Drinking water screening value for perchlorate – Technical summary
November 2020
A drinking water screening value of 0.004 mg/L (4 µg/L) is established for perchlorate.
Screening values
Health Canada's screening values identify limits for contaminants in water that could be used as a source of drinking water. A lifetime of exposure to these contaminants up to the screening value, both by drinking the water or by using it for showering or bathing, is not expected to increase health risks for any Canadian, including children.
Screening values are established for contaminants that are not commonly found in Canadian drinking water (either source or treated) and therefore Guidelines for Canadian Drinking Water Quality are not established. Health Canada establishes screening values for contaminants at the request of federal departments, provinces and territories (jurisdictions). These requests are usually made when there is a concern for human health because the presence of a contaminant is suspected or detected in local source water and that contaminant does not have an established limit in drinking water. Since 2020, the technical summaries for screening values are typically published online when Health Canada expects that screening values may be needed by more than one stakeholder or jurisdiction.
Screening values do not replace or supersede existing regulations. However, screening values may help jurisdictions and the public understand the potential health effects of a contaminant.
Screening values are based on a review of scientific research and international regulatory information available at the time of their development. In addition, screening values are externally peer-reviewed to ensure scientific integrity.
Health Canada is committed to keeping pace with new science, including the potential health risks from contaminants that are not typically found in drinking water and do not have Guidelines for Canadian Drinking Water Quality. To this end, Health Canada includes contaminants with screening values in its cyclical prioritization of contaminants for full guideline development.
Exposure considerations
Identity and sources
Perchlorate is used in various industrial and military applications including nitrate fertilizers, solid rocket fuels, munitions, explosives, fireworks, road flares and air bags (ATSDR, 2008; WHO, 2016). Perchlorate can also occur naturally in nitrate deposits and potash ore, but generally in low concentrations (ATSDR, 2008). In water, perchlorate salts easily dissolve to anions that are stable and mobile (Table 1; Cal EPA, 2015). In soil, the salts do not adsorb to soil particles or sediments (NRC, 2005; Cal EPA, 2015). Vaporization of the salts is not expected based on their vapour pressure (Table 1). However, the anions may be found in the air as a result of military activities (Cal EPA, 2015).
Property |
Ammonium perchlorate |
Sodium perchlorate |
Potassium perchlorate |
Perchloric acid |
|---|---|---|---|---|
Chemical Abstract Service Register Number |
7790-98-9 |
7601-89-0 |
7778-74-7 |
7601-90-3 |
Molecular Weight (g/mol) |
117.49 |
122.44 |
138.55 |
100.47 |
Water Solubility (g/L at 25°C) |
249 |
2100 |
20.9 |
Miscible in water |
Vapour Pressure (at 25°C) |
Very low |
Very low |
Very low |
6.8 mm Hg |
Log Kow |
-5.84 |
-7.18 |
-7.18 |
-4.63 |
ATSDR, 2008.
Exposure
The primary route of exposure to perchlorate is ingestion of contaminated drinking water or food grown with contaminated water (Cal EPA, 2015). Food is estimated to contribute more to the total perchlorate exposure (Cal EPA, 2015). Perchlorate has been detected in a wide variety of foods including fruits, vegetables, and grains as well as in dairy and human breast milk (Pearce et al., 2007; Murray et al., 2008; EFSA et al., 2017). Exposure can also occur through consumption of drinking water even though perchlorate levels are usually low. A survey of Canadian drinking water supplies found that over 99% of samples had perchlorate levels below 1 μg/L with a maximum concentration of 3.2 μg/L in a domestic well (Environment Canada, 2009).
Health considerations
Kinetics
In both humans and experimental animals, ingested perchlorate is readily absorbed through the gastrointestinal tract into the systemic circulation (WHO, 2016). Absorption of perchlorate through skin is very limited (US EPA, 2009). Within the body, the chemical is distributed to the gastric mucosa, salivary glands, thyroid, mammary glands, placenta, choroid plexus and kidney (Cal EPA, 2015; WHO, 2016). Perchlorate can also pass through the placental barrier (Zhang et al., 2016). Perchlorate is not metabolized and more than 60% to 90% of the ingested dose is primarily excreted in the urine (Greer et al., 2002; NRC, 2005; Braverman et al., 2006; WHO, 2016). The elimination half-life of ingested perchlorate in humans ranges from 6.0 to 9.3 hours (Lamm et al., 1999; Greer et al., 2002; NRC, 2005).
Health effects
Perchlorate blocks iodide uptake in the thyroid gland in both humans and experimental animals. Thus, the primary effects observed following perchlorate exposure pertain to altered thyroid functions, including changes in the production of thyroid hormones, and potentially adverse neurological effects. Data from human and animal studies do not suggest an increased risk of cancer from perchlorate exposure.
Clinical studies in humans describe the inhibition of iodide uptake in the thyroid caused by oral exposure to perchlorate (Brabant et al., 1992; Lawrence et al., 2000; Lawrence et al., 2001; Greer et al., 2002; Braverman et al., 2006). The most comprehensive study included multiple testing doses in healthy human volunteers (Greer et al., 2002). In this study, daily oral doses of 0.007, 0.02, 0.1 and 0.5 mg perchlorate/kg body weight (bw) per day were administered in drinking water in 37 healthy male and female volunteers for 14 days. Four males and four females for each group were exposed to 0.02, 0.1 and 0.5 mg perchlorate/kg bw per day in 400 mL drinking water. An additional male and female for each group was exposed to the same doses at four set times throughout the day (in 100 mL drinking water each) along with seven other individuals (six females and one male) exposed to 0.007 mg perchlorate/kg bw per day. Measurements of iodide uptake were taken before (8 and 24 hours before), during (on days 2 and 14) and after (on day 15) exposure to perchlorate. Decreases in iodide uptake in comparison to baseline values taken before perchlorate exposure were measured at all doses, with significant findings at the three highest doses. The effects were similar on days 2 and 14, but not significantly different from the baseline on day 15. Levels of total thyroxin (T4), free T4, total triiodothyronine (T3) and thyroid stimulating hormone (TSH) measured throughout the experiments were in the normal range except for one woman in the lowest exposure dose group.
Environmental studies found associations between increased urinary perchlorate levels and either increased TSH or decreased thyroid hormones (T3, T4) (NRC, 2005; SAB, 2013; Cal EPA, 2015). Occupational exposure studies found no significant effects of perchlorate exposure and markers of thyroid function in exposed workers at levels up to 0.48 mg/kg bw per day (Gibbs et al., 1998; Lamm et al., 1999).
Several studies in humans have investigated the effects of maternal perchlorate exposure through drinking water on newborns by measuring thyroid hormone levels (NRC, 2005; SAB, 2013; Cal EPA, 2015). Of these studies, Brechner et al. (2000) reported effects at the lowest concentrations, as measured by significant decreases in T4 and a 27% increase in TSH in newborns exposed in utero to 4-6 ppb perchlorate in drinking water. The long-term consequences of perchlorate exposure in utero and early in life are not known. However, alterations in fetal thyroid hormone have been associated with adverse cognitive developmental effects (Pop et al., 2003; Vermiglio et al., 2004; Kooistra et al., 2006).
The carcinogenicity of perchlorate has been examined in two human studies of exposure through drinking water. Morgan and Cassady (2002) investigated cancer outcome in the greater Redlands area between 1988 and 1998, where drinking water is known to be contaminated with perchlorate (5-98 ppb) and trichloroethylene (0.09-97 ppb). There were no significant differences between the overall numbers of observed cancers in comparison to expected cancers (standardized incidence ratio [SIR] = 0.97, 99% confidence interval [CI] = 0.93-1.02). Modest increases in skin melanoma and uterine cancer were observed, whereas fewer cases were observed for cancers of the lung and bronchus, as well as for cancers of the colon and rectum. All other cancers, including thyroid, bladder, breast, cervix, kidney and prostate cancers, were not affected. Li et al. (2001) investigated the prevalence of thyroid cancer in Clark County, where drinking water contains 4-24 ppb perchlorate, in comparison to a region with no perchlorate contamination (Washoe County). The relative risk was 0.75 (95% CI = 0.35-1.59) indicating no increased risk of thyroid cancer.
While the mechanisms involved in the function and regulation of the thyroid in rats are qualitatively similar to those in humans, biochemical and physiological differences affect their responses to perchlorate (Cal EPA, 2015). Therefore, studies in rats are not great models for quantitatively assessing human health risk associated with perchlorate exposure and are not discussed in detail here, rather they provide qualitative support on potential adverse effects. Animal exposure to perchlorate results in adverse effects on the thyroid (changes in hormone regulation, thyroid weight, hyperplasia, hypertrophy and neurological effects); these changes are generally not associated with carcinogenic outcome or reproductive effects (NRC, 2005; Cal EPA, 2015; WHO 2016).
Mode of action
Like thiocyanate, nitrate and selenium cyanates, perchlorate competitively binds to the Sodium-Iodide-Symporter to block iodide uptake (Schlosser, 2016). In humans and animals, the proposed mode of action (MOA) for perchlorate exposure is iodide uptake inhibition to altered thyroid hormone concentrations (TSH, T3 and T4) and potentially to neurodevelopmental outcomes, with fetuses and infants being the most vulnerable (NRC, 2005; SAB, 2013; Cal EPA, 2015). Other vulnerable populations include people with thyroid disease and individuals requiring more iodine, including pregnant and lactating women.
Iodide is an essential component of thyroid hormones, which are necessary in controlling basal metabolism and proper differentiation and development of neuronal cells; inhibition of iodide uptake is associated with decreased thyroid hormones production that results in adverse neurodevelopmental effects (SAB, 2013). However, there is no human study that evaluated the direct relationship between environmental perchlorate exposure and adverse neurodevelopmental outcomes, although scarce rodent data suggested an inverse association (SAB, 2013). Using the key precursor event of inhibiting iodide uptake (rather than neurological effects) is a conservative approach to derive a screening value.
Selection of key study
Neurodevelopmental effects are the key endpoints of concern based on the database of information in humans and experimental animals on perchlorate exposure and the risk of cancer is limited. No studies on direct perchlorate exposure and human neurodevelopmental effects in the vulnerable populations (fetuses and infants) are available. However, based on the MOA for perchlorate, Greer et al. (2002) was selected as the key study since it showed alterations in an early precursor key event (inhibition of iodide uptake) that could lead to neurodevelopmental effects and this would result in a more conservative point of departure (POD) concentration for perchlorate exposure than a concentration from later key events or neurodevelopmental endpoints. Greer et al. (2002) was also selected as the key study for other agencies (NRC, 2005; Cal EPA, 2015).
Derivation of the screening value
A tolerable daily intake (TDI) for perchlorate in drinking water was first calculated based on decreased iodide uptake in 37 healthy adults as a conservative precursor event to neurodevelopmental effects (Greer et al., 2002). Benchmark dose (BMD) modelling was applied to the dose-response data from Greer et al. (2002). A benchmark response (BMR) of 2 standard deviation (SD) for continuous data (representing a 20% decrease in iodide uptake compared to baseline) was selected since 20% (1) represents a change in intra-individual iodide uptake in humans that is biologically meaningful, considers inherent daily variability, and lower BMRs would be affected by background variation, as determined by the analysis of the clinical studies (Bruce et al., 2018); (2) is more conservative than the estimated 50% BMR level of inhibition not associated with changes in thyroid hormone levels (WHO, 2016); and (3) is within the observable range of significant effects reported in Greer et al. (2002). A lower BMR would result in a POD that is below the range of significant effects, would be overly conservative and beyond the power of the study size. A benchmark dose for a 20% response (BMD2SD) of 0.0297 mg/kg bw per day and the lower 95% confidence limit on the BMD2SD (BMDL2SD) of 0.0109 mg/kg bw per day were modelled (US EPA BMDS: version 2.7.0.4). A tolerable daily intake (TDI) is calculated as follows:
Equation 1

Equation 1 - Text description
This equation calculates the tolerable daily intake for Perchlorate. The tolerable daily intake is calculated by dividing the lowest observed adverse effect level for Perchlorate (0.0109 micrograms per kilogram of body weight per day) by the uncertainty factor of 10, which equals 0.00109 micrograms per kilogram of body weight per day.
where:
- 0.0109 mg/kg bw per day is the BMDL2SD for decreased iodide uptake in adults (Greer et al., 2002).
-
10 is the uncertainty factor: X10 for intraspecies variation to account for variations in the general population, including vulnerable populations. An uncertainty factor for duration of study is not necessary since iodide uptake is inhibited fairly quickly after exposure begins and the inhibition does not increase, or increases only slightly, as exposure continues (Greer et al., 2002). Also, if there is no reduction in thyroidal iodide uptake, there will be no reduction in stored iodide, and extending the exposure duration is not likely to have an impact on the thyroid function. An uncertainty factor for database deficiencies is not needed since human data is consistent across multiple studies and the POD is based on a precursor, non-adverse, biochemical event. Clinical studies in healthy adults, studies of long-term treatment of hyperthyroidism with perchlorate and studies of occupational/environmental exposures add confidence to the overall database. Since rats are poor models for quantitative assessments, further studies in these models would not be beneficial.
Based on the above TDI, a screening value can be derived as follows:
Equation 2
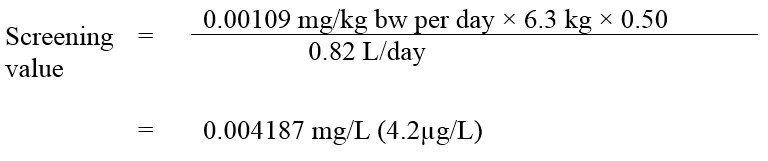
Equation 2 - Text description
This equation calculates the perchlorate screening value. The screening value is reached by multiplying the tolerable daily intake of 0.00109 mg/kg bw-day by the mean body weight estimated for a Canadian 0-5 month old infant, which is 6.3 kg, multiplied by 50%, which is the allocation factor, then dividing that number by 0.82 L, which is the estimated daily volume of tap water consumed by an 0-5 month old infant. This equals a screening value of 0.004187 mg/L (4.2 µg/L).
where:
- 0.00109 mg/kg bw per day is the TDI, as derived above.
- 6.3 kg is the average body weight of a 0-5 month old infant (Health Canada, 2019).
- 0.50 is the allocation factor: the proportion of exposure to perchlorate from drinking water, as opposed to other environmental media (i.e., food, air, soil, consumer products); an allocation factor of 50% is recommended when there are only 2 potential significant sources of exposure (Krishnan and Carrier, 2013). For perchlorate, 50% is appropriate since potential exposure is through two main sources: drinking water and consumption of food grown with contaminated water.
- 0.82 L/day is the estimated daily volume of tap water consumed by a 0-5 month old infant (Health Canada, 2019); multi-route exposure assessment (Krishnan and Carrier, 2008) found that dermal and inhalation exposures through showering or bathing represent negligible routes of exposure to perchlorate.
A screening value of 0.004 mg/L (4 µg/L) for perchlorate in drinking water is recommended by Health Canada.
International considerations
Drinking water quality guidelines, standards and/or guidance established by foreign governments or international agencies may vary due to the science available at the time of assessment, as well as the utilization of different policies and approaches, such as the choice of key study, and the use of different consumption rates, body weights and allocation factors. The World Health Organization (WHO) has a regulatory value while the United States (U.S.) and California (Cal) Environmental Protection Agencies (EPA) have non-regulatory values (Table 2). All values are based on inhibitions of iodide uptake in healthy adults (Greer et al., 2002) and an uncertainty factor of 10, but differ in their selection of the POD, use of adult or infant body weight/consumption values and relative source contribution values (RSC; corresponds to Health Canada's allocation factor).
Source |
Value (mg/L) |
Key Effect |
POD (mg/kg per day) |
UF |
Other Details |
|---|---|---|---|---|---|
Regulatory |
|||||
WHO, 2016 |
0.07 |
Inhibition of iodide thyroid uptake in humans (Greer et al., 2002) |
BMR 50% |
10x intraspecies (account for vulnerable population) |
Used adult body weight and consumption values; RSC 0.20 |
Non-regulatory |
|||||
Cal EPA, 2015 |
0.001 (PHG) |
Same as above |
BMR 5% |
Same as above |
Used infant body weight and consumption values; RSC 0.74 |
US EPA, 2009 |
0.015 (interim HA) |
Same as above |
NOEL |
Same as above |
Used adult body weight and drinking water intake; RSC 0.62 |
Health advisory (HA); Public health goal (PHG); Relative source contribution (RSC); Point of departure (POD); No observed effect level (NOEL); Uncertainty factor (UF).
Contact us
For more information, please contact us at hc.water-eau.sc@canada.ca.
References
ATSDR. (2008). Toxicological profile for perchlorates. Agency for Toxic Substances and Disease Registry. United States Department of Health and Human Services, Atlanta, GA.
Brabant, G., Bergmann, P., Kirsch, C.M., Kohrle, J., Hesch, R.D. and von zur Muhlen, A. (1992). Early adaptation of thyrotropin and thyroglobulin secretion to experimentally decreased iodine supply in man. Metabolism, 41(10): 1093-1096.
Braverman, L.E., Pearce, E.N., He, X., Pino, S., Seeley, M., Beck, B., Magnani, B., Blount, B.C. and Firek, A. (2006). Effects of six months of daily low-dose perchlorate exposure on thyroid function in healthy volunteers. J. Clin. Endocrinol. Metab., 91(7): 2721-2724.
Brechner, R.J., Parkhurst, G.D., Humble, W.O., Brown, M.B. and Herman, W.H. (2000). Ammonium perchlorate contamination of Colorado River drinking water is associated with abnormal thyroid function in newborns in Arizona. J. Occup. Environ. Med., 42: 777-772.
Bruce, G.M., Corey, L.M., Pearce, E.N., Braverman, L.E. and Pleus, R.C. (2018). Determination of thresholds of radioactive iodine uptake response with clinical exposure to perchlorate: A pooled analysis. J. Occup. Environ. Med., 60(4): e199-e206.
Cal EPA. (2015). Public health goal for perchlorate in drinking water. Office of Environmental Health Hazard Assessment. California Environmental Protection Agency, California, USA.
EFSA, Arcela, D., Marco, B. and Francesco, V. (2017). Dietary exposure assessment to perchlorate in the European population. EFSA journal: 15(10):5043. European Food Safety Authority. doi: 10.2903/j.efsa.2017.5043.
Environment Canada. (2009). National assessment of groundwater quality in Canada (Pre-release version for government review and comment). National Water Research Institute, Groundwater Assessment and Remediation Section and the University of Waterloo, Department of Earth Sciences. Pages: 1-54.
Gibbs, J.P., Ahmad, R., Crump, K.S., Houck, D.P., Leveille, T.S., Findley, J.E. and Francis, M. (1998). Evaluation of a population with occupational exposure to airborne ammonium perchlorate for possible acute or chronic effects on thyroid function. J. Occup. Environ. Med., 40: 1072-1082.
Greer, M.A., Goodman, G., Pleus, R.C. and Greer, S.E. (2002). Health effects assessment for environmental perchlorate contamination: The dose response for inhibition of thyroidal radioiodine uptake in humans. Environ. Health Perspect., 110(9): 927-937.
Health Canada. (in preparation). Canadian exposure factors used in human health risk assessments.
Kooistra, L., Crawford, S., van Baar, A.L., Brouwers, E.P. and Pop, V.J. (2006). Neonatal effects of maternal hypothyroxinemia during early pregnancy. Pediatrics, 117(1): 161-167.
Krishnan, K. and Carrier, R. (2008). Approaches for evaluating the relevance of multiroute exposures in establishing guideline values for drinking water contaminants. J. Environ. Sci. Health. C. Environ. Carcinog. Ecotoxicol. Rev., 26(3): 300-316.
Krishnan, K. and Carrier, R. (2013). The use of exposure source allocation factor in the risk assessment of drinking-water contaminants. J. Toxicol. Environ. Health B Crit. Rev., 16(1): 39-51.
Lamm, S.H., Braverman, L.E., Li, F.X., Richman, K., Pino, S. and Howearth, G. (1999). Thyroid health status of ammonium perchlorate workers: a cross-sectional occupational health study. J. Occup. Environ. Med., 41: 248-260.
Lawrence, J.E., Lamm, S.H., Pino, S., Richman, K. and Braverman, L.E. (2000). The effect of short-term low-dose perchlorate on various aspects of thyroid function. Thyroid, 10(8): 659-663.
Lawrence, J., Lamm, S. and Braverman, L.E. (2001). Low dose perchlorate (3 mg daily) and thyroid function. Thyroid, 11(3): 295-295.
Li, F.X., Squartsoff, L. and Lamm, S.H. (2001). Prevalence of thyroid diseases in Nevada counties with respect to perchlorate in drinking water. J. Occup. Environ. Med., 43: 630-634.
Morgan, J.W. and Cassady, R.E. (2002). Community cancer assessment in response to long-term exposure to perchlorate and trichloroethylene in drinking water. J. Occup. Environ. Med., 44(7): 616-621.
Murray, C.W., Egan, S.K., Kim, H., Beru, N. and Bolger, P.M. (2008). U. S. Food and Drug Administration's Total Diet Study: dietary intake of perchlorate and iodine. J. Expo. Sci. Environ. Epidemiol., 18(6): 571-580.
NRC. (2005). Health implications of perchlorate ingestion. National Academies Press, Washington, DC.
Pearce, E.N., Leung, A.M., Blount, B.C., Bazrafshan, H.R., He, X., Pino, S., Valentin-Blasini, L. and Braverman, L.E. (2007). Breast milk iodine and perchlorate concentrations in lactating Boston-area women. J. Clin. Endocrinol. Metab., 92(5): 1673-1677.
Pop, V.J., Brouwers, E.P., Vader, H.L., Vulsma, T., van Baar, A.L. and de Vijlder, J.J. (2003). Maternal hypothyroxinaemia during early pregnancy and subsequent child development: A 3-year follow-up study. Clin. Endocrinol. (Oxf), 59(3): 282-288.
SAB. (2013). SAB advice on approaches to derive a maximum contaminant level goal for perchlorate. Science advisory board. United States Environmental Protection Agency, EPA-SAB-13-004, Washington, D.C.
Schlosser, P.M. (2016). Revision of the affinity constant for perchlorate binding to the sodium-iodide symporter based on in vitro and human in vivo data. J. Appl. Toxicol., 36(12): 1531-1535.
US EPA. (2009). Interim drinking water health advisory for perchlorate. United States Environmental Protection Agency, Office of Water, Washington, D.C. EPA 822-R-08-025.
Vermiglio, F., Lo Presti, V.P., Moleti, M., Sidoti, M., Tortorella, G., Scaffidi, G., Castagna, M.G., Mattina, F., Violi, M.A., Crisa, A., Artemisia, A. and Trimarchi, F. (2004). Attention deficit and hyperactivity disorders in the offspring of mothers exposed to mild-moderate iodine deficiency: A possible novel iodine deficiency disorder in developed countries. J. Clin. Endocrinol. Metab., 89(12): 6054-6060.
WHO. (2016). Perchlorate in drinking-water. Background document for development of WHO guidelines for drinking-water quality. World Health Organization, WHO/SDE/FWC/16.46, Geneva, Switzerland.
Zhang, T., Ma, Y., Wang, D., Li, R., Chen, X., Mo, W., Qin, X., Sun, H. and Kannan, K. (2016). Placental transfer of and infantile exposure to perchlorate. Chemosphere, 144: 948-954.
