2020-21 Departmental Plan: Patented Medicine Prices Review Board
The Honourable Patty Hajdu
Minister of Health
ISSN: 2371-7807
Table of contents
- From the Chairperson
- Plans at a glance
- Core responsibilities: planned results and resources, and key risks
- Planned results for Internal Services
- Spending and human resources
- Corporate information
- Supporting information on the program Inventory
- Supplementary information tables
- Federal tax expenditures
- Organizational contact information
- Appendix: definitions
- Endnotes
From the Chairperson
The Patented Medicine Prices Review Board (PMPRB) is an independent quasi-judicial body established by Parliament in 1987 under the Patent Act (the “Act”). The PMPRB is a consumer protection agency with a dual regulatory and reporting mandate. Its regulatory mandate is to ensure that the prices of patented medicines sold in Canada are not excessive. Its reporting mandate is to provide stakeholders with pharmaceutical trends information to help them make informed choices.
In 2020-21 the PMPRB will formalize its long awaited move to a modern, progressive and risk-based approach to regulating the ceiling prices of patented medicines. This will close the chapter on the PMPRB’s previous three year strategic plan and cap off a period of unprecedented change in the organizations’ history. Having transformed our legal framework, office space and corporate culture over the course of the previous planning cycle, we intend to take somewhat of a a lighter touch to 2020-23.
Our immediate focus for 2020-21 is to finalize and implement the new pricing Guidelines and to support the Government, in whatever capacity is considered appropriate, in moving forward with the other foundational pieces of national pharmacare. With respect to the former, we will also ensure that a comprehensive monitoring and evaluation plan is in place so that we can assess as quickly as practicable whether the new regulatory framework is working as intended.
Like any government organization, the PMPRB’s foremost asset is its people. Priorities may change over time but reaching them is only possible if employees continue to feel engaged in their work and workplace. Although the present document is focused on operational priorities relating to its Core Responsibility, employee engagement is an enduring goal of the PMPRB and a permanent part of every strategic planning cycle.
The modernization of the PMPRB’s regulatory framework will almost certainly see it occupy a more prominent place in the Canadian pharmaceutical pricing and reimbursement system. I would like to express my appreciation for the steadfast support of our federal, provincial and territorial health partners and their abiding belief in our potential throughout this process. I would also like to offer my reassurance to our stakeholders, and those who may be concerned about the impact a more prominent PMPRB will have on access to new and innovative medicines in Canada, that we will continue to hold the welfare of consumers foremost in our minds and the sustainability of the Canadian health system at heart.
Dr. Mitchell Levine
Chairperson
Plans at a glance
Priority 1 – Implement new pricing framework and begin evaluating its impact
This priority arises from the fact that the PMPRB must finalize its Guidelines and a have a plan in place to assess the impact of the new framework prior to its coming into force in July 1, 2020. Priority 1 objectives include:
- Completing consultations with stakeholders and finalizing the new Guidelines;
- Developing and implementing an outreach strategy so that patentees understand how to comply with the new Guidelines;
- Establishing a comprehensive Framework Monitoring and Evaluation Plan (FMEP) to assess whether the new pricing framework is operating as intended.
The implementation of the PMPRB’s new pricing framework is the final step in a multi-year effort to give effect to the Government of Canada’s commitment to make prescription drugs more affordable for Canadians. With the coming into force of the new pricing framework in July 2020, the Government will be in a position to move forward with other foundational pieces that are believed necessary for the eventual establishment of a national pharmacare program.
Priority 2 –Support the Government’s high level priorities for the future of pharmaceutical management in Canada.
This priority reflects ongoing work to better align and integrate the roles, responsibilities and processes of the various participants in the pan-Canadian pharmaceutical pricing and reimbursement system and to implement national universal pharmacare, including the potential establishment of a Canadian drug agency, a national formulary and a rare disease drug strategy.
Priority 2 objectives include:
- Working with F/P/T health partners to align and optimize our respective processes in the context of the new framework and other recent or ongoing reforms that impact pricing and reimbursement.
- Providing analytical support and expertise to health partners, as appropriate, in efforts to advance policy work relating to the foundational elements of national pharmacare and other pan-Canadian initiatives to improve the pricing and reimbursement of pharmaceuticals in Canada.
In the course of carrying out its regulatory and reporting mandate, the PMPRB has developed considerable policy and analytical capacity and is a frequently used as a resource to support broader efforts by its federal health portfolio and pan-Canadian partners to foster a modern and sustainable health system. At a time of unprecedented change in the Canadian pharmaceutical ecosystem, the PMPRB will continue to leverage its resources and expertise to optimize its ability to protect consumers from excessive prices and maximize its value proposition to its F/P/T health partners and the health system as a whole.
For more information on the PMPRB’s plans, priorities and planned results, see the “Core responsibilities: planned results and resources, and key risks” section of this report.
Core responsibilities: planned results and resources, and key risks
This section contains detailed information on the department’s planned results and resources for each of its core responsibilities. It also contains information on key risks related to achieving those results.
Regulate Patented Medicine Prices
Description
The Patented Medicine Prices Review Board (PMPRB) regulates the prices of patented medicines by setting non-excessive price ceilings and taking enforcement action before the Board in the event of non-compliance.
Planning highlights
In August 2019, Treasury Board approved Health Canada’s amendments to the Patented Medicines Regulations (Regulations). Passage of the amendments was necessary to give effect to the Government’s commitment to improve the affordability of prescription medicines by making the following three changes to the way in which the PMPRB sets ceiling prices for patented medicines:
- Having the PMPRB compare Canadian list prices to list prices in countries that are more like Canada from a health system standpoint (i.e, removing Switzerland and the US from the group and adding countries with similar consumer protection priorities, economic output and access to medicines as Canada)
- Introducing factors beyond just domestic and international list prices that the PMPRB can consider in determining whether the Canadian price of a patented medicine is excessive (i.e, pharmacoeconomic value, market size and GDP/GDP per capita [Gross Domestic Product])
- Providing the PMPRB with information it needs to calculate the true price pharmaceutical companies are charging public and private payers (i.e., net of confidential rebates)
Significant changes to the PMPRB’s pricing Guidelines are necessary in order to implement the Regulations. In November 2019, the PMPRB published new Draft GuidelinesFootnote i and is currently seeking feedback on them from stakeholders and interested members of the public. The Guidelines must be finalized prior to the July 2020 coming into force date of the Regulations.
Once the new Guidelines have been finalized, the PMPRB will develop and implement an outreach strategy that will ensure patentees have the information they need to comply with the requirements of the new pricing framework. The PMPRB will offer information sessions, webinars and bilateral meetings with individual patentees as part of its outreach efforts.
While the changes to the Regulations and Guidelines do not alter the PMPRB’s regulatory mandate, they do introduce and address important new factors for it to consider in determining where to draw the line between an excessive and non-excessive price. Like any significant set of changes to a regulatory regime, there is always the potential for unintended consequences in the early days following coming into force. The PMPRB will develop and implement a comprehensive Framework Monitoring and Evaluation plan (FMEP) to assess, on an ongoing basis, the application and impact of the new regime in order to identify the need for any corresponding adjustments.
This is a time of unprecedented change in the pan-Canadian pharmaceutical and pricing reimbursement ecosystem in Canada. The past several years have seen a sustained effort on the part of the PMPRB and its health partners to better align and integrate their respective roles, responsibilities and processes in order to ensure a more sustainable Canadian health system. In addition, Budget 2019 and the Minister’s mandate letter identify a number of important commitments for the Govermment of Canada to pursue on the road to implementing national universal pharmacare.
In the coming year, the PMPRB will continue to host regular working group meetings with its health partners in an effort to sort through the various adjustments in our respective processes required to support the new pricing regime. It will also, as appropriate, provide continued analytical support and expertise to advance policy work relating to the foundational elements of national pharmacare and other pan-Canadian initiatives to improve the pricing and reimbursement of pharmaceuticals in Canada.
Experimentation
As part of its ongoing consultation process on the Draft Guidelines, the PMPRB has struck a working group of federal and provincial health partners to look at case studies and engage in modeling exercises to ensure that the new pricing regime functions as intended. In addition, as mentioned above, prior to the coming into force of the new regime, the PMPRB will put in place a comprehensive FMEP to assess the impact of the new regime on prices, access to medicines, research and development (R&D) and other important metrics for evaluating its effectiveness. The PMPRB will report its findings under the FMEP on a regular basis.
Key risk(s)
The PMPRB has identified four potential risks to the achievement of results for its Core Responsibility. The first such risk is that patentees may threaten to delay bringing new medicines to Canada or to bypass it entirely to discourage other countries that may be contemplating similarly intentioned drug pricing reforms. This is a common tactic used by the pharmaceutical industry internationally in seeking to defeat or dilute policy initiatives perceived to be inimical to their interests. For example, when Germany introduced much stricter price controls for new prescription drugs through its AMNOG legislation in 2011, a number of pharmaceutical companies announced that they would be delaying or pulling their products from the German market, citing the attendant uncertainty of the pending changes on drug prices. However, in the first four years the new rules were in effect, 124 new medicines completed the assessment process and launched in Germany and only 13 were withdrawn from the market due to “no additional benefit” ratings or failed price negotiations. The German government credits AMNOG for over €2 billion in annual savings to its health care system and Germany remains a top tier country in terms of new medicine launches.
In consulting on its new Guidelines, the PMPRB has sought to reassure its stakeholders that it intends to use its new regulatory tools responsibly and in a manner that reflects its role as a ceiling price regulator, not a price setter. As before, the viability of the regime administered by the PMPRB depends on patentees complying voluntarily with the Guidelines in the majority of cases. For this to occur, the ceiling prices that result from the PMPRB’s application of the new schedule of comparator countries and excessive pricing factors in the Regulations must be reasonable from a profitability standpoint and consistent with the continuing imperative of providing Canadian patients with timely access to effective new health technologies at a price they can afford. The PMPRB is working closely with its stakeholders and health partners to ensure that this is the effective result of its ongoing consultations on the new Guidelines. However, given that the inevitable end result will still be lower ceiling prices for many patented medicines, it is unlikely that any amount of reassurance will persuade industry to renounce its public opposition to the reforms.
To ensure that the new regulatory framework does not have unintended consequences in terms of its impact on the market entry of important new and innovative medicines, once the Guidelines are finalized the PMPRB will convene experts to help it develop a comprehensive plan for monitoring and evaluating their effect on such things as price, access and research and development in Canada, so that corrective action can be taken quickly if warranted.
The second risk is that implementation of the new regulatory framework may cause confusion for patentees about what data to file and how to transition between the old and the new regulatory regimes. To mitigate this risk, the PMPRB will make every effort to assist patentees in understanding the new framework and its application. In addition, under subsection 98(4) of the Act, the Board may provide advance guidance to a patentee on its price if there is sufficient information to do so.
A third risk is that compliance with the PMPRB’s new Guidelines may go down initially as patentees test the boundaries of the new regime, resulting in more investigations and hearings into prices of patented medicines. Through Budget 2017, the PMPRB received additional funding to ensure it would have sufficient resources to implement the new framework and contend with any corresponding increase in contested pricing matters. Completing construction of the PMPRB’s own dedicated hearing room will ensure that it has the capacity to accommodate multiple parallel hearings.
A fourth and final risk is that patentees may challenge the new Regulations and/or Guidelines in court and seek to have them quashed or overturned. However, this risk is considered to be an unavoidable consequence of any reforms to the PMPRB’s regulatory framework to make patented prescription medicines more affordable. The PMPRB is working with the federal Attorney General to ensure that any such challenges are properly defended.
| Departmental result | Departmental result indicator | Target | Date to achieve target | 2016-17 actual result | 2017–18 actual result | 2018-19 actual result |
|---|---|---|---|---|---|---|
| Affordable patented medicine prices | % of patented medicine prices in Canada are below the median price of the PMPRB’s comparator countries | 50% | March 31, 2021 | 58% | 56.4% | 57.1%(a) |
| % of patented medicine prices in Canada within the thresholds set out in the Guidelines | 95% | March 31, 2021 | 92.3% | 91.0% | 90.5%(b) | |
|
(a) The 57.1% of patented medicine prices in Canada reported as being below the median international price includes a significant number of patented medicines being sold in fewer than five countries and therefore are not being compared to the actual median international price. Of the 1,399 patented medicines sold in Canada in 2018, only 830 were sold in five or more countries. Of this 830, only 395 patented medicines (48%) had a Canadian price below the median price. This is a significant difference from the reported 57.1%. (b) As of March 31, 2019, 51 patented medicines were still under review, 128 were under investigation, two were the subject of a hearing and one was subject to a Stay Order. |
||||||
| 2020–21 budgetary spending (as indicated in Main Estimates) | 2020–21 planned spending | 2021–22 planned spending | 2022–23 planned spending |
|---|---|---|---|
| 14,728,961 | 14,728,961 | 15,755,364 | 13,807,960 |
The PMPRB’s funding includes a Special Purpose Allotment (SPA) to conduct Public Hearings. The SPA can only be used to cover the costs of public hearings, such as external legal counsel and expert witnesses, etc. Any unspent amount is returned to the Consolidated Revenue Fund.
For purposes of forecasting Planned Spending for 2020-21 and future years, the PMPRB assumes that the entire SPA funding for hearings will be spent. This is because these expenditures are dependent on the number of hearings, and the length and complexity of the hearings held, which are difficult to predict. The amount of the SPA for 2020-21 is $5,257,786; 2021-22 is $6,206,486; and, for 2022-23 and beyond the amount of the SPA is $4,463,361. This is the reason for declined planned spending in 2022-23.
Planned human resources for Regulate Patented Medicine Prices
| 2020–21 planned full-time equivalents | 2021–22 planned full-time equivalents | 2022–23 planned full-time equivalents |
|---|---|---|
| 65 | 65 | 65 |
Financial, human resources and performance information for the PMPRB’s Program Inventory is available in the GC InfoBase Footnote ii.
Planned results for Internal Services
Description
Internal Services are those groups of related activities and resources that the federal government considers to be services in support of Programs and/or required to meet corporate obligations of an organization. Internal Services refers to the activities and resources of the 10 distinct services that support Program delivery in the organization, regardless of the Internal Services delivery model in a department. These services are:
- Management and Oversight Services
- Communications Services
- Legal Services
- Human Resources Management Services
- Financial Management Services
- Information Management Services
- Information Technology Services
- Real Property Management Services
- Materiel Management Services
- Acquisition Management Services
Planning highlights
The PMPRB recently completed a refit of its existing workspace to Workplace 2.0 Fit-up Standards. In 2020-21 working in collaboration with Public Services and Procurement Canada, the PMPRB will complete work on the construction of its own dedicated hearing room. The PMPRB has a detailed plan for the hearing room construction, which includes the ability to quickly take corrective action if unforeseen issues arise that have the potential to delay or impair the successful completion of the project.
In 2020-21, the PMPRB will complete implementation of the Automatic Classification and Metadata Enhancements (ACME) Project to reduce the manual information end-users are required to input into the Electronic Document and Records Management System (EDRMS). The ACME will automate processes such as: classification; metadata generation and tagging; and, information governance. It is intended to improve user experience by being more user-friendly and facilitating e-discovery and search ability.
The PMPRB’s Compliance Information Management System (“CIMS”) is a web-based application used to review and analyze data filed by patentees, and assess patentees’ compliance. In 2020-21, the PMPRB will make a number of enhancements to CIMS so that it can accept and process the additional information patentees must provide under the new Regulations and Guidelines. These enhancements include updating price tests to conduct new medicine reviews, modernizing the online filling tool and upgrading several data management components of the application.
Modernization of the CIMS application is intended to fully integrate the new regulatory framework into the existing system to ensure that the Regulatory Affairs and Outreach branch (RA&O) can continue to manage information filed by patentees as well as monitor compliance.
| 2020–21 budgetary spending (as indicated in Main Estimates) | 2020–21 planned spending | 2021–22 planned spending | 2022–23 planned spending |
|---|---|---|---|
| 3,075,439 | 3,075,439 | 3,050,215 | 3,035,389 |
| 2020–21 planned full-time equivalents | 2021–22 planned full-time equivalents | 2022–23 planned full-time equivalents |
|---|---|---|
| 22 | 22 | 22 |
Spending and human resources
This section provides an overview of the department’s planned spending and human resources for the next three consecutive fiscal years, and compares planned spending for the upcoming year with the current and previous years’ actual spending.
Planned spending
Departmental spending 2017–18 to 2022–23
The following graph presents planned (voted and statutory) spending over time.
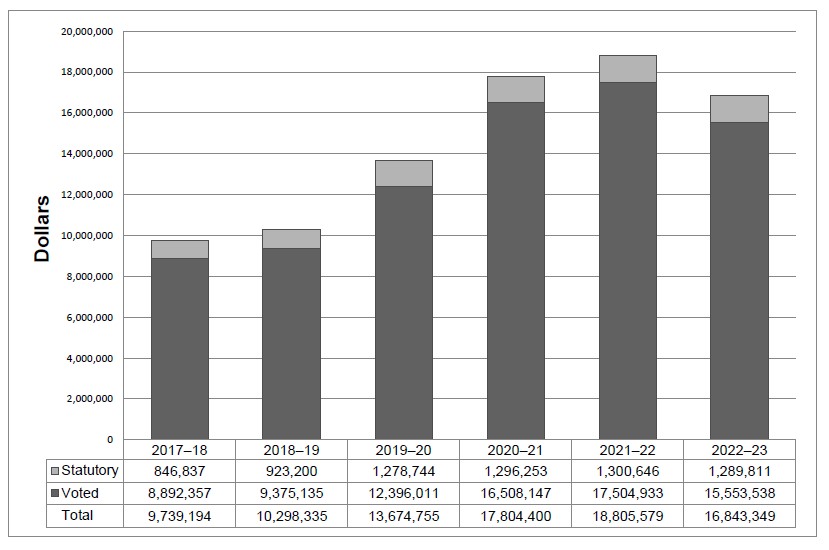
Long description
The graph above shows the PMPRB's planned and actual statutory and voted spending trend over time. The bars for 2017-18 and 2018-19 show actual spending; the bar for 2020-21 shows forecasted spending to the end of that fiscal year; and, the bars for 2021-22 and beyond show planned spending.
As announced in the 2017 Budget, the PMPRB received additional funding for future years; $3,849,215 in 2018-19, $5, 694,677 in 2019-20, $6,671,853 in 2020-21, $7,668,725 in 2021-22 and $5,680,633 in 2022-23 and ongoing, including EBP and increased funding for the SPA.
Forecasted spending for 2019-20 is significantly higher than actual spending in 2018-19 because of increased funding and hiring of new staff to prepare for implementation of the new Guidelines. Forecasted spending for 2020-21 and 2021-22 increases significantly from 2019-20 due to continued funding increases related to the enforcement of the new regulatory framework.
The following table shows actual, forecast and planned spending for each of the PMPRB’s core responsibilities and to Internal Services for the years relevant to the current planning year.
Budgetary planning summary for Core Responsibilities and Internal Services (dollars)
| Core responsibilities and Internal Services | 2017–18 expenditures | 2018–19 expenditures | 2019–20 forecast spending | 2020–21 budgetary spending (as indicated in Main Estimates) | 2020–21 planned spending | 2021–22 planned spending | 2022–23 planned spending |
|---|---|---|---|---|---|---|---|
| Regulate Patented Medicine Prices | 7,109,924 | 7,343,076 | 9,839,415 | 14,728,961 | 14,728,961 | 15,755,364 | 13,807,960 |
| Subtotal | 7,109,924 | 7,343,076 | 9,839,415 | 14,728,961 | 14,728,961 | 15,755,364 | 13,807,960 |
| Internal Services | 2,629,270 | 2,955,259 | 3,835,340 | 3,075,439 | 3,075,439 | 3,050,215 | 3,035,389 |
| Total | 9,739,194 | 10,298,335 | 13,674,755 | 17,804,400 | 17,804,400 | 18,805,579 | 16,843,349 |
The forecast spending for 2019-20 is based on actual spending and anticipated spending to year-end; which does not anticipate full spending of the SPA. At the time of preparing this report, forecasted spending of the SPA amounted to $1,371,745. This and an anticipated surplus in the salary and O&M spending account for the variance in 2019-20 forecast spending and 2020-21 planned spending.
For purposes of forecasting Planned Spending for 2020-21 and future years, the PMPRB assumes the entire SPA funding for hearings will be spent. This is because these expenditures are dependent on the number of hearings, and the length and complexity of the hearings held, which are difficult to predict. The amount of the SPA for 2020-21 is $5,257,786; 2021-22 is $6,206,486; and, for 2022-23 and beyond the amount of the SPA is $4,463,361. Because of the implementation of the new Guidelines in 2020, it is anticipated that by 2022-23, Staff and patentees will have a better understanding of the how to apply the new Guidelines and there will be fewer contested pricing matters.
Planned human resources
The following table shows actual, forecast and planned full-time equivalents (FTEs) for each core responsibility in the PMPRB’s departmental results framework and to Internal Services for the years relevant to the current planning year.
| Core responsibilities and Internal Services | 2017–18 actual full-time equivalents | 2018–19 actual full-time equivalents | 2019–20 forecast full-time equivalents | 2020–21 planned full-time equivalents | 2021–22 planned full-time equivalents | 2022–23 planned full-time equivalents |
|---|---|---|---|---|---|---|
| Regulate Patented Medicine Prices | 48 | 47 | 58 | 65 | 65 | 65 |
| Subtotal | 48 | 47 | 58 | 65 | 65 | 65 |
| Internal Services | 20 | 19 | 21 | 22 | 22 | 22 |
| Total | 68 | 66 | 79 | 87 | 87 | 87 |
The increase in planned FTEs for 2019-20 and beyond is a result of the additional funding received in the 2017 Budget and the need for additional staff and expertise to address the requirements of framework modernization.
Estimates by vote
Information on the PMPRB’s organizational appropriations is available in the 2020–21 Main Estimates.Footnote iii
Condensed future-oriented statement of operations
The condensed future‑oriented statement of operations provides an overview of the PMPRB’s operations for 2019–20 to 2020–21.
The amounts for forecast and planned results in this statement of operations were prepared on an accrual basis. The amounts for forecast and planned spending presented in other sections of the Departmental Plan were prepared on an expenditure basis. Amounts may therefore differ.
A more detailed future‑oriented statement of operations and associated notes, including a reconciliation of the net cost of operations to the requested authorities, are available on the PMPRB’s websiteFootnote iv.
| Financial information | 2019–20 forecast results | 2020–21 planned results | Difference (2020–21 planned results minus 2019–20 forecast results) |
|---|---|---|---|
| Total expenses | 15,182,072 | 19,613,907 | 4,431,835 |
| Total revenues | 867 | 0 | (867) |
| Net cost of operations before government funding and transfers | 15,181,205 | 19,613,907 | 4,432,702 |
For purposes of forecasting Planned Spending for 2020-21, the PMPRB assumes the entire SPA funding for hearings will be spent. This is because these expenditures are dependent on the number of hearings, and the length and complexity of the hearings held, which are difficult to predict. The amount of the SPA for 2020-21 is $5,257,786; spending of SPA funds in 2019-20 to date is $1,371,745. This accounts for a great portion of the variance.
Corporate information
Organizational profile
Appropriate minister(s): The Honourable Patty Hajdu
Institutional head: Dr. Mitchell Levine, Chairperson
Ministerial portfolio: Health
Enabling instrument(s): Patent ActFootnote vand Patented Medicines RegulationsFootnote vi
Year of incorporation / commencement: 1987
Other: The Minister of Health is responsible for the pharmaceutical provisions of the Patent Act set out in sections 79 to 103. Although the PMPRB is part of the Health Portfolio, because of its quasi-judicial responsibilities the PMPRB carries out its mandate at arm’s length from the Minister. It also operates independently of Health Canada, which approves drugs for safety, efficacy and quality; other Health Portfolio members, such as the Public Health Agency of Canada, the Canadian Institutes of Health Research and the Canadian Food Inspection Agency; and FPT public drug plans, which approve the listing of drugs for their respective formularies for reimbursement purposes; and the Common Drug Review, administered by the CADTH, which recommends drugs that should qualify for reimbursement purposes by participating public drug plans.
Raison d’être, mandate and role: who we are and what we do
“Raison d’être, mandate and role: who we are and what we do” is available on the PMPRB’s websiteFootnote vii.
For more information on the department’s organizational mandate letter commitments, see the Minister’s mandate letterFootnote viii.
Operating context
Information on the operating context is available on the PMPRB’s websiteFootnote ix.
Reporting framework
The PMPRB’s approved Departmental Results Framework and Program Inventory for 2020–21 are as follows.
| Departmental Results Framework | Core Responsibility: Regulate Patented Medicine Prices | Internal Services | |
| Departmental Result: Affordable patented medicine prices | Indicator 1: % of patented medicine prices in Canada are below the median price of the PMPRB comparator countries | ||
| Indicator 2: % of patented medicine prices in Canada within the thresholds set out in the Guidelines | |||
| Program Inventory | Patented Medicine Price Regulation Program | ||
| Pharmaceutical Trends Program | |||
Supporting information on the program inventory
Supporting information on planned expenditures, human resources, and results related to the PMPRB’s Program Inventory is available in the GC InfoBaseFootnote x.
Supplementary information tables
The following supplementary information tables are available on the PMPRB’s website:
Federal tax expenditures
The PMPRB’s Departmental Plan does not include information on tax expenditures that relate to its planned results for 2020–21.
Tax expenditures are the responsibility of the Minister of Finance, and the Department of Finance Canada publishes cost estimates and projections for government‑wide tax expenditures each year in the Report on Federal Tax Expenditures.Footnote xiii This report provides detailed information on tax expenditures, including objectives, historical background and references to related federal spending programs, as well as evaluations, research papers and gender-based analysis. The tax measures presented in this report are solely the responsibility of the Minister of Finance.
Organizational contact information
Mailing address
The Patented Medicine Prices Review Board
Box L 40
Standard Life Centre
333 Laurier Avenue West
Suite 1400
Ottawa, Ontario
Toll Free number: 1 877 861-2350
Fax: 613 288-9643
TTY: 613 288-9654
Email: PMPRB.Information-Renseignements.CEPMB@pmprb-cepmb.gc.ca
Appendix: definitions
- appropriation (crédit)
- Any authority of Parliament to pay money out of the Consolidated Revenue Fund.
- budgetary expenditures (dépenses budgétaires)
- Operating and capital expenditures; transfer payments to other levels of government, organizations or individuals; and payments to Crown corporations.
- core responsibility (responsabilité essentielle)
- An enduring function or role performed by a department. The intentions of the department with respect to a core responsibility are reflected in one or more related departmental results that the department seeks to contribute to or influence.
- Departmental Plan (plan ministériel)
- A report on the plans and expected performance of a department over a 3‑year period. Departmental Plans are tabled in Parliament each spring.
- departmental priority (priorité ministérielle)
- A plan or project that a department has chosen to focus and report on during the planning period. Departmental priorities represent the things that are most important or what must be done first to support the achievement of the desired departmental results.
- departmental result (résultat ministériel)
- A consequence or outcome that a department seeks to achieve. A departmental result is often outside departments’ immediate control, but it should be influenced by program-level outcomes.
- departmental result indicator (indicateur de résultat ministériel)
- A factor or variable that provides a valid and reliable means to measure or describe progress on a departmental result.
- departmental results framework (cadre ministériel des résultats)
- A framework that consists of the department’s core responsibilities, departmental results and departmental result indicators.
- Departmental Results Report (rapport sur les résultats ministériels)
- A report on a department’s actual accomplishments against the plans, priorities and expected results set out in the corresponding Departmental Plan.
- experimentation (expérimentation)
- The conducting of activities that seek to first explore, then test and compare, the effects and impacts of policies and interventions in order to inform evidence-based decision-making, and improve outcomes for Canadians, by learning what works and what doesn’t. Experimentation is related to, but distinct form innovation (the trying of new things), because it involves a rigorous comparison of results. For example, using a new website to communicate with Canadians can be an innovation; systematically testing the new website against existing outreach tools or an old website to see which one leads to more engagement, is experimentation.
- full‑time equivalent (équivalent temps plein)
- A measure of the extent to which an employee represents a full person‑year charge against a departmental budget. Full‑time equivalents are calculated as a ratio of assigned hours of work to scheduled hours of work. Scheduled hours of work are set out in collective agreements.
- gender-based analysis plus (GBA+) (analyse comparative entre les sexes plus [ACS+])
- An analytical process used to assess how diverse groups of women, men and gender-diverse people experience policies, programs and services based on multiple factors including race, ethnicity, religion, age, and mental or physical disability.
- government-wide priorities (priorités pangouvernementales)
- For the purpose of the 2020–21 Departmental Plan, government-wide priorities refers to those high-level themes outlining the government’s agenda in the 2015 Speech from the Throne, namely: Growth for the Middle Class; Open and Transparent Government; A Clean Environment and a Strong Economy; Diversity is Canada's Strength; and Security and Opportunity.
- horizontal initiative (initiative horizontale)
- An initiative in which two or more federal organizations are given funding to pursue a shared outcome, often linked to a government priority.
- non‑budgetary expenditures (dépenses non budgétaires)
- Net outlays and receipts related to loans, investments and advances, which change the composition of the financial assets of the Government of Canada.
- performance (rendement)
- What an organization did with its resources to achieve its results, how well those results compare to what the organization intended to achieve, and how well lessons learned have been identified.
- performance indicator (indicateur de rendement)
- A qualitative or quantitative means of measuring an output or outcome, with the intention of gauging the performance of an organization, program, policy or initiative respecting expected results.
- performance reporting (production de rapports sur le rendement)
- The process of communicating evidence‑based performance information. Performance reporting supports decision-making, accountability and transparency.
- plan (plan)
- The articulation of strategic choices, which provides information on how an organization intends to achieve its priorities and associated results. Generally a plan will explain the logic behind the strategies chosen and tend to focus on actions that lead up to the expected result.
- planned spending (dépenses prévues)
-
For Departmental Plans and Departmental Results Reports, planned spending refers to those amounts presented in the Main Estimates.
A department is expected to be aware of the authorities that it has sought and received. The determination of planned spending is a departmental responsibility, and departments must be able to defend the expenditure and accrual numbers presented in their Departmental Plans and Departmental Results Reports.
- program (programme)
- Individual or groups of services, activities or combinations thereof that are managed together within the department and focus on a specific set of outputs, outcomes or service levels.
- program inventory (répertoire des programmes)
- Identifies all of the department’s programs and describes how resources are organized to contribute to the department’s core responsibilities and results.
- result (résultat)
- An external consequence attributed, in part, to an organization, policy, program or initiative. Results are not within the control of a single organization, policy, program or initiative; instead they are within the area of the organization’s influence.
- statutory expenditures (dépenses législatives)
- Expenditures that Parliament has approved through legislation other than appropriation acts. The legislation sets out the purpose of the expenditures and the terms and conditions under which they may be made.
- strategic outcome (résultat stratégique)
- A long-term and enduring benefit to Canadians that is linked to the organization’s mandate, vision and core functions.
- target (cible)
- A measurable performance or success level that an organization, program or initiative plans to achieve within a specified time period. Targets can be either quantitative or qualitative.
- voted expenditures (dépenses votées)
- Expenditures that Parliament approves annually through an Appropriation Act. The vote wording becomes the governing conditions under which these expenditures may be made.