Determination of triacetin in whole tobacco: T-311
1 Scope of application
1.1
This method is used to determine the amount of triacetin added to whole tobacco as a flavorant by Gas Chromatography with Flame Ionization Detection.
1.2
The method is designed to be used as a routine analysis without the need for derivatization.
1.3
Potential interference of vanilla could have an impact on the quantification of the triacetin for other matrices (e.g. cigars, smokeless tobacco) than the Canadian cigarette tobacco. All triacetin values above the method limit of quantification (LOQ) should be confirmed by gas chromatography/mass spectrometer (GC/MS).
2 Normative references
2.1
Health Canada Official Method T-115. Determination of Tar, Water, Nicotine and Carbon Monoxide in Mainstream Tobacco Smoke, 2016.
2.2
Health Canada Official Method T-402. Preparation of Sample for Testing of Cigarettes, Tobacco Sticks, Cigarette Tobacco, Cigars, Little Cigars, Kreteks, Bidis, Leaf, Pipe and Smokeless Tobacco, 2016.
2.3
International Organization for Standardization, ISO 8243 Cigarettes - Sampling. 2013.
2.4
International Organization for Standardization, ISO 15592-1 Fine-Cut tobacco and smoking articles made from it - Methods of sampling, conditioning and analysis - Part 1: Sampling. 2001.
2.5
AOAC INTERNATIONAL, AOAC Official Method 966.02. Moisture in Tobacco, Gravimetric Method. Official Methods of Analysis of AOAC INTERNATIONAL 20th Ed., 2016.
3 Definitions
3.1
Refer to T-301 for definitions of terms used in this document.
4 Method summary
4.1
Tobacco is extracted with methanol on a wrist-action shaker using dimethyl phthalate as an internal standard (ISTD).
4.2
The methanol extract is diluted with dilute acetic acid solution to remove compounds that elute near triacetin.
4.3
The triacetin is extracted into dichloromethane as methanol is a poor solvent for injection into a gas chromatograph (GC).
4.4
The combined extract is then rotary-evaporated to near dryness before dilution to volume.
4.5
The sample is then analyzed by GC using a split mode of injection with a flame ionization detector (FID).
4.6
Quantification is achieved by internal standard calibration (dimethyl phthalate), where the relative response of the samples is compared against a multi-point calibration.
Warning: The testing and evaluation of certain products against this test method may require the use of materials and/or equipment that are potentially hazardous and this document does not purport to address all the safety aspects associated with its use. Anyone using this test method has the responsibility to consult with the appropriate authorities and to establish health and safety practices in conjunction with all existing applicable regulatory requirements prior to its use.
5 Apparatus and equipment
5.1
Erlenmeyer flasks, 250 mL.
5.2
Volumetric flasks, 10, 100, 2000 mL.
5.3
Separatory funnels, 500 mL or equivalent.
5.4
Graduated cylinder, 50 mL.
5.5
Rotary evaporator, Buchi R24 Rotovap or equivalent.
5.6
Batch processor, Robot Coupe RS1-2V or equivalent.
5.7
Micropipettes, 50–500 μL or equivalent.
5.8
Pipettes, class A, 1.0 mL.
5.9
Round bottom flasks with ground glass joints, 250 mL.
5.10
Gas chromatograph with split/splitless injector and flame ionization detector (FID) or equivalent.
5.11
Fused silica capillary column, OV101 or equivalent.
5.12
GC/MS system with an autosampler, a split/splitless injector and ion trap detector or equivalent.
5.13
ZB-1 capillary column, 15 m × 0.25 mm × 0.25 µm or equivalent.
5.14
Analytical balance reading to at least 4 decimal places.
5.15
Autosampler vials with screw caps.
5.16
Top-loading balance.
5.17
Glass funnels.
5.18
Glass wool.
5.19
Glass Pasteur pipettes, disposable.
5.20
Glass beads.
5.21
Parafilm® or equivalent.
5.22
Rubber bulbs.
5.23
Septa, 8 mm Teflon-faced, 60 mil.
5.24
Filter paper.
6 Reagents and supplies
6.1
All reagents shall be at least analytical reagent grade.
Note: Wherever possible, reagents are identified by their Chemical Abstract Service [CAS] registry numbers in square brackets.
6.2
Acetic acid, glacial - [64-19-7] chromatography grade or equivalent.
6.3
Dichloromethane - [75-09-2] Distilled-In-Glass (DIG).
6.4
Dimethyl phthalate (liquid) (DMP) - [131-11-3].
6.5
Extra-dry air (FID) - [132259-10-0].
6.6
Helium (Carrier Gas) UHP - [7440-59-7].
6.7
Hydrogen (FID) UHP - [1333-74-0].
6.8
Methanol - [67-56-1] DIG.
6.9
Nitrogen (Makeup) UHP - [7727-37-9].
6.10
Triacetin (liquid) - [102-76-1] > 99 % purity.
6.11
Water, Type I (as outlined in ASTM D1193, Table 1: Processes for Reagent Water Production, Note A).
7 Preparation of glassware
7.1
Clean and dry glassware in a manner to ensure that contamination from residues on glassware does not occur.
8 Preparation of solutions
8.1
0.1N Acetic Clean-up Solution
8.1.1
Add approximately 500 mL Type I water to a clean 1 L volumetric flask and place on a top-loading balance.
8.1.2
Carefully add 6.24 g of 99.7 % glacial acetic acid.
8.1.3
Add Type I water to below the mark, mix and allow to come to room temperature.
8.1.4
Make volume to the mark with Type I water.
9 Preparation of standards
9.1
Primary Stock Triacetin Standard
9.1.1
Weigh 100 mg of the liquid triacetin into a 100 mL volumetric flask.
9.1.2
Dilute to volume with dichloromethane.
9.2
Primary Stock Dimethyl Phthalate Standard
9.2.1
Weigh 100 mg of the dimethyl phthalate into a 100 mL volumetric flask.
9.2.2
Dilute to volume with dichloromethane.
9.3
Triacetin Calibration Standards
9.3.1
Take appropriate volumes (0.100-2.0 mL) of the triacetin primary stock solution.
9.3.2
Add 1 mL of dimethyl phthalate primary stock solution.
9.3.3
Dilute to 10 mL with dichloromethane to give calibration standards with approximate triacetin concentrations in the ranges detailed below.
9.3.4
Calibration Standards
| Standard No. |
Volume of Primary Triacetin Std. (mL) |
Final Volume (mL) |
Triacetin [μg/mL] |
|---|---|---|---|
| 1 | 0.10 | 10 | 10 |
| 2 | 0.25 | 10 | 25 |
| 3 | 0.50 | 10 | 50 |
| 4 | 1.00 | 10 | 100 |
| 5 | 2.00 | 10 | 200 |
Note: The concentration of triacetin will vary depending on the exact concentration of primary stock prepared.
Note: Additional standards may have to be prepared to cover the range of anticipated responses for test samples.
9.3.5
Transfer to 1.5 mL amber autosampler vials. Rinse the vial first and then fill to minimize head space.
9.3.6
Place vials in a vial file and store at 4 °C, protected from light, until instrument analysis is performed.
9.3.7
Prepare triacetin calibration standards fresh every 5 days.
10 Sampling
10.1
The sampling of cigarettes for the purpose of testing shall be in accord with ISO 8243.
10.2
The sampling of kreteks, little cigars, bidis, tobacco sticks for the purpose of testing shall be in accord with ISO 8243, but modified such that the term "cigarette" is substituted with "kreteks", "little cigars", "bidis" or "tobacco sticks", whereby the term "carton" is equivalent to 200 units
10.3
The sampling of cigars for the purpose of testing shall be in accord ISO 8243, but modified such that the term "cigarette" is substituted with "cigar", whereby 200 units of cigarette is equivalent to 200 grams of cigar.
10.4
The sampling of cigarette tobacco for the purpose of testing shall be in accord with ISO 15592-1.
10.5
The sampling of leaf tobacco, pipe tobacco or smokeless tobacco shall be in accord with ISO 15592-1 but modified such that the term "fine-cut" is substituted with "leaf tobacco", "pipe tobacco" or "smokeless tobacco".
11 Tobacco product preparation
11.1
Preparation of Test Sample
11.1.1
Remove the tobacco required for analysis from its original package and inspect for extraneous materials.
11.1.2
The preparation of tobacco products for the purpose of testing shall be as specified in T-402.
12 Sample preparation
12.1
Accurately weigh 2 g of test sample into a 125 mL (or 250 mL) Erlenmeyer flask.
12.2
Add 49 mL of methanol to the Erlenmeyer flask.
12.3
Add 1 mL of dimethyl phthalate (ISTD), stopper and shake for 30 minutes on a wrist-action shaker.
12.4
Transfer the extract to a 1 L extraction flask through a funnel equipped with a plug of glass wool.
12.5
Add approximately 1 L of 0.1N acetic acid to the extraction flask of the Labline extractor.
12.6
Add 50 mL of dichloromethane to the Labline extractor.
12.7
Shake the separatory funnel and then drain off the dichloromethane through anhydrous sodium sulphate retained by a filter paper (to avoid particles that might clog the injector).
12.8
Repeat the extraction procedure (12.6 to 12.7) a total of 3 times.
12.9
Evaporate the 150 mL combined dichloromethane extract at 30 °C on a rotary evaporator to approximately 5 mL.
12.10
Quantitatively transfer the residual liquid to a 10 mL volumetric flask and dilute to volume with dichloromethane.
12.11
Using a disposable Pasteur pipette, transfer the sample to a 1.5 mL amber autosampler vial.
12.12
Place the vial in a vial file and store at 4 °C, protected from light, until instrument analysis is performed.
13 Sample analysis
13.1
Gas Chromatograph Configuration
- Column: HP–101 fused silica capillary column
- Detector: Flame Ionization (FID)
- Carrier: He at 40.0 psi, flow = 1.0 mL/minute at 70 °C
- Injector: 270 °C
- Detector: 300 °C
- Column Oven Temperature Profile:
- Start Temp.: 70 °C, hold for 2.0 minutes
- Rate: 5.0 °C/minute to 135 °C, hold for 9.5 minutes
- Rate: 7.0 °C/minute to 270 °C, hold for 5.22 minutes
- Total Run Time: 45.00 minutes
- Injection volume: 1.0 μL (splitless injection)
Note: Adjustment to the operating conditions may be required, depending on instrument and column conditions as well as resolution of the analyte peak.
13.2
Analysis
13.2.1
The total time (sample generation plus analysis time) must not exceed 48 hours (ideally 24 hours) at room temperature.
13.2.2
Inject 1 μL of each sample vial onto the GC column and analyze.
14 Calculations
14.1
Construct a Calibration Curve
14.1.1
Inject 1 μL of each calibration standard onto the GC column and analyze as per the chromatographic conditions specified.
14.1.2
Prepare a calibration curve of triacetin by plotting the concentration of the standards versus their respective peak areas relative to the inverse of the internal standard (dimethyl phthalate) peak area and concentration.
14.1.3
Determine the response factor from the calibration curve.
14.2
Sample Quantification
14.2.1
Quantify the amount of the triacetin in tobacco samples by the internal standard method. (See appendix 1, figure 3.)
14.2.2
The identification of peaks is by comparison of retention times with standards and the spiking of samples.
14.3
Determination of Triacetin Deliveries in [μg/g]
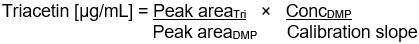
Figure 14-3-a: Text description
Triacetin [μg/mL] =
Peak areaTri
divided by
Peak areaDMP
×
ConcDMP
divided by
Calibration slope
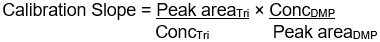
Figure 14-3-b: Text description
Calibration Slope =
Peak areaTri
divided by
ConcTri
×
ConcDMP
divided by
Peak areaDMP
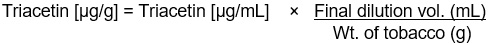
Figure 14-3-c: Text description
Triacetin [μg/g] =
Triacetin [μg/mL]
×
Final dilution vol. (mL)
divided by
Wt. of tobacco (g)
14.4
By entering the correct multiplier (final volume (10 mL) to which the original sample is diluted in mL) and divisor (the original sample weight in g) the concentration of triacetin is automatically calculated in μg/g.
14.5
To convert this concentration to a percentage, divide the μg/g result by 10,000.
14.6
All results are expressed on an 'as received' basis. These may be expressed on a 'dry matter' basis using the appropriate moisture result determined by AOAC Official Method 966.02.
15 Quality control
15.1
For typical chromatograms, see appendix 1, figure 1, figure 2 and figure 3.
15.2
Typical Control Parameters
Note: If the control measurements are outside the tolerance limits of the expected values, appropriate investigation and action must be taken.
15.2.1
Laboratory Reagent Blank (LRB)
To detect potential contamination during the sample preparation and analysis processes, include a laboratory reagent blank (LRB). The LRB consists of all reagents and materials used in performing the analysis on test samples and is analyzed as a test sample.
15.2.2
Laboratory Fortified Blank (LFB)
To detect potential loss of analyte during the sample preparation and analysis processes, include a laboratory fortified blank (LFB). The LFB consists of all reagents and materials used in performing the analysis on test samples plus fortification with a known concentration of at least one of the analytes of interest. The level of fortification should reflect the range of typical results for that sample. The LFB is then analyzed as a test sample.
15.2.3
Laboratory Fortified Matrix (LFM)
To detect potential matrix interferences, include a laboratory fortified matrix (LFM). During the sample preparation and/or analysis processes, divide a test sample and fortify an aliquot with at least one of the analytes of interest in known concentration. The level of fortification should reflect the range of typical results for that sample. The LFM is then analyzed as a test sample.
15.2.4
Laboratory Control Sample
To assess the overall performance of an analysis, a control sample is analyzed. The results of the control sample should be compared, using appropriate statistical techniques, to 'expected values' generated by the laboratory or, if none exist, to values found in literature. This provides information to the laboratory, on test accuracy and precision.
15.2.5
Standard as Sample
To assess the stability of the analytical system, a standard is analyzed as a sample. The results of this standard should be compared, using appropriate statistical techniques, to expected concentrations.
15.3
Recoveries and Levels of Contamination
15.3.1
Typical LRB values average 0.1 ± 0.7 μg/g.
15.3.2
Typical LFB and LFM recoveries fall in the range 85-105 % recovery.
15.4
Limit of Detection (LOD) and Limit of Quantification (LOQ)
15.4.1
The LOD can be determined as 3 times the standard deviation of results obtained by analyzing the lowest standard level a minimum of 10 times over several days.
15.4.1.1
A typical value for LOD, determined by the above method, is 7.3 μg/g as analyzed.
15.4.2
The LOQ can be determined as 10 times the standard deviation of results obtained by analyzing the lowest standard level a minimum of 10 times over several days.
15.4.2.1
A typical value for LOQ, determined by the above method, is 24 μg/g as analyzed.
15.5
Stability of Reagents and Supplies
15.5.1
Prepare all primary stock triacetin standards fresh weekly.
15.5.2
Prepare all work standards and extraction solvents fresh weekly.
15.5.3
The total time from adding the methanol to the sample until injection should not exceed 48 hours (ideally 24 hours) at room temperature.
16 References
16.1
Bohlander, P. J.; McKee, J. L. and Bowermaster, J. TCRC #3263. Celanese Hoechst. Rapid GC method for quantifying plasticizers in cigarette filter rods. 1978.
16.2
Frank, M. S. and Lin, O. C. TCRC # 4147. Brown & Williamson. The analysis of propylene glycol, menthol, triacetin, glycerine and triethyleneglycol diacetate in cigarettes. 1987.
16.3
Gilleland, H. L.; Kimel M. B. and Rush, K. L. TCRC #3148. Reynolds Tobacco. Automated GC method for the determination of plasticizers in cigarette filter rods. 1977.
16.4
McMurtrie, A. and Canon, A. B. TCRC # 3339. Brown & Williamson. Rapid quantitative determination of menthol/triacetin in smoke. 1979.
16.5
ASTM International, ASTM Standard D1193-06(2011). Standard Specifications for Reagent Water.
Appendix 1
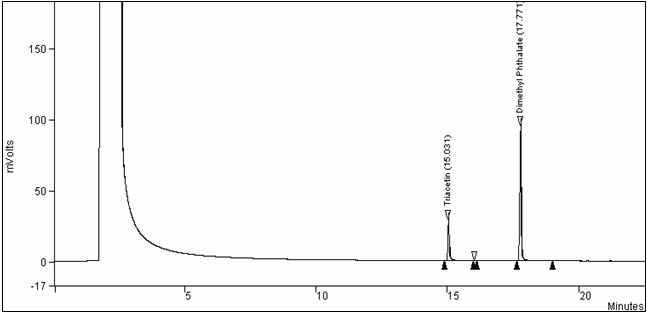
Figure 1. Chromatogram of a typical triacetin calibration standard: Text description
This figure is a chromatogram of a typical triacetin calibration standard. The voltage was measured in mVolts over a period of time, in minutes.
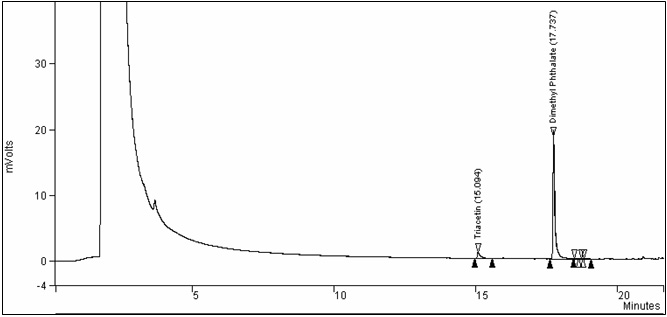
Figure 2. Chromatogram of the analysis of whole tobacco (Control): Text description
This figure is a chromatogram of the analysis of whole tobacco (Control). The voltage was measured in mVolts over a period of time, in minutes.
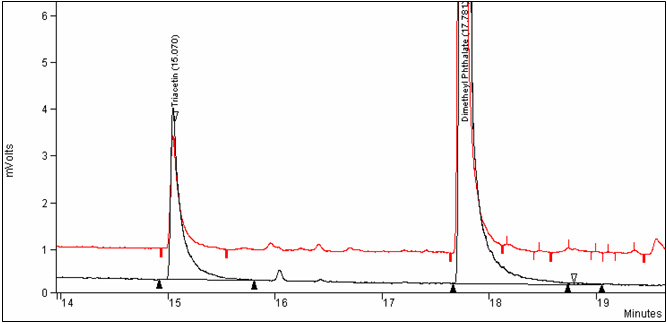
Figure 3. Overlay chromatograms of control whole tobacco and triacetin calibration standard: Text description
This figure is an overlay of chromatograms of control whole tobacco and triacetin calibration standard.