Ethylene glycol final content: chapter 3
3. Risk Assessment under CEPA 1999
- 3.1 CEPA 1999 64(A): Environment
- 3.1.1 Environmental Exposure
- 3.1.2 Environmental Effects
- 3.1.3 Risk Quotient Calculations
- 3.1.4 Characterization of Ecological Risk
- 3.1.5 Uncertainties in Evaluation of Ecological Risk
- 3.2 CEPA 1999 64(c): Human Health
- 3.2.1 Population exposure
- 3.2.2 Hazard Characterization
- 3.2.3 Exposure Response Analysis
- 3.2.4 Risk Characterization
- 3.2.5 Uncertainties and Degree of Confidence in Human Health Risk Characterization
- 3.3 Conclusion
3.1.1 Environmental Exposure
Given that releases from airports (releases to land, with subsequent movement to receiving waters) are by far the largest releases of ethylene glycol in Canada and that those releases occur over a limited part of the year (as opposed to industrial releases that occur over the whole year), the highest environmental exposures are expected in the winter and spring in receiving waters adjacent to airports. The ecological assessment therefore focuses on potential exposure resulting from releases from airports.
Figure 2 illustrates the distribution of 3254 individual measurements of ethylene glycol sampled in stormwater at airports across Canadaover the combined 2003-2004 and 2004-2005 deicing seasons. Key percentiles in this distribution and the breakdown by season are summarized in Table 5. Generally, mean glycol concentrations measured over these two years were very similar to the 1997 to 1999 data (as reported in the 2000 SoS Report).
Figure 2: Frequency Distribution of Glycol Concentrations in Airport Stormwater

3.1.2 Environmental Effects
The critical toxicity values presented in the SoS Report (Environment Canada and Health Canada 2000), namely the IC25 of 3268 mg/L for the green alga Selenastrum capricornutum and the subchronic toxicity value of 4732 mg/L for the amphibian Xenopus laevis, still represent the most sensitive measurements of toxicity of ethylene glycol to aquatic organisms. Using IC25 of 3268 mg/L from the most conservative algae test results as the critical toxicity value (CTV) and applying an application factor of 5 to account for interspecies variability and laboratory to field extrapolation, the estimated no-effect value (ENEV) (or predicted no-effect concentration [PNEC])of 654 mg/L is obtained. As well, using 4732 mg/L as a CTV and applying an application factor of 10 to account for the fact that the amphibian study used a non-native species and for uncertainty in laboratory to field extrapolation, the subchronic ENEV becomes 473 mg/L.
3.1.3 Risk Quotient Calculations
Aquatic Biota -- Direct Effects
Based on the most recent available data from all airports for the 2004-2005 season, the levels of ethylene glycol in effluent streams are below 136 mg/L 99% of the time and below 200 mg/L 99.5% of the time from the over 1728 measurements taken (Table 5), with the reported maximum of 2560 mg/L measured in the spring of 2005. The concentrations of ethylene glycol in effluent streams, measured from all the airports, for the most recent 2004-2005 season, were below 100 mg/L 97.6% of the time.
Natural dilution of effluents occurs in the receiving waters and must be considered in calculating the risk to aquatic organisms. The level of dilution varies with each location; therefore, a generic and conservative dilution factor of 10 is applied. The effluent concentrations are assumed to be reduced by one order of magnitude--that is, the isolated maximum effluent concentration of 2560 mg/L referred to above becomes 256 mg/L in the receiving waters.
It is assumed that, under worst-case conditions, high levels of ethylene glycol will be found in the effluent for a period of several days.
Conservative risk quotients for the most recent 2004-2005 season presented in Table 6 and Table 7 are all less than 1; however, the conservative risk quotients using the highest reported concentration for the 1997-1998 and 1998-1999 seasons were about 1 (0.99) for amphibians and 0.71 for algae.
Risk quotients at the 95th and 99th percentile values for the two comparison periods are quite comparable in the case of both algae and amphibians. At the maximum recorded values, the risk quotient is of course lower, again for both algae and amphibians.
Aquatic Biota -- Indirect Effects
One of the potential indirect effects of ethylene glycol is the depletion of dissolved oxygen levels in receiving waters resulting from microbiological degradation. For characterization of risk from indirect effects, the input parameters applied to the Streeter-Phelps oxygen sag model (Streeter and Phelps 1925) include the assumption of complete ice cover and therefore no re-aeration, a dilution factor of 10, an initial DO concentration of 12.4 mg/L and concentrations of ethylene glycol in stormwater (maximums and percentiles) released from 32 airports during the 1997-1998 and 1998-1999 seasons. Oxygen deficit quotients are provided in Table 8. This table has been updated to include comparative data from the latest available Transport Canada Airport Glycol Monitoring Report for the 2003-2004 and 2004-2005 seasons.
From the quotients presented in Table 8, depletion of oxygen below the Canadian Council of Ministers of the Environment (CCME) guideline of 9.5 mg/L is not expected to occur, based on the 99th percentile of effluent releases. However, the potential for oxygen depletion appears to exist when the analysis assumes the worst-case maximum levels for both pre- and post-2000 Transport Canada data. The worst-case risk quotient for the 1997-1999 season was 16.1, whereas for the 2003-2005 period it was lower at 9.1.
Results of a separate probabilistic modeling study that used maximum loadings from individual airport facilities during the 1997-1998 or 1998-99 season and assumed complete ice cover, predicted that DO levels below the CCME DO guideline would occur about 17% of the time under worst-case conditions (Parker 1999). In a more recent study, using actual geophysical and precipitation data from several major Canadian airports, it was shown that ethylene glycol could reduce oxygen levels below the CCME guideline in the airports’ receiving watercourses, even under some conditions of no ice cover, especially at higher concentrations (> 500 mg/L) (Chaulk 2003). It should be pointed out that concentrations of 500 mg/L or higher represent less than 0.5% of recently recorded values at these airports, thus indicating a very low probability for this scenario.
Terrestrial Wildlife -- Direct Effects
The SoS Report (Environment Canada and Health Canada 2000) reported the following toxicity information, which is pertinent to the potential effects of ethylene glycol on terrestrial wildlife. Ethylene glycol poisoning is common among domestic animals and has been reported in cats, pigs, poultry, wildlife and calves (Kersting and Nielsen 1965; Riddell et al. 1967; Black 1983; Amstrup et al. 1989). Ethylene glycol is a slow-acting poison. Even after a massive dose, an animal will be unaffected for 0.5-2 hours post-exposure (Penumarthy and Oehme 1975; Oehme 1983; Beasley 1985; Grauer and Thrall 1986). The oral toxicity of ethylene glycol varies among species. Cats were reported to be the most susceptible to poisoning (Osweiler et al. 1985). The reported lethal dose for cats is only 1.5 mL/kg-bw (1650 mg/kg-bw) (Black 1983), whereas for dogs it is 4.2-6.6 mL/kg-bw (4620-6600 mg/kg-bw) (Beasley and Buck 1980; Oehme 1983; Grauer and Thrall 1986). Osweiler et al. (1985) reported a lethal dose of 2-4 mL/kg-bw (2200-4400 mg/kg-bw) in cats, 4-5 mL/kg-bw (4400-5500 mg/kg-bw) in dogs and 7-8 mL/kg-bw (7700-8800 mg/kg-bw) in poultry. Mallard ducks (Anas platyrhynchos) exposed orally to ethylene glycol demonstrated adverse toxic effects (lowest-observed-effect dose, or LOED) at 2.3 mL/kg-bw (2530 mg/kg-bw) (Stowe et al. 1981). A no-observed-effect level (NOEL) for orally dosed ducks at 1221 mg/kg-bw and lethal doses for poultry at approximately 8000 mg/kg-bw were reported in CA/ICCA (2000).
For the assessment of very short-term exposure to ethylene glycol, applying an assessment factor of 10 to the LD50 for cats of 1650 mg/kg-bw, to account for interspecies and intraspecies variability in sensitivity, gives an ENEV of 165 mg/kg-bw. The 99th-percentile concentration in stormwater runoff from airports from all data from the 1997-1999 seasons is 200 mg/L. Therefore, an animal would have to drink its own weight of stormwater runoff in a short period of time in order to attain the ENEV dose of 165 mg/kg-bw. Assuming a dilution factor of 10, an animal would have to drink about 10 times its own weight of water from a receiving stream in order to attain the ENEV dose.
Elevated concentrations of ethylene glycol in receiving waters may persist for several days. In a 16-week study with rats, the lowest-observed-effect level, based on increased incidence of calcium oxalate crystals, was 150 mg/kg-bw/day (Cruzan et al. 2004). Dividing this value by an assessment factor of 10, to account for interspecies and intraspecies variability in sensitivity, gives an ENEV of 15 mg/kg-bw/day. A 1-kg animal would have to drink about 75 mL/day of airport stormwater runoff containing 200 mg ethylene glycol/L, or 7.5% of its body weight per day, to attain this ENEV. Assuming a dilution factor of 10, an animal would have to drink about 75% of its own weight of water per day from a receiving stream in order to attain the ENEV dose.
Given that a 70-kg human drinks about 3 kg of water per day, or about 4% of his or her body weight and extrapolating this to other species and considering that high levels of ethylene glycol occur for only a few days at a time, it is unlikely that terrestrial wildlife would be harmed by drinking water from receiving waters in the vicinity of airports for a period of time ranging from a few days to several weeks.
3.1.4 Characterization of Ecological Risk
Ethylene glycol is not persistent in air, water or soil and does not accumulate in organisms. The substance has a low inherent toxicity; that is, it causes adverse effects in organisms only at relatively high doses or concentrations.
With respect to releases of ethylene glycol from all sources, as reported to the National Pollutant Release Inventory (NPRI), there is a general downward trend both in total amounts of untreated releases and in the fraction of untreated releases relative to total releases (including disposed of or recycled releases). Based on data from Table 3, the following graph shows the downward trend in total untreated releases from all sources.

In Figure 3 above, “Total Untreated” is the sum of all ethylene glycol released untreated to air, water, land, and underground injection. The largest portion of untreated releases of ethylene glycol is to land, with 95% of those releases originating from airports. Much of the ethylene glycol released subsequently moves to watercourses, such that airports are ultimately the greatest source to water as well. For airports, improved management practices have led to increases in the amounts of ethylene glycol that are treated or disposed of, such that the proportion of untreated glycol to total glycol releases has steadily declined from 53% in 1998 to 18% in the 2005 reporting year.
Based on extensive monitoring data from airports, the risk quotients for effects in freshwater ecosystems indicate that ethylene glycol concentrations likely do not exceed concentrations associated with effects, based on the 99th percentile glycol concentration measured at airports for both direct and indirect effects. Examination of potential indirect effects through oxygen depletion suggests a low potential for dissolved oxygen (DO) levels to drop below the Canadian water quality guideline value (9.5 mg/L DO) under very infrequent maximal loading conditions. It is thus expected that direct or indirect effects are unlikely, especially when consideration is given to the seasonal and transient nature of the releases, the short duration of exposure, and the low ambient temperatures and metabolic rates at the periods of maximum release.
3.1.5 Uncertainties in Evaluation of Ecological Risk
As reported in the SoS Report (Environment Canada and Health Canada 2000), there is a lack of measurement data for ethylene glycol in the ambient environment. However, a large data set of measurements of ethylene glycol in effluents from Canadian airports provides a very good indication of the concentrations being released. Because the conditions of the receiving waters can be highly variable across Canada, some conservative assumptions, including the use of a dilution factor of 10, have been applied in estimating concentrations in waterways. It is expected that an adequate estimate of the concentrations of ethylene glycol in the receiving waters has been obtained.
The use of deicing and anti-icing fluids at airports across Canada will vary from year to year and from region to region, depending on climatic factors. Through control programs by Transport Canada, the Air Transport Association of Canada and local airports and airlines, including the implementation of glycol mitigation plans and glycol operational management plans at the major airports in Canada, the amounts of untreated ethylene glycol being released to the ambient environment have been declining over the past years, and in particular since 2000 with improvements in glycol application and control methods.
While this assessment addressed the potential ecological impacts of ethylene glycol, it is recognized that, while ethylene glycol is the principal ingredient in airplane deicing and anti-icing formulations in Canada, other constituents can be present and can increase the toxicity of the formulation. For example, some deicing formulations can be three to ten times more toxic to certain organisms than ethylene glycol alone (Pillard 1995).
Based on the present analyses, harmful ecological effects are unlikely to result from exposure to ethylene glycol in Canada. Similarly, effects related to reductions in concentrations of dissolved oxygen are unlikely. However, concentrations could reach levels of concern near some Canadian airports a very small percentage of the time under conditions of maximum loading. Continued monitoring of stormwater effluents and receiving waters at airports would permit an ongoing determination of the occurrence or frequency of such occasional high releases.
3.2.1 Population Exposure
The following presentation is limited to identifying recent data considered critical to the quantitative estimation of the exposure to ethylene glycol for various age groups in the general population of Canada and, hence, to a risk assessment under paragraph 64(c) of the Canadian Environmental Protection Act, 1999 (CEPA 1999) (Canada 1999).
The Environmental Monitoring and Reporting Branch of the Ontario Ministry of Environment (formerly the Ontario Ministry of Environment and Energy) used Trace Atmospheric Gas Analyzer (TAGA) 6000 units to carry out several mobile air-monitoring surveys in Windsor, Ontario, in 1991 and 1992 (OMEE 1994). The surveys were performed in order to determine the concentrations of certain substances in air that are not usually measured in routine monitoring programs (OMEE 1994). Ethylene glycol was measured during the 1992 survey that focused on determining concentrations of industrial chemicals found in areas where the general population could be exposed, such as in city parks and near schools (OMEE 1994). Levels of ethylene glycol ranged from < 1 µg/m3 to 75 µg/m3 (detection limit of 1 µg/m3) (OMEE 1994). Based on the maximum value, the upper-bounding estimates of intake for ambient air range from 1.9 to 5.6 µg/kg-bw/day and are shown in Table 9.
Zhu et al. (2004) developed an analytical method for measuring ethylene glycol and propylene glycol in indoor air. This Canadian study involved sampling 9 residential homes (2 apartments and 7 single detached houses), 1 attached residential garage, 1 office and 2 laboratories. The samples were collected from an area that had no industrial point source releases nearby. Ethylene glycol was detected in all locations with levels from the residences (including the attached garage) ranging from 2.0 to 223 µg/m3 and levels from the office and laboratories, ranging from 1.9 to 4.4 µg/m3 (method detection limit of 0.07 µg/m3). Hodgson et al. (2000) sampled indoor air in 11 newly built houses (4 manufactured and 7 site-built houses) in the eastern and southeastern United States. Levels of ethylene glycol were measured at least one month after the completion of the house and ranged from < 23.4-1247 µg/m3. Latex paint was indicated as a source of ethylene glycol emissions. It is noteworthy that in a study of ethylene glycol emission rates from carpets (possibly from an adhesive), the emission rate decreased from 949 µg/m2/h to less than 50 µg/m2/h (detection limit) in 28 days (Wilke et al. 2002).
Based on the maximum value found in residences from the Canadian study (Zhu et al. 2004), the upper-bounding estimates of daily intake from indoor air range from 39 to 117 µg/kg-bw/day and are shown in Table 9.
As no new data on levels of ethylene glycol in food or food packaging were identified, it is assumed that the level of ethylene glycol intake through food has not changed since the publication of the 2000 SoS Report. This source of intake is summarized in Table 9 and Table 10.
Upper-bounding estimates of total daily intake of ethylene glycol for the general Canadian population range from 53 µg/kg-bw/day in adults (60+ years) to 157 µg/kg-bw/day in children (0.5-4 years), as shown in Table 9. For each age group, indoor air is the primary source of exposure.
Sciences International, Inc. was commissioned by the Ethylene Glycol Panel of the American Chemistry Council to characterize potential exposures of the general population to ethylene glycol near an ethylene-glycol manufacturing plant located in Red Deer, Alberta (Sciences International, Inc. 2003). The purpose of this study was in part to respond to the need for more research on exposure to humans located near point source releases, which was an uncertainty identified in the SoS Report.
Sciences International, Inc. used ISC PRIME (Industrial Source Complex Plume Rise Model Enhancements), a site-specific air dispersion model, to estimate potential human exposure to ethylene glycol. The model incorporated ethylene glycol emission data acquired from the facility, as well as five years of meteorological data (Sciences International, Inc. 2003). The predicted maximum 24-hr concentration at nearby residences was 154 µg/m3 (60 ppb). Based on this output, the upper-bounding estimates of daily intake from ambient air ranges from 3.82 in adults (60+ years) to11.55 µg/kg-bw/day (0.5 - 4 years) for individuals living near point sources (Table 10).
The intake values from exposure to soils located near a point source remain the same as those reported in the SoS Report.
Upper-bounding estimates of daily intake of ethylene glycol for a highly exposed population in the immediate vicinity of an industrial point source range from 57 µg/kg-bw/day in adults (60+ years) to 191 µg/kg-bw/day in children (0.5-4 years), as shown in Table 10. For most age groups, the present estimation is approximately two to three times higher than the estimate presented in the SoS Report. Indoor air is the primary source of exposure for all age groups living in the vicinity of a point source.
In the SoS Report, the highest exposure to the general population was considered to be from dermal exposure to ethylene glycol from use of tub and tile cleaners. Dermal exposure estimates were also derived for latex paints, floor polish/wax and auto polish/wax. According to the Canadian Consumer Specialty Products Association (CCSPA) (2002), tub and tile cleaners found in the Canadian market do not contain ethylene glycol. CCSPA (2007) has also confirmed that floor polish/wax products containing ethylene glycol are for use in commercial and institutional settings and are not for consumer use. Therefore, the daily intake of ethylene glycol through dermal exposure is considered to be mainly from auto polish/wax and latex paints. The estimates of exposure to adults from dermal contact to these consumer products were 0.56 and 1.9 mg/kg-bw/day, respectively (Table 11). These estimates would be lower if dermal absorption is taken into account.
Information on levels of ethylene glycol in latex paint in Canadaas well as on types of paint sold (primer versus topcoat) was submitted by the Canadian Paint and Coatings Association (CPCA) (2008). CPCA surveyed ten architectural paint manufacturers in Canada asking them to report the maximum content of ethylene glycol in their interior paint products, and to provide more detailed information on the average ethylene glycol content found in all paint products, including primers, as well as the ratio of sales within various concentration ranges. The results of this survey indicated that the average ethylene glycol content in consumer paints sold in Canada is 1.9%; that approximately 85% of paints contain less than 3% ethylene glycol; and 99.6% of latex paints contain less than 5% ethylene glycol (i.e., less than 0.4% of consumer paints sold in Canada exceed 5% ethylene glycol content). The ethylene glycol levels in primer were considered to be lower than 5%, with an estimated average ethylene glycol content of 1.57%. The survey also showed that based on paint sold in Canada in 2007, 1.2 gallons (4.5 L) of interior primer were sold for every 8.8 gallons (33.3 L) of interior topcoat, indicating that primers are not used as often as topcoats (ratio of approximately 1:9). Based on the information submitted by CPCA, the following concentration values were used to estimate exposures to latex paint: 1.9% (average), 3% and 5% (maximum).
Inhalation exposure to consumer products was not estimated in the SoS Report; however, based on the levels identified in indoor air from Zhu et al., (2004) and Hodgson et al. (2000), it was determined that this route of exposure should be investigated.
Table 11 shows estimated air concentrations from use of latex paint using the US EPA’s Wall Paint Exposure Model (WPEM), information from CPCA on ethylene glycol content in paint and primers, as well as ethylene glycol-specific default values for mass recovery rate (Chang et al. 1997; Chang 2001) and emission decay rate constant for desorption (k1) (US EPA 2001). The highest 8-hr average concentration for a do-it-yourself painter painting a room with two coats of latex paint was 2.5 mg/m3 for paint containing 1.9% ethylene glycol; 4.0 mg/m3 for paint containing 3% ethylene glycol; and 6.7 mg/m3 for paint containing 5% ethylene glycol. The highest 8-hr average concentration for child occupants of the home being painted was 0.7 mg/m3 for paint containing 1.9% ethylene glycol; 1.1 mg/m3 for paint containing 3% ethylene glycol; and 1.8 mg/m3 for paint containing 5% ethylene glycol (Table 11). It should be noted that all Canadian paint products are sold with the following label warning: “Use only under well-ventilated conditions” (CPCA 2008) and the model used to estimate indoor air concentrations assumed a low ventilation rate of 0.45 air changes per hour.
Although CCSPA (2007) has confirmed that floor polish/wax products containing ethylene glycol are restricted to commercial and institutional settings, inhalation exposure to occupants of these facilities is possible and was therefore investigated using the ConsExpo model developed by The National Institute for Public Health and the Environment (RIVM 2006). The mean event concentration while applying floor polish in a residential setting was 2.1 mg/m3 (see Table 11) and is considered an upper-bounding estimate of the concentrations that occupants may be exposed to. It was considered that auto polish/wax is used primarily outdoors and inhalation exposure would be negligible (US EPA 1986).
In the SoS Report, exposure estimates from use of consumer products were derived through amortization of daily exposures over a one-year period, taking event frequency into account. However, to characterize risk from short-term exposures, daily exposure estimates during product use may be a more appropriate basis for comparison. As such, daily exposure estimates are considered the most appropriate exposure metric for the current assessment.
3.2.2 Hazard Characterization
In the period following the release of the 2000 SoS Report, a number of studies have been reported on ethylene glycol toxicity, including those conducted specifically to address uncertainties identified in the report. Key recent mammalian in vitro and in vivo toxicity studies and human studies are presented below for the hazard characterization of ethylene glycol.
In human acute toxicokinetic studies, when four male volunteers were exposed to 31 mg/m3 vaporized labelled ethylene glycol through inhalation for four hours (16 times at 15-minute intervals), 71% to 85% uptake (equivalent to 0.92-1.46 mg/kg-bw) and complete bioavailability were reported. The half-lives of labelled ethylene glycol and glycolic acid were 1.6-2.6 and 1.8-2.9 h, respectively, and urine excretion rates for ethylene glycol, glycolic acid and oxalic acid were 5.5%, 0.77% and 0.10% of inhaled ethylene glycol, respectively (Carstens et al. 2002, 2003; Upadhyay et al. 2008). The half-lives described in those studies are shorter than those derived from cases of ethylene glycol intoxication (3.0-8.6 h) and Upadhyay et al. 2008 propose that this is because ethylene glycol metabolism is not saturated at the lower level exposures. No adverse effect related to exposure was reported by the volunteers.
Dermal absorption was examined in vitro using split-thickness human skin exposed to various aqueous formulations of labelled ethylene glycol for 24 hours. The percent dose absorbed was 0.84%, 1.04% and 0.94% of the applied dose for 100% (undiluted), 50% and 10% ethylene glycol, respectively, and the skin permeability coefficient at all three concentrations was estimated to range from 1.5 x 10-4 to 2.6 x 10-4 cm/h. The average, steady-state flux of ethylene glycol through the skin was essentially linear with concentration at 217, 129 and 15 μg equivalents/cm2xh for the 100%, 50% and 10% aqueous formulations, respectively (Jovanovic 2008 unpublished). Dermal uptake of ethylene glycol was also determined in three male volunteers. Dermal exposure (skin area 66 cm2) to labelled liquid ethylene glycol (undiluted) for up to six hours resulted in a maximum ethylene glycol plasma concentration of 1.1-2.0 μmol/l, and 8.1% and 0.4% of the dose was excreted in urine as ethylene glycol and glycolic acid, respectively. In addition, the skin permeability constant of liquid ethylene glycol was determined to be 2.7 x 10-5 cm/h (Upadhyay et al. 2008). No exposure-related adverse effect was reported.
In short-term toxicity studies, when Wistar and Sprague-Dawley rats were exposed to 1050 mg/kg-bw/day of ethylene glycol through drinking water for 42 and 28 days, respectively, both strains showed increased urinary oxalate and renal calculi (Huang et al. 2000, 2002, 2003; Green et al. 2005). Khan et al. (2002) conducted a similar study using Sprague-Dawley rats exposed to the same dose of ethylene glycol in their drinking water for eight weeks. Calcium oxalate crystal deposition in the kidneys was present in all treated animals. In another study, when mice were orally administered 2200 mg/kg-bw ethylene glycol for seven days, decreased body weight was reported (Mohanasundari et al. 2005).
A recent 16-week subchronic (Cruzan et al. 2004) and a 12-month chronic (ACC 2005, Corley et al. 2008) dietary study on rats allowed for a more in-depth characterization of the repeat dose toxicity of ethylene glycol to mammals. The subchronic toxicity study (Cruzan et al. 2004) was conducted using both Wistar and Fischer 344 (F-344) rats to further investigate strain differences in toxicity. When rats were exposed to 0, 50, 150, 500 and 1000 mg/kg-bw/day ethylene glycol for 16 weeks under identical dietary exposure conditions, the no-observed-adverse-effect level (NOAEL) and the lowest-observed-adverse-effect level (LOAEL) for both strains were reported as 150 and 500 mg/kg-bw/day, respectively. At the LOAEL, increased incidence of calcium oxalate crystals was found in both strains.
Wistar rats were more sensitive to ethylene glycol at higher doses than were F-344 rats; their sensitivity to ethylene glycol at 500 mg/kg-bw/day was comparable to that of F-344 rats at 1000 mg/kg-bw/day. Toxicokinetic studies showed significant strain difference in oxalic acid levels in the kidney at an ethylene glycol dosage level of 500 mg/kg-bw/day and above. At the end of a 16-week exposure period, the oxalic acid levels in kidney tissues of F-344 rats at 500 and 1000 mg/kg-bw/day were 0.033 and 20.6 mg/g, respectively, whereas in Wistar rats these levels were 33.1 and 100.8 mg/g, respectively (Cruzan et al. 2004). There was a clear strain difference in elimination of oxalic acid in urine. Wistar rats exposed to 500 and 1000 mg/kg-bw/day had 21 and 14 times less elimination in urine, respectively, compared to F-344 rats.
A chronic toxicity study (ACC 2005: Corley et al. 2008) with male Wistar rats, the most sensitive strain, is considered a key study for the human health risk assessment of ethylene glycol for chronic exposures. When rats were exposed to 0, 50, 150, 300 and 400 mg/kg-bw/day in their diet for 12 months, the NOAEL was 150 mg/kg-bw/day, and the LOAEL was 300 mg/kg-bw/day based on renal toxicity (e.g., crystal nephropathy) as well as a significant increase in plasma glycolic acid. The highest dose exceeded the maximum tolerable dose (MTD), as all the surviving rats had to be euthanized before the scheduled termination due to excessive weight loss.
Analysis of blood, urine and kidney samples for ethylene glycol metabolites showed a rapid and non-linear increase in oxalic acid levels in the kidneys. At 0, 50, 150, 300 and 400 mg/kg-bw/day, the oxalic acid levels were 5.31, 16.07, 8.72, 6561 and 18 789 µg/g, respectively. While elimination of ethylene glycol followed a linear dose-response relationship, elimination of glycolic acid (GA) was linear up to 150 mg/kg-bw/day. However, it increased non-linearly at 300 mg/kg-bw/day and urinary elimination of oxalic acid was similar to control animals across all the doses.
In the same study, the renal clearance of oxalic acid in Wistar rats was compared with F-344 rats. A significantly higher renal clearance was present in young F-344 rats (6.06 mL/min/kg-bw) compared to young Wistar rats (3.8 mL/min/kg-bw). There was no age difference in renal clearance of oxalic acid among Wistar rats.
Poldelski et al. (2001) exposed isolated mouse proximal tubular segments (PTSs) to ethylene glycol or its main metabolites (glycolate, glycoaldehyde, glyoxylate or oxalate) for 15 or 60 minutes and cell injury was measured by the percentage of lactate dehydrogenase (LDH) release, LDH destruction, adenosine triphosphate (ATP) depletion or membrane phospholipid degradation. Only glyoxalate and glycoaldehyde resulted in significant ATP depletion and LDH release causing cytotoxicity. Ethylene glycol, glycolate and oxalate were not injurious to PTSs. The authors concluded that glyoxalate and glycoaldehyde are the principle metabolites responsible for ethylene-glycol-induced nephrotoxicity.
Similarly, glyoxylate-treated C57BL/6 mice showed decreased superoxide dismutase and increased malondialdehyde expression, markers for oxidative stress and renal epithelial cell injury, respectively, in renal epithelial cells. Mitochondria structure in renal tubular cells was also severely disrupted in regions with calcium oxalate crystals. These particular changes or crystal formation were not observed in ethylene glycol or glycolic acid-treated mice despite induction of hyperoxaluria in the former (Hirose et al. 2008). The lack of oxalate crystal formation in these mice differs from what has been observed in rats and in cases of acute poisoning in humans. Hirose et al. (2008) propose that this may be due to quantitative differences in the metabolic pathway for oxalate precursors and the presence of a defense mechanism against cell injury in mice.
Guo et al. (2005, 2007) conducted in vitro studies with human proximal tubule (HPT) cells and showed that calcium oxalate monohydrate (COM) dose-dependently increased the LDH release while glycolic acid, glyoxylic acid or glycoaldehyde did not increase LDH release at any of the pH levels tested (pH 6, 6.5, 7 or 7.4). The results of these studies suggest that COM, not the other metabolites of ethylene glycol, is toxic to HPT cells at the relevant concentrations.
In another in vitro study (Guo and McMartin 2005), the toxicity of oxalate and calcium oxalate monohydrate (COM) to rat proximal tubular (PT) cells and human proximal tubular (HPT) cells was investigated. COM, not oxalate ion, caused the cytotoxicity in HPT cells, assessed by the release of LDH and percentage of cell death. Similar results were observed in Wistar and F-344 rat PT cells. This study also showed that HPT cells are less sensitive than rat PT cells to COM-induced cytotoxicity. This difference in sensitivity may be due to the ability of rat PT cells to bind to and internalize five times more COM crystals compared to HPT cells (McMartin and Guo 2007). Furthermore, McMartin and Wallace (2005) found that inhibition of mitochondrial respiratory function in PT cells by COM crystals is an important factor for the renal toxicity of ethylene glycol. These in vitro findings of COM toxicity are in agreement with the findings of Cruzan et al. (2004).
As reported in the SoS Report, slight reproductive effects and developmental toxicity, including teratogenicity, have been observed in rodents exposed to ethylene glycol by the oral route at doses greater than those associated with renal effects. For example, developmental toxicity was observed in mice when 11 090 mg/kg-bw/day ethylene glycol was administered orally during gestational day (GD) 7-14 (Schuler et al. 1984). Similar observations were made from other developmental toxicity studies.
Maternal toxicity and fetal malformations were detected when pregnant rats were exposed to ethylene glycol through a subcutaneous (≥1000 mg/kg-bw/day) bolus injection during GD 7-15, but not when the same doses were administered at a slower rate via a subcutaneous implanted infusion pump (Carney et al. 2001, 2002, as cited in the OECD 2009). This difference in toxicity is considered to be due to the saturation of an intermediary step in ethylene glycol metabolism, resulting in the accumulation of glycolic acid in the bolus exposure. Previous investigations have shown that ethylene glycol-induced developmental toxicity is caused by the intermediate metabolite glycolic acid (Carney et al. 1999, 2001).
Wistar rat whole embryo (on GD 9.5-11.5) cultures were exposed to ethylene glycol and its metabolites (glycoaldehyde, glycolic acid, glyoxale, glyoxylic acid and oxalic acid), and the LOAELs for developmental toxicity were reported as 200 mM for ethylene glycol and 3 mM for glycolic acid. Decreased growth parameters (protein content and crown-rump length) were reported at the LOAEL (Klug et al. 2001).
In a toxicokinetic study, when ethylene glycol was administered orally to pregnant and non-pregnant Sprague-Dawley rats, the pregnancy status did not have any impact on the pharmacokinetic parameters of ethylene glycol and its metabolites. The LOAEL for developmental toxicity was 1000 mg/kg-bw/day based on abnormal embryos and a peak blood glycolic acid level of 363 µg/g or 4.8 mM (Pottenger et al. 2001).
No evidence of developmental or reproductive effects was observed in rabbits in the only such study for this species, as reported in the SoS Report. This species difference has been recently addressed and was attributed to toxicokinetics. Glycolic acid levels in rabbit maternal blood and embryo were only 46% and 10% of the respective values in rats. This is possibly due to a slower rate of maternal metabolism and to fundamental differences in disposition of glycolic acid to the embryo (Carney et al. 2008).
In a metabolism study, Booth et al. (2004) exposed rat, rabbit and human liver slices to ethylene glycol. Liver glycolic acid levels in rats were approximately 10 times higher than in rabbits and were not detected in humans. Human liver tissues were also more efficient in further metabolizing glycolic acid to glyoxylic acid. There is therefore less chance for glycolic acid accumulation in humans compared to rats and rabbits, clearly suggesting that, of the three species, humans may be less sensitive because of this difference in hepatic metabolism.
There are no human developmental or reproductive toxicity data available on ethylene glycol. However, physiologically based pharmacokinetic (PBPK) models have been developed for ethylene glycol and its metabolite, glycolic acid, in rats and humans (Corley et al. 2005a, 2005b; Corley and McMartin 2005). They were developed to integrate partition coefficients and metabolic rate constants for ethylene glycol and glycolic acid determined in rat and human tissues in vitro as well as the estimated clearance of each metabolite in urine from applicable in vivo studies. These PBPK models have predicted that it is unlikely to achieve levels of human blood glycolic acid concentrations that could lead to developmental toxicity. Humans would only achieve the threshold for developmental effects determined in rats of 2 mM if they consumed bolus oral doses greater than 350 mg/kg (> 20 g ethylene glycol for a 58 kg female) during the critical window of susceptibility based on simulations of peak maximum blood concentrations of glycolic acid. In addition, the low volatility of ethylene glycol and its potential irritancy limits the possibility of achieving such high blood levels in humans following inhalation exposure (Corley et al. 2005a).
Similar conclusions were drawn in by the U.S. National Toxicology Programme’s Center for the Evaluation of Risks to Human Reproduction. In 2004, they released an Expert Panel Report on the reproductive and developmental toxicity of ethylene glycol (NTP 2004). In this report, by considering all the available data from animal studies and in vitro metabolism studies, the panel concluded that “there is negligible concern of adverse developmental toxicity in humans from ethylene glycol at exposure levels below 125 mg/kg-bw/day.” The Expert Panel also concluded that the concerns for reproductive toxicity in humans are negligible due to lack of evidence for reproductive toxicity in experimental animals.
Available data suggest that there are qualitative similarities in toxicokinetic parameters and mode of action of nephrotoxicity among experimental animals and humans (Cruzan et al. 2004: Corley et al. 2005b and 2008). Furthermore, there is evidence that humans are more efficient in metabolizing ethylene glycol into less toxic metabolites (Hess et al. 2004) and are more effective in eliminating oxalate metabolites than experimental animals (Corley et al. 2008). Although the mode of action of developmental toxicity is not fully understood, the qualitative similarity in toxicokinetics of ethylene glycol and glycolic acid in humans and experimental animals has been well characterized. Hence, this information could be of value in refining the toxicokinetic uncertainty factor in risk assessment.
There is an extensive database on accidental or intentional ingestion of large quantities of ethylene glycol, in the form of anti-freeze, among humans. These case studies (Leth and Gregersen 2005; Krenova and Pelclova 2005; Huttner et al. 2005; Caravati et al. 2005; Morfin and Chin 2005) have reported severe metabolic acidosis, elevated serum anion and osmolar gap and calcium oxalate crystalalluria in individuals acutely exposed to large quantities of ethylene glycol. A more thorough overview of systemic toxicity associated with ingestion of ethylene glycol is described in the SoS Report. While the ingested mixture prevents us from concluding that the effects are solely due to ethylene glycol, the observed toxicity is consistent with those observed in animal studies. The pathological examination of renal tissues showed widespread necrosis of the tubular epithelium and deposition of oxalate crystals in the proximal and distal tubules and collecting ducts. Renal function usually returns to normal in survivors, but permanent renal damage has occurred in some cases (Rumack 2003).
Chronic exposure to lower levels of ethylene glycol, however, is more relevant to this assessment. Renal toxicity has not been observed at relatively low exposure levels related to potential occupational or environmental exposure. For example, no kidney damage was reported in 19 adult male volunteers exposed to 3 to 67 mg/m3 ethylene glycol for 30 days, 20-22 h daily (Wills et al. 1974), in 33 adult male Canadian aircraft deicing workers (Gerin et al. 1997) or in 10 male Finnish auto mechanics (Laitien et al. 1995).
3.2.3 Exposure Response Analysis
Chronic Exposure
Table 12 presents the benchmark dose05 (BMD05) (i.e., the dose estimated to cause a 5% increase in incidence over the background response rate) and the corresponding 95% lower confidence limit (BMDL05) for key toxicity studies presented in the SoS Report (Environment Canada and Health Canada 2000) and the current follow-up report.
The following polynomial model, which describes the probability of the occurrence of the given health effect, was used to derive the BMD05 from the dose-response data:
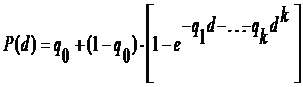 ,
,
where d is dose, k is the number of dose groups in the study, P(d) is the probability of the animal developing the effect at dose d and qi>0, i=1,...,k and d0 are parameters to be estimated.
The models were fit to the data using THRESH (Howe 1995) and the BMD05s were calculated as the dose D which satisfies
![]() .
.
A chi-square lack of fit test was performed for each of the model fits. The degrees of freedom for this test are equal to k minus the number of qis whose estimates are non-zero. A p-value of less than 0.05 indicates a significant lack of fit. The BMDL05 is defined as the lower 95% confidence limit on the BMD05.
The BMD05 of 120 mg/kg/day, based on incidence of crystal nephropathy in male Wistar rats exposed to ethylene glycol for 12 months through diet (ACC 2005: Corley et al. 2008), was considered the most appropriate for use in establishing the tolerable intake (TI). The primary reason for selecting the 12-month study over the 16-week study (Cruzan et al. 2004) is to reduce the uncertainty associated with less-than-chronic exposure. Furthermore, the results of the chronic study indicate that the kidney lesions are not progressing over long-term exposure. The BMDL05 value for this end point was less than twofold lower than the central estimate of the BMD05; the latter, therefore, was used in the calculation of TI.
In the chronic study (ACC 2005; Corley et al. 2008), renal changes were reported in terms of crystal nephropathy only. In contrast, the provisional tolerable intake reported in the SoS Report was established based on the BMD05 for incidence of total tubular damage (i.e., oxalate crystals in kidney, dilated tubules, protein casts) in a subchronic study (Gaunt et al. 1974). Incidence of crystal nephropathy is a more compound-specific histopathological effect, which is considered adverse. For comparison purposes, the BMD05 for incidence of exclusive oxalate crystal formation in the kidney (as opposed to total tubular damage) from Gaunt et al. (1974) is 173.4 mg/kg/day (95% LCL 67.3 mg/kg-bw/day). Similarly, the BMD05 from the 16-week study (Cruzan et al. 2004) is 161 mg/kg-bw/day based on incidence of crystal nephropathy (95% LCL of 72 mg/kg/day).
Based on the BMD05 of 120 mg/kg-bw/day, the tolerable intake (TI) has been derived as follows:
where
120 mg/kg-bw/day is the BMD05 for the incidence of compound-induced crystal nephropathy in male Wistar rats after dietary exposure to ethylene glycol for 12 months (ACC 2005) and
100 is the default uncertainty factor (x 10 for interspecies variation, x 10 for intraspecies variation). Regarding the factors for interspecies and intraspecies variation, the toxicokinetic and toxicodynamic aspects of these uncertainty factors based on available data were not further refined. However, the additional uncertainty factor of 10x, which was applied to account for less-than-chronic exposure in the SoS Report, is no longer warranted given the availability of a chronic study in the sensitive rat strain.
3.2.4 Risk Characterization
The above estimated tolerance intake (TI) for chronic exposure remains protective for potential developmental effects. In the SoS Report, TIs for this end point were derived based on i) the NOAEL for developmental effects in mice (i.e., 500 mg/kg bw/day) divided by an uncertainty factor of 100; and ii) the NOEL of 150 mg/kg-bw/day in the same mice study divided by an uncertainty factor of 100. These values exceed the TI, based on renal effects, of 1.2 mg/kg-bw per day. Furthermore, NTP (2004) has concluded that there is negligible evidence of adverse developmental toxicity in humans from ethylene glycol at exposure levels below 125 mg/kg-bw/day. This is further supported by PBPK model predictions that human blood glycolic acid concentrations are unlikely to reach levels that could lead to developmental toxicity (Corley et al. 2005a).
Based on the information available, upper-bounding estimates of daily intake of ethylene glycol for the general population of Canada, up to 157 µg/kg-bw per day (Table 9), and for a highly exposed population in the immediate vicinity of an industrial point source, up to 191 µg/kg-bw per day (Table 10), are well below the TI of 1200 µg/kg-bw per day.
The SoS Report did not estimate inhalation exposure to consumer products. However, based on the levels identified in indoor air, this route of exposure has been investigated. The predicted highest 8-hr average air concentrations from the use of latex paint ranged from 2.5-6.7 mg/m3 for adult do-it-yourself painters and from 0.7-1.8 mg/m3 for child occupants. Indoor air concentrations would be lower if a higher ventilation rate was used in the model, which might be likely since paint labels instruct users to paint in well-ventilated areas. In contrast, these concentrations could be higher if levels in paint exceed 5% (however, less than 0.4% of consumer paints sold in Canadaexceed this value) or if primer containing similar levels of ethylene glycol is used. These predicted highest air concentration ranges are tenfold or greater below the NOAEL for irritation of 67 mg/m3 identified in a human study (Wills et al. 1974), and are therefore considered protective. Use of this NOAEL as a basis for risk characterization is also considered to be protective of developmental effects as NTP (2004) has concluded that there is negligible concern for adverse developmental toxicity in humans from ethylene glycol at exposure levels below 125 mg/kg-bw/day, and the calculated oral equivalent doses for the ranges of predicted air concentrations would be approximately two orders of magnitude below this level. Confidence in the use of the NOAEL of 67 mg/m3 is further strengthened by PBPK model predictions that human blood glycolic acid concentrations are unlikely to reach levels that could lead to developmental toxicity (Corley et al. 2005a). Results from an inhalation study conducted in CD-1 mice (Tyl et al. 1995), summarized in the SoS Report, were not considered appropriate for use in characterizing risk from inhalation exposure, due to significant study limitations. These limitations included the confounding effects caused by exposure through ingestion and the stress induced by use of a restraint for a nose-only exposure.
According to CCSPA (2007), floor polish/wax products containing ethylene glycol are only used in commercial and institutional settings and are not for consumer use. However, it is possible that the general population may be exposed to ethylene glycol via inhalation in institutional settings. The upper bounding estimate used to represent this route of exposure (2.1 mg/m3) is well below the NOAEL of 67 mg/m3 (Wills et al. 1974) and is not, therefore, of concern. It was considered that auto polish/wax is used primarily outdoors and inhalation exposure would therefore be negligible. (Note: As the dermal exposure estimate from use of auto polish/wax is below the TI, this consumer product scenario is not of concern.)
3.2.5 Uncertainties and Degree of Confidence in Human Health Risk Characterization
In the SoS Report, limitations of the available data precluded development of upper-bounding estimates of daily intake of ethylene glycol by the general population. In the current assessment, additional data permitted derivation of upper-bounding estimates of daily intake of ethylene glycol. However, the intakes from ambient air and from indoor air are each based on a single Canadian study with insufficient samples to ensure representativeness of the Canadian population. No monitoring data for drinking water and soils were identified. No new data on levels of ethylene glycol from food or food packaging were identified either and this remains an area of uncertainty. Thus, estimated total daily intake values for the general population, based on the limited data presented above, could change in cases where drinking water contributes any significant levels of ethylene glycol.
There is moderate confidence in the estimated intake in the vicinity of point sources. The computer air dispersion model (ISC PRIME) is considered to provide a reliable indication of ambient air concentrations of ethylene glycol at the dwelling and property boundary in the vicinity of industrial point sources. With respect to indoor air, it is possible that the indoor air intake value for those residents living near the vicinity of a point source is underestimated, as these values were measured in dwellings not located near industrial point sources. There is a high degree of certainty that the estimates of intake from ingestion of soil by a population exposed due to its proximity to a source of discharge to the atmosphere are upper-bounding.
Overall, there is low to moderate confidence in the estimates of exposure to ethylene glycol from use of consumer products. Based on the information provided by CCSPA (2002), there is high confidence that tub and tile cleaners do not contain ethylene glycol and that floor polish/waxes containing ethylene glycol are targeted to commercial and institutional uses (CCSPA 2002, 2007). However, there is uncertainty about occupant exposures following commercial and institutional uses. There is also some uncertainty regarding the appropriateness of using the ConsExpo residential floor polish scenario to represent potential inhalation exposures to ethylene glycol to occupants of institutional settings. Additionally, data on Canadian-specific ranges and distributions of concentrations in the various products were not available. Information on dermal absorption, reported above, indicates that the assumption of 100% dermal absorption is very conservative and that actual dermal absorption is likely more than an order of magnitude lower than this. As noted in the SoS Report, estimated daily intakes of ethylene glycol by the dermal route from use of consumer products would be several orders of magnitude less than the values presented in Table 11 if skin permeability were taken into account (Environment Canada and Health Canada 2000). There is moderate confidence in the exposure estimates via the inhalation route from latex paint using the US EPA’s Wall Paint Exposure Model and ethylene glycol-specific model parameter inputs. The model is relatively robust and has incorporated results of chamber tests involving ethylene glycol (US EPA 2001). This model was designed specifically to model indoor air concentrations and exposures from painting applications (US EPA 2001). As this model predicts that the highest air concentrations could occur approximately 1.5 days after painting has commenced, other occupant scenarios (e.g., child sleeping or playing in a freshly painted room) could result in higher exposures than the one presented. However, taking into consideration the conservative nature of inputs to the exposure assessment, exposure to ethylene glycol from consumer products is not considered to be of concern.
The degree of confidence in the database on toxicity that serves as the basis for development of the TI for ethylene glycol has increased from the database presented in the SoS Report, but remains in the moderate range. Confidence that the TI developed on the basis of renal effects is protective of other adverse effects of ethylene glycol, such as developmental effects, also remains moderate. The lack of data on progression of renal lesions following chronic exposure in the most sensitive animal model was an area of considerable uncertainty identified in the SoS Report. This has been addressed through conduct of a 12-month study in Wistar rats. It is considered that the chronic study used to derive BMD05 has comprehensively covered the renal histopathology, as renal tissues are the primary target sites for ethylene glycol toxicity. Furthermore, the study was conducted on Wistar rats, the strain of rats most sensitive to ethylene glycol.
3.3 Conclusion
Based on the information presented in this report, ethylene glycol is not entering the environment in a quantity or concentration or under conditions that have or may have an immediate or long-term harmful effect on the environment or its biological diversity, or that constitutes or may constitute a danger to the environment on which life depends.
Based on the information presented in this report, ethylene glycol is not entering the environment in a quantity or concentration or under conditions that may constitute a danger to human life or health.
Therefore, ethylene glycol does not meet any of the criteria under section 64 of CEPA 1999.