Toward Front-of-Package Nutrition Labels for Canadians
Consultation Document
Food Directorate, Health Products and Food Branch, Health Canada
Table of Contents
- A. Introduction
- B. Background
- C. Proposed Approach and Consultation
- Element 1 - An FOP labelling approach for foods high in sodium, sugars and saturated fat
- Element 2 - Thresholds for FOP labelling approach for foods high in sodium, sugars and saturated fat
- Element 3 - Symbols for a high in FOP labelling approach.
- Element 4 - Updating nutrient content claims and other nutrition-related statements
- Element 5 - High-intensity sweetener labelling
- D. Conclusion
- E. Openness and Transparency
- F. References
A. Introduction
Purpose
Health Canada is proposing to introduce mandatory front-of-package (FOP) labelling requirements for foods high in nutrients of public health concern - sodium, sugars and saturated fat - due to excessive intakes. In addition, we are proposing to concurrently update requirements for other information on the front of food packages, including certain claims and sweetener labelling. The purpose of this document is to describe the rationale and elements for Health Canada's proposals and to solicit input from interested Canadians and stakeholders on these proposals.

Download the entire report
(PDF format, 1.04 MB, 22 pages)
Organization: Health Canada
Type: Consultation
Current context
On October 24, 2016 the Minister of Health launched a Healthy Eating Strategy as part of the Government's vision for a healthy Canada (the Strategy)Footnote 1 in response to several food and nutrition commitments identified in her 2015 Mandate Letter from the Prime Minister of CanadaFootnote 2. The Strategy unites Health Canada's ongoing nutrition efforts and successes to date with new, complementary initiatives to help create a food environment that makes healthier eating choices easier for Canadians. The Strategy is part of the Government of Canada's broader commitment to promoting public health and healthy lifestyles.
One of the key initiatives under the Strategy, FOP aims to improve food labelling to help make it easier for Canadians to make healthier food choices at the grocery store. While existing nutrition labelling tools are very useful to many consumers when making food purchasing decisions, some consumers find the information provided too complex to understand and use. To help address this, Health Canada is proposing to introduce FOP labelling requirements on prepackaged foods high in sodium, sugars and saturated fat.
The objectives of FOP labelling are to:
- Provide quick and easy guidance to encourage consumers to make informed choices about foods in relation to sodium, sugars and saturated fat; and
- Encourage the availability of foods lower in these nutrients, thereby reducing risks to health.
B. Background
Diet as a risk factor for chronic disease
Chronic non-communicable disease is a major public health concern in Canada. The incidence of type 2 diabetes continues to increaseFootnote 3 and cardiovascular disease (heart disease and stroke) is among the leading causes of death in CanadaFootnote 4. With high rates of obesityFootnote 5 and high blood pressure, along with an aging Canadian populationFootnote 6, the impact of chronic diseases is likely to continue to increase unless action is taken to reduce modifiable risk factors.
An unhealthy diet is one of the top risk factors for obesity and chronic disease burden in CanadaFootnote 7. In Canada, the annual economic burden of unhealthy eating was estimated at $6.6 billion, including direct health care costs of $1.3 billionFootnote 8. Obesity itself is estimated to create an economic burden of between $4.6 billion to $7.1 billion annuallyFootnote 9. Diets high in sodium, saturated fat and sugars are strongly linked to obesity and/or major chronic diseasesFootnote 10. Taking action to reduce intakes of these nutrients can help reduce important risk factors for chronic disease burden in Canada.
Food labels as a public health tool
Food labels serve an important public health function by providing the information that consumers need to make informed food choices relevant to their health (e.g., nutrition and allergen information). Other core mandatory information (e.g., common name, net quantity, list of ingredients and best before date) is also necessary to assist consumers in their food choices and to protect a fair and secure marketplace.
In Canada, responsibility for food labelling at the federal level is shared between Health Canada and the Canadian Food Inspection Agency (CFIA). The CFIA administers and enforces food labelling policies and regulations and is currently consulting on modernizing non-health and safety aspects of food labellingFootnote 11.
Health Canada establishes policies, regulations and standards relating to the health, safety and nutritional quality of food sold in Canada. Nutrition information on food labels provides product-specific nutrition information to help Canadians make informed food choices, in planning healthy meals and in the dietary management of diseases of public health concern. In Canada, the Food and Drug Regulations address three main types of nutrition information on foods labels: the Nutrition Facts table (NFt); nutrient content claims; and some types of health claims.
The NFt became mandatory on most prepackaged foods in 2007. The NFt provides information on the amount of calories and 13 core nutrients in a given serving of food. Sodium and saturated fat are two of several nutrients whose amounts must be declared both in grams and as a percent of the Daily Value (% DV). Currently, sugars must be declared in grams only. The % DV was developed to help consumers understand the significance of the amount of a nutrient in the context of daily intake. Benchmarks have been developed to assist consumers in interpreting the information on the amount of a nutrient, i.e., 5% or less is "a little" and 15% or more is "a lot" of a nutrient. Results of an analysis of simulated diets, compatible with Canada's Food Guide and reflecting common food choices, suggest that, compared to higher thresholds, choosing foods below 15% DV per serving would help Canadians keep their intakes of nutrients of concern, such as sodium, below levels that contribute to diet-related chronic diseasesFootnote 12.
In 2014, Health Canada engaged Canadians and stakeholders on ways to improve the NFt. Based on the feedback receivedFootnote 13, Health Canada proposed updates to the NFt regulations that were pre-published in Canada Gazette, Part I, in June 2015Footnote 14 and open for public comments for a period of 75 days. As part of the updates, Health Canada proposed establishing a DV for sugars based on 100 grams and requiring that the % DV for sugars be declared in the NFt. Adjustments to the proposals have been made based on the feedback received during the comment period and the final regulatory amendments are being developed.
The NFt is a useful tool, and the updates will improve its scientific accuracy and usability; however, its location (being on the back or side) may not facilitate consumers' ability to make informed, healthy food purchases, particularly when time or motivation are limited. Additionally, the NFt can be complex for some consumers to understand and use at the point of purchase due to the amount of nutrition information providedFootnote 15. Many Canadians do not routinely choose foods that support good health, in particular when it comes to limiting foods high in nutrients of public health concern (i.e., sodium, sugars, and saturated fat)Footnote 16.
Nutrient content claims and health claims, the other types of regulated nutrition information on food labels, help consumers in their decision making process. Nutrient content claims are voluntary statements or expressions which describe, directly or indirectly, the level of a nutrient or energy in a food or a group of foods; for example, "low in sodium", "free of saturated fat", or "excellent source of calcium". Nutrient content claims further enable consumers to make informed food choices by interpreting the nutrient values. Health claims go one step further by stating that a relationship exists between consumption of a food and a person's health; for example, "A healthy diet containing foods high in potassium and low in sodium may reduce the risk of high blood pressure, a risk factor for stroke and heart disease."
However, there are limitations to these claims. These statements focus on positive attributes of the food, but do not signal when foods are high in nutrients with negative public health impacts. For example, a product that is "an excellent source of calcium" may still be high in saturated fat, sodium or sugars, and the consumer would still need to refer to the NFt to make an informed choice. Furthermore, because they are voluntary (i.e., manufacturers can choose which products to use the claim for), they do not appear on all foods in the food supply in a consistent manner.
FOP systems: science and international context
FOP labelling has the potential to build on existing nutrition labelling tools and address their limitations. FOP systems use symbols and nutrient criteria to indicate that a product has certain nutritional characteristics. It is a tool that can complement the NFt and offer consumers a simplified and visible indicator to help them make informed food choices when they are limited by time, motivation, or other factors. There has been a growing interest worldwide to capitalize on this approach to provide simplified information to help people make informed choices.
For the last decade, many different types of FOP nutrition labelling systems have proliferated. FOP symbols can be classified into two systems: nutrient-specific and summary systems. Nutrient-specific FOP systems focus on a limited number of key nutrients. For example, Chile recently introduced a mandatory FOP warning label on foods high in calories, sugars, sodium or saturated fat. The United Kingdom has a voluntary "traffic light" system that uses colours (red, amber, green) to convey a ranking for total fat, saturated fat, sugars and salt (sodium) in a food. Summary systems provide an overall nutritional score based on a variety of complex nutrient criteria. The Health Star Rating System in Australia and New Zealand is an example of a graded summary system (i.e., score on a 5-point scale), whereas the Nordic Keyhole system is an example of a binary summary system (i.e., foods that meet certain criteria can use the logo).
While FOP systems are a relatively recent development in food labelling, there is evidence to support the role of FOP labelling in helping consumers identify healthier food options. Current evidence suggests that nutrient-specific interpretive approaches, such as the United Kingdom's multiple traffic light system, are more likely to help consumers identify healthier products compared to summary systems. A review of 28 studies noted that FOP labels should convey specific nutrient levels with descriptive text (i.e., high/medium/low) and that the highlighted nutrients should be associated with the most prevalent health problemsFootnote 17. A systematic review of 38 studies found that FOP labelling can help consumers make better food choices and that, in general, nutrient-specific FOP labels, rather than summary systems, more easily help consumers identify healthier productsFootnote 18. A 2015 study of four types of FOP systems in Europe concluded that any structured and legible presentation of key nutrients on the FOP label is sufficient to enable consumers to detect a healthier alternative within a food categoryFootnote 19. Similarly, a 2016 systematic review and meta-analysis on the impacts of food labelling systems shows that interpretive nutrition labels may be an effective approach to empowering consumers in choosing healthier productsFootnote 20.
There is also evidence that FOP labelling can motivate food manufacturers and processors to develop products with lower levels of nutrients that contribute to chronic diseases. In New Zealand, the "Pick the Tick" FOP program succeeded in the removal of 33 tonnes of salt from the food supply over the course of a year through product reformulationFootnote 21.
In the Netherlands, the Choices logo motivated food manufacturers to formulate products with a healthier product compositionFootnote 22. In Finland, a warning label is required on products high in salt. Since the warning was implemented in the early 1990s, along with other related policies, the salt content of studied foods has decreased by 20-25%Footnote 23.
The Institute of Medicine of the National Academies (IOM) has also supported FOP labelling. An IOM committee, established to assess FOP systems, recommended the use of a single, standardized FOP system that can be easily understood by most consumers to help simplify and clarify nutrition informationFootnote 24. The expert committee also concluded that the best use of FOP labelling would be to help consumers identify and select foods based on the nutrients most strongly linked to public health concerns.
In 2016, the CodexFootnote i Committee on Food Labelling supported the need to assist consumers in making healthier choices through the use of simplified, science-based nutrition information on the front of food packages and agreed to establish a working group to take stock of current FOP systems and consider the need to develop global principles for FOP systemsFootnote 25. The Codex Committee emphasized that, while it initiates this work, countries that have started or are planning to implement FOP labelling should still proceed with their work, as this will provide valuable information for the working group.
In Canada, there is considerable interest in FOP nutrition labelling from consumers and health-focused organizations. During Health Canada's 2014 consultations with consumers and parents on ways to improve nutrition information on food labels, consumers expressed confusion with the array of information on food labels and asked for simple, consistent and credible information on the front of food labels. The Provincial and Territorial Governments and several Canadian health stakeholders have long advocated for federal government action on FOP labelling to help consumers make more informed food choices.
In addition, between October 2014 and June 2015, the Standing Senate Committee on Social Affairs, Science and Technology heard testimony from a broad range of stakeholders on the increasing incidence of obesity in Canada. In its final report, Obesity in Canada, the Senate Committee recommended that the federal government undertake a regulatory approach to mandate the use of FOP labelling on prepackaged foods displaying a NFtFootnote 26.
Nutrients of public health concern
FOP labelling can be used to highlight nutrients of public health concern among Canadians. Sodium, sugars and saturated fat are considered nutrients of concern, because they are associated with increased risk of chronic disease and dietary survey data indicates that Canadians consume them in excess of recommended limits.
Sodium is an essential nutrient found in salt and many other foods. The IOM, which develops nutrient reference values that underpin dietary guidance in both the United States and Canada, concluded that the amount of sodium considered sufficient to promote good health in adults is 1500 milligrams per day. The IOM also concluded that sodium intakes above 2300 milligrams per day (equivalent to about 1 teaspoon of salt) increase the risk of hypertension, which is a major cause of cardiovascular diseaseFootnote 27. Health Canada's recent Evidence Review for Dietary Guidance concluded that the evidence for excess sodium intake and high blood pressure continues to be convincingFootnote 28. The daily average sodium consumption of Canadians is currently estimated to be 3400 milligramsFootnote 29, well above the recommended limit.
Sugars are a class of carbohydrate that include sugars naturally occurring in fruits, vegetables and dairy products as well as sugars added to foods during processing. Excess sugars intake can lead to excess calorie consumption, a contributing factor to overweight and obesity. Obesity is a risk factor for cardiovascular disease, type 2 diabetes and cancerFootnote 3. Health Canada's Evidence Review for Dietary Guidance found that the evidence base was convincing for the relationship between sugars-sweetened beverages and adiposity (fat mass) in children and the relationship between added sugars and obesity or type 2 diabetesFootnote 27. The World Health Organization (WHO) recommends that individuals reduce their intake of free sugarsFootnote ii to less than 10% of total energy intakeFootnote 30, which is equivalent to about 50 grams per day based on a 2000 calorie reference diet. Data suggests that most Canadians have intakes above the WHO recommendationFootnote 31.
Saturated fat is one type of fat found in foods. Major sources include certain cheese, pizza, grain- and dairy-based desserts, chicken mixed dishes, sausages, bacon and burgers. It is also found in certain vegetable oils such as coconut and palm kernel oil. The IOM recommends that saturated fat intake be as low as possible while consuming a nutritionally adequate dietFootnote 32. Health Canada's Evidence Review for Dietary Guidance concluded that lower intakes of saturated fat, through replacement with unsaturated fat, help reduce the risk of cardiovascular diseaseFootnote 27. The Joint Expert Consultation of the Food and Agriculture Organization of the United Nations (FAO) and WHO recommends that saturated fat intake not exceed 10% of total energy intakeFootnote 33, which is approximately 20 grams per day for a 2000 calorie reference diet. The estimated Canadian population average intake of saturated fat from the Canadian Community Health Survey Cycle 2.2 is approximately 10% of energy (20 grams)Footnote 34 and has remained relatively stable in subsequent yearsFootnote 35. These intake data mean that many Canadians have saturated fat intakes above the FAO/WHO recommendation. The 2015 United States Dietary Guidelines Advisory Committee also concluded that Americans overconsumed saturated fat, based on intake data for 2007-2010 and the 2010 dietary guidelines of less than 10% of total energy from saturated fatFootnote 36.
C. Proposed Approach and Consultation
Element 1 - An FOP labelling approach for foods high in sodium, sugars and saturated fat
Health Canada is proposing to introduce a new, mandatory FOP nutrition symbol on prepackaged foods. Foods that exceed a predetermined threshold for sodium, sugars or saturated fat would be required to place a symbol on the principal display panel to indicate that the food is high in that nutrient(s). The analysis of different types of FOP systems and the selection of the proposed approach was guided by an assessment of whether the approach would achieve the stated objectives and consistency with existing labelling policies. The proposed approach was selected based on consideration of international models, the IOM recommendations and an evaluation of the intersection between various FOP systems and current Canadian food labelling policies.
By implementing a mandatory approach, rather than a voluntary approach, Health Canada could create a consistent and credible system that consumers could rely on for quick information on key nutrients of concern that would allow them to more easily compare products. It would also maintain a level playing field for all food products, whether domestically produced or imported.
By highlighting sodium, sugars and saturated fat, consumers’ attention would be drawn to the nutrients of public health concern related to excessive intakes. A nutrient-specific approach would build on and complement existing nutrition labelling tools in Canada, including the NFt and nutrient content claims. The proposed approach would be transparent, as consumers would see which nutrient is flagged in the FOP symbol and they could refer to the NFt to see the actual nutrient amounts. In addition, should consumers be limited by time, motivation or other factors FOP symbols would give consumers an additional tool to help them make informed choices about individual foods and overall diet. For example, FOP symbols may help a consumer choose more easily between two similar products and may also be an easy visual cue to help consumers balance their overall diet.
Furthermore, food manufacturers may take the opportunity to reformulate their foods to reduce the amount of sodium, sugars and saturated fat below the established thresholds to avoid the use of the FOP symbol. This could support a shift in the food supply toward foods with lower amounts of nutrients of concern. The implementation of the proposed FOP labelling would align with updates to the NFt and list of ingredients, as well as changes under consideration by the Canadian Food Inspection Agency as part of the Food Labelling Modernization project. This will minimize the number of times label changes are needed.
The proposed interpretive, nutrient-specific FOP approach could serve as an important part of a comprehensive suite of policies to help make healthier choices easier for Canadians and to help combat the contribution of excess sodium, sugars and saturated fat to diet-related chronic diseases.
Why a "high in" nutrient-specific approach?
- It attracts consumer attention
- It explicitly highlights nutrients of public health concern
- It supports and simplifies decision-making for consumers
- It helps consumers avoid less healthy food options
- It is transparent - consumers can link it to the information in the NFt
- It offers industry an incentive to manufacture foods lower in nutrients of concern
- It complements and reinforces other healthy eating initiatives
Consultation Question
1. Do you support Health Canada's proposed nutrient-specific "high-in" FOP labelling approach?
Please explain.
Element 2 - Thresholds for FOP labelling approach for foods high in sodium, sugars and saturated fat
2.1 Thresholds
The proposed FOP approach uses nutrient thresholds to determine whether or not a food would be required to carry the FOP symbol. The thresholds that would trigger the FOP symbol are provided in Table 1. A food composition database of approximately 350 indicator foods was used to assess different options, ranging from 10% of the DV to 25% of the DV. In selecting the best option, consideration was given to consistency with Canadian dietary guidance. An appropriate threshold should trigger the FOP label on foods that contain relatively high levels of one (or more) nutrient of concern, but not on foods that would be recommended as part of a healthy diet. Compatibility with existing nutrition labelling policies and regulations, such as the NFt and nutrient content claims was also an important consideration.
The proposed thresholds for a "high in" FOP label for prepackaged foods represent 15% of the DV for sodium, sugars and saturated fat. For sugars, the FOP label would apply to foods containing free sugars, including fruit juice. This means that unsweetened fruits, vegetables and dairy products would not be required to carry a FOP sugars label. The thresholds for prepackaged meals and combination dishes would represent 30% of the DV because they are generally consumed as a meal, or a major part of a meal, that contributes a higher proportion of calories and nutrients than individual foods.
The proposed thresholds are consistent with a recommended overall healthy eating pattern. Foods to choose more often (such as fruit and vegetables), would not be required to display a FOP symbol, whereas many foods to limit (such as soft drinks, cookies, ice cream and sausages) would likely display one (or more) symbols. They are also consistent with the level required for manufacturers to make "high in" claims for positive nutrients, such as calcium, and with Health Canada's messaging around the use of the % DV in the NFt, which says that 15% of the DV or more is "a lot" of that nutrient. For sodium, the proposed approach would also complement Health Canada's sodium reduction efforts by creating a stronger incentive for food industry to reduce sodium levels in their foods.
While the thresholds would apply to foods for most Canadians, Health Canada has recently proposed (in Canada Gazette, Part I, June 2015) to establish lower DVs for foods intended solely for children 1-3 years than those intended for individuals aged 4 years and older. This is to account for the differences in nutritional needs of children. By extension, the criteria that would trigger a FOP symbol on foods intended for this age group would also be lower. Proposed thresholds for these foods are provided in Table 2, which are aligned with 15% of the DV for young children.
| Nutrient | Prepackaged foods (15% of the DV) | Prepackaged mealsTable 1 Footnote a (30% of the DV) |
|---|---|---|
Table 1 Footnotes
|
||
| High in sodium | 345 milligrams or more per reference amountTable 1 Footnote b and per serving of stated sizeTable 1 Footnote c | 690 milligrams or more per reference amount and per serving of stated size |
| High in saturated fatTable 1 Footnote d | 3 grams or more per reference amount and per serving of stated size | 6 grams or more per reference amount and per serving of stated size |
| High in sugars | 15 grams or more total sugars per reference amount and per serving of stated size | 30 grams or more total sugars per reference amount and per serving of stated size |
| Nutrient | Prepackaged foodsTable 2 Footnote b |
|---|---|
|
|
| High in sodium | 225 milligrams per reference amount and per serving of stated size |
| High in saturated fat | 1.5 grams per reference amount and per serving of stated size |
| High in sugars | 7.5 (rounded to 8) grams per reference amount and per serving of stated size |
Consultation Questions
2.1a. Do you support Health Canada's proposed thresholds for triggering FOP labelling? Yes or no. Please explain.
2.1b. If your answer to 2.1a is "no", please suggest alternative thresholds along with a rationale and evidence to support your proposal.
2.2 Food with small reference amounts
Some foods that have high concentrations of one or more nutrients of concern would not trigger the FOP label based on the thresholds proposed, simply due to their small reference amounts. Examples include condiments, coffee cream, butter, margarine, some breakfast cereals, cookies and bars. While the reference amounts are based on amounts of food typically consumed in one sitting, consumers may not be aware that these foods can be important contributors of one or more nutrients of concern if consumed frequently. Health Canada is proposing that the threshold be based on 50 grams (or 50 millilitres for liquids) for products with reference amounts that are 50 grams or 50 millilitres or less. This proposal differs from the adjustment made for "low in" nutrient content claims for a few food categoriesFootnote iii; however, Health Canada considers this necessary to help consumers make more informed choices with respect to foods with smaller reference amounts and that should be consumed less often, or that consumers should limit, such as cookies, granola bars and chocolate bars.
Health Canada is also proposing that for oils and oil-based derivatives such as margarines and salad dressings with less than 30% of total fat as saturated and trans fat would be exempt from this adjustment. This will ensure that "healthy" oils, consistent with dietary guidance that suggests replacing saturated fat with unsaturated fat, will not require FOP labelling.
Consultation Questions
2.2a. Do you support Health Canada's proposed approach for foods with small reference amounts?
Yes or no. Please explain.
2.2b. If your answer to 2.2a is "no", please suggest an alternative approach along with a rationale and evidence to support your proposal.
2.3 Exemptions from FOP labelling
Under the current nutrition labelling regulations, several categories of prepackaged foods are usually exempted from displaying a NFt for technical or practical reasonsFootnote iv. Health Canada is proposing to apply the same exemptions to FOP labelling. Prepackaged foods exempt from displaying a NFt include: products with very small packages (e.g., one-bite confections, after dinner candies, bite size chocolate bars); small individual packages usually served in restaurants (e.g., packets of coffee creamers, crackers, jams, cheese, or margarine); food produced and prepackaged by retailers (e.g., meat cuts, sausages, deli meats, bakery products); and alcoholic beverages. Health Canada is also proposing to extend the exemptions to packages of sugar and salt (e.g. sugar, brown sugar, table salt, sea salt, and gourmet salts).
In addition, certain product categories in Divisions 24 and 25 of the Food and Drug Regulations have nutrition labelling requirements different from those of the NFt, including: infant formula (and foods containing infant formula), formulated liquid diets and foods represented for use in a very low energy diet. These are foods targeted to specific population groups with special nutritional needs and their nutritional composition is regulated.
Consultation Questions
2.3a. Do you support Health Canada's proposed approach to exempt foods from FOP labelling if the current Food and Drug Regulations do not require the food to carry a Nutrition Facts table?
Please explain.
2.3b. Do you support Health Canada's proposal to exempt packages of sugar and salt from FOP labelling?
Please explain.
Element 3 - Symbols for a "high in" FOP labelling approach
Health Canada is considering a variety of symbols that could be used to convey the FOP "high in" message. One strategy is to use shapes and symbols that are already meaningful to consumers (e.g., stop signs, yield signs, arrows, images, exclamation marks) combined with text. The symbol should be simple and intuitive for most Canadians - it should not require an explanation for consumers to understand. Health Canada is planning to conduct focus groups to assess how consumers understand and use a range of proposed FOP symbols. This will help determine which symbol best helps Canadians identify foods high in nutrients of concern. Examples of FOP symbols that Health Canada is considering are shown in Figure 1. Please note that the symbol would only include the nutrient(s) that exceeds the established threshold(s).
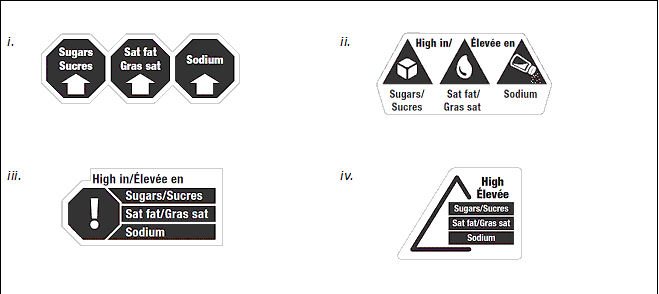
Text equivalent - Figure 1
The figure is comprised of 4 different options for front-of-package symbols, labelled "a-d". Symbol "a" shows three black octagons, side-by-side. Inside the first octagon, the words "Sugars" and "Sucres" in white font sit above a thick white arrow pointing up. Inside the second octagon, the words "Sat fat" and "Gras sat" in white font sit above a thick white arrow pointing up. Inside the third, the word "Sodium" in white font sits above a thick white arrow pointing up. The three black octagons together are outlined by a thin grey line.
Symbol "b" shows three black equilateral triangles, side-by-side, pointed up. The first triangle contains a white cube image. Under the first black triangle, the words "Sugars" and "Sucres" are shown in black font. The second triangle contains a white fat droplet image. Under the second black triangle, the words "Sat fat" and "Gras fat" are shown in black font. The third black triangle contains a white salt shaker, on an angle upside down with salt coming out of it. Under the third black triangle, the word "Sodium" is shown in black font. Between the first and second black triangles the words "High in/" are shown in black font. Between the second and third black triangles, the words "Élevée en" are shown in black font. The three black triangles along with the words between and under them are outlined by a thin grey line.
Symbol "c" shows one black octagon with a large white exclamation mark in the middle. Three black bars extend out from the side of the octagon. The top black bar contains the words "Sugars/Sucres" in white font, the middle black bar contains the words "Sat fat/Gras sat" in white font, and the bottom black bar contains the word "Sodium" in white font. The words "High in/Élevée en" in black font sit above the octagon and bars. The octagon, bars, and the words together are outlined by a thin grey line.
Symbol "d" shows an equilateral triangle, pointed up. There is a break in the right side of the triangle with three black bars cutting through the break. The top black bar contains the words "Sugars/Sucres" in white font, the middle black bar contains the words "Sat fat/Gras sat" in white font, and the bottom black bar contains the word "Sodium" in white font. The words "High/Élevée" in black font sit above the bars. The triangle, the bars, and the words together are outlined by a thin grey line.
Consultation Questions
3a. Do you support Health Canada's approach to choosing a FOP symbol for foods high in sodium, sugars and saturated fat? Please explain.
3b. Which symbol shown in Figure 1 would best help inform Canadians about foods high in sodium, sugars, and saturated fat? Please explain.
3c. If you do not agree that any of the symbols in Figure 1 would help inform Canadians, please propose an alternative symbol along with a rationale.
Element 4 - Updating nutrient content claims and other nutrition-related statements
Health Canada is proposing to update regulations related to a) nutrient content claims and b) other nutrition-related statements. Nutrient content claims and other label statements are regulated to enable consumers to make informed food choices in order to prevent injury to health and to ensure that criteria are applied and are consistent and not deceptive. These proposed updates are needed to bring some of the criteria for nutrient content claims (e.g., claims related to sugars) in line with the proposed FOP approach to nutrition labelling. In addition, Health Canada has assessed other outstanding nutrition-related statements and determined that the regulations should be amended. A summary of the proposed changes is provided in Table 3.
| Subject | Rationale for change | Proposed change and intended impact |
|---|---|---|
| Nutrient content claims | ||
i. No added sugars ii. Unsweetened |
Currently, foods that would be required to display the "high in sugars" FOP symbol may be eligible for the "no added sugars" and the "unsweetened" claims. |
Change conditions of use for the "no added sugars" and "unsweetened" claims by (a) aligning the meaning of "added sugars" with the new definition of "sugars-based ingredients" and (b) not permitting the claim on fruit juices that meet the threshold for "high in sugars". These changes would help ensure that sugars information on product labels is not contradictory. |
iii. Free of sugars |
The current conditions for using the "free of sugars" claim require that the food also be free of energy. Foods containing some types of high-intensity sweeteners cannot use this claim because these sweeteners contribute small amounts of calories. |
Change the condition of use of the claim from "energy free" to "low in energy". This change would offer industry an opportunity to use the "free of sugars" claim on products that contain high-intensity sweeteners. This in turn would provide consumers with more product choices as alternatives to foods high in sugars. |
iv. Low in sugars v. Lightly sweetened |
Currently, the Food and Drug Regulations do not permit industry to state that a food is low in sugars or lightly sweetened. |
Introduce a new "low in sugars" claim for foods that have no more than 5 grams sugars (a) per reference amount and per serving, (b) per 50 grams if the reference amount is small (i.e., 30 grams or 30 millilitres or less), or (c) per 100 grams if the food is a prepackaged meal. Introduce a new "lightly sweetened" claim for foods that have at least 50% less sugars-based ingredients than those added to the similar reference food that is not "low in sugars". These changes would encourage industry to formulate foods that are lower in sugars than those currently available in the Canadian food supply. |
| Nutrition-related statements | ||
vi. Quantitative statements outside the Nutrition Facts table |
Currently, foods that do not meet the conditions for the "free of sugars" or "free of trans fatty acids" claims are able to declare "0 g trans fat" and "0 g sugars", respectively. |
Prohibit the quantitative declaration of "0 g sugars" and "0 g trans fat" outside the NFt for foods that do not meet requirement for "free of sugars" or "free of trans fatty acids", respectively. This would help prevent misleading consumers when "0 g" declaration for sugars and trans fat is made on food labels because "0 g" may be misunderstood by consumers to be equivalent to "0" or "zero" as synonyms for "free". |
vii. Lean claim with respect to prepackaged meals |
Currently, many portion-controlled foods for weight maintenance are not eligible to use the term "lean" in English. |
Allow products to carry the "lean" claim without meeting the regulated definition of "prepackaged meal" if they are represented for use in a weight maintenance diet. This will provide more choices for consumers who are interested in this type of product. |
viii. Representation of the amount of alcohol in beverages |
Currently there are restrictions on the representation of the amount of alcohol in beverages containing less than 0.5% alcohol. |
Permit representation of the amount of alcohol in beverages containing 0-0.5% alcohol. This will allow products such as non alcoholic beers and wines and mocktails to be represented as "alcohol free". |
ix. Foods intended solely for young children |
There are several references to "food intended solely for children under two years of age" in the Food and Drug Regulations related to claims. This age range is not consistent with the age range in the nutrition labelling amendments proposed in 2015. |
Amend applicable sections of the regulations to "food intended solely for children under four years of age". |
Consultation Questions
4a. Do you support the changes proposed to update claims and other nutrition-related statements described in Table 3?
Please explain.
4b. If you do not support one or more of the proposed changes, please identify the subject of the proposed change (e.g., "i. no added sugar" claim) and explain why, along with a rationale and evidence to support your comments.
Element 5 - High-intensity sweetener labelling
5.1 Current high-intensity sweetener regulations
Like most prepackaged foods, foods containing high-intensity sweeteners (e.g., "artificial" sweeteners) are required to adhere to certain core labelling requirements, including the requirement that the label bear a list of all the ingredients found in that food.
In addition, foods containing any one of the following four sweeteners: aspartame, sucralose, acesulfame-potassium or neotame are subject to additional mandatory labelling requirements, including:
- A FOP statement on the principal display panel that the food contains or is sweetened with the sweetener, e.g., "contains aspartame". This information must be shown in the same type height as the net quantity.
- Where applicable, a statement on the principal display panel of any other sweeteners or sweetening agents used in conjunction with the sweetener, e.g., "sweetened with aspartame and xylitol" or "sweetened with sucralose, fructose and sugar".
- A quantitative declaration of the content of the sweetener in the food (in mg per serving size), grouped with the list of ingredients.
- In the case of aspartame, a statement to the effect that aspartame contains phenylalanine, grouped with the list of ingredients.
These additional labelling requirements were first introduced in 1981 with the approval of aspartame, the first high-intensity sweetener approved in Canada for use in non-dietetic foods. Aspartame contains the amino acid phenylalanine, the consumption of which must be avoided by individuals with phenylketonuria (PKU). At the time of its approval, aspartame was a new and unfamiliar dietary source of phenylalanine. For this reason, the requirement to declare phenylalanine was put in place as a means of alerting consumers with PKU to the fact that aspartame is a source of this amino acid. The remaining additional labelling requirements were put in place with the intent of giving consumers in general the information necessary to use this new high-intensity sweetener in an informed manner. For consistency, these additional labelling requirements (with the exception of the phenylalanine statement) were applied to the artificial sweeteners sucralose (approved in 1991), acesulfame-potassium (approved in 1994) and neotame (approved in 2007).
5.2 Issues with sweetener labelling regulations
As far back as the 1990s, certain stakeholders began to question the need for some of these additional labelling measures, noting that the mandatory list of ingredients already provides a mechanism for communicating the presence of these sweeteners to consumers.
Concerns over technical challenges for creating compliant food labels, particularly for foods sold in small or irregularly-shaped packages, have also been raised as have been the inconsistencies between the labelling requirements for these particular sweeteners and the requirements for other sweeteners and other ingredients of concern, such as priority allergens, for which neither principal display panel nor content declarations are required.
Stakeholders have also noted that Canada's requirements are, for the most part, inconsistent with comparable international regulators, such as the United States, United Kingdom, European Union and Australia/New Zealand, which do not require a declaration on the principal display panel nor a declaration of the content (in mg per serving) for foods containing these four sweeteners. However, like Canada, these jurisdictions do require a phenylalanine statement on the labels of foods containing aspartame.
5.3 Proposed changes to sweetener labelling regulations
Health Canada has undertaken a review of the current additional labelling requirements for the four high-intensity sweeteners in question and is proposing to eliminate the requirement for the principal display panel declaration and the quantitative declaration on foods containing sucralose, acesulfame-potassium and neotame. This will remove redundant labelling requirements and bring consistency to the labelling of sweeteners, as well as align our requirements with international regulators. These sweeteners will continue to be declared in the list of ingredients, alerting consumers to their presence in foods. Eliminating these requirements will also serve to alleviate some of the technical challenges faced by industry in labelling their foods and help to facilitate their compliance with other nutritional and legibility requirements for prepackaged food labels. With respect to foods containing aspartame, consideration is also being given to eliminating the principal display panel declaration and quantitative declaration, on the basis that the original intent of these particular requirements was to help the general population make an informed decision about foods containing the first high-intensity sweetener permitted for food additive use in Canada.
Given the risks for individuals with PKU, further consultation with professionals directly involved in the care of individuals with PKU as well as PKU advocacy groups is required in order to better understand how those with PKU use aspartame labelling information. It should be noted that Health Canada is not proposing to change the requirement to declare the presence of phenylalanine in foods containing aspartame. The feedback received will be taken into consideration by Health Canada as it considers eliminating the principle display panel and the quantitative declaration requirements for aspartame. These targeted consultations are expected to take place over the coming months.
Consultation Questions
5a. Do you support the changes proposed to eliminate the requirements for the principal display panel declaration and the quantitative declaration on foods containing sucralose, acesulfame-potassium and neotame? Yes or no. Please explain.
5b. If your answer to 5a. is "no", please provide your recommended approach along with a rationale and evidence to support your proposal.
5c. If you are someone who either has PKU, cares for someone with PKU, or provides dietetic advice to those with PKU, what are your views concerning the principal display panel and quantitative declaration labelling requirements for aspartame?
D. Conclusion
Health Canada values input on its proposed policies and regulations from interested Canadians and stakeholders. We welcome your comments on our proposal to implement FOP labelling requirements on prepackaged foods deemed high in sodium, sugars and saturated fat. We also welcome information and evidence from consumer-based research studies that could inform further development of the proposed approach. The information we receive will help inform decisions on the best approach to provide Canadians with an FOP nutrition labelling system that will help make healthy eating easier and have a meaningful impact on public health.
To submit your input, please visit the Footnote 37. If unpublished information is submitted, it will remain the property of the submitting organization or individual and its confidentiality will be safeguarded in so far as it is possible to do so within current regulations governing such issues. To safeguard privacy, you should ensure that any written comments you may provide are sufficiently general that you cannot be identified as the author and that individual identities are not disclosed.
E. Openness and Transparency
The Government of Canada is committed to Footnote 38. Formal written submissions in response to this consultation will be summarized in a report (e.g., Summary of Comments, What was Heard Report) that will be made publicly available. However, the individual submissions may be released upon request under the Access to Information Act.
All other correspondence and all meetings with stakeholders will be published monthly online in list format including the organization name, date, subject and purpose of Footnote 39.
For more information on Health Canada's new approach to openness and transparency please visit: http://healthycanadians.gc.ca/healthy-canada-vision-canada-en-sante/transparency-stakeholder-communications-transparence-intervenants-eng.php