Recovery Strategy for the Northern Saw-whet Owl brooksi subspecies (Aegolius acadicus brooksi) in Canada [proposed] 2013
- PREFACE
- RECOMMENDATION AND APPROVAL STATEMENT
- ACKNOWLEDGEMENTS
- EXECUTIVE SUMMARY
- RECOVERY FEASIBILITY SUMMARY
- 1. COSEWICSpecies Assessment Information
- 2. Species Status Information
- 3. Species Information
- 4. Threats
- 5. Population and Distribution Objectives
- 6. Broad Strategies and General Approaches to Meet Objectives
- 7. Critical Habitat
- 8. Measuring Progress
- 9. Statement on Action Plans
- 10. References
- APPENDIX A: Effects on the Environment and Other Species
- APPENDIX B: Recovery Team Members
- LIST OF TABLES
- LIST OF FIGURES
- Figure 1. Range of Saw-whet Owl on Haida Gwaii and the Global distribution of Northern Saw-whet Owl (acadicus and brooksi subspecies).
- Figure 2. Critical habitat for Saw-whet Owl at Eli Tingley site on Haida Gwaii (Queen Charlotte Islands)
- Figure 3. Critical habitat Saw-whet Owl at Dat 1/5 (west polygon) and Dat ¾ (east polygon) sites on Haida Gwaii (Queen Charlotte Islands)
- Figure 4. Critical habitat for Saw-whet Owl at QC 11 km North site on Haida Gwaii (Queen Charlotte Islands)
- Figure 5. Critical habitat for Saw-whet Owl at Begbie site on Haida Gwaii (Queen Charlotte Islands)
- Figure 6. Critical habitat for Saw-whet Owl at Bragg site on Haida Gwaii (Queen Charlotte Islands)
- Figure 7. Critical habitat for Saw-whet Owl at QC 33 km North site on Haida Gwaii (Queen Charlotte Islands)
- Figure 8. Critical habitat for Saw-whet Owl at Ghost (west polygon) and QC 32 km South (east polygon) sites on Haida Gwaii (Queen Charlotte Islands)
- Figure 9. Critical habitat for Saw-whet Owl at Riley Creek site on Haida Gwaii (Queen Charlotte Islands)
- Figure 10. Critical habitat for Saw-whet Owl at 19 km Lake site on Haida Gwaii (Queen Charlotte Islands)
- Figure 11. Critical habitat for Saw-whet Owl at Hangover Creek site on Haida Gwaii.

For copies of the recovery strategy, or for additional information on species at risk, including COSEWICStatus Reports, residence descriptions, action plans, and other related recovery documents, please visit the Species at Risk Public Registry (http://www.sararegistry.gc.ca/).
Cover illustration: Matthew Rowe
Également disponible en français sous le titre
« Programme de rétablissement de la Petite Nyctale de la sous-espèce brooksi (Aegolius acadicus brooksi) au Canada [PROPOSITION] »
© Her Majesty the Queen in Right of Canada, represented by the Minister of the Environment, 2013. All rights reserved.
ISBN: ISBN no. to come
Catalogue no.: Catalogue no. to come
Content (excluding the illustrations) may be used without permission, with appropriate credit to the source.
The federal, provincial, and territorial government signatories under the Accord for the Protection of Species at Risk (1996) agreed to establish complementary legislation and programs that provide for effective protection of species at risk throughout Canada. Under the Species at Risk Act (S.C. 2002, c.29) (SARA), the federal competent ministers are responsible for the preparation of recovery strategies for listed Extirpated, Endangered, and Threatened species and are required to report on progress within five years.
The Minister of the Environment and the Minister responsible for the Parks Canada Agency are the competent ministers for the recovery of the Northern Saw-whet Owl brooksi subspecies and have prepared this strategy, as per section 37 of SARA. It has been prepared in cooperation with the Province of British Columbia and with input from the Northern Saw-whet Owl Recovery Team and Haida Nation's Forest Guardians. Nevertheless, the opinions and interpretations contained herein may or may not be shared by participating organizations and individuals.
Success in the recovery of this species depends on the commitment and cooperation of many different constituencies that will be involved in implementing the directions set out in this strategy and will not be achieved by the Parks Canada Agency, Environment Canada, or any other jurisdiction alone. All Canadians are invited to join in supporting and implementing this strategy for the benefit of the Northern Saw-whet Owl brooksisubspecies and Canadian society as a whole.
This recovery strategy will be followed by one or more action plans that will provide information on recovery measures to be taken by the Parks Canada Agency, Environment Canada and other jurisdictions and/or organizations involved in the conservation of the species. Implementation of this strategy is subject to appropriations, priorities, and budgetary constraints of the participating jurisdictions and organizations.
The Parks Canada Agency led the development of this federal recovery strategy, working together with the other competent minister(s) for this species under the Species at Risk Act. The Chief Executive Officer, upon recommendation of the relevant Park Superintendent(s) and Field Unit Superintendent(s), hereby approves this document indicating that Species at Risk Act requirements related to recovery strategy development (sections 37-42) have been fulfilled in accordance with the Act.
Recommended by:
Ernie Gladstone
Field Unit Superintendent, Gwaii Haanas National Park Reserve
Approved by:
Alan Latourette
Chief Executive Officer, Parks Canada
All competent ministers have approved posting of this recovery strategy on the Species at Risk Public Registry.
This recovery strategy was written by Carmen Holschuh of WRG Westland Resource Group Inc. and Ross Vennesland of the Parks Canada Agency. The strategy was greatly improved by technical input, advice and comments provided by the brooksi Northern Saw-whet Owl Recovery Team. In particular, Frank Doyle shared important climate and habitat related information from his recent analyses. Berry Wijdeven, the recovery team chair, kindly helped keep the development of this recovery strategy on track. Detailed reviews of parts of the recovery strategy were provided by Carita Bergman, Diane Casimir, Manon Dubé, Richard Pither, Lucy Reiss and Kara Vlasman. The recovery strategy was reviewed on behalf of the Province of British Columbia by Jeff Brown, Laura Darling, Stephanie Hazlitt and Leah Westereng.
The Northern Saw-whet Owl, brooksi subspecies, (hereafter 'Saw-whet Owl') is an endemic subspecies to Haida Gwaii (Queen Charlotte Islands). Saw-whet Owls are small, brown owls with broad reddish brown stripes on their chest. The brooksisubspecies was designated as Threatened by the Committee on the Status of Endangered Wildlife in Canada (COSEWIC) in April 2006 because they are a distinct subspecies endemic to Canada, have a small world population that is restricted to Haida Gwaii, and are a specialist of older forests that are declining in abundance because of threats such as forest harvest (COSEWIC2006).
Threats to Saw-whet Owls include the loss and degradation of habitat related to forest removal (medium and low levels of concern), the introduction of exotic wildlife species (medium level of concern), roadkill (low level of concern), and potentially global warming (medium level of concern). Forest removal, primarily from forest harvesting, reduces the availability of nesting trees, and alters forest habitats in a manner that may reduce prey availability and ability for Saw-whet Owls to hunt (among other effects). Forest removal also fragments the landscape, which may cause increased predation of Saw-whet Owls, reduced pairing opportunities, and increased energy expenditure for foraging (among other effects). Exotic species compete with Saw-whet Owls for nest sites and food, and potentially depredate nests. The introduction of Sitka black tailed deer in particular has led to a loss of understory forest habitat, which is thought to affect small mammal populations (prey for Saw-whet Owls).
There is some uncertainty about historic and current population sizes and habitat needs for Saw-whet Owls. Additional analyses and information are required to before it will be possible to determine an appropriate, long-term recovery target for the species. At this time it is only possible to set a short-term objective that focuses on preventing further population decline. The short-term population and distribution objective for Saw-whet Owl is therefore to maintain approximately 1800 adults across the species' extent of occurrence (10,000 km2across the Haida Gwaii archipelago) until more precise population and distribution targets can be formulated.
While some information is available on the general habitat needs of the Saw-whet Owl, more definitive information is required to understand and identify the habitat that is necessary for the survival and recovery of the species. Recent surveys identified some locations occupied by Saw-whet Owls and which likely provide suitable habitat for nesting. Based on these surveys, 12 habitat polygons are identified as critical habitat for the species in this recovery strategy. Critical habitat for the species will be revised when additional information becomes available.
One or more action plans for Saw-whet Owl will be completed by December 2018.
| Recovery criteria | Northern Saw-whet Owl, brooksisubspecies |
|---|---|
| 1. Are individuals capable of reproduction currently available to improve the population growth rate or population abundance? | Yes |
| 2. Is sufficient habitat available to support the species, or could it be made available through habitat management or restoration? | Yes |
| 3. Can significant threats to the species or its habitat be avoided or mitigated through recovery actions | Yes |
| 4. Do the necessary recovery techniques exist and are they known to be effective. | Yes |
Date of Assessment: April 2006
Common Name (population): Northern Saw-whet Owl brooksi subspecies
Scientific Name: Aegolius acadicus brooksi
COSEWICStatus:Threatened
Reason for Designation: This is a distinct subspecies endemic to Canada, with a small world population (ca. 1900 adults) restricted to the Queen Charlotte Islands. It is a forest specialist, preferring older forests with abundant nesting snags and an open understory in a landscape where such resources are continually becoming scarcer to forest harvest.
Canadian Occurrence: British Columbia
COSEWICStatus History: Designated Threatened in April 2006. Assessment based on a new status report.
| Natureserve (2006) conservation status ranks: | ||
|---|---|---|
| Global | G5T2T3 | Species globally secure (G5), but subspecies imperiled (T2) to vulnerable (T3) |
| National | N2N3 | Imperiled (N2) to vulnerable (N3) |
| Sub-national (B.C.) | S2S3 | Imperiled (S2) to vulnerable (S3) |
The Northern Saw-whet Owl brooksi subspecies (hereafter referred to as 'Saw-whet Owl') is a small, buffy-brown coloured owl (Fleming 1916). Their heads are relatively large with a prominent facial disk, and white streaking on the crown that converges in a 'V' between the eyes. The belly is broadly striped with dark reddish brown. Compared to the acadicus subspecies, the brooksi subspecies is darker and buffier in coloration. Saw-whet Owls are approximately 20 cm in length, and weigh between 75 g and 145 g, with females approximately 20% larger than males (Cannings 1993).
Range and distribution: Although the distribution of the Northern Saw-whet Owl is widespread throughout North America, the brooksi subspecies is globally restricted to Haida Gwaii (Queen Charlotte Islands) of northwestern British Columbia. A full 100% of the global range of the brooksi subspecies occurs in Canada. There is not thought to be any overlap between the A. a. acadicus and A. a. brooksi subspecies, and the brooksi subspecies is not thought to breed or occur outside of Haida Gwaii (Cannings 1993). On Haida Gwaii, Saw-whet Owls occur on provincial Crown lands, national park lands, private lands and Indian Reserves. Recent studies using radio telemetry (brooksi Northern Saw-whet Owl Recovery Team, unpubl. data) have found home range sizes on Haida Gwaii ranging from 153 to 410 ha in the breeding season and 178 to 908 ha during the non-breeding season. Elsewhere in North America, home range estimates have varied from 61 to 383 ha in the breeding season and from 9 to 249 ha in the non-breeding season (Rasmussen et al. 2008).
Population abundance: The estimated number of mature individuals is approximately 1,852 ± 580 (COSEWIC2006). There is only one population of the Saw-whet Owl.
Population trend: Population trends for Saw-whet Owls on Haida Gwaii have been projected using habitat as a surrogate for population size. Based on habitat availability, populations are estimated to have declined 13% in the last 15 years (3 generations), and are forecasted to decline a further 9% in the next 15 years if management practices remain at the status quo (COSEWIC2006).
Saw-whet Owls are found primarily in mature and old forests, and often near riparian areas (Holschuh and Hobbs 2002, COSEWIC2006). Saw-whet Owls have only been detected below 300 m in elevation, and have occurred in the submontane wet hypermaritime (CWHwh1), central very wet hypermaritime (CWHvh2), and montane wet hypermaritime (CWHwh2) variants of the coastal western hemlock biogeoclimatic zone1 (COSEWIC2006). Though there has been some search effort in habitats above 300 m in elevation, the bulk of survey effort has focused on lower elevations due to greater accessibility (Holschuh, unpub. data).
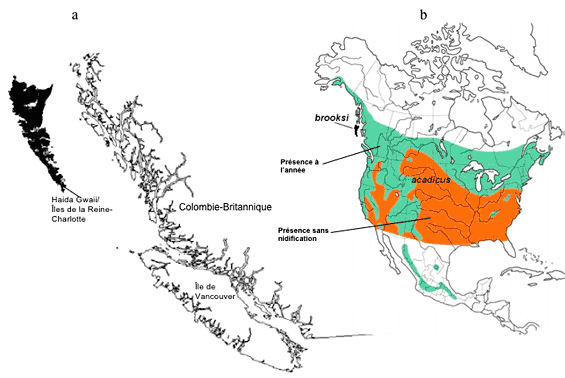
Figure 1. (a) Range of Saw-whet Owl on Haida Gwaii. This is the full Canadian range of the brooksi subspecies. (b) Global distribution of Northern Saw-whet Owl (acadicus and brooksi subspecies). Adapted from COSEWIC2006.
Forests used by Saw-whet Owls during the breeding season are typically composed of Western Hemlock (Tsuga heterophylla), Sitka Spruce (Picea sitchensis), Western Redcedar (Thuja plicata), and occasionally Mountain Hemlock (Tsuga mertensiana), Yellow Cedar (Chamaecyparis nootkatensis), and Shore (Lodgepole) Pine (Pinus contorta) (Holschuh 2004).
High quality nesting habitat is structurally complex forest that has a high relative abundance of nesting snags, features found primarily in mature and old forests. Saw-whet Owls are likely detected more often in mature and old forests due to their reliance on these features. Saw-whet Owls are secondary cavity nesters, and availability of suitable nesting trees is thought to be limiting for Saw-whet Owls on Haida Gwaii. Suitable nesting cavities are more frequently observed in Sitka Spruce leading stands than Western Hemlock leading stands. Western Redcedar stands have the lowest relative availability of suitably sized cavities (F. Doyle, unpub. data). Four active Saw-whet Owl nests have been recorded on Haida Gwaii, though nest searching effort for Saw-whet Owls has been limited. The nests were likely excavated by Northern Flickers or Hairy Woodpeckers, and were located in Western Hemlock and Sitka Spruce trees. The nest trees were all dead, had lost the majority of their branches and bark, and showed signs of internal decay (including breaking the top portion of the tree). The diameter at breast height (DBH) of the four nest trees ranged from 70 cm to 130 cm, and cavity openings were approximately 7-9 cm in diameter (Holschuh 2004). The timing of breeding has been recorded to be quite asynchronous, with nesting starting between early March and early May, and fledging continuing until August (COSEWIC2006). Areas dominated by young regenerating forests have been observed to have reduced detections of Saw-whet Owls during the breeding season (several watersheds on Graham and northern Moresby Islands), whereas the detection rates and apparent densities of Saw-whet Owls in areas with greater amounts of mature and old growth forests are higher (e.g., in Gwaii Haanas National Park Reserve and Haida Heritage Site) (COSEWIC2006). Mature and old forests are considered a limiting factor for Saw-whet Owls.
Being secondary cavity nesters, Saw-whet Owls are dependent on primary cavity excavators, including Northern Flicker (Colaptes auratus), Hairy Woodpecker (Picoides villosus picoideus), and Red-breasted Sapsucker (Sphyrapicus ruber) (Cannings 1993). When first excavated, the woodpecker cavities are often not of sufficient diameter for Saw-whet Owl nesting .As cavities increase in size due to decay and other excavators, they eventually may provide suitable cavities for Saw-whet Owl nesting. Cavity availability is therefore a limiting factor to Saw-whet Owls.
Limited detections in young forest suggest that foraging habitat is largely in mature and old forests, riparian areas and forest edges during the breeding season (Holschuh 2004). Young, regenerating forests likely are less suitable for foraging, due to the high density of trees, and low food species availability.
Outside the breeding season, habitat use has not been systematically documented for Saw-whet Owls. Unlike its continental counterparts, Saw-whet Owls do not migrate; however, dietary analysis and surveys suggest some birds may shift to more coastal areas outside of the breeding season. Birds collected during fall indicated high consumption of marine invertebrates (Hobson and Sealy 1991, Sealy 1999). Road collision rates on the highway on the eastern coastline of Graham Island are typically greater during fall, also suggestive of a seasonal shift.
Saw-whet Owls rely on a variety of foods throughout the year, and are thought to be opportunistic feeders. Dietary analyses have been limited to sampling stomach contents of road-killed individuals during fall, and opportunistic observations. Saw-whet Owls appear to rely heavily on invertebrates in their diet, including amphipods (Orchestria traskiana, O. californiana), isopods (Ligia pallasii), and diptera (Coelopa vanduzeei). Vertebrate food items that have been documented include Western Toad (Bufo boreas), Ancient Murrelet (Synthliboramphus antiquus), Chestnut-backed Chickadee (Poecile rufenscens), Golden-crowned Kinglet (Regulus satrapa), Hermit Thrush (Catharus guttatus), Dusky Shrew (Sorex monticola), and Deer Mouse (Peromyscus maniculatus) (Sealy 1991, Sealy 1999, Gaston 1992).
1 Biogeoclimatic zones are ecosystem classification units used to describe areas with broadly homogeneous macroclimates. They are further subdivided into subzones and variant based on precipitation and temperature regimes, and elevation or latitude (Meidinger and Pojar 1991).
Habitat loss caused by removal of mature and old forest, primarily through industrial forest harvesting, is the largest general threat to the Saw-whet Owl (COSEWIC2006). Forestry is a major industry on Haida Gwaii, with approximately 25% of the forested landscape having been previously logged (A. Cober, pers. comm.). Land development for other reasons also occurs, but is very localized due to a generally small human population.
Given that 25% of Haida Gwaii forests have previously been harvested, forests across most of the archipelago are at various stages of regeneration (recently harvested to young and maturing forests). Most of the productive watersheds on Graham Island have been extensively logged, resulting in a mosaic of patches of mature and old forests surrounded by younger forests. Conversion of forest to younger seral stages potentially affects Saw-whet Owls because it causes (a) a loss of nesting habitat, (b) a loss of old and mature forest habitat for foraging, and (c) fragmentation of suitable habitat.
The negative effects of habitat loss and fragmentation are demonstrated by a reduced detection frequency of Saw-whet Owls during the breeding season in areas dominated by young regenerating forests (several watersheds on Graham and northern Moresby Islands), compared to higher detection rates of Saw-whet Owls observed in areas with greater amounts of old growth forests (e.g., Gwaii Haanas National Park Reserve and Haida Heritage Site) (COSEWIC2006).
Approximately 3,962 km2 of Haida Gwaii (39% of the land base) was in conservation areas (up to 2012), the largest of which are Gwaii Haanas National Park Reserve and Haida Heritage Site and Naikoon and Pure Lake Provincial Parks. In addition, under the Haida Gwaii Strategic Land Use Agreement, an additional 2,641 km2 (26% of land base) has now been included in new conservancies. And through an Ecosystem Based Management (EBM) initiative, an additional 616 km2 (6% of land base) of reserves have been set aside through the Haida Gwaii Land Use Order Objective. In total, approximately 7,219 km2 (71%) of the Haida Gwaii land base is now reserved from forestry operations. This extensive level of protection will contribute to recovery of the species, lessening the impact of the forest removal threat.
The introduction of a number of exotic species has significantly affected the ecology and native species of Haida Gwaii. Sitka Black-Tailed Deer are established throughout Haida Gwaii, with the exception of a few smaller islands, and their high numbers, largely due to a lack of predators, have led to over-browsing. As a result of this over-browsing, the understory of Haida Gwaii forests is very sparse compared to similar coastal regions that have more predators. This has the potential to reduce food availability for Saw-whet Owls including rodents and forest invertebrates that are important to Saw-whet Owls during the breeding season (Cannings 1993, RGIS-FRBC 2001, Allombert et al. 2005).
Saw-whet Owls are vulnerable to nest predation by mammals introduced to Haida Gwaii, including Red Squirrels (Tamiasciurus hudsonicus) (Martin and Joron 2003), Common Raccoons (Procyon lotor), and Norway Rats (Rattus norvegicus) (Golumbia et al. 2002). European Starling populations are established near human settlements on Haida Gwaii, and have been documented harassing Saw-whet Owls on the nest (COSEWIC2006). The quantitative effect of these threats on the population size and dynamics of Saw-whet Owls has not been documented.
The introduction of Black Rats (Rattus rattus) and later Norway Rats may have had direct and indirect effects on Saw-whet Owls. Both species have been documented to predate nests of a variety of sea birds, and their presence has been linked to a decline in various bird species (Golumbia et al. 2002). Rats have also been linked to a decrease in deer mice and shrews, which are important food species to Saw-whet Owls (Kaiser et al. 1997).
Though Barred Owls (Strix varia) have not expanded their range to Haida Gwaii, they have the potential to do so. Barred Owls are known to predate on small owl species (Elliot 2006), and a range expansion to Haida Gwaii has the potential to increase Saw-whet Owl mortality.
Collision with vehicles is a less severe threat to Saw-whet Owls. Vehicle collisions generally only occur during fall along the coastal highways (Sealy 1999). The effect of this accidental mortality on the population as a whole has not been quantified, and based on incidental reports, the proportion of the population that is affected is assumed to be relatively low.
The Haida Gwaii landscape is dynamic with forest ecosystem change being driven by forest harvest and climate change, in addition to other natural processes. The potential effects of climate change on Haida Gwaii are not yet well understood, but may have large effects on the fauna and flora of the archipelago. Climate data trends from north coastal and north central British Columbia indicate a trend toward increased precipitation and temperatures over the past 20 years (Frank Doyle, unpub. data). Such change has the potential to affect the forest ecosystem composition and function. For example, wetter conditions may lead to greater abundance of western redcedar dominated stands across the landscape, which have the lowest availability of nesting cavities.
In Canada, the brooksi subspecies of Saw-whet Owl is found only on the Haida Gwaii archipelago and therefore has a naturally restricted distribution. COSEWIC(2006) reported that suitable habitat for the species is decreasing. However, preliminary studies from ongoing fieldwork indicate that habitat use may be more flexible in terms of suitable habitat requirements. For example, Saw-whet Owls appear in preliminary analyses to use a wider age range of habitat class, however the winter samples are small and we have none on female summer habitat use. Nor do we know if the birds using mostly second growth are breeding successfully or whether these areas are population sinks (brooksi Northern Saw-whet Owl Recovery Team, unpubl. data).
COSEWICestimated the historic and current population sizes to be 3,100 ± 840 and 1,852 ± 580 mature individuals, respectively (COSEWIC2006). However, population estimates are extrapolated from measures of density and estimates of the amount of habitat available, a method that is recognized to have numerous limitations (COSEWIC2006). Additional data and analyses (e.g., population viability analyses) are required before it will be possible to determine an appropriate, quantitative, long-term recovery target for Saw-whet Owls. The section on broad strategies and general approaches to meet objectives (see Section 6) responds to knowledge gaps so that long-term, feasible and quantitative objectives can be set in the future.
Given the uncertainty about population size and habitat needs for the Saw-whet Owl, at this time it is only possible to set a short-term objective that focuses on preventing further population decline for the species across its range. The short-term population and distribution objective for Saw-whet Owl is therefore to maintain approximately 1800 adults across the species' extent of occurrence (10,000 km2across the Haida Gwaii archipelago) until more precise population and distribution targets can be formulated.
The following recovery and management actions for Saw-whet Owl have been conducted or initiated to date:
- Habitat is conserved in the Gwaii Haanas National Park Reserve and Haida Heritage Site, Naikoon and Pure Lake Provincial Parks, and other previously existing conservation areas on Haida Gwaii (approximately 39% of the Haida Gwaii land base; see Section 4.2). It is not known how much of this conserved habitat is suitable for Saw-whet Owls.
- In addition, new conservation areas and management zones, representing approximately 32% of Haida Gwaii, have been established through the Haida Gwaii Strategic Land Use Agreement (see Section 4.2). This will result in a total of 71% of Haida Gwaii being conserved (including both new and previously enacted conservation areas), including large areas of mature and old forests throughout the archipelago. As above, it is not known how much of this conserved habitat is suitable for Saw-whet Owls.
- Under the Haida Gwaii Strategic Land Use Agreement, , Ecosystem Based Management (EBM)2 was adopted as a framework for guiding future land management on Haida Gwaii. The intent of EBM is to "maintain spatial and temporal characteristics of ecosystems such that component species and ecological processes can be sustained, and human well-being supported and improved" (LUP 2006).The recovery team believes that defining habitat suitability and availability for Saw-whet Owls within each landscape unit3 will be crucial to help facilitate effective implementation of the Haida Gwaii Strategic Land Use Agreement and future forest management activities and thereby ensure a constant availability of suitable habitat for this species.
- Twelve Saw-whet Owl habitat areas (at 11 locations), ranging in size from 38 to 111 ha, are currently conserved under the Council of the Haida Nation's Haida Stewardship Law and the Province of British Columbia's Haida Gwaii Land Use Objectives Order.
- Stewardship activities including volunteer based monitoring in road accessible areas on Haida Gwaii.
- Opportunistic surveys to better refine habitat specific densities for Saw-whet Owls in Gwaii Haanas National Park Reserve and Haida Heritage Site are being completed and will continue into the future as resources are available.
- Research is currently being conducted to assist in determination of the characteristics of critical habitat (e.g., call playback surveys and radio telemetry to better determine home range and nest site characteristics), identify individuals based on vocalizations and determine diet in winter and summer.
2 EBM, under the definition of the Haida Gwaii Strategic Land Use Agreement, has the intent of "maintain[ing] spatial and temporal characteristics of ecosystems such that component species and ecological processes can be sustained, and human well-being supported and improved".
3 Landscape units are areas of land used for long-term planning of resource management activities. They are usually 50,000 to 100,000 hectares in size (ILMB 2008).
The recovery planning table (Table 3) identifies activities to meet the short term population and distribution objectives and to identify appropriate, long-term quantitative population and distribution targets for the species. To meet population objectives, the most urgent activities address knowledge gaps about population size and trends. It is necessary to gain a better understanding of the effects of invasive wildlife species on Saw-whet Owls, and beneficial to monitor additional direct threats on the population size. Filling these knowledge gaps should allow for interim measures to allow for conservation of the population while research continues.
To address short and long term distribution objectives, the most urgent activities include analyzing existing habitat information about Saw-whet Owls, developing a habitat suitability model, and conducting more detailed research about important habitat attributes. This information can be incorporated in the process to define habitat needed for survival or recovery of the species. Where appropriate, habitat may be secured for Saw-whet Owls through various land management approaches.
Conducting fall and winter habitat use surveys is necessary to determine the importance of these habitats on the viability of Saw-whet Owls on Haida Gwaii. It is also necessary to gain a better understanding of prey species selection, and whether population cycles of these prey occur. As possible, the recovery strategy and species management in general should be integrated with other recovery plans and land use planning processes on Haida Gwaii (e.g., ecosystem level management) to ensure maximum conservation for the Saw-whet Owl.
It would be beneficial to work with land use proponents to provide advice about mitigating potential effects on the Saw-whet Owl.
Under SARA, critical habitat is defined as "the habitat that is necessary for the survival or recovery of a listed wildlife species and that is identified as the species' critical habitat in the recovery strategy or in an action plan for the species". For Saw-whet Owl, critical habitat is identified to the extent possible, based on the best information available as of November 2010. The critical habitat identified within this recovery strategy is necessary for the survival and recovery of the species, but is most likely only a part of what ultimately needs to be identified as required for the survival and recovery of the Saw-whet Owl in Canada. Critical habitat for the species will be revised when additional information becomes available, for example through the studies outlined in Section 7.2.
While some information is available on the general habitat needs of the Saw-whet Owl, more definitive information is required to understand and identify the habitat that is necessary for the survival and recovery of the species. Habitat suitability modeling has been initiated with the primary objective of identifying critical habitat for the species, but currently the model is not sufficiently developed to achieve accurate delineation of critical habitat boundaries. For example, the model has not yet been tested in the field and computer analysis has shown that it currently excludes some breeding habitat that is known to be occupied. Only four Saw-whet Owl nests have been found, so knowledge on breeding habitat requirements is sparse. In addition, habitat use outside of the breeding season has not been well documented. Currently, information about fall movements to coastal areas is deduced primarily from dietary and isotope analyses (Hobson and Sealy 1991) and limited surveys in 2009/10 (brooksi Northern Saw-whet Owl Recovery Team, unpubl. data). It is not known how critical overwintering habitat attributes are to the viability of Saw-whet Owl populations.
However, recent surveys identified some locations occupied by Saw-whet Owls and which likely provide suitable habitat for nesting. Holschuh and Hobbs (2002) surveyed 133 sites on Graham Island by listening for Saw-whet Owls that were spontaneously calling or using a call playback device and waiting for a response from nearby Saw-whet Owls. Owls were detected at 21 of the surveyed locations.
Holschuh and Hobbs (2002) subsequently produced habitat polygons for 12 locations where owl presence was repeatedly confirmed and suitable nesting habitat was present, towards identification of these polygons as Wildlife Habitat Areas under B.C.'s Identified Wildlife Management Strategy (IWMS) and Forest and Range Practices Act. Habitat polygons were created using Saw-whet Owl location data to, where appropriate, identify a daytime roost or suspected nest location as a centre-point. Information from digital ortho-photo coverage was used to ensure that the polygon captured surrounding mature and old forest. An attempt was also made to capture forest that was likely to contain a greater density of wildlife trees (e.g., large dead trees or snags). Although mature forest is the species' preferred habitat, some areas of younger forest were included to provide habitat for recruitment or foraging opportunities.
In 2009, eight of the 12 locations were re-surveyed and Saw-whet Owls were detected again at all eight locations (brooksiNorthern Saw-whet Owl Recovery Team, unpubl. data). No survey data were available for the other four locations; three could not be accessed due to a heavy snow pack and one occurred on private property. Given that all 12 habitat polygons were developed based in part on repeated detections of Saw-whet Owls, and that the eight re-surveyed locations were still occupied, it is reasonable to assume that the four sites that could not be re-surveyed also continue to be occupied by the species.
As of November 2010, these 12 habitat polygons represented the best available information on the geographical location of habitat that is necessary for the survival and recovery of Saw-whet Owls. As such, they are identified as critical habitat for the species in this recovery strategy. The remaining nine locations where Saw-whet Owls were detected by Holschuh and Hobbs (2002) are included in widespread surveys for the species scheduled for 2009 through 2013. If analysis of data compiled from these surveys determines that the locations are still occupied by Saw-whet Owls they will be considered for addition to the critical habitat identification for the species.
Biophysical Attributes of Critical Habitat
Critical habitat includes forested habitat of all ages within the polygons, but excludes roads and water. Forested habitat is required by Saw-whet Owls to provide viable areas for nesting, roosting, cover and foraging. The required biophysical attributes of forest stands include unfragmented stands of structurally complex forest, with a relatively intact and multi-storied canopy, open forest interior, abundant large diameter snags for nesting (i.e., > 50 cm diameter at breast height), abundant trees with dense foliage for roosting, a productive understory that provides high quality habitat for prey species (e.g., abundant shrubs and woody debris), abundant prey, and the presence of cavity excavators (e.g., woodpeckers). Some areas of sub-optimal habitat (e.g., young regenerating stands) have been included in critical habitat polygons because they still serve a biological function for the species (e.g., foraging habitat or protection from edge effects) and will mature into higher quality habitat.
Geographical Location of Critical Habitat
Critical habitat is identified at the following 12 locations. All location names and critical habitat polygons are as per Holschuh and Hobbs (2002). Field notes describing activity at the locations are primarily from Holschuh and Hobbs (2002), except for information relating to 2009 surveys at the same locations (brooksi Northern Saw-whet Owl Recovery Team, unpubl. data). All but one location (19 kmLake) are on B.C. Crown land and fall within the conservancies established under the Haida Gwaii Strategic Land Use Agreement. The 19 km Lake location is on private land. This partial identification of critical habitat is not sufficient to meet the objectives of this recovery strategy. Total area included in this identification is 908 ha.
Eli Tingley Creek
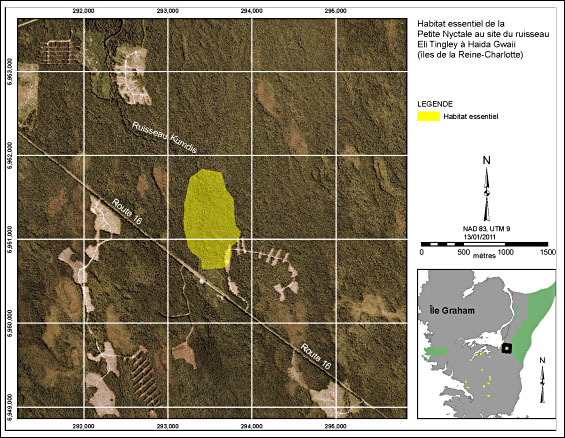
The Eli Tingley Creek site is located where the Eli Tingley Creek crosses Highway 16 (Figure 2). A Saw-whet Owl was detected at this location on four occasions between March and June in 2002 (Holschuh and Hobbs 2002) and on at least one occasion in 2009 (brooksi Northern Saw-whet Owl Recovery Team, unpubl. data). This critical habitat polygon is about 59 ha in area and consists of upland and riparian old-growth forest with high canopy complexity. The site has a sufficient snag density for nesting.
Dat-1/5
The Dat 1/5 site is located on the Datlamen Main Forest Service Road in Tree Farm License (TFL) 60 (the west polygon on Figure 3). A Saw-whet Owl was detected at this location on eight occasions between March and May in 2002 (Holschuh and Hobbs 2002) and on at least one occasion in 2009 (brooksi Northern Saw-whet Owl Recovery Team, unpubl. data). On one survey in 2002, two Saw-whet Owls were detected simultaneously, on the south-east side of the lake shown on Figure 3 (within the west polygon) and to the north of the lake. Having two individuals detected simultaneously indicates that there likely were two bordering territories in the remaining patch of mature forest to the north and to the south-east of the lake. The critical habitat polygon is about 72 ha and consists of mature and old-growth forest, consistently with a multi-storied canopy.
Dat 3/4
The Dat 3/4 site also is located on the Datlamen Main Forest Service Road in TFL 60 (the east polygon on Figure 3). A Saw-whet Owl was detected at this location on five occasions between March and June in 2002 (Holschuh and Hobbs 2002) and on at least one occasion in 2009 (brooksi Northern Saw-whet Owl Recovery Team, unpubl. data). On one occasion in 2002, two Saw-whet Owls were detected simultaneously, so the critical habitat polygon for this area includes an area that both Saw-whet Owls were thought to be using. The critical habitat polygon is about 87 ha and is characterised by mature second-growth Western Hemlock forest. The area has been selectively logged, but several veteran trees remain, as well as a sufficient snag density for nesting. A riparian strip with old growth characteristics also is present in the polygon, further increasing habitat suitability and providing gap openings that may have been useful for foraging as well as potential nesting habitat.
QC 11 km North
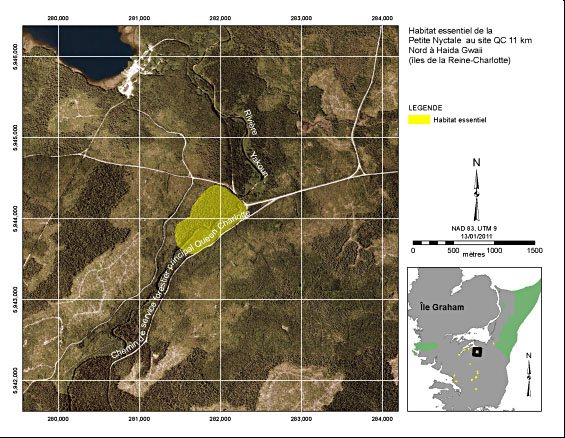
The QC 11 km North site is located on the Queen Charlotte Main Forest Service Road in TFL 60 (Figure 4). A Saw-whet Owl was detected calling spontaneously (i.e., no Saw-whet Owl recording had yet been played on the survey) on two occasions between March and May in 2002 (Holschuh and Hobbs 2002) and on at least one occasion in 2009 (brooksi Northern Saw-whet Owl Recovery Team, unpubl. data). The spontaneous observations in 2002 confirmed the presence of an actively defended territory in the area that was likely used for nesting. The critical habitat polygon is about 43 ha and consists of a mosaic of forest ages, centering on a mature and old-growth forest riparian buffer adjacent to the Yakoun River that runs through the middle of the polygon. Surrounding this is younger forest, which with time and proper management would begin to show important habitat characteristics. These areas also are captured in the critical habitat polygon.
Begbie
The Begbie site is located on the Begbie Forest Service Road in TFL 60 (Figure 5). A Saw-whet Owl was detected near Juskatla Inlet on six occasions between March and May in 2002 (Holschuh and Hobbs 2002) and on at least one occasion in 2009 (brooksi Northern Saw-whet Owl Recovery Team, unpubl. data). Call playback on one occasion in 2002 elicited a response in the form of ksew calls, as described by Cannings (1993), followed by short and rapid territorial calling. This behaviour may have indicated the proximity of a nest site. Although repeated searching for the nest site in the area failed, the Saw-whet Owl most likely was nesting in the area due to a continual presence and territorial behaviour. A roost site also was found in a large (70 cm diameter at breast height) Western Hemlock tree. The critical habitat polygon is about 72 ha and includes the roost site and the surrounding old-growth forest on the west side of the road near the mouth of a creek. The forest at the site has been selectively harvested, but only a small percentage of the volume was removed and the old growth forest structure largely is intact. The stand features many large trees and snags that offered potential nesting opportunities.
Bragg
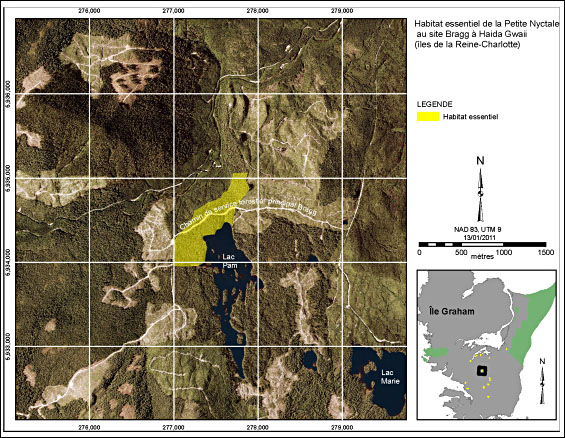
The Bragg site is located on the Bragg Forest Service Road in TFL 60 (Figure 6). A Saw-whet Owl was detected calling spontaneously on four occasions between April and June 2002 near the north end of Pam Lake (located south of the polygon; Holschuh and Hobbs 2002). No follow up surveys were completed at this site in 2009 because the site could not be accessed due to a large snow pack. In 2002, a strong territorial response was observed on one occasion as a Saw-whet Owl approached within 30 m of the surveyors. The critical habitat polygon is about 38 ha and consists of mature forest on the north end of Pam Lake further north across the road that passes through the polygon. This stand was the main apex of Saw-whet Owl activity for this location. The old-growth forest present has a sufficient snag density for nesting, especially the forest adjacent to Pam Lake.
QC 33 km North
The QC 33 km North site is located on the Queen Charlotte Main Forest Service Road in TFL 60 (Figure 7). This site has two critical habitat polygons. A Saw-whet Owl was detected at this site on five occasions between March and May in 2002 (Holschuh and Hobbs 2002) and on at least one occasion in 2009 (brooksiNorthern Saw-whet Owl Recovery Team, unpubl. data). The critical habitat polygons total to about 64 haand include mostly mature forest, but also a small area of regenerating forest that contained several large veteran trees and a small creek.
QC 32 km South
The QC 32 km South site is located on the Queen Charlotte Main Forest Service Road in TFL 60 (north polygon on Figure 8). A Saw-whet Owl was detected on five occasions between March and May in 2002, including an observation on one occasion of a male and female vocalizing spontaneously (Holschuh and Hobbs 2002). The repeated detections and confirmation of both male and female presence in the area provided evidence that a pair was nesting in the area. The site was re-visited in 2009 and on at least one occasion a Saw-whet Owl was detected (brooksi Northern Saw-whet Owl Recovery Team, unpubl. data). The critical habitat polygon is about 105 ha and consists of mature and old-growth riparian forest habitat with sufficient snag density for nesting. Red-breasted Sapsucker (Sphyrapicus ruber) sign was abundant in the area in 2002, indicating the presence of cavity excavators. This species acts as one of the primary excavators of cavities that are later used by Saw-whet Owls. Several small streams and boggy areas surround the river, adding to the habitat complexity (and thus quality) of the site.
Ghost
The Ghost site is located on the Ghost Main Forest Service Road in TFL 60 (south polygon on Figure 8). A Saw-whet Owl was detected on two occasions between April and June in 2002 (Holschuh and Hobbs 2002) and on at least one occasion in 2009 (brooksi Northern Saw-whet Owl Recovery Team, unpubl. data). The critical habitat polygon is about 111 ha and consists of old-growth cedar (Western Red and Yellow), Western Hemlock, and Sitka Spruce forest with an intact understory (including Vaccinium spp.that are commonly absent from Haida Gwaii forests due to deer browsing). Interspersed at the site were open boggy areas as well. This productive mosaic habitat likely supports a healthy rodent population. Snag density is sufficient for nesting.
Riley Creek
The Riley Creek site is located near the Rennel Sound shoreline near the Riley Creek trailhead (Figure 9). A Saw-whet Owl was detected on seven occasions between March and June in 2002 (Holschuh and Hobbs 2002). No follow up surveys were completed at this site in 2009 because the site could not be accessed due to a large snow pack. On one occasion in 2002, two males were heard counter-singing. Hence, two males seemed to be defending active territories in this area. As well, Gill and Cannings (1997) previously detected a Saw-whet Owl in the area. In 2002, both a roost and a nest site were found at the site. The nest was located 25 m up a 28 m tall western hemlock snag (150 cm diameter), in line with the rest of the canopy. The critical habitat polygon is about 100 ha and is characterized mostly by mature and old-growth forest dominated by Sitka Spruce, Western Hemlock and Western Redcedar. Sections of low lying, poorly drained and nutrient poor habitat are interspersed within the mature forest and provide valuable habitat complexity.
19 km Lake
The 19 km Lake site is located on the Queen Charlotte Main Forest Service Road on land owned by Island Timberlands (Figure 10). A Saw-whet Owl was detected on five occasions between March and May in 2002 (Holschuh and Hobbs 2002) and on at least one occasion in 2009. A daytime roost site was discovered on one survey in 2002. The critical habitat polygon is about 94 ha and, although adjacent areas have been extensively harvested, the polygon still has mature forest remaining with some old-growth characteristics (as of 2010). The site has an abundance of snags, many of which have potential nesting cavities. At least eight Red-breasted Sapsuckers were noted to be in this area in 2002. This species acts as one of the primary excavators of cavities that are later used by Saw-whet Owls. The critical habitat polygon also includes some younger forest that may provide good feeding resources, and over time will develop the structural complexity important to Saw-whet Owl breeding habitat.
Hangover Creek
The Hangover Creek site is located on the Rennel Shore Mainline near the Bonanza Creek bridge (Figure 11). A Saw-whet Owl was detected on three occasions between March and May in 2002 (Holschuh and Hobbs 2002). No follow up surveys were completed at this site in 2009 because the site could not be accessed due to a large snow pack. This location also is in the proximity of Saw-whet Owl detections observed by Gill and Cannings (1997). The critical habitat polygon is about 63 ha and is characterized by mature and old-growth riparian forest that parallels the road along the eastern margin of the site. The Saw-whet Owl detection sites in 2002 were spread along a fairly long area, which was consistent with the shape of remaining mature forest habitat.







The primary threats to Saw-whet Owl critical habitat are through alterations to forest structure and function (Table 5). On Haida Gwaii, this occurs primarily through forest harvesting, but could also occur from other activities (e.g., private land development, mining, power line construction, tree cutting for firewood, etc.). These and other activities can remove occupied or potential nest trees, increase mortality from predation, reduce pairing opportunities, increase competition, reduce populations of primary nest site excavating species (e.g., woodpeckers), affect foraging success (e.g., through changes in prey populations or increased energy expenditure for foraging), among other effects.
Destruction must be determined on a case-by-case basis. Destruction would result if part of the critical habitat were degraded, either permanently or temporarily, such that it would not serve its required biological function for Saw-whet Owls (to provide viable habitat for nesting, roosting, cover and foraging). Destruction may result from single or multiple activities at one point in time or from the cumulative effects of one or more activities over time. Destruction may originate from activities inside or outside of critical habitat polygons.
It is recognized that existing facilities and land uses in and adjacent to critical habitat already affect critical habitat, to various degrees, and may decrease the quality of certain portions of critical habitat. Lower quality or sub-optimal areas may nonetheless be included as critical habitat because they serve a biological function for the species (e.g., foraging habitat or protection from edge effects). Any new, additional, or increases in activity (including the construction of new facilities) may cause destruction of critical habitat. Some human activities in or adjacent to critical habitat will require careful assessment for possible effects, including cumulative effects on critical habitat and the potential for destruction.
The performance indicators presented below provide a way to define and measure progress toward achieving the population and distribution objectives. Specific progress towards implementing the recovery strategy also will be measured against indicators outlined in subsequent action plans.
Performance measures for population objectives:
- The location and amount of current suitable habitat across the extent of occurrence of Saw-whet Owl is determined by 2015, and suitable habitat is maintained at 2015 levels by 2018.
- A population estimate of Saw-whet Owl is calculated by 2015, and shows evidence of remaining stable or increasing by 2018.
- Appropriate, long-term quantitative population and distribution targets are determined for Saw-whet Owl by 2018.
Performance measures for distribution objectives:
- A process for identifying habitat needed for survival and recovery of the species is outlined and shared with recovery partners by 2015, and habitat required to achieve long-term population and distribution targets is identified by 2018.
- A conservation gap analysis of the Haida Gwaii Strategic Land Use Agreement compared to the amount and spatial configuration of habitat needed to achieve long-term population and distribution targets is completed, and any further conservation measures needed to meet the targets are forwarded to relevant decision makers or planning initiatives by 2019.
- These habitat requirements of Saw-whet Owls are incorporated into federal, provincial, and local guidelines by 2028.
- Effects of invasive species on Saw-whet Owls are determined, and strategies to manage or mitigate these effects are developed and implemented by 2028.
- An effects assessment of Saw-whet Owl road mortality on population viability and persistence is completed, and mitigation or reduction strategy is implemented by 2028.
It should be noted that these performance measures have been developed assuming that the listed threats in Table 1 continue to have significant population level effects.
One or more action plans will be completed by March 2018.
Allombert, S., A.J. Gaston, and J. Martin. 2005. A Natural Experiment on the Impact of Overabundant Deer on Songbird Populations. Biological Conservation 126:1-13.
BC CDC. 2008. BC Species and Ecosystems Explorer. BC Ministry of Environment - Conservation Data Centre. Victoria, BC. Available: http://a100.gov.bc.ca/pub/eswp/(Accessed August 2008).
Cannings, R.J. 1993. Northern Saw-whet Owl (Aegolius acadicus). PP 20 in Poole, A. and F. Gill (eds.). The Birds of North America. The American Ornithologists Union, Washington, DC.
Cober, Alvin. Personal Communication. B.C. Ministry of Forests, Lands and Natural Resource Operations, Queen Charlotte, B.C.
COSEWIC. 2006. COSEWIC assessment and status report on the Northern Saw-whet Owl brooksi subspecies Aegolius acadicus brooksi in Canada. Committee on the Status of Endangered Wildlife in Canada. Ottawa. Vi+23pp. (www.sararegistry.gc.ca/status/status_e.cfm).
Elliott, K. 2006. Declining Numbers of Western Screech-owl in the Lower Mainland of British Columbia. British Columbia Birds. 14: 2-11.
Environment Canada, Parks Canada Agency, and Fisheries and Oceans Canada. 2005. Species at
Risk Act Policy: recovery - draft policy on the feasibility of recovery. April 15, 2005.
Ottawa, ON.
Fleming, J.H. 1916. The Saw-whet Owl of the Queen Charlotte Islands. The Auk. 33: 420-423.
Gaston, A.J. 1992. The Ancient Murrelet: a natural history in the Queen Charlotte Islands, British Columbia. Occasional papers from the British Columbia Provincial Museum. Victoria, BC.
Golumbia, T., L. Bland, K. Moore, and P. Bartier. 2002 History and Current Status of Introduced Vertebrates on Haida Gwaii (Queen Charlotte Islands). Unpublished Technical Report. 55 pp.
Government of Canada 2009. Species at Risk Act Policies, Overarching Policy Framework [Draft]. Species at Risk Act Policy and Guidelines Series. Environment Canada, Ottawa. 38 pp.
Hobson, K.A. and S.G. Sealy. 1991. Marine protein contribution to the diet of Northern Saw-whet Owls on the Queen Charlotte Islands: a stable isotope approach. Auk 108: 437-440.
Holschuh, C.I. 2004. Monitoring habitat quality and condition of Queen Charlotte Saw-whet Owls (Aegolius acadicus brooksi) using vocal individuality. M.Sc. dissertation, University of Northern British Columbia, Prince George, BC. 77 pp.
Holschuh, C.I., and J. Hobbs. 2002. Queen Charlotte Saw-Whet Owl: An Inventory of Occupied Sites On Graham Island. Unpublished report to the BC Ministry of Environment, Smithers, BC.
ILMB. 2008. Integrated Land Use Management Bureau. Land Use Planning Questions and Answers. Available online at: http://ilmbwww.gov.bc.ca/slrp/srmp/Background/docs/GuideQAs.pdf(Accessed January 2009).
Kaiser, G.W., R.H. Taylor, P.D. Buck, J.E. Elliott, G.R. Howald and M.C. Drever. 1997. The Langara Island Seabird Habitat Recovery Project: eradication of Norway rats, 1993-1997. Prepared by Pacific Wildlife Research Centre, Canadian Wildlife Service, Environment Canada, Delta, BC, R.H. Taylor Associates, Nelson, New Zealand, Systems Forestry Consulting, Masset, BC for Canadian Wildlife Service, Pacific and Yukon Region, BC Canadian Wildlife Service Technical Report Series No. 304. 81 pp.
LUP. 2006. Haida Gwaii/ QCI Land Use Plan Recommendations Report. BC Integrated Land Use Management Bureau.
Martin, J.L. and M. Joron. 2003. Nest predation in forest birds: influence of predator type and predator's habitat quality. Oikos 102: 641-653.
Meidinger, D. and J. Pojar. 1991. Ecosystems of British Columbia. B.C. Ministry of Forests and Range. Victoria, BC.
NatureServe. 2006. NatureServe Explorer: An online encyclopedia of life [web application]. Version 7.0. NatureServe, Arlington, Virginia. Available http://www.natureserve.org/explorer. (Accessed: Oct 20, 2010).
Rasmussen, Justin Lee, Spencer G. Sealy and Richard J. Cannings. 2008. Northern Saw-whet Owl (Aegolius acadicus), The Birds of North America Online (A. Poole, Ed.). Ithaca: Cornell Lab of Ornithology; Retrieved from the Birds of North America Online: http://bna.birds.cornell.edu/bna/species/042. doi:10.2173/bna.42
RGIS-FRBC. 2001. Forest ecology, forest renewal, and introduced species in Haida Gwaii, interim report. (April - September 2001). Prep. By Sean Sharpe. Science Council of British Columbia, Forest Renewal British Columbia, Burnaby, BC, Canada. 27pp.
Sealy, S.G. 1999. Further data on food items of Northern Saw-whet Owls (Aegolius acadicus brooksi) on the Queen Charlotte Islands, British Columbia. Western Birds 30: 200-205.
Wijdeven, Berry. Personal Communication. B.C. Ministry of Forests, Lands and Natural Resource Operations, Queen Charlotte, B.C.
In accordance with The Cabinet Directive on the Environmental Assessment of Policy, Plan and Program Proposals, 2004, a strategic environmental assessment (SEA) is conducted on all Species at Risk Act (SARA) recovery strategies. The purpose of a SEA is to incorporate environmental considerations into the development of public policies, plans, and program proposals to support environmentally sound decision-making.
Recovery planning is intended to benefit species at risk and biodiversity in general. However, it is recognized that strategies may also inadvertently lead to environmental effects beyond the intended benefits. Environmental effects, including impacts to non-target species and the environment, were considered during recovery planning. The SEA is not a separate document, but is incorporated directly into the recovery strategy and is summarized here.
This recovery strategy will clearly benefit the environment by promoting the recovery of the Northern Saw-whet Owl, brooksi subspecies (Aegolius acadicus brooksi) through addressing knowledge gaps associated with population size, dispersal / seasonal distribution, and suitable habitat (short term), and reduction / mitigation of threats and maintenance of sufficient habitat on the landscape (long term).
The SEAconcluded that this recovery strategy would have several positive effects and not cause any important negative effects. Other wildlife with similar habitat requirements would stand to benefit from this recovery strategy. Further project-specific environmental assessments of actions identified as a result of research conducted in this recovery strategy, may be required.
The potential for important negative effects from this recovery strategy on other species or ecological processes is negligible.
Carita Bergman, Parks Canada Agency
Frank Doyle, Wildlife Dynamics
Sue McDonald, Western Forest Products
Sean Muise, B.C. Ministry of Forests and Range
Nick Reynolds, Haida Nation
Ross Vennesland, Parks Canada Agency
Louise Waterhouse, B.C.Ministry of Forests and Range
Berry Wijdeven, B.C. Ministry of Environment (Chair)
Jeff Brown, B.C. Ministry of Environment
Carmen Holschuh, WRGWestland Resource Group Inc.
Lana Wilhelm, Haida Forest Guardians