Barium in Drinking Water - Guideline Technical Document for Public Consultation

Download the alternative format
(PDF format, 545 KB, 52 pages)
Organization: Health Canada
Date published: 2018-11-23
Consultation period ends February 15, 2019
Table of Contents
- Purpose of consultation
- Part I. Overview and Application
- 1.0 Proposed guideline
- 2.0 Executive summary
- 3.0 Application of the guideline
- Part II. Science and Technical Considerations
- 4.0 Identity, use and sources in the environment
- 5.0 Exposure
- 6.0 Analytical methods
- 7.0 Treatment technology and distribution system considerations
- 8.0 Kinetics and metabolism
- 9.0 Health effects
- 10.0 Classification and assessment
- 11.0 Rationale
- 12.0 References
- Appendix A: List of acronyms
Purpose of consultation
The available information on barium has been assessed with the intent of establishing a drinking water guideline and guideline technical document. This draft guideline technical document proposes a new maximum acceptable concentration (MAC) of 2.0 mg/L (2,000 µg/L) for barium in drinking water, based on kidney effects in male mice.
The document is being made available for a 75-day public consultation period. The purpose of this consultation is to solicit comments on the proposed guideline, on the approach used for its development and on the potential economic costs of implementing it, as well as to determine the availability of additional exposure data. Comments are appreciated, with accompanying rationale, where required. Comments can be sent to Health Canada via email at HC.water-eau.SC@canada.ca. If this is not feasible, comments may be sent by mail to the Water and Air Quality Bureau, Health Canada, 269 Laurier Avenue West, A.L. 4903D, Ottawa, Ontario K1A 0K9. All comments must be received before February 15, 2019.
The existing guideline on barium, last updated in 1990, established a maximum acceptable concentration (MAC) of 1.0 mg/L (1,000 µg/L) based on the absence of adverse effects on blood pressure in humans. This new document provides updated scientific data and information related to the health effects of barium, and now focuses on kidney effects in male mice. It outlines analytical methods and treatment technologies available at the municipal and residential scales.
Comments received as part of this consultation will be shared with members of the Federal-Provincial-Territorial Committee on Drinking Water (CDW), along with the name and affiliation of their author. Authors who do not want their name and affiliation to be shared with CDW should provide a statement to this effect along with their comments.
It should be noted that this guideline technical document on barium in drinking water will be revised following evaluation of comments received, and a drinking water guideline will be established. This document should be considered as a draft for comment only.
Part I. Overview and Application
1.0 Proposed guideline
A maximum acceptable concentration (MAC) of 2.0 mg/L (2,000 µg/L) is proposed for total barium in drinking water.2.0 Executive summary
Barium occurs in various compounds in the environment either naturally or from human activities. While the main use of barium is as a drilling fluid additive in oil and gas exploration, it is also used as a contrast agent in X-ray diagnostic tests and in a wide array of products, including plastics, rubbers, paint, glass, carpets, ceramics, sealants, furniture, fertilizers and pesticides.
Naturally occurring barium can be found in most types of rocks and can enter surface and groundwater by leaching and eroding from sedimentary rocks. A total of over 20 radioactive barium isotopes, with various degrees of stability and radioactivity, have been identified in the environment. However, the focus of this document is limited to barium’s chemical properties.
This guideline technical document reviews and assesses all identified health risks associated with barium in drinking water. It assesses new studies and approaches and takes into consideration the availability of appropriate treatment technology. Based on this review, the proposed guideline for barium in drinking water is a maximum acceptable concentration of 2.0 mg/L.
During its fall 2017 meeting, the Federal-Provincial-Territorial Committee on Drinking Water reviewed the guideline technical document for barium and gave its endorsement for this document to undergo public consultation.
2.1 Health effects
The International Agency for Research on Cancer has not classified barium as to its carcinogenicity. The U.S. Environmental Protection Agency concluded that barium is not likely to be carcinogenic to humans from exposure through ingestion; other international agencies agree that there is no evidence showing that exposure to barium through ingestion could cause cancer.
Studies have found links between the ingestion of barium and unwanted effects on blood pressure in animals and humans. However, adverse effects on the kidneys have shown the strongest association with chronic oral exposure to barium. In humans, effects have been observed on the kidneys following exposure to high levels of barium in poisoning events; in animals, kidney effects are considered the most sensitive health effect associated with long-term ingestion of barium, especially in mice, the most sensitive species. Consequently, the proposed MAC of 2.0 mg/L has been established to be protective of the general population, based on studies of kidney effects in mice.
2.2 Exposure
Canadians are primarily exposed to barium through food and drinking water, with food being the main source of exposure. Concentrations of barium in food items in Canada vary widely, depending on the food item and the soil conditions. Barium levels can also vary greatly in
Canadian drinking water, depending on geological formations and anthropogenic activities surrounding the source water. Intake of barium from drinking water is not expected to occur through either skin contact or inhalation.2.3 Analysis and treatment
Several analytical methods are available for the analysis of total barium in drinking water at levels well below the proposed MAC. Total barium in a water sample includes both its dissolved and particulate forms. Therefore, if the two forms are measured separately, the two concentrations must be added before comparison with the MAC.
Although conventional coagulation treatment is not effective for barium removal, there are several effective methods for its removal from municipal drinking water supplies. These technologies include lime softening and ion exchange softening, as well as high-pressure membrane separation processes such as reverse osmosis and nanofiltration. Other control strategies include switching to a new source, blending, and interconnecting with another water system.
At the residential level, there are certified residential treatment devices for removing barium from drinking water. Drinking water treatment technologies able to effectively remove barium include ion exchange and reverse osmosis. It is important to note that reverse osmosis systems should be installed only at the point of use, as the treated water may be corrosive to internal plumbing components.
2.4 International considerations
Drinking water quality guidelines, standards and/or guidance established by foreign governments or international agencies may vary due to the science available at the time of assessment, as well as the utilization of different policies and approaches, such as the choice of key study, and the use of different consumption rates, body weights and allocation factors.
Other organizations have set guidelines or regulations pertaining to the concentration of barium in drinking water. The World Health Organization established a guideline for drinking- water quality of 1.3 mg/L for barium. The United States Environmental Protection Agency’s standard and the guideline established by the Australia National Health and Medical Research Council are both set at 2.0 mg/L. The European Union has not established a limit for barium in drinking water.
3.0 Application of the guideline
Note: Specific guidance related to the implementation of drinking water guidelines should be obtained from the appropriate drinking water authority in the affected jurisdiction.
For drinking water supplies that occasionally experience short-term exceedances of barium above the guideline value, it is suggested that a plan be developed and implemented to address these situations. For more significant, long-term exceedances that cannot be addressed through treatment, it is suggested that alternative sources of drinking water be considered.
3.1 Monitoring
3.1.1 Source characterization
Source water should be characterized to determine if barium is present. If the barium concentration is approaching the proposed MAC and/or the concentration is known to be or expected to be changing with time (e.g., anthropogenic activities are introduced), monitoring of the source water should be conducted annually. Authorities may consider reduced monitoring when there is sufficient data demonstrating that barium is present at concentrations below 2.0 mg/L in the source water and/or appropriate treatment is in place.
Utilities practising control options for addressing the barium concentration in source water should assess the water quality of new sources to ensure that it does not interfere with the existing treatment processes, impact the distribution system, and/or cause other water quality issues.
3.1.2 Operational monitoring
Utilities that treat their water for barium removal need to conduct frequent monitoring of the treated water in order to make necessary process adjustments and to ensure that treatment processes are effectively removing barium and other co-contaminants below their respective MACs.
Utilities using lime softening for barium removal should conduct operational monitoring of pH. Utilities using ion exchange water softening (i.e., a strong-acid cation exchange resin in sodium form) for hardness and barium removal in their source water should monitor for hardness breakthrough in each ion exchange vessel to identify the timing for resin regeneration and achieve effective barium removal. An operational consideration when using strong-acid cation resins in hydrogen form includes chromatographic peaking. Since barium and calcium are the cations most preferred by these ion exchange resins, chromatographic peaking may be observed for ions such as sodium and magnesium in the product water. The hydrogen form of strong-acid cation and weak-acid cation exchange resins must be followed by a carbon dioxide stripping process and a pH or alkalinity adjustment step to reduce the corrosivity of the product water. Utilities using strong-acid cation exchange resins in sodium form should be aware that this process may introduce undesirable quantities of sodium into the treated water.
Reverse osmosis, ion exchange and electrodialysis/electrodialysis reversal are often operated with a bypass blending of a portion of the influent (incoming) flow with the treated water to obtain the desired water quality. It is important to monitor blended treated water to determine final barium concentrations when this control option is used.
3.1.3 Compliance monitoring
When treatment is in place for barium reduction (including control options), it is recommended that compliance monitoring be conducted annually, at minimum, to confirm that the proposed MAC is not exceeded. Samples should be collected after treatment prior to distribution (typically at the entry point to the distribution system). Paired samples of source and treated water should be taken to confirm the efficacy of the treatment or control option.
3.1.4 Distribution system
Like other inorganics, barium may accumulate in distribution systems and be intermittently released. Consequently, monitoring should also be conducted throughout the distribution system when barium is or was historically present in the source water. Since the stability of metals accumulated in distribution system piping scales is unpredictable, it is difficult to establish a monitoring program for barium in distribution systems. The number and location of barium monitoring sites in the distribution system should take into consideration the site-specific accumulation and release risk factors. Factors that influence barium accumulation (i.e., manganese deposits, iron corrosion products and phosphate precipitates) and remobilization (i.e., changes to water chemistry and physical/hydraulic disturbances in the distribution system) could be used as indicators of when and where to monitor for barium releases.
When water quality changes or physical disruptions occur in the system, the release of barium and other contaminants may be indicated by the presence of discoloured water or increased turbidity resulting from the release of deposits or scales present on the pipe wall. When this occurs, monitoring for barium and other contaminants should be conducted when the system is back to normal operation. However, the absence of discoloured water should not be interpreted as the absence of metal release. Monitoring for barium should be done in conjunction with other metals that can co-occur in the distribution system (e.g., iron, manganese, arsenic, lead).
Homeowners with private wells are encouraged to have their water tested for barium to ensure that the concentration in their water supply is below the MAC. Homeowners with private wells using residential treatment devices should conduct routine testing on both the water entering the treatment device and the treated water to verify that the treatment device is effective.
Homeowners using ion exchange softeners should be aware that the treatment unit may introduce undesirable quantities of sodium into the treated water.
Part II. Science and Technical Considerations
4.0 Identity, use and sources in the environment
Barium (Ba; CAS Registry No. 7440-39-3) is a soft, silvery white element. It is a divalent cation and belongs to the Group IIA (alkaline-earth metal) of the periodic table of elements. It has a specific gravity of 3.6 at 25°C, a melting point of 727°C and a boiling point of 1897°C (CRC, 2017). Barium is widely distributed across the earth's crust (0.04%) with an average concentration of 10–425 mg/kg, and ranks 16th in abundance amongst non-gaseous elements (Taylor, 1964; Schroeder et al., 1972). Coal can contain barium in concentrations of up to 3,000 mg/kg (WHO, 2001; ATSDR, 2007). Naturally occurring barium is a combination of seven stable isotopes. However, more than 20 isotopes have been identified, most of them being highly radioactive and with half-lives ranging from several milliseconds to several minutes (WHO, 1990; Boffito, 1991; U.S. EPA, 2005). Only non-radiological forms of barium will be assessed in this document.
Barium does not occur in its elemental form in nature; it occurs as a divalent cation associated with other elements and is mostly found in igneous, sedimentary and metamorphic rocks (Boffito, 1991). Common barium compounds include barium sulphate (barite), barium carbonate (witherite), barium chloride, barium hydroxide, barium oxide, and barium sulphide. Barium reacts with water to form barium hydroxide. Water-soluble barium compounds include salts of acetate, hydroxide, silicate, chlorate, nitrate, thiocyanate, cyanide, oxide and halides (excluding fluoride); salts of arsenate, carbonate, citrate, fluoride, oxalate, and tartrate are only slightly soluble in water but are soluble in acids. Barium chromate and phosphate are insoluble in water but are soluble in acids, whereas barium sulphate is insoluble in water and is only slightly soluble in acids. All barium salts, excluding sulphate, show increasing solubility with decreasing pH (WHO, 1990). Table 1 provides the physicochemical properties of common barium compounds.
| Substance | Chemical formula | Physical description | Molecular weight (g/mol) | Vapour pressure (mm Hg)Footnote a | Solubility (g/100 g water) |
|---|---|---|---|---|---|
| Barium | Ba | Silvery yellow metal (cubic) | 137 | 6.65 × 10-4 at 630°C |
Reacts with water |
| Barium carbonate | BaCO3 | White orthorhombic crystals | 197.34 | Essentially zero | 0.0014 at 20°C |
| Barium chloride | BaCl2 | White orthorhombic crystals; hygroscopic | 208.3 | Essentially zero | 37.0 at 25°C |
| Barium sulphate | BaSO4 | White orthorhombic crystals | 233.39 | No data | 0.00031 at 20°C |
| Barium hydroxide | Ba(OH)2 | White powder | 171.34 | 0 at 15°C | 4.91 at 25°C |
| Barium oxide | BaO | White, yellowish powder; cubic and hexagonal |
153.33 | Essentially zero | 1.5 at 20°C |
| Barium sulphide | BaS | Colourless cubic crystals or gray powder |
169.39 | No data | 8.94 at 25°C |
Footnotes
|
|||||
The main use of barium (as barite) is as a drilling fluid additive to increase fluid density (hydrostatic pressure) in applications such as oil and gas exploration; this represents 85% of the global use. Barite is also used as a contrast agent to improve the visibility of certain organs in X-ray diagnostic tests; in the chemical sector it is used in plastic and rubber products, paints, glass, carpets, ceramics, sealants, furniture, cement vessels, superconducting films, and nuclear reactors (Dumont, 2007; CCME, 2013). Barium nitrate is used in the manufacturing of pyrotechnics. Barium chloride is used in the production of sodium hydroxide, aluminum alloys, pigments and textiles, and in the treatment of boiler water (Dibello et al., 2003). Barium is also used in some pesticide applications, such as barium metaborate (used as a microbiocide/ microbiostat in paints, paper/paper products, industrial adhesives and coatings), and barium carbonate is used as a rodenticide (U.S. EPA, 1993; ATSDR, 2007).
Barite and witherite are two commercial barium compounds widespread throughout Canada, with more than 150 deposits identified and four main orebodies (Giant Mascot and Mineral King in BC; Buchans in NL; and Walton in NS) (CCME, 2013). Small deposits of witherite have been found in Thunder Bay (Dumont, 2007). In 2007, Canadian production of barite and witherite was estimated to be 7,196 tonnes, while 68,971 tonnes were used in Canada (Dumont, 2007).
4.1 Environmental fate
Barium is naturally present in metamorphic, igneous and sedimentary rocks at a wide range of concentrations; however, anthropogenic activities (such as the dispersal of barium-rich fertilizers and insecticides, drilling mud, and shale gas development) can also increase its concentration. Sulphates and carbonates precipitate barium in the soil, and metal oxides and hydroxides also favour its adsorption onto soil particles. The mobility of barium in soil is generally low, given that it adsorbs easily to clay minerals and organic matter; it tends to form insoluble salts and not soluble humic complexes. Under acidic, anaerobic and high chloride/low sulphate conditions, as well as conditions of reduced reduction–oxidation potential, barium mobility is increased, favouring its migration to groundwater (Kravchenko et al., 2014).
The release of barium into the air can result from electric power generation, heating plant operation, gasoline combustion, metal smelting and kiln operation in cement plants, as well as from mining and refuse incineration (CCME, 2013). Point sources of barium include effluents and cuttings from oil drilling sites, since barite is often used as a main constituent in drilling mud suspensions (Breuer et al., 2004). Barium in the air is associated with particulate matter and mainly occurs in insoluble forms (e.g., sulphate) (Kravchenko et al., 2014).
Naturally occurring barium can be found in igneous, metamorphic and sedimentary rocks. Leaching and eroding of barium from sedimentary rocks can be a substantial source of barium in surface and groundwater; the barium concentrations are determined by dissolved ion concentrations (mainly sulphate and carbonate) and by adsorption of barium ions onto suspended particulate matter, clay, metal oxides, and hydroxides (Taylor, 1964; WHO, 2001; Colbert and McManus, 2005; CCME, 2013). The formation of barium sulphate leads to its precipitation (CCME, 2013). As pH decreases, the sulphur species present in water will be reduced to lower oxidation states, increasing the water solubility of barium compounds, with barium sulphate (BaSO4) and carbonate (BaCO3) being more dependent on pH than barium chloride (BaCl2) (ATSDR, 2007; Kravchenko et al., 2014). The presence of chloride and other anions such as nitrate (NO3) and carbonate (CO32-) generally increases the solubility of barium sulphate, and when present in water with a pH less than 9.3, 98% of barium will be found as the free divalent cation (Ba2+), while less than 2% will be present as other chemical species, including BaB(OH)4+, BaCl+, BaCO3, BaNO3+ and BaOH+ (Venugopal and Luckey, 1978; ATSDR, 2007; Tudorache et al., 2010; CCME, 2013). In water with a pH greater than 9.3, the dominant species of barium is BaCO3, limiting its solubility (ATSDR, 2007). Substantial quantities of barium can be released due to IX reactions or oxide dissolution surrounding deep submarine springs (Charette and Sholkovitz, 2006; Moore, 2010). Barium and barium sulphide (BaS) may be solubilized from barite during anaerobic respiration by sulphate-reducing bacteria (Baldi et al., 1996).
5.0 Exposure
Food and drinking water represent the main sources of exposure to barium for the general population; however, the available data indicate that contributions from these sources can be highly variable. In drinking water, barium content can also vary greatly, depending on the geological formations and the anthropogenic activities surrounding the source water. Exposure from air, consumer products or soil is expected to be negligible. Allocating a 20% source contribution to drinking water is deemed appropriate, given that the available exposure data reported below suggest that food represents the main source of exposure to barium; drinking water represents a significant, but lesser, source.
5.1 Water
In Prince Edward Island, 14 236 samples from private drilled wells and municipal wells using the same aquifer were analyzed for barium over a 7-year period (June 2010 to February 2017) (PEI Department of Communities, Land and Environment, 2017). Of 14,236 samples, 1,006 were equal to or less than the detection limit (DL) of either 10 µg/L or 2 µg/L, depending on when the samples were taken; the mean concentration of barium was 290 µg/L, the median was 17 µg/L and the maximum level detected was 5,452 µg/L.
In Newfoundland and Labrador, barium levels in 483 public drinking water systems were reported from 2012 through 2016 using a DL of 1 µg/L (Newfoundland and Labrador Department of Environment and Conservation, 2017). For raw surface water, 169 out of 762 samples were below the DL, as were 25 out of 101 samples from raw groundwater. Raw surface water had a mean level of 20 µg/L, a median of 10 µg/L and a maximum of 130 µg/L; raw groundwater had a mean and median level of 90 µg/L and a maximum of 560 µg/L. In treated surface water, 2,590 out of 3,259 samples were below the DL; the mean was 20 µg/L, the median was 10 µg/L and the maximum level observed was 140 µg/L. In treated groundwater, 567 out of 1,683 samples were below the DL; the mean was 90 µg/L, the median was 60 µg/L and the maximum level detected was 700 µg/L.
In New Brunswick, barium in drinking water was measured in 231 Crown (groundwater wells only) and 476 municipal water supply systems (ground and surface water) between 2007 and 2017 using a DL of 10 µg/L (New Brunswick Department of Health, 2017). In the Crown systems, 364 out of 1,544 samples were below the DL. In treated groundwater, the mean level found was 111 µg/L, the median was 54 µg/L and the maximum was 1,300 µg/L. In raw groundwater, the mean level was 52 µg/L, the median was 53 µg/L and the maximum was 301 µg/L. In municipal systems, 590 out of 4,203 samples were below the DL. In the municipal water distribution systems, the mean level observed was 70 µg/L, the median was 25 µg/L and the maximum was 3,330 µg/L. In municipal raw water, the mean level found was 120 µg/L, the median was 65 µg/L and the maximum was 986 µg/L. In municipal surface water sources, the mean was 28 µg/L, the median was 15 µg/L and the maximum was 746 µg/L; for groundwater sources, the mean was 126 µg/L, the median was 76 µg/L and the maximum was 986 µg/L.
In Quebec, 11,889 samples of treated drinking water were taken from 3,178 distribution systems between 2013 and 2016 (Ministère du Développement durable, de l’Environnement et de la Lutte contre les changements climatiques, 2017). There were 3,843 samples below the DL of 0.2–50 µg/L (32%); the mean level observed was 69 µg/L, the median was 20 µg/L and the maximum was 11,000 µg/L. As part of a long-term groundwater survey, more than 1,719 barium analyses were conducted between 1971 and 2014 in various regions of Quebec; these barium data are from various research projects with varying analytical methodologies. From a total of 1,490 observation wells, individual wells and drinking water facilities, the mean barium level observed was 153 µg/L and the maximum concentration was 10,000 µg/L.
The Ambient Groundwater Geochemistry project characterizes the chemical state of groundwater for southern Ontario (an area of 96,000 km2), with about 2,300 samples taken between 2007 and 2014 (Hamilton, 2015). There were 40 out of 2,255 samples below the DL of 0.1 µg/L (1.8%); the mean level was 128 µg/L, the median was 64.5 µg/L and the maximum was 4,940 µg/L.
In Manitoba, from 2009 to 2017, 1,478 raw and 1,722 treated water samples were analyzed for total barium using a DL of 0.3 µg/L (Manitoba, 2017). For raw water, the mean level found was 70 µg/L, the median was 30 µg/L and the maximum was 2,473 µg/L; for treated water (which includes distribution system data), the mean was 50 µg/L, the median was 20 µg/L and the maximum was 2,170 µg/L. Manitoba surface waters are monitored through a series of monitoring programs, such as the Long‐term Water Quality Monitoring Program, the Lake Winnipeg Monitoring Program and the Coordinated Aquatic Monitoring Program. Between 2006 and 2017, the mean and median total barium concentrations in surface waters were 52 µg/L and 43 µg/L, respectively, with a maximum value of 1,720 µg/L; only one sample out of 6,777 samples was below the DL of 0.2 µg/L. In northern Manitoba lakes and rivers, an average concentration of less than 10 µg/L (range of 4.6 to 8.7 µg/L; n = 5) has been reported (CCME, 2013).
In Saskatchewan, total drinking water barium levels were reported for 2007–2017 using a DL of 0.1–0.5 µg/L (Saskatchewan Water Security Agency, 2017). Groundwater (raw) (n = 1243) had a mean level of 51 µg/L, a median of 14 µg/L and a maximum of 1,210 µg/L. Surface water (raw) (n = 451) had a mean concentration of 51 µg/L, a median of 46 µg/L and a maximum of 920 µg/L. Treated water (n = 223) had a mean of 78.3 µg/L, a median of 64 µg/L and a maximum of 1,440 µg/L.
An average concentration of 91 µg/L was reported for five river basins in Alberta (range of 7 to 389 µg/L; n = 748) (CCME, 2013).
In British Columbia, barium is not included in the typical drinking water testing; however, the Interior Health Authority has reported 11 elevated barium results ranging from 1,500 to 106,000 µg/L in six treated drinking water sites (British Columbia Ministry of Health, 2016).
In Yukon, levels of barium in drinking water were reported for samples taken from 2009 to 2017 (Yukon Environmental Health Services, 2017). Overall, the mean level observed was 59 µg/L, the median was 27 µg/L and the maximum was 819 µg/L; 29 out of 315 samples were below the DL of 0.1–25 µg/L. The mean and median levels, respectively, were 95 µg/L and 80 µg/L for raw water; for groundwater, these levels were 50 µg/L and 25 µg/L. The mean concentration observed was 57 µg/L in treated water (n = 30) and 26 µg/L in surface water (n = 5).
As part of the National Survey of Disinfection By-Products and Selected Emerging Contaminants, Health Canada collected drinking water at three points throughout the distribution systems from 65 sites across Canada during the 2009 and 2010 summer and winter months (DL = 5 µg/L) (Health Canada, 2016). In raw surface water samples (n = 89), barium had a mean concentration of 27 µg/L, a median of 19 µg/L and a maximum of 120 µg/L. In treated surface water (n = 70), barium had a mean concentration of 24 µg/L, a median of 16 µg/L and a maximum of 120 µg/L. In raw well water samples (n = 35), the mean concentration of barium was 103 µg/L, the median was 52 µg/L and the maximum was 500 µg/L. In treated well water (n = 27), barium had a mean concentration of 105 µg/L, a median of 80 µg/L and a maximum of 510 µg/L.
Between 2000 and 2016, Environment Canada collected freshwater quality data from over 200 federal and federal–provincial sampling sites at various locations and sampling frequencies throughout Canada's aquatic ecosystems (ECCC, 2017). For total barium, all 18,985 samples were above the DL of 0.02–1 µg/L; the mean level observed was 40.1 µg/L, the median was 25.3 µg/L and the maximum was 1,900 µg/L.
The Canadian Total Diet Study (CTDS) is a Health Canada initiative that measures the concentrations of different chemicals in foods and uses these data to estimate dietary exposures for different age–sex groups of the Canadian population (Health Canada, 2012b). In this study barium concentrations were measured in the tap water of nine cities between 2000 and 2012. In St. John’s the mean level observed was <3.6–57 µg/L, in Halifax it was 5.9–73 µg/L, in Quebec City 21–61 µg/L, in Montreal 15–247 µg/L, in Toronto <1–14 µg/L, in Ottawa 13–42 µg/L, in Winnipeg 1.2–14 µg/L, in Calgary 31–46 µg/L, and in Vancouver <11–62 µg/L.
From the Canadian Provincial/Territorial monitoring data reported above, as well as data from the National Survey of Disinfection By-Products and Selected Emerging Contaminants, an average of approximately 88 µg/L can be derived using the mean levels for treated drinking water; using an adult body weight of 70 kg and a drinking water consumption rate of 1.5 L/day (Health Canada, 1994), an estimated intake of 2 µg/kg bw per day (rounded) for barium from drinking water can be calculated for adults in the general Canadian population.
5.2 Food
Diet is estimated to be the primary route of barium exposure for the general Canadian population. The average dietary exposures to barium in Canada have been assessed through the CTDS (Health Canada, 2012a). Based on the CTDS data collected between 1993 and 2010 from nine Canadian cities (St. John’s, Halifax, Montreal, Ottawa, Toronto, Winnipeg, Calgary,Vancouver, and Whitehorse), the average dietary exposures to barium expressed on a body weight basis were estimated to be 12.4–27.7 µg/kg bw per day for infants aged 0–6 months, 16.6–26.6 µg/kg bw per day for children 7 months to 4 years old, 14.5–19.9 µg/kg bw per day for 5- to 11-year-olds, 9–12.4 µg/kg bw per day for 12- to 19-year-olds, and 5.1–10.1 µg/kg bw per day for persons aged 20 and above.
Based on the 1993–2012 CTDS data, average barium concentrations in common food items varied between the different years and cities in Canada. Sources of dietary exposure include food commodities such as grain-based products, fruits, vegetables, nuts, and herbs and spices (Health Canada, 2012b). Average barium concentrations from the CTDS were 19–5,403 μg/kg in grain-based products, 15–3,169 μg/kg in fruits, 3–4,023 μg/kg in vegetables, 1,755–5,033 μg/kg in nuts, and 20,750–38,172 μg/kg in herbs and spices. Average barium concentrations in infant milk and soy formulas ranged from16 µg/L to 216 µg/L. The Canadian Council of Ministers of the Environment (CCME) estimated the average barium concentrations in human breast milk to be 3.61 µg/L, based on an average of barium concentrations from six international studies (Canadian, Austrian, Italian, and Emirati) (CCME, 2013). Environment and Climate Change Canada (ECCC) (2016) reported average and 95th percentile intakes of 0.21 μg/kg bw per day and 0.24 μg/kg bw per day, respectively, for infants, based on concentrations in breast milk from 2001 Canadian mothers measured between 2008 and 2011 as part of the core Maternal–Infant Research on Environmental Chemicals (MIREC) study.
5.3 Air
In air, barium is associated with dust and suspended particulates. Barium occurs mainly as the sulphate or carbonate form and possibly as other insoluble forms (WHO, 1990; ATSDR, 2007). Data from the National Air Pollution Surveillance (NAPS) Program suggest that much of the barium in ambient air is bound to the coarse fraction of particulate matter (PM) (2.5–10 μm) (CCME, 2013). NAPS collects data on more than 300 ambient air pollutants across Canada (Environment Canada, 2013). For fine particulates (PM2.5), the mean concentration was 7 ng/m3, the median was 4 ng/m3, and the maximum was 58 ng/m3 (n = 157). For coarse particulates (PM10), the mean was 9 ng/m3, the median 6 ng/m3, and the maximum was 50 ng/m3 (n = 1,735). A mean of 2.7 ng/m3 was reported for the background concentration in indoor air in Canada based on two small monitoring studies (two Canadian homes in Alberta and one American retirement facility; standard deviation (SD) = 4.2, n = 40 samples) (CCME, 2013). In a Science Approach Document (SciAD) by ECCC (2016), median barium levels in 1,025 household dust samples were reported as 277 μg/g (95th percentile, 528 μg/g); median barium levels for outdoor and indoor air associated with PM2.5 were reported as 0.94 ng/m3 (range, 0.04–18.89 ng/m3; n = 910) and 1.06 ng/m3 (95th percentile, 4.71 ng/m3), respectively (Environment Canada, 2011; Rasmussen et al., 2016).
5.4 Consumer products
Barite is licensed in different oral natural health products in Canada and as a radiological contrast media for opacification of the gastrointestinal tract; however, the compound is insoluble and absorption is considered minimal (Health Canada, 2017). Skin products, cosmetics and tattoo inks can also contain barium (CCME, 2013; New Zealand Ministry of Health, 2013). However, daily intakes from these products were not estimated, as there are no available data on the proportion of the general population using these items, and absorption through the skin is considered negligible, based on the physicochemical properties of the element.
5.5 Soil
Soil concentrations of barium vary widely, depending on geological formations and anthropogenic inputs ranging from 15 to 3,000 mg/kg (up to 37,000 mg/kg near barite deposits). The Geological Survey of Canada reported a mean concentration in Canadian soil of 140 mg/kg (SD = 120; n = 7,397) (CCME, 2013). Moreover, a mean concentration of barium in indoor dust was estimated at 305.3 mg/kg (SD = 311.3; n = 165) based on the Ottawa residential study of Rasmussen et al. (2001).
5.6 Biomonitoring data
The most common biological matrix used as an indicator of barium exposure in biomonitoring studies is urine. In the U.S., the National Health and Nutrition Examination Survey (NHANES) III (1988–1994) as well as seven consecutive cycles of the Centers for Disease Control (CDC)-NHANES (up to 2012) provide data on urinary barium concentrations (normalized to creatinine concentrations) based on a representative sample of the U.S. population of all ages (n = 2,502 during the 2011–2012 cycle) (Paschal et al., 1998; CDC, 2015). Generally, urinary barium concentrations have remained relatively stable over the years, with children (aged 6–11) having significantly higher urinary barium levels (median, 2.18 µg/g creatinine (95% confidence interval (CI) = 1.70–2.61) than adolescents (aged 12–19; median, 1.42 µg/g creatinine (95% CI = 1.24–1.78) and adults (aged ≥20; median, 1.31 µg/g creatinine (95% CI = 1.20–1.43). In the 2011–2012 cycle, females had a significantly higher median urinary barium concentration than males, at 1.51 versus 1.17 mg/g creatinine, respectively; however, this difference was not found to be statistically significant at the 95th percentile level. Similarly, a German study by Heitland and Koster (2006) reported mean urinary barium levels of 1.2 µg/g creatinine for 72 children (aged 2–17) and 1.3 µg/g creatinine for 87 adults (aged 18–65) and a French study by Goullé et al. (2005) reported a median of 0.89 mg/L (corrected for creatinine enzymatic determination) in the urine of 100 healthy adults.
5.7 Multi-route exposure through drinking water
Barium can be absorbed via the inhalation route; however, exposure to barium vapours while showering or bathing is not expected to occur, since barium is not volatile (vapour pressure is close to null). Dermal absorption of barium during showering and bathing is considered negligible, since the skin permeability constant is 1 × 10–3 cm/h (U.S. EPA, 2004), indicating that the dermal route of exposure would contribute less than 10% of the drinking water consumption level (Krishnan and Carrier, 2008). Hence, the inhalation and dermal routes during showering and bathing are unlikely to contribute significantly to the total exposure.
6.0 Analytical methods
The U.S. Environmental Protection Agency (EPA) currently has three approved analytical methods (Method 200.5 revision 4.2, Method 200.7 revision 4.4, and Method 200.8 revision 5.4) for the analysis of total barium in drinking water. The U.S. EPA also approved several methods (SM 3111D, SM 3113B, and SM 3120B) developed by a voluntary consensus standard organization (U.S. EPA, 2016).
Method detection limits (MDLs) are dependent on the sample matrix, instrumentation, and selected operating conditions and vary between individual laboratories.
| Method (Reference) | Methodology | MDL (µg/L) |
Interferences/Comments |
|---|---|---|---|
| EPA 200.5 Rev. 4.2 (U.S. EPA, 2003) |
Axially viewed induct- ively coupled atomic emission spectrometry (AVICP-AES) |
0.05 | Subject to spectral, physical, chemical and memory interferences. Matrix interferences: Ca, Mg and Na >125 mg/L and Si >250 mg/L |
| EPA 200.7 Rev. 4.4 and SM 3120B (U.S. EPA, 1994Footnote a; APHA et al., 1992, 1995, 1998, 2005, 2012) |
Inductively coupled plasma-atomic emission spectrometry (ICP-AES) | 1.0 | Subject to spectral, physical, chemical and memory interferences. Matrix interferences: TDS>0.2% (w/v) for EPA 200.7 and TDS>1500 mg/L for SM 3120B |
| EPA 200.8 Rev. 5.4 (U.S. EPA, 1994b) |
Inductively coupled plasma-mass spectrometry (ICP-MS) | 0.04Footnote a –0.8Footnote b |
Subject to isobaric elemental and polyatomic ion interferences, and physical. Matrix interferences: TDS>0.2% (w/v) |
| SM 3111D (APHA et al., 1992, 1995, 2005, 2012) | Flame atomic absorption spectrometry | 30.0 | Subject to several interferences; primarily chemical interference (lack of adsorption by atoms). To prevent barium ionization Na+, K+ and Li+ cations are added in excess. |
| SM 3113B (APHA et al., 1992, 1995, 2005, 2012) | Electrothermal atomic absorption spectrometry |
2.0 | Subject to molecular absorption, chemical and matrix interferences. |
Footnotes
|
|||
The current U.S. EPA practical quantitation limit (PQL) for barium is 0.15 mg/L; it is based on the capability of laboratories to measure the concentrations of barium within reasonable limits of precision and accuracy. In the second six-year review of existing National Primary Drinking Water Regulations, the performance evaluation and proficiency testing data suggested that the current PQL could be lower; however, this reduction might be small, due to the lower passing rate of the laboratories at the lowest spike concentration. No new or revised methods have been approved that may be expected to improve analytical performance in the vicinity of the current PQL for the detection of barium in water (U.S. EPA, 2009).
Sample preservation and preparation
Generally, operational considerations for analysis of barium in drinking water (i.e., sample preservation, storage) can be found in the references listed in Table 2 above. Accurate quantification of dissolved, particulate (suspended) and total barium in samples is dependent on proper sample preservation and preparation steps. The SM 3030B method provides guidance on filtration and preservation procedures for determining dissolved or particulate metals (APHA et al., 2012). To determine dissolved barium concentrations, samples should be filtered (0.45 µm pore diameter) at the time of collection using preconditioned plastic filtering devices under either vacuum or pressure. The filtrate should be acidified to pH <2 with concentrated nitric acid. To determine particulate barium, the filter should be retained and the particulate material on it digested in the laboratory using appropriate methods (APHA et al., 2012).
Currently, EPA methods 200.7 and 200.8 and SM 3111D, SM 3113B, SM 3120B do not require hot acid digestion for total recoverable metals unless the turbidity of the sample is greater than 1 nephelometric turbidity unit (NTU). Digestion for EPA methods is performed by transferring an aliquot of the sample into a solution of nitric acid and hydrochloric acid followed by gentle heating to a temperature of approximately 85°C (i.e., hot digestion) (U.S. EPA, 1994a, 1994b). Microwave-assisted digestion (SM 3030 K) is recommended for analyzing total recoverable metals using SM methods that are based on ICP-MS. Although these methods do not require hot acid digestion unless turbidity is greater than 1 NTU, research conducted on other metals, such as lead and chromium, has indicated that this does not accurately quantify the total metal concentration in a sample. As such, the current protocol may underestimate total barium in drinking water when particulate barium is present. For example, analytical requirements under UCMR 3 include solubilizing the acid-preserved sample by gentle heating using nitric acid, regardless of the sample turbidity or the method used (U.S. EPA, 2012a).
7.0 Treatment technology and distribution system considerations
7.1 Barium in water
In aqueous environments, barium typically exists in divalent form as Ba2+. Under certain conditions, barium may form mineral precipitates involving sulphate, carbonate or chromate (Friedman et al., 2010). In the presence of a sulphate (SO42–) concentration of ≥2 mg/L and a pH level below 9.3, insoluble barite (BaSO4) may precipitate and become the dominant barium species. Most water sources contain sufficient SO42– concentration to precipitate BaSO4, thus maintaining barium concentrations at low levels. In the presence of carbonate ions (CO32–)and at a pH greater than 9.3, witherite (BaCO3) exhibits fast precipitation kinetics and becomes the dominant species, limiting barium solubility (Rai et al., 1984; Snyder et al., 1986; McComish and Ong, 1988; ATSDR, 2007; Menzie et al., 2008). Barium ions form weak complexes with Cl–, OH– and NO3– and the solubilities of these complexes are less dependent on pH than that of barite or witherite (Menzie et al., 2008; Tang and Johannesson, 2010; Kravchenko et al., 2014).
Barium is not readily oxidized or reduced (Menzie et al., 2008; Groschen et al., 2009). Kravchenko et al. (2014) indicated that changes in the oxidation state do not directly affect barium solubility in natural water. However, the redox potential may impact the species of sulphur. For example, if the SO42– oxidation state is reduced to a lower oxidation state (e.g., H2S), the depleted SO42– concentration will result in an increase of barium solubility.
Due to the cationic nature of soluble barium species, barium may be associated with various solid matrices, such as metal oxides, clay and organic matter. A study found that barium may displace other sorbed alkaline earth metals from manganese dioxide (MnO2), silicon dioxide (SiO2) and titanium dioxide (TiO2). However, these alkaline metals may displace barium from aluminium oxide (Al2O3) (Rai et al., 1984). Similarly to most cationic trace metals, the sorption properties of barium are enhanced at higher pH (Friedman et al., 2010).
7.2 Municipal scale treatment
Limited data exist on the removal of naturally occurring barium in drinking water. U.S. EPA (1998) lists lime softening, ion exchange (IX), reverse osmosis (RO) and electrodialysis/electrodialysis reversal (ED/EDR) as the most effective treatment processes for removing barium from drinking water.
Conventional coagulation/filtration techniques showed low barium removal from drinking water (up to 30%). Two-stage coagulation processes may achieve greater effectiveness (approximately 80% removal) than conventional coagulation; however, they are not common practice in Canada (Sorg and Logsdon, 1980; Lovins et al., 1999).
Early pilot- and full-scale lime softening studies reported up to 95% barium removal in drinking water at a pH range of 10.0–10.5. Dissolved barium is precipitated as barium carbonate, a mechanism that occurs for calcium removal during treatment of hard water (Sorg and Logsdon, 1980).
One of the most effective technologies for reducing barium concentration in drinking water is based on IX softening. Limited full-scale data and a number of comprehensive bench- scale studies indicated that strong-acid cation (SAC) and weak-acid cation (WAC) exchange resins remove 95–97% of barium to achieve a treated water concentration of below 1.0 mg/L (Kojola et al., 1978; Snoeyink et al., 1984, 1987a, 1987b; Myers et al., 1985; Snyder et al., 1986).
High-pressure membrane separation processes such as RO and nanofiltration (NF) are proven technologies for hardness removal from drinking water and are therefore expected to be very efficient for barium removal. ED/EDR is less commonly reported; however, it is also effective for barium removal from drinking water. Electrodialysis is typically applied for desalination of seawater and brackish water with a low turbidity (Sorg and Logsdon, 1980; Krause and Stover, 1982).
The selection and effectiveness of each treatment strategy are driven by several factors, including source water chemistry, the concentration of barium, pre-existing treatment processes, operational conditions of a specific treatment method, the utility’s treatment goals, and residual handling concerns and costs. Careful selection of the appropriate technology for a specific application is important, as the performance of each treatment technology is impacted by the specific chemical quality of the water being treated.
Where applicable, the appropriate authorities should be consulted to ensure that the disposal of liquid and solid waste residuals from the treatment of drinking water meet applicable regulations.
7.2.1 Control options
Typical control options for reducing excess barium levels in drinking water include switching to a new source, blending, and interconnecting with and/or purchasing water from another water system (Willey, 1987; U.S. EPA, 2012b). Attention must be given to the water quality of a new source prior to making any changes to an existing supply. Characterization of the water quality must be carried out to ensure that changes in water quality resulting from control options are assessed and that potential impacts on the existing treatment processes and distribution system are determined. For example, if the new water source is more aggressive, it may cause leaching of lead or copper in the distribution system. Any change in water quality should not result in other compliance issues. The disinfectant type (chlorine or chloramine) should be the same when blending two different water sources, to avoid water quality and disinfection issues.
Switching to another source may involve drilling a new well in an aquifer containing low barium levels, sealing off water-producing zones containing high barium levels, or finding an uncontaminated surface water source. Switching to another source may also be limited by the availability of new sources, existing water rights, and/or costs for transporting the new source water to the treatment plant.
Blending involves diluting the barium concentrations of a contaminated source with another source containing low or no barium. To minimize the piping required to carry the sources to a common mixing point, it would be ideal for the sources to be close to each other. Blending usually occurs in a storage tank or a common header, with resulting barium concentrations below the proposed MAC. Corrosion issues should be considered when blending different water qualities.
When interconnecting with another water system, the recipient system must consider a number of factors, including whether there is a nearby water supply that meets the proposed MAC for barium, whether this other system is willing to interconnect or consolidate and whether the interconnecting system can handle the increased demand resulting from additional customers. Costs are an additional consideration in the decision-making process for interconnection.
7.2.2 Conventional coagulation
Conventional coagulation with alum and iron salts provides low levels of barium removal (Krause and Stover, 1982). In a jar-test study, conventional coagulation has been shown to achieve an approximately 30% removal of total barium, reducing concentrations to approximately 5.0 mg/L from 7.0 to 8.5 mg/L in groundwater with either alum or ferric sulphate doses as high as 120.0 mg/L and a pH range of from 7.5 to 8.0 (Sorg and Logsdon, 1980). The test indicated that the formation of BaSO4 was a very slow process and conventional coagulation was not an effective mechanism for barium removal. Due to the high solubility of barium hydroxide [Ba(OH)2] and barium chloride (BaCl2) the formation of these complexes was also reported to be an ineffective factor in the removal of barium by a conventional coagulation treatment (Sorg and Logsdon, 1980).
A two-stage coagulation process may achieve a more efficient removal of barium; however, it is not recommended, since the second step is costly and requires additional space to store treated water between the two coagulation steps. In a bench-scale two-stage coagulation study, up to 80% of barium was removed, reducing approximately 8.0 mg/L barium in the feed water to less than 1.6 mg/L. The process used 100.0 mg/L of either alum or ferric sulphate and allowed for 1 h of sedimentation, followed by a second stage using 20.0 mg/L doses of coagulant and another 1 h of sedimentation (Sorg and Logsdon, 1980).
7.2.3 Chemical precipitation
A chemical precipitation process is commonly used in water treatment utilities to remove hardness and heavy metals such as iron and manganese, as well as barium, radium, cadmium, arsenic, lead, strontium and uranium. Chemical precipitation can also be used as a pretreatment or intermediate treatment in membrane systems to reduce the potential for scaling/fouling of membranes by mineral salts.
Lime softening is the most common application of chemical precipitation. During lime softening, dissolved barium is precipitated as BaCO3, the same mechanism that occurs in calcium removal from hard water. Jar-test, pilot-scale and limited full-scale data indicated that barium was effectively removed from water and that the efficiency was pH dependent. Jar tests revealed that a barium concentration of 7.0–8.0 mg/L in groundwater was effectively reduced by lime softening. The removal was pH dependent and increased from 65% at pH 8.7 to a peak of 98% at pH 10.5 and then declined to 55% at pHs greater than 11.6. Barium and hardness removal curves follow the same trend except in a pH range of 10.6–11.4, when the hardness removal curve developed a sharp increase due to the magnesium removal. At a pH greater than 11.0, barium can become more soluble as barium hydroxide and therefore becomes more challenging to remove. Pilot- and full-scale treatment data have confirmed these trends with respect to the dependence of barium removal on pH. In pilot-scale tests, raw water barium concentrations of 12.6 mg/L, 10.1 mg/L and 10.4 mg/L were reduced to 2.0 mg/L, 0.7 mg/L and 1.6 mg/L in the filtered water at pH levels of 9.2, 10.5 and 11.6, respectively. The hardness reductions achieved were, respectively, from 214 mg/L to 109 mg/L, from 216 mg/L to 86 mg/L and from 209 mg/L to 42 mg/L. Grab samples from two full-scale lime softening plants operating at pH levels of 10.5 and 10.3 showed 88.0% and 95.3% barium removal, respectively. The source waters had average influent barium concentrations of 7.5 mg/L and 17.4 mg/L and average hardness of 272 mg/L and 246 mg/L as CaCO3, respectively (Sorg and Logsdon, 1980).
Although it is not a conventional softening method, Parks and Edwards (2006) found that precipitative softening using sodium carbonate (Na2CO3) only was highly effective for barium removal. The primary objective of their study, which sampled 370 raw waters (⅔ groundwater, ⅓ surface water), was to survey the range of inorganic contaminant removals achieved by precipitative softening using Na2CO3 They reported that raising the pH of water samples to 10.3 using a fixed dose of Na2CO3 achieved an approximately 100% removal of barium from 145 raw water samples containing barium levels of 10–884 mg/L. Their findings suggest that further research should be done on softening water using Na2CO3 when requiring the removal of barium alone, rather than total hardness.
Since BaSO4 is relatively insoluble in water under alkaline conditions, it may precipitate when a sulphate-containing compound, such as gypsum [calcium sulphate (CaSO4)], is used as a precipitating chemical and sodium hydroxide is used for a pH adjustment (Krause and Stover, 1982). Krause and Stover (1982) reported on the effectiveness of a pilot-scale system using chemical precipitation followed by direct filtration. The system demonstrated that with an addition of 100 mg/L of CaSO4 and 352 mg/L NaOH, a barium concentration of approximately 6.0 mg/L was reduced to 0.5 mg/L in the treated water at a pH of 11.0 (91.0% removal). The removal occurred at a filter hydraulic loading rate of 1.5 gpm/ft2 (2.4 mm/s) with a filter head-loss of 27 in. (69 cm) after an 8-h run (end of the run). The precipitation process removed BaSO4 along with calcium hardness in drinking water. Other chemicals (such as alum, calcium hydroxide and ferric sulphate) used for precipitation of barium were less effective. Since the sodium concentration in the finished water increases, due to pH adjustment with sodium hydroxide, the authors indicated that modification of the chemicals used for pH adjustment would be required. Although the authors tested the precipitation of BaSO4 at a pH of 11, such a high pH is likely not necessary, based on several kinetic studies of BaSO4 (Aoun et al., 1996; van Leeuwen et al., 1996; Kugler et al., 2015). In addition, the high chemical doses used would affect the cost and sludge production. Bench, pilot- and full-scale studies are needed on the precipitation of BaSO4 from drinking water supplies.
7.2.4 Ion exchange
The most common application of IX in drinking water treatment is water softening. Extensive research has been conducted on the applicability of SAC and WAC exchange resins for the removal of scale-forming calcium (Ca2+) and magnesium (Mg2+) cations and, to lesser extent, of other alkaline earth metals (including Ba2+ and radium (Ra2+)) in drinking water (Sorg and Logsdon, 1980; Krause and Stover, 1982; Snoeyink et al., 1984, 1987a, 1987b; Myers et al., 1985; Snyder et al., 1986; Clifford, 1999; Elder and Budd, 2011). These types of IX resins come in different forms. SAC exchange resins in sodium (Na+), hydrogen (H+) and Ca2+ forms as well as WAC in H+ form can be used for removing barium in drinking water.
Clifford et al. (2011) compared separation factors of a number of cations on SAC resins and produced an ion selectivity sequence for ten divalent cations. The sequence describes the selectivity order in which cations are preferred by SAC resins and suggests that during water softening Ba2+ cations are preferentially removed compared with Ca2+ and Mg2+ forms. The WAC resins exhibit the same selectivity sequence as SAC resins except that the H+ ion is the most preferred cation (Clifford et al., 2011).
7.2.4.1 SAC resins in sodium and hydrogen forms
The SAC resins in Na+ or H+ forms exchange the Na+ or H+ cations for Ca2+, Mg2+ and other cations such as Ba2+ in the water, either as carbonate hardness or noncarbonate hardness, in a pH range of 2–11. The pH and alkalinity of the water treated with SAC (Na+) remained approximately unchanged throughout the production run. However, the SAC (H+) resin is rarely used in water softening because it produces acidic and corrosive water (Clifford, 1999) requiring acid-resistant materials, CO2 stripping and pH adjustment of the product water (Snoeyink et al., 1984, 1987a, 1987b; Myers et al., 1985; Snyder et al., 1986).
Barium showed similar breakthrough curves for both SAC (Na+) and SAC (H+) resins; hardness showed similar trends as well (Snyder et al., 1986; Snoeyink et al., 1987a). One of the major operational considerations when using IX treatment, especially with SAC (H+) resins, includes chromatographic peaking, whereby the less preferred ions (i.e., Na+ and Mg2+) are displaced by more preferred ions (Ba2+ and Ca2+), causing the effluent of Na+ and Mg+ concentrations to be greater than the influent concentration (Snoeyink et al., 1987a; Clifford, 1999). For conventional SAC (Na+) softening, chromatographic peaking is not a major operational consideration if the treatment run is terminated at hardness breakthrough.
With resins such as SAC (Na+) that have a strong affinity for barium, it may be difficult to remove barium from the exhausted resin. Barium accumulates on resin with repetitive exhaustion–regeneration cycles, and higher doses of regenerant are required to regain the resin’s capacity. The regeneration curve of the SAC (H+) resin regenerated using hydrochloric acid (HCl) follows the same trend as the regeneration curve of SAC (Na+) conducted with sodium chloride (NaCl) regenerant. Although barium can be precipitated in the spent NaCl brine before disposal, the authors stated that this brine cannot be reused due to the depletion of Na+ ions (Snoeyink et al., 1984, 1987a, 1987b; Myers et al., 1985). However, recent research has demonstrated that a spent brine can be reused (after barium removal) by adding NaCl to maintain the Na+ concentration of the brine (Clifford et al., 2011).
An early study reported that two full-scale IX softening plants were capable of achieving a barium concentration of 1.0 mg/L in treated water (Sorg and Logsdon, 1980). Plant 1 had two IX beds operating in a parallel mode, which treated a groundwater barium concentration of 10.0 mg/L (hardness 218 mg/L as CaCO3). Plant 2 had three parallel resin beds, which treated groundwater with a barium concentration of 19.0 mg/L (hardness 230 mg/L as CaCO3). Samples were collected from raw and treated water through one treatment cycle for one IX bed from each treatment plant. The plant 1 treatment run was terminated before either hardness or barium achieved breakthrough. The plant 2 run was terminated beyond both hardness and barium breakthrough. The tested IX beds reduced the barium concentrations to 1.0 mg/L for 153 bed volumes (BVs) (7.14 × 105 L) and 100 BVs (7.87 × 105 L) for plants 1 and 2, respectively. Plant 1 achieved 94–99% barium removal and 95–99% hardness removal. However, plant 2 reduced hardness and barium concentration to 142 mg/L and 5.8 mg/L, respectively, when the process was terminated. Both treatment plants practised blending of a portion of the raw water with the treated water to increase the hardness and to stabilize the distributed water. Since the blended waters had barium concentrations of 1.5–4.5 mg/L for both treatment plants, the authors concluded that blending was not a feasible practice at these two locations because the high influent barium concentrations at both locations would increase barium levels above the treatment goal of 1.0 mg/L even if only a small portion of the raw water was blended with the treated water (Sorg and Logsdon, 1980).
In comprehensive laboratory studies, a SAC (Na+) resin with an exchange capacity of 1.8 mEq/mL (4.8 mEq/g resin) was tested in repetitive exhaustion–regeneration cycles for barium, radium and hardness reduction in drinking water (Snoeyink et al., 1984; 1987a). The virgin resin was capable of reducing an influent barium concentration of 20.0 mg/L to 1.0 mg/L for approximately 1,200 BVs. The results indicated that the magnesium concentration in the treated water reached a peak concentration of approximately two times the influent concentration at approximately 600 BVs and subsequently reached the influent concentration after approximately 1,200 BVs. The calcium effluent concentration was also greater than the influent concentration at the end of the same process conducted with the virgin resin. Each exhaustion run had been terminated at a hardness breakthrough of 40.0 mg as CaCO3/L, and a regenerant dose of 1.8 mEq NaCl/mL resin [6.5 lb NaCl/ft3 resin (3 kg NaCl/m3 resin)], was used in the regeneration cycle. During several repetitive exhaustion–regeneration cycles, the regenerated resin was capable of treating approximately 225 BVs of water before both barium and hardness achieved breakthrough (together) at 1.0 mg/L and 40.0 mg as CaCO3/L, respectively. A regenerant dose of 2.7 mEq NaCl/mL resin [9.75 lb NaCl/ft3 resin (4 kg NaCl/m3 resin)] was capable of increasing the number of the BVs processed per exhaustion cycle to 260–270 BVs. However, an increase of the regenerant dose from 1.8 mEq NaCl/mL resin to 2.7 mEq NaCl/mL decreased the regeneration efficiency from 60% to 46%. The early breakthrough of barium indicated that it was accumulating on the resin. The virgin resin concentration was increased from 0.15 mEq Ba2+/mL to a constant value of 0.38 mEq Ba2+/mL after several regeneration cycles with a regenerant dose of 1.8 mEq NaCl/mL resin. At steady-state (i.e., when the barium uptake by the resin was equal to the barium removed by regeneration), the regenerant dose of 1.8 mEq NaCl/mL removed only 20% of the accumulated barium from the resin. Despite the barium accumulation, the number of BVs to hardness breakthrough remained constant. The study found that the barium concentration was effectively reduced to below 1.0 mg/L as long as the SAC (Na+) resin was not exhausted for hardness ions (Snoeyink et al., 1984; 1987a). These results have been confirmed by full-scale SAC (Na+) columns used to remove hardness and barium in groundwater (Snoeyink et al., 1987a). One of the SAC (Na+) columns was run beyond the hardness breakthrough and achieved water hardness and barium concentrations of 153 mg as CaCO3/L and 6.4 mg/L, respectively. However, another column, treating source water from another well at the same site and operating the exhaustion run to hardness breakthrough, was capable of reducing hardness and barium concentrations to 7.0 mg as CaCO3/L and 0.3 mg/L, respectively.
Snoeyink et al. (1987a) concluded that for raw water quality similar to the water used in the laboratory study cited above, two parallel SAC (Na+) columns operating in a staggered regeneration mode with a regenerant dose of 1.8 mEq NaCl/mL resin could effectively reduce the barium concentration below 1.0 mg/L in a blended treated water, if the exhaustion cycles were terminated at the hardness breakthrough. An important advantage of operating several columns in parallel with staggered regeneration is that product water quality is less variable compared with single-column operation. This can be a major consideration when the contaminant leakage and/or chromatographic peaking are high during a portion of the exhaustion run (Clifford et al., 2011).
It should be noted that using SAC (Na+) resins may result in undesirable quantities of sodium in the treated water. Therefore, SAC (Ca2+ and H+) and WAC (H+) resins are alternatives that can be used for the production of sodium-free treated water. However, proper corrosion control in the finished water should be practised.
7.2.4.2 SAC resin in calcium form
Several studies reported that the SAC (Ca2+) resin was capable of effectively reducing barium and radium concentrations in drinking water (Myers et al., 1985; Snoeyink et al., 1987b; Clifford, 1999; Atassi et al., 2007). However, until approximately 100–200 BVs of SAC (Ca2+) exhaustion, calcium ions are exchanged for all the cations in the feed water, resulting in finished water that is very high in hardness; after that point the total hardness decreases to the influent level. An exhausted SAC (Ca2+) resin requires calcium chloride (CaCl2) brine to be regenerated, and the spent brine may be reused after precipitating and removing barium. Proper doses of CaSO4 are needed to precipitate barium as BaSO4 in the spent brine and to prevent fouling of the resin during the following regeneration cycle. If the brine also contains Ra2+, it will coprecipitate on the BaSO4 and will also be removed (Myers et al., 1985).
A pilot-scale SAC (Ca2+) system was tested in several repetitive exhaustion–regeneration cycles. The regenerated resin was capable of reducing an influent barium concentration of 12.0 mg/L to 1.0 mg/L for run lengths of approximately 1,300 BVs during the third loaded (exhaustion) run. The regeneration has been conducted with a regenerant dose of 6.0 eq CaCl2/L resin (compared with only 1.8 eq NaCl/L resin [Snoeyink et. al., 1987]) and the spent CaCl2 brine was reclaimed and reused. The exhausted SAC (Ca2+) resin required less frequency of regeneration than the calcium sulphate SAC (Na+) resin (Atassi et al., 2007).
In laboratory tests, a virgin SAC (Ca2+) resin was capable of reducing barium concentration of up to 23.0 mg/L to below 1.0 mg/L in the treated water for a run length of approximately 1,200 BVs, at a loading rate of 5.4 gpm/ft2 (13.2 m/h) and an empty bed contact time of 2.5 min (Myers et al., 1985). The hardness, alkalinity and pH of the water were nearly unchanged throughout the treatment runs. The exhausted SAC (Ca2+) resin was effectively regenerated (92–100% removal of barium from the column) with a regenerant dose of 6 eq 0.85 CaCl2/L. Both regenerant concentration and regenerant dose affected the barium concentration in the treated water in the following exhaustion runs. When the regenerant doses were increased from 4.0 to 6.0 and to 8.0 eq CaCl2/L resins, a barium concentration of 1.0 mg/L was achieved for run lengths of 500, 900 and 1,100 BVs, respectively. The study reported that a mole ratio of 1.1:1 (CaSO4:barium) reduced barium concentration in the spent brine and the brine was successfully reused (Myers et al., 1985). Snoeyink et al. (1987b) indicated that the use of the SAC (Ca2+) resin in parallel with SAC (Na+) may produce treated water with the desired level of hardness in addition to barium removal. However, to prevent an increased sodium concentration in the treated water, SAC (Ca2+) can be use in parallel with SAC (H+) and WAC (H+), followed by carbon dioxide stripping.
7.2.4.3 WAC resin in hydrogen form
The WAC (H+) resins have weak-acid functional groups and only exchange ions in the neutral to alkaline pH range. The IX process results in partial softening and produces treated water with a low alkalinity and low total dissolved solids (TDS) levels. WAC (H+) resins also require acid-resistant material, CO2 stripping and pH adjustment of the product water (Snoeyink et al., 1984, 1987a, 1987b; Myers et al., 1985; Snyder et al., 1986). Although WAC (H+) resins can be regenerated by weak acids that are only slightly stronger than the resin functional group, strong acids such as HCl are usually applied. However, sulphuric acid (H2SO4) should not be used, as BaSO4 is precipitated on the resin and destroys its capacity. Since barium is easily removed from the exhausted WAC (H+) resin with approximately 5–10% excess regenerant, it does not accumulate on the resin. The regeneration of WAC (H+) resin produced less spent regenerant per unit volume of treated water than the regeneration of the SAC column (Snyder et al., 1986; Clifford, 1999; Elder and Budd, 2011). WAC (H+) systems can be complex to operate and maintain, and they may have increased costs related to the chemicals and materials used (Snoeyink et al., 1984, 1987a, 1987b; Clifford, 1999; Clifford et al., 2011).
In laboratory tests, WAC (H+) resin was found to effectively remove barium (22.0 mg/L), radium and hardness without increasing the sodium concentration in the treated water . A virgin WAC (H+) resin (capacity of 11.5 mEq/g resin) showed an approximately identical selectivity for barium and calcium ions, as both contaminants break through at approximately 800 BVs, while magnesium ions break through earlier, at 650 BVs. The virgin WAC (H+) resin showed a lower capacity for barium but greater capacities for hardness and alkalinity than the virgin SAC (Hm+) resin under similar operating conditions. Through several repetitive exhaustion–regeneration cycles, the WAC (H+) resin was capable of producing treated water with a barium concentration below 1.0 mg/L (22.0 mg/L influent) for run lengths of 600–650 BVs. A regenerant dose of 8.5 mEq HCl/g resin (i.e., only 75% of the stoichiometric amount) was capable of achieving 93–95% regeneration efficiency and greater than 99% removal of divalent ions from the resin. However, regenerating the resin with a dose of 7.6 mEq HCl/g resin (i.e., 66% of the stoichiometric amount) produced higher barium concentrations ranging from 0.5 to 1.3 mg/L in the treated water as a result of incomplete removal of barium from the resin during the regeneration cycles (Snyder et al., 1986).
A common practice in water softening is bypass blending, which involves diverting a portion of the influent flow around the treatment vessel and blending the diverted water with the treated water. Blending of finished water with raw water may stabilize finished water and decrease the cost of treatment by reducing the volume of water treated, which results in less frequent regeneration and therefore a savings in chemical and brine disposal costs (Clifford, 1999). However, the barium concentration in the bypass water needs to be considered to ensure that the finished water concentration is not above the MAC.
7.2.5 Membrane technology
Effective membrane technologies for barium removal in drinking water include RO, NF, as well as ED/EDR (U.S. EPA, 1998; Odell, 2010). The primary difference between RO and NF is the size of dissolved contaminants that can be removed. RO membranes effectively reduce TDS and monovalent ions while NF membranes are mainly used for the removal of hardness (Ca2+, Mg2+) and organics (e.g., precursors of disinfection byproducts). ED/EDR is most typically used for TDS reduction and inorganic ion removal (U.S. EPA, 2012b).
RO treatment systems typically require prefiltration for particle removal and often include other pretreatment steps, such as the addition of anti-scaling agents, dechlorination and/or softening. Pretreatment is required to preserve membrane life because the presence of chlorine residuals, particulates, and scale-forming ions (i.e., Ca2+, Ba2+, iron, and silica) in the feed water can adversely affect the performance of RO processes.
Post-treatment for RO permeate (i.e., finished water) typically includes pH adjustment, addition of corrosion inhibitors and disinfection. RO concentrate disposal must also be considered in the design and operation of RO plants. Systems that integrate two or more membrane processes or combine a membrane process with other treatment processes (i.e., integrated membrane system [IMS]) are implemented to improve overall process water recovery and reduce waste stream concentrations (Ning et al., 2006; Gabelich et al., 2007).
The performance of an IMS was tested for barium removal from May 1998 to January 1999 at a 56.6 MGD water treatment plant located in East St. Louis, Illinois (Lovins et al., 1999). The IMS was supplied with surface water pretreated by conventional coagulation/sedimentation/ filtration processes (CSF). The raw water had a low barium concentration (average of 0.11 mg/L) and the CSF pretreatment achieved an average barium concentration of 0.07 mg/L. The IMS unit consisted of three spiral-wound, crossflow NF membranes with a maximum pressure of 400 psi (27 bars); a composite thin-film membrane with a cut-off rating of 100–200 Da and 85 m2 of filtration area (CSF-NF1); a composite thin-film membrane with a cut-off rating of 150–300 Da and 97 m2 of filtration area (CSF-NF2); and a cellulose acetate membrane with a cut-off rating of 300 Da and 123 m2 of filtration area (CSF-NF3). Inorganic permeate water quality varied by membrane type and produced water with average barium concentrations of 0.002, 0.026 and 0.021 mg/L, respectively, corresponding to average barium removals of 97.1%, 62.9% and 70.0%, respectively. Since CSF-NF1 also decreased hardness and alkalinity to below DLs (not provided) it required corrosion control measures to stabilize treated water. By contrast, the CSF- NF2 and the CSF-NF3 membranes allowed more alkalinity and hardness to pass into the permeate, which produced more stable and less corrosive product water. The CSF-NF3 membrane was more resistant to fouling than both the CSF-NF1 and CSF-NF2 membranes, while CSF-NF1 and CSF-NF2 were more sensitive to the operating conditions (e.g., flux and recovery) and feed water chemistry (e.g., pH) (Lovins et al., 1999).
7.2.5.1 Improving recovery of reverse osmosis
Inorganic scale formation (e.g. silica, barium sulphate and calcium carbonate) remains a serious impediment to achieving high RO recovery. Scaling resulting from the precipitation of salts within the membrane module leads to permeate flux decline and shortening of the membrane life. As water passes through the RO membrane, rejected Ba2+ ions accumulate near the membrane surface, and its concentration may increase to the point where precipitation of barium salts occurs (Boerlage et al., 2000; Gabelich et al., 2007). Barium sulphate can be problematic for any drinking water treatment methods that concentrate barium (Boerlage et al., 2000; Ning et al., 2006; Gabelich et al., 2007; WQA, 2014). For example, membrane scaling by barium sulphate can reduce membrane recovery and also cause flux decline and potentially severe membrane damage (Boerlage et al., 2000; Ning et al., 2006).
Lime softening followed by filtration and pH adjustment is an effective pretreatment to improve the performance of RO for enhanced removal of mineral salt scaling from water sources. Intermediate concentrate chemical stabilization (ICCS) strategies apply conventional or pellet softening to a primary (first pass) RO concentrate to remove scale-forming compounds such as barium, followed by secondary RO treatment to improve the overall system recovery (He et al., 2011). An implementation of conventional lime-softening-based ICCS technology (a primary RO concentrate followed by a secondary RO system) was capable of improving the overall system recovery from 85% to 92.5–95.5% (He et al., 2011). Using a lime dose of 1,220 mg/L, the ICCS reduced an average barium concentration of 0.34 mg/L in the RO concentrate to an average of 0.05 mg/L (86% reduction) at pH 10.5. At this pH level silica, strontium and calcium concentrations were also reduced to 76%, 84% and 77%, respectively. The authors found that a pelletized-based ICCS technique removed less barium, silica, strontium and calcium from the primary RO concentrate when compared with conventional lime-softening-based ICCS (He et al., 2011).
Ning et al. (2006) reported the results of a bench-scale and pilot-scale study at a large inland desalination plant in El Paso, Texas. Precipitative softening by magnesium hydroxide [Mg(OH)2] powder and conventional lime softening using calcium hydroxide [Ca(OH)2] powder were assessed for the removal of silica and barium from RO concentrate that was subsequently treated by secondary RO to demonstrate improved RO recovery. Treatment with Mg(OH)2 powder reduced a barium concentration from 0.9 mg/L to 0.34 mg/L in the concentrate (64% reduction) within 1 h of processing time at a pH range of 7.8–9.1 but did not provide sufficient silica reduction (only 16%). Conventional lime softening conducted with Ca(OH)2 reduced a barium concentration from 0.9 mg/L to 0.43 mg/L (50%) and silica from 143 mg/L to 48 mg/L (66%) at pH 10.3–10.6 after 1 h. Excess lime softening at pH 12.0 improved the settleability of the sludge due to the coagulative effect of Mg(OH)2 precipitates. However, an influent barium concentration of 0.9 mg/L was reduced by only 30% and 67% after 1 and 3 h, respectively. The silica concentration of 155 mg/L was reduced by 96% and 98% after 1 and 3 h, respectively. It was concluded that concentrate from primary (i.e., first-pass) RO membranes would be most effectively treated by conventional lime softening at a pH of 11.5 for 1 to 3 h. In the pilot-scale study, the authors found that the performance of the primary RO had an 85–90% recovery level, which was limited by the presence of silica and precipitated barium sulphate. Treatment of the primary RO concentrate by lime softening followed by a secondary RO sufficiently reduced membrane fouling through reduction of silica and barium concentrations and also resulted in an increased overall recovery of the system to approximately 97%.
Similarly, both Rahardianto et al. (2007) and Gabelich et al. (2007) demonstrated that two-stage desalination RO, supplemented with precipitative softening treatment of primary RO concentrate, was an effective approach to remove mineral salt scaling including barium sulphate, as well as improving process recovery to 95–98%. Gabelich et al. (2007) suggested that barium removal during precipitative softening was enhanced by inclusion into or onto calcium carbonate crystalline lattice during precipitation.
7.2.6 Emerging technologies
7.2.6.1 Adsorptive media
Araissi et al. (2016) tested an adsorptive zeolite 4A medium for barium removal in batch experiments using feed water with barium concentrations of from 0.3 mmol (41.1 mg/L) to 25 mmol (3,432 mg/L) at pH levels of 8.2–11.0. The samples were stirred for 14 h in contact with 0.15 g of the media. The maximum observed adsorption capacity was 2.25 mmol Ba2+/g. The authors indicated that the mechanism of barium removal was considered an IX process. The batch experiments were also carried out using a binary system of barium and strontium, and the authors found that strontium was preferentially removed by zeolite over barium. They also indicated that zeolite may provide an effective IX material for removing barium in water. Sato et al. (2011) reported on the use of zeolite 4A media filters for the removal of radioactive contaminants including barium from drinking waters associated with the the severe incident at the Fukushima Daiichi Nuclear Power Station. The authors found that synthetic zeolite 4A efficiently removed cesium, strontium and barium but gave no details regarding the capacity of the filters or the operating conditions.
7.2.7 Distribution system considerations
The accumulation of trace inorganic contaminants (TICs) in the drinking water distribution system is a complex function of numerous factors, including contaminant concentration in treated water, pH and redox conditions in the distribution system and pipe material. Iron oxyhydroxides and hydrous manganese oxides are significant sinks for TIC accumulation because of their adsorptive affinity for them. Water quality changes or physical disruptions in the distribution system can remobilize contaminants into the bulk water. Indicators of potential release of TICs in the distribution system include the presence of discoloured water or increased turbidity.
Barium has been repeatedly detected in samples from both distribution system piping scales and solids mobilized during hydrant flushing (Schock et al., 2008; Freidman et al., 2010; Peng et al., 2012; Lytle et al., 2014). Barium deposition in the distribution system occurs primarily due to the surface adsorption and/or co-precipitation reactions involving soluble barium species (Ba2+), which are enhanced at elevated pH (Friedman et al., 2010). Barium accumulation is strongly influenced by the co-occurrence of manganese deposits, phosphate precipitates and/or phosphate surface groups and to lesser extent by the concentration of barium in the water (McComish and Ong, 1988; Sugiyama et al., 1992; Charette and Sholkovitz, 2006; Schock et al., 2008; Friedman et al., 2010; Peng et al., 2012). Barium may also enter the distribution system water through leaching from cement-based materials and linings under aggressive water chemistry and/or hydraulic conditions. Leaching of barium from cement-mortar linings can result in the precipitation of BaCO3 and BaSO4 in the distribution system (Friedman et al., 2010). Guo et al. (1998) conducted laboratory tests to determine the extent of leaching from ductile iron pipes lined in situ with Portland cement (type I) mortar. The pipes were lined, cured and subsequently disinfected in accordance with ANSI/AWWA standards. The tests were performed using tap water from a New Jersey water distribution system. Under static conditions barium concentration was increased gradually by up to 18% of its respective U.S. EPA drinking water standard of 2.0 mg/L during the first 14 days of water stagnation. The cement used to line the test pipes contained a lower amount of metals than most commercially available cements.
Barium impurities may be present in various chemicals (such as alum, calcium hydroxide, calcium hypochlorite and polyaluminium chloride) that are used in drinking water treatment (NHMRC, 2011). NSF/ANSI standards 60 and 61 each set a maximum concentration for barium that can be present in materials and treatment chemicals in contact with drinking water. This maximum concentration is known as the single product allowable concentration (SPAC). Both current SPACs for barium under NSF/ANSI standards 60 and 61 are 0.2 mg/L (NSF/ANSI, 2015; 2016d).
Lead-pipe scale samples collected from 91 pipe specimens of lead and lead-lined service lines from 26 different water distribution systems in the U.S. had an average barium concentration of 199.0 mg/kg scale (range of 1.0 mg/kg to 2,850 mg/kg scale). Barium was detected in concentrations greater than the reporting level of 5.0 mg/kg in 87 of the 91 samples (95%) (Schock et al., 2008).
Barium was the most concentrated inorganic contaminant detected in scale samples and sediments collected from the distribution systems of 20 U.S. drinking water utilities supplied by groundwater, surface water and blended water sources (Friedman et al., 2010; Peng et al., 2012). The distributed water had barium concentrations ranging from 0.002 to 0.6 mg/L, pH levels of 7.1 to 8.5 and alkalinity of 48 to 289 mg/L as CaCO3. Friedman et al. (2010) reported that the median barium concentration of all scale deposits and sediment samples was 94 µg/g. Specifically, median barium concentrations in scale deposits and hydrant-flush solids were 88 µg/g and 104 µg/g, respectively. Five of the deposit samples with barium concentrations ranging from 460 µg/g to 2,400 µg/g also had high manganese (372–46,700 µg/g) and phosphorous concentrations (2,000–12,300 µg/g). Zasoski and Burau (1988) indicated that manganese dioxide has been extremely effective for adsorption of cationic species similar to barium. Similarly, Peng et al. (2012) observed a notable correlation between barium and manganese concentration in scale deposits. This correlation supported the data from previous studies (Murray, 1975; Sugiyama et al., 1992) showing that manganese oxides formed in corrosion products interact with barium more strongly than iron oxides.
Barium concentrations in 22 hydrant flush solids and two pipe specimen samples from 12 different water utility distribution systems ranged from 60.8 µg/g to 9,276 µg/g (Lytle et al., 2014). All waters had a pH range of 7.0–8.0, alkalinity of 251–476 mg/L as CaCO3 and were sourced from groundwater.
Friedman et al. (2010) reported an estimated barium mass of 76.0 lb accumulated on a 100-mile pipe length (based on a 12-in. diameter pipe). The authors noted that theoretically 16– 26% of the scale deposit would need to be released to exceed the U.S. EPA drinking water standard for barium of 2.0 mg/L.
7.3 Residential scale
In cases where barium removal is desired at the household level, for example when a household obtains its drinking water from a private well, a residential drinking water treatment device may be an option for decreasing barium concentrations.
Health Canada does not recommend specific brands of drinking water treatment devices, but it strongly recommends that consumers use devices that have been certified by an accredited certification body as meeting the appropriate NSF International (NSF)/American National Standards Institute (ANSI) drinking water treatment unit standards. These standards have been designed to safeguard drinking water by helping to ensure the material safety and performance of products that come into contact with drinking water. Certification organizations provide assurance that a product conforms to applicable standards and must be accredited by the Standards Council of Canada (SCC). In Canada, the following organizations have been accredited by the SCC to certify drinking water devices and materials as meeting NSF/ANSI standards (SCC, 2018):
- CSA Group (www.csagroup.org);
- NSF International (www.nsf.org);
- Water Quality Association (www.wqa.org);
- UL LLC (www.ul.com);
- Bureau de normalisation du Québec (www.bnq.qc.ca);
- International Association of Plumbing & Mechanical Officials (www.iapmo.org); and
- Truesdail Laboratories Inc. (www.truesdail.com).
An up-to-date list of accredited certification organizations can be obtained from the SCC
(2018).
Water treatment technologies able to be certified to NSF standards for barium reduction include cation exchange, RO and distillation. Applicable NSF/ANSI standards are No. 44: Cation Exchange Water Softeners (NSF/ANSI, 2016a); No. 58: Reverse Osmosis Drinking Water Treatment Systems (NSF/ANSI, 2016b) and No. 62: Drinking Water Distillation Systems (NSF/ANSI, 2016c).
For drinking water treatment devices to be certified to NSF/ANSI standards 44, 58 and 62 for the reduction of barium concentration, the devices must be capable of reducing an average influent (challenge) concentration of 10.0 mg/L to a maximum final concentration of 2.0 mg/L or less. In addition, treatment devices certified to standards 44 and 62 can be certified either specifically for barium reduction (as noted above) or for the removal of hardness and TDS, respectively, which are used as a surrogate for barium in these standards. If water softeners (cation exchange systems using 100% sulphonated polystyrene divinyl benzene resin), certified to NSF/ANSI Standard 44 and reduced hardness concentration to below 1.0 gpg (17.1 mg/L) from an influent hardness of 20 gpg (342 mg/L) will be able effectively to reduce barium in drinking water. Treatment devices certified to NSF/ANSI Standard 62 using TDS as a surrogate must achieve a minimum TDS reduction of 99% from an influent concentration of 1,000 mg/L.
RO systems are intended for point-of-use (POU) installation, as larger quantities of influent water are needed to obtain the required volume of treated water, which is generally not practical for residential-scale point-of-entry systems. RO systems should only be installed at POU, as the water they have treated may be corrosive to internal plumbing components. A consumer may need to pretreat the influent water to reduce fouling and extend the service life of the membrane. Distillation systems are also intended for POU installation only. The distillation process is effective for the removal of inorganic contaminants but requires an electrical energy input.
Selection of the most effective treatment system for a household will depend on a variety of factors, including the concentration of barium, and such other parameters as hardness, alkalinity and the pH of the source water. Before a treatment device is installed, the water should be tested to determine the general water chemistry and verify the presence and concentration of barium. Periodic testing by an accredited laboratory should be conducted on both the water entering the treatment device and the finished water to verify that the treatment device is effective. Treatment devices lose their removal capacity through usage and time and need to be maintained and/or replaced. Consumers should verify the expected longevity of the components in their treatment device according to the manufacturer’s recommendations and service it when required.
Various household water softeners have been randomly sampled to examine their barium removal efficiency. The influent barium concentrations ranged from 0.5 mg/L to 6.4 mg/L and influent hardness varied from 205 mg/L to 248 mg/L as CaCO3. All tested water softeners reduced barium concentration to below 1.0 mg/L and hardness to between 2 mg/L to 35 mg/L as CaCO3 (Snoeyink et al., 1987a).
Homeowners with private wells using IX softeners in sodium form should be aware that the treatment unit may introduce undesirable quantities of sodium in the treated water.
8.0 Kinetics and metabolism
8.1 Absorption
Barium absorption from the diet in humans was reported to vary widely, generally ranging from 1% to 60% (LeRoy et al., 1966; Schroeder et al., 1972; Legget, 1992; Kravchenko et al., 2014). Factors influencing absorption include age, chemical species/solubility, fasting status, vitamin D status, and the presence of other ions in the diet (Legget, 1992). Despite the variability and uncertainty of the results, Legget (1992) proposed an absorption of 20% for water-soluble forms of barium by the gastrointestinal tract, which at the time of the review aligned with the value applied by the International Commission on Radiological Protection (ICRP, 1980). Peak blood level was reported to occur 2 h after ingestion of barium chloride, with a serum half-life of 3 h (Downs et al., 1995).
A wide range of oral absorption values have also been reported in laboratory animals (1–95%), with more water-soluble forms having higher absorption (Venugopal and Luckey, 1978; Kravchenko et al., 2014). In rats, 85% of BaCl2 was absorbed at 14–18 days of age, 63% at 22 days of age, and 6% at age 6 weeks and older (Taylor et al., 1962). Having adult rats fast for 18 h pre-exposure increased absorption by a factor of three. Rats receiving 10 mg /L of barium as barium sulphate by intubation exhibited peak barium blood levels 15 min after dosing, with 50% remaining in the blood 4 h post exposure (McCauley and Washington, 1983). The maximum blood concentration was higher with barium sulphate than with barium chloride or barium carbonate. In dogs, oral absorption of barium chloride was reported to range between 1–7% and 50% (Cuddihy and Griffith, 1972).
Legget (1992) reported several unpublished studies with oral absorption rates ranging from 1% to 50%. In rats administered barium chloride via gavage, younger rats (14–22 days old) absorbed barium more efficiently than older animals (6–70 weeks old) (i.e., 63–84% in young animals versus 7–8% in older rats) (Taylor et al., 1962). In this same study, fasting was also found to increase absorption from 7–8% to 20%. Absorption after the first 7 h following administration was not investigated. The ICRP (1993) and the World Health Organization (WHO, 1990) also reported that children may absorb barium to a greater extent than adults; the ICRP (1993) has estimated oral absorption of soluble barium to be 60% in infants, 30% in children 1–15 years old and 20% in adults.
The solubility of barium compounds under environmental conditions does not necessarily influence their absorption in the body. McCauley and Washington (1983) compared absorption efficiencies of different barium compounds and found that single gavage doses of 131Ba-labelled barium sulphate and barium chloride (amounting to 10 mg of barium) were absorbed at similar rates, as indicated by blood and tissue levels in rats. The authors suggested that the similar absorption efficiencies may be attributed to the solubilization of the low dose of barium sulphate by hydrochloric acid in the stomach. Barium carbonate in a vehicle containing sodium bicarbonate was poorly absorbed, most likely owing to the buffering capacity of sodium bicarbonate, which may have impaired the hydrochloric-acid-mediated conversion to barium chloride. These results suggest that soluble barium compounds or compounds generating barium ions in the acidic environment of the stomach have similar absorption efficiencies.
8.2 Distribution
Barium mainly deposits in bone and connective tissues, with bones containing approximately 66–90% of the total body burden (Schroeder et al., 1972; Venugopal and Luckey 1978; Tardiff et al., 1980). Barium can also accumulate in teeth, as shown in a study by Miller et al. (1985) in which the ratio of barium to calcium observed in the teeth of children from a community with high levels of barium in drinking water (10 mg/L) was five times higher than among children from another community with barium levels of 0.2 mg/L, despite their similar ethnic and socioeconomic status. Barium also distributes widely into different tissues, with lungs, fat, muscle, skin, connective and soft tissues being found to contain 0.1–0.5 mg of barium in adult humans in the United States (Schroeder et al., 1972).
No differences in distribution were found between sexes of rats exposed to 10–250 mg/L of barium as barium chloride for 4–13 weeks (Tardiff et al., 1980). Barium is also known to cross the placental barrier (Venugopal and Luckey, 1978; McCauley and Washington, 1983). The highest concentrations of barium in rats intubated with 10 mg/L of barium chloride for 24 h were found in the heart, muscles, eye, liver, and kidney (McCauley and Washington, 1983).
8.3 Metabolism
Barium is not metabolized in the human body. As an ion it is monovalent and not subject to changes in oxidation state.
8.4 Excretion
In humans barium is excreted in feces and urine, with feces representing the primary route of excretion; Schroeder et al. (1972) reported that for a total intake of 1.33 mg/day (1.24, 0.086, and 0.001 mg/day from food, water, and air, respectively) approximately 90% of the barium is excreted in the feces and 2% in the urine. Similar results were observed by Tipton et al. (1969); two men excreted 95%–98% and 2%–5% of a daily barium intake via feces and urine, respectively.
8.5 Physiologically based pharmacokinetic (PBPK) models
No PBPK modelling for barium was identified in the currently available literature.
9.0 Health effects
Barium is not considered to be an essential element (Venugopal and Luckey, 1978; Ferrante et al., 2014; Chellan and Sadler, 2015).
Different barium compounds have differing solubilities in water and body fluids, which influences their toxicity. The Ba2+ ion and the soluble compounds of barium (mainly chloride, nitrate, and hydroxide) are toxic to humans and animals. Barium carbonate, although relatively insoluble in water, is soluble in the gastrointestinal tract, allowing uptake into serum and tissues thereby capable of causing effects. Insoluble barium compounds, such as barium sulphate, serve as inefficient sources of Ba2+ ion and are therefore generally nontoxic to humans. The nontoxic nature of barium sulphate has made it useful in medical applications; for example, it serves as a contrast medium for X-ray examination of the gastrointestinal tract. However, barium sulphate or other insoluble barium compounds can become toxic if the gastrointestinal tract is compromised (e.g., in the case of colon cancer), thereby allowing barium to enter the bloodstream (ATSDR, 2007).
9.1 Effects in humans
The database on human health effects from barium exposure is limited. Health effects reported from acute exposure to high levels of barium carbonate or chloride are largely related to hypokalemia, which can lead to complications such as ventricular tachycardia, hypertension and/or hypotension, muscle weakness and paralysis. Portal of entry gastrointestinal effects such as vomiting, abdominal cramps, and watery diarrhea have also been reported shortly after ingestion of high doses. Associations between barium in drinking water and mortality from cardiovascular disease are largely negative; however, one retrospective study reported a positive association, although a lack of controlling for confounding factors and poor exposure characterization render the results unreliable. The impacts of barium exposure on developmental and reproductive toxicity, as well as hearing loss, have been investigated; however, results are inconclusive, largely due to poor exposure characterization. The carcinogenicity of barium has not been evaluated in humans.
9.1.1 Acute toxicity
Acute poisoning with barium salts can occur (levels unreported) in cases of accidental human oral ingestion and suicide attempts. In these situations, barium has caused hypokalemia (low serum potassium levels), leading to neuromuscular (e.g., muscle weakness and paralysis, abnormal reflexes, tingling, dizziness) and cardiovascular effects (e.g., arrhythmias, electrocardiogram abnormalities) (McNally, 1925; Diengott et al., 1964; Lewi et al., 1964; Ogen et al., 1967; Talwar and Sharma, 1979; Deng et al., 1991; Downs et al., 1995; Koch et al., 2003; Rhyee and Heard, 2009; Payen et al., 2011; Bhoelan et al., 2014). In one case study, the absorbed barium came from a carbonate salt present in flour that was solubilized as a result of the acidic stomach pH.
Gastrointestinal effects (gastric pain, vomiting, diarrhea, tightness in the throat, dryness of the mouth, hemorrhage) are reported as acute effects following barium ingestion. The acute responses can progress to increased blood pressure, progressive muscular paralysis, cardiovascular and respiratory failure, and even death (Downs et al., 1995; Jourdan et al., 2001; Bhoelan et al., 2014). Hemoglobin in the urine and renal failure have also been observed (Morton, 1945; Gould et al., 1973; Wetherill et al., 1981; Phelan et al., 1984; Jha et al., 1993; Silva et al., 2003; Koch et al., 2003; Lukasik-Glebocka et al., 2014). Doses of 1–15 g of barium sulphide have caused death. However, in another case medical treatment following exposure to up to 133 g of barium carbonate prevented a lethal response (Downs et al., 1995).
9.1.2 Subchronic and chronic toxicity and carcinogenicity
9.1.2.1 Cardiovascular toxicity
A retrospective study found higher mortality rates (p < 0.05) from all cardiovascular diseases and for heart diseases (data retrieved from death certificates for the years 1971–1975) in Illinois communities with barium concentrations of 2–10 mg/L in drinking water compared with communities having <0.2 mg/L in their drinking water (25,433 adults of four high-barium communities and 46,905 adults of seven low-barium communities) (Brenniman and Levy, 1979; Brenniman et al., 1985). The communities were matched for demographic characteristics and socioeconomic status; however, there was no adjustment for the use of water softeners, medication, smoking, diet and exercise. Moreover, the rate of population change was about 70% in the communities with the highest concentrations during the decade preceding the study dates, and no information was provided on the length of time individuals lived in a community. Additionally, exposure was poorly characterized; no information was provided on tap water consumption rates. In another study that evaluated cardiovascular morbidity, no differences in blood pressure, stroke, heart or kidney disease were observed (p < 0.05) between an Illinois community with a mean of 7.3 mg/L in drinking water (n = 1,175 adults in West Dundee) and another one with a mean of 0.1 mg/L (n = 1,203 adults in McHenry) for the years 1976–1977 (Brenniman and Levy, 1981; as summarized in Brenniman et al., 1985).
No association between barium intake from drinking water and cardiovascular markers (i.e., cholesterol, glucose, and triglyceride levels, blood pressure, heart rate, electrocardiographic endpoints) were found in 11 American male volunteers (27–61 years old) (Wones et al., 1990). Barium was administered via the drinking water at successive barium concentrations of 0 ppm for the first 2 weeks, 5 ppm for the next 4 weeks and 10 ppm (as barium chloride) for the final 4 weeks. Factors associated with cardiovascular risk (such as exercise and food intake) were controlled. However, the small number of subjects, the duration of exposure, and the lack of absorption data limit an interpretation of the results of this study.
9.1.3 Developmental and reproductive toxicity
There is little information from human studies on reproductive and developmental effects from soluble barium compounds. In a small prospective epidemiological study, there were no differences (in gestational age, birth weight, and major or minor malformations; p = 1.0) in babies (n = 32) born to mothers who inadvertently swallowed barium sulphate used in radiography procedures during the first trimester of pregnancy when compared with 94 control mothers (Han et al., 2011). Moreover, there was no evidence of teratogenicity in babies (n = 5) born to mothers who were inadvertently exposed to barium via enema (Han et al., 2010). Nevertheless, the low solubility and absorption rates of barium sulphate used in medical procedures confound the interpretation of these results with respect to soluble barium compounds.
A small, single-site case–control study reported that barium was among the metals that were reported to be elevated in the hair (p = 0.003) and urine (p = 0.002) of 25 children (mean age of 5 years) with autism spectrum disorder in Saudi Arabia compared with 25 non-autistic children matched for age and sex. However, this study had several shortcomings: mothers of autistic children were exposed to second-hand smoke, controls were chosen through case referrals, and no attempt was made to identify which other trace elements and heavy metals could be responsible for the associations (Blaurock-Busch et al., 2011).
9.1.4 Other effects
In a pilot cross-sectional study, Ohgami et al. (2016) reported an association between hearing loss (auditory thresholds) and concentrations of barium in hair (odds ratio (OR) = 4.75 (95% confidence interval (CI): 1.44, 17.68) at 8 kHz and OR = 15.48 ( 95% CI: 4.04,79.45) at 12 kHz) and toenails (OR = 3.20 (95% CI: 1.35, 7.85)) at 8 kHz and OR = 3.63 (95% CI: 1.58, 8.55) at 12 kHz) in Bangladesh (n = 145 individuals of both sexes aged 12 to 55); however, with no associations with urine levels being observed and sources of exposure not reportedly characterized, the results of this study limit any inferences that can be made with respect to barium exposure and hearing loss.
Utilizing the data from NHANES 1999–2002 to explore the association between waist circumference and body mass index with the body burdens of various toxic metals, Padilla et al. (2010) reported that urinary barium showed a direct positive association with body mass index and waist circumference (p < 0.05), suggesting that environmental exposure to barium (and other metals) may influence variations in weight gain or loss in humans.
9.2 Effects on experimental animals
The database on health effects in animals following barium exposure is also somewhat limited. Investigations into cardiovascular function generally did not find any significant alterations in blood pressure or electrocardiogram readings following low-dose oral exposure. Some studies did find significant increases in blood pressure; however, the use of a low mineral diet with less than adequate levels of calcium may have influenced the study results.
The most sensitive adverse effect of barium appears to be renal toxicity. Nephropathy has been observed in rats and mice following short- and long-term oral exposure to barium, with steep dose–response curves being observed in both species; data in mice suggest that the severity and sensitivity to renal lesions is related to the duration of exposure.
Reproductive and developmental studies are limited, with results generally being negative for impacts on reproductive tissues or reproductive performance in rats and mice. Decreases in sperm number and quality as well as shortened estrous cycles and morphological alterations in the ovaries have been observed in rats; however, study weaknesses render these results inconclusive. Decreased pup birth weight and a nonsignificant decrease in litter size have been reported in the offspring of rats exposed to barium chloride in drinking water prior to mating. Finally, several studies found that oral exposure to barium did not significantly increase tumour incidence.
9.2.1 Acute toxicity
Fluid in the trachea, inflammation of the small and large intestine, decreased liver/brain weight ratio, increased kidney/brain weight ratio, darkened liver, ocular discharge and decreased body weight were reported in rats administered by gavage 60–960 mg Ba/kg as barium chloride in water (Borzelleca et al., 1988). In this study the LD50 (median lethal dose) values were reported as 419 and 408 mg BaCl2/kg bw in male and female Sprague-Dawley rats, respectively. Another study reported an LD50 of 132 mg Ba/kg bw and 220 mg Ba/kg bw for adults and weanling Charles River rats (strain not specified), respectively, gavaged with barium chloride in water (Tardiff et al., 1980). Also, ECG abnormalities, tachycardia, ventricular fibrillation, muscle paralysis, salivation, diarrhea, hypertension, respiratory paralysis, hypokalemia, and death were observed following intravenous infusion of barium chloride in mongrel dogs (Roza and Berman, 1971).
9.2.2 Short-term exposure
9.2.2.1 Kidney effects
The U.S. National Toxicology Program (NTP, 1994) conducted toxicity studies in F344/N rats and B6C3F1 mice by administering barium chloride dihydrate (99% pure) in drinking water for 15 days and 13 weeks. In the 15-day studies, rats (70 days old; 5/sex/dose) were administered barium at concentrations of 0, 125, 250, 500, l,000, or 2,000 ppm (10, 15, 35, 60, or 110 mg/kg bw per day for both males and females), and mice (77 days old; 5/sex/dose) received 0, 40, 80, 173, 346, or 692 ppm (10, 30, 65, 110, or 200 mg/kg bw per day for males; 10, 35, 65, 115, or 180 mg/kg bw per day for females). In rats, no chemical-related deaths, differences in final mean body weights or clinical findings of toxicity were observed; in addition, no significant differences in absolute or relative organ weights, hematology, clinical chemistry, or neurobehavioral parameters were reported. Water consumption by male and female rats in the highest dose group was slightly less (≤16%) than controls during the second week. In mice, no chemical-related deaths, differences in mean body weights and water consumption, or clinical/histopathological evidence of toxicity were observed; in the highest dose group, relative liver weight of males and absolute and relative liver weights of females were significantly higher than those of the controls.
In the 13-week studies, F344/N rats and B6C3F1 mice (43 days old; 10/sex/dose) were administered barium (as barium chloride in drinking water ad libitum) at concentrations of 0, 125, 500, 1,000, 2,000 and 4,000 mg/L (0, 10, 30, 65, 110 or 200 mg Ba/kg bw per day for male rats; and 0, 10, 35, 65, 115 or 180 mg Ba/kg bw per day for female rats; 0, 15, 55, 100, 205 or 450 mg Ba/kg bw per day for male mice; and 0, 15, 60, 110, 200 or 495 mg Ba/kg bw per day for female mice, as determined by the study authors) (NTP, 1994). In rats, water consumption by males and females in the high dose group was 30% lower than controls; chemical-related kidney lesions (three males and three females) and death (three males and one female) were also observed in this dose group. No changes in blood pressure were observed throughout the 13-week period in any of the dose groups. Absolute and relative kidney weights in females of the 115 or 180 mg/kg bw per day dose groups as well as in males of the 200 mg/kg bw per day dose group were significantly higher than controls and were related to chemical-induced renal lesions; a no-observed-adverse- effect level (NOAEL) of 65 mg/kg bw per day for increased kidney weights in female rats can be identified from this study. In mice, final mean body weights for males and females of the highest dose group were significantly lower than controls, with males consuming 18% less water than controls. More severe kidney lesions were observed in mice than in rats, with multifocal to diffuse nephropathy (i.e., tubule dilatation, regeneration and atrophy), crystals and eosinophilic casts in atrophic tubules and increased mortality being reported in both sexes in the highest dose group; a NOAEL of 200 mg/kg bw per day for kidney lesions in female mice can be identified from this study.
Fisher-344/N rats and B6C3F1 mice received barium chloride dihydrate (10 animals/sex/ species/dose) via drinking water at concentrations of 0, 1,000, 2,000 or 4,000 mg Ba/L (equivalent to doses of 0, 65, 110 and 200 mg Ba/kg bw per day for males; and 0, 65, 115 and 180 mg Ba/kg bw per day for females, as estimated by the study authors) for 13 weeks (Dietz et al., 1992). Mortality was 10–30% for rats and 60–70% for mice in the highest dose groups. Mortality in mice was attributed to treatment-related renal toxicity, whereas in rats, renal lesions were less severe and were not attributed mortality in the high dose group. Toxic nephrosis and crystals in the lumen of renal tubules (postulated by authors as insoluble barium salts) were observed in more than 80% of the mice of both sexes in the highest dose group. Renal lesions were characterized by dilatation (containing eosinophilic granular casts and crystals), regeneration, and atrophy (lined with epithelial cells with stained basophilic cytoplasm) of the tubular cells in mice. There was an irregular depression of the renal capsule near the collapsed tubules. Also, an increase in fibrous tissue was observed between the tubules in the cortex and outer medulla. In rats, only a few foci of dilated tubules were observed in the outer medulla in both sexes at the highest dose. A NOAEL of 2,000 mg Ba/L (110 mg Ba/kg bw per day) was identified by the authors of this study.
In a study investigating the protective effects of pomegranate peel against barium- mediated kidney damage, a single group of six adult Wistar rats exposed to barium chloride (67 ppm, equivalent to 10 mg/kg bw per day, as reported by the study authors) for 21 days reduced creatinine clearance (indicator of glomerular dysfunction) compared with controls (Elwej et al., 2016a). The barium group also had various hemorrhage foci and leucocyte infiltration that were evident in the Bowman’s space portion of the Bowman’s capsule that surrounds the glomeruli.
Female Sprague-Dawley rats gavaged for 10 days with 100, 145, 209 and 300 Ba/kg bw per day in water had a decreased kidney/brain weight ratio in all dose groups except the highest; however, the authors concluded that these results were likely not barium-related, due to the absence of effects in the highest dose group (Borzelleca et al., 1988).
No adverse effects were observed in young adult rats (Charles River rats; 30 animals/sex/ dose) exposed to 0, 10, 50 or 250 mg Ba/L as barium chloride in drinking water (equivalent to 2– 3, 6–13, or 28–64 mg Ba/kg bw per day for males; and 2–3, 7–15, or 36–68 mg Ba/kg bw per day for females, as reported by the study authors) for 13 weeks (Tardiff et al., 1980).
9.2.2.2 Cardiovascular effects
An increase in barbiturate-induced myocardial contractile depression was observed in young female Long-Evans hooded rats exposed for 16 months to 100 ppm barium chloride via drinking water (Kopp et al., 1985). An increase in systolic blood pressure was measured in barium-treated rats compared with controls (n = 12). Reduced myocardial contractile velocity and conduction system in the atrioventricular nodal region and disturbances in energy metabolism were also observed after 16 months of exposure.
9.2.2.3 Other effects
A decrease in motor activity (mice and rats), weaker grip strength (mice), and a decrease in thermal sensitivity (mice) were also observed at the 4,000 ppm dose in the NTP (1994) study, as described above; forelimb grip strength of female mice receiving 4,000 ppm was also lower following 90 days of exposure.
Female ICR mice (n = 5) exposed to barium chloride in drinking water for 2 weeks or 2 months (equivalent to doses of 0.14 and 1.4 mg Ba/kg bw per day, as indicated by the authors) had severe hearing loss in both dose groups after 2 weeks, and severe degeneration of the inner ear after 2 months (Ohgami et al., 2012) when compared with controls. Several study weaknesses limit the interpretation of these findings. These weaknesses include the use of a strain of mouse that has been shown to suffer from progressive hearing loss (thus this study requires replication in other species); the measurement of inner ear barium levels was done at a time (11 weeks) long after hearing analysis was performed (5 weeks); no differences in bone-barium levels were observed between the treatment groups; and levels of barium in other tissues (including kidney, liver and heart) were undetectable, which is inconsistent with what would be expected following barium exposure.
9.2.3 Long-term exposure and carcinogenicity
9.2.3.1 Kidney effects
As part of the NTP study described above (NTP, 1994), rats and mice (60/sex/dose) were also exposed to barium chloride in drinking water at concentrations of 0, 500, 1,250 and 2,500 mg/L (as barium) (0, 15, 30, 60 mg Ba/kg bw per day for male rats; 0, 15, 45, 75 mg Ba/kg bw per day for female rats; 0, 30, 75 or 160 mg Ba/kg bw per day for male mice; and 0, 40, 90 or 200 mg Ba/kg bw per day for female mice, as estimated by the study authors) for 103–105 weeks. In rats, no chemically related effects were observed at any dose. In mice, mild to severe nephropathy in males and females (i.e., tubule regeneration of cortical and medullary epithelium, dilatation, hyaline cast formation, multifocal interstitial fibrosis, and glumerulosclerosis in some animals; brown crystals within tubule lumens and interstitium and renal cysts in males) and increased mortality due to kidney lesions was reported in the high dose group. In the 15-month interim evaluation, urea nitrogen levels were elevated in mice of all dosage groups (although the finding was not statistically significant, according to the authors) indicating that some animals were beginning to experience changes in kidney function consistent with the observed pathology, even at the lower doses. A NOAEL of 75 mg/kg bw per day can be identified for nephropathy in male mice.
Long-Evans rats exposed to 5 mg Ba/L (52/sex; as barium acetate) via drinking water had increased proteinuria in males only after lifetime exposure, whereas females showed no significant difference from controls; no kidney lesions in treated rats were noted by the authors (Schroeder and Mitchener, 1975a). A reduction in survival was observed in male Swiss mice of the Charles River CD strain (but not in females) exposed to 5 mg Ba/L (as barium acetate) via drinking water; however, no kidney lesions in treated mice were noted by the authors (Shroeder and Mitchener, 1975b). Rats administered barium chloride via drinking water at concentrations of 1–1,000 mg Ba/L (the authors only indicated that 10 ppm was equivalent to 1.5 mg Ba/kg bw per day) for 16– 68 weeks exhibited structural changes to the glomeruli at the highest dose of 1,000 ppm (or approximately 150 mg/kg bw per day) (McCauley et al., 1985); thus a lowest-observed-adverse- effect level (LOAEL) of 150 mg/kg bw per day can be identified from this study.
9.2.3.2 Cardiovascular effects
Perry et al. (1985, 1989) exposed female Long-Evans weanling rats (13 per dose group) to barium chloride in drinking water at concentrations of 0, 1, 10 or 100 ppm for 1–16 months. An increase in mean systolic blood pressure was observed in the 10 ppm group at 8–16 months. The increase in the 100 ppm dose group was observed from the first month to the end of the study. Other studies have not observed these associations following exposures as high as 150 mg/kg bw per day (1,000 ppm) (McCauley et al., 1985) and 180 mg/kg bw per day (4,000 ppm) (NTP 1994). Animals in the Perry et al. (1985, 1989) studies were fed a rye-based diet with calcium levels below the recommended daily requirement (NRC, 1995), which may have rendered them more sensitive to the cardiovascular effects of barium; since some evidence in humans indicates that reduced dietary calcium is a risk factor for hypertension (McCarron et al., 1984), the relevance of the results from these studies is uncertain.
9.2.3.3 Other effects
Lymphoid depletion was observed in the spleen, thymus and lymph nodes of mice and rats exposed to 2,500 ppm barium chloride dihydrate for 103–105 weeks (NTP, 1994). A NOAEL of 180 mg/kg bw per day for rats and 205 mg/kg bw per day for mice can be identified from this study.
9.2.3.4 Carcinogenicity
No increase in tumour incidence was observed in Fisher-344/N rats or B6C3F1 mice exposed to concentrations of up to 2,500 mg/L of barium (60–75 mg/kg bw per day in rats; 160– 200 mg/kg bw per day in mice) via drinking water for 2 years (NTP, 1994). Statistically significant decreases were observed in the trends of mononuclear cell leukemia and neoplasms as well as non-neoplastic lesions of the adrenal gland in male rats and of the mammary gland in female rats. Also, statistically significant decreases in hepatocellular adenoma were observed in male mice. Moreover, no increase in tumours was found in Long-Evans rats or Swiss mice (52 animals/species/sex) exposed to 5 ppm barium acetate in drinking water for 540 days (Schroeder and Mitchener, 1975a, 1975b), or after male SD rats were exposed to 1–100 ppm barium chloride for 68 weeks (McCauley et al., 1985).
9.2.4 Genotoxicity
In vitro studies have generally not found evidence of barium genotoxicity; one in vivo study found an increase in one type of somatic mutation at a high dose.
9.2.4.1 In vitro findings
Barium chloride (10 to 10,000 µg/plate) caused no significant increase in gene mutation frequency in Salmonella typhimurium (TA97, TA98, TA100, TA1535, and TA1537) with and without Aroclor 1254-induced rat or hamster liver S9 fractions (Rossman et al., 1991; NTP, 1994). Moreover, no increase in gene mutation or DNA damage was found (via assay with H17 and M45 strains of Bacillus subtilis, DNA polymerase test from avian myoblastosis virus, and microscreen assay with Escherichia coli WP2) with barium nitrate and barium chloride (Nishioka, 1975; Sirover and Loeb, 1976a, 1976b; Kanematsu et al., 1980; Rossman et al., 1991). Also, barium chloride did not induce sister chromatid exchanges, chromosomal aberrations or cell cycle delay in Chinese hamster ovary cells (NTP, 1994). However, barium chloride (at 250 µg/ml and above) induced an increase in gene mutations in L5178Y mouse lymphoma cells in the presence of metabolic activation (no increases in mutant colonies were observed without S9 activation) (NTP, 1994).
9.2.4.2 In vivo findings
Yesilada (2001) reported increases in small single wing spots (the wing spot test is indicative of somatic mutations) in Drosophila melanogaster exposed to high concentrations of barium nitrate (10 mM), but not at lower concentrations (1 mM). The effect of barium on large single spots and twin spots was inconclusive at both 1 and 10 mM.
9.2.5 Reproductive and developmental toxicity
In the NTP (1994) study, as described above, significantly increased testicular (p ≤ 0.01) and uterine (p ≤ 0.05) weights were observed in rats of the 2,500 ppm dose group (60 mg Ba/kg bw per day for males and 75 mg Ba/kg bw per day for females) following 15 months of exposure via drinking water; no significant changes in the weights of reproductive organs were observed in mice. A NOAEL of 60 mg/kg bw per day for rats and 160 mg/kg bw per day for mice can be identified from this study. However, the significance of these findings is unknown, since reproductive and developmental toxicity was not assessed. Dietz et al. (1992) conducted a single-generation mating trial in which groups of male and female F-344/N rats and B6C3F1 mice (20/sex/species/group) were exposed to barium chloride dihydrate in the drinking water at 0, 1,000, 2,000, or 4,000 ppm for the rats and 0, 500, 1,000, or 2,000 ppm for the mice for 60 days (males) or 30 days (females). Estimated doses (from a subchronic study by the same authors described in Section 9.2.2) for rats were 0, 65, 110, and 200 mg/kg per day for males and 0, 65, 115, and 180 mg/kg per day for females; for mice the estimated doses were 0, 55, 100, and 205 mg/kg per day for males and 0, 60, 110, and 200 mg/kg per day for females. After the exposure period, males and females from the same exposure groups were housed together until there was evidence of mating or until the end of the mating period (8 days). Numerous reproductive parameters were assessed. For both rats and mice no changes in epididymal sperm counts, sperm motility, sperm morphology, testicular or epididymal weights or vaginal cytology were observed. In rats, pregnancy rates were below historically normal values for the laboratory; however, they were not treatment related. No significant alterations in gestation length, pup survival, or occurrence of external abnormalities were observed. A statistically significant (p < 0.01) decrease in live pup weight at birth was observed in the 4,000 ppm group; however, after 5 days of age no significant alterations in pup body weight were observed. In mice, no alterations in maternal weight gain, average length of gestation, pup survival or pup weights were observed. A statistically significant (p < 0.05) decrease in average litter size occurred on days 0 and 5 in the 1,000 ppm treatment group but not in the 2,000 ppm treatment group. No external abnormalities were observed in the offspring. A NOAEL of 115 mg/kg bw per day for rats and 200 mg/kg bw per day for mice can be identified from this study.
Decreased ovary weight and ovaries/brain weight ratio were observed in 29- to 37-day-old Sprague-Dawley rats (10 animals/sex/dose) exposed to 198 mg Ba/kg from barium chloride administered by gavage in water once daily for 10 days, but not at 138 mg Ba/kg (Borzelleca et al., 1988); thus a NOAEL of 138 mg Ba/kg can be identified from this study.
9.3 Mode of action
As reported in Section 9.2, chronic and subchronic drinking water studies in rats and mice indicate that the kidney is a sensitive target of barium toxicity; however, the mode of action for barium-mediated kidney toxicity has not been fully elucidated. Available data indicate that kidney toxicity may be associated with oxidative processes. Wistar rats receiving 67 mg/L barium chloride for 21 days had an increase in kidney and liver markers of oxidative stress (tissue lipoperoxide, lipid hydroperoxides, advanced oxidation protein product, malondialdehyde, and H2O2 levels), a decrease in catalase, superoxide dismutase and glutathione peroxidase enzyme activities, vitamin C, and glutathione and non-protein thiol. An increase in metallothionein content was also observed (Elwej et al., 2016a, 2016b). Thus, oxidative stress could be responsible for damaged cellular macromolecules, such as proteins, cell membranes and/or disruption of the mitochondrial respiration chain (Storz et al., 2005). The oxidative stress hypothesis is supported by high levels of H2O2, malondialdehyde and advanced oxidation protein product. More specifically, H2O2 can be converted to the hydroxyl radical, which can cause rapid lipid peroxidation and the depletion of glutathione levels, increasing the susceptibility of organs to oxidative stress. Moreover, the oral administration of pomegranate peel powder (5% of diet) improved all barium-induced renal and liver damage, possibly by acting as an electron donor (antioxidant).
The acute cardiovascular and neuromuscular toxicity of barium reported in case studies has been attributed to its hypokalemia effect (Roza and Berman, 1971; Koch et al., 2003). As a potassium channel antagonist, barium increases intracellular potassium and decreases its extracellular levels by blocking the efflux of cellular potassium and increasing the sodium- potassium-ATPase pump activity (Payen et al., 2011). It is possible that the increased intracellular potassium concentrations may result in a decreased resting membrane potential; however, there is also evidence of a barium-induced neuromuscular blockade and membrane depolarization (Phelan et al., 1984; Thomas et al., 1998). In addition, since barium has chemical properties similar to calcium, they can interact through biochemical pathways involving calcium binding proteins and compete for binding sites (IPCS, 1990). Hypertensive effects of barium in rats reported by Perry et al. (1989) may have been due to inadequate calcium levels in the diet.
10.0 Classification and assessment
The International Agency for Research on Cancer (IARC) has not classified barium as to its carcinogenicity. The U.S. EPA (2005) concluded that barium is considered not likely to be carcinogenic to humans via oral intake. Other agencies have concluded that there is no evidence that barium is carcinogenic via the oral route (IPCS, 2001; WHO, 2016). Several animal studies found no increase in tumours following long-term exposure to barium in drinking water at levels as high as 2,500 mg/L (60–75 mg/kg bw per day in rats; 160–200 mg/kg bw per day in mice) (Schroeder and Mitchener, 1975a, 1975b; McCauley et al., 1985; NTP, 1994).
Nephropathy in mice is considered to be the most sensitive health effect associated with chronic oral exposure to soluble salts of barium. In humans, renal failure has been observed following exposure to high levels of barium in poisoning events (see Section 9.1). The chronic mouse study by the NTP (1994) was chosen as the key study for deriving a health-based value (HBV) for barium in drinking water for the following reasons: it used an adequate number of animals (60/sex/dose); administration of barium was via drinking water; histopathological analysis indicated that the observed renal lesions were morphologically different from the spontaneous degenerative renal lesions commonly observed in aging mice; and mice appear to be the most sensitive of the animal species tested, exhibiting the lowest NOAEL for kidney effects (75 mg/kg bw per day). Additionally, a dose–response relationship was observed; a statistically significant number of mice in the high dose group exhibited mild to severe cases of nephropathy (along with a significant increase in mortality due to treatment-related renal lesions), and within the second highest dose group, one female and two males (out of 60) exhibited mild to moderate chemical-related nephropathy.
Hypertension has also been reported in animals and humans following exposure to barium; however, the evidence is conflicting. In humans, the dose–response data are insufficient to support an association between chronic barium exposure and hypertension. Results from epidemiological studies are limited either by their small sample sizes, short durations of exposure, poor exposure characterization, inadequate controlling of important risk factors for hypertension, or a combination thereof. In animals the results are equivocal; the positive findings are potentially influenced by an experimental diet low in calcium, which has been identified as a possible risk factor for hypertension in humans. As a result the data on hypertensive effects were considered inadequate for deriving an HBV.
Benchmark dose (BMD) modelling was performed using the nephropathy incidence in mice as reported in the 2-year NTP (1994) study at a 10% and 5% increased incidence over background rates (the benchmark response; BMR). Male and female mouse data were modelled separately as well as combined, with both average dose and individual gender doses giving similar modelling results. Of the models that provided a reasonable fit (via evaluation of goodness of fit p-value > 0.1; BMD/BMDL (benchmark dose lower confidence limit) ratio < 5; and visual inspection of the curve), the model providing the best fit (i.e., lowest Akaike information criterion) as well as the most conservative BMD/BMDL values was the multistage 3 model for male mice. A BMR of 5% was selected due to the following statistical and biological considerations: the study design provides sufficient statistical power (e.g., 50–100 animals/dose/sex); the BMR falls near the low end of the observable dose range; the BMD and BMDL values are similar; and the health endpoint is severe. As the lower 95% confidence limit on the benchmark dose for a 5% response (BMDL5) of 58 mg/kg bw per day for male mice is lower than the corresponding values for both females and males and females combined, this has been selected as the point of departure for the calculation of the HBV for barium in drinking water.
Limited information is available on the age-related differences in susceptibility to barium. Although adults were symptomatic, children did not appear to be affected in two food poisoning incidents involving barium carbonate (Lewi and Bar-Khayim, 1964; Deng et al., 1991); however, the lack of examination of children and the uncertainty regarding their barium intake limits the interpretation of these results. Age-related differences in the absorption of ingested barium are reported for both animals and humans. Studies by Taylor et al. (1962) and Cuddihy and Griffith (1972) indicate that oral absorption of barium in younger animals may be an order of magnitude greater than in older animals; additionally, the ICRP (1993) estimates the oral absorption of soluble barium to be 60% in infants, 30% in children 1–15 years old and 20% in adults. With increased uptake of barium and higher bone remodelling rates in infants and children than in adults, there is a greater potential for barium accumulation in bone; however, the significance of this not known and requires further investigation.
Using the BMDL5 identified above, a tolerable daily intake (TDI) can be calculated as follows:
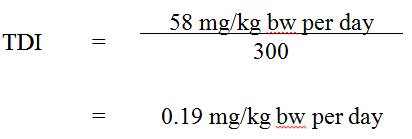
Text description - Equation 1
The TDI for Barium is 0.19 mg/kg body weight per day. This is calculated by dividing the BMDL5 of 58 mg/kg body weight per day by the uncertainty factor of 300.
where:
- 58 mg/kg bw per day is the lower 95% confidence limit on the benchmark dose (BMDL5) for increased incidence of nephropathy in male mice as described above; and
- 300 is the uncertainty factor (10 for interspecies and 10 for intraspecies variation, as well as 3 for database deficiencies, including the lack of a two-generation reproduction toxicity study).
Using this TDI, the HBV for barium in drinking water for non-cancer effects is derived as follows:
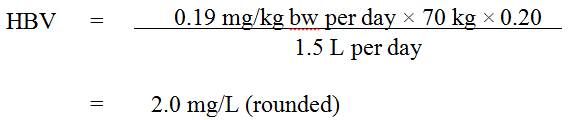
Text description - Equation 2
The HBV for barium in drinking water is 2.0 mg/L (rounded). This is calculated by multiplying the TDI for barium of 0.19 mg/kg body weight per day by the average body weight for an adult of 70 kg, then by 0.2, the allocation factor denoting the contribution of drinking water to the tolerable daily intake, and dividing the resulting product by 1.5 L per day, the drinking water intake rate for an adult.
where:
- 0.19 mg/kg bw per day is the TDI, as derived above;
- 70 kg is the average body weight for an adult (Health Canada, 1994);
- 0.2 is the allocation factor for drinking water, since food represents the main source of exposure and drinking water represents a significant but lesser source of exposure (see Section 5.0); and
- 1.5 L/day is the drinking water intake rate for an adult.
10.1 International considerations
The U.S. EPA (1995) established a maximum contaminant level of 2 mg/L based on the possibility of increased blood pressure in humans. In a more recent evaluation, the U.S. EPA IRIS derived an RfD of 200 µg/kg bw per day for nephrotoxicity based on the benchmark dose lower 95% confidence limit (BMDL5) of 63 mg/kg bw per day derived from a 2-year mouse study (U.S. EPA, 2005); using this recent evaluation, the U.S. EPA updated the drinking water equivalent level for barium to 7 mg/L (U.S. EPA, 2012c).
The WHO (2016) established a drinking water guideline value of 1.3 mg/L for barium based on the same uncertainty factor (300) and BMDL5 of 63 mg/kg bw per day as determined by the U.S. EPA (2005). An allocation factor of 20%, an adult body weight of 60 kg and a drinking- water consumption rate of 2 L per day were used in calculating the final guideline value.
The Australian government has established a drinking water guideline of 2.0 mg/L based on a NOAEL of 0.2 mg/kg bw/day from Brenniman and Levy (1985) for no observed adverse effects on blood pressure and kidney disease (NHMRC, 2011).
11.0 Rationale
Barium is present in many drinking water sources, both naturally and as a result of human activities. The barium levels in Canadian drinking water will vary greatly, depending on geological formations as well as anthropogenic activities, including oil and gas exploration and the use of fertilizers and pesticides. Barium exists as a mixture of over 20 naturally occurring radioisotopes with various levels of radioactivity. The focus of this document is limited to barium’s chemical properties. Based on these chemical properties, exposure to barium from drinking water would only be a concern from ingestion—it is not expected to be a concern through either inhalation or dermal absorption.
IARC has not classified barium as to its carcinogenicity. The U.S. EPA concluded that barium is not likely to be carcinogenic to humans via ingestion, and other international agencies have stated that there is no evidence showing that barium could cause cancer via ingestion.
The kidneys are considered to be the major target for barium toxicity. In humans, renal failure has been observed following exposure to high levels of barium in poisoning events; in animals, kidney effects are considered the most sensitive health effect associated with chronic oral exposure, especially in mice, the most sensitive species. For these reasons, an HBV of 2.0 mg/L was derived to be protective of the general population, based on kidney effects from a study in mice.
A MAC of 2.0 mg/L is proposed for barium in drinking water. The proposed MAC is protective of potential health effects, can be reliably measured by available analytical methods and is achievable by municipal and residential scale treatment technologies. As part of its ongoing guideline review process, Health Canada will continue to monitor new research in this area and recommend any change to the guideline that is deemed necessary.
12.0 References
Aoun, M., Plasari, E., David, R. and Villermaux, J. (1996). Are barium sulfate kinetics sufficiently well known for testing precipitation reactor models? Chem. Eng. Sci., 51(10): 2449–2458.
APHA, AWWA and WEF (1992). Standard methods for the examination of water and wastewater, 18th edition. American Public Health Association, American Water Works Association and Water Environment Federation, Washington, DC.
APHA, AWWA and WEF (1995). Standard methods for the examination of water and wastewater 19th edition. American Public Health Association, American Water Works Association and Water Environment Federation, Washington, DC.
APHA, AWWA and WEF (1998). Standard methods for the examination of water and wastewater 20th edition. American Public Health Association, American Water Works Association and Water Environment Federation, Washington, DC.
APHA, AWWA and WEF (2005). Standard methods for the examination of water and wastewater, 21st edition. American Public Health Association, American Water Works Association and Water Environment Federation, Washington, DC.
APHA, AWWA and WEF (2012). Standard methods for the examination of water and wastewater, 22nd edition. American Public Health Association, American Water Works Association and Water Environment Federation, Washington, DC.
Araissi, M., Ayed, I., Elaloui, E. and Moussaoui, Y. (2016). Removal of barium and strontium from aqueous solution using zeolite 4A. Water Sci. Technol., 73(7): 1628–1636.
Atassi, A., Feizoulof, C., Lecuyer, E., Nebel, J. and Medler, S. (2007). Barium removal using selective ion exchange with calcium chloride regeneration for reducing chloride discharge. Proceedings of the American Water Works Association Water Quality Technology Conference, Toronto, Ontario.
ATSDR (2007). Toxicological profile for barium. Agency for Toxic Substances and Disease Registry, U.S. Department of Health and Human Services, Public Health Service, Atlanta, Georgia.Available at: www.clearwater.asn.au/user-data/resource-files/Aust_drinking_water_guidelines.pdf
Baldi, F., Pepi, M., Burrini, D., Kniewald, G., Scali, D. and Lanciotti, E. (1996). Dissolution of barium from barite in sewage sludges and cultures of desulfovibrio desulfuricans. Appl. Environ. Microbiol., 62(7): 2398–2404.
Bhoelan, B. S., Stevering, C. H., van der Boog, A. T. J. and van der Heyden, M. A. G. (2014). Barium toxicity and the role of the potassium inward rectifier current. Clin. Toxicol., 52(6): 584–593.
Blaurock-Busch, E., Amin, O.R. and Rabah, T. (2011). Heavy metals and trace elements in hair and urine of a sample of arab children with autistic spectrum disorder. Mædica, 6(4): 247–257.
Boerlage, S., Kennedy, M.D., Bremere, I., Witkamp, G.J., Van Der Hoek, J.P. and Schippers, J.C. (2000). Stable barium sulphate supersaturation in reverse osmosis. J. Membr. Sci., 179(1–2): 53–68.
Boffito, C. (1991). Barium. Kirk-Othmer encyclopedia of chemical technology. 4th Edition. John Wiley and Sons., Hoboken, New Jersey, 3: 902–908.
Borzelleca, J.F., Condie, L.W. and Egle, J.L. (1988). Short-term toxicity (one-and ten-day gavage) of barium chloride in male and female rats. Int. J. Toxicol., 7(5): 675–685.
Brenniman, G.R., Namekata, T., Kojola, W.H., Carnow, B.W. and Levy, P.S. (1979). Cardiovascular disease death rates in communities with elevated levels of barium in drinking water. Environ. Res., 20(2): 318–324.
Brenniman, G.R., Kojola, W.H., Levy, P.S., Carnow, B.W. and Namekata, T. (1981). High barium levels in public drinking water and its association with elevated blood pressure. Arch. Environ. Health, 36(1): 28–32.
Breuer, M., Ditrich, K., Habicher, T., Hauer, B., Kesseler, M., Sturmer, R. and Zelinski, T. (2004). Industrial methods for the production of optically active intermediates. Angew. Chem. Int. Ed. Engl., 43(7): 788–824.
British Columbia Ministry of Health (2017). Personal communication from D. Fishwick.
CCME (2013). Canadian soil quality guidelines for barium: Protection of human health. Scientific criteria document PN 1493 ISBN 978-1-896997- 97-1 PDF. Canadian Council of Ministers of the Environment. Available at: www.ccme.ca/files/Resources/supporting_scientific_documents/pn_1493_basqg_scd_prob_1.0.pdf
CDC (2015). Fourth national report on human exposure to environmental chemicals. Updated tables, February. U.S. Department of Health and Human Services. Centers for Disease Control and Prevention, Atlanta, Georgia. Available at: www.cdc.gov/biomonitoring/pdf/FourthReport_UpdatedTables_Feb2015.pdf
Charette, M.A. and Sholkovitz, E.R. (2006). Trace element cycling in a subterranean estuary: Part 2. Geochemistry of the pore water. Geochim. Cosmochim. Acta, 70(4): 811–826.
Chellan, P. and Sadler, P.J. (2015). The elements of life and medicines. Philos. Trans. A Math. Phys. Eng. Sci., 373(2037).
Clifford, D.A. (1999). Ion exchange and inorganic adsorption. Chapter 9 in: Water quality and treatment: a handbook of community water supplies, 5th edition. Letterman, R.D. (ed). American Water Works Association. Denver, Colorado.
Clifford, D., Sorg, T. and Ghurye, G. (2011). Ion exchange and adsorption of inorganic contaminants. Chapter 12 in: Water quality and treatment: A handbook of drinking water. 6th edition. Edzwald, J.K. (ed.). American Water Works Association, Denver, Colorado.
Colbert, D. and McManus, J. (2005). Importance of seasonal variability and coastal processes on estuarine manganese and barium cycling in a Pacific Northwest estuary. Cont. Shelf Res., 25(11): 1395–1414.
CRC (2017). CRC handbook of chemistry and physics. Rumble, J.R.. (ed.). 98th edition. Available at: http://hbcponline.com/faces/contents/ContentsSearch.xhtml
Cuddihy, R.G. and Griffith, W.C. (1972). A biological model describing tissue distribution and whole-body retention of barium and lanthanum in beagle dogs after inhalation and gavage. Health Phys., 23(5): 621–633.
Deng, J.F., Jan, I.S. and Cheng, H.S. (1991). The essential role of a poison center in handling an outbreak of barium carbonate poisoning. Vet. Hum. Toxicol., 33(2): 173–175.
Dibello, P. M., Manganaro, J. L., Aguinaldo, E. R., Mahmood, T. and Lindahl, C. B. (2003). Barium compounds. Kirk-Othmer encyclopedia of chemical technology. Available at: http://onlinelibrary.wiley.com/doi/10.1002/0471238961.0201180904090205.a01.pub2/pdf
Diengott, D., Rozsa, O., Levy, N. and Muammar, S. (1964). Hypokalaemia in barium poisoning. Lancet, 284(7355): 343–344.
Dietz, D.D., Elwell, M.R., Davis, W.E. and Meirhenry, E.F. (1992). Subchronic toxicity of barium chloride dihydrate administered to rats and mice in the drinking water. Fundam. Appl. Toxicol., 19:527–537.
Downs, J.C.U., Milling, D. and Nichols, C.A. (1995). Suicidal ingestion of barium-sulfide-containing shaving powder. Am. J. Forensic Med. Pathol., 16(1): 56–61.
Dumont, M. (2007). Barite and witherite. Canadian minerals yearbook. Natural Resources Canada, Minerals and Metals Sector. [as cited in CCME, 2013]
ECCC (2016). Science approach document biomonitoring-based approach 2 for barium-containing substances molybdenum-containing substances silver-containing substances thallium-containing substances inorganic tin- containing substances. Environment and Climate Change Canada. Available at: www.ec.gc.ca/ese-ees/default.asp?lang=En&n=D335D89F-1#toc03
ECCC (2017). National long-term water quality monitoring data. Environment and Climate Change Canada. Available at: http://open.canada.ca/data/en/dataset/5133b19b-6a92-44b2-8b12-a49731b56421
Elder, D. and Budd, G.C. (2011). Overview of water treatment processes. Chapter 5 in: Water quality and treatment: a handbook on drinking water. 6th edition. Edzwald, J.K. (ed.). American Water Works Association, Denver, Colorado.
Elwej, A., Ghorbel, I., Marrekchi, R., Boudawara, O., Jamoussi, K., Boudawara, T., Zeghal, N. and Sefi, M. (2016a). Improvement of kidney redox states contributes to the beneficial effects of dietary pomegranate peel against barium chloride-induced nephrotoxicity in adult rats. Arch. Physiol. Biochem., 122(3): 130–140.
Elwej, A., Grojja, Y., Ghorbel, I., Boudawara, O., Jarraya, R., Boudawara, T. and Zeghal, N. (2016b). Barium chloride induces redox status unbalance, upregulates cytokine genes expression and confers hepatotoxicity in rats— alleviation by pomegranate peel. Environ. Sci. Pollut. Res., 23(8): 7559–7571.
Environment Canada (2011). National Air Pollution Surveillance Program. PM Speciation - WICPMS metals data (9 files). Available at: http://maps-cartes.ec.gc.ca/rnspa-naps/data.aspx?lang=en
Environment Canada (2013) National air pollution surveillance (NAPS) monitoring results. Environment Canada. Available at: http://maps-cartes.ec.gc.ca/rnspa-naps/data.aspx?lang=en
Ferrante, M., Conti, G.O., Rasic-Milutinovic, Z. and Jovanovic, D. (2014). Health effects of metals and related substances in drinking water. Ferrante, M., Conti, G.O., Rasic-Milutinovic, Z. and Jovanovic, D. (eds.). EWA Publishing, London, United Kingdom.
Friedman, M.J., Hill, A.S., Reiber, S.H., Valentine, R.L., Larsen, G., Young, A., Korshin, G.V. and Peng, C-Y. (2010). Assessment of inorganics accumulation in drinking water system scales and sediments. Water Research Foundation and United States Environmental Protection Agency, Denver, Colorado.
Gabelich, C.J., Williams, M.D., Rahardianto, A., Franklin, J.C. and Cohen, Y. (2007). High-recovery reverse osmosis desalination using intermediate chemical demineralization. J. Membr. Sci., 301(1–2): 131–141.
Gould, D.B. (1973). Barium sulfide poisoning. Some factors contributing to survival. Arch. Intern. Med., 132(6): 891–894.
Goullé, J.P., Mathieu, L., Castermant, J., Neveu, N., Bonneau, L., Laine, G., Bouige, D. and Lacroix, C. (2005). Metal and metalloid multi-elementary ICP-MS validation in whole blood, plasma, urine and hair. Reference values. Forensic Sci. Int. 153 (1): 39–44.
Groschen, G.E., Arnold, T.L., Morrow, W.S. and Warner, K.L. (2009). Occurrence and distribution of iron, manganese, and selected trace elements in ground water in the glacial aquifer system of the Northern United States.
U.S. Geological Survey. Available at: https://pubs.usgs.gov/sir/2009/5006/pdf/SIR2009-5006.pdf
Hamilton, S.M. (2015). Ambient groundwater geochemistry data for southern Ontario, 2007–2014; Ontario Geological Survey, Miscellaneous Release—Data 283–Revised. Available at: www.geologyontario.mndm.gov.on.ca/mndmaccess/mndm_dir.asp?type=pub&id=MRD283-REV
Han, B.H., Han, J.Y., Ahn, H.K. and Nava-Ocampo, A.A. (2010). Conventional barium enema in early pregnancy. J. Obstet. Gynaecol., 30(6): 559–562.
Han, B.H., Lee, K.S., Han, J.Y., Choi, J.S., Ahn, H.K., Ryu, H.M., Yang, J.H., Han, H.W. and Nava-Ocampo, A.A. (2011). Pregnancy outcome after 1st-trimester inadvertent exposure to barium sulphate as a contrast media for upper gastrointestinal tract radiography. J. Obstet. Gynaecol., 31(7): 586–588.
He, C., Gross, M., Westerhoff, P.K., Fox, P. and Li, K. (2011). Comparing conventional and pelletized lime softening concentrate chemical stabilization. Water Research Foundation and City of Phoenix Water Services Department, Denver, Colorado.
Health Canada (1994). Canadian Environmental Protection Act. Human health risk assessment for priority substances. Available at: www.hc-sc.gc.ca/ewh-semt/pubs/contaminants/approach/index-eng.php
Health Canada (2012a). Dietary intakes of contaminants and other chemicals for different age-sex groups of Canadians for Total Diet Study (1993–2007). Bureau of Chemical Safety, Health Products and Food Branch, Health Canada, Ottawa, Ontario. Available at: www.canada.ca/en/health-canada/services/food-nutrition/food-nutrition-surveillance/canadian-total-diet-study.html
Health Canada (2012b). Concentrations (ng/g) of trace elements in total diet study composites (1992–2012). Bureau of Chemical Safety, Health Products and Food Branch, Health Canada, Ottawa, Ontario. Available at: www.canada.ca/en/health-canada/services/food-nutrition/food-nutrition-surveillance/canadian-total-diet-study.html
Health Canada (2016). Personal communication from A. M. Tugulea.
Health Canada (2017). Licensed Natural Health Products Database.Available at: http://webprod.hc-sc.gc.ca/nhpid-bdipsn/search-rechercheReq.do
Heitland, P. and Koster, H.D. (2006). Biomonitoring of 30 trace elements in urine of children and adults by ICP-MS. Clin. Chim. Acta, 365 (1-2): 310–318.
ICRP (1980). Limits for intakes of radionuclides by workers. International Commission on Radiation Protection, Oxford, Permagon Press, ICRP Publication 30, Part 2.
ICRP (1993). Age-dependent doses to members of the public from intake of radionuclides - Part 2, Ingestion dose coefficients. International Commission on Radiation Protection, Publication 67, Ann. ICRP, 23 (3–4).
IPCS (1990). Barium. Geneva, World Health Organization, International Programme on Chemical Safety (Environmental Health Criteria 107). Available at: www.inchem.org/documents/ehc/ehc/ehc107.htm
Jha, S.K., Kumar, R. and Verma, B.S. (1993). A case of barium carbonate poisoning. J. Assoc. Physicians India, 41(11): 750–751.
Jourdan, S., Bertoni, M., Sergio, P., Michele, P. and Rossi, M. (2001). Suicidal poisoning with barium chloride. Forensic Sci. Int., 119(2): 263–265.
Kanematsu, N., Hara, M. and Kada, T. (1980). Rec assay and mutagenicity studies on metal compounds. Mutat. Res./Gen. Toxicol., 77(2): 109–116.
Koch, M., Appoloni, O., Haufroid, V., Vincent, J. and Lheureux, P. (2003). Acute barium intoxication and hemodiafiltration. J. Toxicol. Clin. Toxicol., 41(4): 363–367.
Kojola, W.H., Brenniman, G.R. and Carnow, B.W. (1979). A review of environmental characteristics and health effects of barium in public water supplies. Rev. Environ. Health, 3(1): 79–95.
Kopp, S.J., Perry, H.M., Feliksik, J.M., Erlanger, M. and Perry, E.F. (1985). Cardiovascular dysfunction and hypersensitivity to sodium pentobarbital induced by chronic barium chloride ingestion. Toxicol. Appl. Pharmacol., 77(2): 303–314.
Krause, T.L. and Stover, E.L. (1982). Evaluating water treatment techniques for barium removal. J. Am. Water Works Assoc., 74(9): 478–485.
Kravchenko, J., Darrah, T.H., Miller, R.K., Lyerly, H.K. and Vengosh, A. (2014). A review of the health impacts of barium from natural and anthropogenic exposure. Environ. Geochem. Health, 36(4): 797–814.
Krishnan, K. and Carrier, R. (2008). Approaches for evaluating the relevance of multiroute exposures in establishing guideline values for drinking water contaminants. J. Environ. Sci. Health C, 26: 300–316.
Kugler, R.T., Doyle, S. and Kind, M. (2015). Fundamental insights into barium sulfate precipitation by time-resolved
in situ synchrotron radiation wide-angle X-ray scattering (WAXS). Chem. Eng. Sci, 133: 140–147.
Legget, R.W. (1992). Fractional absorption of ingested barium in adult humans. Health Phys., 62(6): 556–561.
LeRoy, G.V., Rust, J.H. and Hasterlik, R.J. (1966). The consequences of ingestion by man of real and simulated fallout. Health Phys., 12(4): 449–473.
Lewi, Z., Warsaw, D.M. and Bar-Khayim, Y. (1964). Food-poisoning from barium carbonate. Lancet, 284(7355): 342–343.
Lovins, W., Taylor, J., Kozic, R.M., Abbasedegan, M., LeChevallier, M. and Ajy, K. (1999). Multi-contaminant removal by integrated membrane systems. Proceedings of the American Water Works Association Water Quality and Technology Conference, Denver, Colorado.
Lukasik-Głębocka, M., Sommerfeld, K., Hanć, A., Grzegorowski, A., Barałkiewicz, D., Gaca, M. and Zielińska- Psuja, B. (2014). Barium determination in gastric contents, blood and urine by inductively coupled plasma mass spectrometry in the case of oral barium chloride poisoning. J. Anal. Toxicol., 38(6): 380–382.
Lytle, D.A., Sorg, T., Wang, L. and Chen, A. (2014). The accumulation of radioactive contaminants in drinking water distribution systems. Water Res., 50: 396–407.
Manitoba Conservation and Water Stewardship (2017). Personal communication with J. Muenster and K. Philip.
McCarron, D.A., Morris, C.D., Henry, H.J. and Stanton, J.L. (1984). Blood pressure and nutrient intake in the United States. Science, 224: 1392–1398.
McCauley, P.T. and Washington, I.S. (1983). Barium bioavailability as the chloride, sulfate, or carbonate salt in the rat. Drug Chem. Toxicol., 6(2): 209–217.
McCauley, P.T., Douglas, B.H., Laurie, R.D. and Bull, R.J. (1985). Investigations into the effect of drinking water barium on rats. In: Advances in modern environmental toxicology. Vol. IX. Princeton Publishing Co., Princeton, NJ. (ed.). pp. 197.
McComish, M.F. and Ong, J.H. (1988). Trace metals. Chapter 7 in: Environmental inorganic chemistry. Bodek, I., Lyman, W.J., Reehl, W.F. and Rosenblatt, D.H. (eds.). Pergamon Press, Amsterdam.
McNally, W.D. (1925). Two deaths from the administration of barium salts. J. Am. Med. Assoc., 84(24): 1805–1807.
Menzie, C.A., Southworth, B., Stephenson, G. and Feisthauer, N. (2008). The importance of understanding the chemical form of a metal in the environment: The case of barium sulfate (barite). Hum. Ecol. Risk Assess. 14(5): 974–991.
Miller, R., Featherstone, J., Curzon, M., Mills, T., and Shields, C. (1985). Barium in teeth as indicator of body burden. Adv. Mod. Env., 9: 211.
Ministère du Développement durable, de l’Environnement et de la Lutte contre les changements climatiques (2017). Personal communication from C. Robert and A. Bolduc.
Moore, W.S. (2010). A reevaluation of submarine groundwater discharge along the southeastern coast of North America. Global Biogeochem. Cycles, 24(4): n/a-n/a.
Morton, W. (1945). Poisoning by barium carbonate. Lancet, 246(6380): 738–739.
Murray, J.W. (1975). The interaction of metal ions at the manganese dioxide-solution interface. Geochim. Cosmochim. Acta, 39(4): 505–519.
Myers, A.G., Snoeyink, V.L. and Snyder, D.W. (1985). Removing barium and radium through calcium cation exchange. J. Am. Water Works Assoc., 77(5): 60–66.
New Brunswick Department of Health (2017). Personal communication with K. Gould.
New Zealand Ministry of Health (2013). Survey of selected samples of tattoo inks for the presence of heavy metals. Ministry of Health, Wellington.
Newfoundland and Labrador Department of Environment and Conservation (2017). Personal communication from H. Khan.
NHMRC (2011). Australian drinking water guidelines. National Health and Medical Research Council. Available at: www.nhmrc.gov.au/_files_nhmrc/file/publications/nhmrc_adwg_6_version_3.3_2.pdf
Ning, R.Y., Tarquin, A., Trzcinski, M. and Patwardhan, G. (2006). Recovery optimization of RO concentrate from desert wells. Desalination, 201(1–3): 315–322.
Nishioka, H. (1975). Mutagenic activities of metal compounds in bacteria. Mutat. Res. Envir. Muta., 31(3): 185–189.
NRC (1995). Nutrient requirements of laboratory animals. National Research Council. National Academy Press, Washington, DC, p. 13.
NSF/ANSI (2015). Standard 60: Drinking water treatment chemicals—health effects. NSF International/American National Standards Institute, Ann Arbor, Michigan.
NSF/ANSI (2016a). Standard 44: Residential cation exchange water softeners. NSF International/American National Standards Institute, Ann Arbor, Michigan.
NSF/ANSI (2016b). Standard 58: Reverse osmosis drinking water treatment systems. NSF International/American National Standards Institute, Ann Arbor, Michigan.
NSF/ANSI (2016c). Standard 62: Drinking water distillation systems. NSF International/American National Standards Institute, Ann Arbor, Michigan.
NSF/ANSI (2016d). Standard 61: Drinking water system components—health effects. NSF International/American National Standards Institute, Ann Arbor, Michigan.
NTP (1994). Toxicology and carcinogenesis studies of barium chloride dihydrate (CAS No. 10326-27-9) in: F344/N rats and B6C3F1 mice (Drinking Water Studies). National Toxicology Program, Tech. Rep. Ser., 432: 1–285.
Odell, L. (2010). Treatment technologies for groundwater. American Water Works Association. Denver, Colorado.
Ogen, S., Rosenbluth, S. and Eisenberg, A. (1967). Food poisoning due to barium carbonate in sausage. Isr. J. Med. Sci., 3(4): 565–568.
Ohgami, N., Hori, S., Ohgami, K., Tamura, H., Tsuzuki, T., Ohnuma, S. and Kato, M. (2012). Exposure to low-dose barium by drinking water causes hearing loss in mice. Neurotoxicol., 33(5): 1276–1283.
Ohgami, N., Mitsumatsu, Y., Ahsan, N., Akhand, A.A., Li, X., Iida, M., Yajima, I., Naito, M., Wakai, K., Ohnuma,
S. and Kato, M. (2016). Epidemiological analysis of the association between hearing and barium in humans. J. Expos. Sci. Environ. Epidemiol., 26(5): 488–493.
Padilla, M.A., Elobeid, M., Ruden, D.M. and Allison, D.B. (2010). An examination of the association of selected toxic metals with total and central obesity indices: NHANES 99-02. Int. J. Environ. Res. Public Health, 7: 3332– 3347.
Parks, J.L. and Edwards, M. (2006). Precipitative removal of As, Ba, B, Cr, Sr and V using sodium carbonate, ASCE J. Environ. Eng., 132(5): 489–496.
Paschal, D.C., Ting, B.G., Morrow, J.C., Pirkle, J.L., Jackson, R.J., Sampson, E.J., Miller, D.T. and Caldwell, K.L. (1998). Trace metals in urine of United States residents: reference range concentrations. Environ. Res., 76 (1): 53–59.
Payen, C., Dellinger, A., Pulce, C., Cirimele, V., Carbonnel, V., Kintz, P. and Descotes, J. (2011). Intoxication by large amounts of barium nitrate overcome by early massive K supplementation and oral administration of magnesium sulphate. Hum. Exp. Toxicol., 30(1): 34–37.
PEI Department of Communities, Land and Environment (2017). Personal communication from G. Somers.
Peng, C., Hill, A.S., Friedman, M.J., Valentine, R.L., Larson, G.S., Romero, A.M.Y., Reiber, S.H. and Korshin, G.V. (2012). Occurrence of trace inorganic contaminants in drinking water distribution systems. J. Am. Water Works Assoc., 104(3): 53–54.
Perry, H.M., Perry, E.F., Erlanger, M.W. and Kopp, S.J. (1985). Barium-induced hypertension. Chapter 20 in: Inorganics in drinking water and cardiovascular disease. Calabrese, E. (ed). Princeton, New Jersey, Princeton Publishing Co., pp. 221–229.
Perry Jr, H.M., Perry, H.M., Kopp, S.J., Kopp, S.J., Perry, E.F., Perry, E.F., Erlanger, M.W. and Erlanger, M.W. (1989). Hypertension and associated cardiovascular abnormalities induced by chronic barium feeding. J. Toxicol. Environ. Health, 28(3): 373–388.
Phelan, D.M., Hagley, S.R. and Guerin, M.D. (1984). Is hypokalaemia the cause of paralysis in barium poisoning? Br. Med. J., 289(6449): 882.
Rahardianto, A., Gao, J., Gabelich, C.J., Williams, M.D. and Cohen, Y. (2007). High recovery membrane desalting of low-salinity brackish water: Integration of accelerated precipitation softening with membrane RO. J. Membr. Sci., 289(1–2): 123–137.
Rai, D., Zahara, J., Schwab, A., Schmidt, R., Girvin, D. and Rogers, J. (1984). Chemical attenuation rates, coefficients and constant in leachate migration. Vol I: A critical review. Pacific Northwest Laboratories, Richland, Washington.
Rasmussen, P.E., Subramanian, K.S. and Jessiman, B.J. (2001). A multi-element profile of housedust in relation to exterior dust and soils in the city of Ottawa, Canada. Sci. Total Environ., 267(1–3): 125–140.
Rasmussen et al. (2016). Preliminary exposure data for five metals from 2014-2017 CMP(3) research. February 15, 2016. Exposure and Biomonitoring Division, Health Canada, Ottawa, Ontario. Personal communication, unpublished data [as cited in ECCC, 2016].
Rhyee, S.H. and Heard, K. (2009). Acute barium toxicity from ingestion of "snake" fireworks. J. Med. Toxicol., 5(4): 209–213.
Rossman, T.G., Molina, M., Meyer, L., Boone, P., Klein, C.B., Wang, Z., Li, F., Lin, W.C. and Kinney, P.L. (1991). Performance of 133 compounds in the lambda prophage induction endpoint of the Microscreen assay and a comparison with S. typhimurium mutagenicity and rodent carcinogenicity assays. Mutat. Res./Gen. Toxicol., 260(4): 349–367.
Roza, O. and Berman, L.B. (1971). The pathophysiology of barium: hypokalemic and cardiovascular effects. J. Pharmacol. Exp. Ther., 177(2): 433–439.
Saskatchewan Water Security Agency (2017). Personal communication from S. Ferris.
Sato, I., Kudo, H. and Tsuda, S. (2011). Removal efficiency of water purifier and adsorbent for iodine, cesium, strontium, barium and zirconium in drinking water. J. Toxicol. Sci., 36(6): 829–834.
SCC (2018). Directory of accredited product, process and service certification bodies. Standards Council of Canada, Ottawa, Ontario. Available at: www.scc.ca/en/accreditation/product-process-and-service-certification/directory-of-accredited-clients
Schock, M. Hyland, R. and Welch, M. (2008). Occurrence of contaminant accumulation in lead pipe scales from domestic drinking-water distribution systems. Environ. Sci. Technol, 42(12): 4285–4291.
Schroeder, H.A., Tipton, I.H. and Nason, P. (1972). Trace metals in man: strontium and barium. J. Chronic Dis., 25: 491–517.
Schroeder, H.A. and Mitchener, M. (1975a). Life-term studies in rats: effects of aluminum, barium, beryllium, and tungsten. J. Nutr., 105(4): 421–427.
Schroeder, H.A. and Mitchener, M. (1975b). Life-term effects of mercury, methyl mercury, and nine other trace metals on mice. J. Nutr, 105(4): 452–458.
Silva, T.A., Freitas, K.A. and Damasceno, J.J.R. (2003). Experimental evaluation of conical-cylindrical thickeners operating with aqueous barium sulfate suspensions. Mater. Sci. Forum, 416–418(1): 731–736.
Sirover, M.A. and Loeb, L.A. (1976a). Infidelity of DNA synthesis in vitro: screening for potential metal mutagens or carcinogens. Science, 194(4272): 1434–1436.
Sirover, M.A. and Loeb, L.A. (1976b). Metal-induced infidelity during DNA synthesis. Proc. Natl. Acad. Sci. U. S. A., 73(7): 2331–2335.
Snoeyink, V., Jongeward, C., Myers, A. and Richter, S. (1984). Barium and radium in water treatment plant wastes.
U. S. Environment Protection Agency. Municipal Environmental Research Laboratory, Cincinatti, Ohio. EPA 600- S2-85-006.
Snoeyink, V.L., Cairns-Chambers, C. and Pfeffer, J.L. (1987a). Strong-acid ion exchange for removing barium, radium, and hardness. J. Am. Water Works Assoc., 79(8): 66–72.
Snoeyink, V., Chambers, C., Schmidt, C., Manner, r., Myers, A., Pfeffer, J., Richter, J., Richter, S. and Snyder, D. (1987b). Removal of barium and radium from groundwater. Water Engineering Research Laboratory, U. S. Environment Protection Agency, Cincinatti, Ohio. EPA 600-M-86-021.
Snyder, D.W., Snoeyink, V.L. and Pfeffer, J.L. (1986). Weak-acid ion exchange for removing barium, radium, and hardness. J. Am. Water Works Assoc., 78(9): 98–104.
Sorg, T.J. and Logsdon, G.S. (1980). Treatment technology to meet the interim primary drinking water regulations for inorganics: Part 5. J. Am. Water Works Assoc., 72(7): 411–422.
Storz, P., Döppler, H. and Toker, A. (2005). Protein kinase d mediates mitochondrion-to-nucleus signaling and detoxification from mitochondrial reactive oxygen species. Mol. Cell. Biol. 25(19): 8520–8530.
Sugiyama, M., Hori, T., Kihara, S. and Matsui, M. (1992). A geochemical study on the specific distribution of barium in Lake Biwa, Japan. Geochim. Cosmochim. Acta, 56(2): 597–605.
Talwar, K.K. and Sharma, B.K. (1979). Myocardial damage due to barium chloride poisoning. Indian Heart J., 31(4): 244–245.
Tang, J. and Johannesson, K.H. (2010). Ligand extraction of rare earth elements from aquifer sediments: implications for rare earth element complexation with organic matter in natural waters. Geochim. Cosmochim. Acta, 74(23): 6690–6705.
Tardiff, R.G., Robinson, M. and Ulmer, N.S. (1980). Subchronic oral toxicity of BaCl2 in rats. J. Environ. Pathol. Toxicol., 4(5–6): 267.
Taylor, D.M., Bligh, P.H. and Duggan, M.H. (1962). The absorption of calcium, strontium, barium and radium from the gastrointestinal tract of the rat. Biochem. J., 83: 25–29.
Taylor, S.R. (1964). Abundance of chemical elements in the continental crust: A new table. Geochim. Cosmochim. Acta, 28: 1273–1285.
Thomas, M., Bowie, D. and Walker, R. (1998). Acute barium intoxication following ingestion of ceramic glaze. Postgrad. Med. J., 74(875): 545–546.
Tipton, I. H., Stewart, P. L. and Dickson, J. (1969). Patterns of elemental excretion in long term balance studies. Health. Phys. 16: 455–462.
Tudorache, A., Marin, C., Badea, I.A. and Vladescu, L. (2010). Barium concentrations and speciation in mineral natural waters of central Romania. Environ. Monit. Assess., 165(1–4): 113–123.
U.S. EPA (1993). Barium metaborate R.E.D. facts. U.S. Environmental Protection Agency. Available at: www3.epa.gov/pesticides/chem_search/reg_actions/reregistration/fs_PC-011101_1-Dec-93.pdf
U.S. EPA. (1994a). Method 200.7 Revision 4.4. Determination of metals and trace elements in water and wastes by inductively coupled plasma-atomic emission spectrometry. Environment Monitoring Systems Laboratories, Office of Research and Development, U.S. Environmental Protection Agency, Cincinnati, Ohio.
US EPA. (1994b). Method 200.8 Revision 5.4. Determination of trace elements in waters and wastes by inductively coupled plasma-mass spectrometry. Environment Monitoring Systems Laboratories, Office of Research and Development, U.S. Environmental Protection Agency, Cincinnati, Ohio.
U.S. EPA (1995). National primary drinking water regulations. Barium. Office of Water, U.S. Environmental Protection Agency. EPA-811-F-95-002b-T. Available at: www.epa.gov/ground-water-and-drinking-water/national-primary-drinking-water-regulations#Inorganic
U.S. EPA. (1998). Small system compliance technology list for the non-microbial contaminants regulated before 1996. Office of Water, U.S. Environmental Protection Agency, Washington, DC. EPA 815-R-98-002.
U.S. EPA. (2003). Method 200.5 Revision 4.2. Determination of trace elements in drinking water by axially viewed inductively coupled plasma - atomic emission spectrometry. National Exposure Research Laboratory, Office of Research and Development, U.S. Environmental Protection Agency, Cincinnati, Ohio. EPA 600-R-06-115.
U.S. EPA (2004). Risk assessment guidance for superfund. Volume I: Human health evaluation manual (Part E, Supplemental guidance for dermal risk assessment). Office of Superfund Remediation and Technology Innovation, U.S. Environmental Protection Agency, Washington, DC. EPA/540/R/99/005 OSWER 9285.7-02EP PB99-963312.
U.S. EPA (2005). Toxicological review of barium and compounds (CAS No. 7440-39-3) In support of summary information on the Integrated Risk Information System (IRIS). U.S. Environmental Protection Agency, Washington, DC. EPA/635/R-05/001.
U.S. EPA. (2009). Analytical feasibility support document for the second six-year review of existing national primary drinking water regulations. Office of Water, U.S. Environmental Protection Agency, Washington, DC. EPA 815-B- 09-003.
U.S. EPA. (2012a). Federal Registry Part II. Revisions of the unregulated contaminant monitoring regulation (UCMR
3) for public water systems; final rule. Vol 77, No 85. May 2, 2012. U.S. Environmental Protection Agency, Washington, DC.
U.S. EPA. (2012b). Drinking water treatability database. Office of Research and Development, U.S. Environmental Protection Agency, Washington, DC. Available at: http://iaspub.epa.gov/tdb/pages/general/home.do
U.S. EPA (2012c). 2012 Edition of the drinking water standards and health advisories. Office of Water, U.S. Environmental Protection Agency, Washington, DC. Available at: https://nepis.epa.gov/Exe/ZyPDF.cgi/ P100N01H.PDF?Dockey=P100N01H.PDF
U.S. EPA. (2016). Analytical methods approved for drinking water compliance. Monitoring of inorganic contaminants and other inorganic constituents. Office of Water (MS-4606M), U.S. Environmental Protection Agency, Washington, DC. EPA 815-B-16-014.
Van Leeuwen, M.L.J., Bruinsma, O.S.L. and van Rosmalen, G.M. (1996). Three zone approach for precipitation of barium sulphate. J. Crystal Growth, 166: 1004–1008.
Venugopal, B. and Luckey, T.D. (1978). Metal toxicity in mammals, Volume 2: Chemical toxicity of metals and metalloids. Plenum Press, New York.
Wetherill, S.F., Guarino, M.J. and Cox, R.W. (1981). Acute renal failure associated with barium chloride poisoning. Ann. Intern. Med., 95(2): 187–188.
WHO (1990). Environmental health criteria 107. Barium. World Health Organization, International Program on Chemical Safety. Available at: www.inchem.org/documents/ehc/ehc/ehc107.htm
WHO (2001). Barium and barium compounds. Concise international chemical assessment document, World Health Organization, Geneva, Switzerland.
WHO (2016). Barium in drinking-water. Background document for development of WHO guidelines for drinking- water quality. World Health Organization, Geneva, Switzerland. Available at: www.who.int/water_sanitation_health/ water-quality/guidelines/chemicals/barium-background-jan17.pdf
Willey, B.R. (1987). Finding treatment options for inorganics. Water Eng. Manage., 134(10): 28–31.
Wones, R.G., Stadler, B.L. and Frohman, L.A. (1990). Lack of effect of drinking water barium on cardiovascular risk factors. Environ. Health Perspect., 85: 355–359.
WQA. (2013). Barium fact sheet: Technical application bulletin.Water Quality Assocation, National Headquarters & Laboratory, Lisle, Illinois. Available at: www.wqa.org/Portals/0/Technical/Technical%20Fact%20Sheets/ 2014_Barium.pdf
Yesilada, E. (2001). Genotoxicity testing of some metals in the drosophila wing somatic mutation and recombination test. Bull. Environ. Contam. Toxicol., 66(4): 464–469.
Yukon Environmental Health Services (2017). Personal communication with P. Brooks.
Zasoski, R.J. and Burau, R.G. (1988). Sorption and sorptive interaction of cadmium and zinc on hydrous manganese oxide. Soil Sci. Soc. Am. J., 52:81–87.
Appendix A: List of acronyms
- ATSDR
- Agency for Toxic Substances and Disease Registry
- BMD
- benchmark dose
- BMDL
- benchmark dose lower confidence limit
- BW
- body weight
- CCME
- Canadian Council of Ministers of the Environment
- CDC
- Centers for Disease Control
- CI
- confidence interval
- CSA
- Canadian Standards Association
- CTDS
- Canadian Total Diet Study
- DL
- detection limit
- DNA
- deoxyribonucleic acid
- ECG
- electrocardiogram
- ED/EDR
- electrodialysis/electrodialysis reversal
- EPA
- Environmental Protection Agency (U.S.)
- HBV
- health-based value
- IARC
- International Agency for Research on Cancer
- ICRP
- International Commission on Radiological Protection
- IPCS
- International Programme on Chemical Safety
- IX
- ion exchange
- LD50
- median lethal dose
- LOAEL
- lowest-observed-adverse-effect level
- MDL
- method detection limit
- NF
- nanofiltration
- NHANES
- National Health and Nutrition Examination Survey
- NOAEL
- no-observed-adverse-effect level
- NRC
- National Research Council
- NSF
- NSF International
- NTP
- National Toxicology Program (U.S.)
- OR
- odds ratio
- RO
- reverse osmosis
- SAC
- strong-acid cation
- SCC
- Standards Council of Canada
- TDI
- tolerable daily intake
- TDS
- total dissolved solids
- WAC
- weak-acid cation
- WHO
- World Health Organization