northern Mountain), high-elevation windswept alpine ridges with abundant terrestrial lichens (Central and Northern Mountain), high-elevation mature subalpine forests with abundant arboreal lichens (all DUs) and low-elevation cedar-hemlock forests (Southern Mountain only). For Central and Northern DU caribou, snow depth or hardness can limit access to terrestrial lichens (Cichowski 1993, Johnson et al. 2001).
Relative to the winter season, the diet of caribou is general in other seasons, reflecting the use of a broader range of habitats during the spring, summer and fall. Typically, caribou calve in areas that are isolated or distant from predators (e.g. high-elevation alpine, subalpine parkland, subalpine forests, islands in lakes). Females often forgo forage quality at low elevations to calve in high-elevation habitat in late May/early June where forage at that time of year is limited (Bergerud et al. 1984). Those that used lower elevations in May had higher fecal nitrogen concentrations than caribou that remained in the subalpine habitat (Seip 1992). For those in northern BC, calving sites are associated with high-quality forage, and higher calf survival corresponded with movements away from calving sites to increase access to forage and/or minimize predation risk (Gustine et al. 2006). Relatively undisturbed “matrix range” (sensu Environment Canada 2014) is necessary for seasonal migrations and connectivity between ranges that maintain or increase genetic diversity, reduce predation risk, and provide potential movement corridors to facilitate response to changing conditions caused by climate change.
Northern Mountain DU (7)
Northern Mountain caribou generally live in relatively shallow snow areas where they forage primarily on terrestrial lichens, either in low-elevation mature coniferous forests or on windswept alpine slopes (Cichowski 1989, Wood and Terry 1999, Gullickson and Manseau 2000). These animals prefer older pine or spruce forests where terrestrial lichens are abundant, but they also forage on arboreal lichens, especially during late winter or when snow crusting affects their ability to crater (Cichowski 1989, Johnson et al. 2001). Some subpopulations forage on arboreal lichens in subalpine habitat during portions of, or all of the winter. At low elevations, caribou use frozen lakes where they crater for ice and/or free water.
The Northern Mountain DU extends from central BC to the NT and spans a variety of ecological conditions. As such, variation in seasonal behaviour reflects differences in topography, snow accumulation, and availability of low-elevation winter ranges between areas. In Yukon, caribou use various winter range strategies; for example, the Chisana, Kluane, Aishihik, and Klaza subpopulations use alpine ranges, whereas seven subpopulations to the east forage in lower-elevation forests (Kuzyk et al. 1999). In BC, the Telkwa and Takla caribou subpopulations in the central part of the province primarily use high-elevation subalpine and alpine habitats throughout the year (Stronen 2000, Poole et al. 2000). Subpopulations in north-central BC use low-elevation forests in early winter, then generally move to higher-elevation subalpine and alpine habitat in late winter (Terry and Wood, 1999, Wood and Terry 1999), although individuals may spend the whole winter in alpine habitat or low-elevation habitat (Johnson et al. 2002). Individual caribou in subpopulations in west-central and northern BC use low elevations or high-elevation subalpine/alpine most of the winter although during some years, a portion of the Tweedsmuir-Entiako subpopulation move into high-elevation subalpine/alpine habitat from mid-February to mid-March (Cichowski 1989, Young and Roorda 1999, Cichowski and MacLean 2005).
Some Northern Mountain subpopulations, or portions of subpopulations, migrate long distances between winter and summer ranges, whereas others do not. During calving (late May to early June), female caribou generally move to high-elevation mountain areas away from predators and other prey (Bergerud et al. 1984). They may also use islands in lakes as an anti-predator strategy (Seip and Cichowski 1996, Cichowski and MacLean 2005). Caribou that calve at higher elevations and caribou that calve at low elevations on islands in lakes tend to have higher neonatal calf survival than those that calve below treeline but not on islands (Seip and Cichowski 1996). During summer, habitat use varies and caribou can be found in a variety of habitats ranging from low-elevation forests to alpine habitat. During the rut, some subpopulations move to rutting areas at higher elevations while others rut on their summer ranges.
Current understanding of caribou habitat use patterns in the Northern Mountain DU based on scientific information is consistent with ATK of caribou habitat use. In north-central BC, Takla, Tl’azt’en, Nak’azdli and Tsay Keh Dene described seasonal habitat use where caribou began appearing at low elevations in April and May and used pine-dominated areas with abundant “white moss”, lakes to “lick the clear ice”, and mineral licks where they were available (McKay 1997, McNay et al. 2008, Santomauro et al. 2012). Caribou then disappeared in late May, presumably moving to higher elevations for calving because caribou were seen with calves at higher elevations during summer (McNay et al. 2008). In early summer, caribou were seen lying on snow patches, which was likely a strategy to avoid flies, and caribou used lakes during ice-free times as a refuge from wolves (Canis lupus; McKay 1997, McNay et al. 2008). Caribou stayed at high elevations during the rut to “dry their horns” then moved back down to low elevations for early winter to avoid fresh snow at higher elevations; some caribou, but not always all caribou, disappeared in January, presumably to use higher elevations for the rest of the winter (McNay et al. 2008). In the Atlin area, Taku River Tlingit members indicated that during winter, caribou selected low-elevation forests, especially mature Lodgepole Pine (Pinus contorta) stands with high lichen ground cover, and during summer, caribou were wide-ranging and used mountain sides where they forage on grass, willow, and lichen (Polfus et al. 2014).
Central Mountain DU (8)
Like Northern Mountain DU caribou, caribou in the Central Mountain DU also live in relatively shallow snow areas where, during winter, they forage primarily on terrestrial lichens in lower-elevation forests or on windswept alpine slopes (Edmonds and Bloomfield 1984, Brown et al. 1994, Szkorupa 2002, Jones 2007, Shepherd et al. 2007).
In Alberta, caribou in the Central Mountain DU typically winter in old pine or mixed pine/spruce/fir forests in the foothills of the Rocky Mountains where they predominantly crater for terrestrial lichens, and then move in spring to summer range in the mountains. During calving, female caribou are highly dispersed and use habitats in mountainous terrain. In recent years, the migratory behaviour of the Jasper subpopulations (Tonquin, Maligne, Brazeau), as well as the A La Peche and Redrock-Prairie Creek subpopulations has changed. Most caribou in those subpopulations no longer use the low-elevation foothills portions of their ranges and are living in the mountains all year round (Smith 2004, L. Neufeld, pers. comm. 2013). Elders from one community in west-central Alberta observed that the A La Peche subpopulation stays around all year and that grizzly bears (Ursus arctos), wolves, and cougars (Puma concolor) have increased in the area, which may further affect caribou (West Central Alberta Caribou Landscape Planning Team 2008). Currently, adult survival is lower for caribou that migrate to low-elevation foothills areas for winter than for those that live year-round in the mountains, suggesting selection against the migratory behaviour (Hebblewhite et al. 2010a).
Caribou in the BC portion of the Central Mountain DU exhibit varying seasonal habitat use patterns (Jones 2007, Williamson-Ehlers 2012). The Kennedy Siding subpopulation uses low-elevation mature pine forests during the rut and early winter then moves to higher-elevation subalpine and alpine habitat for mid- to late winter. The Moberly, Burnt Pine, and Quintette subpopulations are primarily found in alpine and subalpine habitat throughout the year, except during calving and summer/fall when they use more subalpine forested habitat.
Southern Mountain DU (9)
Range use by deep snow/arboreal lichen feeding caribou varies seasonally (Seip 1990, 1992a, Jones 2007). In early winter, caribou use valley bottoms and lower slopes where they forage primarily on arboreal lichens on fallen trees and lichen litterfall, but also on shrubs and forbs that remain accessible in snow wells (Seip 1992a, Mowat et al. 1998, Terry et al. 2000). In early winter, the snowpack is soft and deep so habitats with conifer canopies that intercept snow, enabling access to feeding sites, are important. Use of early winter habitats varies across the range of caribou in the Southern Mountain DU. In general, caribou that live in rugged mountainous terrain make more pronounced migrations to lower-elevation cedar/hemlock and mid-elevation spruce/subalpine fir (Abies lasiocarpa) forests, whereas caribou that live in more highland type terrain or at the north and south ends of the range, primarily use mid- and upper-elevation forests (Terry et al. 1996, Apps et al. 2001).
In mid- and late winter, caribou move to upper slopes and ridge tops after the snow pack deepens and hardens, where they forage on arboreal lichens in subalpine parkland habitats dominated by subalpine fir (Seip 1990, 1992a, Simpson et al. 1997, Hamilton et al. 2000, Apps et al. 2001). During winter, caribou forage almost exclusively on arboreal lichens, predominantly Bryoria spp. (Terry et al. 2000). Foraging may be more difficult in years with relatively low snow packs or poor snow consolidation and when snowpack is >1.5 m lower than normal, caribou may use habitats 300-600 m lower than normal during late winter (Kinley et al. 2007).
In spring, caribou descend to access new green vegetation and use ranges similar to early and late winter ranges (Seip 1990, 1992a, Hamilton et al. 2000). Pregnant caribou move upwards again in May to higher-elevation calving habitats where predators are less abundant, although forage is limited (Seip 1990, 1992a, Hamilton et al. 2000, Stotyn 2008). During spring/calving, males generally remain at lower elevations where food is more available than at higher elevations. During summer, caribou use subalpine, subalpine parkland, and alpine areas where they forage on a variety of herbs and shrubs (Seip 1992a).
Habitat Trends
Change in habitat quality or availability has both direct and indirect impacts for caribou populations from the three DUs (see Threats and Limiting Factors). A reduction in habitat will directly influence the movement, distribution, and foraging ecology of caribou with a potential decrease in nutritional intake and an increase in the energetic costs of seasonal habitat use (Johnson et al. 2002). The link between these costs and reproduction and survival for small low-density populations of caribou are less certain (McLellan et al. 2012). Indirectly, habitat change may support other ungulate species and larger or more widely distributed predator populations across caribou range (i.e., apparent competition; Serrouya et al. 2011). Habitat change may be the result of both natural disturbance, such as fire and insect outbreaks, and anthropogenic activities including forestry, mining, and energy development. Also, disturbance related to human activities may force caribou to move away from otherwise functional habitat (Seip et al. 2006). For many caribou subpopulations, the long-term cumulative impacts of habitat change are more notable than any one disturbance type or event (Nitschke 2008, Williamson-Ehlers et al. 2013).
For western mountain caribou, habitat change has been related empirically to reduced spatial separation between caribou and other prey or predators (Peters 2010, Robinson et al. 2012), changes in distribution including smaller seasonal ranges or abandonment of large areas of habitat (Smith et al. 2000, Apps and McLellan 2006, Seip et al. 2006, Wittmer et al. 2007), reduced survival of caribou (Smith 2004, Wittmer et al. 2007), and to population declines (Wittmer et al. 2007). The fragmentation of the once continuous distribution of western mountain caribou into multiple small and isolated subpopulations (particularly the Southern and Central Mountain DUs; Figure 2) is indicative of a long-term (>3 generations) decline in the area and connectivity of functional habitat for these animals (Spalding 2000).
Most habitat patches within the Central and Southern Mountain DUs apparently do not support viable populations (see Population Abundance and Trends), although two subpopulations (Hart Ranges and Wells Gray) comprise more than half the number of individuals in the Southern Mountain population. By contrast, all individuals in the Central Mountain DU belong to non-viable subpopulations, hence meeting the IUCN definition of “severely fragmented” (IUCN Standards and Petitions Subcommittee 2013). Similarly, extensive alteration of caribou habitat in the Peace-Moberly region of north-eastern BC (within the Central Mountain DU) has occurred as a result of energy development and commercial logging activities that increased greatly in the early 1990s (Nitschke 2008, Williamson-Ehlers et al. 2013). Land-use changes are locally significant, but cover a much smaller proportion of the range of the Northern Mountain DU. The recent Mountain Pine Beetle (Dendroctonus ponderosae) epidemic in BC and Alberta has resulted in wide-scale salvage logging in beetle-killed areas (McNay 2011). This has the largest impact on habitats for the Northern Mountain DU particularly for subpopulations in the southern and western portions of the DU; there are also significant areas of dead pine and salvage logging across the Central Mountain DU. Proposed development of wind energy may have significant impacts on the quality or availability of alpine habitats. There are also a number of oil and gas pipelines that have been proposed for the region (British Columbia Environmental Assessment Office 2014, Energy BC 2014, Lamers 2014).
Northern Mountain DU (7)
Many caribou subpopulations in the Northern Mountain DU are in relatively remote areas. Forest harvesting is currently the most significant industrial activity in the southern portion of the Northern Mountain DU. At a more localized scale, mineral exploration and development activities occur throughout the DU. The level of anthropogenic disturbance in this DU is expected to increase. Forest harvesting is continuing on ranges in west-central and north-central BC and in some areas harvesting has increased to salvage Mountain Pine Beetle-killed stands (Bogdanski et al. 2011). Mineral exploration and development activities have increased on most ranges in the southern Yukon and in portions of central and northwestern BC (PWC 2012; see Threats and Limiting Factors). In an area known as the “Golden Triangle” in north-western BC, there are over 30 known large mineral deposits and at least five giant pit mines in the planning process. The 344 km, 287 kV Northwest Transmission line is being built from Terrace to Bob Quinn Lake for the purposes of supplying power to planned industrial developments and remote communities in the area. In 2013, BC Hydro announced it would extend the transmission line to the Red Chris mine, which lies close to if not within the western boundary of the Spatsizi subpopulation range. Expected to be in operation by mid-2014, the new power supply is likely to increase the feasibility of potential projects in and adjacent to caribou ranges in northwestern BC (BC Hydro 2013).
Central Mountain DU (8)
Forest harvesting and mineral and hydrocarbon exploration and development have all contributed to habitat change in the Central Mountain DU (ASRD&ACA 2010, Ehlers et al. 2014). Williamson-Ehlers et al. (2013) recently assessed landscape level habitat change for four of the eleven subpopulations in the Central Mountain DU. The footprint area of industrial disturbance included 39% of the Moberly/Burnt Pine ranges, 33% of the Quintette range, 19% of the Bearhole/Redwillow portion of the Narraway range, and 11.5% of the rest of the BC portion of the Narraway range. They used positions from 139 GPS-collared caribou to model avoidance behaviour and calculate zones of influence for individual populations relative to wild fire, coal mining, oil and gas exploration and development, and forest harvesting. The greatest amount of habitat change created by industrial development occurred between 2000 and 2009; this time period coincided with known caribou declines on most ranges (Seip and Jones 2013).
The Forest Practices Board of BC recently undertook a cumulative effects assessment of the Kiskatinaw River Watershed, which included caribou habitat in the Bearhole/Redwillow portion of the Narraway range. They found that 50% of the core habitat present in the winter range in the mid-1980s had been lost by 2007 (Forest Practices Board 2011). No animals were found in the Burnt-Pine range in 2012 (Seip and Jones 2013) when at least 39.4% of the neighbouring annual range (with Moberly) contained industrial developments.
Southern Mountain DU (9)
In the Southern Mountain DU, forest harvesting has been the primary source of habitat alteration (MCTAC 2002). Although logging took place in southeastern BC (Southern Mountain DU) from the early 1900s, it was not until the 1970s that large amounts of habitat were lost (Spalding 2000). MCTAC (2002) compared the estimated amount of suitable historical and current habitat. Habitats were rated in 6 classes ranging from Very High to Nil. The amount of historical habitat was calculated based on the assumption that habitat was in the old-growth forest state, the optimal state for caribou. Current suitability was rated based on estimates of the current amount of old-growth forests and habitat fragmentation associated with industrial development. For the top three suitability classes combined (Very High, High, Medium), the extent of currently suitable habitat was 38% lower than the extent of historically suitable habitat. The greatest decline was in the High class, where the amount of currently suitable habitat was 71% lower than the historical level.
Biology
Life Cycle and Reproduction
Detailed background information on the general biology of caribou can be found in Banfield (1974), Kelsall (1984), Miller (1982), Bergerud (2000), and COSEWIC (2011).
The reproductive rate of caribou is low compared to other members of the deer family. Caribou usually have only one calf per year and females do not generally breed until they are at least 2 years old (Bergerud et al. 2008). Where data on age-specific reproduction are available among caribou ecotypes, they suggest an early age of primiparity (e.g. Rettie and Messier 1998). Pregnancy rates are often high for western mountain caribou (> 90%, Seip and Cichowski 1996; ~92 ± 2%, Wittmer et al. 2005a) and do not differ among subpopulations in BC (Wittmer et al. 2005a) or elsewhere in North America (Bergerud et al. 2008). High pregnancy rates suggest that nutrition is not limiting because caribou pregnancy rate is highly sensitive to forage availability (Cameron et al. 1993) and delay in primiparity is among the most sensitive indicators of food limitation in mid-sized ungulates (Gaillard et al. 1998, 2000). Pregnant females travel to isolated, relatively predator-free areas in the mountains to calve in late May or early June (Edmonds 1988, Farnell and McDonald 1990, Seip 1990, 1992a, Jones 2007). Neonatal calf survival is higher for females that calve at high elevations in mountainous terrain or below treeline on islands in lakes (where predators are less abundant), than for females that calve below treeline but not on islands (Seip and Cichowski 1996). Tsay Key Dene ATK holders noted that caribou use high-elevation sites near the treeline for calving. Caribou prefer sites with a south aspect and deep snow conditions, providing protection from harassment/disturbance by either humans or predators (McKay 1997, McNay et al. 2008).
Calf survival during the first few months after birth tends to be low, with most calf mortality occurring by the fall after birth. For example, a number of caribou herds in the Northern Mountain DU show a consistent pattern of a high level of calf mortality over the first two months of life, followed by summer and winter calf mortality rates that vary among herds. While most calf mortality has occurred by the fall after birth, for herds studied in northern BC (Swan Lake, Little Rancheria and Horseranch) a significant level of calf mortality can occur over the winter and the timing and extent of calf mortality varies among years and among herds (MFLNRO unpublished data). Calf mortality during the first two months of life ranged between 21% and 46% for the Pink Mountain subpopulation (Gustine et al. 2006a). By fall, 3 to 39% of radio-collared female caribou still had calves in the Spatsizi, Tweedsmuir, Wells Gray (south), and Wells Gray (north) subpopulations (Hatler 1986, Seip 1990, 1992a, Cichowski and MacLean 2005). In late winter, reported % calves has ranged from 1 to 23% (average 12%) for Southern Mountain DU subpopulations (Wittmer et al. 2005a), 9 to 15% for subpopulations in the Central Mountain DU (Seip and Jones 2013), and 10 to 18% for subpopulations in the Northern Mountain DU (Young and Freeman 2001, Cichowski and MacLean 2005, Tripp et al. 2006, Florkiewicz 2008, McNay et al. 2010, BC MFLNRO unpublished data). According to Tlingit ATK holders, the Carcross caribou subpopulation has appeared to have low recruitment of calves starting in the late 1970s (Botkin et al. 2005). Low calf recruitment in other herds in northern BC (MFLNRO unpublished data) has also been documented with only 12 of 20 late winter composition surveys on the Atlin, Swan Lake, Little Rancheria and Horseranch herds between 1995 and 2008 showing calf:100 cow ratios of fewer than 25 calves:100 cows. This lies below the 15% theoretical threshold that Bergerud (1996) suggests is needed to stabilize numbers in forest-dwelling caribou subpopulations. This recruitment threshold is, however, tightly linked to a high (>90%) female survival and could sometimes be insufficient to balance high adult mortality rates (Wittmer et al. 2005a).
Adult female (>1 year) survival rate of female caribou varies among subpopulations (see Appendix 1). In the Northern Mountain DU, mean annual survival rates of adult radio-collared caribou ranged from 83% to 90%, and in the Central Mountain and Southern Mountain DUs, mean annual female survival rates ranged from 74% to 92%, and 55% to 96% respectively (Hayes et al. 2003, Cichowski and MacLean 2005, Wittmer et al. 2005a, Tripp et al. 2006, ASRD&ACA 2010, Seip and Jones 2013, BC MFLNRO, unpublished data). Predation has been the major cause of death of radio-collared adults and calves and much of the behaviour of caribou is related to reducing risk of predation (see Threats - Predation).
Calculations of generation length for subpopulations with available data from the Northern Mountain, Central Mountain and Southern Mountain DUs (using a formula from IUCN Standards and Petitions Subcommittee 2013) ranged between 8 and 9 years (see Appendix 1).
Physiology and Adaptability
During winter, caribou in the Northern Mountain, Central Mountain, and Southern Mountain DUs forage primarily on lichens, although conifers, shrubs, graminoids, forbs, horsetails (Equisetum spp.), and mosses are also ingested (Seip 1990, Farnell and McDonald 1990, Farnell et al. 1991, Cichowski 1993, Thomas et al. 1996, Gullickson and Manseau 2000). Together with specialized bacteria and protozoa in their rumens for efficient digestion of lichens, caribou are able to recycle urea to conserve nitrogen, of which preferred lichens are deficient (Parker et al. 2005). During winter, they use fat reserves to meet energy requirements and catabolize body protein when fat reserves are depleted or their diet is nitrogen deficient (Taillon et al. 2013).
In the Northern Mountain and Central Mountain DUs, terrestrial lichens are the primary winter forage except in west-central BC, where arboreal lichens also make up a significant component of the diet (Farnell and McDonald 1990, Farnell et al. 1991, Cichowski 1993, Thomas et al. 1996, Gullickson and Manseau 2000). Horsetails comprise up to 7% of the winter diet (Farnell and McDonald 1990, Thomas et al. 1996, Gullickson and Manseau 2000) and extensive foraging for horsetail in seepage spruce forests has been observed (Cichowski and MacLean 2013). In the Southern Mountain DU, arboreal lichens are the primary winter forage; however, in early winter, caribou also use lower-elevation cedar/hemlock forests where they forage primarily on arboreal lichens (both on standing and fallen trees), but also on terrestrial lichens, conifers, forbs and shrubs (Seip 1992a, Mowat et al. 1998, Terry et al. 2000, Kinley et al. 2003).
During spring, summer and fall, use of lichens decreases while use of vascular and other plants increases. On the Itcha-Ilgachuz caribou range in the Northern Mountain DU, lichens made up over 60% of the diet except from July to October, when they fed primarily on graminoids, forbs, shrubs, and mosses (Cichowski 1993). In the Central Mountain DU, caribou in Jasper Park consumed primarily terrestrial lichens, willows, gramminoids, and forbs (Thomas et al. 1996). In the Southern Mountain DU, caribou that moved into lower-elevation cedar/hemlock forests during spring commonly used gramminoids, horsetails, mosses, and conifers (Simpson 1987, Seip 1990), and during summer, forbs, gramminoids, shrubs, and lichens were predominately used (Seip 1990). ATK holders from the Taku River Tlingit First Nation indicated that Atlin area caribou are wide-ranging during summer and use mountainsides and slopes where they forage on grass, willow, and lichen (Polfus et al. 2014).
Caribou and reindeer can withstand severe cold because their thick winter coat contains insulating semi-hollow hair (Timisjärvi et al. 1984), but they are susceptible to heat stress (Yusef and Luik 1975). The dark summer coat absorbs all wavelengths and suggests the importance of shade and cool forest cover types. Adaptations to snow include large feet and a furred muzzle.
Interspecific Interactions
Caribou are usually one of several prey species in multiple predator-prey systems in the Northern Mountain, Central Mountain, and Southern Mountain DUs. Wolves are the primary predator of caribou (Edmonds 1988, Farnell and McDonald 1988, Seip 1992b, Hayes et al. 2003, McNay 2009, Whittington et al. 2011); however, bears, cougars, golden eagles (Aquila chrysaetos), and wolverine (Gulo gulo) also contribute to adult and calf mortality in some areas (Kinley and Apps 2001, Cichowski and MacLean 2005, Wittmer et al. 2005b, Gustine et al. 2006a, McNay 2009, Milakovic and Parker 2013). Habitat changes brought about by industrial development and increases in early seral forests have led to changing community structure of predators and ungulates, affecting ecological interactions (see Threats – Predation). In a multiple predator-prey system, caribou are seldom the primary prey source for predators, but suffer significant mortality.
In the western mountains, caribou co-occur with several ungulate species including moose (Alces americanus), elk (Cervus elaphus), deer (Odocoileus sp.), and wood bison (Bos bison athabascae). There is no evidence that caribou actively avoid other ungulates yet separation usually is the case because their use of habitat differs. Nevertheless, as caribou share common predators with many other prey species, predation risk is minimized through spatial segregation of prey species, where the less competitive prey (i.e. caribou) does not use areas where the most competitive prey (e.g. moose) is more abundant (Holt 1984). In western mountains, caribou seem to seek habitat types that are unsuitable to other prey species to ultimately segregate from wolves (Seip 1992). Although competition between caribou and Wood Bison has been a cause for concern by local communities, focused research has failed to find substantial diet, habitat, or spatial niche overlap between these species (Jung and Czetwertynski 2013). In a shared landscape, they employ significantly different positions along the grazer-intermediate-browser gradient (Hofmann 1989), and as such have different dietary requirements, which translate to the use of different habitats and spatial distribution (Jung and Czetwertnyski 2013).
Dispersal and Migration
Northern Mountain DU
In the Northern Mountain DU, caribou generally migrate between summer and winter ranges (Farnell and Russell 1984, Culling et al. 2005, Parker and Gustine 2007). However, in some subpopulations, individual caribou may remain on the winter range (Gullickson and Manseau 2000, Culling et al. 2005) or on the summer range (Cichowski 1993, 2010, Backmeyer 2000) all year. For example, some collared individuals of the Redstone showed seasonal migratory movements while others were sedentary year round; ATK holders from the Sahtu speak of a sedentary herd (Olsen et al 2001, J. Nagy pers. comm.) Individuals may also exhibit plasticity in migrational behaviour. On the Tweedsmuir range, for example, one caribou moved to low-elevation winter range for two winters, then remained on high-elevation summer range during the third winter (Cichowski 2010). During spring migration, caribou mainly use low-elevation snowfree areas in valley bottoms (Farnell and Russell 1984, Cichowski 1993, Steventon 1996, Backmeyer 2000) and females initiate spring migration earlier than males (Hatler 1986). Fidelity to calving areas is high but fidelity to winter areas is more variable (Wood and Terry 1999, Gullickson and Manseau 2000, Tripp et al. 2006). Although caribou return to the same general area during winter, they may use different parts of the winter range in different years and/or move between portions of the winter range during each winter (Hatler 1986, Cichowski 1993, Terry and Wood 1999, Culling et al. 2005). Some subpopulations or portions of subpopulations may share wintering areas with other subpopulations but move back to their summer/rutting ranges (e.g. Swan Lake, Little Rancheria, Horseranch, Level Kawdy; BC MFLNRO, unpublished data). Few incidents of dispersal between subpopulations have been documented.
Central Mountain DU
In the Central Mountain DU, movement between summer and winter ranges is variable. In Alberta, the Narraway, Redrock-Prairie Creek, and A La Peche subpopulations migrate between summer and winter ranges (Edmonds 1988, Saher 2005), but more recently, the migratory behaviours of the Redrock-Prairie Creek, A La Peche, and Jasper subpopulations (Tonquin, Maligne, Brazeau) have changed; most caribou no longer use the low-elevation foothills portions of their ranges and are living in the mountains all year round (Smith 2004, L. Neufeld, pers. comm. 2013). The subpopulations in BC generally move from wintering areas on the eastern side of the Rocky Mountains to summering areas in the central part of the Rocky Mountains, although some animals remain in wintering areas all year (D. Seip, pers. comm., 2013).
During spring migration, caribou use lower-elevation portions of drainages for travel, and females begin spring migration earlier than males (Edmonds 1988, Saher 2005). Fidelity to calving areas is high (Brown et al. 1994, Edmonds 1988). ATK elders have noted that the A La Peche subpopulation has a strong fidelity to their home range, staying around all year despite an increase of grizzly, wolves, and cougars in the area (West Central Alberta Caribou Landscape Planning Team 2008). In BC, some range overlap occurs mostly during summer in the Scott, Kennedy Siding, Moberly, Burnt Pine, Quintette, and Bearhole/Redwillow portion of the Narraway subpopulation (Seip and Jones 2013).
There is little evidence of dispersal between subpopulations. One incidence of dispersal observed between ranges was of the one remaining radio-collared caribou in the Burnt Pine subpopulation, which moved to the Scott range in October 2012, but then returned to the Burnt Pine range in summer 2013 (D. Seip, pers. comm. 2013). The three caribou ranges in Jasper National Park are now recognized as separate subpopulations because caribou no longer move among them (L. Neufeld, pers. comm. 2013).
Southern Mountain DU
In the Southern Mountain DU, migration is generally altitudinal, although some subpopulations also migrate between winter and summer ranges (Seip 1990), and caribou exhibit greater fidelity to calving/summer areas than they do to wintering areas (Wittmer et al. 2006). No dispersal between subpopulations has been documented for juveniles, and adult dispersal rate between subpopulations was <0.5% (van Oort et al. 2011).
Population Sizes and Trends
Population estimates are difficult to obtain for forest-dwelling caribou as they live in remote areas and occupy large ranges at relatively low densities. Survey techniques have been developed for most subpopulations, but for some, population estimates may be based on extrapolations from other types of data or expert opinion.
Population trends in this status report are based on available survey data and/or population growth rate data. Population estimates for each DU are not compared to previous summaries (e.g. Williams and Heard 1986, Edmonds 1991, Ferguson and Gauthier 1992, Mallory and Hills 1998, COSEWIC 2002) because most of those summaries do not include all of the subpopulations currently recognized in the 3 DUs covered in this report. For most surveys, the number of adults counted during the survey was used to estimate mature individuals (see Biology). Most surveys did not classify beyond adult male, adult female, unidentified adult and calves, and most provided no data on yearlings.
Northern Mountain DU (7)
Sampling Effort and Methods
In the Northern Mountain DU a variety of methods were used to estimate population numbers. For subpopulations where a large proportion of the population uses high-elevation alpine/subalpine habitat seasonally, surveys of the open areas provide minimum counts. When available, such as in a mark-resight framework (Hegel et al. 2012), the proportion of radio-collared caribou seen is used to correct for caribou missed during the survey and to provide confidence limits. Stratified random quadrat surveys are another method used to estimate forest-dwelling caribou numbers (Gasaway et al. 1986); in contrast to minimum counts, confidence limits around the subpopulation estimates are produced. Distance sampling (Nielsen et al. 2006) has also been used for the Carcross subpopulation in Yukon (Environment Yukon, unpublished data). For some subpopulations, the only estimates available are based on sightings of caribou during surveys conducted for other species (e.g. Thiessen 2009). In addition to population surveys, fall composition surveys are conducted to determine age/sex ratios (e.g. Young and Freeman 2001, McNay et al. 2010, Kerckhoff 2013, BC MFLNRO unpublished data, Environment Yukon unpublished data). For the Bonnet Plume and Redstone subpopulations, fall composition is based on hunter observations that have been collected since 1991 (Larter 2012).
Population size and trend data are limited for the Northern Mountain DU. Only one estimate is available for 18 of 45 subpopulations; some early surveys for the other subpopulations did not always include all of the range and are not comparable to more recent estimates (Appendix 2). The most consistently surveyed subpopulation has been the Itcha-Ilgachuz subpopulation in west-central BC, which was surveyed almost every year from 1978 to 2003 and then on average every 2 years afterwards (Young and Freeman 2001, BC MFLNRO, unpublished data). For that subpopulation, population surveys are conducted in June when most of the females are in the alpine zone and supplemented with fall composition data (Young and Freeman 2001).
Abundance and Trends
About 50 000 - 55 000 caribou occur in the Northern Mountain DU, of which there are an estimated 43,443 to 47,752 mature individuals (Table 1; Appendix 2). These animals account for about 95% of western mountain caribou in Canada. Although most of the subpopulation estimates have been derived from surveys, 29 of the 45 estimates are already over 5 years old or based solely on expert opinion, and may not reflect the current status. Over half (26 of 45) of the subpopulations contain more than 500 mature individuals, while 13 subpopulations contain fewer than 250 (Table 1). Nine of the 15 subpopulations that consist of more than 1000 mature individuals are located in Yukon and NT. Combined, the Bonnet Plume and Redstone subpopulations, the two largest in the DU, comprise >15 000 animals, or 26-29% of the Northern Mountain DU. The four subpopulations that consist of fewer than 50 mature individuals are located in the southern part of the DU in west-central BC (Charlotte Alplands, Rainbows, Telkwa) and northeastern BC (Finlay).
Table summarizing data on trends in numbers of mature caribou within the past three generations (27 years) in subpopulations of the Northern Mountain designatable unit. Information includes subpopulation name, percentage change, most recent estimate of population, current trend, and notes on population management.
| Subpopulation | Footnote1table 1 |
Footnote1table 1 |
Footnote1table 1 |
Footnote1table 1 |
Footnote1table 1 |
Footnote1table 1 |
Footnote1table 1 |
Footnote1table 1 |
Footnote1table 1 |
Footnote1table 1 |
Footnote1table 1 |
Footnote1table 1 |
Footnote1table 1 |
Footnote1table 1 |
Footnote1table 1 |
Footnote1table 1 |
Footnote1table 1 |
Footnote1table 1 |
Footnote1table 1 |
Footnote1table 1 |
Footnote1table 1 |
Footnote1table 1 |
Footnote1table 1 |
Footnote1table 1 |
Footnote1table 1 |
Footnote1table 1 |
Footnote1table 1 |
|
|
Footnote1table 1 Footnote2table 1 |
Population Management |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Northwest Territories / Northern Yukon Hart River |
- | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | X | - | - | - | - | - | - | - | - | 1 853 | U | - |
| Northwest Territories / Northern Yukon Clear Creek |
- | - | - | - | - | - | - | - | - | - | - | - | - | - | X | - | - | - | - | - | - | - | - | - | - | - | - | - | 801 | U | - |
| Northwest Territories / Northern Yukon Bonnet Plume |
O | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 4 200 | =Footnote3table 1 | - |
| Northwest Territories / Northern Yukon Redstone |
- | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | X | - | - | 7 300-10 000 | =Footnote4table 1 | - |
| Northwest Territories / Northern Yukon South Nahanni |
- | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | X | - | - | - | - | +10%Footnote5table 1 | 1 886 | = | - |
| Northwest Territories / Northern Yukon Coal River |
- | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | X | - | - | - | - | - | - | 413 | U | - |
| Northwest Territories / Northern Yukon La Biche |
- | - | - | - | - | - | X | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 388 | U | - |
| Southwest Yukon Chisana |
- | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | X | - | - | - | -3% | 587 | = | Captive rearing 2003-2006; Closure of licensed and First Nation (voluntary) harvest, mid-1990s to present |
| Southwest Yukon Kluane |
- | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | X | - | - | - | - | -23% | 163 | ↓ | Wolf removal and sterilization on adjacent Aishihik subpopulation 1993-1997Footnote6table 1; Closure of licensed and First Nation (voluntary) harvest, mid-1990s to present |
| Southwest Yukon Aishihik |
- | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | X | - | - | - | - | +30% | 1 813 | ↑ | Wolf removal and sterilization 1993-1997Footnote6table 1; Currently under a permit-based licensed harvest |
| Southwest Yukon Klaza |
- | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | X | - | - | 1 065 | U | Wolf removal and sterilization on adjacent Aishihik subpopulation 1993-1997Footnote6table 1; Currently under a permit-based licensed harvest |
| Central Yukon Ethel Lake |
- | - | - | - | - | - | X | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 289 | U | Voluntary harvest closure |
| Central Yukon Moose Lake |
- | - | - | - | X | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 270 | U | - |
| Central Yukon Tay River |
- | - | - | - | X | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 2 907 | U | - |
| Central Yukon Tatchun |
- | - | - | - | - | - | - | - | - | - | - | - | - | X | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 415 | U | - |
| Central Yukon Pelly Herds |
- | - | - | - | - | - | - | - | - | - | - | - | - | - | - | X | - | - | - | - | - | - | - | - | - | - | - | - | 876 | U | - |
| Central Yukon Finlayson |
- | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | X | - | - | - | - | - | - | +13%Footnote7table 1 | 2 657 | ↓ | Wolf removal 1983-1989; Currently under a permit-based licensed harvest |
| Central Yukon Wolf Lake |
- | - | - | - | - | - | - | - | - | - | - | X | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | -Footnote8table 1 | 1 240 | U | - |
| Southern Lakes Yukon LabergeFootnote9table 1 |
- | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | X | - | - | - | - | - | - | - | - | - | - | - | 176 | U | - |
| Southern Lakes Yukon Ibex |
- | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | X | - | - | - | - | - | +127% | 748 | ↑ | Closure of Yukon licensed and First Nation (voluntary) harvest, mid-1990s to present |
| Southern Lakes Yukon CarcrossFootnote9table 1 |
- | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | X | - | - | - | - | - | - | +116% | 674 | = | Closure of Yukon licensed and First Nation (voluntary) harvest |
| Southern Lakes Yukon AtlinFootnote9table 1 |
- | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | X | - | - | - | - | - | - | -2% | 514-857 | = | Closure of Yukon licensed and First Nation (voluntary) harvest |
| Northwest BC Swan LakeFootnote9table 1 |
- | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | X | - | - | - | - | - | - | - | 515-686 | U | - |
| Northwest BC Little RancheriaFootnote9table 1 |
- | - | - | - | - | - | - | - | - | - | - | - | X | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 672-1 342 | U | - |
| Northwest BC HorseranchFootnote9table 1 |
- | - | - | - | - | - | - | - | - | - | - | - | - | X | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 680-850 | U | Wolf removal 1977-1980, 1985 |
| Northwest BC Level Kawdy |
- | - | - | - | - | - | - | - | - | - | - | X | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 1 239 | U | - |
| Northwest BC Edziza |
- | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | X | - | - | - | - | - | - | - | - | 140 | U | - |
| Northwest BC Tsenaglode |
- | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | O | - | - | - | - | - | - | 85-340 | U | - |
| Northwest BC Spatsizi |
- | - | - | - | - | - | - | X | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 2 258 | U | - |
| Northeast BC Liard PlateauFootnote9table 1 |
- | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | X | - | - | - | +15% | 140 | ↓Footnote10table 1 | - |
| Northeast BC Rabbit |
- | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | X | - | - | - | - | - | - | - | 1095 | U | Wolf removal 1982-1985 |
| Northeast BC Muskwa |
- | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | X | - | - | - | - | - | - | - | 828 | U | Wolf removal 1984,1985, 1987 |
| Northeast BC Gataga |
- | - | - | - | - | - | - | - | - | - | - | - | - | X | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 220 | U | - |
| Northeast BC Frog |
- | - | - | - | - | - | - | - | - | - | - | - | - | - | X | - | - | - | - | - | - | - | - | - | - | - | - | - | 199 | U | - |
| Northeast BC Finlay |
- | - | - | - | - | - | - | - | - | - | - | - | - | - | - | X | - | - | - | - | - | - | - | - | - | - | - | -89% | 19 | ↓ | - |
| Northeast BC Pink Mountain |
- | - | - | - | - | - | X | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 1 145 | U | - |
| North-central BC Graham |
- | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | X | - | - | - | - | -Footnote11table 1 | 637 | = | - |
| North-central BC Chase |
- | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | X | - | - | - | - | - | 404 | U | - |
| North-central BC Wolverine |
- | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | X | - | - | - | -9% | 298 | U | - |
| North-central BC Takla |
- | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | X | - | - | - | - | - | - | - | - | - | - | 98 | U | - |
| West-central BC Telkwa |
- | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | X | -64% | 19 | ↓ | Augmented 1997-1999Footnote12table 1 |
| West-central BC Tweedsmuir |
- | - | - | - | - | - | - | - | - | - | - | - | - | - | - | X | - | - | - | - | - | - | - | - | - | - | - | -47%Footnote13table 1 | 248 | ↓ | - |
| West-central BC Itcha-Ilgachuz |
- | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | X | - | +81%Footnote14table 1 | 1 220 | ↓ | - |
| West-central BC Rainbows |
- | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | X | - | - | - | - | - | -53% | 43 | ↓ | - |
| West-central BC Charlotte Alplands |
- | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | X | - | -84% | 6 | ↓ | Transplants 1984-1991Footnote15table 1 |
| Total | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 43 443- 47 752 |
- | - |
Trend data are even more limited for this DU than population size data: Long-term (three-generation) trend is known for only 15 of 45 subpopulations and more recent trends are available for 18 subpopulations (Table 2). Over the last three generations, the best available information (including expert opinion in many cases) indicates that 4 subpopulations have increased, 5 remained stable, and 6 have decreased. Current trends indicate that 2 subpopulations are increasing, 7 are stable and 9 are decreasing. The 2 currently increasing and 6 of the 7 currently stable subpopulations are located in the northern part of the DU in Yukon and NT and along the Yukon/BC border. The Kluane and Finlayson are the only 2 subpopulations known to be declining in Yukon. The Finlayson subpopulation increased in the late 1980s during wolf removal efforts, but has been decreasing since that program ended. The Aishihik subpopulation decreased from the early 1980s until the 1990s but started increasing during wolf removal efforts in the mid-1990s, and has continued to increase. Members of the Champagne and Aishihik First Nation in the southwestern Yukon began noticing a decline in the Aishihik caribou subpopulation in the 1980s, which was believed to have resulted from increased predation and overhunting (Hayes and Couture 2004).