Rusty blackbird (Euphagus carolinus): COSEWIC assessment and status report 2017
Rusty Blackbird

Photo: © Carl Savignac
Special concern
2017
Table des matières
- Table des matières
- COSEPAC Sommaire de l’évaluation
- COSEPAC Résumé
- Résumé technique
- Préface
- Wildlife species description and significance
- Distribution
- Habitat
- Biology
- Population sizes and trends
- Threats and limiting factors
- Threats
- Residential and commercial development (iucn threat #1); agriculture and aquaculture (iucn threat #2); energy production and mining (iucn threat #3)
- Biological resource use (iucn threat #5)
- Natural system modifications (iucn threat #7)
- Pollution (iucn threat #9)
- Invasive and other problematic species and genes (iucn threat #8)
- Climate change and severe weather (iucn threat #11)
- Limiting factors
- Number of locations
- Threats
- Protection, status and ranks
- Acknowledgements
- Authorities contacted
- Information sources
- Biographical summary of report writer
List of figures
- Figure 1. Adult male Rusty Blackbird in breeding plumage
- Figure 2. North American distribution of the Rusty Blackbird in the breeding (light grey) and wintering (dark grey) seasons.
- Figure 3. Breeding distribution (shaded area) and extent of occurrence (area bounded by the line) of the Rusty Blackbird in Canada.
- Figure 4. Relative suitability of Rusty Blackbird breeding habitat in Canada, based on models using data from the Boreal Avian Modelling project’s dataset in ecoregions that intersect the southern extent of the hemi-boreal region.
- Figure 5. Density model within the breeding range of Rusty Blackbirds in the boreal forest (males/ha), for the period 1961-1990.
- Figure 6. Median number of Rusty Blackbirds detected per party hour on the Christmas Bird Count in North America between 1970 and 2012.
- Figure 7. Annual abundance indices of Rusty Blackbirds for Canada between 1970 and 2014 (with 95% confidence intervals), according to a hierarchical Bayesian model of Breeding Bird Survey data.
- Figure 8. Distribution of Rusty Blackbird in Ontario during the period 2001-2005.
- Figure 9. Comparison of the range of the Rusty Blackbird in Quebec between the first (1985-1990, left) and second (2010-2015, right) breeding bird atlas periods.
- Figure 10. Comparison of the range of the Rusty Blackbird in the Maritimes between the first (1985-1990) and second (2010-2015) breeding bird atlas periods.
- Figure 11. Annual abundance index of the Rusty Blackbird in Quebec between 1975 and 2013 based on the ÉPOQ database.
List of tables
- Table 1. estimated population size of the rusty blackbird in Canada based on breeding bird survey, atlas and nt/nu data (partners in flight science committee 2013).
- Table 2. annual population trends of rusty blackbird in Canada, based on the breeding bird survey for the long-term (1970-2014) and 10-year short-term (2004-2014) periods, along with the lower and upper limits of the 95% credible intervals. results in bold are statistically significant declines (i.e., 95% credible intervals do not overlap zero; a. smith unpubl. data).
- Table 3. short- and long-term population trends and 95% credible intervals (cis) of migrating rusty blackbirds counted at migration monitoring stations in eastern Canada (cmmn 2016). trends in bold are statistically significant.
- Table 4. natureserve conservation status ranks for rusty blackbird (natureserve 2015)
List of appendices
Document information
COSEWIC Assessment and status report on the Rusty Blackbird (Euphagus carolinus) in Canada, 2017

COSEWIC
Committee on the Status
of Endangered Wildlife
in Canada

COSEPAC
Comité sur la situation
des espèces en péril
au Canada
Committee on the Status of Endangered Wildlife in Canada (COSEWIC) status reports are working documents used in assigning the status of wildlife species suspected of being at risk. This report may be cited as follows:
COSEWIC. 2017. COSEWIC assessment and status report on the Rusty Blackbird Euphagus carolinus in Canada. Committee on the Status of Endangered Wildlife in Canada. Ottawa. xi + 64 pp. (Species at Risk Public Registry website).
Previous report(s):
COSEWIC 2006. COSEWIC assessment and status report on the Rusty Blackbird Euphagus carolinus in Canada. Committee on the Status of Endangered Wildlife in Canada. Ottawa. vi + 28 pp.
Production note:
COSEWIC acknowledges Dendroica Environnement et Faune (Carl Savignac) for writing the status report on the Rusty Blackbird, Euphagus carolinus, in Canada, prepared with the financial support of Environment and Climate Change Canada. This report was overseen and edited by Jon McCracken and Richard Elliot, Co-chairs of the COSEWIC Birds Specialist Subcommittee.
For additional copies contact:
COSEWIC Secretariat
c/o Canadian Wildlife Service
Environment Canada
Ottawa, ON
K1A 0H3
Tel.: 819-938-4125
Fax: 819-938-3984
E-mail: COSEWIC E-mail
Website: COSEWIC
Également disponible en français sous le titre Ếvaluation et Rapport de situation du COSEPAC sur le Quiscale rouilleux (Euphagus carolinus) au Canada.
Cover illustration/photo:
Rusty Blackbird - Photo by Carl Savignac.
COSEWIC assessment summary
Assessment summary - April 2017
- Common name
- Rusty Blackbird
- Scientific name
- Euphagus carolinus
- Status
- Special concern
- Reason for designation
- Factors that threaten the persistence of this species in Canada have not been reversed or effectively managed since it was assessed as Special Concern in 2006. This species experienced steep population declines through the twentieth century, which may have stabilized recently. This may only be a temporary reprieve, as many important threats contributing to these declines have not been corrected, particularly on the U.S. wintering range. These problems include loss and degradation of wintering habitat due to wetland conversion and dam construction, blackbird control programs in agricultural areas, and impacts from the use of agricultural pesticides. Continuing threats on Canadian breeding grounds include mercury contamination and degradation of wetland habitat due to warming, acidification, and drying climates.
- Occurrence
- Yukon, Northwest Territories, Nunavut, British Columbia, Alberta, Saskatchewan, Manitoba, Ontario, Quebec, New Brunswick, Prince Edward Island, Nova Scotia, Newfoundland and Labrador.
- Status history
- Designated Special Concern in April 2006. Status re-examined and confirmed in April 2017.
COSEWIC executive summary
Rusty Blackbird
Euphagus carolinus
Wildlife Species Description and Significance
The Rusty Blackbird (Euphagus carolinus) is a medium-sized songbird. Both sexes have pale yellow eyes and a black, slightly curved bill. During the breeding season, the male is uniformly black, with a faint greenish gloss to its body and slight violet gloss to its head and neck. The female is brownish grey with no gloss. In winter, the plumage of both sexes is more rust-coloured. In the western provinces, Rusty Blackbirds can easily be confused with Brewer’s Blackbirds (E. cyanocephalus), which have stouter bills, longer legs and tail, and glossier plumage. Because 85% of the Rusty Blackbird’s breeding range is located in Canada, it is a species for which Canada has a major conservation responsibility.
Distribution
The Rusty Blackbird has a wide distribution across boreal regions of Canada, and breeds in all Canadian provinces and territories. The winter range includes most of the central and eastern United States, although it also winters irregularly in extreme southern Canada.
Habitat
The breeding range of the Rusty Blackbird in Canada is almost entirely within the boreal forest. Breeding habitat there is characterized by coniferous-dominated forests adjacent to wetlands, such as slow-moving streams, peat bogs, sedge meadows, marshes, swamps and beaver ponds. On migration, the Rusty Blackbird is primarily associated with wooded wetlands. In winter, it occurs primarily in lowland forested wetlands, cultivated fields and pecan groves. Suitable habitat for the species appears to be decreasing on its breeding range and wintering grounds, due mainly to the loss and degradation of wetlands by human activities.
Biology
Rusty Blackbirds nest in isolated pairs on the margins of wetlands, but sometimes in loose colonies. Nests are located in shrubs or small trees over or near water. The female lays three to seven eggs. Incubation lasts 14 days, and the chicks remain in the nest for about 13 days. Birds from eastern Canada winter mostly east of the Appalachian Mountains, while those breeding in central and western Canada winter mostly in the Mississippi Valley.
Population Sizes and Trends
The Canadian breeding population, which includes approximately 87% of the global population, is estimated at 4.4 million birds. According to the Christmas Bird Count and the Breeding Bird Survey, the Rusty Blackbird has suffered one of the greatest population declines of birds in Canada. It has seen an annual rate of decline of 5.5% and 6.3% in these surveys, respectively, for a total reduction of the population of 85-90% since 1970. This decline appears to have begun as early as the 1920s. However, analyses of short-term trends in Canada indicate that the population has been fairly stable between 2004 and 2014.
Threats and Limiting Factors
The most serious threats to the Rusty Blackbird are thought to be on the wintering grounds in the southeastern U.S. They include loss and degradation of wintering habitat as a result of the conversion of wetlands for agricultural and residential purposes, changes in surface hydrology due to damming activities, large-scale blackbird control programs in agricultural areas, and mortality due to agricultural pesticides. On the breeding grounds, threats include contamination of wetlands by mercury, wetland acidification, and wetland degradation due to climate change.
Protection, Status and Ranks
Although the Rusty Blackbird is a migratory bird, it is not protected in Canada under the Migratory Birds Convention Act, 1994. It is currently listed as a species of special concern on Schedule 1 of the Species at Risk Act. It receives general protection under wildlife legislation of all provinces and territories, and three provinces have listed it under species-at-risk legislation: New Brunswick, Nova Scotia, and Newfoundland and Labrador. In Quebec, the Rusty Blackbird is on the list of wildlife species likely to be designated as threatened or vulnerable.
Technical summary
- Scientific name:
- Euphagus carolinus
- English name:
- Rusty Blackbird
- French name:
- Quiscale rouilleux
- Range of occurrence in Canada:
- Yukon, Northwest Territories, Nunavut, British Columbia, Alberta, Saskatchewan, Manitoba, Ontario, Quebec, New Brunswick, Prince Edward Island, Nova Scotia, Newfoundland and Labrador
Demographic information
| Summary items | Information |
|---|---|
| Generation time (usually average age of parents in the population) | 2 to 3 years |
| Is there an [observed, inferred, or projected] continuing decline in number of mature individuals? | No; declines may have levelled off between about 2004 - 2014 |
| Estimated percent of continuing decline in total number of mature individuals within [5 years or 2 generations | Unknown; no statistically significant change observed |
| [Observed, estimated, inferred, or suspected] percent [reduction or increase] in total number of mature individuals over the last [10 years, or 3 generations]. | No statistically significant change observed, estimated, inferred, or suspected |
| [Projected or suspected] percent [reduction or increase] in total number of mature individuals over the next [10 years, or 3 generations]. | Unknown |
| Observed, estimated, inferred, or suspected] percent [reduction or increase] in total number of mature individuals over any [10 years, or 3 generations] period, over a time period including both the past and the future. | Unknown |
| Are the causes of the decline a. clearly reversible and b. understood and c. ceased? | a. No b. No; largely unknown c. No; may not have ceased |
| Are there extreme fluctuations in number of mature individuals? | No |
Extent and occupancy information
| Summary items | Information |
|---|---|
| Estimated extent of occurrence (EOO) | 9.9 million km2; based on a minimum convex polygon of the species’ range map (see Figure 3). |
| Index of area of occupancy (IAO) | Unknown, but much greater than 2000 km2 |
| Is the population “severely fragmented” i.e., is >50% of its total area of occupancy in habitat patches that are (a) smaller than would be required to support a viable population, and (b) separated from other habitat patches by a distance larger than the species can be expected to disperse? | a. No b. No |
| Number of “locations” See Definitions and Abbreviations on COSEWIC website and IUCN (Feb 2014) for more information on this term.(use plausible range to reflect uncertainty if appropriate) |
Unknown, but much greater than 10 |
| Is there an [observed, inferred, or projected] decline in extent of occurrence? | Unknown |
| Is there an [observed, inferred, or projected] decline in index of area of occupancy? | Possibly inferred; although apparent declines in the south may be balanced by increases in the north. |
| Is there an [observed, inferred, or projected] decline in number of subpopulations? | N/A |
| Is there an [observed, inferred, or projected] decline in Number of “locations” See Definitions and Abbreviations on COSEWIC website and IUCN (Feb 2014) for more information on this term. ? |
N/A |
| Is there an [observed, inferred, or projected] decline in [area, extent and/or quality] of habitat? | Yes; observed decline in quality of habitat |
| Are there extreme fluctuations in number of subpopulations? | No |
| Number of “locations” See Definitions and Abbreviations on COSEWIC website and IUCN (Feb 2014) for more information on this term. ? |
No |
| Are there extreme fluctuations in extent of occurrence? | No |
| Are there extreme fluctuations in index of area of occupancy? | No |
Number of mature individuals (in each population)
| Subpopulations (give plausible ranges) | N Mature Individuals |
|---|---|
| Total | 4.4 million; based on Partners in Flight estimate (see Abundance section) |
Quantitative analysis
| Summary items | Information |
|---|---|
| Is the probability of extinction in the wild at least [20% within 20 years or 5 generations, or 10% within 100 years]? | Unknown; relevant quantitative analysis not conducted |
Threats (direct, from highest impact to least, as per IUCN threats calculator)
| Summary items | Information |
|---|---|
| Was a threats calculator completed for this species? | Yes, on 5 May 2016 (Suzanne Carrière, Kaytlin Cooper, Bruno Drolet, Richard Elliot, Dave Fraser, Marcel Gahbauer, Joanna James, Jeff Keith, Jon McCracken, Carl Savignac, Pam Sinclair, Karen Timm, Steve Van Wilgenberg, Alana Westwood) Overall threat level: Medium. The most significant direct threats identified include:
Other likely threats include climate change effects on wetland hydrology and changes in the timing of insect emergence on the breeding grounds. What additional limiting factors are relevant? N/A |
Rescue effect (immigration from outside Canada)
| Summary items | Information |
|---|---|
| Status of outside population(s) most likely to provide immigrants to Canada. | The U.S. has less than 15% of the global breeding population, and declining habitat quality. U.S. population trends are difficult to evaluate statistically, but are probably declining. |
| Is immigration known or possible? | Yes; possible |
| Would immigrants be adapted to survive in Canada? | Yes |
| Is there sufficient habitat for immigrants in Canada? | Likely; assuming that habitat remains stable |
| Are conditions deteriorating in Canada? | Yes; habitat quality is decreasing |
| Are conditions for the source population deteriorating? | Yes; habitat quality is decreasing in Alaska and northeastern U.S. |
| Is the Canadian population considered to be a sink? | No |
| Is rescue from outside populations likely? | No |
Data sensitive species
| Summary items | Information |
|---|---|
| Is this a data sensitive species? | No |
Status history
| Summary items | Information |
|---|---|
| COSEWIC: Designated Special Concern in April 2006. | Status re-examined and confirmed in April 2017. |
| blank cell | blank cell |
Status and reasons for designation
| Summary items | Information |
|---|---|
| Status | Special Concern |
| Alpha-numeric codes | N/A |
| Reasons for designation | Factors that threaten the persistence of this species in Canada have not been reversed or effectively managed since it was assessed as Special Concern in 2006. This species experienced steep population declines through the twentieth century, which may have stabilized recently. This may only be a temporary reprieve, as many important threats contributing to these declines have not been corrected, particularly on the U.S. wintering range. These problems include loss and degradation of wintering habitat due to wetland conversion and dam construction, blackbird control programs in agricultural areas, and impacts from the use of agricultural pesticides. Continuing threats on Canadian breeding grounds include mercury contamination and degradation of wetland habitat due to acidification, warming, and drying climates. |
Applicability of criteria
| Summary items | Information |
|---|---|
| Criterion A (Decline in Total Number of Mature Individuals) | Not applicable; estimated reduction in total number of mature individuals does not meet thresholds. |
| Criterion B (Small Distribution Range and Decline or Fluctuation) | Not applicable; range exceeds all thresholds. |
| Criterion C (Small and Declining Number of Mature Individuals) | Not applicable; number of mature individuals exceeds all thresholds. |
| Criterion D (Very Small or Restricted Population) | Not applicable; number of mature individuals exceeds all thresholds. |
| Criterion E (Quantitative Analysis) | Analysis not conducted. |
Preface
During the past decade, new research efforts for the Rusty Blackbird have been initiated, largely by the International (Canada-U.S.A.) Rusty Blackbird Working Group. These efforts have focused on documenting: 1) habitat use and reproductive success on the breeding grounds; 2) distribution and habitat use on the wintering grounds and at migratory stopover sites; 3) migratory connectivity between breeding and wintering regions; 4) threats and limiting factors on breeding and wintering grounds in the U.S.; and 5) population trends on the breeding grounds.
Work on the breeding grounds was mainly conducted in Alaska and New England. Data on habitat use on the wintering grounds were collected in the Mississippi alluvial valley and on the Atlantic coastal plain. Since the 2006 species’ assessment, significant knowledge on threats and limiting factors was gained throughout the species’ breeding and wintering ranges, with a general focus on the impacts of climate change, contamination by mercury and pesticides, forest management practices, and nest parasitism. Rusty Blackbird population trends in Canada were studied in the Northwest Territories, where shifts in abundance between the 1970s and 2006 were documented, and in Quebec using migration count data from Observatoire d’oiseaux de Tadoussac.
COSEWIC history
The Committee on the Status of Endangered Wildlife in Canada (COSEWIC) was created in 1977 as a result of a recommendation at the Federal-Provincial Wildlife Conference held in 1976. It arose from the need for a single, official, scientifically sound, national listing of wildlife species at risk. In 1978, COSEWIC designated its first species and produced its first list of Canadian species at risk. Species designated at meetings of the full committee are added to the list. On June 5, 2003, the Species at Risk Act (SARA) was proclaimed. SARA establishes COSEWIC as an advisory body ensuring that species will continue to be assessed under a rigorous and independent scientific process.
COSEWIC mandate
The Committee on the Status of Endangered Wildlife in Canada (COSEWIC) assesses the national status of wild species, subspecies, varieties, or other designatable units that are considered to be at risk in Canada. Designations are made on native species for the following taxonomic groups: mammals, birds, reptiles, amphibians, fishes, arthropods, molluscs, vascular plants, mosses, and lichens.
COSEWIC membership
COSEWIC comprises members from each provincial and territorial government wildlife agency, four federal entities (Canadian Wildlife Service, Parks Canada Agency, Department of Fisheries and Oceans, and the Federal Biodiversity Information Partnership, chaired by the Canadian Museum of Nature), three non-government science members and the co-chairs of the species specialist subcommittees and the Aboriginal Traditional Knowledge subcommittee. The Committee meets to consider status reports on candidate species.
Definitions (2016)
- Wildlife species
- A species, subspecies, variety, or geographically or genetically distinct population of animal, plant or other organism, other than a bacterium or virus, that is wild by nature and is either native to Canada or has extended its range into Canada without human intervention and has been present in Canada for at least 50 years.
- Extinct (X)
- A wildlife species that no longer exists.
- Extirpated (XT)
- A wildlife species no longer existing in the wild in Canada, but occurring elsewhere.
- Endangered (E)
- A wildlife species facing imminent extirpation or extinction.
- Threatened (T)
- A wildlife species likely to become endangered if limiting factors are not reversed.
-
Special concern (SC)
(Note: Formerly described as “Vulnerable” from 1990 to 1999, or “Rare” prior to 1990.) - A wildlife species that may become a threatened or an endangered species because of a combination of biological characteristics and identified threats.
-
Not at risk (NAR)
(Note: Formerly described as “Not in any category”, or “No designation required.”) - A wildlife species that has been evaluated and found to be not at risk of extinction given the current circumstances.
-
Data deficient (DD)
(Note: Formerly described as “Indeterminate” from 1994 to 1999 or “ISIBD” [insufficient scientific information on which to base a designation] prior to 1994. Definition of the [DD] category revised in 2006.) - A category that applies when the available information is insufficient (a) to resolve a species’ eligibility for assessment or (b) to permit an assessment of the species’ risk of extinction.
The Canadian Wildlife Service, Environment and Climate Change Canada, provides full administrative and financial support to the COSEWIC Secretariat.
Wildlife species description and significance
Name and classification
The English name for Euphagus carolinus is the Rusty Blackbird. The French name is Quiscale rouilleux. Its taxonomy is as follows:
Class: Aves
Order: Passeriformes
Family: Icteridae
Genus: Euphagus
Species: Euphagus carolinus
Two species of blackbirds of the genus Euphagus occur in North America: Rusty Blackbird and Brewer’s Blackbird (E. cyanocephalus; American Ornithologists’ Union [AOU] 1957).
Morphological description
The Rusty Blackbird is a medium-sized songbird (body length: 21-25 cm; Godfrey 1986; Figure 1). Both sexes have pale yellow eyes, black bills, and black feet. During the breeding season, the male is uniformly black, with a faint greenish gloss on the body and a slight violet gloss on the head and neck. The plumage differs in the fall, with rust-coloured edges of the tertiary feathers, scapulars, wing coverts and the head, breast and back feathers (Pyle 1997). During the breeding season, the plumage of the female is brownish grey, with no gloss (Pyle 1997). The winter plumage of the female is generally rust coloured, with a grey rump. In fall, immature birds are similar to adults (Pyle 1997), although juveniles have a dark iris.
In summer, the Rusty Blackbird can be confused with the Brewer’s Blackbird, which is similar in size and colour (Avery 2013). The plumage of the male Brewer’s Blackbird is glossier than that of the Rusty Blackbird, and the former has a shorter, straighter, and thicker bill. The Brewer’s Blackbird’s legs and tail are also generally longer than those of the Rusty Blackbird. In summer, the females differ in the colouring of their iris; the Rusty Blackbird’s iris is yellow, whereas the Brewer’s Blackbird’s is dark. In mixed flocks of migrating and wintering blackbirds, Rusty Blackbirds can also be confused with longer-tailed Common Grackles (Quiscalus quiscula).

Long description for Figure 1
Photo of a male Rusty Blackbird in breeding plumage. This bird is uniformly black, with a faint greenish gloss to its body and slight violet gloss to its head and neck.
Population spatial structure and variability
Few molecular or genetic studies have been conducted on the relationships between the Rusty Blackbird and other species. Sequencing of mitochondrial cytochrome-b suggests that Rusty Blackbird is most phylogenetically related to the Brewer’s Blackbird (Lanyon and Omland 1999). Otherwise, there is no information on spatial structure or variability for the Rusty Blackbird.
Designatable units
There are two recognized subspecies of Rusty Blackbird. E. c. carolinus occurs throughout most of the species’ range and winters from southern British Columbia east to southern Ontario and south (generally east of the Great Plains) to eastern Texas and northern Florida. E. c. nigrans (Burleigh and Peters 1948) breeds in Newfoundland and Labrador, Nova Scotia, the Magdalen Islands in Quebec and possibly eastern New Brunswick (AOU 1957; Godfrey 1986), and apparently winters in the Carolinas and Georgia (AOU 1957), likely overlapping with wintering grounds of E. c. carolinus. Compared to E. c. carolinus, male E. c. nigrans are generally darker on the body and have a blue sheen to the head, while breeding females are darker grey (Peters and Burleigh 1951), with darker rusty feather edgings in autumn. Overall, differences in morphology between the two subspecies are subtle when individual variation is taken into consideration (Pyle 1997).
This status report considers E. carolinus as a single designatable unit, as the weak scientific evidence for two distinct subspecies (i.e., minor differences in plumage) with only limited segregation on the breeding and wintering ranges indicates that differences between the subspecies are insufficiently discrete and evolutionarily significant for them to be considered as separate DUs.
Special significance of the species
There are no studies that demonstrate the economic importance of the Rusty Blackbird. On its wintering grounds, the Rusty Blackbird often mixes with flocks of other species of blackbirds, in roosts containing many thousands of birds, which can cause a nuisance to human infrastructure due to the accumulation of droppings, as well as depredation of certain crops (Dolbeer et al. 1997; Avery 2013). Along with other blackbirds, it has long been considered a nuisance species, though for the Rusty Blackbird this reputation is largely ill-deserved. In terms of sources of Aboriginal Traditional Knowledge relating to the Rusty Blackbird, a study of the available sources did not find any in Canada (Chiblow 2013).
Distribution
Global range
The Rusty Blackbird is an exclusively North American species. Its breeding range includes all of the Canadian provinces and territories, most of Alaska, and some parts of Minnesota, Michigan, Vermont, New Hampshire, Maine, New York and Massachusetts (Avery 2013; Figure 2).
The winter range includes Massachusetts, New York, Pennsylvania, Virginia, Ohio, Michigan, Wisconsin, Iowa, Kansas, Oklahoma, and the Atlantic coast states (Figure 2). Core wintering areas are thought to occur within the southern portion of the Mississippi Valley, as well as the Coastal Plain to the east of the Appalachians (Niven et al. 2004; Hamel and Ozdenerol 2008; Greenberg et al. 2011; Avery 2013). High concentrations are also present in winter in Alabama, Georgia, Texas and North and South Carolina (Smithsonian Institution 2015). Small numbers also winter locally and sporadically in the southern part of most Canadian provinces (Avery 2013; Figure 2).
Canadian range
The Canadian range of the Rusty Blackbird extends from Newfoundland and Labrador to the Yukon, and includes all Canadian provinces and territories (Avery 2013; Figure 3). The northern range limit is delineated by the Old Crow region in northern Yukon (Sinclair et al. 2003), the Mackenzie River delta, Great Bear Lake and Great Slave Lake in the Northwest Territories (Norment et al. 1999; Avery 2013), the southern Kivalliq region in Nunavut (eBird Canada 2015), the south shore of Hudson Bay from Manitoba to Ontario (Cadman et al. 2007; BBAMB 2015), Guillaume-Delisle Lake and Kuujjuaq in northern Quebec (BBAQ 2015), Davis Inlet in Labrador, and the north coast of Newfoundland (eBird Canada 2015). The southern limit of its Canadian range includes south-central British Columbia east of the Coast Mountains, central Alberta, central Saskatchewan, southern Manitoba (e.g., Delta Beach), southern Ontario (e.g., Bruce Peninsula), southern Quebec (e.g., the Outaouais and Eastern Townships), and southern New Brunswick and Nova Scotia (Figure 3).
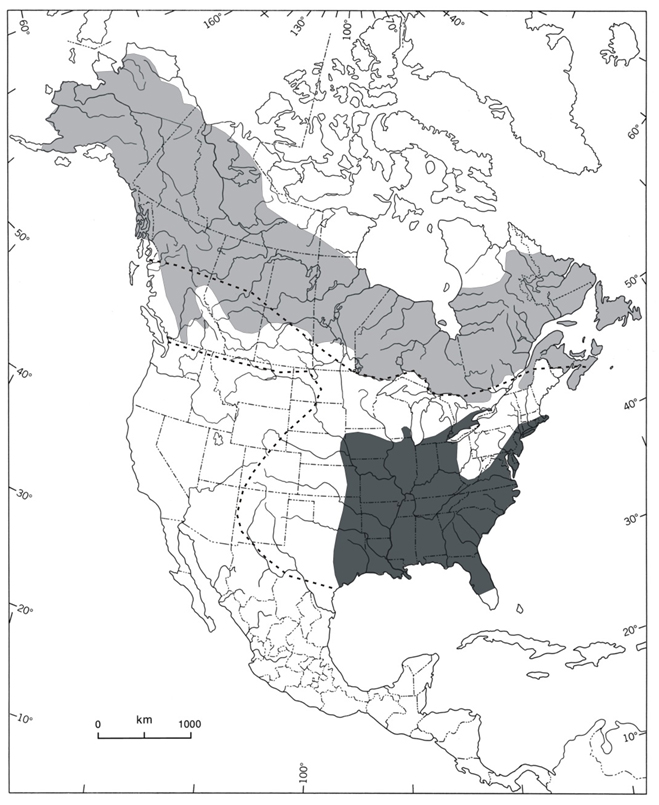
Long description for Figure 2
Map of the North American distribution of the Rusty Blackbird in the breeding and wintering seasons. The breeding range includes all of the Canadian provinces and territories, most of Alaska, and some parts of Minnesota, Michigan, Vermont, New Hampshire, Maine, New York, and Massachusetts. The winter range includes Massachusetts, New York, Pennsylvania, Virginia, Ohio, Michigan, Wisconsin, Iowa, Kansas, Oklahoma, and the Atlantic coast states.
Extent of occurrence and area of occupancy
The Canadian extent of occurrence, based on a minimum convex polygon of the species’ range, covers roughly 9.9 million km2 (Figure 3). This value is significantly higher than that initially calculated by COSEWIC (2006) and is explained in part by the large number of atlas squares with records in northern Quebec identified during the work of the second Quebec Breeding Bird Atlas (BBAQ 2015), as well as the availability of northern records elsewhere in Canada from eBird Canada (2015). The biological area of occupancy of the Rusty Blackbird is about 242,000 km2. This value is based on an average territory size of 0.11 km2 (see Home Range and Territory section), multiplied by a population estimate of 2.2 million breeding pairs (Partners in Flight Science Committee 2013). The index of the area of occupancy based on a 2 km x 2 km grid intersecting the species’ known areas of occupancy cannot be calculated due to lack of detailed information on all breeding sites, but it certainly exceeds COSEWIC’s minimum threshold value of 2000 km2.
The range of the Rusty Blackbird has undergone a contraction in the southern portions of several Canadian provinces, including Alberta, Saskatchewan and Manitoba (Federation of Alberta Naturalists 2007; Greenberg et al. 2011; BBAMB 2015; Government of Saskatchewan 2015). Powell (2008) also notes a 160-km contraction of the species’ breeding range in Maine since 1983, and breeding bird atlas data in Vermont also suggest a reduction in the breeding range in that state (Fisher and Powell 2013). The comparison of recent breeding bird atlas work carried out in the 2010s with corresponding data compiled in the 1980s in Ontario, Quebec and the Maritimes suggests a range contraction in southern Ontario, Quebec, New Brunswick and Nova Scotia (Cadman et al. 2007; BBAQ 2015; Stewart et al. 2015). However, the contraction of the range may be less significant in the Canadian northwest, particularly along the Mackenzie River valley, where occupancy of wetlands monitored in the early 1970s and in 2006 has remained the same (Machtans et al. 2007).
Search effort
The breeding range of the Rusty Blackbird in Canada has been determined mainly on the basis of work carried out in connection with various breeding bird atlases (Sinclair et al. 2003; Cadman et al. 2007; Federation of Alberta Naturalists 2007; BBABC 2015; BBAMB 2015; BBAQ 2015; Government of Saskatchewan 2015; Stewart et al. 2015) as well as from data from eBird Canada (2015) and the Boreal Avian Modeling Project (BAM 2015). Even so, data gaps remain in the northern portions of the species’ breeding range, including northern British Columbia where much of the population occurs in inaccessible higher elevation wetlands (David Fraser pers. comm.).
For the wintering grounds, an analysis of Christmas Bird Count (CBC) data for the period 1946-2001 made it possible to determine the general distribution as well as concentration areas (Hamel and Ozdenerol 2008). The International Rusty Blackbird Working Group(IRBWG) was established in 2005 (Greenberg et al. 2011; International Rusty Blackbird Working Group 2013). In conjunction with the eBird program, the IRBWG conducted the Rusty Blackbird Winter Hotspot Blitz, a 2-week period (January 31 through February 14) of surveys covering the eastern United States between 2009 and 2011. This program collected information on the species’ concentration areas on its wintering grounds (Evans et al. 2014) and tracked broad-scale migration patterns (LaSorte et al. 2016). In 2014, the IRBWG also initiated the Rusty Blackbird Spring Migration Blitz, a 3-year survey that relies on the efforts of amateur birders in all the Canadian provinces and territories (except Nunavut) and 38 American states, between early March and mid-June.
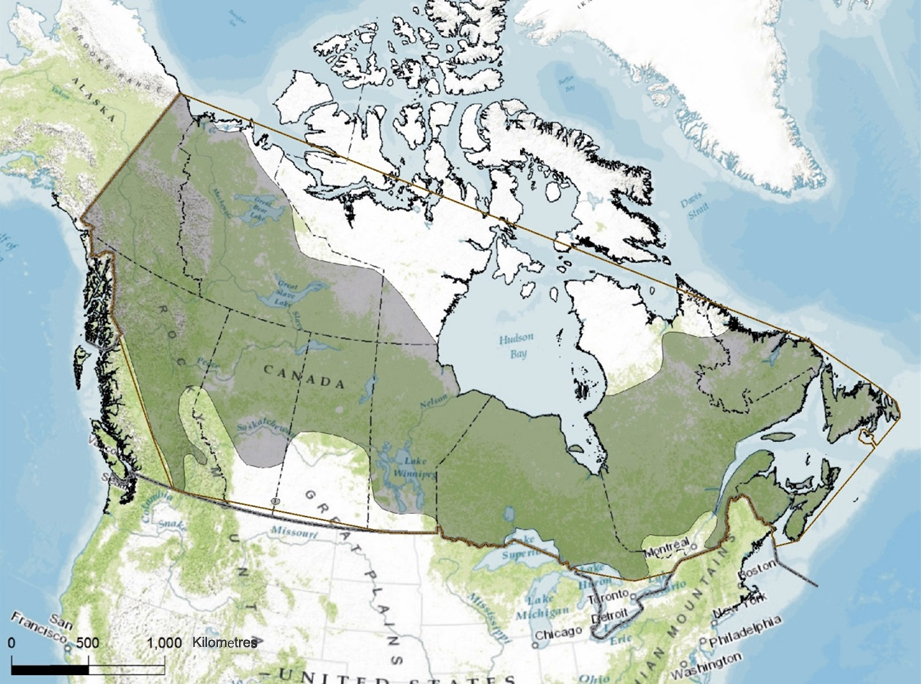
Long description for Figure 3
Map illustrating the breeding range of the Rusty Blackbird in Canada, which extends from Newfoundland and Labrador to Yukon and includes all Canadian provinces and territories.
Habitat
Habitat requirements
Breeding habitat
In Canada, the breeding range of the Rusty Blackbird overlaps almost entirely with the boreal forest and taiga terrestrial ecozones (Erskine 1992; Gauthier and Aubry 1996; Campbell et al. 1997; Sinclair et al. 2003; Cadman et al. 2007; Avery 2013; Boreal Avian Modeling Project 2015). Within these ecozones, the Rusty Blackbird occurs in sedge meadows, beaver ponds, muskegs, swamps, scrub riparian habitats of islands, lakes, rivers and streams, as well as alder and willow thickets (DesGranges and Houde 1989; Darveau et al. 1995; Larue et al.1995; Gauthier and Aubry 1996; Campbell et al. 1997; Whitaker and Montevecchi 1999; Sinclair et al. 2003; Machtans et al. 2007; Matsuoka et al. 2008, 2010a,b; Powell et al. 2010a,b; McCormack 2012; Scarl 2013).
In the southeastern part of its breeding range, the Rusty Blackbird has also been recorded nesting in early successional habitats created by natural disturbances such as fire and windthrow (Ellison 1990; Powell et al. 2010b). In Nova Scotia, and more specifically Cape Breton, it also occasionally occurs in drier environments, such as pasture (Erskine 1992). In the Northwest Territories, the Rusty Blackbird mainly occupies lake edges (Machtans et al. 2007). In the northwestern portion of its range, the parameters that best predict breeding habitat are the regional proportion of freshwater ponds and lakes and the presence of shallow wetlands with emergent vegetation (Matsuoka et al. 2008, 2010b). This species generally avoids dry upland forests for breeding (DesGranges and Houde 1989; Larue et al. 1995; Whitaker and Montevecchi 1999; Matsuoka et al. 2010a). In New England, wetland occupancy was best explained by variables describing Rusty Blackbird foraging habitat (shallow water ponds), nesting habitat (adjacent coniferous uplands), and evidence of beavers (Powell et al. 2014).
At the microhabitat scale, the Rusty Blackbird selects breeding sites in areas dense in regenerating conifers 1-3 m in height, but generally with an open canopy (Ellison 1990; Matsuoka et al. 2010b; Powell et al. 2010b; Buckley 2013). In Alaska, it nests in sites dominated by Black Spruce (Picea mariana) or willows (Salix sp.; Matsuoka et al. 2010a).
Rusty Blackbirds forage in shallow water, where they probe for aquatic prey in leaf litter (Avery 2013; Powell et al. 2014). It is strongly suspected that the area of shallow water (≤ 6 cm deep) is an important driver of habitat selection. Shallow water unconnected to flowing tributaries often teems with prey items (e.g., tadpoles and Odonates; L. Powell pers. obs.), presumably because of a lack of predatory fish and evaporation by seasonal drying. In New England, amphibian eggs and larvae, as well as invertebrate larvae, likely provide an easily accessed, calorie-rich food source in sites with shallow water (Powell et al. 2014).
Creation of wetlands by beavers is known to influence invertebrate community structure by replacing fast running-water taxa by shallow-water taxa such as dragonflies (Naiman et al. 1988), one of the main prey items consumed by Rusty Blackbirds (Avery 2013). Powell et al. (2014) suggest that beaver presence increases the probability of Rusty Blackbird occupancy. Beavers create both foraging and nesting habitat for Rusty Blackbirds by selectively removing hardwoods, which increases light gaps and encourages growth of trees such as spruces (Johnston and Naiman 1990). McDowell and Naiman (1986) also found that beavers increase the density of coarse particulate organic matter and the biomass of invertebrates by 2–5 times, and specifically increased the abundance of four of the Rusty Blackbird’s main prey items: dragonflies (Odonata), damselflies (Odonata), caddisflies (Trichoptera), and snails (Gastropoda) (Avery 2013).
Predictive models of the relative suitability of Rusty Blackbird breeding habitat indicate that habitat suitability increases with latitude (Figure 4; Boreal Avian Modeling Project 2015).
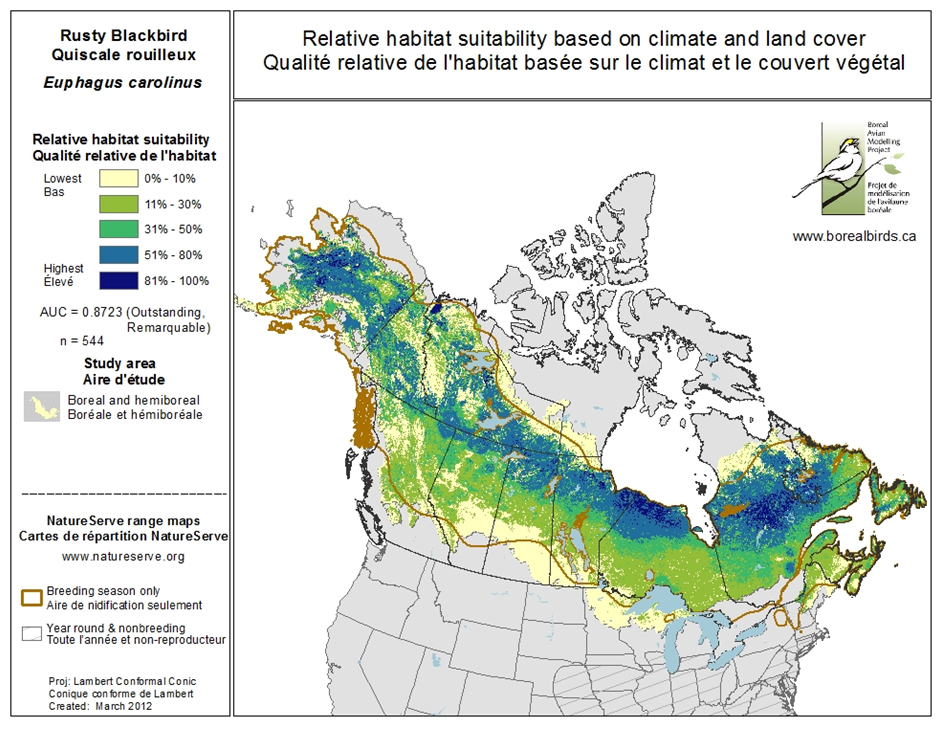
Long description for Figure 4
Map illustrating the relative suitability of Rusty Blackbird breeding habitat in Canada. Five habitat suitability levels are indicated, with habitat suitability generally increasing with latitude.
Migration and wintering habitat
On migration, the Rusty Blackbird is associated with wooded wetlands, such as flooded forests, beaver ponds and scrub along the edges of lakes, rivers and streams (Cyr and Larivée 1995; Campbell et al. 1997; Avery 2013). It occurs to a lesser degree in human-made habitats, such as pastures, ploughed fields, sewage treatment ponds and small landfill sites (Campbell et al. 1997; Sinclair et al. 2003; Avery 2013; Canham 2014). In Missouri, most reports of migrating Rusty Blackbirds are from herbaceous-dominated wetlands, small water bodies, grasslands and agricultural crops (Canham 2014). Three Rusty Blackbirds fitted with geolocators migrated from Alaska to the Great Plains, using the following habitats during their migration: cropland (60%), grasslands (36%), water bodies (2%) wetlands and other habitats (1%) (Johnson et al. 2012).
On wintering sites, the Rusty Blackbird occupies a wide variety of habitats, but is more abundant in semi-flooded continuous forest than in drier habitats (Dias 2008; Luscier et al. 2010). There is a positive association between flock size and the use of flooded lowland forests (Evans et al. 2014). In South Carolina, significant use of dry, wet and recently drained corn fields, as well as former rice field impoundments, was also observed (Dias 2008). In the Mississippi Valley, agricultural fields appear to be an important habitat for wintering Rusty Blackbirds, because they provide alternative food sources, such as remnant corn seeds, when aquatic prey and acorns and other nuts are not available (Luscier et al. 2010). Agricultural fields used by blackbirds are generally near wetlands (Luscier et al. 2010).
The species also makes significant use of Pecan (Carya illinoiensis) orchards and fragments of riparian forests (Mettke-Hofmann et al. 2015). Within these habitats, it feeds near edges of shallow water bodies, regardless of the density of the shrub cover or the distance from forest edges. In Louisiana, the presence of stagnant water and wet leaf litter appear to be the two main variables predicting the species’ occupation of a site (DeLeon 2014). It has also been observed that, in winter, the Rusty Blackbird selects habitats containing a high density of mature trees producing acorns and other nuts, as well as habitats containing regularly or permanently flooded wet soil with an adequate population of invertebrate prey (Newell 2013; Borchert and Stauffer 2014).
Unlike foraging habitats, winter roosts tend to be characterized by habitats containing dense vegetation near the ground, as well as fields with short vegetation or coniferous shrub and tree habitats in residential areas (Dias 2008). In South Carolina, roosts are mainly in cattail (Typha sp.) marshes adjacent to salt marshes (Dias 2008).
Segregation by sex and age apparently occurs on the wintering sites; females and young appear to winter farther south than older males (DeLeon 2012; Mettke-Hofmann et al. 2015). In addition, foraging sites used by males are characterized by greater availability and higher quality of food (Mettke-Hofmann et al. 2015; DeLeon 2012).
Habitat trends
Breeding habitat
Due to its close association with beaver ponds, Rusty Blackbird’s habitat might have been historically impacted by the large-scale loss of wetlands caused by commercial beaver harvesting activities, which drove that species almost to extinction across North America from the 17th century to the early 20th century (Novak 1987; Naiman et al. 1988). A study conducted in Elk Island National Park in Alberta over 54 years reported a 9-fold increase in open water area when compared to a period when beaver were absent. Since the 1950s, beaver populations seem to have recovered well across Canada (Hood and Bayley 2008) and this type of habitat for Rusty Blackbirds should therefore be increasing.
In parts of the species’ Canadian breeding range, large areas of wetlands have been lost through conversion for agricultural and industrial purposes (Greenberg and Droege 1999; Hobson et al. 2002; Greenberg and Matsuoka 2010; Greenberg et al. 2011). For example, Greenberg et al. (2011) estimated that more than 1 million ha of forested wetlands have been flooded in connection with the creation of hydroelectric reservoirs in northern Quebec. It is estimated that 73% of the boreal transition zone in Saskatchewan has been converted to agricultural land since European settlement (Hobson et al. 2002). Similar levels of habitat change are reported for Alberta (Young et al. 2006).
In terms of the impact of oil and gas exploration and extraction activities, it is estimated that, as of 2003, 8% of the boreal forest biome in Canada has been affected to a fairly significant degree (Greenberg et al. 2011). The drainage and pumping of enormous quantities of freshwater from groundwater and surface water reserves for the purposes of oil and gas extraction can also affect Rusty Blackbird habitat (Griffiths and Woynillowicz 2003). Rusty Blackbird habitat in Canada is believed to have decreased by 5% by 2000, and a further reduction of 4% by 2050 is projected (Global Forest Watch Canada 2000; Hayeur 2001; Griffiths and Woynillowicz 2003; Kling et al. 2003).
Climate change, which will result in a substantial increase in temperature in the boreal forest by 2100, is believed to be affecting the amount of potentially suitable breeding habitat. A study using satellite images covering the boreal, subarctic and arctic zone demonstrated that, between 2000 and 2009, the total area of shallow ponds and boreal lakes in Canada decreased by 6700 km2 (Carroll et al. 2011). This reduction in boreal wetlands is due in part to permafrost melting caused by the increase in temperature (Yoshikawa and Hinzman 2003). Several climate modelling scenarios based on change in the climate envelope and existing distribution of habitat predict a 64-90% reduction in core habitat for Rusty Blackbirds by the year 2100 (Stralberg et al. 2015a). Given the recent predictions of warming and drying trends for boreal regions, beavers will likely play an important role in maintaining open water and mitigating the impact of drought (Hood and Bayley 2008).
In summary, there have been declines in the total area of Rusty Blackbird habitat in the species’ Canadian breeding range in recent decades due to the expansion of agriculture in the southern boreal forest, wetland loss due to climate change and increasing numbers of industrial development projects in the boreal forest. Furthermore, future habitat will likely be reduced further due to the anticipated increase of the effects of climate change.
Wintering habitat
A large portion of the Rusty Blackbird’s winter range includes the floodplains of the Mississippi Valley from southern Illinois to the Louisiana coast (Hamel and Ozdenerol 2008; Avery 2013). Conversion of wetland forests in wintering areas in the southern United States is considered one of the most significant factors in the decline of this species (Greenberg and Droege 1999; Hamel et al. 2009). Forested wetlands have been converted to pine (Pinus sp.) plantations and residential development (Hamel et al. 2009; Greenberg and Matsuoka 2010). It is estimated that between 1780 and 1980, approximately 57% of wetlands in Rusty Blackbird wintering grounds were lost in the Mississippi Lowlands, while in the South Atlantic Coastal Plain, wetland loss was 36% (Dahl 1990).
Wetland loss in the southeastern United States between 1950 and 1980 owing to conversion to agricultural land or plantation forests was estimated at 1.3 million ha (Hefner et al. 1994) and is recognized as a significant factor in the decline of the Rusty Blackbird (Greenberg et al. 2011). According to Greenberg et al. (2011), prior to 1970, the forested wetlands of the Mississippi Valley were infrequently used for agriculture, because they were unprofitable owing to the high cost of land conversion. Beginning in 1970, however, higher market prices for soybeans made it profitable to convert these habitats to agricultural land. In the late 1980s, when soybean prices fell below the threshold of profitability, reforestation efforts were undertaken in the southeastern United States (King et al. 2006) and it is estimated that 162,000 ha of unprofitable agricultural land was converted back to forested wetland habitats between 1990 and 2005 (Ducks Unlimited 2007). At the same time, carbon sequestration programs were introduced, which also encouraged the restoration of large areas of forested wetlands in the southeastern United States (Houghton 2002; Shoch et al. 2009).
To conclude, despite some recent habitat gains, there has been a net habitat loss on the wintering grounds since European colonization.
Biology
Life cycle and reproduction
The Rusty Blackbird is usually monogamous, although polygamy exists (Scurr 2013). Pairs nest solitarily or rarely in loose colonies (Spindler and Kessel 1980; Orians 1985; Avery 2013). Generally, nesting begins in early May in the southern part of the breeding range and mid- to late May in the northern part, and continues until mid-July (Matsuoka et al. 2010b; Rousseu and Drolet 2015). Nests are typically placed in thickets of small conifers, deciduous shrubs or dead trees, usually over or close to water (Campbell et al. 1997; Matsuoka et al. 2008, 2010b). The Rusty Blackbird produces one clutch per year, but replacement nests may be built if the first nest is destroyed (Avery 2013).
The number of eggs per clutch is usually 3 to 7, with an average of 4.5 eggs per nest (Matsuoka et al. 2008; Avery 2013; Loomis, 2013). Incubation lasts 14 days (Matsuoka et al. 2010b). Young remain in the nest for 11-13 days (Gauthier and Aubry 1996; Campbell et al. 1997; Matsuoka et al. 2010b).
There are no Canadian studies of Rusty Blackbird nest survival. In Alaska, the daily survival rate of nests is generally high (0.98 ± 0.003), resulting in mean nest success of 0.56 (95% CI 0.47–0.64; Matsuoka et al. 2010b; Loomis 2013). In that state, out of 69 nests that survived through the incubation period, 90% (n= 373 eggs) hatched. In addition, out of 63 nests where at least one young had left the nest, 96% of 312 young survived to fledging. Of 150 nests whose survival was recorded, 96 (64%) fledged young and 54 (36%) failed (Matsuoka et al. 2010b). Of the nests that did not survive, 89% (48 nests) were lost to predators, four were abandoned for unknown reasons and two fell into the water (Matsuoka et al. 2010b). Generally, the average rate of nest success is similar for Alaska (56%; Matsuoka et al. 2010b) and New England (62%; Powell 2008), and much higher than nest success in other blackbird species (30-39%, Martin 1995).
Generation time for Rusty Blackbird is assumed to be 2 to 3 years (COSEWIC 2006), typical of medium-sized passerine birds.
Survival
There are no data on survival of adults on the breeding grounds. On wintering sites, survival of adults and immature individuals for a 12-week period was estimated at 0.84 and 0.53, respectively (Wohner et al. 2014). According to these authors, these survival rates are comparable to those of other North American songbirds. It is currently unclear why these relatively high survival rates on the wintering ground do not translate into an increase in population. Survival studies on the breeding grounds are needed in order to assess adult post-migration survival.
The oldest known individual was a banded bird recovered in 1939, with a life span of at least 8 years, 9 months (Cooke 1942, cited in Avery 2013).
Dispersal and migration
Depending on latitude, fall migration begins between late July and September (Campbell et al. 1997; Sinclair et al. 2003; Johnson et al. 2012; Scarl et al. 2014). In Canada, spring migration occurs principally between mid-March and the first week of May, with the bulk of the birds migrating during April (Campbell et al. 1997; Sinclair et al. 2003; Johnson et al. 2012). In the northern portion of the species’ breeding range in Canada, migrating groups of up to several hundred individuals have been observed (Manitoba Avian Research Committee 2003). In the United States, flock size ranges up to 275 individuals (Scarl et al. 2014), with the average size being 20- 25 birds (DeLeon 2012; Canham 2014).
Returns of banded birds in North America, as well as a study examining the stable-hydrogen isotope ratio of feathers, suggest that birds originating from Alaska to central Canada migrate southeastward toward the southern Mississippi Valley area, whereas populations breeding in northeastern regions (Quebec and the Maritimes) tend to winter in the Atlantic Coastal Plain (Brewer et al. 2006; Hobson et al. 2010). The Appalachians may form a natural barrier separating overwintering populations of Rusty Blackbirds (Hamel and Ozdenerol 2008).
Seventeen adult Rusty Blackbirds fitted with tracking devices (light-sensitive “geolocators”) in the fall migrated from Alaska to the southeastern United States, passing through and/or periodically stopping over in northeastern British Columbia, northwestern Alberta, central Saskatchewan, North Dakota, South Dakota and Iowa. After an average distance of 4400 km, these birds reached their wintering sites, which extended from South Dakota to northern Louisiana (Johnson et al. 2012). In spring, the route followed was generally the same as the previous fall for the three birds tracked. All three birds that were tracked into the spring returned to their previous breeding sites (Johnson et al. 2012).
An analysis of eBird data from Louisiana suggests that on the wintering grounds, the same foraging sites appear to be regularly used by groups of Rusty Blackbirds within and between wintering seasons (DeLeon 2012).
There are no data available on natal dispersal.
Diet and foraging behaviour
On the breeding grounds, Rusty Blackbirds feed mostly on invertebrates, particularly dragonfly nymphs (97%; Loomis 2013). They also eat salamanders, water beetles, spiders, small fish, crustaceans, snails, and mosquitoes (Cade 1953; Matsuoka et al. 2010a; Powell et al. 2010a; Avery 2013).
During fall and winter, the diet is more varied and is composed of aquatic invertebrates, seeds and berries (Avery 2013;Newell 2013;Mettke-Hofmann et al. 2015). Monitoring at two important wintering sites in South Carolina showed that the species foraged in dry, wet and recently drained corn fields, former rice field impoundments (drained or flooded) and forested wetlands (Dias 2008). Rusty Blackbirds also make irregular use of bird feeders in winter (Cyr and Larivée 1995; Campbell et al. 1997).
Acorns are also consumed, but must have had their husks removed by squirrels, or larger birds such as Common Grackle or Blue Jay (Cyanocitta cristata; Newell 2013). According to Newell (2013), Rusty Blackbirds wintering in South Carolina and Georgia often feed on fallen pecans. Owing to their high fat content (Fredrickson and Reid 1988), pecans are eaten during cold winters, and help blackbirds survive when acorns and wetland invertebrates are less available. In order to be consumed by Rusty Blackbirds, fallen pecans must first be crushed, for example by cars, or being stepped on by people or deer (Newell 2013). The Rusty Blackbird also feeds on residential lawns, where they consume mainly aquatic worms and earthworms (Newell 2013). Rusty Blackbirds that winter on the mountainous plateau areas of the Appalachians reportedly incorporate more acorns and other nuts in their diet than birds wintering on the Coastal Plain (34% vs 19%; Newell 2013).
Interspecific interactions
Predation of blackbird eggs by Red Squirrels (Tamiasciurus hudsonicus)and White-tailed Deer (Odocoileus virginianus) has been reported frequently (Buckley 2013). Aggressive behaviour observed toward the American Marten (Martes americana) suggests that this species is a potential predator (Avery 2013). Potential avian predators of nests, adults and fledglings include the Gray Jay(Perisoreus canadensis; Campbell et al. 1997), Black-billed Magpie (Pica hudsonia), Common Raven (Corvus corax), Bald Eagle (Haliaeetus leucocephalus), Northern Harrier (Circus cyaneus), Sharp-shinned Hawk (Accipiter striatus), Northern Goshawk (A. gentilis), Red-tailed Hawk(Buteo jamaicensis) and Merlin (Falco columbarius), as well as Blue Jay and American Crow (Corvus brachyrhynchos; Matsuoka et al. 2010b; Powell et al. 2010b; Buckley 2013).
Rusty Blackbirds might also be naturally limited by the occurrence of more dominant species such as the Red-winged Blackbird (Agelaius phoeniceus) and the Common Grackle on the breeding grounds (Ellison 1990; Erskine 1992). In Ontario, for example, areas where Red-winged Blackbirds and Common Grackles are absent have the highest density of Rusty Blackbirds (Cadman et al. 2007).
On its migration routes and wintering areas, the Rusty Blackbird regularly joins mixed flocks composed of other blackbirds, such as the Red-winged Blackbird, European Starling (Sturnus vulgaris) and Common Grackle (Dolbeer et al. 1997; Luscier et al. 2010; Greenberg et al. 2011; Avery 2013). The formation of mixed groups is believed to reduce the risk of predation to individual birds (Morse 1977).
Home range and territory
No studies concerning the size of the home range and territory of the Rusty Blackbird have been conducted in Canada. However, in Maine, average home range size was estimated at 37.5 +/-12.6 ha, and average territory size was estimated at 11.1 +/-2.8 ha (Powell et al. 2010a). Home range includes lowland forested habitats with scattered unconnected or connected wetlands and upland forest habitats with small wet patches (Powell et al. 2010a). Home range is larger than that of other blackbird species because Rusty Blackbirds often use multiple wetlands for foraging (Powell et al. 2010a). Rusty Blackbirds also nest in small loose colonies (Matsuoka et al. 2010b; Powell et al. 2010c). The formation of colonies is believed to provide better protection of nests against predators (Powell et al. 2010c). In addition, Powell et al. (2010a) suggest that adults nesting in colonies follow one another to feed on the unpredictable emergence of aquatic insect prey.
Behaviour and adaptability
Rusty Blackbirds will frequent wetlands disturbed by human activities (Darveau et al. 1995; Campbell et al. 1997; Whitaker and Montevecchi 1999; Powell et al. 2010b; Buckley 2013), sewage ponds in forest environments (R. Popko pers. comm., 2015), and banks of hydroelectric reservoirs (J. Gauthier pers. comm. 2015), but breeding productivity in human-transformed habitats is unknown in Canada.
The Rusty Blackbird appears to use habitats created by natural disturbances that occur frequently and on large spatial scales, including floods, insect outbreaks and forest fires (Ellison 1990; Machtans et al. 2007; Matsuoka et al. 2010a). Buckley (2013) and others have noted a strong association with the presence of beaver ponds.
Monitoring of migrating Rusty Blackbirds at Tadoussac on the Quebec north shore between 1996 and 2010 suggests that the species follows a 5-year abundance cycle in the boreal forest (Savard et al. 2011). This cycle appears to be positively correlated with the North Atlantic Oscillation (NAO), a climatic phenomenon that reportedly affects the 4-year cycle of small rodents in the boreal forest, which influences predator/prey cycles (Savard et al. 2011).
On its wintering grounds, the Rusty Blackbird regularly forages in former agricultural lands that have been restored to wetlands (Hamel et al. 2009), as well as on wet residential lawns and Pecan plantations (Newell 2013). The Rusty Blackbird appears to base its foraging behaviour on a complex interaction of previous, current and expected weather in the short term as well as on the availability of resources (Newell 2013). Hamel and Ozdenarol (2008) have also hypothesized that precipitation affects interannual variations in the distribution of the Rusty Blackbird in the Mississippi Valley.
Population sizes and trends
Sampling effort and methods
Christmas bird count (cbc)
The Christmas Bird Count documents Rusty Blackbird population trends in North America in winter (Audubon 2015). Volunteer participants record all species observed within a 24-km diameter circle on a single day between December 14 and January 5 (Sauer et al. 1996). The CBC provides a means of monitoring boreal species, such as the Rusty Blackbird, that nest in inaccessible northern regions, when the majority of the population winters in more southerly accessible regions (Audubon 2015). Hence, the main advantage of this method lies in the fact that it surveys Rusty Blackbird populations across the species’ wintering range (Sauer et al. 1996) and is considered to be the most reliable source of population trend information for this species.
One of the major limitations of the CBC is that Rusty Blackbird abundance may be underestimated because it often mixes with groups of similar species on the wintering grounds. However, this should not necessarily influence the trend estimate, unless other species are on different population trajectories.
Breeding bird survey (bbs)
The Breeding Bird Survey has been conducted in Canada since 1970 (Environment Canada 2014). Data on the abundance of breeding birds are collected by volunteers on 39.2-km routes consisting of 50 stops with a 400-m radius spaced 0.8 km apart. The surveys are generally conducted in June, during the breeding period of most birds. Canadian BBS data are analyzed using hierarchical Bayesian models (Smith et al. 2014).
The BBS has three main disadvantages in terms of monitoring Rusty Blackbird population trends: 1) the species is detected on only about a third of routes across Canada (i.e., 150 routes), most of which have been visited sporadically and have had a number of different observers over time (Greenberg et al. 2011); 2) survey coverage is limited to less than 30% of the species’ range and is concentrated in the southern portion of the boreal forest; and 3) the survey is typically conducted as much as a month after the peak singing period for the species, which means that BBS results may not be representative of the trends observed at more northerly latitudes (Greenberg et al. 2011).
Since COSEWIC produced its original estimate of abundance in 2006, the Partners in Flight (PIF) population estimate, which uses BBS raw data, has been improved with data from the daily bird checklist program of the Northwest Territories, in order to increase the accuracy of the estimate for the boreal forest (Blancher et al. 2013).
Breeding bird atlases
The work carried out in connection with breeding bird atlases conducted in Ontario during 1981-1985 and 2001-2005 provides data that make it possible to compare the distribution and probability of observation of the Rusty Blackbird over a 20-year period (Cadman et al. 2007). Atlas data were gathered by volunteers who surveyed 10 x 10 km squares during the breeding season. For Ontario, the percent change in the distribution of Rusty Blackbirds over a 20-year period was calculated by comparing the percentage of 10-km squares with breeding evidence in the first atlas period to the percentage of squares with breeding evidence in the second period, adjusting for observation effort (Cadman et al. 2007). During the period 2001-2005, relative abundance was also estimated by 5-minute point counts.
Likewise, fieldwork for second breeding bird atlas projects in Quebec and the Maritimes was also recently completed (BBAQ 2015; Stewart et al. 2015). Several other atlases have been completed in the northeast U.S. (e.g., New York, Vermont), while first breeding bird atlas projects have been recently completed in British Columbia and Manitoba.
Atlas projects provide very good information on breeding distributions, and more recently on breeding abundance. The main constraint of this method is that comparisons between time periods are usually made on the basis of occurrence, rather than changes in species abundance. Nonetheless, adjustments to account for the probability of observation between the two survey periods are considered adequate for broadly estimating Rusty Blackbird trends (e.g., Cadman et al. 2007).
étude des populations d’oiseaux du québec (époq)/study of quebec bird populations (sqbp)
This program began in 1969, and manages thousands of checklists produced by volunteers in Quebec (Cyr and Larivée 1995). The database covers all regions south of the 52nd parallel and includes all seasons (Cyr and Larivée 1995). The main disadvantage of this method is that it mainly covers inhabited and easily accessible areas. This program nonetheless makes it possible to estimate population trends using observations of Rusty Blackbirds during migration through southern Quebec (Cyr and Larivée 1995; Scarl et al. 2014).
Canadian migration monitoring network
This program is designed to monitor populations of migratory songbirds at a network of more than 20 monitoring stations across Canada (Crewe et al. 2008). Fall migration records, in particular, can provide information on population trends for birds breeding in the boreal forest and farther north, as they move from their northern breeding grounds to their southern wintering grounds. The primary activities carried out at these stations are bird banding and daily visual counts of birds during migration.
Interpretation of population trends obtained from CMMN stations could be improved by new approaches to approximate the origins (or potential ‘catchment areas’) of migrating birds that are counted at CMMN stations using a combination of stable hydrogen isotope analyses of feathers (δ2Hf) and data on potential bird migration trajectories predicted from band recovery data (Hobson et al. 2015).
One issue with this monitoring program is the assumption that the variation in number of birds counted at a site is representative of the regional trend of the population, and not just a reflection of local conditions. Another limitation is the potential for misidentifying or missing migrating Rusty Blackbirds that often occur in large, mixed flocks with other blackbird species.
Abundance data from historical ornithological accounts
In order to determine whether the decline of the Rusty Blackbird over the last 40 years is part of a longer-term trend, Greenberg and Droege (1999) and Greenberg et al. (2011) analyzed qualitative abundance data from ornithological accounts obtained on wintering sites in the southeastern United States since 1890.
Abundance
According to the Partners in Flight database (Partners in Flight Science Committee 2013), which is based on 1998-2007 BBS data and corrected using data from thousands of other point-count stations established in the boreal forest, Canada’s Rusty Blackbird population is estimated at about 4.4 million breeding individuals (i.e., 2.2 million pairs), which represents about 86% of the global population (Table 1; Partners in Flight Science Committee 2013). This does not reflect a true increase from the previous maximum estimate of 1.4 million birds (COSEWIC 2006), but rather the incorporation of newer information and analysis techniques. The provinces with the largest Rusty Blackbird populations are Quebec, Ontario, Manitoba, and Newfoundland and Labrador (Table 1).
Based on data from 40,500 point count stations in the boreal and hemi-boreal region of North America, Rusty Blackbird distribution and density models for the 1961-1990 period suggest that densities (males/ha) are low in all of Canada, except in west-central Quebec (east of James Bay) and the Hudson Plain in northern Ontario, where densities exceed 0.04 males/ha (Stralberg et al. 2015b; Figures 4 and 5).
| Province / Territory | Population size (individual adults) |
|---|---|
| YK | 200,000 |
| NWT/NU | 180,000 |
| BC | 40,000 |
| AB | 40,000 |
| SK | 10,000 |
| MB | 700,130 |
| ON | 1,000,000 |
| QC | 1,517,000 |
| NB | 4,000 |
| NS | 11,000 |
| PEI | 300 |
| NF-LB | 700,000 |
| Total | 4,402,7301a |
a Partners in Flight (2013) rounded this figure down to 4,000,000 individuals.
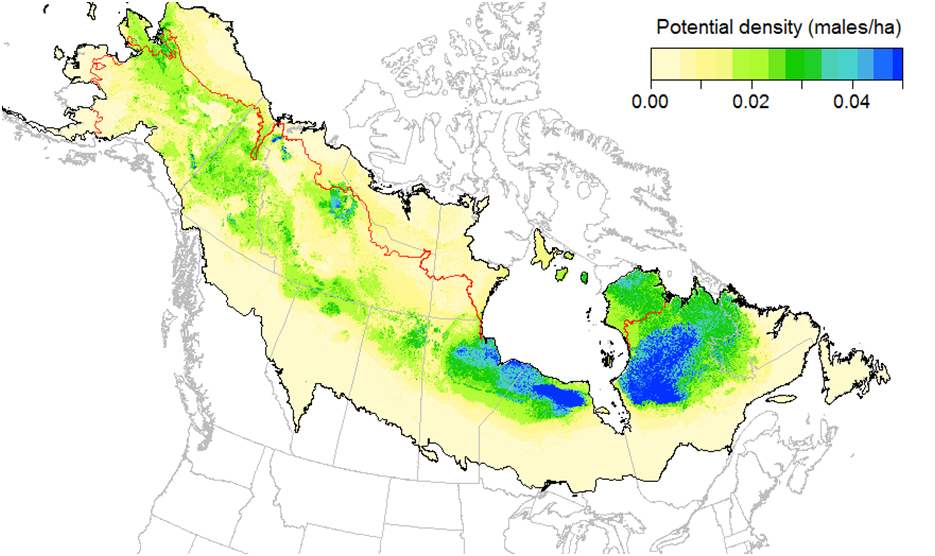
Long description for Figure 5
Map illustrating the relative densities of adult males within the breeding range of the Rusty Blackbird in the boreal forest for the period 1961 to 1990.
Fluctuations and trends
Christmas bird count (cbc)
As noted above, CBC data are judged to be the most reliable for population trend analysis for this species. According to the CBC analysis for North America for the 1970-2012 period, Rusty Blackbird numbers showed a statistically significant annual decline of -2.5 % a year (95% CI: -4.9, -0.5; Figure 6; A. Smith unpubl. data), which is equivalent to a total decline of 65.5% over 42 years. For a slightly longer period from 1966-2013, Soykan et al. (2016) estimated an annual decline of -3.1% (95% CI: -5.7, -1.1). This is equivalent to a continental decline of 77.2% over 48 years.
CBC results indicate a long-term decline in all regions where Rusty Blackbirds were detected, but the rate of decline has diminished since 1990 (Greenberg et al. 2011). For the short-term period 2002-2012, the overall population trend has been uncertain but apparently increasing, though there are very wide confidence intervals (16.9% a year; 95% CI: -29.6, 111; A. Smith unpubl. data).
An analysis of CBC records performed by Hamel et al. (2009) for the period 1953-2001 indicated that Rusty Blackbird numbers detected during the 1950s and early 1960s were high and variable, although they had probably declined since the preceding decades. A marked decline in the overwintering population occurred about 1965 (Hamel et al. 2009).
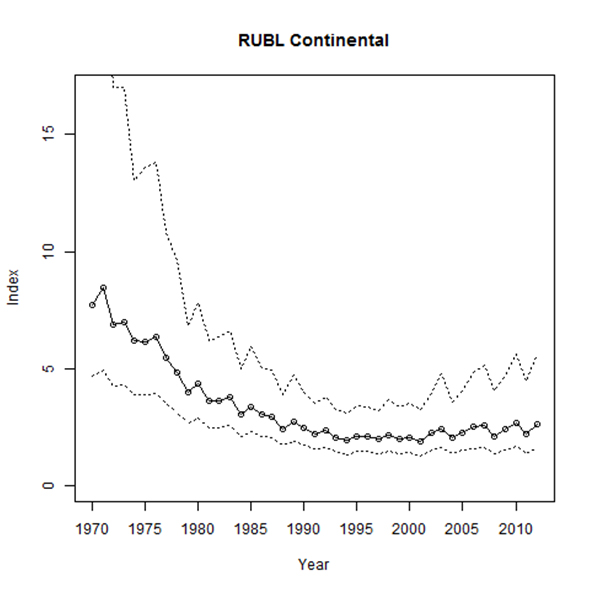
Long description for Figure 6
Chart illustrating median numbers of Rusty Blackbirds detected per party hour on the Christmas Bird Count in North America between 1970 and 2012.
North american breeding bird survey (bbs)
In Canada, there is a statistically significant negative trend of 3.5% per year (n=191 routes, 95% credible interval [CI]: -6.5, -0.6) for the long-term period from 1970-2014 (Figure 7, Table 2; A. Smith unpubl. data), which represents a 79.1% population decline over the last 44 years. For the 10-year (2004-2014) short-term period, a non-significant decline of 2.6% a year was estimated (n=158 routes, 95% CI: -8.4, 4.1; Table 2). At the provincial level, significant long-term declines ranging from -4.6% to -6.7% a year were estimated for the eastern provinces of Ontario, Quebec, Newfoundland and Labrador, New Brunswick and Nova Scotia (Table 2). The species also shows smaller, statistically non-significant declines in all provinces and territories from 2004-2014 (Table 2).
In the United States, statistically non-significant annual declines are apparent for both the long term (1966-2013: -2.1%) and short term (2003-2013: -1.5%), although sample sizes are much smaller (n = 59 routes) than in Canada (Sauer et al. 2014).

Long description for Figure 7
Chard illustrating annual abundance indices of Rusty Blackbirds in Canada between 1970 and 2014, based on a hierarchical Bayesian model of Breeding Bird Survey data.
| Province | Time Period | Annual Trend | Limits Lower |
Limits Upper |
Number of survey routes |
|---|---|---|---|---|---|
| Canada | 1970-2014 | -3.5 | -6.55 | -0.58 | 191 |
| Canada | 2004-2014 | -2.58 | -8.43 | 4.06 | 158 |
| Alberta | 1971-2014 | -4.62 | -11.5 | 2.13 | 19 |
| Alberta | 2004-2014 | -3.46 | -11.8 | 3.85 | 24 |
| British Columbia | 1970-2014 | -3.17 | -8.25 | 3.38 | 21 |
| British Columbia | 2004-2014 | -1.5 | -19.8 | 28.4 | 18 |
| Northwest Territories | 1989-2014 | -2.72 | -6.46 | 1.64 | 15 |
| Northwest Territories | 2004-2014 | -2.62 | -8.78 | 4.68 | 14 |
| Manitoba | 1970-2014 | -3.42 | -12.2 | 6.64 | 6 |
| Manitoba | 2004-2014 | -2.65 | -8.68 | 4.7 | 6 |
| New Brunswick | 1970-2014 | -6.71 | -9.01 | -4.35 | 24 |
| New Brunswick | 2004-2014 | -6.59 | -11.8 | 1.09 | 19 |
| Newfoundland and Labrador | 1976-2014 | -6.33 | -9.35 | -3.58 | 23 |
| Newfoundland and Labrador | 2004-2014 | -2.73 | -8.52 | 4.17 | 21 |
| Ontario | 1970-2014 | -4.61 | -7.68 | -1.4 | 37 |
| Ontario | 2004-2014 | -2.68 | -8.61 | 4.23 | 31 |
| Quebec | 1970-2014 | -5.32 | -7.88 | -2.71 | 30 |
| Quebec | 2004-2014 | -2.61 | -8.62 | 4.47 | 21 |
| Nova Scotia/ Prince Edward Island | 1970-2014 | -5.89 | -7.99 | -3.87 | 29 |
| Nova Scotia/ Prince Edward Island | 2004-2014 | -6.17 | -13.8 | 0.84 | 26 |
| Yukon | 1973-2014 | -2.65 | -5.87 | 0.96 | 25 |
| Yukon | 2004-2014 | -2.59 | -7.67 | 3.75 | 23 |
Breeding bird atlases
In Ontario, a comparison of Rusty Blackbird distribution between the first (1981-1985) and second (2001-2005) survey periods shows a non-significant decline of 5% in the probability of observation for the province as a whole, for the entire 20-year period (Figure 8; Cadman et al. 2007). The decline was statistically significant in the Southern Shield region and in the eastern part of the Northern Shield region, at -30% and -32% respectively. The species also declined non-significantly in the Lake Simcoe region (-49%; Cadman et al. 2007). However, significant increases were calculated for the Hudson’s Bay Lowlands (+37%; Cadman et al. 2007).
In Quebec, a visual comparison of the number of occupied atlas squares between the 1980s and the 2010s suggests a reduction in the number of squares occupied by the species in some southern parts of the province, including the Outaouais, the Laurentians and the Appalachians (Figure 9; BBAQ 2015). For Figure 9, note that areas north of 50 degrees latitude were not covered in the first atlas. Corrections for differences in effort have not yet been completed.
In the Maritimes, a comparison of the number of occupied atlas squares between the 1980s and the 2010s suggests a reduction of 55% (442 squares vs 275 squares; Figure 10; Stewart et al. 2015). Again, corrections for differences in effort have not been carried out.
Atlas work done in Vermont and New York in the 1980-1985 and 2000-2005 periods indicates declines of 23% and 42%, respectively, in the number of squares occupied by breeding Rusty Blackbirds (McCormack 2012).
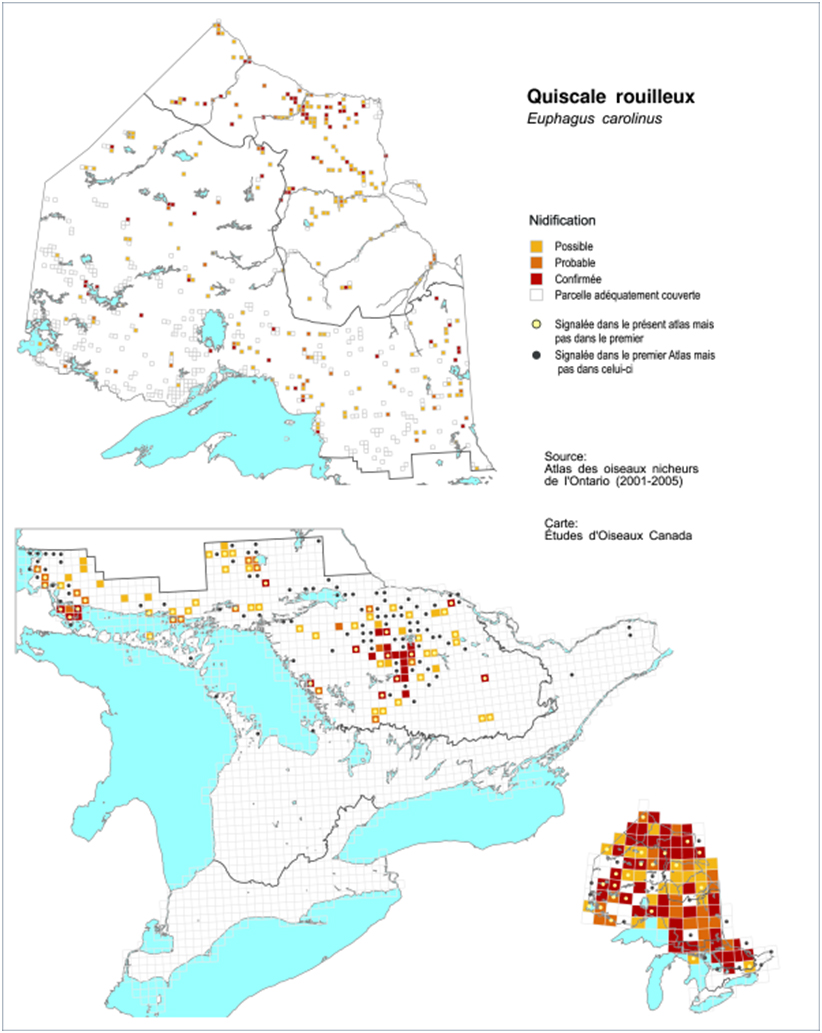
Long description for Figure 8
Maps illustrating the range of the Rusty Blackbird in Ontario in the first (1980 to 1985) and second (2001 to 2005) breeding bird atlas periods.
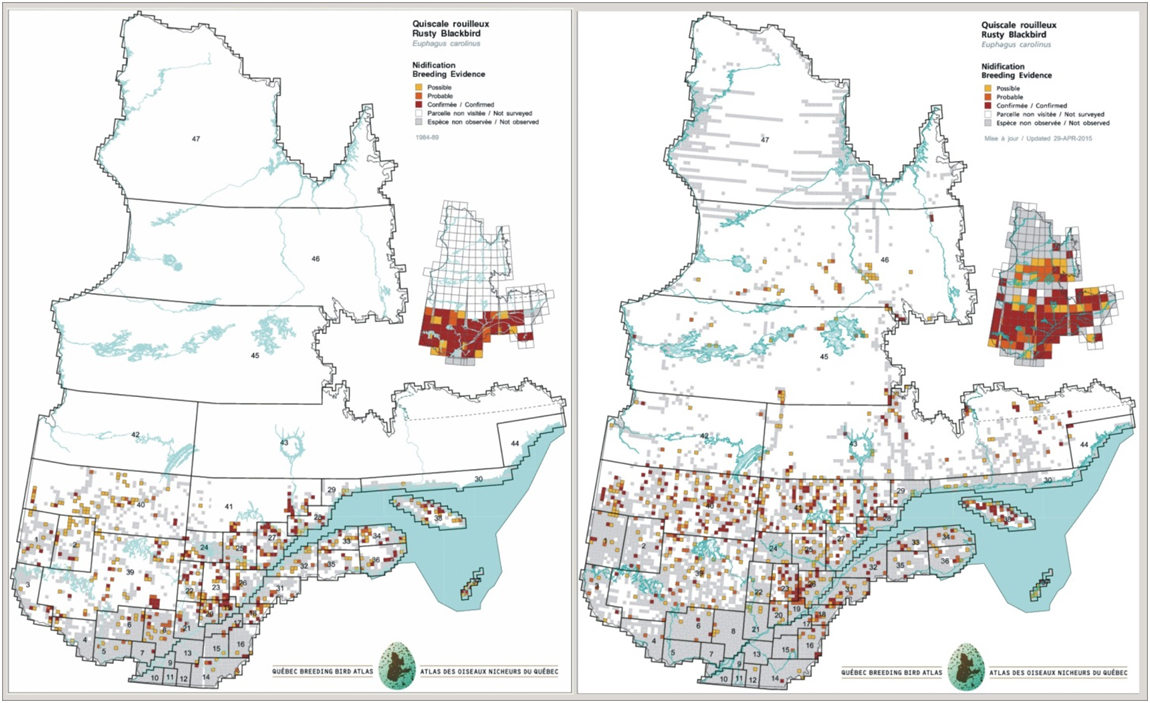
Long description for Figure 9
Maps illustrating the range of the Rusty Blackbird in Quebec in the first (1985 to 1990) and second (2010 to 2015) breeding bird atlas periods.
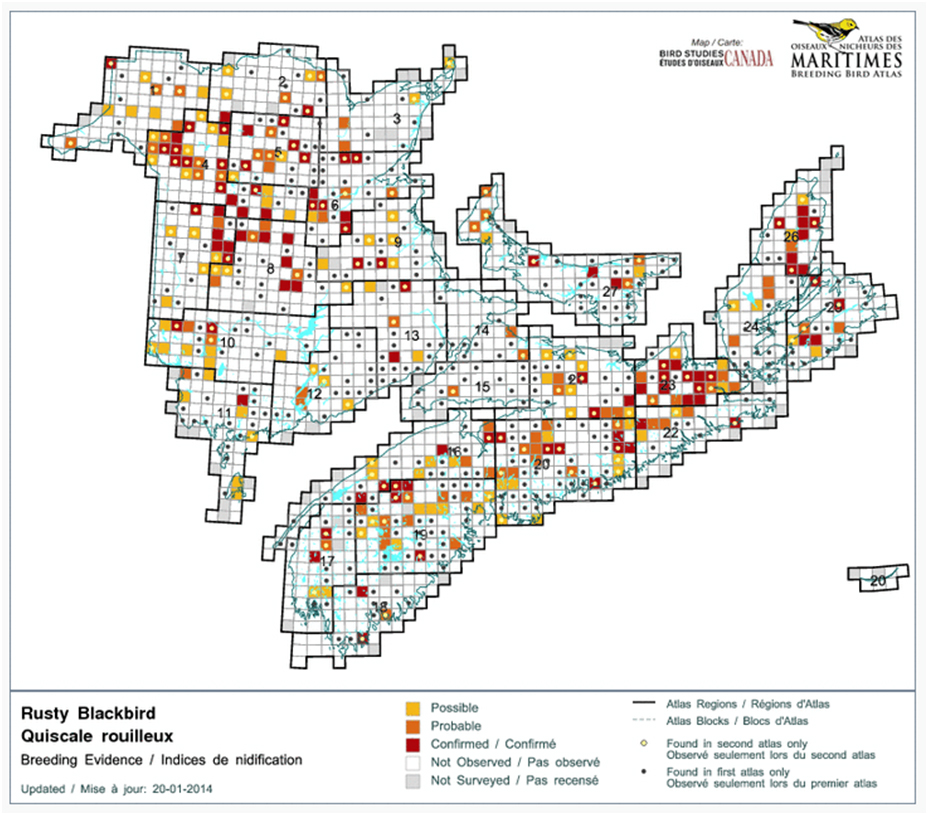
Long description for Figure 10
Map illustrating the range of the Rusty Blackbird in the Maritimes in the first (1985 to 1990) and second (2010 to 2015) breeding bird atlas periods.
étude des populations d’oiseaux du québec (époq)/ study of quebec bird populations (sqbp)
The ÉPOQ spring abundance index of the Rusty Blackbird in Quebec based on data for the period 1975-2013 period suggests a statistically non-significant annual decline of 0.9% (Figure 11; Larivée 2015). The abundance index for the short term estimate for 2003-2013 shows a statistically significant decline (R2=0.57, p=0.01).
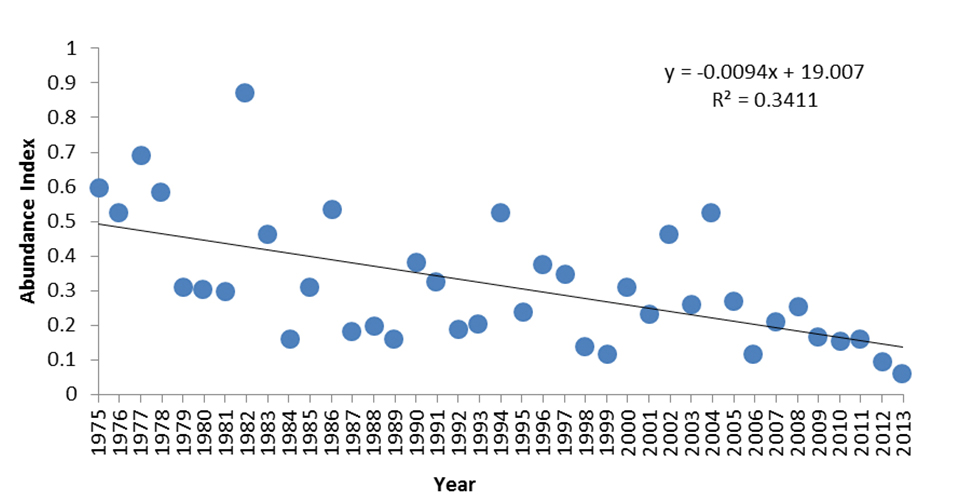
Long description for Figure 11
Chart illustrating annual abundance indices of Rusty Blackbirds in Quebec between 1975 and 2013, based on the Étude des populations d’oiseaux du Québec (ÉPOQ) database.
Canadian migration monitoring network (cmmn)
Population trends are available from five migration monitoring stations in Ontario and Quebec, where Rusty Blackbird is considered to be well-sampled (data for at least 10 years and covering at least 75% of their local migration; Dunn et al. 2016). All stations show high annual variability in counts of Rusty Blackbirds. Based on counts from Observatoire d’oiseaux de Tadoussac, Savard et al. (2011) suggested that the Rusty Blackbird population breeding in northern Quebec fluctuates at 4-5 year intervals.
Migration monitoring stations show mixed results, with both positive and negative short-term trends from 2004-2014 (Table 3), including a statistically significant annual increase of 27% from Tadoussac in eastern Quebec and a significant annual decrease of 17% from Pelee Island in southwestern Ontario (CMMN 2016). However, short-term trends may not be representative of current status for a species with cyclical fluctuations in abundance (Savard et al. 2011). Long-term trends from daily counts are generally positive, but in all cases statistically non-significant (CMMN 2016; Table 3).
| Station | Area | Season | Short-term | Trend (%) | CI-Low (%) | CI-High (%) | Long-term | Trend (%) | CI-Low (%) | CI-High (%) | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| PEPBO | EOnt | Fall | 2004-2014 | 11.40 | -14.71 | 38.79 | blank cell | 2001-2014 | 14.68 | -3.03 | 24.02 |
| IPBO | EOnt | Spring | 2004-2014 | 6.48 | -27.2 | 47.3 | blank cell | 1997-2014 | 4.07 | -13.76 | 23.1 |
| OOT | WQc | Fall | 2004-2014 | 27.05 | 3.69 | 60.51 | blank cell | 1996-2014 | 4.49 | -6.17 | 18.54 |
| PIBO | SWOnt | Fall | 2004-2014 | -17.49 | -28.64 | -3.49 | blank cell | blank cell | blank cell | blank cell | blank cell |
| TCBO | NWOnt | Fall | 2004-2014 | -7.66 | -24.44 | 8.06 | blank cell | 1995-2014 | -0.86 | -9.95 | 6.44 |
PEPBO: Prince Edward Point Bird Observatory; IPBO: Innis Point Bird Observatory, OOT: Observatoire d’oiseaux de Tadoussac, PIBO: Pelee Island Bird Observatory, TCBO: Thunder Cape Bird Observatory
Qualitative analyses of historical accounts in the usa
Two qualitative analyses of ornithological accounts published in the U.S.A. before modern monitoring programs (i.e., prior to 1950) indicate a long-term change in the description of the species’ abundance, changing the descriptive terminology from ‘common - abundant’ to ‘rare - uncommon’ (Greenberg et al. 2011). According to this analysis, the decline of the species began in the early 20th century, which is well before the decline illustrated by the BBS and the CBC. A second study using a larger sample of bird checklists arrived at the same conclusion, namely that the species was recognized as very common or abundant in 56% of the published accounts prior to 1920, but this declined to 19% between 1921 and 1950, and then to 7% after 1950 (Greenberg and Droege 1999). Greenberg et al. (2011) argue that results of these two studies suggest that this long-term decline is more consistent with the known habitat loss on the wintering grounds than environmental changes in the boreal forest, which are more recent phenomena. However, there were indeed wide-scale changes in habitats across the Rusty Blackbird breeding range from 1700-1900 due to large-scale commercial beaver trapping activities by Europeans that hunted beavers nearly to extinction across much of their range (Naiman et al. 1988; Humphries and Winemiller 2009). This might have directly reduced availability of forested wetlands for Rusty Blackbirds prior to 1950.
Population trend summary
The two largest data sources tracking Rusty Blackbird population trends (CBC and BBS) suggest that the Rusty Blackbird population has undergone a long-term decline of 66-80% over the last 44 years. Over the most recent 10-year period, however, the BBS and CBC both indicate that the population has been more or less stable, although there are statistical uncertainties. Various breeding bird atlas projects also show a long-term decline of the species between the 1990s and the 2010s, and in some cases an apparent contraction of the species’ range in the south over the last two decades. Trend signals are mixed from CMMN stations, with increases being noted in some places and declines at others. Analyses of historical records in the United States suggest that the long-term decline in Rusty Blackbird populations probably originated in the early 1900s. On balance, it appears that the pattern of long-term decline may have moved more towards stability over the last decade.
Rescue effect
In the event of extirpation of the Canadian Rusty Blackbird population, immigration of individuals from the New England States appears unlikely to be sufficient to rescue the Canadian population, owing to the small portion of the overall population in these U.S. states, and the significant decline of the species there between 1966 and 2013, particularly in Vermont (-11.5 % yr-1; 95% CI: -20.7, -2.6 % yr-1) and New Hampshire (-9.5 % yr-1; 95% CI: -15.4, -3.9% yr-1; Sauer et al. 2014). Immigration of individuals from Alaska also appears to be unlikely owing to current habitat loss there due to climate change (Riordan et al. 2006). The U.S. population of Rusty Blackbirds is only about 16% of the size of the Canadian population (Partners in Flight Science Committee 2013), further reducing the potential for a rescue effect, particularly if populations in both countries decline.
Threats and limiting factors
Threats
The threat assessment indicated a Medium level impact overall (see Appendix A for details). Threats are ordered below from highest to lowest impact. Greenberg and Matsuoka (2010) state that there are still many unknowns about the reasons for the decline of the Rusty Blackbird. Some of the threats that scored as unknown are considered below, as they may actually have significant impacts on Rusty Blackbird populations, but require further study to assess their importance.
Habitat loss on the wintering grounds of the Rusty Blackbird, through the conversion of 75-80% of forested wetlands in the southern United States, is thought to be one of the most significant factors in the long-term decline of this species (Greenberg and Droege 1999; Greenberg et al. 2011). However, according to Hamel et al. (2009), recent loss of Rusty Blackbird wintering habitat is not as pronounced as the population decline of the species estimated using CBC data. Hence, they felt that it was premature to conclude that this loss is the primary cause of recent population declines.
Residential and commercial development (iucn threat #1); agriculture and aquaculture (iucn threat #2); energy production and mining (iucn threat #3)
All three of these threats contribute directly to the loss of Rusty Blackbird habitat at some point in the species’ life cycle, and are lumped together here (see Appendix A for details). The impact calculated for each of these threats was Low.
On the wintering grounds, wetland forests have been converted to agriculture, pine plantations and residential neighbourhoods (Hamel et al. 2009; Greenberg and Matsuoka 2010). It is estimated that between 1780 and 1980, the area of wetlands on the wintering grounds decreased by 57% (Mississippi Lowlands) and 36% (southern Atlantic Coastal Plain; Hamel et al. 2009). More recently, habitat loss of the remaining wetland forests has also been significant, particularly between 1970 and 1990, owing to the conversion to agricultural land for the production of soybeans, when that crop became highly profitable (Greenberg et al. 2011). However, the area of blackbird habitat subsequently increased following the drop in soybean prices in the late 1980s, when large-scale reforestation programs occurred (Greenberg et al. 2011).
On the breeding range, habitat loss of the Rusty Blackbird is occurring at a slow but increasing rate in parts of Canada. For example, in the southern portion of the species’ Canadian breeding range, large areas of wetlands have been converted for agricultural and industrial operations (Greenberg and Droege 1999; Hobson et al. 2002; Young et al. 2006; Greenberg et al. 2011). In total, breeding habitat in Canada is believed to have decreased by 5% since European settlement, and a further reduction of 4% by 2050 is projected (Global Forest Watch Canada 2000; Griffiths and Woynillowicz 2003; Kling et al. 2003; Rooney et al. 2012). It is estimated that, as of 2003, 8% of the boreal forest in Canada has been affected by oil and gas exploration and extraction activities (Greenberg et al. 2011).
Biological resource use (iucn threat #5)
The overall impact calculated for this threat is Low, as the scope is considered small, but severity is considered to be extreme.
Hunting and collecting terrestrial animals
Bird control programs designed to reduce populations of “nuisance” blackbirds that damage crops are a cause of mortality for overwintering populations of Rusty Blackbirds in the eastern United States (Avery 2013). Between 1974 and 1992, Red-winged Blackbird and European Starling control programs (Heisterberg et al. 1987) using PA-14 Avian Stressing Agent at 83 roosts located in Kentucky, Tennessee and Alabama exterminated 38 million birds, including an estimated 120,000 Rusty Blackbirds (Meanley 1976; Dolbeer et al. 1997). The period of use of this product, which has since been taken off the market, generally coincides with the period of greatest decline of Rusty Blackbird populations (Greenberg et al. 2011).
Migrating and wintering populations of Rusty Blackbirds could also be affected by bird control programs using DCR-1339 (Starlicide®), currently in use in the central and southeastern U.S. states, particularly in sunflower and rice crops (Linz et al. 2002; Blackwell et al. 2003; Custer et al. 2003). In addition, the use of poison bait to control the presence of blackbirds in cattle feedlots in the U.S. could adversely affect migrating and wintering Rusty Blackbirds (Greenberg and Matsuoka 2010).
Logging and wood harvesting
In New England, logging was identified as a threat to the Rusty Blackbird by Powell et al. (2010b). Their study of 43 nests demonstrated that blackbirds that nested in wetlands with recent logging in the vicinity had 2.3 times greater probability of being preyed upon than those nesting in wetlands where there had been no logging in the past 20 years. In this area, the potential predators are the Gray Jay, Blue Jay and Red Squirrel (Powell et al. 2010b), which, at least for Blue Jays, are generally more abundant in fragmented forests (Robinson et al. 1995). However, another New England study, which was based on a larger sample of nests, demonstrated that Rusty Blackbird nest success was high in a managed forest landscape (Buckley 2013). It is therefore premature to conclude that logging has an overall negative impact on Rusty Blackbird breeding populations. Moreover, these two studies are not directly applicable to the different ecological context experienced by the Rusty Blackbird in the boreal forest of Canada. Overall impact calculated for this threat is considered Low, as the scope is small, and severity is moderate.
Natural system modifications (iucn threat #7)
The overall impact calculated for this threat is Low, as the scope is considered large, and severity is considered to be moderate-slight.
The primary threat in this category is related to dams and water management and uses. On its wintering sites, the Rusty Blackbird appears to be sensitive to changes in surface hydrology caused by activities that result in wetland drainage, stream diversion and water control, and displacement of groundwater (Greenberg et al. 2011). Rusty Blackbirds prefer wetlands that have water levels that fluctuate regularly (Mettke-Hofmann et al. 2015); they are less commonly found in reservoirs that are flooded for long periods and have deep water (Batema et al. 1985). Industrial activities on the breeding and migratory grounds (e.g., oil sands development and hydroelectric projects) could also alter the hydrology in Rusty Blackbird habitat (Greenberg et al. 2011).
Pollution (iucn threat #9)
The impact calculated for this threat is ranked Low, as the scope is considered to be large to restricted, and severity is slight.
Agricultural and forestry effluents
Several recent studies have suggested that pesticides are a significant cause of direct mortality for a number of bird species that occur in agricultural areas (Boatman et al. 2004; Mineau 2005; Mineau and Whiteside 2013). The Rusty Blackbird migrates through and winters in heavily farmed areas, where it feeds on grains such as corn (Dias 2008).
A national analysis of the risk of bird mortality due to exposure to pesticides currently used in the U.S. indicated that the greatest number of bird kills likely occurs in the southeastern states, owing to the high proportion of farmlands there using pesticides (Mineau and Whiteside 2013). This region is in the centre of the Rusty Blackbird wintering grounds (Hamel and Ozdenerol 2008).
Between 1970 and 1980, the widespread use of granular carbofuran pesticides on row crops in the North American prairies resulted in an estimated annual loss of 17-91 million birds of various species in corn fields alone (Mineau et al. 2005). Granular carbofuran formulations are no longer in use in Canada and the U.S., but probably contributed to declines in Rusty Blackbird populations prior to their being taken off the market.
DDT (dichloro-diphenyl-trichloroethane) was used on a large scale, including in Spruce Budworm control programs in eastern Canada between 1952 and 1967 (Environment Canada 1991; Walker et al. 2012). In Canada, an estimated 67 million ha of forest were treated with pesticides (mainly DDT) to control budworm outbreaks between 1952 and 1990, with the peak occurring in the mid-1970s (Walker et al. 2012). Even at low concentrations, DDT is very toxic to aquatic invertebrates, including larval odonates (Ide 1967; Wilkes and Weiss 1971; World Health Organization 1989), on which Rusty Blackbirds mainly feed during the breeding period (Loomis 2013). Rusty Blackbirds breeding throughout the Canadian boreal forest could have been also affected by DDT, even in the northern part of the boreal forest where DDT was not historically used, due to DDT’s tendency to enter the atmosphere through volatilization and contaminate food chains in areas far from the original source of emission (Braune 1993; Muir et al.1995). Studies have shown that forest soil that received DDT treatments may remain contaminated decades after treatment (Lichenstein and Shulz 1959; Stewart and Chisholm 1971). DDT was also used intensively in the species’ wintering range (especially in the Mississippi Valley) between 1947 and 1951 in order to eradicate malaria (Centers for Disease Control and Prevention 2012). Although DDT was banned in North America in the 1970s, it could have contributed to the species’ decline.
More recently, neonicotinoid insecticides were introduced in the 1990s. More than 11 million ha of agricultural lands have been treated with these insecticides in the Canadian prairies (Main et al. 2014). These pesticides have been associated with the decline of several farmland bird species in Europe, owing to declines in the insect prey base that they cause (Hallmann et al. 2014). The effect of neonicotinoids on the Rusty Blackbird is unknown, but any effects would be expected to occur outside the breeding season.
Airborne pollutants
The deposition of mercury by industrial activities (Desgranges et al. 1998; Fitzgerald et al. 1998; Porvari et al. 2003; Wiener et al. 2003) and the release of methylmercury by permafrost melting in the boreal forest (Klaminder et al. 2008; Edmonds et al. 2010) are a major source of contamination of aquatic environments in Canada. Exposure to mercury can reduce the reproductive success of birds by altering their immune response and can also cause behavioural and physiological impairments (Scheuhammer et al. 2007). Studies conducted in several parts of the Rusty Blackbird’s range have found high mercury concentrations in the diet, due to the species’ habit of feeding in acidic wetlands where mercury is readily converted to methylmercury, which is the more toxic form (Evers et al. 2005; Edmonds et al. 2010; Greenberg and Matsuoka 2010). Rusty Blackbirds breeding in the boreal forest of Canada, Alaska and New England also have significantly higher blood levels of mercury than birds wintering in the southern United States (Matsuoka et al. 2008; Edmonds et al. 2010), suggesting that birds on the breeding grounds may be more subject to mercury exposure than wintering birds, owing to their stricter diet of insects during the breeding season (Evers et al. 2005).
It is also believed that the Rusty Blackbird is negatively affected by acidification due to acid rain (Schindler 1988), particularly in the eastern part of its breeding range (DesGranges and Houde 1989; Greenberg and Droege 1999). Greenberg and Droege (1999) hypothesized that wetland acidification may affect Rusty Blackbirds through loss of calcium and other minerals essential for eggshell and bone formation. Rusty Blackbirds feed on snails and molluscs, which could make them sensitive to a lack of calcium if those food sources decline owing to acidification (see Graveland et al. 1994). Acidification also results in higher releases of mercury into wetland systems (see above).
Invasive and other problematic species and genes (iucn threat #8)
The overall impact of this threat is unknown. Recent analyses of blood samples of breeding adults from Alaska, Maine and on the wintering sites (Mississippi and Arkansas) reveal that prevalence of hematozoan infection (mainly Leucocytozoon) was 44% (43 birds) in Alaska and 67% (12 birds) in Maine (Barnard et al. 2010). In winter, the overall prevalence was estimated at 49% (also mainly Leucocytozoon), which is considered higher than the anticipated values for this period of the year, when parasite transmission is generally low (Barnard et al. 2010). Although the parasites are not considered to be directly hazardous to the blackbirds’ health, a high prevalence during winter suggests that Rusty Blackbirds may be stressed or have a compromised immune system (Barnard et al. 2010). This could cause an increase in the impact of diseases and other, more harmful, parasitic infections (Barnard et al. 2010). However, the extent to which this poses a bona fide threat versus a limiting factor is currently unknown.
Climate change and severe weather (iucn threat #11)
The overall impact calculated for this threat is unknown, as the scope is considered pervasive to large, but severity is unknown.
Habitat shifting and alteration
The effects of climate change in boreal wetlands could contribute to the species’ decline (Matsuoka et al. 2010a), both historically and in the future. Stralberg et al. (2015b) incorporated climatic data and point count data for 80 Canadian boreal-breeding bird species across Canada, to model potential population impacts of climate change. According to their models, the projected future decline in the abundance of the Rusty Blackbird could be 55% by the year 2100 (LCI, 5th percentile: -150%, UCI, 95th percentile: -18%; Stralberg et al. 2015b).
It is predicted that climate change in the boreal region will result in a substantial increase in mean annual temperatures by 2100 (Price et al. 2013), which will have negative impacts on the Rusty Blackbird, including a contraction of its breeding range. Indeed, climate change might be an underlying cause of an apparent northward retraction of the species’ breeding range reflecting a loss of Rusty Blackbird habitat (McClure et al. 2012; Stralberg et al. 2015a,b). These authors concluded that the probability of local extirpation was greatest in the southern portion of the species’ breeding range and that the southern boundary of the Rusty Blackbird’s breeding range has contracted northward by about 143 km since 1966.
Drying and degradation of wetlands across much of the boreal forest, owing to lowering of the water table associated with thawing permafrost and increased evapotranspiration, have already been documented (Yoshikawa and Hinzman 2003; Klein et al. 2005; Riordan et al. 2006; Matsuoka et al. 2010a; Carroll et al. 2011). In Canada, a study concluded that the area of shallow ponds and lakes in the boreal, subarctic and arctic zones decreased by 6700 km2 between 2000 and 2009 (Carroll et al. 2011). This drying of boreal wetlands is causing changes in communities of aquatic invertebrates, which could include a reduction in the biomass of food resources important to Rusty Blackbirds, such as snails, amphipods and chironomid larvae (Corcoran et al. 2009). McClure et al. (2012) also hypothesized that climate change and the drying of wetlands in the boreal forest could shift the timing of emergence of insect prey, particularly odonates, out of phase with the phenology of breeding Rusty Blackbirds.
Limiting factors
Predation
While not considered a threat, predation is a relatively important limiting factor for the Rusty Blackbird during the nesting period (Powell et al. 2010b; Savard et al. 2011). The monitoring of blackbird migrations at Tadoussac, Quebec, suggests that the peaks of abundance of the Rusty Blackbird in the fall are closely associated with fluctuations in the pressure exerted by predators in the boreal forest (i.e., small mustelids and corvids), which in turn fluctuates according to the 4-5 year cycle of abundance of the Red-backed Vole (Clethrionomys gapperi; Savard et al. 2011). These authors hypothesize that when small rodents are abundant in the boreal forest, predation pressure on Rusty Blackbird nests is greatly reduced, which would result in a greater abundance of blackbirds the following fall.
In Alaska, nesting success of the Rusty Blackbird fluctuated markedly, from 21% in 2006 to 64% in 2007 and 2008 (Matsuoka et al. 2010a). These authors attributed the low success rate in 2006 to heavy predation pressure (89% of nests lost). In New England, the Red Squirrel is the main predator of Rusty Blackbird nests, and nest predation is believed to increase with the rise in the squirrel population during years following high cone production (Buckley 2013).
On the wintering grounds, predation of Rusty Blackbirds being tracked by telemetry was mainly associated with hawks (Newell 2013), whose populations are increasing due to the banning of DDT (Greenberg and Matsuoka 2010).
Number of locations
The number of locations for the Rusty Blackbird is currently unknown but, considering its wide distribution in Canada and the large number of breeding pairs, there are far more than 10 locations at which a single most likely or imminent threatening event could rapidly affect all individual Rusty Blackbirds present. This figure is a threshold for a potential Threatened status under COSEWIC’s quantitative guidelines.
Protection, status and ranks
Legal protection and status
Although the Rusty Blackbird is a migratory species, it is not formally protected in Canada under the Migratory Birds Convention Act, 1994 (Environment Canada 2015). Most blackbirds were excluded from the Act in 1916, along with other birds considered at the time to be pests. As a consequence, it is normally protected in all Canadian jurisdictions under provincial and territorial wildlife legislation.
The Rusty Blackbird is currently listed on Schedule 1 of the Species at Risk Act as Special Concern, and a management plan has been developed for this species (Environment Canada 2015). It is listed as Vulnerable under the Newfoundland and Labrador Endangered Species Act, a Species of Special Concern under New Brunswick’s Species at Risk Act, and Endangered under Nova Scotia’s Endangered Species Act. In Quebec, the Rusty Blackbird is on the list of wildlife species likely to be designated threatened or vulnerable, under the Loi sur les espèces menacées ou vulnérables (RLRQ, c E-12.01) (LEMV) (Act respecting threatened or vulnerable species) (CQLR, c E-12.0).
Non-legal status and ranks
NatureServe (2015) assigns the Rusty Blackbird a global rank of G4 (Apparently Secure; Table 4). According to the IUCN Red List, the species is considered Apparently Secure (NatureServe 2015). In the U.S., it has a national rank of N4 and is considered Apparently Secure (NatureServe 2015), even though it is considered Critically Imperilled (S1) in one state, Imperilled (S2) in five states and Vulnerable (S3) in 15 states (NatureServe 2015). Partners in Flight’s North American Landbird Conservation Plan (Rich et al. 2004) lists it as a species of “Continental Concern”.
In Canada, the Rusty Blackbird is considered Apparently Secure (N4) by NatureServe (NatureServe 2015; Table 4). It is considered Imperilled (S2) in Prince Edward Island and Nova Scotia, and Vulnerable (S3) in the Yukon, Northwest Territories, British Columbia, Manitoba, Quebec, New Brunswick, Newfoundland and Labrador. In Saskatchewan and Ontario, the species is considered uncommon (S4; Table 4).
Habitat protection and ownership
In Canada, suitable breeding habitat for Rusty Blackbird is found primarily on public land in the boreal region (Boreal Avian Modeling Project 2015). It is estimated that 9% of the Rusty Blackbird’s range is currently protected by national and provincial parks, migratory bird sanctuaries and national wildlife areas (Wells et al. 2011), but even larger areas are expected to be protected in Ontario and Quebec within the next decade (D. Kraus pers. comm. 2016). The Rusty Blackbird occurs regularly in at least 23 protected areas managed by the Parks Canada Agency (Parks Canada 2015), including 12 national parks where the species is considered a breeding bird. The species is also present in many other federal lands administered by other departments and Aboriginal governments, as well as in numerous provincial parks and ecological reserves, and other kinds of nature reserves and conservation areas.
| Region | Rankb |
|---|---|
| Global | G4 |
| United States | N4 |
| Canada | N4 |
| Yukon | S3B |
| Nunavut | SNRB |
| Northwest Territories | S3B |
| British Columbia | S3S4B |
| Alberta | S4 |
| Saskatchewan | S4B |
| Manitoba | S3S4B |
| Ontario | S4B |
| Quebec | S3S4 |
| New Brunswick | S3B |
| Nova Scotia | S2S3B |
| Prince Edward Island | S2B |
| Newfoundland | S2S3B |
| Labrador | S3B |
b G = is a global status rank; S = rank assigned to a province or state; N= is a national status rank. S1 indicates that a species is critically imperilled because of extreme rarity (often 5 or fewer occurrences) or because of other factor(s) such as a steep decline, making it especially vulnerable to extirpation; S2 indicates that a species is imperilled because of rarity or other factors making it very vulnerable to extirpation, usually with 6 to 20 occurrences or few individuals remaining (i.e., 1,000 to 3000); S3 indicates that a species is vulnerable at the subnational level because it is rare or uncommon, or found only in a restricted range, or because of other factors making it vulnerable to extirpation; S4 indicates that the species is uncommon but not rare, and that there is some cause for long-term concern due to declines or other factors; S5 indicates that a species is secure, because it is common, widespread and abundant globally.
Acknowledgements
The report writer wishes to extend thanks to Christian Artuso, Ruben Boles, Mike Burrell, Suzanne Carrière, Kaytlin Cooper, Todd Copeland, Dean Demarest, Bruno Drolet, Richard Elliot, Dave Fraser, Marcel Gahbauer, Eric Gross, Samuel Hache, Jessica Humber, Colin Jones, Lisa Keable, Jeff Keith, Dan Kraus, Steven Matsuoka, Jon McCracken, Claudia Mettke-Hofmann, Greg Mitchell, Brian Naylor, Luke Powell, Chris Risley, Jean-Pierre Savard, Ken De Smet, Pam Sinclair, Adam Smith, Diana Stralberg, Don Sutherland, Marc-André Villard, Alana Westwood, and Steven Van Wilgenburg for their comments on earlier drafts of this report and/or for their input on the threats assessment. Thanks are offered by the report writer to the Regroupement QuébecOiseaux, Environment and Climate Change Canada’s Canadian Wildlife Service and Bird Studies Canada for supplying data from the atlas of the breeding birds of Quebec, and to the thousands of participants who helped gather Atlas data. Funding for this report was provided by Environment and Climate Change Canada.
Authorities contacted
Canadian wildlife service – environment and climate change Canada
- Yves Aubry – Wildlife biologist - Migratory Birds, Québec, QC
- Luc Belanger – Manager, Conservation Planning and Stewardship, Québec, QC
- Ruben Boles – Species at Risk Scientific Project Officer, Gatineau, QC
- Andrew Boyne – Head, Conservation Planning and Engagement, Dartmouth, NS
- Shelagh Bucknell – Administrative Services Assistant, Delta, BC
- Syd Cannings – Species at Risk Biologist, Whitehorse, YK
- Vanessa Charlwood – Northwest Territories Section Head, Yellowknife, NWT
- Bruno Drolet – Senior Forest Bird Biologist, Québec, QC
- David Duncan – Manager, Population Status and Assessment, Edmonton, AB
- Samara Eaton – Species at Risk Biologist, Sackville, NB
- Jean Gauthier – Avian biologist, Québec, QC
- Siu-Ling Han – Nunavut Section Head, Iqaluit, NU
- Bruce Macdonald – Northern Manager, Yellowknife, NWT
- Rhonda Millikin – Section Head, Delta, BC
- Michel Robert – Wildlife biologist - Migratory Bird Surveys, Québec, QC
- Rich Russell – Wildlife biologist, Ottawa, ON
Parks Canada agency
- Patrick Nantel – Office of the Chief Ecosystem Scientist, Parks Canada Agency, Gatineau, QC
Provincial and territorial species at risk representatives
- David Fraser – Scientific Authority Assessment, Ecosystems Protection and Sustainability Branch, BC Ministry of Environment, Victoria, BC
- Gordon Court – Provincial Wildlife Status Biologist, Fish and Wildlife Division, Dept. of Sustainable Resource Development, Edmonton, AB
- Richard Quinlan – Biologist, Fish and Wildlife Division, Dept. of Sustainable Resource Development, Edmonton, AB
- Steven Blaney, Biologist, Wildlife and Ecosystem Protection Branch, Manitoba Department of Conservation, Winnipeg, MB
- William Watkins – Wildlife and Ecosystem Protection Branch, Manitoba Department of Conservation, Winnipeg, MB
- Vivian Brownell – Biologist, Ontario Ministry of Natural Resources and Forestry, Peterborough, ON
- Michael Oldham, Biologist, Ontario Ministry of Natural Resources and Forestry, Peterborough, ON
- Isabelle Gauthier, Coordonnatrice provinciale des espèces fauniques menacées ou vulnérables, Direction générale de la gestion de la faune et des habitats, Ministère des Forêts, de la Faune et des Parcs de Québec, Québec, QC
- Jérôme Lemaître, Avian Biologist, Direction générale de la gestion de la faune et des habitats, Ministère des Forêts, de la Faune et des Parcs de Québec, Québec, QC
- Annie Lévesque – Biologist, Direction générale de la gestion de la faune et des habitats Ministère des Forêts, de la Faune et des Parcs de Québec, Québec, QC
- Pascal Giasson – Manager, Species at Risk Program, Fish and Wildlife Branch Department of Natural Resources, Fredericton, NB
- Mary Sabine – Biologist, Species at Risk Program, Fish and Wildlife Branch Department of Natural Resources, Fredericton, NB
- Sherman Boates, Manager – Biodiversity, Wildlife Division, Nova Scotia Department of Natural Resources, Kentville, NS
- Rosemary Curley – Biologist, Forests, Fish and Wildlife Division, Prince Edward Department of Communities, Lands and Environment, Charlottetown, PEI
- Shelley Pardy – Ecosystem Management Ecologist, Endangered Species and Biodiversity Section, Newfoundland and Labrador Department of Environment and Conservation, Corner Brook, NL
- Suzanne Carrière – Biologist (Biodiversity), Wildlife Division, Northwest Territories Department of Environment and Natural Resources, Yellowknife, NWT
- Thomas Jung, Biologist - Senior Wildlife Biologist, Fish and Wildlife Branch, Environment Yukon, Whitehorse, YK
Cosewic secretariat, environment and climate change Canada
- Alain Filion – Scientific and GIS Project Officer, Gatineau, QC
- Jenny Wu – Scientific Project Officer, Gatineau, QC
Other authorities contacted
- R. Anderson – Wildlife Biologist, Canada Museum of Nature, Ottawa, ON
- Robert Bellizi – Wildlife Biologist, Department of National Defence, Ottawa, ON
- Rick Page – Wildlife Biologist, Victoria, BC
- Tim Birt – Wildlife Biologist, Queen’s University, Kingston, ON
- Jean-Pierre Savard - Scientist Emeritus, Wildlife and Landscape Science, Environment and Climate Change Canada, Québec, QC
- Diana Stralberg – Wildlife biologist, Boreal Avian Modelling Project, University of Alberta, Edmonton, AB
Information sources
American Ornithologists' Union (AOU). 1957. Check-list of North American Birds. 5th edition, Washington, D.C.
Atlas of the Breeding Birds of Québec (BBAQ). 2015. Data obtained in response to a request made to the atlas office. Regroupement QuébecOiseaux, Environment Canada’s Canadian Wildlife Service and Bird Studies Canada. Québec, Québec, Canada. [accessed October 2014].
Audubon. 2015. Christmas Bird Count. Web site: https://www.audubon.org/conservation/science/christmas-bird-count [accessed December 2015].
Avery, M.L. 2013. Rusty Blackbird. The Birds of North America Online (A. Poole, Ed.). Ithaca: Cornell Lab of Ornithology. Retrieved from the Birds of North America Online: http://bna.birds.cornell.edu/bna/species/200
Barnard, W.H., C. Mettke-Hofmann, and S.M. Matsuoka. 2010. Prevalence of hematozoa infections among breeding and wintering Rusty Blackbirds. Condor 112:849–853.
Batema, D.L, G.S., Henderson, and L.H. Frederickson. 1985. Wetland invertebrate distribution in bottomland hard- woods as influenced by forest type and flooding regime. Fifth Central Hardwood Conference, University of Illinois, Urbana. Available: www.ncrs.fs.fed.us/pubs/ch/ch05/CHvolume05page196.pdf. [accessed 27 October 2014].
Blackwell, B.F., E. Huszar, G.M. Linz, and D.R.A. Dolbeer. 2003. Lethal control of Red-winged Blackbirds to manage damage to sunflower: an economic evaluation. Journal of Wildlife Management 67:818-828.
Blancher, P.J., K.V. Rosenberg, A.O. Panjabi, B. Altman, A.R. Couturier, W.E. Thogmartin, and the Partners in Flight Science Committee. 2013. Handbook to the Partners in Flight Population Estimates Database, Version 2.0. PIF Technical Series No 6. http://www.partnersinflight.org/pubs/ts/
Boatman, N.D., N.W. Brickle, J.D. Hart, T.P. Milsom, A.J. Morris, A.W. Murray, K A. Murray, and P.A. Robertson. 2004. Evidence for the indirect effects of pesticides on farmland birds. Ibis 146:131-143.
Borchert, S.M., and P.C. Stauffer. 2014. Site-specific habitat and landscape associations of Rusty Blackbirds wintering in Louisiana. PowerPoint presentation to the Rusty Blackbird Working Group at the AOU/COS/SCO Joint Meeting: recent advances in Rusty Blackbird research, Estes Park, Colorado.
Boreal Avian Modelling Project (BAM). 2015. Edmonton, AB. Available: www.borealbirds.ca. [accessed August 2015].
Braune, B.M. 1993. Trends and effects of environmental contaminants in arctic seabirds, waterfowl, and other wildlife. Study I. Contaminants in waterfowl: Native harvest in Labrador. In: Synopsis of research conducted under the 1992/93 Northern Contaminants Program, Environmental Studies #70, J.L. Murray and R.G. Shearer, eds. Indian Affairs and Northern Development Canada, Northern Affairs Program, Ottawa.
Breeding Bird Atlas of British Columbia (BBABC). 2015. British Colombia Breeding Bird Atlas. Web site: http://www.birdAtlas.bc.ca/bcdata/maps.jsp?lang=fr. [accessed August 2015].
Breeding Bird Atlas of Manitoba (BBAMB). 2015. Manitoba Breeding Bird Atlas. Web site: http://www.birdAtlas.mb.ca/index_en.jsp. [accessed July 2015],
Brewer, D., A. Diamond, E.J. Woodsworth, B.T. Collins and E.H. Dunn. 2000. Canadian Atlas of Bird Banding. Volume 1: Doves, Cuckoos, and Hummingbirds through Passerines, 1921-1995, Special Publication, Canadian Wildlife Service.
Buckley, S.H. 2013. Rusty Blackbirds in northeastern U.S. industrial forests: a multi-scale study of nest habitat selection and nest survival. MSc thesis, State University of New York, Syracuse, New York. 151 pp.
Burleigh, T.D., and H.S. Peters. 1948. Geographic variation in Newfoundland birds. Proc. Biol. Soc. Wash. 61:111-126.
Cade, T.J. 1953. Aerial feeding of the Rusty Blackbird on mosquitoes. Wilson Bulletin 65:52-53.
Cadman, M.D., P.F.J. Eagles and F.M. Helleiner (eds.). 1987. Atlas of the Breeding Birds of Ontario, University of Waterloo Press, Waterloo, Ontario.
Cadman, M.D., D.A. Sutherland, G.G. Beck, D. Lepage, and A.R. Couturier (eds). 2007. Atlas of the Breeding Birds of Ontario, 2001-2005. Bird Studies Canada, Environment Canada, Ontario Field Ornithologists, Ontario Ministry of Natural Resources, and Ontario Nature. Toronto. xxii + 706 pp.
Campbell, R.W., N.K. Dawa, I. McTaggart-Cowan, J.M. Cooper, G.W. Kaiser, M.C.E. McNall, and G.E.J. Smith. 1997. The Birds of British Columbia. Volume 3: Passerines, Flycatchers through Vireos. UBC Press, Vancouver. 252 pp.
Canadian Migration Monitoring Network (CMMN). 2016. Population trends and seasonal abundance. NatureCounts, Bird Studies Canada. Web site: http://www.bsc-eoc.org/birdmon/cmmn/popindices.jsp [accessed November 2016].
Canham, L. 2014. Rusty Blackbird (Euphagus carolinus) fall migratory habitat dynamics in Missouri. MSc Thesis, Truman State University, Kirksville, Missouri. 48 pp.
Carroll, M.L., J.R.G. Townshend, C.M. DiMiceli, T. Loboda, and R.A Sohlberg. 2011. Shrinking lakes of the Arctic: spatial relationships and trajectory of change. Geophysical Research Letters 38: L20406, doi:10.1029/2011GL049427
Centers for Disease Control and Prevention. 2012. Elimination of Malaria in the United States (1947-1951). Web site: www.cdc.gov. [accessed May 2016].
Chiblow, S. 2013. ATK Source Report on the Rusty Blackbird Euphagus carolinus in Canada. Prepared for Aboriginal Traditional Knowledge subcommittee of the Committee on the Status of Endangered Wildlife in Canada. 7 pp.
Cooke, M. T. 1942. Returns from banded birds-some longevity records of wild birds. Bird-Banding no. 13:110-119.
Corcoran, R. M., J. Lovvorn, and P. Heglund. 2009. Long-term change in limnology and invertebrates in Alaskan boreal wetlands. Hydrobiologia 620:77-89.
COSEWIC. 2006. COSEWIC assessment and status report on the Rusty Blackbird Euphagus carolinus in Canada. Committee on the Status of Endangered Wildlife in Canada. Ottawa. vi + 28 pp. (www.sararegistry.gc.ca/status/status_e.cfm).
Crewe, T.L., J.D. McCracken, P.D. Taylor, D. Lepage, and A.E. Heagy. 2008. The Canadian Migration Monitoring Network - Réseau Canadien de Surveillance Des Migrations: Ten-Year Report on Monitoring Landbird Population Change. CMMN-RCSM Scientific Technical Report #1.
Custer, T.W., C.M. Custer, P.M. Dummer, G.M. Linz, L. Sileo, R.S. Stahl, and J.J. Johnston. 2003. Nontarget bird exposure to DRC-1339 during fall in North Dakota and spring in South Dakota, pp. 64-70, in G.M. Linz (ed.), Management of North American Blackbirds, National Wildlife Research Center, Fort Collins, Colorado.
Cyr, A., and J. Larivée. 1995. Atlas saisonnier des oiseaux du Québec. Les Presses de l’Université de Sherbrooke et la Société de Loisir ornithologique de l’Estrie, Sherbrooke, Québec.
Dahl, T.E. 1990. Wetlands Losses in the United States, 1780s to 1980s. U.S. Department of Interior, U.S. Fish and Wildlife Service, Washington, DC.
Darveau, M., P. Beauchesne, L. Bélanger, J. Huot, and P. Larue. 1995. Riparian forest strips as habitat for breeding birds in boreal forest. Journal of Wildlife Management 59:67-78.
DeLeon, E.E. 2012. Ecology of Rusty Blackbirds wintering in Louisiana: seasonal trends, flock composition and habitat associations. MSc. thesis, Louisiana State University. 106 pp.
DesGranges, J.-L., and B. Houde. 1989. Influence de l’acidité et d’autres paramètres environnementaux sur la distribution des oiseaux lacustres au Québec, pp. 7-44, in J.-L. DesGranges (éd.), Étude des effets de l’acidification sur la faune aquatique au Canada : les oiseaux lacustres et leurs habitats au Québec, Environnement Canada, Service canadien de la faune, Région du Québec, Publication hors série no 67, 73 p.
DesGranges, J.-L., J. Rodrigue, B. Tardif, and M. Laperle. 1998. Mercury accumulation and biomagnification in Ospreys (Pandion haliaetus) in the James Bay and Hudson Bay regions of Québec. Archives of Environmental Contamination and Toxicology 35:330-341.
Dias, N.W. 2008. Reliable locations of significant Rusty Blackbird flocks in South Carolina: characteristics and shared features of the most reliable sites. PowerPoint presentation to the Rusty Blackbird Working Group. Web site: http://rustyblackbird.org/wp-content/uploads/7-RUBL_Dias.pdf [accessed August 2015].
Dolbeer, R.A., D.F. Mott, and J.L. Belant. 1997. Blackbirds and starlings killed at winter roosts from PA-14 applications, 1974-1992: implications for regional population management. Proceedings Eastern Wildlife Damage Control Conference. 7:77-86.
Ducks Unlimited. 2007. Wetlands Reserve Program: helping to restore bottomland hardwoods in the Mississippi Alluvial Valley. Ducks Unlimited. <http://www.ducks.org/Conservation/Government Affairs/1622/WetlandsReserveProgram.html, July 2007>.
Dunn, E., B. Drolet, D. Collister, D. Okines, and J. McCracken. 2016. Canadian Migration Monitoring Network status assessment. Unpublished report. 9 pp. + Appendices.
eBird Canada. 2015. Web site: http://ebird.org/content/canada/ [accessed August 2015].
Edmonds, S.T., D.C. Evers, D.A. Cristol, C. Mettke-Hofmann, L. L., Powell, A. J. McGann, J.W. Argimer, O.P. Lane, D.F. Tessler, and P. Newell, K. Heyden, and N.J. O’Driscoll. 2010. Geographic and seasonal variation in mercury exposure of the declining Rusty Blackbird. Condor 112:789–799.
Ellison, W.G. 1990. The status and habitat of the Rusty Blackbird in Caledonia and Essex counties. Vermont Fish and Wildlife Department, Woodstock, Vermont.
Environment Canada. 1991. The State of Canada’s Environment - 1991. Environment Canada. Minister of Supply and Services, Ottawa.
Environment Canada. 2009. Status of Birds in Canada website: http://ec.gc.ca/soc-sbc/index-eng.aspx?sY=2011&sL=e [accessed December 2015].
Environment Canada. 2014. Breeding Bird Survey Results. Trend results for Rusty Blackbird. Web site http://www.ec.gc.ca/ron-bbs/P004/A001/?lang=f&m=s&r=RUBL&p=L [accessed in July 2015].
Environment Canada. 2015. Management Plan for the Rusty Blackbird (Euphagus carolinus) in Canada. Species at Risk Act Management Plan Series. Environment Canada, Ottawa. iv + 26 pp.
Erskine, A.J. 1992. Atlas of Breeding Birds of the Maritime Provinces. Nimbus Publ. Ltd. and Nova Scotia Mus., Halifax.
Evans, B.S., L.L. Powell, and R. Greenberg. 2014. The distribution of Rusty Blackbirds on their wintering grounds: potential hotpots and habitat associations. PowerPoint presentation to the Rusty Blackbird Working Group at the AOU/COS/SCO Joint Meeting: Recent advances in Rusty Blackbird research. Estes Park, Colorado. Web site: http://rustyblackbird.org/wp-content/uploads/EvansRustyBlackBirdAOU.pdf [accessed in August 2015].
Evers, D.C., N.M. Burgess, L. Champoux, B. Hoskins, A. Major, W.M. Goodale, R.J. Taylor, R. Poppenga, and T. Daigle. 2005. Patterns and interpretation of mercury exposure in freshwater avian communities in northeastern North America. Ecotoxicology 14:193–221.
Federation of Alberta Naturalists (FAN). 2007. The Atlas of Breeding Birds of Alberta: a second look. Federation of Alberta Naturalists, Edmonton, vii + 626 p.
Fisher, C., and L. Powell, 2013. Rusty Blackbird. In The Second Atlas of Breeding Birds of Vermont. Rosalind B. Renfrew, editor. University Press of New England. 576 pp.
Fitzgerald, W.F., D.R. Engstrom, R.P. Mason, and E.A. Nater. 1998. The case for atmospheric mercury contamination in remote areas. Environmental Science & Technology 32:1-7.
Fraser, D., pers. comm. 2017. Email correspondence to R.D. Elliot. April 2017.
Fredrickson, L.H., and F.A Reid. 1988. Nutritional values of waterfowl foods. Waterfowl Management Handbook. Fish and Wildlife Leaflet 13.1.1, U.S. Fish and Wildlife Service, Washington, DC. 6 pp.
Gauthier, J., and Y. Aubry. (Eds). 1996. The Breeding Birds of Québec: Atlas of the Breeding Birds of Southern Québec. Association québécoise des groupes d’ornithologues, Société québécoise de protection des oiseaux, Service Canadien de la faune, Environment Canada, Montréal, xviii + 1302 pp.
Gauthier, J., pers. comm. 2004. Communication with C. Savignac. July 2004.
Global Forest Watch Canada. 2000. Canada’s Forests at a Crossroads: An Assessment in the Year 2000. World Resources Institute, Washington, DC.
Godfrey, W.E. 1986. The Birds of Canada. Revised Edition. National Museum of Natural Sciences, National Museums of Canada, Ottawa, ON. 595 pp.
Gouvernement du Québec. 2006. Liste des espèces désignées menaces ou vulnérables au Québec. Web site: http://www3.mffp.gouv.qc.ca/faune/especes/menacees/liste.asp#oiseaux. accessed June 2016.
Government of Saskatchewan. 2015. Saskatchewan bird atlas. Web site: http://gisweb1.serm.gov.sk.ca/imf/imf.jsp?site=birds accessed June 2015.
Graveland, J., R. van der Wal, J.H. van Balen, and J. van Noordwijk. 1994. Poor reproduction in forest passerines from decline of snail abundance on acidified soils. Nature 368:446-448.
Greenberg, R., and S. Droege. 1999. On the decline of the Rusty Blackbird and the use of ornithological literature to document long-term population trends. Conservation Biology 13:553-559.
Greenberg, R., and S.M. Matsuoka. 2010. Rusty Blackbird: mysteries of a species in decline. Condor 112: 770-777.
Greenberg, R., D.W. Demarest, S.M. Matsuoka, C. Mettke-Hofmann, D. Evers, P.B. Hamel, J. Luscier, L.L. Powell, D. Shaw, M.L. Avery, K.A. Hobson, P.J. Blancher, and D.K. Niven. 2011. Understanding declines in Rusty Blackbirds. Pp. 107–126 in J.V. Wells (editor). Boreal birds of North America: a hemispheric view of their conservation links and significance. Studies in Avian Biology (no. 41). University of California Press, Berkeley, CA.
Griffiths, M., and D. Woynillowicz. 2003. Oil and troubled waters: Reducing the impact of the oil and gas industry on Alberta’s water resources. Pembina Institute for Appropriate Development. 35 pp.
Hallmann, C.A., R.P. Foppen, C.A. van Turnhout, H. de Kroon, and E. Jongejans. 2014. Declines in insectivorous birds are associated with high neonicotinoid concentrations. Nature 511:341-343.
Hamel, P.B., and E. Ozdenerol. 2008. Using the spatial filtering process to evaluate the nonbreeding range of Rusty Blackbird Euphagus carolinus. Proceedings of the Fourth International Partners in Flight Conference: Tundra to the Tropics. Pp. 334–340.
Hamel, P.B., D. DeSteven, T. Leininger, and R. Wilson. 2009. Historical trends in Rusty Blackbird nonbreeding habitat in forested wetlands. Proceedings of the Fourth International Partners in Flight Conference: Tundra to the Tropics. pp. 341–353.
Hayeur, G. 2001. Synthèse des connaissances environnementales acquises en milieu nordique de 1970 à 2000, Montréal, Hydro-Québec. 110 pp.
Hefner, J.M., B.O. Wilen, T.E. Dahl, and W.E. Frayer. 1994. Southeastern wetlands: status and trends, mid-1970s to mid-1980s. U.S. Fish and Wildlife Service and U.S. Environmental Protection Agency, Atlanta, GA.
Heisterberg, J.F., A.R. Stickley, Jr., K.M. Garner, and P.D. Foster, Jr. 1987. Controlling blackbirds and starlings at winter roosts using PA-14. Proceedings of the Eastern Wildlife Damage Control Conference 3:177–183.
Hobson, K.A., E.M. Bayne, and S.L. Van Wilgenburg. 2002. Large-scale conversion of forest to agriculture in the Boreal Plains of Saskatchewan. Conservation Biology 16:1530-1541.
Hobson, K.A., R. Greenberg, S.l. Van Wilgenburg, and C. Mettke-Hofmann. 2010. Migratory connectivity in the Rusty Blackbird: isotopic evidence from feathers of historical and con- temporary specimens. Condor 112:778–788.
Hobson, K. A., S. L. Van Wilgenburg, E. H. Dunn, D. J. T. Hussell, P. D. Taylor, and D. M. Collister. 2015. Predicting origins of passerines migrating through Canadian migration monitoring stations using stable-hydrogen isotope analyses of feathers: a new tool for bird conservation. Avian Conservation and Ecology 10: 3. http://dx.doi.org/10.5751/ACE-00719-100103
Hood, G.A., and S.E. Bayley. 2008. Beaver (Castor canadensis) mitigate the effects of climate on the area of open water in boreal wetlands in western Canada. Biological Conservation 141:556-567
Houghton, R.A. 2002. Magnitude, distribution and causes of terrestrial carbon sinks and some implications for policy. Climate Policy 2:71–88.
Humphries, P., and K.O. Winemiller. 2009. Historical impacts on river fauna, shifting baselines, and challenges for restoration. BioScience 59:673-684.
Ide, F.P. 1967. Effects of forest spraying with DDT on aquatic insects of salmon streams in New Brunswick. Journal of the Fisheries Research Board of Canada 24:769-805.
International Rusty Blackbird Working Group. 2013. Web Site:http://rustyblackbird.org [accessed August 2015].
Johnson, J.A., S.M. Matsuoka, D.F. Tessler, R. Greenberg, and J.W. Fox. 2012. Identifying migratory pathways used by Rusty Blackbirds breeding in southcentral Alaska. The Wilson Journal of Ornithology 124:698–703.
Johnston, C.A., and R.J. Naiman. 1990. Browse selection by beaver: effects on riparian forest composition. Canadian Journal of Forest Research 20:1036–1043.
Kennard, F.H. 1920. Notes on the breeding habits of the Rusty Blackbird in northern New England. Auk 37:412-422.
King, S.L., D.J. Twedt, and R.R. Wilson. 2006. The role of the Wetland Reserve Program in conservation efforts in the Mississippi River Alluvial Valley. Wildlife Society Bulletin 34:914–920.
Klaminder, J., K. Yoo, J. Rydberg, and R. Giesler. 2008. An explorative study of mercury export from a thawing palsa mire. Journal of Geophysical Research Biogeosciences 113:1–9.
Klein, E., E.E. Berg, and R. Dial. 2005. Wetland drying and succession across the Kenai Peninsula Lowlands, south-central Alaska. Canadian Journal of Forest Research 35:1931–1941.
Kling, G.W., K. Hayhoe, L.B. Johnson, J.J. Magnuson, S. Polasky, S.K. Robinson, B.J. Shuter, M.M. Wander, D.J. Wuebbles, D.R. Zak, R.L. Lindroth, S.C. Moser, and M.L. Wilson. 2003. Confronting Climate Change in the Great Lakes Region: Impacts on our Communities and Ecosystems. The Union of Concerned Scientists, Cambridge (Massachusetts), and The Ecological Society of America, Washington, DC. 92 pp.
Kraus, D. pers. comm. 2016. Nature Conservancy of Canada. Email communication with J. McCracken. November 2016.
Lanyon, S.M., and K.E. Omland. 1999. A molecular phylogeny of the blackbirds (Icteridae): five lineages revealed by cytochrome-B sequence data. Auk 116:629-639.
Larivée, J. 2015. Étude des populations d'oiseaux du Québec (Version 2015-05-15) [base de données]. Rimouski, Québec : Regroupement QuébecOiseaux.
Larue, P., L. Bélanger, and J. Huot. 1995. Riparian edge effects on boreal balsam fir bird communities. Canadian Journal of Forest Research 25:555-566.
La Sorte, F.A., D. Fink, W.M. Hochachka, and S. Kelling. 2016. Convergence of broad-scale migration strategies in terrestrial birds. Proceedings of the Royal Society B.
Linz, G.M., M.J. Kenyon, H.J. Homan, and W.J. Bleier. 2002. Avian use of rice baited corn stubble in east-central South Dakota. International Biodeterioration and Biodegradation 49:179-184.
Loomis, D. 2013. Reproductive Success and Foraging Ecology of the Rusty Blackbird on the Copper River Delta, Alaska. MSc thesis, Oregon State University. 85 pp.
Luscier, J.D., S.E. Lehnen, and K.G. Smith. 2010. Habitat occupancy by Rusty Blackbirds wintering in the Lower Mississippi Alluvial Valley. Condor 112:841–848.
Machtans, C.S., S.L.V. Wilgenburg, L.A. Armer, and K.A. Hobson. 2007. Retrospective comparison of the occurrence and abundance of Rusty Blackbird in the Mackenzie Valley, Northwest Territories. Avian Conservation and Ecology 2(3): 1. Available at http://www.ace-eco.org/vol2/iss1/art3/.
Main, A.R., J.V. Headley, K.M. Peru, N.L. Michel, A.J. Cessna, and C.A. Morrissey. 2014. Widespread use and frequent detection of neonicotinoid insecticides in wetlands of Canada's prairie pothole region. PLoS One 9(3): e92821.
Manitoba Avian Research Committee. 2003. The Birds of Manitoba. Winnipeg. 600 pp.
Martin, T.E. 1995. Avian life history evolution in relation to nest sites, nest predation, and food. Ecological Monographs 65:101–127.
Matsuoka, S.M., D. Shaw, and J.A. Johnson. 2008. Assessing the value of Department of Defense lands in Alaska to a declining species, the Rusty Blackbird. National and International Initiatives. U.S. Fish and Wildlife Service, Migratory Bird Management, Anchorage, AK. 27 pp.
Matsuoka, S.M., D. Shaw, and J.A. Johnson. 2010a. Estimating the abundance of nesting Rusty Blackbirds in relation to wetland habitats in Alaska. Condor 112:825–833.
Matsuoka, S.M., D. Shaw, P.H. Sinclair, J.A. Johnson, R.M. Corcoran, N.C. Dau, P.M. Meyers, and N.A. Rojek. 2010b. Nesting ecology of the Rusty Blackbird in Alaska and Canada. Condor 112:810–824.
McClure, C.J.W., B.W. Rolek, K. McDonald, and G.E. Hill. 2012. Climate change and the decline of a once common bird. Ecology and Evolution 2:370–378.
McCormack, M. 2012. Occupancy of Rusty Blackbirds (Euphagus carolinus) in the Adirondack Region of New York State. MSc thesis, Green Mountain College, Poultney, VT. 47 pp.
McDowell, D.M., and R.J. Naiman. 1986. Structure and function of a benthic invertebrate stream community as influenced by beaver (Castor canadensis). Oecologia 68:481-489.
Meanley, B. 1976. Distribution and ecology of blackbird and starling roosts in the United States. Progress report of the U.S. Fish and Wildlife Service.
Mettke-Hofmann, C., P.B. Hamel, G. Hofmann, T.J. Zenzal Jr., A. Pellegrini, J. Malpass, M. Garfinkel, N. Schiff, and R. Greenberg. 2015. Competition and habitat quality influence age and sex distribution in wintering Rusty Blackbirds. PLoS ONE 10(5): e0123775.
Mineau, P. 2005. Direct losses of birds to pesticides - beginnings of a quantification. Pp. 1065-1070 In Bird Conservation Implementation and Integration in the Americas: Proceedings of the Third International Partners in Flight Conference 2002. USDA Forest Service, GTR-PSW-191. Albany, CA.
Mineau, P., C.M. Downes, D.A. Kirk, E. Bayne, and M. Csizy. 2005. Patterns of bird species abundance in relation to granular insecticide use in the Canadian prairies. Ecoscience 12:267-278.
Mineau, P., and M. Whiteside. 2013. Pesticide acute toxicity is a better correlate of US grassland bird declines than agricultural intensification. PLoS One 8(2): e57457.
Morse, D.H. 1977. Feeding behaviour and predator avoidance in heterospecific groups. BioScience 27:332-339.
Muir, D.C., R, Wagemann, B.T. Hargrave, D.J. Thomas, D.B. Peakall, and R.J. Norstrom. 1995. Spatial trends and historical profiles of organochlorine pesticides in Arctic lake sediments. Science of the Total Environment 160/161:444-457.
Naiman, R.J., C.A. Johnston, and J.C. Kelley. 1988. Alteration of North American streams by beaver. Bioscience 39:753-762.
NatureServe. 2015. NatureServe Explorer: An online encyclopedia of life [application Web], version 4.0. NatureServe, Arlington, Virginia. Available at http://www.natureserve.org/explorer accessed July 2015.
Newell, P.J. 2013. Winter Ecology of the Rusty Blackbird (Euphagus carolinus). PhD Thesis, University of Georgia, Athens, Georgia. 182 pp.
Niven, D.K., J.R. Sauer, G.S. Butcher, and W.A. Link. 2004. Christmas Bird Count provides insights into population change in land birds that breed in the boreal forest. American Birds 58:10–20.
Norment, C.J., A. Hall, and P. Hendricks. 1999. Important bird and mammal records in the Thelon River Valley, Northwest Territories: range expansions and possible causes. Canadian Field-Naturalist 113:375-385.
Novak, M., 1987. Beaver. pp. 283–312 In: Novak, M., Baker, J.A., Obbard, M.E., Malloch, B. (eds.), Wild Furbearer Management and Conservation in North America. Ontario Trappers Association, Toronto.
Orians, G.H. 1985. Blackbirds of the Americas. University of Washington Press, Seattle.
Parks Canada. 2015. Biotic Web Explorer. Web site: http://www.pc.gc.ca/apps/bos/bosfieldselection_e.asp?oqqc=aqs [accessed August 2015].
Partners in Flight Science Committee. 2013. Population Estimates Database, version 2013. Web site http://rmbo.org/pifpopestimates. [accessed July 2015].
Peters, H.S. and T.D. Burleigh. 1951. The Birds of Newfoundland. Houghton Mifflin Co. Boston, MA.
Popko, R. pers. comm. 2014. Wildlife Technician, Resources, Wildlife Ann Economic Development, Northwest Territories government, Norman Wells. Meeting with C. Savignac. July 2014.
Porvari, P., M. Verta, J. Munthe, and M. Haapanen. 2003. Forestry practices increase mercury and methylmercury output from boreal forest catchments. Environmental Science & Technology 37:2389-2393.
Powell, L.L. 2008. Habitat occupancy, status, and reproductive ecology of Rusty Blackbirds in New England. M.S. thesis, University of Maine, Orono, Maine.
Powell, L.L., T.P. Hodgman, and W.E. Glanz. 2010a. Home ranges of Rusty Blackbirds breeding in wetlands: how much would buffers from timber harvest protect habitat? The Condor 112:834–840.
Powell, L.L., T.P. Hodgman, W.E. Glanz, J.D. Osenton, and C.M. Fisher. 2010b. Nest-site selection and nest survival of the Rusty Blackbird: does timber management adjacent to wetlands create ecological traps? Condor 112:800–809.
Powell, L.L., T.P. Hodgman, W.E. Glanz, J.D. Osenton, and D.M. Ellis. 2010c. A loose colony of Rusty Blackbirds nesting in northern Maine. Northeastern Naturalist 17:639-646.
Powell L.L., T.P. Hodgman, I.J. Fiske, and W.E. Glanz. 2014. Habitat occupancy of Rusty Blackbirds (Euphagus carolinus) breeding in northern New England, USA. The Condor 116:122-133.
Price, D.T., R. Alfaro, K. Brown, M. Flannigan, R. Fleming, E. Hogg, M. Girardin, T. Lakusta, M. Johnston, and D. McKenney. 2013. Anticipating the consequences of climate change for Canada’s boreal forest ecosystems. Environmental Reviews 21:322-365.
Pyle, P. 1997. Identification Guide to North American Birds. Slate Creek Press, Bolinas, California.
Rich, T.D., C.J. Beardmore, H. Berlanga, P.J. Blancher, M.S.W. Bradstreet, G.S. Butcher, D.W. Demarest, E.H. Dunn, W.C. Hunter, E.E. Iñigo-Elias, J.A. Kennedy, A.M. Martell, A.O. Panjabe, D.N. Pashley, K.V. Rosenberg, C.M. Rustay, J.S. Wendt, and T.C. Will. 2004. Partners in Flight North American Landbird Conservation Plan. Cornell Lab of Ornithology. Ithaca, NY.
Riordan, B., D. Verbyla, and A.D. McGuire. 2006. Shrinking ponds in subarctic Alaska based on 1950–2002 remotely sensed images. Journal of Geophysical Research, Biogeosciences 111:G04002.
Robinson, S.K., F.R. Thompson, T.M. Donovan, D.R. Whitehead, and J. Faaborg. 1995. Regional forest fragmentation and the nesting success of migratory birds. Science 267:1987-1990.
Rooney, R.C., S.E. Bayley, and D.W. Schindler. 2012. Oil sands mining and reclamation cause massive loss of peatland and stored carbon. Proceedings of the National Academy of Sciences 109, no. 13 (2012). http://www.pnas.org/content/early/2012/03/06/11176 93108.full.pdf
Rousseu, F., and B. Drolet. 2015. Prediction of the nesting phenology of birds in Canada. In J. Hussell and D. Lepage. 2015. Bird Nesting Calendar Query Tool. Project NestWatch. Bird Studies Canada, URL: http://www.birdscanada.org/volunteer/pnw/rnest/warning.jsp [accessed July 2015].
Sauer, J.R., J.E. Hines, J.E. Fallon, K.L. Pardieck, D.J. Ziolkowski, Jr., and W.A. Link. 2014. The North American Breeding Bird Survey, Results and Analysis 1966 - 2012. Version 02.19.2014 USGS Patuxent Wildlife Research Center, Laurel, MD.
Sauer, J.R., S. Schwartz, and B. Hoover. 1996. The Christmas Bird Count home page. Version 95.1. Patuxent Wildlife Research Center, Laurel, MD.
Savard, J-P. L., M. Cousineau, and B. Drolet. 2011. Exploratory analysis of correlates of the abundance of Rusty Blackbirds (Euphagus carolinus) during fall migration. Ecoscience 18:402-408.
Savard, J.-P.L., B. Drolet, and M. Cousineau. 2006. Evidence of a four-year population cycle for the Rusty Blackbird (Euphagus carolinus). Wildlife and Landscape Science Directorate & Canadian Wildlife Service. Presentation to the Rusty Blackbird Working Group. Web site: http://rustyblackbird.org/wp-content/uploads/0945_Savard_Rusty-Blackbirds_BD-2.pdf. [accessed August 2015].
Scarl, J. 2013. Rusty Blackbirds 2012: Building Connections for a Declining Species. Vermont Center for Ecostudies. 15 pp.
Scarl, J.C., C. Foss, D. Demarest, K. Heyden, and P. Sinclair. 2014. Mobilizing Two Countries for Conservation: Rusty Blackbird Spring Migration. PowerPoint presentation to the Rusty blackbird Working Group at the AOU/COS/SCO Joint Meeting: Recent advances in Rusty Blackbird Research. Estes Park, Colorado. Web site: http://rustyblackbird.org/wp-content/uploads/Scarl-AOU-2014-Rusties-FINAL-smaller.pdf [accessed August 2015].
Scheuhammer, A.M., M.W. Meyer, M.B. Sandheinrich, and M.W. Murray. 2007. Effects of environmental methylmercury on the health of wild birds, mammals, and fish. Ambio 36:12-19.
Schindler, D.W. 1988. Effects of acid rain on freshwater ecosystems. Science 239:149-239.
Scurr, A. 2013. Mating system and social behavior of Rusty Blackbirds on Yukon Flats National Wildlife Refuge. MSc. thesis, Humboldt State University. 31 pp.
Shoch, D.T., G. Kaster, A. Hohl, and R. Souter. 2009. Carbon storage of bottomland hardwood afforestation in the Lower Mississippi Valley, USA. Wetlands 29:535–542.
Sinclair, P.H., W.A. Nixon, C.D. Eckert, and N.L. Hughes. 2003. Birds of the Yukon Territory. UBC Press, Vancouver. 595 pp.
Smith, A.C., M.-A.R. Hudson, C. Downes, and C.M. Francis. 2014. Estimating breeding bird survey trends and annual indices for Canada: how do the new hierarchical Bayesian estimates differ from previous estimates? Canadian Field-Naturalist 128:119–134.
Smithsonian Institution. 2015. Smithsonian Migration Bird Center. The Rusty Blackbird Blitz--Year 3. Web site http://nationalzoo.si.edu/scbi/migratorybirds/blog/?id=333 [accessed July 2015].
Soykan, C.U., J. Sauer, J.G. Schuetz, G.S. LeBaron, K. Dale, and G.M. Langham. 2016. Population trends for North American winter birds based on hierarchical models. Ecosphere 7(5):e01351. 10.1002/ecs2.1351
Spindler, M.A., and B. Kessel. 1980. Avian population and habitat use in interior Alaska taiga. Syesis 13:61-104.
Stewart, D., and D. Chisholm. 1971. Long-term persistence of BHC, DDT and chlordane in sandy loam soil. Canadian Journal of Soil Science 51:379–383.
Stewart, R.L.M., K.A. Bredin, A.R. Couturier, A.G. Horn, D. Lepage, S. Makepeace, P.D. Taylor, M.-A. Villard, and R.M. Whittam (eds.). 2015. Second Atlas of Breeding Birds of the Maritime Provinces. Bird Studies Canada, Environment Canada, Natural History Society of Prince Edward Island, Nature New Brunswick, New Brunswick Department of Natural Resources, Nova Scotia Bird Society, Nova Scotia Department of Natural Resources, and Prince Edward Island Department of Agriculture and Forestry, Sackville, NB. 528 + 28 pp.
Stralberg, D., E.M. Bayne, S.G. Cumming, P. Solymos, S.J. Song, and F.K.A. Schmiegelow. 2015a. Conservation of future boreal forest bird communities considering lags in vegetation response to climate change: a modified refugia approach. Diversity and Distributions 1–17.
Stralberg, D., S.M. Matsuoka, A. Hamann, E.M. Bayne, P. Solymos, F.K.A. Schmiegelow, X. Wang, S.G. Cumming, and S.J. Song. 2015b. Projecting boreal bird responses to climate change: the signal exceeds the noise. Ecological Applications 25:1112-1128.
Walker, C.H., R.M. Sibly, S.P. Hopkin and D.B. Peakall. 2012. Principles of Ecotoxicology, Fourth Edition. CRC Press. 386 pp.
Wells, J.V. 2011. Boreal forest threats and conservation status. Pp. 1-6 in J.V. Wells (editor). Boreal birds of North America: a hemispheric view of their conservation links and significance. Studies in Avian Biology (no. 41). University of California Press, Berkeley, CA.
Whitaker, D.H., and W.A. Montevecchi. 1999. Breeding bird assemblages inhabiting riparian buffer strips in Newfoundland, Canada. Journal of Wildlife Management 63:167-179.
Wiener, J.G., D.P. Krabbenhoft, G.H. Heinz, and A.M. Scheuhammer. 2003. Ecotoxicology of mercury. Pp. 407-461 In D.J. Hoffman, B.A. Rattner, G.A. Burton and J. Cairns (eds.). Handbook of Ecotoxicology, 2nd edition. CRC Press. Boca Raton, Florida.
Wilkes, F.G., and C.M. Weiss. 1971. The accumulation of DDT by the dragonfly nymph, Tetragoneuria. Transactions of the American Fisheries Society 100:222-226.
Wohner, P.N., C.R. Foss, R. Greenberg, and R. Cooper. 2014. Rusty Blackbird survivorship and habitat selection during the postfledging period. PowerPoint presentation to the Rusty Blackbird Working Group at the AOU/COS/SCO Joint Meeting: Recent advances in Rusty Blackbird Research. Estes Park, Colorado.
World Health Organization. 1989. DDT and its derivatives: Environmental aspects. Environmental health criteria 83. Geneva.
Yoshikawa, K. and L. Hinzman. 2003. Shrinking thermokarst ponds and groundwater dynamics in discontinuous permafrost. Permafrost Periglacial Process. 14:151-160.
Young, J. E., G. A. Sánchez-Azofeifa, S. J. Hannon, and R. Chapman. 2006. Trends in land cover change and isolation of protected areas at the interface of the southern boreal mixedwood and aspen parkland in Alberta, Canada. Forest Ecology and Management 230:151-161.
Biographical summary of report writer
Carl Savignac is the director of Dendroica Environnement et Faune, an avian ecology consulting firm specializing in studies on conservation of species at risk, wetlands conservation and the assessment of the impacts of industrial development projects on avian wildlife and species at risk. Carl has been studying birds for over 25 years and has conducted numerous field studies in several Canadian provinces and territories. He has written a number of scientific reports and publications, including more than a dozen bird species status reports in Canada and Quebec. Finally, he has led a number of stewardship projects on species at risk in southern Quebec, including the Red-headed Woodpecker, Golden-winged Warbler, Cerulean Warbler, Canada Warbler and Peregrine Falcon.
Appendix a. threat calculator for the rusty blackbird (version june 2016)
Threats assessment worksheet
- Species or ecosystem scientific name:
- Rusty Blackbird
- Date:
- 30 May 2016
- Assessor(s):
- Carl Savignac, Jon McCracken, Dave Fraser, Jeff Keith, Suzanne Carrière, Kaytlin Cooper, Marcel Gahbauer, Richard Elliot, Pam Sinclair, Bruno Drolet, Alana Westwood, Steven Van Wilgenberg, Karen Timm, Joanna James
| Threat impact | Threat impact (descriptions) | Level 1 Threat impact counts: high range |
Level 1 Threat impact counts: low range |
|---|---|---|---|
| A | Very high | 0 | 0 |
| B | High | 0 | 0 |
| C | Medium | 0 | 0 |
| D | Low | 6 | 6 |
| - | Calculated overall threat impact: | Medium | Medium |
- Overall threat comments
- As the overall threat is in line with the existing trend, there is no need to make changes.
| Threat | Threat | Impact (calculated) | Impact (calculated) | Scope (next 10 Yrs) | Severity (10 Yrs or 3 Gen.) | Timing | Timing | Comments |
|---|---|---|---|---|---|---|---|---|
| 1 | Residential & commercial development | D | Low | Small (1-10%) | Moderate (11-30%) | High (Continuing) | High (Continuing) | High (Continuing) |
| 1,1 | Housing & urban areas | D | Low | Small (1-10%) | Moderate (11-30%) | High (Continuing) | Habitat loss and degradation in wintering range. Applies only in wintering range (U.S.), as this threat would be negligible on Boreal forest breeding grounds. RUBL uses suburban areas in wintering range, but threat is difficult to assess without more data from the US. | Habitat loss and degradation in wintering range. Applies only in wintering range (U.S.), as this threat would be negligible on Boreal forest breeding grounds. RUBL uses suburban areas in wintering range, but threat is difficult to assess without more data from the US. |
| 1,2 | Commercial & industrial areas | blank cell | Negligible | Negligible (<1%) | Serious (31-70%) | High (Continuing) | Habitat loss and degradation in wintering range. Factories, shopping malls, etc. do not affect breeding individuals, but are a factor on wintering grounds. Serious severity was assigned because species can still use suburban areas but not industrial areas, with considerable industrial/commercial development and expansion associated with urban expansion in the U.S. southeast (the principal wintering range). | Habitat loss and degradation in wintering range. Factories, shopping malls, etc. do not affect breeding individuals, but are a factor on wintering grounds. Serious severity was assigned because species can still use suburban areas but not industrial areas, with considerable industrial/commercial development and expansion associated with urban expansion in the U.S. southeast (the principal wintering range). |
| 1,3 | Tourism & recreation areas | blank cell | Negligible | Negligible (<1%) | Serious (31-70%) | High (Continuing) | Unlikely to be a threat on breeding grounds; perhaps minor on wintering grounds, but tourism and recreation are positively associated with protection/conservation of natural RUBL habitats (e.g., for fishing, hunting) or do not occur where RUBL impacts might be substantial. | Unlikely to be a threat on breeding grounds; perhaps minor on wintering grounds, but tourism and recreation are positively associated with protection/conservation of natural RUBL habitats (e.g., for fishing, hunting) or do not occur where RUBL impacts might be substantial. |
| 2 | Agriculture & aquaculture | D | Low | Large - Restricted (11-70%) | Slight (1-10%) | High (Continuing) | High (Continuing) | High (Continuing) |
| 2,1 | Annual & perennial non-timber crops | D | Low | Large - Restricted (11-70%) | Slight (1-10%) | High (Continuing) | High (Continuing) | Habitat loss and degradation in wintering range. This threat applies to migration and wintering grounds, mostly in the U.S.; does not affect boreal forest breeding grounds. Scope: could be one of major threats due to conversion of natural habitat to agriculture, and changes to existing agricultural land and practices, which occur frequently owing to changes in commodity prices. Uncertainty categories for scope reflect need for more information from U.S. As the species spends time in North Dakota etc. on migration, need to consider agricultural lands in these areas as well. Data on eastern Canadian population (Manitoba eastward) are lacking. Scope includes all birds that experience agricultural practices - affects all individuals as all migrate through agricultural areas. However, conversion of natural habitat to agriculture is less prevalent now than in past. Data in status report show that population levels have been fairly stable over past 10 years, following large population declines. Low-level effects of habitat conversion could still have an effect as habitat has already been degraded. |
| 2,2 | Wood & pulp plantations | blank cell | Negligible | Negligible (<1%) | Serious (31-70%) | High (Continuing) | High (Continuing) | Logging is not considered a direct threat, with mixed effects depending on circumstances. Conversion of natural habitat to pine plantations is a threat where commercial wood and pulp production affects availability and condition of suitable RUBL wintering habitat, within up to 1% of the range. In these areas, it can have serious impacts on RUBL. |
| 2,3 | Livestock farming & ranching | blank cell | blank cell | blank cell | blank cell | blank cell | blank cell | May apply to migrating population in central US. Not included as a threat, due to lack of supporting data. |
| 2,4 | Marine & freshwater aquaculture | blank cell | blank cell | blank cell | blank cell | blank cell | blank cell | Catfish and crayfish farming are two forms of aquaculture that might have impacts on wintering RUBL in the southeast US, due to habitat alteration/degradation, and associated changes in hydrology. However, in some instances, provision of shallowly-flooded wetland habitats associated with crayfish farming are positive for RUBL, in areas where shallowly-flooded wintering habitat would otherwise not be available. |
| 3 | Energy production & mining | D | Low | Small (1-10%) | Serious (31-70%) | High (Continuing) | High (Continuing) | High (Continuing) |
| 3,1 | Oil & gas drilling | D | Low | Small (1-10%) | Serious (31-70%) | High (Continuing) | High (Continuing) | This is not considered a threat for most of the breeding population as this threat is not found east of Manitoba. This threat would be greatest in Alberta, where the population there has been reduced. This threat only incorporates new drilling activities, which are not likely to occur in boreal wetlands. Drilling might occur only on drier sites (small scope). Seismic lines can also cause ecosystem change through linear development and changes in drainage and permafrost regimes. These lines may be maintained and used for other reasons after seismic use is completed. |
| 3,2 | Mining & quarrying | blank cell | Negligible | Negligible (<1%) | Extreme (71-100%) | High (Continuing) | High (Continuing) | Negligible scope with extreme severity as open pit mines and oil sands development could have a marked local negative impact. |
| 3,3 | Renewable energy | blank cell | Negligible | Negligible (<1%) | Unknown | High (Continuing) | High (Continuing) | Hydro power generation is not included here. No data available on wind farm impacts, but thought to have a negligible overall population impact. Although overall impact is unclear due to lack of data, anecdotal evidence suggests that very few birds are affected. |
| 4 | Transportation & service corridors | blank cell | Negligible | Pervasive (71-100%) | Negligible (<1%) | High (Continuing) | High (Continuing) | High (Continuing) |
| 4,1 | Roads & railroads | blank cell | Negligible | Pervasive (71-100%) | Negligible (<1%) | High (Continuing) | High (Continuing) | Impacts include loss of habitat and collisions with vehicles, which only pose a (slight) threat in wintering areas, as there are few roads in wetland breeding grounds. |
| 4,2 | Utility & service lines | blank cell | Negligible | Pervasive (71-100%) | Negligible (<1%) | Unknown | Unknown | Over 70% of population is likely to encounter utility lines, especially in non-breeding seasons. |
| 4,3 | Shipping lanes | blank cell | blank cell | blank cell | blank cell | blank cell | blank cell | blank cell |
| 4,4 | Flight paths | blank cell | blank cell | blank cell | blank cell | blank cell | blank cell | blank cell |
| 5 | Biological resource use | D | Low | Small (1-10%) | Extreme (71-100%) | High (Continuing) | High (Continuing) | blank cell |
| 5,1 | Hunting & collecting terrestrial animals | D | Low | Small (1-10%) | Extreme (71-100%) | High (Continuing) | Includes pest control (e.g., avicide) programs, such as the U.S. blackbird control program. RUBL has been removed from the list of birds that can be destroyed, so impacts are difficult to estimate, because RUBL may still fall victim through close association with other targeted blackbird species. Control programs which exist for roosting birds and in sunflower and rice fields can affect large number of birds, but there are few data on mortality of this species. Not all RUBL winter in regions where this method is used. | blank cell |
| 5,2 | Gathering terrestrial plants | blank cell | blank cell | blank cell | blank cell | blank cell | blank cell | blank cell |
| 5,3 | Logging & wood harvesting | D | Low | Small (1-10%) | Moderate - Slight (1-30%) | High (Continuing) | Part of boreal forest and some of the wintering range are exposed to this threat. Riparian areas are usually, but not always protected (may be harvested during "salvage" logging operations after forest fire or beetle kill). There is evidence that this species uses freshly-logged habitat more than unlogged, but information is lacking on reproductive success in these habitats. Data are needed to confirm effect of logging on breeding, because it could result in an ecological sink. Logging activities may be close to 1% per year by commercial forestry in the boreal forest. Tree harvesting on wintering grounds (e.g., cypress) may have a negative effect. More information is needed on forestry in U.S., but most southeast U.S. commercial timber operations are unlikely to substantively impact the integrity of those habitats for RUBL. Commercial timber operations are often beneficial to RUBL, as they retain forested land a near-natural state, which might otherwise be converted to non-compatible land uses. Many hardwood silviculture practices improve forest structure and heterogeneity as habitat for RUBL. | blank cell |
| 5,4 | Fishing & harvesting aquatic resources | blank cell | blank cell | blank cell | blank cell | blank cell | blank cell | blank cell |
| 6 | Human intrusions & disturbance | blank cell | blank cell | blank cell | blank cell | blank cell | blank cell | blank cell |
| 6,1 | Recreational activities | blank cell | blank cell | blank cell | blank cell | blank cell | blank cell | blank cell |
| 6,2 | War, civil unrest & military exercises | blank cell | blank cell | blank cell | blank cell | blank cell | blank cell | blank cell |
| 6,3 | Work & other activities | blank cell | blank cell | blank cell | blank cell | blank cell | blank cell | blank cell |
| 7 | Natural system modifications | D | Low | Large (31-70%) | Slight (1-10%) | High (Continuing) | High (Continuing) | blank cell |
| 7,1 | Fire & fire suppression | blank cell | Unknown | Small (1-10%) | Unknown | High (Continuing) | Apart from the natural fire regime, fire frequency may be elevated by climate change or other human-related causes. This species may not be negatively affected by forest fires (appears to nest in wetlands in burned as well as unburned forest areas), but this threat included here due to the large range of RUBL. The effect of fire on this species (i.e., severity) is unknown, with more research required. | blank cell |
| 7,2 | Dams & water management/use | D | Low | Large (31-70%) | Slight (1-10%) | High (Continuing) | Only includes new developments -- both flooded and impounded areas and drained sites. Effects may be mixed as management could create more riparian habitat, but may also destroy small wetlands and create larger wetlands that are not as useable by this species. | blank cell |
| 7,3 | Other ecosystem modifications | blank cell | Unknown | Pervasive (71-100%) | Unknown | High (Continuing) | Various activities (logging, etc.) could cause larger ecosystem changes (floods, droughts) hundreds of km away from point source. Linear disturbances (e.g., boreal seismic lines and roads) may present indirect threats by changing hydrology. Reduction of insect prey could occur due to toxins (neonics, mercury, acid rain), which can be carried into wetland systems (on migration and wintering grounds). Acid rain lowers the amount of calcium in prey species (breeding grounds). | blank cell |
| 8 | Invasive & other problematic species & genes | blank cell | Unknown | Unknown | Unknown | Unknown | blank cell | blank cell |
| 8,1 | Invasive non-native/alien species | blank cell | Unknown | Unknown | Unknown | Unknown | Lack of data on avian diseases. More information is needed on potential impact of West Nile virus. | blank cell |
| 8,2 | Problematic native species | blank cell | Unknown | Unknown | Unknown | Unknown | Parasite loads that are higher than expected could indicate stressed individuals. There could be result of increased competition with other blackbirds in boreal due to expanding ranges. Red Squirrel predation may be increasing. Mosquitoes and black flies may be emerging earlier in the spring and preying on nestlings. Raptor populations are increasing because of DDT ban, but presumably returning to near-historical levels. | blank cell |
| 8,3 | Introduced genetic material | blank cell | blank cell | blank cell | blank cell | blank cell | blank cell | blank cell |
| 9 | Pollution | blank cell | Low | Large - Restricted (11-70%) | Slight (1-10%) | High (Continuing) | blank cell | blank cell |
| 9,1 | Household sewage & urban waste water | blank cell | blank cell | blank cell | blank cell | blank cell | blank cell | blank cell |
| 9,2 | Industrial & military effluents | blank cell | blank cell | blank cell | blank cell | blank cell | blank cell | blank cell |
| 9,3 | Agricultural & forestry effluents | blank cell | Low | Large - Restricted (11-70%) | Slight | High (Continuing) | Ingesting corn treated with pesticides - most of population is exposed while migrating and in wintering grounds. Neonics - need to be looked at in wintering grounds - see ecosystem modification section. | blank cell |
| 9,4 | Garbage & solid waste | blank cell | blank cell | blank cell | blank cell | blank cell | blank cell | blank cell |
| 9,5 | Air-borne pollutants | blank cell | Unknown | Pervasive - Large (31-100%) | Unknown | High (Continuing) | Mercury pollution and acid rain. High levels of mercury occur in the eastern part of the range, linked to high levels of acid rain. This species accumulates high levels of mercury from eating insects. | blank cell |
| 9,6 | Excess energy | blank cell | blank cell | blank cell | blank cell | blank cell | blank cell | blank cell |
| 10 | Geological events | blank cell | blank cell | blank cell | blank cell | blank cell | blank cell | blank cell |
| 10,1 | Volcanoes | blank cell | blank cell | blank cell | blank cell | blank cell | blank cell | blank cell |
| 10,2 | Earthquakes/tsunamis | blank cell | blank cell | blank cell | blank cell | blank cell | blank cell | blank cell |
| 10,3 | Avalanches/landslides | blank cell | blank cell | blank cell | blank cell | blank cell | blank cell | blank cell |
| 11 | Climate change & severe weather | blank cell | Unknown | Pervasive - Large (31-100%) | Unknown | High (Continuing) | High (Continuing) | blank cell |
| 11,1 | Habitat shifting & alteration | blank cell | Unknown | Pervasive - Large (31-100%) | Unknown | High (Continuing) | Could be a larger impact in the longer term, beyond 10 years. Wetlands are likely to shrink, but less than 10% decline in 10-yr period. Melting permafrost could create new wetlands, but in the north bogs are turning into fens, which do not suit this species. Impacts could include shifts in insect emergence, potentially causing a timing mis-match with nesting season. | blank cell |
| 11,2 | Droughts | blank cell | Unknown | Unknown | Unknown | Unknown | May be more of an issue in the north. Need more information on droughts in wintering grounds. | blank cell |
| 11,3 | Temperature extremes | blank cell | blank cell | blank cell | blank cell | blank cell | blank cell | blank cell |
| 11,4 | Storms & flooding | blank cell | Unknown | Unknown | Unknown | Unknown | Possible risks of flooding/late snowstorms on nests, which are often over or near water. | blank cell |