Ethylene glycol final content: chapter 2
2. Summary of Information Critical to the Risk Assessment Under CEPA 1999
2.1 Identity and Physical/Chemical Properties
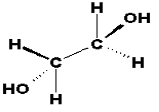
Ethylene glycol (CAS No. 107-21-1) belongs to the simplest group of organic chemicals of the chemical family of glycols, which are characterized by two hydroxyl (OH) groups at adjacent positions in a hydrocarbon chain (see Figure 1).
The physical and chemical properties of ethylene glycol, also known as monoethylene glycol and 1,2-ethanediol, are presented in Table 1. Ethylene glycol is a clear, colourless, odourless, relatively non-volatile, viscous liquid (Nielsen et al. 1993). It has a sweet taste and imparts a warming sensation to the tongue when swallowed (Beasley and Buck 1980). Ethylene glycol has a relatively low vapour pressure (7-12 Pa at 20°C) (Verschueren 1983; Howard 1990) and a low Henry’s Law constant of 5.8 × 10-6 to 6.0 × 10-3 Pa·m3/mol (Hine and Mookerjee 1975; Howard 1990). It is completely miscible in water (Canada 1985; Budavari et al. 1989). It is very hygroscopic and will absorb up to 200% of its weight in water at 100% relative humidity (Budavari et al. 1989). The octanol/water partition coefficient of ethylene glycol is very low (i.e., log Kow = -1.36) (Verschueren 1983; Budavari et al. 1989; Howard 1990).
2.2.1 Production, Importation and Use
The worldwide production capacity of ethylene glycol in 2006 was approximately 19 500 kilotonnes (kt), where the majority went into the production of polyethylene terephthalate (81.5%) (Chinn 2007).
In 2006, there were three companies (in four locations, all in Alberta) manufacturing ethylene glycol in Canada: Alberta & Orient Glycol (in Prentiss), MEGlobal Limited (in Fort Saskatchewan and Prentiss) and Shell Chemicals (in Scotford) (Chinn 2007). The total annual capacity in 2006 from these three companies was 1540 kt: 370 kt for Alberta & Orient Glycol, 720 kt for the two MEGlobal Limited plants combined and 450 kt for Shell Chemicals (Chinn 2007).
Canadaproduced 1410 kt and imported 3 kt of ethylene glycol from the United Statesin 2006. In total, 1200 kt of ethylene glycol were exported in the same year, mainly to China(Chinn 2007).
In Canadain 2006, ethylene glycol was mainly used for anti-freeze mixtures and deicing fluids, and in the production of polyethylene terephthalate (PET) (Chinn 2007). The ethylene glycol used for anti-freeze mixtures and deicing fluids accounted for 70 kt (33% of the domestic demand) (Chinn 2007). This amount has slowly decreased since 2000 with a slight increase from 2005 (Chinn 2007). The amount of ethylene glycol used for the production of PET has increased since 2000 and was 69 kt (31% of the domestic demand) in 2006 (Chinn 2007). PET is manufactured into resins, fibres and films that are subsequently converted into numerous products such as bottles, packaging, and textiles (Chinn 2007). The amount of ethylene glycol used in other applications such as for oil and gas processing and in miscellaneous industrial applications, including its use as a solvent, totalled 70 kt in 2006 (Chinn 2007; CIS 2003).
1. Ethylene glycol is present in various surface coatings, including latex paint, as a slow-evaporating solvent and/or freeze-thaw stabilizer, as well as to provide coalescence and wet-edge control (Chinn 2007; US EPA 1986; NLM 2009). It is noteworthy that information indicates a trend in Canada towards substitution of ethylene glycol in paint formulations with other solvents/stabilizers (ICI Canada 2007). Ethylene glycol is used as a formulant in pesticide products in Canada(e-mail from Pest Management Regulatory Agency [PMRA] to Healthy Environments and Consumer Safety [HECS] September 2009, unreferenced). This substance can also be used in a variety of other products such as in automotive anti-freeze/coolant and wax/polish; brake fluid; as a solvent in adhesives; in floor wax/polish; in caulks and grout, spackling compound, concrete sealers, and shoe polish (NLM 2009; Chinn 2007; US EPA 1986). In addition, ethylene glycol may be used in asphalt emulsion paints; as a coolant and heat transfer fluid; in low-pressure laminates; in glycol diacetate production; in low-freezing dynamite; as a solvent mixture for cellulose esters and ether (a softening agent in cellophane in particular), printing inks, stamp pad inks, ballpoint pen inks, and wood stains; in lacquers; in alkyd resins; in leather dyeing; in textile processing; in humectants; and as a foam stabilizer (ATSDR 2007; Lewis 2007; O’Neil et al. 2006). The quantities used in Canadafor most of these products are unknown.
Ethylene glycol may also be present in pharmaceutical products as a residual solvent. It is classified by the International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use (ICH) as a Class 2 solvent or a solvent that is to be limited in pharmaceutical products (ICH 1997). The concentration limit of 620 ppm of ethylene glycol in pharmaceutical products has been set by both the ICH and the United States Pharmacopoeia (ICH 1997; USPC 2007).
2.2.2 Sources and Releases
Table 2 presents data from the National Pollutant Release Inventory (NPRI) on total industrial annual volumes of releases, disposal and recycling of ethylene glycol from all reporting sources from 1994 to 2005. There are year-to-year fluctuations, but the total of releases, disposal and transfers for recycling (together taken as an indirect measure of usage) generally increased while untreated releases dropped. Thus, untreated releases have declined in this period, both numerically and proportionally.
Annual volumes of untreated releases from all reporting sources of ethylene glycol to air, water and land for the period 1994 to 2005 are shown in Table 3. Since peaking in 1997 at 4698 tonnes, total untreated releases have trended downwards, reaching 2263 tonnes in 2005. While both air and land releases continue to show a downward trend, reported releases to water have increased fivefold since 2000.
Releases to Air
The amount of untreated ethylene glycol released to air has gone down since 1995 (Table 3). While petrochemical manufacture is still the biggest contributor (144 tonnes, or 49%), significant amounts are also released by conventional oil and gas extraction (85 tonnes, or 29%), as well as from the paint and coating industry (20 tonnes, or 7%), resin and synthetic rubber manufacturing (11 tonnes, or 4%), and petroleum refineries (10 tonnes, or 3.5%) (NPRI 2005).
Other contributing industrial activity includes motor vehicle brake and motor manufacturing, printing, and iron ore mining. Together these account for approximately 7% of the total releases to air (NPRI 2005). Use of consumer products containing ethylene glycol may also contribute to releases to air.
Releases to Water
Releases (untreated) to water have gone up significantly since 1994. Releases then were reported at 91 tonnes, mostly from the paper products and the primary steel industry sectors. A sharp increase in releases occurred in 2003 and continued increases were reported up to 2005. Total 2005 releases to water were reported as 572 tonnes, with oil and gas accounting for 446 tonnes (78%). The paper products sector, including pulp mills, reported a significant drop. While this sector was previously reported as the biggest contributor of releases to water, it accounted for only 8 tonnes (1.4%) in 2005. Iron and steel mills accounted for 44 tonnes (8%).
As indicated in the SoS Report, ethylene glycol is used in large volumes for aircraft deicing/anti-icing practices and these volumes are reportedly released to land (see below); however, airport collection facilities and drainage systems may divert substantial quantities to the aquatic environment. This point is presented further in Appendix D.
Releases to Land
NPRI data show that land releases are the biggest component of total untreated releases in 2005. For that year, scheduled air transportation and support activities for air transport account for 95% of untreated releases to land. Other sources of untreated releases to land include chemical pulp mills, diamond mines and cement manufacturing.
Since 2000, several major federal airports have built and/or improved their glycol handling facilities. These include the following locations:
- Ottawa Macdonald-Cartier International: A new biological treatment facility was opened in 2003.
- Toronto Pearson International: The Central Deicing Facility (CDF), built in 1998, was expanded in 1999-2000. Three additional deicing pads were built in 2004. An on-site glycol recycling facility was commissioned in 2005 (GTAA 2005).
- Winnipeg James Armstrong Richardson International: A central deicing facility was opened in 2005. Aeration to Truro Creek, one of two receiving water bodies for airport runoff, was started in 2001.
- Edmonton International: A subsurface-flow (SSF) wetland facility was commissioned in 2000-2001 to treat ethylene glycol-containing aircraft deicing/anti-icing fluids (ADAFs) on-site.
- Vancouver International: work began on a new deicing pad in 2005 and the pad was operational in 2006.
- Montréal-Trudeau: extensive improvements have been made since 2000, including a new enlarged deicing pad.
Releases, disposal and recycling of ethylene glycol from airport operations for the years 1998 to 2005 are shown in Table 4. As can be seen, there is an increase in the total for these three categories from 4577 tonnes in 1998 to 6745 tonnes in 2005. This was due to an increase in the amount of ethylene glycol that was either recycled or disposed of. The term “disposal” indicates that the glycol received some form of treatment before being either released or sent to a municipal wastewater treatment system.
For the same 1998 to 2005 period, untreated releases of ethylene glycol dropped from 2450 tonnes to 1232 tonnes. This represents a decrease of 50%. For the same period, the fraction of ethylene glycol that was released with no treatment compared with the total amount (including recycling and disposal) declined steadily from 53% in 1998 to 18% in 2005.
Releases Underground
Table 3 shows that compared with 1994, when underground injections (mostly on-site) amounted to 77 tonnes, some 93 tonnes were disposed of in this manner in the 2005 reporting year. There was a peak of 422 tonnes injected underground in the 2000 reporting year. The natural gas industry in western Canadais the biggest user of this type of disposal method (NPRI 2005).
2.3 Environmental Fate
The SoS Report presents a detailed discussion of fate in the environment. Once released into the environment, ethylene glycol partitions mainly into surface water or groundwater. It does not bioaccumulate or persist in the environment, primarily due to biodegradation. Half-lives are estimated to typically range from 0.35 to 3.5 days in air, 2 to 12 days in water, 4 to 24 days in groundwater and 2 to 12 days in soil, but may exceed these ranges, depending on environmental conditions. Ethylene glycol has been found to biodegrade rapidly in the aquatic environment and therefore has the potential to induce depletion of the dissolved oxygen (DO) in receiving waters.