Controlling Insect Pests with Low Temperature – Canadian Conservation Institute (CCI) Notes 3/3
(PDF Version, 64 KB)
CCI Note 3/3 is part of CCI Notes Series 3 (The Museum Environment: Biological Factors)
Introduction
Low temperature control is currently used along with other pest management techniques to kill insects within collections. Many insects adapt in order to survive cold temperatures, yet low temperatures have been used to successfully control insect populations in the fur and food industries for over a century. While there is still some lack of knowledge on the specific responses to cold by certain museum insect pests, enough is known to establish guidelines for treatment (Strang, ). These guidelines are based on the lethal temperatures and exposure times published in the entomological literature, on the knowledge of how insects survive cold, and on successful treatment over past decades.
Response to Cold
Cooling a room below +15°C is often very effective in slowing or stopping the growth, feeding, and breeding of museum insect pests, and can be used as a temporary measure to help combat an infestation (Strang, ).
Closer to 0°C, insects become comatose. Cooling to just above 0°C is also known to have a fatal effect on some insect species (e.g., Lasioderma serricorne, Cigarette Beetle), provided the exposure to cold lasts many days. However, most museum insect pests must be cooled to well below 0°C to reduce the time of exposure needed to completely kill the insects populating the object within a reasonably short time (Strang, ).
The two major insect adaptations to cold are to avoid freezing or to tolerate freezing. Insects that avoid freezing do so by increasing the concentration of glycerol and sugars in their tissues in order to lower their freezing point. Sufficient time for metabolic changes (acclimation) and habitat preparation is necessary for these insects to survive such low temperatures. Low temperature extermination methods usually eliminate this period of adaptation.
Insects that tolerate freezing do so by controlling ice formation in their bodies by infusing a special nucleating protein into their body fluid. These insects can remain frozen for months and still recover on thawing.
Insects generally do not maintain these cold protective systems when their environment remains relatively warm (late spring to early autumn conditions) because there is a metabolic cost involved. For this reason, determine the past temperature history of the artifact and allow a preconditioning quarantine period in warm temperatures if the object has come from cool or cold conditions. As a rough guide, allow one month at room temperature to induce insects to flush protective substances from their bodies.
As well, many insects quickly achieve short-term cold adaptation by voiding excreta and water from their bodies. Insects commonly protect themselves this way against temperatures as low as -15°C. The more effective temperatures for killing insects lie below -15°C.
As far as is known, common museum insect pests are neither sufficiently freeze tolerant nor freeze avoidant to withstand the low temperatures recommended for eradication (Strang, ). The museum community's use of freezing to kill cockroaches, clothes moths, silverfish, wood borers, and dermestid beetles has been quite successful. Reported failures are attributable to not reducing the temperature low enough to kill the specific pest, or not maintaining sufficient exposure time (Strang, ).
To kill insects with cold, the artifact has to be cooled to a point lower than which the insects can protect themselves; otherwise, the insects will recover on warming.
A major guideline to follow when using freezing to control insect pests is to expose them to temperatures that drop as low as possible, as quickly as possible, for as long as possible. A practical recommended treatment is -20°C for one week.
Choosing Equipment and Establishing Procedures
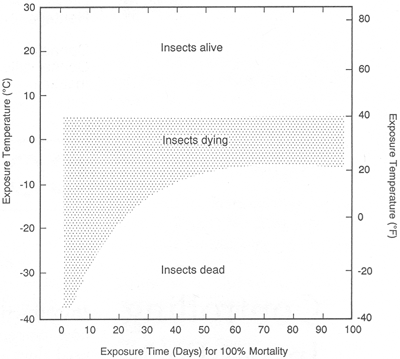
In practice, the most effective freezer systems for killing insect pests operate between −30°C and −40°C. However, common household deep-freezers operating between −20°C and −25°C can be very effective, and are usually less expensive than commercial freezers. Commercial freezers or "top of the line" household chest freezers can usually achieve these temperatures (−29°C). Commercial "ice-cream hardening" freezers in vertical or horizontal configurations are designed to operate at −42°C. They are not much more expensive than standard commercial freezers. Some walk-in laboratory freezers allow you to set the operating temperature. "Air-blast"Endnote 1 freezers are preferable because their forced air circulation increases the cooling rate. It is not necessary to set temperatures below −40°C to increase the effectiveness of using freezing to kill insects.
Once the minimum operating temperature of a freezer is established, choose an appropriate exposure period for insect eradication by referring to Figure 1.
In a freezer, the temperature at the centre of an object drops as a cooling front moves through the object.Endnote 2 A high rate of cooling decreases the likelihood that insects will be able to adapt for cold temperature survival. However, there are limits to how quickly one can lower the temperature.
The only way to have any practical control over the cooling rate is to make an object thinner; for example, lay a textile flat rather than roll it up in the freezer. It is not uncommon for massive or well-insulated artifacts to take half a day to achieve the minimum temperature of the freezer. Minimizing the number of artifacts, increasing air circulation in the freezer, reducing insulating padding and supports, and presenting the greatest surface area are all techniques that increase the rate of cooling and enhance the killing of insects by low temperature.
If a chest freezer is used on a continuous basis, installing interior lids of rigid polystyrene foam will reduce the inflow of warm air when the main door is opened. The foam lids can be lifted slightly and slid to the side for access.
Obtain a record of the minimum temperature for each freezing treatment. There are moderately priced digital indoor/outdoor thermometers with long wire leads that are suitable for determining interior temperature without opening the freezer. (Prevent ice formation and improve cooling by covering the gap with tape where the wire crosses a door seal.)
Occasionally, concern has been expressed over the potential damage to artifacts from exposing them to sub-zero temperatures. Ice damage will not occur within objects that have equilibrium moisture contents (EMC) that conform to normal museum environmental conditions (Strang, ).
The risk of damage increases when some materials are cooled because they lose flexibility and become more brittle. There are some common materials, such as rubber, linseed oil films (oil paint), and synthetic polymers (acrylic paint, soft vinyl), that embrittle when cooled between room temperature and -30°C (Michalski, ; Mecklenburg, ). For this reason, do not cool objects such as paintings on canvas. There is no benefit to going below -40°C, and there is some increased risk to the object. Between -20°C and -40°C there is a very small, but real, possibility of increased craquelure to painted and varnished objects. If the temperature is set between 0°C and -20°C, the risk is negligible (Michalski, ).
Other materials will not become significantly more brittle when the temperature is lowered to -30°C. In general, textiles, furs, feathers, leather, paper, and wood are not threatened by low temperature exposure (Strang, ). Note: Exercise great care when handling cooled objects. There maybe risk of frostbite for the handler, and of physical damage to the object if the potentially slippery object is dropped when moved from the freezer.
Another concern is damage to the object when a change in the relative humidity modifies the object's moisture content, which thereby alters the dimensions of the object and possibly causes cracks. The strongly recommended practice of sealing artifacts in polyethylene bags before cooling allows organic artifacts (e.g., books, fur, wood, etc.) to control their own environment (Strang, ). Bagging to control moisture content is important in order to isolate the artifact from mechanical failure in the refrigeration unit and from condensation once removed from the freezer. Bagging also protects thin objects from the high relative humidity produced by defrosting cycles.Endnote 3
Reducing the volume of air in the bag decreases the amount of moisture that could condense and freeze on the object, but this reduction is not usually necessary and overly tight bags could damage fragile objects. (Sucking the air out of a bag with a straw is strongly discouraged because of the potential to inhale contaminates.) During freezing, the bag cools first and any condensate forms and freezes on it, not on the artifact. In practice, such condensate is rarely seen. If there is concern about this possibility, wrap tissue around water-sensitive objects to reduce the chance of any damage.
Summary
Inspect artifacts, packing material, cases and surroundings periodically. Use sticky trap zone monitors throughout the collection area to improve chances of detection (CCI Notes 3/1, Detecting Infestations. Part I: Detection Methods, and 3/2, Detecting Infestations. Part II: Facility Inspection Procedure and Checklist).
Isolate infested artifacts from the collection to prevent further insect infestation in the museum. Quarantine all incoming artifacts. This is essential for early detection and prevention of infestation. Wrap artifacts in bags before transporting them from collections; this will help contain insects while moving the artifact. As well, it will be easier to associate evidence of excreta and insect skins to a particular object. Note infestations on a plan of the room in order to guide cleaning procedures and to document the incident.
Seal the object in clear polyethylene to allow inspection of the process at all times. Close all bag openings using heat sealing or polyester adhesive tape. Using heat sealing is preferred because it ensures that the air inside the bag is isolated from its surroundings. Adhesive tapes, self-sealing bags or wire ligatures are also used to close bags, but are more prone to failure, and can create a less effective seal. Complete sealing allows organic objects to control their environment within the bag, and prevents moisture gain from condensation during defrost cycles or mechanical failure, or on removal from the freezer (Strang, ).
Precondition the insects. If the object is coming from cool or cold weather outside, some insects may be adapting to cold in autumn or may still be adapted in spring. It may take several weeks of exposure to constant room temperature to completely reduce the insect's ability to withstand cold exposure. If the object has been subject to comfortable room temperatures for weeks, then this preconditioning should not be necessary. As mentioned above, a month's exposure at room temperature should be sufficient.
Cool the object in a freezer. Use a freezer that achieves the coldest temperature available. Household horizontal chest freezers operate between -18°C and -28°C. Commercial freezers for frozen foods will operate near -30°C or -42°C. Even if an immediately fatal temperature is not achieved, many insects will die if they are cooled and maintained somewhat above the temperature at which they freeze. The longer the insects are held in the freezer, the greater the chance that they will be killed. As a minimum measure, keep the artifact in the freezer, set below -20°C, for one week. If possible, include live pest insects in a vial as an indicator. Electronic indoor/outdoor thermometers with a probe on a wire lead are useful for measuring the core temperature of textiles or boxes of books.
Remove bagged artifacts from the freezer. Do not damage the seal on the bags. Allow one day for the object to come to room temperature. All condensation will occur on the outside of the bag if the seal was correctly made and preserved.
Quarantine bagged and sealed artifacts at room temperature to check for reviving insects (several days) or development from unseen eggs and pupae (three weeks or longer). Refer to pest control publications to determine the full period of time to allow insects to emerge from eggs and pupae.
Clean artifacts that are obviously contaminated. Perform this task in an appropriate place (e.g., fumehood, quarantine room, outdoors). This prevents false infestation alarms arising from the sight of insect bodies, or other signs, once the artifacts have been placed in storage.
Final Notes
There is no risk of mould damage to materials sealed in bags provided that the materials were dry (i.e., not damp, at equilibrium with less than 65% relative humidity) when enclosed, and that there is no temperature gradient during storage (e.g., having sat on a cold floor in a warm room). In temperate climates, bagged artifacts can be safely returned to storage in their bags, which will greatly reduce the chances of reinfestation by insects still roaming the storage facility. For further discussion on mould growth on objects stored in plastic bags, see Strang, .
If live insects are found when inspecting the artifact, the object may be cooled a second time. Repeat cooling was used from the s to s when freezers did not normally operate below −10°C to −15°C (Strang ). In practice, a second cooling is often unnecessary if lower temperatures are used. However, this step re-exposes any resistant insects to freezing and should kill them. Failure to kill the insects after this stage indicates that the insects were previously acclimated, the temperature to which they were cooled must be lowered, or the time of exposure must be prolonged.
Further Reading
-
Michalski, S. Retrieval From Cold Storage. Report Nº EDR-1612. Ottawa: Canadian Conservation Institute, .
-
Michalski, S. "Paintings: Their Response to Temperature, Relative Humidity, Shock, and Vibration." In, Art in Transit: Studies in the Transport of Paintings, M.F. Mecklenburg, ed. Washington, D.C.: National Gallery of Art, , pp. 223-248.
-
Michalski, S. "Freezing of Wood and Parchment," Paper Conservation News, nº 80 (), pp. 11-12.
-
Ring, R.A. and D. Tesar. "Adaptations to Cold in Canadian Arctic Insects," Cryobiology, vol. 18 (), pp. 199-211.
-
Strang, T.J.K. "A Review of Published Temperatures for the Control of Pest Insects in Museums," Collection Forum, vol. 8, nº 2 (), pp. 41-67.
-
Strang, T.J.K. "The Effect of Thermal Methods of Pest Control on Museum Collections." In, Biodeterioration of Cultural Property 3. C. Aranyanak and C. Singhasiri, eds. Bangkok: Thailand, (), pp. 334-353.
Written by Thomas J.K. Strang.
Copies are also available in French.
Texte également publié en version française.
© Canadian Heritage,
Cat. Nº NM95-57/3-3-1997E
ISSN 0714-6221