Guidance Document for Cabinet X-Ray Equipment: Guide-001
2013
Foreword
Guidance documents are meant to provide general information to industry and others on how to comply with governing statutes and regulations. Guidance documents also provide assistance to Departmental staff on how Health Canada mandates and objectives should be implemented in a manner that is fair, consistent and effective.
Guidance documents are administrative instruments not having force of law and, as such, allow for flexibility in approach. Alternate approaches should be discussed in advance with the relevant program area to avoid the possible finding that applicable statutory or regulatory requirements have not been met.
As a corollary to the above, it is equally important to note that Health Canada reserves the right to request information or material, or define conditions not specifically described in this document, in order to allow the Department to adequately assess a radiation emitting device. Health Canada is committed to ensuring that such requests are justifiable and that decisions are clearly documented.
This document should be read in conjunction with the accompanying notice and the relevant sections of other applicable regulatory documents.
Table of Contents
- 1. Introduction
- 1.1. Purpose
- 1.2. Background
- 1.2.1. Roles and Responsibilities
- 1.2.2. Safety Standards
- 1.3. Scope and Application
- 1.4. Definitions
- 2. Guidance for Implementation
- Section 2.2: Device Design and Construction
- Section 2.3: Installation and Maintenance Instructions
- Section 2.4: Device Labelling
- Section 2.5: Additional Labeling
- Section 2.6: Shielding
- Section 2.7: Design Safety Features
- Section 2.8: Design Safety Features
- Section 2.9: Design Safety Features
- Section 2.10: Standards of Functioning
- Section 2.11: Warning Sign Specification
- 3. Enquiries
- 4. References
1. Introduction
1.1 Purpose
This guidance document is intended to inform manufacturers, importers and distributors and others of cabinet x-ray equipment of the requirements of the Radiation Emitting Devices (RED) Act and its Regulations as well as to identify best practices that are highly recommended. It is not intended to substitute for, supersede or limit the requirements under the RED Act and its Regulations. In case of any discrepancy between this summary and the legislation, the legislation will prevail. In addition, other laws, whether federal, provincial or territorial, may also apply to the devices covered by this law.
1.2 Background
1.2.1 Roles and Responsibilities
Health Canada is a regulatory authority for radiation emitting devices and is responsible for administering the Radiation Emitting Devices (RED) Act and its Regulations. The Consumer and Clinical Radiation Protection Bureau (CCRPB) is part of the Healthy Environments and Consumer Safety Branch (HECSB) of Health Canada. The CCRPB carries out inspections (compliance verification) and can apply various subsections of the RED Act to verify and enforce compliance of radiation emitting devices.
It consults or collaborates and, where appropriate, with branches of the federal government where other legislation applies to a radiation emitting device for protection of health. It does not approve radiation emitting devices for sale or importation, or perform pre-market assessment of such devices on behalf of manufacturers, importers or distributors, or issue letters, certificates or licenses regarding compliance of such devices.
All manufacturers, sellers, importers and distributors of radiation emitting devices must ensure that devices comply with the applicable requirements of the RED Act and, including, if applicable, other federal legislation and associated regulations.
Provincial, territorial and federal contacts, who could answer questions concerning their respective legislative regimes, are provided in the Reference Section of this document.
1.2.2 Safety Standards
As radiation emitting devices have the potential to harm human health and safety, they must meet the general requirements of Sections 4 and 5 of the RED Act as well legislative requirements set out elsewhere in the Act and the Regulations.
The RED Regulations prescribe specific safety standards governing the design, construction and function of various classes of radiation emitting devices, including certain medical devices (e.g. medical x-ray and ultrasound equipment), certain consumer products (e.g. microwave ovens and laser pointers), or certain industrial/commercial products (e.g. airport security screening equipment and tanning beds). This guidance document focuses on cabinet x-ray equipment, for which the applicable RED Regulations fall under Schedule II, Part XV: Cabinet X-Ray Equipment.
Certification of compliance with a foreign radiation safety standard is not a substitution for compliance with the RED Act and its regulations.
1.3 Scope and Application
This guidance document is intended to provide general information to manufacturers, importers, distributors and others about the RED Act and applicable Regulations as well as to identify best practices that are highly recommended with respect to cabinet x-ray equipment (as defined under Schedule I of the RED Regulations). It is not intended to substitute for, supersede or limit the requirements under the RED Act and its Regulations. In case of any discrepancy between this summary and the legislation, the legislation will prevail.
1.4 Definitions
For the purpose of this guidance document, the subsequent terms are defined as follows:
- Class/Classification:
- The 15 types of radiation emitting devices, as listed in Schedule I (Parts 1-15) of the RED Regulations. These device classes are subject to specific regulations outlined in Schedule II, Parts I-XV. « Classe ou Classification »
- Cabinet X-Ray Equipment:
-
X-ray generating devices, not including analytical x-ray equipment or baggage inspection x-ray devices, that have the x-ray tube permanently installed in a cabinet and are designed primarily for the examination of material, part or all of which is placed within the cabinet. For cabinet x-ray equipment, the applicable
RED Regulation is Schedule II, Part XV.
« Équipement à rayons X logé dans une enceinte » - Collimator:
- A device or mechanism that limits the size and shape of the primary beam by the use of radiation shields. « Collimateur »
- Compliance Evaluation:
- A generic term that means evaluating, assessing, testing or modifying a product to ensure or demonstrate conformance with an applicable statute or prescribed standard. « Évaluation de la conformité »
- Compliance Verification:
- Actions undertaken by Health Canada to verify compliance with the RED Act and applicable government regulations in respect of radiation emitting devices sold (including re-sale or donated), leased, imported or distributed in Canada. « Vérification de la conformité »
- Door or Panel:
- A physical barrier that prevents or inhibits human intrusion into hazardous areas of the cabinet x-ray equipment and provides shielding of radiation. « Porte ou panneau »
- Failsafe:
- Having the property that any failure causes an action or actions which always result in a safe situation. « Sécurité intégrée »
- Imaging Receptor or Radiation Detector:
- The device that interacts with the x-rays to produce a signal corresponding to the intensity of the x-rays incident on it. « Récepteur d'image ou Détecteur de rayonnements »
- Irradiation Chamber :
- That portion of the cabinet x-ray equipment that facilitates irradiation of materials, contains the imaging receptor or its accessories thereof, and is enclosed by radiation shields, doors or panels and by mechanisms that facilitate moving, positioning or controlling materials for irradiation in the primary beam. « Chambre d'irradiation »
- Interlock:
- A device that precludes radiation exposure to an individual by preventing entry to a hazardous area or by automatically removing the hazard. « Dispositif de verrouillage »
- Label:
- This includes any legend, word or mark attached to, included in, belonging to or accompanying any radiation emitting device or package. « Étiquette »
- Leakage Radiation:
- All radiation coming from a radiation source, except primary radiation. It includes secondary radiation. « Rayonnement de fuite »
- Leakage Radiation Test or Radiation Survey:
- An evaluation of the levels of radiation associated with the use, operation, maintenance or testing of the x-ray equipment. It is intended to assess compliance with the regulatory limits stated in Section 10 of the Regulations. « Essai des rayonnements de fuite ou Contrôle des rayonnement »
- Leakage Radiation Rejection Criteria or Rejection Criteria :
- The lower limit of leakage radiation exposure rate requiring investigation by the manufacturer and corrective action. Rejection criteria should be conservatively determined to be lower than the regulatory limit stated in Section 10 of the Regulations. [Section 10 prescribes the leakage radiation as follows: when averaged over a detection area of ten square centimetres, the exposure rate from leakage radiation, averaged over a time period of no less than five minutes, shall not exceed 0.5 milliroentgen per hour at a distance of five centimetres from any accessible external surface.] « Critère de rejet des rayonnements de fuite ou Critère de rejet »
- Manual or Operational Manual:
- The reference material or book accompanying a radiation emitting device, explaining its functions, components, safety features as well as how to install, commission, operate and maintain that device. Such materials are to be provided to clients in both English and French. « Manuel ou Manuel d'utilisation »
- Panel see Door
- « Panneau voir Porte »
- Permanent Installation:
- A shielded enclosure that is equipped with safety features and is in a fixed location. « Installation Permanente »
- Port:
- Access openings or that portion of the cabinet x-ray equipment which facilitates the entry and exit of materials in the irradiation chamber. « Port »
- Primary Beam or Primary Radiation:
- Radiation that emanates from a radiation source or target and passes through the radiation source assembly via a collimator or other beam shaping device, and that is intended to irradiate materials . « Faisceau primaire ou Rayonnement primaire »
- Radiation Shield:
- Materials that attenuate or absorb ionizing radiation to acceptable levels. « Écran de protection radiologique ou Blindage »
- Radiation Survey
- see Leakage Radiation Test « Contrôle des rayonnements voir Essai des rayonnements de fuite »
- Redundant:
- The use of more than the minimum number of items needed to accomplish a given safety function, and is intended to improve the reliability of systems important to safety. « Redondant »
- Secondary Radiation:
- Ionizing radiation emitted by matter as a result of the interaction of primary radiation with that matter. « Rayonnement secondaire »
- Shroud:
- A structure that comprises one or more panels or doors and is configured at the ports of the cabinet x-ray equipment. A shroud is subject to the radiation limit of Section 10, including the imaginary vertical plane surface at its end which is furthest from the primary beam, even when flexible materials create intermittent gaps that allow secondary radiation to emerge. « Écran protecteur »
- X-Ray Detector
- see Imaging Receptor « Détecteur de rayons X voir Récepteur d'image »
2. Guidance for Implementation
2.1 General Regulatory Requirements for Cabinet X-Ray Equipment
2.1.1 Radiation Emitting Devices Act
Under the RED Act, it is incumbent upon the manufacturer, importer or distributor, as the case may be, to ensure that they meet the requirements of Sections 4 and 5 of the RED Act, as well as the other provisions of the Act and regulations applicable to their devices.
In addition, as per Section 6 of the RED Act, the Minister shall be notified by the manufacturer or importer of a radiation emitting device, where that person becomes aware, after the device has left the person's premises, of the fact that the device
- does not comply with the standards, if any, prescribed under paragraph 13(1)(b) and applicable thereto, or
- creates a risk to any person of genetic or personal injury, impairment of health or death from radiation by reason of the fact that it
- does not perform according to the performance characteristics claimed for it,
- does not accomplish its claimed purpose, or
- emits radiation that is not necessary in order for it to accomplish its claimed purpose.
Refer to Section 6 of the RED Act for additional details concerning Notification requirements.
2.1.2 Applicable Regulations
For cabinet x-ray equipment, each requirement of the applicable RED Regulation (Schedule II, Part XV) must also be met. It is good practice that industry carry out relevant compliance evaluations of their products and to retain all supportive documentation thereof. Such evaluations may be done in house by the industry or by a qualified 3rd party on its behalf.
Additional explanation and guidance for the industry relating to the Regulations (Schedule II, Part XV) are detailed below.
Sections 2.2-2.11 of this Guidance Document are numbered in accordance with the corresponding sections of Part XV of the Regulations (Sections 2-11) and should be read concurrently. Note that this Guidance Document does not replace the Regulations nor does it contain line by line substantiation of all parts or items of the Regulations. Reference should always be made to the official text of the RED Act and Regulations.
Items denoted with the preceding check box symbol indicate best practices that are highly recommended.
Certification of compliance with a foreign radiation safety standard is not a substitution for compliance with the RED Act and its regulations.
Section 2.2: Device Design and Construction
The following elements are recommended for inclusion in the operational manual:

- A general description of the design of the device and of the materials used in its construction, including its principles of operation. Each of the functional and safety components of the device should be clearly described as to function, with labelled pictorial representation of the device (e.g. diagrams, photographs or drawings).
- A section on how radiation is generated and produced in order to facilitate the intended use or application of the device, including a section solely dedicated to radiation protection and safety.
Section 2.3: Installation and Maintenance Instructions
The following elements are recommended to be included in the manual:

- Clear and explicit instructions regarding the installation, commissioning, operation and maintenance of the x-ray equipment, including protocol necessary in order to maintain compliance and functioning as intended. A complete list of installation and replacement components accessories that can be sold separately and used with other devices, systems or units should be identified.
- Manufacturer quality assurance (QA) testing procedures, including testing of all safety components and leakage radiation testing. Manufacturer-recommended maintenance checks should be specified, including suggested scheduling (i.e. daily, weekly, monthly, quarterly and annually). Leakage radiation rejection criteria should be explicit and conservatively determined to be lower than the regulatory limit stated in Section 10 of the Regulations.
- Specific instruction that a radiation re-survey should be carried out by qualified and authorized personnel in the event that any critical component (i.e. x-ray tube, detector, generator, shielding or any mechanism that controls the generation, emission, collimation, transmission or attenuation of x-rays) is serviced, repaired or replaced. (See Section 2.10 of this document for additional information on radiation surveys.)
Section 2.4: Device Labelling
Section 4 (a) of the Regulations requires clear and legible radiation warning signs and labels that shall be provided in the form of a tag or label permanently affixed or inscribed on the control panel of the device.
Radiation warning labels in respect of paragraph 4 (a)(i) and (ii) of the Regulations may be provided separately or in a single composite label (e.g. Figure 1).
Figure 1. Example of Composite Label.
CAUTION - X-RADIATION THIS EQUIPMENT PRODUCES HIGH INTENSITY X-RADIATION WHEN ENERGIZED. TO BE OPERATED ONLY BY QUALIFIED PERSONNEL.
Text description
It consists of two parts: (I) the X-Radiation warning sign and (II) additional caution notification in both official languages: THIS EQUIPMENT PRODUCES HIGH INTENSITY X-RADIATION WHEN ENERGIZED. TO BE OPERATED ONLY BY QUALIFIED PERSONNEL. / CET APPAREIL PRODUIT DES RAYONNEMENTS X À HAUTE INTENSITÉ LORSQU'IL EST SOUS TENSION. SON UTILISATION EST RÉSERVÉE AU PERSONNEL QUALIFIÉ.
ATTENTION - RAYONNEMENTS X CET APPAREIL PRODUIT DES RAYONNEMENTS X À HAUTE INTENSITÉ LORSQU'IL EST SOUS TENSION. SON UTILISATION EST RÉSERVÉE AU PERSONNEL QUALIFIÉ.
The manufacturer label, paragraph 4 (a)(iii), shall explicitly include:
- the full name of the manufacturer;
- the model designation;
- the serial number;
- the date of manufacture (the month and year [four-digit] should be explicit); and
- the city and country of manufacture.
It is recommended that all labels be provided in both English and French. Only the x-radiation warning sign (See Figure 1, above) shall be used throughout. (The trefoil symbol implies continuous radiation.)
It is recommended that radiation warning labels and manufacturer labels be placed on the front of the control panel in direct view of the operator.

Clear texts and/or copies of all labels (including manufacturer label and radiation warning labels) are recommended to be provided in the operational manual.

Section 2.5: Additional Labeling
Subsection 5 (a) of the Regulations requires that all marks, labels and signs are securely affixed to the device and are clearly visible, legible and easily identifiable.
Subsection (b) requires that all controls, meters, lights or other indicators are easily identifiable and clearly labelled or marked with respect to function.
It is recommended that a labelled replica or diagram of the components referenced in Subsection 5 (b) should be included in the operational manual, including functional and safety descriptions where applicable.

Section 2.6: Shielding
Section 6 of the Regulations requires that sufficient shielding is used so that the device functions within the standards of functioning described in Section 10 of the Regulations (i.e. radiation emission limits are met).
It is recommended that details concerning the shielding materials used in the construction of the device be included in the operational manual.

Section 2.7: Design Safety Features
Section 7 of the Regulations requires that the device design and construction include necessary safety features as described in the associated subsections (a) - (f).
Subsection (a) lists the safety features that shall be included on the control panel. It should be noted that:
- Item (i) refers to a power "ON/OFF" switch. This switch should not be used in lieu of an emergency switch.
- Item (iv) refers to an x-ray warning indicator which is of a failsafe design, unless there is a warning light or other indicator on the control panel. The latter means a redundant indicator is required.
- Item (v) refers to redundancy of the x-ray warning indicator described in Item (iv). Where redundancy is utilized, x-ray generation should not be possible when these redundant components fail.
Subsection (b) requires shielded doors or panels over all access openings designed for insertion or removal of any material to be examined except where the design of the device prevents the insertion of any part of the human body into the primary x-ray beam.
Subsection (c) requires at least two independent interlocks be connected to the doors or panels described in subsection (b) in order to prevent the generation of x-rays should any such door or panel be opened.
Subsection (d) requires at least one interlock on all doors or panels that provide access to any critical component associated with the generation, production, collimation, transmission or detection of radiation.
With respect to subsection (e), unless the conditions of Section 8 of the Regulations are met, that is, the device is comprised of a conveyor, the device shall include one or more controls requiring separate operator action to initiate each x-ray exposure.
The beam limiting device described in subsection (f) should ensure that the size of the divergent primary x-ray beam at the plane of the imaging receptor does not exceed the maximum size of the imaging receptor/x-ray detector.
It is recommended that labeled pictorial diagrams of each component and safety feature be captured in the operational manual, including a brief functional description.

Section 2.8: Design Safety Features
Subparagraph 8 (b)(ii)(A) of the Regulations requires that a remote warning light indicating when x-rays are being generated is to be included on any x-ray equipment controlled by a central processing unit and that does not require the continuous presence of an operator (e.g. a beacon light on top of the device would satisfy this requirement as it is viewable at 360 degrees).
Subparagraph 8 (b)(ii)(B), considers that a similar mechanism, a control or switch to stop the conveyor or other automatic feed system and terminate x-ray generation, already exists on the control panel. The safety rationale for the duplicate control or switch is that, in case of an emergency, personnel positioned on either lateral side of the x-ray equipment should have easy access to the stop mechanism. This mechanism shall be designed to perform its required functions (promptly terminating x-ray exposure and stopping the conveyor) and, subsequently, to require specific manual operator action(s) in order to re-establish conditions conducive to x-ray generation.
A note about x-ray devices containing a conveyor system: Flexible materials, which may or may not contain lead, may be utilized at the entrance and exit ports of a conveyor system in order to reduce x-ray emissions. However, such flexible materials are deflected as items pass through, creating intermittent gaps where the radiation limits of Section 10 of the Regulations could be exceeded (i.e. exposure from leakage radiation should not exceed 0.5 milliroentgen per hour at a distance of five centimetres from the external surface of the device). It should be noted that the radiation emission limits of Section 10 apply at the plane of entry/exit ports even when items are translated for examination in the system.
Subparagraph 8(b)(iii) requires that devices containing a conveyor or other automatic feed system should include shrouds (solid panels or doors), which function as radiation shields preventing any person from reaching inside the irradiation chamber or near the intermittent gaps of the flexible materials or areas where radiation emission levels exceed the regulatory limit.
These shrouds or panels:

- are to be permanently affixed to the cabinet x-ray equipment and the conveyor chassis; and
- if designed to be opened (e.g. to remove product jams or address problems associated with the automatic feed system), shall be equipped with two independent interlocks to prevent the generation of x-rays if any such shroud or panel is open in view of the fact that they provide access to the conveyor area or irradiation chamber where radiation levels are high [See also Subsections 7(b) and (c) of the Regulations]. The closure of any such shroud or panel shall not result in the re-generation of x-rays, unless another specific action is taken by the operator.
Clear and unambiguous photographs and/or diagrams of the location of each of these components and a brief description of their functions are recommended to be provided in the operational manual.

Section 2.9: Design Safety Features
There are cabinet x-ray systems designed with large entrance and exit ports that can accommodate heavy or bulky materials, which are placed on pallets or specialized containers and which are translated into or out of the path of the primary beam by conveyor or other translational mechanisms (e.g. rollers, rail, manually or automatically propelled vehicles). Owing to the configuration, if human access is physically possible beyond the entrance and exit ports and into the path of the primary beam or into any part of the irradiation chamber thereof, the requirements of Section 9 apply.
Examples that may fall into this category include cabinet x-ray systems designed with:
- a conveyor or other translational mechanism, which is at a height of 80 centimetres or less from the surface on which that cabinet x-ray system is positioned for operation, and
- entrance and exit ports that have internal dimensions greater than 150 centimetres in height (measured vertically) and 100 centimetres in width (measured horizontally).
With respect to subsection 9 (a) of the Regulations, any such switch or control is to be clearly identifiable and marked as to function [Refer to subsection 5 (b) of the Regulations].
It is recommended that the switches be positioned in such a way that activation (i) can occur without passing through the primary beam, and (ii) is easy for a person who inadvertently falls onto the conveyor or other translational mechanisms.

It is recommended that clear and unambiguous photographs and/or diagrams of the location of each of the safety feature components, (a) - (e), and a brief description of their functions be provided in the operational manual.

Section 2.10: Standards of Functioning
Section 10 of the Regulations specifies the maximum exposure rate for leakage radiation as follows:
When averaged over a detection area of 10 square centimetres, the exposure rate from leakage radiation (averaged over a period not less than five minutes), should not exceed 0.5 milliroentgen per hour at a distance of five centimetres from any accessible external surface.
For the exposure rate measurement, the radiation sensitive volume of the quantitative instrument shall have a cross section aligned parallel to the external surface of the x-ray machine with an area of 10 square centimetres and no linear dimension more than 5 centimetres.
Radiation surveys should incorporate the use of both qualitative and quantitative instrumentation, and the corresponding survey report are recommended to include:

- the x-ray tube kilovoltage and tube current used (maximum design ratings of the x-ray tube should be used);
- the nominal size of the radiation field typically used to test materials;
- the characteristics and dimensions of the scatter (media which should mimic the scattering properties of soft tissue);
- the quantitative instrument used should be calibrated over the x-ray energy range consistent for the cabinet x-ray system;
- the quantitative instrument used, as well as a copy of its validated calibration status and operational characteristics;
- leakage measurements (in units of milliroentgen per hour) from all external surfaces and panels of the x-ray system; and
- the date and signature of the person who performed that survey.
It is recommended that detailed quality assurance (QA) testing procedures, including testing of all safety components and radiation leakage testing (along with associated signed and dated test reports) be documented and kept on file. Detailed QA procedures should also be captured in the operational manual, as indicated in Section 2.3 of this guidance.

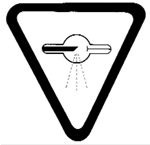
Section 2.11: Warning Sign Specification
Section 11 of the Regulations specifies requirements for the radiation warning signs referred to in Section 2.4 of this guidance.
The radiation warning symbol is to:
- be displayed in two contrasting colours;
- be clearly visible and recognizable from a distance of one metre;
- have no outer dimensions less than two centimetres, while remaining consistent with the size of the object to which it is affixed;
- bear the words "CAUTION, X-RAYS" and the French equivalent "ATTENTION, RAYONS X"; and
- be designed in accordance with the following diagram (Figure 2):
Figure 2. Radiation warning symbol.

3. Enquiries
For questions, clarification, and further information concerning compliance requirements for cabinet x-ray equipment, contact Health Canada, Consumer and Clinical Radiation Protection Bureau (CCRPB) at the following e-mail address: CCRPB-PCRPCC@hc-sc.gc.ca
Or by mail at:
Consumer and Clinical Radiation Protection Bureau
Health Canada
P.L. 6301A
775 Brookfield Road
Ottawa, ON
K1A 1C1