Screening assessment carboxylic acids group
Official title: Screening assessment carboxylic acids group
Chemical Abstracts Service Registry Numbers 79-09-4, 107-92-6, 112-05-0, 144-62-7
Environment and Climate Change Canada
Health Canada
March 2019
Cat. No.: En14-364/2019E-PDF
ISBN 978-0-660-29735-4
Synopsis
Pursuant to section 74 of the Canadian Environmental Protection Act, 1999 (CEPA), the Minister of the Environment and the Minister of Health have conducted a screening assessment of four substances referred to collectively as the Carboxylic Acids Group. Substances in this group were identified as priorities for assessment as they met categorization criteria under subsection 73(1) of CEPA. The Chemical Abstracts Service Registry Numbers (CAS RNFootnote 1 ), their Domestic Substances List (DSL) names and their common names are listed in the table below.
| CAS RN | DSL name | Common name |
|---|---|---|
| 79-09-4 | Propanoic acid | Propionic acid |
| 107-92-6 | Butanoic acid | n-Butyric acid |
| 112-05-0 | Nonanoic acid | Nonanoic acid |
| 144-62-7 | Ethanedioic acid | Oxalic acid |
In 2011, imported quantities of n-butyric acid and oxalic acid were reported to range from 10 000 to 100 000 kg, imported quantities of nonanoic acid were reported to be 28 925 kg and imported quantities of propionic acid were reported to range from 1 000 000 to 10 000 000 kg. None of these substances were reported to be manufactured in Canada in 2011 above the reporting threshold of 100 kg.
The substances in the Carboxylic Acids Group are reported to be used commercially in Canada in a number of applications such as processing aids, plastic and rubber materials, industrial intermediates, lubricants, solvents, and non-pesticidal agricultural products.
These substances are naturally occurring compounds. Propionic and n-butyric acid are endogenous to humans as they are produced through microbial fermentation in the gastrointestinal tract. Propionic acid, n-butyric acid, and nonanoic acid occur naturally in a variety of foods and may also be used as food flavouring agents. In Canada, propionic acid is also an approved food additive. Propionic acid and oxalic acid are used as components in the manufacture of a variety of food packaging materials. Nonanoic and oxalic acid are used as components in incidental additives for use in food processing establishments. Oxalic acid is also a naturally occurring substance in some foods and has been identified as an ingredient in cleaning products available to consumers in Canada.
All of the substances in the Carboxylic Acids Group are registered pesticide formulants in Canada. They are also all permitted ingredients in natural health products, and propionic, n-butyric, and oxalic acids have been identified in natural health products. Some of these substances are also present in products available to consumers, such as nonanoic acid in eye make-up and cleaning products, and oxalic acid in cleaning products.
The ecological risks of substances in the Carboxylic Acids Group in this screening assessment were characterized using the ecological risk classificationof organic substances (ERC) approach, which is a risk-based approach that employs multiple metrics for both hazard and exposure with weighted consideration of multiple lines of evidence for determining risk classification. Hazard profiles are established based principally on metrics regarding mode of toxic action, chemical reactivity, food web-derived internal toxicity thresholds, bioavailability, and chemical and biological activity. Metrics considered in the exposure profiles include potential emission rate, overall persistence, and long-range transport potential. A risk matrix is used to assign a low, moderate or high level of potential concern for substances on the basis of their hazard and exposure profiles. Based on the outcome of the ERC analysis, propionic acid, n-butyric acid, nonanoic acid, and oxalic acid are considered unlikely to cause ecological harm.
Considering all available lines of evidence presented in this screening assessment, there is low risk of harm to the environment from propionic acid, n-butyric acid, nonanoic acid, and oxalic acid. It is concluded that propionic acid, n-butyric acid, nonanoic acid, and oxalic acid do not meet the criteria under paragraphs 64(a) or (b) of CEPA as they are not entering the environment in a quantity or concentration or under conditions that have or may have an immediate or long-term harmful effect on the environment or its biological diversity or that constitute or may constitute a danger to the environment on which life depends.
With respect to human health, propionic acid has been assessed by the Organization for Economic Cooperation and Development (OECD) in 2007 and by the European Food Safety Authority (EFSA) in 2014; n-butyric acid was assessed by OECD in 2003, and nonanoic acid has been assessed by the European Chemicals Agency (ECHA 2013). Potential sources of exposure of the general population to propionic and n-butyric acid would be from their natural occurrence in the environment and in foods, their uses as food additives or flavouring agents, as well as their uses in natural health products and/or homeopathic products. Potential sources of exposure of the general population to nonanoic acid include its natural occurrence in the environment and in foods, and from use as a food flavouring substance, and uses as an ingredient in an eye make-up product and as an ingredient in a liquid disinfectant solution. On the basis of the information from the above-noted international assessments, propionic acid, n-butyric acid, and nonanoic acid are considered to be substances of low hazard potential and therefore risk to human health is considered to be low.
Exposure to oxalic acid can occur from its use as an ingredient in cleaning products and its natural presence in foods. The available health effects information on oxalic acid indicates potential effects on the reproductive system. The margins of exposure between estimated exposures of oxalic acid and the critical effect level in laboratory studies are considered adequate to address uncertainties in the health effects and exposure databases.
On the basis of the information presented in this screening assessment, it is concluded that propionic acid, n-butyric acid, nonanoic acid, and oxalic acid do not meet the criteria under paragraph 64(c) of CEPA as they are not entering the environment in a quantity or concentration or under conditions that constitute or may constitute a danger in Canada to human life or health.
It is therefore concluded that propionic acid, n-butyric acid, nonanoic acid, and oxalic acid do not meet any of the criteria set out in section 64 of CEPA.
1. Introduction
Pursuant to section 74 of the Canadian Environmental Protection Act, 1999 (CEPA) (Canada 1999), the Minister of the Environment and the Minister of Health have conducted a screening assessment of four substances referred to collectively as the Carboxylic Acids Group to determine whether these substances present or may present a risk to the environment or to human health. The substances in this group were identified as priorities for assessment as they met categorization criteria under subsection 73(1) of CEPA (ECCC, HC [modified 2017]).
The ecological risks of the four substances in the Carboxylic Acids Group were characterized using the ecological risk classification of organic substances (ERC) (ECCC 2016a). The ERC describes the hazard of a substance using key metrics, including mode of action, chemical reactivity, food-web derived internal toxicity threshold, bioavailability, and chemical and biological activity, and considers the possible exposure of organisms in the aquatic and terrestrial environments on the basis of factors including potential emission rates, overall persistence and long-range transport potential in air. The various lines of evidence are combined to identify substances as warranting further evaluation of their potential to cause harm to the environment or as having a low likelihood of causing harm to the environment.
Substances in the Carboxylic Acids Group have been reviewed by various international bodies, such as the Organisation for Economic Cooperation and Development (OECD) (through its Cooperative Chemicals Assessment Programme), the European Chemicals Agency (ECHA) and the European Food Safety Authority (EFSA). Environment and Climate Change Canada and Health Canada consider these assessments as reliable. The data in these assessments are used to inform the health effects characterization for some of the substances in the Carboxylic Acids Group.
This screening assessment includes consideration of information on chemical properties, environmental fate, hazards, uses and exposure, including additional information submitted by stakeholders. Relevant data were identified up to October 2016. However, more recent studies or information provided via internal and external peer consultation may also be cited. Empirical data from key studies as well as results from models were used to reach proposed conclusions.
This screening assessment was prepared by staff in the CEPA Risk Assessment Program at Health Canada and Environment and Climate Change Canada and incorporates input from other programs within these departments. The ecological portion of this assessment is based on the ERC document (published July 30, 2016), which has undergone external review, and was subject to a 60-day public comment period. The human health portions of this assessment have undergone external peer review and/or consultation. Comments on the technical portions relevant to human health were received from Lynne Haber (Department of Environmental Health, College of Medicine, University of Cincinnati), Michael Jayjock (Jayjock & Associates LLC), and Raymond York (RG York & Associates LLC). Additionally, the draft of this screening assessment published on December 30, 2017, was subject to a 60-day public comment period. While external comments were taken into consideration, the final content and outcome of the screening assessment remain the responsibility of Environment and Climate Change Canada and Health Canada.
This screening assessment focuses on information critical to determining whether substances meet the criteria as set out in section 64 of CEPA, by examining scientific information and incorporating a weight-of-evidence approach and precaution.Footnote 2 The screening assessment presents the critical information and considerations on which the conclusions are based.
2. Identity of substances
The Chemical Abstracts Service Registry Numbers (CAS RNFootnote 3 ), Domestic Substances List (DSL) names and common names for the individual substances in the Carboxylic Acids Group are presented in Table 2‑1. A list of additional chemical names (e.g., trade names) is available from the National Chemical Inventories (NCI 2015).
| CAS RN | DSL name (common name) | Chemical structure and molecular formula | Molecular weight (g/mol) |
|---|---|---|---|
| 79-09-4 | Propionic acid (Propionic acid) | 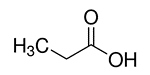 C3H6O2 C3H6O2 |
74.08 |
| 107-92-6 | Butanoic acid (n-Butyric acid) | 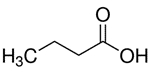 C4H8O2 C4H8O2 |
88.11 |
| 112-05-0 | Nonanoic acid | 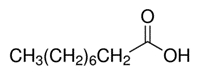 C9H18O2 C9H18O2 |
158.24 |
| 144-62-7 | Ethanedioic acid (Oxalic acid) | 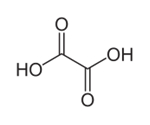 C2H2O4 C2H2O4 |
90.04 |
3. Physical and chemical properties
A summary of physical and chemical properties of the substances in the Carboxylic Acids Group are presented in Table 3‑1, with the range in values indicated for each property. Additional physical and chemical properties are presented in ECCC (2016b).
| Property | Propionic acid | n-Butyric acid | Nonanoic acid | Oxalic acid | Key references |
|---|---|---|---|---|---|
| Physical state | oily colourless liquid | oily colourless liquid | oily colourless liquid | white solid | ChemIDplus 1993-a,b,c,d |
| Melting point (°C) | -20.7 | -5.7 | 12.3 | 189.5 | ChemIDplus 1993-a,b,c,d |
| Vapour pressure (Pa) | 470 | 220 | 2.20 | 0.031 | ChemIDplus 1993-a,b,c,d |
| Henry’s law constant (atm·m3/mol) | 4.45 x 10 -7 | 5.35 x 10 -7 | 1.62 x 10 -6 | 1.43 x 10 -10 | ChemIDplus 1993-a,b,c,d |
| Water solubility (mg/L) | 1.0 x 10 6 | 60 000 | 284 | 220 000 | ChemIDplus 1993-a,b,c,d |
| log Kow (dimensionless) | 0.33 | 0.79 | 3.42 | -2.22* | ChemIDplus 1993-a,b,c,d |
| pKa1 (dimensionless) | 4.87 | 4.82 | 4.95 | 1.46 | HSDB 1983-a,b,c, Haynes 2016 |
| pKa2 (dimensionless) | NA | NA | NA | 4.40 | Haynes 2016 |
Abbreviations: Kow, octanol–water partition coefficient; pKa, acid dissociation constant
*; reported value was estimated
4. Sources and uses
Propionic acid, n-butyric acid, and nonanoic acid occur naturally in a wide range of foods, including fruits, vegetables, dairy products, meats, coffee, and nuts and grains; n‑butyric acid and nonanoic acid occur naturally in some essential oils (Burdock 2009; HSDB 1983- a,b,c). The main source of dietary oxalic acid is from its natural occurrence in plants and plant products, primarily seeds and leafy plants related to spinach and rhubarb (Holmes and Kennedy 2000). Certain substances may also be present in food as a result of their use as food flavouring agents or food additives, as elaborated upon below.
Propionic and n-butyric acid are considered to be volatile fatty acids (VFAs) and can be produced metabolically from the fermentation of plant materials, such as cellulose, fibres, starches, and sugars (Bergman 1990). They are produced in the greatest amounts in herbivorous animals and, to a lesser extent, in humans through microbial fermentation in the gastrointestinal tract (Bergman 1990). In mammals, oxalic acid is produced as the end product of metabolism of amino acids, glycolate and ascorbic acid (EMA 2003). Oxalic acid may be present in the atmosphere through photochemical oxidation of other hydrocarbons and cyclic olefins in the atmosphere (Kawamura and Kaplan 1987). The substance was also reported to be present in vehicle exhaust (Kawamura and Kaplan 1987).
Use of a compound structurally similar to oxalic acid, i.e., oxalic acid dihydrate (CAS RN 6153-56-6), may represent a source of exposure to oxalic acid. Oxalic acid dihydrate is registered as an active ingredient for pesticide use in Canada and is regulated under the Pest Control Products Act. It is also present in cleaning products available to consumers in Canada.
All substances in this grouping have been included in surveys issued pursuant to a CEPA section 71 notice (ECCC 2013). None of these substances were reported to be manufactured in Canada in 2011. Table 4‑1 presents a summary of the total manufacture and import quantities for the Carboxylic Acids Group in 2011.
| Common name | Total importsa (kg) |
|---|---|
| Propionic acid | 1 000 000–10 000 000 |
| n-Butyric acid | 10 000–100 000 |
| Nonanoic acid | 28 925 |
| Oxalic acid | 10 000–100 000 |
a Values reflect quantities reported in response to the survey[s] conducted under section 71 of CEPA (ECCC 2013). See survey for specific inclusions and exclusions (Schedules 2 and 3).
In Canada, propionic acid is used as a formulant in pest control products. Propionic acid and its sodium and calcium salts are permitted for use in food as a food additive as prescribed in Health Canada’s List of Permitted Preservatives, functioning as preservatives in certain food items such as breads and certain types of cheese. It is also identified as an ingredient in the manufacture of some food packaging materials (inks on the exterior of materials used to package foods and in pipe coverings) (personal communications, emails from Food Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, dated 2017; unreferenced).
In the United States, the national production volume for propionic acid, n-butyric acid and oxalic acid ranged from approximately 9 to 204 million kg (between 20 and 450 million pounds), respectively, for the year 2011 (ChemView 2013-a,b,c). Manufacture quantities of nonanoic acid in 2011 were over 9 million kg (20 million pounds) (Chemview 2013-a,b,c).
In Europe, propionic acid may also be used as a food flavouring substance and a food additive (EU Food Flavouring Database [modified 2016], EU Food Additives Database [modified 2018]), and in the United States, the substance is permitted under 21 CFR 184.1081 as an ingredient in food with no limitation other than current good manufacturing practice and is considered “generally recognized as safe” (GRAS) (US FDA 2016a; FCC 2016). Propionic acid is listed in the Natural Health Products Ingredients Database (NHPID) with a medicinal role and classified as a natural health product (NHP) substance falling under item 2 (an isolate) of Schedule 1 to the Natural Health Products Regulations (NHPR), as well as listed with a non-medicinal role for use as a flavour enhancer or preservative antimicrobial (NHPID 2018). It is listed in the Licensed Natural Health Products Database (LNHPD) as being present as non-medicinal ingredient in a currently licensed NHP (LNHPD 2018). There are several commercial uses of propionic acid in Canada, including use as a processing aid in paints and coatings, water treatments, and plastic and rubber materials (ECCC 2013).
n-Butyric acid is used as a formulant in pest control products in Canada. It is listed in the NHPID with a medicinal role and classified as a NHP substance falling under item 2 (an isolate) of Schedule 1 to the NHPR, and with a non-medicinal role for use as a flavour enhancer, as well as with a homeopathic role for use as a medicinal ingredient in NHPs licensed as homeopathic medicines (NHPID 2018). It is also listed in the LNHPD as being present as a medicinal or non-medicinal ingredient in a limited number of currently licensed NHPs, including homeopathic medicines (LNHPD 2018). According to information submitted pursuant to a CEPA section 71 notice, n-butyric acid was reported to have food and beverage uses in Canada as an ingredient in food flavours (ECCC 2017). n-Butyric acid is used as a food flavouring agent and regulated in the United States under 21 CFR 182.60 as a GRAS synthetic flavour (US FDA 2016b). In Europe, it is regulated under EU No. 872/2012 as a flavouring substance (EU 2012). Commercial uses of n-butyric acid in Canada include non-pesticidal agricultural products (ECCC 2013). The majority of n-butyric acid manufactured in the United States is used as a chemical intermediate in the production of cellulose acetate butyrate (CAB) (OECD 2003). CAB sheets are used in a variety of applications, such as thermoplastics, face shields, goggles, and automotive coatings (OECD 2003). Other global uses of n-butyric acid include its use as a chemical intermediate in the production of pharmaceuticals, emulsifiers and disinfectants, leather tanning agents, butyrate ester perfumes, and food flavouring agents (SRI 1999; Hawley 1981; Reimenshneider 1986; Furia 1972, as cited in OECD 2003).
In Canada, nonanoic acid is used as a formulant in pest control products and is also present as an ingredient in an eye make-up product and in a liquid ready-to-use disinfectant solution (personal communications, emails from Pest Management Regulatory Agency to Existing Substances Risk Assessment Bureau, Health Canada, dated 2016; unreferenced; personal communications, emails from Consumer Product Safety Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, dated 2016; unreferenced; personal communications, emails from Therapeutic Products Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, dated 2016; unreferenced). Nonanoic acid has been identified as an ingredient in sanitizers for use in food processing establishments (personal communications, emails from Food Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, dated 2017; unreferenced). There are several commercial uses of nonanoic acid in Canada, including its use in the manufacture of intermediates, lubricants and lubricant additives, and pest control products (ECCC 2013). Nonanoic acid is listed in the NHPID with a non-medicinal role for use as a flavour enhancer; however, it is not listed in the LNHPD as being present in currently licensed NHPs (LNHPD 2018; NHPID 2018). Nonanoic acid is not listed as a permitted food additive in Canada; however, it is approved for use in the United States and Europe as a synthetic flavour and was reported to have food uses in alcoholic beverages, baked goods, dairy products, meat products, and snacks, among others (US FDA 2016c; EU Food Flavourings Database [modified 2016]; Burdock 2009). Global uses also include pest control applications (EPA 1998).
In Canada, oxalic acid is listed as a formulant in several pest control products (personal communications, emails from Pest Management Regulatory Agency to Existing Substances Risk Assessment Bureau, Health Canada, dated 2016; unreferenced). Use of a compound structurally similar to oxalic acid, i.e., oxalic acid dihydrate (CAS RN 6153-56-6), may represent a source of exposure to oxalic acid. Oxalic acid dihydrate is registered as an active ingredient for pesticide use in Canada and is regulated under the Pest Control Products Act. It is also present in consumer cleaning products in Canada. Oxalic acid has been identified as an ingredient used in the manufacture of a limited number of food packaging materials (e.g., inks used on the exterior surface of food packaging materials and interior coating or lining of cans) (personal communications, emails from Food Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, dated 2017; unreferenced). It may also be used in cleaners and laundry detergents for food processing establishments (personal communications, emails from Food Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, dated 2016; unreferenced). Oxalic acid was not identified as a food additive in Canada or elsewhere. Oxalic acid is listed in the NHPID with a homeopathic role for use as a medicinal ingredient in NHPs licensed as homeopathic medicines. It is listed in the LNHPD as being present in a limited number of NHPs currently licensed as homeopathic medicines (NHPID 2018, LNHPD 2018). It was also identified as being used in cleaning products available to consumers in Canada, namely all-purpose sprays designed for household rust stains, a powdered abrasive cleaner, boat hull-cleaning solutions to remove waterline stains, and a boat bottom cleaner to remove scum line and rust stains (SDS 2014a; SDS 2014b; SDS 2015a; SDS 2015b; SDS 2015c). Industrial applications of oxalic acid in Canada include uses as a solvent and corrosion inhibitor in automotive and cleaning and furnishing care, as well as uses in paint and coating applications, among others (ECCC 2013). Globally, oxalic acid is used in cleaning and washing products, pH regulators and water treatment products, metal and non-metal surface treatment products, laboratory chemicals, coating products, and as an intermediate in the manufacture of other chemicals (ECHA c2007-2016).
A summary of these uses can be found in Table 4‑2 below.
| Use | Propionic acid | Butyric acid | Nonanoic acid | Oxalic acid |
|---|---|---|---|---|
| Food additivea | Y | N | N | N |
| Food packaging materialsb | Y | N | N | Y |
| Incidental food additiveb | N | N | Y | Y |
| Medicinal or non-medicinal ingredients in disinfectant, human or veterinary drug productsc | N | N | Y | N |
| Natural Health Products Ingredients Databased | Y | Y | Y | Y |
| Medicinal or non-medicinal ingredients in licensed natural health productse | Y | Y | N | Y |
| Present in cosmetics, based on notifications submitted under the Cosmetic Regulationsf | N | N | Y | N |
| Formulant in registered pest control productsg | Y | Y | Y | Y |
Abbreviations: Y = yes this use was reported for this substance; N = no this use was not reported for this substance
a personal communications, emails from Food Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, dated 2016; unreferenced.
b personal communications, emails from Food Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, dated 2016; unreferenced.
c personal communications, emails from Therapeutic Products Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, dated 2016; unreferenced.
d personal communications, emails from Natural and Non-prescription Health Products Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, dated 2016; unreferenced.
e personal communications, emails from Natural and Non-prescription Health Products Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, dated 2016; unreferenced.
f personal communications, emails from Consumer Product Safety Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, dated 2016; unreferenced.
g personal communications, emails from Pest Management Regulatory Agency to Existing Substances Risk Assessment Bureau, Health Canada, dated 2016; unreferenced.
5. Potential to cause ecological harm
5.1 Characterization of ecological risk
The ecological risks of substances in the Carboxylic Acids Group were characterized using the ecological risk classification of organic substances (ERC) approach (ECCC 2016a). The ERC is a risk-based approach that considers multiple metrics for both hazard and exposure, with weighted consideration of multiple lines of evidence for determining risk classification. The various lines of evidence are combined to discriminate between substances of lower or higher potency and lower or higher potential for exposure in various media. This approach reduces the overall uncertainty with risk characterization compared to an approach that relies on a single metric in a single medium (e.g., LC50) for characterization. The following summarizes the approach, which is described in detail in ECCC (2016a).
Data on physical-chemical properties fate (chemical half-lives in various media and biota, partition coefficients, fish bioconcentration), acute fish ecotoxicity, and chemical import or manufacture volume in Canada were collected from scientific literature, from available empirical databases (e.g., OECD QSAR Toolbox), and from responses to surveys under section 71 of CEPA, or they were generated using selected quantitative structure-activity relationship (QSAR) or mass-balance fate and bioaccumulation models. These data were used as inputs to other mass-balance models or to complete the substance hazard and exposure profiles.
Hazard profiles were established principally on the basis of metrics regarding mode of toxic action, chemical reactivity, food web-derived internal toxicity thresholds, bioavailability, and chemical and biological activity. Exposure profiles were also established using multiple metrics, including potential emission rate, overall persistence, and long-range transport potential. Hazard and exposure profiles were compared to decision criteria in order to classify the hazard and exposure potentials for each organic substance as low, moderate, or high. Additional rules were applied (e.g., classification consistency, margin of exposure) to refine the preliminary classifications of hazard or exposure.
A risk matrix was used to assign a low, moderate, or high classification of potential risk for each substance on the basis of its hazard and exposure classifications. ERC classifications of potential risk were verified using a two-step approach. The first step adjusted the risk classification outcomes from moderate or high to low for substances that had a low estimated rate of emission to water after wastewater treatment, representing a low potential for exposure. The second step reviewed low risk potential classification outcomes using relatively conservative, local-scale (i.e., in the area immediately surrounding a point-source of discharge) risk scenarios, designed to be protective of the environment, to determine whether the classification of potential risk should be increased.
ERC uses a weighted approach to minimize the potential for both over- and under-classification of hazard, exposure and subsequent risk. The balanced approaches for dealing with uncertainties are described in greater detail in ECCC 2016a. The following describes two of the more substantial areas of uncertainty. Error in empirical or modeled acute toxicity values could result in changes in classification of hazard, particularly metrics relying on tissue residue values (i.e., mode of toxic action), many of which are predicted values from QSAR models. The impact of this error is mitigated, however, by the fact that overestimation of median lethality will result in a conservative (protective) tissue residue value used for critical body residue (CBR) analysis. Error in underestimation of acute toxicity will be mitigated through the use of other hazard metrics, such as structural profiling of mode of action, reactivity and/or estrogen binding affinity. Changes or errors in chemical quantity could result in differences in classification of exposure as the exposure and risk classifications are highly sensitive to emission rate and use quantity. The ERC classifications thus reflect exposure and risk in Canada based on what is believed to be the current use quantity and may not reflect future trends.
Critical data and considerations used to develop the substance-specific profiles for the four substances in the Carboxylic Acids Group and the hazard, exposure and risk classification results are presented in ECCC (2016b).
The hazard, exposure and risk classifications for the four substances in the Carboxylic Acids Group are summarized in Table 5‑1.
| Common name | ERC hazard classification | ERC exposure classification | ERC risk classification |
|---|---|---|---|
| Propionic acid | low | high | low |
| n-Butyric acid | low | low | low |
| Nonanoic acid | low | low | low |
| Oxalic acid | low | low | low |
According to information considered under ERC, propionic acid was classified as having a high exposure potential on the basis of high reported use quantities and long-range transport potential. Given its low hazard potential, propionic acid was classified as having a low potential for ecological risk. It is unlikely that propionic acid results in concerns for the environment in Canada given current use patterns.
On the basis of low hazard and low exposure classifications according to the information considered under ERC, n-butyric acid, nonanoic acid, and oxalic acid were classified as having a low potential for ecological risk. It is therefore unlikely that these substances result in concerns for the environment in Canada.
6. Potential to cause harm to human health
6.1 Propionic acid
6.1.1 Exposure assessment
The following section provides general information on exposure to propionic acid. As propionic acid is considered to be of low hazard potential (Section 6.1.2), quantitative estimates of exposure to the general population were not derived.
No Canadian or recent international data on levels in indoor air, drinking water, or soil were identified for propionic acid.
Inhalation exposure to propionic acid may occur from its presence in ambient air. Propionic acid was measured in ambient air near three intensive livestock operations in southern Alberta, Canada (McGinn et al. 2003). The authors noted a reduction in the total volatile fatty acids (VFAs), including propionic acid (which can be released from manure) concentrations, when samples were taken 100 m and 200 m downwind of the feedlots.
In Canada, propionic acid and its sodium and calcium salts are permitted food additives in certain foods such as breads and certain types of cheese, which may result in dietary exposure to the substance. Depending on the category of food to which it is permitted to be added, propionic acid can be used up to a maximum of 2 000 ppm or 3 000 ppm (2 000 mg/kg or 3 000 mg/kg) (Health Canada 2016). Propionic acid has also been identified as being used in the manufacture of some food packaging materials, but for which there would be no contact with food. Dietary exposure to propionic acid from its use in food packaging materials is therefore not expected (personal communications, emails from Food Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, dated 2017; unreferenced).
Oral exposure to propionic acid may also occur from its presence as a non-medicinal ingredient in a liquid glucosamine natural health product (LNHPD 2018).
6.1.2 Health effects assessment
Propionic acid has been reviewed by the OECD (2007) and EFSA (2014), and these reviews were used to inform the health effects characterization of this substance. A literature search was conducted for the period of January 2006 to October 2016, and no studies that could result in a different health effects characterization from those of OECD (2007) and EFSA (2014) assessments were identified.
Propionic acid is found naturally in humans as a normal intermediary metabolite that represents up to 4% of the normal total plasma fatty acids (OECD 2007).
Repeated dose toxicity
Both the OECD and EFSA concluded that the only reported adverse effect to propionic acid exposure was observed at the site of contact and was a consequence of its irritating property, and that no systemic toxicity was seen in repeated dose studies.
Several repeated dose studies were identified by the OECD. In one study, male and female dogs were administered doses up to 3% (approximately 1 800 mg/kg bw/day) of propionic acid in the diet for 100 days. There was no mortality and no clinical signs of toxicity. Three high-dose animals had point-of-contact effects that included diffuse epithelial hyperplasia of the esophageal mucosa. The incidence of lesions of the esophagus in the high-dose animals after a 6-week recovery period was similar to controls (BASF 1988, as cited in OECD 2007).
In another repeated dose oral study, male and female Sprague-Dawley rats were administered up to 5%, or approximately 3 300 mg/kg bw/day, propionic acid in a pulverized diet for 91 days. There was no mortality. Males in the high-dose group exhibited decreased body weight gain. No other clinical signs of toxicity were observed. Point-of-contact effects were observed in the epithelium of the rat forestomach mucosa in rats in the high-dose group; these changes were not observed after a 6-week recovery period (BASF AG 1971, as cited in OECD 2007).
Additional feeding studies in rats ranging from 28 days to lifetime exposure were identified. These studies focused only on point-of-contact effects in the forestomach. The outcome of the studies varied with the type of diet (i.e., pelleted vs. powdered), but were consistent with point-of-contact effects observed in other studies.
Carcinogenicity
A lifetime feed study in which male rats were administered doses up to 4% (approximately 2 700 mg/kg bw/day) propionic acid was identified by the OECD (2007). On the basis of the information from this study, OECD concluded that propionic acid is not predicted to have carcinogenic potential at the point of contact (forestomach). The changes observed upon feeding of high doses of propionic acid are the result of chronic irritation and inflammation and the associated hyperplastic proliferative repair response.
The EFSA (2014) also concluded that propionic acid was of no concern with respect to carcinogenicity.
Genotoxicity
Propionic acid tested negative in in vitro and in vivo assays. Therefore, both OECD (2007) and EFSA (2014) concluded that propionic acid was of no concern with respect to genotoxicity.
Reproductive/developmental toxicity
With respect to reproductive and developmental toxicity, no reproductive studies were identified for propionic acid. However, in a 100-day feeding study in dogs fed up to 1 800 mg/kg bw/day propionic acid, no evidence of toxicity to male or female reproductive organs was found (OECD 2007).
The OECD also reviewed a 1971 study in which groups of male and female Sprague-Dawley rats were administered a daily diet containing up to 50 000 ppm propionic acid (or 5% of the diet). There were no effects observed on male or female reproductive organs (OECD 2007).
No developmental studies were identified for propionic acid. However, both OECD and EFSA considered results from a study conducted with calcium propionate. The study showed that there was no effect on litter size or viability and no increase in fetal abnormalities in mice and rats administered calcium propionate during gestation at doses up to 300 mg/kg bw/day or in hamsters and rabbits at doses up to 400 mg/kg bw/day (OECD 2007; EFSA 2014).
6.1.3 Characterization of risk to human health
The Joint FAO/WHO Expert Committee on Food Additives (JECFA) evaluated a flavouring group of saturated aliphatic acyclic linear primary alcohols, aldehydes, and acids, including propionic acid, at its 49th meeting (WHO 1998). It concluded that substances in that flavour grouping could be predicted to undergo complete metabolism to endogenous products via the fatty acid and tricarboxylic acid pathways following ingestion. Given that endogenous levels of metabolites from these substances would not give rise to perturbations outside the physiological range, JECFA concluded that their use as food flavouring agents would not be a safety concern. In considering this, JECFA also maintained the group acceptable daily intake (ADI) of “not limited” that had previously been established for food additive uses of propionic acid and its sodium, potassium, and calcium salts.
In 2014, the European Food Safety Authority (EFSA) re-evaluated the safety of propionic acid and its salts, which are authorized food additives in the EU. The EFSA Panel concluded that food additive uses of propionic acid and its salts are the major contributor to dietary exposure, but that there would be no safety concern from the maximum concentrations (up to 3 000 ppm) authorized for use in the EU (EFSA 2014). Like JECFA, EFSA established a group ADI for propionic acid and its salts of “not specified.”
The OECD (2007) and the EFSA (2014) have concluded that no systemic toxicity was seen in animals after repeated dose exposure to propionic acid (animals were tested with doses as high as 3 300 mg/kg bw/day). The OECD and EFSA concluded that propionic acid is not predicted to have carcinogenic or genotoxic potential. No evidence of adverse effects on reproductive organs or of developmental effects was observed in repeated dose studies on various species. In addition, propionic acid is found naturally in humans as a normal intermediary metabolite that represents up to 4% of the normal total plasma fatty acids. Propionic acid is therefore considered to be of low hazard potential.
As propionic acid is considered to be of low hazard potential, derivation of estimates of exposure from sources and uses identified in Section 4 were not considered meaningful, and risk to human health is considered to be low.
6.2 n-Butyric acid
6.2.1 Exposure assessment
The following section provides general information on exposure to n-butyric acid. As n-butyric acid is considered to be of low hazard potential (Section 6.2.2), quantitative estimates of exposure to the general population were not derived.
Inhalation exposure to n-butyric acid may occur from its presence in ambient air. As with propionic acid, n-butyric acid was measured in ambient air (which can be released from manure) near intensive livestock operations in southern Alberta, Canada. McGinn et al. (2003) identified n-butyric acid in outdoor air near three cattle feedlots, noting that total VFA concentrations decreased when measured at increasing distances from the facilities.
No Canadian or recent international data on levels in indoor air, drinking water, or soil were identified for n-butyric acid.
Dietary exposure to n-butyric acid may occur from its use as a food flavour ingredient in flavouring concentrates in Canada (ECCC 2017). In addition, oral exposure to n-butyric acid may also occur from its presence as a non-medicinal ingredient in a limited number of natural health products in Canada (LNHPD 2018).
6.2.2 Health effects assessment
n-butyric acid was reviewed by the OECD (2003), and its review was used to inform the health effects characterization of this substance.
A literature search was conducted for the period of January 2002 to October 2016, and no studies that could result in a different health effects characterization from OECD (2003) assessment were identified.
The OECD assessed n-butyric acid and n-butyric anhydride as a group as these two substances are closely related. Anhydride rapidly hydrolyzes in the presence of water to form the acid and these substances share toxicity characteristics. The OECD also used information from n-butanol and n-butyl acetate to identify the hazards associated with systemic exposure to n-butyric acid since n-butyl acetate and n-butanol are metabolic precursors of n-butyric acid.
n-butyric acid can be produced metabolically from the fermentation of plant materials, such as cellulose, fibres, starches, and sugars. It is produced in the greatest amounts in herbivorous animals and, to a lesser extent, in humans through microbial fermentation in the gastrointestinal tract (Bergman 1990, as cited in OECD 2003).
Repeated dose toxicity
The OECD identified several repeated dose studies. In an inhalation study, male and female Sprague-Dawley rats were exposed to concentrations of 0, 500, 1 500 or 3 000 ppm (approximately 0, 2 376, 7 128, or 14 256 mg/m3) of n-butyl acetate, 6 hours per day, 5 days per week for 13 weeks. Systemic effects including decreased body weight and feed consumption, increased testes weights, increased adrenal weights, and signs of localized necrosis of olfactory epithelium were observed at the mid and high doses. Minimal narcosis and sedation effects were also observed in rats in the mid- and high-dose groups. In addition, reduced spleen weights and increased lung weights were observed in the high-dose group (David et al. 2001).
The OECD noted that degeneration of the olfactory epithelium following exposures of > 1 500 ppm (7 128 mg/m3) was observed. This is considered to represent a common lesion in rats exposed to acetate esters of alcohols caused by the liberation of acetic acid in these cells from the hydrolysis of the ester linkage. Since rats are obligate nose-breathers, the delivered dose to this portion of the nose is higher in rats than humans, and the significance of this lesion in human health is questionable.
In an earlier study by the same author and with the same study design, rats developed central nervous system effects (reduced activity) during exposure, but with rapid recovery once the exposure ended (David et al. 1998, as cited in OECD 2003). The OECD also identified a 13-week oral study in rats using dose levels of n-butanol up to 500 mg/kg bw/day by gavage, and reported a NOAEL of 125 mg/kg/day and a LOAEL of 500 mg/kg bw/day, based on transient post-dose ataxia and hypo activity (TRL 1986, as cited in OECD 2003). These effects are transient and therefore not considered truly adverse.
Carcinogenicity
No data has been identified by the OECD (2003) or in the literature since 2003.
Genotoxicity
n-butyric acid was not mutagenic in Ames tests with or without metabolic activation. There was also no mutagenic effect observed in cultured Chinese hamster lung (CHL) cells. In an in vivo assay, male and female NMRI mice were exposed to n-butanol via gavage at doses of up to 2 000 mg/kg. No chromosome-damaging effects were observed; and there were no indications of any impairment of chromosome distribution in the course of mitosis (no spindle poison effect) (OECD 2003).
Reproductive/developmental toxicity
Several reproductive toxicity studies were identified by the OECD. In an inhalation study, female Sprague-Dawley rats were exposed to n-butanol at concentrations up to 6 000 ppm (approximately 18 192 mg/m3), 7 hours per day on gestation day (GD) 1 to 20. No detectable effect on pregnancy rate was observed. In another study conducted by the same author, male rats were exposed to n-butyl acetate, via inhalation, at concentrations up to 3 000 ppm (approximately 14 256 mg/m3), 6 hours per day, for 14 weeks. No testicular toxicity was observed (Nelson et al. 1989a, as cited in OECD 2003).
Two developmental studies were reported by OECD (2003) using n-butanol. In one study, pregnant Sprague-Dawley rats were exposed via inhalation to n-butanol concentrations of 0, 3 500, 6 000 or 8000 ppm (approximately 0, 10 612, 18 192 or 24 256 mg/m3), 7 hours per day during GD 1 to 19. Maternal effects such as decreased feed consumption were observed in the 6 000 and 8 000 ppm groups, and narcosis and mortality were observed in the 8 000 ppm group only. Very slight decreases in fetal body weights were observed in the 6 000 and 8 000 ppm groups. The NOAEL determined by the author, both for maternal and fetal effects, was 3 500 ppm (10 612 mg/m3) (Nelson et al. 1989b, as cited in OECD 2003). In a developmental neurotoxicity study, male and female rats were exposed via inhalation to n-butanol concentrations as high as 6 000 ppm (approximately 18 192 mg/m3). Males were exposed for 6 weeks prior to mating with unexposed females, and females were exposed only during GD 1 to 20 for 3 weeks. The NOAEL for developmental neurotoxicity was determined by the author to be the highest dose tested (6 000 ppm, or 18 192 mg/m3) for both male and female rats (Nelson et al. 1989a, as cited in OECD 2003)
Studies conducted with n-butyl acetate in rats and rabbits were also identified as supporting data for n-butyric acid. No developmental effects were observed in these studies (Hackett et al. 1982, as cited in OECD 2003).
6.2.3 Characterization of risk to human health
JECFA evaluated a group of flavouring agents that included n-butyric acid (WHO 1998). It concluded that substances in that flavour grouping (including n-butyric acid) could be predicted to undergo complete metabolism to endogenous products via the fatty acid and tricarboxylic acid pathways following ingestion. Given that endogenous levels of metabolites from these substances would not give rise to perturbations outside the physiological range, JECFA concluded that their use as food flavouring agents would not be a safety concern.
On the basis of the available information on precursors of n-butyric acid (n-butanol and n-butyl acetate), n-butyric acid is not predicted to have carcinogenic or genotoxic potential and is not expected to cause reproductive and developmental effects. Systemic effects such as increased testes weights and increased adrenal weights were observed in experimental animals following repeated inhalation exposure for 13 weeks to n-butyric acid, but only at high doses, as high as 7 128 or 14 256 mg/m3 (equivalent to approximately 2 210 or 4 420 mg/kg bw/day, respectively), doses that are considered irrelevant to potential human exposure levels, especially since inhalation is not expected to be a predominant route of exposure to n-butyric acid considering its use pattern for the general population. In addition, only transient effects were observed at the highest dose tested in a repeated dose oral study. As such, n-butyric acid is considered to be of low hazard potential.
As n-butyric acid is considered to be of low hazard potential, derivation of estimates of exposure from sources and uses identified in Section 4 were not considered meaningful and risk to human health is considered to be low.
6.3 Nonanoic acid
6.3.1 Exposure assessment
The following section provides general information on exposure to nonanoic acid. As nonanoic acid is considered to be of low hazard potential (Section 6.3.2), quantitative estimates of exposure to the general population were not derived.
No Canadian or recent international data on levels in ambient air, indoor air, or drinking water were identified for nonanoic acid, however inhalation and incidental oral exposures to nonanoic acid may occur from its natural presence in soil and ambient air (Rogge et al. 1993; ECHA 2013). Nonanoic acid was identified to be present in household dust in a 2011 National Research Council of Canada (NRC) report (NRC 2011). Concentrations of nonanoic acid in dust samples were reported to range from 0.67 to 654.93 μg/g dust (n=37) with an arithmetic mean of 36.83 μg/g dust.
Although there is no definitive information on the use of nonanoic acid as a food flavouring agent in Canada, it is possible that the substance is present as a flavouring agent in foods sold in Canada based on use of the substance as a synthetic food flavouring agent in the US and Europe, resulting in dietary exposure. In Canada, nonanoic acid has been identified to be used as an ingredient in sanitizers for use in food processing establishments however dietary exposure, if any, from this use is expected to be negligible (personal communication, emails from Food Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, dated January 2017; unreferenced).
Nonanoic acid was reported as an ingredient in a limited number of cosmetics and products available to consumers in Canada that may result in dermal exposure to the substance. A non-oily non-permanent eye make-up product was reported to contain nonanoic acid (personal communications, emails from Consumer Product Safety Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, dated January 2016; unreferenced). The substance was also reported to be present at low concentrations in a ready-to-use disinfectant solution used to disinfect and sanitize surfaces (MSDS 2011; TDS 2011; personal communications, emails from Therapeutic Products Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, dated March 2016).
6.3.2 Health effects assessment
Nonanoic acid was reviewed internationally by ECHA (2013), and its review was used to inform the health effects characterization of this substance. A literature search was conducted for the period of January 2012 to October 2016, and no studies that could result in a different health effects characterization from that of ECHA (2013) assessment was identified.
Repeated dose toxicity
Several repeated dose toxicity studies were identified. In a 28-day oral study, male and female Wistar rats were administered nonanoic acid via gavage at 0, 50, 150 or 1 000 mg/kg bw/day in concentrations of 0%, 1%, 3% or 20% in propylene glycol as vehicle. Slight to marked hyperplasia of the squamous epithelium of the forestomach was observed in the high-dose group only. Therefore, a NOAEC of 150 mg/kg bw/day was established. However, ECHA indicated that this effect was assumed to be associated with its local irritant property rather than by systemic action (ECHA 2013).
In addition, ECHA described several repeated studies with medium chain triglycerides to further support their conclusion that there were no effects toxicologically relevant at very high doses in the available oral repeated dose studies from nonanoic acid (ECHA 2013). ECHA concluded that there was no need to conduct further studies because of a number of factors, including the ubiquity of nonanoic acid and other similar fatty acids in nature, the knowledge of metabolic pathways—similar for all fatty acids—which is complete catabolism for energy supply or conversion to fat suitable for storage, and because of the lack of systemic effects in the available repeated studies.
Carcinogenicity
Nonanoic acid was tested for the dermal carcinogenicity in a mouse bioassay. Pure nonanoic acid was applied on the back of the clipped skin of male C3H/HeJ mice, at doses of 25 mg or 50 mg, twice weekly for 80 weeks. Controlled groups included one untreated group, one negative control group treated with 50 mg mineral oil, and one positive control group treated with 0.05% benzo(a)pyrene (BaP) in mineral oil. There was no evidence of gross skin tumours in mice treated with nonanoic acid or in the two negative control groups. Non-neoplastic skin lesions were observed in the exposed group at a rate as high as in the negative control groups (ECHA 2013).
No oral carcinogenicity studies were identified. However, ECHA (2013) indicated that the hyperplasia of the squamous epithelium of forestomach observed in the above 28-day gavage study is not considered to be of relevance for human cancer risk assessment. This conclusion is supported by the absence of genotoxic effects and the knowledge of kinetics and metabolism of fatty acids (ECHA 2013).
Genotoxicity
Nonanoic acid tested negative in bacteria mutation assays with or without metabolic activation. It tested negative in gene mutation and chromosome aberration assays. It also tested negative in an in vivo mouse micronucleus assay (ECHA 2013).
Reproductive/developmental toxicity
No reproductive studies were identified for nonanoic acid. However, studies with medium chain triglyceride showed no effects on fertility in rats fed up to 8 000 mg/kg bw/day (ECHA 2013).
In a developmental toxicity study, pregnant CD rats were exposed to nonanoic acid in corn oil by oral intubation at 0 and 1 500 mg/kg bw/day on days 6 through 15 of gestation. No maternal or developmental effects were observed (ECHA 2013).
6.3.3 Characterization of risk to human health
JECFA (1997) evaluated a group of flavouring agents that included nonanoic acid. It concluded that substances in that flavour grouping (including nonanoic acid) could be predicted to undergo complete metabolism to endogenous products via the fatty acid and tricarboxylic acid pathways following ingestion. Given that endogenous levels of metabolites from these substances would not give rise to perturbations outside the physiological range, JECFA concluded that their use as food flavouring agents would not be a safety concern.
ECHA (2013) has concluded that no systemic toxicity was seen in experimental animals after repeated dose exposure to levels up to 1 000 mg/kg bw/day of nonanoic acid. It is considered to have no carcinogenic or genotoxic potential. No reproductive studies were identified, but studies with medium-chain triglycerides showed no effects on fertility in rats fed up to 8 000 mg/kg bw/day, and no evidence of developmental effects were observed in a rat study. ECHA (2013) concluded that the knowledge of metabolic pathways for fatty acids in general, which is complete catabolism for energy supply or conversion to fat suitable for storage, is consistent with the expectation that there would be no systemic effects observed in repeated studies even at very high doses. As such, nonanoic acid is considered to be of low hazard potential.
As nonanoic acid is considered to be of low hazard potential, derivation of estimates of exposure from sources and uses identified in Section 4 were not considered meaningful, and risk to human health is considered to be low.
6.4 Oxalic acid
6.4.1 Exposure assessment
Environmental media and food
No recent Canadian or international data on oxalic acid in soil, dust, or water was identified. In a study on oxalic acid and vehicle emissions, Huang and Yu (2007) summarized various aerosol oxalic acid concentrations from different countries, where oxalic acid was detected in PM0.7-2.5 or total suspended particles (TSP). Aerosol oxalic acid concentrations ranged from <0.004 (Germany) to 0.78 μg/ m3 (West Los Angeles, U.S.).
On the basis of the physical-chemical properties (high water solubility) and uses of oxalic acid (cleaning products available to consumers), releases to water may be a source of exposure. However, potential concentrations in water are likely to be low as the substance shows ready biodegradability in water (ECHA 2017).
On the basis of the available information on concentrations of oxalic acid in environmental media, indirect exposure to oxalic acid from environmental media for the general population in Canada is expected to be minimal.
No food additive or food flavouring uses of oxalic acid have been identified. However, the substance is reported to occur naturally in a variety of foods, such as nuts, fruits, vegetables, grains, and legumes (Tang et al. 2008), with higher levels found in seeds and leafy plants related to spinach and rhubarb (Holmes and Kennedy 2000). Under physiological conditions in plants and animals, oxalic acid exists in its anionic form, oxalate. Oxalate content in foods has been reported to vary widely even within the same foods (Holmes and Kennedy 2000) as it is influenced by factors such as plant variety, plant development, season, and growth conditions (Libert and Franceschi 1987, as cited in Holmes and Kennedy 2000). In a report on oxalate intake, Taylor and Curhan (2007) conducted a prospective study of three large cohorts: the Health Professionals Follow-up Study (HPFS, n = 45 985 men) and the Nurses’ Health Studies (NHS) I (n= 92 872) and II (n=101 824). In this study, on the basis of food frequency questionnaires administered every 4 years during the combined 44-year follow-up, mean oxalate intakes in men (HPFS), older women (NHS I), and younger women (NHS II) were estimated at 214, 185, and 183 mg/day, respectively.
In Canada, oxalic acid has been identified as an ingredient used in the manufacture of some food packaging materials (e.g., inks applied to the exterior surface of food packaging and the interior coatings or liners of cans). It may also be used as an ingredient in cleaners and laundry detergents for use in food processing establishments. The contribution of these uses is expected to be negligible compared to background exposure from the diet and levels of oxalic acid that can be produced endogenously (personal communications, emails from Food Directorate, Health Canada, to Existing Substances Risk Assessment Bureau, Health Canada, dated 2016; unreferenced).
Products available to consumers
Oxalic acid was identified in an all-purpose rust spray for use in removing household rust stains, and in an abrasive powder cleaner (SDS 2015a; SDS 2015b). It was also reported to be present in boat hull-cleaning solutions to remove waterline stains from the hulls of boats as well as boat bottom cleaners to remove scum line and rust stains (SDS 2014a; SDS 2014b; SDS 2015c).
The highest estimated dermal exposure to oxalic acid from products available to consumers was from the use of the boat hull-cleaning solution. The dermal exposure from this use was estimated assuming the entire surface area of one hand would be exposed to the product (applying the solution with a sponge/cloth). Dermal exposure from this use was estimated to be 1.09 mg/kg bw, assuming a concentration of 8% in the product (SDS 2014a). Inhalation exposure from this use was not considered as the product would typically be applied outdoors. Inhalation exposure to oxalic acid from cleaning a surface sprayed with an all-purpose rust cleaner (SDS 2015a) was estimated using ConsExpo Web (ConsExpo Web 2016). Assuming this product contained oxalic acid at 10% (SDS 2015a), inhalation exposure was estimated to be 0.033 mg/m3, which is equivalent to a systemic exposure of 0.00028 mg/kg bw/day. Details on the calculations of these exposure scenarios can be found in Appendix A.
Use of a structurally similar compound, oxalic acid dihydrate (CAS RN 6153-56-6), may result in potential exposure to oxalic acid. The dihydrate was identified to be used in cleaning products available to consumers, including deck cleaners, polishes, and toilet bowl cleaners. Exposure to oxalic acid from the use of products containing oxalic acid dihydrate would be lower or approximately equivalent to exposure from the use of products containing oxalic acid described above.
6.4.2 Health effects assessment
Repeated dose toxicity
In a chronic oral study, male and female Osborne-Mendel rats were administered oxalic acid in their diet at 0%, 0.1%, 0.5%, 0.8% or 1.2% for two years. No significant adverse effects were observed at the highest dose tested (Fitzhugh and Nelson 1947).
Adverse effects of oxalic acid on humans were reported, such as the formation of renal oxalate stones in individuals consuming a diet high of oxalate-rich foods, such as spinach, rhubarb, or parsley, combined with a deficiency in calcium-containing dairy foods (Hodgkinson and Zarembski 1968).
Genotoxicity
Oxalic acid was negative in Ames tests (Rossman et al. 1991) and in a chromosome aberration assay in Chinese hamster lung fibroblasts (V79) (Ishidate et al. 1984). No in vivo genotoxicity studies were identified.
Reproductive/developmental toxicity
The US National Toxicology Program conducted a study of reproduction and fertility in mice administered oxalic acid (NTP 1985). Male and female mice were randomly paired and exposed to oxalic acid in drinking water at 0.0%, 0.05%, 0.1% or 0.2% (approximately 100, 200 or 400 mg/kg bw/day). The pairs were housed together for 98 days, followed by a 21-day segregation period to allow for delivery of the final litters. In the 0.05% and 0.1% dose groups, no significant (p > 0.05) effect with respect to the number of live pups per litter, sex ratio and average live pup weight were observed. In the group exposed to 0.2% oxalic acid in drinking water, a significant reduction (p < 0.05) in number of litters per fertile pair and decreased average pup weight (adjusted for litter size) were observed.
The reproductive performance of the control and high-dose offspring was further evaluated. Group A consisted of 20 pairs of control males and females, and Group B consisted of 20 pairs of high-dose (0.2%) group males and females. After a 7-day cohabitation period, the pairs were separated and the females were allowed to deliver their litters. The total number of live pups and of live female pups delivered by the second-generation breeding pairs was significantly lower (p < 0.05) than that of the corresponding control group.
The prostate gland of adult animals administered 0.2% oxalic acid was smaller as evidenced by significantly reduced absolute and adjusted weights at necropsy. For second-generation mice, adjusted kidney weight for female mice and absolute kidney weight for male mice were significantly increased (p < 0.05).
Sperm morphology and vaginal cytology evaluation studies indicated that prolonged oxalic acid treatment may interfere with the relative frequency of estrus as evidenced by the data from first-generation mice. The incidence of abnormal sperm was increased in second-generation mice receiving 0.2% oxalic acid in drinking water. Since the increase in the percentage of abnormal sperm was observed in both treated second-generation animals and adult mice, it is possible that oxalic acid interferes with spermiogenesis. The NTP concluded that oxalic acid administration in drinking water at up to the 0.1% dose level does not affect the fertility in adult or second-generation CD-1 mice (NTP 1985). Therefore, a NOAEL of 0.1% oxalic acid in drinking water (or 200 mg/kg bw/day) was identified on the basis of the occurrence of reproductive effects at 0.2% oxalic acid.
In an oral developmental toxicity study, female rabbits were exposed to oxalic acid, via gavage, at 0, 2.5, 12, 54 and 250 mg/kg bw/day on GD 6 to 18. No developmental effects were observed at the highest dose tested (ECHA c2007-2016).
6.4.3 Characterization of risk to human health
Exposure of the general population to oxalic acid is expected to occur mainly from food and from the use of a boat cleaner and household rust cleaning spray. The predominant source of long-term exposure to oxalic acid for the general population is expected to be through the diet, as a naturally occurring substance in food. On the basis of the available data indicating the wide range of levels of oxalic acid in different foods, and within the same foods, the derivation of margins of exposure from its natural occurrence in foods was not considered to be meaningful.
A NOAEL of 200 mg/kg bw/day identified in a reproductive effects study on mice was identified as the most relevant endpoint for characterization of the human health risk from intermittent exposure to oxalic acid in products available to consumers.
Table 6‑1 provides the relevant estimates of exposure and critical effect levels for oxalic acid, as well as the resultant MOEs.
| Exposure scenario | Exposure estimate | Critical level | Critical effect | MOE |
|---|---|---|---|---|
| Intermittent dermal exposure from boat cleaner | 1.09 mg/kg bw (per application) | NOAEL = 200 mg/kg bw/day (oral administration in drinking water, in a reproduction and fertility study) | Reduction of total number of live pups and number of live female pups, increased abnormal sperm in both exposed generations of mice at the next dose (400 mg/kg bw/day) | 183a |
| Intermittent inhalation exposure from all-purpose rust spray | 0.00028 mg/kg bw/day (estimated concentration converted into systemic exposure) | NOAEL = 200 mg/kg bw/day (oral administration in drinking water, in a reproduction and fertility study) | Reduction of total number of live pups and number of live female pups, increased abnormal sperm in both exposed generations of mice at the next dose (400 mg/kg bw/day) | 714 285 |
aAssuming dermal absorption is equivalent to oral bioavailability
The risk characterization for oxalic acid is considered conservative; the duration of the reproductive and fertility study was much longer than the anticipated duration of the intermittent exposures from the use of the boat cleaner and all-purpose rust spray reported to contain oxalic acid. Additionally, conservative default values, such as 100% dermal absorption, and use of maximum concentrations reported in safety data sheets were used in estimating exposures.
Comparison of the NOAEL of 200 mg/kg bw/day based on reproductive effects in mice at the next dose tested to the estimates of exposure from using a boat cleaner (1.09 mg/kg bw) and from using an all-purpose rust spray (0.00028 mg/kg bw/day) resulted in MOEs of approximately 183 and >710 000, respectively. These margins are considered adequate to address uncertainties in the health effects and exposure databases.
6.4.4 Uncertainties in evaluation of risk to human health
The key sources of uncertainty associated with the risk characterization of oxalic acid are presented in Table 6‑2 below.
| Key source of uncertainty | Impact |
|---|---|
| Assumptions of 100% dermal absorption when using an oral endpoint for characterizing the risk from dermal exposure to oxalic acid from use in boat cleaners | + |
| Limited hazard database for oxalic acid, lack of short-term dermal and inhalation studies. | +/- |
+ = uncertainty with potential to cause over-estimation of exposure/risk; +/- = unknown potential to cause over or under estimation of risk.
7. Conclusion
Considering all available lines of evidence presented in this screening assessment, there is low risk of harm to the environment from propionic acid, n-butyric acid, nonanoic acid, and oxalic acid. It is concluded that propionic acid, n-butyric acid, nonanoic acid, and oxalic acid do not meet the criteria under paragraphs 64(a) or (b) of CEPA as they are not entering the environment in a quantity or concentration or under conditions that have or may have an immediate or long-term harmful effect on the environment or its biological diversity or that constitute or may constitute a danger to the environment on which life depends.
On the basis of the information presented in this screening assessment, it is concluded that propionic acid, n-butyric acid, nonanoic acid, and oxalic acid do not meet the criteria under paragraph 64(c) of CEPA as they are not entering the environment in a quantity or concentration or under conditions that constitute or may constitute a danger in Canada to human life or health.
Therefore, it is concluded that propionic acid, n-butyric acid, nonanoic acid, and oxalic acid do not meet any of the criteria set out in section 64 of CEPA.
References
Bergman EN. 1990. Energy contributions of volatile fatty acids from the gastrointestinal tract in various species. Physiol Rev. 70(2):567-590.
Burdock GA. 2009. Fenaroli’s handbook of flavour ingredients. 6th ed. Boca Raton (FL): CRC Press
Canada. 1999. Canadian Environmental Protection Act, 1999. S.C. 1999, c. 33. Canada Gazette Part III, vol. 22, no. 3.
Canada, Dept. of the Environment. 2012. Canadian Environmental Protection Act, 1999: Notice with respect to certain substances on the Domestic Substances List [PDF, 1.98 MB]. Canada Gazette, Part I, vol. 146, no. 48, Supplement.
ChemIDplus [database]. 1993-a. Bethesda (MD): US National Library of Medicine. Search results for CAS RN [79-09-4]. [accessed 2016 Sept 15].
ChemIDplus [database]. 1993-b. Bethesda (MD): US National Library of Medicine. Search results for CAS RN [107-92-6]. [accessed 2016 Sept 15].
ChemIDplus [database]. 1993-c. Bethesda (MD): US National Library of Medicine. Search results for CAS RN [112-05-0]. [accessed 2016 Sept 15].
ChemIDplus [database]. 1993-d. Bethesda (MD): US National Library of Medicine. Search results for CAS RN [144-62-7]. [accessed 2016 Sept 15].
ChemView [database]. 2013-a. Washington (DC): US Environmental Protection Agency. Search results for CAS RN [79-09-4]. [accessed 2016 December 01].
ChemView [database]. 2013-b. Washington (DC): US Environmental Protection Agency. Search results for CAS RN [107-92-6]. [accessed 2016 December 01].
ChemView [database]. 2013-c. Washington (DC): US Environmental Protection Agency. Search results for CAS RN [144-62-7]. [accessed 2016 December 01].
[ConsExpo Web] Consumer Exposure Model. 2016. Web version. Bilthoven (NL): Rijksinstituut voor Volksgezondheid en Milieu [National Institute for Public Health and the Environment].
David RM, Tyler TR, Ouellette T, Faber WD, Banton MI. 2001. Evaluation of subchronic toxicity of n-butyl acetate vapor. Food Chem Toxicol. 39:877-886.
[ECCC] Environment Canada and Climate Change. 2013. DSL Inventory Update data collected under the Canadian Environmental Protection Act, 1999, section 71: Notice with respect to certain substances on the Domestic Substances List. Data prepared by: Environment Canada, Health Canada; Existing Substances Program.
[ECCC] Environment and Climate Change Canada. 2017. Data collected from a targeted information gathering initiative for assessments under the Chemicals Management Plan (Feb 2017). Data prepared by: ECCC, Health Canada; Existing Substances Program.
[ECCC] Environment and Climate Change Canada. 2016a. Science Approach Document: Ecological Risk Classification of Organic Substances.
[ECCC] Environment and Climate Change Canada. 2016b. Gatineau (QC): Data used to create substance-specific hazard and exposure profiles and assign risk classifications in the Ecological Risk Classification of organic substances. Gatineau (QC). Available from: substances@ec.gc.ca.
[ECCC] Environment and Climate Change Canada. 2017. Data submitted pursuant to the Canadian Environmental Protection Act, 1999, section 70. Data prepared by ECCC, Health Canada; Existing Substances Program.
[ECCC, HC] Environment and Climate Change Canada, Health Canada. [modified 2017 Mar 12]. Categorization. Ottawa (ON): Government of Canada. [accessed 2017 Feb 10].
[ECHA] European Chemicals Agency. c2007-2016. Registered substances database; search results for CAS RN [144-62-7]. Helsinki (FI): ECHA. [updated 2016 Jan 14; accessed 2016 Dec].
[ECHA] European Chemicals Agency, 2013. [PDF, 768 KB] Annex 1 Background document to the Opinion proposing harmonised classification and labelling at Community level of nonanoic acid. EC number: 203-931-2; CAS number: 112-05-0. Committee for Risk Assessment (RAC).
[ECHA] European Chemicals Agency. 2017. Brief Profile: Oxalic acid. Helsinki (FI): ECHA [updated 2017 May 12; accessed 2017 May 15].
[EFSA] European Food Safety Authority. 2014. Scientific opinion on the re-evaluation of propionic acid (E 280), sodium propionate (E281), calcium propionate (E 282) and potassium propionate (E 283) as food additives. EFSA Journal. 12(7):3779.
[EMA] European Agency for the Evaluation of Medicinal Products. [PDF, 151 KB] 2003. Committee for veterinary medicinal products: oxalic acid summary report. London (UK): EMA. [updated 2003 Dec 1; accessed 2017 March 17].
[EPA] Environmental Protection Agency. 1998. EPA pesticide fact sheet. Pelargonic acid. Washington (DC): Environmental Protection Agency, Biopesticides and Pollution Prevention Division. Available from: NTIS, Springfield VA; 199823. Sponsored by the Office of Pesticide Program.
[EPI Suite] Estimation Program Interface Suite for Microsoft Windows [estimation model]. c2000-2012. Ver. 4.11. Washington (DC): US Environmental Protection Agency, Office of Pollution Prevention and Toxics; Syracuse (NY): Syracuse Research Corporation.
EU Food Flavourings Database [database]. [modified 2016]. Brussels (BE): European Commission Directorate-General for Health and Consumers. [accessed 2015].
EU Food Additives Database [database]. [modified 2018]. Brussels (BE): European Commission Directorate-General for Health and Consumers. [accessed 2015].
Fitzhugh OG, Nelson AA. 1947. The comparative chronic toxicities of fumaric, tartaric, oxalic and maleric acids. J Am Pharm Assoc. 36(7):217-219.
[FCC] Food Chemical Codex. 2016. United States Pharmacopeial Convention. Rockville (MD).
Haynes W, editor. 2016. CRC handbook of chemistry and physics, 97th ed. [database]. Search results for CAS RN [144-62-7]. Boca Raton (FL). accessed 2016 Sept 15.
Health Canada. [modified 2015 Dec 14]. Cosmetic Ingredient Hotlist: the list of prohibitied and restricted cosmetic ingredients. Ottawa (ON): Government of Canada. [accessed 2016 March 01].
Health Canada. [modified 2016 Dec 14] List of Permitted Preservatives (Lists of Permitted Food Additives). Ottawa (ON): Government of Canada. [accessed 2017 Feb 01].
Hodgkinson A, Zarembski PM. 1968. Oxalic acid metabolism in man: a review. Calcif Tissue Res. 2:115-132.
Holmes RP, Kennedy M. 2000. Estimation of the oxalate content of foods and daily oxalate intake. Kidney Int. 57:1662-1667.
[HSDB] Hazardous Substances Data Bank [database]. 1983-a . search results for CAS RN [144-62-7]. Bethesda (MD): National Library of medicine (US). [updated 2005 Jun 24; accessed 2016 December 01].
[HSDB] Hazardous Substances Data Bank [database]. 1983-b. search results for CAS RN [112-05-0]. Bethesda (MD): National Library of medicine (US). [updated 2008 Oct 07; accessed 2016 December 01].
[HSDB] Hazardous Substances Data Bank [database]. 1983-c. search results for CAS RN [107-92-6]. Bethesda (MD): National Library of medicine (US). [updated 2006 Dec 20; accessed 2016 December 01].
Huang XF, Yu JZ. 2007. Is vehicle exhaust a significant primary source of oxalic acid in ambient aerosols? Geophys Res Lett. 34:1-5.
Ishidate M Jr, Sofuni T, Yoshikawa K, Hayashi M, Nohmi T, Sawada M, Matsuoka A. 1984. Primary mutagenicity screening of food additives. Food Chem Toxicol. 22(8):623-636.
Kawamura K, Kaplan IR. 1987. Motor exhaust emissions as a primary source of dicarboxylic acids in Los Angeles ambient air. Environ Sci Technol. 21:105-110.
[LNHPD] Licensed Natural Health Products Database [database]. [modified 2018 Feb 6]. Ottawa (ON): Health Canada. [accessed 2017 Jan 15].
[MSDS] Material Safety Data Sheet. [PDF, 18 KB] 2011. CleanCide. Kirkwood (MO): Wexford Labs Inc. [accessed 2016 Dec 15].
McGinn SM, Janzen HH, Coates T. 2003. Atmospheric ammonia, volatile fatty acids, and other odorants near beef feedlots. J Environ Qual. 32:1173-1182.
[NCI] National Chemical Inventories Global [database]. 2015. Columbus (OH): American Chemical Society, Chemical Abstracts Service.
Nelson BK, Brightwell WS, Robertson SK, Kahn A, Krieg EF Jr, Massari VJ. 1989a. Behavioral teratology investigation of 1-butanol in rats. Neurotoxicol Teratol. 11(3):313-315.
Nelson BK, Brightwell WS, Kahn A, Burg JR, Goad PT. 1989b. Lack of selective developmental toxicity of three butanol isomers administered by inhalation to rats. Fundam Appl Toxicol. 12(3):469-479.
[NHPID] Natural Health Products Ingredients Database [database]. [modified 2018 October 18 ]. Ottawa (ON): Health Canada. [accessed 2017 Jan 15].
[NRC] National Research Council of Canada. 2011. Data gathering on chemicals released to indoor air of residences from building materials and furnishings. NRC-Institute for Research in Construction Report no. B3332.2. Prepared for Health Canada, Ottawa (ON).
[NTP] US National Toxicology Program. 1985. Oxalic acid: reproduction and fertility assessment in CD-1 mice when administered in drinking water. National Toxicology Program. National Institute of Environmental Health Science. NTP-85-322.
[OECD] Organisation for Economic Co-operation and Development. 2003. SIDS initial assessment report: n-butyric acid. CAS No. 107-92-6. [PDF, 150 KB] SIDS Initial Assessment Meeting 16; May 27-30, 2003; Helsinki, Finland.
[OECD] Organisation for Economic Co-operation and Development. 2007. SIDS initial assessment report: propionic acid. CAS No. 79-09-4. [PDF, 170 KB] SIDS Initial Assessment Meeting 25; October 16-19, 2007; Helsinki, Finland.
Rogge WF, Mazurek MA, Hildemann LM, Cass GR. 1993. Quantification of urban organic aerosols at a molecular level: Identification, abundance and seasonal variation. Atmospheric Environment. 27(8):1309-1330
Rossman TG, Molina M, Meyer L, Boone P, Klein CB, Wang Z, Li F, Lin WC, Kinney PL. 1991. Performance of 133 compounds in the lambda prophage induction endpoint of the Microscreen assay and a comparison with S. typhimurium mutagenicity and rodent carcinogenicity assays. Mutat Res. 260(4):349-367.
[SDS] Safety Data Sheet. 2014a. Star brite Instant Hull Cleaner. Fort Lauderdale (FL): Star brite Inc. [accessed 2016 Dec 15].
[SDS] Safety Data Sheet. 2014b. Star brite Rust Stain Remover. For Lauderdale (FL): Star brite Inc. [accessed 2016 Dec 15].
[SDS] Safety Data Sheet. 2015a. Iron OUT (liquid). [PDF, 184 KB] Fort Wayne (IN): Iron Out dba Summit Brands. [accessed 2016 Dec 15]..
[SDS] Safety Data Sheet. 2015b. Bar Keepers Friend Cleanser & Polish. [PDF, 490 KB] Indianapolis (IN): SerVaas Laboratories Inc. [accessed 2016 Dec 15].
[SDS] Safety Data Sheet. 2015c. Boat Bottom Cleaner. Fort Lauderdale (FL): Star brite Inc. [accessed 2016 Dec 15].
[TDS] Technical Data Sheet. [PDF, 151 KB] 2011. Cleancide Kirkwood (MO): Wexford Labs Inc. [accessed 2016 Dec 15].
Tang M, Larson-Meyer DE, Liebman M. 2008. Effect of cinnamon and tumeric on urinary oxalate excretion, plasma lipids, and plasma glucose in healthy subjects. Am J Clin Nutr. 87:1262-1267.
Taylor EN, Curhan GC. 2007. Oxalate intake and the risk for nephrolithiasis. J Am Soc Nephrol. 18(7):2198-2204.
[US FDA] US Food and Drug Administration. 2016a. [revised as of 2016 Apr 1]. Code of Federal Regulations Title 21, Volume 3: Section 184.1081 Propionic acid. [online] [accessed 2016 Sept 15]
[US FDA] US Food and Drug Administration. 2016b. [revised as of 2016 Apr 1]. Code of Federal Regulations Title 21, Volume 3: Section 182.60 Synthetic flavoring substances and adjuvants. [online]
[US FDA] US Food and Drug Administration. 2016c. [revised as of 2016 Apr 1]. Code of Federal Regulations Title 21, Volume 3: Section 172.515 Synthetic flavoring substances and adjuvants. [online]
Versar. 1985. Methods of assessing exposure to chemical substances. Volume 7. Methods for assessing consumer exposure to chemical substances. Washington D.C.: U.S. Environmental Protection Agency. EPA 560/5-85-007. [WHO] World Health Organization. 1998. Evaluation of certain food additives and contaminants: forty-ninth meeting of the Joint FAO/WHO Expert Committee on Food Additives. WHO Technical Report Series. Geneva (CH): World Health Organization. 104 pages. Report No.: 884.
Appendices
Appendix A. Estimated exposures to oxalic acid
Exposures to oxalic acid from products available to consumers were estimated on the basis of the assumed weight, i.e., 70.9 kg (Health Canada 1998) and the use behaviours of an adult. Exposures were estimated using ConsExpo Web (ConsExpo Web 2016) or algorithms from the model (RIVM 2007). In the absence of dermal absorption data, dermal absorption was assumed to be 100%.
| Exposure scenario | Assumptions |
|---|---|
| Boat hull cleaner (dermal) | Surface film thickness: 0.00213 cm (VERSAR 1985) Hand surface area: 455 cm2 (Health Canada 1998) Product density: 1.0 g/cm3 Concentration of substance in product: 8% (SDS 2014a) |
| All-purpose rust spray (inhalation) | Model: ConsExpo Bathroom cleaning spray: spraying (RIVM 2007) Application duration: 1.5 min (RIVM 2007) Exposure duration: 25 min (RIVM 2007) Room volume: 10 m3 (RIVM 2007) Ventilation rate: 2.0 per hour (RIVM 2007) Concentration of substance in product: 10% (SDS 2015a) |
