Caribou (Rangifer tarandus) specific populations: COSEWIC assessment and status report 2014, part 2
COSEWIC Assessment and Status Report
Wildlife Species Description and Significance
Name and Classification
Class: Mammalia
Order: Artiodactyla
Family: Cervidae
Scientific name: Rangifer tarandus (Linnaeus 1758)
Common Names: Caribou (English and French); Qalipu/Xalibu (Mi’kmaq); Minunasawa atikw (Innu); Ahtik/Atik (Cree); Tǫdzi (Tłįchǫ); T’onzi/Tohzi (North Slavey); Vadzaih (Gwichin); Ch’atthaii (Vuntut Gwichin); (see COSEWIC 2012 regarding Aboriginal names).
Caribou are an inseparable component of many Aboriginal societies and the species has names in many languages. COSEWIC (2012) contains an extensive list of Caribou names used with permission from Aboriginal traditional knowledge (ATK) collection exercises. Reindeer is the common name in Eurasia. Reindeer have been introduced in some parts of Alaska and Newfoundland. All Caribou and Reindeer, despite physical differences like size and colouration, are the same species and able to interbreed and produce fertile offspring.
Terminology in Caribou ecology is complex and confusing because similar terms (e.g., woodland, boreal, and forest-dwelling) have been used to describe ecotypes and subspecies interchangeably. This report follows the designatable unit (DU) structure outlined in COSEWIC (2011). The Boreal population (BP) refers to ‘Boreal Caribou’ (Environment Canada 2012) or relatively non-migratory Caribou (‘sedentary ecotype’) of mainly forested regions (COSEWIC 2011). Within each DU, the term ‘Caribou’ refers to information collected within that DU, unless otherwise noted. Thus, ‘Caribou’ in the BP DU refers to Boreal or Woodland Caribou (sensu Banfield 1961), unless otherwise noted. Regulations within the BP DU often apply to ‘Boreal Caribou’ and this term is used if regulations are discussed.
Morphological Description
Caribou exhibit tremendous variation in morphology, ecology, and behaviour across their range (Geist 1998). Caribou are medium-size deer with relatively long legs, large crescent-shaped hooves, and broad muzzles with large nostrils. Both hooves and muzzles are covered by hairs as adaptations to cold. The hooves constitute one of the most obvious adaptations to their environment: they are very large and often wider than they are long, and thus are well-suited to walk on deep soft snow, dig through crusted snow for winter forage, and swim across large or turbid bodies of water. Caribou are the strongest swimmers among the deer family (Kelsall 1968; COSEWIC 2012). A Pikangikum elder noted that:
“When they fall through the ice … they have the ability to get out of the water because of the way their hooves are structured… When there is no snow on the ice, the caribou can run… It will not slip. It is even able to gallop…” (O’Flaherty et al. 2007).
Coat colouration varies seasonally and between DUs. NP Caribou have pale fur with a very long, white neck mane in bulls (see cover page) although their antlers are similar to those of the sedentary ecotype (Geist 1998). During winter, GP Caribou are pale grey or almost white, while in summer, the coat turns dark brown. During the rut, male bodies are brown with a very long beige mane and brown face (see cover page). BP Caribou typically are brownish-grey along the back, sides, and legs, and with white or pale underparts and rump. The genus Rangifer is the only cervid where females usually possess antlers, although they are smaller than male antlers. Antler appearance is highly variable between ecotype, sex, age, and reproductive season, and antler mass and size are sensitive to changing environmental and nutritional conditions (Bergerud et al. 2008).
Population Spatial Structure and Variability
Climate-driven range fluctuations during the Pleistocene epoch reshaped Rangifer distribution after the last glaciation (e.g., Grayson and Delpeche 2005; Sommer et al. 2011). A phylogenetic study by Røed et al. (1991) concludes that Caribou recolonized North America and Eurasia from at least two refugia: one north of the Beringia-Eurasia ice sheet and one south of the North American ice sheets (Yannic et al. 2013). Flagstad and Røed (2003) suggested that the North America refugium was composed of several separate refugial populations, which was confirmed by a recent phylo-geographical analysis that suggested that postglacial expansion of Caribou dates back 14,000-22,000 years from three separate refugia located south of the ice sheets: the Rocky Mountains; east of the Mississippi; and the Appalachian Mountains (Klütsch et al. 2012). These separate refugia corresponded to distinct genotypic lineages that diverged before the last glacial maximum (38,000-48,000 years).
All three DUs were named by Banfield (1961) as part of the same subspecies, Rangifer tarandus caribou; but Newfoundland Caribou were considered a distinct subspecies (R. t. terraenovae; Bangs 1896) until the reclassification by Banfield (1961). Geist (2007) proposed retention of subspecies status for NP Caribou based on nuptial characteristics (coat colour, antler shape). The population does contain several haplotypes not found in other Rangifer populations (Cronin et al. 2005) but the subspecies issue is not resolved. Within the NP, mtDNA analyses suggest there is minor genetic structure, except between the sub-population on the Avalon Peninsula and remaining sub-populations (Wilkerson 2010).
COSEWIC uses the term ‘sub-population’ for populations within a species’ or DU range. In Caribou literature, other terms, such as herd, range, and local population are often used for groupings below the DU level. Delineation of these ‘sub-units’ can be difficult (Environment Canada 2011; Nagy 2011). In BP Caribou, sites are reused annually but females disperse from one another at calving, and population structure can be described as a continuum of local populations or groups of Caribou that may exchange a small percentage of individuals within the continuous range and change areas over time (Couturier et al. 2009). Delineating populations in the BP range is thus a challenge, and Schaefer et al. (2001) proposed the use of fuzzy cluster classification to identify the spatial structure of populations. Fuzzy cluster analysis allows group identity to be a matter of degree of membership (Schaefer and Wilson 2002), which fits well to the ecology of the BP. Using the same methods, Rudolph et al. (2012) confirmed the spatial structure of three populations in northern Québec. Nagy (2011) identified two populations in the NT; however, these clusters may have been an artefact of lack of data from the centre of the NT range. BP are treated as one continuous distribution of individuals within their range in the NT (NT Species at Risk Committee 2012). When a local population becomes isolated, the population structure changes from a diffuse continuum of individuals to a distinct group that lacks movement between groups. Thus, distinct BP local ranges, such as in Alberta, are likely a product of anthropogenic landscape disturbance as opposed to how the animals organize themselves on an intact landscape.
Delineation of GP and NP Caribou into identifiable subpopulations is less of a concern because a large proportion is restricted to mountain tops (GP) or plateaus (NP), and both generally use more open habitat (see Habitat Use section).
For the BP, the Federal Recovery Strategy, developed in association with provincial agencies, recognizes population ranges, defined as the geographic area occupied by a group of Caribou subject to similar factors affecting their demography and used to satisfy their life history processes over a defined time frame (Environment Canada 2012). Within the population range, three types of ranges were identified based on the certainty of their boundary delineation: ‘conservation unit’ (low certainty); ‘improved conservation unit’ (medium certainty); and ‘local population unit’ (high certainty). ‘Local population’ refers to a group of Boreal Caribou occupying any of the three types of Boreal Caribou ranges (Environment Canada 2011, 2012). Some of these ranges are remnant, isolated populations and easily delineated (i.e., Coastal Range in Ontario, Charlevoix Range in Québec, Little Smoky in Alberta), while most others exist in a continuous distribution and are less easily delineated (e.g., ‘improved conservation units’ in Ontario; OMNR 2014; Elder pers. comm.). The term ‘herd’ is used in the GP and NP (Weir et al. 2013).
Designatable Units
Twelve DUs (one extinct) were defined for Caribou in Canada based on guidelines proposed in COSEWIC (COSEWIC 2011). DU delineation was based on five lines of evidence: (1) phylogenetics; (2) genetic diversity and structure; (3) morphology; (4) movements, behaviour, and life history strategies; and (5) distribution (COSEWIC 2011). Isolated sub-populations are located in BC, Alberta, Ontario, and Québec but these are not valid DUs because they exist due to anthropogenic disturbances, rather than as a DU that has evolved from local conditions (Environment Canada 2012). This report will assess the status of three DUs of Caribou: the Newfoundland population (NP); Gaspésie population (GP); and the Boreal population (BP). The NP is identified as a DU based on evidence of phylogenetic divergence and genetic discreteness from mainland populations due to isolation, as well as distinctive morphology. The GP is identified as a DU based on genetic differentiation from nearest DUs, facilitated by the movement barrier of the St. Lawrence River, and its status as the last sub-population from the Maritime region. The BP is identified as a DU based on aggregation and migration strategies adapted to the boreal forest landscape, and a lack of genetic flow between adjacent Caribou DUs (COSEWIC 2011).
Special Significance
Caribou play a significant role in the ecology, economy, and culture of much of Canada (Festa-Bianchet et al. 2011). This animal was the most abundant large mammal in much of its range, providing food, tools and clothes to people for thousands of years (Gordon 2003). Caribou continue to play a vital role in societal cohesion and form the basis of many legends and spiritual practices that depict the strong relationships linking Caribou and Aboriginal people (Hummel and Ray 2008; Vors and Boyce 2009). For example, a Gwich’in elder living in the Yukon explained that Caribou and Gwich’in always carry a part of one another’s hearts deep within them; she added that Caribou blood runs through her blood and that of all Arctic peoples (Hummel and Ray 2008). Caribou are hunted for subsistence and sport hunting, both of which generate significant economic contributions (Wells et al. 2011).
Caribou have been proposed as a keystone species of the boreal forest because of their role as a prey species, and because of their impact on vegetation and nitrogen cycling (Festa-Bianchet et al. 2011). Maintaining a healthy Caribou presence in the boreal ecosystems may therefore ensure that their biodiversity is perpetuated while maintaining their ecological services (Fortin et al. 2011).
Distribution
Global Range
Rangifer has a widespread circumpolar distribution in boreal, subarctic, and arctic biomes. Most Reindeer populations are found in Norway, Sweden, Finland, and Russia, while Caribou occupy large portions of northern Canada, Greenland and Alaska. The three DUs are found entirely within Canada although the southern portion of the BP/GP was in the northern United States; Caribou disappeared from Wisconsin, Vermont, Maine, and New Hampshire by the turn of the 20th century (Banfield 1974).
Canadian Range
Newfoundland population
Caribou originally inhabited the entire island of Newfoundland, although three areas of higher abundance were identified in the early 20th century: the Humber River Valley; the central portion of the island south of the railway; and the Avalon Peninsula (Prichard 1910, cited in Banfield 1961). Twelve Caribou sub-populations were present before additional sub-populations were established through a series of relocations made in the 1960s-70s (Mercer et al. 1985). Up to 36 sub-populations have existed (Figure 1) but there appear to be approximately 14 sub-populations presently (Pardy Moores pers. comm.). Shifts in Caribou occupancy have been observed in some sub-populations; anecdotal evidence suggests that a small number of Caribou have begun to reoccupy areas (Newfoundland and Labrador Department of Environment and Conservation (NLDEC), unpubl. data 2013).
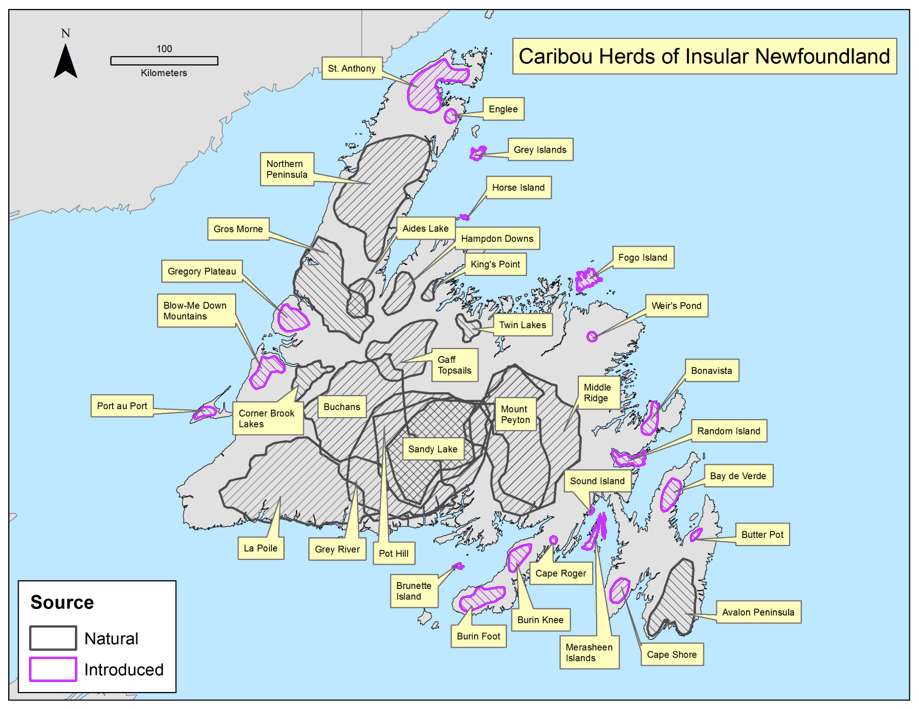
Photo: G. Kuehl © Environment Canada, 2015
Long description for Figure 1
Map of showing the distribution of 36 Caribou subpopulations throughout the island of Newfoundland during the 1990s. Areas containing naturally occurring subpopulations are distinguished from areas with introduced populations.
Gaspésie population
In the 19th century, Caribou were found in New England and the Canadian Maritime Provinces. Caribou were extirpated from Prince Edward Island in 1874, from Nova Scotia in 1925, and from New Brunswick in 1927 (Banfield 1974; Bergerud and Mercer 1989). COSEWIC (2012) reported from ATK that Nova Scotia Caribou had been adversely impacted by railroad establishment across migration routes. The southern limit of Caribou range gradually receded northward and the GP became the only remnant Caribou population south of the St. Lawrence River. In the mid-19th century, Caribou were irregularly present throughout the Gaspésie Peninsula, from the town of Gaspé to the east, to the Matapédia valley to the west, and south to the Baie des Chaleurs, an area spanning approximately 30,000 km2 (Guay 1983; St-Laurent et al. 2009). By 1953-1955, Caribou range was mainly confined to higher elevation areas.
The GP is currently found mostly on the McGerrigle and Chic-Chocs Mountains, and are largely confined to Gaspésie National Park (802 km2) (Figure 2). The proportion within the Park was > 75% in the 1950s (Moisan 1958), 91% during 1987-1992 (Ouellet et al. 1996), and 83% during 1998-2001 (Mosnier et al. 2003).
The GP may be divided into three sub-populations, each associated with different mountain summits: Mount Logan, Mont Albert, and the McGerrigle Mountains (Figure 2; see Dispersal and Migration section – Gaspésie population). The McGerrigle Mountains sector includes Mount Jacques-Cartier and Mount Vallières-de-Saint-Réal.
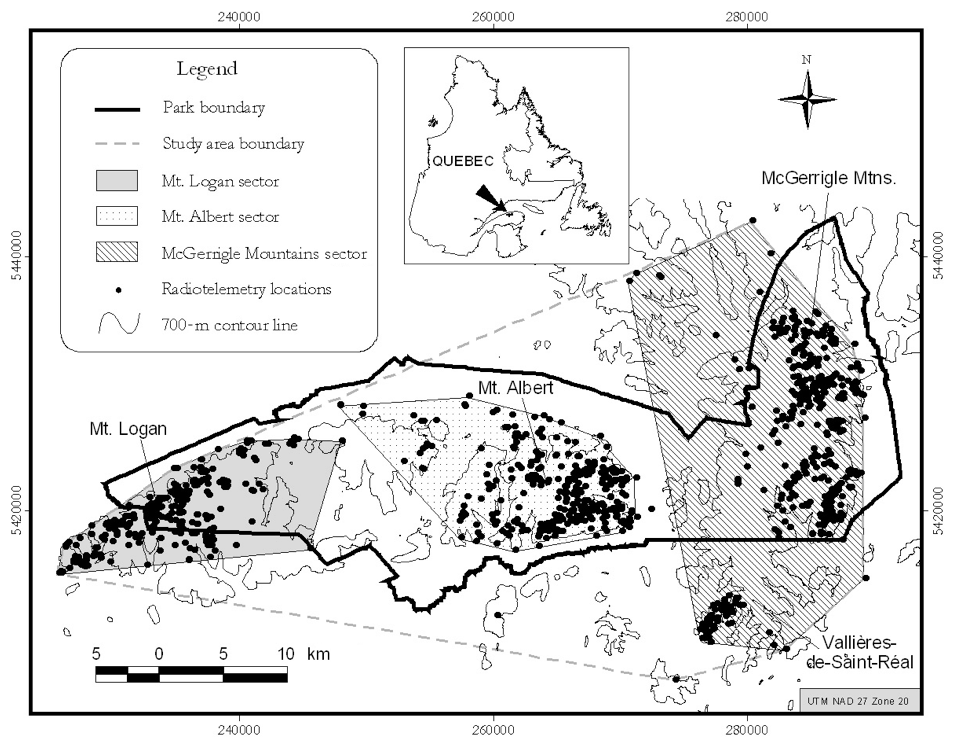
Long description for Figure 2
Map showing the distribution of Caribou in the Atlantic-Gaspésie population, much of which occurs in the Gaspésie National Park. Information on the map includes park boundary, study area boundary, radiotelemetry locations, and the areas covered by the Mount Logan, Mount Albert, and McGerrigle Mountains sectors.
Boreal population
Boreal Caribou occupy boreal forests from Labrador to eastern Yukon. In the northern portion of their range, the BP overlaps with the Eastern migratory (DU4) and Barren-ground (DU3) designatable units. For example, in northern Ontario, forest-associated Caribou have moved north, and tundra-associated Caribou have moved south during winter (Abraham et al. 2012; Wilson 2013). Boreal Caribou are found in three large ecozones including the Boreal Shield, the Boreal Plains, and the Taiga Plains ecozone (Wiken 1996). Together, these ecozones cover more than 3 million km2, stretch more than 4,000 km across Canada, and encompass more than one third of Canada’s land mass (Wiken 1996). In British Columbia (BC), Northwest Territories (NT), and the Yukon, the ranges of the BP adjoin with those of the Northern Mountain designatable unit (DU7).
Caribou are extirpated from half of their historic range in Canada in a pattern that mirrors the last century’s expansion of human settlement and resource exploitation (Laliberté and Ripple 2004). Caribou are extirpated from approximately 60% of their historic range in Alberta, 50% in Ontario, and 40% in BC (Hummel and Ray 2008). In Quebec, Caribou have been extirpated from the southern townships and north to the 50th parallel, except for a few isolated sub-populations (Courtois et al. 2003a). The Charlevoix sub-population, which had a population of 10,000 in the 19th century was extirpated by hunting and poaching by 1920, before a reintroduction in the 1960s (St-Laurent and Dussault 2012). In Ontario, range recession has been documented for over 80 years (De Vos and Peterson 1951), and corresponds closely to the geography of human activity, particularly forest harvest (Schaefer 2003). The process of extirpation noted in northwestern Ontario (Racey and Armstrong 2000) likely exemplifies the process of range recession for the DU. In northwestern Ontario, over 150 years, Caribou declined in phases-- there was a period of mature forest loss from large fires, forestry and agriculture, which resulted in remnant populations persisting in relatively inaccessible areas (e.g. poor quality soil, limited river access to remove timber). In the 1950s, forest access roads into these areas facilitated tree harvest, Wolf and human activity, and the spread of White-tailed Deer (and presumably Meningeal Brainworm, Parelaphostrongylus tenuis, which kills Caribou; see Threats – Problematic Native Species section). Ranges of the BP in Ontario have receded approximately 34 km per decade and the estimated time to extirpation, inferred from a sustained rate of disappearance, is 91 years (95% CI: 57-149 years) (Schaefer 2003).
The rate of this northward recession is not constant across BP range in Canada. Using range maps in COSEWIC (2011), the authors estimated the range contraction based on the historical distribution limit (estimated at 1850) and the current distribution limit (estimated at 2010). For five provinces, starting from Québec to Alberta, the authors computed northward recession rates of 36 (QC), 28 (ON), 14 (MB), 8 (SK), and 11 (AB) km per decade, respectively (BC was not included in this analysis). The northward BP recession has progressed at a greater rate than what would be expected from climate warming alone; on average, the loss of Caribou range attributed to climate change is approximately 6 km per decade (Parmesan and Yohe 2003). The observed range recession (8-36 km per decade) is more plausibly linked to an increase in anthropogenic disturbances (see Threats section). Range recession continues, with Caribou recently confirmed as absent in the Swan-Pelican Lakes area, Manitoba (MBWCMC 2014).
As the range retracted northward, populations of Caribou became separated from contiguous Caribou range in Québec (e.g., Charlevoix and Val-d’Or), in Ontario (e.g., Lake Superior Coast Range, including Slate Islands, Michipicoten Island), and in Alberta (e.g., Little Smoky).
Extent of Occurrence and Area of Occupancy
The smallest range is the GP, which is estimated at 1500 km2 (EO) and <1000 km2 (IAO). The NP EO is much of the island, at 112,000 km2. Combining telemetry locations (11,656 blocks or 13%) with “known caribou locations” (23,421 blocks or 27%) equals IAO of 35,077 blocks, or 44,781 km2, approximately 40% of the island of Newfoundland. The BP EO encompasses much of Canada, at approx. 3 million km2, and an IAO nearly as large at 2.45 million km2.
Search Effort
The overall distribution of Caribou is well known. Delineating ranges for a highly mobile species like Caribou can be a difficult and expensive task. Consequently, budget constraints and large range size may preclude identification of some Caribou population ranges. The apparent large sizes of some Manitoba or Québec Caribou ranges should not be taken as a biological entity but may be simply an indication that more studies have yet to be conducted in these large, continuous ranges. For example, all BP Caribou in NT are delineated in a single distribution of 44.1 million ha, while the size of the Val-d’Or range is 347,000 ha (Environment Canada 2012). Five ranges were newly delineated in Ontario in 2012 and they reflect geographical units used for assessment purposes (improved conservation units), rather than discrete population ranges (Elder pers. comm.).
The NP and GP are more readily delineated because they occur in relatively smaller areas and the high proportion of radio-collared animals has facilitated documentation of movement and distribution.
Although recent studies have improved our understanding of the spatial ecology of Caribou, most research has focused on females in order to find calving areas; relatively less is known about adult male space use.
Newfoundland population
The NP is one of the best-studied DUs in Canada (Mahoney and Weir 2009). Information on distribution, abundance, and annual movement has been collected since 1902 (Bergerud 1971), while scientific surveys began by the 1950s. In 1996, the Caribou Data Synthesis Project was initiated to centralize, compile, and evaluate all Caribou-related data from the 1950s to the 1990s. In the early 2000s, population monitoring revealed low recruitment, which resulted in calf mortality research in 2003, followed by research on adult mortality and distribution. Current efforts to monitor the abundance of the NP involve mark-resight aerial surveys for each major sub-population at least once every 4 years.
Gaspésie population
The GP were studied in the 1950s (Moisan 1958; Bergerud 1973). Autumn aerial surveys have been conducted since the early 1970s in order to assess population size, as well as determine sex and age ratios. Aerial surveys have been conducted in the Mount Albert and McGerrigle sectors since the early 1970s, but more rigorous methods have been applied since 1983. Surveys in the Mount Logan sector began in 1997.
Radio-tracking of the GP was initiated between 1975 and 1977 using aerial surveys (Georges et al. 1976; Rivard 1978). From 1987-1992, 28 Caribou (mostly adult females and some calves) were monitored to study space and habitat use during a period of low recruitment attributed to high predation (Ouellet et al. 1996). Another 35 VHF radio-tagged adults of both sexes were monitored from 1998-2001 (Mosnier et al. 2003), and monitoring of 43 GPS radio-tagged adults began in 2013 (St-Laurent pers. comm.).
Boreal population
Relatively little scientific research was conducted on the BP prior to the 1990s, but hundreds of papers and reports have since been produced. Research has often focused on the interrelationships between anthropogenic and natural landscape disturbance, predation, survival, and habitat use. Much of the last decade’s research was conducted in Alberta (e.g., Dyer et al. 2002; Latham et al. 2011b), Québec (e.g., Courtois et al. 2008; Fortin et al. 2008; Briand et al. 2009; Moreau et al. 2012; Lesmerises et al. 2013), and Ontario (OMNR 2014), but many projects are underway in most jurisdictions.
In Labrador, Schmelzer (2013) compiled a comprehensive synthesis of demographic data collected on three Caribou sub-populations since the 1970s. In Québec, research has quantified relationships between Caribou habitat selection, distribution, and anthropogenic disturbance (e.g., Courtois et al. 2002, 2008; Faille et al. 2010; Renaud et al. 2010; Fortin et al. 2011, 2013; Moreau et al. 2012; Pinard et al. 2012; Lesmerises et al. 2013). Data from radio-tagged Caribou during 2004-2011 were used to delineate local population ranges and examine habitat selection in the Cree territory in Québec (Rudolph et al. 2012).
In Ontario, most research since the 1990s has addressed conservation and management priorities. Radio-collaring had focused on individuals at the southern margins and central core of the continuous Caribou range (e.g., Brown et al. 2003; Elder pers. comm.). Shuter and Rodgers (2010) used movement data derived from 73 radio-collared Caribou from 1995-2008 to delineate demographic units, particularly along the southern range. More recent research has occurred across BP range in Ontario. A major collaborative research programme is underway among universities and government to evaluate alternative hypotheses regarding mechanisms that influence probability of Caribou persistence, such as apparent competition (see Threats section), predator road use, prey escape, sensory disturbance, and energetics/nutrition (Elder pers. comm.; OMNR 2014). Study areas were selected for key habitat attributes (e.g., conifer cover, linear feature density, Wolf (Canis lupus) and Moose (Alces alces) density) (Rodgers et al. 2009). GPS telemetry data was being collected from 423 Caribou and 56 Wolves during 2009-2013 (Shuter pers. comm.). Extensive fecal surveys and systematic occupancy surveys have occurred over much of the range since 2009 (Elder pers. comm.; OMNR 2012).
Caribou research in Manitoba and Saskatchewan focused on range use, distribution, and movements (e.g., Rettie and Messier 2000; Arsenault and Manseau 2011; Arlt and Manseau 2011). Research in Manitoba has integrated genetic techniques to delineate local ranges and quantify genetic population structure (Ball et al. 2007; 2010, Galpern et al. 2012; Hettinga et al. 2012). Habitat analysis and landscape connectivity modelling have been studied in Manitoba and Saskatchewan (e.g., O’Brien et al. 2006; Fall et al. 2007; Koper and Manseau 2009; Galpern and Manseau 2013a, b). There has been long-term monitoring of Caribou in the Owl Lake region (Brannen pers. comm. 2014). Manitoba Hydro has collaborated with Manitoba Conservation since 2007 in the collaring and monitoring of Caribou in ranges intersected by a transmission power line project; a radio-collaring study was initiated in 2007 and expanded in 2010 (78 radio-collars) and 2011 (70 radio-collars) (Joro Consultants Inc. 2011). Rettie and Messier (2000) radio-monitored 40 adult female Caribou in the southcentral portion of their range in Saskatchewan and delineated seasonal movements, habitat selection, and assessed vital rates.
In Alberta, vital rates (e.g., adult female survival, calf recruitment, finite rate of population growth, calf:cow ratios) and movement data have been collected from radio-collared Caribou since 2001 (Alberta Caribou Committee). A 4-year research program was initiated which studied relationships between Caribou, Wolf, Moose, and Grizzly Bear (Ursus arctos) using GPS collaring and data collection (Decesare et al. 2012).
In BC, a Caribou monitoring project was conducted in 2008 and 2009 using radio-telemetry (Thiessen 2009), with further monitoring of 160 collared caribou throughout provincial Boreal Caribou range since 2012 (Culling and Culling 2013).
Since 2002, NT has radio-collared > 180 Boreal Caribou. Vital rates (e.g., adult female survival, calf recruitment, finite rate of population increase) and movement data have been collected from collared Caribou from four different study areas: Gwich’in North, Gwich’in South, South Slave, and Dehcho (Nagy 2011; Kelly and Cox 2001; Nagy et al. 2011; Larter and Allaire 2014). Location data from collared female caribou were used to assess demographic and behavioural responses of Caribou to anthropogenic disturbance and to define secure habitat (Nagy 2011).
Habitat
In this report, habitat is defined as the vegetative structures (e.g., old coniferous forest), as well as factors that influence survival and productivity (e.g., predation levels). The Habitat section is divided into habitat use and habitat avoidance components because avoidance of certain habitats is pronounced in Caribou. There is strong agreement on which factors constitute important habitat to Caribou among ATK and Western science.
Habitat Requirements
Caribou habitat selection is complex; it occurs at multiple spatial and temporal scales, and the avoidance of predation appears to dictate habitat use more than food availability. The emphasis on predation is integral to Caribou management because predator densities increase with anthropogenic and natural disturbances (Bergerud 1974; Vors et al. 2007; Wittmer et al. 2005; Bastille-Rousseau et al. 2012). Separation of proximate from ultimate factors is difficult, and there is variation in behaviour within each DU. Caribou will select small sites to be used for short periods annually (e.g., nursery areas), as well as large areas used for generations (e.g., seasonal ranges), but use of either can shift depending on changing forest conditions. In Manicouagan, Québec, Caribou use of residual patches of old forest stands increased as they became less available (Moreau et al. 2012). Selection also varies with spatial scale. For example, nursery areas contained relatively higher abundance of groundcover vegetation, terrestrial lichens, lower shrub density and higher densities of mature confer trees in northern Ontario, when compared to random sites, but at the landscape scale, nursery sites were in remote areas with overall high refuge value from predators and less forage (Lantin et al. 2003; Carr et al. 2011; Dyke and Manseau 2011; Pinard et al. 2012).
Habitat Avoidance
The ultimate factor in Caribou habitat use is predator avoidance, which is achieved by existing at low density, and by avoiding areas with predators, particularly Wolves. A ‘stabilizing density’ of 0.06 Caribou/km2 has been suggested as a threshold density wherever Wolf predation is the major mortality factor (Bergerud and Elliot 1986; Bergerud et al. 2008). At this density, Caribou can space themselves to balance recruitment and mortality (Schmelzer 2014). BP Caribou in Labrador are typically found at densities of 0.03-0.06 Caribou/km2 in core areas of their range and 0.004 Caribou/km2 in peripheral areas (Schmelzer et al. 2004; Bergerud et al. 2008). The Federal Recovery Strategy estimates an average Boreal Caribou density of 0.02 to 0.03 Caribou/km2 across BP range (Environment Canada 2012).
Caribou also minimize predation risk by “spatially separating” themselves from predators by using less productive, old forest habitat that does not support sizable Moose or deer (White-tailed [Odocoileus virginianus] or Mule [O. hemionus]) populations and, consequently, does not support dense predator populations (Bergerud 1974; Rettie and Messier 2000; Racey and Arsenault 2007; Environment Canada 2012; OMNR 2014). At larger spatial scales, Moose, deer, and large predators are more common in areas in deciduous forest containing large amounts of browse for ungulates (Hall 1984; Peek 2007). By contrast, during snow-free seasons, Caribou use barren lands, alpine tundra, islands in large lakes, and wetlands, all of which are sites that have relatively fewer predators and therefore reduced predation on calves (Mosnier et al. 2003; Carr et al. 2011; Schaefer and Mahoney 2013). Elders in Tłįchǫ (northeast of Ft. Simpson, NT), Little Red Cree River, and Tallcree (both in Alberta) First Nations note that Caribou prefer thick bush, particularly in winter when it is more difficult to remain camouflaged from predators, and that Caribou prefer to be near water in summer in order to access greater abundance of food, cool off, and to avoid insects and Wolves (WRRB 2013; Schramm and Krogman 2001).
Although logged areas and regenerating forest stands can be utilized for foraging in some areas (Hins et al. 2009; Hébert and Weladji 2013), Caribou have been found to generally avoid such areas by an average of 1.2 km in Alberta (Smith et al. 2000), 4.5 km in Québec (Fortin et al. 2013), and 9.2 km in Newfoundland (Schaefer and Mahoney 2007). Miller (2010) quotes Anishinaabe elders as stating that Caribou only enter cut blocks when being chased by Wolves. In Alberta, Caribou daily movement rates and individual winter range sizes decreased in response to timber harvest (Smith et al. 2000). Other patterns of habitat use include Caribou only using disturbed sites at nighttime (Beauchesne et al. 2013), and use of forest cutovers only when they were adjacent to mature residual stands (Briand et al. 2009; Hins et al. 2009).
A study in northwestern Ontario using tracks and pellets in winter concluded that Caribou and Wolves were spatially separated, while Moose and Wolves were not (Cumming et al. 1996). More recent work using telemetry over a larger area found Wolf territories were smaller in areas with higher Moose density, and Wolves selected habitat types that were selected by Moose (e.g., stands with deciduous or regenerating forest) and avoided some habitat types (e.g., dense conifer) that are selected by Caribou (Anderson 2012; Shuter pers. comm.). In a disturbed landscape in Alberta, Peters et al. (2013) reported a significant positive relationship between spatial overlap of Moose and Caribou, and the degree of human landscape alteration. They found that locations of Caribou mortalities corresponded with areas of high resource use by Moose in summer, suggesting that early successional forest stages may decrease spatial separation between Caribou and Moose, resulting in increased predation risk for Caribou. Traditional knowledge indicates that industry within BP habitat diminishes this spatial separation and alters predation rates on Caribou (Dehcho First Nations 2011, cited in COSEWIC 2012).
Forestry operations can directly remove lichen biomass available to Caribou (Fisher and Wilkinson 2005) but the limited use of recently disturbed sites likely is related mainly to predator avoidance. The abundance of forage within disturbed habitats can be attractive to Caribou and make them more prone to predation (Beauchesne et al. 2014) because Black Bears (Ursus americanus), Wolves, and Coyotes (Canis latrans) frequent cutovers (James et al. 2004; Kays et al. 2008; Mahoney and Virgl 2003; Mosnier et al. 2008b; Boisjoly et al. 2010; Bowman et al. 2010). Moose and deer populations thrive in disturbed areas and support larger Wolf populations than would be present if Caribou were the primary prey species (Bergerud and Elliott 1986; James et al. 2004; Latham et al. 2011b). In the Charlevoix sub-population in Québec, regenerating stands (6-20 years old) had the greatest ground vegetation cover, providing a high biomass of berries and other vegetation for Black Bear (Brodeur et al. 2008). Mosnier et al. (2008b) reported similar positive effects of logging on the Black Bear habitat in the GP range. Caribou selected forest habitat that provided seclusion from Black Bears and Wolves. Pinard et al. (2012) and Dussault et al. (2012) reported that variation in habitat selection tactics among females with calves resulted in different Black Bear predation risks for their calves. Recruitment rate is inversely linked to the proportion of early seral stage forest stands in managed landscapes (Environment Canada 2008).
In the NT and northern Alberta, Caribou avoided linear features such as seismic lines by 400 m (Nagy 2011). Travel rates were inversely related to seismic line densities; local Caribou movements may have become increasingly constrained as seismic line densities increased, and Caribou avoided seismic lines during periods when they were vulnerable to predation and/or harvest. In Alberta, Dyer et al. (2001) reported that female Caribou avoided areas within 1 km of well sites, and avoided areas within 250 m of roads and seismic lines. This avoidance behaviour was more pronounced in winter and lowest in summer. Wasser et al. (2011) reported that during winter, Caribou in northeastern Alberta did not avoid unused secondary linear features, but did avoid primary roads. Near Lake Nipigon, Cumming and Hyer (1998) recorded avoidance of a log haul road in use by 25 trucks working 24 hours/day, but not in the year before and after the road was active, suggesting the traffic was a concern and not the road. In Québec, Caribou strongly avoided road networks by at least 2 km (Rudolph et al. 2012; Dussault et al. 2012), and up to 10 km (Rudolph 2011). Tertiary forest roads are avoided by 750 m, primary roads by 1.25 km, and highways by 5 km (Leblond et al. 2011, 2013a). These metrics are comparable to a 4 km threshold of road avoidance in Ontario (Vors et al.’s 2007), and a 4.5 km avoidance in Québec (Fortin et al. 2013). Leblond et al. (2013a) estimated a zone of influence (ZOI) of 5 km from roads and reported that within this ZOI, Caribou avoided habitat types that were otherwise selected at the home range scale, and displayed higher movement rates, especially when traffic density was high.
The avoidance of linear features appears to be related to predation risk. Seismic lines (McLoughlin et al. 2003; Latham et al. 2011a) and roads (Whittington et al. 2011) may be used by Wolves as travel routes (which facilitate access into once-secluded Caribou habitat), can provide barriers to Caribou movement (Dyer et al. 2002), and change encounter rates between Caribou and their predators. In the NT, predation mortalities were closer to linear features (Larter and Allaire 2014) than locations of live Caribou (James and Stuart-Smith 2000). Computer simulations suggested that Wolf-Caribou encounters increased with the density of linear disturbances in Caribou habitat (McCutchen 2006; Whittington et al. 2011).
Anthropogenic disturbances cause direct loss of Caribou habitat through range modification. Noise pollution from trucks and other industrial activity may drive Caribou away from areas actively used by humans (COSEWIC 2012). Female Caribou establish nursery areas in areas with lower levels of sensory disturbance from development or recreational activities (Carr et al. 2007; Schaefer and Mahoney 2007; Vors et al. 2007; Vistnes and Nellemann 2008). A critical threshold of 10-15 km from disturbance was recommended for parturient Caribou (Carr et al. 2011). Caribou may also experience chronic stress and negative energetic consequences when fleeing disturbances (Bradshaw et al. 1998). Individuals have been found to become confined into smaller, less productive habitats if disturbance is severe (Beauchesne et al. 2014). Caribou often show diminished use of areas within a 1-10 km radius of these disturbed landscapes (Duchesne et al. 2000, Mahoney and Schaefer 2002a; Cameron et al. 2005; Vors et al. 2007; Weir et al. 2007; Hins et al. 2009; Faille et al. 2010; Mahant 2013).
The probability of remaining and surviving in an area decreases with the quantity and severity of the disturbance (Vors et al. 2007, Environment Canada 2011). Lesmerises et al. (2013) reported that in landscapes with small patches (<100 km2) of habitat, Caribou became concentrated, which increased their vulnerability to predation and acted as ecological traps. To increase the likelihood of Caribou occurrence, they found that core forests must be greater than 1000 km2 in size and not surrounded by a dense network of roads, cutovers, and cabin developments.
Habitat Use
Selection for rich lichen feeding areas, particularly during winter when lichen may be the only forage available, can be an important driver of Caribou distribution within the boreal forest (Mayor et al. 2009). In Ontario, ‘Winter Use Areas’ are associated with soil and forest cover conditions that provide abundant ground lichen (Cladina and Cladonia species) (OMNR 2014). Ground lichens are low in protein but are an important winter source of carbohydrates (Schaefer and Pruitt 1991). Caribou may dig through snow to access terrestrial lichens, or forage on arboreal lichens that grow on old trees. Mature and old coniferous forests generally have shallower snow and less crust compared to open areas and are thus used for access to lichens, and as shelter from harsh winter conditions (Mosnier et al. 2003; Ferguson and Elkie 2004; Mayor et al. 2009).
Mature and old coniferous forests are generally recognized as important habitats for Caribou and are utilized throughout the year (Festa-Bianchet et al. 2011; Bastille-Rousseau et al. 2012). It is difficult to assess how old a stand should be to provide adequate habitat to Caribou but research in Ontario suggests that Caribou will use natural stands aged ≥ 50-60 years (Elkie et al. 2009). In the Assinica region (Québec), Caribou occur in areas with a fire cycle of 67 years (Girard pers. comm. 2014). Caribou demonstrate habitat selection in response to the natural heterogeneity of the boreal forest (e.g., tree age and species composition) (Racey and Arsenault 2007). For example, Caribou will use smaller, discrete patches of young forest and hardwood forest, provided these sites are near larger patches of conifer-dominated forest (Elder pers. comm.).
The size of forest stands also is important. Lesmerises et al. (2013) demonstrated in Québec that forest stand size must reach approximately 270 km2 to attain a 50% probability of use by Caribou. They concluded that a range composed of stand sizes from 100 to 250 km2 is too small to support BP Caribou, and that the matrix composition and structure of the forest is important. Nagy (2011) hypothesized that Caribou in the NT can be sustained in areas where ≥46% of the land is unburned habitat found in patches >500 km2 in a relatively pristine range, and with low predator and alternate prey diversity. These habitat components may be important for a number of activities, including foraging, calving, and insect relief, or simply to provide connectivity among preferred habitats (Nagy 2011).
Newfoundland population
The habitats available to the NP are composed of a mosaic of small to extensive patches of coniferous forests, barren lands, shrub lands, and bog/wetland complexes. Winter snow conditions strongly influenced Caribou habitat selection in the central portion of the island, and coniferous forests were important habitats all year (Hébert 2012). Wetlands, barren lands, and shrub lands are generally preferred during snow-free seasons, and mature-old coniferous forests are used in winter to minimize energetic stress related to snow accumulation (Mahoney and Virgl 2003; Wells et al. 2011). Females tend to migrate and aggregate at traditional calving grounds, characterized as barrens surrounded by contiguous coniferous forests and broken by areas of wind-scarred trees or old coniferous stands, from April to mid-May (Saunders 2007). Females of the Middle Ridge sub-population have a stronger preference for bogs than do males, which may serve to reduce calf predation risk (Schaefer and Mahoney 2007). In winter, female Caribou of the same sub-population selected high terrestrial lichen abundance at a broad scale and, within lichen-rich areas, selected microsites with soft and shallow snow (Mayor et al. 2007, 2009).
Fecal samples from 1990-1997 and 2010-2011 showed that NP consume a wide variety of plants throughout the year, with terrestrial lichens being a large portion (33% in summer, and 62% in autumn) and arboreal lichens being relatively rare in NP diet (< 4%) (Soulliere and Mahoney 2014). Despite this use, terrestrial lichens are not thought to be a limiting food resource in Newfoundland (Humber et al. 2009).
While some of the sub-populations occupy relatively undisturbed landscapes, others occupy areas disturbed by industrial forestry, access development, and other anthropogenic footprints. Some sub-populations avoid recently harvested areas, and females with calves displayed heightened sensitivity (Chubbs et al. 1993; Mahoney and Virgl 2003; Schaefer and Mahoney 2007). Areas with timber harvesting were associated with reduced calf recruitment rates for NP Caribou (McCarthy et al. 2011).
Gaspésie population
The GP extensively use alpine habitat on mountain plateaus and mature Balsam Fir (Abies balsamea) and spruce stands on forested mountain slopes at >700 m asl (St-Laurent et al. 2009). Alpine habitats are sometimes abandoned when snow conditions impede access to forage (Ouellet et al. 1996). During summer, GP often use high altitude areas where stronger winds and remnant snow patches provide relief from insect harassment (Boileau 1996). Caribou also congregate on open habitats at high elevations during rut, which facilitates mate access (Bergerud 1973; Ouellet et al. 1996). In the spring, females with calves remain at high elevations consuming winter forage despite availability of new green forage at lower elevations; this may reduce risk of calf predation (Ouellet et al. 1996).
During winter, the GP may seek mature Fir stands to access other winter forage, such as arboreal lichens (Boileau 1996; Mosnier et al. 2003), which can comprise up to 53% of GP winter’s diet (27% in summer, St-Laurent et al. 2009). Arseneau et al. (1997) estimated the arboreal lichen biomass (primarily Usnea spp., Alectoria sarmentosa, and Bryoria spp.) in the GP range in three altitudinal belts ranging from 720 to 1068 m, and reported biomasses of 1306, 150 and 11 kg/ha for the mountain (<900 m), subalpine and alpine (>1000 m) belts, respectively. Within the coniferous forest, dense snowpack can facilitate Caribou access to higher arboreal lichens, thereby increasing forage availability. Mosnier et al. (2003) found that GP selected forest patches that had more arboreal lichen, larger trees, and harder snowpack. Terrestrial lichens are relatively absent in GP range and are rarely consumed (St-Laurent pers. comm.).
Boreal population
Local variations in habitat selection occur within ecozones (Environment Canada 2011), but BP habitat consists chiefly of mature or old forest (i.e., >80 years old) spruce (Picea spp.) and pine (Pinus spp.) stands intermixed with peatlands and muskegs, with abundant lichens and low predator densities (O’Brien et al. 2006; Brown et al. 2007; Courtois et al. 2007). In northeastern BC, Caribou primarily use large peatlands throughout the year, and closed-canopy mature Lodgepole Pine (Pinus contorta) and Black Spruce (Picea mariana) stands during late winter and periods of high snow accumulation (BC Ministry of the Environment 2010). In Ontario and Québec, females used habitats which are composed primarily of mature or old coniferous forest (Brown et al. 2003; Courbin et al. 2009). The BP caribou generally do not use recent burns, perhaps due to low lichen availability (Rettie and Messier 2000; Vors et al. 2007; Sorensen et al. 2008). In BC, recent burns were used during the spring for access to non-lichen forage (Boonstra and Sinclair, cited in Fisher and Wilkenson 2005). BP wintering locations vary little from year to year (Cumming et al. 1996; Ferguson and Elkie 2004), and typically consist of lichen-rich ranges characterized by shallow snow (Barrette and Vandal 1986; Courbin et al. 2009; Moreau et al. 2012). Site fidelity decreased in areas disturbed by natural or anthropogenic events (Faille et al. 2010).
Habitat Trends
The section below outlines trends in habitat that have occurred recently and typically are ongoing. The Threats section of the report outlines present threats, many of which relate to change in habitat, but also predictions of the future change in habitat.
A) Change in Structural Habitat
The response of Caribou to forestry seems to be similar across the species’ range, likely because clear-cut harvest is common within the range of each DU. An important aspect of forestry is the temporal nature of the threat. Widespread re-occupancy of clear-cut sites has not been documented (Wilkinson 2010) but succession in disturbed forests will result in increased food and cover over time, suggesting Caribou should reoccupy clear-cut harvested sites. Some Caribou recovery plans and forest management plans rely on harvested areas to eventually become Caribou habitat (see Habitat Protection and Ownership section). In Ontario, for example, three logged sites clear-cut between 1952 and 1970 were replanted with pine or spruce and are now used by Boreal Caribou in both summer and winter, a finding in large part attributed to the refuge habitats that remained in the vicinity (Racey et al. 1996; Racey et al. 2010; Rose and Racey 2010; OMNR 2012). Reoccupation of cutovers can occur if the cutovers regain characteristics similar to the original stand (Fisher and Wilkinson 2005; Faille et al. 2010) but not all cutovers return to similar condition; Hébert and Weladji (2013) showed cutovers (>40 years) did not develop into forests with similar stand characteristics as the coniferous forests selected by Caribou in central Newfoundland. The regenerated canopy in the cutover sites was more closed, and supported less forage, than the uncut coniferous forests.
Reoccupancy may also be influenced by predation, at least in the BP. Caribou may be absent from older cuts if Wolf and Moose densities remain higher (Boertje et al. 1996; Rettie and Messier 2000), and lichen availability is lower (Johnson et al. 2004). Vors et al. (2007) reported that the Caribou inhabiting areas within 13 km of clear-cuts in Ontario became extirpated within 20 years. Faille et al. (2010) showed that Caribou displayed high fidelity to logged areas, which created an ecological trap of increased predation. Vors et al. (2007) reported that Caribou were extirpated within 20 years from areas within 13 km of clear-cuts. In summary, reoccupancy is difficult to predict, and given the multiple scales and factors related to occupancy by Caribou, it is likely that use of any particular harvested area is associated with the amount and configuration of forest at larger scales that retained Caribou, as well as predator densities.
Newfoundland population
Habitat use by the NP changed when the decline began after the 1990s. A study reviewing 30 years of habitat selection data of one NP sub-population (i.e. Middle Ridge) found that Caribou shifted their habitat preferences by avoiding cutovers as well as open and closed forests, and instead selecting barrens, shrubs and wetlands (Mahoney and Schaefer 2011). The changes in habitat use may be indicative of a response to predators, but also limited lichen abundance. The portion of moss in NP diets has increased since the 1990s and Horsetail (Equisetum sp.) did not appear in their diet until the 2010s. Increased tooth wear may have resulted from an overall decrease in diet quality, or may be related specifically to high silica content in horsetails. Preliminary analyses indicate reduced range quality since the peak of the NP (Soulliere and Mahoney 2014). Caribou are exhibiting density-dependent morphological responses, such as decreases in jaw bone size, birth weight, and antler quality, which are characteristic of low nutrient availability (Mahoney and Weir 2009; Mahoney et al. 2011; Weir et al. 2014). Some of these indices are returning to pre-decline levels (see Fluctuations and Trends section).
Caribou habitat may be adversely affected by the high density of introduced Moose. An overall density of approximately 1.3 Moose/km2 and a maximum density of approximately 15 Moose/km2 in local areas are some of the highest densities in the world (McLaren et al. 2004). High densities of Moose negatively affect the capacity of Balsam Fir to regenerate following canopy disturbance (Gosse et al. 2011). Overabundant Moose on the island are affecting the landscape by slowing the succession rate of the forest after a disturbance, which can influence Caribou habitat over the long term. However, at present there is no conclusive evidence that Moose are strongly impacting NP Caribou.
Gaspésie population
At the beginning of the 20th century, the pre-industrial forest of the Gaspésie region consisted primarily of >100-year-old stands of Balsam Fir and White Spruce (Picea glauca). Extensive forestry activity over the past 50-70 years changed mature forest structure into young forests (Lesmerises 2012) and the new forest generally did not retain all attributes of the natural ecosystem. For example, Boucher et al. (2009) concluded that 20th century logging practices strongly altered the eastern Québec region’s pre-industrial vegetation patterns to the point that ecosystem-based forest management strategies should be developed to restore conifer dominance, altitudinal gradients, as well as the irregular structure similar to old forest stands.
In the GP range, Stone et al. (2008) reported that arboreal lichen biomass increased slowly through time, with negligible biomass production <50 years and greatest biomass at 70-90 years in the Balsam Fir-dominated stands. They suggested that lichen biomass will plateau at the same time that trees are senescing, which is about 100 years.
Forestry within Gaspésie National Park was permitted from 1938-1977 and mining was conducted between 1943 and 1963 (St-Laurent et al. 2009). Natural resource exploitation continues within the Chic-Chocs Wildlife Reserve and Matane Wildlife Reserve, which are contiguous to Gaspésie National Park. Consequently, much of the surrounding habitat for GP is continually impacted by resource development. The disturbed landscape maintains high predator abundance, primarily of Black Bear and Coyote (St-Laurent et al. 2009).
The GP co-occurs with a high Moose population. In 2007, Moose density was estimated at 4.8 Moose/km2 in the Matane Wildlife Reserve (Lamoureux et al. 2007), a Moose density never reached before in Québec and which negatively affected regeneration of Balsam Fir following forest harvest (Gingras 2013). Moose may have a detrimental impact on the GP habitat via high levels of grazing, but also the transmission of parasites (see Threats – Problematic Native Species section).
Habitat use by female Caribou, particularly those with calves, has changed over recent decades. GP are not showing seasonal differences in habitat use; they remain on mountain summits during the spring, where the likelihood of predator encounters is low, predator detection is high, but nutritious forage is scarce (Ouellet et al. 1996). The GP have demonstrated increasing use of open habitats since the 1970s. Ouellet et al. (1996) reported that 31% of the radio-locations were found in alpine habitat between 1975 and 1977, compared with 45% between 1987 and 1992. During 1987-1992, 43% of summer and 62% of autumn locations were in alpine habitat (Ouellet et al. 1996), compared to 65% (summer) and 70% (autumn) of locations from 1998 to 2001 (Mosnier et al. 2003). Increased use of alpine habitat could be an anti-predator strategy but changes in snow conditions, disturbances in areas located outside the park, avoidance of linear features within the park, or changes in interspecific competition may also explain these changes in habitat use. Escaping toward higher ground may also be a parasite avoidance strategy to space out from Moose and White-tailed Deer that may carry Meningeal Brainworm (Kutz pers. comm.).
Boreal population
Forestry, roads, utility corridors, petroleum infrastructure, multi-use trails, and mining all have been recorded as having negative impacts on BP habitat (e.g., James and Stuart-Smith 2000; Rettie and Messier 2000; Dyer et al. 2001; 2002, McLoughlin et al. 2003; Vors et al. 2007; Arsenault and Manseau 2011; Latham et al. 2011a; Dussault et al. 2012; see Threats section). Disturbed habitat is defined in the National Recovery Strategy (Environment Canada 2012) as habitat showing: i) anthropogenic disturbance visible on Landsat at a scale of 1:50,000, including habitat within a 500 m buffer of the anthropogenic disturbance; and/or ii) fire disturbance in the last 40 years, as identified in data from each provincial and territorial jurisdiction (without buffer) (Environment Canada 2012). The greatest industrial disturbance footprint in BP range occurs in the southern boreal regions across Canada (Figure 3). In Alberta, all BP ranges are highly disturbed (range: 57-95%, Environment Canada 2012). In BC, Thiessen (2009) reported high disturbance levels (range: 58-87%) caused especially by oil and gas development. Less is known about Caribou in Saskatchewan, and in some regions of Manitoba and Ontario (Environment Canada 2012). In the NT, the BP range is less disturbed (31%), and most disturbance (24%) is caused by fires (Environment Canada 2012). At least half of BP range across northern Ontario, Québec, and Labrador remains relatively undisturbed from anthropogenic sources. Figure 3 illustrates a typical spatial pattern of anthropogenic disturbance in the southern Caribou range, and natural disturbance from fire and blowdown in northern parts. In eastern Canada, the BP range is receding northward, while in western provinces, it is becoming highly fragmented and receding northward.
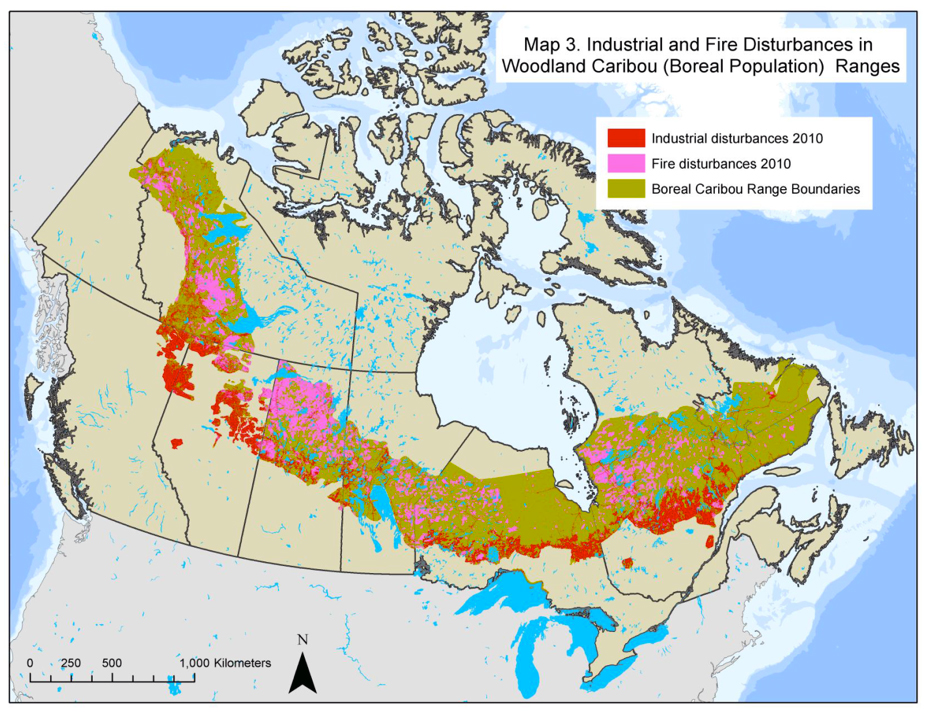
Long description for Figure 3
Map illustrating the location of anthropogenic (forest harvest, mining, roads, with 500-metre buffers) and natural (fire, blowdown) disturbance in remaining range of the Boreal population (BP) of Caribou as of 2010. The greatest industrial disturbance footprint in BP range occurs in the southern boreal regions across Canada.
B) Change in Predation Rates
Predation rates can increase to unsustainable levels when changes in forest habitat increase Moose or White-tailed Deer populations, or new predators arrive. Predation rates on Caribou can increase after land use change (e.g., roads, forestry) because of increased access (see Threats section) but also because of increased predator densities due to the arrival of alternative prey. The term ‘apparent competition’ describes population decline of one prey species concurrent with population increase of another prey species, not because of direct competition for a shared food resource, but because of a shared predator (Holt 1977). Apparent competition begins when natural and/or anthropogenic disturbances alter forest structure into ideal habitats for Moose and deer (Latham et al. 2011b). In turn, these ungulates support higher predator populations than would Caribou alone, given their younger age at first reproduction and multiple births, and Caribou suffer more from predation impact. Typically, the novel prey species (i.e., Moose or deer) is a habitat generalist with high reproductive output, and predator numbers do not respond numerically to declines of the original prey species (i.e., Caribou) (Holt 1977). The resultant inverse density-dependent relationship between predator and original prey can result in local extirpation of Caribou, or may suppress it well below carrying capacity.
There are alternative hypotheses from apparent competition for Caribou declines. It is possible that predation rates have increased because linear features, such as roads, seismic lines, and hydro corridors have increased the efficiency of predator foraging. It is also possible that Caribou populations are more vulnerable to predation because Boreal Caribou are being confined to a restricted number of undisturbed areas (Elder pers. comm.). Research is underway in Ontario to assess these hypotheses, as well as identify potential additive and interacting factors in Caribou declines (OMNR 2014).
Newfoundland population
Predation accounted for 59% of calf deaths from 1979-1997 (Mahoney and Weir 2009), and 90% between 2003 and 2012 (Lewis and Mahoney 2014). The role of apparent competition is unknown in the NP but it is not considered to be as significant as it is in the other DUs. It is unknown whether forest harvest has increased Moose populations in NF, and secondly whether predator density has increased because of Moose. There is evidence that Coyote and Black Bear consume a great deal of Moose meat, primarily as carrion. However, it is not clear if Moose carrion has an influence on population levels of Coyote and Bear (Pardy Moores pers. comm.; ENVC unpubl. data).
Our understanding of the impact of apparent competition is based mainly in areas where Wolves are the primary predator on Caribou, but Wolves are absent from the NP and Coyote do not appear to be depredating large numbers of adult Caribou. Of 730 neonate calves radio-tagged from 2003-2012, 57% died from predation within 6 months (Lewis and Mahoney 2014). Most (90%) of mortalities were due to predation. Cause of death was Black Bear (34%), Coyote (28%), Bald Eagle or Lynx (16%), predator not determined (16%), and remaining due to starvation, accident, or orphaning. Coyotes reached Newfoundland in 1985 (Mahoney and Schaefer 2002b). Lynx were historically a main predator of calves (Bergerud 1971) but are less so with the arrival of Coyote; it is possible that Coyote depredation is compensatory to Lynx depredation. There is little evidence that Coyote are significant predators of adults in winter, with only 18 of 424 radio-collared adults killed by Coyote between 2004-2011(Lewis and Mahoney 2014). Adult survival rates pre- and post-Coyote colonization are not significantly different, suggesting that Coyote predation is not additive to other causes of adult mortality (Weir et al. 2014).
Gaspésie population
Wolves were extirpated from the region by 20th century and the smaller Coyote was established in the 1980s (Crête and Desrosiers 1995). Although Coyotes are smaller than Wolves, Coyote predation on GP Caribou is significant enough to affect population abundance, and Coyotes are increasing with disturbance (see Threats – Problematic Native Species section).
Boreal population
Apparent competition is considered a primary contributor to the decline of Caribou in parts of BP range (Stuart-Smith et al. 1997; Rettie and Messier 1998; Schaefer et al. 1999; Vors et al. 2007). Apparent competition in Wolf-Caribou-Moose systems is well studied in western Canada, but less so in eastern Canada.
Wolves are considered the primary predator of Caribou in the BP range (Rettie and Messier 2000; McLoughlin et al. 2003; Wittmer et al. 2005). Wolves may actively seek the less competitive prey species (e.g., Caribou) (Tremblay-Gendron 2012) while Black Bears are more opportunistic predators (Bastille-Rousseau et al. 2011). Wolves are predicted to cause population decline in Caribou population if their density is > 6.5 wolves/1000km2 (Bergerud and Elliot 1998).
Caribou persistence in the boreal forest will depend on whether their space needs for predator avoidance are met (Bergerud 1985, 1988). Smith et al. (2000) reported that if fragmentation of Caribou winter range in Alberta continued via timber harvesting and other industrial activities, the “spacing out” anti-predator strategy used by Caribou may be compromised.
In parts of the BP range in Alberta, Wolf densities have increased from 6 wolves/1000km2 in 1994-1997 (James et al. 2004) to 11.5 wolves/1000km2 in 2001-2009. Caribou declines accelerated during higher Wolf density (Hervieux et al. 2013). Changes in Wolf density in other parts of the range are not available.
No published studies on Caribou reoccupying disturbed areas that contain high predator and alternative prey populations are known. Apparent competition may irrevocably compromise the ability of Caribou to spatially separate themselves from predators and alternative prey (Losier 2013; Peters et al. 2013).
Biology
Life Cycle, Demographic Parameters and Reproduction
Maximum recorded longevity is 22 years in captivity (Müller et al. 2010) and estimates of 19.5 (Schmelzer pers. comm.) and 17 (Larter and Allaire 2014) years have been made for wild BP Caribou, but few males and females exceed 10 years and 15 years, respectively (Thomas and Kiliaan 1998; Larter and Allaire 2014). Age structure within a Caribou population may change over time because survival and fertility rates within each age class change over time. For example, the average age of females in NP was 3.8 years in 1980, and 6.2 years in 2008 (Lewis and Mahoney 2014). Generation length in this report is based on the average age of parents within the population and therefore reflects the turnover rate of breeding individuals in a population (IUCN Standards and Petitions Subcommittee 2013). For the NP, average survival and fertility rates for 2004 and 2010 were 0.86 and 0.77, respectively. Generation length was estimated at 6.2 years for the NP, assuming that senescence starts at 12 years for survival and 9 years for fertility, and maximum life span is 17 years (Neville, J., NLDEC, unpubl. data 2013). A generation length of 6 years is used in this report. Generation lengths are not calculated for the GP and BP but are likely to be similar to the 6 years.
Caribou populations are usually female-biased, possibly due to preferential sport hunting of males in some areas. Bergerud (1980) reported a mean sex ratio among adults of 36 males per 100 females for the GP, 32 for ‘interior sub-populations’ in NP, 40 for the NP Avalon sub-population, and 40 for the BP Pukaskwa sub-population in Ontario. Hettinga et al. (2012) estimated a sex ratio of 76 males:100 females for Manitoba’s North Interlake BP sub-population. In NP, adult sex ratio has become increasingly female-biased since the 1960s (Mahoney and Weir 2009; Weir et al. 2014). Harvest rate of males was estimated at 16% during the 2000s. The proportion of males in the population has been increasing since 2009 with the adoption of ‘either sex’ licences, which reduced hunting pressure on males (NLDEC, unpubl. data 2013). In the non-hunted GP, sex ratio has been near equal since 1983 (Lalonde pers. comm. 2014).
Females first produce young between 24-48 months, depending on quality of range (Bergerud 1971; Crête et al. 1996; Larter pers. comm.). Male Caribou can be sexually active at 2 years, but usually > 4 years of age. In the NT, BP female Caribou have produced calves between 2-16 years of age (Larter and Allaire 2014). Caribou give birth to a single offspring. Parturition is highly synchronized within a Caribou population but varies geographically; for example, parturition peaked in mid-May in BC (BP) (BC Ministry of Environment 2010), mid-late May in NT (BP) (Nagy 2011; Larter and Allaire 2014), end of May in Newfoundland (NP) (Bergerud 1975), and early to mid-June in Labrador (BP) (Schmelzer 2014).
Caribou are polygynous; males gather females into harems, though not as pronounced as in other gregarious ungulates, and tend to guard several females at a time and prevent other males from approaching (Kelsall 1968; L’Italien et al. 2012). The rutting period for the NP was mid-October (Bergerud 1975) and late September to mid-October in the BP Labrador range (Schmelzer 2014). In the GP, the rutting period was first three weeks of October (Bergerud 1973).
Gestation length for Caribou is 215-230 days (McEwan and Whitehead 1972; Bergerud 1975). Females may conceive in alternate years when foraging conditions are poor, or a female’s body fat and protein reserves are diminished from rearing previous offspring (Gerhart et al. 1997). Pregnancy and productivity rates are usually high (75-100%) within the BP (Bergerud 1974; Nagy 2011; Rettie and Messier 1998). In the NP, productivity ranged from 70-100% from 1960 to 2012, with most years falling between 70-90% (Weir et al. 2014). Productivity in the NP has displayed minor variability, especially when compared to other life history parameters (e.g., abundance, mean age, calf survival, etc.). Pregnancy rates for the GP ranged between 60% (in 2013) and 89% (in 2014) (M.-H. St-Laurent, unpubl. data).
Physiology and Adaptability
Caribou often experience marked seasonal fluctuations in body fat and protein reserves, which reflect differences in forage quality and energetic stressors (e.g., deep snow, insect harassment) (Barboza et al. 2004; Barboza and Parker 2008; Vors 2013). During the snow-free season, Caribou consume a wide variety of nitrogen-rich herbaceous vegetation, which is essential for protein synthesis. Male Caribou may lose up to 25% of their protein reserves during the rut (Barboza et al. 2004) and female protein stores are allocated to gestation and lactation (Gerhart et al. 1997). Caribou winter diet is nitrogen-poor because of higher dependence on lichens, but Caribou are able to conserve protein to cope with this dietary deficiency (Parker et al. 2005). Caribou also reduce forage intake during winter in response to reduced forage quality and availability (i.e., under the snow), as well as to lower energy requirements.
Dispersal and Migration
Newfoundland population
The NP aggregate into social groups of tens to hundreds of individuals, and most sub-populations undertake small seasonal migrations. Some NP sub-populations undertake short seasonal migrations to calving grounds, and summer and wintering ranges, while other sub-populations remain within the same general area throughout the year. The timing of NP migration varies within and between sub-populations and may even vary on an individual scale due to environmental factors (e.g., snow), or may be consistent from year to year, regardless of environmental factors (Mahoney and Schaefer 2002a).
Seven migratory corridors are consistently used by the NP (Wells et al. 2011). Caribou remain at calving grounds until late June before migrating to summer ranges. Site fidelity is strong but varies with season and density (Weir et al. 2014). Aggregation is most pronounced during post-calving (Schaefer and Mahoney 2013). NP display less movement and stronger site fidelity during summer, and possess less fidelity to traditional calving grounds following a population peak (Schaefer and Mahoney 2013). Following the rutting season, Caribou migrate to wintering areas with better access to forage and cover. In contrast, females exhibit much weaker philopatry during winter and individuals were typically 35-45 km away from their previous year’s location (Schaefer and Mahoney 2013). This pattern may indicate that the NP is more flexible about foraging conditions (e.g., abundance and availability) in winter than in spring and summer because they may face more stringent habitat requirements during these seasons.
Gaspésie population
The GP undertakes altitudinal migrations on a limited spatial scale, from closed coniferous forest to open alpine sites (Moisan 1958). The home range size of adult females is small (mean 148 km2, from 1987-1992) and does not vary seasonally or annually (Ouellet et al. 1996). The GP use alpine habitats in the fall, as open areas seem to facilitate mate interactions (Bergerud 1973). Females use alpine habitat in spring and at calving to avoid predation. Nevertheless, no distinct seasonal migration pattern exists for females (Ouellet et al. 1996).
Dispersal is limited for the GP because habitat around their annual range has been highly modified by forestry. It appears that Caribou on the three summits (Logan, Albert and McGerrigle; Figure 2) are relatively isolated and that the DU is acting as a metapopulation composed of three sub-populations. The Mount Logan sub-population was previously abandoned by Caribou in the 1980s and later recolonized starting in 1997, likely by Caribou that dispersed from Mount Albert (Fournier and Faubert 2001; Mosnier et al. 2003). Movement is limited though; there have not been any observations of movement from one summit to another during telemetry surveys (1975-1977; 1987-1992, 1998-2001), or when nearly half (45%) of the population have been collared (2013-present). As well, resource selection modelling suggests that the matrix between the summits is a functional barrier and movement is not expected to be significant (Gaudry 2013). There have been cases of solitary Caribou, mostly males, moving approximately 200 km away, up to the Matapédia Valley in the southwest, and to the tip of the Gaspésie Peninsula to the northeast (St-Laurent pers. comm. 2014).
Boreal population
BP Caribou group size is typically < 50 animals, with the smallest number occurring during calving when females disperse, and the largest number during the rut and winter (Bergerud 1985). For example, in BC, mean group size was 6 Caribou in October and March, but females in the same range were by themselves (or with newborn) during calving (BC Ministry of Environment 2010).
Caribou in the BP frequently move over large areas to access predator-free areas, and food. Changes in movement patterns and behaviour suggest that there are 4-6 distinct seasons; a summer calving and post-calving season, a late winter season where Caribou were more sedentary, and spring and early winter seasons where Caribou made much larger and directional movements (Brown et al. 2003; Ferguson and Elkie 2004; OMNR 2014). Rudolph and Drapeau (2012) suggest there are three seasons; winter, spring dispersal, and calving.
Annual home range size varies greatly across the range (Rasiulis et al. 2012). Home range size for female Caribou ranged from 3312-4790 km2 in northeastern Ontario (Brown et al. 2003). Caribou home range varied from 1148 km2-5000 km2 and average movement was 37-53 km between winter and summer range in the Ontario Shield Ecozone, while those in the James Bay Ecozone had home ranges of 15000-75000 km2, and moved up to 384 km between summer and winter ranges (OMNR 2014). Mean annual home range size ranged from 776-2796 km2 among four local populations in Québec (Bastille-Rousseau et al. 2012). Of these populations, the northernmost Jamésie population displayed the largest home ranges. In Labrador, average home range size was 5650 +/- 259 km2 for the Red Wine Mountain sub-population, compared to 4866 ± 256 km2 for the Lac Joseph sub-population, monitored for 4-11 years (Rasiulis et al. 2012). Larter and Allaire (2014) reported that the mean home range size for 82 female Caribou was 2989 km2 (range 261-14,420 km2; median 2328 km2) in the NT.
The annual movements of Caribou consist of small-scale shifts between seasonal ranges that are embedded in a larger annual home range (Brown et al. 2003). Rasiulis et al. (2012) reported on the importance of long-term monitoring because monitoring lasting ≤ 3 years captured < 65% of the sub-population range for Caribou in Labrador. They also demonstrated that range size increased with each additional year of monitoring, for up to seven years. Home range sizes for individual BP in Labrador indicate that animals increase their range over time and make use of large regions (~5000 km2) throughout their lifetime, a strategy consistent with a long-lived animal at low densities in a landscape with varying environmental conditions (Schmelzer 2014).
Although some areas have recorded low site fidelity (e.g. in Saskatchewan; Rettie and Messier 2001), most studies indicate site fidelity is strong in BP, particularly by females to calving grounds (Schaefer et al. 2000; Faille et al. 2010; BC Ministry of the Environment 2010; Shuter and Rogers 2012). For example, in the Mealy Mountain sub-population, Labrador, site fidelity to calving grounds was strong among 12 females that calved an average 3.9 km (year 1) and 11.5 km (year 2) from sites used the previous year. Lower snow levels in year 2 may explain lower fidelity. Post-calving fidelity averaged 7.7 km, and winter fidelity was weakest, at 17 and 19 km (Popp et al. 2011). Females in BC made pre-calving movement of 90 km each year to calving areas (BC Ministry of the Environment 2010).
Monitoring in Labrador established that approximately 10% of individuals move among adjacent sub-populations per year, or for consecutive years, but most of the sub-population remains with the same animals and reuses parts of the landscape each year (Schmelzer pers. comm.). The sub-population can shift to from 10 km to > 100 km in response to natural (e.g., snow, forage, and fires) and anthropogenic (e.g., forestry) factors (Stuart-Smith et al. 1997; Dyer et al. 2001; Rettie and Messier 2000). Caribou are most sedentary during winter, tend to reoccupy the same wintering areas (Faille et al. 2010), and often use frozen bodies of water as travel corridors (Ferguson and Elkie 2004). Movement rates are greatest in late spring (Ferguson and Elkie 2004), when females disperse from one another to give birth. In the NT, Nagy (2011) monitored 140 boreal adult female and reported that their daily movement rate varied between 1.1 km/day in late winter to 5.0 km/day in late fall. In Québec and Labrador, daily movements of the Red Wine Mountains and Lac Joseph sub-populations were 4-7 times lower than those of sympatric migratory sub-populations (Couturier et al. 2010). For the same sub-populations during the snow-bound period (December 26-April 30), movement rates were 1.4 and 0.9 km/day, respectively, while it was 2.1 and 1.6 km/day during the rest of the year (Couturier et al. 2010). Schmelzer (2012) reported that daily movements for these sub-populations from 2007-2012 were lowest during late winter and, secondarily, calving, and highest during spring and fall migration where females travelled an average of 3.5 km/day. Movement rates are greatest in late spring (Ferguson and Elkie 2004), when females disperse from one another to give birth.
Interspecific Interactions
Caribou share their range with other large herbivores across Canada. BP range overlaps slightly with Muskox (Ovibos moschatus) range in the NT. The following interactions with Caribou were described in the Sahtu Settlement Area in NT:
“Some people claimed that muskox cause boreal caribou to abandon areas because of their hair, the noise they make, or because of parasites they transmit in their feces. Other people said they have seen boreal woodland caribou and muskox feeding on the same plants in the same places without competition or exclusion.” COSEWIC (2012; p. 68).
West of Manitoba, BP range overlaps slightly with Wood Bison (Bison bison) range. Fischer and Gates (2005) reported strong space use differences between Bison and Caribou, including elevation, slope, and distance from permanent water bodies. Bison showed a strong preference for graminoids while Caribou preferred lichens. As only 10% of their winter diet overlapped, they concluded that exploitative competition between Caribou and Bison was unlikely.
Caribou range reaches the northern limit of many cervids in Canada. Though Caribou spatially separate themselves from other cervids at the stand scale (see Habitat Requirements section), their ranges overlap with Moose, White-tailed Deer, Mule Deer, and Elk (Cervus elaphus). Caribou habitat needs are different but these species may be found in close proximity, particularly in the southern portion of Caribou range. Overlap of Caribou with some cervids is predicted to increase with a warming climate (Vors and Boyce 2009). These interactions could be highly important with respect to disease/parasite transmission (see Threats – Problematic Native Species section)
Caribou are an important food item for numerous predators and a source of scavenged meat. Wolves are the most important predators of Caribou, although Black Bear, Grizzly Bear, Coyote, Cougar (Puma concolor), Lynx (Lynx canadensis), and Wolverine (Gulo gulo) are predators of both adult and calf Caribou (Bergerud 1974; Gustine et al. 2006; Boisjoly et al. 2010; Pinard et al. 2012). Golden eagles (Aquila chrysaetos) (Crête and Desrosiers 1995) and Bald Eagles (Haliaeetus leucocephalus) may also prey on Caribou calves (Mahoney and Weir 2009; McCarthy et al. 2011). Scavenging by Wolverine on Caribou carcasses is important to Wolverine across much of their range.
There has been limited research on subclinical parasitism in Caribou, until recently (Gunn and Irvine 2003; Gunn et al. 2011). Gastrointestinal parasites are universally prevalent in Rangifer and while the infections may not cause obvious symptoms, they are costly to Caribou (Gunn and Irvine 2003) by reducing fecundity and sometimes even regulating caribou abundance, as suggested by a study on Svalbard Reindeer (Albon et al. 2002). Trends in gastrointestinal parasites are unknown but climate warming will likely influence the host-parasite relationship (Gunn et al. 2011).
Liver Fluke (Fasciola hepatica) is only found in migratory Caribou of northern Québec and Labrador (Lankester and Luttich 1988), where it can reach prevalence of close to 100% (Côté, S.D., and Couturier, S., unpubl. data). However, these migratory sub-populations are overlapping their seasonal range with BP and future transmission remains possible.
Besnoitia tarandi has been documented in Caribou and Reindeer for almost a century, but little is known about its epidemiology, life cycle (Ducrocq et al. 2012, 2013) and transmissibility (Kutz et al. 2009). This protozoan parasite has emerged as a significant disease-causing agent in migratory Caribou of Quebec (DU4) (Kutz et al. 2009) and could be an issue in the adjacent BP. Numerous other parasites and pathogens are suspected to impact, or potentially impact Caribou, such as Toxoplasma gondii, Neospora caninum, Babesia sp., Giardia sp., Mycobacterium avium, Erysipelothrix rhusiopathiae, Trypanosoma sp., Cryptosporidium sp. (Johnson et al. 2010; Kutz pers. comm.). Research is underway to establish the effect on productivity and mortality, and the extent of sub-populations impacted (Kutz pers. comm.).
Population Sizes and Trends
Sampling Effort and Methods
Numerous aerial surveys and radio-collaring studies have been conducted in each DU to document Caribou abundance, space use, habitat selection, mortality rates, cause of death, and demographic trends. However, estimating Caribou numbers is challenging because of large ranges, low densities, and forest cover that hinders visual surveys. These challenges are most profound for BP across their large range, but GP and NP also have census difficulties because of weather and remoteness. Caribou counts are conducted with aerial surveys and a standardized census method has been proposed (Courtois et al. 2003b). Some jurisdictions record animals as calves and mature animals during aerial surveys. Calves in their second autumn are of similar appearance as mature animals, and mature is defined as > 1 year old (Lalonde pers. comm. 2014). New techniques using fecal DNA have been successfully tested (Hettinga et al. 2012). Demographic models are commonly used to determine recruitment and survival rates, based on radio-collared animals, and/or aerial surveys.
Few jurisdictions attempt to determine % population change using aerial surveys because of concerns over bias between surveys, particularly if surveys are few over many years. Jurisdictions conducting rigorous surveys over multiple years can more confidently detect trends and % change data exist for Labrador, the GP and NP. If % change data are lacking, the future trend can be predicted from population growth rate data, and the amount of disturbance in each range. Indices of population decline are widely used in Caribou management. The population finite growth rate (Lambda, λ) using a measure of recruitment and mortality (e.g., Hatter and Bergerud 1991; λ=(1-M)/(1-R), where M is adult female mortality and R is population recruitment) is often considered the best method for detecting declines (Bergerud 2000). A λ of < 1.0 indicates population decline; a population with a Lambda of 0.90 will decline by 50% in approximately 7 years. A recruitment rate of 15% is considered a threshold for a stable Caribou population when it is coupled with an 85% adult survival rate and normal sex ratios (Bergerud 1980). Bergerud and Elliot (1986) report that ‘negative recruitment’ (or recruitment that fails to balance adult mortality), occurs at values of < 10-12%, and a preferred value of 15% is used because it offsets uncertainty. Schmelzer (2013) examined population dynamics of three Labrador Caribou sub-populations from 1997- 2012 and indicated that a survival to recruitment ratio of 90/20 was much more likely to result in demographic stability. Calf recruitment rates have been established to be a minimum of 25 calves per 100 cows in order to avoid population decline (Bergerud and Elliot 1998). Environment Canada (2008) suggests a minimum recruitment rate of 28.9 calves per 100 cows.
Newfoundland population
The distribution of the NP has been monitored since the 1800s and abundance and demographic characteristics have been researched since the 1950s. Extensive censuses have been conducted on major sub-populations throughout the island, and Bergerud (1971) compiled and analyzed data collected from the 1900s to 1967. Radio-collaring was initiated in 1979 with more intensive, large-scale sampling in the mid-1990s. Researchers deployed 100 GPS and 99 satellite collars in 2007 to augment an already large (>1,200) sample of collared Caribou; over 2300 Caribou have been radio-collared since 1979 (Pardy Moores pers. comm.). Until 2013, sampling effort remained intensive, with large-scale satellite collar deployments, extensive surveys of sub-population composition, and mortality investigations.
A calf mortality study initiated in 2003 included sub-populations representing different ecological regions and their predators. The ‘Caribou Strategy’, which ran from 2008- 2013, was a coordinated effort to determine factors driving the decline in the island’s Caribou population (Mahoney and Weir 2009), as well as implement predator ecology studies, evaluate Caribou-predator-habitat interactions, conduct predator reduction experiments, increase public knowledge and engage the public. In addition to collaring, aerial surveys and fall classifications have been completed and repeated for all major sub-populations over the duration of the Caribou Strategy (Weir et al. 2014). Hunting information is also collected to determine the quality and state of the animals, but also to detect hunting trends.
Gaspésie population
During the 1970s, the GP was studied using sporadic surveys. Since 1983, annual aerial surveys were used to estimate population size, sex ratio, and calf abundance. Radio-tracking occurred from 1975-1977, 1987-1992, 1998-2001, and 2013-present, and data gathered contribute to a better assessment of population size and trends.
All annual population estimates include a correction factor of 0.70 to account for visibility bias, as not all Caribou are seen on the barren lands during the census (Lalonde 2013; Lalonde and Michaud 2013). This correction factor was estimated from 1999 to 2005 when some radio-collars were in use in the three subpopulations. However, visibility and census conditions vary with time, suggesting that this average correction factor may no longer be accurate. Indeed, it may be too low, resulting in an overestimation of the sub-population size (Lalonde pers. comm.).
Boreal population
Population information is vague or absent for many BP ranges. Population estimates are hindered by a lack of standardized animal location data, limited understanding of Caribou movement within and between ranges, and the difficulty in delineating population ranges because occupancy is generally continuous for much of the range (Schmelzer et al. 2004; Couturier 2007; Couturier et al. 2009; Environment Canada 2011). The lack of known critical habitat in Caribou range in northern Saskatchewan prompted a large population and habitat research program in the Boreal Shield range in 2014 (USask 2014). The National Recovery Strategy has maximum and minimum population estimates derived by aerial surveys. Often, the best available data are a minimum number of Caribou counted during aerial surveys. Correction factors for visibility bias (due to weather or forest cover) are not equally applied over years and ranges, resulting in limited use of minimum counts as a measure of population trend.
Population trend data can be estimated by comparing aerial survey counts conducted over time but this type of data is uncommon in the BP because the large area, low density, and forest cover result in weak confidence in comparing surveys over time. It is more common to assess population status by using aerial surveys, and survival measures to establish population growth rate (Lambda; λ). This depends on surveys conducted several years in succession, which is not common in some jurisdictions. In BC, the objective is to radio-collar 15% of the animals in each range in order to determine calf recruitment and mortality rates (Culling and Culling 2013). In Alberta, Hervieux et al. (2013) used adult female and calf survival rates from radio-collared animals to determine Lambda for 11 ranges. Lambda also is used in Ontario (OMNR 2014), Northwest Territories (Larter and Allaire 2014), and Labrador (Schmelzer 2013).
Abundance
Newfoundland population
In 2013, the total population (including calves) was estimated at 31,980 Caribou (NL Gov., unpubl. data). The population of mature Caribou is about 28,240, based on applying this calf recruitment mean to the total population estimated in 2013. Estimates of mature individuals were 74,912 in 1995 (18.7% recruitment) and 66,623 in 2001 (11.7% recruitment) (Weir et al. 2014).
Gaspésie population
The total number recorded in 2013 was 91 (all ages), which increases to an estimate of 130 with use of the visibility correction factor. The number of mature animals is estimated as 112, based on subtracting the proportion of calves (14.3%) counted in the autumn survey. The number of Caribou observed during surveys has declined on Mount Albert since 1996. No animals were recorded on Mount Logan in 2012 or 2013. Most (87%) of the animals were recorded on Mount McGerrigle (St-Hugues pers. comm.)
Boreal population
Historical population estimates are only available in some regions (e.g., Lake Superior Ontario; Cringan 1956; Bergerud et al. 2007). Organized censuses and studies of the BP occurred within the last 20 years. Based on present scientific and traditional knowledge of its ecology, it is likely that BP was never abundant and never approached the high densities of Caribou recorded in northern tundra. BP numbers vary widely among jurisdictions (Table 1).
| Jurisdiction | Population estimate (min.-max.) | Range size (km2) | Local ranges delineated | Local ranges surveyed |
|---|---|---|---|---|
| Alberta | 2,074-2,315 | 146047 | 12 | 12 |
| British Columbia | 1,040-1,110 | 25999 | 5 | 5 |
| Labrador | 2,983 [2909]Noteaof Table 1 | 155895 | 3 | 3 |
| Manitoba Notebof Table 1Notecof Table 1 | 1,063-1,543 | 211865 | 13 | 10 |
| NT | 6500 | 441665 | 1 | 1 |
| Ontario Note b of Table 1Note c of Table 1Notedof Table 1 | 1,284-5000 | 497554 | 9 | 3 |
| Québec Notebof Table 1Noteeof Table 1 | 7091 | 680433 | 6 | 6 |
| Saskatchewan Notebof Table 1Note c of Table 1Notefof Table 1 | No data | 286273 | 2 | No data |
| Canada (Total) | 24,722-30,513 | 2445731 | 51 | 40 |
| Viability | Range ID | Range name | Range area (ha) | Range area (km2) | Population size (min) | Population size (max) | Max. Density (#/100 km2) |
|---|---|---|---|---|---|---|---|
| SELF-SUSTAINING | MB10 | Manitoba South | 1867255 | 18673 | ? | ? | - |
| SELF-SUSTAINING | MB11 | Manitoba East | 6612782 | 66128 | ? | ? | - |
| SELF-SUSTAINING | MB12 | Atikaki-Berens | 2387665 | 23877 | 300 | 500 | 2.09 |
| SELF-SUSTAINING | MB4 | Reed | 357425 | 3574 | 100 | 150 | 4.20 |
| SELF-SUSTAINING | MB7 | Wabowden | 628938 | 6289 | 200 | 225 | 3.58 |
| SELF-SUSTAINING | MB8 | Wapisu | 565044 | 5650 | 110 | 125 | 2.21 |
| SELF-SUSTAINING | NT1 | NT | 44166546 | 441665 | 6500 | 6500 | 1.47 |
| SELF-SUSTAINING | ON3 | ChurchillNotegof Table 2 | 2150490 | 21505 | ? (262)Notehof Table 2 | ? | - |
| SELF-SUSTAINING | ON5 | NipigonNotegof Table 2 | 3885026 | 38850 | 300 (172)Notehof Table 2 | 300 | 0.77 |
| SELF-SUSTAINING | ON6 | Coastal | 376598 | 3766 | 492 | 492 | 13.06 |
| SELF-SUSTAINING | ON7 | PagwachuanNotegof Table 2 | 4542918 | 45429 | ? (164)Notehof Table 2 | ? | - |
| SELF-SUSTAINING | ON9 | Far North | 28265143 | 282651 | ? | ? | - |
| SELF-SUSTAINING | QC5 | Manicouagan | 1134129 | 11341 | 181 | 181 | 1.60 |
| SELF-SUSTAINING | QC6 | Québec | 62156186 | 621562 | 9000 | 9000 | 1.45 |
| SUB-TOTAL | - | - | 159096145 | 1,590,961 (65.1%)Noteiof Table 2 | 17183 | 17,473 (68.5%)Noteiof Table 2 | - |
| NOT SELF-SUSTAINING | AB1 | Chinchaga (with BC) | 3162612 | 31626 | 250 | 250 | 0.79 |
| NOT SELF-SUSTAINING | AB10 | Cold Lake | 672422 | 6724 | 150 | 150 | 2.23 |
| NOT SELF-SUSTAINING | AB11 | Nipisi | 210771 | 2108 | 55 | 55 | 2.61 |
| NOT SELF-SUSTAINING | AB12 | Slave Lake | 151904 | 1519 | 65 | 65 | 4.28 |
| NOT SELF-SUSTAINING | AB2 | Bistcho | 1436555 | 14366 | 195 | 195 | 1.36 |
| NOT SELF-SUSTAINING | AB3 | Yates | 523094 | 5231 | 350 | 350 | 6.69 |
| NOT SELF-SUSTAINING | AB4 | Caribou Mountains | 2069000 | 20690 | 315 | 394 | 1.90 |
| NOT SELF-SUSTAINING | AB5 | Little Smoky | 308606 | 3086 | 78 | 78 | 2.53 |
| NOT SELF-SUSTAINING | AB6 | Red Earth | 2473729 | 24737 | 172 | 206 | 0.83 |
| NOT SELF-SUSTAINING | AB7 | West Side Athabasca | 1572652 | 15727 | 204 | 272 | 1.73 |
| NOT SELF-SUSTAINING | AB8 | Richardson | 707350 | 7074 | 150 | 150 | 2.12 |
| NOT SELF-SUSTAINING | AB9 | East Side Athabasca | 1315980 | 13160 | 90 | 150 | 1.14 |
| NOT SELF-SUSTAINING | BC1 | Maxhamish | 710105 | 7101 | 300 (132)Notejof Table 2 | 300 | 4.22 |
| NOT SELF-SUSTAINING | BC2 | Calendar | 496393 | 4964 | 290 (135)Notejof Table 2 | 290 | 5.84 |
| NOT SELF-SUSTAINING | BC3 | Snake-Sahtahneh | 1198752 | 11988 | 360 (321)Notejof Table 2 | 360 | 3.00 |
| NOT SELF-SUSTAINING | BC4 | Parker | 75222 | 752 | 40 (59)Notejof Table 2 | 60 | 7.98 |
| NOT SELF-SUSTAINING | BC5 | Prophet | 119396 | 1194 | 50 (35)Notejof Table 2 | 100 | 8.38 |
| NOT SELF-SUSTAINING | MB2 | Kississing | 317029 | 3170 | 50 | 75 | 2.37 |
| NOT SELF-SUSTAINING | MB3 | Naosap | 456977 | 4570 | 100 | 200 | 4.38 |
| NOT SELF-SUSTAINING | MB6 | William Lake | 488219 | 4882 | 25 | 40 | 0.82 |
| NOT SELF-SUSTAINING | NL2 | Red Wine Mountain | 5838594 | 58386 | 97 | 97 | 0.17 |
| NOT SELF-SUSTAINING | ON1 | Sydney | 753001 | 7530 | ? (55)Notehof Table 2 | ? | - |
| NOT SELF-SUSTAINING | ON8 | Kesagami | 4766463 | 47665 | 492 (164)Notehof Table 2 | 492 | 1.03 |
| NOT SELF-SUSTAINING | QC1 | Val-d’Or | 346861 | 3469 | 30 | 30 | 0.86 |
| NOT SELF-SUSTAINING | QC2 | Charlevoix | 312803 | 3128 | 75 | 75 | 2.40 |
| NOT SELF-SUSTAINING | QC3 | Pipmuacan | 1376899 | 13769 | 134 | 134 | 0.97 |
| SUB-TOTAL | - | - | 31861389 | 318,614 (13%)Noteiof Table 2 | 4117 | 4,568 (17.9%)Noteiof Table 2 | - |
| UNCERTAIN STATUSNotekof Table 2 | MB1 | The Bog | 446383 | 4464 | 50 | 75 | 1.68 |
| UNCERTAIN STATUS | MB13 | Owl-Flinstone | 363570 | 3636 | 78 | 78 | 2.15 |
| UNCERTAIN STATUS | MB5 | North Interlake | 489680 | 4897 | 50 | 75 | 1.53 |
| UNCERTAIN STATUS | MB9 | Manitoba North | 6205520 | 62055 | ? | ? | - |
| UNCERTAIN STATUS | NL1 | Lac Joseph | 5802491 | 58025 | 1282 | 1282 | 2.21 |
| UNCERTAIN STATUS | NL3 | Mealy Mountain | 3948463 | 39485 | 1604 | 1604 | 4.06 |
| UNCERTAIN STATUS | ON2 | Berens | 2794835 | 27948 | ? (237)Notehof Table 2 | ? | - |
| UNCERTAIN STATUS | ON4 | Brightsand | 2220921 | 22209 | ? (224)Notehof Table 2 | ? | - |
| UNCERTAIN STATUS | QC4 | Manouane | 2716449 | 27164 | 358 | 358 | 1.32 |
| UNCERTAIN STATUS | SK1 | Boreal ShieldNotelof Table 2 | 18034870 | 180349 | ? | ? | - |
| UNCERTAIN STATUS | SK2 | Boreal Plain | 10592463 | 105925 | ? | ? | - |
| SUB-TOTAL | - | - | 53615645 | 536,156 (21.9%)Noteiof Table 2 | 3422 | 3,472 (13.6%)Noteiof Table 2 | |
| TOTAL | - | - | 244573179 | 2445732 | 24722 | 25513 |
The BP was estimated at 33,000 in 2002 (COSEWIC 2002), and 34,000 in 2012 (Environment Canada 2012). A maximum of 25,513 is estimated from those sub-populations with data (Table 2), and an additional 8000 animals are estimated for the remaining sub-populations. Although these numbers were based on best available information, they are only a crude approximation and the population is actually unknown. In Quebec, for example, the estimate of 7,091 animals is based on extrapolation of density from several study areas to the known range in the province. Data quality on density and abundance are variable across BP range and some range population estimates were missing.
Survival and recruitment
Predation is the main proximate factor limiting Caribou population growth because the survival of calves to one year of age is usually low and is often insufficient to compensate for annual adult mortality in declining populations (Bergerud 1974; Stuart-Smith et al. 1997; DeMars et al. 2011). Recruitment rates are expressed as actual survival derived from mortality records and survival models, or as an index of the ratio of cows to calves recorded in aerial surveys conducted in the fall (see Sampling Effort and Methods section for stability thresholds).
Newfoundland population
In the NP, adult survival is high (combined mean of 86%; range: 82-92%) for all sub-populations that have been studied between 2004 and 2010. Calf survival is not significantly different between males and females, though males do have a slightly lower survival rate than females (Lewis and Mahoney 2014).
Calf survival rates were estimated in the 1980s and 1990s for many NP sub-populations (Table 3) and averaged 62% for the first six months before declining to a mean of 35% between 2003 and 2012 (Lewis and Mahoney 2014; Figure 4). Trindade et al. (2011) reported an extremely low annual calf survival rate of 4% (SD: 5%) for the Middle Ridge, Gaff Topsails, and Mount Peyton sub-populations between 2003 and 2007. Calf survival has greatly increased since 2007, with rates near 50% in 2009 and 2012 (Figure 4). Data from 2003 to 2012 (Figures 4, 5) suggest the decline in recruitment has halted (Weir et al. 2014; Lewis and Mahoney 2014).
| Population Area | Designatable Unit/ Sub-population |
Years Notemof Table 3 | Calf survival (%) Note n of Table 3 | Recruitment (%, or calf/100 females)Noteoof Table 3 | Source |
|---|---|---|---|---|---|
| Newfoundland Population (NP) | Several sub-populations | 1979-97 | 67 (to 6 months) | - | Lewis and Mahoney 2014 |
| Newfoundland Population (NP) | 3 sub-populations | 2003-07 | 4 ± 5 (SD) | - | Trindade and al. 2011 |
| Newfoundland Population (NP) | Middle Ridge | 1990s | - | 25-30% Notepof Table 3 | Mahoney and Weir 2009 |
| Newfoundland Population (NP) | Middle Ridge North | 2003-05 | 5 (1-12) | - | Lewis and Mahoney 2014 |
| Newfoundland Population (NP) | Middle Ridge North | 2006-11 | 20 (6-36) | - | - |
| Newfoundland Population (NP) | Middle Ridge North | 2012 | 38 | - | - |
| Newfoundland Population (NP) | Middle Ridge South | 2003-05 | 13 (11-15) | - | - |
| Newfoundland Population (NP) | Middle Ridge South | 2006-11 | 0.5 (0-2) | - | - |
| Newfoundland Population (NP) | Middle Ridge South | 2012 | 23 | - | - |
| Newfoundland Population (NP) | LaPoile | 2007-11 | 24 (5-40) | - | - |
| Newfoundland Population (NP) | LaPoile | 2012 | 26 | ||
| Newfoundland Population (NP) | Mount Peyton | 2003 | 11 | ||
| Newfoundland Population (NP) | Northern Peninsula | 2008-11 | 25 (3-41) | ||
| Newfoundland Population (NP) | Northern Peninsula | 2012 | 41 | ||
| Newfoundland Population (NP) | NP all sub-populations | 1966-97 | - | 40.8 c/100F Notepof Table 3 | Weir and al. 2014 |
| Newfoundland Population (NP) | NP all sub-populations | 2003-12 | 35 | 16.3 c/100F Notepof Table 3 | Lewis and Mahoney 2014 |
| Boreal Population (BP) | 6 ranges in British Columbia |
2009-10 2013 |
- | 17 c/100F 21 c/100F, 13.4% |
Thiessen 2009, BC MoE 2010 Culling and Culling 2013 |
| Boreal Population (BP) | Alberta (8-13 sub-populations) | 2001-08 | - | 18 c/100F | Alberta Caribou Committee |
| Boreal Population (BP) | Alberta (11 sub-populations)Noteqof Table 3 | 1998-12 | - | 15 c/100F | Hervieux and al., 2013 |
| Boreal Population (BP) | Saskatchewan | 1993-96 | - | 28 c/100F | Rettie and Messier, 1998 |
| Boreal Population (BP) | NT - Dehcho | 2005-14 | - | 36 c/100FNotemof Table 3 | Larter and Allaire, 2014 |
| Boreal Population (BP) | NT - South Slave | 2004-10 | - | 24 c/100F | Kelly and Cox, 2011 |
| Boreal Population (BP) | Ontario; Berens | 2011-12 | - | 10.7 c/100F | Ontario Ministry of Natural Resources, 2014 |
| Boreal Population (BP) | Sydney | - | - | 15.7 c/100F | |
| Boreal Population (BP) | Churchill | - | - | 15.6 c/100F | |
| Boreal Population (BP) | Brightsand | 2010-12 | - | 22 c/100F | |
| Boreal Population (BP) | Nipigon | 2009-12 | - | 33.1 c/100F | |
| Boreal Population (BP) | Pagwachuan | - | - | 21.6 c/100F | |
| Boreal Population (BP) | Kesagami | - | - | 19.4 c/100F | |
| Boreal Population (BP) | Swan | - | - | 16.1 c/100F | |
| Boreal Population (BP) | Spirit | 2008-11 | - | 20.1 c/100F | |
| Boreal Population (BP) | Kinlock | - | - | 12.2 c/100F | |
| Boreal Population (BP) | Ozhiski | - | - | 17.2 c/100F | |
| Boreal Population (BP) | James Bay | - | - | 18.8 c/100F | |
| Boreal Population (BP) | Missisa | - | - | 14.2 c/100F | |
| Boreal Population (BP) | Avg. 13 sub-populations | 2008-12 | - | 18.2 c/100F | |
| Boreal Population (BP) | Québec | - | 53 (to 30 d) 43 (to 90 d) |
- | Leclerc et al., 2012 |
| Boreal Population (BP) | Charlevoix | - | 46 (to 50 d) | - | Pinard et al., 2012 |
| Boreal Population (BP) | Labrador; Mealy Mtns. | 1971 | - | 26.1% | Schmelzer, 2013 |
| Boreal Population (BP) | Labrador; Mealy Mtns. | 1974-77 | - | 18.9% (14.8-21.1) | |
| Boreal Population (BP) | Labrador; Mealy Mtns. | 1987 | - | 17.7% | |
| Boreal Population (BP) | Labrador; Mealy Mtns. | 1994 | - | 12.8% | |
| Boreal Population (BP) | Labrador; Mealy Mtns. | 2002 | - | 28.8% | |
| Boreal Population (BP) | Labrador; Mealy Mtns. | 2005 | - | 16.3% | |
| Boreal Population (BP) | Labrador; Mealy Mtns. | 2008-09 | - | 16.0% (13.8-18.2)Noterof Table 3 | |
| Boreal Population (BP) | Labrador; Mealy Mtns. | 2011-12 | - | 12.5% (10.5-14.5)Noterof Table 3 | |
| Boreal Population (BP) | Red Wine Mountains | 1974 | - | 12.5% | Bergerud et al. 2008 |
| Boreal Population (BP) | Red Wine Mountains | 1984, 1987, 2009 | - | 16.4%, 19.2%, 11.8% | Schmelzer 2013 |
| Boreal Population (BP) | Red Wine Mountains | 2001-03 | - | 19.3% (12.3-26.9) | |
| Boreal Population (BP) | Lac Joseph | 1984-86 | - | 19.5% (15.2-23.1) | St-Martin 1987 |
| Boreal Population (BP) | Lac Joseph | 1998-01 | - | 14.9% (11.9-15.7) | Schmelzer 2013 |
| Boreal Population (BP) | Lac Joseph | 2002-05 | - | 16.8% (15-19.7) | |
| Boreal Population (BP) | Lac Joseph | 2007-09 | - | 20.9% (1.5-23.6) |
Long description for Figure 4
Chart illustrating the trend in the first six-month survival of Newfoundland population Caribou calves from 1979 to 2012, with 95 percent confidence intervals. Estimates of calf survival rates in the 1980s and 1990s averaged 62 percent for the first six months before declining to a mean of 35 percent between 2003 and 2012 (as discussed in the report text). Calf survival has greatly increased since 2007, with rates near 50 percent in 2009 and 2012.
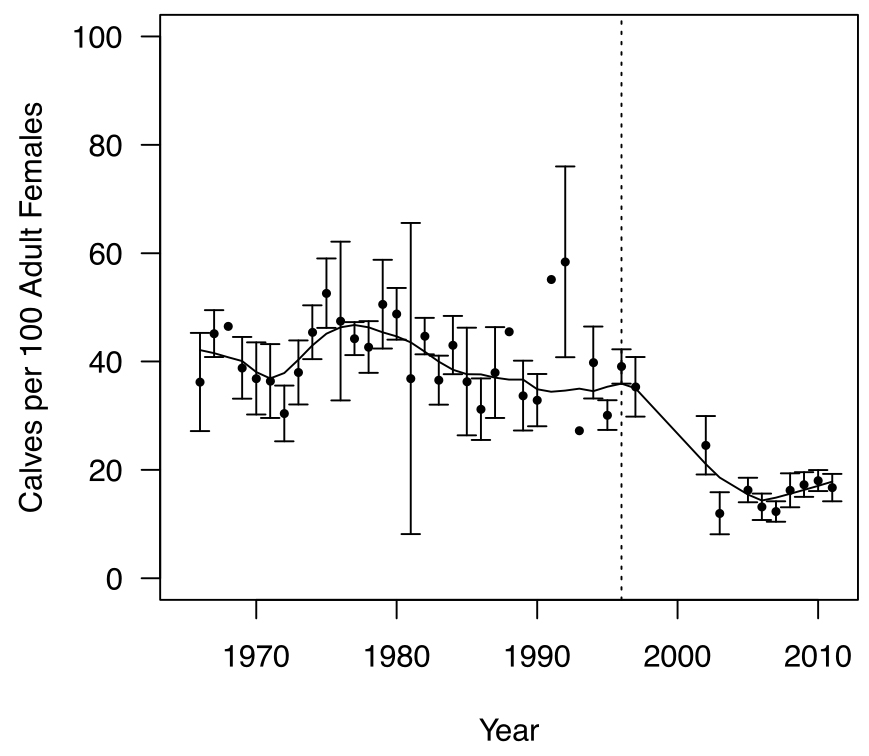
Long description for Figure 5
Chart illustrating the trend in Caribou calf abundance (number of calves per 100 adult females) recorded in autumn on the island of Newfoundland, in the years 1966 to 2011. The trend line indicates that the decline in recruitment has halted since the mid-2000s.
Sub-population composition surveys across Newfoundland indicate that the proportion of females giving birth declined slightly, but considerable variation exists among sub-populations (Mahoney and Weir 2009). The Pot Hill and Grey River sub-populations both showed steady declines, whereas other sub-populations only showed slight declines in calf production. These sub-population-specific trends may reflect the differential availability of forage resources, which results in differences in the female reproductive potential and lower calf weight (Mahoney and Weir 2009). A greater weight at birth has been correlated with increased calf survival in BP range (Pinard et al. 2012) and recent analyses on a larger data set (2003-2011) indicated a similar relationship in Newfoundland (Lewis and Mahoney 2014).
Predator vulnerability is inversely related to prey age. For example, the majority of deaths due to predation occur within the first 12 weeks of a Caribou calf’s life, and >70% of calves that survive until fall will reach one year of age (Mahoney and Weir 2009).
Gaspésie population
Female adult survival was 92% from 1987 to 1992, but later declined to 83% in 1999-2000 (Table 4). More recent data for adult mortality suggests that more adults than expected are subject to predation by Coyote (St-Laurent pers. comm. 2014). Predator control programs were initiated if these annual surveys indicated low recruitment. Between 1990 and 1996, a predator control program was implemented with short term, positive results (Crête and Desrosiers 1995). Predator control resumed in 2001 and has continued since then. Until 2011, calf recruitment values were often far below the 17% threshold set as a goal for a stable sub-population by the Équipe de rétablissement du caribou de la Gaspésie (Figure 6). Lesmerises (2012) underlined that this threshold could be underestimated and, considering the current rate of adult mortality, the threshold should be a minimum of ~21% to reach population stability. In 2012 and 2013, the proportion of juveniles was high, which may result in increased recruitment if they survive. The proportion of juveniles probably increased because of the indirect effect of climate on cow’s condition during pregnancy, and predator’s access to the summit during calving (M.-H. St-Laurent, unpubl. data).
| Province/ Regions | Sub-populations | YearsNotetof Table 4 | Mean Survival Rate %Noteuof Table 4 | Source |
|---|---|---|---|---|
| NP DU5 | 5 sub-populations | 2004-11 | 87 (CI: 83-91) | Lewis and Mahoney 2014 |
| GP DU11- Gaspésie | - | 1987-92 | 92 | Crête and Desrosiers 1995 |
| GP DU11- Gaspésie | - | 1999-00 | Males: 79 (CI: 57-100) Fem.: 83 (CI: 67-95) |
Fournier and Faubert 2001 |
| BP DU6 - Alberta | 8-13 | 2001-08 | 86 (range 82-91) | Alberta Caribou Committee |
| BP DU6 - Alberta | 11 | 1994-12 | 85 (range 77-91) | Hervieux et al. 2013 |
| BP DU6 - NT | Dehcho | 2005-14 | 77 (range 62-88) | Larter and Allaire 2014 |
| BP DU6 - NT | South Slave | 2004-10 | 85 (range 76-91) | Kelly and Cox 2011 |
| BP DU6 - Sask. | 5 sub-populations | 1993-96 | 84 (CI: 75-93) | Rettie and Messier 1998 |
| BP DU6 - Manitoba | North Interlake | 2005-09 | Males: 65 (CI: 54-75) Fem.: 76 (CI: 65-85) |
Hettinga et al. 2012 |
| BP DU6 - Québec | 3 sub-populations | 2003-11 | 87 (range 75-100) | Rudolph et al. 2012 |
| BP DU6 - Québec | 3 sub-populations | - | 90 (range 75-100) (hunting excluded) | Rudolph et al. 2012 |
| BP DU6 - Labrador | Mealy Mtn. | 1985-87 | 85 | Schmelzer 2013 |
| BP DU6 - Labrador | Mealy Mtn. | 2002-05 | 92 (range 83-97) | Schmelzer 2013 |
| BP DU6 - Labrador | Mealy Mtn. | 2006-09 | 92 (range 91-93) | Schmelzer 2013 |
| BP DU6 - Labrador | Mealy Mtn. | 2010-12 | 93 (range 92-93) | Schmelzer 2013 |
| BP DU6 - Labrador | Red Wine M. | 1981-88 | 80 (SE=5.2) | Schaefer et al. 1999 |
| BP DU6 - Labrador | Red Wine M. | 1993-97 | 70 (SE=5.4) | Schaefer et al. 1999 |
| BP DU6 - Labrador | Red Wine M. | 1997-00 | 75 (range 60-92) | Schmelzer 2013 |
| BP DU6 - Labrador | Red Wine M. | 2001-04 | 84 (range 77-95) | Schmelzer 2013 |
| BP DU6 - Labrador | Red Wine M. | 2005-09 | 82 (range 70-89) | Schmelzer 2013 |
| BP DU6 - Labrador | Lac Joseph | 1984-87 | 95 (CV=0.05, range 89-100) |
St-Martin 1987 |
| BP DU6 - Labrador | Lac Joseph | 1998-09 | 84 (CV=0.05, range 69-100) | Schmelzer 2013 |
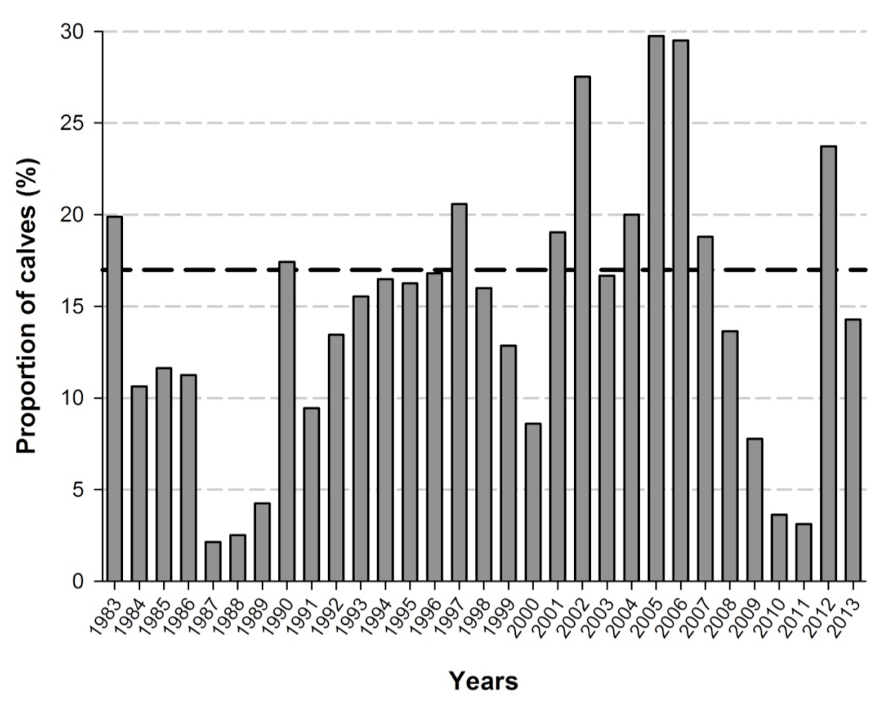
Long description for Figure 6
Chart illustrating the percentage of calves recorded in autumn surveys of Atlantic-Gaspésie population Caribou from 1983 to 2013. The chart indicates the years when this percentage exceeded the target of 17 percent in 1983,1990, 1997, 2001, 2002, 2004, 2005, 2006, 2007, set in the Recovery Plan to achieve a stable population.
Boreal population
Annual adult female survival generally is high (i.e., >75%; Table 4) but fluctuates depending upon levels of hunting. For example, adult female survival for the Lac Joseph sub-population in Labrador was significantly higher between 1984 and 1987, when compared to estimates from 1998-2009 (Table 4; Schmelzer 2013). Using knowledge that deaths from hunting accounted for 30% of all known mortalities, Schmelzer (2013) calculated that mean survival of females over the monitoring period increased by 6% when hunting-related mortality was excluded and this difference in mean survival rates could have significant consequences for long-term population growth. Under natural conditions, adult female survival is characterized by higher survival rates and lower variation. In the Nottaway, Témiscamie, and Assinica sub-populations in Québec, Rudolph et al. (2012) calculated female survival at 87% with and 90% without hunting effects for the Témiscamie and Assinica sub-populations in Québec; at this rate, their models predicted declining adult survival over time (Table 3).
Recruitment rates (often expressed as # calves per 100 females) are highly variable (Table 3). In the Alberta populations, mean recruitment from 2001 to 2008 was 18 calves:100 females, but ranged from 3 to 52 calves:100 females. Populations in southern parts of BP range generally have rates below a sustainability threshold (i.e., 29 calves per 100 females) while rates in northern parts of the range are higher (i.e., 36 calves per 100 females (range: 17-67) in the NT (Larter and Allaire 2014).
Fluctuations and Trends
Newfoundland population
Dramatic population fluctuations have occurred in the NP since surveys were first conducted. Anecdotal estimates on the NP date to the early 19th century and historic documents show that the NP peaked in the early 1900s at an estimated 100,000 individuals and declined to 10,000-15,000 between 1925 and 1935 (Peek et al. 2012). Systematic surveys and research began in the 1950s. The NP slightly increased from 1930 to 1950 but did not approach the same abundance as in the early 1900s, as suggested by historical hunting records and ecological knowledge (Peek et al. 2012). The NP increased until about 1975 when the population reached approximately 22,500 individuals (16,589 mature) (Peek et al. 2012) and from this point, the population increased to 94,000 in 1996 then declined to 68,000 in 2002, and further to approximately 33,000 Caribou in 2012 (Weir et al. 2014) (Figure 7). This represents a decline of 58% and 65% in the past 12 and 18 years respectively (2 and 3 generation times; see Life Cycle and Reproduction section). Similar patterns were observed for most individual sub-populations in the NP.
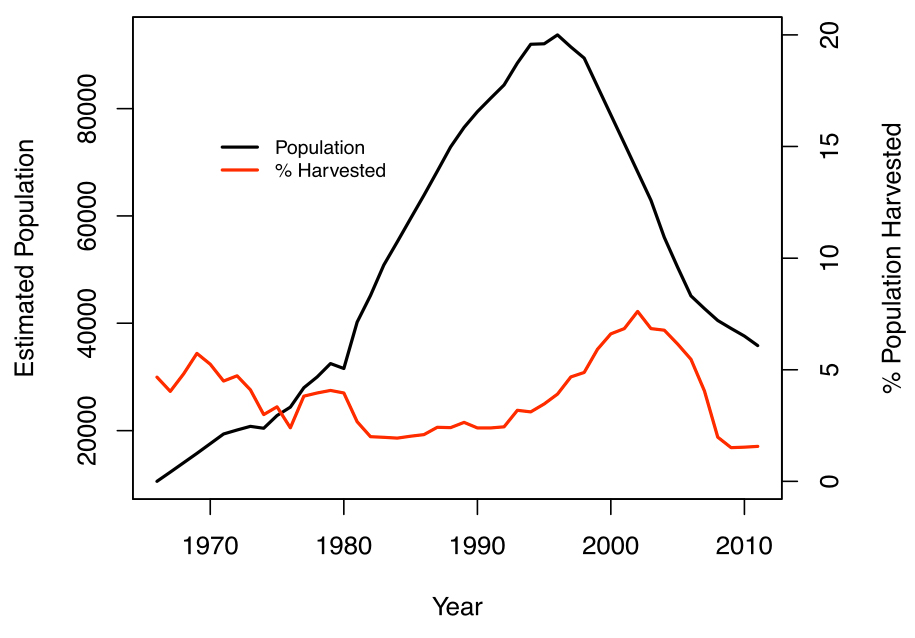
Long description for Figure 7
Chart illustrating changes in the population abundance of Newfoundland population (NP) Caribou and hunting harvest rate from 1966 to 2011. The NP increased until about 1975 when it reached approximately 22,500 individuals. From this point the population increased to 94,000 in 1996 then declined to 68,000 in 2002 and to approximately 33,000 in 2012. The highest rates of harvest (5.9 to 7.6 percent from 1999 to 2005) coincide with the period of rapid population decline.
It is believed that the decline of Caribou in Newfoundland is part of a natural fluctuation and that population size will increase (Bastille-Rousseau et al. 2013; Lewis and Mahoney 2014; Weir et al. 2014). The decline was related to density- dependence effects associated with poor range conditions (Bastille-Rousseau et al. 2013), and excessive hunting levels occurring as the population was beginning to decline (Weir et al. 2014). The conclusion that density-dependence effects exist is based on decreased calf survival as the population increased and increased calf weight when Caribou density was lower, both of which suggest populations were being limited because of high Caribou densities. Increased calf weight was likely due to better condition of breeding females (Trindade et al. 2011; Weir et al. 2014), and larger calves had a better chance of survival (Mahoney and Weir 2009; Lewis and Mahoney 2014). Other indices suggestive of density-dependence effects included altered habitat use (Mahoney and Schaefer 2011), as well as increased tooth wear and body morphology changes (Mahoney et al. 2011). Changes to the timing of annual migrations also were noted during high and lower density periods, with some sub-populations spending 5 weeks less on summer range when density was high, possibly due to competition for limited forage (Mahoney and Schaefer 2002b; Weir et al. 2014). Body size of female Caribou decreased through time, and some sub-populations exhibited substantial reductions in male stature and antler size, changes typically associated with limited forage quality and quantity (Mahoney and Schaefer 2002b; Mahoney and Weir 2009). Peek et al. (2012) concluded that although predation is the main proximate cause of the NP decline, density-dependence through summer range deterioration may constitute an ultimate cause.
Hunting is also believed to have been a factor in the recent decline. Between the 1960s and 2012, 3.8% of the population was harvested annually (Figure 7; Weir et al. 2014). However, hunting in the early years of the population decline exacerbated the decline. Licence sales increased steadily until 2004, resulting in an 8-year lag from the population peak to quota reduction that resulted in the highest rates of harvest (5.9%–7.6% from 1999 to 2005) during the period of rapid population decline. The overall rate of harvest peaked at > 12% for some individual sub-populations, and > 18% when considering only the adult male segment of the population (Weir et al. 2014). Mean harvest rate was highest (7.6%) during the steepest rate of decline (1999-2005), and maximum harvest rates were recorded in sub-populations such as La Poile at 12.3% in 2004 and Grey River at 17.8% in 2007 (Weir, J. and Luther, G., NLDEC, unpub. data).
The future trend in the NP has been predicted based on scenarios with, and without, increased calf recruitment. Leslie matrix population models, which use calf survival, productivity, adult survival, and age of breeding females to forecast future population abundance, suggested that if calf recruitment remained low, the NP would decline by 90% to approximately 5,074 individuals by 2035, whereas an increase in recruitment following higher calf survival would stabilize population change or initiate population increase by 2035 (Weir et al. 2014; Figure 8). Based on these models, a 40-45% annual calf survival is required to stabilize the NP. Note that the average calf survival during the population increase period (1980-1996) was 67.4 +/- 9.1% (Lewis and Mahoney 2014).
The rate of decline is slowing, with a decline of 9%/year between 2000 and 2006 to 5%/year since 2007, and this change is likely due to improved calf survival and reduced hunting pressure (Weir et al. 2014). Calf survival has increased to 48% in 2012 and is expected to be stable (see Survival and Recruitment section). Body size (jawbone length and antler size) have increased since the mid-2000s and have returned to pre-decline levels (Weir et al. 2014). Weight of male and female calves has returned to pre-decline levels (Weir et al. 2014).
In summary, the present decline is density-dependent in nature and believed to ultimately be due to nutritional stress from food limitation. Caribou calf vulnerability increased with density, allowing exploitation by predators to be a driving mechanism of decline. The rate of population decrease was exacerbated by hunting in the early part of the decline, but quotas have since been decreased. Various indices suggest a healthier population and that the decline is slowing and may be halting.
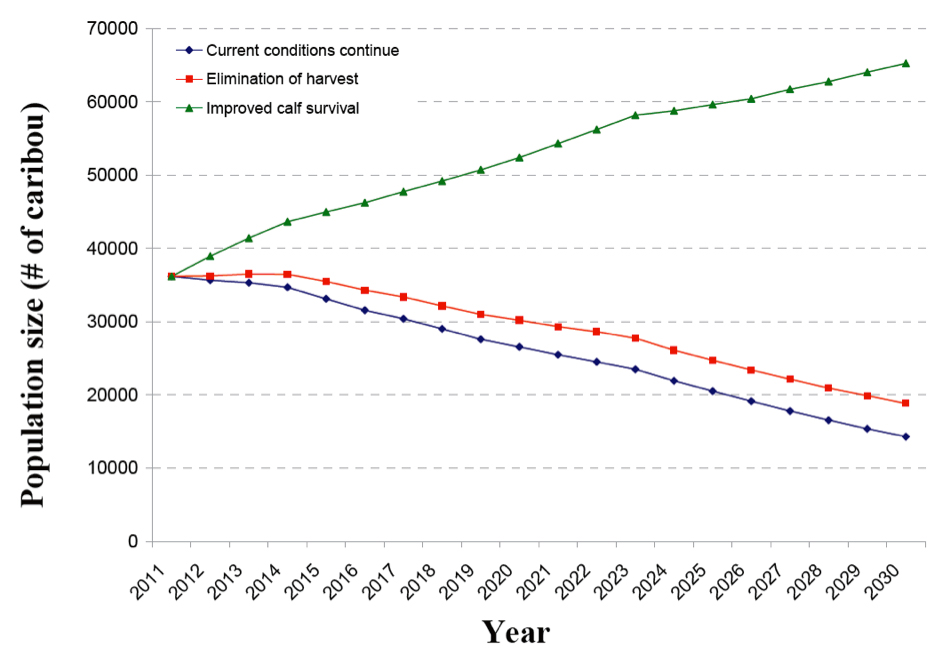
Long description for Figure 8
Chart illustrating a population simulation for the Newfoundland population (NP) of Caribou using Population Viability Analysis. Three projections of population size are shown for the years 2011 to 2035: one assumes continuing current low calf survival rates, another assumes elimination of hunting, and the third assumes improved calf survival rates. The projection that assumes improved calf survival is the only one showing an increase in Caribou numbers.
Gaspésie population
The GP has declined since population size was first estimated in the 1950s at 700-1,500 individuals (calves and mature animals combined) (Moisan 1958). The population was estimated at 274 (219 mature) in 1983, declined later to a low of 100 (88 mature) in 1999, and increased again to 214 (174 mature) animals in 2007 (St-Laurent pers. comm.; Figure 9). The GP was at its lowest recorded levels in recent years, with estimates of 92 (89 mature) animals in 2011, and 85 (65 mature) animals in 2012. A population trend is difficult to assess over any single time period because the fluctuations mean that comparisons within the dataset can yield dramatic increases or decreases. The two-generation trend (12 years; see Life Cycle and Reproduction section; 2001-2013) is highly variable depending upon which 12-year period is used. An averaged rate over 5, 12-year periods from 1997-2013 was -10% (range: +1, to -47%). Similarly, an averaged rate over 5, 18-year periods from 1991-2013 for the three generation trend is -11% (range: +15, to -34%). The trend since 1983 has been -49%. The approximate decline since the 1950s likely is 81-91% (i.e., 700-1500 Caribou in the 1990s, to 130 Caribou in 2013).
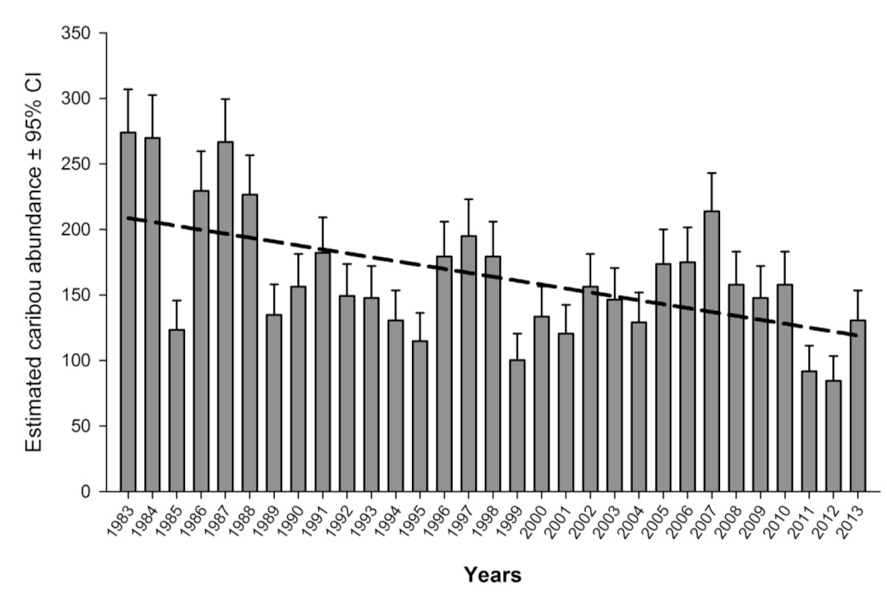
Long description for Figure 9
Chart illustrating autumn population estimates for all ages of Caribou within the Atlantic-Gaspésie (GP) population from 1983 to 2013. The population was estimated at 274 in 1983, declined to a low of 100 in 1999, and increased to more than 214 animals in 2007. The GP was at its lowest recorded levels in recent years, with estimates of 92 animals in 2011 and 85 animals in 2012.
The future trend in the GP has been estimated using a population viability analysis (PVA). Lesmerises (2012) demonstrated that the average time to extinction would be 20.5 years (± 6.7 years) if calf mortality of the GP remains at 89%, which is the mean level reported in 2009-2011. No iteration goes beyond 2057 with this modelling scenario. This PVA suggested that calf survival must remain between 41 and 43%, or approximately 33 calves:100 females to assure sustainability for the next 100 years, which is similar to the rate of 40 to 45% reported for the NP population (Randell et al. 2012). Lesmerises (2012) also demonstrated the importance of adult survival on the GP trends, but results should then be interpreted with caution as some missing data from this analysis was acquired from other Caribou populations. Recent surveys suggested that adult mortality could be higher than expected, and that adult females could be more vulnerable to predation by Coyote (M.-H. St-Laurent, unpubl. data).
Boreal population
The % change trend in the last 3 generations (18 years – see Life Cycle and Reproduction section) for the BP population as a whole is unknown. A large part of the DU remains unsurveyed, and surveys within ranges over long periods are difficult to compare with one another. The last status report (COSEWIC 2002) estimated there were 33,000 Caribou, which is similar to the 2012 National Recovery Report (Environment Canada 2012) estimate of 34,000 BP Caribou. These are considered to be crude estimates of population size, particularly in the large areas of northern ranges, where abundance is believed to be high (i.e., 6,500 in NT, and 7,000 in Quebec; Table 1). An earlier (early 2000s) crude estimate for four sub-populations in southern James Bay was 700 Caribou (Rudolph et al. 2012). Some regions have undertaken new surveys since the National Recovery report, but trend data is still difficult to obtain. For example, in BC, a recent (2013) minimum population count of all ranges recorded 952 Caribou (816 adults), which is a decline of 37% (all ages) from the 2004 estimate, and a decline of 29% from the lower confidence interval estimate (Culling and Culling 2013). However, population density may have been overestimated for some ranges used in the 2004 estimate, and comparisons are thus difficult. Even though percent decline is equivocal in BC, a decline still appears evident because 6 of 8 ranges and sub-ranges had unsustainable calf recruitment rates.
Trend data using population growth rate exists for some jurisdictions and indicates population decline in almost all ranges (Table 5). Lambda values for a period approximating the last 3 generations (1996-present) are available for 37 sub-populations covering 46 time periods. A ‘time period’ is a short period of several years, which was used to limit masking of possible years where Lambda was positive. The sub-populations represent > 90% of the BP range. (Note: some sub-populations do not correspond to population ranges in the National Recovery Strategy, i.e., BP South and BP North in NT are separated here, and the five sub-populations in Saskatchewan equate to a single population range in the National Recovery Strategy). Only some Labrador sub-populations in some years, and a single population (northern NT section range), indicated population growth. In the Sahtu Settlement Area in the NT, there is a general consensus among Aboriginal users that local Caribou populations are currently healthy, as reported by SRRB (2010). In Labrador, even with higher Lambda values, the Red Wine Mountain sub-population has not recovered from a major decline during the 1990s (Schaefer et al. 1999; Schmelzer 2013) and a recent assessment of the Mealy Mountain and Lac Joseph sub-populations also indicates decline (Schmelzer 2013).
| Province/ Regions | Sub-populations | Years Notevof Table 5 | Lambda (range) Notewof Table 5 | Source |
|---|---|---|---|---|
| Alberta | 13 sub-populations | 2001-08 | 0.93 (0.69-1.19) | LINK NOT WORKING http://www.albertacariboucommittee.ca/ |
| Alberta | Bistcho | 5 years | 0.84 (-58 %) Notezof Table 5 | Hervieux et al. 2013 |
| Alberta | Chinchaga | 10 years | 0.89 (-70%) | - |
| Alberta | Cold Lake | 12 years | 0.86 (-87%) | - |
| Alberta | Cold L. Sask | 12 years | 0.92 (-63%) | - |
| Alberta | Caribou Mtn | 17 years | 0.92 (-76%) | - |
| Alberta | ESAR | 17 years | 0.92 (-78%) | - |
| Alberta | Little Smoky | 13 years | 0.97 (-33%) | - |
| Alberta | Red Earth | 15 years | 0.88 (-84%) | - |
| Alberta | Richardson | 3 years | 0.98 (-5%) | - |
| Alberta | WSAR | 18 years | 0.93 (-71%) | - |
| Alberta | Yates | 5 years | 1.00 (+1%) | - |
| Alberta | Average | 1994-12 | 0.92 (- 57%) | - |
| NT | Dehcho | 2005-14 | 0.97 (0.72-1.29) | Larter and Allaire 2014 |
| NT | South Slave | 2004-10 | 0.96 (0.83-1.06) | Kelly and Cox 2011 |
| NT | Gwich'in S. | 2003-07 | 1.08 | Nagy 2011 |
| NT | Gwich'in N. | 2005-07 | 1.2 | Nagy 2011 |
| NT | BP South | - | 0.87-0.97 | - |
| Sask. | Mossy | 1993-96 | 1.1 | Rettie and Messier 1998 |
| Sask. | Montreal | 1993-96 | 0.91 | - |
| Sask. | Nemeiben | 1993-96 | 0.84 | - |
| Sask. | Clarke | 1993-96 | 0.92 | - |
| Sask. | Weyakin | 1993-96 | 0.98 | - |
| Sask. | Avg. Noteaaof Table 5 | 1993-96 | 0.95 | - |
| Manitoba | N. Interlake | 2005-09 | 0.90 (0.82-0.99) | Hettinga et al. 2012 |
| Ontario | Berens | 2011-12 | 0.93 (0.89-0.98) | OMNR 2014 |
| Ontario | Sydney | 2011-12 | 0.98 (0.97-0.99) | - |
| Ontario | Churchill | 2011-12 | 0.96 (0.94-0.98) | - |
| Ontario | Brightsand | 2010-12 | 0.87 (0.86-0.90) | - |
| Ontario | Nipigon | 2009-12 | 0.98 (0.96-0.99) | - |
| Ontario | Pagwachuan | 2009-12 | 0.94 (0.72-1.05) | - |
| Ontario | Kesagami | 2009-12 | 0.94 (0.88-1.01) | - |
| Ontario | Spirit | 2008-11 | 0.95 (0.82-1.06) | - |
| Ontario | Kinlock | 2008-11 | 0.95 (0.91-1.01) | - |
| Ontario | James Bay | 2008-13 | 0.91 | - |
| Ontario | Missisa | 2008-13 | 0.86 | - |
| Ontario | Average | 2008-13 | 0.93 (0.82-1.06) | - |
| Québec | Assinica | 2003-12 | 0.98 | Rudolph et al. 2012 |
| Québec | Nottaway | 2002-11 | 0.98 | - |
| Québec | Temiscamie | 2002-12 | 0.98 | - |
| Labrador | Mealy Mtns. | 1960 | 0.78 Notexof Table 5 | Schmelzer 2013 |
| Labrador | Mealy Mtns. | 1963 | 0.81 Notexof Table 5 | - |
| Labrador | Mealy Mtns. | 1970-71 | 1.0 (0.99-1.02) Notexof Table 5 | - |
| Labrador | Mealy Mtns. | 1974-75 | 0.86 (0.69-1.08) Notexof Table 5 | - |
| Labrador | Mealy Mtns. | 1981 | 1.16 Notexof Table 5 | - |
| Labrador | Mealy Mtns. | 1987 | 1.18 Notexof Table 5 | - |
| Labrador | Mealy Mtns. | 2002 | 1.17 Noteyof Table 5 | - |
| Labrador | Mealy Mtns. | 2005-06 | 1.12 (1.11-1.13) Noteyof Table 5 | - |
| Labrador | Mealy Mtns. | 2008-10 | 1.14 (1.07-1.25) Noteyof Table 5 | - |
| Labrador | Mealy Mtns. | 2011-12 | 1.06 (1.04-1.08) Noteyof Table 5 | - |
| Labrador | Average | 2002-12 | 1.13 (1.07-1.25) Noteyof Table 5 | - |
| Labrador | Red Wine | 1997 | 0.69 Noteyof Table 5 | - |
| Labrador | Mtns. | 2001-03 | 1.03 (0.88-1.12) Noteyof Table 5 | - |
| Labrador | Mtns. | 2009 | 1.1 Noteyof Table 5 | - |
| Labrador | Average | 1997-09 | 1.05 (0.69-1.16) Noteyof Table 5 | - |
| Labrador | Lac Joseph | 1984-86 | 1.2 (1.16-1.19) Notexof Table 5 | - |
| Labrador | Lac Joseph | 1998 | 0.95 Noteyof Table 5 | - |
| Labrador | Lac Joseph | 2000-02 | 0.99 (0.84-1.18) Noteyof Table 5 | - |
| Labrador | Lac Joseph | 2003 | 0.87 d | - |
| Labrador | Lac Joseph | 2005 | 0.97 Noteyof Table 5 | - |
| Labrador | Lac Joseph | 2007-09 | 1.15 (0.88-1.31) Noteyof Table 5 | - |
| Labrador | Average | 1998-09 | 1.08 (0.84-1.3) Noteyof Table 5 | - |
The averaged Lambda for the 37 subpopulations is 0.96, indicating population decline (Table 5). The declines may be short term but this is unlikely because data sets covered short, 2-4 year periods and positive Lambda would be noted. Negative Lambda existed in 30 sub-populations (81% of all sub-populations with data) and 30 time periods. In Alberta, enough data from 998 radio-collared Caribou in 11 of the 13 Albertan ranges indicated 10 ranges were in decline, with mean adult female survival of 85%, mean number of 14.5 calves/100 females, and mean λ = 0.92 (Hervieux et al. 2013). In Ontario, Lambda values indicate a decline in all 11 ranges with data, and a mean Lambda of 0.93, from 2008-2012 (OMNR 2014). Lambda is < 1 in regions with highly disturbed ranges (e.g. Alberta) but Lambda also was lower in areas considered to be relatively less disturbed (e.g., Ontario and Québec, southern NT). A Lambda of < 0.90 equates to a 50% decline in 7 years; 9 sub-populations over 11 time periods had Lambda < 0.9. In Alberta, the mean Lambda of 0.93 for the 11 sub-populations equated to a 57% cumulative decline from 1994-2012.
The present and future trend in the BP can also be inferred from a recent assessment of population size, size of range, and disturbance levels. The Federal Recovery Strategy (Environment Canada 2012) identified 51 local populations (see Designatable Units section) and assessed the likelihood of 50 ranges to maintain Boreal Caribou (1 range [SK1] in northern Saskatchewan was omitted from assessment because it is a unique situation and lacked critical habitat analysis; Environment Canada 2012). A self-sustaining local population is a local population of Boreal Caribou that on average demonstrates stable or positive population growth over the short-term (≤20 years), and is large enough (minimum of 10 females) to withstand stochastic events and persist over the long-term (≥50 years), without the need for ongoing active management intervention (Environment Canada 2012). A total of 14 local populations were predicted to be ‘self-sustaining’ (65% of entire range), while 26 (13% of entire range) were ‘likely not self-sustaining’, and 10 (22% of entire range) are ‘as likely as not self-sustaining’ (Table 2). The classification of ‘as likely as not self-sustaining’ indicates uncertainty about the viability of the population; because of this uncertainty such ranges are not considered as part of the contribution towards population objectives in the National Recovery Strategy, and are labelled as ‘not self-sustaining’ in the Strategy (Environment Canada 2012). Population estimates are available for 40 local populations, usually expressed as a minimum and maximum estimate. Most (69% of maximum population size estimate) of the known Caribou population was predicted to exist in 9 self-sustaining local populations, while most of the local populations (25; 13% of the total population) were not self-sustaining, and 6 local populations (14% of total population) were classified as likely as not self-sustaining. The ‘likely as not’ ranges exceed the disturbance management threshold of >35% disturbance (Figure 10) and based on the precautionary principle are considered in this assessment to likely experience population declines. These ranges are combined with the ‘likely not self-sustaining ranges’ for a total estimate of amount of area and population in decline. Therefore, 35% of the BP range area, and 27% of the population are inferred to be in decline. The proportion may be higher; a 2014 fire event burned 3 million hectares in NT and there is concern that the range is not as self-sustaining as it is presently assessed (Boyan and Carrière pers. comm. 2014).
Long description for Figure 10
Chart illustrating the relationship between the amount of disturbance and the probability of a Caribou subpopulation within the Boreal population being self-sustaining. Certainty of outcome, ecological risk, and management scenarios are illustrated along a continuum of conditions.
Based on the National Recovery Strategy, Lambda values, and recent population updates, ranges in most jurisdictions along the southern portion of the DU are in population and habitat decline, such as Alberta (all 9 ranges, monitored from 2001-2011), Manitoba (1 range, 2005-2009), Ontario (7 ranges, 2009-2013), Québec (3 ranges, 2002-2012), and Labrador (2 ranges, 2002-2012) (Table 2, 5). A more recent assessment of BP range in Ontario (OMNR 2014), suggests that only 2 of 13 ranges (15% of total BP area, and 19% of minimum population count, in Ontario) have Lambda > 1.0, a decrease of 3 ranges and 105,784 km2, or 7% of the 65% of total BP range that is defined as self-sustaining in the National Recovery Strategy (Environment Canada 2012). A similar issue may apply to Quebec; research on three sub-populations in a large area (~ 112,000 km2) southeast of James Bay indicates population decline even when disturbance levels are at the 35% maximum threshold (Rudolph et al. 2012).
In summary, approximately 1/3 of the area and population of BP Caribou are in decline, based on recorded declines, negative Lambda for most sub-populations, and the percentage of ranges that have too much disturbance to be self-sufficient. Lambda rates indicate declines of up to 50% are likely to continue in the most affected areas, and lesser declines will occur in sub-populations with negative Lambda. An ongoing decline of > 30% is inferred.
Rescue Effect
As per COSEWIC guidelines, rescue effect can only occur within a DU; rescue of one DU by another DU is not supported for purposes of status assessment. All three DUs are endemic to Canada, therefore rescue of outside populations within the same DU is not possible. Rescue of isolated ranges within a DU is possible but these events relate to recovery, rather than status, and are not discussed in this report.
Threats and Limiting Factors
Boreal Population
Threats of high concern identified in the National Recovery Strategy (Environment Canada 2012) for BP are habitat alteration from anthropogenic disturbance, and predation. Threats of medium concern are habitat alteration from fire, climate change that increases fire, hunting, and noise disturbance. Jurisdictions with recovery plans typically identify the same threats (e.g., BC Ministry of the Environment 2010; Labrador (Schmelzer et al. 2004), Manitoba (MBWCMC 2014). Some threats interact and can have cumulative impacts that may not be evident when threats are examined individually (Weclaw and Hudson, 2004; Boreal Caribou ATK Reports, 2010-2011; Badiou et al., 2011; Environment Canada 2012).
The National Recovery Strategy for the BP identifies a disturbance management threshold of 65% undisturbed habitat in a range as a measurable probability (60%) for a local population to be self-sustaining. A population of 100 animals provides a 0.7 probability of not reaching a quasi-extinction threshold of less than 10 reproductively active females under stable conditions (Environment Canada 2011; Figure 10). The 65% threshold is considered a minimum threshold because at 65% undisturbed habitat, there remains a significant risk (40%) that a local population will not be self-sustaining (i.e., < 10 breeding females) (Environment Canada 2012). The definition of disturbed habitat is the presence of fire < 40 years old and or anthropogenic disturbance visible on Landsat at a scale of 1:50,000, including habitat within a 500 m buffer of the anthropogenic disturbance (Environment Canada 2012). Figure 11 uses BP range in Ontario to illustrate the extent of disturbance when both anthropogenic and natural disturbances are mapped, following the methodology in Environment Canada (2012).
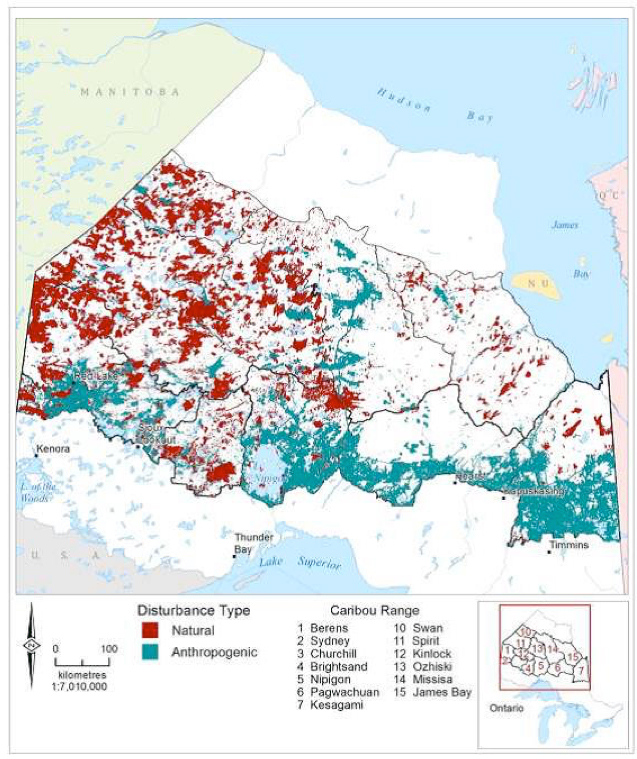
Long description for Figure 11
Map illustrating the extent of disturbance in the Ontario range of the Boreal population of Caribou. The map shows areas of forest cover of less than 36 years of age due to natural (fire, blowdown) and anthropogenic (resource extraction, roads) disturbance.
Sorensen et al. (2008) identified a strong negative relationship between recruitment rate of Caribou in Alberta and the level of disturbance from both anthropogenic and natural origin. Sleep and Loehle (2010) questioned the simplicity of these models and suggested that they overestimated Caribou population growth rate and possessed low predictive power. The Science Review for the Identification of Critical Habitat conducted by Environment Canada (2008) extended Sorensen et al.’s (2008) study. This meta-analysis was further improved by Environment Canada (2011) and their best model (i.e., fire + 500 m buffered anthropogenic disturbance, exclusion of reservoirs) explained 69% of the variation in calf recruitment across a sample of 24 Caribou ranges based on the percent total disturbance in each range. The Hervieux et al. (2013) method, which was based on known survival rates of females and calves (see Sampling Effort and Methods section), came to the same conclusion on extent of declines as the method used in the National Recovery Strategy, suggesting there is value in the disturbance threshold method of Environment Canada (2011, 2012).
The IUCN Threats calculator for BP indicates an overall threat score of Very High-High (Appendix 1). The High threats were Logging and Predation.
High and High-Medium Threats
Logging and Wood Harvesting (IUCN 5.3)
Loss of mature forest (mainly by harvesting, but also mining, hydroelectric and oil and gas development) is the most pervasive method of habitat alteration in the boreal forest (Gagnon and Morin 2001; McRae et al. 2001; Burton et al. 2003). ATK states that the BP has altered their diet in response to forestry disturbance and, as a result, their meat has a different taste (McDonald et al. 1997; Huntington et al. 2005, cited in COSEWIC 2012, p. 65).
The limit of timber harvest is moving northwards to reach unharvested forests, thereby increasing access to other resource development via road construction (Festa-Bianchet et al. 2011). Local forestry expansion rates are unknown but declines in Caribou range of 8-36 km/decade correspond to resource development (see Canadian Range section). Industrial forest activity has been expanding northward into BP range. Provincial land planning exercises in the last 20 years (e.g., Ontario Lands for Life; Watton and Dunn 2003) and auctioning of large areas (e.g., 68,000 km2 forest management agreement in northeastern Alberta in 1991; Alpac 2014) have extended industrial forest management northward. Much of the disturbed land identified in the National Recovery Strategy (Environment Canada 2012) corresponds with the location of the new forestry licences. In Labrador, forest harvest operations as of 2004 were restricted to a small portion of the Red Wine Mountain range but further harvest has been proposed within the range (Schmelzer et al. 2004).
The northward extension of large-scale industrial forestry is limited by provincial government forest licence planning restrictions (Greenpeace 2014). However, industrial forestry has not yet reached these northern limits (Figure 12) and impacts of forestry on Caribou are projected into the future. The long-term (i.e., 25-year) leases require companies to practise sustained yield harvest; areas harvested will eventually be re-harvested, concurrent with maintained access roads. In Quebec, Leblond et al. (2014), using an expert-based habitat suitability model, showed that very few suitable habitats are still available for caribou below the northern limit of commercial forestry.
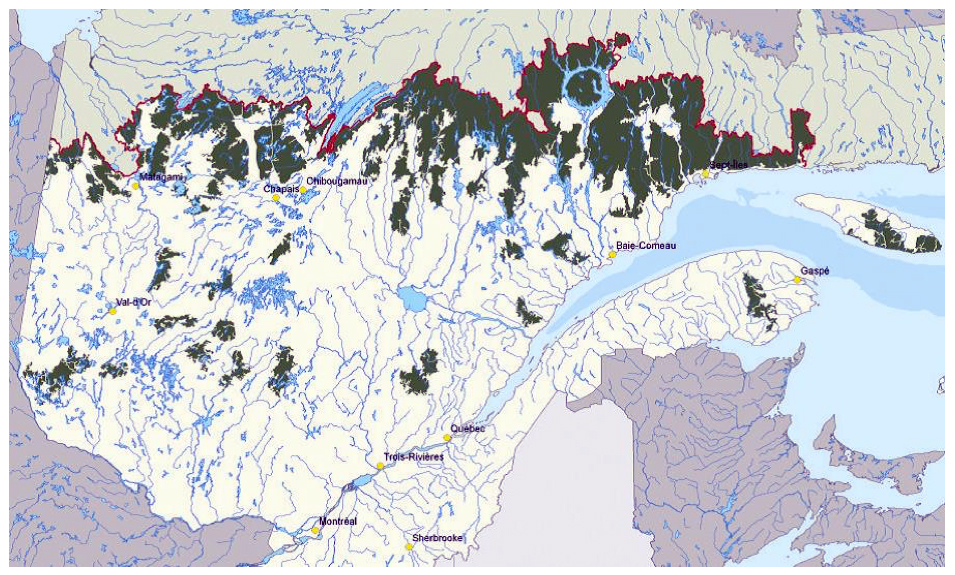
Long description for Figure 12
Map illustrating the location of the northern limit of industrial forestry in Quebec and remaining forested land existing in blocks of greater than 500 square kilometres as of 2009.
Caribou appear to be sensitive to noise from snowmobiles, motorized boats, vehicles, and aircraft (e.g., low-level flight training in Labrador, Schmelzer et al. 2004), which may increase chronic stress. Renaud (2012) recorded higher cortisol levels in Caribou with higher percentage of clear-cut harvest in their range compared to Caribou in undisturbed habitats. Caribou have been shown to flee from areas with noise disturbance from these sources (COSEWIC 2012). SRRB (2010) reported that noises, including industrial noises, may represent a threat to Caribou:
“Noise is given as a major factor that impacts boreal woodland caribou. These disturbances include drilling, seismic cut-line activities, slashing, and machines including helicopters and all-terrain vehicles that conduct work during the summer. Caribou prefer old growth areas and have a tendency to stay away from winter roads because of noise pollution.”
In Labrador, the Red Wine Mountain sub-population was exposed to low-level military jet training in the 1990s-2000s, which was associated with changes in movement patterns (Harrington and Veitch 1991; 1992) and lower calf survival (Harrington and Veitch 1992). Some Aboriginal experts reported to COSEWIC (2012) that Caribou were more likely to be initially frightened by industrial or vehicular noises but eventually habituated over time.
Problematic Native Species (IUCN 8.2) - predation
Caribou declines have been associated with increased predation rates, which are associated with anthropogenic disturbance (see Habitat Trends section). Leblond et al. (2013b) showed that the probability of an adult Caribou dying from predation increased with the proportion of recent disturbances in their annual home range. Changing predator-prey dynamics are associated with density of alternate prey species increasing after forest clearing (i.e., apparent competition; see Habitat Trendssection). Although predation is a natural process, predation rates appear to be unsustainable in disturbed parts of the BP range.
Increased predation rates via apparent competition are widely accepted as the primary driver of BP decline across disturbed parts of their range (e.g., Cumming et al. 1996; Rettie and Messier 2000; James et al. 2004; Courbin et al. 2009; Whittington et al. 2011) although alternative hypotheses are being studied (see Change in Predation Rates section).
Apparent competition within the BP range typically involves Wolves as the primary predator and Moose as the alternative prey species, although White-tailed Deer constitute alternative prey species in some regions (e.g., Alberta; Latham et al. 2011b). Black Bear also are an important predator; Leclerc et al. (2014) recorded that Black Bear depredation accounted for 52% of Caribou calf deaths in the Charlevoix and Saguenay region of Quebec. In areas with a large industrial footprint, Nagy (2011) suggested that aggressive management actions, including predator control, may be required to provide secure habitat for Caribou. In Alberta, 733 Wolves in the Little Smoky Caribou range were culled from 2005 to 2012 to help with Caribou recovery. Predator control in this area is ongoing (Smith and Pittaway 2011; Hervieux pers. comm.) but has not resulted in population increase, suggesting habitat management also is necessary (Hervieux et al. 2014).
Medium – Low Threats
Energy Production and Mining (IUCN 3)
Few studies have examined the effects of mining operations on Caribou. Information from the migratory Bathurst sub-population (DU 3) in the NT may be relevant. In this study, the combination of direct (i.e., physical footprint) and indirect (i.e., noise, dust, and other sensory disturbances) effects created a ZOI around a mine site that can change the behaviour and occurrence of the animals. Studies in the vicinity of two diamond mines north of Yellowknife in the NT showed that migratory Caribou occurrence decreased with increasing proximity to the mine (Boulanger et al. 2012). Studying the same migratory Caribou sub-population, Johnson et al. (2005) reported that habitat quality was most severely reduced during the post-calving season; modelled coefficients effected a 37% reduction in high-quality habitats, and an 84% increase in low-quality habitats.
Wilson et al. (2010) modelled management actions with different scenarios for the next 50 years that could mitigate the impacts of petroleum and natural gas (PNG) development on the BP in British Columbia. In the scenario simulating no further PNG exploration and development (i.e., equivalent to a moratorium on future PNG industry), the estimated Caribou population would be approximately stable at 1300 animals although two local ranges (out of six) could be extirpated. In the scenario wherein only committed PNG tenures (i.e., accepting no new tenures for the future) were allowed to proceed, the BC sub-population would be about 800 animals in 50 years, and three local ranges would have high probability (>40%) of extirpation. The third scenario modelled a situation wherein no deferrals or management actions were implemented (i.e., PNG development continued to increase as in recent years). In this scenario, the BP would contain 250 Caribou and there is a very high probability (>60%) that they would be extirpated from all but one range (Maxhamish range).
Although also present in BC and Saskatchewan, petroleum and natural gas (PNG) infrastructures are more frequent in Alberta, where some form of PNG activities is present in nearly all remaining Caribou ranges. In Alberta, PNG infrastructures and forest harvest are frequently conducted in close proximity to one another; the resulting degree of disturbance in Caribou habitat is often significant, as in the Little Smoky Caribou sub-population range, which is 95% disturbed and at immediate risk of extirpation (Schneider et al. 2010).
As of 2008, there were 105 active mines in the Boreal Forest, covering 44% of all mines in Canada, and an area of 583,000 km2 (BorealCanada 2008). In Quebec, for example, expenditures for mining exploration and appraisal were approximately $200 million in 2003, and > $800 million in 2011 and 2012 (Government of Quebec 2014). Predicting the location and extent of future mining activity is difficult because development of new mines is strongly dependent on the changing market value of minerals. Mining claims indicate potential development, rather than a confirmed threat. Figure 13 illustrates potential location of mining relative to Caribou distribution and protected areas in Canada.
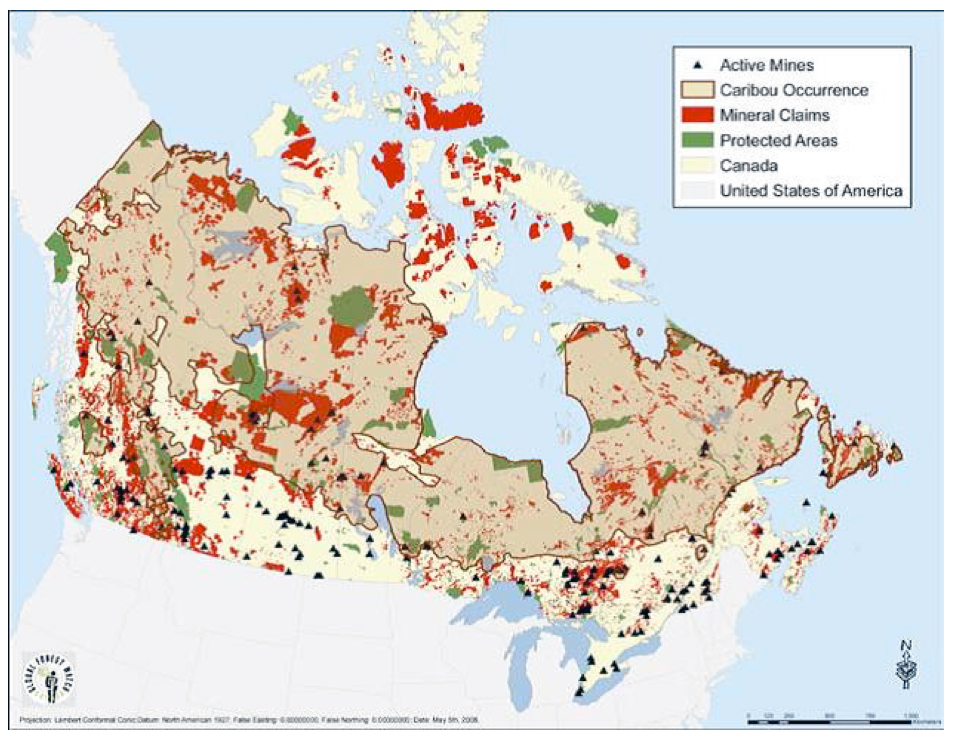
Long description for Figure 13
Map illustrating the potential location of mining (active mines and mineral claims) relative to Caribou distribution and protected areas in Canada.
In BP range on Tłįchǫ territory in NT, a proposed nickel copper mine will result in the south NT range (Environment Canada 2011) exceeding the 65% disturbance threshold for sustainability. Most previous disturbance had been related to fire (MVRB 2013). In the Sahtu Settlement Area in the NT, an ATK process noted that many changes are occurring that could impact Boreal Caribou and cited climate change and industrial activities as causes for concern SRRB (2010).
Peat mining is a growing industry in northern Alberta and Saskatchewan, with numerous proposals and permit applications ongoing. The removal of peat would represent a loss of habitat, as well as a source of road and infrastructure development.
Hydroelectric projects may result in habitat loss due to flooding, infrastructure and linear features. For example, the Red Wine Mountain sub-population in Labrador occurs in the Lower Churchill Falls project, which will flood 126 km2 of forest, create 345 km of temporary roads for the estimated 10-year construction period and 30 km of permanent roads connecting to the Trans-Labrador Highway, as well as 263 km of transmission lines (Nalcor 2013). Habitat suitability analysis using a ZOI (see Habitat Avoidance section) of 2-4 km width suggested a cumulative impact to 16% of primary calving range, and 22% of primary winter range (Nalcor date unknown). Numerous hydroelectric projects are proposed; for example, in Ontario, 10 facilities are planned for the region west of James Bay in the next 15 years (Carlson and Chetkiewicz 2013).
Linear Features (Roads, utility lines; IUCN 4.1, 4.2)
Direct mortality as roadkill is not a significant threat from roads. Linear features, such as roads and seismic lines, represent both functional habitat loss because Caribou appear to avoid them, and predation risk because mortality is increased (see Habitat Avoidance section). Building of new roads during industrial development provides access to people using trucks, snowmobiles, or ATV to a new territory, which represents an additional source of disturbance and increased hunter presence in Caribou range where hunting is permitted.
Projected road density is unknown over the large BP range. Many roads are associated with mining development but specific projects are dependent on economic factors and the certainty of projects is difficult to predict. However, there is expected growth in mining and forestry activities throughout much of BP range and it is likely that road density will increase. In Manitoba, for example, construction is underway on a 1000 km, all-season road network to connect remote communities in the region east of Lake Winnipeg, and there are proposals for connecting many northern Manitoba communities (Government of Manitoba 2014). Another indication of potential disturbance in BP range is the 2011 (renewed in 2014) Plan Nord, an $80 billion proposal for investment in roads, airports, mining, and forestry in central and northern Quebec (Northern Miner 2014). In 2014, $63 million was budgeted to extend access along the north shore of the St. Lawrence (Highway 138) and repair Highway 389 in the Côte-Nord region (Baie-Comeau to Labrador) and the James Bay Highway (Matagami to Radisson) (Resource Clips 2014).
Hunting (IUCN 5.1)
In the NT, evidence suggests that the current harvest of the BP is relatively low because most Aboriginal communities rely primarily on Barren-ground Caribou (DU3), or Moose for sustenance. Aboriginal harvest rates are not believed to be significant in NT (NT Species at Risk Committee 2012). Sport hunting of Boreal Caribou has been banned across the range, first in Ontario (in 1929), then other provinces in the last 20 years; Alberta (1985), Saskatchewan (1987), BC (2001), Manitoba (2006), NT (Festa-Bianchet et al. 2011). In Québec, sport hunting in Zone 19 (north shore Québec to Labrador) took an average of 56 Caribou annually between 1980 and 2000. This hunt was permanently closed in 2001 and, since then, no sport harvests have directly targeted Boreal Caribou in Québec. However, harvest of migratory Caribou (DU4) is ongoing and the two DUs overlap in some areas for parts of the year. In Labrador, sport hunting was closed in 1986 or earlier, while subsistence hunting was closed in 2002 following listing of Boreal Caribou under the Endangered Species Act.
Illegal and accidental harvest of Boreal Caribou may occur where their range overlaps with the range of migratory Caribou that are hunted in Saskatchewan, Ontario (native hunt only), Québec, and Labrador (Schmelzer et al. 2004; Courtois et al. 2007, Comité de rétablissement du caribou forestier 2007). Subsistence hunting, while closed, continues to occur in Labrador where mortality from hunting represented 29% and 11% of all known mortalities of collared Caribou for Lac Joseph and Red Wine Mountain sub-populations, respectively, between 1997 to 2009 (Schmelzer 2013). In April 2003, 15% of the total Red Wine Mountain sub-population were poached in one incident (Schmelzer et al. 2004). Mortality estimates from poaching of Boreal Caribou are unknown but illegal hunting may be present in some regions. With current declines of large migratory sub-populations in eastern Canada and corresponding hunting closures or reductions, subsistence hunting of Boreal Caribou has increased substantially.
Russell (2011) reported the assessment done by the Labrador Metis:
“The top three threats to boreal woodland caribou in Labrador include: illegal hunting, technology and predation. Concerns regarding overhunting were primarily concerning Innu hunters from Québec and observations of hunting using spotlights and aircraft spotting of animals for hunters….Modern vehicles (skidoos, trucks, planes, satellite tracking & GPS) are thought to provide easier access to caribou for hunting than traditionally used methods by foot or dog team. Generally people are less respectful to caribou than in the past and kill as many as they want without fully utilizing the animal.”
Aboriginal people have unique harvesting rights for fish and wildlife under Canadian constitutional law. However, these harvest rights may be altered by land claim agreements where conservation goals must take precedence (e.g., Labrador Inuit) and some communities agreed to stop or limit their harvest of Caribou. Aboriginal harvests remain a source of mortality for many Boreal Caribou populations but there is little information on its extent or impact (Hayes et al. 2003; Courtois et al. 2007), because reporting is limited. Action 4b of the 2012-2023 Quebéc Caribou Recovery Plan identifies the need to identify harvest management terms and conditions between Aboriginal communities and the provincial government. In the James Bay region of Québec, Rudolph et al. (2012) demonstrated that Aboriginal hunting had a significant negative impact on Boreal Caribou. Schmelzer et al. (2004) reached the same conclusion in Labrador. Hunting of boreal caribou is facilitated by construction of roads and other linear features and by use of off-road vehicles that permit access to previously inaccessible areas.
In conclusion, although the extent of hunting is poorly described in some regions of the BP range across Canada, many observations indicate that it remains an important component of Caribou mortality and hence may represent a threat to some local populations (Callaghan et al. 2011), such as in Alberta (Dzus 2001) and Labrador (Schmelzer et al. 2004).
Low Threats
Fire and Fire Suppression (IUCN 7.1)
Many native elders note that lands are now drier, which has increased the frequency and severity of forest fires, and has reduced the winter range available for Caribou (Northern River Basins Study 1996 quoted in COSEWIC 2012, p. 99).
“Recent changes in climate are significant which include warmer temperatures, increased rain in November, milder winters and increasing summer storms. Boreal woodland caribou food sources are affected by precipitation. During colder times, food becomes less accessible as it is covered by more snow, making it harder for caribou to access.” (SRRB (2010), in COSEWIC (2012, p. 100).
Caribou co-evolved with forest fires, which are a natural component of the boreal forest, but the effect of fires on Caribou range occupancy is complex and subject to conflicting reports in the literature (Schaefer and Pruitt 1991). Forest fires initially diminish the forest habitats of Caribou because they result in loss of mature conifer stands, loss of lichens and other forage plants, and act as barriers to movement (Thomas and Gray 2002; Dalerum et al. 2007; Dzus et al. 2010; Environment Canada 2012). Caribou generally do not return to burned areas for several decades until the forest is old enough to support lichens and other food sources, although they may make limited use of burned areas to feed on new growth (Boreal Caribou ATK Reports 2010-2011; CRA 2010).
Fire can also be beneficial because it helps generate coniferous trees (i.e., Jack Pine, Pinus banksiana) and prevent replacement of lichens by inedible feather mosses (Hypnaceae) in mature or old forest (Schaefer and Pruitt 1991). Fires reduce lichen biomass but may increase the amount of summer green forage (Fisher and Wilkinson 2005). Elders of the Anishinaabe Nation state that Caribou will use habitats affected by forest fires only once the vegetation regrowth begins (Miller 2010). Although fires initially decrease Caribou habitat, balancing the short-term detriments of fire against the long-term benefits is the key to the understanding of the effects of fire on Caribou (Schaefer and Pruitt 1991). Caribou shift their use of habitat from the burned areas to areas that are more suitable (Environment Canada 2011). However, suitable new areas may be limited in areas with fire and increased forest harvest and disturbance. As a result, forest fires can threaten Boreal Caribou recovery, even though they are a natural component of the boreal forest ecosystem (Environment Canada 2012).
The fire cycle follows a gradient from west to east where it is shorter in western Canada than in eastern Canada (Bergeron et al. 2001). In the eastern provinces, the fire rotation is about 111-139 years in western and central Québec’s Black Spruce (Picea mariana) forests on xeric soils, but increases to about 500 years in eastern Québec and southeastern Labrador’s mesic Black Spruce forests (Bergeron et al. 2001; Bergeron and Le Goff 2005). The Boreal Caribou ranges in Saskatchewan are particularly affected by forest fires and the Boreal Shield range fire rotation can be about 70 years. Figure 11 displays the extent of fire within Caribou range in Ontario. At the other extreme in fire rotation, fires have not played a major role in the regional forests around the GP in recent history (1952-1998), the fire cycle being currently estimated at more than 2,700 years (Parisien et al. 2004). In summer 2014, an unprecedented 3 million hectares of Caribou habitat was burned (Carriere pers. comm. 2014).
An increase in fire periodicity above natural intervals could reduce lichen-bearing forests. The regeneration time of lichen after burns will influence the length of time before sites become suitable again; in forests of the NT near the Saskatchewan border, ‘caribou lichen’ (Cladina spp, Cetraria nivalis) biomass stabilized between 40-60 years (Thomas et al. 1995). Predictions for future fire impact vary from a 1.9X increase in area burned in western mixed boreal forest (Krawchuk et al. 2009), to significant increase in fire severity in parts of central and western Ontario (Colombo et al. 1998), a 7X increase in BP range in central Quebec (Le Goff et al. 2009), to a minor impact in eastern boreal forest (Bergeron et al. 2001). Modelling based on a scenario of 3X CO2 increase suggests that, compared to fires between 1959-97, there will be an approximately 50% increase in fire over the BP range by 2100 (Flannigan et al. 2005).
Unknown Threats
Recreational Activities (IUCN 6.1)
Recreational activities, such as snowmobiling, hiking, skiing and cabins can have significant impact on Caribou by displacing them, forcing them to use lower quality habitats, or changing their behaviour (Duchesne et al. 2000; Mahant 2013). Each of these responses can impact body condition, recruitment, individual survival, and vulnerability to predation (Bergerud 1988; Vistnes and Nelleman 2008; Bowman et al. 2010).
Problematic Native Species (IUCN 8.2) – parasites and pathogens
Caribou parasites can influence host population dynamics, and quality and safety of the meat consumed by people (Kutz et al. 2009). Potential changes in the distribution of alternative prey species could have negative consequences for the BP. The presence of abundant Deer is considered one of the factors in Caribou range recession, as a food item for Wolves and vector for disease (Dumont and Crête 1996; Racey and Armstrong 2000; Pitt and Jordan 2004). Meningeal Brainworm (Parelaphostrongylus tenuis), which is non-lethal to White-tailed Deer, can be transmitted via gastropods on vegetation to Caribou and causes death (Anderson and Strelive 1968). P. tenuis exists from Saskatchewan eastward (Wasel et al. 2003). Re-introduction attempts into historical southern range have failed, likely because of the establishment of infected Deer populations (Bergerud and Mercer 1989). In Maine, for example, 32 Caribou (originally from stock from Newfoundland) were released in 1989; of 25 known mortalities, 26% died from Brainworm (McCullough and Connery 1991).
The historical northward expansion of White-tailed Deer into Caribou range was in response to widespread creation of younger forest and associated preferred browse species (Hall 1987). The northern limit of deer range is limited by severe winters (Hall 1987) and it is likely that deer populations will expand northward into Caribou range because of climate change (Thompson et al. 1998; Pickles et al. 2013). White-tailed Deer have expanded into boreal forest due to warmer winters (Côte et al. 2004; Dawe 2011), including a 17.5X increase within BP Caribou range of northeastern Alberta since the 1990s (Latham et al. 2011b). Parelaphostrongylus tenuis presently is absent in deer in Alberta.
Although Moose can be severely affected by the Winter Tick (Dermacenter albipictus), Caribou are also a host of this parasite (Samuel 2004). Kutz et al. (2009) reported that Winter Tick range is expanding into the Canadian North (Girard pers. comm. 2014; Larter and Allaire 2014), possibly due to warmer spring weather (Drew and Samuel 1986). In BC, Culling and Culling (2013) observed an increasing number of Caribou with hair loss and patches of bald skin caused by adult Winter Ticks.
Chronic wasting disease (CWD) may spread to Caribou in western Canada with increasing range overlap with Deer and Elk. This prion disease is highly transmissible and it has been detected in 18 states in USA, and in Saskatchewan and Alberta (Tapscott 2011). A recent experimental study confirmed that reindeer are susceptible to CWD by oral inoculation, implicating the potential for transmission to other Rangifer subspecies (Mitchell et al. 2012).
In 2013, unexpected high levels of Caribou mortality were recorded in northeastern BC, which may have been due to an outbreak of Erysipelothrix rhusiopathiae (Macbeth et al. 2014). This is the first record this pathogenic bacterium in North American Caribou, but it is implicated in mortality events in Musk-ox (Kutz pers. comm.).
Habitat Shifting (IUCN 11.1)
The Dehcho region is warmer and wetter, which results in ice formation over ground lichens preventing successful foraging. Winter thaws create ice, which reduces available Caribou forage and increases the energy used by Caribou to obtain forage (COSEWIC 2012, p. 99). There is growing concern that large areas of boreal peatland are converting from forest to wetlands. Permafrost covers 37% of boreal peatland across BP range, and is melting due to climate warming; 9% loss was recorded in one study (Baltzer et al. 2014) in the last 40 years, with the rate tripling since 2000. Compared to forest, the wetlands may become relatively unused habitat.
Pollution (IUCN 9.2, 9.5)
Aboriginal users of Caribou have raised concerns that pollution and other environmental contaminants are negatively affecting this species (COSEWIC 2012). However, studies that assessed contaminant levels in Caribou tissues have not suggested that these pose a risk to Caribou survival (for example in the Yukon, see Gamberg 2004).
Gaspésie Population
Using metrics from the National Recovery Strategy for the BP (Environment Canada 2011) of a 500-m buffer zone on clear-cuts and roads, and no buffer on fire and windthrow areas, St-Laurent (unpub. data) determined that the disturbance level of the GP area as 75% (Figure 14), well above the 35% threshold determined necessary for sustainability for the BP (Environment Canada 2011). The disturbance was mainly from roads, and forest harvest. The National Recovery Strategy framework likely applies to the GP because the response of the GP to forest change, linear features, and predators is similar to responses recorded by the BP (see Habitat Trends section).
The IUCN Threats calculator for GP indicates an overall threat score of Very High – Very High (Appendix 2). The High threats were Logging and Predation.
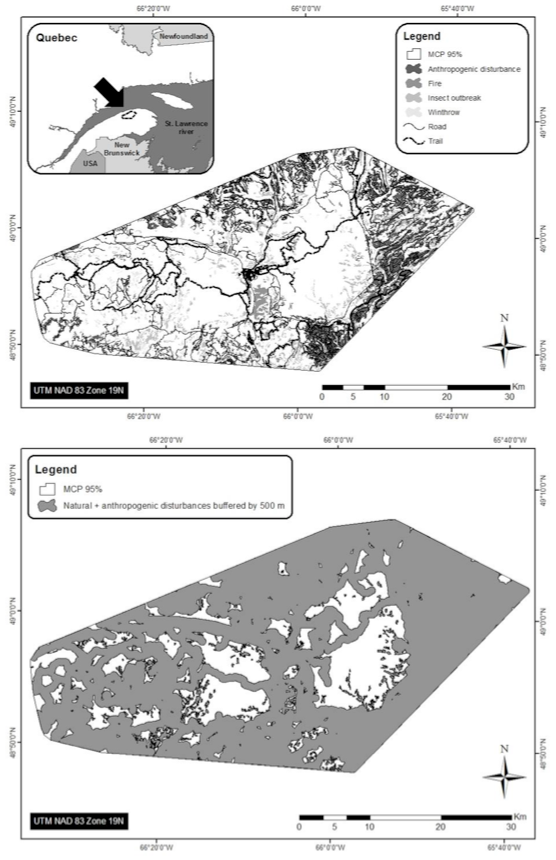
Long description for Figure 14
Two map panels illustrating the extent of anthropogenic and natural disturbance within the range of the Atlantic-Gaspésie population of Caribou. One panel indicates anthropogenic disturbance (including roads and trails) and types of natural disturbance (fire, insect outbreak, and windthrow). The other panel shows extent of disturbance when 500-metre buffer zones are included.
Very High Threats
Renewable Energy (IUCN 3.3)
Wind turbine farms exist within GP range and three more farms are in development, with turbines close enough that most of the intervening forest likely will be removed (Lalonde pers. comm.).
Logging and Wood Harvesting (IUCN 5.3)
Logging within the Park from 30 years ago, and in GP range more recently, has created young forest that is generally poor Caribou habitat. Specialized forestry practices are applied to most of the GP range (see Habitat Protection and Ownership section), with the goal of maintaining lichen availability and connectivity, but also minimizing creation of habitat that could increase predator density. Roads and harvest contribute to a disturbance level of approximately 75% (M.-H. St-Laurent, unpub. data) in the greater GP areas. Future trends in forestry activity are unknown.
Problematic Native Species (IUCN 8.2) - predation
Extinction is predicted in 20 years if calf mortality remains at present rates (Lesmerises 2012; see Fluctuations and Trends section). A study of 25 radio-collared calves in 1989 and 1990 identified Coyote predation as causing 64% of calf deaths, while Black Bears and Golden Eagles caused 27%, and 4%, respectively (Crête and Desrosiers 1995). Low calf survival in the GP is attributed to predation (St-Laurent et al. 2009) and predator removal programs were put in place from 1990 to 1996 (Pilon 1997) and from 2001 to present. Coyotes and Black Bears were culled at varying intensities and locations (Équipe de rétablissement du caribou de la Gaspésie 2011). Predator control temporarily improved calf recruitment in some years but these trends stopped when predator control ceased (Lalonde 2013).
Apparent competition in the GP is likely a contributing factor to Caribou decline via a complex Coyote, Bear, Moose, Deer and Caribou system evolving in disturbed range. Similar to the BP, timber harvesting in the GP range has created favourable Moose habitat, augmented Coyote populations, and created negative consequences for Caribou (Crête and Desrosiers 1995). Coyote were first sighted in Gaspésie in 1973 (Georges 1976) and by the 1990s, they were the main cause of calf mortality (Fournier and Faubert 2001). Hunting may support predator populations because it provides food to Coyotes and Black Bears via injured animals and carcasses dumped at the edge of the Chic-Chocs and Matane wildlife reserves (Boisjoly 2007; Mosnier et al. 2008a; Boisjoly et al. 2010). Forestry-mediated habitat alteration fosters populations of Coyotes and Black Bears around the Gaspésie Park as logging modifies those landscapes once dominated by old forest. Black Bears selected clear-cuts in summer and autumn (Mosnier et al. 2008b). These regenerating stands also support higher Snowshoe Hare densities, which are an important prey species for the Coyote in Gaspésie (St-Laurent et al. 2009). Moose and White-tailed Deer populations have also increased in the Gaspésie region over the last decade (Lamoureux et al. 2007), which in turn contributes to increased predator densities.
High Threats
Tourism and Recreation Areas (IUCN 1.3)
Numerous hunting/fishing/outdoor recreation camps and lodges exist in the area, with several proposals for large resorts within the GP range.
Linear Features (Roads, utility lines; IUCN 4.1, 4.2)
The reason for increased use of alpine habitats over time by the GP is unknown but the avoidance of linear structures (paved and unpaved roads, hiking and skiing trails) caused a functional habitat loss of about 30 to 50% (Gaudry 2013), and may have resulted in the greater use of open habitats where there are fewer linear structures.
The abundance of roads is an important factor in the 75% disturbance level for the GP. Future road building or decommissioning activity is unknown.
Medium Threats
Mining and Quarrying (IUCN 3.2)
Mining is not permitted within the area containing most of the GP. There are numerous claims for mining in areas adjacent to the GP (e.g. Mount Lyall, 10 km south) but new mines are not expected in the near future (Lalonde pers. comm.). Hydroelectric facilities are not expected in the area, and there presently is a moratorium on shale gas hydrofracturing in Quebec. A water pumping project is proposed adjacent to the Park, within the range of GP (Lalonde pers. comm.).
Low Threats
Commercial and Industrial areas (IUCN 1.2)
A large water pumping station is in development adjacent to the largest remaining GP sub-population.
Hunting (IUCN 5.1)
Sport hunting of the GP ceased from 1929 to 1934 but subsequently resumed until ceasing definitively in 1949; since then, sport hunting of Caribou has been banned in GP range (St-Laurent et al. 2009). With the support of local Aboriginal groups, no subsistence hunting occurs in the GP. Some poaching cases were reported and mortality from any sources could be significant with such a small population.
Recreational Activities (IUCN 6.1)
Winter ecotourism can negatively influence Caribou survival and recruitment because Caribou spend less time resting and foraging, and increase vigilant behaviours in the presence of hikers within the GP (Dumont 1993). There are over 9000 hikers on the Mount Jacques-Cartier within the Gaspésie Park and the presence of these hikers caused Caribou to shift locations from alpine tundra areas to subalpine forests where they were more heavily predated on by Coyotes and Black Bears (Dumont 1993). Hiking trails were recognized as inducing functional habitat loss for Caribou in the Gaspésie National Park (Gaudry 2013) because Coyote and Black Bear use hiking trails to access the high plateaus where Caribou are located, and Caribou are avoiding areas surrounding these trails.
Avalanches (IUCN 10.3)
Parts of the GP range experience avalanches, on the scale of every 2 years. There is the potential for a significant proportion of the remaining animals to be killed in one of these stochastic events.
Unknown Threats
Fire and Fire Suppression (IUCN 7.1)
Comments regarding climate change and the effect of an increase in fires are discussed in the Threats section for the BP. The predicted future fire regime for the Gaspésie peninsula is unknown.
Problematic Native Species (IUCN 8.2) – parasites and pathogens
Near the GP range, the prevalence of Winter Ticks on harvested Moose was high (84-96%) in three hunting areas in 2009 and 2010 while it was absent in 2000 (Gingras 2013); Winter Ticks were found on most GP Caribou during the capture sessions in 2013 and 2014 (M.-H. St-Laurent, unpubl. data). In captivity, Winter Tick infections on Caribou can lead to mortality (Welch et al. 1990). Studies are underway to assess Toxoplasma gondii and Neospora caninum (St-Laurent pers. comm.).
Temperature Extremes/Forage Limitation (IUCN 11.3)
Forage limitation may pose a threat to the GP because Gaspésie Park is essentially an island surrounded by abundant summer forage, but low winter forage, and abundant predators. The GP is dependent on the forested mountain slopes for their winter forage, yet these forests may only be able to support a population of 400 Caribou (Ouellet et al. 1996). However, the functional loss of alpine and subalpine habitat associated with the avoidance of linear structure (Gaudry 2013) suggests that this carrying capacity could be overestimated. Forage may thus be sufficient both in quantity and quality, but Caribou may face nutritional restrictions when functional access to forage is limited. Indeed, forage limitation is suspected because pregnancy rates were low in the last two years, especially for young females (M.-H. St-Laurent, unpubl. data). Ice events reduce access to lichen, and are a growing concern because an extensive, extended ice event could impact a larger percentage of the small population (Lalonde pers. com.).
Newfoundland Population
The IUCN Threats calculator for NP indicates an overall threat score of High-Medium, due to cumulative impact of several medium and low threats (Appendix 3). The highest ranked threat was predation (medium threat) and energy production, hunting, logging, recreation, and invasive threats were ranked as low threats. The disturbance threshold used in the National Recovery Strategy framework for assessing sustainability of BP likely applies to the NP at some level but the absence of Wolves, or large fires and intensive forestry over large parts of the range suggests a different threshold of sustainability needs to be determined. In the NP, the human footprint is relatively low and is located mostly in the coastal areas (NL Gov., unpubl. data 2013; Figure 15).
Long description for Figure 15
Map illustrating a human footprint index (range 0 to 10) for the island of Newfoundland. The index shows that human disturbance is relatively low and located mostly in the coastal areas.
Medium Threats
Problematic Native Species (IUCN 8.2) - predation
Black Bear are native to Newfoundland and Coyote are considered naturally invasive because they arrived in 1985 as part of a continental-scale expansion. Predation of calves is an important factor in the population of the NP but the role of predation in the recent decline is less clear (see Fluctuations and Trends section). The potential for Wolves establishing in Newfoundland is unknown; there have been two Wolf records since 2009 (Adams et al. 2012; Pilgrim et al. 2012). If Wolves re-established on Newfoundland, it is likely that predation would become a more significant factor in the NP.
A diversionary food experiment and experimental cull of 40 Coyote over 2 years was conducted in one sub-population, with calf survival rates increasing from approximately 5% before the removal, to 30% afterwards (Lewis et al. 2014).
Low Threats
Energy Production and Mining (IUCN 3)
In the NP, the La Poile sub-population showed a general avoidance of a gold mine across all seasons. The avoidance of the gold mine for the La Poile sub-population was most prominent during the pre-calving season and the number, and group size of Caribou decreased with proximity of the mining operation (Weir et al. 2007). The ZOI on Caribou varied in size among DUs and ranged from about 4 km to 30 km from a mine site (Weir et al. 2007; Boulanger et al. 2012). The ZOI appears to be larger for mines with a large geographic footprint and higher levels of activity, and diminished around smaller mines.
Migration patterns can be influenced by anthropogenic disturbance. For example, the timing of migration for the Buchans Plateau sub-population was disrupted by the construction phase of a hydroelectric development but returned to pre-disturbance timing once the construction phase was finished (Mahoney and Schaefer 2002b).
Linear Features (Roads, utility lines; IUCN 4.1, 4.2)
McCarthy et al. (2011) found that there was a significant negative relationship between recruitment and the amount of disturbance due to recreation, mining, hydroelectric development, roads, and logging on Newfoundland. It is not possible to determine the impact of only linear features because linear features are associated with these developments.
Hunting (IUCN 5.1)
Hunters typically harvest prime-aged individuals (Mahoney and Weir 2009) and the potential for negative impact on population growth is much greater than that of predators, which are more likely to remove old individuals or young of the year (Wright et al. 2006). Hunting of the NP is still permitted in most local populations, with the exception of the Avalon Peninsula, Grey River, Burnin Knee, Burnin Foot, and the Blow Me Down Mountains sub-populations (Newfoundland and Labrador Hunting and Trapping Guide 2012). Sport hunting of the NP is closely regulated and the sub-population harvest rates varied from zero to 17.8%, depending on the Caribou status within the management areas (Weir, J. and Luther, G., NLDEC).
Survey effort was relatively limited in the 1990s and managers did not detect that a population decline was underway. Harvest quotas were not lowered soon enough and hunting was found to substantially contribute to the decline of Caribou in late 1990s (Weir, J. and Luther, G., NLDEC; seeFluctuations and Trends section). Since 2006, the percent of decline caused by hunting has decreased due to stringent hunting quotas, coupled with a harvest management strategy that is intrinsically tied to population demographics (e.g., size, calf recruitment, adult male component and total harvest) (NLDEC, unpubl. data 2013).
Areas such as Newfoundland with extensive data on harvest rates and population estimates demonstrate that harvesting by humans can have a significant impact on Caribou. Both males and females are hunted, but harvest is male-biased and linked to a skewed sex ratio in some Caribou sub-populations, although males have increased in the NP recently (Weir et al. 2014). Annual sales of hunting licences reached an average maximum of about 6,800 licences sold from 1999 to 2003, but have decreased to less than 1000 licences since 2009 because of a lower quota set by the Newfoundland government to stop the decline of Caribou. A decrease in the hunter success rate was observed as the NP declined, with lowest hunter success (60%) observed in 2005. Since 2005, the hunter success rate appears to be increasing, despite the continued decline of the Caribou population.
Logging and Wood Harvesting (IUCN 5.3)
There is evidence of negative impact of forestry on Newfoundland Caribou; fewer females and calves exist near clear-cuts (Chubbs et al. 2007) and female Caribou on Newfoundland avoided recent clear-cuts by an average of 9.2km (Schaefer and Mahoney 2007). However, forest harvesting has slowed greatly in recent years in NP range (DNR 2014a) and many Caribou are not dependent upon mature forests, but instead calve and winter in open areas (see Habitat Requirements section). Large portions of the land are still relatively undisturbed in the northern boreal regions and on the Avalon Peninsula and southern and central Newfoundland.
Recreational Activities (IUCN 6.1)
In the NP, Caribou were displaced 60-237 m by snowmobile activity, with adult-only groups responding sooner, and travelling farther than groups with calves, and movement was generally less during periods of deeper snow (Mahoney et al. 2001). Density of roads and trails is not known but considered to be high in parts of NP range. Access is not limited by roads in open areas, such as the barrens and plateaus.
Forage Limitation (IUCN 7.3)
The amount and quality of food is considered to be a main factor in recent declines (see Fluctuations and Trends section). Forage decreased because of high population density of Caribou in the 1990s. Substantial habitat is available and intact, therefore it is unlikely that conditions of decreased forage will persist indefinitely.
Negligble Threats
Fire and Fire Suppression (IUCN 7.1)
In the NP, fire is a rare event and the average amount of land burned annually from 2011-2013 was 632 ha (range 38-1781 ha), which is 0.006% of the island (DNR 2014b). The dynamics of mature forest in western parts of NP range is driven by insect epizootics; the role of climate change on these dynamics is unknown.
Problematic Native Species (IUCN 8.2) – parasites and pathogens
In Newfoundland, the Brainworm, Elaphostrongylus rangiferi, was responsible for at least two epizootics of a debilitating neurologic disease, which affects Moose as well as Caribou (Ball et al. 2001). E. rangiferi was introduced in Newfoundland with Reindeer brought from Norway in 1908 (Lankester and Fong 1998). Caribou appear to be developing Brainworm immunity in NP and outward signs of the disease are less frequently observed. The Brainworm has been recorded in all major sub-populations but in five years (2007-2012) of carcass inspection, few cases were noted and the threat is considered to be minor.
Introduced Genetic Material (IUCN 8.3)
The introduction of Reindeer from Europe in the early 1900s does not appear to have altered genotype of Newfoundland Caribou. Wilkerson (2010) determined that there has been little to no genetic interchange with Norwegian reindeer and island Caribou.
Number of Locations
Newfoundland population
There are an estimated 14 sub-populations on Newfoundland (see Canadian Range section) and each varies in their exposure to predation, hunting, poaching, food availability and forestry activities. There is no single threat dominating the population. The number of locations would at least be 14.
Gaspésie population
The GP exists as a small metapopulation of three sub-populations within a small and isolated location (see Dispersal and Migration section). There is no single threat that encompasses the three sub-populations. Three locations are recognized because predation and disturbance threats are similar within each sub-population.
Boreal population
The Federal Recovery Strategy (Environment Canada 2012) identified 51 Boreal Caribou ranges from Labrador to the Yukon, and each varies in its exposure to predation, hunting, poaching, food availability and forestry activities. There is no single threat dominating the population. The number of locations would be ‘many’.
Protection, Status and Ranks
Legal Protection and Status
All three populations are found only within Canada. The status of the NP was assessed by COSEWIC as Not at Risk in 1984, 2000, and 2002.
In 1984, the GP was listed as Threatened and uplisted in 2000 to Endangered under the Species at Risk Act (SARA) due to a sharp decline in recruitment. This status was reconfirmed in 2002 (Table 6). The Québec government also prepared a Recovery Plan for the period 2002-2012 (Comité de rétablissement du caribou de la Gaspésie 2002) and this plan was evaluated for the period 1990-2009 in an interim report published in 2011 (Équipe de rétablissement du caribou de la Gaspésie 2011). GP are listed as Threatened in Québec.
The BP was first assessed in 2000 as Threatened, which was reconfirmed in 2002. Each province within BP range lists Boreal Caribou as a special designation conservation status (i.e., vulnerable, threatened, red-listed), except Saskatchewan and the Yukon, where they are listed as Not at Risk (Table 6). The range of BP is very small in the Yukon and much of the range in Saskatchewan is relatively undisturbed. Boreal Caribou are listed as Vulnerable in Québec. In Ontario, Boreal Caribou in central Ontario (‘forest-dwelling woodland caribou’) are listed as Threatened, while Boreal Caribou in northern forests (forest-tundra woodland caribou’) are not facing similar disturbance threats, and are listed as Not at Risk (OMNR 2012).
| Designatable UnitsFootnotebb | Federal status: SARA | Province/Territories | Provincial status | Nature Serve rankFootnotecc |
|---|---|---|---|---|
| Newfoundland | Not at risk (2002) | Newfoundland & Labrador (island only) | Not at risk | S4 |
| Boreal | Threatened (2002) | NT | Threatened (2012) | S3 |
| Boreal | Threatened (2002) | Yukon | Not at risk | S1 |
| Boreal | Threatened (2002) | British Columbia (2010) | Red-listed (Threatened-Endangered) | S2 |
| Boreal | Threatened (2002) | Alberta | Threatened | S2 |
| Boreal | Threatened (2002) | Saskatchewan | Not at risk | SNR |
| Boreal | Threatened (2002) | Manitoba | Endangered (1994) | SNR |
| Boreal | Threatened (2002) | Ontario | Threatened (2007) | S4 |
| Boreal | Threatened (2002) | Québec | Vulnerable (2005) | S2S3 |
| Boreal | Threatened (2002) | Newfoundland & Labrador (Labrador only) | Threatened (2002) | S2S3 |
| Gaspésie | Endangered (2002) | Québec | Threatened (2009) | N1T1Footnotedd |
Non-Legal Status and Ranks
The global status of Rangifer is listed by IUCN (2008) as least concern because of their wide circumpolar distribution and large populations, although population declines have been documented for many sub-populations worldwide (Vors and Boyce 2009). However, the taxonomic classification used in this global status is much coarser than what is used in Canada and includes semi-domestic European Reindeer sub-populations as well. Nature Serve (2013) determined the status for the GP as critically imperilled (N1, T1). Nature Serve (2013) also lists the conservation status of Boreal Caribou for each province/territory (Table 6). Caribou are classified as being globally (G5) and nationally (N5) secure in 2006 and 2012 respectively. Wild Species (2010) ranks the Canadian population of Caribou as well as the populations in the NT, Nunavut, Manitoba, Québec, Labrador, and Newfoundland populations as 4 (secure), the Yukon, British Columbia, Saskatchewan, and Ontario populations as 3 (sensitive), and the Alberta population as 1 (at risk).
Habitat Protection and Ownership
Habitat protection is managed by creation of protected areas, but mainly by establishing policy and best-management practices for mitigating the impact of forestry and other resource development on Caribou. Appendix I of the Federal Recovery Strategy outlines generic mitigation practices (Environment Canada 2012). Courtois et al. (2008) reported that Caribou would remain in a 2,800 km2 area where the forest management plan included the preservation of large forest blocks (35-182 km2) linked with >400 m corridors. It has been noted that the efficacy of these forest range strategies for long-term conservation remain unresolved and may not be apparent for decades because time lags exist between habitat disturbance and extirpation (Vors et al. 2007). Most practices are embedded in forest management plans and Caribou conservation plans developed by jurisdictions.
Newfoundland population
Insular Newfoundland has large amounts of undisturbed landscape, most of which is Crown land. A total of 770,000 hectares (11%) in Newfoundland are located within protected areas, which covers 6.7% of the most intensively used areas (core areas) of Caribou on the island, mainly within Gros Morne National Park (Wells et al. 2011). Most Caribou Management Areas (CMA) have less than 3% of protection, two CMAs have between 3 and 25% and one CMA has more than 25% of its land protected. There are still sufficient large blocks of undisturbed landscapes to maintain large populations of Caribou on the island; however, there is little habitat protection within these areas (Wells et al. 2011).
Caribou are widely distributed island-wide and have seasonal migrations, which make protection of large areas difficult. The Avalon and Bay du Nord Wilderness Reserves were specifically established to be large enough to protect large mobile animals. Nevertheless, these reserves did not insulate the Avalon sub-population from the most recent epizootic of Elaphostrongylus rangiferi in 1996-1998 (Ball et al. 2001). The Middle Ridge sub-population, which overlaps the Bay du Nord Reserve and Middle Ridge Wildlife Reserve, is the most abundant sub-population on the island.
Much of Newfoundland is unlikely to be developed for commercial forestry because some regions (e.g., southcentral) are largely bog/barren lands and are of little commercial value (NLDEC, unpubl. data 2013). Cranberry and peat farming could occur on bog areas. Parks and reserves are a small part of Caribou conservation on the island because few parks exist where Caribou sub-populations are located. Environmental assessment processes and guidelines exist to reduce impacts of forestry and mining on Caribou (NLDEC, unpubl. data 2013).
Gaspésie population
In 1937, the Québec government established the Gaspésie Park to protect the alpine and sub-alpine landscape of the Mount Albert (1157 m) and other Chic-Chocs mountains like Mount Jacques-Cartier (1270 m) in the vicinity. Caribou habitat was legally defined and protected. In 2006, 657 km2 was protected, most of which was located within the Gaspésie Park. In 2011, the protected, legally defined habitat was increased to 1,035 km2, of which 233 km2 is outside the Gaspésie Park (802 km2). The remaining legal habitat is found in the Chic-Chocs and Matane Wildlife Reserves and provides a specific protection status for Caribou habitat, including that used by Caribou outside the park that are subject to anthropogenic disturbances like logging, mining and recreational activities (St-Laurent et al. 2009).
Since 1997, managers have established silvicultural methods that may preserve Caribou habitat quality outside the Gaspésie Park, while also permitting as much land use and forest exploitation as possible. A first special Forest Management Plan (1999-2004) in the GP range aimed to protect summits with tundra habitats, movement corridors and control deciduous regeneration that is beneficial to Moose and also experiment with partial and irregular logging practices (Champagne et al. 1999). The conservation value of some elements of this strategy, like irregular and partial cuts, is not completely known yet (St-Laurent et al. 2009). A second Forest Management Plan to help Caribou range conservation outside the park was published in 2007 and was effective until 2012-2013 (Turcotte et al. 2007). Most of the areas covered by these two plans were within the legal habitat delineation adopted in 2006. The first two plans were made for a relatively small area of 290 km2 used by GP outside the park limits while a third plan recently published will cover an area of 2,857 km2 (Chouinard and Lalonde 2013). The goals of these Forest Management Plans are to: 1) protect tundra summits and their forested high-altitude slopes; 2) maintain arboreal lichen production; 3) limit development of predator habitats; 4) maintain connectivity between Caribou subpopulations; 5) limit disturbance to Caribou; and 6) maintain logging activities for the continuation of forestry. For example, within a 15-km buffer surrounding the park, clear-cutting is not allowed > 700m asl, and < 700 m, partial cutting is used, and fruit-bearing trees and deciduous stems are reduced as a means of decreasing attractiveness of the area to Moose, White-tailed Deer, and Black Bear (QMRN 2013). The impacts of these Plans on Caribou conservation while simultaneously permitting logging activities remain unknown.
Boreal population
The National Recovery Strategy (Environment Canada 2012) was developed over several years and published in 2012. Conservation and recovery plans have been undertaken in the NT (NT Department of Environment and Natural Resources 2010, NT Species at Risk Committee 2012), in British Columbia (BC Ministry of Environment 2011), in Alberta for the period 2004-2014 (Alberta Woodland Caribou Recovery Team 2005), in Manitoba (MBWCMC 2014), in Ontario (Ontario Woodland Caribou Recovery Team 2008, OMNR 2009, 2012), in Québec for the period 2005-2012 (Équipe de rétablissement du caribou forestier du Québec 2008, 2010) and for the period 2013-2023 (Équipe de rétablissement du caribou forestier du Québec 2013), and in Labrador (Schmelzer et al. 2004). Mitigation measures, such as two-pass harvest, road layout to avoid important habitat, and harvesting schedules avoiding critical periods (i.e., calving) are common to forestry plans in areas containing Caribou.
In Labrador, incidental hunting of BP Caribou is minimized in areas that overlap with the hunted but larger George River sub-population (DU4) by delineating ‘extension zones’ wherein hunting is closed unless large numbers of the migratory George River sub-population are in the area (Schmelzer et al. 2004).
The Québec Boreal Caribou 2005 - 2012 Recovery Plan and the more recent 2013-2023 Plan (Équipe de rétablissement du caribou forestier du Québec 2013) are based on a rotation logging method where larger stands (100-250 km2) were protected until they became quality habitat for Caribou, but also contained commercially harvestable volumes (>75 years). This approach assumes that Caribou will move away from recent cutovers in search of quality habitat elsewhere within their range. Another hypothesis is that Caribou can simply move north and further away from disturbances. The forest harvesting framework in the Québec recovery plan does not consider the proximate factor of avoidance of disturbed habitat, namely predation risk. It remains unknown what predation risks will be present in these future coniferous forests and whether they will comprise suitable habitat for Caribou. As well, there is a potential conflict between Caribou and Moose habitat management in the James Bay region of Québec, where a priority for Moose could impact Caribou (Girard pers. comm. 2014).
Ontario’s Boreal Caribou population has been classified as a Threatened species since 2007. Ontario’s Woodland Caribou Conservation Plan serves as the provincial government’s response to a recovery strategy (OMNR 2009, 2012). The plan’s goal is to maintain self-sustaining, genetically connected local populations of Woodland Caribou where they currently exist, improve security and connections among isolated mainland local populations, and facilitate the return of Caribou to strategic areas near their current extent of occurrence. Habitat has been delineated into three categories that reflect seasonal habitat use, function, and risk to disturbance (OMNR 2013a). High Use areas (nursery, winter habitat) typically are used for multiple years, are more sensitive to disturbance, and should receive the highest level of protection. Category 2 areas are Seasonal Range that contain travel corridors, but it is recognized that Caribou effectively space themselves from threats that may change annually, and therefore require areas larger than existing, known annual home ranges (Racey and Arsenault 2007; Avgar et al. 2013). High Use areas are typically nested within these Seasonal Ranges, and are dependent on the refuge function provided at this larger spatial scale (OMNR 2013a). Category 3 areas support Caribou indirectly by maintaining the overall refuge function within the range. Category 3 areas are currently young or disturbed (< 40 years old) but are expected to become used habitat in the future as the forest matures and the areas become connected with seasonal range (OMNR 2013a). Critical habitat for BP is defined in the National Recovery Strategy as the area within the boundary of each range that provides an overall ecological condition that will allow for an ongoing recruitment and retirement cycle of habitat, which maintains a perpetual state of a minimum of 65% of the area as undisturbed habitat; and biophysical attributes required by Boreal Caribou to carry out life processes, such as calving, rutting, and winter areas (see Appendix H in Environment Canada 2012). Decisions on resource development relate to how much disturbance each category could withstand but continue to support Caribou (OMNR 2012, 2013b). One category in the conservation plan is based on the expectation of forest harvested areas becoming future Caribou habitat, which is under debate (see Threats - Loss or Change in Forest section).
In Manitoba and Saskatchewan, numerous actions, similar to other jurisdictions, are in development and await acceptance of the draft provincial recovery strategy (MBWCMC 2014; Saskatchewan Environment 2014). In Alberta, various restrictions have been developed, including that new exploration and construction activities must apply for permits to minimize impact to calving and other special sites (Alberta Government 2014). The Alberta Woodland Caribou Recovery Plan 2004/05 – 2013/14 proposes actions to preserve Boreal Caribou and classified four categories based on risk of extirpation (Alberta Woodland Caribou Recovery Team 2005). For the highest risk category “Immediate Risk of Extirpation”, the Plan proposes a moratorium on further mineral and timber resource allocation be put in place until a land use and range plan is completed, evaluated, and implemented. As of 2013, the plan has not been implemented.
In British Columbia, Boreal Caribou are provincially red-listed (Threatened to Endangered), and are identified as Priority 1 under the BC Conservation Framework. The goals of the BC Implementation Plan for the management of the BP are intended to maximize conservation efforts to benefit Boreal Caribou and support future recovery efforts while providing resource development opportunities (BC Ministry of Environment 2011). Numerous protections have been established in BC, including 13% of the boreal range as Resource Review Areas where a 5-year moratorium exists on new oil and gas leases, 73 sites (Wildlife Habitat Areas, Ungulate Winter Ranges) totalling 977,000 ha where no new roads or harvesting will occur, and > 800,000 ha more where resource activities are altered to mitigate impact to Caribou. An additional 748,000 ha of similar mitigation has been proposed for another part of the range. Additional protected areas comprise 1.4% of the range, and 2% of identified core habitat (BC Ministry of the Environment 2010).
Numerous projects to protect Caribou are underway in various First Nations, and management in most of the remaining BP range involves First Nations. For example, in Ontario, the Lac Seul, Attawapiskat, Cat Lake, Slate Falls, Mishkeegogamang, and Eabametoong First Nations are integrating ATK and scientific data towards identifying critical habitat, stewardship and involvement in land use planning that affects Boreal Caribou (OMNR 2012). In Eeyou Istchee (the southern James Bay region), First Nation communities and government agencies are collaborating on conservation of Caribou, including community-based practices to reduce hunting levels where needed, and to establish protected areas (Saganash 2013; Girard pers. comm. 2014). In Labrador, the Labrador Inuit Association is incorporating components of Inuit ‘Customary Law’ into resource management policy and to revive traditional concepts of stewardship related to Caribou (Schmelzer et al. 2004). The Wek’èezhìi, Sahtu, and Gwich’in Renewable Resource Boards in NT (WRRB 2013; SRRB 2010), and the Cree in southern James Bay region (CRA 2010) are very active in ATK gathering and publications on BP Caribou.
Relatively few protected areas exist in the boreal forest and the majority of these are not large or specific enough to accommodate the space needs of Boreal Caribou. Proposals exist for the creation of large parks or reserves in some provinces or territories with areas >10,000 km2, as Environment Canada (2012) reported that 300 Caribou are needed for a self-sustaining local population, thereby requiring ranges of at least 10,000 to 15,000 km2 of habitat to meet their space requirements. Parks Canada and Newfoundland and Labrador’s Department of Environment and Conservation announced the imminent establishment of a new national park reserve in the Mealy Mountain (Akamiuapishkua) area of Labrador. The park reserve will protect roughly 10,700 km2, which will make it the largest national park in eastern Canada. The provincial government also announced its intent to establish a waterway provincial park to protect the Eagle River, adjacent to the proposed national park reserve. Together these areas would protect 13,668 km2 in Boreal Caribou range, primarily of the Mealy Mountain sub-population.
In Québec, protected areas that facilitate exchanges and connectivity between local sub-populations have been proposed as an essential tool to maintain Caribou (Rudolph et al. 2012). The area traditionally used by the Charlevoix Caribou sub-population covers approximately 5,500 km2 and overlaps the Laurentides Wildlife Reserve and three parks, but it is the most heavily disturbed (80-99%) Caribou range in Québec (Environment Canada 2011; St-Laurent pers. comm. 2014). Extensive forestry activities in the Laurentides Wildlife Reserve and on Crown land may threaten the sub-population’s persistence (St-Laurent and Dussault 2012). The process of creating the biodiversity reserve project of Albanel-Témiscamie-Otish is ongoing in Québec (11,871 km2) (Équipe de rétablissement du caribou forestier du Québec 2010), but a small proportion will offer protection to Caribou (Girard pers. comm. 2014). A future park (3200 km2) in Assinica region would offer protection to portions of Caribou range in the area (Girard pers. comm. 2014).
In Ontario and Manitoba, several protected areas are found within the continuous BP range, and a World Heritage site has been proposed (Brannen pers. comm. 2014). However, even the largest of these parks, such as Wabakimi (8,920 km2), Atikaki (3,980 km2), and Woodland Caribou Provincial Parks (4,500 km2) may not be large enough to effectively conserve more than a few sub-populations (MBWCMC 2014). Elsewhere, smaller protected areas will be impacted by habitat change associated with the northward advancing front of forest harvest (Vors et al. 2007).
The creation of five large national wildlife areas in the NT is currently being studied by the Canadian Wildlife Service (Bigelow pers. comm.). The Dehcho First Nations interim land withdrawal agreement and the Wood Buffalo National Park would protect 32,633 km2, the largest in BP range in Canada. Another protected area project will have a total contiguous area of 14,688 km2. It should be noted, however, that these proposed protection areas may have some levels of anthropogenic and natural (fire) disturbance already present.
Acknowledgements and Authorities Contacted
The authors thank Environment Canada for funding the preparation of this status report. Meghan Andersen contributed to the writing of the Apparent Competition section and Dr. Mariana Trindade reviewed a preliminary version of the report. Members of the COSEWIC Terrestrial Mammals and ATK Subcommittees, and numerous reviewers from provinces and territories provided valuable comments, in particular Martin-Hugues St-Laurent, Justina Ray, and Shelley Pardy Moores. The authors would like to thank the following people who were contacted and provided information and comments.
List of authorities contacted
| Name | Affiliations |
|---|---|
| Maria Alt | Manitoba Dept. of Conservation and Water Stewardship, Winnipeg, MB |
| Ted Armstrong | Species at Risk biologist (past), Government of Ontario, Ontario |
| Al Arsenault | Wildlife biologist (past), Government of Saskatchewan |
| Walter Bezha | Sahtu Renewable Resources Board, NT |
| John Blake | Dept. of Environment & Conservation, Wildlife Division, Corner Brook, NL |
| Ruben Boles | Canadian Wildlife Service, Environment Canada, Gatineau, QC |
| Tracz Boyan | Wek’èezhìi Renewable Resources Board, Yellowknife, NT |
| Dennis Brannen | Manitoba Dept. of Conservation and Water Stewardship, Winnipeg, MB |
| Vivian R. Brownell | Ontario Ministry of Natural Resources, Peterborough, ON |
| Kristen Callaghan | Gwich’in Renewable Resources Board, Inuvik, NT |
| Suzanne Carrière | Dept. of Environment & Natural Resources, Yellowknife, NT |
| Karin Clark | Wek’èezhìi Renewable Resources Board, Yellowknife, NT |
| Gordon Court | Alberta Dept. of Sustainable Resource Dev., Edmonton, AB |
| Vince Crichton | Wildlife Biologist (retired), Manitoba Dept. of Conservation, MB |
| Christine Doucet | Dept. of Environment & Conservation, Wildlife Division, Corner Brook, NL |
| Christian Dussault | Ministère des Forêts, de la Faune et des Parcs, Québec, QC |
| Darren Elder | Ontario Ministry of Natural Resources Thunder Bay ON |
| Jonathan Feldgajer | NF Regional Advisor to the Canadian Boreal Initiative |
| David Fifield | Dept. of Environment & Conservation, SDSS, St. John’s, NL |
| David Fraser | British Columbia Ministry of Environment, Victoria, BC |
| Rob Gau | Manager, Biodiversity Conservation, Yellowknife, NT |
| Isabelle Gauthier | Ministère des Forêts, de la Faune et des Parcs, Québec, QC |
| Caroline Girard | Hunting, Fishing and Trapping Coordinating Ctte., Montréal, QC |
| Nicole Gougeon | Hunting, Fishing and Trapping Coordinating Ctte., Montréal, QC |
| Emily Herdman | Dept. of Environment and Conservation, Wildlife Division, Corner Brook, NL |
| Dave Hervieux | Alberta Dept. of Sustainable Resource Dev., Edmonton, AB |
| Susan Kutz | University of Calgary, Calgary, AB |
| Gerry Kuzyk | BC Ministry of Forests, Lands and Natural Resource Operations, BC |
| Mélinda Lalonde | Ministère des Forêts, de la Faune et des Parcs, Québec, QC |
| Nic Larter | Dept. of Environment and Natural Resources, Fort Simpson, NT |
| Keith Lewis | Dept. of Environment & Conservation, SDSS, St. John’s, NL |
| Glenn Luther | Dept. of Environment & Conservation, SDSS, St. John’s, NL |
| Shane Mahoney | Dept. of Environment & Conservation, SDSS, St. John’s, NL |
| Julien Mainguy | Ministère des Forêts, de la Faune et des Parcs, Québec, QC |
| Micheline Manseau | Park Canada and Manitoba University, Winnipeg, MB |
| Katherine Mehl | Dept. of Environment & Conservation, Wildlife Division, Corner Brook, NL |
| Natalka Melnycky | Gwich’in Renewable Resources Board, Yellowknife, NT |
| Patrick Nantel | Parks Canada 25 Eddy St., Gatineau, QC |
| Dean Nernberg | National Defence, Ottawa, ON |
| Shelley Pardy Moores | Dept. of Environment & Conservation, Wildlife Division, Corner Brook, NL |
| Chris Pasztor | BC Ministry of the Environment, BC |
| Claudel Pelletier | Ministère des Forêts, de la Faune et des Parcs, Québec, QC |
| John Pisapio | Dept. of Environment & Conservation, Wildlife Division, Corner Brook, NL |
| Gigi Pittoello | Saskatchewan Ministry of Environment, Regina, SK |
| Paul Saunders | Dept. of Environment & Conservation, Wildlife Division, Corner Brook, NL |
| Isabelle Schmelzer | Dept. of Environment & Conservation, Wildlife Division, Corner Brook, NL |
| Deborah Simmons | Executive Director, Sahtu Renewable Resources Board |
| Jody Snortland | Wek’eezhii Renewable Resources Board, Yellowknife, NT |
| Colleen Soulliere | Dept. of Environment & Conservation, SDSS, St. John’s, NL |
| Susan Squires | Dept. of Env. & Conservation, Park & Natural areas Div., Corner Brook, NL |
| Martin-H. St-Laurent | Université du Québec à Rimouski, Rimouski, QC |
| Amy Thompson | Gwich’in Renewable Resources Board, Yellowknife, NT |
| Vicki Trim | Manitoba Dept. of Conservation and Water Stewardship, Winnipeg, MB |
| Tim Trottier | Saskatchewan Ministry of Environment, Regina, SK |
| Gord Vaadeland | Canadian Parks and Wilderness Society. PAWS, Saskatoon, SK |
| Denis Vandal | Ministère des Ressources naturelles, QC |
| Stephen Virc | Canadian Wildlife Service, Environment Canada, Ottawa, ON |
| Bill Watkins | Manitoba Dept. of Conservation and Water Stewardship, Winnipeg, MB |
Information Sources
Abraham, K.F., (and 6 co-authors). 2012. Recent changes in summer distribution and numbers of migratory caribou on the southern Hudson Bay coast. Rangifer 32: 269-276.
Adams, J., M. Mumma, and L. Waits. 2012. Species identification of unknown canid sample collected in Newfoundland. Laboratory for Ecological, Evolutionary and Conservation Genetics, University of Idaho.
Alberta Caribou Committee. http://www.albertacariboucommittee.ca/ [Accessed July 2014].
Alberta Government 2014. Caribou protection plan guidelines. http://esrd.alberta.ca/fish-wildlife/wildlife-management/caribou-management/caribou-protection-plans/documents/CaribouProtectionPlan-CaribouCalvingInfo-2012.pdf [Accessed August 2014].
Alberta Woodland Caribou Recovery Team. 2005. Alberta woodland caribou recovery plan 2004/05-2013/14. Alberta Sustainable Resource Development, Fish and Wildlife Division, Alberta Species at Risk Recovery Plan No. 4. Edmonton, AB. 48 pp.
Albon, S.D., (and 5 co-authors). 2002. The role of parasites in the dynamics of a reindeer population. Proceedings of the Royal Society B-Biological Sciences 269: 1625-1632.
Alpac. 2014. Forest management agreement area. http://alpac.ca/forest-sustainability/forest-management-agreement-area/. [Accessed August 2014].
Anderson, M. 2012. Wolf responses to spatial variation in moose density in northern Ontario. M.Sc. Thesis, University of Guelph, Guelph, Ontario.
Anderson, R., and U. Strelive. 1968. The experimental transmission of Pneumostgrongylus tenuis to caribou (Rangifer tarandus terranovae). Canadian Journal of Zoology 46:503-510.
Arlt, M., and M. Manseau. 2011. Changes in caribou distribution and landcover in and around Prince Albert National Park: Different management strategies and different landscapes. Rangifer Special Issue 19: 17-31.
Arsenault, A., and M. Manseau. 2011. Land management strategies for the recovery of boreal woodland caribou in central Saskatchewan. Rangifer Special Issue 19: 33-48.
Arseneau, M.-J., L. Sirois and J.-P. Ouellet. 1997. Effects of altitude and tree height on the distribution and biomass of fruticose arboreal lichens in an old growth balsam fir forest. Écoscience 4: 206-213.
Avgar, T., A. Mosser, G.S Brown, and J.M. Fryxell. 2013. Environmental and individual drivers of animal movement patterns across a wide geographical gradient. Journal of Animal Ecology 82: 96-106.
Badiou, P., (and 22 co-authors). 2011. Keeping woodland caribou in the boreal forest: Big challenge, immense opportunity. International Boreal Conservation Science Panel. 12 pp.
Ball, M.C., L. Finnegan, M. Manseau, and P. Wilson. 2010. Integrating multiple analytical approaches to spatially delineate and characterize genetic population structure: an application to boreal caribou (Rangifer tarandus caribou) in central Canada. Conservation Genetics 11: 2131-2143.
Ball, M.C., M.W. Lankester, and S.P. Mahoney 2001. Factors affecting the distribution and transmission of Elaphostrongylus rangiferi (Protostrongylidae) in caribou (Rangifer tarandus caribou) of Newfoundland, Canada. Canadian Journal of Zoology 79: 1265-1277.
Ball, M.C., (and 7 co-authors). 2007. Characterization of target nuclear DNA from faeces reduces technical issues associated with the assumptions of low-quality and quantity templates. Conservation Genetics 8: 577-586.
Baltzer, (and 4 co-authors). 2014. Forests on thawing permafrost; fragmentation, edge effects and net forest loss. Global Change Biology 20:824-834.
Banfield, A.W.F. 1961. A revision of the reindeer and caribou genus Rangifer. Ottawa, Canada, Dept. of Northern Affairs and National Resources.
Banfield, A.W.F. 1974. The Mammals of Canada, Published for the National Museum of Natural Sciences. University of Toronto Press.
Barboza, P.S., and K.L. Parker. 2008. Allocating protein to reproduction in arctic reindeer and caribou. Physiological and Biochemical Zoology 81: 835-855.
Barboza, P.S., D.W. Hartbauer, W.E. Hauer, and J.E. Blakes. 2004. Polygynous mating impairs body condition and homeostasis in male reindeer (Rangifer tarandus tarandus). Journal of Comparative Physiology B 174: 309-317.
Barrette, C., and D. Vandal. 1986. Social rank, dominance, antler size, and access to food in snow-bound wild woodland caribou. Behaviour 97: 118-146.
Bastille-Rousseau, G., (and 7 co-authors). 2012. Sélection d’habitat du caribou forestier en forêt boréale québécoise. Ministère du Développement durable, de l’Environnement, de la Faune et des Parcs, Québec. 66 p.
Bastille-Rousseau, G., D. Fortin, C. Dussault, R. Courtois, and J.-P. Ouellet. 2011. Foraging strategies by omnivores: are black bears actively searching for ungulate neonates or are they simply opportunistic predators? Ecography 34: 588-596.
Bastille-Rousseau, G., J. Schaefer, S. Mahoney, and D. Murray. 2013. Population decline in semi-migratory caribou (Rangifer tarandus): intrinsic or extrinsic drivers? Canadian Journal of Zoology 91:820-828.
Beauchesne, D., J.A.G. Jaeger, and M.-H. St-Laurent. 2013. Disentangling woodland caribou movements in response to clearcuts and roads across temporal scales. PLoS ONE 8: e77514.
Beauchesne, D., J.A.G., Jaeger & M.-H. St-Laurent 2014. Thresholds in the capacity of boreal caribou to cope with cumulative disturbances: Evidence from space use patterns. Biological Conservation 172: 190-199
Bergeron, Y., S. Gauthier, V. Kafka, P. Lefort, and D. Lesieur. 2001. Natural fire frequency for the eastern Canadian boreal forest: consequences for sustainable forestry. Canadian Journal of Forest Research 31: 384–391
Bergeron, Y., and E. Le Goff. 2005. Doit-on remettre en question notre façon d’aménager la forêt boréale canadienne? VertigO – La revue en sciences de l’environnement 6: 1-7.
Bergerud, A.T. 1971. The population dynamics of Newfoundland caribou. Wildlife Monographs (25): 3-55.
Bergerud, A.T. 1973. Movement and rutting behavior of caribou (Rangifer tarandus) at Mount Albert, Québec. Canadian Field-Naturalist 87: 357-369.
Bergerud, A.T. 1974. Decline of caribou in North America following settlement. Journal of Wildlife Management 38: 757-770.
Bergerud, A.T. 1975. The reproductive season of Newfoundland caribou. Canadian Journal of Zoology 53: 1213-1221.
Bergerud, A.T. 1980. A review of the population dynamics of caribou and wild reindeer in North America. Proc. 2nd Int. Reindeer/Caribou Symposium, Roros, Norway. pp 556-581.
Bergerud, A.T. 1985. Antipredator strategies of caribou: dispersion along shorelines. Canadian Journal of Zoology 63: 1324-1329.
Bergerud, A.T. 1988. Caribou, wolves and man. Trends in Ecology and Evolution 3: 68-72.
Bergerud, A.T. 2000. Caribou. pp. 658-693 in Ecology and Management of Large Mammals in North America. S. Demarais and P. Krausman (eds.). Prentice Hall, NJ. 778 p.
Bergerud, A.T., and J.P. Elliott. 1986. Dynamics of caribou and wolves in northern British Columbia. Canadian Journal of Zoology 64: 1515-1519.
Bergerud, A.T., and J.P. Elliott. 1998. Wolf predation in a multiple-ungulate system in northern British Columbia. Canadian Journal of Zoology 76: 1551-1569.
Bergerud, A.T., and W.E. Mercer. 1989. Caribou introductions in eastern North America. Wildlife Society Bulletin 17: 111-120.
Bergerud, A.T., S.N. Luttich, and L. Camps. 2008. The return of caribou to Ungava. Montréal, McGill Queen University Press.
Bergerud, A.T., W.J. Dalton, H. Butler, L. Camps, and R. Ferguson. 2007. Woodland caribou persistence and extirpation in relic populations on Lake Superior. Rangifer Special Issue No. 17: 57-78.
Bigelow, D. Species at Risk Biologist, Canadian Wildlife Service, Yellowknife, NT. Email correspondence with S. Couturier, December 2013.
Boertje, R.D., P. Valkenburg, and M.E. McNay. 1996. Increases in moose, caribou, and wolves following wolf control in Alaska. Journal of Wildlife Management 60: 474-489.
Boileau, F. 1996. Rapport sur la situation du caribou (Rangifer tarandus caribou) du Parc de conservation de la Gaspésie. Min. Environnement et Faune, Québec, QC. 49 p.
Boisjoly, D. 2007. Sélection de l’habitat par le coyote, Canis latrans, dans le contexte de la conservation du caribou de la Gaspésie. M.Sc., Université du Québec à Rimouski.
Boisjoly, D., J.-P. Ouellet, and R. Courtois. 2010. Coyote habitat selection and management implications for the Gaspésie caribou. Journal of Wildlife Management 74: 3-11.
BorealCanada 2008. Mining exploration conflicts in Canada’s boreal forest. http://www.borealcanada.ca/documents/MiningExplorationConflicts-Report-May2008.pdf [Accessed July 2014].
Boreal Caribou ATK Reports 2010-2011. Boreal Caribou Aboriginal Traditional Knowledge (ATK) Reports. 2010-2011. Compiled June 2011. Ottawa: Environment Canada.
Boucher, Y., D. Arsenault, L. Sirois, and L. Blais. 2009. Logging pattern and landscape changes over the last century at the boreal and deciduous forest transition in Eastern Canada. Landscape Ecology 24: 171-184.
Boulanger, J., K.G. Poole, A. Gunn, and J. Wierzchowski. 2012. Estimating the zone of influence of industrial developments on wildlife: a migratory caribou Rangifer tarandus groenlandicus and diamond mine case study. Wildlife Biology 18: 164-179.
Bowman, J., J.C. Ray, A.J. Magoun, D.S. Johnson, and F.N. Dawson. 2010. Roads, logging, and the large-mammal community of eastern Canadian boreal forest. Canadian Journal of Zoology 88: 454-467.
Boyan, Tracz. 2014. Wek’èezhìi Renewable Resources Board, Yellowknife, NT. Email correspondence with G. Forbes, October 2014.
Bradshaw, C.J.A., S. Boutin, and D.M. Hébert. 1998. Energetic implications of disturbance caused by petroleum exploration to woodland caribou. Canadian Journal of Zoology 76: 1319-1324.
Brannen, Dennis. 2014. Manitoba Dept. Conservation and Water Stewardship. Email correspondence with G. Forbes, November 2014.
Briand, Y., J.-P. Ouellet, C. Dussault, and M.-H. St-Laurent. 2009. Fine-scale habitat selection by female forest-dwelling caribou in managed boreal forest: Empirical evidence of a seasonal shift between foraging opportunities and antipredator strategies. Écoscience 16: 330-340.
British Columbia Ministry of Environment. 2010. Science update for the boreal caribou (Rangifer tarandus caribou pop. 14) in British Columbia. Victoria, BC, Ministry of Environment, Victoria, BC. 54 pp.
British Columbia Ministry of Environment. 2011. Implementation plan for the ongoing management of boreal caribou (Rangifer tarandus caribou pop. 14) in British Columbia. Victoria, BC, Ministry of Environment, Victoria, BC. 17 p.
Brodeur, V., J.-P. Ouellet, R. Courtois, and D. Fortin. 2008. Habitat selection by black bears in an intensively logged boreal forest. Canadian Journal of Zoology 86: 1307-1316.
Brown, G.S., F.F. Mallory, and W.J. Rettie. 2003. Range size and seasonal movement for female woodland caribou in the boreal forest of northeastern Ontario. Rangifer Special Issue No. 14: 227-233.
Brown, G.S., W.J. Rettie, R.J. Brooks, and F.F. Mallory. 2007. Predicting the impacts of forest management on woodland caribou habitat suitability in black spruce boreal forest. Forest Ecology and Management 245: 137-147.
Burton, P.J., (and 5 co-authors). 2003. The current state of boreal forestry and the drive for change. In Towards Sustainable Management of the Boreal Forest. P.J. Burton, C. Messier, D.W. Smith, and W.L. Adamowicz, eds., National Research Press: 1-40. Ottawa, ON.
Callaghan, C., S. Virc, and J. Duffe. 2011. Woodland caribou, boreal population, trends in Canada. Canadian Biodiversity: Ecosystem Status and Trends 2010. Ottawa, ON. Technical Thematic Report No. 11: 36 p.
Cameron, R.D., W.T. Smith, R.G. White, and B. Griffith. 2005. Central Arctic caribou and petroleum development: distributional, nutritional, and reproductive implications. Arctic 58: 1-9.
Carlson, M., and C. Chetkiewicz. 2013. A fork in the road: future development in Ontario’s far north. Wildlife Conservation Society and Canadian Boreal Forest Initiative. Ottawa, ON. 72 p.
Carr, N.L., A.R. Rodgers, and S.C. Walshe. 2007. Caribou nursery site habitat characteristics in two northern Ontario parks. Rangifer 27(4):167-179.
Carr, N.L., A.R. Rodgers, S.R. Kingston, and D.J. Lowman. 2011. Use of island and mainland shorelines by woodland caribou during the nursery period in two northern Ontario parks. Rangifer 19: 49-61.
Carrière, Suzanne. 2014. Dept. of Environment & Natural Resources, Yellowknife, NT Email correspondence with G. Forbes, October 2014.
Champagne, S., (and 5 co-authors). 1999. Plan d’aménagement de l’aire du caribou de la Gaspésie. Ministère des Ressources naturelles et Société de la faune et des parcs du Québec, 34 p.
Chouinard, D., and M. Lalonde. 2013. Plan d’aménagement forestier de l’aire de fréquentation du caribou de la Gaspésie (3e édition) 2013-2018. Ministère des Ressources naturelles, Caplan, QC. 29 p.
Chubbs, T.E., L.B. Keith, S.P. Mahoney, and M.J. McGrath. 1993. Responses of woodland caribou (Rangifer tarandus caribou) to clear-cutting in east-central Newfoundland. Canadian Journal of Zoology 71: 487-493.
Colombo, S., (and 9 co-authors). 1998. The impacts of climate change in Ontario forests. Ontario Ministry Natural Resources Forest Research Information Report No. 143. 50 p.
Comité de rétablissement du caribou de la Gaspésie. 2002. Plan de rétablissement du caribou de la Gaspésie (2002-2012) (Rangifer tarandus caribou). Société de la faune et des parcs du Québec, Québec. 47 p.
COSEWIC. 2011. Designatable units for caribou (Rangifer tarandus) in Canada. Ottawa, Committee on the status of Endangered Wildlife in Canada (COSEWIC). 88 p.
COSEWIC. 2002. COSEWIC assessment and update status report on the woodland caribou Rangifer tarandus caribou in Canada. Committee on the Status of Endangered Wildlife in Canada. Ottawa. 98 pp.
COSEWIC 2012. Aboriginal Traditional Knowledge assessment report on caribou Rangifer tarandus in Canada ATK Subcommittee of COSEWIC. Environment Canada, Ottawa.
Côte, S., (and 4 co-authors). 2004. Ecological impacts of deer overabundance. Annual Review Ecology and Evolutionary Systematics 35:113-147.
Courbin, N., D. Fortin, C. Dussault, and R. Courtois. 2009. Habitat management for woodland caribou: the protection of forest blocks influences wolf-caribou interactions, Landscape Ecology 24: 1375-1388.
Courtois, R., (and 5 co-authors). 2003a. Historical changes and current distribution of Caribou (Rangifer tarandus), in Quebec. Canadian Field-Naturalist 117:399-414.
Courtois, R., A. Gingras, C. Dussault, L. Breton, and J.-P. Ouellet. 2003b. An aerial survey technique for the forest-dwelling ecotype of woodland caribou, Rangifer tarandus caribou. The Canadian Field-Naturalist 117: 546-554.
Courtois, R., A. Gingras, D. Fortin, A. Sebbane, B. Rochette, and L. Breton. 2008. Demographic and behavioural response of woodland caribou to forest harvesting. Canadian Journal of Forest Research 38: 2837–2849.
Courtois, R., J.-P. Ouellet, L. Breton, A. Gingras, and C. Dussault. 2002. Effet de la fragmentation du milieu sur l’utilisation de l’espace et la dynamique de population chez le caribou forestier. Québec, QC, Société de la faune et des parcs du Québec: 44 p.
Courtois, R., J.-P. Ouellet, L. Breton, A. Gingras, and C. Dussault. 2007. Effects of forest disturbance on density, space use and mortality of woodland caribou. Écoscience 14: 491–498.
Couturier, S. 2007. Génétique et condition physique des trois écotypes de caribou du Québec-Labrador. Ph.D., Université Laval, Québec, QC.
Couturier, S., J. Huot, S.D. Côté, Q. van Ginhoven, R. Otto, and D. Jean. 2009. Populations, metapopulations, ecotypes and subspecies of caribou in Québec-Labrador: An exploratory discussion. Proceedings from Caribou Genetics and Relationships Workshop, University of Alberta, Edmonton, March 8-9, 2003, Dept. of Natural Resources and Environment, Gov. of the NT.
Couturier, S., R.D. Otto, S.D. Côté, G. Luther, and S.P. Mahoney. 2010. Body size variations in caribou ecotypes and relationships with demography. Journal of Wildlife Management 74: 395-404.
Cree Regional Authority. 2010. Woodland Caribou (Boreal Population). A Portrait of Cree Knowledge in Eeyou Istchee. From Communities to Science. Cree Regional Authority, Montreal.
Crête, M., S. Couturier, B.J. Hearn, and T.E. Chubbs. 1996. Relative contribution of decreased productivity and survival to recent changes in the demographic trend of the Rivière George sub-population. Rangifer Special Issue No. 9: 27-36.
Crête, M., and A. Desrosiers. 1995. Range expansion of coyotes (Canis latrans), threatens a remnant sub-population of caribou (Rangifer tarandus), in southeastern Québec. Canadian Field-Naturalist 109: 227-235.
Cringan, A.T. 1956. Some aspects of the biology of caribou and a study of the woodland caribou range of the Slate Islands, Lake Superior, Ontario. M.A. (Zoology), Univ. of Toronto.
Cronin, M., M. MacNeil, and J. C. Patton. 2005. Variation in mitochondrial DNA and microsatellite DNA in caribou (Rangifer tarandus) in North America. Journal of Mammalogy 86:495-505.
Culling, D., and B. Culling. 2013. BC Boreal caribou implementation plan: 2012-13 collar deployment and late winter recruitment survey. Diversified Environmental Services, Fort St. John, BC. 29 p + appendices.
Cumming, H., D. Beange, and G. Lavoie. 1996. Habitat partitioning between woodland caribou and moose in Ontario: the potential role of shared predation risk. Rangifer 9: 81-94.
Cumming, H. G., and B.T. Hyer. 1998. Experimental log hauling through a traditional caribou wintering area. Rangifer Special Issue No. 10: 241-258.
Dalerum, F., S. Boutin, and J.S. Dunford. 2007. Wildfire effects on home range size and fidelity of boreal caribou in Alberta, Canada. Canadian Journal of Zoology 85: 26-32.
DeMars, C., C. Thiessen, and S. Boutin. 2011. Assessing Spatial Factors Affecting Predation Risk to Boreal Caribou Calves: Implications for Management. University of Alberta and BC Ministry of Natural Resource Operations 1-35.
Dawe, K. L. 2011. Factors driving range expansion of white-tailed deer, Odocoileus virginianus, in the boreal forest of northern Alberta, Canada. Doctoral thesis, University of Alberta, Edmonton, AB, Canada.
Decesare, N., W. Peters, C. Semeniuk, M. Musiani, and M. Hebblewhite. 2012. Scaling up the role of predation in caribou declines in west-central Alberta (Redwillow, Narraway, Redrock Prairie Creek, A la Pêche and Little Smoky ranges). 2012 AUPRF project status update. Interim, internal report to the Petroleum Technology Alliance of Canada (PTAC).
Department of Natural Resources 2014a. Forestry statistics. Government of Newfoundland and Labrador. http://www.nr.gov.nl.ca/nr/forestry/statistics/commercial_production.html. [Accessed August 2014].
Department of Natural Resources 2014b. Forest fire statistics. Government of Newfoundland and Labrador. http://www.nr.gov.nl.ca/nr/forestry/fires/fire_stats.html#Fire%20Reports. [Accessed August 2014].
De Vos, A., and R.L. Peterson. 1951. A review of the status of woodland caribou (Rangifer tarandus) in Ontario. Journal of Mammalogy 32: 329-337.
Drew, M.L., and W.M. Samuel. 1986. Reproduction of the winter tick, Dermacentor albipictus, under field conditions in Alberta, Canada. Canadian Journal of Zoology 64: 714-721.
Duchesne, M., S.D. Côté, and C. Barrette. 2000. Responses of woodland caribou to winter ecotourism in the Charlevoix Biosphere Reserve, Canada. Biological Conservation 96: 311-317.
Ducrocq, J., (and 12 co-authors). 2012. Comparison of gross visual and microscopic assessment of four anatomic sites to monitor Besnoitia tarandi in barren-ground caribou (Rangifer tarandus). Journal of Wildlife Diseases 48: 732-738.
Ducrocq, J., (and 7 co-authors). 2013. Variables associated with Besnoitia tarandi prevalence and cyst density in barren-ground caribou (Rangifer tarandus) populations. Journal of Wildlife Diseases 49: 29-38.
Dumont, A. 1993. Impact des randonneurs sur les caribous Rangifer tarandus caribou du parc de la Gaspésie. M.Sc., Université Laval. Québec, QC.
Dumont, A., and M. Crête. 1996. The meningeal worm, Parelaphostrongylus tenuis, a marginal limiting factor for moose, Alces alces. Canadian Field-Naturalist 110: 413-418.
Dussault, C., V. Pinard, J.-P. Ouellet, R. Courtois, and D. Fortin. 2012. Avoidance of roads and selection for recent cutovers by threatened caribou: fitness-rewarding or maladaptive behaviour? Proceedings of the Royal Society B 279: 4481-4488.
Dyer, S.J., J.P. O’Neill, S.M. Wasel, and S. Boutin. 2001. Avoidance of industrial development by woodland caribou. Journal of Wildlife Management 65: 531-542.
Dyer, S. J., J.P. O’Neill, S.M. Wasel, and S. Boutin. 2002. Quantifying barrier effects of roads and seismic lines on movements of female woodland caribou in northeastern Alberta. Canadian Journal of Zoology 80: 839-845.
Dyke, C. and M. Manseau. 2011. Characterization of woodland caribou (Rangifer tarandus caribou) calving habitat in the boreal plains and boreal shield ecozones of Manitoba and Saskatchewan. Rangifer 31(2):151.
Dzus, E. 2001. Status of the woodland caribou (Rangifer tarandus caribou) in Alberta. Edmonton, AB, Alberta Environment, Fisheries and Wildlife Management Division, and Alberta Conservation Association. Wildlife Status Report No. 30. 47 p.
Dzus, E., J. Ray, I. Thompson, and C. Wedeles. 2010. Caribou and the National Boreal Standard: Report of the FSC Canada Science Panel. https://ca.fsc.org/preview.caribou-and-the-national-boreal-standard-report-of-the-fsc-canada-science-panel.a-498.pdf. 73 pp.
Elder, Darren. Species at Risk Branch, Ontario Ministry of Natural Resources. Email correspondence with G. Forbes, June 2014.
Elkie, P., (and 8 co-authors). 2009. Science and Information in support of the Forest Management Guide for Landscapes: Package Caribou Habitat Definitions, Habitat Tracts, Habitat Models and Simulation Results. Ontario Ministry of Natural Resources. Forest Policy Section.
Environment Canada. 2008. Scientific review for the identification of critical habitat for woodland caribou (Rangifer tarandus caribou), Boreal population, in Canada. E. Canada. Ottawa: 72 pp. plus 180 pp. Appendices.
Environment Canada. 2011. Scientific assessment to inform the identification of critical habitat for woodland caribou (Rangifer tarandus caribou), Boreal population, in Canada – 2011 Update. Environment Canada, Ottawa, 104 p + appendices.
Environment Canada. 2012. Recovery strategy for the Woodland Caribou (Rangifer tarandus caribou), Boreal population in Canada. Species at Risk Act Recovery Strategy Series. Ottawa, ON, Environment Canada, 138 p.
Équipe de rétablissement du caribou de la Gaspésie. 2011. Bilan du rétablissement du caribou de la Gaspésie (Rangifer tarandus caribou) pour la période 1990-2009, Ministère des Ressources naturelles et de la Faune du Québec, Québec. 25 p.
Équipe de rétablissement du caribou forestier du Québec. 2008. Plan de rétablissement du caribou forestier (Rangifer tarandus) au Québec -- 2005-2012. Québec, QC, Ministère de Ressources naturelles et de la Faune, 77 p.
Équipe de rétablissement du caribou forestier du Québec. 2010. Bilan intérimaire du Plan de rétablissement du caribou forestier (Rangifer tarandus) au Québec pour la période 2005-2010. Québec, QC, Ministère des Ressources naturelles et de la Faune, 27 p. + Appendices.
Équipe de rétablissement du caribou forestier du Québec. 2013. Plan de rétablissement du caribou forestier (Rangifer tarandus caribou) au Québec -- 2013-2023. Québec, QC, Ministère Développement durable, Environnement, Faune et Parcs, 110 p.
Faille, G., (and 6 co-authors). 2010. Range fidelity: The missing link between caribou decline and habitat alteration? Biological Conservation 143: 2840-2850.
Fall, A., M.-J. Fortin, M. Manseau, and D. O’Brien. 2007. Spatial graphs: principles and application for habitat connectivity. Ecosystems 10: 448-461.
Ferguson, S.H., and P.C. Elkie. 2004. Seasonal movement patterns of woodland caribou (Rangifer tarandus caribou). Journal of the Zoological Society of London 262: 125-134.
Festa-Bianchet, M., J.C. Ray, S. Boutin, S. Côté, and A. Gunn. 2011. Conservation of caribou (Rangifer tarandus) in Canada: an uncertain future. Canadian Journal of Zoology 89: 419-434.
Fischer, L.A., and C.C. Gates. 2005. Competition potential between sympatric woodland caribou and wood bison in southwestern Yukon, Canada. Canadian Journal of Zoology 83: 1162-1173.
Fisher, J.T., and L. Wilkinson. 2005. The response of mammals to forest fire and timber harvest in the North American boreal forest. Mammal Review 35: 51-81.
Flagstad, Ø., and K.H. Røed. 2003. Refugial origins of reindeer (Rangifer tarandus L.) inferred from mitochondrial DNA sequences. Evolution 57: 658-670.
Flannigan, M., (and 4 co-authors). 2005. Future area burned in Canada. Climatic Change 72:1-16.
Fortin, D., (and 7 co-authors). 2013. Movement responses of caribou to human-induced habitat edges lead to their aggregation near anthropogenic features. The American Naturalist 181: 827-836.
Fortin, D., R. Courtois, P. Etcheverry, C. Dussault, and A. Gingras. 2008. Winter selection of landscapes by woodland caribou: behavioural response to geographical gradients in habitat attributes. Journal of Applied Ecology 45: 1392-1400.
Fortin, D., C. (and 10 co-authors). 2011. Partial harvesting in old-growth boreal forests and the preservation of animal diversity from ants to woodland caribou. Woodlands: Ecology, Management and Conservation. E. B. Wallace, Nova Science Publishers.
Fournier, N., and R. Faubert. 2001. Évaluation du troupeau de caribous de la Gaspésie. Direction de la Recherche sur la Faune et Direction de l’Aménagement de la Faune de la Gaspésie-Îles-de-la-Madeleine, Société de la Faune et des Parcs du Québec, Sainte-Anne-des-Monts, Canada.
Gagnon, R., and H. Morin. 2001. Les forêts d’épinette noire du Québec: dynamique, pertubations et biodiversité. Naturaliste canadien 125: 26-35.
Galpern, P., and M. Manseau. 2013a. Modeling the influence of landscape connectivity on animal distribution: a functional grain approach. Ecography 36: 1-13.
Galpern, P., and M. Manseau. 2013b. Finding the functional grain: comparing methods for scaling resistance surface. Landscape Ecology 28: 1269-1281.
Galpern, P., M. Manseau, and P. Wilson. 2012. Grains of connectivity: analysis at multiple spatial scales in landscape genetics. Molecular Ecology 21: 3996-4009.
Gamberg, M. 2004. Contaminants in Yukon moose and caribou – 2003. Prepared for Yukon Contaminants Committee, and Dept. Indian and Northern Affairs. Whitehorse, Yukon. 24 p.
Gaudry, W. 2013. Impact des structures anthropiques linéaires sur la sélection d’habitat du caribou, de l’ours noir et du coyote en Gaspésie. Mémoire de maîtrise, Université du Québec à Rimouski, Rimouski, 113 p.
Geist, V. 1998. Deer of the World: Their Evolution, Behavior, and Ecology, Stackpole Books.
Geist, V. 2007. Defining subspecies, invalid taxonomic tools, and the fate of the woodland caribou. Rangifer Special Issue No. 17:25-28.
Georges, S., G. Rivard, and G. Lemay. 1976. Détermination de l’aire de distribution du caribou (Rangifer tarandus) dans le Parc de la Gaspésie et ce en fonction de l’utilisation optimale de la matière ligneuse. Québec, Service de la faune, Ministère du Tourisme, de la Chasse et de la Pêche.
Georges, S. 1976. A range extension of the coyote in Québec. Canadian Field-Naturalist 90: 78-79.
Gerhart, K.L., R.G. White, R.D. Cameron, D.E. Russell, and D. van de Wetering. 1997. Pregnancy rate as an indicator of nutritional status in Rangifer: Implications of lactational infertility. Rangifer 17: 21-24.
Gingras, J. 2013. Condition corporelle et fécondité des orignaux de l’Est du Québec en réponse à la variation de la densité. M. Sc. Thesis, Laval University, Québec, Québec. 61 p. + appendix.
Girard, Caroline. 2014. Chairperson. Hunting, Fishing and Trapping Coordinating Committee, Montreal, QC. Letter to Neil Jones, November 2011.
Gordon, B. 2003. Rangifer and man: An ancient relationship. Rangifer Special Issue No. 14: 15-28.
Gosse, J., L. Hermanutz, B. McLaren, P. Deering, and T. Knight. 2011. Degradation of boreal forests by non-native herbivores in Newfoundland’s National Parks: Recommendations for ecosystem restoration. Natural Areas Journal 31: 331-339.
Government of Manitoba 2014. Transportation systems planning and development. http://www.gov.mb.ca/mit/tspd/current.html. [Accessed August 2014].
Government of Quebec 2014. Report on mineral activities in Quebec 2012. http://www.mern.gouv.qc.ca/english/publications/mines/publications/publication-2012-chapter1.pdf. [Accessed August 2014].
Grayson, D., and F. Delpeche. 2005. Pleistocene reindeer and global warming. Conservation Biology 19: 557-562.
Greenpeace 2010. Boreal refuge. http://www.greenpeace.org/canada/Global/canada/report/2010/5/Boreal_refuge/QC%20Intact%20Factsheet.pdf. [Accessed August 2014].
Guay, D. 1983. Histoires vraies de la chasse au Québec. Montréal, VLB Éditeur.
Gunn, A., and R.J. Irvine. 2003. Subclinical parasitism and ruminant foraging strategies - A review. Wildlife Society Bulletin 31: 117-126.
Gunn, A., D. Russell, and J. Eamer. 2011. Northern caribou population trends in Canada. Canadian Biodiversity: Ecosystem Status and Trends 2010, Technical Thematic Report. Ottawa, ON, Canadian Council of Resource Ministers. 10: 71.
Gustine, D.D., K.L. Parker, R.J. Lay, M.P. Gillingham, and D.C. Heard. 2006. Calf survival of woodland caribou in a multi-predator ecosystem. Wildlife Monographs 165: 1-32.
Hall, L. 1984. White-tailed Deer Ecology and Management. Stackpole Books, Harrisburg, PA. 870 p.
Harrington, F.H. and A.M. Veitch. 1991. Short-term impacts of low-level jet fighter training on Caribou in Labrador. Arctic 44(4):318-327.
Harrington, F.H. and A.M. Veitch. 1992. Calving Success of woodland caribou exposed to low-level jet fighter overflights. Arctic, 45(3): 213-218.
Hatter, I. W. & Bergerud, A.T. 1991. Moose recruitment, adult mortality, and rate of change. Alces 27: 65-73.
Hayes, R.D., (and 8 co-authors). 2003. Experimental reduction of wolves in the Yukon: Ungulate responses and management implications. Wildlife Monographs 152: 1-35.
Hébert, I. 2012. The habitat preferences of Newfoundland woodland caribou across range components and scales: implications for management. M.Sc. thesis. Biological Sciences. Concordia University, Montréal.
Hébert, I., and R.B. Weladji. 2013. The use of coniferous forests and cutovers by Newfoundland woodland caribou. Journal of Forest Ecology and Management 291: 318-325.
Hervieux, D., (and 6 co-authors). 2013. Widespread declines in woodland caribou (Rangifer tarandus caribou) continue in Alberta. Canadian Journal of Zoology 91:872-882.
Hervieux, D., (and 4 co-authors). 2014. Managing wolves (Canis lupus) to recover threatened woodland caribou (Rangifer tarandus caribou) in Alberta. Canadian Journal of Zoology 92:1029-1037.
Hettinga, P.N., (and 5 co-authors). 2012. Estimating size and trend of the North Interlake woodland caribou population using fecal-DNA and capture–recapture models. Journal of Wildlife Management 76: 1153-1164.
Hins, C., J.-P. Ouellet, C. Dussault, and M.-H. St-Laurent. 2009. Habitat selection by forest-dwelling caribou in managed boreal forest of eastern Canada: Evidence of a landscape configuration effect. Forest Ecology and Management 257: 636-643.
Holt, R. D. 1977. Predation, apparent competition, and the structure of prey communities. Theoretical Population Biology 12: 197-229.
Humber, J., C. Doucet, and C. Smith. 2009. Body condition of Newfoundland’s woodland caribou: A factor in current population declines? Department of Environment and Conservation, Wildlife Division. 48 p. + 3 Appendixes.
Hummel, M., and J. C. Ray. 2008. Caribou and the North - A shared future. Toronto, ON, Dundurn Press.
Huntington, H. P., S. Fox, F. Berkes, I. Krupnik, and et al. 2005. The changing Arctic: Indigenous perspectives. Pages 61-98 in Arctic Climate Impact Assessment (ACIA). Cambridge University Press, Cambridge.
IUCN. 2008. IUCN Red List. http://www.iucnredlist.org/details/29742/0 Accessed 21 February 2013.
IUCN Standards and Petitions Subcommittee. 2013. Guidelines for Using the IUCN Red List Categories and Criteria. Version 10.1. Prepared by the Standards and Petitions Subcommittee. From http://www.iucnredlist.org/documents/RedListGuidelines.pdf.
James, A.R.C., and A.K. Stuart-Smith. 2000. Distribution of caribou and wolves in relation to linear corridors. Journal of Wildlife Management 64: 154-159.
James, A.R.C., S. Boutin, D.M. Hébert, and A.B. Rippin. 2004. Spatial separation of caribou from moose and its relation to predation by wolves. Journal of Wildlife Management 68: 799-809.
Johnson, C.J., and 6 co-authors. 2005. Cumulative effects of human developments on arctic wildlife. Wildlife Monographs: 1-36.
Johnson, C.J., K.L. Parker, D.C. Heard, and D.R. Seip. 2004. Movements, foraging habits, and habitat use strategies of northern woodland caribou during winter: Implications for forest practices in British Columbia. BC Journal of Ecosystems and Management 5: 22-35.
Johnson, D., (and 5 co-authors). 2010. Serum biochemistry, serology, and parasitology of boreal caribou (Rangifer tarandus caribou) in the Northwest Territories, Canada. Journal of Wildlife Diseases 46:1096-1107.
Joro Consultants inc. 2011. Bipole III Transmission Project - Caribou Technical report - Manitoba Hydro. 179 p. + 3 appendices.
Kays, R.W., M.E. Gompper, and J.C. Ray. 2008. Landscape ecology of eastern coyotes based on large-scale estimates of abundance. Ecological Applications 18: 1014-1027.
Kelly, A., and K. Cox. 2011. Boreal Caribou Progress Report: Hay River Lowlands and Cameron Hills Study Areas, 2008-2010. Dept. Environment and Natural Resources, Govt. Northwest Territories, Fort Smith, NT. 29 pp.
Kelsall, J.P. 1968. The migratory barren-ground caribou of Canada. Ottawa, ON, Queen’s Printer.
Klütsch, C.F.C., M. Manseau, and P.J. Wilson. 2012. Phylogeographical analysis of mtDNA data indicates postglacial expansion from multiple glacial refugia in woodland caribou (Rangifer tarandus caribou). PLoS ONE 7: e52661.
Koper, K., and M. Manseau. 2009. Generalized estimating equations and generalized linear mixed-effects models for modeling resource selection. Journal of Animal Ecology 46: 590-599.
Krawchuk, M., S, Cumming, and M. Flannigan. 2009. Predicted changes in fire weather suggests increases in lightning fire initiation and future area burned in the mixedwood boreal forest. Ambio 92:83-97.
Kutz, S. J. Faculty of Veterinary Medicine. University of Calgary, Calgary, AB. Personal communication to G. Forbes, August 2014.
Kutz, S.J., (and 6 co-authors). 2009. The Arctic as a model for anticipating, preventing, and mitigating climate change impacts on host-parasite interactions. Veterinary Parasitology 163: 217-228.
Laliberte, A.S., and W.J. Ripple. 2004. Range contractions of North American carnivores and ungulates. Bioscience 54: 123-138.
Lalonde, M. 2014. Ministère du Développement durable, de l’Environnement, de la Faune et des Parcs, Ste-Anne-des-Monts, QC. Voice and email correspondence with G. Forbes July-September 2014.
Lalonde, M. 2013. Inventaire aérien de la population de caribou de la Gaspésie (Rangifer tarandus caribou), automne 2013, Ministère du Développement durable, de l’Environnement, de la Faune et des Parcs, Ste-Anne-des-Monts, QC. 20 p.
Lalonde, M., and J. Michaud. 2013. Inventaire aérien de la population de caribou de la Gaspésie (Rangifer tarandus caribou), automne 2012. Ministère des Ressources naturelles, Ste-Anne-des-Monts, QC. 19 p.
Lamoureux, J., A. Pelletier, M. Bélanger, and C. Larocque. 2007. Inventaire aérien de l’orignal dans les réserves fauniques de Matane et de Dunière à l’hiver 2007. Ministère des Ressources naturelles et de la Faune, Rimouski, QC. 30 p.
Lankester, M.W., and D. Fong. 1998. Protostrongylid nematodes in caribou (Rangifer tarandus caribou) and moose (Alces alces) of Newfoundland. Rangifer Special Issue No. 10: 73-83.
Lankester, M.W., and S. Luttich. 1988. Fascioloides magna (Trematoda) in woodland caribou (Rangifer tarandus caribou) of the George River sub-population, Labrador. Canadian Journal of Zoology 66: 475-479.
Lantin, É., P. Drapeau, M. Paré, and Y. Bergeron. 2003. Preliminary assessment of habitat characteristics of woodland caribou calving areas in the Claybelt region of Québec and Ontario, Canada. Rangifer Special Issue 14:247–254.
Larter, N. C., and D. G. Allaire. 2014. Dehcho Boreal Caribou Study Progress Report, April 2014. Fort Simpson, Dept. Environment and Natural Resources, Govt. Northwest Territories, Fort Simpson, NT. 35 pp.
Latham, A.D.M., M.C. Latham, M.S. Boyce, and S. Boutin. 2011a. Movement responses by wolves to industrial linear features and their effect on woodland caribou in northeastern Alberta. Ecological Applications 21: 2854-2865.
Latham, A.D.M., M.C. Latham, N.A. McCutchen, and S. Boutin. 2011b. Invading white-tailed deer change wolf-caribou dynamics in northeastern Alberta. Journal of Wildlife Management 75: 204-212.
Leblond, M., C. Dussault, and J.-P. Ouellet. 2013a. Avoidance of roads by large herbivores and its relation to disturbance intensity. Journal of Zoology 289: 32-40.
Leblond, M., C. Dussault, and J.-P. Ouellet. 2013b. Impacts of human disturbance on large prey species: Do behavioral reactions translate to fitness consequences? PLos ONE 8: e73695.
Leblond, M., (and 5 co-authors). 2011. Assessing the influence of resource covariates at multiple spatial scales: an application to forest-dwelling caribou faced with intensive human activity. Landscape Ecology 26: 1433-1446.
Leblond, M., C. Dussault & M.-H. St-Laurent (2014). Development and validation of an expert-based habitat suitability model to support boreal caribou conservation. Biological Conservation 177: 100-108.
Leclerc, M., C. Dussault, and M.-H. St-Laurent. 2012. Multi-scale assessment of the impacts of roads and cutovers on calving site selection in woodland caribou. Forest Ecology and Management 286: 59-65.
Leclerc, M., C. Dussault & M.-H. St-Laurent. 2014. Behavioural strategies towards human disturbances explain individual performance in woodland caribou. Oecologia 176:297-306.
Le Goff, H., M. Flannigan, and Y. Bergeron. 2009. Potential changes in monthly fire risk in the eastern Canadian boreal forest under future climate change. Canadian Journal of Forest Research 39: 2369-2380.
Lee, P.G. 2012. Canada’s woodland caribou: Industrial disturbances in their ranges and implications for their survival. Global Forest Watch Canada International Year of Sustainable Energy, Publication 1. Edmonton. 13 pp. ISBN 978-0-9867907-9-9.
Lesmerises, F. 2012. Analyses de viabilité de la population de caribou des bois (Rangifer tarandus caribou) de la Gaspésie. Rimouski, QC. 34 p.
Lesmerises, R., J.-P. Ouellet, C. Dussault, and M.-H. St-Laurent. 2013. The influence of landscape matrix on isolated patch use by wide-ranging animals: conservation lessons for woodland caribou. Ecology and Evolution 3: 2880-2891.
Lewis, K. P., S. Gullage, D. Fifield, D. Jennings, and S. Mahoney. 2014. Experimental manipulations of black bear (Ursus americanus) and eastern coyote (Canis latrans) to improve caribou calf survival in southern Newfoundland 2008–2013. Technical Bulletin No. 010. Sustainable Development and Strategic Science. Department of Environment and Conservation, Newfoundland, Canada. 52 p.
Lewis, K. P. and S. Mahoney. 2014. Caribou survival, fate, and cause of mortality in Newfoundland: a summary and analysis of the patterns and causes of caribou survival and mortality in Newfoundland during a period of rapid population decline (2003-2012). Technical Bulletin No. 009, Sustainable Development and Strategic Science. Government of Newfoundland and Labrador, St. John’s, NL. 62 p.
L’Italien, L., (and 5 co-authors). 2012. Mating group size and stability in reindeer, Rangifer tarandus: the effects of male characteristics, sex ratio, and male age structure. Ethology 118:783-792.
Losier, C. 2013. Les réponses fonctionnelles dans la sélection de l’habitat influencant la survie du caribou forestier en forêt boréale. M.Sc., Université Laval, Québec, QC.
Macbeth, B., (and 5 co-authors). 2014. The role of health in management and conservation of Boreal Caribou herds in northeastern British Columbia. Poster presentation. 15th North American Caribou Conference. May 2014, Whitehorse, YK.
Mackenzie Valley Review Board. 2013. Fortune NICO Mine. Report of Environmental Assessment and Reasons for Decision. http://www.reviewboard.ca/upload/project_document/EA0809-004_NICO_Report_of_EA_and_Reasons_for_Decision_corrected.PDF [Accessed July 2014].
Mahant, S.P. 2013. Effects of snowmobile trails on woodland caribou habitat selection in Gros Morne National Park. Honours thesis, Trent University, Peterborough, Canada.
Mahoney, S.P., (and 4 co-authors). 2001. Caribou reactions to provocation by snowmachines in Newfoundland. Rangifer 21:35-43.
Mahoney, S.P., and J.A. Schaefer. 2002a. Hydro-electric development and the disruption of migration in caribou. Biological Conservation 107: 147-153.
Mahoney, S.P., and J.A. Schaefer. 2002b. Long-term changes in demography and migration of Newfoundland caribou. Journal of Mammalogy 83: 957-963.
Mahoney, S., and J.A. Schaefer. 2011. Three decades of habitat selection by Newfoundland caribou. Peterborough, ON. 19 p.
Mahoney, S.P., and J.A. Virgl. 2003. Habitat selection and demography of a nonmigratory woodland caribou population in Newfoundland. Canadian Journal of Zoology 81: 321-334.
Mahoney, S.P., and J.N. Weir. 2009. Caribou data synthesis – Progress report – Overview of the status of woodland caribou in insular Newfoundland: Research methodology, results, interpretations and future projections. St. John’s, NL, Sustainable Development and Strategic Science, Gov. of Newfoundland and Labrador.
Mahoney, S.P., (and 4 co-authors). 2011. Morphological change in Newfoundland caribou: Effects of abundance and climate. Rangifer 31:21-34.
Manitoba Government. 2006. Manitoba’s Conservation and Recovery Strategy for Boreal Woodland Caribou (Rangifer tarandus caribou). 20 pp.
Manitoba Boreal Woodland Caribou Management Committee. 2014. Conserving the Icon of the Boreal, Manitoba’s Boreal Woodland Caribou (Rangifer tarandus caribou) Recovery Strategy. Manitoba Conservation and Water Stewardship. Winnipeg, Manitoba. 30 pp.
Mayor, S.J., J.A. Schaefer, D.C. Schneider, and S.P. Mahoney. 2007. Spectrum of scale selection: new approaches to detecting the scale-dependent response to habitat. Ecology 88: 1634-1640.
Mayor, S.J., J.A. Schaefer, D.C. Schneider, and S.P. Mahoney. 2009. The spatial structure of habitat selection: A caribou’s-eye-view. Acta Oecologica 35:253-260.
McCarthy, S.C., R.B. Weladji, C. Doucet, and P. Saunders. 2011. Woodland caribou calf recruitment in relation to calving/post-calving landscape composition. Rangifer 31: 35-47.
McCollough, M., and B. Connery. 1991. An attempt to reintroduce woodland caribou to Maine 1986-1990. Final Report. Maine Caribou Project, 1986-1990. Orono: University of Maine.
McCutchen, N.A. 2006. Factors affecting caribou survival in northern Alberta: the role of wolves, moose and linear features. Ph.D. Thesis, Univ. of Alberta, Edmonton, AB.
McDevitt, A., (and 7 co-authors). 2009. Survival in the Rockies of an endangered hybrid swarm from diverged caribou (Rangifer tarandus) lineages. Molecular Ecology 18: 665-679.
McDonald, M., L. Arragutainaq, and Z. Novalinga. 1997. Voices from the Bay: Traditional Ecological Knowledge of Inuit and Cree in the James Bay Bioregion. Ottawa, Ontario.
McEwan, E. H. and P. E. Whitehead. 1972. Reproduction in female reindeer and caribou. Canadian Journal of Zoology 504: 3-46.
McLaren, B.E., B.A. Roberts, N. Djan-Chékar, and K.P. Lewis. 2004. Effects of overabundant moose on the Newfoundland landscape. Alces 40: 45-59.
McLoughlin, P.D., E. Dzus, B. Wynes, and S. Boutin. 2003. Declines in populations of woodland caribou. Journal of Wildlife Management 67: 755-761.
McRae, D.J., L.C. Duchesne, B. Freedman, T.J. Lynham, and S. Woodley 2001. Comparisons between wildfire and forest harvesting and their implications in forest management. Environmental Reviews 9: 223-260.
Mercer, E., S.P. Mahoney, K. Curnew, and C. Finlay. 1985. Distribution and abundance of insular Newfoundland caribou and the effects of human activities. Proceedings of the Second North American Caribou Workshop, (Meredith, T.C., and Martell, A.M., eds.), Val Morin, Québec.
McGill Subarctic Res. Paper 40: 15-32.
Miller, A.M. 2010. Living with Boreal Forest Fires: Anishinaabe Perspectives on Disturbance and Collaborative Forestry Planning, Pikangikum First Nation, Northwestern Ontario. University of Manitoba, Winnipeg, Manitoba.
Mitchell, G.B., (and 7 co-authors). 2012. Experimental oral transmission of Chronic Wasting Disease to reindeer (Rangifer tarandus tarandus). PLoS ONE 7: e39055.
Moisan, G. 1958. Le caribou de la Gaspésie. Extrait du Naturaliste canadien No 83: 225-234, 262-274, No 84: 5-27. Québec, Qc. 83, 84: 225-234, 262-274, 225-227.
Moreau, G., D. Fortin, S. Couturier, and T. Duchesne. 2012. Multi-level functional responses for wildlife conservation: the case of threatened caribou in managed boreal forests. Journal of Applied Ecology 49: 611–620.
Mosnier, A., D. Boisjoly, R. Courtois, and J.-P. Ouellet. 2008a. Extensive predator space use can limit the efficacy of a control program. Journal of Wildlife Management 72: 483-491.
Mosnier, A., J.-P. Ouellet, and R. Courtois. 2008b. Black bear adaptation to low productivity in the boreal forest. Écoscience 15: 485-497.
Mosnier, A., J.-P. Ouellet, L. Sirois, and N. Fournier. 2003. Habitat selection and home-range dynamics of the Gaspé caribou: a hierarchical analysis. Canadian Journal of Zoology 81: 1174-1184.
Müller, D., (and 4 co-authors). 2010. Relevance of management and feeding regimens on life expectancy in captive deer. American Journal of Veterinary Research 71:275-280.
Nagy, J. 2011. Use of space by caribou in northern Canada. Ph.D. Thesis, Biological Sciences, University of Alberta.
Nagy, J.A., and 8 co-authors. 2011. Subpopulation structure of caribou (Rangifer tarandus L.) in arctic and subarctic Canada. Ecological Applications 21: 2334-2348.
Nalcor 2013. Lower Churchill Hydroelectric Generation Project. http://muskratfalls.nalcorenergy.com/wp-content/uploads/2013/03/Plain-Lang-Summary-ENG-HI-RES.pdf. [Accessed August 2014].
Nalcor (date unknown). Red wine mountain caribou habitat suitability modeling. http://ceaa-acee.gc.ca/050/documents/48786/48786F.pdf. [Accessed August 2014].
NatureServe. 2013. NatureServe Explorer: An online encyclopedia of life [web application]. Version 7.1. NatureServe, Arlington, Virginia. Available http://www.natureserve.org/explorer. [Accessed: October 28, 2013].
Newfoundland and Labrador Government. 2012. 2012-2013 Hunting and Trapping Guide. Department of Environment and Conservation, Wildlife Division. St. John’s, NF www.env.gov.nl.ca/env/publications/wildlife.
Northern Miner 2014. Quebec government relaunches Plan Nord. June 18, 2014. http://www.northernminer.com/news/quebec-relaunches-plan-nord/1003103496/ [Accessed August 2014].
NT Species at Risk Committee. 2012. Species status report Boreal caribou (Rangifer tarandus caribou) - Threatened December 2012. Government of the NT, Yellowknife, NT. 148 p.
NT Department of Environment and Natural Resources. 2010. Action Plan - Boreal Woodland caribou conservation in the Northwest Territories 2010-2015. Yellowknife, NT, NT Dept. of Environment and Natural Resources.
O’Brien, D., M. Manseau, A. Fall, and M.-J. Fortin. 2006. Testing the importance of spatial configuration of winter habitat for woodland caribou: An application of the graph theory. Biological Conservation 130: 70-83.
O’Flaherty, R.M., I. Davidson-Hunt, and M. Manseau. 2007. Keeping woodland caribou in the Whitefeather Forest. Edmonton, Alberta. SFM Network, University of Alberta.
Ontario Woodland Caribou Recovery Team. 2008. Woodland Caribou (Rangifer tarandus caribou) (Forest-dwelling, Boreal Population) in Ontario. Queen’s Printer, Toronto, ON. 93 pp.
Ontario Ministry of Natural Resources. 2009. Ontario’s Woodland Caribou Conservation Plan. Queen’s Printer, Toronto, ON. 24 pp.
Ontario Ministry of Natural Resources. 2012. Ontario’s Woodland Caribou Conservation Plan Progress Report Winter 2012. Queen’s Printer, Toronto, ON. 17 pp.
Ontario Ministry of Natural Resources. 2013a. General habitat description of the forest-dwelling woodland caribou (Rangifer tarandus caribou). Species at Risk Branch, Peterborough, Ontario. 15 pp. [http://files.ontario.ca/environment-and-energy/species-at-risk/mnr_sar_ghd_car_en.pdf]. Accessed 24 July 2014.
Ontario Ministry of Natural Resources. 2013b. Range Management Policy in Support of Woodland Caribou Conservation and Recovery. Ontario Ministry of Natural Resources, Species at Risk Branch, Peterborough, Ontario. 24pp.
Ontario Ministry of Natural Resources. 2014. State of Woodland Caribou Resources Report (DRAFT). Ontario Ministry of Natural Resources, Species at Risk Branch, Peterborough, Ontario. 172pp.
Ouellet, J.-P., J. Ferron, and L. Sirois. 1996. Space and habitat use by the threatened Gaspé caribou in southeastern Québec. Canadian Journal of Zoology 74: 1922–1933.
Pardy Moores, Shelley. Department of Environment and Conservation, Corner Brook, NFL. Email correspondence with G. Forbes, August 2014.
Parisien, M.A., L. Sirois, and M. Babeau. 2004. Distribution and dynamics of jack pine at its longitudinal range limits in Québec. Proceedings of the 22nd Tall Timbers Fire Ecology Conference, 1999, Kananaskis.
Parker, K.L., Barboza, P.S., & Stephenson, T.R. 2005. Protein conservation in female caribou (Rangifer tarandus): effects of decreasing diet quality during winter. Journal of Mammalogy 86(3): 610-622.
Parmesan, C., and G. Yohe. 2003. A globally coherent fingerprint of climate change impacts across natural systems. Nature 421: 37-42.
Peek, J, 2007. Habitat requirements. pp. 351-376 in Ecology and Management of North American Moose. A. Franzmann and C. Schwartz (eds.). University of Colorado Press, CO. 733 p.
Peek, J., (and 9 co-authors). 2012. Management of Large Mammalian Carnivores in North America. The Wildlife Society Technical Review 12-1. The Wildlife Society, Bethesda, Maryland, USA.
Peters, W., M. Hebblewhite, N. DeCesare, F. Cagnacci, and M. Musiani. 2013. Resource separation analysis with moose indicates threats to caribou in human altered landscapes. Ecography 36: 487-498.
Pickles, R., (and 4 co-authors). 2013. Predicting shifts in parasite distribution with climate change: a multitrophic level approach. Global Change Biology 19 :2645-2654.
Pilgrim, B., E. Perry, and D. Marshall. 2012. Species Identification of the Bonavista Canid Collected 12 March 2012 on the Island of Newfoundland. Final Report for the Department of Environment and Conservation, St. John’s, NL.
Pilon, E. 1997. Bilan du contrôle de prédateurs effectué de 1990 à 1996 dans le cadre du plan de soutien du caribou (Rangifer tarandus) du parc de la Gaspésie. Société de la Faune et des Parcs du Québec, Ste-Anne-des-Monts, QC.
Pinard, V., C. Dussault, J.-P. Ouellet, D. Fortin, and R. Courtois. 2012. Calving rate, calf survival rate, and habitat selection of forest-dwelling caribou in a highly managed landscape. Journal of Wildlife Management 76: 189-199.
Pitt, W.C. and P.A. Jordan. 1994. A survey of the nematode parasite Parelaphostrongylus tenuis in the white-tailed deer, Odocoileus virginianus, in a region proposed for caribou, Rangifer tarandus caribou, re-introduction in Minnesota. Canadian Field-Naturalist 108(3):341-346.
Popp, J., J. Schaefer, and F. Mallory. 2011. Female site fidelity of the Mealy Mountain caribou sub-population in Labrador. Rangifer 19:87-95.
Quebec Ministère des Ressources naturelles 2013. Plan d’aménagement forestier de l’aire de frequentation du caribou de la Gaspésie. 3e édition. 2013-2018. 36 p.
Racey, G.D., and T. Armstrong. 2000. Woodland caribou range occupancy in northwestern Ontario: past and present. Rangifer Special Issue 12:173-184.
Racey, G.D., and A.A. Arsenault. 2007. In search of a critical habitat concept for woodland caribou, boreal population. Rangifer Special Issue No. 17: 29-37.
Racey, G., A. Harris, and R. Foster. 1996. Caribou winter habitat in the new forest: lessons from Lucy Lake. OMNR NWST Technical Report TR-103. OMNR Queen’s Printer for Ontario.
Racey, G., E. McCaul, R. Hartley, C. Leale, and M. Rose. 2010. Caribou in the new forest: lessons from Castlewood Lake. OMNR NWST Technical Report TR-144. OMNR Queen’s Printer for Ontario.
Randell, H., J.N. Weir, J.G. Luther, and S.P. Mahoney. 2012. Population projections of Newfoundland caribou using Population Viability Analysis. St. John’s, NL, Sustainable Development and Strategic Science, Gov. of Newfoundland and Labrador, Technical Bulletin No. 4.
Rasiulis, A.L., I. Schmelzer, and C.G. Wright. 2012. The effect of temporal sampling regime on the characterization of home range for female boreal woodland caribou (Rangifer tarandus caribou) in Labrador, Canada. Rangifer Special Issue No. 20: 227-239.
Renaud, L.-A., M. Leblond, D. Beauchesne, and M.-H. St-Laurent. 2010. Synthèse des connaissances relatives aux impacts du réseau routier sur l’écologie du caribou forestier. Ministère des Ressources naturelles et de la Faune, Québec: 21 p.
Renaud, L.-A. 2012. Impacts de l’aménagement forestier et des infrastructures humaines sur les niveaux de stress du caribou forestier. M.Sc. Thesis, Université du Québec à Rimouski.
Resource Clips 2014. Quebec Liberals revive Plan Nord infrastructure and development program. June 6 2014. http://resourceclips.com/2014/06/06/quebec-liberals-revive-plan-nord-infrastructure-and-development-program/ [Accessed August 2014].
Rettie, W.J., and F. Messier. 1998. Dynamics of woodland caribou populations at the southern limit of their range in Saskatchewan. Canadian Journal of Zoology 76: 251-259.
Rettie, W.J., and F. Messier. 2000. Hierarchical habitat selection by woodland caribou: its relationship to limiting factors. Echography 23: 466-478.
Rettie, W.J. and F. Messier. 2001. Range use and movement rates of Woodland Caribou in Saskatchewan. Can. J. Zool. 79:1933–1940.
Rivard, G. 1978. Étude du caribou de la Gaspésie en considérant l’habitat. Québec, Ministère du Tourisme, de la Chasse et de la Pêche.
Rodgers, A.R., (and 5 co-authors). 2009. Forest-dwelling woodland caribou in Ontario: Candidate study areas report. Information paper CNFER IP-004. Thunder Bay, Ontario, Ontario Ministry of Natural Resources, Centre for Northern Forest Ecosystem Research: 49 p.
Røed, K.H., M. Ferguson, M. Crête, and A.T. Bergerud. 1991. Genetic variation in transferrin as a predictor for differentiation and evolution of caribou from eastern Canada. Rangifer 11: 65-74.
Rose, M. and G. Racey. 2010. Caribou in the new forest: Lessons from South Allely lake and 20 years of caribou habitat management in the English River Forest. OMNR DRAFT NWST Technical Report TR-145. OMNR Queen’s Printer for Ontario.
Rudolph, T.D. 2011. Mouvements et sélection d’habitat lors des déplacements printaniers du caribou (Rangifer tarandus caribou) dans le Nord-du-Québec. M.Sc. Thesis, Université du Québec à Montréal, Montréal.
Rudoplh, T., and P. Drapeau. 2012. Using movement behaviour to define biological seasons for woodland caribou. Rangifer 20:295-307.
Rudolph, T.D., P. Drapeau, M.-H. St-Laurent, and L. Imbeau. 2012. Status of woodland caribou (Rangifer tarandus caribou) in the James Bay region of Northern Québec. Report presented to the Ministère des Ressources Naturelles et de la Faune du Québec and the Grand Council of the Crees (Eeyou Istchee), Montréal, Qc. 72 p.
Russell, W. 2011. Labrador Boreal caribou Aboriginal Traditional Knowledge gathering workshop - Final Report. NunatuKavut Community Council inc. 23 p. + 23 Appendices.
Saganash, N. 2013. Woodland Caribou (Boreal Population). Fostering community involvement in the acquisition of knowledge and conservation planning. Activity Report 2012-2013. Cree Regional Authority, Montreal. 90 p.
Sahtu Renewable Resources Board. 2010. Boreal caribou Traditional Knowledge collection study - The Sahtu settlement area. Tulita, NT, Sahtu Renewable Resources Board.
Samuel, W.M. 2004. White as a ghost: Winter ticks and moose. Natural History Series, Volume 1, Federation of Alberta Naturalists, Edmonton, Alberta. 100 p.
Saskatchewan Environment. 2014. Woodland caribou. http://www.environment.gov.sk.ca/woodlandcaribou. [Accessed August 2014]/
Saunders, P. 2007. Guidelines for the protection of caribou and caribou calves in or around forest harvesting operations. Dept. Environment and Conservation, Wildlife Division.
Schaefer, J.A. 2003. Long-term range recession and the persistence of caribou in the taiga. Conservation Biology 17: 1435-1439.
Schaefer, J.A., and S.P. Mahoney. 2007. Effects of progressive clearcut logging on Newfoundland caribou. Journal of Wildlife Management 71: 1753-1757.
Schaefer, J.A., and S.P. Mahoney. 2013. Spatial dynamics of the rise and fall of caribou (Rangifer tarandus) in Newfoundland. Canadian Journal of Zoology 91: 767-774.
Schaefer, J.A., and W.O.J. Pruitt. 1991. Fire and Woodland Caribou in Southeastern Manitoba. Wildlife Monographs 116: 3-39.
Schaefer, J.A., A.M. Veitch, F.H. Harrington, W.K. Brown, J.B. Theberge, and S.N. Luttich. 1999. Demography of decline of the Red Wine Mountains caribou sub-population. Journal of Wildlife Management 63: 580-587.
Schaefer, J.A., A.M. Veitch, F.H. Harrington, W.K. Brown, J.B. Theberge, and S.N. Luttich. 2001. Fuzzy structure and spatial dynamics of a declining woodland caribou population. Oecologia 126: 507-514.
Schaefer, J.A., and C.C. Wilson. 2002. The fuzzy structure of populations. Canadian Journal of Zoology 80: 2235-2241.
Schaefer, J. A., C. Bergman, and S. Luttich. 2000. Site fidelity of female caribou at multiple spatial scales. Landscape Ecology 15:731-739.
Schmelzer, I. Dept. of Environment & Conservation, Wildlife Division, Corner Brook, NL Email correspondence to G. Forbes, August 2014.
Schmelzer, I. 2014. Range use, life history and trends in abundance of forest-dwelling threatened caribou populations in Labrador: An overview. Wildlife Division, Dept. of Wildl. and Conserv., Newfoundland and Labrador Gov. 24 p.
Schmelzer, I. 2013. Survival, recruitment and population trajectory for Lac Joseph and Red Wine Mountain caribou sub-populations - A compilation to inform the COSEWIC Review process November 2012. Newfoundland and Labrador Gov., Department of Environment and Conservation, Corner Brook, NL.
Schmelzer, I., (and 15 co-authors). 2004. Recovery strategy for three woodland caribou sub-populations (Rangifer tarandus caribou; Boreal population) in Labrador. Department of Environment and Conservation, Corner Brook. Newfoundland and Labrador.
Schneider, R.R., G. Hauer, W. Adamowicz, and S. Boutin. 2010. Triage for conserving populations of threatened species: the case of woodland caribou in Alberta. Biological Conservation 143: 1603-1611.
Schramm, N., and N. Krogman. 2001. Final project report: 2001-2008 Caribou Mountain critical wildlife habitat and traditional ecological knowledge study. Sustainable Forest Management Network. Edmonton, AB.
Shuter, Jennifer. Centre for Northern Ecosystem Research, Ontario Ministry of Natural Resources. Email correspondence with G. Forbes, June 2014.
Shuter, J.L., and A.R. Rodgers. 2010. Delineating demographic units of woodland caribou (Rangifer tarandus caribou) in Ontario: cautions and insights. Rangifer Special Issue 20: 159-181.
Sleep, D.J., and C. Loehle. 2010. Validation of a demographic model for woodland caribou. Journal of Wildlife Management 74: 1508-1512.
Smith, K.G., E.J. Ficht, D. Hobson, T.C. Sorensen, and D. Hervieux. 2000. Winter distribution of woodland caribou in relation to clear-cut logging in west-central Alberta. Canadian Journal of Zoology 78: 1433-1440.
Smith, K., and L. Pittaway. 2011. Little Smoky woodland caribou calf survival enhancement project. Rangifer Special Issue No. 19: 97-102.
Sommer, R.S., (and 5 co-authors). 2011. When the pond turtle followed the reindeer: effect of the last extreme global warming event on the timing of faunal change in Northern Europe. Global Change Biology 17: 2049-2053.
Sorensen, T., (and 6 co-authors). 2008. Determining sustainable levels of cumulative effects for boreal caribou. Journal of Wildlife Management 72: 900-905.
Soulliere, C., and S. Mahoney. 2014. Food habits of Newfoundland caribou. Draft report. Newfoundland and Labrador Department of Environment and Conservation. St. John’s, NL.
St-Laurent, Martin-Hugues. Université du Québec à Rimouski, Rimouski, QC. Email correspondence with G. Forbes, June 2014.
St-Laurent, M.-H., and C. Dussault. 2012. The reintroduction of boreal caribou as a conservation strategy: A long-term assessment at the southern range limit. Rangifer Special Issue No. 20: 127-138.
St-Laurent, M.-H., J.-P. Ouellet, A. Mosnier, D. Boisjoly, and R. Courtois. 2009. Le parc national de la Gaspésie est-il un outil de conservation efficace pour maintenir une population menacée de caribou? Le Naturaliste canadien 133: 6-14.
St-Martin, G. 1987. The ecology of eastern-central Quebec and western Labrador caribou populations as it relates to proposed road development. MSc. Thesis, University of Waterloo, Waterloo, Ontario.
Stone, I., J.-P. Ouellet, L. Sirois, M.J. Arseneau, and M.-H. St-Laurent. 2008. Impacts of silvicultural treatments on arboreal lichen biomass in balsam fir stands on Québec’s Gaspé Peninsula: Implications for a relict caribou sub-population. Forest Ecology and Management 28: 2733-2742.
Stuart-Smith, A.K., C.J. Bradshaw, S. Boutin, D.M. Hébert, and A.B. Rippin. 1997. Woodland caribou relative to landscape patterns in northeastern Alberta. Journal of Wildlife Management 61: 622-633.
Tapscott, B. 2011. Chronic Wasting Disease - Fact sheet. Ontario Ministry of Agriculture and Food. 7 p.
Thiessen, C. 2009. Peace Region Boreal caribou monitoring: Annual report 2008-09. Ministry of Environment, Fort St-John, BC. 27 p + appendices.
Thomas, D., S. Barry, and G. Alaie. 1995. Fire-caribou-winter range relationships in northern Canada. Rangifer 16:57-67.
Thomas, D. C., and H. Kiliaan. 1998. Fire-caribou relationships: (ll) Fecundity and physical condition of the Beverly sub-population. Technical Report Series. Edmonton, AB, Canadian Wildlife Service, Prairie and Northern Region. 310: 96.
Thompson, I.D., M.D. Flannigan, B.M. Wotton, and R. Suffling. 1998. The effects of climate change on landscape diversity: an example in Ontario forests. Environmental Monitoring and Assessment 49: 213-233.
Tremblay-Gendron, S. 2012. Influence des proies sur le déplacement d’un prédateur : étude du système loup-orignal-caribou. M.Sc. Thesis, Université du Québec à Rimouski. Rimouski, QC. 68 p.
Trindade, M., F. Norman, K.P. Lewis, S.P. Mahoney, J. Weir, and C. Soulliere. 2011. Caribou Calf Mortality Study. A summary and analysis of the patterns and causes of caribou calf mortality in Newfoundland during a period of rapid population decline: 2003-2007. Gov. of Newfoundland and Labrador, Technical Bulletin No. 2, St. John’s, NL.
Turcotte, C., (and 5 co-authors). 2007. Plan d’aménagement de l’aire du caribou de la Gaspésie (Rangifer tarandus caribou), 2e édition. Ste-Anne-des-Monts, QC, Ministère des Ressources naturelles et de la Faune du Québec: 85 p.
USask. 2014. U of S to lead $2 million study of woodland caribou in northern Saskatchewan. [http://words.usask.ca/news/2014/04/25/u-of-s-to-lead-2-m-study-of-woodland-caribou-in-northern-saskatchewan/]. Accessed 28 July 2014.
Vistnes, I., and C. Nellemann. 2008. The matter of spatial and temporal scales: a review of reindeer and caribou response to human activity. Polar Biology 31: 399-407.
Vors, L. S. 2013. Caribou in Canada: Ecology and policy. Ph.D. Thesis, University of Alberta, Edmonton, AB.
Vors, L.S., J.A. Schaefer, B.A. Pond, A.R. Rodgers, and B.R. Patterson. 2007. Woodland caribou extirpation and anthropogenic landscape disturbance in Ontario. Journal of Wildlife Management 71: 1249-1256.
Vors, L. S., and M. Boyce. 2009. Global decline of caribou and reindeer. Global Change Biology 15: 2626–2633.
Wasel, S., W. Samuel, and V. Crichton. 2003. Distribution and ecology of Parelaphostryongylus tenuis (Nematoda), in northcentral North America. Journal of Wildlife Diseases 39:338-346.
Wasser, S.K., J.L. Keim, M.L. Taper, and S.R. Lele. 2011. The influences of wolf predation, habitat loss, and human activity on caribou and moose in the Alberta oil sands. Frontiers in Ecology and the Environment.
Watton, D., and F. Dunn 2003. The Ontario Forest Accord: A landmark in forest policy. Proceedings XII World Forestry Congress, Quebec City, QC. http://www.fao.org/docrep/ARTICLE/WFC/XII/0557-C1.HTM. [Accessed June 2014].
Weclaw, P., and R.J. Hudson. 2004. Simulation of conservation and management of Woodland Caribou. Ecological Modelling 157: 75-94.
Wek’èehzìi Renewable Resources Board. 2013. Overview: Boreal caribou habitat use in Wek’èehzìi. http://www.wrrb.ca/sites/default/files/Boreal%20Caribou%20in%20Wekeezhii%20FINAL%20REPORT%203may13_0.pdf. [Accessed July 2014].
Weir, J., S.P. Mahoney, B. McLaren, and S.H. Ferguson. 2007. Effects of mine development on woodland caribou Rangifer tarandus distribution. Wildlife Biology 13: 66-74.
Weir, J. N., S. F. Morrison, J. G. Luther, and S. P. Mahoney. 2014. Caribou Data Synthesis – Progress Report #2. Status of the Newfoundland population of woodland caribou. Technical Bulletin No. 008, Sustainable Development and Strategic Science, Government of Newfoundland and Labrador, St. John’s, NL. 52 p.
Welch, A., M. Samuel, and J. Wilke. 1990. Dermacentor albipictus (Acari, Ixodidae) on captive reindeer and free-ranging woodland caribou. Journal of Wildlife Diseases 26: 410–411.
Wells, J., J. Jacobs, I. Goudie, and J. Feldgajer. 2011. Intact habitat landscapes and woodland caribou on the island of Newfoundland. Canadian Boreal Initiative, Ottawa, Canada.
Wiken, E. B. 1996. Ecozones of Canada. Ottawa, ON, Environment Canada.
Wild Species. 2010. http://www.wildspecies.ca/ResultSimple.cfm?lang=e Accessed 8 January 2014.
Wilkerson, C. 2010. Population genetics of woodland caribou (Rangifer tarandus caribou) on the island of Newfoundland. M.Sc. Thesis, Memorial University of Newfoundland.
Wilkinson, C.J.A. 2010. An analysis of government actions for the protection and recovery of forest-dwelling woodland caribou (Rangifer tarandus caribou) in Ontario, Canada. Rangifer 30: 67-77;
Wilkinson, C.J.A. 2010. An analysis of government actions for the protection and recovery of forest-dwelling woodland caribou (Rangifer tarandus caribou) in Ontario, Canada. Rangifer 30: 67-77;
Wilson, K. 2013. Temporal and spatial variation in home range size for two woodland caribou ecotypes in Ontario. Master’s Thesis Trent University, Peterborough, ON.
Wilson, S.F., C. Pasztor, and S. Dickinson. 2010. Projected boreal caribou habitat conditions and range populations for future management options in British Columbia, Prepared for Ministry Energy, Mines and Petroleum Resources, and Ministry of Environment, Prince George, BC. 11 p.
Wittmer, H., A. Sinclair, and B. McLellan. 2005. The role of predation in the decline and extirpation of woodland caribou. Oecologia 144:257-267.
Whittington, J., (and 6 co-authors). 2011. Caribou encounters with wolves increase near roads and trails: a time-to-event approach. Journal of Applied Ecology 48: 1535-1542.
Yannic, G., (and 20 co-authors). 2013. Genetic diversity in caribou linked to past and future climate change. Nature Climate Change DOI 10.1038/nclimate2074.
Biographical Summary of Report Writers
Serge Couturier received his BSc in Biology at Université du Québec à Rimouski, and his MSc and PhD in Biology at Université Laval. His PhD dissertation focused on the ecology of Caribou from three ecotypes. In 1985, he joined the Québec government, first in the Northern Québec Regional office, and later in the Wildlife Research Department with an emphasis on monitoring of Caribou populations. Since 2012, he has worked as a consultant on projects dealing with Caribou. Serge is an Associate Professor at Université Laval and has co-authored many scientific papers and reports focusing on Caribou.
Liv S. Vors received her BSc from the University of Saskatchewan, and MSc at Trent University on the relationship between anthropogenic landscape change and Boreal Caribou habitat occupancy in Ontario. She received a PhD from the University of Alberta in 2013 studying the population ecology of migratory and Boreal Caribou. She has additionally worked as an independent consultant on Caribou for Environment Canada, the Environmental Commissioner of Ontario, Yukon College, and other institutions.
Issac Hébert received his B. Agri and Env. Sc. at McGill University and MSc at Concordia University on the spatial structure and habitat selection of Newfoundland Caribou.
Collections Examined
No collection was examined during the preparation of this status report.