Wild Species 2005: chapter 8
Amphibians
Amphibia - The class of vertebrate chordates that contains the frogs, toads, newts and salamanders. The amphibians evolved in the Devonian period (about 370 million years ago) as the first vertebrates to occupy the land, and many of their characteristics are adaptations to terrestrial life. - Oxford Dictionary of Biology.

Quick facts
- There are roughly 5700 species of amphibians worldwide, 46 of which are found in Canada.
- More than two-thirds (65%) of amphibian species have Canada General Status Ranks (Canada ranks) of Secure, but 20% have Canada ranks of At Risk and 15% have Canada ranks of Sensitive. No amphibians have Canada ranks of May Be At Risk since all the species ranked May Be At Risk in Wild Species 2000 have since been the subject of detailed status assessments by the Committee on the Status of Wildlife in Canada (COSEWIC).
- Since Wild Species 2000, the Rocky Mountain Tailed Frog has been declared a separate species from the Coast Tailed Frog, increasing the total number of amphibian species in Canada to 46.
- Compared to Wild Species 2000, the Canada ranks of 77% of amphibian species remain the same, 11% of amphibian species have been moved to a Canada rank with an increased level of risk, and 11% of amphibian species have been moved to a Canada rank with a reduced level of risk. Most of the changes were due to COSEWIC assessments (80%). None of the changes were due to biological changes in species abundance, distribution or threats.
- On a global scale, many amphibian species are at a high level of risk of extinction; the recent Global Amphibian Assessment ranked nearly one-third (32%) of the world's amphibians as Threatened, compared with 23% of all mammal species and 12% of all bird species.
- The Wood Frog has the most northerly distribution of any North American amphibian, and is the only North American amphibian found north of the Arctic Circle.
Background
Canadian amphibians include frogs, toads, newts and salamanders. These cold-blooded vertebrates can be recognized by their soft, moist skin, without scales, feathers or fur. Many amphibians spend the first part of their life cycle as aquatic, gill-breathing larvae (also known as tadpoles) before they metamorphose into terrestrial, air-breathing adults. This dual life cycle allowed ancestral amphibians to be the first vertebrates to inhabit dry land more than 300 million years ago, giving rise to the modern amphibians, reptiles, birds and mammals.
Many people are familiar with the typical life cycle of frogs and toads, in which an aquatic larva with gills metamorphoses into a terrestrial air-breathing adult. However, in the process of adapting to a wide range of habitats, amphibians have developed a variety of different life cycles, ranging from completely aquatic (e.g. Mudpuppy, Necturus maculosus), to completely terrestrial. For example, the Northern Red-backed Salamander (Plethodon cinereus) lays its eggs on land and guards them until they hatch into juveniles, which look and behave much like the adults. Newts, such as the Roughskin Newt (Taricha granulosa) of British Columbia, have an additional stage in their life cycle, known as the eft. Aquatic larvae with gills metamorphose into terrestrial air-breathing efts, which live up to four years in moist terrestrial habitats. Efts must then metamorphose into amphibious adults to breed and complete the life cycle. The amazing diversity of life cycles displayed by amphibians is not matched in any other group of vertebrates.
Unlike reptiles, birds and mammals, adult amphibians do not have waterproof skin. This is advantageous for amphibians because it allows them to breathe through their skin as well as through their lungs, but it makes amphibians prone to dehydration. So how do amphibians survive on dry land? Many amphibians have special skin on their underside through which they can absorb moisture. This allows them to re-hydrate simply by sitting on moist soil or in a small puddle. To reduce water loss, many amphibians are nocturnal. During the day they remain under logs and rocks. At night, when the air is cooler and less evaporation occurs, they emerge to hunt for food or mates. These physical and behavioural adaptations allow amphibians to survive away from the water, where they can take advantage of many different habitats and food sources.
Like reptiles, amphibians are cold-blooded (ectothermic), meaning they rely on external heat sources (like the sun) to warm their body, rather than producing heat from food energy, like birds and mammals. However, amphibians can survive much further north than reptiles. The distribution of amphibians in northern habitats is largely related to winter temperature and the ability of individual species to tolerate cold. The champion of cold-tolerant amphibians is the Wood Frog (Rana sylvatica), the only North American amphibian or reptile found north of the Arctic Circle. Wood Frogs survive cold temperatures by hibernating frozen underground for several months of the year. Normally cells rupture and die when they are frozen, but Wood Frogs produce a special 'anti-freeze' chemical called a cryoprotectant that protects their cells when frozen solid! Cryoprotectants are of great interest to scientists, who have studied Wood Frogs to develop new methods of freezing mammalian organs, so they can be stored before transplantation.
Status of knowledge
People have been studying amphibians for centuries, so the basic biology, physiology and developmental biology of many species, particularly the frogs, is well known. The natural history of most amphibians in Canada is also generally well understood, but the distribution, population size and population structure of amphibians in some regions is not well known. This is partly due to the difficulties in monitoring amphibians which can include their nocturnal and secretive behaviours, their small size and their cryptic appearance. Initiatives such as 'Frogwatch', a program that uses volunteers to monitor amphibian populations across the country, are providing data which will increase our understanding of amphibian distributions, and provide baseline data to monitor population changes.
Genetic tools are becoming increasingly important in amphibian research. For example, in 1997, genetic analysis was used to distinguish the Oregon Spotted Frog (Rana pretiosa) as a separate species from the Columbia Spotted Frog (Rana luteiventris). Genetic tools have also been used to study Bullfrog (Rana catesbeiana) dispersal in Ontario, the impact of clear-cutting on the Coastal Giant Salamander (Dicamptodon tenebrosus) in British Columbia and the evolution of new species of salamanders (speciation) in the Rocky Mountains.
In recent years, the impacts of environmental contaminants on the growth and development of amphibians has been studied across Canada. Chemicals and fertilizers, which collect in some aquatic habitats used by amphibians, can cause a range of negative effects including deformities, reduced immune system activity, abnormal behaviours and, in extreme cases, death. However, it is difficult to link these impacts with population declines.
Richness and diversity in Canada
Canada has 46 species of amphibians including one mudpuppy, two newts, seven toads, 18 frogs and 18 salamanders. The most species rich provinces are Ontario (25 species), British Columbia (22 species) and Quebec (21 species) (Figure 2-7-i, Table 2-7-i). British Columbia has the most species found nowhere else in Canada (13 species). All the amphibian species found in Canada are also found in the USA, but several species including the Canadian Toad (Bufo hemiophrys) and the Mink Frog (Rana septentrionalis), have the majority of their range in Canada.
Species spotlight - Northern Leopard Frog
Northern Leopard Frogs, Rana pipiens, (Canada General Status Rank (Canada rank): Secure) are found in every province and territory except the Yukon. This medium-sized frog breeds in shallow, warm ponds and produces egg masses of 600 to 7000 eggs. Eggs hatch into tadpoles, which graze on algae for about 9 to 12 weeks, until they are ready to metamorphose into adults. Adults spend the summer feeding away from the water, but return to deep, well-oxygenated water to hibernate.
Northern Leopard Frogs were once common throughout their Canadian range, but during the late 1970s they underwent rapid, widespread population declines in British Columbia, Alberta, Saskatchewan, and Manitoba. In fact, Northern Leopard Frogs had virtually disappeared from Manitoba by 1976 and from Alberta by 1979. Lack of monitoring before this period makes population trends difficult to interpret, and scientists are still uncertain of the reason for the declines. Since the 1980s, Northern Leopard Frog populations in Alberta and Saskatchewan have been recovering slowly, whereas Manitoba's populations have recovered relatively quickly. In British Columbia, populations have not substantially recovered and are now restricted to a single Wildlife Management Area.
The story of the Northern Leopard Frog demonstrates that even widespread, numerous species are vulnerable to catastrophic population declines and local extirpation. Scientists are now focussing on captive breeding and release in Alberta and British Columbia and population monitoring in Alberta and Saskatchewan to attempt to restore this species to its former range and to improve our knowledge of the Northern Leopard Frog.
Species spotlight - Oregon Spotted Frog
The Oregon Spotted Frog, Rana pretiosa, was described as a distinct species, separate from the Columbia Spotted Frog, in 1997. In the same year, the Oregon Spotted Frog was the first species to be given an emergency listing of Endangered by the Committee on the Status of Endangered Wildlife in Canada (COSEWIC). This small frog was once distributed from southwest British Columbia to northwest California, but is now restricted to small, isolated populations and is estimated to have been lost from more than 90% of its historic range. Population declines and range reduction have been linked to habitat loss, changes in hydrology, introduction of exotic predators and vegetation, and isolation of remaining populations. In addition, they are vulnerable to pollution and climate change. Now known from only three populations in southwest British Columbia and less than 30 populations in the United States, this species has a Canada rank of At Risk, and an IUCN Red List rank of Vulnerable, meaning it is considered vulnerable to extinction on a global scale.
Since the emergency designation by COSEWIC, work has begun on a recovery plan for the Oregon Spotted Frog with the co-operation of government agencies, universities, local native groups and the public. Captive breeding, habitat mapping and habitat remediation have already begun. Although the three small remaining Canadian populations are isolated from each other and from populations in the United States, the development of the recovery plan and the cooperation between different agencies and groups gives hope that this species can be preserved into the future.
Species spotlight - Western Toad
The Western Toad, Bufo boreas, is the only toad found in the Yukon, and it is also found in the Northwest Territories, British Columbia and Alberta. This large toad breeds in the shallow margins of ponds, streams and lakes. Females can produce clutches of up to 15 000 eggs, but may breed only once in their lifetime. Adult toads frequently wander long distances from water and are usually nocturnal, especially at low elevations. In the winter, Western Toads hibernate in animal burrows or under loose debris. Adult Western Toads are carnivorous and eat a wide range of invertebrates including earthworms, beetles, spiders and ants. Despite their ability to release a mild poison, Western Toads are preyed on by reptiles, mammals and birds.
Due to a COSEWIC status assessment (Special Concern, 2002), the Canada rank of the Western Toad has changed from Secure in Wild Species 2000 to Sensitive in this report. The COSEWIC status assessment found that populations within the Georgia Basin of south-costal British Columbia are of special concern, due to evidence of population declines and at least one example of a local extirpation. The rest of the Canadian population was considered 'likely not at risk'. Canadian populations of Western Toads are not only a unique component of the fauna of western Canada, they are also important to the global survival of this species, due to declining populations in the United States. Careful monitoring and research are needed to help maintain healthy Canadian populations of Western Toads.
Results of general status assessmentFooetnote1
Of the 46 species of amphibians found in Canada, nine species have Canada ranks of At Risk (20% of all species), including two toads, three frogs, and four salamanders (Figures 2-7-i and 2- 7-ii, Table 2-7-i). Within Canada, all nine species with Canada ranks of At Risk have fairly restricted ranges; none are found in the territories or in more than one province.
Seven species of amphibian have Canada ranks of Sensitive (15%) and 30 species have Canada ranks of Secure (65%). Canada has no Exotic or Accidental amphibian species and no species have Canada ranks of May Be At Risk, Undetermined or Not Assessed.
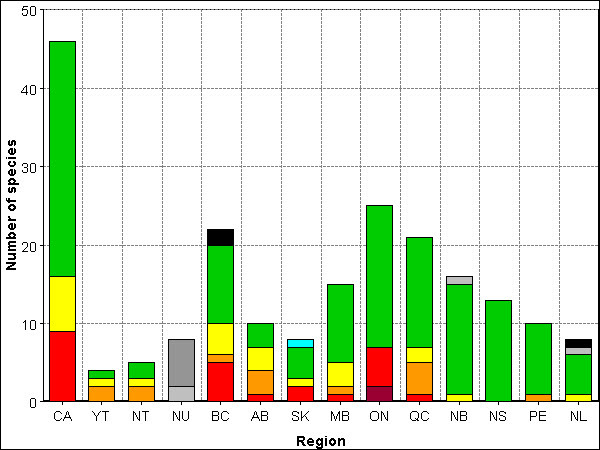
Long description for Figure 2-7-i
Figure 2-7-i illustrates the total number of amphibian species in Canada and per region, broken down into status rank. In Canada there were 9 species at risk, 7 sensitive, and 30 secure for a total of 46 amphibian species. In the Yukon there were 2 species ranked may be at risk, 1 sensitive, and 1 secure, for a total of 4 species. In the Northwest Territories there were 2 species ranked may be at risk, 1 sensitive, and 2 secure for a total of 5 species. In Nunavut there were 2 species that were ranked undetermined and 6 not assessed for a total of 8 species. In British Columbia there were 5 species ranked at risk, 1 may be at risk, 4 sensitive, 10 secure, and 2 exotic for a total of 22 species. In Alberta there was 1 species that was ranked at risk, 3 may be at risk, 3 sensitive, and 3 secure for a total of 10 species. In Saskatchewan there were 2 species that were ranked at risk, 1 sensitive, 4 secure and 1 accidental for a total of 8 species. In Manitoba there was 1 species ranked at risk, 1 may be at risk, 3 sensitive, and 10 secure for a total of 15 species. In Ontario there were 2 species that were ranked extirpated, 5 at risk, and 18 secure for a total of 25 species. In Quebec there was 1 species ranked at risk, 4 may be at risk, 2 sensitive, and 14 secure for a total of 21 species. In New Brunswick there was 1 species that was ranked sensitive, 14 secure, and 1 undetermined for a total of 16 species. In Nova Scotia there were 13 species that were ranked secure for a total of 13 species. In Prince Edward Island there was 1 species that was ranked may be at risk and 9 secure for a total of 10 species. In Newfoundland and Labrador there was 1 species that was ranked sensitive, 5 were secure, 1 undetermined, and 1 exotic for a total of 8 species.
| Rank | CA | YT | NT | NU | BC | AB | SK | MB | ON | QC | NB | NS | PE | NL |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Extirpated | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 2 | 0 | 0 | 0 | 0 | 0 |
| Extinct | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| At risk | 9 | 0 | 0 | 0 | 5 | 1 | 2 | 1 | 5 | 1 | 0 | 0 | 0 | 0 |
| May be at risk | 0 | 2 | 2 | 0 | 1 | 3 | 0 | 1 | 0 | 4 | 0 | 0 | 1 | 0 |
| Sensitive | 7 | 1 | 1 | 0 | 4 | 3 | 1 | 3 | 0 | 2 | 1 | 0 | 0 | 1 |
| Secure | 30 | 1 | 2 | 0 | 10 | 3 | 4 | 10 | 18 | 14 | 14 | 13 | 9 | 5 |
| Undetermined | 0 | 0 | 0 | 2 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 1 |
| Not assessed | 0 | 0 | 0 | 6 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Exotic | 0 | 0 | 0 | 0 | 2 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 |
| Accidental | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Total | 46 | 4 | 5 | 8 | 22 | 10 | 8 | 15 | 25 | 21 | 16 | 13 | 10 | 8 |
| Number | Canada rank | Number and percentage of species in each in Wild Species 2000 | Number and percentage of species in each rank in Wild Species 2005 | Summary of change | Reason(s) for change(s) |
|---|---|---|---|---|---|
| 0 | 0 | 0 | - a | - | - |
| 0.2 | Extinct | - | - | ||
| 0.1 | Extirpated | - | - | ||
| 1 | At Risk | 4 (9%) | 9 (20%) | ↑ | COSEWIC assessmentb, Change in taxonomyc |
| 2 | May be at risk | 6 (13%) | 0 | COSEWIC assessmentb, Improved knowledged | |
| 3 | Sensitive | 6 (13%) | 7 (15%) | ↑ | COSEWIC assessmentb, Improved knowledged, Change in taxonomyc |
| 4 | Secure | 29 (64%) | 30 (65%) | ↑ | COSEWIC assessmentb, Improved knowledged, Errore |
| 5 | Undetermined | 0 | 0 | = | - |
| 6 | Not Assessed | 0 | 0 | = | - |
| 7 | Exotic | 0 | 0 | = | - |
| 8 | Accidental | 0 | 0 | = | - |
a The single category of Extinct/extirpated in Wild Species 2000, was replaced with two separate categories in 2005; Extinct and Extirpated. See the Background section for details.
b A formal COSEWIC assessment has been conducted, and used as evidence for a change in rank. A biological change (i.e. a change in species population, distribution or threats) since 2000
c The Rocky Mountain Tailed Frog (Ascaphus montanus), ranked At Risk was split from the Coast Tailed Frog (Ascaphus truei) after the completion of the Wild Species 2000 ranks.
d New information has been collected or brought to light, and used as evidence for a change in rank. A biological change (i.e. a change in species population, distribution or threats) since 2000 is not suggested.
e An error was detected in one of the Wild Species 2000 ranks, after its publication; the national
Key to symbols: ↑Number of species in this category has increased. ↓Number of species in this category has decreased. ↔ An equal number of species have been added and removed from this category; no net change. = No species have been added or removed from this category.
| 2005 Canada rank | 2000 Canada rank | English name | Scientific name | Reason for changea |
|---|---|---|---|---|
| At Risk | May be at risk | Allegheny Mountain Dusky Salamander | Desmognathus ochrophaeus | C |
| At Risk | May be at risk | Coastal Giant Salamander | Dicamptodon tenebrosus | C |
| At Risk | May be at risk | Jefferson Salamander | Ambystoma jeffersonianum | C |
| At Risk | Sensitive | Great Basin Spadefoot | Spea intermontana | C |
| Sensitive | May be at risk | Great Plains Toad | Bufo cognatus | C |
| Sensitive | May be at risk | Spring Salamander | Gyrinophilus porphyriticus | C |
| Sensitive | May be at risk | Coeur d`Alene Salamander | Plethodon idahoensis | I |
| Sensitive | Secure | Western Toad | Bufo boreas | C |
| Secure | Sensitive | Plains Spadefoot | Spea bombifrons | C |
| Secure | Sensitive | Canadian Toad | Bufo hemiophrys | C/I/E |
a C: change due to new COSEWIC assessment. I: change due to improved knowledge of the species. E: change partially due to error in the 2000 ranks.
In Wild Species 2000, species assessment results were presented as the proportion of resident species ('resident species' excludes species with Canada ranks of Extirpated, Extinct and Accidental). In this report, we have used the more straightforward method of presenting results as a proportion of total species richness. Therefore, proportions given in the 'Results of assessment' sub-sections can not be directly compared to those given in the text of Wild Species 2000. To compare results for amphibians directly between the text of Wild Species 2000 and this report please use the following figures, which represent the 2000 results as a proportion of total species richness: Total species richness: 45 species, At Risk: 9%, May Be At Risk: 13%, Sensitive: 13%, Secure: 64%.
Comparison with Wild Species 2000
In late 2000, the Rocky Mountain Tailed Frog (Ascaphus montanus) was described as a separate species from the Coast Tailed Frog (Ascaphus truei), so there are now 46 species of amphibians in Canada, compared to 45 listed in Wild Species 2000. Both species are excluded from further comparisons with Wild Species 2000.
Since Wild Species 2000, the Canada ranks of 10 species (23%) have been changed (Figure 2-7- iii, Table 2-7-ii and 2-7-iii); half of the changes placed species in ranks with a higher level of risk and half placed species in ranks with a lower level of risk than their original rank. The remaining 34 species (77%) retained the Canada rank they were given in Wild Species 2000. The changes have led to an increase in the percentage of species with Canada ranks of At Risk and a reduction in the percentage of species with Canada ranks of May Be At Risk, while the percentage of species with Canada ranks of Sensitive and Secure has remained similar between 2000 and 2005 (Table 2-7-ii).
Changes in Canada rank were due to COSEWIC status assessments (80%), improvements in knowledge (10%) and a combination of a formal COSEWIC assessment, improvement in knowledge and an error detected in the 2000 report following its publication (10%). Most of the changes (60%) were due to reclassification of species previously ranked May Be At Risk to At Risk or Sensitive, reflecting improved knowledge rather than biological changes in species' population, distribution or threats since 2000 (Figure 2-7-iii, Table 2-7-iii). Therefore, the large increase in species with Canada ranks of At Risk (Table 2-7-ii), does not reflect a true decline in overall amphibian status since 2000. Rather, by reclassifying species previously ranked May Be At Risk, the reassessment simply presents a clearer picture of the true status of amphibians in Canada.
Threats to Canadian amphibians
Global amphibian declines over the last 20 years, have spurred considerable discussion of threats to amphibians. Major threats include habitat loss and degradation, introduction of exotic species, over-harvesting (for commercial and recreational use), increases in UV radiation, pollution, disease and climate change. In addition, road mortality is also a threat to some amphibian populations.
Habitat loss is one of the leading threats to amphibians in Canada. In parts of southern Canada, 90% of wetlands have been drained or otherwise destroyed. Remaining wetlands within agricultural or urban landscapes may be polluted and often retain a reduced abundance and diversity of amphibians. In addition, fragmentation of remaining habitat can reduce or prevent the movement of individuals between populations, leading to reduced population stability and reduced flow of genes between populations.
Fungal and viral diseases have been implicated in some global amphibian declines, even in pristine habitats. Research is showing that disease acts on populations in combination with other stresses. For example, incidence of disease may be increased in populations stressed by other factors such as pollutants or increased UV-B radiation.
Conclusion
This reassessment of Canada's amphibians resulted in an increase in the percentage of amphibians with a Canada rank of At Risk, compared to Wild Species 2000. However, this change results not from biological changes in species abundance, distribution or threats, but largely from new COSEWIC assessments and improvements in our knowledge of Canadian amphibians. The majority of changes reclassified species from May Be At Risk to At Risk or Sensitive, reflecting the emphasis on improved knowledge rather than biological changes in species status. Therefore, despite the increased proportion of species with a Canada rank of At Risk, this report does not reflect a true, biological decline in overall amphibian status since 2000, but instead simply presents a more accurate picture of the true status of Canada's amphibians than was available in 2000.
Further information
AmphibiaWeb: Information on amphibian biology and conservation. (Accessed September 23, 2005).
Canadian Amphibian and Reptile Conservation Network. (Accessed February 10, 2006).
Conant, R. and Collins, J. T. 1998. A field guide to reptiles and amphibians of eastern and central North America (3rd Ed.). Houghton Mifflin Co., Boston. 616 pp
EMAN. 2004. Status of amphibian and reptile populations in Canada. (Accessed September 23, 2005).
Frogwatch. (Accessed September 23, 2005).
Froom, B. 1982. Amphibians of Canada. McClelland and Stewart, Toronto, Ontario. 120 pp
IUCN, Conservation International, and NatureServe. 2004. Global amphibian assessment. Amphibian Red List Authority (Accessed 15 October 2005).
The Tree of Life. 1995. Living amphibians. (Accessed 15 October 2005).
Stebbins, R. C. and Cohen, N. W. 1995. A natural history of amphibians. Princeton University Press, Princeton, New Jersey. 316 pp
Virtual exhibit on Canada's biodiversity: focus amphibians. (Accessed 15 October 2005).
References
COSEWIC. 2000. COSEWIC assessment and update status report on the northern leopard frog Rana pipiens (Southern Mountain and Prairie populations) in Canada. Committee on the Status of Endangered Wildlife in Canada, Ottawa. vi + 40 pp
COSEWIC. 2000. COSEWIC assessment and status report on the Oregon spotted frog Rana pretiosa in Canada. Committee on the Status of Endangered Wildlife in Canada, Ottawa. vi + 22 pp
COSEWIC. 2002. COSEWIC assessment and status report on the western toad Bufo boreas in Canada. Committee on the Status of Endangered Wildlife in Canada, Ottawa. vi + 31 pp
Fahrig, L., Pedlar, J. H., Pope, S. E., Taylor, P. D. and Wegner, J. F. 1995. Effect of road traffic on amphibian density. Biological Conservation 73(3):177-182
Green, D. M. (Ed) 1997. Amphibians in decline, Canadian studies in a global problem. Herpetological Conservation 1:1-338
Gibbons, J. W., Scott, D. E., Ryan, T. J., Buhlmann, K. A., Tuberville, T. D., Metts, B. S., Greene, J. L., Mills, T., Leiden, Y., Poppy, S. and Winne, C. T. 2000. The global decline of reptiles, déjà vu amphibians. BioScience 50(8): 653-666
Hine, R. S. and Martin, E. Eds. 2004. The dictionary of biology. Oxford University Press, Oxford, England. 698 pp
Oldham, M. J. 1996. Amphibians and reptiles. In Assessment of species diversity in the mixedwood plains ecozone. Edited by I. M. Smith, Ecological Monitoring and Assessment Network, Environment Canada. (Accessed 15 October 2005).
Ouellet, M., Bonin, J., Rodrigue, J., DesGranges, J. and Lair., S. 1997. Hindlimb deformities (ectromelia, ectrodactyly) in free-living anurans from agricultural habitats. Journal of Wildlife Diseases 33:95-104
Seburn, D. and Seburn, C. 2000. Conservation priorities for the amphibians and reptiles of Canada. World Wildlife Fund Canada and the Canadian Amphibian and Reptile Conservation Network. Toronto. 92 pp (Accessed 15 October 2005).
Storfer, A. 2003. Amphibian declines: future directions. Diversity and Distributions 9:151-163