Draft screening assessment thiophosphate alkyl esters group
Official title: Draft Screening Assessment - Thiophosphate Alkyl Esters Group
Confidential Accession Numbers:
- 11145‑3
- 11105‑8
Environment and Climate Change Canada
Health Canada
March 2021
Synopsis
Pursuant to section 74 of the Canadian Environmental Protection Act, 1999 (CEPA), the Minister of the Environment and the Minister of Health have conducted a screening assessment of two substances referred to collectively as the Thiophosphate Alkyl Esters (TPAEs) Group. Substances in this group were identified as priorities for assessment as they met categorization criteria under subsection 73(1) of CEPA. Their International Union of Pure and Applied Chemistry (IUPAC) names, or trade names and their Chemical Abstracts Service Registry Numbers (CAS RNFootnote 1) as well as their Confidential Accession Numbers (CAN) are listed in the table below.
| Acronym | Substance name | CAS RNb | CANc |
|---|---|---|---|
| TPAE-1a | Reaction products of 4-methyl-2-pentanol and diphosphorus pentasulfide, propoxylated, esterified with diphosphorous pentaoxide, and salted by amines, C12-14- tert-alkyl | 91745-46-9 | 11145-3 |
| TPAE-2a | Phosphorothioic acid, O,O-dibutyl ester, mixed (C8,C16,C18) alkylamine saltd | Not available | 11105-8 |
a Substance is a UVCB (unknown or variable composition, complex reaction products, or biological materials).
b CAS RN, Chemical Abstracts Service Registry Number.
c CAN, Confidential Accession Number.
d This is a trade name; the substance has IUPAC names for each of its two main constituents.
The two substances described above and addressed in this screening assessment will hereinafter be referred to as TPAE-1 and TPAE-2. It was determined through the Confidential Substance Identity Claim Review Project that the identities of these substances are no longer considered to be confidential business information and are therefore disclosed in this document.
According to information submitted in response to a CEPA section 71 survey, import quantities of 100 000 to 1 000 000 kg for TPAE-1 and 500 kg for TPAE-2 were reported in Canada in 2011. These substances are used in Canada in industrial and automotive lubricants. TPAE-1 is also used in metalworking fluids.
The two substances in this group are composed primarily of alkyl dithiophosphate or thiophosphate anions with primary aliphatic amine counterions. TPAE-1 is the more complex of the two substances, with about ten major components that include dithiophosphate, thiophosphate and phosphate alkyl ester salts, in addition to a neutral thiophosphate component. With the exception of the neutral component of TPAE-1, all components of these substances are ionized under ambient environmental conditions, and some components, notably the aliphatic amine counterions, have surfactant properties.
The whole-substance empirical ecotoxicity data, as well as ecotoxicity data for aliphatic amines, were used to derive critical toxicity values for the substances in this group. These data indicate that both the alkyl (di)thiophosphate ester components and the aliphatic amine components of the TPAEs have high chronic toxicity to aquatic organisms, and the aliphatic amines components have moderate to low chronic toxicity to soil organisms. TPAE-1 was also found to biodegrade very slowly. These substances are expected to persist in the environment long enough to cause chronic toxicity; however, they were not found to be highly bioaccumulative.
Two industrial uses of TPAEs were identified as having the highest potential for releases to the environment: formulation of lubricants and use in metalworking fluids. Environmental concentrations of representative components of TPAE-1 in the aquatic environment associated with releases from these uses, following wastewater treatment, were estimated and compared to predicted no-effect concentrations for aquatic organisms. In addition, the concentration of the aliphatic amine components of TPAE-1 in soils following the application of biosolids from wastewater treatment facilities to soil were estimated and compared to a predicted no-effect concentration for soil organisms. On the basis of these comparisons, TPAE-1 may pose a risk to aquatic and soil organisms from its use in metalworking fluids. TPAE-2 is unlikely to pose a risk to aquatic or soil organisms based on current usage patterns.
Considering all available lines of evidence presented in this draft screening assessment, there is risk of harm to the environment from TPAE-1. It is proposed to conclude that TPAE-1 meets the criteria under paragraph 64(a) of CEPA as it is entering or may enter the environment in a quantity or concentration or under conditions that have or may have an immediate or long-term harmful effect on the environment or its biological diversity. However, it is proposed to conclude that TPAE-1 does not meet the criteria under paragraph 64(b) of CEPA as it is not entering the environment in a quantity or concentration or under conditions that constitute or may constitute a danger to the environment on which life depends.
Considering all available lines of evidence presented in this draft screening assessment, there is low risk of harm to the environment from TPAE-2. It is proposed to conclude that TPAE-2 does not meet the criteria under paragraphs 64(a) or (b) of CEPA as it is not entering the environment in a quantity or concentration or under conditions that have or may have an immediate or long-term harmful effect on the environment or its biological diversity or that constitute or may constitute a danger to the environment on which life depends.
From a human health perspective, TPAE-2 was previously evaluated in the Rapid Screening of Substances with Limited General Population Exposure, which determined that the potential for direct and indirect exposure to the general population was negligible. Therefore, TPAE-2 is considered to be of low concern for human health at current levels of exposure.
No measured data regarding concentrations of TPAE-1 in environmental media were identified in Canada, or elsewhere. However, releases to the environment have been estimated for use of the substance in formulation of lubricants and in metalworking fluids. Critical health effects included effects in the adrenal glands and effects on the nervous system. Comparison of estimates of oral exposure via drinking water with levels associated with critical effects in laboratory studies resulted in margins of exposure that were considered to be adequate to address uncertainties in the exposure and health effects databases.
Considering the information presented in this draft screening assessment, it is proposed to conclude that the two substances in the Thiophosphate Alkyl Esters Group do not meet the criteria under paragraph 64(c) of CEPA as they are not entering the environment in a quantity or concentration or under conditions that constitute or may constitute a danger in Canada to human life or health.
It is therefore proposed to conclude that TPAE-1 meets one or more of the criteria set out in section 64 of CEPA and that TPAE-2 does not meet any of the criteria set out in section 64 of CEPA.
It is also proposed that TPAE-1 meets the persistence criteria but not the bioaccumulation criteria as set out in the Persistence and Bioaccumulation Regulations of CEPA.
1. Introduction
Pursuant to section 74 of the Canadian Environmental Protection Act, 1999 (CEPA) (Canada 1999), the Minister of the Environment and the Minister of Health have conducted a screening assessment of two substances referred to collectively as the Thiophosphate Alkyl Esters (TPAEs) Group to determine whether these substances present or may present a risk to the environment or to human health. The substances in this group were identified as priorities for assessment as they met categorization criteria under subsection 73(1) of CEPA (ECCC, HC [modified 2017]). The substances in this group have similar chemical structures, as they both have alkyl dithiophosphate or alkyl thiophosphate anions with primary aliphatic amine counterions. The first of the two substances addressed in this screening assessment has Chemical Abstracts Service Registry Number (CAS RN) 91745-46-9, and Confidential Accession Number (CAN) 11145-3. The second substance has CAN 11105-8 but does not have a CAS RN. Hereinafter the substances will be referred to as TPAE-1 and TPAE-2, respectively. More information on the identity of these substances, including their International Union of Pure and Applied Chemistry (IUPAC) names or trade names, is provided in Section 2.
It was determined through the pilot phase of the Confidential Substance Identity Claim Review Project that the identities of these substances are no longer considered to be confidential business information. This project stems from the Approach to disclose confidential information and promote transparency in chemicals management (the Approach) (Canada 2018). This policy document sets out several measures to improve the transparency of chemicals management in Canada, including the introduction of a review of confidentiality claims for substance identities after a period of ten years. To implement this part of the Approach, a pilot project was initiated to address 24 substances on the Domestic Substances List (DSL), including TPAE-1 and TPAE-2. Through this process, the nominators of TPAE-1 and TPAE-2 confirmed that they no longer required confidential substance identities for these two substances, and as such, their identities are disclosed in this document.
TPAE-2 was evaluated under the approach applied in the Rapid Screening of Substances with Limited General Population Exposure (ECCC, HC 2018), and was found to be of low concern to human health.
This draft screening assessment includes consideration of information on chemical properties, environmental fate, hazards, uses and exposures, including additional information submitted by stakeholders. Relevant data were identified up to September 2018, with additional targeted literature searches up to March 2020. Empirical data from key studies as well as results from models were used to reach proposed conclusions. When available and relevant, information presented in assessments from other jurisdictions was considered.
This draft screening assessment was prepared by staff in the CEPA Risk Assessment Program at Health Canada and Environment and Climate Change Canada and incorporates input from other programs within these departments. The ecological portions of this assessment have undergone external review. Comments on the technical portions relevant to the environment were received from James Armitage (AES Environmental Services, Inc.), Connie Gaudet, and Meredith Curren. While external comments were taken into consideration, the final content and outcome of the screening assessment remain the responsibility of Health Canada and Environment and Climate Change Canada.
This draft screening assessment focuses on information critical to determining whether substances meet the criteria as set out in section 64 of CEPA by examining scientific information and incorporating a weight of evidence approach and precaution.Footnote 2 This draft screening assessment presents the critical information and considerations on which the proposed conclusions are based.
2. Identity of substances
Both substances in this grouping are of Unknown or Variable composition, Complex reaction products or Biological materials (UVCBs). UVCBs are derived from natural sources or complex reactions and cannot be characterized in terms of constituent chemical compounds because their composition is too complex or variable. A UVCB is not an intentional mixture of discrete substances and is considered a single substance. In Table 2-1 the CAN of the substances are shown alongside the descriptive chemical names, and where available, the CAS RNs.
| Acronym | Substance name | CAS RN | EC No. | CAN |
|---|---|---|---|---|
| TPAE-1 |
Reaction products of 4-methyl-2-pentanol and diphosphorus pentasulfide, propoxylated, esterified with diphosphorous pentaoxide, and salted by amines, C12-14- tert-alkyla (Amines, C12-14-alkyl, reaction products with hexanol, phosphorus oxide (P2O5), phosphorus sulfide (P2S5) and propylene oxide)b |
91745-46-9 | 294-716-2d, 931-384-6 | 11145-3 |
| TPAE-2 | Phosphorothioic acid, O,O-dibutyl ester, mixed (C8,C16,C18) alkylamine saltc | Not available | Not available | 11105-8 |
Abbreviations: CAS RN, Chemical Abstracts Service Registry Number; EC No., European Community number; CAN, Confidential Accession Number.
a Substance name used in REACH substance evaluation report.
b Substance name associated with the CAS RN.
c This is a trade name; the substance has IUPAC names for each of its two main constituents (see Table 2-3).
d This EC No. is linked with the CAS RN; the other EC No. is not linked with the CAS RN.
The composition of TPAE-1 includes a variety of phosphorodithioate/thioate alkyl esters and phosphate alkyl ester structures, all of which are anions with C12-14 tert-alkyl amine counterions, with the exception of one neutral component (Table 2-2). Industry information identifies about 10 major structures in this substance that are present in the commercial product in variable amounts, along with the aliphatic amine cation (Study Submission 2018; Table 2-2). The structures present in the largest molar quantity are the aliphatic amines, as they are the counterion to all other ionic structures present in this UVCB and are estimated to comprise about 40% of the mixture by molar quantity, or about 25% by weight. The data on the substance composition provided to Environment and Climate Change Canada (ECCC) and Health Canada (HC), which was based on chemical analyses of a single sample of this substance using liquid chromatography-mass spectrometry as well as phosphorus nuclear magnetic resonance (Study Submission 2018; ECHA 2019a), did not give the percentages of each of the major structures in the substance; rather percent ranges were assigned to groupings of these structures.
| Structure number | Molecular weight (g/mol)a | Chemical structure drawing |
|---|---|---|
| Aliphatic amine componentb | 185.35 | 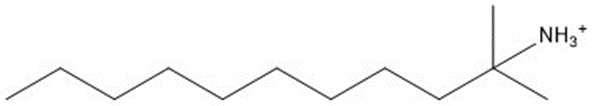 |
| 1 | 356.52 | 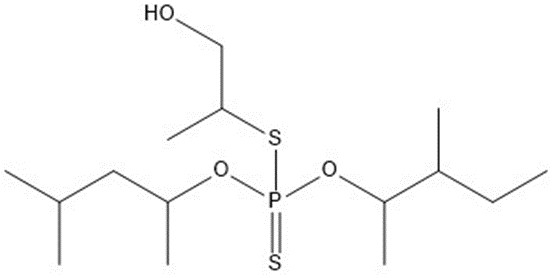 |
| 2 | 436.51 | 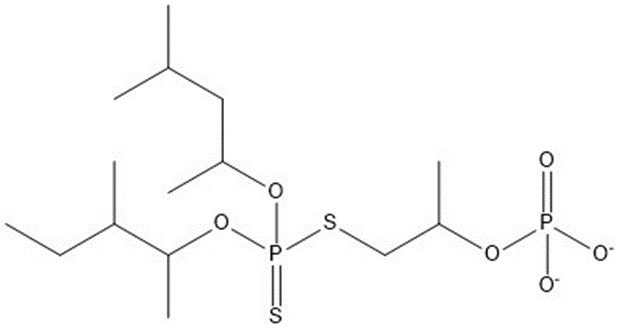 |
| 3 | 775.02 | 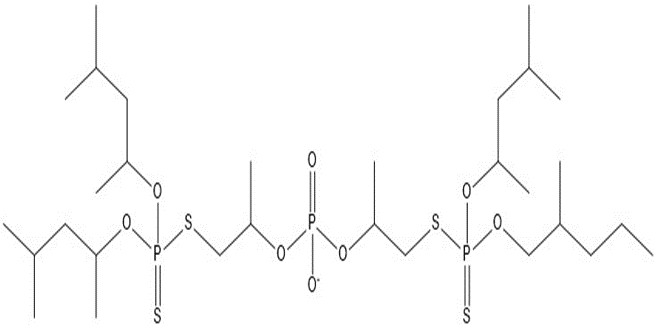 |
| 4 | 520.67 | 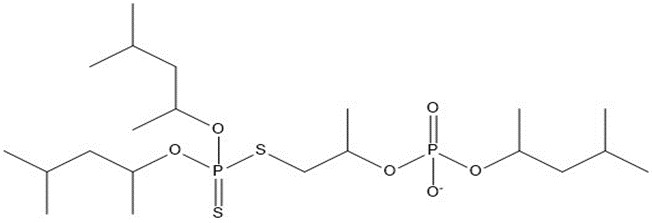 |
| 5 | 282.38 | 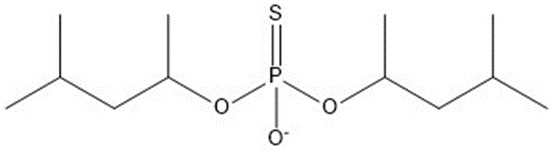 |
| 6 | 346.29 | 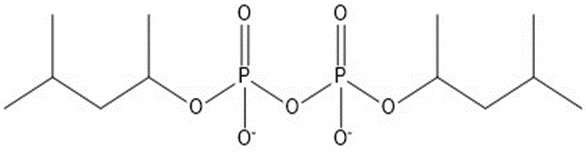 |
| 7 | 586.62 | 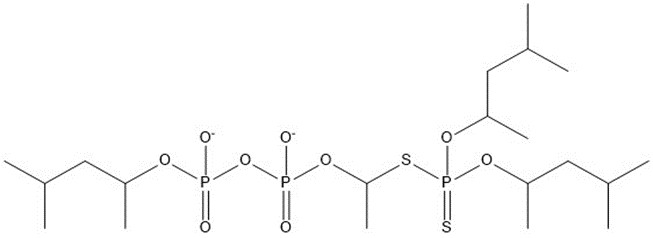 |
| 8 | 855.00 | 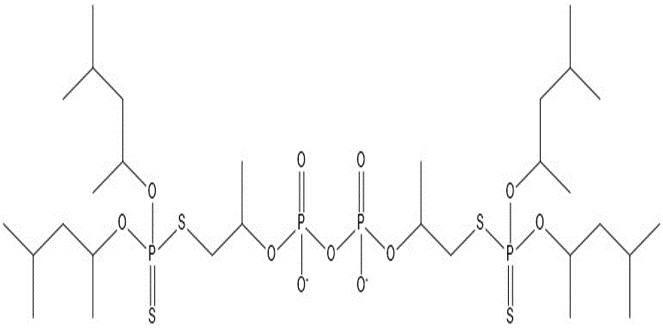 |
| 9 | 182.15 | 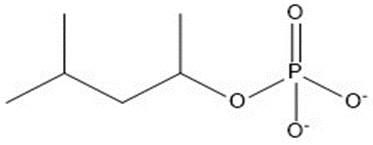 |
| 10 | 250.31 | 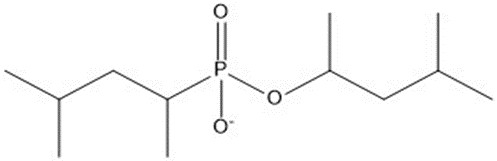 |
a The representative structure weights do not include the weight of the aliphatic amine counterions.
b The representative structure depicted is C12 tert-alkyl amine. The actual aliphatic amine component is a UVCB, C12-14 tert-alkyl amines.
| Constituent | IUPAC name | CAS RN or EC No. |
|---|---|---|
| constituent A | Amines, C16-18-(even numbered, saturated and unsaturated) alkyl, O,O-di-Bu phosphorothioates | EC No. 947-129-7a |
| constituent B | O,O-dibutyl hydrogen thiophosphate, compound with 1-octylamine (1:1) | 93964-99-9 |
Abbreviation: CAS RN, Chemical Abstracts Service Registry Number; EC No., European Community number; IUPAC, International Union of Pure and Applied Chemistry.
a CAS RN has not been assigned.
TPAE-2 is registered under the European Union’s Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH) regulation under each of its two main constituents, as provided in Table 2-3, rather than as a single substance, as described in Table 2-1. However, the individual constituents are only manufactured and distributed together and are not offered for commercial use separately (personal communication, letter from industry stakeholder to Environment and Climate Change Canada, dated November 20, 2017; unreferenced). Constituent A (Table 2-3) accounts for the major portion of TPAE-2, comprising approximately 84% by weight of this substance, while constituent B accounts for approximately 12% by weight. The alkyl thiophosphate component is found in both the A and B constituents of TPAE-2 with the composition of the aliphatic amine component of the constituents differing (Table 2-4). The aliphatic amine component of constituent A is variable; however, the composition of constituent A with the C18 amine representative structure as depicted in Table 2-4 represents 60% to 80% of this constituent (ECHA 2018a). The aliphatic amine component of constituent B is comprised of one major component with less than 10 wt.% impurity. More detailed information on the composition of constituents A and B of TPAE-2 can be found in their REACH dossiers (ECHA 2017, 2018a). The remainder of TPAE-2 (about 4 wt.%) is a mixture of unreacted alkylamines including CAS RN 40165-68-2 [9-octadecen-1-amine, N-(9Z)-9-octadecen-1-yl-, (9Z)-], CAS RN 112-90-3 [9-octadecen-1-amine, (9Z)-] and CAS RN 111-86-4 [1-octanamine] (personal communication, email from industry stakeholder to Environment and Climate Change Canada, dated April 25, 2018; unreferenced).
| Structure | Molecular weight (g/mol)a | Chemical structure drawing |
|---|---|---|
| 11105-8, alkyl thiophosphate (constituent A & B) | 226.27 | ![O-]P(OCCCC)(OCCCC)=S](/content/dam/eccc/images/pded/tpaes/20210118-Table2-4-1.jpg) |
| 11105-8, C18 amine (constituent A) | 269.52 | 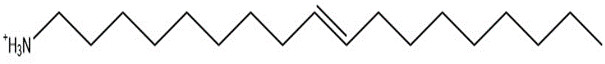 |
| 11105-8, C8 amine (constituent B) | 129.24 | 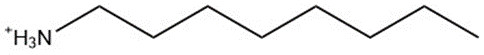 |
a The representative structure weights do not include the weight of the aliphatic amine counterions
2.1 Selection of analogues and use of (Q)SAR models
A read-across approach using data from analogues and the results of (quantitative) structure-activity relationship ((Q)SAR) models, where appropriate, has been used to inform the ecological assessment. The analogues selected were structurally similar and/or functionally similar to substances within this group (e.g., similar physical-chemical properties, same mechanism of toxic action) and had relevant empirical data that could be used to read across to substances in the TPAEs Group, which have limited empirical data. The names and structures of the analogue substances discussed in this report are confidential as they were notified under the New Substances Notification (NSN) Regulations. However, they have structures quite similar to some of the representative structures of TPAE-1 in that they both have ionized alkyl phosphorodithioate species in association with branched aliphatic amine salts. It is noted, however, that analogue selection for UVCBs carries greater uncertainty than for discrete chemicals due to the diversity of structural components and variability in compositions. The use of analogue ecotoxicity data is further discussed in Section 6.1.
The applicability of (Q)SAR models was determined on a case-by-case basis. Details of the read-across data and (Q)SAR models chosen to inform the ecological assessment of the TPAEs Group are further discussed in the relevant sections of this report.
| Analogue | Water solubility | Kow | Biodegradation data | Ecotoxicity data | Human health data |
|---|---|---|---|---|---|
| NSN substance 1 | Y | Y | N | N | NR |
| NSN substance 2 | Y | Y | Y | Y | NR |
Abbreviations: Y = yes; N = no; NR = not required; NSN = New Substances Notification
3. Physical and chemical properties
Measured and modelled physical and chemical property data for the two substances in the TPAEs Group are presented in Table 3-1, while data for the analogue NSN substances are found in Appendix A. Given the complexity of the TPAEs, as well as the NSN analogue substances, as they are ionizing UVCBs, there are inherent challenges in measuring the physical/chemical properties of these substances, meaning that these data may be associated with significant uncertainty.
| - | TPAE-1 | TPAE-2, Constituent A | TPAE-2, Constituent B |
|---|---|---|---|
| Physical state (20°C) | liquid | liquid | liquid |
| Melting point (°C) | -6 (pour point) | <-20 | <-20 |
| Boiling point (°C at 101.3 kPa) | 198 (initial) | 215 (initial) | 201 (initial) |
| Vapour pressure (Pa) | 3.2 x 10-2 | 2.2 x 10-2 | 1.8 |
| Relative density (unitless) | 1.02 @ 15.6°C | 0.938 @ 20°C | 0.969 @ 20°C |
| Water solubility (mg/L at loading rate in mg/L) | 39.5 @ 100 LR | 20.7 @ 10 000 LR | 4.9 @ 10 000 LR |
| Water solubility (mg/L at loading rate in mg/L) | 138 @ 1000 LR | 15.4 @ 1000 LR | 4.7 @ 100 000 LR |
| log Kow (dimensionless) | Range: <0.30 to >7.1a | C16-18 aliphatic amines compt.: 4.61 (±5.2) | C8 aliphatic amines compt.: 1.07 (±0.24) |
| log Kow (dimensionless) | NA | Dibutylphos-phorothioate compt: 2.73 (±1.8) | Dibutylphos-phorothioate compt.: 1.37 (±0.36 |
| log Koc (dimensionless) | <1.25 (to ≤5.09b | C16-18 aliphatic amines compt: 3.42c | NA |
| log Koc (dimensionless) | NA | Dibutylphosphorothioate compt: 2.51c | NA |
Notes: The references for constituents A and B of TPAE-2 are ECHA (2018a) and ECHA (2017), respectively. The reference for data for TPAE-1 is ECHA (2019a).
Abbreviations: Compt., component; NA, not available; Kow, octanol-water partition coefficient; Koc, organic carbon-water partition coefficient; LR, loading rate.
a 38% of area had Kow of >7.1, 24% of area had Kow of 1.8, and 9.8% of area had Kow of 4.9.
b 68% of area had Koc <1.25; 32% of area had Koc of 1.36 to ≤5.09.
c Calculated based on QSARs (ECHA 2017, 2018a).
Analytical characterization of TPAE-2 reported in ecotoxicity studies suggests the amine-thiophosphate salts dissociate in aqueous systems (Section 6.1.2), and it is assumed that TPAE-1 also dissociates in water. The physical-chemical properties of representative thiophosphate and amine components of the TPAEs were considered in addition to those of the whole-substance UVCBs included in Table 3-1. All of the representative structures of TPAE-1 and TPAE-2 are ionizing, with the exception of one neutral component of TPAE-1. The alkyl dithio/thio/phosphate ester components of these structures are all predicted to be strong acids, with predicted log pKa values in the range of 0.8 to 2.7 (ACD/Percepta c1997-2012), and so they will be almost completely ionized (negatively charged) at the environmentally relevant pH range of 5 to 9. All of the primary aliphatic amines have a pKa of about 10.5 (ECCC, HC 2021) and, therefore, are nearly 100% ionized (positively charged) over the environmentally relevant pH range of 5 to 9.
The alkyl (di)thiophosphate structures are predicted to have surfactant properties on the basis that the representative structures/components all have predicted surface tensions of about 28-29 dyn/cm (TEST 2016), which is well below the 60 dyn/cm value below which substances are considered to have surfactant properties (EC 1992). The aliphatic amines also have surfactant properties (ECCC, HC 2021).
(Q)SAR models were also used to generate predicted physical/chemical property values for the alkyl (di)thiophosphate and aliphatic amine components of these substances (Table 3-3). It should be noted that these models (i.e., EPI Suite c2000-2012, AIEPS c2010-2012, CATALOGIC 2014), with the exception of ACD/Percepta (c1997-2012), do not model the ionized forms of the substances, which is the form for most of these representative structures, at ambient pH range for natural waters (pH 5 to 9). Toxicity Estimation Software Tool (TEST) (2016) generates predictions based on the most similar substances available in its training set, which may or may not be ionizing.
Given the uncertainties inherent in modelling physical/chemical properties of ionizing substances and ones with surfactant properties, it was decided to use a representative alkyl dithiophosphate structure (structure 1) of TPAE-1, which is the only structure that is not ionizing, to represent the physical/chemical properties of the alkyl (di)thiophosphate and alkyl phosphate ester components of this substance. As this structure has a predicted log Kow of 5.4 to 5.9, this is considered to be a conservative choice for modelling endpoints such as bioaccumulation. A C12 tert-alkyl amine is used to represent the counterions of TPAE-1, which will be referred to as the aliphatic amine component of TPAE-1.
Physical-chemical properties for the other structures of TPAE-1 were modelled; however, it is unclear how valid some of these model results are for these ionizing substances, and therefore these values were not included in Table 3-2. The molecular weights and sizes of these structures vary widely (Table 2-2), and therefore their predicted physical-chemical properties also vary considerably. The major structures range in log D at pH 7 from -3.8 (Str. 10) to 6.9 (Str. 3) and in water solubility at pH 7 from 1.5 mg/L (Str. 3) to 1 x 106 mg/L (Str. 10) (ACD/Percepta c1997-2012). However, they are all predicted to have low vapour pressures (<0.13 Pa) (ACD/Percepta c1997-2012).
| - | Structure 1, TPAE-1 | Alkyl thiophosphate component, TPAE-2a | References |
|---|---|---|---|
| log Kow (dimensionless) | 5.9 | NA | KOWWIN (2010) |
| log D @ pH 7 (dimensionless) | 5.4 | -1.1 | ACD/Percepta (c1997-2012 ) |
| Water solubility (mg/L) | 0.055 – 1.1b | 3.7 x 105 | ACD/Percepta (c1997-2012 ) |
| Vapour pressure (Pa, 20 °C) | 5.2 x 10-5 | 6.6 x 10-3 | EPI Suite (c2000-2012) |
| Henry’s law constant (Pa m3/mol) | 7.0 x 10-3 | 2.0 x 10-1 | HENRYWIN (2008) Bond method |
| log Koc (dimensionless) | 3.1-3.7c | 2.6 | KOCWIN (2010), MCI method |
Abbreviations: log D, log octanol-water distribution ratio; Kow, octanol-water partition coefficient; Koc, organic carbon-water partition coefficient; MCI, molecular connectivity index.
a Modelling is based on the neutral form of this component, with the exception of log D and water solubility.
b Water solubility values estimated using the following models: WSKOWWIN (2010), WATERNT (2010) and ACD/Percepta (c1997-2012).
c 3.7 estimate is based on Kow.
Determining physical-chemical properties for aliphatic amines is complicated by their surfactant and ionizing properties, as discussed in more detail in the assessment report for this substance group (ECCC, HC 2021). ACD/Percepta (c1997-2012) was used to model the water solubility of the aliphatic amines, in order to supplement the limited experimental data (ECCC, HC 2021). Since the bioconcentration and partitioning behaviour of ionizing and surfactant substances does not correlate well with Kow (ECCC, HC 2021), other metrics were used to describe the partitioning behaviour of aliphatic amines, including the solids-water distribution coefficient (Kd), an organic carbon-water distribution coefficient specific to cations (Koc-cation), a distribution coefficient for ionic interactions with clays (KCEC), and the membrane-water partition coefficient (Kmw) (ECCC, HC 2021). The general trend in these partitioning coefficients is that the partitioning of aliphatic amines to soil, clay or membranes increases with increasing chain length. Modelled and measured physical-chemical properties for aliphatic amine representative structures for the TPAEs Group are summarized in Table 3-3.
| Structure | pKaa | Water solubility (mg/L) at pH 7 (pH 6-9)a | Vapour pressure at 20 °C (Pa)b |
|---|---|---|---|
| TPAE-1 aliphatic amine (C12) | 10.5 | 18400 (941 - 22 300) | 6.1 |
| TPAE-2, Constituent B, C8 aliphatic amine | 10.5 | 197 000 (10 500 – 235 000) | 129(E), 131 |
| TPAE-2, Constituent A, C18 amine | 10.5 | 4.6 (0.21 -5.6) | 0.012 |
Abbreviations: pKa, acid dissociation constant; E, experimental value (from EPI Suite c2000-2012 database).
a Modelled with ACD/Percepta c1997-2012.
b Modelled with EPI Suite c2000-2012.
4. Sources, uses and releases
The two substances in the TPAEs Group were included in a survey issued pursuant to a CEPA section 71 notice (Canada 2012). Both substances were reported to have been imported into Canada and neither substance was manufactured in Canada in 2011 at a reporting threshold of 100 kg (Table 4-1).
| Substance | Total importsa (kg) | Total manufacturea(kg) |
|---|---|---|
| TPAE-1 | 100 000 to 1 000 000 | NR |
| TPAE-2 | 500 | NR |
Abbreviations: NR, not reported at reporting threshold of 100 kg.
a Values reflect quantities reported in response to a CEPA section 71 survey (Environment Canada 2013). See survey for specific inclusions and exclusions (schedules 2 and 3).
Both substances in this group are used in Canada in industrial and automotive lubricants and lubricant additives (ECCC 2015). One of the reported uses of TPAE-1 is as a lubricant in the manufacture of machinery (ECCC 2015). For this assessment, this has been interpreted as use in metalworking fluids, to provide anti-oxidation, anti-corrosion, or anti-extreme temperature/pressure protection. Metalworking fluids may be used in various industries including aviation, ship building, automotive, electronic equipment manufacturing, transportation, and other industries. TPAEs might also be used as an additive in re-refined lubricant products, which are produced from waste oils that are re-processed at specialized facilities.
The only identified use of TPAE-2 in Canada is as a component of hydraulic oils, with a recommended content of less than 1% in the formulated hydraulic oil (personal communication, email from industry stakeholder to Environment and Climate Change Canada, dated September 12, 2018; unreferenced). However, TPAE-2 may also be used as an ashless antiwear additive for use in gear oils (automotive and industrial), hydraulic oils, compressor oils and engine oils for light duty trucks (personal communication, email from industry stakeholder to Environment and Climate Change Canada, dated September 12, 2018; unreferenced). In Europe, it is used in the manufacturing of vehicles and machinery, as a processing aid, and as a laboratory reagent (ECHA 2017, 2018a).
The uses of TPAEs that have potential releases to the environment are product formulation and, for TPAE-1, use in metalworking fluids. Most facilities using TPAEs would release their effluents to off-site wastewater treatment systems (WWTS), where the TPAEs may partition to the sludge. Therefore, there is also the potential for TPAEs contained in biosolids produced by WWTSFootnote 3 to be spread on to agricultural land. These potential release scenarios are discussed in Section 6.2.
Spent oil-based metalworking fluids are collected and disposed of when they are no longer suitable for service in accordance with provincial/territorial requirements. Small amounts of the spent metalworking fluids, whether they are oil-based or water-based, may enter into wastewater streams and be discharged to WWTS. Disposal of TPAEs in other lubricant oils are not expected to result in significant releases to the environment, as there are provincial/territorial regulations that require recycling of used lubricating oils (Environment Canada, Health Canada 1994, Environment Canada 2011, ChemInfo Services 2016).
5. Environmental fate and behaviour
5.1 Environmental distribution
The distribution of components of TPAEs, including long-chain aliphatic amines,Footnote 4 among the environmental compartments (air, water, soil, sediments, and biota) is qualitatively described below, based on measured/modelled physical and chemical properties. Current fugacity models are not designed to address the environmental fate of ionizing and surface-active substances such as TPAEs. Therefore, fugacity modelling was not performed.
The components of the TPAEs, most of which are ionized under typical conditions, are predicted to have low vapour pressures and will not be volatile (US EPA 2010). Volatilization of a chemical from water and moist soils is described by its Henry’s law constant (HLC), which can be measured experimentally, but is typically estimated as the ratio of vapour pressure to water solubility. Although QSAR models are available for generating HLCs, measured values for ionizing substances may differ significantly from the estimates (ECETOC 2003).Footnote 5 No experimental HLC data, or data calculated from high-quality experimental water solubility and vapour pressure data, was found for TPAEs or long-chain aliphatic amines. In general, negligible volatilization can be expected for surfactants (Boethling and Mackay 2000).
When released to water, the aliphatic amine components (due to their positive charge and surfactant properties) will be adsorbed onto the suspended solids, organic matter, and sediments. Organic cations that are charged at environmentally relevant pHs, such as aliphatic amines, sorb via ionic and non-ionic interactions with negatively charged cation exchange sites in both organic matter and clays (Droge and Goss 2013a, 2013b). The uncharged structure 1 of TPAE-1 is also predicted to sorb strongly to organic matter due to its high predicted log KOC value. Some of the larger alkyl (di)thiophosphate components may also partition to sediment despite their negative charge, as they have moderate to high modelled log D values (Table 3-3). The smaller thiophosphate components, however, are expected to remain predominantly in water given their high predicted water solubilities (greater than 1000 to greater than
10 000 mg/L) and low log D values (Table 3-3).
Likewise, when released to soil (e.g., via the application of sludge/biosolids on agricultural lands), structure 1 and some of the larger structures of TPAE-1 are expected to adsorb to soil particles. The long-chain aliphatic amine components will also sorb strongly to soil particles. However, the smaller, more water soluble and negatively charged alkyl thiophosphate and alkyl phosphate components of the TPAEs are not expected to bind to soil particles. Therefore, some of the TPAEs components will bind to soil and others will not.
Long-chain aliphatic amine components of TPAEs are not expected to undergo
long-range transport in air or water due to their low to moderate volatility, their binding to soils and sediments, and their relatively rapid biodegradability in the environment. In addition, TPAEs and aliphatic amines are predicted to have very short half-lives in the atmosphere as they react with hydroxyl radicals (Section 5.2.1), thus it appears unlikely that they would undergo long-range atmospheric transport. It may be possible for some of the alkyl thiophosphate components of TPAE-1 to undergo long-range transport in water, as some are highly soluble, and TPAE-1 appears to be persistent in water (Section 5.2.2). TPAE-2 is not persistent in water (Section 5.2.2), so is unlikely to undergo long-range transport in water.
5.2 Environmental persistence
5.2.1 Abiotic degradation
For TPAE-1, a hydrolysis study was not performed for the REACH registration, as it was not considered to be technically feasible (ECHA 2018a). No reliable data was obtained for the hydrolysis of constituent A of TPAE-2 due to its surface effects and poor instrument response (ECHA 2018a). No hydrolysis study was performed for constituent B of TPAE-2. However, the alkyl thiophosphate component of TPAE-2 was stable in solution for 48 to 72 hours during ecotoxicity testing (Section 6.1.2). Based on this information, the alkyl (di)thiophosphate components of the TPAEs do not seem to hydrolyse quickly. The aliphatic amine components will also not hydrolyse due to the lack of hydrolysable groups (ECCC, HC 2021).
TPAEs are not expected to be released in air given their intended uses and physical-chemical properties. The modelled atmospheric half-life of structure 1 of TPAE-1 due to reaction with hydroxyl ions is 0.6 hours (AOPWIN 2010), indicating that it is not persistent in air. The neutral forms of the other alkyl (di)thiophosphate components of TPAE-1 and TPAE-2 also have half-lives of less than 2 hours in air due to reactions with hydroxyl ions (AOPWIN 2010), and so are also not considered to be persistent in air. However, structure 1 is predicted to highly sorb (68% to 84%) to airborne particulates (AEROWIN 2010), and thus may be resistant to atmospheric degradation. The larger structures of TPAE-1 that have higher Kow values may also highly sorb to airborne particulates.
Aliphatic amines are also not predicted to be persistent in air, as they are likely to be degraded due to reactions with the hydroxyl radical and with ozone (AOPWIN 2010). The half-lives of aliphatic amines reacting with the hydroxyl radical vary from 0.4 to 3.2 hours, while for the reactions with ozone, the estimated half-lives are in the range of 0.7 to 2.1 hours (AOPWIN 2010). Therefore, components of TPAEs are not likely to be persistent in air.
5.2.2 Biodegradation in water
TPAE-1
In whole substance aerobic biodegradation tests, TPAE-1 was found to have very low ready biodegradability in water, with values ranging from -10.3% to 9.8% CO2 evolution over 28 days, even though the microbes were pre-adapted to the test substance in two of the tests (Table 5-1). NSN analogue 2, which is similar to TPAE-1, also showed low biodegradation of 20% over 28 days (Table 5-1).
As the TPAEs are expected to dissociate in aquatic environments, biodegradation of the components of TPAE-1 was examined separately in addition to the whole substance data. Modelled data for structure 1 support the empirical biodegradation data for the whole substance. CATALOGIC (2014) predicts a 28-day biological oxygen demand (BOD) test result (% degraded) of 4.0% (± 3.0%) for structure 1, with an ultimate half-life of over 1 year, 3 months. The BIOWIN 5 (MITI Linear model) and BIOWIN 6 (MITI non-linear model) (BIOWIN 2008) results indicate that structure 1 has low biodegradability, though the BIOWIN 3 (ultimate biodegradation model) result indicates a biodegradation timeframe of weeks to months. The C10-14 tert-alkyl amine component is not readily biodegradable with biodegradation measured by O2 consumption or CO2 evolution of 22% or less following 28-day tests (Table 5-1); however, some of these tests used concentrations of test substance of 10 mg/L or more which was hypothesized to have been toxic to the microbes. It is possible that C12-14 tert-alkyl amines could biodegrade over a longer time-frame. The predicted ready
(28-day) biodegradation for C12-14 tert-alkyl amines is 18% to 22%, and their ultimate biodegradation half-lives are predicted to be approximately 3 months (CATALOGIC 2014). In summary, the empirical and modelled data suggest that the alkyl (di)thiophosphate components of TPAE-1 will be persistent in water, while the aliphatic amine components may biodegrade on the order of months.
TPAE-2
In aerobic CO2 evolution tests, constituents A and B were found to have biodegradation of 75% and 80% over 28 days, respectively (Table 5-1). The higher biodegradation observed for this substance may be attributable to its components being considerably smaller than those of TPAE-1, several of which have molecular weights in the 700 to 1000 g/mol range, versus 494 g/mol for the largest structure of TPAE-2 (i.e., constituent A with C18 aliphatic amine). TPAE-1 also has much more branching in both the alkyl (di)thiophosphate and amine components than TPAE-2, a feature which may hinder biodegradation.
Modelling for the alkyl thiophosphate component of TPAE-2 generally supports the experimental data for the whole substance. The CATALOGIC (2014) 28-day BOD prediction is only 10% (±28%); however, this prediction is not considered robust as 54% of substance fragments were not in the model training set. The BIOWIN 3 estimate indicates an ultimate biodegradation timeframe of days to weeks (BIOWIN 2008).
The aliphatic amine components of TPAE-2 were also found to be highly biodegraded. Both octadecylamine (C18 primary amine; CAS RN 124-30-1) and octylamine (C8 primary amine; CAS RN 111-86-4) were reported as being readily biodegradable in OECD guideline tests (Table 5-1). For C8-C18 cocoamine (CAS RN 61788-46-3), there were two ready biodegradation studies available with values ranging from 56% to 67% (Table 5-1), which are close to or above the ready biodegradation pass value of 60%. Model results also support that these components will have ultimate half-lives in the order of weeks in water (BIOWIN 2008; CATALOGIC 2014).
In summary, existing data suggest that both the alkyl thiophosphate and aliphatic amine components of TPAE-2 will degrade rapidly in the environment.
| Substance | Test conditions (method) | Test result (28 day) | Reference |
|---|---|---|---|
| TPAE-1, whole substance | Aerobic ready biodegradability in water, CO2 evolution test (ASTM D-5864-95) | 3.6% +/- 4.5% a | ECHA 2019a |
| TPAE-1, whole substance | Aerobic ready biodegradability in water, CO2 evolution test (OECD 301B) | -10% +/- 0.3% a | ECHA2019a |
| TPAE-1, whole substance | Aerobic ready biodegradability in water, CO2 evolution test (OECD 301B) | 7.4% +/- 2.4% | ECHA 2019a |
| TPAE-1, whole substance | Aerobic ready biodegradability in water, CO2 evolution test (OECD 301B, EU Method C.4-C, and ISO 10634:1995(E)) |
-4.7% b -2.7% c |
ECHA 2019a |
| TPAE-2 constituent A | Aerobic ready biodegradability in water, CO2 evolution test (OECD 301B) | 75% | ECHA 2018a |
| TPAE-2, constituent B | Aerobic ready biodegradability in water, CO2 evolution test (OECD 301B) | 80% | ECHA 2017 |
| NSN analogue 2 | OECD 301B | 20% (CO2 evolution) | CEPA New Substances Program |
| EC 701-175-2, C10-14 tert-alkyl amines, constituentd | Ready biodegradability: Closed bottle test, O2 consumption (OECD 301D) | 22% | ECHA 2019b |
| EC 701-175-2, C10-14 tert-alkyl amines, constituente | Ready biodegradability: O2 consumption (OECD 301D, OECD 301F), CO2 evol. (ASTM D 5864-95) | -18.1% to 4.3% | ECHA 2019b |
| Octadecylamine (C18) (CAS RN 124-30-1) | OECD 301F | 70% | ECHA 2018c |
| Coco amine (C8-C18) (CAS RN 61788-46-3) | OECD 301B, OECD 301D | 56% (O2 consumption), 58%, 67.3% (CO2 evolution) | Akzo Nobel 1992a, ECHA 2018c |
| Octylamine (C8) (CAS RN 111-86-4) | Various OECD methods | 73% to 99% | ECHA 2019c |
a In these studies, the test sludge was pre-adapted to the test material for 14 days before commencement of the 28-day biodegradability tests. Pre-adaptation was not performed in the other studies with TPAE-1.
b Test material was applied to test medium by pre-application on silica gel.
c Test material was added to test medium by pre-dissolving in silicone oil.
d No analysis of test material provided.
e Analysis of test material, PRIMENE 81-R, provided for two of the tests: 74% C12, 14% C11, 11% C10.
5.2.3 Biodegradation in soil
No data on biodegradation in soil was available for TPAEs or NSN analogues. Data were available on the biodegradation of the aliphatic amine hexadecanamine (C16), as well as for two analogue substances for aliphatic amines, quaternary ammonium compounds, C12-C18 alkyl (hydroxyethyl) dimethyl, chlorides, and C22 trimethyl ammonium chloride (ECCC, HC 2021). It was determined, because of the relatively high percentage of the formation of radioactive 14CO2 in tests with these substances (55% to 73%), as well as 50% dissipation times in soils of 8 to 41 days, that biodegradation is a predominant process of the dissipation of these substances, and that soil microorganisms are capable of degrading long-chain aliphatic amines (ECCC, HC 2021).
Based on the above data, the aliphatic amine components of the TPAEs are not expected to be persistent in soil. However, the highly branched alkyl (di)thiophosphate components in TPAE-1 are expected to be persistent in soil.
5.2.4 Biodegradation in sediments
No studies were found on the biodegradation of TPAEs or of long-chain aliphatic amines in sediments.
Under aerobic conditions in the top few centimeters of sediments, half-lives of the alkyl (di)thiophosphate and aliphatic amine components in sediments could be expected to be similar to those in soils. Thus, the aliphatic amine components are not expected to be persistent, whereas the half-lives of the alkyl (di)thiophosphate components would depend on the level of branching, where the smaller less branched components of TPAE-2 would be expected to degrade quickly while the larger more highly branched components of TPAE-1 may persist.
The representative structures of TPAE-1, including the aliphatic amine structure, are expected to undergo very slow anaerobic biodegradation (BIOWIN 2008). No tests on biodegradation of the TPAEs or primary aliphatic amines in sediments under anaerobic conditions are available (ECCC, HC 2021). Most sediments are anoxic at a depth greater than a few centimeters. It is conservatively assumed that these substances are persistent under anaerobic conditions such as those found in anoxic sediment layers. This assumption is consistent with the European risk assessment report on primary aliphatic amines (EC 2008). For the aliphatic amines components, it is also consistent with observations of accumulated and elevated concentrations of cationic surfactants in field sediments (Lara-Martin et al. 2010).
5.3 Potential for bioaccumulation
5.3.1 Bioaccumulation potential of alkyl (di)thiophosphate components
A bioconcentration factor (BCF) study with rainbow trout (Oncorhynchus mykiss) was conducted with structure 1 of TPAE-1, which is a neutral (unionized) structure, as a surrogate for the whole substance TPAE-1 (Study Submission 2018; REACH registration dossier, unreferenced). The study was not conducted on the UVCB itself as it has a complex composition which is not suited for conventional bioconcentration tests. In the BCF assay, the radiolabelled surrogate molecule of 99% purity was tested at two concentrations in water, 0.25 and 2.8 μg/L (mean measured values), using a solvent carrier (dimethylformamide at 0.1 mL/L). There was a 35-day uptake period and a 62-day depuration period, during which the fish appeared normal and healthy. Steady state concentrations in the fish tissues were achieved after 31 days. The results from this study showed that this substance had a steady state BCF of 436 and a kinetic BCF of 834, in whole fish. These results show that this substance does not bioconcentrate significantly in fish. It was noted that the measured concentrations of test substance were substantially lower (39% to 67%) than the target concentrations, indicating that sorption of the test substance may have been an issue.
Bioaccumulation modelling was performed for TPAE-1 representative structure 1 using BCFBAF (2010). This substance has a predicted log Kow of 5.9 (Table 3-2). For a mid-trophic level fish, the BCF and BAF were estimated as 1015 and 2023 L/kg wet wt., respectively, indicating that this structure is predicted to have moderate bioaccumulation (between 1000 and 5000 L/kg).
There is some uncertainty related to whether the BCF and BAF values for structure 1 may apply to some of the other ionized structures of TPAE-1. Some of the larger structures of this substance, despite being ionized, are predicted to have low water solubilities and high log D values, suggesting they may have bioaccumulative properties.
No bioaccumulation testing was performed with TPAE-2, or the NSN analogues 1 and 2. However, the alkyl thiophosphate component of TPAE-2 has a measured Kow less than 5 (Table 3-1). As well, this substance is ionizing, so is not expected to highly bioaccumulate.
5.3.2 Bioaccumulation potential of long-chain aliphatic amines
A single experimental BCF study by Akzo Nobel (2006) with hexadecylamine in carp (Cyprinus carpio) was found in which the whole-fish BCF value obtained in this long-term (11 month) study (2400 L/kg) is considered a conservative whole-body estimate (ECCC, HC 2021). There is also a study on the uptake and tissue distribution of surfactants in rainbow trout, including C9 to C16 aliphatic amines (McLachlan 2018). While the study duration was too short to determine steady-state BCFs, 7-day BCFs for individual substances ranged from approximately 3 to 1000 L/kg ww, which are consistent with BCFs predicted by others using the BIONIC model (McLachlan 2018). Analogue data on monoalkyl trimethyl ammonium compounds (TMAC) were also considered. A review on the bioaccumulation of surfactants by Krop and de Voogt (2007) shows that TMAC (Monoalkyl trimethyl ammonium compounds), with alkyl chains of C12 or less, have BCF values of approximately 100 L/kg or less in fresh water, where these values were determined indirectly (Versteeg and Shorter 1991; Schlechtriem et al. 2015). Schlechtriem et al. (2015) estimated the BCF for a C16-18 TMAC as 1960 L/kg using uptake and depuration rates from Versteeg and Shorter (1991). Considering all of the available information, it was determined that long-chain aliphatic amines do not have high bioaccumulation potential in aquatic organisms (ECCC, HC 2021).
Table 5‑2 summarizes the key data regarding the bioconcentration potential of the TPAEs components in aquatic organisms.
| Substance | Test organism | Experimental concentration, duration or model information | BCF or BAF (L/kg) |
|---|---|---|---|
| Structure 1 of TPAE-1 | Rainbow trout (Oncorhynchus mykiss) | 0.25 and 2.8 µg/L (35 days uptake, 62 day depuration) | BCF=834 |
| Structure 1 of TPAE-1 | NA | Middle trophic level fish, BCFBAF (2012) model, including biotransformation | BCF=1015, BAF=2023 |
| Hexadecylamine (C16) | Carp (Cyprinus carpio) | 3 µg/L, 11 months | BCF=2400 |
Abbreviations: NA, not applicable
The measured, modelled and analogue data all indicate that the alkyl (di)thiophosphate components of TPAEs have low potential for bioaccumulation. The experimental and modelled data for long-chain aliphatic amines and their analogues show moderate potential for bioaccumulation.
6. Potential to cause ecological harm
6.1 Ecological effects assessment
6.1.1 Mode/mechanism of action
The alkyl (di)thiophosphate components of the TPAEs bear structural similarity to certain thiophosphate, dithiophosphate, and organophosphate (OP) pesticides (e.g., ethion, disulfoton, dimethoate). These pesticides act as neurotoxins by deactivating the enzyme acetylcholinesterase (AChE) that hydrolyzes the neurotransmitter acetylcholine (Sparling 2016). While very toxic to insects, most OPs have moderate to low toxicity to birds and mammals, with fish tending to be more sensitive than mammals (Sparling 2016).
However, the alkyl (di)thiophosphate and OP components of the TPAEs have significant structural differences relative to these pesticide compounds, which introduces uncertainty into whether they will also exhibit an AChE inhibition mode of action (MoA). These differences include molecular weight, length of the alkyl chains as well as their degree of branching, and ionization (i.e., the majority of the TPAEs structures are ionized whereas the pesticides are not), all of which could affect the potential for the TPAEs to interact with and inhibit AChE. Furthermore, the toxicity tests for the TPAEs indicate a lower aquatic hazard profile relative to the most comparable (di)thiophosphate pesticides (see Section 6.1.2), suggesting there may be differences in MoA, or at least in potencies. The profiles for ethion and dimethoate in the OECD QSAR Toolbox (2017) indicate that they have a mechanism of action related to covalent binding, including protein binding related to skin sensitization, and DNA binding (SN2 nucleophilic substitution), and they were classified as having a specific MoA according to the Verhaar classification scheme. In contrast, the alkyl (di)thiophosphate components of TPAE-1 do not have alerts for protein binding, though they do for DNA binding (SN2), and are also classified as having a specific MoA by Verhaar.
There is also uncertainty about the MoA of long-chain aliphatic amine components of the TPAEs. While there is evidence to suggest that these substances have a non-specific narcotic MoA, there is uncertainty as to whether this MoA could be expected for all organisms, or whether some may be more susceptible to these substances due to specific ionic interactions (ECCC, HC 2021).
In conclusion, there is uncertainty about the MoAs of both the alkyl (di)thiophosphate and aliphatic amine components of the TPAEs. For the purpose of this assessment, both of these components were considered to have MoAs with higher toxicity than nonpolar narcosis.
6.1.2 Effects on aquatic organisms
Aquatic toxicity studies for TPAEs include acute fish studies for TPAE-1, and acute Daphnia magna and algae studies for both TPAEs. Also available for TPAE-1 is a chronic D. magna study. Robust study summaries are available for all of these studies through ECHA (ECHA 2017, 2018a, 2019a), and they are briefly summarized in Table 6-1. All studies mentioned above were conducted with water-accommodated fractions (WAFs) of test substance, and all of the results were reported as nominal loading rates. For TPAE-1, the studies did not include chemical analyses, except for the acute D. magna study, which analyzed the total organic carbon (TOC) levels of the test solutions. Tests with constituents A and B of TPAE-2 included chemical analyses of test substance components, as discussed below.
| Substance | Test organism | Endpoint | Value (mg/L) | Reference |
|---|---|---|---|---|
| TPAE-1 | Rainbow trout (Oncorhynchus mykiss) | 96 h LL50 | 24 (nominal) | Study Submission 2018 |
| TPAE-1 | Fathead minnow (Pimephales promelas) | 96 h LL50 | 8.5 (nominal) | Study Submission 2018 |
| TPAE-2 (CAS RN 97808-07-6), constituent A | Rainbow trout (Oncorhynchus mykiss) | 96 h LL50 | >0.028 (nominal) | ECHA 2018a |
| TPAE-1 | Daphnia magna | 48 h EL50 (immobilization) | 91.4 (nominal) | Study Submission 2018 |
| TPAE-1 | Daphnia magna | 21 d EL50 (immobilization) | 0.91 (nominal) | Study Submission 2018 |
| TPAE-1 | Daphnia magna | 21 d EL50 (reproduction) | 0.66 (nominal) | Study Submission 2018 |
| TPAE-2, constituent A | Daphnia magna | 48 h EL50 (immobilization) | 0.071 (nominal) | ECHA 2018a |
| TPAE-2, constituent B | Daphnia magna | 48 h EL50 (immobilization) | 31 (nominal) | ECHA 2017 |
| TPAE-1 | Algae (Selenastrum capricornutum)a | 72 h EL50 (growth rate) | 16 (nominal) | Study Submission 2018 |
| TPAE-1 | Algae (Selenastrum capricornutum)a | 72 h EL50 (yield) | 6.3 (nominal) | Study Submission 2018 |
| TPAE-2, constituent A | Algae (Pseudokirchneriella subcapitata)a | 72 h EL50 (growth rate) | 0.028 (nominal) | ECHA 2018a |
| TPAE-2, constituent A | Algae (Pseudokirchneriella subcapitata)a | 72 h EL50 (yield) | 0.026 (nominal) | ECHA 2018a |
| TPAE-2, constituent A | Algae (Pseudokirchneriella subcapitata)a | 72 h EL10 (yield) | 0.022 (nominal) | ECHA 2018a |
| TPAE-2, constituent B | Algae (Pseudokirchneriella subcapitata)a | 72 h EL50 (growth rate) | 3 (nominal) | ECHA 2017 |
| TPAE-2, constituent B | Algae (Pseudokirchneriella subcapitata)a | 72 h EL50 (yield) | 1.8 (nominal) | ECHA 2017 |
Abbreviations: ELX, loading rate of test substance resulting in a specified effect (e.g., immobilization, growth) in X% of the test species exposed to the WAF; LL50, median lethal loading rate, loading rate of the test substance that results in 50% mortality in a population of test organisms exposed to the WAF; WAF, water-accommodated fraction, aqueous medium containing only the fraction of the substance that is dissolved or present as a stable dispersion or emulsion.
a P. subcapitata and S. capricornutum are the same species.
For the acute D. magna study with TPAE-1, the test solutions were analyzed for TOC in the fresh test solutions only. These analyses indicated that the 30.5 mg/L (nominal) test solution contained only 3.8 times as much TOC as the 1 mg/L test solution (0.53 mg/L TOC), and that the 99.5 mg/L test solution contained only 9.6 times as much TOC as the 1 mg/L test solution (ECHA 2019b), indicating low levels of dissolution of test substance in these WAF solutions relative to the 1 mg/L (nominal) test solution. In the fathead minnow study with TPAE-1, it was noted that most of the solutions in these test vessels were cloudy with surface films (Study Submission 2018), also indicating a low level of test substance dissolution.
For TPAE-2, aquatic toxicity studies included chemical analyses performed by high performance liquid chromatography with mass spectrometry (HPLC-MS) (ECHA 2017, 2018a). The tests involving constituent A measured the concentration of the unsaturated C18 amine, and the tests involving constituent B measured the concentration of dibutylthiophosphoric acid (DBPA). These substances were measured in the test solution at the beginning and end of the test periods (ECHA 2017, 2018a). The C18 amine component was not stable during the assays; its concentration declined during the acute semi-static fish test in fresh test preparations at 0 h and 72 h from 0.013 and 0.017 mg/L, respectively, to 0.0062 mg/L and 0.0085 mg/L at 24 h and 96 h, respectively, representing declines of about 48% and 50%, over the course of a 24-h period. During the static D. magna test, the C18 amine declined in concentration after 48 hours by about 94%, from 0.083 mg/L (nominal: 0.10 mg/L) to 0.005 mg/L. From a measured concentration of 0.042 mg/L at 0 h, the concentration of the C18 amine dropped below the limit of quantification of the analytical method (0.0019 mg/L). This is not unexpected, as it is known that aliphatic amines can sorb to containers as well as to biota (ECCC, HC 2021). Unlike the amine, the concentrations of DBPA appeared to be stable over the test periods of 48 to 72 h in the static D. magna and algae studies where this component was measured (ECHA 2017, 2018a).
Based on the analyses in the tests with TPAE-2, it is hypothesized that the aliphatic amine components in the aquatic toxicity studies with TPAE-1 also declined greatly in concentration during the tests, while the concentrations of the alkyl (di)thiophosphate components were stable over time.
A comparison of the acute toxicity (EL50) of these substances shows that constituent A of TPAE-2 is more toxic to D. magna by 3 orders of magnitude (0.071 mg/L) than constituent B (31 mg/L) and TPAE-1 (91 mg/L). As with the D. magna studies, constituent A was shown to have a toxicity to algae of about 2 orders of magnitude greater than constituent B and TPAE-1. The differences in the toxicity of these substances may be attributable to the differences in the toxicities of the aliphatic amine components with differing chain lengths (C16/18 vs. C8; with increasing toxicity with increasing chain length), but may also be attributable to experimental variation.
6.1.2.1 Analogue data
Ecotoxicity data were submitted for NSN analogue 2 under the New Substances Notification Regulations, including acute data for fish and daphnia, and chronic data for daphnia and algae. This alkyl dithiophosphate ester is structurally similar to some of the representative structures of TPAE-1, despite differences in physical/chemical property values (Section 3). Similar issues were reported for tests with this substance as for the tests with the TPAEs, as described above. A WAF protocol was used due to the low aqueous solubility and complex nature of the test material, so results were reported as nominal loading rates. The WAF solutions were filtered as the test media were observed to be cloudy white dispersions, with globules of test material at the bottom of the mixing vessels, at the water surface and adhered to the sides of the mixing vessels at the end of the 24-h mixing period. No chemical analyses were done for the toxicity studies, so only nominal endpoint values were reported.
NSN analogue 2 was shown to have similar nominal whole-substance toxicity to TPAE-1 for all of the organisms studied. In the tests involving O. mykiss, NSN analogue 2 had a 96-h LL50 value of 56 mg/L. In a chronic study with D. magna, the 21-d LL50 values were 2.5 mg/L and 3.2 mg/L for reproduction and immobilization, respectively. The acute daphnia 48-h EL50 value was 15 mg/L. The 21-d D. magna reproduction EL50 was the most sensitive endpoint for both substances. With green algae, the 72-h EC50 value (biomass) was 5.1 mg/L.
6.1.2.2 Pesticide data
Toxicity data for the pesticides ethion and sulfotep, which have alkyl thiophosphate ester structures similar to some of the representative structures of TPAE-1, were compared with empirical whole-substance toxicity data for TPAEs. These two pesticides are not registered for use in Canada (Health Canada 2019). Arthropods, such as insects, or for aquatic organisms, crustaceans, such as daphnids, are expected to be the most sensitive class of organisms for these pesticides. Daphnid data for ethion and sulfotep were available from the ECOTOX (2000-2019) database. For each of these substances, there were two acute studies with D. magna. Ethion 48 h EC50 values were 4.6 x 10-5 and 5.6 x 10-5 mg/L, while sulfotep had 48 h EC50 or LC50 values of 2.3 x 10-3 and 2.5 x 10-3 mg/L. The acute D. magna values for the TPAEs were 7.1 x 10-2 mg/L for TPAE-2 and 91.4 mg/L for TPAE-1. Given the differences in chemical structures of the TPAEs and these two pesticides, the hazard data for these pesticides are considered in the weight of evidence for hazard potential rather than used as direct read-across for the TPAEs.
6.1.2.3 Modelled data
Modelling the toxicity of the alkyl (di)thiophosphate/organophosphate components of the TPAEs was undertaken. However, the model results were ultimately not included in this report due to uncertainties with the accuracy of the predictions. Toxicity modelling of ionizing substances is generally problematic, as the available models are mainly KOW-based (e.g., ECOSAR c2000-2016, AIEPS c2010-2012, CATALOGIC 2014), and do not take into account the ionizing nature of the substances. In addition, the model training sets were not suitable for the alkyl (di)thiophosphate components of the TPAEs. TEST (2016) and AIEPS (c2010-2012) did not have substances similar to the alkyl (di)thiophosphate components in the model training sets, and the training sets of ECOSAR (c2000-2016) for the applicable substance classes, “esters, dithiophosphate” and “esters, thiophosphate,” were based largely upon pesticide data, which as discussed earlier, have uncertainty as to whether they are appropriate surrogate substances for the TPAEs as they are considerably more toxic. CATALOGIC (2014) was not able to produce toxicity estimates for the alkyl (di)thiophosphate components, predicting only that these substances have a mode of action more reactive than baseline narcosis, so only “less than” values were produced.
There is a large data set of empirical data for long-chain aliphatic amines (ECCC, HC 2021), so modeling the toxicity of the aliphatic amine components was not necessary, though these substances would also be difficult to model due to their ionizing and surfactant properties. Therefore, models were not used to estimate the toxicity of the components of the TPAEs.
6.1.2.4 Selection and standardization of critical toxicity values (CTVs) and derivation of predicted no-effect concentrations (PNECs)
Two CTVs were selected for TPAEs: one for the alkyl (di)thiophosphate components and one for the aliphatic amines components.
6.1.2.4.1 CTV and PNEC derivation for the thiophosphate components
Because of the lack of appropriate analogue or modelled data for the alkyl (di)thiophosphate components, the CTV was based on the empirical whole-substance toxicity data for TPAE-1 and TPAE-2. These data were considered as a group, since TPAE-1 has components that contain either an alkyl dithiophosphate ester or an alkyl thiophosphate ester (Table 2-2), and the structures of these substances are similar. The most sensitive endpoint for these two substances is the 72 h algae EL10 for yield with TPAE-2 of 0.022 mg/L (Table 6-1). The quality of this study was evaluated and it was found to be acceptable.
To derive the PNEC for the alkyl (di)thiophosphate components, the CTV was divided by an assessment factor (AF). AFs account for various extrapolations and sources of uncertainty. An endpoint standardization factor (FES) is considered for extrapolation from a short-term (acute) to a long-term (chronic) time-frame, from lethal effects (i.e., mortality) to sublethal effects (e.g., growth, reproduction), and from median effect levels (e.g., EC50) to low effect levels (e.g., EC10). The AF also accounts for the number of species and organism categories that are represented in the toxicity dataset (species variation factor; FSV), and whether the substance has a mode of action that is more toxic than baseline narcosis (mode of action factor, FMOA). The final AF is derived by multiplying the FES, FSV and the FMOA. Since the CTV is a chronic study with a low-effects sublethal endpoint, the FES is equal to one. Since the mode of action for the alkyl (di)thiophosphate components of the TPAEs is suspected to be more toxic than non-polar narcosis (Section 6.1.1), a FMOA of 2 was applied to the CTV. This is a relatively low FMOA, as the dataset includes what is considered to be the most sensitive class of organisms for the AChE inhibition MoA, arthropods, such as insects, or for aquatic organisms, crustaceans, such as daphnids. The combined aquatic toxicity data sets for TPAE-1 and TPAE-2 include a total of four species, covering the three species categories (plants, invertebrates and vertebrates), as previously described, which is a moderately rich data set, therefore an FSV of 2 was used. The overall AF of 4 (FES × FSV × FMoA = 1 × 2 × 2) was applied to the CTV of 22 µg/L, giving a PNEC of 5.5 µg/L.
6.1.2.4.2 CTV and PNEC derivation for the aliphatic amine components
The CTV for the aliphatic amine components of the TPAEs was taken from the assessment of aliphatic amines (ECCC, HC 2021). One aquatic CTV and PNEC was used for all of the long chain aliphatic amines, based on the similar toxicity observed for these substances (ECCC, HC 2021). Aquatic toxicity data for long chain aliphatic amines were found for 11 species covering the three organism categories of fish, invertebrates, and algae. The CTV is from an algae study with Desmodesmus subspicatus with hexadecyldimethylamine (CAS 112-69-6), a tertiary amine with a C16 alkyl chain, which has an EC10 for biomass of 0.43 µg/L (measured) (ECHA 2019d). To convert this CTV to a PNEC value, the CTV was divided by the following factors: a FES of 1 as this endpoint is for a low effect value from a chronic study, a FSV of 1 due to the relatively large and complete toxicity dataset, and a mode of action factor (FMOA) of 2, to account for uncertainty in the mode of action of aliphatic amines (ECCC, HC 2021), thus giving a PNEC of 0.22 µg/L.
6.1.3 Effects on sediment organisms
No sediment toxicity data (empirical or modelled) were available for the two TPAEs, or for the NSN analogue substances. Therefore, it was not possible to derive a sediment PNEC for the alkyl (di)thiophosphate components of the TPAEs. However, sediment data were available for long-chain aliphatic amines.
Sediment toxicity data for long-chain aliphatic amines and long-chain quaternary ammonium compounds (analogue substances) were available, including five chronic studies covering three species: three studies with the nematode Caenorhabditis elegans, one study with the midge Chironomus riparius, and one study with the worm Lumbriculus variegatus. These studies are further described in ECCC, HC (2021). The toxicities of long-chain aliphatic amines and analogue substances were found to be similar. The most sensitive endpoint from an acceptable study was a nominal EC10 value of 11 mg/kg dw for effects on reproduction and biomass with L. variegatus and the long-chain aliphatic amine N-(Hydrogenated tallow)-1,3-diaminopropane (CAS RN 68603-64-5). The CTV represents a long-term, low-effect, and sub-lethal toxicity value; therefore, no assessment factor is needed to account for acute-to-chronic extrapolation. However, since the dataset represents only three species from one category of organisms (invertebrates), an assessment factor of 20 is used to account for species variation. In addition, an assessment factor of 2 is used to account for the uncertainty in the mode of action of these substances. Thus, the CTV was divided by an overall assessment factor of 40 to derive a PNEC of 0.275 mg/kg dw.
6.1.4 Effects on soil-dwelling organisms
No soil toxicity data (empirical or modelled) were available for the two TPAEs, or for the NSN analogue substances. Therefore, it was not possible to derive a soil PNEC for the alkyl (di)thiophosphate components of the TPAEs. However, soil toxicity data were available for long-chain aliphatic amines.
Soil toxicity data for aliphatic amines were available for six species from two categories of organisms (primary producers and invertebrates), as described further in ECCC, HC (2021). The most sensitive value was a 21-day EC25 value with C12-14 DMAFootnote 6 of 52 mg/kg for rapeseed (shoot length) (ECHA 2015), which was selected as the CTV for PNEC derivation. Since the CTV represents an acute (short duration) exposure for plants, an assessment factor of 5 was applied to extrapolate to a long-term (chronic) value. Since a moderate amount of toxicity data are available, a species-variation factor of 5 was used, in addition to a FMoA of 2. An AF of 50 was applied to this value (FES × FSV × FMoA = 5 × 5 × 2), giving a PNEC of 1 mg/kg dry wt. This PNEC will be used for the aliphatic amines components of the TPAEs.
6.2 Ecological exposure assessment
Quantitative exposure scenarios were prepared only for TPAE-1, due to the much lower usage quantity of TPAE-2 (Table 4-1). Also, TPAE-2 is not used in metalworking fluids. The exposure/risk for TPAE-2 is qualitatively described in Section 6.3.1.
Exposure scenarios were prepared for: 1) formulation of lubricants, 2) use of metalworking fluids, and 3) application of biosolids from WWTSs receiving wastewater from industrial users of metalworking fluids.
An exposure scenario was not prepared for the use of TPAEs in other lubricants and greases, as it was determined that there would be little to no environmental exposure from this use, as these products get recycled or disposed of at waste facilities and are therefore not discharged to the environment.
6.2.1 Measured concentrations in environmental media and wastewater
No data were found on environmental concentrations of TPAEs or aliphatic amines, in Canada or elsewhere.
6.2.2 Calculation of predicted exposure concentrations (PECs) and general assumptions
Exposure modelling of TPAE-1 was performed based on two representative structures of this substance, structure 1 (neutral organic), representing the alkyl (di)thiophosphate components of this substance, and the aliphatic amine component (ionizable) (Table 2-2). The rationale for the choice of structure 1 is discussed in Section 3. The aliphatic amine component was chosen as it is the counterion for every other major structure in this substance (Section 2). Both of these structures were considered individually for the WWTS modelling. Assumptions were made about the weight percentages of these representative structures of TPAE-1, which were based on the chemical analysis of a single sample of this substance, as described in Section 2. Based on this analysis, it was assumed that the alkyl (di)thiophosphate ester components, as represented by structure 1, account for roughly 75% by weight of this substance, with the aliphatic amine component comprising the remaining 25% by weight.
For both industrial scenarios described below, the model SimpleTreat 3.1 (2003) was used to model the on-site secondary WWTS and gives a removal rate of 76% for structure 1. For the aliphatic amine component, the WWTS removal rate of 96% was based on empirical data for aliphatic amines (ECCC, HC 2021) and is assumed to represent the combined primary and secondary removal rate. In addition, pre-treatment from an on-site oil/water separator was also assumed, with a removal value of 50% for TPAEs given their solubility and density (OECD 2011).
The aquatic PECs for the aliphatic amines were further adjusted to calculate the freely dissolved fraction that will be bioavailable, in recognition that once released to surface waters a fraction of the aliphatic amines will sorb to organic matter and not be bioavailable. As described in the Aliphatic Amines Group assessment (ECCC, HC 2021), assuming a mean TOC concentration of 13.05 mg/L in Canadian surface waters, the freely dissolved fraction for the C12-C14 aliphatic amine component of TPAE-1 would be 93.7%.
The exposure scenarios are briefly described below. More detail is available in ECCC (2020).
6.2.3 Exposure scenario 1: lubricant formulation
Releases to the environment may occur during the blending of TPAEs with other components to make lubricant products at industrial facilities. Companies that formulate lubricant products are situated in various locations across Canada. Information on lubricant formulation facilities in Canada, such as their blending capacities, maximum potential use quantities, wastewater flows, discharge methods (direct or indirect), on-site or off-site treatment systems and dilution factors for the receiving bodies of water, were used to estimate their releases.
The scenario is based on the potential quantity handled at a Canadian facility. It is assumed that the substance is used at a facility continually throughout the year. The mass of TPAE-1 handled was provided by the importing companies (personal communication, emails from industry stakeholders to Environment and Climate Change Canada, February 2018; unreferenced). An emission factor of 0.25% was used for this scenario based on OECD (2004), which considers substance releases to wastewater from raw materials handling, blending, packaging, floor cleaning, and other processes. The removal rate for an on-site oil/water separator (50%) was taken from OECD (2011). The removal rate for a secondary WWTS (located on site or off site), as discussed above, was also used. A facility can discharge effluent directly to surface water or to a WWTS and then to surface water.
Applying facility specific information and the above information, the highest aquatic PECs for this scenario are 1.9 µg/L for structure 1 and 0.106 µg/L for the aliphatic amine component. Adjusting to a freely dissolved concentration, the aliphatic amine component PEC is 0.099 µg/L.
6.2.4 Exposure scenario 2: use in metalworking fluids
Use of TPAEs in metalworking fluids may occur in multiple facilities located across Canada, ranging in operation size and location. Specific information on the users of metalworking fluids containing TPAEs including use quantities are unknown. To develop a PEC in surface water, the following parameter values were used: the volume of metalworking fluids handled at a facility, the concentration of TPAE-1 in various metalworking fluids, the emission factor for this substance released to wastewater, the substance removal rates by a WWTS, the effluent flow, and the dilution factor from a receiving water body.
This scenario considers a representative situation where an industrial facility uses oil-based metalworking fluids and handles TPAE-1 continuously throughout the year. The volume of metalworking fluids handled at the facility was taken from the U.S. geometric mean value (OECD 2011). The concentration of TPAEs in oil-based metalworking fluids varies from 5% to 20% (Brinksmeier et al. 2015); a concentration of 10% was used in this scenario. An emission factor of 11% was used, which considers releases of the substance to wastewater from raw materials handling, residual oil cleaning on metal surfaces, finishing, and other processes (OECD 2011). Average removal rates were used for the on-site oil/water separator and for applicable secondary treatment systems as most facilities discharge wastewater to a WWTS with secondary treatment. In addition, the effluent from the WWTS is diluted by the receiving water body. The daily dilution volumeFootnote 7 for the receiving water body was taken as the 10th percentile from a distribution for industrial facilities potentially handling the substance in metalworking fluids, which was based on industrial data taken from Canadian government databases and on measured or calculated data on receiving water body flows.
The above scenario resulted in aquatic PECs of 10.1 µg/L for structure 1 and 0.56 µg/L for the aliphatic amine component. Adjusting to a freely dissolved concentration, the aliphatic amine component PEC is 0.52 µg/L.
6.2.5 Exposure scenario 3: Biosolids application
The soil PEC was calculated as an extension of the aquatic scenario for metalworking fluids described above, as these facilities discharge their wastewater to an off-site WWTS. Some lubricant blending facilities also discharge wastewater to off-site WWTSs; however, the discharges are much less than from metalworking facilities, so this biosolids application scenario is based on the aquatic PEC from the metalworking fluid scenario. Biosolids produced from WWTSs may be applied to agricultural fields as a soil amendment.
The amount of the aliphatic amine component of TPAE-1 accumulated within the top 20 cm of soil over 10 consecutive years of once-yearly biosolids application was estimated using an approach described by ECHA (2016). This was based on the quantity released to a WWTS from an industrial user. A soil PEC for structure 1 was not developed due to the lack of empirical, analogue or modelled soil toxicity data needed to derive a PNEC. A biodegradation half-life in soil of 96 days for the aliphatic amine component was used based on biodegradation modelling performed with CATALOGIC (2014), and the degradation was assumed to have first order kinetics. Concentrations were determined on a yearly basis immediately after application and at the end of the year (365 days) prior to the subsequent application, over a 10-year period. The calculations assumed no losses of TPAEs via volatilization, leaching or run-off (ECCC 2020).
Due to lack of information, it was assumed that all of the aliphatic amine component removed within the WWTS would remain in sludge. The 10th percentile flow rate from Canadian WWTSs with secondary treatment that receive wastewater from metalworking facilities was used in the PEC calculation.
Following this approach, the soil PEC value for the aliphatic amine component is 1.20 mg/kg dry wt.
6.2.6 Sediment
As no sediment exposure data were available for the TPAEs, and only very limited data were available for aliphatic amines (ECCC, HC 2021), PECs for sediment were not derived for the components of TPAE-1. It is not currently possible to model the distribution of concentrations in sediment for the aliphatic amines and alkyl (di)thiophosphate components (as represented by structure 1), due to the unique properties of surfactants and lack of field measurements. While sorption to sediment particles is expected and there is potential for exposure to benthic organisms, there is uncertainty about the level of exposure to sediment organisms and a need for environmental sampling to quantitatively determine this. However, given the substance properties (i.e., expected persistence of components of TPAEs in sediment and expected partitioning of the neutral and large alkyl (di)thiophosphate components and of the aliphatic amine components to sediment), as well as calculated concentrations in aquatic systems, it is expected that sediment organisms will likely be exposed to TPAEs.
6.3 Characterization of ecological risk
The approach taken in this ecological screening assessment was to examine assessment information and develop proposed conclusions using a weight-of-evidence approach and precaution. Evidence was gathered to determine the potential for TPAEs or their components to cause harm in the Canadian environment. Lines of evidence considered include those evaluated in this assessment that support the characterization of ecological risk in the Canadian environment. Secondary or indirect lines of evidence are considered when available, including regulatory decisions and classification of hazard or fate characteristics made by other regulatory agencies.
6.3.1 Risk quotient analysis
Risk quotient analyses for TPAE-1 were performed by comparing the various estimates of exposure (Section 6.2) with ecotoxicity information (Section 6.1) to determine whether there is potential for ecological harm in Canada. Risk quotients (RQs) were calculated separately for two representative components of TPAE-1, the alkyl (di)thiophosphate components, represented by structure 1 and a C12 tert-alkyl amine structure, representing the aliphatic amine counterions. RQs were calculated by dividing the PEC by the PNEC for relevant environmental compartments and associated exposure scenarios, as shown in tables 6-2 and 6-3.
| Exposure scenario (compartment) | PEC | PNEC | RQ |
|---|---|---|---|
| Lubricant formulation (water) | 1.9 µg/L | 5.5 µg/L | 0.35 |
| Metalworking fluids (water) | 10.1 µg/L | 5.5 µg/L | 1.8 |
Abbreviation: PEC, predicted exposure concentration; PNEC, predicted no-effect concentration; RQ, risk quotient
a PEC modelling used physical and chemical properties of Structure 1, while PNEC is based on whole-substance toxicity data.
| Exposure scenario (compartment) | PECa | PNEC | RQ |
|---|---|---|---|
| Lubricant formulation (water) | 0.099 µg/L | 0.22 µg/L | 0.45 |
| Metalworking fluids (water) | 0.52 µg/L | 0.22 µg/L | 2.4 |
| Metalworking fluids (soil, from biosolids) | 1.2 mg/kg d.wt. | 1 mg/kg d.wt. | 1.2 |
Abbreviation: PEC, predicted exposure concentration; PNEC, predicted no-effect concentration; RQ, risk quotient
a The PEC values for the exposure scenarios for aliphatic amine components in water have been adjusted to freely dissolved concentrations at a median TOC concentration.
The aquatic RQs are less than 1 for both representative structures for the lubricant formulation scenario, and they are greater than 1 for both representative structures for the metalworking fluids scenario (tables 6-2 and 6-3). As well, the soil RQ for the aliphatic amine component is greater than 1 for the spreading of biosolids on land for the metalworking scenario.
The usage quantity of TPAE-2 in Canada is at least 200 times lower than that of TPAE-1 and TPAE-2 is not used in metalworking fluids (Section 4). It is therefore expected that TPAE-2 would have RQs lower than 1 for its use in lubricant formulation.
6.3.2 Consideration of the lines of evidence
To characterize the ecological risk of TPAEs, technical information for various lines of evidence was considered (as discussed in the relevant sections of this report) and qualitatively weighted. The key lines of evidence supporting the assessment conclusion are presented in Table 6-4 with an overall discussion of the weight of evidence provided in Section 6.3.3. The level of confidence refers to the combined influence of data quality and variability, data gaps, causality, plausibility and any extrapolation required within the line of evidence. The relevance refers to the impact the line of evidence has when determining the potential to cause harm in the Canadian environment. Qualifiers used in the analysis ranged from low to high, with the assigned weight having five possible outcomes.
| Line of evidence | Level of confidencea | Relevance in assessmentb | Weight assignedc |
|---|---|---|---|
| Similarity in chemical structure for read-across purposes | moderate | high | moderate to high |
| Environmental fate and behaviour (ionic nature) | moderate | moderate | moderate |
| Persistence in the environment, alkyl (di)thiophosphate and aliphatic amines components | high | high | high |
| Long-range transport | moderate | low | low to moderate |
| Bioaccumulation in aquatic organisms, alkyl (di)thiophosphate and aliphatic amines components | moderate | moderate | moderate |
| Mode of action, alkyl (di)thiophosphate and aliphatic amines components | moderate | moderate | moderate |
| PNEC for aquatic organisms, alkyl (di)thiophosphate components | moderate | high | moderate to high |
| PNEC for aquatic organisms, aliphatic amines component | high | high | high |
| PNEC for soil organisms, aliphatic amines component | moderate | high | moderate to high |
| Aquatic PECs | moderate | high | moderate to high |
| Soil PEC | moderate | high | moderate to high |
| RQs for water | moderate | high | moderate to high |
| RQ for soil | moderate | high | moderate to high |
Abbreviation: PEC, predicted exposure concentration; PNEC, predicted no-effect concentration; RQ, risk quotient
a Level of confidence is determined according to data quality, data variability, data gaps (i.e., are the data fit for purpose).
b Relevance refers to the impact of the evidence in the assessment.
c Weight is assigned to each line of evidence according to the overall combined weights for level of confidence and relevance in the assessment.
6.3.3 Weight of evidence for determining potential to cause harm to the Canadian environment
The PNEC for the alkyl (di)thiophosphate and organophosphate components of TPAE-1 is based on whole-substance toxicity data for both TPAEs, while PNECs for the aliphatic amine component of TPAE-1 are based on empirical data for long-chain aliphatic amines. The whole-substance toxicity data endpoints are expressed as nominal loading rates. It is not known what component(s) of the TPAEs are responsible for the toxicity (e.g., aliphatic amine vs. alkyl (di)thiophosphate esters) giving only moderate confidence in this PNEC. The aquatic PNEC for the aliphatic amines component of TPAE-1 is based on a large empirical aquatic toxicity data set for aliphatic amines and is therefore considered to be reliable. The soil toxicity data set for aliphatic amines is not as large, thus giving only moderate confidence in the soil PNEC. Both the alkyl (di)thiophosphate and aliphatic amine components have high chronic toxicity to aquatic organisms, while the aliphatic amines have moderate to low chronic toxicity to soil organisms. There are empirical data on the environmental fate and behaviour of aliphatic amines in the environment (e.g., their strong sorption to environmental matrices, empirical soil-water partition coefficient); however, no empirical data were found on the environmental fate and behaviour of the alkyl (di)thiophosphate components of the TPAEs (other than whole-substance biodegradation data). The empirical and modelled biodegradation data indicate that TPAE-1 will persist in the environment long enough to cause chronic toxicity. In addition, TPAE-1 is released continuously from industrial facilities.
Components of TPAEs are not considered to be persistent in air and are not released to air; therefore, they do not undergo long-range atmospheric transport to remote regions. TPAE-1 was found to be persistent in water due to its very slow biodegradation rate, thus increasing the duration and spatial extent of exposure to organisms. TPAE-2 was found to biodegrade much faster than TPAE-1, probably due to structural considerations as discussed in Section 5.2, and therefore is not persistent. The TPAEs were not found to be bioaccumulative based on empirical and modelled data for structure 1 of TPAE-1 and for aliphatic amines.
The reliability of aquatic PECs considers a number of factors, including the WWTS removal rate, the usage quantity, the emission factor, and the daily dilution water volume of the receiving environment. The WWTS removal rate for structure 1, as modelled with SimpleTreat (2003), is considered to be moderately reliable. The performance of SimpleTreat (2003) to model Kd (the solids-water partition coefficient) has been verified with measured data for PAHs (ECCC study, unreferenced) and is considered to be very reliable. However, this also depends on the reliability of the physical/chemical property data of the modelled substance, which are only considered to be moderate for structure 1, as they are based entirely on modelled, rather than measured data. The WWTS removal rate for the aliphatic amines component is considered to be reliable, as this is based on reliable empirical data. The industrial usage volume data for the TPAEs for the lubricant formulation scenario are considered to be reliable, as they were obtained directly from industry stakeholders, while the usage quantity data for the metalworking scenario are based on an OECD (2011) emission scenario document, representing generic practices. The daily dilution water volume data are considered to be reliable, as they are based on industrial data taken from Canadian government databases and on measured or calculated data on receiving water body flows. The emission factors are estimates from generic sources (i.e., OECD publications), rather than site-specific data. The above factors lead to the confidence in the aquatic PECs being moderate.
The soil PEC was determined using a generic approach that uses the highest allowable provincial biosolids application rate in Canada. The approach incorporates biodegradation in soil, but no additional losses due to volatilization, leaching or runoff, over a 10-year period. The soil PEC is also based on some of the same parameters as the aquatic PECs such as physical/chemical properties, industrial usage volume, and the industrial emissions factor. Therefore, the confidence in the soil PEC is also moderate.
The risk quotients indicate that TPAE-1 has the potential to cause ecological harm to aquatic and soil organisms in Canada, resulting from its use in metalworking fluids. The formulation of lubricants containing TPAEs was not found to pose a risk to the environment. Use of TPAEs in lubricants is not likely causing ecological harm in Canada, as the majority of these products are recycled in dedicated facilities.
Some components of TPAEs released to water are expected to partition to sediments (e.g., aliphatic amines) and may accumulate over time, as they are expected to be persistent in sediment. The industrial uses of TPAEs considered in the exposure scenarios are assumed to result in continuous releases to the environment. Empirical sediment toxicity data for aliphatic amines and their analogues indicate relatively low chronic toxicity; however, information on the toxicity of the TPAEs (whole substance), the alkyl (di)thiophosphate components or analogues through sediment exposure, as well as information on the concentration of components of TPAEs in sediments was not available.
TPAE-1 has been given the following ecological hazard classification by the European Chemicals Agency (ECHA): “toxic to aquatic life with long lasting effects” (ECHA 2018b). Constituent A of TPAE-2 has been given the following hazard classification by ECHA: “This substance is very toxic to aquatic life, is toxic to aquatic life with long lasting effects” (ECHA 2018e). Constituent B of TPAE-2 does not have hazard labelling based on ecotoxicity, but does have hazard labelling for human health–related effects (ECHA 2019e).
While current exposures of the Canadian environment to TPAE-2 are unlikely to be of concern, TPAE-2 is considered to have a high level of hazard for aquatic organisms. Therefore, there may be a concern for the Canadian environment should exposures increase.
6.3.4 Sensitivity of conclusion to key uncertainties
Substances with ionizing and surfactant properties pose a challenge for risk assessment because their physical-chemical properties and toxicities are difficult to measure in empirical studies and also to model, which adds uncertainty to the assessment conclusions. This challenge was addressed by modelling a non-ionizing representative structure of TPAE-1 and considering only valid empirical studies for TPAEs, aliphatic amines and analogues. An additional source of uncertainty in the RQs is that the PNEC for the alkyl (di)thiophosphate components of TPAE-1 is based on whole-substance toxicity data, whereas the PEC was modelled based on the physical-chemical properties of structure 1 only. Therefore, the PEC for the alkyl (di)thiophosphate components is not representative of the full range of physical-chemical properties present in this substance. The choice of structure 1 for PEC modelling is considered to be a conservative choice due to its relatively high log Kow value of 5.4 to 5.9.
Although there is limited ecotoxicity data for the two TPAEs, a large data set of empirical toxicity data for the long-chain aliphatic amines (including analogue substances) shows that components of TPAEs have high toxicity to aquatic and soil organisms.
The exposure scenarios for the TPAEs were developed on the basis of information obtained through CEPA section 71 surveys and follow-up with industrial stakeholders. In the absence of data, realistic assumptions were made in order to estimate PECs. For the exposure scenarios, empirical data on the physical and chemical properties of the components of the TPAEs, especially the alkyl (di)thiophosphate ester components, as well as more specific data for industrial emission factors would have increased the certainty in the PECs. However, this information would not likely have changed the risk conclusion for these exposure scenarios given the low PNECs.
Some components of TPAEs released to water are expected to partition to sediments and may accumulate over time, with some representative structures expected to be persistent in sediment. A determination of risk to sediment organisms could not be made due to the lack of information on environmental concentrations of TPAEs components in sediment near points of effluent discharge, as well as a lack of information on the toxicity of TPAEs components other than aliphatic amines to sediment organisms. If this information had been available, it may have added confidence to the lines of evidence which were used to arrive at the proposed conclusion, but would not have resulted in a change to the overall conclusion.
7. Potential to cause harm to human health
7.1 Exposure assessment
TPAE-2 was included in the Rapid Screening of Substances with Limited General Population Exposure document (ECCC, HC 2018). In this approach, the potential for direct exposure was determined if a substance was present or used in a product used by the general population. Subsequently, substances considered to have low potential for direct exposure were assessed for their potential for indirect exposure. As a result of this analysis, the potential for exposure of the general population to TPAE-2 was considered to be negligible.
According to information submitted in response to a CEPA section 71 survey, there was no reported manufacturing of TPAE-1 in Canada for the 2011 calendar year. The reported use for this substance was manufacturing machinery; therefore, exposure of the general population to the substance from this use is not expected (Environment Canada 2013).
No measured data regarding concentrations of TPAE-1 in environmental media were identified in Canada, or elsewhere.
Estimates of exposure to TPAE-1 through drinking water were based on the sum of the aquatic PECs for both components (described in Table 6‑2 and Table 6‑3) associated with environmental releases of this substance from metalworking facilities (see Section 6.2.4 for detailed discussion). The highest total daily intake (via drinking water consumption) of TPAE-1 by the general population was 1.39 µg/kg bw/day (for infants less than 6 months of age). Total daily intake for other age categories and parameters for estimating exposure are described in Appendix B. Estimated exposures to TPAE-1 via drinking water consumption may be highly conservative as the PEC was derived to represent the concentration in the water close to the point of discharge, and potential additional dilution and removal during drinking water treatment was not taken into consideration when estimating exposure.
7.2 Health effects assessment
Data on the health effects of TPAE-1 were available from the submission under REACH for EC Number 931-384-6 (ECHA 2007-2018a).
A short-term repeat-dose study was available for TPAE-1 (Unnamed study report 2002a). In the study, four groups of 10 female and 10 male CD Sprague-Dawley rats were administered the substance via gavage daily for 28 days at doses of 0, 50, 150 or 500 mg/kg bw/day. Increases in absolute and relative adrenal and liver weights and absolute heart weights were observed in the high-dose females. Histopathological changes were observed in the mid- and high-dose groups, including effects on the adrenal glands (increased cortical vacuolation and hypertrophy) and kidneys (increased incidence of hyaline droplets). Neurobehavioural testing revealed decreased landing foot splay in females at the mid- and high-dose levels. No treatment-related changes were observed during the microscopic examination of the central nervous system. Based on the effects observed in the mid-dose group, a NOAEL of 50 mg/kg bw/day has been identified for this study.
Reproductive toxicity data from an OECD 421 screening test is available for TPAE-1. In the study (Unnamed study report 2010), Wistar rats in groups including 10 males and 10 females each, were administered 0, 15, 150 or 750 mg/kg bw/day via gavage for up to 56 days. Based on clinical observations observed in the high-dose animals, the dose level was reduced to 500 mg/kg bw/day from Day 5 onwards; females treated at 750 mg/kg bw/day had occasional tremors prior to reduction in dose level to 500 mg/kg bw/day. Animals of both sexes in the high-dose group showed statistically significant reduction in cumulative body weight gain in the final week of gestation. No effects on body weight were observed at the mid or low dose in either females or males. An increase in the incidence of hypertrophy/vacuolation of the interstitial glands of the ovaries was observed in the high-dose females only, although this increase was not found to be statistically significant. At the high-dose level there was a statistically significant decrease in the live birth index. There were no statistically significant increases in pre- and post-implantation losses observed, nor treatment-related effects on conception rates. The study authors indicated that the substance showed adverse reproductive effects only at maternally-toxic doses in this study. The study authors reported the NOAEL to be 150 mg/kg bw/day. The authors also indicated that a NOEL of 15 mg/kg bw/day was appropriate for reproductive and developmental toxicity based on a LOEL of 150 mg/kg bw/day for non-statistically significant reproductive and developmental effects (Unnamed study report 2010).
The substance was not found to be genotoxic based on available data for the substance from ECHA 2007-2018a. The results were negative for in vitro Ames assays in five strains of Salmonella typhimurium, with or without metabolic activation (Unnamed study report 1996). In addition, the substance was found to be non-mutagenic in the OECD Test No. 476, In Vitro Mammalian Cell Gene Mutation test (Unnamed study report 2009). The available data also indicates that the substance is not genotoxic (Chromosome aberration) in vivo, based on the results of a Mammalian Erythrocyte Micronucleus Test (Unnamed study report 2002b).
7.3 Characterization of risk to human health
The exposure potential for TPAE-2 was evaluated in the Rapid Screening of Substances with Limited General Population Exposure (ECCC, HC 2018). The exposure potential for this substance was considered to be negligible and as a result, this substance was found to be of low concern to human health.
The critical health effect endpoint for TPAE-1 was identified to be 50 mg/kg bw/day, based on observed effects in the adrenal glands and potential neurotoxic and/or behavioural effects from a short-term repeated dose study. Exposure to the general population is possible from ingestion of drinking water containing the substance as a result of industrial releases. Comparison of the critical effect level with the highest estimated potential exposure (1.39 x 10-3 mg/kg bw/day for infants less than 6 months of age) results in a margin of exposure of more than 35 000. Margins of exposure for other age groups (not shown) are greater.
On the basis of the conservative parameters used in modelling human exposure to TPAE-1 from potential environmental releases, the calculated margins are considered adequate to address uncertainties in the health effects and exposure databases.
7.4 Uncertainties in evaluation of risk to human health
The key sources of uncertainty are presented in the table below.
| Key source of uncertainty | Impact |
|---|---|
| There is a lack of occurrence data for TPAE-1 in the Canadian environment; therefore, there is uncertainty surrounding the occurrence and predicted environmental concentrations of this UVCB, as discussed in Section 6.3.4 | +/- |
| There is a lack of chronic toxicity data for TPAE-1 | +/- |
+/- = unknown potential to cause over or under estimation of risk.
8. Conclusion
Considering all available lines of evidence presented in this draft screening assessment, there is risk of harm to the environment from TPAE-1. It is proposed to conclude that TPAE-1 meets the criteria under paragraph 64(a) of CEPA as it is entering or may enter the environment in a quantity or concentration or under conditions that have or may have an immediate or long-term harmful effect on the environment or its biological diversity. However, it is proposed to conclude that TPAE-1 does not meet the criteria under paragraph 64(b) of CEPA as it is not entering the environment in a quantity or concentration or under conditions that constitute or may constitute a danger to the environment on which life depends.
Considering all available lines of evidence presented in this draft screening assessment, there is low risk of harm to the environment from TPAE-2. It is proposed to conclude that TPAE-2 does not meet the criteria under paragraphs 64(a) or (b) of CEPA as it is not entering the environment in a quantity or concentration or under conditions that have or may have an immediate or long-term harmful effect on the environment or its biological diversity or that constitute or may constitute a danger to the environment on which life depends.
Considering all the information presented in this draft screening assessment, it is proposed to conclude that TPAE-1 and TPAE-2 do not meet the criteria under paragraph 64(c) of CEPA as they are not entering the environment in a quantity or concentration or under conditions that constitute or may constitute a danger in Canada to human life or health.
It is therefore proposed to conclude that TPAE-1 meets one or more of the criteria set out in section 64 of CEPA and that TPAE-2 does not meet any of the criteria set out in section 64 of CEPA.
It is also proposed that TPAE-1 meets the persistence criteria but not the bioaccumulation criteria as set out in the Persistence and Bioaccumulation Regulations of CEPA.
References
ACD/Percepta [prediction module]. c1997-2012. Toronto (ON): Advanced Chemistry Development, Inc.
[AEROWIN] Atmospheric particulate sorption program for Microsoft Windows [estimation model]. 2010. Ver. 1.00. Washington (DC): US Environmental Protection Agency, Office of Pollution Prevention and Toxics; Syracuse (NY): Syracuse Research Corporation.
[AIEPS] Artificial Intelligence Expert Predictive System. c2010-2012. Ver. 3.0. Gatineau (QC): Environment Canada. Model developed by Stephen Niculescu.
Akzo Nobel. 1992. Biodegradability of Armeen CD. Report No.: CRL F91125. Arnhem (NL): Akzo Research Laboratories. [cited in US EPA 2003].
Akzo Nobel. 2006. Bioconcentration of 1-Hexadecylamine (C16 primary amine) in the carp, Cyprinus carpio, under flow-through conditions. Arnhem (NL): Akzo Research Laboratories. [cited in EC 2008].
[AOPWIN] Atmospheric Oxidation Program for Microsoft Windows [estimation model]. 2010. Ver. 1.92a. Washington (DC): US Environmental Protection Agency, Office of Pollution Prevention and Toxics; Syracuse (NY): Syracuse Research Corporation.
Armitage J, Arnot J, Wania F, Mackay D. 2013. Development and evaluation of a mechanistic bioconcentration model for ionogenic organic chemicals in fish. Environ Tox Chem. 32(1):115-128.
[ASTM] American Standard Test Method International. 2000. ASTM D5864-95 Standard Test Method for Determining Aerobic Aquatic Biodegradation of Lubricants or Their Components (account required). West Conshohocken, PA. [accessed 2020 Mar 12].
[BCFBAF] Bioaccumulation Program for Microsoft Windows [estimation model]. 2010. Ver. 3.01. Washington (DC): US Environmental Protection Agency, Office of Pollution Prevention and Toxics; Syracuse (NY): Syracuse Research Corporation.
[BIOWIN] Biodegradation Probability Program for Microsoft Windows [estimation model]. 2008. Ver. 4.10. Washington (DC): US Environmental Protection Agency, Office of Pollution Prevention and Toxics; Syracuse (NY): Syracuse Research Corporation. Blakemore G, Forbis A. 1991. Chronic toxicity of Di-Syston technical to Daphnia magna under flow-through test conditions. Data evaluation record. Kansas City (MO): Mobay Corporation. Report No.: 032501. Study ID No.: 33896. MRID No.: 419358-02.
Boethling R, Mackay D. 2000. Handbook of property estimation methods for chemicals. New York (NY): Lewis Publisher.
Canada. 1999. Canadian Environmental Protection Act, 1999. S.C. 1999, c. 33. Canada Gazette Part III, vol. 22, no. 3.
Canada. 2000. Canadian Environmental Protection Act, 1999: Persistence and Bioaccumulation Regulations. P.C. 2000-348, 23 March, 2000, SOR/2000-107.
Canada, Dept. of the Environment. 2012. Canadian Environmental Protection Act, 1999: Notice with respect to certain substances on the Domestic Substances List [PDF]. Canada Gazette, Part I, vol. 146, no. 48, Supplement.
Canada. 2018. Approach to disclose confidential information and promote transparency in chemicals management [PDF]. [updated 2018 Oct 23; accessed 2020 June 10].
CATALOGIC [environmental fate and ecotoxicity model]. 2014. Ver. 5.11.15. Bourgas (BG): University “Prof. Dr. Assen Zlatarov”, Laboratory of Mathematical Chemistry.
ChemInfo Services 2016. Technical Study on the Use, Exposure, and Release Potential of Used Lubricating Oils and Associated Substances. Prepared for Health Canada. Confidential Report.
Droge ST, Goss KU. 2013a. Development and evaluation of a new sorption model for organic cations in soil: Contributions from organic matter and clay minerals. Environ Sci Technol. 47(24):14233-14241.
Droge STJ, Goss KU. 2013b. Ion-exchange affinity of organic cations to natural organic matter: influence of amine type and nonionic interactions at two different pHs. Environ Sci Technol. 47:798-806.
[EC] European Communities. 1992. A.5. Surface Tension [PDF]. Report No.: 67/548/EEC Method, L383 A. Official Journal of the European Communities. p.47-53. [accessed 2020 Mar 12].
[EC] European Commission. 2008. European Union draft risk assessment report: Primary aliphatic amines [PDF]. Luxembourg: Office for Official Publications of the European Communities. [accessed 2020 Mar 12].
[ECCC] Environment and Climate Change Canada. 2015. DSL Inventory Update II data collected under the Canadian Environmental Protection Act, 1999, section 71: Notice with respect to certain substances on the Domestic Substances List. Data prepared by: ECCC, Health Canada, Existing Substances Program.
[ECCC] Environment and Climate Change Canada. 2020. Supporting Documentation for the Ecological Exposure Assessment of TPAEs. Gatineau (QC). Available from: substances@ec.gc.ca.
[ECCC, HC] Environment and Climate Change Canada, Health Canada. 2015. Identification of risk assessment priorities: results of the 2015 review. Ottawa (ON): Government of Canada. [accessed 2020 Mar 12].
[ECCC, HC] Environment and Climate Change Canada, Health Canada. [modified 2017 Mar 12]. Categorization. Ottawa (ON): Government of Canada. [accessed 2020 Mar 12].
[ECCC, HC] Environment and Climate Change Canada, Health Canada. 2018. Rapid screening of substances with limited general population exposure. Ottawa (ON): Government of Canada. [accessed 2020 Mar 12].
[ECCC, HC] Environment and Climate Change Canada, Health Canada. 2021. Screening assessment: Aliphatic Amines. Ottawa (ON): Government of Canada.
[ECETOC] European Centre for Ecotoxicology and Toxicology of Chemicals. 2003. Environmental risk assessment of difficult substances. Report No. 88. Brussels (BE): ECETOC. 109 p.
[ECHA] European Chemicals Agency.2015. Registration Dossier: Hexadecyldimethylamine CAS RN 112-69-6. Helsinki (FI): ECHA. [updated 2015 Apr 13; accessed 2018 Oct].
[ECHA] European Chemicals Agency. 2016. Guidance on Information Requirements and Chemical Safety Assessment. Chapter R. 16: Environmental Exposure Estimation. Version 3.0 [PDF]. [accessed 2020 Mar 12].
[ECHA] European Chemicals Agency. 2017. Registration Dossier: O,O-dibutyl hydrogen thiophosphate, compound with 1-octylamine (1:1) CAS RN 93964-99-9. Helsinki (FI): ECHA. [updated 2017 Aug 14; accessed 2018 Nov 19].
[ECHA] European Chemicals Agency. 2018a. Registration Dossier: Amines, C16-18-(even numbered, saturated and unsaturated) alkyl, O,O-di-Bu phosphorothioates. EC No. 947-129-7. Helsinki (FI): ECHA. [updated 2018 Mar 13; accessed 2018 Nov 19].
[ECHA] European Chemicals Agency. 2018b. Substance Information for Reaction products of bis(4-methylpentan-2-yl) dithiophosphoric acid with phosphorus oxide, propylene oxide and amines, C12-14-alkyl (branched). EC No. 931-384-6. Helsinki (FI): ECHA. [updated 2018 Aug 15; accessed 2019 Mar 13].
[ECHA] European Chemicals Agency. 2018c. Registration Dossier: Octadecylamine, CAS RN 124-30-1. Helsinki (FI): ECHA. [updated 2018 Jan 10; accessed 2018 Apr 6].
[ECHA] European Chemicals Agency. 2019a. Registration Dossier: Reaction products of bis(4-methylpentan-2-yl) dithiophosphoric acid with phosphorus oxide, propylene oxide and amines, C12-14-alkyl (branched). EC No. 931-384-6. Helsinki (FI): ECHA. [updated 2019 Mar 6; accessed 2019 Mar 13].
[ECHA] European Chemicals Agency. 2019b. Registration Dossier: Amines, C10-14 tert-alkyl. EC 701-175-2. Helsinki (FI): ECHA. [updated 2019 Feb 7; accessed 2019 Mar 27].
[ECHA] European Chemicals Agency. 2019c. Registration Dossier: Octylamine. CAS RN 111-86-4. Helsinki (FI): ECHA. [updated 2019 Feb 6; accessed 2019 Mar 27].
[ECHA] European Chemicals Agency. 2019d. Registration Dossier: Hexadecyldimethylamine, CAS RN 112-69-6. Helsinki (FI): ECHA. [updated 2019 Feb 9; accessed 2020 Feb 18].
[ECHA] European Chemicals Agency. 2019e. Registration Dossier: O,O-dibutyl hydrogen thiophosphate, compound with 1-octylamine (1:1) CAS RN 93964-99-9. Helsinki (FI): ECHA. [updated 2019 Jan 25; accessed 2019 Mar 13].
[ECOSAR] ECOlogical Structure-Activity Relationship Model Class Program [estimation model]. c2000-2016. Ver. 2.0. Washington (DC): US Environmental Protection Agency, Office of Chemical Safety and Pollution Prevention [accessed 2020 Mar 12].
[ECOTOX] ECOTOXicology database [database]. 2000-2019. Ver. 5.1. Washington (DC): US Environmental Protection Agency, Office of Research and Development; National Health and Environmental Effects Research Laboratory; Mid-Continent Ecology Division. [updated 2019 Mar 14; accessed 2019 Mar 14].
Environment Canada. 2011. Follow up on the Final Decision on the Assessment of Releases of Used Crankcase Oils to the Environment [PDF]. [accessed 2020 Mar 12].
Environment Canada. 2013. DSL Inventory Update data collected under the Canadian Environmental Protection Act, 1999, section 71: Notice with respect to certain substances on the Domestic Substances List. Data prepared by: Environment Canada, Health Canada; Existing Substances Program.
Environment Canada, Health Canada. 1994. Waste Crankcase Oils. Priority Substances List Assessment Report [PDF]. Ottawa (ON): Government of Canada. [accessed 2020 Mar 12].
[EPI Suite] Estimation Program Interface Suite for Microsoft Windows [estimation model]. c2000-2012. Ver. 4.11. Washington (DC): US Environmental Protection Agency, Office of Pollution Prevention and Toxics; Syracuse (NY): Syracuse Research Corporation. [accessed 2020 Mar 12].
Health Canada. 2015. Food Consumption Table derived from Statistics Canada, Canadian Community Health Survey, Cycle 2.2, Nutrition (2004), Share file. Ottawa (ON).
Health Canada. 2017. Water Consumption Table derived from Statistics Canada, Canadian Community Health Survey, Cycle 2.2, Nutrition (2004). Share file. Ottawa.
Health Canada. 2018. Draft backgrounder document on default values for breast milk and formula intakes. Unpublished report. Ottawa (ON): Government of Canada.
Health Canada. 2019. Pesticide Product Information Database, Active Ingredient Search. [accessed 2019 July 25].
[HENRYWIN] Henry’s Law Constant Program for Microsoft Windows [estimation model]. 2008. Ver. 3.20. Washington (DC): US Environmental Protection Agency, Office of Pollution Prevention and Toxics; Syracuse (NY): Syracuse Research Corporation.
Johnson WW, Finley MT. 1980. Handbook of aquatic toxicity of chemicals to fish and aquatic invertebrates. Washington (DC): U.S. Fish and Wildlife Service. Resource Publication No.: 137. 98 p.
[KOCWIN] Organic Carbon Partition Coefficient Program for Microsoft Windows [estimation model]. 2010. Ver. 2.00. Washington (DC): US Environmental Protection Agency, Office of Pollution Prevention and Toxics; Syracuse (NY): Syracuse Research Corporation.
[KOWWIN] Octanol-Water Partition Coefficient Program for Microsoft Windows [estimation model]. 2010. Ver. 1.68. Washington (DC): US Environmental Protection Agency, Office of Pollution Prevention and Toxics; Syracuse (NY): Syracuse Research Corporation.
Krop H, de Voogt P. 2007. Bioconcentration factors of surfactants in seawater. Amsterdam (NL): IVAM UvA BV.
Lara-Martin PA, Li X, Bopp RF, Brownawell BJ. 2010. Occurrence of alkyltrimethylammonium compounds in urban estuarine sediments: Behentrimonium as a new emerging contaminant. Environ Sci Technol. 44:7569-7575.
McLachlan M. 2018. CEFIC ECO37 D-BASS: Development of a bioaccumulation assessment strategy for surfactants. Deliverable D8: Report on tissue-distribution for 2 surfactant mixtures. Stockholm (SE): Stockholm University, Department of Environmental Science and Analytical Chemistry (ACES). 17 p.
[OECD] Organisation for Economic Co-operation and Development. 1992. Test No. 301: Ready Biodegradability, OECD Guidelines for the Testing of Chemicals, Section 3, OECD Publishing, Paris.
[OECD] Organisation for Economic Co-operation and Development. 2004. Emission scenario document on lubricants and lubricant additives [PDF]. Paris (FR): OECD, Environment Directorate. (Series on Emission Scenario Documents No. 10; Report No.: ENV/JM/MONO(2004)21, JT00174617). [accessed 2020 Mar 12].
[OECD] Organisation for Economic Co-operation and Development. 2011. Emission scenario document on the use of metalworking fluids [PDF]. Paris (FR): OECD, Environment Directorate. (Series on Emission Scenario Documents No. 28, Report No. ENV/JM/MONO(2011)18. [accessed 2020 Mar 12].
OECD QSAR Toolbox. [read-across tool]. 2017. Ver. 4.1. Paris (FR): Organisation for Economic Co-operation and Development, Laboratory of Mathematical Chemistry.
Owojori OJ, Waszak K, Roembke J. 2014. Avoidance and reproduction tests with the predatory mite Hypoaspis aculeifer: Effects of different chemical substances. Environ Toxicol Chem. 33(1):230-237.
Schlechtriem C, Nendza M, Hahn S, Zwintscher A, Schüürmann G, Kühne R. 2014. Contribution of non-lipid based processes to the bioaccumulation of chemicals. Dessau-Roßlau (DE): Environmental Research of the Federal Ministry for the Environment, Nature Conservation and Nuclear Safety. Report No.: UBA-FB 00. 92 p.
SimpleTreat [sewage treatment plant removal model]. 2003. Ver. 3.1. Bilthoven (NL): Rijksinstituut voor Volksgezondheid en Milieu (RIVM) [National Institute for Public Health and the Environment]. RIVM, Laboratory for Ecological Risk Assessment, PO Box 1, 3720 BA Bilthoven, The Netherlands.
Sparling DW. 2016. Ecotoxicology Essentials: Environmental contaminants and their biological effects on animals and plants. Academic Press, Elsevier Inc., USA. Chapter 5, Current Use Pesticides. p. 109-147.
Study Submission. 2018. Unpublished confidential studies submitted to Environment and Climate Change Canada (ECCC) under the Chemicals Management Plan initiative. Gatineau (QC): ECCC, Program Development and Engagement Division. Submission received on 2018 Apr 4.
[TEST] Toxicity Estimation Software Tool. 2016. Ver. 4.2.1 Washington (DC): US Environmental Protection Agency.
Unnamed study report. 1996. OECD Guideline 471 Bacterial Reverse Mutation Assays Study. (as cited in ECHA 2019a). [accessed 2018 Nov 15].
Unnamed study report. 2002a. Short-term Repeated Dose Toxicity Study in Rats. (as cited in ECHA 2019a). [accessed 2018 Nov 15].
Unnamed study report. 2002b. EPA OPPTS 870.5395 In Vivo Mammalian Cytogenetics Tests: Erythrocytes Micronucleus Assay. (as cited in ECHA 2019a). [accessed 2018 Nov 15].
Unnamed study report. 2009. OECD Guideline 476 In Vitro Mammalian Cell Gene Mutation Test. (as cited in ECHA 2019a). [accessed 2018 Nov 15].
Unnamed study report. 2010. OECD Guideline 421 Reproduction/Developmental Toxicity Screening Test. (as cited in ECHA 2019a). [accessed 2018 Nov 15].
[US EPA] US Environmental Protection Agency. 2003. Appendix A – Robust Summaries for Reliable Studies, FND Ether Amines category HPV Chemicals Challenge. Washington (DC): US EPA, Toxicology/ Regulatory Services, Inc. Docket No.: 201-14978.
[US EPA] US Environmental Protection Agency. 2010. Screening-level hazard characterization: Fatty Nitrogen Derived amines category. Washington (DC): US EPA, Office of Pollution Prevention and Toxics.
Versteeg DJ, Shorter SJ. 1991. Effect of organic carbon on the uptake and toxicity of quaternary ammonium compounds to the fathead minnow, pimephales promelas. Cincinnati (OH): The Procter & Gamble Company, Environmental Safety Department.
Versar, Inc. 1986. Standard scenarios for estimating exposure to chemical substances during use of consumer products. Vol. II. Consumer use of used motor oil. Springfield (VA): Versar, Inc. Prepared for US Environmental Protection Agency.
[WATERNT] Water Solubility Program [estimation model]. 2010. Ver. 1.01. Washington (DC): US Environmental Protection Agency, Office of Pollution Prevention and Toxics; Syracuse (NY): Syracuse Research Corporation.
[WSKOWWIN] Water Solubility for Organic Compounds Program for Microsoft Windows [estimation model]. 2010. Ver. 1.42. Washington (DC): US Environmental Protection Agency, Office of Pollution Prevention and Toxics; Syracuse (NY): Syracuse Research Corporation.
Appendices
Appendix A. Physical and chemical properties of analogue substances from New Substances Notification (NSN) Program
| Property | NSN analogue 1 | NSN analogue 2 |
|---|---|---|
| Physical state (20°C) | liquid | liquid |
| Melting point (°C) | -12 (pour point) | -20.2 (pour point) |
| Boiling point (°C at 101.3 kPa) | 180 (initial) – 569 (final) | 240 |
| Vapour Pressure (Pa) | 2.95 @ 24°C | 1.8 x 10-4 |
| Relative Density (unitless) | 1.014 @ 15.6°C | 1.02 @ 20°C |
| Water solubility (mg/L) | 70.1 @ 500 LR | P compt 838, S compt 938; amine compt 2140; @ 5500 LR |
| Water solubility (mg/L) | 729 @ 10,000 LR | MSDS: insoluble |
| log Kow (dimen-sionless) | Majority of components >6, minor components 3.0, 4.2, 4.3 | 0.76 |
| log Koc (dimen-sionless) | 1.9 – 2.4 | NA |
Appendix B. Estimated daily intake from oral exposures of humans to TPAE-1
| Age categories | Drinking watera |
|---|---|
| 0 to 5 monthsb (formula fed)c | 1.39 |
| 6 to 11 monthsd | 0.89 |
| 1 yeare | 0.35 |
| 2 to 3 yearsf | 0.30 |
| 4 to 8 yearsg | 0.24 |
| 9 to 13 yearsh | 0.19 |
| 14 to 18 yearsi | 0.19 |
| Greater than or equal to 19 yearsj | 0.22 |
a 10.62 µg/L as reported as the sum of PECs for releases of TPAE-1 at industrial facilities, assuming a 10th percentile dilution value (see Section 6.2.4).
b Assumed to weigh 6.3 kg (Health Canada 2015). It is assumed that no soil ingestion occurs due to typical caregiver practices.
c Exclusively for formula-fed infants, assumed to drink 0.826 L of water per day (Health Canada 2018), where water is used to reconstitute formula. See footnote on drinking water for details.
d Assumed to weigh 9.1 kg (Health Canada 2015), to drink 0 L of water per day (Health Canada 2017). For breast milk-fed infants, assumed to consume 0.632 L of breast milk per day (Health Canada 2018). For formula-fed infants, assumed to drink 0.764 L of water per day (Health Canada 2018), where water is used to reconstitute formula. See footnote (a) for drinking water details.
e Assumed to weigh 11.0 kg (Health Canada 2015), to drink 0.36 L of water per day (Health Canada 2017).
f Assumed to weigh 15 kg (Health Canada 2015), to drink 0.43 L of water per day (Health Canada 2017).
g Assumed to weigh 23 kg (Health Canada 2015), to drink 0.53 L of water per day (Health Canada 2017).
h Assumed to weigh 42 kg (Health Canada 2015), to drink 0.74 L of water per day (Health Canada 2017).
i Assumed to weigh 62 kg (Health Canada 2015), to drink 1.09 L of water per day (Health Canada 2017).
j Assumed to weigh 74 kg (Health Canada 2015), to drink 1.53 L of water per day (Health Canada 2017).
