Measles surveillance in Canada: 2017

Download the alternative format
(PDF format, 580 MB, 18 pages)
Organization: Health Canada
Published: 2019-08-14
Table of contents
- Introduction
- Methods
- Results
- Discussion
- Limitations
- Conclusion
- Acknowledgements
- Appendix A
- References
List of figures
- Figure 1: Number of reported measles cases (n=45), by end date of epidemiologic week of rash onset and reporting province or territory, Canada, 2017
- Figure 2: Confirmed measles cases (n=45) and incidence rates (per 1,000,000 population) by age group, Canada, 2017
- Figure 3: Vaccination status of confirmed measles cases (n=45) reported in Canada in 2017
- Figure 4: Distribution of measles cases (n=29) for Outbreak Two by reporting province, epidemiological week of rash onset and setting of exposure
List of tables
Introduction
Measles is a highly contagious infectious disease caused by the measles virus and was responsible for an estimated 2.6 million deaths annually before the introduction of the measles vaccine in 1963Footnote 1. Symptoms of measles include: fever; cough; runny nose; inflamed eyes; fatigue; small, white spots in the mouth and throat; and a blotchy (maculopapular) rash that develops on the face and bodyFootnote 2. Measles can be especially dangerous for infants and those with weakened immune systems. Complications of measles can include ear infections, pneumonia, and encephalitisFootnote 2. Measles continues to be an important public health issue globally. In 2017, a total of 895 cases of measles were reported to the World Health Organization (WHO) in the Region of the Americas and 149,357 cases of measles were reported to the WHO worldwideFootnote 3. Approximately 110,000 people died from measles worldwide in 2017, with most of them being children under 5 years of ageFootnote 1.
Although Canada's measles elimination status was achieved in 1998Footnote 4Footnote 5, Canada's estimated first dose measles-containing vaccine coverage rate among two year olds is 90%Footnote 6. This is below the minimum 95% vaccination coverage for all population cohorts needed to maintain measles eliminationFootnote 5. Therefore, measles remains a disease of concern in Canada due to the risk of importation from other countries, where the disease is still endemic. In order to provide evidence for measles elimination, timely and enhanced national surveillance of measles in Canada is conducted through the Canadian Measles and Rubella Surveillance System (CMRSS), which is coordinated by the Public Health Agency of Canada's (PHAC) Centre for Immunization and Respiratory Infectious Diseases (CIRID) and the National Microbiology Laboratory (NML). This involves weekly collection of data on confirmed measles cases in Canada from all 13 provinces and territories.
The objective of this report is to provide an epidemiologic summary of measles activity reported in Canada for 2017.
Methods
Surveillance data: Confirmed cases of measles meeting the national case definitionFootnote 7 were reported weekly to PHAC by provinces and territories through CMRSS. Cases with rash onset between January 1, 2017 and December 31, 2017 were included in this report. Epidemiological weeks of rash onset are assigned by CIRID with week 1 beginning on the first Sunday of the year. A data validation process was conducted, with all provinces and territories, that included querying for blank fields, identifying illogical field entries and confirming values with reporting jurisdictions.
Genotyping: Measles virus genotyping was performed at the NML by sequencing of specific measles genome targets in accordance with WHO guidelinesFootnote 8Footnote 9. Measles viral sequences were deposited in the WHO measles nucleotide surveillance database (MeaNS) and compared to so called "named strains" as well as sequences deposited by other members of the global measles laboratory networkFootnote 9Footnote 10.
Analysis: Descriptive epidemiologic analyses were performed for age, gender, location, vaccination status, hospitalization, exposure setting and genotype. Cases that were age-ineligible for routine vaccination (i.e., aged less than 1 year or born before 1970) were classified as up-to-date, regardless of reported statusFootnote 11. Those aged 7 years or more and born after 1970 were classified as up-to-date with two doses. Those aged 1 to 6 years were classified as up-to-date with at least one dose. Measles outbreaks are defined as two or more confirmed cases linked, either epidemiologically, virologically, or bothFootnote 12.
Results
In 2017, the incidence of measles in Canada was 1.2 cases per 1,000,000 population, with a total of 45 reported cases. The distribution of measles cases by week of rash onset and reporting province or territory are presented in Figure 1. All cases were laboratory-confirmed and 37 (82%) were genotyped. The genotypes detected were B3 (n=8 cases) and D8 (n=29 cases). Three outbreaks accounted for the majority of cases (84%, 38/45), while the remaining seven cases were sporadic without further spread.
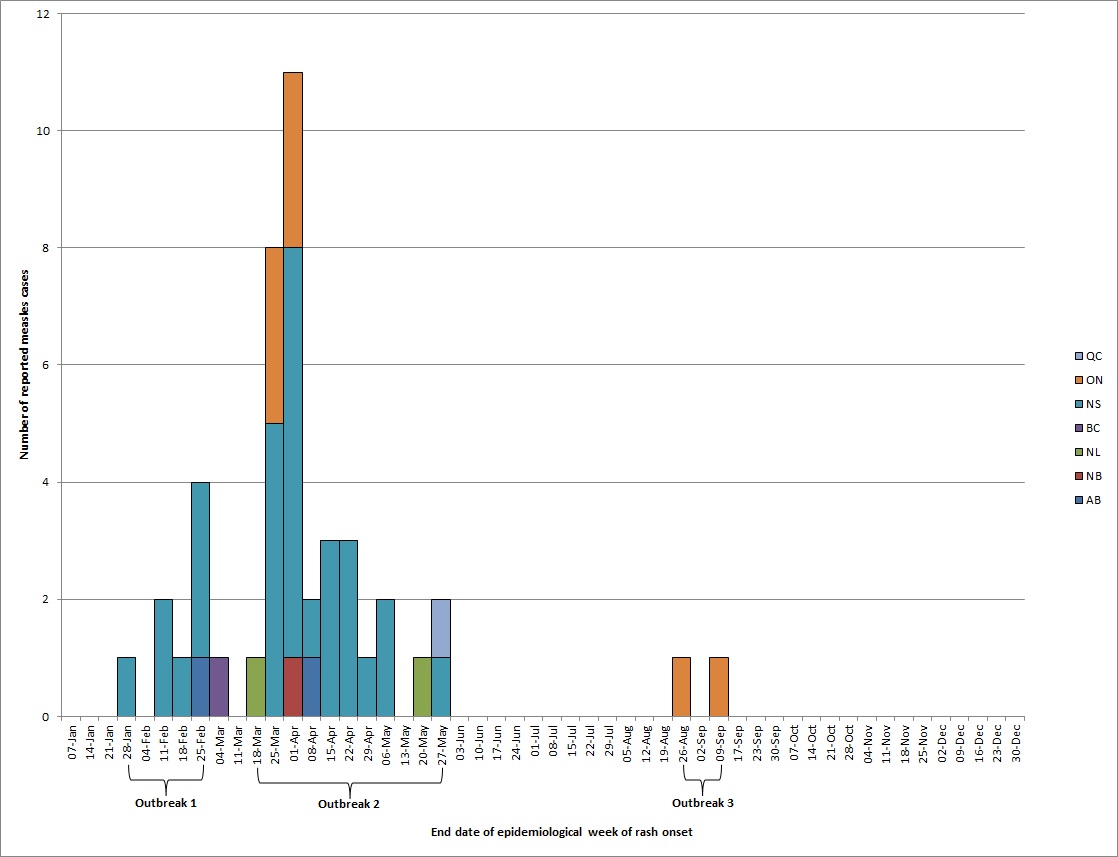
Figure 1 - Text description
There were no measles cases in Canada with rash onset between January 1, 2017 and epidemiological week of rash onset ending January 21, 2017.
Outbreak 1 began in epidemiological week of rash onset ending in January 28 and ended in epidemiological week of rash onset ending in February 25.
Outbreak 2 began in epidemiological week of rash onset ending in March 18 and ended in epidemiological week of rash onset ending in May 27.
Outbreak 3 began in epidemiological week of rash onset ending in August 26 and ended in epidemiological week of rash onset ending in September 9.
The following are the number of measles cases received by end date of epidemiological week of rash onset in 2017:
January 28, 1 in Nova Scotia
February 11, 2 in Nova Scotia
February 18, 1 in Nova Scotia
February 25, 1 in Alberta, 3 in Nova Scotia
March 4, 1 in British Columbia
March 18, 1 in Newfoundland & Labrador
March 25, 5 in Nova Scotia, 3 in Ontario
April 1, 1 in New Brunswick, 7 in Nova Scotia, 3 in Ontario
April 8, 1 in Alberta, 1 in Nova Scotia
April 15, 3 in Nova Scotia
April 22, 3 in Nova Scotia
April 29, 1 in Nova Scotia
May 6, 2 in Nova Scotia
May 20, 1 in Newfoundland & Labrador
May 27, 1 in Nova Scotia, 1 in Québec
August 26, 1 in Ontario
September 9, 1 in Ontario
No measles cases were reported from epidemiological week of rash onset ending in September 17th to December 31st.
Information on age, gender and reporting province or territory were available for all cases reported in 2017. Cases were aged from 2 to 52 years, with a median age of 28 years. The most frequently reported age group was 25 to 44 years (58%, n=26), followed by those aged 15 to 24 years (16%, n= 7). The highest incidence rate was reported in those aged 25 to 44 years at 2.6 cases per 1,000,000 population (Figure 2). Approximately half of the reported cases (53%, n=24) were male. Seven Canadian provinces reported measles cases in 2017: British Columbia, Alberta, Ontario, Quebec, New Brunswick, Nova Scotia, and Newfoundland and Labrador. Incidence rates ranged from 0.2 cases to 31.3 cases per 1,000,000 population, compared to the national incidence rate of 1.2 cases per 1,000,000 population.
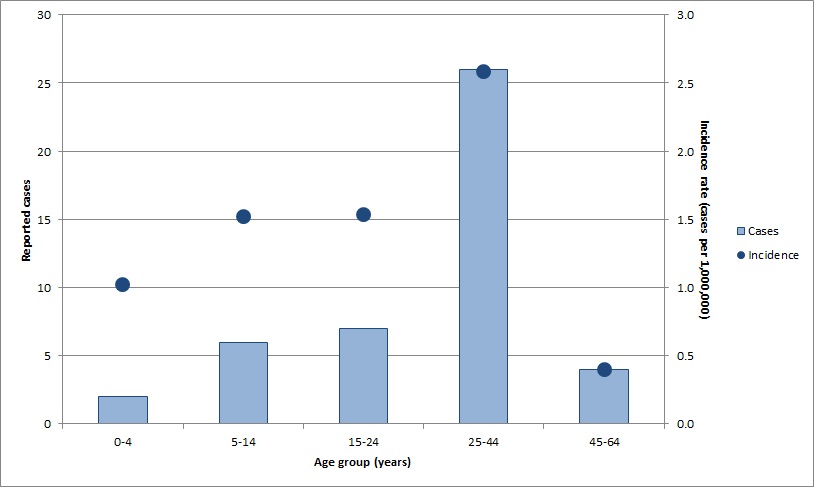
Figure 2 - Text description
| Age group (years) | Reported cases | Incidence rate (cases per 1,000,000) |
|---|---|---|
| 0-4 | 2 | 1.0 |
| 5-14 | 6 | 1.5 |
| 15-24 | 7 | 1.5 |
| 25-44 | 26 | 2.6 |
| 45-64 | 4 | 0.4 |
Hospitalization
Of the 45 cases reported in 2017, 41 cases (91%) had hospitalization information. Of those 41 cases, six cases (15%) were hospitalized at a rate of 0.16 per 1,000,000 population. The age distribution of cases hospitalized was 3 to 43 years. Of the six cases that were hospitalized, one case (17%) had up-to-date vaccination. Four hospitalized cases were travel-related and were hospitalized shortly after returning to Canada.
Vaccination
Of the 45 cases in 2017, documented vaccination information was available for 32 cases (71%) and was unknown or missing for 13 cases (29%). Of those 32 cases, 17 (53%) cases were fully immunized, nine (28%) cases were under-immunized, and six (19%) cases were unimmunized (Figure 3). Three of the cases with unknown or missing vaccination information were adults born before 1970 (aged 49 or older) and do not require MMR vaccination, as they can be presumed to have acquired natural immunity to measlesFootnote 13. The other ten cases with unknown or missing vaccination information were born in or after 1970 and presumed to be susceptible to measles. Overall, 44% (20/45) of cases had up-to-date vaccination status; this includes the 17 cases that reported having received at least two documented doses and the three cases that are presumed to have acquired natural immunity to measles.
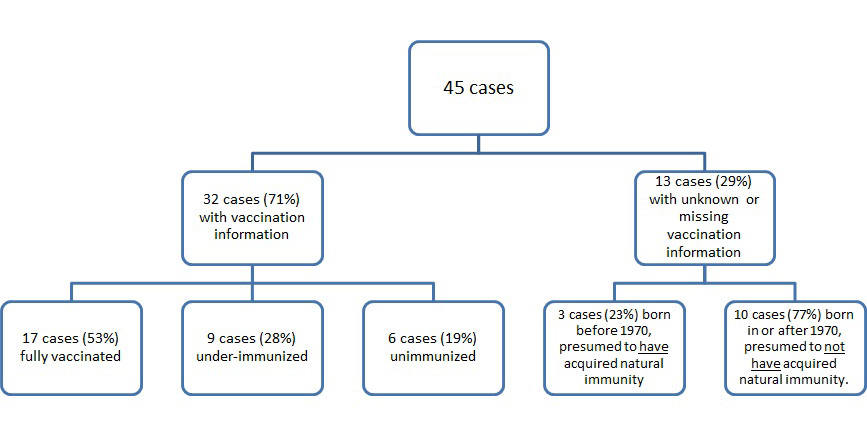
Figure 3 - Text description
The flow chart for the vaccination status of confirmed measles cases reported in Canada is shown in figure 3. Of the 45 cases in 2017, 32 cases (71%) had vaccination information and 13 cases (29%) had unknown or missing vaccination information. Of the 32 cases (71%) with vaccination information, 17 cases (53%) were fully vaccinated, 9 cases (28%) were under-immunized, and 6 cases (19%) were unimmunized. Of the 13 cases (29%) with unknown or missing vaccination information, 3 cases (23%) were born before 1970 and presumed to have acquired natural immunity and 10 cases (77%) were born in or after 1970 and presumed to not have acquired natural immunity.
Importations
Nine cases (20%) were imported into Canada in 2017, having been exposed to measles during travel to the following countries or regions: India (n=3), Indonesia (n=2), Pakistan (n=1), United States (n=1) and Europe (n=1). One case reported travel to both Mexico and France. Of the nine imported cases, three of these resulted in three separate outbreaks and an additional 35 cases. Six of the nine imported cases were sporadic cases without further spread. In total, imported and import-related cases accounted for 98% (n=44) of cases in 2017. The remaining case had no recent history of travel or contact, so the source of exposure was unknown. This case was also sporadic with no further spread. The median age of the seven sporadic cases was 20 years. All seven cases were genotyped: six cases were D8 and one case was genotype B3. Six of the seven cases reported travel history while the final case did not travel (genotype D8). Four cases were imported from the South East Asia region, and were all genotype D8, which is endemic in the regionFootnote 10. All four had distinct viral sequences that did not match any WHO named strains. One case was imported from Pakistan, and the locally endemic genotype, B3, was identified. The last case, genotype D8, traveled to both France and Mexico. None of the cases had a documented dose of measles-containing vaccine prior to exposure.
Outbreaks
Three measles outbreaks were identified in 2017, spanning epidemiological weeks 4 to 36 (January 22nd to September 9th) (Figure 1) and they ranged in size from two to 29 cases. Of the 45 cases reported in 2017, 38 (84%) were outbreak related. The outbreaks are described in more detail below.
Outbreak One
The first outbreak was reported by Nova Scotia and resulted in seven cases (with onset dates spanning epidemiological weeks 4-8) (Figure 1). The median age was 24 years (range: 21-30 years). All cases were male and no hospitalizations or deaths were reported. The index case, with disease onset in week 4, reported travel to the United States to a region with no known measles activity at the time of the exposure. Two secondary cases, with onset dates in epidemiological week 6, were subsequently reported. These two cases resulted in four other transmission cases with onset dates in epidemiological weeks 7 and 8. Vaccination history was available for all cases in this outbreak. Five cases, including the index case, had two documented doses of measles-containing vaccine prior to exposure. The other two cases had one documented dose of measles containing vaccine prior to exposure. Five of the seven outbreak cases were genotyped. All were genotype B3 and had sequences identical to one another and the WHO named strain MVs/Dublin.IRL/8.16/ (GenBank accession number KY013331).
Outbreak Two
The second outbreak was a multi-jurisdictional outbreak, with no deaths reported. Cases were reported by Nova Scotia, Newfoundland and Labrador, Ontario, New Brunswick, and Alberta. Outbreak Two resulted in 29 cases, with onset dates spanning epidemiological weeks 11-21 (Figure 4). The median age was 29 years (range: 6-52 years). Fifty-five percent of the cases were female. Of the 29 cases, the index case and two other cases were hospitalized. The index case, with disease onset in epidemiological week 11, was reported by Newfoundland and Labrador. The index case had travelled from India to Nova Scotia via a connecting flight in Ontario, while infectious. Five secondary cases, with onset dates from epidemiological weeks 12 to 14, who were exposed to the index case during air travel or in an airport setting were reported in Ontario, Alberta, Nova Scotia and New Brunswick. These included three passengers on the same flight as the index case, one flight attendant, and an exposure in an airport terminal. The index case was hospitalized, in Nova Scotia, and further transmission took place in a healthcare setting, including 10 healthcare workersFootnote 12. Subsequent exposures in the following weeks were identified. Four cases, with onset dates in epidemiological weeks 15, 18, 20 and 21, were determined as part of the outbreak but the exact setting of exposure was unknown. Fifteen cases (52%) did not have up-to-date vaccination status prior to exposure, including the index case. Of the 10 healthcare workers, five (50%) had up-to-date immunization. Of the other 5 healthcare workers, two were born before 1970. The median age of the 10 healthcare workers was 36 years (range: 26-49). The genotype associated with the outbreak was D8 (23 cases sequenced), which is endemic in IndiaFootnote 10, and the sequence was not identical to any WHO named strain.
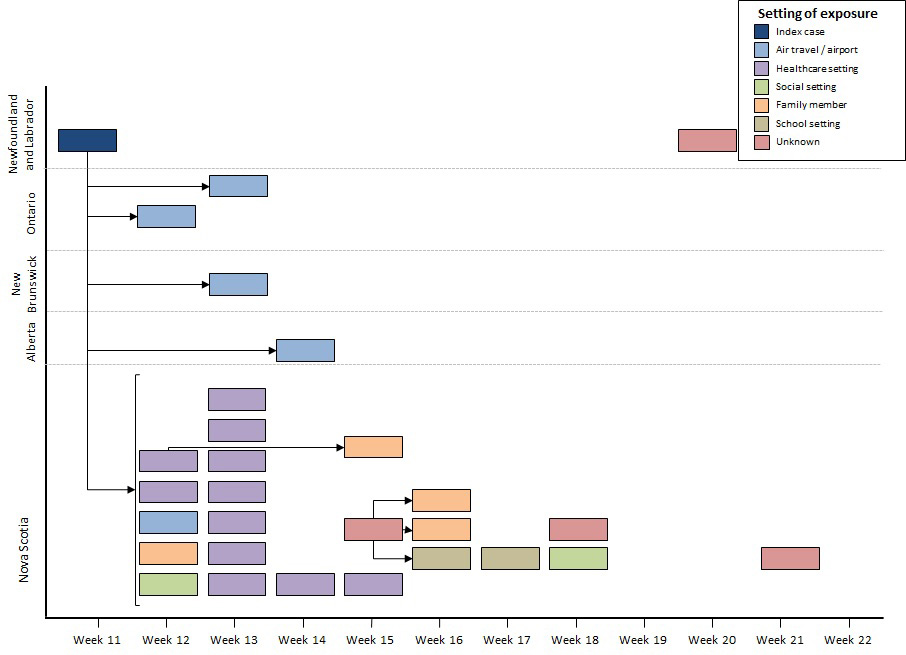
Figure 4 - Text description
The distribution of measles cases for Outbreak Two by reporting province, epidemiological week of rash onset and source of exposure is shown in figure 4. The index case with rash onset in epidemiological week 11 was reported by Newfoundland and Labrador. Five secondary cases, with rash onset dates spanning epidemiological weeks 12 to 14, who were exposed to the index case during air travel or in an airport setting, were reported in Ontario, Alberta, Nova Scotia and New Brunswick. The index case was hospitalized in Nova Scotia and 11 cases of further transmission took place in a healthcare setting, with rash onset spanning epidemiological weeks 12 to 15. Subsequent exposures took place in a school setting (n=2), in social settings (n=2) and among family members (n=4) in the weeks that followed, with rash onset spanning epidemiological weeks 12 to 18. Four cases, with rash onset dates in epidemiological weeks 15, 18, 20 and 21, were determined to be part of the outbreak but the exact setting of exposure was exposed to an unknown source.
Outbreak Three
Outbreak Three, reported by Ontario, resulted in two cases (with onset dates in epidemiological weeks 34 and 36) (Figure 1). The median age was 27.5 years and both cases were females. The index case reported travel to Europe. The country of exposure is unknown due to a lack of information about the index case's location during the incubation period. One case was hospitalized and no deaths were reported. Vaccination history was available for both cases: neither case had a documented dose of measles-containing vaccine prior to exposure. The genotype associated with the outbreak was B3, and the sequence was identical to the WHO named strain MVs/Dublin.IRL/8.16/ (GenBank accession number KY013331).
Discussion
There were 45 confirmed cases of measles reported in Canada in 2017, which is the lowest number of cases and incidence rate after 2016 (n=11; 0.3 cases per 1,000,000 population), since annual publication of reports began in 2013. The median age (28 years) of cases indicate that a large portion falls within the cohort that received one dose of measles-containing vaccine, prior to the introduction of the second dose in routine immunization schedules in 1996-1997Footnote 14. Over twenty percent of all cases were among healthcare workers, of which half of these cases had up-to-date vaccination status. The age range of these healthcare worker cases fall in the cohort that received one dose of measles-containing vaccine. An overwhelming majority of Canadian cases were genotype D8 and all but one of these cases were imported or import-related cases from the South East Asia region, where this genotype is endemic. Only one case was exposed in Canada to an unknown source. Measles importations are to be expected due to the high frequency of persons travelling from measles endemic areas into Canada. Therefore, Canada's maintenance of measles elimination status will continue to be challenged. Furthermore, over half of the 2017 cases were not fully immunized against the measles virus; Canada will remain vulnerable to importations and thus, there is a need to maintain high vaccination coverage in Canada. This highlights that Canada needs to continue to promote vaccination for those at risk, including potential travellers. The 17 confirmed cases of measles that were fully immunized may be the result of vaccine failure, which may include failure to develop an appropriate immune response, waning of vaccine-induced immunity to non-protective levels, and improper vaccine storage, handling and administration of the vaccineFootnote 15Footnote 16. A large majority of cases did not require hospitalization, indicating that most of the cases in 2017 did not result in serious illness.
There are four criteria set out by the Pan American Health Organization (PAHO) for the ongoing verification of measles eliminationFootnote 17 (Appendix A). The indicators, established by PAHO, of a well-performing surveillance system are based on investigation of measles-like illness (i.e., suspected cases), whereas only confirmed cases are nationally notifiable in Canada. As such, these data can only indirectly address the PAHO criteria. In 2017, Canada met or partially met three of four PAHO essential indicators for maintenance of measles elimination. Canada has a high performing national surveillance system for measles, which is able to detect imported and import-related cases, as well as cases with unknown source of exposure.
However, Canada falls short of the criterion regarding measles-containing vaccine coverage. Canada currently measures (biennially) measles vaccination coverage rates at 2 and 7 years of age, and therefore is unable to assess measles vaccination coverage for all ages 1 through 40 years as set out in the PAHO elimination framework. The 2017 estimate for 2 year olds receiving measles-containing vaccine is 90%, below the PAHO indicator of 95%. Despite this, based on a combination of epidemiologic and laboratory data, there is no evidence that endemic transmission of the measles virus has been re-established in Canada.
Limitations
These data have limitations that merit consideration. Only confirmed cases are nationally notifiable in Canada; therefore, suspected cases of measles or cases of measles that do not present to the healthcare system are not captured by CMRSS. Moreover, information on mortality and detailed information on morbidity (e.g., length of hospitalization, sequelae) are not currently captured by CMRSS, limiting the ability to completely describe the burden of illness due to measles in Canada.
Conclusion
Both in Canada and abroad, maintaining high vaccination coverage rate with measles-containing vaccine requires a sustained public health effort, and is an essential component of a strategy for achieving and maintaining measles elimination. Although importations and areas of low vaccination coverage continue to challenge Canada's elimination status, surveillance data indicates that measles elimination has been maintained.
Acknowledgements
The authors gratefully acknowledge the continued cooperation and efforts of provincial and territorial surveillance and laboratory partners for providing and validating data captured by CMRSS, for referring specimens for molecular surveillance (genotyping) and for their review of the report content.
The authors are also grateful to NML staff for their contribution in generating the genotype data.
Appendix A
| Criterion | Indicator | Description |
|---|---|---|
| Verify the interruption of endemic measles cases for a period of at least three years from the last known endemic case, in the presence of high-quality surveillance. | Zero cases of endemic transmission. | Criterion met. Canada achieved measles elimination status in 1998. Since then, molecular and epidemiological data continue to demonstrate that no viral strain has circulated for a period of one year or more in Canada Footnote a. |
| Maintain high-quality surveillance sensitive enough to detect imported and import-related cases. | > 2 suspect cases per 100,000 population adequately investigated. | Criterion partially met. CMRSS allows the identification of imported and import-related cases but not suspected cases. |
| Verify the absence of endemic measles virus strains through viral surveillance. | Measles genotype assessed in 80% of outbreaks. | Criterion met. Genotype information was available for 100% (3/3) of outbreaks reported in 2017. |
| Verify adequate immunization in the population. | 95% of population cohorts aged 1 to 40 years have received a measles-containing vaccine. | Criterion not met. Canada currently measures (biennially) measles vaccination coverage rates at 2 and 7 years of age, and therefore is unable to assess measles vaccination coverage for all ages 1 through 40 years. The 2017 childhood National Immunization Coverage survey estimated first dose measles-containing vaccine coverage among 2 year olds to be 90%Footnote b. The coverage rates remained unchanged since 2013Footnote c. |
|
||
References
- Footnote 1
-
World Health Organization (WHO). Measles. [Updated September 2018; Access October 2018] http://www.who.int/news-room/fact-sheets/detail/measles
- Footnote 2
-
Public Health Agency of Canada. Symptoms of measles. [Updated October 2016; Accessed December 2018] https://www.canada.ca/en/public-health/services/diseases/measles/symptoms-measles.html
- Footnote 3
-
World Health Organization (WHO). Immunization, Vaccines and Biologics, Measles Surveillance Data, Regional summary of reported measles cases [Updated September 2018, Accessed October 2018] http://www.who.int/immunization/monitoring_surveillance/burden/vpd/surveillance_type/active/measles_monthlydata/en/
- Footnote 4
-
King A, Varughese P, De Serres G, Tipples GA, Waters J, Working Group on Measles Elimination. Measles elimination in Canada. J Infect Dis. 2004 May 1;189 Suppl 1:S236-42.
- Footnote 5
-
Public Health Agency of Canada. Elimination of Measles Rubella and Congenital Rubella Syndrome in Canada, Documentation and Verification Report, submitted to PAHO December 2011.
- Footnote 6
-
Public Health Agency of Canada. Preliminary Results from the 2017 childhood National Immunization Coverage Survey (cNICS) [Internet]. [updated 2019 Mar; cited 2019 Mar]. Ottawa: PHAC; 2019. https://www.canada.ca/en/services/health/publications/vaccines-immunization/vaccine-uptake-canadian-children-preliminary-results-2017-childhood-national-immunization-coverage-survey.html
- Footnote 7
-
Public Health Agency of Canada. Case definitions for diseases under national surveillance. Can Comm Dis Rep 2009;35-Suppl 2:71-72.
- Footnote 8
-
World Health Organization (WHO). Standardization of the nomenclature for describing the genetic characteristics of wild-type measles viruses. Wkly Epidemiol Rec 1998;73:265.
- Footnote 9
-
Mulders M, Rota P, Brown K, Goodson J. Genetic diversity of wild-type measles viruses and the global measles nucleotide surveillance database (MeaNS). Wkly Epidemiol Rec 2015;90(30):373.
- Footnote 10
-
Rota PA, Brown K, Mankertz A, et al. Global distribution of measles genotypes and measles molecular epidemiology. J Infect Dis 2011;204-Suppl 1:8514.
- Footnote 11
-
Government of Canada. Canada's provincial and territorial routine (and catch-up) vaccination programs for infants and children [Internet]. Ottawa: PHAC; 2018. [updated 2018 Aug; cited 2018 Oct]. https://www.canada.ca/en/public-health/services/provincial-territorial-immunization-information/provincial-territorial-routine-vaccination-programs-infants-children.html
- Footnote 12
-
Measles and Rubella Elimination Working Group (MREWG), Health Canada, Public Health Agency of Canada. Guidelines for the prevention and control of measles outbreaks in Canada. Can Comm Dis Rep 2013;39(ACS-3).
- Footnote 13
-
Government of Canada. Page 13: Canadian immunization guide: Part 1 - key immunization information [Internet]. Ottawa: PHAC; 2018. [updated 2018 Mar; cited 2018 Oct]. https://www.canada.ca/en/public-health/services/publications/healthy-living/canadian-immunization-guide-part-1-key-immunization-information/page-13-recommended-immunization-schedules.html
- Footnote 14
-
Canadian Public Health Association. Immunization timeline [Internet]. [cited 2019 Mar]. https://www.cpha.ca/immunization-timeline
- Footnote 15
-
Centre for Disease Control. Measles outbreak associated with vaccine failure in adults - Federated States of Micronesia, February-August 2014 [Internet]. U.S.: CDC; 2015. [updated 2015 Oct; cited 2019 May]. https://www.cdc.gov/mmwr/preview/mmwrhtml/mm6438a7.htm
- Footnote 16
-
Hinman A, Orenstein W, Mortimer E Jr. When, where, and how do immunizations fail?: 1992. Annals of Epidemiology;2(6):805-12.
- Footnote 17
-
Pan-American Health Organization (PAHO). Plan of action. Documentation and verification of measles, rubella and congenital rubella syndrome elimination in the region of the Americas. Technical Document. 2010.