Drinking water screening value for hexahydro-1,3,5-trinitro-1,3,5-triazine (RDX): Technical summary

Download the alternative format
(PDF format, 683 KB, 10 pages)
Organization: Health Canada
A drinking water screening value of 0.1 mg/L (100 µg/L) is established for RDX.
Screening values
Health Canada's screening values identify limits for contaminants in water that could be used as a source of drinking water. A lifetime of exposure to these contaminants up to the screening value, both by drinking the water or by using it for showering or bathing, is not expected to increase health risks for any Canadian, including children.
Screening values are established for contaminants that are not commonly found in Canadian drinking water (either source or treated) and therefore Guidelines for Canadian Drinking Water Quality are not established. Health Canada establishes screening values for contaminants at the request of federal departments, provinces and territories (jurisdictions). These requests are usually made when there is a concern for human health because the presence of a contaminant is suspected or detected in local source water and that contaminant does not have an established limit in drinking water. Since 2020, the technical summaries for screening values are typically published online when Health Canada expects that screening values may be needed by more than one stakeholder or jurisdiction.
Screening values do not replace or supersede existing regulations. However, screening values may help jurisdictions and the public understand the potential health effects of a contaminant.
Screening values are based on a review of scientific research and international regulatory information available at the time of their development. In addition, screening values are externally peer-reviewed to ensure scientific integrity.
Health Canada is committed to keeping pace with new science, including the potential health risks from contaminants that are not typically found in drinking water and do not have Guidelines for Canadian Drinking Water Quality. To this end, Health Canada includes contaminants with screening values in its cyclical prioritization of contaminants for full guideline development.
Exposure considerations
Identity and sources
Hexahydro-1,3,5-trinitro-1,3,5-triazine (CAS # 121-82-4) is a heterocyclic nitramine used in military and industrial applications. It is commonly known as RDX (British code for Research Department Explosive or Royal Demolition eXplosive), cyclonite, hexogen, hexolite, 1,3,5-trinitro-1,3,5-triazine, cyclotrimethylenetrinitramine, trimethylenetrinitramine, or C-4 (Etnier et al., 1989; HSDB, 2005; ATSDR, 2012). As an explosive, RDX is generally used in mixtures with other explosives and plasticizers or desensitizers (HSDB, 2005; ATDSR, 2012).
RDX is a synthetic compound and does not occur naturally in the environment (ATSDR, 2012). RDX is not produced commercially in the United States or in Canada. Production, handling and packaging of RDX are limited to ammunition plants (HSDB, 2005). In the United States, waste waters resulting from the manufacture of RDX are classified as hazardous wastes and are subject to regulations by the United States Environmental Protection Agency (US EPA) (ATSDR, 2012). RDX can enter the environment from effluents and emissions at ammunition plants, from use at military facilities, and from spills or leaks from improper disposal at plants or hazardous waste sites (ATSDR, 2012).
RDX is a white crystalline solid with a melting point of 205.5°C. RDX is weakly volatile and therefore can only exist in the particulate phase in the ambient atmosphere, which is evidenced by an estimated vapour pressure of 4.10 × 10-9 mm Hg at 25°C (HSDB, 2005; Oxley et al., 2007; Reifenrath et al., 2008). If released to soil, the compound is expected to have high to moderate mobility based upon a Koc range of 42-167. Volatilization from water is not expected to be an important fate process based upon the estimated Henry's Law constant of 2.0 ×10-11 atm-cu m/mole (HSDB, 2005; Oxley et al., 2007; Reifenrath et al., 2008). RDX has moderate water solubility (59.7 mg/L at 25°C). It does not bind significantly to soils and can leach to groundwater from soil (ATSDR, 2012). Aerobic biodegradation in aquatic environments is unlikely to occur; however, RDX undergoes biodegradation in water and soil under anaerobic conditions to form several biodegradation products (ATSDR, 2012). RDX is not expected to bioaccumulate (Bannon et al., 2009; ATSDR, 2012).
Exposure
There is evidence of RDX contamination of surface and groundwater near Canadian and United States military installations (Lapointe et al., 2006; Parker et al., 2006). Additionally, RDX was detected in surface water, groundwater, sediment or soil at 34 US EPA National Priorities List sites (US EPA, 2018). For the general population, including children, exposure to RDX is limited to areas around military plants and facilities where it is manufactured, used or disposed of. Monitoring data indicate that the most likely route of exposure is ingestion of contaminated drinking water or agricultural crops irrigated with contaminated water (Gadagbui et al., 2012). Exposure can also occur by inhaling contaminated particulate matter produced during incineration of RDX-containing waste material. Children playing in contaminated water or soil may also be exposed via ingestion (ATDSR, 2012).
Health considerations
Kinetics
There is little empirical evidence regarding the toxicokinetics of RDX in humans (by the oral route), but reports of adverse effects following accidental or intentional ingestion of RDX, as well as results from animal studies, indicate that RDX is slowly absorbed from the gastrointestinal tract after ingestion (Etnier et al., 1989; ATDSR, 2012). Skin absorption of RDX is very unlikely since it is not particularly lipid soluble (Etnier et al., 1989), as evidenced by its low octanol-water partition coefficient (Log Kow = 0.87) (HSDB, 2005; Krishnan et al., 2009).
In laboratory animals, RDX is metabolized primarily in the liver to several kinds of one-carbon fragments: carbon dioxide, bicarbonate ion and formic acid. No larger intermediates have been identified (Etnier et al., 1989). There is no evidence that RDX accumulates appreciably in any tissue. RDX and its metabolites are eliminated in the urine and exhaled air as carbon dioxide in a few days (Etnier et al., 1989; ATSDR 2012).
A physiologically based pharmacokinetic (PBPK) model was developed to simulate the pharmacokinetics of RDX in rats (Krishnan et al., 2009). The PBPK model was later modified and extended for simulations of human exposure; oral absorption and liver metabolism rates were estimated by optimizing the fit of the model to serum RDX time course data following accidental RDX exposures (Sweeney et al., 2012a). To support interspecies extrapolations for toxicity studies using mice, Sweeney et al. (2012b) further revised the PBPK model. The model consists of five compartments (liver, brain, adipose, slowly perfused, and richly perfused tissue compartments connected via blood flow) and has been designed to simulate RDX exposure via intravenous or oral routes. The model represents oral absorption as first-order uptake from the gastrointestinal tract into the liver with 100% of the dose being absorbed. The model also assumes clearance by first order metabolism in the liver. The kinetics of RDX metabolites are not represented in the model. The US EPA further modified the models to improve animal to human extrapolation for use in the dose-response modeling in their 2018 assessment of RDX (US EPA, 2018).
Health effects
Studies in humans are limited to studies on worker populations exposed to higher levels of RDX than the general population. RDX exerts its primary toxic effect on the central nervous system, but also involves effects on testicles, liver, blood, kidney and the gastrointestinal system in humans (Etnier et al., 1989; Parker et al., 2006; Gong et al., 2009; ATSDR, 2012) and animals (Table 1). The main neurotoxic effect following ingestion of RDX in humans (and animals) is the induction of hyperactivity manifested as convulsions or seizures.
No data are available on the reproductive and developmental toxicity of RDX in humans via ingestion. Results from animal studies are ambiguous with prostate effects and testicular degeneration seen in some rodent studies but not others (Levine et al. 1983; Lish et al. 1984). A two-generation study in rats revealed non-significant decreases in fertility but only at doses that also resulted in decreased body weight (bw) and increased mortality (Cholakis et al. 1980). Developmental studies in rats or rabbits either showed no dose-response effects or showed effects in offspring only at doses that were considered to be maternally toxic (Cholakis et al. 1980, Angerhofer et al. 1986).
The International Agency for Research on Cancer has not classified RDX with respect to carcinogenicity. No studies were located regarding cancer in humans following exposure to RDX. Conclusions from rodent bioassays provide very weak support for carcinogenicity (Table 1). Based on results of the study by Lish et al. (1984), the US EPA has determined that there is suggestive evidence of carcinogenic potential for RDX and calculated an oral slope factor of 0.08 (mg/kg bw per day)-1 (2018). No evidence of genotoxicity of RDX was found in various short- and long-term studies in in vitro or in vivo test systems (Parker et al., 2006; Krishnan et al., 2009).
| Study | Strain, Species (number of males/females), Exposure type, Duration | Dosimetry (mg/kg bw per day) | Critical effect(s) | Point of departure (mg/kg bw per day) |
|---|---|---|---|---|
Crouse et al., 2006 |
F344, Rat (10/sex/group), gavage, 90 days |
0, 4, 8, 10, 12, 15 |
Death, tremors/convulsions, increased erythrocyte mean cell volume and decreased serum cholesterol in males. |
NOAEL= 4 LOAEL= 8 |
Lish et al., 1984 |
B6C3F1, Mouse (85/sex), diet, 2 years |
0, 1.5, 7, 35, 175 (Highest dose group was reduced to 100 mg/kg bw per day during week 11 due to high mortality) |
Elevated kidney and heart weights at some doses, as well as elevated serum triglyceride and cholesterol levels (females) at some doses. Possible behavioural changes. Increased testicular degeneration. Increased incidence of hepatocellular neoplasms and alveolar/bronchiolar adenomas or carcinomas in female mice at doses≥7 mg/kg bw per day. Note: A re-evaluation of histological sections found only one female group (35 mg/kg bw per day) to have significant increase in tumours as compared to controls. Further, when evaluated against the historical control database, the incidence of hepatocellular neoplasms for all groups was within the spontaneous range (Parker et al., 2006). |
NOAEL= 1.5 LOAEL= 7 NOAEL= 1.5 LOAEL= 7 (authors' conclusions) NOAEL= 100 LOAEL= N/A (Parker et al., 2006 |
Levine et al., 1983 |
Fischer 344, Rat 85/sex), diet, 2 years |
0, 0.3, 1.5, 8.0, 40 |
Tremors, convulsions, and hyper-responsiveness; decreased hematocrit, haemoglobin, and erythrocyte levels; hepatomegaly and decreased serum cholesterol and triglycerides; renal papillary necrosis (males only) and increased blood urea nitrogen levels; testicular degeneration; and cataracts (females only); 88% mortality rate. Increased incidence of suppurative inflammation in the prostate of rats exposed to ≥ 1.5 mg/kg bw per day. Note: Inflammation of the prostate gland is a common condition in older rodents (Parker et al. 2006) and may be secondary to a bacterial infection in older rats dying early (ATSDR, 2012). No incidence of cancer. |
NOAEL= 0.3 NOAEL= 8 LOAEL= 40 (ATSDR NOAEL= 40 LOAEL= N/A |
Hart, 1976 |
Sprague-Dawley, Rat (100/sex), diet, 2 years |
0, 1, 3.1, 10 |
Decreased body weight (deemed toxicologically insignificant by authors). No adverse histological effects (did not examine the prostate). No incidence of cancer. |
NOAEL= 10 LOAEL= NA |
Sweeney et al. (2012a) conducted a re-evaluation of the health effects of RDX by using PBPK modeling and benchmark dose (BMD) analyses of previously published data. An array of candidate reference doses (RfDs) were derived for various endpoints including neurotoxicity endpoints (convulsions observed in Crouse et al., 2006 and Levine et al., 1983) and endpoints for which the mode of action is not well elucidated. Applying an uncertainty factor of 30, the RfDs based on seizures ranged from 0.05 to 0.14 mg/kg bw per day. Ultimately, a chronic oral RfD of 0.07 mg/kg bw per day was recommended and considered to be protective of various effects, including neurotoxicity.
Mode of action
RDX's action as a gamma-amino butyric acid (GABA) receptor antagonist is reported to lead to effects on the central nervous system. RDX crosses the blood-brain barrier to antagonize GABA receptors resulting in decreased conduction of chloride through the ion channel; reduced chloride conduction results in depolarization of the neuronal membrane, which reduces spontaneous inhibitory postsynaptic currents; reduction in spontaneous inhibitory postsynaptic currents results in overall reduction in inhibitory inputs to the nervous system (US EPA, 2018). There are no studies available that inform mechanistically how RDX might lead to other health effects.
Sweeney et al. (2012b) conducted a re-evaluation of the cancer weight of evidence and possible mode of action (MOA) for RDX, and concluded that the available data do not suggest a reasonably plausible MOA and that RDX is unlikely to be mutagenic or genotoxic.
Selection of key study
There are no human studies suitable for estimating a point of departure (POD). In animals, the non-cancer effect observed at the lowest concentration is suppurative prostatitis in male F344 rats exposed to RDX in their diet for 2 years (Levine et al., 1983). However, this finding was not observed in other studies in rats exposed to RDX for only 90 days (via oral gavage or in their diet) nor in a 2-year dietary study in mice (Lish et al., 1984). Furthermore, the MOA by which RDX may induce prostate effects is unknown (US EPA 2018).
Evidence for nervous system effects (i.e., convulsions), was observed in multiple studies, multiple species (including humans), and following a range of exposure durations.
Additionally, mechanistic evidence supports the biological plausibility of these effects. Therefore, the health-based value is calculated based on the incidence of convulsions in F344 rats exposed to RDX via oral gavage for 13 weeks (Crouse et al., 2006). The US EPA (2018) also selected neurotoxicity, represented by convulsions, as the critical endpoint for dose-response assessment.
Crouse et al. (2006) administered F344 rats (10/sex/group) 0, 4, 8, 10, 12, or 15 mg/kg bw per day pure (99.9%) RDX in a water suspension via gavage for 7 days/week for 90 days. There were significant differences in body weights and food consumption between dosed animals and controls. Death was observed in all dose groups above 4 mg/kg bw per day, with significant increases in mortality rates (25%) at ≥10 mg/kg bw per day. Deaths were observed as early as day 8 of this 90-day gavage study. The tremors/convulsions were observed within the first week of exposure in the two highest dosed groups and persisted throughout the study. The incidences of convulsions in males and females combined were 0/20, 0/20, 3/20, 6/20, 13/20, and 12/20 in the 0, 4, 8, 10, 12, or 15 mg/kg bw per day dose groups, respectively. The incidence and severity of transient clinical signs (such as changes in arousal, inflammation of eyelash follicles, increased salivation, tremors, and convulsions) increased with dose. Hematological tests showed significant increases in erythrocyte mean cell volume at 8 (males only), 10, and 12 mg/kg bw per day and significant decrease in serum cholesterol in males exposed to ≥8 mg/kg bw/day. No significant increases in the incidence of histopathological alterations were observed.
Given the proximity in dose and time at which mortality and nervous system effects were observed in several studies, including Crouse et al. (2006), the US EPA (2018) conducted an analysis to assess the relationship between convulsions/tremors/seizure-like activity and mortality. The analysis concluded although death may occur without seizures/convulsions - this may be due to the low frequency of observations; however, based on the available data it is appropriate to conclude that convulsions represent a severe endpoint for a human health risk assessment.
Derivation of the screening value
The derivation of the tolerable daily intake (TDI) was based on the incidence of convulsions in male and female rats following a 13-week exposure to RDX via gavage (Crouse et al., 2006). As described by the US EPA (2018), benchmark dose (BMD) modeling was applied to the dose-response data. The data supported a benchmark response (BMR) of 5%, which provides a margin of protection against the severity of the endpoint. A BMDL05 of 2.66 mg/kg bw per day (BMD05 of 5.19 mg/kg bw per day) was identified as the POD. A human equivalent POD (BMDL05-HED) of 1.3mg/kg bw per day was derived using the rat PBPK model (Krishnan et al., 2009, as modified by US EPA, 2018).
Using the human equivalent POD of 1.3 mg/kg bw per day a TDI is calculated as follows:

Equation 1 - Text description
Equation for TDI:
This equation calculates the tolerable daily intake for Hexahydro-1,3,5-trinitro-1,3,5-triazine (RDX). The tolerable daily intake is calculated by dividing the lowest observed adverse effect level for RDX (1.3 micrograms per kilogram of body weight per day) by the uncertainty factor of 300 which equals 0.004 micrograms per kilogram of body weight per day.
where:
- 1.3 mg/kg bw/day is the human equivalent BMDL05 for incidence of convulsions using the combined incidence data from male and female rats (Crouse et al., 2006) as calculated by the US EPA (2018) using PBPK modeling.
- 300 is the uncertainty factor: ×3 for interspecies variability in toxicodynamics, ×10 for intraspecies variability and ×10 for database deficiencies (data gaps in developmental neurotoxicity, lack of incidence data for less severe effects).
Based on the above TDI, a screening value can be derived as follows:
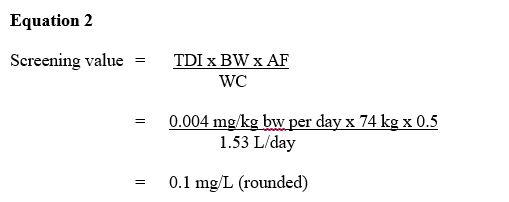
Equation 2 - Text description
Screening value:
This equation calculates the RDX. The screening value for total for RDX is multiplied tolerable daily intake of 0.004 mg/kg bw-day by the mean body weight estimated for a Canadian adult is 74 and by 50% based on usage patterning, environmental fate and physical-chemical properties then dividing it by 1.53 L the estimated daily volume of tap water consumed by an adult. Which equals a screening value of 0.1 mg/L (100 µg/L)
where:
TDI = tolerable daily intake of 0.004 mg/kg bw-day as derived above.
BW = the mean body weight estimated for a Canadian adult is 74 kg (Health Canada, in preparation).
AF = allocation factor: the proportion of exposure to RDX from drinking water, as opposed to other environmental media (i.e., food, air, soil, consumer products). Based on usage patterning, environmental fate and physical-chemical properties, a value of 50% is applied (Gadagbui et al., 2012; Krishnan and Carrier, 2013).
WC = water consumption: the estimated daily volume of tap water consumed by an adult is 1.53 L (Health Canada, in preparation); multi-route exposure assessment (Krishnan and Carrier, 2008) found that dermal and inhalation exposures through showering or bathing represent negligible routes of exposure to RDX.
A screening value of 0.1 mg/L (100 µg/L) for RDX is recommended by Health Canada.
International considerations
Drinking water quality guidelines, standards and/or guidance established by foreign governments or international agencies may vary due to the science available at the time of assessment, as well as the utilization of different policies and approaches, such as the choice of key study, and the use of different consumption rates, body weights and allocation factors.
International jurisdictions such as the World Health Organization, the US EPA, Australia and the European Union have not established regulatory guidelines for RDX in drinking water. The US EPA has established a chronic RfD of 0.004 mg/kg bw per day based on nervous system effects (i.e., convulsions) in rats (US EPA, 2018). Based on a 1988 assessment, the US EPA also has a non-regulatory lifetime health advisory for RDX of 0.002 mg/L. This value is derived from an older dataset and is based on an RfD of 0.003 mg/kg bw per day where the effect was suppurative prostatitis in rats (US EPA, 1988).
Contact us
For more information, please contact us at hc.water-eau.sc@canada.ca.
References
Angerhofer, R., Davis, G., Balezewski, L. (1986). Teratological assessment of trinitro - RDX in rats. ADA166249. Aberdeen Proving Ground, MD, Department of the Army, U.S. Army Environmental Hygiene Agency.
ATSDR. (2012). Toxicological profile for RDX. Agency for Toxic Substances and Disease Registry, Public Health Services, U.S. Department of Health and Human services. Available at: www.atsdr.cdc.gov/toxprofiles/tp78.pdf
Bannon, D.I., Dillman, J.F., Hable, M.A., Phillips, C.S., Perkins, E.J. (2009). Global gene expression in rat brain and liver after oral exposure to the explosive hexahydro-1,3,5-trinitro-1,3,5-triazine (RDX). Chem. Res. Toxicol., 22:620-625.
Cholakis, J.M., Wong, L.C.K., Van Goethem, D.L., Minor, J., Short, R., Sprinz, H., Ellis, H.V. III. (1980). Mammalian toxicological evaluation of RDX. Contract No. DAMD17-78-C-8027. Kansas City, MO, Midwest Research Institute.
Crouse, L.C.B., Michie, M.W., Major, M.A., Johnson, M.S., Lee, R.B. and Paulus, H.I. (2006). Subchronic oral toxicity of RDX in rats. (Toxicology study no. 85-XC-5131-03), Aberdeen Proving Ground, MD, U.S. Army Center for Health Promotion and Preventative Medicine. [as cited in ATSDR, 2012]
Etnier, E.L. (1989). Water quality criteria for hexahydro-1,3,5-trinitro-1,3,5-triazine (RDX). Regul. Toxicol. Pharmacol., 9: 147-157.
Gadagbui, B., Patterson, J., Rak, A., Kutzman, R.S., Reddy, G. and Johnson, M.S. (2012). Development of a relative source contribution factor for drinking water criteria: The case of hexahydro-1,3,5-trinitro-1,3,5-triazine (RDX). Hum. Ecol. Risk Assess. 18(2): 338-354.
Gong, P., Inouye, L.S. and Perkins, E.J. (2009). Comparative neurotoxicity of two energetic compounds, hexanitrohexaazaisowurtzitane and hexahydro-1,3,5-trinitro-1,3,5-triazine, in the earthworm eisenia fetida. Environ. Toxicol. Chem., 26(5): 954-959.
Hart, E.R. (1976). Two year feeding study in rats. Contract No. N00014-73-C-0162. ADAO40161.Office of Naval Research. Kensington, MD, Litton Biotetics Inc.
Health Canada. (1994). Human health risk assessment for priority substances. Canadian Environmental Protection Act. Available from
www.hc-sc.gc.ca/ewh-semt/pubs/contaminants/approach/index-eng.php
Health Canada. (in preparation). Canadian exposure factors used in human health risk assessments. Health Canada, Ottawa, Ontario.
HSDB. (2005). Cyclonite CASRN: 121-82-4. Hazardous Substances Data Bank.
Krishnan, K. and Carrier, R. (2008). Approaches for evaluating the relevance of multiroute exposures in establishing guideline values for drinking water contaminants. J. Environ. Sci. Health. C. Environ. Carcinog. Ecotoxicol. Rev., 26(3): 300-316.
Krishnan, K. and Carrier, R. (2013). The use of exposure source allocation factor in the risk assessment of drinking-water contaminants. J Toxicol Environ Health B Crit Rev., 16(1):39-51.
Krishnan, K., Crouse, L.C.B., Bazar, M.A., Major, M.A. and Reddy, G. (2009). Physiologically based pharmacokinetic modeling of cyclotrimethylenetrinitramine in male rats. J. Appl. Toxicol., 29: 629-637.
Lapointe, M-C., Martel, R. and Diaz, E. (2017). A Conceptual Model of Fate and Transport Processes for RDX Deposited to Surface Soils of North American Active Demolition Sites. J. Environ. Qual., 46(6):1444-1454.
Levine, B.S., Furedi-Machacek, E.M., Rac, V.S., Gordon, D.E. and Lish, P.M. (1983). Determination of the chronic mammalian toxicological effects of RDX: twenty-four month chronic toxicity/carcinogenicity study of hexahydro-1,3,5-trinitro-1,3,5-triazine (RDX) in Fischer 344 rats. Phase V. Vol. 1. Contract No. DAMD 17-79-C-9161. ADA 160774. U.S. Army Medical Research and Development Command. Chicago, IL, IIT Research Institute.
Lish, P.M., Levine, B.S., Marianna-Furedi, E.M., Sagartz, J.M. and Rac, V.S. (1984). Determination of the chronic mammalian toxicological effects of RDX: twenty-four month chronic toxicity/carcinogenicity study of hexahydro-1,3,5-trinitro-1,3,5-triazine (RDX) in the B6C3F1 hybrid mouse. Final report. Phase VI, Vol.1. Contract No. DAMD 17-79-C-9161. ADA 160774. U.S. Army Medical Research and Development Command. Chicago, IL, IIT Research Institute.
Oxley, J.C., Smith, J.L., Kirschenbaum, L.J. and Marimganti, S. (2007). Accumulation of explosive in hair - Part II: factors affecting sorption. J. Forensic Sci., 52(6): 1291-1296.
Parker, G.A., Reddy, G. and Major, M.A. (2006). Reevaluation of a twenty-four-month chronic toxicity/carcinogenicity study of hexahydro-1,3,5-trinitro-1,3,5-triazine (RDX) in the B6C3F1 hybrid mouse. Int. J. Toxicol., 25: 373-378.
Reifenrath, W.G., Kammen, H.O., Reddy, G., Major, M.A. and Leach, G.J. (2008). Interaction of hydration, aging, and carbon content of soil on the evaporation and skin bioavailability of munition contaminants. J. Toxicol. Environ. Health A, 71(8): 486-494.
Sweeney, L.M., Gut Jr, C.P., Gargas, M.L., Reddy, G., Williams, L.R. and Johnson, M.S. (2012a). Assessing the non-cancer risk for RDX (hexahydro-1,3,5-trinitro-1,3,5-triazine) using physiologically based pharmacokinetic (PBPK) modeling. Reg. Toxicol. Pharmacol. 62:107-114.
Sweeney, L.M., Okolica, M.R., Gut Jr, C.P. and Gargas, M.L. (2012b). Cancer mode of action, weight of evidence, and proposed cancer reference value for hexahydro-1,3,5-trinitro-1,3,5-triazine (RDX). Reg. Toxicol. Pharmacol. 64:205-224.
US EPA. (1988). Health Advisory for hexahydro-1,3,5-trinitro-1,3,5-triazine (RDX). United States Environmental Protection Agency, Office of Drinking Water, Washington, D.C.
US EPA. (2018). Toxicological review of hexahydro-1,3,5-trinitro-1,3,5-triazine (RDX) (CASRN 121-82-4) in support of the Integrated Risk Information System (IRIS). United States Environmental Protection Agency, Office of Research and Development, National Center for Environmental Assessment. Washington, D.C.