ARCHIVED - Priority Substances List Assessment Report for Road Salts
Environment Canada
Health Canada
2001
ISBN: 0-662-31018-7
Cat. No.: En40-215/63E
Canadian Environmental Protection Act 1999
Table of Contents
- List of Acronyms and Abbreviations
- Synopsis
- 1.0 Introduction
- 2.0 Summary of Background Information for the Assessment of Road Salts
- 2.1 Identity, properties, production and sources
- 2.2 Road Salt loadings
- 2.3 Snow disposal
- 2.4 Patrol Yards
- 2.5 Environmental fate and pathways
- 2.6 Chloride concentrations in surface waters across Canada
- 2.6.1 Mapping of chloride concentrations in watersheds across Canada
- 2.6.2 Environmental concentrations: Lakes and rivers
- 2.6.3 Environmental concentrations: Benthic sediments
- 2.6.4 Environmental concentrations: Wetlands
- 2.6.5 Environmental concentrations: Urban lakes and ponds
- 2.6.6 Chloride concentrations based on mass balance calculations
- 2.6.7 Summary
- 3.0 Assessment of Critical Information and Assessment of "Toxic" under CEPA 1999
- 3.1 CEPA 1999 64(a): Environment and CEPA 1999 64(b): Environment on which life depends
- 3.2 Groundwater
- 3.3 Aquatic ecosystems
- 3.3.1 Scoping and assessment approach
- 3.3.2 Laboratory studies
- 3.3.3 Assessment of toxicity to aquatic ecosystems
- 3.3.3.1 Quotient-based risk characterization
- 3.3.3.2 Tier 1 and Tier 2 assessments
- 3.3.3.3 Tier 3 assessments with field validation
- 3.3.3.3.1 Case 1: Road salt runoff and urban creeks, streams and small rivers in densely populated areas
- 3.3.3.3.2 Case 2: Road salt runoff and creeks, streams and rivers in less densely populated areas
- 3.3.3.3.3 Case 3: Road salt runoff and urban ponds and lakes
- 3.3.3.3.4 Case 4: Highway runoff and rural ponds and lakes
- 3.3.3.3.5 Case 5: Salt storage depots and aquatic ecosystems
- 3.3.3.3.6 Case 6: Snow dumps
- 3.3.3.4 Other effects
- 3.3.3.5 Conclusions for aquatic ecosystems
- 3.4 Soils
- 3.5 Terrestrial vegetation
- 3.5.1 Conservative environmental assessment
- 3.5.2 Detailed environmental assessment - threshold-based analysis
- 3.5.3 Detailed environmental assessment - reference site analysis
- 3.5.3.1 Spread of halophytic plant species
- 3.5.3.2 Community changes in a bog impacted by road salts
- 3.5.3.3 Effects of salt spray from a four-lane highway on peach and plum orchards in Grimsby, Ontario
- 3.5.3.4 Effects of salt spray from a four-lane highway on peach and plum orchards near St. Catharines, Ontario
- 3.5.3.5 Effects of salt spray from a two-lane highway on blueberries in western Nova Scotia
- 3.5.3.6 Concentrations in soil and woody plants adjacent to highways in interior British Columbia
- 3.5.3.7 Concentrations in soil and woody foliage adjacent to a two-lane highway in British Columbia
- 3.5.3.8 Concentrations in woody foliage directly adjacent to a two-lane highway in Connecticut
- 3.5.3.9 Concentrations in soil and woody and herbaceous foliage along roadsides in Massachusetts
- 3.5.3.10 Concentrations in soil and woody plants downslope from a four-lane highway in British Columbia
- 3.5.3.11 Concentrations in soil and woody foliage adjacent to a patrol yard
- 3.5.4 Changes in application rates and current injury in Canada
- 3.5.5 Estimate of area of potential injury
- 3.5.6 Risk assessment summary and conclusions
- 3.6 Terrestrial wildlife
- 3.7 Ferrocyanides
- 3.8 Overall conclusions
- 3.9 Considerations for follow-up (further action)
- 4.0 References
Appendices
- Appendix A Search Strategies Employed for Identification of Relevant Data
- Appendix B Supporting Documents Prepared for fhe PSL Assessment of Road Salts
List of Tables
- Table 1 Physical and chemical properties of road salts
- Table 2 Total loading of sodium chloride road salt, winter 1997-98 (from Morin and Perchanok, 2000)
- Table 3 Estimated quantities of calcium chloride used on roadways in a typical year (from Morin and Perchanok, 2000)
- Table 4 Total loadings of chloride (from Morin and Perchanok, 2000)
- Table 5 Total volume of waste snow and quantity of salt used in certain Canadian cities, winter 1997-98 (from Delisle and Dériger, 2000)
- Table 6 Snow disposal methods used in certain Canadian cities, winter 1997-98 (from Delisle and Dériger, 2000)
- Table 7 Number of provincial patrol yards and sodium chloride and abrasive use, by province (from Snodgrass and Morin, 2000)
- Table 8Estimate of magnitude of salt loss at patrol yards (from Snodgrass and Morin, 2000)
- Table 9 Chloride concentrations in various streams in the Toronto Remedial Action Plan watershed for 1990-96 (from Toronto and Region Conservation Authority, 1998)
- Table 10 Toxicity responses of organisms to sodium chloride for exposures less than 1 day (from Evans and Frick, 2001)
- Table 11 Toxicity responses of organisms to sodium chloride for exposures of 1 day (from Evans and Frick, 2001)
- Table 12Four-day LC50s of various taxa exposed to sodium chloride (from Evans and Frick, 2001)
- Table 13 Seven- to 10-day LC50s and EC50s of various taxa exposed to sodium chloride (from Evans and Frick, 2001)
- Table 14 Summary of Tier 2 calculations (from Evans and Frick, 2001)
- Table 15 pH, total phosphorus and chloride optima for selected diatom species in the northeastern United States (from Dixit et al., 1999)
- Table 16 Sodium chloride tolerance of selected plant species in the salt-impacted mat zone of Pinhook Bog, Indiana (from Wilcox, 1982)
- Table 17 Predicted cumulative percentage of species affected by chronic exposures to chloride (from Evans and Frick, 2001)
- Table 18 Modelled electrical conductivity values and chloride concentrations for Corbett Brook, downstream from Fredericton's salt depot
- Table 19 Species list of roadside trees and shrubs rated for their resistance to airborne highway salt spray (from Lumis et al., 1983)
- Table 20 Species list of native forest tree species rated for their resistance to highway salt spray
- Table 21 Range of threshold values estimated for soil and water for various forms of plants (from Cain et al., 2001)
- Table 22 Range of threshold values estimated for herbaceous tissue concentrations following soil or water solution exposure (from Cain et al., 2001)
- Table 23 Range of threshold values estimated for woody tissue concentrations following aerial exposure (from Cain et al., 2001)
- Table 24 Calculation of the number of particles of salt that need to be ingested in order to reach CTVs assuming a model 28-g house sparrow consuming particles at the upper end of its known preference range
- Table 25 Calculation of the number of particles of salt that need to be ingested in order to reach CTVs assuming a model 28-g house sparrow consuming particles of average size
- Table 26 Quotient calculation results for aquatic biota - Tier 1 (from Letts, 2000a)
- Table 27 Quotient calculation results for terrestrial biota - Tier 1 (from Letts, 2000a)
- Table 28 Quotient calculation results for aquatic biota - Tier 2 (from Letts, 2000a)
- Table 29 Quotient calculation results for terrestrial biota - Tier 2 (from Letts, 2000a)
List of Figures
- Figure 1 Provincial road network area fraction (from Morin and Perchanok, 2000)
- Figure 2 Recommended provincial application rates for sodium chloride road salts, 1998 (from Morin and Perchanok, 2000)
- Figure 3 Average recommended municipal application rates for sodium chloride road salts, 1998 (from Morin and Perchanok, 2000)
- Figure 4 Total amount of sodium chloride used, by maintenance district (from Morin and Perchanok, 2000)
- Figure 5 Historical salt use by provincial agencies (based on surveys conducted by the Salt Institute, 1964-1983, and Morin and Perchanok, 2000)
- Figure 6 Average daily concentration of leachate from salt-treated sand pile, 1975-76 (from NB DOE and DOT, 1978)
- Figure 7 Chloride concentrations in Heffley Creek municipal wells (from AGRA, 1999)
- Figure 8 Road salts transport pathway
- Figure 9 Observed chloride concentrations in Canadian watersheds (from Mayer et al., 1999)
- Figure 10 Estimated road salt chloride concentrations by watershed, calculated from average annual road salt loadings and average annual runoff (from Mayer et al., 1999)
- Figure 11 Chloride concentrations in runoff for provincial roads in Canada
- Figure 12 Estimated chloride concentrations in groundwater for various chloride application rates and a groundwater recharge rate of 10 cm/year (from Johnston et al., 2000)
- Figure 13 Estimated chloride concentrations in groundwater for various chloride application rates and a groundwater recharge rate of 20 cm/year (from Johnston et al., 2000)
- Figure 14 Estimated chloride concentrations in groundwater for various chloride application rates and a groundwater recharge rate of 40 cm/year (from Johnston et al., 2000)
- Figure 15 Chloride concentrations in groundwater from municipal production wells in southern Ontario (from Johnston et al., 2000)
- Figure 16 Species diversity across a salinity and chloride gradient (from Wetzel, 1983)
- Figure 17 Experimental acute toxicity and predicted chronic toxicity for aquatic taxa (from Evans and Frick, 2001)
- Figure 18 Representative short-term chloride concentrations in the Canadian aquatic environment associated with contamination by road salts and concentrations causing adverse biological effects following brief exposures
- Figure 19 Representative long-term chloride concentrations in the Canadian aquatic environment associated with contamination by road salts and concentrations causing adverse biological effects following prolonged exposures
- Figure 20 Areas with low to medium to high road salt application hazards, based on product of soil clay content with exchangeable sodium ratio (from Morin et al., 2000)
- Figure 21 Box plot showing 10th, 25th, 75th, and 90th percentile plus individual data above and below the 10th and 90th percentile for ditch water electrical conductivity and chloride concentrations along two highways near Fredericton's salt depot, outside the depot's catchment area for representative days in June, July, October 1999, and January 2000. "Well-drained", "cross-flow", and "stagnant" refer to three conditions: ridges from which water drains, stream/road crossings, and ditch depressions with no visible outflow, respectively (from Arp, 2001)
- Figure 22 Estimates for total dissolved solids (TDS) and electrical conductivity (EC) of average soil solution and surface waters, by level 2 watersheds and by municipal boundary, Ontario and Quebec (from Morin et al., 2000)
- Figure 23 Exchangeable sodium percentage (ESP) along roadsides, by provincial road maintenance district (from Morin et al., 2000)
- Figure 24 Salt tolerance and crop yield relative to soil salinity (electrical conductivity, EC) and the estimated percentage of roadsides with corresponding levels of electrical conductivity (from Bresler et al., 1982)
List of Acronyms and Abbreviations
CAS
Chemical Abstracts Service
CEPA
Canadian Environmental Protection Act
CEPA 1999
Canadian Environmental Protection Act, 1999
CTV
Critical Toxicity Value
EC
electrical conductivity
EC50
median effective concentration
EEV
Estimated Exposure Value
ENEV
Estimated No-Effects Value
ESA
environmental site assessment
kg-bw
kilogram body weight
LC50
median lethal concentration
LD50
median lethal dose
LOEC
Lowest-Observed-Effect Concentration
LOEL
Lowest-Observed-Effect Level
NOEC
No-Observed-Effect Concentration
NOEL
No-Observed-Effect Level
PSL
Priority Substances List
Synopsis
Road salts are used as de-icing and anti-icing chemicals for winter road maintenance, with some use as summer dust suppressants. Inorganic chloride salts considered in this assessment include sodium chloride, calcium chloride, potassium chloride and magnesium chloride. In the environment, these compounds dissociate into the chloride anion and the corresponding cation. In addition, ferrocyanide salts, which are added as anti-caking agents to some road salt formulations, were assessed. It is estimated that approximately 4.75 million tonnes of sodium chloride were used as road salts in the winter of 1997-98 and that 110 000 tonnes of calcium chloride are used on roadways in a typical year. Very small amounts of other salts are used. Based on these estimates, about 4.9 million tonnes of road salts can be released to the environment in Canada every year, accounting for about 3.0 million tonnes of chloride. The highest annual loadings of road salts on a road-length basis are in Ontario and Quebec, with intermediate loadings in the Atlantic provinces and lowest loadings in the western provinces.
Road salts enter the Canadian environment through their storage and use and through disposal of snow cleared from roadways. Road salts enter surface water, soil and groundwater after snowmelt and are dispersed through the air by splashing and spray from vehicles and as windborne powder. Chloride ions are conservative, moving with water without being retarded or lost. Accordingly, all chloride ions that enter the soil and groundwater can ultimately be expected to reach surface water; it may take from a few years to several decades or more for steady-state groundwater concentrations to be reached. Because of the widespread dispersal of road salts through the environment, environmental concerns can be associated with most environmental compartments.
In water, natural background concentrations of chloride are generally no more than a few milligrams per litre, with some local or regional instances of higher natural salinity, notably in some areas of the Prairies and British Columbia. High concentrations of chloride related to the use of road salts on roadways or releases from patrol yards or snow dumps have been measured. For example, concentrations of chloride over 18 000 mg/L were observed in runoff from roadways. Chloride concentrations up to 82 000 mg/L were also observed in runoff from uncovered blended abrasive/salt piles in a patrol yard. Chloride concentrations in snow cleared from city streets can be quite variable. For example, the average chloride concentrations in snow cleared from streets in Montréal were 3000 mg/L for secondary streets and 5000 mg/L for primary streets. Waters from roadways, patrol yards or snow dumps can be diluted to various degrees when entering the environment. In the environment, resulting chloride concentrations have been measured as high as 2800 mg/L in groundwater in areas adjacent to storage yards, 4000 mg/L in ponds and wetlands, 4300 mg/L in watercourses, 2000-5000 mg/L in urban impoundment lakes and 150-300 mg/L in rural lakes. While highest concentrations are usually associated with winter or spring thaws, high concentrations can also be measured in the summer, as a result of the travel time of the ions to surface waters and the reduced water flows in the summer. Water bodies most subject to the impacts of road salts are small ponds and watercourses draining large urbanized areas, as well as streams, wetlands or lakes draining major roadways. Field measurements have shown that roadway applications in rural areas can result in increased chloride concentrations in lakes located a few hundred metres from roadways.
The potential for impacts on regional groundwater systems was evaluated using a mass balance technique that provides an indication of potential chloride concentrations downgradient from saltable road networks. The mass balance modelling and field measurements indicated that regional-scale groundwater chloride concentrations greater than 250 mg/L will likely result under high-density road networks subject to annual loadings above 20 tonnes sodium chloride per two-lane-kilometre. Considering data on road salt loadings, urban areas in southern Ontario, southern Quebec and the Atlantic provinces face the greatest risk of regional groundwater impacts. Groundwater will eventually well up into the surface water or emerge as seeps and springs. Research has shown that 10-60% of the salt applied enters shallow subsurface waters and accumulates until steady-state concentrations are attained. Elevated concentrations of chlorides have been detected in groundwater springs emerging to the surface.
Acute toxic effects of chloride on aquatic organisms are usually observed at relatively elevated concentrations. For example, the 4-day median lethal concentration (LC50) for the cladoceran Ceriodaphnia dubia is 1400 mg/L. Exposure to such concentrations may occur in small streams located in heavily populated urban areas with dense road networks and elevated road salt loadings, in ponds and wetlands adjacent to roadways, near poorly managed salt storage depots and at certain snow disposal sites.
Chronic toxicity occurs at lower concentrations. Toxic effects on aquatic biota are associated with exposures to chloride concentrations as low as 870, 990 and 1070 mg/L for median lethal effects (fathead minnow embryos, rainbow trout eggs/embryos and daphnids, respectively). The No-Observed-Effect Concentration (NOEC) for the 33-day early life stage test for survival of fathead minnow was 252 mg chloride/L. Furthermore, it is estimated that 5% of aquatic species would be affected (median lethal concentration) at chloride concentrations of about 210 mg/L, and 10% of species would be affected at chloride concentrations of about 240 mg/L. Changes in populations or community structure can occur at lower concentrations. Because of differences in the optimal chloride concentrations for the growth and reproduction of different species of algae, shifts in populations in lakes were associated with concentrations of 12-235 mg/L. Increased salt concentrations in lakes can lead to stratification, which retards or prevents the seasonal mixing of waters, thereby affecting the distribution of oxygen and nutrients. Chloride concentrations between 100 and 1000 mg/L or more have been observed in a variety of urban watercourses and lakes. For example, maximum chloride concentrations in water samples from four Toronto-area creeks ranged from 1390 to 4310 mg/L. Chloride concentrations greater than about 230 mg/L, corresponding to those having chronic effects on sensitive organisms, have been reported from these four watercourses through much of the year. In areas of heavy use of road salts, especially southern Ontario, Quebec and the Maritimes, chloride concentrations in groundwater and surface water are frequently at levels likely to affect biota, as demonstrated by laboratory and field studies.
Application of road salts can also result in deleterious effects on the physical and chemical properties of soils, especially in areas that suffer from poor salt, soil and vegetation management. Effects are associated with areas adjacent to salt depots and roadsides, especially in poorly drained depressions. Effects include impacts on soil structure, soil dispersion, soil permeability, soil swelling and crusting, soil electrical conductivity and soil osmotic potential. These can have, in turn, abiotic and biotic impacts on the local environment. The primary abiotic impact is the loss of soil stability during drying and wetting cycles and during periods of high surface runoff and wind. Biological impacts relate primarily to osmotic stress on soil macro- and microflora and macro- and microfauna, as well as salt-induced mobilization of macro- and micronutrients that affect flora and fauna.
A number of field studies have documented damage to vegetation and shifts in plant community structure in areas impacted by road salt runoff and aerial dispersion. Halophytic species, such as cattails and common reed-grass, readily invade areas impacted by salt, leading to changes in occurrence and diversity of salt-sensitive species. Elevated soil levels of sodium and chloride or aerial exposures to sodium and chloride result in reductions in flowering and fruiting of sensitive plant species; foliar, shoot and root injury; growth reductions; and reductions in seedling establishment. Sensitive terrestrial plants may be affected by soil concentrations greater than about 68 mg sodium/kg and 215 mg chloride/kg. Areas with such soil concentrations extend linearly along roads and highways or other areas where road salts are applied for de-icing or dust control. The impact of aerial dispersion extends up to 200 m from the edge of multi-lane highways and 35 m from two-lane highways where de-icing salts are used. Salt injury to vegetation also occurs along watercourses that drain roadways and salt handling facilities.
Behavioural and toxicological impacts have been associated with exposure of mammalian and avian wildlife to road salts. Ingestion of road salts increases the vulnerability of birds to car strikes. Furthermore, intake calculations suggest that road salts may poison some birds, especially when water is not freely available during severe winters. Road salts may also affect wildlife habitat, with reduction in plant cover or shifts in communities that could affect wildlife dependent on these plants for food or shelter. Available data suggest that the severity of road kills of federally protected migratory bird species (e.g., cardueline finches) and the contribution of road salts to this mortality have been underestimated.
Ferrocyanides are very persistent but are of low toxicity. However, in solution and in the presence of light, they can dissociate to form cyanide. In turn, the cyanide ion may volatilize and dissipate fairly quickly. The ultimate effects of ferrocyanides therefore depend on the complex balance between photolysis and volatilization, which in turn depends on environmental factors. Modelling studies undertaken in support of this assessment indicate that there is a potential for certain aquatic organisms to be adversely affected by cyanide in areas of high use of road salts.
Based on the available data, it is considered that road salts that contain inorganic chloride salts with or without ferrocyanide salts are entering the environment in a quantity or concentration or under conditions that have or may have an immediate or long-term harmful effect on the environment or its biological diversity or that constitute or may constitute a danger to the environment on which life depends. Therefore, it is concluded that road salts that contain inorganic chloride salts with or without ferrocyanide salts are "toxic" as defined in Section 64 of the Canadian Environmental Protection Act, 1999 (CEPA 1999).
The use of de-icing agents is an important component of strategies to keep roadways open and safe during the winter and minimize traffic crashes, injuries and mortality under icy and snowy conditions. These benefits were recognized by the Ministers' Expert Advisory Panel on the Second Priority Substances List, even as they recommended that this assessment of potential impacts on the environment be conducted. Any measures developed as a result of this assessment must never compromise human safety; selection of options must be based on optimization of winter road maintenance practices so as not to jeopardize road safety, while minimizing the potential for harm to the environment. Any action taken to reduce impacts on the environment is also likely to reduce potential for contamination of groundwater-based drinking water supplies, which is clearly desirable.
Future management should focus on key sources in areas where the assessment has indicated concerns. These relate to the following:
- Patrol yards: Key concerns relate to the contamination of groundwater at patrol yards and the discharge to surface water. In addition, overland flow of salty snowmelt waters can result in direct impacts to surface water and near-field vegetation. Based on surveys and reviews, salt losses from patrol yards are associated with loss at storage piles (which include salt piles as well as piles of sand and gravel to which salts have been added) and during the handling of salts, relating to both storage and loading and unloading of trucks. The discharge of patrol yard washwater is also a potential source of release of salts. Measures and practices should therefore be considered to ensure storage of salts and abrasives to reduce losses through weathering, to reduce losses during transfers and to minimize releases of stormwater and equipment washwater.
- Roadway application: Key environmental concerns have been associated with areas of high salt use and high road density. Regions of southern Ontario and Quebec and the Atlantic provinces have the highest rate of salt use on an area basis and as such have the highest potential for contamination of soils, groundwater and surface water by road salts as a result of roadway applications. In addition, urban areas in other parts of the country where large amounts of salts are applied are of potential concern, especially for streams and aquifers that are wholly surrounded by urban areas. In rural areas, surface waters receiving drainage from roadways may also be susceptible to contamination. Areas where splash or spray from salted roads can be transported through air to sensitive vegetation are a potential concern. Wetlands that directly adjoin roadway ditches and that receive runoff in the form of salty snowmelt waters are also potential management concerns. Therefore, measures should be considered to reduce the overall use of chloride salts in such areas. The selection of alternative products or of appropriate practices or technology to reduce salt use should be considered while ensuring maintenance of roadway safety.
- Snow disposal: Key environmental concerns relate to eventual loss of meltwater into surface water and into soil and groundwater at snow disposal sites. Measures to minimize percolation of salty snowmelt waters into soil and groundwater at snow disposal sites should be considered. Practices to direct the release of salty snowmelt waters into surface waters that have minimal environmental sensitivity or into storm sewers could be considered. Measures should also be considered to ensure sufficient dilution before release.
- Ferrocyanides: This assessment indicates that there is a possible adverse exposure for the more sensitive aquatic vertebrates in areas of very high use of road salts. Risks could be reduced by reducing total salt use or reducing content of ferrocyanides in road salt formulations. To reduce the possibility of exposure, producers of road salts could consider reducing the addition rate of ferrocyanide to road salts. Any reduction in total salt use would be expected to result in an equivalent reduction in release of ferrocyanides.
1.0 Introduction
The Canadian Environmental Protection Act, 1999 (CEPA 1999) requires the federal Ministers of the Environment and of Health to prepare and publish a Priority Substances List (PSL) that identifies substances, including chemicals, groups of chemicals, effluents and wastes, that may be harmful to the environment or constitute a danger to human health. The Act also requires both Ministers to assess these substances and determine whether they are "toxic" or capable of becoming "toxic" as defined in Section 64 of the Act, which states:
...a substance is toxic if it is entering or may enter the environment in a quantity or concentration or under conditions that
- have or may have an immediate or long-term harmful effect on the environment or its biological diversity;
- constitute or may constitute a danger to the environment on which life depends; or
- constitute or may constitute a danger in Canada to human life or health.
Substances that are assessed as "toxic" as defined in Section 64 may be placed on the List of Toxic Substances in Schedule I of the Act and considered for possible risk management measures, such as regulations, guidelines, pollution prevention plans or codes of practice to control any aspect of their life cycle, from the research and development stage through manufacture, use, storage, transport and ultimate disposal.
Based on initial screening of readily accessible information, the rationale for assessing road salts provided by the Ministers' Expert Advisory Panel on the Second Priority Substances List (Ministers' Expert Advisory Panel, 1995) was as follows:
The Panel recognized the benefits associated with the use of road salts. However, these substances have negative effects on the environment. Large volumes are released through road salting, particularly in Ontario, Quebec and the Atlantic provinces. There is evidence of adverse local environmental effects to groundwater and to plant and animal life following exposure. Algae and benthic fauna have been shown to be particularly sensitive to changes in chloride ion concentrations, resulting in a reduction of fish populations. The Panel recognizes that there has been considerable progress in upgrading storage facilities. However, given the widespread exposure to these substances, and their release in large volumes into the Canadian environment, the Panel believes that an assessment is needed to determine their ecological effects.
The basis for inclusion of road salts by the Ministers' Expert Advisory Panel on the Second Priority Substances List was limited to environmental effects and did not identify concerns with respect to human health. Humans are exposed to road salts principally through the contamination of roadside well waters, where chloride and sodium levels can be increased and taste adversely affected. These elements are not considered toxic; in fact, the Canadian drinking water guidelines for chloride and sodium are based on taste, which is affected at levels well below those that might be of concern for toxicity (Health Canada, 1996). Other substances contained in road salts, including ferrocyanide compounds and certain metals, are present only at trace levels.
With respect to the potential for health effects of sodium chloride road salts, an extensive literature search failed to identify any studies that were adequate to serve as the basis for a health risk assessment. In one study dermal exposure to concentrated solutions of road salt caused dermal irritation, but no sensitization (Cushman et al., 1991). There is a single correlational study in which a statistical association between road salt use and mortality from a number of types of cancer in the United States was identified (Foster, 1993). However, in this study, road salt use would have varied in parallel with a number of other factors; for example, larger amounts of salt are used in urban centres, where there is also more exposure to air pollution. There would also have been differences among states in smoking patterns, diet and a host of other factors. The design of this exploratory study does not permit adjustment for these other factors, many of which are more plausible causes of cancer than is road salt.
In view of the focus of this assessment on environmental effects defined by the Ministers' Expert Advisory Panel and the extremely limited and inadequate data available related to the potential impacts of road salts on human health, this assessment solely addresses effects on the environment (i.e., the determination of whether road salts are "toxic" under Paragraphs 64(a) and 64(b) of CEPA 1999).
Descriptions of the approaches to assessment of the effects of Priority Substances on the environment are available in a published companion document. The document entitled "Environmental Assessments of Priority Substances under the Canadian Environmental Protection Act. Guidance Manual Version 1.0 -March 1997" (Environment Canada, 1997a) provides guidance for conducting environmental assessments of Priority Substances in Canada. This document may be purchased from:
- Environmental Protection Publications
Environmental Technology Advancement Directorate
Environment Canada
Ottawa, Ontario
K1A 0H3
It is also available on the Existing Substances Branch web site at www.ec.gc.ca/cceb1/eng/psap.htm under the heading "Technical Guidance Manual." It should be noted that the approach outlined therein has evolved to reflect changes in the Canadian Environmental Protection Act and to incorporate recent developments in risk assessment methodology, which will be addressed in future releases of the guidance manual for the assessment of effects of Priority Substances on the environment.
The search strategies employed in the identification of data relevant to the assessment of potential effects on the environment (prior to May 2001) are presented in Appendix A. Review articles were consulted where appropriate. However, all original studies that form the basis for determining whether road salts are "toxic" under CEPA 1999 have been critically evaluated.
Preparation of the environmental components of the assessment was led by B. Elliott under the direction of R. Chénier. Extensive supporting documentation (Appendix B) related to the environmental assessment of road salts was prepared and reviewed by the Environmental Resource Group established in June 1997 by Environment Canada to support the environmental assessment:
- P. Arp, University of New Brunswick
- M. Barre, Environment Canada (until December 1999)
- Y. Bourassa, Environment Canada (since December 1999)
- L. Brownlee, Environment Canada (until April 2000)
- B. Butler, University of Waterloo
- N. Cain, Cain Vegetation Inc.
- R. Chénier, Environment Canada
- C. Delisle, École Polytechnique de Montréal
- M. Eggleton, Environment Canada (until February 2000)
- B. Elliott, Environment Canada
- M. Evans, Environment Canada
- K. Hansen, Ontario Ministry of the Environment
- J. Haskill, Environment Canada
- K. Howard, University of Toronto (until January 2000)
- A. Letts, Morton International Inc.
- B. Mander, Environment Canada
- T. Mayer, Environment Canada
- P. Mineau, Environment Canada (since April 2000)
- D. Morin, Environment Canada
- M. Perchanok, Ontario Ministry of Transportation
- R. Smith, Transportation Association of Canada
- W. Snodgrass, Snodgrass Consultants
- K. Taylor, Environment Canada
- M. Weese, Ontario Ministry of Transportation (until January 1999)
Environmental Resource Group members also reviewed the Assessment Report prepared by Environment Canada. Conclusions and interpretations in this report are those of Environment Canada and do not necessarily represent those of all members of the Environmental Resource Group.
Environmental supporting documentation was also reviewed by:
- K. Adare, Environment Canada
- J. Addison, Royal Roads University
- J. Van Barneveld, British Columbia Ministry of Environment, Lands and Parks
- Y. Bédard, Ministère des Transports du Québec
- D. Belluck, Minnesota Department of Transport
- Y. Blouin, Ville de Cap-de-la-Madeleine
- R. Brecher, Global Tox
- C. Chong, University of Guelph
- A. Decréon, Ministère des Transports du Québec
- R. Delisle, Ministère des Transports du Québec
- G. Duval, Ministère des Transports du Québec
- A. El-Shaarawi, Environment Canada
- A. Fraser, Environment Canada
- M. Frénette, Ville de Montréal
- D. Gutzman, Environment Canada
- G. Haines, New Brunswick Department of Transportation
- R. Hodgins, Ecoplans Limited
- M. Kent, British Columbia Ministry of Transportation and Highways
- C. MacQuarrie, New Brunswick Department of Transportation
- B. Mason, City of Toronto
- L. McCarty, L.S. McCarty Consulting
- G. McRae, Ontario Ministry of Transportation
- T. Pollock, Environment Canada
- M. Roberts, University of New Brunswick
- D. Rushton, Nova Scotia Department of Transportation and Public Works
- K. Solomon, University of Guelph
- R. Stemberger, Dartmouth College, New Hampshire
- T. Young, Clarkson University, New York
The Assessment Report was reviewed and approved by the Environment Canada/Health Canada CEPA Management Committee.
The Assessment Report for Road Salts was released in August 2000 for a 60-day public comment period. Following consideration of comments received, the Assessment Report was revised. A summary of the public comments and responses from Environment Canada and Health Canada is available by contacting the Existing Substances Branch (see address below) or on the Internet at:
Copies of this Assessment Report are available upon request from:
- Inquiry Centre
Environment Canada
Main Floor, Place Vincent Massey
351 St. Joseph Blvd.
Hull, Quebec
K1A 0H3
or on the Internet at:
Unpublished supporting documentation, which presents additional information, is available upon request from:
- Existing Substances Branch
Environment Canada
14th Floor, Place Vincent Massey
351 St. Joseph Blvd.
Hull, Quebec
K1A 0H3
e-mail: PSL.LSIP@ec.gc.ca
2.0 Summary of Information Critical to Assessment of "Toxic" Under CEPA 1999
2.1 Identity, properties, production and sources
2.1.1 Substance characterization
Road salts can refer to any salt applied to roadways for roadway maintenance. As outlined below, road salts are mainly used in Canada as de-icing and anti-icing agents during winter road maintenance, but smaller quantities are also used as dust suppressants. "Salt" can refer to any compound consisting of the cation from a base and the anion from an acid and which is readily dissociated in water. While sodium chloride (NaCl) is by far the most frequently used road salt in Canada, other inorganic salts used in Canada include calcium chloride (CaCl2), magnesium chloride (MgCl2) and potassium chloride (KCl).
Few additives are used in Canada as part of road salt formulations; sodium ferrocyanide (Na4Fe(CN)6·10H2O) is the only product regularly added as an anti-caking agent in Canada. Organic salts and a few other products are essentially used in Canada for maintenance of airports and for plane de-icing or limited roadway trials.
Abrasives such as sand are used for winter roadway maintenance; while these are not salts, chloride salts are frequently mixed with abrasives. Accordingly, abrasives blended with salts can be a source of chloride salts to the environment.
This assessment focuses on the inorganic chloride salts used for roadway maintenance. This is based on the following considerations:
- The original recommendations of the Ministers' Expert Advisory Panel called for an ecological assessment of the most commonly used de-icing chloride salts.
- Sodium chloride and, to a lesser extent, calcium chloride are by far the most commonly used salts on roadways in Canada.
- All inorganic chloride salts have broadly similar behaviour and effects in the environment. Notably, effects related to the toxicity of chloride can be considered to depend on the cumulative input of all chloride salts.
In addition, public concerns have been expressed regarding the use of ferrocyanide salts in formulations of road salts, notably given that, in solution, they can photolyse to yield free cyanide ions, which are highly toxic to aquatic organisms. Since ferrocyanides are commonly used in road salt formulations, their entry, exposure and effects are also considered as part of the assessment of road salts.
Organic salts are not used in Canada or are used in specific circumstances, such as at airports (rather than on roadways), and they are not assessed in this report. While abrasives are used in large quantities in Canada, the nature and potential for environmental effects of these compounds are distinct from those of road salts and are not considered in this assessment.
Limited data were available for parking lots, industrial, commercial and other private properties. However, the assessment was based essentially on uses and releases from public roadways, snow disposal sites and patrol yards.
2.1.2 Physical and chemical properties
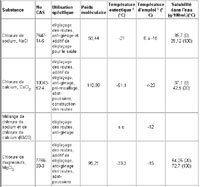
The Chemical Abstracts Service (CAS) registry numbers and physical-chemical properties of four inorganic salts (sodium chloride, calcium chloride, magnesium chloride and potassium chloride) and ferrocyanides used as road salts are given in Table 1.
Table 1 Physical-chemical properties of road salts
The eutectic temperature is the lowest freezing temperature that can be achieved for water by adding a given salt to it. The greater the difference between the ambient temperature and eutectic temperature, the higher the rate of melting (OECD Scientific Expert Group, 1989). Thus, from the salts presented in Table 1, calcium chloride would produce the highest melting rate. The rate of reaction is observed to be approximately the same for sodium chloride and calcium chloride at temperatures between -1 and -4°C; from -5°C downwards, however, sodium chloride acts more slowly than calcium chloride when equal quantities are applied (OECD Scientific Expert Group, 1989).
Salinity is defined as the total dissolved solids in water after all carbonates have been converted to oxides, all bromides and iodides have been replaced by chlorides and all organic matter has been oxidized (Stumm and Morgan, 1981). Since road salts in this assessment are chloride salts, chloride (Cl-) is the principal contributing anion to salinity resulting from the application of these salts, but other contributing anions in the environment include bicarbonate (HCO3-), carbonate (CO32-) and sulphate (SO42-). Cations that contribute significantly to salinity include calcium (Ca2+), magnesium (Mg2+), sodium (Na+) and potassium (K+). Salinity is closely related to the total halide concentration, which is often called chlorinity. The relation between the two is described by the empirically derived Knudson equation (Mayer et al., 1999).
Ferrocyanide is a very complex anion of limited solubility composed of a central iron atom surrounded by an octahedral configuration of cyanide ligands (Letts, 2000a).
2.1.3 Production and use
Road salts (mostly sodium chloride) have been used as ice-disbonding and ice-melting agents in Canada since the 1940s (Perchanok et al., 1991).
2.1.3.1 Sodium chloride
The predominant chloride salt used as a de-icer in North America is sodium chloride, which is composed of about 40% sodium and 60% chloride by weight. Trace elements, including trace metals, may represent up to 5% of the total salt weight. Substances potentially present include phosphorus (14-26 mg/kg), sulphur (6.78-4200 mg/kg), nitrogen (6.78-4200 mg/kg), copper (0-14 mg/kg) and zinc (0.02-0.68 mg/kg) (MDOT, 1993).
World production of sodium chloride totalled 189 000 kilotonnes in 1995. The largest producers are the United States (21%), China (15%), Germany (8%) and Canada (7%) (Natural Resources Canada, 1998). The largest global market for sodium chloride is the chemical industry (60%), followed by table salt (20%) and road de-icing (10%) (CIS, 1994).
As of 1993, there were 12 manufacturers of sodium chloride in Canada, with 24 plants in seven provinces. Total nameplate capacity was 13 645 kilotonnes per year, and total domestic production was 10 895 kilotonnes. A further 1010 kilotonnes were imported from the United States and Mexico, making a total supply of 11 905 kilotonnes. Of this, Canada exported 3106 kilotonnes, mainly to the United States. Canadian domestic demand was 8799 kilotonnes (CIS, 1994). The largest market for sodium chloride in Canada is snow and ice control, which accounts for about half the domestic demand (4240 kilotonnes in 1993). Total amounts used for de-icing fluctuate from year to year, depending on weather conditions (CIS, 1994).
2.1.3.2 Calcium chloride
Calcium chloride is the second most commonly used road salt in North America. Calcium chloride is the leading chemical used for dust suppression in Canada. Liquid calcium chloride is applied primarily to gravel roads to consolidate aggregates and control dust. Calcium chloride is also used to pre-wet salt or sand in winter highway maintenance and to stabilize road base mixtures after pulverization. Of these three uses, dust control accounts for approximately 97% of total use (Morin and Perchanok, 2000). The quantity of calcium chloride used for winter road maintenance may increase as different agencies start experimenting with pre-wetting techniques.
In 1995, there were three calcium chloride producers in Canada, with one plant in Ontario and four brine wells in Alberta. Total nameplate capacity for calcium chloride in Canada for that year was 629 kilotonnes. At that time, domestic production totalled 399 kilotonnes and 25 kilotonnes were imported, creating a total supply of 424 kilotonnes. Of this, 156 kilotonnes were exported, while Canadian domestic demand was 268 kilotonnes. The total amount used in 1995 in road dust suppression and in road construction was reported at 201 kilotonnes (CIS, 1996).
2.1.3.3 Mixture of sodium chloride and calcium chloride
Sodium chloride pre-wetted with calcium chloride brine has been recommended for reducing total salt applications (Gooding and Bodnarchuk, 1994). The total amount used for roads in Canada is included in the above estimates for sodium chloride and calcium chloride.
2.1.3.4 Magnesium chloride
Although not recommended for dust control on roads, magnesium chloride is an alternative dust suppressant for use on material piles, road shoulders or material transfer ponds (OMEE, 1993). Using data provided by industry, Morin and Perchanok (2000) estimated that approximately 25 000-35 000 tonnes of magnesium chloride are used annually for roadway de-icing activities in Canada. No information was found concerning magnesium chloride production volumes in Canada, but volumes are likely quite low.
2.1.3.5 Mixture of sodium chloride and magnesium chloride
While sodium chloride-based de-icing products can contain magnesium chloride as well as some corrosion-inhibiting additives (MDOT, 1993), there is no evidence of their current use in Canada.
2.1.3.6 Potassium chloride
There is limited use of potassium chloride in road de-icing chemicals; however, potash mine tailings containing 0.5-2% potassium chloride (the rest is primarily sodium chloride) are applied to some Canadian roads for de-icing (CFI, 1997). The total amount of potassium chloride used annually for roadway de-icing in Canada has been estimated to be 2000 tonnes (Morin and Perchanok, 2000) and 3300 tonnes (CFI, 1997). It is estimated that total Canadian production of potassium chloride in 1999 was 13.6 million tonnes, and the potash producing industry operated at 61% of production capacity (Prud'homme, 2000). Most of the potash produced was used for agricultural purposes.
2.1.3.7 Brines
Oil field brine is a traditional dust suppressant containing calcium, magnesium, sodium and chloride. Oil field brine is obtained as formation water generated from oil exploration operations at several locations in southwestern Ontario (OMEE, 1993).
2.1.3.8 Ferrocyanide
To prevent the clumping of chloride salts during storage and de-icing operations, sodium ferrocyanide and ferric ferrocyanide can be added to these salts. In Canada, sodium ferrocyanide is added to sodium chloride in amounts of 30-124 mg/kg (Letts, 2000a).
Sodium ferrocyanide is not currently produced in Canada. However, approximately 300-350 tonnes per year are imported from European and Asian manufacturers for use as an anti-caking agent. Road salts containing sodium ferrocyanide are used in Ontario, Quebec and the Atlantic provinces. From Manitoba to the interior of British Columbia, the by-product salt from Saskatchewan potash mines that is used as road salt is not treated with sodium ferrocyanide. On the west coast, rock salt imported from Chile is treated with sodium ferrocyanide at a rate of 60 mg/kg. The quantity of ferrocyanide used in various jurisdictions can be estimated from the amount of salt used and its average ferrocyanide content.
2.1.4 Sources and releases
2.1.4.1 Natural sources
Natural sources of sodium chloride, calcium chloride, magnesium chloride and potassium chloride salts in the environment include weathering and erosion of rocks and soils, atmospheric precipitation and groundwater discharge. The composition of the rock material, relief and climate are important factors that largely control the rates of weathering and dissolution. In aquatic ecosystems, cations such as calcium, magnesium and potassium are not uniquely associated with chlorides but can be derived from natural sources such as carbonates from soils and bedrock in the watershed (Mayer et al., 1999). Igneous rocks and associated soils have generally lower salt contents than shales and limestones (Pringle et al., 1981). The contribution of salinity from atmospheric sources is particularly important in the coastal maritime regions. Areas that are dry with low rates of precipitation, such as the Canadian Prairies, have landscapes that tend to concentrate salts in surface soils because of high net evapotranspiration from the land. Natural sources of salts in aquatic ecosystems are reviewed by Evans and Frick (2001).
Sodium and chloride are stored in vegetation, but they are also easily lost by ion uptake, leaf washing and litter decomposition. Chloride is an important inorganic solute of osmotic regulation. The chloride concentration in the environment is not affected by chemical or biological reactions. Chloride does not adsorb onto particulate matter. Potassium ion tends to be the most constant quantity in animal cells. Calcium is the most reactive ion of all the major cations contributing to salinity; it is required as a nutrient in higher plants and is one of the basic inorganic elements present in algae. Magnesium is important in enzymatic transformations and is required by chlorophyll-bearing plants.
While ferrocyanides do not occur naturally in the environment, cyanide compounds are naturally occurring substances reported from over 2000 plant species (Conn, 1980). Sodium ferrocyanide and ferric ferrocyanide are highly stable and relatively immobile in the environment. When they are exposed to sunlight, the ferrocyanide complex decomposes and releases cyanide ions (CN-) (Meeussen et al., 1992a). Under natural conditions, the cyanide ion hydrolyses to the volatile hydrogen cyanide (HCN) molecule (Shifrin et al., 1996).
2.1.4.2 Anthropogenic sources
Anthropogenic sources of inorganic chloride salts include domestic sewage and industrial processes (Sonzogni et al., 1983), such as effluent from the chemical and petrochemical industry (Johnson and Kauss, 1991), gas manufacturing and acid mine drainage (U.S. EPA, 1973; Pringle et al., 1981). Once these substances enter the environment, they remain in aqueous phase until their concentrations exceed their solubility products, when crystallization and subsequent sedimentation of mineral salts might occur.
Salts may be released to the environment during their production, mining, mixing, and bulk transportation and storage. Since these industrial sources are not all limited to ultimate use as road salts, they are not considered further in this assessment.
Ultimately, all road salts enter the environment as a result of:
- storage at patrol yards (including losses from storage piles and during handling);
- roadway application (at the time of application as well as subsequent movement of the salts off the roadways); and
- disposal of waste snow.
Releases are therefore associated with both point sources (storage and snow disposal) and line sources (roadway application). The following sections review material pertinent to entry into the environment, notably loadings (amounts applied to roadways), disposal of waste snow and patrol yards.
2.2 Road salt loadings
This section summarizes findings in a report prepared by Morin and Perchanok (2000), which characterizes the use of road salts in Canada. Information describing application rates and annual mass loadings per highway length and per geographic district area is provided. These detailed, geographically based data were used to calculate exposure and to support subsequent sections of the assessment. Data in this section focus on the use of sodium chloride and calcium chloride salts. Magnesium chloride and potassium chloride salts are used to a lesser extent. Industry representatives estimated that approximately 25 000-35 000 tonnes of magnesium chloride and 2000 tonnes of potassium chloride are used for roadway de-icing in Canada annually.
2.2.1 Surveys
Information on the use of road salts in Canada was obtained from surveys conducted by the Transportation Association of Canada, the Ontario Ministry of Transportation and Environment Canada. The Environment Canada survey included data from provinces, municipalities and the private sector. Details on the methodology used in the surveys are presented in Morin and Perchanok (2000).
2.2.1.1 Provinces
Information on salt loadings and road network length was obtained from all provincial and territorial departments of transportation. The 1997-98 winter had the most complete data nationwide; these data were used to estimate total salt use. Comparisons between data from salt suppliers and data from provincial agencies suggest that the quantities reported are reliable (Morin and Perchanok, 2000).
2.2.1.2 Municipalities and regional governments
Information on salt use and road network length was obtained from 104 municipalities across Canada. Data on salt use were also obtained from all regional municipalities and counties in Ontario. Some respondents provided information on salt use and road network length for each of the past 5 years; others provided information on salt use for the 1997-98 winter only.
2.2.1.3 Salt industry data
Salt suppliers provided purchase and tender data for 500 municipalities in Canada. A comparison between tender data and purchase data indicates a strong concordance between the two.
2.2.1.4 Municipal estimates
Information on salt use by road type is available only for 104 municipalities. Even by combining data obtained from municipalities (104) and salt suppliers (500 municipalities), this does not represent total municipal salt use. Estimates were made to account for salt use by municipalities where no data were submitted.
Population was chosen as the variable to predict municipal salt use. This was done by deriving an average annual loading per person for municipalities with survey or purchase/tender data and multiplying this by the population of all other municipalities in a maintenance district. By summing known municipal salt use and estimated municipal salt use, it was possible to estimate total municipal salt use by maintenance district. After estimates were made, salt use was mapped by maintenance district.
Figure 1 Provincial road network area fraction (from Morin and Perchanok, 2000)

2.2.1.5 Provincial and municipal use of dust suppressant salts
The major producer of calcium chloride provided market estimates of calcium chloride use on roadways by province. Recommended application rates for calcium chloride were obtained from industry. Rates vary depending on the type of gravel road, traffic and policies regarding roadway maintenance. Minimum and maximum loadings for a gravel road 7.4 m wide were calculated.
2.2.2 Results
Detailed results are presented in Morin and Perchanok (2000).
Using survey information, maps depicting the distribution of provincial roads were created. A provincial road network area fraction was calculated and mapped (Figure 1 indicates the area of saltable roads maintained by provincial agencies per district area). This map can be used to estimate salt loadings to surface water in drainage areas of a similar size and shape. Districts with the highest proportion of saltable road to district area are in parts of the Atlantic provinces, southern Quebec, southern Ontario and parts of British Columbia.
Figure 2 Recommended provincial application rates for sodium chloride road salts, 1998 (from Morin and Perchanok, 2000)

2.2.2.1 Sodium chloride
Information on recommended provincial application rates is depicted in Figure 2; rates for select municipalities are presented in Figure 3. Application rates cannot be used to quantify annual loadings; they provide information on the quantity of salt applied with each application, not the number of applications and resulting total mass applied. Loadings refer to average total annual mass, not application rates.
Detailed data were analysed separately for provinces, municipalities and regional governments, including calculations of mass of salts per length and surface area of two-lane saltable roads and per area of maintenance district (Morin and Perchanok, 2000). Areas with highest loadings were in central and southern Ontario and Quebec, followed by the Atlantic provinces; lowest loadings were in the Prairies.
Municipal loadings were combined with provincial and territorial loadings to estimate total salt use by maintenance district for the 1997-98 winter season (Figure 4); it is estimated that 4 418 462 tonnes of sodium chloride were used as de-icers on roadways in Canada.
The above estimates of total loading include data on the use of road salts by municipal, provincial, territorial and regional governments only. Private contractors, industry and agencies such as port and transit authorities also use road salts. Cheminfo (1999) estimated that the total use by commercial and industrial customers is 5-10% of the road salt market. A midpoint value of 7.5% was used to estimate the quantity of road salts used by commercial and industrial sources in Canada (Table 2). If these quantities are included, it is estimated that 4 749 847 tonnes of sodium chloride road salts were used during the 1997-98 winter season.
Figure 3 Average recommended municipal application rates for sodium chloride road salts, 1998 (from Morin and Perchanok, 2000)

Figure 4 Total amount of sodium chloride used, by maintenance district (from Morin and Perchanok, 2000)


- Commercial and industrial road salt use is assumed to be at 7.5% of provincial and municipal use (7.5% based on estimate by Cheminfo, 1999).
- Including Nunavut.
2.2.2.2 Calcium chloride
The quantity of calcium chloride used by province and territory is listed in Table 3. While Ontario is the province where the largest quantity of calcium chloride is used on roadways, analyses by Morin and Perchanok (2000) indicate that Nova Scotia is the province with the highest usage per unit area of land.
2.2.2.3 Total chloride loadings
Estimates of the quantity of sodium chloride and calcium chloride used on roadways in Canada are combined to estimate total chloride use on roadways by province. Table 4 presents total chloride use by province, based on total sodium chloride loadings for the 1997-98 winter season and the estimated use of calcium chloride in a typical year. Mass of chloride used per area of province was calculated to provide a basis on which to compare loadings. Thus, while Ontario and Quebec are the provinces where the most chloride is used on roadways, Nova Scotia has the highest loading per unit area of province (Morin and Perchanok, 2000).
2.2.2.4 Historic trends
Changes in road salt loadings over time were considered. A few agencies that maintain roadways provided long-term information on total salt use. While these data suggest that total road salt loadings have generally increased over time, they also indicate that there are annual fluctuations. Furthermore, it is difficult to conclude that loadings have increased without information on the length of the road network that is maintained.
1 Including Nunavut.
1 Including Nunavut.
One way to roughly ascertain if annual road salt loadings have changed is by comparing survey data compiled by the Salt Institute from the mid 1960s to early 1980s (Salt Institute, 1964-1983) with data collected for the 5-year survey period (1993-94 to 1997-98). This comparison suggests that road salt loadings per two-lane-kilometre have not decreased.
While some agencies used similar quantities of salt per two-lane-kilometre over both time periods, road salt loadings by other agencies increased slightly from the early 1980s to mid 1990s. Figure 5 shows average provincial loadings per two-lane-kilometre of provincial road for Ontario, Quebec, New Brunswick and Nova Scotia.
Figure 5 Historical salt use by provincial agencies (based on surveys conducted by the Salt Institute, 1964-1983, and Morin and Perchanok, 2000)

The comparison and merger of data from different sources for different time periods were assessed by comparing the correspondence of different data sets for provincial agencies in Ontario and Nova Scotia. This type of comparison could not be done for the other provinces, because overlapping data were not available. When data could be compared, however, there was a concurrence between the quantities reported. For example, data obtained from the Salt Institute indicate that the Ontario Ministry of Transportation used 418 997, 486 648 and 402 346 tonnes of sodium chloride for the winters of 1978-79, 1981-82 and 1982-83, respectively (Salt Institute, 1964-1983). Data obtained from the Ontario Ministry of Transportation indicate that it used 415 381, 486 648 and 402 346 tonnes of sodium chloride for the same years, respectively. Data obtained from the Salt Institute and Nova Scotia Department of Transportation and Public Works both indicate that 201 989 and 129 453 tonnes of sodium chloride were used during the winters of 1981-82 and 1982-83, respectively. While this comparison is limited in scope, it suggests that data obtained from the Salt Institute and provincial agencies concur.
2.2.3 Reliability of loadings data
Although a concerted effort was made to obtain data from the most accurate sources, errors in data and calculations are possible. Quantities of materials applied may have been inaccurately recorded by agencies, or there may be non-reported salt usage. There could also be inconsistencies in the data that were provided. For example, some provinces purchase salts for use by municipalities or maintain municipal roads. Hence, municipal data may be included in the provincial loadings for certain districts. Another problem could be that survey data generally refer to salt purchased and not salt used. While all the salts purchased will eventually be used, there is no way of determining the quantity of salt that is applied each year. Potential errors with data from salt suppliers were assessed by correlating provincial bids and usage. Overall, both data sets are highly correlated.
One possible source of error could be the use of 1997-98 data to characterize salt loadings. While these quantities are a best estimate, available provincial data indicate that loadings varied by year. A comparison of the total salt loadings per unit length of road for all agencies that provided 5 years of data suggests that the 1997-98 winter season is not atypical (Figure 5). To validate assumptions that the 1997-98 winter season was representative, two statistical analyses were done (Collins, 2000). Data were analysed using an analysis of variance (ANOVA), and, since data were not normally distributed, they were also analysed using the non-parametric Friedman's two-way analysis by ranks. Whenever there was a significant difference among years, Ryan's step-down procedure tested for differences among years. Results of these analyses indicate that the loadings were somewhat lower for the 1997-98 period (Collins, 2000).
A potential limitation of analyses may be the use of a "saltable" category to characterize road salt loadings. Since highways generally receive higher loadings, it could be argued that the saltable category underestimates salt loadings on high-volume roads. Another limitation may be the use of population to estimate salt loadings for municipalities with no data. While there is a good correlation between municipal population and salt use at the provincial level, the strength of this relation varies by maintenance district.
Despite these potential limitations, data presented are good indicators of the quantities of salt used on roadways in Canada. This is particularly so when considering that there is a difference of 9.4% (417 501 tonnes) between known and estimated usage of sodium chloride by municipal, county and provincial agencies for the 1997-98 winter.
2.2.4 Summary
The estimated annual use of sodium chloride and calcium chloride road salts in Canada was determined from surveys of provincial, territorial and municipal agencies, industry data and estimates based on population. It is estimated that approximately 4.75 million tonnes of sodium chloride de-icers were used in the 1997-98 winter and that 110 000 tonnes of calcium chloride are used on roadways in a typical year. When combined, it is estimated that 2.95 million tonnes of chloride were used on roads in Canada during the 1997-98 year. It is recognized that amounts used will vary on a yearly basis, notably because of variation in climatic conditions.
Annual loadings vary geographically, with the highest loadings on a length of road basis occurring in Ontario and Quebec, intermediate loadings in the Atlantic provinces and lowest loadings in the western provinces. Overall, data presented in this section indicate that road salt loadings have not decreased over the past 20 years. Furthermore, data for some provinces suggest that loadings have increased since the late 1970s (Figure 5).
2.3 Snow disposal
Delisle and Dériger (2000) reviewed the physicochemical and ecotoxicological characteristics of roadside snow and the different methods used for removing snow from roads and sidewalks.
2.3.1 Characterization of snow from roadways
Road salts contribute to the presence of sodium, chloride and cyanide ions in snow. Other major contaminants also found in urban snow include debris, suspended solids, oil and grease, and metals (lead, manganese, iron, chromium) (Gouvernement du Québec, 1997). Average concentrations of chlorides in roadside snow or its meltwater for certain municipalities in Quebec ranged from 3.8 to 5689 mg/L (see Delisle and Dériger, 2000).
Chenevier (1997) analysed the physicochemical parameters of roadside snow from Montréal's primary and secondary streets in 1997. Concentrations for all parameters were higher for the primary streets. Average concentrations of chloride were 3115 mg/L for secondary streets and 5066 mg/L for primary streets (Delisle and Dériger, 2000). This difference is probably due to the higher frequency or rates of application of road salts. A similar study by Delisle et al. (1997) monitored the physicochemical characteristics of roadside snow for two snowstorms in January 1997. Mean chloride concentrations for samples taken during the first and second storms were 7716 and 3663 mg/L, respectively. Chloride concentrations for the 24 samples taken during both storms ranged between 1366 and 18 230 mg/L.
The characteristics of roadside snow depend on a wide range of factors, including community size, the land occupancy factor, traffic density, quantities of de-icing salt and abrasives used, and duration of snow clearing (Malmqvist, 1985; Delisle et al., 1995). Contaminant content generally changes with time. For example, concentrations of chloride were 530 mg/L after 72 hours for snow from residential city streets and 7496 mg/L after 72 hours for snow from commercial city streets (Delisle and Leduc, 1987). Concentrations subsequently diminished to 590 mg/L after more than 1 week, presumably due to loss of ions though meltwater.
2.3.2 Volumes of snow in Canadian cities
Removal and disposal of snow may be required when the accumulation of snow on or along roadways may hamper traffic or safety. As such, the quantity of snow to be removed and disposed of depends on the volume of snow and the extent of urban development (Table 5). Given the large amount of snowfall in Montréal (annual snowfall in southern Quebec is between 200 and 350 cm) and the size and density of the city, about 11.258 million cubic metres of snow were brought to snow disposal sites during the winter of 1997-98. This quantity can be compared with 1.5 million cubic metres for Toronto (before municipal amalgamation) and a total of about 30 million cubic metres of snow for all municipalities in Quebec.
2.3.3 Snow clearing and snow disposal methods
Clearing of snow generally involves plowing snow to the side of the roadway. In cities, snow clearing can begin as snowfall reaches 2.5 cm, with the snow being pushed to the sides of streets and onto sidewalks. However, when there is considerable accumulation (more than 10 cm) or when necessary, snow is cleared and transported to various disposal sites (City of Montréal, 1998). The various snow disposal methods have been reviewed by Delisle and Dériger (2000) and can be grouped into three categories, as described below.
2.3.3.1 Methods not involving snow removal
Methods for clearing snow from the roadway without transporting it to snow disposal sites typically involve plowing snow to the side of the road or blowing it onto land adjacent to the roadway. These methods are generally not effective in areas with a high land occupancy factor (Delisle, 1994). These methods are, however, the most prevalent approach to the clearing of snow from roadways in non-urban areas. This type of approach contributes to potential impacts of salts on roadside soils, vegetation, surface water and groundwater. Data collected by Watson (2000) characterized chloride concentrations in ponds and wetlands adjacent to roadways in southern Ontario. Results of this study indicate that the chloride concentrations are variable, but concentrations greater than 4000 mg/L were observed. This study indicates that runoff from roadways can adversely affect aquatic environments adjacent to roadways.
A study by Delisle (1999), which associated elevated chloride concentrations in municipal wells with the use of road salts, also indicated that high chloride concentrations were observed in an area where ditch waters were left to percolate through the soils. Concentrations of chloride at three municipal production wells in the study area increased by 102, 116 and 145 mg/L between 1983 and 1994.
- These quantities are approximate and vary yearly with snowfall amount. N/A = not available.
- Plus 100 000 tonnes of a sand/salt mix containing 5% NaCl.
- Plus 39 000 tonnes of a sand/gravel mix (1:9 for sidewalks and 5:5 for streets) during freezing rain.
2.3.3.2 Methods involving release into waterways after processing or treating meltwater
Roadway snow can be transported to snow disposal sites where snow melts and the meltwater is treated. This typically involves dumping snow at surface sites or in quarries where runoff is channelled to treatment facilities. Generally, snow disposal sites are located on impermeable or slightly permeable ground or must be equipped with a geotextile membrane. Some sites are also equipped with sedimentation facilities or are designed to direct the meltwater towards a wastewater treatment system (Gouvernement du Québec, 1991). These sites should not be located next to watercourses that could be affected by runoff.
Pinard et al. (1989) characterized chloride concentrations in runoff from snow disposal sites. This study indicated that only 2% of the salt spread on city streets was present in meltwater from snow disposal sites, with most of the salt likely released to the environment from the roadway or roadside. This concurs with Delisle and Leduc (1987), who indicated that chloride concentrations in roadside snow initially increase then decrease with increasing time. The concentration of chloride in snow removed from roadways will, however, vary by street type; concentrations in snow collected from primary streets can be an order of magnitude higher than concentrations in snow from secondary streets.
While the percentage of salt present in snow transferred to snow depots may be low (e.g., 2%), the concentration of chloride in runoff is still elevated. A study by Péloquin (1993) indicated that the average chloride concentration in meltwater from a snow disposal site was 414 mg/L. Pinard et al. (1989) monitored chloride concentrations in runoff from a Québec City snow disposal site from April 18 to the end of June 1988. Concentrations in runoff ranged from approximately 100 to 1100 mg/L. Concentrations were highest in the early sampling and gradually decreased throughout the spring. It is not known if more elevated chloride concentrations were present in runoff prior to the monitoring program or if the concentrations increased throughout the summer due to a decrease in the volume of runoff water.
The potential impact of snowmelt on groundwater quality will vary by disposal site. A study described in Morin (2000) indicates that chloride concentrations between 233 and 1820 mg/L were measured at monitoring wells installed to assess the impact of a snow disposal site on shallow groundwater quality.
Other agencies (e.g., City of Montréal) dump snow through chutes linked to municipal sewer networks (Godbout, 1996; Couture, 1997). Certain agencies use snow melters. The underlying principle of melters is to dump water rather than snow into the sewer system. Melters can be stationary or mobile. Snow is dumped by trucks into preheated water tanks equipped with oil or gas burners. The City of Toronto uses this technique occasionally. So-called "geothermal" snow melting, using geological formations as a natural storage reservoir for water during the summer and keeping the water sufficiently hot to melt the snow during the winter, has been used since January 1998 in Cap-Rouge, Quebec (Bilodeau, 1999).
2.3.3.3 Methods involving release to surface water without water treatment
This method of disposing of snow involves dumping snow directly into a waterway or onto its banks. Snow can also be dumped down sewer chutes that are not linked to treatment plants. This results in the release of waste snow and any contaminants to surface water directly (dumping into rivers or into the ocean), with some removal of large debris (dumping onto banks) or with possible dilution by stormwater (dumping into sewer chutes not linked to treatment plants).
2.3.4 Use of disposal methods by Canadian municipalities
The use of snow disposal approaches by selected Canadian municipalities is shown in Table 6. The City of Montréal uses the greatest variety of disposal methods. In 1997-98, the City of Montréal used surface sites (7 sites), sewer chutes (11 sites), dumping into the St. Lawrence River (3 sites) and quarry dumps (2 sites) (City of Montréal, 1998).
Surface sites are clearly the most common method of disposal in Canada. For example, Regina uses between two and five surface disposal sites, depending on the year (City of Regina, 1997). In the case of the Regional Municipality of Ottawa-Carleton, several studies have been conducted to determine the number of surface sites needed to accommodate snow removal in that region (McNeely Engineering, 1990; McNeely-Tunnock Ltd., 1995). A few municipalities (e.g., St. John's, Halifax and Vancouver) dump snow into the sea at harbours. The direct dumping of snow to fresh surface water is restricted and will not be permitted in Quebec as of 2002 (Gouvernement du Québec, 2001).
In the past, municipalities such as Montréal blew snow onto private property and occasionally used snow melters. Blowing onto private land was virtually abandoned by Montréal because of social and political pressure, and snow melters proved to be too expensive to operate because of high fuel costs (City of Montréal, 1998). In certain cities like Halifax, large quantities of de-icing salt are used, but little snow is removed; roadway meltwater is channelled to storm sewer systems (Delisle and Dériger, 2000).
2.3.5 Summary
In addition to inorganic ions from road salts, waste snow can contain a broad range of physical and chemical contaminants. Chloride ions are dissolved and ultimately transported in meltwater and are largely not affected by physical or biological water treatment. Impacts depend on total amounts and concentrations of the ions at the point of release of meltwater into surface water or into soil and groundwater. While chloride concentrations in waste snow and its meltwater can be quite variable, they can be as high as 18 000 mg/L. Average chloride concentrations in snow from streets in Montréal were approximately 3000 and 5000 mg/L for secondary and primary streets, respectively. Canadian municipalities use a variety of snow disposal techniques, but surface sites are the most common. While there is uncertainty regarding the portion of salt that may be transported to snow disposal sites, it is clear that runoff from these sites has elevated chloride concentrations.
2.4 Patrol yards
Patrol yards (also referred to as storage yards or maintenance yards) are used to store road maintenance materials before their application to roadways. The following sections describe and characterize these facilities, outline environmental exposure pathways and present measured concentrations from specific points around patrol yards. Most of the data are based on pre-1998 standards for patrol yard design and the storage of road salts. While more effective designs are currently being promoted to reduce salt loss (TAC, 1999), current design and management are largely similar to those considered in this assessment. Data in the following sections were summarized from a report prepared by Snodgrass and Morin (2000).
2.4.1 Number of patrol yards and quantity of materials stored
Data compiled by Morin and Perchanok (2000) were used to estimate the number of provincial and territorial patrol yards and the quantity of road salts and abrasives stored at these yards. There is no definite information on the numbers of patrol yards in Canada, but it is estimated that there are 1300 provincial patrol yards. Assuming that total salt and abrasive use by jurisdictions is distributed equally among patrol yards, the average quantities used at patrol yards were estimated (Table 7). This table does not include municipal and county yards and storage facilities used by private contractors and hence significantly underestimates the number of patrol yards in Canada. The average amount of salts and abrasives stored at patrol yards varies considerably across Canada. Table 7 suggests that the average amount of salts stored at patrol yards in Ontario, Quebec and the Maritime provinces ranges between 1300 and 3800 tonnes. The average amount of abrasives for these provinces ranges between 1000 and 8600 tonnes. The number of patrol yards by district and the average tonnage of sodium chloride and abrasives that would be used by patrol yards for selected provinces are presented in Snodgrass and Morin (2000).
1 Including Nunavut.
Data for the winter of 1997-98 from 117 patrol yards administered by the Ontario Ministry of Transportation indicate that the quantity of sodium chloride stored at these patrol yards ranges between 45 and 21 400 tonnes per yard; the quantity of abrasives stored ranges between 10 and 13 200 tonnes per yard.
2.4.2 Patrol yard design and management
Patrol yards can be located in a variety of settings, and patrol yard design and standards vary substantially across Canada. Accordingly, the degree of protection against weathering varies considerably. Covered facilities used to store road salts can include domes/igloos, sheds and lean-tos. Doors or walls may or may not be present, and storage under an overpass can be considered as covered by some agencies. Salt may be stored on asphalt or concrete pads or outside on a thick plastic tarp and covered by another tarp. While storage in covered facilities can protect against precipitation, which leaches the salt, there is no universal standard design for storage facilities. Use of best management practices that promote good housekeeping at patrol yards is equally important to reduce salt loss. The effectiveness of best practices, however, depends on the rigour with which they are implemented.
Salts can be released from mixed abrasive/salt piles as well as from salt piles. A mixed abrasive/salt pile consists of either sand or gravel mixed with salt to prevent the abrasive from freezing and to keep the abrasive in a flowing state (NB DOE and DOT, 1978). The percentage of salt varies - most agencies have 5% mixtures, but this can range between 2.5 and 15% salt. A New Brunswick study (NB DOE and DOT, 1978) monitored the quantity and quality of leachate from a 2000-tonne abrasive pile with a 2.5% salt content. During the first year, 420 m3 of leachate passed through the monitoring system. Sodium and chloride concentrations in the leachate are depicted in Figure 6. The maximum concentrations for sodium and chloride were 37 000 and 66 000 mg/L, respectively. It was estimated that total salt loss through leaching during the first year was 18.2 tonnes, or over a third of total salt added to the pile, even though 80% of the abrasive pile had already been removed for sanding operations by the end of January. Results of the second year also indicated high concentrations in leachate from the pile. A total of 255 m3 of leachate was measured. The maximum concentrations for sodium and chloride were 49 000 and 82 000 mg/L, respectively. Considering these concentrations and the results of a survey suggesting that few provincial agencies cover mixed abrasive/salt piles (Snodgrass and Morin, 2000), the potential for salt loss from mixed abrasive/salt piles is high.
Spillage during stockpiling and loading of spreaders is a major source of salt loss (TAC, 1999), as confirmed by a review of electromagnetic surveys done at patrol yards (Snodgrass and Morin, 2000). These surveys, which typically identify subsurface materials and groundwater (within 6 m of the surface) that are highly conductive (e.g., soils and groundwater with elevated levels of salts), indicated high conductivity in areas adjacent to driveways and around storage facilities where salts are handled.
Another potential source of contamination is from washwater used to wash winter road maintenance vehicles. At patrol yards in urban areas, the washwater is likely released to a municipal sewage treatment system; in rural areas, discharge is probably to a dry well and left to percolate through the soil. Washwater samples from seven patrol yards had chloride concentrations between 3500 and 37 000 mg/L; the average concentration was 16 000 mg/L (Beck et al., 1994). This study also estimated that between one-third and two-thirds of patrol yards in Ontario discharge washwater to dry wells. Chloride concentrations in washwater can also be estimated by analysing chloride concentrations in oil/water interceptors. These devices have been installed at some patrol yard garages to remove oil, grease and similar compounds from washwater waste streams. Since oil/water interceptors do not remove salt, concentrations in discharges will be the same as in the influent. A study by INTERA Consultants (1996) indicated that chloride concentrations in oil/water interceptors at three patrol yards ranged between 1100 and 35 400 mg/L. These concentrations roughly concur with those monitored by Beck et al. (1994).
2.4.3 Estimation of releases of salts from patrol yards
Estimates of releases of salts from patrol yards are addressed in this section; details of the assumptions used to calculate these releases are in Snodgrass and Morin (2000). Estimates of releases to the environment were made by considering a hypothetical scenario of releases from 190 active yards. Total estimated releases from patrol yards were then compared with a total roadway application of approximately 590 000 tonnes annually. This hypothetical scenario corresponds roughly to a major province or several smaller provinces as the aggregate basis for the calculations.
Figure 6 Average daily concentration of leachate from salt-treated sand pile, 1975-76 (from NB DOE and DOT, 1978)

Table 8 provides three estimates based on the following scenarios: (1) a patrol yard with best management practices where salt and mixed abrasive piles are stored indoors; (2) a patrol yard where salt piles are stored indoors and abrasive piles are stored outdoors; and (3) a patrol yard where neither abrasive piles nor salt piles are stored indoors. Of the three scenarios, options 1 and 2 are probably most representative of patrol yards. The last column of Table 8 estimates the percentage of total road salt use that may be lost at patrol yards. Depending on the facility and salt management options, 0.2-20% of total salt use can be lost at patrol yards.
2.4.4 Salt concentrations at different areas around patrol yards
The following sections provide an overview of concentrations observed at surface and subsurface endpoints at patrol yards.
2.4.4.1 Concentrations at surface points around patrol yards
A limited database for surface drainage systems around patrol yards is available from environmental site assessments (ESAs) done at some Ontario Ministry of Transportation patrol yards (MTO, 1993-1999). Chloride concentrations at 29 yards where samples were taken ranged between 4 and 4880 mg/L; the mean chloride concentration was 659 mg/L.
A similar type of study was done for a yard in Alberta. A sample taken from the evaporation pond located on this property had a chloride concentration of 32 100 mg/L (Alberta Infrastructure, 2000). A study by the Nova Scotia Department of Transportation and Public Works indicated that runoff and shallow groundwater flowing from a patrol yard to a small brook increased chloride concentrations in a pond located approximately 250 m downgradient from less than 75 mg/L to approximately 1000 mg/L (Rushton, 1999).
2.4.4.2 Concentrations in patrol yard well water
A study by the Ontario Ministry of Transportation (MTO, 1997) graphed chloride concentrations from over 300 patrol yards in Ontario over a 20- to 30-year period ending in 1988. A 5-year period, 1983-1988, was selected to characterize the range of concentrations that could be observed in patrol yard wells. Of the yards in the analysis, most (229 yards, 74%) had chloride concentrations below 200 mg/L. A smaller number of yards (82 yards, 26%) had concentrations above 200 mg/L, including 30 yards (9%) with concentrations greater than 500 mg/L and 14 yards (4%) with concentrations greater than 1000 mg/L.
ESAs done for patrol yards in the Ontario Ministry of Transportation's Eastern Region tested the quality of water in potable water wells (MTO, 1993-1999). Concentrations of chloride in samples from 36 wells ranged between 1.5 and 5050 mg/L; the mean concentration was 722 mg/L. Sixty-four percent of the samples had chloride concentrations greater than 100 mg/L, and 50% of the samples had chloride concentrations that exceeded 250 mg/L. Thirty-five percent of the samples had concentrations greater than 500 mg/L. The depth of the wells sampled ranged from 12 to 113 m, with an average depth of 39.2 m. Elevated chloride concentrations at wells do not necessarily indicate that the entire aquifer is contaminated. For example, faulty well construction can cause salts to flow down around the well casing to the groundwater.
2.4.4.3 Concentrations in shallow groundwater
The same set of ESAs (MTO, 1993-1999) was also used to assess the quality of shallow groundwater. At several patrol yards, monitoring wells were installed to depths between 0.26 and 5.25 m and groundwater was sampled. Chloride concentrations ranged between 1 and 24 000 mg/L; the mean chloride concentration for the 102 samples was 2600 mg/L. A summary of the data indicated that 75% of the samples had chloride concentrations greater than 100 mg/L and 69% of the samples had chloride concentrations that exceeded 250 mg/L.
In similar types of analyses, chloride concentrations in groundwater at three Alberta patrol yards ranged between 26 and 26 400 mg/L (Alberta Infrastructure, 2000). Chloride concentrations in shallow groundwater at three patrol yards managed by the Nova Scotia Department of Transportation and Public Works ranged between 254 and 38 600 mg/L (Rushton, 1999).
2.4.4.4 Concentrations in patrol yard soils
At several patrol yards subjected to ESAs, test pits and bore holes were dug and soil samples were analysed (MTO, 1993-1999). Chloride concentrations from 53 test pits ranged between 5 and 14 500 µg/g soil; sodium concentrations for 46 samples ranged between 39 and 13 100 µg/g soil. The average concentrations for chloride and sodium were 2100 and 2600 µg/g soil, respectively.
Chloride concentrations in soil samples from 46 bore holes ranged between 2 and 13 300 µg/g soil. The average chloride concentration was 1500 µg/g soil. Sodium concentrations for 98 samples ranged between 86 and 6720 µg/g soil; the average sodium concentration was 870 µg/g. Bore holes were between 0.1 and 5 m deep, with an average depth of 1.6 m.
2.0 Summary of Information Critical to Assessment of "Toxic" Under CEPA 1999 (Continued)
2.4.5 Off-site concentrations at patrol yards
A review of select case studies by Morin (2000) indicated that there have been high off-site concentrations of salts at several locations where road salts are stored. A few examples are presented in the following paragraphs.
Arp (2001) examined how the presence of a salt depot can be associated with elevated levels of salinity in surface waters. Sodium concentrations in areas surrounding the depot varied by type (pond and stream water), location and season. Sodium concentrations were highest at and near the salt depot, ranging from 3000 to more than 10 000 mg/L. Runoff and seepage from the depot were quickly diluted by flowing surface water next to the depot, with sodium concentrations decreasing to about 100-300 mg/L. There were also seasonal variations: salt concentrations near the salt depot were lowest during snowmelt and the rainy fall season. During summer, salt concentrations increased with reduced precipitation and increasing air temperature. Highest concentrations occurred during summer months. At this time, the high salt concentration envelope that surrounds the salt depot spread into the adjacent pond and caused elevated salt concentrations in the main drainage stream.
Exposed abrasive piles and salt reserves stored in sheds that offered poor protection from the elements resulted in the contamination of the municipal water supply for Heffley Creek, British Columbia (BC MoTH, 1993). Road salts and unprotected abrasive piles with a 3% salt mixture had been stored at this site since 1973 (AGRA, 1995). (Prior to 1975, concentrations of chloride had been less than 5 mg/L.) In 1993, the B.C. Ministry of Transportation and Highways received an official complaint from the Municipality of Heffley Creek that groundwater for one of its two wells had been contaminated by road salts stored at the Heffley Creek patrol yard. Water quality tests indicated that chloride concentrations at this well were approximately 1000 mg/L. By 1994, there were elevated chloride concentrations at both municipal wells located between 175 and 200 m from the patrol yard (Figure 7). Chloride concentrations eventually peaked at approximately 3200 mg/L in 1995. While chloride concentrations have decreased since 1996, well #1 still had chloride concentrations higher than 500 mg/L in 1998 (AGRA, 1999).
In 1996, complaints were registered with the Nova Scotia Departments of Transportation and Public Works and the Environment regarding salt contamination of residential wells in Bible Hill, Nova Scotia. Tests undertaken at three wells that supplied six duplexes indicated that chloride and sodium concentrations ranged from 130 to 640 mg/L (Rushton, 1999). The Bible Hill patrol yard, which is located relatively close to these residences, was determined to be the source of salt contamination. While road salts are stored in a salt dome, abrasive mixtures have traditionally been stored outside. The Nova Scotia Department of Transportation and Public Works indicated that groundwater contamination was typical of locations where mixed abrasive/salt piles are left uncovered (Rushton, 1999).
Figure 7 Chloride concentrations in Heffley Creek municipal wells (from AGRA, 1999)

A study by Ohno (1990) conducted on wetlands near sand/salt storage sites in Maine showed that concentrations of chloride and sodium at the affected sites were 2-3 orders of magnitude higher than those of control sites. The concentrations at the control sites were 6 mg sodium/L and 8 mg chloride/L, while the concentrations at the affected sites ranged from 16 to 8663 mg sodium/L and from 14 to 12 463 mg chloride/L.
Pinhook Bog, LaPorte County, Indiana, was adversely impacted by contamination from a salt storage pile from the late 1960s to the early 1980s (Wilcox, 1982). Chloride concentrations at control sites in 1980 and 1981 were 5-6 mg/L. This compares with a maximum single daily reading for salt-impacted locations of 1468 mg chloride/L in 1979, 982 mg chloride/L in 1980 and 570 mg chloride/L in 1981. The total chloride inputs to the bog over the 10-year period when salt storage occurred were estimated as follows: 2.3 million kilograms from the salt pile, 0.4 million kilograms from road salting and 0.012 million kilograms from direct precipitation.
2.5 Environmental fate and pathways
Road salts can enter and move through the environment in their salt form or as their dissociated ions. In aquatic systems, chloride salts are present in dissociated form as chloride ions and corresponding cations (sodium, potassium, calcium, magnesium). The circulation of chloride through the hydrological cycle is due mostly to physical rather than chemical processes. The chloride ions pass readily though soil, enter groundwater and eventually drain into surface waters. Because chloride ions are persistent and are entrained in the hydrological cycle, all chloride ions applied to roadways as road salts or released from patrol yards or disposal sites can be expected to be ultimately found in surface water. This section presents information on the fate and transportation of road salts in the environment and their potential pathways.
2.5.1 Fate/transport
Once road salts enter surface waters, they remain in solution unless their concentrations are very high and exceed their solubility, when crystallization and subsequent sedimentation of mineral salts could occur. When water containing road salts percolates through soil, positively charged ions (i.e., sodium, calcium, magnesium, potassium) are attracted to and bond with the inherently negatively charged soil surfaces. The extent of bonding depends on the cation exchange capacity and the number of negatively charged sites in the soil. The sodium ion has a high solubility and may, once dissolved, remain in solution; however, since it is readily adsorbed onto soil particles, it is less probable that it will reach groundwater and surface waters. Nevertheless, in cases with limited adsorption or where extreme leaching from the soil occurs, the sodium ion will follow water pathways and eventually find its way to groundwater and surface waters (MDOT, 1993).
There are no major removal mechanisms, such as volatilization, degradation (photodegradation, biodegradation), sorption (to particulates) or oxidation, that would remove the salts from surface waters. Dilution resulting from mixing with low-salinity water would decrease the salt concentrations in the aqueous phase. Hence, the salts will always be present in the aqueous phase rather than in the particulate phase (suspended or bottom sediments). In the case of benthic sediments, salts may accumulate in sediment interstitial water (pore water).
Dissolved salts may alter the physical properties of water by changing its density. Density increases linearly with increasing salinity (Ruttner, 1963). The increases in the density resulting from salinity changes are large in comparison with density changes associated with temperature and have important implications for lakes, as inorganic salts can accumulate temporarily or permanently in deeper strata and prevent lake waters from mixing (Section 3.3.3).
The chemical and biochemical behaviour of major ions from four inorganic salts and the ferrocyanide anti-caking agent considered in this report are discussed below.
2.5.1.1 Chloride
Chloride is the principal contributing anion to salinity from application of road salts and is influential in general osmotic salinity balance (Wetzel, 1975; Hammer, 1977). Chloride is a highly soluble and mobile ion that does not volatilize or easily precipitate or adsorb onto surfaces of particulates. In freshwater ecosystems, chloride behaves conservatively (i.e., its concentration in water is not affected by chemical or biological reactions) (Wetzel, 1975; Pringle et al., 1981). Chloride levels in surface water will largely change only in response to the addition of chloride, dilution by precipitation or inflow, or concentration by evaporation of water. In the majority of inland surface waters, the concentrations of chloride and the respective cations rarely reach the solubility products of the respective chloride salts, and so precipitation rarely occurs.
2.5.1.2 Sodium and potassium
In surface waters, the monovalent cations sodium and potassium are relatively conservative in their chemical reactivity and low biotic requirements. Because of this, the spatial and temporal distributions of sodium and potassium in unimpacted freshwater systems are uniform, with little seasonal variation.
2.5.1.3 Calcium
Calcium is the most reactive ion of all the major cations contributing to salinity. Its concentrations in surface waters are affected by chemical and biological processes within the aquatic systems. It is required as a micronutrient for higher plants and is one of the basic inorganic elements of algae. Calcium concentrations in lakes with hard water undergo seasonal changes, with a decrease in calcium concentrations and total alkalinity in the summer, when biogenically induced decalcification removes calcium from the water column. Decreased concentrations of inorganic carbon in the epilimnion, resulting from increased rates of photosynthesis, are responsible for the precipitation of calcium with bicarbonate. Some of the precipitated calcium carbonate is resolubilized in the hypolimnion, and some is entrained in sediments. Decalcification of the trophogenic zone changes monovalent:divalent cation ratios, which has an effect on the distribution and dynamics of algae and larger aquatic plants in freshwater ecosystems (Wetzel, 1975).
2.5.1.4 Magnesium
In general, magnesium compounds are more soluble than other compounds. In hardwater systems, calcium carbonates precipitate before more soluble magnesium carbonates. Significant precipitation of magnesium carbonate and hydroxide occurs only at very high pH (>10). Magnesium is a micronutrient in enzymatic transformations of organisms. It is required by chlorophyll-bearing plants as the magnesium porphyrin of the chlorophyll molecules (Wetzel, 1975). However, the metabolic requirements for magnesium are small in comparison with its availability in freshwater systems.
2.5.1.5 Sodium ferrocyanide
Sodium ferrocyanide dissociates to yield sodium and ferrocyanide ions. In the ferrocyanide ion, the chemical bond between the cyanide group and the iron is very strong; in water, however, it can be photolysed to release cyanide ions (Hsu, 1984). Laboratory tests determined that a 15.5 mg/L solution of ferrocyanide would produce 3.8 mg cyanide/L upon exposure to sunlight for 30 minutes (U.S. EPA, 1971).
2.5.2 Environmental pathways
The transport pathway for road salts applied to roadways is illustrated in Figure 8. The following sections explain the different environmental compartments that can be affected by release of road salts via roadway applications, snow disposal sites and salt storage sites (patrol yards).
2.5.2.1 Roadway applications
Road salts applied to roadways can enter surface water from:
- salt-laden runoff from roadways into drainage ditches, watercourses or wetlands soon after application;
- salt-contaminated meltwater from roadway surfaces or from snow plowed to roadsides after application of salts; and
- the release of salts stored in surface soils (Scott, 1980).
Factors affecting the degree of roadway runoff into aquatic ecosystems include a) the length of major road treated and drained, b) the amount of salts applied prior to the thaw period, c) road drainage pattern and topography, d) level of discharge of the receiving stream, e) degree of urbanization, f) rate of rise and duration of temperatures above freezing and g) precipitation (Scott, 1981).
Road salts applied to roadways can enter soils and groundwater from meltwater runoff from roadways and wet and dry deposition of airborne salt.
Road salts applied to roadways can enter air from windborne vehicle spray or splash and windborne dry powder from residue on roads. Windborne vehicle spray or splash can be affected notably by vehicle speed, wind, road gradient and geometrical features of the road (McBean and Al-Nassri, 1987).
2.5.2.2 Snow disposal sites
Road salts released through snow disposal will enter surface water from:
- direct release when dumping into surface waters, releasing to untreated storm sewers or melting following disposal on banks of water bodies;
- indirect releases via wastewater treatment plants in the case of dumping to treated storm sewers or of surface disposal sites draining to a treated water system;
- overland runoff of stormwater and snowmelt water; and
- stormwater pipage, which drains part of the disposal area to a surface water body.
Road salts released through snow disposal will enter soil and groundwater from direct percolation of precipitation and meltwater from snow disposal sites.
Figure 8 Road salts transport pathway
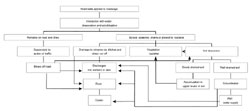
2.5.2.3 Storage sites/patrol yards
Road salts released from storage sites or patrol yards will enter surface water from:
- salt-laden leachate from unprotected material piles;
- overland runoff of stormwater that has dissolved salts and of snowmelt water; and
- vehicle washwater.
Road salts released from storage sites or patrol yards will enter groundwater from:
- groundwater recharge from salt-laden soil and surface water;
- releases from vehicle washwater where the effluent is discharged to a dry well; and
- entry through wells or around well casings.
Road salts released from storage sites or patrol yards will enter soil from:
- spillage of salts at storage sheds during stockpiling or transferring of salts or blended abrasives/salts;
- spillage of salts or blended abrasives/salts during loading and unloading of maintenance vehicles;
- spillage of salts in the remainder of the yard as maintenance vehicles drive through;
- wind-driven erosion of salts from piles and from uncovered winter maintenance vehicles;
- salt-laden leachate from unprotected salt and blended abrasive/salt piles;
- precipitation or snowmelt waters, which dissolve salts and infiltrate into the soil; and
- release from vehicle washwater discharging onto the soil.
2.6 Chloride concentrations in surface waters across Canada
This section presents information on the environmental concentrations of constituents of road salts in Canadian surface waters and presents estimates of chloride concentrations in surface waters across Canada based on mass balance modelling methods. Information in this section is summarized in Mayer et al. (1999) and Evans and Frick (2001) and is based on a review of numerous agency reports, scientific publications and monitoring surveys.
The surface water ecosystems covered include running (lotic) waters, such as rivers and creeks, and standing (lentic) waters, which include lakes, ponds and wetlands. Both the pelagic and the benthic compartments of these ecosystems are discussed. Also presented is a review of background concentrations and concentrations for water bodies that have been impacted by the release of road salts. Examples of water bodies that have been impacted by the storage of road salts were included in the section on patrol yards (see Section 2.4). The data suggest that the surface waters most sensitive to road salt impacts are low-dilution environments such as wetlands, small urban lakes and ponds with long residence times and small streams draining large urbanized areas.
2.6.1 Mapping of chloride concentrations in watersheds across Canada
Concentrations of chloride in water across Canada were mapped by watershed. Observed chloride concentrations in Canadian watersheds are provided in Figure 9. The concentrations of chloride in surface water are based on mean values obtained from federal and provincial water quality monitoring stations in Canada. The methods and database used in generating the map are provided in Mayer et al. (1999). Basically, monitoring data from near the mouth of each watershed are combined to provide estimates of the concentration for the entire watershed. The amount of data are varied, ranging from many stations and a monthly sampling interval to intermittent spot samples. Accordingly, data in Figure 9 should be used as an indication of the relative variations in the concentrations of chloride across Canada.
Watersheds containing major urban areas and areas of intense road salt loadings in southern Quebec, Ontario and the Maritime provinces have elevated chloride concentrations. Note that these concentrations represent averages over a watershed and, as such, do not indicate actual exposure concentrations of a specific watercourse. The map also indicates that some watersheds in the Prairie provinces have elevated ambient concentrations. These concentrations are related to the arid climate of the Prairies and are associated with the saline lakes and rivers situated in this region.
Road salts are not the only input that influences the concentrations in Figure 9. Effluents from various industries (e.g., pulp and paper mills) and releases from private septic systems and municipal wastewater treatment plants can influence chloride concentrations. It has been estimated for the lower Great Lakes that road salts contribute approximately 20% of the chloride load, with natural and other anthropogenic sources contributing the balance (Jones et al., 1986).
2.6.2 Environmental concentrations: Lakes and rivers
In the absence of anthropogenic sources, surface waters acquire their characteristics by dissolution and chemical reactions with solids, liquids and gases with which they come into contact during various phases of the hydrological cycle (Stumm and Morgan, 1981). For instance, stream water chemistry can be related to the chemistry of the source rock and to equilibria controlling the formation of solid phases (Feth et al., 1964; Garrels and Mackenzie, 1967). Watershed bedrock and surficial geology play a dominant role in determining the salt concentrations in surface waters. In addition, the climate and the proximity of the surface waters to the sea will be important in regulating the background salt concentrations. Thus, the background salt concentrations (salinity) of surface waters vary.
Surface waters range from fresh waters, with salinities lower than 500 mg/L, to saline waters, with salinities equal to or greater than 3000 mg/L (Hammer, 1986). While enclosed lakes and ponds may become saline, streams and rivers will rarely have natural salinity high enough to be classified as saline.
For the purpose of this Assessment Report, surface water quality is discussed by geographic region rather than by hydrogeological region in order to better address the effects of human factors on surface water quality. Both naturally occurring concentrations and those impacted by human activity are presented. While anthropogenic sources such as road salts, sewage and industrial effluents can all increase salinity, the main emphasis for data on impacted areas relates to the releases of road salts.
2.6.2.1 Atlantic region (Newfoundland, Nova Scotia, New Brunswick and Prince Edward Island)
The median concentrations of chloride and calcium from recent surveys (1985 and later) were compiled by Jeffries (1997). The data set includes 38 lakes from Labrador, 63 lakes from Newfoundland, 150 lakes from Nova Scotia and 166 lakes from New Brunswick. The median chloride and calcium concentrations in lakes in largely unimpacted areas ranged between 0.3 and 4.5 mg/L and between 0.82 and 1.10 mg/L, respectively.
Comprehensive studies by Kerekes et al. (1989), Kerekes and Freedman (1989) and Freedman et al. (1989) at Kejimkujik National Park, Nova Scotia, investigated the water quality of several lakes and rivers in the park. These studies concluded that sea salt influence is significant in Nova Scotia. The concentration ranges were 3.6-5.4 mg/L for chloride, 2.3-3.4 mg/L for sodium and 0.36-0.96 mg/L for calcium. The levels of these ions were comparable with those measured in other surface waters in the Atlantic region (Jeffries, 1997).
Figure 9 Observed chloride concentrations in Canadian watersheds (from Mayer et al., 1999)

Studies on the Chain Lakes near Halifax indicate that winter de-icing activities can have an impact on water quality. Thirmurthi and Tan (1978) explained that, from June to September 1975, the chloride concentration was approximately 19 mg/L. Concentrations began to increase in November and eventually peaked in May 1976, at 43 mg chloride/L. The chloride concentration then dropped rapidly through the rest of May and June, decreasing to 19 mg/L in July. The increase in chloride concentrations for both lakes represents an approximate addition of 19 440 kg of chloride to First Chain Lake and 7920 kg to Second Chain Lake. The Chain Lakes continued to be studied through the early 1980s (Hart, 1985, 1988). Chloride concentrations in both lakes increased gradually from 1976 to reach a maximum in the mid-1980s of approximately 120 mg/L in First Chain Lake and about 170 mg/L in Second Chain Lake. Highest chloride concentrations were observed during spring runoff.
Road salts have impacted Chocolate Lake in Nova Scotia. The lake is small (82.7 ha) and shallow (mean depth 3.9 m, maximum depth 12.2 m), with an estimated volume of 350 000 m3 (Kelly et al., 1976). The watershed is large (900 ha), approximately 11 times as great as the lake surface area. Chloride concentrations were highly elevated in the lake between April and August 1975. Average summer chloride concentrations were 207.5 mg/L compared with an estimated background value of 15-20 mg/L for non-impacted lakes. Chloride concentrations varied with depth, ranging from 199 to 224 mg/L at the surface, from 189 to 217 mg/L at 6.1 m, and from 225 to 330 mg/L at 12.1 m. Sodium concentrations ranged from 108 to 125 mg/L, from 102 to 125 mg/L and from 116 to 183 mg/L at the same depths. The gradient in salt concentrations and the absolute concentration of salt in deep waters were large enough to prevent complete vernal vertical mixing of the water column; deep waters became anoxic in summer. The primary source of chloride was attributed to highway runoff containing road salts. Kelly et al. (1976) estimated that 58.4 tonnes of road salt (representing 35 409 kg of chloride and 22 737 kg of sodium) were applied to the Chocolate Lake drainage basin during the winter of 1974-75.
A study on the water quality of 50 lakes in the Halifax/Dartmouth metropolitan area noted that most lakes experienced a very pronounced increase in the concentrations of sodium and chloride between 1980 and 1991 (Keizer et al., 1993). Of the lakes sampled in 1980, 12 had chloride concentrations greater than 50 mg/L and 3 had concentrations greater than 100 mg/L (the maximum concentration was 125 mg/L). In 1991, 22 of the 50 lakes sampled had chloride concentrations greater than 50 mg/L, 12 had concentrations greater than 100 mg/L and 4 had concentrations greater than 150 mg/L (the maximum concentration was 197 mg/L). The concentrations of sodium in these lakes showed similar trends. Of the lakes sampled in 1980, 9 lakes had sodium concentrations greater than 40 mg/L and 3 had concentrations greater than 60 mg/L (the maximum concentration of sodium was 73 mg/L). In 1991, 21 lakes had sodium concentrations greater than 40 mg/L, 12 had concentrations greater than 60 mg/L and 7 had concentrations greater than 80 mg/L (the maximum concentration was 114 mg/L). It is noteworthy that sampling was at the surface in the middle of the lakes and may not be characteristic of bays, where higher concentrations may occur. Furthermore, since denser, salt-laden water generally sinks to the bottom, these samples may be a low estimate of the overall lake concentrations.
Keizer et al. (1993) found that lakes in more developed watersheds had higher chloride and sodium concentrations than lakes in rural areas. In addition, salt concentrations from lakes in less developed areas did not increase markedly between the two sampling periods, unlike lakes in urban areas. For example, 19 of the rural lakes sampled had chloride concentrations below 25 mg/L in both 1980 and 1991; 23 of the lakes sampled had sodium concentrations below 20 mg/L in 1980 and 1991. Three of the lakes with the lowest concentrations had chloride concentrations of 5.1, 3.9 and 5 mg/L in 1980 and 4.5, 3.4 and 5 mg/L in 1991. Sodium concentrations were 3.4, 2.5 and 3.2 mg/L in 1980 and 2.9, 2.2 and 2.9 in 1991.
In general, the chloride concentrations in unimpacted water bodies in the Atlantic region do not exceed a concentration of 20 mg/L. Lower concentrations (<10 mg/L) are generally observed in inland water bodies, while the higher concentrations occur in coastal areas.
2.6.2.2 Central region (Ontario and Quebec)
The concentrations of chloride, sodium and calcium in lakes and rivers in the Canadian Shield, which covers a large area of Quebec and Ontario, including the upper Great Lakes, are very low (e.g., 1-10 mg chloride/L) (Jeffries, 1997). According to Weiler and Chawla (1969), the average annual concentrations of chloride in lakes Erie and Ontario were 24.6 and 27.5 mg/L, respectively, while the corresponding chloride concentration in Lake Superior was only 1.3 mg/L. Earlier studies, which investigated water quality trends in the Great Lakes, reported a rapid increase in chloride concentrations related to human activities (Kramer, 1964; Beeton, 1965; Dobson, 1967). The largest increase was observed for Lake Ontario. Later data (Williams et al., 1998), however, showed a decline in chloride concentrations from about 22 mg/L in 1977 to about 16 mg/L in recent years (1985-1993). The decline in the lower Great Lakes can probably be ascribed to lower loadings from industrial and domestic sources, resulting from improved control/treatment of industrial and domestic effluents. The tributaries to Lake St. Clair (e.g., Sydenham and Thames rivers) have somewhat higher chloride concentrations (about 30-40 mg/L) than other southern Ontario rivers because they drain the Paleozoic sedimentary rock adjacent to Michigan Basin, which contains saline formations such as salt (sodium chloride) and anhydrite (calcium sulphate) bearing strata. These strata also contribute to salt (sodium chloride and calcium sulphate) inputs to Lake St. Clair and western Lake Erie.
In urbanized areas, application of de-icing salts contributes significantly to chloride inputs. Long-term monitoring locations in the Greater Toronto Area show a gradual increase in chloride concentrations. In Highland Creek, a highly degraded urban watercourse, the statistical trend analysis shows an increase in chloride concentrations from 150 mg/L in 1972 (presumably already impacted) to over 250 mg/L by 1995 (Bowen and Hinton, 1998). In Duffin Creek, the median chloride concentrations increased from 10-20 mg/L in the 1960s to 30-40 mg/L in the early 1990s (Bowen and Hinton, 1998). In urban streams and rivers, such as the Don River in Toronto, concentrations of chloride increased to more than 1000 mg/L in winter (Scott, 1980; Schroeder and Solomon, 1998), with increases appearing to coincide with thaws. The autumn baseline values in the Don River were 100-150 mg chloride/L. Similar baseline concentrations of chloride (50-100 mg/L) were reported in another Toronto watercourse, Black Creek (Scott, 1980). Chloride concentrations 50 times higher than baseline values were measured during the thaw periods in this creek. Crowther and Hynes (1977) reported high winter concentrations of chloride (1770 mg/L), sodium (9550 mg/L) and calcium (4890 mg/L) in Laurel Creek, which passes through urban Waterloo. Rodgers (1999) observed high concentrations of chloride (4355 mg/L) in Red Hill Creek in Hamilton, Ontario.
The Ontario Ministry of Transportation has monitored water quality in a number of streams in Ontario, including Toronto-area watersheds, between 1990 and 1996. Results for four watercourses in urban areas are shown in Table 9. Highest concentrations were recorded during the winter months, indicating peak inputs of road salts. Chloride concentrations frequently exceeded 250 mg/L and often exceeded 500 mg/L (Table 9).
Little Round Lake in central Ontario was affected by cultural disturbances that included urbanization, septic tanks, highway road salt runoff and seepage from a salt storage depot (Smol et al., 1983). This small lake (7.4 ha, maximum depth 16.8 m) became meromictic. Meromictic conditions were attributed by Smol et al. (1983) to input of road salts from runoff and storage. Salt concentrations in the monimolimnion or deep layer were 58.4 mg sodium/L and 103.7 mg chloride/L, well in excess of that explainable by the natural geology of the region. Road salt additions apparently had resulted in the formation of meromixis in the 30 years prior to the study in 1981.
The Ministère des Transports du Québec (1980,1999) investigated the impacts of road salt on Lac à la Truite, near Sainte-Agathe-des-Monts. The lake drainage area, estimated at 728 ha, was affected by a 7-km stretch of highway (approximately 30 lane-kilometres). At certain places, the highway was located as close as 250 m from the lake. The lake has a surface area of 48.6 ha, a mean depth of 21.5 m and an estimated volume of 486 000 m3. In 1972, the average chloride concentration for the lake was 12 mg/L. This increased through the 1970s to reach a maximum concentration of 150 mg/L in 1979. This corresponds to an addition of 67 068 kg of chloride to the lake. To prevent further increases, abrasives (containing some salt to prevent caking) were used instead of road salts. Chloride concentrations fell through the 1980s to reach 45 mg/L in 1990 and have remained at that level since.
Table 9 Chloride concentrations in various streams in the Toronto Remedial Action Plan watershed for 1990-96 (from Toronto and Region Conservation Authority, 1998)
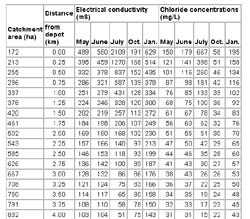
In the United States, significant increases in chloride concentrations observed in four Adirondack streams were attributed to winter salt application (Demers and Sage, 1990). There was a significant difference in chloride concentrations between samples upstream and downstream from roadways. Demers (1992) reported that chloride concentrations were as much as 66 times higher in downstream samples. The overall mean chloride level was 0.61 mg/L upstream and 5.23 mg/L downstream. The terrain characteristics of Adirondack streams (New Hampshire) are similar to those of streams in the Eastern Townships of Quebec. Chloride concentrations of 11 000 mg/L (Hawkins and Judd, 1972) were measured in winter of 1969 in Meadowbrook, which drains about 10 km2 of suburbs in Syracuse, New York (Hawkins and Judd, 1972). Also in the United States, chloride concentrations of about 400 mg/L were measured in the bottom waters of Irondequoit Bay, a small bay on Lake Ontario near Rochester, New York (Bubeck et al., 1971, 1995).
In summary, the chloride concentrations of unimpacted water bodies in the Shield region of Central Canada are among the lowest in the country and vary within the range <1-5 mg/L. Higher background concentrations of up to 10-30 mg/L are observed in water bodies situated within the St. Lawrence Lowlands region, which includes the basin of the lower Great Lakes and the St. Lawrence River. A marked increase in chloride concentrations above background levels is particularly noticeable in many small urban lakes and watercourses in this region, as it is the most densely populated area of Canada. Seasonal and spatial trends indicate the important contribution of road salts to these increases.
2.6.2.3 Prairie region (Manitoba, Saskatchewan and Alberta)
Most naturally saline lakes in Canada are located within two regions. The first region encompasses several endorheic drainage basins on the Canadian Prairies (Hammer, 1984). The area where saline lakes are most numerous stretches over southern Alberta and Saskatchewan. The Alberta-Saskatchewan saline lake region is largely underlain by Cretaceous bedrock, composed mainly of shales, silts and sandstones (Hammer, 1984). Of the Prairie provinces, Saskatchewan has by far the greatest number and volume of saline lakes (Hammer, 1986). The relative proportions of cations in most Prairie lakes are as follows: Na > Mg > Ca. Of the lakes investigated, there are only two known meromictic lakes on the Canadian Prairies, Waldsea Lake and Deadmoose Lake (Hammer, 1984). Numerous potholes (shallow lakes or ponds), which range in salinity from fresh to saline, can be found in the southern Prairies.
There are fewer naturally saline lakes (about 30) in Alberta (Hammer, 1984). Most of them are concentrated in the Provost and Hanna regions, although they occur as far north as Edmonton. Even fewer saline lakes are in Manitoba. Barica (1978) investigated a group of over 100 small potholes, which varied in salinity from fresh to moderately saline, near Erickson in southwestern Manitoba. The concentration ranges were 1-448 mg/L for chlorine, 0.8-1075 mg/L for sodium and 27-380 mg/L for calcium. Twenty-three sodium chloride-dominated sites along the western shore of Lake Winnipegosis were studied by McKillop et al. (1992). The concentration ranges for chloride, sodium and calcium were 861-33 750 mg/L, 587-21 313 mg/L and 59-1400 mg/L, respectively.
Much lower concentrations of chloride and calcium are reported for 27 and 193 lakes in northern portions of Saskatchewan and Alberta, respectively (Jeffries, 1997). The median concentrations of chloride were 0.2 and 0.5 mg/L, respectively. The calcium concentrations of Saskatchewan and Alberta lakes were 2.0 and 14.0 mg/L, respectively. The chloride and calcium concentrations of 26 lakes in Manitoba were 2.0 and 7.5 mg/L, respectively (Jeffries, 1997).
In summary, many of the saline water bodies in Canada are located within the Interior Plains, which cover the southern portions of the Prairie provinces. In general, dissolved salt concentrations, including chloride, are higher in this geographical region because of the underlying geology. However, low concentrations of chloride (<5 mg/L) are reported for unimpacted water bodies situated in the northern region of these provinces, outside of the Interior Plains region.
2.6.2.4 Pacific region (British Columbia)
The second region in Canada where a large number of naturally saline lakes are located is the Southern Interior Plateau (Fraser Plateau) of British Columbia, in the rain shadow of the Coast Mountains. Saline lakes in British Columbia are small and shallow (Topping and Scudder, 1977). The limnology of four of these lakes was described by Northcote and Halsey (1969), who showed not only that these lakes differed in total dissolved solids concentrations among themselves, but also that there were differences within the lakes (surface to near-bottom). Concentrations ranging from 5.1 to 800 mg/L were reported. The meromixis in three of these lakes was maintained by chemical density gradients, while in the fourth, the mixing was inhibited by morphometric features. In non-saline regions, the reported median chloride and calcium concentrations of six acid-sensitive lakes located in the southwest of the province were 2.5 and 3.7 mg/L, respectively (Phippen et al., 1996; Jeffries, 1997).
The impact of road salts on water quality was monitored for the Serpentine River in British Columbia's Lower Fraser Valley. Electronic data recorders monitored water temperature, pH, water level and conductivity every 15 minutes. Three-fold increases in conductivity were observed over 10- to 20-hour periods during thaw periods following a cold period when roads were sanded and salted (Whitfield and Wade, 1992). Furthermore, periods of elevated conductivity were followed by increased water levels, which are characteristic of snowmelt. Results of this study indicate that organisms living in certain streams during winter months may be subject to fluctuations in salt concentrations.
In summary, there are several lakes located in the Southern Interior Plateau of British Columbia whose chloride concentrations are higher than 100 mg/L, and several of these lakes are naturally saline. The chloride concentrations of unimpacted water bodies in the remaining parts of British Columbia are generally less than 5 mg/L.
2.6.2.5 Yukon, Northwest Territories and Nunavut
Few data were identified from the Yukon, Northwest Territories and Nunavut.
2.6.3 Environmental concentrations: Benthic sediments
Because inorganic chloride salts used for road maintenance are highly soluble and interactions with sediment particulates are minimal, inorganic salts are expected to accumulate in sediment pore water rather than in the solid phase. The study by Mayer et al. (1999) of an urban pond (Rouge River Pond) shows that the sediment pore water, which is in equilibrium with the overlying water, enriched in inorganic salts, may attain high salt concentrations (see Section 2.6.5). High concentrations of salts in sediment pore water can result not only in osmotic stress and direct toxic effects on benthic biota but also in complexation of metals with chloride, augmenting the concentrations of dissolved heavy metals (e.g., cadmium), which are toxic to benthic organisms.
2.6.4 Environmental concentrations: Wetlands
Wetlands are an important landscape feature in Canada. Their most distinguishing features are presence of standing water, unique wetland soils and vegetation adapted to or tolerant of saturated soils (Mitsch and Gosselink, 1986). Canada has a great variety of wetlands; detailed descriptions of various types of wetlands can be found in Mitsch and Gosselink (1986).
There is only limited information on the chemical characteristics of coastal and inland freshwater wetlands with respect to chloride, for much of the work on wetlands is concerned with nutrient cycling, acidification and geochemistry of metals. A 1987 study of wetlands within Kejimkujik National Park (Wood and Rubec, 1989) assessed the chemical characteristics of wetlands. The authors reported significant differences in peat chemistry between the bogs and fens, in particular in sodium and calcium concentrations. The concentrations of calcium, sodium and chloride in peat from bogs were 0.17, 0.8 and 0.15 mg/L, respectively. The concentrations of calcium, sodium and chloride in peat from fens were 0.32, 1.5 and 0.12 mg/L. There was no difference in the surface water chemistry between bog and fen. The concentrations of calcium, sodium and chloride in water of bogs were 6 x 10-4, 3.0 x 10-3 and 3.5 x 10-3 mg/L, respectively, and the concentrations of calcium, sodium and chloride in water of fens were 6 x 10-4, 2.8 x 10-3 and 3.9 x 10-3 mg/L, respectively.
Bourbonniere (1985, 1998) investigated the geochemistry of wetlands in Nova Scotia and Ontario. He measured the major ion chemistry in pore water of the ombrotrophic bog in Barrington County in Nova Scotia. The measured concentrations were 9.6-24 mg/L for chlorine, 5.2-12.8 mg/L for sodium and 0.47-1.5 mg/L for calcium. The concentrations of calcium, sodium and chlorine in Beverly Swamp, Ontario, were 58.1-95.3 mg/L, 5.1-20.0 mg/L and 8.7-41.2 mg/L, respectively. The water composition of potholes described in Section 2.6.3.3 may be considered to be characteristic of the prairie wetlands. No Canadian studies were found that address the impact of road salt application on the chemistry of wetlands.
Two studies were presented in the patrol yard section (Section 2.4) that focus on the impact of salt depots on wetlands.
2.6.5 Environmental concentrations: Urban lakes and ponds
Natural small urban lakes and ponds that receive road runoff are susceptible to changes in ion concentrations. There are only limited data on the chloride concentrations in such bodies. In the absence of such data, ponds that are part of an urban stormwater management system can be used as a reasonable surrogate. Numerous ponds have been constructed and remain a widely used form of watershed management in urban areas. Although these ponds are engineered water bodies designed primarily for hydraulic flow management and stormwater treatment, they have developed aquatic ecology similar to natural small urban ponds. Like natural urban aquatic systems, these ponds are wetland habitats for many species of flora and fauna. They are likely candidates for showing maximum chloride concentrations, since they represent the type of water body that receives the maximum exposure to chloride loadings from winter maintenance activities.
A study of Lake Wabukayne, a small human-made lake in Mississauga, Ontario, reported chloride concentrations between 200 and 2000 mg/L and temporary meromixis during the winter and early spring (Free and Mulamoottil, 1983). Chloride concentrations of 1100-2000 mg/L (Vickers, 2000) were measured in the Harding Pond in Richmond Hill in the Greater Toronto Area. Seasonal surveys of the stormwater retention ponds in the Greater Toronto Area (Mayer et al., 1996; Mayer, 2000) showed higher concentrations of chloride during the winter months. Chloride concentrations of 380 and 800 mg/L, respectively, were measured in the Heritage and Unionville ponds, which are situated in residential settings (Mayer, 2000), while chloride concentrations as high as 5910 mg/L were measured in the Col. S. Smith Reservoir, receiving runoff from a multi-lane highway (Queen Elizabeth Way) in February of 1999.
Both a mass balance study and a sediment study were performed on the Rouge River Pond, situated in the Rouge River Valley, Scarborough, Ontario, near Highway 401 and Port Union Road. This pond is an urban stormwater management facility designed to treat highway stormwater by removing 70% of suspended solids. The mass balance study was conducted to measure the pollutant removal and the dynamics of several water quality constituents between 1995 and 1998 (Liang, 1998). The initial studies during 1995, 1996 and 1997 focused on balances during the ice-free season. A chloride imbalance related to permanent meromictic conditions in the pond was shown, with meromixis likely due to a combination of the pond design and high road salt loadings. The salt concentrations calculated as sodium chloride in the bottom layer of the pond were of the order of 5000 mg/L. Elevated concentrations of metals and ammonia were also measured in this bottom layer. The sediment study (Mayer et al., 1999) investigated the chemistry and toxicity of sediment pore water and assessed the solid-phase metal chemistry. A geochemical model was formulated to assess the impact of salts on metal speciation. The investigation revealed that sediment pore water, which is in equilibrium with the overlying water, enriched in inorganic salts, may itself attain high salt concentrations. At the sediment-water interface, concentrations of chloride and sodium greater than 3000 mg/L and 2000 mg/L, respectively, were measured in the pore water. A gradual decrease with depth in sediment was observed for both ions; however, even at a depth of 40 cm below the sediment-water interface, the chloride concentrations were about 1500 mg/L.
A study by Bishop et al. (2000) focused on the presence of contaminants in six stormwater retention ponds in the Greater Toronto Area and nine stormwater retention ponds in Guelph, Ontario. Of the 15 ponds in this study, 4 ponds in Guelph were sampled between 19 and 21 times each to determine the concentration of chloride in stormwater. These samples were taken at different times during the months of September, October and November 1997 and April, May, June, July, August, September and November 1998. Samples were also taken as close to the ponds' outfalls as possible (Struger, 2000). Mean concentrations of chloride in stormwater samples from these four ponds were between 120 and 282 mg/L; the maximum concentrations observed at the four ponds were between 416 and 1230 mg/L (Struger, 2000). While samples were not taken from December to March, the average chloride concentrations of samples for November, April and May were higher than the average chloride concentrations of samples for the other months at each of the ponds (336 vs. 153 mg/L; 183 vs. 79 mg/L; 469 vs. 82 mg/L; 444 vs. 188 mg/L).
The samples were taken close to the ponds' outfalls because these concentrations are probably indicative of the concentrations entering receiving environments. The stormwater retention ponds treat suspended solids and other attached constituents, but do not remove dissolved salts from runoff (OME, 1991; Bishop et al., 2000). Depending on the volume of runoff entering these ponds, they are typically designed to retain runoff for a period ranging between 24 and 72 hours; retention time may be as low as 10 hours during peak periods of large events.
Watson (2000) sampled the quality of water in 89 ponds located near roadways in southern Ontario. Multiple samples were taken during spring and summer of 2000, and the ponds were categorized according to the number of lanes of the nearest road. The mean concentration of chloride in ponds located near two-lane roads was 95 mg/L (range 0-368 mg/L). The mean concentration of chloride in ponds located near roads with more than two lanes but fewer than six lanes was 124 mg/L (range 0-620 mg/L). The mean concentration of chloride in ponds located near roads with six lanes or more was 952 mg/L (range 49-3950 mg/L). Watson (2000) does not provide an indication of background concentrations, but indicates that the main source of chlorides would be from road salts.
2.6.6 Chloride concentrations based on mass balance calculations
2.6.6.1 Calculation of watershed chloride concentrations resulting from the use of road salts
A mass balance model was used to estimate chloride concentrations in Canadian watersheds from the use of road salts. The calculated chloride concentrations were compared with the observed concentrations presented in Section 2.6.1. More details of this comparison and the analysis are presented in Mayer et al. (1999).
While there are some simplifications inherent to the model used (e.g., assumption of similar road salt application and volume of runoff from year to year), the model provides a reasonable estimate of the potential chloride concentrations resulting from road salt use. The model is also useful for identifying the relative concentrations of chlorides in the different watersheds. Sub-basin watersheds are used in the calculations; a more accurate comparison of calculated chloride concentrations would have been obtained if finer-scale resolution watershed maps had been used.
The results are presented in Figure 10. The Island of Montréal has the highest modelled concentration of chloride in runoff. Figure 10 also indicates that watersheds on the north shore of Lake Ontario, notably those largely influenced by municipalities in the Toronto area, also have high chloride concentrations related to road salts. The watersheds in urban areas, particularly those in southern Ontario, southern Quebec, the Maritime provinces and larger urban centres in the Prairie provinces, have the highest modelled concentrations of chloride in runoff.
The results are presented in Figure 10. The Island of Montréal has the highest modelled concentration of chloride in runoff. Figure 10 also indicates that watersheds on the north shore of Lake Ontario, notably those largely influenced by municipalities in the Toronto area, also have high chloride concentrations related to road salts. The watersheds in urban areas, particularly those in southern Ontario, southern Quebec, the Maritime provinces and larger urban centres in the Prairie provinces, have the highest modelled concentrations of chloride in runoff.
It is noteworthy that the concentrations presented in Figure 10 are modelled averages for watersheds. Accordingly, the concentrations do not indicate actual exposure concentrations for specific watercourses or water bodies. Data in Figure 10 can be used to identify areas that face the greatest risk of adverse effects from the use of road salts.
Figure 10 Estimated road salt chloride concentrations by watershed, calculated from average annual road salt loadings and average annual runoff (from Mayer et al., 1999)

2.6.6.2 Calculation of chloride concentrations in roadway runoff
The previous section assessed chloride concentrations caused by road salts for entire watersheds. At a watershed scale, modelled concentrations are diluted by runoff from areas other than just the roadway. This section estimates the concentration of chlorides at a roadway scale, using a mass balance approach.
For these estimates, it was assumed that the mass of salt used per two-lane-kilometre of provincially maintained road was dissolved and diluted by the volume of average annual runoff that would accumulate on the roadway surface (i.e., in 7400 m2, based on an assumed width of a two-lane kilometre of 7.4 m).
Figure 11 presents estimated concentrations of chloride and the length of provincial roads associated with these concentrations. Concentrations of chloride range between 30 and 31 000 mg/L. Based on these estimates, the majority of the concentrations (85%) range between 1000 and 10 000 mg/L. These estimates are of the concentrations of chloride in runoff from roadways; various application factors can be used to account for dilution in receiving waters. The accuracy of results depends on the quality of data used to calculate concentrations. One limitation is that estimates of average annual runoff were derived using a map at a scale of 1:7 500 000. More detailed maps (i.e., 1:50 000) might result in slightly different results. Furthermore, estimates of average annual runoff were used. Different values for runoff may have been calculated if runoff associated with winter months or spring thaw had been used.
Figure 11 Chloride concentrations in runoff for provincial roads in Canada

The concentrations of chloride observed in a recent study (Mayer et al., 1998) of highway runoff from three sites in southern Ontario are consistent with the above estimated chloride concentrations. The study reported chloride concentrations in runoff from roadways at three sites: a) the Burlington Skyway Bridge (a four-lane road with 92 000 cars per day), b) Highway 2 east of Brantford (a two-lane road with 31 100 cars per day) and c) Plains Road in Burlington (a two-lane roadway with 15 460 cars per day). Highest chloride concentrations (up to 19 135 mg/L) were observed in winter runoff from the Skyway Bridge, although high concentrations were also observed at Plains Road. The study, which investigated runoff toxicity, showed that salt-laden runoff has the potential to adversely affect aquatic organisms in low-dilution aquatic systems. The high chloride concentrations reported in this study are representative of concentrations at point of discharge and represent the worst-case scenario, which may be found along roadside ditches or small wetlands adjacent to major roadways.
2.6.7 Summary
Data show a broad range of background chloride concentrations in Canadian surface waters. Differences stem from the variance in bedrock and surficial geology, climate and proximity to the sea. Impacts of high use of road salts are evident from the data, particularly in highly urbanized areas. Water bodies most sensitive to the releases of road salts are low-dilution environments, such as small urban lakes and ponds with long residence times, urban stormwater ponds with short residence times, streams draining large urbanized areas and wetlands adjacent to major roadways.
In terms of natural concentrations across Canada, the following regional trends have evolved:
- In general, the chloride concentrations in unimpacted water bodies in the Atlantic provinces do not exceed about 20 mg/L. Lower concentrations (<10 mg/L) are generally observed in inland water bodies.
- Chloride concentrations of unimpacted water bodies in the Canadian Shield region of Central Canada are among the lowest in the country and vary within the range <1-5 mg/L. A marked increase in chloride concentrations is noticeable in many small urban lakes and streams.
- Many of the naturally saline water bodies in Canada are in the southern portions of the Prairie
provinces. However, naturally low concentrations of chloride (<5 mg/L) are reported for unimpacted water bodies in the northern region of these provinces. - There are several naturally saline lakes located in the Southern Interior Plateau of British Columbia (chloride concentrations higher than 100 mg/L). The chloride concentrations of unimpacted water bodies are generally less than 5 mg/L.
A spatial analysis of chloride concentrations was done based on two approaches. The first aggregated monitoring data to a watershed scale; the second used a mass balance analysis to model chloride concentrations at a watershed level. Mass balance techniques were also used to compute average annual concentrations in runoff from specific highways across Canada. The calculated concentrations range up to 31 000 mg/L, but mainly lie between 1000 and 10 000 mg/L. These estimates are consistent with measured undiluted runoff from roadways.
Elevated chloride concentrations were also measured in ponds and wetlands adjacent to roadways. Concentrations up to 4300 mg/L were observed in watercourses, and concentrations between 150 and 300 mg/L were observed in rural lakes that had been impacted by the use of road salts. While highest concentrations are usually associated with winter or spring thaws, high concentrations can also be measured in the summer, as a result of the travel time of the ions to surface waters and the reduced water flows in the summer.
3.0 Summary of Critical Information and Assessment of "Toxic" under CEPA 1999 (Continued)
3.1 CEPA 1999 64(a): Environment and CEPA 1999 64(b): Environment on which life depends
The approach for the environmental assessment of PSL substances involves characterizing the entry of, exposure to and effects of a substance, which ultimately permits the characterization of the risk posed by the substance (Environment Canada, 1997a). This may involve the use of both laboratory and field-derived data. Empirical field evidence linking environmental changes to entry of the substance can provide strong indications of actual risk.
It can be assumed that all road salts eventually enter the environment, whether from losses from storage or patrol yards, from roadway application or from disposal of snow. Although all chloride ions will ultimately be found in surface waters, ions from inorganic chloride road salts and ferrocyanides will be released to or move through soil, groundwater, surface water and air, and organisms can be exposed to road salts through all these media. Therefore, all environmental compartments are of concern for the assessment of road salts. Environmental effects can be either biotic or abiotic. Thus, endpoints of concern can include effects on individual organisms, populations or communities, as well as abiotic endpoints, such as deterioration of soil structure or lake stratification, which disrupts seasonal mixing of waters.
While there has not been systematic assessment of exposure to road salts for any given compartment across Canada that would permit detailed quantitative risk characterization at the national scale, many individual studies are available that permit the establishment of relationships between the use of road salts and their impacts on biotic and abiotic systems.
The following sections characterize the risks associated with releases of road salts into the environment, by reviewing the entry, exposure and effects related to five environmental compartments or biotic groups with regard to inorganic chloride salts. Data pertinent to ferrocyanides and their environmental risks are reviewed in a separate section (Section 3.7).
3.2 Groundwater
3.2.1 Introduction
This section summarizes the fate and impact of road salts on groundwater quality. Discussion in this section is based on a report prepared by Johnston et al. (2000). A summary of case studies (Morin, 2000) is also presented.
Data on concentrations of salts in groundwater are frequently discussed in published and unpublished literature in terms of exceedences of drinking water quality guidelines. These guidelines are set on the basis of taste thresholds at 200 mg sodium/L and 250 mg chloride/L (CCME, 1998). Although the use of groundwater as a drinking water resource is not considered in this assessment, a review of reported exceedences of a chloride concentration of 250 mg/L is pertinent to the assessment of potential impacts on aquatic biota. As reviewed in Section 3.3, the No-Observed-Effect Concentration (NOEC) for the 33-day survival of early life stage fathead minnow was 252 mg chloride/L. Furthermore, lethality data modelled for chronic exposure indicated that 10% of aquatic species would be affected at chloride concentrations of about 240 mg/L.
3.2.2 Groundwater principles
3.2.2.1 Hydrological cycle
The degree to which road salts may have an impact on the groundwater environment will vary significantly, depending on the road salt loading, climatic conditions, surface and subsurface soil conditions and location of the site within the overall hydrogeological environment.
Precipitation that falls across an area as rain or snow contributes to a number of hydrological processes. A portion of the precipitation will run off as overland flow from impervious surfaces, such as roadways and parking lots, and is eventually directed by open ditches or subsurface piped systems that discharge to streams, wetlands, lakes or other surface water features. Overland flow will result in the direct migration of surficial contaminants, like road salts, to surface water features, typically in the time frame of hours.
In certain settings, a portion of precipitation will infiltrate the shallow soil zone and move laterally through the unsaturated zone above the water table as interflow to the local discharge area. The transport of contaminants to surface water features as interflow typically occurs in the time frame of hours to days.
The balance of precipitation is subject to evapotranspiration or infiltrates as groundwater recharge to the water table. Contaminants introduced to the local, intermediate or regional groundwater flow systems may take several years to several hundred years before discharging as baseflow to surface water features.
3.2.2.2 Unsaturated flow and groundwater recharge
The amount of precipitation that infiltrates to the water table depends on a number of factors, including the duration and intensity of the rainfall event, the initial soil moisture content and the soil moisture characteristics. Based on the general temperature and precipitation pattern in Canada, the majority of groundwater recharge in a year is expected to occur in the late winter and early spring, as a result of winter snowmelt and spring rains (Gerber and Howard, 1997; Singer et al., 1997).
Vertical travel times through the unsaturated zone may be expected to range from less than 0.5 m/year to more than 20 m/year. In areas with thick unsaturated zones (i.e., 20-30 m), travel times for infiltrating precipitation to reach the water table may be in the range of 5-20 years in sandy soils. These travel times are important when considering the impacts to downgradient receptors and the time required before remedial measures implemented at the source would have noticeable effects.
3.2.2.3 Saturated flow and contaminant migration
Once below the water table, the movement of water is controlled by the fluid potential and the permeability of the geological material. When evaluating the migration of solutes, such as sodium, calcium and chloride ions, within groundwater environments, several other physical and chemical processes must be considered. Mechanical dispersion, molecular diffusion and density effects represent the primary physical transport mechanisms that affect the migration of sodium, calcium and chloride ions.
3.2.3 Impacts of road salts on groundwater quality
3.2.3.1 Factors controlling the migration of road salts
One of the primary concerns associated with inorganic sodium chloride road salts is their high solubility in water. The dissociated ions will migrate with infiltrating precipitation to the water table. The migration rate of chloride ion will be the same as that of the water. This is not the case for cations such as the sodium ion, which can be affected by cation exchange reactions in certain geological environments. The following equation presents the ion exchange reaction for sodium ion:
2Na+ + Ca(adsorbed) <> Ca2+ + 2Na(adsorbed)
In the equation, sodium ion is exchanged with calcium adsorbed to clay-rich materials. The net effect of this reaction is a decrease in sodium ion concentrations within the infiltrating groundwater and a Na:Cl ratio of less than 1. Reduced sodium ion concentrations in groundwater impacted by road salts have been documented by Howard and Beck (1993) and Pilon and Howard (1987).
3.2.3.2 Predicted road salt concentrations in groundwater
To provide an understanding of the impact that road salting may have on groundwater quality, an estimate of the amount of road salt available for dissolution in infiltrating precipitation is required. A simple spreadsheet mass balance model was developed to provide an indication of the equilibrium chloride ion concentration in shallow groundwater (Johnston et al., 2000). The model is based on the dissolution of the total chloride ion retained within the soil by recharging precipitation and does not consider any additional chloride ion from other sources. The calculated chloride ion concentrations can therefore be considered representative of regional-scale equilibrium chloride ion concentrations downgradient of a saltable road network. It should be noted that the model does not provide an indication of potential chloride ion concentrations immediately downgradient of a source area, such as an individual roadway, where higher chloride ion concentrations may be expected.
Input to the model includes groundwater recharge rates, road salt loadings and road densities typical of southern Ontario. Recharge rates ranging from 10 to 40 cm/year were selected based on a range of soil types. The chloride ion loadings were based on typical road salt application rates for both provincial and municipal road networks, as discussed in Section 2.2. The road salt available for infiltration to the water table was estimated based on the ranges reported by Wulkowicz and Saleem (1974), Scott (1980) and Howard and Haynes (1993). Road densities in the range of 1-15 two-lane-kilometres per square kilometre were selected based on the data presented in Morin and Perchanok (2000) and Stantec Consulting Ltd. (2000). Road densities of 0.01-0.1 two-lane-kilometres per square kilometre were assumed to be characteristic of low-density rural land use, road densities of 1-5 two-lane-kilometres per square kilometre were assumed to be characteristic of medium-density rural to semi-rural land use, and road densities of 10-15 two-lane-kilometres per square kilometre were assumed to be characteristic of urban land use.
Predicted chloride ion concentrations for a given recharge rate increase with increased loading, road network density and percentage chloride ion retained. Chloride concentrations decrease with increasing recharge rates due to the dilution effects of increased recharge. The highest chloride ion concentration, 3600 mg/L, was predicted for soil with a recharge rate of 10 cm/year, a loading of 40 tonnes chloride ion per two-lane-kilometre and 60% of the chloride ion applied available for dilution in recharging precipitation. For comparison, the chloride ion concentration for the same loading and percentage retained is 900 mg/L for soils with a recharge rate of 40 cm/year. It is expected that the percentage of chloride ion retained will vary significantly between a site with a recharge rate of 10 cm/year versus 40 cm/year. This is due to the fact that runoff would be higher for the site with a recharge rate of 10 cm/year, and therefore less chloride ion should be retained for dissolution in infiltrating precipitation.
Figures 12-14 present regional-scale groundwater chloride ion concentrations that were predicted for various road salt loadings, road densities and recharge rates. In each figure, an envelope corresponding to the maximum and minimum chloride ion concentrations is presented. Average chloride ion concentration envelopes for medium- and high-density road networks are also presented, assuming the average percentage chloride ion retained of 30% and 40%.
Figure 12 Estimated chloride concentrations in groundwater for various chloride application rates and a groundwater recharge rate of 10 cm/year (from Johnston et al., 2000)

Figure 13 Estimated chloride concentrations in groundwater for various chloride application rates and a groundwater recharge rate of 20 cm/year (from Johnston et al., 2000)

Figure 14 Estimated chloride concentrations in groundwater for various chloride application rates and a groundwater recharge rate of 40 cm/year (from Johnston et al., 2000)

For soils with recharge rates of 10 cm/year, typical of clay-rich soils, chloride ion concentrations may exceed 250 mg/L for chloride ion loadings of approximately 5 tonnes chloride per two-lane-kilometre under high-density road networks (Figure 12). Under medium road densities, chloride ion concentrations are predicted to remain below 250 mg/L for road salt loadings of approximately 10-15 tonnes chloride per two-lane-kilometre. For low-density road networks, typical of a rural setting, regional-scale chloride concentrations are predicted to remain well below 250 mg/L (Johnston et al., 2000).
Figure 13 presents the estimated groundwater concentrations for a soil with a recharge rate of 20 cm/year, typical of sand. Due to dilution from the increased recharge, slightly higher chloride ion loadings can be supported before a concentration of 250 mg/L is exceeded. Under high-and medium-density road networks, chloride ion loadings above 11 tonnes chloride per two-lane-kilometre and 25-34 tonnes chloride per two-lane-kilometre, respectively, may result in regional-scale chloride ion concentrations above 250 mg/L.
At recharge rates typical of very permeable soils, such as gravels or fractured rock, groundwater chloride ion concentrations exceed 250 mg/L only for high-density road networks. In this case, chloride ion loadings above about 16-22 tonnes chloride per two-lane-kilometre are predicted to result in concentrations above 250 mg/L (Figure 14).
The mass balance modelling completed for this section indicates that road salt loadings above approximately 15 tonnes chloride per two-lane-kilometre, or 24 tonnes sodium chloride per two-lane-kilometre, will result in regional-scale groundwater concentrations greater than 250 mg/L in any soil condition under high-density land uses. Considering that road salt loadings in southern Ontario, southern Quebec and the Maritime provinces are in the range of 20-50 tonnes sodium chloride per two-lane-kilometre, highly developed urban areas in these provinces are at the greatest risk of wide-scale groundwater impacts associated with road salting. In addition to groundwater impacts, surface water quality within these areas of Canada will be subject to the discharge of groundwater with elevated sodium and chloride ion concentrations as baseflow, particularly during the late summer and fall.
The mass balance modelling indicates that under low-density land use, chloride ion concentrations will remain well below 250 mg/L, reaching a maximum concentration of 24 mg/L for road salt loadings of 40 tonnes sodium chloride per two-lane-kilometre. It should be noted, however, that this concentration represents an average regional concentration, and higher concentrations may be expected immediately downgradient from individual roadways.
3.2.3.3 Plume migration and impact assessment
The above section provides an overview of the regional impact associated with non-point source loading from road salting in rural and urban areas. The impact on groundwater quality and plume development from point source loading associated with road salting along an individual roadway is discussed below.
To provide an indication of maximum chloride ion concentrations and the effects of dispersion on plume concentrations, a numerical solute transport model was developed for a simple unconfined sand aquifer system using the finite difference modelling programs MODFLOW (McDonald and Harbaugh, 1988) and MT3D (Zheng, 1990). The simplified aquifer was assigned a lateral hydraulic conductivity of 1 x 10-4 m/s, vertical hydraulic conductivity of 1 x 10-5 m/s, specific yield of 0.25, effective porosity of 0.30, longitudinal dispersivity of 10 m, and lateral and vertical transverse dispersivities of 1 m and 0.1 m, respectively. Linear constant-head boundaries were assigned at the upgradient (17.5 m) and downgradient (15 m) extents of the model to simulate a lateral hydraulic gradient of 0.005 m/m across the 500-m model length. Recharge was assigned at 30 cm/year to the entire model domain.
Within the simulated steady-state flow field, a square constant concentration source term of 1000 mg/L was assigned at the water table to represent the chloride ion loading from a section of roadway. This concentration is within the range (323-2930 mg/L, average 923 mg/L) for shallow groundwater beneath a four-lane arterial road in Kitchener, Ontario (Stantec Consulting Ltd., 2000). The formation and migration of an unretarded, dissolved-phase chloride ion plume was simulated for 5000 days, followed by the dissipation of the plume after removing the concentration source term.
At a simulated time of 100 days, typical of early summer following recharge of groundwater with elevated chloride ion concentrations from winter road salting, groundwater concentrations parallel to the plume showed the formation of a typical "cigar-shaped" plume as defined by the 10 mg/L contour. At 100 days, the 10 mg/L contour has migrated approximately 40 m downgradient from the source area, with the 250 mg/L contour restricted to within approximately 10 m of the source area. After a period of approximately 5 years, the plume has reached a steady-state condition as defined by the 10 mg/L contour in which the flux from the source area is balanced by the flux across the plume boundary. The 10 mg/L contour under steady-state conditions is located approximately 200 m downgradient from the source, while the 250 mg/L contour moved 20 m downgradient from the source area.
After a simulation period of approximately 5 years, the chloride ion source was removed to provide an indication of the time required for concentrations to dissipate. Chloride concentrations decreased rapidly after 50 days, with no simulated concentrations above 250 mg/L.
The above simulations indicate that impacts associated with road salting along individual highways may be limited in extent due to groundwater flow and transport processes, depending on the aquifer properties. Based on the modelling results, shallow wells and surface water features located within 20 m of salted roads are at the greatest risk of chloride ion impacts. The modelling does suggest that if road salting activities are discontinued, chloride ion concentrations within the shallow aquifer may improve significantly over time frames of months to years.
3.2.4 Case studies
This section presents modelling and field measurements for specific areas in Canada. Additional case studies involving impacts on groundwater quality associated with roadways and maintenance salt storage yards are presented in Morin (2000).
One method for assessing the regional-scale impacts associated with road salting was that applied by Howard and Haynes (1993) in the Highland Creek catchment located near Toronto, Ontario. Through detailed measurements of stream flow and chloride ion concentrations over a 3-year period, it was estimated that only 45% of the chloride ion applied as road salt within the basin was removed by surface water runoff. The remaining 55% was interpreted to be stored within the groundwater system. At current salt application rates, Howard and Haynes (1993) estimated an average chloride ion concentration of 426 ± 50 mg/L in groundwater discharging as baseflow to Highland Creek. While road salt application rates were not presented, the total loading over the basin was approximately 200 g sodium chloride/m2. Assuming a road density typical of an urban environment, the road salt application rate over the study area is estimated to be in the range of 20-30 tonnes sodium chloride per two-lane-kilometre, which is similar to that predicted based on the mass balance modelling completed as part of this report (Section 3.2.3.2).
Further evidence of the impact on groundwater quality and discharge to surface water features due to road salting in urban areas is presented in Howard and Beck (1993) and Eyles and Howard (1988). Chloride concentrations in springs discharging to Lake Ontario, along the Scarborough Bluffs near Toronto, were measured and found to be approximately 400 mg/L, similar to those estimated by Howard and Haynes (1993) for the Highland Creek watershed.
Long-term chloride ion concentrations from municipal supply wells provide further evidence of impacts on groundwater quality associated with winter road salting (Johnston et al., 2000). Figure 15 presents chloride ion concentrations from municipal water supply wells in an urban centre of southern Ontario. Data on current chloride ion application rates in the study area near the wells indicate that application rates have ranged from 18 to 36 tonnes sodium chloride per two-lane-kilometre, with an average application rate of 28 tonnes sodium chloride per two-lane-kilometre. Chloride concentrations beginning in the early 1960s were less than 50 mg/L and increased steadily to 250-300 mg/L in the late 1990s (Figure 15) (data after 1998 not plotted). One of the key issues with respect to the trends indicated in Figure 15 is that chloride ion concentrations are continuing to rise and have not reached steady state. Since 1998, chloride concentrations at these wells have increased at a rate similar to that seen since 1993 on Figure 15, with Well 3 now over 350 mg/L. However, it is noted that the groundwater being pumped by these wells represents precipitation that recharged the aquifer in the mid to late 1970s, with travel times from ground to surface on the order of 30 years. Hence, increased chloride concentrations observed at these production wells are the result of road salts that were spread 30 years ago. Given that these wells pump on average 3.3 million cubic metres per year and have 20-year capture zones that extend out over 12 km2, the concentrations predicted at these wells are considered to reflect regional-scale chloride concentrations. A comparison of the current concentrations of approximately 350 mg/L with the predicted concentrations of 300-450 mg/L indicates that the mass balance modelling approach presented in the previous sections provides reasonable estimates of chloride concentrations (Johnston et al., 2000).
Figure 15 Chloride concentrations in groundwater from municipal production wells in southern Ontario (from Johnston et al., 2000)

The time to steady state (the time when salt inputs are balanced by salt output) depends on local hydrogeological conditions and the size of the catchment area (Howard et al., 1993). Howard et al. (1993) used two numerical models to demonstrate temporal and spatial changes in water quality. The model FLOWPATH showed that chemically conservative contaminants such as chloride released within a few kilometres of rivers and Lake Ontario will discharge in about 5 years. Contaminants that are released in more central areas will take more than 100 years to be flushed from the groundwater system. When road salt is evenly distributed in a representative 460-km2 region of the Greater Toronto Area, Howard et al. (1993) demonstrated, using the FLOWPATH model, that the average chloride concentrations will reach steady-state values within 200 years of initial salt application. Using another model, AQUA, in a smaller area near Toronto, steady state was reached in just 30 years. At steady state, the levels of chloride at a distance of 200 m from the salted highways are 2-3 times the levels of chloride observed in the discharging baseflow.
Williams et al. (1997) investigated chloride concentrations in 20 springs in southeastern Ontario. Chloride concentrations ranged from 8.1 to 1149 mg/L. The higher chloride concentrations originated from groundwater that may be contaminated by road salt. Williams et al. (1999) continued this research, noting that the mean chloride concentrations were 2.1 mg/L at the Glen Majors Conservation Area and about 100 mg/L in rural areas. Chloride concentrations in springs near urban areas were higher (>200 mg/L), with the maximum concentration 1345 mg/L and mean 1092 mg/L. The spring with highest concentrations was adjacent to a highway and bridge. Chloride concentrations at these urban sites also increased between November 1996 and November 1997. Salinity contamination was related to road salt. Williams et al. (1999) noted that spatial patterns in road salt contamination were more readily detected by sampling springs than by sampling creeks, because chloride concentrations in spring waters (i.e., discharging groundwater) exhibited relatively little seasonal variability. In contrast, chloride concentrations in creeks were highly variable seasonally.
Howard and Taylor (1998) reported high concentrations of chloride in springs discharging from aquifers along the Scarborough Bluffs, a stretch of Lake Ontario shoreline east of Toronto, and along an urban-rural transect from the Oak Ridges Moraine to Metropolitan Toronto. Concentrations exceeding 2800 mg chloride/L were detected in springs issuing from shallow aquifers in the Oak Ridges Moraine-Toronto area. The primary source of the chloride contamination was road de-icing chemicals (Howard and Beck, 1993).
In 1970, an expressway was constructed at a distance of 50-200 m from wells used by the municipality of Trois-Rivières-Ouest to obtain groundwater. By 1976, an increase in salinity was noted (Gélinas and Locat, 1988). In the early 1980s, a plume of salty groundwater was identified. Chloride concentrations in samples from piezometers ranged between 3 and 1500 mg/L. Data collected in 1985 revealed that the shape and extent of the plume varied depending on the time of year and the quantity of water pumped from each of the six municipal pumping stations. Data for 1985-86 indicate that concentrations gradually increase throughout the spring (during snowmelt) to peak in August and September. Higher concentrations were observed for a municipal pumping station located downgradient from the highway. Concentrations at most wells upgradient from the highway were less than 5 mg/L. Mean chloride concentrations in one station downgradient (80 mg/L) were about 6 times higher than the average concentration in a nearby station upgradient (14 mg/L).
The municipality of Cap-de-la-Madeleine pumps water from 27 wells. While the quality of this water was considered to be extremely good, certain wells have experienced a gradual increase in the concentration of chloride since the 1970s that is attributed to the use of de-icing road salts. In areas where ditches were not connected to a drainage system, runoff is left to percolate through the soils (Delisle, 1999).
3.2.5 Conclusions
The potential for impacts on groundwater and surface water quality was evaluated using a mass balance technique that provides an indication of potential regional-scale chloride ion concentrations downgradient of saltable road networks. The mass balance modelling indicates that for road salt application rates above 20 tonnes sodium chloride per two-lane-kilometre, regional-scale groundwater chloride ion concentrations greater than 250 mg/L will likely result under high-density road networks, typical of urban areas. Considering road salt application rates throughout Canada, urban areas in southern Ontario, southern Quebec and the Maritime provinces are at the greatest risk of wide-scale groundwater impacts associated with road salting. The mass balance modelling completed as part of this report indicates that rural and semi-rural areas are not as likely to be impacted by chloride ion concentrations above 250 mg/L on a regional scale. However, local impacts along individual roadways have been documented.
Groundwater containing road salts will eventually upwell into the surface water or emerge as springs. Research has shown that only a portion of the road salts that are applied to roads are removed by surface water runoff, and a significant proportion of the salt may be stored within the groundwater system. Elevated concentrations of chlorides have been detected in both groundwater and groundwater that has emerged to the surface as springs. Predicted and measured concentrations in groundwaters and springs exceed those identified as lethality thresholds for many organisms, as identified in Section 3.3 (e.g., 10% of species can be expected to be affected by concentrations greater than 240 mg/L).
3.3 Aquatic ecosystems
3.3.1 Scoping and assessment approach
Aquatic ecosystems are vulnerable to various impacts from road salts. Evans and Frick (2001) assessed such impacts, focusing on a) a literature review of the effects of road salt applications on aquatic ecosystems and b) the lethal and sublethal effect levels of sodium chloride and other chloride salts as determined from laboratory studies. These studies were then used in the characterization of the risks of road salts. Highlights of this report are presented in this section of the Assessment Report.
The aquatic ecosystems that were considered in this assessment included a wide variety of habitats, including streams, rivers, wetlands, ponds and lakes. These ecosystems were located across Canada and in a wide variety of regions, some experiencing minimal and others intense anthropogenic stress. Many aquatic ecosystems are located in relatively pristine areas of Canada, where anthropogenic impacts are minimal. Nevertheless, such impacts are of concern, because pristine habitats are vulnerable to relatively minor anthropogenic stresses. Small shifts in species composition, particularly for phytoplankton, are highly indicative of such impacts. This is because the species assemblage of pristine environments is, in large measure, composed of taxa that are adapted to unperturbed environments; pollution-tolerant species generally are minor constituents of such assemblages. Therefore, there is the concern that small increases in chloride concentrations from highway runoff or loss from salt storage depots may have measurable impacts on such communities.
Other aquatic ecosystems are located in rural and agricultural areas. Taxa inhabiting these environments are experiencing a variety of stresses as a result of a variety of anthropogenic activities, including habitat loss and increased chemical inputs. With increased development in the Canadian countryside, including the loss of naturally occurring ponds and wetlands to agricultural usage, the narrow tens of metres of space on the sides of roadways may provide important habitat for displaced plant and wildlife communities. Moreover, this land can serve as migration corridors between larger, relatively undisturbed areas (Nadec, 2001).
Aquatic ecosystems are also found in urban areas, with many such aquatic ecosystems gradually being incorporated into the growing urban landscape. In such areas, provision is made to protect these ecosystems by limiting development around their immediate shoreline and by minimizing anthropogenic impacts in their immediate watershed. Well-known examples of such incorporated parkland areas include those established around the many creeks running through the Metropolitan Toronto area and parks such as Stanley Park in Vancouver. These aquatic systems, while no longer pristine, nevertheless merit protection against further environmental degradation. Increasingly, the design of subdivisions includes the incorporation of rural features such as lakes and creeks. Many of these engineered ecosystems are built within the natural landscape over existing aquatic habitats (e.g., ponds, wetlands, sloughs, creeks), which are rapidly recolonized by temporarily displaced aquatic organisms.
Stormwater detention ponds, which are often constructed in a natural aquatic ecosystem, e.g., a creek or wetland, are engineered habitats. Nevertheless, these ponds do provide habitat for a wide variety of organisms (Bishop et al., 2000). While clearly not ideal because of the variety of contaminants associated with them and altered hydrological regime, they provide aquatic habitat and resources for both the organism and the population (which requires a critical area for self-maintenance) in an increasingly fragmented urban and rural landscape.
Aquatic organisms considered in the assessment included all components of the aquatic ecosystem, i.e., bacteria, fungi, protozoans, zooplankton, benthic invertebrates, fish and amphibians. However, not all groups were equally well represented in the literature. For example, laboratory studies investigating chloride toxicity were heavily weighted towards fish, zooplankton and benthic invertebrates and for short-term exposures. Such studies, by their nature, focus on the capacity of species to endure short-term osmotic stress due to elevated salinities. Few studies investigated chronic toxicity; chronic toxicity was estimated using approaches followed by the U.S. EPA (1988) for setting water quality guidelines for chloride.
Relatively few field studies have been conducted specifically investigating the impacts of road salts on aquatic ecosystems. The majority of these studies were conducted in the 1970s and 1980s. Many of the studies that were found were conducted in the United States but have general application to the Canadian environment.
As provided for in CEPA 1999 and as noted in Environment Canada (1997a), a weight of evidence approach involving consideration of several lines of evidence is used to strengthen the confidence in assessment conclusions. These lines of evidence include consideration of short-term toxicity studies, estimates of acute toxicity, field studies, emerging information and the general knowledge of the use patterns of road salts in the Canadian environment. In addition, a tiered approach, using quotient-based assessments, is used to determine the likelihood of adverse effects occurring in aquatic ecosystems as a result of release of road salts into the environment.
3.3.2 Laboratory studies
The literature review of laboratory studies of chloride toxicity focused on obtaining information on sodium chloride toxicity, but also collected data for calcium, potassium and magnesium chloride toxicity, since these chlorides are also present in various road salt formulations. Toxicity data are essential for assessing the environmental concentrations and exposure durations that may be harmful. Both short-term (hours to days) toxicity and long-term (weeks to months) toxicity are of concern. In the following paragraphs, the highlights of these determinations are presented.
3.3.2.1 Short-term or acute toxicity studies
One exposure scenario that is of concern is short-term exposures (hours to days) to elevated chloride concentrations, particularly when the dilution capacity of the receiving water body is weak. Direct highway runoff, particularly into creeks, streams and other roadside waterways, is of particular concern. Organisms inhabiting larger water bodies such as rivers and lakes, with their greater dilution capacity, are less likely to be exposed to conditions in which acute toxicity is of concern. Organisms inhabiting wetlands, which are relatively small, stationary water bodies with slow water exchange, may experience significant acute and chronic toxicity.
An extensive review of the literature revealed several dozen studies that investigated the short-term or acute toxicity of sodium chloride to aquatic organisms; fewer studies were located for magnesium, calcium and potassium salts (Evans and Frick, 2001). These acute studies were subsequently grouped into four time intervals to estimate toxicity following various exposures to elevated chloride concentrations:
- <24 hours: For exposures of less than 1 day, four studies were located for fish (3) and a benthic organism (1) (Table 10). The LC50s ranged from 6063 to 30 330 mg chloride/L, with a geometric mean of 12 826 mg chloride/L. Natural salinities in this range are associated with estuaries, tidal marshes, oceans and inland saline lakes. Salinities in this range have also been associated with highway runoff (and spray) from multiple-lane highways, waste snow from urban areas, leachate and groundwater from patrol yards. Elevated chloride concentrations at the lower end of this range have also been observed in well water, a stream and a wetland near a road salt depot (see Sections 2.3, 2.4, 2.6 and 3.4).
- 24 hours: For exposures of 24 hours, seven studies were located testing fish (4) and cladocerans (3) (Table 11). The LC50s ranged from 1652 to 8553 mg chloride/L, with a geometric mean of 3746 mg chloride/L. Salinities in this range are associated with estuaries, tidal marshes and inland saline lakes. Salinities in this range have also been associated with highway runoff (and spray) from multiple-lane highways, waste snow from urban areas and leachate from patrol yards. Elevated chloride concentrations at the lower end of this range have also been observed in well water, a stream and a wetland near a road salt depot (see Sections 2.3, 2.4, 2.6 and 3.4).
- 3 or 4 days: Several studies were found investigating the 4-day toxicity of sodium chloride to a variety of organisms, and a smaller set of 3-day exposures was also found. The 3-day exposure data were converted into 4-day estimates using a conversion factor as described in Evans and Frick (2001). This resulted in 28 observations, including fish (13), cladocerans (7) and other invertebrates (8) (Table 12). Some species were the subject of several studies, e.g., Daphnia magna and fathead minnow (Pimephales promelas). There were differences in some LC50s, probably as a result of test conditions (e.g., use of reconstituted water in Birge et al., 1985) and/or differences in the physiological tolerances of the test organisms. In general, fish had greater tolerances than invertebrates. The LC50s ranged from 1400 to 13 085 mg chloride/L, with a geometric mean of 4033 mg chloride/L. The higher 4-day than 1-day tolerances is due, in large measure, to the inclusion of mosquito fish (Gambusia affinis, a hardy fish that is used worldwide for mosquito control) and American eel (Anguilla rostrata, a fish that spends its adult life in the ocean) in the data set. Salinities in this range are associated with estuaries, tidal marshes and inland saline lakes. Salinities in this range have also been associated with highway runoff (and spray) from multiple-lane highways, waste snow from urban areas and leachate from patrol yards. Elevated chloride concentrations at the lower end of this range have also been observed in well water, a stream and a wetland near a road salt depot (see Sections 2.3, 2.4, 2.6 and 3.4). Moreover, salinities in the 1000-4000 mg/L range have been reported for several creeks in the Metropolitan Toronto area.
- 7-10 days: Seventeen studies were found estimating chloride toxicity at exposure times of 7-10 days (Table 13). Exposures of this duration are at the upper end of acute exposure times but are not sufficiently long to be considered chronic for long-lived organisms such as amphibians and fish. These studies examined responses such as mortality, reduced growth, fecundity and failure to complete development, e.g., from egg to embryo. There were five fish tests, two amphibian tests, nine zooplankton tests and one algal test. The LC50s and EC50s ranged from 874 to 3660 mg chloride/L, with a geometric mean of 1840 mg chloride/L.
1 LC50/EC50 ratio calculated for studies where both parameters were estimated (i.e., Cowgill and Milazzo, 1990; Beak, 1999).
This salinity range is defined as subsaline (Hammer, 1986). Such salinities are found at the interface between the marine and freshwater realms and in inland subsaline lakes. Species diversity declines rapidly with increasing salinity in this range (Wetzel, 1983). Salinities in this range have been associated with highway runoff (and spray), waste snow, leachate from patrol yards, well water, a stream, a wetland near a road salt depot, and creeks and rivers in the Metropolitan Toronto area.
3.3.2.2 Chronic toxicity
Long-term or chronic toxicity to sodium chloride, which may be expected to occur over weeks to months, has seldom been investigated in the laboratory. Only two studies were located in the literature review.
Birge et al. (1985) estimated the 4-day LC50 toxicity of chloride to fathead minnow as 6570 mg/L. The NOEC for the 33-day early life stage test was 252 mg chloride/L and the Lowest-Observed-Effect Concentration (LOEC) was 352 mg/L, giving a geometric mean of 298 mg chloride/L as the estimated chronic value and an acute to chronic ratio (ACR) of 22.1. Birge et al. (1985) also tested Daphnia pulex. The 4-day LC50 was 1470 mg chloride/L. For the 21-day test, the chronic toxicity, calculated as the geometric mean of the NOEC (314 mg/L) and the LOEC (441 mg/L), was 372 mg chloride/L. This value is similar to that for fathead minnow. However, because of the lower acute toxicity value for Daphnia, the ACR was 3.95. Birge et al. (1985) conducted a second 4-day toxicity test with D. pulex using natural stream water and reported an EC50 of 3050 mg chloride/L, or twice the tolerance of that using reconstituted water. (If this value were used to estimate the ACR, the value would become 8.20.) Birge et al. (1985) calculated the geometric mean of the Daphnia and fathead minnow chronic toxicity values to estimate a chronic chloride toxicity concentration of 333 mg/L, although they also indicated some uncertainty in the D. pulex values.
In a later study, the U.S. EPA (1988) developed water quality criteria for chloride, relying largely on the results of the Birge et al. (1985) study for acute toxicity and ACR estimates. They used the same toxicity data and ACR (3.95) for Daphnia pulex. However, for fathead minnow, they estimated the chronic toxicity as 433.1 mg/L (based on the geometric mean of 9% impaired survival at 352 mg/L and 15% impaired survival at 533 mg/L), giving an ACR of 15.17. They also cited a rainbow trout (Salmo gairdneri, now Oncorhynchus mykiss) study conducted by Spehar (1987), who reported an acute toxicity of 6743 mg chloride/L, a chronic toxicity of 922.7 mg chloride/L and an ACR of 7.31. The geometric mean of these three ACRs was used to develop a final ACR of 7.59. A final acute genus toxicity value of 1720 mg/L, based on the most sensitive genus (Daphnia), was divided by this ACR to give a final chronic toxicity value of 226.5 mg/L.
A modification of the ACR approach was used to estimate the chronic toxicity of the organisms for which there were 4-day acute toxicity data. Two approaches could have been used. The first was to apply the Daphnia ACR (3.95) as developed by Birge et al. (1985) and later used by the U.S. Environmental Protection Agency (U.S. EPA, 1988) to the invertebrate data and the geometric mean (10.53) of the fish ACR data reported by the U.S. EPA. However, Birge et al. (1985) considered the acute toxicity data (and ACR) to be too low. Application of two different ratios also changes the general ordering of species sensitivities based on the 4-day acute toxicity data, so that fish now appear more sensitive and aquatic invertebrates more tolerant to long-term elevated chloride exposures. This seems highly unlikely, with aquatic organisms such as cladocerans and insect larvae being poorly represented in the marine and estuarine environment, while fish taxa such as the salmonids have strong marine affinities. A more suitable approach, then, is to follow the U.S. EPA approach and to use the mean ACR of 7.59 based on the geometric mean of the three ACR studies.
Using a mean ACR of 7.59, the acute toxicity of chloride was estimated as ranging from 184.5 mg/L for Ceriodaphnia dubia to 1724 mg/L for the black eel stage of the American eel, with a geometric mean value of 512.6 mg/L chloride (Evans and Frick, 2001). The lower value is approximately 24 times greater than the average chloride concentration (8 mg/L) of the world's river waters (Wetzel, 1983). The geometric mean of the chronic toxicities, 551.9 mg chloride/L, is at the lower end of the 500-1000 mg chloride/L range, where many researchers have reported losses in freshwater species with increasing salinity (Williams, 1987; Hart et al., 1990; Leland and Fend, 1998; Evans and Frick, 2001). A marked drop in the species diversity of freshwater ecosystems occurs as salinity increases from about 2000 mg/L to about 5000 mg/L (Figure 16; see also Wetzel, 1983). In general, for freshwater species, the number of species decreases as salinity increases, with the greatest and most rapid decrease in species numbers at chloride ion concentrations of 1000-3000 mg/L. Salinities in this 185-1724 mg/L range have been associated with highway runoff (and spray), waste snow, leachate from patrol yards and creeks in the Metropolitan Toronto area. The lower salinity values have been associated with chloride-impacted lakes.
3.3.3 Assessment of toxicity to aquatic ecosystems
Three approaches were used to assess the toxicity of road salts in the aquatic environment. These are a) toxicity thresholds and quotient-based risk characterization, as described in Environment Canada (1997a); b) field evidence of population or ecosystem effects; and c) consideration of abiotic effects. Briefy, these approaches can be described as follows:
- Toxicity thresholds and quotient-based risk characterization: This is a quotient-based approach, which is used to determine the likelihood of adverse effects occurring in aquatic ecosystems as a result of release of road salts into the environment. This approach is based on comparisons of concentrations of chloride in the aquatic environment resulting from the release of road salts (based on literature and analyses done for this assessment) and toxicity thresholds for chloride salts as identified in laboratory studies.
- Field evidence of population or ecosystem effects: The quotient-based approach generally focuses on the consideration of toxicity thresholds. However, community structure can be affected by environmental conditions that, while not necessarily exposing organisms to concentrations of contaminants that are themselves toxic, can result in selective advantage for certain populations or species and thereby result in shifts in populations and changes in community structure. Thus, field studies are particularly well suited for investigating the chronic effects of elevated chloride concentrations in the environment. However, because not all variables are controlled in such studies, it is possible that other, covarying variables affect some observed responses.
- Abiotic effects: Through their interactions with physical, chemical or biological components of the environment, substances may alter the abiotic environment and thereby affect ecosystems. Road salts may operate in two important ways. The first is by affecting the density of water and hence its mixing properties. This is especially important in relatively stationary water bodies such as lakes, ponds and wetlands. Second, chloride may increase the solubility of a variety of compounds, including metals.
3.3.3.1 Quotient-based risk characterization
The quotient-based environmental risk assessment is based on the procedures outlined in Environment Canada (1997a). Based on possible exposure scenarios, a hyperconservative or conservative Estimated Exposure Value (EEV) is selected. Next, an Estimated-No-Effects Value (ENEV) is determined by dividing an experimental Critical Toxicity Value (CTV) by an application factor to account for the uncertainty surrounding the extrapolation to chronic or pulsed exposure, the extrapolation from laboratory to field conditions, and interspecies and intraspecies variations in sensitivity. The CTV can be based on a wide range of toxicity responses, including mortality, growth, reproduction, fecundity, longevity, productivity, community structure and diversity.
Figure 16 Species diversity across a salinity and chloride gradient (from Wetzel, 1983)

After calculating ENEVs, a hyperconservative (Tier 1) quotient (EEV/ ENEV) is calculated for pertinent sensitive taxa in order to determine whether there is potential environmental risk from the release of that substance. If these quotients are less than 1, it can be concluded that the substance poses little risk to the environment, and the risk assessment is completed. If, however, the quotient is greater than 1 for a particular assessment endpoint, then the risk assessment for that endpoint proceeds to a Tier 2 analysis, where a more realistic but conservative quotient is calculated for each pertinent taxon. If these quotients are greater than 1, the assessment proceeds to a Tier 3 level. Here, the broader likelihood and magnitude of effects are considered. This latter approach involves a more thorough consideration of sources of variability and uncertainty in the risk analysis.
3.3.3.2 Tier 1 and Tier 2 assessments
A Tier 1 assessment is based on a hyperconservative exposure scenario, generally assuming that the EEV is the maximum concentration measured or likely to be encountered in the Canadian environment. The highest reported sodium chloride concentrations in the aquatic environment have been associated with highway runoff, salt-contaminated snow and contaminated waters near salt storage depots.
In Ontario, chloride concentrations as high as 19 135 mg/L were reported in highway runoff from the Skyway Bridge (Mayer et al., 1998). Delisle et al. (1995) assessed the concentration of chloride in snow cleared from city streets in Montréal. The average concentration of chloride was 3851 mg/L, with a maximum reported concentration of 10 000 mg/L. Concentrations of chloride in waste snow in Montréal averaged 3115 mg/L for secondary streets and 5066 mg/L for primary streets (Delisle and Dériger, 2000; Section 2.3.1). Chloride concentrations in leachate from poorly maintained salt storage depots may reach 66 000 mg/L (Section 2.4.2.1). While no Canadian wetland studies have been identified, in Maine, chloride concentrations in surface waters of a bog located near a salt storage depot reached 13 500 mg/L; concentrations remained elevated throughout the March to October sampling period (Ohno, 1990). Therefore, for the purposes of Tier 1 assessments, an EEV of 10 000 mg/L was used to represent estimated maximum chloride concentrations. Such a concentration is representative of snowmelt draining from highways into small receiving water bodies such as roadside creeks and wetlands and approximates that observed in wetlands near poorly designed road salt storage depots.
Tier 2 assessments involve a further analysis of exposure and/or effects to calculate a quotient that is still conservative, but is more realistic than the hyperconservative quotient calculated in Tier 1 (Environment Canada, 1997a). In Canada, the best-documented impacts of road salt on chloride levels in groundwater and other aquatic systems have been for the Metropolitan Toronto area. Williams et al. (1997, 1999) demonstrated the impact of road salts on groundwater and the invertebrate communities inhabiting groundwater-fed springs. There is also a comprehensive water quality data set for the numerous creeks flowing through the Metropolitan Toronto area. The southern Ontario region is also an area of high road salt application rates. Hence, this data set was examined to estimate a Tier 2 EEV.
Chloride concentrations were measured in several streams in the Toronto Remedial Action Plan watershed over 1990-1996 (see Evans and Frick, 2001; Table 9). Data were collected seasonally. Maximum chloride concentrations reported at individual stations included 2140-3780 mg/L for three stations on Etobicoke Creek, 3470 mg/L at Mimico Creek, 4310 mg/L at Black Creek, 96-4310 mg/L at five stations on the Humber River, 960-2610 mg/L at three stations on the Don River and 1390 mg/L at Highland Creek. Seasonal plots of chloride concentration variations in Highland Creek over 1990-1993 show several winter sampling periods in which chloride concentrations exceeded 1000 mg/L for what appear to be several periods of 1 week. Mean chloride concentrations at these sites ranged from 278 to 553 mg/L, with winter concentrations approximately twice those of summer. Williams et al. (1999) reported that an Ontario spring located near a highway and bridge had a mean chloride concentration of 1092 mg/L as a probable result of road salt contamination of groundwater. While the maximum chloride value observed in creeks and rivers in the Toronto area that have been contaminated by road salts is in the range of 2000-4000 mg/L, the frequency and duration of such occurrences are likely variable. Therefore, a less conservative estimate of 1000 mg/L was used as the EEV in the Tier 2 assessments. Chloride concentrations in this range have been commonly observed in Toronto-area creeks and rivers, in a contaminated spring in the Toronto area and in a bog contaminated by a salt storage depot (Wilcox, 1982). It is assumed that, for these scenarios, exposure times would be in the order of days.
Data used for generating the CTVs used in Tier 1 and 2 assessments were obtained during the literature review as described above and presented in detail in Evans and Frick (2001). Suitable data were not found for all trophic groups. The most comprehensive data were from the 2- to 4-day studies. The endpoint most typically investigated was mortality (zooplankton, benthos), unsuccessful development of eggs to larval stages (EC50 data) and reduced growth (phytoplankton, macrophytes).
To calculate ENEVs, an application factor of 100 was used for toxicity data based on LC50 and EC50 data as recommended in the guidance manual (Environment Canada, 1997a) and 10 for toxicity data based on LC25 and EC25 data.
The Tier 1 assessments indicated that risk quotients were greater than 1 for all taxa considered. Accordingly, a Tier 2 assessment was conducted for all taxa. Tier 2 assessments were conducted by reducing the EEV from 10 000 mg chloride/L to 1000 mg chloride/L. Results are presented in Table 14 (given the assumed differences in exposure scenarios between Tier 1 and Tier 2, all Tier 1 quotients are 10 times greater than the Tier 2 quotients). Even if the Tier 2 application factors were reduced by a factor of 10, most quotients would still exceed 1. Tier 3 assessments were therefore done for all groups of organisms considered in Tiers 1 and 2.
3.3.3.3 Tier 3 assessments with field validation
Tier 3 assessments include considerations of the distributions of exposures and/or effects in the environment (Environment Canada, 1997a). This is a challenging task, given the wide variety of road salt entries into the Canadian environment, e.g., leakage from salt storage depots, road and highway runoff (and spray) and transport via creeks into larger aquatic ecosystems, such as wetlands, rivers, ponds and lakes. It is also a complex task, as watercourses vary tremendously in their size, location in relation to a potential source of road salt source, flow regimes, etc.
While data on chloride concentrations have been collected as part of routine water quality monitoring programs, most data have not been subjected to rigorous analysis for spatial and temporal distributions and for causal factors. Thus, while Evans and Frick (2001) conducted an extensive review of the literature, a relatively small number of case history studies were located, especially for the Canadian environment. This situation may begin to change as researchers and agencies devote more effort to better understanding the impacts of road salts on chloride concentrations in the environment.
In order to conduct the Tier 3 assessment, a scenario approach is employed to describe the different settings in which road salts have or may have impacted the Canadian aquatic environment. This approach is based on information on road salt usage patterns across Canada, known information on chloride levels in the aquatic environment, toxicity data as described previously and reported environmental impacts or case studies. Many of the case studies are drawn from literature in the United States, where a greater number of studies have been conducted, but are representative of use in Canada.
3.3.3.3.1 Case 1: Road salt runoff and urban creeks, streams and small rivers in densely populated areas
As cities expand into the rural landscape, they enclose an increasingly large network of creeks, streams and rivers. Moreover, two-lane roads increase in density and, for larger cities, four-lane and larger highways are constructed, resulting in a highly altered watershed. Unlike the rural landscape, where a significant fraction of the runoff may be retained in relatively close proximity to the roadway, in the highly paved urban setting, much of the runoff is delivered in pulses into creeks, streams and rivers. Moreover, the absolute volume of road salt used is higher.
The Metropolitan Toronto area represents the clearest example of the impacts of a dense network of highways on the numerous creeks, streams and, eventually, rivers flowing through this densely populated urban area. As they flow south towards Lake Ontario, these creeks pick up more and more of the urban runoff. As previously noted, chloride concentrations can exceed several hundred milligrams per litre for much of the winter, with peak concentrations reaching 1000-4000 mg/L. Watercourses with high chloride concentrations include Etobicoke, Mimico, Black and Highland. The Don River can also have high chloride concentrations during much of the winter.
Schroeder and Solomon (1998) conducted a formal investigation of chloride concentrations and toxicity at three stations on the Don River. Chloride concentrations from November 1995 to April 1996 ranged up to about 950 mg/L at site 1, up to about 2600 mg/L at site 2 and up to about 1150 mg/L at site 3. These researchers also collected acute lethal toxicity data (LC50 and EC50) for 13 fish and 7 invertebrate taxa; values ranged from about 1000 to 30 000 mg/L. Next, the researchers superimposed the cumulative toxicity data on the cumulative chloride distribution data. They then estimated that 10% of the species would be expected to experience acute effects of chloride toxicity at least 90% of the time at two of the sites and at least 85% of the time at the third site.
Several field studies were located that provided corroborating evidence that chloride concentrations in the 500-1000 mg/L range can impact stream and creek ecosystems. The following paragraphs summarize some of these studies.
Williams et al. (1997) detected significant variations in macroinvertebrate community structure related to different concentrations of chloride in 20 groundwater-fed springs in southeastern Ontario in the Metropolitan Toronto area. The chloride content of these springs ranged from 8.1 to 1149 mg/L. Tipulidae and Ceratopogonidae were associated with springs containing higher chloride concentrations, whereas taxa such as Gammarus pseudolimnaeus (an amphipod) and Tubellaria (a flatworm) were found only in springs with low chloride concentrations. Crowther and Hynes (1977) reported that 20% of a test population of G. pseudolimnaeus dies with a 1-day exposure to 4121 mg sodium chloride/L (2500 mg chloride/L). The higher chloride concentrations in some of the springs originated from groundwater that apparently had been contaminated by road salt.
Crowther and Hynes (1977) experimentally investigated the effect of chloride concentrations on the drift of benthic invertebrates in Lutteral Creek, a trout stream located in southern Ontario. This creek is a small, spring-fed tributary of the Speed River. They also investigated chloride concentrations in Laurel Creek (annual discharge 19 x 106 m3), a tributary to the Grand River, which passes through urban Waterloo. At that time, Waterloo had a population of 32 000. Therefore, the general results of the study by Crowther and Hynes (1977) may apply to the creeks flowing through the numerous small towns in such regions as southern Ontario, Quebec and the Maritimes, where road salt is heavily used. Chloride levels in Laurel Creek varied seasonally, with peak concentrations as high as 1770 mg/L being observed for a 2-week period between January 5 and February 6, 1975.
Considering the elevated concentrations in Laurel Creek, a series of experiments was conducted in Lutteral Creek to assess the impacts of pulses of sodium chloride-laden water on benthic drift. Fifteen metres of Lutteral Creek were divided longitudinally by sheets of corrugated steel and benthic drift assessed on the control side and experiment side of the partition. Highlights are presented in Evans and Frick (2001).
Crowther and Hynes (1977) concluded their study by noting that, as chloride levels in streams approach 1000 mg/L, impacts on benthic drift may begin to appear in streams.
During their study, they noted that most of these effects would be expected to occur during winter and early spring. However, they warned that, as groundwater continued to become contaminated with chloride salts, stream flows might be contaminated by chloride-rich groundwater inflows during summer low-flow periods.
Dickman and Gochnauer (1978) studied the effects of exposure to sodium chloride on the density of bacteria and algae in Heyworth Stream, near Heyworth, Quebec, between July 24 and October 1, 1973. Sodium chloride was added at one location along the stream to maintain chloride concentrations of 1000 mg/L (1653 mg sodium chloride/L). This concentration was selected to simulate the impact of road salt from storm sewers on a receiving stream microflora ecosystem. It would also simulate the impacts of elevated chloride concentrations in groundwater of spring ecosystems. Sodium chloride concentrations at the control site were 2-3 mg/L. The stream was described as shallow, fast flowing and densely shaded. Artificial substrates (tiles) were placed in the creek at the experimental site and at an upstream control and recovered at weekly intervals. The treated site had lower algal diversity and lower standing crops of photosynthetic periphyton or algae, which was related to osmotic stress. Auxospore (a resting stage) formation of the diatom Cocconeis placentula was noted at the treated site on day 28 but not at the control site; auxospore formation is often triggered by environmental stress. Density of bacteria was greater at the treated site, which was attributed to a reduction in the grazing pressure on the bacterial population because of the reduced number of grazers, such as flagellates, ciliates and rhizopods. The incidence of diatom parasitism was lower at the treated site than at the control site, possibly because the sodium chloride inhibited fungal growth, as indicated by other researchers (Kszos et al., 1990; Rantamaki et al., 1992).
Other studies investigated the effects of salinity of benthic and algal community structure. Although the salinity was not due to road salt, these studies provide further support for the hypothesis that stream community composition is sensitive to salinity, with species composition changing as salinity increases towards 1000 mg chloride/L.
Leland and Fend (1998) investigated benthic distributions in the San Joaquin River Valley, California. Total dissolved solids concentrations ranged from 55 to 1700 mg/L. Distinct assemblages were observed at the high and low salinities, and the distribution of many taxa indicated salinity optima. Ephemeroptera (mayfly larvae) rarely occurred at salinities above 1000 mg/L (600 mg chloride/L). While other, covarying factors may have affected these relationships, the authors noted that patterns were not related to pesticide distributions or to water discharge and irrigation regime.
Short et al. (1991) investigated benthic invertebrates in a Kentucky stream subject to chloride seepage from nearby oil field operations. Ephemeropterans were the group least tolerant of elevated sodium chloride levels and were absent in regions where salinity exceeded 2000 mg chloride/L. Fish appeared to be more tolerant of these elevated salinities.
Blinn et al. (1981) investigated the seasonal dynamics of phytoplankton at three locations along the Chevelon Creek system in Arizona. The chloride concentration at site 1 was 88 mg/L at baseflow compared with 900-1100 mg/L at site 3, located 10.8 km downstream of site 1 and 3.5 km downstream of site 2 (chloride concentration at site 2 was not given). The elevated chloride concentration at site 3 was due to numerous seeps and springs from the canyon wall. There were marked differences in phytoplankton between the three sites, with species associated with differences in salinities.
Overall, these studies suggest that continuous exposure to chloride concentrations as low as 1000 mg/L (1653 mg sodium chloride/L) for time periods as short as 1 week can result in changes in stream periphyton and benthic communities. Furthermore, if these exposures persist, community composition remains different from lower-salinity upstream and downstream sites. Several severely chloride-contaminated streams and ponds adjacent to roadways have been identified in the Metropolitan Toronto area and surrounding municipalities. The number of affected streams and creeks is likely to increase with increased population growth in southern Ontario. Chloride-contaminated streams may also occur in other similar areas of Quebec and elsewhere where there is a dense network of highways and heavy road salt use. Areas in which continuous (or long-term) exposures are most likely to occur are seepages from road salt storage yards and contaminated groundwaters that later emerge as springs. Data suggest that pulses of sodium chloride-laden water during spring melt will have pronounced effects on stream communities, as will the continued release of high concentrations of sodium chloride from stream banks during summer and autumn months. Periphyton form the base of the food web in many creeks, being grazed upon by invertebrates, which in turn serve as forage for fish. Reduced fungal biomass resulting from increased sodium chloride concentrations may also impact invertebrates, forage fish and, ultimately, predatory fish communities.
Substantially lower chloride concentrations have been observed in creeks flowing through less densely populated areas with a smaller highway network. Moreover, at these low levels, which may be only 10-100 mg/L above background concentrations, many anthropogenic activities may be associated with this increase. For example, within the Waterford River Basin (Newfoundland), chloride concentrations averaged 70 mg/L at the Donovans Station (located in an industrial park), compared with 17 mg/L in an area that had no known industrial activities (Arsenault et al., 1985). High concentrations at the Donovans Station were related to industrial activities in addition to road salting and contamination from two road salt depots. Unfortunately, no study was specifically designed to investigate chloride concentrations immediately following road salt application and the following snowmelt.
Several studies conducted in the United States have shown similarly low chloride concentrations. One study shows how, in these low-impact streams, relatively high chloride pulses may occur. Boucher (1982) conducted a study investigating the impacts of road salt runoff on Penjajawoc Stream in Maine.
Chloride concentrations upstream of a commercial development averaged 5-15 mg/L over 1979-1982; within the development, concentrations averaged 10-50 mg/L. However, chloride levels rose during runoff events to reach a maximum concentration of 621 mg/L. Other studies have also associated the use of road salts with increased chloride concentrations. These concentrations have ranged between 10 and 150 mg/L in small urban and rural streams (Cherkauer and Ostenso, 1976; Gosz, 1977; Hoffman et al., 1981; Smith and Kaster, 1983; Prowse, 1987; August and Graupensperger, 1989; Demers and Sage, 1990; Mattson and Godfrey, 1994; Herlighy et al., 1998). Other factors may have affected some of the increases in average chloride concentrations in these streams.
However, where studies have been conducted to assess the effects of snowmelt on chloride levels, a clear pulse has been observed (Prowse, 1987). Prowse (1987) and Mattson and Godfrey (1994) also noted a relationship between the concentration of chloride in streams and highway density and length of the roadway that drains directly into the watercourse (e.g., where the road is adjacent to the watercourse).
The impacts on stream and creek communities of relatively small increases in chloride concentrations or larger increases over a short duration (hours to days) are uncertain.
Demers (1992) investigated the effects of elevated chloride levels on the aquatic macroinvertebrates inhabiting four streams near the town of Newcomb in the Adirondack region of northern New York. All four streams were located along a 2-km stretch of a state highway. Chloride concentrations in the creeks were measured weekly, yielding an overall mean concentration of chloride in upstream locations of 0.61 mg/L compared with 5.23 mg/L in downstream areas. Artificial substrates were placed in riffle or fast-flowing sections of the stream upstream and downstream of the highway. Samplers were within 50-100 m of the road and were left in place between April 22 and June 3, 1988, at which time they were recovered to characterize the colonizing invertebrates.
Benthic diversity was lower in downstream than in upstream sites. There was an increase of Chironomidae in downstream sites, while Perlodidae (a stonefly family) and Ephemerellidae (a mayfly family) declined downstream of the highway. A similar increase in chironomid and decrease in mayfly and stonefly dominance were observed in the Humber River (Ontario) between November, when road salt had not yet been applied, and the following March, after road salt had been applied (Kersey and Mackay, 1981).
Other studies have reported differences in benthic communities upstream and downstream of highways, but these differences were related to the larger differences in flow regime, sediment suspended load, habitat and inorganic and organic contaminants (Molles and Gosz, 1980; Kersey and Mackay, 1981; Smith and Kaster, 1983; Maltby et al., 1995). In the absence of carefully designed field studies, the impacts of small increases in chloride concentrations on stream, creek and river ecosystems remain unknown. An increase in mean chloride concentration from about 3 mg/L (world average from rivers) to 30-300 mg/L may affect succession of species with different salinity optima. Recent paleolimnological studies are showing that algal species, in fact, have distinct salinity optima, and changes in lake salinity are being inferred based on long-term changes in algal communities (Dixit et al., 1999).
Some researchers are beginning to investigate the role that water chemistry plays in determining community structure in rivers.
For example, Rott et al. (1998) investigated periphyton assemblages in the Grand River, Ontario, where chloride concentrations ranged from 7.7 to 85.0 mg/L and conductivity from 180 to 540 µS/cm. Canonical correspondence analysis showed that the largest portion of variability in species composition in the river over the study period could be explained by a seasonal gradient related to temperature and latitudinal gradients of nitrite-nitrate, conductivity and chloride. Phosphorus, ammonia, pH, turbidity and oxygen were of lesser importance.
Chetelat et al. (1999) investigated periphyton biomass and community composition in 13 rivers in southern Ontario that differed in their nutrient concentrations and conductivity. Biomass was strongly correlated with total phosphorus (r 2 = 0.56) and even more strongly correlated with conductivity (r 2 = 0.71). They suggested that the predominance of Cladophora at high-conductivity sites was related to the higher calcium concentrations at this site. Conductivity ranged from 65 to 190 µS/cm. Major differences in species composition were observed between low- and high-conductivity sites.
It is not clear whether periphyton communities were responding directly to chloride or whether chloride is an indicator of other perturbations in the watershed. However, given newer studies involving algal community composition in lakes (e.g., Dixit et al., 1999), it is probable that at least some of the responses are directly due to small differences in salt concentrations.
Ponds and lakes differ from creeks, streams and rivers in three important ways. First, ponds and lake systems do not have the pronounced and seasonally varying flow velocities characteristic of streams, creeks and rivers. Second, water residence time in ponds and lakes is longer, ranging from weeks to years. Third, ponds and lakes receive water from a variety of sources, and so their dilution capacity for contaminants can be higher than that for streams and creeks located near similar contaminant sources. Thus, chloride levels tend to be substantially lower in road salt-contaminated ponds, although the exposure time is generally much longer. The degree of contamination also depends on the degree of dilution that can be afforded. Generally, larger water bodies have a greater capacity for dilution.
There is a tremendous amount of variation in the degree to which road salt runoff affects the chloride concentration of urban ponds and lakes. However, in general, impacts on the Canadian environment have been modest, with chloride increases limited to the tens to hundreds of milligrams per litre rather than the thousands of milligrams per litre as observed for creeks in the Metropolitan Toronto area.
In Newfoundland, Arsenault et al. (1985) investigated chloride concentrations in five ponds in the Waterford River basin. Average concentrations ranged from 6 to 24 mg chloride/L, with the higher values associated with highway runoff and possibly runoff from a salt storage depot.
In Halifax, the First and Second Chain lakes have shown seasonal increases in chloride concentration, apparently related to road salting; chloride concentrations increased through the early 1980s to peak at 120-170 mg/L in the mid 1980s. Both lakes are shallow (mean depths of 4.1 and 3.2 m, respectively) and with a large urban watershed. More recently, Keizer et al. (1993) investigated the water quality of 51 lakes in the Halifax/Dartmouth Metro Area, comparing data collected in April 1991 with those collected in April 1980. The most pronounced change in these lakes was a near doubling in conductivity, with the majority of the increase due to increased concentrations of sodium and chloride. Whereas only 3 lakes had chloride concentrations greater than 100 mg/L in 1980, 12 had chloride concentrations exceeding 100 mg/L in 1991, with one lake reaching 197 mg/L.
In southeastern Ontario, Little Round Lake apparently became meromictic (or more strongly meromictic) in the 1950s as a result of road salt entering the lake from two highways and seepage from a salt storage shed (Smol et al., 1983). Chloride concentrations in the monimolimnion were 104 mg/L in about the early 1970s. This meromixis resulted in a reduction in seasonal nutrient exchange from deep to surface waters, a reduction in lake productivity and a change in algal community structure.
In Ontario, an ongoing study is providing evidence of elevated chloride concentrations in ponds next to highways. Chloride concentrations in ponds within 10 m of the highway average 88 mg/L (5-150 mg/L) for two-lane highways and 970 mg/L (84-2630 mg/L) for four-lane highways (Watson, 2000). Chloride concentrations remained elevated 50 m from the highway, with concentrations averaging 115 mg/L (35-368 mg/L) for two-lane and 1219 mg/L (50-3950 mg/L) for four-lane highways. Free and Mulamoottil (1983) reported chloride concentrations of up to 282 mg/L in Lake Wabekayne, a stormwater impoundment in Mississauga.
The Ontario Ministry of Transportation has unpublished data on the mean (May-October 1995) chloride concentrations in eight water bodies in the Humber River watershed (Scanton, 1999). Chloride concentrations averaged 10.6 mg/L at Lake St. George, 26.8 mg/L at Preston Lake, 47.0 mg/L at Wilcox Lake, 60.8 mg/L at Heart Lake, 107.9 mg/L at Claireville Reservoir, 110.3 mg/L at G. Ross Lord Dam, 174.0 mg/L at Lake Aquitanne and 408.9 mg/L at Grenedier Pond. The Humber River, like Highland Creek, is a major urban catchment basin. Road salt applications may be impacting the Humber River basin, as has been observed for the Highland Creek basin (Howard and Haynes, 1997).
More extreme examples of increased chloride concentrations in ponds and creeks in urban areas are to be found in U.S. studies. Cherkauer and Ostenso (1976) reported that Northridge Lakes in Milwaukee, Wisconsin, were affected by elevated chloride concentrations during winter ice cover. These lakes are shallow (9.2 m), artificial and interconnected. Chloride concentrations reached 2500 mg/L. Another extreme example is Irondequoit Bay, a small embayment located on the southern shore of Lake Ontario. This is a moderately deep bay (maximum depth 23.8 m, mean depth 6.8 m) surrounded by a dense network of highways. Chloride concentrations began increasing in this bay in the 1940s and continued increasing into the early 1970s. Chloride concentrations in deep waters reached more than 400 mg/L; vertical mixing of the water column was prevented (Bubeck et al., 1971). Remedial actions were developed; by the early 1980s, chloride concentrations in Ides Cove, within the bay, declined to a maximum of 140-150 mg/L, and vertical mixing was improved (Bubeck et al., 1995).
In Ann Arbor, Michigan, Fonda Lake was adversely affected by highway runoff that entered the lake via two drainage pipes. Chloride concentrations reached 177 mg/L in the mid 1960s, and vertical mixing was disrupted (Judd, 1969). After this was learned, the city reduced its salt usage in the subdivision and redirected runoff. However, chloride concentrations continued to increase because of increased inputs from the greater number of highways. Chloride concentrations reached a maximum of 720 mg/L in deep waters (Judd and Stegall, 1982). However, because the salt entered the lake in diffuse sources rather than through two drainage pipes, this salt mixed into the lake more effectively, and vertical mixing was not disrupted.
As already noted, few biological studies have accompanied these urban pond and lake studies. In addition to the Smol et al. (1983) study, Free and Mulamoottil (1983) noted a decrease in benthic invertebrate densities that occurred when bottom water became anoxic. Based on estimates of chronic toxicity, direct chloride effects on the individual species may be expected to occur at concentrations as low as 200 mg/L for individual species. However, recent studies involving phytoplankton suggest that for algae, at least, impacts may occur at even lower levels. This is discussed in the next section.
Chloride increases due to road salt have also been reported in rural ponds and lakes. As in the urban setting, the increase is related to pond/lake size and the degree of contact with the roadway.
In Terra Nova National Park in Newfoundland, chloride concentrations in Pine Hill Pond increased nearly 7-fold in spring from an average of 14 mg/L over April-December 1969 to 94 mg/L in April (Kerekes, 1974). This increase was related to road salt usage.
Underwood et al. (1986) surveyed 234 Nova Scotia lakes. The background chloride concentration was 8.1 mg/L, while the maximum concentration was 28.1 mg/L. The Nova Scotia Department of the Environment (Briggins et al., 1989) indicated that waters with chloride concentrations above 25 mg/L must have been receiving chloride inputs from anthropogenic sources. In watersheds with numerous highways, road salt would have been the probable source.
The Ministère des Transports du Québec (1980, 1999) investigated the impacts of road salt on Lac à la Truite, by Highway 15 and near Sainte-Agathe-des-Monts. The lake's drainage area, estimated at 728 ha, was affected by a 7-km stretch of highway, with a maximum slope of 5%. The lake had a surface area of 48.6 ha, a mean depth of 21.5 m and an estimated volume of 486 000 m3. In 1972, the average chloride concentration for the lake was 12 mg/L. This increased through the 1970s to reach a maximum concentration of 150 mg/L in 1979. This corresponds to an addition of approximately 67 068 kg of salt to the lake. The amount of salt applied to the roads was reduced, and chloride concentrations fell through the 1980s to reach 45 mg/L in 1990. While concentrations declined, they remained elevated at 42 mg/L at 3 m and 49 mg/L at 10 m.
In the United States, as in Canada, there is growing evidence of increasing chloride levels in lakes, with these increases being related, at least in part, to road salt application.
Several studies conducted in the United States provide further information on the probable impact of road salts on rural lakes and ponds that may be applicable to the Canadian environment. In Maine, Hanes et al. (1970) discussed the effects of road salt on a variety of aquatic ecosystems. They noted that farm ponds located near highways had sodium concentrations ranging from 1.4 to 115 mg/L and chloride concentrations ranging from <1 to 210 mg/L. Salt concentrations were higher in April 1966 than in July 1965. In 1967, chloride concentrations ranged from 1.4 to 221 mg/L, suggesting that chloride levels were increasing.
Sparkling Lake in the northern Wisconsin Lake District experienced an increase in chloride concentration due to contamination from road salt-laden groundwater (Bowser, 1992). The morphometry of Sparkling Lake was not reported. Road salt was initially applied to roads above the lake, and the salt apparently leached into the groundwater prior to entering the lake. Chloride concentrations in unaffected lakes and groundwater in the area were 0.3-0.5 mg/L compared with 2.61 mg/L in 1982 and 3.68 mg/L in 1991 in Sparkling Lake. The load of chloride required to produce such an increase in chloride concentration between 1982 and 1991 was calculated by Bowser (1992) to be 1200 kg/year.
Eilers and Selle (1991) compared conductivity, alkalinity, calcium and pH data collected at 149 northern Wisconsin lakes over 1925-1931 with data collected over 1973-1983. All parameters increased between the two time periods, with the greatest increases associated with increased land development on lake shorelines. Mean conductivity increased from 31.5 µS/cm in 1925 to 44.3 µS/cm in 1983 in developed watersheds and from 14.3 µS/cm in 1925 to 14.7 µS/cm in 1983 in undeveloped watersheds. Increased conductivity appeared to be associated with a combination of factors, including road salt, cultural eutrophication and changes in hydrology. The strongest increases were associated with lakes located near highways or paved roads.
The impacts of small increases in chloride concentration from road salt on lakes and ponds were not investigated in these studies. However, increased chloride (and other salts) concentrations traditionally have been viewed with concern (Beeton, 1969; Pringle et al., 1981) and actions taken to protect aquatic environments from these increased salt levels.
Small increases in chloride concentration are most likely to impact species composition and productivity. There is emerging evidence that microorganisms such as fungi, plankton and macroinvertebrates have salinity optima. Changes in salinity that alter the competitive balance between species should result in compositional changes at the base of the food web. Productivity may also increase. Evans and Frick (2001) addressed this issue by investigating what is known about the effects of salinity and conductivity on freshwater systems in general.
The addition of road salts to aquatic environments may enhance productivity through a variety of means. Road salt contains trace nutrients, including phosphorus (14-26 mg/kg) and nitrogen (7-4200 mg/kg). The addition of 100 mg chloride salt/L could result in an increase in phosphorus concentration of 1.4-2.6 µg/L and an increase in nitrogen concentration of 0.7-420 µg/L. Metals such as copper and zinc are also essential elements, and the addition of these metals to aquatic ecosystems may also enhance productivity. The systems that would be most vulnerable to a road salt-stimulated eutrophication would be low-productivity ecosystems. Paleolimnological studies in the Great Lakes have demonstrated enhanced productivity in the Bay of Quinte as early as the late 1600s, which was associated with land clearing and the increased inputs of nutrients (and trace metals) from the watershed (Schelske et al., 1983, 1985; Stoermer et al., 1985).
More recent limnological studies are investigating community structure in a wide variety of lakes and as a function of a number of limnological variables, such as conductivity, nutrient concentrations and major ions. This literature was briefly examined to determine whether additional information could be found on the responses of aquatic organisms to small gradients in salinity in addition to the expected responses to phosphorus concentrations. Since these studies were conducted in freshwater lakes, conductivity was dominated primarily by calcium carbonates. The following are noted.
Dixit et al. (1999) assessed changes in water quality in the northeastern United States using diatoms that had become deposited in lake sediments as indicators of change. The composition of diatoms in the surface sediments was related to current water quality parameters, including pH, chloride concentration and total phosphorus concentration. Total phosphorus and chloride optima were developed for 235 of the common species (Table 15). Mathematical models were then developed to predict the pH, chloride concentration or total phosphorus concentration of the study lakes based on the diatom assemblage (i.e., not individual taxa) found on surface sediments. The diatom assemblage was then examined at the 30-cm sediment depth in core samples to determine whether or not the water quality had changed since before the 1850s. Marked deterioration in water quality was noted in hundreds of lakes. In these lakes, phosphorus and chloride concentrations clearly were higher in modern than in preindustrial times. Moreover, there has been a marked increase in the number of eutrophic lakes in the Coastal Lowlands/Plateau. This increase in the trophic status of these lakes was associated not only with increasing phosphorus concentrations but also with increasing chloride ion concentrations. Sodium and chloride concentrations were highly correlated in these lakes, suggesting that increased chloride concentrations were associated with road salting. However, it was also recognized that agriculture, silviculture and urbanization can contribute to increases in chloride concentration. Future research, based on more detailed paleolimnological studies, will resolve these issues. However, it is already apparent that diatoms have optimal chloride concentrations, and deviations in these concentrations are associated with changes in species composition. This also must occur for other organisms, such as other algal groups, plants, benthic invertebrates and zooplankton.
Studies, while few in number, strongly suggest that small increases in chloride concentration will result in shifts in phytoplankton community composition in ponds and lakes. Furthermore, it is highly likely that standing stocks of plants and animals will also be affected. Productivity is likely to be enhanced through increased phosphorus, nitrogen and trace element inputs.
The actual number of road salt storage depots in Canada is unknown. Morin and Perchanok (2000) estimated that there were more than 1300 patrol yards at the provincial level and an unknown number of municipal and privately maintained yards. Snodgrass and Morin (2000) also noted that the design of these yards varies considerably across Canada. Some are well designed, with minimal salt leaching through the winter. Poorly designed yards may lose 22% or more of their salt through leaching. Salt concentrations in such leachate can exceed 80 000 mg/L (see Section 2.4). Ontario Ministry of Transportation (MTO, 1997) studies conducted in Ontario determined that 26% of patrol yards had well water concentrations that exceeded 500 mg/L. In another study, 69% of shallow groundwater samples had chloride concentrations that exceeded 250 mg/L. While leachate eventually will be diluted on entering a receiving water body, considerable damage may be inflicted on the aquatic environment on the dilution path. This is of particular concern for wetlands and small streams such as investigated by Dickman and Gochnauer (1978). Moreover, even after dilution, chloride concentrations may remain high enough to create meromixis in small lakes and/or to significantly raise their salinity.
Poorly designed or maintained salt storage depots clearly present a threat to the Canadian environment. Brief (hours to a few days) exposures to undiluted leachate are well within the toxic range for a variety of aquatic organisms. Where saline water collects for extended periods of time, as in a bog, sensitive marsh vegetation species may be killed and replaced by more salt-tolerant forms (Wilcox, 1982). Where saline water flows into lakes, the normal circulation patterns of the water column may be disrupted, and the lake becomes more strongly stratified. Relatively small increases in salinity to as little as 200-300 mg chloride/L fall within the chronic toxicity range for certain aquatic organisms.
Some studies have implicated road salt storage depots as having adversely affected chloride levels in the Canadian environment. Arsenault et al. (1985) related small increases in chloride concentrations at the Donovans Station on the Waterford River, Newfoundland, to salt storage depots in addition to potential impacts from the highway. In addition, Arp (2001) (Section 3.4.1.2) reported elevated sodium and chloride concentrations in creek and ditch water located downstream of a salt storage depot. Highest chloride levels occurred in summer. In southeastern Ontario, Smol et al. (1983) conducted a paleolimnological study on Little Round Lake, noting that the lake was oligotrophic prior to European settlement in the mid 1800s. With increased logging in the watershed, the lake became eutrophic, presumably because of increased nutrient and mineral inputs associated with land disturbance. A railroad and two highways were later built adjacent to the lake. The lake returned to oligotrophic conditions in the late 1960s as an apparent result of the lake becoming meromictic. The deep-water chloride concentration in the early 1970s was 104 mg/L, well above background levels. This meromixis prevented nutrients regenerated in the deep layer of the lake from being mixed back into surface waters. The elevated chloride concentrations (and meromixis) were related to highway runoff and seepage from a salt storage shed. Unfortunately, very few studies have been conducted around salt storage depots in Canada, despite the fact that there are more than a thousand such facilities in Canada and the fact that many clearly are contaminating groundwater and well water with chloride.
More limnological studies have been conducted around road salt storage depots in the United States. Such studies have shown a variety of impacts, which indicates that the proper management of such facilities is of environmental concern. Thus, the results of these studies have broad application to the Canadian environment, particularly for storage depots that are not properly maintained (i.e., covered and on an asphalt pad) and where best practices are not followed with respect to patrol yard washwater waste.
Wilcox (1982) investigated the effects of sodium chloride contamination by a road salt storage pile at Pinhook Bog in Indiana. In 1963, an uncovered sodium chloride storage depot was established overlooking the bog. Salt-laden runoff from the storage pile resulted in major alterations in the bog vegetation within a 2-ha area, as did runoff from the highway. The salt pile was covered in 1972; after winter 1980-81, road salt ceased to be stored at this site. Impacts were studied from 1979 to 1983. Sodium concentrations as high as 468 mg/L and chloride concentrations as high as 1215 mg/L were recorded in interstitial waters of the bog mat in areas of the strongest road salt impact (Wilcox, 1982). These readings were made in 1979, so higher concentrations may have occurred in earlier years. Chloride concentrations at control sites in 1980 and 1981 were 5-6 mg/L. The maximum single daily readings for salt-impacted locations were 1468 mg chloride/L in 1979, 982 mg chloride/L in 1980, 570 mg chloride/L in 1981 and 610 mg chloride/L in 1983. This indicates that decommissioning of the storage facility resulted in reductions in loadings and resulting concentrations in the bog, but that the chloride was subsequently largely retained in the wetland.
Native species, such as Sphagnum spp. and Larix laricina, were absent from the impacted areas of the bog, while salt-tolerant species, such as Typha angustifolia, invaded the bog. Salinity tolerances of various plant species were defined (Wilcox, 1982), and taxa were shown to be sensitive to sodium chloride concentrations in bog water as low as 280 mg/L (170 mg chloride/L) (Table 16). As salt concentrations decreased some 50% over 1980-1983, many endemic bog plants, including Sphagnum, recolonized the bog (Wilcox, 1982). Sphagnum began growing on low hummocks in areas where interstitial chloride concentrations had dropped to approximately 300 mg/L.
Tuchman et al. (1984) used core samples from lake sediments to investigate the impacts of historic salinization on diatoms of Fonda Lake, Michigan. A salt storage facility has been located adjacent to Fonda Lake since 1953; an asphalt pad was added at the salt storage facility in the early 1970s to reduce loss of salt. A reduction in diversity of algal species began in 1960. Diatom diversity reached a minimum in 1968, when a variety of salt-tolerant (or halophilic) taxa attained their highest relative abundance. In later years, diversity increased slightly and some halophilic taxa decreased in relative abundance, suggesting a decrease in salt loading to the lake. Unfortunately, lake salinity was not determined as part of this study. Zeeb and Smol (1991) extended the study of Fonda Lake to scaled chrysophytes. Mallamonas caudata, a chloride-indifferent taxon, dominated at all depths in the core. Mallamonas elongata, a widely distributed taxon found more commonly in eutrophic and alkaline waters, and Mallamonas pseudocoronata, a chloride-intolerant taxon, declined during the period when chloride concentrations apparently were highest.
Mallamonas tonsurrata, which occurs mainly in eutrophic lakes, became more abundant during this period. Major shifts in diatom and chrysophyte assemblages were associated with relatively small changes in salinity (i.e., from about 12 to 235 mg chloride/L, or about 20-387 mg sodium chloride/L).
Overall, these studies show that poorly designed or managed road salt storage depots can have a number of adverse impacts on the aquatic environment. This is of particular concern because biological monitoring programs have not been established around these yards to ensure that the local aquatic and terrestrial environments are not being damaged.
Snow collected from roadways to which road salts have been applied has been reported to contain chloride at concentrations reaching up to 2000-10 000 mg/L. Chloride concentrations in this range can be acutely toxic to a variety of organisms for exposures of 1 day or less. The majority of municipalities that remove roadside snow use surface dumps (Delisle and Dériger, 2000). The actual location of snow dumps is subject to significant variation and variable environmental regulation. The number of snow dumps is also unknown.
Snow deposited on low-lying lands can result in the increased chloride content of underlying soils and wetlands when the snow melts. Similarly, snow deposited on elevated areas eventually melts, transporting salt into the groundwater and into aquatic ecosystems, such as streams, wetlands and ponds.
Few studies have been conducted around snow dumps. However, elevated chloride concentrations (233-994 mg/L) have been reported in monitoring wells established to assess the impact of snow disposal on shallow groundwater (Morin, 2000). This contaminated groundwater could, in turn, contaminate groundwater-fed springs, as observed by Williams et al. (1999).
In British Columbia, Warrington and Phelan (1998) reported that the ionic composition of two lakes was shifted from carbonate dominated to chloride dominated as a result of salt-laden snow being pushed into the lakes from the highway. Salt-laden highway runoff may also have been a contributing factor. The biological effects were not investigated, but some could be inferred from recent paleolimnological studies of the effects of increased chloride concentrations on phytoplankton communities.
No other studies were located on the adverse impacts of surface snow dumps on aquatic ecosystems. However, given the probable thousands of snow dumps across Canada and the high chloride concentration in meltwaters, the potential harm to the Canadian aquatic environment of poorly located dumps is of concern. This is particularly so because there are not uniform regulations regarding snow dump practices and very few monitoring programs in place to ensure that the environment is not being harmed by these chloride releases. Unsuitable locations for snow dumps include wetlands, small ponds and lakes, and elevated areas that drain into small creeks. Groundwater contamination also needs to be considered.
3.3.3.4 Other effects
Road salts can have adverse effects on the aquatic environment in addition to their apparent direct effects on species composition and abundance. Chloride salts can affect aquatic systems through interactions with abiotic components of the environment. Notably, chloride salts tend to be more soluble than carbonate salts and can thus, through various reactions, enhance the mobility of trace metals in aquatic ecosystems. Increased salt loadings can affect water density, thereby affecting mixing processes in lakes and thus disrupting the ecological functioning of that ecosystem.
Increased concentrations of chloride salts in surface water systems can lead to the release of metals from sediments and suspended particulate matter. By competing for particulate binding sites, sodium chloride acts as an enhancer of dissolved and potentially bioavailable trace metals, such as cadmium, copper and zinc (Warren and Zimmerman, 1994). Cadmium, copper and zinc are acutely toxic to aquatic organisms at concentrations as low as 1 µg cadmium/L (rainbow trout), 6.5 µg copper/L (Daphnia magna) and 90 µg zinc/L (rainbow trout) (CCME, 1991). In addition, since road salt contains a variety of contaminants, including metals such as copper (0-14 µg/g), zinc (0.02-0.68 µg/g) and cadmium (MDOT, 1993; Maltby et al., 1995), snowmelt containing road salt is a potential source of metal contamination to receiving water bodies. Furthermore, because the highway surface contains a variety of metal contaminants from the automobile itself (lead, copper, zinc and cadmium), road salt can facilitate the mobilization and transport of these contaminants into the aquatic ecosystem (Maltby et al., 1995). These metals may accumulate in various reaches of streams on sedimentary particles (Maltby et al., 1995) or in stationary waters such as ponds. There, they have the potential to exert toxic effects, especially through processes involving sediments.
High concentrations of salts in sediment pore water were shown to augment the concentrations of dissolved heavy metals (e.g., cadmium) and resulting toxicity to a benthic invertebrate, Hyalella azteca (Mayer et al., 1999). Wang et al. (1991) found that 709 mg chloride/L (or 0.02 mol/L) substantially enhanced the release of mercury from freshwater sediments. Mercury can be acutely toxic to invertebrate species and fish in concentrations as low as 0.002 mg/L and 0.02 mg/L, respectively (CCME, 1991) and, when present in its methylated form, accumulates in the tissues of fish. Sodium chloride also enhances mercury mobilization from soils (MacLeod et al., 1996).
Road salt can also affect ponds and lakes by reducing their capacity for vertical mixing. Most lakes undergo vertical mixing, resulting in an exchange of deep and surface waters. Such exchanges are important for transferring oxygen-rich surface water to the deeper regions of the lake. In the absence of such exchanges, deep waters can become anoxic. Vertical exchanges are also important in transferring nutrients regenerated in the deeper regions of the lake to the surface, where they become available to the phytoplankton community. Vertical mixing is driven by the winds, which mix surface waters down to depths dependent on lake fetch, surrounding topography and water temperature. Vertical mixing is also driven by changes in water temperature that affect the density of water. Since water has a maximum density at 4°C, spring warming from freezing to 4°C is accompanied by an increase in water density, promoting the vertical exchange of warmer waters with colder and less dense deep waters. Lake cooling in autumn and lake warming to 4°C in spring are accompanied by a change in water density, which promotes the vertical exchange of waters.
Meromixis occurs in lakes when conditions develop that prevent the vertical exchange of surface and deep waters (Wetzel, 1983). This occurs when a sufficient gradient exists in salinity to override the effects of seasonal variations in water temperature on density and lake mixing. Road salts, by entering lakes through surface flow (overland runoff, ditches, streams) or groundwater discharge (seeps, springs), have the potential to impair the normal circulation of lakes. Small, moderately deep lakes will be the most vulnerable to such meromixis, especially in areas of heavy road salt application. Larger lakes are less vulnerable, because the intruding saltwater plumes experience greater dilution as the denser salt-laden water flows along the lake floor towards the deeper regions of the lake. In addition, larger lakes have greater fetches and hence more powerful wind-driven currents and other water exchanges.
The formation of meromixis can have a number of impacts on lakes. The low oxygen conditions that develop below the chemocline can result in the loss of all but the most resilient of deep-water benthic species. Zooplankton may be excluded from their deep-water daytime refuges, being forced to live in the brighter surface layers, where they may be more vulnerable to size-selective fish predation. Hypolimnetic fish such as lake trout may also be adversely affected. The onset of lake meromixis will affect sediment-water exchanges. Phosphorus and various metals are more readily released from poorly oxygenated than from well-oxygenated sediments (Wetzel, 1983). This increase in phosphorus release from the sediments may enhance the productivity and eutrophication of the lake, particularly if there is sufficient exchange at the chemocline. Alternatively, if there is limited exchange at the chemocline, production may be reduced, and a eutrophic lake may become oligotrophic (Smol et al., 1983).
3.3.3.5 Conclusions for aquatic ecosystems
Short-term acute toxic effects have been associated in the laboratory with high chloride concentrations. However, as exposure time increases, sensitivity to chloride increases.
For example, the 4-day LC50 is 1400 mg/L for Ceriodaphnia dubia (a cladoceran) and 1261 mg/L at 9 days (Cowgill and Milazzo, 1990). Exposure to such high lethal concentrations would most likely be associated with poorly managed salt storage depots, inappropriately placed snow dumps on wetland areas, roadside ditches in areas of high use, contaminated groundwater springs and small watercourses in heavily populated urban areas (e.g., Metropolitan Toronto) that have a dense network of highways and road salt application.
Longer-term toxicity occurs at substantially lower levels, estimated to begin at concentrations as low as 210 mg/L. Chloride concentrations at these levels have been observed in a variety of urban creeks, streams and lakes, although primarily in highly populated areas and small water bodies near highways with high use. Such levels are also expected to occur in the vicinity of poorly managed road salt storage depots and in inappropriately placed snow dumps, e.g., on wetlands, in small ponds, near headwater creeks and near contaminated groundwater-fed springs. Aquatic ecosystems experiencing such chloride levels are expected to be impaired.
A family of curves was prepared based on chloride toxicity data following 4-day, 7-day and predicted long-term (chronic) exposures (Figure 17). The toxicity data used to develop Figure 17 can be found in Evans and Frick (2001). One-day and less than 1-day data are excluded because studies were few in number. Data are plotted on a linear-log scale with 95% confidence intervals around each curve. A log-logistic model was used in the curve fitting (Environment Canada, 1997b). Based on the predicted data, it can be expected that 5% of species would be affected (median lethal concentration) at chloride concentrations of about 210 mg/L (Table 17), while 10% of species would be affected at chloride concentrations of about 240 mg/L.
Figures 18 and 19 show various chloride concentrations in the Canadian aquatic environment juxtaposed with concentrations causing adverse biological effects. The environmental chloride concentrations specified are assumed to arise from road salt contamination. Unless otherwise specified, all values for effects relate to lethality.
Figure 18 presents data on short-term exposure pertinent to a period of 5 days or less. The concentrations given are for rivers and creeks where chloride peaks associated with events such as freeze/thaw occurrences would not be expected to last for more than a few days at any given sampling location. The biological effects specified are for toxicity tests with an endpoint of 5 days or less (e.g., 4-day LC50).
Figure 19 presents data pertinent to exposure for a period of more than 5 days. The environmental concentrations given are mostly for lakes and ponds where chloride levels are generally expected to remain stable over a longer period. The biological effects specified are for toxicity tests with an endpoint of more than 5 days (e.g., 33-day LC50).
As previously noted, there is growing evidence of a widespread increase in chloride levels in rivers and lakes in the northeastern and midwestern United States, including lakes in a non-urban setting. For example, Dixit et al. (1999) investigated 257 lakes and reservoirs in the northeastern United States and noted that while 34% of the reservoirs and 6% of the lakes had background chloride concentrations of >8 mg/L prior to the 1850s, this had increased to 46% and 18%, respectively, by present times. Such increases are related, in part, to the number of highways in the area, land disturbance and road salt use. Furthermore, researchers are beginning to note changes in algal communities as these chloride levels increase from background levels of 1.8-3.6 mg/L to over 7 mg/L (Dixit et al., 1999). Studies conducted in Canada, although less detailed, are also pointing towards increased chloride levels, in the range at which changes in algal composition may be expected, in small ponds and lakes located near certain highways and in a variety of streams and lakes in an urban setting. However, it is important to note that the vast majority of these studies are limited to areas of heavy road salt usage, primarily southern Ontario, the Maritimes and Quebec.
1 Data are from Figure 17.
The U.S. EPA (1988) developed water quality guidelines for chloride and concluded that, except possibly where a locally important species is very sensitive, freshwater organisms and their uses should not be affected unacceptably if:
- the 4-day average concentration of chloride, when associated with sodium, does not exceed 230 mg/L more than once every 3 years on average;
- the 1-hour average chloride concentration does not exceed 860 mg/L more than once every 3 years on average.
They noted that these criteria will not be adequately protective when the chloride is associated with potassium, calcium or magnesium, and that because animals have a narrow range of acute sensitivities to chloride, excursions above this range might affect a substantial number of species. Evans and Frick (2001) also found evidence that potassium chloride and magnesium chloride salts were more toxic than sodium chloride. In addition, fish appeared to be less sensitive to calcium chloride than to sodium chloride, and the converse was true for invertebrates, although the data sets were small. Although the focus of the assessment was primarily on sodium chloride, they concluded that potassium chloride and magnesium chloride salts appear to be as toxic as or even more toxic than sodium chloride.
To conclude, it is considered that high concentrations of chloride associated with road salts may have immediate or long-term harmful effects on surface water systems, based on several exceedences of effect thresholds in the environment and on field evidence of ecosystem population effects. These effects are most likely to occur at improperly managed road salt storage depots, at inappropriately placed snow dumps and in small watercourses along a dense network of highways. At lower concentrations, increased chloride concentrations may affect community structure, diversity and productivity. Elevated chloride levels have been shown to increase metal bioavailability; by affecting density gradients in lakes, road salts may have a major impact on lake ecosystems, notably in terms of depth-dependent availability of oxygen and nutrients. Most instances in which this occurs are in areas of high road salt usage, primarily southern Ontario, Quebec and the Maritimes.
Figure 17 Experimental acute toxicity and predicted chronic toxicity for aquatic taxa (from Evans and Frick, 2001)

Figure 18 Representative short-term chloride concentrations in the Canadian aquatic environment associated with contamination by road salts and concentrations causing adverse biological effects following brief exposures


Figure 19 Representative long-term chloride concentrations in the Canadian aquatic environment associated with contamination by road salts and concentrations causing adverse biological effects following prolonged exposures


3.4 Soils
3.4.1 Soil salinization resulting from road salt application
This section summarizes the information presented in the report prepared by Morin et al. (2000) on the extent of soil salinization due to road salt application. The report presented soil maps for all provinces in Canada. This mapping effort is complemented with a few case studies designed to show how the salinity of soil solutions changes dynamically in salt-affected areas, including short distances from point and line salt sources, and with season. The salt ions examined are sodium, calcium, sulphate and chloride. Contributions due to other ions such as potassium and magnesium are neglected, because these generally contribute little to overall soil salinity in most locations.
Mapping of road salt inputs (annual inputs) and salt concentrations in soils was done as spatial averages compiled for Level 2 watersheds, as spatial averages at the municipality level and as spatial averages at the road level (mainly principal roads and expressways). These three levels of analysis were done because of the general connection between soil salinity and surface water salinity, i.e., most surface waters receive direct input from soils as a result of runoff, interflow, soil percolation and baseflow. Similarly, downslope soils often receive seepage water from higher surface water locations. Gradual dissolution of easily weathered soil and rock minerals further contributes to downslope soil salinization, as do agricultural and horticultural fertilizer applications and seepage from domestic and industrial sewage. Evaporation of water from dry soils in arid regions such as the Prairie provinces also contributes to soil salinity, which, in the extreme, leads to above- and below-ground salt accumulations, especially in landscape depressions and on hillslope seeps. Industrial activities, especially those associated with salt mining and with the sodium hydroxide-based extraction of fossil fuels from tar and oil sands, add further to environmental salt loading in areas specific to these activities. This section, however, focuses on the extent to which road salt applications in combination with atmospheric deposition contribute to the overall salinity in soils next to roads, and how these applications may affect the general background salinity of water as it reemerges from the soil in various downslope locations. In general, calculated average Level 2 soil salt concentrations from road salt loadings are expected to be lower than area-wide municipal averages, and area-wide municipal averages are expected to be lower than roadside soil averages. Hence, a three-way mapping effort was done to estimate:
- expected average salt concentrations in soil solutions based on total road and atmospheric salt inputs per Level 2 watershed;
- expected average salt concentrations in soil solutions based on total road and atmospheric salt inputs per municipal area; and
- expected average salt concentrations in soil solutions based on total road and atmospheric salt inputs in immediate roadside vicinities.
To quantify these averages, information contained in various national, provincial and municipal databases was compiled to determine:
- local average annual road salt applications, by municipality and county;
- local average annual atmospheric deposition regarding precipitation and wet sodium, calcium, chloride and sulphate ion deposition (a dry deposition database does not yet exist; including dry deposition further adds to overall salt loadings, especially in the Atlantic region on account of sea spray);
- average annual runoff; and
- local soil depth, clay content and organic matter content.
For the mapping effort, seasonal aspects are not considered, but soil salinities would vary depending on the volume of road salt loadings, weather and topography. Some of the seasonal aspects are addressed in this section by way of special case studies that deal with soil and surface water salinity in downslope locations from a salt depot.
3.4.1.1 Definitions and equations
Compiled data on atmospheric sodium, calcium, chloride and sulphate ion loadings refer to volume-weighted ion concentrations (wet deposition only), either in mg/L or in meq/L. Salt loadings, in contrast, are generally expressed in terms of g/m2 per year or kg/ha per year. In this report, atmospheric and road salt loadings are converted into total dissolved salt concentrations in soil solutions by dividing the calculated combined annual salt loadings by the expected annual soil percolation rate, where:
soil percolation rate (mm/year) = precipitation rate (mm/year)– actual evapotranspiration rate (mm/year) – surface runoff (mm/year)
For simplicity, annual soil percolation rates (mm/year) are estimated to be equivalent to stream discharge rates (mm/year), where stream discharge rates are obtained from watershed-specific hydrometric discharge observations. For the mapping effort, expected soil percolation rates are therefore set equal to stream discharge rates. Maps located in Morin et al. (2000) depict:
- millimetres of stream water discharge at a national scale, as obtained from Natural Resources Canada; and
- cubic metres of stream water discharge per Level 2 watershed (as obtained from the national stream discharge map).
Watersheds with highest runoff coefficients occur along the Pacific and the Atlantic coasts. Watersheds located in the southern part of the Prairie provinces have the lowest quantity of runoff to dissolve and dilute the salts. Areas with low rates of runoff are important to identify because these areas have high road salt concentrations in runoff.
For the mapping of these parameters, it was also essential to obtain national soil survey information on:
- soil clay content (as a % of mineral portion of soil) (particulate matter <2 mm);
- soil organic matter (in %); and
- cation exchange capacity (in meq/100 g oven-dry soil).
The soil solution estimates for total dissolved salt loadings and related ion concentrations were used to calculate:
- total cations and total anions (in meq/L);
- soil electrical conductivity (in mmho/cm or mS/cm);
- osmotic potential (in bar);
- sodium absorption ratio (dimensionless);
- exchangeable sodium ratio (dimensionless);
- exchangeable sodium percentage (in %);
- exchangeable sodium content (in meq/100 g oven-dry soil);
- soil clay and silt dispersion (as % of soil clay content); and
- relative changes in soil hydraulic conductivity (dimensionless).
Likely salt-induced effects on soils include substantial lowering of soil osmotic potentials, increased soil swelling, reduced soil stability (loss of soil structure), decreased soil permeability, increased potential for soil erosion, increased clay and silt dispersion, and increased turbidity in surface waters. All of these impacts get worse with increasing sodium content in the soil. This is because increased sodium ion contents decrease the affinity that soil particles have for each other on account of the strong affinity between sodium ions and water molecules. To some extent, salt-induced effects depend strongly on soil clay mineralogy (e.g., soil swelling and shrinking in soils containing large portions of montmorillonite). In some other respects, salt-induced effects are independent of soil mineralogy and are simply affected by electrolyte-mediated affinity among individual particles in the soil or in suspension (e.g., affinity is regulated by interparticulate electrolyte forces that determine the degree of particle coagulation and dispersion; see Ali et al., 1987). With increasing soil dispersion (e.g., when salinized soils receive low electrolyte irrigation water and are then subjected to water and wind erosion), not only does dispersion include fine soil particles, but these particles may also be carriers for environmental contaminants such as nutrients, heavy metals and microbiota by way of water-and wind-induced soil erosion.
Soils that have a relatively high clay content and continuously or periodically receive high sodium inputs will likely show the strongest impacts in the long run (i.e., should receive a high salt hazard rating). To map areas with a high salt hazard rating, a simple salt hazard index was obtained by setting:
salt hazard index = exchangeable sodium ratio x soil clay content (in %)
In this way, areas with highest hazard ratings were located for southern Ontario near the Greater Toronto Area and in southern Quebec around the Island of Montréal (Figure 20). Other urban areas in southern Ontario and southern Quebec also have high ratings.
Additional areas are found in the southern portions of the Prairie provinces, where soil salinization on account of natural processes is a major regional concern (see Eilers et al., 1995).
3.4.1.2 Case studies
Three case studies were conducted at Kejimkujik National Park, Nova Scotia, along select streets in Fredericton, New Brunswick, and in a forested area next to Fredericton's municipal salt depot (University of New Brunswick woodlot). These studies focused on:
- providing numbers for the general turnover rate of sodium ions in the soils of headwater basins at Kejimkujik National Park (an environment that is essentially free of road salts but is influenced by sea spray);
- assessing the status of soil salinity underneath lawns next to the curb of select roads in Fredericton during the fall season;
- examining how the salinity of surface water changes with distance from Fredericton's salt depot, by season; and
- examining the salinity of ditch water along the Fredericton-New Maryland Highway.
The results of these case studies indicated that the ability of soils and watersheds in the Kejimkujik National Park to retain incoming sodium and chloride ions is very low, i.e., sodium and chloride ions are readily flushed through the soils and through the basins of the park. Ion concentrations are highest in soils and streams during early fall, and lowest concentrations occur at the time of snowmelt.
Along the city streets of Fredericton, generally little sodium remains on the ion exchange sites in soils next to the road, as observed in early fall. The data indicate that ion exchangeable sodium is particularly low along the most frequently travelled roads at this time of year. Furthermore, exchangeable sodium concentrations are lower next to curbs than 2-3 m away from curbs. This difference indicates that repeated and substantial traffic splashing and road runoff during spring, summer and fall are flushing sodium out of the roadside soils.
Compared with Kejimkujik National Park, background sodium and chloride ion concentrations in soil solutions and in surface waters are somewhat smaller at the University of New Brunswick's woodlot (2 mg/L versus 3 mg/L, respectively). Chloride concentrations in ditch and stream water around the University of New Brunswick's woodlot, however, vary by location. Arp (2001) measured the electrical conductivity (later related to chloride concentrations) of ditch water along the Fredericton-New Maryland Highway and Bishop Drive for representative days in June, July, October (1999) and January (2000). Results are summarized in Figure 21 by way of box plots, according to the hydrological condition at each point of measurement. Ditches in this analysis were categorized according to the following three location characteristics:
- well-drained locations where the ditches tend to run dry (crest locations);
- poorly drained areas where water accumulates and becomes stagnant; and
- areas of continuous water flow, i.e., brook road crossings (culverts).
Figure 21 shows that electrical conductivities of ditch water peak in July, especially in areas where water is not stagnant.
Figure 20 Areas with low to medium to high road salt application hazards, based on product of soil clay content with exchangeable sodium ratio (from Morin et al., 2000)

At stagnant locations, electrical conductivity readings remain fairly high regardless of season and tend to be highest in June and July, but are lowest and generally least variable in the fall. Where streams, brooks and/or ditch water cross underneath the highways (by way of culverts), conductivities are lowest, at or near background level, due to continuous flushing - i.e., ditch water, as it enters streams and brooks, is quickly diluted, on a year-round basis. However, where ditch water drains into poorly drained, water-accumulating depressions and where the water turnover rate is low, dilution does not occur, and electrical conductivity levels (and, therefore, salinity levels) remain elevated year-round.
Chloride concentrations are highest at and near the salt depot, ranging from 300 to more than 1000 mg/L. Listed in Table 18 are chloride concentrations for stream and pond water with increasing stream catchment area of the Corbett Brook downstream from Fredericton's salt depot at various times of the year (May, June, July, October, January). At first, considerable dilution occurs in the nearby "Larch Swale Pond" (0 km in Table 18), which receives water from the 172-ha headwater system. Below this area, dilution continues either gradually or in jumps, with each jump being specific to the catchment area of each of the adjoining brooks. For example, at about 2 km away from the depot, Corbett Brook is joined by the O'Leary Brook. At that point, dilution increases sharply, but in proportion with the combined catchment area for O'Leary Brook and Corbett Brook.
Figure 21 Box plot showing 10th, 25th, 75th, and 90th percentile plus individual data above and below the 10th and 90th percentile for ditch water electrical conductivity and chloride concentrations along two highways near Fredericton's salt depot, outside the depot's catchment area for representative days in June, July, October 1999, and January 2000. "Well-drained", "cross-flow", and "stagnant" refer to three conditions: ridges from which water drains, stream/road crossings, and ditch depressions with no visible outflow, respectively (from Arp, 2001).

Season has a strong effect on electrical conductivity measurements and the related stream dilution factor. In principle, conductivity measurements are related to stream discharge rates: high discharge rates imply increased dilution, and therefore decreased conductivity values; low discharge rates imply less dilution, and therefore increased conductivity values. For intermittent stream flow conditions (as may occur in summer and early fall), the overall influence of the salt depot appears to be confined to the vicinity of the salt depot, while areas further removed from the salt depot receive less salt-enriched water. This "apparent" dilution with increasing catchment area at lower stream locations is greater during times of intermittent flow (such as July) than during times of continuous flow. In July, concentrations greater than 250 mg/L were observed up to 500 m from the depot and were still 100 mg/L at 1250 m from the depot.
3.4.1.3 Discussion
Road salt application effects on physical and chemical soil parameters such as osmotic potential, electrical conductivity, soil permeability, soil dispersion and exchangeable sodium ratio (see Morin et al., 2000, for detailed information on other physical and chemical parameters) are all calculated to be highest in areas next to salt depots and roads receiving salt inputs. At the municipal level, impacts of soil waters on downslope positions can be expected to vary greatly depending on the density of the road network, on local conditions, on the actual location of road salt applications and on the physical condition of salt storage facilities. For example, in highly paved urban areas, road salt applications are expected to produce higher overall salt concentrations in soils and surface waters than in less developed areas. Williams et al. (1999), for example, showed that salt concentrations in springs within the Metropolitan Toronto area increase as predominant land use changes from rural to urban. These authors also showed that:
- these salt concentrations peak during the time of salt application;
- some of these peaks have chloride ion concentrations in excess of 1000 mg/L; and
- secondary salt concentration peaks occur in midsummer (thereby supporting the results of one of the above case studies), with highest chloride ion concentrations in excess of 200 mg/L.
Table 18 Modelled electrical conductivity values and chloride concentrations for Corbett Brook, downstream from Fredericton's salt depot Catchment area (ha) Distance Electrical conductivity (mS) Chloride concentrations (mg/L)

Salt concentrations in surface water, groundwater and soil solutions, therefore, may be high at the time of road salt application, but elevated concentrations may recur later in summer during periods of drought when water evaporates and when salty water from, for example, subsoils seeps back into downslope locations. During times of high precipitation, low electrical conductivity water will help to flush briny water from these areas. Along roadsides, frequent splashing during rain events may further accelerate the displacement of briny water and the return to low soil exchangeable sodium percentage values, but this can occur effectively only in soils of high permeability in areas where water does not accumulate (i.e., does not stagnate for significant parts of the year).
Underneath paved surfaces (as in urban areas), soils may not benefit from seasonal salt flushing. Salt in these locations likely may remain concentrated or may accumulate even more with additional salt applications, because these locations are, at least in part, outside the paths of percolating soil water. These locations, therefore, become a source of soil and surface water salinity, by way of either slow seepage or high-salinity flushes during times of pavement repair or unusually wet weather.
The overall salt impacts on soils need to be evaluated in terms of landscape-level source/sink positioning. For example, the location of the roadside in relation to the road (above road, below road, same level as road, upwind from road, downwind from road) and terrain conditions along roads (e.g., open versus forested, well-drained versus poorly drained) have to be considered. Typically, salts are spread from road to roadside by vehicle spray, dust and runoff. Accordingly, soil salinity is expected to increase from ridge tops (recharge areas) to poorly drained depressions.
The process of salt dilution through subsequent precipitation and traffic splashing is important in terms of allowing much of the Canadian soil roadscape to recover partially or completely from seasonal road salt applications. Apparently, splashing from roadsides accelerates the leaching loss of winter-accumulated salt from the roadsides, as noted in one of the above case studies. However, during times of high exchangeable sodium percentage values and low electrical conductivity splash water, this effect could have deleterious effects on soil aggregation, e.g., soil clay and silt dispersion, which, in turn, can lead to reduced soil infiltration rates and thereby enhanced soil erosion by water and enhanced dispersion of soil particles by wind. This effect would be highest where roadsides are not covered by vegetation.
Figure 22 shows expected average values for total dissolved solids, osmotic potential and electrical conductivity of soil solutions and surface water at the Level 2 watershed level and at the municipal area level. An inspection of the total dissolved solids results indicates that these values are all higher than the "background" levels that would be expected due to local atmospheric deposition and subsequent evapotranspiration alone. These area-averaged concentrations, however, are far from being of concern, except for highly urbanized areas. As expected, calculated values for total dissolved solids, osmotic potential and electrical conductivity increase with decreasing area averaging.
Values for total dissolved solids, osmotic potential and electrical conductivity for soil solutions and surface water for roadway rights-of-way were calculated. These values are based on provincial road salt loading data, by maintenance district. Overall, these calculations indicate that areas immediately surrounding the right-of-way tend to have high annual averages for total dissolved solids, osmotic potential and electrical conductivity. Seasonally, these values would be higher through periods of salt application and substantially lower during periods of high flushing. High peaks, however, can be expected to return during midsummer in areas of water accumulation and may remain high in stagnant water pools or depressions with limited surface runoff, as discussed above.
The map in Figure 23 suggests that certain areas in Ontario and Quebec may have roads with elevated exchangeable sodium percentage values. These values are primarily associated with areas that have high road salt loadings. Figure 23 suggests that certain areas in the Prairie provinces may also have high exchangeable sodium percentage values. While roads in the Prairie provinces have low road salt loadings compared with most municipal areas in Ontario and Quebec, there is less precipitation to dissolve and dilute the salts. The situation is such that many soils in southern Alberta and southern Saskatchewan and some soils in southern Manitoba are already saline due to natural causes, especially in depressions and near wetlands and drainage courses. Here, road salts would add to the total electrolyte loads of soils, as these would subsequently accumulate in the depressed areas next to the roads. In general, the spatial patterns of the background levels of electrolyte ions in the soil solutions based on atmospheric deposition alone match the spatial patterns of soil salinity as displayed by Eilers et al. (1995) fairly well, thereby confirming the simple mass balance protocol of the model that produced the maps of this report.
Figure 22 Estimates for total dissolved solids (TDS) and electrical conductivity (EC) of average soil solution and surface waters, by level 2 watersheds and by municipal boundary, Ontario and Quebec (from Morin et al., 2000)
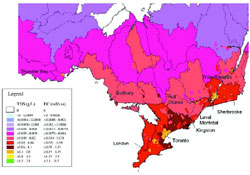
Increased soil salinization through road salt applications may affect soil biota (macro- and microflora and macro- and microfauna). For a simplified summary of expected plant sensitivities and related species-specific threshold tolerances to falling soil osmotic potentials, refer to Figure 24. Figure 24 associates the sensitivity of crops to soil salinity by demonstrating how crop yield decreases with increasing levels of electrical conductivity. The dashed boxes in this figure show the percentage of provincial roadways with certain levels of estimated soil salinity. Data in this graph suggest that salinity in soil solution alongside the majority of provincial roads may adversely affect many shrub and tree species. This graph also suggests that certain roadsides may have levels of soil salinity that can adversely affect salt-tolerant plants like wheatgrass. Impacts on terrestrial vegetation are discussed more comprehensively in Section 3.5.
Figure 23 Exchangeable sodium percentage (ESP) along roadsides, by provincial road maintenance district (from Morin et al., 2000)
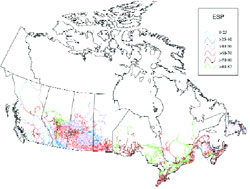
Road salt effects on soils are expected to be highly dependent on the extent of vegetative cover. Soils that are bare likely suffer the worst impacts in terms of surface crusting, surface runoff, and clay and silt dispersal by water and wind. Soils that are originally covered by vegetation will also suffer impacts once the surface vegetation reacts negatively to high negative osmotic potentials in the soil.
Here, gradual loss of surface vegetation will lead to increased mineral soil exposure; this leads to gradually decreasing levels of soil organic matter and, therefore, increasing soil dispersion, and thus decreasing soil permeability and increasing soil erosion. Proper vegetation management, salt application and road maintenance practices would obviously go a long way to reducing any such effects. Additional details on soil salinity and many other aspects related to soil management and soil conservation can be found in, for example, Anon. (1992), Holms and Henry (1982), Steppuhn and Curtin (1992) and Acton and Gregorich (1995).
Other impacts of road salts not covered in this section deal with salt-induced release of heavy metals (e.g., zinc, copper, cobalt, mercury, cadmium) and base cations (calcium, magnesium, potassium). All of these may have positive or negative effects on soil flora and fauna, including:
- encouraging the growth of certain species in otherwise nutrient-poor ecosystems;
- enhancing the loss of plant nutrients such as calcium, magnesium and potassium due to ion exchange with sodium ions; and
- heavy metal mobilization in soil and aquatic habitats, depending on local circumstances.
Heavy metal mobilization occurs through formation of water-soluble chloride-metal cation complexes, thereby rendering many heavy metal cations more phyto-available. This would increase the entrance of heavy metals into the local food chain by way of plant uptake. The plants themselves could experience heavy metal toxicity symptoms in the worst-case scenarios. For an example of the processes involved, see Smolders and McLaughlin (1996) regarding the case of chloride-enhanced cadmium mobilization and subsequent phyto-uptake. For additional details regarding soil salinization effects, see, for example, Bresler et al. (1982), Eilers et al. (1995), Butler and Addison (2000) and Cain et al. (2001).
3.4.1.4 Conclusions
Application of road salts can result in deleterious effects on physical and chemical parameters of soils, especially in salt-affected areas that also suffer from general salt, soil and vegetation management neglect. Highest effects are calculated in areas that would be directly impacted by salt depots and along roadsides, especially in poorly drained depressions.
Electrolyte-related effects impact on soil structural stability, soil dispersion, soil permeability, soil swelling and crusting, soil electrical conductivity and soil osmotic potential. Surface waters downslope from salt-affected soils are also affected by briny seepage water or briny runoff with salt dispersed sediments. All of this has, in turn, abiotic and biotic impacts on the local environment. Abiotic impacts primarily deal with loss of soil stability during drying and wetting cycles and during periods of high surface runoff and wind; biological impacts primarily deal with osmotic stressors on soil macro-and microflora and macro- and microfauna and salt-induced mobilization of macro- and micronutrients, which, in turn, affect flora and fauna in situ and in downslope seepage areas.
3.0 Summary of Critical Information and Assessment of "Toxic" under CEPA 1999
3.4 Soils
3.4.2 Biological effects of road salts on soils
A comprehensive review of the effects of salt on soil microflora and microfauna was compiled by Butler and Addison (2000). There appear to be very few data dealing with road salt exposure and soil microbial communities and/or their activities. Thus, available literature on the metabolic adaptations required for microbial salt tolerance and on the effects of high soil salinity on agriculturally important processes was reviewed and addressed for this assessment, although the originators of the quoted data had purposes very different from those of this document. An additional difficulty is the varied ways of reporting salt exposures in the literature. Soil-to-soil comparisons are typically based on electrical conductivity, water potential or osmotic potential, not added salt concentration, because the ionic composition of different soil solutions varies. However, salt concentrations (mass/liquid volume) are more usually reported in the microbiology literature. Studies involving soil fauna typically report salt inputs as amount per unit area.
3.4.2.1 Ecological role of soil biotic communities
The biological communities of soils and sediments are responsible for the degradation and mineralization of organic matter and cycling of elements (e.g., carbon, nitrogen, phosphorus, sulphur) in these environments. In terrestrial systems, there is only a minor direct contribution to primary production by photo- and chemolithotrophic microbes. However, experiments have indicated that the primary productivity of higher plants is greatly enhanced by the activities of the below-ground microbial and faunal biota (Faber and Verhoef, 1991; Setälä and Huhta, 1991; Setälä et al., 1998). Mycorrhizae are features of nearly all terrestrial plants, with the fungal partner of this symbiotic plant-fungus relationship contributing to nutrient (especially phosphorus) uptake and translocation and potential protection against plant pathogens, nematodes and toxicants such as heavy metals in the rooting zone (Paul and Clark, 1996). The enzymatic capabilities of the soil microfauna (e.g., protozoa, nematodes) and mesofauna (e.g., mites, wingless insects) are more restricted than those of the prokaryotes and microfungi, so that most of the chemical transformations involved in organic matter decomposition and nutrient cycling are mediated by soil microbes. The contributions of the soil fauna to these processes include more indirect effects such as physical breakdown of litter, dispersal of microbial propagules (including mycorrhizae and pathogens), selective grazing on microbial species and, in the case of macrofauna, modification of soil structure. For example, earthworms, where present, can exert a tremendous impact on soil aeration, aggregate structure, water-holding capacity and bulk density of soils, as well as influence nitrogen mineralization rates (Lee, 1985).
Figure 24 Salt tolerance and crop yield relative to soil salinity (electrical conductivity, EC) and the estimated percentage of roadsides with corresponding levels of electrical conductivity (from Bresler et al., 1982)

3.4.2.2 Effects on soil biota
Data on effects of sodium chloride on soil organisms are presented by Butler and Addison (2000). Soil bacteria can be quite sensitive to sodium chloride. Hattori & Hattori (1980) studied the growth of 40 oligotrophic soil bacteria isolated from various Japanese soils. Two of the 40 bacteria were moderately inhibited by 0.15 g sodium chloride/L (corresponding to approximately 60 mg sodium/L and 90 mg chloride/L). One bacterium was strongly inhibited by 1 g sodium chloride/L (approximately 400 mg sodium/L and 600 mg chloride/L), while 17 were strongly inhibited by 5 g sodium chloride/L (approximately 2000 mg sodium/L and 3000 mg chloride/L). Eleven of the 40 bacteria were strongly inhibited by 10 g sodium chloride/L, while the remaining 9 were moderately inhibited at 10 g/L. McCormick and Wolf (1980) found that soil nitrification was significantly reduced when 0.25 g/kg (approximately 100 mg sodium/kg and 150 mg chloride/kg) or more sodium chloride was present. Sodium chloride at -10 g/kg completely inhibited nitrification. Some rhizobia, soil bacteria that form nitrogen-fixing nodules with leguminous plants (including important crops like soybean, pea and clover), tend to be salt-sensitive. The generation times of salt-sensitive Bradyrhizobia japonicum strains were increased with exposure to 2.6 g sodium chloride/L, while there was little effect on salt-insensitive variants (Upchurch and Elkan, 1977). Earthworms can also be quite sensitive to salt. Earthworms of the species Eisenia fetida died within 2 weeks when exposed to≥5 g sodium chloride/kg (Kaplan et al., 1980). McCormick and Wolf (1980) reported a fairly sharp decline in carbon dioxide evolution from a sandy loam treated for 14 weeks with sodium chloride when sodium chloride exceeded 10 g/kg (corresponding to about 4000 mg sodium/kg and 6000 mg chloride/kg). Soil receiving 100 g sodium chloride/kg did not release detectable carbon dioxide.
3.4.2.3 Exposure of soil organisms to road salt
Soil concentrations of sodium and chloride are presented in Cain et al. (2001). The highest soil chloride concentration reported was 1050 mg/kg in a sample taken in the highway median, and the highest soil sodium concentration was 890 mg/kg in a sample taken 10 m from the highway, both from a study along a four-lane highway in Ontario (Hofstra and Smith, 1984). Concentrations of sodium and chloride decreased rapidly with distance from the road. Concentrations of sodium were below 100 mg/kg at distances greater than about 20 m from the road at all reported Canadian sites, while concentrations of chloride were below 300 mg/kg at distances greater than about 30 m from the road.
Sodium concentrations of up to 13 100 mg/kg and chloride concentrations up to 14 500 mg/kg have been reported in soils at patrol yards (Section 2.4.4.2).
3.4.2.4 Environmental assessment
Sensitive soil bacteria are moderately inhibited by sodium chloride at concentrations as low as 150 mg sodium chloride/L (corresponding to approximately 60 mg sodium/L and 90 mg chloride/L). Soil nitrification can be significantly reduced at a sodium chloride concentration of 250 mg/kg (approximately 100 mg sodium/kg and 150 mg chloride/kg). Soil concentrations of sodium exceeding 60 mg/kg have been reported within about 30 m from the edges of roads in Canada. Soil concentrations of chloride exceeding 200 mg/kg have been reported within about 200 m from the edges of roads in Canada.
Concentrations high enough to adversely affect microorganisms and other soil organisms have also been reported for soils at patrol yards.
3.4.2.5 Conclusions
Concentrations of sodium and chloride in soil near the edges of roads in Canada and in patrol yards are high enough to have adverse effects on sensitive soil organisms. How serious these effects might be to soil ecosystem function is very difficult to gauge. Soil microorganisms may have considerable ability to adapt to increased salinity or to reestablish from unaffected areas. The dispersal capabilities of soil fauna such as earthworms are low, as most species are adapted to the relatively high humidity of the soil environment. Where affected areas are small enough - for example, strips of soil adjacent to roads - populations of impacted soil organisms may be able to be reestablished quite quickly if the unfavourable salt conditions are abated.
3.5 Terrestrial vegetation
Information presented in this risk assessment section is taken from Cain et al. (2001).
Elevated levels of sodium and chloride in the substrate or soil affect plants in various ways:
- inhibition of water and nutrient absorption by plants due to osmotic imbalances, resulting in reduced shoot and root growth and drought-like symptoms;
- nutritional imbalances for some species by disrupting uptake of other nutrients;
- long-term growth inhibition and, at higher levels, direct toxicity to the plant cells, manifested in leaf burn symptoms and tissue death; and
- deterioration of soil structure.
Excess soil salinity leads to deterioration of soil structure due to soil crusting and clogging of soil pores by entrapment of dispersed soil clay and silt particles (Morin et al., 2000). Soil crusting reduces shoot emergence of subsurface seeds and root penetration of both surface and subsurface seeds, resulting in reduced plant establishment. Clogging of soil pores reduces a) soil space available for air and water retention and b) air and water penetration and permeation. Reduced soil aeration is a concern, since reduced oxygen supply to plant roots affects root growth.
An additional impact of elevated chloride in roadside soils is to increase the availability of heavy metals. Chloride forms complexes with heavy metals, rendering many of them more water soluble and therefore more available for uptake by plant roots. This would increase plant uptake of such metals, possibly resulting in plant toxicity, depending on the type and amount of metals present in the soil. Smolders and McLaughlin (1996), for example, reported on chloride-related enhancement of cadmium mobilization through the soil and increased plant uptake.
Laboratory and experimental field studies (done away from roadside locations) have demonstrated that soil and spray applications of sodium and calcium chloride severely injure plants and have characterized the injury symptoms (Hall et al., 1972; Cordukes and Maclean, 1973; Dirr, 1975, 1978; Hofstra and Lumis, 1975; Paul et al., 1984; Headley and Bassuk, 1991; Dkhili and Anderson, 1992; Harrington and Meikle, 1994).
Plant symptoms following exposure to elevated soil levels of sodium and chloride include general plant decline, reduction in leaf size and plant growth, leaf chlorosis, leaf burn and tissue death. Seed germination can be reduced or delayed as well.
Injury of woody plants along roadsides due to salt spray is the result of tissue drought or desiccation and is related to the penetration of phytotoxic ions of sodium and chloride through the stem, bud and leaf tissues (Barrick and Davidson, 1980; Chong and Lumis, 1990). Death of stems and death of leaf and flower buds, usually on first-year shoots, are the predominant symptoms on deciduous woody plants. Flower buds are more sensitive to salt injury than leaf buds. Conifers exhibit premature leaf drop and browning of needles.
Salt levels in soil or plant tissue are generally highest in the spring and decline with rainfall (leaching of soil) or plant growth. Some species may be able to recover or regrow following injury early in the growing season. However, many herbaceous plant species germinate or regrow from dormant propagules in the spring due to specific day length, temperature and moisture requirements. Once a bank of seeds or propagules on a site is killed by elevated salt levels, the affected species may not regrow, resulting in a change in the plant community.
With woody species, even though plant injury is often worst in the spring and some plants recover later in the growing season, repeated injury, year after year, reduces the vigour and growth of the plants. If the soil or tissue levels are high enough, a plant may be killed outright or may have extensive dieback, which results in plant weakening and death over a number of years (Sucoff, 1975; Lumis et al., 1976). In addition, woody plants that are weakened by the repeated stress of road salt are more sensitive to disease, insects or abiotic stresses, such as winter injury (Sucoff and Hong, 1976) or drought.
For woody species that flower in the spring, flower buds lost due to salt damage will not be recovered later in the season. If this damage occurs year after year, there will be a negative impact on the reproductive capability of spring flowering species.
Dirr (1978) pointed out that the pattern of plant injury and elevated soil or tissue levels should be used to confirm that the cause of the damage is de-icing salts. The following injury patterns are associated with road salt injury to plants (Lumis et al., 1973; Dirr, 1976):
- injury occurs in a linear pattern along roads or in areas where runoff from roads collects;
- injury is more severe on the side of the plant facing the road;
- injury decreases with distance from the road;
- injury is worse on the downwind side of the road;
- parts of woody plants that are covered by snow or were sheltered from spray lack injury symptoms;
- parts of trees that are above the salt spray zone are not injured or are injured less;
- salt spray injury extends only a short distance into dense plants;
- injury in coniferous trees becomes evident in late winter and continues into the growing season; and
- injury in deciduous trees becomes evident in spring when growth resumes and continues into the growing season.
Additional information about injury to plants characteristic of road salt exposure is presented in Cain et al. (2001).
There have been extensive compilations of ratings of sensitivity of woody plants from various observational and experimental studies. A survey of these woody plant ratings (ornamental and forest species) in the literature indicated that 29.6-50.8% of the plants listed in each report were deemed sensitive to salt in at least one rating (Sucoff, 1975; Dirr, 1976; Lumis et al., 1977; Thuet, 1977; Beckerson et al., 1980; Dobson, 1991).
Lumis et al. (1977), making field observations of plants on Ontario roadsides, rated 30% of 73 species as sensitive to road salt; 7% had severe injury symptoms that could result in plant death. A review of sensitivity ratings of plants commonly used in Canadian landscape plantings (Beckerson et al., 1980) found that 40% of 89 species had been rated as sensitive to road salt.
A survey of the salt tolerance ratings of 32 cultivars or varieties of fruit trees, vines or shrubs, representing commercial crop and landscape plants (Appendix 2 in Cain et al., 2001), indicated that 65% were considered sensitive to salt in at least one rating.
There are 15 principal genera of trees (over 25 species) within Canadian forested regions, according to Rowe (1972), and 11 of these have been rated as sensitive to road salt (Section 4.5 in Cain et al., 2001). Looking at individual species listed by Rowe (1972), 8 of 15 (53%) have been rated as sensitive to road salt in at least one rating.
A list of roadside trees and shrubs rated for their resistance to airborne highway salt spray, based on Lumis et al. (1983), is presented in Table 19. A list of native forest trees rated for their resistance to airborne highway salt spray is presented in Table 20.
Additional information about the sensitivity of various types of plants is included in Cain et al. (2001).
3.5.1 Conservative environmental assessment
EC25 and CTV threshold values were estimated from the scientific literature for chloride, sodium and sodium chloride in growing media (soil, soil water or water); for sodium chloride in foliar spray solution; and for chloride and sodium in plant tissue (Cain et al., 2001). Lowest-Observed-Effect Level (LOEL) threshold values were identified for chloride and sodium chloride in growing media, and No-Observed-Effect Level (NOEL) threshold values were identified for sodium chloride in growing media.
1 A rating of 1 indicates no twig dieback or needle browning of conifers and no dieback, tufting or inhibitions of flowering of deciduous trees and shrubs. Ratings of 5 represent complete branch dieback and needle browning of conifers, and complete dieback, evidence of previous tufting and lack of flowering of deciduous trees and shrubs. Under severe conditions, plants rated 5 will eventually die. Ratings of 2, 3 and 4 encompass slight, moderate and extensive gradations of the above injury symptoms.
1S - sensitive, MS - moderately sensitive, M - intermediate, MT - moderately tolerant, T - tolerant.
3.5.1.1 Effects threshold values
Threshold values, derived from experimental data or roadside studies, have been tabulated in Tables 21, 22 and 23 and in Section 3.6 in Cain et al. (2001). The plant species represented include wetland species (e.g., rushes, sphagnum moss, bulrush, cattails and duckweed), herbaceous species (e.g., sedges, grasses and beets), coniferous species (e.g., pine and spruce) and deciduous species (e.g., maple, peach and plum). The threshold values ranged from 67.5 to 300 ppm sodium, 215 to 1500 ppm chloride and 280 to 66 600 ppm sodium chloride in growing media (applied soil solution) (Table 21); from <280 to 17 800 ppm sodium and <4100 ppm chloride in herbaceous tissue following soil or water solution exposure (Table 22); and from 200 to 16 100 ppm sodium and 800 to 70 700 ppm chloride in woody tissue following aerial exposure and 7140 ppm sodium chloride foliar spray concentration (Table 23).
For the conservative assessment, the values used were the lowest concentrations that were identified in the threshold value analysis (Section 3.6 in Cain et al., 2001) for each type of threshold described above from experimental evaluations or from sampling done beyond the road or highway right-of-way limits.
3.5.1.2 Estimated Exposure Values (EEVs)
The EEVs used in the conservative assessment were the highest values extracted from the literature dealing with environmental concentrations in Canada of chloride or sodium in growing media (soil or aqueous) in roadside conditions from sampling done beyond the highway right-of-way or the highest values extracted from literature dealing with concentrations of chloride or sodium found in vegetation growing along salted roadsides in Canada beyond the highway right-of-way.
The exposure values used for the assessment were 240 ppm soil chloride and 45 ppm soil sodium in samples taken outside of the highway right-of-way, 40 m from the highway, both from a study along a four-lane highway in Ontario (Hofstra and Smith, 1984).
The highest soil chloride concentration reported along a highway was 1050 ppm in a sample taken in the highway median, and the highest soil sodium concentration was 890 ppm in a sample taken 10 m from the highway, both from the same study.
No Canadian references reported soil concentrations of sodium chloride. A value of soil sodium chloride was estimated based on the concentration of chloride found in the soil 40 m from the highway, 240 ppm, which was multiplied by a factor of 1.6485, resulting in a value of 396 ppm sodium chloride. A second value of soil sodium chloride was estimated based on the concentration of sodium found in the soil 40 m from the highway, 45 ppm, which was multiplied by a factor of 2.542, resulting in a value of 114 ppm sodium chloride. The average of these two values, 255 ppm sodium chloride, was used in the conservative estimation.
In order to estimate the EEVs for aerial exposure of plants to sodium chloride following road de-icing, references were reviewed that reported concentrations of chloride or sodium in plant tissues at given distances from the road or highway, following exposure to road salt aerial dispersion (refer to Section 2.3 of Cain et al., 2001). Three references were reviewed for tissue concentrations of chloride and sodium. These data included plant tissue samples taken from 10 to 200 m from the road or highway. These references reported studies from Ontario on Prunus (peach and plum) and unspecified vegetation.
The tissue concentrations used for the assessment were 4600 ppm chloride and 3500 ppm sodium reported in peach (Prunus persica) growing 40 m from a four-lane highway, outside the highway right-of-way, in southern Ontario (Northover, 1987). By comparison, the highest tissue chloride concentration was 11 000 ppm, reported in unspecified vegetation growing 60 m from a four-lane highway in Ontario (Hofstra and Smith, 1984). Northover (1987) also reported 9000 ppm chloride in peach growing 20 m from a four-lane highway in southern Ontario (Northover, 1987). The highest tissue sodium concentration was 6900 ppm, reported in twig tissue of peach growing 20 m from a four-lane highway in southern Ontario (Northover, 1987).
3.5.1.3 Quotient estimation
From Section 3.5.1.2, it can be seen that chloride and sodium have been reported in Canadian soils and in plant tissues outside of the highway right-of-way at concentrations higher than or, in one case, in the range of the effects threshold values presented in Section 3.5.1.1 and in Section 3.6 of Cain et al. (2001). Therefore, a detailed assessment of the effects of road salts on terrestrial vegetation is warranted.
3.5.2 Detailed environmental assessment -threshold-based analysis
This risk characterization reviews concentrations of chloride and sodium in soils or tissue that produce biologically relevant effects on vegetation and relates these to concentrations found in the Canadian environment. The data presented represent primarily Canadian literature and incorporate threshold values that were presented in Section 3.5.1.1 above and in Section 3.6 of Cain et al. (2001).
3.5.2.1 Response to sodium in soil
The range of threshold values for sodium chloride applied to roots was 67.5-300 ppm sodium (Table 21 above and Section 3.6 of Cain et al., 2001). The most sensitive plants reported were 2-year-old ponderosa pine (Pinus ponderosa) seedlings, with a threshold value of 67.5 ppm, based on foliar injury following soil application of a sodium chloride solution. Mortality occurred at a concentration of 140 ppm sodium (Bedunah and Trlica, 1979). Seed germination rates of prairie grass and broad-leaved wildflower species were reduced by about 60-70% and root growth was reduced by up to 73% when plants were exposed to 100-400 ppm sodium chloride (about 40-160 ppm sodium) (Harrington and Meikle, 1994). The EC25 threshold values that were estimated from this study for sodium concentration in soil were 202-270 ppm.
The concentration of sodium in soil exceeded the estimated threshold range of 67.5-300 ppm in a number of soil samples collected in Canada at a distance of about 30 m or less from the roads (Figure 2.17 in Cain et al., 2001). A concentration of 890 ppm sodium was found in soil 10 m from a four-lane highway in southern Ontario (Hofstra and Smith, 1984). A concentration of 45 ppm sodium was found 40 m from the highway, outside the right-of-way, in the same study.
3.5.2.2 Response to chloride in soil
The range of threshold values for sodium chloride applied to roots was 215-1500 ppm chloride (Table 21 above and Section 3.6 of Cain et al., 2001). The most sensitive plants reported were 2-year-old ponderosa pine seedlings, with a threshold value of 215 ppm chloride, based on foliar injury following soil application of a sodium chloride solution. Mortality occurred at a concentration of 350 ppm chloride (Bedunah and Trlica, 1979).
The estimated LOEL for reduction in mean length growth of sphagnum moss, Sphagnum recurvum, was 300 ppm chloride in a hydroponic solution (Wilcox, 1984). Wilcox and Andrus (1987) reported reproductive effects, reduced gametophore production, in Sphagnum fimbriatum at 300 ppm chloride.
The concentration of chloride in soil samples collected in Canada at a distance of up to 200 m from the roads exceeded the estimated lowest threshold of 215-500 ppm for a number of samples (Figure 2.16 in Cain et al., 2001). Concentrations of chloride in soils exceeded 500 ppm only at the edge of the roads. A soil concentration of 240 ppm chloride was found in samples taken 40 m from a four-lane highway, beyond the right-of-way, in southern Ontario (Hofstra and Smith, 1984). A soil concentration of 1200 ppm chloride was found in the soil behind a patrol yard located along a highway in northwestern Ontario (Racette and Griffin, 1989).
3.5.2.3 Response to sodium chloride in soil
The range of threshold values for sodium chloride applied to roots was 280-66 600 ppm sodium chloride (Table 21 above and Section 3.6 of Cain et al., 2001). The most sensitive reported species were two wetland species, three-fruited sedge (Carex trisperma) and common mountain holly (Nemopanthus mucronata [mucronatus]), which had salt tolerance limits of 280 ppm based on sodium chloride concentration gradients in bog water (Wilcox, 1986a). Survival of Colorado blue spruce (Picea pungens) and growth of Scots pine (Pinus sylvestris) were affected by sodium chloride concentrations of 600 ppm (Werkhoven et al., 1966).
Schauffler (1993) reported sublethal effects, biomass increase and tiller production of cottongrass (Eriophorum vaginatum var. spissum) with a concentration of 400 ppm sodium chloride in a hydroponic culture, and reproductive effects, reduced flower production, with a concentration of 1600 ppm.
No Canadian references reported soil concentrations of sodium chloride; however, values have been estimated using reported concentrations of sodium and chloride. A value of soil sodium chloride of 1978 ppm was estimated by multiplying the highest Canadian concentration of chloride found in the soil adjacent to a patrol yard, 1200 ppm, by a factor of 1.6485. A second value of soil sodium chloride of 2262 ppm was estimated by multiplying the highest concentration of sodium found in the soil 10 m from the highway, 890 ppm, by a factor of 2.542. The average of these two values, 2120, was about 10 times the lowest threshold value of 280 ppm.
Using the same method of calculation, a value of 255 ppm sodium chloride outside of the highway right-of-way was calculated from soil values of 240 ppm chloride and 40 ppm sodium, found 40 m from a four-lane highway in southern Ontario (Hofstra and Smith, 1984). This value is in the range of the threshold value of 280 ppm.
3.5.2.4 Response to sodium and chloride by aerial dispersion
The range of threshold values for aerial exposure to sodium chloride was 800-70 700 ppm chloride and 200-16 100 ppm sodium (Table 23 above and Section 3.6 of Cain et al., 2001). The most sensitive species reported in a Canadian environment were peach (Prunus persica) and plum (Prunus domestica), with a threshold value of 800 ppm chloride and 650 ppm sodium, based on twig dieback (a sublethal effect) following aerial exposure (McLaughlin and Pearson, 1981). Mortality of multiple pine species (Pinus spp.) had threshold values of 13 000 ppm chloride and 8000 ppm sodium (Townsend and Kwolek, 1987). Reproductive impacts, evaluated as peach fruit yield, had threshold values of 1000 ppm chloride and 1700 ppm sodium (Northover, 1987).
Tissue concentrations exceeding the lowest threshold values of 800 ppm chloride and 650 ppm sodium estimated for sensitive plant species (Section 3.6.2 of Cain et al., 2001) have been reported at distances up to about 120 m from highways for sodium and up to 200 m for chloride. Tissue concentrations of 4600 ppm chloride and 3500 ppm sodium were reported in peach growing 40 m from a four-lane highway, outside the highway right-of-way, in southern Ontario (Northover, 1987). The highest concentrations reported in woody plants growing along a road were 9000 ppm chloride and 6900 ppm sodium in peach twig tissue growing 20 m from a four-lane highway in southern Ontario (Northover, 1987).
3.5.2.5 Detailed environmental assessment -summary of threshold-based analysis
Elevated soil levels of sodium and chloride have been recorded up to 30 m from four-lane highways (Hofstra and Smith, 1984; Davis et al., 1992) and up to 15 m from two-lane highways (Davis et al., 1992; Soilcon Laboratories Ltd., 1995). The threshold range of 67.5-300 ppm sodium and 215-500 ppm chloride in soil would be exceeded in a zone up to about 30 m from highways (Figures 2.7 and 2.8 in Cain et al., 2001), depending on the class of road, application rates, local topography and climate. Threshold values have also been exceeded on sites where salty runoff has collected (Racette and Griffin, 1989).
Similarly, elevated tissue concentrations of sodium and chloride have been reported for vegetation growing adjacent to highways in southern Ontario. Tissue concentrations exceeding the lowest threshold values of 650 ppm sodium and 800 ppm chloride estimated for sensitive plant species (Section 3.6.2 of Cain et al., 2001) have been reported at distances up to about 120 m from highways for sodium and up to 200 m for chloride.
3.5.3 Detailed environmental assessment -reference site analysis
Reference sites are presented that document effects on vegetation that can be traced to application of road salts. A number of Canadian studies have evaluated environmental concentrations of sodium and chloride and plant impacts as a result of de-icing salt application to roads or runoff as a result of handling activities in highway maintenance patrol yards. Some U.S. studies that are relevant to Canada have also been included in this section. Details about these studies can be found in Section 4.4 of Cain et al. (2001).
3.5.3.1 Spread of halophytic plant species
The spread of halophytic species, characteristic of sea coasts and salt marshes, has been recorded along roadsides in Ontario. Catling and McKay (1980) reported the occurrence of 20 salt-tolerant plant species along roadsides in Ontario, of which 4 species were reported for the first time in the province. The range of an additional four species was extended significantly. The species discussed by Catling and McKay (1980) were naturally occurring plants, not ones planted by the road authorities for roadside stabilization or landscaping. These species were spreading naturally along highways in response to the use of de-icing salts and the decline of non-adapted planted vegetation. The sites studied had sodium concentrations in the range of 1000 ppm and growing conditions that ranged from aquatic to moist to periodically moist to dry. These sites included roadside ditches, road and highway medians, areas adjacent to highways that receive salty runoff, and areas adjacent to salt storage depots and snow dumps. These included areas within the engineered right-of-way, within the planted right-of-way and outside the right-of-way.
Species that were tolerant of soil sodium levels of 500-1000 ppm and were often associated with halophytic species included narrow-leaved cattail (Typha angustifolia), common reed-grass (Phragmites australis) and weedy species such as annual and perennial sow-thistle (Sonchus oleraceus, Sonchus arvensis, Sonchus uliginosus), common ragweed (Ambrosia artemisiifolia), wild carrot (Daucus carota) and kochia (Kochia scoparia).
Reznicek et al. (1976) reported the increase in distribution of Carex praegracilis, an alkali-tolerant western sedge species, since its first observation in Ontario in 1973. The plant was observed in 28 highway locations in 14 counties, generally within highway rights-of-way. Additional reports of roadside and highway median occurrences of this species were reported by Catling and McKay (1980).
Similar observations of the spread of salt-tolerant species have been made in Michigan, which has a climate and flora similar to those of southern Ontario (Reznicek, 1980).
3.5.3.2 Community changes in a bog impacted by road salts
The effects of sodium and chloride contamination were studied in Pinhook Bog, located in northwestern Indiana, southeast of Michigan City, which had been impacted by road salt runoff (Wilcox, 1982, 1986a,b). This study provides evidence of plant community impacts by elevated salt levels. Many of the predominant plant species in this bog are native wetland species that are found in Canada. This part of Indiana is part of the Great Lakes basin and has the same climatic zone as parts of southern Ontario, the Montréal region, the Atlantic provinces and British Columbia.
An uncovered road salt storage pile had been located on an embankment adjacent to the bog from 1963 to 1972. A protective dome was placed over the salt pile in 1972 to limit runoff, and salt storage was discontinued on this site in mid 1981. The bog also received runoff from a 1-km stretch of a four-lane highway. The annual road salt application for the adjacent highway was 11 300 kg/km (5650 kg per two-lane-kilometre).
The most severely impacted area had mean salt concentrations as high as 486 ppm sodium and 1215 ppm chloride in the interstitial waters of the peat mat. The pattern of elevated sodium and chloride concentrations in the bog was correlated with zones of affected vegetation. The salt concentrations in the root zone decreased each year from 1979 to 1981.
Vegetation evaluations were carried out on the unimpacted and impacted portions of the bog from 1980 to 1983. The salt contamination had resulted in a shift in vegetation species in the impacted areas. Nearly all of the endemic plant species were absent from the portion of the bog with elevated salt levels. They had been replaced with non-bog species dominated by narrow-leaved cattail.
As salt levels declined by 50% over the 4-year study period, many endemic species returned to the impacted bog. Some of the species that had invaded during the high salt conditions declined, while others, such as narrow-leaved cattail and purple chokeberry (Pyrus floribunda), remained dominant in parts of the impacted bog. This study provides evidence that wetland communities can recover once a source of salt is removed, although certain invasive species may remain, resulting in a permanent change in community composition.
3.5.3.3 Effects of salt spray from a four-lane highway on peach and plum orchards in Grimsby, Ontario
The effect of distance on the impact of salt spray was studied in a number of peach and plum plantings along the Queen Elizabeth Way, a four-lane, controlled-access highway in the Grimsby and Beamsville area of Ontario (McLaughlin and Pearson, 1981). These plantings were on grower properties outside of the highway right-of-way. Peach and plum trees were evaluated for terminal twig dieback in April 1980 and 1981. Twig samples were evaluated for chloride and sodium content as well.
The chloride and sodium contents of the twigs were highest closest to the highway and generally decreased with increasing distance from the highway (Figures 2.11 and 2.12 in Cain et al., 2001). Twig tissue sodium concentration was 2030 ppm at 45 m from the highway, and chloride concentration was 2300 ppm at 40 m from the highway.
Twig dieback was greatest in the plants closest to the highway and decreased with increasing distance from the highway (Figure 4.5 in Cain et al., 2001); however, the degree of dieback differed between years and species. Injury was greater on the south side of the highway in April 1981; peach twig dieback was 288 mm at 22 m from the highway, decreasing to 97 mm at 82 m from the highway. In April 1980, peach twig dieback on the south side of the highway fell from 91 mm at 36 m from the highway to 8 mm at 82 m from the highway. Twig dieback was correlated with tissue concentrations of sodium and chloride. Twig dieback increased with increasing concentrations of sodium and chloride in the combined peach and plum data (threshold evaluation, Appendix 8 in Cain et al., 2001).
3.5.3.4 Effects of salt spray from a four-lane highway on peach and plum orchards near St. Catharines, Ontario
The effect of salt spray was evaluated on Loring peach trees during the 1973-74 winter (Northover, 1987). The trees were located on the south side of an east-west section of the Queen Elizabeth Way, a busy, four-lane, controlled-access highway near St. Catharines, Ontario. The orchard was located at the same elevation as the highway, on a grower property outside of the highway right-of-way.
Sodium and chloride tissue content of shoot tissue decreased with increasing distance from the highway. The chloride content of the peach twig tissue, from trees located 20 m from the highway, was greater than 4 times the levels found in trees 120 m from the highway (9000 ppm compared with 1900 ppm chloride). The sodium content was elevated more than 7 times the levels detected 120 m from the highway (6900 ppm compared with 900 ppm sodium).
Fruit yield was reduced at distances less than 80 m from the highway, and peach fruit number per shoot increased with increasing distance from the highway. Shoots in the tree canopy were 97% dead 20 m from the highway. Shoot death decreased with increasing distance from the highway, falling to 8% at 80 m from the highway and decreasing to a background level of less than 1% at 120 m from the highway. The sodium and chloride contents of the shoots were positively correlated with the percentage of dead wood in the canopy of the trees.
The soil levels of sodium and chloride were not elevated on this site, indicating that the elevated tissue levels and plant effects were primarily due to aerial salt spray from the highway.
3.5.3.5 Effects of salt spray from a two-lane highway on blueberries in western Nova Scotia
Eaton et al. (no date) evaluated the effects of road salt from a two-lane highway in the Folly Mountain area, west of Truro, over the winters from 1993 to 1995 on naturally occurring managed lowbush blueberry stands on adjacent lands. Salt application in this area was 33 and 40 tonnes of sodium chloride per kilometre of highway during the winters of 1993-94 and 1994-95, respectively.
Blueberry blossom number and yield were reduced within 35 m of the two-lane highway and approached background levels beyond 50 m from the highway. Blossom number was reduced by 60-90% within 10 m of the highway, and yield was reduced by 50% or more compared with samples taken at 50 m from the highway. Yield under protective structures of nursery film was 4224 kg/ha of fresh fruit in 1994 compared with less than 2000 kg/ha at 30 m and less than 200 kg/ha at 5 m from the highway. In 1995, yield under the protective structures was 8092 kg/ha compared with less than 3000 kg/ha at 35 m and less than 1500 kg/ha at 5 m from the highway.
Stem tissue salt levels taken under the protective structures were 0.53 and 0.3 g sodium chloride per 20 stems in 1993-94 and 1994-95, respectively. This compared with levels from 6 to over 10 g at 10 m from the road in spring 1994 and 3 to over 8 g in spring 1995. Tissue levels were elevated up to 45 or 50 m from the highway. Reductions in both blossom number and yield were negatively correlated with elevated salt content of the blueberry stems taken in winter and early spring.
3.5.3.6 Concentrations in soil and woody plants adjacent to highways in interior British Columbia
Davis et al. (1992) reported on the causes of observed roadside tree injury and decline on 17 forested sites in south-central and eastern British Columbia, located on two-, three- or four-lane highways.
At 15 of the sites, sodium chloride was used for winter maintenance; application rates varied between 60 and 130 kg per lane-kilometre as conditions dictated. On two of the sites that had gravel roads, sodium chloride was not used for de-icing, although sand with 3-5% sodium chloride was applied in the winter. In the summer, calcium chloride or magnesium chloride was used for dust suppression, initially at 2 L/m2, but at 1.6 L/m2 at the time of the study.
A detailed field evaluation was undertaken on the 17 sites. Three sample plots were established, including two injury plots with moderate to severe injury symptoms and one control plot with no visible injury symptoms. A detailed assessment was made of plant salt injury symptoms and injury due to other causes, such as abiotic stress, disease, insects or breakage. Soil and vegetation samples were analysed for sodium and chloride content and other parameters, and a detailed history was completed for each site.
The pattern of injury to conifer trees on 14 of the sites was similar. At these sites, injury decreased with increasing distance from the highway and was typical of road salt injury in woody plants, including leaf scorch, leaf loss, thinning of crowns and severe defoliation and dieback in severely injured trees. In most of the sites, the injury occurred on a narrow strip, within 5-15 m downslope of the highway. At other sites, injury occurred as close as 2 m from the road, since the forest stand was close to the road. At one site, injury was evident for 40 m into a stand that received road runoff from a culvert. At another site, minor injury was evident 50 m from the highway and was attributed to salt spray from the road. Injury occurred within the highway right-of-way of many of the sites, since the typical vegetated right-of-way in British Columbia is 4.5-5 m along a two-lane highway or up to 10 m on some highways. At other sites, injury was outside the highway right-of-way, more than 5 or 10 m from the highway.
Soil chloride concentrations at 0- to 10-cm depth in the control plots ranged from <1 to 12 ppm. Soil concentrations of chloride on the plots with plant injury ranged from 0.9 to 230 ppm chloride and were greater than 25 ppm on six of the sites. Two of the sites that were adjacent to a four-lane highway had soil chloride levels ranging from 3.0 to 198 ppm.
Soil sodium concentrations at 0- to 10-cm depth in the control plots ranged from <1 to 8.5 ppm. Soil concentrations of sodium ranged from 0.02 to 50.2 ppm on the plots with plant injury. At 13 sites, soil sodium concentrations were greater than the control on one or both of the injury plots and were greater than 17 ppm on 7 of the sites. Two of the sites that were adjacent to a four-lane highway had soil sodium levels as high as 43 ppm.
The chloride content of the needles from the injury plots was elevated up to 3.4 times that found in the control needle samples. The sodium content of needles from the injury plots was elevated up to 206 times that found in the control needles.
Sodium tissue levels were not elevated on the two sites where sodium chloride was not used for winter de-icing, but tissue chloride levels were 2-2.4 times greater than in the control plots on at least one of the injury plots. Plant injury occurred less than 10 m from the roadside on these sites, both inside and outside the highway right-of-way. On one site, soil chloride was elevated to 225 ppm, while soil sodium was not elevated. The use of calcium chloride or magnesium chloride for dust suppression as part of summer maintenance programs was implicated in the injury of trees on these two sites.
In this study, sodium chloride used for de-icing and calcium chloride or magnesium chloride used for summer dust control were strongly implicated as major contributors to the observed roadside tree injury and decline on 16 of the sites. This conclusion is based on plant injury symptoms, the pattern of injury and elevated sodium and chloride concentrations in soil and vegetation.
3.5.3.7 Concentrations in soil and woody foliage adjacent to a two-lane highway in British Columbia
Soilcon Laboratories Ltd. (1995) investigated the levels of salt in soil and plant tissue with respect to plant injury levels along a 2-km section of Loon Lake Road, a two-lane highway northwest of Kamloops in British Columbia. The dominant forest species on the site were ponderosa pine and Douglas-fir (Pseudotsuga menziesii).
The individual rock salt application rates were 22, 88 or 153 kg per two-lane-kilometre, depending on whether low, average or heavy applications were applied. The total salt loading over the winter was 1561 kg per two-lane-kilometre, consisting of 1031 kg applied as rock salt and 530 kg applied as sand with 5% salt. Salt or sand applications were made from November 15, 1993, to February 23, 1994.
Nine sites were evaluated within a 2-km section along Loon Lake Road, where trees displayed low, moderate or severe salt injury symptoms. Trees that fell in the moderate and severe injury classes were located from 3 to 8.4 m from the road, both within and outside the highway right-of-way. In comparison, trees that fell in the low injury class were located 10-20 m from the road outside of the highway right-of-way.
Soil concentrations of chloride and sodium were sampled on the nine sites in December 1993 and April 1994 and compared with control values sampled in April 1991. On sites with moderate and severe tree injury, soil concentrations of chloride measured in the 0- to 10-cm layer were 31.3 ppm chloride (range 84.5-4.1 ppm) in December. By April, the concentrations fell to 5.12 ppm chloride (range 12.6-1.6 ppm). On sites with low tree injury, soil concentrations of chloride measured in the 0- to 10-cm layer were 7.2 ppm chloride (range 23.5-1.4 ppm) in December. By April, the concentrations fell to 2.9 ppm chloride (range 4.5-1.3 ppm). This compared with background levels of 3.8 ppm chloride (range 7.5-0.2 ppm).
On sites with moderate and severe tree injury, soil concentrations of sodium measured in the 0- to 10-cm layer were 15.9 ppm (range 28.2-1.9 ppm) in December. By April, the concentrations fell to 9.1 ppm sodium (range 30.7-1.0 ppm). On sites with low tree injury, soil concentrations of sodium measured in the 0- to 10-cm layer were 4.7 ppm (range 11.7-5.6 ppm) in December. By April, the concentrations fell to 1.2 ppm sodium (range 1.8-0.5 ppm). This compared with background levels of 2.3 ppm sodium (range 2.7-2.0 ppm).
Soil concentrations of chloride and sodium were higher at sites with plants in the moderate- and severe-injury class than at sites with plants in the low-injury class in both December and April. Soil concentrations were approaching background levels by the April sampling period. It was likely that sodium and chloride had leached from the soil by April by rainfall, since the last salt application was in February.
Over the December to April sampling period, foliar chloride concentrations ranged from 110 to 560 ppm in trees in the low-injury class, from 110 to 3180 ppm in trees in the moderate-injury class and from 870 to 5590 ppm in trees in the severe-injury class.
During the same period, foliar sodium concentrations ranged from 0.5 to 70 ppm in trees in the low-injury class, from 0.5 to 190 ppm in trees in the moderate-injury class and from 30 to 920 ppm in trees in the severe-injury class.
3.5.3.8 Concentrations in woody foliage directly adjacent to a two-lane highway in Connecticut
Button and Peaslee (1966) reported foliar concentrations of chloride and sodium in mature (about 70-year-old) sugar maple (Acer saccharum) leaves. The trees were growing close (from 1 to 4 m) to Route 17 between Middletown and Durham, a two-lane highway in Connecticut, likely within the highway right-of-way. The research is relevant to Canadian conditions since sugar maple is a native forest species, used for maple syrup production and planted as a landscape tree. This study also provides an example of injury due to soil exposure to road salt via runoff.
Although salt load is not provided in the paper, the authors indicate that 63 000 tonnes of rock salt and 2250 tonnes of calcium chloride were used annually on 7180 km of highway in Connecticut. This converts to an annual application of 9850 kg rock salt/km and 350 kg calcium chloride/km.
The trees growing on the west side of the highway had road surface drainage flowing directly towards the trees. The trees growing on the east side of the highway had drainage diverted by a curb that had been installed during the summer prior to the study. Foliar concentrations of chloride at the different sampling periods averaged 5200-6700 ppm on the west side of the road. These were significantly higher than the concentrations found on the east side of the road, averaging 1600-5400 ppm. This compared with levels ranging from 200 to 1400 ppm chloride in control trees of the same species and age, located away from a road. Foliar sodium levels ranged from an average of 1100-1600 ppm on the west side of the highway compared with non-detectable to 200 ppm on the east side of the highway and 0 ppm in the control trees.
The higher concentrations of chloride and sodium found in the foliage of the trees on the west side of the highway with overland surface flow were correlated with moderate to severe injury symptoms in the trees on the west side of the road. This compared with no or light injury symptoms in the trees on the east side of the road and no injury in the control plants.
3.5.3.9 Concentrations in soil and woody and herbaceous foliage along roadsides in Massachusetts
Soil and vegetation samples were taken at different distances along numerous highways in inland (central and western) Massachusetts during June 2000 (Barker, 2000; Mills and Barker, 2001). The research is relevant to Canadian conditions, since most of the sampled species are native or naturalized species in Canada and since the application rates used in Massachusetts in 1999 were comparable to application rates in central and eastern Canada. Application rates in Massachusetts were 240 lb./lane-mile (136 kg per two-lane-kilometre), compared with rates in Ontario ranging from 130 to 170 kg per two-lane-kilometre.
Samples of eight of the woody plant species and a range of herbaceous species were taken at 1.5-12 m from different highways and were analysed for sodium concentrations. Plant injury was observed within 4-9 m from the highways and was correlated with increased sodium concentrations in the plant tissues in multiple pine species (Pinus spp.) and sumac (Rhus sp.). In pine, the mean foliar concentration of sodium was 2139 ppm in damaged foliage compared with 28 ppm in a sample collected from an off-roadside site. In sumac, the mean foliar concentration of sodium was 209 ppm in damaged foliage compared with 177 ppm in healthy foliage. There was not a great difference found in sodium concentrations in a single sample of poplar (Populus sp.). The sodium concentration was 310 ppm in damaged foliage compared with 338 ppm in healthy foliage.
The foliar sodium concentrations in plants were elevated between 3 and 7.6 m from the road and continued to fall off up to 12 m from the road. Extractable sodium measured in the roadside soils was elevated within 6.1 m from the road, as high as 154 ppm, falling off to 26 ppm at 12 m from the road. Soil pH was elevated within 3 m from the road, above 7, compared with less than 6.5 at greater than 6 m from the road. There was not a consistent pattern to electrical conductivity, which ranged from 0.12 dS/m at 9.1 m to 0.30 dS/m at 12 m from the road.
3.5.3.10 Concentrations in soil and woody plants downslope from a four-lane highway in British Columbia
A site investigation conducted in 1989 in Boitanio Park in British Columbia investigated the sources of tree injury and decline that were evident on trees in the east section of the park near Highway 97 (Van Barneveld and Louie, 1990). Boitanio Park is located in Williams Lake, southwest of a four-lane section of Highway 97. The dominant tree species in the park was rocky mountain Douglas-fir (Pseudotsuga menziesii var. glauca).
The area of concern was near the highway and sloped towards the southwest with a grade of 5-15%. Two culverts were located along the highway above the park, so the water was channelled through the park. The highway had a traffic volume of approximately 12 000 vehicles per day in this area, and sodium chloride de-icing salt was applied to the highway at a rate of 60-130 kg salt per lane-kilometre.
Trees along the drainage channels and adjacent to the highway displayed symptoms of severe injury and rapid decline that were reported in the spring of 1989. Preliminary soil samples found high levels of sodium in the soil, which led to a comprehensive sampling and field analysis of vegetation and soil within the park in July 1989. Samples were taken using an incomplete grid pattern running away from the highway, with sampling locations every 50 m along the grid. Vegetation was evaluated for plant injury symptoms and insect and disease status. Foliar tissue was sampled and analysed for chloride and sodium content. Soil samples were taken from successive soil layers of 25 cm down to 150 cm.
Plant injury was evaluated using a rating scale of low, moderate and severe. Trees in the low-injury class had less than 10% discoloration and dead branches with healthy leaders.
Trees in the moderate-injury class had 10-50% discoloration, defoliation and some dead branches with leaders alive. Trees in the severe-injury class were dead or had greater than 50% foliar discoloration, defoliation and dead tops.
The pattern of tree injury was associated with roadside locations and with the drainage patterns from the two culverts along drainage channels within the park. The injury was most severe along these drainage pathways.
Soil concentrations of sodium and chloride were elevated on sites with severe or moderate injury compared with those with low injury. The average soil concentration of sodium was 108.7 ppm on the severe-injury sites, 37.9 ppm on the moderate-injury sites and 29.0 ppm on the low-injury sites. The average soil concentration of chloride was 52.1 ppm on the severe-injury sites, 50.0 ppm on the moderate-injury sites and 20.3 ppm on the low-injury sites. Severe-injury sites had soil sodium concentrations that were 3.7 times the concentration in low-injury sites and soil chloride concentrations that were 2.6 times the concentration in low-injury sites.
Foliar concentrations of sodium and chloride were higher in the plants with a severe or moderate injury rating compared with the plants with a low injury rating. The average concentration of sodium in the 0- to 3-year needles was 113 ppm on the severe-injury sites, 54.8 ppm on the moderate-injury sites and 59.7 ppm on the low-injury sites. This compared with average concentrations of sodium in the 4-to 6-year needles of 208 ppm on the severe-injury sites, 72.7 ppm on the moderate-injury sites and 47.9 ppm on the low-injury sites.
The average concentration of chloride in the 0- to 3-year needles was 3560 ppm on the severe-injury sites, 2630 ppm on the moderate-injury sites and 572 ppm on the low-injury sites. A similar pattern was observed in the average concentrations of chloride in the 4- to 6-year needles: 4180 ppm on the severe-injury sites, 2667 ppm on the moderate-injury sites and 1060 ppm on the low-injury sites.
Other factors such as recent drought, presence of other contaminants in the runoff, insects and recent brush removal may have aggravated the decline of the trees. However, it was determined that these factors were not the primary cause of the decline in the park. This site has a dry climate, with annual May to August precipitation of 158 mm of rain. This low rainfall amount and recent drought would have aggravated the problem, in that elevated levels of sodium and chloride in the soil would not be leached out.
The injury symptoms on the Douglas-fir in this site were consistent with salt injury and supported by the pattern of more severe plant injury associated with roadside locations and with the drainage patterns from the two culverts along drainage channels within the park. In these areas, elevated soil and tissue concentrations of sodium and chloride were correlated with more severe injury symptoms in the trees.
Injury due to highway runoff following a drainage course into forest stands has been reported by Kliejunas et al. (1989). They recorded conifer needle tip dieback typical of salt absorption from the soil in forest stands 91-122 m from Highway 89 at Emerald Bay and from Interstate 80 at Donner Summit in the Lake Tahoe area of Nevada. High levels of sodium and chloride (5800 ppm sodium and 13 700 ppm chloride) were found in injured white fir (Abies concolor) and Jeffrey pine (Pinus jeffreyi) foliage at Donner Summit located within a drainage channel 76 m downslope from Interstate 80.
3.5.3.11 Concentrations in soil and woody foliage adjacent to a patrol yard
Racette and Griffin (1989) reported on the investigation of the cause of severe woody plant injury in the vicinity of a highway maintenance patrol yard east of Kenora, Ontario. Trees in the area behind the sand dome and salt storage sheds and along a drainage channel that received drainage from the highway and patrol yard area had severe injury symptoms.
In a nearby sand and gravel storage area, salt-treated sand had been stored on at least one occasion. In this area, jack pine (Pinus banksiana), spruce (Picea sp.), balsam fir (Abies balsamea) and poplar (Populus sp.) trees had died a number of years before. Existing vegetation was damaged along a drainage course in this area. Grasses had been killed, and trembling aspen (Populus tremuloides) had symptoms of severe marginal necrosis.
Plant tissue levels of sodium and chloride were sampled in September 1988 in the areas where the injury was evident. Foliar concentrations of chloride ranged from 1800 ppm (serviceberry [Amelanchier sp.] foliage) to 22 000 ppm (white birch [Betula papyrifera] foliage). Foliar concentrations of sodium ranged from 10 ppm (black spruce [Picea mariana] foliage) to 86 ppm (serviceberry foliage). Background foliar concentrations of chloride ranged from less than 20 ppm (serviceberry and white birch) to 1900 ppm (trembling aspen). Background foliar concentrations of sodium ranged from 7 ppm (black spruce) to 19 ppm (serviceberry).
Twig concentrations of sodium and chloride were also elevated in these areas. Twig concentrations of chloride ranged from 900 ppm (trembling aspen) to 9100 ppm (black spruce). Twig concentrations of sodium ranged from 9 ppm (trembling aspen) to 78 ppm (black spruce). Background twig concentrations of chloride ranged from less than 20 ppm (trembling aspen) to 200 ppm (black spruce). Background twig concentrations of sodium ranged from 7 to 17 ppm (both in black spruce).
Surface soil concentrations of sodium and chloride at these two sites, taken at 0- to 5-cm depth, were elevated compared with a control site remote from the area. The soil concentration of chloride ranged from 780 to 1200 ppm, and the soil concentration of sodium ranged from 370 to 680 ppm. Background concentrations were 15 ppm chloride and 91 ppm sodium.
Changes were initiated in the winter of 1988-89 to reduce losses of salt from the patrol yard operations. The area and severity of the vegetation injury had decreased by August 1989. The concentrations of sodium and chloride in the foliage also declined, although levels were still elevated in comparison with the control.
3.5.4 Changes in application rates and current injury in Canada
The Canadian field studies discussed in Section 3.5.3 and the environmental concentrations discussed in Section 3.5.1 use data on plant or soil chloride and sodium concentrations and plant injury data from field sampling done between 1980 and 1995. Application rates of sodium chloride have decreased by 20-30% in some jurisdictions since the 1980s; however, total annual loadings have not declined (Morin and Perchanok, 2000).
The total amount applied during a winter (annual loading) and the total amount of soil or aerial deposition are the critical values for plants (Hofstra and Lumis, 1975). The total amount of salt applied during a season will be higher in winters with more snow events, with more days with snowfall, with higher snowfall amounts per event or with more freezing rain events. As well, plants are exposed to more aerial salt spray in years with more salt applications, more early-and late-season storms, and freezing conditions requiring salt application that begin earlier than usual in the fall and extend later into spring.
Current data from two municipalities in Ontario and Alberta demonstrate the variation in total application of road salts per winter season. In the North District of the Region of Peel in Ontario, total application amounts per year per lane-kilometre varied by 22% over 1995-2000, from 22 to 27 tonnes per lane-kilometre (Zidar, 2000). Over the years of 1996-97 to 1998-99, the City of Edmonton practised "sensible salting" and "best management practices" to minimize the effect of road salt on the environment. They found that total road salt usage during this period ranged from 11 000 tonnes sodium chloride plus 69 400 L calcium chloride to 21 300 tonnes sodium chloride plus 40 900 L calcium chloride. This represents a variation of more than 90% for sodium chloride and 70% for calcium chloride (Neehall, 2000).
In other scenarios, with early- or late-season storms that occur when the frost is out of the ground, more salty runoff would penetrate the soil around plant roots rather than run off over frozen ground (Headley and Bassuk, 1991). Spring salt applications would also result in aerial salt deposition on expanding woody buds and succulent young shoots as well as soil exposure of germinating seedlings. In years with dry springs, salt deposition on woody plant stems and leaves may not be washed off by rainfall (Hofstra et al., 1979; Simini and Leone, 1986), and salt present in soil may not be leached as much as in years with wetter springs. All these variables mean that salt exposure of plants via soil or aerial deposition goes up and down depending on climatic conditions each year (Hofstra et al., 1979).
Salt injury on roadside plants remains evident along rural and urban roadside conditions in Canada. The City of Edmonton has observed negative impacts attributable to salt on roadside grass, shrub and tree vegetation, but notes that there are few alternatives to road salt to achieve safe winter driving conditions (Neehall, 2000). Although city staff practise "sensible salting" and "best management practices" to minimize the effect of road salt on the environment, they have found that soil chloride concentrations are exacerbated in years with drought conditions that reduce soil moisture and flushing of soluble salts from the soil. Negative impacts on roadside trees in Edmonton varied in severity from year to year and were more severe in drought years.
Salt injury to trees has also been observed in rural areas of Ontario during the 2000 growing season - for example, along County Road 124 in Dufferin County (Figure 3.4 in Cain et al., 2001, and accompanying discussion) and along Highway 401. The Region of Peel in Ontario evaluated salt damage to trees along roads in the northern part of their district on roads maintained by the Region of Peel and the Ontario Ministry of Transportation (Thompson, 2000; Zidar, 2000). Injury seemed to be more severe than usual following the 1999-2000 winter. Salt injury was observed most often on sensitive tree species, such as eastern white cedar (Thuja occidentalis), red pine (Pinus resinosa) and Scots pine (Pinus sylvestris). Other contributing factors included summer drought conditions during the previous two summers, which contributed to tree stress, and the occurrence of mild winter days over the 1999-2000 winter, which possibly resulted in wet road conditions, contributing to salt spray.
The occurrence of injury also depended on the thickness of forest along the roads, tree distance from the road, soil type and slope, speed of road and volume of traffic.
3.5.5 Estimate of area of potential injury
The zone of impact of road salts on susceptible plants extends generally up to 80 m from the edge of multi-lane highways or freeways and up to 35 m from two-lane highways. Using highway length data, one could estimate the area of land in Canada where sensitive vegetation could be affected by road salts. Using data published by Transport Canada (2001), the length of freeways in Canada in 1995 was 16 571 two-lane-equivalent kilometres. This number could be divided by 2 to approximate the linear length of highway. The potential area of land where sensitive plants would be affected up to 80 m from the edge of the highway would be 132 568 ha. In the same year, the length of urban and rural paved roads in Canada was 301 348 two-lane-equivalent kilometres, providing a linear length of the same amount. Along these roads, the potential area of land where sensitive plants would be affected up to 35 m from the edge of the roads would be 2 109 436 ha.
In total, sensitive vegetation could be affected by road salts on an estimated 2 242 000 ha of land along Canadian roadsides, using road length data for 1995. Not all of these roads and highways are salted, however, so the actual amount of potentially affected land would be substantially less.
3.5.6 Risk assessment summary and conclusions
Sodium chloride applied as a de-icer can have a harmful effect on plants growing along roads or in areas that have localized contamination due to runoff with elevated salt levels. Elevated soil levels of sodium and chloride or aerial dispersion of sodium and chloride results in severe reductions in flowering and fruiting of sensitive plant species; severe foliar, shoot and root injury; growth reductions; and reductions in germination and seedling establishment. The species affected include native grass species; wetland species, such as sphagnum moss and sedges; and woody species, such as maple, pine, Douglas-fir, dogwood, peach and plum.
Severe effects may result in plant mortality, while repeated (chronic) exposure of perennial plants to elevated salt levels, year after year, results in reduced plant growth and death of shoot and leaf tissue. Repeated stress on plants reduces plant vigour and the ability to deal with insects, disease and abiotic stresses. These effects can lead to loss of plants within a community as well as impacts on other organisms that rely on plants for food and cover.
Changes in plant communities in response to elevated levels of de-icing salts have been recorded in areas impacted by road salt runoff and aerial dispersion. Halophytic species, such as cattails and common reed-grass, readily invade areas impacted by salt, leading to the changes in occurrence and diversity of salt-sensitive species. The composition of non-salt-tolerant communities changes due to the loss of sensitive species and replacement by salt-tolerant species.
The zone of impact is usually linear, along roads and highways or other areas where sodium chloride or other chloride salts are applied for de-icing or dust control. Salt injury also occurs along drainage courses that direct salty runoff from roads and salt handling facilities. Aerial dispersal of sodium and chloride occurs along roads during the winter months when de-icing is required and impacts mainly woody plants. The impact of aerial dispersion of sodium and chloride extends up to about 80 m from the edge of multi-lane highways and up to about 35 m of two-lane highways where de-icing salts are used. The degree and distance of dispersion depend on the salt load, traffic volume and velocity, local topography and prevailing winds.
Impacts of road salts on forest species adjacent to roadsides have been documented in central and southern British Columbia. In these areas, the zone of impact was in the first 10-15 m from two-lane highways. Adjacent to a four-lane highway in British Columbia, the zone of severe injury of forest trees covered the first 50 m from the highway. Areas of high forest density in Canada that experience medium to high salt loadings (based on average annual loadings, by maintenance district) include Vancouver Island, southwestern and central Alberta, central Ontario and parts of southern Ontario, southern and central Quebec, and areas in New Brunswick and Nova Scotia. Fifty-three percent of the primary species of forest trees in Canada have been rated as sensitive to road salt.
Fruit crops experience severe plant injury and reduced fruit production in a zone up to 50-80 m from four-lane highways in southern Ontario and about 35 m from two-lane highways in Nova Scotia. Sixty-five percent of 32 cultivars or varieties of fruit trees, vines or shrubs have been rated as sensitive to salt. The sensitivity of these species provides an indication of sensitivity of native fruiting plants that occur in natural communities along roadsides.
Sensitive landscape plantings and nursery crops suffer severe plant injury and plant death due to elevated soil levels of road salt and salt spray. From 30 to 40% of common landscape species used in Ontario and throughout Canada have been rated as sensitive to road salts.
3.6 Terrestrial wildlife
The effects of laying down a road network on vertebrate wildlife, both positive and negative, have been the subject of numerous scientific reviews (e.g., Bennett, 1991), and these aspects will not be covered here. Impacts specific to the use of road salts may be mediated through a reduction in plant cover or community shifts as described in Section 3.5. Under some conditions, especially where habitat is limiting, road verges are valuable, both as breeding habitat for wildlife and as a corridor between suitable habitat patches (e.g., Oetting and Cassell, 1971; Drake and Kirchner, 1987; Bennett, 1991; Camp and Best, 1993a,b). Efforts are currently under way in several jurisdictions to improve roadside management in order to increase the value of this habitat. Road salts can have an adverse effect on these management efforts. Also, concerns about the impact of road salts on species with an aquatic life stage such as frogs and other amphibians were covered in Section 3.3. This section addresses the direct toxicological effects of road salts on terrestrial wildlife, namely birds and mammals.
3.6.1 Exposure characterization
Sodium is an indispensable component of the physiological processes of all vertebrates, but it is toxic when taken in excess. Typically, surplus sodium is removed by an increase in glomerular filtration rate and a decrease in the percentage of sodium reabsorbed by the kidneys. Terrestrial birds, especially herbivorous and granivorous species, are more likely to be salt deficient and are poorly equipped to deal with excess sodium. Any animal attempting to satisfy a "salt hunger" generally overshoots its deficit by a substantial amount, and the increase persists for some time after the deficit is replaced (Schulkin, 1991). Overconsumption of salt is in turn regulated via a feedback mechanism: the thirst response. The absence of a source of water therefore increases the toxicity of ingested salt (see Section 3.6.2.1). Sodium-deficient wildlife are known to travel great distances to obtain and ingest salt and to visit roads where salt has been applied (Schulkin, 1991). One concern raised by this assessment is that the "footprint" of a salted road may be quite large when highly mobile wildlife species are attracted from great distances.
3.6.1.1 Mammals
The most visible interaction between road salt and wildlife concerns large ungulates, such as moose (Alces alces), mule deer (Odocoileus hemionus), white-tailed deer (Odocoileus virginianus), elk (Cervus canadensis) and bighorn sheep (Ovis canadensis). Although there may be other reasons for mammalian wildlife to visit roadsides, the use of road salt has repeatedly been identified as a major factor contributing to car strikes. In Ontario, the peak of moose-vehicle accidents corresponds to the period of highest sodium hunger, not the period of highest vehicular traffic (Fraser and Thomas, 1982). Most sightings of moose and about half of all moose-vehicle accidents occurred at or very near salt pools (Fraser and Thomas, 1982). A technical report published by the Ontario Ministry of Transportation states that deer and moose drinking salty water tend to lose their fear of humans and vehicles and are prone to bolt, sometimes in the path of a vehicle, rather than move away as they normally would (Jones et al., 1986). In one Quebec study, moose visitation to roadside ponds increased with sodium and calcium levels, and the number of car strikes was higher near ponds receiving higher moose visitation (Grenier, 1973). Overall, there were twice as many moose killed per kilometre of road when roadside pools were present than where there were no pools. Miller and Litvaitis (1992) found that radio-collared moose in northern New Hampshire extended their home ranges in order to include roadside pools heavily contaminated by salt. Road salt attraction has been identified as a main reason for kills of bighorn sheep and a minor reason for kills of elk in Jasper National Park (Bradford, 1988). The role that road salt could play in the mortality of other wildlife species such as small mammals commonly killed by traffic has not, to our knowledge, been the subject of formal study, but another Ontario Ministry of Transportation report states that woodchucks (Marmota monax), snowshoe hares (Lepus americanus) and porcupines (Erethizon dorsatum) are frequently observed feeding on roadside salt (Hubbs and Boonstra, 1995). Trainer and Karstad (1960) reported a case of poisoning by road salt in eastern cottontails (Sylvilagus floridanus).
3.6.1.2 Birds
Twelve published reports of bird kills associated with salted roads were identified (Brownlee et al., 2000). Two incidents were formally diagnosed as salt poisoning (Trainer and Karstad, 1960; Martineau and Lair, 1995), and observations of aberrant behaviour suggestive of toxicosis were made in several other cases. Sodium chloride was usually identified as the substance to which birds were exposed, although in one case the road salt applied was calcium chloride (Meade, 1942). Geographically, these kills occurred at several sites within the Canadian and U.S. snow belt from 1942 to 1999. One report originated from Germany. At least two reported kills were large, involving more than a thousand birds each. Given the difficulty of finding avian carcasses (Mineau and Collins, 1988), the high rate of scavenging reported from sites where birds are routinely killed at roadside (Benkman, 1998; Woods, 1998) and the low rate of reporting for wildlife mortality events in general, the number of reports of bird mortality associated with the use of road salt is significant and suggests that kills are more widespread and frequent than indicated by documented reports alone. Supporting this view is the fact that other researchers and biologists, when contacted, were very familiar with kills of birds on salted highways, even though their observations had not been published. In one location (Mount Revelstoke Park), mortality of siskins and other winter finches (see below) has been seen frequently enough over the last 25 years for the birds to be called "grill birds" by the local inhabitants, in reference to their propensity to be collected by the front end of moving vehicles (Woods, 1998). The kills often occur in sections of the highway with curves or where there are many cracks and crevasses in the pavement where salt can accumulate, although they may occur anywhere the roads have been sanded and salted. Baker (1965) stated that the inhabitants of one Maine community "looked upon this bird slaughter as a natural and everyday occurrence." Another researcher reported seeing "hundreds of dead crossbills that were hit by vehicles while on the road eating salt" (Benkman, 1998).
Cardueline finches (family Fringillidae, subfamily Carduelinae: crossbills, grosbeaks and siskins) were involved in 11 out of 12 published incidents. All cardueline finches are protected species listed under the Migratory Birds Convention Act. This group of seed-eating birds ranges throughout the boreal forest, moving to more southern latitudes when seed yield is low. The extent of these "invasions" is best visualized through animated computer-generated maps of yearly abundance (see Cornell Laboratory of Ornithology, 2000a,b). Although finches will eat deciduous seed, insects and berries, their primary diet, particularly in the winter, consists of coniferous seeds. Their attraction to salt is well known. Red crossbills (Loxia curvirostra) have been trapped using salt as bait (Dawson et al., 1965), and cardueline finches were found to prefer sodium over other minerals regardless of grit size (Bennetts and Hutto, 1985). Fraser and Thomas (1982) identified three finch species - evening grosbeaks (Hesperiphona vespertina), purple finches (Carpodacus purpureus) and pine siskins (Carduelis pinus) - as the main visitors to roadside salt pools in northern Ontario.
The information from reported kills and the behavioural/social characteristics of the cardueline finches suggest that this is the group most at risk of acute salt toxicity from ingesting road salt.
However, because the birds tend to travel and forage in large flocks in the winter, there is undoubtedly a bias for a higher detectability and reporting of kills. Trainer and Karstad (1960) reported bobwhite quail (Colinus virginianus), ring-necked pheasant (Phasianus colchicus) and feral dove (Columba livia) as well as eastern cottontails being poisoned.
It is often assumed that salt is ingested in order to fulfil a physiological need associated with a largely vegetarian diet. However, the taking of salt crystals as grit cannot be ruled out. Grit is used to aid in the grinding of food and to provide supplementary minerals to bird species with low-calcium diets (Gionfriddo and Best, 1995). Across species, grit size preferences are linearly related to bird size. The size of road salt particles from a salt mine in Ontario ranged from 0.6 to 9.5 mm (Canadian Salt Company Limited, 1991). Because salt particles applied to roads undergo a gradual size reduction as they melt (Letts, 1999), salt particles overlap broadly with the preferred grit size for any size of bird from the time of application to some undetermined time after application. The large variation in the starting size of road salt particles ensures that a suitable size of grit will be available for an extended period.
3.6.2 Effects characterization
3.6.2.1 Acute toxicity of sodium chloride
Incidents of vertebrate salt toxicity in domestic or captive stock fall under several main categories: accidental overdose in feed beyond a level that can be compensated for by drinking more water (Sandals, 1978; Swayne et al., 1986; Howell and Gumbrell, 1992; Wages et al., 1995; Khanna et al., 1997), exposure to saline drinking water (Franson et al., 1981), exposure to saline lakes (Windingstad et al., 1987; Meteyer et al., 1997) or the provision of salt supplements with deprivation of water (Trueman and Clague, 1978; Scarratt et al., 1985). Reports of salt toxicosis in free-ranging vertebrate wildlife have been the result of exposure to road salt, drought, ice, hypersaline lakes and discharge ponds from various industries (Baeten and Dein, 1996).
Sodium chloride has an oral LD50 of 3000 mg/kg-bw in rats and 4000 mg/kg-bw in mice (Bertram, 1997). Sodium chloride administered as a saturated water solution (approximately 2100 mg/kg-bw) usually resulted in death within 24 hours (Trainer and Karstad, 1960). Repeated small doses of salt produced no illness if water was not restricted.
The Canadian Cooperative Wildlife Health Centre in Saskatoon and the National Wildlife Research Centre of Environment Canada recently conducted a joint study on the acute effects of salt on wild-caught house sparrows (Passer domesticus) (Wickstrom et al., 2001). The "up-and-down" method was used in a pilot study to estimate the approximate lethal oral dose of granular sodium chloride in birds that were non-fasted but without access to water for 6 hours post-dose. Results indicated an approximate LD50 of 3000-3500 mg/kg-bw, which is similar to the rodent values. A tentative no-effect level (mortality) was estimated to be 2000 mg/kg-bw in this pilot study, although mortality was seen at 1500 mg/kg-bw in a subsequent phase of the study.
3.6.2.2 Sublethal effects of excess salt ingestion in birds and mammals
Several authors reporting wildlife incidents associated with road salt made note of behavioural deficits in exposed birds. The most common observation is that the birds appeared unusually fearless and could be approached easily (Meade, 1942; Trainer and Karstad, 1960; Baker, 1965; Thiel, 1980; Smith, 1981; Woods, 1999). Others described the birds as "weak and slow" (Martineau and Lair, 1995) or appearing sick (Meade, 1942). Trainer and Karstad (1960) reported "depression, tremors, torticollis, retropulsion, and partial paralysis" for birds and mammals affected in one incident.
These signs are closely matched by observations made in the course of laboratory studies. Pheasants fed increased amounts of sodium in their mash exhibited signs of intoxication when the drinking water was restricted. These included depression followed by excitement, tremors, torticollis, opisthotonos, retropulsion, complete incoordination and coma (Trainer and Karstad, 1960). Not all signs were seen in the same animal. Frequent signs reported in poultry and in swine include salivation, diarrhea, a staggering gait, muscular spasms, twitching and prostration (Humphreys, 1978). In Wickstrom et al. (2001), following the pilot study reported above, house sparrows were dosed orally with granular sodium chloride at 0, 500, 1500, 2500 or 3500 mg/kg-bw. Overt clinical signs were observed at 1500 mg/kg-bw or higher and included rapid onset (<30 minutes) of depression, ataxia and inability to fly or perch, with death in as little as 45 minutes. Birds that survived for 6 hours usually recovered. Plasma sodium concentrations above 200 mmol/L were consistently associated with overt clinical signs. Lesions in the form of gizzard edema were observed after 1 hour in most birds dosed with 500 mg/kg-bw or higher, and brain stem vacuolation, also consistent with edema, was seen in some birds showing clinical signs.
There is no record of moose or deer suffering from salt toxicosis, although there are anecdotal observations that moose drinking salty water tend to lose their fear of humans and vehicles (Jones et al., 1992), in contrast to moose not drinking salty water, which move away from approaching humans. The only documented case of small mammals exhibiting signs of salt toxicosis is eastern cottontails, reported during a severe winter in Wisconsin (Trainer and Karstad, 1960).
3.6.3 Risk characterization
Mortality has been documented and is clearly much more frequent and widespread than is reflected by the published record. The risk to cardueline finches at least is significant. Locally, the risk to large mammals is also high. It is clear that the mere presence of road salt contributes to vehicle collisions by increasing the attractiveness of roadsides. The main uncertainty in the risk characterization is whether salt toxicity increases the vulnerability of some species to vehicle collisions. Also, if salt toxicity is occurring, it is relevant to ask whether wildlife, cardueline finches in particular, could be killed by salt ingestion, even in the absence of a vehicle strike.
3.6.3.1 Estimating the contribution of salt toxicity to vehicle strikes
The severe behavioural impairments (depression, tremors, torticollis, retropulsion and partial paralysis) observed at one site, as well as the finding of elevated brain sodium levels at another, argue strongly for a contributory role of salt toxicity to roadside bird kills. The more common observation of unusual fearlessness documented in both birds and mammals could be an early symptom of intoxication or the manifestation of animals under extreme salt hunger modifying their usual behaviour towards potential danger.
Although overt signs of intoxication were seen in the house sparrow at 1500 mg/kg-bw and higher, the level of dosing at which a more subtle behavioural impairment could be manifest is not known. A lower limit for potential impairment, using the house sparrow as a model, was estimated as follows.
The distribution of normal sodium values in the plasma of the sparrows is very narrow (mean 163.2 mmol/L, range 158-168 mmol/L, n = 12). The assumption is that adverse effects (such as reduced coordination and weakness) may begin when sodium homeostasis is breached and elevated sodium levels are being delivered to target organs. This level in the house sparrow model was determined to be 266 mg/kg-bw (Brownlee et al., 2000).
To estimate exposure, we assume that salt crystals are most likely to be picked up whole, as are grit particles. A finch trying to satisfy a salt hunger might pick up a granule of a size that it is "comfortable" with. The maximum recorded size for grit in the house sparrow is 2.4 mm (Gionfriddo and Best, 1995). Table 24 shows the number of particles that need to be ingested by birds in order to attain specific CTVs. This calculation suggests that birds could be sublethally impaired following ingestion of a single large salt particle and could be killed by as few as two large particles.
On the other hand, the average size of grit ingested by the house sparrow was 0.5 mm (Gionfriddo and Best, 1995). Assuming birds were intent on picking up salt particles of this "preferred" size for digestion, the above calculations were repeated for average-sized grit. In fact, this average grit size is likely an underestimate, because it is based on "worn" grit extracted from sparrow gizzards. The average also encompasses very fine mineral particles, which were probably ingested accidentally along with food. These results are given in Table 25.
Although the number of particles that need to be ingested to reach CTVs is much higher than that calculated in Table 24, this number of particles can easily be ingested based on a comparison with average grit counts. Therefore, birds in need of replenishing their grit supply may easily achieve doses that would be toxicologically relevant.
3.6.3.2 Conclusions and discussion of uncertainty
Road salt is widely acknowledged to be an important factor in attracting large mammals to roads, and it does appear to increase the frequency of vehicular collisions.
It is clear that, even without road salt, some birds would be coming to highways for grit, clay, road-killed prey, etc. However, observations made at roadside and the biology of cardueline finches especially suggest that road salt is an important attractant. The evidence to date suggests that road salt increases the vulnerability of birds to car strikes by causing impairment. It is common sense that birds at the roadside that are "weak and slow" (Martineau and Lair, 1995), that appear sick (Meade, 1942) or that show "depression, tremors, torticollis, retropulsion, and partial paralysis" (Trainer and Karstad, 1960) have a diminished capacity to avoid moving vehicles. The biological effects of road salts differentiate them from, for example, sand, which is known to attract birds in search of grit to highways but which does not cause impairment. Furthermore, intake calculations suggest that road salt may poison some birds directly, especially when water is not freely available during severe winters. Birds have been observed eating snow, catching snowflakes during a storm and chipping ice to obtain water (Wolfe, 1996). Oeser (1977) reported that crossbills were eating snow in the reported German kill incident. There could be negative energetic consequences of snow ingestion in response to salt overconsumption. Furthermore, melted snow on the roads may be the most obtainable source of water, but, depending on the concentration of salt in the snowmelt, consumption of such water may increase salt ingestion rather than alleviate salt toxicosis.
A remaining uncertainty is whether the house sparrow is a suitable toxicological model for cardueline finches and other native birds. The house sparrow has Middle Eastern origins and may therefore be genetically predisposed to be more salt tolerant than cardueline finches. On the other hand, consumption rates of grit may be high in house sparrows relative to other bird species (Gionfriddo and Best, 1995). The scenarios presented above assumed a consumption of salt based on grit need (Table 25) as well as on optimizing salt intake by ingesting larger particles at the limit of tolerated grit size (Table 24). Regardless of the exact particle size taken, salt-deprived birds could ingest toxicologically relevant doses before feedback mechanisms reduce the desire for salt.
The overlap between the distribution of winter finches and the Canadian road system receiving road salt was determined to be extensive when mapped by Brownlee et al. (2000). This is especially true during "invasion" years when the finches descend out of the boreal forest. It is reasonable to assume that finches will be attracted more to some types of roads than to others. For example, a busy multi-lane highway servicing a large metropolitan area may not offer many opportunities for a flock of birds to alight, regardless of how much salt is available. However, the data needed to assess the probability that a flock will land on any given roadway or the proportion of those visits that result in kills (whether fatal attraction, toxicosis-induced car strikes or lethal ingestion) are not available. The length of time the applied salt is retained on or near the road surface (e.g., in the soil), where it could create an artificial "lick" attractive to both birds and mammals even after granules have disappeared, is highly stochastic and difficult to assess, but undoubtedly important.
Despite these uncertainties, the scientific data available to date suggest that the number of road kills of federally protected migratory bird species (especially the cardueline finches) and the contribution of road salt to this mortality have been underestimated by wildlife managers and transport officials. On the other hand, the importance of road salt in increasing the number of collisions between large mammals and motor vehicles has long been recognized and acknowledged by transport officials.
3.7 Ferrocyanides
This section is based on a detailed review of ferrocyanides presented by Letts (2000a).
3.7.1 Exposure characterization
Sodium ferrocyanide dissolves in water to release the stable ferrocyanide anion (Fe(CN)64-), which is not volatile and resists further breakup unless illuminated. The anion is fairly mobile in groundwater but reacts readily with iron to form the precipitate ferric ferrocyanide, a very stable, non-toxic, relatively immobile compound, which is a major removal pathway of ferrocyanide from groundwater. Under favourable conditions, including non-illumination, the half-life of ferrocyanide is approximately 2.5 years (Higgs, 1992), while that of ferric ferrocyanide can extend to a thousand years (Meeussen et al., 1992a).
Two characteristics of the ferrocyanide anion are paramount. It is of extremely low toxicity in the complexed form (Rader et al., 1993), but in solution under direct light it photodecomposes, liberating free cyanide, which rapidly reacts to produce the highly toxic and volatile hydrogen cyanide (HCN). Outside of solution, photolysis of ferrocyanide is slight (American Cyanamid Company, 1953).
Photolysis of the ferrocyanide anion is a well-studied phenomenon (Broderius, 1973; Broderius and Smith, 1980; Otake et al., 1982; Clark et al., 1984; Meeussen et al., 1992a). It can liberate as many as five of the six cyanide ions associated with the parent compound. The reaction is a function of the initial concentration, intensity of illumination, pH and temperature, among other factors.
Free cyanide in the environment is subject to a number of attenuation processes. It is readily chelated with numerous transition metals to form more stable, less toxic materials (Chatwin, 1990). The presence of clay or organic matter removes free cyanide by surface adsorption (Theis and West, 1986). Additionally, it is oxidized to the relatively innocuous cyanates and thiocyanates in the presence of sulphur.
3.7.1.1 Soil
The largest portion of research covering the behaviour of non-illuminated ferrocyanides in soil has focused on waste compounds from former manufactured gas plant sites. Studies at such sites indicate that the presence of ferrocyanide is tightly restricted around the source site (Effenberger, 1964; Parker and Mather, 1979). This low mobility is apparently due to the presence of sulphur and transition metals in the soil. Iron under pH conditions of about 4-7 combines with the ferrocyanide anion to produce the sparingly soluble ferric ferrocyanides known as prussian blue (Meeussen et al., 1992b). This compound is evident in the blue coloration of the soil at many abandoned manufactured gas plant sites. In alkaline soils (pH above 7), ferrocyanide in solution retains the anion form and is fairly mobile. It has been noted that under strongly acidic conditions, ferrocyanide anions dissociate to release cyanide ion (Belluck, 2000). It has been suggested that the appropriate conditions may occur in the laboratory but seldom or never in the environment (Chatwin, 1990).
Retention and immobilization in soils in low-toxicity forms are the major attenuation pathways limiting the potential toxic effects of ferrocyanide. Particle adsorption in clay-bearing moderate-pH soils may effectively contain ferrocyanide anions. Goethite (--FeOOH), a common surface coating of soil particles, removes as much as 95% of the ferrocyanide anion through adsorption at pH 4 (Theis and West, 1986). These removal methods are abetted by moderate pH levels. Soils in Canada are acidic in forested regions, and especially so in Eastern Canada, where salting activities are heavy. In contrast, soils are neutral to alkaline in the Prairies, where soils are naturally salt enriched (Acton and Gregorich, 1995; see also CANSIS at http://sis.agr.gc.ca/cansis/intro.html).
Immobilized ferrocyanide is also subject to chemical conversion to compounds of low toxicity in the presence of sulphur, and microorganisms that degrade metal-complexed cyanides have been identified (Mudder, 1991).
Photolysis of ferrocyanide under illumination is a complex, intensively studied reaction (Broderius, 1973; Broderius and Smith, 1980; Otake et al., 1982; Clark et al., 1984; Meeussen et al., 1992a). Factors affecting the process include pH, temperature, initial concentration, season and time of day. In their study of photolytic reactions, Miller et al. (1989) reported that free cyanide release is restricted to a soil depth of 1 mm. This suggests that only ferrocyanide anions within the top 1 mm of soil are subject to the process.
Hydrogen cyanide produced within the soil is mildly persistent. It has an estimated half-life of 4 weeks to 6 months (Howard et al., 1991). It is adsorbed at the rates of 0.05 mg/g by clays and 0.5 mg/g by organic matter (Chatwin, 1989). It is also subject to hydrolysis to formic acid up to a rate of 4% in saturated soils (Chatwin, 1990). Volatilization of hydrogen cyanide from soils accounts for as much as 10% of losses, but saturation of the soil retards this process by a factor of 104 (Chatwin, 1990). Biological degradation of various cyanides by soil microbes (Towill et al., 1978; Dubey and Holmes, 1995) or assimilation by plants (Fuller, 1984; Knowles and Bunch, 1986) is an efficient removal system that is strongly diminished under saturated anaerobic conditions (Strobel, 1967).
3.7.1.2 Water
Ferrocyanide dissociates very slowly under non-illumination. Under this condition, the amount of free cyanide released from concentrations of ferrocyanide in the range of 1-100 000 mg/L varies from 1.2 to 6.1 µg/L. However, under environmental illuminated conditions, rapid conversion of ferrocyanide to free cyanide has been reported (Ferguson, 1985; Singleton, 1986).
In water, free cyanide is converted to hydrogen cyanide, a weak acid, through the equilibrium reaction H+ + CN- - HCN. The hydrogen cyanide state is highly favoured below pH 6. Hydrogen cyanide is highly volatile and, under environmental conditions, quickly dissipates from water bodies, its volatilization rate being a function of concentration, temperature, pH and water flow rate (Schmidt et al., 1981; Melis et al., 1987). Secondary cyanide removal processes include complexation and precipitation, biological degradation, adsorption by organic material and uptake by plants. A number of naturally occurring organisms are known to produce cyanides (Knowles, 1976).
3.7.1.3 Air
Ferrocyanide itself does not volatilize. Hydrogen cyanide is highly volatile and is expected to rapidly dissipate in the atmosphere, with an estimated half-life of 89 days to 2.4 years (Howard et al., 1991).
3.7.1.4 Biomagnification
Towill et al. (1978) found no studies reporting biomagnification of cyanide.
3.7.2 Measured and estimated environmental concentrations
Reported cyanide concentrations measured in runoff waters range from 2.3 to 22 µg/L for free cyanide and from 3 to 270 µg/L for total cyanide (Hsu, 1984; Ohno, 1989; Novotny et al., 1997; Mayer et al., 1998). Since actual data are limited, estimated concentrations were also derived.
Morin and Perchanok (2000) documented road salting activity in Canada. The highest level of use in Canada is 10 150 g salt/m2, and the highest addition rate of sodium ferrocyanide to road salt is reported to be 124 mg/kg (Letts, 2000b). This corresponds to 86 mg ferrocyanide/kg, or a stoichiometric cyanide content of 63.7 mg/kg. From these values, the distribution rate is calculated as being 877.8 mg ferrocyanide/m2 or 646.6 mg cyanide ion/m2. Using annual precipitation data, the concentration range of ferrocyanide anion in runoff water can be determined to be 0.000 142-1.42 mg/L, which corresponds to a stoichiometric content of 0.000 104-1.04 mg cyanide ion/L.
In soils, since local conditions will dictate the mobility of ferrocyanide, two sets of assumptions are proposed. Under Case I, ferrocyanide is precipitated out of solution and the annual application is retained in the upper 2 cm of soil. Under Case II, ferrocyanide remains in solution and 20% of the annual application is retained in the first 20 cm of soil. For Case I, the resultant soil concentration ranges from 0.0043 to 43.8 mg ferrocyanide/kg soil, which corresponds to a stoichiometric cyanide ion content of 0.0032-32.3 mg/kg soil. Under Case II, it ranges from 0.000 086 to 0.88 mg ferrocyanide/kg soil, which corresponds to a stoichiometric cyanide ion content of 0.000 064-0.64 mg/kg soil.
3.7.3 Effects characterization
Initial assessment suggests that the greatest inherent danger in the use of sodium ferrocyanide lies with the ability of the ferrocyanide anion to photodecompose, resulting in free cyanide. Therefore, the following sections summarize the known effects of both these compounds on terrestrial and aquatic biota. The limitations of determining toxicity under artificial laboratory conditions must be taken into account.
3.7.3.1 Aquatic biota
The main factors affecting toxicity in water are concentration and water temperature. Toxicity is also modified by the level of dissolved oxygen and salinity. The literature contains a number of comprehensive reviews on the toxicity of cyanide to freshwater organisms (Doudoroff, 1966, 1976, 1980; Leduc et al., 1982; U.S. EPA, 1985). Some organisms display reversible acclimatization to the toxic effects.
The effects of cyanide are quite diverse among various microbial species. Many species exhibit remarkable resistance to noxious effects; indeed, some species are employed in cyanide detoxification processes (Chatwin, 1990; Fallon et al., 1991). On the other hand, adverse effects on Pseudomonas putida have been reported at the 1 µg/L level (U.S. EPA, 1985). This is the lowest concentration at which adverse effects on aquatic microorganisms have been reported (Letts, 2000a).
Data on toxicity to plants are scarce. The most sensitive species, Scenedesmus quadricauda, exhibits incipient cell inhibition under a 96-hour exposure to 31 µg free cyanide/L (U.S. EPA, 1985).
In invertebrates, onset of ferrocyanide toxicity generally exceeds 1000 µg/L. On the other hand, the lowest free cyanide 96-hour LC50 reported in the literature is 83 µg/L for Daphnia magna (U.S. EPA, 1985; Eisler, 1991). Divergent sensitivity in mixed subspecies groups may cause a population structure shift following exposure in some communities (Smith et al., 1979). For additional data, see Letts (2000a).
Toxicity to vertebrates has been intensively studied - for example, by Leduc et al. (1982). Vertebrates are less sensitive to ferrocyanide (Schraufnagel, 1965) than to free cyanide (Leduc et al., 1982). Fish as a group appear more sensitive than invertebrates to free cyanide.
The 96-hour LC50 estimates lie in the range of 40-200 µg hydrogen cyanide/L (U.S. EPA, 1985). A wide variety of physiological and behavioural effects has been reported. Life stage investigation suggests that the juvenile phase is most sensitive, with a negative impact on growth commonly reported. Adverse effects on predation and avoidance behaviours and swimming ability have been reported. Adverse reproductive effects have been selected as a toxicity target indicator. Schraufnagel (1965) and Smith et al. (1979) found spawning to be inhibited at concentrations of 5.0 and 5.2 µg/L, respectively.
3.7.3.2 Terrestrial biota
The primary effect of cyanide is an interference with respiration as a result of disruption of a mitochondrial enzyme (Towill et al., 1978; Way et al., 1988). If the effect is not rapidly fatal, recovery is generally quick.
Microorganisms show few adverse effects as a result of cyanide exposure. Some show adaptations, such as alternative respiratory pathways. Evidence suggests the existence of a "cyanide microcycle" enabling microorganisms as well as plants to use hydrogen cyanide as a source of nitrogen and carbon (Allen and Strobel, 1966).
As a group, terrestrial plants are fairly insensitive to ferrocyanide and free cyanide. As much as 10 g ferrocyanide/kg soil mix is needed to inhibit growth. Like microorganisms, some plants possess alternative respiratory pathways and/or the ability to metabolize hydrogen cyanide. However, some plants do exhibit adverse effects. A 25% inhibition of seedling emergence was observed in radish plants at concentrations as low as 1.2 mg/kg (Environment Canada, 1995). This represents the lowest concentration at which adverse effects on terrestrial plants have been reported.
While some invertebrates are resistant to cyanide or produce cyanides as a defensive mechanism, most demonstrate some level of sensitivity. The lowest free cyanide concentration inducing an adverse effect in terrestrial invertebrates is 4 mg/kg, which caused mortality in 25% of earthworms (Eisenia fetida) exposed for 14 days (Environment Canada, 1995).
Ferrocyanides have been approved as food additives and used as therapeutics. Rats exposed to 3200 mg/kg-bw per day were unaffected (Dvorak et al., 1971). For hydrogen cyanide, existing standards with a 7- to 8-fold safety margin limit exposure to 5 mg/m3 (Klaassen, 1966). The U.S. EPA developed a subchronic reference dose of 0.08 mg/kg-bw per day for free cyanide from available research. From the available literature, the mammal most sensitive to adverse effects is the rabbit. Ballantyne (1987) observed an LD50 value of 0.6 mg/kg-bw. Among birds, mallards (Anas platyrhynchos) appear to be the most sensitive, exhibiting an LD50 for oral ingestion of sodium cyanide of 2.7 mg/kg-bw (Henny et al., 1994).
3.7.4 Risk characterization
As previously stated, the greatest environmental danger from the use of sodium ferrocyanide is the ability of the ferrocyanide anion in solution to undergo photodecomposition, resulting in the release of cyanide ion and its subsequent hydrolysis to hydrogen cyanide. The process has been extensively examined by Broderius and Smith (1980), who reported the rate of reaction to be generally a direct function of light intensity (which depends on time of year, time of day, sky conditions), temperature and concentration and an inverse function of pH and latitude. A maximum of five out of six of the available molecular cyanides can be released.
For assessment under Tiers 1 and 2 assumptions, quotient values were calculated for biota by dividing the highest measured and/or estimated free cyanide concentrations in water, air or soil by the individual ENEVs. The ENEVs used correspond to the CTVs, which are the lowest effect concentrations found in toxicity studies from the available literature, divided by appropriate application factors.
3.7.4.1 Tier 1 risk characterization
The Tier 1 assessment applied worst-case condition correction factors in conjunction with typical Canadian location values to compute the photolysis rate. The reaction is proposed to occur at latitude 45°N at noon on a cloudless day of 4°C. The rate under these conditions is 28.5% per hour. The maximum number of cyanide anions that can be released from the ferrocyanide molecule through the photolysis process is five out of six. The cyanide so produced is considered to be conserved entirely within the considered environmental compartment.
Calculating from the range of annual mean peak cyanide concentrations expected in runoff water that was determined above, it can be seen that the maximum potential cyanide concentration after total potential cyanide anion release through photolysis ranges from 0.087 to 870 µg cyanide ion/L. Given a 1-hour reaction window, the cyanide ion released to water through photolysis gives a concentration ranging from 0.025 to 250 µg/L.
Using the maximum annual salt application rate (i.e., 10 150 g/m2) and Tier 1 and soil Case I assumptions, the maximum potential cyanide concentration after total potential cyanide anion release through photolysis would be 1.35 mg cyanide ion/kg soil. After a 1-hour reaction window, the cyanide ion released through photolysis would give a concentration of 0.39 mg/kg.
Using the maximum annual salt application rate (i.e., 10 150 g/m2) and Tier 1 and soil Case II assumptions, the maximum potential cyanide concentration after total potential cyanide anion release through photolysis would be 0.027 mg cyanide ion/kg soil. After a 1-hour reaction window, the cyanide ion released through photolysis would give a concentration of 0.0078 mg/kg.
The volatilization rate of cyanide at concentrations of 0.1-0.5 mg/L is 0.22 mg cyanide ion/m2 per hour (Higgs, 1992). For Tier 1 purposes, a closed-box model corresponding to a 10-cm layer of air above the roadway was used. After 1 hour, the maximum cyanide concentration in this layer would be 2.2 mg cyanide ion/m3.
Quotient calculations were based on the highest estimated free cyanide and measured total cyanide (assumed to be free cyanide for the Tier 1 analysis) concentrations in water (i.e., 250 and 270 µg/L) (Novotny et al., 1997) and were calculated as follows: Quotient = EEV/ENEV.
1 (m) = Using measured water concentration; (e) = Using estimated water concentration.
For terrestrial organisms, quotient calculations were undertaken using the highest estimated free cyanide concentration in soil under Tier 1 assumptions (i.e., 0.39 mg/kg soil). No measured free cyanide soil concentrations were available in the literature. For birds and mammals, EEVs were calculated using a multipath model incorporating intake from soil, air and water.
With the exception of terrestrial vertebrates and invertebrates, all quotients exceed 1 (Tables 26 and 27). Aquatic organisms appear particularly susceptible. A Tier 2 assessment is warranted.
3.7.4.2 Tier 2 risk characterization
The Tier 2 assessment, while still conservative, is based on more representative scenarios and assumptions. Typical and measured variables are generally employed.
The rate of addition of sodium ferrocyanide has decreased over the past years. The 1997 addition rate of 80 mg sodium ferrocyanide/kg salt corresponding to 55.8 mg ferrocyanide ion/kg and 41.1 mg cyanide ion/kg road salt is more typical of present-day use and will be used in Tier 2.
Labadia and Buttle (1996) measured the pattern of road salt dispersal along a section of southern Ontario highway. Their observations show that slightly more than 50% of applications are redistributed to the adjacent snowpack within a 3.7-m splash zone. This will also be taken into consideration in Tier 2.
The ferrocyanide photolysis rate calculation was corrected to reflect an average daily light intensity and a condition of 70% clear sky effect. Corrections were also made to account for changes in initial ferrocyanide concentration. Overall Tier 2 assumptions produced an adjusted photolysis rate of 13.8% per hour.
Based on Tier 2 assumptions and the annual mean peak chloride ion concentrations compiled by Morin and Perchanok (2000), the cyanide ion concentration range in water after 1 hour was calculated to be 0.0039-39 µg/L.
Using the maximum annual salt application rate (i.e., 10 150 g/m2) and Tier 2 and soil Case I assumptions, the soil concentration following the photolytic release of cyanide ion from the available ferrocyanide would be 0.12 mg cyanide ion/kg soil after 1 hour.
Using the maximum annual salt application rate (i.e., 10 150 g/m2) and Tier 2 and soil Case II assumptions, the soil concentration following the photolytic release of cyanide ion from the available ferrocyanide would be 0.0024 mg cyanide ion/kg soil after l hour.
The volatilization rate of cyanide at concentrations of 0.1-0.5 mg/L is 0.22 mg cyanide ion/m2 per hour (Higgs, 1992). For this assessment, it is assumed the available cyanide is completely transferred to the air above the roadway without dispersal and that the gaseous diffusion rate is the same as the interfacial diffusion rate. Under these conditions, after 1 hour, the maximum cyanide concentration in this layer would be 0.22 mg/m3.
Quotient calculations were undertaken using the highest estimated free cyanide concentration in runoff water under Tier 2 assumptions (i.e., 39 µg/L) and the highest measured concentration of 41 µg/L derived as the geometric mean of measured values reported in the literature. The CTVs used in the Tier 1 assessment are used again in Tier 2, but some application factors were modified to reflect a more realistic analysis.
Assessment of terrestrial organisms was conducted using the highest estimated free cyanide concentration in soil under Tier 2 assumptions (i.e., 0.12 mg/kg soil). No measured free cyanide soil concentrations were available in the literature. Calculation of EEVs for birds and mammals was the same as in Tier 1.
Based on the calculated quotients (Tables 28 and 29), a Tier 3 assessment should be conducted for aquatic organisms.
3.7.4.3 Tier 3 risk characterization
Tier 1 and Tier 2 analyses have shown that, through the formation of cyanide, sodium ferrocyanide can have the potential to negatively impact the aquatic environment. The Tier 3 assessment quantifies the probability of adverse effect within the aquatic compartment.
As already stated, photolytic release of free cyanide from ferrocyanide anion remains the most serious concern from ferrocyanide releases to the environment. Total photolysis across the relevant wavelengths and losses due to volatilization can be mathematically modelled; from these models, the concentration of free cyanide in solution can be calculated for any given time after the salt application event. These calculations were part of the Tier 3 assessment.
1 (m) = Using measured water concentration; (e) = Using estimated water concentration.
For purposes of Tier 3, the following assumptions and correction factors were used in estimating ferrocyanide photolysis and volatilization rates. Because photolysis is a function of water depth and clarity, ferrocyanide was considered to be contained within the first 20 cm, and clarity measurements from a range of water bodies were used. Likewise, the reaction rate is a function of date, time of day, sky conditions and temperature. Calculations were therefore made for every hour of the day for January 1, April 1, June 21 and September 21 under conditions of 70% clear sky effect with Henry's law constant corrected for an average temperature of 4°C. The observations of Labadia and Buttle (1996), who reported that approximately 50% of an initial salt application is redistributed to the adjacent snowpack, were, as previously, taken into consideration. Both stream and wind velocities were estimated from direct Canadian source measurements. Mean stream current was set at 0.52 m/s, and wind velocity was set to 5.0, 4.7, 3.8 and 3.8 m/s for the months of January, April, June and September, respectively. The chloride ion runoff water concentrations were selected from the range of values collected by Morin and Perchanok (2000). The probability of selection of a given concentration for inclusion in a calculation was a function of the size of the area reporting that concentration. Sodium ferrocyanide addition rates were selected for inclusion in calculations on a probability basis reflecting the percentage of the market served by the corresponding source mine.
Calculations were conducted by randomly selecting each variable from the appropriate frequency distribution. Variables included chloride concentration in runoff water, concentration of ferrocyanide in salt, current speed, wind speed and attenuation coefficient. Photodecomposition- volatilization curves were generated for each date. The maximum free cyanide value so produced was then recorded. This procedure was repeated 10 000 times, and the resulting "maximum free cyanide observed" values (EEVmax) were sorted into ascending order. The CTVs for each aquatic organism were then compared with these randomly generated distributions, and the probability that an exposure event would equal or exceed the CTV was determined. The procedure was run in duplicate using data derived from across Canada and from the heaviest use region, the Québec City to Windsor corridor. As extensive data on salt use by many Canadian municipalities were available, the above procedure was repeated using the salting data from three municipalities selected as representative of the highest, average and lowest Canadian municipal use. The selected municipalities were La Tuque, Quebec; Newmarket, Ontario; and White River, Ontario. Results are briefly summarized below. Full results are presented in the supporting document by Letts (2000a).
1) Aquatic microorganisms
Analyses indicate that Pseudomonas putida would be adversely affected in 100% of modelled exposures. In the municipality selected to represent a case of highest use, Microregma heterostoma would be expected to be affected adversely in as many as 60% of cases. However, the use of road salts in that municipality is twice or greater than the level in all but the two next highest users in 90 Canadian cities. For other cities, effects would be expected in 10 to 20% of scenarios. No adverse effects are indicated for any of the other five aquatic microorganisms considered. Thus, while ferrocyanide from road salts may be viewed as posing little threat to five of seven microorganisms for which data are available, the most sensitive species are susceptible to harm.
2) Aquatic plants
Results demonstrate that Scenedesmus quadricauda, the most sensitive alga identified, could be adversely affected in 16-28% of possible exposure scenarios, with a value as high as 82% in the municipality with highest use. Values therefore indicate that there is a potential for moderate effects at certain times for certain sensitive species, with lower levels of risk for most aquatic plants at most sites and times.
3) Aquatic invertebrates
Adverse effects on the doughboy scallop (Chlamys asperrimus) may occur on as many as 34% of exposures during April in the Québec City to Windsor corridor. The likelihood of negative effects on this organism falls to less than 25% of cases for other locations and dates. For the municipality with highest use, adverse effects were predicted for 88% of occurrences. No other organism is shown to risk harm at greater than 3.5% of exposure levels. Generally, members of this group are not considered at serious risk, but significant exposure of sensitive species may occur.
4) Aquatic vertebrates
The highest potential for significant exposures was indicated for aquatic vertebrates. Among the studies reviewed by Letts (2000a), the 96-hour LC50 for rainbow trout was reported to be 17 µg/L (Higgs, 1992), 27 µg/L (Smith et al., 1979) and 28 µg/L (Kovacs, 1979; Kovacs and Leduc, 1982). The probability of occurrence of concentrations equal to or greater than these is, respectively, 52.6%, 26.8% and 25.2% for January and 67.2%, 36.2% and 33.8% for April for locations within the Québec City to Windsor corridor. This is the area of higher concentrations of the two broad-based sources of data.
Concentrations at these levels are most likely to occur in areas where annual mean peak road salts chloride concentrations in runoff are 1000 mg/L or more (modelled as 53 out of 90 cities). Thus, in these areas, ferrocyanide in undiluted runoff could affect aquatic invertebrates.
3.7.5 Conclusion for ferrocyanides
The assessment indicates that, at present levels of road salt use, potential adverse effects related to sodium ferrocyanide are limited to aquatic biota, with possible negative consequences to sensitive species of aquatic microorganisms, aquatic plants and aquatic invertebrates. The more sensitive aquatic vertebrates in roadside ditches and water courses in areas of high use would be most susceptible to harmful exposures. The modelling of likelihood of effects was based on exposures predicted in roadside ditches, and did not account for further dilution by receiving waters, which would tend to overestimate exposure. At the same time, modelling was based on dilution of salts by total yearly precipitation, which would underestimate exposure at any given time. The ultimate impact of ferrocyanides on fish populations depends on a wide range of parameters, including those influencing the quantities of ferrocyanides entering water courses (e.g., mass and timing of road salts application, runoff), the photolysis of the ferrocyanides to cyanide (e.g., insolation, water turbidity, temperature), volatilization of cyanide (e.g., water flow, temperature), status of fish or amphibian populations in receiving waters, extent of dilution in receiving waters, etc. Accordingly, while available data indicate a potential for adverse effects, it is not possible to predict reliably the impact of ferrocyanide on vertebrate populations in water bodies adjacent to or near roadways.
3.8 Overall conclusions
3.8.1 Considerations
As provided by Paragraph 76(1) of CEPA 1999, PSL substances are to be assessed to determine whether they are "toxic or capable of becoming toxic." In this assessment, road salts are assessed as to whether they are "toxic or capable of becoming toxic" as defined in Paragraphs 64(a) and 64(b) of CEPA 1999, namely:
... a substance is toxic if it is entering or may enter the environment in a quantity or concentration or under conditions that
- have or may have an immediate or long-term harmful effect on the environment or its biological diversity;
- constitute or may constitute a danger to the environment on which life depends.
3.8.1.1 Environment
CEPA 1999 defines "environment" quite broadly, to include all biotic and abiotic components of the biosphere (CEPA 1999, Section 3):
"environment'' means the components of the Earth and includes
- air, land and water;
- all layers of the atmosphere;
- all organic and inorganic matter and living organisms; and
- the interacting natural systems that include components referred to in paragraphs (a) to (c).
This definition does not distinguish between "natural" and "non-natural" environments. Indeed, all ecosystems can be considered to have been subjected to stress or modifications by human activity, be it at a local (e.g., habitat destruction, emission of non-persistent pollutants in liquid effluent), regional (e.g., dispersion of atmospheric pollutants) or global scale (e.g., persistent organic pollutants, global atmospheric effects).
Road salts are stored or used and snow is disposed of in engineered environments -namely, storage or patrol yards, roadways and snow dumps. The degree to which these are in fact "engineered" is quite variable: salts can be stored in specially designed facilities at patrol yards or in temporary piles at the margins of roadways or patrol yards; roadways can be part of complex corridors with rights-of-ways with lined ditches or run parallel to and drain directly into streams, lakes or wetlands; snow dumps can range from secure facilities with meltwater retention and treatment to dumps on available vacant lots. Thus, the degree to which road salts are released to well-circumscribed engineered systems is quite variable, as is the level of concern or need for protection at the periphery of these engineered systems.
In some cases, elements of the environment that are part of or immediately adjacent to the engineered systems can be ecologically significant (Nadec, 2001). Roadsides have been shown to be extensively used as habitat by a considerable diversity of species (Perring et al., 1964; Oetting and Cassel, 1971; Way, 1977; Laursen, 1981; Adams and Geiss, 1983; Auestad et al., 1999; Bellamy et al., 2000). Accordingly, roadsides play two ecologically significant roles:
- Roadsides contribute to maintaining biodiversity by providing habitat for some species. For example, 35 of the 257 nationally rare plant species in Great Britain are found in roadsides, including some for which roadsides are the main remaining habitat (Way, 1977). In Quebec, 75 species of birds were observed in road rights-of-way, among which 29 were long-range migratory birds and 10 were year-round local residents (Lacroix and Bélanger, 2000). Migratory birds are protected species, subject to the provisions of the Migratory Birds Convention Act. In parts of Quebec, roadside ditches were reported to be the only site where the frog Pseudacris triseriata, identified as vulnerable on the provincial endangered species list, was observed to breed (Saumure, 2001). In southwestern Ontario, only 0.5-3.8% of the original tallgrass prairie or oak savanna ecosystems remain that existed at the time of land surveys during the 1700s and 1800s (Bakowsky and Riley, 1994). At least 35 native tallgrass prairie plant species persist in prairie-like conditions found along roadsides in southern and central Ontario (Woodliffe, 2001). Thirteen of these species are on the provincial "rare, threatened, and endangered" list (Argus et al., 1987; Woodliffe, 2001).
- Roadsides contribute in reducing the probability of local species extirpation due to habitat fragmentation. While habitat fragmentation has been shown to lead to species extinction (Wilcox and Murphy, 1985), research has also shown that, given a fragmentation event, the linking of fragments by strips of hospitable habitat allows the maintenance of biodiversity within the fragments (Noss, 1983; Lefkovitch and Fahrig, 1985; Bennett, 1990; Bélanger, 2000).
Roadsides have been shown to act as strips of hospitable habitat linking fragments and ensuring habitat continuity (Huey, 1941; Getz et al., 1978; Lewis, 1991; Andreassen et al., 1996).
Changes to roadside habitats that could result from contamination by road salts could therefore compromise their ecological value.
3.8.1.2 Harmful effects
It is recognized that any environmental change is not necessarily adverse, nor does it necessarily result in harmful effects. Changes within the range of normal variation may not necessarily constitute or result in harm, but more substantive ones may be harmful. The potential for harm resulting from increases in salt concentrations in the environment would depend, among other factors, on the magnitude, duration and frequency of changes.
Effects observed at the population, community or ecosystem level are generally considered more environmentally harmful and are of more concern than those observed only at lower levels of organization. Few studies have directly tested Priority Substances for effects at the population, community or ecosystem level of organization. Most toxicity studies are conducted in the laboratory using relatively small sample sizes relative to population sizes in natural communities. However, many of the effects measured in laboratory and field studies have implications for populations, communities and ecosystems. Effects on processes such as growth, reproduction or survival are closely related to the viability of natural populations and are used as an indication of the potential for environmental harm. While quantification of likely ecosystem impacts is often not feasible, it can be assumed that where ambient concentrations approach those associated with serious effects on individual organisms (such as median lethal concentrations), harmful environmental impact may also be expected.
3.8.1.3 Weight of evidence
CEPA 1999, Section 76.1, provides that, when conducting and interpreting the results of an assessment to determine whether a PSL substance is "toxic or capable of becoming toxic," "the Ministers shall apply a weight of evidence approach and the precautionary principle."
It is impossible to specify a rigid cut-off point where effects are considered sufficient in extent or harm to declare a substance "toxic or capable of becoming toxic." In using a weight of evidence approach, several lines of evidence and endpoints are considered. This includes consideration of several lines of evidence for a given environmental compartment (e.g., surface water assessment considering toxic thresholds for a range of aquatic biota, potential for shifts in freshwater algal populations, changes in physical structure of lakes, exposure resulting from releases of contaminated groundwater through springs, etc.). Ultimate overall conclusions must be based on consideration of the full array of lines of evidence. While some lines of evidence may provide for only tenuous conclusions, others will provide stronger evidence. By using a weight of evidence approach, risk assessment can reduce the biases and uncertainties associated with using only one approach to estimate risk.
3.8.2 Summary of assessment
Road salts are used as de-icing and anti-icing chemicals for winter road maintenance, with some use as summer dust suppressants. Inorganic chloride salts considered in this assessment include sodium chloride, calcium chloride, potassium chloride and magnesium chloride. In the environment, these compounds dissociate into the chloride anion and the corresponding cation. In addition, ferrocyanide salts, which are added as anti-caking agents to some road salt formulations, were assessed. It is estimated that approximately 4.75 million tonnes of sodium chloride were used as road salts in the winter of 1997-98 and that 110 000 tonnes of calcium chloride are used on roadways in a typical year. Very small amounts of other salts are used. Based on these estimates, about 4.9 million tonnes of road salts can be released to the environment in Canada every year, accounting for about 3.0 million tonnes of chloride. The highest annual loadings of road salts on a road-length basis are in Ontario and Quebec, with intermediate loadings in the Atlantic provinces and lowest loadings in the western provinces.
Road salts enter the Canadian environment through their storage and use and through disposal of snow cleared from roadways. Road salts enter surface water, soil and groundwater after snowmelt and are dispersed through the air by splashing and spray from vehicles and as windborne powder. Chloride ions are conservative, moving with water without being retarded or lost. Accordingly, all chloride ions that enter the soil and groundwater can ultimately be expected to reach surface water; it may take from a few years to several decades or more for steady-state groundwater concentrations to be reached. Because of the widespread dispersal of road salts through the environment, environmental concerns can be associated with most environmental compartments.
In water, natural background concentrations of chloride are generally no more than a few milligrams per litre, with some local or regional instances of higher natural salinity, notably in some areas of the Prairies and British Columbia. High concentrations of chloride related to the use of road salts on roadways or releases from patrol yards or snow dumps have been measured. For example, concentrations of chloride over 18 000 mg/L were observed in runoff from roadways. Chloride concentrations up to 82 000 mg/L were also observed in runoff from uncovered blended abrasive/salt piles in a patrol yard. Chloride concentrations in snow cleared from city streets can be quite variable. For example, the average chloride concentrations in snow cleared from streets in Montréal were 3000 mg/L (secondary streets) and 5000 mg/L (primary streets). Waters from patrol yards, roadways or snow dumps can be diluted to various degrees when entering the environment. In the environment, resulting chloride concentrations have been measured as high as 2800 mg/L in groundwater in areas adjacent to storage yards, 4000 mg/L in ponds and wetlands, 4300 mg/L in watercourses, 2000-5000 mg/L in urban impoundment lakes and 150-300 mg/L in rural lakes. While highest concentrations are usually associated with winter or spring thaws, high concentrations can also be measured in the summer, as a result of the travel time of the ions to surface waters and the reduced water flows in the summer. Water bodies most subject to the impacts of road salts are small ponds and watercourses draining large urbanized areas, as well as streams, wetlands or lakes draining major roadways. Field measurements have shown that roadway applications in rural areas can result in increased chloride concentrations in lakes located a few hundred metres from roadways.
The potential for impacts to regional groundwater systems was evaluated using a mass balance technique that provides an indication of potential chloride ion concentrations downgradient from saltable road networks. The mass balance modelling and field measurements indicated that regional-scale groundwater chloride concentrations greater than 250 mg/L will likely result under high-density road networks subject to annual loadings above 20 tonnes sodium chloride per two-lane-kilometre. Considering data on loadings of road salts, urban areas in southern Ontario, southern Quebec and the Atlantic provinces face the greatest risk of regional groundwater impacts. Groundwater will eventually well up into the surface water or emerge as seeps and springs. Research has shown that 10-60% of the salt applied enters shallow subsurface waters and accumulates until steady-state concentrations are attained. Elevated concentrations of chlorides have been detected in groundwater springs emerging to the surface.
Acute toxic effects of chloride on aquatic organisms are usually observed at relatively elevated concentrations. For example, the 4-day median lethal concentration (LC50) for the cladoceran Ceriodaphnia dubia is 1400 mg/L. Exposure to such concentrations may occur in small streams located in heavily populated urban areas with dense road networks and elevated road salt loadings, in ponds and wetlands adjacent to roadways, near poorly managed salt storage depots and at certain snow disposal sites.
Chronic toxicity occurs at lower concentrations. Toxic effects on aquatic biota are associated with exposures to chloride concentrations as low as 870, 990 and 1070 mg/L for median lethal effects (fathead minnow embryos, rainbow trout eggs/embryos and daphnids, respectively). The NOEC for the 33-day early life stage test for survival of fathead minnow was 252 mg chloride/L.
Furthermore, it is estimated that 5% of aquatic species would be affected (median lethal concentration) at chloride concentrations of about 210 mg/L, and 10% of species would be affected at chloride concentrations of about 240 mg/L. Changes in populations or community structure can occur at lower concentrations. Because of differences in the optimal chloride concentrations for the growth and reproduction of different species of algae, shifts in populations in lakes were associated with concentrations of 12-235 mg/L. Increased salt concentrations in lakes can lead to stratification, which retards or prevents the seasonal mixing of waters, thereby affecting the distribution of oxygen and nutrients. Chloride concentrations between 100 and 1000 mg/L or more have been observed in a variety of urban watercourses and lakes. For example, maximum chloride concentrations in water samples from four Toronto-area creeks ranged from 1390 to 4310 mg/L. Chloride concentrations greater than about 230 mg/L, corresponding to those having chronic effects on sensitive organisms, have been reported from these four watercourses through much of the year. Available studies indicate that, in areas of heavy use of road salts, especially southern Ontario, Quebec and the Maritimes, chloride concentrations in groundwater and surface water are frequently at levels likely to affect biota, as demonstrated by laboratory and field studies.
Application of road salts can also result in deleterious effects on the physical and chemical properties of soils, especially in areas that suffer from poor salt, soil and vegetation management. Effects are associated with areas adjacent to salt depots and roadsides, especially in poorly drained depressions. Effects include impacts on soil structure, soil dispersion, soil permeability, soil swelling and crusting, soil electrical conductivity and soil osmotic potential. These can have, in turn, abiotic and biotic impacts on the local environment. The primary abiotic impact is the loss of soil stability during drying and wetting cycles and during periods of high surface runoff and wind. Biological impacts relate primarily to osmotic stress on soil macro- and microflora and macro- and microfauna, as well as salt-induced mobilization of macro- and micronutrients that affect flora and fauna.
A number of field studies have documented damage to vegetation and shifts in plant community structure in areas impacted by road salt runoff and aerial dispersion. Halophytic species, such as cattails and common reed-grass, readily invade areas impacted by salt, leading to changes in occurrence and diversity of salt-sensitive species. Elevated soil levels of sodium and chloride or aerial exposure to sodium and chloride results in reductions in flowering and fruiting of sensitive plant species; foliar, shoot and root injury; growth reductions; and reductions in seedling establishment. Sensitive terrestrial plants may be affected by soil concentrations greater than about 68 mg sodium/kg and 215 mg chloride/kg. Areas with such soil concentrations extend linearly along roads and highways or other areas where road salts are applied for de-icing or dust control. The impact of aerial dispersion extends up to 200 m from the edge of multi-lane highways and 35 m from two-lane highways where de-icing salts are used. Salt injury to vegetation also occurs along watercourses that drain roadways and salt handling facilities.
Behavioural and toxicological impacts have been associated with exposure of mammalian and avian wildlife to road salts. Ingestion of road salts increases the vulnerability of birds to car strikes. Furthermore, intake calculations suggest that road salts may poison some birds, especially when water is not freely available during severe winters. Road salts may also affect wildlife habitat, with reduction in plant cover or shifts in communities that could affect wildlife dependent on these plants for food or shelter. Available data suggest that the severity of road kills of federally protected migratory bird species (e.g., cardueline finches) and the contribution of road salts to this mortality have been underestimated.
Ferrocyanides are very persistent but are of low toxicity. However, in solution and in the presence of light, they can dissociate and form cyanide. In turn, the cyanide ion may volatilize and dissipate fairly quickly. The ultimate effects of ferrocyanides therefore depend on the complex balance between photolysis and volatilization, which in turn depends on environmental factors. Modelling studies undertaken in support of this assessment indicate that there is a potential for certain aquatic organisms to be adversely affected by cyanide in areas of high use of road salts.
3.8.3 Uncertainties
There are a number of uncertainties and assumptions inherent in the environmental risk characterization of road salts presented in this report. These uncertainties can typically be associated with knowledge and data gaps in laboratory and field data that were used to characterize the use of road salts in Canada and assess the impact of road salts on different environmental endpoints. In addition to discussions of uncertainties in previous sections, the following paragraphs present further discussions of uncertainties pertinent to the conclusions for this assessment.
A concerted effort was made to obtain data on road salt loadings from the most accurate sources. Data were also analysed to determine if the base year used to characterize road salt loadings was abnormal. Results of this analysis indicate that the 1997-98 winter season was not characterized by elevated loadings (in comparison with the 5-year window for which data were collected). One limitation, however, is that data are characterized by provincial maintenance district. While these estimates are reliable, it is uncertain how the use of road salts is distributed within a maintenance district. It was assumed that road salts were used equally throughout the maintenance districts. Certain roads, however, are subject to higher loadings, and the density of roads will vary within a maintenance district. Estimates were made to account for municipalities where data were not available and for the amount of salt that would be used for commercial purposes (e.g., in parking lots). Estimates were not made to account for the volume of salt that would be used by individuals or other agencies, such as transit or port authorities.
Most of the data presented in the Assessment Report relate to the use of road salts on roadways. Road salts are also used in parking lots and other commercial and industrial properties. Data on chloride concentrations in runoff from such properties and in receiving waters were not presented in this report; however, there is a potential for receiving waters to be impacted by such releases.
The number of active patrol yards in Canada is uncertain. Information from provincial and territorial agencies indicates that they manage approximately 1300 patrol yards. Considering that other levels of government (e.g., counties and municipalities) and private sector agencies also maintain roadways, it is probable that this number considerably underestimates the number of patrol yards in Canada. Another uncertainty is the amount of salt that may be stored at patrol yards. Data and estimates of the amount of salt that may be stored at patrol yards were presented, but these are rough estimates. For example, it was estimated that the amount of salt stored at patrol yards ranges between 45 and 21 000 tonnes. Other uncertainties relate to the type of structures used to store salts, the number of agencies that store blended abrasive/salt piles outside, the percentage of salt in blended piles, the discharge of washwater and the range of practices used to reduce the impact of road salt storage on the environment.
Another uncertainty relates to the relevance of the use of exposure data obtained from engineered environments such as ditches, rights-of-ways and stormwater retention ponds to estimate possible environmental effects. Modified environments can be ecologically important. In areas heavily impacted by urbanization, industrialization or agriculture, modified landscapes such as rights-of-ways or impoundments can in fact offer the only habitats available for many species locally or regionally, including sensitive species as well as protected organisms such as migratory birds. As such, consideration of potential impacts in these modified environments may be justifiable. In some cases, concentrations in engineered environments can be used as surrogates for concentrations that may be found in adjacent areas. Thus, concentrations in ditches can be representative of environmental concentrations likely to occur in streams or wetlands immediately adjacent to roadways, and concentrations in groundwater below storage facilities may be indicative of groundwater concentrations immediately off-site. In yet other instances, it may be more appropriate to consider data from engineered environments as an estimate of concentrations that may be released to the environment following little or extensive dilution.
There is also a certain degree of uncertainty regarding the models used to estimate the impact of road salts on soils, surface water and groundwater resources. At the outset, any assumptions made when characterizing road salt loadings (e.g., equal use over a maintenance district) were carried forward to these models. Furthermore, these models were designed to be as simple as possible to derive reasonable approximate estimates of the spatial effects of road salts on the endpoints at a national scale. Because of the numerous and often confounding factors at play in the environment, it was not possible to develop one model that would provide site-specific results. The calculations and procedures used in these models are based on regional-scale assumptions. If the different parameters included in these models were measured on a site-specific basis, more accurate estimates of the impacts of road salts on environmental endpoints could be modelled. Accordingly, the estimates generated from these models should be viewed as characteristic of the range of impacts that can be expected at a national scale.
Some of the exposure data presented in the Assessment Report were collected as part of routine water quality monitoring; samples were not necessarily collected within a framework designed to assess the impact of road salts on water quality. Studies like the one by Whitfield and Wade (1992) demonstrate the importance of continuous monitoring to characterize the temporal impact of road salts on watercourses. The temporal aspect of these concentrations is important to determine if organisms are exposed to concentrations that cause acute or chronic effects. Continuous monitoring data were seldom available; however, several years of monitoring data were used to characterize concentrations that would be typical for certain locations at different times. A few studies with continuous or extended sampling were available that permitted corroboration of other studies.
Without constant monitoring data, it is difficult to determine if the grab samples are representative of peak concentrations or are more characteristic of ambient concentrations that are typical of certain time periods. Recent unpublished monitoring data for six watercourses in the Toronto area from September 2000 to April 2001 suggest that elevated chloride concentrations can be observed throughout winter months. For example, samples taken from October 30 to the end of April for Etobicoke Creek, Mimico Creek, Humber River, Don River, Highland Creek and Rouge River had chloride concentrations that ranged between 448 and 3771, 419 and 5376, 264 and 2110, 87 and 2247, 51 and 1755 and 101 and 434 mg/L, respectively (City of Toronto, 2001). These data generally concur with other data that were used to characterize chloride concentrations in watercourses in the Greater Toronto Area. Furthermore, this information (and monitoring data presented previously) seems to suggest that aquatic organisms in these watercourses would be exposed to elevated chloride concentrations throughout winter and spring. Other work in New Brunswick indicated maximum concentrations in summer.
There is also a certain degree of uncertainty regarding the species that may be impacted by the use of road salts. The sections on aquatic ecosystems, vegetation, wildlife and ferrocyanides compared toxicity values with exposure concentrations that are characteristic of the same region. Within a region, however, the distribution of these species and the corresponding exposure concentrations may vary. While there is a certain degree of uncertainty, case studies were used to reduce the degree of uncertainty and highlight the type of impact that can be expected.
Case studies, which document the impact of road salts on environmental endpoints, are presented in this Assessment Report and supporting documentation. The review of case studies should not be viewed as exhaustive. The number of case studies presented likely underestimates the number of instances where concentrations of concern for salts have been exceeded. Case studies in this report have generally been brought to the forefront because the taste of groundwater-based drinking water supplies has been affected as a result of an elevation in salt concentrations from contamination by road salts. It is expected that contamination in areas away from drinking water sources would frequently not be detected or reported.
It is not possible to predict the impact of potential climate change on the net use of road salts in the future, notably as this relates to the frequency and extent of precipitation events at air temperatures of about -10°C to 0°C. However, it can be expected that any yearly increase in evapotranspiration or decrease in precipitation would result in an increase in concentrations of chloride and other solutes in surface water and shallow groundwater. Field evidence in the Experimental Lakes Area in western Ontario indicated that an increase in average air temperature from 14°C to 16°C caused an increase in evaporation of 30% (Schindler et al., 1996). Despite predicted increases in precipitation in parts of the Great Lakes basin, greatly increased evapotranspiration in the catchment of the Great Lakes is expected, resulting in reductions in wetlands (Mortsch and Quinn, 1996) and a corresponding increase in concentrations of solutes.
3.8.4 Conclusions
Almost 5 million tonnes of road salts that contain inorganic chloride salts with or without ferrocyanides are used every year in Canada. All of these salts are ultimately found in the environment, whether as a result of dispersive use on roadways or through losses from patrol yards and snow dumps. The report has identified situations where the resulting environmental concentrations approach or exceed those associated with harmful effects on physical properties of soils or water bodies or on organisms associated with freshwater and terrestrial habitats at numerous sites across Canada.
Local and regional contamination of groundwater can lead, sometimes after several years or decades, to high concentrations of chloride, including concentrations that exceed acute and chronic toxicity values for organisms that may occur in springs.
For surface waters associated with roadways or storage facilities, episodes of salinity have been reported during the winter and spring in some urban watercourses in the range associated with acute toxicity in laboratory experiments. Reports of lower levels of exposure ranging from near those associated with chronic toxicity in laboratory experiments to those modestly above background salinity levels are more frequent and widespread. Some measurable changes in aquatic biota and/or communities have been found in laboratory experiments and in field studies at these lower concentrations, including possible impacts on lake stratification.
Increased salt loadings into soils can lead to modification of soil properties critical for soil health. A number of field studies have documented damage to vegetation and shifts in plant community structure in areas impacted by road salt runoff and aerial dispersion. Elevated soil levels of sodium and chloride or aerial exposure to sodium and chloride have been recorded frequently at levels impacting growth, reproduction and survival of sensitive plant species along roadways and watercourses that drain roadways and salt handling facilities.
Behavioural and toxicological impacts have been associated with exposure of mammalian and avian wildlife to road salts. Road salts may also affect wildlife habitat, with reduction in plant cover or shifts in communities that could affect wildlife dependent on these plants for food or shelter.
Since ferrocyanides can dissociate in the environment to form cyanide, there is a potential for certain aquatic organisms to be adversely affected by cyanide in areas of high use of road salts.
While the ecological significance of many of the potential impacts outlined above cannot be quantified, it is clear from available information that there is a reasonable probability that road salts that contain inorganic chloride salts with or without ferrocyanide salts may be having an immediate or long-term harmful effect on some Canadian surface water organisms, terrestrial vegetation and wildlife and may also constitute a danger to the environment on which life depends through its impacts on aquatic systems and soils and terrestrial habitats. Thus, road salts that contain inorganic chloride salts with or without ferrocyanide salts should be considered "toxic" under CEPA 1999 because of tangible threats of serious or irreversible environmental damage.
CEPA 1999 64(a): Based on the available data, it is considered that road salts that contain inorganic chloride salts with or without ferrocyanide salts are entering the environment in a quantity or concentration or under conditions that have or may have an immediate or long-term harmful effect on the environment or its biological diversity. Therefore, it is concluded that road salts that contain inorganic chloride salts with or without ferrocyanide salts are "toxic" as defined in Paragraph 64(a) of CEPA 1999.
CEPA 1999 64(b): Based on the available data, it is considered that road salts that contain inorganic chloride salts with or without ferrocyanide salts are entering the environment in a quantity or concentration or under conditions that constitute or may constitute a danger to the environment on which life depends. Therefore, it is concluded that road salts that contain inorganic chloride salts with or without ferrocyanide salts are "toxic" as defined in Paragraph 64(b) of CEPA 1999.
Overall conclusion: Based on critical assessment of relevant information, road salts that contain inorganic chloride salts with or without ferrocyanide salts are considered to be "toxic" as defined in Section 64 of CEPA 1999.
3.9 Considerations for follow-up (further action)
The use of de-icing agents is an important component of strategies to keep roadways open and safe during the winter and minimize traffic crashes, injuries and mortality under icy and snowy conditions. These benefits were recognized by the Ministers' Expert Advisory Panel on the Second Priority Substances List, even as they recommended that this assessment of potential impacts on the environment be conducted. Any measures developed as a result of this assessment must never compromise human safety; selection of options must be based on optimization of winter road maintenance practices so as to not jeopardize road safety, while minimizing the potential for harm to the environment. Any action taken to reduce impacts on the environment is likely also to reduce potential for contamination of groundwater-based drinking water supplies, which is clearly desirable.
Future management should focus on key sources in areas where the assessment has indicated concerns. These relate to the following:
- Patrol yards: Key concerns relate to contamination of groundwater at patrol yards and the discharge to surface water. In addition, overland flow of salty snowmelt waters can result in direct impacts on surface water and near-field vegetation. Based on surveys and reviews, salt losses from patrol yards are associated with loss at storage piles (which include salt piles as well as piles of sand and gravel to which salts have been added) and during the handling of salts, relating to both storage and loading and unloading of trucks. The discharge of patrol yard washwater is also a potential source of release of salts. Measures and practices should therefore be considered to ensure storage of salts and abrasives to reduce losses through weathering, to reduce losses during transfers and to minimize releases of stormwater and equipment washwater.
- Roadway application: Key environmental concerns have been associated with areas of high salt use and high road density. Regions of southern Ontario and Quebec and the Atlantic provinces have the highest rate of salt use on an area basis and as such have the highest potential for contamination of soils, groundwater and surface water by road salts as a result of roadway applications. In addition, urban areas in other parts of the country where large amounts of salts are applied are of potential concern, especially for streams and aquifers that are wholly surrounded by urban areas. In rural areas, surface waters receiving drainage from roadways may also be susceptible to contamination. Areas where splash or spray from salted roads can be transported through air to sensitive vegetation are a potential concern. Wetlands that directly adjoin roadway ditches and that receive runoff in the form of salty snowmelt waters are also potential management concerns. Therefore, measures should be considered to reduce the overall use of chloride salts in such areas. The selection of alternative products or of appropriate practices of technology to reduce salt use should be considered while ensuring maintenance of roadway safety.
- Snow disposal: Key environmental concerns relate to eventual loss of meltwater into surface water and into soil and groundwater at snow disposal sites. Measures to minimize percolation of salty snowmelt waters into soil and groundwater at snow disposal sites should be considered. Practices to direct the release of salty snowmelt waters into surface waters with minimal environmental sensitivity or into storm sewers could be considered. Measures should also be considered to ensure sufficient dilution before release.
- Ferrocyanides: This assessment indicates that there is a possible adverse exposure for the more sensitive aquatic vertebrates in areas of very high use of road salts. Risks could be reduced by reducing total salt use or reducing content of ferrocyanides in road salt formulations. To reduce the possibility of exposure, producers of road salts could consider reducing the addition rate of ferrocyanide to road salt. Any reduction in total salt use would be expected to result in an equivalent reduction in release of ferrocyanides.
4.0 References
Acton D.F. and L.J. Gregorich (eds). 1995. The Health of Our Soils. Toward sustainable agriculture in Canada. Centre for Land and Biological Resources Research. Research Branch, Agriculture and Agri-Food Canada. Publication 1906/E.
Adams, L.W. and A.D. Geiss. 1983. Effects of roads on small mammals. J. Appl. Ecol. 20: 403-415.
Adelman, I.R., L.L.J. Smith and G.D. Siesennop. 1976. Acute toxicity of sodium chloride, pentachlorophenol, guthion and hexavalent chromium to fathead minnows (Pimephales promelas) and goldfish (Carassius auratus). J. Fish. Res. Board Can. 33: 203-208.
AGRA. 1995. Phase 1 of groundwater flow and contaminant assessment, Heffley Creek, British Columbia. Report prepared by AGRA Earth and Environmental for the British Columbia Ministry of Transportation and Highways, Kamloops, British Columbia (Project No. KX11322).
AGRA. 1999. Chloride plume monitoring -October 1998 and February 1999, Heffley Creek, British Columbia. Report prepared by AGRA Earth and Environmental for the British Columbia Ministry of Transportation and Highways, Kamloops, British Columbia (Project No. KX11538).
Alberta Infrastructure. 2000. Draft Phase 3 environmental site assessments for the Worsley Satellite Yard, Obed Satellite Yard and 25th Baseline Satellite Yard. Report prepared by Green Plan Limited for Alberta Infrastructure, Civil Projects Branch, Edmonton, Alberta.
Ali, O.M., M. Yousaf and J.D. Rhoades. 1987. Effect of exchangeable cation and electrolyte concentration on mineralogy of clay dispersed from aggregates. Soil Sci. Soc. Am. J. 51: 896-900.
Allen, J. and G.A. Strobel. 1966. The assimilation of H14CN by a variety of fungi. Can. J. Microbiol. 12: 414-416 [cited in Towill et al., 1978].
American Cyanamid Company. 1953. The chemistry of the ferrocyanides. The Beacon Press, Inc., New York, N.Y.
Anderson, B.G. 1948. The apparent thresholds of toxicity to Daphnia magna for chlorides of various metals when added to Lake Erie water. Am. Fish. Soc. 78: 96-113.
Andreassen, H.P., R.A. Ims and O.K. Steinset. 1996. Discontinuous habitat corridors: effects on male root vole movements. J. Appl. Ecol. 33: 555-560.
Anon. 1992. Dryland salinity investigations procedures manual. Conservation and Development Branch, Alberta Agriculture, and Prairie Farm Rehabilitation Administration, Agriculture and Agri-Food Canada. 110 pp.
Arambasic, M.B., S. Bjelic and G. Subakov. 1995.
Acute toxicity of heavy metals (copper, lead, zinc), phenol and sodium on Allium cepa L., Lepidium sativum L. and Daphnia magna St.: comparative investigations and the practical applications. Water Res. 29: 497-503.
Argus, G.W., K.M. Pryer, D.J. White and C.J. Keddy (eds.). 1987. Atlas of the rare vascular plants of Ontario, 1982-1987. National Museum of Natural Sciences, Ottawa, Ontario.
Arp, P.A. 2001. Electrical conductivity and chloride contents of stream, pond and ditchwater locations near a salt depot: a case study. Faculty of Forestry and Environmental Management, University of New Brunswick, Fredericton, New Brunswick. 14 pp.
Arsenault, R.A., T.L. Pollock, S. Ratnam and D. Hansen. 1985. Surface water quality study in the Waterford River Basin. Urban Hydrology Study of the Waterford River Basin. 91 pp. (Technical Report No. UHS-WRB1.4).
Auestad, I., A. Norderhaug and I. Auestad. 1999. Road verges - Species rich habitats. Aspects Appl. Biol. 54: 269-275.
August, L.L. and T.A. Graupensperger. 1989. Impacts of highway deicing programs on groundwater and surface water in Maryland. In: Groundwater issues and solutions in the Potomac River Basin. Chesapeake Bay Region. National Water Well Association Conference, May 14-16, 1989, Washington, D.C. pp. 37-394.
Baeten, L.A. and F.J. Dein. 1996. Salt toxicosis in wild birds. Am. Assoc. Zoo Vet. Annu. Proc. 1996: 296-297.
Baker, K.D. 1965. An observation of bird mortality on highways. Blue Jay 23(2): 79-80.
Bakowsky, W. and J.L. Riley. 1994. A survey of the prairies and savannas of southern Ontario. In: R.G. Wickett, P.D. Lewis, A. Woodliffe and P. Pratt (eds.), Proceedings of the 13th North American Prairie Conference, 1992. Department of Parks and Recreation, Windsor, Ontario.
Ballantyne, B. 1987. Clinical and experimental toxicology of cyanides. In: B. Ballantyne and T.C. Marrs (eds.), Clinical and experimental toxicology of cyanides. John Wright, Bristol, U.K. pp. 41-126.
Barica, J. 1978. Variability in ionic composition and phytoplankton biomass of saline eutrophic lakes within a small geographic area. Arch. Hydrobiol. 81(3): 304-326.
Barker, A.V. 2000. Search of literature and planning of research on salt damage along highways in Massachusetts. Massachusetts Highway Department and University of Massachusetts Cooperative Transportation Research Program. 44 pp. (Research Quarterly Report).
Barrick, W.E. and H. Davidson. 1980. Deicing salt spray injury in Norway maple as influenced by temperature and humidity treatments. HortScience 15(2): 203-205.
BC MoTH (British Columbia Ministry of Transportation and Highways). 1993. Groundwater contamination study: Heffley Creek Pit, Thompson-Okanagan Region. Report prepared by R. Buchanan, Geotechnical and Materials Engineering, Victoria, British Columbia.
Beak. 1999. Ecotoxicity test results. Unpublished data for M.S. Evans. Beak International Inc., Brampton, Ontario.
Beck, P., S. Kok and W.J. Snodgrass. 1994. Technology assessment - removal of total dissolved solids from salty washwaters generated from vehicle washing operations. Report prepared for Ontario Ministry of Transportation, Toronto, Ontario.
Beckerson, D.W., N.P. Cain, G. Hofstra, D.P. Ormrod and P.A. Campbell. 1980. A guide to: plant sensitivity to environmental stress. Landscape Archit. May: 299-303.
Bedunah, D. and M.J. Trlica. 1979. Sodium chloride effects on carbon dioxide exchange rates and other plant and soil variables of ponderosa pine. Can. J. For. Res. 9(3): 349-353.
Beeton, A.M. 1965. Eutrophication of the St. Lawrence Great Lakes. Limnol. Oceanogr. 10: 240-254.
Beeton, A.M. 1969. Changes in the environment and the biota of the Great Lakes. In: Eutrophication: Causes, consequences, correctives. Printing and Publishing Office, National Academy of Sciences, Washington, D.C. pp. 150-187.
Bélanger, L. 2000. Personal communication. Section Faune et Habitats, Environnement Canada, Sainte-Foy, Québec.
Bellamy, P.E., R.F. Shore, D. Ardeshir, J.R. Treweek and T.H. Sparks. 2000. Road verges as habitat for small mammals in Britain. Mammal Rev. 30(2): 131-139.
Belluck, D.A. 2000. Personal communication. Minnesota Department of Transport, St. Paul, Minnesota.
Benkman, C.W.1998. [Crossbills hit by vehicles while eating salt]. Personal communication from C.W. Benkman (Dept. of Biology. New Mexico State University, Las Cruces, New Mexico) to Lorna Brownlee (Canadian Wildlife Service, National Wildlife Research Centre, Ottawa). Email dated 18 February 1998.
Benkman, C.W. 2000. [The scavenging of finches killed at roadside in Algonquin Park] Personal communication from Craig W. Benkman (Dept. of Biology. New Mexico State University, Las Cruces, New Mexico) to P. Mineau (Canadian Wildlife Service, National Wildlife Research Centre, Ottawa). Email dated 11 May 2000.
Bennett, A.F. 1990. Habitat corridors and the conservation of small mammals in a fragmented forest environment. Landscape Ecol. 4: 109-122.
Bennett, A.F. 1991. Roads, roadsides and wildlife conservation: a review. In: D.A. Saunders and R.J. Hobbs (eds.), Nature conservation 2: The role of corridors. Surrey Beatty and Sons, Victoria, Australia. pp. 99-118.
Bennetts, R.E. and R.L. Hutto. 1985. Attraction of social fringillids to mineral salts: An experimental study. J. Field Ornithol. 56(2): 187-189.
Bertram, B.M. 1997. Sodium chloride. In: J.I. Kroschwitz (exec. ed.), M. Howe-Grant (ed.), Kirk-Othmer encyclopedia of chemical technology. 4th edition. Vol. 22. John Wiley & Sons, New York, N.Y. p. 371.
Bilodeau, S. 1999. La fondeuse à neige de Cap-Rouge, un an après son démarrage. Vecteur Environnement 32(2): 70-73.
Birge, W.J., J.A. Black, A.G. Westerman, T.M. Short, S.B. Taylor, D.M. Bruser and E.D. Wallingford. 1985. Recommendations on numerical values for regulating iron and chloride concentrations for the purpose of protecting warmwater species of aquatic life in the Commonwealth of Kentucky. Kentucky Natural Resources and Environmental Protection Cabinet, Lexington, Kentucky (Memorandum of Agreement No. 5429).
Bishop, C.A., J. Struger, L.J. Shirose, L. Dunn and G.D. Campbell. 2000. Contamination and wildlife communities in stormwater detention ponds in Guelph and the Greater Toronto Area, Ontario, 1997 and 1998. Part 2 - Contamination and biological effects of contamination. Water Qual. Res. J. Can. 35(3): 437-474.
Blinn, D.W., M. Hurley and L. Brokaw. 1981. The effects of saline seeps and restricted light upon the seasonal dynamics of phytoplankton communities within a southwestern (USA) desert canyon stream. Archiv. Hydrobiol. 92: 287-305.
Boucher, R.J. 1982. Penjajawoc Stream Salt Study. Materials and Research Division, Maine Department of Transportation. 15 pp. (Technical Paper 82-85).
Bourbonniere, R.A. 1985. Unpublished data. National Water Research Institute, Burlington, Ontario.
Bourbonniere, R.A. 1998. Unpublished data. National Water Research Institute, Burlington, Ontario.
Bowen, G.S. and M.J. Hinton. 1998. The temporal and spatial impacts of road salt on streams draining the Greater Toronto Area. In: Groundwater in a watershed context: proceedings. Ontario Ministry of the Environment and Geological Survey of Canada, Burlington, Ontario. pp. 303-309.
Bowser, C.J. 1992. Groundwater pathways for chloride pollution of lakes. In: F.M. D'Itri (ed.), Chemical deicers and the environment. Lewis Publishers, Boca Raton, Florida. pp. 283-301.
Bradford, W. 1988. A plan to reduce wildlife mortality on transportation corridors in Jasper National Park. Unpublished management plan. Natural Resources Conservation, Jasper National Park, Jasper, Alberta.
Bresler, E., B.L. McNeil and D.L. Carter. 1982. Saline and sodic soils. Advanced Series in Agriculture Sciences 10. Springer-Verlag, Berlin. 236 pp.
Briggins, D., J. Walsh, C.L. Lin, K.A. Macdermid and R.H. Johnson (eds.). 1989. The environmental implications of road salting in Nova Scotia. Joint release of the Nova Scotia Department of the Environment and the Nova Scotia Department of Transportation and Communication. 40 pp.
Broderius, S.J. 1973. Determination of molecular hydrocyanic acid in water and studies of the chemistry and toxicity to fish of metal cyanide complexes. Ph.D. thesis, Oregon State University, Corvallis, Oregon [cited in Doudoroff, 1976; U.S. EPA 1985].
Broderius, S.J. and L.L. Smith, Jr. 1980. Direct photolysis of hexacyanoferrate complexes: proposed applications to the aquatic environment. Environmental Research Laboratory, U.S. Environmental Protection Agency, Duluth, Minnesota (Technical Report EPA-600/3-80-003).
Brownlee, L., P. Mineau and A. Baril. 2000. Risk characterization for wildlife. Report submitted to the Environment Canada CEPA Priority Substances List Environmental Resource Group on Road Salts, May 2000. Commercial Chemicals Evaluation Branch, Environment Canada, Hull, Quebec.
Bubeck, R.C., W.H. Diment, B.L. Deck, A.L. Baldwin and S.D. Lipton. 1971. Runoff of deicing salt: Effect on Irondequoit Bay, Rochester, New York. Science 172: 1128-1132.
Bubeck, R.C., W.W. Staubitz, A.D. Weidemann and L.P. Spittal. 1995. Physical and chemical limnology of Ides Cove near Rochester, New York, 1970-1982. U.S. Geological Survey, Ithaca, New York. 44 pp. (Water-Resources Investigations Report 92-4052).
Butler, B.J. and J. Addison. 2000. Biological effects of road salts on soils. Report submitted to the Environment Canada CEPA Priority Substances List Environmental Resource Group on Road Salts, March 2000. Commercial Chemicals Evaluation Branch, Environment Canada, Hull, Quebec.
Button, E.F. and D.E. Peaslee. 1966. The effect of rock salt upon roadside sugar maples in Connecticut. Highw. Res. Rec. 161: 121- 131.
Cain, N.P., B. Hale, E. Berkelaar and D. Morin. 2001. Critical review of effects of NaCl and other road salts on terrestrial vegetation in Canada. Report submitted to the Environment Canada CEPA Priority Substances List Environmental Resource Group on Road Salts. July 2001. Existing Substances Branch, Environment Canada, Hull, Quebec.
Camp, M. and L.B. Best. 1993a. Nest density and nesting success of birds in roadsides adjacent to rowcrop fields. Am. Midl. Nat. 131: 347-358.
Camp, M. and L.B. Best. 1993b. Bird abundance and species richness in roadsides adjacent to Iowa rowcrop fields. Wildl. Soc. Bull. 21: 315-325.
Canadian Salt Company Limited. 1991. Windsor salt, Safe-T-Coarse - Highway. Point Claire, Quebec (PDS VIII-3 5/91).
Catling, P.M. and S.M. McKay. 1980. Halophytic plants in southern Ontario. Can. Field-Nat. 94(3): 248-258.
CCME (Canadian Council of Ministers of the Environment). 1991. Canadian water quality guidelines. Environment Canada, Ottawa, Ontario.
CCME (Canadian Council of Ministers of the Environment). 1998. Guidelines for Canadian drinking water quality. Environment Canada, Ottawa, Ontario.
CFI (Canadian Fertilizer Institute). 1997. [Regarding report on potassium chloride in potash mine tailings used for salting roads]. Personal communication from J. Farrell (Manager, Technical Affairs, CFI) to R. Chenier (Chemicals Evaluation Division, Environment Canada, Hull). Letter dated April 18, 1997.
Chang, N.-Y., W. Pearson, J.I.J. Chang, A. Gross, M. Meyer, M. Jolly, B. Vang and H. Samour. 1994. Environmentally sensitive sanding and deicing practices. Colorado Transportation Institute and Colorado Department of Transportation, Denver, Colorado. 116 pp.
Chatwin, T.D. 1989. Cyanide attenuation/ degradation in soil (final report). Resource Recovery and Conservation Company (R2C2), Salt Lake City, Utah.
Chatwin, T.D. 1990. Cyanide attenuation/ degradation in soil (Appendix). Resource Recovery and Conservation Company (R2C2), Salt Lake City, Utah.
Cheminfo. 1999. Socio-economic background and options study on the Canadian salt industry. Report prepared by Cheminfo, Toronto, Ontario, for Regulatory and Assessment Branch, Environment Canada, Hull, Quebec.
Chenevier, C. 1997. Synthèse des données physico-chimiques sur les neiges usées au Québec et leurs impacts environnementaux. M.Sc.A., Département des génies civile, géologique et des mines, École Polytechnique de Montréal, Montréal, Québec.
Cherkauer, D.S. and N.A. Ostenso. 1976. The effect of salt on small artificial lakes. Water Res. Bull. 12(6): 1259-1266.
Chetelat, J., F.R. Pick, A. Morin and P.B. Hamilton. 1999. Periphyton biomass and community composition in rivers of different nutrient status. Can. J. Fish. Aquat. Sci. 56: 560-569.
Chong, C. and G.P. Lumis. 1990. Reduction of salt build-up and twig injury in roadside peach trees with film-forming sprays. Transp. Res. Rec. 1279: 45-53.
CIS (Camford Information Services). 1994. Sodium chloride. CPI Product Profiles. Don Mills, Ontario. 3 pp.
CIS (Camford Information Services). 1996.
Calcium chloride. CPI Product Profiles. Don Mills, Ontario. 3 pp.
City of Montréal. 1998. Actualisation et complément d'information relatifs au programme d'assainissement des lieux d'élimination de neige de la Ville de Montréal. Montréal, Quebec. 28 pp.
City of Regina. 1997. Soil investigation at city snow dump sites 1996-1997. City of Regina Municipal Engineering Department, Regina, Saskatchewan. 11 pp. + appendices.
City of Toronto. 2001. [Unpublished monitoring data for Toronto area watercourses]. Personal communication from G. Gris (Works and Emergency Services, City of Toronto) to D. Morin (Chemicals Evaluation Division, Environment Canada). April and May 2001.
Clark, M.J.R., H. Hanssen, G. Van Aggelen and S. Horvath. 1984. Acute toxicity of iron cyanide species to rainbow trout and to Daphnia magna under exposure to different light intensities. In: Proceedings of the 11th Annual Aquatic Toxicity Workshop. British Columbia Ministry of the Environment, Vancouver, British Columbia [cited in Higgs, 1992].
Collins, B. 2000. Comparison of salt use among years. Memorandum dated November 3, 2000. Migratory Bird Populations Division, National Wildlife Research Centre, Environment Canada.
Conn, E.E. 1980. Cyanogenic compounds. Annu. Rev. Plant Physiol. 31: 433 [cited in Knowles and Bunch, 1986].
Cordukes, A.J. and W.E. Maclean. 1973. Tolerance of some turfgrass species to different concentrations of salt in soils. Can. J. Plant Sci. 53: 69-73.
Cornell Laboratory of Ornithology. 2000a. Biennial irruptions of pine siskins across North America. http://birdsource.cornell.edu/results/irruptions.htm
Cornell Laboratory of Ornithology. 2000b. Population trends in evening grosbeak. http://birdsource.cornell.edu/features/evegro
Couture, P. 1997. La gestion des neiges usées : Dilemmes et contradictions. Vecteur Environnement 30(6): 18-20.
Cowgill, U.M. and D.P. Milazzo. 1990. The sensitivity of two cladocerans to water quality variables: Salinity and hardness. Arch. Hydrobiol. 120(2): 185-196.
Crowther, R.A. and H.B.N. Hynes. 1977. The effect of road deicing salt on the drift of stream benthos. Environ. Pollut. 14: 113-126.
Cushman, J.R., V.A. Duff, G.H. Buteau, L.B. Aust, N. Caldwell and W. Lazer. 1991. Evaluation of calcium magnesium acetate and road salt for contact hypersensitivity potential and dermal irritancy in humans. Contact Dermatitis 24: 289-292.
Davis, G., S. Krannitz and M. Goldstein. 1992. Phase II: A reconnaissance study of roadside tree injury and decline at 17 sites in interior British Columbia for the Roadside Tree Injury Committee. British Columbia Ministry of Environment, Lands and Parks, Victoria, British Columbia. 98 pp. plus appendices.
Dawson, W.R., V.H. Shoemaker, H.B. Tordoff and A. Borut. 1965. Observations on metabolism of sodium chloride in the red crossbill. Auk 82: 606-623.
Delisle, C.E. 1994. La gestion des neiges usées. Routes et transports 24(4): 20-27.
Delisle, C.E. and L. Dériger. 2000. Caracterisation et élimination des neiges usées : impacts sur l'environnement. Report submitted to the Environment Canada CEPA Priority Substances List Environmental Resource Group on Road Salts. Commercial Chemicals Evaluation Branch, Environment Canada, Hull, Quebec.
Delisle, C.E. and A. Leduc. 1987. Évolution dans le temps et dans l'espace de la qualité de la neige usée et de l'eau de ruissellement de pluie du territoire de la Ville de Montréal. Centre de développement technologique de l'École Polytechnique de Montréal. Final report. 155 pp. + annexes.
Delisle, C.E., J.-F. Paradis and P. André. 1995. The Montréal, Quebec experience in used snow disposal and treatment. In: Ecological reclamation in Canada at century's turn. Proceedings of the 35th Annual Meeting of the Canadian Society of Environmental Biologists. Canadian Plains Research Center, Regina, Saskatchewan. pp. 10-18.
Delisle, C.E., C. Chenevier and P. André. 1997. Synthèse des données physico-chimiques sur les neiges usées au Québec et leurs répercussions environnementales. Vecteur Environnement 30(6): 33-44.
Delisle, R. 1999. Autoroute 40 - Cap-de-la-Madeleine : Étude de la contamination de la nappe aquifère par les sels déglacants. Ministère des transports du Québec (Projet # 20-6373-9501).
Demers, C.L. 1992. Effects of road deicing salt on aquatic invertebrates in four Adirondack streams. In: F.M. D'Itri (ed.), Chemical deicers and the environment. Lewis Publishers, Boca Raton, Florida. pp. 245-252.
Demers, C.L. and R.W. Sage, Jr. 1990. Effects of road deicing salt on chloride levels in four Adirondack streams. Water Air Soil Pollut. 49: 369-373.
Dickman, M.D. and M.B. Gochnauer. 1978. Impact of sodium chloride on the microbiota of a small stream. Environ. Pollut. 17: 109-126.
Dirr, M.A. 1975. Effects of salts and application methods on English ivy. HortScience 10(2): 182-184.
Dirr, M.A. 1976. Selection of trees for tolerance to salt injury. J. Arboric. 2(11): 209-216.
Dirr, M.A. 1978. Tolerance of seven woody ornamentals to soil-applied sodium chloride. J. Arboric. 4(7): 162-165.
Dixit, S.S., J.P. Smol, D.F. Charles, R.M. Hughes, S.G. Paulsen and G.B. Collins. 1999. Assessing water quality changes in the lakes of the northeastern United States using sediment diatoms. Can. J. Fish. Aquat. Sci. 56: 131-152.
Dkhili, M. and B. Anderson. 1992. Salt effects on seedling growth of switchgrass and big bluestem. In: D. Smith and C.C. Jacobs (eds.), Proceedings of the 12th North American Prairie Conference 1990. University of Northern Iowa, Cedar Falls, Iowa. pp. 13-16.
Dobson, H.H. 1967. Principal ions and dissolved oxygen in Lake Ontario. In: Proceedings of the 10th Conference on Great Lakes Research. International Association for Great Lakes Research, Ann Arbor, Michigan. Braun-Brumfield, Inc., Ann Arbor, Michigan. pp. 337-356.
Dobson, M.C. 1991. De-icing salt damage to trees and shrubs. Forestry Commission, Surrey, British Columbia. 64 pp. (Bulletin 101).
Doudoroff, P. 1966. Some experiments on the toxicity of complex cyanides to fish. Sewage Ind. Wastes 28(8): 1020.
Doudoroff, P. 1976. Toxicity to fish of cyanides and related compounds. A review. Environmental Research Laboratory, Office of Research and Development, U.S. Environmental Protection Agency, Duluth, Minnesota (EPA-600/3-76-038).
Doudoroff, P. 1980. A critical review of recent literature on the toxicity of cyanides to fish. American Petroleum Institute, Washington, D.C.
Doudoroff, P. and M. Katz. 1953. Critical review of literature on the toxicity of industrial wastes and their components to fish. Sewage Ind. Wastes 25: 802-839 [cited in Evans and Frick, 2001].
Dowden, B.F. and H.J. Bennett. 1965. Toxicity of selected chemicals to certain animals. J. Water Pollut. Control Fed. 37: 1308-1316 [cited in Evans and Frick, 2001].
Drake, L. and B. Kirchner. 1987. Protecting remnant natural communities along rural roadsides. Nat. Areas J. 7(2): 79-80.
Dubey, S.K. and D.S. Holmes. 1995. Biological cyanide destruction mediated by microorganisms World J. Microbiol. Biotechnol. 11: 257.
Dvorak, P., M. Gunther, U. Zorn and A. Catsch. 1971. Metabolisches verhalten von kolloidalem ferrihexacyanoferrat (II). Naunyn-Schmiedebergs Arch. Pharmakol. 269: 48-56 [cited in Shifrin et al., 1996].
Eaton, L.J., J. Hoyle and A. King. No date. The impact of road salt on lowbush blueberries. Nova Scotia Agricultural College and Nova Scotia Department of Agriculture and Marketing, Truro, Nova Scotia.
Effenberger, E. 1964. Verunreinigunen eives grund-wasser durch cyanide. Arch. Hyg. Bakteriol. 148(4-5): 271-278 [cited in Chatwin, 1990].
Eilers, J.M. and A.R. Selle. 1991. Changes in the conductivity, alkalinity, calcium, and pH during a 50-year period in selected Wisconsin lakes. Can. J. Fish. Aquat. Sci. 46: 1929-1944.
Eilers, R.G., W.D. Eilers, W.W. Pettapiece and G. Lelyk. 1995. Salinization of soils. In: D.F. Acton and L.J. Gregorich (eds.), The health of our soils. Agriculture and Agri-Food Canada. pp. 77-86 (Publication 1906/E).
Eisler, R. 1991. Cyanide hazards to fish, wildlife, and invertebrates: A synoptic review. U.S. Fish and Wildlife Service (FWS Biol. Rep. 85(1.23); NTIS No. PB94-120144).
Environment Canada. 1995. Toxicity testing of National Contaminated Sites Remediation Program priority substances for the development of soil quality criteria for contaminated sites. Retesting Volatile Substances Guidelines Division, Evaluation and Interpretation Branch, Ecosystem Conservation Service.
Environment Canada. 1997a. Environmental assessments of Priority Substances under the Canadian Environment Protection Act. Guidance manual Version 1.0 - March 1997. Environmental Protection Series. Commercial Chemicals Evaluation Branch, Environment Canada (EPS/2/CC/3E).
Environment Canada. 1997b. Canadian Environmental Protection Act - Problem formulation for the environmental assessment of the Priority Substance road salts. Government of Canada.
Evans, M. and C. Frick. 2001. The effects of road salts in stream, lake and wetland ecosystems. National Water Research Institute, Saskatoon, Saskatchewan.
Eyles, N. and K.W.F. Howard. 1988. Urban landsliding caused by heavy rains: Geochemical identification of recharge waters along the Scarborough Bluffs, Toronto, Ontario. Can. Geotech. J. 25: 455-466.
Faber, J.H. and H.A. Verhoef. 1991. Functional differences between closely-related soil arthropods with respect to decomposition processes in the presence or absence of pine roots. Soil Biol. Biochem. 23(1): 15-23.
Fallon, R.D., D.A. Cooper, R. Speece and M. Henson. 1991. Anaerobic biodegradation of cyanide under methanogenic conditions. Appl. Environ. Microbiol. 57(6): 1656-1662.
Ferguson, K.D. 1985. The photolysis of iron-cyanide in a stream receiving effluent from a gold cyanidation mill. Environmental Protection Service, Pacific Region, Environment Canada.
Feth, J.H., C.E. Roberson and W.L. Polzer. 1964. Sources of mineral constituents in water from granitic rocks, Sierra Nevada, California and Nevada. U.S. Geological Survey, Washington, D.C. (Water Supply Papers No. 1535-I).
Foster, H.D. 1993. Road de-icing salt and cancer: the need for further study. J. Natl. Cancer Inst. 85(19): 1603-1605.
Franson, J.C., L. Sileo and W.J. Flemming. 1981. Iatrogenic salt poisoning in captive sandhill cranes. J. Am. Vet. Med. Assoc. 179(11): 1211-1213.
Fraser, D. and E.R. Thomas. 1982. Moose-vehicle accidents in Ontario: Relation to highway salt. Wildl. Soc. Bull. 10(3): 261-265.
Free, B.M. and G.G. Mulamoottil. 1983. The limnology of Lake Wabukayne, a storm-water impoundment. Water Res. Bull. 19(5): 821-827.
Freedman, B., J. Kerekes and G. Howell. 1989. Patterns of water chemistry among twenty-seven oligotrophic lakes in Kejimkujik National Park, Nova Scotia. Water Air Soil Pollut. 46: 119-130.
Fuller, W.H. 1984. Cyanides in the environment with particular attention to soil. In: D. Van Zyl (ed.), Cyanide and the environment. Vol. 1. Geochemical Engineering Program, Colorado State University, Fort Collins, Colorado. pp. 19-46.
Garrels, R.M. and F.T. Mackenzie. 1967. Origin of the chemical compositions of some springs and lakes. In: Equilibrium concepts in natural water systems. American Chemical Society, Washington, D.C. pp. 222-242 (Advances in Chemistry Series No. 67).
Gélinas, P. and J. Locat. 1988. Effets des sels déglaçants sur la qualité de l'eau de l'aquifère de Trois-Rivières Ouest. Ministère des transports du Québec (Report No. RTQ-87-05).
Gerber, R.E. and K.W.F. Howard. 1997. Ground-water to the Oak Ridges Moraine. In: N. Eyles (ed.), Environmental geology of urban areas. Geological Association of Canada, St. John's, Newfoundland. pp. 173-192.
Getz, L.L., F.R. Cole and D.L. Gates. 1978. Interstate roadsides as dispersal routes for Microtus pennsylvannicus. J. Mammal. 59: 208-212.
Gionfriddo, J.P. and L.B. Best. 1995. Grit use by house sparrows: Effects of diet and grit size. The Condor 97: 57-67.
Godbout, P. 1996. Chute à neige à l'égout motorisée et asservie - Une innovation québécoise unique en Amérique du Nord. Les consultants BGH - Planning, Inc. Symposium, Hôtel Le Méridien, November 19-21, 1996. Montréal, Quebec. 27 pp.
Gonzalez-Moreno, S., J. Gomez-Barrera, H. Perales and R. Moreno-Sanchez. 1997. Multiple effects of salinity on photosynthesis of the protist Euglena gracilis. Physiol. Plant. 101: 777-786.
Gooding, D. and A.J. Bodnarchuk. 1994. Field trials of prewetting salt and sand with MgCl2 and CaCl2 brines: Efficiency and effects. Highway Environment Branch, British Columbia Ministry of Transportation and Highways. 7 pp. (MoTH-ENV-94-01).
Gosh, A.K. and R.N. Pal. 1969. Toxicity of four therapeutic compounds to fry of Indian major carps. Fish. Technol. 6: 120-123 [cited in Hammer, 1977].
Gosz, J.R. 1977. Effects of ski area development and use in stream water quality on the Santa Fe Basin, New Mexico. For. Sci. 23(2): 167-179.
Gouvernement du Québec. 1991. Guide pour l'aménagement des lieux d'élimination des neiges usées. Ministère de l'Environnement du Québec. 95 pp.
Gouvernement du Québec. 1997. Gestion des neiges usées : Guide pour l'aménagement des lieux d'élimination de neige et mise en oeuvre du règlement sur les lieux d'élimination de neige. Ministère de l'Environnement du Québec. 56 pp.
Gouvernement du Québec. 2001. Guide d'aménagement des lieux d'élimination de neige et mise en oeuvre du Règlement dur les lieux d'élimination de neige. Ministère de l'environnement du Québec. Downloaded on June 13, 2001, from: www.menv.gouv.qc.ca/matieres/neiges_usees/ index.htm
Grenier, P. 1973. Moose killed on the highway in the Laurentides Park, Quebec, 1962 to 1972. Proc. N. Am. Moose Conf. Workshop 9: 155-193.
Hall, R., G. Hofstra and G.P. Lumis. 1972. Effects of deicing salt on eastern white pine: foliar injury, growth suppression, and seasonal changes in foliar concentrations of sodium and chloride. Can. J. For. Res. 2: 224-249.
Hamilton, R.W., J.K. Buttner and R.G. Brunetti. 1975. Lethal levels of sodium chloride and potassium chloride for an oligochaete, a chironomid midge, and a caddisfly of Lake Michigan. Environ. Entomol. 4: 1003-1006.
Hammer, U.T. 1977. The effects of alkali halides on fish and other aquatic fauna. In: The effects of alkali halides in the Canadian environment. Associate Committee on Scientific Criteria for Environmental Quality, National Research Council of Canada, Ottawa, Ontario. pp. 99-115 (NRCC Publication No. 15019).
Hammer, U.T. 1984. The saline lakes of Canada. In: F.B. Taub (ed.), Lakes and reservoirs. Elsevier Scientific Publishing Company, Amsterdam. pp. 521-540.
Hammer, U.T. 1986. Saline lake resources of the Canadian Prairies. Revue canadienne des ressources en eau 11(1): 43-57.
Hanes, R.E., L.W. Zelazny and R.E. Blaser. 1970. Effects of deicing salts on water quality and biota: Literature review and recommended research. Highway Research Board, National Academy of Sciences/National Academy of Engineering, Washington, D.C. 70 pp.
Harrington, J.A. and T. Meikle. 1994. Road salt effects on the germination of eight select prairie species. In: R.G. Wickett, P.D. Lewis, A. Woodliffe and P. Pratt (eds.), Proceedings of the 13th North American Prairie Conference, 1992. Department of Parks and Recreation, Windsor, Ontario. pp. 183-192.
Hart, W.C. 1985. Effects on the chemistry of water supply lakes due to chloride inputs. Centre for Water Resources Research, Technical University of Nova Scotia, Halifax, Nova Scotia. 16 pp. [cited in Briggins et al., 1989].
Hart, W.C. 1988. Chloride measured in the vicinity of Second Chain Lake, 1987-1988. Centre for Water Resources Research, Technical University of Nova Scotia, Halifax, Nova Scotia [cited in Briggins et al., 1989].
Hart, B.T., P. Bailey, R. Edwards, K. Hortle, K. James, A. McMahon, C. Meredith and K. Swadling. 1990. Effects of salinity on river, stream, and wetland ecosystems in Victoria, Australia. Water Res. 24: 1103-1107.
Hattori, R. and T. Hattori. 1980. Sensitivity to salts and organic compounds of soil bacteria isolated on diluted media. J. Gen. Microbiol. 26: 1-14.
Hawkins, R.H. and J.H. Judd. 1972. Water pollution as affected by street salting. Water Resour. Bull. 8: 1246-1252.
Headley, D. B. and N. Bassuk. 1991. Effect of time and application of sodium chloride in the dormant season on selected tree seedlings. J. Environ. Hortic. 9(3): 130-136.
Health Canada. 1996. Guidelines for Canadian drinking water quality. 6th edition. Ottawa, Ontario.
Henny, C.J., R.J. Hallock and E.F. Hill. 1994. Cyanide and migratory birds at gold mines in Nevada, USA. Ecotoxicology 3: 45-58.
Herlihy, A.T., J.L. Stoddard and C.B. Johnson. 1998. The relationship between stream chemistry and watershed cover data in the mid-Atlantic region, U.S. Water Air Soil Pollut. 105: 377-386.
Higgs, T.W. 1992. Technical guide for the environmental management of cyanide in mining. Prepared for Cyanide Subcommittee, British Columbia Technical and Research Committee on Reclamation, by T.W. Higgs Associates Ltd., Vancouver, British Columbia.
Hinton, M.J. and A.G. Eversole. 1978. Toxicity of ten commonly used chemicals to American eels. Agricultural Experiment Station, U.S. Department of Agriculture (Technical Contribution 1595).
Hoffman, R.W., C.R. Goldman, S. Paulson and G.R. Winters. 1981. Aquatic impacts of deicing salts in the central Sierra Nevada Mountains, California. Water Res. Bull. 17: 280-285.
Hofstra, G. and G.P. Lumis. 1975. Levels of deicing salt producing injury on apple trees. Can. J. Plant Sci. 55(1): 113-115.
Hofstra, G. and D.W. Smith. 1984. The effects of road deicing salt on the levels of ions in roadside soils in southern Ontario. J. Environ. Manage. 19: 261-271.
Hofstra, G., R. Hall and G.P. Lumis. 1979. Studies of salt-induced damage to roadside plants in Ontario. J. Arboric. 5: 25-31.
Holms, H.M. and J.L. Henry. 1982. Understanding salt-affected soils. Plant Industry Branch, Saskatchewan Agriculture. 14 pp.
Howard, K.W.F. and P.J. Beck. 1993.
Hydrogeochemical implications of groundwater contamination by road de-icing chemicals. J. Contam. Hydrol. 12: 245-268.
Howard, K.W.F. and J. Haynes. 1993. Groundwater contamination due to road de-icing chemicals - salt balance implications. Geosci. Can. 20(1): 1-8.
Howard, K.W.F. and J. Haynes. 1997. Contamination of urban groundwater by road de-icing chemicals. In: N. Eyles (ed.), Environmental geology of urban areas. Geological Association of Canada, St. John's, Newfoundland.
Howard, K.W.F. and L.C. Taylor. 1998.
Hydrogeochemistry of springs in urban Toronto, Canada. In: J. Van Brahana, Y. Eckstein, L.K. Ongley, R. Scheinder and J.E. Moore (eds.), Gambling with groundwater - physical, chemical, and biological aspects of aquifer-stream relations. American Institute of Hydrology, St. Paul, Minnesota. pp. 639-644.
Howard, K.W.F., J.I. Boyce, S.J. Livingstone and S.L. Salvatori. 1993. Road salt impacts on ground-water quality - the worst is still to come. GSA Today 3(12): 301-321.
Howard, P.H., R.S. Boethling, W.F. Jarvis, W.M. Meylan and E.M. Michalenko. 1991.
Handbook of environmental degradation rates. Lewis Publishers, New York, N.Y. pp. 132-133.
Howell, J. and R.C. Gumbrell. 1992. Salt poisoning in broiler chickens. N. Z. Vet. J. 40: 85.
Hsu, M. 1984. Anticaking agents in deicing salt. Materials and Research Division, Maine Department of Transportation. 18 pp. (Technical Paper No. 84-4).
Hubbs, A.H. and R. Boonstra. 1995. Study design to assess the effects of highway median barriers on wildlife. Research and Development Branch, Ontario Ministry of Transportation (MAT-94-03).
Huey, L.M. 1941. Mammalian invasion via the highway. J. Mammal. 22: 383-385.
Humphreys, D.J. 1978. A review of recent trends in animal poisoning. Br. Vet. J. 134: 128-141.
INTERA Consultants. 1996. Application guidelines for installation of oil/water separators at existing MTO patrol yards. Report submitted to Environmental Office, Ontario Ministry of Transportation, Toronto, Ontario.
Jeffries, D.S. 1997. 1997 Canadian acid rain assessment: The effects on Canada's lakes, rivers and wetlands. Vol. 3. Aquatic effects. Environment Canada, Ottawa, Ontario.
Johnson, G.D. and P.B. Kauss. 1991. Tributary contaminant inputs on the St. Clair and Detroit rivers and Lake St. Clair 1984-1985. Water Resources Branch, Ontario Ministry of the Environment, Toronto, Ontario.
Johnston, C.T., R.C. Sydor and C.L.S. Bourne. 2000. Impact of winter road salting on the hydrogeologic environment - an overview. Stantec Consulting Ltd., Kitchener, Ontario.
Jones, P.H., B.A. Jeffrey, P.K. Watler and H. Hutchon. 1986. Environmental impact of road salting - State of the art. Ontario Ministry of Transport and Communications (MTC No. RR237).
Jones, P.H., B.A. Jeffery, P.M. Watler and H. Hutchon. 1992. Environmental impact of road salting. In: F.M. D'Itri (ed.), Chemical deicers and the environment. Lewis Publishers, Chelsea, Michigan.
Judd, J.H. 1969. Lake stratification caused by runoff from street deicing, Milwaukee, Wisconsin. Center for Great Lakes Studies. 25 pp.
Judd, J.H. and J.W. Stegall. 1982. Reevaluation of the effects of deicing salt runoff on a small lake. National Technical Information Service, Springfield, Virginia. 28 pp. (A-114-MICH).
Kaplan, D.L., R. Hartenstein, E.F. Neuhauser and M.R. Malecki 1980. Physicochemical requirements in the environment of the earthworm (Eisenia foetida). Soil Biol. Biochem. 12: 347-352.
Keizer, P.D., D.C. Gordon, J.W. Rowell, R. McCurdy, D. Borgal, T.A. Clair, D. Taylor, J.D. Ogden and G.E.M. Hall. 1993. Synoptic water quality survey of Halifax/Dartmouth Metro Area lakes on April 16, 1991. Biological Services Branch, Scotia-Fundy Region, Department of Fisheries and Oceans.
Kelly, D., J. Underwood and J. Hansen. 1976. Influence of highway deicing salts upon water quality in Chocolate Lake, Halifax, Nova Scotia. Nova Scotia Department of the Environment, Halifax, Nova Scotia. 118 pp.
Kerekes, J.J. 1974. Limnological conditions in five small oligotrophic lakes in Terra Nova National Park, Newfoundland. J. Fish. Res. Board Can. 31: 555-583.
Kerekes, J. and B. Freedman. 1989. Seasonal variation of water chemistry in oligotrophic streams and rivers in Kejimkujik National Park, NS. Water Air Soil Pollut. 46(1-4): 131-144.
Kerekes, J., B. Freedman, S. Beauchamp and R. Tordon. 1989. Physical and chemical characteristics of three acidic oligotrophic lakes and their watersheds in Kejimkujik Park, Nova Scotia. Water Air Soil Pollut. 46: 99-117.
Kersey, K. and R.J. Mackay. 1981. Laboratory and field studies on the effects of road de-icing salt on stream invertebrates SIC-9. Snow and Ice Control Working Group, Institute for Environmental Studies, University of Toronto, Toronto, Ontario.
Khanna, C., H.J. Boermans and B. Wilcock. 1997. Fatal hypernatremia in a dog from salt ingestion. J. Am. Anim. Hosp. Assoc. 33: 113-117.
Klaassen, C.D. (ed.). 1996. Casarett and Doull's toxicology. The basic science of poisons. 5th edition. McGraw-Hill, New York, N.Y.
Kliejunas, J., M. Marosy and J. Pronos. 1989. Conifer damage and mortality associated with highway de-icing and snow removal in the Lake Tahoe area. Forest Pest Management, U.S. Department of Agriculture Forest Service. 18 pp. (Report No: 89-11).
Knowles, C.J. 1976. Microorganisms and cyanide. Bacteriol. Rev. 40: 652-680.
Knowles, C.J. and A.W. Bunch. 1986. Microbial cyanide metabolism. Adv. Microbial Physiol. 27: 73-111.
Kovacs, T.G. 1979. The effect of temperature on the cyanide toxicity to rainbow trout: I, Acute effects. II, Chronic effects. M.Sc. thesis, Concordia University, Montréal, Quebec. 69 pp. [cited in Leduc et al., 1982].
Kovacs, T.G. and G. Leduc. 1982. Sublethal toxicity of cyanide to rainbow trout (Salmo gairdneri) at different temperatures. Can. J. Fish. Aquat. Sci. 39: 1389-1395.
Kramer, J.R. 1964. Theoretical model for the chemical composition of fresh water with application to the Great Lakes. In: Proceedings of the 7th Conference on Great Lakes Research. Great Lakes Research Division, Institute of Science and Technology, University of Michigan, Ann Arbor, Michigan. pp. 147-160 (Publication No. 11).
Kszos, L.A., J.D. Winter and T.A. Storch. 1990. Toxicity of Chautauqua Lake bridge runoff to young-of-the-year sunfish (Lepomis macrochirus). Bull. Environ. Contam. Toxicol. 45: 923-930.
Labadia, C.F. and J.M. Buttle. 1996. Road salt accumulation in highway snow banks and transport through the unsaturated zone of the Oak Ridges Moraine, southern Ontario. Hydrol. Processes 10: 1575-1589.
Lacroix, G. and L. Bélanger. 2000. Suivi écologique du projet-pilote de gestion différenciée des emprises autoroutières: volet avifaune et mortalité par collision. Prepared for the Department of Biological Chemistry of the Université du Québec at Trois-Rivières and the Quebec Ministry of Transportation by the Canadian Wildlife Service, Environment Canada, Sainte-Foy, Quebec.
Laursen, K. 1981. Birds on roadside verges and the effect of mowing on frequency and distribution. Biol. Conserv. 20: 59-68.
Leduc, G., R.C. Pierce and I.R. McCracken. 1982. The effects of cyanides on aquatic organisms with emphasis on freshwater fishes. Associate Committee on Scientific Criteria for Environmental Quality, National Research Council of Canada, Ottawa, Ontario (NRCC No. 19246).
Lee, K. 1985. Earthworms: Their ecology and relationships with soils and land use. Academic Press, New York, N.Y.
Lefkovitch, L.P. and I. Fahrig. 1985. Spatial characteristics of habitat patches and population survival. Ecol. Model. 30: 297-308.
Leland, H.V. and S.V. Fend. 1998. Benthic invertebrate distributions in the San Joaquin River, California, in relation to physical and chemical factors. Can. J. Fish. Aquat. Sci. 55:1051-1067.
Letts, A. 1999. [The melting of salt applied to roads]. Personal communication from A. Letts (Technical Manager, Morton International Inc., Chicago, Illinois) in conversation with L. Brownlee (Canadian Wildlife Service, National Wildlife Research Centre, Ottawa).
Letts, A.D. 2000a. Effects of sodium ferrocyanide derived from road salting on the ecosystem. Morton Salt, Morton International Inc., Chicago, Illinois.
Letts, A.D. 2000b. A model to predict chloride concentrations in meltwater runoff from road salt. Morton Salt, Morton International Inc., Chicago, Illinois.
Lewis, S.A. 1991. The conservation and management of roadside vegetation in South Australia. In: D.A. Saunders and R.J. Hobbs (eds.), Nature conservation 2: The role of corridors. Beatty & Sons, Surrey. pp. 314-318.
Liang, W. 1998. Performance monitoring of the Rouge River stormwater management facility. Draft report. Stormwater Assessment Monitoring and Performance Program, Ontario Ministry of the Environment, Etobicoke, Ontario.
Lumis, G.P., G. Hofstra and R. Hall. 1973. Sensitivity of roadside trees and shrubs to aerial drift of deicing salt. Can. J. Plant Sci. 8(6): 475-477.
Lumis, G.P., G. Hofstra and R. Hall. 1976. Roadside woody plant susceptibility to sodium and chloride accumulation during winter and spring. Can. J. Plant Sci. 56: 853-859.
Lumis, G.P., G. Hofstra and R. Hall. 1977. Salt damage to roadside plants. Ontario Ministry of Agriculture and Food Factsheet. 3 pp. (Order No. 77-063 Dec. 1977).
Lumis, G.P., G. Hofstra and R. Hall. 1983. Salt damage to roadside plants. Ontario Ministry of Agriculture and Food Factsheet. 3 pp.
MacLeod, C.L., M.P. Borcsik and P.R. Jaffe. 1996. Effect of infiltrating solutions on the desorption of mercury from aquifer sediments. Environ. Technol. 17: 465-475.
Malmqvist, P.-A. 1985. Environmental effects of snow disposal in Sweden. Molndalsvagen, Göteborg, Sweden. 10 pp.
Maltby, L., D.M. Farrow, A.B.A. Boxall, P. Calow and C.I. Betton. 1995. The effects of motorway runoff on freshwater ecosystems: 1. Field study. Environ. Toxicol. Chem. 14: 1079-1092.
Martineau, D. and S. Lair. 1995. Nécropsie. Canadian Cooperative Wildlife Health Centre, Saint-Hyacinthe, Quebec (unpublished pathology report 95-1893).
Mattson, M.D. and P.J. Godfrey. 1994. Identification of road salts contamination using multiple regression and GIS. J. Environ. Manage. 18: 767-773.
Mayer, T. 2000. Unpublished data. National Water Research Institute, Burlington, Ontario.
Mayer, T., J. Marsalek and E. Delos Reyes. 1996. Nutrients and metal contaminants status of urban stormwater detention ponds. Lake Reserv. Manage. 12(3): 348-368.
Mayer, T., Q. Rochfort, J. Marsalek, M. Servos, A. Jurkovic and R. McKinnis. 1998. Effects of de-icing salts on the chemistry and toxicology of highway runoff. National Water Research Institute, Burlington, Ontario.
Mayer, T., W.J. Snodgrass and D. Morin. 1999. Spatial characterization of the occurrence of road salts and their environmental concentrations as chlorides in Canadian surface waters and benthic sediments. Water Qual. Res. J. Can. 34(4): 545-574. See also: Mayer, T., W.J. Snodgrass and D. Morin. 1999. Spatial characterization of the occurrence of road salts and their environmental concentrations as chlorides in Canadian surface waters and benthic sediments. Environment Canada. NWRI contribution No. 99-097 (revised 2001). 44 pp.
McBean, E. and S. Al-Nassri. 1987. Migration pattern of de-icing salts from roads. J. Environ. Manage. 25: 231-238.
McCormick, R.W. and D.C. Wolf. 1980. Effects of sodium chloride on CO2 evolution, ammonification and nitrification in a sassafras sandy loam. Soil Biol. Biochem. 12: 153-157.
McDonald, M.G. and A.W. Harbaugh. 1988. A modular three-dimensional finite-difference ground-water flow model. U.S. Geological Survey, Washington, D.C. 586 pp. (Techniques of Water Resources Investigations, Book 6).
McKee, J.E. and H.W. Wolf. 1963. Water quality criteria. 2nd edition. California State Water Resources Control Board. 542 pp.
McKillop, W.B., R.T. Patterson, L.D. Delorme and T. Nogrady. 1992. The origin, physico-chemistry and biotics of sodium chloride dominated saline waters on the western shore of Lake Winnipegosis, Man. Can. Field-Nat. 106(4): 454-473.
McLaughlin, D.L. and R.G. Pearson. 1981. Effectiveness of snow fence barriers in reducing salt spray injury to fruit orchards adjacent to the Queen Elizabeth Highway: 1978-1981. Phytotoxicology Section, Air Resources Branch, Ontario Ministry of the Environment, Brampton, Ontario.
McNeely Engineering. 1990. Snow disposal study for the designated growth centres. Report presented to the Transportation Department, Regional Municipality of Ottawa-Carleton, Ottawa, Ontario.
McNeely-Tunnock Ltd. 1995. Strategic plan for snow disposal inside the Greenbelt. Final report presented to the Regional Municipality of Ottawa-Carleton, Ottawa, Ontario.
MDOT (Michigan Department of Transportation). 1993. The use of selected de-icing materials on Michigan roads: Environmental and economic impacts. 105 pp.
Meade, G.M. 1942. Calcium chloride - a death lure for crossbills. Auk 59: 439-440.
Meeussen, J.C.L., M.G. Keizer and F.A.M. De Haan. 1992a. Chemical stability and decomposition rate of iron cyanide complexes in soil solutions. Environ. Sci. Technol. 26(3): 511-516.
Meeussen, J.C.L., M.G. Keizer, W.H. van Riemsdijk and F.A.M. de Haan. 1992b. Dissolution behavior of iron cyanide (prussian blue) in contaminated soils. Environ. Sci. Technol. 26(9): 1832-1838.
Melis, L.A., P.I. Tones and S.M. Swanson. 1987. Identification and evaluation of impacts resulting from the discharge of gold mill effluents. Report prepared for Environment Canada. 121 pp. (Project No. 52SS.KE145-6-095) [cited in Higgs, 1992].
Meteyer, C.U., R.R. Dubielzig, F.J. Dein, L.A. Baeten, M.K. Moore, J.R. Jehl, Jr. and K. Wesenberg. 1997. Sodium toxicity and pathology associated with exposure of waterfowl to hypersaline playa lakes of southeast New Mexico. J. Vet. Diagn. Invest. 9: 269-280.
Miller, B.K. and J.A. Litvaitis. 1992. Use of roadside salt licks by moose, Alces alces, in northern New Hampshire. Can. Field-Nat. 106: 112-117.
Miller, G.C., V.R. Hebert and W.W. Miller. 1989. Effect of sunlight on organic contaminants at the atmosphere-soil interface. Soil Science Society of America, Madison, Wisconsin. pp. 99-110. (SSSA Special Publication No. 22).
Mills, G.E. and A.V. Barker. 2001. Sodium accumulation in soils and plants along Massachusetts roadsides. In: Proceedings of the Annual Meeting of Northeast Region American Society for Horticultural Science (Abstract).
Mineau, P. and B.T. Collins. 1988. Avian mortality in agro-ecosystems 2. Methods of detection. In: M.P. Greaves, B.D. Smith and P.W. Greig-Smith (eds.), Field methods for the study of environmental effects of pesticides. British Crop Protection Council, Croydon, U.K. pp. 13-27.
Ministère des Transports du Québec. 1980. Prévention contre la pollution du Lac à la Truite en hiver. Document prepared by François Berard, Office des autoroutes du Québec. Document obtained from the Quebec Ministry of Transportation's document centre.
Ministère des Transports du Québec. 1999. Personal communication from G. D'Astous (Direction des Laurentides-Lanaudière) and A. Décréon (Service de l'aménagement des infrastructures et de l'environnement) to D. Morin (Environment Canada) regarding the impact of road salt loadings on lac à la Truite.
Ministers' Expert Advisory Panel. 1995. Report of the Ministers' Expert Advisory Panel on the second Priority Substances List under the Canadian Environmental Protection Act (CEPA). Government of Canada, Ottawa, Ontario. 26 pp.
Mitsch, W.J. and J.G. Gosselink. 1986. Wetlands. Van Nostrand Reinhold Company, New York, N.Y. 539 pp.
Molles, M.C. and J.R. Gosz. 1980. Effects of ski area on the water quality and invertebrates of a mountain stream. Water Air Soil Pollut. 14: 187-205.
Morin, D. 2000. Environmental impacts from the spreading and storage of road salts: Case studies. Supporting document for the road salts PSL assessment. Report submitted to the Environmental Resource Group for Road Salts, Commercial Chemicals Evaluation Branch, Environment Canada. May 2000. 84 pp.
Morin, D. and M. Perchanok. 2000. Road salt loadings in Canada. Supporting document for the road salts PSL assessment. Report submitted to the Environment Canada CEPA Priority Substances List Environmental Resource Group on Road Salts, May 2000. Commercial Chemicals Evaluation Branch, Environment Canada, Hull, Quebec. 85 pp.
Morin, D., W. Snodgrass, J. Brown and P.A. Arp. 2000. Impacts evaluation of road salt loadings on soils and surface waters. Supporting document for the road salts PSL assessment. Report submitted to the Environment Canada CEPA Priority Substances List Environmental Resource Group on Road Salts, June 2000. Commercial Chemicals Evaluation Branch, Environment Canada, Hull, Quebec. 76 pp.
Mortsch, L.D. and F.H. Quinn. 1996. Climate change scenarios for Great Lakes Basin ecosystem studies. Limnol. Oceanogr. 41: 903-911.
MTO (Ontario Ministry of Transportation). 1993-1999. Phase 1 and/or Phase 2 environmental site assessments done by DST Consulting Engineers, Oliver Mangione McCalla & Associates, NAR Environmental Consultants, Conestoga-Rovers & Associates or Ecoplans for the following patrol yards owned and operated by the Ontario Ministry of Transportation: Alexandria, Almonte, Apsley, Barriefield, Belleville Sub PY, Bloomfield, Bobcaygeon, Brighton, Calabogie, Camden East, Carlsbad Springs, Cloyne, Coboconk, Crystal Beach, Denbigh, Eganville, Erinsville, Grafton, Greely, Hawkesbury, Johnstown, Kemptville, Lansdowne, Lorneville, Madawaska, Madoc, Marysville, Morrisburg, Navan, Norwood, Pembroke, Roseneath, Sharbot Lake, Summerstown, Toledo, Tory Hill, Trenton Sub PY, Vankleek Hill, Verona, Winchester, Wolf Island, Youngs Point.
MTO (Ontario Ministry of Transportation). 1997. Graphical compilation of chloride concentrations in patrol yards. Managed by the Ontario Ministry of Transportation, Toronto, Ontario.
Mudder, T. 1991. Personal communication. Steffen Robertson and Kristen, Seattle, Washington [cited in Higgs, 1992].
Nadec (Nadec Experts & Consultants Inc.). 2001. The ecological significance of roadside habitat - Revised final draft. Presented to Commercial Chemicals Branch, Environment Canada, Hull, Quebec. March.
Natural Resources Canada. 1998. Mineral and metal commodity reviews - salt. In: Canadian minerals yearbook, 1998. Minerals and Metals Sector, Natural Resources Canada.
NB DOE and DOT (New Brunswick Department of Environment and Department of Transportation). 1978. An investigation of the quantity and quality of leachate from a highway salt treated sand pile under normal operating conditions. Prepared by the Water Resources Branch, New Brunswick Department of Environment, for and in cooperation with the New Brunswick Department of Transportation.
Neehall, R. 2000. Comments on the draft assessment report on salts, City of Edmonton, Office of the Environment, dated October 11, 2000.
Northcote, T.G. and T.G. Halsey. 1969. Seasonal changes in the limnology of some meromictic lakes in southern British Columbia. J. Fish. Res. Board Can. 26: 1763-1787.
Northover, J. 1987. NaCl injury to dormant roadside peach trees and its effect on the incidence of infections by Leucostoma spp. Phytopathology 77(6): 835-840.
Noss, R.F. 1983. A regional landscape approach to maintain diversity. Bioscience 33: 700-706.
Novotny, V., D. Muehring, D.W. Smith, D.H. Zitomer and R. Facey. 1997. Impact of nonpoint source snowmelt on receiving waters. In: Proceedings of the Water Environmental Federation 70th Annual Conference, October 19, Chicago, Illinois (Workshop #118).
OECD Scientific Expert Group. 1989. Curtailing usage of de-icing agents in winter maintenance. Organisation for Economic Cooperation and Development, Paris, France.
Oeser, R., Jr. 1977. The crossbill (Loxia curvirostra) - Highway mortalities in the Fichtelbers area. Beitr. Vogelkd. 23(5): 278-280.
Oetting, R.B. and J.F. Cassel. 1971. Waterfowl nesting on interstate highway right-of-way in North Dakota. J. Wildl. Manage. 35(4): 774-781.
Ohno, T. 1989. Determination of levels of free cyanide in surface and in ground waters affected by MDOT salt storage facilities. Technical Services Division, Maine Department of Transportation, Orono, Maine (Technical Report 86C:16-17).
Ohno, T. 1990. Levels of total cyanide and NaCl in surface waters adjacent to road salt storage facilities. Environ. Pollut. 67: 123-132.
OME (Ontario Ministry of the Environment). 1991. Stormwater quality best management practices. Environmental Sciences & Standards/Water Resources, Ontario Ministry of the Environment. June (ISBN 0-7729-8478-6).
OMEE (Ontario Ministry of Environment and Energy). 1993. A study of dust suppressants in Ontario. Final report. Prepared by CH2M Hill Engineering Ltd. for Waste Management Branch, Ontario Ministry of Environment and Energy, Toronto, Ontario.
Otake, T., S. Tone, I. Komasawa and M. Kawashima. 1982. Mechanism and kinetics of decomposition of water soluble cyanoferrates by ultraviolet radiation. Osaka University, Osaka, Japan [translation].
Parker, A. and J.D. Mather. 1979. An investigation into pollution from a disused gasworks site near Ladybank, Fife. Atomic Energy Research Establishment, Harwell, Berkshire. 19 pp. (AERE-R 9213) [cited in Chatwin, 1990].
Patrick, R., J.J. Cairns and A. Scheier. 1968. The relative sensitivity of diatoms, snails and fish to twenty common constituents of industrial wastes. Prog. Fish-Cult. 30: 137-140.
Paul, E.A. and F.E. Clark. 1996. Soil microbiology and biochemistry. 2nd edition. Academic Press, San Diego, California.
Paul, R., M. Rocher and R. Impens. 1984. [Influence of winter deicing with CaCl2 on mountain ash, maple, lime and plane.] Bull. Soc. R. Bot. Belg. 117: 277-284 (in French).
Péloquin, Y. 1993. Qualité des eaux de fonte provenant d'un site de surface pour l'élimination des neiges usées à la Ville de Laval. Université du Québec à Montréal, Montréal, Québec. 91 pp.
Perchanok, M.S., D.G. Manning and J.J. Armstrong. 1991. Highway deicers: Standards, practice and research in the Province of Ontario. Research and Development Branch, Ontario Ministry of Transportation, Downsview, Ontario. 53 pp. (MAT-91-13).
Perring, F.H., P.D. Sell, S.M. Walters and H.L.K. Whitehouse. 1964. A flora of Cambridgeshire. Cambridge University Press, Cambridge, U.K.
Phillips, A.M.J. 1944. The physiological effect of sodium chloride upon brook trout. Trans. Am. Fish. Soc. 74: 297-309.
Phippen, B.W., D.C. Parks, L.G. Swain, R. Nordin, C.J.P. McKean, J.B. Holms, P.D. Warrington, R. Nijman, J. Deniseger and L. Erickson. 1996. A ten year assessment of water quality in six acid-rain sensitive British Columbia lakes (1984-1994). British Columbia Ministry of Environment, Lands and Parks. 250 pp.
Pilon, P. and K.W.F. Howard. 1987. Origins of elevated chloride concentrations in groundwater systems. Ontario Ministry of the Environment Technology Transfer Conference No. 7, Toronto, December; Part C - Liquid and Solid Waste Research. pp. 376-398 (ISSN 0-825-4591).
Pinard, D., J.B. Sérodes and P.A. Côté. 1989. Charactérisation des eaux de fonte d'un dépot à neiges usées. Sci. Tech. Eau 22(3): 211-215.
Pringle, C.M., D.S. White, C.P. Rice and M.L. Tuchman. 1981. The biological effects of chloride and sulfate with the special emphasis on the Laurentian Great Lakes. University of Michigan, Ann Arbor, Michigan. pp. 1-51.
Prowse, C.W. 1987. The impact of urbanization on major ion flux through catchments: a case study in southern England. Water Air Soil Pollut. 32: 277-292.
Prud'homme, M. 2000. Potash. In: Canadian minerals yearbook, 1999. Minerals and Metals Sector, Natural Resources Canada. Downloaded on November 10, 2000, from www.nrcan.gc.ca/mms/cmy/1999CMY_E2.html.
Racette, D.J. and H.D. Griffin. 1989. Vegetation assessment surveys near the Ministry of Transportation's Longbow Lake Patrol Yard 1988-1989. Ontario Ministry of the Environment, Technical Support Section, N.W. Region. 9 pp.
Rader, W.S., L. Solujic, E.B. Milosavljevic and J.L. Hendrix. 1993. Sunlight-induced photochemistry of aqueous solutions of hexaferrocyanate (II) and (III) ions. Environ. Sci. Technol. 27(9): 1875-1879.
Rantamaki, J., L. Cerenius and K. Soderhall. 1992. Prevention of transmission of the crayfish plague fungus (Aphanomyces astaci) to the freshwater crayfish (Astacus astacus) by treatment with MgCl2. Aquaculture 104: 11-18.
Reznicek, A.A. 1980. Halophytes along a Michigan roadside with comments on the occurrence of halophytes in Michigan. Mich. Bot. 19: 23-30.
Reznicek, A.A., P.M. Catling and S.M. McKay. 1976. Carex praegracilis W. Boott, recently adventive in southern Ontario. Can. Field-Nat. 90: 180-183.
Rodgers, G.K. 1998. Winter stratification in Hamilton Harbour. National Water Research Institute, Burlington, Ontario (NWRI Contribution 98-231).
Rodgers, G.K. 1999. Personal communication regarding data in NWRI Contribution 98-231 [see Rodgers, 1998]. National Water Research Institute, Burlington, Ontario.
Rott, E., H.C. Duthie and E. Pipp. 1998. Monitoring organic pollution and eutrophication in the Grand River, Ontario, by means of diatoms. Can. J. Fish. Aquat. Sci. 55: 1443-1453.
Rowe, J.S. 1972. Forest regions of Canada. Canadian Forestry Service, Department of the Environment, Ottawa, Ontario. 172 pp.
Rushton, D. 1999. [Data regarding Bible Hill, Saulnierville and Oxford patrol yards]. Personal communication from D. Rushton (Environmental Services, Nova Scotia Department of Transportation and Public Works) to D. Morin (Chemicals Evaluation Division, Environment Canada). October and November, 1999.
Rushton, D. 2000. [Data regarding Bible Hill, Saulnierville and Oxford patrol yards]. Personal communication from D. Rushton (Environmental Services, Nova Scotia Department of Transportation and Public Works) to D. Morin (Chemicals Evaluation Division, Environment Canada). Winter, 2000.
Ruttner, F. 1963. Fundamentals of limnology. Translated by D.G. Frey and F.E.J. Fry. University of Toronto Press, Toronto, Ontario. 295 pp.
Salt Institute. 1964-1983. Survey of salt, calcium chloride, and abrasives use in the United States and Canada for 1964-65, 1966-67, 1969-70, 1973-74, 1978-79, 1982-83. Alexandria, Virginia.
Sandals, W.C.D. 1978. Acute salt poisoning in cattle. Can. Vet. J. 19: 136-137.
Saumure, R.A. 2001. Personal communication.
Shark Reef at Mandalay Bay, Las Vegas, Nevada.
Scanton, D. 1999. Personal communication (letter dated May 19, 1999). Ontario Ministry of Transportation, Toronto, Ontario.
Scarratt, W.K., T.J. Collins and D.P. Sponenberg. 1985. Water deprivation-sodium chloride intoxication in a group of feeder lambs. J. Am. Vet. Med. Assoc. 186(9): 977-978.
Schauffler, M. 1993. Capture of road-salt aerosols in an acidic peatland in central Maine. MA thesis, University of Maine. 41 pp.
Schelske, C. L., E. F. Stoermer, D. J. Conley, J. A. Robbins, and R. M. Glover. 1983. Early eutrophication in the lower Great Lakes: new evidence from biogenic silica in sediments. Science (Wash., DC) 222:320-322.
Schelske, C. L., D. J. Conley, and W. F. Warwick. 1985. Historical relationships between phosphorus loading and biogenic silica accumulation in Bay of Quinte sediments. Can. J. Fish. Aquat. Sci. 42:1401-1409.
Schindler, D.W., S.E. Bayley, B.R. Parker, K.G. Beaty, D.R. Cruikshank, E.J. Fee, E.U. Schindler and M.P. Stainton. 1996. The effects of climatic warming on the properties of boreal lakes and streams at the Experimental Lakes Area, northwestern Ontario. Limnol. Oceanogr. 41: 1004-1017.
Schmidt, J.W., L. Simovic and E. Shannon. 1981. Natural degradation of cyanide in gold mill effluents. Presented at Cyanide in Gold Mining Seminar, January 1981, Ottawa, Ontario [cited in Higgs, 1992].
Schraufnagel, F.H. 1965. Chlorides. Committee on Water Pollution, Wisconsin Public Health Service, Madison, Wisconsin. pp. 1-13 plus appendix.
Schroeder, J.E. and K.R. Solomon. 1998. Impact of road salts on an urban watershed. Poster presented at 19th Annual Meeting of the Society of Environmental Toxicology and Chemistry (SETAC), Charlotte, North Carolina.
Schulkin, J. 1991. Sodium hunger: The search for a salty taste. Cambridge University Press, Cambridge, U.K. pp. 458-499.
Scott, W.S. 1980. Road salt movement into two Toronto streams. J. Environ. Eng. Div. EE3: 547-560.
Scott, W.S. 1981. An analysis of factors influencing de-icing salt levels in streams. J. Environ. Manage. 13: 269-287.
Setälä, H. and V. Huhta. 1991. Soil fauna increase Betula pendula growth - Laboratory experiments with coniferous forest floor. Ecology 72(2): 665-671.
Setälä, H., J. Laakso, J. Mikola and V. Huhta. 1998. Functional diversity of decomposer organisms in relation to primary production. Appl. Soil Ecol. 9: 25-31.
Shifrin, N.S., B.D. Beck, T.D. Gauthier, S.D. Chapnick and G. Goodman. 1996. Chemistry, toxicology, and human health risk of cyanide compounds in soil at former manufactured gas plant sites. Regul. Toxicol. Pharmacol. 23: 106-116.
Short, T.M., J.A. Black and W.J. Birge. 1991. Ecology of a saline stream: community responses to spatial gradients of environmental conditions. Hydrobiologia 226: 167-178.
Simini, M. and I.A. Leone. 1986. The response of dormant Norway and sugar maples to simulated de-icing salt spray. J. Arboric. 12(1): 1-5.
Singer, S.N., C.K. Cheng and M.G. Scafe. 1997. The hydrogeology of southern Ontario. Vol. 1. Ontario Ministry of Environment and Energy, Toronto, Ontario. 80 pp.
Singleton, H.J. 1986. Water quality criteria for cyanide. Report and technical appendix. Water Management Division, British Columbia Ministry of Environment, Victoria, British Columbia. 84 pp.
Smith, L.L., Jr., S.J. Broderius, D.M. Oseid, G.L. Kimball, W.M. Koenst and D.T. Lind. 1979. Acute and chronic toxicity of HCN to fish and invertebrates. U.S. Environmental Protection Agency. 129 pp. (EPA-600/3-79-009).
Smith, M.E. and J.L. Kaster. 1983. Effect of rural highway runoff on stream benthic macroinvertebrates. Environ. Pollut. Ser. A 32: 157-170.
Smith, W.G. 1981. Observations on a large highway kill of Evening Grosbeaks in British Columbia. Syesis 14: 163.
Smol, J. P., S.R. Brown and R.N. McNeely. 1983. Cultural disturbances and trophic history of a small meromictic lake from central Canada. Hydrobiologia 103: 125-130.
Smolders, E. and M.J. McLaughlin. 1996. Chloride increases cadmium uptake in Swiss chard in a resin-buffered nutrient solution. Soil Sci. Soc. Am. J. 60: 1443-1447.
Snodgrass, W.J. and D. Morin. 2000. Patrol (maintenance/works) yards. Supporting document for the road salts PSL assessment. Report submitted to the Environment Canada CEPA Priority Substances List Environmental Resource Group on Road Salts, July 2000. Commercial Chemicals Evaluation Branch, Environment Canada, Hull, Quebec.
Soilcon Laboratories Ltd. 1995. Detailed study of de-icing salt in the Loon Lake environment. Draft prepared for Highway Environmental Branch, Ministry of Transportation and Highways, Victoria, British Columbia. 97 pp.
Sonzogni, W.C., W. Richardson, P. Rodgers and T.J. Monteith. 1983. Chloride pollution of the Great Lakes. J. Water Pollut. Control Fed. 55: 513-521.
Spehar, R.L. 1987. Memorandum to C. Stephan, U.S. Environmental Protection Agency, Duluth, Minnesota, June 24 [cited in U.S. EPA, 1988].
Sridhar, K.R. and F. Barlocher. 1997. Water chemistry and sporulation by aquatic hyphomycetes. Mycol. Res. 101(5): 591-596.
Stantec Consulting Ltd. 2000. Calculation of historical road densities within well field capture zones. Kitchener, Ontario. Steppuhn, H. and D. Curtin (eds.). 1992.
Salinity and sustainable agriculture. Swift Current Research Station, Research Branch, Agriculture Canada, Swift Current, Saskatchewan. 192 pp. (Prairie Salinity Publication No. 1).
Stoermer, E. F., J. A. Wolin, C. L. Schelske, and D. J. Conley. 1985. Postsettlement diatom succession in the Bay of Quinte, Lake Ontario. Can. J. Fish. Aquat. Sci. 42: 754-767.
Strobel, G.A. 1967. Cyanide utilization in soil.
Soil Sci. 103(4): 299-302 [cited in Chatwin, 1990].
Struger, J. 2000. Data on Cl concentrations in the outfall from water detention ponds. Data sent by e-mail on December 15, 2000.
Stumm, W. and J.J. Morgan. 1981. Aquatic chemistry. John Wiley & Sons, New York, N.Y. 780 pp.
Sucoff, E. 1975. Effect of deicing salts on woody vegetation along Minnesota Roads. Minn. Agric. Exp. Stn. Tech. Bull. 303, For. Ser. 20. 49 pp.
Sucoff, E. and S.G. Hong. 1976. Effect of NaCl on cold hardiness of Malus spp. and Syringa vulgaris. Can. J. Bot. 54: 2816-2819.
Sutcliffe, D.W. 1961. Studies on salt and water balance in caddis larvae (Trichoptera): II. Osmotic and ionic regulation of body fluids in Limnephilus stigma Curtis and Anabolia nervosa Leach. J. Exp. Biol. 38: 521-530.
Swayne, D.E., A. Shlosberg and R.B. Davis. 1986. Salt poisoning in turkey poults. Avian Dis. 39(4): 847-852.
TAC (Transportation Association of Canada). 1999. Salt management guide. Ottawa, Ontario.
Theis, T.L. and M.J. West. 1986. Effects of cyanide complexation on adsorption of trace metals at the surface of goethite. Environ.
Technol. Lett. 7: 309-318 [cited in Shifrin et al., 1996].
Thiel, R.P. 1980. Unusual vehicle caused mortality in red crossbills. Passenger Pigeon 42(1): 23-24.
Thirmurthi, D. and K.-H. Tan. 1978. Establishing baseline data for management of Halifax, N.S., lake watersheds. J. Am. Water Works Assoc. 70(8): 436-445.
Thompson, P. 2000. Salt damage to trees along roads in the Regional Municipality of Peel. Report for Regional Municipality of Peel, July 10, 2000.
Thorton, K.W. and J.R. Sauer. 1972. Physiological effects of NaCl on Chironomus attenuatus (Diptera: Chironomidae). Ann. Entomol. Soc. Am. 65: 872-875.
Thuet, J.H. 1977. Environment. In: Economic impact of highway snow and ice control: state of the art. Federal Highway Administration, Washington, D.C. pp. 146-168, 231-247 (Report No. FHWA-RD-77-20).
Topping, M.S. and G.G.E. Scudder. 1977. Some physical and chemical features of saline lakes in central British Columbia. Syesis 10: 145-166.
Toronto and Region Conservation Authority. 1998. 1990 to 1996 Water Quality Data from the Toronto RAP Watershed; based on data from the Ontario Ministry of the Environment's Provincial Water Quality Monitoring Network. Toronto, Ontario.
Towill, L.E., J.S. Drury, B.L. Whitfield, E.B. Lewis, E.L. Galyan and A.S. Hammons. 1978. Reviews of the environmental effects of pollutants: V. Cyanide. 190 pp. (ORNL/EIS-81; EPA-600/1-78-027).
Townsend, A.M. and W.F. Kwolek, 1987. Relative susceptibility of thirteen pine species to sodium chloride spray. J. Arboric. 13(9): 225-228.
Trainer, D.O. and L. Karstad. 1960. Salt poisoning in Wisconsin wildlife. J. Am. Vet. Med. Assoc. 13: 614-617.
Trama, F.B. 1954. The acute toxicity of some common salts of sodium, potassium, and calcium to the common bluegill (Lepomis macrochirus). Proc. Acad. Nat. Sci. Philadelphia 196: 1885 [cited in McKee and Wolf, 1963].
Transport Canada. 2001. Length of public roads. Table presented on website www.tc.gc.ca/pol/en/ExcelSpreadsheets2/main.asp on May 28, 2001.
Trueman K.F. and D.C. Clague. 1978. Sodium chloride poisoning in cattle. Aust. Vet. J. 54: 89-91.
Tuchman, M.L., E.F. Stoermer and H.J. Carney. 1984. Effects of increased salinity on the diatom assemblage in Fonda Lake, Michigan. Hydrobiologia 109: 179-188.
Underwood, J.K., J.G. Ogden and D.L. Smith. 1986. Contemporary chemistry of Nova Scotian lakes. Water Air Soil Pollut. 30: 991-999.
Upchurch, R.G. and G.H. Elkan. 1977. Comparison of colony morphology, salt tolerance, and effectiveness in Rhizobium japonicum. Can. J. Microbiol. 23: 1118-1122.
U.S. EPA (United States Environmental Protection Agency). 1971. Water quality research. Environmental impact of highway deicing. Edison Water Quality Laboratory, Edison, New Jersey (NJ 11040 GKK).
U.S. EPA (United States Environmental Protection Agency). 1973. Water quality criteria. 1972. National Academy of Science and National Academy of Engineering, Washington, D.C. (EPA-R3-73-03) [cited in Singleton, 1986].
U.S. EPA (United States Environmental Protection Agency). 1985. Ambient water quality criteria for cyanide, 1984. Criteria and Standards Division, U.S. Environmental Protection Agency, Washington, D.C.
(EPA/440/5-84-028).
U.S. EPA (United States Environmental Protection Agency). 1988. Ambient water quality criteria for chloride.
U.S. Environmental Protection Agency, Washington, D.C.
Van Barneveld, J.W. and R.H. Louie. 1990. Boitanio Park, causes of tree decline, soil and vegetation analysis. Working report. British Columbia Ministry of Environment, Victoria, British Columbia. 34 pp. plus appendices.
Vickers, S. 2000. Metal accumulation and sediment-water exchange in an urban wet detention pond. M.Sc. thesis, Geography Department, University of Toronto, Toronto, Ontario.
Wages, D.P., M.D. Ficken, M.E. Cook and J. Mitchell. 1995. Salt toxicosis in commercial turkeys. Avian Dis. 39: 158-161.
Wallen, I.E., W.C. Greer and R. Lasater. 1957. Toxicity to Gambusia affinis of certain pure chemicals in turbid waters. Environ. Future 29: 695 [cited in McKee and Wolf, 1963].
Waller, D.L., S.W. Fisher and H. Dabrowska. 1996. Prevention of zebra mussel infestation and dispersal during aquaculture operations. Prog. Fish Cult. 58(2): 77-84.
Wang, J.S., P.M. Huang, W.K. Liaw and U.T. Hammer. 1991. Kinetics of the desorption of mercury from selected freshwater sediments as influenced by chloride. Water Air Soil Pollut. 56: 533-542.
Warren, L.A. and A.P. Zimmerman. 1994. The influence of temperature and NaCl on cadmium, copper and zinc partitioning among suspended particulate and dissolved phases in an urban river. Water Res. 28(9): 1921.
Warrington, P. and C. Phelan. 1998. Road salts and winter maintenance - a best management practices guide to protecting water quality. Water Management Section, British Columbia Ministry of Environment, Lands and Parks, Victoria, British Columbia.
Watson T. 2000. [Data on chloride concentrations in ponds adjacent to roadways in Southern Ontario]. Personal communication from T. Watson (University of Toronto, Dept. of Biology) to D. Morin (Chemicals Evaluation Division, Environment Canada). Communication between October, 2000, and January, 2001.
Way, J.L., P. Leung, E. Cannon, R. Morgan, C. Tamulinas, J. Leong-Way, L. Baxter, A. Nagi and C. Chui. 1988. The mechanism of cyanide intoxication and its antagonism. In: Proceedings of the Symposium on Cyanide Compounds in Biology, March 15-17. Ciba Foundation, London, U.K. pp. 233-243.
Way, J.M. 1977. Roadside verges and conservation in Britain: a review. Biol. Conserv. 12: 65-74.
Weast, R.C., D.R. Lide, M.J. Astle and W. Beyer (eds.). 1989. CRC handbook of chemistry and physics, 1989-1990. 70th ed. CRC Press, Boca Raton, Florida.
Weiler, R.R. and V.K. Chawla. 1969. Dissolved mineral quality of Great Lakes waters. In: Proceedings of the 12th Conference on Great Lakes Research. Vol. 1. International Association for Great Lakes Research, Ann Arbor, Michigan. Braun-Brumfield, Inc., Ann Arbor, Michigan. pp. 801-818.
Werkhoven, C.H.E., P.J. Salisbury, and W.H. Cram. 1966. Germination and survival of Colorado spruce, Scots pine, caragana, and Siberian elm at four salinity and two moisture levels. Can. J. Plant Sci. 46(1): 1-7.
Wetzel, R.G. 1975. Limnology. W.B. Saunders Co., Philadelphia, Pennsylvania. 743 pp.
Wetzel, R.G. 1983. Limnology. 2nd edition. Saunders College Publishing, Toronto, Ontario.
Whitfield, P.H. and N.L. Wade. 1992. Monitoring transient water quality events electronically.
Water Resour. Bull. 28(4): 703-711.
Wickstrom, M.L., P. Mineau, L.J. Brownlee and T.K. Bollinger. 2001. Toxicity of road salt to house sparrows. Draft report. University of Saskatchewan, Saskatoon, Saskatchewan.
Wilcox, B.A. and D.D. Murphy. 1985 Conservation strategy: the effects of fragmentation on extinction. Am. Nat. 125: 879-887.
Wilcox, D.A. 1982. The effects of deicing salts on water chemistry and vegetation in Pinhook Bog, Indiana Dunes National Lakeshore. Ph.D. dissertation, Purdue University, West Lafayette, Indiana. 134 pp.
Wilcox, D.A. 1984. The effects of NaCl deicing salt on Sphagnum recurvum P. Beauv. Environ. Exp. Bot. 24(4): 295-304.
Wilcox, D.A. 1986a. The effects of deicing salts on vegetation in Pinhook Bog, Indiana. Can. J. Bot. 64(4): 865-874.
Wilcox, D.A. 1986b. The effects of deicing salt on water chemistry in Pinhook Bog, Indiana. Water Resour. Bull. 22(1): 57-64.
Wilcox, D.A. and R.E. Andrus. 1987. The role of Sphagnum fimbriatum in secondary succession in a road salt impacted bog. Can. J. Bot. 65: 2270-2275.
Williams, D.D., N.E. Williams and Y. Cao. 1997. Spatial differences in macroinvertebrate community structure in springs in southeastern Ontario in relation to their chemical and physical environment. Can. J. Zool. 75: 1404-1414.
Williams, D.D., N.E. Williams and Y. Cao. 1999. Road salt contamination of groundwater in a major metropolitan area and development of a biological index to monitor its impact. Water Res. 34(1): 127-138.
Williams, D.J., K.W. Kuntz, S. L'Italien and V. Richardson. 1998. Lake Ontario Surveillance Program. Spatial and temporal trends of selected parameters, with emphasis on 1992-93 results. Environmental Conservation Branch/Ontario Region, Ecosystem Health Division, Environment Canada. 24 pp. (Report No. EHD/ECB-OR/98-04/l).
Williams, W.D. 1987. Salinity as a determinant of salt lake fauna: a question of scale. Hydrobiologia 210: 151-160.
Windingstad, R.M., F.X. Kartch, R.K. Stroud and M.R. Smith. 1987. Salt toxicosis in waterfowl in North Dakota. J. Wildl. Dis. 23(3): 443-446.
WI SLOH (Wisconsin State Laboratory of Hygiene). 1995. Unpublished data on chloride toxicity to aquatic species. From A. Letts (Technical Manager, Morton International Inc. Chicago, Illinois) to M.S. Evans (National Hydrology Research Institue, Environment Canada), August 11, 1998.
Wolfe, D.F.G. 1996. Opportunistic winter water acquisition by pine grosbeaks. Wilson Bull. 108(1): 186-187.
Wood, J.A. and C.D.A. Rubec. 1989. Chemical characterization of several wetlands in Kejimkujik National Park, Nova Scotia. Water Air Soil Pollut. 46: 177-186.
Woodliffe, P.A. 2001. Personal communication. Ontario Ministry of Natural Resources-Aylmer District, Chatham, Ontario.
Woods, J. 1998. [The recurrent kills of pine siskins witnessed by local inhabitants in Revelstoke National Park]. Personal communication from John Woods (Parks Canada, Mount Revelstoke & Glacier National Parks, Revelstoke, British Columbia) with P. Mineau and G. Fox (Canadian Wildlife Service, National Wildlife Research Centre, Ottawa). Meeting, December 4, 1998.
Wulkowicz, G.M. and Z.A. Saleem. 1974. Chloride balance of an urban basin in the Chicago area. Water Resour. Res. 10(5): 974-982.
Zeeb, B.A. and J.P. Smol. 1991. Paleolimnological investigation of the effects of road salt seepage on scaled chrysophytes in Fonda Lake, Michigan. J. Paleolimnol. 5: 263-266.
Zheng, C. 1990. MT3D: A modular three-dimensional transport model for simulation of advection, dispersion and chemical reactions of contaminant in groundwater systems. Report to the U.S. Environmental Protection Agency, Ada, Oklahoma. 170 pp.
Zidar, L. 2000. Public comment. Salt damage to trees in North District of the Region of Peel. Letter to J. Buccini, dated October 11, 2000. System Operations, Region of Peel, Brampton, Ontario.
Appendix A Search Strategies Employed for Identification of Relevant Data
Data relevant to the assessment of whether road salts are "toxic" to the environment under CEPA 1999 were identified from existing review documents, published reference texts and on-line searches of the following databases, conducted between January and May 1996: Aqualine (1990-1996), ASFA (Aquatic Sciences and Fisheries Abstracts, Cambridge Scientific Abstracts; 1996), BIOSIS (Biosciences Information Services; 1990-1996), CAB (Commonwealth Agriculture Bureaux; 1990-1996), CESARS (Chemical Evaluation Search and Retrieval System, Ontario Ministry of the Environment and Michigan Department of Natural Resources; 1996), Chemical Abstracts (Chemical Abstracts Service, Columbus, Ohio; 1990-1996), CHRIS (Chemical Hazard Release Information System; 1964-1985), Current Contents (Institute for Scientific Information; 1990-1992, 1996), ELIAS (Environmental Library Integrated Automated System, Environment Canada library; January 1996), Enviroline (R.R. Bowker Publishing Co.; November 1995 - June 1996), Environmental Abstracts (1975 - February 1996), Environmental Bibliography (Environmental Studies Institute, International Academy at Santa Barbara; 1990-1996), GEOREF (Geo Reference Information System, American Geological Institute; 1990-1996), HSDB (Hazardous Substances Data Bank, U.S. National Library of Medicine; 1990-1996), Life Sciences (Cambridge Scientific Abstracts; 1990-1996), NTIS (National Technical Information Service, U.S. Department of Commerce; 1990-1996), Pollution Abstracts (Cambridge Scientific Abstracts, U.S. National Library of Medicine; 1990-1996), POLTOX (Cambridge Scientific Abstracts, U.S. National Library of Medicine; 1990-1995), RTECS (Registry of Toxic Effects of Chemical Substances, U.S. National Institute for Occupational Safety and Health; 1996), Toxline (U.S. National Library of Medicine; 1990-1996), TRI93 (Toxic Chemical Release Inventory, U.S. Environmental Protection Agency, Office of Toxic Substances; 1993), U.S. EPA-ASTER (Assessment Tools for the Evaluation of Risk, U.S. Environmental Protection Agency; up to December 21, 1994), WASTEINFO (Waste Management Information Bureau of the American Energy Agency; 1973 - September 1995) and Water Resources Abstracts (Geological Survey, U.S. Department of the Interior; 1990-1996). Reveal Alert was used to maintain an ongoing record of the current scientific literature pertaining to the potential environmental effects of road salts.
In addition, the Transportation Association of Canada has reviewed information on sodium/calcium road salt as part of its Road Salt Management Initiative (TAC, 1999).
Data searches were done by individual working groups preparing separate components of the supporting documentation. Search strategies or data sources are provided in individual reports.
Data obtained after May 2001 were not considered in this assessment.
Appendix B Supporting Documents prepared for the PSL Assessment of Road Salts
Brownlee, L., P. Mineau and A. Baril. 2000. Risk characterization for wildlife. Report submitted to the Environment Canada CEPA Priority Substances List Environmental Resource Group on Road Salts, May 2000. Commercial Chemicals Evaluation Branch, Environment Canada, Hull, Quebec.
Butler, B.J. and J. Addison. 2000. Biological effects of road salts on soils. Report submitted to the Environment Canada CEPA Priority Substances List Environmental Resource Group on Road Salts, March 2000. Commercial Chemicals Evaluation Branch, Environment Canada, Hull, Quebec.
Cain, N.P., B. Hale, E. Berkelaar and D. Morin. 2001. Critical review of effects of NaCl and other road salts on terrestrial vegetation in Canada. Report submitted to the Environment Canada CEPA Priority Substances List Environmental Resource Group on Road Salts. July 2001. Existing Substances Branch, Environment Canada, Hull, Quebec.
Delisle, C.E. and L. Dériger. 2000. Caracterisation et élimination des neiges usées: impacts sur l'environnement. Report submitted to the Environment Canada CEPA Priority Substances List Environmental Resource Group on Road Salts. Commercial Chemicals Evaluation Branch, Environment Canada, Hull, Quebec.
Environment Canada. 1997. Canadian Environmental Protection Act - Problem formulation for the environmental assessment of the Priority Substance road salts. Government of Canada.
Evans, M. and C. Frick. 2001. The effects of road salts aquatic ecosystems. National Water Research Institute, Saskatoon, Saskatchewan.Report submitted to the Environment Canada CEPA Priority Substances List Environmental Resource Group on Road Salts. August 2001. Existing Substances Branch, Environment Canada, Hull, Quebec.
Johnston, C.T., R.C. Sydor and C.L.S. Bourne. 2000. Impact of winter road salting on the hydrogeologic environment - an overview. Stantec Consulting Ltd., Kitchener, Ontario. Report submitted to the Environment Canada CEPA Priority Substances List Environmental Resource Group on Road Salts, May 2000. Commercial Chemicals Evaluation Branch, Environment Canada, Hull, Quebec.
Letts, A.D. 2000. Effects of sodium ferrocyanide derived from road salting on the ecosystem. Morton Salt, Morton International Inc., Chicago, Illinois. Report submitted to the Environment Canada CEPA Priority Substances List Environmental Resource Group on Road Salts, May 2000. Commercial Chemicals Evaluation Branch, Environment Canada, Hull, Quebec.
Mayer, T., W.J. Snodgrass and D. Morin. 1999b. Spatial characterization of the occurrence of road salts and their environmental concentrations as chlorides in Canadian surface waters and benthic sediments. Environment Canada. NWRI contribution No. 99-097 (revised 2001). 44 pp.
Morin, D. 2000. Environmental impacts from the spreading and storage of road salts: Case studies. Supporting document for the road salts PSL assessment. Report submitted to the Environmental Resource Group for Road Salts, Commercial Chemicals Evaluation Branch, Environment Canada. May 2000. 84 pp.
Morin, D. and M. Perchanok. 2000. Road salt loadings in Canada. Supporting document for the road salts PSL assessment. Report submitted to the Environment Canada CEPA Priority Substances List Environmental Resource Group on Road Salts, May 2000. Commercial Chemicals Evaluation Branch, Environment Canada, Hull, Quebec. 85 pp.
Morin, D., W. Snodgrass, J. Brown and P.A. Arp. 2000. Impacts evaluation of road salt loadings on soils and surface waters. Supporting document for the road salts PSL assessment. Report submitted to the Environment Canada CEPA Priority Substances List Environmental Resource Group on Road Salts, June 2000. Commercial Chemicals Evaluation Branch, Environment Canada, Hull, Quebec. 76 pp.
Snodgrass, W.J. and D. Morin. 2000. Patrol (maintenance/works) yards. Supporting document for the road salts PSL assessment. Report submitted to the Environment Canada CEPA Priority Substances List Environmental Resource Group on Road Salts, July 2000. Commercial Chemicals Evaluation Branch, Environment Canada, Hull, Quebec.