Wild Species 2005: chapter 2
Vascular plants
Plante vasculaire : Spermatophytes; plante caractérisée par ses racines, pousses et feuilles contenant des tissus vasculaires spécialisés dans lesquels circulent les liquides - Dictionary of the Environment.

Quick facts
- There are over 260 000 species of vascular plants in the world. More than 95% of vascular plants are flowering plants, also called angiosperms (e.g. grasses, orchids, maple trees). The other types of vascular plants are gymnosperms (cone-bearing trees, e.g. pine trees, spruce trees) and seedless plants (e.g. ferns, horsetails).
- 5074 species of vascular plants have been found in Canada, of which 51% have Canada General Status Ranks (Canada ranks) of Secure, 11% have Canada ranks of May Be At Risk, 9% have Canada ranks of Sensitive and 2% have Canada ranks of At Risk.
- 24% of vascular plant species have a Canada rank of Exotic (not native to Canada), the highest proportion of Exotic species of any group covered in this report.
- 194 species of ferns and orchids were ranked in both 2000 and 2005. The Canada ranks of 18 of these species (9%) have been changed since 2000; 11 species have been moved into categories with a higher level of risk, six species have been moved into categories with a lower level of risk and one species has been moved into the Extirpated category. The majority of changes were due to improved information; none of the changes were due to biological changes in species distribution, abundance or threats.
Background
Green plants play a critical role in maintaining life on earth, because they are one of the few groups of organisms that can make their own food. Through the chemical process of photosynthesis, green plants use energy from the sun to convert water and carbon dioxide into oxygen and sugar, which is used as a food source by plants, and by plant-eating animals. Therefore, green plants produce two of the resources that all animals need to survive; food and oxygen. In addition, plants play important roles in the environment by helping to regulate climate, providing habitat for wildlife, contributing to nutrient cycles and soil creation, improving air and water quality, and reducing soil erosion.
Most familiar plants, such as ferns, orchids, herbs, grasses, shrubs and trees are vascular plants. All vascular plants have roots, leaves and a vascular system, which transports water, sugars and nutrients throughout the plant. Vascular plants are the largest group of green plants in the world, and form the dominant vegetation over much of the earth's landmass.
The oldest vascular plants are the seedless plants, including the ferns, club mosses, and horsetails. Seedless plants dominated the world in the Carboniferous period, approximately 300 million years ago. Dead plants from this period formed some of the coal beds from which coal is still mined today. All seedless plants reproduce using spores. For more information about seedless plants, please visit the Ferns section in Wild Species 2000.
From the seedless plants evolved two groups of seed plants; the cone-bearing plants (the gymnosperms, e.g. pine trees, spruce trees) and the flowering plants (the angiosperms e.g. grasses, orchids, maple trees, oak trees). As their name suggests, seed plants use seeds rather than spores for reproduction. Seeds are simply embryos surrounded by a seed coat, which protects the embryo from drought, extreme temperatures and other harsh conditions. Most seeds also contain a food source for the developing plant. Angiosperms surround their seeds with an additional layer of protection, the fruit. The fruit protects the seeds, and often provides a mechanism for spreading them over long distances. Fruits can be fleshy (e.g. blueberries, cranberries) or dry (e.g. the keys of the Sugar Maple, Acer saccharum, are actually a type of fruit!) vascular plants are literally rooted to one spot. So, how do plants find a mate and reproduce? Gymnosperms produce pollen (male sex cells) and eggs in separate male and female cones. The pollen is released into the air and carried by the wind to a female cone, where it will fertilize the eggs. The process of transporting pollen from the male cone to the female cone is called pollination. The chance of successful pollination is fairly small, so gymnosperms produce large amounts of pollen to increase the chance that some of it will meet with a female cone of the same species.
In angiosperms, all the sex organs are located within the flowers (although male and female organs are not necessarily found within the same flower, or even on the same plant). While some angiosperms rely on wind pollination (e.g. grasses), most rely on animal pollinators such as insects, birds and even bats, to carry pollen between their flowers. Angiosperms attract potential pollinators with brightly coloured petals, sweet fragrances, or by producing nectar. Some species of angiosperms have evolved to attract very specific pollinators. For example, the main pollinators of the Cardinal Flower (Lobelia cardinalis) are hummingbirds, which are attracted to the plant by the bright red flowers. The long, narrow cardinal flowers are the perfect shape for a hummingbird to insert its bill and retrieve the nectar. As the hummingbird feeds, pollen is deposited on its head; the pollen will be brushed off at the next flower, where it will fertilize the eggs. By attracting specific types or species of pollinators, plants can increase the chances that their pollen reaches another flower of the same species.
Plants are amazing chemical factories that make a variety of different products, from defensive chemicals that protect the plant from predation, disease and parasites, to hormones that control the plant's growth. Humans have long known that many of the chemicals that plants produce have useful medicinal properties. For example, Common Yarrow (Achillea millefolium) is a well-known traditional cure for staunching wounds and treating fevers, colds and other ailments. Today, Common Yarrow can be found in more than 20 pharmaceutical products sold in Canada.
Status of knowledge in Canada
Even before plants were first domesticated, roughly 10 000 years ago, they provided humans with food and raw material. Today, plants form the basis of our agricultural and forestry systems, provide raw materials for clothing, buildings, paper products and medicines and play an important role in the biotechnology industry.
The study of plants has a long history in Canada, from Aboriginal Peoples who relied on plants for food, shelter, clothing, raw materials and medicines, to the early European settlers, some of whom became famous botanists (e.g. Catherine Parr Trail, John Macoun). Much of today's research is centered on plants that are important for agriculture, forestry or medicine, using new genetic and molecular tools to study a huge variety of topics including plant physiology, genetics, and interactions between plants and pests.
Relative to other species groups covered in this report, such as marine fishes, the distribution and status of many vascular plant species in Canada is fairly well known, particularly in southern Canada. Nevertheless, systematic surveys are still uncovering new information, such as the discovery of a new tree species for Canada, the Swamp Cottonwood (Populus heterophylla, see species spotlight for more details). As well as discovering new species, systematic surveys improve information on the distribution and abundance of vascular plants. For example, in New Brunswick, the first systematic rare plant survey of the upper St. John River in 2001 and 2002, showed that two species of grasses, Mat Muhly (Muhlenbergia richardsonis) and Little Bluestem (Schizachyrium scoparium), thought to be rare in the province, were actually more common than previously believed. Within New Brunswick, these species are ranked Secure and Sensitive respectively.
The distribution of vascular plants in remote areas, and in northern Canada is less well known than that of plants in southern Canada. This is partly because fewer people (both amateurs and professionals) are studying plants in these regions, despite the presence of unique plant communities and endemic species found nowhere else on earth. In addition, many plant specimens from northern Canada have been housed in national collections in southern Canada. While some of these collections have been well documented and catalogued (e.g. the National Herbarium at the Canadian Museum of Nature), others have only recently been fully catalogued to reveal new information about northern vascular plants.
Plant ecology is the study of how plants interact with, and are affected by, the world around them; both the physical world (e.g. temperature, soil type, light levels) and the 'biological' world (interactions with other plants, animals, fungi etc). This is important for understanding a variety of problems including plant distribution, how plants survive in different environments, and plant productivity. Plant ecology also helps researchers understand how changes in the environment (e.g. climate change, invasion of exotic species) might affect plant communities. For example, researchers in Quebec, working on grasses in pastureland, have shown that exposure to increased levels of carbon dioxide can influence plant succession (changes in community composition through time) and species richness. Knowledge of plant ecology can also help conserve and restore native plant communities. For example, Canadian researchers are working on restoring surface vegetation to bogs that have been mined for peat. This is the first step in restoring mined bogs back to a functioning ecosystem.
Richness and diversity in Canada
Relative to other groups covered in this report, the species richness of vascular plants is high across the country (Figure 2-1-i, Table 2-1-i), peaking in British Columbia (2133 native species). The flora of British Columbia is particularly diverse within a Canadian context, because 557 species of native vascular plants found there are found nowhere else in Canada. Other regions of Canada known for being local centers of vascular plant species diversity, and for concentrations of endemic plants include the Central Yukon Plateau, Ellesmere and Baffin Islands, the sand-dune region of Lake Athabasca, Saskatchewan, and the Gulf of St Lawrence.
The proportion of Exotic plant species is high across the country, but tends to be highest in the provinces of eastern Canada (22% to 36%) and lowest in the territories (2% to 11%).
Species spotlight - Showy Lady's-slipper, Cypripedium reginae
Known as the 'queen' of the orchids, the Showy Lady's-slipper, Cypripedium reginae, has beautiful pink and white flowers, and grows up to 80 cm tall. Each flower has three petals, the lowest of which is folded into a pouch. This pouch is said to resemble a slipper, giving the lady's-slipper orchids (genus Cypripedium) their name. Showy Lady's-slippers require very nutrient-rich soil and are found in fens and wet, open forests throughout eastern and central Canada.
Like all orchids, Showy Lady's-slippers have an intriguing and complicated life cycle. Orchid seeds are very small, almost microscopic, and do not contain a food source to nourish the germinating seed. In order to survive and grow, the seed must come in to contact with a specific soil fungus, which provides enough nutrients for it to grow into a small plant. Once the plant produces leaves, it can make its own food through photosynthesis. However, it can take up to 12 years for this slow-growing plant to produce flowers! To protect the plant from hungry predators during its long life cycle, the shoots and leaves of Showy Lady's-slippers are covered in stinging hair-like structures. The stinging 'hairs' strongly discourage invertebrates, and larger predators, such as White-tailed Deer (Odocoileus virginianus), from eating the plant.
Showy Lady's-slippers are pollinated by insects, typically small bees or flies. However, unlike many other angiosperms, lady's-slipper orchids do not produce nectar to attract visiting insects. Instead, insects are thought to be attracted to the flower by the colours and patterns of its petals, and by its scent. Once an insect enters the flower, it becomes trapped within the folded lower petal, or slipper. To escape, the insect must push past the pistil (the female part of the flower), where pollen is brushed off the insect's body, to fertilize the eggs. Finally, the insect pushes past the stamen (the male part of the flower), where it picks up more pollen, before leaving the flower.
Due in part to its long, complicated life cycle, this species is particularly vulnerable to increases in rates of adult mortality. For example, harvesting by gardeners and other collectors has led to the loss of entire populations, despite the fact that this species does not grow well in artificial settings. Other concerns include habitat loss, changes in the abundance or distribution of insect pollinators or soil fungi, and trampling of the inconspicuous young shoots by humans attracted by the beauty of the adult plants.
Showy Lady's-slipper is widespread and locally common in much of eastern Canada, and has a Canada rank of Secure. This rank has not changed since Wild Species 2000, although, of the eight provinces in which it is found, two provinces have changed its rank to reflect a lower level of risk and one province has changed its rank to reflect a higher level of risk.
Species spotlight - Carnivorous plants
Carnivorous plants have the fascinating ability to capture and kill insects and other small animals. Carnivorous plants live primarily in nutrient-poor bogs and other habitats with acidic or wet soils. In these habitats, essential nutrients such as nitrogen, are difficult to obtain, so carnivorous plants supplement their nutrient supply by digesting the insects that they capture. Interestingly, the ability to capture and digest prey has evolved separately in several different plant families, so modern carnivorous plants are quite varied in structure and the methods they use for capturing insects.
There are 20 different species of carnivorous vascular plants in Canada, representing four different groups; pitcher plants (genus Sarracenia, one species), sundews (genus Drosera, five species), butterworts (genus Pinguicula, three species) and bladderworts (genus Utricularia, 11 species). Each group has its own unique method of capturing and digesting prey. For example, sundews have modified leaves covered in red, hair-like structures, each topped with a glistening drop of sticky mucus. Insects are attracted by the sundew's colourful appearance and sweet-smelling secretions, but once they step onto a leaf, they quickly become stuck. As the insect struggles, more hairs are drawn inwards to help secure the insect. Glands on the hairs secrete enzymes, which digest the prey, allowing the leaf to absorb the nutrients.
The most complicated active traps developed by carnivorous plants are found in the bladderworts, which capture tiny aquatic prey. Aquatic bladderworts float freely in shallow water, without the benefit of roots to draw nutrients from the soil. Their leaves are very finely divided and contain numerous tiny chambers or bladders. Each bladder operates as a vacuum trap, whose door is triggered by hair-like structures. When a prey item brushes against the 'hairs', the door of the bladder flips open and the prey is sucked into the trap along with the surrounding water. Once inside, the prey is digested and the water is pumped back outside, re-creating the vacuum and leaving the trap ready for the next victim. Amazingly, the door of the bladder trap opens in less than 0.002 seconds, one of the fastest response-times in the plant world!
The majority of Canada's carnivorous plants are ranked Secure or Sensitive, but one species (Yellowish-white Bladderwort, Utricularia ochroleuca) has a Canada rank of May Be At Risk and one species (Thread-leaved Sundew, Drosera filiformis) has a Canada rank of At Risk. Carnivorous plants are an important component of nutrient-poor wetlands across the country. The most important threat to carnivorous plants is habitat destruction through peat mining, wetland drainage and succession, although collecting for the commercial plant trade is also a concern for all species of carnivorous plants.
Species spotlight - Tamarack, Larix laricina
Tamarack (Larix laricina), also known as Hackmatack or Eastern Larch, is found in every province and territory of Canada, and is the official tree of the Northwest Territories. Tamaracks are unusual in the plant world because they are deciduous conifers! Like other conifers, Tamaracks have cones and needle-like leaves, but each autumn their soft, flexible needles turn a beautiful golden colour and fall off, to be replaced again in spring.
Tamaracks grow in a range of soil conditions, but are typically found in cold, poorly drained soils, in bogs and other peatlands. A small to medium sized tree, mature plants are typically 15 to 23 m tall, up to 40 cm in diameter and can live for about 150 to 180 years. Tamaracks are common in the boreal forest and are considered a very cold-hardy tree. In order to survive the cold winter, Tamararks take advantage of a process called extracellular freezing. As water freezes, ice-crystals are formed which can damage cells irreparably. However, during extracellular freezing, water is squeezed out of the tree's cells and stored in the air spaces between the cells. This prevents the cells from being damaged when ice crystals form, allowing Tamaracks to survive as far north as the tree line.
Although Tamaracks are not an important commercial species, they are harvested and sold to make pulp products. The hard, rot-resistant wood is also used to make poles, fence posts and railway ties, while in the past its roots were prized for shipbuilding. Aboriginal Peoples have used Tamarack for many purposes including food, medicine, and construction of canoes and snowshoes. The roots can be used for weaving bags and for sewing bark canoes together. Tamarack branches and twigs arevariety of animals feed on the leaves, cones, seeds or bark of Tamarack trees, such as Sharp-tailed Grouse (Tympanuchus phasianellus), American Black Bear (Ursus americanus), Snowshoe Hare (Lepus americanus), North American Porcupine (Erethizon dorsatum), and Red Squirrel (Tamiasciurus hudsonicus). Major pests of the Tamarack include Larch Sawfly (Pristiphora erichsonii) and Eastern Larch Beetle (Dendroctonus simplex).
Tamarack has a Canada rank of Secure and is also ranked Secure in each of the provinces and territories. Its native cousins, Subalpine Larch (Larix lyallii) and Western Larch (Larix occidentalis), found only in Alberta and British Columbia, also have Canada ranks of Secure.
Species spotlight - Swamp Cottonwood, Populus heterophylla
In 2002, while carrying out a biological survey of Bickford Woods in southern Ontario, researchers were amazed to discover a new species for Canada. This new species is not small or easily overlooked, but is in fact a stand of over 60 mature trees, growing up to 27 m tall! The new species is Swamp Cottonwood, Populus heterophylla, a deciduous tree belonging to the willow family (family Salicaceae) and closely related to the poplars, aspens and other cottonwoods (genus Populus). Swamp Cottonwood occurs fairly commonly in the southeast United States, but is rarer in the northeast United States.
This medium-sized deciduous tree grows up to 40 m in height, in wet soils of swamps and floodplains. Its leaves are large and rounded, and the bark is thick and rough with a reddish colour. As with other poplars, Swamp Cottonwood flowers grow very early in the spring, even before the leaves appear. The flowers grow in the form of dangling catkins and each tree has either male or female flowers, never both. Pollen is carried by the wind from the male flowers to the female flowers, where the eggs are fertilized and seeds begin to develop. The seeds are light with hair-like tufts, so they can be carried by wind or float on water. The Swamp Cottonwood's habitat is often flooded early in the spring, when the seeds are produced. The seeds fall into the water and float until water levels decrease, at which time the seeds are deposited on the wet soil where they can germinate and grow. Swamp Cottonwoods grow best in open areas with little shade, and they are often found along the edges of swamps and rivers. Mature trees occur in low numbers throughout the species' range and are not a major component of any forest-type.
The story of the discovery of the Swamp Cottonwood in Canada reminds us that there are many discoveries still to be made about Canadian wildlife, even in densely populated regions like southern Ontario. Due to its highly restricted Canadian distribution and small population size, Swamp Cottonwood has a Canada rank of May Be At Risk.
Results of assessment
51% of Canada's 5074 species of vascular plants have Canada ranks of Secure (2572 species), while 11% have Canada ranks of May Be At Risk (552 species), 9% have Canada ranks of Sensitive (460 species) and 2% have Canada ranks of At Risk (110 species, Figures 2-1-i and 2-1-ii, Table 2-1-i). Less than 1% of vascular plant species have Canada ranks of Extirpated (22 species), and none have Canada ranks of Extinct. 24% of vascular plant species have a Canada rank of Exotic (1216 species), the highest proportion of Exotic species of any species group covered in this report. Finally, 2% of Canada's vascular plant species have Canada ranks of Undetermined (112 species), and 1% have Canada ranks of Not Assessed (30 species).
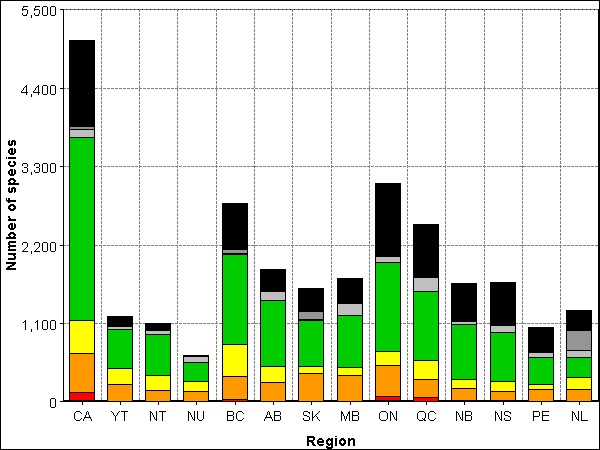
Long description for Figure 2-1-i
Figure 2-1-i illustrates the total number of vascular plant species in Canada and per region, broken down into rank status. In Canada there were 22 extirpated, 110 at risk, 552 may be at risk, 460 sensitive, 2572 secure, 112 undetermined, 30 not assessed, and 1216 exotic species for a total of 5074 vascular plant species. In the Yukon there were 249 species that may be at risk, 227 sensitive, 542 secure, 36 undetermined, 20 not assessed, and 132 exotic for a total of 1206 vascular plant species. In the Northwest Territories there were 167 species that may be at risk, 199 sensitive, 576 secure, 63 undetermined, 3 not assessed, and 95 exotic for a total of 1103 vascular plant species. In Nunavut there were 148 species that may be at risk, 137 sensitive, 269 secure, 86 undetermined, and 13 exotic for a total of 653 vascular plant species. In British Columbia there were 2 extirpated species, 37 at risk, 323 may be at risk, 440 sensitive, 1265 secure, 10 undetermined, 56 not assessed, and 657 exotic for a total of 2790 vascular plant species. In Alberta there was 1 extirpated species, 6 at risk, 268 may be at risk, 220 sensitive, 923 secure, 135 undetermined, 1 not assessed, and 298 exotic for a total of 1852 vascular plant species. In Saskatchewan there were 7 species at risk, 395 may be at risk, 90 sensitive, 649 secure, 16 undetermined, 104 not assessed, and 330 exotic for a total of 1591 vascular plant species. In Manitoba there were 11 species at risk, 364 may be at risk, 110 sensitive, 732 secure, 161 undetermined, and 353 exotic for a total of 1731 vascular plant species. In Ontario there were 22 extirpated species, 55 at risk, 441 may be at risk, 184 sensitive, 1255 secure, 81 undetermined, and 1017 exotic for a total of 3055 vascular plant species. In Quebec there were 11 extirpated species, 48 at risk, 259 may be at risk, 259 sensitive, 974 secure, 191 undetermined, 5 not assessed, and 745 exotic for a total of 2491 vascular plant species. In New Brunswick there were 9 extirpated species, 8 at risk, 168 may be at risk, 126 sensitive, 774 secure, 41 undetermined, 2 not assessed, and 536 exotic for a total of 1664 vascular plant species. In Nova Scotia there were 14 extirpated species, 8 at risk, 127 may be at risk, 136 sensitive, 694 secure, 96 undetermined, and 596 exotic for a total of 1671 vascular plant species. In Prince Edward Island there were 6 extirpated species, 1 at risk, 168 may be at risk, 70 sensitive, 383 secure, 65 undetermined, and 359 exotic for a total of 1052 vascular plant species. In Newfoundland and Labrador there were 4 species at risk, 173 may be at risk, 161 sensitive, 291 secure, 88 undetermined, 280 not assessed, and 284 exotic for a total of 1281 vascular plant species.
| Rank | CA | YT | NT | NU | BC | AB | SK | MB | ON | QC | NB | NS | PE | NL |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Extirpated | 22 | 0 | 0 | 0 | 2 | 1 | 0 | 0 | 22 | 11 | 9 | 14 | 6 | 0 |
| Extinct | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| At risk | 110 | 0 | 0 | 0 | 37 | 6 | 7 | 11 | 55 | 48 | 8 | 8 | 1 | 4 |
| May be at risk | 552 | 249 | 167 | 148 | 323 | 268 | 395 | 364 | 441 | 259 | 168 | 127 | 168 | 173 |
| Sensitive | 460 | 227 | 199 | 137 | 440 | 220 | 90 | 110 | 184 | 259 | 126 | 136 | 70 | 161 |
| Secure | 2572 | 542 | 576 | 269 | 1265 | 923 | 649 | 732 | 1255 | 974 | 774 | 694 | 383 | 291 |
| Undetermined | 112 | 36 | 63 | 86 | 10 | 135 | 16 | 161 | 81 | 191 | 41 | 96 | 65 | 88 |
| Not assessed | 30 | 20 | 3 | 0 | 56 | 1 | 104 | 0 | 0 | 5 | 2 | 0 | 0 | 280 |
| Exotic | 1216 | 132 | 95 | 13 | 657 | 298 | 330 | 353 | 1017 | 745 | 536 | 596 | 359 | 284 |
| Accidental | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Total | 5074 | 1206 | 1103 | 653 | 2790 | 1852 | 1591 | 1731 | 3055 | 2491 | 1664 | 1671 | 1052 | 1281 |
Comparison with Wild Species 2000
Two groups of vascular plants, the ferns and the orchids, were assessed in Wild Species 2000. At that time, there were 122 species of ferns and 78 species of orchids known to occur in Canada. Since 2000, three new fern species have been added to the national list, all belonging to the Moonwort genus (genus Botrychium); Narrow-leaved Moonwort (Botrychium lineare), Tunux Moonwort (Botrychium tunux) and Yaa Xu da Keit Moonwort (Botrychium yaaxudakeit). In addition, California Sword Fern (Polystichum californicum), ranked May Be At Risk in 2000, has been removed from the national list since the single Canadian collection was re-identified to another species, meaning that this species has never been documented in Canada. Therefore, the total number of fern species ranked in 2005 is 124.
No new species of orchids have been added to the national list since 2000, but some taxonomic changes have occurred, resulting in the merging of two sets of species; Dactylorhiza fuchsia with Dactylorhiza majalis, and Platanthera leucostachys with Platanthera dilatata. Therefore the total number of orchids ranked has changed from 78 in 2000, to 76 in 2005. Species that have undergone changes in taxonomy or that have been added to or removed from the species list since 2000 are not included in further discussions of rank changes.
A total of 195 species of ferns and orchids were ranked in both 2000 and 2005. Of these, changes in Canada rank were made for 18 species (9%). Eleven species moved into categories with an increased level of risk (61%), six species moved into categories with a reduced level of risk (33%) and one species moved to the Extirpated category (6%). Changes in Canada rank were due to improved knowledge (67%, 12 changes), process changesFootnote1 (17%, three changes) or detailed status assessments by the Committee on the Status of Endangered Wildlife in Canada (COSEWIC) (17%, three changes) and not to biological changes in species distribution, threats or population (Tables 2-1-ii and 2-1-iii). Changes in Canada ranks had very little effect on the overall proportion of species ranked in each category.
| Number | Canada rank | Number and percentage of species in each rank in 2000 | Number and percentage of species in each rank in Wild Species 2005 | Summary of change | Reasons for change |
|---|---|---|---|---|---|
| 0 | Extinct/Extirpated | 0 | --a | - | |
| 0.2 | Extinct | --a | 0 | - | |
| 0.1 | Extirpated | --a | 1 (1%) | ↑ | Improved knowledgeb |
| 1 | At Risk | 10 (5%) | 13 (7%) | ↑ | COSEWIC assessmentc |
| 2 | May Be At Risk | 28 (14%) | 29 (15%) | ↑ | Improved knowledgeb, Species addedd |
| 3 | Sensitive | 26 (13%) | 25 (13%) | ↓ | Improved knowledgeb, Process changee |
| 4 | Secure | 129 (65%) | 127 (64%) | ↓ | Improved knowledgeb, Process changee Taxonomy changef |
| 5 | Undetermined | 0 | 0 | = | - |
| 6 | Not Assessed | 0 | 0 | = | - |
| 7 | Exotic | 6 (3%) | 5 (3%) | ↓ | Taxonomy changef |
| 8 | Accidental | 1 (1%) | 0 | ↓ | Improved knowledgeb |
Key to symbols: ↑ Number of species in this category has increased. ↓ Number of species in this category has decreased. ↔ An equal number of species have been added and removed from this category; no net change. = No species have been added or removed from this category.
a The single category of Extinct/Extirpated in Wild Species 2000, was replaced with two separate categories in 2005; Extinct and Extirpated. See the Background section for details.
b New information has been collected or brought to light, and used as evidence for a change in rank. A biological change (i.e. a change in species population, distribution or threats) since 2000 is not suggested.
c A formal COSEWIC assessment has been conducted, and used as evidence for a change in rank. A biological change (i.e. a change in species population, distribution or threats) since 2000 is not suggested.
d Three new species have been added to the national list.
e Rank change is due to a change in the process of assigning Canada ranks; the provincial/territorial rank with the lowest level of risk is used as the Canada rank.
f A change in taxonomy has lead to the merging of two species. For details see the 'Comparison with Wild Species 2000' section of the Vascular Plants General Status Summary.
| 2005 Canada rank | 2000 Canada rank | English name | Scientific name | Reason for changea |
|---|---|---|---|---|
| Extirpated | Accidental | Yellow Fringed-orchid | Platanthera ciliaris | I |
| At Risk | May Be At Risk | Lemmon's Holly Fern | Polystichum lemmonii | C |
| At Risk | May Be At Risk | Mountain Holly Fern | Polystichum scopulinum | C |
| At Risk | May Be At Risk | Eastern Prairie Fringed-orchid | Platanthera leucophaea | C |
| May Be At Risk | Sensitive | Mountain Moonwort | Botrychium montanum | I |
| May Be At Risk | Sensitive | Gastony's Cliff-brake | Pellaea gastonyi | I |
| May Be At Risk | Sensitive | Alaska Holly Fern | Polystichum setigerum | I |
| May Be At Risk | Sensitive | Upswept Moonwort | Botrychium ascendens | I |
| May Be At Risk | Sensitive | Stalked Moonwort | Botrychium pedunculosum | I |
| May Be At Risk | Secure | Hairy Water Fern | Marsilea vestita | I |
| Sensitive | May Be At Risk | Large Round-leaved Orchid | Platanthera macrophylla | I |
| Sensitive | May Be At Risk | Yellow Ladies'- tresses | Spiranthes ochroleuca | I |
| Sensitive | Secure | Giant Chain Fern | Woodwardia fimbriata | I |
| Sensitive | Secure | Hillside Rein Orchid | Piperia elegans | I |
| Sensitive | May Be At Risk | Ram's-head Lady'sslipper | Cypripedium arietinum | P |
| Sensitive | May Be At Risk | Bog Adder's-mouth | Malaxis paludosa | P |
| Secure | Sensitive | Nahanni Oak Fern | Gymnocarpium jessoense | I |
| Secure | Sensitive | Large Purple Fringed-orchid | Platanthera grandiflora | P |
a C: change due to new COSEWIC assessment I: change due to improved knowledge of the species. P: change due to procedural change.
Threats to Canadian vascular plants
With such a wide diversity of vascular plant species in Canada, it is not surprising that the threats to vascular plants are similarly varied. As with other species groups, habitat loss and degradation are major problems. Habitat loss occurs when natural habitats are replaced with human landuses such as agriculture or housing, or as a result of natural processes such as succession, fire or flooding. Habitat degradation can occur in many forms, including pollution, changes in drainage patterns, or trampling by humans or animals. Over-harvesting is another threat for some species, particularly for plants that are valued for their beauty (e.g. Showy Lady's-slipper), or for medicinal properties.
In recent years, the impact of Exotic species has become recognized as a serious threat to native wildlife. Exotic plants can compete with native plants for space to grow and for resources. A wellknown example of this is Purple Loosestrife (Lythrum salicaria), which was introduced from Europe in the 1800s and has altered many wetlands from systems of high plant diversity to systems dominated almost entirely by a small number of Exotic species. This change affects many species including the mammals, reptiles, amphibians and invertebrates that rely on wetlands for survival. For example, Muskrats (Ondatra zibethicus) will not eat Purple Loosestrife, and many birds will not nest in it. Exotic species can also introduce new diseases, which can reduce the health of native plants. Another problem is hybridization, in which an Exotic plant interbreeds with a native plant, weakening its gene pool. The native Red Mulberry (Morus rubra), ranked At Risk, has declined partly due to hybridization with the Exotic White Mulberry (Morus alba). Every year, millions of dollars are spent on trying to control Exotic species like Purple Loosestrife, Spotted Knapweed (Centaurea biebersteinii) and European Buckthorn (Rhamnus cathartica) in natural habitats.
Conclusion
This general status assessment of all Canada's 5074 species of vascular plants is an important achievement, involving input from botanists across the country using the most current information to assess the distribution and general status of Canada's vascular plants. This compilation of knowledge will form a platform for further monitoring and research, as well as highlighting areas where knowledge is lacking. The results of this general status assessment indicate that the majority (51%) of vascular plant species in Canada are considered Secure, although 11% of vascular plants have Canada ranks of May Be At Risk and 2% have Canada ranks of At Risk. The results also highlight the large proportion of Exotic species; 24% of Canada's vascular plants are ranked Exotic, a much higher proportion than for any other group covered in this report. Finally, the update of the fern and orchid ranks demonstrates the importance of improved knowledge in increasing our understanding of the status of vascular plants in Canada.
Further information
Adrian, S. 2000. Carnivorous plants. Marston House, England. 240 pp
Agriculture and Agri-food Canada. The vascular plant herbarium. (Accessed February 1, 2006).
Ames, D., Bainard Acheson, P., Heshka, L., Joyce, B., Neufeld, J., Reeves, R., Reimer, E. and Ward, I. 2005. Orchids of Manitoba. Native Orchid Conservation Inc., Canada. 158 pp
Bruce-Grey Plant Committee. 1997. A guide to the orchids of Bruce and Grey counties, Ontario. 2nd edition. Stan Brown Printers, Owen Sound, Ontario. 105 pp
Burchill, C. 2005. Vascular flora of Manitoba. (Accessed February 9, 2005).
Canadian Botanical Association. (Accessed February 1, 2006).
Cody, W. J. 2000. Flora of the Yukon Territory. National Research Press, Ottawa. 669 pp
Davis, S. D., Heywood, V. H., Herrera-MacBryde, O., Villa-Lobos, J. and Hamilton, A. (eds.). 1997. Centres of plant diversity: A guide and strategy for their conservation. Volume 3: The Americas. IUCN Publications Unit, Cambridge, England. (Accessed March 2, 2006)
E-Flora BC. (Accessed February 1, 2006).
Eastman, J. 1992. The book of forest and thicket. Trees shrubs and wildflowers of eastern North America. Stackpole Books, Harrisburg, Pennsylvania, USA. 212 pp
Farrar, J. L. 1995. Trees in Canada. Fitzhenry & Whiteside, Ontario and Natural Resources Canada, Ottawa. 502 pp
Flora of North America. (Accessed February 1, 2006).
Foss, K. 2004. Our lady of the bog. Canadian Geographic Jul/Aug 2004. (Accessed March 14, 2006).
Henry, J. D. 2002. Canada's boreal forest. Smithsonian natural history series. Smithsonian Institute Press. 176 pp
Hinds, H. R. 2000. Flora of New Brunswick: A manual for the identification of the vascular plants of New Brunswick. University of New Brunswick, Fredericton. 699 pp
Johnston, W. F. 1990. Tamarack. In Burns, R. M., and Honkala, B. H. (technical coordinators). 1990. Silvics of North America: 1. Conifers; Agriculture Handbook 654. U.S. Department of Agriculture, Forest Service, Washington, DC. vol. 2, 877 pp (Accessed December 22, 2005).
L'Union des prodcuteurs agricoles. Gaspé Peninsula's Non-Timber Forest Products Fact Sheets. Common Yarrow. (Accessed April 5, 2006).
Library and Archives Canada. 2001. Susanna Moodie and Catherine Parr Traill. (Accessed April 5, 2006).
Maunder, J. E. 2001. A digital flora of Newfoundland and Labrador vascular plants. (Accessed February 1, 2006).
McMaster, R. T. 2003. Populus heterophylla L. Swamp cottonwood. Conservation and research plan for New England. New England Wild Flower Society, Massachusetts. (Accessed January 10, 2006).
Morris, A. 2003. New tree for Carolinian Canada. Carolinian Canada Newsletter. Winter 2003-4. (Accessed January 10, 2006).
National herbarium of Canada. (Accessed February 1, 2006).
PlantWatch. (Accessed February 1, 2006).
Prindle, T. 2000. NativeTech: Native American technology and art. An introduction to tamarack trees & traditions. (Accessed December 22, 2005).
Rice, B. A. 2004. Carnivorous Plant FAQ v10.0. (Accessed February 1, 2006).
Roland, A. E. 1998. The flora of Nova Scotia. Nimbus, Halifax. 1350 pp
Scoggan, H. J. 1978. The flora of Canada. National Museum of Natural Sciences, National Museum of Canada, Publications in Botany 7(1-4)
The Nature Trust of New Brunswick. Rare flora of the Upper St. John River. (Accessed April 5, 2006).
Victorin, M. and Rouleau, E. 1964. Flore Laurentienne, 2nd ed. University of Montreal Press, Montreal. 925 pp (Accessed March 13, 2006).
Waldron, G., Ambrose, J. and Rodger, L. 2003. Swamp cottonwood (Populus heterophylla), another new tree for Canada. Ontario Natural Heritage Information Centre Newsletter 8(1):6. (Accessed January 10, 2006).
White, D. J., Haber, E. and Keddy, C. 1993. Invasive plants of natural habitats in Canada: an integrated review of wetland and upland species and legislation governing their control. Canadian Wildlife Service, Ottawa, Canada. 121 p (Accessed April 5, 2006).
References
Allaby, M. 1989. Dictionary of the environment. New York University Press, New York. 423 pp
Nature Trust of New Brunswick and Atlantic Canada Conservation Data Centre. 2003. Rare plant survey of the Upper St. John River with focus on Furbish's Lousewort. The Nature Trust of New Brunswick, INC. Fredericton, NB. 61 pp.
Smith, D. 2001. Documenting plant domestication: The consilience of biological and archaeological approaches. PNAS 98(4):1324-1326
Vasseur, L. and Potvin, C. 1997. Natural pasture community response to enriched carbon dioxide atmosphere. Plant Ecology 135:31-41