Draft Guidance Document Electronic Canadian Drug Facts Table (eCDFT) Technical Standards
Consultation on the Draft Guidance Electronic Canadian Drug Facts Table (eCDFT) Technical Standards
Health Canada is pleased to announce that the Draft Guidance eCDFT Technical Standards, and the accompanying technical implementation package, are open for a 30-day comment period starting June 1, 2017 until June 30, 2017.
This guidance describes how Health Canada's Health Products and Food Branch (HPFB) interprets the flexibility allowing for the use of a Uniform Resource Locator (URL) on Category IV products, mouthwash and toothpaste as described in the Good Label and Package Practices Guide for Non-prescription Drugs and Natural Health Products (GLPPG).
These flexibilities are intended to provide consumers with access to on-line information presented in a standardised, mobile-friendly format that can be easily located, read and understood.
This guidance must be used in conjunction with the GLPPG, the Guidance Document: Drug Facts Table for Non-prescription Drugs and the relevant sections of other applicable Health Canada regulations, guidance documents, and policies for non-prescription drugs. The GLPPG outlines the formatting specifications to be applied by sponsors and the detailed implementation dates for the Facts Table requirement.
The PLL Regulations aim to improve the safe use of drugs by making drug labels and packages easier to read and understand. Following consultation with the non-prescription drug industry, a series of progressive design, layout and content flexibilities are available to sponsors to reduce the need to increase package sizes or introduce innovative labels to comply with the new facts table requirement.
Foreword
Guidance documents are meant to provide assistance to industry and health care professionals on how to comply with the policies and governing statutes and regulations. They also serve to provide review and compliance guidance to staff, thereby ensuring that mandates are implemented in a fair, consistent and effective manner.
Guidance documents are administrative instruments not having force of law and, as such, allow for flexibility in approach. Alternate approaches to the principles and practices described in this document may be acceptable provided they are supported by adequate scientific justification. Alternate approaches should be discussed in advance with the relevant program area to avoid the possible finding that applicable statutory or regulatory requirements have not been met.
As a corollary to the above, it is equally important to note that Health Canada reserves the right to request information or material, or define conditions not specifically described in this document, in order to allow the Department to adequately assess the safety, efficacy or quality of a therapeutic product. Health Canada is committed to ensuring that such requests are justifiable and that decisions are clearly documented.
This document should be read in conjunction with the accompanying notice and the relevant sections of other applicable guidance documents.
Table of Contents
- Introduction
- Technical Requirements For Electronic Canadian Drug Fact Table
- 2.1 URL (Uniform Resource Locator) conventions
- 2.1.1 Selecting or Deciding on an Appropriate URL Convention
- 2.1.2 Canadian DINholderwebsite.ca(com)/brandname, or brandname.ca(com)/cdft guideline
- 2.1.3 Canadian DINholderwebsite.ca(com)/search and DINholderwebsite.ca(com)/recherche, or brandname.ca(com)/search and brandname.ca(com)/recherche
- 2.1.4 CanadianDINholderwebsite.ca(com)/info, or brandname.ca(com)/info guideline
- 2.1.5 URL Choices for Private Label Products
- 2.1.6 URL placement
- 2.2 Use of two-dimensional (2D) barcodes
- 2.3 Technical approaches for accessing individual eCDFT via Search Functions
- 2.4 WET publication template
- 2.5 Using a third party
- 2.1 URL (Uniform Resource Locator) conventions
- 3. Important Considerations When Preparing Electronic Canada Drug Facts Table
- 4. Submission Process
1. Introduction
1.1 Objectives
The objective of the Electronic Canadian Drug Facts Table (eCDFT) Technical Standards Guidance document is to provide direction to sponsors, manufacturers and license holders (to be referred to as 'sponsors' within this guide) on the technical web publication standards including , naming conventions for "user-friendly" URLs, implementation flexibilities, and the use of other URL technologies.
This guidance document supports implementation of the Plain Language Labelling (PLL) Regulations for non-prescription drugs by outlining approaches intended to provide consumers with access to on-line information presented in a standardised, mobile-friendly format that can be easily located, read and understood.
1.2 Policy Statements
This document describes how Health Canada's Health Products and Food Branch (HPFB) interprets the flexibility allowing for the use of a URL on Category IV products, mouthwash and toothpaste as described in the Good Label and Package Practices Guide for Non-prescription Drugs and Natural Health Products (GLPPG)Footnote 1.
1.3 Scope and Application
Category IV products as well as mouthwash and toothpaste that limit information on the physical label (as described in the GLPPG) are required to include either a patient insert or a URL linking consumers to a complete Canadian Drug Facts Table (CDFT). The inclusion of a URL will be optional for products that include a full CDFT on the physical label or within an insert. This document outlines the parameters of how a standard Canadian Drug Facts Table will be provided to the consumer via a URL in an eCDFT format. The scope of this document is limited to technical standards associated with eCDFT.
This guidance must be used in conjunction with the GLPPG, the Guidance Document: Drug Facts Table for Non-prescription Drugs and the relevant sections of other applicable Health Canada regulations, guidance documents, and policies for non-prescription drugs. The GLPPG outlines the formatting specifications to be applied by sponsors and the detailed implementation dates for the Facts Table requirement.
The guidance document is intended to be used with the entire contents of the implementation package that includes templates and style sheets for posting eCDFT on sponsor websites.
1.4 Background
The PLL Regulations aim to improve the safe use of drugs by making drug labels and packages easier to read and understand. For non-prescription drugs and contact lens disinfectants, the PLL regulations will apply as of June 13, 2017 and impose new obligations on sponsors to include a standard Facts Table on the outer label. Following extensive consultation with the non-prescription drug industry, a series of progressive design, layout and content flexibilities are available to sponsors to reduce the need to increase package sizes or introduce innovative labels to comply with the new this facts table requirement. Linking consumers of Category IV products as well as mouthwash and toothpaste to point of use information via a URL is one such flexibility.
The principles to be respected in making the standard CDFT available to consumers via an electronic format are as follows:
- Readable
- Factual/non-promotional
- Accessible
- Mobile friendly
- Built on an Open standards
- Multilingual
- Consistency
2. Technical Requirements For Electronic Canadian Drug Fact Table
2.1 URL (Uniform Resource Locator) conventions
Uniform Resource Locators (URL), more commonly known as a web address, are a reference to a web page and a mechanism for accessing it.
An eCDFT URL is a URL printed on the physical label of a non-prescription drug, directing consumers to a location at which they can view a complete CDFT. A meaningful eCDFT URL needs to be:
- User-friendly: using words rather than a series of numbers or random alphanumeric characters.
- Not too long, and be easy for the consumer to enter in a browser.
- Bilingual.
2.1.1 Selecting or Deciding on an Appropriate URL Convention
Multiple URL conventions are provided for sponsors to address different market and logistical realities. For example, although the choice of a brandname in the URL may be appropriate for some products, it may not be a good fit for others due to length and complexity of a brand name containing multiple chemical names. There are six different approaches available for selecting an appropriate URL naming convention:
- Canadian DINholderwebsite.ca(com)/brandname
- Canadian DINholderwebsite.ca(com)/search and DINholderwebsite.ca(com)/recherche -
- Canadian DINholderwebsite.ca(com)/info
- brandname.ca(com)/cdft (for English) and brandname.ca(com)/tcim (for French)
- brandname.ca(com)/search and brandname.ca(com)/recherche
- brandname.ca(com)/ info
The scenarios presented in the table below should be considered in choosing an appropriate URL convention :
| Scenario | Suggested approach |
|---|---|
| Brand name length is greater than 10 characters | Use info or search URL |
| Brand name contains multiple ingredients | Use info or search URL |
| DINholderwebsite.ca(com)/search, or brandname.ca(com)/search already exists | Use info or cdft URL |
| there are multiple variants of a particular drug (such as for children, different drug strength, etc) | Use info URL |
2.1.2 Canadian DINholderwebsite.ca(com)/brandname, or brandname.ca(com)/cdft guideline
These are considered the default approaches. It uses the brand name or product name to take the consumer directly to the eCDFT. It should be used if the brand name is relatively small (less than 10 characters) and easy to enter.
An example of this URL could be: http://DINholdercompany.com/ lozenge
2.1.3 Canadian DINholderwebsite.ca(com)/search and DINholderwebsite.ca(com)/recherche, or brandname.ca(com)/search and brandname.ca(com)/recherche
Most consumers are familiar with search feature and how to use it. As such this approach is a good fit for longer or complicated brand names.
Examples of this URL could be: https://www.pharmacompany.com/search and https://www.pharmabrand.ca/recherche
It is important to note that the search results should have URLs that are language appropriate. They should either be bilingual or be in the language that the consumer intiaited the search. For example, if the consumer searches in French, the page containing the eCDFT for the searched drug should have URL that is either bilingual or in French.
2.1.4 CanadianDINholderwebsite.ca(com)/info, or brandname.ca(com)/info guideline
Ideal for brands with multiple line extensions. This approach using search functions provides multiple mechanisms for accessing an eCDFT.
Example of this URL could be: http://www.DINholderwebsite.ca/info
It is important to note that the results should have URLs that are language appropriate. They should either be bilingual or be in the language that the consumer intiaited the search . For example, if the consumer searches in French, the page containing the eCDFT for the searched drug should have an URL that is either bilingual or in French.
2.1.5 URL Choices for Private Label Products
For private labels, it is recommended to use the search approach i.e. Canadian DINholderwebsite.ca(com)/search or CanadianDINholderwebsite.ca(com)/recherche
2.1.6 URL placement
The CDFT format specificationsFootnote 2 do not prescribe a specific location for a user-friendly URL.
2.2 Use of two-dimensional (2D) barcodes
Category IV products as well as mouthwash and toothpaste that limit information on the physical label (as described in the GLPPG) and do not include an insert, are required to include a URL linking consumers to a complete CDFT. In addition to a URL, sponsors are permitted to include a two dimensional bar code. A 2D barcode cannot be a replacement for a URL.
Automatic identification and data capture (AIDC) refers to the method of automatically identifying objects, collecting data about them, and entering them directly into computer systems, without human involvement. Barcodes represent one such example of this technology.
2D barcodes (for example QR codes) store information in a 2D pattern of black and white squares. These 2D patterns are able to store more information per unit area than a 1-dimensional barcode. With the aid of a scanning device, such as a scanning application on a mobile phone, the matrix 2D barcode can be converted into a URL for a website. This offers consumers an alternative means of accessing a website other than manually typing the URL into a browser.
If a sponsor chooses to use a matrix 2D code, the following recommendations should be considered:
- The information contained within the automated identifier should not be considered a substitute for providing all required information directly on inner and outer labels.
- Legibility and readability of key information on the label should not be impeded by the presence of the matrix 2D barcode.
- Information embedded within the matrix 2D barcode should not include anything other than approved product information; be non-promotional in nature; and be focused on taking the consumer to the brand name specific page for the eCDFT.
- Information contained in the eCDFT accessible via the matrix 2D barcode must be compliant with the regulatory requirements.
- Sponsors should ensure that quality assurance processes are in place, including verification of the accuracy (e.g., the right barcode appearing on the right label) and readability of automated identifiers on health product labels.
- Sponsors should ensure patients are aware that by accessing 2D barcodes with their mobile phones their personal information may be exposed or collected (e.g., location, purchases, spending habits). Sponsors are therefore advised to consider their obligations under The Personal Information Protection and Electronic Documents Act (PIPEDA).
2.2.1 2D Barcode to brand name specific page or for accessing individual eCDFT
Sponsors should consider including a callout near the 2D barcode to make consumers aware of what the 2D barcode will do once scanned; For example, 'Scan here to go to [insert brand page url]'.
2.2.3 2D Barcode code placement
The CDFT format specifications do not prescribe a specific location for a 2D code. However, the following recommendations should be considered:
- The location of the 2D barcode should take into account the overall readability of the labeling.
- The inclusion of a 2D barcode should not compromise the readability of prescribed information and should be located in an area with minimal or no impact on readability.
- Inclusion of more than one 2D barcode is not recommended.
2.3 Technical approaches for accessing individual eCDFT via Search Functions
2.3.1 Search page
When a URL does not link directly to an individual eCDFT, search functions must be used to direct consumers in an intuitive and seamless manner to the relevant information. This process should not require more than three "clicks" and contain one or more mechanisms for navigating to a product-specific eCDFT, including text search, filter, and drop down menu.
The search page should use the following URL formats: DINholderwebsite.ca(com)/search and DINholderwebsite.ca(com)/recherche, or brandname.ca(com)/search and brandname.ca(com)/recherche. The other acceptable form is DINholderwebsite.ca(com)/info (the consumer can select "Search" or "Recherche" from the menu).
The content of the search page must be language appropriate. They should either be bilingual or be in the language that the consumer initiated the search or accessed the page. For example, if the consumer visits http://www.Canadian DINholderwebsite.ca(com)/recherche, the content should be in French.
The main search page will contain a simple search such as in Figure A.
Figure A: Search page
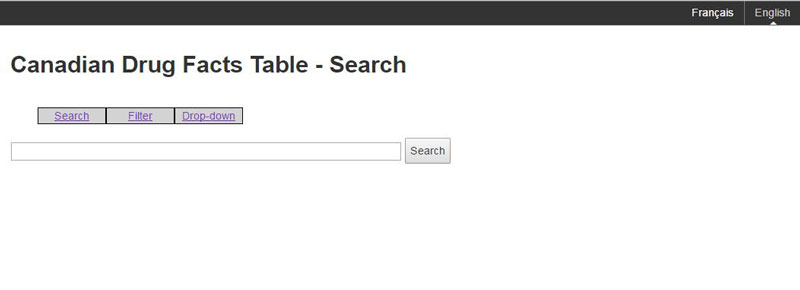
Consumers can search for a particular brand name and the search will display the closest matching eCDFT.
Figure B: The result page associated with a search
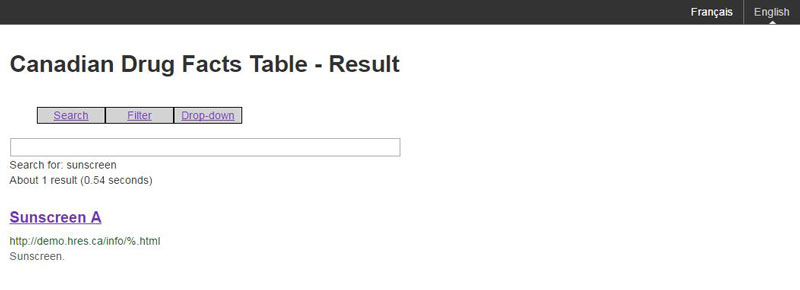
Any search should result in accessing an eCDFT. The search mechanism should only search for eCDFT, it should not search for other website content or company information.
2.3.2 Filter for brand name
The filter page is accessed via: DINholderwebsite.ca(com)/info or brandname.ca(com)/info. The consumer can select "Filter" or "Filtrer" from the menu.
The filter page will list all brand names.
Figure C: Filter mechanism
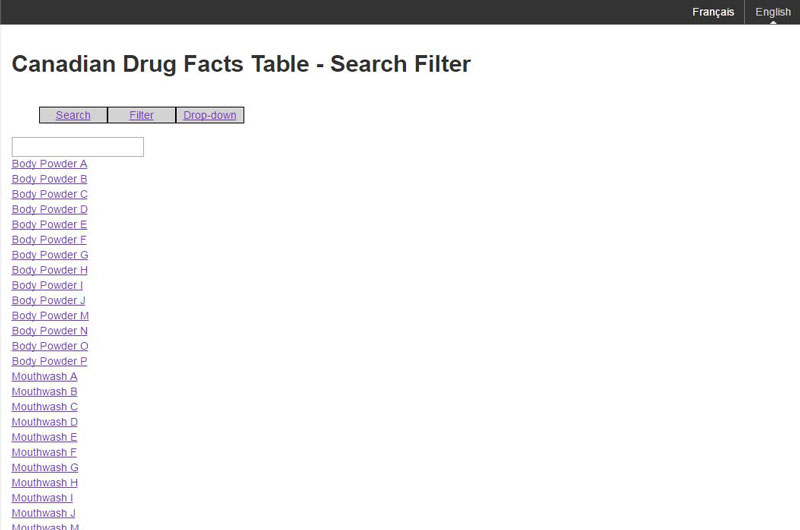
The text box will allow the brand names to be filtered on the value the consumer has entered.
Figure D: Filtering for a particular brand name
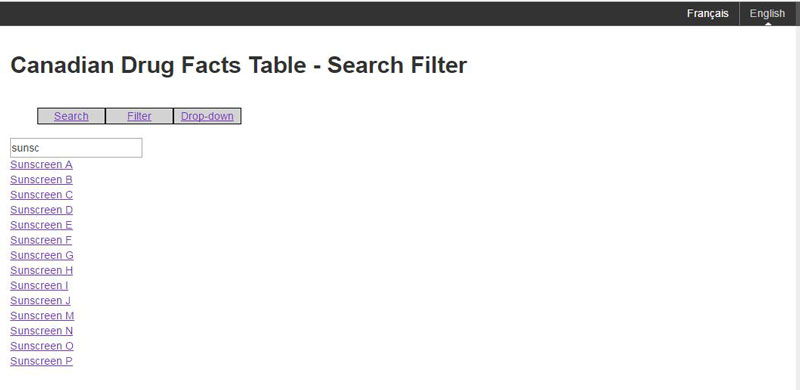
2.3.3 Drop down menu for brand name
The filter page is accessed via: DINholderwebsite.ca(com)/info or brandname.ca(com)/info. The consumer can select "Drop-down" or "Menu déroulant" from the menu.
The consumer can select the brand name from a drop down.
Figure E: Drop down mechanism.

Figure F: Selecting a particular brand name using the filter mechanism

By clicking on "Search" or "Recherche", the consumer will be taken to the eCDFT for the selected brand name.
2.4 WET publication template
The Web Experience Toolkit (WET)Footnote 3 is a front-end framework that is used to provide accessible, usable, multilingual and mobile-friendly websites.
Each sponsor will be provided access tothe followingWET templates:
- Search page
- Results page
- Filter page
- Drop-down page
- eCDFT content page
Per the GLPPG and the Guidance Document: Drug Facts Table for Non-prescription Drugs, the eCDFT content must use plain language and must not be combined with additional non-approved statements that are promotional in nature. If eCDFT are being used, the Health Canada approved template files must be used.
2.4.1 Using search page template
The search page has an input button labelled "Search" or "Recherche". The HTML associated with the button is:
<input value="Search" aria-label="Search" name="btnK" nClick="document.location.href='result.html';" type="submit">
The onClick should be redirected to the actual search mechanism which will search for the appropriate eCDFT. The technology to use for the search mechanisms is outside the scope of this document. It is left to the sponsor to determine what programming language stack to use (e.g., .Net, LAMP, Java) and which technological tools (e.g., ElasticSEarch, Solr, HubbleDoNet) if any, they prefer.
The results of the search should be presented in a results page such as Figure G.
Figure G: The result page associated with a search
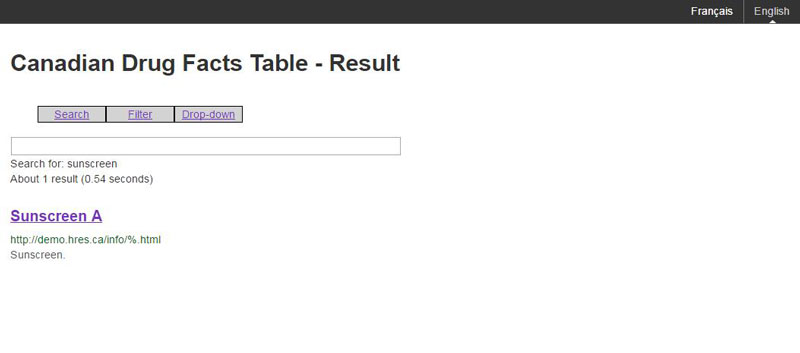
The result page should have the following results presented dynamically:
- search query
- number of results (and optional query search time)
- brand name that is a link to the eCDFT of the brand name
- URL to the eCDFT of the brand name
- purpose of the drug
An example of the dynamic result would be:
<p>Search for: Brand name
<br />About 1 result (0.54 seconds)
</p> <h3><a href="brandname.html">Brand Name</a></h3>
<div class="search_link">http://www.DINholderwebsite.ca/brandname.html</div>
<div class="search_result">Fever and pain relief medication.</div>
2.4.3 Using filter for brand name template
The filter page can be dynamically generated or can be manually updated as changes are required.
The following code needs to be added for each eCDFT:
<div class="wrapper"><span class="medicine"><a href="brandname.html">Brand name</span></a></div>
Where "brandname.html" is the filename of the eCDFT HTML file.
The added code needs to be inside of the gs_lc0 div, below the search input element.
An example of the content could be:
<div id="gs_lc0">
<input id="search" maxlength="2048" name="q" autocomplete="off" title="Search" value="" aria-label="Search" aria-haspopup="false" role="combobox" aria-autocomplete="both" dir="ltr" spellcheck="false" type="text">
<div class="wrapper"><span class="medicine"><a href="brandname.html">Brand name</span></a></div>
</div>
2.4.4 Using drop down for brand name template
The drop down page can be dynamically generated or can be manually updated as changes are required.
Each brand name should have an option element which specifies:
* filename of the eCDFT HTML file
* the brand name
An example of the content would be:
<select id="brand" class="entry-box"">
<option value"All">All</option>
<option value="brandname.html">Brandname</option>
</select>
Where "brandname.html" is the filename of the eCDFT HTML file and "Brandname" is the brand name.
2.4.1 Using eCDFT content template
The content template is used to create an eCDFT for a particular brand name. The French and English version of each brand must follow the requirements established for the standard CDFT.
The "Date modified" field should be updated each time there is a change.
The content pages can be dynamically generated or can be manually updated as changes are required.
Clicking on the language button on the upper right hand side must toggle the language between English and French
2.4.1.1 Name of drug in page header
The eCDFT must not be combined with additional non-approved statements . As such, it is permissible to list the drug name in the header of the eCDFT only.
The format of the header should be "Canadian Drug Facts Table - BRAND NAME" where "BRAND NAME" is the proprietary name (or product name, if there is no brand name).
2.4.5 Use of pictures/images/branding
Sponsors are permitted to include an image of the currently approved principal display (branding) panel on the eCDFTpage below the Drug Facts title i.e. Drug Facts Table –BRAND NAME), as long as the image is accurate, truthful and not misleading. No other promotional content in the form of logos, wording or images are permitted. An example of using the brand picture is in Figure H.
Figure H: Example of permissible brand picture

2.5 Using a third party
In some cases, Canadian DIN holders may choose to contract the services of a third party to host their eCDFTs. It is important to note that contracting a third party to host the eCDFT does not change the sponsors regulatory obligations. Sponsors are still accountable for ensuring their eCDFT is compliant with the regulations regardless of its location or the host.
The PLL Regulations also apply to the eCDFT content hosted by the third party. In particular, this also means third party's are subject to the rules on branding (e.g., the eCDFT is to be devoid of any branding or promotional material other than what is prescribed); and redirection (e.g., consumers should be brought directly to the eCDFT).
3. Important Considerations When Preparing Electronic Canada Drug Facts Table
3.1 Accessibility considerations
Accessibility of content for all users is an important consideration when creating web content. While much of the web template is already accessible, there are some content considerations when making accessible pages, following the level AA standard of the World Wide Web Consortium (W3C) Web Content Accessibility Guidelines (WCAG)Footnote 4
3.2 Non-text Content
From the WCAG principle 1.1.1:"All non-text content that is presented to the user has a text alternative that serves the equivalent purpose."
For the eCDFT, the product image, if included, must be descriptively labelled to help visually impaired consumers. This achieved by appropriately labelling the image using the alt tag in the template with an appropriate description. Examples of appropriate labelling include:
"Primary Label of < full drug name>"
"Image of <drug name /strength > in blue container"
"Image of <brand name> <drug name> <size>"
3.3 Sensory Characteristics
From the WCAG principle 1.3.3: "Instructions provided for understanding and operating content do not rely solely on sensory characteristics of components such as shape, size, visual location, orientation, or sound."
Any information conveyed on the web page should not rely solely on visual cues such as:
- "as shown below",
- "as per the next section "
- "as described in the red (text)"
Information should either appropriately describe the information or provide programmatic links to the content. Since the location of the content can vary, identification based on location is best avoided.
3.4 Language of Parts
From the WCAG principle 3.1.2: "The human language of each passage or phrase in the content can be programmatically determined except for proper names, technical terms, words of indeterminate language, and words or phrases that have become part of the vernacular of the immediately surrounding text."
The template, by default has a language tag set at the beginning of the web page (the language of the page, WCAG principle 3.1.1). If there is no change in language no further action is required. If however, another language is used anywhere in the template that differs from the base page language, it would need to be identified by the appropriate language identifier. For example, the French language selection on the English template is the following:
<a lang="fr" href="content-fr.html">Français</a>
Identifying the language allows screen readers to correctly pronounce the words in the intended language.
4. Submission Process
During the filing of a submission, there should be a placeholder for the URL. For the content of the URL, a "standard CDFT" must be submitted at the time of filing.
Activation of the URL is not required at the time of submission filing, but must be live at the time of marketing the product so that consumers can access it at the time of purchase. The URL can only be activated once the "standard CDFT" is approved by HC.
There is no requirement to make a submission for the review of the live-URL.