Guidelines for Canadian Drinking Water Quality: Guideline Technical Document – Chromium

Download the alternative format
(PDF format, 835 kB, 91 pages)
Organization: Health Canada
Type: Guidelines
Date published: 2018-08-17
Related Topics
Table of Contents
- Part I. Overview and Application
- 1.0 Guideline
- 2.0 Executive summary
- 3.0 Application of the guideline
- Part II. Science and Technical Considerations
- 4.0 Identity, use and sources in the environment
- 5.0 Exposure
- 6.0 Analytical methods
- 7.0 Treatment technology
- 8.0 Kinetics and metabolism
- 9.0 Health effects
- 10.0 Classification and assessment
- 11.0 Rationale for guideline
- 12.0 References
- Appendix A: List of abbreviations
Part I. Overview and Application
1.0 Guideline
A maximum acceptable concentration (MAC) of 0.05 mg/L (50 µg/L) is established for total chromium in drinking water.
2.0 Executive summary
Chromium occurs naturally in small amounts in rocks and soils, some of which is released into the aquatic environment through weathering and erosion processes. More than 70% of chromium in the environment comes from anthropogenic sources, such as non-ferrous base metal smelters, refineries, leather tanning industries, urban storm water runoff, effluent streams from pulp and paper mills and discharges from thermal generating stations. Chromium can exist in nine different oxidation states, with the trivalent [Cr(III)] and hexavalent [Cr(VI)] forms being the most common in the environment.
This guideline technical document reviews and assesses all identified health risks associated with chromium in drinking water. It incorporates new studies and approaches and takes into consideration the availability of appropriate treatment technology. Based on this review, the drinking water guideline for total chromium is a maximum acceptable concentration (MAC) of 0.05 mg/L (50 µg/L).
2.1 Health effects
Chromium toxicity in humans varies depending on the form of the compound, its oxidation state and the route of exposure. Studies show that there is little or no toxicity associated with the trivalent form of chromium, whereas hexavalent chromium compounds are classified as carcinogenic to humans by the inhalation route of exposure, based on sufficient evidence in both humans and animals.
The critical health effect on which to establish a guideline for chromium in drinking water is diffuse hyperplasia of the small intestine, as it is the most sensitive endpoint and a precursor of tumour formation. The physiologically based pharmacokinetic (PBPK) models for mice and humans and benchmark dose (BMD) modelling were used to determine appropriate external doses in humans from animal data. The MAC for chromium in drinking water is based on the health effects of Cr(VI) and considers the cancer and non-cancer effects together.
2.2 Exposure
Background levels of chromium in surface water and groundwater are a direct function of regional geology, mineral weathering processes, sediment loading rates and precipitation patterns. Average concentrations of total chromium in uncontaminated surface waters are generally below 1 µg/L. Chromium levels in groundwater can be significantly higher than levels in surface water.
Canadians can be exposed to total chromium through its presence in food, drinking water, dust, soil and air. The single most important source of exposure to Cr(VI) is drinking water. In order to be most protective of human health, this assessment assumes that all the chromium present in drinking water is in the form of Cr(VI).
2.3 Analysis and treatment
There are several approved analytical methods available to measure total chromium (i.e., the sum of Cr(III) and Cr(VI)) in drinking water at levels well below the MAC.
Given the presence of oxidants and disinfectants in treated water, Cr(III) is likely to be oxidized to Cr(VI) after treatment. For this reason, it is important to ensure the removal of both chromium species. At the municipal level, the best available technologies for the treatment of total chromium are coagulation/filtration, ion exchange, reverse osmosis and lime softening. Reduction/coagulation/filtration, weak-base ion exchange and strong-base ion exchange are effective technologies for removing Cr(VI) from drinking water .
At the residential level, drinking water treatment technologies able to be certified to NSF International (NSF) standards for reduction of total chromium, as well as Cr(VI) and Cr(III) individually, include adsorption, reverse osmosis and distillation. It is important to note that reverse osmosis and distillation systems should be installed only at the point of use, as the treated water may be corrosive to internal plumbing components.
3.0 Application of the guideline
Note: Specific guidance related to the implementation of drinking water guidelines should be obtained from the appropriate drinking water authority in the affected jurisdiction.
The MAC for total chromium is based on the health effects of Cr(VI), which is more toxic than Cr(III). Utilities need to ensure that treatment adequately removes both forms of chromium, as the water chemistry in the distribution system will encourage the oxidation of Cr(III)to the more toxic form.
For drinking water supplies that occasionally experience short-term exceedances above the guideline values, it is suggested that a plan be developed and implemented to address these situations. For more significant, long-term exceedances that cannot be addressed through treatment, it is suggested that alternative sources of drinking water be considered.
3.1 Monitoring
Utilities should characterize their source water to assess chromium concentration. In source waters where chromium is present at levels exceeding the MAC, quarterly monitoring of surface water and semi-annually monitoring of groundwater should be conducted. Utilities that treat their water to remove chromium need to frequently monitor the treated water to ensure that the treatment process is effective and to make necessary adjustments to the process to maintain chromium concentrations below the MAC. If the treatment system performs reliably and consistently, utilities may reduce monitoring frequency.
As with other inorganics, chromium can be accumulated in distribution systems and later released. Consequently, monitoring should also be conducted throughout the distribution system for systems in which chromium is present in the source water. Since the stability of metals accumulated in the scales is unpredictable, it is difficult to establish a monitoring program for chromium in distribution system. Factors that influence chromium accumulation and release in distribution system (presence of galvanized pipes, co-occurrence of manganese in the deposits, low pH conditions and hydraulic disturbance) could be used as indictors of when and where to monitor for chromium releases in distribution systems. Where total chromium is not detected, or is detected below the MAC, in the source water, utility may conduct less frequent monitoring.
Analytical sample preservation and dechlorination procedures should be done at the time of sample collection. The samples for total chromium need to be acid digested before analysis, regardless of turbidity.
Part II. Science and Technical Considerations
4.0 Identity, use and sources in the environment
Chromium is a transitional metal (Group 6 of the periodic table). It is typically present only in trace amounts, with an average concentration of 100 mg/kg in Earth’s crust; it ranks 21st in elemental abundance (Hammond, 2002). More than 40 chromium-containing minerals have been identified, with chromite (FeCr2O4) being the most common in crustal rock (Shiraki, 1978). Chromium occurs naturally in small amounts in rocks and soils as relatively inert Cr(III) solid phases. It is released into the aquatic environment in limited quantities by weathering and erosion of these materials. Windblown dusts are the primary natural source of chromium in Earth’s atmosphere, and wind erosion of prairie soils may be a significant natural source of airborne chromium in central Canada (Nriagu, 1990). Volcanic emissions, sea salt aerosols, dusts from wildfires and vegetative debris are other natural sources of chromium in the atmosphere.
The only commercial source of chromium is chromite ore. Approximately 95% of the world’s chromium resources are geographically concentrated in Kazakhstan and South Africa (Cary, 1982), but ore-grade chromite has been identified at more than 250 locations in Canada (EMR, 1989). The principal deposits occur in Quebec, Ontario, British Columbia, Manitoba and Newfoundlandand Labrador (Phillips, 1988). More than 70% of chromium in the environment comes from anthropogenic sources, such as non-ferrous base metal smelters, refineries, leather tanning industries, urban storm water runoff,effluent streams from pulp and paper mills and discharges from thermal generating stations (Merian, 1984; Environment Canada et al., 1988; OMOE, 1991a, 1991b; MacLatchy, 1992). Ferrochromium production is the most important industrial source of atmospheric chromium (U.S. EPA, 1984b).
Chromium is mainly used in the metallurgical industry (production of ferrochromium alloys, such as stainless steel, high-speed steel, alloy cast irons and non-ferrous alloys; Stoecker, 2004), in electrical applications (copper–chromium; Nriagu, 1988), in the automobile industry (chromium alloys in the form of stainless steel components, catalytic converters, chrome trim, and other control and decorative systems; Nriagu, 1988) and wood preservation (copper chrome arsenate, allowed in Canada for industrial uses and still used for the treatment of wooden poles; Health Canada, 2005). Chromium is also used in the production of fungicides, drilling muds, water treatment, textiles, catalysts, synthetic rubies for lasers, chromium dioxide magnetic tapes, clinical medicine (labelling of red blood cells), toner for copying machines, montan wax manufacturing and vitamin K manufacturing and as a mordant in wool dyeing, photography and manufacturing of activated carbon (Taylor et al., 1979; U.S. EPA, 1984a; Nriagu, 1988; ATSDR, 2012).
A search of the national pollutant release inventory database (NPRI, 2012) yielded 358 facilities reporting chromium (and its compounds) releases across Canada in 2012. On-site releases totaled 81 tonnes with 2.4 tonnes released into water. A total of 26,568 tonnes were disposed of on-site while 3,138 tonnes and10,346 tonnes were sent off-site for disposal and recycling, respectively.
Although chromium is a naturally occurring element, elemental chromium (Cr(0)) does not appear in nature (Shupak, 1991); it is found complexed with oxygen, iron or lead (Williams, 1988). Chromium can exist in nine different oxidation states, from −II to +VI, but the common valence states are +II, +III and +VI (Hammond, 2002). Because of their stability in the environment, the trivalent [Cr(III)] and hexavalent [Cr(VI)] forms are the most common (Cary, 1982; U.S. EPA, 1984a; WHO, 1988; Shupack, 1991).
Cr(III) is generally considered to be the most thermodynamically stable oxidation state under ambient redox conditions. It is a positively charged ion that has a strong tendency to form hexacoordinate octahedral complexes with a variety of ligands (oxygen, nitrogen or sulphur atoms). These stable complexes can prevent the precipitation of Cr(III) above pH 5–6, where it would otherwise precipitate (U.S. EPA, 1990).
Cr(VI) is not thermodynamically stable. It is a strong oxidizing agent, existing only as tetrahedral oxo species such as chromium oxide (CrO3), chromyl chloride (CrO2Cl2) and chromate ion (CrO42−) (Nieboer and Jusys, 1988). It is produced during the reduction of chromite ore to obtain chromium metal (WHO, 1988; Shupack, 1991). The principal source of Cr(VI) in the environment is anthropogenic pollution. Cr(VI) was thought to rarely occur naturally due to its affinity for organic matter and other reducing substances (U.S. EPA, 1984c; Jaworski, 1985; Bartlett and James, 1988; Hammond, 2002); however, more recent studies have demonstrated the occurrence of Cr(VI) in the absence of anthropogenic sources of chromium, potentially due to oxidation of Cr(III) to Cr(VI) under certain geological conditions (Oze et al., 2007; Kaprara et al., 2015). In solution, Cr(VI) exists as an anion and is thus quite mobile in the environment; the dissolved species of Cr(VI) are hydrogen chromate (HCrO4−), dichromate (CrO2−, more commonly presented as Cr2O72−) and chromate (CrO42−) (Saleh et al.,1989). For example, chromium oxide, the ammonium salts of chromic acid and the alkali salts of chromic acid are readily soluble in water (Theopold, 1994).
Cr(VI) is the dominant form of dissolved chromium in surface waters. At the normal pH of drinking water (around pH 7), Cr(III) is generally not soluble (Costa and Klein, 2006). The proportion of Cr(III) could nevertheless be elevated in some deep, anoxic waters and in waters receiving direct discharges of Cr(III)-containing wastes. In contrast, nearly all of the chromium in soils (excluding those contaminated with Cr(VI)), sediments (excluding those immediately below the interface with overlying aerobic waters) and biological tissues is likely to be present as Cr(III) (Anderson, 1981; Bartlett and James, 1988; Nieboer and Jusys, 1988; Nriagu et al., 1993).
The main physicochemical properties of a selection of chromium coumpounds are presented in Table 1. As the compounds present in the environment are not documented, the compounds presented in Table 1 are those used to study the toxicity of ingested chromium (see Section 9.0).
5.0 Exposure
Canadians can be exposed to chromium through its presence in food, drinking water, dust, soil and air. For Cr(VI), drinking water is the main source of exposure, followed by food, dust, air and soil.
5.1 Water
Background levels of chromium in surface water and groundwater aquifers are a direct function of regional geology, mineral weathering processes, sediment loading rates and precipitation patterns. Average concentrations of total chromium (including Cr(III) and Cr(VI) in dissolved and particulate phases) in uncontaminated surface and marine waters are generally below 1 µg/L (Erickson and Fowler, 1987; Mayer, 1988; Rossmann and Barres, 1988; Beaubien, 1993). Between 10% and 60% of the total chromium content in Canadian rivers may be present as dissolved Cr(VI). This range is based on measurements of filtered and unfiltered North American river water (Merritt, 1975; Gibbs, 1977; Campbell and Yeats, 1984; Allan, 1986; Kauss et al.,
| Name | Chromium (0) | Chromium (III) chloride | Chromium (III) oxide | Potassium chromate [Cr(VI)] | Sodium chromate [Cr(VI)] | Potassium dichromate [Cr(VI)] | Sodium dichromate dihydrate [Cr(VI)] |
|---|---|---|---|---|---|---|---|
| Synonym(s) | Chrome | Chromium trichloride | Chromic oxide Dichromium trioxide |
Potassium chromate Dipotassium chromate Chromic acid, dipotassium salt |
Chromic acid, disodium salt | Potassium dichromate Dipotassium dichromate Dichromic acid, dipotassium salt |
Dichromic acid, disodium salt, dihydrate SDD |
| CAS number | 7440-47-3 | 10025-73-7 | 1308-38-9 | 7789-00-6 | 7775-11-3 | 7778-50-9 | 7789-12-0 |
| Chemical formula | Cr | CrCl3 | Cr2O3 | K2CrO4 | Na2CrO4 | Na2Cr2O7 | Na2Cr2O7·2H2O |
| Relative molecular mass | 51.996 | 158.35 | 151.99 | 194.19 | 161.97 | 294.19 | 298.00 |
| Melting point | 1900 ± 10°C | ~1150°C | 2435°C | 975°C | 792°C | 398°C | 357°C |
| Boiling point | 2642°C | Decomposes at 1300°C | 3000°C | No data | No data | Decomposes at 500°C | Decomposes at 400°C |
| Density | 7.14 g/cm3 (28°C) | 2.87 g/cm3 (25°C) | 5.22 g/cm3 (25°C) | 2.73 g/cm3 (18°C) | 2.71–2.74 g/cm3 (temperature not reported) |
2.68 g/cm3 (25°C) | 2.35 g/cm3 |
| Water solubility | Insoluble | Slightly soluble in hot water | Insoluble | 62.9 g/100 g (20°C) 65.0 g/100 g (25°C) |
87.3 g/100 mL (30°C) | 15.1 g/100 g (25°C) | 272.9 g/100 g (20°C) |
| Log Kow | Not applicable | Not applicable | Not applicable | Not applicable | Not applicable | Not applicable | Not applicable |
| Henry's law constant at 25°C | Not applicable | Not applicable | Not applicable | Not applicable | Not applicable | Not applicable | Not applicable |
| Vapour pressure at 25°C | 1 mmHg (0.13 kPa) | No data | No data | 0 | No data | No data | No data |
CAS: Chemical Abstracts Service; Kow: octanol–water partition coefficient |
|||||||
1988) and data obtained from studies on the speciation of dissolved chromium in aerobic lake waters (Balistrieri et al., 1992; Johnson et al., 1992; Beaubien, 1993).
Canadian data on chromium levels in drinking water were provided by several provinces and territories. The vast majority of the samples analysed across the country were below the detection limits (DL) for chromium. Average detected values and maximum values are reported for each province or territory, when available.
For Prince Edward Island (PEI), of 7622 samples from private wells tested for total chromium from June 2005 to June 2010, 3 were above the DL of 50 µg/L, with concentrations of 60, 80 and 234 µg/L (PEI Department of Environment, Energy and Forestry, 2010).
In Newfoundland and Labrador, 3946 and 1910 drinking water samples were analysed for total chromium from surface water and groundwater sources, respectively, between 2004 and 2010. The average concentration of total chromium reported above the method detection limit (MDL) (n = 157, MDL = 1 µg/L) in surface water samples was 2 µg/L, with a maximum value of 13 µg/L. The average concentration of total chromium reported above the MDL (n = 417, MDL = 1 µg/L) in groundwater samples was also 2 µg/L, with a maximum value of 26 µg/L (Newfoundland and Labrador Department of Environment and Conservation, 2010).
In Nova Scotia, 118 raw and 292 treated water samples were analysed for total chromium between 2004 and 2009. In raw water samples, the average concentration of total chromium reported above the MDL (n = 12, MDL = 0.6–2.0 µg/L) was 2.5 µg/L, with a maximum value of 4 µg/L. The average concentration of total chromium in treated water reported above the MDL (n = 9, MDL = 1.0–2.0 µg/L) was 2.7 µg/L, with a maximum value of 5.0 µg/L (Nova Scotia Department of Environment and Labour, 2010).
In Quebec, 17 005 results for total chromium in drinking water were reported between 2005 and 2010, of which 14 263 were below the DL (DL = 0.1–30 µg/L). The average concentration of total chromium reported above the DL was 4 µg/L, with a total of 11 samples above 50 µg/L (Ministère du Développement durable, de l’Environnement et des Parcs du Québec, 2010). The maximum value reported for years 2005–2009 was 992 µg/L (Ministère du Développement durable, de l’Environnement et des Parcs du Québec, 2012).
In Ontario, 6101 results were reported for total chromium in drinking water between 2009 and 2014, of which 4038 were below the DL (DL = 0.6–5.0 µg/L). The average concentration of total chromium reported above the DL was 1.2 µg/L, with a maximum of 41.3 µg/L (OMOE, 2014).
In Manitoba, 220 raw and 212 treated water samples were analyzed for total chromium between 2009 and 2010. In raw water samples, the average concentration of total chromium in samples above the DL (n = 26, DL = 1 µg/L) was 3 µg/L, with a maximum value of 14 µg/L. In treated water samples, the average concentration of total chromium in samples above the DL (n = 19, DL = 1 µg/L) was also 3 µg/L, with a maximum value of 13 µg/L (Manitoba Water Stewardship, 2010).
In Saskatchewan, 2013 results were reported for total chromium in drinking water between 2002 and 2010, of which 1760 were below the DL (DL = 0.03–5.0 µg/L). The average concentration of total chromium reported above the DL was 5.4 µg/L, with a maximum of 29.0 µg/L (Saskatchewan Ministry of Environment, 2010).
In British Columbia, 645 facilities reported results for chromium levels in drinking water between 2004 and 2010. The data for the Greater Vancouver Regional District and member municipalities and the City of Abbotsford indicate total chromium levels below 1 µg/L from all their source waters. Analytical results from the most populated drinking water systems indicated a maximum chromium concentration of 5 µg/L (B.C. Ministry of Health, 2010).
In Yukon, 22 results were reported for total chromium in drinking water between 2007 and 2010, of which 15 were below the DL (DL = 0.2–5.0 µg/L). The average detected concentration of total chromium was 0.7 µg/L, with a maximum of 1.2 µg/L (Government of Yukon, 2010).
In the Northwest Territories, levels of total chromium in drinking water in 2010 (n=53) were all below the reportable detection limit (RDL) of 1 µg/L or 10 µg/L, except for four sites at 20 µg/L and two sites at the RDL of 1 µg/L (Government of the Northwest Territories, 2010).
The Ontario Drinking Water Surveillance Program for 2000–2002 reported a mean total chromium concentration of 1.4 µg/L in drinking water in Ontario (OMOE, 2004). A more recent Ontario survey of total chromium concentrations in unfiltered distributed drinking water (1997–2007) reported average concentrations ranging from ≤ 0.5 to 18.9 µg/L (n=52), from 1.08 to 1.73 µg/L (n=4), from 0.42 to 6.92 µg/L (n=49) and from 0.49 to 3.82 µg/L (n=83) in drinking water originating from groundwater, lake water, river water and surface water, respectively; the mean concentration was 2.0 µg/L (OMOE, 2008). These concentrations are similar to those measured, in 2005, at a Montréal drinking water treatment plant supplied from the St. Lawrence River (mean total chromium concentration: 1 µg/L; range: < 1–3 µg/L; Ville de Montréal, 2005). They are also similar to those documented in earlier monitoring programs (i.e.,< 2–5 µg/L, median 2.0 µg/L, in raw water from 71 cities across Canada in 1977, Méranger et al., 1981; and 0.51–18 µg/L, average 2.4–2.6 µg/L, for treated and distributed drinking water from over 110 sampling sites in Ontario in 1994–1995, McGrachan, 1996).
In the United States, drinking water data indicate that 71% of the population is exposed to chromium concentrations below 10 µg/L, and 29% receive drinking water containing chromium at concentrations between 10 and 100 µg/L; only 0.001% receives drinking water containing chromium at concentrations greater than 100 µg/L (U.S. EPA, 2003a). Another study reported that approximately 18% of the U.S. population is exposed to chromium concentrations in drinking water between 2 and 60 µg/L, and less than 0.1% of the population is exposed to concentrations between 60 and 120 µg/L (Hirose et al., 2002). Chromium concentrations were measured in 10 groundwater sources from California, Nevada and Oklahoma. The total chromium concentrations ranged from 1.9 to 48 µg/L, and virtually all the chromium was present as Cr(VI) (Najm et al., 2014).
Currently, the third Unregulated Contaminant Monitoring Rule (UCMR3) requires monitoring for total chromium and Cr(VI) in the raw water, at the entry points to the distribution system and in the distribution system. Since Cr(III) can transform into Cr(VI) in the distribution system due to the presence of oxidants, monitoring for Cr(VI) in the distribution system should be done at locations with maximum residence time. This is consistent with the monitoring goals for disinfection by-products.
Once available, the data from UCMR3 and other studies will be used to inform the best approach for sampling (U.S. EPA, 2012a). In the interim, the U.S. Environmental Protection Agency (U.S. EPA, 2014b) recommends that water systems with surface water sources collect samples quarterly and that ground water systems be sampled twice per year and that these samples (raw, entry point to the distribution system and distribution system) be collected on the same day.
Considering the whole data set, a concentration of 2.0 µg/L, based on the most recent survey (mean concentration in unfiltered distributed drinking water according to the Ontario Drinking Water Surveillance Program for 1997–2007; OMOE, 2008), is used to represent the total chromium concentration in Canadian drinking water.
All of the chromium in drinking water is assumed to be in the form of Cr(VI) (Sanexen, 2009). This conservative approach is supported by the fact that different forms of chromium can interconvert in water and in the human body, depending on the conditions. It is further supported by the redox chemistry of chromium, whereby Cr(VI) is expected to predominate in the dissolved fraction of oxygenated water or in drinking water disinfected with chlorine or chloramines (Brandhuber et al., 2004).
5.2 Food
Food is generally considered to be the main source of chromium exposure, except in situations where a population is living near a point source. Food has been found to contain chromium at concentrations ranging from < 0.0005 to 1.3 µg/g (UK Ministry of Agriculture, Fisheries and Food, 1985; Sloof, 1989; Anderson et al., 1992; Mann Testing Laboratories, 1992; Schuhmacher et al., 1993; UK Food Standards Agency, 1999; Ferre-Huguet et al., 2008; Jorhem et al., 2008; Rose et al., 2010; ATSDR, 2012). The highest concentrations (> 0.1 µg/g) have been found in meat, fish, seafood, cereal products, tea, black pepper, cheese, wheat germ and some fruits and vegetables (Toepferetal.,1973; UK Ministry of Agriculture, Fisheries and Food, 1985; Copat et al., 2012). However, total chromium levels in most fresh foods can be extremely low (vegetables, 0.02–0.05 µg/kg; fruits, 0.02 µg/kg; and grains and cereals, 0.04 µg/kg) (Fishbein, 1984). Beer, wine and spirits contain chromium concentrations of approximately 450, 300 and 135 µg/L, respectively (U.S. EPA, 1984a). Chromium may also be released during food preparation (stainless steel utensils), processing and packaging (Offenbacher and Pi-Sunyer, 1983; Maher, 2008).
Average daily exposure of Canadians to chromium was estimated on the basis of the most relevant Canadian results to date (1970 Nutrition Canada Survey) and the ranges of chromium concentrations obtained in a survey of 108 Canadian foodstuffs conducted in 1992 for Health Canada (Mann Testing Laboratories, 1992). This survey revealed that approximately half of the foodstuff samples analyzed contained chromium at levels below the DL, with concentrations ranging from < 0.004 to 0.100 µg/g. The estimated daily intake of chromium for the 7-month to 4-year, 5- to 11-year, 12- to 19-year and 20+-year age groups are 11.2, 15.0, 19.9, and 16.4 µg/day, respectively. These estimates were used to calculate the total daily intakes of Cr(VI) in the Canadian population, which are presented in Table 2. Although data on the speciation of chromium in all food items are not available, it is not expected that Cr(VI) will be found in food not contaminated by a local source (Sanexen, 2009). However, Cr(VI) was found to represent approximately 15% of total chromium in both white bread and whole wheat bread samples (Soares et al., 2010). In addition, it is not known if, in a region affected by sources of Cr(VI), local food could be enriched in Cr(VI) (e.g., atmospheric deposits on vegetables). For the purpose of calculating the intake of Cr(VI) from food, it is assumed that 10% of total chromium is in the form of Cr(VI) (Sanexen, 2009).
5.3 Air
The National Air Pollution Surveillance program measured mean atmospheric chromium concentrations of 3–9 ng/m3 (1987–1990; Dann, 1991), < 1–28 ng/m3 (1993; Dann, 2007) and 0.21–0.8 ng/m3 (2004–2007; Dann, 2007) in 13 Canadian cities. Concentrations of airborne total chromium as high as 1250 ng/m3 have been measured near point sources of chromium discharge (Environment Canada, 1991).
Generally, indoor concentrations of total chromium are related to and lower than outdoor concentrations. However, as a result of smoking, indoor air concentrations can be 10–400 times greater (up to 1000 ng/m3) than outdoor concentrations (WHO, 1996). The total chromium concentration of 0.8 ng/m3 is the highest mean value reported in samples from the ambient air of 13 Canadian cities collected from 2004 to 2007 (Dann, 2007). This concentration is used to calculate the total daily intakes of Cr(VI) for the Canadian population, which are presented in Table 2. Concentrations in indoor air were generally reported to be lower (but in the same order of magnitude) than concentrations in outdoor air. Thus, the selected value can be considered representative of average concentrations both indoors and outdoors. For the purpose of calculating the intake of Cr(VI) from air, it is assumed that 25% of total chromium is in the form of Cr(VI) (Sanexen, 2009).
5.4 Soil
Total chromium concentrations in soil vary greatly and depend on the composition of the parent rock from which the soil was formed. However, industrial activities, such as wood treatment with chromium-containing preservatives, may significantly increase the levels of chromium in soil.
The mean background concentrations of total chromium reported throughout Canada range from 13 to 78 mg/kg (range of individual values: 1–540 mg/kg) (McKeague et al., 1979; Choinière and Beaumier, 1997; B.C. Ministry of Environment, 2005). Similar concentrations were reported in soils from rural and agricultural areas (range of means: 15–85 mg/kg; range of values: < 0.5–510 mg/kg; Whitby et al., 1978; Soon and Abboud, 1990; Giroux et al., 1992; Gizyn, 1994; OMEE, 1994; Mermut et al., 1996; Pilgrim, 1996; Sharpe and Rasmussen, 1996; Haluschak et al., 1998) and from urban areas (range of means: 19–44 mg/kg; range of values: < 0.5–82 mg/kg; Gizyn, 1994; OMEE, 1994; Pilgrim, 1996; Kuja et al., 2000; Rasmussen et al., 2001; Penney, 2004). However, in areas contaminated by wood treatment activities, average chromium concentrations of 200–1760 mg/kg were reported, with a maximum individual value of 5280 mg/kg (Henning and Konasewich, 1984; Manitoba Environment and Workplace Safety and Health, 1989; Bamwoya et al., 1991). Naturally high concentrations of chromium (> 1000 mg/kg) have also been reported in soils associated with chromium-enriched serpentine bedrock in western Newfoundland (Roberts, 1980).
A mean concentration of 50 µg/g of total chromium in most uncontaminated soils across Canada can be used to estimate the intake of chromium from soil for the general Canadian population (Sanexen, 2009). As Cr(III) is the major form of chromium in most unpolluted soils (Bartlett and James, 1988), the proportion of Cr(VI) in soil/dust was set at 1% of the total chromium levels (Sanexen, 2009). This estimate is in agreement with the position of Bartlett and James (1988) that anthropogenic inputs should be suspected in soils containing Cr(VI) concentrations above a few tenths of a microgram per kilogram and with the findings of OMEE (1994), which reported Cr(VI) concentrations in soils of rural parkland ranging from < 0.5 to 0.9 mg/kg, with a 98th percentile concentration of 0.5 mg/kg.
In addition, exposure through house dust was estimated to be 6.1 µg/day for toddlers and 1.5 µg/day for the other age classes (based a mean total chromium concentration of 87 µg/g in house dust samples collected in 48 Canadian homes; Radimer et al., 2004). Similar chromium concentrations were reported in dust samples from 78 classrooms in California (median: 33.1 µg/g; 95th percentile: 72.8 µg/g; CARB, 2003).
5.5 Consumer products
The consumption of multivitamins may lead to an additional chromium intake of 500 µg/day, from chromium picolinate (Health Canada, 2009).
Higher than average chromium exposures can be estimated for smokers. Assuming mainstream exposure of 0.147 µg total chromium per cigarette, adults and teenagers smoking a pack of cigarettes (n = 20) per day would have an additional chromium intake of 0.0422 and 0.04952 µg/kg body weight (bw) per day, respectively (Sanexen, 2009). About 0.8–1.2% of the total chromium content of the cigarette is in the hexavalent form in the smoke (Sanexen, 2009), corresponding to a Cr(VI) exposure of 0.0003–0.0006 µg/kg bw per day. This exposure is 7–15 times higher than exposure due to inhalation of ambient air (as estimated in Table 2).
5.6 Multi-route exposure through drinking water
Given the physicochemical properties of chromium, a multi-route exposure assessment, as outlined by Krishnan and Carrier (2008), could not be performed. Exposure to chromium vapours while showering is not expected to occur, as chromium is not volatile. Paustenbach et al. (2003) experimentally determined that approximately 5–10 ng of Cr(VI) would be inhaled during a 10-minute shower with water containing Cr(VI) at a concentration of 1 mg/L, and bathing in water containing Cr(VI) at a concentration of ≤ 10 mg/L for 30 years would be similar to a continuous 30-year exposure to background Cr(VI) levels in outdoor air. Chromium penetration through skin is expected to be minimal (Section 8.1). When human volunteers were immersed for 3 hours in water containing Cr(VI) at a concentration of 22 mg/L, penetration was estimated to be less than 10% of daily ingestion of water, based on blood and urine samples (Paustenbach et al., 2003).
Thus, dermal and inhalation routes of exposure to chromium in drinking water were not considered to be significant in this assessment.
5.7 Total daily intake
Most available data on chromium levels in the environment pertain to total chromium. However, chromium in drinking water occurs mainly as Cr(VI), which is also the form used as the basis for the calculation of the health-based value (HBV). For this reason, the estimated total daily intakes are presented only for Cr(VI). In order to calculate the total daily intakes for Cr(VI) using the available exposure data, it was assumed that Cr(VI) represents 100% of total chromium in drinking water, 25% in air, 1% in soil/dust, 10% in food and 0% in breast milk, based on the findings of various reports (Sanexen, 2009). The estimated total daily intakes of Cr(VI) from drinking water, food, air, soil and dust for five age groups (0–6 months, 7 months– 4 years, 5–11 years, 12–19 years and 20+ years) in the Canadian population are shown in Table 2. Daily chromium intakes from consumer products were not estimated, as there are no available data on the proportion of the general population using these products. Reliable data were also not available to estimate distinct exposure levels for populations living in mineralized areas and those living in the vicinity of chromium-related industrial activities.
As presented in Table 2, the proportion of the total daily intake of Cr(VI) coming from drinking water represents 99%, 0%, 51%, 51%, 50% and 64% for the age groups non-breastfed infants 0–6 months of age, breastfed infants 0–6 months of age, 0.5–4 years, 5–11 years, 12–19 years and 20+ years, respectively. Based on these estimates, Sanexen (2009) suggested the use of 0.5 as an allocation factor to derive the HBV for chromium in drinking water. The proportion of Cr(VI) intake from drinking water for the adult population is estimated to be 64%. Because food is the second major source of exposure and assuming that 10% of total chromium in food is Cr(VI), exposure through food may represent up to 50% of the total daily intake (Sanexen, 2009). Hence, the allocation factor of 0.5 estimated for drinking water refers to the minimum contribution of drinking water to the total daily intake of Cr(VI) for Canadians.
| Age group | Estimated total daily intake of Cr(VI) (µg/kg bw per day) | ||||||
|---|---|---|---|---|---|---|---|
| Drinking waterFootnote a |
FoodFootnote b | AirFootnote c | SoilFootnote d | DustFootnote e | Total | % intake from drinking water |
|
| 0–6 months non-breastfed infantsFootnote f | 0.18 | 0 | 0.000 049 | 0.000 16 | 0.001 8 | 0.18 | 99 |
| 0–6 months breastfed infantsFootnote g | 0 | 0 | 0.000 049 | 0.000 16 | 0.001 8 | 0.002 0 | 0 |
| 7 months – 4 yearsFootnote h | 0.073 | 0.067 | 0.000 12 | 0.000 30 | 0.003 7 | 0.14 | 51 |
| 5–11 yearsFootnote i | 0.049 | 0.046 | 0.000 09 | 0.000 040 | 0.000 46 | 0.096 | 51 |
| 12–19 yearsFootnote j | 0.034 | 0.034 | 0.000 054 | 0.000 022 | 0.000 25 | 0.068 | 50 |
| 20+ yearsFootnote k | 0.042 | 0.023 | 0.000 045 | 0.000 018 | 0.000 21 | 0.065 | 64 |
Source: Adapted from Sanexen (2009). |
|||||||
6.0 Analytical methods
There are several analytical methods that are relevant for the quantification of total chromium and Cr(VI) in drinking water. However, only methods for the analysis of total chromium are approved by the U.S. EPA (2014a).
6.1 Total chromium
Total chromium is defined as the sum concentration of both Cr(III) and Cr(VI) (the most common oxidation states) in the dissolved and suspended fractions of a water sample and is analyzed using methods to determine total recoverable chromium.
The concentration of dissolved chromium is determined after filtration and acid preservation with nitric acid to a pH level below 2.0. For total chromium analysis of the dissolved and suspended fraction, the water sample is not filtered but is acidified to dissolve the suspended fractions. Current methods require an acid digestion when the turbidity of the acid-preserved sample is greater than 1 nephelometric turbidity unit (NTU). Following the preservation procedure, the sample is analyzed using inductively coupled plasma–atomic emission spectroscopy (ICP-AES), inductively coupled plasma–mass spectrometry (ICP-MS) or graphite furnace atomic absorption (GFAA) spectrometry.
The following analytical methods have been approved by the U.S. EPA, with detection limits that vary between 0.08 and 7 µg/L:
- EPA Method 200.5 Rev. 4.2 uses axially viewed ICP-AES and has an MDL of 0.2 µg/L (U.S. EPA, 2003b).
- EPA Method 200.7 Rev.4.4 uses ICP-AES and has an MDL of 4 µg/L (U.S. EPA, 1994a).
- EPA Method 200.8 Rev. 5.4 uses ICP-MS and has an MDL of 0.08 µg/L and a minimum reporting level (MRL) of 0.2 µg/L (U.S. EPA, 1994b).
- EPA Method 200.9 Rev. 2.2 uses stabilized temperature GFAA spectrometry and has an MDL of 0.1 µg/L (U.S. EPA, 1994c).
- Standard Method (SM) 3113B uses electrothermal atomic absorption spectrometry and has an MDL of 0.1 µg/L (APHA et al., 1992, 1995, 2005, 2012).
- SM 3120B uses ICP-AES and has an MDL of 7 µg/L (APHA et al., 1992, 1995, 1998, 2005, 2012).
- The online versions of SM 3113B-04, 99 and SM 3120B-99 are also approved methods.
Studies have indicated that total chromium analysis can be complicated using these analytical procedures. Eaton et al. (2001) simultaneously analyzed drinking water samples for both total chromium (using ICP-MS) and Cr(VI) (using ion chromatography). The results indicated that many of the samples had soluble Cr(VI) concentrations that were higher than total chromium concentrations. The authors postulated that a possible difference in the instrument calibration for Cr(III) and Cr(VI) species analysis or a problem with the sample acid preservation may explain this discrepancy (Eaton et al., 2001). However, Zimmer (2014) undertook a study to assess chromium analysis for compliance under the U.S. EPA and California regulations and concluded that the difference between Cr(VI) and total chromium is due to method variability and accuracy.
It is well established that the ICP-MS method is prone to polyatomic interference, which is caused when ions consisting of more than one atom have the same nominal mass-to-charge ratio as the chromium isotopes 52Cr and 53Cr. Carbon in the form of alkalinity or natural organic matter (NOM) generates a “false positive” for the 52Cr isotope by forming the 40Ar12C ion. The presence of chlorine generates a “false positive” for both 52Cr and 53Cr by forming 35Cl16O1H and 37Cl16O ions, respectively (Inoue et al., 1995; Powell et al., 1995; McNeill et al., 2013; Parks et al., 2013).
Total chromium determination may also be complicated by the presence of iron particles in the sample. Soluble chromium species in water may sorb to the iron hydroxide solids (“sorbed chromium”) or become incorporated within the iron hydroxide crystalline structure (“fixed chromium”). In some cases, the filtration and acidification procedures are unable to achieve total chromium recovery, and a hydroxylamine or microwave-assisted acid digestion is required for full recovery of chromium (Eaton et al., 2001; Frey et al., 2004; Parks et al., 2004; APHA et al., 2012; McNeill et al., 2013).
The current U.S. EPA practical quantitation limit (PQL) for total chromium is 10 μg/L (U.S. EPA, 1991). In the second 6-year review of existing National Primary Drinking Water Regulations, the U.S. EPA (2009) concluded that it may not be appropriate to lower the PQL, given the lack of data below the current PQL.
A U.S. EPA (2010a) report indicated that the PQL could be problematic for practical purposes, as different methods have been used for its determination. The report noted that the MRL may be useful as an alternative to the PQL for setting future regulatory limits. The MRL for an analyte is measured using a specific analytical method and is defined as an estimate of the lowest concentration minimum reporting level (LCMRL) that is achievable by the analyst with 95% confidence at least 75% of the time (U.S. EPA, 2012a).
6.2 Hexavalent chromium
The current recommended U.S. EPA method for the analysis of low-level Cr(VI) in drinking water is EPA Method 218.7, which uses ion chromatography with post-column derivatization and ultraviolet (UV)–visible spectroscopy. The method is based on a modified version of EPA Method 218.6 and uses two ion chromatography systems with different eluents (ammonium sulphate/ammonium hydroxide and sodium carbonate/sodium bicarbonate).
EPA Method 218.7 has an MDL ranging from 0.0044 to 0.015 µg/L, and the lowest concentration MRL ranges from 0.012 to 0.036 µg/L, depending on the type of preservative and the eluent system used (U.S. EPA, 2011a). As Cr(III) and Cr(VI) can interconvert, depending on the water quality and presence of various constituents (oxidizing or reducing agents), the proper preservation of the chromium species in the collected samples is critical for accurate analysis. The samples are preserved with a combination of buffer/dechlorinating agent. Preservation is accomplished by raising the pH of the sample above 8.0 with either liquid (ammonium sulphate/ammonium hydroxide) or solid (sodium carbonate/sodium bicarbonate/ammonium sulphate) buffers. Following the preservation step, Cr(VI) is separated from other components in the sample by the ion chromatography column and then derivatized in the post-column reactor. Cr(VI) is then analyzed spectrophotometrically at a wavelength of 530 nm. EPA Method 218.7 has a holding time of 14 days for the preserved sample (U.S. EPA, 2011a).
EPA Method 218.6 Rev. 3.3 also uses ion chromatography determining Cr(VI), but was not specifically developed for drinking water analysis (U.S. EPA, 1994d). This method has an MDL and MRL of 0.3 µg/L and 0.4 µg/L, respectively. It requires that the sample be filtered at the time of collection and the pH of the filtrate be adjusted to 9.0–9.5 using an ammonium sulphate/ammonium hydroxide buffer. At this pH, Cr(VI) exists as a CrO42−anion and is separated from other ionic species present in the water sample on an anion exchange column. Following the derivatization of Cr(VI), the sample is measured spectrophotometrically at 530 nm. The holding time of the preserved samples is 5 days (U.S. EPA, 1994d, 2011b).
Cr(VI) can also be measured by ion chromatography using a modified version of EPA Method 218.6 to achieve lower DLs. The modifications (Dionex, 2003) will achieve an MDL of 0.018 µg/L and a reporting limit of 0.06 µg/L. Further modifications published by Dionex (2011) reported an MDL of 0.001 µg/L and a quantitation limit of 0.003 µg/L (Dionex, 2011; McNeill et al., 2013).
6.2.1 Other methods for determination of Cr(VI)
6.2.1.1 Cr(VI) analysis by HPLC-ICP-MS
Several researchers have conducted speciation analyses for chromium using high-performance liquid chromatography (HPLC) coupled with ICP-MS(Inoue et al., 1995; Powell et al., 1995; Barnowski et al., 1997). As the Cr(III) species are positively charged ions and the Cr(VI) species are negatively charged ions, HPLC can only separate one of the chromium species, depending on the type of ion exchange column used. ICP-MS is used to quantify the concentration of the chromium species before and after the separation step, with the difference providing the concentration of the other chromium species retained on the column. The reported MDLs for Cr(III) ranged from 0.005 to 0.5 µg/L, and for Cr(VI), from 0.009 to 1.0 µg/L, depending on the column types, eluents used, pH and injection volume (McNeill et al., 2013).
6.2.1.2 Field speciation method for Cr(VI)
This method uses a cation exchange column coupled with GFAA spectrometry and has a DL of 0.05 µg/L for Cr(VI). Limitations of the method include a need for larger amounts of cation exchange resin when analyzing samples with a high ionic strength or high Cr(III) concentration of the water sample (Ball and McCleskey, 2003). Previous work suggests that organic ligands may complex Cr(III)and convert it into an anion that can pass through the column, giving a “false positive” result for Cr(VI) (Icopini and Long, 2002). The presence of particulate iron can result in lower recovery of chromium due to sorption or co-precipitation. There is a need to develop a method for total chromium method that overcomes iron interference (Parks et al., 2004; McNeill et al., 2013).
6.3 Sample preservation and preparation
The stability of total chromium and Cr(VI) samples is dependent on the proper preservation and/or dechlorination steps. The third Unregulated Contaminant Monitoring Rule (UCMR3) defined specific analytical requirements for the monitoring of total chromium and Cr(VI).
The methods currently approved by the U.S. EPA only require acid digestion when the turbidity of the acid-preserved sample is greater than 1 NTU. However, the analytical requirements under UCMR3 include solubilizing the acid preserved sample by gentle heating (i.e., hot digestion) using nitric acid, regardless of the sample turbidity or the method used. Sample preservation and dechlorination procedures are required when the sample is collected (U.S. EPA, 2012c).
For Cr(VI) samples, the proper preservation of the chromium species in the collected samples is critical for accurate analysis since Cr(III) and Cr(VI) can interconvert, depending on the water quality and presence of various constituents (oxidizing or reducing agents). As outlined above, EPA Method 218.7 requires that the samples be preserved with a combination of buffer/dechlorinating agent by raising the pH above 8.0 (U.S. EPA, 2011a).
7.0 Treatment technology
7.1 Chromium redox chemistry
The two most common oxidation states for chromium in natural water are Cr(III) and Cr(VI). Other oxidation states, such as Cr(IV) and Cr(V), are known to form as intermediates in redox reactions, but they are unstable and subsequently disproportionate to Cr(III) and Cr(VI) species (Kotas and Stasicka, 2000; Frey et al., 2004; Lai and McNeill, 2006).
The simple ionic form of Cr(III) is Cr3+, which predominates in water below pH 4. Above pH 4, Cr3+ gradually forms the hydroxide complexes Cr(OH)2+, Cr(OH)2+, Cr(OH)30 and Cr(OH)4−, and the ionic charge is changed from +3 to a mix of charges ranging from +2 to −1 in the pH range of 4–10. Cr(III) exhibits very low water solubility (less than 20 µg/L) in the pH range of 7.0–10.0, and minimum solubility (approximately 1 µg/L) is reported around pH 8 (Rai et al., 1987; Frey et al., 2004; McNeill et al., 2012).
At chromium concentrations typically found in drinking water, Cr(VI) occurs as oxyanions: hydrogen chromate (HCrO4−) and chromate (CrO42−). These anions are considered to be highly soluble in water, and their concentrations are pH dependent. In natural water sources, HCrO4− is the dominant anion below pH 6.5, while CrO42− is the dominant anion above pH 6.5 (Sengupta et al., 1986; Brandhuber et al., 2004; Sharma et al., 2008).
A survey by Frey et al. (2004) reported that total chromium occurred equally in surface waters and groundwaters. However, Cr(VI) was not found in surface waters to nearly the same degree as in the groundwater. Because of its relative insolubility under typical groundwater conditions, Cr(III) is not a significant groundwater contaminant. The survey reported that total chromium concentrations in surface waters were composed primarily of Cr(III), whereas total chromium concentrations in groundwater were composed predominantly of Cr(VI) (Frey et al., 2004). These findings were corroborated in the recent study conducted by Seidel and Corwin (2013).
The redox chemistry of chromium is of utmost importance in the treatment and removal of chromium from drinking water. The oxidation reaction of soluble Cr(III) to Cr(VI) is of concern, because even if Cr(VI) is completely reduced to Cr(III) at the treatment plant, it may potentially re-form in the distribution system when oxidants such as chlorine and chloramine are in contact with soluble Cr(III) or plumbing surfaces that contain chromium (Ulmer, 1986; Clifford and Chau, 1988; Brandhuber et al., 2004; Saputro et al., 2011; Lindsay et al., 2012). The rate of oxidation of soluble Cr(III) to Cr(VI) by free chlorine is dependent on water pH and the chlorine doses (Ulmer, 1986; Clifford and Chau, 1988). The oxidation of soluble Cr(III) at a concentration of 100 µg/L was investigated under different water quality conditions (Brandhuber et al., 2004). Experiments conducted for 24 hours with deionized water and chlorine at 1.0 mg/L showed less than 50% oxidation of Cr(III) at both pH 5 and pH 7 and minimal oxidation at pH 9. Experiments conducted for 140 hours with synthetic water showed that the oxidation was > 90% and > 80% at pH 5 and pH 7, respectively. However, in water containing manganese and NOM, no oxidation was observed at both pH 5 and pH 7, suspected to be due to the reaction between chlorine and manganese and/or NOM. Raw water with high total dissolved solids (TDS), high alkalinity, high hardness and low total organic carbon (TOC) that contained Cr(III) at 90 µg/L was also oxidized with chlorine at 1 mg/L at pH 5, 7 and 9. A graphical representation showed that a greater degree of oxidation was observed at pH 7 (at 50 hours); the lowest was observed at pH 9 owing to the precipitation of Cr(III), which was unable to be oxidized by the chlorine (Brandhuber et al., 2004). A study by Lindsay et al. (2012) demonstrated that chlorine oxidized soluble Cr(III) to Cr(VI) on the time scale of hours. The experiments were conducted with both distilled, deionized water (DDW) and tap water (1.7 mg/L TOC) samples spiked with a high concentration of Cr(III) (100 µM; 5,200 µg/L). As the theoretical stoichiometric molar ratio of chlorine to Cr(III) is 1.5:1, full oxidation of Cr(III) to Cr(VI) is expected to occur with chlorine doses higher than 10.6 mg/L. According to a kinetic model, a chlorine dose of 10 mg/L resulted in a maximum formation of 15 µM (780 µg/L) and 31 µM (1612 µg/L) of Cr(VI) in the DDW (pH 6.98) and in the tap water (pH 5.88), respectively. The study indicated that the oxidation reactions slowed and that Cr(VI) concentrations reached a plateau. The consumption of chlorine via reactions with intermediate oxidation states of chromium, such as Cr(IV) and Cr(V), was a possible explanation for the observed plateau (Lindsay et al., 2012). However, Clifford and Chau, (1988) reported no measurable oxidation of Cr (III) in tap water in experiments conducted on water spiked with 200 µg/L Cr(III), chlorine dose of 3 mg/L, TOC concentration of 3.8 mg/L at a pH ranging from 5 to 8. Brandhuber et al. (2004) reported that a water sample containing Cr(VI) at 100 µg/L was partially reduced (70%) to Cr(III) by stannous chloride, yielding Cr(VI) concentrations in the range of 25–30µg/L. However, Cr(VI) concentrations increased to approximately 48–51 µg/L in the 48 hour period following application of chloramine doses ranging from 0.5 to 2.0 mg/L, at pH 7.0. After this 48 hour period, negligible change was observed to the end of the study (168 hours).
Potassium permanganate was also found to be an effective oxidant for soluble Cr(III), and nearly complete oxidation was observed at neutral or low pH conditions within 60 minutes (Brandhuber et al., 2004). Manganese oxides (Fendorf and Zasoski, 1992; Nico and Zasoski, 2000; McNeill et al., 2012) and hydrogen peroxide (Rock et al., 2001) have also been shown to mediate the oxidation of Cr(III). However, dissolved oxygen was reported to be ineffective for oxidizing soluble Cr(III). It was reported that the particulate Cr(III) species, such as those formed from reduction of Cr(VI) by ferrous iron, could not be oxidized by chlorine-based oxidants (Brandhuber et al., 2004).
Reduction of Cr(VI) to Cr(III) is a water treatment strategy used for removing Cr(VI) from drinking water. The reduction of Cr(VI) to Cr(III) by ferrous iron was reported to be highly effective, with reaction kinetics ranging from seconds to hours, depending on the pH of the water and Cr(VI) concentrations; it can occur in groundwater with low dissolved oxygen, in water treatment plants and in distribution systems (Philipot et al., 1984; Fendorf and Li, 1996; Buerge and Hug, 1997, 1999; Schlautman and Han, 2001; Lee and Hering, 2003; McNeill et al., 2013). Iron solids present in the water distribution pipes, such as hematite, magnetite, ilmenite and green rust, may serve as a source of ferrous iron for the reduction of Cr(VI) (Peterson et al., 1997; Kiyak et al., 1999; Loyaux-Lawniczak et al., 2000). Cr(VI) can also be reduced by many sulphur compounds, including thiols, iron sulphide, metabisulphite, sodium sulphide and sodium sulphite (Kim et al., 2001; Lai and McNeill, 2006). Other potential reducing agents include a variety of organic compounds (Brandhuber et al., 2004; Xu et al., 2004). Cr(VI) can be reduced by microbes under both aerobic and anaerobic conditions through direct reduction by chromium-reducing bacteria or indirect reduction via production of hydrogen sulphide or ferrous iron by sulphate-reducing bacteria and iron-reducing bacteria, respectively (Vainshtein et al., 2003).
7.2 Municipal scale
Management strategies for total chromium in municipal drinking water include source water treatment, treatment at the well head or at the water treatment plant, and non-treatment options (i.e., blending). The U.S. EPA (2012b) lists coagulation/filtration, ion exchange, reverse osmosis and lime softening as the best available technologies (BATs) for the control of total chromium in drinking water. More recently, the state of California has identified ion exchange, reduction/coagulation/filtration (RCF), and reverse osmosis as BATs for the removal of Cr(VI) from drinking water (CDPH, 2013).
The treatment strategies or methods generally used for removal of Cr(VI) include 1) direct removal of Cr(VI) and 2) reduction of Cr(VI) to Cr(III), followed by removal of Cr(III). Direct removal of Cr(VI) can be achieved by ion exchange and potentially by adsorptive media. However, reduction methods need to be followed by coagulation/filtration to remove the precipitated Cr(III). Other technologies for treatment of chromium include conventional treatment (Cr(III)), high pressure membrane processes (Cr(III) and Cr(VI)) and reductive media (Cr(III) and Cr(VI)).
Given the presence of oxidants and disinfectants in the water distribution system, Cr(III) is likely to be oxidized to Cr(VI). For this reason, the removal of both chromium species is necessary to achieve the total chromium objectives in treated drinking water.
A broad four-phase program was originally initiated in Glendale, California, in 2002 to develop full-scale treatment processes capable of removing Cr(VI). This program also included the assessment of treatment technology for total chromium removal from drinking water. The program consisted of bench-scale, pilot-scale and demonstration-scale studies. Interim reports were published for each phase (Brandhuber et al., 2004; Qin et al., 2005; McGuire et al., 2006, 2007) and subsequently collated into one final report along with subsequent findings (Blute et al., 2013a), and one peer-reviewed publication (Blute et al., 2014).
A number of bench-scale, pilot-scale and demonstration-scale studies demonstrated that RCF and single-use weak base anion exchange (WBA) processes were very successful in removing Cr(VI)from drinking water (Zotter and Licsko, 1992; Lee and Hering, 2003; Qin et al., 2005; McGuire et al., 2006, 2007; Blute, 2011; Blute et al., 2013a, 2014; Chowdhury et al., 2014; Najm et al., 2014). Pilot-scale strong base anion exchange (SBA) resin was also found to be an effective technology for Cr(VI) removal (McGuire et al., 2006). The selection and effectiveness of each treatment strategy are driven by several factors, including source water chemistry, pre-existing treatment processes and facilities, chromium concentrations, treatment goals, residual handling concerns and costs. Careful selection of the appropriate technology for a specific application is important, as the performance of each treatment technology is impacted by the specific chemical quality of the water being treated. Due to the operational complexity, the RCF process is listed in the California regulation as a BAT only for systems with more than 500 service connections. The treatment performance of the three leading technologies tested at Glendale, California and Coachella Valley Water District is summarized in Table 3.
| Technology | Total chromium | Cr(VI) |
|---|---|---|
| RCF/granular media filtration | < 5 µg/L | < 1 µg/L |
| RCF/microfiltration | < 1 µg/L | < 1 µg/L |
| WBA resin | < 1 µg/L | <1 µg/L |
| SBA resin | 1 µg/L | < 1 µg/L |
|
Note: Cr(VI) and total chromium levels in the treated water from each treatment systems may vary depending on the design and the operation of the treatment system. |
||
Cost curves developed for Glendale, California formed the basis for the cost-benefit analysis in setting the maximum contaminant level (MCL) for Cr(VI) in California (Blute et al., 2013a). An on-line cost estimation tool is available to help utilities estimate a range of potential costs to remove Cr(VI) from their drinking water based on system-specific information, water quality and residual handling. This tool estimates potential cost ranges for three Cr(VI) technologies that have emerged as the leading approaches with respect to feasibility and cost—RCF (reduction with ferrous iron), WBA and SBA—and is available at www.CrVITreatmentCosts.com.
7.2.1 Reduction/coagulation/filtration of Cr(VI)
The removal of Cr(VI) by reduction to Cr(III) with ferrous iron and subsequent coagulation with ferric iron and filtration have long been used in industrial treatment processes and have been demonstrated in Glendale, California, to be an effective technology in drinking water applications (Blute et al., 2013a). Pilot testing at the Metropolitan Water District of Southern California (Najm et al., 2014) and the Coachella Valley Water District (Chowdhury et al., 2014) also confirmed the effectiveness of the RCF process for Cr(VI) removal. The RCF process typically includes an oxidation step upstream of the filters to oxidize the excess ferrous iron to ferric iron, followed by filtration (e.g., dual-media or microfiltration) to remove the formed ferric iron and chromium hydroxide particles. Polymer addition may also be used to enhance the formation of large particles for granular media filtration. Periodically, the filters require backwashing to restore the hydraulic capacity by removing trapped particulate. Waste backwash water processing facilities, including water recovery tanks and solid processing equipment, can be utilized to increase the water efficiency and reduce the volume of residuals for disposal.
A demonstration-scale RCF system with a flow rate of 100 gallons per minute (gpm) (6.3 L/s) achieved Cr(VI) concentrations below 1 µg/L and total chromium concentrations below 5 µg/L in drinking water at Glendale, California. The system consisted of ferrous (Fe(II)) sulphate addition and three reduction tanks in series (each providing 15 minutes of reduction time) to reduce Cr(VI) to Cr(III). Following the reduction tanks, the water passed through an aeration tank to oxidize residual ferrous iron, then a rapid mixing tank where polymer was added for floc formation. Finally, the water was pumped through two parallel dual-media filters (anthracite/sand) with a hydraulic loading rate of 3 gpm/ft2 (7.3 m/h) each, to remove the chromium-containing flocs.
A demonstration study observed better performance when the time for reduction of Cr(VI) to Cr(III) was 45 and 30 minutes compared with 15 minutes. At a Fe(II):Cr(VI) mass ratio of 25:1 constant, both reduction times of 45 and 30 minutes were able to reduce an influent Cr(VI) concentration of approximately 80 µg/L to a range from below the MRL of 0.02 to 0.21 µg/L in the filtered water (filter run length of 48 hours). A decrease of the reduction time to 15 minutes resulted in an increase in the Cr(VI) concentration up to 0.63 µg/L in the filtered water. A similar pattern was observed for the total chromium concentrations. When the reduction times were 45 and 15 minutes, the total chromium concentration in the filtered water (filter run length of 48 hours) ranged from below the MRL of 1 µg/L to 2.9 µg/L and 5.0 µg/L, respectively. A study by Najm et al. (2014) reported that the shorter reduction time may be sufficient when higher ferrous doses are used.
The demonstration study indicated that, depending on the influent chromium concentration and the iron dose, an aeration step may be used to fully oxidize the excess ferrous iron and facilitate the coagulation of ferric iron with Cr(III). It was observed that approximately 21% of the ferrous iron remained in the water after 45 minutes of reduction time, whereas 26% and 60% of the ferrous iron were present after 30 and 15 minutes of reduction, respectively (Blute et al., 2013a).
The impact of raw water pH on Cr(VI) reduction efficiency was studied by several researchers (Blute et al., 2013a; Chowdhury et al., 2014; Najm et al., 2014). Demonstration-scale results showed that when the RCF process was conducted with an Fe(II):Cr(VI) ratio of 34:1 and a raw water pH of 8.2 and 7.5, the influent Cr(VI) concentration of approximately 80 µg/L was reduced to 23 µg/L and < 0.02 µg/L, respectively, in the filtered water(Blute et al., 2013a). Bench-scale jar testing results (Blute et al., 2013b) indicated that Fe(II):Cr(VI) ratios of 25:1 and 50:1 at a raw water pH of 7.87 were insufficient to reduce an influent Cr(VI) concentration of 13 µg/L to below 1 µg/L in the treated water. However, when the pH was reduced to 7.35, the Fe(II):Cr(VI) ratio of 50:1 effectively reduced the influent Cr(VI) concentration to 0.04 µg/L in the filtered water, indicating that a higher Fe(II):Cr(VI) ratio and lower pH levels facilitated Cr(VI) reduction in the RCF process. Similar to the Cr(VI) results, Fe(II):Cr(VI) ratios of 50:1 and 75:1 reduced the total chromium concentration to below 1 µg/L in the same sample. A pH higher than 7.5 may accelerate the reaction rate of ferrous iron oxidation by oxygen and result in less ferrous iron being available for Cr(VI) reduction (Fendorf and Li, 1996; Lee and Hering, 2003). The initial findings at Glendale, California indicated that pH can impact reduction rate; however the follow-up study demonstrated that higher ferrous concentrations can overcome these pH impacts (Blute et al., 2013b). The necessary Fe(II):Cr(VI) ratio and pH levels will depend on the target Cr(VI) and total chromium removal.
Brandhuber et al. (2004) found that the presence of co-occurring contaminants, such as phosphate, sulphate, arsenate and silica, may have a varying impact on the rate of reduction of Cr(VI) by ferrous iron. The authors reported that the presence of sulphate ions had no impact on the rate of reduction of Cr(VI) and that phosphate and arsenate ions slightly decreased the rate. Blute et al. (2013b) found that the presence of silica inhibited the reduction of Cr(VI) when the silicon dioxide concentration was increased from 29 to 76 mg/L, and hypothesized that the impact of silica on Cr(VI) reduction efficiency may be due to less effective coagulation.
As Cr(III) precipitates are associated with the ferric iron particles, total chromium removal depends on the effectiveness of the filtration process. A pilot-scale study (Qin et al., 2005) using an Fe(II):Cr(VI) ratio of 50:1 effectively reduced an influent Cr(VI) concentration of 100 µg/L to below detectable levels (MDL was not cited) and the total chromium concentration to below 1 µg/L (99.1–100% removal) in the treated water. Conditions were optimized with filter loading rates ranging from 3 to 4 gpm/ft2 (7.3–9.8 m/h) and a water pH below 7.5 during the filtration step (Qin et al., 2005). Pilot-scale tests showed that total chromium concentrations at or below 1 µg/L could be achieved with the RCF process (Qin et al., 2005; Blute et al., 2013c). However, demonstration-scale tests conducted with granular media filtration yielded fluctuations in the filter effluent turbidity, resulting in total chromium levels in the filtered water ranging from 1 to 5 µg/L (Blute et al., 2013a). Blute et al. (2013d) investigated the effectiveness of membrane filtration in achieving lower total chromium concentrations through enhanced particle removal. Previous studies have suggested that the performance of direct membrane filtration on contaminant removal was site specific as a result of differences in feed water quality, membrane material and membrane systems used (Blute et al., 2013c).
Two membrane systems, a pressure microfiltration (MF) with flow rates of 20 gpm and submerged ultrafiltration (UF) membranes (11 gpm), were integrated into the existing demonstration-scale RCF facility at a flow rate of 100 gpm (6.3 L/s) (Blute et al., 2013d). A pretreatment applied prior to the membranes included a reduction of Cr(VI) to Cr(III) and an aeration step. A chlorine dose was added to the aeration tank to oxidize ferrous residual and minimize the membrane fouling. Two different Cr(VI) concentrations, approximately 80 µg/L and 15 µg/L, representing high and low Cr(VI) levels in the source water, respectively, were tested. The influent total chromium concentrations similarly ranged from 84 to 89 µg/L and from 2.8 to 16 µg/L for the high and low concentrations, respectively. The raw water (influent water to the RCF process) alkalinity ranged from 210 to 220 mg/L as CaCO3; total hardness ranged from 330 to 360 mg/L as CaCO3; and TOC ranged from < 0.3 to 0.4 mg/L. A ferrous iron dose of 2 mg/L effectively reduced both low and high Cr(VI) concentrations to Cr(III). This study concluded that a minimum threshold of iron (e.g., 2 mg/L as Fe) may be necessary for removal of low and high Cr(VI), rather than a specific Fe(II):Cr(VI) ratio. The Cr(VI) concentrations in both membrane influents ranged from <0.02 to 0.12 µg/L, whereas the total chromium concentrations were similar to the levels observed in the raw water. Both membrane systems achieved Cr(VI) concentrations ranging from 0.02 to 0.26 µg/L and total chromium concentrations were <1 µg/L. Turbidity levels were below 0.04 NTU in the filtered water, and alkalinity, total hardness and TOC levels were similar to those of the raw water. Free chlorine residual in the feed water to the membrane systems ranged from < 0.02 to 0.66 mg/L. It was reported that minor reoxidation of Cr(III) to Cr(VI) (concentrations below 0.3 µg/L) occurred when the chlorine was added upstream of the membrane systems and that at full scale, the chlorine dose would need to be carefully controlled in order to minimize the oxidation of Cr(III) to Cr(VI) (Blute et al., 2013d).
A pilot-scale study (Najm et al., 2014) indicated that an RCF process can be integrated into a conventional water treatment process to remove Cr(VI) or total chromium from surface water. Surface water was spiked with Cr(VI) at 25 µg/L and split between two parallel treatment trains (control and test) at a flow rate of approximately 3 gpm (0.2 L/s) per train. TOC ranged from 2.93 to 3.06 mg/L; total alkalinity and total hardness varied from 107 to 120 mg/L and from 218 to 278 mg/L as CaCO3, respectively. Alum coagulant at 10 mg/L was added to the rapid mix tanks of both trains to provide adequate coagulation of the surface water. Ferrous sulphate at 2 mg/L (Fe(II):Cr(VI) mass ratio of 80:1) was added at the flocculation basin influent of the test train to reduce Cr(VI) to Cr(III). A chlorine dose of 0.5 mg/L was added upstream of the granular media filter to oxidize any residual ferrous iron to ferric iron, while minimizing the reoxidation of Cr(III) to Cr(VI). The control train (rapid mix, flocculation, sedimentation, granular media filtration) showed no removal of Cr(VI) or total chromium, whereas the test train (conventional surface water treatment in combination with the RCF process) achieved a Cr(VI) concentration of < 0.15 µg/L (> 99% removal) and a total chromium concentration of 1.5 µg/L (93% removal) in the filtered water. Replacing the aeration step with a low dose of chlorine helped reduce the footprint of the RCF process (Najm et al., 2014). The turbidity goal of < 0.15 NTU was achieved after a filter ripening period of > 2 hours. An increase of the alum dose from 10 to 15 mg/L and a relocation of the chlorine feed location from the filter influent to the flocculation basin effluent improved the filter performance by reducing the filter ripening period to < 20 minutes; decreased the average headloss accumulation rate from 0.39 to 0.33 feet per hour (0.12 to 0.10 m/hour); and increased the filter run length from 14.6 to 16 hours. Relocating the feed chlorine increased the chlorine contact time and resulted in minor increases of Cr(VI) concentrations: from < 0.03 to 0.28 µg/L in the sedimentation basin influent; from 0.13 to 0.35 µg/L in the filter influent; and from 0.15 to 0.38 µg/L in the filter effluent. Nonetheless, all Cr(VI) concentrations in the filtered water were below 1 µg/L. The integration of an RCF process into a conventional treatment train did not adversely affect ferrous iron or total iron limits. No measurable increase in the formation of total trihalomethanes or halogenated acetic acids was observed (Najm et al., 2014).
The RCF techniques result in chromium-rich sludge, which must be disposed of appropriately. The RCF process results in backwash water that contains chromium-rich solids. Treatment and disposal of this waste will depend on a jurisdiction’s regulations. Some municipalities may allow sewer discharge of the unsettled backwash water if sewage treatment plants can accept the waste. Otherwise, solids can be settled, and liquid backwash water can either be returned to the head of the treatment plant (minimizing waste) or disposed of as non-hazardous waste. However, it is recommended that filter backwash water not be recirculated through the treatment plant without additional treatment. Testing at Glendale, California found that settled solids were found to be hazardous based on California’s classification and non-hazardous according to U.S. EPA classification.
7.2.2 Anion exchange
Ion exchange is a physicochemical process in which there is an exchange of ions in the raw water with ions within the solid phase of a resin. As raw water ions displace ions on the resin, the capacity of the resin is gradually exhausted, resulting in finished water concentrations that increase (i.e., contaminant breakthrough). Once the resin has reached its capacity (i.e., when all the available resin sites have been occupied by the contaminant ion), the resin must be regenerated to reverse the process. Exchange resins exhibit a degree of selectivity for various ions, depending on their type and their concentration in solution, and the type of resin selected.
Two types of anion exchange resins have been shown to be effective for Cr(VI) removal from drinking water: WBA and SBA resins.
7.2.2.1 Weak base anion exchange resin
WBA resin represents a new application in drinking water treatment. Bench-scale, pilot-scale and demonstration-scale studies have shown that some WBA resins exhibited a high capacity for Cr(VI). For optimum Cr(VI) removal with WBA resin, the pH of the water must be reduced to pH 6. In the acidic pH range (using HCl), the resin is converted to its hydrochloric form and the functional groups of the resins are protonated, and act as positively charged exchange sites where the chloride ions (Cl–) are replaced with the Cr(VI) (hydrogen chromate) anions. Decreasing the pH also reduces the competition between hydroxyl ion and Cr(VI) for the exchange sites on the WBA resin (Blute et al., 2007). The traditional ion exchange mechanism is not solely responsible for the high Cr(VI) removal. Although the true mechanism of Cr(VI) removal by WBA resins is not fully understood, it has been observed that a reduction process is involved in which the adsorbed Cr(VI) is converted to Cr(III) (McGuire et al., 2006; Blute, 2011). Following the WBA treatment, the treated water pH needs to be adjusted to reduce the potential for distribution system issues (e.g., corrosion). The WBA resin is operated as a single-pass resin, eliminating the need for resin regeneration with brine. The WBA resins exhibit very high Cr(VI) removal capacities which result in the cost-effective use of the resins as disposable media (McGuire et al., 2007; Blute et al., 2013a).
A 425 gpm (26.8 L/s) demonstration-scale system (Blute et al., 2013a) treated an influent Cr(VI) concentration of 40 µg/L in groundwater to achieve a target concentration of 5 µg/L in the lag bed effluent. The system included two bag filters for raw water particle removal and two ion exchange vessels in a lead/lag configuration. The influent water was pH-adjusted from the initial pH of approximately 6.8 to 6.0 by the addition of carbon dioxide. The lead vessel was replaced at 172 000 bed volumes (BVs) (after 1 year of operation) when the lag vessel reached the target Cr(VI) concentration of 5 µg/L (86 000 BVs). At that point, the lead vessel effluent reached a Cr(VI) concentration of 15–20 µg/L. After the lead vessel resin was replaced, the lag vessel was put in the lead position. The total BVs of water treated by the initial lag vessel was 364 000 (approximately 940 days), with the effluent reaching a Cr(VI) concentration of 14 µg/L.
The removal of Cr(VI) by WBA resins is strongly dependent on water pH (McGuire et al., 2006; Blute et al., 2013a; Najm et al., 2014). A pilot-scale WBA unit in lead/lag configuration was operated with an empty bed contact time (EBCT) ranging from 2 to 3 minutes per column under different pH conditions (McGuire et al., 2006). The ion exchange unit treated a Cr(VI) concentration of 100 µg/L in groundwater samples to achieve a target concentration of 5 µg/L in the lead column effluent. The average Cr(III) and Cr(VI) concentrations achieved in the lead column effluent were 8.6 µg/L and 4.9 µg/L, respectively, when pH was below 5.5.When the pH was increased to 6.0, the Cr(III) and Cr(VI) concentrations were decreased to 1.7 µg/L and 4.1 µg/L, respectively. The authors reported that the Cr(VI) concentration in the lead column effluent was significantly higher (14.8 µg/L) when the pH was above 6.0. As the dominant chromium species in the influent was Cr(VI) and as Cr(III) was present in the treated water, it was suggested that a reduction of Cr(VI)to Cr(III) occurred on the resin surface or in the resin matrix. Cr(III) concentrations exceeded 5 µg/L in the effluent when the ion exchange unit operated below pH 5.5 (McGuire et al., 2006). Another pilot-scale WBA column was capable of reducing an influent total chromium concentration of approximately 35 µg/L in groundwater to a target concentration of 5 µg/L for 45 000 BVs, approaching 15 µg/L at 113 000 BVs. The column operated at a hydraulic loading rate of 4 gpm/ft2 (9.8 m/h), an EBCT of 2 minutes and pH 6.0. The same resin was used to treat groundwater at pH 6.8 and achieved a total chromium concentration of 5 µg/L in the treated water after only 2300 BVs, gradually increasing to 25 µg/L after 80 000 BVs (McGuire et al., 2007). A bench-scale study (Najm et al., 2014) evaluated the performance of two types of WBA resins at pH 5.5.The tests were conducted on source water with an influent Cr(VI) concentration of 17 µg/L. Both WBA resins achieved complete removal of Cr(VI) (MRL = 0.02 µg/L). However, Cr(III) concentrations ranged from 1 to 4 µg/L in all samples(based on graphical representation) for 100 000 BVs. An increase in Cr(III) solubility at a lower pH and the formation of positively charged hydroxide complexes suggested that operating at pH 6.0 was optimal to maximize the exchange capacity of WBA resins without increasing the solubility of Cr(III) (Rai et al., 1987; Najm et al., 2014).
Bench-scale tests (Najm et al., 2014) evaluated the applicability of two types of WBA resins for Cr(VI) removal under a wide range of water quality conditions. The overall impacts of water quality on resin performance as well as the impacts of influent Cr(VI) and sulphate concentrations were also evaluated. The tests were conducted under 16 different conditions, using 10 groundwater sources. The setup for each condition included two continuous-flow packed columns in series. Each column operated at pH 6.0 and an EBCT of 1.5 minutes. During the 24-week period of testing, approximately 150 000 and 75 000 BVs were treated through the columns, with EBCTs of 1.5 and 3.0 minutes, respectively. The water quality parameters of the raw groundwater sources included total chromium at concentrations ranging from 1.9 to 48.0 µg/L (present as Cr(VI)); uranium at concentrations ranging from < 1.0 to 11 µg/L; pH ranging from 7.8 to 8.9; total alkalinity ranging from 88.0 to 426.0 mg/L as CaCO3; and sulphate and nitrate at concentrations ranging from 6.0 to 59.0 mg/L and from 0.3 to 46.0 mg/L, respectively. The study objective was to achieve Cr(VI) concentrations ranging from 1 to 2 µg/L (breakthrough) in the treated water. The study reported an MRL of 0.02 µg/L for Cr(VI) and an MRL of 1.0 µg/L for total chromium (Najm et al., 2014). According to the authors, both tested WBA resins demonstrated high capacity for Cr(VI) removal, as no breakthrough was experienced up to 150 000 BVs for several different source waters (Najm et al., 2014).
Both resins performed similarly when tested in parallel under the same operating conditions and were capable of reducing an average influent Cr(VI) concentration of 10.0 µg/L to below 0.02 µg/L in the presence of nitrate at 2.5 mg/L and sulphate at 19.0 mg/L, achieving 150 000 BVs with an EBCT of 1.5 minutes. Paired samples were collected, and the total chromium concentration was measured. Both resins reduced the total chromium concentration to the MRL of 1 µg/L for 150 000 BVs, with an EBCT of 1.5 minutes. According to the authors, when the source water was spiked with Cr(VI) at 35 and 60 µg/L, both resins showed good removal of Cr(VI) for both concentrations (Najm et al., 2014).
Sulphate ions significantly influence the removal of trace constituents by SBA resins. However, the bench-scale study (Najm et al., 2014) demonstrated that the sulphate ions had a minor effect on Cr(VI) and total chromium removal by WBA resins for up to 150 000 BVs. In the presence of sulphate at 167 mg/L, an average influent Cr(VI) concentration of 10.0 µg/L was reduced to 0.02 µg/L for 100 000 BVs and an EBCT of 1.5 minutes. The breakthrough curve demonstrated that Cr(VI) concentrations increased slowly and gradually after 100 000 BVs and reached approximately 1 µg/L in the treated water after 150 000 BVs, while total chromium concentrations ranged from 1 to2 µg/L. The impact of nitrate ions on the WBA resin performance was tested on source water with ambient concentrations of 40 µg/L for Cr(VI), 40 mg/L for nitrate and 35–40 mg/L for sulphate. The graphical representation of the breakthrough profile showed an early breakthrough. Both Cr(VI) and total chromium concentrations increased at approximately 10 000 BVs in the finished water and gradually reached 10 µg/L at 100 000 BVs, with an EBCT of 1.5 minutes (Najm et al., 2014).
Both resins achieved complete removal of uranium under all test conditions. Minimal removal was achieved for nitrate, perchlorate and arsenic ions. Uranium is of significance, as it can accumulate on the WBA resin and pose a significant disposal challenge. Two water sources with influent nitrate concentrations of approximately 45 mg/L experienced chromatographic peaking of nitrate at approximately 150 BVs of operation (Najm et al., 2014). Chromatographic peaking of competitive anions may occur when the less preferred anions, such as nitrate, collect on the resin and are then displaced by more preferred anions.
WBA resins exhibit the potential to release nitrosamine species, N-nitrosodimethylamine and N-nitrosopiperidine, during start-up, and appropriate pre-treatment or rinsing of the resin may be necessary to remove them. Organic resin by-products, such as formaldehyde and acetaldehyde, have also been shown to leach from WBA resins and may require pre-treatment prior to resin installation and/or rinsing after installation (McGuire et al., 2007; Blute, 2011; Najm et al., 2014).
In addition to removing Cr(VI), WBA resins also remove other inorganic contaminants, such as uranium. If uranium is present in the raw water, and depending on the level of treatment needed for Cr(VI) removal, the residual from the spent WBA resin may be regulated as a radioactive or hazardous waste and require further processing or hazardous waste disposal, depending on the jurisdiction’s regulations. Testing at Glendale, CA found that spent WBA resin was classified as hazardous, low level radioactive waste (LLRW) or technologically enhanced naturally occurring radioactive material (TENORM) waste in California and non-hazardous, TENORM or LLRW according to U.S. EPA classification (Blute et al, 2013b). The presence of radioactive contaminants may result in operation of the WBA resin that is driven or limited by the solid-phase radioactive contaminant concentration (McGuire et al., 2007; Blute, 2011).
7.2.2.2 Strong base anion exchange resin
SBA exchange technology is commonly used in groundwater treatment for the removal of inorganic anions, such as nitrate and arsenic. SBA resins remove Cr(VI) by exchanging chloride ions on the functional groups of the resin beads. SBA resins are regenerated with a salt (i.e., brine) solution to restore their exchange capacity. A benefit of using SBA resin is the ability to operate without pH adjustment if the calcium carbonate precipitation potential is not significant and bed plugging is not anticipated (Blute et al., 2012). The greatest challenge facing the use of SBA resin for Cr(VI) removal is the handling and disposal of the waste brine.
Other operational considerations when using SBA treatment include chromatographic peaking of the treated water. An additional consideration when using SBA resins is the potential for the release of nitrosamines from the resin. Research has found that new resin or resin that is exposed to disinfectants (chlorine and chloramines) may release nitrosamines due to shedding of manufacturing impurities (Najm and Trussell, 2001;Kemper et al., 2009).
A field scale SBA resin was evaluated to remove total chromium from two water sources. Influent concentrations of 20 µg/L and 18 µg/L of total chromium were reduced to the target concentration of 8 µg/L in the treated water with an EBCT of 45 seconds. Co-occurring contaminants appeared to have substantially impacted the resin performance. For the source water containing nitrate concentration of 6 mg/L and sulphate concentration of 20 mg/L (influent 18 µg/L), the target treatment goal was achieved for 20,000 BVs, while for the source water containing nitrate concentration of 27 mg/L and sulphate concentration of 30 mg/L (influent 20 µg/L), the treatment goal was achieved for 5,500 BV. Site-specific testing of SBA treatment is suggested (Seidel et al., 2014).
A full scale SBA resin was used to remove both arsenic and Cr(VI) from Coachella Valley Water District groundwater using three different treatment facilities with capacities ranging from 1,000 to 4,000 gpm. Resin vessels were run in parallel operation with several of the vessels being regenerated at a time, as driven by arsenic breakthrough which occurs earlier than that of Cr(VI). These facilities show that Cr(VI) can be reliably removed from approximately 10 µg/L to <1 µg/L using SBA resin (Water Research Foundation, 2013).
Two pilot-scale ion exchange columns in lead/lag configuration demonstrated that an SBA type II chloride-based resin was capable of decreasing a spiked influent Cr(VI) concentration of 100 µg/L in groundwater to below 5 µg/L (McGuire et al., 2006). The breakthrough curves of Cr(VI) showed that the first column exceeded 5 µg/L after 1900 BVs, whereas the breakthrough in the lag column occurred at 3800 BVs. Each column operated with an EBCT of 3–4 minutes, and no changes in the oxidation state of chromium were detected throughout the pilot unit. The study also reported the removal of bicarbonate, nitrate, phosphate and sulphate ions by the SBA resin. Chromatographic peaking of nitrate occurred at 410 BVs with a nitrate-nitrogen concentration of 15 mg/L and at 450 BVs with a phosphate concentration of 0.8 mg/L. The raw water concentrations of nitrate and phosphate were 5 mg/L (as nitrate-nitrogen) and 0.2 mg/L, respectively, demonstrating that the chromatographic peaking increased the concentrations of nitrate and phosphate in the treated water (McGuire et al., 2006).
Although chromate anion is strongly preferred by most anion exchange resins, Clifford (1999) reported that the number of BVs that can be treated before chromium breakthrough occurs depends on the resin matrix.
A pilot-scale column with microporous polystyrene resin reduced a Cr(VI) concentration of 42 µg/L to 10 µg/L and achieved 32 000 BVs, whereas ion exchange columns with isoporous polystyrene and macroporous polyacrylic resins achieved 20 700 BVs and 14 600 BVs, respectively. Generally, the resin with the highest affinity for the contaminant and the longest run length is the hardest to regenerate (Clifford, 1999).
Bench-scale tests (Najm et al., 2014) evaluated the applicability of two types of SBA resins for Cr(VI) removal from 10 groundwater sources. The setup for each condition included a single column with a flow rate of 5.9 mL/min, an EBCT of 3.0 minutes and a run length of 12 000 BVs. The water quality parameters of the tested groundwater sources included total chromium at concentrations ranging from1.9 to 48.0 µg/L, as virtually all chromium was present as Cr(VI); uranium at concentrations ranging from < 1.0 to 11 µg/L; pH ranging from 7.8 to 8.9; total alkalinity ranging from 88.0 to 426.0 mg/L as CaCO3; and sulphate and nitrate at concentrations ranging from 6.0 to 59.0 mg/L and from 0.3 to 46.0 mg/L, respectively. The bench-scale study was targeted to achieve Cr(VI) concentrations ranging from 1 to2 µg/L (breakthrough) in the treated water. The MRLs were 0.02 µg/L and 1.0 µg/L for Cr(VI) and total chromium, respectively (Najm et al., 2014). The results of this bench-scale study are discussed in the following four paragraphs.
Both SBA resins showed no differences in chromium removal when they were tested in parallel under the same operating conditions. Each resin was tested with two different source waters with influent concentrations of total chromium ranging from 8.8 to16 µg/L; sulphate concentrations ranging from 19 to 22 mg/L; and nitrate concentrations ranging from 2.2 to 2.8 mg/L. Each resin effectively reduced the total chromium concentration to the MRL of 1.0 µg/L for 5000 BVs and an EBCT of 3.0 minutes (Najm et al., 2014).
Traditional SBA resins are impacted by sulphate and nitrate ions. To evaluate the impact of these ions on SBA resin performance for chromium removal, tests were conducted under ambient and spiked conditions. SBA resin was capable of decreasing an influent total chromium concentration of 10 µg/L to 1 µg/L for greater than 12 000 BVs and an EBCT of 3 minutes in the presence of sulphate at 16 mg/L. Increasing the sulphate concentration to approximately 165 mg/L reduced the column run length to approximately 2000 BVs. When studying the impact of nitrate anions on source water with an initial total chromium concentration of 16.0 µg/L and spiked nitrate concentration of 45 mg/L, no breakthrough (1µg/L) was observed for 5000 BVs and an EBCT of 3 minutes (Najm et al., 2014).
Several source waters were tested under ambient conditions, and the results varied. There was no breakthrough for the entire 12 000 BVs for a source water with a total chromium concentration of 7.3 µg/L and with low nitrate and sulphate concentrations (2.7 mg/L and 5.5 mg/L, respectively).However, breakthrough occurred as early as 4000 BVs when the source water had a total chromium concentration of 11 µg/L and high nitrate and sulphate concentrations (47 mg/L and 60 mg/L, respectively). Source water with a total chromium concentration of 3.2 µg/L, a nitrate concentration of 30 mg/L and a sulphate concentration of 40 mg/L experienced a breakthrough at approximately 9,000 BVs; whereas source water with a total chromium concentration of 53 µg/L, a nitrate concentration of 2.4 mg/L and a sulphate concentration of 16 mg/L also ran for 9,000 BVs before chromium breakthrough. The authors concluded that the breakthrough time for chromium was more affected by the competing nitrate and sulphate anions than by the chromium levels in the source water (Najm et al., 2014).
SBA resins removed other inorganic anions, such as nitrate, arsenic, uranium and perchlorate, when present in the water. Other than uranium, the breakthrough of these anions was observed before that of chromium. The study reported complete removal of uranium for all tested waters for up to 12,000 BVs (Najm et al., 2014).
A pilot-scale magnetic ion exchange system (MIEX ®) was tested for Cr(VI) removal from drinking water. The system consisted of a continuously stirred mixing tank for anion exchange. The magnetic particles, embedded in the SBA resin structure, allowed rapid agglomeration and quick settling of the resin to the bottom of the reactor, which was removed, regenerated and then continuously returned to the reactor. The pilot-scale unit achieved removal efficiency ranging from 92% to 97% of the influent Cr(VI) concentration of 100 µg/L. Optimal Cr(VI) removal occurred with an SBA resin dose between 50 and 60 mL per litre of water and a 30-minute contact time. The tests were interfered with by high levels of sulphate (90 mg/L) (McGuire et al., 2006).
Since brine disposal is one of the most challenging aspects of the SBA technology, studies have evaluated the opportunities for minimizing brine volume by reusing the brine multiple times before disposal. Early pilot-scale work in Glendale, California showed that untreated brine could be recycled several times prior to replacement with a slight decrease in Cr(VI) breakthrough curve length (McGuire et al., 2006). Later testing by Gorman et al. (2014) also showed that recycling is feasible, and that brine minimization techniques such as brine reuse could be effective. A pilot scale study, using three SBA exchange columns operated in parallel, reported that direct reuse of the brine was effective. Regenerant brine was used 8 times consecutively. Each reuse cycle achieved runs of at least 15,000 BVs prior to reaching the treatment goal of 8 µg/L. Seidel et al. (2014) indicated that additional confirmation is needed at full-scale. Regenerant brine management options include discharge to a sewer or septic system, off-site approved land application, ocean discharge through a coastal pipeline, deep well injection, and advanced treatment (Seidel et al., 2014). In the absence of the ability to dispose of the brine to municipal sewer or a brine line, the regenerant brine used in the SBA process typically needs to be disposed of off-site and can potentially be classified as hazardous waste due to the enhanced levels of Cr(VI) and other co-occurring contaminants. For example, the Coachella Valley Water District used SBA to remove arsenic and Cr(VI), and the brine requires treatment with a reduction/coagulation process to remove hazardous arsenic and chromium constituents in the form of solid waste and remaining brine as non-hazardous liquid waste (Water Research Foundation, 2013). A careful evaluation of the viability and long-term reliability of the waste brine disposal options is needed when considering SBA technology for chromium removal (Najm et al., 2014).
7.2.3 Membrane filtration
Reverse osmosis (RO) and nanofiltration (NF) membranes reject water constituents on the basis of their molecular size and charge characteristics. These processes are based on forcing water across a membrane under pressure while the ionic species, such as chromium, are retained in the waste stream. RO systems typically require prefiltration for particle removal to protect the membranes and often include other pre-treatment steps, such as the addition of anti-scaling agents, dechlorination and softening. Post-treatment typically includes pH adjustment, addition of corrosion inhibitors and disinfection (Cevaal et al., 1995). Concentrate disposal must also be considered in the design and operation of RO systems.
Several authors reported that RO and potentially NF effectively removed chromium from drinking water (Hafiane et al., 2000; Taleb-Ahmed et al., 2002; Brandhuber et al., 2004; Muthukrishnan and Guha, 2008; Rad et al., 2009;Yoon et al., 2009; Barikbin et al., 2011). RO was listed as one of the BATs for chromium removal (U.S. EPA, 2003c) and has demonstrated excellent rejection of Cr(VI) at benchscale (Brandhuber et al., 2004). However, it was not selected for further evaluation at pilot or demonstration scale at Glendale, California, as it results in the loss of large volumes of water.
In a pilot scale RO study, a polyamide thin film composite membrane was capable of reducing a feed Cr(VI) concentration of 5.0 mg/L to 0.01 mg/L (10 µg/L), achieving greater than 99% rejection, at a permeate flux rate of 58.8 L/m2.h and a system recovery greater of 42.5% (Rad et al., 2009).
Pilot-scale tests evaluated the effectiveness of NF membranes for simultaneous removal of Cr(VI) and sulphate (SO42−) anions from brackish groundwater (Barikbin et al., 2011). The study reported that a polyamide spiral-wound membrane rejected more than 94% of Cr(VI) from 100 µg/L in the feed water to a concentration of approximately 5 µg/L at pH 8.2 and an operating pressure of 4 bars (400 kPa). The pilot-scale testing showed that NF is efficient and applicable for the removal of Cr(VI) from drinking water. The results indicated that factors such as TDS, transmembrane pressure and pH have an impact on Cr(VI) removal, whereas the concentration of Cr(VI) was less important. The pilot-scale tests also demonstrated that NF simultaneously removed Cr(VI) and sulphate from water, but that an increase in sulphate concentration resulted in a corresponding decrease in Cr(VI) rejection (Barikbin et al., 2011).
7.2.4 Conventional treatment and lime softening
Conventional treatment (coagulation/sedimentation/filtration/disinfection) can remove Cr(III) from water, but not Cr(VI). Cr(III) is removed as a result of precipitation as Cr(OH)3 and co-precipitation with Al(OH)3 and Fe(OH)3. Several authors have reported removal efficiencies ranging from 50% to 98% for Cr(III) by conventional coagulation/flocculation techniques followed by filtration (Philipot et al., 1984; Fatoki and Ogunfowokan, 2002; Lee and Hering, 2003; Brandhuber et al., 2004; Qin et al., 2005).
Jar tests investigated the coagulant efficiency of aluminum sulphate and ferric sulphate for removal of metals, including Cr(III), from surface water. An influent Cr(III) concentration of 0.48 mg/L was reduced to 0.07 mg/L (85.4% removal) with an aluminum sulphate dose of 10 mg/L at pH 7.7 and to 0.05 mg/L (89.6% removal) with a ferric sulphate dose of 13 mg/L at pH 7.4 (Fatoki and Ogunfowokan, 2002).
Lime softening is widely used to treat hard water and is effective at removing inorganic contaminants, such as arsenic, barium, chromium, strontium and vanadium, from drinking water sources. Studies on chromium removal by lime softening have indicated that the removal is species dependent (Sorg, 1979; Philipot et al., 1984). Lime softening during pilot plant testing resulted in greater than 97% removal of an average raw water Cr(III) concentration of 0.15 mg/L at pH 11.3, whereas removal of Cr(VI) at a concentration of 0.13 mg/L was not effective (5% at pH 9.5) (Sorg, 1979). Lime softening is an expensive process and is not recommended unless there is also a need to reduce hardness in the raw water feed (U.S. EPA, 2002).
7.2.5 Emerging technologies
New drinking water treatment technologies for chromium are being studied but are still primarily in the experimental stage and/or have no published information on the effectiveness of full-scale application.
7.2.5.1 Adsorptive/reductive media
Two adsorptive media, zeolite media and granular activated carbon (GAC), were tested at pilot scale (Blute et al., 2013a). Both media were capable of decreasing the concentration of Cr(VI) from 100 µg/L to concentrations <5 µg/L and experienced breakthrough in the range of 600-620 BVs.
Iron-based reductive media have been shown to remove Cr(VI) from drinking water in pilot-scale and bench-scale applications. There are multiple manufacturers of reductive media targeted for chromium removal. These media are proprietary, and the treatment processes are patented. Although the exact removal mechanisms are uncertain, they are thought to involve a combination of reduction, adsorption and precipitation/filtration of Cr(III).A typical reduction media system for Cr(VI) would include prefiltration and pressure vessels loaded with the media. Other process equipment may include post-filtration, pH adjustment and residual processing equipment.
A bench-scale study (Brandhuber et al., 2004) reported that sulphur modified iron (SMI) was effective for Cr(VI) removal from drinking water, however, leaching of iron and increased headloss were observed. Pilot testing using porous iron composite media demonstrated removal of Cr(VI) from 80 µg/L to an average concentration of 0.5 µg/L (Hu and Luk, 2011).The media also simultaneously removed arsenic and phosphate along with Cr(VI).In Ripon, California, demonstration-scale testing of reductive media (iron-based media) for nitrate removal showed the ability to reduce the total chromium concentration to below the DL of 1 µg/L from a concentration of 4 µg/L in the raw water (DSWA and City of Ripon, 2010).
Pilot-scale testing of SMI-III® and Cleanit® technologies was conducted to evaluate these adsorptive media for Cr(VI) removal from drinking water andto determine their effectiveness and any operational issues associated with the technologies. Due to a number of operational issues, the pilot testing for SMI-III® was abandoned. Cleanit® pilot testing showed effective removal of Cr(VI) and total chromium. However, relatively fast breakthrough occurred which would necessitate frequent media replacement or regeneration. There was also significant iron leaching from the media, necessitating iron removal downstream. Overall, the adsorptive media requires a larger footprint than SBA and brine disposal is difficult (Blute et al., 2015).
7.2.5.2 Biological treatment
Biological treatment is an emerging water treatment technology that may be particularly attractive for treating water supplies requiring treatment for the removal of multiple inorganic and organic contaminants, such as nitrate, perchlorate, Cr(VI) and phenol (Nkhalambayausi-Chirwa and Wang, 2001; Drago et al., 2013). The biological treatment process is based on the removal of Cr(VI) from source water through the biological reduction of Cr(VI) to Cr(III) using non-pathogenic microbes under either aerobic or anaerobic conditions. The treatment systems can be designed as fixed bed reactors, fluidized bed reactors (FBRs), membrane bioreactors or membrane biofilm reactors. In general, biological treatment systems require post-treatment to remove biomass and biodegradable organic materials that are present in the reactor effluent. Typical post-treatment includes aeration, filtration, activated carbon and disinfection. Several studies reported on the reduction of Cr(VI) under aerobic and anaerobic conditions by microbes (Wang et al., 2000; Nkhalambayausi-Chirwa and Wang, 2001; Vainshtein et al., 2003; Drago et al., 2013; Najm et al., 2013). It was reported that biological treatment can reduce Cr(VI) concentrations as high as 45 µg/L in the source water to concentrations below 5 µg/L in the filtered water (Drago et al., 2013; Najm et al., 2013).
Drago et al. (2013) reported data from a pilot-scale FBR operated under anoxic conditions and using an electron donor (acetic acid) and nutrients (phosphoric acid) to achieve a biological reduction of Cr(VI) to Cr(III). The bacteria attached to granular activated carbon consistently reduced Cr(VI) concentrations ranging from 40 to 45 µg/L to less than 5 µg/L in the FBR effluent. The study showed that altering the concentration of the electron donor affected the percent reduction of Cr(VI). The Cr(VI) concentrations measured the FBR effluent ranged from 3.7 to 4.1 µg/L and from < 0.2 to1.3 µg/L when the added acetic acid concentrations were 13.1 mg/L and 16 µg/L, respectively, with a hydraulic retention time (HRT) of 40 minutes. Increasing the electron donor concentration up to 17.5 mg/L decreased the Cr(VI) concentration in the FBR effluent to 0.27 µg/L at an HRT of 20 minutes. The study also conducted bench scale experiments with two different size filter papers to simulate granular media filtration and membrane filtration following the FBR process. The results indicated little removal of total chromium and the authors suggested that the residual chromium (following biological process) was likely in a dissolved form and require coagulation to obtain removal. Jar tests demonstrated that a ferric chloride coagulant dose of 4 mg/L reduced the total chromium concentration to below 5 µg/L in the filtered sample. A chlorine dose of 1.5 mg/L applied to the filtered water increased the Cr(VI) concentration from 0.5 to 1.8 µg/L after 3 days of retention (Drago et al., 2013).
7.2.5.3 Electrochemical processes
An electrochemical process relies on the redox reactions taking place at the surface of conductive electrodes immersed into water (Mukhopadhyay et al., 2007; Vasudevan et al., 2010). An electrochemical process has been studied for the removal of Cr(VI) from groundwater (Mukhopadhyay et al., 2007). During the process, ferrous iron released from the anode caused a reduction of Cr(VI) species to Cr(III). The dissolved and oxidized iron reacts with the hydrogen ions produced at the cathode to form the insoluble ferric hydroxide necessary for the adsorption and precipitation of Cr(III) species. Laboratory experiments reported removal of Cr(VI) at a concentration of 3.3 mg/L in groundwater to levels below the DL (0.01 mg/L) in the filtered water with an electrochemical ferrous iron dose of 25 mg/L (Mukhopadhyay et al., 2007).
7.3 Cr(VI) levels in the distribution system
In addition to the chromium present in source water, the water treatment process and the distribution system can contribute additional chromium (Cr(III) and Cr(VI)) to the finished water. Chromium accumulation in distribution system piping can occur and is influenced by a variety of factors, including source water quality, treatment techniques, pipe material, co-occurrence of manganese in the pipe deposits, pH and redox conditions in the distribution system. Changes in treatment type, chemical use and source water characteristics can all lead to the release of chromium (both Cr(III) and Cr(VI); Shock et al., 2008; Friedman et al., 2010).
In a Chicago, Illinois, survey in the 1960s, it was found that 17% of samples had “pickup” of total chromium after leaving the water treatment plant (Craun and McCabe, 1975). In the Water Research Foundation Cr(VI) occurrence study (Frey et al., 2004), it was observed that there was little difference between Cr(VI) levels in samples of the raw water, at the entry point and in the distribution system for most systems. However, results varied in two groundwater systems sampled in this study. In one groundwater system, the authors stated that Cr(VI) concentrations increased from 1.3 µg/L in the raw water to 11.9 µg/L in the distribution system; in the second system, Cr(VI) concentrations decreased from 22.2 µg/L in the raw water to 0.4 µg/L in the distribution system. The study indicated that the sampling locations chosen in the distribution system may have been subject to blending from other source waters. The authors noted that additional monitoring of chromium in the distribution system was needed.
Chromium can also be a trace contaminant in chemicals commonly used for drinking water treatment. Chromium was detected in alum coagulants and had the potential to add 0.24 µg/L of total chromium to the water at commonly applied alum doses (Eyring et al., 2002). Cr(VI) concentrations increased from 0.1 to 0.6 µg/L in a Missouri water treatment plant due to the contribution from alum or lime. The Louisville Water Company discovered that up to 0.4 µg/L of Cr(VI) was being added to its treated water through lime softening treatment at the plant (Song, 2012). This Cr(VI) was carried through the distribution system, and similar concentrations were measured at the distribution system sampling locations.
Total chromium concentrations may also increase in drinking water through leaching from the distribution system materials or premise plumbing. Typical materials used in water system infrastructure include cast iron and stainless steel, both of which contain chromium. Impurities in galvanized iron pipes may be another potential source of chromium. Chromium can be released into the water through leaching or corrosion from these materials (Friedman et al., 2010; McNeill et al., 2013).
Chromium released in the distribution system may be in either the trivalent or hexavalent form. However, given the presence of oxidants and disinfectants such as chlorine or chloramines in the water, Cr(III) is likely to be oxidized to Cr(VI). The reaction kinetics of Cr(III) to Cr(VI) oxidation can vary from minutes to days, depending on the water characteristics (Lai and McNeill, 2006). Chromium accumulation in deposits can occur as a result of surface adsorption and/or co-precipitation reactions involving the soluble Cr(VI). Alternatively, unlined iron pipes may release ferrous iron to water due to corrosion, which can subsequently reduce Cr(VI) to Cr(III) ,be precipitated and accumulate in the distribution system. Friedman et al., (2010) indicated that chromium accumulation in distribution system appears to be influenced by chromium concentration in the water, presence of galvanized pipes, co-occurrence of manganese in the deposits and low pH conditions (below 7.6).
Similar to other inorganics, chromium can be released from distribution system pipes to the drinking water through a desorption process, physical and hydraulic disturbances (Schock et al., 2008; Friedman et al., 2010; Lytle et al., 2010). Friedman et al. (2010) found a higher chromium concentration (of more than an order-of-magnitude) in hydrant flush solids than in the pipe specimen scale. Since hydrant flush solids are generally superficial and more readily mobilized than pipe scale, this may present exposure implications following any hydraulic disturbances (e.g., hydrant flushing, main breaks).
The overall sinks and sources of Cr(VI) and total Cr in the distribution system are not well characterized. Brandhuber et al. (2017) investigated the fate of total chromium in bulk water in ductile iron pipe loops, a commonly used distribution systems pipe material. Results indicated that Cr(VI), under certain water quality conditions, decreased in the bulk water which was consistent with predictions that Cr(VI) sorption to iron oxide surfaces occurs at pH less than 7.
7.4 Residential scale
Generally, it is not recommended that drinking water treatment devices be used to provide additional treatment of municipally treated water. In cases where an individual household obtains its drinking water from a private well, a private residential drinking water treatment device may be necessary for the removal of contaminants such as Cr(VI) from drinking water.
Health Canada does not recommend specific brands of drinking water treatment devices, but it strongly recommends that consumers use devices that have been certified by an accredited certification body as meeting the appropriate NSF/American National Standards Institute (ANSI) drinking water treatment unit standards. These standards have been designed to safeguard drinking water by helping to ensure the material safety and performance of products that come into contact with drinking water. Certification organizations provide assurance that a product conforms to applicable standards and must be accredited by the Standards Council of Canada (SCC). In Canada, the following organizations have been accredited by the SCC to certify drinking water devices and materials as meeting NSF/ANSI standards (SCC, 2014):
- CSA Group (www.csagroup.org);
- NSF International (www.nsf.org);
- Water Quality Association (www.wqa.org);
- UL LLC (www.ul.com);
- Bureau de normalisation du Québec (www.bnq.qc.ca); and
- International Association of Plumbing & Mechanical Officials (www.iapmo.org).
An up-to-date list of accredited certification organizations can be obtained directly from the SCC (2014).
Drinking water treatment technologies able to be certified to NSF standards for removal of total chromium as well as Cr(VI) and Cr(III) individually include adsorption, reverse osmosis and distillation.
NSF/ANSI Standards 53, 58 and 62 for chromium removal currently require testing of a device for the individual reduction of each chromium species from 0.3 to 0.1 mg/L. If the influent challenge total chromium concentration of 0.3 mg/L is prepared as 0.15 mg/L for both Cr(VI) and Cr(III), the devices certified to NSF/ANSI Standards 53 and 58 must be capable of reducing the individual chromium species to a maximum of 0.05 mg/L each (NSF/ANSI, 2013a, 2013b, 2013c). However, treatment devices certified to NSF/ANSI Standard 62 for chromium removal can be certified either specifically for chromium, as noted above, or for the removal of total chromium, using TDS as a surrogate for chromium. Treatment devices certified to NSF/ANSI Standard 62 using TDS as a surrogate must achieve an individual maximum treated water concentration of 0.05 mg/L from an influent (challenge) concentration of 0.15 mg/L for each chromium species (NSF/ANSI, 2013c).
Devices certified to NSF/ANSI Standard 58 and NSF/ANSI Standard 62 are intended for point-of-use installation only. RO and distillation systems should be installed only at the point of use, as the treated water may be corrosive to internal plumbing components. RO systems are also intended for point-of-use installation, as larger quantities of influent (incoming) water are needed to obtain the required volume of treated water, which is generally not practical for residential-scale point-of-entry systems. A consumer may need to pretreat the influent water to reduce fouling and extend the service life of the membrane.
Ion exchange treatment technology using anion based resins may also be a feasible for chromium removal in residential scale applications. Ion exchange technology is typically designed and constructed for residential use by drinking water treatment system providers or dealer. Health Canada strongly recommends that homeowners ensure that these systems are constructed using materials certified to NSF/ANSI Standard 61 (NSF/ANSI, 2013). If an ion exchange system is used, the water needs to be filtered through a GAC filter to remove any chlorine or chloramine from the water before it reaches the resin. It is important to routinely monitor the chromium concentration in the water treated by ion exchange to ensure that the system is effectively removing chromium and that chromatographic peaking is not occurring.
8.0 Kinetics and metabolism
8.1 Absorption
The water solubility and oxidation state of the different chromium compounds are important factors influencing their absorption rates via oral, inhalation and dermal routes. Cr(VI) is absorbed across cell membranes to a higher degree than Cr(III); however, its absorption is effectively reduced because of reduction to Cr(III) at the application sites (Donaldson and Barreras, 1966; De Flora et al., 1997).
Absorption of chromium following oral administration is relatively low in both rodents and humans. The absorption rates reported are generally < 2% for Cr(III) and ~7% for Cr(VI) (WHO, 1988; Kerger et al., 1996; ATSDR, 2012). Estimates of total chromium uptake in human volunteers ranged from 3% to 7% after ingesting 5–10 mg Cr(VI)/L in drinking water and 0.13% and 0.6% after ingesting Cr(III) chloride in drinking water, as determined by urinary excretion (Kerger et al., 1996, 1997). However, these values are based on urinary excretion and may underestimate the actual uptake, as they do not account for chromium retained in tissues. The higher rate of absorption of Cr(VI) than Cr(III) is largely explained by the capacity of Cr(VI) to enter the cells through anion transporter (sulphate/phosphate) channels, whereas Cr(III) enters (more slowly) by passive diffusion only (O'Brien et al., 2003; Collins et al., 2010). Extracellular reduction of Cr(VI) to Cr(III) at lower concentrations thereby limits chromium absorption.
The extent of Cr(VI) absorption from the gastrointestinal tract is a function of three competing rates: gastrointestinal transport, gastric reduction (both of which are a function of fasting state) and absorption. If the rate of reduction is far faster than the rate of transit and absorption, then little Cr(VI) will be absorbed. However, it was suggested that absorption from the gastrointestinal tract is so rapid that it is able to effectively compete with reduction in the stomach (Zhitkovich, 2011). In humans, the amount of Cr(VI) escaping reduction in the stomach after ingestion of water without food was estimated to be in the 10–20% range on the basis of both in vivo bioavailability estimates at low doses and in vitro gastric reduction rate estimates (Zhitkovich, 2011). When the capacity of reduction is exceeded (e.g., at high doses) or when the stomach emptying time is short (e.g. when water is ingested on an empty stomach), a possibly higher proportion of Cr(VI) may be available for absorption in the gastrointestinal tract and further enter portal venous blood, likely to be reduced in the liver (Kerger et al., 1997; O’Flaherty et al., 2001). Under such conditions, it is anticipated that the dose leading to saturation represents an inflection point for a sublinear exposure–response for blood and/or tissue concentrations, with doses at this point demonstrating a greater rate of response than lower doses (Collins et al., 2010).
Pulmonary absorption of inhaled chromium is believed to be greater than absorption via the gastrointestinal tract, with an estimated 20–30% of highly water-soluble Cr(VI) entering the bloodstream following exposure (European Chemicals Bureau, 2005). As with oral exposure, Cr(VI) is generally more readily absorbed than the trivalent form (Suzuki et al., 1984; Wiegand et al., 1988). Extracellular reduction of Cr(VI) to Cr(III) in the respiratory tract constitutes a line of defence against pulmonary chromium toxicity (De Flora et al., 1997; U.S. EPA, 1998a; De Flora, 2000).
Both Cr(III) and Cr(VI) can penetrate human skin to some extent, especially if the skin is damaged (ATSDR, 2012). In contrast to chromium salts, chromium metal is not considered likely to penetrate intact human skin under normal physiological conditions (Larese et al., 2007). In an in vitro permeation study performed with human skin, Cr(VI) passed through the skin more easily than Cr(III) and was also absorbed into the skin to a greater extent, both effects being attributed to rejection of positively charged Cr(III) by the skin barrier (Van Lierde et al., 2006). The permeation rates for human skin after 168 hours of exposure to chromium salts (1.7 g Cr/L) were < 0.09 µg/cm2 of skin for Cr(III) (chloride and nitrate) and 0.18 µg/cm2 of skin for Cr(VI)(potassium dichromate). The permeation rates for human skin (168 hours of exposure to an aqueous solution of Cr(VI) at 0.25–5%, with or without incubation in simulated sweat) ranged from < 0.09 to 0.23 µg/cm2 of skin (Van Lierde et al., 2006). Dermal absorption of Cr(VI) was also assessed in vivo with four human volunteers immersed in a bath containing 22 mg Cr(VI)/L (as potassium dichromate) for 3 hours (Paustenbach et al., 2003). Dermal penetration was estimated to be < 10% of daily ingestion of water, based on blood and urine samples. Dermal uptake rates ranged from 3.5 × 10−5 to 4.1 × 10−4 µg Cr/cm2 of skin per hour, with an average value of 1.4 × 10−4 µg Cr/cm2 of skin per hour.
8.2 Distribution
Tissue distribution of chromium depends on several factors, including the chemical form, the solubility and the route of exposure. When Cr(III) salts are administered orally or by inhalation, Cr(III) is presumed to be present in plasma as a stable mix of organic complexes with amino acids and other low molecular weight organic acids and with proteins, primarily globulins. The small fraction of chromium complexed with low molecular weight ligands is able to slowly diffuse into and out of plasma, blood and cells (O’Flaherty, 1996; Kerger et al., 1996, 1997; O’Flaherty et al., 2001; Paustenbach et al., 2003). Under physiological conditions, Cr(VI) as chromate is isostructural with sulphate and phosphate, thereby actively entering many types of cells by means of the anion exchange carrier pathway (Connett and Wetterhahn, 1983; Buttner and Beyersmann, 1985; Wiegand et al., 1985). However, the abundance of sulphate transporters varies by cell type (higher in mature differentiated cells), thereby modulating distribution of Cr(VI) (Silberg et al., 1995; Markovich, 2001).
Once Cr(VI) enters the red blood cells, it is reduced to Cr(III) by glutathione (GSH) or hemoglobin (Ebaugh et al., 1961; Aaseth et al., 1982; Wiegand et al., 1984). Plasma also reduces Cr(VI) but not as effectively as red blood cells(Korallus et al., 1984; Minoia and Cavalleri, 1988; Corbett et al., 1998). The newly formed Cr(III) is bound to hemoglobin, which has a higher affinity for Cr(III) than for Cr(VI) (Gray and Sterling, 1950; Wiegand et al., 1988), or to low molecular weight ligands (likely GSH; Aaseth et al., 1982; Wiegand et al., 1984) and is slowly lost from the cell, with a half-life of approximately 30 days and a mean residence time of 43 days (Eadie and Brown, 1954; Read, 1954).
Once in the bloodstream, absorbed chromium may be widely distributed throughout the body. The iron transport protein, transferrin, maintains chromium levels in the blood and transfers chromium to tissues in an insulin-responsive manner. Absorbed chromium distributes to nearly all tissues, with the highest concentrations being found in the kidneys, spleen and liver. Bone is also a major depot and may contribute to the long-term retention kinetics of chromium (ATSDR, 2012). A number of studies have shown that chromium can enter the central nervous system (as reviewed by Costa and Klein, 2006). Chromium can also cross the placental barrier in both rodents administered chromium intraperitoneally (Kirpnick-Sobol et al., 2006) and humans, as demonstrated in pregnant women with a metal-on-metal hip arthroplasty (Ziaee et al., 2007).
The distribution of chromium after oral exposure was recently studied in both male rats and female mice administered soluble Cr(VI) in drinking water (as SDD) or Cr(III) complexes in feed (as chromium picolinate monohydrate [CPM]) for 4, 11, 180 or 369 days (NTP, 2008; Collins et al., 2010). The highest concentration of SDD (516 mg SDD/L, 182 mg Cr(VI)/L) and the lowest concentration of CPM (2000 mg/kg in diet) provided quite similar oral doses of chromium (9 and 13 mg Cr(VI)/kg bw per day compared with 15 and 37 mg Cr(III)/kg bw per day in rats and mice, respectively). Total chromium concentrations measured in various tissues and in excreta 48 hours after cessation of exposure (i.e., on days 6, 13 and 182) revealed that exposure to Cr(VI),compared with exposure to Cr(III), led to 1) higher chromium concentrations in red blood cells (by 6- to 16-fold in rats and by 7- to 22-fold in mice), 2) similar chromium concentrations in plasma, 3) higher chromium concentrations in the stomach (by 2- to 14-fold in rats and by 6- to 28-fold in mice), 4) higher chromium concentrations in the liver (by 11- to 13-fold in rats and by 15- to 40-fold in mice) and the kidney (by 4- to 6-fold in rats and by 6- to 22-fold in mice) and 5) similar fecal and urinary excretion (by species). Species-specific differences were also revealed, with a higher absorption of chromium in mice than in rats (higher fecal excretion in rats) and, after normalization to external Cr(VI) dose, statistically higher chromium concentrations in the glandular stomach (by 4-fold) and liver (by 5-fold) of mice compared with rats and statistically significantly higher (by 1.4-fold) chromium concentrations in the kidney of rats. In both species and with both treatments, the tissue chromium concentrations increased with the duration of exposure up to 6 months (some exceptions with Cr(III) in the stomach), but with Cr(VI), the rate of accumulation decreased with longer (1year) exposure in all tissues except red blood cells and plasma (no 1-year data for Cr(III)) (Collins et al., 2010).
8.3 Metabolism
Reduction by fluids in the digestive tract (saliva 0.7-2.1mg/day and gastric juice 84-88 mg/day) and sequestration by intestinal bacteria (11-24 mg eliminated daily in feces) account for poor intestinal absorption of Cr(VI) in humans (De Flora et al., 1997). After oral exposure, most Cr(VI) that escapes reduction in the digestive tract is likely reduced in the blood of the portal vein system or in the liver, having an overall reducing capacity of 3300 mg (De Flora et al., 1997). Reduction of Cr(VI) also occurs in body fluids (e.g., alveolar fluids), red blood cells and nucleated cells.
Ex vivo studies using stomach contents from rats and mice found 1) that Cr(VI) reduction followed a mixed second-order process that is concentration and capacity limited (i.e., reduction capacity is not infinite); 2) a greater proportional reduction of Cr(VI) in rats compared with mice; 3) a non-linear reduction rate as a function of pH; and 4) support for exceedance of reducing capacity at ≥ 20 mg Cr(VI)/L (Proctor et al., 2012).
After Cr(VI) crosses cellular membranes, its reduction to Cr(III) may occur at various sites within the cell, including cytoplasm, endoplasmic reticulum, mitochondria and the nucleus. Cr(VI) tends to be reduced either directly or via intermediates in a network of mechanisms, leading to the generation of variable amounts of transient products, such as Cr(V), Cr(IV), and sulphur- and carbon-based radicals. In the presence of hydrogen dioxide, intermediate forms of chromium can generate highly reactive hydroxyl radicals (HO•) through the Fenton reaction (De Flora, 2000; Costa and Klein, 2006; Zhitkovich, 2011). Intracellular reduction of Cr(VI) to Cr(III) may involve small molecules (ascorbate, GSH and cysteine), soluble proteins (hemoglobin, glutathione reductase) and microsomal proteins (NADPH–cytochrome P450 reductase and cytochrome P450 transport systems) (Connett and Wetterhahn, 1983).
Reduction may result in either activation or detoxification, depending on the nature of the cellular components reducing Cr(VI), the site of the intracellular reaction and its proximity to deoxyribonucleic acid (DNA) (Bianchi and Levis, 1988). Reduction is a detoxification process when it occurs far away from DNA and when the reactive oxygen species (ROS) can be trapped by a large number of ligands, nucleophiles and antioxidants that are present in the intracellular environment (De Flora, 2000). Alternatively, when intracellular reduction occurs in the proximity of DNA, it may be an activation process, as reduction of Cr(VI) to Cr(III) in the cell is thought to be a prerequisite for the genotoxic action of chromium salts due to the generation of unstable chromium reduced intermediates (Cr(V) and Cr(IV)), ROS and Cr(III), which can react with other cellular constituents, such as proteins and DNA (De Flora, 2000; Zhitkovich, 2011).
8.4 Excretion
Whether originating from exposure to Cr(III) or to Cr(VI), absorbed chromium is largely eliminated in the urine in the trivalent form (Suzuki et al., 1984).
Following oral administration of soluble inorganic chromium salts in water, absorbed inorganic Cr(VI) originating from aqueous solutions behaves as Cr(III). However, the rate of elimination of chromium was found to be slower after ingestion of Cr(VI) as potassium dichromate (half-life ~40 hours) than after ingestion of soluble Cr(III) as chromium chloride (half-life ~10 hours) (ATSDR, 2012). Similarly, stabilization of Cr(VI) prior to ingestion (e.g., conversion to Cr(III) by orange juice) may result in more rapid excretion (O’Flaherty et al., 2001). Urinary clearance of chromium administered in orange juice more closely approximated chromium clearances seen in the general population exposed to ambient environmental sources of chromium. The similarity between Cr(III) and Cr(VI) kinetics in experimental models may thus not be applicable for environmental exposures to chromium or for other routes of exposure (e.g., inhalation) for which there are far fewer human kinetic data (O’Flaherty et al.,2001).
Elimination of chromium in feces and urine is similar in both rats and mice administered Cr(VI) as SDD in drinking water for 4, 11, 180 or 369 days (Collins et al., 2010). Within 48 hours after cessation of exposure, about 49% of the ingested dose was recovered in the feces of both species. In rats, the recovery in urine was 0.6–2.4% at the lowest exposure (5–20 mg Cr(VI)/L) and 0.2–0.95% at the higher exposures(60–180 mg Cr(VI)/L) (no detailed data for mice). When chromium was administered as a Cr(III) complex (CPM), fecal excretion was lower, especially in mice (42% in rats, 20% in mice).
8.5 PBPK models
Two multi-compartment physiologically based pharmacokinetic (PBPK) models, developed to describe the internal behaviour of Cr(III) and Cr(VI) in mice and rats (Kirman et al., 2012) and humans (Kirman et al., 2013), were used in this assessment. Although other models (Schlosser and Sasso, 2014, 2015) were subsequently developed and are discussed below, the small quantitative changes they made to the results did not warrant remodelling.
8.5.1 Models used in this assessment
The Kirman et al. (2012, 2013) models included compartments for oral mucosa, gastrointestinal lumen, forestomach/stomach, small intestine, blood, liver, kidney, bone and a combined compartment for remaining tissues. As chronic exposure to high Cr(VI) concentrations in drinking water causes small intestinal cancer in mice (NTP, 2008), the toxicokinetics of Cr(VI) in the upper gastrointestinal tract of rodents and humans is important for assessing internal tissue dose in risk assessment.
For the rodent model development, ex vivo Cr(VI) reduction data were used to characterize reduction of Cr(VI) in fed rodent stomach fluid as a second-order, pH-dependent process (Proctor et al., 2012); tissue time-course data for total chromium were collected from rats and mice exposed to Cr(VI) in drinking water for 90 days at six concentrations ranging from 0.1 to 180 mg Cr(VI)/L (Thompson et al., 2011); and tissue time-course data were collected from chronic oral Cr(III) and Cr(VI) bioassays (NTP, 2007, 2008). For model validation, the data sets for rats and mice exposed to SDD in drinking water for 21 days (NTP, 2007) were used. Sensitivity analyses were conducted by adjusting model parameter values by 5% and then reporting the relative impact on several predicted measures of internal dose. Overall, the PBPK rodent model provides a good description of chromium toxicokinetics, with model predictions within a factor of 3 or less for approximately 90% of rat data points and 80% of mouse data points. The model predicted 1) species differences for chromium delivery to the target tissue (small intestine), with higher concentrations in mice than in rats, consistent with small intestinal tumour formation in mice, but not in rats; 2) a concentration gradient in the small intestine (duodenum > jejunum > ileum), consistent with the tumour response gradient observed in mouse small intestine; and 3) that Cr(VI) enters portal circulation in rodents at drinking water concentrations ≥ 60 mg/L based on erythrocyte:plasma chromium ratios (however, this might be a limited measure of systemic absorption, if Cr(VI) is systemically absorbed but reduced to Cr(III) in erythrocytes).
The rodent model was adapted to develop the human model, based on 1) ex vivo Cr(VI) reduction studies conducted using fasted human stomach fluid to characterize the reduction of Cr(VI) in human stomach fluid as a mixed second-order, pH-dependent process; and 2) toxicokinetic data for total chromium in human tissues and excreta from published literature. For model validation, the data sets of Finley et al. (1997) were used. Sensitivity analyses were conducted by adjusting model parameter values one at a time by 5% and then reporting the relative impact on several predicted measures of internal dose. Overall, the PBPK model provides a good description of chromium toxicokinetics—with model predictions within a factor of 3 or less for approximately 90% of Cr(VI) data—and is consistent with the available total chromium data from Cr(III) and Cr(VI) exposures in typical humans.
For application of the model for risk assessment, exposures to Cr(VI) in drinking water were modelled assuming 1) three exposure events per day during meals (fed state) and three exposure events per day between meals (fasted state); 2) six exposure events per day during meals (fed state); and 3) six exposure events per day between meals (fasted state). Assuming that all exposures occurred during a fasted state represents the worst-case scenario.
Although two internal dose measures were evaluated for calculating human equivalent dose (HED) values (pyloric flux, defined as the amount of Cr(VI) leaving the stomach lumen divided by the volume of small intestine per day; small intestine flux, which is defined as the amount of Cr(VI) entering the small intestinal epithelium divided by the volume of small intestine per day), greater confidence is placed on pyloric flux. Pyloric flux values have less uncertainty, as they are dependent primarily on well-characterized processes (gastric lumen transit, gastric lumen reduction). Pyloric flux and small intestine flux yielded nearly identical HED results.
The authors indicated several sources of uncertainty in the models, as follows. The models did not consider potential changes in gastrointestinal (GI) tract kinetics due to toxic effects in the tissues (e.g., hyperplasia, altered villus structure) or contributors to chromium reduction other than stomach acids (e.g., bile, pancreatic secretions, intestinal secretions, enzymatic activity, bacterial flora). Erythrocyte Cr(VI) reduction data were used as a surrogate for reduction in GI tissues, due to an absence of data on the latter. Cr(VI) reduction rates for humans were based on samples only from fasted individuals. For simplicity and based on data limitations, Cr(VI)-reducing agents were modelled as a single lumped pool, whereas multiple reducing agents—likely with different rates and capacities of Cr(VI) reduction—exist in the GI lumen. pH dependence of Cr(VI) reduction rates in humans was characterized quantitatively using a simple empirical calculation, based on data from pooled gastric fluid samples of humans with normal gastric pH (separate pools for pH values of approximately 1 [n = 3], 2 [n = 2], and 4 [n = 5]) and pH altered by use of proton pump inhibitors (pH of approximately 7; n = 5).
8.5.2 Newer models
Newer models (Schlosser and Sasso, 2014, 2015) have been developed, building on the Kirman et al. (2012, 2013) models. They address the last two sources of uncertainty described above. A mechanistic description of pH dependence was added to the Kirman et al. (2012, 2013) model, using assumptions of shifting relative abundance of Cr(VI) species due to pH-dependent chemical equilibria—along with differing reduction rates for each species—at varying pH levels. They also assumed that the rate constant was unchanged with dilution of human gastric juice, whereas Kirman et al. (2013) assumed proportional increase in rate constant with increases in dilution. The newer models also described multiple pools of reducing agents, used to reflect the variety in reducing agents in the GI tract (e.g., ascorbate, NADH, and glutathione). They included up to three generic pools (an initial pool that reacts and is depleted quickly, a second pool that reacts and is depleted more slowly, and a third pool that reacts and is depleted very slowly), with kinetic parameters for each estimated by model optimization. The use of multiple pools of reducing agents improved model fits in rat and mouse data at higher doses (the Kirman et al. [2012] model demonstrated good fit to the data a low doses).
The Schlosser and Sasso model also predicted a large decrease in—but not complete depletion of—Cr(VI) reduction capacity in rats and mice. In humans, the single pool model was sufficient to fit the data; however, improved fit to human data was obtained due to the pH model inclusion and reoptimization of parameters for the single-pool model. The latest work also updated the GI kinetics into more consistent model for all species.
8.5.3 Comparison of models
Although the fit of the (Schlosser and Sasso, 2014, 2015) models was improved over that of the original models (Kirman et al., 2012, 2013), the dose–response analysis data provided to Health Canada from the original models (Summit Toxicology, 2014) was retained for this assessment, as fit of the original models was still considered to be within an acceptable range, particularly at lower doses. Moreover, Sasso and Schlosser (2015) identified that application of the updated model to the point of departure used in a dose–response assessment by Thompson et al. (2014) resulted in a human equivalent dose that was within an order of magnitude of those calculated using the original model, with the original model more conservative at a pH of 2.5, and the updated model more conservative at a pH of 5.
Older PBPK models were developed for ingestion of Cr(VI) and Cr(III) by humans (O’Flaherty et al., 2001) and rats (O’Flaherty, 1996). However, they did not include parameterization of the gastrointestinal tract (target tissue for Cr(VI)) and were developed using few oral exposure data.
By modelling key species differences, sources of saturable toxicokinetics and sources of uncertainty and variation, the rodent and human PBPK models can provide a robust characterization of toxicokinetics in the target tissue (small intestine).
9.0 Health effects
9.1 Effects in humans
Chromium toxicity in humans varies depending on the form of the compound, its valence state and the route of exposure. Little information has been reported on the trivalent form of chromium, and available data, mainly relating to mixtures with Cr(VI) and Cr(III), show little or no toxicity associated with the trivalent form. However, several studies agree on the toxicity of the hexavalent form, which is soluble in water and represent the vast majority of the chromium present in drinking water.
9.1.1 Essentiality
Adequate intakes (AIs) have been proposed by the U.S. National Academy of Sciences in partnership with Health Canada (Institute of Medicine, 2001), reflecting current estimates of average chromium intake from well-balanced diets. These AIs range from 0.2 µg chromium per day (for infants) to 45 µg chromium per day (for lactating women). There is currently no consensus concerning the essentiality of chromium in humans. Chromium was considered essential based on its regulation of glucose metabolism and lipids (Broadhurst and Domenico, 2006). However, such data were found in diabetics or total parenteral nutrition patients only, and no measurable benefits, such as the popularly promoted conversion of fat into muscle, were demonstrated in healthy people (Pittler et al., 2003; Trumbo and Ellwood, 2006; Balk et al., 2007;Vincent and Stallings, 2007). Stearns (2000) stated that “there is no direct evidence of chromium deficiencies in humans,” and the U.S. Food and Drug Administration concluded that there was no evidence of a beneficial effect of Cr(III) supplementation with chromium picolinate (Trumbo and Ellwood, 2006).
9.1.2 Acute toxicity
A small number of cases of fatal oral ingestion of chromium have been documented. In all cases, highly water-soluble forms of Cr(VI) were implicated (e.g., sodium/potassium dichromate). About 1 g of potassium dichromate is considered a lethal dose (ATSDR, 2012), and deaths of children and adults have been reported at doses ranging from 4.1 to 357 mg/kg bw. Symptoms of acute chromium intoxication associated with the ingestion of a lethal dose of chromate include gastrointestinal, respiratory, liver and kidney injury, as well as cardiovascular collapse due to severe hypovolemia; fatty degeneration and metabolic acidosis were also observed at 357 mg/kg bw (Baresic et al., 2009; ATSDR, 2012).
No apparent clinical changes or health effects were reported in several studies performed in human volunteers exposed to lower doses, such as 0.03–4 mg Cr(VI)/day via drinking water for at least 3 days (Paustenbach et al., 1996; Finley et al., 1997; Kerger et al., 1997) or 5 mg of Cr(III) or Cr(VI) as a single dose via drinking water or orange juice (Kerger et al., 1996).
9.1.3 Subchronic and chronic toxicity and carcinogenicity
Some cross-sectional and ecological studies have found associations between exposure to Cr(VI) in drinking water and the odds of lung, liver and stomach cancer, while others found no associations. An association has also been observed between mean chromium blood concentration and oral cancer. Occupational studies have observed an increase in the risk of lung and stomach cancer following inhalation of Cr(VI). The weight of evidence for these effects is presented in detail in the subsequent sections. The epidemiological database on chromium does not allow drawing definitive conclusions on cancer effects following oral exposure considering the results inconsistencies, the difference in the route of exposure (most cancer studies were inhalation occupational), and the major limitations of the individual studies; however, it is supportive of the choice of an animal carcinogenicity key endpoint.
9.1.3.1 Environmental exposure
Oral ulcers, diarrhea, abdominal pain, indigestion and vomiting, leukocytosis and presence of immature neutrophils were reported in a Chinese population using a well (20 mg Cr(VI)/L) adjacent to a chromium alloy plant as a source of drinking water (Zhang and Li, 1987). The study also reported elevated mortality rates for stomach cancer and lung cancer in communities with Cr(VI)-contaminated well water in the vicinity of the chromium alloy plant when compared with unexposed regions. However, this ecological study did not report statistical measures of association or precision or individual exposures or account for numerous confounding factors. A reanalysis of the original data from Zhang and Li (1987) found significantly increased stomach cancer mortality in five regions with chromium reported in the drinking water (downstream from a ferrochromium alloy plant) compared with four nearby regions without contaminated water (rate ratio = 1.82; 95% confidence interval [CI] = 1.11–2.91) and compared to the whole province (rate ratio = 1.69; 95% CI = 1.12–2.44) (Beaumont et al., 2008; Wilcox et al., 2008). All-cancer and lung cancer mortality rates were also elevated compared with the rates in the whole province (rate ratio = 1.23, 95% CI = 0.97–1.53, and rate ratio = 1.78, 95% CI = 1.03–2.87). However, this study did not consider important covariates, such as the demographic differences between the agricultural villages and the industrial town (both combined as unexposed) and employment at the nearby plant; moreover, there was no dose–response data, the ecological study design did not allow determination of individual exposure bias is possible because some data from the original analysis had to be reconstructed by the authors, and only data on mortality—and not incidence of cancer—were available. A second reanalysis found that the average mortality rates from all cancer, lung cancer or stomach cancer were not significantly different between chromium-exposed and unexposed villages using three surrogates of exposure (distance from the alloy plant, water Cr(VI) concentration in the 1965 survey, and percentage of wells exceeding 0.05 ppm) (Kerger et al., 2009). However, the relative risk of stomach cancer was increased in the exposed villages compared with the industrial town. Weaknesses for this reanalysis include: 1) absence of adjustment for many confounders, such as other groundwater contaminants, smoking, and infections; 2) absence of data on chromium exposure characterization; 3) evaluation of health effects was performed in relatively short period of time after the beginning of the exposure period (~15 years), which might be insufficient latency duration for cancers to occur; and 4) the study has been criticized based on the possibility of conflict of interest.
In Taiwan, mean blood chromium concentrations were about 2‑fold higher in oral cancer patients (0.795 ± 0.26 µg/L; n = 79 patients) than in non-cancer residents (0.44 ± 0.392 µg/L; n = 641 residents) in an agricultural region of Taiwan where soils were contaminated by chromium and other heavy metals (Chiang et al., 2011). Weaknesses of the study include an absence of assessment of total environmental and drinking water exposures, co-occurrence of nickel exposures, and concerns that the two groups might differ in other ways than Cr exposure, especially since the authors indicated that most cancer patients were smokers and chewed betel.
In Greece, a population exposed to drinking water contaminated by industrial waste (with Cr(VI) at concentrations up to ~50 μg/L) since the 1990s demonstrated an association between Cr(VI) exposure and an increase in liver cancer mortality (standardized mortality ratio [SMR] = 1104, 95% CI = 405–2403) (Linos et al., 2011). However, the results are very likely to be confounded by other chemical contaminants (e.g., arsenic) in the water.
Other studies have found no association between exposure to chromium and cancer prevalence. In central Mexico, no adverse health effects (including septum perforation, lung and unusual cancers, and birth defects, ascertained by self-report in a door survey) were associated with exposure to 0.5 mg Cr(VI)/L (Armienta-Hernandez and Rodriguez-Castillo, 1995).in drinking water However, the methodology was poorly detailed, especially for the investigation of the health outcomes. An ecological study in Nebraska looking at correlations between various drinking water contaminants and multiple health conditions found no evidence of carcinogenicity of chromium in drinking water (Bednar and Kies, 1991). The authors noted that the MCL for chromium in drinking water was 50 µg/L in this state. Moreover, another ecological study found no increase in lung or total cancer mortality risk in residents possibly exposed to Cr in California (living near an industry using and disposing of Cr in ponds near their water source) compared with other areas in the same county. Conclusions are limited since exposure levels were not reported, mortality rates were based on census data, and there were no adjustment for covariates other than age (Fryzek et al., 2001).
9.1.3.2 Occupational exposure
Numerous occupational studies have reported that exposure to Cr(VI) by inhalation was associated with an increased risk of lung cancer (Hayes et al., 1979; Sorahan et al., 1987; Pastides et al., 1994; Mancuso, 1997a, 1997b; Kimbrough et al., 1999; Gibbet al., 2000; Crumpet al., 2003; Park et al., 2004; Cole and Rodu, 2005; Park and Stayner, 2006).
There is currently no consensus regarding a possible association between occupational exposure to Cr(VI) by inhalation and the occurrence of cancer in the gastrointestinal tract. Two reviews have reported that occupational inhalational exposure to Cr(VI) may induce cancer of the stomach, larynx, kidney, prostate, bladder, brain, small intestine and genital organs, as well as Hodgkin’s disease, lymphoma and leukemia (Siemiatycki, 1991; Costa and Klein, 2006). Conversely, a meta-analysis including 29 risk estimates on the associations between the gastrointestinal (GI) tract cancers (oral cavity, oesophagus, stomach, small intestine, colon, and rectum) found no increase in the risk of GI tract cancer at the levels of exposure experienced (estimated to be ~0.25 mg Cr(VI)/day, based on the previous occupational limit of 50 µg/m3 and an ingested fraction of 50% of inhaled Cr(VI)), except for oesophageal cancer (SMR = 1.49, 95% CI = 1.06–2.09) when grouping highly exposed workers in the U.S. in 4 studies, of which only one observed a significant association (Gatto et al., 2010). The conclusions based on this study are limited because the results are likely to be biased by the healthy worker effect (most individual studies used exterior groups as control), there was high heterogeneity across studies, the exposure was poorly characterized with industries reporting different grouping categories, and the included studies did not control for major confounders. Also, the positive relationship was driven by only one exploratory study in U.S. workers.
A recent meta-analysis has reported an association between stomach cancer and occupational inhalation exposure to Cr(VI) after combining 74 risk estimates from 56 cohort and case–control peer-reviewed studies (RR = 1.27, 95% CI = 1.17–1.38) (Welling et al.,2015). The authors included all occupations known for their exposure to chromium and risk estimates for the highest exposure categories available. Cement and leather work were associated with the highest RR for stomach cancer. The authors were confident in the results because the risk of publication bias, occurring when positive results tend to be more published, and of chance findings were calculated to be low. However, there was significant heterogeneity across studies (48% of the observed variations can be attributed to heterogeneity rather than chance), not all included studies adjusted for important confounders (e.g., Helicobacter pylori infection, lifestyle, exposure to asbestos) other than age and sex, case-control studies are susceptible to recall bias, and the exposure metrics varied across studies (no exposure estimates was provided by the authors). In support of this association, three studies found an association between occupational exposure to Cr(VI) and stomach cancer in the Portland cement manufacturing, chromate production or concrete mixing industries (McDowall, 1984; Rosenman and Stanbury, 1996; Knutsson et al., 2000). However, conclusions based on these studies are limited since the risk did not increase with years of exposure, the workers were co-exposed to other carcinogens, the concentration of Cr(VI) is low in cement, and other studies conducted on Cr(VI) workers did not report associations with stomach cancer (Davies et al., 1991; Satoh et al., 1991).
Non-neoplastic effects on the respiratory system have been reported in chrome platers exposed subchronically to chromic acid mists in air containing Cr(VI) at concentrations higher than 0.001 mg/m3 (Kleinfeld and Rosso, 1965; Hanslian et al., 1967; Gomes, 1972; Cohen et al., 1974; Lucas and Kramkowski, 1975; Royle, 1975; Kuo et al., 1997; U.S. EPA, 1998a). Various gastrointestinal, hepatic and renal effects have also been observed in workers in the chromate production and chromium plating industries (Wang et al., 2011a, 2011b; ATSDR, 2012). In addition, excess deaths related to mental, psychoneurotic and personality disorders were reported in an epidemiological study of several thousand chrome workers, with an observed/expected ratio of 2.41 for all races (ratio ranging from 1.78 to 5.61, depending on race groups) (Gibb et al., 2000).
9.1.4 Genotoxicity
Numerous studies have been conducted in vivo (in humans exposed occupationally, mainly by inhalation) and in vitro (human cell lines) to assess the genotoxicity of both trivalent and hexavalent chromium compounds, as reviewed by the International Agency for Research on Cancer (IARC, 1990), De Flora et al. (2008), Sanexen (2009), the Agency for Toxic Substances and Disease Registry (ATSDR, 2012) and Urbano et al. (2012).
With Cr(III) compounds, increased micronucleus frequency and DNA–protein crosslinks were observed in the peripheral lymphocytes of tanners primarily exposed to Cr(III) compounds (Medeiros et al., 2003), and some in vitro studies yielded positive responses in human lymphocytes (Nakamuro et al., 1978; Stella et al., 1982; Blasiak and Kowalik, 2000). However, it was further suggested that positive results in vitro in intact cells could be due to artifacts such as contamination of the test compounds with traces of Cr(VI), non-specific effects at very high doses, experimental conditions that would increase the penetration of Cr(III) into cells (e.g., detergents) or a technical artifact formed during the extraction procedures (De Flora et al., 1990;IARC, 1990; De Flora, 2000).
In workers exposed occupationally to Cr(VI), several studies reported increased levels of chromosomal aberrations, sister chromatid exchanges, DNA strand breaks, DNA–protein crosslinks or micronuclei in peripheral lymphocytes or buccal cells (Sarto et al., 1982; Stella et al., 1982; Koshi et al., 1984; Deng et al., 1988; IARC, 1990; Lai et al., 1998; Werfel et al., 1998; Vaglenov et al.,1999; Dana Devi et al., 2001; Halasová et al., 2001; Wu et al., 2001; Benova et al., 2002; Gambelunghe et al., 2003; Medeiros et al., 2003). Direct correlations have been reported between levels of workplace chromium or duration of chromium exposure (inhalation) and amount of genetic damage detected (IARC, 1990). However, some of these studies have limitations, such as possible co-exposure to other genotoxic factors or lack of correlation between the effect and exposure to chromium. In contrast, no increased number of chromosomal aberrations, sister chromatid exchanges, DNA strand breaks, oxidative DNA damage (8-hydroxydeoxyguanosine [8-OHdG]) or DNA–protein crosslinks were reported by Husgafvel-Pursiainen et al. (1982), Littorin et al. (1983), Nagaya (1986), Nagaya et al. (1991), Kuykendall et al. (1996) and Benova et al. (2002). No correlation was found between unscheduled DNA synthesis in pleural mesothelial cells and chromium concentrations in urine of workers from chromium plating factories (Pilliere et al., 1992).
9.1.5 Reproductive and developmental toxicity
Some occupational studies have found associations between Cr(VI) exposure via inhalation and reproductive health outcomes in males, including an increased number of morphologically abnormal sperm in workers in a chromate industry (duration of employment not reported; Kumar et al., 2005) and decreased sperm counts and motility, increased serum follicle stimulating hormone concentration and a significant decrease in semen lactate dehydrogenase activity in workers employed for 1–15 years in an electroplating factory (Li et al., 2001). The levels of exposure were not available. Other studies have found no effects of Cr(VI) occupational inhalation on male reproduction. No significant differences in semen quality biomarkers or levels of sex hormones (follicle stimulating hormone, luteinizing hormone, and testosterone) were observed in a cohort of welders compared with non-metal workers (Hjollund et al., 1998) and in a cross-sectional study comparing welders with non-welder workers (Bonde and Ernst, 1992). No reliable studies were located for female reproduction or risk of human birth defects (i.e., available studies were of poor quality and were poorly reported).
Although the epidemiological evidence as a whole supports an association between exposure to chromium and an increased risk of cancer, especially for occupational inhalation exposure and lung cancer, significant study limitations prevent its use for the derivation of a guideline.
9.2 Effects on experimental animals
The toxicity of chromium compounds depends principally on valency, as well as on the physicochemical properties of specific compounds, with Cr(VI) being more toxic than Cr(III). The studies summarized below are the most relevant ones for drinking water exposure.
9.2.1 Acute toxicity
In general, the acute toxicity of chromium compounds in experimental animals increases with their solubility in water. The oral median lethal dose (LD50) values reported for trivalent compounds vary between 183 and 422 mg Cr(III)/kg bw for chromium nitrate (rats) and between 140 (rats) and 390 mg Cr(III)/kg bw (mice) for chromium phosphate in aqueous solution; the LD50 is 2365 mg Cr(III)/kg bw for the less soluble chromium acetate (Fairhurst and Minty, 1989; ATSDR, 2012). Exposure to Cr(VI) compounds, such as potassium dichromate, sodium dichromate, ammonium dichromate and sodium chromate, generated LD50 values ranging from 13 to 20 mg Cr(VI)/kg bw for female rats and from 23 to 28 mg Cr(VI)/kg bw for male rats (Fairhurst and Minty, 1989; ATSDR, 2012). For chromium trioxide, the LD50 values range from 27 to 59 mg Cr(VI)/kg bw for rats and from 70 to 91 mg Cr(VI)/kg bw for mice (European Chemicals Bureau, 2005). Exposure to calcium chromate yielded LD50 values of 108 and 249 mg Cr(VI)/kg bw for female and male rats, respectively (ATSDR, 2012). A higher LD50 value (811 mg Cr(VI)/kg bw) was reported for strontium chromate in male rats (ATSDR, 2012).
After acute oral exposure to Cr(VI), the toxicity signs observed include hypoactivity, lacrimation, diarrhea, pulmonary congestion and corrosion of mucosa in the gastrointestinal tract (European Chemicals Bureau, 2005; ATSDR, 2012). Recent acute toxicity studies also reported oxidative stress, apoptosis and hepatotoxicity in rats (Soudani et al., 2011a, 2013) and mice (Wang et al., 2009).
9.2.2 Short-term exposure
No toxic effects were observed following the administration of chromium chloride (Cr(III)) to rats in drinking water (25 mg/L for 1year) or in the diet (100 mg/kg for 90 days or 9 mg/kg bw per day for 20 weeks) (Fairhurst and Minty, 1989; Anderson et al., 1997). No toxic effects were observed in rats or mice following administration of organic forms of Cr(III), such as chromium picolinate, in their diet at doses ranging from 9 mg/kg bw per day for 20 weeks to 1415 mg/kg bw per day for 14 weeks (Anderson et al., 1997; Rhodes et al., 2005; NTP, 2008). No toxic effects were observed in female rats fed chromium nicotinate for 90 days (1.5 mg Cr(III)/kg bw per day) or for 38 weeks (0.25 mg/kg bw per day) (Shara et al., 2005). No effect on body weight was observed in mice after exposure to drinking water containing 500 mg Cr(III)/L (corresponding to 140 or 165 mg Cr(III)/kg bw per day for females and males, respectively) as chromium potassium sulphate for 210 days (De Flora et al., 2006). In contrast, administration of Cr(III) as chromium chloride in drinking water for 12 weeks decreased the body weight of male mice at 5 mg/kg bw per day (no effect at 14 mg/kg bw per day in females) (Elbetieha and Al-Hamood, 1997) as well as the body weight of rats (decreased by 24%) at 40 mg Cr(III)/kg bw per day (Bataineh et al., 1997). Decreased spleen and liver weights were reported in rats ingesting chromic oxide in the diet (1806 mg Cr(III)/kg bw per day) for 90 days (Ivankovic and Preussmann, 1975).
Most short-term oral toxicity studies have been conducted in animals administered Cr(VI) by gavage, through the diet or, more recently, through drinking water. The most sensitive endpoint, lesions in the small intestine, was observed in the drinking water studies, which are summarized below.
Three recent subchronic (90-day) studies conducted using a similar study design withF344 rats and B6C3F1, BALB/c and C57BL/6 mice administered SDD in drinking water revealed non-neoplastic microscopic lesions in the small intestine of both sexes of both species (NTP, 2007; Thompson et al., 2011, 2012c). The SDD concentrations tested by Thompson et al. were 0, 0.3, 4, 14 [mice only], 60, 170 and 520 mg/L (corresponding to 0, 0.1, 1.4, 4.9 [mice only], 21, 60 and 182 mg Cr(VI)/L), and those tested by the NTP (2007) were 0, 62.5, 125, 250, 500 and 1000 mg/L (corresponding to 0, 22, 44, 88, 175 and 350 mg Cr(VI)/L). In the small intestine, the lesions were observed in the duodenum and jejunum; for each lesion, there was a dose-related increased incidence or severity. The lesions included villous atrophy, crypt cell hyperplasia, apoptosis, histiocytic infiltration in the villous lamina propria, micronucleated syncytia in the villous lamina propria (mice only) and villous cytoplasmic vacuolization (mice only). In both species, the lowest-observed-adverse-effect level (LOAEL) for the observation of non-neoplastic lesions in at least one of these studies was 21–22 mg Cr(VI)/L (corresponding to 2.9 mg Cr(VI)/kg bw per day for rats and to 2.6–4.6 mg Cr(VI)/kg bw per day for mice). In rats, the first lesions to appear were apoptosis, hyperplasia and histiocytic infiltration in the duodenum. In mice, the first lesions were hyperplasia and histiocytic infiltration in the duodenal villi as well as villous cytoplasmic vacuolization in both the duodenum and the jejunum. The lesions observed in the duodenum of mice were “considered consistent with regenerative hyperplasia secondary to previous epithelial cell injury” (NTP, 2007). Qualitatively, the only notable species-specific difference observed at the histopathological level was the lack of cytoplasmic vacuolization in the villous epithelium of the duodenum and jejunum in rats, whereas this lesion was among the most sensitive in mice. The study also reported lesions on the liver and the pancreatic lymph node (histiocytic infiltration from 22 mg Cr(VI)/L) and in the glandular stomach and bone marrow (350 mg Cr(VI)/L) of rats, as well as lesions in the mesenteric lymph node (histiocytic infiltration from 44 mg Cr(VI)/L) of mice. No lesions were reported in the oral mucosa of both species in any of the three studies (NTP, 2007).
In addition to the effects observed after 3 months of continuous exposure, Thompson et al. (2011, 2012c) reported histopathological changes in the duodenum and jejunum of mice and rats after only 1 week of exposure; no lesion was detected in the oral cavity of either species. In mice, the results indicated the occurrence of villous cytoplasmic vacuolization in both the duodenum and jejunum from 60 mg Cr(VI)/L, as well as villous atrophy in the duodenum and crypt cell hyperplasia in both the duodenum and the jejunum at 182 mg Cr(VI)/L (Thompson et al., 2011). In rats, apoptosis and crypt cell hyperplasia occurred in the duodenum from 21 mg Cr(VI)/L and in the jejunum from 60 mg Cr(VI)/L, whereas villous atrophy and histiocytic infiltration in the villi occurred in both the duodenum and jejunum from 60 mg Cr(VI)/L; there was no cytoplasmic vacuolization (Thompson et al., 2012c).
Subchronic oral exposure to Cr(VI) has also been shown to significantly decrease body weight in mice administered SDD in drinking water for 3 months (22 mg Cr(VI)/L, corresponding to 3.1 mg Cr(VI)/kg bw per day; NTP, 2007). Other studies also reported body weight decreases in mice and rats administered higher doses of Cr(VI) compounds (SDD, potassium dichromate) in drinking water for 4–30 weeks (Bataineh et al., 1997; Elbetieha and Al-Hamood, 1997; De Flora et al., 2006; NTP, 2007; Quinteros et al., 2007; Thompson et al., 2011, 2012c). No effect on body weight was observed in rabbits (3.6 mg Cr(VI)/kg bw per day), rats (9.8 mg Cr(VI)/kg bw per day for 9 weeks) or mice (48 mg Cr(VI)/kg bw per day for 9 weeks) administered potassium dichromate by gavage (NTP,1996a, 1996b; Yousef et al., 2006). However, dramatic decreases in body weight of rats (by 57% and 59%) were observed after administration of sodium dichromate by gavage (40 or 60 mg Cr(VI)/kg bw per day) for 3 months (Chowdhury, 1995).
Effects on the liver and hematology were reported in the U.S. National Toxicology Program (NTP) dietary studies conducted with potassium dichromate. A possible bone marrow/erythroid response was observed at a high dose, and effects in the liver were observed at a lower dose (second study). In the first study (NTP, 1996b), Sprague-Dawley rats received chromium in the diet (0, 15, 50, 100 and 400 mg/kg) for 9 weeks, followed by an 8-week recovery period. Slightly reduced mean red cell volume (MCV) and mean red cell hemoglobin (MCH) were observed at the highest dose, leading to a no-observed-adverse-effect level (NOAEL) of 100 mg/kg (equivalent to 2.1 and 2.45 mg Cr(VI)/kg bw per day for male and female rats, respectively). The authors considered the results suggestive of a potential bone marrow/erythroid response, as revealed by anemia due to slightly reduced MCV and MCH. In the second study (NTP, 1996a), potassium dichromate was administered to mice in the diet (0, 15, 50, 100 and 400 mg/kg) for 9 weeks, followed by a recovery period of 9 weeks. The achieved dose levels were 0, 1.1, 3.5, 7.4 and 32 mg Cr(VI)/kg bw per day for males and 0, 1.8, 5.6, 12 and 48 mg Cr(VI)/kg bw per day for females, respectively. A maximum tolerated dose was achieved at 400 mg/kg (males) and 100 mg/kg (females). No treatment-related effects were noted for clinical signs, necropsy findings, microscopic evaluation or hematology findings, except for decreased MCV and possibly MCH in the 400 mg/kg males and females, suggesting a possible bone marrow/erythroid response. This effect returned to normal by week 17 in the 400 mg/kg female mice but increased in the 400 mg/kg male mice (no hematological effects at ≤ 7.4 and ≤ 12 mg Cr(VI)/kg bw per day in male and female mice, respectively). Cytoplasmic vacuolization in hepatocytes was noted in the 50, 100 and 400 mg/kg males and females, leading to a NOAEL of 1.1 and 1.8 mg Cr(VI)/kg bw per day for male and female mice, respectively.
9.2.3 Long-term exposure and carcinogenicity
No definitive evidence of toxicity or carcinogenicity following chronic oral exposure to Cr(III) was reported. No effects were found after a 2-year exposure of F344/N rats and B6C3F1 mice to Cr(III) administered as dietary chromium picolinate (up to 50 000 mg/kg; average doses: up to 316 and 788 mg Cr(III)/kg bw per day for rats and mice, respectively), except for an increased incidence of preputial gland adenoma in male rats at the middle dose only (55 mg Cr(III)/kg bw per day) (NTP, 2010). In addition, no dose-related changes were reported in ddY mice (25–100 mg Cr(III)/L drinking water for 1year; Maruyama, 1982), rats (fed bread with up to 2040 mg Cr(III)/kg bw per day for 2years; Ivankovic and Preussmann, 1975), Long-Evans rats (up to 0.46 mg Cr(III)/kg bw per day in drinking water for 2–3 years; Schroeder et al., 1965) or mice(0.48 mg Cr(III)/kg bw per day in drinking water for 2–3 years; Schroeder et al., 1964).
The NTP demonstrated that Cr(VI) is carcinogenic to rodents when administered in drinking water for 2 years as SDD, inducing neoplasms of the oral cavity and small intestine in rats and mice, respectively (NTP, 2008; Stout et al., 2009). Cr(VI) induction of histiocytic cellular infiltration in the liver, small intestine, and pancreatic and mesenteric lymph nodes of rats and mice and diffuse epithelial hyperplasia in the small intestine of mice were also reported in the NTP study. The details of these studies are provided below.
In the rat study, groups of 50 male and 50 female F344/N rats received 0, 14.3, 57.3, 172 or 516 mg SDD/L drinking water for 2 years (equivalent to 0, 5, 20, 60 and 180 mg Cr(VI)/L or 0, 0.2, 0.8, 2.1 and 5.9 mg Cr(VI)/kg bw per day for males and 0, 0.2, 0.9, 2.4 and 7.0 mg Cr(VI)/kg bw per day for females, respectively; NTP, 2008; Stout et al., 2009). Survival of exposed groups was similar to that of the control groups. Mean body weights of 516 mg SDD/L males and females were reduced, thought to be partly due to poor palatability of the dosed water. The incidences of squamous cell carcinoma in the oral mucosa of 516 mg SDD/L male and female rats were significantly higher than those in the controls. The incidence of oral tumours in 172 mg SDD/L females exceeded the historical control ranges for drinking water studies. The incidences of squamous cell papilloma or squamous cell carcinoma (combined) of the oral mucosa or tongue of 516 mg SDD/L male and female rats were significantly higher than those in the controls. Exposure concentration–related non-neoplastic liver lesions were observed in males and females exposed to 57.3 mg/L and above. These included histiocytic cellular infiltration, chronic inflammation, fatty change and clear cell focus (females), and basophilic focus (males). Increased incidences of histiocytic cellular infiltration also occurred in the small intestine (duodenum), mesenteric lymph node and pancreatic lymph node of males or females exposed to 57.3 mg/L and above.
In the mouse study, groups of 50 male B6C3F1 mice received 0, 14.3, 28.6, 85.7 or 257.4 mg SDD/L drinking water for 2 years (equivalent to 0, 5, 10, 30 and 90 mg Cr(VI)/L or 0, 0.4, 0.9, 2.4 and 5.9 mg Cr(VI)/kg bw per day, respectively). Groups of 50 female mice received 0, 14.3, 57.3, 172 or 516 mg SDD/L drinking water for 2 years (equivalent to 0, 5, 20, 60 and 180 mg Cr(VI)/L or 0, 0.4, 1.4, 3.1 and 8.7 mg Cr(VI)/kg bw per day, respectively; NTP, 2008; Stout et al., 2009). Survival of exposed groups was similar to that of the control groups. The mean body weight of 172 mg/L females was 8% less than that of the controls, and the mean body weight of 516 mg/L females was 15% less than that of the controls, partly attributed to poor palatability of the dosed water. The mean body weights of the 257.4 mg/L males was only slightly less than that of the control group. The incidences of neoplasms of the small intestine (duodenum, jejunum or ileum) were increased in exposed groups of male and female mice. The incidences of adenoma of the duodenum in 257.4 mg/L males and 172 and 516 mg/L females were significantly higher than those in the controls. The incidence of carcinoma of the duodenum was significantly increased in 516 mg/L females. The incidence of adenoma of the jejunum in 516 mg/L females was significantly increased compared with that in the controls. When the incidences of adenoma and carcinoma were combined for all sites of the small intestine, the incidences were significantly increased in 85.7 and 257.4 mg/L males and in 172 and 516 mg/L females compared with those in the controls. The incidences in 57.3 mg/L females exceeded the historical control ranges for drinking water studies. The incidences of diffuse epithelial hyperplasia were statistically significantly increased in the duodenum of all exposed groups of male and female mice. The incidences of histiocytic cellular infiltration were significantly increased in the duodenum of 85.7 and 257.4 mg/L males and in 172 and 516 mg/L females. In the jejunum, the incidences of diffuse epithelial hyperplasia and histiocytic cellular infiltration were significantly increased in 516 mg/L females. The incidences of histiocytic cellular infiltration of the liver in all exposed groups of females (but not males), of the mesenteric lymph node in all exposed groups of males and females and of the pancreatic lymph node of 85.7 and 257.4 mg/L males and 172 and 516 mg/L females were significantly increased.
Conversely, no variation of tumour yield in skin, lungs, forestomach, glandular stomach or duodenum was observed in mice administered SDD in drinking water for 9 months (5 and 20 mg Cr(VI)/L; De Flora et al., 2008). Perhaps tumour development requires more time. Also, no significant pathological changes in the blood, liver, kidneys or femur were observed in rats after a 1-year exposure to Cr(VI) as potassium dichromate in drinking water (up to 25 mg/L, corresponding to 2.5 mg Cr(VI)/kg bw per day; MacKenzie et al., 1958). An excess of benign and malignant forestomach tumours was seen in female mice administered potassium dichromate in drinking water (500 mg/L, corresponding to 134 mg Cr(VI)/L and to a dose of 9 mg Cr(VI)/kg bw per day) for 880 days. However, these studies presented several limitations (e.g., infection caused early mortality, small number of animals per treatment group and lack of individual animal data) that reduced their reliability (Flegal et al., 2001).
Based on the strength of the NTP studies and the limitations of the other long-term studies, the weight of evidence suggests that there is clear evidence of the carcinogenic activity of Cr(VI) as SDD in rodents. Significant increases in the incidences of oral tumours in rats and small intestinal tumours in mice were seen at doses of ≥ 2.1 mg/kg bw per day and ≥ 1.4 mg/kg bw per day, respectively. Also in the small intestine, histiocytic cellular infiltration (rat) and diffuse epithelial hyperplasia (mouse) were dose-dependently increased starting at 0.8 and 0.4 mg/kg bw per day, respectively. Non-neoplastic liver lesions were also observed in rat liver at doses of ≥ 0.8 mg/kg bw per day.
9.2.4 Genotoxicity
Trivalent chromium compounds are considered non-genotoxic by IARC (2012). According to a review by De Flora (2000), most of the assays conducted with Cr(III) compounds (361/403 valid studies, i.e., 90%) were negative, and the positive results were obtained at doses 2 or 3 orders of magnitude higher than the doses required to obtain positive results with Cr(VI) compounds. In vivo studies regarding DNA crosslinks, DNA–protein crosslinks, DNA strand breaks, DNA fragmentation and micronuclei generally led to negative results, except for one DNA deletion study at high doses (> 1875 mg Cr(III)/L; Kirpnick-Sobol et al., 2006). In addition, the NTP (2010) found no clear evidence of genotoxicity of Cr(III) as CPM (NTP, 2010).
Many studies have performed detailed reviews of the genotoxicity of Cr(VI). Cr(VI) has been identified to be mutagenic in a large number of in vitro and in vivo studies in a variety of test systems, which has been thoroughly reviewed by several authors (Sedman et al., 2006; McCarroll et al., 2010; Zhitkovich, 2011). However, the genotoxicity data obtained with Cr(VI) compounds must be interpreted with caution, because the genotoxic potential of Cr(VI) is influenced by various factors, including the availability of Cr(VI) to target cells (influenced by toxicokinetic patterns) and the availability of Cr(VI) to DNA (influenced by metabolic patterns) (De Flora, 2000; De Flora et al., 2006, 2008). Indeed, Cr(VI) does not interact directly with DNA; it must first be reduced to Cr(III), as only intracellular Cr(III) can interact with DNA. However, extracellular reduction of Cr(VI) to Cr(III) limits chromium absorption into cells (see Section 8.0). For this reason, the focus of this section is on in vivo studies performed by the oral route.
The results of genotoxicity assays conducted with Cr(VI) compounds in vivo and in vitro were compared by De Flora (2000). The authors found that 1) most of the in vitro assays assessed (384/436 data, i.e., 88%) were positive, whereas only 43% (30/70) of the in vivo assays were positive (De Flora, 2000); and 2) most of these positive in vivo assays were conducted by routes of administration not relevant to human exposure (injection or instillation, thus bypassing important detoxification mechanisms), and all positive results in vivo were rather weak and generated at high doses, with a lack of dose–response relationship when several doses were tested (De Flora, 2000; Thompson et al., 2013). Despite evidence of Cr(VI) genotoxicity in vitro, more recent in vitro studies suggest that at lower Cr(VI) concentrations, oxidative stress and oxidative DNA damage, and not direct DNA reactivity, may be primary drivers of genotoxicity (Thompson et al., 2012a). However, in vitro studies for Cr(VI) demonstrating an absence of genotoxicity do not necessarily indicate a complete absence of effect, as ascorbate levels under standard culture conditions can be insufficient to result in the intracellular Cr(VI) reduction that is required for mutagenicity (Luczak et al., 2015).
The genotoxicity studies conducted by the oral route have shown either a lack of genotoxicity of Cr(VI) or the occurrence of genotoxic effects in distant tissues. Indeed, in the first study investigating the genotoxicity of Cr(VI) in the gastrointestinal tract (De Flora et al., 2008), female SKH-1 mice were administered Cr(VI) as SDD in drinking water (5 and 20 mg Cr(VI)/L) for 9 months. Results indicated that Cr(VI) did not induce DNA damage in the gastrointestinal tract (no DNA–protein crosslinks and no oxidative DNA damage [8‑OHdG] in the forestomach, the glandular stomach or the duodenum); however, ex vivo treatment of intestinal tissue from exposed animals with Cr(VI) resulted in DNA–protein crosslinks and formation of 8-oxodeoxyguanosine (8-oxodG) (De Flora et al., 2008). Similarly, Thompson et al. (2011b, 2012a) did not detect any increases in oxidative DNA damage in oral or duodenal mucosae of rats or mice following 90 days of exposure to ≤ 520 mg SDD/L. In the second drinking water study (3 months of exposure), ingested Cr(VI) was found to be genotoxic (micronuclei) in the villi of the duodenum from 60 mg Cr(VI)/L, but not in the proliferative crypt cells of mice (O’Brien et al., 2013).
In the review of oral genotoxicity studies by Sedman et al. (2006), evidence of genotoxicity was found in the liver, brain, bone marrow and leukocytes—that is, in tissues where no tumours were observed in the 2‑year carcinogenicity study (NTP, 2008). They included chromosomal aberrations in bone marrow of rats and mice (gavage; Bigaliev et al., 1977; Sarkar et al., 1993), DNA–protein crosslinks in rat liver (100–200 mg Cr(VI)/L in drinking water for 3 weeks; Coogan et al., 1991), DNA single-strand breaks in rat liver and brain (gavage; Bagchi et al., 1995a, 1995b, 1997), DNA fragmentation in mouse liver and brain (gavage; Bagchi et al., 2001, 2002) and DNA single- and double-strand breaks in leukocytes of mice (gavage; Dana Devi et al., 2001).These studies were conducted at doses ranging from 0.3 to 25 mg Cr(VI)/kg bw for durations varying from 2 hours to 1 year. Some of the damage observed (e.g., DNA single-strand breaks) was related to oxidative stress and not to a direct interaction of chromium with DNA (Bagchi et al., 1995a, 1995b, 1997). In vivo micronucleus assays in rodents exposed to Cr(VI) via gavage or drinking water were negative with doses ranging from 0.007 to 165 mg Cr(VI)/kg bw for 2–210 days (Shindo et al., 1989; Mirsalis et al., 1996; De Flora et al., 2006; NTP, 2007).
No treatment-related effect on K-ras codon 12GAT mutation frequency (a marker of early intestinal tumour formation) was observed, even at high Cr(VI) doses that were carcinogenic in the 2-year bioassay and that increased crypt proliferation in mice after 7 or 90 days of exposure to 0.3–520 mg SDD/L drinking water (O’Brien et al., 2013); however, these analyses revealed limited evidence for DNA damage responses (Kopec et al., 2012a, 2012b) and genomic responses induced by 520 mg SDD/L drinking water at day 8 were more similar to those induced by non-mutagenic than by mutagenic carcinogenic compounds (Thompson et al., 2012b).
An in vivo investigation in male Big Blue® transgenic F344 rats identified an absence of mutagenicity of Cr(VI) in oral tissues at doses that were previously identified as being tumourigenic to oral tissues (Thompson et al., 2015a). The study was performed following the protocols of the Organisation for Economic Co-operation and Development (OECD) Test Guideline No. 488 (OECD, 2013), with the exception of a single dose group (180 ppm SDD [10–12 mg Cr(VI)/kg bw per day], i.e., the high dose in the NTP [2008] study). Methods were developed to ensure sufficient and consistent removal of gingival/palate (inner gingiva of upper jaw and hard palate) and gingival/buccal (gingiva covering maxillary alveolar process, from incisors to molars) tissues, as well as to ensure sufficient extraction of DNA from the cartilaginous tissues (Young et al., 2015). These methods were identified as being sufficient to identify increased mutant frequency in oral tissues of Big Blue® rats for both direct-acting and metabolism-dependent mutagens (N-ethyl-N-nitrosourea and benzo[a]pyrene, respectively) (Young et al., 2015). The study found similar levels of mutant frequency in both gingival/palate and gingival/buccal tissues in rats exposed to 180 ppm SDD and those exposed to untreated drinking water; however, the study was sufficient to identify markedly increased mutant frequency in a model oral carcinogen (10 ppm 4-NQO, which is thought to act via an oxidative stress mechanism) (Thompson et al., 2015a). These results therefore suggest that oral tumours observed in F344 rats in the NTP (2008) study did not result from mutagenic processes.
9.2.5 Reproductive and developmental toxicity
Although few studies of the effects of Cr(III) on reproductive and developmental effects have been performed, the effects of Cr(VI) have been investigated in primates, rabbits, rats, and mice. Effects on male reproductive toxicity (particularly on spermatogenesis) occurred in all species, at ≥2.1 mg Cr(VI)/kg-bw per day. Effects on reproduction in female mice (placental effects and fetal resorption and loss) were observed only at doses of ≥46 mg Cr(VI)/kg bw per day, and not in a lower dose multigenerational study.
Oral administration of 3.4 mg Cr(III)/kg bw per day was found to have effects on spermatogenesis in mice (Zahid et al., 1990). Other reproductive and developmental toxicity studies were not relevant, as they involved complexes of dietary supplements that are not anticipated to be present in drinking water (Bailey et al., 2006, 2008; Staniek and Krejpcio, 2009; McAdory et al., 2011).
The effects of Cr(VI) on reproduction were studied in primates (Aruldhas et al., 2004, 2005, 2006; Subramanian et al., 2006) and rodents (Yousef et al., 2006). The lowest LOAEL of 2.1 mg/kg bw per day was reported in male macaca monkeys exposed to potassium dichromate via drinking water for 180 days; the observed effects included histopathological changes of epididymis, decreased testis weight and decreased (by 25%) sperm count and motility (Aruldhas et al., 2006). A similar LOAEL (2.6 mg/kg bw per day) was found in rabbits administered potassium dichromate by gavage for 10 weeks, for decreased plasma testosterone (by 20.8%), sperm count (by 18%) and total mobile sperm (by 34%) and increased proportion (by 24%) of dead sperm (Yousef et al., 2006). Subramanian et al. (2006) concluded that Cr(VI)’s toxic effects on male reproduction may be reversible and prevented by antioxidant vitamin supplementation.
The LOAELs were higher in male rats than in mice, ranging from 20 to 45 mg/kg bw per day compared with 6.4 mg/kg bw per day (for decreased spermatogenesis) (Zahid et al., 1990; U.S. EPA, 2010b). In mice, the LOAELs were higher in females than in males, with values ranging from 46 to 120 mg/kg bw per day for various effects such as increased fetal resorption, increased pre- and post-implantation loss, decreased placental weight and decreased number of follicles at different stages of maturation (Trivedi et al., 1989; Junaid et al., 1996; Murthy et al., 1996; NTP, 2007). In the NTP (1997) multigenerational study conducted in mice, doses up to 36.7 mg Cr(VI)/kg bw per day as potassium dichromate in the diet did not generate reproductive effects. Another study conducted in rats administered potassium dichromate in drinking water (700 mg/L) from gestation day 14 until 14 days after delivery revealed that repeated exposure to a high dose of Cr(VI) (245 mg/L or 9.2 mg/kg bw per day) led to a decrease in body weight (by 25%), delayed bone growth and altered antioxidant system in the bones of offspring (Soudani et al., 2011b).
Lactational exposure to Cr(VI) was found to impair ovarian development, steroidogenesis and pituitary hormone synthesis in developing rats. The pups of lactating Wistar rats exposed to potassium dichromate at a concentration of 200 mg/L via drinking water during postnatal days (PNDs) 1–21 experienced decreased steroidogenesis, growth hormone and prolactin, an increase in follicle stimulating hormone, delayed puberty, decreased follicles and an extended estrous cycle (Banu et al., 2008). Similarly, exposure of lactating rats to 50 or 200 mg Cr(VI)/L on PNDs 1–21 led to delayed puberty and altered levels of steroids and gonadotrophin associated with an induction of oxidative stress caused by a decrease in antioxidant enzyme activities (Samuel et al., 2011). However, the results of steroid and pituitary hormones from Banu et al. (2008; Table 2) and Samuel et al., (2011; Table 3) should be considered with caution, as the data for PND45 and PND65 are repeated in both studies, with some discrepancy between the two studies—although exposure in Banu et al. (2008) was stated to be 200 ppm, the results for two of the hormones (luteinizing hormone and follicle stimulating hormone) were presented as 50 ppm in Samuel et al. (2011).
Rat dams (n=12, eight pups per litter) receiving potassium dichromate in drinking water at a dose of 67 mg/kg bw from gestation day 14 until PND 14 experienced hepatotoxicity, as did their progeny (Soudani et al., 2013).
9.3 Mode of action
A review of the health effects of ingested chromium identified Cr(VI) as the toxic form and the small intestine as the most sensitive tissue. Small intestinal tumours were the most sensitive chronic carcinogenic endpoint (observed at doses of ≥ 1.4 mg Cr(VI)/kg bw per day in mice; NTP, 2008). The most sensitive non-neoplastic chronic effects were also in the small intestine, histiocytic cellular infiltration in the rat and diffuse epithelial hyperplasia in the mouse, starting at 0.8 and 0.4 mg Cr(VI)/kg bw per day, respectively (NTP, 2008). Intestinal tumour development is likely related to the occurrence of previous changes in the small intestine. Although oral mucosal tumours occurred in rats, they occurred at higher doses than mouse intestinal tumours (2.1 mg/kg bw per day) and are unlikely to be relevant to humans at low concentrations. Hence, we present the full mode of action (MOA) analysis for small intestinal tumours and evidence for lack of relevance of oral tumours. The weight of evidence points to the occurrence of a threshold MOA for Cr(VI) carcinogenesis. However, it should be noted that debates are still on-going on the MOA and appropriate model for chromium; current information relies primarily on work conducted by a single group of researchers (Thompson et al., 2013)
9.3.1 Cytotoxic MOA for intestinal tumours in mice
A MOA analysis was conducted for intestinal carcinogenesis (Thompson et al., 2013) based on an established MOA framework (Meek et al., 2003; Boobis et al., 2006). Guidance on the protocol for the series of studies investigating the MOA and review of the final MOA by seven risk assessments experts with expertise in MOA analysis was provided by a science advisory board convened by an independent group (TERA, 2009). The overall weight of evidence supports a cytotoxic MOA for intestinal tumours in mice, with the following four key events: 1) absorption of Cr(VI) from the intestinal lumen; 2) villous cytotoxicity; 3) sustained compensatory crypt hyperplasia to repair/replace the damaged intestinal mucosa; and 4) mutagenesis within crypt cells, ultimately leading to tumorigenesis. The mode of action analysis was also peer reviewed prior to publication by seven experts (TERA, 2012); although one reviewer recommended consideration of a mixed MOA (i.e., including some role for mutagenicity within the existing MOA), the reviewers generally identified the presented data and analyses supported the authors’ conclusion and did not reasonably support alternative MOAs. However,
A summary of the MOA analysis by Thompson et al. (2013) is provided below:
Key event 1: Absorption of Cr(VI) from the intestinal lumen. Under physiological conditions, Cr(VI) exists primarily as anions that are structurally similar to sulphate and phosphate and therefore enter intestinal cells through anion transporters. Cr(III) is not structurally similar to anions, and therefore its entry into cells is limited to passive diffusion. Extracellular reduction of Cr(VI) to Cr(III) prevents absorption through these transporters, thus limiting toxicity. Although the reduction stoichiometry is not fully known, the reduction capacity is known to vary depending on stomach conditions (pH, reducing agents and fed versus fasted). Cr(VI) escaping reduction in the stomach transits further along the gastrointestinal tract lumen, and is either absorbed into mucosa of the small intestine or excreted unabsorbed. In vivo and ex vivo pharmacokinetic data collected in the alimentary tract of mice provide evidence that the carcinogenic doses in the NTP drinking water study decrease the reductive capacity of the mouse gastric contents, resulting in higher chromium concentrations in the duodenum and the jejunum. Consistent with these data, tissue damage and tumour formation are increased mostly in the duodenum and slightly in the jejunum and are not increased in the ileum or large intestine. After 90 days of exposure to Cr(VI) in drinking water, the mouse PBPK model (see Section 8.5 for details) predicts that Cr(VI) exposure produces a substantial decrease in the reducing equivalents (lumped concentration of reducing agents) present in the gastrointestinal lumen at the three highest doses that were carcinogenic in the 2-year NTP study, resulting in increased duodenal chromium concentrations. Additional evidence of chromium uptake into the intestine is provided by toxicogenomics. The number of gene changes in the mouse small intestine after 90 days of exposure to chromium was correlated with tissue dose and histopathological evidence. Although this decrease in Cr(IV) reduction has been posited as a complete depletion in reduction capacity (Kirman et al., 2012, 2013; Thompson et al., 2013), other authors have provided evidence to indicate that depletion in this capacity is not complete. This includes a supralinear trend in liver and kidney concentrations in mice exposed at the doses in the subchronic NTP (2007) study (Stern, 2010) and in dose–response in the chronic NTP (2008) study (Collins et al., 2010). Moreover, improved PBPK model data fits were observed when Cr(VI) reduction capacity is represented by several different pools of reducing compounds, including more slowly depleted pools (Schlosser and Sasso, 2014; Sasso and Schlosser, 2015). Some Cr(VI) also appears to escape reduction even at lower doses (Sedman et al., 2006; Stern, 2010; Zhitkovich, 2011; McLean et al., 2012). Nevertheless, even if Cr(VI) reduction does not become depleted at higher doses, it does clearly decrease with increasing doses.
Key event 2: Villous cytotoxicity. The weight of evidence indicates that Cr(VI) induces toxicity in the non-proliferating, non-pluripotent cells of the intestinal villi. The non-neoplastic lesions observed in the mouse small intestine following Cr(VI) exposure were characterized by NTP (2007, 2008) pathologists as being secondary to previous epithelial cell injury. Histological evidence of cytotoxicity (cytoplasmic vacuolization) in the duodenal villi occurred at lower doses than those associated with atrophy of the villi and crypt hyperplasia, suggesting a mechanism whereby toxicity originates at the point of contact (i.e., villi) and subsequently triggers compensatory cell proliferation of crypt enterocytes. Further, the absence of aberrant nuclei in the crypt and the presence of a small number of aberrant nuclei in villi at the highest Cr(VI) exposure imply that the latter were likely a manifestation of a dose-dependent increase in villous cytotoxicity and not the result of direct genotoxicity or cytotoxicity in the crypt. In vivo evidence suggests that oxidative stress with adequate tissue doses of Cr(VI) likely contributes to cytotoxicity in the intestinal villi. Cr(VI) significantly decreased the ratio of GSH to glutathione disulphide (GSSG), a key indicator of cellular redox status, in the mouse small intestine in a time- and dose-dependent manner. Toxicogenomic analyses also reported increased expression of genes involved in oxidative stress signalling (at the lowest doses after 90 days of Cr(VI) exposure). These data indicate the occurrence of oxidative stress in the villous cells at low doses, even if no oxidative DNA damage (8-OHdG) could be detected at higher doses. Similar results (elevated markers of oxidative stress in the small intestine from 21 mg Cr(VI)/L and lack of oxidative DNA damage) were also obtained in rats. More recent studies provide data that further support this key event. After exposure to tumour-relevant Cr(VI) concentrations (180 mg Cr(VI)/L) for 13 weeks, mice exhibited blunted villi and elongated crypts (which were also increased in number) in the duodenum (Thompson et al., 2015b, 2015c). Moreover, increase in chromium concentrations and a marker of DNA damage (phosphorylated H2AX) was observed in the villi but not the crypts of mouse duodenum (Thompson et al., 2015b). An absence of micronuclei in duodenal crypts was also observed (Thompson et al., 2015c). Chromium concentrations in villi were observed to form a gradient, with the highest concentrations in villous tips. These data provide support to the argument that damage is occurring to the short-lived villi and not to the pluripotent stem cells of the crypt.
Key event 3: Sustained compensatory crypt hyperplasia to repair/replace the damaged intestinal mucosa. Chronic cell proliferation, a well-known risk factor for carcinogenesis, occurred in the duodenum of mice at all SDD concentrations examined in the 2-year bioassay and in the 90-day NTP study. Evaluations at both day 8 and day 91 of exposure revealed that duodenal crypt cell hyperplasia was present in three of five mice exposed to 520 mg SDD/L; by day 91, crypt hyperplasia was present in almost all animals at 170 mg SDD/L. Notably, focal hyperplasia (or other preneoplastic lesions) was not observed in any animals in the 90-day drinking water studies. The presence of diffuse hyperplasia without focal hyperplasia is consistent with proliferation that is secondary to mucosal injury. Rats did get hyperplasia, but only at a high dose, suggesting that mice and rats are qualitatively similar.
Key event 4: Crypt cell mutagenesis. The weight of evidence supports that Cr(VI) is not acting via a mutagenic MOA, specifically where mutation is an early key event, in the mouse small intestine. In addition to the evidence from Sections 9.1.4 and 9.2.4, absence of cytogenetic damage in the duodenal crypts, observations of early tumours, metastases and mortality in the 2-year NTP bioassay, as well as of preneoplastic (e.g., focal hyperplasia) or neoplastic lesions in any of the 90-day Cr(VI) drinking water studies is consistent with a non-mutagenic carcinogen. In addition, genomic profiles induced by 520 mg SDD/L at day 8 were more similar to those induced by non-mutagenic than by mutagenic carcinogens.
The presence of diffuse hyperplasia with and without tumour formation suggests that Cr(VI)-induced cell proliferation is independent of mutagenesis. Given the sustained pressure for cell proliferation at high doses (hyperplasia occurred from 1 week of exposure at 180 mg Cr(VI)/L and from 60 mg Cr(VI)/L over 3 months of exposure), mutations leading to tumour formation may be spontaneous.
9.3.1.1 Concordance of dose–response and temporal association
As summarized from Thompson et al. (2013), following 90 days of exposure, chromium levels in duodenal tissue significantly increased at ≥ 14 mg SDD/L, and the GSH/GSSG ratio significantly decreased at these concentrations. Cytoplasmic vacuolization and other signs of villous toxicity significantly increased at ≥ 60 mg SDD/L, and crypt cell proliferation significantly increased at ≥ 170 mg SDD/L, ultimately resulting in adenoma formation (which typically precedes carcinoma formation). A similar pattern occurs in the jejunum at day 91 and in the 2-year NTP bioassay, although in the NTP studies, the term ‘‘diffuse hyperplasia’’ represented both villous cytotoxicity and crypt hyperplasia. The incidence of diffuse hyperplasia can be seen to precede the incidence of tumour formation.
Key events that occur at the same time as tumours are not likely to contribute to tumour development (Boobis et al., 2006). In this regard, oxidative stress, villous toxicity and crypt hyperplasia were seen after 7 days of Cr(VI) exposure. After 90 days of exposure, damage to villi and crypt hyperplasia can be seen across multiple doses without tumours or preneoplastic lesions. Diffuse hyperplasia was reported in the 90-day NTP bioassay. After 2 years of exposure, diffuse hyperplasia was observed in all treatment groups (≥ 14 mg SDD/L), and tumours were observed at ≥ 172 mg SDD/L relative to concurrent controls (≥ 57 mg/L relative to historical controls). Thus, mice experienced increased cell proliferation and redox changes beginning within the first week of exposure. The fact that neither preneoplastic lesions nor tumours were observed in the 90-day studies and that tumours did not occur until 450 days or later suggests that it takes the majority of the 2-yearmouse lifespan for these events to contribute to tumour formation. A highly proliferative tissue (like the small intestine) experiencing oxidative stress is expected to provide the ideal environment for an early mutation induced by a mutagenic compound—yet no genetic damage of the crypt cells was observed with Cr(VI)administration.
9.3.1.2 Human relevance and potentially susceptible subpopulations
Evidence suggests that the MOA is relevant to humans, provided that the dose is sufficient (higher than environmentally relevant doses):
Key event 1: It is likely that uptake of Cr(VI) by intestinal cells is relevant to humans, as the process of intestinal absorption is, at least qualitatively, similar in mice and humans, and there is evidence that ingested Cr(VI) is absorbed by the human gastrointestinal tract (see Section 8.1). Stern (2010) estimated that the fraction of Cr(VI) escaping gastric reduction might be lower in humans than in mice, but data currently available (limited ex vivo data in humans) do not allow a quantitative estimation of the actual fraction of Cr(VI) that may reach the small intestine after ingestion of drinking water in both mice and humans. Estimates of total chromium uptake in humans range from 3 to 20% (Kerger et al., 1996, 1997; Zhitkovich, 2011). Estimates of the reductive capacity in the fed and fasted state were approximately 30 and 7 mg/L, respectively (Kirman et al., 2013). PBPK modelling predicts that the fraction of total chromium absorbed is low, approximately 0.01-0.02 (Kirman et al., 2013). Nevertheless, escaping reduction appears to be relevant in humans, as Cr(VI) was found to be absorbed even at very low doses, such as 6.4 ng Cr(VI)) in water administered orally to fasting patients (Donaldson and Barreras, 1966).
Key event 2: It is reasonable to assume that villous cytotoxicity is relevant to humans. Limited epidemiological data suggest a possible link between the consumption of Cr(VI)-contaminated drinking water and abdominal pain and diarrhea (Zhang and Li, 1987). Also, it is likely that the effects of Cr(VI) in the villi (oxidative stress, cytotoxicity) and the response of the villi may be, at least qualitatively, similar in both mice and humans. However, there may be some species-specific changes (e.g., lesions in rats and mice were slightly different).
Key event 3: It is reasonable to assume that crypt cell hyperplasia may be relevant to humans, as the regenerative response to villous cytotoxicity and crypt cell proliferation may be, at least qualitatively, similar in both mice and humans.
Key event 4: It is reasonable to assume that crypt cell mutagenesis may be relevant to humans, as prolonged pressure for an increased rate of crypt cell proliferation preceding an increased rate of spontaneous mutations is unlikely to be qualitatively different between mice and humans.
Several factors influence Cr(VI)’s toxicity, including reduction rate (pH dependent),fed versus fasted conditions, gastric motility and ascorbate availability (Thompson et al., 2013). For example, higher gastric pH (individuals taking proton pump inhibitors and children less than 2 or 3 years of age) might reduce Cr(VI) less efficiently and result in higher Cr(VI) concentrations in the small intestine. In addition, intracellular ascorbate-mediated Cr(VI) reduction may be more deleterious than other forms of Cr(VI) reduction (Reynolds et al., 2012). Whereas rodents can synthesize their own ascorbate, humans acquire ascorbate through their diet (Linster and Van Schaftingen, 2007), thus potentially limiting Cr(VI) toxicity.
9.3.1.3 Alternative carcinogenic MOAs
The alternative MOA for intestinal tumours that has been the greatest focus of discussion—and that presents the greatest implications for dose–response assessment approaches—is direct-acting mutagenicity. An analysis of this MOA was developed by U.S. EPA staff (McCarroll et al., 2010); although this analysis precedes the publication of many of the MOA studies described in the cytotoxicity MOA review (Thompson et al., 2013), the results of the mutagenicity MOA analysis are summarized below to ensure consideration of this much-discussed MOA.
Key event 1: Reduction of Cr(VI) to Cr(III) via interaction of cellular components (DNA) with Cr(VI). The initial step of this key event, cellular absorption, is as described above, with Cr(VI) being transported into the cell due to its structural similarity to phosphate and sulphate ions, and Cr(III) not being readily absorbed. Intracellular Cr(VI) then becomes reduced to Cr(III) by enzymes bound to the endoplasmic reticulum; this reduction process is thought to cause damage to the DNA, either by generation of reactive oxygen species or by direct binding of Cr(III) to DNA. Evidence of oxidative damage in acute oral in vivo studies was provided by increased DNA fragmentation in mouse leukocytes in a Comet assay at ≥0.59 mg/kg, and DNA fragmentation and production of oxidative products in livers and brains of mice at ≥1.9 mg/kg. Cr(III)-DNA adducts have also been noted in the absence of oxidative stress in in vitro studies, with the majority of point mutations as G:C base pair substitutions, a high percentage of which resulted in G:CàT:A transversions. An in vivo study also noted DNA–protein cross-links in rat livers after three weeks of drinking water exposure to both doses (6.1 or 8.7 mg K2CrO4/kg bw per day).
Key event 2: Mutagenesis. Evidence of mutagenesis in in vivo studies includes induction of micronuclei and chromosome aberration, which was observed in mice exposed via i.p. (at 40 mg K2CrO4/kg bw per day) or oral gavage (at 20 mg K2CrO4/kg bw per day), but not in those exposed through drinking water. Although the effects were investigated in liver and bone marrow, uptake and reduction of Cr(VI) could plausibly occur in all cells and organs.
Key event 3: Cell proliferation (hyperplasia). Duodenal hyperplasia was observed at all doses in the 90 day (≥10 mg Cr(VI)/kg bw per day) and 2-year (≥0.4 mg Cr(VI)/kg bw per day) NTP mouse studies. Hyperplasia was not observed in gastrointestinal tissues in rats in the study.
McCarroll et al. (2010) demonstrated dose–response and temporal concordance, with the earlier key events occurring at lower doses and after shorter durations than later key events. The authors also stated that although oxidative DNA damage and DNA-protein crosslinks were not observed in the duodenum of mice exposed to low chromium doses in drinking water, the use of doses and durations of exposure that were tumourigenic in the NTP study might plausibly exhaust cellular reductive capacity and result in DNA damage. An absence of site concordance data was noted by the authors, who stated that the weight of evidence of overwhelming mutagenicity in other cells and tissues overcame this observation. As DNA adducts and micronuclei were observed in tissues of occupationally-exposed individuals, the authors concluded the effects were plausible in humans.
An additional alternative MOA for Cr(VI)-induced intestinal cancer includes a mitogenic MOA. Evidence for a mitogenic MOA is weak, as crypt hyperplasia occurs after villous cytotoxicity in both dose and time (hyperplasia not occurring in the absence of cytotoxicity), and a mitogenic effect would likely lead to elongated crypts without the blunted villi that were seen in the 2-year NTP study (Thompson et al., 2013).
9.3.1.4 Comparative weight of evidence for potentially relevant MOAs
Neither of the two major proposed MOAs for chromium-induced intestinal tumours in rats can be fully confirmed, as data gaps and/or inconsistencies in the data exist for both MOAs. In the absence of complete confirmation of the effect, the available weight of evidence for the two effects can be compared to identify the most likely MOA, based on existing data. This process impacts the dose–response analysis for the endpoint, as linear low-dose extrapolation is warranted if stronger weight of evidence is assumed for the mutagenic MOA, and a non-linear evaluation (i.e. a TDI approach) is warranted if cytotoxicity and regenerative hyperplasia are considered to be the most relevant MOA.
Several key events for both MOAs are similar. Both MOAs require some amount of Cr(VI) escaping reduction to Cr(III) to allow for absorption of Cr(VI) into intestinal cells. Furthermore, both MOAs propose that the observed intestinal hyperplasia is a final key event, whereby clonal expansion of genetic mutations leads to the development of intestinal adenomas and carcinomas. The difference between the two MOAs is whether Cr(IV)-induced gene mutations trigger the clonal expansion (i.e. DNA mutation preceding hyperplasia), or whether the hyperplasia is regenerative and triggered by cytotoxicity, resulting in the propagation of unrepaired spontaneous mutations.
Studies of Cr(VI) appear to clearly indicate that the compound is mutagenic; however, whether mutagenicity is relevant for drinking water conditions—and whether it was a driver of the development of intestinal tumours observed in the NTP (2008) study—is less evident. This MOA was based primarily on mutation data from non-target tissues, in vitro systems and mutations in K-ras/Apc genes that could not be replicated (McCarroll et al., 2010; O’Brien et al., 2013).
Therefore, the major weakness to the mutagenic MOA is the absence of evidence of mutagenicity in relevant tissues, and the absence of site concordance of mutagenicity and tumour development. The absence of observed mutagenesis from drinking water exposures at concentrations relevant to the NTP (2008) study is a particularly large gap. Although micronuclei and chromosomal aberrations were observed in the tissue after exposure via i.p. or oral gavage, these routes of exposure are not necessarily relevant due to the likelihood of an increased proportion of Cr(VI) escaping reduction after a large bolus dose (gavage) or bypassing reduction in the GI tract (i.p.). Studies designed to measure mutagenesis of Cr(VI) in intestinal tissues after drinking water exposure did not identify the adverse effects by this route (Shindo et al., 1989; De Flora et al., 2008; Thompson et al., 2011b, 2012a). Because the small intestine is a highly proliferative tissue, it is likely that evidence of DNA damage and mutation would have been readily apparent if Cr(VI) tumourigenicity were mediated through a direct genotoxic or mutagenic MOA (most in vivo micronucleus studies are conducted in proliferative tissues such as bone marrow, skin and intestine, because proliferation facilitates the detection of genotoxicity) (Thompson et al., 2013). An additional major weakness of the mutagenicity MOA is that the presence of tumours has been restricted to GI tissues, despite evidence of mutagenicity and high levels of Cr absorption in other tissues. DNA damage has been demonstrated to occur in a variety of tissues, including liver and blood cells, subsequent to Cr(VI) exposure; although these studies are primarily via non-drinking water routes of exposure, DNA-protein crosslinks were measured in liver in rats exposed to 100 or 200 ppm for 3 weeks (Coogan et al., 1991). Cr(VI) was also measured to be reaching tissues systemically, as concentrations in mouse and rat liver and kidney after exposure to drinking water were similar to concentrations the glandular stomach (no measurements in intestines) (Collins et al., 2010), and treatment-related effects were observed in the livers of female mice and rats (NTP, 2007; NTP, 2008; Witt et al., 2013).
Moreover, Thompson et al. (2013) believe that the high incidence (60%) of crypt proliferation after only 7 days of Cr(VI) exposure is unlikely to be the result of a fixed mutation—especially considering the lack of neoplasms at day 90, the late tumour onset in the 2-year NTP mouse studies and the crypt hyperplasia in rats after 7 days of Cr(VI) exposure, but no hyperplasia or tumours after 2 years (suggesting that hyperplasia is reversible and not the result of a fixed mutation).
Lastly, Thompson et al. (2013) compared available Cr(VI) data with the key events and characteristics reported for chemicals with a mutagenic MOA (U.S. EPA, 2007; Boobis et al., 2009) and found that Cr(VI) data do not share these key events and characteristics. For example, 1) mutations in genes associated with carcinogenesis and in the presence of low cytotoxicity increase the weight of evidence for a mutagenic MOA; however, no increases in markers of intestinal tumours (K-ras mutation frequency or changes in Apc/Wnt/b-caten in signalling) were detected, even at cytotoxic concentrations; 2) mutagens often elicit tumour responses early in chronic studies (e.g., within 52 weeks), but tumours were observed late in the NTP study (450 days) and did not increase mortality; 3) mutagens produce tumours in many tissues, but Cr(VI) exposure is associated only with tumours in tissues related to the portal of entry (GI tract, including mouth, for oral exposure, and respiratory tract for inhalation exposure); and 4) clonal expansion of mutated cells often increases mutations in other key genes and leads to preneoplastic lesions; thus, even if Cr(VI) did not specifically target K-ras, a general increase in mutations would likely lead to increases in additional mutations in K-ras codon 12, which were measurable, but unchanged by Cr(VI). Further, the lack of preneoplastic lesions suggests that clonal expansion of cells with growth advantages were not present.
In contrast, stronger tissue-relevant dose–response and temporal concordance was observed for the non-mutagenic MOA. Whereas there was no evidence of increases in mutagenic effects in response to increasing concentrations or duration of exposure to Cr(VI), endpoints for the non-mutagenic MOA (villous cytotoxicity, oxidative stress and crypt hyperplasia) were all increased in a dose-dependent manner, and were worse (or more prevalent) at day 91 relative to day 8 (Thompson et al., 2013). The diffuse hyperplasia that was observed was consistent with tissue damage. In addition to the above evidence, Thompson et al. (2013) provide further evidence for the MOA’s plausibility. This includes the recognition that cytotoxicity and subsequent regenerative hyperplasia is a well-known MOA; the MOA has also been relevant for intestinal tumours for other compounds, including captan and folpet (Gordon, 2007; Cohen et al., 2010). Additionally, cytotoxic and proliferative effects observable by 13 weeks of exposure can be predictive of effects in 2-year bioassays.
One gap in the MOA is that no studies identifying the essentiality of early key events (i.e. studies measuring the presence or absence of tumours after one of the early key events, such as cytotoxicity, is prevented). The majority of other data gaps for the non-mutagenic MOA are more of a deficiency in the detailed mechanism of action rather than the MOA needed to inform human health risk assessment. Additional measures 1) to distinguish between Cr(III) and Cr(VI) in biological samples, 2) of oxidative status (in addition to GSH/GSSG), 3) of chromium–DNA adducts, which can differentiate between crypt and villi enterocytes in vivo, and 4) of DNA methylation might provide useful information on chromium toxicity, but would not change the MOA. It is also unclear whether the Cr(VI)-induced tumours are due to expansion of pre-existing initiated cells due to constant proliferative pressure or whether cells become initiated due to new mutations that arise from constant proliferative pressure. Further, the exact Cr(VI) reductive stoichiometry in rodents and humans is unknown. These data gaps, when filled, are not expected to alter the MOA.
9.3.2 Oral cavity neoplasms in rats
Data are insufficient to thoroughly evaluate the MOA for oral tumours observed in F344 rats in the 2-year NTP (2008) study; however, several recent studies have further investigated some aspects of two potential modes of action for the effect—mutagenicity, and alteration of iron homeostasis.
As described in Section 9.2.4, an in vivo study in transgenic F344 rats did not identify mutagenicity in oral tissues at a dose of 10–12 mg Cr(VI)/kg bw per day, which is a tumourigenic dose in the NTP (2008) study, whereas the same protocols were able to identify increased mutant frequency in model mutagens and oral carcinogens (Thompson et al., 2015; Young et al., 2015). These results therefore suggest that oral tumours observed in F344 rats in the NTP (2008) study did not result from mutagenic processes. The duration of the study is shorter than the first observed incidence of oral tumours in rats; however, a 28-day mutagenicity study in transgenic rodents is thought to be of sufficient duration to allow for accumulation of mutations even from weak mutagens or in slowly proliferating tissues (OECD, 2013). Moreover, although a small sample size (n = 6) was used, the number of animals was higher than that considered to be sufficient by OECD (n = 5).
Despite the suggestion that oral tumourigenic doses of Cr(VI) are not mutagenic in rat species, few additional details of non-mutagenic MOAs exist for the oral tumours. The oral tumours were not preceded by non-neoplastic or pre-neoplastic lesions (NTP, 2008); therefore, the progression of the tumours is difficult to ascertain.
A potential MOA for rat oral tumours that has recently been posited is chromium-induced alteration of iron homeostasis (Suh et al., 2014). The MOA has not been fully analyzed; however, supporting evidence has been demonstrated. Cr(IV) exposure might potentially result in iron depletion and anaemia. Cr(VI) is known to oxidize ferrous iron to ferric iron (Fe2+ - Fe3+), and only ferrous iron can be absorbed from the intestinal lumen; therefore, Cr(VI) is hypothesized to interfere with iron absorption (Suh et al., 2014). Intracellular Cr(III) can also competitively bind with ferritin, and extracellular Cr(III) can bind to transferrin, impacting iron homeostasis (Suh et al., 2014). In support of chromium’s effect on iron absorption and homeostasis, microcytic hypochromic anaemia (characterized by dose-related trends in decreased hematocrit values, hemoglobin concentrations, and erythrocyte counts, and suspected by authors to be due to alteration in iron metabolism) was observed in rats (at all doses; ≥ 22 mg Cr(VI)/L), whereas the effect was much less severe in mice in the 90 day NTP (2007) study, as well as in other studies (NTP, 1996a, 1996b, 1997). Haematological effects observed in the 2-year study occurred at ≥ 20 mg Cr(VI)/L and improved with time, but were still observed in rats and mice at ≥ 60 mg Cr(VI)/L at the last time point for haematological evaluation (at one year) (NTP, 2008); however, NTP pathologists believe this was an adaptive response, due to the transient nature of effects (Witt et al., 2013). Moreover, dose-dependent decreases in iron concentrations were observed in serum (significant at ≥7.2 mg Cr(VI)/kg bw per day in rats, and not significantly decreased at any dose in mice), liver (significant at ≥2.9 mg Cr(VI)/kg bw per day in rats and ≥4.6 mg Cr(VI)/kg bw per day in mice), and duodenum (significant at ≥20.5 mg Cr(VI)/kg bw per day in rats and ≥4.6 mg Cr(VI)/kg bw per day in mice) in a 90-day study (Suh et al., 2014). Dose-related effects on the incidence and severity of iron content in bone marrow of rats in the study was observed, with decreases in all rats at 20.5 mg Cr(VI)/kg bw per day, which corresponded with fewer iron-containing bone marrow macrophages (Suh et al., 2014). Moreover, expression of genes involved in iron transport and absorption was altered in mice and rats exposed for 8 or 91 days, with the effects in rats being dose-dependent and stronger than in mice (Suh et al., 2014). Suh et al. (2014) outlined many references that demonstrate oral carcinogenicity from altered iron metabolism, impairment of salivary peroxidase activity in iron-deficient rats, and effects of nutritional status on saliva, salivary glands, oral health, and oral carcinogenesis.
The described MOA for oral tumours based on disruption of iron homeostasis is potentially qualitatively relevant in humans, as certain types of anemia have been associated with oral cancers (as described in Suh et al., 2014). However, as iron depletion only occurs at high concentrations, the MOA might not be relevant to humans exposed to Cr(VI) at low doses in drinking water.
Although more data would be required to perform a thorough MOA analysis for oral tumours, data on the absence of mutagenicity in the oral tissues of rats, accompanied by evidence of impact of Cr(VI) on iron homeostasis (particularly in the rat), a low-dose linear extrapolation does not appear to be relevant for oral tumours.
10.0 Classification and assessment
10.1 Cancer and non-cancer assessment
No definitive evidence of toxicity or carcinogenicity following short-term or chronic oral exposure to Cr(III) has been reported. Indeed, IARC (1990) classified metallic chromium and Cr(III) compounds as “not classifiable as to their carcinogenicity to humans” (Group 3) based on inadequate evidence in both humans and animals. Thus, a guideline based on Cr(III) is not feasible.
In contrast, Cr(VI) compounds were classified as “carcinogenic to humans” (Group 1) based on sufficient evidence for carcinogenicity in humans (lung cancer) and sufficient evidence in experimental animals (IARC, 2012). Although the epidemiological evidence as a whole supports an association between exposure to chromium and an increased risk of cancer, especially for occupational inhalation exposure and lung cancer, significant limitations prevent its use for the derivation of a guideline. Data on human carcinogenicity via the oral route are still lacking, however, there is sufficient carcinogenic evidence in experimental animals on which to base an HBV.
Small intestinal tumours were the most sensitive chronic carcinogenic endpoint (observed at doses of ≥ 1.4 mg Cr(VI)/kg bw per day in mice; NTP, 2008). Two of the most sensitive non-neoplastic chronic effects were also in the small intestine, histiocytic cellular infiltration in the rat and diffuse epithelial hyperplasia in the mouse, starting at 0.8 and 0.4 mg Cr(VI)/kg bw per day, respectively (NTP, 2008). Liver inflammation was also observed in female rats at all doses (i.e. ≥ 0.2 mg Cr(VI)/kg bw per day (NTP, 2008). Although oral mucosal tumours occurred in rats, they occurred at higher doses than mouse intestinal tumours (2.1 mg/kg bw per day) and are unlikely to be relevant to humans at low concentrations. The MOA analysis (Section 9.3) indicates progression from non-cancer to cancer effects as the critical effect after exposure to Cr(VI) in drinking water. Thus, the assessment of Cr(VI) in drinking water focuses on cancer and non-cancer effects of chromium together. The MOA analysis supports hyperplasia as a key precursor event to tumour development and a threshold approach for the risk assessment for ingested Cr(VI).Thus, the HBV for Cr(VI) in drinking water is based on diffuse hyperplasia of the small intestine, as it is the most sensitive endpoint, is a precursor of tumour formation and thus will be protective of both non-cancer and cancer effects.To derive a HBV for Cr(VI), benchmark doses (BMD) and the lower 95% confidence limits on those BMDs (BMDL) were obtained from Thompson et al. (2014). These BMDs/BMDLs were derived using the rodent PBPK model (Section 8.5) to estimate the amount of Cr(VI) entering each intestinal tissue section (duodenum, and jejunum only) from the lumen per day (normalized to intestinal tissue weight) in both sexes of mice. Then, using BMD modelling of the internal doses versus incidences of diffuse hyperplasia, BMD and BMDLs for the benchmark response (BMR) rates of 10%, 5%, and 1% were derived. Although a 10% response rate serves as the default value, the application of a PBPK model to estimate internal dose metrics provides internal dose gradients for each section of the small intestine; the additional dose–response data for each dose resulted in approximately 1500 data points (Thompson et al., 2014), which can support the selection of lower BMRs. A BMR of 1% falls well within the range of observation of the data set, and its selection is strengthened by the similarity between the BMD and BMDL values (i.e. indicating a low degree of uncertainty in the calculated values). Moreover, the selection of a BMR of 1% is considered conservative, and provides an additional degree of health protection for cancer effects. The BMD/BMDL values for Cr(VI) based on mouse small intestine hyperplasia are 4.8/3.8 mg/kg small intestine section–day at a BMR of 10%, 3.2/2.2 mg/kg small intestine section–day at a BMR of 5%, and 1.2/0.67 mg/kg small intestine section–day at a BMR of 1%.
Next, the human PBPK model (Section 8.5) and Health Canada specific parameters were used to convert the animal BMD/BMDLs into human equivalent doses (HED) for Health Canada (Summit Toxicology, 2014). Health Canada parameters for adult exposure (70 kg body weight and 1.5 L/day drinking water consumption) and the fasted exposure scenario (worst-case scenario; Section 8.5) were used. Based on the adult human PBPK predictions, as a function of external dose, the HED fell within the range of doses associated with non-linear toxicokinetics (due to decreases in reducing agent) in humans. The HEDs of 0.14, 0.11, and 0.054 mg Cr(VI)/kg bw per day correspond to the BMDL10, BMDL05, and BMDL01, respectively.
Based on the most conservative HED, the tolerable daily intake (TDI) for Cr(VI) is calculated as follows:
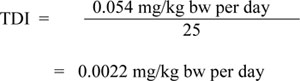
Equation 1 - Text description
The TDI of chromium (VI) is 0.0022 mg/kg body weight per day. This is calculated by dividing the human equivalent dose (HED) of 0.054 mg/kg body weight per day by the uncertainty factor of 25.
where:
- 0.054 mg/kg bw per day is the HED for the lower 95% confidence limit on the benchmark dose for a 1% response (BMDL01) for diffuse epithelial hyperplasia as a precursor to intestinal tumours; and
- 25 represents the uncertainty factor (only the pharmacodynamic component of the interspecies uncertainty factor [×2.5] is used because pharmacokinetic differences between mice and humans were already quantitatively accounted for with the application of the PBPK model; ×10 to account for intraspecies variability). A database uncertainty factor was deemed unnecessary due to the availability of reproductive and developmental toxicity studies—including multigenerational studies—in addition to lifetime bioassays in multiple species; adverse effects from these studies were less sensitive than effects on the small intestine. No uncertainty factor for severity of effect was used, as the TDI is based on the precursor event to cancer.
Using this TDI, the HBV for Cr(VI) in drinking water is derived as follows:
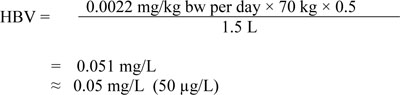
Equation 2 - Text description
The HBV for chromium (VI) in drinking water is 0.05 mg/L (rounded). This is calculated by multiplying the TDI for chromium of 0.0022 mg/kg body weight per day by the average adult body weight of 70 kg, then by 0.5, the allocation factor denoting the contribution of drinking water to the tolerable daily intake, and dividing the resulting product by 1.5 L, the daily average volume of drinking water ingested by an adult.
where:
- 0.0022 mg/kg bw per day is the TDI derived above;
- 70 kg is the average body weight of an adult;
- 0.5 is the allocation factor estimated for drinking water; it refers to the contribution of drinking water to the estimated total daily intake for Canadians (Section 5.7); and
- 1.5 L is the daily average volume of drinking water ingested by an adult.
10.2 International considerations
The assessments for Cr(VI) in drinking water by the World Health Organization’s (WHO) International Programme on Chemical Safety (IPCS, 2013) and California’s Office of Environmental Health Hazard Assessment (CalEPA, 2011) are based on the NTP (2008) study. IPCS (2013) derived a tolerable daily intake (TDI) of 0.9 µg Cr(VI)/kg bw per day based on the lower 95% confidence limit on the benchmark dose for a 10% response (BMDL10) for diffuse epithelial hyperplasia in female mice. However, owing to uncertainties, they did not derive a quantitative assessment of the carcinogenic risk to humans from ingesting Cr(VI). This same value was also derived by ATSDR (2012), using intestinal hyperplasia as a basis. CalEPA (2011) established a public health goal of 0.02 µg Cr(VI)/L based on an oral cancer slope factor of 0.5 (mg/kg bw per day)-1 for tumours of the small intestine in mice (NTP, 2008). CalEPA (2011) also derived a health protective goal of 0.002 mg/L based on liver toxicity in female rats in the NTP (2008) study. The maximum contaminant level for Cr(VI) adopted by the California Environmental Protection Agency in 2014 was 0.01 mg/L (10 µg/L), which was derived based on technological and economic capabilities that allowed the maximum contaminant level to be as close to the public health goal as possible (CalEPA, 2015). The current WHO (1996) provisional guideline value of 0.05 mg/L for total chromium was based on the carcinogenicity of Cr(VI) by the inhalation route, but does not account for new research. The current U.S. EPA maximum contaminant level for total chromium of 0.1 mg/L was established in 1991.
11.0 Rationale for guideline
Small amounts of naturally occurring chromium are released from rocks and soils into water. However, more than 70% of chromium in the environment comes from a wide array of anthropogenic sources. Chromium can exist in trivalent and hexavalent forms in water; Cr(VI) may represent up to 100% of the chromium present in drinking water. Exposure to chromium from drinking water is limited to the ingestion route.
Chromium toxicity in humans varies depending on the form of the compound, its valence state and the route of exposure. Studies show that there is little or no toxicity associated with the trivalent form, whereas IARC has classified Cr(VI) compounds as carcinogenic to humans by the inhalation route of exposure, based on sufficient evidence in both humans and animals. Although data on human carcinogenicity via the oral route are still lacking for Cr(VI), there is sufficient carcinogenic evidence in experimental animals to establish a drinking water guideline for chromium.
The MOA analysis supports a progression from non-cancer to cancer effects after exposure to Cr(VI) in drinking water. Thus, the assessment of chromium in drinking water, which is based on the health effects of Cr(VI), considers the cancer and non-cancer effects together. The critical health effect on which to establish a guideline for chromium in drinking water is diffuse hyperplasia of the small intestine, as it is a precursor of tumour formation in the mode of action with the greatest weight of evidence. The mouse and human PBPK models and BMD modelling were used to determine appropriate external doses in humans from animal data.
A MAC of 0.05 mg/L (50 µg/L) is for total chromium in drinking water. This MAC is achievable by available treatment technology and measurable by available analytical methods, and is protective of both cancer and non-cancer effects. As part of its ongoing guideline review process, Health Canada will continue to monitor new research in this area and recommend any change to the guideline that is deemed necessary.
12.0 References
Aaseth, J., Alexander, J. and Norseth, T. (1982). Uptake of 51Cr-chromate by human erythrocytes—a role of glutathione. Acta Pharmacol. Toxicol., 50(4): 310–315 [cited in O’Flaherty et al., 2001].
Allan, R.J. (1986). The role of particulate matter in the fate of contaminants in aquatic systems. Environment Canada, Burlington, Ontario. 128 pp. (Scientific Series No. 142).
Anderson, R.A. (1981). Nutritional role of chromium. Sci. Total Environ., 17: 13–28.
Anderson, R.A., Bryden, N.A. and Polansky, M.M. (1992). Dietary chromium intake: freely chosen diets, institutional diets, and individual foods. Biol. Trace Elem. Res., 32: 117–121 [cited in Stoecker, 2004].
Anderson, R.A., Bryden, N.A. and Polansky, M.M. (1997). Lack of toxicity of chromium chloride and chromium picolinate in rats. J. Am. Coll. Nutr., 16(3): 273–279 [cited in ATSDR, 2012].
Anger, G., Halstenberg, J., Hochgeschwender, K., Scherhag, C., Korallus, U., Knopf, H., Schmidt, P. and Ohlinger, M. (2005). Chromium compounds. In: Ullman’s encyclopedia of industrial chemistry. John Wiley & Sons, New York, New York. pp. 1–36.
APHA, AWWA and WEF(1992). Standard methods for the examination of water and wastewater. 18th edition. American Public Health Association, American Water Works Association and Water Environment Federation, Washington, DC.
APHA, AWWA and WEF (1995). Standard methods for the examination of water and wastewater. 19th edition. American Public Health Association, American Water Works Association and Water Environment Federation, Washington, DC.
APHA, AWWA and WEF(1998). Standard methods for the examination of water and wastewater.20th edition. American Public Health Association, American Water Works Association and Water Environment Federation, Washington, DC.
APHA, AWWA and WEF(2005). Standard methods for the examination of water and wastewater. 21st edition. American Public Health Association, American Water Works Association and Water Environment Federation, Washington, DC.
APHA, AWWA and WEF(2012). Standard methods for the examination of water and wastewater. 22nd edition. American Public Health Association, American Water Works Association and Water Environment Federation,Washington, DC.
Armienta-Hernandez, M.A. and Rodriguez-Castillo, R. (1995). Environmental exposure to chromium compounds in the valley of Leon, Mexico. Environ. Health Perspect., 103(Suppl. 1): 47–51.
Aruldhas, M.M., Subramanian, S., Sekhar, P., Chandra Hasan, G., Govindarajulu, P. and Akbarsha, M.A. (2004). Microcanalization in the epididymis toovercome ductal obstruction caused by chronic exposure to chromium—a study in the mature bonnet monkey (Macaca radiata Geoffroy).Reproduction, 128: 127–137 [cited in ATSDR, 2012].
Aruldhas, M.M., Subramanian, S., Sekar, P., Vengatesh, G., Chandrahasan, G., Govindarajulu, P. and Akbarsha, M. A. (2005). Chronic chromium exposure-induced changes in testicular histoarchitecture are associated with oxidative stress: study in a non-human primate (Macaca radiata Geoffroy). Hum. Reprod., 20(10): 2801–2813.
Aruldhas, M.M., Subramanian, S., Sekhar, P., Vengatesh, G., Govindarajulu, P. and Akbarsha, M.A. (2006). In vivo spermatotoxic effect of chromium as reflected in the epididymal epithelial principal cells, basal cells, and intraepithelial macrophages of a nonhuman primate (Macaca radiata Geoffroy). Fertil. Steril., 86 (Suppl. 3) [cited in ATSDR, 2012].
ATSDR (2012). Toxicological profile for chromium. Agency for Toxic Substances and Disease Registry,U.S. Department of Health and Human Services, Atlanta, Georgia. Available at:www.atsdr.cdc.gov/toxprofiles/tp.asp?id=62&tid=17
Bagchi, D., Hassoun, E.A., Bagchi, M., Muldoon, D.F. and Stohs, S.J. (1995a). Oxidative stress induced by chronic administration of sodium dichromate. Comp. Biochem. Physiol. C, 110: 281–287 [cited in Sedman et al., 2006].
Bagchi, D., Hassoun, E.A., Bagchi, M. and Stohs, S.J. (1995b). Chromium-induced excretion of urinary lipid metabolites, DNA damage, nitric oxide production, and generation of reactive oxygen species in Sprague-Dawley rats. Comp. Biochem. Physiol. C, 110: 177–187 [cited in Sedman et al., 2006].
Bagchi, D., Vuchetich, P.J., Bagchi, M., Hassoun, E.A., Tran, M.X., Tang, L. and Stohs, S.J. (1997). Induction of oxidative stress by chronic administration of sodium dichromate [chromium VI] and cadmium chloride [cadmium II] to rats. Free Radic. Biol. Med., 22(3): 471–478.
Bagchi, D., Bagchi, M. and Stohs, S.J. (2001). Chromium (VI)–induced oxidative stress, apoptotic cell death and modulation of p53 tumor suppressor gene. Mol. Cell. Biochem., 222(1–2): 149–158 [cited in Sedman et al., 2006].
Bagchi, D., Stohs, S.J., Downs, B.W., Bagchi, M. and Preuss, H.G. (2002). Cytotoxicity and oxidative mechanisms of different forms of chromium. Toxicology, 180(1): 5–22 [cited in Sedman et al., 2006]. Comment in: Toxicology, 2003, 186(1–2):171–173; author reply:Toxicology, 2003, 186(1–2):175–177.
Bailey, M.M., Boohaker, J.G., Sawyer, R.D., Behling, J.E., Rasco, J.F., Jernigan, J.J., Hood, R.D. and Vincent, J.B. (2006). Exposure of pregnant mice to chromium picolinate results in skeletal defects in their offspring. Birth Defects Res. B Dev. Reprod. Toxicol., 77(3): 244–249.
Bailey, M.M., Boohaker, J.G., Jernigan, P.L., Townsend, M.B., Sturdivant, J., Rasco, J.F., Vincent, J.B. and Hood, R.D. (2008). Effects of pre- and postnatal exposure to chromium picolinate or picolinic acid on neurological development in CD-1 mice. Biol. Trace Elem. Res., 124(1): 70–82.
Balistrieri, L.S., Murray, J.W. and Paul,B. (1992). The biogeochemical cycling of trace metals in the water column of Lake Sammamish, Washington: response to seasonally anoxic conditions. Limnol. Oceanogr., 37: 529–548.
Balk, E.M., Tatsion, I.A., Lichtenstein, A.H., Lau, J. and Pittas, A.G. (2007). Effect of chromium supplementation on glucose metabolism and lipids: a systematic review of randomized controlled trials. Diabetes Care, 30(8): 2154–2163.
Ball, J.W. and McCleskey, R.B. (2003). A new cation-exchange method for accurate field speciation of hexavalent chromium. Talanta, 61(3):305–313.
Bamwoya, L.A., Rutherford, P.L., Henninger, A.A. and Home, W.H. (1991). Toxic contaminants in soils and sediments at four wood preservation facilities in Atlantic Canada. Environment Canada, Environmental Protection, Conservation and Protection, Dartmouth, Nova Scotia. 52 pp.(EPS-5-AR-91-2).
Banu, S.K., Samuel, J.B., Arosh, J.A., Burghardt, R.C. and Aruldhas, M.M. (2008). Lactational exposure to hexavalent chromium delays puberty by impairing ovarian development, steroidogenesis and pituitary hormone synthesis in developing Wistar rats. Toxicol. Appl. Pharmacol., 232(2): 180–189.
Baresic, M., Gornik, I., Radonic, R., Zlopasa, O., Gubarev, N. and Gasparovic, V. (2009). Survival after severe acute chromic acid poisoning complicated with renal and liver failure. Intern. Med., 48(9): 711–715.
Barikbin, B., Mortazavi, S.B. and Moussavi, G. (2011). Removal of hexavalent chromium from brackish groundwater by nanofiltration: a case study in Iran. J. Water Supply Res. Technol.Aqua, 60(2): 121–126.
Barnowski, C., Jakubowski, N.,Stuewer, D. and Broekaert, J. (1997). Speciation of chromium by direct coupling of ion exchange chromatography with inductively coupled plasma mass spectrometry. J. Anal. Atom. Spectrom., 12(10):1155–1161.
Bartlett, R.J. and James, R.B. (1988). Mobility and bioavailability of chromium in soils. In: Nriagu, J.O. and Nieboer, E. (eds.),Chromium in the natural and human environments. John Wiley and Sons, New York, NY. pp. 267–304.
Bataineh, H., Al-Hamood, M.H., Elbetieha, A. and Bani Hani, I. (1997). Effect of long-term ingestion of chromium compounds on aggression, sex behavior and fertility in adult male rat. Drug Chem. Toxicol., 20(3): 133–149 [cited in ATSDR, 2012].
B.C. Ministry of Environment (2005). Technical guidance on contaminated sites 17—Soil quality database. Environmental Management Branch. Available at: www.env.gov.bc.ca/epd/remediation/guidance/index.htm#1
B.C. Ministry of Health (2010). Personal communication with B. Boettger, Office of the Provincial Health Officer.
Beaubien, S. (1993). Chromium in Lake Ontario: speciation and geochemical cycling. University of Waterloo, Waterloo, Ontario.
Beaumont, J.J., Sedman, R.M., Reynolds, S.D., Sherman, C.D., Li, L.-H., Howd, R.A., Sandy, M.S., Zeise, L. and Alexeeff, G.V. (2008). Cancer mortality in a Chinese population exposed to hexavalent chromium in drinking water. Epidemiology, 19(1): 12–23.
Bednar, C.M. and Kies, C. (1991). Inorganic contaminants in drinking water correlated with disease occurrence in Nebraska. J. Am. Water Resources Assoc., 27: 631–635.
Benova, D., Hadjidekova, V., Hristova, R., Nikolova, T., Boulanova, M., Georgieva, I., Grigorova, M., Popov, T., Panev, T., Georgieva, R., Natarajan, A.T., Darroudi, F. and Nilsson, R. (2002). Cytogenetic effects of hexavalent chromium in Bulgarian chromium platers. Mutat. Res., 514(1–2): 29–38. Comment in: Mutat. Res., 2003, 538(1–2):181–1812; author reply: Mutat. Res., 2003, 538(1–2):183–184.
Bianchi, V. and Levis, A.G. (1988). Review of genetic effects and mechanisms of action of chromium compounds. Sci. Total Environ., 71(3): 351–355.
Bigaliev, A., Turebaev, M. and Elemesova, M. (1977). [Cytogenetic study of the in vivo mutagenic properties of chromium compounds.] In: Dubinin, N.P. (ed.),Genet Posledstviya Zagryanenia Okruzhayushehei Sredy (Proceedings of the Symposium). Nauka, Moscow. pp. 173–176 (in Russian) [cited in Sedman et al., 2006].
Blasiak, J. and Kowalik, J. (2000). A comparison of the in vitro genotoxicity of tri- and hexavalent chromium. Mutat. Res., 469(1): 135–145.
Blute, N. (2011). Cr(VI) treatment options and costs. Water Research Foundation Technology Transfer Workshop on Hexavalent Chromium, August 18, 2011, Sacramento, CA. Water Research Foundation, Denver, CO.
Blute, N., McGuire, M., Qin, G., Brabander, D, Newille, M. and Kavounas,P. (2007). State of art geochemical techniques in evaluating drinking water treatment contaminant removal processes. In: Proceedings of the American Water Works Association Water Quality Technology Conference, Charlotte, North Carolina.American Water Works Association, Denver, Colorado.
Blute, N., Wu, X., Alspach, B., Seidel, C., Brandhuber, P. and Najm, I. (2012). Guidelines for hexavalent chromium treatment testing. Water Research Foundation, Denver, Colorado (Web Report No. 4418). Available at: www.waterrf.org/PublicReportLibrary/4418.pdf
Blute, N., Wu, X., Porter, K. and Imamura, G. (2013a).Hexavalent chromium removal research. Project report to the California Department of Public Health. Research managed by Department of Water and Power,City of Glendale, CA. Report prepared by Hazen and Sewer, Arcadis U.S./Malcolm Pirnie.
Blute, N., Wu, X., Porter, K. and Imamura, G. (2013b). Appendix G. In: Hexavalent chromium removal research. Project report to the California Department of Public Health. Research managed by Department of Water and Power,City of Glendale, CA. Report prepared by Hazen and Sewer, Arcadis U.S./Malcolm Pirnie.
Blute, N., Wu, X., Porter, K. and Imamura, G. (2013c). Appendix H1. In: Hexavalent chromium removal research. Project report to the California Department of Public Health. Research managed by Department of Water and Power,City of Glendale, CA. Report prepared by Hazen and Sewer, Arcadis U.S./Malcolm Pirnie.
Blute, N., Wu, X., Cron, C., Fong, L., Froelich, D. and Abueg, R. (2013d). Microfiltration in the RCF process for hexavalent chromium removal from drinking water. Water Research Foundation, Denver, CO (Report No.4365).
Blute, N.B., Wu, X., Cron, C., Abueg, R., Froelich, D., and Fong, L. (2014). Hexavalent chromium treatment implementation in Glendale, Calif. J. Am. Water Works Assoc., 106 (3): E160-E175.
Blute, N., Wu, X., Imamura, G., Song, Y., Porter, K. Cron, C., Fong, L Froelich, D., Abueg, R., Henrie, T., Ramesh, S. and Vallejo, F. (2015). Assessment of single-pass ion exchange and adsorptive media for hexavalent chromium removal from drinking water. Water Research Foundation. Denver, Colorado. (Project #4423). Available at: www.waterrf.org/Pages/Index.aspx
Bonde, J,P, and Ernst, E. (1992). Sex hormones and semen quality in welders exposed to hexavalent chromium. Hum. Exp. Toxicol. 11 (4):259–263.
Boobis, A.R., Cohen, S.M., Dellarco, V., McGregor, D., Meek, M.E., Vickers, C., Willcocks, D. and Farland, W. (2006). IPCS framework for analyzing the relevance of a cancer mode of action for humans. Crit. Rev. Toxicol., 36(10): 781–792.
Boobis, A.R., Daston, G.P., Preston, R.J. and Olin, S.S. (2009). Application of key events analysis to chemical carcinogens and noncarcinogens. Crit. Rev. Food Sci. Nutr., 49(8): 690–707.
Brandhuber, P., Frey, M., McGuire, M., Chao, P.F., Seidel, C., Amy, G., Yoon, J., McNeill, L. and Banerjee, K. (2004). Low-level hexavalent chromium treatment options: bench-scale evaluation. American Water Works Association Research Foundation, Denver, CO.
Brandhuber, P., McNeill, L., McLean, J., Rogers, N. and Bukhari, Z. (2017). Sources, chemistry, fate, and transport of chromium in drinking water treatment plants and distribution systems. Water Research Foundation. Denver, Colorado. (Project #4497). Available at: www.waterrf.org/Pages/Index.aspx
Broadhurst, C.L. and Domenico, P. (2006). Clinical studies on chromium picolinate supplementation in diabetes mellitus—a review. Diabetes Technol. Ther., 8(6): 677–687.
Buerge, I.J. and Hug, S. (1997). Kinetics and pH dependence of chromium (III) reduction by iron (II). Environ. Sci. Technol., 31(5): 1426–1432.
Buerge, I.J. and Hug, S. (1999). Influence of mineral surface on chromium (VI) reduction by iron (II). Environ. Sci. Technol., 33(23): 4285–4291.
Buttner, B. and Beyersmann, D. (1985). Modification of the erythrocyte anion carrier by chromate. Xenobiotica, 15(8–9): 735–741.
CalEPA (2011). Public health goals for chemicals in drinking water—hexavalent chromium (CrVI). Pesticide and Environmental Toxicology Branch, Office of Environmental Health Hazard Assessment, California Environmental Protection Agency, Sacramento, California. 155pp. Available at: http://oehha.ca.gov/water/phg/pdf/Cr6PHG072911.pdf
CalEPA (2015). Chromium-6 drinking water MCL. California Environmental Protection Agency, Sacramento, California. Available at: www.waterboards.ca.gov/drinking_water/certlic/drinkingwater/Chromium6.shtml
Campbell, J.A. and Yeats, P.A. (1984). Dissolved chromium in the St. Lawrence estuary. Estuar. Coast. Shelf Sci., 19: 513–522.
CARB (2003). California portable classrooms study project executive summary. Final report, Vol. III. RTI International, Research Triangle Park, NC; California Air Resources Board, Sacramento, CA; and California Department of Health Services, Berkeley, CA. Availablr at: www.arb.ca.gov/research/indoor/pcs/pcs-fr/pcs_v3_es_03-23-04.pdf
Cary, E.E. (1982). Chromium in air, soil and natural waters. In: Langard, S. (ed.), Topics in environmental health 5: Biological and environmental aspects of chromium. Elsevier Biomedical Press, New York, New York. pp. 49–64 [cited in ATSDR, 2012].
CCME (2006). A Protocol for the derivation of environmental and human health soil quality guidelines. Canadian Council of Ministers of the Environment.
CDPH (2013). Notice of proposed rulemaking. Title 22, California Code of Regulations. Subject: hexavalent chromium MCL (DPH-11-005). California Department of Public Health. Available at: www.cdph.ca.gov/services/DPOPP/regs/Documents/DPH-11-005HCPublicNotice.pdf
Cevaal, J.N., Suratt, W.B. and Burke, J.E. (1995). Nitrate removal and water quality improvements with reverse osmosis for Brighton, Colorado. Desalination, 103: 101–111.
Chiang, C.T., Chang, T.K., Hwang, Y.H., Su, C.C., Tsai, K.Y., Yuan, T.H. and Lian,leB. (2011). A critical exploration of blood and environmental chromium concentration among oral cancer patients in an oral cancer prevalent area of Taiwan. Environ. Geochem. Health, 33(5): 469–476.
Choinière, J. and Beaumier, M. (1997). Bruits de fond géochimiques pour différents environnements géologiques au Québec. Ministère des Ressources Naturelles, Service des minéraux industriels et de l’assistance à l’exploration. pp. 1–28.
Chowdhury, A.R. (1995). Spermatogenic and steroidogenic impairment after chromium treatment in rats. Indian J. Exp. Biol., 33: 480–484.
Chowdhury, Z., Bigley, S., Gonzalez, W., Porter, K., Francis, C., Imamura, G., Blute, N.K., Rhoades, J., Westerhoff, P. and Bowen, A. (2014). Evaluation of technologies for hexavalent chromium removal. Water Research Foundation. Denver, Colorado. Project #4516 (In progress).
Clifford, D. and Chau, J. (1988). The fate of chromium (III) in chlorinated water. Water Engineering Research Laboratories, U.S. Environmental Protection Agency, Cincinnati, Ohio (EPA/600/S2-87/100).
Clifford, D.A (1999). Ion exchange and inorganic adsorption. Chapter 9 in: Letterman,R.D. (ed.), Water quality and treatment: a handbook of community water supplies.5th edition. American Water Works Association, Denver, CO; McGraw-Hill, New York, New York.
Cohen, S.R., Davis, D.M. and Kramkowski, R.S. (1974). Clinical manifestations of chromic acid toxicity: nasal lesions in electroplate workers. Cutis, 13: 558–568.
Cohen, S.M., Gordon, E.B., Singh, P., Arce, G.T. and Nyska, A. (2010). Carcinogenic mode of action of folpet in mice and evaluation of its relevance to humans.
Cole, P. and Rodu, B. (2005). Epidemiologic studies of chrome and cancer mortality: a series of meta-analyses. Regul. Toxicol. Pharmacol., 43(3): 225–231.
Collins, B.J., Stout, M.D., Levine, K.E., Kissling, G.E., Melnick, R.L., Fennell, T.R., Walden, R., Abdo, K., Pritchard, J.B., Fernando, R.A., Burka, L.T. and Hooth, M.J. (2010). Exposure to hexavalent chromium resulted in significantly higher tissue chromium burden compared with trivalent chromium following similar oral doses to male F344/N rats and female B6C3F1 mice. Toxicol. Sci., 118(2): 368–379. Comment in: Toxicol. Sci., 2011, 119(2):423–424; author reply:Toxicol. Sci., 2011, 119(2):425.
Connett, P. and Wetterhahn, K.E. (1983). Metabolism of the carcinogen chromate by cellular constituents. Struct. Bonding, 54: 93–124.
Coogan, T.P., Motz, J., Snyder, C.A., Squibb, K.S. and Costa, M. (1991). Differential DNA–protein crosslinking in lymphocytes and liver following chronic drinking water exposure of rats to potassium chromate. Toxicol. Appl. Pharmacol., 109: 60–72.
Copat, C., Bella, F., Castaing, M., Fallico, R., Sciacca, S. and Ferrante, M. (2012). Heavy metals concentrations in fish from Sicily (Mediterranean Sea) and evaluation of possible health risks to consumers. Bull. Environ. Contam. Toxicol., 88(1): 78–83.
Corbett, G.E., Dodge, D.G., O’Flaherty, E.J., Liang, J., Throop, L., Finley, B.L. and Kerger, B.D. (1998). In vitro reduction kinetics of hexavalent chromium in human blood. Environ. Res., 78(1): 7–11 [cited in O’Flaherty et al., 2001].
Costa, M. and Klein, C.B. (2006). Toxicity and carcinogenicity of chromium compounds in humans. Crit. Rev. Toxicol., 36(2): 155–163.
Craun, G.F. and McCabe, L.J. (1975). Problems associated with metals in drinking water.J. Am. Water Works Assoc.,67(11): 593–599.
Crump, C., Crump, K., Hack, E., Luippold, R., Mundt, K., Liebig, E., Panko, J., Paustenbach, D. and Proctor, D. (2003). Dose–response and risk assessment of airborne hexavalent chromium and lung cancer mortality. Risk Anal., 23(6): 1147–1163.
Dana Devi, K., Rozati, R., Saleha Banu, B., Jamil, K. and Grover, P. (2001). In vivo genotoxic effect of potassium dichromate in mice leukocytes using comet assay. Food Chem. Toxicol., 39(8): 859–865.
Dann, T. (1991). Measurement program for toxic contaminants in Canadian urban cities. Pollution Division, Environment Canada, Ottawa, Ontario (PMD 91-2).
Dann, T. (2007). Personal communication. November.
Davies, J.M., Easton, D.F. and Bidstrup, P.L. (1991). Mortality from respiratory cancer and other causes in United Kingdom chromate production workers. Br J Ind Med, 48: 299–313..
De Flora, S. (2000). Threshold mechanisms and site specificity in chromium(VI) carcinogenesis. Carcinogenesis, 21(4): 533–541.
De Flora, S., Bagnasco, M., Serra, D. and Zanacchi, P. (1990). Genotoxicity of chromium compounds. A review. Mutat. Res., 238: 99–172 [cited in Environment Canada and Health Canada, 1994].
De Flora, S., Camoirano, A., Bagnasco, M., Bennicelli, C., Corbett, G.E. and Kerger, B.D. (1997). Estimates of the chromium(VI) reducing capacity in human body compartments as a mechanism for attenuating its potential toxicity and carcinogenicity. Carcinogenesis, 18(3): 531–537.
De Flora, S., Iltcheva, M. and Balansky, R.M. (2006). Oral chromium(VI) does not affect the frequency of micronuclei in hematopoietic cells of adult mice and of transplacentally exposed fetuses. Mutat. Res., 610(1–2): 38–47.
De Flora, S., D’Agostini, F., Balansky, R.M., Micale, R., Baluce, B. and Izzotti, A. (2008). Lack of genotoxic effects in hematopoietic and gastrointestinal cells of mice receiving chromium(VI) with the drinking water. Mutat. Res., 659(1–2): 60–67.
Deng, C., Lee, H.H. and Xian, H. (1988). Chromosomal aberrations and sister chromatid exchanges of peripheral blood lymphocytes in Chinese electroplating workers: effect of nickel and chromium. J. Trace Elem. Exp. Med., 1: 57–62.
Dionex (2003). Determination of hexavalent chromium in drinking water using ion chromatography. Dionex Corporation,Sunnyvale, CA(Application Update 144;www.dionex.com/en-us/webdocs/4242-AU144_LPN1495.pdf).
Dionex (2011). Sensitive determination of hexavalent chromium in drinking water. Dionex Corporation,Sunnyvale, CA(Application Update 179;www.dionex.com/en-us/webdocs/110230-AU179-IC-Hexavalent-Chromium-Water-AU70415_E.pdf).
Donaldson, R.M.J. and Barreras, R.F. (1966). Intestinal absorption of trace quantities of chromium. J. Lab. Clin. Med., 68: 484–493.
Drago, J., Webster, T., Williams, T., Laybourne, S. and Debra, J. (2013). Fluidized bed bioreactor treatment of hexavalent chromium, nitrate and selenium: results of pilot scale testing at Davis, California. In: Proceedings of the American Water Works Association Water Quality Technology Conference, Long Beach, California. American Water Works Association, Denver, Colorado.
DSWA and City of Ripon (2010). City of Ripon, California Proposition 50 Project—Integrated nitrate and arsenic treatment demonstration. Damon S. Williams Associates and City of Ripon, California(ID No. P50-3910007-055).
Eadie, G.S. and Brown, I.W.,Jr.(1954). The potential life span and ultimate survival of fresh red blood cells in normal healthy recipients as studied by simultaneous Cr51 tagging and differential hemolysis. J. Clin. Invest., 34: 629–636 [cited in O’Flaherty et al., 2001].
Eaton, A., Ramirez, L. and Haghani, A. (2001). The Erin Brockovich factor—analysis of total chromium and hexavalent chromium in drinking water. In: Proceedings of the American Water Works Association Water Quality Technology Conference, Nashville, TN. American Water Works Association, Denver, Colorado.
Ebaugh, J.F.G., Samuels, A.J., Dobrowski, P. and Heisterkamp, D. (1961). The site of the CrO4=–hemoglobin bond as determined by starch electrophoresis and chromatography. Fed. Proc., 20: 70 [cited in O’Flaherty et al., 2001].
Elbetieha, A. and Al-Hamood, M.H. (1997). Long-term exposure of male and female mice to trivalent and hexavalent chromium compounds: effect on fertility. Toxicology, 116: 39–47 [cited in ATSDR, 2012].
EMR (1989). Canadian mineral deposits not being mined in 1989. National Mineral Inventory, Energy, Mines and Resources Canada, Ottawa, Ontario.
Environment Canada (1991). Toxic chemicals in the Great Lakes and associated effects. Vol. 1. Contaminant levels and trends. Environment Canada, Fisheries and Oceans Canada and Health and Welfare Canada, Ottawa, Ontario. 448 pp.
Environment Canada and Health Canada (1994). Canadian Environmental Protection Act. Priority Substances List assessment report. Chromium and its compounds. Government of Canada, Ottawa, Ontario (www.hc-sc.gc.ca/ewh-semt/alt_formats/hecs-sesc/pdf/pubs/contaminants/psl1-lsp1/chromium_chrome/chromium_chrome_e.pdf).
Environment Canada, Environment Ontario, Ontario Ministry of Natural Resources, and Metropolitan Toronto and Region Conservation Authority(1988). Metro Toronto remedial action plan: environmental conditions and problem definition. Ontario Ministry of the Environment, Toronto, Ontario.
Erickson, P.E. and Fowler, B.R. (1987). The flux of suspended particulates, petroleum related hydrocarbons, trace metals and nutrients from the Mackenzie River during the winter season: a pilot study of the East Channel Ottawa. Indian and Northern Affairs Canada, Ottawa, Ontario. 124 pp. (Environmental Studies No. 48) [cited in Environment Canada, 1996].
European Chemicals Bureau (2005). European Union risk assessment report—chromium trioxide, sodium chromate, sodium dichromate, ammonium dichromate, potassium dichromate. Joint Research Centre, European Commission (EUR 21508 EN; www.echa.europa.eu/documents/10162/3be377f2-cb05-455f-b620-af3cbe2d570b).
Eyring, A., Weinberg,H. and Singer, P.(2002).Measurement and effect of trace element contaminants in alum. J. Am. Water Works Assoc., 94(5):135–146.
Fairhurst, S. and Minty, C.A. (1989). The toxicity of chromium and inorganic chromium compounds. HSE Books, UK Health and Safety Executive (Toxicity Review 21).
Fatoki, O.S. and Ogunfowokan, A.O. (2002). Effect of coagulant treatment on the metal composition of raw water. Water SA, 28(3): 293–298.
Febel, H., Szegedi, B. and Huszar, S. (2011). Absorption of inorganic, trivalent and hexavalent chromium following oral and intrajejunal doses in rats. Acta Vet. Hung., 49(2): 203–209.
Fendorf, S.E. and Li, G. (1996). Kinetics of chromate reduction by ferrous iron. Environ. Sci.Technol., 30:1614–1617.
Fendorf, S.E. and Zasoski, R.J. (1992). Chromium(III) oxidation by δ-MnO2.1. Characterization. Environ. Sci. Technol., 26:79–85.
Ferre-Huguet, N., Marti-Cid, R., Schuhmacher, M. and Domingo, J.L. (2008). Risk assessment of metals from consuming vegetables, fruits and rice grown on soils irrigated with waters of the Ebro River in Catalonia, Spain. Biol. Trace Elem. Res., 123(1–3): 66–79.
Finley, B., Kerger, B.D., Corbett, G.E., Katona, M., Gargas, M., Reitz, R. and Paustenbach, D.J. (1997). Human ingestion of chromium(VI) in drinking water: pharmacokinetics following repeated exposure. Toxicol. Appl. Pharmacol., 142: 151–159 [cited in Paustenbach et al., 2003].
Fishbein, L. (1984). Overview of analysis of carcinogenic and/or mutagenic metals in biological and environmental samples: I. Arsenic, beryllium, cadmium, chromium and selenium. Int. J. Environ. Anal. Chem., 17: 113–170.
Flegal, R., Last, J., McConnell, E.E., Schenker, M. and Witschi, H. (2001). Scientific review of toxicological and human health issues related to the development of a public health goal for chromium(VI). Report prepared by the Chromate Toxicity Review Committee, Office of Environmental Health Hazard Assessment, California Environmental Protection Agency (www.oehha.org/public_info/facts/pdf/CrPanelRptFinal901.pdf).
Frey, M., Seidel, C., Edwards, M., Parks, J. and McNeell, L (2004). Occurrence survey of boron and hexavalent chromium. American Water Works Association Research Foundation,Denver, CO (Report No. 91044F).
Friedman, M., Hull, A., Reiber, S., Valentine, R., Larsen, G., Young, A., Korshin, G. and Peng, C.-Y. (2010). Assessment of inorganic accumulation in drinking water system scale and sediments. Water Research Foundation, Denver, CO (Report No.3118).
Fryzek, J.P., Mumma, M.T., McLaughlin, J.K., Henderson, B.E. and Blot, W.J. (2001). Cancer mortality in relation to environmental chromium exposure. J. Occup. Environ. Med., 43(7): 635–640.
Gambelunghe, A., Piccinini, R., Ambrogi, M., Villarini, M., Moretti, M., Marchetti, C., Abbritti, G. and Muzi, G. (2003). Primary DNA damage in chrome-plating workers. Toxicology, 188(2–3): 187–195.
Gammelgaard, B., Jensen, K. and Steffansen, B. (1999). In vitrometabolism and permeation studies in rat jejunum: organic chromium compared to inorganic chromium. J. Trace Elem. Med. Biol., 13(1–2): 82–88.
Gatto, N.M., Kelsh, M.A., Mai, D.H., Suh, M. and Proctor, D.M. (2010). Occupational exposure to hexavalent chromium and cancers of the gastrointestinal tract: a meta-analysis. Cancer Epidemiol., 34(4): 388–399.
Gibb, H.J., Lees, P.S.J., Pinsky, P.F. and Rooney, B.C. (2000). Lung cancer among workers in chromium chemical production. Am. J. Ind. Med., 38: 115–126.
Gibbs, R.J. (1977). Transport phases of transition metals in the Amazon and Yukon rivers. Geol. Soc. Am. Bull., 88: 829–843.
Giroux, M., Rompré, M., Carrier, D., Audesse, P. and Lemieux, M. (1992). Caractérisation de la teneur en métaux lourds totaux et disponibles des sols du Québec. Agrosol, 5(2): 46–55.
Gizyn, W.I. (1994). Windsor Air Quality Study: soil and garden produce survey results. Phytotoxicology Section, Standards Development Branch, Ontario Ministry of Environment and Energy. 15 pp.
Gordon, E. (2007). Captan: transition from 'B2' to 'not likely'. How pesticide registrants affected the EPA Cancer Classification Update. J. Appl. Toxicol., 27(5):519–526.
Gomes, E. (1972). Incidence of chromium-induced lesions among electroplating workers in Brazil. Ind. Med., 41(12): 21–25.
Government of the Northwest Territories (2010). Personal communication with D. Fleming, Department of Health and Social Services..
Government of Yukon (2010). Personal communication with P. Brooks, Health and Social Services, Environmental Health Services.
Gray, S.J. and Sterling, K. (1950). The tagging of red cells and plasma proteins with radioactive chromium. J. Clin. Invest., 29(12): 1604–1613.
Hafiane, A., Lemordant, D. and Dhahbi, M. (2000). Removal of hexavalent chromium by nanofiltration. Desalination, 130: 305–312.
Halasová, E., Bukovska, E., Kukura, F., Cervenová, T., Oravec, P. and Kereskéni, J. (2001). Do works concerning ferrochromium alloys mean risk for the inhabitants living in their surrounding? A cytogenetic study. Biologia, 56(6): 679–683.
Haluschak, P.W., Mills, G.F., Eilers, R.G. and Grift, S. (1998). Status of selected trace elements in agricultural soils of southern Manitoba.Land Resource Unit, Brandon Research Centre, Research Branch, Agriculture and Agri-Food Canada (Research Branch Technical Bulletin 1998-6E; www.gov.mb.ca/agriculture/land/soil-survey/pubs/fss03s00a.pdf).
Hammond, C. (2002). The elements. In: Lide, D. (ed.), CRC handbook of chemistry and physics, 2002–2003. CRC Press, New York, New York. pp. 1–10 [cited in Stoecker, 2004].
Hanslian, L., Navratil, J. and Jurak, J. (1967). [Damage to the upper respiratory tract by a chromic acid aerosol.] Pracovni Lekarstvi, 19: 294–298 (in Czechoslovakian).
Hayes, R.B., Lilienfeld, A.M. and Snell, L.M. (1979). Mortality in chromium chemical production workers: aprospective study. Int. J. Epidemiol., 8(4): 365–374.
Health Canada. (2005). Fact sheet on chromated copper arsenate (CCA) treated wood. Pest Management Regulatory Agency, Health Canada, Ottawa, Ontario. 12 pp. (Report No. H113-2/11-2005E-PDF; http://dsp-psd.pwgsc.gc.ca/collection_2007/hc-sc/H113-2-11-2005E.pdf).
Health Canada. (2009). Monograph: Chromium (from chromium picolinate). Natural Health Products Ingredient Database. http://webprod.hc-sc.gc.ca/nhpid-bdipsn/monoReq.do?id=65
Henning, F.A. and Konasewich, D.E. (1984). Characterization and assessment of wood preservation facilities in British Columbia. Environmental Protection Service, Pacific Region, Environment Canada, West Vancouver, B.C. 206 pp.
Hirose, T., Kondo, K., Takahashi, Y., Ishikura, H., Fujino, H., Tsuyuguchi, M., Hashimoto, M., Yokose, T., Mukai, K., Kodama, T. and Monden, Y. (2002). Frequent microsatellite instability in lung cancer from chromate-exposed workers. Mol. Carcinog., 33(3): 1772–1780.
Hjollund, N. H. I., Bonde, J. P. E., Jensen, T. K., Ernst, E., Henriksen, T. B., Kolstad, H. A., Giwercman, A., Skakkebæk, N. E. and Olsen, J. (1998). Semen quality and sex hormones with reference to metal welding. Reprod. Toxicol. 12(2): 91– 95.
Hu, B. and Luk, S. (2011). Removal of contaminants from groundwater by a porous iron composite media with high permeability.In: Proceedings of the American Water Works Association Annual Conference and Exposition, Washington, DC. American Water Works Association, Denver, Colorado.
Husgafvel-Pursiainen, K., Kalliomaki, P.L. and Sorsa, M. (1982). A chromosome study among stainless steel workers. J. Occup. Med., 24: 762–766.
IARC (1990). Chromium and chromium compounds. Chromium [VI] (Group 1). Metallic chromium and chromium [III] compounds (Group 3). IARC Monogr. Eval. Carcinog. Risks Hum., 49: 49–256. Available at: http://monographs.iarc.fr/ENG/Monographs/vol49/index.php
IARC (2012). A review of human carcinogens: arsenic, metals, fibres, and dusts. IARC Monogr. Eval. Carcinog. Risks Hum., 100C: 147–167. Available at: http://monographs.iarc.fr/ENG/Monographs/vol100C/index.php
Icopini, G.A. and Long,D.T. (2002). Speciation of aqueous chromium by use of solid-phase extractions in the field. Environ. Sci. Technol., 36(13):2994–2999.
Inoue, Y., Sakai, T. and Kumagai, H.(1995). Simultaneous determination of chromium(III) andchromium(VI) by ion chromatography with inductively coupled plasma mass spectrometry. J. Chromatogr. A, 706(1–2):127–136.
Institute of Medicine (2001). Dietary reference intakes for vitamin A, vitamin K, arsenic, boron, chromium, copper, iodine, iron, manganese, molybdenum, nickel, silicon, vanadium and zinc. A report of the Panel of Micronutrients, Subcommittees on Upper Reference Levels of Nutrients and of Interpretation and Uses of Dietary Reference Intakes, and the Standing Committee on the Scientific Evaluation of Dietary Reference Intakes, Food and Nutrition Board, Institute of Medicine. National Academy Press, Washington, DC. Available at: www.nap.edu/books/0309072794/html/
IPCS (2013). Inorganic chromium(VI) compounds. International Programme on Chemical Safety, World Health Organization, Geneva, Switzerland (Concise International Chemical Assessment Document 78). Available at: www.inchem.org/documents/cicads/cicads/cicad_78.pdf
Ivankovic, S. and Preussmann, R. (1975). Absence of toxic and carcinogenic effects after administration of high doses of chromic oxide pigment in subacute and long-term feeding experiments in rats. Food Cosmet. Toxicol., 13: 347–351.
Jaworski, J.F. (1985). Chromium update 1984: Environmental and nutritional effects of chromium. National Research Council of Canada, Ottawa, Ontario. 56 pp. (NRCC No. 23917).
Johnson, C.A., Sigg, L. and Lindauer, U. (1992). The chromium cycle in a seasonally anoxic lake. Limnol. Oceanogr., 37: 315–321.
Jorhem, L., Astrand, C., Sundstrom, B., Baxter, M., Stokes, P., Lewis, J. and Grawe, K.P. (2008). Elements in rice on the Swedish market: Part 2. Chromium, copper, iron, manganese, platinum, rubidium, selenium and zinc. Food Addit. Contam. A Chem. Anal. Control Expo. Risk Assess., 25(7): 841–850.
Junaid, M., Murthy, R.C. and Saxena, D.K. (1996). Embryo- and fetotoxicity of chromium in pregestationally exposed mice. Bull. Environ. Contam. Toxicol., 57: 327–334.
Kaprara, E., Kazakis, N., Simeonidis, K., Coles, S., Zouboulis, A.I., Samaras, P. and Mitrakas, M. (2015). Occurrence of Cr(VI) in drinking water of Greece and relation to the geological background. J. Hazard. Mater., 281: 2–11.
Kauss, P.B., Hamdy, Y.S. and Hamma,B.S. (1988). St. Lawrence River environmental investigations. Vol. 1.Background: Assessment of water, sediment and biota in the Cornwall, Ontario and Massena, New York section of the St. Lawrence River, 1979–1982. Ontario Ministry of the Environment, Toronto, Ontario.
Kemper, J.M., Westerhoff, P., Dotson, A. and Mitch, W.A. (2009). Nitrosamine, dimethylnitramine and chloropicrin formation during strong base anion-exchange treatment. Environ. Sci. Technol., 43(2):466–472.
Kerger, B.D., Paustenbach, D., Corbett, G.E. and Finley, B.L. (1996). Absorption and elimination of trivalent and hexavalent chromium in humans following ingestion of a bolus dose in drinking water. Toxicol. Appl. Pharmacol., 141: 145–158.
Kerger, B.D., Finley, B., Corbett, G.E., Dodge, D.G. and Paustenbach, D. (1997). Ingestion of chromium(VI) in drinking water by human volunteers: absorption, distribution, and excretion of single and repeated doses. J. Toxicol. Environ. Health, 50: 67–95.
Kerger, B.D., Butler W.J., Paustenbach, D., Zhang, J., and Li, S. (2009). Cancer mortality in chinese populations surrounding an alloy plant with chromium smelting operations. J. Toxicol. Environ. Health. A., 72(5):329-44.
Kim, C., Zhou, Q.,Deng, B., Thornton, E.C. and Xu, H. (2001). Chromium(VI) reduction by hydrogen sulfide in aqueous media: stoichiometry and kinetics. Environ. Sci. Technol., 35(11):2219–2225.
Kimbrough, D.E., Cohen, Y.E., Winer, A.M., Creelman, L.M. and Mabuni, C.M. (1999). A critical assessment of chromium in the environment. Crit. Rev. Environ. Sci. Technol., 29(1): 1–46.
Kirman, C.R., Hays, S.M., Aylward, L.L., Suh, M., Harris, M.A., Thompson, C.M., Haws, L.C. and Proctor, D.M. (2012). Physiologically based pharmacokinetic model for rats and mice orally exposed to chromium. Chem. Biol. Interact., 200(1): 45–64.
Kirman, C.R., Aylward, L.L., Suh, M., Harris, M.A., Thompson, C.M., Haws, L.C., Proctor, D.M., Lin, S.S., Parker, W. and Hays, S.M. (2013). Physiologically based pharmacokinetic model for humans orally exposed to chromium. Chem. Biol. Interact., 204(1): 13–27.
Kirpnick-Sobol, Z., Reliene, R. and Schiestl, R.H. (2006). Carcinogenic Cr(VI) and the nutritional supplement Cr(III) induce DNA deletions in yeast and mice. Cancer Res., 66(7): 3480–3484.
Kiyak, B., Ozer, A., Altundogan, H.S., Erdem, M. and Tumen, F. (1999). Reduction in aqueous solution by using copper smelter slag. Waste Manage., 19(5): 333–338.
Kleinfeld, M. and Rosso, A. (1965). Ulcerations of the nasal septum due to inhalation of chromic acid mist. Ind. Med. Surg., 24: 242–243.
Knutsson, A., Damber, L. and Järvholm, B. (2000). Cancers in concrete workers: results of a cohort study of 33 668 workers. Occup. Environ. Med., 57(4): 264–267.
Kopec, A.K., Kim, S., Forgacs, A.L., Zacharewski, T.R., Proctor, D.M., Harris, M.A., Haws, L.C. and Thompson, C.M. (2012a). Genome-wide gene expression effects in B6C3F1 mouse intestinal epithelia following 7 and 90 days of exposure to hexavalent chromium in drinking water. Toxicol. Appl. Pharmacol., 259(1): 13–26.
Kopec, A.K., Thompson, C.M., Kim, S., Forgacs, A.L. and Zacharewski, T.R. (2012b). Comparative toxicogenomic analysis of oral Cr(VI) exposure effects in rat and mouse small intestinal epithelia. Toxicol. Appl. Pharmacol., 262(2): 124–138.
Korallus, U., Harzdorf, C. and Lewalter, J. (1984). Experimental bases for ascorbic acid therapy of poisoning by hexavalent chromium compounds. Int. Arch. Occup. Environ. Health, 53(3): 247–256 [cited in O’Flaherty et al., 2001].
Koshi, K., Yagami, T. and Nakanishi, Y. (1984). Cytogenetic analysis of peripheral blood lymphocytes from stainless steel welders. Ind. Health, 22: 305–318.
Kotas, J. and Stasicka, Z. (2000). Chromium occurrence in the environment and methods of its speciation. Environ.Pollut., 107:263–283.
Krishnan, K. and Carrier, R. (2008). Approaches for evaluating the relevance of multiroute exposures in establishing guideline values for drinking water contaminants. J. Environ. Sci. Health C Environ. Carcinog. Ecotoxicol. Rev., 26(3): 300–316.
Kuja, A., Jones, R. and McLaughlin, D. (2000). Soil contamination in Port Colborne woodlots: 2000. Ontario Ministry of the Environment, Port Colborne, Ontario. 30 pp. (Report No. SDB-012-3511-2001).
Kumar, S., Sathwara, N.G., Gautam, A.K., Agarwal, K., Shah, B., Kulkarni, P.K., Patel, K., Patel, A., Dave, L.M., Parikh, D.J. and Saiyed, H.N. (2005). Semen quality of industrial workers occupationally exposed to chromium. J. Occup. Health, 47(5): 424–430.
Kuo, H.W., Lai, J.S. and Lin,T.I. (1997). Nasal septum lesions and lung function in workers exposed to chromic acid in electroplating factories. Int. Arch. Occup. Environ. Health, 70: 272–276.
Kuykendall,J.R., Kerger B.D., Jarvi E.J., Corbett G.E., and Paustenbach D.J. (1996). Measurement of DNA-protein cross-links in human leukocytes following acute ingestion of chromium in drinking water. Carcinogenesis, 17(9):1971–1977.
Lai, H. and McNeill, L. (2006). Chromium redox chemistry in drinking water systems. J. Environ. Eng., 132(8): 841–852.
Lai, J.S., Kuo, H.W. and Liao, F.C. (1998). Sister chromatid exchange induced by chromium compounds in human lymphocytes. Int. Arch. Occup. Environ Health, 71: 550–553.
Larese, F., Gianpietro, A., Venier, M., Maina, G. and Renzi, N. (2007). In vitro percutaneous absorption of metal compounds. Toxicol. Lett., 170(1): 49–56.
Lee, G. and Hering, G. (2003).Removal of chromium (VI) from drinking water by redox-assisted coagulation with iron (II). J.Water Supply Res. Technol. Aqua, 52: 319–332.
Li, H., Chen, Q., Li, S., Yao, W., Li, L., Shi, X., Wang, L., Castranova, V., Vallyathan, V., Ernst, E. and Chen, C. (2001). Effect of Cr(VI) exposure on sperm quality: human and animal studies. Ann. Occup. Hyg., 45(7): 505–511. Comment in: Ann. Occup. Hyg., 2002, 46(2):269–270.
Lide, D.R. (ed.) (2008). Chromium and compounds. In: CRC handbook of chemistry and physics. CRC Press, Boca Raton, FL.
Lindsay, D., Farley, K. and Carbonaro, R.(2012). Oxidation of Cr(III) to Cr(VI) during chlorination of drinking water. J. Environ. Monit., 14:1789–1797.
Linos, A., Petralias, A., Christophi, C.A., Christoforidou, E., Kouroutou, P., Stoltidis, M., Veloudaki, A., Tzala, E., Makris, K.C. and Karagas, M.R. (2011). Oral ingestion of hexavalent chromium through drinking water and cancer mortality in an industrial area of Greece—an ecological study. Environ. Health, 10: 50.
Linster, C.L. and Van Schaftingen, E. (2007). Vitamin C biosynthesis, recycling and degradation in mammals. FEBSJ., 274(1): 1–22.
Littorin, M., Hogstedt, B. and Stromback, B. (1983). No cytogenetic effects in lymphocytes of stainless steel welders. Scand. J. Work Environ. Health, 9: 259–264.
Loyaux-Lawniczak, S., Refait, P., Ehrhardt, J., Lecomte, P. and Génin, J. (2000). Trapping of Cr by formation of ferrihydrite during the reduction of chromate ions by Fe(II)–Fe(III) hydroxysalt green rusts. Environ. Sci. Technol., 34(3): 438–443.
Lucas, J.B. and Kramkowski, R.S. (1975). Health hazard evaluation determination. Center for Disease Control, National Institute for Occupational Safety and Health,U.S. Department of Health, Education, and Welfare, Cincinnati, OH (Report No. 74-87-221).
Luczak, M.W., Green, S.E. and Zhitkovich, A. (2015). Different ATM signaling in response to chromium(VI) metabolism via ascorbate and nonascorbate reduction: implications for in vitro models and toxicogenomics. Environ. Health Perspect. (in press).
Lytle, D.A., Sorg, T.J.,Muhlen, C. and Wang,L. (2010). Particulate arsenic release in a drinking water distribution system. J. Am. Water Works Assoc., 102(3):87–98.
MacKenzie, R.D., Byerrum, R.U., Decker, C.F., Hoppert, C.A. and Langham, R.F. (1958). Chronic toxicity studies. II. Hexavalent and trivalent chromium administered in drinking water to rats. AMA Arch. Ind. Health, 18: 232–234.
MacLatchy, J. (1992). Metal data from base metal smelters and refineries. Environment Canada.
Maher, T.J. (2008). Chromium and other minerals in diabetes mellitus. U.S. Pharmacist (http://legacy.uspharmacist.com/oldformat.asp?url=newlook/files/Alte/minerals.cfm&pub_id=8&article_id=439).
Mancuso, T.F. (1997a). Chromium as an industrial carcinogen: Part I. Am. J. Ind. Med., 31: 129–139.
Mancuso, T.F. (1997b). Chromium as an industrial carcinogen: Part II. Chromium in human tissues. Am. J. Ind. Med., 31: 140–147.
Manitoba Environment and Workplace Safety and Health (1989). Chromium concentrations in Manitoba soil. Internal report provided by D. McEachem, Manitoba Environment and Workplace Safety and Health.
Manitoba Water Stewardship (2010). Personal communication with K. Phillip, Office of Drinking Water.
Mann Testing Laboratories (1992). Analysis of chromium in Canadian foods. Conducted for Health and Welfare Canada by Mann Testing Laboratories Ltd.
Markovich, D. (2001). Physiological roles and regulation of mammalian sulfate transporters. Physiol. Rev., 81(4): 1499–1533.
Maruyama, Y. (1982). The effect of mice given oral administration of trivalent and hexavalent chromium over a long-term. Gifu Daigaku Igakubo Kiyo, 314: 25–46 [cited in Environment Canada and Health Canada, 1994].
Mayer, L.M. (1988). Geochemistry of chromium in ocean.In: Nriagu, J.O. and Nieboer, E. (eds.),Chromium in the natural and human environments. John Wiley and Sons, New York, NY. pp. 173–187.
McAdory, D., Rhodes, N.R., Briggins, F., Bailey, M.M., Di Bona, K.R., Goodwin, C., Vincent, J.B. and Rasco, J.F. (2011). Potential of chromium(III) picolinate for reproductive or developmental toxicity following exposure of male CD-1 mice prior to mating. Biol. Trace Elem. Res., 143(3): 1666–1672.
McCarroll, N., Keshava, N., Chen, J., Akerman, G., Kligerman, A. and Rinde, E. (2010). An evaluation of the mode of action framework for mutagenic carcinogens case study. II: Chromium (VI). Environ. Mol. Mutagen., 51(2): 89–111.
McDowall, M.E. (1984). A mortality study of cement workers. Br. J. Ind. Med., 41(2): 179–182.
McGrachan, J. (1996). Drinking water surveillance program summary for 1994–1995 for chromium. Unpublished Ontario Ministry of Environment and Energy data. Personal communication dated February 14, 1996.
McGuire, M., Blute, N., Seidel, C., Qin, G. and Fong, L. (2006). Pilot scale studies of hexavalent chromium removal from drinking water. J. Am. Water Works Assoc., 98(2): 134–143.
McGuire, M., Blute, N. and Qin, G. (2007). Hexavalent chromium removal using anion exchange and reduction with coagulation and filtration. Water Research Foundation, Denver, Colorado (Report No. 91193).
McLean, J.E., McNeill, L.S., Edwards, M.A. and Parks, J.L. (2012). Hexavalent chromium review, part 1: health effects, regulations, and analysis. J. Am. Water Works Ass., 104(6): E348–E357.
McKeague, J.A., Desjardin, J.G. and Wolynetz, M.S. (1979). Minor elements in Canadian soils. Agriculture and Agri-Food Canada.
McNeill, L., McLean, J., Perks, J. and Edwards, M. (2012). Hexavalent chromium review. Part 2: Chemistry, occurrence and treatment. J. Am. Water Works Assoc., 104(7): E395–E405.
McNeill, L., McLean, J., Edwards, M. and Perks, J. (2013). Trace level hexavalent chromium. Occurrence and analysis. Water Research Foundation, Denver, CO (Project No. 4404).
Medeiros, M.G., Rodrigues, A.S., Batoreu, M.C., Laires, A., Rueff, J. and Zhitkovich, A. (2003). Elevated levels of DNA–protein crosslinks and micronuclei in peripheral lymphocytes of tannery workers exposed to trivalent chromium. Mutagenesis, 18(1): 19–24.
Meek, M.E., Bucher, J.R., Cohen, S.M., Dellarco, V., Hill, R.N., Lehman-McKeeman, L.D., Longfellow, D.G., Pastoor, T., Seed, J. and Patton, D.E. (2003). A framework for human relevance analysis of information on carcinogenic modes of action. Crit. Rev. Toxicol., 33(6): 591–653.
Méranger, J.C., Subramanian, K.S. and Chalifoux, C. (1981). Survey for cadmium, cobalt, chromium, copper, nickel, lead, zinc, calcium, and magnesium in Canadian drinking water supplies. J. Assoc. Off. Anal. Chem., 64(1): 44–53.
Merian, E. (1984). Introduction of environmental chemistry and global cycle of arsenic, beryllium, cadmium, chromium, cobalt, nickel, selenium, and their derivatives. Toxicol. Environ.Chem., 8: 9–38 [cited in Stoecker, 2004].
Mermut, A.R., Jain, J.C., Song, L., Kerrich, R., Kozak, L. and Jana, S. (1996). Trace element concentrations of selected soils and fertilizers in Saskatchewan, Canada. J. Environ. Qual., 25: 845–853.
Merritt, W.F. (1975). Variation in trace element concentrations along the length of the Ottawa River. Can. J. Earth Sci., 12: 850–857.
Ministère du Développement durable, de l’Environnement et des Parcs du Québec. (2010). Personal communication with C. Robert, Direction des politiques de l’eau.
Ministère du Développement durable, de l’Environnement et des Parcs du Québec. (2012). Bilan de la qualité de l’eau potable au Québec 2005–2009. Gouvernement du Québec, Québec.
Minoia, C. and Cavalleri, A. (1988). Chromium in urine, serum and red blood cells in the biological monitoring of workers exposed to different chromium valency states. Sci. Total Environ., 71(3): 323–327.
Mirsalis, J.C., Hamilton, C.M., O’Loughlin, K.G., Paustenbach, D.J., Kerger, B.D. and Patierno, S. (1996). Chromium (VI) at plausible drinking water concentrations is not genotoxic in the in vivo bone marrow micronucleus or liver unscheduled DNA synthesis assays. Environ. Mol. Mutagen., 28: 60–63.
Mukhopadhyay, B., Sundquist, J. and Schmitz, R. (2007). Removal of chromium (VI) from Cr-contaminated groundwater through electrochemical addition of Fe (II). J. Environ. Manage., 82:66–76.
Murthy, R.C., Junaid, M. and Saxena, D.K. (1996). Ovarian dysfunction in mice following chromium(VI) exposure. Toxicol. Lett., 89: 147–154.
Muthukrishnan, M. and Guha, B.K. (2008). Effect of pH on rejection of hexavalent chromium by nanofiltration. Desalination, 219: 171–178.
Nagaya, T. (1986). No increase in sister-chromatid exchange frequency in lymphocytes of chromium platers. Mutat. Res., 170: 129–132.
Nagaya, T., Ishikawa, N. and Hata, H. (1991). Sister-chromatid exchanges in lymphocytes from 12 chromium platers: a5-year follow-up study. Toxicol. Lett., 58: 329–335 [cited in ATSDR, 2012].
Najm, I. and Trussell, R. (2001). NDMA formation in water and wastewater. J. Am. Water Works Assoc., 93(2).
Najm, I., Kim, T. and Ajvani, C. (2013). Optimizing biological denitrification of groundwater—Recovering waste backwash water and co-removal of hexavalent chromium. In: Proceedings of the American Water Works Association Water Quality Technology Conference, Nashville, TN. American Water Works Association, Denver, CO.
Najm, I., Brown,N., Seo, E., Gallagher,B., Gramith,K., Blute,N., Wu,X., Yoo,M., Liang,S., Maceiko,S., Kader,S. and Lowry,J. (2014). Impact of water quality on hexavalent chromium removal efficiency and cost. Water Research Foundation, Denver, Colorado (Web Report No. 4450).
Nakamuro, K., Yoshikawa, K., Sayato, Y. and Kurata, H. (1978). Comparative studies of chromosomal aberration and mutagenicity of the trivalent and hexavalent chromium. Mutat. Res., 58(2–3): 175–181.
Newfoundland and Labrador Department of Environment and Conservation (2010). Personal communication with H. Khan, Water Resources Management Division.
Nico, P. and Zasoski, R. (2000). Importance of Mn (III) availability on the rate of Cr (III) oxidation on δ-MnO2. Environ. Sci. Technol., 34: 3363–3367.
Nieboer, E. and Jusys, A.A. (1988). Biologic chemistry of chromium. In: Nriagu, J.O. and Nieboer, E. (eds.), Chromium in the natural and human environments. John Wiley and Sons, New York, New York. pp. 21–79.
Nkhalambayausi-Chirwa,E. and Wang, Y.-T. (2001). Simultaneous chromium (VI) reduction and phenol degradation in a fixed-film coculture bioreactor: reactor performance. Water Res., 35(8):1921–1932.
Nova Scotia Department of Environment and Labour (2010). Personal communication with J. MacDonald, Water and Wastewater Branch.
NPRI (2012). Reviewed facility data release for chromium and its compounds. National Pollutant Release Inventory. Environment Canada, Ottawa. Available at: www.ec.gc.ca/inrp-npri/donnees-data/index.cfm?lang=En
Nriagu, J.O. (1988). Production and uses of chromium. In: Nriagu, J.O. and Nieboer, E.(eds.),Chromium in the natural and human environments. John Wiley and Sons, New York, New York. pp. 125–172.
Nriagu, J.O. (1990). Global metal pollution: poisoning the biosphere. Environ. Health Perspect., 32: 7–11, 28–33.
Nriagu, J.O., Beaubien, S. and Blowes, D. (1993). Chemistry of chromium in lakes. Environ. Rev., 1(2): 104–120.
NSF/ANSI (2013a). Section 7. In: NSF/ANSI Standard 53: Drinking water treatments units—Health effects. NSF International, Ann Arbor, Michigan.
NSF/ANSI (2013b).Section 7. In: NSF/ANSI Standard 58: Reverse osmosis drinking water treatment systems. NSF International, Ann Arbor, Michigan.
NSF/ANSI (2013c). Section 5. In: NSF/ANSI Standard 62: Drinking water distillation system. NSF International, Ann Arbor, Michigan.
NSF/ANSI (2014). Standard 61: Drinking water system components—health effects. NSF International/American National Standards Institute, Ann Arbor, Michigan.
NTP (1996a). Potassium dichromate (hexavalent): the effects of potassium dichromate in BALB/c mice when administered in the diet. National Toxicology Program, U.S. Department of Health and Human Services [cited in U.S. EPA, 1998b].
NTP (1996b). Potassium dichromate (hexavalent): the effects of potassium dichromate on Sprague-Dawley rats when administered in the diet. National Toxicology Program, U.S. Department of Health and Human Services [cited in U.S. EPA, 1998b].
NTP (1997). Potassium dichromate (hexavalent): reproductive assessment by continuous breeding when administered to BALB/c mice in the diet. National Toxicology Program, U.S. Department of Health and Human Services.
NTP (2007). NTP technical report of the toxicity studies of sodium dichromate dihydrate (CAS No. 7789-12-0) administered in drinking water to male and female F344/N rats and B6C3F1 mice and male BALB/c and am3-C57BL/6 mice. National Toxicology Program, U.S. Department of Health and Human Services (NTP TR 72). Available at: http://ntp.niehs.nih.gov/ntp/htdocs/st_rpts/tox072.pdf
NTP (2008). NTP technical report of the toxicology and carcinogenesis of sodium dichromate dihydrate (CAS No. 7789-12-0) in F344/N rats and B6C3F1 mice (drinking water studies). National Toxicology Program, U.S. Department of Health and Human Services.192 pp. (NTP TR 546). Available at: http://ntp.niehs.nih.gov/ntp/htdocs/LT_rpts/tr546.pdf
NTP (2010). NTP toxicology and carcinogenesis studies of chromium picolinate monohydrate (CAS No. 27882-76-4) in F344/N rats and B6C3F1 mice (feed studies). National Toxicology Program, U.S. Department of Health and Human Services. 194 pp. (NTP TR 556). Available at: http://ntp.niehs.nih.gov/ntp/htdocs/lt_rpts/tr556.pdf
O’Brien, T.J., Ceryak, S. and Patierno, S.R. (2003). Complexities of chromium carcinogenesis: role of cellular response, repair and recovery mechanisms. Mutat. Res., 533(1–2): 3–36.
O’Brien, T.J., Ding, H., Suh, M., Thompson, C.M., Parsons, B.L., Harris, M.A., Winkelman, W.A., Wolf, J.C., Hixon, J.G., Schwartz, A.M., Myers, M.B., Haws, L.C. and Proctor, D.M. (2013). Assessment of K-ras mutant frequency and micronucleus incidence in the mouse duodenum following 90-days of exposure to Cr(VI) in drinking water. Mutat. Res., 754(1–2): 15–21.
OECD (2013). Test No. 488: Transgenic rodent somatic and germ cell gene mutation assays, OECD Guidelines for the Testing of Chemicals, Section 4, OECD Publishing, Paris. Available at: http://dx.doi.org/10.1787/9789264203907-en
Offenbacher, E.G. and Pi-Sunyer, F.X. (1983). Temperature and pH effects on the release of chromium from stainless steel into water and fruit juices. J. Agric. Food Chem., 31: 89–92.
O’Flaherty, E.J. (1996). A physiologically based model of chromium kinetics in the rat. Toxicol. Appl. Pharmacol., 138: 54–64.
O’Flaherty, E.J., Kerger, B.D., Hays, S.M. and Paustenbach, D.J. (2001). A physiologically based model for the ingestion of chromium(III) and chromium(VI) by humans. Toxicol. Sci., 60(2): 196–213.
OMEE (1994). Ontario typical range of chemical parameters in soil, vegetation, moss bags and snow. April, 1994 (Version 1.0a). Ontario Ministry of the Environment and Energy, Toronto, Ontario. 212 pp. + appendices (PIBS 2792).
OMOE (1991a). The preliminary report on the first six months of the process effluent monitoring in the MISA pulp and paper sector: January 1 to June 30. Ontario Ministry of the Environment, Toronto, Ontario.
OMOE (1991b). The preliminary report on the second six months of the process effluent monitoring in the MISA pulp and paper sector:July 1 to December 30. Ontario Ministry of the Environment, Toronto, Ontario.
OMOE (2004). Data obtained from the Drinking Water Surveillance Program, Ontario Ministry of the Environment, on November 30, 2004.
OMOE (2008). Chromium level in Ontario drinking water. Personal communication with P. Cheung. Drinking Water Unit, Drinking Water Surveillance Program, Ontario Ministry of the Environment.
OMOE (2014). Personal communication with S. Deshpande, Standards Development Branch, Ontario Ministry of the Environment.
Oze, C., Bird, D.K. and Fendorf, S. (2007). Genesis of hexavalent chromium from natural sources in soil and groundwater. Proc. Natl. Acad. Sci. U.S.A., 104(16): 6544–6549.
Park, R.M. and Stayner, L.T. (2006). A search for thresholds and other nonlinearities in the relationship between hexavalent chromium and lung cancer. Risk Anal., 26(1): 79–88.
Park, R.M., Bena, J.F., Stayner, L.T., Smith, R.J., Gibb, H.J. and Lees, P.S.J. (2004). Hexavalent chromium and lung cancer in the chromate industry: aquantitative risk assessment. Risk Anal., 24(5): 1099–1108.
Parks, J., McNeill, L., Frey M., Eaton, A., Haghani, A., Ramirex, L. and Edwards, M. (2004). Determination of total chromium in environmental water samples. Water Res., 38(12):2827–2838.
Parks, J., McNeill, L. and Edwards, M. (2013). Effect of alkalinity and NOM on trace level of hexavalent chromium analysis. In: Proceedings of the American Water Works Association Water Quality Technology Conference,Long Beach, CA. American Water Works Association, Denver, CO.
Pastides, H., Austin, R.I. and Lemeshow, S. (1994). A retrospective-cohort study of occupational exposure to hexavalent chromium. Am. J. Ind. Med., 25: 663–675 [cited in U.S. EPA, 1998b].
Paustenbach, D., Hays, S., Brien, B., Dodge, D.G. and Kerger, B.D. (1996). Observation of steady state in blood and urine following human ingestion of hexavalent chromium in drinking water. J. Toxicol. Environ. Health, 49: 453–461.
Paustenbach, D.J., Finley, B.L., Mowat, F.S. and Kerger, B.D. (2003). Human health risk and exposure assessment of chromium (VI) in tap water. J. Toxicol. Environ. Health A, 66(14): 1295–1339.
PEI Department of Environment, Energy and Forestry (2010). Personal communication with G. Somers, Drinking Water, Land and Systems Protection Section.
Penney, D. (2004). The micronutrient and trace element status of forty-three soil quality benchmark sites in Alberta. Prepared for the Alberta Environmentally Sustainable Agriculture (AESA) Soil Quality Monitoring Program,Conservation and Development Branch,Alberta Agriculture, Food and Rural Development. 16 pp.
Peterson, M.L., White, A.F., Brown, G.E. and Parks, G.A (1997). Surface passivation on magnetite by reaction with aqueous chromium (VI). Environ. Sci. Technol., 31(5): 1573–1576.
Philipot, J.M., Chaffange, F. and Sibony, J. (1984). Hexavalent chromium removal from drinking water. Water Sci. Technol., 17: 1121–1132.
Phillips, D.R. (1988). Chromium. In: Canadian minerals yearbook. Energy, Mines and Resources Canada, Ottawa, Ontario. pp. 20.1–20.10.
Pilgrim, W. (1996). Ecosystem aspects of mercury pollution. In: Haya, K. and Niimi, A.J. (eds.),Proceeding of the 22nd Annual Toxicity Workshop. Department of Fisheries and Oceans, Biological Station, St. Andrews, New Brunswick. pp. 64–81.
Pilliere, F., Levy, F. and Renier, A. (1992). Induction of DNA-repair synthesis (UDS) in rat pleural mesothelial cells by urine of subjects exposed to genotoxic agents. Clin. Toxicol., 30(2): 223–238.
Pittler, M.H., Stevinson, C. and Ernst, E. (2003). Chromium picolinate for reducing body weight: meta-analysis of randomized trials. Int. J. Obes., 27: 522–529.
Powell, M.J., Boomer, D.W. and Wiederin, D.R. (1995). Determination of chromium species in environmental samples using high-pressure liquid chromatography direct injection nebulization and inductively coupled plasma mass spectrometry. Anal. Chem., 67(14):2474–2478.
Proctor, D.M., Thompson, C.M., Suh, M. and Harris, M.A. (2011). A response to “A quantitative assessment of the carcinogenicity of hexavalent chromium by the oral route and its relevance to human exposure.” Environ. Res., 111(3): 468–470. Comment on: Environ. Res., 2010, 110(8):798–807;discussion in: Environ. Res., 2011, 111(3): 471–472.
Proctor, D.M., Suh, M., Aylward, L.L., Kirman, C.R., Harris, M.A., Thompson, C.M., Gurleyuk, H., Gerads, R., Haws, L.C. and Hays, S.M. (2012). Hexavalent chromium reduction kinetics in rodent stomach contents. Chemosphere, 89(5): 487–493.
Qin, G., McGuire, M., Blute, N., Seidel, C. and Fong, L. (2005). Hexavalent chromium removal by reduction with ferrous sulphate, coagulation, and filtration: apilot-scale study. Environ. Sci. Technol., 39: 6321–6327.
Quinteros, F.A., Poliandri, A.H.B., Machiavelli, L.I., Cabilla, J.P. and Duvilanski, B.H. (2007). In vivo and in vitro effects of chromium VI on anterior pituitary hormone release and cell viability. Toxicol. Appl. Pharmacol., 218: 79–87.
Rad, M., Mirbagheri, S. and Mohammadi, T. (2009). Using reverse osmosis membrane for chromium removal from aqueous solution. World Acad. Sci. Eng. Technol., 3: 332–336.
Radimer, K., Bindewald, B., Hughes, J., Ervin, B., Swanson, C. and Picciano, M.F. (2004). Dietary supplement use by US adults: data from the National Health and Nutrition Examination Survey, 1999–2000. Am. J. Epidemiol., 160(4): 339–349.
Rai, D., Saas, B.M. and Moore,D.A. (1987). Chromium(III) hydrolysis constants and solubility of chromium(III) hydroxide. Inorg. Chem., 26(3):345–349.
Rasmussen, P.E., Subramanian, K.S. and Jessiman, B.J. (2001). A multi-element profile of house dust in relation to exterior dust and soils in the city of Ottawa, Canada. Sci. Total Environ., 267:125–140.
Read, R.C. (1954). Studies of red-cell volume and turnover using radiochromium. N. Engl. J. Med., 250: 1021–1027 [cited in O’Flaherty et al., 2001].
Reynolds, M., Armknecht, S., Johnston, T. and Zhitkovich, A. (2012). Undetectable role of oxidative DNA damage in cell cycle, cytotoxic and clastogenic effects of Cr(VI) in human lung cells with restored ascorbate levels. Mutagenesis, 27(4): 437–443.
Rhodes, M.C., Hébert, C.D., Herbert, M.A., Morinello, E.J., Roycroft, J.H., Travlos, G.S. and Abdo, K.M. (2005). Absence of toxic effects in F344/N rats and B6C3F1 mice following subchronic administration of chromium picolinate monohydrate. Food Chem. Toxicol., 43: 21–29 [cited in Costa and Klein, 2006].
Roberts, B.A. (1980). Some chemical and physical properties of serpentine soils from western Newfoundland. Can. J. Soil Sci., 60: 231–240.
Rock, M., Lames, B. and Helz, G. (2001). Hydrogen peroxide effects on chromium oxidation state and solubility in four diverse chromium enriched soils. Environ. Sci. Technol., 35:4054–4059.
Rose, M., Baxter, M., Brereton, N. and Baskaran, C. (2010). Dietary exposure to metals and other elements in the 2006 UK Total Diet Study and some trends over the last 30 years. Food Addit. Contam. A Chem. Anal. Control Expo. Risk Assess., 27(10): 1380–1404.
Rosenman, K.D. and Stanbury, M.S. (1996). Risk of lung cancer among former chromium smelter workers. Am. J. Ind. Med., 29: 491–500 [cited in U.S. EPA, 1998b;CalEPA, 2011].
Rossmann, R. and Barres,J. (1988). Trace element concentrations in near-surface waters of the Great Lakes and methods of collection, storage, and analysis. J. Great Lakes Res., 14(2): 188–204.
Royle, H. (1975). Toxicity of chromic acid in the chromium plating industry (2). Environ. Res., 10: 141–163.
Saleh, F.Y., Parkerton, T.F. and Lewis, R.V. (1989). Kinetics of chromium transformations in the environment. Sci. Total Environ., 86: 25–41.
Samuel, J.B., Stanley, J.A., Roopha, D.P., Vengatesh, G., Anbalagan, J., Banu, S.K. and Aruldhas, M.M. (2011). Lactational hexavalent chromium exposure-induced oxidative stress in rat uterus is associated with delayed puberty and impaired gonadotropin levels. Hum. Exp. Toxicol., 30(2): 91–101.
Sanexen (2009). Canadian soil quality guidelines for contaminated sites—Human health effects. Scientific supporting document. Report prepared by Sanexen Environmental Services Inc. for Health Canada.
Saputro, S., Yoshimura, K., Takehara, K., Matsuoka, S. and Narsito. (2011). Oxidation of chromium (III) by free chlorine in tap water during the chlorination process studied by an improved solid-phase spectrometry. Anal. Sci., 27: 649–652.
Sarkar, D., Sharma, A. and Talukder, G. (1993). Differential protection of chlorophyllin against clastogenic effects of chromium and chlordane in mouse bone marrow in vivo. Mutat. Res., 301: 33–38.
Sarto, F., Cominato, I. and Bianchi, V. (1982). Increased incidence of chromosomal aberrations and sister chromatid exchanges in workers exposed to chromic acid (CrO3) in electroplating factories. Carcinogenesis, 3(9): 1011–1016.
Saskatchewan Environment (2010). Personal communication with S. Ferris, Municipal Branch.
Sasso, A.F. and Schlosser, P.M. (2015). An evaluation of in vivo models for toxicokinetics of hexavalent chromium in the stomach. Toxicol. Appl. Pharmacol., 287(3): 293–298.
Satoh, K., Fukuda, Y., Torii, K. and Katsuno, N. (1981). Epidemiological study of workers engaged in the manufacture of chromium compounds. J. Occup. Med., 23: 835–838.
SCC (2014). Directory of accredited product, process and service certification bodies. Standards Council of Canada, Ottawa, Ontario . Available at: www.scc.ca/en/accreditation/product-process-and-service-certification/directory-of-accredited-clients
Schlautman, M.A. and Han, I. (2001). Effect on pH and dissolved oxygen on the reduction of hexavalent chromium by dissolved ferrous iron in poorly buffered aqueous solution. Water Res., 35(6):1534–1546.
Schlosser, P.M. and Sasso, A.F. (2014). A revised model of ex-vivo reduction of hexavalent chromium in human and rodent gastric juices. Toxicol. Appl. Pharmacol., 280(2): 352–361.
Schock , M., Hyland, R. and Welch, M.(2008). Occurrence of contaminant accumulation in lead pipe scales from domestic drinking water distribution systems.. J. Environ. Sci. Technol. 42:4285-4291.
Schroeder, H.A., Balassa, J.J. and Vinton, W.H. (1964). Chromium, lead, cadmium, nickel and titanium in mice: effect on mortality, tumors and tissue levels. J. Nutr., 83: 239–250 [cited in Environment Canada and Health Canada, 1994].
Schroeder, H.A., Balassa, J.J. and Vinton, W.H. (1965). Chromium, cadmium and lead in rats: effects on life span, tumors and tissue levels. J. Nutr., 86: 51–66 [cited in Environment Canada and Health Canada, 1994.
Schuhmacher, M., Domingo, J.L. and Llobet, J.M. (1993). Chromium, copper, and zinc concentrations in edible vegetables grown in Tarragona Province, Spain. Bull. Environ. Contam. Toxicol., 50: 514–521 [cited in ATSDR, 2012].
Sedman, R.M., Beaumont, J., McDonald, T.A., Reynolds, S., Krowech, G. and Howd, R. (2006). Review of the evidence regarding the carcinogenicity of hexavalent chromium in drinking water. J. Environ. Sci. Health C, 24(1): 155–182.
Seidel, C.J. and Corwin, C.J., (2013) Total chromium and hexavalent chromium occurrence analysis. J.Am.Water Works Assoc., 105 (6), E310-319.
Sengupta, A., Clifford, D. and Subramonian, S. (1986). Chromate ion-exchange process at alkaline pH. Water Res., 20(9):1177–1184.
Shara, M., Yasmin, T., Kincaid, A.E., Limpach, A.L., Bartz, J., Brenneman, K.A., Chatterjee, A., Bagchi, M., Stohs, S.J. and Bagchi, D. (2005). Safety and toxicological evaluation of a novel niacin-bound chromium (III) complex. J. Inorg. Biochem., 99(11): 2161–2183.
Sharma, S.K., Petrusevski, B. and Amy, G. (2008). Chromium removal from water. J. Water Supply Res. Technol. Aqua, 57(8): 541–553.
Sharpe, D.R. and Rasmussen, P.E. (1996). Soil geochemical survey of southern Ontario. Geological Survey of Canada, Ottawa, Ontario.
Shindo, Y., Toyoda, Y., Kawamura, K., Kurebe, M., Shimada, H., Hattori, C. and Satake, S. (1989). Micronucleus test with potassium chromate(VI) administered intraperitoneally and orally to mice. Mutat. Res., 223: 403–406 [cited in Sedman et al., 2006].
Shiraki, K. (1978). Chromium. Springer-Verlag, Berlin, Federal Republic of Germany.
Shock, M., Hyland, R. and Welch, M. (2008). Occurrence of contaminant accumulation in lead pipe scales fromdomestic drinking water distribution systems. Environ. Sci. Technol., 42(12):4285–4291.
Shupack, S.I. (1991). The chemistry of chromium and some resulting analytical problems. Environ. Health Perspect., 92: 7–11.
Siemiatycki, J. (1991). Risk factors for cancer in the workplace. CRC Press, Boca Raton, FL [cited in Environment Canada and Health Canada, 1994].
Silberg, D.G., Wang, W., Moseley, R.H. and Traber, P.G. (1995). The down regulated in adenoma (dra) gene encodes an intestine-specific membrane sulfate transport protein. J. Biol. Chem., 270: 11897–11902.
Sloof, W. (1989). Integrated criteria document chromium. National Institute of Public Health and Environmental Protection, Bilthoven, Netherlands (Report No. 758701002) [cited in WHO, 1996].
Soares, M.E., Vieira, E. and de Lourdes Bastos, M. (2010). Chromium speciation analysis in bread samples. J. Agric. Food Chem., 58(2): 1366–1370.
Song, R. (2012). Cr-6 removal/minimization at Louisville Water Company: a successful story. In: Proceedings of the American Water Works Association Annual Conference and Exposition, Dallas, TX. American Water Works Association, Denver, Colorado.
Soon, Y.K. and Abboud, S. (1990). Trace elements in agricultural soils of northwestern Alberta. Can. J. Soil Sci., 70: 277–288.
Sorahan, T., Burgess, D.C. and Waterhouse, J.A. (1987). A mortality study of nickel/chromium platers. Br. J. Ind. Med., 44: 250–258 [cited in U.S. EPA, 1998b].
Sorg, T.J. (1979).Treatment technologies to meet the interim primary drinking water regulation for organics: Part 4. J. Am. Water Works Assoc., 71(8):454–460.
Soudani, N., Ben Amara, I., Sefi, M., Boudawara, T. and Zeghal, N. (2011a). Effects of selenium on chromium (VI)–induced hepatotoxicity in adult rats. Exp. Toxicol. Pathol., 63(6): 541–548.
Soudani, N., Ben Amara, I., Troudi, A., Bouaziz, H., Boudawara, T. and Zeghal, N. (2011b). Oxidative stress induced by chromium (VI) in bone of suckling rats. Toxicol. Ind. Health, 27(8): 724–734.
Soudani, N., Bouaziz, H., Sefi, M., Chtourou, Y., Boudawara, T. and Zeghal, N. (2013). Toxic effects of chromium (VI) by maternal ingestion on liver function of female rats and their suckling pups. Environ. Toxicol., 28(1):11–20.
Staniek, H. and Krejpcio, Z. (2009). The effects of tricentric chromium(III) propionate complex supplementation on pregnancy outcome and maternal and foetal mineral status in rat. Food Chem. Toxicol., 47(10): 2673–2678.
Stearns, D.M. (2000). Is chromium a trace essential metal? Biofactors, 11(3): 149–162.
Stella, M., Montaldi, A. and Rossi, R. (1982). Clastogenic effects of chromium on human lymphocytes in vitro and in vivo. Mutat. Res., 101: 151–164.
Stern, A.H. (2010). A quantitative assessment of the carcinogenicity of hexavalent chromium by the oral route and its relevance to human exposure. Environ Res., 110(8): 798–807. Comment by Proctor et al. in: Environ Res., 2011, 111(3):468–470; reply by authorin: Environ. Res., 2011, 111(3):471–472.
Stoecker, B. (2004). Chromium. In: Merian, E.,Anke, M., Ihnat, M. and Stoeppler, M. (eds.),Elements and their compounds in the environment: occurrence, analysis and biological relevance. 2nd edition.Wiley-VCH. pp. 709–729.
Stout, M.D., Herbert, R.A., Kissling, G.E., Collins, B.J., Travlos, G.S., Witt, K.L., Melnick, R.L., Abdo, K.M., Malarkey, D.E. and Hooth, M.J. (2009). Hexavalent chromium is carcinogenic to F344/N rats and B6C3F1 mice after chronic oral exposure. Environ. Health Perspect., 117(5): 716–722.
Subramanian, S., Rajendiran, G., Sekhar, P., Gowri, C., Govindarajulu, P. and Aruldhas, M.M. (2006). Reproductive toxicity in adult bonnet monkeys (Macaca radiata Geoffrey). Reversible oxidative stress in the semen. Toxicol. Appl. Pharmacol., 215: 237–249.
Suh, M., Thompson, C.M., Kirman, C.R., Carakostas, M.C., Haws, L.C., Harris, M.A. and Proctor, D.M. (2014). High concentrations of hexavalent chromium in drinking water alter iron homeostasis in F344 rats and B6C3F1 mice. Food Chem. Toxicol., 65: 381–388.
Summit Toxicology (2014). Physiologically based pharmacokinetic (PBPK) modeling support for the assessment of hexavalent chromium. Report prepared for Health Canada. Available upon request from water_eau@hc-sc.gc.ca
Suzuki, Y., Homma, K., Minami, M. and Yoshikawa, H. (1984). Distribution of chromium in rats exposed to hexavalent chromium and trivalent chromium aerosols. Ind. Health, 22: 261–267.
Taleb-Ahmed, M., Taha, S., Maachi, R. and Dorange, G. (2002). The influence of physico-chemistry on the retention of chromium ions during nanofiltration. Desalination, 145: 103–108.
Taylor, M.C., Reeder, S.W. and Demayo, A. (1979). Chromium. In: Guidelines for surface water quality. Vol.1. Inorganic chemical substances. Water Quality Branch, Inland Waters Directorate, Environment Canada, Ottawa, Ontario.
TERA (2009). Independent peer consultation for hexavalent chromium mode of action research. Toxicology Excellence for Risk Assessment. Available at: www.tera.org/Peer/Chromium/Chromium.htm
TERA (2012). Peer review report on the chromium mode of action studies: mode of action analysis. Expert review organized by Toxicology Excellence for Risk Assessment (TERA). December. Available at: www.tera.org/Peer/MOA analysis peer review report.pdf
Theopold, K. (1994). Chromium: inorganic and coordination chemistry. In: King, R.B. (ed.), Encyclopedia of inorganic chemistry. John Wiley &Sons, New York, NY.pp. 666–678.
Thompson, C.M., Proctor, D.M., Haws, L.C., Hébert, C.D., Grimes, S.D., Shertzer, H.G., Kopec, A.K., Hixon, J.G., Zacharewski, T.R., and Harris M.A. (2011). Investigation of the mode of action underlying the tumorigenic response induced in B6C3F1 mice exposed orally to hexavalent chromium. Toxicol Sci., 123(1):58-70.
Thompson, C.M., Fedorov, Y., Brown, D.D., Suh, M., Proctor, D.M., Kuriakose, L., Haws, L.C. and Harris, M.A. (2012a). Assessment of Cr(VI)-induced cytotoxicity and genotoxicity using high content analysis. PLoS One, 7(8): e42720.
Thompson, C.M., Hixon, J.G., Proctor, D.M., Haws, L.C., Suh, M., Urban, J.D. and Harris, M.A. (2012b). Assessment of genotoxic potential of Cr(VI) in the mouse duodenum: anin silico comparison with mutagenic and nonmutagenic carcinogens across tissues. Regul. Toxicol. Pharmacol., 64(1): 68–76.
Thompson, C.M., Proctor, D.M., Suh, M., Haws, L.C., Hébert, C.D., Mann, J.F., Shertzer, H.G., Hixon, J.G. and Harris, M.A. (2012c). Comparison of the effects of hexavalent chromium in the alimentary canal of F344 rats and B6C3F1 mice following exposure in drinking water: implications for carcinogenic modes of action. Toxicol. Sci., 125(1): 79–90.
Thompson, C.M., Proctor, D.M., Suh, M., Haws, L.C., Kirman, C.R. and Harris, M.A. (2013). Assessment of the mode of action underlying development of rodent small intestinal tumors following oral exposure to hexavalent chromium and relevance to humans. Crit. Rev. Toxicol., 43(3): 244–274.
Thompson, C.M., Kirman, C.R., Proctor, D.M., Haws, L.C., Suh, M., Hays, S.M., Hixon, J.G. and Harris, M.A. (2014). A chronic oral reference dose for hexavalent chromium–induced intestinal cancer. J. Appl. Toxicol., 34(5): 525–536.
Thompson, C.M., Young, R.R., Suh, M., Dinesdurage, H.R., Elbekai, R.H., Harris, M.A., Rohr, A.C. and Proctor, D.M. (2015a). Assessment of the mutagenic potential of Cr(VI) in the oral mucosa of Big Blue® transgenic F344 rats. Environ. Mol. Mutagen, 56: 621–628.
Thompson, C.M., Seiter, J., Chappell, M.A., Tappero, R.V., Proctor, D.M., Suh, M., Wolf, J.C., Haws, L.C., Vitale, R., Mittal, L., Kirman, C.R., Hays, S.M. and Harris, M.A. (2015b). Synchrotron-based imaging of chromium and gamma-H2AX immunostaining in the duodenum following repeated exposure to Cr(VI) in drinking water. Toxicol Sci., 143(1): 16–25.
Thompson, C.M., Wolf, J.C., Elbekai, R.H., Paranjpe, M.G., Seiter, J.M., Chappell, M.A., Tappero, R.V., Suh, M., Proctor, D.M., Bichteler, A., Haws, L.C. and Harris, M.A. (2015c). Duodenal crypt health following exposure to Cr(VI): micronucleus scoring, gamma-H2AX immunostaining, and synchrotron X-ray fluorescence microscopy. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 789–790: 61–66.
Toepfer, E.W., Mertz, W., Roginski, E.E. and Polansky, M.M. (1973). Chromium in foods in relation to biological activity. J. Agric. Food Chem., 21: 69–73 [cited in WHO, 1988].
Trivedi, B., Saxena, D.K., Murthy, R.C. and Chandra, S.V. (1989). Embryotoxicity and fetotoxicity of orally administered hexavalent chromium in mice. Reprod. Toxicol., 3: 275–278.
Trumbo, P.R. and Ellwood, K.C. (2006). Chromium picolinate intake and risk of type 2 diabetes: an evidence-based review by the UnitedStates Food and Drug Administration. Nutr. Rev., 64(8): 357–363.
UK Food Standards Agency (1999). 1997 Total Diet Study—Aluminium, arsenic, cadmium, chromium, copper, lead, mercury, nickel, selenium, tin and zinc. Food Standards Agency, London, UK(Report No. 191).
UK Ministry of Agriculture, Fisheries and Food (1985). Survey of aluminium, antimony, chromium, cobalt, indium, nickel, thallium and tin in food.15th report of the Steering Group on Food Surveillance, Working Party on the Monitoring of Foodstuffs for Heavy Metals. Her Majesty’s Stationery Office, London, UK [cited in WHO, 1996].
Ulmer, N.S. (1986). Effect of chlorine on chromium speciation in tap water. Water Engineering Research Laboratories,U.S. Environmental Protection Agency, Cincinnati, OH (EPA/600/M-86/015).
Urbano, A.M., Ferreira, L.M.R. and Alpoim, M.C. (2012). Molecular and cellular mechanisms of hexavalent chromium–induced lung cancer: an updated perspective. Curr. Drug Metab., 13: 284–305.
U.S. EPA (1984a). Health assessment document for chromium. U.S. Environmental Protection Agency, Research Triangle Park, NC (EPA 600/8-83-014F).
U.S. EPA (1984b). Health effects assessment for trivalent chromium. Office of Research and Development, Office of Health and Environmental Assessment, Environmental Criteria and Assessment Office, U.S. Environmental Protection Agency, Washington, DC.
U.S. EPA (1984c). Health effects assessment for hexavalent chromium.Office of Research and Development, Office of Health and Environmental Assessment, Environmental Criteria and Assessment Office, U.S. Environmental Protection Agency, Washington, DC (EPA/540/1-86/019).
U.S. EPA (1990). Noncarcinogenic effects of chromium: update to health assessment document. U.S. Environmental Protection Agency, Research Triangle Park, NC (EPA 600/8-87/048F).
U.S. EPA (1991). National Primary Drinking Water Regulations—synthetic organic chemicals and inorganic chemicals; monitoring for unregulated contaminants; National Primary Drinking Water Regulations implementation; National Secondary Drinking Water Regulations; final rule. Fed. Regist., 56(30): 3526.
U.S. EPA (1994a). Method 200.7 Revision 4.4. Determination of metals and trace elements in water and wastes by inductively coupled plasma–atomic emission spectrometry. Environment Monitoring Systems Laboratories, Office of Research and Development, U.S. Environmental Protection Agency, Cincinnati, Ohio.
U.S. EPA (1994b).Method 200.8 Revision 5.4. Determination of trace elements in waters and wastes by inductively coupled plasma–mass spectrometry. Environment Monitoring Systems Laboratories, Office of Research and Development, U.S. Environmental Protection Agency, Cincinnati, Ohio.
U.S. EPA (1994c).Method 200.9 Revision 2.2. Determination of trace elements by stabilized temperature graphite furnace atomic absorption. Environment Monitoring Systems Laboratories, Office of Research and Development, U.S. Environmental Protection Agency, Cincinnati, Ohio.
U.S. EPA (1994d). Method 218.6Revision 3.3. Determination of dissolved hexavalent chromium in drinking water, groundwater and industrial wastewater effluents by ion chromatography. U.S. Environmental Protection Agency, Washington, DC.
U.S. EPA (1998a). Toxicological review of hexavalent chromium (CAS No. 18540-29-9)—In support of summary information on the Integrated Risk Information System (IRIS). U.S. Environmental Protection Agency, Washington, DC. Available at: www.epa.gov/iris/toxreviews/0144tr.pdf
U.S. EPA (1998b). Integrated Risk Information System (IRIS)—Chromium (VI) (CASRN 18540-29-9). U.S. Environmental Protection Agency, Washington, DC. Available at: www.epa.gov/iris/subst/0144.htm
U.S. EPA (2002). Review of literature on removal of fluoride from drinking water, draft report. Office of Water, United States Environmental Protection Agency, Cincinnati, Ohio.
U.S. EPA (2003a). Occurrence estimation methodology and occurrence findings report for the six-year review of existing National Primary Drinking Water Regulations. Office of Water, U.S. Environmental Protection Agency, Washington, DC (EPA-815-R-03-006).
U.S. EPA (2003b). Method 200.5.Determination of trace elements in drinking water by axially viewed inductively coupled plasma–atomic emission spectrometry. National Exposure Research Laboratory, Office of Research and Development, U.S. Environmental Protection Agency, Cincinnati, Ohio (EPA 600/R-06/115).
U.S. EPA (2003c). Water treatment technology feasibility support document for chemical contaminants; in support of EPA six-year review of National Primary Drinking Water Regulations. Targeting and Analysis Branch, Standards and Risk Management Division, Office of Ground Water and Drinking Water, U.S. Environmental Protection Agency, Washington, DC (EPA 815-R-03-004)
U.S. EPA (2007). Draft framework for determining a mutagenic mode of action for carcinogenicity. U.S. Environmental Protection Agency, Washington, DC. Available at: www.epa.gov/osa/mmoaframework/.[As cited in Thompson et al., 2013].
U.S. EPA (2009). Analytical feasibility support document for the second six-year review of existing National Primary Drinking Water Regulations. U.S. Environmental Protection Agency, Washington, DC (EPA 815/B-09/003).
U.S. EPA (2010a). Technical basis for the lowest concentration minimum reporting level (LCMRL) calculator. Office of Water, U.S. Environmental Protection Agency, Washington, DC (EPA 815-R-11-001;http://water.epa.gov/scitech/drinkingwater/labcert/upload/LCMRLTechRpt.pdf).
U.S. EPA (2010b). Toxicological review of hexavalent chromium (CAS No. 18540-29-9): in support of summary information on the Integrated Risk Information System (IRIS)—Draft. U.S. Environmental Protection Agency, Washington, DC. Available at: http://cfpub.epa.gov/ncea/iris_drafts/recordisplay.cfm?deid=221433
U.S.EPA (2011a). Method 218.7. Determination of hexavalent chromium in drinking water by ion chromatography with post-column derivatization and UV–visible spectroscopic detection. Office of Water, U.S. Environmental Protection Agency, Washington, DC (EPA 815-R-11-005).
U.S. EPA (2011b). EPA’s recommendations for enhanced monitoring for hexavalent chromium (chromium-6) in drinking water. U.S. Environmental Protection Agency, Washington, DC. Available at: http://water.epa.gov/drink/info/chromium/guidance.cfm
U.S. EPA (2012a). Revisions of the unregulated contaminant monitoring regulation (UCMR 3) for public water systems; final rule. U.S. Environmental Protection Agency, Washington, DC. Fed. Regist.,77(85).
U.S. EPA (2012b). Drinking water treatability database. Office of Research and Development, U.S. Environmental Protection Agency, Washington, DC.Available at: http://iaspub.epa.gov/tdb/pages/general/home.do
U.S. EPA (2012c). UCMR3 laboratory approval requirements and information document. Version 2. Technical support center. Standards and risk management division. Office of groundwater and drinking water. U.S. Environmental Protection Agency, Cincinnati, Ohio 45268
U.S. EPA (2014a). Analytical methods approved for drinking water compliance monitoring of inorganic contaminants. U.S. Environmental Protection Agency, Washington, DC. Available at: http://water.epa.gov/scitech/drinkingwater/labcert/upload/methods_inorganic.pdf
U.S. EPA (2014b). EPA’s recommendations for enhanced monitoring for hexavalent chromium (Chromium-6) in drinking water. Available at: http://water.epa.gov/drink/info/chromium/guidance.cfm#three
Vaglenov, A., Nosko, M., Georgieva, R., Carbonell, E., Creus, A. and Marcos, R. (1999). Genotoxicity and radioresistance in electroplating workers exposed to chromium. Mutat. Res., 446(1): 23–34.
Vainshtein,M.,Kuschk,P., Mattush,J.,Vatsourina,A. and Wiessner,A.(2003). Model experiments on the microbial removal of chromium from contaminated groundwater. Water Res., 37:1401–1405.
Van Lierde, V., Chéry, C.C., Roche, N., Monstrey, S., Moens, L. and Vanhaecke, F. (2006). In vitro permeation of chromium species through porcine and human skin as determined by capillary electrophoresis–inductively coupled plasma–sector field mass spectrometry. Anal. Bioanal. Chem., 384(2): 378–384.
Vasudevan,S., Lakshmi,J. and Vanath,R. (2010). Electrochemical coagulation for chromium removal: process optimization, kinetics, isotherms and sludge characterization. Clean, 38(1): 9–16.
Vincent, J.B. and Stallings, D. (2007). Introduction: ahistory of chromium studies (1955–1995). In: Vincent, J.B. (ed.),The nutritional biochemistry of chromium(III). Elsevier, Amsterdam, Netherlands.pp. 1–42.
Wang, T., Jia, G., Zhang, J., Ma, Y., Feng, W., Liu, L., Zhang, N., Yan, L., Wang, X., Zhang, X., Liu, Z., Du, X. and Zhen, S. (2011a). Renal impairment caused by chronic occupational chromate exposure. Int. Arch. Occup. Environ. Health, 84(4): 393–401.
Wang, T.C., Jia, G., Zhang, J., Ma, Y.H., Liu, L.Z., Zhang, N., Feng, W.Y., Zhou, J.W., Song, Y.S., Yan, L. and Du, X.M. (2011b). Vitamin B12 and folate deficiency and elevated plasma total homocysteine in workers with chronic exposure to chromate. Occup. Environ. Med., 68(12): 870–875.
Wang, X.F., Lou, X.M., Shen, Y., Xing, M.L. and Xu, L.H. (2009). Apoptotic-related protein changes induced by hexavalent chromium in mice liver. Environ. Toxicol., 25(1): 77–82.
Wang,Y.-T., Chirwa,E. and Shen,H.(2000). Cr(VI) reduction in continuous-flow coculture bioreactor. J. Environ. Eng., 126(4):300–306.
Water Research Foundation (2013). Coachella Valley Water District treatment of hexavalent chromium. HexChrome 2013 Symposium. February 4, 2013. Sacramento, CA. Available at: www.waterrf.org/resources/expertsymposiums/Pages/hexchromesymposium2013.aspx
Welling, R., Beaumont, J.J., Petersen, S.J., Alexeeff, G.V. and Steinmaus, C. (2015). Chromium VI and stomach cancer: a meta-analysis of the current epidemiological evidence. Occup. Environ. Med., 72(2): 151–159.
Werfel, U., Langen, V. and Eickhoff, I. (1998). Elevated DNA single-strand breakage frequencies in lymphocytes of welders exposed to chromium and nickel. Carcinogenesis, 19(3): 413–418.
Whitby, L.M., Gaynor, J.D. and Maclean, A.J. (1978). Metals in soils of some agricultural watersheds in Ontario. Can. J. Soil Sci., 58: 325–330.
WHO (1988). Chromium. International Programme on Chemical Safety, World Health Organization, Geneva, Switzerland (Environmental Health Criteria 61). Available at: www.inchem.org/documents/ehc/ehc/ehc61.htm
WHO (1996). Chromium in drinking-water. Background document for development of WHO Guidelines for Drinking-water Quality. World Health Organization, Geneva, Switzerland. 8 pp. (WHO/SDE/WSH/03.04/04). Available at: www.who.int/water_sanitation_health/dwq/chemicals/chromium.pdf
Wiegand, H.J., Ottenwälder, H. and Bolt, H.M. (1984). The reduction of chromium(VI) to chromium (III) by glutathione: an intracellular redox pathway in the metabolism of the carcinogen chromate. Toxicology, 33: 341–348 [cited in O’Flaherty et al., 2001].
Wiegand, H.J., Ottenwälder, H. and Bolt, H.M. (1985). Fast uptake kinetics in vitro of 51Cr (VI) by red blood cells of man and rat. Arch. Toxicol., 57(1): 31–34.
Wiegand, H.J., Ottenwälder, H. and Bolt, H.M. (1988). Recent advances in biological monitoring of hexavalent chromium compounds. Sci. Total Environ., 71(3): 309–315.
Wilcox, A.J., Savitz, D.A. and Samet, J.M. (2008). A tale of two toxicants (lessons from Minamata and Liaoning). Epidemiology, 19(1): 1–2.
Williams, J.H. (1988). Chromium in sewage sludge applied to agricultural land. Office of Official Publications for the Commission of the European Communities, Brussels, Belgium.
Witt, K.L., Stout, M.D., Herbert, R.A., Travlos, G.S., Kissling, G.E., Collins, B.J. and Hooth, M.J. (2013). Mechanistic insights from the NTP studies of chromium. Toxicol. Pathol. 41(2): 326-42.
Wu, F.Y., Wu, W.Y., Kuo, H.W., Liu, C.S., Wang, R.Y. and Lai, J.S. (2001). Effect of genotoxic exposure to chromium among electroplating workers in Taiwan. Sci. Total Environ., 279(1–3): 21–28.
Xu, X.-R., Li, H.-B., Li, X.-Y. and Gu, J.-D. (2004). Reduction of hexavalent chromium byascorbic acid in aqueous solutions. Chemosphere, 57(7):609–613.
Yoon, J., Amy, G., Chung, J., Soha, J. and Yoon, Y. (2009). Removal of toxic ions (chromate, arsenate and perchlorate) using reverse osmosis, nanofiltration, and ultrafiltration membranes. Chemosphere, 77: 228–235.
Young, R.R., Thompson, C.M., Dinesdurage, H.R., Elbekai, R.H., Suh, M., Rohr, A.C. and Proctor, D.M. (2015). A robust method for assessing chemically induced mutagenic effects in the oral cavity of transgenic Big Blue® rats. Environ. Mol. Mutagen., 56: 629–636.
Yousef, M.I., El-Demerdash, F.M., Kamil, K.I. and Elaswad, F.A. (2006). Ameliorating effect of folic acid on chromium(VI)-induced changes in reproductive performance and seminal plasma biochemistry in male rabbits. Reprod. Toxicol., 21(3): 322–328.
Zahid, Z.R., Al-Hakkak, Z.S., Kadhimm, A.H.H., Elias, E.A. and Al-Jumaily, I.S. (1990). Comparative effects of trivalent and hexavalent chromium on spermatogenesis of the mouse. Toxicol. Environ. Chem., 25: 131–136 [cited in Environment Canada and Health Canada, 1994].
Zhang, J.D. and Li, X.L. (1987). Chromium pollution of soil and water in Jinzhou. Zhonghua Yu Fang Yi Xue Za Zhi, 21: 262–264 [cited in Beaumont et al., 2008].
Zhitkovich, A. (2011). Chromium in drinking water: sources, metabolism, and cancer risks. Chem. Res. Toxicol., 24(10): 1617–1629.
Ziaee, H., Daniel, J., Datta, A.K., Blunt, S. and McMinn, D.J. (2007). Transplacental transfer of cobalt and chromium in patients with metal-on-metal hip arthroplasty: a controlled study. J. Bone Joint Surg. Br., 89(3): 301–305.
Zimmer, R. (2014). Chromium analysis for MCL compliance. CA/NV AWWA Chromium Seminar Series, California, January.
Zotter, K. and Licsko, I. (1992). Removal of chromium (VI) and other heavy metals from groundwater and alkaline media. Water Sci. Technol., 26(1–2):207–216.
Appendix A: List of acronyms
- 8-OHdG
- 8-hydroxydeoxyguanosine
- 8-oxodG
- 8-oxodeoxyguanosine
- AI
- adequate intake
- ANSI
- American National Standards Institute
- ATSDR
- Agency for Toxic Substances and Disease Registry
- BAT
- best available technology
- BMD
- benchmark dose
- BMDL
- lower 95% confidence limit on the benchmark dose
- BMDL01
- lower 95% confidence limit on the benchmark dose for a 1% response
- BMDL05
- lower 95% confidence limit on the benchmark dose for a 5% response
- BMDL10
- lower 95% confidence limit on the benchmark dose for a 10% response
- BMR
- benchmark response
- BV
- bed volume
- bw
- body weight
- CI
- confidence interval
- CPM
- chromium picolinate monohydrate
- DDW
- distilled, deionized water
- DL
- detection limit
- DNA
- deoxyribonucleic acid
- EBCT
- empty bed contact time
- EPA
- Environmental Protection Agency (U.S.)
- FBR
- fluidized bed reactor
- GAC
- granular activated carbon
- GFAA
- graphite furnace atomic absorption
- gfd
- gallons per square foot per day
- GI
- gastrointestinal
- gpm
- gallons per minute
- GSH
- glutathione
- GSSG
- glutathione disulphide
- HBV
- health-based value
- HED
- human equivalent dose
- HPLC
- high-performance liquid chromatography
- HRT
- hydraulic retention time
- IARC
- International Agency for Research on Cancer
- ICP-AES
- inductively coupled plasma–atomic emission spectroscopy
- ICP-MS
- inductively coupled plasma–mass spectrometry
- LD50
- median lethal dose
- LOAEL
- lowest-observed-adverse-effect level
- MAC
- maximum acceptable concentration
- MCH
- mean cell hemoglobin
- MCV
- mean cell volume
- MDL
- method detection limit
- MF
- microfiltration
- MOA
- mode of action
- MRL
- minimum reporting level
- NADPH
- nicotinamide adenine dinucleotide phosphate (reduced)
- NF
- nanofiltration
- NOAEL
- no-observed-adverse-effect level
- NOM
- natural organic matter
- NSF
- NSF International
- NTP
- National Toxicology Program (U.S.)
- NTU
- nephelometric turbidity unit
- OECD
- Organisation for Economic Co-operation and Development
- PBPK
- physiologically based pharmacokinetic
- PND
- postnatal day
- PQL
- practical quantitation level
- RCF
- reduction/coagulation/filtration
- RDL
- reportable detection limit
- RO
- reverse osmosis
- ROS
- reactive oxygen species
- SBA
- strong base anion exchange
- SCC
- Standards Council of Canada
- SDD
- sodium dichromate dihydrate
- SM
- Standard Method
- SMR
- standardized mortality ratio
- TDI
- tolerable daily intake
- TDS
- total dissolved solids
- TOC
- total organic carbon
- UCMR3
- third Unregulated Contaminant Monitoring Rule
- UF
- ultrafiltration
- WBA
- weak base anion exchange