Peary caribou and barren-ground caribou COSEWIC assessment and status report: chapter 10
Limiting Factors and Threats
Industrial activities
Miller (1991) concluded that industrial activities have the potential to reduce or prevent access to important habitats, and some Inuit believe that this has been the case at least in some situations (Freeman 1975). However, a study of interactions between seismic operations and ungulate behaviour (F.F. Slaney & Co. Ltd. 1975a, b) found that:
- 15-20 caribou were in the vicinity of seismic vehicles during the mid-winter portion of the study and some of them were seen repeatedly.
- Caribou continued foraging when seismic shots were detonated 3.5 km distant and remained in the vicinity for at least several hours.
- Caribou ceased foraging and walked away when approached by seismic vehicles closer than 0.8 km. Individuals remained for more than 1 day within about 4.0 km sight or sound of seismic activity.
- Caribou moving through the area by-passed the seismic camp within 0.8 km without altering their direction of movement.
- Caribou showed neither preference nor avoidance of seismic lines, which became partially obscured by blowing snow within a few hours.
While some displacement from foraging areas and increased energy expenditure occurred, these were within the ranges of normal activities such as predator avoidance; more intensive disturbance in areas of ungulate concentrations during critical periods, such as late winter and calving times, could be harmful (F.F. Slaney & Co. Ltd. 1975a, b). These recorded observations support Amagoalik’s (in Freeman 1975) opinion that seismic activities can disrupt the activities of caribou, but not to the extent that disturbance would cause caribou to abandon the island.
Miller and Gunn (1978, 1980) noted the potential threat of ship traffic interrupting inter-island caribou movements, which are necessary, at some times, both to escape unfavourable environmental conditions and to repopulate previously abandoned ranges. Inuit and biologists, at a Peary caribou population and habitat workshop in Yellowknife, echoed these concerns in regard to migration of the Dolphin and Union herd (Gunn et al. 1998). More recently, hunters have voiced similar concerns at meetings of the Kitikmeot Hunters and Trappers Association in regard to shipping traffic associated with the proposed Bathurst Road and Port Development (M. Wheatley, Nunavut Wildlife Management Board, pers. comm. June 30, 2003).
Interspecific competition
Some Inuvialuit of Victoria Island do not believe that competition with muskoxen is a factor in caribou declines, because of different food preferences and different feeding areas (Elias 1993). Residents of Resolute Bay also doubt that there is direct competition, noting that the 2 species do not feed together (Nunavut Tusaavut Inc. 1997). Parker (1978) suggested, based on rumen and fecal samples of both species collected after the 1973-1974 crash in the Western Queen Elizabeth Islands, that, while there was some overlap in forage species, there was no interspecific competition. He found that in severe winters, however, both ungulates sought out willows on exposed slopes or ridges and suggested that when densities are high, they may compete for forage. In contrast, Thomas et al. (1999) found almost no dietary overlap and different use of micro-habitats (caribou used moist and dry sites while muskox preferred wet meadows), and concluded that, at low densities, competition was not a factor. Miller’s (1991) review supported the Aboriginal traditional knowledge on this point, stating that caribou and muskoxen use somewhat different habitats, prefer different foods, and segregate their ranges, although there is overlap on all 3 points. In the western Queen Elizabeth Islands, neither the amount or quality of the absolute forage supply nor competition with other grazers made any significant contribution to the documented declines of Peary caribou (Miller and Gunn 2003a).
On Banks Island, the caribou population decreased from about 12 000 in 1972 to about 700 in 1995, while muskox populations increased from about 4000 to about 65 000 (Larter and Nagy 1997). Several studies (Wilkinson and Shank 1974, Wilkinson et al. 1976, Shank et al. 1978, Oakes et al. 1992, Mulder and Harmsen 1995, Smith 1996) examined this issue and found that while overlap of feeding areas and forage species occurred, there was no compelling evidence of large-scale impact of muskoxen on forage conditions that could affect caribou. Larter and Nagy (1997) found that caribou and muskoxen had considerable similarity in their diets, and diet similarity was more pronounced in areas of high muskox densities. Larter and Nagy (1997) also found that deep snow caused muskoxen to crater for forage on the same slopes where caribou foraged and suggested that muskox browsing on willows in such situations could reduce its availability for caribou. In recent years, however, the caribou population trend reversed and began to rise at a high annual rate, while muskox populations have also continued to increase. Consequently, in 3 decades of study, detrimental effects of high muskox densities on caribou have not been demonstrated.
On Prince of Wales and Somerset islands, Gunn and Dragon (1998) found that while the caribou declined, muskoxen increased from about 1155 in 1980 to 6399 in 1995 (Figure 14). (Confidence intervals can not be given because they were calculated separately for Prince of Wales and Somerset surveys; for the Somerset population only 29 muskoxen were seen and no estimate was calculated, so for the graph, 29 was added to the Prince of Wales population for 1980.) The trend lines are based on the exponential model (Appendix 1), but with only 2 estimates at the start and end, sudden changes are equally plausible.
Figure 14. Prince of Wales-Somerset caribou and muskox trends, 1980-1995 (data from Gunn and Dragon 1998).
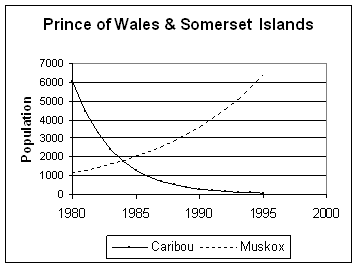
Gunn and Dragon (1998), reviewing the possible causes of the caribou decline on Prince of Wales and Somerset islands, made the following points about the possibility of competition with muskoxen having affected the caribou adversely:
- There has been a long-standing controversy about the relationship between muskoxen and caribou, particularly among the Banks and Victoria islands Inuvialuit who hunt them and the biologists who study them.
- Muskox-caribou relationships operate at different scales of space and time; on an evolutionary time scale they have long coexisted, but also have evolved in more complex communities of large herbivores and predators.
- In the Arctic Archipelago, their population growth and decline trajectories are spatially inconsistent: mostly synchronous in the western Queen Elizabeth Islands and south central Victoria Island and asynchronous on Banks Island, northwest Victoria Island, and in the Prince of Wales-Somerset islands complex. There are region-wide climatic, terrain and vegetative differences that may contribute to these differences (Banks Island being the only well-vegetated Arctic island).
- Previous studies of muskox-caribou relationships did not specifically address competition. They compared diet or habitat use, but have not addressed behavioural exclusion (or avoidance), although Inuit have often stated that caribou avoid muskoxen or muskox foraging areas and suggested several reasons why this is so.
- The studies of distribution and diet that point to little overlap between caribou and muskox did not deal with high muskox densities or deep snow foraging conditions (e.g. Larter and Nagy 1997). Russell et al. (1978 cited in Gunn and Dragon 1998), however, documented little overlap between caribou and muskox seasonal ranges based on distribution of fecal pellets, except on Prince of Wales Island where both herbivores fed in summer on willow-moss-lichen patterned ground, 1 of the most common plant communities.
- Dietary overlap, in itself, is not evidence of competition.
- The time scales of the effects of competition vary. For example, browsing at different seasons can either damage shrubs or stimulate compensatory growth.
The above evidence suggests that in most circumstances there is no substantial competition between muskoxen and caribou for space or forage, but more investigation is needed into the space and time scales of potential competition (Ferguson 1996), as well as the mechanisms of competition (Gunn and Dragon 1998). The possibility remains that when muskoxen are at high densities, the caribou may be at a disadvantage, particularly in severe winters when forage for both species is limited by snow and ice. When muskoxen are at high densities, correspondingly higher wolf populations may take enough caribou to depress their numbers and either exacerbate a decline or dampen a recovery.
Predation
Arctic grey wolves (Canis lupus arctos) are present throughout Peary caribou and barren-ground caribou range in the Arctic Archipelago (Miller 1992, Miller and Reintjes 1995, Larter and Nagy 1998b). Wolves feed on caribou and muskoxen approximately according to their availability when both prey species are present (Gunn et al. 2000b).
Inuvialuit of Victoria Island do not believe that wolf predation is a significant factor in caribou declines on northwest Victoria Island (Gunn 1993). Wolf predation may have been a factor in the 1993-1994 caribou decline because of the high density of wolf dens and large wolf packs (up to 20 wolves per pack) adjacent to the traditional caribou calving and summering grounds on Banks Island (Larter and Nagy 2000b).
Miller and Reintjes (1995), based on 373 reports involving 1203 wolf sightings from 1967 to 1991, documented observations of at least 572 different wolves in 118 separate packs and 116 single wolves throughout the Arctic Archipelago. Most (81%) of the wolves were seen on the Queen Elizabeth Islands, but not all population areas were sampled equally. Pack sizes averaged 4.8±0.28 and ranged from 2 to 15 wolves per pack. Pups averaged 3.9±2.24 SD (range 1-10) per pack. On a decadal scale, the frequency of wolf sightings increased and decreased with the populations of Peary caribou and muskoxen in the Queen Elizabeth Islands. (This association did not hold up, however, in the “southern tier” of islands, most likely due to the relatively small data sets from those regions (F.L. Miller, pers comm. May 21, 2003).
Miller (1998) concluded that if the wolf population responded to the increasing prey base (caribou and muskoxen) in the Bathurst Island complex as expected, then there would have been about 45 wolves there when the populations crashed during 1995-1997. That many wolves would pose “a serious threat to…the remnant caribou and muskox populations” Miller (1998 p. 55). Gunn et al. (2000b) agreed that wolf predation could accelerate a decline.
Hunting
The southern tier of islands and parts of the eastern High Arctic were more or less continuously occupied by caribou hunting pre-Dorset, Dorset, and Thule cultures from about 4500 ybp onwards (McGhee 1976, 1997). Although the Melville Island complex and the northwestern portions of the Bathurst Island complex were beyond the margins of consistent Inuit occupation, the other parts of the Queen Elizabeth Islands had long periods of occupation interspersed with periods of abandoned or limited use, so that hunting pressure probably ranged from moderate to non-existent in those areas during the latter half of the Holocene. The western Queen Elizabeth Islands were unpopulated prior to 1953 when a community of Inuit was established at Resolute Bay.
Although hunting has not been implicated as a causative factor in any of the die-offs (Gunn et al. 2000b), it may have contributed to long-term caribou decline in several cases. The demise, between about 1900 and 1920, of the Dolphin and Union herd, was thought due to hunting following the introduction of firearms (Anderson 1934, Manning 1960). On Banks Island, throughout the 1980s, harvest was female-selective, which likely skewed the population towards males and younger animals (Larter and Nagy 2000b). After 1991, the caribou population on Banks Island continued to decline until 1998 despite severely restricted hunting by Sachs Harbour residents (Larter and Nagy 2000b). More recently, the population increased significantly as noted previously.
Hunting may also have been a factor in the decline of caribou on Victoria Island (both Peary and Dolphin and Union populations), and is of concern on the Boothia Peninsula (Gunn et al.1998).
The harvest of the Dolphin and Union herd is relatively high compared to the 1997 estimated size of the herd. As the herd migrates close to Cambridge Bay and Holman in the fall and over-winters on the mainland, increased access by hunters from other communities including Kugluktuk, Umingmaktok, and Bathurst Inlet contribute to the total harvest. In addition to the subsistence harvest, the herd provides the opportunity for guided hunts when they mass along the north coast of Dolphin and Union Strait prior to fall migration. Currently, 50 tags are allotted (Mathieu Dumond, Government of Nunavut, pers. comm. February 9, 2004).The extrapolated harvest is 2000-3000 caribou (based on the reported harvest from the Kitikmeot Harvest Study (Gunn et al. 1986, Nishi and Gunn 2004).
The Dolphin and Union herd is also hunted commercially. Irregularly, a few (currently 30 for Kagluktuk and 40 for Cambridge Bay) commercial caribou tags are issued for the sale of meat. Although the commercial harvest is intended for barren-ground caribou, the hunt occurs in winter when the Dolphin and Union caribou are on the mainland and they account for a large proportion of the harvest. Probably fewer than 100 Dolphin and Union caribou are harvested in these hunts (Mathieu Dumond, Government of Nunavut, pers. comm. February 9, 2004).
Inuit hunters, through their local associations, have voluntarily curtailed hunting of Peary caribou throughout the period of investigation into caribou population declines from 1974 to present (Miller and Gunn 1978, Ferguson 1987, Larter and Nagy 1995, 2000b, Ferguson et al. 2001). For example, from 1974 to 1989, the Resolute Bay Hunters and Trappers Organization prohibited hunting of the Bathurst Island local population (the Melville-Prince Patrick herd is too far for access by snow machine). From 1989 to 1996, as the caribou population increased in the Bathurst complex, it allowed limited harvesting in consultation with government biologists. After the 1994-97 die-off, however, it again curtailed the harvest. Inuit hunters from Grise Fiord halted caribou hunting on most of southern Ellesmere Island from 1986 to 1996 while caribou numbers were low, and then began again in response to the increasing caribou population. At times of unavailability of local (Bathurst) caribou, the Resolute Bay hunters made arrangements with Inuit groups at Grise Fiord and Cambridge Bay to hunt in their territories, but this was not a satisfactory solution for several reasons (T. Mullen pers. comm. March 20, 2002; N. Amarualik Resolut Bay Hunters and Trappers Association, pers. comm. March 21, 2002). From these observations it seems clear that excessive hunting can at least exacerbate, if not cause, declines, and would also dampen recovery rates. Peary caribou are under no serious threat from hunting as long as local community associations and biologists continue to cooperate on conservation.
Freezing fall rain and snow/icing events
Weather events that result in a hard glaze of ice forming on the ground, or as a crust or in layers in the snow, are implicated in a number of the major die-offs of Peary caribou. The mechanism is warming in spring or fall when there is already snow on the ground, which then melts or absorbs rain, followed by re-freezing to form a hard crust in the snow or a glaze covering the ground.
These situations contrast with the more usual condition in which temperatures from the first snowfall onward throughout winter remain below freezing and the snow remains powdery. Normally, the wind erodes snow away from topographic elevations and packs it into topographic depressions and the lee sides of geographic features. Both caribou and muskoxen feed on these exposed, snow-free terrain features, but muskoxen have more of an ability to dig craters in the snow and make more use of mesic sites, while caribou make more use of the xeric sites. A hard crust or glaze disadvantages both species; this is why muskoxen and caribou populations decline more or less in synchrony during these events in the areas where they occur, and not in other areas.
Measures of winter severity such as temperature and snowfall are difficult to associate with events such as winter die-offs, but several authors (Miller 1998, Larter and Nagy 2000b, Gunn and Dragon 2002) have found snowfall to be the best predictor.
Miller and Gunn (2003b) summarized the evidence as follows:
“We know of four major die-offs and associated subsequent major to near total calf crop losses plus one additional major calf crop reduction on, at least, the WQEI: 1973/74, 1989/90, 1994/95, 1995/96, and 1996/97…. All 5 of those winters and springs experienced significantly greater (p < 0.005) than average total snowfall between 1 September-21 June of each year and ranked at the top of 50 years for which weather records exist at Resolute, Cornwallis Island (Miller, 1998: 1st, 1995/96; 2nd, 1994/95; 3rd, 1996/97; 4th, 1989/90; and 5th, 1973/74). It is more the extent and characteristics of the snow cover than snow depth per se which result from heavy snowfall and high winds that cause widespread or in the few worst years range-wide prolonged and extreme relative unavailability of forage. This condition is especially compounded when associated with extensive icing that will ‘lock in’ the forage even further and prevent the animals from obtaining an adequate supply of food. However, so far, there has been a direct correlation between total snowfall and die-offs, particularly when heavy snowfall occurs in early winter. Therefore, total snowfall is the best indicator that we have to date of the potential for an extremely severe ‘weather-year’ causing die-offs and calf crop failures. The timing, duration, types, and amounts of icing compound the impact of deep snow and tends to cloud the relative importance of the role of deep snow vs. icing in these drastic die-off years.”
They further noted that the die-offs occurred in the western Queen Elizabeth Islands when the caribou were at low mean overall densities, implying that density-dependent mechanisms such as overgrazing were not involved (Miller and Gunn 2003b):
- 1973-74, 68% reduction at a starting density of 0.04 caribou ·km-2 (19 199 km²) and a 46% decline at 0.7 caribou ·km-2 (61 237 km²);
- 1994-95, 30% decrease at 0.16 caribou ·km-2(19 199 km²);
- 1995-96, 75% drop at 0.11 caribou ·km-2 (1 199 km²); and
- 1996-97, 83% loss at 0.03 caribou ·km-2 (19 199 km²).
All 4 of the above Peary caribou die-offs also involved similar high annual rates and timing of deaths among muskoxen (Miller and Gunn 2003b), supporting snow/ice conditions as the causative factor.
Snowfall and snow depth records and notes on their relationship to caribou die-off years are given in Appendix 2. Neither total annual snowfall (Appendix 2, Figure 19) nor total monthly snowfall during September-May (Appendix 2, Figure 20) are exclusively associated with caribou die-off years, but snow depth at month end (Appendix 2, Figure 21) is. Interestingly, snow depth at month end is not correlated with total monthly snowfall. This suggests that snow depth may be a better predictor of die-off years than snowfall (Appendix 2, Figure 22).
The Banks Island-Victoria Island region is in a different climate region from the Queen Elizabeth Islands, with warmer annual mean temperatures and higher precipitation (Maxwell 1980, MacIver and Isaac 1989). Larter and Nagy (2000b) reported that severe winters with heavy snow caused caribou die-offs in 1987-1988, 1988-1989, and 1990-1991. A severe winter with deep, hard snow and freezing rain in autumn also occurred in 1993-1994. The freezing rain effectively eliminated approximately 50% of the available range and the local Inuvialuit voiced concern in November 1993 that the caribou were undernourished (Larter and Nagy, 1995). Although overwinter growth occurred in caribou calves and there was no die-off, during that winter (Larter and Nagy 1995), subsequent analysis showed that in a 5-year period, calf production was highest following the winter with the shallowest snow, and lowest following the winter with the hardest snow (Larter and Nagy 2000b).
To evaluate other possible explanations for caribou scarcity during those years, Miller (1998) and Gunn and Dragon (2002) counted caribou and muskox carcasses along with their population surveys in the Bathurst complex. In June 1995, Miller (1998) counted 1984 live and 56 dead caribou in intensive but unsystematic (hence, no population estimate) searches and estimated that 25% to 30% of the 3000+ Peary caribou there in 1994 had died. However, Resolute Bay hunters reported that significant numbers of caribou moved that fall from Bathurst Island to Cornwallis, where they killed between 50 and 100. In July 1996, Miller (1998) estimated from 143 caribou carcasses counted on transect, that 1143±164 had died the previous winter. He saw no calves that summer, and estimated 452±108 living caribou. This left about 400 caribou unaccounted for by deaths, suggesting that some had also emigrated during 1995-1996, which was confirmed by (a) reports by hunters of more caribou appearing on Cornwallis Island that year, and (b) a movement of a satellite-collared cow leaving from Bathurst to Borden Island, where it died (Miller 1997b, 1998). Miller (1998) estimated, from interviews with a number of Inuit hunters, that 85±25 of those 400 caribou were shot on Cornwallis Island that winter, leaving about 315 unaccounted for.
In 1997 Gunn and Dragon (2002) estimated 78±26 live caribou and 408±53 carcasses in the Bathurst Island complex, suggesting that most of the caribou alive there in 1996 had died by the following spring. They also found that death rates varied by island complex and by island based on the ratio of dead to living caribou in 1997. The Bathurst Island complex had a higher death rate (83%) than the Melville Island complex (30%). Their tabulation of caribou group size further supported the hypothesis that the declines were due to deaths and not emigrations (because when emigrating, usually a herd or family group goes together): the mean group size for the Melville, Prince Patrick and Bathurst groups was 4.3±0.34, compared to 6.7±0.36 in 1974, another die-off year. Although they could not fully discriminate among deaths, reproductive failure or emigrations as the cause of the 1996-1997 decline in the Melville-Prince Patrick complex, a 30% decline was due directly to deaths.
In summary, severe winters characterized by deep snow and icing resulting from rain or unseasonably warm weather, either in fall or spring, have been the cause of the best documented, sudden population declines, notably those that occurred in the western Queen Elizabeth Islands during 1973-1974 and again during 1995-1997. Other declines, including those of the Banks Island-northwestern Victoria Island and Prince of Wales-Somerset Island populations, have been more gradual and without a clear, single cause. In those latter cases, weather played a role in some cases (e.g., Banks Island) and in other cases may have, but the evidence is lacking. Contributing factors in these latter cases included excessive hunting (Banks Island, northwestern Victoria Island, Prince of Wales-Somerset and Boothia Peninsula) and may also have included summer weather effects on plant growth (Prince of Wales-Somerset) or interaction with muskoxen through some as yet undetermined mechanism, possibly related to either predator-prey interactions or competition for space or forage, or both.
Climate change
The susceptibility of Peary caribou to population-wide decimation caused by weather events has led to speculation that climate change has caused the observed 3-decade overall decline in the subspecies (Miller 1991, Ferguson 1996, Gunn 1998b, Miller and Gunn 2003b). Gunn (2003) noted that,
“The current patterns of morphological and genetic variation are also a consequence of recent as well as past conditions. The North American Arctic and subarctic climate is strongly regionalized with east-west and north-south gradients in variables such as snow depth which is related to probabilities of incursions of Pacific and/or Atlantic maritime air masses (Maxwell 1981). Annual and seasonal variability is high and unpredictable. Consequently, caribou ranges especially on the Arctic islands are a non–equilibrium ecological system with environmental extremes causing unpredictable declines in relative forage supplies (Caughley and Gunn 1993, Behinke 2000).”
The notion of caribou existing in a non-equilibrium situation with respect to their range conditions explains their extreme susceptibility to climatic factors that may alter their food supplies or access to them. Brown and Alt (2001) documented extensive high latitude warming in the Northern Hemisphere in recent decades, consistent with predictions of greenhouse gas-forced global warming (Figure 15). These data confirm reports by Inuit from the Queen Elizabeth Islands of warming and associated ecological changes (Nunavut Tusaavut Inc. 1997). Much of the warming and reduction in snow and sea ice has occurred during the 1980s and 1990s, and 1998 was conspicuous for the unprecedented warmth and length of the melt season over the Canadian Arctic. Accompanying the 1998 weather anomalies, Brown and Alt (2001) found changes in:
- Snow melt: The 1998 melt season was characterized by an initial early retreat of the spring snow-line over the entire North American continent in April and May. Early spring warming coupled with below-average winter snow accumulation over western Canada and the Mackenzie Basin, led to a record (1955-2000) early melt of snow over the western Arctic.
- Break-up: In the west, break-up was early and there was twice as much open water in the southern Beaufort Sea as normal; distance to the ice edge was 46% greater than for the previous record year of 1954. Minimum ice extent conditions extended into the Queen Elizabeth Islands where the latest date for minimum ice extent in the 40 years was recorded.
- Active layer: In the west, permafrost temperatures and thaw penetration were generally the highest in the 10-year record and there was evidence of ground ice wedge melt and increased active layer detachment slide activity.
They also found that 1998 was the warmest year in the instrumental record for the Canadian Arctic (since ~1950 for the High Arctic climate station network), and was also the warmest in the instrumental record for the Northern Hemisphere land area (since ~1850). Other years have experienced similar cryospheric conditions to 1998, notably 1962.
Current Environment Canada global warming projections from the Canadian General Circulation Model (CCGM) (Environment Canada 2002) show that both maximum and minimum winter temperatures will rise more than summer temperatures. At Eureka, for example, the maximum extreme temperatures during 1961–1990 remained below freezing from November through April (Figure 16), but with global warming, temperature extremes of 0° C or above may occur in every month except March (Figure 17). (Extreme temperatures are important because although these events are rare, it only takes 1 event to cause the thawing and re-freezing that is associated with caribou die-offs). This would increase the frequency of winter rain and consequent snow/icing events.
Such dramatic climate changes portend equally profound ecological changes that could affect caribou. Effects could include:
- Warmer winter weather could reduce caribou energy expenditure, but is also likely to increase the frequency of rain and consequent snow/icing events, a known cause of starvation and population declines.
- Warmer summer weather could lead to greater plant growth, a benefit to caribou. It could, however, be a disadvantage to Peary caribou if it and other weather-related ecosystem changes allowed barren-ground caribou to invade the Arctic Archipelago. Warmer spring weather and earlier snowmelt can hasten plant green-up and flowering phenology. This may benefit caribou during late 3rd trimester gestation in spring. However, disruption of the synchrony between plant phenology and Peary caribou life cycle could be detrimental (Gunn 1998b)
- Warmer and wetter summer weather could lead to greater exposure to diseases and parasites, which have not previously been implicated in population declines (Miller and Gunn 2003b). Inuit from the Queen Elizabeth Islands report that mosquitoes and black flies have increased along with warmer weather in certain areas (Nunavut Tusaavut Inc. 1997).
Figure 15. 1998 Departures from normal in selected cryosphere parameters. The thin verticle line highlights 1998 (from Brown and Alt 2001).
Figure 16. Mean monthly temperature at Eureka, 1961 to 1990 (Environment Canada 2002).
Figure 17. Mean monthly temperature projections for 2040-2069 at Eureka based on General Circulation Model (Environment Canada 2002).

- A greater extent or longer periods of open water in summer, combined with higher evaporation rates, could increase cloud cover, limit solar radiation and reduce plant growth.
- Increased soil temperatures and greater depth of the active soil layer above the permafrost could increase plant productivity with benefits to caribou. Also soil subsidence caused by ice lens melting could increase the availability of seed beds for some plant species, such as grasses and forbs, resulting in changes in plant community structure. Shrub dominance could also move northward with an increased duration of warmer >5 °C) temperatures.
- Changes in the timing of freeze-up and break-up of sea-ice and a reduction in the extent of sea-ice will disrupt caribou inter-island and island-mainland migrations. Caribou deaths may increase if caribou cross on thin ice.
- In the eastern Queen Elizabeth Islands, Peary caribou could subsist at higher elevation than those currently used if glaciers were to retreat.
In experiments on the effect of global warming on Ellesmere Island, Greg Henry (University of British Columbia, pers. comm., November 20, 2002; unpubl. data) maintained a long-term warming experiment at a site on Ellesmere Island since 1992. He increased summer growing season temperatures by 1°C to 3°C in open-top chambers made of greenhouse grade fibreglass. The higher temperatures were associated with a change in species composition and abundance in all 7 of the tundra plant communities examined. There was an increase in the abundance of non-woody (herbaceous) plants (graminoids and forbs) in all sites and an increase in the only deciduous shrub (Salix arctica) in those communities where it dominates. The evergreen shrubs did not show any strong response. He concluded that in terms of Peary caribou forage, climate warming should increase some caribou food species and cause earlier flowering of those species.
If global warming were to prove favourable to barren-ground caribou, their expanding range northward could put Peary caribou at a competitive disadvantage. For example, in the Yukon, some lichens could expand their range northward and this could favour barren-ground caribou, which can prosper on lichens (Harding and McCullum 1997). How Peary caribou, which include little lichen in their diet because of the paucity of lichens on their range, would adapt to a new vegetation profile (species assemblage, forage biomass and phenology) compared to barren-ground is unknown. Miller (1991) suggested that Peary caribou would not compete well with barren-ground caribou.
Unfortunately, the hypothesis of climate change impacts on Peary caribou has not been tested. To do so would require a detailed analysis of weather records focusing on the specific variables that may change (or that have changed) with greenhouse-induced global warming in relation to Peary caribou demographics and associated ecological conditions over the period of record. This should be a priority, because if global warming increases the frequency or severity of spring and fall icing events, Peary caribou could go extinct within a relatively few years. Conversely, if climate turns out not to be implicated, then it becomes urgent to develop and test alternative hypotheses to explain the observed population declines.
Genetic diversity
Mitochondrial DNA shows that Peary caribou have low genetic diversity, the Prince of Wales population having a very low index of gene diversity (0.5714, compared to 0.791 to 1.000 for other North American caribou populations, where 1.0 means that all haplotypes in the sample are different) (Eger et al.1999).
Zittlau (2003), using microsatellite analyses of nuclear DNA, which reflects a shorter evolutionary scale, found considerable genetic variation within the Peary and Dolphin and Union populations, sufficient to recognize distinct genotypes. However, the Canadian Arctic islands samples were significantly less variable than mainland barren-ground and woodland caribou populations. The Bathurst Island complex, Melville Island complex, and Prince of Wales-Somerset populations had the lowest levels of genetic diversity. Each population of Arctic islands caribou that they sampled had experienced recent genetic bottlenecks, raising concerns of increased risk of inbreeding depression as well as a loss of adaptive potential. Zittlau (2003) concluded that,
“The Arctic Island caribou populations have already experienced a number of declines and may not be able to adapt to changes in their environment if their genetic diversity is not preserved.”
Parasites and diseases
Few parasites and diseases have been found in Peary caribou and they are not thought to be a serious conservation issue (Miller 1991, Gunn 1993, Gunn and Dragon 1998, Larter and Nagy 1998d, Miller and Gunn 2003b).
Other threats
Dolphin and Union caribou, although relatively numerous are at risk, because of their behaviour of staging along the coast until freeze-up, of reduced forage availability, deaths from drowning through being too anxious to move across the ice, and a high susceptibility to hunting at that time (A. Gunn, Government of the Northwest Territories, pers. comm. June 12, 2003).