Fact Sheet - Facts Table for Non-prescription Drugs
Backgrounder
June 2016
For any health product you buy for your family, reading the label is important for helping you choose the right product and know how to use it properly and safely. Labels provide important information, but it's not always clearly organized or easy to understand. The new Facts Table for non-prescription drugs aims to make information already found on product labels more uniform, and easier to find, read and understand.
Through Health Canada's Plain Language Labelling Initiative, regulations have been introduced with new industry requirements for health product labelling and packaging, including a requirement for a Facts Table on the outer label of non-prescription drugs. While companies may choose to incorporate the table sooner, the requirement is effective for new products starting June 2017 with all non-prescription products expected to carry a Facts Table by June 30, 2021. The table will apply to thousands of non-prescription drug products in Canada, from cough syrups and allergy remedies to pain and fever relievers, and will be called the "Drug Facts" table.
Companies producing natural health products (e.g., vitamins, herbal products and minerals), while not subject to the regulations, are being provided with guidance for adopting a Facts Table, called the "Product Facts" table, on their product labels as a best practice.
Modelled after Canada's Nutrition Facts table on foods and the Drug Facts box used on non-prescription drugs in the U.S., the new table uses simple language and an easy-to-read format to help consumers:
- compare and choose products
- identify the active ingredient(s) and their purpose, including the amount in each dosage unit which can help avoid the risk of accidentally overdosing by taking more than one product with the same active ingredient
- quickly know what the product is used for
- quickly locate the warnings, including when the product should not be used
- easily locate the directions for use, including when, how and how often to take the product
- identify inactive ingredients, which is important for avoiding potential allergic reactions
- know how to contact the manufacturer with product questions or concerns
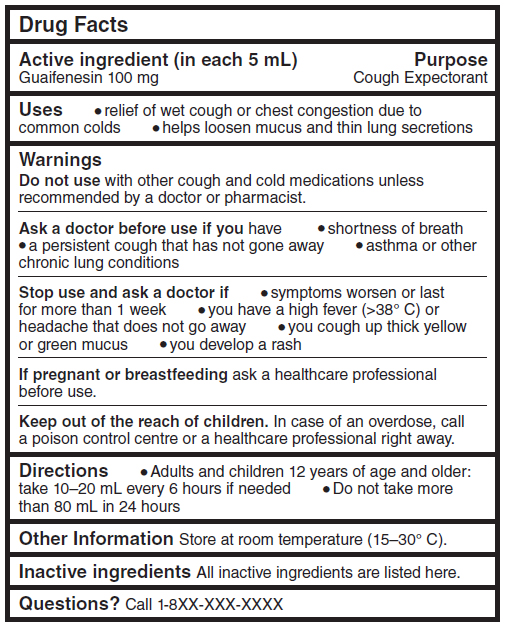
Shown above: Sample Drug Facts table for a product containing the active ingredient guaifenesin
If you read a health product label and still have questions about the product, talk to your pharmacist, doctor, or other health care professional.
Related Links
Search for related information by keyword