Committee report – February 5 and 6, 2020
Chemicals Management Plan Science Committee
Considerations for identifying potential risks from exposure to chemicals in the workplace
On this page
Introduction
Throughout the Chemicals Management Plan (CMP), stakeholders have often identified occupational exposure as a gap in risk assessments under the Canadian Environmental Protection Act, 1999 (CEPA 1999), as the assessments focus only on the risks to the environment and the general population. This limited focus was also identified as an area of misalignment internationally through the United States (U.S.)-Canada Regulatory Cooperation Council (RCC) Chemicals Management project and through several program evaluations of the CMP. In 2019, United Nations (UN) Special Rapporteur on toxics and human rights Baskut Tuncak undertook an official country visit to Canada. He expressed concern that workers are currently not considered as a vulnerable class under the CMP (UNHR 2019). This concern further stimulated Health Canada (HC) to continue exploring ways to reduce the risk to Canadians from chemical exposure by considering workplace exposures and enhancing worker protections. One way that this could be accomplished is by using information, tools and expertise of the CMP. To discuss these opportunities, a meeting of the CMP Science Committee (SC) was held in Ottawa on February 5 and 6, 2020.
Prior to the February meeting, the SC heard presentations from the Government of Canada, the European Chemicals Agency (ECHA), the U.S. Environmental Protection Agency (EPA), and the Australian National Industrial Chemicals Notification and Assessment Scheme (NICNAS). The Ottawa meeting opened with presentations by the Government of Canada and then heard several presentations from members of the SC.
This report summarizes the discussions held by the SC during this 2-day meeting. The SC's discussions were guided by 2 charge questions presented by the Government in a background document entitled "Considerations for identifying potential risks from exposure to chemicals in the workplace."
Overall, the SC identified the movement of CMP into the occupational area as a great opportunity to achieve the goal stated above. Many non-federal initiatives across multiple sectors and jurisdictions in Canada have been active over the years in gathering and analyzing information aimed at protecting workers from occupational chemical exposure risks. However, most such initiatives have been challenged by inconsistent and often diminished resources. The lack of resources extends to research funds available to investigate workplace health and safety. Changes at provincial and territorial levels, such as shifting responsibility for workplace monitoring from public sector organizations to employers, have created an additional challenge of reducing publicly available data (Hall et al. 2014). Thus, the CMP's involvement presents an opportunity to learn from past lessons, integrate knowledge country-wide and possibly stimulate funding initiatives to sustain and develop such initiatives.
Charge Question 1
Considering what was presented at the international government science forum at the pre-meeting webinars:
- What are the key scientific lessons learned from international agencies?
- What considerations does the SC recommend as HC explores ways to contribute to the protection of workers from exposure to chemicals in Canada?
The SC discussed Charge Question 1 covering a number of specific topics that were also outlined in the discussion paper.
1.1 Hazard classification
The SC discussed that availability of reliable information in the supply chain is fundamental to the protection of workers. A key piece of information, instrumental for the safe use of chemicals, is information on potential chemical hazards, specifically the Globally Harmonised System (GHS) classification that are found on the labels and safety data sheets (SDS) for hazardous workplace products (CCOHS 2020a). However, Canada lacks a centralized repository of classification information. The SC agreed that an active role for HC, possibly together with Environment and Climate Change Canada (ECCC), in making reliable hazard classification information centrally available would substantially improve the safe use of chemicals in the workplace and throughout supply chains in Canada. A policy decision would likely be required to determine how GHS classification would be obtained/provided, how these classifications would be made publicly available (for example, including a quality check by authorities), and how this information could be used in the regulatory process, including risk management decisions.
In making information more available, Canada could learn from other domestic and international organizations that publish classification information under the GHS. The SC discussed taking approaches similar to those of the European Union (EU) and Australia's NICNAS, of collating and making hazard data broadly available, specifically classification information. Australia's approach involves the formal recognition of hazard conclusions done in other jurisdictions, specified in detail in their legal framework.
Until now, HC has classified and made publicly available only a fraction of the chemicals currently in use in Canadian workplaces, and has done so on an ad hoc basis. Classifying chemicals in the workplace could be intensified and done in a more systematic manner, making more hazard classifications publicly available in a centralized repository.
HC should avail itself of information gathered by other jurisdictions, including knowledge of gaps in chemical classification. Such gaps could be filled by the use of, for example, read-acrossFootnote 1 or other New Approach Methodologies (NAMs)Footnote 2 and other available sources such as the Danish QSAR Database and the U.S. EPA's CompTox Chemicals Dashboard. Making such information publicly available in a centralized, easy-to-access fashion will aid the creation of high quality SDSs.
1.2 Supply chain
The presentation by ECHA to the SC highlighted the European approach that aims for a full understanding of the supply chain of chemicals used in different professional/occupational settings, from production to the waste stage. Generally speaking, chemicals are manufactured and supplied through a supply chain that can contain multiple steps (for example, preparation of mixtures), until the point that the chemical is used in a product (for example, either as a mixture or an article). This product might find its use in an occupational setting. Under REACH in the EU, the manufacturer or importer of a chemical should be aware of all these steps, until the chemical ceases to exist in a legal sense (reacts into something else, or becomes waste). ECHA highlighted in their presentation the challenges faced by both industry and authorities with regard to getting a clear picture of all these steps over a chemical's life cycle. Despite the legal requirements as laid down in EU REACH, in practice, getting this information is often very difficult, especially for chemicals that have many uses and at high total volume.
The SC discussed that getting clarity on the uses of the chemicals through the supply chain can be challenging within the current Canadian legislative framework, especially for existing chemicals. It can be expected that for new chemicals, notifications will usually be for specific or limited uses of a chemical in a relatively limited supply chain. These uses are assumed to be fewer in number and less diverse than for existing chemicals. Also, the new chemical notification scheme has options that allow for a better understanding of the uses of the chemical. Data available for new chemicals could presumably allow government officials to understand how a new chemical could result in various occupational exposures under the CMP.
In contrast, existing chemicals on the Canadian market can have more diverse uses and complex supply chains than new chemicals. Provisions do not include a systematic and periodic reporting scheme keeping volumes and uses up to date, unlike the U.S. Chemical Data Reporting (CDR) approach in which the U.S. EPA collects basic exposure-related information, including information on the types, quantities and uses of chemical substances produced domestically and imported into the U.S. every 4 years.
In Canada, information on the uses of existing chemicals can be collected through provisions of sections 46 and 71 of CEPA 1999. Information on new chemicals that are not on Canada's Domestic Substance List (DSL) can be collected by means of Significant New Activity (SNAc) provisions. The limited information available and impediments to gathering further information on existing chemical uses create a number of complexities and limitations. For example, the SC discussed the case in which substances, both on the DSL and new substances, are marketed in preparations that fall beneath the reporting threshold for the SDS, with the consequence that they are often not declared on the SDS, and hence users are unaware that such chemicals are present in the products used.
The SC recognized that interpreting and using the hazard and safety information contained in an SDS could be complex in terms of using the information to apply risk management measures on-site. To address the challenges posed by the use of hazard and safety information, it was suggested that HC could play a coordinating role in delivering training to improve employers' and workers' understanding of SDS information. Such training could include interpreting hazard information, implementing engineering controls, exposure mitigation measures, use of appropriate personal protective equipment, worker safety education, and hazard and risk communication. Improved understanding of SDS could also aid in developing new SDS in cases where chemicals are used as raw materials to make other chemical products.
For employers to protect workers, they must consider the complex mixtures to which workers are exposed, including mixtures of substances in products and not just the main ingredients that may or may not be disclosed. The SC discussed the issue of confidential business information (CBI). As HC notes, "A supplier or employer who wants to protect CBI must file a claim for exemption with Health Canada." The CBI process includes an HC review of the SDS and/or label to verify that the hazard and safe use information complies with Hazardous Products Act and the Hazardous Materials Information Review Act requirements. This mechanism aims to balance workers' right to know with industry's need to protect trade secrets (CCOHS 2020b). The SC also noted that while governments may have full information on hazardous chemical constituents in the products as they are used in the supply chain, CBI claims may limit disclosure of hazardous chemicals to the user.
To overcome obstacles related to the disclosure and understanding of chemicals present in products, the SC suggested leveraging information from other jurisdictions and considering adapting the modelling work done on chemical use by the U.S. EPAFootnote 3 (Dionisio et al. 2018, Isaacs et al. 2020, Williams et al. 2017). Another suggestion was to invest in compositional product testing to gain knowledge of the chemicals present in products used in occupational settings. Such testing and analysis should not only consider the downstream use of the product, but should also consider upstream, occupationally relevant activities which could result in exposures throughout the chemical's life cycle, for example, during chemical manufacturing and formulation before the chemical enters the product/article through to end-of-life (see section 2.3).
The SC raised the issue of products available to consumers not intended for occupational use but that are used in occupational settings. This issue was raised to illustrate 1 of the implications of the lack of full disclosure of chemicals in such products. According to the Consumer Packaging and Labelling Act, "right-to-know" labelling and information on chemical ingredients in consumer chemical products (not including cosmetics) is not mandatory for products available to consumers, resulting in an information gap for occupational exposure (Government of Canada 2012, 2015a). Disclosure of hazardous ingredients that are present in the hazardous products at greater than 1% (Government of Canada 2015b) and used in the workplace (as defined by the Hazardous Products Act) is required. In contrast, Ontario's Occupational Health and Safety Act gives workers the right to know about hazards they may encounter in the workplace, through SDSs as one example (Government of Ontario 2012). Such concerns are mitigated in part by the Canadian Consumer Chemicals and Containers Regulations, 2001 under the Canada Consumer Product Safety Act (CCPSA). The Regulations outline requirements for identification of hazards in consumer chemical products with respect to toxicity, flammability, corrosiveness and quick skin-bonding adhesives. The regulations use scientifically derived classification criteria to dictate a chemical product's classification and determine labelling and packaging requirements, "including hazard symbols, warning statements, safety instructions and first aid statements." (Government of Canada 2015b). As noted, while such labelling exists, only the hazardous ingredients that contribute to the classification results and that are present at a concentration of 1% or greater are required to be disclosed, rather than the full ingredient list.
1.3 Monitoring
Presentations by the U.S. EPA and ECHA highlighted the value of good-quality monitoring data for their chemicals management programs. The U.S. EPA makes use of data collected by the National Institute for Occupational Safety and Health (NIOSH) and Occupational Safety and Health Administration (OSHA). ECHA uses monitoring data combined with modelled data to determine risk management measures for substances of high concern in occupational settings.
It was noted that Canada lacks a coordinated program for workplace exposure monitoring. Monitoring has been conducted through specific research activities, but these activities are not ongoing or necessarily coordinated (Davies 2019, Hall et al. 2014). Also, occupational health and safety regulators do not currently have a mechanism to influence Health Canada research and monitoring projects. A specific program to nationally coordinate and fund projects related to occupational exposure or effects of chemicals used in the workplace would add significant information to the management of chemicals in Canada.
In terms of monitoring, cancer has been the health effect that has received the most attention. The SC heard that effects monitoring studies that go beyond cancer should be considered to include other effects such as sensitization, dermatitis, and reproductive and neurotoxicological effects.
The SC noted that monitoring data could be used to develop and evaluate models specific to workers' exposure. The SC saw the benefits of promoting research to support the development and use of exposure models that would allow estimates to be made for activity-specific uses and processes (for example, spray painting).
1.4 Prioritization
The SC learned that the U.S. EPA, NICNAS and ECHA consider occupational exposure as part of an integrated prioritization scheme for risk evaluation and, where warranted, risk management. Specific activities for the occupational use of chemicals are integrated within the purview of these agencies, as occupational uses have historically fallen within the scope of their mandates. The SC heard that ECHA recently began to undertake a separate prioritization exercise for Occupational Exposure Limits (OELs), for which data are not taken from the registration dossiers held by ECHA. The specific topic of prioritization and directions that HC might pursue was discussed in more detail in the context of Charge Question 2b (see section 2).
1.5 Risk assessment and risk management
Occupational exposure, including during manufacturing and use stages, is an integrated part of the authority risk assessment and risk management activities conducted by other jurisdictions outside of Canada. Indeed, risk management measures often relate to occupational safety due to elevated exposures in occupational settings. The SC discussed the benefit of taking such an integrated approach.
Within more specific discussions, a number of suggestions were made:
- To rationalize efforts, government officials could explore opportunities for taking an approach using exposure and hazard bands or categories under the CMP, as done by Australia's NICNAS rather than setting explicit limits on a chemical-by-chemical basis. HC could play a role in developing ways to categorize chemicals into bands, possibly based on chemical classification without setting OELs.
- Occupational uses, including factors such as the size of the workforce, should be included as a factor to prioritize chemical risk assessment and risk management. This could require additional information, since occupational uses of a chemical/product are not always known or intended by a product manufacturer. For example, the SC noted the use of some consumer products in occupational settings (for example, consumer nail products used by workers in nail salons, consumer cleaning products used by cleaners).
- An iterative approach could be used from prioritization to risk management, starting with exposure modelling and moving towards measurement and more data collection if warranted. The challenge is the course of action in case of knowledge and data gaps, both in hazard and in use and subsequent exposure.
1.6 Occupational Exposure Limits (OEL) development and use
The SC discussed this topic at some length. In terms of "lessons learned" from other jurisdictions, among the organizations that presented in the pre-meeting webinars, only ECHA develops OELs. Their use within the European community involves a relatively complex system of pan-European binding and indicative limits. In addition, most member states have "local" OELs, often themselves having multiple lists (binding and indicative). The complexity of this system is a consequence of the historical development of different laws in parallel at different levels (Member States versus a pan-European approach). In addition, the introduction of REACH created a partial overlap and therefore complications in terms of implementation. Within REACH, registrants are obliged to set Derived No Effect Levels (DNEL) that function similarly to OELs as limit values. It is unclear which limits should be used in cases where there are both a DNEL and an OEL, or even multiple DNELs and OELs.
The SC discussed the difference between using a purely health-based, scientifically derived limit versus the use of limits that give additional consideration to technical feasibility and socio-economic aspects. It was suggested that HC could disseminate health-based "Occupational Exposure Guidelines" as advisory guidelines as opposed to strict implementation of OELs that would incorporate technical feasibility and socio-economic considerations. Any such guideline would require appropriate scientific derivation and transparency and, importantly, a well-thought-out communications plan.
Charge Question 2a
Given the summary of types of data and sources identified below that could inform a risk-based prioritization exercise like the Identification of Risk Assessment Priorities (IRAP), is anything missing? Comment on the importance and relevance of the various types of data and sources (for example, relevance of total quantities used in workplaces across Canada, number of workers, work tasks; relevance of changes to/new hazard classifications)
Main sources of information
Figure 1 provides a summary of 3 main feeders of information discussed by the SC. These are discussed below along with additional considerations.

Figure 1: Text description
Figure 1 depicts a Venn diagram of three intersecting sources of information for prioritizing chemicals for occupational concern. These include CMP evaluations, workplace prioritization information, and high-throughput modelling.
2.1 Chemicals Management Plan (CMP) evaluations
To identify information that could inform a risk-based prioritization for occupational settings, the SC considered sources used in developing existing CMP evaluations and identified several sources of information that could be consulted:
- Chemicals listed as CEPA toxic on Schedule 1 where 1 question is whether existing risk management measures are sufficient to be protective in the occupational setting
- New substances for which SNAc orders and notices have been issued, and again the concern is whether existing risk management measures are adequate for occupational settings
- Information gathered from section 71 requests
The SC noted that in the U.S., Significant New Use Rules (SNURs, similar to SNAcs) are often issued based on considerations of potential occupational health concerns.
CMP evaluations themselves contain a wealth of information that could be used in the occupational health context. However, the SC noted the different exposure circumstances between current CMP versus occupational health considerations. Additionally, CMP takes a chemical-by-chemical approach and does not explicitly consider combined exposures to multiple chemicals that may be of greater concern in occupational settings due to the higher exposure levels as compared to the general population. As well, CMP considerations factor in chemical usage according to volumes but not the number of people who could be affected.
- consider the occupational exposures of those chemicals
- consider the combined exposures in the workplace
- consider the number of workers
- identify priorities more broadly using a public health approach
Indeed, the SC's previous discussion on taking a public health approach to chemical management could provide useful guidance in this instance as there is an intersection between considering occupational and public health, both of which deal with exposures of a wide population to complex mixtures, which can include simultaneous exposures to different substances from different sources.
The SC discussed what additional information could be gathered from section 71 requests. Such information could include data on acute sensitization and genotoxicity that is now reported under the U.S. Toxic Substances Control Act (TSCA). Information could also be requested on how the substance is actually used in the workplace (for example, open versus closed system), the number of workers who might be exposed, and if personal protective equipment is used.
2.2 Information gathered from workplaces
The SC considered multiple sources of information. The SC raised many examples in which few data were available, and when available, the data were outdated or fragmented and/or difficult to access (as noted in section 1.3). In addition to recommending a nationally coordinated program for monitoring, the SC suggested looking to the provinces and territories and their agencies for prioritized occupational health data, since occupational health is primarily under their jurisdiction. However, some (but not all) provinces and territories collect data. WorkSafeBC was raised as an example of an agency holding occupational exposure data. Some SC members cautioned that even if such data exist, they may not be readily available or current. For example, British Columbia and Ontario collect data on incidents and disease surveillance; however, such information was not thought to be useful in this context.
The SC discussed other sources of information that may or may not be useful. The discussion illustrated the fragmentary nature of information on occupational exposure and health in Canada. Some useful information could be gained from occupational health clinics that operate across North America and are mostly run through academic institutions. An example is the Occupational Health Clinics for Ontario Workers (OHCOW), which provides a variety of services to workers, health and safety committees, unions, employers, legal clinics, and members of the public. Poison control centres across the country gather data on acute effects which could be examined for a trend analysis as one example, but again, these data may or may not be useful. Another example is in Ontario, the Occupational Disease Surveillance Program created by the Occupational Cancer Research Centre, based at Ontario Health. It routinely analyzes occupational patterns of disease but lacks occupational exposure data, as do all similar efforts (Jung et al. 2018).
The SC noted that information could be pulled from studies that have been conducted on prioritized sectors. Examples of such studies include exposure of firefighters to polycyclic aromatic hydrocarbons (Keir et al. 2017), and e-waste worker exposure to flame retardants (Nguyen et al. 2019, Gravel et al. 2019, 2020).
Other jurisdictions, such as the U.S., could have useful information in cases where exposure conditions are similar to those in Canada (for example, DTSC 2020). If data were obtained from other jurisdictions, efforts need to be made to either obtain comparable data for Canada or to ensure that data from another jurisdiction are relevant to Canada.
Another source of information is SDSs; the SC discussed their use but also limitations to such use. SDSs are available for existing and known hazardous products and thus cannot be used to prioritize chemicals with unknown hazards. SDSs are not systematically available for consumer products sold in Canada that can be used in occupational situations. Another limitation is simply the effort required to mine this source of information, although this is being done by the U.S. EPA (Dionisio et al. 2018). The SC commented that disclosure of constituents of consumer products could be achieved through the CCPSA, noting that this Act does not provide for disclosure of chemicals occurring at less than 1% of the formulation (section 1.2). The Hazardous Products Act, which outlines supplier requirements to provide information on the safe use of hazardous products in Canadian workplaces, needs to remove the exclusion for consumer products that can be used in occupational settings.
As noted in section 1.3, measurement data would be useful but are often not available or are available for some locations in some years. Gaining access to workplaces where measurement campaigns could be conducted is not always possible (for example, air quality monitoring, biomonitoring). Modelling could be used to fill data gaps, recognizing that data and significant resources are required to parameterize and evaluate such models. Even if measurements are available, the SC commented that modelling such situations would be useful in order to gain additional insights and experience that could be translated to other situations.
An impediment noted with respect to gathering information from industry is the lack of a proactive periodic reporting scheme for basic exposure-related information, including information on the types, quantities and uses of existing chemical substances produced domestically and imported into Canada, as discussed under section 1.2. This translates to significant unknowns and uncertainties with respect to chemical usage and exposure.
The use of Incident Reports (that is, acute effects) was discussed as a source of information by the SC but with a cautionary note that reviewing such reports could be time-consuming and yield minimal information, as they often lack the information to be useful such as specific exposure information. The SC was not aware of examples where the review of Incident Reports yielded useful information. Assessing disease records for chronic effects could be even more labour intensive, with the risk of minimal return on resources invested, as determining causal relationship between disease and exposure to chemicals is very challenging.
2.3 High throughput exposure modelling extended to incorporate a life cycle approach
This modelling approach is a hybrid of material flow analysis, life cycle thinking and risk assessment, as illustrated in figure 2. Occupational exposure can be a concern throughout a chemical's life cycle. For example, occupational exposure can occur during resource extraction, as well as chemical, product and item manufacturing. Occupational exposure can also occur during the use stage with the service sector raised as an example. Finally, occupational exposure can be of concern at the end-of-life stage, either through waste management and/or recycling. Csiszar et al. (2016) and Meyer et al. (2020) have outlined the hybrid modelling approach between life cycle analysis and risk assessment (LCA-RA) that could be modified to consider occupational exposures. Li et al. (2016, 2017, 2020) have also developed mass flow analysis models for chemicals in products and articles that are then linked to fate and exposure and PBTK models for production-to-exposure and dose simulations. These frameworks are well suited for application in occupational settings where the emissions estimates in occupational settings could then be used to parameterize occupational-setting fate and exposure models. Diamond and Kvasnicka (2020) provided a qualitative example of using this approach that includes occupational exposure, based on the case study of polybrominated diphenyl ethers (PBDEs) used in electronics cases. This example considered occupational exposure during the production/manufacturing and end-of-life stages. These approaches could benefit from adding work on Quantitative Structure Use Relationships (QSURs) that consider "functional use" (Phillips et al. 2017). As well, the OECD's published exposure scenarios could be helpful towards extending the High Throughput LCA-RA into the occupational realm.
An advantage of the High Throughput LCA-RA approach is that it can be used to identify priorities for further assessment and for data gathering. Such a system could be used in "batch mode" to consider numerous chemicals at a screening level. Another advantage is that the model could be run for a suite of chemicals that could be of concern in an occupational setting, thus moving away from the limitation of taking a chemical-by-chemical approach. There are clear advantages in adopting this approach for non-occupational exposures as well as the information collected, evaluated and corroborated with mass-flow analysis tools, which can address data gaps on chemical emissions throughout their life-cycle. The limitation of implementing this approach includes the availability of Canada-specific data, as discussed throughout this report.
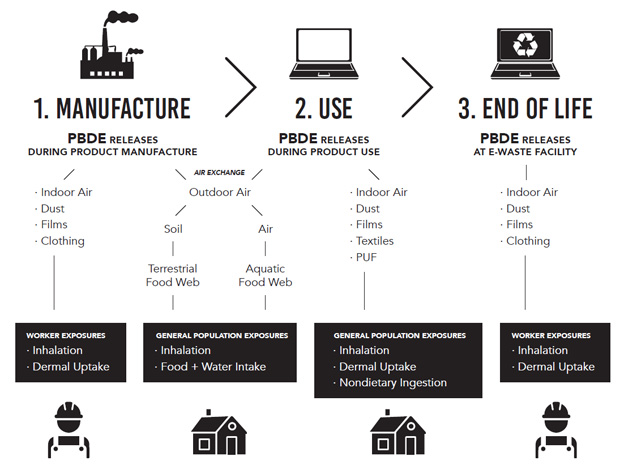
Figure 2: Text description
Figure 2 uses PBDEs in computer cases as an example to illustrate how three lifecycle stages (manufacture, use and end of life) contribute to a risk analysis approach to consider occupational exposures.
The manufacturing stage can result in releases of PBDEs to indoor air, and the presence of PBDEs in dust, films and on clothing; which contribute to worker exposures through inhalation and/or dermal uptake.
Manufacturing and product use may result in releases of PBDEs to outdoor air, where PBDEs may end up in soil, and then the terrestrial food web, or in air, and then the aquatic foodweb. These releases contribute to general population exposures through inhalation and/or food and water intake. Product use may also result in releases of PBDEs to indoor air, and the presence of PBDEs in dust, films, textiles and PUF. These can result in general population exposures through inhalation, dermal uptake and/or nondietary ingestion.
Lastly, in the end of life stage, PBDEs may be released at e-waste facilities to indoor air, and may result in the presence of PBDEs in dust, films and on clothing; which contribute to worker exposures through inhalation and/or dermal uptake.
2.4 Other sources of information
In addition to the sources of information illustrated in figure 1, the SC considered information gained from biomonitoring. The SC discussed how biomonitoring could be used for surveillance purposes. Specifically, non-target analysis could be used to potentially identify a wide range of chemicals to which individuals were exposed, acknowledging the challenge of relating that broad range to the workplace (this challenge exists for targeted screening as well). Several groups are making progress with the use of non-target analysis from biomonitoring sampling, which is easier among highly impacted groups by virtue of the occurrence of higher concentrations in occupational than non-occupational settings. While biomonitoring may be advantageous, obtaining biological samples from occupationally exposed groups can be difficult, especially for non-unionized and potentially vulnerable workers such as those in the service sector or who work as contractors. The SC considered the opportunity to broaden the Canadian Health Measures Survey (CHMS) to include questions regarding work and workplaces.
Applying mass balance exposure models that include toxicokinetics models with calculations for blood and urine concentrations (for example, Li et al. 2018) with occupational monitoring (for example, air, dust) and biomonitoring data (for example, hand-wipes, urine) provides an opportunity to build and test exposure modelling capacity while concomitantly aiding the interpretation of occupational biomonitoring data.
Charge Question 2b
- What guiding principles or factors could help prioritize substances or sectors that have been identified to date?
- If sectors are identified as priorities, what considerations are important for prioritizing the chemicals for further work within those sectors?
Data collection using the 3 main data sources shown in Figure 1 needs to be followed by prioritization. The SC supported the approach used in Australia by NICNAS of binning chemicals according to priorities of tier I, II and III based on the need for additional information gathering.
To further address this question, the SC discussed criteria to help prioritize substances and sectors. Discussions then turned to identifying who might be involved with prioritization and how the prioritization process could be evaluated.
In terms of who should be involved in prioritizing substances or sectors, the SC suggested that the prioritization process could be aided by input from a multi-party committee that could draw members from labour, representatives from the federal, provincial and territorial governments, academics, clinicians and non-governmental organizations and, potentially, industry. The SC discussed at some length that different jurisdictional authorities within Canada could pose a significant challenge to bringing occupational considerations under a national umbrella. Labour, and thus oversight of workplaces, is mainly under the jurisdiction of the provinces and territories, with differential capacity amongst these governments. The SC discussed the advantages of having a federal role in coordinating and enhancing efforts to reduce risks to workers from chemical exposures in the workplace.
For "how" to prioritize substances or sectors, the SC recommended that the prioritization process be "tested" through the use of case studies such as occupational exposure to silica and diesel exhaust. Extensive information on occupational exposure and risk evaluation is available for both these cases. Further, CMP has a mandate and the instruments to work on these substances.
Criteria
The SC discussed principles or criteria that could be used to prioritize substances or sectors:
- Priorities that have been identified in the information sources listed under Charge Question 2a such as Schedule 1 toxic substances determined through CMP. In the case of priorities identified through CMP, actions to reduce worker exposure are possible given the existence of regulations.
- Sectors and substances for which a large number of individuals could be exposed. This could start by looking at High Production Volume (HPV) chemicals where a variety of sectors could be involved. It could also start with sectors that are dominant in the Canadian economy such as the resource extraction sector.
- New and emerging sectors could be prioritized. Several examples were given such as cannabis growers and nail salons.
- Sectors that are data-rich or that could be modelled could also be prioritized. The OECD Emission Scenario Documents were raised as being potentially useful.
- Prioritization could be given to situations where gaps have been identified.
- One gap discussed was that of consumer products that are used in occupational settings (see section 1.5). The SC heard several examples where this occurs, including office cleaners using cleaning products and the beauty sector using cosmetics. Some have observed that although these workers might formally be subject to the "workers' right to know" provisions under the Workplace Hazardous Materials Information System (WHMIS), in practice these workers lack access to safety information, such as a SDS.
- Another gap that was identified was product classification. For example, welding rods are not always considered hazardous until used, which reduces the information available for worker protection.
- Priority could be given where workers are vulnerable. The SC discussed examples of workers who are particularly vulnerable, such as low-income individuals, those working on contract (for example, office cleaners), and those employed where labour regulations are poorly enforced.
- Situations where alternatives could be assessed could be prioritized. In assessing and acting on occupational risks, it is important to avoid "regrettable substitutions," whereby a known toxic substance is replaced with one for which less information is available but which could be of similar or greater concern.
Summary
The SC supported HC in seeking ways to reduce risks in workplaces arising from chemical exposures. The SC considered lessons learned from other jurisdictions that have addressed worker protection, while being aware of jurisdictional arrangements in Canada. The SC saw parallels between the discussion of occupational exposure and the previous discussion of taking a public health approach to chemical management, where both need to consider complex exposure scenarios to chemical mixtures.
The following is a summary of the main points from the 2-day meeting held to discuss considerations that could inform HC moving forward to reduce workplace risks due to chemical exposures:
- The SC supported the recommendation of the UN Special Rapporteur to incorporate workplace considerations under the CMP.
- HC can play an essential role in coordinating and sharing information at a national level, as well as training, with the provinces and territories, with the goal of enabling best practices, but not necessarily harmonization. Jurisdictional conflict should be avoided through, for example, not being overly prescriptive but rather by fostering inter-jurisdictional cooperation.
- HC can play a key role in convening a multi-party committee to recommend priorities, drawing members from labour, representatives from the federal, provincial and territorial governments, academics, clinicians and non-governmental organizations, and possibly industry.
- It will be important for HC, in working to forge partnerships and coordinate funding efforts, to operationalize an integrated and national system of surveillance, prioritize substances for regulatory action (for example, risk assessment/management, OEL setting, hazard classification), and be involved with information gathering, education, and action.
- HC can take a more proactive role in centralizing information related to emerging or rapidly changing types of work (like nail salons, cannabis growing, recycling).
- Modelling should be investigated as a means of providing estimates of workplace exposure in the absence of monitoring data and for building generalizations. The SC discussed the merits of modelling that could consider substances using a life cycle assessment-risk assessment framework.
- The SC identified the need for better disclosure and availability of information for prioritization and hazard classification. In addition, "workers' right to know" and worker education should be enhanced to ensure that workers are sufficiently informed of the hazards associated with various chemicals and substances with which they work or may come in contact with in the workplace, including products not intended for occupational use and products for which full disclosure of hazardous substances is lacking.
- Information for hazard classification and prioritization can be obtained from other international programs such as Europe's REACH, the U.S.'s TSCA and the Australian program under the authority of NICNAS. Canadian sources of information include CMP risk assessments, data from the provinces and territories, academia, and other Canadian sources such as occupational health clinics. Information can include measurements where the SC saw a role for HC to coordinate data curation and availability.
- Case studies conducted or coordinated by HC could inform how to best move forward to consider workplace exposures under CMP and to prioritize further activities.
References
- CCOHS (Canadian Centre for Occupational Health and Safety). 2020a.
Globally Harmonized System (GHS) (accessed 07-08-2020). - CCOHS (Canadian Centre for Occupational Health and Safety). 2020b.
WHMIS 2015 - Confidential Business Information (CBI) (accessed 07-08-2020). - Csiszar, S. A., Meyer, D. E., Dionisio, K. L., Egeghy, P., Isaacs, K. K., Price, P. S., Scanlon, K. A., Tan, Y., Thomas, K., Vallero, D. Bare, J. C. 2016. Conceptual Framework To Extend Life Cycle Assessment Using Near Field Human Exposure Modeling and High-Throughput Tools for Chemicals. Environ Sci Technol 50(21), 11922–11934.
- DTSC (Department of Toxic Substances Control). 2020. Safer Products.
- Davies, H.W. 2019. The Canadian Workplace Exposure Database. Presentation at the National Occupational Disease and Exposure Surveillance Workshop, November 7, 2019.
- Diamond, M.L.; Kvasnicka, J. 2020. Developing an exposure workflow to consider PBDE in electronic cases across the life cycle. Report prepared for U.S. EPA.
- Dimich-Ward, H.; Beking, K.J.; Dybuncio, A.; Bartlett, K.H.; Karlen, B.J.; Chow, Y.; Chan-Yeung, M. 2011. Respiratory health of two cohorts of terminal grain elevator workers studied 30 years apart. Am J Ind Med 54(4):263.
- Dionisio K.L.; Phillips K.; Price, P.S.; Grulke, C.; Williams, A.; Biryol, D. et al. 2018. The Chemical and Products Database, a resource for exposure-relevant data on chemicals in consumer products. Scientific Data. 5:1–9.
- Gravel, S.; Lavoué J.; Bakhiyi, B.; Diamond, M.L.; Jantunen, L.M.; Lavoie, J.; Roberge, B.; Verner, M.-A.; Zayed, J.; Labrèche, F. 2019. Halogenated flame retardants and organophosphate esters in the air of electronic waste recycling facilities: Evidence of high concentrations and multiple exposures. Environ Int 128: 244-253.
- Gravel, S.; Lavoué J.; Bakhiyi, B.; Lavoie, J.; Roberge, B.; Patry, L.; Bouchard, M.F.; Verner, M.-A.; Zayed, J.; Labrèche, F. 2020. Multi-exposures to suspected endocrine disruptors in electronic waste recycling workers: Associations with thyroid and reproductive hormones. Int J Hygiene Environ Health 225: 113445.
- Government of Canada. 2012. Guide to Cosmetic Ingredient Labelling.
- Government of Canada. 2015a. Guide to the Consumer Packaging and Labelling Act and Regulations.
- Government of Canada. 2015. Frequently Asked Questions – Consumer Chemicals and Containers Regulations, 2001.
- Government of Ontario, 2012. WHMIS and the worker (accessed 07-08-2020).
- Hall AL, Peters CE, Demers PA, Davies H. 2014. Exposed! Or not? The diminishing record of workplace exposure in Canada. Can J Public Health 105(3):e214-e217.
- Isaacs, K.K., Dionisio, K., Phillips, K. et al. 2020. Establishing a system of consumer product use categories to support rapid modeling of human exposure. J Expo Sci Environ Epidemiol 30, 171–183.
- Jung JKH, Feinstein SG, Palma Lazgare L, Macleod JS, Arrandale VH, McLeod CB, Peter A, Demers PA. 2018. Examining lung cancer risks across different industries and occupations in Ontario, Canada: the establishment of the Occupational Disease Surveillance System. Occup Environ Med 75(8):545-552.
- Keir JLA, Akhtar US, Matschke DMJ, Kirkham TL, Chan HM, Ayotte P, White PA, Blais JM. 2017. Elevated Exposures to Polycyclic Aromatic Hydrocarbons and Other Organic Mutagens in Ottawa Firefighters Participating in Emergency, On-Shift Fire Suppression.
Environ Sci Technol. 2017 Nov 7;51(21):12745-12755. doi: 10.1021/acs.est.7b02850. - Li, L.; Wania, F., 2016. Tracking chemicals in products around the world: introduction of a dynamic substance flow analysis model and application to PCBs. Environ. Int. 94, 674-686.
- Li, L.; Liu, J. G.; Hu, J. X.; Wania, F.. 2017. Degradation of Fluorotelomer-Based Polymers Contributes to the Global Occurrence of Fluorotelomer Alcohol and Perfluoroalkyl Carboxylates: A Combined Dynamic Substance Flow and Environmental Fate Modeling Analysis. Environ. Sci. Technol. 51, (8), 4461-4470.
- Li, L.; Hoang, C.; Arnot, J. A.; Wania, F. 2020. Clarifying Temporal Trend Variability in Human Biomonitoring of Polybrominated Diphenyl Ethers through Mechanistic Modeling. Environ. Sci. Technol. 51(1): 166-175.
- Li, L., Westgate, J. N., Hughes, L., Zhang, X., Givehchi, B., Toose, L., Armitage, J. M., Wania, F., Egeghy, P., Arnot, J. A. 2018. A model for risk-based screening and prioritization of human exposure to chemicals from near-field sources. Environ Sci Technol 52(24), 14235-14244.
- Meyer, D.E.; Bailin, S.C.; Vallero, D; Egeghy P.P.; Liu, S.V.; Cohen Hubal, E.A., 2020. Enhancing life cycle chemical exposure assessment through ontology modeling. Sci Tot Env 2020 712, 136263.
- Nguyen, L.V.; Diamond, M.L.; Venier, M.; Stubbings, W.A.; Guo, J.H.; Romanek, K.; Bajard, L.; Melymuk, L.; Jantunen, L.M.; Arrandale, V.H. 2019. Exposure of Canadian e-waste dismantlers to flame retardants. Environ Int 129: 95-104.
- Phillips, K. A., Wambaugh, J. F., Grulke, C. M., Dionisio, K. L., & Isaacs, K. K. 2017. High throughput screening of chemicals as functional substitutes using structure-based classification models. Green Chemistry 19(4), 1063–1074.
- UNHR (United Nations Human Rights), Office of the High Commissioner. 2019. End-of-visit statement by the United Nations Special Rapporteur on human rights and hazardous substances and wastes, Baskut Tuncak, on his visit to Canada, 24 May to 6 June 2019.
- Williams AJ, Grulke CM, Edwards J, McEachran AD, Mansouri K, Baker NC, et al. 2017. The CompTox Chemistry Dashboard: a community data resource for environmental chemistry. J Cheminformatics. 9:61.
Footnotes
- Footnote 1
-
Read-across is a technique for predicting endpoint information for 1 substance (target substance), by using data from the same endpoint from (an)other substance(s), (source substance(s)).
- Footnote 2
-
NAMs are any non-animal technology, methodology, approach, or combination thereof that can be used to provide information on chemical hazard and risk assessment.
- Footnote 3