Little brown myotis, northern myotis and tri-coloured bat: proposed recovery strategy 2015
Species at Risk Act
Recovery Strategy Series

Left Photo: © Jordi Segers, 2015; Center Photo: © Hugh Broders, 2015; Right Photo: © Hugh Broders, 2015;
Table of Contents
- Document Information
- Preface
- Acknowledgments
- Executive Summary
- Recovery Feasibility Summary
- 1. COSEWIC Species Assessment Information
- 2. Species Status Information
- 3. Species Information
- 4. Threats
- 5. Population and Distribution Objectives
- 6. Broad Strategies and General Approaches to Meet Objectives
- 7. Critical Habitat
- 8. Measuring Progress
- 9. Statement on Action Plans
- 10. References
- Appendix A: Effects on the Environment and Other Species
- Appendix B: Additional Research Needs Related to Known and Suspected Threats

Recommended citation:
Environment Canada. 2015. Recovery Strategy for Little Brown Myotis (Myotis lucifugus), Northern Myotis (Myotis septentrionalis), and Tri-colored Bat (Perimyotis subflavus) in Canada [Proposed]. Species at Risk Act Recovery Strategy Series. Environment Canada, Ottawa. ix + 110 pp.
For copies of the recovery strategy, or for additional information on species at risk, including on the Status of Endangered Wildlife in Canada (COSEWIC) Status Reports, residence descriptions, action plans, and other related recovery documents, please visit the Species at Risk (SAR) Public Registry.
Cover illustrations: Left: Northern Myotis, © Jordi Segers; Center: Little Brown Myotis, © Hugh Broders; Right: Tri-colored Bat © Hugh Broders
Également disponible en français sous le titre
« Programme de rétablissement de la petite chauve souris brune (Myotis lucifugus), de la chauve souris nordique (Myotis septentrionalis) et de la pipistrelle de l’Est (Perimyotis subflavus) au Canada [Proposition] »
Content (excluding the illustrations) may be used without permission, with appropriate credit to the source.
Preface
The federal, provincial, and territorial government signatories under the Accord for the Protection of Species at Risk (1996) agreed to establish complementary legislation and programs that provide for effective protection of species at risk throughout Canada. Under the Species at Risk Act (S.C. 2002, c.29) (SARA), the federal competent ministers are responsible for the preparation of recovery strategies for listed Extirpated, Endangered, and Threatened species and are required to report on progress within five years after the publication of the final document on the SAR Public Registry.
The Minister of the Environment and Minister responsible for Parks Canada Agency is the competent minister under SARA for the Little Brown Myotis, Northern Myotis, and Tri-colored Bat and has prepared this recovery strategy, as per section 37 of SARA. To the extent possible, it has been prepared in cooperation with the provinces of British Columbia, Alberta, Saskatchewan, Manitoba, Ontario, Quebec, New Brunswick, Prince Edward Island, Nova Scotia, and Newfoundland and Labrador, as well as the territories of Yukon and Northwest Territories, and the Sahtu Renewable Resources Board, and Wek’èezhìi Renewable Resources Board.
Success in the recovery of these species depends on the commitment and cooperation of many different constituencies that will be involved in implementing the directions set out in this strategy and will not be achieved by Environment Canada and the Parks Canada Agency, or any other jurisdiction alone. All Canadians are invited to join in supporting and implementing this strategy for the benefit of the Little Brown Myotis, Northern Myotis, and Tri-colored Bat and Canadian society as a whole.
This recovery strategy will be followed by one or more action plans that will provide information on recovery measures to be taken by Environment Canada, the Parks Canada Agency, and other jurisdictions and/or organizations involved in the conservation of the species. Implementation of this strategy is subject to appropriations, priorities, and budgetary constraints of the participating jurisdictions and organizations.
The recovery strategy sets the strategic direction to arrest or reverse the decline of the species, including identification of critical habitat to the extent possible. It provides all Canadians with information to help take action on species conservation. When the recovery strategy identifies critical habitat, there may be future regulatory implications, depending on where the critical habitat is identified. SARA requires that critical habitat identified within federal protected areas be described in the Canada Gazette, after which prohibitions against its destruction will apply. For critical habitat located on federal lands outside of federal protected areas, the Minister of the Environment must either make a statement on existing legal protection or make an order so that the prohibition against destruction of critical habitat applies. For critical habitat located on non-federal lands, if the Minister of the Environment forms the opinion that any portion of critical habitat is not protected by provisions in or measures under SARA or other Acts of Parliament, and not effectively protected by the laws of the province or territory, SARA requires that the Minister recommend that the Governor in Council make an order to extend the prohibition against destruction of critical habitat to that portion. The discretion to protect critical habitat on non-federal lands that is not otherwise protected rests with the Governor in Council.
Acknowledgments
This recovery strategy was prepared by Krista Baker, David Andrews, Kathy St. Laurent (Environment Canada-Canadian Wildlife Service (EC-CWS) – Atlantic Region), Ryan Zimmerling, and Sybil Feinman (EC-CWS – National Capital Region).
Andrew Boyne (EC-CWS – Atlantic Region), Robert McLean, Manon Dubé, Carolyn Seburn, Veronica Aponte, Marc-André Cyr (EC-CWS – National Capital Region), Diana Ghikas, Donna Bigelow, Lynne Burns, Amy Ganton, Dawn Andrews (EC-CWS – Prairie Northern Region), Syd Cannings, Tanya Luszcz, Megan Harrison (EC-CWS – Pacfic Yukon Region), Sylvain Giguère, Pierre-André Bernier (EC-CWS – Quebec Region), Krista Holmes (EC-CWS – Ontario Region), Hugh Broders (Saint Mary`s University), Craig Willis (University of Winnipeg), Jordi Segers (Canadian Wildlife Health Cooperative), Derek Morningstar (Myotistar / Golder Associates), Juliet Craig (British Columbia Community Bat Project Network), Laura Kaupas (University of Calgary), Thomas Jung (Yukon Department of Environment), Joanna Wilson, Ashley McLaren, Stephanie Behrens (Government of Northwest Territories), Purnima Govindarajulu (British Columbia Ministry of Environment), Lisa Wilkinson (Alberta Environment and Parks), Daphne Murphy (Saskatchewan Ministry of Environment), Jay Fitzsimmons, Lesley Hale, Chris Risley, Brian Naylor, Jim Saunders, Vivian Brownell, Katie Paroschy, Sue Russell, Aileen Wheeldon, Todd Copeland, Peter Davis, Chuck McCridden, Megan Rasmussen, Valerie Vaillancourt, Laura Darbey, Bill Greaves, Christopher Martin, April Mitchell (Ontario Ministry of Natural Resources and Forestry), Nathalie Desrosiers, Ariane Masse, Anouk Simard, Isabelle Gauthier (Ministère des Fôrets, de la Faune et des Parcs [Quebec]), Mary Sabine, Maureen Toner (New Brunswick Department of Natural Resources), Garry Gregory (Prince Edward Island Department of Communities, Land, and Environment), Mark Elderkin (Nova Scotia Department of Natural Resources), Shelley Pardy, Bruce Rodrigues, Allysia Park, Sara McCarthy (Newfoundland and Labrador Department of Environment and Conservation), Marie-Josée Laberge, Pippa Shepherd (Parks Canada Agency), and staff at the Department of National Defence provided expertise, document reviews, and/or data during the development of this recovery strategy.
Executive Summary
Little Brown Myotis (Myotis lucifugus), Northern Myotis (Myotis septentrionalis), and Tri-colored Bat (Perimyotis subflavus) are small, insectivorous species of the Family Vespertilionidae. The three species were emergency listed as Endangered on Schedule 1 of the federal Species at Risk Act (SARA) in 2014 because of sudden and dramatic declines across the eastern portions of the ranges of Little Brown Myotis and Northern Myotis, and throughout the entire Canadian range of Tri-colored Bat. These declines are the direct result of white-nose syndrome (WNS).
Little Brown Myotis and Northern Myotis have been confirmed in every province and territory except Nunavut. Approximately 50% and 40% of their global ranges occur in Canada, respectively. Tri-colored Bat has been recorded in Ontario, Quebec, New Brunswick, and Nova Scotia, and 10% of its global population is estimated to occur in Canada.
The single greatest threat to Little Brown Myotis, Northern Myotis, and Tri-colored Bat is WNS. Because of the significance of WNS, where appropriate, this recovery strategy differentiates between areas affected by WNS and those not yet affected (e.g., within population and distribution objectives, threats, and recovery approaches). In areas already affected by WNS, the significance of other threats to the three species of bats is heightened because the mortality of a small number of the remaining individuals (particularly adults) has the ability to impact the survival of local populations, their recovery, and, perhaps, the development of resistance to the fungus that causes WNS. Threats other than WNS include habitat loss and degradation (e.g., destruction or degradation of hibernacula, maternity roosts, and foraging areas), disturbance or harm (e.g., collisions with or barotraumaFootnote1 from wind turbines, intentional harm to individuals, recreational or scientific disturbance, and industrial disturbance), pollution, and climate change.
The feasibility of recovery for Little Brown Myotis, Northern Myotis, and Tri-colored Bat in Canada is unknown. In keeping with the precautionary principle, this recovery strategy has been prepared as per section 41(1) of SARA.
Within WNS-affected areas, the short-term (12-18 years) population objective for both Little Brown Myotis and Northern Myotis is to maintain (and where feasible increase) the population compared to its current (2015) level. Within WNS-affected areas, the long-term (many generations) population objective is a self-sustaining, resilient, and redundant population (see section 5: Population and Distribution Objectives for further details and definitions). Within areas not yet affected by WNS, the population objective is to maintain (and where feasible increase) the population compared to its current level. The distribution objective for both the Little Brown Myotis and Northern Myotis is to maintain (or where applicable restore to) the pre-WNS extent of occurrence.
The short-term population objective for Tri-colored Bat is to maintain (and where feasible increase) the population compared to its current (2015) level over the next 10 years. The long-term population objective is a self-sustaining, resilient, and redundant population. The distribution objective is to restore (then maintain) the pre-WNS extent of occurrence.
Broad strategies aimed at supporting the survival and recovery of Little Brown Myotis, Northern Myotis, and Tri-colored Bat are presented in section 6.2: Strategic Direction for Recovery. Approaches required to meet population and distribution objectives will be different in areas where WNS has already caused dramatic declines, compared to areas not yet affected by WNS.
Critical habitat for these three species is partially identified in this recovery strategy. A schedule of studies is included to obtain the information needed to complete the identification of critical habitat.
One or more action plans for Little Brown Myotis, Northern Myotis, and Tri-colored Bat will be posted on the Species at Risk Public Registry within three years following the final posting of this recovery strategy.
Recovery Feasibility Summary
Based on the following four criteria that the Government of Canada uses to determine recovery feasibility, the feasibility of recovery for Little Brown Myotis, Northern Myotis, and Tri-colored Bat is unknown. In keeping with the precautionary principle, a recovery strategy has been prepared as per section 41(1) of SARA, as would be done when recovery is determined to be feasible. This recovery strategy addresses the lack of knowledge related to the feasibility of recovery. Tri-colored Bat’s recovery feasibility is assessed separately because, unlike Little Brown Myotis and Northern Myotis, WNS encompasses Tri-colored Bat’s entire Canadian range.
Little Brown Myotis and Northern Myotis
1. Individuals of the wildlife species that are capable of reproduction are available now or in the foreseeable future to sustain the population or improve its abundance.
Yes. In the provinces and territories where WNS is absent, these species are apparently abundant throughout much of their range. At present, it is believed that there are currently adequate numbers of breeding individuals in the Prairies and western Canada to sustain the species in Canada or increase their abundances. The number of individuals remaining in most parts of eastern Canada and the United States (U.S) is very small. Hence, rescue from outside populations (i.e., the U.S.) is not considered likely. However, there are early indications in some portions of their range that a very small proportion of individuals may survive WNS.
2. Sufficient suitable habitat is available to support the species or could be made available through habitat management or restoration.
Unknown. Sufficient suitable summering habitat (i.e., roosting and foraging habitat) is likely available, and it is possible that more could be made available through management or restoration. However, not all locations are known. Suitable wintering habitat (i.e., hibernacula) that is free of the spores that cause WNS is probably sufficient in the Prairies and western and northern Canada, but has substantially declined in many areas of eastern Canada. Although the habitat characteristics of hibernacula are generally known in eastern Canada, the locations of some hibernacula there remain unknown. In the Prairies, western Canada, and northern Canada, the habitat characteristics and locations of hibernacula are not well understood. Currently, it is not possible to identify available, but currently unoccupied wintering habitat that may be needed for recovery for either of these two species.
3. The primary threats to the species or its habitat (including threats outside Canada) can be avoided or mitigated.
Unknown. The primary threat to these species is WNS which is caused by an invasive fungus (Pseudogymnoascus destructans). Spores that cause WNS are introduced to uninfected hibernacula. Limiting the human-assisted spread of WNS by avoiding activities that can cause the spores to spread to uninfected hibernacula may be possible through education and adherence to handling protocols and beneficial management practices (BMPs). Nevertheless, WNS is predicted to continue to spread across Canada from bat-to-bat contact. It is unknown if natural barriers (e.g., Rocky Mountains) may halt or slow the spread of the disease by bats. No widely tested treatment for WNS exists, but intensive research is currently focused on potential treatment and mitigation measures. New research has found substances from certain bacterium and yeast that inhibit the growth of P. destructans in the laboratory. This research is still in early stages of development and its effectiveness of application in the field is unknown.
4. Recovery techniques exist to achieve the population and distribution objectives or can be expected to be developed within a reasonable timeframe.
Unknown. Although WNS has primarily spread through Canada by bats, habitat management and stewardship to prevent (or slow) the human-assisted spread of the spores that cause WNS may be effective recovery techniques for these species. Potential treatments and mitigation measures for WNS are currently being researched and tested. Habitat enhancement techniques that increase the availability of optimal roosting locations in areas where bats are known to raise young may aid in the recovery of these species. In addition, limiting access and disturbance to hibernacula can have positive effects on population growth.
Tri-colored Bat
1. Individuals of the wildlife species that are capable of reproduction are available now or in the foreseeable future to sustain the population or improve its abundance.
Yes. In eastern Canada, where this species and WNS are present, Tri-colored Bat continues to persist at very low population levels. Populations also continue to exist in the U.S., although rescue from outside populations is not considered likely. There are no reliable Canadian estimates of population size. At present, it is believed that there are limited individuals capable of reproduction available to increase population abundances.
2. Sufficient suitable habitat is available to support the species or could be made available through habitat management or restoration.
Unknown. Sufficient suitable summering habitat (i.e., roosting and foraging habitat) is likely available, although not all locations are known. Suitable wintering habitat (i.e., hibernacula) that is free of the fungus that causes WNS may be non-existent because Tri-colored Bat’s entire Canadian range overlaps with the areas affected by WNS. Not all locations of hibernacula are known for this species. It is currently not possible to identify available but currently unoccupied habitat that may be needed for recovery.
3. The primary threats to the species or its habitat (including threats outside Canada) can be avoided or mitigated.
Unknown. The primary threat to these species is an invasive fungus (Pseudogymnoascus destructans). No widely tested treatment for WNS exists, but intensive research is currently focused on potential treatment and mitigation measures. New research has found substances from certain bacterium and yeast that inhibit the growth of P. destructans in the laboratory. This research is still in early stages of development and its effectiveness of application in the field is unknown.
4. Recovery techniques exist to achieve the population and distribution objectives or can be expected to be developed within a reasonable timeframe.
Unknown. Because it is presumed that WNS encompasses the entire range of Tri-colored Bat in Canada, it will be challenging to conduct the necessary research for the treatment of WNS within a reasonable timeframe for recovery. This is an area of intensive ongoing research.
1. COSEWICFootnote* Species Assessment Information
- Date of Assessment:
- November 2013
- Common Name (population):
- Little Brown Myotis
- Scientific Name:
- Myotis lucifugus
- COSEWIC Status:
- Endangered
- Reason for Designation:
- Approximately 50% of the global range of this small bat is found in Canada. Sub-populations in the eastern part of the range have been devastated by White-nose Syndrome, a fungal disease caused by an introduced pathogen. This disease was first detected in Canada in 2010, and to date has caused a 94% overall decline in known numbers of hibernating Myotis bats in Nova Scotia, New Brunswick, Ontario, and Québec. The current range of White-nose Syndrome has been expanding at an average rate of 200-250 kilometres per year. At that rate, the entire Canadian population is likely to be affected within 12 to 18 years. There is no apparent containment of the northward or westward spread of the pathogen, and proper growing conditions for it exist throughout the remaining range.
- Canadian Occurrence:
- Yukon, Northwest Territories, British Columbia, Alberta, Saskatchewan, Manitoba, Ontario, Quebec, New Brunswick, Prince Edward Island, Nova Scotia, Newfoundland and Labrador
- COSEWIC Status History:
- Designated Endangered in an emergency assessment on February 3, 2012. Status re-examined and confirmed in November 2013.
- Date of Assessment:
- November 2013
- Common Name (population):
- Northern Myotis
- Scientific Name:
- Myotis septentrionalis
- COSEWIC Status:
- Endangered
- Reason for Designation:
- Approximately 40% of the global range of this northern bat is in Canada. Sub-populations in the eastern part of the range have been devastated by White-nose Syndrome, a fungal disease caused by an introduced pathogen. This disease was first detected in Canada in 2010 and to date has caused a 94% overall decline in numbers of known hibernating Myotis bats in Nova Scotia, New Brunswick, Ontario, and Québec hibernacula compared with earlier counts before the disease struck. Models in the northeastern United States for Little Brown Myotis predict a 99% probability of functional extirpation by 2026. Given similar life history characteristics, these results are likely applicable to this species. In addition to its tendency to occur in relatively low abundance levels in hibernacula, there is some indication this species is experiencing greater declines than other species since the onset of White-nose Syndrome. The current range of White-nose Syndrome overlaps with approximately one third of this species' range and is expanding at an average rate of 200 to 250 kilometres per year. At that rate, the entire Canadian population will likely be affected within 12 to 18 years. There is no apparent containment of the northward or westward spread of the pathogen, and proper growing conditions for it exist throughout the remaining range.
- Canadian Occurrence:
- Yukon, Northwest Territories, British Columbia, Alberta, Saskatchewan, Manitoba, Ontario, Quebec, New Brunswick, Prince Edward Island, Nova Scotia, Newfoundland and Labrador
- COSEWIC Status History:
- Designated Endangered in an emergency assessment on February 3, 2012. Status re-examined and confirmed in November 2013.
- Date of Assessment:
- November 2013
- Common Name (population):
- Tri-colored Bat
- Scientific Name:
- Perimyotis subflavus
- COSEWIC Status:
- Endangered
- Reason for Designation:
- This bat is one of the smallest bats in eastern North America. Approximately 10% of its global range is in Canada, and it is considered rare in much of its Canadian range. Declines of more than 75% have occurred in the known hibernating populations in Québec and New Brunswick due to White-nose Syndrome. This fungal disease, caused by an invasive pathogen, was first detected in Canada in 2010, and has caused similar declines in Little Brown Myotis and Northern Myotis in eastern Canada and the northeastern United States. Most of the Canadian range of the species overlaps with the current White-nose Syndrome range, and further declines are expected as more hibernacula continue to become infected.
- Canadian Occurrence:
- Ontario, Quebec, New Brunswick, Nova Scotia
- COSEWIC Status History:
- Designated Endangered in an emergency assessment on February 3, 2012. Status re-examined and confirmed in November 2013.
2. Species Status Information
Approximately 50%, 40%, and 10% of the global ranges of Little Brown Myotis, Northern Myotis, and Tri-colored Bat, respectively, occur in Canada (COSEWIC 2013).
Little Brown Myotis, Northern Myotis, and Tri-colored Bat were listed as Endangered in Canada under Schedule 1 of the Species at Risk Act (SARA, c. 29) in November 2014 by an emergency listing order. Little Brown Myotis and Northern Myotis were listed as Endangered in January 2013 on the Species at Risk in Ontario (SARO) List (O. Reg. 230/08) under Ontario’s Endangered Species Act, 2007 (OMNRF 2015). The three species also receive protection under Ontario’s Fish and Wildlife Conservation Act, 1997 as specially protected mammals. All three species were listed as Endangered by the New Brunswick Species at Risk Act in June 2013 and were added to the list of animals protected under the Nova Scotia Endangered Species Act in July 2013. Little Brown Myotis and Northern Myotis were listed as Endangered under Manitoba’s Endangered Species and Ecosystems Act in June 2015.
These species are not listed under the provincial and territorial endangered species legislations of Yukon, British Columbia, Northwest Territories, Alberta, Saskatchewan, Quebec, or Newfoundland and Labrador, although they are afforded protections under the Wildlife Acts of these provinces and territories. In Quebec, Tri-colored Bat is listed on the Liste des espèces susceptibles d’être désignées menacées ou vulnérables (list of wildlife species likely to be designated threatened or vulnerable). This list is produced according to the Loi sur les espèces menacées ou vulnérables (RLRQ, c E-12.01) (Act respecting threatened or vulnerable species) (CQLR, c E-12.01), but it does not afford any extra protection to the species. In Saskatchewan, The Wildlife Amendment Regulation, 2013 removed all Chiroptera in Saskatchewan from the list of unprotected wildlife, thereby granting them protection from unlicensed hunting. These species are not listed, nor do they receive protection under the Wildlife Conservation Act in Prince Edward Island. There are no confirmed records of these species in Nunavut, but there has been limited survey coverage.
NatureServe (2015) ranks for Canada and the United States (U.S.) are presented in Table 1.
| Areas | Little Brown Myotis | Northern Myotis | Tri-colored Bat | Presence of WNS (winter discovered) |
|---|---|---|---|---|
| Global | G3G4 | G1G2 | G3G4 | - |
| United States | N3 | N1N2 | N3N4 | Yes (2006-2007) |
| Canada | N3 | N2N3 | N2N3 | - |
| Newfoundland | S4 | S2S3 | Not Applicable | - |
| Labrador | S4 | SNR | Not Applicable | - |
| Nova Scotia | S1 | S2 | S1 | Yes (2010-2011) |
| PEI | S1 | S1 | Not Applicable | Yes (2012-2013) |
| Quebec | S1 | S1 | S1 | Yes (2009-2010) |
| New Brunswick | S1 | S1 | S1 | Yes (2010-2011) |
| Ontario | S4 | S3 | S3? | Yes (2009-2010) |
| Manitoba | S2N, S5B | S3S4N, S4B | Not Applicable | - |
| Saskatchewan | S5B, S5N | S4B, SNRN | Not Applicable | - |
| Alberta | S5 | S2S4 | Not Applicable | - |
| British Columbia | S4 | S3S4 | Not Applicable | - |
| NT | S2 | S2 | Not Applicable | - |
| Yukon | S1S3 | S1S2 | Not Applicable | - |
- Types of ranks: G = global conservation status rank, N = national conservation status rank, and S = sub-national (provincial or territorial) ranks.
- Definitions of rank: 1 = critically imperiled; 2 = imperiled; 3 = vulnerable; 4 = apparently secure; 5 = secure; SNR = unranked; SU = unrankable; B = breeding; N = non-breeding.
3. Species Information
3.1. Species Description
Little Brown Myotis
Little Brown Myotis is a small (7-9 g) brown bat with black ears, black wings, and a black tail membrane (van Zyll de Jong 1985). Its wingspan is approximately 22-27 cm. Females tend to be slightly larger than males (Harvey et al. 2011). Compared to other mammals, Little Brown Myotis has a long lifespan; some individuals live more than 30 years (Davis and Hitchcock 1995).
Northern Myotis
Northern Myotis, also known as Northern Long-eared Bat, is a small bat (5-8 g) similar in size and colouration to Little Brown Myotis, but is generally distinguishable by its longer ears that extend beyond the nose when pressed forward, longer tail, and larger wing area (Caceres and Barclay 2000, Harvey et al. 2011). It can also be distinguished by its tragusFootnote2, which is long and thin with a pointed tip (van Zyll de Jong 1985). Northern Myotis has similar life history characteristics to Little Brown Myotis; the longevity record in the wild is 18.5 years (Caceres and Barclay 2000).
Tri-colored Bat
Tri-colored Bat, formerly known as Eastern Pipistrelle (Pipistrellus subflavus), has a distinct colouration; each hair is black at the base, yellow in the middle, and brown at the tip giving the bat an overall reddish-brown to yellowish-brown colour (Harvey et al. 2011). Its ears and face are brown, forearms are orange-red or pinkish, and its wings and flight membranes are blackish (Fujita and Kunz 1984, Naughton 2012). Tri-colored Bat is similar in size and weight (5-9 g) to Little Brown Myotis and Northern Myotis (Fujita and Kunz 1984, van Zyll de Jong 1985, Farrow and Broders 2011).
3.2. Population and Distribution
Distribution
Little Brown Myotis
Little Brown Myotis has been confirmed in every province and territory throughout Canada with the exception of Nunavut where no known observations meeting evidentiary standards exist (i.e., recorded but not confirmed) (COSEWIC 2013). In general, its Canadian distribution includes the boreal forest south of the treeline through to the U.S. border (van Zyll de Jong 1985, Grindal et al. 2011, Burles et al. 2014) (Figure 1).
The northern limit of its distribution is difficult to define because of limited survey effort and difficulties related to survey logistics (e.g., large area, few roads – see Jung et al. 2014 for a more comprehensive explanation). Few maternity roosts or hibernacula have been located in the northern portions of the range (COSEWIC 2013); however, Wilson et al. (2014) observed reproductive females and maternity colonies of Little Brown Myotis in southwest and south central Northwest Territories. The species is also found south of 64° in Yukon (Slough and Jung 2008). Hibernacula have been confirmed in the Northwest Territories though no hibernacula have been found in Yukon (Slough and Jung 2008, Wilson et al. 2014). Scattered records from Nunavut and northern Northwest Territories exist (i.e., north of the defined range in Figure 1); however, it is unclear if these records represent resident breeding individuals or extralimitalFootnote3 observations (COSEWIC 2013).
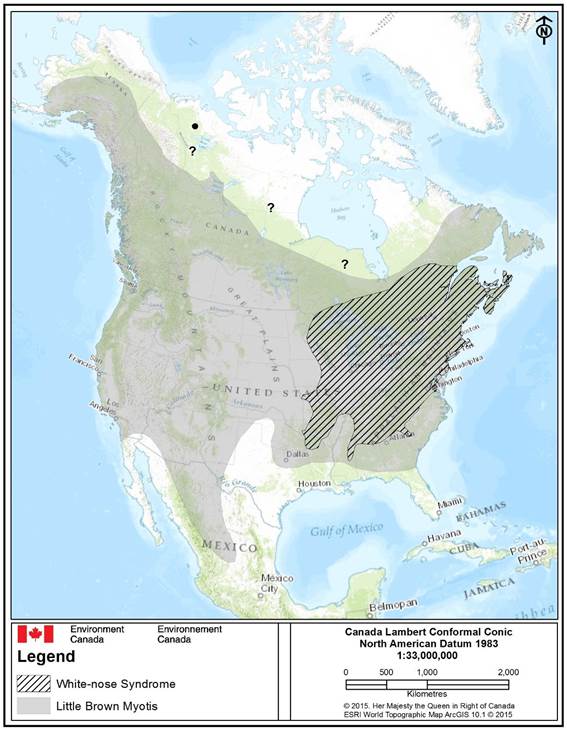
Long description for Figure 1
Approximate North American distribution (range) map of Little Brown Myotis (Myotis lucifugus) (grey-filled area) and overlap with white-nose syndrome, Pseudogymnoascus destructans, (WNS) (black-diagonal cross-fill) as of May 2015 ((Naughton 2012, COSEWIC 2013, Wilson et al. 2014, CWHC 2015, GNWT 2015a). Recorded, but unconfirmed locations of Little Brown Myotis in the Northwest Territories and Nunavut are indicated by question marks (“?”), whereas an extralimital observation (outside defined range as represented by the grey-filled area) is represented as a black dot. The species has confirmed occurences in every Canadian province and territory except Nunavut (unconfirmed).
The continental range of the species is continuous, and extends from a northern-most limit of central Alaska, north-central Yukon, southern Northwest Territories following the boreal forest treeline into the northern portions of Saskatchewan, Manitoba, Ontario, Quebec, Newfoundland and Labrador. The western range limit extends from the Alaskan coastline to northmost tip of the Baja California Peninsula in Mexico. The southwestern range limit extends from Baja California Peninsula along the US-Mexico border extending into north-central Mexico. The south-central rangle limit extends from northern Texas into the northern portions of the US gulf states including Louisiana, Mississipi, and southern borders of Alabama and Georgia. The eastern range limits extend northern Newfoundland and Labrador along the Atlantic coastline to southern Georgia in the US. The distribution of White-nose syndrome overlaps the central-eastern Atlantic range of the species.
Northern Myotis
Northern Myotis has been confirmed in every province and territory throughout Canada with the exception of Nunavut (van Zyll de Jong 1985, Brown et al. 2007, Henderson et al. 2009, Park and Broders 2012, Broders et al. 2013, Reimer and Kaupas 2013) (Figure 2). Its Canadian distribution includes the boreal forest south of the treeline and into the montane forests of the west and deciduous and mixedwood forests of the east. It is mostly absent from the Canadian Prairies, and when it is found outside of forested regions, it is found in forest remnants or at hibernacula (Turner 1974).
Similar to Little Brown Myotis, the northern limit of Northern Myotis’ range is difficult to determine due to limited survey effort and difficulties related to survey logistics (e.g., large area, few roads). However, Northern Myotis has been confirmed breeding in Yukon (Lausen et al. 2008) and the Northwest Territories (Wilson et al. 2014). Hibernation sites have not been recorded in Yukon (Jung et al. 2006, Slough and Jung 2007), but likely exist in the Northwest Territories (Wilson et al. 2014).
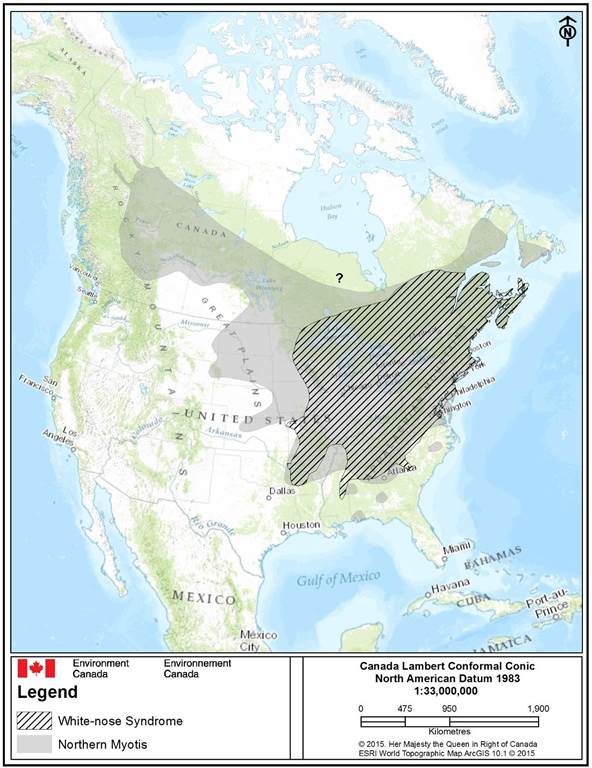
Long description for Figure 2
Approximate North American distribution (range) map of Northern Myotis (Myotis septentrionalis) (grey-filled area) and overlap with white-nose syndrome, Pseudogymnoascus destructans, (WNS) (black-diagonal cross-fill) as of May 2015 (Naughton 2012, COSEWIC 2013, Wilson et al. 2014, CWHC 2015, GNWT 2015b)). Recorded, but unconfirmed occurrence of Northern Myotis in Ontario is indicated by a question mark (“?”). The species has confirmed occurences in every Canadian province and territory except Nunavut (unconfirmed)(van Zyll de Jong 1985, Brown et al. 2007, Henderson et al. 2009, Park and Broders 2012, Broders et al. 2013, Reimer and Kaupas 2013).
The continental range of the species is near-continuous, and extends from a northern-most limit of southeast Yukon/southwest Northwest Territories into the north-central portions of the Canadian Praires and Ontario, central Quebec, Newfoundland and Labrador. The southern range of the species covers south-central US states though largely absent from US-midwest and central prairies. The west to eastern range limits of the species extends from the boreal forest of western Canada (British Columbia) across the central-northern portions of the Canadian prairie provinces, northern Ontario, north-central Quebec to the far southeast of Newfoundland. The eastern limits of the species extend from the interior of central Georgia and northwest South Carolina in the US, along the Atlantic coastline into southeast Newfoundland and southeast Labrador. The species has also has a discontinous range in the southeast Us observed by isolated occurrence in Alabama, Florida, Georgia and central South Carolina. Overlap with White-nose-syndrome covers the eastern North American portion of the species range.
Tri-colored Bat
The Canadian range of Tri-colored Bat encompasses mainland Nova Scotia, southern New Brunswick, Quebec and Ontario (van Zyll de Jong 1985, Fraser et al. 2012) (Figure 3), although some populations may be isolated within this range (Broders et al. 2003). Breeding records for Tri-colored Bat have been confirmed in Nova Scotia (Broders et al. 2003) though breeding was not observed in New Brunswick (Broders et al. 2001). Although detections of Tri-colored Bat have been recorded from Ontario and Quebec in the summer (COSEWIC 2013), it is unclear if breeding has been confirmed. It has been recorded in hibernacula in all provinces within its range, albeit rarely and/or in relatively small numbers (COSEWIC 2013).
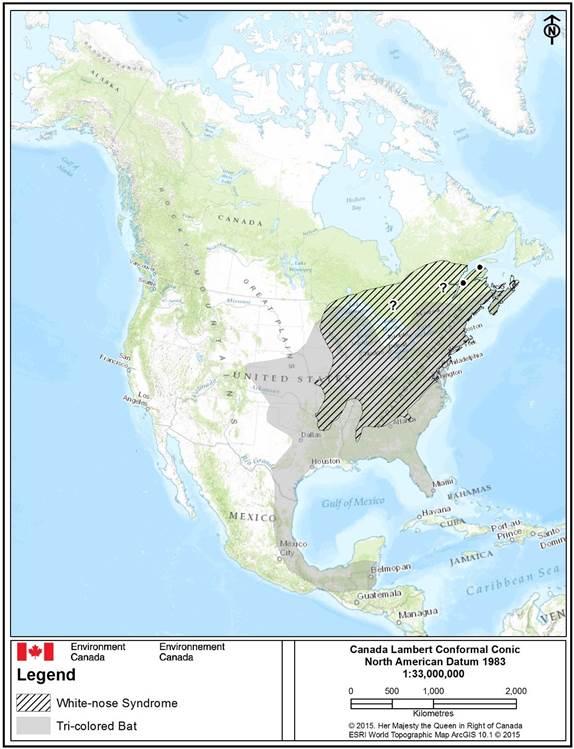
Long description for Figure 3
Approximate North American distribution (range) map of Tri-colored Bat (Perimyotis subflavus) (grey-filled area) and overlap with white-nose syndrome, Pseudogymnoascus destructans, (WNS) (black-diagonal cross-fill) as of May 2015 (Naughton 2012, COSEWIC 2013, CWHC 2015). Recorded, but unconfirmed occurrences of Tri-colored Bat in Ontario and Quebec are indicated by a question mark (“?”), whereas northern range extralimital records are indicated by black dots. The species ocucrence in Canda is limited to central-eatern provinces including Ontario, Quebec, southern New Brunswick and Nova Scotia.
The continental range of the species is continuous, but confined from central to eastern US and Canada with a northern limit along the Great-Lakes and Laurentiens in Canada. The species range narrows/contracts from southern US Gulf states into Mexico where its southern range follows the western Gulf of Mexico coastline as far south as Guatemala. Overlap of the species range with White-nose syndrome covers the northeast portion of the species range.
Population
This recovery strategy defines two periods relevant to the discussion of bat populations in Canada: the period prior to the arrival of WNS in Canada (up to and including 2009) and the period after the arrival of WNS in Canada (2010 and onwards). However, it should be noted that the population sizes and the relative abundance of the three bat species in Canada are unknown pre- and post-WNS (both in Canada and in North America) and, therefore, it is challenging to obtain accurate estimates of species-specific declines in the Canadian populations.
In the northeastern U.S., Langwig et al. (2012) estimated that bat populations (all species) in general, prior to the arrival of WNS, were growing at an average rate of 8% per year. Population trend analyses of hibernacula data from across the U.S. indicated these three species of bat specifically were relatively stable (i.e., a positive or negative trend was unable to be detected) (Ellison et al. 2003, Frick et al. 2010a, Frick et al. 2010b). Substantial intra- and inter-annual variation in the number of hibernating bats (and species) can exist within a hibernaculum (Trombulak et al. 2001); nonetheless, winter hibernacula data are likely an accurate reflection of the population status in all three bat species’ populations (COSEWIC 2013). In addition, summer survey data corroborate observations collected at hibernacula (COSEWIC 2013, Natureserve 2015).
Since the arrival of WNS to North America in 2006, the most precipitous declines in North American wildlife in recorded history have been observed (Kunz and Tuttle 2009). An estimated one million bats (multiple species) died in the northeastern U.S. within three years of the arrival of WNS (Kunz and Tuttle 2009), and an estimated 5.7 to 6.7 million bats died within six years of its arrival (U.S. Fish and Wildlife Service 2012). In Canada, the total number of Myotis spp. bats recorded in Nova Scotia, New Brunswick, Ontario, and Quebec hibernacula declined by approximately 94% between 2010 and 2012 (COSEWIC 2013). In Quebec, Nova Scotia, and New Brunswick, some hibernacula no longer have any individuals of these species of bats present (McAlpine and Vanderwolf, unpub. data in COSEWIC 2013, Mainguy and Desrosiers 2011, H. Broders pers. comm. 2015).
There is limited information related to the proportion of bat populations found in eastern Canada versus western Canada. In Canada, 95% of records of hibernating Myotis spp. bats are from Nova Scotia to Manitoba, while relatively few had been recorded west of Manitoba (COSEWIC 2013). Fewer hibernacula have been found in the western provinces and northern territories as compared to the east (excluding Newfoundland and Labrador where few hibernacula are known); furthermore, hibernacula in the east tended to have more individuals per site (>10,000) compared to hibernacula in the north and west (<1000 per site) (Nagorsen and Brigham 1993, Olson et al. 2011). Some researchers suggest that a large proportion of the Little Brown Myotis population prior to WNS resided in the northeastern U.S. (Kunz and Reichard 2010). Genetic evidence also exists to suggest populations in the east were larger than populations in the west (Wilder 2014). However, bats in northern and western Canada may not necessarily be less abundant but instead tend to overwinter singly or in small numbers, making it more difficult to obtain accurate population estimates (i.e., many sites with small numbers of bats in the north and west compared to few sites with large numbers in the east). Comparisons between eastern and western population levels should be interpreted with caution because of the survey limitations previously mentioned for the north in addition to issues in the west (i.e., British Columbia) in differentiating Little Brown Myotis from Yuma Myotis (Myotis yumanensis) (COSEWIC 2013).
Little Brown Myotis
Pre-WNS
Evidence from multiple surveys suggests that Little Brown Myotis was probably the most common bat throughout much of Canada, and still is in many areas outside of those impacted by WNS (COSEWIC 2013). Little Brown Myotis is thought to be relatively common in the northern limits of its range, although abundance is difficult to estimate because of previously discussed issues with surveying populations in the north (COSEWIC 2013, Jung et al. 2014).
The Canadian population size of Little Brown Myotis prior to the arrival of WNS is unknown but likely exceeded one million individuals (COSEWIC 2013). Frick et al. (2010a) estimated the population was 6.5 million individuals in the northeastern U.S. as of 2006 which further supports numbers in excess of 1 million for Canada. NatureServe (2015) estimated the global population size to be 100,000 to >1,000,000 individuals prior to WNS. Data from known hibernacula are incomplete but, prior to the arrival of WNS, some known hibernacula were used by thousands to tens of thousands of bats in southern Ontario, Quebec, Nova Scotia, New Brunswick, and Manitoba (Fenton 1970, Scott and Hebda 2004, Mainguy and Desrosiers 2011).
Post-WNS
NatureServe (2015) states that the current global population size is unknown but likely still exceeds 100,000 individuals. Model results predicted that Little Brown Myotis will be functionally extirpatedFootnote4 (i.e., 1% of pre-WNS population or 65,000 individuals) in the northeastern U.S. by 2026 (Frick et al. 2010a). However, new evidence suggests that some individuals are surviving the infection and survival rates have increased at locations previously decimated by WNS; however, the increased rates of survival are not sufficient to support a positive population growth trend (Maslo et al. 2015). The entire Canadian range of Little Brown Myotis is expected to be impacted by WNS between 2025 and 2028 (COSEWIC 2013).
Northern Myotis
Pre-WNS
Evidence suggests that Northern Myotis is less common than Little Brown Myotis, in part, because they have a more restricted distribution within Canada and are reliant on forested areas (COSEWIC 2013). Observations made during summer indicate that the species is relatively common in the southern Northwest Territories and uncommon at the western and northern edges of their range (Jung et al. 2014, Wilson et al. 2014). At some eastern sites (e.g., Newfoundland and Labrador, Prince Edward Island, and Nova Scotia), numbers were more or less equal to Little Brown Myotis (Park and Broders 2012). However, counts obtained from individual hibernacula in the winter indicate relatively few (i.e., <100) Northern Myotis (Barbour and Davis 1969, Amelon and Burhans 2006). This may be a result of difficulties in detecting Northern Myotis in hibernacula because they are often found in deep cracks (COSEWIC 2013).
The Canadian population size of Northern Myotis is unknown but is believed to have also exceeded one million individuals before the arrival of WNS (COSEWIC 2013). In contrast, NatureServe (2015) estimated that the global population size was relatively small prior to WNS (2,500 to 100,000 individuals). However, NatureServe notes that this estimate is supported by low counts at hibernacula (which may be related to issues with detectability). In addition, the NatureServe estimate appears to be primarily based on counts within the U.S. part of the range. Based on Harvey (1992) and D. Morningstar (pers. comm. 2015), the species is probably more common and abundant in the northern part of its global range (i.e., boreal) than in the south (COSEWIC 2006).
Post-WNS
Declines of Northern Myotis populations in the northeastern U.S. have occurred at the same rate as Little Brown Myotis; an expected result given the two species have similar life history traits and often share same hibernacula. Thus, it was predicted that Northern Myotis will likely also be functionally extirpated (i.e., 1% of pre-WNS population) in the northeastern U.S. by 2026 (Frick et al. 2010a). However, new evidence for Little Brown Myotis suggests that low numbers of individuals are surviving the infection and survival rates have increased at locations previously decimated by WNS, a trend which may also apply to Northern Myotis. The entire Canadian range of Northern Myotis is expected to be impacted by WNS between 2025 and 2028 (COSEWIC 2013).
Tri-colored Bat
Pre-WNS
Although too little data exist to reliably estimate the population size of Tri-colored Bat, the species was relatively rare in the Maritimes, Quebec, and in parts of Ontario (COSEWIC 2013). It is also rare in the adjacent states of Vermont (Darling and Smith 2011) and Maine (Zimmerman and Glanz 2000). In addition to being uncommon, Tri-colored Bats tend to hibernate solitarily within hibernacula, often within the deepest parts of the cave where human access is limited, and thus, may be more difficult to detect during hibernation than other species (Hitchcock 1949, Fujita and Kunz 1984, Sandel et al. 2001, COSEWIC 2013).
The Canadian population size of Tri-colored Bat prior to WNS is unknown; however, the COSEWIC (2013) status report provides <20,000 individuals in Canada as a rough estimate. NatureServe (2015) estimated that the global population size was between 10,000 to 1,000,000 individuals prior to WNS. In Nova Scotia, it was estimated that between 1,000 and 2,000 adult females existed (H. Broders, pers. comm. in COSEWIC 2013). Across the Canadian range, the species accounted for between 0.2 to 4.5% of individuals counted at various hibernacula (Hitchcock 1949, 1965, Mainguy and Desrosiers 2011, Vanderwolf et al. 2012).
Post-WNS
The Tri-colored Bat population declines in areas affected by WNS in Canada and the U.S. are likely similar to that observed in Little Brown Myotis and Northern Myotis, though the declines observed in this species are less straightforward (COSEWIC 2013). In part, this is because of the small numbers of Tri-colored Bats that have been recorded from monitored Canadian hibernacula and because the species shows evidence of a seasonal latitudinal migration (Fraser et al. 2012), both of which may affect interpretation of Canadian population trends.
Declines observed since the arrival of WNS have been variable. The average population decline in five northeastern states was 75% (range 16 to 95%) between 2006 and 2010, with 13 of 36 hibernacula declining 100% (Turner et al. 2011). Acoustic monitoring during the summers of 2007-2009 in New York yielded similar population declines to those listed above (i.e., 78% between 2008 and 2009) (Dzal et al. 2011). In eastern Ontario, Quebec, and Nova Scotia there are indications the Tri-colored Bat population may have declined by as much as 94% compared to pre-WNS populations (Mainguy and Desrosiers 2011, L. Hale, pers. comm. in COSEWIC 2013). In southern Ontario, significant declines were also noted at eight locations where acoustic monitoring was performed prior to the arrival of WNS and again in 2014 (D. Morningstar, pers. comm.). In New Brunswick, declines at individual hibernacula have ranged from 30% to more than 75% (D. McAlpine and K. Vanderwolf, unpub. data in COSEWIC 2013, D. McApline pers. comm. in COSEWIC 2013). NatureServe (2015) states that the current global population size is unknown but likely still exceeds 10,000 individuals.
3.3 Needs of Little Brown Myotis, Northern Myotis, and Tri-colored Bat
The habitat requirements of temperate-region bats vary by season. The habitat is composed of (1) overwintering habitat (i.e., hibernacula, such as caves, abandoned mines, and wells) used for hibernation and overwinter survival, (2) summering habitat that includes roosting habitat (for maternity roosts and males) and foraging habitat within commuting range of the roosts (Sasse and Perkins 1996, Norquay et al. 2013), and (3) swarming habitat used in the late summer and early fall for mating and socializing (Fenton 1969, Randall and Broders 2014). Swarming sites are also typically used as hibernacula (Fenton 1969, Randall and Broders 2014).
Overwintering and Swarming Habitat
Hibernating bats survive the winter using stored fat reserves accumulated during the summer and autumn (Jonasson and Willis 2011). Hibernation allows year-round resident, insect-eating bats to persist in a region when ambient temperature declines and insects are not available in winter. Hibernating bats minimize use of fat reserves by decreasing metabolic rate and body temperature to within a few degrees of the ambient temperature in the hibernaculum (i.e., they enter torporFootnote5) (Henshaw and Folk 1966).
Hibernacula for Little Brown Myotis, Northern Myotis, and Tri-colored Bat are generally underground openings, including caves, abandoned mines, wells, and tunnels, but at some sites only specific sections of the site will be used for hibernation. The sections used for hibernating typically have a temperature range between 2˚C and 10˚C (Fenton 1970, Anderson and Robert 1971, Vanderwolf et al. 2012), and relative humidity levels > 80% to reduce evaporative water loss (Cryan et al. 2010, but see Kurta 2014). Structural features, such as number of openings, cave size and length, and angle of chambers can influence the stability and levels of humidity and temperature (Davis 1970, Raesly and Gates 1987). Because of the specific, stable microclimates required by bats, hibernacula are typically used year after year by overwintering bats.
Stable microclimates are preferred by bats because temperature fluctuations can cause arousals from torpor. Bats will arouse from torpor to access water, groom, and mate (Whitaker and Rissler 1993, Thomas 1995), but they consume a significant portion of their limited energy reserve during arousals (Thomas et al. 1990). Relocating to more suitable sites can accelerate the depletion of limited energy reserves, but may also be used as an adaptation for long-term energy conservation.
Where their distributional ranges overlap, all three bat species may occur in the same hibernaculum, but may be found in different sections. Northern Myotis and Tri-colored Bat do not typically overwinter in buildings, but Little Brown Myotis may overwinter in buildings in western Canada (C. Lausen, pers. comm.), where winter temperatures are relatively high. Little is known regarding the overwintering habitat of Little Brown Myotis and Northern Myotis in western Canada.
Bats congregate in swarming habitat in the late summer and early fall. Swarming sites may function as mating sites, stopover locations during migration, social sites for information transfer, and/or allow individuals to assess potential sites for overwintering (Fenton 1969, Randall and Broders 2014). Swarming behaviour often occurs in and around entrances or openings of hibernacula. Both Little Brown Myotis and Northern Myotis often swarm and hibernate at the same locations (Randall and Broders 2014), but the proportion of bats that visit a swarming site in relation to those that stay at the site for hibernation is unknown (Johnson et al. 2015). Swarming groups of Little Brown Myotis and Northern Myotis in eastern Canada are comprised of individuals from various summering sites and therefore gene pools may span relatively large areas (Burns et al. 2014, Johnson et al. 2015, Segers and Broders 2015).
Few studies have attempted to characterize external habitat features that predict selection of hibernacula and swarming sites by bats. In Nova Scotia, a survey of natural and anthropogenic swarming sites (abandoned mines), also assumed to be hibernacula, revealed that the amount of entrance shelter (i.e., canopy cover and/or rock faces that provide protection from weather elements), watercourse length within a 2 km radius of the site, and total chamber length of the hibernaculum were significant predictors of Little Brown Myotis and Northern Myotis swarming activity (Randall and Broders 2014). Generally, swarming sites had more exposed entrances, greater total length of rivers, creeks, and streams within a 2 km radius, and deeper / longer chamber lengths, than sites surveyed where swarming activity levels were low or absent (Randall and Broders 2014). For example, an 10% increase in the degree of entrance shelter resulted the probability of swarming declining by 50% and the probability of swarming doubled with an increase of 10 km of stream length within a 2 km radius (Randall and Broders 2014).
Little Brown Myotis
Little Brown Myotis appear to derive energetic and water conservation benefits from clustering while hibernating. In Michigan, Kurta and Smith (2014) found that 78% of individuals hibernated in clusters and were more likely to be found in clusters (rather than solitarily) at lower temperatures. In western Canada, the number of Little Brown Myotis hibernating together may be substantially less than in northeastern North America; bats likely hibernate singly or in small groups west of the Rocky Mountains (Jung et al. 2014). A recent radio-telemetry study in the temperate rainforest of southeast Alaska found Little Brown Myotis hibernating solitarily in rock scree on steep, forested hillsides and beneath root wads of trees and stumps (K. Blejwas, pers. comm.). Little Brown Myotis exhibit high fidelity to hibernacula (Norquay et al. 2013). A mark-recapture study in Manitoba and northwestern Ontario found only 4% of marked individuals relocated to an alternate hibernaculum within the study period (Norquay et al. 2013).
Northern Myotis
Northern Myotis may hibernate in cooler sections of a cave, compared to Little Brown Myotis (Barbour and Davis 1969). In a study of abandoned mines in northern Michigan, Northern Myotis and Little Brown Myotis co-occurred in 92% of the mines occupied by Myotis spp., but 75% of Northern Myotis individuals hibernated alone (Kurta and Smith 2014). Northern Myotis will generally return to the same hibernaculum, but not always in consecutive years (Caceres and Barclay 2000). Naughton (2012) noted that “they are loyal to a group of hibernacula rather than a single one”.
Tri-colored Bat
Tri-colored Bat is considered to have the most rigid overwintering habitat requirements of the three species. They often select the deepest part of caves or mines where temperature is the least variable, have strong humidity level preferences, and use warmer walls than other species (Fujita and Kunz 1984, Raesly and Gates 1987, Briggler and Prather 2003, Kurta and Smith 2014). A study of hibernacula in New Brunswick noted Tri-colored Bats hibernating low on the cave walls (Vanderwolf et al. 2012). Although Tri-colored Bats tend to use the same hibernacula as Little Brown Myotis and Northern Myotis, relatively few (i.e., ≤10) Tri-colored Bats have been recorded within any one hibernacula in Canada, possibly because they tend to hibernate solitarily (i.e., not in clusters) in the deepest sections of the caves/mines. Tri-colored Bats exhibit high fidelity to hibernacula (Sandel et al. 2001, Damm and Geluso 2008).
Summering Habitat
Roosting habitat
Roosts provide thermal regulation, shelter from weather and predation, and can be sites for social interaction (Kunz 1982, Barclay and Kurta 2007). Individuals may switch roosts regularly and therefore, may use a network of roosts in a roosting area (Barclay and Brigham 1996, Sasse and Perkins 1996, Caceres and Barclay 2000). The tendency to switch roosts may depend on species, sex, age, reproductive status, and roost type (e.g., natural or anthropogenic) (Garroway and Broders 2008, Randall et al. 2014).
Roost selection is a function of numerous characteristics occurring at a range of spatial scales (Fabianek et al. 2011). For example, at the scale of the roosting structure, tree species, diameter, height, stage of decay, availability of roosting medium, sun exposure, and other characteristics may affect roost selection (Garroway and Broders 2008, Slough 2009, Poissant et al. 2010, Olson and Barclay 2013). At the stand scale, roost selection may be a function of canopy gaps, number of available snags, tree density, proximity to water, etc. (Kalcounis-Rüppell et al. 2005, Garroway and Broders 2008, Henderson and Broders 2008). At the landscape scale, characteristics such as forest age, composition, and degree of fragmentation may affect roost selection (Henderson and Broders 2008, Fabianek et al. 2011). The species may also use treed and forested habitat in urban and suburban areas for roosting, in addition to man-made structures found within urban and suburban landscapes (Little Brown Myotis, in particular).
Many bat species (including Little Brown Myotis, Northern Myotis, and Tri-colored Bat) preferentially roost in older forest stands, compared to young forests (Barclay and Brigham 1996). Older forests likely provide increased snag availability for roosting (Crampton and Barclay 1996, Krusic et al. 1996) and foraging habitat under a relatively closed canopy (Jung et al. 1999).
Females generally give birth and raise pups in maternity colonies in the spring/summer. Because of roost switching behavior, a colony can be defined as an assemblage of roosting groups comprised of individuals that regularly associate and groups that intermix (Olson and Barclay 2013). Roosting in groups likely aids social thermoregulation and energy savings (Willis and Brigham 2007).
Males of all three species roost during the daytime in a variety of structures, and often switch sites during the summer. Male roosting habitat includes rock crevices, raised bark, foliage, and tree cavities (Fenton and Barclay 1980, Caceres and Barclay 2000, Broders and Forbes 2004, Huynh 2009, Randall et al. 2014, Fabianek et al. 2015). Male Little Brown Myotis and Northern Myotis often roost in tall snags with large diameters in the early to middle stages of decay and located in or near small open patches within mature to over mature forest (Broders and Forbes 2004, Jung et al. 2004, Fabianek et al. 2015).
Little Brown Myotis
Little Brown Myotis is one of the few bat species that uses buildings and other anthropogenic structures (e.g., bat boxes, bridges, and barns) to roost (particularly for maternity roosting), but it will also use cavities of canopy trees, foliage, tree bark, crevices on cliffs, and other structures (Fenton and Barclay 1980, Slough 2009, Coleman and Barclay 2011, Randall et al. 2014).
Maternity colonies may include hundreds of females with young. Females show a strong tendency to roost in large-diameter trees, although roost properties may vary significantly throughout the summer (Olson & Barclay 2013). Females are thought to select a preferred maternity roost at the expense of travelling longer distances to forage, possibly indicative of a limited number of suitable maternity roosting sites (Broders et al. 2006, Randall et al. 2014). Female Little Brown Myotis show a relatively high degree of philopatryFootnote6 (Frick et al. 2010b). Roosting areas are generally used annually and individual natural roost sites can be used for upwards of 10 years (M. Brigham, pers. comm.). Little Brown Myotis are particularly loyal to anthropogenic structures and sites may be used for 50 years or more (M. Brigham, pers. comm.). They also exhibit strong within-year site fidelity to anthropogenic structures; Randall et al. (2014) found that most females using anthropogenic structures in Yukon did not switch roosts throughout the summer. Nevertheless, Little Brown Myotis have been documented switching anthropogenic sites between and within years to meet their needs (e.g., thermoregulation) (e.g., Syme et al. 2001). For example, using passive integrated transponder tags, a mix of use was documented at an anthropogenic maternity roost in Ontario; some individuals remained at the roost for several nights, others visited the roost on occasion, and many only visited the roost at night, but did not stay throughout the day, suggesting that individuals were also using other roosts in the area (D. Morningstar, unpub. data.).
Males roost individually or in small groups and periodically switch roosts. In Quebec, males switched roosts approximately every 2 days (Fabianek et al. 2015). Males use a variety of roost structures, including buildings, rock crevices, foliage, raised bark, and tree cavities (Huynh 2009, Randall et al. 2014). In New Brunswick and Quebec, male Little Brown Myotis primarily roosted in coniferous or conifer-dominated mixedwood stands with a large number of snags (Broders and Forbes 2004, Fabianek et al. 2015).
Northern Myotis
Northern Myotis roost singly or in small groups and favour tree roosts (under raised bark and in tree cavities and crevices), but they can also be found in anthropogenic structures (e.g., under shingles) (Sasse and Perkins 1996, Foster and Kurta 1999, Caceres and Barclay 2000, Carter and Feldhamer 2005).
Northern Myotis’ maternity roosts are strongly associated with forest cover, streams, and tree characteristics (e.g., species, height, diameter, age, and decay) (Caceres and Barclay 2000, Broders and Forbes 2004, Broders et al. 2006). Females prefer to roost in tall, large diameter trees in early- to mid-stages of decay (Sasse and Perkins 1996, Caceres and Barclay 2000, Silvis et al. 2015a). Maternity colonies in Newfoundland and Labrador, Nova Scotia, and New Brunswick were generally in larger-than-average trees (Broders and Forbes 2004, Garroway and Broders 2008, Park and Broders 2012). In New Brunswick and Prince Edward Island, female Northern Myotis primarily roosted in trees in the mid-stages of decay within mature, shade-tolerant deciduous stands (Broders and Forbes 2004, Henderson and Broders 2008). Broders and Forbes (2004) attributed this preference to these tree species’ susceptibility to limb breakage and decay (creating available habitat for roosting), long-lived characteristics (permitting repeated use by bats), and their upland habitats with increased solar radiation (reducing energy costs to maintain thebat’s body temperature). Female Northern Myotis are more likely to resort to anthropogenic structures where habitat is fragmented and few potential roost trees exist (Henderson and Broders 2008). In Nova Scotia, Northern Myotis maternity colonies consisted of females with a high degree of maternal relatedness, likely caused by female philopatry (Patriquin et al. 2013). Females switch maternity roost trees approximately every 1-5 days, but roosts are commonly clustered in roosting areas (Sasse and Perkins 1996, Caceres and Barclay 2000, Carter and Feldhamer 2005, Broders et al. 2006, Olson 2011). The largest roosting area recorded in Canada was 300 ha in Alberta (Olson 2011).
Males generally roost alone under raised bark or within cavities of trees in mid-stages of decay (Broders and Forbes 2004). In New Brunswick and Quebec, male Northern Myotis roosted in coniferous or conifer-dominated mixedwood stands (Broders and Forbes 2004, Fabianek et al. 2015). In Quebec, males switched roosts approximately every 2 days (Fabianek et al. 2015).
Tri-colored Bat
Less is known about roosts of Tri-colored Bats. Most roost sites are found within forested habitats, where this species also forages. Tri-colored Bats may roost in clumps of dead foliage and lichens (Veilleux et al. 2003, Perry and Thill 2007, Poissant et al. 2010). In Nova Scotia, 30 radio-tagged bats had roosts in large clumps of arboreal lichens (Usnea spp.) that grew on coniferous or deciduous trees relatively close to water features (Poissant et al. 2010).
Females roost alone or in small colonies. In Nova Scotia, as many as 18 Tri-colored Bats were found in a cluster (Poissant et al. 2010). In more anthropogenically modified landscapes, maternity roosts may be barns or similar human-made structures (Fujita and Kunz 1984). In Nova Scotia, Tri-colored Bats exhibit fidelity to small (<78 ha) roosting areas within and between years (Poissant 2009). In Indiana, females returned to the same area (0.4 ha) each summer and used the same 4-6 trees each year, suggesting value in familiar (and possibly limited) structures (Veilleux and Veilleux 2004).
Males roost individually (Veilleux and Veilleux 2004, Perry and Thill 2007, Poissant 2009). A single male tracked in Nova Scotia roosted alone in arboreal lichen (Usnea trichodea) (Poissant 2009). In Arkansas, males preferentially roosted in dead leaves of oak trees (Quercus spp.) in sites with less canopy cover, more midstory hardwoods, and more overstory large pines than randomly available (Perry and Thill 2007).
Foraging Needs
Little Brown Myotis, Northern Myotis, and Tri-colored Bat are insect predators and will exploit locally abundant patches of prey that may be temporally and spatially scattered. Identification of foraging areas for bats is complicated by sex biases, differences between species, seasonal variations of habitat use by females (e.g., pregnant, lactating, or non-reproducing), and foraging habitat availability and configuration (Henry et al. 2002, Owen et al. 2003, Broders et al. 2006, Randall et al. 2014).
Little Brown Myotis
Little Brown Myotis feed nocturnally on insects (e.g., moths, mayflies, flies, beetles, and caddisflies) and spiders (Moosman et al. 2012, Thomas et al. 2012, Clare et al. 2014). Nevertheless, the diet of Little Brown Myotis can vary significantly based on seasonal, geographic, and environmental factors (Moosman et al. 2012, Clare et al. 2014). On a successful night during peak summer activity, males eat approximately half of their body weight and lactating females may eat their entire body weight in insects (Anthony and Kunz 1977). Peak foraging activity occurs several hours after dusk and often again before sunrise (Fenton 1970, Kunz 1973, Broders et al. 2003). In northern areas (above 60°N) where summer night length is short, pregnant females appeared to alter their foraging behavior by exhibiting only one bout of peak activity. In addition, they foraged for fewer hours than their southerly counterparts though they compensated for reduced time foraging by exhibiting a higher rate of insect capture (Talerico 2008, Reimer 2013).
Foraging Little Brown Myotis are most often associated with open habitats, such as ponds and roads and open canopy (0-50%) forests (Segers and Broders 2014), but have also been recorded gleaningFootnote7 prey within forests (Ratcliffe and Dawson 2003, Jung et al. 2014) and using vegetation along lake and stream margins (Fenton and Barclay 1980). Little Brown Myotis in Yukon boreal habitat travelled 3.8 ± 0.7 km from their daytime roosts to foraging areas, with females travelling significantly farther than males (Randall et al. 2014). In Quebec, lactating females had home ranges 42% smaller (mean: 17.6 ha) than pregnant females (mean: 30.1 ha) (Henry et al. 2002).
Northern Myotis
Northern Myotis feed on insects (e.g., moths, beetles, wasps, and flies) and spiders (Lacki et al. 2009, Dodd et al. 2012, Thomas et al. 2012) that are primarily terrestrial in origin (Broders et al. 2014). Unlike Little Brown Myotis, which most often forage over water and are aerial hawkers, Northern Myotis forage more frequently along and within forests and although they feed on flying insects, they also glean prey (Caceres and Barclay 2000, Ratcliffe and Dawson 2003).
Female Northern Myotis foraged along forest-covered creeks in Prince Edward Island (Henderson and Broders 2008). In West Virginia, female Northern Myotis mainly foraged in 70-90 year-old hardwood stands with road corridors (Owen et al. 2003), and in Kentucky, Northern Myotis were found foraging along ridges and midslopes, rather than lower slopes (Lacki et al. 2009). In an intensively managed forest of West Virginia, the mean home range for lactating or pregnant Northern Myotis was 65 ha (Owen et al. 2003). In New Brunswick, Broders et al. (2006) found males and females travel significantly different distances between roost sites and foraging areas. The distance travelled by females between successive roosts was twice as far as males on average (457 m vs. 158 m) (Broders et al. 2006). The authors suggested that females travelled farther because suitable maternity sites were located in poor foraging habitat (Broders et al. 2006).
Tri-colored Bat
Similar to Little Brown Myotis, Tri-colored Bats feed on insects (e.g., flies, beetles, wasps, and moths) after dusk and before dawn using echolocation (Fujita and Kunz 1984, Naughton 2012). Each night, males consume at least half of their body weight in insects and pregnant and nursing females may eat more than their body weight (Naughton 2012).
Foraging predominately occurs in forested riparian areas, over water (e.g., ponds and rivers), and in relatively open areas (Ethier and Fahrig 2011). In Nova Scotia, Farrow and Broders (2011) found Tri-colored Bats foraging at river sites, but found more activity in areas with greater forest cover at a landscape scale, suggesting that this species may avoid landscapes that are cleared for agriculture, urban development, and forest harvesting. The distances between roost sites and foraging areas are generally unknown, but in some locations may be up to 5 km (Quinn and Broders 2007).
Migration
Little Brown Myotis, Northern Myotis, and Tri-colored Bat are considered short-distance migrants, radiating annually from overwintering areas to summering areas in any direction (Fraser et al. 2012, COSEWIC 2013). In Manitoba and Ontario, Little Brown Myotis migrated regionally 35 to 554 km (median 463 km) (Fenton 1970, Dubois and Monson 2007, Norquay et al. 2013). Migratory movements by Northern Myotis are not well understood, but are likely similar to the Little Brown Myotis. Tri-colored Bat have been recorded moving 53-780 km (Griffin 1940, COSEWIC 2013). In addition, Fraser et al. (2012) found that some Tri-colored Bats engage in annual latitudinal migrations, especially those at the northern extent of the range, which may be related to their need to keep warm since they often hibernate individually (COSEWIC 2013). This is further supported by Thorne (2015), who found increased detections of Tri-colored Bats later in the season (i.e., August – September) on islands in the Great Lakes of Ontario.
As noted above, swarming sites may serve as migratory stopover locations (Fenton 1969) and are likely used annually (Rydell et al. 2014). When travelling over large waterbodies, peninsulas and islands may function as stopover sites (Dzal et al. 2009, Thorne 2015). For example, Tri-colored Bats may use Amherst Island, Lake Ontario and Long Point, Lake Erie for migration and stopover (Dzal et al. 2009, Thorne 2015).
Limiting Factors
All three species are long-lived and females produce only one (Little Brown Myotis and Northern Myotis) or two (Tri-colored Bat) young annually. Such life-history traits heighten the vulnerability of these bat populations to increases in adult mortality rates. In addition, yearling survival is low (0.23 to 0.46) (Frick et al. 2010b). In a recent pre-WNS study from New Hampshire, the annual population growth rate of Little Brown Myotis over 16 years was estimated to be 1.008 (Frick et al. 2010b). In 22 subpopulations in the northeastern U.S., the population growth rate was estimated to be 0.98-1.2 (Frick et al. 2010a). Similarly, the population growth rates of Northern Myotis and Tri-colored Bat were estimated to be 1.03 and 1.04, respectively (Langwig et al. 2012). Predicted population growth rates for Little Brown Myotis in the northeastern U.S. post-WNS was 0.95 (Maslo et al. 2015).
These species are socially gregarious which increases their susceptibility to the spread of pathogens (such as WNS) (Langwig et al. 2012). It is unknown colonial and social nature of these species creates subpopulation or colony size thresholds below which the survival or reproductive success of individuals decline and /or the population will be unable to recover.
It is also unknown if available overwintering sites with suitable microclimatic conditions are limiting in Canada.
4. Threats
4.1. Threat Assessment
In areas where local bat populations have significantly declined as a result of WNS, the relative magnitudes of other threats increase because the mortality of a small number of the remaining individuals (particularly adults) has the ability to impact the survival of local populations, their recovery, and, perhaps, the development of resistance to the fungus that causes WNS. Therefore, the level of concern and severity of the impact on bat populations from threats other than WNS will generally be elevated in areas of Canada affected by WNS, compared to areas that have not yet been affected by WNS. These dissimilarities in the threat assessments are identified in Table 2.
| Threat Category | Threat | Area | Level of ConcernNoteaof Table 2 | Extent | Occurrence | Frequency | SeverityNotebof Table 2 | Causal CertaintyNotecof Table 2 |
|---|---|---|---|---|---|---|---|---|
| Exotic, Invasive, or Introduced Species/Genome | White-nose syndrome (Pseudogymnoascus destructans) | WNS-affected areas | Very High | Widespread | Current | Continuous | High | High |
| Exotic, Invasive, or Introduced Species/Genome | White-nose syndrome (Pseudogymnoascus destructans) | Areas not yet affected by WNS | Very High | Widespread | Imminent | Continuous | High | High |
| Exotic, Invasive, or Introduced Species/Genome | Feral and free-roaming cats | - | Unknown | Localized | Current | Continuous | Unknown | Low |
| Habitat Loss or Degradation | Destruction or degradation of hibernacula | WNS-affected areas | High | Localized | Current | Continuous | High | High |
| Habitat Loss or Degradation | Destruction or degradation of hibernacula | Non-WNS affected areas | High | Localized | Current | Continuous | High | High |
| Habitat Loss or Degradation | Destruction or degradation of roosts | WNS-affected areas | High | Localized | Current | Continuous | High | High |
| Habitat Loss or Degradation | Destruction or degradation of roosts | Non-WNS affected areas | Medium | Localized | Current | Continuous | Moderate | High |
| Habitat Loss or Degradation | Destruction, degradation, or conversion of foraging habitats | WNS-affected areas | Medium | Widespread | Current | Continuous | Unknown | Medium |
| Habitat Loss or Degradation | Destruction, degradation, or conversion of foraging habitats | Non-WNS affected areas | Medium-Low | Widespread | Current | Continuous | Unknown | Medium |
| Disturbance or Harm | Collisions with or barotrauma from wind turbinesNotedof Table 2 | WNS-affected areas | High | Localized | Current | Seasonal (spring, summer, fall) | High | High |
| Disturbance or Harm | Collisions with or barotrauma from wind turbinesNotedof Table 2 | Non-WNS affected areas | Medium | Localized | Current | Seasonal (spring, summer, fall) | Moderate-Low | Medium |
| Disturbance or Harm | Intentional harm to individuals | WNS-affected areas | High | Localized | Current | Continuous | High | High |
| Disturbance or Harm | Intentional harm to individuals | Non-WNS affected areas | Medium-Low | Localized | Current | Continuous | Moderate-Low | Medium |
| Disturbance or Harm | Recreational or scientific disturbance of individuals | WNS-affected areas | Medium-High | Localized | Current | Continuous | High | Medium |
| Disturbance or Harm | Recreational or scientific disturbance of individuals | Non-WNS affected areas | Medium | Localized | Current | Continuous | Moderate | Medium |
| Disturbance or Harm | Industrial disturbance of individuals (e.g., mining and forestry practices) | WNS-affected areas | Medium-Low | Localized | Current | Continuous | Moderate | Low |
| Disturbance or Harm | Industrial disturbance of individuals (e.g., mining and forestry practices) | Non-WNS affected areas | Low | Localized | Current | Continuous | Low | Low |
| Pollution | Mercury | - | Unknown | Widespread (Eastern Canada) | Current | Continuous | Unknown | Low |
| Pollution | Other toxic chemicals | - | Unknown | Widespread | Current | Continuous | Unknown | Low |
| Pollution | Light pollution | - | Unknown | Widespread | Current | Continuous | Unknown | Low |
| Climate and Natural Disasters | Habitat or prey dynamic alterations resulting from climate change | - | Unknown | Widespread | Current | Continuous | Unknown | Low |
| Accidental Mortality | Collisions with vehicles | - | Unknown | Widespread | Current | Continuous | Unknown | Low |
4.2. Description of Threats
Threats are listed here in the order in which they are presented in Table 2.
White-nose Syndrome (Pseudogymnoascus destructans)
White-nose syndrome (WNS), caused by the dermatophyteFootnote8 fungus Pseudogymnoascus destructans (formerly called Geomyces destructans), is the greatest threat to the survival and recovery of Little Brown Myotis, Northern Myotis, and Tri-colored Bat. Most of the known hibernacula in the northeastern U.S. and eastern Canada (except in Newfoundland and Labrador) have experienced massive declines resulting from WNS (U.S. Fish and Wildlife Service 2015).
The fungus is believed to have originated in Europe (Lindner et al. 2011, Pikula et al. 2012, Ren et al. 2012, Warnecke et al. 2012, Leopardi et al. 2015) and was first detected in the U.S. in 2006 (Lorch et al. 2011) and in Canada in 2010 (U.S. Fish and Wildlife Service 2015). The fungus grows in the same microclimate conditions that occur within the hibernacula where the three species of bats overwinter. The fungus colonizes the bat’s skin, causes erosions of the epidermis, and damages sweat glands, oil-producing glands, muscles, connective tissue, blood vessels, and hair follicles (Meteyer et al. 2009, Cryan et al. 2010). The wings and ears develop white-grey blotches on their surfaces and the muzzle often turns fuzzy white.
The early stages of WNS may not be visible in all affected individuals. WNS is characterized by an elevated metabolic rate associated with the epidermal fungal growth, stimulating hyperventilation, and results in increased arousals from torpor that contribute to dehydration and electrolyte loss (Warnecke et al. 2012, Warnecke et al. 2013, Verant et al. 2014). Later stages of WNS are associated with more extensive and severe tissue lesions, further increasing arousal frequency, water loss, and energy use (Warnecke et al. 2012, Warnecke et al. 2013, Verant et al. 2014). Energy reserve depletion is accelerated by reduced torpor bout lengths and through acute physiological changes as WNS pathology progresses, eventually leading to mortality (Frank et al. 2014, Verant et al. 2014). WNS-infected bats are more likely to fly (and fly erratically) during winter (Carr et al. 2014). Bats that survive until spring may have damaged wings with numerous holes, and may exhibit signs of physiological stress, and reduced reproductive success (Reeder and Turner 2008, Meteyer et al. 2009, Reichard and Kunz 2009, Powers et al. 2012). However, almost all of the identified mortality associated with WNS has been during hibernation when the immune functions of bats are reduced (Cryan et al. 2010). Additionally, Fuller et al. (2011) tracked individual Little Brown Myotis and found wing damage caused by WNS healed to some degree throughout the summer. Prevalence decreases during the summer months, likely as a result of body temperatures above that required for P. destructans (Langwig et al. 2015b).
In eastern Canada and the northeastern U.S., WNS mortality rates are typically low (i.e., 20%) in the first year of detection, followed by high levels (i.e., >70%) within two years (Frick et al. 2010a). At known hibernacula in eastern Canada, the number of hibernating Little Brown Myotis and Northern Myotis bats has declined by an estimated 94% (COSEWIC 2013). The number of Tri-colored Bats has also shown a precipitous decline of approximately 75% at known hibernacula (Turner et al. 2011, COSEWIC 2013).
Rate of spread
From the epicenter in Albany, New York, the rate of spread of WNS in Canada has been approximately 200 to 250 km per year (COSEWIC 2013). As of 29 May 2015, WNS was recorded in 26 states and 5 provinces; the presence of P. destructans was confirmed in 2 additional states (U.S. Fish and Wildlife Service 2015) (Figure 1; Table 1). This represents approximately 17% of the Canadian range of Little Brown Myotis, 28% of Northern Myotis and 100% of Tri-colored Bat that have been impacted by WNS (Figure 1). Additional sites are being detected each year in Canada, with the most recent (i.e., 2015) detection of P. destructans in northwestern Ontario, near Atikokan (U.S. Fish and Wildlife Service 2015). It is uncertain if WNS has spread to Labrador, as well as the northern-most parts of Ontario and Quebec because there is limited information related to the hibernacula in these regions. To date, testing of hibernacula on the island of Newfoundland has shown negative results (S. Pardy Moores, pers. comm. 2015). The Côte-Nord region of Quebec is also currently WNS-free.
Transmission
Transmission occurs as a result of bat-to-bat contact and contact with contaminated hibernacula, as well as human-assisted mechanisms (e.g., tourists, spelunkers, and researchers that do not follow proper decontamination protocols) (Lorch et al. 2011, Lorch et al. 2013). The amount of physical contact among hibernating bats varies by species, and does not correlate well with infection rates (Kilpatrick 2013, Langwig et al. 2015b). Transmission of P. destructans does not appear to be associated with winter colony sizes or influx of susceptible individuals after the mating season (Langwig et al. 2015b).
Pseudogymnoascus destructans spores in the soil and on the walls of hibernacula may remain viable for years, if not decades, potentially impeding recovery of these species (Langwig et al. 2012, Hoyt et al. 2014).
Infection of the remaining Canadian range of Little Brown Myotis and Northern Myotis
If WNS continues to spread at its current rate, all hibernacula in Canada and the U.S. will be infected by 2025 to 2028 (Maher et al. 2012, COSEWIC 2013, O'Regan et al. 2014).
In Canada, as WNS approaches less forested regions of southwestern Manitoba, the relative dryness and few trees in the Prairies suggests that transmission may occur at a slower rate. But it is possible that WNS may spread westward using alternative routes: from the south and /or from forested regions north of the Prairies. Moreover, despite some evidence that the Rocky Mountains historically restricted bat gene flow between eastern and western areas, mixing exists and therefore it is unlikely that the mountains will be a physical barrier that prevents WNS from reaching the western coastline (Russell et al. 2012).
Samples of Little Brown Myotis collected from insular-Newfoundland exhibited genetic differentiation and less genetic connectivity when compared to other samples collected in eastern Canada, indicating few movements of bats between Newfoundland and other areas (McLeod et al. 2015). This suggests that the probability of transmission is lower to the island than it is between other sampling sites. Nevertheless, bats from other regions separated by similarly-wide oceanic straits are not genetically isolated and therefore bat-assisted spread to Newfoundland remains possible (McLeod et al. 2015).
WNS may reach WNS-free populations faster than would be expected from bat-to-bat transmission because of human-assisted transmission of the P. destructans spores. Pseudogymnoascus destructans was probably brought to North America on the clothing of tourists who had visited caves in Europe (Okoniewski et al. 2010). People who visit multiple caves without decontaminating their clothing or gear substantially increase the risk that P. destructans will be transmitted to WNS-free hibernacula. In addition, bats can be inadvertently transported. For example, there are reports of bats being transported to British Columbia within the cargo hold of a ship and in the awnings of camper vans (P. Govindarajulu, pers. comm.). Such incidences have the ability to greatly increase the rate at which WNS spreads.
Resistance and treatment
A small percentage of individuals may have a genetically-based resistance or immunity to the effects of P. destructans that would be passed to their offspring. In Central Europe, P. destructans has been recorded in approximately 63% of sampled hibernacula, and on several bat species, yet mortality due to WNS has not been observed suggesting that populations of European species may be resistant to, or tolerant of, WNS (Wibbelt et al. 2010, Horacek et al. 2012). Reichard et al. (2014) found a small number (113 / 2095 banded individuals) of Little Brown Myotis in New England survived 1 to 6 winters since the arrival of WNS and some showed signs of reproductive success. Researchers in Ontario have also documented small numbers of Little Brown Myotis surviving WNS infection and reproducing (D. Morningstar, pers. comm.).
Substantial research is ongoing into the ultimate causes, treatment, and mitigation of WNS. The major molecular component responsible for the effects of P. destructans has been identified and could represent a target for WNS intervention (O’Donoghue et al. 2015). Promising new research has also isolated an enzyme that naturally occurs on the skin of bats that appears to inhibit the invasion of tissue by P. destructans in laboratory tests (Hoyt et al. 2015). Additionally, research has demonstrated that substances produced by the soil bacterium Rhodococcus rhodochrous and yeast Candida albicans may have potential as biological control agents of P. destructans (Cornelison et al. 2014, Raudabaugh and Miller 2015).
Feral and Free-roaming Cats
Domestic and feral cats are known to prey upon a substantial number of birds (Calvert et al. 2013), small mammals (Loss et al. 2013), reptiles, and amphibians (Loyd et al. 2013). Ancillotto et al. (2013) suggested that cats may be a significant threat to bats. The Community Bat Programs of British Columbia regularly receives calls and anecdotal reports of cats killing bats in British Columbia (J. Craig, pers. com.). Species that roost within anthropogenic structures (e.g., barns), such as Little Brown Myotis, are likely more susceptible to this threat because of their potential close proximity to cats. In Italy, adult female bats in rural or sparsely urban areas were most likely to be preyed upon by cats (Ancillotto et al. 2013). In Yukon, cats tend to kill juvenile Little Brown Myotis that have recently become capable of flying (T. Jung, pers. com.).
Destruction or Degradation of Hibernacula
Little Brown Myotis, Northern Myotis, and Tri-colored Bat require specific microclimatic conditions throughout hibernation. Therefore, any activity that results in a hibernaculum being inaccessible to bats or alters a hibernaculum’s temperature, humidity, airflow, or other microclimatic characteristics may destroy or degrade the habitat.
The practice of sealing the entrances of mines, caves, and dug wells has become increasingly common during the past few decades, likely because of health and safety concerns and potential associated liabilities. If the structures are being used as hibernacula, sealing the entrance can represent a potentially significant source of habitat loss. For example, when a hibernaculum in Kentucky was blocked by a new gift shop, thousands of Indiana Bats (Myotis sodalis) clung to the walls of the building, rather than search for an alternative hibernaculum (Murphy 1987). Even blockages of small entrances can alter airflow patterns and change internal temperatures (U.S. Fish and Wildlife Service 2007).
Industrial activities, such as quarrying, mining exploration and development, agriculture, dam construction, and forestry outside of hibernacula can also cause, exacerbate, or accelerate blockages of airflow, create changes to hydrology or microclimatic conditions, potentially cause flooding, or directly degrade the habitat (McAlpine 1983, U.S. Fish and Wildlife Service 2007). Also, mine or cave entrances may collapse if heavy machinery (e.g., timber harvesting equipment or mining equipment) is used near weak areas of the hibernaculum (McAlpine 1983, U.S. Fish and Wildlife Service 2007). Once bats are blocked from entry, the hibernaculum can no longer be used. Rehabilitation activities at old mine sites can also affect hibernacula and the bats using them. While removing a head frame during rehabilitation activities at an old mine in Ontario during January 2011, 50-60 bats were uncovered, flew away, and subsequently died (P. Davis, pers. comm.). Conversely, the decommissioning of active or semi-active mines (e.g., shutting off water pumps) can result in flooding of hibernacula. Mining companies may reactivate previously abandoned mines for extraction purposes as a result of fluctuations in mineral prices, but there is little information on the frequency of this practice in Canada. The prevalence is assumed to be low, but may be a concern for particular areas of the country.
Modifications for tourists (e.g., observation platforms), intentional vandalism, or the erection of physical barriers (e.g., gates) can cause restricted or altered airflow or modify other microclimatic characteristics of the hibernaculum (U.S. Fish and Wildlife Service 2007). Gates are often considered the most efficient and effective technique to control human access to hibernacula, but even so-called bat-friendly gating may cause bats to avoid the hibernaculum, collide with the gate, or cause significant changes in bat behaviour (Spanjer and Fenton 2005, U.S. Fish and Wildlife Service 2007, Derusseau and Huntly 2012, Diamond and Diamond 2014). Gating should be completed using well-supported designs in conjunction with bat monitoring to ensure no negative impacts to bats.
Destruction or Degradation of Roosts
Of the three species of bats, Little Brown Myotis most regularly uses buildings and bat boxes for maternity colonies. The number of bat colonies in buildings may be declining as a result of limited available structures and attempts by landowners to exclude bats (Kunz and Reynolds 2003). Because females tend to show a relatively high degree of fidelity, excluding bats from previously occupied maternity roosts in anthropogenic structures would be considered habitat loss. Similarly, habitat loss would occur if a previously occupied bat box is removed from a site.
Any type of development activity that results in the removal of trees or forested landscapes (e.g., land development, geological, exploration, and oil and gas wells) has the potential to destroy or degrade roosts for the three bat species. For example, forestry and timber harvesting operations (e.g., salvage logging) may remove tracts of mature forests, as well as individual snags that may be used by male and female bats for roosting.
The effects of tree removal or roost exclusion may depend on the availability of other suitable habitat, timing, bat species, bat sex, and other factors. Roost exclusions may lower reproductive success, alter home range size, change mean colony size, and decrease site fidelity (Brigham and Fenton 1986, Neilson and Fenton 1994, Borkin et al. 2011, Chaverri and Kunz 2011). Little Brown Myotis may abandon roosting areas after being excluded from roost sites (Neilson and Fenton 1994). However, depending on habitat availability, bats may use another tree for roosting if a previous roosting tree is removed outside the breeding season (Silvis et al. 2015b). For Northern Myotis roosting in Kentucky, the number of roosts, roost site characteristics, and overall space used did not change after single highly-used roosts and 24% of secondary roosts were experimentally removed prior to roosting (Silvis et al. 2015b). However, the distances bats moved between sequential roosts doubled within areas where secondary roosts were removed (Silvis et al. 2015b). Nevertheless, Silvis et al. (2015b) noted that tolerance limits of roost loss may be influenced by local forest conditions and the social / behavioral characteristics of the species using the roost.
Destruction, Degradation, or Conversion of Foraging Habitat
The removal, degradation, and fragmentation of foraging habitat (e.g., forests, wetlands and riparian areas) can be caused by a variety of anthropogenic sources, including (but not limited to) forestry practices, agriculture, oil and gas development, mining exploration and development, and urban and residential development. Harvest rates associated with forestry practices in Canada are highest in Quebec, British Columbia, and Ontario. The harvest rates were relatively stable in Canada from the 1980s to 2008 (Masek et al. 2011), but have been lower since 2008 (NFD 2014). Land conversion has been intensive in some portions of these species ranges. For example, 73% of the boreal hardwood transition zone in Saskatchewan has been converted to agriculture, with 25% lost between 1966 and 1994 (Hobson et al. 2002). Young et al. (2006) calculated an annual rate of change in forest cover along the southern boreal edge of Alberta to be -0.82% per year. Wetland loss in Canada is estimated at approximately 70% within settled areas, with draining for agriculture accounting for the majority (85%) of known conversions (Haak 2008).
In general, many bat species avoid large clearcuts and open areas (e.g., Henderson and Broders 2008), but forested and vegetated edges may provide foraging opportunities for some bats (Krusic et al. 1996, Grindal and Brigham 1998). These edges may provide protection from predators and wind and/or concentrate prey (Swystun et al. 2001, Henderson and Broders 2008). Little Brown Myotis, Northern Myotis, and Tri-colored Bat are all associated with forests or trees for at least some portion of their annual cycles and therefore are likely affected by activities that result in forest fragmentation, degradation, and removal. However, research findings related to potential effects vary in relation to species, amount of available habitat, cause of forest degradation, spatial scale examined, and availability of insects (Grindal and Brigham 1999, Swystun et al. 2001, Henderson et al. 2008, Ethier and Fahrig 2011, Segers and Broders 2014, Pauli et al. 2015).
Forest fragmentation can create linear elements beneficial for Little Brown Myotis, but may decrease the availability of habitat for Northern Myotis (Broders and Forbes 2004, Broders et al. 2006, Segers and Broders 2014). Conversely, Ethier and Fahrig (2011) found that forest fragmentation (independent of forest amount) was positive for both Little Brown Myotis and Northern Myotis (Ethier and Fahrig 2011). In Alberta, Little Brown Myotis and Northern Myotis were active along forest edges and residual patches, but activity was significantly reduced in the center of cutblocks (Hogberg et al. 2002). In Prince Edward Island, Northern Myotis exhibited a significant preference for commuting and foraging under forest cover compared to open areas (Henderson and Broders 2008). In Quebec, Myotis spp. and Tri-colored Bat were most active in green spaces within the least-fragmented forested areas (Fabianek et al. 2011). In Ontario, the abundance of Tri-colored Bat was negatively related to the amount of forest cover (Ethier and Fahrig 2011), but in Nova Scotia, Tri-colored Bats were less active in forest-cleared landscapes (e.g., cleared for agriculture, settlements, and timber production) (Farrow and Broders 2011). Farrow and Broders (2011) concluded that Tri-colored Bat is a forest-associated species negatively impacted by practices that reduce the extent of forests on a landscape scale.
In agriculturally-dominated landscapes, some species follow linear forest features for commuting and foraging (Henderson and Broders 2008). Northern Myotis traveled following a hedgerow of trees in a agriculturally dominated landscape in Prince Edward Island (Henderson and Broders 2008). Myotis spp. and Tri-colored Bat were not directly observed using open areas (such as cultivated and fallow fields and golf courses) in Quebec, but were active in the wooded areas adjoining these features (Fabianek et al. 2011). Agricultural intensification that removes hedgerows and field margins could be reducing foraging and commuting habitat (Wickramasinghe et al. 2003).
Wetlands and areas around waterbodies (e.g., riparian areas and forest edges) are important foraging habitat for Little Brown Myotis, Northern Myotis and Tri-colored Bat. Activities that degrade or remove wetlands have the potential to have negative impacts to foraging habitat availability and quality. Wetland loss in southern Ontario, where all three species occur, has been extensive and continues (additional losses of 3.5% between 1982 and 2002) (Federal Provincial and Territorial Governments of Canada 2010). Within the range of Little Brown Myotis, estimates of wetland losses in the Prairie Pothole region of Canada vary between 40 to 71% (Federal Provincial and Territorial Governments of Canada 2010). Wetland loss and degradation continues within this region mainly due to agricultural intensification.
Roads can also act as barriers to bats by restricting movements and changing habitat use (Abbott et al. 2012, Bennett and Zurcher 2013, Kitzes and Merenlender 2014, Abbott et al. 2015). Bats are less likely to fly across a road as traffic noise increases (Bennett and Zurcher 2013). Traffic noise may also reduce flight activity and foraging efficiency by interfering with echolocation and hearing (Abbott et al. 2015).
Collisions with or Barotrauma from Wind Turbines
Bats can be killed either through direct collisions with turbine blades (Horn et al. 2008) or barotrauma caused by the sudden drop in air pressure behind the blades (Baerwald et al. 2008, Grodsky et al. 2011, Rollins et al. 2012). Wind turbines represent one of the largest sources of anthropogenic mortality for bats (Cryan and Brown 2007, Cryan 2011). Results from mortality studies at various sites in the U.S. and Europe suggest that annual bat mortality ranges from 0 to over 50 deaths per turbine, but data collection protocols, experimental design, and analysis methods varied substantially among wind farms (Kunz et al. 2007, Arnett et al. 2008, Cryan 2011, Hayes 2013, Smallwood 2013). Kunz et al. (2007) predicted that by 2020 as many as 110,000 bats may be killed annually in the U.S. based on an average of 2.3 bats / turbine / year. Cryan (2011) suggested that this may be a considerable underestimate, and provided an estimate of 450,000 bats annually based on an average published mortality rate of 11.6 bats / megawatts / year. Smallwood (2013) estimated approximately 888,000 bats killed each year.
Relative to long-distance migrants, such as Hoary Bat (Lasiurus cinereus), Eastern Red Bat (Lasiurus borealis), and Silver-haired Bat (Lasionycteris noctivagans), Myotis species are killed by turbines at lower rates (e.g., 0 to 13% of fatalities) (Arnett et al. 2008). This is likely because they migrate shorter distances and generally fly below turbine height during summer (Reynolds 2006).
In Canada, it is estimated that 15.5 ± 3.8 bats per turbine are killed annually (Zimmerling and Francis, unpublished data). Based on 4019 installed turbines (the number installed in Canada as of December 2013), an estimated 52,100 bats (95% C.I. Footnote928,690 - 75,316) are killed by wind turbines annually (Zimmerling and Francis, unpublished data). In Canada, using data pre- and post-WNS, Little Brown Myotis accounted for 13% of all bat mortalities from wind turbines (approximately 7,000 individuals), with most (88%) mortality occurring in Ontario (Zimmerling and Francis, unpublished data). At one wind power facility in Ontario, bat mortality was dominated by Little Brown Myotis; this species accounted for 46% of all bat mortality in July and 38% over the period of April to September (OMNRF, unpublished data). Northern Myotis account for slightly less than 1% of all mortality from wind turbines (approximately 440 individuals) in Canada (Zimmerling and Francis, unpublished data). At a wind power facility in British Columbia, Little Brown Myotis and Northern Myotis comprised 44% of all bat mortalities recorded with most fatalities occurring in July and August (Hemmera 2011). At some wind facilities in the eastern U.S., Tri-colored Bats accounted for as many as 25.4% of fatalities (Arnett et al. 2008), whereas in Canada, fewer than five carcasses (i.e., less than 0.1% of all carcasses) have been found during carcass searches (Zimmerling and Francis, unpublished data). It is unknown if these species are less vulnerable to impacts from wind turbines because of differences in flight, foraging behavior, or habitat, or if they simply have smaller populations and are therefore generally uncommon around wind farms. Nevertheless, even low rates of mortality have the potential to be biologically significant for relatively rare species; it is possible that future wind farms, if accidentally located near important concentration areas such as maternity roosts or hibernacula, could cause high mortality.
Mortality rate is anticipated to increase as the number of turbines increases. In WNS affected areas, the biological significance of any mortality could increase considerably. The mortality of a few individuals has the potential to impact the ability of local populations to recover and develop resistance to the fungus.
Presently, mitigation measures to reduce bat mortality related to wind turbine development may include the feathering of wind turbine blades or increasing the cut-in speed when the risk to bats is particularly high (e.g., at night during peak migration) (Baerwald et al. 2009). Baerwald et al. (2009) demonstrated that these mitigation techniques reduced bat fatalities by approximately 60% at a site in southwestern Alberta. Arnett et al. (2009, 2013b) found that increasing the speed at which the turbine starts to rotate and generate power reduced bat mortalities by approximately 73% (range 44% to 93%) at a wind farm in Pennsylvania with a marginal (~1%) loss in annual power. The use of ultrasonic broadcasts may also reduce bat fatalities at wind turbines by deterring bats from approaching the sound source (Arnett et al. 2013a). In some circumstances, operational mitigation techniques may include the periodic shutdown of select turbines during the highest risk periods.
Intentional Harm to Individuals
Some species of bats, including Little Brown Myotis, often use anthropogenic structures as maternity roosts or hibernacula. Noise, the accumulation of feces (guano), and fears of contracting histoplasmosisFootnote10 and rabies may cause maternity colonies of bats to be exterminated. Few data are available to determine the prevalence and impact of maternity colony removals; most data are anecdotal because not all jurisdictions have reporting requirements for nuisance wildlife control companies (COSEWIC 2013). Sealing the entrance(s) of an occupied maternity roost will most likely result in the death of all individuals inside the roost site. Some maternity colonies may contain most of the breeding females and offspring within a large area, so colony removal can have a significant impact on local populations.
Intentional harm to individuals within hibernacula has also been reported. For example, all bats (~800 Little Brown Myotis and Northern Myotis) were removed for incineration from the only known hibernacula of Northern Myotis on Prince Edward Island in 1989 (Brown et al. 2007). Although the bats were rescued from incineration, attempts to keep them over winter failed and they all perished (Brown et al. 2007).
Some provinces and territories have taken measures to reduce the risk of intentional harm. For example, New Brunswick Department of Natural Resources has removed these species from the list of nuisance wildlife under the New Brunswick Fish and Wildlife Act, thereby providing support for non-lethal alternatives in their management. See section 2: Species Status Information for more legislation protecting individuals.
Recreational or Scientific Disturbance of Individuals
Visitation by people (Thomas 1995) or handling of hibernating bats can result in arousal from torpor (Speakman et al. 1991). When in deep torpor, bats are generally unaffected by ambient noise (Harrison 1965), but some individuals may respond to noise and light, arouse from torpor, and begin to fly (Thomas 1995). These individuals can then cause a cascade of arousals in nearby bats, resulting from their tactile activities (e.g., attempted copulation, rejoining the cluster of bats) (Thomas 1995). Even non-intrusive visitations can cause severe fat consumption (premature energy depletion), starvation, reduced energy reserves for reproduction, and death (Gaisler et al. 1981, Boyles and Brack 2009, Olson et al. 2011). The population of Little Brown Myotis significantly increased in an Alberta cave after winter and autumn access was restricted to reduce disturbance during hibernation and swarming (Olson et al. 2011). Disturbance tolerance is related to the length of winter and number and rate of visits; repeated visits over several consecutive days have the most severe impacts (Boyles and Brack 2009). Because bats with WNS have more frequent arousal episodes (Reeder et al. 2012), the additive effect of human-caused arousals within WNS-affected hibernacula may be significant.
Tourists, spelunkers, recreational users, and researchers are the main visitors to hibernacula. Visitation in the summer (when most occur) likely has less direct impact on bats because the site is not being used or bats can replenish fat reserves. To minimize visitation, year-round gates have been installed at some hibernacula, spelunking societies have posted guidelines (e.g., Manitoba Speleological Society) (SSM 2015), and bat researchers have minimized the number and duration of their visits.
Activities in a maternity roost may be a concern if the activities disturb bats, or result in the bats abandoning the roost and relocating to other sites. Handling bats for research may also have energetic or fitness consequences, however, data on the extent of such activities are unavailable and the significance of this activity is unknown, but assumed to be small.
Industrial Disturbance of Individuals
Mining exploration activities, forestry operations, or other industrial activities may threaten bats if the activities cause noise, light, and vibrations near hibernacula that disturb hibernating bats and cause them to arouse from torpor. For example, during the fall of 2008, a mineral exploration company conducting exploration activities in an old mine adit in Ontario woke numerous bats from torpor (Anonymous 2008).
Similarly, noise and vibrations in areas where maternity colonies are found may result in reduced reproductive success, roost abandonment, and relocation to other sites (McCracken 2011). Since bats rely on echolocation or prey-generated sounds to forage, anthropogenic noise could also interfere with foraging and affect prey detection (Bunkley et al. 2015). However, a study of compressor stations associated with natural gas extraction in New Mexico revealed no significant difference in the activity level of Little Brown Myotis at loud compressor sites compared to quieter well pads (Bunkley et al. 2015).
Mercury
Mercury is a naturally occurring element that is enriched in the environment by human activities. Long-range atmospheric transport and deposition is the dominant source of mercury to many aquatic habitats over much of the landscape (Fitzgerald et al. 1998, U.S. Geological Survey 2000). Bio-available mercury is also mobilized within watersheds by forestry activities, hydroelectric reservoir creation, and various industrial-related activities (Porvari et al. 2003, Vuori et al. 2003, Wiener et al. 2003). Mercury concentrations in aquatic food webs are usually correlated with low pH levels, and as a result mercury concentrations increase from west to east across Canada, along with pH levels, in freshwater food webs (Depew et al. 2013).
Bat species appear to be particularly susceptible to heavy metal accumulation because most species are long-lived, occupy high trophic levels, feed on aquatic emergent insects, and sustain high metabolic rates and food intake. Recent studies in the northeastern U.S. found significantly higher accumulation of mercury in blood and fur samples in Little Brown Myotis and Northern Myotis in all age classes near WNS contaminated sites than in non-contaminated locations (Karouna-Renier et al. 2014, Yates et al. 2014). However, Karouna-Renier et al. (2014) did not find significant differences in the genotoxic effects (i.e., DNA damage) of mercury between contaminated and non-contaminated sites. Mercury concentrations in Little Brown Myotis sampled across Nova Scotia varied among colonies in relation to nearby lake acidity, and 48% of the individuals sampled had concentrations in excess of a threshold associated with neurochemical changes in other bat species from Virginia (Little et al. 2015b). Of 344 Little Brown Myotis individuals sampled from maternity roosts across Atlantic Canada (Nova Scotia, Prince Edward Island, and Newfoundland and Labrador), 37% had concentrations exceeding the neurochemical threshold (Little et al. 2015a). These recent studies raise concerns regarding the effects of mercury and other environmental contaminants on reproductive success, physiological responses (e.g., immune system responses), and survival.
Other Toxic Chemicals
Reported sub-lethal effects of chemical contaminants in bats include impairment of flight and foraging ability, resulting in higher predation risk and lower energy accumulation, immunosuppression, reduced reproductive success, and change in metabolic activity (Clark and Lamont 1976, Eidels et al. 2007, Kannan et al. 2010). The effect of sub-lethal contamination on the resulting susceptibility of bats to WNS is still unclear, but high concentration of organic contaminants in Little Brown Myotis fat tissues were found both in healthy populations and populations affected by WNS (Kannan et al. 2010).
Pesticide spraying in agricultural or forested landscapes has the potential to reduce the abundance of insects on which bats feed. Widespread and/or continuous application of pesticides (such as that which might occur for Spruce Budworm - Choristoneura fumiferana, Mountain Pine Beetle - Dendroctonus ponderosae, or on agricultural landscapes) could potentially have substantial impacts on food availability and physiology. Even at local scales in the United Kingdom, bat activity was significantly higher in aquatic habitats of organic farms versus conventional farms, suggesting greater prey availability in areas with lower levels of agrochemical use (Wickramasinghe et al. 2003).
Neonicotinoid insecticides were introduced in the 1990s, are currently the most widely used class of insecticides globally, and their use is continuing to increase (Sparks 2013, Douglas and Tooker 2015). They are generally used on agricultural lands, but have been detected in wetlands (Main et al. 2014) and watercourses in Canada (Environment Canada 2011, Xing et al. 2013) and are frequently found at levels that exceed water quality guidelines (Morrissey et al. 2015). Neonicotinoids adversely affect insect populations (and therefore potential prey of bats) (Goulson 2013); some of the most important prey (flies, caddisflies, and mayflies) of Little Brown Myotis, Northern Myotis, and Tri-colored Bats are among the most sensitive aquatic insects to neonicotinoids (Morrissey et al. 2015). Reduced prey availability could potentially result in increased time spent foraging, less fat stores and/or increased time spent in torpor, ultimately leading to poor body condition and resulting in reduced reproductive and/or survival rates (Talerico 2008, Reimer 2013). In addition to reduced prey populations, neonicotinoids also cause direct sub-lethal effects on the reproductive success, development, immune function, and growth in numerous vertebrates (Gibbons et al. 2015). Mason et al. (2013) hypothesized that the thousands of invertebrates consumed by bats would inevitably expose bats to small cumulative doses of these toxins. To date, no research has explored the direct or indirect effects of neonicotinoids on bats.
Increased incidence of seasonal algal blooms may also pose a threat to these species. Algal blooms can be a natural occurrence, however, there appears to be an escalation in the global occurrence of blooms that are harmful or toxic (Hallegraeff 1993, Anderson et al. 2002). Algal blooms occur as a result of increased nutrient inputs to waterbodies; phosphorus and nitrogen from industrial, agricultural and sewage sources are the two most important human-derived inputs (Anderson et al. 2002). These inputs allow for the production of harmful and/or toxic algae, including a chemical called microcystin. Microcystin is known to cause skin irritations, vomiting, cancer of the liver and death in humans, livestock, pets and many aquatic organisms (Kuiper-Goodman et al. 1999, Sivonen and Jones 1999). Researchers have found the transfer of microcystin from aquatic to terrestrial ecosystems, namely through the emergence of aquatic insects (Smith et al. 2008) which in turn are ingested by bats. Microcystin was detected in all fecal samples (n=20) of Little Brown Myotis near a lake in Michigan that experiences seasonal algal blooms (Woller-Skar et al. 2015). Further study is required to understand the impacts to individual bats and populations, however, this toxin may represent a previously unrecognized threat to bat populations (Woller-Skar et al. 2015). Another toxin associated with algal blooms (Anabaena flos-aquae) was implicated in a mass mortality event of bats (including Myotis spp.) in Alberta (Pybus et al. 1986).
Light Pollution
The alteration (e.g., timing, spatial extent, and spectral signature) of natural light regimes from artificial light sources can impact species in various direct and indirect ways related to foraging, reproduction, communication, habitat use, and movement behavior (Stone et al. 2009, Gaston et al. 2013, Mathews et al. 2015). Impacts may be beneficial (e.g., increased foraging opportunities), neutral, or detrimental (e.g., increased susceptibility to predation, collisions with lighted structures) (Kyba et al. 2011).
For bats in particular, when insect prey becomes concentrated around light sources, foraging efficiency can increase which has been viewed as beneficial to some species (Entwistle et al. 2001, Lacoeuilhe et al. 2014, Mathews et al. 2015). In southwestern Ontario, Furlonger (1987) found all species of bats encountered exploited concentrations of insects around artificial light sources though this was not significant for Myotis spp.; Tri-colored Bats were not encountered. In general, it has been found that bats with high or medium wing-loading and fast flight exploit insects at street lamps while gleaners and flutter-detectorsFootnote11 rarely, if ever, forage at street lights (Mathews et al. 2015). Similarly, in France, aerial hawkersFootnote12 were light-tolerant, while slow fliers would experience a high predation risk at high light levels and thus do not use lit areas to forage (Lacoeuilhe et al. 2014). Little Brown Myotis are aerial hawkers and efficient, maneuverable fliers, and are therefore expected to benefit from lights; Northern Myotis are slow fliers that often hover hunt and Tri-colored Bats are slow, erratic, flutter fliers, and are therefore not expected to forage at lights (Naughton 2012).
Though seemingly beneficial to some species of bats, light pollution has been attributed to reductions in many insect populations that are attracted to lights, including moths, aquatic insects, and other terrestrial insects (Frank 1988, Perkin et al. 2014, MacGregor et al. 2015). Many insects are attracted to artificial lights, affecting their dispersal and navigation as well as reproduction, mating, crypsis, and ability to evade predators. Individuals experience direct mortality from flying to exhaustion, burning to death, or becoming trapped in light receptacles (Frank 1988, Horváth et al. 2009, Perkin et al. 2014). This in turn reduces the biomass and abundance, and can change the relative composition of insect populations, creating implications in the food chain through disruption of predator-prey relationships, pollination services, and ecosystem function (Hölker et al. 2010, Kyba et al. 2011).
In Hungary, illumination of the roosts of house-dwelling Myotis species resulted in the collapse of entire colonies and reduced growth rates of juveniles (Boldogh et al. 2007). This was due to delayed emergence from roosts resulting in missed opportunities for foraging during peak insect activity; presumably, the avoidance of lighted areas is due to increased predation risk and/or negative effects on the bats’ orientation ability (Boldogh et al. 2007, Lacoeuilhe et al. 2014).
The impacts of artificial light appear to be variable and species-specific. Little direct research dedicated to understanding the effects of light pollution has been done on these three species of bats in Canada and further study is required.
Habitat or Prey Dynamic Alterations resulting from Climate Change
The effects of climate change on bats are unknown. Bats (particularly lactating females) are more susceptible to evaporative water loss than other mammals, suggesting that they may be vulnerable to increased temperatures associated with climate change (Webb et al. 1995, Chruszcz and Barclay 2002, Adams and Hayes 2008). Adams (2010) found significant reproductive declines in Little Brown Myotis in years that mimicked predicted conditions related to future climate change scenarios (i.e., reduced availability of water).
Other direct effects include the destruction of roosts and/or hibernacula as a result of increased storm frequency that is predicted to occur in the future (Jones and Rebelo 2013). Although warmer temperatures as a result of climate change could benefit hibernating bats in Canada, it may also lead to a disruption of hibernation, a reduction in water, and increased disease (Sherwin et al. 2013). Humphries et al. (2002) predicted climate change to cause a northward range expansion of Little Brown Myotis within 80 years.
The extent, intensity, and frequency of forest fires are projected to further increase because of warmer and drier springs and summers (Flannigan et al. 2009, de Groot et al. 2013, Girardin et al. 2013). Similarly, forest insect outbreaks (e.g., Spruce Budworm, and Mountain Pine Beetle) may intensify with the changing climate (Mattson and Haack 1987). These processes have the ability to alter large forested areas and cause whole tree mortality (Fleming et al. 2002), but may also create available snags for bat roosting and/or increase local prey availability (Wilson and Barclay 2006). Therefore, the ultimate impacts on bat populations and their habitats are unknown.
In addition to direct effects, climate change is predicted to indirectly affect bat survival through its effect on insect populations (Arlettaz et al. 2001). In northeastern regions of the continent, climate change is expected to cause wetter winters and drier summers (Hayhoe et al. 2007, Huntington et al. 2009). Adult female Little Brown Myotis have reduced annual survival in dry years, presumably related to the link between moisture availability and emergent insect availability (Frick et al. 2010b). The timing of peak abundances in some insects have also become earlier (Both et al. 2009). This may affect the synchronicity of peak prey densities and bat breeding (Jones et al. 2009) and therefore pup survival; sufficient prey is needed for pups to gain the fat tissue that is necessary for overwinter survival (Kunz et al. 1998).
Collisions with Vehicles
Collisions with vehicles can cause direct mortality to bats (Medinas et al. 2013). Seasonal timing, surrounding habitat, and level of vehicular traffic affect the number of collisions with vehicles (Lesiński et al. 2011, Medinas et al. 2013). Mortality rates are highest near roosts and active foraging areas (Medinas et al. 2013) and forest-adapted species, such as Northern Myotis and Tri-colored Bat, have the highest risk due to their characteristic low and slow flight (Abbott et al. 2015). There are also anecdotal reports of bats colliding with non-traditional / recreational vehicles and devices, such as water crafts (e.g. boats, personal watercrafts, and wind surfers), fishing lines, and all-terrain vehicles. The severity of this threat on the populations of Little Brown Myotis, Northern Myotis, and Tri-colored Bat in Canada is unknown.
5. Population and Distribution Objectives
There are no reliable current or past population estimates for Little Brown Myotis, Northern Myotis, or Tri-colored Bat in Canada. As such, population trends will be estimated using data from known and previously surveyed hibernacula and roosts throughout the species’ Canadian range (as presented in COSEWIC 2013) as an index of the total population trends, and augmented by surveys and monitoring presented in section 6.2: Strategic Direction for Recovery of this recovery strategy.
Little Brown Myotis and Northern Myotis
Area-specific population objectives are defined based on the current (2015) status of WNS across the country: WNS-affected areas and areas that are not yet affected by WNS (see Figure 1). The current invasion frontFootnote13 is in northwestern Ontario, near Atikokan (as of 29 May 2015) (U.S. Fish and Wildlife Service 2015).
Distribution objective:
- The distribution objective for both the Little Brown Myotis and Northern Myotis is to maintain (or where applicable restore to) the pre-WNS extent of occurrence (the area that encompasses the known geographic distribution of the species in Canada as depicted in Figures 1 and 2).
Population objectives (within WNS-affected areas):
- Within WNS-affected areas, the short-term (12-18 years) population objective is to maintain (and where feasible increase) the current (2015) level of the population.
- Within WNS-affected areas, the long-term (many generations) population objective is a self-sustainingFootnote14, resilientFootnote15, and redundantFootnote16 population.
Population objective (areas not yet affected by WNS):
- Within areas not yet affected by WNS, the population objective is to maintain (and where feasible increase) the currentFootnote17 level of the population.
The range of WNS overlaps with approximately 28% and 17% of Little Brown Myotis and Northern Myotis’ Canadian ranges, respectively, and is expanding at an average rate of 200 to 250 kilometers per year (COSEWIC 2013). Since conditions suitable for P. destructans growth exist in areas not yet affected by WNS, without mitigation, the entire Canadian population of both species will likely be affected by 2025 to 2028 years (COSEWIC 2013). Therefore, a 12-18 year timeframe was deemed appropriate for the short-term population objective.
The long-term population objective is based on the slow population growth rate of these two species, which implies that populations would require many generations (i.e., hundreds of years) to recover (see Limiting Factors section). The degree to which the Canadian (and continental) populations of Little Brown Myotis and Northern Myotis will ever be able to fully recover to their historical levels in WNS-affected areas is uncertain. The objective for areas not yet affected by WNS recognizes that increasing populations (where feasible) will improve the recovery potential of the populations (e.g., ability to re-populate areas affected by WNS and increase the likelihood of finding individuals with resistance to WNS).
Preventing the introduction of WNS to hibernacula in areas not yet affected (e.g., Prairies, northern Canada, western Canada) is the most important factor for preventing further loss of individuals. In Canada, as WNS approaches less forested regions of southeastern Manitoba, the relative dryness and lower density of trees suggest that transmission may occur at a slower rate. Nevertheless, it is acknowledged that it may not be possible to prevent the spread of WNS.
Tri-colored Bat
Distribution objective:
- The distribution objective for Tri-colored Bat is to restore (then maintain) the pre-WNS extent of occurrence (the area that encompasses the known geographic distribution of the species in Canada as depicted in Figure 3).
Short-term (10 years) population objective:
- The short-term population objective is to maintain (and where feasible increase) the population at its current (2015) level over the next 10 years.
The 10-year time frame was deemed appropriate for the short-term population objective because determining if a population has stabilized or is increasing will take multiple years of data acquisition. During this timeframe, it is expected that extensive research into treatments for WNS on individuals or in hibernacula will be explored. Furthermore, the COSEWIC criteria for assessment include reviewing population change within 10-year windows.
Long-term population objective:
- The long-term population objective is a self-sustaining, resilient, and redundant population.
The degree to which the Tri-colored Bat population will be able to fully recover to its historical levels is uncertain. The long-term population objective is based on the expectation that, even if individuals develop resistance or a treatment for WNS is found, the slow population growth rate of this species means populations would require many generations (i.e., hundreds of years) to recover (see Limiting Factors section).
Because the Canadian population of Tri-colored Bat occurs at the northeastern part of its continental range, and the vast majority of its population and distribution occurs in the U.S. to the eastern coast of Central America, population changes at the continental level will have a significant effect on recovery feasibility in Canada.
These objectives will be reviewed during the development of the report required within five years after this strategy is posted on the Species at Risk Public Registry to assess the implementation of this strategy and the progress towards meeting its objectives (s. 46 of SARA). These objectives may be revised as new information related to WNS and bat populations across Canada become available.
6. Broad Strategies and General Approaches to Meet Objectives
6.1. Actions Already Completed or Currently Underway
The following list of actions is not exhaustive, but is meant to illustrate the main areas where work is already underway and to give context to the broad strategies to recovery outlined in section 6.2. Actions completed or underway include the following:
International:
- In April 2015, a ‘Letter of Intent Related to Efforts to Promote Conservation of Bats in the United Mexican States, the United States of America and Canada’ was signed by representatives of each of the three countries to increase collaboration and coordination of bat conservation across North America.
- The North American Bat Monitoring Program (NABat) is creating a continental-wide coordinated effort for monitoring bats at local to range-wide scales to provide reliable data that will promote effective conservation decision-making. A Plan for the North American Bat Monitoring Program was released in June 2015 (Loeb et al. 2015).
- North American Bat Conservation Alliance (NABCA) facilitates communication between bat organizations across North America, develops conservation priorities, and addresses conservation issues.
- The Northeast Bat Working Group and Western Bat Working Group enable information exchange between agencies, organizations, industry, and individuals interested in bat research, management, and conservation, and facilitates multi-state collaborations (Northeast Bat Working Group 2015, Western Bat Working Group 2015).
- The North American Society for Bat Research promotes and develops research of bats and coordinates an annual Northern American Symposium on Bat Research (NASBR 2015).
- The U.S. Fish and Wildlife Service provides funding for research and coordinates an annual WNS workshop or conference that brings together international researchers to present new results and develop management strategies.
National:
- Canadian Wildlife Health Cooperative (CWHC) works in collaboration with federal, provincial and territorial, academic, and non-governmental organizations to develop national decontamination protocols and product assessment, coordinate national-level monitoring of bats and WNS and timely reporting of WNS, and identify data gaps and data management needs in Canada. CWHC leads Canada’s Inter-agency White Nose Syndrome Committee which contains five technical working groups. This group updated A National Plan to Manage White Nose Syndrome in Bats in Canada in February 2015 and will likely continue to serve as one of the main avenues for national implementation efforts related to WNS (Canadian Wildlife Health Cooperative 2015a).
- Efforts are underway by CWHC, NABCA, and Environment Canada to expand the Edubat project into Canada to increase bat education and outreach (BatsLive 2015).
- Federally, provincially and territorially, bats are considered for land-use development project screening and permitting and during environmental assessments across Canada. Mitigations measures are established as necessary to minimize adverse effects.
- Environment Canada is developing beneficial management practices (BMPs) for the forestry, wind energy, mining, and nuisance wildlife control industries, as well as for bats in buildings.
- Researchers at University of Winnipeg and Trent University are investigating the genetic response of bats pre- and post-WNS across Canada.
- The Canadian Wildlife Federation has several outreach activities related to bats (Canadian Wildlife Federation 2015). For example, they have a national bat box program used to distribute bat boxes and encourage citizens to track occupancy.
- Several national parks across Canada are conducting bat monitoring using a national protocol developed by Parks Canada Agency and contributing to NABat.
- Parks Canada Agency has produced a video with support from CWHC to illustrate the use of decontamination protocols, along with information related to bats and WNS.
- Under the National Park General Regulations, caves within Canada's national park system are closed unless specifically opened by a park superintendent.
- Parks Canada Agency has drafted guidance for hibernacula and for roosting bats during the reproductive season.
Western and northern Canada:
- The Western Canada Bat Network (WCBN) facilitates information exchange between groups and individuals involved in bat research, management, and conservation in western Canada, Alaska, and some northwestern states, primarily through semi-annual newsletters.
- In British Columbia, the Community Bat Programs of British Columbia promotes conservation of bats on private lands, provides a resource to landowners dealing with bat issues, and engages citizen scientists to collect data on bat populations (Community Bat Programs of BC 2014).
- The Community Bat Programs of BC has developed BMPs for pest control techniques and has started outreach initiatives with provincial pest management companies (Community Bat Programs of BC 2014).
- The British Columbia Bat Action Team, in collaboration with the British Columbia Ministry of Environment is currently finalizing BMPs for caving activities and mining and wind energy industries.
- Winter bat activity is being monitored throughout southern British Columbia by Cori Lausen (Wildlife Conservation Society Canada), Environment Canada-Canadian Wildlife Service, the British Columbia Ministry of Environment (Dr. Purnima Govindarajulu) and others.
- The British Columbia Ministry of Environment and Dr. Cori Lausen have developed appropriate hygiene protocols for bat researches, cavers and others visiting/working in underground bat habitats.
- The British Columbia Ministry of Environment and the Royal British Columbia Museum are archiving incidental mortality of bats and bat DNA to establish baseline information for British Columbia. A complete health assessment including testing to detect WNS, rabies and other parasites is conducted by Dr. Chelsea Himsworth (British Columbia Ministry of Agriculture) on all the incidental mortality bat carcasses that are recovered.
- Dr. Cori Lausen is leading a liaison project (Bats and Cavers Project) with the caving community in British Columbia and Alberta to map bat use in caves and other underground habitats.
- Alberta Environment and Sustainable Resource Development has developed guidelines for minimizing the impacts of wind energy development on bats and protocols for pre- and post-construction surveys.
- The Alberta Bat Action Team develops protocols, identifies research and management priorities, and facilitates information exchange.
- The Government of Yukon surveys Little Brown Myotis and Northern Myotis to better document their occurrence, reproductive status, and range in Yukon.
- The Government of Yukon and Government of the Northwest Territories conduct public education through interpretative events and brochures.
- The Government of Yukon engages in various research projects including studying the effects of natural disturbances, the importance of old-growth riparian forest, and long-term monitoring at select maternity colonies.
- The Government of the Northwest Territories is gathering baseline information on bats throughout Northwest Territories, searching for undiscovered hibernacula, and conducting WNS surveillance, population monitoring, and management planning at known hibernacula.
- Various partners (including Government of the Northwest Territories, Sahtu Renewable Resources Board, industry, academic researchers and community members) have erected acoustic bat recorders at several locations in the Sahtu, Dehcho, South Slave and North Slave regions of the Northwest Territories to learn more about the distribution and activity patterns of bats in these regions.
Central Canada:
- The Ontario Ministry of Natural Resources and Forestry has developed guidelines for minimizing the impacts of wind energy development on bats and is working with the Ministry of Northern Development and Mines and the mining industry to minimize industrial disturbance to bats and their habitats.
- Ontario’s Forest Management Guide for Conserving Biodiversity at the Stand and Sites Scales includes direction designed to maintain suitable habitats and habitat features, including hibernacula (Ontario Ministry of Natural Resources 2010).
- The Ontario Ministry of Natural Resources and Forestry has developed Ontario’s White-nose Syndrome Response Plan (Ontario Ministry of Natural Resources and Forestry 2015).
- The Ontario Ministry of Natural Resources and Forestry acoustically monitors bat populations during summer and winter throughout Ontario.
- The Ministère des Forêts, de la Faune et des Parcs of Québec in collaboration with the Centre de la science de la biodiversité du Québec (CSBQ) and the University of Winnipeg developed a website (English: BatWatch; French : chauve-souris.ca) to promote a citizen science project for maternity roost monitoring in central Canada (Quebec, Ontario and Manitoba). The website presents documentation about bats, their conservation, and BMPs for bats in buildings. The website also hosts a maternity roost database, where citizens can enter a roost and its annual counts.
- Environment Canada has collected radar and acoustic data in southern Ontario to determine if bats exhibit broad-front migratory movements or concentrate along linear landscape features.
- Summer population monitoring is underway in Ontario by the University of Guelph and Myotistar.
- Myotistar and Gray Owl Environmental Inc. (with support from Environment Canada and the National Speleological Society) are conducting advanced roost use monitoring at a significant summer roost near Cambridge, Ontario where bats are surviving and reproducing despite presence of WNS.
Eastern Canada:
- The Ministère des Forêts, de la Faune et des Parcs of Québec continues to track the spread of WNS and to monitor bat populations during summer using acoustic surveys and maternity roost counts. More intensive monitoring is planned in the Côte-Nord region where WNS has not been detected.
- A bat recovery team for Québec was created in 2014. The team is developing a recovery plan for the province and coordinating provincial-level recovery actions.
- Bat populations are being monitored regularly by the Department of National Defence at Canadian Forces Base Valcartier.
- St. Mary’s University and Environment Canada are investigating mercury levels in Little Brown Myotis in Atlantic Canada.
- The New Brunswick Museum has monitored the spread of WNS, and associated rate of mortality at known hibernacula throughout the province during pre- and post-WNS years, and continues to provide research in the pervasiveness of the pathogen, the cave microfauna, and environmental conditions.
- The New Brunswick Museum, in collaboration with New Brunswick Department of Natural Resources, have established a reporting program to collect observations of winter day-flying bats, leading to new information on the distribution of these species in the province.
- New Brunswick Department of Natural Resources has developed survey guidelines for bat and bird mortality during pre- and post-construction periods of wind farm developments in New Brunswick.
- Prince Edward Island’s Forest, Fish and Wildlife Division promotes the reporting of bat sightings and works with the CWHC to implement bat monitoring programs.
- Nova Scotia Bat Conservation by the Nova Scotia Department of Natural Resources monitors bats during the spring, summer, and fall.
- St. Mary’s University is monitoring winter counts at hibernacula in Nova Scotia.
- St. Mary’s University is researching demographics and social behavior of Little Brown Myotis in collaboration with Newfoundland and Labrador Department of Environment and Conservation in Newfoundland and Labrador.
- The Government of Newfoundland and Labrador continues WNS surveillance and monitors known populations. In collaboration with Memorial University of Newfoundland and Parks Canada, acoustic monitoring takes place to determine species’ presence and abundance. Surveys for Little Brown Myotis and Northern Myotis in Labrador are conducted to better document the occurrence, reproductive status and range.
6.2. Strategic Direction for Recovery
The Canadian Wildlife Health Cooperative (Canadian Inter-agency White Nose Syndrome Committee) released a revised version of A National Plan to Manage White Nose Syndrome in Bats in Canada in February 2015 (Canadian Wildlife Health Cooperative 2015b). Whenever possible, the approaches in Table 3 were developed to align with initiatives outlined in that plan. When appropriate, the approaches were categorized into those that are applicable to non-WNS affected areas and those that are applicable to WNS-affected areas (and the invasion front).
| Threat or Limitation | Broad Strategy to Recovery | PriorityNoteeof Table 3 | General Description of Research and Management Approaches |
|---|---|---|---|
| Knowledge gaps to recovery | Monitoring and surveys | High | Entire range:
|
| Knowledge gaps to recovery | Monitoring and surveys | Medium |
|
| Knowledge gaps to recovery | Monitoring and surveys | Low |
|
| Knowledge gaps to recovery and all threats | Research | High | Entire range:
WNS-affected areas (and invasion front):
Non-WNS affected areas:
|
| Knowledge gaps to recovery and all threats | Research | Medium |
|
| Knowledge gaps to recovery and all threats | Education and awareness, and partnerships | High |
|
| All threats | Habitat and species conservation and management | High | Entire range:
WNS-affected areas (and invasion front):
Non-WNS affected areas:
|
| All threats | Habitat and species conservation and management | Medium |
Non-WNS affected areas:
|
| All threats | Law and policy | High |
|
6.3. Narrative to Support the Recovery Planning Table
Recovery of Little Brown Myotis, Northern Myotis, and Tri-colored Bat will require commitment, resources, collaboration, and cooperation among federal, provincial, and territorial jurisdictions, wildlife management boards, Aboriginal people, species experts and researchers, local communities, landowners, industry, and other interested parties from Canada and the U.S.. Because of their widespread range across the country, it will be important to monitor the spread of WNS, hibernacula, maternity roosts, population trends, and the distributions of the three species of bats so that the effectiveness of the recovery efforts can be evaluated, and adjusted as necessary.
A comprehensive approach to research and monitoring (which includes all stages of the annual life cycles and the entire extent of occurrence) will be required to more completely understand the status, limiting factors, significant threats, and habitat use of each species. Currently, there are no reliable population estimates for any of the three bat species and, in the short-term, it is not realistic to expect that population estimates will be derived. To determine the success of conservation and management efforts, and determine if the population and distribution objectives have been met, an effective monitoring program is needed to measure relative changes in bat abundance over time. To maintain a uniform sampling method across Canada, the protocol of the North American Bat Monitoring Program (NABat) should be used, to the extent possible, as the Canadian national sampling framework (Loeb et al. 2015).
Because WNS is spreading quickly across Canada, research and monitoring pertinent to the fungus must be performed promptly and efficiently before opportunities are lost. This will likely require a large body of researchers, governments, industry, Aboriginal communities, and volunteers to coordinate efforts, communicate effectively, pool resources, and share findings efficiently (Langwig et al. 2015a). Whenever possible, research efforts and priorities should be coordinated through established groups (e.g., North American Bat Conservation Alliance, and CWHC) to avoid duplicated effort and resources. To succeed in preventing the spread of WNS (see section 5: Population and Distribution Objectives), research aimed at a treatment for individuals and/or hibernacula infected with WNS or methods to significantly reduce or prevent the spread of P. destructans will continue to be required.
As noted in the threats section (section 4: Threats), in areas where local bat populations have significantly declined as a result of WNS, the level of concern related to other potential threats increases (Langwig et al. 2015a). Identifying the importance and risk factors associated with these threats on each of the three species becomes increasingly important (see Appendix B), so that appropriate mitigation measures can be adopted in a timely manner.
While necessary monitoring and research occurs across North America, the current state of available science can provide a base of knowledge to conserve known habitats and mitigate threats to the species.
7. Critical Habitat
Critical habitat is the habitat that is necessary for the survival or recovery of the species. Section 41(1)(c) of SARA requires that the recovery strategy include an identification of the species’ critical habitat, to the extent possible, as well as examples of activities that are likely to result in its destruction.
7.1. Identification of the Species’ Critical Habitat
The critical habitat identified in this document is considered a partial identification, insufficient to meet the population and distribution objectives. A schedule of studies has been developed to provide the information necessary to complete the identification of critical habitat (see section 7.2: Schedule of Studies). In this recovery strategy critical habitat is partially identified for hibernacula, based on the best available information for each species as of October 2015.
Hibernacula
Hibernacula are used by Little Brown Myotis, Northern Myotis, and Tri-colored Bat to survive when ambient temperatures decline and insects are unavailable, and as such are necessary for the survival and recovery of these species. Suitable hibernacula may be limiting for these species (OMNR 2010).
As a result of human safety concerns, efforts to minimize bat disturbance, and the difficulty in locating hibernating bats within complex structures, the number of bats using a hibernaculum often cannot be fully counted. In addition, the discovery of one hibernating individual can sometimes indicate the occurrence of more undetected individuals using the hibernaculum.
Occupancy
- Any site where Little Brown Myotis, Northern Myotis, or Tri-colored Bat has been observed hibernating during the winter at least once since 1995 is identified as critical habitat.
The time period (≥ 1995) recognizes that Little Brown Myotis, Northern Myotis, and Tri-colored Bat exhibit strong site fidelity to hibernacula, individual sites may be used by hibernating bats for decades, and suitable hibernacula may be limiting.
At sites where swarming (an activity that typically occurs adjacent to or within a hibernaculum) was observed and there was appropriate habitat for hibernation but hibernation could not be verified (e.g., chambers are inaccessible, or entry is avoided to minimize disturbance), the swarming activity is considered an indicator of a site’s use for hibernation. As such, these sites were included in the identification of critical habitat.
Sites that were deemed to have unsuitable habitat (e.g., suitable habitat no longer exists, or hibernating bats did not survive winter) were not identified as critical habitat; this did not include sites affected by WNS. The long-term persistence of the WNS pathogen, P. destructans, in hibernacula affects the suitability of caves and mines for maintaining self-sustaining and resilient bat populations. Nevertheless, WNS-affected hibernacula are considered critical habitat because they need to be preserved from loss or modification to aid population recovery in the scenario that a treatment or decontamination measures are discovered, or natural resistance is developed.
Knowledge regarding the importance of habitat associations, their configuration surrounding the hibernacula, and in what geographical scale they exert an influence, if any, on the predictability of bat occurrence at these sites, is limited. At present, knowledge related to the locations of hibernacula is also limited throughout the range of these species. In addition, difficulties in confirming the identity of Little Brown Myotis in areas of British Columbia where Yuma Myotis also exist, limits the ability to confirm hibernacula use by this species. Filling these knowledge gaps is necessary to complete the identification of critical habitat for hibernacula and has been addressed in the schedule of studies (section 7.2).
Biophysical attributes
- Typically, hibernacula for these species are subterranean features, such as caves, abandoned mines, hand-dug wells, cellars, or tunnels where light and noise levels are low.
- Hibernacula typically contain sections that have relatively stable temperatures (2-10 ˚C) and stable, high humidity levels (>80 %).
A single hibernaculum may include multiple entry and exit points and vast underground networks of chambers. The full extent of these features are included in the definition of hibernacula as critical habitat (regardless of the location of hibernating bats within the structure) because (1) individuals may use multiple areas within these structures and it is not always possible to determine their usage, and (2) the entire, intact network is generally required to maintain microclimatic conditions (e.g., air flow, temperature, and humidity).
Sites used for hibernation in western Canada, particularly along the Pacific Coast, may differ substantially from those elsewhere in the species Canadian range, and our knowledge of the location and biophysical attributes of these hibernacula is very limited (Jung et al. 2014). These knowledge gaps have been addressed in the schedule of studies (section 7.2) and will allow for the complete identification of critical habitat for hibernacula in these areas.
Geographical Location
The areas containing critical habitat for Little Brown Myotis, Northern Myotis, and/or Tri-colored Bat are presented in Figures 4-20. Critical habitat occurs within the standardized UTM grids where the critical habitat criteria (occupancy and biophysical attributes) described in this section are met. Critical habitat is displayed as UTM grids that represent all species that occur within a particular province or territory and identified critical habitat may include one or more of the species of interest. The UTM grid squares shown on these figures are part of a standardized national grid system that highlights the general geographic area containing critical habitat. Critical habitat is presented within standardized UTM grids, in order to respect protocols for provincial and territorial species at risk data use (and related agreements) and to protect the species and their habitats from disturbance and the potential introduction of WNS. The grid size used to display the critical habitat (i.e., 10, 50, or 100 km2) is relative to the ability and risks associated with the location of critical habitat being discovered (as perceived by the data owners). More detailed information on the location of critical habitat to support protection of the species and its habitat may be requested, on a need-to-know basis, by contacting Environment Canada’s Recovery Planning section at: ec.planificationduretablissement-recoveryplanning.ec@canada.ca.
A total of 192 hibernacula were identified as critical habitat in Newfoundland and Labrador, Nova Scotia, New Brunswick, Quebec, Ontario, Manitoba, Alberta, British Columbia, and Northwest Territories based on data as of October 2015. The locations of hibernacula are not known in Yukon, Saskatchewan, and Nunavut. Data on the locations of hibernacula in Prince Edward Island are unavailable because of landowner privacy concerns.
Maternity Roosts
Maternity roosts are used for giving birth and rearing young, and clearly contribute to the survival and recovery of these three species of bats. However, the locations of the vast majority of maternity roosts are currently either unknown or undocumented, or the data are unavailable to Environment Canada. Given this, it is not possible to determine which maternity roosts are necessary for the survival or recovery of these species; therefore, maternity roosts are not identified as critical habitat in this recovery strategy.
Once additional information on the locations and attributes of maternity roosts is available, it will be possible to set criteria that would identify the roosts that are necessary for the survival or recovery of the species as critical habitat. Criteria for identifying which maternity roosts are critical habitat would likely consider species, number of individuals using the roost, whether the roost is within a WNS-affected area, and the number of other known maternity roosts in the vicinity.
The work required to obtain the necessary information and establish criteria for the identification of maternity roosts as critical habitat are included in the schedule of studies (section 7.2).
Similar to hibernacula, knowledge of the location and biophysical attributes of maternity roosts in western Canada is very limited and habitat characteristics may differ substantially from those elsewhere in the species’ Canadian range (Jung et al. 2014). Filling these knowledge gaps is necessary to complete the identification of critical habitat for maternity roosts and is addressed in the schedule of studies (section 7.2).
Landscape-scale identification
The geographic range of the species, threats, and habitat specificity of Little Brown Myotis (and possibly Northern Myotis) suggest the critical habitat associated with summering habitat (i.e., roosting and foraging) should eventually be identified at a landscape scale. This would permit the long-term management of the habitat needed for survival and recovery of the species. This type of identification would reflect the dynamic mosaic of habitat conditions available and required on the landscape. Nevertheless, the available information is not adequate to currently identify critical habitat at a landscape scale for the following reasons:
- There is a lack of data related to bat presence and abundance in large portions of their ranges.
- Habitat requirements may vary across the ranges of the species. Management units (i.e., geographic units within which critical habitat would be managed) need to be identified in such a way to best reflect variation in habitat use and management patterns.
- There is a lack of understanding and data to indicate the appropriate configuration of important landscape biophysical attributes.
- It is unclear whether certain summering habitats with specific biophysical attributes may be functionally more important than others. For example, specific habitats may have greater densities of individuals and/or result in higher reproductive success.
- The relationships between anthropogenic disturbance and habitat quality are poorly known. A better understanding of these relationships is needed to ensure sufficient suitable habitat is available for these species and to identify at what scale and intensity activities would be likely to destroy the critical habitat.
It will take many years to gather sufficient data to address the knowledge gaps above. Therefore, a long-term schedule of studies is presented in section 7.2 to address the landscape-scale identification of summering habitat.
Other Habitats
Currently, male roosting sites, migration routes, and swarming sites are not identified as critical habitat. It is unclear whether the habitats that support male roosts or migration routes would be required for the survival or recovery of each species. At present, the knowledge of habitat requirements for the selection of significant swarming sites is insufficient to identify the biophysical attributes. Determining if male roosting sites and migration routes warrant identification as critical habitat and studies on the importance and biophysical attributes of swarming sites is included in the schedule of studies (section 7.2).
In summary, the critical habitat of Little Brown Myotis, Northern Myotis, and Tri-colored Bat can be partially identified at this time. A schedule of studies has been developed to provide the information necessary to complete the identification of critical habitat that will be sufficient to meet the population and distribution objectives. Although the short-term schedule of studies spans 10 years, the critical habitat can be updated as new information becomes available, either in a revised recovery strategy or action plan(s).
Long description for Figure 4
Standardized universial transverse mercator (UTM) grid square (red-border and semi-transparent fill) at a 50 x 50 resolution that contains critical habitat for Little Brown Myotis (Myotis lucifugus) and Northern Myotis (Myotis septentrionalis) in Newfoundland and Labrador. The grid square covers both the eastern and western portion of White Bay on the Island of Newfoundland. The standardized national grid system indicates the geographic area containing critical habitat but mapping at finer resolution is not shown. Critical habitat for these species occurs within the UTM grid square where the description of critical habitat is met (i.e., hibernacula have been identified).
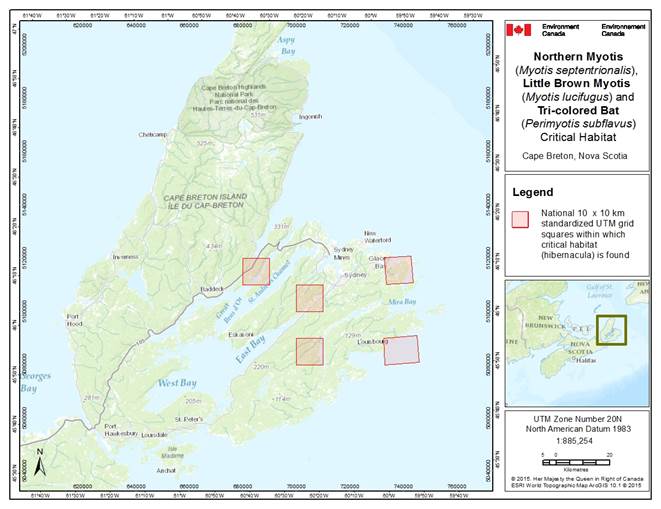
Long description for Figure 5
Grid squares (red-outline and semi-transparent fill) that contain critical habitat for Little Brown Myotis (Myotis lucifugus), Northern Myotis (Myotis septentrionalis), and Tri-colored Bat (Perimyotis subflavus) in northern Nova Scotia on Cape Breton Island. Critical habitat for these species occurs within the 10 x 10 km standardized UTM grid squares where the description of critical habitat is met (i.e., hibernacula have been identified). Standardized UTM grid squares at the intersection of UTM zones are merged with their adjacent grid squares. This standardized national grid system indicates the general geographic area containing critical habitat; detailed critical habitat mapping is not shown. Grids (four) containing critical habitat for these species are found on south-east of Cape Breton (Island) and north of Saint Andrew’s Channel (one grid).
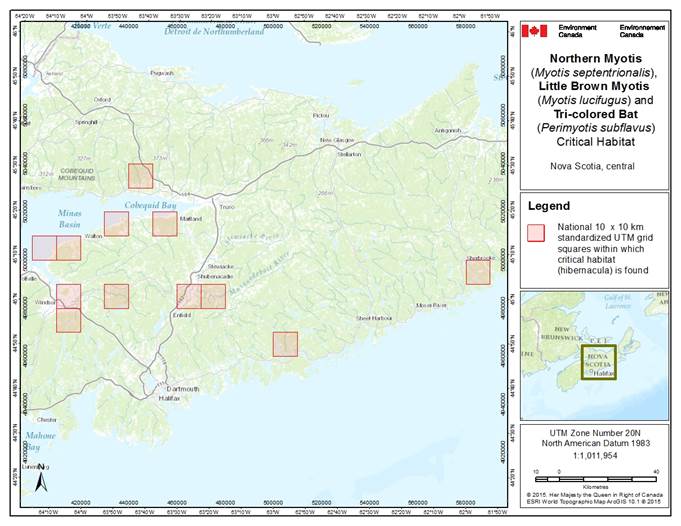
Long description for Figure 6
Grid squares (twelve) (red-outline and semi-transparent fill) that contain critical habitat for Little Brown Myotis (Myotis lucifugus), Northern Myotis (Myotis septentrionalis), and Tri-colored Bat (Perimyotis subflavus) in central, mainland Nova Scotia . Critical habitat for these species occurs within these 10 x 10 km standardized UTM grid squares where the description of critical habitat is met (i.e., hibernacula have been identified). This standardized national grid system indicates the general geographic area containing critical habitat; detailed critical habitat mapping is not shown. Most grid squares (ten) containing critical habitat are located on the eastern portion of mainland Nova Scotia, south of the Minas Basin and both north and south of Cobequid Bay, as well as north of Enfield. Isolated grid squares are also found in the south-central mainland coast and south of Sherbrooke.
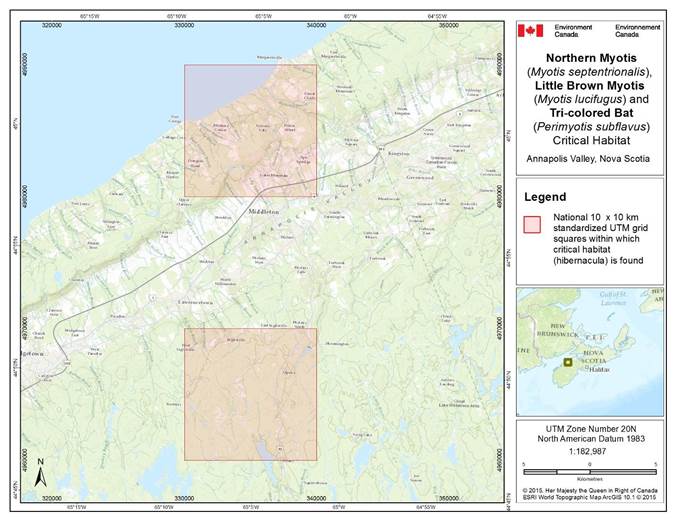
Long description for Figure 7
Grid squares (two) (red-outline and semi-transparent fill) that contains critical habitat for Little Brown Myotis (Myotis lucifugus), Northern Myotis (Myotis septentrionalis), and Tri-colored Bat (Perimyotis subflavus) in Annapolis Valley of mainland Nova Scotia. Critical habitat for these species occurs within the 10 x 10 km standardized UTM grid square where the description of critical habitat is met (i.e., hibernacula have been identified). This standardized national grid system indicates the general geographic area containing critical habitat; detailed critical habitat mapping is not shown. One grid square is located north of Middelton and and the second is located south of Middleton.
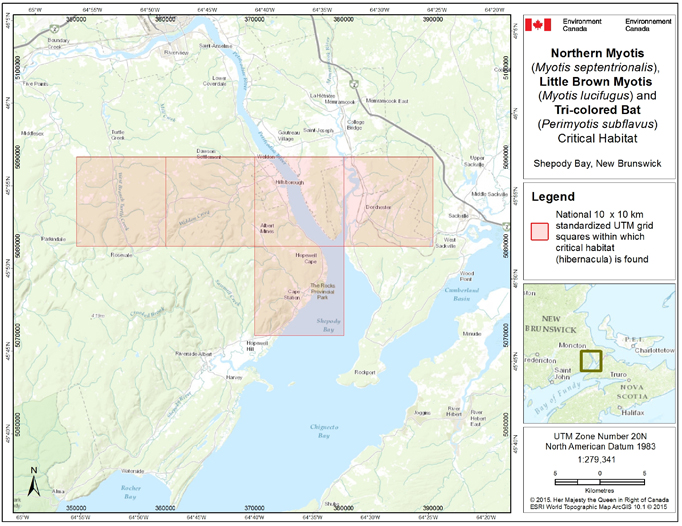
Long description for Figure 8
Grid squares that contain critical habitat for Little Brown Myotis (Myotis lucifugus), Northern Myotis (Myotis septentrionalis), and Tri-colored Bat (Perimyotis subflavus) in southern New Brunswick (Shepody Bay area). Critical habitat for these species occurs within these 10 x 10 km standardized UTM grid squares where the description of critical habitat is met (i.e., hibernacula have been identified). This standardized national grid system indicates the general geographic area containing critical habitat; detailed critical habitat mapping is not shown. Critical habitat grid squares are continuous from the mouth of Shepody Bay and west from Peticodiac east to Dorchester.
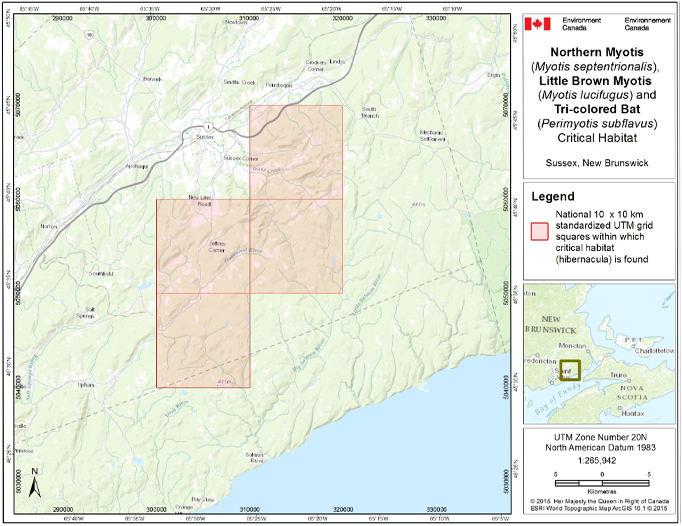
Long description for Figure 9
Grid squares (five) that contain critical habitat for Little Brown Myotis (Myotis lucifugus), Northern Myotis (Myotis septentrionalis), and Tri-colored Bat (Perimyotis subflavus) in southeastern New Brunswick (Sussex area). Critical habitat for these species occurs within these 10 x 10 km standardized UTM grid squares where the description of critical habitat is met (i.e., hibernacula have been identified). This standardized national grid system indicates the general geographic area containing critical habitat; detailed critical habitat mapping is not shown. Critical habitat grid squares encompass an area south of Jefferies Corner along the Hammond River and an area east of Sussex Corner along Trout Creek.
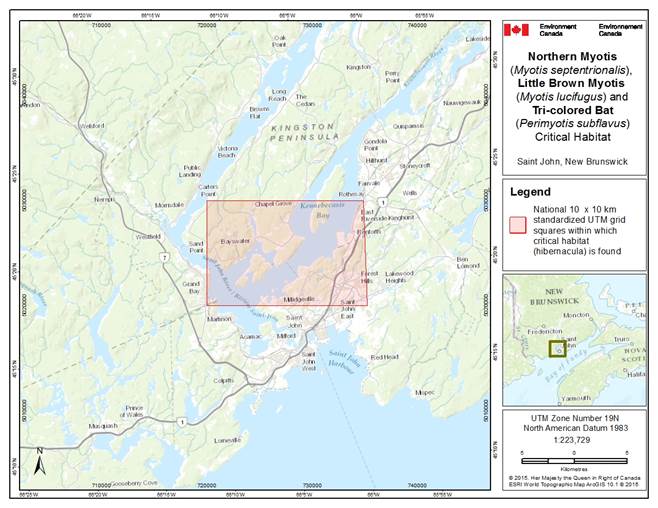
Long description for Figure 10
Grid square (red-outline and semi-transparent fill) that contains critical habitat for Little Brown Myotis (Myotis lucifugus), Northern Myotis (Myotis septentrionalis), and Tri-colored Bat (Perimyotis subflavus) in south-central New Brunswick, encompassing and greater Saint region, Grand Bay-Westfield region and southern portion of the Kingston Peninsula. Critical habitat for these species occurs within this 10 x 10 km standardized UTM grid square where the description of critical habitat is met (i.e., hibernacula have been identified). This standardized national grid system indicates the general geographic area containing critical habitat; detailed critical habitat mapping is not shown. Standardized UTM grid squares at the intersection of UTM zones are merged with their adjacent grid squares.

Long description for Figure 11
Grid that contain critical habitat for Little Brown Myotis (Myotis lucifugus) and Northern Myotis (Myotis septentrionalis) in Quebec, along the southern shoreline of the mouth of the St. Lawrence River (Bas-Saint-Laurent area). The locations of the critical habitat grid squares are isolated and discontinuous from one another. Critical habitat for these species occurs within these 10 x 10 km standardized UTM grid squares where the description of critical habitat is met (i.e., hibernacula have been identified). This standardized national grid system indicates the general geographic area containing critical habitat; detailed critical habitat mapping is not shown.
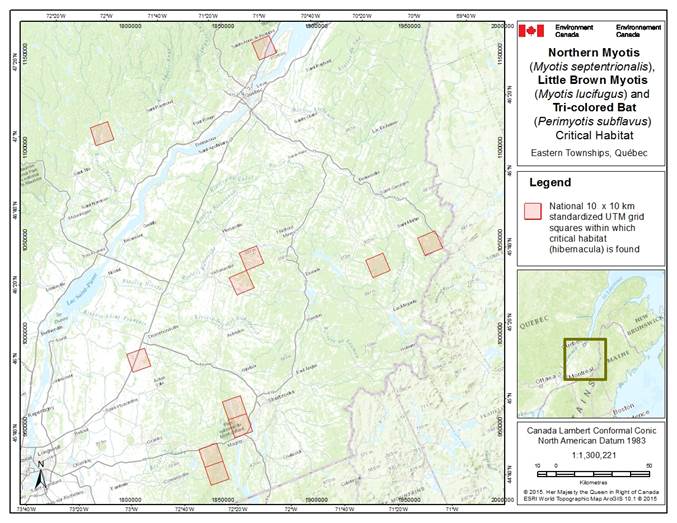
Long description for Figure 12
Grid squares) that contain critical habitat for Little Brown Myotis (Myotis lucifugus), Northern Myotis (Myotis septentrionalis), and Tri-colored Bat (Perimyotis subflavus) in Quebec, Eastern Townships region. Critical habitat grid squares are widely dispersed in the region, with three grids representing adjacent geographic areas where critical habitat has been identified. Critical habitat for these species occurs within these 10 x 10 km standardized UTM grid squares where the description of critical habitat is met (i.e., hibernacula have been identified). This standardized national grid system indicates the general geographic area containing critical habitat; detailed critical habitat mapping is not shown.

Long description for Figure 13
Grid squares that contain critical habitat for Little Brown Myotis (Myotis lucifugus) and Northern Myotis (Myotis septentrionalis) in Quebec (Nord-du-Quebec region). Critical habitat grid squares are highly dispersed in northern Quebec, located near Val-d’Or, north of Lebel-sur Quevillon, east Chibougamau, and southeast Lac Saint-Jean. Critical habitat for these species occurs within these 10 x 10 km standardized UTM grid squares where the description of critical habitat is met (i.e., hibernacula have been identified). This standardized national grid system indicates the general geographic area containing critical habitat; detailed critical habitat mapping is not shown.

Long description for Figure 14
Grid squares (ten) (red-outline and semi-transparent fill) that contain critical habitat for Little Brown Myotis (Myotis lucifugus), Northern Myotis (Myotis septentrionalis), and Tri-colored Bat (Perimyotis subflavus) in Quebec (Outaouais region). Critical habitat grid squares are primarily located in the southern and eastern Outaouais with moderate dispersion in the southern portion and higher dispersion in the northern portion. Critical habitat for these species occurs within these 10 x 10 km standardized UTM grid squares where the description of critical habitat is met (i.e., hibernacula have been identified). This standardized national grid system indicates the general geographic area containing critical habitat; detailed critical habitat mapping is not shown.
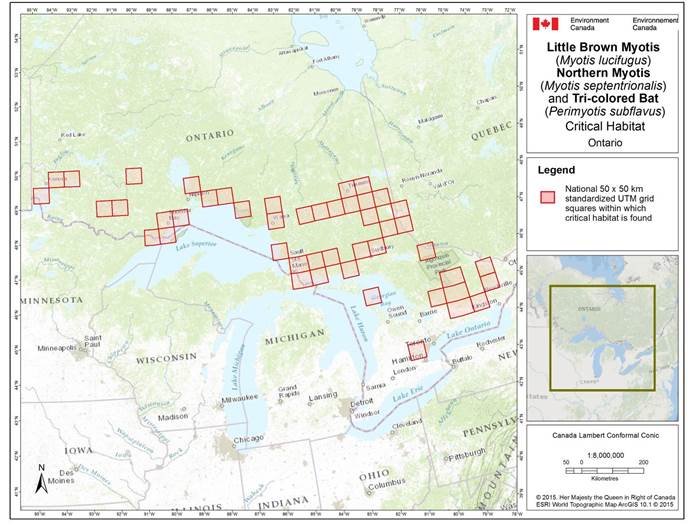
Long description for Figure 15
Grid square (47) (red-outline and semi-transparent fill) that contains critical habitat for Little Brown Myotis (Myotis lucifugus), Northern Myotis (Myotis septentrionalis), and Tri-colored Bat (Perimyotis subflavus) in Ontario. Critical habitat grid squares widely distributed in the southern boreal forest of Ontario with a high concentration in northeastern Ontario along the Quebec border, north shore of Lake Huron and central Algonquin-Ottawa Valley, and single grid in southern Ontario between Lake Erie and Lake Ontario. Critical habitat for these species occurs within these 50 x 50 km standardized UTM grid squares where the description of critical habitat is met (i.e., hibernacula have been identified). This standardized national grid system indicates the general geographic area containing critical habitat; detailed critical habitat mapping is not shown. Standardized UTM grid squares at the intersection of UTM zones are merged with their adjacent grid squares.
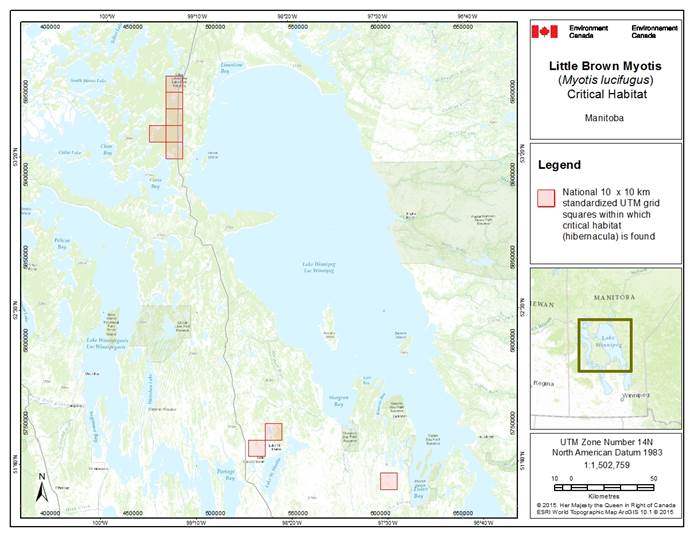
Long description for Figure 16
Grid squares (9) (red-outline and semi-transparent fill) that contain critical habitat for Little Brown Myotis (Myotis lucifugus) in Manitoba. Critical habitat grid squares are highly concentrated in the northwest of Lake Winnipeg, north of Grand Rapids, with isolated grids more isolated grids located north of Lake St. Martin and west of Fisher Bay. Critical habitat for this species occurs within these 10 x 10 km standardized UTM grid squares where the description of critical habitat is met (i.e., hibernacula have been identified). This standardized national grid system indicates the general geographic area containing critical habitat; detailed critical habitat mapping is not shown.

Long description for Figure 17
Grid square that contains critical habitat for Little Brown Myotis (Myotis lucifugus) in Alberta (Wood Buffalo National Park area). Critical habitat is mapped as single isolated grid in the northeast portion of Wood Buffalo National Park. Critical habitat for this species occurs within this 10 x 10 km standardized UTM grid squares where the description of critical habitat is met (i.e., hibernacula have been identified). This standardized national grid system indicates the general geographic area containing critical habitat; detailed critical habitat mapping is not shown.
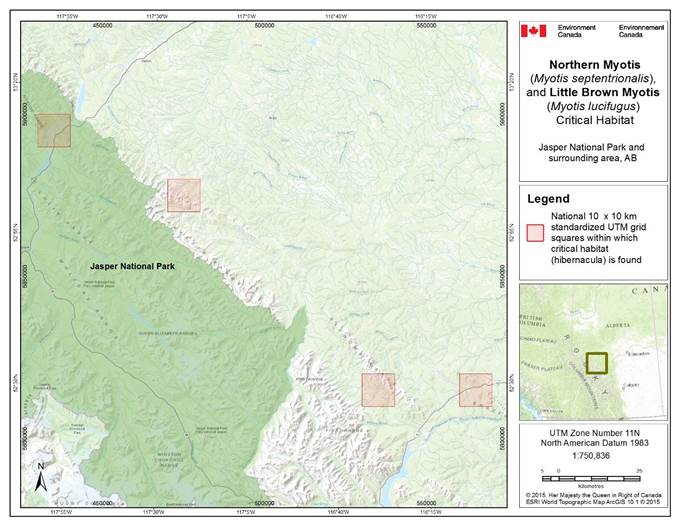
Long description for Figure 18
Grid squares (4) (red-outline and semi-transparent fill) that contains critical habitat for Northern Myotis (Myotis septentrionalis) and Little Brown Myotis (Myotis lucifugus) in Alberta (Jasper National Park and surrounding area). Critical habitat grid squares are widely dispersed along the eastern border of Jasper National Park, including locations near Pochahontas, north Mountain Park, and more southeastern locations along the Largehron Peak range and Nordegg. Critical habitat for these species occurs within these 10 x 10 km standardized UTM grid squares where the description of critical habitat is met (i.e., hibernacula have been identified). This standardized national grid system indicates the general geographic area containing critical habitat; detailed critical habitat mapping is not shown.

Long description for Figure 19
Grid square (red-outline and semi-transparent fill) that contains critical habitat for Northern Myotis (Myotis septentrionalis) in northeast British Columbia. The area bounded by the critical habitat grid encompasses an area southwest of Fort St. John. Critical habitat for this species occurs within this 50 x 50 km standardized UTM grid squares where the description of critical habitat is met (i.e., hibernacula have been identified). This standardized national grid system indicates the general geographic area containing critical habitat; detailed critical habitat mapping is not shown.
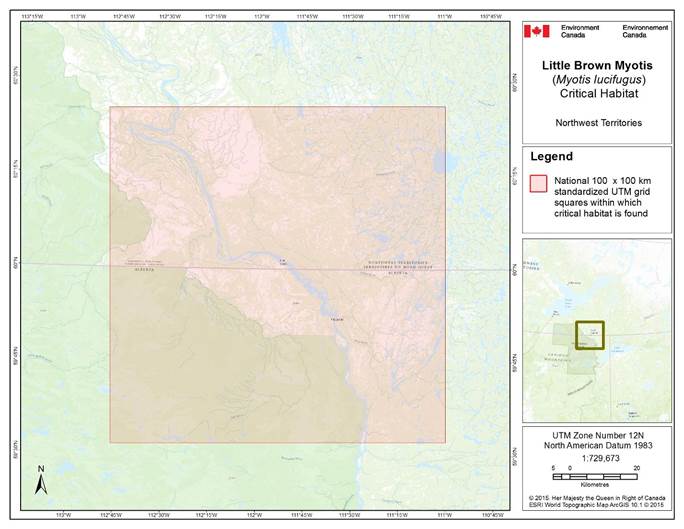
Long description for Figure 20
Grid square that contains critical habitat for Little Brown Myotis (Myotis lucifugus) in Northwest Territories. Critical habitat for this species occurs within these 100 x 100 km standardized UTM grid square where the description of critical habitat is met (i.e., hibernacula have been identified). This standardized national grid system indicates the general geographic area containing critical habitat; detailed critical habitat mapping is not shown.
7.2. Schedule of Studies to Identify Critical Habitat
A schedule of studies has been developed to provide the information necessary to complete the identification of critical habitat (Table 4 and 5).
| Description of Activity | Rationale | Timeline |
|---|---|---|
| Work with researchers, provinces / territories, Aboriginal communities, and non-government organizations to determine how sensitive data can be made available and used for critical habitat identification. | Lack of data availability (particularly related to anthropogenic structures) is a major impediment to critical habitat identification. | 2016-2017 |
| Conduct surveys in areas where hibernacula and maternity roosts are suspected but unconfirmed. | Known locations of hibernacula and maternity roosts are limited. In addition, confirmation of use by Little Brown Myotis versus Yuma Myotis is required in British Columbia. This information is needed to fully identify critical habitat. | 2016-2025 |
| Refine biophysical attributes for hibernacula and maternity roosts (particularly in western and northern parts of the species’ ranges). | Although general biophysical attributes are understood, further refining these is deemed essential for full identification of critical habitat. The limited knowledge is especially evident in western and northern parts of the species’ ranges. | 2016-2025 |
| Identify the location, characteristics, and biophysical attributes of swarming sites. | The importance of individual swarming sites to survival and recovery and the important biophysical attributes of these sites are unknown. | 2016-2025 |
| Determine criteria for identifying maternity roosts as critical habitat. | There is a lack of appropriate data to develop science-based criteria. | As appropriate |
| Description of Activity | Rationale | Timeline |
|---|---|---|
| Increase surveys and monitoring of summering habitat at strategic locations. | Information on abundance and other measures of summering habitat quality is poor in many regions of the country. Increased surveys and monitoring in pre-determined locations is necessary. | 2016-2031 |
| Determine if specific male roosting sites and migration routes are considered necessary for recovery and survival, and thus would warrant critical habitat identification. | It is currently unclear if these habitats (or even a subset of these) should be considered critical habitat. | 2019-2025 |
| Determine the appropriate configuration of landscape biophysical attributes. | To identify critical habitat at a landscape scale it is necessary to understand the biophysical attributes required by the species’ at this scale and to determine how these should be configured to meet the species’ needs. | 2016-2031 |
| Determine habitat quality across the species’ ranges. | Information on abundance, productivity and other measures of habitat quality may lead to the identification of areas that contribute disproportionately to the survival or recovery of these species. | 2016-2031 |
| Determine the scale and intensity at which suitable habitat would likely be destroyed by anthropogenic activities. | A better understanding of the relationship between anthropogenic disturbance and habitat quality is needed to ensure sufficient suitable habitat is available for these species and to identify at what scale and intensity activities would be likely to destroy critical habitat. | 2016-2031 |
| Determine how much suitable summering habitat is required to meet the population and distribution objectives. | It is uncertain whether summering habitat is limiting in Canada. An assessment of whether there is sufficient habitat in Canada to meet the population and distribution objectives is required. | 2032 |
| Develop and validate summering habitat models to determine where biophysical attributes are present in required quantity, quality and configuration across the species’ ranges to meet population and distribution objectives. | Results from studies listed above, will allow models to be built to identify the location, quantity, and quality of habitat that should be identified as summering critical habitat. | 2032-2036 |
7.3. Activities Likely to Result in the Destruction of Critical Habitat
This subsection of a recovery strategy describes the kinds of activities that are likely to cause the destruction of critical habitat and provides examples of such activities. Information is provided on potential impacts to critical habitat and species populations that may result from these example activities.
Destruction of critical habitat is determined on a case-by-case basis. Destruction would result if part of the critical habitat were degraded, either permanently or temporarily, such that it would not serve its function when needed by the species. Destruction may result from single or multiple activities at one point in time or from the cumulative effects of one or more activities over time. Activities described below include those likely to cause destruction of critical habitat for the species; however, destructive activities are not limited to those listed.
Hibernacula
Activities likely to result in the destruction of hibernacula identified as critical habitat include, but are not limited to, the following: activities resulting in the introduction of WNS into hibernacula previously free of WNS, activities that result in collapsed walls or ceilings, or activities that result in the hibernaculum being inaccessible or unavailable to bats or alters the hibernaculum’s temperature, humidity, airflow, or other microclimatic characteristics outside of the range acceptable to the bat species for which critical habitat is identified.
| Description of activity | Description of effect |
|---|---|
| Visitors (e.g., researchers, cavers, tourists) that do not follow proper decontamination protocols | May result in permanent or temporary direct destruction of habitat by introducing WNS. |
| Modifications to accommodate visitors (e.g., observation platform, altering entrance) | May result in permanent or temporary direct destruction of habitat or indirect effects (e.g., changes to microclimatic conditions, limit bat access to site). |
| Erection of physical barriers (e.g., doors, or gates) leading to reduced bat access and/or useFootnotef | May result in permanent or temporary direct destruction of habitat and/or indirect effects (e.g., restricting bat access to site, changes to airflow, temperature, or other microclimatic characteristics). |
| Filling wells | May result in permanent direct destruction of habitat. |
| Intentional vandalism | May result in permanent or temporary direct destruction of habitat and/or indirect effects (e.g., restricting access to site, changes to airflow, temperature, or other microclimatic characteristics). |
| Activities that cause excessive disturbance (e.g., light, noise, vibrations or visitation) could result in arousal from torpor which in turn could cause a cascade of arousals in nearby bats and/or could cause the abandonment of a site. Repeated visits over several consecutive days can have the most severe impacts. | May result in temporary destruction of habitat by reducing the functionality of the hibernacula to provide for the survival of bats during overwintering periods. Arousals from excessive disturbance causes increased fat consumption (and premature energy depletion), starvation, reduced energy reserves for reproduction, and ultimately, death. |
| Quarrying, mining exploration and development, agriculture, forestry, dam construction, and other industrial practices that cause, exacerbate, or accelerate blockages of airflow or flooding of hibernaculaFootnoteg | May result in permanent or temporary direct destruction of habitat and/or indirect effects (e.g., changes to microclimatic characteristics, ceiling or wall collapse, limit access to site). |
8. Measuring Progress
The performance indicators presented below provide a way to define and measure progress toward achieving the population and distribution objectives.
Little Brown Myotis and Northern Myotis
- Little Brown Myotis’ and Northern Myotis’ extent of occurrence is maintained (or where applicable restored to) the pre-WNS extent (to be verified every 5 years).
- In the short-term (12-18 years) within WNS-affected areas, the population of each species is maintained (and where feasible increased) at its current (2015) level.
- In the long-term (many generations) within WNS-affected areas, the population of each species is self-sustaining, resilient, and redundant.
- Within areas not yet affected by WNS, the population of each species is maintained (and where feasible increased) at its current level (to be verified every 5 years).
Tri-colored Bat
- Tri-colored Bat’s extent of occurrence is restored (then maintained) to the pre-WNS extent of occurrence (to be verified every 5 years).
- In the short-term (next 10 years), the population is maintained (and where feasible increased) at its current (2015) level.
- In the long-term (many generations), the population is self-sustaining, resilient, and redundant.
9. Statement on Action Plans
One or more action plans for Little Brown Myotis, Northern Myotis, and Tri-colored Bat will be posted on the Species at Risk Public Registry within 3 years of the final posting of the recovery strategy.
10. References
Abbott, I. M., A. Berthinussen, E. Stone, M. Boonman, M. Melber, and J. Altringham. 2015. Bats and Roads. Pages 290-299 In R. van der Ree, D. J. Smith, and C. Grilo (eds.). Handbook of Road Ecology. John Wiley & Sons, Ltd. Chichester, UK.
Abbott, I. M., S. Harrison, and F. Butler. 2012. Clutter-adaptation of bat species predicts their use of under-motorway passageways of contrasting sizes – a natural experiment. Journal of Zoology 287(2): 124-132.
Adams, R. A. 2010. Bat reproduction declines when conditions mimic climate change projections for western North America. Ecology 91(8): 2437-2445.
Adams, R. A. and M. A. Hayes. 2008. Water availability and successful lactation by bats as related to climate change in arid regions of western North America. Journal of Animal Ecology 77(6): 1115-1121.
Amelon, S. and D. Burhans. 2006. Conservation assessment: Myotis septentrionalis (northern long-eared bat) in the Eastern U.S.. Pages 69-82 In F. Thompson (ed.). Conservation Assessements of Five Forest Bat Species in Eastern U.S.. United States Department of Agriculture Forest Service General Technical Report. NC.
Ancillotto, L., M. T. Serangeli, and D. Russo. 2013. Curiosity killed the bat: domestic cats as bat predators. Mammalian Biology 78(5): 369-373.
Anderson, D. M., P. M. Glibert, and J. M. Burkholder. 2002. Harmful algal blooms and eutrophication: nutrient sources, composition and consequences. Estuaries 25(4): 704-726.
Anderson, J. and C. Robert. 1971. A new unipolar electrode for electrocardiography in small mammals. Journal of Mammalogy 52: 469-471.
Anonymous. 2008. Sarissa Trip Report - Chapleau / Nemegosenda Property. [accessed: 21 July 2015].
Anthony, E. L. and T. H. Kunz. 1977. Feeding strategies of the little brown bat, Myotis lucifugus, in southern New Hampshire. Ecology: 775-786.
Arlettaz, R., P. Christe, A. Lugon, N. Perrin, and P. Vogel. 2001. Food availability dictates the timing of parturition in insectivorous mouse-eared bats. Oikos 95(1): 105-111.
Arnett, E., C. D. Hein, M. Schirmacher, M. Huso, and J. M. Szewczak. 2013a. Evaluating the effectiveness of an ultrasonic acoustic deterrent for reducing bat fatalities at wind turbines. PLoS One 8(9): e65794.
Arnett, E., G. Johnson, W. Erickson, and C. Hein. 2013b. A synthesis of operational mitigation studies to reduce bat fatalities at wind energy facilities in North America. National Renewable Energy Laboratory, Austin, TX.
Arnett, E. B., W. Brown, W. P. Erickson, J. K. Fiedler, B. L. Hamilton, T. H. Henry, A. Jain, G. D. Johnson, J. Kerns, and R. R. Koford. 2008. Patterns of bat fatalities at wind energy facilities in North America. The Journal of Wildlife Management 72(1): 61-78.
Arnett, E. B., M. Schirmacher, M. Huso, and J. Hayes. 2009. Effectiveness of changing wind turbine cut-in speed to reduce bat fatalities at wind facilities. Bats and Wind Energy Cooperative, Bat Conservation International, Austin, TX.
Baerwald, E. F., G. H. D'Amours, B. J. Klug, and R. M. Barclay. 2008. Barotrauma is a significant cause of bat fatalities at wind turbines. Current Biology 18(16): R695-R696.
Baerwald, E. F., J. Edworthy, M. Holder, and R. M. Barclay. 2009. A large-scale mitigation experiment to reduce bat fatalities at wind energy facilities. The Journal of Wildlife Management 73(7): 1077-1081.
Barbour, R. W. and W. H. Davis. 1969. Bats of America. University Press of Kentucky. Lexington, KY. 286 pp.
Barclay, R. and A. Kurta. 2007. Ecology and behaviour of bats roosting in tree cavities and under bark. Pages 17-60 In M. J. Lacki, J. P. Hayes, and A. Kurta (eds.). Bats in Forests: Conservation and Management. John Hopkins University Press. Baltimore, MD.
Barclay, R. R. and R. M. Brigham. 1996. Bats and Forests Symposium. British Columbia, Ministry of Forests Research Program. Victoria, BC.
BatsLive. 2015. Project EduBat. [accessed: 18 June 2015].
Bennett, V. J. and A. A. Zurcher. 2013. When corridors collide: road-related disturbance in commuting bats. The Journal of Wildlife Management 77(1): 93-101.
Boldogh, S., D. Dobrosi, and P. Samu. 2007. The effects of the illumination of buildings on house-dwelling bats and its conservation consequences. Acta Chiropterologica 9(2): 527-534.
Borkin, K., C. O’Donnell, and S. Parsons. 2011. Bat colony size reduction coincides with clear-fell harvest operations and high rates of roost loss in plantation forest. Biodiversity and Conservation 20(14): 3537-3548.
Both, C., C. A. Van Turnhout, R. G. Bijlsma, H. Siepel, A. J. Van Strien, and R. P. Foppen. 2009. Avian population consequences of climate change are most severe for long-distance migrants in seasonal habitats. Proceedings of the Royal Society B: Biological Sciences 277: 1259-1266.
Boyles, J. G. and V. Brack. 2009. Modeling survival rates of hibernating mammals with individual-based models of energy expenditure. Journal of Mammalogy 90(1): 9-16.
Briggler, J. T. and J. W. Prather. 2003. Seasonal use and selection of caves by the eastern pipistrelle bat (Pipistrellus subflavus). The American Midland Naturalist 149(2): 406-412.
Brigham, R. M. and M. B. Fenton. 1986. The influence of roost closure on the roosting and foraging behaviour of Eptesicus fuscus (Chiroptera: Vespertilionidae). Canadian Journal of Zoology 64(5): 1128-1133.
Broders, H. G., L. E. Burns, and S. C. McCarthy. 2013. First records of the Northern Myotis (Myotis septentrionalis) from labrador and summer distribution records and biology of little brown bats (Myotis lucifugus) in southern Labrador. Canadian Field-Naturalist 127(3): 266-269.
Broders, H. G., L. J. Farrow, R. N. Hearn, L. M. Lawrence, and G. J. Forbes. 2014. Stable isotopes reveal that little brown bats have a broader dietary niche than northern long-eared bats. Acta Chiropterologica 16(2): 315-325.
Broders, H. G. and G. J. Forbes. 2004. Interspecific and intersexual variation in roost-site selection of northern long-eared and little brown bats in the Greater Fundy National Park ecosystem. Journal of Wildlife Management 68(3): 602-610.
Broders, H. G., G. J. Forbes, S. Woodley, and I. D. Thompson. 2006. Range extent and stand selection for roosting and foraging in forest-dwelling Northern Long-Eared Bats and Little Brown Bats in the Greater Fundy Ecosystem, New Brunswick. The Journal of Wildlife Management 70(5): 1174-1184.
Broders, H. G., D. F. McAlpine, and G. J. Forbes. 2001. Status of the eastern pipistrelle (Pipistrellus subflavus) (Chiroptera: Vespertilionidae) in New Brunswick. Northeastern Naturalist 8(3): 331-336.
Broders, H. G., G. M. Quinn, and G. J. Forbes. 2003. Species status, and the spatial and temporal patterns of activity of bats in southwest Nova Scotia, Canada. Northeastern Naturalist 10(4): 383-398.
Brown, J. A., D. F. McAlpine, and R. Curley. 2007. Northern Long-eared Bat, Myotis septentrionalis (Chiroptera: Vespertilionidae), on Prince Edward Island: first records of occurrence and over-wintering. The Canadian Field-Naturalist 121(2): 208-209.
Bunkley, J. P., C. J. McClure, N. J. Kleist, C. D. Francis, and J. R. Barber. 2015. Anthropogenic noise alters bat activity levels and echolocation calls. Global Ecology and Conservation 3: 62-71.
Burles, D. W., M. B. Fenton, R. M. Barclay, R. M. Brigham, and D. Volkers. 2014. Aspects of the winter ecology of bats on Haida Gwaii, British Columbia. Northwestern Naturalist 95(3): 289-299.
Burns, L. E., T. R. Frasier, and H. G. Broders. 2014. Genetic connectivity among swarming sites in the wide ranging and recently declining little brown bat (Myotis lucifugus). Ecology and Evolution 4(21): 4130-4149.
Caceres, M. C. and R. M. Barclay. 2000. Myotis septentrionalis. Mammalian Species 634: 1-4.
Calvert, A. M., C. A. Bishop, R. D. Elliot, E. A. Krebs, T. M. Kydd, C. S. Machtans, and G. J. Robertson. 2013. A synthesis of human-related avian mortality in Canada. Avian Conservation and Ecology 8(2): 11.
Canadian Wildlife Federation. 2015. Help the Bats. Kanata, ON. [accessed: 19 June 2015].
Canadian Wildlife Health Cooperative. 2015a. Bat White Nose Syndrome. [accessed: 22 April 2015].
Canadian Wildlife Health Cooperative. 2015b. A national plan to manage White Nose Syndrome in bats in Canada. Canadian Wildlife Health Cooperative: Canada's Inter-agency White Nose Syndrome Committee, Saskatoon, SK.
Carr, J. A., R. F. Bernard, and W. H. Stiver. 2014. Unusual bat behavior during winter in Great Smoky Mountains National Park. Southeastern Naturalist 13(2): N18-N21.
Carter, T. C. and G. A. Feldhamer. 2005. Roost tree use by maternity colonies of Indiana bats and northern long-eared bats in southern Illinois. Forest Ecology and Management 219(2–3): 259-268.
Chaverri, G. and T. H. Kunz. 2011. Response of a specialist bat to the loss of a critical resource. PLoS One 6(12): e28821.
Chruszcz, B. and R. Barclay. 2002. Thermoregulatory ecology of a solitary bat, Myotis evotis, roosting in rock crevices. Functional Ecology 16(1): 18-26.
Clare, E. L., W. O. C. Symondson, H. Broders, F. Fabianek, E. E. Fraser, A. MacKenzie, A. Boughen, R. Hamilton, C. K. R. Willis, F. Martinez-Nuñez, A. K. Menzies, K. J. O. Norquay, M. Brigham, J. Poissant, J. Rintoul, R. M. R. Barclay, and J. P. Reimer. 2014. The diet of Myotis lucifugus across Canada: assessing foraging quality and diet variability. Molecular Ecology 23(15): 3618-3632.
Clark, D. R. and T. G. Lamont. 1976. Organochlorine residues in females and nursing young of the big brown bat (Eptesicus fuscus). Bulletin of Environmental Contamination and Toxicology 15(1): 1-8.
Coleman, J. L. and R. M. Barclay. 2011. Influence of urbanization on demography of little brown bats (Myotis lucifugus) in the prairies of North America. PLoS One 6(5): e20483.
Community Bat Programs of BC. 2014. Community Bat Programs Help People and Bats. [accessed: 22 April 2015].
Cornelison, C. T., M. K. Keel, K. T. Gabriel, C. K. Barlament, T. A. Tucker, G. E. Pierce, and S. A. Crow. 2014. A preliminary report on the contact-independent antagonism of Pseudogymnoascus destructans by Rhodococcus rhodochrous strain DAP96253. BMC Microbiology 14(1): 246.
COSEWIC. 2006. COSEWIC assessment and status report on the Rusty Blackbird Euphagus carolinus in Canada. Committee on the Status of Endangered Wildlife in Canada, Ottawa, ON.
COSEWIC. 2013. COSEWIC assessment and status report on the Little Brown Myotis Myotis lucifugus, Northern Myotis Myotis septentrionalis and Tri-colored Bat Perimyotis subflavus in Canada. Committee on the Status of Endangered Wildlife in Canada, Ottawa, ON.
Crampton, L. and R. Barclay. 1996. Habitat selection by bats in fragmented and unfragmented aspen mixedwood stands of different ages Page 292 In M. Brigham and R. Barclay, eds. Bats and Forests Symposium. BC Ministry of Forests Victoria, BC.
Cryan, P. M. 2011. Wind turbines as landscape impediments to the migratory connectivity of bats. Environmental Law 41: 355.
Cryan, P. M. and A. C. Brown. 2007. Migration of bats past a remote island offers clues toward the problem of bat fatalities at wind turbines. Biological Conservation 139(1): 1-11.
Cryan, P. M., C. U. Meteyer, J. G. Boyles, and D. S. Blehert. 2010. Wing pathology of white-nose syndrome in bats suggests life-threatening disruption of physiology. BMC Biology 8(1): 135.
CWHC. 2015. Canadian Wildlife Health Cooperative. CWHC, Saskatoon, SK. [accessed: 19 July 2015].
Damm, J. P. and K. Geluso. 2008. Use of a mine by eastern pipistrelles (Perimyotis subflavus) in east central Nebraska. Western North American Naturalist 68(3): 382-389.
Darling, S. and R. Smith. 2011. Assessment of Vermont cave bat populations and proposal to list bat species. Vermont Fish and Wildlife Department, VT.
Davis, W. and H. Hitchcock. 1995. A new longevity record for the bat Myotis lucifugus. Bat Research News 36(1): 6.
Davis, W. H. 1970. Hibernation: ecology and physiological ecology. Biology of Bats 1: 265-300.
de Groot, W. J., M. D. Flannigan, and A. S. Cantin. 2013. Climate change impacts on future boreal fire regimes. Forest Ecology and Management 294: 35-44.
Depew, D. C., N. M. Burgess, and L. M. Campbell. 2013. Modelling mercury concentrations in prey fish: derivation of a national-scale common indicator of dietary mercury exposure for piscivorous fish and wildlife. Environmental Pollution 176: 234-243.
Derusseau, S. N. and N. J. Huntly. 2012. Effects of gates on the nighttime use of mines by bats in northern Idaho. Northwestern Naturalist 93(1): 60-66.
Diamond, G. F. and J. M. Diamond. 2014. Bats and mines: evaluating Townsend's Big-Eared Bat (Corynorhinus townsendii) maternity colony behavioral response to gating. Western North American Naturalist 74(4): 416-426.
Dirzo, R., H. S. Young, M. Galetti, G. Ceballos, N. J. Isaac, and B. Collen. 2014. Defaunation in the Anthropocene. Science 345(6195): 401-406.
Dodd, L. E., M. J. Lacki, E. R. Britzke, D. A. Buehler, P. D. Keyser, J. L. Larkin, A. D. Rodewald, T. B. Wigley, P. B. Wood, and L. K. Rieske. 2012. Forest structure affects trophic linkages: how silvicultural disturbance impacts bats and their insect prey. Forest Ecology and Management 267: 262-270.
Douglas, M. R. and J. F. Tooker. 2015. Large-scale deployment of seed treatments has driven rapid increase in use of neonicotinoid insecticides and preemptive pest management in US field crops. Environmental Science & Technology 49(8): 5088-5097.
Dubois, J. E. and K. M. Monson. 2007. Recent distribution records of the Little Brown Bat, Myotis lucifugus, in Manitoba and Northwestern Ontario. The Canadian Field-Naturalist 121(1): 57-61.
Dzal, Y., L. A. Hooten, E. L. Clare, and M. Brock-Fenton. 2009. Bat activity and genetic diversity at Long Point, Ontario, an important bird stopover site. Acta Chiropterologica 11(2): 307-315.
Dzal, Y., L. P. McGuire, N. Veselka, and M. B. Fenton. 2011. Going, going, gone: the impact of white-nose syndrome on the summer activity of the little brown bat (Myotis lucifugus). Biology Letters 7(3): 392-394.
Edmonds, S. T., D. C. Evers, D. A. Cristol, C. Mettke-Hofmann, L. L. Powell, A. J. McGann, J. W. Armiger, O. P. Lane, D. F. Tessler, and P. Newell. 2010. Geographic and seasonal variation in mercury exposure of the declining Rusty Blackbird. The Condor 112(4): 789-799.
Eidels, R. R., J. O. Whitaker Jr, and D. W. Sparks. 2007. Insecticide residues in bats and guano from Indiana. Proceedings of the Indiana Academy of Science 116(1): 50-57.
Ellison, L., T. O’Shea, M. Bogan, A. Everette, and D. Schneider. 2003. Existing data on colonies of bats in the U.S.: summary and analysis of the US Geological Survey’s bat population database. Pages 127-237 In T. J. O'Shea and M. A. Bogan (eds.). Monitoring trends in bat populations of the United States and territories: problems and prospects. U.S. Geological Survey. Fort Collins, CO.
Entwistle, A. C., S. Harris, A. M. Hutson, P. Racey, A. Walsh, S. D. Gibson, I. Hepburn, and J. Johnston. 2001. Habitat management for bats: a guide for land managers, land owners and their advisors. Joint Nature Conservation Committee, Peterborough, UK.
Environment Canada. 2011. Presence and levels of priority pesticides in selected Canadian aquatic ecosystems. Water Science and Technology Directorate, Ottawa, ON.
Environment Canada. 2015a. Recovery Strategy for Canada Warbler (Cardellina canadensis) in Canada [Proposed]. Ottawa, ON.
Environment Canada. 2015b. Recovery Strategy for the Common Nighthawk (Chordeiles minor) in Canada [Proposed]. Ottawa, ON.
Ethier, K. and L. Fahrig. 2011. Positive effects of forest fragmentation, independent of forest amount, on bat abundance in eastern Ontario, Canada. Landscape Ecology 26(6): 865-876.
Fabianek, F., D. Gagnon, and M. Delorme. 2011. Bat distribution and activity in Montréal Island green spaces: responses to multi-scale habitat effects in a densely urbanized area. Ecoscience 18(1): 9-17.
Fabianek, F., M. A. Simard, E. B. Racine, and A. Desrochers. 2015. Selection of roosting habitat by male Myotis bats in a boreal forest. Canadian Journal of Zoology 93(7): 539.
Farrow, L. J. and H. G. Broders. 2011. Loss of forest cover impacts the distribution of the forest-dwelling tri-colored bat (Perimyotis subflavus). Mammalian Biology 76(2): 172-179.
Federal Provincial and Territorial Governments of Canada. 2010. Canadian Biodiversity: Ecosystem Status and Trends 2010. Ottawa, ON. [accessed: October 2015].
Fenton, M. B. 1969. Summer activity of Myotis lucifugus (Chiroptera: Vespertilionidae) at hibernacula in Ontario and Quebec. Canadian Journal of Zoology 47(4): 597-602.
Fenton, M. B. 1970. A technique for monitoring bat activity with results obtained from different environments in southern Ontario. Canadian Journal of Zoology 48(4): 847-851.
Fenton, M. B. and R. M. Barclay. 1980. Myotis lucifugus. Mammalian Species 142: 1-8.
Fitzgerald, W. F., D. R. Engstrom, R. P. Mason, and E. A. Nater. 1998. The case for atmospheric mercury contamination in remote areas. Environmental Science & Technology 32(1): 1-7.
Flannigan, M., B. Stocks, M. Turetsky, and M. Wotton. 2009. Impacts of climate change on fire activity and fire management in the circumboreal forest. Global Change Biology 15(3): 549-560.
Fleming, R., J.-N. Candau, and R. McAlpine. 2002. Landscape-scale analysis of interactions between insect defoliation and forest fire in central Canada. Climatic Change 55(1-2): 251-272.
Foster, R. and A. Kurta. 1999. Roosting ecology of the northern bat (Myotis septentrionalis) and comparisons with endangered Indiana bat (Myotis sodalis). Journal of Mammalogy 80: 659-672.
Francl, K. E., W. M. Ford, D. W. Sparks, and V. Brack Jr. 2012. Capture and reproductive trends in summer bat communities in West Virginia: assessing the impact of white-nose syndrome. Journal of Fish and Wildlife Management 3(1): 33-42.
Frank, C. L., A. Michalski, A. A. McDonough, M. Rahimian, R. J. Rudd, and C. Herzog. 2014. The resistance of a North American bat species (Eptesicus fuscus) to white-nose syndrome (WNS). PLoS One 9(12): e113958.
Frank, K. D. 1988. Impact of outdoor lighting on moths: an assessment. Journal of the Lepidopterists' Society 42(2): 63-93.
Fraser, E. E., L. P. McGuire, J. L. Eger, F. J. Longstaffe, and M. B. Fenton. 2012. Evidence of latitudinal migration in tri-colored bats, Perimyotis subflavus. PLoS One 7(2): e31419.
Frick, W. F., J. F. Pollock, A. C. Hicks, K. E. Langwig, D. S. Reynolds, G. G. Turner, C. M. Butchkoski, and T. H. Kunz. 2010a. An emerging disease causes regional population collapse of a common North American bat species. Science 329(5992): 679-682.
Frick, W. F., D. S. Reynolds, and T. H. Kunz. 2010b. Influence of climate and reproductive timing on demography of little brown myotis Myotis lucifugus. Journal of Animal Ecology 79(1): 128-136.
Fujita, M. S. and T. H. Kunz. 1984. Pipistrellus subflavus. Mammalian Species 228: 1-6.
Fuller, N. W., J. D. Reichard, M. L. Nabhan, S. R. Fellows, L. C. Pepin, and T. H. Kunz. 2011. Free-ranging little brown myotis (Myotis lucifugus) heal from wing damage associated with white-nose syndrome. EcoHealth 8(2): 154-162.
Furlonger, C., H. Dewar, and M. Fenton. 1987. Habitat use by foraging insectivorous bats. Canadian Journal of Zoology 65(2): 284-288.
Gaisler, J., V. Hanak, and I. Horacek. 1981. Remarks on the current status of bat populations in Czechoslovakia. Myotis 18(19): 68-75.
Garroway, C. J. and H. G. Broders. 2008. Day roost characteristics of northern long-eared bats (Myotis septentrionalis) in relation to female reproductive status. Ecoscience 15(1): 89-93.
Gaston, K. J., J. Bennie, T. W. Davies, and J. Hopkins. 2013. The ecological impacts of nighttime light pollution: a mechanistic appraisal. Biological Reviews 88(4): 912-927.
Gibbons, D., C. Morrissey, and P. Mineau. 2015. A review of the direct and indirect effects of neonicotinoids and fipronil on vertebrate wildlife. Environmental Science and Pollution Research 22(1): 103-118.
Girardin, M. P., A. A. Ali, C. Carcaillet, O. Blarquez, C. Hély, A. Terrier, A. Genries, and Y. Bergeron. 2013. Vegetation limits the impact of a warm climate on boreal wildfires. New Phytologist 199(4): 1001-1011.
GNWT. 2015a. NWT Species at Risk - Little Brown Myotis. Government of Northwest Territories, Yellowknife, NT. [accessed: 21 July 2015].
GNWT. 2015b. NWT Species at Risk - Northern Myotis. Government of the Northwest Territories, Yellowknife, NT. [accessed: 22 July 2015].
Goulson, D. 2013. Review: An overview of the environmental risks posed by neonicotinoid insecticides. Journal of Applied Ecology 50(4): 977-987.
Griffin, D. 1940. Migrations of New England bats. Bulletin of the Museum of Comparative Zoology 86: 217-246.
Grindal, S. D. and R. M. Brigham. 1998. Short-term effects of small-scale habitat disturbance on activity by insectivorous bats. The Journal of Wildlife Management: 996-1003.
Grindal, S. D. and R. M. Brigham. 1999. Impacts of forest harvesting on habitat use by foraging insectivorous bats at different spatial scales. Ecoscience: 25-34.
Grindal, S. D., C. I. Stefan, and C. Godwin-Sheppard. 2011. Diversity, distribution, and relative abundance of bats in the oil sands regions of northeastern Alberta. Northwestern Naturalist 92(3): 211-220.
Grodsky, S. M., M. J. Behr, A. Gendler, D. Drake, B. D. Dieterle, R. J. Rudd, and N. L. Walrath. 2011. Investigating the causes of death for wind turbine-associated bat fatalities. Journal of Mammalogy 92(5): 917-925.
Haak, D. 2008. Sustainable Agriculture Land Management around Wetlands on the Canadian Prairies.In A. a. A.-F. Canada, ed. Agriculture and Agri-Food Canada, Ottawa, ON.
Hallegraeff, G. M. 1993. A review of harmful algal blooms and their apparent global increase. Phycologia 32(2): 79-99.
Harrison, J. B. 1965. Temperature effects on responses in the auditory system of the little brown bat Myotis l. lucifugus. Physiological Zoology: 34-48.
Harvey, M. J. 1992. Bats of the eastern United States. Arkansas Game and Fish Commission and United States Fish and Wildlife Service, Center for the Management, Utilization, and Protection of Water Resources, Tennessee Technological University, Cookeville, TN.
Harvey, M. J., J. S. Altenbach, and T. L. Best. 2011. Bats of the United States and Canada. Johns Hopkins University Press. Baltimore, MD. 202 pp.
Hayes, M. A. 2013. Bats killed in large numbers at United States wind energy facilities. BioScience 63(12): 975-979.
Hayhoe, K., C. P. Wake, T. G. Huntington, L. Luo, M. D. Schwartz, J. Sheffield, E. Wood, B. Anderson, J. Bradbury, and A. DeGaetano. 2007. Past and future changes in climate and hydrological indicators in the US Northeast. Climate Dynamics 28(4): 381-407.
Hemmera. 2011. Raptor and Migratory Bird and Bat Monitoring and Follw-up Report 2010 and Recommendations for 2011. Hemmera, Vancouver, BC.
Henderson, L. E. and H. G. Broders. 2008. Movements and resource selection of the Northern Long-Eared Myotis (Myotis septentrionalis) in a forest-agriculture landscape. Journal of Mammalogy 89(4): 952-963.
Henderson, L. E., L. J. Farrow, and H. G. Broders. 2008. Intra-specific effects of forest loss on the distribution of the forest-dependent northern long-eared bat (Myotis septentrionalis). Biological Conservation 141(7): 1819-1828.
Henderson, L. E., L. J. Farrow, and H. G. Broders. 2009. Summer distribution and status of the bats of Prince Edward Island, Canada. Northeastern Naturalist 16(1): 131-140.
Henry, M., D. W. Thomas, R. Vaudry, and M. Carrier. 2002. Foraging distances and home range of pregnant and lactating Little Brown Bats (Myotis lucifugus). Journal of Mammalogy 83(3): 767-774.
Henshaw, R. E. and G. E. Folk. 1966. Relation of thermoregulation to seasonally changing microclimate in two species of bats (Myotis lucifugus and M. sodalis). Physiological Zoology: 223-236.
Hitchcock, H. B. 1949. Hibernation of bats in southeastern Ontario and adjacent Quebec. Canadian Field-Naturalist 63(1): 47-59.
Hitchcock, H. B. 1965. Twenty-three years of bat banding in Ontario and Quebec. Canadian Field-Naturalist 79: 4-14.
Hobson, K. A., E. M. Bayne, and S. L. Van Wilgenburg. 2002. Large-scale conversion of forest to agriculture in the boreal plains of Saskatchewan. Conservation Biology 16(6): 1530-1541.
Hogberg, L. K., K. J. Patriquin, and R. M. Barclay. 2002. Use by bats of patches of residual trees in logged areas of the boreal forest. The American Midland Naturalist 148(2): 282-288.
Hölker, F., C. Wolter, E. K. Perkin, and K. Tockner. 2010. Light pollution as a biodiversity threat. Trends in Ecology & Evolution 25(12): 681-682.
Horacek, I., J. Zukal, T. Bartonika, and N. Martinkova. 2012. Ecology of geomycosis in central Europe. 5th Annual White-nose Syndrome Symposium, Madison, WI.
Horn, J. W., E. B. Arnett, and T. H. Kunz. 2008. Behavioral responses of bats to operating wind turbines. The Journal of Wildlife Management 72(1): 123-132.
Horváth, G., G. Kriska, P. Malik, and B. Robertson. 2009. Polarized light pollution: a new kind of ecological photopollution. Frontiers in Ecology and the Environment 7(6): 317-325.
Hoyt, J. R., T. L. Cheng, K. Langwig, M. M. Hee, W. F. Frick, and A. M. Kilpatrick. 2015. Bacteria isolated from bats inhibit the growth of Pseudogymnoascus destructans, the causative agent of white-nose syndrome. PLoS One 10(4): e0121329.
Hoyt, J. R., K. E. Langwig, J. Okoniewski, W. F. Frick, W. B. Stone, and A. M. Kilpatrick. 2014. Long-term persistence of Pseudogymnoascus destructans, the causative agent of white-nose syndrome, in the absence of bats. EcoHealth: 1-4.
Humphries, M. M., D. W. Thomas, and J. R. Speakman. 2002. Climate-mediated energetic constraints on the distribution of hibernating mammals. Nature 418(6895): 313-316.
Huntington, T. G., A. D. Richardson, K. J. McGuire, and K. Hayhoe. 2009. Climate and hydrological changes in the northeastern United States: recent trends and implications for forested and aquatic ecosystems. Canadian Journal of Forest Research 39(2): 199-212.
Huynh, H. M. 2009. Another record of foliage roosting in the Little Brown Bat, Myotis lucifugus, in Canada. The Canadian Field-Naturalist 123(3): 265.
Johnson, L. N., B. A. McLeod, L. E. Burns, K. Arseneault, T. R. Frasier, and H. G. Broders. 2015. Population genetic structure within and among seasonal site types in the little brown bat (Myotis lucifugus) and the northern long-eared bat (M. septentrionalis). PLoS One 10(5): e0126309.
Jonasson, K. A. and C. K. Willis. 2011. Changes in body condition of hibernating bats support the thrifty female hypothesis and predict consequences for populations with white-nose syndrome. PLoS One 6(6): e21061.
Jones, G., D. S. Jacobs, T. H. Kunz, M. R. Willig, and P. A. Racey. 2009. Carpe noctem: the importance of bats as bioindicators. Endangered Species Research 8(1-2): 93-115.
Jones, G. and H. Rebelo. 2013. Responses of bats to climate change: learning from the past and predicting the future. Pages 457-478 In R. A. Adams and S. C. Pedersen (eds.). Bat Evolution, Ecology, and Conservation. Springer.
Jung, T. S., K. M. Blejwas, C. L. Lausen, J. M. Wilson, and L. E. Olson. 2014. Concluding remarks: what do we need to know about bats in northwestern North America? Northwestern Naturalist 95(3): 318-330.
Jung, T. S., B. G. Slough, D. W. Nagorsen, T. A. Dewey, and T. Powell. 2006. First records of the Northern Long-eared Bat, Myotis septentrionalis, in the Yukon Territory. The Canadian Field-Naturalist 120(1): 39-42.
Jung, T. S., I. D. Thompson, and R. D. Titman. 2004. Roost site selection by forest-dwelling male Myotis in central Ontario, Canada. Forest Ecology and Management 202(1): 325-335.
Jung, T. S., I. D. Thompson, R. D. Titman, and A. P. Applejohn. 1999. Habitat selection by forest bats in relation to mixed-wood stand types and structure in central Ontario. The Journal of Wildlife Management: 1306-1319.
Kalcounis-Rüppell, M. C., J. M. Psyllakis, and R. M. Brigham. 2005. Tree roost selection by bats: an empirical synthesis using meta-analysis. Wildlife Society Bulletin 33(3): 1123-1132.
Kannan, K., S. H. Yun, R. J. Rudd, and M. Behr. 2010. High concentrations of persistent organic pollutants including PCBs, DDT, PBDEs and PFOS in little brown bats with white-nose syndrome in New York, USA. Chemosphere 80(6): 613-618.
Karouna-Renier, N. K., C. White, C. R. Perkins, J. J. Schmerfeld, and D. Yates. 2014. Assessment of mitochondrial DNA damage in little brown bats (Myotis lucifugus) collected near a mercury-contaminated river. Ecotoxicology 23(8): 1419-1429.
Kilpatrick, M. 2013. Transmission of Geomyces destructans between bats and the environment. International Bat Research Conference San Jose, Costa Rica.
Kitzes, J. and A. Merenlender. 2014. Large roads reduce bat activity across multiple species. PLoS One 9(5): e96341.
Krusic, R. A., M. Yamasaki, C. D. Neefus, and P. J. Pekins. 1996. Bat habitat use in white mountain national forest. The Journal of Wildlife Management: 625-631.
Kuiper-Goodman, T., I. Falconer, and J. Fitzgerald. 1999. Human health aspects. Pages 113-153 In I. Chorus and J. Bartram (eds.). Toxic Cyanobacteria in Water, A Guide to Their Public Health Consequences, Monitoring and Management. Spon Press. New York, Ny.
Kunz, T. and M. Tuttle. 2009. White-nose syndrome science strategy meeting II., Austin, TX.
Kunz, T. H. 1973. Resource utilization: temporal and spatial components of bat activity in central Iowa. Journal of Mammalogy: 14-32.
Kunz, T. H. 1982. Roosting ecology in bats. Pages 1-56 In T. H. Kunz (ed.). Ecology of Bats. Plenum Press. New York, NY.
Kunz, T. H., E. B. Arnett, W. P. Erickson, A. R. Hoar, G. D. Johnson, R. P. Larkin, M. D. Strickland, R. W. Thresher, and M. D. Tuttle. 2007. Ecological impacts of wind energy development on bats: questions, research needs, and hypotheses. Frontiers in Ecology and the Environment 5(6): 315-324.
Kunz, T. H. and J. D. Reichard. 2010. Status review of the little brown myotis (Myotis lucifugus) and determination that immediate listing under the Endangered Species Act is scientifically and legally warranted. Boston University, Boston, MA.
Kunz, T. H. and D. S. Reynolds. 2003. Bat colonies in buildings. Pages 91-102 In T. J. O'Shea and M. A. Bogan, eds. Monitoring Trends in Bat Populations of the United States and Territories: Problems and Prospects. US Geological Survey, Springfield, VA.
Kunz, T. H., J. A. Wrazen, and C. D. Burnett. 1998. Changes in body mass and fat reserves in prehibernating little brown bats (Myotis lucifugus). Ecoscience: 8-17.
Kurta, A. 2014. The misuse of relative humidity in ecological studies of hibernating bats. Acta Chiropterologica 16(1): 249-254.
Kurta, A. and S. M. Smith. 2014. Hibernating bats and abandoned mines in the Upper Peninsula of Michigan. Northeastern Naturalist 21(4): 587-605.
Kyba, C. C., T. Ruhtz, J. Fischer, and F. Hölker. 2011. Cloud coverage acts as an amplifier for ecological light pollution in urban ecosystems. PLoS One 6(3): e17307-e17307.
Lacki, M. J., D. R. Cox, L. E. Dodd, and M. B. Dickinson. 2009. Response of Northern Bats (Myotis septentrionalis) to prescribed fires in Eastern Kentucky forests. Journal of Mammalogy 90(5): 1165-1175.
Lacoeuilhe, A., N. Machon, J.-F. Julien, A. Le Bocq, and C. Kerbiriou. 2014. The influence of low intensities of light pollution on bat communities in a semi-natural context. PLoS One 9(10): e103042.
Langwig, K., J. Voyles, M. Q. Wilber, W. F. Frick, K. A. Murray, B. M. Bolker, J. P. Collins, T. L. Cheng, M. C. Fisher, J. R. Hoyt, D. L. Lindner, H. I. McCallum, R. Puschendorf, E. B. Rosenblum, M. Toothman, C. K. R. Willis, C. J. Briggs, and A. M. Kilpatrick. 2015a. Context dependent conservation responses to emerging wildlife diseases. Frontiers in Ecology and the Environment 13: 195-202.
Langwig, K. E., W. F. Frick, J. T. Bried, A. C. Hicks, T. H. Kunz, and A. Marm Kilpatrick. 2012. Sociality, density-dependence and microclimates determine the persistence of populations suffering from a novel fungal disease, white-nose syndrome. Ecology Letters 15(9): 1050-1057.
Langwig, K. E., W. F. Frick, R. Reynolds, K. L. Parise, K. P. Drees, J. R. Hoyt, T. L. Cheng, T. H. Kunz, J. T. Foster, and A. M. Kilpatrick. 2015b. Host and pathogen ecology drive the seasonal dynamics of a fungal disease, white-nose syndrome. Proceedings of the Royal Society of London B: Biological Sciences 282(1799): 20142335.
Lausen, C. L., T. S. Jung, and J. M. Talerico. 2008. Range extension of the northern long-eared bat (Myotis septentrionalis) in the Yukon. Northwestern Naturalist 89(2): 115-117.
Leopardi, S., D. Blake, and S. J. Puechmaille. 2015. White-Nose Syndrome fungus introduced from Europe to North America. Current Biology 25(6): R217-R219.
Lesiński, G., A. Sikora, and A. Olszewski. 2011. Bat casualties on a road crossing a mosaic landscape. European Journal of Wildlife Research 57(2): 217-223.
Lindner, D. L., A. Gargas, J. M. Lorch, M. T. Banik, J. Glaeser, T. H. Kunz, and D. S. Blehert. 2011. DNA-based detection of the fungal pathogen Geomyces destructans in soils from bat hibernacula. Mycologia 103(2): 241-246.
Little, M. E., N. M. Burgess, H. Broders, and L. M. Campbell. 2015a. Distribution of mercury in archived fur from little brown bats across Atlantic Canada. Environmental Pollution 207: 52-28.
Little, M. E., N. M. Burgess, H. G. Broders, and L. M. Campbell. 2015b. Mercury in little brown bat (Myotis lucifugus) maternity colonies and its correlation with freshwater acidity in Nova Scotia, Canada. Environmental Science & Technology 49(4): 2059-2065.
Loeb, S. C., T. J. Rodhouse, L. E. Ellison, C. L. Lausen, J. D. Reichard, K. M. Irvine, T. E. Ingersoll, J. T. Coleman, W. E. Thogmartin, J. R. Sauer, C. M. Francis, M. L. Bayless, T. R. Stanley, and D. H. Johnson. 2015. A Plan for the North American Bat Monitoring Program (NABat). United States Department of Agriculture: Forest Service Research & Development, Asheville, NC.
Lorch, J. M., C. U. Meteyer, M. J. Behr, J. G. Boyles, P. M. Cryan, A. C. Hicks, A. E. Ballmann, J. T. Coleman, D. N. Redell, and D. M. Reeder. 2011. Experimental infection of bats with Geomyces destructans causes white-nose syndrome. Nature 480(7377): 376-378.
Lorch, J. M., L. K. Muller, R. E. Russell, M. O'Connor, D. L. Lindner, and D. S. Blehert. 2013. Distribution and environmental persistence of the causative agent of white-nose syndrome, Geomyces destructans, in bat hibernacula of the eastern United States. Applied and Environmental Microbiology 79(4): 1293-1301.
Loss, S. R., T. Will, and P. P. Marra. 2013. The impact of free-ranging domestic cats on wildlife of the United States. Nature 4: 1396.
Loyd, K. A. T., S. M. Hernandez, J. P. Carroll, K. J. Abernathy, and G. J. Marshall. 2013. Quantifying free-roaming domestic cat predation using animal-borne video cameras. Biological Conservation 160(0): 183-189.
MacGregor, C. J., M. J. Pocock, R. Fox, and D. M. Evans. 2015. Pollination by nocturnal Lepidoptera, and the effects of light pollution: a review. Ecological Entomology 40(3): 187-198.
Maher, S. P., A. M. Kramer, J. T. Pulliam, M. A. Zokan, S. E. Bowden, H. D. Barton, K. Magori, and J. M. Drake. 2012. Spread of white-nose syndrome on a network regulated by geography and climate. Nature 3: 1306.
Main, A. R., J. V. Headley, K. M. Peru, N. L. Michel, A. J. Cessna, and C. A. Morrissey. 2014. Widespread use and frequent detection of neonicotinoid insecticides in wetlands of Canada's prairie pothole region. PLoS One 9(3): e92821.
Mainguy, J. and N. Desrosiers. 2011. Cave-dwelling bats in the province of Quebec: historical data about hibernacula population surveys. Ministère des Ressources naturelles et de la Faune, QC.
Masek, J. G., W. B. Cohen, D. Leckie, M. A. Wulder, R. Vargas, B. de Jong, S. Healey, B. Law, R. Birdsey, and R. Houghton. 2011. Recent rates of forest harvest and conversion in North America. Journal of Geophysical Research: Biogeosciences (2005–2012) 116(G4).
Maslo, B., M. Valent, J. F. Gumbs, and W. F. Frick. 2015. Conservation implications of ameliorating survival of little brown bats with white-nose syndrome. Ecological Applications 25(7): 1832-1840.
Mason, R., H. Tennekes, F. Sánchez-Bayo, and P. U. Jepsen. 2013. Immune suppression by neonicotinoid insecticides at the root of global wildlife declines. Journal of Environmental Immunology Toxicology 1(1): 3-12.
Mathews, F., N. Roche, T. Aughney, N. Jones, J. Day, J. Baker, and S. Langton. 2015. Barriers and benefits: implications of artificial night-lighting for the distribution of common bats in Britain and Ireland. Philosophical Transactions of the Royal Society of London B: Biological Sciences 370(1667): 20140124.
Mattson, W. J. and R. A. Haack. 1987. The role of drought in outbreaks of plant-eating insects. BioScience: 110-118.
McAlpine, D. F. 1983. Status and conservation of solution caves in New Brunswick. The New Brunswick Museum, St. John, NB.
McCracken, G. F. 2011. Cave conservation: special problems of bats. Pages 68-95 In J. Tyburec, J. Chenger, T. Snow, and C. Geiselman, eds. Bat Conservation International: Bat Conservation and Management Workshop. Bat Conservation International, Portal, AZ.
McLeod, B. A., L. E. Burns, T. Frasier, and H. Broders. 2015. Effect of oceanic straits on gene flow in the recently endangered little brown bat (Myotis lucifugus) in maritime Canada: implications for the spread of white-nose syndrome. Canadian Journal of Zoology 93(999): 427-437.
Medinas, D., J. T. Marques, and A. Mira. 2013. Assessing road effects on bats: the role of landscape, road features, and bat activity on road-kills. Ecological Research 28(2): 227-237.
Meteyer, C. U., E. L. Buckles, D. S. Blehert, A. C. Hicks, D. E. Green, V. Shearn-Bochsler, N. J. Thomas, A. Gargas, and M. J. Behr. 2009. Histopathologic criteria to confirm white-nose syndrome in bats. Journal of Veterinary Diagnostic Investigation 21(4): 411-414.
Moosman, P. R., H. H. Thomas, and J. P. Veilleux. 2012. Diet of the widespread insectivorous bats Eptesicus fuscus and Myotis lucifugus relative to climate and richness of bat communities. Journal of Mammalogy 93(2): 491-496.
Morrissey, C. A., P. Mineau, J. H. Devries, F. Sanchez-Bayo, M. Liess, M. C. Cavallaro, and K. Liber. 2015. Neonicotinoid contamination of global surface waters and associated risk to aquatic invertebrates: A review. Environment International 74: 291-303.
Murphy, M. 1987. Vandals destroy hibernating Indiana Bats. BATS Magazine 5(2): 5-8.
Nagorsen, D. and R. Brigham. 1993. Bats of British Columbia: Royal British Columbia museum handbook. University of British Columbia Press, Vancouver, BC.
NASBR. 2015. North American Society for Bat Research. Ypsilanti, MI. [accessed: 1 October 2015].
Natureserve. 2015. NatureServe Explorer: An online encyclopedia of life [web application]. Version 7.0. Natureserve, Arlington, VA. U.S.A. [accessed: 20 April 2015].
Naughton, D. 2012. The Natural History of Canadian Mammals. University of Toronto Press. Toronto, ON. 784 pp.
Neilson, A. L. and M. B. Fenton. 1994. Responses of little brown myotis to exclusion and to bat houses. Wildlife Society Bulletin: 8-14.
NFD. 2014. National Forestry Database. Natural Resources Canada and Canadian Forest Service, Ottawa, ON. [accessed: 20 August 2014].
Norquay, K. J. O., F. Martinez-Nunez, J. E. Dubois, K. M. Monson, and C. K. R. Willis. 2013. Long-distance movements of little brown bats (Myotis lucifugus). Journal of Mammalogy 94(2): 506-515.
Northeast Bat Working Group. 2015. Northeast Bat Working Group. [accessed: 22 April 2015].
O'Regan, S. M., K. Magori, J. T. Pulliam, M. A. Zokan, R. R. B. Kaul, H. D. Barton, and J. M. Drake. 2014. Multi-scale model of epidemic fadeout: Will local extirpation events inhibit the spread of white-nose syndrome? Ecological Applications 25: 621-633.
O’Donoghue, A. J., G. M. Knudsen, C. Beekman, J. A. Perry, A. D. Johnson, J. L. DeRisi, C. S. Craik, and R. J. Bennett. 2015. Destructin-1 is a collagen-degrading endopeptidase secreted by Pseudogymnoascus destructans, the causative agent of white-nose syndrome. Proceedings of the National Academy of Sciences: 201507082.
Okoniewski, J., J. Haines, A. Hicks, K. Langwig, R. VonLinden, and C. Dobony. 2010. Detection of the conidia of Geomyces destructans in northeast hibernacula, at maternal colonies, and on gear – some findings based on microscopy and culture. Page 18 WNS Symposium, Pittsburg, Pennsylvania.
Olson, C. R. 2011. The roosting behaviour of Little Brown Bats (Myotis lucifugus) and Northern Long-eared Bats (Myotis septentrionalis) in the boreal forest of northern Alberta. University of Calgary, Calgary, AB.
Olson, C. R. and R. M. Barclay. 2013. Concurrent changes in group size and roost use by reproductive female little brown bats (Myotis lucifugus). Canadian Journal of Zoology 91(3): 149-155.
Olson, C. R., D. P. Hobson, and M. J. Pybus. 2011. Changes in population size of bats at a hibernaculum in Alberta, Canada, in relation to cave disturbance and access restrictions. Northwestern Naturalist 92(3): 224-230.
OMNRF. 2015. Ontario: Species at Risk. [accessed: 3 May 2015].
Ontario Ministry of Natural Resources. 2010. Forest Management Guide for Conserving Biodiversity at the Stand and Site Scales. Ontario Ministry of Natural Resources, Ontario.
Ontario Ministry of Natural Resources and Forestry. 2015. Ontario's White-nose Syndrome Response Plan. Wildlife Section, Species Conservation Policy Branch, Ontario.
Owen, S. F., M. A. Menzel, W. M. Ford, B. R. Chapman, K. V. Miller, J. W. Edwards, and P. B. Wood. 2003. Home-range size and habitat used by the northern myotis (Myotis septentrionalis). The American Midland Naturalist 150(2): 352-359.
Park, A. C. and H. G. Broders. 2012. Distribution and roost selection of bats on Newfoundland. Northeastern Naturalist 19(2): 165-176.
Patriquin, K. J., F. Palstra, M. L. Leonard, and H. G. Broders. 2013. Female northern myotis (Myotis septentrionalis) that roost together are related. Behavioral Ecology 24(4): 949-954.
Pauli, B., P. Zollner, G. Haulton, and G. Shao. 2015. The simulated effects of timber harvest on suitable habitat for Indiana and northern long-eared bats.
Perkin, E. K., F. Hölker, and K. Tockner. 2014. The effects of artificial lighting on adult aquatic and terrestrial insects. Freshwater Biology 59(2): 368-377.
Perry, R. and R. Thill. 2007. Tree roosting by male and female eastern pipistrelles in a forested landscape. Journal of Mammalogy 88: 974-981.
Pikula, J., H. Bandouchova, L. Novotný, C. U. Meteyer, J. Zukal, N. R. Irwin, J. Zima, and N. Martínková. 2012. Histopathology confirms white-nose syndrome in bats in Europe. Journal of Wildlife Diseases 48(1): 207-211.
Poissant, J. 2009. Roosting and social ecology of the tri-colored bat, Perimyotis subflavus, in Nova Scotia. St. Mary's University, Halifax, NS.
Poissant, J. A., H. G. Broders, and G. M. Quinn. 2010. Use of lichen as a roosting substrate by Perimyotis subflavus, the tricolored bat, in Nova Scotia. Ecoscience 17(4): 372-378.
Porvari, P., M. Verta, J. Munthe, and M. Haapanen. 2003. Forestry practices increase mercury and methyl mercury output from boreal forest catchments. Environmental Science & Technology 37(11): 2389-2393.
Powers, L., E. Pritchard, K. Lau, and B. Francis. 2012. Does Geomyces destructans infection reduce female fertility in Myotis lucifugus? . North America Symposium on Bat Research, Puerto Rico.
Pybus, M., D. Hobson, and D. Onderka. 1986. Mass mortality of bats due to probable blue-green algal toxicity. Journal of Wildlife Diseases 22(3): 449-450.
Quinn, G. M. and H. G. Broders. 2007. Roosting and foraging ecology of eastern pipistrelle (Perimyotis subflavus) bats in SW Nova Scotia. Nova Scotia Habitat Conservation Fund c/o NS Department of Natural Resources, Halifax, NS.
Raesly, R. L. and J. E. Gates. 1987. Winter habitat selection by north temperate cave bats. American Midland Naturalist: 15-31.
Randall, J. and H. G. Broders. 2014. Identification and characterization of swarming sites used by bats in Nova Scotia, Canada. Acta Chiropterologica 16(1): 109-116.
Randall, L. A., T. S. Jung, and R. M. Barclay. 2014. Roost-site selection and movements of Little Brown Myotis (Myotis lucifugus) in southwestern Yukon. Northwestern Naturalist 95(3): 312-317.
Ratcliffe, J. M. and J. W. Dawson. 2003. Behavioural flexibility: the little brown bat, Myotis lucifugus, and the northern long-eared bat, M. septentrionalis, both glean and hawk prey. Animal Behaviour 66(5): 847-856.
Raudabaugh, D. B. and A. N. Miller. 2015. Effects of trans, trans-farnesol on Pseudogymnoascus destructans and several closely related species. Mycopathologia.
Reeder, D. and G. Turner. 2008. Working together to combat white-nose syndrome: a report of a meeting on 9–11 June 2008. Bat Research News 49(7).
Reeder, D. M., C. L. Frank, G. G. Turner, C. U. Meteyer, A. Kurta, E. R. Britzke, M. E. Vodzak, S. R. Darling, C. W. Stihler, and A. C. Hicks. 2012. Frequent arousal from hibernation linked to severity of infection and mortality in bats with white-nose syndrome. PLoS One 7(6): e38920.
Reichard, J. D., N. W. Fuller, A. B. Bennett, S. R. Darling, M. S. Moore, K. E. Langwig, E. D. Preston, S. v. Oettingen, C. S. Richardson, and D. Scott Reynolds. 2014. Interannual survival of Myotis lucifugus (Chiroptera: Vespertilionidae) near the epicenter of white-nose syndrome. Northeastern Naturalist 21(4): N56-N59.
Reichard, J. D. and T. H. Kunz. 2009. White-nose syndrome inflicts lasting injuries to the wings of little brown myotis (Myotis lucifugus). Acta Chiropterologica 11(2): 457-464.
Reimer, J. and L. Kaupas. 2013. South Slave region bat research summary 2013: University of Calgary. Environment and Natural Resources, Government of Northwest Territiories, Fort Smith, NT.
Reimer, J. P. 2013. Nocturnality under the mignight sun: behavioural adaptations of the little brown bat (Myotis lucifugus) at 60° N latitude, Calgary, AB.
Ren, P., K. H. Haman, L. A. Last, S. S. Rajkumar, M. K. Keel, and V. Chaturvedi. 2012. Clonal spread of Geomyces destructans among bats, Midwestern and Southern United States. Emergent Infectious Disease 18(5): 883-885.
Reynolds, D. S. 2006. Monitoring the potential impact of a wind development site on bats in the northeast. Journal of Wildlife Management 70(5): 1219-1227.
Rollins, K. E., D. K. Meyerholz, G. D. Johnson, A. P. Capparella, and S. S. Loew. 2012. A forensic investigation into the etiology of bat mortality at a wind farm: barotrauma or traumatic injury? Veterinary Pathology Online 49(2): 362-371.
Russell, A., M. Vonhof, and E. Claire. 2012. Phylogeographic analyses reveal cryptic subdivisions and unexpected connections among Myotis lucifugus populations. North America Symposium on Bat Research, Puerto Rico.
Rydell, J., L. Bach, P. Bach, L. G. Diaz, J. Furmankiewicz, N. Hagner-Wahlsten, E.-M. Kyheröinen, T. Lilley, M. Masing, and M. M. Meyer. 2014. Phenology of migratory bat activity across the Baltic Sea and the south-eastern North Sea. Acta Chiropterologica 16(1): 139-147.
Sandel, J. K., G. R. Benatar, K. M. Burke, C. W. Walker, J. Lacher, Thomas E, and R. L. Honeycutt. 2001. Use and selection of winter hibernacula by the eastern pipistrelle (Pipistrellus subflavus) in Texas. Journal of Mammalogy 82(1): 173-178.
Sasse, D. and P. Perkins. 1996. Summer roosting ecology of northern long-eared bats (Myotis septentrionalis) in the White Mountain National Forest. Pages 91-101 In Bats and Forests Symposium. British Columbia Ministry of Forests Victoria, BC.
Scott, F. W. and A. J. Hebda. 2004. Annotated list of the mammals of Nova Scotia. Proceedings of the Nova Scotian Institute of Science (NSIS) 42(2).
Segers, J. and H. Broders. 2014. Interspecific effects of forest fragmentation on bats. Canadian Journal of Zoology 92(8): 665-673.
Segers, J. L. and H. G. Broders. 2015. Carbon (δ 13 C) and nitrogen (δ 15 N) stable isotope signatures in bat fur indicate swarming sites have catchment areas for bats from different summering areas. PLoS One 10(4): e0125755.
Sherwin, H. A., W. I. Montgomery, and M. G. Lundy. 2013. The impact and implications of climate change for bats. Mammal Review 43(3): 171-182.
Silvis, A., W. M. Ford, and E. R. Britzke. 2015a. Day-roost tree selection by northern long-eared bats--What do non-roost tree comparisons and one year of data really tell us? Global Ecology and Conservation 3(0): 756-763.
Silvis, A., W. M. Ford, and E. R. Britzke. 2015b. Effects of hierarchical roost removal on Northern Long-Eared Bat (Myotis septentrionalis) maternity colonies. PLoS One 10(1).
Sivonen, K. and G. Jones. 1999. Cyanobacterial toxins. Pages 41-111 In I. Chorus and J. Bartram (eds.). Toxic Cyanobacteria in Water, a Guide to Their Public Health Consequences, Monitoring and Management. Spon Press. New York, NY.
Slough, B. G. 2009. Behavioral thermoregulation by a maternity colony of little brown bats in the Yukon. Northwestern Naturalist: 47-51.
Slough, B. G. and T. S. Jung. 2007. Diversity and distribution of the terrestrial mammals of the Yukon Territory: A review. The Canadian Field-Naturalist 121(2): 119-127.
Slough, B. G. and T. S. Jung. 2008. Observations on the natural history of bats in the Yukon. Northern Review(29): 127-150.
Smallwood, K. S. 2013. Comparing bird and bat fatality-rate estimates among North American wind-energy projects. Wildlife Society Bulletin 37(1): 19-33.
Smith, J. L., G. L. Boyer, E. Mills, and K. L. Schiulz. 2008. Toxicity of microcystin-LR, a cyanobacterial toxin, to multiple life stages of the burroeing mayfly, Hexagenia, and possible implications for recruitment. Environmental Toxicoloogy 23(4): 499-506.
Spanjer, G. R. and M. B. Fenton. 2005. Behavioral responses of bats to gates at caves and mines. Wildlife Society Bulletin 33(3): 1101-1112.
Sparks, T. C. 2013. Insecticide discovery: an evaluation and analysis. Pesticide biochemistry and physiology 107(1): 8-17.
Speakman, J., P. Webb, and P. Racey. 1991. Effects of disturbance on the energy expenditure of hibernating bats. Journal of Applied Ecology: 1087-1104.
SSM. 2015. Speleological Society of Manitoba. Speleological Society of Manitoba. [accessed: 16 July 2015].
Stone, E. L., G. Jones, and S. Harris. 2009. Street lighting disturbs commuting bats. Current Biology 19(13): 1123-1127.
Swystun, M. B., J. M. Psyllakis, and R. M. Brigham. 2001. The influence of residual tree patch isolation on habitat use by bats in central British Columbia. Acta Chiropterologica 3(2): 197-201.
Syme, D. M., M. B. Fenton, and J. Zigouris. 2001. Roosts and food supplies ameliorate the impact of a bad summer on reproduction by the bat, Myotis lucifugus LeConte (Chiroptera: Vespertilionidae). Ecoscience: 18-25.
Talerico, J. M. 2008. The behaviour, diet and morphology of the little brown bat (Myotis lucifugus) near the northern extent of its range in Yukon Canada. University of Calgary, Calgary, AB.
Thomas, D. W. 1995. Hibernating bats are sensitive to nontactile human disturbance. Journal of Mammalogy 76(3): 940-946.
Thomas, D. W., M. Dorais, and J.-M. Bergeron. 1990. Winter energy budgets and cost of arousals for hibernating little brown bats, Myotis lucifugus. Journal of Mammalogy: 475-479.
Thomas, H. H., P. R. Moosman, J. P. Veilleux, and J. Holt. 2012. Foods of bats (Family Vespertilionidae) at five locations in New Hampshire and Massachusetts. The Canadian Field-Naturalist 126(2): 117-124.
Thorne, T. J. 2015. The role of islands in the migration of bats across Lake Erie and Lake Ontario: Lasiurus borealis, Lasiurus cinereus and Perimyotis subflavus. The University of Western Ontario, London, ON.
Trombulak, S. C., P. E. Higuera, and M. DesMeules. 2001. Population trends of wintering bats in Vermont. Northeastern Naturalist 8(1): 51-62.
Turner, G. G., D. Reeder, and J. T. Coleman. 2011. A five-year assessment of mortality and geographic spread of white-nose syndrome in North American bats, with a look at the future. Update of white-nose syndrome in bats. Bat Research News: 13.
Turner, R. W. 1974. Mammals of the Black Hills of South Dakota and Wyoming. University of Kansas. Lawrence, KS. pp.
U.S. Fish and Wildlife Service. 2007. Indiana Bat (Myotis sodalis) draft recovery plan: first revision. U.S. Fish and Wildlife Service, Fort Snelling, MN.
U.S. Fish and Wildlife Service. 2012. North American bat death toll exceeds 5.5 million from white-nose syndrome. U.S. Fish and Wildlife Service, Arlington, VA.
U.S. Fish and Wildlife Service. 2015. White-nose Syndrome. Hadley, MA. [accessed: 3 May 2015].
U.S. Geological Survey. 2000. Mercury in the environment, fact sheet 146-00. [accessed: August 2014].
van Zyll de Jong, C. G. 1985. Handbook of Canadian Mammals: Bats. National Museum of Natural Sciences. Ottawa, ON. 212 pp.
Vanderwolf, K. J., D. F. McAlpine, G. J. Forbes, and D. Malloch. 2012. Bat populations and cave microclimate prior to and at the outbreak of white-nose syndrome in New Brunswick. The Canadian Field-Naturalist 126(2): 125-134.
Veilleux, J. P. and S. L. Veilleux. 2004. Intra-annual and interannual fidelity to summer roost areas by female eastern pipistrelles, Pipistrellus subflavus. The American Midland Naturalist 152(1): 196-200.
Veilleux, J. P., J. O. Whitaker Jr, and S. L. Veilleux. 2003. Tree-roosting ecology of reproductive female eastern pipistrelles, Pipistrellus subflavus, in Indiana. Journal of Mammalogy 84(3): 1068-1075.
Verant, M. L., M. U. Carol, J. R. Speakman, P. M. Cryan, J. M. Lorch, and D. S. Blehert. 2014. White-nose syndrome initiates a cascade of physiologic disturbances in the hibernating bat host. BMC Physiology 14(1): 10.
Vuori, K.-M., O. Siren, and H. Luotonen. 2003. Metal contamination of streams in relation to catchment silvicultural practices: a comparative study in Finnish and Russian headwaters. Boreal Environment Research 8(1): 61-70.
Warnecke, L., J. M. Turner, T. K. Bollinger, J. M. Lorch, V. Misra, P. M. Cryan, G. Wibbelt, D. S. Blehert, and C. K. Willis. 2012. Inoculation of bats with European Geomyces destructans supports the novel pathogen hypothesis for the origin of white-nose syndrome. Proceedings of the National Academy of Sciences 109(18): 6999-7003.
Warnecke, L., J. M. Turner, T. K. Bollinger, V. Misra, P. M. Cryan, D. S. Blehert, G. Wibbelt, and C. K. Willis. 2013. Pathophysiology of white-nose syndrome in bats: a mechanistic model linking wing damage to mortality. Biology Letters 9(4): 20130177.
Webb, P., J. Speakman, and P. Racey. 1995. Evaporative water loss in two sympatric species of vespertilionid bat, Plecotus auritus and Myotis daubenton i: relation to foraging mode and implications for roost site selection. Journal of Zoology 235(2): 269-278.
Western Bat Working Group. 2015. Western Bat Working Group. [accessed: 22 April 2015].
Whitaker, J. O. and L. J. Rissler. 1993. Do bats feed in winter? American Midland Naturalist: 200-203.
Wibbelt, G., A. Kurth, D. Hellmann, M. Weishaar, A. Barlow, M. Veith, J. Prüger, T. Görföl, L. Grosche, and F. Bontadina. 2010. White-nose syndrome fungus (Geomyces destructans) in bats, Europe. Emerging Infectious Diseases 16(8): 1237.
Wickramasinghe, L. P., S. Harris, G. Jones, and N. Vaughan. 2003. Bat activity and species richness on organic and conventional farms: impact of agricultural intensification. Journal of Applied Ecology 40(6): 984-993.
Wiener, J. G., D. P. Krabbenhoft, G. H. Heinz, and A. M. Scheuhammer. 2003. Ecotoxicology of mercury. Pages 409-463 In D. J. Hoffman, B. A. Rattner, G. A. Burton, Jr., and J. Cairns, Jr. (eds.). Handbook of Ecotoxicology, 2nd Edition. CRC Press. Boca Raton, FL.
Wilder, A. P. 2014. Spatial, ecological and genetic correlates of the geographic expansion of an infectious disease, white-nose syndrome in bats. University of California, Santa Cruz, CA.
Willis, C. K. and R. M. Brigham. 2007. Social thermoregulation exerts more influence than microclimate on forest roost preferences by a cavity-dwelling bat. Behavioral Ecology and Sociobiology 62(1): 97-108.
Wilson, J. M. and R. M. Barclay. 2006. Consumption of caterpillars by bats during an outbreak of western spruce budworm. The American Midland Naturalist 155(1): 244-249.
Wilson, J. M., J. P. Reimer, D. Allaire, and C. L. Lausen. 2014. Diversity and distribution of bats in the Northwest Territories. Northwestern Naturalist 95(3): 197-218.
Woller-Skar, M. M., D. N. Jones, M. R. Luttenton, and A. L. Russell. 2015. Mycrocystin detected in little brown bats (Myotis lucifugus). The American Midland Naturalist 174(2): 331-334.
Xing, Z., L. Chow, H. Rees, F. Meng, S. Li, B. Ernst, G. Benoy, T. Zha, and L. M. Hewitt. 2013. Influences of sampling methodologies on pesticide-residue detection in stream water. Archives of Environmental Contamination and Toxicology 64(2): 208-218.
Yates, D. E., E. M. Adams, S. E. Angelo, D. C. Evers, J. Schmerfeld, M. S. Moore, T. H. Kunz, T. Divoll, S. T. Edmonds, and C. Perkins. 2014. Mercury in bats from the northeastern United States. Ecotoxicology 23(1): 45-55.
Young, J. E., G. A. Sánchez-Azofeifa, S. J. Hannon, and R. Chapman. 2006. Trends in land cover change and isolation of protected areas at the interface of the southern boreal mixedwood and aspen parkland in Alberta, Canada. Forest Ecology and Management 230(1–3): 151-161.
Zimmerman, G. S. and W. E. Glanz. 2000. Habitat use by bats in eastern Maine. The Journal of Wildlife Management 64(4): 1032-1040.
Personal Communications
Blejwas, K. 2015. Regional Wildlife Biologist, Alaska Department of Fish and Game, Juneau, AK.
Brigham, M. 2015. Professor, University of Regina, Regina, SK.
Broders, H. 2015. Associate Professor, St. Mary’s University, Halifax, NS.
Govindarajulu, P. 2015. Amphibian, Reptile, and Small Mammal Specialist, British Columbia Ministry of Environment, Victoria, BC.
Jung, T. 2015. Senior Wildlife Biologist, Whitehorse, YK.
Lausen, C. 2015. Birchdale Ecological Ltd, Kaslo, BC.
Morningstar, D. 2015. Terrestrial Ecologist, Golder Associates Ltd, Cambridge, ON.
Pardy Moores, S. 2015. Senior Manager – Endangered Species and Biodiversity, Department of Environment and Conservation, Cornerbrook, NL.
Appendix A: Effects on the Environment and Other Species
A strategic environmental assessment (SEA) is conducted on all SARA recovery planning documents, in accordance with the Cabinet Directive on the Environmental Assessment of Policy, Plan and Program Proposals. The purpose of a SEA is to incorporate environmental considerations into the development of public policies, plans, and program proposals to support environmentally sound decision-making and to evaluate whether the outcomes of a recovery planning document could affect any component of the environment or any of the Federal Sustainable Development Strategy’s (FSDS) goals and targets.
Recovery planning is intended to benefit species at risk and biodiversity in general. However, it is recognized that strategies may also inadvertently lead to environmental effects beyond the intended benefits. The planning process based on national guidelines directly incorporates consideration of all environmental effects, with a particular focus on possible impacts upon non-target species or habitats. The results of the SEA are incorporated directly into the strategy itself, but are also summarized below in this statement.
WNS affects other bat species not considered in this recovery strategy (e.g., Eastern Small-footed Myotis – Myotis leibii) (U.S. Fish and Wildlife Service 2015). Therefore, any approaches that mitigate the impact or spread of WNS will most likely also benefit these species. In contrast, it is possible that other species of bats with populations not heavily impacted by WNS (e.g., Big Brown Bat – Eptesicus fuscus) may be benefiting from the declining populations of Little Brown Myotis, Northern Myotis, and Tri-colored Bat by filling the niche recently vacated (Francl et al. 2012). It is unknown how recovery of the three at-risk species will affect these other bat species that have recently increased.
Many potential threats identified in this recovery strategy have also been identified as threats for other species at risk. Approaches that help to minimize these threats may also benefit other species. For example, Rusty Blackbird (Euphagus carolinus) is thought to be susceptible to mercury contamination in Eastern Canada (Edmonds et al. 2010), and feral and free-roaming cats have been identified as a potential threat to numerous bird species (Calvert et al. 2013), including other species at risk, such as Common Nighthawk (Chordeiles minor) (Environment Canada 2015b). Conservation of forests surrounding hibernacula and roosts may positively affect other species (at local scales) that are also threatened by forest removal (e.g., Canada Warbler – Cardellina canadensis) (Environment Canada 2015a).
Bat populations consume substantial quantities of insects each night and therefore control the local populations of insects. Initiatives that result in the recovery of bat populations may cause local declines in insect populations (some of which may have already exhibited drastic declines) (Dirzo et al. 2014). In contrast, strategies that investigate the declining insect prey, or research, mitigate, or educate the public about mutual potential threats may aid in the recovery of possible depleted insect populations.
The possibility that the present recovery strategy inadvertently generates negative effects on the environment and on other species was considered. The majority of recommended actions are non-intrusive in nature, including surveys, research, and outreach. It is unlikely that the present recovery strategy will produce significant negative effects.
Appendix B: Additional Research Needs Related to Known and Suspected Threats
The following list is not exhaustive, but illustrates some of the research required to understand the threats (other than WNS) to Little Brown Myotis, Northern Myotis, and Tri-colored Bat and their habitats.
Disturbance or harm
- Determine the effects of different levels and types of noise on behavior and biology of bats throughout their life-cycles.
- Determine the effects of research activities on bat stress and survival.
- Further investigate the location and characteristics of wind turbines in Canada that may significantly affect bat populations.
- Further investigate the timing and population-level effects of wind turbines.
- Continue to monitor frequency of wind turbine collisions in Canada, and develop a consistent range-wide monitoring program for wind energy facilities.
- Investigate the effects and characteristics of vehicle-bat collisions in Canada.
- Investigate the effects of bat collisions related to non-traditional / recreational vehicles and devices (e.g., boats, unmanned aerial vehicles, and fishing lines).
Habitat loss or degradation
- Determine the effects of common forestry operations (e.g., silviculture, and selective cutting) on roost tree availability, behavior, biology, and movement patterns across the range of the species.
- Determine the amount (and characteristics) of forest removal, harvesting, and silviculture that can be completed while maintaining enough suitable habitat for bat populations across the range of the species.
- Continue to investigate the effects of forest fragmentation from various sources (e.g., agriculture and road development).
- Investigate the significance of habitat loss due to insect outbreaks (e.g., Mountain Pine Beetle).
- Further investigate the effects of exclusion of maternity colonies from anthropogenic structures.
- Further investigate the use of bat boxes to mitigate loss of anthropogenic or natural roosting structures.
Pollution
- Further investigate the bats’ exposure to mercury and other pollutants across their ranges.
- Determine the potential effects of mercury on biology, survival, and behavior.
- Determine the effects of neonicotinoids and other widely-used pesticides on bats.
- Determine the effects of techniques to reduce the spread of spruce budworm on bats.
- Determine the effects of light pollution on bat behavior, foraging efficiency, and their prey.
Problematic non-native species
- Determine human-related predation risk in urban and rural areas (e.g., by cats).
Climate and natural disasters
- Determine the impacts of climate change on these species, their prey, and their habitat.
- Investigate the potential impact of insect outbreaks and forest fires on these species, their prey, and their habitat.