Annual Report on Compliance and Enforcement Activities (Tobacco Control), 2014–2015
Table of Contents
What We Do
Mission and Vision
Health Canada is the federal department responsible for helping the people of Canada maintain and improve their health.
Health Canada is committed to improving the lives of all of Canada’s people and to making this country’s population among the healthiest in the world as measured by longevity, lifestyle, and effective use of the public health care system.
Tobacco Control in Canada
Tobacco use is the leading preventable cause of disease and premature death in Canada. It causes many serious chronic illnesses, including cancer, respiratory ailments, and heart disease. Half of long-term smokers die prematurely from smoking-related diseases. Each year, about 37,000 Canadians die from tobacco use.
Since 2001, the Government of Canada has been addressing this national public health problem through the Federal Tobacco Control Strategy (FTCS). Built on the principles of prevention, protection, cessation, and product regulation, the FTCS aims to reduce smoking prevalence and the resulting disease and death among Canadians.
Provinces and territories also have tobacco control strategies, a situation which reflects the shared responsibility in tobacco control among levels of government.

Canada’s Tobacco Act
The Tobacco Act is a key component of the FTCS. The Act regulates the manufacture, sale, labelling, and promotion of tobacco products. The Act recognizes the inherent harmful nature of tobacco products; even when tobacco manufacturers comply with all the provisions of the Tobacco Act, their products remain harmful.
Within Health Canada, the Tobacco Act and its regulations are administered by the Tobacco Control Directorate of the Healthy Environments and Consumer Safety Branch, while the related inspection and investigation activities are carried out by the Regions and Program Bureau (which became the Regulatory Operations and Regions Branch in April 2016).
Canada and the World Health Organization Framework Convention on Tobacco Control
Canada, together with 179 other jurisdictions, is a Party to the World Health Organization Framework Convention on Tobacco Control (FCTC). The FCTC is the first international treaty negotiated under the auspices of the World Health Organization.
The FCTC, which came into force in 2005, was developed in response to the global tobacco epidemic. It reaffirms the right of all people to the highest standard of health. Based on scientific evidence, it sets out minimum requirements for action; Parties to the treaty are invited to exceed those requirements.
Executive Summary
The purposes of this report are to inform interested parties about Health Canada’s level of effort in monitoring compliance with the Tobacco Act and its regulations, and to identify areas where the tobacco industry has not complied with the applicable legislation.
Tobacco control initiatives in Canada have led to a significant reduction in smoking prevalence. However, despite decades of effort, there are still over 5 million tobacco users in Canada, including 4.2 million current smokers.
Canada’s current tobacco control efforts are grounded in the Federal Tobacco Control Strategy. Actions taken pursuant to the Tobacco Act contribute to preventing people—in particular youth—from taking up tobacco use, and to helping tobacco users quit.
Health Canada actively monitors tobacco manufacturers and importers, retailers, and others to identify cases of non-compliance with the Act and regulations. Health Canada takes enforcement actions where warranted.
Key Statistics for 2014–2015
Through its compliance monitoring and enforcement activities, between April 2014 and March 2015 (i.e. fiscal year 2014–2015) Health Canada:
- Collected and analyzed over 1300 samples from the tobacco manufacturing and importing sector. Among the subset of 457 samples where labelling requirements were assessed, 9% were found to be in non-compliance with the Tobacco Products Labelling Regulations (Cigarettes and Little Cigars).
- Completed 6774 inspections of tobacco retailers. The most frequently observed infractions were the presence of self-service tobacco displays (where consumers were able to handle tobacco products before purchase), and promotions of tobacco products that are not permitted under the Tobacco Act.
- Reviewed 1607 reports from tobacco manufacturers and importers—reports that are required under the Tobacco Reporting Regulations of the Tobacco Act. During the year, 81 letters of deficiency were issued due to incomplete or missing reports. These letters indicate to the manufacturer or importer which missing information is to be submitted to Health Canada, along with the time-frame for doing so.
It is important to note that tobacco manufacturers, importers, and retailers who comply with the different provisions of the Tobacco Act still manufacture or sell a harmful product: every year, 37,000 people die in Canada from tobacco use, which is about one of every six deaths.
Compliance and Enforcement Activities
Scope of Tobacco Control Efforts
Current tobacco control legislation supports the Federal Tobacco Control Strategy in helping prevent people from taking up tobacco use, and helping users quit. Specifically, the Tobacco Act is intended to protect young people and others from inducements to use tobacco products, restrict youth access to these products, and enhance public awareness of the health hazards of using tobacco products. The Act does not directly address the harmful effects of tobacco products.
Since its coming into force in 1997, the Tobacco Act has been amended twice, and a number of regulations have been adopted.
Key Measures
- Protecting youth from inducements to use tobacco products by banning additives, including flavours, which contribute to making these products attractive: With the Cracking Down on Tobacco Marketing Aimed at Youth Act, which amended the Tobacco Act in 2009, Canada became the first country in the world to ban the use of certain additives in cigarettes, little cigars, and blunt wraps. The banned additives include flavours like chocolate and bubble gum that were making tobacco products more appealing to youth.
- Restricting advertising for tobacco products: The promotion of tobacco products by means of “information advertising” or “brand-preference advertising” is limited to publications delivered by mail to a named adult and on signs in places where young persons are not permitted by law.
- Labelling requirements, such as health warnings: Canada was the first country to require pictorial health warnings on tobacco packages in 2000. In June 2011, the Tobacco Products Labelling Regulations (Cigarettes and Little Cigars) were adopted to renew and strengthen these messages. These Regulations increased the size of the health warnings from 50% to 75% of the main panels of cigarette and little cigar packages.
- Disclosure requirements for the tobacco industry: Under the Tobacco Reporting Regulations, tobacco manufacturers and importers must submit regular reports that include sales data, manufacturing information, information on the ingredients used in their products, constituents and emissions information, as well as information on their research and promotional activities.
Three Pillars of Compliance and Enforcement Activities
Compliance and enforcement activities are based on three pillars, as described below.
- Compliance Promotion
- Compliance Monitoring
- Enforcement
1. Compliance Promotion
To promote industry’s compliance with the Tobacco Act and its regulations, Health Canada carries out a range of activities, including publishing public notices and conducting presentations with businesses. By sharing information on the applicable restrictions and requirements, Health Canada expects that manufacturers, importers and retailers will make well-informed decisions regarding how they manufacture, sell, label and promote their products.
Compliance promotion initiatives may be stand-alone activities (such as information provided in trade publications, direct e-mail communication to affected industry members, information sessions) or they may be part of a planned inspection.
During on-site inspections at both manufacturer/importer and retail locations, inspectors provide, as needed, information on the provisions being monitored. They also answer questions and listen to concerns that company representatives raise.
When new provisions are adopted, Health Canada informs those sectors of the industry that will be affected by these new measures.
- In fiscal year 2014–2015 there were no legislative amendments to the Tobacco Act or regulations that came into force. The Department’s compliance promotion initiatives focused on general compliance promotion of the Tobacco Act and its regulations during planned inspections.
2. Compliance Monitoring
Through its compliance monitoring work, Health Canada oversees regulated businesses to verify that their activities are carried out in accordance with the Tobacco Act and its regulations and to identify and assess cases of non-compliance. Both inspectors and reviewers play a role in the Department’s tobacco compliance monitoring activities:
- Inspectors conduct inspections at manufacturer/importer facilities and retailer locations across Canada. During an inspection, inspectors may perform audits, collect samples, and carry out analysis of tobacco product packaging and of promotional materials.
- Reviewers examine reports received from tobacco companies to determine if they comply with the Tobacco Reporting Regulations.
3. Enforcement
Health Canada uses a progressive enforcement regime, in which activities range from promoting compliance up to and including possible prosecution and court ordersFootnote 1. If a business is found to be non-compliant with a specific section of the Tobacco Act or its regulations, Health Canada considers a range of actions to induce, encourage, or compel the business to correct the observed non-compliance. The Department considers many factors in determining the appropriate enforcement action. Among these are: how severe the alleged non-compliance is; how likely it is to reoccur; how likely it is that the business will cooperate and become compliant; and, the expected impact of a possible enforcement action.
The main enforcement actions taken when Health Canada observes non-compliance include negotiating compliance with the regulated business, issuing warning letters, including those that outline deficiencies in industry reports, and seizing products.
Compliance Promotion, Monitoring, and Enforcement Activities involving the Manufacturing/Importing Sector
In Canada, about 60 manufacturers and importers are actively involved in the sale of tobacco products. The majority are located in Ontario and Québec. The main measures Health Canada monitors compliance with are:
- The cigarette ignition propensity standard, as set out in the Cigarette Ignition Propensity Regulations (“CIPR”);
- The prohibition on the use of certain additives in cigarettes, little cigars and blunt wraps, as per sections 5.1 and 5.2 of the Tobacco Act (“Prohibited additives”);
- The prohibition on promoting, by means of cigarette, little cigar and blunt wrap packaging, the presence of additives that cannot be in used in said products, as per section 23.1 of the Tobacco Act (“Prohibition of Promotion of Banned Additives on Packaging”);
- The minimum packaging requirements for cigarettes, little cigars and blunt wraps, as per section 10.1 of the Tobacco Act (“Minimum Packaging”); and,
- The labelling requirements (specifically, health warnings, toxic emissions statements, and health information messages) as set out in the Tobacco Products Labelling Regulations (Cigarettes and Little Cigars) (“TPLR”).
Inspections in fiscal year 2014–2015, and trends over time
Tobacco inspections in the manufacturing/importing sector consist of sampling tobacco products, which are then analyzed for compliance with the applicable provisions of the Tobacco Act and its regulations. Once the analyses are completed, the determination of an appropriate enforcement action is made for all cases of observed non-compliance.
Non-compliance rates based on samples from the manufacturing/importing sector analyzed against specific provisions are shown in the table below. The non-compliance rate reflects the number of samples found to be non-compliant with the provisions in question.
| Provisions | Number of Samples Analyzed | Non-Compliance Rate (%) |
|---|---|---|
| Cigarette Ignition Propensity Regulations (CIPR) | 20 | 0% |
| Prohibited Additives | 197 | 0% |
| Prohibition of Promotion of Banned Additives on Packaging | 436 | 0% |
| Minimum Packaging | 247 | 0% |
| Tobacco Products Labelling Regulations (Cigarettes and Little Cigars) (TPLR) | 457 | 9% |
In fiscal year 2014–2015, 9% of the tobacco products analyzed against the labelling provision did not meet the requirements set forth in the TPLR.
The number of warning letters issued and the number of seizures conducted by Health Canada in the manufacturing/importing sector in fiscal year 2014–2015 are shown in the table below.
| Enforcement Action | Health Canada Region | Number of Actions | ||||
|---|---|---|---|---|---|---|
| AtlanticFootnote 1 | Québec | Ontario | PrairiesFootnote 2 | British Columbia | ||
| Number of seizures conducted | 0 | 0 | 0 | 0 | 0 | 0 |
| Number of warning letters issued to manufacturersFootnote * | 0 | 12Footnote 3 | 8Footnote 3 | 0 | 0 | 20 |
|
||||||
Non-compliance rates based on samples from the manufacturing/importing sector analyzed over the last three fiscal years are set out in the following table. It shows trends over time for non-compliance with specific regulatory provisions. The non-compliance rate for each provision is shown as the percent of the number of non-compliant samples identified (shown in brackets), over the number of samples analyzed.
| Provisions | 2012–2013 | 2013–2014 | 2014–2015 | |||
|---|---|---|---|---|---|---|
| Number of Samples Analyzed (779) | Non-Compliance Rate (%) | Number of Samples Analyzed (1556) | Non-Compliance Rate (%) | Number of Samples Analyzed (1357) | Non-Compliance Rate (%) | |
| CIPR | 108 | 0% | 20 | 0% | 20 | 0% |
| Prohibited Additives | 60 | 0% | 318 | 0% | 197 | 0% |
| Prohibition of Promotion of Banned Additives on Packaging | 200 | 5% | 468 | 3% | 436 | 0% |
| Minimum Packaging | 200 | 5% | 262 | 3% | 247 | 0% |
| TPLR | 211 | 35% | 488 | 21% | 457 | 9% |
The non-compliance rates (from the above table) are presented in graph form below to show trends for fiscal years 2012–2013 to 2014–2015. The percentages represent the non-compliance rate for samples analyzed for each provision.
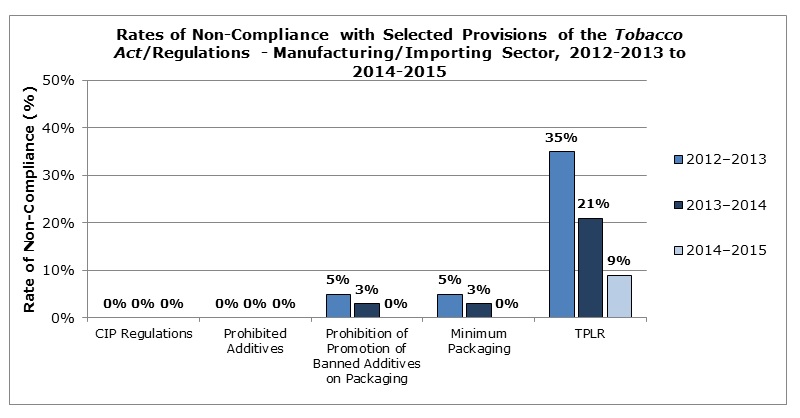
Figure 1 - Text Description
Description – Rates of Non-Compliance with Selected Provisions of the Tobacco Act/Regulations – Manufacturing/Importing Sector, 2012-2013 to 2014-2015.
Bar chart showing rates of non-compliance for selected provisions of the Tobacco Act and Regulations for fiscal years 2012-2013, 2013-2014, and 2014-2015.
CIP Regulations = 0% non-compliance for 2012-2013, 0% non-compliance for 2013-2014 and 0% non-compliance for 2014-2015; Prohibited Additives = 0% non-compliance for 2012-2013, 0% non-compliance for 2013-2014 and 0% non-compliance for 2014-2015; Prohibition of Promotion of Banned Additives on Packaging = 5% non-compliance for 2012-2013, 3% non-compliance for 2013-2014 and 0% non-compliance for 2014-2015; Minimum Packaging = 5% non-compliance for 2012-2013, 3% non-compliance for 2013-2014 and 0% non-compliance for 2014-2015; TPLR = 35% non-compliance for 2012-2013, 21% non-compliance for 2013-2014 and 9% non-compliance for 2014-2015.
In fiscal year 2012–2013, 5% of the 200 samples analyzed against the minimum packaging requirements showed non-compliance. For the same period, 5% of the 200 samples analyzed were found to be promoting prohibited additives on the packaging.
The TPLR came into force in 2011 and included a transition period until March 2012 for manufacturers and importers to introduce the new labelling elements on tobacco product packages. In fiscal year 2012-2013, 35% of the samples analyzed against these Regulations were found to be non-compliant.
In fiscal year 2013–2014, non-compliance was seen mainly with respect to the TPLR, where 21% of samples analyzed were found to be non-compliant.
Compliance Promotion, Monitoring, and Enforcement Activities involving the Retail Sector
There are about 30,000 to 35,000 points of sale for tobacco products across Canada. Health Canada monitors their compliance with the following key measures:
- Minimum packaging requirements
- Prohibition of promotion of banned additives on packaging
- Labelling requirements
- Prohibited promotional activities.
Inspections in fiscal year 2014–2015, and trends over time
Health Canada conducts inspections of tobacco retailers in order to promote and assess retailers’ compliance with the Tobacco Act and its regulations. A retailer is identified as non-compliant if at least one case of non-compliance that relates to the above-mentioned key measures is observed during the inspection.
The retail environment is where tobacco users have access to tobacco products. Taking timely action on observed non-compliance in the retail sector prevents the sale and promotion of tobacco products that are appealing to youth. It also helps prevent the sale of tobacco products that do not display the required health warnings.
The number of inspections and the non-compliance rate based on samples from the retail sector analyzed in fiscal year 2014–2015 are shown in the table below. The non-compliance rate is based on the percentage of retailers inspected where at least one case of non-compliance was observed.
| Provisions | Number of Inspections | Non-Compliance Rate (%) |
|---|---|---|
|
6774 | 12% |
In fiscal year 2014–2015, Health Canada completed 6774 retail tobacco inspections, with an overall non-compliance rate of 12% with respect to the provisions listed in the above table.
The number of enforcement activities (both seizures at retail and warning letters issued) in each region is shown in the table below.
| Enforcement Action | Atlantic | Québec | Ontario | Prairies | British Columbia | Total Number of Actions |
|---|---|---|---|---|---|---|
| Number of seizures at retail | 48 | 48 | 29 | 50 | 21 | 196 |
| Number of warning letters issued to retailersFootnote * | 0 | 0 | 2 | 0 | 6 | 8 |
|
||||||
The number of inspections conducted in the retail sector over the last three fiscal years is shown in the table below. The non-compliance rate for each year is expressed as the percentage of retailers inspected where at least one case of non-compliance was observed.
| Fiscal Year | Number of InspectionsFootnote 1 | Non-Compliance Rate |
|---|---|---|
| 2012–2013 | 9382 | 16% |
| 2013–2014 | 7724 | 14% |
| 2014–2015 | 6774 | 12% |
|
||
In recent years, the number of inspections conducted at the retail level has declined from 9382 (fiscal year 2012–2013) to 6774 (fiscal year 2014–2015). This decrease was due to a reduction in inspection resources.
Over the same period, the overall non-compliance rate has decreased by 4 percentage points (from 16% to 12%).
Compliance Promotion, Monitoring and Enforcement Activities Concerning Industry Reporting
As noted above, the Tobacco Reporting Regulations require tobacco manufacturers and importers to regularly submit reports to the Minister of Health (see previous section on Disclosure requirements for the tobacco industry for a list of the information to be submitted). Health Canada assesses the reports to verify that the required information is provided. When a report is assessed as missing required information, Health Canada issues a letter outlining the deficiency to the company who submitted the incomplete report.
As the information contained in the industry reports is used to inform policies and support the development of tobacco regulations, it is critical that the information received be complete and accurate.
In fiscal year 2014–2015, Health Canada reviewed 1607 reports from manufacturers and importers. Of the reports reviewed, 172 (11%) were determined to be incomplete, and a total of 81 letters of deficiency were issued. (In a number of cases, one letter referred to more than one deficiency.)
The number of reports reviewed, the number of these deemed incomplete and the number of letters of deficiency issued over the last three fiscal years are displayed in the table below to show the trend over time.
| Fiscal Year | Number of Reports Reviewed | Number (and Percentage) of Reports Deemed Incomplete | Number of Letters of Deficiency Issued |
|---|---|---|---|
| 2012–2013 | 1490 | 213 (14%) | 107 |
| 2013–2014 | 1186 | 139 (12%) | 108 |
| 2014–2015 | 1607 | 172 (11%) | 81 |
Prosecution
In 2006, Health Canada investigated Compagnie de tabac Dynasty Inc. for alleged failure to comply with the requirements set out in the Tobacco Reporting Regulations. On April 17, 2007, 35 charges were filed before the Court of Québec against the company and its majority shareholder. The trial has been postponed several times and is still ongoing.
Litigation
Canadian tobacco control legislation has been challenged by members of the tobacco industry for most of the period since 1988. The most recent case involves the Tobacco Products Labelling Regulations (Cigarettes and Little Cigars), which were challenged by Imperial Tobacco Canada and JTI-Macdonald in April 2012. The case is still before the courts.
Conclusion
Despite decades of effort, there are still over 5 million tobacco users in Canada (including 4.2 million current smokers).
Canada’s Tobacco Act is a key component of the Federal Tobacco Control Strategy. Health Canada actively monitors compliance with the Tobacco Act and its regulations and responds to non-compliance.
In fiscal year 2014–2015, a non-compliance rate of 12% was observed at retail. Retail establishments are identified as non-compliant when at least one instance of non-compliance is observed for that establishment.
With respect to the importing and manufacturing sector, non-compliance was mainly the result of a lack of proper disclosure of information (11% of reports deemed incomplete) and inadequate labelling of tobacco products (9% of analyzed samples deemed non-compliant).
The Tobacco Act recognizes the inherent harmful nature of tobacco products; even when tobacco manufacturers comply with all the provisions of the Tobacco Act, their products remain harmful.
Key Priorities for fiscal year 2015–2016
Health Canada’s key priorities with respect to compliance and enforcement activities in relation to the Tobacco Act for fiscal year 2015–2016 were:
- To continue to promote compliance with the Tobacco Act and its regulations and actively monitor the market.
- To promote and monitor compliance with the Order Amending the Schedule to the Tobacco Act, in force as of December 2015, which prohibits the manufacture and sale of certain types of cigars that contain prohibited additives, including flavouring additives.