Caribou (Rangifer tarandus) barren-ground population COSEWIC assessment and status report 2016: chapter 3
Wildlife species description and significance
Name and classification
Caribou are members of the deer family (Order: Artiodactyla, Family: Cervidae, Subfamily: Capreolinae, Genus: Rangifer, Species: tarandus; Gilbert et al. 2006). Banfield (1961) classified Rangifer into subspecies and “intergrades”, based largely on morphometric measurements of skulls and leg bones as well as pelage colouration and antler form. This taxonomy has not been updated, even though it was often based on a few specimens and techniques have changed, including DNA analyses that are revealing evolutionary relationships among caribou. COSEWIC (2011) compiled recent information to describe and classify caribou diversity below the species levels as separate DUs.
This report refers to the barren-ground subspecies (Rangifer tarandus groenlandicus), including the migratory Porcupine subpopulation, which Banfield (1961) classified as R. t. granti “intergrades” with features resembling R. t. groenlandicus (pure R. t. granti were from the Alaskan Peninsula). COSEWIC (2011) combined R. t. groenlandicus and R. t. granti intergrades in Canada based on shared phylogenetic lineage and adaptive behaviours. In this report, caribou in DU 3 (COSEWIC 2011) are referred to as Barren-ground Caribou (See Designatable Units).
Based on nuclear and mitochondrial DNA analyses, Barren-ground Caribou are from the Beringian lineage which includes other Alaskan caribou and the Northern Mountain Caribou (Zittlau 2004; Weckworth et al. 2012; COSEWIC 2014a). Caribou colonization of north-central Canada occurred as recently as 6,000 years before present (ybp) with the Ahiak and Qamanirjuaq subpopulations established as recently as 2,000 to 3,000 ybp (Zittlau 2004).
Morphological description
Barren-ground Caribou is a medium-sized member of the deer family with characteristically large hooves, broad muzzle and a distinct brown and white coat pattern in the fall. The large dew claws are prominent in appearance, giving these caribou a large and flexible weight-bearing surface on ice and in snow. The coat pattern is the most pronounced for males during the rut when they have a white and dark brown flank stripe setting off the brown back from the white belly and brown legs. The neck often has a prominent white mane contrasting with a dark brown head. Newborn calves are typically ruddy. Pelage pattern and colouration from light to dark vary seasonally as the timing of shedding the winter pelage varies between sex and age classes. Caribou are the only member of the deer family where the females are antlered. Some females may not grow antlers in every year, which may reflect varying nutritional state (Reimers 1993; Bergerud et al. 2008).
Adult females can weigh 90-135 kg in the fall and lose about 10% of their weight during the winter, although this varies greatly depending on the winter foraging conditions as body weight tends to increase and decrease with environmental conditions and population abundance (see Biology) (Boertje 1996; Miller 2003). Mature males are 10-15% larger and weigh 10-50% more than adult females, their antlers are more massive and their heads have a more convex profile than females. The females have a less developed mane and their antlers are smaller with shorter tines and a less massive main beam. Juvenile males are a similar size as females and so have to be distinguished by their external genitalia (Miller 2003).
Caribou appearance varies seasonally as the pelage becomes more bleached and faded during the winter until it is shed during the summer, earlier for males and calves or later for lactating females (Cuyler et al. 2012). Mature males shed their antlers after the rut while barren females retain their antlers until late winter and pregnant females generally do not shed their antlers until just after their calves are born. The size and branching of the antlers are influenced by the individual’s body condition (Bergerud et al. 2008).
Barren-ground Caribou differ in size, body proportions, pelage and behaviour from other caribou subspecies. While they are not as long-legged as Boreal Caribou DU individuals, they are not as short-legged or as small-bodied as Peary Caribou or Dolphin and Union Caribou DUs. A clear distinction during summer into early fall is their characteristic dark brown antler velvet compared to the grey velvet of Peary Caribou. The legs and backs are typically darker brown than Peary or Dolphin and Union Caribou although when pelage is faded later in the season, the differences are less apparent. However, the characteristically longer legs and face of Barren-ground Caribou remain distinct in late winter. Boreal Caribou have similar darker pelage as Barren-ground Caribou but their relatively longer legs and behaviour distinguish them (COSEWIC 2014b).
Population spatial structure and variability
Across the continental mainland, there are few conspicuous geographic barriers (mountain ranges or major rivers) that could impose subpopulation structure by limiting movements of Barren-ground Caribou. A possible exception is the Mackenzie River valley – a low-lying valley with dense boreal forests that may be a partial barrier to the Porcupine subpopulation whose annual range lies west of the Mackenzie River. On the eastern part of Barren-ground Caribou range, Southampton Island, Coats Island and Mansel Island in the Hudson Bay are generally isolated by strong currents and high tides. Recently, however, sea ice in winter has linked Southampton to the mainland (Campbell and Boulanger 2016). Baffin Island is mostly isolated from the mainland and has mountainous terrain that may contribute to subpopulation structure across the island (Campbell et al. 2014).
The current state of knowledge of Barren-ground Caribou supports a continuum between sympatric discrete subpopulations and spatially complex subpopulations (demes with or without source-sink dynamics) rather than fixed categories (Ciannelli et al. 2013). Spatially complex subpopulations could vary over time as abundance and distribution change and calving becomes more dispersed or gregarious. However, gaps in information are evident, as described in the following sections.
Behavioural basis for subpopulation structure
Since the 1960s, the behavioural mechanism for subpopulation structure has been based on the annual return of females to specific geographic areas for calving, a traditional behaviour that it is passed between generations (Lent 1964; Kelsall 1968; Thomas 1969; Bergerud 1974; Gunn and Miller 1986). The association between females on particular calving grounds persists year-round despite often overlapping winter distribution between neighbouring subpopulations. For example, based on over 10,000 ear-tag and visual collar returns, Heard (1983) recorded 93–95% fidelity among the Beverly, Bathurst and Qamanirjuaq subpopulations between 1959 and 1981. Likewise, Nagy et al. (2011) found that calving ground associations of satellite-collared females persisted during the rest of the year. Their statistical cluster analyses of the year-round movements of satellite-collared females supported the same subpopulations as those based on fidelity to calving grounds. Females associated during calving were also associated during the rut, which is support for using fidelity to a geographic calving ground to indicate subpopulation identity (Gunn et al. 2013a).
Calving strategies and subpopulation structure vary when relative population densities are low (see Habitat), or when abundance changes. Calving is more dispersed on the northeast mainland – an area characterized by low vegetation productivity where calving densities are relative low (Heard and Stenhouse 1992; Nagy et al. 2011; Appendix B.). Responses to lower vegetation productivity may be compounded by how changing abundance affects association between females. For example, on northern Baffin Island, gregarious migration has shifted to dispersed individual home ranges concomitant with a large decrease in abundance (Baffinland 2012).
Available data on fidelity to a geographic calving area are uneven across the range of Barren-ground Caribou. For the subpopulations identified according to fidelity to calving grounds, the fidelity has been sampled over an average of 35 years (2.7 years SE). Fidelity of individual adult females to a specific geographic calving ground is typically high (~95%; Gunn et al. 2012). Births are highly synchronized as most calves are born during a peak of a few days. This means that females and calves on their calving and post-calving grounds are exposed to similar environmental conditions of foraging and predation for the first few months of their lives. This also helps explain differences in vital rates among subpopulations (see Fluctuations and Trends). Such behaviour results in sympatric discrete subpopulation structures; these are not closed subpopulations, but immigration/emigration is typically less a driver of abundance than births and deaths (Boulanger et al. 2011).
Gregarious calving and post-calving is likely promoted by the predictability of the learned location (Gunn et al. 2012), hence the advantage of fidelity to a geographic location. The geographic location used for calving can shift but the association of females remains constant. The overlap between consecutive calving grounds annually varies (Griffith et al. 2002; Gunn et al. 2007, 2008; Nagy 2009a). Directional shift over a number of years can lead to a geographically changed location of a calving ground, as was the case for the Bathurst subpopulation’s calving ground that shifted at peak abundance. This latter shift probably was related to high densities on the calving grounds. Conversely, exceptionally low densities of calving females may be a mechanism for switches in calving ground location (Gunn et al. 2012; Adamczewski et al. 2015). Environmentally forced changes such as icing (Nagy 2009a) can lead to temporary switches in the fidelity of individual caribou to specific calving grounds (see Distribution).
Genetic basis for subpopulation structure
Techniques to identify subpopulation structure for Barren-ground Caribou other than behavioural isolation, such as analyses of non-metric skeletal morphology, have not been undertaken. Interest in assessing patterns of genetic variation relative to subpopulation structure has been strong but genetic variation and demographic isolation operate at different spatial and temporal scales (for example, Esler et al. 2006). The extent to which genetic variation can be expected among subpopulations is tempered by the recent post-glacial spread and the absence of geographic barriers, both of which reduce the likelihood of strong genetic differentiation for Barren-ground Caribou (McFarlane et al. 2016).
Limitations to genetic analyses include uneven geographic distribution of sampling and uneven sample sizes, especially for males. The ability to discern subpopulation genetic structure also depends on analytical techniques, as mitochondrial DNA, unlike nuclear DNA analyses, is only inherited from females (Zittlau 2004; Cronin et al. 2005; Weckworth et al. 2012). Current genetic analyses depend on sampling selection-neutral genes, but functional genes (Kennedy et al. 2010) may possibly detect finer-scale genetic structure at the subpopulation scale.
Gene flow is characterized by sex-biased dispersal. Male dispersal within Canada is measurable from DNA sampling, except for the Southampton Island and Porcupine subpopulations (McFarlane et al. 2016). Some of the annual variation in gene flow likely reflects caribou movement when abundance was high and the likelihood of dispersal was greatest (McFarlane et al. 2016). Occasional female dispersal has occurred but the movements are not ongoing based on DNA sampling.
Subpopulations considered in this report
Currently, there are 14-15 subpopulations considered in this report, based on fidelity of females during calving, clustering of year-round association of satellite-collared females and isolated or semi-isolated geographic areas (Table 1; see Canadian Range). Some were combined for the purposes of population trends analyses, for a total of 13 units (see Population Sizes and Trends). Although major subpopulations (commonly referred to as “herds”) have been consistently recognized, the total number of subpopulations of Barren-ground Caribou is uncertain due to evolving information, re-analyses, re-naming and changes in definitions. Infrequent surveys and low numbers of marked individuals have led to different interpretations for identifying subpopulations, especially for the Beverly, Ahiak, and northeast mainland subpopulations (Appendix B).
Parker (1971) referred to “four major populations on mainland Canada”, but also several “herds” within each. In 1991, Ferguson and Gauthier (1992) identified 11 loosely defined “populations”. Between 1991 and 2009, six additional subpopulations on the northeast mainland were recognized based on aerial surveys and telemetry. A re-analysis of aerial survey and telemetry data (Nagy et al. 2005; Nagy 2009a) led to a split of the previously recognized Bluenose subpopulation into three units: Cape Bathurst, Bluenose-West and Bluenose-East. The Tuktoyaktuk subpopulation received recognition in 2005 following the removal of domesticated reindeer (Branigan 2005; Nagy and Johnson 2006; Davison and Branigan 2011).
Campbell et al. (2015b) summarized the information for three subpopulations on Baffin Island. Although earlier aerial surveys had revealed migratory caribou returning to calving grounds, other caribou were non-migratory and calving was dispersed. Ferguson and Gauthier (1992) initially delineated three Baffin Island “populations” (South, North, and Northeast), based on ear-tag returns and satellite collars and on where caribou congregated in the fall for the rut. Jenkins et al. (2012) noted that available information and Inuit Qaujimajatuqangit (IQ, or Inuit Knowledge) support those three “demographically independent populations”. Campbell et al. (2015b) used the 1987-1994 survey and 2008-2011 satellite telemetry data to map these.
| Subpopulation (or area) | Duration of study (years) | No. surveys | Telemetry | Genetic (DNA sampled) |
|
|---|---|---|---|---|---|
| Reasonable certainty | Porcupine | 38 | 20 | Yes | Yes |
| Reasonable certainty | Cape Bathurst a | 32 | 20 | Yes | Yes |
| Reasonable certainty | Bluenose-West a | 32 | 22 | Yes | Yes |
| Reasonable certainty | Bluenose-East a | 32 | 22 | Yes | Yes |
| Reasonable certainty | Bathurst a | 52 | 24 | Yes | Yes |
| Reasonable certainty | Beverly a, b | 37 | 23 | Yes | Yes |
| Reasonable certainty | Qamanirjuaq a | 45 | 19 | Yes | Yes |
| Reasonable certainty | Ahiak b | 25 | 7 | Yes | Yes |
| Reasonable certainty | Wager Bay c | 28 | 4 | blank | Unknown |
| Reasonable certainty | Lorillard a,c | 25 | 4 | blank | Unknown |
| Geographical isolation (island) | Southampton | blank | blank | No | Yes |
| Geographical isolation (island) | Coats | blank | blank | No | Unknown |
| Uncertain (limited information) | Tuktoyaktuk Peninsula | 2 | 2 | Unreported | Unknown |
| Uncertain (limited information) | Boothia Peninsula | 36 | 5 | Low number | Unknown |
| Uncertain (limited information) | Arrowsmith Lowlands d | 3 | 6 | Low number | Unknown |
| Uncertain (limited information) | Keith Bay d | 20 | 3 | Low number | Unknown |
| Uncertain (limited information) | Simpson Lake d | 26 | 4 | Low number | Unknown |
| Uncertain (limited information) | Simpson Peninsula d | 22 | 3 | Low number | Unknown |
| Uncertain (limited information) | South Melville Peninsula d | 1 | 1 | Low number | Unknown |
| Uncertain (limited information) | Baffin Island e | 8 | 2 | Yes | Yes |
a Subpopulation considered robust (Nagy et al. 2011).
b After 2011, considered as combined Beverly-Ahiak population unit in this report. See discussion of alternative interpretations of population structure in Subpopulations and Subpopulation-specific trends.
cPopulation estimates combined for Lorillard and Wager Bay in this report.
d Renamed as Ahiak by Campbell et al. (2012) and included in this report within a combined Beverly-Ahiak population unit.
e Subpopulation structure uncertain (Campbell et al. 2015b).
There are two interpretations for the subpopulation structure of the Beverly and Ahiak (sometimes referred to as Queen Maud Gulf) subpopulations, which largely rest on different explanations of the timing, extent and underlying mechanism for emigration of females from inland to coastal calving grounds (see Dispersal and Migration and Subpopulation-specific trends). One hypothesis is that the Beverly subpopulation is no longer identifiable, as numbers on the inland (traditional) calving grounds declined between 1994 and 2006 to the point when the few remaining females joined the Ahiak subpopulation’s calving grounds along the Queen Maud Gulf coast (Gunn et al. 2012, 2013a; Adamczewski et al. 2015). An alternative explanation (e.g., Nagy et al. 2011 and Campbell et al. 2014) is that both subpopulations remain extant with contiguous or partially overlapping calving grounds along the Queen Maud Gulf coast. In this interpretation, the Beverly subpopulation shifted from farther inland and started to calve along the unoccupied western part of the Queen Maud Gulf coast in the mid-1990s. Those authors assert that the shift in calving distribution would have taken place over many years.
These interpretations are limited by significant gaps in both survey and radio-collaring information, and so the prospects for resolving this debate are clearly limited at present. With a satellite radio-collaring program for the Ahiak and Beverly subpopulations not having been initiated until 2001 (and with low numbers of collars; Gunn and D’Hont 2002). The first stratified survey was in 1986 but not repeated until 2008 (see Subpopulation-specific trends). Hence, there are significant gaps in information (Campbell et al. 2014; Adamczewski et al. 2015) that make it impossible to resolve this debate. For this assessment, information related to Beverly and Ahiak subpopulations prior to 2011 is presented as originally designated and referred to in government reports and publications. After 2011, this report refers to “Beverly/Ahiak” as a combined unit, encompassing both the traditional Beverly inland calving ground and the one along the Queen Maud Gulf coast.
Designatable units
COSEWIC (2011) recognized Barren-ground Caribou with all its subpopulations as one of 11 extant caribou DUs. The Barren-ground Caribou is a discrete and significant unit phylogenetically different from Eastern Migratory Caribou of Québec/Labrador and Ontario/Manitoba, which have similar aggregating calving behaviour (COSEWIC in prep.). The aggregating behaviour distinguishes Barren-ground from Peary (COSEWIC 2016) and Dolphin and Union Caribou. Banfield (1961) recognized subpopulations in this unit as belonging to two subspecies (R. t. groenlandicus, R. t. granti), which COSEWIC (2011) combined into a single DU because of their genetic and behavioural similarities (COSEWIC 2011).
Barren-ground Caribou may overlap periodically and/or seasonally with other DUs, including Northern Mountain Caribou (COSEWIC 2014a), Dolphin and Union Caribou, Peary Caribou (COSEWIC 2016), Boreal Caribou (COSEWIC 2014b) and Eastern Migratory Caribou (COSEWIC in prep.). Peary Caribou and Barren-ground Caribou periodically occupy adjacent ranges on western and eastern Boothia Peninsula, respectively (Thomas and Everson 1982; Gunn et al. 2000a). Dolphin and Union Caribou migrate to the mainland from Victoria Island after the rut and winter along the coast with possible overlap with tundra-wintering Barren-ground Caribou (Gunn et al. 2000b; McFarlane et al. 2016; COSEWIC in prep.). Information is lacking as to the extent and frequency of the spatial and temporal overlap. The seasonal overlapping distribution of the Bluenose-West, Bluenose-East, Bathurst, Beverly and Qamanirjuaq subpopulations with Boreal Caribou and Eastern Migratory Caribou is in boreal forests during winter, especially when abundance is high. Movements of radio-collared individuals from the Qamanirjuaq caribou to the range of the Cape Churchill caribou (Eastern Migratory) are known although the extent and frequency is unreported (COSEWIC 2011).
The annual range of the Porcupine subpopulation is separated from neighbouring subpopulations in Canada by the Mackenzie River valley and delta (see Canadian Range), but it exhibits no significant genetic or behavioural differentiation that might compel the designation of a separate DU (COSEWIC 2011). COSEWIC (2011) tentatively included the Fortymile subpopulation in Barren-ground Caribou, and singled it out as requiring further consideration. It is not, however, included in this assessment as part of the Barren-ground Caribou DU, nor is it currently assigned to any identified caribou DU in Canada. Similar to the Nelchina subpopulation, Fortymile caribou often winter in central-western Yukon (Gross 2011), but the annual range of this subpopulation occurs within a transboundary mountain range; unlike all other subpopulations considered in this assessment, the Fortymile and Nelchina subpopulations do not migrate north of tree-line. Instead, pre-calving migration is elevational to alpine tundra. The peak of calving for the Fortymile subpopulation is approximately May 17th – two weeks earlier than Barren-ground subpopulations.
Special significance
The ecological significance of Barren-ground Caribou largely stems from their abundance. Common species (or populations) can shape ecosystems by their sheer strength of numbers (Gaston and Fuller 2008). Barren-ground Caribou is also often considered a keystone species. The boreal and Arctic food webs have relatively few relationships among the organisms as compared to more southern systems. Northern ecosystems are nutrient-limited because so much carbon and nitrogen is inaccessible, with only a shallow active layer of the soil thawing each year. Caribou, through their forage intake and output (fecal pellets), have complex effects that are strongly patterned over time and space. As caribou travel and rest on frozen waterways, the nutrient returns from fecal pellets to aquatic ecosystems where filter-feeding larvae contribute to nutrient cycling in aquatic systems. Adult female mosquitoes, in turn, feed on caribou for blood, joining a complex and diverse community of parasites dependent on caribou (Kutz et al. 2012). Additionally, Barren-ground Caribou support predators and scavengers.
The significance of Barren-ground Caribou to the peopling of northern Canada is evident from archaeological findings tracking how the distribution of people and Barren-ground Caribou were affected by the retreating glaciers some 8,000 years ago in the central barrens (Gordon 2005) and as long ago as 24,000 cal BP (calibrated years Before Present) in the central range of the Porcupine subpopulation (Bourgeon et al. 2017). Many Indigenous peoples recognize the central role of caribou in tundra and taiga ecology and revere Barren-ground Caribou beyond simply depending on meat and hides. Barren-ground Caribou have been and continue to serve as a key resource for people in northern Canada; in some cases these animals have such importance that families would follow their migration (Benson 2015). Caribou have significant direct economic value from harvest, primarily for subsistence use. They also contribute to the northern economy through wildlife tourism and recreational hunting. Beyond this, Barren-ground Caribou have incalculable cultural value for Indigenous peoples throughout the range (Gunn et al. 2011b; InterGroup Consultants Ltd. 2013).
Distribution
Global range
Because COSEWIC (2011) did not consider subpopulations with entire ranges located outside Canada, the global range of this DU is uncertain. The western extent of the current range of Barren-ground Caribou as identified by COSEWIC (2011) is marked by the seasonal extensions of the Porcupine subpopulation into eastern Alaska. Ecological similarities of the Alaskan large coastal subpopulations (e.g., Western Arctic, Teshekpuk, and Central Arctic) with the Canadian Barren-ground Caribou DU suggest the possibility that the global range of this DU extends to the Bering Sea coastline of northern Alaska (Figure 1). Likewise, the eastern extent may be in western Greenland. The ecological counterpart of Barren-ground Caribou in northern Russia was identified as a different subspecies (R. t. sibiricus) by Banfield (1961).
Canadian range
The combined range of 14-15 subpopulations of Barren-ground Caribou (see Subpopulations and Subpopulation-specific trends) is widespread and continuous across northern continental mainland Canada, Hudson Bay islands and Baffin Island. The western extent is the northwest Yukon boundary with Alaska. The range extends eastwards to the western coast of Baffin Island (Figures 1 and 2). The larger islands of Hudson Bay (Southampton, Coats and Mansel) have caribou, although recent information on Mansel Island is lacking, and not considered here. The Belcher Islands have introduced domestic reindeer (R. t. tarandus). The islands off the west coast of Baffin Island in Hudson Strait (Mill, Salisbury, and Nottingham) and Foxe Basin (Koch and Rowley) are currently not occupied by caribou (Jenkins et al. 2012). The southern extent of Barren-ground Caribou range is northern Saskatchewan, Alberta, and Manitoba. The range of the Porcupine subpopulation extends into Alaska.
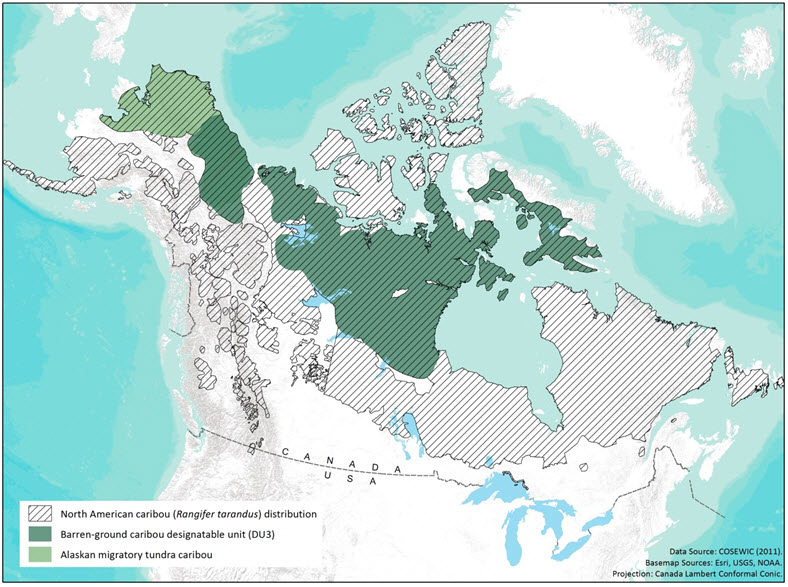
Long description for Figure 1
Map of Barren-ground Caribou distribution in North America relative to Caribou from other designatable units.
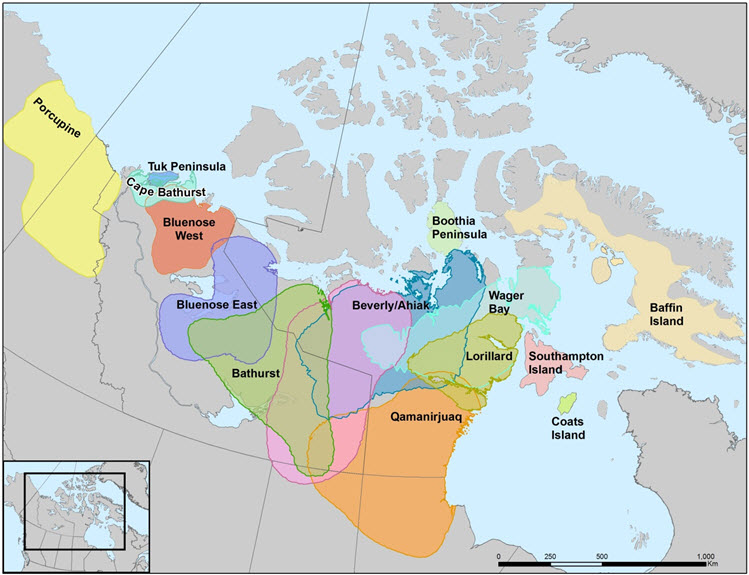
Long description for Figure 2
Map delineating subpopulations in the Barren-Ground Caribou designatable unit.
The Mackenzie River valley and delta likely cause a disjunction between the western distribution of Barren-ground Caribou, isolating the Porcupine subpopulation from the subpopulations to the east (WMAC (North Slope) and Aklavik HTC 2009). Genetic analysis of some caribou sampled in the foothills of the Mackenzie Mountains, NWT show that they were more similar to Barren-ground Caribou than both Boreal Caribou and Northern Mountain Caribou sampled further up the mountains and west (Polfus et al. 2016). This fits with descriptions by a local resident of historical movements of Barren-ground Caribou that crossed the Mackenzie River from the east to the west shore (TK section, SARC 2016). South of Norman Wells, in 1954, caribou crossed the Mackenzie River from the east (J. Antoine reported in Beaulieu 2012). Ice conditions fragment the distribution of Barren-ground Caribou within Hudson Bay restricting caribou to islands; similarly, marine ice conditions mostly restrict caribou to Baffin Island.
Historical trends in distribution
Available information to assess historical trends (ca. 1950s) is patchy for the northern distribution of Barren-ground Caribou. These historical trends are primarily based on infrequent surveys and mapping of low numbers of satellite-collared females (see Search Effort) and ATK, particularly through subsistence harvest (e.g., SARC 2016). Compiled ATK indicates a marked contraction away from the southern portion of Barren-ground historical range over the past 60 years, which extended as far south as Fort Smith, Fort Resolution, and the Rocher River in the NWT, as well as Fort Fitzgerald and Fort Chipewyan in Alberta (ACFN Elders et al. 2003a,b; WRRB 2010b; Beaulieu 2012; BQCMB 2014a). Distribution trends can be influenced by fluctuations in abundance and their corresponding changes in seasonal distribution. Longer-term information (< 1950s) for calving and summer ranges from archaeological sites and hoof-scars on spruce roots document long-term fidelity for pre-calving migratory routes, post-calving areas and water-crossings (Gordon 2005; Zalatan et al. 2006). Blythe and Bath (in SARC 2016) mapped the Barren-ground Caribou range in the 1990s, the most recent population peak, based on data compiled from cited traditional knowledge sources (Figure 3).
Overall, there is some evidence for a contraction of the southern and southwest distribution of Barren-ground Caribou since 1935 (Banfield 1954, 1961; Kelsall 1968; SARC 2016). Kelsall (1968) mapped Barren-ground Caribou west of the Slave River into Wood Buffalo National Park since 1935 and Beaulieu (2012) reported caribou as far south as Fort Chipewyan for the period 1948-58, suggesting that the southern winter distribution has contracted to the north for the Beverly and Bathurst subpopulations. (Thomas et al. 1998; Gunn et al. 2011a). Thomas et al. (1998) summarized historical information to suggest a contraction of at least 200 to 300 km between the 1960s and the 1990s. Based on distribution information from government surveys (1940s-2011), radio-telemetry (1993-2012) and traditional knowledge of caribou harvesters, BQCMB (2014a) documented cessation of use of much of the former winter range in Alberta and Saskatchewan for the combined Beverly and Qamanirjuaq ranges.
Assessing annual trends in distribution for individual subpopulations is limited because annual variation is high and winter ranges often overlap between neighbouring subpopulations (Thomas et al. 1998; Gunn and D’Hont 2002; Nagy et al. 2005; Nagy and Campbell 2012). The Porcupine subpopulation, which has not overlapped range with neighbouring subpopulations since monitoring with satellite collars began in 1985 and has somewhat predictable range use patterns (Fancy et al. 1989; McNeil et al. 2005), is an exception. Likewise, there is some evidence for contraction of the southern extent of fall and winter distribution of the Bathurst subpopulation along with declines in abundance. D’Hont (pers. comm. 2005) indexed the contraction by a reduced length of pre-calving migration from the winter range to the calving ground (Figure 4). In addition, while the area of the rut range has declined, based on the straight-line distance between the centroid of the annual calving grounds and the centroid of the rut range, the location of the rut range has also contracted north. Between 1996 and 2011, the reduction was about 200 km.
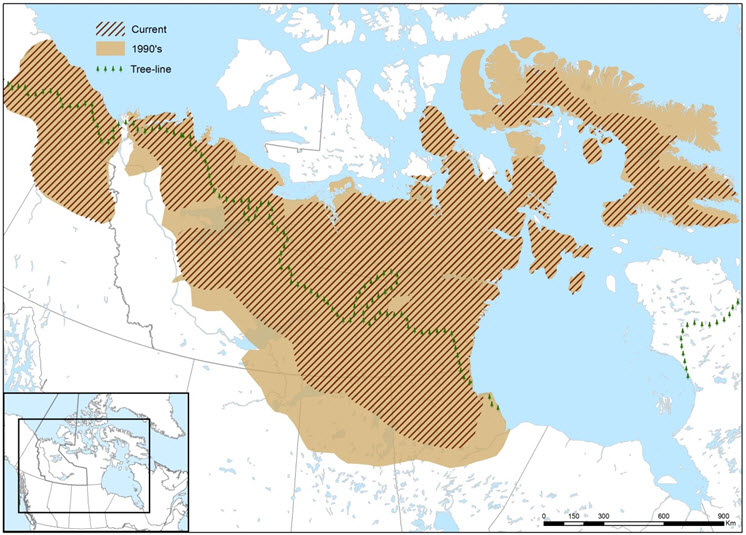
Long description for Figure 3
Map delineating maximum Barren-ground Caribou range in Canada in the 1990s, based on data compiled from cited traditional knowledge sources and current distribution.
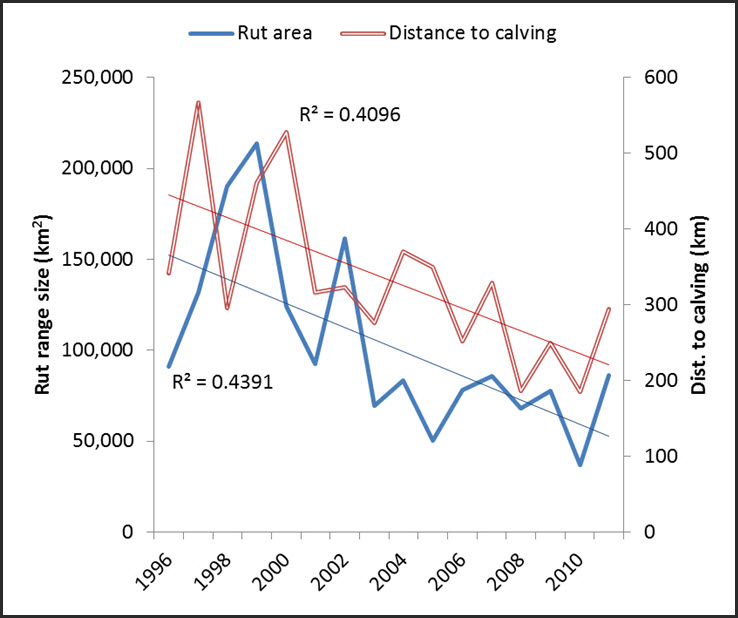
Long description for Figure 4
Chart illustrating trends (1996 to 2011 approx.) for size of the rut range and straight-line distance between the centroid of the annual calving grounds and annual rut ranges for the Bathurst subpopulation of the Barren-ground Caribou.
Baffin Island is the eastern margin of Barren-ground Caribou distribution in Canada. Abundance has sharply declined since the 1990s (Goorts 2014). Historically, when abundance was low, distribution – especially in winter – contracted to traditional refuges, a phenomenon well-known to Indigenous elders (Ferguson and Vivaventsova 2007; DOE-GN 2013). During the 2012 environmental assessment for a large open pit mine, historical information was compiled for northern Baffin (Baffinland 2012; Campbell et al. 2015a). In the current phase of low numbers, distribution had contracted and the females were dispersed in individual home ranges rather than a gregariously used and recognizable calving ground.
Based on the cumulative distribution of satellite-collared females (1996-2011), areas on the northeast mainland (Simpson Peninsula, northern Melville Peninsula, western Boothia Peninsula and King William Island; Appendix B) appear unoccupied (Fig. 2.2 in Nagy and Campbell 2012). However, whether the distribution is a contraction possibly related to change in abundance and/or reflects different techniques to describe distribution is uncertain. For example, in May 2014, Campbell et al. (2015a) report low numbers of caribou on northern Melville Peninsula. Historically, caribou migrated from the mainland to King William Island for calving and summer and were hunted during the fall migration south to Adelaide Peninsula. Although the abundance of caribou on the northeast mainland including Boothia Peninsula increased during the 1980s into the 1990s, this did not include King William Island (summarized in Gunn and Ashevak 1990; Gunn et al. 2000a). Inuit report caribou recently on the island year-round ( Ljubicic pers. comm. 2014).
A reduction in fall and winter distribution during the phases of low abundance compounds difficulty in discerning overall contractions in distribution with the risk that the current distribution is considered ‘normal’; this could be considered an example of a shifting baseline (Pauly 1995), meaning that as populations dwindle, each human generation’s standard for how “it used to be” gradually degrades.
Extent of occurrence and area of occupancy
The extent of occurrence (EO) is calculated as the area included in a convex polygon that encompasses the geographic distribution of all known populations of the species, excluding cases of vagrancy. For most subpopulations, governments generated the distribution maps from satellite telemetry. The EO is based on polygons generated from satellite-collared females. The assumption that the collared females are representative of the subpopulation’s distribution is only well supported for overall calving distribution (see comments below on the AO). Sample size (collar-years) and the reliance on collaring adult females introduces uncertainty into the EO estimates. The number of collar-years is variable among subpopulations and years and has increased since 2005 for some subpopulations during a time of declining abundance. Thus, EO may be biased by sampling constraints and underlying changes in population abundance.
Mapping was not available to compare extent of occurrence for both peak abundance (approximately mid-1990s) and the phase of low abundance (~2008-12). Instead, the extent of occurrence was calculated using GNWT-Environment and Natural Resources (ENR) and CircumArctic Rangifer Monitoring and Assessment (CARMA) data for the period covering roughly 2006-12 (compiled by K. McGreish from NWT’s Wildlife Information Management System [WMIS] and CARMA data). EO at peak population abundance and to account for possible differences in the range of males (because only females have been collared in most subpopulations) may be upwards of 5-10% because typically adult males are distributed further south than females in the boreal forest (Thomas et al. 1998).
The EO calculated using a single polygon encompassing all 17 subpopulations (2006-2012) was 4,253,842 km2. The area of occupancy (AO) is defined by IUCN (2016) as the smallest essential area at any stage for the survival of existing subpopulations. For migratory Barren-ground Caribou, this is their calving grounds, including the area occupied by breeding females up to three weeks post-birth. This has parallels with pelagic migratory mammals such as Northern Fur Seals (Callorhinus ursinus) where breeding sites were assessed as AO (COSEWIC 2010). The AO is measured both as an estimate of the actual area occupied (the ‘biological occupancy’) and as an index of area of occupancy (IAO), which uses a scale-correction factor to standardize this estimate across different spatial scales. The IAO is measured as the surface area of 2 km x 2 km grid cells that intersect the actual area occupied by the wildlife species. The IAO for Barren-ground Caribou was calculated as the cumulative annual calving range of the eight subpopulations between 2006 and 2012 and totalled 247,840 km2.However, NE mainland, Southampton and Baffin were not included in this calculation, due to incomplete knowledge.
Search effort
Historical distribution is largely based on aerial surveys conducted during the 1940s and 1950s that covered large areas of the tundra and boreal forest on the continental mainland (Banfield 1954; Kelsall 1968, Urquhart 1981) and ATK (Figure 3). The southern extent of Barren-ground Caribou range is winter range, but since the 1960s this is only infrequently mapped through aerial surveys (e.g., D’Hont et al. 2009; Gunn 2013).
By the 1960s, efforts were targeted towards mapping calving distribution (Table 2), but search effort has been uneven among subpopulations. Eight subpopulations have been mapped the most frequently and have had higher numbers of satellite-collared individuals (Tables 2, 3). In 2007 and 2008, standardization of calving distribution surveys increased across the NWT and NU (Poole et al. 2013). In 2010 and 2011, relatively widespread aerial surveys were flown in June to map calving distributions for the northeast mainland (Campbell et al. 2014; Appendix B). In earlier surveys, the criteria for delineating the boundaries of calving grounds were not always comparable or reported. Consequently, it is not always clear whether, for example, non-breeding caribou (juveniles and young males) were included in survey estimates.
Search effort to map Barren-ground Caribou distribution changed in the mid-1980s when VHF telemetry was used and then in the mid-1990s, when satellite telemetry was initiated. The numbers of collar-years to assess distribution varies markedly among subpopulations and years and could be a source of bias in determining the size of calving grounds (Table 3). Annual ranges of caribou in the Kivalliq region of Nunavut are mapped in a recent atlas (Campbell et al. 2012) based on aerial surveys and collared females.
Most satellite-collared caribou are adult females. Males were collared for the five western subpopulations for which abundance is estimated from the photography of post-calving aggregations and those aggregations are located through radio and satellite-collared caribou. The distribution of males, as indicated by satellite locations has not been compared to the distribution of females. In winter, males are often found further south than females (Kelsall 1968). AO calculations are also derived from satellite telemetry, which for females is representative of their aggregated distribution during calving, at least for the larger subpopulations (Griffith et al. 2002; Gunn et al. 2008). Kernel analyses (usually set at 95% or 99% but 50-70% are also used among studies and subpopulations) are used to define the boundaries of the AO. However, kernel methods are sensitive to sample size, which adds uncertainty to the AO estimates (Seaman et al. 1999).
| Subpopulation | Period | No. annual surveys | References |
|---|---|---|---|
| Porcupine | 1972-2012 | 41 | Fancy and Whitten 1991 (1983-90), Russell et al. 1992 (1970-1990), Griffith et al. 2002, Caikoski 2011 |
| Cape Bathurst | 1974-2012 | 22 | Nagy and Johnson 2006, Nagy 2009a, Davison et al. 2014, Davison and Veitch in prep. |
| Bluenose-West | 1974-2012 | 24 | Nagy and Johnson 2006, Nagy 2009a, Davison et al. 2014, Davison and Veitch in prep. |
| Bluenose-East | 1974-2013 | 22 | Nagy 2009a, Nagy et al. 2008, Adamczewski et al. 2013, Adamczewski pers. comm. 2014 |
| Bathurst | 1965-2012 | 29 | Sutherland and Gunn 1996; Gunn et al. 2008; Nishi et al. 2007, 2010; Boulanger et al. 2015 |
| Ahiak | 1986-2011 | 8 | Gunn et al. 2013a, Johnson et al. 2008, Campbell et al. 2014 |
| Beverly | 1957-2011 | 29 | Gunn and Sutherland 1997b, Johnson et al. 2009; Campbell et al. 2014 |
| Qamanirjuaq | 1963-2008 | 19 | Campbell et al. 2010 |
| Herd | Mean | SE | Minimum | Maximum | Years |
|---|---|---|---|---|---|
| Porcupine | 77.7 | 2.74 | 53 | 95 | 18 |
| Tuktoyaktuk Pen. | 12.0 | 2.05 | 5 | 20 | 7 |
| Cape Bathurst | 9.9 | 1.80 | 1 | 22 | 17 |
| Bluenose East | 16.5 | 4.23 | 3 | 47 | 15 |
| Bluenose West | 15.3 | 3.22 | 2 | 46 | 17 |
| Bathurst | 13.6 | 1.31 | 3 | 22 | 17 |
| Ahiak f | 13.4 | 2.88 | 2 | 39 | 15 |
| Beverly g | 2.0 | 0.70 | 0 | 8 | 12 |
| Queen Maud Gulf | 6.9 | 3.12 | 1 | 27 | 9 |
| Lorillard | 8.6 | 1.23 | 2 | 12 | 9 |
| Wager Bay | 5.5 | 1.28 | 1 | 11 | 10 |
| Qamanirjuaq | 13.3 | 2.13 | 7 | 31 | 15 |
f Calved along the Queen Maud Gulf coastline.
g Calved on the traditional Beverly calving grounds south of Garry Lake.
Habitat
Habitat requirements
Barren-ground Caribou require relatively large annual ranges which allow individuals to adapt their seasonal distribution to annual and decadal variation in environmental conditions (Bergerud et al. 2008). This includes the direct influences of weather and its indirect effects on habitat attributes, such as snow depth, forest fire pattern and intensity, insect harassment, forage quality or timing of plant growth. Caribou use seasonal migrations and local movements to meet their requirements for forage and to minimize the risk of predation and perhaps parasitism (Heard and Williams 1992; Heard et al. 1996; Kutz et al. 2014; Klaczek et al. 2016).
Caribou occur in four ecozones (Taiga Shield, Taiga Plains, Taiga Cordillera, and Arctic); the Arctic ecozone is further divided into 3 ecoregions (Southern Arctic, Northern Arctic, and Arctic Cordillera; Eamer et al. in press). Most subpopulations of Barren-ground Caribou migrate north toward the Arctic coast to calve, summer and rut on the tundra of the Southern Arctic ecoregion.
The islands within Hudson Bay and Baffin Island are within the Southern Arctic ecoregion of the Arctic ecozone. Northeastern Nunavut mainland, including Boothia and Melville peninsulas, King William Island and most of western and central Baffin Island are located within the Northern Arctic ecoregion, while the eastern edge of Baffin Island falls within the Arctic Cordillera. Many Barren-ground Caribou return south each fall to winter in the boreal forests of the Taiga Plains or Taiga Shield ecozones (Gunn et al. 2011b). West of the Mackenzie River, the Porcupine subpopulation largely uses the Taiga Cordillera ecozone except to calve along the narrow coastal extension of the Southern Arctic ecozone. Most of the Taiga Cordillera is mountainous tundra with coniferous forest mostly confined to lowland areas (ESTR Secretariat 2011).
Forage requirements depend on the timing of the annual breeding cycle relative to the brief plant growing season and long winters characteristic of the sub-arctic and arctic regions (e.g., Russell et al. 1993). Forage requirements are high during spring and early summer after body reserves are depleted during winter and females must confront the metabolic costs of pregnancy, pre-calving migration and lactation. These requirements cannot always be met because of insect harassment and wolf predation risk so late summer fall foraging is important because that is when body reserves are accumulated (Heard et al. 1996). While much is known about requirements for protein and carbohydrates, there is less information about micro-nutrients (Kuropat and Bryant 1980). Both sexes seek out mineral-rich overflow on lakes during spring migration and females use sodium-rich mineral licks on at least the Beverly and Bathurst inland calving grounds (Fleck and Gunn 1982; Heard and Williams 1990).
During non-winter months, caribou select for nutrient content according to the stage of plant growth rather than plant species (Kuropat and Bryant 1980). Plant growth begins as the snow ablates and melts, so Barren-ground Caribou time their pre-calving migrations to the tundra to take advantage of the peak nutritional value of the plants relative to energy and protein requirements which differs between males and non-pregnant females (White and Trudell 1980; Russell et al. 1993; Heard et al. 1996; Chen et al. 2012). Given low food resources characteristic of calving areas and the increasing costs of lactation, pregnant females precede plant development and move to greening-up vegetation after calving (Heard et al. 1996).
Caribou can meet their habitat requirements for calving within relatively diverse tundra landscapes, varying from low-elevation coastal plains to inland elevated rocky and hilly areas (Chen et al. 2012). The strategy is to minimize encounters with predators on calving grounds (Fancy and Whitten 1991; Griffith et al. 2002; Bergerud et al. 2008; Heard and Williams 1992) and minimize exposure to parasitic flies (Folstad et al. 1991).
Habitat requirements during post-calving and summer allow caribou to balance forage intake relative to reduced exposure to biting and parasitic flies (e.g., Russell et al. 1993). Such habitats include higher elevation topography with stronger winds and shallow water where caribou may avoid parasitic flies; the latter may also be a strategy for cooling (see Parasites and diseases). By fall, habitat requirements are tied to selection for nutritious and digestible forage to compensate for foraging time lost as a result of insect harassment and avoidance. This includes habitats with shrubs, grasses and sedges, and mushrooms (Russell et al. 1993). These requirements are met on the tundra and the tree-line transition zone. The timing of the early snowfalls and fall storms influences fall movements (McNeil et al. 2005). Boertje (1984) reported extensive use of mushrooms by Barren-ground Caribou in Alaska.
Habitat requirements during winter include areas with relatively shallow or soft snow to reduce the energetic costs of obtaining lichens, sedges and dried shrub leaves. Lichens occur in mats and are efficient to forage and are high in digestible carbohydrates (Svihus and Holand 2000) but are low in protein. Caribou offset the low protein content by recycling nitrogen and select vascular plants higher in protein (Aagnes et al. 1995; Parker et al. 2005). Caribou can meet their winter habitat requirements on the tundra, in the tree-line transition zone or within the boreal forest (Gunn et al. 2013b).
Caribou select late-succession boreal forest in winter; as lichens do not recover until decades after a fire, they tend to avoid recent burns (Thomas and Kiliaan 1998). However, caribou may still move through such areas early in the winter (Thomas et al. 1998; Barrier and Johnson 2012) and they also select the edges of the burns (Anderson and Johnson 2014).
The breadth of the tundra zone increases between Bathurst Inlet and the west coast of Hudson Bay as the tree-line dips further south (Timoney et al. 1992). Subpopulations on a west to northeast gradient across the mainland meet their habitat requirements with an increasing proportion of their annual range on the tundra. The Cape Bathurst, Tuktoyaktuk, and the northeast mainland subpopulations winter on the tundra and transition zone (the tree line).
Habitat requirements during pre-calving migration favour efficient movement as distances from winter to calving range can be several hundred kilometres. Environmental conditions such as fall icing can cause caribou to shift their winter ranges, which can increase the length of pre-calving migration (McNeil et al. 2005). Caribou select and pack trails along frozen waterways and lakes with relatively harder snow than under a tree canopy. Routes and rates differ between early and late snow melt years, being slower when snow melt is late (McNeill et al. 2005).
Within the Arctic ecozone, there are strong west-east and north-south climatic gradients that influence plant productivity (e.g., the length of the plant growing season decreases on the northeast mainland). Annual net plant productivity varies widely across the Canadian range (Figure 5). The subpopulations with the highest peak abundance (see Abundance) have the majority of their summer range where annual net above and below ground plant productivity is 250-1000 g m-2 yr-1 (from Gould et al. 2003); those subpopulations also winter in the boreal forests with their extensive lichen mats. Calving is more dispersed and at lower densities for the subpopulations where plant productivity on the tundra is <50 g m-2 yr-1 (Figure 5).
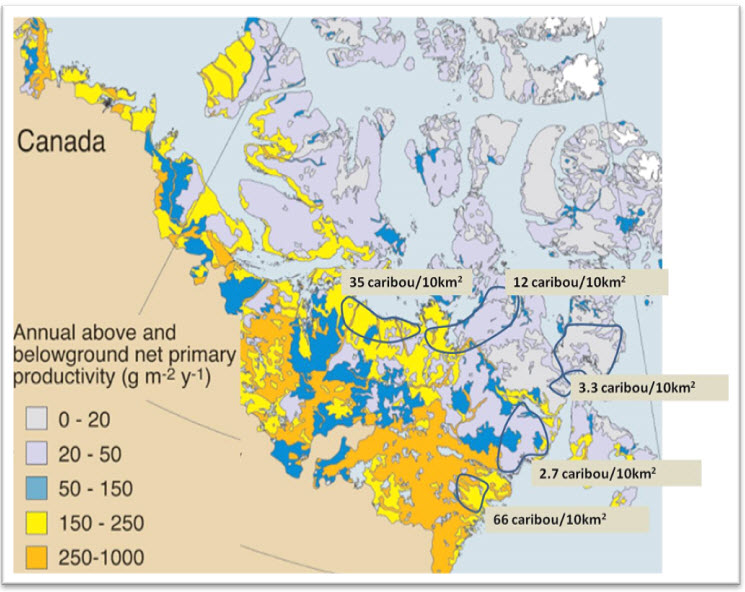
Long description for Figure 5
Map illustrating patterns of above- and below-ground annual net primary productivity in grams per square metre per year. Estimated densities of adult caribou per 10 square kilometres during calving are shown for several subpopulations of Barren-ground Caribou.
Habitat trends
Traditional and community knowledge contains numerous accounts (or descriptions) of habitat change in NWT (e.g., Kendrick 2003; BQCMB 2011; WRRB 2013) and Yukon. The factors that are causing these changes include the combined effects of fire, climate change, anthropogenic development and habitat fragmentation in boreal forest portions of the range, and increased off-road vehicle use damaging vegetation on the summer range (Whaèhdôö Nàowoò Kö [Dogrib Treaty 11 Council] 2001; Kendrick 2003; Tłįchǫ Government 2007; Nesbitt and Adamczewski 2009; Katz 2010; BQCMB 2011; WRRB 2013; ACCWM 2014).
Habitat availability
Information gaps about food availability result partly from varied measuring approaches and also from uneven sampling effort among subpopulations and seasonal ranges. Observational studies of diet and distribution in habitat types have largely shifted to focus on habitat selection through satellite-collared caribou (e.g., Bathurst subpopulation for summer and winter ranges; Gunn et al. 2011c; Barrier 2011). For example, the availability of winter range since the peak of abundance has only been measured for the Bathurst subpopulation (Barrier and Johnson 2012).
Trends in anthropogenic development
Increasing habitat loss associated with human activities is rated as a major threat for migratory ungulates worldwide (Berger 2004). For Barren-ground Caribou, the actual footprint of habitat loss due to development is still small but measurable at each subpopulation level (Figure 6). Increasing concerns are being voiced in public hearings about habitat change within the ranges of several subpopulations (e.g., NWMB 2016; WRRB 2016) and management plans (e.g., BQCMB 2014a).
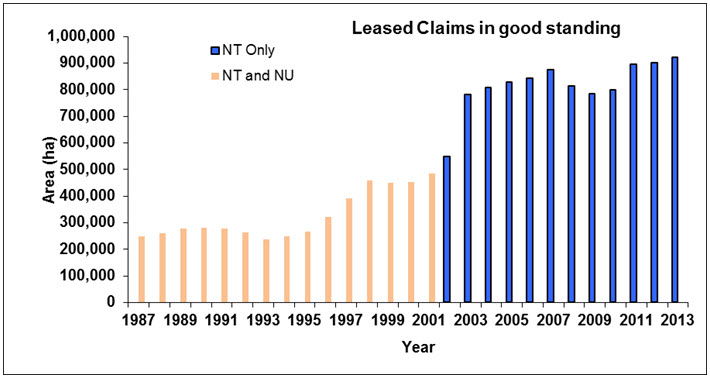
Long description for Figure 6
Chart illustrating the trend in land area covered by leased claims in good standing (developed mines) in the Northwest Territories alone (2002 to 2013) and in the Northwest Territories and Nunavut combined (1987 to 2001).
Development potential, dependent on underlying geology, is relatively straightforward to predict at the broad geographical scale. For example, the mineralization of the Slave Craton (Bathurst subpopulation range) has high mining potential while the sedimentary basins (Bluenose-West and Cape Bathurst) are more associated with oil and gas development. While the rate of exploration has increased (Figure 7), metal and oil price cycles will influence future rates of exploration and development.
Typically, exploration occurs as bursts of activity (Figure 7). An example was for the Bathurst subpopulation tundra ranges when exploration for diamonds peaked in 1993. The effects were not assessed until 1996 (when satellite-collared female locations became available). Exploration sites are now included during cumulative effects assessments for mines (Figure 8a, Figure 8b and Figure 8c). For example, De Beers (2012) reported that the direct footprint of those developments, including the now-permitted Gahcho Kué mine, would cumulatively reduce caribou habitat for the Bathurst subpopulation by up to 7.3% on the autumn range.
While exploration activities are relatively short-lived and most do not lead to full development, exploration can still have an impact, and can induce significant cumulative impacts; this activity is subject to minimal tracking and regulations. Developed mines have projected lives of 15-30 years. The number of proposed or constructed roads as part of mine developments is increasing on tundra ranges, mostly for the Bathurst subpopulation (Figure 8a, 8b and 8c). Over 1,000 km of all-weather roads to service mines have been constructed since 2001 or are proposed (Table 4) with 14 mines operational or currently undergoing project-level environmental assessment (www.NIRB.ca; www.MVEIRB.ca). Of all subpopulations, Bathurst has the most current and potential industrial development on its range (Figure 8a, 8b and 8c), but the ranges of other subpopulations are also demonstrating growing signs of encroachment by development (Figure 9; BQCMB 2014a). Additionally, mines abandoned prior to complete remediation can have persistent effects, mainly local contamination. Twelve of the 22 abandoned mines in the NWT require action; the other mines and one in NU are likely to require remediation (Office of the Auditor General of Canada 2002).
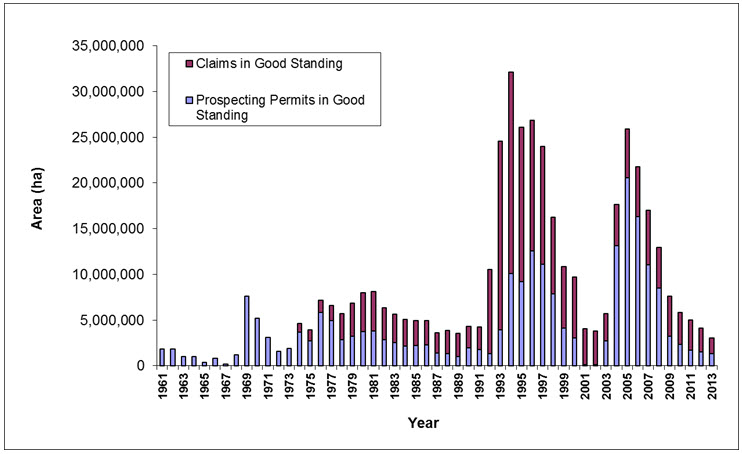
Long description for Figure 7
Chart illustrating trends in land area allotted to prospecting permits and mineral claims (in good standing) per year for the Northwest Territories from 1961 to 2013 and for Nunavut prior to 2001.
In northern Saskatchewan, Peach and Hovdeba (2003) reported 40 abandoned uranium mines and two abandoned mills. Beaverlodge Lake and three adjacent watersheds in the Uranium City area have been seriously contaminated with uranium and selenium mostly dating back to 1952-1982 (Prebble and Coxworth 2013). There are currently four operational uranium mines in the historical winter range of the Beverly subpopulation (Key Lake, McClean Lake, Rabbit Lake, and McArthur River); the Tazi Twe Hydroelectric Project is currently under federal assessment review, and three mines are being decommissioned or are in care-and-maintenance. The trend since the 1980s has been toward increased all-season road access to the caribou ranges. An all-weather road (Highway No. 914) was proposed in 2010 to link two mines, which would complete a circular linkage of highways within central Saskatchewan (Government of Saskatchewan 2010).
Since 2010, all-season roads and hydro-transmission lines have been proposed for winter ranges within the boreal forest. On the western edge of the Bathurst subpopulation’s range, an all-season road is under consideration to replace the current winter road between Tlicho communities (Figure 8a, 8b and 8c). This proposed road is necessary before the Nico Fortune mine can be built, and is currently in the scoping phase of an environmental assessment (MVEIRB Public Registry 2016). On the western edge of the Cape Bathurst, Bluenose-West and Bluenose-East subpopulation’s winter ranges, the Government of Northwest Territories is proposing to build an all-season 818-km road to extend the Mackenzie Valley Highway from Wrigley to the Dempster Highway linking Wrigley to Inuvik (MVEIRB Public Registry 2016). The proposed road is designed for up to 100 vehicles/day although the estimated traffic volumes are predicted to be 50 vehicles/day. The Inuvik to Tuktoyaktuk Highway is currently being constructed within the Cape Bathurst subpopulation winter range. The feasibility of an all-season road from Sundance, Manitoba to Rankin Inlet, Nunavut has been actively assessed since 2007 with completed route selection (2007) and business case study (2010) and community consultations, and would cross hundreds of kilometres of the Qamanirjuaq range (Government of Manitoba 2016). The Dempster Highway is the only all-season road (736 km) crossing the Porcupine subpopulation’s winter range. It increases accessibility to hunters when the subpopulation is in that part of its extensive winter range (Government of Yukon 2011). Traffic on the Dempster Highway was relatively stable from 1993 to 2005 and then increased. Road-related impacts are dependent on the location of the subpopulation; for example, 2015 was the first year in five that members of the Porcupine subpopulation were in the vicinity of this highway.
| Subpopulation | Road type | Operation | Road km | Road km | Start-up year | Start-up year | Access |
|---|---|---|---|---|---|---|---|
| Baffin | Ore haul road mine to port | Mary River (Baffinland)h Tote Road |
100 | (1960s) 2014 |
(1960s) 2014 |
Public | Public |
| Qamanirjuaq | Ore haul road pit to plant | AEM Meliadineh | 24 | Proposed | Proposed | Public | Public |
| Lorillard and Ahiak | Supply road from Baker Lake | AEM Meadowbankh | 107 | 2010 | 2010 | Public | Public |
| blank | Ore haul road pit to plant | AEM Meadowbank (Whale Tail extension)i |
c. 50 | Proposed | Proposed | Private | Private |
| Bathurst | Ore haul road pit to plant | DDEC Ekati (Misery Rd)j | 27 | 2001 | 2001 | Private | Private |
| blank | Ore haul road pit to plant | DDEC Ekati (Sable Rd)j | 20 | 2019 | 2019 | Private | Private |
| blank | Ore haul road pit to plant | DDEC Ekati (Jay Rd)j | 5 | 2022 | 2022 | Private | Private |
| blank | Ore haul road pit to plant & supply road to winter road to Yellowknife | BIPAR Phase 2 to i Contwoyto Lake | 132 | Post-poned | Post-poned | Public | Public |
| blank | Ore haul road pit to plant | MMG Izok to Grays Bay Road and Port i | 80 | Post-poned | Post-poned | Private | Private |
| Beverly-Ahiak | Ore haul road pit to plant & supply road to port | BIPAR Phase 1 Back River2 & Hackett i | 85 | Post-poned | Post-poned | Public | Public |
| Bathurst | Road for resources from Contwoyto L to port | GN and KitIA Grays Bay Road and Port (Phase 1) | 270 | Proposed | Proposed | Public | Public |
| Bathurst | Road for resources from Yellowknife to Contwoyto Lake | GN and KitIA Grays Bay Road and Port (Phase 2) | c. 600 | Proposed | Proposed | Public | Public |
| Beverly-Ahiak | Ore haul road pit to plant & supply road to port | Doris Northh | 16 | 2008 | 2008 | Private | Private |
| Porcupine | Public highway | blank | 736 | 1979 | 1979 | Public | Public |
| blank | Northern Cross k | Oil and gas well development | c. 95 | Proposed | Proposed | Private | Private |
h Listed as completed environmental assessments NIRB 2014
i Listed as active environmental assessments NIRB 2014
j Listed as completed environmental assessments MVEIRB 2014
k Listed as active environmental assessments YESAB 2016
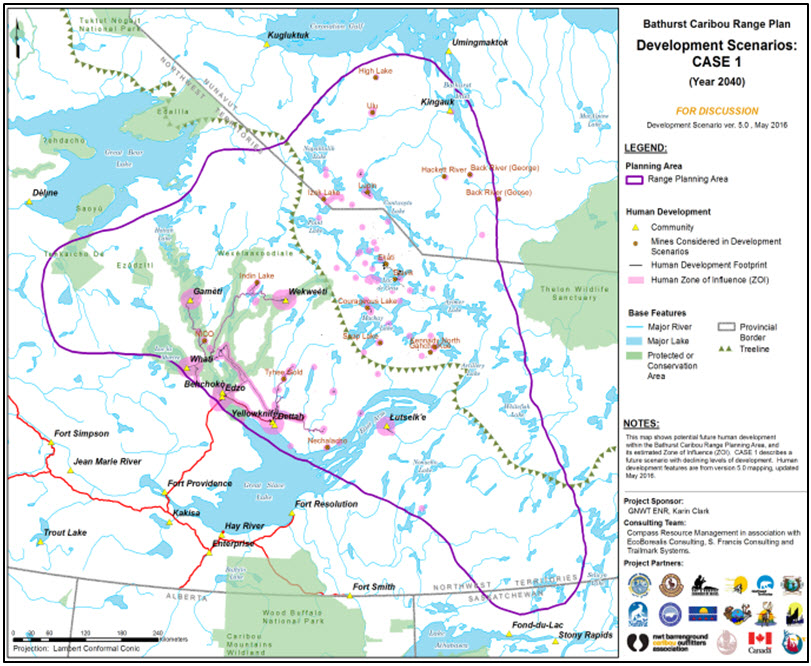
Long description for Figure 8a
Three map panels illustrating human development scenarios for 2040 (declining development, continuing development, and increasing development) in the range of the Bathurst subpopulation of the Barren-ground Caribou, produced to support the Bathurst Caribou Range Planning process.
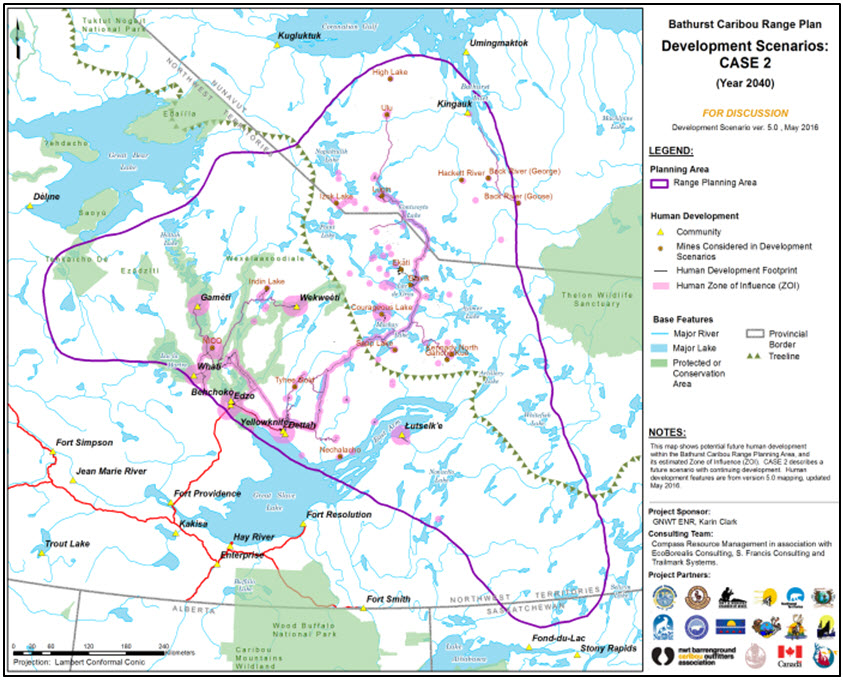
Long description for Figure 8b
Three map panels illustrating human development scenarios for 2040 (declining development, continuing development, and increasing development) in the range of the Bathurst subpopulation of the Barren-ground Caribou, produced to support the Bathurst Caribou Range Planning process.
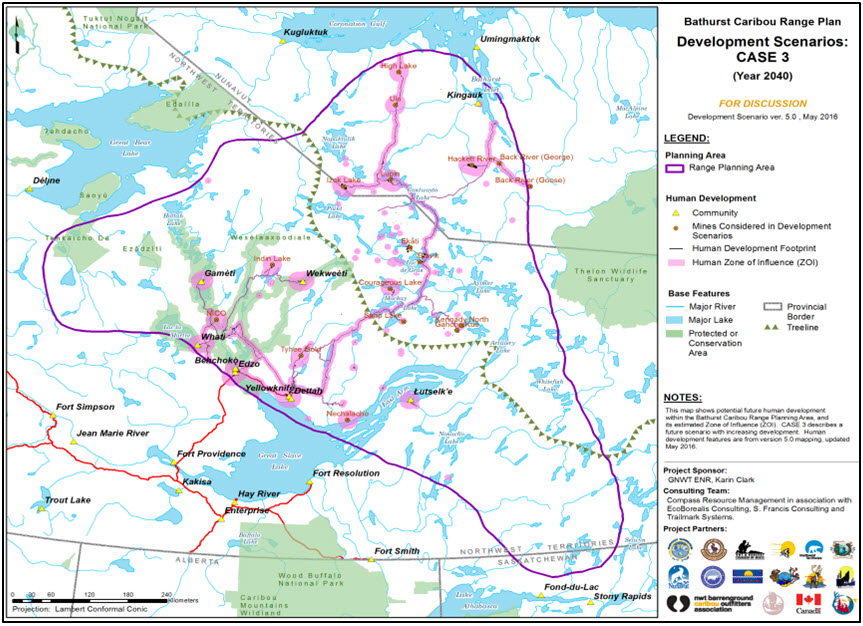
Long description for Figure 8b
Three map panels illustrating human development scenarios for 2040 (declining development, continuing development, and increasing development) in the range of the Bathurst subpopulation of the Barren-ground Caribou, produced to support the Bathurst Caribou Range Planning process.
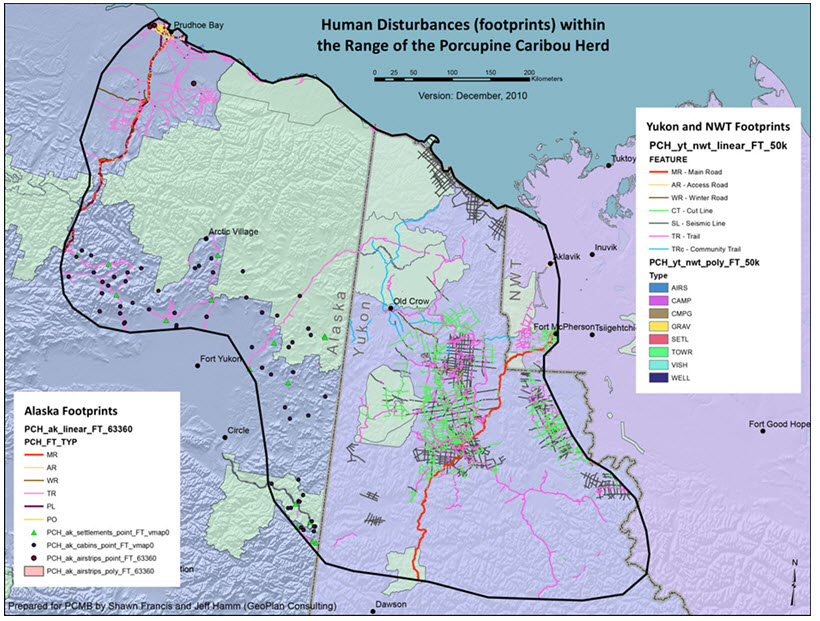
Long description for Figure 9
Map illustrating human disturbance (footprints) within the range of the Porcupine Caribou subpopulation of the Barren-ground Caribou.
Climate-mediated habitat trends
Overall, recent climate and weather trends indicate warmer temperatures, longer snow-free periods, deeper maximum snow depths, warmer ground with associated changes in nitrogen dynamics and increased plant growth. Over the last 30 years, temperatures have risen over 2°C for the Taiga Plains especially in winter and spring (ESTR Secretariat 2011). These temporal trends are based on data from weather stations, which are few across caribou ranges. However, there is also the MERRA spatial dataset, which is applied to caribou seasonal ranges at the scale of 1/2 degrees latitude by 2/3 degrees longitude (Russell et al. 2013).
Chen et al. (2014) found that indicators of summer forage (leaf biomass, phenology, and nitrogen content) derived from remote sensing and weather data explained 59% of the variation in late-winter calf:female ratio for the Bathurst subpopulation between 1985 and 2012. This in turn was correlated to population change in the following seasons, suggesting that weather and summer range conditions can act as severe limiting factors to population-level dynamics for Barren-ground Caribou. It is not known if this is valid for other subpopulations.
Many plant productivity trends for Barren-ground Caribou are driven by decadal climatic patterns, which are the result of larger scale atmospheric patterns, such as the Arctic, North Atlantic (NAO) and Pacific Decadal Oscillations. Since the 1970s, those decadal patterns are superimposed on longer-term trends of a warmer climate. When the Arctic Oscillation is in a negative phase, winters are cooler. From the 1950s to the 1980s, there were frequent shifts between positive and negative phases (Richter-Menge et al. 2006) and then from 1989 to 1995, the Arctic Oscillation was strongly positive – the 1990s on the caribou ranges stand out as a warmer decade. Correlation between changes in caribou abundance and dominant decadal patterns has been mostly documented through the effects of winter foraging conditions (Forchhammer and Post 2004; Zalatan et al. 2006; Joly et al. 2011; Weladji and Holand 2006; Bastille-Rousseau et al. 2013). Weladji and Holand (2006) reported that reindeer age classes born following a high NAO winter index were lighter in summer and early winter, and had a lower absolute growth rate than cohorts born after cold and dry winters (low NAO winter index; State Climate Office of North Carolina 2016).
Plant growth has increased over the last three decades by 20-26% based on satellite imagery (Normalized Difference Vegetation Index; NDVI) correlated with field measurements (Epstein et al. 2012). The increases in plant biomass are strongest along the mainland arctic coast (Cape Bathurst and Queen Maud Gulf areas). However, lichen-dominated communities had consistently lower NDVI trends than vascular-plant-dominated communities (Olthof and Pouliot 2010). In addition, the trends for increasing net plant productivity may not mean an increase in forage quality as, for example, the amount of solar radiation (or cloud cover) and temperature also affect the levels of compounds such as tannins in plants, which affects forage quality (Weladji et al. 2002). Thus the conditions that promote greater primary productivity may also lower the quality of some of the vegetation as food for herbivores (e.g., see Chen et al. 2014).
Forest fires
The long-term trend in area burned for the Taiga Plains and Taiga Shield increased from the 1960s until the 1990s and decreased in the 2000s (Joly et al. 2010). In the Taiga Cordillera, the average annual area burned approximately doubled between the 1960s and 1980s and the 1990s-2000s (Krezek-Hanes et al. 2011). The annual burn rate and the severity of fires is higher in the boreal forests of western Taiga Shield and Taiga Plains (Krezek-Hanes et al. 2011), although most of the area burned has been west of the Bluenose-West winter range. For both the Beverly and Bathurst winter ranges, the prevalence of burns has been higher to the south and west of their ranges (BQCMB 2014a). On the winter ranges of the Bathurst subpopulation, especially south of Great Slave Lake, the trend was for an increase in the area burned from 1947 to 2006, with the 1.06°C increase in the mean June-September temperature a likely driver (Chen et al. 2012). Winter food availability was sufficient for the Bathurst subpopulation at its peak densities of 1.83 ± 0.28 caribou/km2 of total winter range in 1986, but larger and more abundant fires, in addition to fewer older and lichen-rich stands, could reduce future food availability (Barrier 2011). Within the past three generations (24-27 years; see Life Cycle and Reproduction), reduced food availability as a consequence of cumulative disturbance from forest fires may be a concern within the winter ranges of Barren-ground Caribou within the boreal forests (Barrier and Johnson 2012). Figure 10 depicts the history of fires over a 50-year period within caribou ranges in NWT.
In the Arctic ecozone, fires are rare and small, due to lack of fire-prone fuels and small patches of fuels interspersed by rock barrens and surface waters (Krezek-Hanes et al. 2011). However, tundra fires are likely to increase as the temperature-related increase of shrub vegetation cover continues (Elmendorf et al. 2012).
Migration habitat and lake ice
Based on satellite imagery over the past 20 years, the trend for the ice-free period for lakes has been towards a longer ice-free period in the Arctic (Latifovic and Pouliot 2007). The trend is for the ice to form 2-5 days later and breakup is 2-5 days earlier. However, there is little information to describe how and if caribou migration patterns will change in response to the timing of breakup and freeze-up and changing snow conditions, although such changes were modelled by Leblond et al. (2016) for eastern migratory subpopulations.
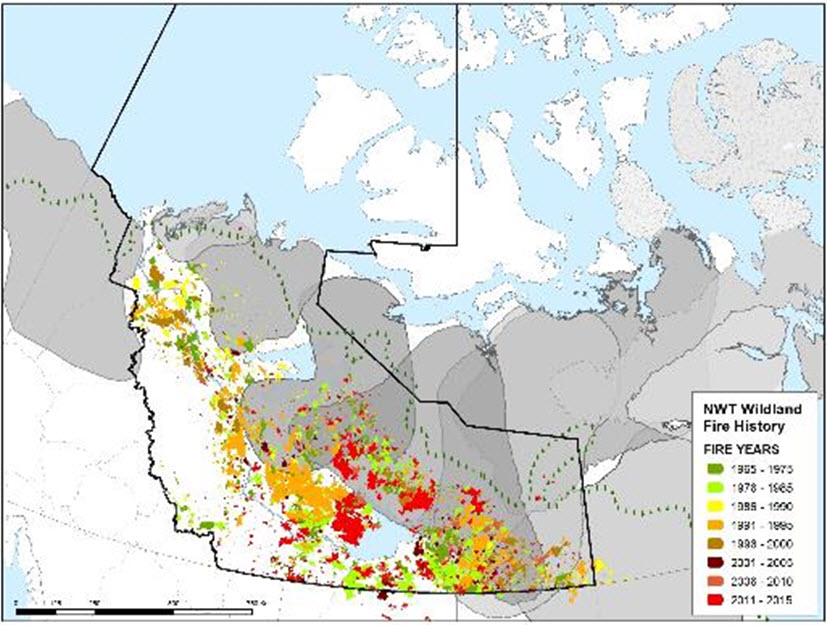
Long description for Figure 10
Map illustrating Northwest Territories’ fire history from 1965 to 2015 superimposed on Barren-ground Caribou subpopulation ranges.
Biology
Some biological information for Barren-ground Caribou (pregnancy rates, calf and adult survival and calving dates) is available from monitoring undertaken by management agencies. Sampling is unevenly distributed among the subpopulations and only for very few subpopulations is the sampling consistent for more than a few years. Understanding the mechanisms for survival and reproductive strategies is mostly dependent on research in Alaska/Yukon (Porcupine subpopulation) and Norway on caribou, domesticated or semi-domesticated reindeer.
Life cycle and reproduction
Barren-ground Caribou are characteristic of many larger-bodied mammals in that adult survival is typically high and reproductive strategies, at least for females, are relatively flexible. At the stages of conception, fetal growth (birth mass) and lactation, a female can trade off between her survival and that for her offspring. This reproductive flexibility is an adaption to annual variations in energetic costs and forage availability.
Generation time is estimated at 8-9 years based on IUCN generation length calculation using mean survival values (generation length = [1/mortality rate] + age at first reproduction; IUCN 2016). Barren-ground Caribou adult survival ranged from 0.66 to 0.85 in the Bathurst subpopulation up to 2009, with age of first reproduction at 3 years (Boulanger et al. 2011). However, calculation of generation time is complicated, as it is not a fixed value but depends on the age structure of the population (Hernandez-Suarez 2011). This generation time conforms with COSEWIC (2014a) and COSEWIC (2016), but Pacifici et al. (2013) and COSEWIC (2014; in prep.) have indicated lower generation times (6-7 years) for caribou in general or other caribou DUs, respectively.
Barren-ground Caribou females usually calve at 3 years of age, although under high forage availability and a corresponding high rate of body growth, they can calve at 2 years of age. The reproductive lifespan is likely about 12 years, with females living as long as 12–16 years, and males a few years less (Thomas and Killiaan 1998). Single births are the rule (Thomas and Killiaan 1998). Initially, calf growth rate depends upon female milk production; lactation demands peak at about 10 days post-partum. After one month old, however, milk production supplies maintenance requirements for the calves and growth rate depends on calf foraging. Underweight calves have a reduced chance of survival (Griffith et al. 2002).
Most calves within a subpopulation are born within a few days of one another, but the timing of the peak of calving may vary annually by a few days. Between 1957 and 1994, the annual peak of calving was a 4-5 day period between 1 and 17 June for the Beverly subpopulation based on sequential counts of calves to females observed on the calving grounds (Gunn and Sutherland 1997b). The technique for determining the peak of calving since 1996 is based primarily on the movements of satellite-collared caribou, as females show a distinctive drop in movement rates upon giving birth. In addition, observations during aerial surveys over calving grounds are used to define that year’s calving peak. At the broad scale of the NWT and NU, the peak of calving is generally earlier for the western subpopulations than for the subpopulations to the east and northeast, based on data from satellite-collared females between 1993 and 2009 (Nagy 2011).
There are relatively few observations of Barren-ground Caribou rutting behaviour (Gunn et al. 2005). Females can have several oestrus cycles of 10-12 days (McEwan and Whitehead 1972; Ropstad 2000), but conceptions are highly synchronous (Dauphiné and McClure 1974). Synchrony during the rut likely leads to birth synchrony, although the timing of birth can change by a few days depending on the condition of females (Bergerud et al. 2008).
Physiology and adaptability
Barren-ground Caribou are adapted to an environment characterized by a long winter season of generally low-quality forage, energetically costly movement and forage accessibility, and low temperatures. The long snow-covered season contrasts with a brief plant growth season when forage is high quality and to which caribou are adapted to take advantage of the brief pulse of green forage. Gregarious and migratory behaviour are key adaptations to a temporally and spatially variable environment. Migration is an adaptation response to seasonal forage availability, predation and parasites. Gregariousness increases individual fitness through predator-swamping (Bergerud et al. 2008; McLellen et al. 2010), increased foraging through reduced vigilance and information exchanges with conspecifics (references in Gunn et al. 2012).
Barren-ground Caribou have numerous physiological adaptations to long and cold winters (reviewed in SARC 2016), including dense pelage consisting of hollow guard hairs and underfur, intricately developed scrolled nasal bones that provide a large surface area to warm and moistened air, and various characteristics to prevent overheating while moving or running through deep snow. They are able to cope with declines in forage quality brought about by the cessation of plant growth through high metabolism of body protein and fat reserves. And during the brief annual pulse of plant growth, they selectively forage on high-protein forage and selectively digest the protein. The ability to digest lichens is a key adaptation in caribou. Lichens have high digestible carbohydrates but low protein content, and unlike most wildlife, caribou have the unique ability to use lichens as an important food resource because they have rumen microflora that can ferment them (Aagnes et al. 1995). Caribou must also seek out evergreen leaves and sedges as well as dried leaves and twigs of shrubs (Russell et al. 1993).
Part of the adaptability of caribou depends on trade-offs between reproduction and survival, which is typical of long-lived species in a variable environment characterized by periods with nutritional stress (Gaillard et al. 1998, 2000). Females can safeguard their own survival in years of restricted forage either by not becoming pregnant, weaning their calf prematurely or extending lactation beyond the rutting period (Russell and White 2000). Less is known about the lifetime reproductive strategies of males, which tend to have more offspring as they mature than females (Melnycky et al. 2013).
Caribou can learn to adapt to human activities (Haskell and Ballard 2008), similar to their responses to predators, ranging from being alert to displacement and avoidance (Stankowich 2008).
Dispersal and migration
Dispersal, or the movement of an individual from its place of birth to where it reproduces, is either innate or driven by environmental changes (Caughley 1977). Relatively little is known about dispersal in Barren-ground Caribou. Immigration and emigration rates between subpopulations are pivotal to understanding the definition of and relationships among subpopulations and changes in abundance (Polfus et al. 2016).
The frequency and rate of innate dispersal in Barren-ground Caribou have not been studied in detail. Typically, juveniles and males of other species are the most likely to disperse, but for Barren-ground Caribou, information is least known for juveniles and males as it is mostly adult females that are collared and their movements tracked. Roffler et al. (2012) reported short-term breeding dispersal of a few collared males but not females between two neighbouring subpopulations in Alaska. Male dispersal likely occurs among the Canadian mainland subpopulations of Barren-ground Caribou (McFarlane et al. 2016).
Information for adult females based on satellite-collaring suggests that switches between calving grounds are infrequent. Of the 153 caribou females monitored between 1996 and 2008, only 7 (4.5%) were found on >1 calving ground (Adamczewski et al. 2009). Specifically for the Bathurst subpopulation, Boulanger et al. (2011) reported that rates of immigration to or emigration from the Bathurst calving ground were low (<5%) and that immigration rates equaled emigration rates. The low rate of switching of adult females between calving grounds may not be true dispersal as the natal calving ground is unknown and unless the female died, it is unknown whether the female switched again after the collar stopped transmitting. An exception to the low rates of switching is the Beverly subpopulation. Between 2007 and 2010, the probability of females returning to the Beverly traditional inland calving ground was 28%, similar to the 31% probability they would switch to calving along the Queen Maud Gulf (Adamczewski et al. 2015).
Annual migrations are one of the most conspicuous characteristics of Barren-ground Caribou. Migration is a directed movement toward a destination and is conventionally applied to movements of the females to and from their calving grounds. Annual ranges of Barren-ground Caribou subpopulations are usually several hundred thousand km2 in size, ranging from 5,700 km2 (Coats Island) to 460,000 km2 (Qamanirjuaq) (Figure 2). Fidelity to the pre-calving post-calving areas is relatively predictable compared to those in fall and winter (Gunn and Miller 1986).
Nagy et al. (2011) estimated that the mean annual cumulative distance travelled by Barren-ground Caribou of the Lorillard and Qamanirjuaq subpopulations was from 1500 km up to 3500 km, respectively. Bathurst caribou on average begin pre-calving migration in mid-April, with peak distances moved in mid- to late May (~14 km/day). During early (or late) snowmelt years, caribou reduced (or increased) movement rates in late May (Gunn et al. 2011a). The extent to which the caribou used stopovers (staging) during pre-calving migration is unmeasured, although it is known for other migratory deer (Sawyer and Kauffman 2011).
Fall migration is less predictable on an annual scale, with timing being affected by environmental conditions (Campbell pers. comm. 2005 in Gunn 2013).
Interspecific interactions
Barren-ground Caribou do not have obligate relationships with other species for survival, although through their migrations and sheer numbers they play a key role in the tundra and boreal forest food webs. The interactions of caribou with vegetation are not just through removal of plant biomass but also extend to the fertilizing effects of fecal pellets transferred between terrestrial systems (Zamin and Grogan 2013) and aquatic systems as caribou migrate and rest on lake ice.
Barren-ground Caribou seasonally overlap with Moose (Alces americanus) and Muskoxen(Ovibos moschatus), as well as other caribou DUs. Muskoxen have re-occupied large parts of their historical tundra ranges since the 1960s. Muskoxen were reintroduced to the Beaufort Coastal Plain in 1969 (Reynolds 1998), and now overlap with portions of the the Porcupine Caribou subpopulation’s range. More recently, they have spread further along the tree-line and penetrated the boreal forest in southwestern NWT where their distribution overlaps the winter ranges for Barren-ground Caribou (Gunn et al. 2009; Adamczewski pers. comm. 2013). Occasional Muskox sightings have been reported in northern Alberta and Saskatchewan. Studies that have described the relationship between caribou and Muskoxen on the mainland have not reported overlap in diet, especially for grasses and sedges (Gunn and Sutherland 1997a). The Porcupine Caribou subpopulation range overlaps also with Thinhorn Sheep (Ovis dalli) in the British, Richardson, and Ogilvie mountain ranges (T. Hegel, personal communication, 2014).
A large part of the annual distribution of Boreal Caribou (COSEWIC 2015) overlaps the winter ranges of Barren-ground Caribou, especially the Bluenose-West and Bluenose-East subpopulations (Gunn et al. 2004) and the historical winter ranges of Beverly, Ahiak, and Bathurst subpopulations in northern Alberta and Saskatchewan (Figure 2). The 2012 SARC assessment of Woodland (boreal) Caribou reports that about 40% of the range of Boreal Caribou overlaps the cumulative winter range of Barren-ground Caribou in NWT (SARC 2012).
Muskoxen and Moose share a variety of helminth and protozoal parasites with caribou, including gastrointestinal nematodes, lungworms, and tapeworms and protozoa such as Toxoplasma gondii and Neospora caninum that are transmitted through carnivore definitive hosts (Kutz et al. 2012). The implications of inter-specific parasite relationships/parasite-mediated competition are unknown (Hughes et al. 2009; Kutz et al. 2012), but range expansion of Winter Tick (Dermacentor albipictus) with Moose has been identified as a potential risk for Barren-ground Caribou under current climate warming scenarios (Kutz et al. 2009).
Predation
Grizzly Bear and Wolves are effective predators of all sex and age classes of caribou (Gau et al. 2002). Across NWT, between 2007 and 2010, almost 75% of Wolves had stomach contents containing 67% by weight of caribou (Davison 2015). Caribou accounted for 10-93% of the diet of Grizzly Bears on the Bathurst subpopulation’s summer range (Gau et al. 2002; see also Mowat et al. 2013). Wolverines (Gulo gulo) scavenge wolf-killed caribou (Lee 1995; van Dijk et al. 2008) and are able to kill caribou through persistent long chases (Dumond 2007). Lynx (Lynx canadensis) are occasional predators. Golden Eagles (Aquila chrysaetos) hunt and kill calves on the calving grounds west of the Coppermine River, NWT (Theberge and Nagy 2001; Nagy and Johnson 2007).
Information on trends in either relative abundance of predators or predation rates is piecemeal. Geographic trends in predator sightings during calving ground aerial surveys between 2007 and 2012 reveal proportionately higher densities of Wolves in more eastern subpopulations, and higher densities of Grizzly Bears in more western subpopulations (e.g., Poole et al. 2013; J. Williams unpubl. data). The Bluenose-East calving grounds had the highest Grizzly Bear sightings among calving grounds (Figure 11).
The trend in wolf sightings on the coastal Ahiak-Beverly calving ground was an increase as Heard (1992) reported an average of 8 wolves/100 hours compared to 17-42 wolves/100 hours 2007-2010 (Poole et al. 2013). Williams (1995) reported 25-54 wolves and 8-12 bears/100 hours for the inland Beverly calving ground in 1993 and 1994 compared to 13 wolves and 2 bears/100 hours in 1988 (Heard and Jackson 1990).
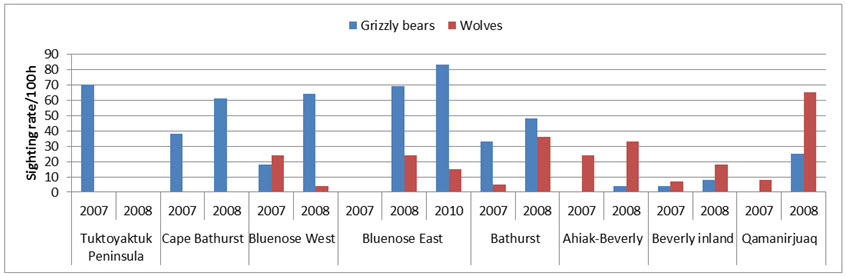
Long description for Figure 11
Chart illustrating Grizzly Bear and Wolf sighting rates during surveys of Barren-ground Caribou calving grounds in the Northwest Territories and Nunavut (2007 to 2008).
The susceptibility of Barren-ground Caribou to predation varies with seasonal distribution. When caribou congregate, such as on the calving grounds, vulnerability to predation is reduced through mutual vigilance and swamping of predators at a time when vulnerable newborn calves first emerge (Heard and Williams 1992). Calving grounds are located north of the tree-line, away from areas where breeding Wolves tend to den (Heard and Williams 1992). It is unclear how effective this is as non-breeding wolves may kill calves surplus to their immediate nutritional needs (Miller et al. 1985), although migratory caribou in general reach densities that are 1 to 2 orders of magnitude higher than non-migratory populations (Seip 1990). On the Beverly subpopulation’s calving grounds, 50-70% of calf deaths were wolf kills, although for 13% of those deaths the calves had underlying health problems such as pneumonia (Miller and Broughton1974; Miller et al. 1988). On the Porcupine subpopulation’s calving grounds, between 25 and 29% of calves died in June; 61% of identified mortality was attributed to predation by Golden Eagle, Grizzly Bear and Wolves (Griffith et al. 2002).
Predation rates have been measured through observation of marked Wolves. In the 1980s, for the Bluenose-West subpopulation the kill rate for radio-collared packs of six and seven Wolves in April 1992 was almost a caribou killed every 2 days with a higher rate during pup rearing (Clarkson and Liepins 1992). Pack size influences kill partly because it interacts with the scavenging loss to Ravens (Corvus corax); Ravens can take 75% of the edible carcass from a pair of Wolves (Kaczensky et al. 2005).
Wolf abundance indices can be approximated through sightings during aerial surveys and harvested numbers (Heard et al. 1996). Trends in wolf numbers relative to caribou numbers were tracked between 1996 and 2012 for the Bathurst subpopulation (D. Cluff, GNWT-ENR, unpubl. data). Trends for the average number of adult Wolves at den sites, the number of occupied dens in August, and average number of pups per den declined during this period.
While the Wolves may numerically respond to the abundance of caribou (Klaczek 2015), the trend for Wolves associated with the caribou on the late winter ranges or the calving grounds has not declined concomitantly with reductions in caribou abundance. Wolf sightings between 1987 and 2015 during late winter aerial surveys of the Bathurst subpopulation suggest an increasing, albeit weak, trend (Figure 12a and Figure 12b; Williams and Fournier 1996; Gunn 2013; Cluff pers. comm. 2015). For caribou from the Northern Mountain DU, predation rates interacted with climate rather than the abundance of prey (Hegel et al. 2010). Wolves can numerically respond to the abundance of caribou as pup survival is dependent on the availability of caribou during the denning period (Heard and Williams 1992; Frame et al. 2008; Klaczek 2015). However, the relationship between wolf abundance and predation rates is complicated due to overlapping caribou winter distributions between neighbouring subpopulations, which may serve to augment predation rates due to Wolves moving in from other areas. In the Porcupine Caribou range, many wolves are not migratory and for these animals moose are more likely the limiting prey item; Barren-ground Caribou provide only a brief pulse of prey (Hayes et al. 2003).
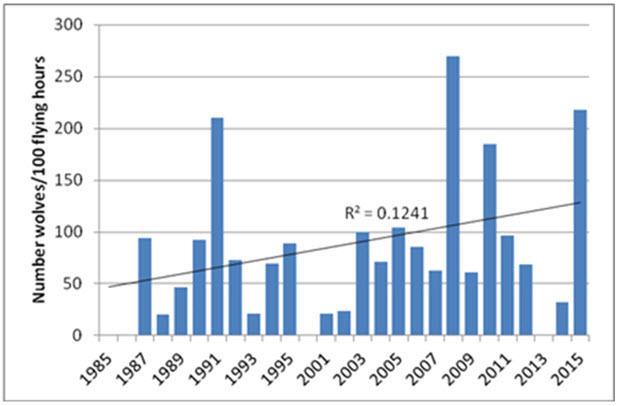
Long description for Figure 12a
Two charts (12a and 12b) illustrating sighting rates for Wolves and Grizzly Bears in the Bathurst subpopulation range of the Barren-ground Caribou. One chart records numbers of Wolves sighted per 100 hours of flying during late winter sex and age composition surveys from 1987 to 2015, and the other records numbers of Wolves and Grizzly Bears sighted per 10 hours of flying during calving ground surveys from 2006 to 2015.
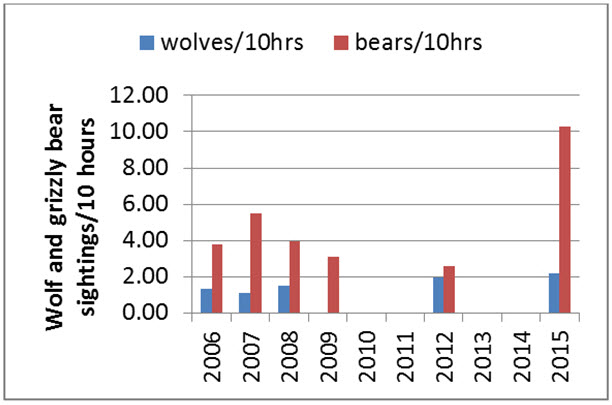
Long description for Figure 12b
Two charts (12a and 12b) illustrating sighting rates for Wolves and Grizzly Bears in the Bathurst subpopulation range of the Barren-ground Caribou. One chart records numbers of Wolves sighted per 100 hours of flying during late winter sex and age composition surveys from 1987 to 2015, and the other records numbers of Wolves and Grizzly Bears sighted per 10 hours of flying during calving ground surveys from 2006 to 2015.
The relatively long generation times of Grizzly Bears means that population responses to changes in caribou abundance will be slow. During recent decades, Grizzly Bears have expanded their distribution on the northeast tundra ranges (COSEWIC 2012), although densities are still low and no overall trend in abundance was apparent between 1991 and 2012 for NU and NWT (COSEWIC 2012). However, there are local changes, for example Grizzly Bear sightings have increased on the ranges of the Bluenose-East subpopulation over the past two decades (Dumond 2007). Sightings during surveys of Bathurst calving grounds (Figure 12) suggest more Grizzly Bears than Wolves are present.
Genetic mark and recapture data revealed that Wolverine numbers on the Bathurst subpopulation’s summer range declined approximately 11% annually between 2004–2011 (Boulanger and Mulders 2013), likely related to reduced wolf-killed caribou available for scavenging.
Parasites and diseases
Pathogens, including viruses, bacteria, helminths and protozoa, together with biting and parasitic insects, are important in caribou ecology and population dynamics (Gunn and Irvine 2003; Kutz et al. 2012,2014). These can act by causing direct mortality and/or reduced fecundity, or through having energetic costs that may also reduce survival and reproduction. Warble Flies and nasal bots, and abomasal nematodes are the most studied pathogens of caribou with respect to impacts at the individual and population level; however, other pathogens are emerging as important disease-causing agents of Barren-ground Caribou.
Warbles (Hypoderma tarandi) and nasal bots (Cephenemyia trompe) have significant energetic costs (Cuyler et al. 2012). Caribou lose substantial foraging time in avoiding the adult flies which deposit eggs on the fur (H. tarandi) or larvae in the nostrils (C. trompe) (Russell et al. 1993; Witter et al. 2012a,b). The larval stages of these parasites in caribou have metabolic costs through the immune responses and the growth of the larvae as they grow and migrate (Thomas and Kiliaan 1990; Cuyler et al. 2012). The activity levels and subsequent infection levels of warble and bot flies, and hence levels of caribou harassment, are very dependent on temperature and wind speed. This serves as the basis for the development of an index for warble fly harassment (Russell et al. 1993; Witter et al. 2012a,b). The warble index on the summer range of the Bathurst and Bluenose-East subpopulations is annually variable, but has increased to some extent between 1979-2014 as the summers became warmer, particularly after the early 1980s (Figure 13; CARMA unpubl.).
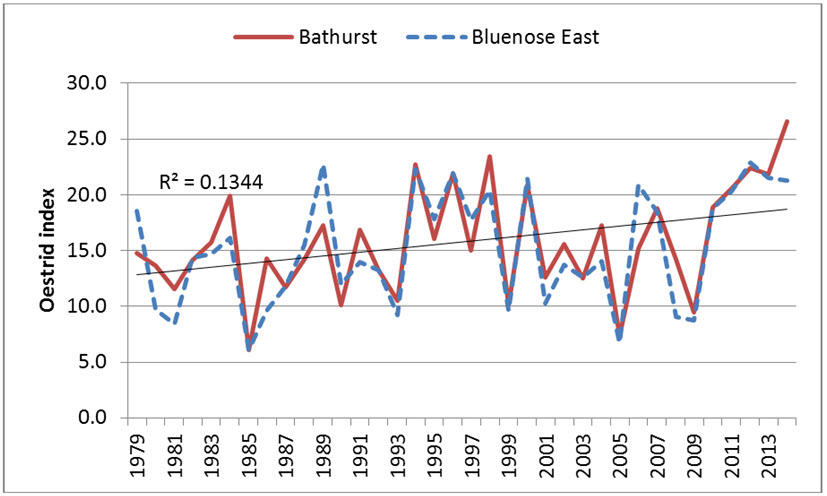
Long description for Figure 13
Chart illustrating the trend in the warble fly (oestrid) index based on daily temperature and wind speed on the summer range of the Bathurst and Bluenose-East subpopulations of the Barren-ground Caribou from 1979 to 2014.
Similarly, abomasal nematodes (primarily Ostertagia gruehneri, but also Marshallagia marshalli and Teladorsagia boreoarcticus) can reduce host body reserves and pregnancy rates with potential population level impacts (Albon et al. 2002; Hughes et al. 2009; Cuyler et al. 2012; Steele 2013). These parasites are sensitive to climate and disease ecology with impacts that are expected to increase with climate warming (Molnár et al. 2013; Kutz et al. 2014). They have direct life cycles with adult nematodes in the abomasum depositing eggs that are shed in the feces. Egg and subsequent larval development and survival to the infective third stage are temperature and humidity dependent. Third-stage larvae are ingested and migrate through the abomasal mucosa and mature to adult parasites. For O. gruehneri in Barren-ground Caribou, larvae typically overwinter and emerge the following spring (Hoar et al. 2012a). This means the lifecycle is 1-2 years, explaining the 2-year delay between host density and parasite infection intensity observed by Albon et al. (2002). Through experimental treatment of Svalbard reindeer, Albon et al. (2002) demonstrated that O. gruehneri can regulate this reindeer population by influencing pregnancy rates.
Seasonal migration by caribou from the calving grounds may reduce parasite exposure (Folstad et al. 1991; Hoar et al. 2012b). At a finer spatial scale, wild Svalbard reindeer reduced exposure by foraging away from the vicinity of fecal pellets containing gastro-intestinal nematode eggs and larvae (van der Waal et al. 2000).
Other parasites of potentially increasing significance include Besnoitia tarandi, Setaria, sp., Toxoplasma gondii and Neospora caninum. Besnoitia tarandi is a protozoan parasite that is well established across most Barren-ground Caribou subpopulations. A recent outbreak of disease causing severe debilitation and probable infertility in the Rivières Georges and Feuilles (George and Leaf River) caribou subpopulations in Quebec and Labrador highlights the potential significance of this parasite. The underlying cause of the disease outbreaks in these subpopulations is unknown and may have been associated with shifting conditions promoting transmission and/or susceptibility and the applicability to Barren-ground Caribou is unknown (Ducrocq et al. 2013).
Toxoplasma gondii and Neospora caninum are protozoan parasites that, in ungulates, can cause infertility (spontaneous abortion, mummified foetuses, fetal defects) or weak calves. They are transmitted generally through a predator-prey cycle (canid definitive hosts for Neospora, felid definitive hosts for Toxoplasma), but also may be transmitted trans-placentally. Both parasites are present in Barren-ground Caribou based on serological assays (Curry et al. 2011; Kutz et al. 2014). The abundance in caribou of these protozoa and other predator transmitted parasites, such as the tapeworms Echinococcus canadensis and Taenia spp. will be influenced by that of their carnivore definitive hosts (Rausch 2003; Kutz et al. 2012).
Foot rot, caused by an interaction between bacteria and environmental conditions, was identified as a cause of severe lameness on the late summer range of Bathurst Caribou in 2001 (Gunn et al. 2005). Foot rot is characterized by swollen feet, and is typically seen in late summer. Warm temperatures and muddy ground are favourable conditions for this soil-borne bacterium, which enters the foot through minor abrasions. This may be why sharp gravel on roads is considered to be a pre-disposing factor (Radostits et al. 2007 in Handeland et al. 2010).
The bacterial disease Brucella suis biovar 4 can cause spontaneous abortion, stillbirth, weak calves, enlarged testes, enlarged joints, lameness, and abscesses in Barren-ground Caribou (Dietrich 1981). The most recent serological survey across Canada (2007-2009) suggests prevalence is low across most subpopulations (Curry 2012). However, on Baffin Island between 1983 and 1986, sero-prevalence for brucellosis was 15% to 43% for south to north Baffin Island (217 caribou sampled) and six caribou showed clinical signs (Ferguson 1997). More recently, B. suis appears to have colonized caribou on Southampton Island, and coincided with reduced pregnancy rates and declining abundance (Campbell 2013).
The bacterium Erysipelothrix rhusiopathiae has recently been identified as one of the causes of mortality events from 2010-2015 for Muskoxen on Banks and Victoria Islands, NT and NU (Kutz et al. 2015). This bacterium is a generalist and can infect all mammal and fish, and is a known cause of mortality for Boreal Caribou. Seropositivity has been confirmed in nine Barren-ground Caribou subpopulations (Kutz pers. comm. 2013); the role in population dynamics remains undetermined.
Caribou are host to a variety of viral pathogens, but knowledge of population-level impacts is limited. In serological surveys of Barren-ground Caribou using tests developed for domestic cattle, seroprevalence to alpha herpes virus and pestiviruses was common (Curry 2012; Carlsson et al. 2015). It is likely that the herpes virus reaction is to Cervid Herpes Virus 2 (CHV2), which has been isolated from reindeer in Norway and is likely in caribou in Alaska (das Neves et al. 2010; Evans et al. 2012). This virus was identified as the primary agent in an outbreak of kerato conjunctivitis in Norwegian reindeer and experimental infections in reindeer have been linked to neonatal death and spontaneous abortion (das Neves et al. 2010). Clinical disease associated with this virus has not been observed in Barren-ground Caribou, but this is more likely due to a lack of detection as opposed to absence of disease.
Population sizes and trends
Sampling effort and methods
Three methods (Table 5) are typically used to estimate the size of Barren-ground Caribou subpopulations. Details can be found in Heard (1985) and Gunn and Russell (2008). Trends in the western subpopulations are estimated through a minimum count of caribou numbers using aerial photography of post-calving aggregations. Post-calving photo surveys were first applied to the Bluenose subpopulation in 1986 and 1987 (McLean and Russell 1992). Post-calving aggregation surveys are possible because all sex and age classes form large aggregations in late June through July in response to insect harassment (Valkenburg et al. 1985). Photography is used to minimize bias (increased accuracy). Locating post-calving aggregations depends on finding radio- or satellite-collared individuals. The precision of the survey is calculated based on the proportion of radio-collars located and is estimated using a Lincoln-Peterson estimator or the Rivest method (Rivest et al. 1998). The latter tends to produce higher estimates and associated variance because it examines how the radio-collared caribou are distributed randomly among the post-calving groups, then models how the groups and collared caribou are detected (Adamczewski et al. 2013). Precision depends on the number of collared caribou, their representativeness of the subpopulation and the search effort to locate them. Survey estimates, especially for post-calving photography, may include calves and subadults. Thus, population estimates provided in this report likely overestimate the number of mature individuals.
The second method is aerial count sampling on calving grounds. Counts are extrapolated to a subpopulation estimate (using sex and age composition) or area estimate of total numbers. Bias is reduced either through aerial transect photography or other techniques, such as double observer method to estimate bias as a correction factor (Campbell et al. 2014). Trends in numbers of caribou on calving grounds can be used as an index to trends in subpopulation size. The sample counts are based on strip transects and there have been major efforts to reduce bias (improving accuracy) and increasing precision (repeatability) of the surveys. In the last 10 years, the use of global positioning system technology has improved data recording and handling, which leads to more repeatable survey design and analyses (Nishi et al. 2010; Poole et al. 2013).
Direct comparisons of calving photo and post-calving surveys have only been carried out twice, with paired surveys of the Bluenose-East subpopulation in 2010 (Adamczewski et al. 2013) and the George River subpopulation (Eastern Migratory Caribou DU) Québec/Labrador in 1993 (Couturier et al. 1996). The June 2010 calving ground survey for the Bluenose-East subpopulation led to an estimate of 114,472 ± 6,908 (SE) caribou at least 1 year old, which was not statistically different from an estimate of 122,697 ± 16,202 (SE) from the post-calving survey in July of that year (Adamczewski et al. 2013).
The third method is strip or line transect surveys using visual counts to obtain a sample count on a predetermined geographic area. This may be an entire island (Southampton), part of a large island (Baffin), or part of the mainland where the survey boundaries are not determined by geographic features or subpopulation ranges. These surveys include both pre-calving (May) and calving (June) aerial surveys on the northeast mainland. The June surveys included techniques to reduce bias and increase precision (Campbell et al. 2014; Jenkins et al. 2012).
| Parameters | Post-calving photography |
Calving ground photography |
Calving ground visual |
Island or geographic area |
|---|---|---|---|---|
| Subpopulations | Porcupine, Cape Bathurst, Bluenose-West and Bluenose-East | Bluenose-East; Bathurst; Qamanirjuaq; Beverly | Ahiak; Lorillard, Wager Bay; Boothia | Southampton; Coats; northeast mainland; Baffin |
| Reducing Bias | Photography | Photography | None or double-observer counting | None or double-observer counting |
| Increasing precision | Dependent on number of and locating radio-collars | Coverage (no. of strip transects); stratification of survey effort | Coverage (strip or line transects); stratification of survey effort | Coverage (strip or line transects); stratification of survey effort |
| End point | Total numbers+variance (assumed for subpopulation) | 1) Breeding females+variance 2) Extrapolate to subpopulation size |
Total numbers+variance | Total numbers+variance |
There has been inconsistent sampling of population vital rates over time among subpopulations, thereby limiting the availability of current (since 2005) information for most subpopulations. Calf survival, pregnancy rates, adult survival and harvest are often reported as summaries, limiting any assessment of trends. Adult sex ratio and calf survival (reported as a ratio), can be influenced by changes in either the numerator or denominator of the ratio (Caughley 1974). Assumptions associated with these ratios are often overlooked. For example, changes in adult female survival of the Bathurst subpopulation likely magnified the calf survival estimates during a few years of low female survival (Boulanger et al. 2011).
Sampling effort to estimate abundance has been uneven since 1983 and varies among the subpopulations. Consistency in survey methods, timing and analyses vary along a gradient from Bathurst, Southampton and Porcupine subpopulations (relatively consistent techniques, timing, and analyses) to northeast mainland and Baffin Island subpopulations (few surveys, different seasons, and various methods). Ten subpopulations have a higher mean survey frequency of 4.8 years (± 0.85 SE) between surveys but the precision of the individual estimates varies widely. There has been a trend towards increased precision, as census design has been adjusted to increase both accuracy and precision (see individual subpopulation accounts).
Estimating trends is dependent on the consistency with which the survey units are defined and consistency of the application of sampling technique. For subpopulations where visual and photographic calving ground surveys were conducted, methods and areas have been relatively consistent over time. For the northeast mainland subpopulations (Appendix B), the area focus of surveys has shifted from calving areas to geographic areas, and from pre-calving to calving, which limits a comparison of estimates over time. Survey areas have remained consistent over time for Southampton Island, Coats Island, and Boothia Peninsula, although the latter two have not been counted for over two decades.
Increasing statistical power to detect trends is attained through increasing precision, more frequent surveys, and better analysis methods (Nishi et al. 2007). Information on statistical power and the likelihood of missing a decline when one has occurred (Type II error) is unevenly distributed among estimates of abundance for the subpopulations and areas surveyed, except for the Bathurst subpopulation (Nishi et al. 2010: see Subpopulation-specific trends; Appendix C.). For other subpopulations, the amount of information is limited and statistical analyses of trends are currently lacking.
The majority of the surveys used to derive total population estimate for this DU were designed to estimate a subpopulation size. The two exceptions have been area-based surveys on Baffin Island in 2014 and the northeast mainland (see Population Spatial Structure; Appendix B). Most recent estimates for some subpopulations (Lorillard/Wager Bay and Beverly/Ahiak) were combined (see Table 6), such that 13 subpopulation units were used as the basis for total population estimates. For nine of the 13 subpopulations the most recent estimate was 2013-2015 (Tables 6, 7).
Population trend analyses
Two methods were used to calculate the overall population trend for Barren-ground Caribou. The first, using the IUCN model (IUCN Method; IUCN 2016, p. 33), was premised on the two population estimates closest in time to the start and end of a 3-generation period. Where survey data did not match the start and end date of that period, the exponential rate of change was calculated and used to project the population estimates to the present (2016) or the past (1989; Generation Time = 9 years). This method assumes that the rate of change was constant between two subsequent estimates. For the second method (Exponential Method), the exponential model (Caughley 1977) was applied to all survey data to project population change over the 3-generation period. Here, a simple exponential model was parameterized using the instantaneous growth rate (r) that was iteratively recalculated with successive population estimates as:

As with the IUCN Method, this approach assumes a constant rate of change when generating a projected estimate for years with no survey data. However, the iterative calculation of r allowed for the use of all survey data that occurred within the 3-generation period, not just the end points as required by the IUCN Method.
A trend was calculated for each individual subpopulation with reliable survey estimates that approximated a 3-generation period (IUCN Method) or with at least four repeated surveys between 1987 and 2015 to allow for an iterative population projection required for the Exponential Method. Given these conditions, the overall trend estimate was based on seven of the 13 subpopulations, representing approximately 68.2% of the current total number of Barren-ground Caribou based on a sum of the most recent individual estimates (Table 8) and ca. 63% of the most recently recorded maximum (1991-1995; Table 6). The seven subpopulations used for this analysis were: Porcupine (N=8; Surveys 1987 to 2013), Cape Bathurst (N=9; Surveys 1984 to 2015), Bluenose-West (N=9; Surveys 1986 to 2015), Bluenose-East (N=6; Surveys 2000 to 2015), Bathurst (N=9; Surveys 1984 to 2015), Qamanirjuaq (N=5; Surveys 1985 to 2014), and Southampton (N=12; Surveys 1987 to 2015). The trend is not calculated for the Tuktoyaktuk Peninsula (N=5; Surveys 2005 to 2015), Beverly-Ahiak (N=1; Survey 2011), Lorillard and Wager Bay (N=2; Surveys 1995, 2002), Boothia Peninsula (N=2; Surveys 1985, 1995), and Baffin Island (N=2; Surveys 1991, 2014) subpopulation (Table 6; Appendix C).
There is some uncertainty in both the generation time for Barren-ground Caribou (see Life Cycle and Reproduction) and the precision of the survey estimates. Thus, population trend was calculated using both methods (Exponential and IUCN) and using generation times of 6, 7, 8, 9, and 10 years. A Monte Carlo simulation was then used to investigate the influence of imprecision in individual population estimates. The trend for each subpopulation was estimated 10,000 times using the Exponential Method with a generation time of 9 years. A number of the population estimates did not have a measure or comparable measure of precision; thus, the Monte Carlo simulation was parameterized using a normal distribution, the reported estimate, and a variance that was 10% of the estimate. Population change is expressed here as the simulated mean and the 95th (5th percentile population estimate in 1989 versus 95th percentile 2016) and 5th percentile (95th percentile population estimate in 1989 versus 5th percentile 2016) of the projected population estimates. The assumption is that the 95th and 5th percentiles represent the possibility of an underestimated population in the past and an overestimated population in the present, respectively. These simulated values are plausible extremes, given a 10% estimate of imprecision, and would potentially dampen a trend in decline over the 3-generation period.
Abundance
Maximum recorded overall abundance is estimated at > 2 million Barren-ground Caribou during the period 1991-1995 (Table 6), followed by a decline to just over 800,000 caribou by 2015 (Table 7). However, on one hand, these numbers overestimate mature individuals as some survey estimates included calves and non-mature adults. On the other hand, estimates were not available for some subpopulations in 1991-1995 or survey methodology was later refined. For example, there are no estimates for Baffin Island prior to 1991 and for Bluenose-East prior to 2000, and Coats Island was only surveyed once (1991) and the last survey for Boothia Peninsula took place even earlier (1995; Table 7). Tables 7, 8 and Appendix C include available details on individual subpopulations. These incorporate a relatively conservative approach by not comparing estimates from pre-calving surveys with estimates from calving surveys, as caribou distribution can change markedly in the weeks leading up to calving.
| Year | PCH | TUK | CBH | BLW | BLE | BCH | QAM | BEV/AHl | BEVl | Ahiakl | Booth | Lorillard/ Wager Bay | Coats | SCH | Baffin |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1984 | blank | blank | 13,476 | blank | blank | 384,000 | blank | blank | blank | blank | blank | blank | 2,130 | blank | blank |
| 1985 | blank | blank | blank | blank | blank | blank | 272,000 | blank | blank | blank | 4,830 | blank | blank | blank | blank |
| 1986 | blank | blank | blank | 88,369 | blank | 472,000 | blank | blank | blank | blank | blank | blank | blank | blank | blank |
| 1987 | 165,000 | blank | 12,516 | 106,887 | blank | blank | blank | blank | blank | 32,000 | blank | blank | blank | 5,400 | blank |
| 1988 | blank | blank | blank | blank | blank | blank | 221,000 | blank | 189,561 | blank | blank | blank | blank | blank | blank |
| 1989 | 178,000 | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank |
| 1990 | blank | blank | blank | blank | blank | 351,683 | blank | blank | blank | blank | blank | blank | blank | 9,000 | blank |
| 1991 | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank | 500 | 13,700 | 235,000 |
| 1992 | 160,000 | blank | 19,278 | 112,360 | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank |
| 1993 | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank |
| 1994 | 152,000 | blank | blank | blank | blank | blank | 495,665 | blank | blank | blank | blank | blank | blank | blank | blank |
| 1995 | blank | blank | blank | blank | blank | blank | blank | blank | 276,000 | blank | 6,658 | 28,336 | blank | 18,275 | blank |
| 1996 | blank | blank | blank | blank | blank | 349,046 | blank | blank | blank | 200,000 | blank | blank | blank | blank | blank |
| 1997 | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank | 30,381 | blank |
| 1998 | 129,000 | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank |
| 1999 | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank |
| 2000 | blank | blank | 11,089 | 76,376 | 104,000 | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank |
| 2001 | 123,000 | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank |
| 2002 | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank | 41,000 | blank | blank | blank |
| 2003 | blank | blank | blank | blank | blank | 186,005 | blank | blank | blank | blank | blank | blank | blank | 17,981 | blank |
| 2004 | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank |
| 2005 | blank | 2,700 | 2,434 | 20,800 | 70,081 | blank | blank | blank | blank | blank | blank | blank | blank | 20,582 | blank |
| 2006 | blank | 2,866 | 1,821 | 18,050 | 66,754 | 128,047 | blank | blank | blank | blank | blank | blank | blank | blank | blank |
| 2007 | blank | blank | blank | blank | blank | blank | blank | blank | blank | 235,000 | blank | blank | blank | 15,452 | blank |
| 2008 | blank | blank | blank | blank | blank | blank | 348,661 | blank | blank | blank | blank | blank | blank | blank | blank |
| 2009 | blank | 2,752 | 1,934 | 17,897 | blank | 31,900 | blank | blank | blank | blank | blank | blank | blank | 13,956 | blank |
| 2010 | 169,000 | blank | blank | blank | 114,472 | blank | blank | blank | blank | blank | blank | blank | blank | blank | blank |
| 2011 | blank | blank | blank | blank | blank | blank | blank | 195,529-208,230m | blank | blank | blank | blank | blank | 7,800 | blank |
| 2012 | blank | 2,192 | 2,427 | 20,465 | blank | 34,690 | blank | blank | blank | blank | blank | blank | blank | blank | blank |
| 2013 | 197,000 | blank | blank | blank | 68,300 | blank | blank | blank | blank | blank | blank | blank | blank | 7,287 | blank |
| 2014 | blank | blank | blank | blank | blank | blank | 264,661 | blank | blank | blank | blank | blank | blank | blank | 4,856 |
| 2015 | blank | 1,701 | 2,259 | 15,268 | 38,592 | 19,769 | blank | blank | blank | blank | blank | blank | blank | 12,297 | blank |
l Population structure, delineation, and estimates of Beverly and Ahiak subpopulations are subject to differing interpretations (see text and footnote 3). They are enumerated separately until 2011 and amalgamated into one unit in 2011.
m This range of estimates represents those generated from two interpretations of survey data: 1) Beverly (124,189) and Ahiak (71,340), as defined by Campbell et al. (2014), and 2) a Beverly-Ahiak unit that calves along the length of the Queen Maud Gulf coast including Adelaide Peninsula and extends to Chantrey Inlet (142,050), plus animals counted east of Chantrey Inlet (66,180), as per rationale presented in Adamczewski et al. (2015).
n 2011 estimate for Beverly-Ahiak is from Campbell et al. (2014). See Table 6 footnote no. 3 for notes on slightly elevated population estimate derived from different interpretation of survey history.)
| Subpopulation/ geographic area |
Maximum | Year | Minimum | Year | Most recent estimate | Year | No. estimates | Period years | Most recent Trend |
|---|---|---|---|---|---|---|---|---|---|
| Porcupine o | 197,000 | 2013 | 123,000 | 2001 | 197,000±28,561p | 2013 | 8 | 27 | Increasing |
| Cape Bathursto | 19,278 | 1992 | 1,821 | 2006 | 2,259±84 | 2015 | 8 | 28 | Declining |
| Bluenose-Westo | 112,360 | 1992 | 15,268 | 2015 | 15,268±1,369 | 2015 | 9 | 27 | Declining |
| Bluenose-Easto | 114,472 | 2010 | 38,592 | 2015 | 38,592±4,733 | 2015 | 7 | 17 | Declining |
| Bathursto | 472,000 | 1986 | 19,769 | 2015 | 19,769±7,420 | 2015 | 9 | 30 | Declining |
| Qamanirjuaqo | 495,000 | 1994 | 221,000 | 1988 | 264,661±44,084 | 2014 | 5 | 22 | Declining |
| Southamptono | 30,381 | 1997 | 5,400 | 1987 | 12,297±1,844 | 2015 | 11 | 26 | Increasing |
| Tuktoyaktuk Pen. | 2,866 | 2006 | 1,701 | 2015 | 1,701 | 2015 | 5 | 8 | Declining |
| Beverly/Ahiak | blank | blank | blank | blank | 195,529q | 2011 | blank | blank | Declining |
| Boothia Pen. | 6,658 | 1995 | 4,830 | 1985 | 6,658 | 1995 | 3 | 20 | Unknown |
| Lorillard + Wager Bay | 41,000 | 2002 | 28,336 | 1995 | 41,000 | 2002 | 3 | 5 | Unknown |
| Coats Island | 4,236 | 1978 | 500 | 1991 | 500 | 1991 | 2 | 11 | Unknown |
| Baffin Island | 235,000 | 1991 | 3,096 | 2014 | 4,856 | 2014 | 2 | 22 | Declining |
| [Total current estimate] | blank | blank | blank | blank | [800,090] | blank | blank | blank | blank |
o Available data sufficient to calculate three-generation population trend.
p 95% CI
q 2011 estimate for Beverly-Ahiak is from Campbell et al. (2014). See Table 6 footnote no. 3 for notes on slightly elevated population estimate derived from different interpretation of survey history.)
Fluctuations and trends
Seven of thirteen subpopulations (representing almost 70% of the total population) had sufficient survey data with which to calculate the three-generation decline; the median decline for these subpopulations was 56.8% (range = -50.8 to -59.0%) after applying the two methods for population projection (IUCN, Exponential) and five estimates of generation time (Figure 14). Four of the seven subpopulations (Cape Bathurst, Bluenose-East, Bluenose-West and Bathurst) declined by >80% during this period, one (Qamanirjuaq) had a median decline of 39.7%, driven by marked variability, whereas the remaining two (Porcupine, Southampton) increased (Figure 15). Of the six subpopulations with insufficient data to calculate trend, available survey data for three with recent surveys (Tuktoyaktuk Peninsula, Baffin, Beverly-Ahiak cluster) and representing 25.8% of the current population have indicated declines; 2014 survey estimates for Baffin Island suggest a steep decline (Tables 6, 7). The trajectories of the remaining three subpopulation units, ca. 5% of the DU, are unknown. For seven subpopulations, the most recent estimate is the same as their minimum recorded counts (Table 7).
Generation time, method and precision strongly influenced the projected trend in decline for those subpopulations that did not demonstrate a linear trajectory over the 3-generation period (Figure 15). For example, the Porcupine, Southampton and Qamanirjuaq subpopulations demonstrated relatively large decreases and increases in abundance; thus, the overall trend in decline was sensitive to the choice of generation time and imprecision in the population estimates. Southampton is a reintroduced population to an island and is consequently characterized by a steep population increase and decline. Likewise, the Qamanirjuaq subpopulation was increasing during the 1980s then declining after 1994. The other four subpopulations had a more linear trend over the 18-, 21-, 24-, 27-, or 30-year periods.
After representing uncertainty in the numerical estimates (N) for each subpopulation, the Monte Carlo simulation (using only one generation time, 9 years, and one method) also supported the conclusion of a >50% decline in the population. However, when the 5th and 95th percentiles of the population projections were applied to the calculation of percent change, the estimates ranged from -17.0 to -76.3% (simulated mean = -54.1%; Table 8). As with generation time, those subpopulations that did not demonstrate consistent linear trends were most susceptible to simulated imprecision in population estimate with variable estimates over time (i.e., Porcupine, Southampton, Qamanirjuaq).
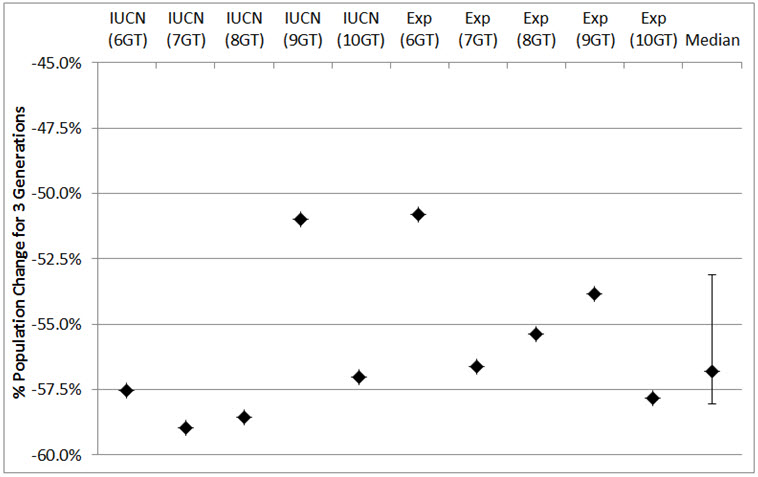
Long description for Figure 14
Chart illustrating estimated percent change in Barren-ground Caribou population size based on population estimates summed over seven subpopulations for each of two estimation methods (International Union for Conservation of Nature and Exponential) and five generation times (6, 7, 8, 9, and 10 years).
| Subpopulation | Mean % Population Change |
95% UCI | 95% LCI |
|---|---|---|---|
| Porcupine | 31% | 132% | -31% |
| Cape Bathurst | -85% | -78% | -90% |
| Bluenose-West | -87% | -81% | -92% |
| Bluenose-East | -89% | -66% | -96% |
| Bathurst | -96% | -93% | -97% |
| Southampton | 113% | 232% | 31% |
| Qamanirjuaq | -4% | 48% | -39% |
| Total Population | -54% | -17% | -76% |
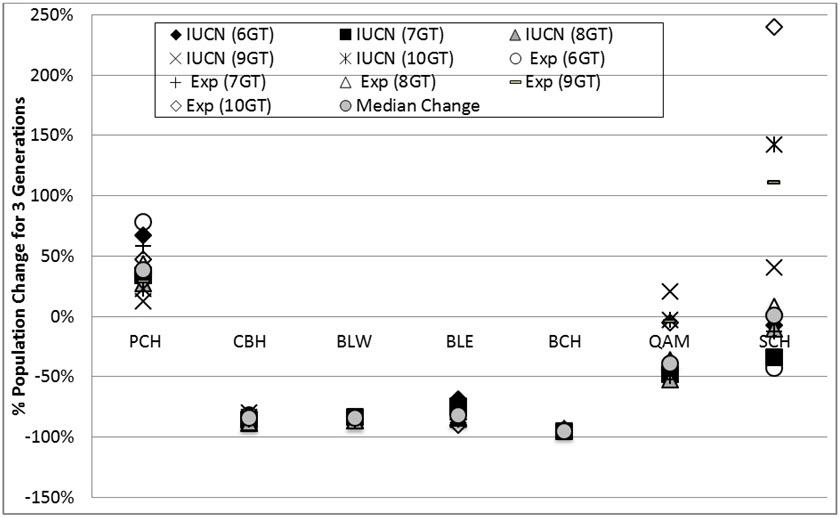
Long description for Figure 15
Chart illustrating estimated percent change in population size for each of seven subpopulations of Barren-ground Caribou for each of two estimation methods for population projection (International Union for Conservation of Nature and Exponential) and five generation times (6, 7, 8, 9, and 10 years).
There is quantitative evidence from some subpopulations that Barren-ground Caribou abundance fluctuates at relatively regular intervals on the continental mainland (Zalatan et al. 2006; Beaulieu 2012). For example, data for the Porcupine subpopulation has a very good fit to a third-order polynomial (R2 = 0.93), which suggests a cycling-type dynamic. A similar pattern is suggested for other large populations of migratory caribou (Eastern Migratory Caribou DU; Messier et al. 1988; COSEWIC, in prep.). ATK from NWT (SARC 2016) indicates fluctuations in abundance of subpopulations but variability in the length of time between times of greater abundance. A 60-80 year cycle on Baffin Island has been proposed (Ferguson et al. 1998; Baffinland 2012; Campbell et al. 2015b). Information on cyclic (regular fluctuations) abundance is from recollections of Indigenous elders (Baffinland 2012; Campbell et al. 2015b) and, for the Beverly and Bathurst subpopulations, from the cumulative frequency of hoof scars on spruce roots exposed on caribou trails in the tree-line transition zone (Zalatan et al. 2006). The scar frequency distribution for both the Bathurst and Beverly subpopulations was high during the mid-1940s and 1990s, and low during the 1920s, 1950s–1970s, and at the turn of the 21st century (Zalatan et al. 2006). However, most evidence for a small subset of Barren-ground Caribou subpopulations only indicates 1-2 cycles.
Evidence has not been compiled for any role of unpredictable stochastic events for Barren-ground Caribou. ATK observations indicate that population highs are not as high as they used to be (Nesbitt and Adamczewski 2009; WMAC (North Slope) and Aklavik HTC 2009). Population declines can be exacerbated by anthropogenic factors (Tłįchǫ Government 2007; WRRB 2010b; Soublière 2011). This includes harvest which, when occurring at constant rate in a declining subpopulation, may result in the mortality of an increasing proportion of that subpopulation (Messier et al. 1988; Adamczewski et al. 2009). As numbers become exceptionally low, susceptibility to unpredictable stochastic events, such as unusual or extreme weather events (e.g., summer heat, icing storms) or disease epidemics becomes increasingly possible, particularly if the caribou continue to aggregate in the same location at certain times of year.
Available information is insufficient to determine whether these population fluctuations meet the IUCN definition of “extreme fluctuations” (IUCN 2016).
At the subpopulation scale (with relatively consistent survey methods and survey areas), Barren-ground Caribou have declined from 3% to 97%. From among the seven subpopulations with sufficient data, only the Porcupine and Southampton of the 14 subpopulations are currently increasing. It should be noted, however, that Southampton Island is a re-introduced subpopulation without predators that declined with high harvest and disease, with the currently increasing trend explained at least in part by immigration from the mainland (see Subpopulation-specific trends).
Subpopulation-specific trends
This section draws on the published and jurisdictionally reviewed summary (Gunn et al. 2011b) produced for the Canadian Biodiversity: Ecosystem Status and Trends 2010 (Federal, Provincial and Territorial Governments of Canada 2010) as well as more updated reports (see Appendix C). The emphasis is on trends in abundance and on vital rates such as adult and calf survival, where information is available. Subpopulation information is presented here from west to east.
Porcupine
a) Trends in subpopulation size
The trend for the Porcupine subpopulation has been estimated from photographing post-calving aggregations since 1972 (Caikoski 2011). The Porcupine subpopulation increased from an estimated 100,000 in 1972 to 178,000 in 1989 at an estimated 0.04 exponential rate of increase (references in Caikoski 2011). After 1989, the subpopulation declined at -0.03 exponential rate of decline, dropping to 123,000 by 2001. A combination of poor weather and lack of post-calving aggregations prevented another estimate until 2010 when the population was estimated at 169,000 (153,493-184,403 95% CI) (Caikoski 2011). A subsequent survey in 2013 indicated that the subpopulation was 197,000 (168,667–225,789 95% CI) (Caikoski 2015), the highest value since standardized population estimates began in the early 1970s. The three-generation trend for the Porcupine subpopulation is estimated at +31%, albeit with a wide confidence interval (-31-132% 95% CI) (Table 8).
b) Trends in vital rates
A large sample of collared females (average 66 + 15.4 relocated on calving grounds) was used annually to estimate calving location, parturition rate, and early calf survival (Caikoski 2011). Between 2002 and 2010, the number of collared females used to calculate mortality averaged 97 + 6.7. Collared females were located within a week of peak calving to determine pregnancy status based on extended udders, presence of antlers or accompanied calf. Between 1987 and 2012, the proportion of females giving birth varied between 0.64 – 0.90, averaging 0.81. Similar values were reported in the decline phase (0.81) and the increase phase (0.81). Early calf survival (averaging 0.72) was higher during the decline phase (0.74) than during the increase phase (0.70). Spring composition counts provided recruitment estimates that were higher in the decline (36 calves per 100 females) than during the increase (34 calves per 100 females). However, because adult female survival varied between phases (violating the assumption of constant survival), these data are not comparable. Further, estimates made between 1987 and 1989, when the subpopulation approached peak numbers were 0.87 – higher than during the decline phase.
Tuktoyaktuk peninsula
a) Trends in subpopulation size
In 2005, hunters reported caribou on the Tuktoyaktuk Peninsula after a resident private herd of reindeer had been moved away in about 2001. Systematic aerial counts started in September 2005 when 2,700 caribou (including calves) were estimated; about 20% of these were reindeer based on their appearance (Branigan 2005). In March 2006, 26 caribou, including 19 females, were fitted with satellite-collars (Nagy and Johnson 2006). These caribou revealed movements that appeared to be restricted to the upper peninsula and since then, there have been four post-calving surveys (Appendix C), with a most recent (2015) estimate of 1,701 animals (T. Davison, pers. comm. 2016).
The trend in late winter recruitment, as indexed by calf:adult female ratios for 2007 to 2015, suggests a low of 30 calves:100 females in 2007, followed by consecutive years of higher calf:female ratios during 2008-2015 (41-52:100) (Davison and Branigan 2011; Davison pers. comm. 2016).
Cape Bathurst
a) Trends in subpopulation size
Cape Bathurst was first surveyed as a distinct subpopulation in 1986, having been considered part of the currently known Bluenose-East and Bluenose-West subpopulations (Nagy et al. 2009). Cape Bathurst subpopulation peaked at 19,300 (± 5,397 95% CI) in 1992, then declined to 11,089 ± 1,756 in 2000 and 2,434 ± 257 in 2005. The decline between 1992 and 2006 was about 85%; the subpopulation stabilized at low numbers with no statistical trend between 2006 and 2015 (Nagy and Johnson 2006; Nagy 2009a; Davison et al. 2014; ENR 2014); the most recent estimate was 2,259 ± 84 in 2015 (Davison 2015; Davison pers. comm. 2016).
The calving grounds for the Cape Bathurst subpopulation were known since the 1970s and were counted using visual stratified surveys but the estimates combined the Cape Bathurst, Bluenose-West and Bluenose-East subpopulations. In 1987, methods changed to post-calving aggregation photography and Nagy (2009a) and Nagy et al. (2009) re-analyzed the 1987 and 1992 surveys to calculate population-specific counts. The three-generation trend for the Cape Bathurst subpopulation is estimated at -85% (-78% - -90% 95% CI) (Table 8).
b) Trends in vital rates
Pregnancy rates from Cape Bathurst caribou sampled in 1995 were high (96%; n = 47 adult females; Larter and Nagy 1996). Calf:adult female ratios after calving were variable over time (Table 9). Calf:female ratios in late winter were low in 2007 (22 calves:100 females), higher for 2008-11 (42-49:100 females), low in 2013 (26 calves:100 females) and high (47.8 ± 2.1 in 2014 (Davison et al. 2015; Davison pers. comm. 2016).
Adult survival is unrecorded, although total harvest rates were high during the decline: for example, 730 caribou mostly females were harvested in 2005 (J. Nagy unpubl. data). Non-aboriginal harvest limits were implemented in 2006, and then in 2007 was closed (Davison 2014).
| Year | Cape Bathurst Post-calving Calves: 100 females Mean ± SE |
Cape Bathurst Late winter Calves: 100 females Mean ± SE |
Bluenose-West Post-calving Calves: 100 females Mean± SE |
Bluenose-West Late winter Calves: 100 females Mean±SE |
|---|---|---|---|---|
| 2000 | 64.4r | 63.5 | 38.7r | 39.8 |
| 2001 | 19.2r ± 8.7 | blank | 54.1r ± 1.76 | blank |
| 2002 | 32.3s ± 5.0 | blank | 53.7s ± 2.3 | blank |
| 2003 | 47.0s ±3.8 | blank | 53.2s ± 1.6 | blank |
| 2004 | 46.5s ±17.6 | blank | 60.9s ± 1.8 | blank |
| 2005 | 52.6r ± 5.5 | blank | 59.4s ± 1.4 | blank |
| 2006 | 32.9r ±7.0 | blank | blank | blank |
| 2007 | 52.6t ±1.6 | 21.8 ± 3.1 | 77.4t ± 7.3 | 25.7 ± 2.8 |
| 2008 | 49.3t ±1.0 | 49.0 ± 3.6 | 59.6t ±1.9 | 41.9 ± 1.4 |
| 2009 | blank | 41.9 ± 3.6 | blank | 43.7 ± 2.3 |
| 2010 | blank | 48.1 ± 4.1 | blank | blank |
| 2011 | blank | 47.4 ± 2.8 | blank | 32.0 |
| 2012 | blank | blank | blank | blank |
| 2013 | blank | 25.9 ± 3.4 | blank | blank |
| 2014 | blank | 47.8 ± 2.1 | blank | blank |
r mid- June;
s late June;
t July
Bluenose-west
a) Trends in subpopulation size
Similar to the Cape Bathurst subpopulation, the Bluenose-West subpopulation was first surveyed in 1986 at about 88,000 individuals (Nagy 2009a). The trends are also similar with numbers likely having peaked in 1992, based on a count of 112,360 (± 25,566 95% CI) caribou, then declining to low levels over the past decade (Nagy and Johnson 2006; Nagy 2009a; Davison et al. 2014; Davison pers. comm. 2016). Numbers remained at about 18,000 to 20,000 for 2005 to 2012 before declining to 15,268 ± 1,369 by 2015 (Davison pers. comm. 2016). The three-generation trend for the Bluenose-West subpopulation is estimated at -87% (-81% - -92% 95% CI) (Table 8).
b) Trends in vital rates
Mid- to late June calf ratios for the Bluenose-West subpopulation were low in 2000 and higher in 2001-08 (Table 9). Late winter calf survival was high in 2000 (40 calves:100 females) and low in 2007 (26 calves:100 females). Calf survival increased in 2008 and 2009 to 42-44 calves:100 females and declined in 2011 to 32 calves:100 females (ENR/ACCWM 2014).
Bluenose-east
a) Trends in subpopulation size
Bluenose-East was first recognized as a distinct subpopulation in 1999, having been included with the Cape Bathurst and Bluenose-West subpopulations prior to that year (Nagy 2009b). The peak estimate was 104,000 (± 22,100 95% CI) from the first post-calving photographic survey in 2000 (Patterson et al. 2004). By 2005, numbers had declined to an estimated 70,081 (± 8,120) in 2005 and 65,119 (± 3,504) in 2006 (Nagy and Tracz 2006; Nagy et al. 2008). In 2010 a calving ground photographic survey resulted in an estimate of 114,472 ± 6,908 SE (Adamczewski et al. 2013), but after 2010, numbers declined again. In 2013, a calving ground survey estimated 68,295 ± 7,610 SE (Davison 2014) followed by an estimate of 38,592 ± 4,733 (95% CI) in 2015 (Boulanger 2015). This represented a -0.29 exponential rate of decline between 2013 and 2015. The three-generation trend for the Bluenose-East subpopulation is estimated at -89% (-66%- -96% 95% CI) (Table 8).
b) Trends in vital rates
Adult survival is not measured directly from collar loss but is estimated from population models (ENR 2014). The limited information on pregnancy rates from hunter samples and serum samples from females from the Bluenose-East subpopulation captured for collaring was relatively inconsistent: hunter samples suggested reduced pregnancy rate (65%) in 2010, while both methods suggested a low rate (69%) in 2012 (ENR 2012). Late winter calf:female ratios were low at 25:100 females in 2001, high at 38–52 calves:100 females between 2004 and 2011, and low again in 2012, 2014 and 2015 at 27:100 females, 30:100 females and 21:100 females (Cluff et al. In prep.). The sex ratio in 2009 was 43 males:100 females and 42.9 males:100 in 2013 (ENR 2014).
Bathurst
a) Trends in subpopulation size
Between 1986 and 2009 the Bathurst subpopulation declined by 93% from a peak of 472,000 (± 72,900 SE) caribou in 1986 to 31,982 (± 5,306 SE) in 2009 (Heard and Williams 1991; Boulanger et al. 2015). The 2012 census estimated 34,690 (± 4,691 SE), which suggested stability although the number of breeding females slightly declined (Boulanger et al. 2015). The June calving ground photographic survey in 2015 estimated 19,769 ± 7,420 caribou and annual decline of 23% breeding females from 2012 to 2015 (Boulanger et al. 2015). The three-generation trend for the Bathurst subpopulation is estimated at -96% (-93% - -97% 95% CI) (Table 8).
The estimates of Bathurst caribou are extrapolations from photographic estimates of the number of breeding females on the calving grounds (Nishi et al. 2010; Boulanger et al. 2011, 2014a,b; 2015). While the precision of the estimates varies, the six surveys that were available were sufficient to derive an overall trend using weighted least squares regression and Monte Carlo simulation to estimate the overall variance in trend. The decline in number of breeding females on the calving ground was non-linear and was increasingly negative after 2003: between 2006 and 2009, the rate of decline was equivalent to a halving time of ca. 1.7 year (Figure 16). The 2012 census found numbers of breeding females had stabilized or slightly declined at 15,935 (95% CI 13,009-18,861; Boulanger et al. in press). However, by 2015, the number of breeding females had sharply declined to 8,075 ± 3,467 (95% CI) (Boulanger 2015).
b) Trends in vital rates
Fragmentary information for the Bathurst subpopulation prevents describing trend in pregnancy rates. A sample of 150 females collected from hunter samples in winter 2005 showed that the proportion of pregnant females was 63% (Gunn 2013). In March 2008 and April 2009, 26 of 26 adult females (100%) and 25 of 28 females (89%) were pregnant during collections (Adamczewski et al. 2009).
Another measure of productivity is calf:female ratio measured during the years when the number of breeding females is estimated on the calving ground. The ratios of calves to total females declined after 2006 (Table 10). The declining ratios since 2006 must be interpreted cautiously because during this same period adult female survival declined, violating the assumption of constancy.
Between 1985 and 1995, mean calf survival (as indexed by late winter calf:female ratios) varied annually, but between 2001 and 2004 survival declined by almost 50% vs. 1991-1995 (Figure 17; Gunn et al. 2005). Based on fall composition surveys in late October 2000, 2001, and 2004, calf survival was lower during the summer than winter (Gunn et al. 2005). Subsequent changes in calf survival from 2006 to 2009 were relatively low, which may have been affected by the declining adult survival (Boulanger et al. 2011). Calf:female ratios (Figure 17) increased 2009-2011 (45±0.03 SE) but was low again in 2012 (25:100), 2014 (32:100) and 2015 (24:100).
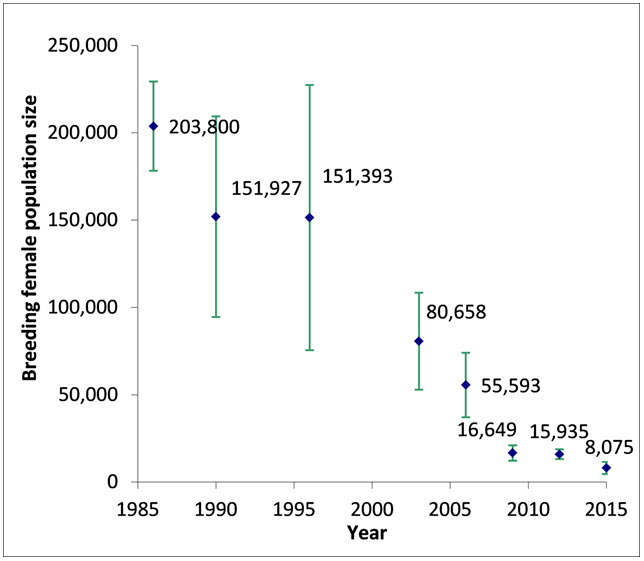
Long description for Figure 16
Chart illustrating the trend in numbers of breeding females in the Bathurst subpopulation of the Barren-ground Caribou, from 1986 to 2015.
| Year | Breeding females | Non-breeding | Total females | Calves | Calves: 100 females |
|---|---|---|---|---|---|
| 1986 | 670 | 157 | 827 | 604 | 73 |
| 1990 | 847 | 158 | 1,005 | 634 | 63 |
| 1996 | 3,273 | 467 | 3,740 | 2,954 | 79 |
| 2003 | 4,016 | 600 | 4,616 | 3,412 | 74 |
| 2006 | 4,373 | 1348 | 5,721 | 2,878 | 50 |
| 2009 | 2,033 | 417 | 2,450 | 1,528 | 62 |
| 2012 | 3,563 | 843 | 4,406 | 2,595 | 59 |
| 2015 | 851 | 587 | 1,438 | 495 | 34 |
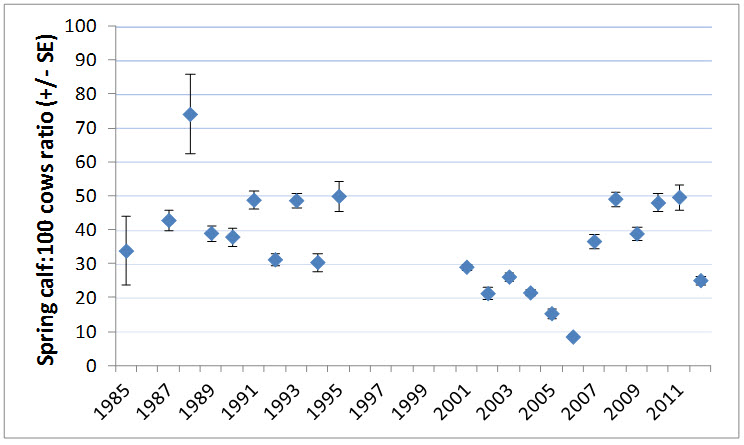
Long description for Figure 17
Chart illustrating the trend in late winter calf to 100 female ratios and standard error for the Bathurst subpopulation of the Barren-ground Caribou, from 1985 to 2012.
Trends in adult survival could not be directly measured because the sample size of satellite-collared females was low, resulting in imprecise estimates of adult female mortality. Demographic modelling using calf survival and subpopulation size suggested that adult female annual survival declined from 86% in 1985 to 76% in 2006 and 78% for 2009 (Boulanger et al. 2011). More recent estimates following harvest restrictions in 2010 were 78% for 2012 and 77% for 2015 (GNWT 2016).
Sample data from 1992 and again 2008 suggested that the age structure had shifted toward more females in the older age classes but sample sizes were small. Demographic modelling suggested that the possible trend toward an older age structure was a likely factor in the later (2006-09) stages of the subpopulation decline (Boulanger et al. 2011).
Beverly and Ahiak
a) Trends in subpopulation size
The Beverly and Ahiak subpopulations are included together when considering population estimates, as there are two interpretations about recent trends in their distributions and abundances (see Subpopulations).
The Beverly subpopulation was previously recognized based on its traditional inland calving ground south of Garry Lake, documented since the 1950s (Thomas 1969; Gunn and Sutherland 1997b; BQCMB 2014a; Adamczewski et al. 2015). The Ahiak subpopulation was described based on information about calving distribution along the Queen Maud Gulf coast from the 1940s to the present (Gunn et al. 2000a, 2013a; Campbell et al. 2014). Systematic aerial surveys using visual counts (termed aerial reconnaissance surveys), flown in 2007-2010, showed that the use of the inland calving ground by caribou declined sharply between 1994 and 2007, while the use of the coastal calving ground, along the Queen Maud Gulf coast, persisted (Johnson et al. 2008, 2009; Johnson and Williams 2008).
The end result of one interpretation of these events (Nagy et al. 2011; Campbell et al. 2014) is that there are currently two distinct subpopulations – Beverly and Ahiak. This interpretation splits the current calving distribution and assigns the western part of the Queen Maud Gulf calving area to the Beverly subpopulation and the eastern part (Adelaide Peninsula to and areas extended east to Simpson Peninsula) to the Ahiak subpopulation (Campbell et al. 2014). In contrast, Gunn et al. (2012, 2013a) and Adamczewski et al. (2015) have concluded that the Beverly subpopulation has disappeared and the Ahiak subpopulation (including individuals from the former Beverly subpopulation) calves along the western Queen Maud Gulf coast extending east only to Chantrey Inlet, with animals located east of this point belonging to two or three other subpopulations (Gunn et al. 2000a; Figure 18).
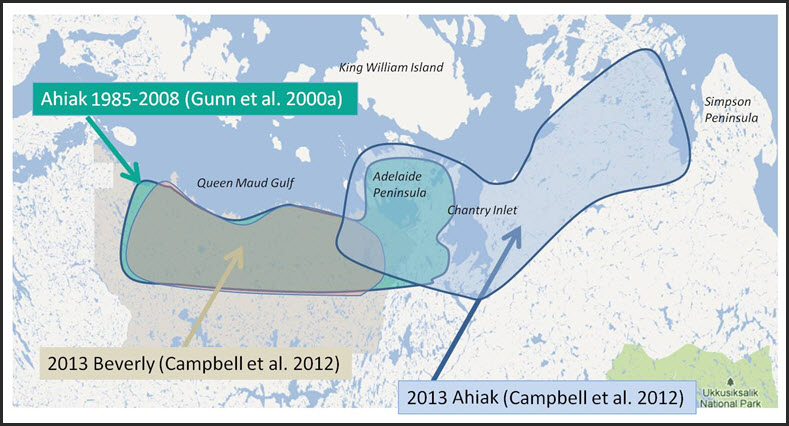
Long description for Figure 18
Schematic figure showing the relative location of calving along the Queen Maud Gulf coast for the Beverly and Ahiak subpopulations of the Barren-ground Caribou.
Accordingly, there are two different interpretations of trends in abundance depending on how the Beverly and Ahiak subpopulations are defined. These different subpopulation delineations, combined with infrequent survey history, complicate the ability to establish trends for either. However, when the two groups are considered together, the overall population estimate and recent trend are similar between the two interpretations (see Table 6 footnotes).
Between 1984-1994 (3 generations prior to the present) the Beverly subpopulation, which was known to use one inland calving ground, was relatively stable; in 1994 it was estimated at 276,000 (± 106,600; Williams 1995). Between 1994 and 2007 the subpopulation declined steeply; 2007-2010 surveys of its inland calving ground had fallen to levels too low to produce estimates for the population unit (Johnson et al. 2009). During this period there was an unknown level of emigration to the coastal calving ground. One interpretation asserts that emigration from the inland calving ground to the coastal calving ground began in the mid-1990s and continued to at least 2009 (Nagy et al. 2011). An alternative explanation is that switching occurred later, based on the finding that nine of 21 collared females (43%) that calved on the traditional inland Beverly calving ground moved to the Queen Maud Gulf coast between 2007 and 2010 (Adamczewski et al. 2015).
Campbell et al. (2014) reported a 2011 population estimate of approximately 124,000 caribou for the Beverly subpopulation, considered to be using the western-central coastal calving ground (Nagy et al. 2011). Campbell et al. (2014: page 97) did not measure the trend in their summary report but stated that the 2011 estimate “was at the lower end of the known population size range of the Beverly subpopulation”. In 2011, no females were calving on the traditional inland calving ground (Nagy and Campbell 2012). This raises the possibility that either the Beverly subpopulation has effectively disappeared (Adamczewski et al. 2015) or that the Beverly subpopulation has declined but also moved its calving ground location further north to the coast (Campbell et al. 2014). A decline (albeit not possible to quantify) is consistent with both perspectives.
For the Ahiak subpopulation, successive surveys changed in coverage and effort. As with the Beverly subpopulation, there are two interpretations of what constitute the Ahiak calving ground: the entire Queen Maud Gulf area or only the eastern section (Figure 18).
The first systematic (stratified visual) calving survey for the Ahiak subpopulation was in 1986. It yielded an estimate of 11,265 ± 1,615 (SE) breeding female caribou on the calving ground (Gunn et al. 2000b). The calving ground was a relatively small area west of Adelaide Peninsula with clearly delineated boundaries. A 1996 survey area was more extensive, with an estimated 83,134 ± 5,298 (SE) breeding female caribou on the calving ground (Gunn et al. 2000b). The entire Ahiak subpopulation estimate was extrapolated to approximately 200,000 caribou based on that latter sample (Gunn et al. 2000b). The next surveys did not occur until 2006 - 2010, when reconnaissance surveys were conducted along the coastal Queen Maud Gulf, including the Adelaide Peninsula. The observed numbers during the 2006-2010 calving surveys of the coastal calving ground were designed to map distribution and relative densities. The density was extrapolated to estimate 235,000 adult caribou in 2007 but coverage was low ( ~7%) and it was concluded that any estimates would be relatively imprecise and lack power to detect trends (Johnson et al. 2009; GNWT unpubl. data).
Campbell et al. (2012) conducted an extensive survey of the central Queen Maud Gulf area to the northeast mainland in June 2011. They used the survey results to estimate the number of adult caribou and yearlings part of the Ahiak subpopulation (as defined by Campbell et al. 2014) at 71,340 ± 3,882 (SE). However, their survey area included Adelaide Peninsula and extended east to Simpson Peninsula, which is considerably larger than previous Ahiak surveys, complicating the ability to derive a trend between successive estimates.
A trend may be inferred from calving densities, which have been consistently measured for adult caribou during aerial surveys in June (see Sampling Effort and Methods). The density increased threefold between 1986 and 1996 in addition to a large western extension of the calving distribution (Gunn et al. 2013a). Relatively stable densities between 2006 and 2011 (Johnson et al. 2009; Campbell et al. 2014) were observed, but during that period an unknown portion of the Beverly females previously calving on the inland grounds moved to calve further north along the coast (see above).
b) Trends in vital rates
Trends in vital rates are difficult to establish as there were two ‘pulses’ of sample frequency. Monitoring was regular and detailed in the 1980s, almost non-existent in the 1990s to mid-2000s, and some data were collected from 2006 onwards.
Pregnancy and productivity
Trends in pregnancy rates are based on annual monitoring for 1980 to 1987, after which monitoring became infrequent. In addition, methods changed from examination of harvested caribou to blood serum progesterone levels in adult females captured to fit satellite collars. The overall pregnancy rate was 87% based on harvested caribou from 1980 to 1987, with annual pregnancy rates in females at least 4 years old annually varying from low averages of 76–78% to highs of 98–100% (Thomas and Barry 1990a). In March 2006, 70% of the adult females fitted with satellite collars on the winter range were pregnant based on progesterone levels from serum samples collected in March (Johnson et al. 2009). However, in April 2008, 43% of 30 captured females were pregnant (Johnson and Williams 2008) and in March 2012, the pregnancy rate was 56% (Williams pers. comm. 2013). Productivity, as indexed by calf:female ratios on the calving ground, declined between 1988 and 2009.
Calf survival and recruitment
The ratio of calves to females during sex and age composition surveys on late winter ranges suggests an increasing trend during 2008-2011, but the apparent trend may also reflect the relatively low survival of adult females. Fall (late October) sex ratios for the mixed Beverly and Ahiak subpopulations were 54 males:100 females (3.6 SE; 3,772 caribou and 41 groups classified) in 2009 and 69 males:100 females (2.4 SE; 12,4221 caribou and 252 groups classified) in 2010 (GNWT unpubl. data).
Age structure and adult survival
For the period 1980-87, age-specific death rates were estimated for the Beverly subpopulation using a large sample of harvested caribou to construct a life-table (Thomas and Barry 1990b). Mortality increased progressively from 10.6% between age 2 and 3 years, 11.3% from 3 to 4 years of age, to 22.4% between age 10 and 11 years of age during a time when the subpopulation was considered stable in numbers. This analysis of age structure also showed the importance of middle-aged females as the age-specific fecundity rates indicated that 54% of all calves born were from females aged 3–6 years. By 2007-2009, Boulanger (in Adamczewski et al. 2015) estimated that for satellite-collared females with at least 1 year of calving on the traditional Beverly calving grounds, survival was low at 0.58 (CI = 0.42-0.72) and was less than the 0.79 (CI = 0.67-0.88) survival rate of Ahiak females for the same period.
Qamanirjuaq
a) Trends in subpopulation size
The Qamanirjuaq subpopulation was first surveyed in the 1960s when numbers were low. The increase that had begun in the 1970s continued through the 1980s until 1994 (495,665 [± 105,426]; CV 21%). The estimates in 1983, 1985, and 1988 had CVs ranging from 26-52%, and indicated 230,000-272,000 caribou (summarized in Campbell et al. 2010, 2015b). In June 2008, the estimate was 348,661 (± 44,861 SE) caribou (Campbell et al. 2010) and 264,718 (95% CI=44,084) in 2014, revealing a significant 23% decline between 2008 and 2014 and a 47% decline since the 1994 peak (Campbell et al. 2015a).
b) Trends in vital rates
The spring female:calf ratios declined between 1994 and at least 2008, which supports the likely decline suggested by the 1994, 2008 and 2014 estimates of abundance (Campbell et al. 2010, 2015a). Trends in adult survival are mostly unreported although for 1993-2003 the annual survival rate for the Qamanirjuaq satellite-collared females was 0.79±0.043 SE during the time period when abundance was starting to decline (Boulanger et al. 2003).
Southampton Island
a) Trends in subpopulation size
Caribou numbers increased following the re-introduction of 48 caribou from nearby Coats Island in 1968: prior to the re-introduction, the last wolf sighting was 1937 and the last caribou had died in 1957 (Heard and Ouellet 1994). The estimates are based on stratified strip surveys Campbell and Boulanger (2016). Caribou numbers reached a peak of 30,381 (± 3,982 SE) in 1997. By 2003, the subpopulation declined to 17,981 (± 2,127 95% CI) before stabilizing at 15,452 (± 1,858 95% CI) in 2007; it declined further to 7,287 (± 1,045 95% CI) in 2013 (Campbell 2006, 2013). The most recent survey in May 2015 estimated 12,297 (95% CI=1,844), a 0.262 exponential rate of increase since the previous survey. This, however, may reflect immigration of individuals from the Nunavut mainland, as indicated by multiple reports of tracks on the sea ice in winter 2013-2014 (Campbell and Boulanger 2016). The three-generation trend for the Southampton subpopulation is estimated at +113% (31% - 232% 95% CI).
b) Trends in vital rates
Southampton is a large and predator-free island (43,000 km2). Monitoring vital rates is largely based on sampling caribou for fat indicators, diet, pregnancy, age (tooth cementum) and diseases and parasites during commercial harvesting. Campbell (2006, 2013) summarized a trend in body condition during a population decline between 1995 and 1998, then an increase by 2000. Fall icing in 1998, 2005 and 2010 may have influenced poor body condition (Campbell 2006) and caused deaths in 2010-2011 winter (Coral Harbour HTO & GN 2013).
Southampton caribou are unusual among migratory tundra caribou as an outbreak of a disease, Brucella suis, affected reproduction and population dynamics. Brucellosis was unrecorded until 2000 when a 1.7% prevalence was found during commercial harvests. The prevalence increased to 51.5% in 2006 and then declined to 37% by 2011 (Campbell and Boulanger 2016). The brucellosis likely contributed to trends in pregnancy rates which were high (80%) in 1997; declined to 60% in 2003 and 36.3% in 2008, 55.6% in 2010 and 37.0% in 2011 (Campbell 2013). The low ratio of prime males (12% prime males and 20% young males in April 2005; Campbell 2006) also likely contributed to the low pregnancy rates.
Coats Island
On Coats Island (5,600 km2), caribou numbers have fluctuated, with two die-offs recorded between 1961 and 1991 (Gates et al. 1986; Ouellet et al. 1996). In winters of 1974-75, and 1978-79, the rate of die-off was about 70%, and 50%, respectively. The estimate in June 1984 was 2,130 ± 228 (SE) (Gates et al. 1986). Although a 20% systematic survey was flown in 1991, an estimate was not provided; however, based on a figure of population density, it would have been roughly 500 caribou (Fig. 1 in Ouellet et al. 1996). The overall trend in numbers from the mid-1970s to 1991 was declining abundance on Coats Island, but there is no information on the status of this subpopulation since this time. There is no information on trends in vital rates.
Northeast mainland (including wager bay and lorillard)
The northeast mainland was initially defined as a geographic survey area in 1983 to encompass several known calving areas (Wager Bay, Lorillard and South Melville as well as areas that had not been previously surveyed; Heard et al. 1986; see Appendix B). Subsequent surveys have been infrequent and have varied in timing and geography of survey areas. Currently, the area surveyed as northeast mainland in 1983 and 1995 overlaps with the Wager Bay, i.e., the Ahiak population (as defined in Campbell et al. 2014) and Lorillard clusters from the limited telemetry (Nagy and Campbell 2012). Calving areas are not resolved.
a) Trends in subpopulation size
The entire northeast mainland was aerially surveyed during pre-calving, early in May 1983 and the methods and survey area were repeated in late May 1995 (Figure 19; Heard et al. 1986, Buckland et al. 2000).
Based on the two pre-calving surveys (from the Queen Maud Gulf coast and east, and from Chesterfield Inlet and north), abundance declined from a May 1983 peak of 120,000 (± 13,900 SE) caribou to 72,395 (± 7,857 SE) in May 1995 (Heard et al. 1986; Buckland et al. 2000). The 1983 and 1995 estimates were based on stratified aerial strip transect surveys, using identical survey areas and methods (Buckland et al. 2000). The decline was unevenly distributed, with essentially no change in the western survey area (Queen Maud Gulf coast and Adelaide Peninsula) and a 66% reduction in abundance north of Wager Bay and Melville Peninsula (Buckland et al. 2000).
Part of the pre-calving area was surveyed in 2010 and 2011 (Campbell et al. 2014). The trend was a 38% increase between May 1995 and June 2011 (71,340 ± 3,882 SE). However, uncertainty is high because the comparison is between a pre-calving and a calving survey and the survey areas incompletely overlapped.
Long description for Figure 19
Schematic illustrating the relationship between the pre-calving 1983 and 1995 survey areas and the June 1999 to 2004 and June 2011 survey areas.
The pre-calving surveys in 1983 and 1995 detected a 66% reduction between 1983 and 1995 in abundance north of Wager Bay and on Melville Peninsula (Heard et al. 1986; Buckland et al. 2000). Subsequently, two calving areas (Wager Bay and Lorillard) were surveyed in alternate years during calving between 1999 and 2004 (Campbell 2005). The resulting estimates based on aerial strip transect surveys were imprecise (CV 15-52%). Averaging the three estimates for each of the two calving areas (1999-2004) suggests that 41,000 (± 8,200 SE) caribou used both calving areas. The overlap between the pre-calving (1995; Buckland et al. 2000) and mean calving (1999-2004) estimates (Campbell 2005) suggests that, between these years, the trend was stable although with a high degree of uncertainty.
Part of the 1983 and 1995 pre-calving area was subsequently surveyed during calving surveys in 2010 and 2011 (Adelaide Peninsula and east of Chantrey Inlet east to and including the Simpson Peninsula). Previously in the 1990s, aerial surveys and satellite collars had supported the identification of Arrowsmith Lowlands, Keith Bay, Simpson Lake and Simpson Peninsula calving grounds. However, in 2010 and 2011, Campbell et al. (2014) did not distinguish these four calving grounds and included them in the Ahiak subpopulation estimated at 71,340 ± 3,882 SE (see above). This is an increase since May 1995 (44,100 ± 8,080) for a mostly overlapping survey area but uncertainty is high because the comparison is between a pre-calving survey and a calving survey 16 years apart.
Boothia Peninsula
A systematic strip transect survey in June 1985 of Boothia Peninsula had a coverage of 10-20% and estimated 4,830 (± 540 SE) caribou, representing a tripling since the mid-1970s (Gunn and Ashevak 1990). The 1985 survey was repeated in July 1995, estimating 6,658 (± 1,728 SE) individuals (Gunn and Dragon 1998). A survey was flown in June 2006 (Dumond 2006), although the results are unavailable.
Baffin Island
a) Trends in subpopulation size
Between 1991 and 2014, caribou on Baffin Island declined by 98% but there is considerable uncertainty in the population estimates and resulting trend. While there is information on relative trends (Ferguson et al. 1998; Baffinland 2012), estimates of abundance are restricted to relatively small survey areas across south and north Baffin Island (507,451 km2). Ferguson and Gauthier (1992) used those surveys and expert knowledge to suggest that in 1991 caribou abundance was stable, with 60,000-180,000 caribou in South Baffin, 50,000-160,000 caribou in North Baffin, and >10,000 caribou in the northeastern portion of the island.
Jenkins and Goorts (2011) flew a systematic reconnaissance survey and counted 47 and 119 caribou during 4,587 and 7,186 km of flying in 2008 and 2009, respectively. The number was considered to be too low to generate an estimate (Jenkins and Goorts 2011). Jenkins et al. (2012) flew an extensive stratified aerial survey of south Baffin using two helicopters with relatively high coverage to increase precision and distance sampling (line transects) to reduce bias. The estimate was 1,065–caribou (+/- 2,067 95% confidence intervals).
In 2014, expanded surveys covered north, northeastern and south Baffin using line transects and the double observer method (Campbell et al. 2015a). The overall estimate for Baffin Island was 4,652 (SE=702.79) caribou. Abundance was low on North Baffin as 315 caribou (159-622 95% confidence intervals) were estimated. About a third of the caribou estimated for southern Baffin were concentrated on Prince Charles Island (9,521 km2) which had an estimated 1,603 (1,158-2,200) caribou (Campbell et al. 2015b).
b) Trends in vital rates
Little information is available on the vital rates of Baffin Island caribou. Pregnancy rates appear to be annually variable but samples were small. Based on collections in April 1992 (n = 15) and April 1993 (n = 19) and March 1999 (n = 13), pregnancy rates were 67%, 79% and 69%, respectively. Pregnancy rates for the caribou females collared in 2009 and 2010 were 89% and 88% for 28 and 16 females, respectively. However, in 2009, none of nine collared females appeared to calve (Baffinland 2012). Mean age increased between 1992 and 1999 from 5.8 to 7.5 years (data from B. Elkin reported in Baffinland 2012). Of 32 females collared in 2008 and 2009, 13 were harvested, five died of natural causes and four died of unknown causes by 2011 (Jenkins and Goorts 2011).
Summary: population abundance and trends
Of 13 subpopulation units used to derive population estimates (Table 8), only two – Porcupine and Southampton Island – are known to be increasing; the latter is likely influenced by immigration from the mainland (Campbell and Boulanger 2016). Eight subpopulations are currently declining, and the trends of three are unknown, due to infrequency of surveys. Most of the large subpopulations have sufficient survey information to discern trends (Figure 20), with the exception of the Beverly-Ahiak unit, where the decline cannot be quantified at this time. Several survey areas have received little survey attention, leaving questions about subpopulation structure and population trends.
Available data were sufficient to model population change for seven of the 13 subpopulations of Barren-ground Caribou, representing about 70% of the total population. Four of these declined by > 80% over the past three-generation period. Other subpopulations suggest declines; some (Baffin Island) appear to have been dramatic. The summed change across the seven subpopulations resulted in an estimated median decline of -56.8%. The large decline was robust to choice of method and generation time (interquartile range: -54.3 to -57.8%; Figure 14), as well as imprecision in survey estimates (simulated 95% confidence interval: -17.0 to -76.3%; Table 8).
ATK and scientific study both suggest that Barren-ground Caribou populations undergo natural fluctuations of low and high abundance; in some subpopulations these fluctuations resemble cycles. At present, available demographic data for the declining subpopulations provide no indication of an impending increase, and suggest that more than one mechanism is operating to affect caribou over time, depending on which subpopulation. Three subpopulations appear to have stabilized at exceptionally low numbers, heightening their susceptibility to unpredictable stochastic events, such as unprecedented heat or icing storms or disease outbreaks. Further, there have been potentially significant cumulative changes to the environment, habitat and harvest regime for many of these subpopulations without historical precedent (see Habitat Trends, Threats). The implications of such changes for population fluctuations and recovery are unknown. Although a number of management actions are being developed and implemented, such as cessation of harvest (see Threats), their effectiveness is unknown at this time. As such, it would be unwise to assume a naturally occurring recovery, at least to historical numbers as recorded in the 1990s for many of the subpopulations (Tables 6, 7).
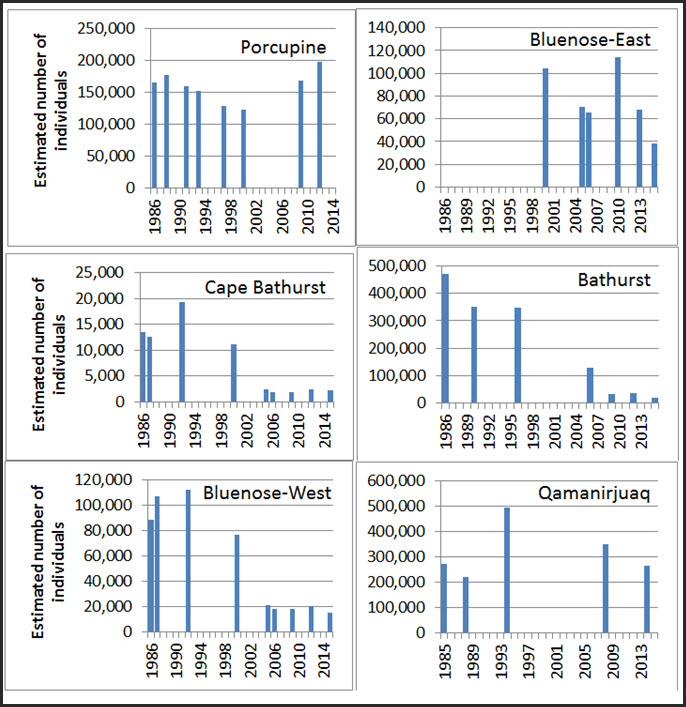
Long description for Figure 20
Six chart panels illustrating available population survey numbers over three generations for six large and well-surveyed Barren-ground Caribou subpopulations, representing roughly 67 percent of the total population.
Rescue effect
Barren-ground Caribou are bounded by five other caribou DUs (as well as the Fortymile Herd, which hasn’t been placed in a DU), each of which include caribou distinct in behaviour and movements, morphology, phylogenetics and/or genetics (COSEWIC 2011). For Peary Caribou (DU1), Dolphin and Union (DU2), Boreal Caribou (DU6), and Northern Mountain Caribou (DU7), seasonal distribution overlaps with Barren-ground Caribou in some areas and the caribou remain distinct (COSEWIC 2011). In such cases, rescue would not apply.
The range of the Porcupine subpopulation seasonally extends into Alaska and adjoins the distribution of other subpopulations that may be part of the same DU (see Global Range). Hence, rescue for the Porcupine subpopulation, if it declined to extremely low numbers and neighbouring subpopulations were at high abundance, cannot be ruled out. The immigrants would be likely to survive despite being on unfamiliar ranges. However, Alaska subpopulations are also declining at present, with the neighbouring Central Arctic population having been counted at 22,000 animals in 2016, a decline from about 70,000 in 2010 and 50,000 in 2013 (USFWS 2017). However, the distribution of the Porcupine subpopulation is limited to the east by the Mackenzie Valley and Delta so any rescue effect would be confined to the western extent of Barren-ground Caribou distribution. However, current trends of the three Alaskan migratory subpopulations that occur west of Porcupine are all currently declining (Parrett et al. 2014), further reducing the likelihood of a rescue effect.
Threats and limiting factors
Natural limiting factors
Despite observed changes in the abundance of caribou, there is still a lack of understanding of how natural limiting factors (predation, forage availability and parasites) interact. Predation and parasites are likely strongest when caribou abundance is declining or at low numbers (constant mortality has a greater effect at lower populations; see Predation). The potential limiting effects of foraging when populations of caribou are at peak numbers (Henry and Gunn 1991; Zamin and Grogan 2013) are important. Forage availability, and the interactions with caribou population dynamics, has been suggested as a limiting factor for the Bathurst subpopulation (Chen et al. 2014) as well as subpopulations in other caribou DUs (Manseau et al. 1996; Mahoney and Schaefer 2002). The growth cycle of lichens is slow and lichens may not be available for caribou until about 50 years post-fire (Barrier and Johnson 2012). Although Barrier (2011) reported that at the current low population density, winter range was not a limiting factor for the Bathurst subpopulation in 2010 and that lichen availability during winter may not be a causal factor for decline (Barrier and Johnson 2012). However, this is a mechanism that has been postulated as important in the dynamics of other migratory caribou (Messier et al. 1988; Joly et al. 2009). Heard and Williams (1992) outlined a mechanism for population dynamics based on the interaction between predation and foraging and caribou movements, the fundamental assumptions of which were supported by Klaczek et al. (2016)’s observations for Bathurst caribou, which suggested that even at low caribou numbers, Wolves still were efficient hunting caribou. The interaction between forage, predation (especially stress) and parasites is also a potential mechanism for caribou declines.
Threats
Disturbance and habitat loss
Disturbance (behavioural responses to and indirect habitat loss from industrial exploration and development) is a threat for many migratory species (Wilcove and Wikelski 2008). Behavioural responses to disturbance from human and industrial activities (e.g., Stankowich 2008) include local displacement (Boulanger et al. 2012) which is measurable as indirect habitat loss (see Habitat Trends). Incremental and cumulative loss of habitat can occur through the footprint of mines, oil and gas fields and roads as partial barriers to movements. Change or loss of migration as a response to human activities is recognized as a threat for other migratory ungulates (Berger 2004; Berger et al. 2008), but for Barren-ground Caribou there are information gaps (WRRB 2016). Studies of migration routes are only reported for subpopulations where environmental assessments for mines have been undertaken (e.g., Baffin, Qamanirjuaq and Bathurst subpopulations).
Habitat loss resulting from the cumulative effects of developments can be a threat, but the exact thresholds are uncertain and difficult to test (e.g., 7-12% for the Bathurst subpopulation) (De Beers 2012). Progress toward managing and mitigating cumulative effects is slow and uneven across the range of this DU (Gunn et al. 2014). For example, the need to manage cumulative effects was emphasized in the 1996 assessment for the first NWT diamond mine (CEAA 1996) and the subsequent assessments for the next three diamond mines between 2000 and 2012 (Lutsel Ké Dene First Nation 2012; Tlicho Government 2012).
While there is no direct evidence about cumulative disturbance and habitat loss as a factor in the decline of the Bathurst subpopulation, the increasing scale of the developments is considered a contributing factor (Lutsel Ke Dene First Nation 2012). Computer modelling (Gunn et al. 2011c; De Beers 2012) projects that the cumulative effects of behavioural responses and local displacements from exposure to current and future mines may have moderate subpopulation-level effects through reductions in pregnancy and calf survival rates within a caribou generation.
Over-hunting
Barren-ground Caribou have been hunted for thousands of years and harvest is a large part of the life of Indigenous peoples (SARC 2016). Harvest opportunities are dictated by the movements and distribution of the subpopulation with respect to the location of communities within and adjacent to the subpopulation’s range, although the numbers taken may also be influenced by harvests for commercial meat or for meat to send to distant communities. The conditions under which hunting becomes or may become a threat are incompletely understood, and data on numbers of harvested animals is often incomplete. The relative synchrony among subpopulations in the timing of peaks, declines and low numbers along with overlapping winter distribution and switching of harvest among neighbouring subpopulations makes harvest management difficult. From a traditional knowledge perspective, the link between harvesting and population dynamics is not well understood (Spak 2005; Sandlos 2007; Wray and Parlee 2013), and is all the more complex because of the cultural perception that harvesting is integral to subpopulation health (Parlee and Furgal 2010).
Understanding of harvest pressure is challenged by the difficulty of obtaining accurate harvest counts for any given subpopulation, which tend to be unevenly harvested by multiple communities. Information is not generally tracked by territorial governments; rather it is collected at the community level in some cases. In Nunavut, harvest reporting is voluntary. For the Bathurst subpopulation, the Dogrib Harvest Study (based on community interviews) ran from 1989-93 and reported an initial increase from 8,000 caribou harvested for the first two years to a stable harvest of about 20,000 caribou for 1989 to 1993 (Boulanger et al. 2011). The resident harvest (non-Indigenous hunters) peaked in 1993 at 1,800 Barren-ground Caribou (data including both NT and NU) before declining to <100 in 2007 in NT only, unknown but likely similarly low in NU; Carrière 2012). Information on subsistence harvesting resumed from road check stations and interviews in 2007/08 and 2008/09. The winter harvest was estimated at 3,380-5,424 caribou, mostly females (Adamczewski et al. 2009). In 2010, harvesting was restricted to 300 caribou and then in 2016, no caribou were to be harvested (WRRB 2016). Harvest rates were annually reported to the Beverly-Qamanirjuaq Caribou Management Board (BQCMB 2006, 2007, 2008, 2009, 2010a), but it is not always clear how the rates were estimated and how they were assigned to individual subpopulations when the Bathurst, Beverly, Ahiak and Qamanirjuaq subpopulations overlapped in winter distribution. After 2008-2009, BQCMB were no longer able to report harvests because information was not received from government agencies and there were difficulties in assigning harvests to subpopulations. Harvest rates on the tundra ranges are also difficult to assign to subpopulations as they are collected for individual communities. Annual caribou harvest for Baker Lake recorded from 1996 to 2001 ranged between 2,230 and 3,116 animals, using upper limits of standard error (Priest and Usher 2004). Trends in harvest from Baker Lake are currently measured from a study that is part of the monitoring for Agnico-Eagle’s Meadowbank gold mine (Gebauer et al. 2013) and tracks changes in harvest as a result of the mine.
Some data collected for some subpopulations indicate clear population-level impacts from harvesting. Of 32 adult caribou females collared on north Baffin Island from 2008 to 2011, local hunters harvested 13 individuals, or 41%, a rate that is indicative of north Baffin Island hunting pressure (Campbell et al. 2014). Campbell et al. (2014) also noted increases in harvesting that occurred as a direct result of research activities, where a number of local hunters travelled to the survey area to hunt the caribou that were observed during the survey. Over-harvest has become the dominant threat to the long term sustainability of the Southampton population, with a growing export market within Nunavut having pushed harvest rates “well beyond sustainable limits” (Campbell and Boulanger 2016).
Harvest is more likely to increase as a threat when monitoring is poorly understood and leads to delays in management. For example, there was a 7-year delay between first measurement of a decline (2003) and subsequent harvest restrictions (2010) for the Bathurst subpopulation (GNWT 2016; see Subpopulation-Specific trends) during which time, the declines accelerated. Lack of monitoring was identified as a factor challenging timely management action in the possible collapse of the use of Beverly subpopulation’s traditional calving grounds (BQCMB 2014b; Adamczewski et al. 2015).
When Barren-ground Caribou population numbers are lower than typical under natural fluctuation regimes, any impact from hunting will exacerbate other threats and recovery can be slower (Beaulieu 2012). “You can’t expect the caribou to go up when we are killing, killing, killing.” (Bluenose Caribou Management Working Group [Tsiigehtchic] in Benson 2015: 55). During declines, hunting is more likely to become a threat when increased access along winter roads combined with changing technology uncouples harvesting effort from caribou abundance and harvest level stays constant despite declining abundance (Mullon et al. 2005; Fonzo et al. 2013; SARC 2016). For example, in the Bathurst subpopulation, the decline accelerated as the constant harvesting level increased from 2-4% to 10-16% of abundance (Boulanger et al. 2011). Recently (within a caribou generation), harvesting technology has changed, for example on Baffin Island (DOE-GN 2013) and in the NWT (Nesbitt and Adamczewski 2009). Such shifts, including high-powered snowmobiles and chartering aircraft, made it easier to increase harvesting effort. Increased road access is often cited as a cause of increased hunting pressure, and first-hand observations of increases in harvest levels and changes in hunting locations facilitated by new road access are reported frequently to caribou management boards (e.g., Wakelyn, pers. comm. 2013). However, generally quantitative information is missing. An exception is the monitoring of harvest on the all-season road built to service the Meadowbank mine north of Baker Lake; harvesting of caribou from Lorillard, Wager Bay and Ahiak subpopulation(s) increased along the road but possibly decreased elsewhere (Gebauer et al. 2013).
Management planning for six subpopulations (Baffin, Cape Bathurst, Bluenose-West, Bluenose-East, Bathurst, and Southampton) has between 2007 and 2015 led to harvest restrictions in response to declines. For example, based on recommendations of the Wildlife Management Advisory Council (NWT), the Gwich'in Renewable Resources Board, and the Sahtu Renewable Resources Board, all non-Indigenous hunting of the Bluenose-West subpopulation ceased in 2006. The co-management boards recommended restricting Indigenous harvesting which was implemented in 2007. The Total Allowable Harvest for the Bluenose-West subpopulation was set at 4% (712 caribou) in 2007/08, with recommendation that the harvest be 80% males (Davison 2014). In response to the decline in the Bluenose-East subpopulation between 2000 and 2010, the Wek’eezhii Renewable Resource Board recommended closing the commercial meat, outfitting and resident harvest and restricting the Indigenous harvest to an annual harvest of 2800 caribou with a 85:15 ratio of males to females and then in 2016, a further reduction to 750 caribou (WRRB 2010a, 2016). In 2010, harvesting of the declining Bathurst subpopulation was subjected to tight restrictions when it was reduced to 300 caribou in the NWT and again in 2014 when it was reduced to a ceremonial harvest of 15 males (GNWT 2015). Following surveys documenting a >90% decline in caribou numbers on Baffin Island, the Government of Nunavut imposed an interim moratorium on the harvest of caribou on Baffin Island effective January 1, 2015, which it replaced with a quota of 250 male caribou in August, 2015. In response to population declines, harvest of Southampton Island caribou were subjected to restrictions in 2012 (Campbell and Boulanger 2015).
Yukon and NWT residents can hunt Porcupine Caribou (two male tags per season) in their respective territories. There is also a resident harvest of any subpopulation allowed in Nunavut of five tags per season. While harvest restrictions (2007-2010) initially halted or reduced the rate of decline, at least for Cape Bathurst and Bluenose-West subpopulations, the declines between 2012 and 2015 continued or resumed for Cape Bathurst, Bluenose-West, Bluenose-East and Bathurst, suggesting limits to management planning that relied on harvest restrictions.
Although the Porcupine Caribou subpopulation currently has an increasing trend, it faced concerns of a potential significant decline in the early 2000s and no reliable population estimate. Consequently, the Porcupine Caribou Management Board worked with the eight governments with management responsibilities for the herd to develop a comprehensive Porcupine Caribou Harvest Management Plan (2010) and an associated Implementation Plan (2010). These plans identified management triggers for different population levels and included requirements for harvest monitoring and reporting, native user allocation agreements, stipulating a greater proportion of males in the harvest rather than females. (PCMB 2010a). The Implementation Plan also established an annual meeting of all the parties to review the status of the herd and determine what actions are required for the upcoming year to help ensure the sustainable management of the herd (PCMB 2010b).
For some subpopulations, the extent of hunting as a threat has been complicated by non-resident and commercial harvests. For example, the harvest of caribou males by outfitters on the Bathurst subpopulation averaged about 825 annually, peaking in 2001 (1,166 males), and was reduced through changes in the quota between 2005 and 2009 (Adamczewski et al. 2009). Outfitter harvesting was terminated in 2010 over concerns about declining subpopulation numbers. Although the Porcupine Caribou Management Agreement (PCMA) states that there shall be no commercial harvest of Porcupine Caribou in Canada, this does not apply to guiding and outfitting. Further to this issue, the PCMB has developed Guidelines for the Sale, Trade and Barter of Porcupine Caribou Meat as directed by the PCMA. Currently, there is no commercial harvesting of any NWT Barren-ground Caribou subpopulations, but limited non-resident hunting occurs within Nunavut. Most harvesting of caribou for commercial purposes outside the Nunavut Settlement Area ended in 2007, although Nunavut and caribou outfitters continue to guide non-resident hunters in northern Manitoba. Caribou harvest for inter-settlement trade also continues to occur and there is significant unregulated inter-regional trade of caribou, primarily from the Kivalliq to Baffin regions (Campbell et al. 2015) and over the Internet (BQCMB 2016a,b).
Also considered a threat to Barren-ground Caribou is non-traditional harvest practices, including reckless shooting, overuse of motorized vehicles, wastage of meat and leaving carcasses on the ground, not sharing meat, and not using the entire carcass. Multiple ATK sources (WMAC (North Slope) and Aklavik HTC 2009; Beaulieu 2012; Sangris 2012; BQCMB 2014b; Benson 2015) indicate that Barren-ground Caribou may abandon an area if such hunting practices are occurring. It is also commonly observed that killing or disturbing the leaders of the migration can be detrimental from a variety of perspectives (Whaèhdôö Nàowoò Kö [Dogrib Treaty 11 Council] 2001).
Climate change
The signals of climate change are especially strong in the Arctic, as measured by reductions in sea ice and warmer temperatures. Although evidence is already strong for changes such as an increase in shrubs (Myers-Smith et al. 2011), changes to the ecology of Barren-ground Caribou will be complex, consisting of positive and negative effects, most of which are interacting and non-linear (Cebrian et al. 2008; Chen et al. 2014). The effects of climate change on forage availability during calving and summer appear important (Griffith et al. 2002; Chen et al. 2014) but are still not well understood. A climate envelope was measured at which lower availability of summer range forage explained some of the variation in productivity 2-3 years later in the Bathurst subpopulation (Chen et al. 2014).
Future climate change may act as a continuing threat for Barren-ground Caribou through a complex mechanism involving shifts in timing of greening, lower summer forage quality, and subsequent lower calf production and reproductive potential of females, then population declines. Unpredictable weather events, which are increasing in frequency in a changing climate, are also implicated in population declines. In August 2016, 47 caribou carcasses were found on Prince Charles Island with unusually low fat reserves indicative of starvation. A storm (e.g., rain on snow) creating a layer of ice and preventing access to forage is the most likely explanation (Van Dusen 2017).
Caribou may be susceptible to heat stress (Soppela et al. 1986): days with mean daily temperatures exceeding 25oC are infrequent for Bluenose-East, Bathurst and Qamanirjuaq subpopulations and the number of days when temperatures exceeded mean+2 SD for the 1990s was similar to the 2000s. 2014 stands out with more high temperature days for the three subpopulations, which exceeded previous totals for 1979 to 2014 (CARMA unpubl. data). As such, there are considerable limits to present understanding and consequent uncertainty until more explorative modelling and discussion takes place.
Climate change is a growing concern for migratory species, where timing of arrival to breeding grounds is critical for survival (Crick 2006). Highly productive seasonal habitats become less food-rich and predictable in space and time and species like caribou are forced to contend with a decoupling of climate variables between seasonal ranges, such that mistimed migration becomes an increasing likelihood (Robinson et al. 2009).
Contaminants
Most contaminant levels in caribou tissue are generally low based on monitoring (Elkin and Bethke 1995; Braune et al. 1999; Gamberg et al. 2005; Gamberg 2009). Determining trends in contaminant levels is difficult as the sample size and frequency varies among subpopulations with the Porcupine subpopulation being sampled annually while the Qamanirjuaq subpopulation was sampled in 1992, 1993, and 2006 (although differences in the season sampled for the Qamanirjuaq subpopulation impedes describing trends) (Gamberg 2009). The Beverly subpopulation was sampled in 1994 and 2000; Cape Bathurst and Bluenose-West between 1994 and 2006, and the Bathurst subpopulation in 2005-06 (Gamberg et al. 2005; Gamberg 2009; Larter et al. 2010).
While most chemicals and metals are at low concentrations, the levels of mercury in the kidneys have increased over time for the Porcupine subpopulation (Gamberg 2009); in contrast, levels of mercury decreased over time for Cape Bathurst caribou (Larter et al. 2010). The contrast highlights uncertainties about atmospheric trends in mercury and implications for Arctic ecosystems (Gamberg 2009). Another potential threat could occur if novel chemicals (from either local sources or from sources subject to long-distance transport) come into common use or are found to accumulate in the Arctic environment. For example, use of brominated flame retardants and fluorinated surfactants has increased since the 1980s (Stow et al. 2004).
Lichens are important in caribou diet and lichens have a propensity to accumulate atmospheric contaminants. Monitoring has revealed increased levels of chromium and manganese in lichens near a large open-pit diamond mine on the summer range of the Bathurst caribou subpopulation (Enns 2012). During the environmental assessments for mines, dust is listed as a potential concern as it is generated by mine activities, including roads, and is carried by wind and rain or snow onto vegetation, including caribou forage. In the vicinity of an abandoned gold mine on the Bathurst winter range, increased levels of some metals were found in the fecal pellets of caribou (Macdonald and Gunn 2004). A similar finding was the elevated levels of metals in the fecal pellets and tissues from caribou of the Western Arctic subpopulation collected near the Red Dog base metal mine (O’Hara et al. 2003). The levels of metals were not considered to be a risk for human health, but adverse effects on caribou health, while unlikely, are unknown. The dust-contaminated forage was proposed as a factor in caribou showing reduced probability of use in the vicinity of the diamond mines on the Bathurst subpopulation’s summer range (Boulanger et al. 2012).
Cumulative impacts
Migratory caribou have demonstrated negative responses in distribution and behaviour to changes in habitat and stimuli that are associated with human activities (Cameron et al. 2005; Boulanger et al. 2012). However, direct threats interact with each other with either additive, synergistic, interactive or nonlinear impacts. Most Barren-ground Caribou subpopulations are now at low points in their abundance and they are facing the cumulative effects from multiple interacting threats that were not present in the past (Campbell et al. 2014). These include increased development and industrial activity, growing human populations with advanced hunting equipment, techniques as well as access yielding increased harvest, and a changing climate. Each of the major development projects that is subject to environmental assessments include cumulative effects assessments but these are proponent rather than issue-driven and have not made a significant contribution to managing cumulative impacts for caribou (Gunn et al. 2011c). Also, these assessments rarely consider the full range of activities that might act on populations of caribou in a cumulative way (Johnson et al. 2005). A key challenge is the lack of overall land use planning, especially in the context of cumulative effects of industrial developments and human activities. In particular, the lack of an overall approach to calving ground management is a specific issue of concern.
Whether limiting factors such as disturbance and harvesting become threats is largely dependent on the corresponding management response. In theory, those limiting factors are typically reversible or their effects can be accommodated through trade-offs between them. In practice, this is more complicated and efforts to manage cumulative impacts are often absent or, at best, delayed. A lack of systems thinking (Richmond 1993) about ecological processes will compound delays in management action or lead to inappropriate assessments and treating or mistreating causes of declines. Although harvesters and managers are aware of cycles in abundance, this has not led to a conceptual framework for monitoring and management based on adaptive behavioural use of space over time relative to cycles of abundance. For example, at extremes of high and low abundance, we must recognize that caribou adapt through changes in gregarious calving behaviour. This lack of understanding has led to gaps in monitoring and limited effective management actions. While there is acknowledgement of the Precautionary Principle, in practice, a lack of certainty often leads to inaction, which is a threat to a subpopulation if a real decline is underway and left unmanaged (an example of a Type II error that involves assuming a decline has not occurred, when it actually has).
The slow development of plans for subpopulation range and harvest management challenges the ability to address cumulative impacts. Although management strategies have been prepared cooperatively between governments including Indigenous governments and wildlife management boards (see ENR 2011; Table 11), their implementation takes time and extensive consultation, and some remain relatively high-level documents that contain few specific actions to manage threats at the subpopulation level. Management of cumulative impacts demands access to credible and current information and shortfalls or delays in access to information may lead to delays in management actions to mitigate threats. For example, when declines are initially reported, the steps to confirm and then consult before taking actions (to avoid Type 1 error, i.e., assuming a decline has occurred when it has not) can be time-consuming. The Bathurst subpopulation’s decline was first identified in 2003, but until 2010 few and limited actions were undertaken, by which time the decline had accelerated and population size had declined from 185,000 to 31,200 individuals. So while uncertainty of information and consequent delays in management actions is rarely, if ever, listed as a threat, experience indicates it does happen. A review of recovery of marine fisheries reported that quickly halting declines influences the timing and extent of recovery (Hutchings and Reynolds 2004).
By contrast, for the Porcupine subpopulation, the experience of insufficient information on population status resulted in anticipatory actions among agencies and user groups. From 2001 until 2010 the trend in subpopulation size was unknown. In the absence of trend information, groups developed and implemented a Harvest Management Plan where subpopulation monitoring was conducted, reported and assessed on an annual basis and full harvest reporting was undertaken. In 2010 the subpopulation was estimated to have increased to 169,000, but the Plan is still in place and annual meetings examine the monitoring data and recommend any harvest management response (First Nation of the NaCho Nyak Dun et al. 2010).
| Subpopulation | Management Authorities u | Management Planning and Process | Reference (link) |
|---|---|---|---|
| Porcupine | US-AK, GRRB, WMAC(NS), WMAC(NWT), YG, GNWT, PCA, PCMB, IPCB, | Porcupine Caribou Harvest Management Plan (2010) and an associated Implementation Plan (2010), Porcupine Caribou Management Board Strategic Framework (2013), and Porcupine Caribou Herd Annual Status Report (2010-2016), Wildlife Conservation and Management Plan for Yukon North Slope (2002); Plan for the International Conservation of the Porcupine Caribou Herd (1987) |
Porcupine Caribou Management Board, Plan for the International Conservation of the Porcupine Caribou Herd (PDF version; 47.9 KB). |
| Tuktoyaktuk | GNWT, WMAC (NWT) | blank | blank |
| Cape Bathurst | GNWT, WMAC(NWT), GRRB, SRRB | Management plan for the Cape Bathurst, Bluenose-West, and BNE herds (2014) | Taking Care of Caribou. Cape Bathurst, Bluenose-West, and Bluenose-East Barren-Ground Caribou Herds Management Plan. |
| Bluenose-West | GNWT, GNU, Parks Canada, WMAC(NWT), GRRB, SRRB, NWMB, TNNPMB | Management plan for the Cape Bathurst, Bluenose-West, and BNE herds (2014), Belarewıle ́ Gots’e Ɂekwe (Caribou for All Time) Délıne Caribou Conservation Plan (2016). | Belarewıle ́ Gots’e Ɂekwe (Caribou for All Time) Délıne Caribou Conservation Plan (2016) |
| Bluenose-East | GNWT, GNU, TG, WRRB, SRRB, NWMB, PCA, TNNPMB, SENHSMB | Management plan for the Cape Bathurst, Bluenose-West, and BNE herds (2014); Draft action plan for the herd (2015), Délıne Caribou Conservation Plan ( 2016). | ACCWM Update: Action Planning for Bluenose Caribou Herds Deline Community (2016) |
| Bathurst | GNWT, GNU, TG, WRRB, SRRB, NWMB | NT: Management plan (2004); Emergency measures enacted (2010), Draft Range Plan (2016), Public Hearing Process (2015-2016) NU: Public Hearing Process (2016), Délıne Caribou Conservation Plan (2016). |
Public Hearing Registry (Bluenose East Caribou Management Hearing - March 2016 and Bluenose West Caribou Management Hearing - November 2007) |
| Beverly | GNU, GNWT, GSK, GMB, Canada, NWMB, BQCMB | Beverly and Qamanirjuaq Caribou Management Plan 2013-2022 | Beverly and Qamanirjuaq Caribou Management Plan 2013-2022 |
| Ahiak | GNU, GNWT, Canada, NWMB | blank | blank |
| Qamanirjuaq | GNU, GNWT, GSK, GMB, Canada, NWMB, BQCMB | Beverly and Qamanirjuaq Caribou Management Plan 2013-2022 | blank |
| Southampton | GNU, NWMB | Management plan (2011); Public Hearings (2011, 2014) | Management plan (2011); Public Hearings (2011, 2014) |
| Baffin Island | GNU, NWMB | Interim measures (2014;2015) | Minister initiates interim moratorium on Baffin Island caribou harvest |
u US-AK (United States – Alaska, Nunavut Wildlife Management Board (NWMB), Gwich’in Renewable Resources Board (GRRB), Sahtu Renewable Resources Board (SRRB), Wek’èezhìi Renewable Resources Board (WRRB), Wildlife Management Advisory Committee North Slope (WMAC -NS), Wildlife Management Advisory Committee NWT (WMAC- NWT), Tłįcho Government (TG), Yukon Government (YG), Government of the Northwest Territories (GNWT), Government of Canada (Canada), Government of Nunavut (GNU), Parks Canada Agency (PCA), Porcupine Caribou Management Board (PCMB), International Porcupine Caribou Board (IPCB), Tuktut Nogait National Park Management Board (TNNPMB), Sayou Edacho National Historic Site Management Board (SENHSMB).
Number of locations
Calving grounds may be considered locations in the context of likely threatening events, as almost all females of subpopulations aggregate together on a calving ground at the time of year when females with newborn calves are especially sensitive to environmental variability and disturbance (Wolfe et al. 2000). The total number of Barren-ground Caribou subpopulations is, however, uncertain (see Subpopulations). Furthermore, at low abundance, calving for some subpopulations is more dispersed, which adds difficulty in assessing the number of locations. Currently, information is unavailable to assess the degree of dispersion of calving on the northeast mainland and Southampton, Coats and Baffin Islands. Given the number of known subpopulations, the number of locations is at least 14, but very likely exceeds this number.
Protection, status and ranks
Legal protection and status
Barren-ground Caribou was assessed for the first time by COSEWIC as Threatened in November 2016, and is currently not scheduled under the Species at Risk Act (SARA).
In 1984, the Inuvialuit Final Agreement identified the establishment of the Porcupine Caribou Management Agreement (IFA 1984, Annex L). This set out the parameters for the cooperative management of the Porcupine Caribou subpopulation. The 1985 Porcupine Caribou Management Agreement (PCMA) between Canada, Yukon, NWT and affected Inuvialuit and First Nation governments established the management structure for the herd and established the Porcupine Caribou Management Board (PCMB). The PCMA is annexed to the Inuvialuit Final Agreement (IFA 1984, Annex L). The mandate of the PCMB includes making recommendations on any matter affecting the Porcupine Caribou and its habitat (PCMA Section E.1.a). Subsequently, the International Porcupine Caribou Agreement (1987) provided for the coordinated management of the subpopulation between the US and Canada and established the International Porcupine Caribou Board and the supporting Porcupine Caribou Technical Committee, which coordinate research and management of the herd between jurisdictions. The Wildlife Management Advisory Council (North Slope) provides advice to these bodies. (IFA 12 (56)a).
Protection of other subpopulations by territorial and provincial jurisdictions is through harvest regulation and habitat protection. Harvest regulation varies with jurisdiction and varies by category. In NWT, the three general categories of hunting licence holders (Aboriginal hunters, resident hunters, and ‘commercial’ hunters (including non-resident hunters)) operate differently on population dynamics given that they target different sex/age categories. Commercial harvesting is mostly quota-based, guided, and focused on prime males. The three categories in Yukon are Aboriginal, licensed resident, and licensed non-resident. The latter can be guided by an outfitter with a quota, or a licensed resident hunter holding a special guiding licence. Both opportunities are limited and harvest is very low relative to other hunter categories. In Nunavut, the categories are Beneficiary (under the Nunavut Land Claims Agreement), Non-Beneficiary and Non-Resident (sport hunt). Currently, there is no legal commercial harvesting of any NWT Barren-ground Caribou subpopulation, but limited non-resident hunting occurs within Nunavut and Yukon and limited sport hunting in Nunavut. Yukon and NWT residents can hunt Porcupine Caribou (two male tags per season) in their respective territories. Non-Indigenous residents in northern Saskatchewan and Manitoba can annually hunt one or two Barren-ground Caribou, respectively.
The large ranges of Barren-ground Caribou mean they cross jurisdictional boundaries and the boundaries of different land claims and groups of people. For example, the subsistence harvesters for Cape Bathurst, Bluenose-West, and Bluenose-East subpopulations are from 14 communities within five land claim/regional areas and two territories. The Beverly and Qamanirjuaq subpopulations at one time included residents of more than 20 communities from three provinces (AB, SK, MB) and two territories (NU, NT) in an area with one established land claim and four more under negotiation. More recently, the Qamanirjuaq subpopulation alone has been harvested by at least 14 communities from four of these jurisdictions. The Porcupine subpopulation ranges across Alaska, Yukon, and the Northwest Territories, requiring a coordinated and cooperative management approach among all user groups and jurisdictions in two countries, two territories, and five Indigenous governments.
The co-management system in which all users and management authorities coordinate monitoring, research and conservation actions is used for the management of all Barren-ground Caribou subpopulations. Shared responsibility is based on land claims legislation in areas with approved land claims and on renewable multi-year agreements where claims have not yet been finalized. The often complex system is designed to work towards consensus on conservation measures, such as hunting bans (Table 11). Various community-based user groups (e.g., hunter and trapper organizations) can self regulate harvest and develop by-laws or other restrictions; however, territorial governments may impose emergency measures in the interest of conservation. Strict harvest limitations have been agreed to for the subpopulations that have shown a steep reduction in abundance.
Non-legal status and ranks
Globally, caribou were listed by the International Union for Conservation of Nature (IUCN) as Least Concern until 2016, when the species was assessed for the first time as Vulnerable (IUCN 2016). Caribou subspecies or ecotypes are not differentiated. NatureServe ranks caribou (R. tarandus) as secure globally and Not Yet Ranked for Barren-ground Caribou (R. t. groenlandicus), other than NWT (S3; Vulnerable), and Saskatchewan (S3S4) (NatureServe 2017). The 2015 national general status for Caribou in Canada will not be available until the 2015 General Status Report is published in August 2017. This Canada-wide rank will apply to all DUs of Caribou combined, with nothing specific to Barren-ground Caribou. The 2015 territorial rank for YT for Barren-ground Caribou is S3S4 (Vulnerable to Apparently Secure) (Yukon CDC 2017, Cannings pers. comm. 2017), and for NWT is S3 (Sensitive) (WGGSNS 2016). At present, there is no specific rank for Barren-ground Caribou for NU; however, for all DUs combined, the territory-specific general status rank for Caribou in Nunavut is S4 (Apparently Secure) (Etiendem, pers. comm. 2017).
Habitat protection and ownership
The majority of Barren-ground Caribou habitat is on public land administered by the territorial and provincial governments, except the Tlicho Government administers Tlicho Lands. Crown lands in Nunavut are currently administered by the Government of Canada. The second category of land ownership is Private Settlement Lands, which were selected during land claims (Inuvialuit Settlement Region, Gwich’in Settlement Area, Sahtu Settlement Area, and Tlicho Lands). A total of 14.4% (315,800 km2) of Barren-land Caribou range is private lands, owned and administered by Indigenous corporations or associations created under various land claim agreements. Land claims negotiations with First Nations in northern Saskatchewan and Manitoba will soon finalize land selection for areas on caribou range in the provinces and adjoining territories.
National parks and national park reserves are categories of protected areas on federal land in Canada that limit development but not traditional harvesting. There are seven such areas, ranging in size from 4,345 km2 (Vuntut) to 22,200 km2 (Sirmilik), covering 104,117 km2. The proposed Thaidene Nëné National Park Reserve, located at the eastern end of Great Slave Lake, NWT, will add 14,000 km2 to the network. Migratory Bird Sanctuaries are an additional federal land protection category, of which there are eight within Barren-ground Caribou ranges, but the scope of protection in these areas is limited to migratory birds and their habitats. These range in size from 2 km2 (Cape Perry) to 61,765 km2 (Queen Maud Gulf). In addition, NWT and NU share the Thelon Game Sanctuary, which also is not open to development. The total area of these protected areas within Barren-ground Caribou range is approximately 180,000 km2, covering approximately 6% of Barren-ground Caribou range.
In the NWT, there are additional protected areas being established which will also provide varying levels of habitat protection for Barren-ground Caribou. There are also several parks, ecological reserves and special management areas on caribou range in Yukon and northern Manitoba. For instance, the Old Crow Flats Special Management Area, which includes a portion of the Porcupine Caribou range and is administered by the federal, Yukon and Vuntut Gwitchin governments, has been recommended for permanent withdrawal from industrial resource development (Government of Yukon 2006). In northern Manitoba, five provincial parks (Caribou River, Colvin Lake, Nueltin Lake, Numaykoos Lake, and Sand Lakes) protect more than 25,000 km2 of Barren-ground Caribou winter range through prohibition and restrictions on certain development activities. Four of the national parks provide year-round protection for part of the current calving grounds of five subpopulations. Tuktuk Nogait National Park was established in 1998, and includes most of the Bluenose-West subpopulation’s calving and post-calving areas although trends in the extent of distribution within the Park have not been assessed. The Lorillard and Wager Bay subpopulations calve partially within Ukkusiksalik National Park, established in 2003. Any trends in the extent of use are unavailable. In Yukon, the Porcupine subpopulation has a variety of habitat protection tenures within their range. Most of the range north of the Porcupine River is protected by two National Parks (Vuntut and Ivvavik) and a 1967 Order-in-Council Withdrawal area east of the parks which prevents exploration and development. Since 2000, the frequency of calving in Ivvavik National Park has been increasing; in 5 of 9 years during 2002-2010, calving occurred on the coastal plain primarily in Yukon within Ivvavik National Park (Caikoski 2011). Three territorial parks (Fishing Branch, Herschel Island and Tombstone) also offer some level of protection.
Land use planning may result in habitat protection through areas of restricted development. In NWT, the Inuvialuit, Gwich’in and Sahtu regions have completed land use plans and the Dehcho plan has been under development for many years. The Sahtu plan includes some protection from industrial activities on areas of Barren-ground Caribou range. The Tlicho Land Use Plan, specific to Tlicho lands that are located within the area known as Wek’èezhìı, was completed in 2013, while a land use plan for the Wek’èezhìı area has not been completed. The Athabasca Interim Advisory Panel completed a draft land use plan that included restrictions on land use activities along a 50-km wide road corridor through Barren-ground Caribou winter range in northern Saskatchewan. The Nunavut Planning Commission is developing a territorial land use plan that will replace the existing Kivalliq and North Baffin regional plans. The 2016 draft plan proposes establishing protected areas that would restrict development over extensive areas, including Nunavut’s caribou calving and post-calving areas (Government of Nunavut 2016).Much of the winter range of the Porcupine Caribou subpopulation in Yukon has no formal protection designations although the area is covered by the North Yukon Land Use Plan and the contested Peel Watershed Land Use Plan.
Acknowledgements and authorities contacted
Members of the COSEWIC Terrestrial Mammal Subcommittee provided careful reviews, advice, and analyses throughout the process of putting this status report together. The writers have been greatly assisted by Environment and Natural Resources (Government of the Northwest Territories) staff: J. Adamczewski, M. Branigan, S. Carrière, D. Cluff, T. Davison, B. Elkin, and R. Gau freely supplied information, advice and help. M. Campbell (Government of Nunavut) is appreciated by the writers for sharing his draft reports. The writers especially thank B. Fournier, ENR, for providing Caribou data from the ENR WMIS database and J. Williams ENR who went to great lengths to answer requests and check information. Special thanks are extended from the writers to S. Carrière, B. Fournier, C. Johnson, and M. Southee for creating the figures and maps and to C. Johnson for conducting analyses that appear in the Abundance and Trends section. J. Ray (WCS Canada) provided valuable assistance in proofing and finalizing Information Sources.
The writers wish to thank the following individuals for furnishing information and analyses, and for providing thoughtful and thorough reviews at various stages:
| Jurisdiction | Name of Contact |
|---|---|
| Government of the Northwest Territories | Jan Adamczewski, Suzanne Carrière, Karin Clark, Tracey Davison, Brett Elkin, Nic Larter, Claire Singer |
| Government of Nunavut | Lisa-Marie Leclerc, Mitch Campbell, Matthieu Dumond, Lynda Ormond |
| Government of Yukon | Troy Hegel, Mike Suitor, Todd Powell, Thomas Jung |
| Canadian Wildlife Service | Amy Ganton, Shannon Stotyn, Keri McFarlane, Syd Cannings, Ruben Boles, Lea Craig-Moore, Dawn Andrews |
| Parks Canada | Patrick Nantel, Peter Sinkins, Ian McDonald, Andrew Maher, Jane Chisholm, Maryse Mahy |
| Gwich’in Renewable Resources Board. | Kaytlin Cooper, Natalka Melnycky |
| Wek’èezhìı Renewable Resources Board | Jody Pellissey, John McCullum, J. Grant Pryznyk |
| Wildlife Management Advisory Council (North Slope) | Lindsay Staples |
| Nunavut Wildlife Management Board | Karla Letto |
| Beverly-Qamanirjuaq Caribou Management Board | Leslie Wakelyn, Ross Thompson |
| Porcupine Caribou Management Board | Joe Tetlichi, Deana Lemke |
| Sahtu Renewable Resources Board | Colin Macdonald |
| Other experts and relevant contacts | Doug Heard, Alan Sinclair |
| COSEWIC Secretariat | Karen Timm, Jenny Wu |
| COSEWIC Terrestrial Mammals Subcommittee |
Justina Ray, Nicolas Lecomte, Susan Kutz, Christopher Kyle, Chris Johnson, Martin-Hughes St.-Laurent, Graham Forbes, Hugh Broders, Scott Gilbert, Stephen Petersen, Ian Thompson, Gloria Goulet |
| COSEWIC Aboriginal Traditional Knowledge Subcommittee | James Pokiak and Norma Kassi |
