Screening assessment thiols group
Official title: Screening assessment thiols group
Chemical abstracts service registry numbers
75-18-3, 150-60-7, 25103-58-6, 71159-90-5
Environment and Climate Change Canada
Health Canada
October 2018
Catalogue: En14-340/2018E-PDF
ISBN: 978-0-660-28182-7
Synopsis
Pursuant to section 68 or 74 of the Canadian Environmental Protection Act, 1999 (CEPA), the Minister of the Environment and the Minister of Health have conducted a screening assessment on four of six substances referred to collectively under the Chemicals Management Plan as the Thiols Group. These four substances were identified as priorities for assessment as they met categorization criteria under subsection 73(1) of CEPA or were considered a priority on the basis of other human health concerns. Two of the six substances were subsequently determined to be of low concern through other approaches, and decisions for these substances are provided in a separate screening assessmentFootnote 1 . Accordingly, this screening assessment addresses the four substances listed in the table below. The four substances addressed in this screening assessment will hereinafter be referred to as the Thiols Group. The Chemical Abstracts Service Registry Numbers (CAS RNFootnote 2 ), their Domestic Substances List (DSL) names and their common names are listed in the table below.
CAS RN |
DSL name |
Common name |
75-18-3a |
Thiobis-methane |
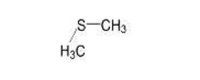
Dimethyl sulfide |
150-60-7 |
Bis(phenylmethyl) disulfide |
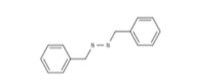
Benzyl disulfide |
25103-58-6b |
tert-Dodecanethiol |
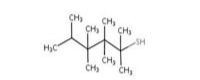
tert-Dodecyl mercaptan |
71159-90-5 |
alpha, alpha, 4-Trimethyl-3- cyclohexene-1-methanethiol |
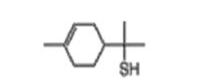
Grapefruit mercaptan |
a This substance was not identified under subsection 73(1) of CEPA but was included in this screening assessment as it was considered a priority on the basis of other human health concerns.
b This CAS RN is a UVCB (unknown or variable composition, complex reaction products, or biological materials).
According to information submitted under section 71 of CEPA, there were no reports of manufacture of tert-dodecyl mercaptan in 2008 or of dimethyl sulfide, benzyl disulfide, or grapefruit mercaptan in 2011 above the reporting threshold of 100 kg in Canada. Dimethyl sulfide and tert-dodecyl mercaptan were imported into Canada in 2011 and 2008 in the ranges of 10 000 to 100 000 kg and 100 000 to 1 000 000 kg, respectively. There were no reports of imports of benzyl disulfide and grapefruit mercaptan above the reporting threshold of 100 kg into Canada in 2011.
Dimethyl sulfide, benzyl disulfide, and grapefruit mercaptan may be used as food flavouring agents. tert-Dodecyl mercaptan has been identified as a component in the manufacture of some food packaging materials and may also be present as an impurity in paints and coatings. Dimethyl sulfide is being imported as an odorant in natural gas, and tert-dodecyl mercaptan is used in vinyl coverings, paper products, plastic and rubber materials, and metal materials.
The ecological risks of the substances in the Thiols Group were characterized using the ecological risk classification of organic substances (ERC), which is a risk-based approach that employs multiple metrics for both hazard and exposure with weighted consideration of multiple lines of evidence for determining risk classification. Hazard profiles are established based principally on metrics regarding mode of toxic action, chemical reactivity, food web-derived internal toxicity thresholds, bioavailability, and chemical and biological activity. Metrics considered in the exposure profiles include potential emission rate, overall persistence, and long-range transport potential. A risk matrix is used to assign a low, moderate or high level of potential concern for substances on the basis of their hazard and exposure profiles. The ERC identified the four substances in the Thiols Group as having low potential to cause ecological harm.
Considering all available lines of evidence presented in this screening assessment, there is low risk of harm to the environment from dimethyl sulfide, benzyl disulfide, tert-dodecyl mercaptan and grapefruit mercaptan. It is concluded that dimethyl sulfide, benzyl disulfide, tert-dodecyl mercaptan and grapefruit mercaptan do not meet the criteria under paragraphs 64(a) or (b) of CEPA as they are not entering the environment in a quantity or concentration or under conditions that have or may have an immediate or long-term harmful effect on the environment or its biological diversity or that constitute or may constitute a danger to the environment on which life depends.
The assessment of dimethyl sulfide by the Organization for Economic Cooperation and Development (OECD) did not identify effects of concern for human health. The Joint (Food and Agriculture Organization/World Health Organization (FAO/WHO)) Expert Committee on Food Additives (JECFA) determined that there are no safety concerns related to the use of dimethyl sulfide, benzyl disulfide and grapefruit mercaptan as food flavouring agents. The assessment of tert-dodecyl mercaptan by OECD noted effects on the liver in laboratory studies.
Dimethyl sulfide is imported as an odorant in natural gas and potential exposure to the general population from this use is expected to be limited. There is potential for dietary exposure to dimethyl sulfide, benzyl disulfide and grapefruit mercaptan from possible uses of these substances as food flavouring agents. While tert-dodecyl mercaptan is used in the manufacture of some food packaging materials such as coatings, the exposures from this use is considered to be negligible. Tert-dodecyl mercaptan may also be present as an impurity in some paints and coatings. There are no reports of benzyl disulfide and grapefruit mercaptan being identified in products available to consumers in Canada. On the basis of a consideration of the assessments by other organizations and their limited uses, the potential risk to human health is considered to be low for the substances in the Thiols Group. For tert-dodecyl mercaptan, based upon a comparison of levels Canadians may be exposed to from its presence as an impurity in some paints and coatings and levels associated with health effects in laboratory studies a concern for human health was not identified.
On the basis of the information presented in this screening assessment, it is concluded that dimethyl sulfide, benzyl disulfide, tert-dodecyl mercaptan and grapefruit mercaptan do not meet the criteria under paragraph 64(c) of CEPA as they are not entering the environment in a quantity or concentration or under conditions that constitute or may constitute a danger in Canada to human life or health.
Therefore, it is concluded that dimethyl sulfide, benzyl disulfide, tert-dodecyl mercaptan and grapefruit mercaptan do not meet any of the criteria set out in section 64 of CEPA.
1. Introduction
Pursuant to section 68 or 74 of the Canadian Environmental Protection Act, 1999 (CEPA) (Canada 1999), the Minister of the Environment and the Minister of Health have conducted a screening assessment on four of six substances, referred to collectively under the Chemicals Management Plan as the Thiols Group, to determine whether these four substances present or may present a risk to the environment or to human health. These four substances were identified as priorities for assessment as they met categorization criteria under subsection 73(1) of CEPA or were considered a priority on the basis of other human health concerns (ECCC, HC [modified 2017]).
The other two substances (CAS RNsFootnote 3 60-24-2, Ethanol, 2-mercapto-; 73984-93-7,1,3,4-Thiadiazole-2(3H)-thione, 5-(tert-dodecyldithio)-) were considered in the ecological risk classification of organic substances (ERC) and the Threshold of Toxicological Concern (TTC)-based Approach for Certain Substances science approach documents (ECCC 2016a; ECCC HC 2017), and were identified as being of low concern to both human health and the environment. As such, they are not further addressed in this report. Conclusions for these two substances are provided in the Substances Identified as Being of Low Concern based on the Ecological Risk Classification of Organic Substances and the Threshold of Toxicological Concern (TTC)-based Approach for Certain Substances Draft Screening Assessment (ECCC, HC 2017). The four substances addressed in this screening assessment will hereinafter be referred to as the Thiols Group.
Two substances in the Thiols Group currently being evaluated have been reviewed internationally through the Organization for Economic Cooperation and Development (OECD) Cooperative Chemicals Assessment Programme, and Screening Information Data Set (SIDS) and Initial Assessment Reports (SIARs) are available. These assessments undergo rigorous review (including peer-review) and endorsement by international governmental authorities. Health Canada and Environment and Climate Change Canada are active participants in these processes, and consider these assessments to be reliable. The dimethyl sulfide and the C8-C12 aliphatic thiols category OECD SIARs were used to inform the health effects characterization in this screening assessment (OECD 2006, OECD 2011). In addition, health effects for three of the substances in this group (dimethyl sulfide, benzyl disulfide, and grapefruit mercaptan) have been evaluated as food flavouring agents by the Joint (Food and Agriculture Organization/World Health Organization (FAO/WHO)) Expert Committee on Food Additives (JECFA), whose evaluations were also used to inform the health effects characterization in this screening assessment (WHO 2000).
The ecological risks of substances in the Thiols Group were characterized using the ERC approach (ECCC 2016a). The ERC describes the hazard of a substance using key metrics including mode of toxic action, chemical reactivity, food web-derived internal toxicity thresholds, bioavailability, and chemical and biological activity and considers the possible exposure of organisms in the aquatic and terrestrial environments on the basis of factors including potential emission rates, overall persistence and long-range transport potential in air. The various lines of evidence are combined to identify substances as warranting further evaluation of their potential to cause harm to the environment or as having a low likelihood of causing harm to the environment.
This screening assessment includes consideration of information on chemical properties, environmental fate, hazards, uses and exposures, including additional information submitted by stakeholders. Relevant data were identified up to November 2016. Empirical data from key studies as well as some results from models were used to reach conclusions. When available and relevant, information presented in assessments from other jurisdictions was considered.
This screening assessment was prepared by staff in the CEPA Risk Assessment Program at Health Canada and Environment and Climate Change Canada and incorporates input from other programs within these departments. The ecological portion of this assessment is based on the ERC document (published July 30, 2016), which was peer-reviewed and subject to a 60-day public comment period. Additionally, the draft of this screening assessment (published July 22, 2017) was subject to a 60-day public comment period. While external comments were taken into consideration, the final content and outcome of the screening assessment remain the responsibility of Environment and Climate Change Canada and Health Canada.
This screening assessment focuses on information critical to determining whether substances meet the criteria as set out in section 64 of CEPA, by examining scientific information and incorporating a weight-of-evidence approach and precautionFootnote 4 . This screening assessment presents the critical information and considerations on which the conclusions are based.
2. Identity of substances
The CAS RNs, Domestic Substances List (DSL) names and common names for the individual substances in the Thiols Group are presented in Table 2-1. A list of additional chemical names (e.g., trade names) is available from the National Chemical Inventories.
| CAS RN | DSL name | Common name | Chemical structure and molecular formula |
Molecular weight (g/mol) |
|---|---|---|---|---|
| 75-18-3 | Thiobis-methane | Dimenthyl sulfide |
C2H6S |
62.13 |
| 150-60-7 | Bis(phenyl,methyl) disulfide | Benzyl disulfide |
C14H14S2 |
246.39 |
| 25103-58-6a | tert-Dodecanethiol | tert-Dodecyl mercaptan |
C12H26S |
202.41 |
| 71159-90-5 | alpha,alpha, 4-trimethyl-3-cyclohexene-1 methanethiol | Grapefruit mercaptan |
C10H18S |
170.31 |
2.1 Selection of analogues
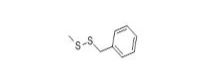
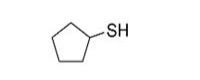
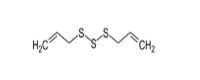
Potential metabolites, as outlined in JECFA’s safety evaluation of flavouring agents comprising substances in the Thiols Group (WHO 2000), were used as analogues to inform the human health assessment of benzyl disulfide and grapefruit mercaptan. Specifically, benzyl methyl disulfide (CAS RN 699-10-5), cyclopentanethiol (CAS RN 1679-07-8), and diallyl trisulfide (CAS RN 2050-87-5) have human health data which informed the assessment. Information on the identities of these analogues is presented in Table 2-2. Analogue identitiesa
CAS RN |
DSL or other name |
Chemical structure and molecular formula |
Molecular weight (g/mol) |
|---|---|---|---|
699-10-5 |
Benzyl methyl disulfide |
|
170.30 |
1679-07-8 |
Cyclopentanethiol |
|
102.20 |
2050-87-5 |
Diallyl trisulfide |
|
178.33 |
a As outlined in WHO (2000)
3. Physical and chemical properties
A summary of physical and chemical properties of the substances in the Thiols Group are presented in Table 3-1, with the range in values indicated for each property. When experimental information was limited or not available for a property, data from analogues were used for read across and/or (quantitative) structure-activity relationship ((Q)SAR) models were used to generate predicted values for the substance. Additional physical and chemical properties are presented in ECCC (2016b).
Property |
Dimethyl sulfidea |
Benzyl disulfideab |
tert-Dodecyl mercaptanc |
Grapefruit mercaptand |
Melting point (°C) |
-98.3 |
71.5 |
3.7 |
3.0 |
Boiling point (°C) |
37.3 |
376-384 |
215 |
295 |
Vapour pressure (Pa) |
66928 |
0.0036 |
22.80 |
14.13 |
Water solubility (mg/L) |
22000 |
0.75 |
0.43 |
5.49 |
Log Kow (dimensionless) |
0.92 |
5.29 |
5.85 |
4.74 |
Abbreviations: Kow, octanol–water partition coefficient;
a Chemidplus 2018
b Chemspider 2015: boiling point is experimentally determined; vapour pressure and water solubility are predicted by EPISuite
c Chemspider 2015: all values are predicted by EPISuite
d Chemspider 2015: boiling point is experimentally determined; melting point, vapour pressure, water solubility, Log Kow are predicted by EPISuite
4. Sources and uses
Dimethyl sulfide is naturally present in many types of food such as milk, eggs, beef, cheese, strawberries, broccoli, and cabbage (Kim and Kim 2014, Klein et al 2016). Benzyl disulfide is naturally present in a variety of foods and grapefruit mercaptan is found in grapefruit juice (WHO 2000). tert-Dodecyl mercaptan does not occur naturally in the environment.
All four of the substances in the Thiols Group have been included in surveys issued pursuant to a CEPA section 71 notice and Table 4-1 presents a summary of information reported on the total manufacture and total import quantities for 2008 or 2011 for tert-dodecyl mercaptan and dimethyl sulfide (Environment Canada 2009, 2014). Manufacturing and import of benzyl disulfide and grapefruit mercaptan were not reported under the 2011 CEPA section 71 survey (Environment Canada 2014).
Common name |
Total manufacturea (kg) |
Total importsa,b (kg) |
Reporting year |
Survey reference |
Dimethyl sulfide |
N/A |
10 000 – 100 000 |
2011 |
Environment Canada 2014 |
tert-Dodecyl mercaptan |
N/A |
100 000 – 1 000 000 |
2008 |
Environment Canada 2009 |
Abbreviations: N/A, Not Applicable
a Values reflect quantities reported in response to a survey conducted under section 71 of CEPA (Environment Canada 2014). See survey for specific inclusions and exclusions (Schedules 2 and 3).
b Import quantities are reported in ranges to mask Confidential Business Information.
With respect to Canadian uses, dimethyl sulfide is used as an odorant in natural gas while tert-dodecyl mercaptan is used as an intermediate, polymerization aid, solids separation agent, viscosity adjustor and process regulator (Environment Canada 2009,
2014). Dimethyl sulfide is also released during the kraft pulping process. Tert-Dodecyl mercaptan is identified as a component in the manufacture of some food packaging materials (personal communication, email from Food Directorate to ESRAB, July 2016) and is also used in the manufacture of other products including metal, paper, plastic and rubber materials (Environment Canada 2009). Tert-Dodecyl mercaptan can be found in products used by consumers including vinyl floor coverings and paper products, and may also be present as an impurity in certain do-it-yourself products (e.g., paints and coatings) (Environment Canada 2009).
None of these four substances are listed in the Drug Product Database or the Therapeutic Products Directorate's internal Non-Medicinal Ingredient Database as being present in prescription, non-prescription, or veterinary drugs in Canada (personal communication Therapeutic Products Directorate to ESRAB, June 2016). Dimethyl sulfide, benzyl disulfide, and grapefruit mercaptan are listed in the Natural Health Products Ingredients Database with a non-medicinal role for use as flavour enhancer in natural health products (NHPs), up to 0.25, 0.0012, and 0.00056 mg/kg- bw/day, respectively. However, none of these substances are listed in the Licensed Natural Health Products Database as being present in currently licensed NHPs (NHPID [modified 2018], LNHPD [modified 2018], personal communication, email from the Natural and Non-prescription Health Products Directorate to ESRAB, June 2016). Three of the four substances in the Thiols Group (i.e., dimethyl sulfide, benzyl disulfide, and grapefruit mercaptan) could possibly be used as food flavouring agents. One of the substances in this group, tert- dodecyl mercaptan, was identified as a component used in the manufacture of some food packaging materials (i.e. coatings) (personal communication, email from Food Directorate to ESRAB, July 2016). None of the substances in this group are approved for use as food additives in Canada (personal communication, email from Food Directorate to ESRAB, July 2016).
None of these substances were reported to be found in cosmetic products in Canada (personal communication Consumer Product Safety Directorate to ESRAB, July 2016). Dimethyl sulfide and benzyl disulfide are on the Pesticide Management Regulatory Agency (PMRA)’s formulants list; however, only dimethyl sulfide is present in registered products. Specifically, dimethyl sulfide is present as a fragrance in several products such as insect repellents and insecticides (personal communication, emails from PMRA to ESRAB, July 2016).
5. Potential to cause ecological harm
5.1 Characterization of ecological risk
The ecological risks of substances in the Thiols Group were characterized using the ecological risk classification of organic substances (ERC) (ECCC 2016a). The ERC is a risk-based approach that considers multiple metrics for both hazard and exposure on the basis of weighted considerations of multiple lines of evidence for determining risk classification. The various lines of evidence are combined to discriminate between substances of lower or higher potency and lower or higher potential for exposure in various media. This approach reduces the overall uncertainty with risk characterization compared to an approach that relies on a single metric in a single medium (e.g., LC50) for characterization. The following summarizes the approach, which is described in detail in ECCC (2016a).
Data on physical-chemical properties fate (chemical half-lives in various media and biota, partition coefficients, fish bioconcentration), acute fish ecotoxicity, and chemical import or manufacture volume in Canada were collected from scientific literature, from available empirical databases (e.g., OECD QSAR Toolbox), and from responses to surveys under section 71 of CEPA, or they were generated using selected QuantitativeStructure-Activity Relationship (QSAR) or mass-balance fate and bioaccumulation models. These data were used as inputs to other mass-balance models or to complete the substance hazard and exposure profiles.
Hazard profiles were established based principally on metrics regarding mode of toxic action, chemical reactivity, food web-derived internal toxicity thresholds, bioavailability, and chemical and biological activity. Exposure profiles were also composed of multiple metrics including, potential emission rate, overall persistence, and long-range transport potential. Hazard and exposure profiles were compared to decision criteria in order to classify the hazard and exposure potentials for each organic substance as low, moderate, or high. Additional rules were applied (e.g., classification consistency, margin of exposure) to refine the preliminary classifications of hazard or exposure.
A risk matrix was used to assign a low, moderate or high classification of potential risk for each substance on the basis of its hazard and exposure classifications. ERC classifications of potential risk were verified using a two-step approach. The first step adjusted the risk classification outcomes from moderate or high to low for substances which had a low estimated rate of emission to water after wastewater treatment, representing a low potential for exposure. The second step reviewed low risk potential classification outcomes using relatively conservative, local-scale (i.e., in the area immediately surrounding a point-source of discharge) risk scenarios, designed to be protective of the environment, to determine whether the classification of potential risk should be increased.
ERC uses a weighted approach to minimize the potential for both over and under classification of hazard and exposure and subsequent risk. The balanced approaches for dealing with uncertainties are described in greater detail in ECCC 2016a. The following describes two of the more substantial areas of uncertainty. Error with empirical or modeled acute toxicity values could result in changes in classification of hazard, particularly metrics relying on tissue residue values (i.e., mode of toxic action), many of which are predicted values from QSAR models. However, the impact of this error is mitigated by the fact that overestimation of median lethality will result in a conservative (protective) tissue residue used for critical body residue (CBR) analysis. Error with underestimation of acute toxicity will be mitigated through the use of other hazard metrics such as structural profiling of mode of action, reactivity and/or estrogen binding affinity. Changes or errors in chemical quantity could result in differences in classification of exposure as the exposure and risk classifications are highly sensitive to emission rate and use quantity. The ERC classifications thus reflect exposure and risk in Canada on the basis of what is believed to be the current use quantity, and may not reflect future trends.
Critical data and considerations used to develop the substance-specific profiles for the four substances in the Thiols Group and the hazard, exposure and risk classification results are presented in ECCC (2016b).
The hazard and exposure classifications for the four substances in the Thiols Group are summarized in Table 5-1.
|
Common name |
ERC hazard classification |
ERC exposure classification |
ERC risk classification |
Dimethyl sulfide |
low |
low |
low |
Benzyl disulfide |
moderate |
low |
low |
tert-Dodecyl mercaptan |
moderate |
low |
low |
Grapefruit mercaptan |
high |
low |
low |
According to information considered under ERC, grapefruit mercaptan was classified as low exposure potential. Grapefruit mercaptan was classified by ERC as having a high hazard on the basis of reactive mode of action and high potential to cause adverse effects in aquatic foodwebs given its bioaccumulation potential. In addition, structural alerts from the OECD toolbox identified grapefruit mercaptan as being a potential DNA and protein binder. This substance was initially classified as having a moderate potential for ecological risk, however, the risk classification was decreased to low potential for ecological risk following the adjustment of risk classification based on current use quantities (see section 7.1.1 of the ERC approach document, ECCC 2016a). The potential effects and how they may manifest in the environment were not further investigated due to the low exposure of this substance. On the basis of current use patterns, this substance is unlikely to result in concerns for the environment in Canada.
According to information considered under ERC, Benzyl disulfide and tert-dodecyl mercaptan were classified as having low exposure potentials. Benzyl disulfide and tert-dodecyl mercaptan were classified as having moderate hazard potential on the basis of reactive mode of toxic action and a moderate potential to cause adverse effects in aquatic foodwebs given their bioaccumulation potential. In addition, structural alerts from the OECD toolbox identified these substances as being potential protein binders. The potential effects and how they may manifest in the environment were not further investigated due to the low exposure of these substances. On the basis of current use patterns, these substances are unlikely to result in concerns for the environment in Canada.
On the basis of low hazard and low exposure classifications according to information considered under ERC, dimethyl sulfide was classified as having a low potential for ecological risk. It is therefore unlikely that this substance will result in concerns for the environment in Canada.
6. Potential to cause harm to human health
6.1 Exposure assessment
Environment media
No concentrations above detection limits of any of the substances in this group in indoor air, outdoor air, drinking water, or soil were measured in Canada. An ambient air quality monitoring program measured specific compounds in air samples collected at upwind and downwind locations relative to a landfill in Ontario, to examine any potential impact of compounds emitted to the air from the landfill. Dimethy sulfide was not present in detectable concentration for the upwind and downwind air samples (City of Hamilton 2009).
Some studies have suggested that dimethyl sulfide may be released from landfills, sewage plants, kraft pulp mills and composting facilities (City of Hamilton 2009, Kotowska et al. 2012, Kangas et al 1984, Drimal et al 2010, Goyer 1990, Catalan et al. 2009), and it has been detected in ppb levels in outdoor air in Korea (Susaya et al 2011). However, given that dimethyl sulfide was not detected in outdoor air in Canada, levels down wind of landfills were below levels of detection and given the limited uses (dimethyl sulfide as an odorant in natural gas, and tert-dodecyl mercaptan is imported in products), environmental releases and as a result environmental media exposure for these substances is expected to be limited. Thus, exposure from environmental media were not quantified.
Food
The Food Chemicals Codex (FCC) indicates that two of the substances in this group, dimethyl sulfide and benzyl disulphide, have the function of a flavouring agent (FCC USP 2016). Dimethyl sulphide, benzyl disulphide and grapefruit mercaptan are listed in Fenaroli’s Handbook of Flavor Ingredients (Burdock 2010). This same reference also reports that dimethyl sulfide occurs naturally in a variety of foods and grapefruit mercaptan naturally is present in grapefruit juice (Burdock 2010). The predominant source of dietary exposure to dimethyl sulfide is reasonably expected to result from its natural occurrence in foods. The European Union also permits the use of dimethyl sulfide, benzyl disulfide, and grapefruit mercaptan as flavouring agents in foods (European Union Food Flavourings Database). No information is available concerning the potential use of these three thiols as food flavourings in Canada (personal communication from Food Directorate, Health Canada to Existing Substances Risk Assessment Bureau, Health Canada, July 2016).
The Joint FAO/WHO Expert Committee on Food Additives (JECFA) evaluated a group of flavouring agents that included thiols at its 53rd meeting (WHO 2000). Estimated intakes of thiols considered by the JECFA committee and included in the current assessment are summarized in Table 6-1 below.
Substance (CAS RN) |
Intakes (ug/kg-bw/day) |
Dimethyl sulfide (75-18-3) |
Europe: 10 USA: 9 |
Benzyl disulfide (150-60-7) |
Europe: 0.0002 USA : 0.002 |
Grapefruit mercaptan (71159-90-5) |
Europe: 0.01 USA: 0.01 |
One of the substances in this group, tert-dodecyl mercaptan, has also been identified as a component used in the manufacture of food packaging. However, dietary exposure from this source is expected to be negligible (personal communication, email from Food Directorate to ESRAB, August 2016).
Products available to consumers
As described in the uses section dimethyl sulfide is used as an odorant in natural gas
(Environment Canada 2014) as it has a low odor threshold (as low as 2 ppb, OCED 2011). Exposure from this source is not expected to be significant for the general population of Canada due to the unpleasant odor of this substance as well as the limited potential for exposure (e.g. accidental gas leakage).
Regarding tert-dodecyl mercaptan, the survey under section 71 of CEPA indicated potential presence, as an impurity, in paints and coatings with concentrations in the final products being less than 0.1 % (Environment Canada 2014). General population exposure was estimated, using ConsExpo version 4.1 (ConsExpo 2006, see appendix A), from use of a waterborne paint containing 0.1 % of tert-dodecyl mercaptan (inhalation mean event concentration: 0.67 mg/m3, dermal incidental exposure: 51 ug/kg-bw). It should be noted that complete dermal absorption was assumed; however, on the basis of flux (Jmax) derived from the physical/chemical properties of tert-dodecyl mercaptan (Kroes et al 2007) dermal absorption is considered to be significantly lower. Exposure from potential presence of tert-dodecyl mercaptan in vinyl coverings was not modelled as this exposure was not expected to be significant (Environment Canada 2014).
For benzyl disulfide and grapefruit mercaptan, no uses in products available to consumers were identified in Canada according to the section 71 survey (Environment Canada 2011).
6.2 Health effects assessment
Dimethyl sulfide
The health effects information for this substance is on the basis of the OECD SIAR for dimethyl sulfide (OECD 2006). A literature review from the year prior to the OECD SIAR to July 2016 did not identify any new studies that would impact the hazard and risk characterization of this substance.
OECD (2006) did not identify any well-conducted studies regarding the toxicokinetics, metabolism, and distribution of dimethyl sulfide. A limited study did identify, following a single acute exposure to dimethyl sulfide vapours, that dimethyl sulfide was widely distributed in all tissues (OECD 2006). Alkyl sulfides are thought to be oxidized to sulfoxides and subsequently to sulfones in mammals (Terazawa et al 1991, as cited in OECD 2006).
Inhalation, oral, and dermal acute toxicity studies were available, but the data was limited for studies of oral and dermal routes. As reported in OECD 2006, rats were exposed for 4 hours to dimethyl sulfide vapor at 0, 800, 3000, 6000, 12000, 24000, 36000, 39000, 42000, 45000, or 48000 ppm (approximately 0, 2.03, 7.61, 15.2, 30.4, 60.9, 91.3, 98.9, 106, 114, or 122 mg/L), and animals were observed for 14 days post- exposure. Mortality was observed in some animals exposed to concentrations equal to or greater than 36000 ppm, and the LC50 was calculated to be 40250 ppm (102 mg/L) mammals (Tansy et al 1981, as cited in OECD 2006). In another acute inhalation toxicity study, mice were dosed with 6.8, 11.6, 23.6, 34.0, or 50.6% (68000, 116000, 236000, 340000, or 506000 ppm), and all animals died within approximately 8 minutes at 6.8% (approximately 173 mg/L) mammals (Terazawa et al 1991, as cited in OECD 2006). Limited acute oral toxicity studies showed that no deaths or other effects occurred in mice treated with 5000 mg/kg-bw, or in pregnant rats dosed with 1000 mg/kg-bw/day for 14 days (Elf Atochem 1995, WIL Research Laboratories 2004, as cited in OCED 2006). A limited dermal exposure study reported an LD50 of >5 g/kg-bw (>5000 mg/kg-bw) in rabbits however no exposure time was noted (Opdyke 1979, as cited in OCED 2006).
As summarized in OECD (2006) in vitro (bacterial reverse mutation assay and DNA damage and repair assay) and in vivo (mouse micronucleus assay) studies show negative results (Nakamura et al 1990, Microbiological Associates Inc. 1995a, Microbiological Associates 1995b as cited in OECD 2006). No carcinogenicity studies were identified.
In an oral (gavage) repeated-dose study, male and female rats were administered 2.5, 25, and 250 mg/kg-bw/day of dimethyl sulfide for 2, 6, or 14 weeks (Butterworth et al. 1975 as cited in WHO 2000 and OECD 2006)Footnote 5 . Increased relative brain weights and decreased absolute heart weights were observed in females in the high-dose group at 2 and 6 weeks respectively. At 14 weeks, male rats had significantly higher absolute small intestine weights (at all dose levels) when compared to control (relative intestine weights were increased at 25 and 250 mg/kg-bw/day). Sex-specific differences in relative thyroid weights were also observed in males and females dosed for 14 weeks at 250 mg/kg-bw/day (decreases in relative and absolute thyroid weights in females and increases in relative thyroid weights in males). No treatment-related histopathological effects were observed in the organs mentioned above. No abnormalities were observed in the testes and ovaries and no treatment-related effects were observed for body weight, food consumption, water consumption, hematology, and blood chemistry. A no-observed-adverse-effect-level (NOAEL), highest dose tested, of 250 mg/kg-bw/day was derived (OECD 2006).
In an oral (gavage) developmental study, pregnant rats (25/group) were dosed 100, 500, or 1000 mg/kg-bw/day of dimethyl sulfide on gestational days (GD) 6 to 19 (WIL Research Laboratories, LLC, 2004 as cited in OECD 2006). No treatment-related effects on maternal body weight, food consumption, intrauterine growth, fetal numbers, fetal weight, and survival were observed. Additionally, no fetal malformation or developmental variations were observed in any of the treatment groups. A NOAEL, highest dose tested, of 1000 mg/kg-bw/day was derived (OECD 2006).
Benzyl disulfide and grapefruit mercaptan
The JECFA has evaluated the safety of a group of flavouring agents, including dimethyl sulfide, benzyl disulfide and grapefruit mercaptan (WHO 2000). A literature review from the year prior to the JECFA safety evaluation to July 2016 identified two additional studies published by the European Food Safety Authority (EFSA) that would have an impact on the hazard and risk characterization of these substances (EFSA 2009, 2014).
The JECFA Committee concluded that flavouring agents comprising thiols, including benzyl disulfide and grapefruit mercaptan were of no safety concern at the estimated levels of intake.
Ames tests, with and without metabolic activation, with administered benzyl disulfide have shown negative results at concentrations of 10, 100, and 300 ug/plate and positive results at a higher concentration of 600 ug/plate (Gao Y et al 1993). However, in contrast, another Ames test (with and without activation) at 6-fold higher concentration of benzyl disulfide (3600 ug/plate) was negative (WHO 2000, EFSA 2009).
WHO (2000) classified benzyl disulfide as a class III substance - “substances that have structural features that permit no strong initial presumption of safety, or may even suggest significant toxicity”, and grapefruit mercaptan as a class II substance - “substances that have structural features that are less innocuous than those of substances in class I, but are not suggestive of toxicity. Substances in this class may contain reactive functional groups”.
In terms of assessing the health effects of these two substances, the JECFA considered benzyl methyl disulfide (CAS RN 699-10-5) and cyclopentanethiol (CAS RN 1679-07-8) as related substances for the safety evaluation of benzyl disulfide and grapefruit mercaptan, respectively (WHO 2000). The assessment of these related substances are on the basis of these substances being potential metabolites of benzyl disulfide andgrapefruit mercaptan. The no-observed-effect-levels (NOEL) used for JECFA’s safety assessment of benzyl disulfide are 1.2 mg/kg-bw/day in a 90-day benzyl methyl disulfide repeated-dose study in rats (only dose tested). The NOEL used for JECFA’s safety assessment of grapefruit mercaptan is 0.56 mg/kg-bw/day in a 90-day cyclopentanethiol repeated-dose study (only dose tested).
In external reviews of JECFA decisions by the European Food Safety Authority (EFSA), this organization agreed with JECFA’s conclusion of “no safety concern” for benzyl disulfide (EFSA 2009). With respect to grapefruit mercaptan EFSA (2011ab) concluded that this substance could not be adequately evaluated through the WHO (2000) procedure as related monothiols have concerns for genotoxicity. This opinion was updated in 2014, after genotoxicity data of a related substance (2-methyl-4-oxopentane-2-thiol) showed negative genotoxic effects, thus addressing EFSA’s concern of potential genotoxicity for grapefruit mercaptan (McGarry 2012 as cited in EFSA 2014). However, the EFSA (2014) did not finalize its evaluation for this substance as the Panel was of the opinion that an adequate NOAEL did not exist for grapefruit mercaptan or from what it considered to be a structurally related analogue.
tert-Dodecyl mercaptan
The health effects information for this substance is on the basis of OECD SIARS for tert- dodecyl mercaptan (OECD 2011). A literature review from the year prior to the OECD SIARs to July 2016 did not identify any studies that would impact the hazard and risk characterization of this substance.
The OECD (2011) did not identify any well conducted studies regarding the toxicokinetics, metabolism, and distribution of tert-dodecyl mercaptan. tert-Dodecyl mercaptan is an irritant to the skin and eyes and may be a dermal sensitizer (OECD
2011).
Dermal, oral, and inhalation acute toxicity studies were available. As reported in OECD (2011), New Zealand white rabbits were administered with 6.83, 10.25, 15.28 and 23.07 g/kg-bw of tert-dodecyl mercaptan by the dermal routeFootnote 6 . All eight rabbits of the 15.28 g/kg-bw dose group died three to four days post-dosing and the LD50 was modelled to be 12.6 g/kg-bw (12 600 mg/kg-bw) (OECD 2011). In an oral acute toxicity study, Sprague-Dawley rats were dosed with 4.56, 6.84, 10.25, 15.38 g/kg-bw. All eight rats in the two highest dose groups died, and the modelled LD50 was 6.8 g/kg-bw (6800 mg/kg- bw) (OECD 2011). Two other studies in rats reported dermal LD50’s of 4380 mg/kg-bw and > 5000 mg/kg-bw (OECD 2011). A limited inhalation acute study (1 dose tested, 12 mg/L) in Sprague-Dawley rats (5 males and 5 females) reported no deaths (OECD 2011). Other limited inhalation acute studies are reported in OECD (2011).
Sub-chronic inhalation studies in rats, mice and dogs are available for tert-dodecyl mercaptan. Charles River CD rats were exposed to 26 ppm (220 mg/m3) and 98 ppm (810 mg/m3, saturated vapour) for six hours a day, 5 days a week for 4 weeks. High-dose males showed statistically significant decreases in body weight and food consumption, and increases in creatinine production. Exposure-related increases in liver weights were also observed; however, no microscopic changes were noted. At both doses, male rats also showed mild renal tubular degeneration and granular cysts, while high-dose females exhibited hydronephrosis (OECD 2011). In CD-1 mice, tert-dodecyl mercaptan was administered at doses of 25 ppm (210 mg/m3) and 109 ppm (900 mg/m3) for six hours a day, five days a week for four weeks. One male and one female mouse died in the high-dose group, and surviving animals at this dose level showed signs of alopecia and peeling skin, and exhibited statistically significant reductions of erythrocyte counts, hematocrit and increases in other red blood parameters. In addition, female mice at the high dose showed elevations of alanine aminotransferase, blood urea nitrogen, and alkaline phosphatase values while also showing depressed ovary weights. Other effects in high-dose mice were also observed (OECD 2011). At the low dose, females showed elevated blood glucose levels, increased liver weights and in both sexes, liver enlargement and discoloration and hepatocellular hypertrophy was also observed at the low dose (in addition to the high dose) (OECD 2011). In a four week inhalation dog study where animals were dosed with 25 ppm (210 mg/m3) and 109 ppm (900 mg/m3) for six hours a day, five days a week for four weeks, increases in alanine aminotransferase and blood urea nitrogen were reported at the high dose.
Dose-related increases in liver weights with hepatocellular hypertrophy, at the high dose, were also observed in both sexes (OECD 2011). Given the similarities, in terms of effects seen in the liver and kidney at the lowest dose in all three studies, lowest- observed-adverse-effect-concentrations (LOAECs) of 25-26 ppm (210-220 mg/m3) were determined (OECD 2011).
In vitro genotoxicity studies showed predominantly negative results (Ames, mouse lymphoma, sister chromatid) and one equivocal result (chromosome aberration test) (OECD 2011). No in vivo genotoxicity or carcinogenicity studies were identified.
Inhalation developmental studies, in rats and mice, were also conducted. Pregnant COBS-CD rats were dosed 22.6 ppm (190 mg/m3) or 88.6 ppm (730 mg/m3) from GD 6 to 19 for six hours a day. Caesarian sections were conducted on day 20. No treatment- related effects were observed in fetuses or mothers; however, maternal body weight gains appeared to have a dose-related decrease (OECD 2011). One rat died at the high-dose level; however, the cause of death was not determined. The OECD (2011) determined a no-observed-adverse-effects-concentration (NOAEC) of 88.6 ppm for both maternal and developmental endpoints. In pregnant mice (CD-1), tert-dodecyl mercaptan was administered at 22.6 ppm (190 mg/m3) and 88.6 ppm (730 mg/m3) from GD 6 to 16 (six hours a day). There were no statistically significant changes in body weights or fetal malformations. Other parameters (e.g., mean number of total implants and fetal body weight) were comparable to control values. No treatment-related fetal malformations were noted; however, one mouse died at the low-dose level, although the cause of death could not be determined. In 2011, the OECD stated “In rats and mice exposed via inhalation to tert-dodecyl mercaptan, no treatment-related maternal or developmental toxicity was observed at concentrations up to 88.6 ppm (730 mg/m3)”(OECD 2011).
6.3 Characterization of risk to human health
Regarding critical studies for hazard characterization; key studies summarized in OECD SIARs (OECD 2011) are used for risk characterization. Table 6-2 provides all relevant exposure and hazard values for the thiol present in this grouping, as well as resultant margin of exposure (MOE), for determination of risk.
| Substance | Exposure scenario[JCV1] | Intakes | Critical effect level | MOE |
|---|---|---|---|---|
| tert-Dodecyl mercaptan | Inhalation exposure from paints | 0.60 mg/m3 a | NOAEC: 730 mg/m3 |
1216 |
Abbreviations: NOAEL: No-observed-adverse-effect-concentration.
a As modelled by ConsExpo for general population exposure
b As reported by OECD 2011
These MOEs are considered adequate to account for uncertainties in the databases. The sources of exposure, for the general population of Canada, to dimethyl sulfide, benzyl disulfide, and grapefruit mercaptan are from uses of these substances as food flavouring agents.
With regards to benzyl disulfide and grapefruit mercaptan, on the basis of the available data on toxicity and metabolism as well as estimated low intakes derived for the US and EU populations, the JECFA committee concluded there to be no safety concerns with their use as food flavouring agents (WHO 2000). Furthermore, it has been reported that grapefruit mercaptan displays a grapefruit-like flavour at low concentration (0.004 ppm), while a 10-fold higher concentration will result in a burning tire smell (Young et al 2006). Because of such properties, the intakes of these flavouring agents are self-limiting. Given that exposures to these substances are considered to be negligible in Canada and other uses have not been identified, the risk to the general population for these substances is considered to be low.
For tert-dodecyl mercaptan, the general population may be exposed through the use of paints and coatings, and the primary route of exposure is inhalation. Acute inhalation studies did not demonstrate endpoints for use in assessing exposure to low airborne concentrations of tert-dodecyl mercaptan from painting activities. As noted in Table 6-2, a MOE of 1216 was obtained from comparing the estimate of exposure from inhaling tert-dodecyl mercaptan during painting with the absence of effects noted at the highest dose tested in an inhalation developmental toxicity study (OECD 2011). This MOE is considered adequate to account for potential uncertainties in the hazard and exposure databases.
The general population may also be exposed to tert-dodecyl mercaptan through the use of paints and coatings by the dermal route (0.22 ug/kg-bw/event). However, this exposure is not expected to be a concern due to the low concentrations in the products (impurity: < 0.1 %). Additionally, as mentioned in the exposure section, this substance has a low predicted dermal absorption value (10 %); therefore, the current modelled dermal exposure estimate is conservative. In addition, the potential risk to human health via exposure from food packaging applications is considered to be negligible due to low exposure.
6.4. Uncertainties in evaluation of risk to human health
The primary uncertainty in the estimation of dietary exposures is that Health Canada does not have data on the levels of these substances, if any that may be present in foods available in the Canadian marketplace today. However, the intake estimates considered by the JECFA committee in their evaluation of these flavouring agents are considered to be representative of exposures in Canada.
The current assessment considered the JECFA’s evaluation that these substances (dimethyl sulfide, benzyl disulfide and grapefruit mercaptan) are safe at the estimated levels of intake as food flavouring agents. JECFA’s evaluation was conducted with the assumption of low toxicity for potential metabolites of these substances, but the toxicity for metabolites of benzyl disulfide and grapefruit mercaptan is not known.
7. Conclusion
Considering all available lines of evidence presented in this screening assessment, there is low risk of harm to the environment from dimethyl sulfide, benzyl disulfide, tert-dodecyl mercaptan and grapefruit mercaptan. It is concluded that dimethyl sulfide, benzyl disulfide, tert-dodecyl mercaptan and grapefruit mercaptan do not meet the criteria under paragraphs 64(a) or (b) of CEPA as they are not entering the environment in a quantity or concentration or under conditions that have or may have an immediate or long-term harmful effect on the environment or its biological diversity or that constitute or may constitute a danger to the environment on which life depends.
On the basis of the information presented in this screening assessment, it is concluded that dimethyl sulfide, benzyl disulfide, tert-dodecyl mercaptan and grapefruit mercaptan do not meet the criteria under paragraph 64(c) of CEPA as they are not entering the environment in a quantity or concentration or under conditions that constitute or may constitute a danger in Canada to human life or health.
Therefore, it is concluded that dimethyl sulfide, benzyl disulfide, tert-dodecyl mercaptan and grapefruit mercaptan do not meet any of the criteria set out in section 64 of CEPA.
References
Burdock, G. 2010. Fenaroli’s Handbook of Flavor Ingredients, sixth ed. CRC Press, Boca Raton, FL.
Butterworth, K.R., Carpanini, F.M.B., Gaunt, I.F. Hardy, J., Kiss, I.S. and Gangolli, S. D. 1975. Short-term toxicity of dimethyl sulphide in the rat. Fd. Cosmet.Toxicol. 13:15-22 [cited in OECD 2006]
Canada. 1999. Canadian Environmental Protection Act, 1999. S.C. 1999, c.33. Canada Gazette Part III, vol. 22, no. 3. [Accessed 2016 Feb 22].
Catalan L, Liang V, Johnson A, Jia C, O'Connor B, Walton C. 2009. Emissions of reduced sulphur compounds from the surface of primary and secondary wastewater clarifiers at a Kraft Mill. Environ Monit Assess. 156(1-4):37-49.
ChemIDplus [database]. 2018. Bethesda, MD: National Institutes of Health; [Accessed 2018 June 25].
Chemspider [database]. 2015. London, UK: Royal Society of Chemistry; [Accessed 2016 Feb 22].
City of Hamilton. 2009. Ambient Air Monitoring Program Year 2009 [PDF], City of Hamilton Glanbrook Landfill, Binbrook, Ontario. [Accessed 2016 Nov 22].
[ConsExpo] Consumer Exposure Model. 2006. Version 4.1. Bilthoven (NL): Rijksinstituut voor
Volksgezondheid en Milieu (National Institute for Public Health and the Environment). [Accessed 2016 Feb 22].
Drimal M, Koppová K, Klöslová Z, Fabiánová E. 2010. Environmental exposure to hydrogen sulfide in central Slovakia (Ruzomberok area) in context of health risk assessment. Cent Eur J Public Health. 18(4):224-9.
[ECCC] Environment and Climate Change Canada. 2016a. Science approach document: ecological risk classification of organic substances. Ottawa (ON): Government of Canada.
[ECCC] Environment and Climate Change Canada. 2016b. Data used to create substance-specific hazard and exposure profiles and assign risk classifications in the Ecological Risk Classification of organic substances. Gatineau (QC). Available from: substances@ec.gc.ca.
[ECCC, HC] Environment and Climate Change Canada, Health Canada. [modified 2017 Mar 12]. Categorization. Ottawa (ON): Government of Canada. [Accessed 2016 Nov 20].
[ECCC, HC] Environment and Climate Change Canada, Health Canada. 2017. Draft screening assessment: substances identified as being of low concern using the ecological risk classification of organic substances and the threshold of toxicological concern (TTC)-based approach for certain substances. Ottawa (ON): Government of Canada.
[EFSA] European Food Safety Authority. 2009. Flavouring Group Evaluation 8 (FGE.08): Aliphatic and alicyclic mono-, di-, tri-, and polysulfide s with or without additional oxygenated functional groups from chemical group 20. [Accessed 2016 Feb 22].
[EFSA] European Food Safety Authority. 2011a. Scientific Opinion on Flavouring Group Evaluation 74, Revision 1 (FGE.74Rev1): Consideration of Simple Aliphatic Sulfide s and Thiols evaluated by the JECFA (53rd and 61st meeting) Structurally related to Aliphatic and Alicyclic Mono-, Di-, Tri-, and Polysulfide s with or without Additional Oxygenated Functional Groups from Chemical Group 20 evaluated by EFSA in FGE.08Rev1 (2009). [Accessed 2016 Feb 22].
[EFSA] European Food Safety Authority. 2011b. Scientific Opinion on Flavouring Group Evaluation 74, Revision 2 (FGE.74Rev2): Consideration of Simple Aliphatic Sulfide s and Thiols evaluated by the JECFA (53rd and 61st meeting) Structurally related to Aliphatic and Alicyclic Mono-, Di-, Tri-, and Polysulfide s with or without Additional Oxygenated Functional Groups from Chemical Group 20 evaluated by EFSA in FGE.08Rev3 (2011). [Accessed 2016 Feb 22].
[EFSA] European Food Safety Authority. 2014. Scientific Opinion on Flavouring Group Evaluation 91, Revision 2 (FGE.91Rev2): Consideration of simple aliphatic and aromatic sulfides and thiols evaluated by the JECFA (53rd and 68th meetings) structurally related to aliphatic and alicyclic mono-, di-, tri-, and polysulfide s with or without additional oxygenated functional groups evaluated by EFSA in FGE.08Rev5 (2012). [Accessed 2016 Feb 22].
Elf Atochem SA. 1995. Biodegradabilite du Dimethyl Sulfure. Report 50794/HT [cited in OECD 2006]
Environment Canada. 2009. DSL Inventory Update data collected under the Canadian Environmental Protection Act, 1999, section 71: Notice with respect to certain inanimate substances (chemicals) on the Domestic Substances List. Data prepared by: Environment Canada, Health Canada; Existing Substances Program.
Environment Canada. 2014. Results from Phase Two of the Domestic Substances List Inventory Update Rapid Screening Assessment of Substances of Lower Ecological Concern – Detailed Spreadsheet. Ecological Assessment Division, Environment Canada, Gatineau, QC, Canada.
[EPI Suite] Estimation Program Interface Suite for Microsoft Windows [estimation model]. c2000-2012. Ver. 4.11. Washington (DC): US Environmental Protection Agency, Office of Pollution Prevention and Toxics; Syracuse (NY): Syracuse Research Corporation. [Accessed 2016 Feb 22].
European Union Food Flavourings Database [Database]. [Accessed 2016 Nov 22]
[FCC] Food Chemicals Codex. 2016 National Academy of Sciences, Washington, DC. In: The United States Pharmacopeia-National Formulary (USP39–NF34). [Accessed 2016 Nov 22].
Gao Y, Wang G, Fan P, Wang T. 1993. Mutagenic and teratogenic effects of BDS. Weisheng Dulixue Zazhi 7(4):214-16.
Goyer N. 1990. Evaluation of occupational exposure to sulfur compounds in paper pulp kraft mills. Am Ind Hyg Assoc J. 51(7):390-4.
Kangas J, Jäppinen P, Savolainen H. 1984. Exposure to hydrogen sulfide, mercaptans and sulfur dioxide in pulp industry. Am Ind Hyg Assoc J. 45(12):787-90.
Kim KH, Kim YH. 2014. Composition of key offensive odorants released from fresh food materials. Atmos Environ 89:443–52.
Klein F, Platt SM, Farren NJ, Detournay A, Bruns EA, Bozzetti C, Daellenbach KR, Kilic D, Kumar NK, Pieber SM, Slowik JG, Temime-Roussel B, Marchand N, Hamilton JF, Baltensperger U, Prévôt AS, El Haddad I. 2016. Characterization of Gas-Phase Organics Using Proton Transfer Reaction Time-of-Flight Mass Spectrometry: Cooking Emissions. Environ Sci Technol. 50(3):1243-50.
Kotowska U, Zalikowski M, Isidorov VA. 2012. HS-SPME/GC-MS analysis of volatile and semi-volatile organic compounds emitted from municipal sewage sludge, Environ. Monit Asses 184(5):2893-2907.
Kroes R, Renwick AG, Feron V, Galli CL, Gibney M, Greim H, Guy RH, Lhuguenot JC, van de Sandt JJ. 2007. Application of the threshold of toxicological concern (TTC) to the safety evaluation of cosmetic ingredients. Food Chem Toxicol. 45(12):2533-62.
[LNHPD] Licensed Natural Health Products Database [database]. [modified 2018 Feb 06]. Ottawa (ON): Government of Canada. [Accessed 2018 Jan 05].
McGarry S, 2012. Audited Draft Report. Reverse mutation in five histidine-requiring strains of Salmonella typhimurium. 2-Mercapto-4-methylpentan-2-one. Covance Laboratories Ltd. Study no. 8261928. September 2012. Unpublished report submitted by EFFA to FLAVIS Secretariat [cited in EFSA 2014]
Microbiological Associates, Inc. 1995a. Micronucleus cytogenetic assay in mice. Laboratory Study No. G95AT25.122. Submitted to Elf Atochem North America, Inc. Philadelphia, PA [cited in OECD 2006]
Microbiological Associates, Inc. 1995b. Salmonella/Escherichia coli closed-phase preincubation mutagenicity assay with an independent repeat assay. Laboratory Study No. G95AT25.502003. Submitted to Elf Atochem North America, Inc. Philadelphia, PA [cited in OECD 2006]
Nakamura, S., Oda, Y. and Ugawa, M. 1990. Induction of umu Gene Expression in Salmonella typhimurium TA1535/p SK1002 by Dimethyl Sulfoxide (DMSO). Mutation Research, 229(1):11-15 [cited in OECD 2006]
[NCI] National Chemical Inventory [Database]. [modified 2017 May 15]. [Accessed 2016 Jul 20]
[NHPID] Natural Health Products Ingredients Database [database].. [modified 2018 Jun 25]. Ottawa (ON): Government of Canada. [Accessed 2018 Jan 05].
[OECD]. 2006. SIDS Initial Assessment Report: Dimethyl Sulfide. [Accessed 2016 Feb 22].
[OECD]. 2011. SIDS initial Assessment Report: C8 - C12 Aliphatic Thiols Category. [Accessed 2017 May 18].
Opdyke, D. L. J. 1979. Fragrance Raw Materials Monograph, Dimethyl Sulfide. Fd. Cosmet. Toxicol. 17:365-367 [cited in OECD 2006]
[RIVM] Rijksinstituut voor Volksgezondheid en Milieu [National Institute for Public Health and the Environment]. 2006. General fact sheet: limiting conditions and reliability, ventilation, room size, body surface area: updated version for ConsExpo 4 [PDF]. Bilthoven (NL): RIVM. Report No.: 320104002/2006. [Accessed 2016 Nov 22].
[RIVM] Rijksinstituut voor Volksgezondheid en Milieu [National Institute for Public Health and the Environment]. 2007. Paint products fact sheet: to assess the risks for the consumer: updated version for ConsExpo 4 [PDF]. Bilthoven (NL): RIVM. Report No.: 320104008/2007. [Accessed 2016 Feb 22].
Susaya J, Kim KH, Phana NT, Kim JC. 2011. Assessment of reduced sulfur compounds in ambient air as malodor components in an urban area Atmos Environ 45(20):3381–90.
Tansy, M.F., Kendall, F.M., Fantasia, J., Landin, W.E. and Oberly, R. 1981. Acute and subchronic toxicity studies of rats exposed to vapors of methyl mercaptan and other reduced-sulfur compounds, J. Toxicol. Environ. Health, 8:71-88 [cited in OECD 2006]
Terazawa K, Mizukami K, Wu B, Takatori T. 1991. Fatality due to inhalation of dimethyl sulfide in a confined space: a case report and animal experiments. Int J Legal Med 104(3):141-4. [cited in OECD 2006]
[WHO]. World Health Organization. 2000. Joint FAO/WHO Expert Committee on Food Additives (JECFA). Safety Evaluation of Certain Food Additives and Contaminants. WHO Food Additives Series 44. [Accessed 2016 Jul 20]
WIL Research Laboratories, LLC. 2004. A prenatal developmental toxicity study of dimethyl sulfide in rats. Report # WIL-495002. Unpublished study sponsored by the Dimethyl Sulfide Producers Association [cited in OECD 2006]
Young KW, Danielewska-Nikiel B, Munro IC. 2006. An evaluation of the maximized survey-derived daily intake (MSDI) as a practical method to estimate intake of flavouring substances. Food Chem Toxicol. 44(11):1849-67.
Appendix A. Dermal and inhalation exposures to humans from wall paint
Human exposures were estimated using the ConsExpo scenario for ‘Brush/roller painting, waterborne wall paint’ (RIVM 2007). All recommended default values from this scenario were used, except for parameters listed below.
Concentration of tert-dodecyl mercaptan in wall paint = 0.1 % (Environment Canada 2009)
Molecular weight of tert-dodecyl mercaptan = 202 g/mol
Vapour pressure of tert-dodecyl mercaptan = 22.8
Pa Kow of tert-dodecyl mercaptan = 5.85 10Log
Parameters used to calculate inhalation and dermal exposure to wall paint
Inhalation model: Exposure to vapour: evaporation
Exposure duration: 132 minutes
Room volume: 20 m3
Ventilation rate: 0.6 /hr
Product amount: 1.25E3 gram
Release area: 10 m2
Application duration: 120 minute
Application temperature: 20 oC
Molecular weight matrix: 120 g/mol
Mass transfer rate: 0.223 m/min (Thibodeaux)
Inhalation absorption: 100 %
Inhalation rate: 16.2 m3/day
Dermal model: direct dermal contact with product: constant rate
Exposed area: 0.367 cm2 (RIVM 2006)
Contact rate: 30 mg/min
Release duration: 120 minutes
Dermal absorption: 100 %