New Health-Related Labelling for Tobacco Products: Consultation Summary

Download the entire report
(PDF format, 1.55 MB, 20 pages)
Organization: Health Canada
Published: August 2019
Pub.: 190112
Table of Contents
Executive Summary
In October, 2018 Health Canada published “New Health-Related Labelling for Tobacco Products: Document for Consultation” and launched a public consultation on this document. This process sought feedback on areas Health Canada is exploring for renewed health labelling for tobacco products including labelling on cigarettes, labelling content and design, labelling size and placement, labelling for tobacco products that do not currently require labels, labelling rotation, and other considerations.
A total of 141 submissions were received from provincial and territorial governments, national and regional non-governmental organizations (NGOs), health associations, regional health authorities, academia, the general public and the tobacco industry.
Overall, there was considerable support for the approaches put forward in the consultation document. Most respondents were supportive of new initiatives such as displaying health messages directly on cigarettes themselves, requiring health labelling on the packaging of all tobacco products and linking the information on tobacco packages by theme. There was also support for extending the 75% health warning coverage on cigarette and little cigar packages to other tobacco products and for establishing minimum size requirements for health warnings for these same products.
Suggestions were received for the design and placement of quitline and website information as well as for health information messages and toxic statements, in order to maximize their noticeability. A number of ideas for the content and design of labelling directly on cigarettes were also received.
Responses received from the tobacco industry were largely in opposition to the approaches put forward in this consultation. Concerns were primarily about a perceived lack of evidence to support the approaches as well as anticipated technical and logistical difficulties in implementing the labelling changes under consideration.
Comments received through this consultation will be carefully considered as Health Canada continues to develop new health-related labelling for tobacco products.
Introduction
Tobacco use in Canada is a deadly and costly problem. It causes dozens of diseases in people who use tobacco as well as those exposed to second-hand smoke. In fact, tobacco use is the leading preventable cause of premature death in Canada. Every year, more than 45,000 Canadians die from illnesses caused by smoking; that is about one Canadian every 12 minutes. In a single year, smoking costs in Canada are over $6.5 billion for direct health care and $16.2 billion in combined health and economic costs.
Health-related labelling on tobacco packages is effective at informing Canadians and raising awareness of the health hazards and health effects of using tobacco products. The current labelling requirements for cigarettes and little cigars have been in place since 2011 while those for most other tobacco products have been in place since 2000.
On October 26, 2018 Health Canada released “New Health-Related Labelling for Tobacco Products: Document for Consultation” and launched a 70-day online consultation period. Interested stakeholders and the general public were invited to submit feedback on new approaches under consideration for:
- Labelling on Cigarettes;
- Labelling Content and Design;
- Labelling Size and Placement;
- Labelling for All Tobacco Products that Do Not Currently Require Labels;
- Labelling Rotation; and
- Other Considerations.
Who Responded
A total of 141 submissions were received in response to the consultation. Respondents included a mix of provincial and territorial governments, national and regional non-governmental organizations (NGOs), health associations, regional health authorities, academia, the general public and the tobacco industry. Please see below for a breakdown of respondents by type:
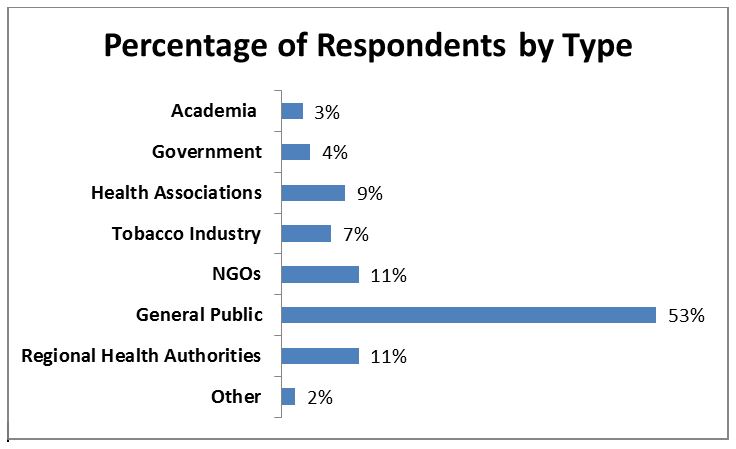
Figure 1: Percentage of Respondents by Type
The figure depicts the percentage of respondents to the consultation on new health-related labelling for tobacco products by stakeholder group.
| Stakeholder Group | Percentage of Submissions |
|---|---|
| Academia | 3% |
| Government | 4% |
| Health Associations | 9% |
| Tobacco Industry | 7% |
| NGOs | 11% |
| General Public | 53% |
| Regional Health Authorities | 11% |
| Other | 2% |
What We Heard
The consultation asked a total of 37 questions on the six distinct topic areas listed previously. Extensive feedback was provided across all topics.
Overall, there was considerable support for the approaches Health Canada is considering for new labelling on tobacco products. Many creative suggestions were received in all areas related to the consultation. A summary of responses for each of the areas under consideration follows.
Due to the nature of the submissions received from the tobacco industry, they are summarized in a separate section below.
1) Labelling on Cigarettes
Currently: There is currently no health labelling on cigarettes themselves.
Approach Under Consideration: Health Canada is considering introducing requirements for Health Warnings directly on cigarettes. All elements of design, such as size, wording, font and colour are being considered.
The vast majority of respondents (120)Footnote 1 supported the approach of displaying health messages directly on cigarettes. Some respondents (2) voiced their opposition to this idea. Suggestions were received for these messages to be short and concise, with information primarily related to the health impacts of smoking. Including pictograms or symbols indicating hazardous or harmful constituents was also suggested by a number of respondents.
Examples of the types of messages suggested included:
- Causes Cancer
- Smoking Kills
- Smoking Causes Heart Disease
- Emphysema
Conversely, some respondents suggested using motivational messages that encouraged smoking cessation.
Respondents suggested using a font that is noticeable and impactful, such as large type in contrasting colours to a white cigarette, like red or black. Another suggestion was to use different colours for different types of messaging, where one colour would be used for health warnings and another colour for positive cessation messages.
Various options for where to place information on cigarettes were put forward, including on the filter, on the cigarette shaft, and written along the length of the cigarette shaft.
One respondent suggested that warnings on cigarettes could help combat smuggling and contraband cigarettes.
The two respondents who were opposed to the approach of displaying messages on cigarettes felt that doing so would be disrespectful towards people who smoke.
2) Labelling Content and Design
Health Information Messages
Currently: Current Health Information Messages focus mainly on the benefits of quitting and provide tips to help people quit. They are either printed on the package or on a leaflet inserted into the package.
Approach Under Consideration: Several options are being explored for maximizing the noticeability of Health Information Messages, including increasing the size of leaflets inserted into packages, requiring them to be stuck onto packages so that they cannot be thrown away, and changing where Health Information Messages are printed on the inside of packages.
There was support expressed for Health Information Messages (HIMs) to have a greater presence on the inside of tobacco packages and for them to be either printed directly on packaging or fixed to packaging so that they could not be removed (12).
Suggestions received regarding the placement of new HIMs in order to maximize the noticeability of the information included putting them on all areas where the user will see them, on both sides of the cigarette package, on the removable foil inner liner of cigarette packages or on the inner flap. It was also suggested that HIMs be sized and affixed inside packaging in a way that does not interfere with the utility of the package. Others commented that HIMs should be located in front of the cigarettes inside the pack (rather than behind them) and that inserts should not be allowed to be folded which prevents them from being seen at a glance.
When asked about topics for future HIMs, some commenters recommended using a mix of positive messages combined with negative health warnings. It was also suggested that some HIMs be tailored to various sub-populations such as Indigenous groups, the LGBTQ+ community and people with mental illness. Specific suggestions for topic areas included:
- Tobacco-related health impacts (e.g. lung diseases, heart diseases, oral disease)
- Effects of second-hand smoke on others (e.g. babies, children, pregnant women and pets)
- Smoking cessation information (e.g. encouragement, resources for quitting, managing relapses)
- Benefits of being smoke-free (e.g. short and long-term health impacts, financial)
- Impact of illnesses on quality of life
- Addiction to nicotine and its effects
- Provincially targeted information (e.g. eligibility for nicotine replacement therapy)
- Personal testimonials about smoking and experiences with quitting
- Harm reduction messages (e.g. reduced risk of vaping products)
- Health risks of co-use of tobacco with other inhaled products (e.g. nicotine, cannabis)
- Negative impacts of tobacco use on the environment
Toxic Statements (Includes Toxic Emissions Statements and Toxic Constituents Statements)
Currently: Toxic Statements are required on packages of cigarettes and little cigars. Certain other tobacco products display a list of some of the toxic emissions or the amount of toxic constituents found in the product.
Approach Under Consideration: Health Canada is considering updating requirements related to current Toxic Statements. This includes considering changes to the messages, design and location of the Toxic Statements.
Suggestions for making new Toxic Statements (TSs) easier to notice and read than current TSs included having statements that are concise as well as written in plain language. It was also suggested that large, bold font or symbols related to the chemical toxicity, health risks, and environmental hazards of toxic components be used. Bolding the names of toxic substances and their adverse health effects, and using colours suitable for individuals with colour blindness were also offered as suggestions.
Regarding the placement of new TSs that would serve to make them even easier to notice and read, options offered were to maintain their current location on the side of cigarette packs, place them on the inner flap, or on the foil inner liner.
Respondents offered the following ideas for consideration for future TSs:
- Give the number of chemicals found in tobacco products
- Avoid referring to chemicals that are too commonly found in everyday life
- Continue to use statements that mention ‘tobacco smoke’ first
- Clarify the nature of dangerous substances (e.g.
‘Tobacco smoke contains arsenic, which is poisonous’) - Be specific about how chemicals impact health (e.g. their effects on specific body parts, how severe the damage would be and how it is caused)
- Make use of phrases such as ‘inhaling tobacco smoke’, ‘releases chemicals into your body’ and ‘any level of exposure’
- Personalize messages to refer to ‘your body’ or ‘your death’
- Use direct statements rather than ‘may’ or ‘can’
Connecting Labelling Elements
Currently: There is currently no requirement for the various health messages on tobacco products and packages to relate to one another.
Approach Under Consideration: Since 2012, Australia has connected labelling elements on tobacco packages so that they have messages that relate to one another. Similar measures to those in Australia could be implemented for future labelling in Canada.
The respondents who commented on this topic (43) all supported the concept of linking the information on tobacco products and packages by theme, particularly for information that was new or that was about complex risks. There were several suggestions related to linking the information by theme that respondents felt would improve the understanding of the information presented. These included:
- Using this as an opportunity to explain how smoking leads to specific health conditions
- Including additional resources (e.g. websites, toll-free quitline) that can be accessed for further depth of information
- Using connecting labelling elements for major health risks that are common for all people
- Using a mix of effective current health themes and new health themes
- Providing themes that appeal to a variety of audiences, such as different age groups
- Using themes that focus on diseases or on demographic concerns
Quitline Information
Currently: Toll-free quitline and web site information is currently on Health Warnings for cigarettes and little cigars, as well as on some Health Information Messages. Other products do not require quitline information.
Approach Under Consideration: Health Canada is considering changes to the size, colour, and placement of quitline and website information on labelling to maximize its noticeability. It is also considering adding quitline and website information to other tobacco products, such as smokeless, heated tobacco, and fine cut tobacco products.
A common suggestion (30) was that the quitline and website information should be larger than its present size, possibly even occupying space beyond the 75% that is currently required for the Health Warnings (HWs) on cigarettes. Increasing the prominence of the information not only through size but also through the use of imagery, bold and bright font, contrasting colours and placement on more prominent locations on packages were also proposed.
Several ideas for improving how quitline and website information is shown on tobacco packages were also put forward, including a QR code (short for "quick response" code - a type of two-dimensional barcode) was the most frequent suggestion (31). Others suggested including a more memorable URL/phone number and displaying related smartphone applications as well as connections to social media.
3) Labelling Size and Placement
Percentage of Coverage of Health Warnings on Tobacco Products Other than Cigarettes and Little Cigars
Currently: Health Warnings on cigarette and little cigar packages must cover at least 75% of the two largest sides of the package or primary display area. For certain other tobacco products, Health Warnings must either cover up to 50% of the main surface or meet size requirements (that are less than 50%), depending on the type of product. Only toxic constituent amounts are required on packages of smokeless tobacco.
Approach Under Consideration: Health Canada is considering expanding the 75% Health Warning coverage requirements for cigarettes and little cigars to other tobacco products.
The vast majority of respondents (42) to this section of the consultation agreed that the amount of space required for HWs should be the same for all tobacco products and that this size should be at least 75% of the package, as is the case for cigarettes and little cigars. Three respondents did not agree that the space requirements should be the same for all products. Two suggested that HW sizes should be at least 75% for all tobacco products except for cigarettes which should have HW up to 90%, and one suggested that the space occupied by HWs should be modified to fit individual package space.
Minimum Size of Health Warnings on Tobacco Products Other Than Cigarettes and Little Cigars
Currently: Labelling requirements for tobacco products other than cigarettes and little cigars are varied, with minimum size requirements (in cm2) applying only to a small number of package types.
Approach Under Consideration: Health Canada is considering establishing a minimum size for Health Warnings on tobacco products other than cigarettes and little cigars. Minimum sizes for Health Warnings for products other than cigarettes and little cigars have also been adopted in Australia and the European Union.
All respondents to this section of the consultation (41) supported the approach of having minimum sizes for HWs on tobacco products other than cigarettes and little cigars.
One suggestion was for Health Canada to have minimum sizes for health warnings and for these sizes to align with the requirements of other countries, such as Australia and the European Union, which have also adopted plain and standardized appearance measures.
4) Labelling for All Tobacco Products that Do Not Currently Require Labels
Currently: Some tobacco products, such as heated tobacco products, water pipe tobacco and blunt wraps, are not currently covered by health labelling requirements.
Approach Under Consideration: Health Canada is considering extending health labelling requirements to all packages of products containing tobacco, including products not currently subject to existing labelling requirements.
All respondents (105) voiced their support for requiring health labelling on the packaging of all tobacco products. There were no responses opposing this approach. There was considerable support for including all types of labelling (HWs, HIMs and TSs), and for including image-based HWs on all packages of products containing tobacco.
It was suggested that messages be developed that are specifically tailored for each product type and that are relevant to the different health risks associated with various products. A common suggestion was for warnings to be developed specifically for heated tobacco products.
Respondents (26) suggested that in order to allow for the design of warnings that are appropriate for new tobacco products, manufacturers should be required to give three years' notice before a new product is marketed for which a mandatory warning does not already exist.
5) Labelling Rotation
Currently: Health messages on tobacco products are not currently required to rotate after a specified period of time.
Approach Under Consideration: Heath Canada is considering implementing several rotating suites of health labelling for tobacco products. Some research suggests that the ideal rotation time between sets of labels is 12–18 months, and the most common rotation period for countries appears to be 12 months per set.
When asked about the ideal time period for rotating health labelling on tobacco products, respondents (14) suggested periods around 12 months, with some suggesting 12 months or less and another suggesting a longer period of up to 12-36 months, citing World Health Organization recommendations.
With regards to the number of labels and the number of sets for rotation, it was suggested by respondents (34) that there should be 16 labels per set. Respondents (32) also suggested that there should be a total of three sets.
It was frequently suggested (24) that the number of HIMs per rotation be increased beyond their current number (there are currently eight HIMs). It was also suggested that combining older health themes with new health themes may strengthen their overall credibility and effectiveness.
Additional measures offered included tracking and monitoring the labels for effectiveness, ensuring that enforcement of label rotation is feasible, and that messages resonating with the greatest number of people who smoke are on higher rotation than others.
6) Other Considerations
In addition to the feedback on the specific elements discussed above, respondents were encouraged to provide additional comments and suggestions for improving labelling for tobacco products.
Some respondents (3) suggested tailoring messages to other high-use populations such as those with mental illness, those living in poverty and members of the LGBTQ+ community. It was also suggested (3) that tobacco health labelling be culturally appropriate for First Nations populations and not reinforce existing stigmas and stereotypes. This includes the recognition of the traditions of tobacco use in First Nations cultural practices and the high rates of commercial tobacco use among this population.
Respondents (28) suggested that a warning be created that highlighted the association between smoking and developing or dying from breast cancer.
Respondents (30) suggested there be the adoption of new legislation that allow Health Canada to make changes to labelling administratively, without need for regulatory changes as is currently the case. Respondents hoped that this could result in more frequent updates to health labelling on tobacco products in Canada.
While not directly related to this consultation, many respondents (73) recommended that the absolute size of HWs on cigarette packs be 4468 mm2, as is required by the province of Quebec. Conversely, an additional suggestion was for federal regulations to require the HW size for cigarettes and little cigars to be different from Quebec’s size regulations, as this differentiation would facilitate the job of inspectors.
While also not related to this consultation, respondents (28) suggested that Health Canada develop specific warnings for the packaging of vaping products. Theproposed Vaping Products Labelling and Packaging Regulations and the accompanying Regulatory Impact Analysis Statement were pre-published in the Canada Gazette, Part I, on June 22, 2019.Footnote 2 The proposed Vaping Products Labelling and Packaging Regulations would require that all vaping substances be labelled with a list of ingredients. In addition, vaping products containing nicotine would be required to display a standardized nicotine concentration statement and a health warning about the addictiveness of nicotine. The proposed regulations would also mandate child-resistant containers and a toxicity warning.
Tobacco Industry Responses
A total of 10 submissions were received from the tobacco industry. These included large multinational tobacco corporations, a First Nations tobacco product manufacturer, representatives from the cigar and smokeless tobacco industries, and a tobacco industry- funded coalition.
Overall, their responses were generally in opposition to the approaches under consideration. Respondents (3) indicated that they felt there was a lack of strong evidence to support these approaches or to justify their implementation, particularly for labelling directly on cigarettes and for requiring larger health warnings.
Respondents (4) raised the Tobacco Products Regulations (Plain and Standardized Appearance) (PSAR) for tobacco products. It was felt that there should be a longer period between the implementation of PSAR regulations, which were registered on April 23, 2019, and any new labelling requirements in order to allow for a clear evaluation of the impact of PSAR requirements. Concerns were also raised about the heavy burden on industry transitioning to meet PSAR requirements and new labelling requirements.
Concerns regarding the implementation of new health labelling requirements included the cost of changing printing machinery, making adjustments to inks and other printing processes, as well as packaging modifications. It was felt that connecting labelling elements and the rotation of labels would be challenging as a result of the complexity of supply chains, labelling and packaging processes, as well as the large number of different package types, particularly for cigars.
There was opposition to increasing the size of HWs as this was considered excessive and unnecessary. Respondents said that size requirements should not be the same for all tobacco products, but that HW sizes and messages should be based on the level of risk of different products. One large multi-national company supported the approach of extending the 75% HW size requirement to other combustible products, and to labelling all tobacco/nicotine products but echoed that HW sizes and messages should be based on level of risk. Adapting to the diversity of package types was also a concern, but primarily for cigars.
Respondents (2) advocated for the government to recognize heated tobacco products (HTPs) as less harmful than combustible tobacco products and to regulate their labelling and packaging accordingly. It was suggested that Health Canada should recognize and communicate the relative risk and harm reduction potential of HTPs and smokeless tobacco. Respondents stated that regulations in promotion, packaging and labelling should reflect the risk continuum of different tobacco products.
Labelling rotation was also not supported by the tobacco industry respondents as it was considered to be unnecessary, overly disruptive to businesses and not feasible due to slow retail turnover of certain products. If implemented, one submission suggested that rotation time periods should be long and that the number of labels per rotation should be small.
Submissions from the cigar industry argued that cigars are important to Canadian culture and that the industry should be protected. They stated that cigars should not be generalized with other tobacco products and that there should be a distinction made between different sizes of cigars. It was felt that the retail market for cigars is small and that changing HWs is unsuitable and not required.
There was some support for refreshing health warning content and for continuing to require quitline information. However, there was some opposition to including quitline information on the grounds that there is not adequate space on smaller packages. A submission from the smokeless tobacco industry opposed the inclusion of quitline information until cessation services tailored for smokeless tobacco users are available.
With regards to having health messaging placed directly on cigarettes, respondents (2) expressed concern about the potential toxicity of the ink during combustion and that this could add additional health risks to smoking.
Conclusion
Health Canada would like to thank all of those who submitted their feedback to this consultation. Comments received as part of this consultation will be taken into careful consideration in the development of new health-related labelling for tobacco products.
Footnotes
- Footnote 1
-
The numbers shown in brackets indicate the number of respondents to a particular question.
- Footnote 2
-
The proposed Vaping Products Labelling and Packaging Regulations are available online - http://gazette.gc.ca/rp-pr/p1/2019/2019-06-22/html/reg4-eng.html