Protecting Canadians from Excessive Drug Prices: Consulting on Proposed Amendments to the Patented Medicines Regulations

Download the alternative format
(PDF format, 1.17 MB, 16 pages)
Organization: Health Canada
Type: Consultation
Date published: 2017-05-16
Table of Contents
- Introduction
- Executive summary of the regulatory proposal
- Consultation details
- Origin and purpose of the PMPRB’s regulatory mandate
- The PMPRB’s regulatory framework
- Canadian drug prices
- Limitations of the current framework
- Moving to a risk-based approach to regulation
- Proposed amendments to the Regulations
- Introducing new factors to help determine whether a price is excessive
- Amending the list of countries used for international price comparisons
- Reducing regulatory burden for generic drugs with a patent
- Modernizing reporting requirements for patentees
- Providing information related to third party rebates
- Submitting Comments to Health Canada
Introduction
Prescription drugs are an increasingly important part of our health care system, helping prevent and cure disease as well as save lives. However, Canada’s drug spending, which is high by international standards, is not producing all of the benefits it should.
Drug spending in Canada has increased from less than 10% of total health expenditure, when Medicare was first established, to about 16% today. Drugs are now the second-largest category of spending in health care, ahead of physician services with per capita spending on drugs second only to the United States. Canada is paying higher prices for prescription drugs than most other developed countries which can result in limited access to innovative medicines, place a financial burden on patients, and mean fewer resources for other critical areas of the health care system.
In January 2016, federal, provincial and territorial Ministers agreed to work together to improve the affordability, accessibility and appropriate use of prescription drugs to better meet health care system needs. The Government of Canada is firmly committed to this work and is taking action to significantly lower the cost of prescription drugs; provide faster access to new drugs that are safe and effective and better meet the needs of the health care system; and support the development of tools for more appropriate prescribing. To support these actions, Budget 2017 outlined an investment of $140.3 million over 5 years, starting in 2017, and $18.2 million ongoing.
This important work includes reducing the cost of patented drugs through the modernization of the pricing framework under the Patented Medicine Prices Review Board (PMRPB). Under the authority of the Patent Act and through the Patented Medicines Regulations (“Regulations”), the Government of Canada regulates the prices of patented medicines in Canada to ensure that prices are not excessive. Despite significant changes in the pharmaceutical market, the regulations have not been substantively changed in over two decades. Health Canada is consulting stakeholders and interested members of the public on proposed amendments to the Regulations, to equip the PMPRB with more relevant and effective regulatory tools to better protect Canadians from excessive prices for patented drugs.
Executive summary of the regulatory proposal
In the last twenty years, the global environment for pharmaceuticals has significantly changed. Two such changes are of particular relevance in this context: 1) the emergence of higher cost drugs, such as biologics and genetic therapies, that are putting increasing pressure on drug spending; and 2) a growing discrepancy between public list prices and lower actual market prices due to the increased use of confidential discounts and rebates.
These changes have led other countries to experiment with new forms of cost containment that are more reliant on assessing the economic value of a new drug to their respective health systems and less on comparing drug prices internationally. The lack of corresponding reform in Canada has contributed to rising Canadian patented drug prices which are among the highest in the world as well as relatively high pharmaceutical spending per capita and as a proportion of GDP.
The amendments that are proposed in this consultation document are intended to reverse these trends by moving to a modern, risk-based approach to drug price regulation.
Five important improvements to the Regulations are proposed that would:
- Introduce new, economics-based price regulation factors that would ensure prices reflect Canada’s willingness and ability-to-pay for drugs that provide demonstrably better health outcomes;
- Update the list of countries used for price comparison so that it is more aligned with the PMPRB’s consumer protection mandate and median OECD prices;
- Formalize a move to a complaints-based system of oversight for patented generics products that are at lower risk of excessive pricing, reducing regulatory burden for patentees;
- Set out the pricing information required of patentees to enable the PMPRB to operationalize the new pricing factors; and
- Require patentees to provide the PMPRB with third party information related to rebates and discounts on domestic prices.
Based on international best practices, the proposed amendments would provide the PMPRB with new regulatory tools and information to better protect Canadian consumers from excessive prices while reducing regulatory burden on patentees.
Consultation details
Health Canada is seeking feedback from provinces and territories, consumer groups, private payers, the pharmaceutical industry, other stakeholders and any interested members of the public on proposed amendments to the Regulations, prior to their pre-publication in Part I of the Canada Gazette (“CGI”).
This consultation builds on the written submissions received by the PMPRB in response to its June 2016 discussion paper on Guidelines modernization. Feedback from stakeholders suggests:
- The PMPRB has a relevant role to play in Canada’s pharmaceutical ecosystem;
- There is a need for greater collaboration and coordination between and among players within that system;
- Not all patented drugs should be subject to the same level of regulatory oversight;
- Systems which recognize and reward therapeutic value encourage the “right kind” of innovation;
- Affordability and sustainability considerations fall within the PMPRB’s regulatory purview;
- The price review process should be more responsive to changes in science and market conditions; and
- To the extent possible, the PMPRB should apply “bright line” rules that are consistent with international best practices and provide predictability to stakeholders.
Stakeholder feedback on the regulatory amendments outlined in this paper will allow for refinement of the regulatory proposal prior to pre-publication and will be reflected in the Regulatory Impact Analysis Statement (RIAS) posted at that time. All interested parties will have a further opportunity to provide input during the 75-day consultation period during CGI pre-publication.
This consultation period is 6 weeks, running from May 16, 2017 to June 28, 2017.
Further consultations on the PMPRB Compendium of Policies, Guidelines and Procedures will follow from the regulatory amendments.
Origin and purpose of the PMPRB’s regulatory mandate
The PMPRB was created in 1987 as the consumer protection “pillar” of a major set of reforms to the Patent Act (Act), which were designed to encourage greater investment in pharmaceutical research and development (“R&D”) in Canada through stronger patent protection.
Consumer Protection
The Act sets out the period of time that patentees of a drug are provided the exclusive rights granted by a patent. The PMPRB’s regulatory mandate is to ensure that patentees do not abuse their patent rights by charging consumers excessive prices during this statutory monopoly period.
In a monopoly situation, a seller with market power can set a higher price than would exist if the product were subject to competition. The risk of excessive pricing is greater when demand for the product is high and there are few, if any, substitutes. This is especially true for drugs that are first of their kind, or for which alternatives are less effective or have less tolerable side effects.
In the patented pharmaceutical sector, the impact of excessive pricing is that consumers are unable to afford the products they need to survive or thrive. The PMPRB’s existence as the only sector-specific regulator under the Act is attributable to this fact and a recognition by policy makers that the unfettered ability to set a price due to patent rights over pharmaceutical products is not in the public interest.
It is recognized that a balance must be struck. There is a need to encourage pharmaceutical innovation by providing patentees with a period of market exclusivity to recoup their investment and turn a profit. At the same time, it is important to ensure that prices charged during that exclusivity period are not so high as to result in limited access to needed medicines. The PMPRB’s role in that balance is to identify and remedy instances of excessive pricing that might otherwise have that effect.
However, the PMPRB’s current regulatory framework does not provide it with adequate tools to effectively protect Canadians from excessive prices, or for optimal price setting in today’s pharmaceutical environment. That is why Health Canada is advancing the proposed amendments for consultation.
The PMPRB’s regulatory framework
The PMPRB’s legal authority to regulate patented drug prices is set out in the Act and the Regulations, and supplemented by the Compendium of Policies, Guidelines and Procedures (“Guidelines”), which explain the policies and procedures normally undertaken in determining whether a price appears to be excessive.
The Patent Act
The Minister of Health is responsible for those sections of the Act (sections 79 to 103) that pertain to the PMPRB. Sections 79 to 103 enable the PMPRB to take remedial action where it finds the patentee of a drug is charging an excessive price. Although no definition is provided for the term “excessive”, section 85 of the Act identifies factors that the PMPRB must take into consideration when evaluating whether a price is excessive. These are:
- The prices at which the same drug has been sold in the relevant market;
- The prices at which other drugs in the same therapeutic class have been sold in the relevant market;
- The prices at which the drug and other drugs in the same therapeutic class have been sold in countries other than Canada; and
- Changes in the Consumer Price Index (“CPI”).
If after considering the above factors, the PMPRB is unable to determine if a price is excessive, section 85 states that the costs of making and marketing the drug may be considered. The Act also provides authority for other factors to be prescribed in regulation.
The Patented Medicines Regulations
The Regulations specify the information that patentees must provide the PMPRB for it to carry out its regulatory mandate effectively. They include requirements relating to the prices of all patented drugs sold in Canada and their prices in foreign countries where they are also sold. The Regulations specify which countries patentees are required to submit pricing information. Currently there are seven countries set out in the Schedule to the Regulations (the PMPRB7): the United States, the United Kingdom, France, Germany, Switzerland, Italy and Sweden. Although section 85 of the Act allows for further excessive pricing factors to be prescribed in the Regulations, none have been proposed for consideration until now.
The Compendium of Policies, Guidelines and Procedures
Many of the core regulatory concepts in the Act and Regulations have been further developed in the Guidelines, which the PMPRB is authorized to make under section 96 of the Act, subject to consultation with relevant stakeholders. The purpose of the Guidelines is to establish and ensure that patentees are generally aware of the policies and procedures normally undertaken in determining whether a price appears to be excessive.
How the current framework works
Under the PMPRB’s current framework, new patented drugs are assessed for the degree of therapeutic benefit they provide relative to existing drugs on the market and, depending on the outcome of that process, are assigned a ceiling price that is based either on the median price of that same drug in the PMPRB7 countries, the highest priced drug in Canada in the same therapeutic class, or some combination of the two. Once a drug’s introductory ceiling price is set and it enters the market, the regulatory framework allows annual price increases in keeping with CPI, provided these increases do not result in the Canadian price becoming greater than the highest price of the same drug among the PMPRB7 countries.
Canadian drug prices
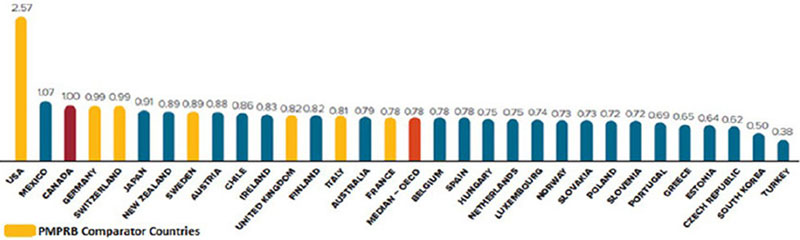
Canadian patented drug prices are among the highest in the world. Among all 35 Organization for Economic Co-operation and Development (OECD) member countries, only the United States and Mexico have higher patented drug prices than Canada. In 2015, median OECD prices for patented drugs were on average 22% below those in Canada, as seen in Figure A.
Figure A. Average Foreign-to-Canadian Price Ratios, Patented Drugs, OECD, 2015
-
Figure A - Long Description
Average Foreign-to-Canadian Price Ratios, Patented Drugs, OECD, 2015 Country Patented drugs prices (percentage) Average (median) percentage of all OECD countries 0.78 United States (PMPRB comparator country) 2.57 Mexico 1.07 Canada (PMPRB comparator country) 1.00 Germany(PMPRB comparator country) 0.99 Switzerland(PMPRB comparator country) 0.99 Japan 0.91 New Zealand 0.89 Sweden(PMPRB comparator country) 0.89 Austria 0.88 Chile 0.86 Ireland 0.83 United Kingdom (PMPRB comparator country) 0.82 Finland 0.82 Italy (PMPRB comparator country) 0.81 Australia 0.79 France (PMPRB comparator country) 0.78 Belgium 0.78 Spain 0.78 Hungary 0.75 Netherlands 0.75 Luxembourg 0.74 Norway 0.73 Slovakia 0.73 Poland 0.72 Slovenia 0.72 Portugal 0.69 Greece 0.65 Estonia 0.64 Czech Republic 0.62 South Korea 0.50 Turkey 0.38
Source: PMPRB 2015 Annual Report
In addition to paying high relative prices, all payers in Canada are struggling with a dramatic increase in the number of high costs drugs on the market. In 2005, there were 20 drugs on the Canadian market with an annual average cost per patient of $10,000 or more. By 2015, that number grew more than five-fold, to 124. That number represents nearly one-quarter of public and private drug plan costs but less than 1% of their active beneficiaries.Footnote 1
Limitations of the current framework
Limitations in the current price regulation framework prevent the PMPRB from effectively regulating high patented drug prices in Canada.
Under the current framework, patented prescription drugs are subject to the same degree of regulatory scrutiny and excessivity is assessed almost entirely on the basis of domestic and international public list prices. This is problematic in an era marked by an influx in high cost specialty drugs and list prices which bear less and less resemblance to what public and private insurers are paying in the market.
The main limitations to the regulatory framework are the following:
- The framework does not provide additional tools, beyond price comparisons and CPI, for the PMPRB to assess whether a price is excessive. It does not consider whether the price of a drug reflects its value to patients or other relevant factors that influence drug prices in different markets such as market size or the relative wealth of a country;
- Canadian prices for new drugs are pegged to countries with high drug prices. Moreover, under the Guidelines, once a new drug enters the market, prices can remain high as long as they do not increase by more than CPI and the highest international price; and
- The Regulations do not require patentees to provide information to the PMPRB on rebates provided to Canadian customers beyond the first point of sale, despite their widespread use. The absence of this information leaves the PMPRB with a limited understanding of the actual prices paid in the market.
Despite periodic adjustment, the current framework remains grounded in an understanding of the Canadian and global pharmaceutical sector of 1987. Reform is needed so that consumers can be effectively protected from any excessive pricing that could result from the current wave of very high cost patented drugs, and to enable the PMPRB to adapt to the modern day reality of non-transparent pricing.
Moving to a risk-based approach to regulation
A risk-based approach is proposed to guide amendments to the Regulations and the modernization of the PMPRB regulatory framework. Central to this approach is the recognition that patented drugs have differing potential to exert market power and charge excessive prices. This potential is largely shaped by the characteristics of the market for each drug, such as the availability of comparator products and the size of the patient population the drug is used to treat. It is proposed that drugs be evaluated against such characteristics to determine their relative risk of excessive prices. Drugs with higher potential to exert market power would face a higher degree of regulatory scrutiny while drugs with medium or lower risk of excessive prices would face respectively lower oversight.
Proposed amendments to the Regulations
1. Introducing new factors to help determine whether a price is excessive
For the past 30 years, domestic and international price referencing has served as the backbone of the PMPRB’s ceiling setting methodology. In this time, many other developed countries have expanded their approach beyond this practice, given the inherent unreliability of international public list prices.
Alternative best practices include the use of long term value for money thresholds, multi-year budget caps, price volume arrangements, systematic therapeutic class and price reviews, and reimbursement and clinical criteria restrictions. However, even with all these tools, no jurisdiction is immune to excessive monopoly pricing in circumstances where a manufacturer holds a patent for a new breakthrough drug for a life-threatening condition.
Taking into consideration “willingness and ability-to-pay” of payers
The assessment of whether a price is excessive is an economic exercise that examines the factors that impact both supply and demand for a particular product in a given market. On the supply side, if the PMPRB is unable to determine whether a price is excessive based on the section 85 factors, it may consider information on the costs of making and marketing the drug. However, the factors do not account for the demand side of the equation, which is driven in large part by the following:
- The relative value of a drug to a patient (drugs that offer substantial clinical benefits to patients or are alone in their class will be in greater demand than drugs that are only marginally better than the standard of care or are one among many in their class);
- The number of patients that can benefit from a drug (the relative size of the market for a drug can have an impact on its expected price and the ability to pay in a given country); and,
- The relative wealth of a country (those with comparatively greater economic resources can afford more or higher cost drugs than countries with fewer resources).
Proposal #1: It is proposed that the Regulations be amended to include the following three new factors for consideration, under s. 85(1), when determining whether a medicine is being or has been sold at an excessive price:
- The pharmacoeconomic evaluation for the medicine and other medicines in the same therapeutic class in Canada and in countries other than Canada;
- The size of the market for the medicine in Canada and in countries other than Canada; and
- Gross Domestic Product in Canada.
i. New factor: the pharmacoeconomic evaluation for the medicine
A pharmacoeconomic evaluation identifies, measures, and compares the costs and benefits of a given drug to patients and the healthcare system.
A cost utility analysis is a type of pharmacoeconomic evaluation that uses a common unit of measurement, to enable comparisons across different types of drugs and health interventions. In this context, the quality-adjusted life year (QALY) is the established benchmark for quantifying benefits by measuring lengthened life and/or improved quality of life. The cost per QALY measure allows for a comparison between drugs to provide an assessment of opportunity cost, i.e., what one must give up to pay for the drug in question.
The price paid for a medicine should take into consideration the value it produces. At the same time, it must recognize the cost to supply the medicine if pharmaceutical manufacturers are to continue to invest in the production of new drugs.
Other developed countries rely to some degree on cost per QALY in determining whether and how much to pay for a new drug. With the addition of a pharmacoeconomic evaluation factor to the Regulations, the PMPRB could introduce, in Guidelines, the concept of a fixed cost per QALY threshold in Canada. In order to minimize regulatory burden on patentees and uphold a common standard of evidence, the definition of a cost per QALY would be consistent with the economic guidance provided by the Canadian Agency for Drugs and Technology in Health’s (CADTH).
ii. New factor: the size of the market for the medicine in Canada and in countries other than Canada
A drug can be considered cost effective yet still be priced at a level that may result in short term rationing by payers because of the prevalence of the condition it is indicated to treat. For example, where public and private payers are called on to cover the cost of a drug for a significant number of patients, the high cost of a drug could render the drug unaffordable, thereby limiting access to only a subset of the population that needs it or for a limited period of time.
Many other developed countries are grappling with this issue and looking for ways to link cost effectiveness considerations with budget impact thresholds that are informed by estimated market size. These thresholds do not always operate as hard caps but rather as triggers for incorporating market size considerations and estimated patient uptake over a particular time horizon into commercial arrangements with manufacturers.
Since monopolies are protected from new entrants, prices tend to remain unaffected from subsequent fluctuations in market size. Seeing that firms are assumed to set their introductory prices at a profitable level to recoup initial investment, a subsequent exponential growth in the market size should align and correct prices downwards to a comparable level. Failure to do so could suggest that the original price, for an expanded market, is now excessive.
The addition of this factor in the Regulations could enable the PMPRB to develop market impact tests for drugs that are likely to pose affordability challenges for payers due to their market size. As the pharmaceutical industry operates in a global context, the Canadian price could be assessed against international prices and prevalence levels in an effort to evaluate the price-volume relationship.
iii. New factor: Gross Domestic Product in Canada
Gross Domestic Product (GDP) is a measure of a country’s economic output. GDP growth measures how much the inflation adjusted market value of the goods and services produced by an economy is increasing over time. Per capita GDP measures how much a country is producing relative to its population. In this context, growth in Canadian GDP can be taken as indicator of the country’s ability to pay year-over-year, whereas per capita GDP is a proxy for buying power at the level of the individual. Both can serve as analytically sound ways to assess a drug’s affordability, as a marker for potentially excessive prices.
Consultation questions:
- Do you agree that a pharmacoeconomic evaluation is an important factor for the PMPRB to consider when determining whether a drug is priced excessively? If so, how should the evaluation be considered?
- Do you agree that the size of the market for the drug in Canada and other countries is an important factor for the PMPRB to consider when determining whether a drug is priced excessively? If so, how should the size of the market be considered?
- Do you agree that Canada’s GDP and GDP growth are important for the PMPRB to consider when determining whether a drug is priced excessively? If so, how should GDP be considered?
- Are there any other factors that should be considered by the PMPRB when determining whether a drug is priced excessively? How should the factor(s) be considered and what information should be required from patentees?
2. Amending the list of countries used for international price comparisons
In determining whether a medicine is being or has been sold at an excessive price in Canada, Section 85 of the Act directs the Board to take into consideration the prices at which a medicine or other medicines in the same therapeutic class have been sold in other countries. The Act and Regulations also require patentees to submit to the Board the “ex-factory” priceFootnote 2 of the medicine in the PMPRB7 countries set out in the Schedule to the Regulations.
The Board uses the public ex-factory prices of the same patented drug products in these countries, where available, to set price ceilings on drug prices in Canada at introduction and in subsequent years. Therefore, the selection of countries can have a significant impact on the price ceilings for patented drugs in Canada.
Origin of the list of countries in the Schedule
When the Regulations were first conceived 30 years ago, policy makers believed that patent protection and price were key drivers of pharmaceutical R&D investment. The choice was thus made to offer a comparable level of patent protection and pricing for drugs as exists in countries with a strong pharmaceutical industry presence, on the assumption that Canada would come to enjoy comparable levels of R&D. However, the percentage of R&D-to-sales by pharmaceutical patentees in Canada has been falling since the late 1990s and is at a historic low. By comparison, and despite Canada having among the highest patented drug prices, industry R&D investment relative to sales in the PMPRB7 countries is on average 22.8%Footnote 3 versus 4.4%Footnote 4 in Canada.
There is no evidence of a determinant link between domestic prices and the location of industry R&D investment. Other factors, such as head office location, clinical trials infrastructure and scientific clusters, appear to be much more influential determinants of where pharmaceutical investment takes place in a global economy. The policy intent of the original Schedule selection has not materialized and is no longer considered to be the most appropriate basis for the composition of Canada’s international price reference Schedule.
Modernizing the Schedule
The proposed amendments to the Regulations would reconstitute the Schedule with countries that are more aligned with Canada economically and from a consumer protection standpoint. The number of countries would also be increased to make the benchmarking process less sensitive to missing or skewed price data from individual countries, but not to the point of imposing undue regulatory burden on patentees.
Three main criteria have been used to select the countries:
- Consumer protection: whether the country has national pricing containment measures in place to protect consumers from high drug prices;
- Economic Standing: whether the country has a similar economic standing to Canada, as measured by GDP per capita; and
- Pharmaceutical market characteristics: whether the country has similar market characteristics to Canada, such as population, consumption, revenues and market entry of new products.
| Current Schedule | Proposed New Schedule |
|---|---|
|
|
| * New countries added to the list | |
Consultation questions:
- Are there other countries that should be considered in revising the Schedule?
- Are there other criteria that should be considered in revising the Schedule?
- Please provide any other comments you may have on the Schedule of comparator countries.
3. Reducing regulatory burden for generic drugs with a patent
A third proposed amendment to the Regulations would remove the requirement for patentees of generic drugs (i.e. drugs that have been authorized for sale by Health Canada through an Abbreviated New Drug Submission (ANDS) and received a Declaration of Equivalence to a Canadian Reference Product) to systematically report information pertaining to the identity and price of these drugs to the PMPRB. Going forward, the Regulations would only require identity and price information in the event of a pricing complaint about a particular patented generic drug or at the request of the PMPRB.
This proposal would be consistent with an increased PMPRB focus on drugs that are at greatest risk of excessive pricing due to the degree of market power held by the patentee. It would spare patentees unnecessary regulatory burden. Patentees of generic drugs typically face greater competition and the risk of excessive pricing due to market power is generally not cause for concern.
Proposal #3: It is proposed that the Regulations be amended to set out that patented generic drugs, which received market authorization from Health Canada through an Abbreviated New Drug Submission, be required only to report identity and price information in the event of a complaint or at the request of the Patented Medicines Prices Review Board.
Consultation question:
- Do you agree that patentees of generic drugs, i.e. drugs that have been authorized for sale by Health Canada through an ANDS should only report information about the identity of the drug and its pricein the event of a complaint or at the request of PMPRB?
4. Modernizing reporting requirements for patentees
The Regulations specify what information is to be provided to the PMPRB by patentees relating to the prices of patented drugs sold in Canada and other countries, patentees’ revenues as well as R&D expenditures.
Information to assess new factors
It is proposed that the Regulations be amended to require patentees to provide the information required by the PMPRB to operationalize the new factors, as well as information to account for the confidential rebates and discounts provided to third party payers.
Proposal #4: It is proposed that the Regulations be amended to set out the information required to enable the PMPRB to consider the new factors as proposed:
- For the new factor – the pharmacoeconomic evaluation for the medicine and other medicines in the same therapeutic class in Canada and in countries other than Canada – the Regulations would be amended to require patentees to submit:
- the cost utility analysis by approved indication of the medicine, where that information is available to the patentee
- For the new factor – the size of the market for the medicine in Canada and in countries other than Canada – the Regulations would be amended to require the patentee to submit:
- the estimated uptake of the medicine, by approved indication, in Canada without restraint on utilization (e.g. market/budget impact analysis in any relevant market), where that information is available to the patentee
No information would be required from patentees on per capita GDP or growth in GDP.
Consultation questions:
- Is the information sought in relation the new factors relevant and sufficient?
- Is this information generally available to patentees?
5. Providing information related to third party rebates
The Regulations currently require patentees to provide the PMPRB with information on the price of a drug sold to different types of customers (e.g. wholesalers, pharmacy chains). While patentees are required to report reductions in prices through rebates and discounts, this information is only collected by the PMPRB for the first point of sale.
Patentees are not required to report the significant discounts and rebates they provide to third party payers such as provincial drug plans, which are their biggest customer. Without this information, the PMPRB is left to set its domestic price ceilings on the basis of information that only includes list prices and does not reflect the actual prices paid in the market.
This information would be considered privileged as per section 87 of the Patent Act and would be taken into consideration by PMPRB when determining whether a patentee is compliant with ceilings set to determine price excessivity.
Proposal #5: It is proposed that the Regulations be amended to require patentees to report to the PMPRB all indirect price reductions, given as a promotion or in the form of rebates, discounts, refunds, free goods, free services, gifts or any other benefit in Canada.
Consultation questions:
- Are there any reasons why patentees should not be required to disclose to the PMPRB information on indirect discounts and rebates provided to third party payers?
Submitting Comments to Health Canada
By Email: Please send your responses to the consultation questions to:
PMR-Consultations-RMB@hc-sc.gc.ca
By Mail: Please send your responses to the consultation questions to:
Attention: Patented Medicines Regulations Consultations
70 Colombine Driveway, Tunney's Pasture
Mail Stop 0910, Floor 10, Building Brooke Claxton Building
Ottawa, Ontario K1A 0K9
Canada
Please do not include any personal information when providing feedback to Health Canada. The Department will not be retaining your email address or contact information when receiving your feedback and will only retain the comments you provide.
Feedback received on or before June 28th, 2017 will be considered in developing a regulatory proposal under the Patent Act to protect Canadians from excessive drug prices. A summary of comments will be published in the RIAS statement at Canada Gazette, Part I.
Thank you for taking the time to consider these proposals and to contribute to further protecting the health of Canadians.