Gaspé shrew (Sorex gaspensis) COSEWIC assessment and status report: chapter 3
Species Information
Name and classification
Sorex gaspensis and S. dispar belong to the order Insectivora and the family Soricidae (subfamily Soricinae, tribe Soricini). The genus Sorex contains 40-50 species and occurs throughout the Holarctic. The Gaspé shrew S. gaspensis Anthony and Goodwin (French, la Musaraigne de Gaspé) belongs in the subgenus Otisorex. S. gaspensis and the long-tailed or rock shrew S. dispar Batchelder (French, la Musaraigne longicaude) are allospecies comprising the S. dispar group. Two subspecies of S. dispar are currently recognized, S. d. dispar Batchelder and S. d. blitchi Schwartz. S. gaspensis is monotypic (Kirkland and Van Deusen 1979).
To dateS. dispar and S. gaspensis have been recognized as different species primarily because of differences in body size. There is a south to north cline of decreasing size in S. dispar, with the southern Appalachian subspecies S. d. blitchi being the largest (Kirkland and Van Deusen 1979). Extrapolating this cline to Quebec, Kirkland and Van Deusen (1979) predicted that if S. gaspensis belonged to thedispar cline, then S. gaspensis should be larger than it is. In fact S. gaspensis is slightly smaller than S. dispar. This discontinuity in body size, and the observed disjunction in the range of the two species, has supported the view that S. gaspensis is a distinct species (Kirkland and Van Deusen 1979). Their conclusions were confirmed by French and Kirkland (1983) and Scott and van Zyll de Jong (1989).
However, similarity in skull shape between S. disparand S. gaspensis challenge the notion that the two are distinct species. Furthermore, a hypothesized steeper cline at the northern edge of the range coinciding with a harsher climate also is suggestive of one species. Small numbers of specimens for analyses from the northern edge of the cline (Scott and van Zyll de Jong 1989; Rhymer et al. 2004) leave the question open.
The following paragraph summarizes recent genetic work and was written by Don Stewart (pers. comm.). Two studies of mitochondrial DNA variation have now been conducted on the S. dispar/S. gaspensis species complex. Recently, Rhymer et al. (2004) used partial mitochondrial control region sequence (284 bp). Don Stewart, Fred Scott and Stephen Petersen (in preparation) used partial mitochondrial cytochrome b sequence (309 bp) amplified from DNA extracted from small (~1x5 mm) pieces of dried skin samples. In addition to having analyzed different mitochondrial genes, the two studies differ in the geographic areas sampled. Rhymer et al. (2004) examined 35 specimens from northwestern Maine, southern Quebec, and Quebec's Gaspé Peninsula. Stewart et al. (in prep) examined specimens of S. dispar from Tennessee, North Carolina, New York, and Atlantic Canada and specimens of S. gaspensisfrom New Brunswick and Cape Breton Island, Nova Scotia. Rhymeret al. (2004) found little polymorphism and no differentiation between the two species. Stewart et al. (in prep) found two clades present among the sample of 18 individuals. One clade consisted of S. dispar specimens from Tennessee, North Carolina, and New York State. The other clade consisted primarily of S. gaspensis specimens but also included three S. dispar specimens from Nova Scotia. There was very little sequence variation in each of these two clades. The average sequence divergence between the two clades was also quite small (~1%), which may be consistent with conspecific status, as suggested by Rhymer et al. (2004).
The amount of divergence noted for this region of cyt b (309 bp) is less than that observed between several other closely related species of shrews (i.e., 3.2% divergence between S. palustris and S. bendirii, 5.7% between S. cinereus and S. haydeni, and 3.9% between S. hoyi and S. thompsoni, two putative pygmy shrew species). At present, the taxonomic status of S. disparand S. gaspensis must be viewed as uncertain. Although Rhymer's work clearly indicated that the two species were indistinguishable, the geographic range of specimens examined was very small (northwestern Maine and the Gaspé region of Quebec). The current work of Stewart et al. (in prep), which includes specimens from the southern portion of the range (Tennessee, North Carolina) and the northeastern portion of the range (mainland Nova Scotia and Cape Breton), suggests that there may be some taxonomically significant divisions within this complex. However, given the amount of cytochrome b sequence divergence, it could be argued that the two clades represent different subspecies rather than different species.
Taken together, the two studies suggest that the geographic boundaries of these two putative species or subspecies (i.e., S. dispar and S. gaspensis or S. dispar dispar and S. dispar gaspensis) will likely have to be re-drawn. It certainly appears unlikely thatS. gaspensis (or S. dispar gaspensis) will continue to be recognized as a Canadian endemic taxon. Further work on this species complex needs to be done focusing on (1) broader sampling of the range of S. dispar in New England, particularly in areas where the two mitochondrial DNA lineages identified by Stewart et al. (in prep) are likely to come into contact (i.e., Vermont, New Hampshire, southern Maine); (2) sequencing of additional mitochondrial DNA gene regions (e.g., the complete cytochrome b gene); and (3) the addition of nuclear DNA markers (e.g., microsatellites)’’.
Morphological description
Both S. dispar and S. gaspensis are small, slender and slate-grey in colour throughout the year (usually on both dorsal and ventral surfaces). The tail is long, well furred and not distinctly bicoloured (Kirkland 1981). S. gaspensis weighs approximately 3 g (range 2.2-4.3 g) and measures 95-127 mm in total body length, with a tail length of 45-55 mm. S. dispar is larger in all respects (3.1-8.3 g, total length 103-136 mm, tail length 46-67 mm). Morphologically, skulls of the two species can be separated based on condylobasal length and the length of the molariform tooth row. In S. gaspensis the condylobasal length is < 16.4 mm (range 15.35-16.35 mm) and the molariform tooth row is < 3.7 mm (range 3.40-3.65 mm), and in S. dispar these features are larger (condylobasal length 16.45-18.70 mm, molariform tooth row 3.75-4.30 mm, Kirkland and Van Deusen 1979, Table 1).
| Measurements | Sorex dispar | Sorex gaspensis | t-test |
|---|---|---|---|
| Total length | 120.26 ± 4.24 (n=5) | 105 ± 8.78 (n=21) |
3.74
|
| Tail length | 57.68 ± 1.85 (n=5) | 49.72 ± 2.65 (n=23) |
6.34
|
| Hind foot length | 13.08 ± 0.78 (n=5) | 11.98 ± 0.57 (n=23) |
3.67
|
| Mass | 4.08 ± 0.37 (n=5) | 2.89 ± 0.63 (n=9) |
3.83
|
| Condylobasal length | 17.12 ± 0.33 (n=33) | 15.88 ± 0.37 (n=18) |
12.29
|
| Length of molariform tooth row | 3.92 ± 0.12 (n=49) | 3.56 ± 0.07 (n=18) |
3.74
|
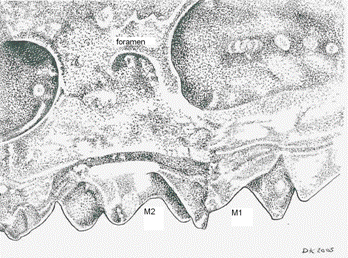
Externally, their dorsal pelage, slender build, unusually long snout and long tail serve to separate S. dispar and S. gaspensis from other sympatric soricids. The smoky shrew (S. fumeus) is chunky and robust, with an incrassate (swollen in mid-length) tail not found in S.dispar or S. gaspensis. As well, tail length of the smoky shrew generally averages 60-70% of head-body length, whereas in both S. dispar and S. gaspensis it is usually larger, averaging 80-90% of head-body length (Kirkland 1981). Both S. dispar and S. gaspensis can be distinguished from other North AmericanSorex based on the position of the posterior border of the intraorbital foramen (posterior to the plane between M1 and M2) and the rostra (which are longer than other Otisorex; see Fig. 1).
Figure 1. Distinguishing features of skull in S. gaspensis (similar to S. dispar) compared to other Otisorex, specifically the position of the intraorbital foramen in relation to M1 and M2, and the rostra (from photograph in Lupien 2001).